Modular Calcar Revision System
|
|
|
- Leo Reed
- 5 years ago
- Views:
Transcription
1 Modular Calcar Revision System
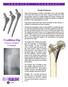
2 Engineered for long term stability, pain reduction, and optimal fixation for revision cases The Mallory/Head modular revision system offers stability at the time of surgery, fixation proximally in a compromised femur, and load transfer from proximal to distal in a physiological manner. Distal stems are offered straight and bowed, porous coated, and splined. Porous Plasma Sprayed for Long Term Stability Biomet s PPS Porous Plasma Spray may provide enhanced fixation and is supported by decades of clinical use. 1 Designed to Reduce Thigh Pain The matte-finish distal tip and coronal slot are designed to reduce the possibility of thigh pain and pedestal formation.
3 Supplied with Canal-Sealing Locking Screw The locking screw secures the proximal body and distal stem connection while also sealing the implants from errant soft tissues or other potential debris. It also helps to seal the modular junction from the joint space. Patented Roller Hardening Technology Roller-hardened tapers provide up to three times more strength in cantilever beam testing than standard, non-roller hardened tapers. 2 Engineered to Preserve Abductor Function A multitude of claws and bolts are available for adjunct support and fixation of the proximal body, and to aid in the preservation of good trochanteric bone. Made to Meet the Patient s Needs Five available resection levels in five different proximal body sizes simplify the process of matching patients anatomies. Proximal calcar components offer 34mm, 45mm, and 55mm resection levels in five sizes designated from small to large as A, B, C, D, and E.
4 Distal Stem Options In complex revision surgery, distal fixation is often needed as an adjunct to proximal fixation. The Mallory/Head modular system incorporates two distal stem designs including porous coated and splined stems. Where additional length is required to span distal cortical defects and provide additional fixation in the distal femur, 200mm to 300mm stems are available in coating levels ranking from 40% to 100% of the stem length. Distal stem diameters range from 11mm to 19mm for optimum patient fit. Macro sizes are available with stem diameters up to 23mm and stem lengths of 350mm. The Mallory/Head Modular Calcar Revision System also offers an STS Splined Tapered Stem. The design of the STS stem incorporates a 2 taper with sharp splines to engage the distal cortices. Its modularity with the load sharing proximal calcar component makes it a good offering in the modular revision system. The design allows for solid fixation in the distal femur with less voluminous metal. This lower modulus construct could potentially reduce end-of-stem pain often associated with distal fixed components. Stem diameters range from 14mm to 20mm in 1mm increments with stem lengths 200mm and 240mm.
5 Roller Hardening Roller hardening is a specialized, patented manufacturing process used to compress or work harden the taper region of the distal stem. This process increases the hardness of the titanium alloy metal at the interface of the taper junction, thus making the construct more resistant to fretting which may lead to fracture. Greater resistance to fretting translates into greater fatigue strength of the modular distal stem. This method of work hardening the taper is a patented process (U.S. patent #6,067,701) developed and owned by Biomet, Inc. Endurance Limit (lbs.) Others Non-Roller Hardened Tapers Biomet Roller Hardened Tapers Mallory/Head Modular Calcar Graph above shows significant increase in taper strength with roller hardened taper. All groups were tested in a cantilever beam testing fixture. As a result, the roller hardening process triples the endurance limit of the taper junction in cantilever beam fatigue testing. 2 This added strength meets the increased demands being placed on the taper junction due to: (A) lack of proximal bone stock; and (B) addition of high demand proximal components (lateralized and/or anteverted).
6 Easy-To-Use Instrumentation Optional Determining Version Distal Canal Reaming Flexible reamers over a guide wire are used to ream distally. Over-reaming is recommended when using a bowed stem. 4 Proximal Rasping The proximal broach prepares the metaphysis and is used in conjunction with a 170mm straight distal pilot that corresponds to the size of the last flexible reamer used. The broach and distal pilot are locked together with the self-contained screw in the proximal segment. The location for the medial keel is marked at this time. Once the broach is removed, a burr prepares the bone for the keel. Trial Implant Insertion Utilizing the correct distal trial, a trial proximal head/neck segment is used to confirm proper fit and anteversion. Once confirmed, the trial is locked together with the self-contained screw in the proximal segment. Be certain to position anteriorly the L or the R etched on the proximal component for the appropriate left or right hip. Position anteriorly the A on the distal component. This will lock the trial components in the proper plane. After extracting the trial stem from the femur, place the trial on the sundial upside down. Let the coronal pin on the sundial descend into the coronal slot in the trial and tighten thumbscrew knob. Reproducing Version Maintain the set version position and place the appropriate sized proximal implant on the sundial. Then, lightly place the appropriate distal implant into the proximal implant s taper being sure to place the A (for anterior) facing the same direction as the L for a left hip or the R for the right hip. Rotate the distal component to align the coronal slot with the coronal pin. This will reproduce the identical version from the trial. Impact the taper with a head pusher and mallet.
7 Bolt Assembly 60mm 55mm 50mm 45mm 40mm Insert the oblong bolt medially and hold into place with finger. An optional 3.5mm angled hex bolt holder may also be utilized. A 5.0mm T-handle hex wrench is then used to tighten the plate/bolt assembly. Final Implant Insertion After components are impacted, insert the segmental locking screw. The modular calcar inserter is then used to seat the implant as a single unit. Bolt and Claw Preparation With the final implant in place, the bolt hole may be drilled from medial to lateral or by utilizing the pictured lateral-to-medial drill guide, depending on the surgical approach. Measuring for Plate or Lateral Claw Connector A bolt hole gauge is used to determine the length of the final assembly for the plate and claw connector. Claw Assembly Insert the oblong bolt medially and hold into place with finger. Determine the claw size, medium or large, and attach to the greater trochanter. The appropriate size lateral claw connector is placed through the claw and tightened into the oblong bolt with a 5.0mm screwdriver. 5
8 Surgical Technique To address complex revision cases, the Mallory/Head modular instruments provide for ease of insertion as well as profound accuracy. With the ability to independently size the distal and proximal femur, the Mallory/ Head modular system provides customized fixation at the time of surgical intervention for revision arthroplasties. Preoperative Planning The femoral component must be stable and have adequate host bone support for predictable success. By utilizing X-rays and implant templates, a plan can be coordinated to ensure that this takes place. The platform area to be fashioned for the proximal calcar replacement can be approximated by utilizing the X-rays with regard to bone loss and bone quality. Final determination frequently cannot be made until the actual time of surgery, however with appropriate planning, a consistent operative plan with alternatives can be formulated. Selection of the proximal resection level can be predicted in order to restore leg length and to adjust the offset. It is desirable to obtain accurate fit and fill with both the proximal and distal portions of the prosthesis. The length of the stem should be such that the stem of the prosthesis extends at least 2cm beyond any perforation or lesion. In situations where the proximal portion of the prosthesis is seated on good quality bone, where the keel is keyed into the medial calcar region, and where good prosthesis/calcar contact is present, the bolt may not be necessary. In situations where there is not a complete proximal platform on which to seat the prosthesis or where bone quality or bone contact is incomplete as a result of bone destruction, the trochanteric bolt should be used. Biomechanical studies have demonstrated that this is the last line of defense in regard to proximal stability. It is not necessary to have complete circumferential bone present for calcar support. It is necessary, however, to have good quality bone for calcar support, making certain that at least 50% of the proximal component is supported on good quality host bone. This brochure demonstrates the surgical technique developed by Thomas H. Mallory M.D. and William C. Head, M.D. Biomet, as the manufacturer of this device, does not practice medicine and does not recommend this or any other surgical technique for use on a specific patient. The surgeon who performs any implant procedure is responsible for determining and utilizing the appropriate techniques for implanting the prosthesis in each individual patient. Biomet is not responsible for selection of the appropriate surgical technique to be utilized for an individual patient. 6
9 Patient Positioning and Surgical Approach The patient should be placed in a full lateral position with positioning devices placed to ensure complete patient stability (Figure 1). An anterolateral approach through a lateral curvilinear incision is recommended (Figure 2). The incision begins superiorly and posteriorly to the greater trochanter and extends distally along the lateral aspect of the femur, providing sufficient length for reconstruction and cement removal. When major acetabular reconstruction is present, a trochanteric osteotomy may need to be performed. Removal of a Cemented Component Care should be exercised when removing a femoral component and proximal cement to prevent a fracture in the trochanteric region or proximal femur. Fractures may be avoided by making certain that the lateral shoulder of the implant is not impinging on or buried into the greater trochanter. Once the implant is removed, the cement in the proximal femur can easily be removed with osteotomes or ultrasonic equipment (Figure 3). If proximal bone removal is needed in debridement, proceed with the proximal resection for the calcar prosthesis. This allows excellent visibility of the upper femur and canal access for cement removal. Controlled anterior femoral perforations are preferable in the debridement of the distal femur. The most distal perforation is made at the inferior pole of the cement column. X-rays are useful in accessing that position. Cement can then be easily removed with osteotomes or ultrasonic equipment (Figures 4a and 4b). Fig. 1 Fig. 3 Fig. 2 Fig. 4a Fig. 4b 7
