|
|
|
- Reynold Eaton
- 6 years ago
- Views:
Transcription
1 Stefania Bracci, Magdalena Ietswaart, Marius V. Peelen and Cristiana Cavina-Pratesi J Neurophysiol 103: , First published Apr 14, 2010; doi: /jn You might find this additional information useful... Supplemental material for this article can be found at: This article cites 49 articles, 18 of which you can access free at: This article has been cited by 1 other HighWire hosted article: Functional Magnetic Resonance Imaging Reveals the Neural Substrates of Arm Transport and Grip Formation in Reach-to-Grasp Actions in Humans C. Cavina-Pratesi, S. Monaco, P. Fattori, C. Galletti, T. D. McAdam, D. J. Quinlan, M. A. Goodale and J. C. Culham J. Neurosci., August 4, 2010; 30 (31): [Abstract] [Full Text] [PDF] Updated information and services including high-resolution figures, can be found at: Additional material and information about Journal of Neurophysiology This information is current as of October 5, can be found at: Journal of Neurophysiology publishes original articles on the function of the nervous system. It is published 12 times a year (monthly) by the American Physiological Society, 9650 Rockville Pike, Bethesda MD Copyright 2010 by the American Physiological Society. ISSN: , ESSN: Visit our website at
2 J Neurophysiol 103: , First published April 14, 2010; doi: /jn Dissociable Neural Responses to Hands and Non-Hand Body Parts in Human Left Extrastriate Visual Cortex Stefania Bracci, 1 Magdalena Ietswaart, 1 Marius V. Peelen, 3,4 and Cristiana Cavina-Pratesi 2 1 School of Psychology and Sport Sciences, Northumbria University, Newcastle upon Tyne; 2 Durham University, Durham, United Kingdom; 3 Center for Mind/Brain Sciences (CIMEC), University of Trento, Rovereto, Italy; and 4 Department of Psychology, Harvard University, Cambridge, Massachusetts Submitted 2 March 2010; accepted in final form 7 April 2010 Bracci S, Ietswaart M, Peelen MV, Cavina-Pratesi C. Dissociable neural responses to hands and non-hand body parts in human left extrastriate visual cortex. J Neurophysiol 103: , First published April 14, 2010; doi: /jn Accumulating evidence points to a map of visual regions encoding specific categories of objects. For example, a region in the human extrastriate visual cortex, the extrastriate body area (EBA), has been implicated in the visual processing of bodies and body parts. Although in the monkey, neurons selective for hands have been reported, in humans it is unclear whether areas selective for individual body parts such as the hand exist. Here, we conducted two functional MRI experiments to test for hand-preferring responses in the human extrastriate visual cortex. We found evidence for a hand-preferring region in left lateral occipitotemporal cortex in all 14 participants. This region, located in the lateral occipital sulcus, partially overlapped with left EBA, but could be functionally and anatomically dissociated from it. In experiment 2, we further investigated the functional profile of hand- and bodypreferring regions by measuring responses to hands, fingers, feet, assorted body parts (arms, legs, torsos), and non-biological handlike stimuli such as robotic hands. The hand-preferring region responded most strongly to hands, followed by robotic hands, fingers, and feet, whereas its response to assorted body parts did not significantly differ from baseline. By contrast, EBA responded most strongly to body parts, followed by hands and feet, and did not significantly respond to robotic hands or fingers. Together, these results provide evidence for a representation of the hand in extrastriate visual cortex that is distinct from the representation of other body parts. INTRODUCTION Address for reprint requests and other correspondence: S. Bracci, School of Psychology and Sport Sciences, Northumbria University, Newcastle upon Tyne, UK ( stefanie.bracci@gmail.com). Social interactions are at the core of our common everyday experience. Identifying other people based on their facial and bodily features and understanding their behaviors and intentions from their movements are highly complex yet mostly effortless tasks (Van Overwalle and Baetens 2009). Within the human cortex several visual areas have been found to be selective for static depictions of human physical features. While the fusiform face area (FFA; Kanwisher et al. 1997; Puce et al. 1996) in the lateral fusiform gyrus and the occipital face area (OFA; Rossion et al. 2003) in the ventral occipital lobe encode visual aspects of human faces, the extrastriate body area (EBA; Downing et al. 2001) in the lateral occipitotemporal cortex and the fusiform body area (FBA; Peelen and Downing 2005) in the lateral fusiform gyrus encode whole bodies and body parts. Converging evidence from lesion and transcranial magnetic stimulation studies indicate that ventrotemporal and lateral occipitotemporal cortices are indeed causally associated with face and body discrimination (Barton 2003; Damasio et al. 1982; Moro et al. 2008; Pitcher et al. 2009; Steeves et al. 2009; Urgesi et al. 2007). One body part in particular, the hand, is of great significance in daily life. For example, while communicating, we use hands to emphasize our speech and to direct people s attention. Moreover, while learning how to use new tools, we closely observe how the model s hands manipulate the object. More crucially, in action execution, on-line visual control is applied to monitor and correct the direction of the hand, even when performing simple reach-to-grasp movements. Humans outstanding manual dexterity is an important capability that sets humans apart from other species (Napier 1956). Considering the unique role played by the hand in our capacity to interact with the external world, the question arises as to whether the hand may be selectively represented in the human visual cortex. Pioneering neurophysiological studies in the inferior temporal (IT) cortex of macaque monkeys (Desimone et al. 1984; Gross et al. 1969) have provided evidence for neurons selectively responding to the vision of the hand. In humans, evidence for hand-selective neural responses is limited, with studies mostly focusing on investigating the selectivity of assorted body parts or whole bodies rather than individual body parts (Peelen and Downing 2007). Nonetheless, initial evidence for hand selectivity comes from functional magnetic resonance imaging (fmri; Op de Beeck et al. 2010) and event-related potential (ERP; McCarthy et al. 1999) studies demonstrating higher responses to hands compared with that to torsos or faces in restricted parts of the human occipitotemporal cortex. Furthermore, it was shown that distributed fmri response patterns in multiple regions of visual cortex discriminated between hands and torsos (Op de Beeck et al. 2010). These results suggest the existence of hand-selective responses in human visual cortex, although further investigations that include additional body and nonbody control conditions are needed to strengthen the evidence for this hypothesis. Furthermore, it is unknown how putative hand-selective responses are related, both functionally and anatomically, to known bodyselective responses in EBA. Finally, response properties of a putative human hand-selective region remain largely unknown. Here, we present two experiments providing evidence for hand-preferring fmri responses in human visual cortex. In experiment 1 we measured fmri responses to hands, whole bodies, assorted body parts, chairs, and handheld tools. This experiment allowed us to test for hand-preferring responses /10 Copyright 2010 The American Physiological Society 3389
3 3390 BRACCI, IETSWAART, PEELEN, AND CAVINA-PRATESI relative to both body and nonbody control conditions. It also allowed for a direct comparison of hand-preferring and known body-preferring responses in EBA. In experiment 2 we further investigated the functional profile of hand- and body-preferring regions by measuring responses to hands, fingers, feet, assorted body parts (arms, legs, torsos), and nonbiological handlike stimuli, such as robotic hands. In addition, this experiment tested for response selectivity to other non-hand body parts (feet, fingers). METHODS Participants Fifteen naive volunteers with no history of neurological diseases took part in experiment 1. Fourteen of the volunteers also took part in experiment 2. All subjects were right-handed as assessed by the Edinburgh Handedness Inventory (Oldfield 1971). Due to excessive head motion one subject was excluded from the data analyses. Participants gave informed consent and the study was approved by the Ethical Committees of The School of Psychology and Sport Sciences of Northumbria University and Newcastle Magnetic Resonance Centre, School of Clinical Medical Sciences, University of Newcastleon-Tyne. Material and apparatus For each stimulus category we presented 65 different grayscale photographs on a white background. Stimulus presentation was controlled by a PC computer running Psychophysics Toolbox package (Brainard 1997) in Matlab (The MathWorks, Natick, MA). Pictures were projected (Canon Xeed SX6 projector) onto a screen located at the foot of the scanner bed and were viewed through a mirror mounted on the head coil. Experimental design Each experiment consisted of two runs lasting 7 min 14 s, corresponding to 217 functional volumes. Each functional run comprised 25 stimulus blocks and 6 fixation blocks. Each stimulus was presented only once within a run. Runs were organized into quasi-random sequences of five stimulus blocks (one for each stimulus category) followed by periods of fixation. In experiment 1 stimulus categories were: hands, whole bodies, body parts, tools, and chairs. In experiment 2 stimulus categories were: body parts, hands, fingers, feet, and robotic hands. Whole body stimuli did not have heads but they did have hands (this choice was made to keep the concept of whole body intact). Body-part stimuli did not include either feet or hands. Hand stimuli depicted both left and right hands in equal amounts and were captured in a resting position viewed from a third-person perspective. In only 4 of 65 pictures the hand posture could imply some sort of grasping pose. Robotic hands were recruited from Internet websites and only a subgroup showed grasping postures (18 of 65). Within each block, stimuli were presented for 800 ms, with a blank interstimulus interval (ISI) of 200 ms for a total block time of 14 s. Subjects were asked 1) to fixate toward the center of the screen during the whole scan and 2) to perform a one-back task by pressing a button with the index finger any time the same image was presented twice in a row (within each block one or two images were randomly repeated). Imaging data acquisition All images were acquired using a Phillips Achieva 3T scanner with a SENSE standard 8-channel birdcage head coil. The functional gradient-echo echoplanar T2*-weighted images (EPI) with BOLD (blood oxygenation level dependent) contrast were acquired using a repetition time (TR) of 2 s, echo time (TE) of 30 ms, film aperture (FA) of 90, field of view (FOV) of 192, and a matrix size of pixels. Each functional image consisted of 30 axial slices (4 mm thickness with no gap), which covered the whole cerebral cortex. T1-weighted anatomical scans were collected using TR of 9.6, TE of 4.6, FA of 8, FOV 256, and a matrix of pixels. We collected 180 slices of 1.0 mm thickness. Imaging data: preprocessing and analysis Imaging data for both experiments were analyzed using Brain Voyager QX (version 1.9; Brain Innovation, Maastricht, The Netherlands) using the following steps. Individual subject s imaging data underwent high-pass temporal frequency filtering to remove frequencies below three cycles per run, linear trend removal, three-dimensional motion correction, spatial smoothing (6 mm Gaussian kernel, for the group-average analysis only), and were transformed into Talairach stereotaxic space (Talairach and Tournoux 1988). Functional volumes were then aligned to the anatomical volumes, thereby transforming the functional data into a common stereotaxic space across subjects. Data were analyzed using a general linear model (GLM) and a random-effect GLM was used for the group-average analysis. The model included five experimental predictors (one for each stimulus category in each experiment) and six motion correction predictors (x, y, z for translation and for rotation). The periods of fixation (14 s) were used as baseline. The experimental predictors were modeled as a transient (14 s) epoch where the square-wave function was convolved with the default Brain Voyager QX two-gamma function designed to estimate the hemodynamic response. In the averaged voxelwise group analysis, statistical activation maps were set to reliable threshold levels and cluster volumes (P 0.005, minimum cluster size 27) using Monte Carlo simulations (performed using Brain Voyager QX) to verify that our regions of interest were unlikely to have arisen by chance as a consequence of multiple comparisons. In single-subject analysis, activations were defined in each individual at a threshold of P 0.05, Bonferroni corrected. In experiment 1, for both the averaged voxelwise group analysis and the individual subject analysis we first identified the active brain areas with a given comparison of interest using odd runs only. Analyses on the percentage bold signal change (%BSC), which were performed on the individual subject data only, were extracted from the even runs only. The use of separate data sets to identify the statistical maps and to evaluate brain activity within them circumvents the problem of circular analysis (Kriegeskorte et al. 2009), where the independence of statistical tests for selective analysis is invalidated by the bias present in the comparisons used to select the areas (procedures also called double dipping ). For each region of interest (ROI) we extracted the event-related time course from a mm cube centered at peak activation and then we computed the %BSC for the peak response (by averaging 3 to 6 volumes after stimulus appearance). The exact number of volumes was chosen in consideration of the shape of the peak activation on the averaged data. %BSC was then analyzed by using repeated-measures ANOVAs and post hoc pairwise t-test comparisons (Šidák corrected). The pattern of brain activity for the stimuli presented in experiment 2 was tested by localizing hand-selective voxels using odd and even runs of experiment 1 and extracting the %BSC by using a mm cube centered at peak activation using odd and even runs from experiment 2. Data were then analyzed by averaging peak response (4 8 volumes) and by using ANOVA statistics as before. With respect to experiment 1, although brain activations related to a given comparison of interest were localized by using odd runs only, the pattern of brain activity was analyzed using even runs.
