TABLE OF CONTENTS. ShurFit Anterior Cervical Interbody Fusion (ACIF) System Overview 2. Implant Specifications 3. Instrument Features 4
|
|
|
- Florence Chase
- 5 years ago
- Views:
Transcription
1 Surgical Technique
2 TABLE OF CONTENTS ShurFit Anterior Cervical Interbody Fusion (ACIF) System Overview 2 Product Highlights 2 Indications 2 Implant Specifications 3 Instrument Features 4 Surgical Technique 5 1. Patient Positioning & Approach 5 2. Exposure & Distraction 5 3. Endplate Preparation 5 4. Implant Sizing 5 5. Implant Insertion 6 6. Implant Positioning 7 7. Closure 7 8. Implant Removal 7 Implant/Instrument Information 8 Indications 12 Contraindications 12 Potential Adverse Effects 12 Warnings 12
3 ShurFit ACIF SYSTEM OVERVIEW The ShurFit Anterior Cervical Interbody Fusion System consists of implants with various heights to accommodate individual patient anatomy and graft material size. It is implanted from the anterior approach. It is to be packed with autogenous bone graft to facilitate fusion. The device is intended to provide mechanical support to the implanted level until biologic fusion is achieved. All components are manufactured from medical grade polyetheretherketone (Using PEEK-OPTIMA polymer from INVIBIO ). The products are supplied clean and NON-STERILE. Product Highlights: Large contact area optimizes vertebral body supports and minimizes risk of subsidence Trapezoidal design allows for proper anterior placement Large graft area provides generous biological coverage Strategically placed tantulum markers (1.9mm from front edge, 1.8mm from side edge) facilitate radiographic implant positioning Aggressive tooth pattern resists expulsion Indications: The ShurFit Anterior Cervical Interbody Fusion System is indicated for use in skeletally mature patients with degenerative disc disease (DDD) of the cervical spine at one disc level. DDD is defined as discogenic pain with degeneration of the disc confirmed by history and radiographic studies. ShurFit Anterior Cervical Interbody Fusion System implants are used to facilitate fusion in the cervical spine and are placed via an anterior approach at the C-3 to C-7 disc levels using autograft bone. Patients should have at least six weeks of non-operative treatment prior to treatment with an intervertebral fusion device. The device should be used with supplemental fixation. Please refer to package insert (LBL-IFU-006) for complete system description, indications and warnings. 2
4 IMPLANT SPECIFICATIONS ShurFit ACIF 14mm Wide, 12mm Deep 16mm Wide, 14mm Deep 5-12mm Heights (1mm increments) 5 and 10 Degree Lordotic Options 5 Degrees 10 Degrees Standard ShurFit ACIF Sizes Part Number Description Width & Depth ACIF05-05P Standard Cervical 5 degree x 5mm 14mm x 12mm ACIF05-06P Standard Cervical 5 degree x 6mm 14mm x 12mm ACIF05-07P Standard Cervical 5 degree x 7mm 14mm x 12mm ACIF05-08P Standard Cervical 5 degree x 8mm 14mm x 12mm ACIF05-09P Standard Cervical 5 degree x 9mm 14mm x 12mm ACIF05-10P Standard Cervical 5 degree x 10mm 14mm x 12mm ACIF05-11P Standard Cervical 5 degree x 11mm 14mm x 12mm ACIF05-12P Standard Cervical 5 degree x 12mm 14mm x 12mm ACIF10-05P Standard Cervical 10 degree x 5mm 14mm x 12mm ACIF10-06P Standard Cervical 10 degree x 6 mm 14mm x 12mm ACIF10-07P Standard Cervical 10 degree x 7 mm 14mm x 12mm ACIF10-08P Standard Cervical 10 degree x 8 mm 14mm x 12mm ACIF10-09P Standard Cervical 10 degree x 9 mm 14mm x 12mm ACIF10-10P Standard Cervical 10 degree x 10 mm 14mm x 12mm ACIF10-11P Standard Cervical 10 degree x 11 mm 14mm x 12mm ACIF10-12P Standard Cervical 10 degree x 12 mm 14mm x 12mm Wide ShurFit ACIF Sizes Part Number Description Width & Depth P Wide Cervical 5 degree 5mm 16mm x 14mm P Wide Cervical 5 degree 6mm 16mm x 14mm P Wide Cervical 5 degree 7mm 16mm x 14mm P Wide Cervical 5 degree 8mm 16mm x 14mm P Wide Cervical 5 degree 9mm 16mm x 14mm P Wide Cervical 5 degree 10mm 16mm x 14mm P Wide Cervical 5 degree 11mm 16mm x 14mm P Wide Cervical 5 degree 12mm 16mm x 14mm P Wide Cervical 10 degree 5mm 16mm x 14mm P Wide Cervical 10 degree 6mm 16mm x 14mm P Wide Cervical 10 degree 7mm 16mm x 14mm P Wide Cervical 10 degree 8mm 16mm x 14mm P Wide Cervical 10 degree 9mm 16mm x 14mm P Wide Cervical 10 degree 10mm 16mm x 14mm P Wide Cervical 10 degree 11mm 16mm x 14mm P Wide Cervical 10 degree 12mm 16mm x 14mm 3
5 INSTRUMENT FEATURES ShurFit ACIF Sizers Used to determine proper implant size Standard variations 14mm width x 12mm depth Wide variations 16mm width x 14mm depth All available 5mm to 12mm in 1mm increments; 5 degree and 10 degree See charts below for part numbers Part Number Description Width & Depth ACIFS05 Standard Cervical Sizer 5mm 14mm x 12mm ACIFS06 Standard Cervical Sizer 6mm 14mm x 12mm ACIFS07 Standard Cervical Sizer 7mm 14mm x 12mm ACIFS08 Standard Cervical Sizer 8mm 14mm x 12mm ACIFS09 Standard Cervical Sizer 9mm 14mm x 12mm ACIFS10 Standard Cervical Sizer 10mm 14mm x 12mm ACIFS11 Standard Cervical Sizer 11mm 14mm x 12mm ACIFS12 Standard Cervical Sizer 12mm 14mm x 12mm Part Number Description Width & Depth Wide Cervical Sizer 5mm 16mm x 14mm Wide Cervical Sizer 6mm 16mm x 14mm Wide Cervical Sizer 7mm 16mm x 14mm Wide Cervical Sizer 8mm 16mm x 14mm Wide Cervical Sizer 9mm 16mm x 14mm Wide Cervical Sizer 10mm 16mm x 14mm Wide Cervical Sizer 11mm 16mm x 14mm Wide Cervical Sizer 12mm 16mm x 14mm ShurFit ACIF Inserter Used to place the implant in intervertebral space Part Number: ShurFit ACIF Tamp Used to assist with implant insertion Part Number:
6 SURGICAL TECHNIQUE 1. Patient Positioning: Position the patient in a supine position on a radiolucent operating table. Ensure that the neck of the patient is in a sagittally neutral position and supported with a cushion. 2. Exposure/Distraction: Obtain anterior exposure per surgeon preference. Perform segmental distraction. Distraction of the segment is essential for restoring disc height as well as for providing optimal access to the intervertebral space. 3. Endplate Preparation: When the discectomy is complete, remove the superficial layers of the cartilaginous endplates to expose bleeding bone. This can be accomplished with a variety of instruments, such as osteotomes, scrapers, curettes and rasps. Adequate preparation of the endplates is important to enhance vascular supply to the fusion site. 4. Implant Sizing: The selection of the trial implant is based on the height, width and depth of the intervertebral space, the preparation technique and the patient s anatomy. Choose either a standard or wide footprint Cervical Sizer (ACIFS05-12 or ) with the appropriate lordosis (5 or 10 degrees) and height (5-12mm). The Cervical Sizers are double sided. Ensure that the side with the appropriate lordosis is selected. The Cervical Sizers do not have a dedicated cranial or caudal surface. They can be inserted into the intervertebral disc space with either surface pointing cranially. 5
7 5. Implant Insertion: Select the ACIF implant that corresponds to the footprint shape and height determined by the Cervical Sizer ( ). Rotate the knurled knob counter clockwise to expand the posts on the ACIF Inserter (Fig. 1). Insert the two posts of the ACIF Inserter into the corresponding slots of the ACIF Implant. Rotate the knurled knob of the ACIF Inserter clockwise to secure the ACIF Implant to the Inserter (Fig. 2). Figure 1 Insert the two posts of the ACIF Inserter into the corresponding slots of the ACIF Implant(Fig. 3). Rotate the knurled knob of the ACIF Inserter clockwise to secure the ACIF Implant to the Inserter (Fig. 4 & 5). Insert the ACIF Implant into the disc space (Fig. 6). If necessary, controlled and light hammering can be used to help advance the ACIF Implant into the intervertebral disc space. Figure 2 Once the ACIF Implant is properly positioned, turn the knurled knob of the ACIF Inserter counter clockwise to release the Inserter from the Implant (Fig. 7). Figure 3 Figure 4 Figure 6 Figure 5 Figure 7 6
