Design and Molecular Docking of Antioxidant Lead Compound and its Analogues Acting as Human Tyrosine Kinase Inhibitors
|
|
|
- Augustine Collins
- 6 years ago
- Views:
Transcription
1 IR Journal of Pharmacy and Biological ciences (IR-JPB) e-i: Volume 5, Issue 4 (Mar. Apr. 2013), PP Design and Molecular Docking of Antioxidant Lead Compound and its Analogues Acting as uman Tyrosine Kinase Inhibitors Ammara Khalid 1, aimakalsoom 2, aveedariaz *1 1 (Department of Microbiology and Molecular Genetics, University of the Punjab, Pakistan) 1 (Department of Bioinformatics and Biotechnology, International Islamic University, Islamabad, Pakistan) 2 (Department of Chemistry, Quaid-i-Azam University, Islamabad, Pakistan) Abstract:Cancer is the 2 nd leading cause of death in humans. Proteine.g. tyrosine kinaseleads to cancer when become constitutively active due to mutation. o there is a need to develop tyrosine kinase inhibitors to avoid cancer.in the present study the problem of designing an anticancer drug with more efficacies was solved by using computer aided drug designing (CADD). Antioxidants were used as tyrosine kinase inhibitors. Molecular docking studies of antioxidants belonging from different classes were carried out with human target protein having pdbid:2ck in order to find out the most active antioxidant drug having high inhibitory activity in cancer.docking of 36 selected ligands, comprised of active drugs for cancer and compounds having anticancer activity, was done with the active site of protein 2CK and a lead compound was recognized on the basis of hydrogen bonding, hydrophobic and ionic interactions. Three analogues of the lead compound were designed to enhance its activity against cancer. Analogues were docked with protein 2CK and their interactions showed that they could possess good inhibitory activity against cancer with suitable drug-like properties as compared to other active drugs for cancer and therefore could be recommended for further studies. Keywords: Antioxidants, Tyrosine Kinase inhibitors, lead compound, Cancer, Molecular docking I. Introduction Antioxidants are the bioactive chemical compounds which are highly important to fight against oxidative stress which is responsible for various diseases including cancer. xidative stress produces due to the accumulation of free radicals. Free radicals interfere with different process in the body by destroying proteins, lipids and DA sugars etc[1]. Antioxidant fight against oxidative stress by neutralizing the effect of free radicals produce during the process of respiration as a by-product of oxygen metabolism and thus can play important role in preventing many life threatening diseases. Antioxidant helps in neutralizing the free radicals before they do harm to our body antioxidant system includes different enzymes such as superoxide dismutase, glutathione reductase, glutathioneperoxidase, -methyl transferase and catalase. mall molecule antioxidants such as ascorbate, glutathione, tocopherol, carotenoids, polyphenols, alkaloids and some other compounds are also important cellular antioxidants [2]. Tyrosine kinases are a family of proteins involved in the transduction of growth factor signals to the nucleus and are located in or near the cell membrane.tyrosine kinases catalyze the phosphorylation of tyrosine residues in proteins. The phosphorylation of tyrosine residues then causes a change in the function of the protein in which they are present in. Due to mutations some tyrosine kinases become constitutively active and nonstop functional state which can result in cancer[3]. Querecetin is an antioxidant compound also used as tyrosine kinase inhibitor. But quercetin appears to be associated with side effects when administered orally and intravenously[4]for example, it may cause kidney failure, gastrointestinal complaints, allergic skin reactions, and increase the risk of kernicterus (brain damage due to severe jaundice) in newborns,mouth sores, headaches, and digestive upset are also the possible side effects of taking quercetin[5]. There is a need to develop more antioxidant drugs which are more specific and significantly fewer side effects, can bind with tyrosine kinase, and inhibits its hyper activity which is one of the reasons for cancer formation. The studies have been carried out in order to identify effective, selective and efficient anticancerous antioxidant Lead compound and its analogues. In short, this work reveals that the current bioinformatics tools used strategically can lead to the discovery of potential Lead compounds and their analogues that can inhibit the process of cancer formation. In current study molecular docking has been carried out on different antioxidants with the aim of identifying their anticancer activity in uman tyrosine kinase binding and inhibition. ChemDraw [6], AutodockVina[7] and visual molecular dynamics (VMD) [8] were used for studying molecular docking and ligand protein interactions, respectively. 75 Page
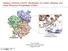
2 C Design and Molecular Docking of Antioxidant Lead compound and its Analogues Acting as uman II. Methodology 1.1. Target selection Identifying and selecting the most appropriate drug target or receptor is the key step in drug designing.bioinformatics tools can be used to identify the required target. For a drug target to be ideal it must be specifically linked to the human disease. Inpresent study protein having pdb id: 2hck shown in Fig.1 is used. Figure 1.Crystal structure of hematopoietic cell kinase (CK) quercetin complex Ligand selection Various antioxidant tyrosine kinase inhibitors have been identified and selected in this study. The data set consisted of 36 antioxidant ligands(a1-a36) have drug like properties against cancer.ligands were drawn by using ChemDraw. All the ligands were selected on the bases of clearance from 2,2-Diphenyl-1-Picrylhydrazyl (DPP) bioassayand the IC50 of all the ligands should lie under 100 μm. tructures and IC50 values of these 36 compounds along with the standard compound Quercetin (A11) are shown in Table 1 [1, 2, 9-14]. Table 1. Chemical structures and IC50 values of selected compounds o. tructure IC 50 o. tructure IC 50 o. tructure IC 50 (µm) (µm) (µm) C(C A1 C 3 12 A2 3) 2 C(C 22 A3 3) 3 3C 22 3C 3C A A A A A A Me Me Me Me A A A C A A A C 3 A16 3C C 2C 6 5 C 2C A17 CK 76.5 A18 CK 168 3C C A19 2CCC 3 C 26 A20 2CCC 3 C 30 A Cl 76 Page