4 NEURAL RESPONSE TO HANDS IN THE VISUAL CORTEX 3391 RESULTS Experiment 1 Experiment 1 of this fmri study served to localize the selective neural substrate for visual coding of the human hand. In this experiment five different categories of visual stimuli were presented using a block design paradigm: whole bodies, body parts, human hands, tools, and chairs (Fig. 1A and METHODS). We first compared whole bodies and body parts versus chairs, given that it is the typical contrast used to localize body-selective voxels in the extrastriate visual cortex (e.g., EBA and FBA; Peelen and Downing 2007). To localize a selective neural response to hands, we similarly compared hands versus chairs. Figure 1B shows bilateral brain activations for both group average contrasts in the occipitotemporal cortex, demonstrating a large degree of overlap between regions activated in both contrasts: whole bodies and body parts versus chairs (body-preferring regions in EBA, depicted in orange) and hands versus chairs (hand-preferring regions, depicted in white; see also Table 1 for the Talairach coordinates of all activated areas). To investigate whether, despite a shared anatomical site, the hand-preferring regions and the bodypreferring regions in the lateral occipitotemporal cortices are functionally dissociated, we analyzed the pattern of brain activity using percentage of bold signal change (%BSC; see METHODS) extracted from these regions localized in individual subjects. Brain activity underwent a repeatedmeasures ANOVA using Hemisphere (left and right), region of interest ROI (EBA, hand-preferring region) and Stimulus type (whole bodies, body parts, hands, tools, and chairs) as withinsubjects factors. ROIs were defined from independent data sets (see METHODS). Results revealed significant interactions for Hemisphere Stimulus type [F(4,52) 15.95, P ] and for ROI Stimulus type [F(4,52) 24.97, P ], suggesting that, despite an anatomical overlap, the different categories of stimuli activated each region of interest and the two hemispheres, to a different extent (Fig. 1, C F). Post hoc pairwise comparisons on the %BSC revealed that, whereas hand-responsive voxels elicited higher activity for the hand FIG. 1. Stimuli, averaged statistical maps and activation levels in experiment 1. A: exemplars of stimuli used in experiment 1: whole bodies, body parts, hands, tools, and chairs. B: group analysis results are shown in the right and left hemisphere of a single subject for the comparison of hands vs. chairs (H C, shown in white) and for whole body and body parts vs. chairs (WB BP C, shown in orange). C F: average peak activity (percentage bold signal change [%BSC]) for each stimulus category extracted from individual-subject brain areas functionally localized with the comparisons illustrated in B. Error bars represent SEs. G: group analysis results are shown in the right and left hemispheres of a single subject for the more selective comparison of hands vs. whole bodies, body parts, and chairs (H WB BP C, shown in yellow) and for whole bodies and body parts vs. hands and chairs (WB BP H C, shown in red). H J: average peak activity (%BSC) for each stimulus category in the individual-subject brain areas localized with the comparisons illustrated in G. Error bars represent SEs.
5 3392 BRACCI, IETSWAART, PEELEN, AND CAVINA-PRATESI TABLE 1. Main contrasts, brain areas, volume, Talairach coordinates, and t values for averaged group data in experiment 1 Talairach Coordinates Contrast Brain Region Volume, mm 3 x y z t Value WB BP C Left occipitotemporal cortex 3, Left middle fusiform gyrus 1, Right occipitotemporal cortex 7, Right middle fusiform gyrus 1, H C Left occipitotemporal cortex 2, Right occipitotemporal cortex 1, WB BP H C Left occipitotemporal cortex 1, Left middle fusiform gyrus Right occipitotemporal cortex 5, Right middle fusiform gyrus 2, H WB BP C Left occipitotemporal cortex 1, H BP Left occipitotemporal cortex WH, whole bodies; BP, body parts; H, hands; C, chairs. stimuli compared with that for body parts [t (13) 5.61, P 0.001; see Fig. 1, C and D], body-responsive voxels in left and right EBA showed a higher response to whole bodies compared with that to hands [t (13) 4.35, P 0.008; see Fig. 1, E and F]. Interestingly, although the response to tools was statistically indistinguishable from that to chairs (P 0.05) within the body-preferring region, it was significantly higher than that to chairs within the hand-preferring region [t (13) 6.16, P ]. In addition, we found a higher response to hand stimuli in the left hemisphere [hands body parts, t (13) 3.65, P 0.028; see Fig. 1, C and E] and for body stimuli in the right hemisphere [whole bodies hands, t (13) 6.79, P ; see Fig. 1, D and F]. These analyses suggest that, despite an anatomical overlap between body- and hand-preferring regions, a different hemispheric lateralization as well as a distinctive pattern of activation to the different stimulus categories could be observed for the two regions (Fig. 1, C F). Next, we tested whether body-responsive and hand-responsive voxels can be anatomically dissociated by adopting more selective contrasts. To this end, we localized body-preferring voxels by comparing whole bodies and body parts versus hands and chairs, and hand-preferring voxels by comparing hands versus whole bodies, body parts, and chairs. The first contrast gave significant body-preferring activation in the right and left lateral occipitotemporal cortex and in the left and right fusiform gyrus (see Talairach coordinates in Table 1; also see Fig. 1G). The localization of the hand-preferring region in this analysis is now much more specific, with the contrast of hands versus whole bodies, body parts, and chairs showing a significant activation only in the left lateral occipitotemporal cortex (Talairach coordinates: x 46, y 65, z 1; see also Fig. 1G). Furthermore, individual-subject analyses revealed that the hand-preferring region was evident in each participant (see Fig. 2, depicted in yellow and Supplemental Table S1). 1 Moreover, hand- and body-preferring activations in the lateral occipitotemporal cortices were segregated from each other, with the hand-preferring response localized significantly more anterior (t-test for the y Talairach coordinates: P 0.002) to the body-preferring response. Interestingly, the hand-preferring region was largely confined to an area along the left lateral occipital sulcus (Damasio 2005). For convenience, in the remainder of this study, we will refer to this highly robust 1 The online version of this article contains supplemental data. hand-preferring response in the left lateral occipital sulcus using its anatomical location (left LOS, Fig. 2). The use of an anatomical landmark to label a functional activation has been previously applied the lateral occipital cortex (LOC) for shape recognition (see Malach et al. 1995) and the middle temporal gyrus (MTG) for tool representation (see Chao et al. 1999) and may also be appropriate here because functional characteristics of the region might change with future studies. Furthermore, this label is intended to describe rather than to identify a distinct functional brain area (see DISCUSSION). Interestingly, contrary to EBA, the hand response in LOS appears to be mainly lateralized in the left hemisphere, with only 6 of 14 subjects showing bilateral hand-preferring activation. To demonstrate how EBA and left LOS, as defined with the more selective contrasts, functionally differ from each other, single subjects %BSC underwent a 3 5 repeated-measures ANOVA using ROI (left-los, right-eba, and left-eba) and Stimulus type (whole bodies, body parts, hands, tools, and chairs) as within-subjects factors. The results revealed a main effect of Stimulus type [F(4,52) 48.9, P ] and, importantly, a significant interaction of Stimulus type ROI [F(8,104) 16.6, P ], demonstrating that the functional properties of these regions differ from each other (Fig. 1, H I). Whereas left LOS elicited a significantly higher response to hands compared with that to whole bodies [t (13) 6.13, P ], body parts [t (13) 6.62, P ], tools [t (13) 7.06, P ], and chairs [t (13) 11.22, P ], without distinguishing between whole bodies, body parts, and tools, activation in the right EBA was higher for whole bodies and body parts compared with that for hands [t (13) 12.35, P ; t (13) 4.38, P 0.001, respectively], tools [t (13) 11.95, P ; t (13) 6.76, P , respectively], and chairs [t (13) 12.26, P ; t (13) 7.75, P , respectively]. In addition, and in line with previous findings (Taylor et al. 2007), whole bodies elicited higher activation than body parts in the right EBA [t (13) 3.65, P 0.05]. Moreover, within right EBA, hands elicited a significantly higher response compared with nonbiological stimuli [tools: t (13) 5.10, P 0.002; chairs: t (13) 5.33, P 0.001]. Within left EBA, we found that hands, whole bodies, and body parts were statistically indistinguishable (P 0.10) and showed similar significantly higher responses compared with the nonbiological stimuli represented by tools and chairs
6 NEURAL RESPONSE TO HANDS IN THE VISUAL CORTEX 3393 FIG. 2. Individual statistical maps for hand-responsive and body-responsive voxels in experiment 1. The position of hand-selective voxels localized by comparing hands vs. whole bodies, body parts, and chairs (H WB BP C, highlighted in yellow) and of body-selective voxels (extrastriate body area [EBA]) localized by comparing whole bodies and body parts vs. hands and chairs (WB BP H C highlighted in red) in experiment 1 is shown in the clearest transversal slice of each participant. Averaged statistical maps (bottom right image) for the hand-responsive area within left lateral occipital sulcus (LOS) and the body-responsive area in right and left EBA are overlaid onto the slice of a single participant for clarity. As clearly shown, although the hand-related response in left LOS is mostly lateralized to the left (left LOS 14/14 participants; right LOS 6/14 participants), EBA is bilaterally represented (left and right EBA 14/14). In addition, in most of the participants (11/14) left LOS is localized anterior to left EBA. L, left; R, right. (for all comparisons, P 0.05). Overall, the above-cited results indicate that the functional profile of left LOS differs from that of EBA, with left LOS being selective for the visual processing of the human hand compared with the rest of the body. Furthermore, whereas tool stimuli activated left LOS significantly above baseline and significantly more than chairs [t (13) 6.56, P ], both tools and chairs did not elicit any detectable activation in either right or left EBA (Fig. 1, H J). To confirm the hand versus nonhand body-part preference of left LOS, we computed the more direct and conservative comparison of hands versus body parts, both at group level and in individual subjects. In the group analysis, results revealed a hand-preferring activation in the left hemisphere only (x 43, y 65, z 3; see also Table 1), overlapping with the left LOS region defined with the contrast hands versus whole body, body parts, and chairs. The same comparison was applied to single subject s data and, as shown in (Supplemental Fig. 1SA and Supplemental Table S1, the activated areas for hands versus body parts were localized in the left hemisphere in all participants (14/14 left hemisphere; 6/14 right hemisphere). Indeed, both contrasts (H BP and H WB BP C) activated highly similar regions, as indicated by nearly identical Talairach coordinates (Supplemental Table S1) that did not significantly differ (P 0.34, for x, y, and z coordinates). Subsequently, the functional profile of the left LOS region defined by hands versus body parts was investigated. Single subjects %BSC underwent a repeated-measures ANOVA using Stimulus type (whole bodies, body parts, hands, tools, and chairs) as within-subjects factor. The results (shown in Supplemental Fig. S1B) revealed a main effect of Stimulus type [F(4,52) 30.20, P ] and post hoc analyses confirmed previous results showing a significantly higher response to hands compared with that to whole bodies [t (13) 5.25, P 0.002], body parts [t (13) 5.76, P 0.001], tools [t (13) 5.61, P 0.001], and chairs [t (13) 8.58, P ]. Importantly, whole bodies and body parts were statistically indistinguishable when compared with each other (P 1) and versus tools (for both comparisons, P 0.925). Interestingly, the two nonbiological stimuli showed different responses in left LOS: whereas tools were significantly higher than chairs [t (13) 5.35, P 0.001], the latter were significantly inferior from baseline [t (13) 5.28, P ] and significantly lower than that of all the other categories of stimuli (for all comparisons, P 0.003). Experiment 2 Questions arise as to the specific properties of the handpreferring region in the left LOS. More in particular: 1) isthe left LOS specifically responsive to hands or more generally to visually similar subcategories of body parts (e.g., feet); 2) does the left LOS respond to the hand as a whole or to a single part of it (e.g., the fingers); and 3) does the left LOS respond to nonbiological stimuli that resemble the hand in terms of their function (e.g., robotic hands)? To answer these questions in experiment 2 we measured the neural response of