8 SURGICAL TECHNIQUE 6. Implant Positioning: Verify the final ACIF Implant position relative to the vertebral bodies in the AP and Lateral directions with the help of an intraoperative x-ray. The ACIF Implant has two tantalum markers incorporated in the implant to enable accurate assessment of the implant position (Fig. 8). Figure 8 7. Closure: After visual and radiographic confirmation of the implant placement, the closure process can proceed. The ShurFit ACIF System surgical technique is a general guide for instrumentation. The surgeon should be familiar with anterior cervical fusion. 8. Implant Removal: If needed, the implant can be removed using the ShurFit ACIF Inserter ( ). 7
9 ShurFit ACIF IMPLANTS & INSTRUMENTS Implants Instruments Item No. Description Item No. Description ACIF05-05P ACIF05-06P ACIF05-07P ACIF05-08P ACIF05-09P ACIF05-10P ACIF05-11P ACIF05-12P ACIF10-05P ACIF10-06P ACIF10-07P ACIF10-08P ACIF10-09P ACIF10-10P ACIF10-11P ACIF10-12P P P P P P P P P P P P P P P P P ACIF Cage Peek 5 degree x 5mm ACIF Cage Peek 5 degree x 6mm ACIF Cage Peek 5 degree x 7mm ACIF Cage Peek 5 degree x 8mm ACIF Cage Peek 5 degree x 9mm ACIF Cage Peek 5 degree x 10mm ACIF Cage Peek 5 degree x 11mm ACIF Cage Peek 5 degree x 12mm ACIF Cage Peek 10 degree x 5mm ACIF Cage Peek 10 degree x 6mm ACIF Cage Peek 10 degree x 7mm ACIF Cage Peek 10 degree x 8mm ACIF Cage Peek 10 degree x 9mm ACIF Cage Peek 10 degree x 10mm ACIF Cage Peek 10 degree x 11mm ACIF Cage Peek 10 degree x 12mm ACIF-Wide 5 degree x 5mm ACIF-Wide 5 degree x 6mm ACIF-Wide 5 degree x 7mm ACIF-Wide 5 degree x 8mm ACIF-Wide 5 degree x 9mm ACIF-Wide 5 degree x 10mm ACIF-Wide 5 degree x 11mm ACIF-Wide 5 degree x 12mm ACIF-Wide 10 degree x 5mm ACIF-Wide 10 degree x 6mm ACIF-Wide 10 degree x 7mm ACIF-Wide 10 degree x 8mm ACIF-Wide 10 degree x 9mm ACIF-Wide 10 degree x 10mm ACIF-Wide 10 degree x 11mm ACIF-Wide 10 degree x 12mm HTR ACIF Rasp, Narrow, 2mm Stop (5mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (6mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (7mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (8mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (7mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (8mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (9mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (10mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (11mm Height) * HTR ACIF Rasp, Narrow, 2mm Stop (12mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (5mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (6mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (7mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (8mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (9mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (10mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (11mm Height) * HTR ACIF Rasp, Wide, 2mm Stop (12mm Height) * ACIFS05 Cervical Sizer 5mm ACIFS06 Cervical Sizer 6mm ACIFS07 Cervical Sizer 7mm ACIFS08 Cervical Sizer 8mm ACIFS09 Cervical Sizer 9mm ACIFS10 Cervical Sizer10mm ACIFS11 Cervical Sizer 11mm ACIFS12 Cervical Sizer12mm ACIF-Wide 5mm Sizer ACIF-Wide 6mm Sizer ACIF-Wide 7mm Sizer ACIF-Wide 8mm Sizer ACIF-Wide 9mm Sizer ACIF-Wide 10mm Sizer ACIF-Wide 11mm Sizer ACIF-Wide 12mm Sizer ACIF Inserter ACIF Tamp * Special order 8
10 INDICATIONS CONTRAINDICATIONS: The ShurFit Anterior Cervical Interbody Fusion System (ACIF) contraindications include, but are not limited to: 1. Prior fusion at the level(s) to be treated 2. Any condition not described in the indications for use 3. Previous vascular approach 4. Iliofemoral arteriosclerosis 5. Morbid obesity 6. Mental illness 7. Pregnancy 8. Local infection or inflammation 9. Any case needing to mix metals from different components 10. Any patient unwilling to cooperate with postoerative instructions 11. All cases not stated in the indications 12. Reuse, or multiple uses POTENTIAL ADVERSE EFFECTS: The following potential adverse effects associated with the procedure have been shown to occur with the use of similar spinal systems. All patients considered candidates for fusion should be informed concerning the pathogenesis of their spinal abnormality, the rationale for fusion with instrumentation, and the potential adverse effects. The following are potential adverse effects, but not limited to: 1. Loss of proper spinal curvature, correction, height, and/or reduction 2. Infection 3. Non-union or delayed union 4. Foreign body reaction to the implants 5. Hemorrhaging 6. Loss of neurological function, dural tear, pain, and/or discomfort 7. Bone graft fracture, vertebral body fracture or discontinued growth of fused at, above and/or below the surgery level 8. Bending, loosening, fracture, disassembly, slippage and/or migration of the components 9. Pain or discomfort 10. Change in mental status 11. Bursitis 12. Bone loss and/or bone fracture due to stress shielding 13. Inability to resume activities of normal daily activities 14. Revision surgery 15. Death WARNINGS: The following are warnings for this device. 1. Patients with previous spinal surgery at the level(s) to be treated may have different clinical outcomes compared to those without previous surgery. 2. Potential risks identified with the use of this device system, which may require additional surgery, include device component fracture, loss of fixation, non-union, fracture of the vertebrae, necrosis of the bone, neurological injury, and/or vascular or visceral injury. 3. The benefit of spinal fusions utilizing any interbody fusion device has not been adequately established in patients with stable spines. 4. Patient selection and compliance will greatly affect the results. Patients suffering from obesity, malnutrition, and/or poor bone quality are poor candidates for spinal fusion. Patients who smoke or abuse alcohol are poor candidates for spinal fusion. 5. Patients who smoke should be advised of the consequences of the fact that an increased incidence of non-union has been reported with patients who smoke. 6. The implants and instruments are provided non-sterile and must be cleaned and sterilized before use. Device components should be sterilized using one of the noted validated sterilization cycle parameters. 7. A successful result is not always achieved in every surgical case due to many extenuating circumstances. This is especially true in spinal surgeries where other patient conditions may compromise the results. 8. Never reuse an internal fixation device under any circumstances. 9. This device is not intended to be the sole means of spinal support. The ShurFit Anterior Cervical Interbody System (ACIF) must be used with additional anterior and/or posterior instrumentation to augment stability. 10. Only surgeons trained and experienced in spinal decompression and bone grafting techniques should use the ShurFit Anterior Cervical Interbody System (ACIF). Preoperative and operating procedures, including knowledge of surgical techniques and proper selection and placement of the implants are essential considerations in the utilization of this device. 11. Physician note: Although the physician is the learned intermediary, the important medical information given in this document should be conveyed to the patient. 12. Do not reuse implants. Discard used, damaged, or otherwise suspect implants. AN IMPLANT SHOULD NEVER BE REUSED. Any implant, once used, should be discarded. Even though it appears undamaged, it may have small defects and internal stress patterns that may lead to failure. Reuse can potentially compromise device performance and patient safety. 9
11 Precision Spine, Inc Executive Drive Pearl, MS Customer Service: Phone: Toll Free: Fax: Caution: Federal (USA) law restricts these devices to sale by or on the order of a physician. ShurFit and Precision Spine are trademarks of Precision Spine, Inc. Copyright 2015 Precision Spine, Inc. All rights reserved. P/N LBL-STG-018 Rev. B 08/2015
TABLE OF CONTENTS. Vault C Anterior Cervical Discectomy 2 and Fusion (ACDF) System Overview. Implants 3. Instruments 5. Surgical Technique 10
 Surgical Technique TABLE OF CONTENTS Vault C Anterior Cervical Discectomy 2 and Fusion (ACDF) System Overview Indications 2 Implants 3 Instruments 5 Surgical Technique 10 1. Preoperative planning 10 2.
