Nasal High-Flow Therapy for Primary Respiratory Support in Preterm Infants
|
|
|
- Shannon French
- 6 years ago
- Views:
Transcription
1 The new england journal of medicine Original Article Nasal High-Flow Therapy for Primary Respiratory Support in Preterm Infants Calum T. Roberts, M.B., Ch.B., Louise S. Owen, M.D., Brett J. Manley, Ph.D., Dag H. Frøisland, Ph.D., Susan M. Donath, M.A., Kim M. Dalziel, Ph.D., Margo A. Pritchard, Ph.D., David W. Cartwright, M.B., B.S., Clare L. Collins, M.D., Atul Malhotra, M.D., and Peter G. Davis, M.D., for the HIPSTER Trial Investigators* ABSTRACT From Neonatal Services and Newborn Research Centre, Royal Women s Hospital (C.T.R., L.S.O., B.J.M., D.H.F., P.G.D.), the Departments of Obstetrics and Gynaecology (C.T.R., L.S.O., B.J.M., P.G.D.) and Paediatrics (S.M.D.) and School of Population and Global Health (K.M.D.), University of Melbourne, Critical Care and Neurosciences (L.S.O., P.G.D.) and Clinical Epidemiology and Biostatistics Unit (S.M.D.), Murdoch Children s Research Institute, Neonatal Services, Mercy Hospital for Women (C.L.C.), and Monash Newborn, Monash Children s Hospital, and Department of Paediatrics, Monash University (A.M.), Melbourne, VIC, and the School of Nursing, Midwifery and Paramedicine, Australian Catholic University (M.A.P.), Mater Research Institute (M.A.P.) and the Department of Paediatrics (D.W.C.), University of Queensland, and Women s and Newborn Services, Royal Brisbane and Women s Hospital (D.W.C.), Brisbane, QLD all in Australia; and the Department of Pediatrics, Innlandet Hospital Trust, Lillehammer, Norway (D.H.F.). Address reprint requests to Dr. Owen at the Newborn Research Centre, Level 7, Royal Women s Hospital, Locked Bag 300, Parkville, VIC 3052, Australia, or at louise.owen@thewomens.org.au. * A complete list of investigators in the High Flow Nasal Cannulae as Primary Support in the Treatment of Early Respiratory Distress (HIPSTER) Trial is provided in the Supplementary Appendix, available at NEJM.org. Drs. Roberts and Owen contributed equally to this article. N Engl J Med 2016;375: DOI: /NEJMoa Copyright 2016 Massachusetts Medical Society. BACKGROUND Treatment with nasal high-flow therapy has efficacy similar to that of nasal continuous positive airway pressure (CPAP) when used as postextubation support in neonates. The efficacy of high-flow therapy as the primary means of respiratory support for preterm infants with respiratory distress has not been proved. METHODS In this international, multicenter, randomized, noninferiority trial, we assigned 564 preterm infants (gestational age, 28 weeks 0 days) with early respiratory distress who had not received surfactant replacement to treatment with either nasal high-flow therapy or nasal CPAP. The primary outcome was treatment failure within 72 hours after randomization. Noninferiority was determined by calculating the absolute difference in the risk of the primary outcome; the chosen margin of noninferiority was 10 percentage points. Infants in whom high-flow therapy failed could receive rescue CPAP; infants in whom CPAP failed were intubated and mechanically ventilated. RESULTS Trial recruitment stopped early at the recommendation of the independent data and safety monitoring committee because of a significant difference in the primary outcome between treatment groups. Treatment failure occurred in 71 of 278 infants (25.5%) in the high-flow group and in 38 of 286 infants (13.3%) in the CPAP group (risk difference, 12.3 percentage points; 95% confidence interval [CI], 5.8 to 18.7; P<0.001). The rate of intubation within 72 hours did not differ significantly between the high-flow and CPAP groups (15.5% and 11.5%, respectively; risk difference, 3.9 percentage points; 95% CI, 1.7 to 9.6; P = 0.17), nor did the rate of adverse events. CONCLUSIONS When used as primary support for preterm infants with respiratory distress, highflow therapy resulted in a significantly higher rate of treatment failure than did CPAP. (Funded by the National Health and Medical Research Council and others; Australian New Zealand Clinical Trials Registry number, ACTRN ) 1142 n engl j med 375;12 nejm.org September 22, 2016