7 3394 BRACCI, IETSWAART, PEELEN, AND CAVINA-PRATESI left LOS to images of not only fingers, feet, robotic hands, but also body parts and hands (Fig. 3A). Given the strong lateralization for the hand response, we focused our analyses in the left hemisphere and for each individual participant we first localized EBA and LOS in the left hemisphere as regions of interest using the main contrasts and data set of experiment 1 (hand contrast: hands vs. whole bodies, body parts, and chairs; body contrast: whole bodies and body parts vs. hands and chairs). The %BSC in individually defined left LOS and EBA underwent a 2 5 ANOVA with ROI (left LOS and left EBA) and Stimulus type (hands, feet, fingers, robotic hands, and body parts; see Fig. 3A) as within-subject factors. Results revealed a main effect of Stimulus type [F(4,48) 9.66, P ], further qualified by an interaction between ROI Stimulus type [F(4,48) 30.15, P ], indicating that the two regions responded differently to the stimuli (see Fig. 3, B and C). Within left EBA, activation for body parts did not differ from that for feet and human hands, but was significantly higher than that for fingers [t (12) 4.45, P 0.01] and robotic hands [t (12) 4.30, P 0.01; see Fig. 3B]. Conversely, within left LOS brain activity for human hands was significantly greater than that for body parts [t (12) 5.71, P 0.001], feet [t (12) 3.40, P 0.05], and fingers [t (12) 3.54, P 0.05] and, despite showing a tendency, did not significantly differ from that for robotic hands [t (12) 3.15, P 0.079] (see Fig. 3C). For completeness, we repeated the analysis by localizing left LOS with the more conservative contrast of hand versus body parts. The %BSC for left LOS underwent a one-way repeated-measures ANOVA with Stimulus type (hands, feet, fingers, robotic hands, and body parts) as within-subjects factor. Results revealed a main effect of Stimulus type [F(4,48) 20.11, P ], indicating that in the left LOS brain activity for A Fingers Body-parts Hands Feet human hands was significantly greater than that for body parts [t (12) 5.98, P 0.001], feet [t (12) 4.18, P 0.013], and fingers [t (12) 3.87, P 0.023] and, despite showing a tendency, did not significantly differ from robotic hands [t (12) 3.22, P 0.068]. In addition we tested whether, as found for hands, feet, and fingers may also activate a separate portion of the visual cortex. To do so, we compared feet versus body parts and fingers versus body parts using odd runs only. No significant activations were found for either contrasts at the chosen threshold (or at a more liberal one, for example, P 0.05, uncorrected for multiple comparison). We also performed the comparison at the level of single subjects. Significant activation in left lateral occipitotemporal cortex was found in 9 of 13 participants for the contrast feet versus body parts and in 12 of 13 participants for the contrast fingers versus body parts. However, %BSC in both areas suggested a lack of selectivity for both feet and fingers stimuli showing that there was no significant difference between feet and hands and between fingers and hands (see Supplemental text and Supplemental Fig. S2, A and B). As expected, the contrast hands versus body parts again gave significant (P 0.01) activation in left occipitotemporal cortex (Talairach coordinates: x 43, y 64, z 2). DISCUSSION The present fmri study provides evidence for a handpreferring region in the human extrastriate visual cortex. Handpreferring responses were strongest in left lateral occipitotemporal cortex. Two experiments demonstrated the functional preference of left LOS to hands relative to various control conditions, including feet and fingers, that share several features with hands (fingers Robotic-hands B % BSC Fingers Body-parts Hands Feet Robotic-hands C 1 Left EBA 0.8 Left LOS FIG. 3. Stimuli and activation levels for experiment 2. A: exemplars of stimuli used in experiment 2: fingers, body parts, hands, feet, and robotic hands. B and C: average time course (%BSC) for each stimulus category in experiment 2 in left LOS, functionally localized in each individual by comparing hands vs. whole bodies, body parts, and chairs (H WB BP C) and left EBA functionally localized in each individual by comparing whole bodies and body parts vs. hands and chairs (WB BP H C) for odd and even runs of experiment 1. Error bars represent SEs Time (s) Time (s)
8 NEURAL RESPONSE TO HANDS IN THE VISUAL CORTEX 3395 are part of the hand and feet have five toes). The hand response was localized anterior and closely adjacent to non-hand body responses. Interestingly, contrary to EBA, the hand response in the LOS was strongly left lateralized. Indeed, all 14 participants showed a hand response in the left hemisphere, whereas only 6 participants showed a similar activation also in the right hemisphere. These findings are in line with previous evidence reporting the existence of 1) hand-selective cells in monkey IT (Desimone et al. 1984; Gross et al. 1969), 2) handresponsive ERP sites in the human left temporal lobe (Mc- Carthy et al. 1999), and 3) hand versus torso specificity in the human visual cortex using fmri (Op de Beeck et al. 2010). Importantly, the present data provide the first evidence for a double dissociation in the lateral occipitotemporal cortex between the representations of a specific body part such as the hand in the left LOS (where activation for hands was higher than that for both whole bodies and body parts) and bodies in general in EBA (where activation for both whole body and body parts were higher than that for hands). Indeed, although previous work (Op de Beeck et al. 2010) showed higher activation for hands compared with that for torsos and faces in the lateral occipitotemporal cortex, it did not find significantly greater responses to torsos than to hands a preferred response to torsos compared with that to hands was reported only in the right fusiform gyrus. The selective response to hands but not torsos is in agreement with our findings and suggests that hands, more than other body parts, could be overrepresented in the lateral occipitotemporal cortex. What do our results suggest about the organization of body part representations in the human lateral occipitotemporal cortex? One possibility is that selective representations exist for some, or perhaps all, individual body parts, with the EBA being the collection of these separate representations. Alternatively, the hand, similar to the face, could be a special body part and distinctive among other body parts in eliciting selective responses in the human high-order visual cortex. Indeed, the way we interact with cospecifics and with the external world is continuously mediated by our hands, starting with learning how to intake food, following by pointing at objects to name them, continuing with learning how to count and write, and then developing into more complex abilities such as gesture communication and tool-use execution/understanding. In consideration of the special role played by hands in our daily experience it is therefore possible that hands, like faces, are overrepresented in visual cortex relative to other body parts. However, it should be pointed out that, despite the absence of a selective response to feet and fingers in the current study, we cannot rule out that a less conservative contrast (e.g., feet vs. chairs and fingers vs. chairs) would uncover such responses. Finally, future experiments using high-resolution fmri (Schwarzlose et al. 2005) and fmri adaptation will be useful to test for the existence of separate body part representations within EBA and the extrastriate visual cortex more generally. Regardless of whether the hand response in left LOS is a unique functional region or should instead be considered a subregion of EBA, it clearly showed a distinct functional specialization in the present experiments. The left LOS, but not EBA, responded to non-human robotic hands and partially to nonbiological stimuli such as tools, but not to other objects such as chairs. This response profile may relate to recent findings suggesting a broader modulation of neural activity in visual areas from other modalities, such as semantics (Mahon et al. 2009), tactile information (Amedi et al. 2001; Burton et al. 2002), and motor action (Astafiev et al. 2004). In this respect, humans unique manual abilities to manipulate external objects and tools might influence the information encoded within left LOS. The relevance of occipitotemporal regions in storing, not only visual object information, but also motion and associated action knowledge with it, gained support in a series of experiments by Beauchamp and colleagues (2002, 2003). These reports showed that although an area in the posterior part of the superior temporal sulcus (STS), anterior and superior to the motion area MT (Grossman and Blake 2002; Grossman et al. 2000), responds to body-related form and motion, a region within the posterior middle temporal gyrus (MTG), anterior and inferior to MT, responds to tool-associated form and motion (Beauchamp et al. 2002, 2003). Interestingly, anterior to left LOS in the left MTG a selective response to tools has been widely reported (Chao et al. 1999; Valyear and Culham 2009; Valyear et al. 2007), but also see the results report by Downing and colleagues (2006). A number of imaging studies support the role of left MTG in coding not only tool visual form but also conceptual knowledge and tool-associated actions (Binder et al. 2009; Devlin et al. 2002; Martin et al. 1995, 1996). In support of these findings, clinical studies (Tranel et al. 1997, 2003) showed that lesions involving left MTG cause impairments in action knowledge associated with objects. In this respect, the observation that left LOS, but not EBA, responds to tools may reflect a coding of motion information of hand actions associated with tool use. Interestingly, in contrast to the predominantly right hemispheric lateralization of other visual body and face selective regions, such as FBA (Peelen and Downing 2005), FFA (Kanwisher et al. 1997), OFA (Puce et al. 1996), and EBA itself (Downing et al. 2001), the hand response in LOS showed a strong left hemispheric lateralization. Neural mechanisms encoding hands being primarily lateralized in the hemisphere dominant for human praxis may symbolize an advantageous structural organization. Indeed, a left lateralized network would allow fast interhemispheric connections between brain regions in left occipitotemporal cortex representing hand visual form, hand motion, and tool actions, and left frontoparietal regions subserving planning and guidance of hand actions (Goodale and Milner 1992; Rizzolatti et al. 1998). In line with this, clinical studies report that, although left frontoparietal and parietal lesions frequently induce postural and spatial temporal errors in gesture imitation and object usage (Buxbaum and Saffran 2002; Haaland et al. 2000), lesions within the left temporoparietal junction compromise the conceptual knowledge of the correct hand movements associated with a specific object (De Renzi and Lucchelli 1988). An interesting avenue for future research is to test the role of left LOS in the observation and execution of object-directed hand actions and how it is functionally connected with other regions implicated in these processes. Indeed, the body is not a passive entity but mediates our interaction with the external world. This bidirectional interactive experience is likely to be critical in modulating and shaping functional brain organization. In conclusion, this report provides evidence for neural representation of the human hand in the left occipitotemporal cortex. EBA has generally been regarded as a uniform neural substrate dedicated to visual body processing. Our results