10 Removal of an Ingrowth Component When removing an ingrowth component, an extended trochanteric osteotomy is usually helpful. If ingrowth is present proximally it is beneficial to proceed with the proximal resection for the calcar prosthesis. This allows for proximal loosening of and better access to the implant. In fully coated devices this procedure is almost always necessary. The fully coated device can then be cut with a carbide bit and a trephine reamer can be placed over the remaining distal portion to remove the femur. It is usually impossible to manually drive out a well-fixed ingrowth stem (Figures 5a 5c). Evaluation of Bone Quality The modular calcar replacement prosthesis should have a stable cortical platform fashioned on to the proximal femur. The shoulder of the prosthesis should be in contact with the cancellous bed of the greater trochanter. Note: It is extremely important to assess the quality of femoral bone and make the platform for the femoral prosthesis as proximal as possible. In the majority of revision situations, the platform resides near the lesser trochanter. (At times, it is possible to make the platform and still have bone atrophy or a bony defect.) In these situations, it is first necessary to ensure that the fashioned platform is adequate for seating of the prosthesis. Then, instead of removing unnecessary bone, cortical strut bone grafting is performed to ensure long-term prosthetic stability and support. In such situations, the patient must be kept to limited weight bearing until bone graft healing has occurred. In situations where strong host cortical bone exists, weight bearing as tolerated may begin based on surgeon discretion. Fig. 5a Fig. 5b Fig. 5c
11 Determination of Resection Level The calcar cutting guide or a trial prosthesis as a guide is used to make the proximal resection (Figures 6a and 6b). The modular calcar proximal component comes in 34, 45, and 55mm resection levels (Figure 7). It is necessary to have a stable platform on which to place the prosthesis. As stated earlier, in some situations proximal bone grafting must be done. At least two-thirds of the prosthetic platform should be supported by quality host bone. Femoral Canal Preparation Flexible reamers over a guide wire are utilized for distal reaming when a bowed distal stem is preferred (Figure 8). The distal femoral reaming for the 220 and 250mm should progress to approximately 1mm over the selected implant size to accommodate the bow of the component. Additional overreaming may be necessary if a distal stem with 80% porous coating is utilized. Cylindrical canal reamers are applied in preparing the distal femur when utilizing a 170mm straight distal component. Over-reaming is not necessary when implanting the 170mm straight, smooth distal stems (Figure 9a). Fig. 6a Fig. 6b Reaming for the STS distal stem requires the use of the STS reamers. STS reamers are advanced into the canal until cortical chatter is obtained. A line-to-line fit is recommended for the STS stem. There are four lines on the reamer to identify the various proximal components and STS stem lengths (Figure 9b). 1st line (most distal): Represents the 200mm STS distal with A Mallory/Head proximal body. 2nd line: Represents the 200mm STS distal with a B, C, D, and E Mallory/Head proximal body. Fig. 7 Fig. 8 Fig. 9a B E (240) A B E (200) A Fig. 9b 9
12 Rasp Insertion and Reduction Rasping the proximal femur may not be necessary or even possible when dealing with a compromised femur. If the proximal femur is not entirely compromised, a rasp can be utilized. Rasp Assembly The 170mm distal pilots are recommended when rasping. The appropriate sized distal pilots are attached to the selected proximal rasp by a self-contained segmental locking screw located in the proximal rasp. The screw is tightened with a 3.5mm screwdriver (Figure 10). Rasp Insertion Fig. 10 Upon assembly of the rasp, insert it into the femur to contour the proximal envelope. The distal pilots help to assure proper alignment with the shaft axis. Carefully advance the rasp until it becomes snug, then withdraw and repeat until the rasp is in position. The medial keel is then marked with methylene blue where it comes in contact with the medial calcar (Figure 11). After marking, the rasp is removed and a groove or recess is made in this same location, using a power burr. With the rasp reinserted, a trial reduction can be performed utilizing trial heads. Trial Insertion and Reduction Trial Assembly The final length distal provisional is attached to the appropriate size trial proximal prosthesis. When trialing with the 220mm, 250mm, and 300mm bowed distal provisionals, an A for anterior is marked on the distal tip of the trial stems. An L for left and an R for right is marked on the opposite sides of the proximal trials. To assemble and trial for a left femur, place the proximal trial segment with the L facing up over the appropriate sized bowed distal trial with the A facing up. Once the trial is assembled, tighten the self-contained locking screw in the proximal trial with a 3.5mm screwdriver (Figure 12). Note: When trialing for a right hip the R is facing up on the proximal trial. Fig. 12 Trial Insertion In order for the trial to be stable, it must be inserted beginning at 90 of anteversion and gradually turned into neutral or slight anteversion as it progresses in the canal. The trial stem should seat firmly on the horizontal femoral Fig. 13 platform with the medial keel in the groove or recess. With the trial in place, a proper reduction can be performed to assess joint stability. If a change in anteversion is required, the locking screw is loosened and the anteversion is adjusted. If the anteversion is adjusted, the recess or groove for the medial keel may need to be enlarged to allow flush seating of the platform (Figure 13). The groove implant stability is an absolute must. Care must be taken to adjust leg length and to evaluate the offset. Fig
13 Optional Version Setting Determining Version After extracting the trial stem from the femur, place the trial on the sundial upside down. Let the coronal pin on the sundial descend into the coronal slot in the trial and tighten the thumbscrew knob (Figure 14). A scale on the base of the sundial will indicate the degrees of version of the trial stem. Stem and Bolt Insertion Stem Assembly Maintain the set version of the sundial and place the appropriate sized proximal implant on the sundial. Like the trial, the proximal implant is marked with an L and an R and the distal stem is marked with an A. After placing the appropriate sized proximal implant on the sundial, lightly place the appropriate distal implant into the proximal implant s taper, being sure to place the A (for anterior) facing the same direction as the L for a left hip or the R for the right hip. Rotate the distal component to align the coronal slot with the coronal pin. This will reproduce the identical version from the trial (Figures 14 and 15). Impact the taper with a head pusher and mallet and insert the segmental locking screw (Figure 16). Stem Insertion The implant must be inserted in the same manner as the trial stem. Begin at 90 of anteversion, gradually turning the implant into the correct position as it is driven into place. The turn should be started after the tip of the stem passes the isthmus of the upper femur (Figure 17). Fig. 14 Fig. 15 Fig.17 Fig
14 Bolt Hole Preparation With the implant in place, the need for a trochanteric bolt or claw can be determined. With an anterior-lateral approach, the bolt hole is prepared by using a 3 /8" drill bit from medial to lateral through the greater trochanter (Figure 18). With a posterior approach, the bolt hole can be prepared using the drill guide which allows drilling from lateral to medial (Figure 19). The correct length of the plate or lateral claw connector can be determined by utilizing the bolt depth gauge. The bolt depth gauge is placed medially through the implant and greater trochanter and the correct distance is measured (Figure 20). Note: All the medial bolts are the same lengths to ensure that the union between the plate or lateral claw connector and the bolt are contained within the housing of the calcar prosthesis. Fig. 18 Fig Fig. 20
15 Bolt Assembly To assemble, the medial bolt is held with finger pressure as the correct length plate is tightened laterally with a 5.0mm T-handle (Figure 21). The medial bolt is oblong in shape to help prevent rotation within the bolt hole (Figure 22). The plate should be screwed down until it is tightened in place (Figure 23). Note: In cases where the trochanter is porotic, care should be taken in tightening. Claw Assembly The claw is available in two sizes. Each claw provides multiple positions where the lateral claw connector can be placed to adjust for optimal claw height. To assemble, the medial bolt is held with finger pressure as the correct length lateral claw connector is tightened laterally with a 5.0mm T-handle (Figure 24). The lateral claw connector incorporates five lengths to attach with the medial oblong bolt. Fig. 21 Final Trial Reduction After insertion of the modular calcar prosthesis, either with or without the optional trochanteric fixation, a final trial reduction is performed. Prosthetic stability, leg length assessment, and femoral offset must be evaluated at this time. Once the correct length head is determined, impact the final femoral head (Figure 25). Fig. 22 Fig. 23 Fig. 24 Fig
16 Mallory/Head Modular Calcar Components Horizontal Offset Horizontal Offset Horizontal Offset Vertical Offset Vertical Offset 34mm 45mm 55mm Vertical Offset Mallory/Head Modular Calcar Metaphyseal Segments Neck Angle mm A B C D E Stem Length Length to Center of Standard Head Modular Head Options CoCr Zirconia Zirconia/CoCr 28mm Neck (-6) Zirconia/CoCr 29mm Neck (-5) 31mm Neck (-3) CoCr 34mm Neck (std) Each proximal resection level may be coupled with three distal stem options for stem lengths of 170, 220, and 250mm. 37mm Neck (+3) 40mm Neck (+6) 43mm Neck (+9) 46mm Neck (+12) 14
17 Mallory/Head Modular Calcar Dimensions & Offsets 34mm Femoral Resection Level Size Neck Angle Horiz. Offset/28mm Vert. Offset/28mm Neck Length/28mm -6-3 STD STD STD A B C D E mm Femoral Resection Level Size Neck Angle Horiz. Offset/28mm Vert. Offset/28mm Neck Length/28mm -6-3 STD STD STD A B C D E mm Femoral Resection Level Size Neck Angle Horiz. Offset/28mm Vert. Offset/28mm Neck Length/28mm -6-3 STD STD STD A B C D E Overall Lengths to Center of Standard Head (28mm) Stem Length (mm) Resection Level mm mm mm