Surgical Technique TABLE OF CONTENTS Vault C Anterior Cervical Discectomy 2 and Fusion (ACDF) System Overview Indications 2 Implants 3 Instruments 5 Surgical Technique 10 1. Preoperative planning 10 2.
C-THRU Anterior Spinal System
 C-THRU Anterior Spinal System Surgical Technique Manufactured From Contents Introduction... Page 1 Design Features... Page 2 Instruments... Page 3 Surgical Technique... Page 4 Product Information... Page
C-THRU Anterior Spinal System Surgical Technique Manufactured From Contents Introduction... Page 1 Design Features... Page 2 Instruments... Page 3 Surgical Technique... Page 4 Product Information... Page
nva Anterior Lumbar Interbody Fusion System
 nva Anterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nva Anterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nvt Transforaminal Lumbar Interbody Fusion System
 nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
nvp Posterior Lumbar Interbody Fusion System
 nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
Apache Cervical Interbody Fusion Device. Surgical Technique. Page of 13. LC-005 Rev F
 LC-005 Rev F Apache Cervical Interbody Fusion Device Page of 13 Surgical Technique INDICATIONS: When used as an intervertebral body fusion device, the Genesys Spine Interbody Fusion System is indicated
LC-005 Rev F Apache Cervical Interbody Fusion Device Page of 13 Surgical Technique INDICATIONS: When used as an intervertebral body fusion device, the Genesys Spine Interbody Fusion System is indicated
EXACTECH SPINE. Operative Technique. Cervical Spacer System. Surgeon focused. Patient driven. TM
 EXACTECH SPINE Operative Technique Cervical Spacer System Surgeon focused. Patient driven. TM ACAPELLA ONE Acapella One Cervical Spacer System is an anterior cervical discectomy and fusion device with
EXACTECH SPINE Operative Technique Cervical Spacer System Surgeon focused. Patient driven. TM ACAPELLA ONE Acapella One Cervical Spacer System is an anterior cervical discectomy and fusion device with
Alamo C. Cervical Interbody System Surgical Technique. An Alliance Partners Company
 Cervical Interbody System Surgical Technique Table of Contents Indications for Use................................1 Device Description............................... 1 Alamo C Instruments..............................
Cervical Interbody System Surgical Technique Table of Contents Indications for Use................................1 Device Description............................... 1 Alamo C Instruments..............................
Cervical Spacer System surgical technique
 Blackhawk TM Cervical Spacer System surgical technique Blackhawk TM The BLACKHAWK Cervical Spacer System is designed to provide biomechanical stabilization as an adjunct to fusion. Spinal fixation should
Blackhawk TM Cervical Spacer System surgical technique Blackhawk TM The BLACKHAWK Cervical Spacer System is designed to provide biomechanical stabilization as an adjunct to fusion. Spinal fixation should
Royal Oak IBFD System Surgical Technique Posterior Lumbar Interbody Fusion (PLIF)
 Royal Oak IBFD System Surgical Technique Posterior Lumbar Interbody Fusion (PLIF) Preoperative Planning Preoperative planning is necessary for the correct selection of lumbar interbody fusion devices.
Royal Oak IBFD System Surgical Technique Posterior Lumbar Interbody Fusion (PLIF) Preoperative Planning Preoperative planning is necessary for the correct selection of lumbar interbody fusion devices.
Surgical Technique. Apache Anterior Lumbar Interbody Fusion
 Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
HawkeyeTM Peek. surgical technique
 HawkeyeTM Peek surgical technique Introduction The ChoiceSpine HAWKEYE Vertebral Body Replacement (VBR) System is intended for use in the thoracolumbar spine (T1 - L5) to replace a collapsed, damaged,
HawkeyeTM Peek surgical technique Introduction The ChoiceSpine HAWKEYE Vertebral Body Replacement (VBR) System is intended for use in the thoracolumbar spine (T1 - L5) to replace a collapsed, damaged,
Surgical Technique. Apache Posterior Lumbar Interbody Fusion Apache Transforaminal Lumbar Interbody Fusion
 Surgical Technique Apache Posterior Lumbar Interbody Fusion Apache Transforaminal Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 5 Disc Exposure 5 Disc and
Surgical Technique Apache Posterior Lumbar Interbody Fusion Apache Transforaminal Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 5 Disc Exposure 5 Disc and
Alamo T Transforaminal Lumbar Interbody System Surgical Technique
 Transforaminal Lumbar Interbody System Surgical Technique Table of Contents Indications and Device Description.............. 1 Alamo T Implant Features and Instruments...........2 Surgical Technique......................
Transforaminal Lumbar Interbody System Surgical Technique Table of Contents Indications and Device Description.............. 1 Alamo T Implant Features and Instruments...........2 Surgical Technique......................
Posterior Lumbar Interbody Fusion System
 Px Posterior Lumbar Interbody Fusion System Px PEEK INTERBODY FUSION SYSTEM INDICATIONS FOR USE The Innovasis Px PEEK IBF System is an intervertebral body fusion device for use in patients with degenerative
Px Posterior Lumbar Interbody Fusion System Px PEEK INTERBODY FUSION SYSTEM INDICATIONS FOR USE The Innovasis Px PEEK IBF System is an intervertebral body fusion device for use in patients with degenerative
DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY
 0086 Instructions for Use ShurFit Anterior Cervical Interbody Fusion System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION
0086 Instructions for Use ShurFit Anterior Cervical Interbody Fusion System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use
 Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
PILLAR AL. Anterior Lumbar Interbody Fusion (ALIF) and Partial Vertebral Body Replacement (pvbr) PEEK Spacer System OPERATIVE TECHNIQUE
 PILLAR AL PEEK Spacer System Anterior Lumbar Interbody Fusion (ALIF) and Partial Vertebral Body Replacement (pvbr) OPERATIVE TECHNIQUE Table of Contents 1 INTRODUCTION 2 PRE-OPERATIVE TECHNIQUE 3 OPERATIVE
PILLAR AL PEEK Spacer System Anterior Lumbar Interbody Fusion (ALIF) and Partial Vertebral Body Replacement (pvbr) OPERATIVE TECHNIQUE Table of Contents 1 INTRODUCTION 2 PRE-OPERATIVE TECHNIQUE 3 OPERATIVE
OPERATIVE TECHNIQUE. CONSTRUX Mini PTC. Mini PTC Spacer System
 OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
product overview Implant heights range from 8mm-20mm in 2mm increments, with two lordocic angle options of 6 and 12.