2 Nasal High-Flow Therapy in Preterm Infants In 2014, there were more than 380,000 preterm births (i.e., births at a gestational age of <37 weeks) in the United States, accounting for approximately 10% of all births that year. 1 Preterm infants have a risk of the respiratory distress syndrome. The introduction of endotracheal ventilation has improved the survival rate among preterm infants but is associated with an increased risk of complications such as bronchopulmonary dysplasia. 2 Clinicians aim to use noninvasive respiratory support to minimize the risk of such complications. The most widely used noninvasive approach, nasal continuous positive airway pressure (CPAP), has been shown to be an effective alternative to endotracheal ventilation as primary respiratory support for preterm infants. 3,4 Treatment with heated, humidified, high-flow nasal cannulae (high-flow therapy) is an increasingly popular means of noninvasive respiratory support. Surveys have shown that approximately two thirds of neonatal intensive care units in the United States 5 and in Australia and New Zealand 6 used high-flow therapy. This approach has several reported advantages over CPAP, including reduced rates of nasal trauma 7-9 and reduced infant pain scores. 10 Surveys show that it is preferred by parents 11 and nursing staff. 12 In a previous randomized trial comparing high-flow therapy with CPAP as respiratory support after extubation in infants born at a gestational age of less than 32 weeks, we found that high-flow therapy was noninferior to CPAP in preventing treatment failure. 8 This finding was consistent with the results of other randomized trials of neonatal respiratory support after extubation. 7,9 Previous studies comparing high-flow therapy with CPAP as primary support have not shown significant differences in treatment-failure or intubation rates. However, these studies were small, single-center trials, 13,14 reported interim data, 15 or constituted a substudy of a larger trial. 9 The authors of a recent Cochrane Review suggested that additional, adequately powered randomized trials assessing high-flow therapy as primary respiratory support should be undertaken. 16 We performed an international, multicenter, randomized, controlled trial to test the hypothesis that high-flow therapy would be noninferior to CPAP as primary respiratory support for preterm infants (gestational age, 28 weeks 0 days) with early respiratory distress. Methods Study Design and Oversight Nine neonatal intensive care units in Australia and Norway participated in the study. The human research ethics committee at each participating center approved the study. All authors vouch for the accuracy and completeness of the data and for the fidelity of the study to the protocol, which was published previously 17 and is available with the full text of this article at NEJM.org. The study had no commercial support, and the respiratory device manufacturers had no input in study design, data accrual, data analysis, or manuscript preparation and no access to the study data. Patients Infants were eligible for inclusion if they were born at a gestational age of 28 weeks 0 days to 36 weeks 6 days, were less than 24 hours old, and had not previously received endotracheal ventilation or surfactant treatment and if the attending clinician had decided to commence or continue noninvasive respiratory support. Infants were ineligible if there was an urgent need for intubation and ventilation (as determined by the attending clinician) or if they had already met the criteria for treatment failure, had a known major congenital abnormality or pneumothorax, or had received 4 hours or more of CPAP support. Recruitment and Consent The parents of all participating infants provided written informed consent. At all sites, antepartum consent was sought when possible. If antepartum consent was obtained, infants were randomly assigned to a study group as soon as they met the eligibility criteria. If antepartum consent was not sought, parents of eligible infants were approached at the earliest opportunity after birth. In addition, at the lead center (the Royal Women s Hospital, Melbourne, Australia), the human research ethics committee approved a retrospective consent process (see Section 2.2 in the Supplementary Appendix, available at NEJM.org). Randomization A computer-generated randomization sequence with variable block sizes was used. Infants were stratified according to gestational age (<32 weeks vs. 32 weeks) and study center. Sequentially n engl j med 375;12 nejm.org September 22,