9 3396 BRACCI, IETSWAART, PEELEN, AND CAVINA-PRATESI indicate that segregated body-part representations may exist within lateral occipitotemporal cortex, with the hand possibly being overrepresented relative to other body parts. Future experiments are needed to address 1) the functional properties of the LOS hand response (e.g, the difference between hands in grasping and resting postures) and 2) how hand-preferring responses found within left LOS relate functionally and anatomically to other nearby functional areas encoding motion (MT; Tootell et al. 1995), biological motion (superior temporal sulcus [STS]; Hoffman and Haxby 2000), and tool action information (left MTG-TA; Chao et al. 1999). ACKNOWLEDGMENTS We thank Dr. Franz Mechsner and Dr. Quoc Vuong for inputs on earlier versions of the manuscript, L. Morris and C. Smith for assistance with fmri data collection, and A. Bester for help in hardware development. GRANTS This work was supported by internal funding from Northumbria University and UK Medical Research Council Grant G DISCLOSURES No conflicts of interest are declared by the authors. REFERENCES Amedi A, Malach R, Hendler T, Peled S, Zohary E. Visuo-haptic objectrelated activation in the ventral visual pathway. Nat Neurosci 4: , Astafiev SV, Stanley CM, Shulman GL, Corbetta M. Extrastriate body area in human occipital cortex responds to the performance of motor actions. Nat Neurosci 7: , Barton JJ. Disorders of face perception and recognition. Neurol Clin 21: , Beauchamp MS, Lee KE, Haxby JV, Martin A. Parallel visual motion processing streams for manipulable objects and human movements. Neuron 34: , Beauchamp MS, Lee KE, Haxby JV, Martin A. FMRI responses to video and point-light displays of moving humans and manipulable objects. J Cogn Neurosci 15: , Binder JR, Desai RH, Graves WW, Conant LL. Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex 19: , Brainard DH. The Psychophysics Toolbox. Spat Vis 10: , Burton H, Snyder AZ, Diamond JB, Raichle ME. Adaptive changes in early and late blind: a fmri study of verb generation to heard nouns. J Neurophysiol 88: , Buxbaum LJ, Saffran EM. Knowledge of object manipulation and object function: dissociations in apraxic and nonapraxic subjects. Brain Lang 82: , Chao LL, Haxby JV, Martin A. Attribute-based neural substrates in temporal cortex for perceiving and knowing about objects. Nat Neurosci 2: , Damasio AR, Damasio H, Van Hoesen GW. Prosopagnosia: anatomic basis and behavioral mechanisms. Neurology 32: , Damasio H. Human Brain Anatomy in Computerized Images. Oxford, UK: Oxford Univ, De Renzi E, Lucchelli F. Ideational apraxia. Brain 111: , Desimone R, Albright TD, Gross CG, Bruce C. Stimulus-selective properties of inferior temporal neurons in the macaque. J Neurosci 4: , Devlin JT, Moore CJ, Mummery CJ, Gorno-Tempini ML, Phillips JA, Noppeney U, Frackowiak RS, Friston KJ, Price CJ. Anatomic constraints on cognitive theories of category specificity. NeuroImage 15: , Downing PE, Chan AW, Peelen MV, Dodds CM, Kanwisher N. Domain specificity in visual cortex. Cereb Cortex 16: , Downing PE, Jiang Y, Shuman M, Kanwisher N. A cortical area selective for visual processing of the human body. Science 293: , Goodale MA, Milner AD. Separate visual pathways for perception and action. Trends Neurosci 15: 20 25, Gross CG, Bender DB, Rocha-Miranda CE. Visual receptive fields of neurons in inferotemporal cortex of the monkey. Science 166: , Grossman E, Donnelly M, Price R, Pickens D, Morgan V, Neighbor G, Blake R. Brain areas involved in perception of biological motion. J Cogn Neurosci 12: , Grossman ED, Blake R. Brain areas active during visual perception of biological motion. Neuron 35: , Haaland KY, Harrington DL, Knight RT. Neural representations of skilled movement. Brain 123: , Hoffman EA, Haxby JV. Distinct representations of eye gaze and identity in the distributed human neural system for face perception. Nat Neurosci 3: 80 84, Kanwisher N, McDermott J, Chun MM. The fusiform face area: a module in human extrastriate cortex specialized for face perception. J Neurosci 17: , Kriegeskorte N, Simmons WK, Bellgowan PS, Baker CI. Circular analysis in systems neuroscience: the dangers of double dipping. Nat Neurosci 12: , Mahon BZ, Anzellotti S, Schwarzbach J, Zampini M, Caramazza A. Category-specific organization in the human brain does not require visual experience. Neuron 63: , Malach R, Reppas JB, Benson RR, Kwong KK, Jiang H, Kennedy WA, Ledden PJ, Brady TJ, Rosen BR, Tootell RB. Object-related activity revealed by functional magnetic resonance imaging in human occipital cortex. Proc Natl Acad Sci USA 92: , Martin A, Haxby JV, Lalonde FM, Wiggs CL, Ungerleider LG. Discrete cortical regions associated with knowledge of color and knowledge of action. Science 270: , Martin A, Wiggs CL, Ungerleider LG, Haxby JV. Neural correlates of category-specific knowledge. Nature 379: , McCarthy G, Puce A, Belger A, Allison T. Electrophysiological studies of human face perception. II: Response properties of face-specific potentials generated in occipitotemporal cortex. Cereb Cortex 9: , Moro V, Urgesi C, Pernigo S, Lanteri P, Pazzaglia M, Aglioti SM. The neural basis of body form and body action agnosia. Neuron 60: , Napier JR. The prehensile movements of the human hand. J Bone Joint Surg Br 38B: , Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9: , Op de Beeck HP, Brants M, Baeck A, Wagemans J. Distributed subordinate specificity for bodies, faces, and buildings in human ventral visual cortex. NeuroImage 49: , Peelen MV, Downing PE. Selectivity for the human body in the fusiform gyrus. J Neurophysiol 93: , Peelen MV, Downing PE. The neural basis of visual body perception. Nat Rev Neurosci 8: , Pitcher D, Charles L, Devlin JT, Walsh V, Duchaine B. Triple dissociation of faces, bodies, and objects in extrastriate cortex. Curr Biol 19: , Puce A, Allison T, Asgari M, Gore JC, McCarthy G. Differential sensitivity of human visual cortex to faces, letterstrings, and textures: a functional magnetic resonance imaging study. J Neurosci 16: , Rizzolatti G, Luppino G, Matelli M. The organization of the cortical motor system: new concepts. Electroencephalogr Clin Neurophysiol 106: , Rossion B, Schiltz C, Crommelinck M. The functionally defined right occipital and fusiform face areas discriminate novel from visually familiar faces. NeuroImage 19: , Schwarzlose RF, Baker CI, Kanwisher N. Separate face and body selectivity on the fusiform gyrus. J Neurosci 25: , Steeves J, Dricot L, Goltz HC, Sorger B, Peters J, Milner AD, Goodale MA, Goebel R, Rossion B. Abnormal face identity coding in the middle fusiform gyrus of two brain-damaged prosopagnosic patients. Neuropsychologia 47: , Talairach J, Tournoux P. Co-Planar Stereotaxic Atlas of the Human Brain. New York: Thieme Medical, 1988.
10 NEURAL RESPONSE TO HANDS IN THE VISUAL CORTEX 3397 Taylor JC, Wiggett AJ, Downing PE. Functional MRI analysis of body and body part representations in the extrastriate and fusiform body areas. J Neurophysiol 98: , Tootell RB, Reppas JB, Kwong KK, Malach R, Born RT, Brady TJ, Rosen BR, Belliveau JW. Functional analysis of human MT and related visual cortical areas using magnetic resonance imaging. J Neurosci 15: , Tranel D, Damasio H, Damasio AR. A neural basis for the retrieval of conceptual knowledge. Neuropsychologia 35: , Tranel D, Kemmerer D, Adolphs R, Damasio H, Damasio AR. Neural correlates of conceptual knowledge for actions. Cogn Neuropsychol 20: , Urgesi C, Candidi M, Ionta S, Aglioti SM. Representation of body identity and body actions in extrastriate body area and ventral premotor cortex. Nat Neurosci 10: 30 31, Valyear KF, Cavina-Pratesi C, Stiglick AJ, Culham JC. Does tool-related fmri activity within the intraparietal sulcus reflect the plan to grasp? NeuroImage 36, Suppl. 2: T94 T108, Valyear KF, Culham JC. Observing learned object-specific functional grasps preferentially activates the ventral stream. J Cogn Neurosci 22: , Van Overwalle F, Baetens K. Understanding others actions and goals by mirror and mentalizing systems: a meta-analysis. Neuroimage 48: , 2009.
Closely overlapping responses to tools and hands in left lateral occipitotemporal cortex
 J Neurophysiol 107: 1443 1456, 2012. First published November 30, 2011; doi:10.1152/jn.00619.2011. Closely overlapping responses to tools and hands in left lateral occipitotemporal cortex Stefania Bracci,
J Neurophysiol 107: 1443 1456, 2012. First published November 30, 2011; doi:10.1152/jn.00619.2011. Closely overlapping responses to tools and hands in left lateral occipitotemporal cortex Stefania Bracci,
Summary. Multiple Body Representations 11/6/2016. Visual Processing of Bodies. The Body is:
 Visual Processing of Bodies Corps et cognition: l'embodiment Corrado Corradi-Dell Acqua corrado.corradi@unige.ch Theory of Pain Laboratory Summary Visual Processing of Bodies Category-specificity in ventral
Visual Processing of Bodies Corps et cognition: l'embodiment Corrado Corradi-Dell Acqua corrado.corradi@unige.ch Theory of Pain Laboratory Summary Visual Processing of Bodies Category-specificity in ventral
Accepted refereed manuscript of:
 Accepted refereed manuscript of: Bracci S, Bracci S, Cavina-Pratesi C, Connolly JD & Ietswaart M (2016) Representational content of occipitotemporal and parietal tool areas, Neuropsychologia, 84, pp. 81-88.
Accepted refereed manuscript of: Bracci S, Bracci S, Cavina-Pratesi C, Connolly JD & Ietswaart M (2016) Representational content of occipitotemporal and parietal tool areas, Neuropsychologia, 84, pp. 81-88.
Impaired face discrimination in acquired prosopagnosia is associated with abnormal response to individual faces in the right middle fusiform gyrus
 Impaired face discrimination in acquired prosopagnosia is associated with abnormal response to individual faces in the right middle fusiform gyrus Christine Schiltz Bettina Sorger Roberto Caldara Fatima
Impaired face discrimination in acquired prosopagnosia is associated with abnormal response to individual faces in the right middle fusiform gyrus Christine Schiltz Bettina Sorger Roberto Caldara Fatima
Are face-responsive regions selective only for faces?