18 Biomet Orthopedics, Inc P.O. Box 587 Date: 09/05 56 East Bell Drive Warsaw, Indiana USA 16 Biomet Hip Joint Replacement Prostheses Attention Operating Surgeon DESCRIPTION Biomet manufactures a variety of hip joint replacement prostheses. Hip joint replacement components include: femoral stems, femoral heads, acetabular shells, and acetabular liners. Components are available in a variety of designs and size ranges intended for both primary and revision applications. Specialty components are available including: acetabular screws, centering sleeves, and canal plugs. Materials Femoral Stems Femoral Heads Acetabular Shells Acetabular Liners Acetabular Screws Centering Sleeves Canal Plugs Porous Coating CoCrMo Alloy or Titanium Alloy CoCrMo Alloy Titanium Alloy Ultra-High Molecular Weight Polyethylene (UHMWPE) Titanium Alloy Polymethylmethacrylate (PMMA) UHMWPE Titanium Alloy INDICATIONS 1. Noninflammatory degenerative joint disease including osteoarthritis and avascular necrosis. 2. Rheumatoid arthritis. 3. Correction of functional deformity. 4. Treatment of non-union, femoral neck fracture, and trochanteric fractures of the proximal femur with head involvement, unmanageable using other techniques. 5. Revision procedures where other treatment or devices have failed. Patient selection factors to be considered include: 1) need to obtain pain relief and improve function, 2) ability and willingness of the patient to follow instructions, including control of weight and activity level, 3) a good nutritional state of the patient, and 4) the patient must have reached full skeletal maturity. CONTRAINDICATIONS Absolute contraindications include: infection, sepsis, and osteomyelitis. Relative contraindications include: 1) uncooperative patient or patient with neurologic disorders who are incapable of following directions, 2) osteoporosis, 3) metabolic disorders which may impair bone formation, 4) osteomalacia, 5) distant foci of infections which may spread to the implant site, 6) rapid joint destruction, marked bone loss or bone resorption apparent on roentgenogram, 7) vascular insufficiency, muscular atrophy, or neuromuscular disease. WARNINGS Improper selection, placement, positioning, alignment, and fixation of the implant components may result in unusual stress conditions which may lead to subsequent reduction in the service life of the prosthetic components. Malalignment of the components or inaccurate implantation can lead to excessive wear and/or failure of the implant or procedure. Inadequate preclosure cleaning (removal of surgical debris) can lead to excessive wear. Improper preoperative or intraoperative implant handling or damage (scratches, dents, etc.) can lead to crevice corrosion, fretting, fatigue fracture, and/or excessive wear. Do not modify implants. The surgeon is to be thoroughly familiar with the implants and instruments, prior to performing surgery. 1. Use Biomet femoral and modular head component with appropriate matching Type I Taper, Type II Taper, or 12/14 Taper. 2. Firmly seat modular head components to prevent dissociation. Thoroughly clean and dry taper prior to attachment of the modular head component to avoid crevice corrosion and improper seating. 3. Acetabular screws are to be fully seated to assure stable fixation and to avoid interference with the acetabular liner component. 4. Prior to seating the liner into the shell component, all surgical debris (tissue fragments, etc.) must be removed from the interior of the shell component, as debris may inhibit the locking mechanism from engaging and securing the liner into the shell component. 5. Perforation entirely through the pelvic bone with dome fixation screws or rim screws is to be completely avoided. Caution is to be used when determining and selecting the length of screws to be used, as perforation through the pelvic bone with screws that are too long can cause damage to body structures (blood vessels, etc.) located on the interior side of the pelvis. 6. Tight fixation of all non-cemented components at the time of surgery is critical to the success of the procedure. Each component must properly press fit into the host bone which necessitates precise operative technique and the use of specified instruments. Bone stock of adequate quality must be present and appraised at the time of surgery. 7. Care is to be taken to assure complete support of all parts of the device embedded in bone cement to prevent stress concentrations, which may lead to failure of the procedure. Complete preclosure cleaning and removal of bone cement debris, metallic debris and other surgical debris at the implant site is critical to minimize wear of the implant articular surfaces. Implant fracture due to cement failure has been reported. 8. Acetabular shells should only be used with compatible FDA cleared acetabular liners. Biomet joint replacement prostheses provide the surgeon with a means of reducing pain and restoring function for many patients. While these devices are generally successful in attaining these goals they cannot be expected to withstand the activity levels and loads of normal, healthy bone and joint tissue. Accepted practices in postoperative care are important. Failure of the patient to follow postoperative care instructions involving rehabilitation can compromise the success of the procedure. The patient is to be advised of the limitation of the reconstruction and the need for protection of the implants from full load bearing until adequate fixation and healing have occurred. Excessive activity, trauma, and weight gain have been implicated with premature failure of the implant by loosening, fracture, and/or wear. Loosening of the implants can result in increased production of wear particles, as well as accelerate damage to bone making successful revision surgery more difficult. The patient is to be made aware and warned of general surgical risks, possible adverse effects as listed, and to follow the instructions of the treating physician including follow-up visits. PRECAUTIONS Specialized instruments are designed for Biomet joint replacement systems to aid in the accurate implantation of the prosthetic components. The use of instruments or implant components from other systems can result in inaccurate fit, sizing, excessive wear, and device failure. Intraoperative fracture or breaking of instruments has been reported. Surgical instruments are subject to wear with normal usage. Instruments, which have experienced extensive use or excessive force, are susceptible to fracture. Surgical instruments should only be used for their intended purpose. Biomet recommends that all instruments be regularly inspected for wear and disfigurement. Do not reuse implants. While an implant may appear undamaged, previous stress may have created imperfections that would reduce the service life of the implant. Do not treat patients with implants that have been, even momentarily, placed in a different patient. POSSIBLE ADVERSE EFFECTS 1. Material sensitivity reactions. Implantation of foreign material in tissues can result in histological reactions involving various sizes of macrophages and fibroblasts. The clinical significance of this effect is uncertain, as similar changes may occur as a precursor to or during the healing process. Particulate wear debris and discoloration from metallic and polyethylene components of joint implants may be present in adjacent tissue or fluid. It has been reported that wear debris may initiate a cellular response resulting in osteolysis or osteolysis may be a result of loosening of the implant. 2. Early or late postoperative infection and allergic reaction. 3. Intraoperative bone perforation or fracture may occur, particularly in the presence of poor bone stock caused by osteoporosis, bone defects from previous surgery, bone resorption, or while inserting the device.
19 4. Loosening or migration of the implants can occur due to loss of fixation, trauma, malalignment, bone resorption, and excessive activity. 5. Periarticular calcification or ossification, with or without impediment of joint mobility. 6. Inadequate range of motion due to improper selection or positioning of components. 7. Undesirable shortening of limb. 8. Dislocation and subluxation due to inadequate fixation and improper positioning. Muscle and fibrous tissue laxity can also contribute to these conditions. 9. Fatigue fracture of component can occur as a result of loss of fixation, strenuous activity, malalignment, trauma, non-union, or excessive weight. 10. Fretting and crevice corrosion can occur at interfaces between components. 11. Wear and/or deformation of articulating surfaces. 12. Trochanteric avulsion or non-union as a result of excess muscular tension, early weight bearing, or inadequate reattachment. 13. Problems of the knee or ankle of the affected limb or contralateral limb aggravated by leg length discrepancy, too much femoral medialization or muscle deficiencies. 14. Postoperative bone fracture and pain. STERILITY Prosthetic components are sterilized by exposure to a minimum dose of 25 kgy of gamma radiation. Do not resterilize. Do not use any component from an opened or damaged package. Do not use implants after expiration date. Caution: Federal law (USA) restricts this device to sale by or on the order of a physician. Comments regarding this device can be directed to Attn: Regulatory Dept., Biomet, Inc., P.O. Box 587, Warsaw, IN USA, Fax Authorized Representative: 0086 Biomet, U.K., Ltd. Waterton Industrial Estates, Bridgend, South Wales CF31 3XA U.K. The information contained in this package insert was current on the date this brochure was printed. However, the package insert may have been revised after that date. To obtain a current package insert, please contact Biomet at the contact information provided herein. References 1. Head,W.C.; Emerson R.H. Jr.; and Higgins L.L.: A Titanium Cementless Calcar Replacement Prosthesis in Revision Surgery of the Femur: 13 Year Experience. J. of Arthroplasty, 16: 183 7, Data on file at Biomet. This material is intended for the sole use and benefit of the Biomet sales force and physicians. It is not to be redistributed, duplicated, or disclosed without the express written consent of Biomet. Mallory/Head, STS, and PPS are trademarks of Biomet Manufacturing Corp. 17
20 DrivenByEngineering P.O. Box 587, Warsaw, IN ext Biomet Orthopedics, Inc. All Rights Reserved Form No. Y-BMT-982/073106/K
Positioning Sleeve Surgical Technique
 Positioning Sleeve Surgical Technique Surgical Technique The Comprehensive Fracture System offers a unique tool to evaluate and re-establish humeral height, which can be critical to restoring proper biomechanics
Positioning Sleeve Surgical Technique Surgical Technique The Comprehensive Fracture System offers a unique tool to evaluate and re-establish humeral height, which can be critical to restoring proper biomechanics
Taperloc Complete Hip System. Surgical Technique
 Taperloc Complete Hip System Surgical Technique One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized care to one patient. The science and art of medical
Taperloc Complete Hip System Surgical Technique One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized care to one patient. The science and art of medical
BIOLOX delta Option Ceramic Femoral Head System. Product Features and Instructions for Use
 BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Product Features BIOLOX delta ceramic heads are composed of 75% aluminum oxide and approximately 25% zirconia.
BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Product Features BIOLOX delta ceramic heads are composed of 75% aluminum oxide and approximately 25% zirconia.
BIOLOX delta Option Ceramic Femoral Head System
 BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Knees Hips Extremities Cement and Accessories PMI Technology Product Features BIOLOX delta ceramic heads are composed
BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Knees Hips Extremities Cement and Accessories PMI Technology Product Features BIOLOX delta ceramic heads are composed
WristMotion Wrist Hemiarthroplasty System Instructions for Use
 WristMotion Wrist Hemiarthroplasty System Instructions for Use Description The Arthrosurface WristMotion Wrist Hemiarthroplasty System consists of a contoured capitate articular implant designed to articulate
WristMotion Wrist Hemiarthroplasty System Instructions for Use Description The Arthrosurface WristMotion Wrist Hemiarthroplasty System consists of a contoured capitate articular implant designed to articulate
Arcos Modular Femoral Revision System. Broach and Calcar Proximal Bodies Surgical Technique
 Arcos Modular Femoral Revision System Broach and Calcar Proximal Bodies Surgical Technique Table of Contents Pre-operative Planning...2 Patient Positioning and Surgical Approach...2 Removal of a Cemented
Arcos Modular Femoral Revision System Broach and Calcar Proximal Bodies Surgical Technique Table of Contents Pre-operative Planning...2 Patient Positioning and Surgical Approach...2 Removal of a Cemented
Active Articulation. Surgical Technique. Dual Mobility Hip System. Knees Hips Extremities Cement and Accessories PMI Trauma Technology
 Active Articulation Dual Mobility Hip System Surgical Technique Knees Hips Extremities Cement and Accessories PMI Trauma Technology Active Articulation Dual Mobility Hip System Contents Preoperative Planning,
Active Articulation Dual Mobility Hip System Surgical Technique Knees Hips Extremities Cement and Accessories PMI Trauma Technology Active Articulation Dual Mobility Hip System Contents Preoperative Planning,
PLR. Proximal Loading Revision Hip System
 PLR Proximal Loading Revision Hip System The PLR splined revision stem is designed to recreate the natural stresses in the revised femur, where proximal bone may be compromised. PLR Hip System Design Considerations
PLR Proximal Loading Revision Hip System The PLR splined revision stem is designed to recreate the natural stresses in the revised femur, where proximal bone may be compromised. PLR Hip System Design Considerations
Arcos Interlocking Distal Stem. Surgical Technique Addendum to the Arcos Modular Femoral Revision System
 Arcos Interlocking Distal Stem Surgical Technique Addendum to the Arcos Modular Femoral Revision System One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized
Arcos Interlocking Distal Stem Surgical Technique Addendum to the Arcos Modular Femoral Revision System One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized
Description Materials Indications Patient selection factors to be considered include: Contraindications Absolute contraindications include:
 Description The Patello-Femoral Wave Arthroplasty Systems incorporate a distal femoral trochlear surface articular component that mates to a taper post via a taper interlock, and an all-polyethylene patella
Description The Patello-Femoral Wave Arthroplasty Systems incorporate a distal femoral trochlear surface articular component that mates to a taper post via a taper interlock, and an all-polyethylene patella
Talar Dome System Surgical Technique
 Talar Dome System Surgical Technique CAP TALAR DOME RESURFACING HEMIARTHROPLASTY IMPLANT Surgical Technique Guide Description The HemiCAP Contoured Articular Prosthetic incorporates an articular resurfacing
Talar Dome System Surgical Technique CAP TALAR DOME RESURFACING HEMIARTHROPLASTY IMPLANT Surgical Technique Guide Description The HemiCAP Contoured Articular Prosthetic incorporates an articular resurfacing
Hip Resurfacing System
 Hip Resurfacing System The Arthrosurface HemiCAP Hip Hemiarthroplasty System restores the articular surface geometry of the femoral head and preserves functional structures using an innovative 3 dimensional
Hip Resurfacing System The Arthrosurface HemiCAP Hip Hemiarthroplasty System restores the articular surface geometry of the femoral head and preserves functional structures using an innovative 3 dimensional
Cementless Tapered Femoral Stem Surgical technique
 Cementless Tapered Femoral Stem Surgical technique Contents Operative summary 4 Pre-operative planning 5 Femoral neck osteotomy 5 Femoral canal preparation 5 Intra-medullary (IM) reamer 6 Sequential rasping
Cementless Tapered Femoral Stem Surgical technique Contents Operative summary 4 Pre-operative planning 5 Femoral neck osteotomy 5 Femoral canal preparation 5 Intra-medullary (IM) reamer 6 Sequential rasping
Optimum implant geometry
 Surgical Technique Optimum implant geometry Extending proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
Surgical Technique Optimum implant geometry Extending proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
Cementless Tapered Femoral Stem Surgical technique
 Cementless Tapered Femoral Stem Surgical technique Contents Operative summary 4 Pre-operative planning 5 Femoral neck osteotomy 5 Femoral canal preparation 5 Intra-medullary (IM) reamer 6 Sequential rasping
Cementless Tapered Femoral Stem Surgical technique Contents Operative summary 4 Pre-operative planning 5 Femoral neck osteotomy 5 Femoral canal preparation 5 Intra-medullary (IM) reamer 6 Sequential rasping
Pinit Plate Small Bone Fusion System Bone Plate & Screw System
 Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
Regenerex Primary Tibial Tray
 Cruciate Retaining and Posterior Stabilized Surgical Technique Addendum to the Vanguard Complete Knee System Knees Hips Extremities Cement and Accessories PMI Technology Contents Microplasty Knee Instrumentation
Cruciate Retaining and Posterior Stabilized Surgical Technique Addendum to the Vanguard Complete Knee System Knees Hips Extremities Cement and Accessories PMI Technology Contents Microplasty Knee Instrumentation
Lapidus Arthrodesis System Instructions for Use
 Lapidus Arthrodesis System Instructions for Use Description The AlignMATE Lapidus Arthrodesis System consists of bone plates and bone screws (locking, non-locking and interfragmentary), which are intended
Lapidus Arthrodesis System Instructions for Use Description The AlignMATE Lapidus Arthrodesis System consists of bone plates and bone screws (locking, non-locking and interfragmentary), which are intended
Bone Preservation Stem
 TRI-LOCK Bone Preservation Stem Featuring GRIPTION Coating Surgical Technique Implant Geometry Extending the TRI-LOCK Stem heritage The original TRI-LOCK Stem was introduced in 1981. This implant was
TRI-LOCK Bone Preservation Stem Featuring GRIPTION Coating Surgical Technique Implant Geometry Extending the TRI-LOCK Stem heritage The original TRI-LOCK Stem was introduced in 1981. This implant was
CAUTION: Ceramic liners are not approved for use in the United States.