3 2 Design and Molecular Docking of Antioxidant Lead compound and its Analogues Acting as uman C A A A24 ( 3C 2C 2C)( 3C)C C C 3 C(C 3)(C 2C 2C 3) A A A ( 3C 2C 2C)( 3C)C tbu C(C 3)(C 2C 2C 3) tbu C 3 3C C 3 A A29 3C 70 A30 46 C 3 C 3 3C C 3 C 3 3C 3C C 3C C 3 3 A A32 70 A C 3 3C A A35 C C 3 3 C A C C 3 3C C 3 C 3 C Molecular docking In molecular docking the ligand and the target protein are two important ingredients. In order to perform docking a specific target protein was chosen for antioxidant compounds. Tyrosine kinase (pdb id: 2CK) was identified as protein target for current studies. Pdb file 2CK was obtained from the protein data bank (rscb.org).autodockvina was used for docking in this study. AutodockVina is the latest version of Autodock it provides speed and accuracy and it accepts the ligand and the target protein in pdb format. The pdb files of the macromolecule 2CK and ligand were placed in a newly made folder. The pdb file of the macromolecule (protein) was given as an input to the software. Protein was colored after that polar hydrogen s were added to it. This macromolecule was then saved as grid output file with.pdbqt extension. Then in grid box different values were set according to the active site of protein. After this ligand pdb file was taken as an input and its torsions were calculated this ligand file was also saved with.pdbqt extension. Another file of.txt extension was made in which receptor and ligand names and there location of binding at x y and z axis with each other were set. The.txt file was used in command prompt as an input file and command prompt itself generates an output log file with.txt extension according to the given input file. After generating log.txt file different docking conformations were obtained. The conformation having lowest energy of all the compounds was chosen to find out the interactions with target protein. This previously mentioned procedure of docking was done on all compounds. The ligand protein interactions were predicted using Visual Molecular Dynamics (VMD). The most active antioxidant tyrosine kinase inhibitor was identified after the detailed analysis of ligand-protein interactions. Binding interactions of all compounds were observed and then the compound showing best interactions among all was selected as Lead. Lead designing is the most crucial step in drug designing and discovery Analogue designing After the identification of Lead compound three structural analogues of the Lead were made by introduction or elimination of various functional groups in the Lead compound. Docking was performed on these analogues and their interactions with protein were identified. 77 Page
4 Design and Molecular Docking of Antioxidant Lead compound and its Analogues Acting as uman III. Results 1.5. Molecular docking The binding interactions of the actively docked conformations of the ligands and the target protein have been identified and marked using VMD. Checking one by one all amino acids within 5 A of the active site of the target protein, the important binding interactions were identified. The identified interactions in all 36 compounds including the standard one include the hydrophobic interactions and hydrogen bonding. The binding interactions of all ligands have been analyzed. Five compounds have been identified as the most active from the set of given ligands A7, A12, A15, A25 and A33. These ligands have shown strong hydrogen bonding and hydrophobic interactions with the target protein than the rest of the ligands. Along with their strong interactions, the IC50 values of them are lower which is the positive sign towards their being the active ones. These three active ligands along with their interactions and IC50 values are shown in Table 2. From the table it is clear that the number of hydrogen bonds in ligands A7, A12, A15, A25 and A32 are 8,4,6,5 and 4 respectively, and the hydrophobic interactions are 2,1,2,2 and 2 respectively and additionally compounds A7 and A12 both showed 2 Vander Waal interactions as well. The lead compound has been identified from the 5 most active compounds, after analyzing them on the basis of their interactions and IC50 values. Among them ligand A7 has been identified as the lead compound. The strong ligand protein interactions, which include 8hydrogen bonds and 2 hydrophobic interactions and 2 Vander Waal interactions is shown in Fig. 2 and the low IC50 amongst the five proved it to be the most active one. The interactions shown in Fig. 2, highlights the important amino acids of the target protein pocket and the atoms of the ligand. Fig. 2 also shows the bonding distances of the ligand with target protein. Compound A7 consisted of 8 hydrogen bonds i.e. hydrogen bond with amino acid ER247 with a distance of 3.56 amino acids TYR136, LY151, VAL137, TYR90, GLU147, TYR90 and VAL137 with the distances of 3.41,2.88, 3.27, 2.76, 3.84, 3.94 and 3.15A 0 respectively, A7 consisted of two hydrophobic interactions with amino acid LEU89 and GLU144 with the distance of 3.64 and 3.85A 0 respectively and two hydrophobic interactions with amino acids PE150 and TYR90 with the distance of 3.52 and 3.78A 0 and respectively. The interactions of the standard compound have also been identified and are shown in Fig. 3. The binding interactions of Quercetin include 4 hydrogen bonds, 2 hydrophobic interactions and 1 Vander Waal interaction. The hydrogen bonds include GLU117 at a distance of 2.96, of GLU117 at a distance of 2.97, of GL318 at a distance of 3.72 and of GL318 at a distance of 3.13with the standard compound. ydrophobic interactions include C of LY257 at a distance of 3.96 and C of I326 at a distance of 3.68 with standard compound. Vander Waal interaction is between hydrophobic rings of ligand with hydrophobic ring containing amino acid TRP254 of active site at a distance of The binding interactions of the standard Quercetin A11 are very much close to the identified lead compound infact the number of interactions showed by lead compound are more than the standard compound and the IC50 of A7 is also lower than the A11. This gives stronger evidence that the identified lead compound has high efficacy and has great potential to be a drug. Table 2. Five active ligands 78 Page
5 Design and Molecular Docking of Antioxidant Lead compound and its Analogues Acting as uman Figure 2. Binding interactions of Lead compound with target 2CK Figure 3. Binding interactions of Quercetin with target 2CK 1.6. Analogues of lead Three structural analogues of the lead compound have been designed. Keeping in view the chemical structure of the lead compound, i.e., the hydrophobic features, BD s, BA s, the analogues have been developed. The analogues of the lead have been shown in Fig. 4. The analogues have been developed by the introduction or elimination of various groups. In the present study new analogues were formed by clammenson reduction, acetylene formation and ester formation. The first analogue oflead has been made by reducing ketones into hydrocarbon so in this way hydrophobic character has been increased.in acetylene formation double bonded hydrogen has been replaced by triple bond in order to increase the hydrophobicity of the compound. In case of third analogue group has been replaced by an ester in order to increase the polarity of the compound. Docking of the analogues through AutodockVina has been