 ETHOS A-Spacer PEEK System Surgical Technique Guide Synchronizing Medical Innovation with Global Markets product overview The SyncMedical Ethos PEEK IBF System is an intervertebral body fusion device for
ETHOS A-Spacer PEEK System Surgical Technique Guide Synchronizing Medical Innovation with Global Markets product overview The SyncMedical Ethos PEEK IBF System is an intervertebral body fusion device for
Solitaire Anterior Spinal System
 Surgical Technique Solitaire Anterior Spinal System Independent Stabilization for the Anterior Column Available in Titanium and Contents Introduction... Page 1 Design Features... Page 2 Instruments...
Surgical Technique Solitaire Anterior Spinal System Independent Stabilization for the Anterior Column Available in Titanium and Contents Introduction... Page 1 Design Features... Page 2 Instruments...
CROSS -FUSE P E E K V B R / I B F SYST E M
 S U R G I C A L T E C H N I Q U E CROSS -FUSE P E E K V B R / I B F SYST E M S U R G I C A L S Y S T E M O V E R V I E W 2 CROSS-FUSE P E E K V B R / I B F S Y S T E M S U R G I C A L T E C H N I Q U E
S U R G I C A L T E C H N I Q U E CROSS -FUSE P E E K V B R / I B F SYST E M S U R G I C A L S Y S T E M O V E R V I E W 2 CROSS-FUSE P E E K V B R / I B F S Y S T E M S U R G I C A L T E C H N I Q U E
AVS ARIA TM Product Overview
 AVS ARIA TM Product Overview Table of Contents Indication 3 Adaptive Sizing 4-5 Wedge Nose Design 6 Graft Volumes 7 Efficient Fixation 8 Lordotic Descriptions 9 Anatomical Descriptions 10 Visualization
AVS ARIA TM Product Overview Table of Contents Indication 3 Adaptive Sizing 4-5 Wedge Nose Design 6 Graft Volumes 7 Efficient Fixation 8 Lordotic Descriptions 9 Anatomical Descriptions 10 Visualization
Fusion Device. Surgical Technique. Cervical Interbody Fusion with Trabecular Metal Technology
 TM-S Fusion Device Surgical Technique Cervical Interbody Fusion with Trabecular Metal Technology 2 TM-S Fusion Device Surgical Technique Disclaimer This surgical technique is not intended for use in the
TM-S Fusion Device Surgical Technique Cervical Interbody Fusion with Trabecular Metal Technology 2 TM-S Fusion Device Surgical Technique Disclaimer This surgical technique is not intended for use in the
Table of Contents.
 surgical technique The Ambassador TM Anterior Cervical Plate System is a versatile system of implants and instruments with a variety of sizes to provide optimal anatomic compatibility. The integrated cam
surgical technique The Ambassador TM Anterior Cervical Plate System is a versatile system of implants and instruments with a variety of sizes to provide optimal anatomic compatibility. The integrated cam
Advantage ALIF. Keith Shevlin Managing Director
 Advantage ALIF Unit 10, 9-11 Myrtle Street, Crows Nest NSW 2065 Keith Shevlin Managing Director keithshevlin@precisionsurgical.com.au Advantage ALIF Introduction & Indications for Use 1 Surgical Technique
Advantage ALIF Unit 10, 9-11 Myrtle Street, Crows Nest NSW 2065 Keith Shevlin Managing Director keithshevlin@precisionsurgical.com.au Advantage ALIF Introduction & Indications for Use 1 Surgical Technique
TM TM Surgical Technique
 TM TM Surgical Technique TABLE OF CONTENTS Reli SP Spinous Plating System Overview Device Description Implant Features Indications Instruments Access Instruments Preparation Instruments Insertion Instruments
TM TM Surgical Technique TABLE OF CONTENTS Reli SP Spinous Plating System Overview Device Description Implant Features Indications Instruments Access Instruments Preparation Instruments Insertion Instruments
Thoracolumbar Solutions. Zyston Curve. Interbody Spacer System. Surgical Technique Guide
 Thoracolumbar Solutions Zyston Curve Interbody Spacer System Surgical Technique Guide 2 Zyston Curve Interbody Spacer System Surgical Technique Guide The Zyston Curve Interbody System is designed to optimize
Thoracolumbar Solutions Zyston Curve Interbody Spacer System Surgical Technique Guide 2 Zyston Curve Interbody Spacer System Surgical Technique Guide The Zyston Curve Interbody System is designed to optimize
The Fusion of. Strength, Design. Performance. and
 The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
Apache Interbody Surgical Technique. LC004 Rev F
 LC004 Rev F Apache Interbody Surgical Technique Apache Anterior Lumbar Interbody Fusion Surgical Technique INDICATIONS: When used as an intervertebral body fusion device, the Genesys Spine Interbody Fusion
LC004 Rev F Apache Interbody Surgical Technique Apache Anterior Lumbar Interbody Fusion Surgical Technique INDICATIONS: When used as an intervertebral body fusion device, the Genesys Spine Interbody Fusion
Cervical Solutions. Trinnect. Hydrated Anterior Cervical Spacer System. Surgical Technique Guide
 Cervical Solutions Trinnect Hydrated Anterior Cervical Spacer System Surgical Technique Guide 2 Trinnect Hydrated Anterior Cervical Spacer System Surgical Technique Guide Trinnect Cervical Allograft Spacers
Cervical Solutions Trinnect Hydrated Anterior Cervical Spacer System Surgical Technique Guide 2 Trinnect Hydrated Anterior Cervical Spacer System Surgical Technique Guide Trinnect Cervical Allograft Spacers
Interbody fusion cage for the transforaminal approach. Travios. Surgical Technique
 Interbody fusion cage for the transforaminal approach Travios Surgical Technique Image intensifier control This description alone does not provide sufficient background for direct use of DePuy Synthes
Interbody fusion cage for the transforaminal approach Travios Surgical Technique Image intensifier control This description alone does not provide sufficient background for direct use of DePuy Synthes
PARADIGM SPINE. Anterior Cervical Fusion Cage. Cervical Interbody Fusion
 PARADIGM SPINE Anterior Cervical Fusion Cage Cervical Interbody Fusion DESIGN RATIONALE The OptiStrain TM C* interbody fusion cage follows well established biomechanical principles: The slot design of
PARADIGM SPINE Anterior Cervical Fusion Cage Cervical Interbody Fusion DESIGN RATIONALE The OptiStrain TM C* interbody fusion cage follows well established biomechanical principles: The slot design of
Zimmer Anterior Buttress Plate System. Surgical Technique
 Zimmer Anterior Buttress Plate System Surgical Technique 2 Zimmer Anterior Buttress Plate System Surgical Technique Zimmer Anterior Buttress Plate System Surgical Technique Description, Indications & Contraindications...
Zimmer Anterior Buttress Plate System Surgical Technique 2 Zimmer Anterior Buttress Plate System Surgical Technique Zimmer Anterior Buttress Plate System Surgical Technique Description, Indications & Contraindications...
Surgical technique. SynCage-C short
 Surgical technique SynCage-C short Table of contents Implants 2 Indications/contra-indications 3 Surgical technique 4 Image intensifier control Warning This description is not sufficient for immediate
Surgical technique SynCage-C short Table of contents Implants 2 Indications/contra-indications 3 Surgical technique 4 Image intensifier control Warning This description is not sufficient for immediate
Surgical Technique INTERSOMATIC CERVICAL CAGE
 R INTERSOMATIC CERVICAL CAGE NEOCIF IMPLANTS NEOCIF is an implant designed to make anterior cervical interbody fusion (ACIF) easier and to remove the need for structural autologous graft. The cage is made
R INTERSOMATIC CERVICAL CAGE NEOCIF IMPLANTS NEOCIF is an implant designed to make anterior cervical interbody fusion (ACIF) easier and to remove the need for structural autologous graft. The cage is made
SynCage-C short. Surgical Technique. This publication is not intended for distribution in the USA.