 Total Hip Prostheses, Self-Centering Hip Prostheses and Hemi-Hip Prostheses IMPORTANT: This essential product information sheet does not include all of the information necessary for selection and use of
Total Hip Prostheses, Self-Centering Hip Prostheses and Hemi-Hip Prostheses IMPORTANT: This essential product information sheet does not include all of the information necessary for selection and use of
SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS
 SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS INTRODUCTION The Summit Tapered Hip System s comprehensive set of implants and instruments
SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS INTRODUCTION The Summit Tapered Hip System s comprehensive set of implants and instruments
ACETABULAR CUP SURGICAL TECHNIQUE
 ACETABULAR CUP SURGICAL TECHNIQUE ACETABULAR CUP DEVICE INDICATIONS FOR USE The ICONACY I-Hip total hip replacement is indicated for the following conditions: 1. A severely painful and/or disabled hip
ACETABULAR CUP SURGICAL TECHNIQUE ACETABULAR CUP DEVICE INDICATIONS FOR USE The ICONACY I-Hip total hip replacement is indicated for the following conditions: 1. A severely painful and/or disabled hip
TaperFill. Surgical Technique
 TaperFill Surgical Technique Table of Contents Indications and Contraindications 3 TaperFill Hip Size Charts 4-5 DJO Surgical 9800 Metric Boulevard Austin, TX (800) 456-8696 www.djosurgical.com Preoperative
TaperFill Surgical Technique Table of Contents Indications and Contraindications 3 TaperFill Hip Size Charts 4-5 DJO Surgical 9800 Metric Boulevard Austin, TX (800) 456-8696 www.djosurgical.com Preoperative
Technique Guide Hip Resurfacing System
 Technique Guide Hip Resurfacing System Approximately 5%-18% of all hip arthroplasties are completed on patients with a primary diagnosis of osteonecrosis. Patients are generally younger adults age 35 years
Technique Guide Hip Resurfacing System Approximately 5%-18% of all hip arthroplasties are completed on patients with a primary diagnosis of osteonecrosis. Patients are generally younger adults age 35 years
Technique Guide Small Bone Fusion System
 Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Answer. Hip System Design Features:
 Table of Contents Alliance Revision Balance Stanmore Bio-Moore II Taperloc Mallory-Head P.O. Box 587, Warsaw, IN 46581-0587 574.267.6639 2003 Biomet, Inc. All Rights Reserved web site: www.biomet.com email:
Table of Contents Alliance Revision Balance Stanmore Bio-Moore II Taperloc Mallory-Head P.O. Box 587, Warsaw, IN 46581-0587 574.267.6639 2003 Biomet, Inc. All Rights Reserved web site: www.biomet.com email:
SURGICAL TECHNIQUE. Alpine Cementless Hip Stem
 SURGICAL TECHNIQUE Alpine Cementless Hip Stem The following technique is a general guide for the instrumentation of the Alpine Cementless Hip Stem. It is expected that the surgeon is already familiar with
SURGICAL TECHNIQUE Alpine Cementless Hip Stem The following technique is a general guide for the instrumentation of the Alpine Cementless Hip Stem. It is expected that the surgeon is already familiar with
Progeny Hip Stem. Surgical Protocol and Product Specifications
 Progeny Hip Stem Surgical Protocol and Product Specifications Progeny Hip Stem Introduction With emphasis on maximum stability and ease of use, the StelKast ProgenyTM Hip System provides the surgeon with
Progeny Hip Stem Surgical Protocol and Product Specifications Progeny Hip Stem Introduction With emphasis on maximum stability and ease of use, the StelKast ProgenyTM Hip System provides the surgeon with
Arcos Modular Femoral Revision System
 Arcos Modular Femoral Revision System Arcos System Simplify the Complex The Arcos Modular Femoral Revision System meets the demands of complex hip revision surgery by offering surgeons and OR staff the
Arcos Modular Femoral Revision System Arcos System Simplify the Complex The Arcos Modular Femoral Revision System meets the demands of complex hip revision surgery by offering surgeons and OR staff the
Clinical Evaluation Surgical Technique
 Clinical Evaluation Surgical Technique Table of Contents EMPERION Specifications 3 EMPERION Surgical Technique 9 EMPERION Catalog 18 Nota Bene: This technique description herein is made available to the
Clinical Evaluation Surgical Technique Table of Contents EMPERION Specifications 3 EMPERION Surgical Technique 9 EMPERION Catalog 18 Nota Bene: This technique description herein is made available to the
Document No Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE
 Document No. 0400-0205 Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE Revision ECO Date ECO Summary of Changes Deann Rector
Document No. 0400-0205 Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE Revision ECO Date ECO Summary of Changes Deann Rector
Technique Guide Wrist Hemiarthroplasty System
 Technique Guide Wrist Hemiarthroplasty System The WristMotion TM Wrist Hemiarthroplasty System restores mobility and maintains native biomechanics using a dual curvature HemiCAPITATE implant that locks
Technique Guide Wrist Hemiarthroplasty System The WristMotion TM Wrist Hemiarthroplasty System restores mobility and maintains native biomechanics using a dual curvature HemiCAPITATE implant that locks
FLH /11
 FLH 225 04/11 This publication has been issued by: European Central Marketing Waterton Industrial Estate Bridgend, South Wales CF31 3XA, United Kingdom Tel: +44 (0)1656 655221 Fax: +44 (0)1656 645454 www.biomet.com
FLH 225 04/11 This publication has been issued by: European Central Marketing Waterton Industrial Estate Bridgend, South Wales CF31 3XA, United Kingdom Tel: +44 (0)1656 655221 Fax: +44 (0)1656 645454 www.biomet.com
Encina Taper Stem. Stinson Orthopedics Inc. 303 Twin Dolphin Drive, Suite 600 Redwood City, CA
 Stinson Orthopedics Inc. 303 Twin Dolphin Drive, Suite 600 Redwood City, CA 94065 info@stinsonortho.com www.stinsonortho.com Table of Contents Introduction 3 Features 4 Surgical Technique 5 Preoperative
Stinson Orthopedics Inc. 303 Twin Dolphin Drive, Suite 600 Redwood City, CA 94065 info@stinsonortho.com www.stinsonortho.com Table of Contents Introduction 3 Features 4 Surgical Technique 5 Preoperative
operative technique Kent Hip
 operative technique Kent Hip The Kent Hip Operative Technique The Kent Hip was developed by Mr Cliff Stossel, FRCS in Maidstone, Kent, UK and first implanted in 1986. It was designed to deal with problems
operative technique Kent Hip The Kent Hip Operative Technique The Kent Hip was developed by Mr Cliff Stossel, FRCS in Maidstone, Kent, UK and first implanted in 1986. It was designed to deal with problems
CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician.
 CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH Mpact 3D Metal Implants and Augments 3D Metal INSTRUCTION FOR USE Important notice: the device(s) can
CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH Mpact 3D Metal Implants and Augments 3D Metal INSTRUCTION FOR USE Important notice: the device(s) can
VerSys LD/Fx Cemented and Press-Fit Hip Prostheses. Surgical Technique IMAGE TO COME. Versatile solutions for total and partial hip replacement
 VerSys LD/Fx Cemented and Press-Fit Hip Prostheses Surgical Technique IMAGE TO COME Versatile solutions for total and partial hip replacement VerSys LD/Fx Cemented and Press-Fit Hip Prostheses VerSys
VerSys LD/Fx Cemented and Press-Fit Hip Prostheses Surgical Technique IMAGE TO COME Versatile solutions for total and partial hip replacement VerSys LD/Fx Cemented and Press-Fit Hip Prostheses VerSys
Approach Patients with Confidence
 Surgical Technique Approach Patients with Confidence The ACTIS Total Hip System is the first DePuy Synthes stem specifically designed to be utilized with tissue sparing approaches, such as the anterior
Surgical Technique Approach Patients with Confidence The ACTIS Total Hip System is the first DePuy Synthes stem specifically designed to be utilized with tissue sparing approaches, such as the anterior
ZMR Over-the-Junction Instruments for Revision Hip Arthroplasty. Surgical Technique IMAGE TO COME
 ZMR Over-the-Junction Instruments for Revision Hip Arthroplasty Surgical Technique IMAGE TO COME ZMR Over-the-Junction Instruments for Revision Hip Arthroplasty Introduction The ZMR Over-the-Junction (OTJ)
ZMR Over-the-Junction Instruments for Revision Hip Arthroplasty Surgical Technique IMAGE TO COME ZMR Over-the-Junction Instruments for Revision Hip Arthroplasty Introduction The ZMR Over-the-Junction (OTJ)
Surgical Technique. Hip System
 Surgical Technique Hip System INDICATIONS FOR USE The TaperSet Hip System is designed for total or partial hip arthroplasty and is intended to be used with compatible components of the Consensus Hip System.
Surgical Technique Hip System INDICATIONS FOR USE The TaperSet Hip System is designed for total or partial hip arthroplasty and is intended to be used with compatible components of the Consensus Hip System.