 Cognitive Neuroscience and Neurophysiology 10, 2945±2950 (1999) TO examine the speci city of face-responsive regions for face processing, we used fmri to measure the response of the fusiform gyrus and
Cognitive Neuroscience and Neurophysiology 10, 2945±2950 (1999) TO examine the speci city of face-responsive regions for face processing, we used fmri to measure the response of the fusiform gyrus and
Separate Face and Body Selectivity on the Fusiform Gyrus
 The Journal of Neuroscience, November 23, 2005 25(47):11055 11059 11055 Brief Communication Separate Face and Body Selectivity on the Fusiform Gyrus Rebecca F. Schwarzlose, 1,2 Chris I. Baker, 1,2 and
The Journal of Neuroscience, November 23, 2005 25(47):11055 11059 11055 Brief Communication Separate Face and Body Selectivity on the Fusiform Gyrus Rebecca F. Schwarzlose, 1,2 Chris I. Baker, 1,2 and
Supplementary information Detailed Materials and Methods
 Supplementary information Detailed Materials and Methods Subjects The experiment included twelve subjects: ten sighted subjects and two blind. Five of the ten sighted subjects were expert users of a visual-to-auditory
Supplementary information Detailed Materials and Methods Subjects The experiment included twelve subjects: ten sighted subjects and two blind. Five of the ten sighted subjects were expert users of a visual-to-auditory
fmri analysis of body and body part representations in the extrastriate and fusiform body areas
 Page 1 of 34 Articles in PresS. J Neurophysiol (June 27, 2007). doi:10.1152/jn.00012.2007 1 Body processing in EBA and FBA fmri analysis of body and body part representations in the extrastriate and fusiform
Page 1 of 34 Articles in PresS. J Neurophysiol (June 27, 2007). doi:10.1152/jn.00012.2007 1 Body processing in EBA and FBA fmri analysis of body and body part representations in the extrastriate and fusiform
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis
 Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Procedia - Social and Behavioral Sciences 159 ( 2014 ) WCPCG 2014
 Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
How do individuals with congenital blindness form a conscious representation of a world they have never seen? brain. deprived of sight?
 How do individuals with congenital blindness form a conscious representation of a world they have never seen? What happens to visual-devoted brain structure in individuals who are born deprived of sight?
How do individuals with congenital blindness form a conscious representation of a world they have never seen? What happens to visual-devoted brain structure in individuals who are born deprived of sight?
Selective Attention to Face Identity and Color Studied With fmri
 Human Brain Mapping 5:293 297(1997) Selective Attention to Face Identity and Color Studied With fmri Vincent P. Clark, 1 * Raja Parasuraman, 2 Katrina Keil, 1 Rachel Kulansky, 1 Sean Fannon, 2 Jose Ma.
Human Brain Mapping 5:293 297(1997) Selective Attention to Face Identity and Color Studied With fmri Vincent P. Clark, 1 * Raja Parasuraman, 2 Katrina Keil, 1 Rachel Kulansky, 1 Sean Fannon, 2 Jose Ma.
Supporting Information
 Supporting Information Lingnau et al. 10.1073/pnas.0902262106 Fig. S1. Material presented during motor act observation (A) and execution (B). Each row shows one of the 8 different motor acts. Columns in
Supporting Information Lingnau et al. 10.1073/pnas.0902262106 Fig. S1. Material presented during motor act observation (A) and execution (B). Each row shows one of the 8 different motor acts. Columns in
Selectivity for the Human Body in the Fusiform Gyrus
 J Neurophysiol 93: 603 608, 2005. First published August 4, 2004; doi:10.1152/jn.00513.2004. Selectivity for the Human Body in the Fusiform Gyrus Marius V. Peelen and Paul E. Downing School of Psychology,
J Neurophysiol 93: 603 608, 2005. First published August 4, 2004; doi:10.1152/jn.00513.2004. Selectivity for the Human Body in the Fusiform Gyrus Marius V. Peelen and Paul E. Downing School of Psychology,
WHAT DOES THE BRAIN TELL US ABOUT TRUST AND DISTRUST? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1
 SPECIAL ISSUE WHAT DOES THE BRAIN TE US ABOUT AND DIS? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1 By: Angelika Dimoka Fox School of Business Temple University 1801 Liacouras Walk Philadelphia, PA
SPECIAL ISSUE WHAT DOES THE BRAIN TE US ABOUT AND DIS? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1 By: Angelika Dimoka Fox School of Business Temple University 1801 Liacouras Walk Philadelphia, PA
The Neural Basis of Body Form and Body Action Agnosia
 Clinical Study The Neural Basis of Body Form and Body Action Agnosia Valentina Moro, 1, * Cosimo Urgesi, 2 Simone Pernigo, 3 Paola Lanteri, 4 Mariella Pazzaglia, 5,6 and Salvatore Maria Aglioti 5,6, *
Clinical Study The Neural Basis of Body Form and Body Action Agnosia Valentina Moro, 1, * Cosimo Urgesi, 2 Simone Pernigo, 3 Paola Lanteri, 4 Mariella Pazzaglia, 5,6 and Salvatore Maria Aglioti 5,6, *
Twelve right-handed subjects between the ages of 22 and 30 were recruited from the
 Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
Normal Body Perception despite the Loss of Right Fusiform Gyrus
 Normal Body Perception despite the Loss of Right Fusiform Gyrus Tirta Susilo 1, Hua Yang 1, Zachary Potter 2, Rachel Robbins 3, and Bradley Duchaine 1 Abstract Human extrastriate cortex contains functional
Normal Body Perception despite the Loss of Right Fusiform Gyrus Tirta Susilo 1, Hua Yang 1, Zachary Potter 2, Rachel Robbins 3, and Bradley Duchaine 1 Abstract Human extrastriate cortex contains functional
Attention to Form or Surface Properties Modulates Different Regions of Human Occipitotemporal Cortex
 Cerebral Cortex March 2007;17:713-731 doi:10.1093/cercor/bhk022 Advance Access publication April 28, 2006 Attention to Form or Surface Properties Modulates Different Regions of Human Occipitotemporal Cortex
Cerebral Cortex March 2007;17:713-731 doi:10.1093/cercor/bhk022 Advance Access publication April 28, 2006 Attention to Form or Surface Properties Modulates Different Regions of Human Occipitotemporal Cortex
Perception of Faces and Bodies
 CURRENT DIRECTIONS IN PSYCHOLOGICAL SCIENCE Perception of Faces and Bodies Similar or Different? Virginia Slaughter, 1 Valerie E. Stone, 2 and Catherine Reed 3 1 Early Cognitive Development Unit and 2
CURRENT DIRECTIONS IN PSYCHOLOGICAL SCIENCE Perception of Faces and Bodies Similar or Different? Virginia Slaughter, 1 Valerie E. Stone, 2 and Catherine Reed 3 1 Early Cognitive Development Unit and 2
Scratching Beneath the Surface: New Insights into the Functional Properties of the Lateral Occipital Area and Parahippocampal Place Area
 8248 The Journal of Neuroscience, June 1, 2011 31(22):8248 8258 Behavioral/Systems/Cognitive Scratching Beneath the Surface: New Insights into the Functional Properties of the Lateral Occipital Area and
8248 The Journal of Neuroscience, June 1, 2011 31(22):8248 8258 Behavioral/Systems/Cognitive Scratching Beneath the Surface: New Insights into the Functional Properties of the Lateral Occipital Area and
Hallucinations and conscious access to visual inputs in Parkinson s disease
 Supplemental informations Hallucinations and conscious access to visual inputs in Parkinson s disease Stéphanie Lefebvre, PhD^1,2, Guillaume Baille, MD^4, Renaud Jardri MD, PhD 1,2 Lucie Plomhause, PhD
Supplemental informations Hallucinations and conscious access to visual inputs in Parkinson s disease Stéphanie Lefebvre, PhD^1,2, Guillaume Baille, MD^4, Renaud Jardri MD, PhD 1,2 Lucie Plomhause, PhD
FAILURES OF OBJECT RECOGNITION. Dr. Walter S. Marcantoni
 FAILURES OF OBJECT RECOGNITION Dr. Walter S. Marcantoni VISUAL AGNOSIA -damage to the extrastriate visual regions (occipital, parietal and temporal lobes) disrupts recognition of complex visual stimuli
FAILURES OF OBJECT RECOGNITION Dr. Walter S. Marcantoni VISUAL AGNOSIA -damage to the extrastriate visual regions (occipital, parietal and temporal lobes) disrupts recognition of complex visual stimuli
Functional topography of a distributed neural system for spatial and nonspatial information maintenance in working memory
 Neuropsychologia 41 (2003) 341 356 Functional topography of a distributed neural system for spatial and nonspatial information maintenance in working memory Joseph B. Sala a,, Pia Rämä a,c,d, Susan M.
Neuropsychologia 41 (2003) 341 356 Functional topography of a distributed neural system for spatial and nonspatial information maintenance in working memory Joseph B. Sala a,, Pia Rämä a,c,d, Susan M.