 SynCage-C short Surgical Technique This publication is not intended for distribution in the USA. Instruments and implants approved by the AO Foundation. Table of contents Implants 2 Indications/contra-indications
SynCage-C short Surgical Technique This publication is not intended for distribution in the USA. Instruments and implants approved by the AO Foundation. Table of contents Implants 2 Indications/contra-indications
IBD PEEK C. Anterior Cervical Spacer System. Surgical Technique
 IBD PEEK C Anterior Cervical Spacer System Surgical Technique Table of Contents Introduction.... 3 Step 1: Exposure... 3 Step 2: Discectomy... 4 Step 3: Endplate Preparation... 5 Step 4: Trialing... 6
IBD PEEK C Anterior Cervical Spacer System Surgical Technique Table of Contents Introduction.... 3 Step 1: Exposure... 3 Step 2: Discectomy... 4 Step 3: Endplate Preparation... 5 Step 4: Trialing... 6
SURGICAL TECHNIQUE MANUAL. InterFuse T
 1 CONTENTS InterFuse T Product Description 3 Indications for Use 3 X-Ray Marker Locations 4 Product Specifications 4 Instrument Set 5 Step 1 Preoperative Planning 8 Patient Positioning 8 Step 2 Disc Removal
1 CONTENTS InterFuse T Product Description 3 Indications for Use 3 X-Ray Marker Locations 4 Product Specifications 4 Instrument Set 5 Step 1 Preoperative Planning 8 Patient Positioning 8 Step 2 Disc Removal
Patient Information MIS LLIF. Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine...2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine...2 General Conditions of the Spine....4 What is Spondylolisthesis....5
O.I.C. PEEK UniLIF Unilateral Lumbar Interbody Fusion
 O.I.C. PEEK UniLIF Unilateral Lumbar Interbody Fusion A new implant for an evolving procedure Unique wedge nose Adaptive longer sizing Large internal graft volume Introduction Stryker Spine enters a new
O.I.C. PEEK UniLIF Unilateral Lumbar Interbody Fusion A new implant for an evolving procedure Unique wedge nose Adaptive longer sizing Large internal graft volume Introduction Stryker Spine enters a new
SURGICAL TECHNIQUE GUIDE BRECKENRIDGE. Intervertebral Body/VBR Fusion System. ACDF Anterior Cervical Discectomy and Fusion
 SURGICAL TECHNIQUE GUIDE TM BRECKENRIDGE Intervertebral Body/VBR Fusion System ACDF Anterior Cervical Discectomy and Fusion The following general Surgical Technique Guide is for illustrative purposes only.
SURGICAL TECHNIQUE GUIDE TM BRECKENRIDGE Intervertebral Body/VBR Fusion System ACDF Anterior Cervical Discectomy and Fusion The following general Surgical Technique Guide is for illustrative purposes only.
ACP1 CERVICAL PLATE SPINAL SYSTEM SURGICAL TECHNIQUE GUIDE II.
 I. ACP1 CERVICAL PLATE II. SPINAL SYSTEM SURGICAL TECHNIQUE GUIDE I. Introduction The Gold Standard Orthopaedics, LLC ACP1 Spinal System was designed with surgeons to incorporate strength, functionality,
I. ACP1 CERVICAL PLATE II. SPINAL SYSTEM SURGICAL TECHNIQUE GUIDE I. Introduction The Gold Standard Orthopaedics, LLC ACP1 Spinal System was designed with surgeons to incorporate strength, functionality,
POSTERIOR SPINE TRUSS SYSTEM
 POSTERIOR SPINE TRUSS SYSTEM ORDERING & SHIPPING ALL FIELDS ARE REQUIRED ORDER FORM PLEASE PRINT LEGIBLY RECEIVING YOUR PSTS TRAYS & IMPLANTS All implant bins and trays have a warehouse number on them
POSTERIOR SPINE TRUSS SYSTEM ORDERING & SHIPPING ALL FIELDS ARE REQUIRED ORDER FORM PLEASE PRINT LEGIBLY RECEIVING YOUR PSTS TRAYS & IMPLANTS All implant bins and trays have a warehouse number on them
SURGICAL TECHNIQUE GUIDE TRESTLE. Anterior Cervical Plating System
 SURGICAL TECHNIQUE GUIDE TRESTLE Anterior Cervical Plating System 2 SURGICAL TECHNIQUE GUIDE SURGICAL TECHNIQUE GUIDE System Features Large window enables visualization of graft site and end plates Screw
SURGICAL TECHNIQUE GUIDE TRESTLE Anterior Cervical Plating System 2 SURGICAL TECHNIQUE GUIDE SURGICAL TECHNIQUE GUIDE System Features Large window enables visualization of graft site and end plates Screw
Imola Lateral IBF System Surgical Technique
 Imola Lateral IBF System Surgical Technique IMOLA CIRCUIT TABLE OF CONTENTS Design Rationale Instructions for Use Surgical Technique 1. Table Mounting 2. Surgical Planning & Targeting 3. Access and Preparation
Imola Lateral IBF System Surgical Technique IMOLA CIRCUIT TABLE OF CONTENTS Design Rationale Instructions for Use Surgical Technique 1. Table Mounting 2. Surgical Planning & Targeting 3. Access and Preparation
Commander Cervical Cage - SURGICAL TECHNIQUE
 Commander Cervical Cage - SURGICAL TECHNIQUE D e s i g n e d to c l o s e l y f i t yo u! Commander Cervical Cage - SURGICAL TECHNIQUE Indications Commander cervical cages are designed primarly for restoring
Commander Cervical Cage - SURGICAL TECHNIQUE D e s i g n e d to c l o s e l y f i t yo u! Commander Cervical Cage - SURGICAL TECHNIQUE Indications Commander cervical cages are designed primarly for restoring
EFSPINE CERVICAL COMBINED SET DISC PROTHESIS ORGANIZER BOX
 EFSPINE CERVICAL COMBINED SET INSTRUMENTS CERVICAL CAGE & DISC PROTHESIS ORGANIZER BOX Cervical Thoracic Thoraco - Lumbar Sacral EFSPINE CERVICAL COMBINED SET CERVICAL IMPLANTS INTRODUCTION Cervical Disc
EFSPINE CERVICAL COMBINED SET INSTRUMENTS CERVICAL CAGE & DISC PROTHESIS ORGANIZER BOX Cervical Thoracic Thoraco - Lumbar Sacral EFSPINE CERVICAL COMBINED SET CERVICAL IMPLANTS INTRODUCTION Cervical Disc
MODULAR DESIGN OFFERS FREEDOM OF CHOICE. Surgical Technique
 MODULAR DESIGN OFFERS FREEDOM OF CHOICE Surgical Technique Joint Spine Sports Med MectaLIF Anterior Surgical Technique 2 INDEX 1. INTRODUCTION 4 1.1 Material & Marker 5 2. INDICATIONS 5 3. CONTRAINDICATIONS
MODULAR DESIGN OFFERS FREEDOM OF CHOICE Surgical Technique Joint Spine Sports Med MectaLIF Anterior Surgical Technique 2 INDEX 1. INTRODUCTION 4 1.1 Material & Marker 5 2. INDICATIONS 5 3. CONTRAINDICATIONS
Oracle Spacer. A PEEK spacer for lateral lumbar interbody fusion.