performed in order to get the active conformations of the analogues. The interactions of the active conformations of each of the analogue bound into the active site of the protein have been obtained using VMD. The interactions of the analogues with the protein have been shown in Table 3. n the basis of the interactions the analogues have been ranked for being the active ligands. Analogue 2 amongst the all is most active one so it is given the first rank, while second rank has been given to analogue 1, and third to analogue 3.The hydrogen bonds identified in analogues 1,2 and 3 are 4,7 and 2 in number, respectively, the hydrophobic interactions are 4,2and 0 respectively and the Vander Waal interactions are2, 3and 0 respectively. The binding interactions of the most active analogue have been shown, in its docked conformation in Fig. 5. Analogues like the lead compound showed high number of binding interactions. This suggests that the designed analogues are also active like the lead compound.an active antioxidant acting as tyrosine kinase inhibitor has been identified as a lead compound and three active novel analogues have been made which after their analysis are being proposed for clinical trials along with the lead compound for the treatment of uman Cancers caused by the disruption of tyrosine kinase. Figure 4. Analogues of lead compound 79 Page
6 Design and Molecular Docking of Antioxidant Lead compound and its Analogues Acting as uman Table 3.Binding interactions of analogues of lead compound Figure 5.Binding interactions of second analogue with target 2CK IV. Conclusion The current study was aimed at finding novel antioxidant molecules as tyrosine kinase inhibitors, hence as anticancer inhibitors.in this study various intermolecular interactions between target protein binding site of 2CK and different antioxidant compounds were observed. The purpose of this study was to evaluate therapeutic applications of antioxidants in cancer through molecular docking. The study identified an antioxidant Lead compound working as tyrosine kinase inhibitor, hence working as anticancer drug. The active novel analogues of the lead compound were also suggested for their anticancerous activity like the lead compound. References [1] Kang T, Jo, Park WK, Kim JP, Konishi Y, Kong JY, Parka and Junga Y, ynthesis and antioxidant activities of 3, 5- dialkoxy-4- hydroxycinnamamides, Bioorganic and [2] Medicinal Chemistry Letters 18, 2008, pp [3] mietana M, Clayette P, Mialocq P, Vasseur JJ and Joe, A ynthesis of new -isobutyryl-l-cysteine/mea conjugates: Evaluation of their free radical-scavenging activities and anti-iv properties in human macrophages, Bioorganic Chemistry 36, 2008, pp [4] Cox, elson M and David R, Principles of Biochemistry (W Freeman & Co,2008). [5] Lamson DW and Brignall M, Antioxidants and Cancer III:Quercetin, Altern Med Rev 5 (3), 2000, pp [6] Minich DM, Quantum upplements: A Total ealth and Wellness Makeover with Vitamins, Minerals, and erbs(conari Press 113, 2010). [7] K.R. Cousins, ChemDraw Ultra 9.0. Cambridge soft, 100 Cambridge ParkDrive, Cambridge, MA 02140, J.Am.Chem.oc., 127, 2005,pp [8] Chang MW, Ayeni C, Breuer, Torbett BE, Virtual creening for IV Protease Inhibitors: A Comparison of AutoDock 4 and Vina, PLo E 5(8), 2010, e [9] umphrey W, Dalke A, chulten K, VMD: visual molecular dynamics. J Mol Graph 14, 1996, pp [10] g L, Ko and Lu TM, Potential antioxidants and tyrosinase inhibitors from synthetic polyphenolicdeoxybenzoins, Journal of Bioorganic and Medicinal Chemistry 17, 2009, pp [11] Calliste CA, Kozlowski D, Duroux JL, Champavier Y, Chulia AJ and Trouillas P, A new antioxidant from wild nutmeg, Journal of Food Chemistry 118, 2009, pp [12] Kumar V and agarajaaik, ynthesis and antioxidant properties of some novel 5-dibenz[b,f]azepinederivatives in different in vitro model systems, European Journal of Medicinal Chemistry 45, 2010, pp [13] Kus C, Ayhan G, zbey, Kaynak FB, Kaya M, Coban T and Eke BC, ynthesis and antioxidant properties of novel methyl-1, 3, 4- thiadiazol-2-amine and 4-methyl-2-1, 2, 4-triazole-3(4)-thione derivatives of benzimidazole class, Journal of Bioorganic & Medicinal Chemistry 16, 2008, pp [14] Rao YK, Geethangili M, Fang and Tzeng YM, Antioxidant and cytotoxic activities of naturally occurring phenolic and related compounds: A comparative study, Journal of Food and Chemical Toxicology 45 (9), 2007, pp [15] tefek M, nirc V, Djoubissie P, Majekova M, Demopoulos V, Rackova L, Bezakova Z, and Wermuth CG, Ganellin, CR, Lindberg P and Mitscher LA, Glossary of terms used in medicinal chemistry, Elsevier 33, 1998, pp Page
Molecular Docking Studies of Flavonoids of Noni Fruit (Morinda citrifolia L.) to Peroxisome Proliferator-Activated Receptor-Gamma (PPARγ)
 3rd International Conference on Computation for Science and Technology (ICCST-3) Molecular Docking Studies of Flavonoids of Noni Fruit (Morinda citrifolia L.) to Peroxisome Proliferator-Activated Receptor-Gamma
3rd International Conference on Computation for Science and Technology (ICCST-3) Molecular Docking Studies of Flavonoids of Noni Fruit (Morinda citrifolia L.) to Peroxisome Proliferator-Activated Receptor-Gamma
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
6. SUMMARY AND CONCLUSION
 6. SUMMARY AND CONCLUSION Free radicals are chemical species containing one or more unpaired electrons, like hydrogen atom, most transition metal ions, nitric oxide and oxygen, with two unpaired electrons.
6. SUMMARY AND CONCLUSION Free radicals are chemical species containing one or more unpaired electrons, like hydrogen atom, most transition metal ions, nitric oxide and oxygen, with two unpaired electrons.
Excerpt from J. Mol. Biol. (2002) 320, :
 Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
INSILICO DOCKING AND INTERACTION ANALYSIS OF ELLAGIC ACID AND CURCUMIN DERIVATIVES AGAINST HUMAN CANCER
 ISSN: 0976-2876 (Print) ISSN: 2250-0138(Online) INSILICO DOCKING AND INTERACTION ANALYSIS OF ELLAGIC ACID AND CURCUMIN DERIVATIVES AGAINST HUMAN CANCER NAYANA MOHAN a AND M.S. LATHA b1 ab Department of
ISSN: 0976-2876 (Print) ISSN: 2250-0138(Online) INSILICO DOCKING AND INTERACTION ANALYSIS OF ELLAGIC ACID AND CURCUMIN DERIVATIVES AGAINST HUMAN CANCER NAYANA MOHAN a AND M.S. LATHA b1 ab Department of
Patrick, An Introduction to Medicinal Chemistry 5e Chapter 1 Drugs and drug targets: an overview
 Patrick, An Introduction to dicinal hemistry 5e Answers to end-of-chapter questions 1) The ability of a molecule to cross the fatty cell membrane has little to do with its size, but more with its hydrophobic
Patrick, An Introduction to dicinal hemistry 5e Answers to end-of-chapter questions 1) The ability of a molecule to cross the fatty cell membrane has little to do with its size, but more with its hydrophobic
Allosteric Inhibition of SHP2: Identification of a Potent, Selective, and Orally Efficacious Phosphatase Inhibitor!