3 The new england journal of medicine numbered, sealed, opaque envelopes containing the treatment assignment were opened as soon as both eligibility and consent criteria had been met. Study Intervention Eligible infants were randomly assigned to treatment with either high-flow therapy or CPAP. Infants weighing 1250 g or less received caffeine (intravenous loading dose, 20 mg per kilogram of body weight) on the first day of life. Other aspects of care were provided according to individual unit protocols. Infants randomly assigned to the high-flow group received an initial gas flow of 6 to 8 liters per minute, from either the Optiflow Junior (Fisher and Paykel Healthcare) or Precision Flow (Vapotherm) device. The size of the nasal cannulae was determined according to the manufacturers instructions in order to maintain a leak at the nares. The maximum permissible gas flow was 8 liters per minute, as recommended by the manufacturer. Infants assigned to highflow therapy who met the criteria for treatment failure could receive CPAP as rescue therapy, initiated at 7 to 8 cm of water. Infants who continued to meet treatment-failure criteria were intubated and ventilated. In the infants randomly assigned to CPAP, the starting pressure was 6 to 8 cm of water, achieved with a ventilator, an underwater bubble system, or a variable-flow device. Treatment was delivered through either short binasal prongs or a nasal mask, according to the protocol at each participating center, with sizing determined according to the manufacturer s recommendations. The maximum permissible pressure was 8 cm of water. Infants treated with CPAP who met the criteria for treatment failure were intubated and ventilated. Changes in respiratory support were made in steps of 1 liter per minute (for high-flow therapy) or 1 cm of water (for CPAP). All infants were evaluated at least daily. Weaning from noninvasive respiratory support was considered if there was clinical improvement and the infants were receiving a fraction of inspired oxygen of 0.3 or lower, whereas discontinuation of noninvasive support was considered in infants who were receiving a fraction of inspired oxygen of 0.3 or lower, with gas flow of 4 liters per minute (in the high-flow group) or pressure of 5 cm of water (in the CPAP group); earlier cessation of support could be ordered at the discretion of the treating clinician. If further support was required after discontinuation of respiratory support, the randomly assigned treatment was reinitiated, except that infants in the high-flow group with previous treatment failure could receive CPAP at the treating clinician s discretion. Study Outcomes The primary outcome was treatment failure within 72 hours after randomization. Treatment was considered to have failed if an infant receiving maximal support (high-flow therapy at a gas flow of 8 liters per minute or CPAP at a pressure of 8 cm of water) met one or more of the following criteria: a fraction of inspired oxygen of 0.4 or higher, a ph of 7.2 or less plus a partial pressure of carbon dioxide greater than 60 mm Hg (8.0 kpa) in a sample of arterial or free-flowing capillary blood obtained at least 1 hour after commencement of the assigned treatment, or either two or more episodes of apnea requiring positive-pressure ventilation within a 24-hour period or six or more episodes requiring any intervention within a 6-hour period. Infants with an urgent need for intubation and mechanical ventilation (as determined by the treating clinician) were also considered to have treatment failure. Prespecified secondary outcomes included the reason (or reasons) for treatment failure, the use of mechanical ventilation within 72 hours after randomization or at any time during admission, nasal trauma, other complications, including complications of prematurity, and other measures of the use of respiratory support and of neonatal health. An additional secondary outcome was the cost of care, calculated on the basis of data for infants at all participating Australian units (435 infants) (Section 4.1 in the Supplementary Appendix). 18 The complete list of prespecified secondary outcomes is provided in the study protocol and in Section 2.3 in the Supplementary Appendix. Serious adverse events were defined as death before hospital discharge and pneumothorax or other air leak during the assigned treatment. Data were collected until death or discharge home. Statistical Analysis On the basis of data from the participating Australian centers, we estimated that treatment failure within 72 hours after randomization would 1144 n engl j med 375;12 nejm.org September 22, 2016