 Oracle Spacer. A PEEK spacer for lateral lumbar interbody fusion. Anatomic shape Self-distracting nose Large central canal Instruments and implants approved by the AO Foundation Oracle Spacer The Oracle
Oracle Spacer. A PEEK spacer for lateral lumbar interbody fusion. Anatomic shape Self-distracting nose Large central canal Instruments and implants approved by the AO Foundation Oracle Spacer The Oracle
XRL A modular expandable radiolucent vertebral body replacement system
 XRL A modular expandable radiolucent vertebral body replacement system This publication is not intended for distribution in the USA. SURGICAL TECHNIQUE Table of Contents Introduction XRL 2 AO Spine Principles
XRL A modular expandable radiolucent vertebral body replacement system This publication is not intended for distribution in the USA. SURGICAL TECHNIQUE Table of Contents Introduction XRL 2 AO Spine Principles
T-PAL Spacer System. Transforaminal posterior atraumatic lumbar spacer system.
 T-PAL Spacer System. Transforaminal posterior atraumatic lumbar spacer system. One instrument, one technique Accommodates both open and MIS approaches A PEEK implant that works with patient anatomy Streamlined
T-PAL Spacer System. Transforaminal posterior atraumatic lumbar spacer system. One instrument, one technique Accommodates both open and MIS approaches A PEEK implant that works with patient anatomy Streamlined
Instructions for Use AccuFit Lateral Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician
 Instructions for Use AccuFit Lateral Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There is no
Instructions for Use AccuFit Lateral Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There is no
Synex System TECHNIQUE GUIDE. An expandable vertebral body replacement device
 Synex System TECHNIQUE GUIDE An expandable vertebral body replacement device Original Instruments and Implants of the Association for the Study of Internal Fixation AO ASIF Synex System Overview The Synex
Synex System TECHNIQUE GUIDE An expandable vertebral body replacement device Original Instruments and Implants of the Association for the Study of Internal Fixation AO ASIF Synex System Overview The Synex
Veyron -C Anterior Cervical System Surgical Technique
 Veyron -C Anterior Cervical System Surgical Technique 2 Veyron-C Anterior Cervical System Surgical Technique Veyron-C Anterior Cervical System Surgical Technique Description, Indications & Contraindications...3
Veyron -C Anterior Cervical System Surgical Technique 2 Veyron-C Anterior Cervical System Surgical Technique Veyron-C Anterior Cervical System Surgical Technique Description, Indications & Contraindications...3
EIT TLIF Cage. For Natural Bone Ingrowth with EIT Cellular Titanium
 EIT TLIF Cage For Natural Bone Ingrowth with EIT Cellular Titanium EIT TLIF Cage Surgical Technique EIT Cellular Titanium provides active fusion area» ~ 80% porosity» ~ 650 µm diamond pore size» open interconnected
EIT TLIF Cage For Natural Bone Ingrowth with EIT Cellular Titanium EIT TLIF Cage Surgical Technique EIT Cellular Titanium provides active fusion area» ~ 80% porosity» ~ 650 µm diamond pore size» open interconnected
Cervical Solutions. Alta. ACDF System. Surgical Technique Guide
 Cervical Solutions Alta ACDF System Surgical Technique Guide 2 Alta ACDF System Surgical Technique Guide A comprehensive system to address a continuum of fixation requirements and anatomic demands. Alta
Cervical Solutions Alta ACDF System Surgical Technique Guide 2 Alta ACDF System Surgical Technique Guide A comprehensive system to address a continuum of fixation requirements and anatomic demands. Alta
Technique Guide. ECD Expandable Corpectomy Device. Continuously Expandable Vertebral Body Replacement for Tumour Cases.
 Technique Guide ECD Expandable Corpectomy Device. Continuously Expandable Vertebral Body Replacement for Tumour Cases. Table of Contents Introduction Overview 2 AO ASIF Principles 4 Indications and Contraindications
Technique Guide ECD Expandable Corpectomy Device. Continuously Expandable Vertebral Body Replacement for Tumour Cases. Table of Contents Introduction Overview 2 AO ASIF Principles 4 Indications and Contraindications
VTI INTERFUSE S SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK.
 VTI INTERFUSE S SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. CONTENTS InterFuse S Product Description Indications for Use X-Ray Marker Locations and Product Specifications Instrument Set 3 4 5-7 STEP
VTI INTERFUSE S SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. CONTENTS InterFuse S Product Description Indications for Use X-Ray Marker Locations and Product Specifications Instrument Set 3 4 5-7 STEP
Royal Oak Cervical Plate System
 Royal Oak Cervical Plate System Manufactured by Nexxt Spine, Inc. Royal Oak Cervical Plate System INTRODUCTION FEATURES AND BENEFITS Table of Contents SURGICAL TECHNIQUE Step 1. Patient Positioning Step
Royal Oak Cervical Plate System Manufactured by Nexxt Spine, Inc. Royal Oak Cervical Plate System INTRODUCTION FEATURES AND BENEFITS Table of Contents SURGICAL TECHNIQUE Step 1. Patient Positioning Step
The Fusion of Power and Performance.
 The Fusion of Power and Performance. Leading the Way in Lumbar Interbody Fusion with 1 Years of Clinical Results. The JAGUAR Lumbar I/F CAGE System, approved in the U.S. in February 1999, has revolutionized
The Fusion of Power and Performance. Leading the Way in Lumbar Interbody Fusion with 1 Years of Clinical Results. The JAGUAR Lumbar I/F CAGE System, approved in the U.S. in February 1999, has revolutionized
Zeus Intervertebral Body Fusion Devices PACKAGE INSERT
 LB-119 Rev 4 Zeus Intervertebral Body Fusion Devices PACKAGE INSERT CAUTION: Federal law (USA) restricts these devices to sale by or on the order of a physician. Implants and disposable instruments single
LB-119 Rev 4 Zeus Intervertebral Body Fusion Devices PACKAGE INSERT CAUTION: Federal law (USA) restricts these devices to sale by or on the order of a physician. Implants and disposable instruments single
The Implant. (Klappen außen sind nur 192 mm breit!)
 SEMIAL product information The Implant SEMIAL implants are designed to restore interbody vertebral disc height and maintain solid bony fusion when used in conjunction with bone graft material and supportive
SEMIAL product information The Implant SEMIAL implants are designed to restore interbody vertebral disc height and maintain solid bony fusion when used in conjunction with bone graft material and supportive
100 Interpace Parkway Parsippany, NJ
 100 Interpace Parkway Parsippany, NJ 07054 www.biometspine.com 800-526-2579 All trademarks are the property of Biomet, Inc. or one of its subsidiaries, unless otherwise indicated. Rx Only. 2009 EBI, LLC.
100 Interpace Parkway Parsippany, NJ 07054 www.biometspine.com 800-526-2579 All trademarks are the property of Biomet, Inc. or one of its subsidiaries, unless otherwise indicated. Rx Only. 2009 EBI, LLC.