 Allosteric Inhibition of SHP2: Identification of a Potent, Selective, and Orally Efficacious Phosphatase Inhibitor Allosteric pocket SHP2 Phosphatase ovel allosteric Phosphatase inhibitor Evan Carder Wipf
Allosteric Inhibition of SHP2: Identification of a Potent, Selective, and Orally Efficacious Phosphatase Inhibitor Allosteric pocket SHP2 Phosphatase ovel allosteric Phosphatase inhibitor Evan Carder Wipf
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Investigation of the binding of spermine and derivative polyamines on the 1U6M active site in the presence of Acetyl coenzyme-a
 Investigation of the binding of spermine and derivative polyamines on the 1U6M active site in the presence of Acetyl coenzyme-a Lindsay Mitchell lm04575n@pace.edu Pace University NYC Department of Chemistry
Investigation of the binding of spermine and derivative polyamines on the 1U6M active site in the presence of Acetyl coenzyme-a Lindsay Mitchell lm04575n@pace.edu Pace University NYC Department of Chemistry
number Done by Corrected by Doctor
 number 19 Done by حسام ابو عوض Corrected by وسيم ابو عبيدة Doctor د.نايف 1 P a g e GAGs and Glycoproteins: GAGs: long, unbranched heteropolysaccharides, made from زunits repeating disaccharide [Acidic
number 19 Done by حسام ابو عوض Corrected by وسيم ابو عبيدة Doctor د.نايف 1 P a g e GAGs and Glycoproteins: GAGs: long, unbranched heteropolysaccharides, made from زunits repeating disaccharide [Acidic
3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B
 3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B 3.2.1 Characterization of the Ligand Binding Site at GPR109A Receptor Ligands of GPR109A Receptor are Carboxylic Acids Nicotinic acid (pyridine-3-carboxylic
3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B 3.2.1 Characterization of the Ligand Binding Site at GPR109A Receptor Ligands of GPR109A Receptor are Carboxylic Acids Nicotinic acid (pyridine-3-carboxylic
LIGAND BASED PHARMACOPHORE DEVELOPMENT FOR COLORECTAL CANCER DRUGS
 The Professional Medical Journal www.theprofesional.com ORIGINAL PROF-2566 LIGAND BASED PHARMACOPHORE DEVELOPMENT FOR COLORECTAL CANCER DRUGS 1. PhD Scholar (Bioinformatics) M.A.J.U. Islamabad. 2. Postgraduate
The Professional Medical Journal www.theprofesional.com ORIGINAL PROF-2566 LIGAND BASED PHARMACOPHORE DEVELOPMENT FOR COLORECTAL CANCER DRUGS 1. PhD Scholar (Bioinformatics) M.A.J.U. Islamabad. 2. Postgraduate
Patrick, An Introduction to Medicinal Chemistry 4e Chapter 5 Receptors and signal transduction
 atrick, An Introduction to Medicinal Chemistry 4e Answers to end-of-chapter questions 1) The diagram in the question shows two important hydrogen bonding interactions where AT acts both as a hydrogen bond
atrick, An Introduction to Medicinal Chemistry 4e Answers to end-of-chapter questions 1) The diagram in the question shows two important hydrogen bonding interactions where AT acts both as a hydrogen bond
Structure and Activation of the Visual Pigment Rhodopsin
 Seminar 02.12.2011 Structure and Activation of the Visual Pigment Rhodopsin by Steven O.Smith, 2010 Constantin Schneider FB Physik, FU Berlin GPCR: Overview Rhodopsin is a G protein-coupled receptor (GPCR)
Seminar 02.12.2011 Structure and Activation of the Visual Pigment Rhodopsin by Steven O.Smith, 2010 Constantin Schneider FB Physik, FU Berlin GPCR: Overview Rhodopsin is a G protein-coupled receptor (GPCR)
List of Figures. List of Tables
 Supporting Information for: Signaling Domain of Sonic Hedgehog as Cannibalistic Calcium-Regulated Zinc-Peptidase Rocio Rebollido-Rios 1, Shyam Bandari 3, Christoph Wilms 1, Stanislav Jakuschev 1, Andrea
Supporting Information for: Signaling Domain of Sonic Hedgehog as Cannibalistic Calcium-Regulated Zinc-Peptidase Rocio Rebollido-Rios 1, Shyam Bandari 3, Christoph Wilms 1, Stanislav Jakuschev 1, Andrea
Page 8/6: The cell. Where to start: Proteins (control a cell) (start/end products)
 Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Docking Analysis of Darunavir as HIV Protease Inhibitors
 Journal of Computational Methods in Molecular Design, 2012, 2 (1):39-43 Scholars Research Library (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231-3176 CODEN (USA): JCMMDA Docking Analysis
Journal of Computational Methods in Molecular Design, 2012, 2 (1):39-43 Scholars Research Library (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231-3176 CODEN (USA): JCMMDA Docking Analysis
In silico approach of compounds in Cissus quadrangularis targeting multiproteins as anticancer agents
 Research Article In silico approach of compounds in Cissus quadrangularis targeting multiproteins as anticancer agents C. N. Hemalatha 1, Elancheziyan K 1, D. Pavithra 2, M. Vijey Aanandhi 3 * ABSTRACT
Research Article In silico approach of compounds in Cissus quadrangularis targeting multiproteins as anticancer agents C. N. Hemalatha 1, Elancheziyan K 1, D. Pavithra 2, M. Vijey Aanandhi 3 * ABSTRACT
Important reactions of lipids
 Taif University College of Medicine Preparatory Year Students Medical chemistry (2) Part II (Lipids) week 4 lectures 1435-36 Important reactions of lipids Lectures outlines Definition and importance of
Taif University College of Medicine Preparatory Year Students Medical chemistry (2) Part II (Lipids) week 4 lectures 1435-36 Important reactions of lipids Lectures outlines Definition and importance of
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Term Definition Example Amino Acids
 Name 1. What are some of the functions that proteins have in a living organism. 2. Define the following and list two amino acids that fit each description. Term Definition Example Amino Acids Hydrophobic
Name 1. What are some of the functions that proteins have in a living organism. 2. Define the following and list two amino acids that fit each description. Term Definition Example Amino Acids Hydrophobic
CHAPTER 21: Amino Acids, Proteins, & Enzymes. General, Organic, & Biological Chemistry Janice Gorzynski Smith
 CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
Introduction. Basic Structural Principles PDB
 BCHS 6229 Protein Structure and Function Lecture 1 (October 11, 2011) Introduction Basic Structural Principles PDB 1 Overview Main Goals: Carry out a rapid review of the essentials of protein structure
BCHS 6229 Protein Structure and Function Lecture 1 (October 11, 2011) Introduction Basic Structural Principles PDB 1 Overview Main Goals: Carry out a rapid review of the essentials of protein structure