Revision. Hip Stem. Surgical Protocol
 U2 TM Revision Hip Stem Surgical Protocol U2 Revision Hip Stem Table of Contents Introduction... 1 Preoperative Planning... 2 Femoral Preparation... 3 Trial Reduction... 5 Implant Insertion... 6 Ordering
U2 TM Revision Hip Stem Surgical Protocol U2 Revision Hip Stem Table of Contents Introduction... 1 Preoperative Planning... 2 Femoral Preparation... 3 Trial Reduction... 5 Implant Insertion... 6 Ordering
THE NATURAL FIT. Surgical Technique. Hip Knee Spine Navigation
 THE NATURAL FIT Surgical Technique Hip Knee Spine Navigation MiniMAX Surgical Technique Hip Knee Spine Navigation INTRODUCTION The MiniMAX TM is a cementless anatomic stem available in 9 right sizes and
THE NATURAL FIT Surgical Technique Hip Knee Spine Navigation MiniMAX Surgical Technique Hip Knee Spine Navigation INTRODUCTION The MiniMAX TM is a cementless anatomic stem available in 9 right sizes and
Revision A Date:
 Biomet Trauma 56 East Bell Drive P.O. Box 587 Warsaw, Indiana 46581 USA 01-50-4053 Revision A Date: 2016-06 Biomet Unite3D Bridge with OsseoTi Technology ATTENTION OPERATING SURGEON DESCRIPTION Biomet
Biomet Trauma 56 East Bell Drive P.O. Box 587 Warsaw, Indiana 46581 USA 01-50-4053 Revision A Date: 2016-06 Biomet Unite3D Bridge with OsseoTi Technology ATTENTION OPERATING SURGEON DESCRIPTION Biomet
SURGICAL TECHNIQUE. Alpine Cemented Hip Stem
 SURGICAL TECHNIQUE Alpine Cemented Hip Stem The following technique is a general guide for the instrumentation of the Alpine Cemented Hip Stem. It is expected that the surgeon is already familiar with
SURGICAL TECHNIQUE Alpine Cemented Hip Stem The following technique is a general guide for the instrumentation of the Alpine Cemented Hip Stem. It is expected that the surgeon is already familiar with
ADDRESSING CLINICAL ISSUES OF CEMENTLESS HIP ARTHROPLASTY
 E C H E L O N P R I M A R Y H I P S Y S T E M P R O D U C T R A T I O N A L E ADDRESSING CLINICAL ISSUES OF CEMENTLESS HIP ARTHROPLASTY Echelon Primary Total Hip System HIGH OFFSET STANDARD OFFSET Cementless
E C H E L O N P R I M A R Y H I P S Y S T E M P R O D U C T R A T I O N A L E ADDRESSING CLINICAL ISSUES OF CEMENTLESS HIP ARTHROPLASTY Echelon Primary Total Hip System HIGH OFFSET STANDARD OFFSET Cementless
Surgical Technique. Cup System
 Surgical Technique Cup System INDICATIONS AND USAGE Indications for the use of the CS2 ACETABULAR CUP SYSTEM must be carefully considered with respect to the patient s entire evaluation and alternative
Surgical Technique Cup System INDICATIONS AND USAGE Indications for the use of the CS2 ACETABULAR CUP SYSTEM must be carefully considered with respect to the patient s entire evaluation and alternative
Echo Bi-Metric. " Hips " Extremities " Cement and Accessories " PMI " Technology
 Echo Bi-Metric " Hips " Extremities " Cement and Accessories " PMI " Technology Echo Bi-Metric Figure Figure Biomets Echo Hip System Offers Three Stem Variations Forged cobalt chromium alloy Interlok gritblasted
Echo Bi-Metric " Hips " Extremities " Cement and Accessories " PMI " Technology Echo Bi-Metric Figure Figure Biomets Echo Hip System Offers Three Stem Variations Forged cobalt chromium alloy Interlok gritblasted
StelKast Acetabular. Surgical Protocol and Product Specifications. Including ProForm, Provident, Provident Apical Threaded Hole & Systems
 StelKast Acetabular Surgical Protocol and Product Specifications Including ProForm, Provident, Provident Apical Threaded Hole & Systems StelKast Acetabular Surgical Protocol Introduction StelKast offers
StelKast Acetabular Surgical Protocol and Product Specifications Including ProForm, Provident, Provident Apical Threaded Hole & Systems StelKast Acetabular Surgical Protocol Introduction StelKast offers
9800 Metric Blvd. Austin, Texas
 rev. A Encore Orthopedics, Inc. 1998 www.encoremed.com 9800 Metric Blvd. Austin, Texas 78758 512-832-9500 1 contents How to use the Foundation Hip 1 2 Plan your approach Select your hardware Preparing
rev. A Encore Orthopedics, Inc. 1998 www.encoremed.com 9800 Metric Blvd. Austin, Texas 78758 512-832-9500 1 contents How to use the Foundation Hip 1 2 Plan your approach Select your hardware Preparing
Technique Guide KISSloc Suture System
 Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
Biomet Large Cannulated Screw System
 Biomet Large Cannulated Screw System s u r g i c a l t e c h n i q u e A Complete System for Simplified Fracture Fixation 6.5mm & 7.3mm The Titanium, Self-drilling, Self-tapping Large Cannulated Screw
Biomet Large Cannulated Screw System s u r g i c a l t e c h n i q u e A Complete System for Simplified Fracture Fixation 6.5mm & 7.3mm The Titanium, Self-drilling, Self-tapping Large Cannulated Screw
OSS Modular Arthrodesis System. Assembly Guide
 OSS Modular Arthrodesis System Assembly Guide One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized care to one patient. The science and art of medical
OSS Modular Arthrodesis System Assembly Guide One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized care to one patient. The science and art of medical
HIP SYSTEM SURGICAL TECHNIQUE
 HIP SYSTEM SURGICAL TECHNIQUE Introduction...2 Preoperative Planning...3 Preoperative Planning...3 Templating and Radiographs...4 Determination of Leg Length Discrepancy...5 Determining Acetabular Cup
HIP SYSTEM SURGICAL TECHNIQUE Introduction...2 Preoperative Planning...3 Preoperative Planning...3 Templating and Radiographs...4 Determination of Leg Length Discrepancy...5 Determining Acetabular Cup
AML Hip System. Design Rationale/ Surgical Technique
 AML Hip System Design Rationale/ Surgical Technique Design Rationale Evolution In 1977, DePuy Synthes Companies introduced the original cementless total hip. The AML Hip launched in order to solve one
AML Hip System Design Rationale/ Surgical Technique Design Rationale Evolution In 1977, DePuy Synthes Companies introduced the original cementless total hip. The AML Hip launched in order to solve one
Dual Mobility System Evaluation surgical technique
 Trinity Dual Mobility System Evaluation surgical technique Contents Operative summary 4 Overview 5 Operative technique 6 1. Acetabular reaming 6 Reamer guide 6 2. Acetabular shell trial 6 3. Acetabular
Trinity Dual Mobility System Evaluation surgical technique Contents Operative summary 4 Overview 5 Operative technique 6 1. Acetabular reaming 6 Reamer guide 6 2. Acetabular shell trial 6 3. Acetabular
Approach Patients with Confidence
 Approach Patients with Confidence The is the first stem specifically designed to be utilized with tissue sparing approaches, such as the anterior approach, as well as traditional approaches. The implant
Approach Patients with Confidence The is the first stem specifically designed to be utilized with tissue sparing approaches, such as the anterior approach, as well as traditional approaches. The implant
Resurfacing Distal Femur. Orthopaedic Salvage System
 Resurfacing Distal Femur Orthopaedic Salvage System Primary Arthroplasty OSS 3cm Resurfacing Distal Femur Distal Femoral Resection Drill and ream the distal femur in the following sequence: (Figure 1)
Resurfacing Distal Femur Orthopaedic Salvage System Primary Arthroplasty OSS 3cm Resurfacing Distal Femur Distal Femoral Resection Drill and ream the distal femur in the following sequence: (Figure 1)
RECOVERY. P r o t r u s i o
 RECOVERY P r o t r u s i o TM C a g e RECOVERY P r o t r u s i o TM C a g e Design Features Revision acetabular surgery is a major challenge facing today s total joint revision surgeon. Failed endo/bi-polars,
RECOVERY P r o t r u s i o TM C a g e RECOVERY P r o t r u s i o TM C a g e Design Features Revision acetabular surgery is a major challenge facing today s total joint revision surgeon. Failed endo/bi-polars,
- October 2013 English
 TOTAL HIP JOINT REPLACEMENT FOR CEMENTED APPLICATIONS FOR THE ATTENTION OF THE OPERATING SURGEON Total hip replacement provides the surgeon with a means of restoring mobility and reducing pain with the
TOTAL HIP JOINT REPLACEMENT FOR CEMENTED APPLICATIONS FOR THE ATTENTION OF THE OPERATING SURGEON Total hip replacement provides the surgeon with a means of restoring mobility and reducing pain with the
FLH183 04/08. Biomet UK Ltd Waterton Industrial Estate Bridgend, South Wales CF31 3XA, United Kingdom. Tel. +44 (0) Fax: +44 (0)
 FLH183 04/08 Biomet UK Ltd Waterton Industrial Estate Bridgend, South Wales CF31 3XA, United Kingdom Tel. +44 (0)1656 655221 Fax: +44 (0)1656 645454 Exceed ABT Operative Technique The Exceed ABT TM acetabular
FLH183 04/08 Biomet UK Ltd Waterton Industrial Estate Bridgend, South Wales CF31 3XA, United Kingdom Tel. +44 (0)1656 655221 Fax: +44 (0)1656 645454 Exceed ABT Operative Technique The Exceed ABT TM acetabular
Scan Bi-Polar 22/28. Operative Technique
 Scan Bi-Polar 22/28 Operative Technique Disclaimer This publication and all content, artwork, photographs, names, logos and marks contained in it are protected by copyright, trademarks and other intellectual
Scan Bi-Polar 22/28 Operative Technique Disclaimer This publication and all content, artwork, photographs, names, logos and marks contained in it are protected by copyright, trademarks and other intellectual
ESC. Enhanced Stability Liners. Design Rationale & Surgical Technique
 ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
Precautionary Statement ( )
 Precautionary Statement (21282008) Biomet Sports Medicine, Inc. 21282008 4861 E. Airport Dr. Rev. A Ontario, CA 91761 Date: 06/07 Sleeve and Button Soft Tissue Devices Utilizing ZipLoop Technology ATTENTION
Precautionary Statement (21282008) Biomet Sports Medicine, Inc. 21282008 4861 E. Airport Dr. Rev. A Ontario, CA 91761 Date: 06/07 Sleeve and Button Soft Tissue Devices Utilizing ZipLoop Technology ATTENTION
Echo. Femoral Hip System. Hip Fracture Surgical Technique. Endo II Uni-polar Acetabular Component RingLoc Bi-polar Acetabular Component
 Echo Femoral Hip System Hip Fracture Surgical Technique Endo II Uni-polar Acetabular Component RingLoc Bi-polar Acetabular Component One Surgeon. One Patient. Over 1 million times per year, Biomet helps
Echo Femoral Hip System Hip Fracture Surgical Technique Endo II Uni-polar Acetabular Component RingLoc Bi-polar Acetabular Component One Surgeon. One Patient. Over 1 million times per year, Biomet helps
OSS Orthopedic Salvage System Compress Device. Surgical Technique
 OSS Orthopedic Salvage System Compress Device Surgical Technique Table of Contents Quick Reference Guide... 2 Device Description... 3 Resection... 4 No Face Adapter Provisional Construct... 4 Canal Preparation...