GIZA Surgical Technique
 GIZA Surgical Technique Vertebral Body Replacement System Manufactured by Titanium alloy material provides mechanical integrity during insertion and distraction, x-ray visibility, and biocompatibility*
GIZA Surgical Technique Vertebral Body Replacement System Manufactured by Titanium alloy material provides mechanical integrity during insertion and distraction, x-ray visibility, and biocompatibility*
Patient Information MIS LLIF. Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine....2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine....2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Patient Information ACDF. Anterior Cervical Discectomy and Fusion
 Patient Information ACDF Anterior Cervical Discectomy and Fusion Table of Contents Anatomy of the Spine...2-3 General Conditions of the Cervical Spine...4 5 What is an ACDF?...6 How is an ACDF performed?...7
Patient Information ACDF Anterior Cervical Discectomy and Fusion Table of Contents Anatomy of the Spine...2-3 General Conditions of the Cervical Spine...4 5 What is an ACDF?...6 How is an ACDF performed?...7
Transforaminal Lumbar Interbody Fusion Cage (TLIF)
 Transforaminal Lumbar Interbody Fusion age (TLIF) 990100010 DOULE ENGINE MEDIL MTERIL O., LTD. No. 218 Houxiang Road, Haicang District, Xiamen 361022, P.R.hina Tel: +86 592 6087101 Fax: +86 592 6587078
Transforaminal Lumbar Interbody Fusion age (TLIF) 990100010 DOULE ENGINE MEDIL MTERIL O., LTD. No. 218 Houxiang Road, Haicang District, Xiamen 361022, P.R.hina Tel: +86 592 6087101 Fax: +86 592 6587078
BONE GRAFT WASHER _Rev A IMPORTANT INFORMATION ON THE BONE GRAFT WASHER
 0381080_Rev A BONE GRAFT WASHER IMPORTANT INFORMATION ON THE BONE GRAFT WASHER 03/2008 Medtronic Sofamor Danek USA, Inc. 1800 Pyramid Place Memphis, Tennessee 38132 Telephone: 800-933-2625 (in U.S.A.)
0381080_Rev A BONE GRAFT WASHER IMPORTANT INFORMATION ON THE BONE GRAFT WASHER 03/2008 Medtronic Sofamor Danek USA, Inc. 1800 Pyramid Place Memphis, Tennessee 38132 Telephone: 800-933-2625 (in U.S.A.)
Technique Guide. Zero-P VA. Variable angle zero-profile anterior cervical interbody fusion (ACIF) device.
 Technique Guide Zero-P VA. Variable angle zero-profile anterior cervical interbody fusion (ACIF) device. Image intensifier control Warning This description alone does not provide sufficient background
Technique Guide Zero-P VA. Variable angle zero-profile anterior cervical interbody fusion (ACIF) device. Image intensifier control Warning This description alone does not provide sufficient background
VTI INTERFUSE T SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. 1/20
 VTI INTERFUSE T SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. 1/20 CONTENTS InterFuse T Product Description Indications for Use X-Ray Marker Locations Product Specifications Instrument Set 3 4 5 STEP
VTI INTERFUSE T SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. 1/20 CONTENTS InterFuse T Product Description Indications for Use X-Ray Marker Locations Product Specifications Instrument Set 3 4 5 STEP
BAK/C Cervical Anterior Interbody Fusion System
 Surgical Technique BAK/C Cervical Anterior Interbody Fusion System The Comfortable Choice for Cervical Fusion BAK/C Cervical Surgical Technique 1 The BAK/C Cervical Fusion System is an alternative to conventional
Surgical Technique BAK/C Cervical Anterior Interbody Fusion System The Comfortable Choice for Cervical Fusion BAK/C Cervical Surgical Technique 1 The BAK/C Cervical Fusion System is an alternative to conventional
ECD EXPANDABLE CORPECTOMY DEVICE Continuously Expandable Vertebral Body Replacement for Tumour Cases
 ECD EXPANDABLE CORPECTOMY DEVICE Continuously Expandable Vertebral Body Replacement for Tumour Cases Instruments and implants approved by the AO Foundation. This publication is not intended for distribution
ECD EXPANDABLE CORPECTOMY DEVICE Continuously Expandable Vertebral Body Replacement for Tumour Cases Instruments and implants approved by the AO Foundation. This publication is not intended for distribution
ACP. Anterior Cervical Plate System SURGICAL TECHNIQUE
 ACP Anterior Cervical Plate System SURGICAL TECHNIQUE ACP TABLE OF CONTENTS INTRODUCTION 4 INDICATIONS AND CONTRAINDICATIONS 5 WARNINGS AND PRECAUTIONS 6 IMPLANT DESCRIPTION 7 INSTRUMENTS 10 SURGICAL
ACP Anterior Cervical Plate System SURGICAL TECHNIQUE ACP TABLE OF CONTENTS INTRODUCTION 4 INDICATIONS AND CONTRAINDICATIONS 5 WARNINGS AND PRECAUTIONS 6 IMPLANT DESCRIPTION 7 INSTRUMENTS 10 SURGICAL
Silverstone Interbody System Surgical Technique
 Silverstone Interbody System Surgical Technique TABLE OF CONTENTS Design Rationale Instructions for Use Surgical Technique 1. Disc Space Preparation 2. Distraction 3. Implant Selection 4. Implant Placement
Silverstone Interbody System Surgical Technique TABLE OF CONTENTS Design Rationale Instructions for Use Surgical Technique 1. Disc Space Preparation 2. Distraction 3. Implant Selection 4. Implant Placement
ACIS Anterior Cervical Interbody Spacer
 Implants and Instruments for Interbody Fusion Available in both PEEK and ProTi 360º Titanium Integrated Technology ACIS Anterior Cervical Interbody Spacer Surgical Technique Image intensifier control This
Implants and Instruments for Interbody Fusion Available in both PEEK and ProTi 360º Titanium Integrated Technology ACIS Anterior Cervical Interbody Spacer Surgical Technique Image intensifier control This
DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY
 0086 Instructions for Use RCS Anterior Buttress Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There
0086 Instructions for Use RCS Anterior Buttress Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There
PLIF/TPLIF Cage System
 PLIF/TPLIF Cage System LnK Lumbar Interbody Fusion Cage System LnK PLIF / T-PLIF Cage The LnK Interbody Fixation System implants are interbody fusion devices intended for use as an aid in spinal fixation.
PLIF/TPLIF Cage System LnK Lumbar Interbody Fusion Cage System LnK PLIF / T-PLIF Cage The LnK Interbody Fixation System implants are interbody fusion devices intended for use as an aid in spinal fixation.
Surgical Technique Manual
 InterFuse S Interbody Fusion System Surgical Technique Manual VERTEBRAL TECHNOLOGIES MS 4043-02 Rev. O Product Overview Introduction The VTI InterFuse S implant is an interbody fusion device that combines
InterFuse S Interbody Fusion System Surgical Technique Manual VERTEBRAL TECHNOLOGIES MS 4043-02 Rev. O Product Overview Introduction The VTI InterFuse S implant is an interbody fusion device that combines
Ardis. Surgical Technique. Interbody System. Solutions by the people of Zimmer Spine. zimmerspine.com
 Ardis Interbody System Surgical Technique Solutions by the people of Zimmer Spine. zimmerspine.com A more advanced interbody solution. From the people of Zimmer Spine. The easily inserted Ardis Interbody
Ardis Interbody System Surgical Technique Solutions by the people of Zimmer Spine. zimmerspine.com A more advanced interbody solution. From the people of Zimmer Spine. The easily inserted Ardis Interbody
Cervical Solutions. Optio-C Anterior Cervical Plate. with Allograft/Autograft. Surgical Technique Guide
 Cervical Solutions Optio-C Anterior Cervical Plate with Allograft/Autograft Surgical Technique Guide 2 Optio-C Anterior Cervical Plate with Allograft/Autograft Surgical Technique Guide The Optio-C System
Cervical Solutions Optio-C Anterior Cervical Plate with Allograft/Autograft Surgical Technique Guide 2 Optio-C Anterior Cervical Plate with Allograft/Autograft Surgical Technique Guide The Optio-C System
 Visit our website on www.biotech-medical.com The DLP - Dorso-Lumbar Polyaxial Screw System has been designed to address the pathologies of the thoracolumbar spine. The DLP System contains a wide range
Visit our website on www.biotech-medical.com The DLP - Dorso-Lumbar Polyaxial Screw System has been designed to address the pathologies of the thoracolumbar spine. The DLP System contains a wide range
Cervical Solutions. Optio-C Anterior Cervical PEEK. Interbody System. Surgical Technique Guide
 Cervical Solutions Optio-C Anterior Cervical PEEK Interbody System Surgical Technique Guide 2 Optio-C Anterior Cervical PEEK Interbody System Surgical Technique Guide The Optio-C System provides a zero-profile
Cervical Solutions Optio-C Anterior Cervical PEEK Interbody System Surgical Technique Guide 2 Optio-C Anterior Cervical PEEK Interbody System Surgical Technique Guide The Optio-C System provides a zero-profile
Replacement Device A modular expandable radiolucent vertebral body replacement system
 XRL Vertebral Body Replacement Device A modular expandable radiolucent vertebral body replacement system SURGICAL TECHNIQUE TABLE OF CONTENTS Introduction XRL System 2 AO Principles 5 Indications and Contraindications
XRL Vertebral Body Replacement Device A modular expandable radiolucent vertebral body replacement system SURGICAL TECHNIQUE TABLE OF CONTENTS Introduction XRL System 2 AO Principles 5 Indications and Contraindications
Cervical Solutions ROI-C. Cervical Cage. with ertebridge PLATING TECHNOLOGY. Surgical Technique Guide
 Cervical Solutions ROI-C Cervical Cage with ertebridge PLATING TECHNOLOGY Surgical Technique Guide 2 ROI-C Cervical Cage Surgical Technique Guide ROI-C featuring VerteBRIDGE plating is a zero-profile,
Cervical Solutions ROI-C Cervical Cage with ertebridge PLATING TECHNOLOGY Surgical Technique Guide 2 ROI-C Cervical Cage Surgical Technique Guide ROI-C featuring VerteBRIDGE plating is a zero-profile,
Aero -C Anterior Cervical Interbody and Fixation System
 Aero -C Anterior Cervical Interbody and Fixation System Table of contents System overview.... 3 Implant overview.... 4 Step 1. Patient positioning and exposure.... 6 Step 2. Discectomy and endplate preparation...