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Design and Modeling Studies on Liriodenine derivatives as novel topoisomerase II inhibitors
 International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol. 3, No.3,pp 1622-1627, July-Sept 2011 Design and Modeling Studies on Liriodenine derivatives as novel topoisomerase II
International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol. 3, No.3,pp 1622-1627, July-Sept 2011 Design and Modeling Studies on Liriodenine derivatives as novel topoisomerase II
Statin inhibition of HMG-CoA reductase: a 3-dimensional view
 Atherosclerosis Supplements 4 (2003) 3/8 www.elsevier.com/locate/atherosclerosis Statin inhibition of HMG-CoA reductase: a 3-dimensional view Eva Istvan * Department of Molecular Microbiology, Howard Hughes
Atherosclerosis Supplements 4 (2003) 3/8 www.elsevier.com/locate/atherosclerosis Statin inhibition of HMG-CoA reductase: a 3-dimensional view Eva Istvan * Department of Molecular Microbiology, Howard Hughes
An Introduction to Enzyme and Coenzyme Chemistry, 2nd Ed. T. D. H. Bugg, Blackwell Science, Oxford, 2004
 Combinatorial synthesis of linchpin β-turn mimic 1 2 DCC, BT 1 2 n -tbu 1 n -tbu 1) 2 FMC DCC, BT 2) piperidine 1 2 2 n -tbu 3 DCC, BT 1 2 n -tbu 3 1) Ph 3 P 2) cyclization 3) CF 3 C 2 2 1 n 3 2 Evaluated
Combinatorial synthesis of linchpin β-turn mimic 1 2 DCC, BT 1 2 n -tbu 1 n -tbu 1) 2 FMC DCC, BT 2) piperidine 1 2 2 n -tbu 3 DCC, BT 1 2 n -tbu 3 1) Ph 3 P 2) cyclization 3) CF 3 C 2 2 1 n 3 2 Evaluated
1. Denaturation changes which of the following protein structure(s)?
 Chem 11 Fall 2008 Examination #5 ASWER KEY MULTIPLE CICE (20 pts. total; 2 pts. each) 1. Denaturation changes which of the following protein structure(s)? a. primary b. secondary c. tertiary d. both b
Chem 11 Fall 2008 Examination #5 ASWER KEY MULTIPLE CICE (20 pts. total; 2 pts. each) 1. Denaturation changes which of the following protein structure(s)? a. primary b. secondary c. tertiary d. both b
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Amino Acids. Review I: Protein Structure. Amino Acids: Structures. Amino Acids (contd.) Rajan Munshi
 Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
A Random Forest Model for the Analysis of Chemical Descriptors for the Elucidation of HIV 1 Protease Protein Ligand Interactions
 A Random Forest Model for the Analysis of Chemical Descriptors for the Elucidation of HIV 1 Protease Protein Ligand Interactions Gene M. Ko, A. Srinivas Reddy, Sunil Kumar, Barbara A. Bailey, and Rajni
A Random Forest Model for the Analysis of Chemical Descriptors for the Elucidation of HIV 1 Protease Protein Ligand Interactions Gene M. Ko, A. Srinivas Reddy, Sunil Kumar, Barbara A. Bailey, and Rajni
Molecular docking analysis of UniProtKB nitrate reductase enzyme with known natural flavonoids
 www.bioinformation.net Volume 12(12) Hypothesis Molecular docking analysis of UniProtKB nitrate reductase enzyme with known natural flavonoids Ayub Shaik 1, Vishnu Thumma 2, Aruna Kumari Kotha 2, Sandhya
www.bioinformation.net Volume 12(12) Hypothesis Molecular docking analysis of UniProtKB nitrate reductase enzyme with known natural flavonoids Ayub Shaik 1, Vishnu Thumma 2, Aruna Kumari Kotha 2, Sandhya
Name. The following exam contains 44 questions, valued at 2.6 points/question. 2. Which of the following is not a principal use of proteins?
 Chemistry 131 Exam 3 Practice Proteins, Enzymes, and Carbohydrates Spring 2018 Name The following exam contains 44 questions, valued at 2.6 points/question 1. Which of the following is a protein? a. Amylase
Chemistry 131 Exam 3 Practice Proteins, Enzymes, and Carbohydrates Spring 2018 Name The following exam contains 44 questions, valued at 2.6 points/question 1. Which of the following is a protein? a. Amylase
Heat Shock Proteins in Targeted Cancer Chemotherapy
 Heat Shock Proteins in Targeted Cancer Chemotherapy Aykut ÖZGÜR 1 and Yusuf TUTAR 2,* 1 Gaziosmanpaşa University, Faculty of Natural Sciences and Engineering, Department of Bioengineering, Tokat, Turkey
Heat Shock Proteins in Targeted Cancer Chemotherapy Aykut ÖZGÜR 1 and Yusuf TUTAR 2,* 1 Gaziosmanpaşa University, Faculty of Natural Sciences and Engineering, Department of Bioengineering, Tokat, Turkey
ANTIOXIDANT ACTIVITY OF β-carboline DERIVATIVES
 Acta Poloniae Pharmaceutica ñ Drug Research, Vol. 68 No. 2 pp. 185ñ189, 2011 ISSN 0001-6837 Polish Pharmaceutical Society DRUG BIOCHEMISTRY ANTIOXIDANT ACTIVITY OF β-carboline DERIVATIVES RENATA FRANCIK
Acta Poloniae Pharmaceutica ñ Drug Research, Vol. 68 No. 2 pp. 185ñ189, 2011 ISSN 0001-6837 Polish Pharmaceutical Society DRUG BIOCHEMISTRY ANTIOXIDANT ACTIVITY OF β-carboline DERIVATIVES RENATA FRANCIK
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Computer Aided Docking Studies on Anticancer Drugs for Breast Cancer
 International Journal of Information & Computation Technology. ISSN 0974-2239 Volume 2, Number 1 (2012), pp. 49-55 International Research Publications House http://www. ripublication.com Computer Aided
International Journal of Information & Computation Technology. ISSN 0974-2239 Volume 2, Number 1 (2012), pp. 49-55 International Research Publications House http://www. ripublication.com Computer Aided
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
Synergistic effects of antioxidative peptides from rice bran
 Synergistic effects of antioxidative peptides from rice bran Pichamon Kiatwuthinon 1,*, Neeracha Lapanusorn 1, Anunyaporn Phungsom 1, Wirawan Tinanchai 1 1 Department of Biochemistry, Faculty of Science,
Synergistic effects of antioxidative peptides from rice bran Pichamon Kiatwuthinon 1,*, Neeracha Lapanusorn 1, Anunyaporn Phungsom 1, Wirawan Tinanchai 1 1 Department of Biochemistry, Faculty of Science,
Macromolecules of Life -3 Amino Acids & Proteins
 Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
7.012 F 04 Problem Set 1 September 10 th 2004
 MIT Biology Department 7.012: Introductory Biology Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. laudette Gardel ame Question 1 TA Section 7.012 F 04 Problem Set 1 September
MIT Biology Department 7.012: Introductory Biology Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. laudette Gardel ame Question 1 TA Section 7.012 F 04 Problem Set 1 September
SID#: Also give full SID# (w/ 9) on your computer grid sheet (fill in grids under Student Number) BIO 315 Exam I
 SID#: Also give full SID# (w/ 9) on your computer grid sheet (fill in grids under Student Number) BIO 315 Exam I Choose an answer of A,B, C, or D for each of the following Multiple Choice Questions 1-35.