OSS Orthopedic Salvage System Compress Device Surgical Technique Table of Contents Quick Reference Guide... 2 Device Description... 3 Resection... 4 No Face Adapter Provisional Construct... 4 Canal Preparation...
U2 PSA. Revision Knee. Surgical Protocol
 U2 PSA TM Revision Knee Surgical Protocol Table of Contents 1 Component Removal... 1 2 Tibial Preparation... 1 2.1 Tibial Canal Preparation... 1 2.2 Proximal Tibial Resection... 2 2.3 Non Offset Tibial
U2 PSA TM Revision Knee Surgical Protocol Table of Contents 1 Component Removal... 1 2 Tibial Preparation... 1 2.1 Tibial Canal Preparation... 1 2.2 Proximal Tibial Resection... 2 2.3 Non Offset Tibial
Fixed and Variable Geometry Total Shoulder Arthroplasty
 Fixed and Variable Geometry Total Shoulder Arthroplasty RECOVERY FUNCTION SURVIVORSHIP DePuy believes in an approach to total shoulder replacement that places equal importance on recovery, function and
Fixed and Variable Geometry Total Shoulder Arthroplasty RECOVERY FUNCTION SURVIVORSHIP DePuy believes in an approach to total shoulder replacement that places equal importance on recovery, function and
Dual Mobility System Surgical technique
 Trinity Dual Mobility System Surgical technique 2 Contents Operative summary 4 Overview 5 Operative technique 6 1. Acetabular reaming 6 Reamer guide 6 2. Acetabular shell trial 7 3. Acetabular shell implantation
Trinity Dual Mobility System Surgical technique 2 Contents Operative summary 4 Overview 5 Operative technique 6 1. Acetabular reaming 6 Reamer guide 6 2. Acetabular shell trial 7 3. Acetabular shell implantation
MIS Cemented Tibial Component
 MIS Cemented Tibial Component NexGen Complete Knee Solution Surgical Technique Table of Contents Surgical Exposure... 2 Finish the Tibia... 2 Position Based on Anatomic Landmarks... 3 Lateral Posterior
MIS Cemented Tibial Component NexGen Complete Knee Solution Surgical Technique Table of Contents Surgical Exposure... 2 Finish the Tibia... 2 Position Based on Anatomic Landmarks... 3 Lateral Posterior
Important notice: the device(s) can be prescribed and implanted only by a doctor legally authorized to perform this type of surgery.
 rev.10 CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH - EVOLIS/GMK KNEE PROSTHESIS - INSTRUCTIONS FOR USE Important notice: the device(s) can be prescribed
rev.10 CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH - EVOLIS/GMK KNEE PROSTHESIS - INSTRUCTIONS FOR USE Important notice: the device(s) can be prescribed
Following a tradition of success. VerSys Heritage Primary Hip Prosthesis Surgical Technique
 Following a tradition of success VerSys Heritage Primary Hip Prosthesis Surgical Technique VerSys Heritage Primary Hip Prosthesis 1 Surgical Technique For VerSys Heritage Primary Hip Prosthesis Dennis
Following a tradition of success VerSys Heritage Primary Hip Prosthesis Surgical Technique VerSys Heritage Primary Hip Prosthesis 1 Surgical Technique For VerSys Heritage Primary Hip Prosthesis Dennis
Restoration Modular. Revision Hip System Surgical Protocol
 Orthopaedics Restoration Modular Revision Hip System Surgical Protocol Restoration Modular Cone Body/Conical Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
Orthopaedics Restoration Modular Revision Hip System Surgical Protocol Restoration Modular Cone Body/Conical Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
Integral 180 Surgical Technique
 Integral 180 Surgical Technique The Integral 180 and 225 are part of the Alliance Family Total Hip System. The Integral 225 femoral component is marketed for use with bone cement in the United States.
Integral 180 Surgical Technique The Integral 180 and 225 are part of the Alliance Family Total Hip System. The Integral 225 femoral component is marketed for use with bone cement in the United States.
Manza Cup HA SURGICAL TECHNIQUE.
 1 PRE-OPERATIVE PLANNING. Preoperative assessment of the appropriate size and position of the acetabular component will provide intraoperative guidance for acetabular reaming. To determine the acetabluar
1 PRE-OPERATIVE PLANNING. Preoperative assessment of the appropriate size and position of the acetabular component will provide intraoperative guidance for acetabular reaming. To determine the acetabluar
Stinson Orthopedics Inc. 303 Twin Dolphin Drive, Suite 600 Redwood City, CA
 Stinson Orthopedics Inc. 303 Twin Dolphin Drive, Suite 600 Redwood City, CA 94065 info@stinsonortho.com www.stinsonortho.com Encina HA Stem Table of Contents Introduction 3 Encina HA Stem Features 4 Surgical
Stinson Orthopedics Inc. 303 Twin Dolphin Drive, Suite 600 Redwood City, CA 94065 info@stinsonortho.com www.stinsonortho.com Encina HA Stem Table of Contents Introduction 3 Encina HA Stem Features 4 Surgical
EXTENDED TROCHANTERIC OSTEOTOMY SURGICAL TECHNIQUE FPO EXTENSIVELY COATED FIXATION
 EXTENDED TROCHANTERIC OSTEOTOMY SURGICAL TECHNIQUE FPO EXTENSIVELY COATED FIXATION SINCE 1983 PREOPERATIVE PLANNING EXPLANTATION OPTIONS the cement from inside the cement canal until the bone/ cement bond
EXTENDED TROCHANTERIC OSTEOTOMY SURGICAL TECHNIQUE FPO EXTENSIVELY COATED FIXATION SINCE 1983 PREOPERATIVE PLANNING EXPLANTATION OPTIONS the cement from inside the cement canal until the bone/ cement bond
Duraloc CONSTRAINED LINER
 SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
Greater Component Accuracy...