4 Nasal High-Flow Therapy in Preterm Infants occur in 17% of infants assigned to receive CPAP. We prespecified a noninferiority margin for high-flow treatment of 10 percentage points above the failure rate for CPAP treatment. Highflow therapy would be considered noninferior to CPAP if the difference in the risk of treatment failure and the upper limit of the two-sided 95% confidence interval were less than 10% and the lower limit of the 95% confidence interval was below zero. For the study to have 90% power, a sample of 750 infants was required. 17,19 We chose this margin of noninferiority after considering the following factors: high-flow therapy is already widely accepted in many neonatal intensive care units; infants in whom high-flow treatment failed could receive CPAP treatment, which we hypothesized would obviate the need for intubation and ventilation in some infants; and the primary study outcome was short-term efficacy, rather than death or disability (a lower margin of noninferiority would be required for death or disability). The neonatologists and parent representatives who were consulted during the design phase of the trial agreed to this noninferiority margin. The primary and secondary outcomes were analyzed on an intention-to-treat basis. A prespecified subgroup analysis on the basis of gestational age (<32 weeks or 32 weeks) and a per-protocol analysis (not prespecified but recommended for noninferiority trials 19 ) were performed for both the primary outcome and the intubation rate within 72 hours after randomization. For the primary outcome and dichotomous secondary outcomes, we calculated a risk difference (with a two-sided 95% confidence interval) in percentage points between treatment groups. We used chi-square tests to compare dichotomous outcomes and the appropriate parametric test (Student s t-test) or nonparametric test (difference in medians estimated by quantile regression) to compare continuous outcomes. All analyses were performed with the use of Stata/IC software, version 13.1 (StataCorp). As specified in the trial protocol, 17 an independent data and safety monitoring committee, consisting of two neonatologists and a statistician, reviewed outcome data when the primary outcome was available for 250 infants and when it was available for 500 infants. The committee could recommend stopping the trial if there were safety concerns or if there was a highly significant difference (P<0.001) in the rate of the primary outcome between treatment groups. Results Duration and Cessation of Recruitment Infants were recruited from May 27, 2013, to June 16, On June 12, 2015, after reviewing the primary outcome data for the first 515 recruited infants, the independent data and safety monitoring committee recommended that the trial be stopped, since there was a highly significant difference (P<0.001) in the rate of the primary outcome between treatment groups and continued recruitment was extremely unlikely to show the noninferiority of high-flow therapy to CPAP. The steering committee stopped recruitment on June 16, Study Patients In total, 583 infants were randomly assigned to a treatment group (289 to the high-flow group and 294 to the CPAP group) (Fig. 1). Nineteen infants were excluded because they did not meet the eligibility criteria or their parents did not provide consent. The remaining 564 infants (278 in the high-flow group and 286 in the CPAP group) were followed until hospital discharge or death and were included in the analysis. Demographic and clinical characteristics of the mothers and infants were similar in the two groups (Table 1). Primary Outcome Treatment failure within 72 hours after randomization occurred in 71 of the 278 infants (25.5%) in the high-flow group and in 38 of the 286 infants (13.3%) in the CPAP group (risk difference, 12.3 percentage points; 95% confidence interval, 5.8 to 18.7; P<0.001). Treatment failure was significantly more common in the high-flow group than in the CPAP group both among infants with a gestational age of less than 32 weeks and among those with a gestational age of 32 weeks or greater at randomization (Table 2). Intubation during the First 72 Hours after Randomization There was no significant between-group difference in intubation rates within 72 hours after randomization, either in the overall study population or in the gestational-age subgroups (Table 2). n engl j med 375;12 nejm.org September 22,