The roles of face and non-face areas during individual face perception: Evidence by fmri adaptation in a brain-damaged prosopagnosic patient
 www.elsevier.com/locate/ynimg NeuroImage 40 (2008) 318 332 The roles of face and non-face areas during individual face perception: Evidence by fmri adaptation in a brain-damaged prosopagnosic patient Laurence
www.elsevier.com/locate/ynimg NeuroImage 40 (2008) 318 332 The roles of face and non-face areas during individual face perception: Evidence by fmri adaptation in a brain-damaged prosopagnosic patient Laurence
Comparing event-related and epoch analysis in blocked design fmri
 Available online at www.sciencedirect.com R NeuroImage 18 (2003) 806 810 www.elsevier.com/locate/ynimg Technical Note Comparing event-related and epoch analysis in blocked design fmri Andrea Mechelli,
Available online at www.sciencedirect.com R NeuroImage 18 (2003) 806 810 www.elsevier.com/locate/ynimg Technical Note Comparing event-related and epoch analysis in blocked design fmri Andrea Mechelli,
Attention: Neural Mechanisms and Attentional Control Networks Attention 2
 Attention: Neural Mechanisms and Attentional Control Networks Attention 2 Hillyard(1973) Dichotic Listening Task N1 component enhanced for attended stimuli Supports early selection Effects of Voluntary
Attention: Neural Mechanisms and Attentional Control Networks Attention 2 Hillyard(1973) Dichotic Listening Task N1 component enhanced for attended stimuli Supports early selection Effects of Voluntary
Bodies capture attention when nothing is expected
 Cognition 93 (2004) B27 B38 www.elsevier.com/locate/cognit Brief article Bodies capture attention when nothing is expected Paul E. Downing*, David Bray, Jack Rogers, Claire Childs School of Psychology,
Cognition 93 (2004) B27 B38 www.elsevier.com/locate/cognit Brief article Bodies capture attention when nothing is expected Paul E. Downing*, David Bray, Jack Rogers, Claire Childs School of Psychology,
DISTINCT BUT OVERLAPPING PATTERNS OF RESPONSE TO WORDS AND FACES IN THE FUSIFORM GYRUS
 DISTINCT BUT OVERLAPPING PATTERNS OF RESPONSE TO WORDS AND FACES IN THE FUSIFORM GYRUS Richard J. Harris, Grace E. Rice, Andrew W. Young & Timothy J. Andrews* Department of Psychology and York Neuroimaging
DISTINCT BUT OVERLAPPING PATTERNS OF RESPONSE TO WORDS AND FACES IN THE FUSIFORM GYRUS Richard J. Harris, Grace E. Rice, Andrew W. Young & Timothy J. Andrews* Department of Psychology and York Neuroimaging
Event-Related fmri and the Hemodynamic Response
 Human Brain Mapping 6:373 377(1998) Event-Related fmri and the Hemodynamic Response Randy L. Buckner 1,2,3 * 1 Departments of Psychology, Anatomy and Neurobiology, and Radiology, Washington University,
Human Brain Mapping 6:373 377(1998) Event-Related fmri and the Hemodynamic Response Randy L. Buckner 1,2,3 * 1 Departments of Psychology, Anatomy and Neurobiology, and Radiology, Washington University,
NeuroImage 70 (2013) Contents lists available at SciVerse ScienceDirect. NeuroImage. journal homepage:
 NeuroImage 70 (2013) 37 47 Contents lists available at SciVerse ScienceDirect NeuroImage journal homepage: www.elsevier.com/locate/ynimg The distributed representation of random and meaningful object pairs
NeuroImage 70 (2013) 37 47 Contents lists available at SciVerse ScienceDirect NeuroImage journal homepage: www.elsevier.com/locate/ynimg The distributed representation of random and meaningful object pairs
fmri Adaptation Studies of Viewpoint Tuning in the Extrastriate and Fusiform Body Areas
 J Neurophysiol 103: 1467 1477, 2010. First published December 23, 2009; doi:10.1152/jn.00637.2009. fmri Adaptation Studies of Viewpoint Tuning in the Extrastriate and Fusiform Body Areas John C. Taylor,
J Neurophysiol 103: 1467 1477, 2010. First published December 23, 2009; doi:10.1152/jn.00637.2009. fmri Adaptation Studies of Viewpoint Tuning in the Extrastriate and Fusiform Body Areas John C. Taylor,
Supplemental Material
 1 Supplemental Material Golomb, J.D, and Kanwisher, N. (2012). Higher-level visual cortex represents retinotopic, not spatiotopic, object location. Cerebral Cortex. Contents: - Supplemental Figures S1-S3
1 Supplemental Material Golomb, J.D, and Kanwisher, N. (2012). Higher-level visual cortex represents retinotopic, not spatiotopic, object location. Cerebral Cortex. Contents: - Supplemental Figures S1-S3
fmr-adaptation reveals a distributed representation of inanimate objects and places in human visual cortex
 www.elsevier.com/locate/ynimg NeuroImage 28 (2005) 268 279 fmr-adaptation reveals a distributed representation of inanimate objects and places in human visual cortex Michael P. Ewbank, a Denis Schluppeck,
www.elsevier.com/locate/ynimg NeuroImage 28 (2005) 268 279 fmr-adaptation reveals a distributed representation of inanimate objects and places in human visual cortex Michael P. Ewbank, a Denis Schluppeck,
Activity patterns in the category-selective occipitotemporal cortex predict upcoming motor actions
 European Journal of Neuroscience European Journal of Neuroscience, Vol. 38, pp. 2408 2424, 2013 doi:10.1111/ejn.12215 COGNITIVE NEUROSCIENCE Activity patterns in the category-selective occipitotemporal
European Journal of Neuroscience European Journal of Neuroscience, Vol. 38, pp. 2408 2424, 2013 doi:10.1111/ejn.12215 COGNITIVE NEUROSCIENCE Activity patterns in the category-selective occipitotemporal
The Role of Working Memory in Visual Selective Attention
 Goldsmiths Research Online. The Authors. Originally published: Science vol.291 2 March 2001 1803-1806. http://www.sciencemag.org. 11 October 2000; accepted 17 January 2001 The Role of Working Memory in
Goldsmiths Research Online. The Authors. Originally published: Science vol.291 2 March 2001 1803-1806. http://www.sciencemag.org. 11 October 2000; accepted 17 January 2001 The Role of Working Memory in
Investigating directed influences between activated brain areas in a motor-response task using fmri
 Magnetic Resonance Imaging 24 (2006) 181 185 Investigating directed influences between activated brain areas in a motor-response task using fmri Birgit Abler a, 4, Alard Roebroeck b, Rainer Goebel b, Anett
Magnetic Resonance Imaging 24 (2006) 181 185 Investigating directed influences between activated brain areas in a motor-response task using fmri Birgit Abler a, 4, Alard Roebroeck b, Rainer Goebel b, Anett
Neural correlates of non-familiar face processing
 Budapest University of Technology and Economics PhD School in Psychology Cognitive Science Krisztina Nagy PhD thesis booklet Supervisor: Prof. Gyula Kovács Budapest, 2013 Contents Abbreviations 2 1 Introduction
Budapest University of Technology and Economics PhD School in Psychology Cognitive Science Krisztina Nagy PhD thesis booklet Supervisor: Prof. Gyula Kovács Budapest, 2013 Contents Abbreviations 2 1 Introduction
Classification and Statistical Analysis of Auditory FMRI Data Using Linear Discriminative Analysis and Quadratic Discriminative Analysis
 International Journal of Innovative Research in Computer Science & Technology (IJIRCST) ISSN: 2347-5552, Volume-2, Issue-6, November-2014 Classification and Statistical Analysis of Auditory FMRI Data Using
International Journal of Innovative Research in Computer Science & Technology (IJIRCST) ISSN: 2347-5552, Volume-2, Issue-6, November-2014 Classification and Statistical Analysis of Auditory FMRI Data Using
Define functional MRI. Briefly describe fmri image acquisition. Discuss relative functional neuroanatomy. Review clinical applications.
 Dr. Peter J. Fiester November 14, 2012 Define functional MRI. Briefly describe fmri image acquisition. Discuss relative functional neuroanatomy. Review clinical applications. Briefly discuss a few examples
Dr. Peter J. Fiester November 14, 2012 Define functional MRI. Briefly describe fmri image acquisition. Discuss relative functional neuroanatomy. Review clinical applications. Briefly discuss a few examples
Representing connected and disconnected shapes in human inferior intraparietal sulcus
 www.elsevier.com/locate/ynimg NeuroImage 40 (2008) 1849 1856 Representing connected and disconnected shapes in human inferior intraparietal sulcus Yaoda Xu Department of Psychology, Yale University, Box
www.elsevier.com/locate/ynimg NeuroImage 40 (2008) 1849 1856 Representing connected and disconnected shapes in human inferior intraparietal sulcus Yaoda Xu Department of Psychology, Yale University, Box
Neuropsychologia ] (]]]]) ]]] ]]] Contents lists available at SciVerse ScienceDirect. Neuropsychologia
![Neuropsychologia ] (]]]]) ]]] ]]] Contents lists available at SciVerse ScienceDirect. Neuropsychologia Neuropsychologia ] (]]]]) ]]] ]]] Contents lists available at SciVerse ScienceDirect. Neuropsychologia](/thumbs/85/91347702.jpg) Neuropsychologia ] (]]]]) ]]] ]]] Contents lists available at SciVerse ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia The response of face-selective cortex with
Neuropsychologia ] (]]]]) ]]] ]]] Contents lists available at SciVerse ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia The response of face-selective cortex with
Virtual Brain Reading: A Connectionist Approach to Understanding fmri
 Virtual Brain Reading: A Connectionist Approach to Understanding fmri Rosemary A. Cowell (r.a.cowell@kent.ac.uk) Computing Laboratory, University of Kent, Canterbury, CT2 7NF, UK David E. Huber (dhuber@psy.ucsd.edu)
Virtual Brain Reading: A Connectionist Approach to Understanding fmri Rosemary A. Cowell (r.a.cowell@kent.ac.uk) Computing Laboratory, University of Kent, Canterbury, CT2 7NF, UK David E. Huber (dhuber@psy.ucsd.edu)
ASSESSING MULTISENSORY INTEGRATION WITH ADDITIVE FACTORS AND FUNCTIONAL MRI
 ASSESSING MULTISENSORY INTEGRATION WITH ADDITIVE FACTORS AND FUNCTIONAL MRI Thomas W. James, Ryan A. Stevenson and Sunah Kim Department of Psychological and Brain Sciences, Indiana University, 1101 E Tenth
ASSESSING MULTISENSORY INTEGRATION WITH ADDITIVE FACTORS AND FUNCTIONAL MRI Thomas W. James, Ryan A. Stevenson and Sunah Kim Department of Psychological and Brain Sciences, Indiana University, 1101 E Tenth
HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006
 MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
Top-Down Activation of Fusiform Cortex without Seeing Faces in Prosopagnosia
 Cerebral Cortex August 2010;20:1878--1890 doi:10.1093/cercor/bhp254 Advance Access publication November 25, 2009 Top-Down Activation of Fusiform Cortex without Seeing Faces in Prosopagnosia Ruthger Righart
Cerebral Cortex August 2010;20:1878--1890 doi:10.1093/cercor/bhp254 Advance Access publication November 25, 2009 Top-Down Activation of Fusiform Cortex without Seeing Faces in Prosopagnosia Ruthger Righart
Face-specific resting functional connectivity between the fusiform gyrus and posterior superior temporal sulcus
 HUMAN NEUROSCIENCE Original Research Article published: 24 September 2010 doi: 10.3389/fnhum.2010.00176 Face-specific resting functional connectivity between the fusiform gyrus and posterior superior temporal
HUMAN NEUROSCIENCE Original Research Article published: 24 September 2010 doi: 10.3389/fnhum.2010.00176 Face-specific resting functional connectivity between the fusiform gyrus and posterior superior temporal
HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2008
 MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
ARTICLE IN PRESS. Neuropsychologia xxx (2009) xxx xxx. Contents lists available at ScienceDirect. Neuropsychologia
 Neuropsychologia xxx (2009) xxx xxx Contents lists available at ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia Abnormal face identity coding in the middle fusiform
Neuropsychologia xxx (2009) xxx xxx Contents lists available at ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia Abnormal face identity coding in the middle fusiform
Functional Magnetic Resonance Imaging of Human Visual Cortex during Face Matching: A Comparison with Positron Emission Tomography
 NEUROIMAGE 4, 1 15 (1996) ARTICLE NO. 0025 Functional Magnetic Resonance Imaging of Human Visual Cortex during Face Matching: A Comparison with Positron Emission Tomography V. P. CLARK, K. KEIL, J. MA.
NEUROIMAGE 4, 1 15 (1996) ARTICLE NO. 0025 Functional Magnetic Resonance Imaging of Human Visual Cortex during Face Matching: A Comparison with Positron Emission Tomography V. P. CLARK, K. KEIL, J. MA.