SID#: Also give full SID# (w/ 9) on your computer grid sheet (fill in grids under Student Number) BIO 315 Exam I Choose an answer of A,B, C, or D for each of the following Multiple Choice Questions 1-35.
Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels. Supplementary Information
 Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels Craig T. Armstrong, Philip E. Mason, J. L. Ross Anderson and Christopher E. Dempsey
Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels Craig T. Armstrong, Philip E. Mason, J. L. Ross Anderson and Christopher E. Dempsey
Structural Analysis of TCRpMHC Complexes Using Computational Tools. Feroze Mohideen Briarcliff High School
 Structural Analysis of TCRpMHC Complexes Using Computational Tools Feroze Mohideen Briarcliff High School TCR-pMHC Complexes Peptide Structure of TCR-pMHC complex PDB AO7 Crossreactivity is the ability
Structural Analysis of TCRpMHC Complexes Using Computational Tools Feroze Mohideen Briarcliff High School TCR-pMHC Complexes Peptide Structure of TCR-pMHC complex PDB AO7 Crossreactivity is the ability
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase. Biol 405 Molecular Medicine
 Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
3150:112 SAMPLE TEST 2. Print out a copy Answer the questions on your own. Check the answers at GOBC Ans.pdf. Good Luck!
 SAMPLE TEST 2 3150:112 Print out a copy Answer the questions on your own. Check the answers at GOBC Ans.pdf. Good Luck! QUESTIONS 1-3 REFER TO TE FOLLOWING: A. C 2 O O B. C 2 O O O C 2 O C. O C 2 O 1.
SAMPLE TEST 2 3150:112 Print out a copy Answer the questions on your own. Check the answers at GOBC Ans.pdf. Good Luck! QUESTIONS 1-3 REFER TO TE FOLLOWING: A. C 2 O O B. C 2 O O O C 2 O C. O C 2 O 1.
Molecular Docking Studies on Pyrazolopyrimidine and their Derivatives as Human Phosphoinositide 3-Kinase Inhibitors
 Cloud Publications International Journal of Advanced Bioinformatics and Computational Biology 2012, Volume 1, Issue 1, pp. 1-11, Article ID Med-30 Research Article Open Access Molecular Docking Studies
Cloud Publications International Journal of Advanced Bioinformatics and Computational Biology 2012, Volume 1, Issue 1, pp. 1-11, Article ID Med-30 Research Article Open Access Molecular Docking Studies
Free Radicals in Biology and Medicine
 Free Radicals in Biology and Medicine 0 \ Second Edition BARRY HALLIWELL Professor of Medical Biochemistry, University of London King's College and JOHN M.C. GUTTERIDGE Senior Scientist, National Institute
Free Radicals in Biology and Medicine 0 \ Second Edition BARRY HALLIWELL Professor of Medical Biochemistry, University of London King's College and JOHN M.C. GUTTERIDGE Senior Scientist, National Institute
A Chemical Look at Proteins: Workhorses of the Cell
 A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
Available Online through
 ISS: 0975-766X CDE: IJPTFI Available nline through Research Article www.ijptonline.com RELATISHIP BETWEE SCRIG FUCTI AD THE IC 50 IHIBITRY F PI3K IHIBITRS Awantika Shrivastava*, Dr.K.Durga Prasad 1, Dr.
ISS: 0975-766X CDE: IJPTFI Available nline through Research Article www.ijptonline.com RELATISHIP BETWEE SCRIG FUCTI AD THE IC 50 IHIBITRY F PI3K IHIBITRS Awantika Shrivastava*, Dr.K.Durga Prasad 1, Dr.
Proteins are sometimes only produced in one cell type or cell compartment (brain has 15,000 expressed proteins, gut has 2,000).
 Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Cbl ubiquitin ligase: Lord of the RINGs
 Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
Chapter 3: Amino Acids and Peptides
 Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2009 Examination #5 ANSWER KEY For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2009 Examination #5 ANSWER KEY For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
1. (38 pts.) 2. (25 pts.) 3. (15 pts.) 4. (12 pts.) 5. (10 pts.) Bonus (12 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
PROTEINS. Amino acids are the building blocks of proteins. Acid L-form * * Lecture 6 Macromolecules #2 O = N -C -C-O.
 Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5
 Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Project Manual Bio3055. Apoptosis: Superoxide Dismutase I
 Project Manual Bio3055 Apoptosis: Superoxide Dismutase I Bednarski 2003 Funded by HHMI Apoptosis: Superoxide Dismutase I Introduction: Apoptosis is another name for programmed cell death. It is a series
Project Manual Bio3055 Apoptosis: Superoxide Dismutase I Bednarski 2003 Funded by HHMI Apoptosis: Superoxide Dismutase I Introduction: Apoptosis is another name for programmed cell death. It is a series
If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out.
 Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
! Proteins are involved functionally in almost everything: " Receptor Proteins - Respond to external stimuli. " Storage Proteins - Storing amino acids
 Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Identification of polyphenolic compound inhibitors for 1, 3 β D- glucan synthase in the treatment of dermatophytoses by molecular docking
 International Journal of Advanced Research in Medical Sciences 2017 Volume 1 Issue 1 Pages: 1-5 Original Research Paper Identification of polyphenolic compound inhibitors for 1, 3 β D- glucan synthase
International Journal of Advanced Research in Medical Sciences 2017 Volume 1 Issue 1 Pages: 1-5 Original Research Paper Identification of polyphenolic compound inhibitors for 1, 3 β D- glucan synthase
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
SUPPORTING INFORMATION FOR. A Computational Approach to Enzyme Design: Using Docking and MM- GBSA Scoring
 SUPPRTING INFRMATIN FR A Computational Approach to Enzyme Design: Predicting ω- Aminotransferase Catalytic Activity Using Docking and MM- GBSA Scoring Sarah Sirin, 1 Rajesh Kumar, 2 Carlos Martinez, 2
SUPPRTING INFRMATIN FR A Computational Approach to Enzyme Design: Predicting ω- Aminotransferase Catalytic Activity Using Docking and MM- GBSA Scoring Sarah Sirin, 1 Rajesh Kumar, 2 Carlos Martinez, 2
Molecular Docking Studies of Shc1 Protein: A Drug Target to Treat Obesity
 IOSR Journal of Pharmacy and Biological Sciences (IOSR-JPBS) e-issn: 2278-3008, p-issn:2319-7676. Volume 9, Issue 6 Ver. III (Nov -Dec. 2014), PP 63-68 Molecular Docking Studies of Shc1 Protein: A Drug
IOSR Journal of Pharmacy and Biological Sciences (IOSR-JPBS) e-issn: 2278-3008, p-issn:2319-7676. Volume 9, Issue 6 Ver. III (Nov -Dec. 2014), PP 63-68 Molecular Docking Studies of Shc1 Protein: A Drug
UNIVERSITY OF GUELPH CHEM 4540 ENZYMOLOGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS
 UNIVERSITY F GUELPH CHEM 4540 ENZYMLGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS Instructions: Time allowed = 80 minutes. Total marks = 30. This quiz represents
UNIVERSITY F GUELPH CHEM 4540 ENZYMLGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS Instructions: Time allowed = 80 minutes. Total marks = 30. This quiz represents
Previous Class. Today. Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism. Protein Kinase Catalytic Properties
 Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
Dr. Nafith Abu Tarboush
 11 Dr. Nafith Abu Tarboush July 3 rd 2013 Marah Dannoun Continuation of amino acids applications in life other than tryptophan and tyrosine: 1. Glutamate (glutamic acid): it s modified to give monosodium
11 Dr. Nafith Abu Tarboush July 3 rd 2013 Marah Dannoun Continuation of amino acids applications in life other than tryptophan and tyrosine: 1. Glutamate (glutamic acid): it s modified to give monosodium
This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is worth 2 points.
 MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
Workshop on Analysis and prediction of contacts in proteins
 Workshop on Analysis and prediction of contacts in proteins 1.09.09 Eran Eyal 1, Vladimir Potapov 2, Ronen Levy 3, Vladimir Sobolev 3 and Marvin Edelman 3 1 Sheba Medical Center, Ramat Gan, Israel; Departments
Workshop on Analysis and prediction of contacts in proteins 1.09.09 Eran Eyal 1, Vladimir Potapov 2, Ronen Levy 3, Vladimir Sobolev 3 and Marvin Edelman 3 1 Sheba Medical Center, Ramat Gan, Israel; Departments
Amino acids. Dr. Mamoun Ahram Summer semester,
 Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Practice Problems 3. a. What is the name of the bond formed between two amino acids? Are these bonds free to rotate?
 Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Ionization of amino acids
 Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site.
 Supplementary Figure 1 Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site. (a) Gel filtration profiles of purified 5 TMD samples at 100 nm, heated beforehand for
Supplementary Figure 1 Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site. (a) Gel filtration profiles of purified 5 TMD samples at 100 nm, heated beforehand for
Chapter 11 Nutrition: Food for Thought
 Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Hydrogen Bonds and Biological Activities
 ugo Kubinyi, www.kubinyi.de ydrogen Bonds and Biological Activities ugo Kubinyi Germany EMail kubinyi@tonline.de omepage www.kubinyi.de ugo Kubinyi, www.kubinyi.de ature of the ydrogen Bond C=... a hydrogen
ugo Kubinyi, www.kubinyi.de ydrogen Bonds and Biological Activities ugo Kubinyi Germany EMail kubinyi@tonline.de omepage www.kubinyi.de ugo Kubinyi, www.kubinyi.de ature of the ydrogen Bond C=... a hydrogen
Pharmacologic Principles. Dr. Alia Shatanawi
 Pharmacologic Principles Dr. Alia Shatanawi Definitions Drug: It is any chemical that affect living processes. It modifies an already existing function, and does not create a new function. 2 What is Pharmacology?
Pharmacologic Principles Dr. Alia Shatanawi Definitions Drug: It is any chemical that affect living processes. It modifies an already existing function, and does not create a new function. 2 What is Pharmacology?
Protein Cleavage Due to Pro-oxidative Activity in Some Spices
 Protein Cleavage Due to Pro-oxidative Activity in Some Spices Sittiwat Lertsiri Department of Biotechnology Faculty of Science, Mahidol University Phayathai, Bangkok 10400 Thailand Kanchana Dumri Department
Protein Cleavage Due to Pro-oxidative Activity in Some Spices Sittiwat Lertsiri Department of Biotechnology Faculty of Science, Mahidol University Phayathai, Bangkok 10400 Thailand Kanchana Dumri Department
Final Report. Identification of Residue Mutations that Increase the Binding Affinity of LSTc to HA RBD. Wendy Fong ( 方美珍 ) CNIC; Beijing, China
 Final Report Identification of Residue Mutations that Increase the Binding Affinity of LSTc to HA RBD Wendy Fong ( 方美珍 ) CNIC; Beijing, China Influenza s Viral Life Cycle 1. HA on virus binds to sialic
Final Report Identification of Residue Mutations that Increase the Binding Affinity of LSTc to HA RBD Wendy Fong ( 方美珍 ) CNIC; Beijing, China Influenza s Viral Life Cycle 1. HA on virus binds to sialic
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination #5: Section Five December 7, Name: (print) Section:
 Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Amino acids & Protein Structure Chemwiki: Chapter , with most emphasis on 16.3, 16.4 and 16.6
 Amino acids & Protein Structure Chemwiki: Chapter 16. 16.1, 16.3-16.9 with most emphasis on 16.3, 16.4 and 16.6 1 1. Most jobs (except information storage) in cells are performed by proteins. 2. Proteins
Amino acids & Protein Structure Chemwiki: Chapter 16. 16.1, 16.3-16.9 with most emphasis on 16.3, 16.4 and 16.6 1 1. Most jobs (except information storage) in cells are performed by proteins. 2. Proteins
The building blocks of life.