 Greater Component Accuracy... StageOne cement spacer molds are designed to provide the surgeon with choices never before offered in two-stage revision knee surgery for an infected total joint. StageOne
Greater Component Accuracy... StageOne cement spacer molds are designed to provide the surgeon with choices never before offered in two-stage revision knee surgery for an infected total joint. StageOne
Zimmer NexGen MIS Tibial Component. Cemented Surgical Technique IMAGE TO COME
 Zimmer NexGen MIS Tibial Component Cemented Surgical Technique IMAGE TO COME Zimmer NexGen MIS Tibial Component Cemented Surgical Technique 1 Zimmer NexGen MIS Tibial Component Cemented Surgical Technique
Zimmer NexGen MIS Tibial Component Cemented Surgical Technique IMAGE TO COME Zimmer NexGen MIS Tibial Component Cemented Surgical Technique 1 Zimmer NexGen MIS Tibial Component Cemented Surgical Technique
CC TRIO VERSAFITCUP. Surgical Technique. each to their own. Hip Knee Spine Navigation
 VERSAFITCUP CC TRIO each to their own Surgical Technique Hip Knee Spine Navigation Versafitcup CC TRIO Surgical Technique Hip Knee Spine Navigation EACH TO THEIR OWN The Versafitcup CC Trio is a range
VERSAFITCUP CC TRIO each to their own Surgical Technique Hip Knee Spine Navigation Versafitcup CC TRIO Surgical Technique Hip Knee Spine Navigation EACH TO THEIR OWN The Versafitcup CC Trio is a range
Design Rationale. ECHELON Primary Hip System
 Design Rationale ECHELON Primary Hip System ECHELON Primary Total Hip System Addressing clinical issues of cementless hip arthroplasty Cementless total hip arthroplasty has provided a proven method of
Design Rationale ECHELON Primary Hip System ECHELON Primary Total Hip System Addressing clinical issues of cementless hip arthroplasty Cementless total hip arthroplasty has provided a proven method of
28 Surgical Technique
 Surgical Technique 10 12 14 16 18 20 22 24 28 26 Technique described by James L. Guyton, MD Campbell Clinic Memphis, Tennessee James W. Harkess, MD Campbell Clinic Memphis, Tennessee David G. LaVelle,
Surgical Technique 10 12 14 16 18 20 22 24 28 26 Technique described by James L. Guyton, MD Campbell Clinic Memphis, Tennessee James W. Harkess, MD Campbell Clinic Memphis, Tennessee David G. LaVelle,
RESTORATION MODULAR. Surgical Protocol REVISION HIP SYSTEM
 RESTORATION MODULAR REVISION HIP SYSTEM Surgical Protocol Restoration Modular Milled Body/Fluted & Plasma Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
RESTORATION MODULAR REVISION HIP SYSTEM Surgical Protocol Restoration Modular Milled Body/Fluted & Plasma Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
RESTORATION MODULAR. Surgical Protocol REVISION HIP SYSTEM
 RESTORATION MODULAR REVISION HIP SYSTEM Surgical Protocol Restoration Modular Cone Body/Fluted & Plasma Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
RESTORATION MODULAR REVISION HIP SYSTEM Surgical Protocol Restoration Modular Cone Body/Fluted & Plasma Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
Flexible Fragment Fixation. Surgical Technique
 Flexible Fragment Fixation Surgical Technique 2 F 3 Flexible Fragment Fixation The F 3 Fragment Plating System offers low profile, yet strong fixation in a locked plating construct that can be contoured
Flexible Fragment Fixation Surgical Technique 2 F 3 Flexible Fragment Fixation The F 3 Fragment Plating System offers low profile, yet strong fixation in a locked plating construct that can be contoured
Bi-Polar 22.2mm & 28mm System - Operative technique
 Disclaimer Biomet UK Ltd, as the manufacturer of this device, does not practice medicine and does not recommend any particular surgical technique for use on a specific patient. The surgeon who performs
Disclaimer Biomet UK Ltd, as the manufacturer of this device, does not practice medicine and does not recommend any particular surgical technique for use on a specific patient. The surgeon who performs
AVANTEON. Operative Technique & Catalogue Information AVANTEON
 AVANTEON Operative Technique & Catalogue Information AVANTEON H I P S Y S T E M Pre-operative Planning The overall aim of pre-operative planning is to establish anatomical data from the patient to guide
AVANTEON Operative Technique & Catalogue Information AVANTEON H I P S Y S T E M Pre-operative Planning The overall aim of pre-operative planning is to establish anatomical data from the patient to guide
EXACTECH KNEE. Operative Technique ADDENDUM. Metaphyseal Cones. Surgeon focused. Patient driven. TM
 EXACTECH KNEE Operative Technique ADDENDUM Metaphyseal Cones Surgeon focused. Patient driven. TM TABLE OF CONTENTS INTRODUCTION...1 DESCRIPTION...1 DETAILED OPERATIVE TECHNIQUE Initial Preparation and
EXACTECH KNEE Operative Technique ADDENDUM Metaphyseal Cones Surgeon focused. Patient driven. TM TABLE OF CONTENTS INTRODUCTION...1 DESCRIPTION...1 DETAILED OPERATIVE TECHNIQUE Initial Preparation and
Ovation Hip System. Surgical Technique
 Ovation Hip System Surgical Technique Ovation Hip System Surgical Technique Ovation Designing Surgeons: Andrew Petrella, M.D. Lecanto, FL Richard Vlasak, M.D. Gainesville, FL Ovation Tribute Designing
Ovation Hip System Surgical Technique Ovation Hip System Surgical Technique Ovation Designing Surgeons: Andrew Petrella, M.D. Lecanto, FL Richard Vlasak, M.D. Gainesville, FL Ovation Tribute Designing
RESTORATION MODULAR. Surgical Protocol REVISION HIP SYSTEM
 RESTORATION MODULAR REVISION HIP SYSTEM Surgical Protocol Restoration Modular Calcar Body/Fluted & Plasma Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
RESTORATION MODULAR REVISION HIP SYSTEM Surgical Protocol Restoration Modular Calcar Body/Fluted & Plasma Distal Stem Femoral Components Using the Restoration Modular Instrument System Restoration Modular
Tradition Hip Primary Surgical Technique
 Design Rationale Many total hip designs in today s marketplace do not take advantage of the known forces present in the femur. Long term stability of a total hip prosthesis requires an implant design and
Design Rationale Many total hip designs in today s marketplace do not take advantage of the known forces present in the femur. Long term stability of a total hip prosthesis requires an implant design and
A novel cementless option. Zimmer NexGen Trabecular Metal Primary Patella Surgical Technique
 A novel cementless option Zimmer NexGen Trabecular Metal Primary Patella Surgical Technique Zimmer Trabecular Metal Primary Patella 1 Zimmer NexGen Trabecular Metal Primary Patella Surgical Technique
A novel cementless option Zimmer NexGen Trabecular Metal Primary Patella Surgical Technique Zimmer Trabecular Metal Primary Patella 1 Zimmer NexGen Trabecular Metal Primary Patella Surgical Technique
SURGICAL TECHNIQUE GUIDE
 DANGER indicates an imminently hazardous situation which, if not avoided, will result in death or serious injury. WARNING indicates a potentially hazardous situation which, if not avoided, could result
DANGER indicates an imminently hazardous situation which, if not avoided, will result in death or serious injury. WARNING indicates a potentially hazardous situation which, if not avoided, could result
Optimum implant geometry
 Design Rationale Optimum implant geometry Extending proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
Design Rationale Optimum implant geometry Extending proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
Surgical Technique r5.indd 1 12/8/10 10:36 AM
 Surgical Technique The science of simplicity With more than 700,000 implantations and two and a half decades of clinical success, the Corail Total Hip System now has the most extensive experience with
Surgical Technique The science of simplicity With more than 700,000 implantations and two and a half decades of clinical success, the Corail Total Hip System now has the most extensive experience with
Comprehensive Reverse Shoulder System Augmented Baseplate
 Comprehensive Reverse Shoulder System Augmented Baseplate Surgical Technique Addendum FPO 1 Comprehensive Reverse Shoulder System Augmented Baseplate Surgical Technique Addendum Adaptive Building on the
Comprehensive Reverse Shoulder System Augmented Baseplate Surgical Technique Addendum FPO 1 Comprehensive Reverse Shoulder System Augmented Baseplate Surgical Technique Addendum Adaptive Building on the
Signature Personalized Patient Care
 Surgical Technique Acetabular Guide System Contents One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized care to one patient. The science and art of medical
Surgical Technique Acetabular Guide System Contents One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized care to one patient. The science and art of medical
Preoperative Planning. The primary objectives of preoperative planning are to:
 Preoperative Planning The primary objectives of preoperative planning are to: - Determine preoperative leg length discrepancy. - Assess acetabular component size and placement. - Determine femoral component
Preoperative Planning The primary objectives of preoperative planning are to: - Determine preoperative leg length discrepancy. - Assess acetabular component size and placement. - Determine femoral component
Pre-Operative Planning. Positioning of the Patient
 Surgical Technique Pre-Operative Planning Decide upon the size and angle of the barrel plate to be used from measuring the x-rays. To maximise the sliding action when using shorter lag screws, the Short
Surgical Technique Pre-Operative Planning Decide upon the size and angle of the barrel plate to be used from measuring the x-rays. To maximise the sliding action when using shorter lag screws, the Short
HELIOS h i p s y s t e m
 HELIOS h i p s y s t e m Design The Helios stem is a highly polished, High Nitrogen Stainless Steel (ISO5832-9) dual tapered cemented stem. The design of the stem is based on the clinically lly successful
HELIOS h i p s y s t e m Design The Helios stem is a highly polished, High Nitrogen Stainless Steel (ISO5832-9) dual tapered cemented stem. The design of the stem is based on the clinically lly successful