Abstract Representations of Object-Directed Action in the Left Inferior Parietal Lobule
 Cerebral Cortex, June 2018;28: 2162 2174 doi: 10.1093/cercor/bhx120 Advance Access Publication Date: 9 June 2017 Original Article ORIGINAL ARTICLE Abstract Representations of Object-Directed Action in
Cerebral Cortex, June 2018;28: 2162 2174 doi: 10.1093/cercor/bhx120 Advance Access Publication Date: 9 June 2017 Original Article ORIGINAL ARTICLE Abstract Representations of Object-Directed Action in
Supplemental Information. Direct Electrical Stimulation in the Human Brain. Disrupts Melody Processing
 Current Biology, Volume 27 Supplemental Information Direct Electrical Stimulation in the Human Brain Disrupts Melody Processing Frank E. Garcea, Benjamin L. Chernoff, Bram Diamond, Wesley Lewis, Maxwell
Current Biology, Volume 27 Supplemental Information Direct Electrical Stimulation in the Human Brain Disrupts Melody Processing Frank E. Garcea, Benjamin L. Chernoff, Bram Diamond, Wesley Lewis, Maxwell
Category-Specific Organization in the Human Brain Does Not Require Visual Experience
 Article Category-Specific Organization in the Human Brain Does Not Require Visual Experience Bradford Z. Mahon,,,3, * Stefano Anzellotti,, Jens Schwarzbach,,4 Massimiliano Zampini,,4 and Alfonso Caramazza,,
Article Category-Specific Organization in the Human Brain Does Not Require Visual Experience Bradford Z. Mahon,,,3, * Stefano Anzellotti,, Jens Schwarzbach,,4 Massimiliano Zampini,,4 and Alfonso Caramazza,,
SUPPLEMENTARY INFORMATION
 doi: doi:10.1038/nature08103 10.1038/nature08103 Supplementary Figure 1. Sample pictures. Examples of the natural scene pictures used in the main experiment. www.nature.com/nature 1 Supplementary Figure
doi: doi:10.1038/nature08103 10.1038/nature08103 Supplementary Figure 1. Sample pictures. Examples of the natural scene pictures used in the main experiment. www.nature.com/nature 1 Supplementary Figure
Visual areas involved in the perception of human movement from dynamic form analysis
 BRAIN IMAGING Visual areas involved in the perception of human movement from dynamic form analysis Lars Michels, 1,CA Markus Lappe 1 and Lucia Maria Vaina 2,3 1 Psychologisches Institut II,WestfÌlische
BRAIN IMAGING Visual areas involved in the perception of human movement from dynamic form analysis Lars Michels, 1,CA Markus Lappe 1 and Lucia Maria Vaina 2,3 1 Psychologisches Institut II,WestfÌlische
Multivariate Patterns in Object-Selective Cortex Dissociate Perceptual and Physical Shape Similarity
 Multivariate Patterns in Object-Selective Cortex Dissociate Perceptual and Physical Shape Similarity Johannes Haushofer 1,2*, Margaret S. Livingstone 1, Nancy Kanwisher 2 PLoS BIOLOGY 1 Department of Neurobiology,
Multivariate Patterns in Object-Selective Cortex Dissociate Perceptual and Physical Shape Similarity Johannes Haushofer 1,2*, Margaret S. Livingstone 1, Nancy Kanwisher 2 PLoS BIOLOGY 1 Department of Neurobiology,
Vision Research 51 (2011) Contents lists available at ScienceDirect. Vision Research. journal homepage:
 Vision Research 51 (2011) 195 202 Contents lists available at ScienceDirect Vision Research journal homepage: www.elsevier.com/locate/visres Differences between perception of human faces and body shapes:
Vision Research 51 (2011) 195 202 Contents lists available at ScienceDirect Vision Research journal homepage: www.elsevier.com/locate/visres Differences between perception of human faces and body shapes:
Specificity of Action Representations in the Lateral Occipitotemporal Cortex
 University of Pennsylvania ScholarlyCommons Departmental Papers (Psychology) Department of Psychology 9-2006 Specificity of Action Representations in the Lateral Occipitotemporal Cortex Joseph W. Kable
University of Pennsylvania ScholarlyCommons Departmental Papers (Psychology) Department of Psychology 9-2006 Specificity of Action Representations in the Lateral Occipitotemporal Cortex Joseph W. Kable
York, YO10 5DD, UK. *Corresponding author: Figures: 4. Tables: 2. Abstract: 291.
 RESPONSES IN THE RIGHT POSTERIOR SUPERIOR TEMPORAL SULCUS SHOW A FEATURE- BASED RESPONSE TO FACIAL EXPRESSION Tessa R. Flack, Timothy J. Andrews, Mark Hymers, Mohammed Al-Mosaiwi, Samuel P. Marsden, James
RESPONSES IN THE RIGHT POSTERIOR SUPERIOR TEMPORAL SULCUS SHOW A FEATURE- BASED RESPONSE TO FACIAL EXPRESSION Tessa R. Flack, Timothy J. Andrews, Mark Hymers, Mohammed Al-Mosaiwi, Samuel P. Marsden, James
Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla
 Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla Atsushi Miki*, Grant T. Liu*, Sarah A. Englander, Jonathan Raz, Theo G. M. van Erp,
Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla Atsushi Miki*, Grant T. Liu*, Sarah A. Englander, Jonathan Raz, Theo G. M. van Erp,
Individuating faces and common objects produces equal responses in putative face-processing areas in the ventral occipitotemporal cortex
 HUMAN NEUROSCIENCE Original Research Article published: 08 October 2010 doi: 10.3389/fnhum.2010.00181 Individuating faces and common objects produces equal responses in putative face-processing areas in
HUMAN NEUROSCIENCE Original Research Article published: 08 October 2010 doi: 10.3389/fnhum.2010.00181 Individuating faces and common objects produces equal responses in putative face-processing areas in
Impaired Face Discrimination in Acquired Prosopagnosia Is Associated with Abnormal Response to Individual Faces in the Right Middle Fusiform Gyrus
 Cerebral Cortex April 2006;16:574-586 doi:10.1093/cercor/bhj005 Advance Access publication July 20, 2005 Impaired Face Discrimination in Acquired Prosopagnosia Is Associated with Abnormal Response to Individual
Cerebral Cortex April 2006;16:574-586 doi:10.1093/cercor/bhj005 Advance Access publication July 20, 2005 Impaired Face Discrimination in Acquired Prosopagnosia Is Associated with Abnormal Response to Individual
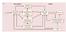 Peripheral facial paralysis (right side). The patient is asked to close her eyes and to retract their mouth (From Heimer) Hemiplegia of the left side. Note the characteristic position of the arm with
Peripheral facial paralysis (right side). The patient is asked to close her eyes and to retract their mouth (From Heimer) Hemiplegia of the left side. Note the characteristic position of the arm with
Nature versus Nurture in Ventral Visual Cortex: A Functional Magnetic Resonance Imaging Study of Twins
 The Journal of Neuroscience, December 19, 2007 27(51):13921 13925 13921 Brief Communications Nature versus Nurture in Ventral Visual Cortex: A Functional Magnetic Resonance Imaging Study of Twins Thad
The Journal of Neuroscience, December 19, 2007 27(51):13921 13925 13921 Brief Communications Nature versus Nurture in Ventral Visual Cortex: A Functional Magnetic Resonance Imaging Study of Twins Thad
FFA: a flexible fusiform area for subordinate-level visual processing automatized by expertise
 commentary FFA: a flexible fusiform area for subordinate-level visual processing automatized by expertise Michael J. Tarr and Isabel Gauthier Much evidence suggests that the fusiform face area is involved
commentary FFA: a flexible fusiform area for subordinate-level visual processing automatized by expertise Michael J. Tarr and Isabel Gauthier Much evidence suggests that the fusiform face area is involved
FINAL PROGRESS REPORT
 (1) Foreword (optional) (2) Table of Contents (if report is more than 10 pages) (3) List of Appendixes, Illustrations and Tables (if applicable) (4) Statement of the problem studied FINAL PROGRESS REPORT
(1) Foreword (optional) (2) Table of Contents (if report is more than 10 pages) (3) List of Appendixes, Illustrations and Tables (if applicable) (4) Statement of the problem studied FINAL PROGRESS REPORT
9.71 Functional MRI of High-Level Vision Fall 2007
 MIT OpenCourseWare http://ocw.mit.edu 9.71 Functional MRI of High-Level Vision Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Differential
MIT OpenCourseWare http://ocw.mit.edu 9.71 Functional MRI of High-Level Vision Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Differential
Chantal E. Stern,*, Adrian M. Owen, Irene Tracey,*, Rodney B. Look,* Bruce R. Rosen,* and Michael Petrides
 NeuroImage 11, 392 399 (2000) doi:10.1006/nimg.2000.0569, available online at http://www.idealibrary.com on Activity in Ventrolateral and Mid-Dorsolateral Prefrontal Cortex during Nonspatial Visual Working
NeuroImage 11, 392 399 (2000) doi:10.1006/nimg.2000.0569, available online at http://www.idealibrary.com on Activity in Ventrolateral and Mid-Dorsolateral Prefrontal Cortex during Nonspatial Visual Working
Men fear other men most: Gender specific brain activations in. perceiving threat from dynamic faces and bodies. An fmri. study.
 Men fear other men most: Gender specific brain activations in perceiving threat from dynamic faces and bodies. An fmri study. Kret, ME, Pichon, S 2,4, Grèzes, J 2, & de Gelder, B,3 Cognitive and Affective
Men fear other men most: Gender specific brain activations in perceiving threat from dynamic faces and bodies. An fmri study. Kret, ME, Pichon, S 2,4, Grèzes, J 2, & de Gelder, B,3 Cognitive and Affective
Predicting Perceptual Performance From Neural Activity
 Predicting Perceptual Performance From Neural Activity Koel Das 2, Sheng Li 3, Barry Giesbrecht 1, 2, Zoe Kourtzi 4, Miguel P. Eckstein 1, 2 1 Institute for Collaborative Biotechnologies 2 Department of
Predicting Perceptual Performance From Neural Activity Koel Das 2, Sheng Li 3, Barry Giesbrecht 1, 2, Zoe Kourtzi 4, Miguel P. Eckstein 1, 2 1 Institute for Collaborative Biotechnologies 2 Department of
Excellent Network Courses. Department of Neurology Affiliated hospital of Jiangsu University
 Excellent Network Courses Department of Neurology Affiliated hospital of Jiangsu University Agnosia Visual Agnosia Lissauer (1890) described 2 types: a) Apperceptive Cannot see objects b) Associative Does
Excellent Network Courses Department of Neurology Affiliated hospital of Jiangsu University Agnosia Visual Agnosia Lissauer (1890) described 2 types: a) Apperceptive Cannot see objects b) Associative Does
Brain activity related to integrative processes in visual object recognition: bottom-up integration and the modulatory influence of stored knowledge
 Neuropsychologia 40 (2002) 1254 1267 Brain activity related to integrative processes in visual object recognition: bottom-up integration and the modulatory influence of stored knowledge C. Gerlach a,,
Neuropsychologia 40 (2002) 1254 1267 Brain activity related to integrative processes in visual object recognition: bottom-up integration and the modulatory influence of stored knowledge C. Gerlach a,,
Ilan Dinstein, Uri Hasson, Nava Rubin and David J. Heeger
 Ilan Dinstein, Uri Hasson, Nava Rubin and David J. Heeger J Neurophysiol 98:1415-1427, 2007. First published Jun 27, 2007; doi:10.1152/jn.00238.2007 You might find this additional information useful...
Ilan Dinstein, Uri Hasson, Nava Rubin and David J. Heeger J Neurophysiol 98:1415-1427, 2007. First published Jun 27, 2007; doi:10.1152/jn.00238.2007 You might find this additional information useful...