 The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
Combining molecular modelling with experiments: Sulfonylureas and glinides as new PPARγ agonists
 Combining molecular modelling with experiments: Sulfonylureas and glinides as new PPARγ agonists Marco Scarsi Biozentrum - Swiss Institute of Bioinformatics Drug discovery and development 12-15 years of
Combining molecular modelling with experiments: Sulfonylureas and glinides as new PPARγ agonists Marco Scarsi Biozentrum - Swiss Institute of Bioinformatics Drug discovery and development 12-15 years of
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Research Article. Insilico predictions of inhibitors of novel statin structural analogues with HMG-CoA reductase
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(3):942-950 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Insilico predictions of inhibitors of novel statin
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(3):942-950 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Insilico predictions of inhibitors of novel statin
Deficiency. - Night blindness - Dry, rough skin - Decreased resistance to infection - Faulty tooth development - Slower bone growth
 MEDCHEM 562 Fall 2013 Vitamin Problem Set 1: 1) Complete the table: Vitamin A D Physiological Function / Therapeutic Uses - Vision - Promotion of cell differentiation - Inhibition of cell proliferation
MEDCHEM 562 Fall 2013 Vitamin Problem Set 1: 1) Complete the table: Vitamin A D Physiological Function / Therapeutic Uses - Vision - Promotion of cell differentiation - Inhibition of cell proliferation
Details of Organic Chem! Date. Carbon & The Molecular Diversity of Life & The Structure & Function of Macromolecules
 Details of Organic Chem! Date Carbon & The Molecular Diversity of Life & The Structure & Function of Macromolecules Functional Groups, I Attachments that replace one or more of the hydrogens bonded to
Details of Organic Chem! Date Carbon & The Molecular Diversity of Life & The Structure & Function of Macromolecules Functional Groups, I Attachments that replace one or more of the hydrogens bonded to
Break Desired Compound into 3 Smaller Ones
 Break Desired Compound into 3 Smaller nes A B C Indole ydrazine Phenol When creating a covalent link between model compounds move the charge on the deleted into the carbon to maintain integer charge (i.e.
Break Desired Compound into 3 Smaller nes A B C Indole ydrazine Phenol When creating a covalent link between model compounds move the charge on the deleted into the carbon to maintain integer charge (i.e.
Basics of Signal Transduction. Ebaa M Alzayadneh, PhD
 Basics of Signal Transduction Ebaa M Alzayadneh, PhD What is signal transduction? Cell signaling The science of understanding how individual cells sense their environments and respond to stimuli... how
Basics of Signal Transduction Ebaa M Alzayadneh, PhD What is signal transduction? Cell signaling The science of understanding how individual cells sense their environments and respond to stimuli... how
N NH 3 NHCNH 2. 3 Lysine (Lys)
 Make sure You have FIVE Different C 110 May, 2007 Pages Including this Cover Page. Cover Page You may pick up your graded exam after 120 minutes Final Exam 5/08/07 until 12/01/07 DETAC TIS SEET AND USE
Make sure You have FIVE Different C 110 May, 2007 Pages Including this Cover Page. Cover Page You may pick up your graded exam after 120 minutes Final Exam 5/08/07 until 12/01/07 DETAC TIS SEET AND USE
Chemical Nature of the Amino Acids. Table of a-amino Acids Found in Proteins
 Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
DEPARTMENT: Chemistry
 CODE: CHEM-236 TITLE: Biochemistry Institute: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: Upon completion of this course the student will be able to recognize and draw the structure and state the nature
CODE: CHEM-236 TITLE: Biochemistry Institute: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: Upon completion of this course the student will be able to recognize and draw the structure and state the nature
CHM333 LECTURE 6: 1/25/12 SPRING 2012 Professor Christine Hrycyna AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID:
 AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
Chemical Basis For Life Open Ended Questions:
 Chemical Basis For Life Open Ended Questions: Answer the following questions to the best of your ability: Make sure you read each question carefully and provide answers to all of the parts of the question.
Chemical Basis For Life Open Ended Questions: Answer the following questions to the best of your ability: Make sure you read each question carefully and provide answers to all of the parts of the question.
The chemistry of life
 The chemistry of life All living organisms are comprised of organic molecules. Organic molecules contain CARBON and HYDROGEN which is not true of inorganic molecules. Carbon is central to life on Earth
The chemistry of life All living organisms are comprised of organic molecules. Organic molecules contain CARBON and HYDROGEN which is not true of inorganic molecules. Carbon is central to life on Earth
BIOCHEMISTRY 460 FIRST HOUR EXAMINATION FORM A (yellow) ANSWER KEY February 11, 2008
 WRITE YOUR AND I.D. NUMBER LEGIBLY ON EVERY PAGE PAGES WILL BE SEPARATED FOR GRADING! CHECK TO BE SURE YOU HAVE 6 PAGES, (print): ANSWERS INCLUDING COVER PAGE. I swear/affirm that I have neither given
WRITE YOUR AND I.D. NUMBER LEGIBLY ON EVERY PAGE PAGES WILL BE SEPARATED FOR GRADING! CHECK TO BE SURE YOU HAVE 6 PAGES, (print): ANSWERS INCLUDING COVER PAGE. I swear/affirm that I have neither given
HOMEWORK II and Swiss-PDB Viewer Tutorial DUE 9/26/03 62 points total. The ph at which a peptide has no net charge is its isoelectric point.
 BIOCHEMISTRY I HOMEWORK II and Swiss-PDB Viewer Tutorial DUE 9/26/03 62 points total 1). 8 points total T or F (2 points each; if false, briefly state why it is false) The ph at which a peptide has no
BIOCHEMISTRY I HOMEWORK II and Swiss-PDB Viewer Tutorial DUE 9/26/03 62 points total 1). 8 points total T or F (2 points each; if false, briefly state why it is false) The ph at which a peptide has no
D. glycerol and fatty acids 4. Which is an example of an inorganic compound?
 Name: ate: 1. Glucose and maltose are classified as organic compounds because they are both 3. Which process is most directly responsible for the production of O 2 in these sugar solutions?. carbon-containing
Name: ate: 1. Glucose and maltose are classified as organic compounds because they are both 3. Which process is most directly responsible for the production of O 2 in these sugar solutions?. carbon-containing