Aero -C Anterior Cervical Interbody and Fixation System Table of contents System overview.... 3 Implant overview.... 4 Step 1. Patient positioning and exposure.... 6 Step 2. Discectomy and endplate preparation...
OPERATIVE TECHNIQUE COVER IMAGE OPTIONAL (DETAIL) IMAGE PEEK PTC PILLAR SA PEEK AND PTC SPACER SYSTEM
 OPERATIVE TECHNIQUE COVER IMAGE OPTIONAL (DETAIL) IMAGE PEEK PTC PILLAR SA PEEK AND PTC SPACER SYSTEM TABLE OF CONTENTS Introduction 1 Operative Technique 2 - Partial Vertebral Body Replacement - Intervertebral
OPERATIVE TECHNIQUE COVER IMAGE OPTIONAL (DETAIL) IMAGE PEEK PTC PILLAR SA PEEK AND PTC SPACER SYSTEM TABLE OF CONTENTS Introduction 1 Operative Technique 2 - Partial Vertebral Body Replacement - Intervertebral
Zavation Z-Link Cervical
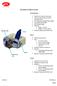 Zavation Z-Link Cervical Interbody Plate: Interbody Plate Screw Quarter turn locks for each screw Locks use same driver as used for inserting screws 30 caudal and cephalad biased angles 15 midline biased
Zavation Z-Link Cervical Interbody Plate: Interbody Plate Screw Quarter turn locks for each screw Locks use same driver as used for inserting screws 30 caudal and cephalad biased angles 15 midline biased
Amendia Interbody Fusion Devices PACKAGE INSERT
 LB-182 Rev 0 Amendia Interbody Fusion Devices PACKAGE INSERT CAUTION: Federal law (USA) restricts these devices to sale by or on the order of a physician. Implants and disposable instruments single use
LB-182 Rev 0 Amendia Interbody Fusion Devices PACKAGE INSERT CAUTION: Federal law (USA) restricts these devices to sale by or on the order of a physician. Implants and disposable instruments single use
Features & Benefits. 1) Wide Bone Graft Space. Anterior Margin 3mm Posterior Margin 1.5mm. Marker Margin
 ALIF Cage System LnK ALIF Cage System The LnK ALIF Cage can make solid inter-body fusion with optimized anatomical position. Broad bone graft area also makes perfect fusion. It can be used with additional
ALIF Cage System LnK ALIF Cage System The LnK ALIF Cage can make solid inter-body fusion with optimized anatomical position. Broad bone graft area also makes perfect fusion. It can be used with additional
A U X I L I A R Y C O N N E C T O R S Surgical Technique
 A U X I L I A R Y C O N N E C T O R S Surgical Technique AUXILIARY CONNECTORS ISSYS LP Auxiliary Connectors The ISSYS LP auxiliary connectors were designed to provide medial-lateral variability for the
A U X I L I A R Y C O N N E C T O R S Surgical Technique AUXILIARY CONNECTORS ISSYS LP Auxiliary Connectors The ISSYS LP auxiliary connectors were designed to provide medial-lateral variability for the
ACIS Anterior Cervical Interbody Spacer
 An enhanced system of implants and instruments for interbody fusion ACIS Anterior Cervical Interbody Spacer Surgical Technique Image intensifier control This description alone does not provide sufficient
An enhanced system of implants and instruments for interbody fusion ACIS Anterior Cervical Interbody Spacer Surgical Technique Image intensifier control This description alone does not provide sufficient
Lapidus Arthrodesis System Instructions for Use
 Lapidus Arthrodesis System Instructions for Use Description The AlignMATE Lapidus Arthrodesis System consists of bone plates and bone screws (locking, non-locking and interfragmentary), which are intended
Lapidus Arthrodesis System Instructions for Use Description The AlignMATE Lapidus Arthrodesis System consists of bone plates and bone screws (locking, non-locking and interfragmentary), which are intended
OPERATIVE TECHNIQUE PTC PEEK FORZA. spacer system
 OPERATIVE TECHNIQUE PTC PEEK FORZA spacer system TABLE OF CONTENTS Introduction 1 Operative Technique 2 Instruments 12 FORZA PEEK Part Numbers 20 FORZA PTC Part Numbers 22 Modular Implant Inserter 24 Disassembly
OPERATIVE TECHNIQUE PTC PEEK FORZA spacer system TABLE OF CONTENTS Introduction 1 Operative Technique 2 Instruments 12 FORZA PEEK Part Numbers 20 FORZA PTC Part Numbers 22 Modular Implant Inserter 24 Disassembly
INTELLIGENT SPINAL SYSTEM
 INTELLIGENT SPINAL SYSTEM I. Introduction II. Product Specification III. Surgical Technique IV. Ordering Information V. IFU for Lospa IS SPINAL SYSTEM The LOSPA IS spinal system consists of
INTELLIGENT SPINAL SYSTEM I. Introduction II. Product Specification III. Surgical Technique IV. Ordering Information V. IFU for Lospa IS SPINAL SYSTEM The LOSPA IS spinal system consists of
Instructions for use Prodisc -C Vivo Cervical disc prosthesis
 Instructions for use Prodisc -C Vivo Cervical disc prosthesis Instructions for use Prodisc-C Vivo Cervical disc prosthesis Safety precautions Please read these instructions for use, the Synthes brochure
Instructions for use Prodisc -C Vivo Cervical disc prosthesis Instructions for use Prodisc-C Vivo Cervical disc prosthesis Safety precautions Please read these instructions for use, the Synthes brochure
PEEK Cage for Posterior Lumbar Interbody Fusion (PLIF) Plivios Revolution. Surgical Technique
 PEEK Cage for Posterior Lumbar Interbody Fusion (PLIF) Plivios Revolution Surgical Technique Image intensifier control This description alone does not provide sufficient background for direct use of DePuy
PEEK Cage for Posterior Lumbar Interbody Fusion (PLIF) Plivios Revolution Surgical Technique Image intensifier control This description alone does not provide sufficient background for direct use of DePuy