Title:Atypical language organization in temporal lobe epilepsy revealed by a passive semantic paradigm
 Author's response to reviews Title:Atypical language organization in temporal lobe epilepsy revealed by a passive semantic paradigm Authors: Julia Miro (juliamirollado@gmail.com) Pablo Ripollès (pablo.ripolles.vidal@gmail.com)
Author's response to reviews Title:Atypical language organization in temporal lobe epilepsy revealed by a passive semantic paradigm Authors: Julia Miro (juliamirollado@gmail.com) Pablo Ripollès (pablo.ripolles.vidal@gmail.com)
Extrastriate body area in human occipital cortex responds to the performance of motor actions
 Extrastriate body area in human occipital cortex responds to the performance of motor actions Serguei V Astafiev 1,Christine M Stanley 1,Gordon L Shulman 2 & Maurizio Corbetta 1 3 A region in human lateral
Extrastriate body area in human occipital cortex responds to the performance of motor actions Serguei V Astafiev 1,Christine M Stanley 1,Gordon L Shulman 2 & Maurizio Corbetta 1 3 A region in human lateral
FRONTAL LOBE. Central Sulcus. Ascending ramus of the Cingulate Sulcus. Cingulate Sulcus. Lateral Sulcus
 FRONTAL LOBE Central Ascending ramus of the Cingulate Cingulate Lateral Lateral View Medial View Motor execution and higher cognitive functions (e.g., language production, impulse inhibition, reasoning
FRONTAL LOBE Central Ascending ramus of the Cingulate Cingulate Lateral Lateral View Medial View Motor execution and higher cognitive functions (e.g., language production, impulse inhibition, reasoning
Representation of Shapes, Edges, and Surfaces Across Multiple Cues in the Human
 Page 1 of 50 Articles in PresS. J Neurophysiol (January 2, 2008). doi:10.1152/jn.01223.2007 Representation of Shapes, Edges, and Surfaces Across Multiple Cues in the Human Visual Cortex. Joakim Vinberg
Page 1 of 50 Articles in PresS. J Neurophysiol (January 2, 2008). doi:10.1152/jn.01223.2007 Representation of Shapes, Edges, and Surfaces Across Multiple Cues in the Human Visual Cortex. Joakim Vinberg
Investigations in Resting State Connectivity. Overview
 Investigations in Resting State Connectivity Scott FMRI Laboratory Overview Introduction Functional connectivity explorations Dynamic change (motor fatigue) Neurological change (Asperger s Disorder, depression)
Investigations in Resting State Connectivity Scott FMRI Laboratory Overview Introduction Functional connectivity explorations Dynamic change (motor fatigue) Neurological change (Asperger s Disorder, depression)
Visual Context Dan O Shea Prof. Fei Fei Li, COS 598B
 Visual Context Dan O Shea Prof. Fei Fei Li, COS 598B Cortical Analysis of Visual Context Moshe Bar, Elissa Aminoff. 2003. Neuron, Volume 38, Issue 2, Pages 347 358. Visual objects in context Moshe Bar.
Visual Context Dan O Shea Prof. Fei Fei Li, COS 598B Cortical Analysis of Visual Context Moshe Bar, Elissa Aminoff. 2003. Neuron, Volume 38, Issue 2, Pages 347 358. Visual objects in context Moshe Bar.
Cortical Organization. Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:.
 Cortical Organization Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:. 2. Secondary cortex: located immediately adjacent to primary cortical areas,
Cortical Organization Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:. 2. Secondary cortex: located immediately adjacent to primary cortical areas,
Personal Space Regulation by the Human Amygdala. California Institute of Technology
 Personal Space Regulation by the Human Amygdala Daniel P. Kennedy 1, Jan Gläscher 1, J. Michael Tyszka 2 & Ralph Adolphs 1,2 1 Division of Humanities and Social Sciences 2 Division of Biology California
Personal Space Regulation by the Human Amygdala Daniel P. Kennedy 1, Jan Gläscher 1, J. Michael Tyszka 2 & Ralph Adolphs 1,2 1 Division of Humanities and Social Sciences 2 Division of Biology California
Supplementary Note Psychophysics:
 Supplementary Note More detailed description of MM s subjective experiences can be found on Mike May s Perceptions Home Page, http://www.senderogroup.com/perception.htm Psychophysics: The spatial CSF was
Supplementary Note More detailed description of MM s subjective experiences can be found on Mike May s Perceptions Home Page, http://www.senderogroup.com/perception.htm Psychophysics: The spatial CSF was
Parietal representations of stimulus features are amplified during memory retrieval and flexibly aligned with top-down goals
 This Accepted Manuscript has not been copyedited and formatted. The final version may differ from this version. Research Articles: Behavioral/Cognitive Parietal representations of stimulus features are
This Accepted Manuscript has not been copyedited and formatted. The final version may differ from this version. Research Articles: Behavioral/Cognitive Parietal representations of stimulus features are
Functional MRI Mapping Cognition
 Outline Functional MRI Mapping Cognition Michael A. Yassa, B.A. Division of Psychiatric Neuro-imaging Psychiatry and Behavioral Sciences Johns Hopkins School of Medicine Why fmri? fmri - How it works Research
Outline Functional MRI Mapping Cognition Michael A. Yassa, B.A. Division of Psychiatric Neuro-imaging Psychiatry and Behavioral Sciences Johns Hopkins School of Medicine Why fmri? fmri - How it works Research
An Integrated Face Body Representation in the Fusiform Gyrus but Not the Lateral Occipital Cortex
 An Integrated Face Body Representation in the Fusiform Gyrus but Not the Lateral Occipital Cortex Michal Bernstein, Jonathan Oron, Boaz Sadeh, and Galit Yovel Abstract Faces and bodies are processed by
An Integrated Face Body Representation in the Fusiform Gyrus but Not the Lateral Occipital Cortex Michal Bernstein, Jonathan Oron, Boaz Sadeh, and Galit Yovel Abstract Faces and bodies are processed by
Functional organization of spatial and nonspatial working memory processing within the human lateral frontal cortex
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 7721 7726, June 1998 Neurobiology Functional organization of spatial and nonspatial working memory processing within the human lateral frontal cortex ADRIAN M. OWEN*,
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 7721 7726, June 1998 Neurobiology Functional organization of spatial and nonspatial working memory processing within the human lateral frontal cortex ADRIAN M. OWEN*,
Mirror Neuron System Differentially Activated by Facial Expressions and Social Hand Gestures: A Functional Magnetic Resonance Imaging Study
 Mirror Neuron System Differentially Activated by Facial Expressions and Social Hand Gestures: A Functional Magnetic Resonance Imaging Study Kimberly J. Montgomery and James V. Haxby Abstract & Facial expressions
Mirror Neuron System Differentially Activated by Facial Expressions and Social Hand Gestures: A Functional Magnetic Resonance Imaging Study Kimberly J. Montgomery and James V. Haxby Abstract & Facial expressions
Pennsylvania; and 5 Maastricht Brain Imaging Center, Maastrict University, Maastrict, The Netherlands
 J Neurophysiol 106: 2720 2736, 2011. First published July 6, 2011; doi:10.1152/jn.00672.2010. Face categorization in visual scenes may start in a higher order area of the right fusiform gyrus: evidence
J Neurophysiol 106: 2720 2736, 2011. First published July 6, 2011; doi:10.1152/jn.00672.2010. Face categorization in visual scenes may start in a higher order area of the right fusiform gyrus: evidence
Left Anterior Prefrontal Activation Increases with Demands to Recall Specific Perceptual Information
 The Journal of Neuroscience, 2000, Vol. 20 RC108 1of5 Left Anterior Prefrontal Activation Increases with Demands to Recall Specific Perceptual Information Charan Ranganath, 1 Marcia K. Johnson, 2 and Mark
The Journal of Neuroscience, 2000, Vol. 20 RC108 1of5 Left Anterior Prefrontal Activation Increases with Demands to Recall Specific Perceptual Information Charan Ranganath, 1 Marcia K. Johnson, 2 and Mark
Key questions about attention
 Key questions about attention How does attention affect behavioral performance? Can attention affect the appearance of things? How does spatial and feature-based attention affect neuronal responses in
Key questions about attention How does attention affect behavioral performance? Can attention affect the appearance of things? How does spatial and feature-based attention affect neuronal responses in
Domain Specificity in Visual Cortex
 Cerebral Cortex Advance Access published December 7, 2005 Cerebral Cortex doi:10.1093/cercor/bhj086 Domain Specificity in Visual Cortex P. E. Downing 1, A. W.-Y. Chan 1, M. V. Peelen 1, C. M. Dodds 2 and
Cerebral Cortex Advance Access published December 7, 2005 Cerebral Cortex doi:10.1093/cercor/bhj086 Domain Specificity in Visual Cortex P. E. Downing 1, A. W.-Y. Chan 1, M. V. Peelen 1, C. M. Dodds 2 and
Selective Attention. Modes of Control. Domains of Selection
 The New Yorker (2/7/5) Selective Attention Perception and awareness are necessarily selective (cell phone while driving): attention gates access to awareness Selective attention is deployed via two modes
The New Yorker (2/7/5) Selective Attention Perception and awareness are necessarily selective (cell phone while driving): attention gates access to awareness Selective attention is deployed via two modes
Functional Magnetic Resonance Imaging with Arterial Spin Labeling: Techniques and Potential Clinical and Research Applications
 pissn 2384-1095 eissn 2384-1109 imri 2017;21:91-96 https://doi.org/10.13104/imri.2017.21.2.91 Functional Magnetic Resonance Imaging with Arterial Spin Labeling: Techniques and Potential Clinical and Research
pissn 2384-1095 eissn 2384-1109 imri 2017;21:91-96 https://doi.org/10.13104/imri.2017.21.2.91 Functional Magnetic Resonance Imaging with Arterial Spin Labeling: Techniques and Potential Clinical and Research
11, in nonhuman primates, different cell populations respond to facial identity and expression12, and
 UNDERSTANDING THE RECOGNITION OF FACIAL IDENTITY AND FACIAL EXPRESSION Andrew J. Calder* and Andrew W. Young Abstract Faces convey a wealth of social signals. A dominant view in face-perception research
UNDERSTANDING THE RECOGNITION OF FACIAL IDENTITY AND FACIAL EXPRESSION Andrew J. Calder* and Andrew W. Young Abstract Faces convey a wealth of social signals. A dominant view in face-perception research
Journal of Neuroscience. For Peer Review Only
 The Journal of Neuroscience Discrimination Training Alters Object Representations in Human Extrastriate Cortex Journal: Manuscript ID: Manuscript Type: Manuscript Section: Date Submitted by the Author:
The Journal of Neuroscience Discrimination Training Alters Object Representations in Human Extrastriate Cortex Journal: Manuscript ID: Manuscript Type: Manuscript Section: Date Submitted by the Author:
Visuo-haptic object-related activation in the ventral visual pathway
 Visuo-haptic object-related activation in the ventral visual pathway Amir Amedi 1, Rafael Malach 2, Talma Hendler 3, Sharon Peled 3 and Ehud Zohary 1 1 Neurobiology Department, Life Science Institute and
Visuo-haptic object-related activation in the ventral visual pathway Amir Amedi 1, Rafael Malach 2, Talma Hendler 3, Sharon Peled 3 and Ehud Zohary 1 1 Neurobiology Department, Life Science Institute and
The Central Nervous System
 The Central Nervous System Cellular Basis. Neural Communication. Major Structures. Principles & Methods. Principles of Neural Organization Big Question #1: Representation. How is the external world coded
The Central Nervous System Cellular Basis. Neural Communication. Major Structures. Principles & Methods. Principles of Neural Organization Big Question #1: Representation. How is the external world coded
Mirror neurons. Romana Umrianova
 Mirror neurons Romana Umrianova The functional role of the parieto-frontal mirror circuit: interpretations and misinterpretations Giacomo Rizzolatti and Corrado Sinigaglia Mechanism that unifies action
Mirror neurons Romana Umrianova The functional role of the parieto-frontal mirror circuit: interpretations and misinterpretations Giacomo Rizzolatti and Corrado Sinigaglia Mechanism that unifies action
Ventral and Dorsal Pathways Differently Related to Visual Awareness of Body Postures under Continuous Flash Suppression
 This Accepted Manuscript has not been copyedited and formatted. The final version may differ from this version. A link to any extended data will be provided when the final version is posted online. Research
This Accepted Manuscript has not been copyedited and formatted. The final version may differ from this version. A link to any extended data will be provided when the final version is posted online. Research
Is a small apple more like an apple or more like a cherry? A study with real and modified sized objects.
 Is a small apple more like an apple or more like a cherry? A study with real and modified sized objects. Valentina Bazzarin (valentina.bazzarin@unibo.it) Department of Psychology, 5 Viale Berti Pichat
Is a small apple more like an apple or more like a cherry? A study with real and modified sized objects. Valentina Bazzarin (valentina.bazzarin@unibo.it) Department of Psychology, 5 Viale Berti Pichat