5 The new england journal of medicine 3349 Infants admitted to NICU were screened for eligibility 2256 Were ineligible 1636 Did not receive noninvasive respiratory support 393 Were intubated, received surfactant, or both 92 Were 24 hr of age 51 Had major congenital anomaly or air leak 46 Met treatment-failure criteria with CPAP of 8 cm water 36 Had received 4 hr of CPAP treatment 2 Received palliative care in the delivery room 1093 Were eligible 510 Did not undergo randomization 395 Had parents who were not approached 264 Had parents who were unavailable or a research team that was not notified 81 Had language or social factors 18 Were in another study, prohibiting participation 17 Were transferred to another hospital 13 Had a clinician who made the decision 2 Were in a NICU that did not have high-flow equipment available 113 Had parents who declined prospective consent 2 Had parents who consented but clinicians who decided against randomization 583 Underwent randomization 289 Were assigned to high-flow treatment 201 Had parents who provided prospective consent 88 Had parents who were approached for retrospective consent 294 Were assigned to CPAP treatment 206 Had parents who provided prospective consent 88 Had parents who were approached for retrospective consent 11 Were excluded 5 Had parents who declined retrospective consent 2 Had parents who withdrew prospective consent 4 Were ineligible and underwent randomization in error 8 Were excluded 5 Had parents who declined retrospective consent 2 Were ineligible and underwent randomization in error 1 Underwent randomization without prospective parental consent 278 Infants were followed until death or discharge and included in the primary intention-to-treat analysis 286 Infants were followed until death or discharge and included in the primary intention-to-treat analysis Figure 1. Numbers of Infants Who Were Screened, Assigned to a Study Group, and Included in the Primary Analysis. Infants born at a gestational age of 28 weeks 0 days to 36 weeks 6 days were screened for eligibility. CPAP denotes continuous positive airway pressure, and NICU neonatal intensive care unit n engl j med 375;12 nejm.org September 22, 2016
6 Nasal High-Flow Therapy in Preterm Infants Table 1. Baseline Characteristics of the Mothers and Infants.* Characteristic High-Flow Group (N = 278) CPAP Group (N = 286) Mothers White race no. (%) 217 (78.1) 225 (78.7) Primigravida no. (%) 117 (42.1) 119 (41.6) Exposure to antenatal glucocorticoids no. (%) 224 (80.9) 229 (80.4) Cesarean section no. (%) 204 (73.4) 200 (69.9) Prolonged rupture of membranes 24 hr before delivery no. (%) 33 (11.9) 37 (12.9) Obstetrical diagnosis of chorioamnionitis no. (%) 22 (8.0) 13 (4.6) Infants Gestational age No. of weeks 32.0± ±2.2 <32 wk no. (%) 140 (50.4) 149 (52.1) Birth weight g 1737± ±599 Male sex no. (%) 157 (56.5) 156 (54.5) Multiple birth no. (%) 109 (39.2) 109 (38.1) Median Apgar score at 5 min (IQR) 8 (8 9) 9 (8 9) Median postnatal age at randomization (IQR) hr 1.4 ( ) 1.3 ( ) Treatment with CPAP before randomization No. of infants (%) 157 (56.5) 166 (58.0) Median duration (IQR) hr 1.6 ( ) 1.5 ( ) Arterial or capillary blood ph before randomization 7.26± ±0.07 Partial pressure of arterial or capillary carbon dioxide before 55.2± ±9.8 randomization mm Hg Median fraction of inspired oxygen at time of randomization (IQR) 0.21 ( ) 0.21 ( ) Caffeine received in first 24 hr of life no. (%) 109 (39.2) 125 (43.7) * Plus minus values are means ±SD. There were no significant differences between the groups. CPAP denotes continuous positive airway pressure, and IQR interquartile range. Race was reported by the investigators. Data on exposure to antenatal glucocorticoids were missing for 1 infant in each treatment group. Obstetrical diagnosis of chorioamnionitis was not known for 2 infants in the high-flow group and 3 in the CPAP group. The Apgar score was not known for 2 infants in the high-flow group. Blood gases were not measured before randomization in 297 infants: 145 in the high-flow group and 152 in the CPAP group. Per-Protocol Analysis Results were similar in an analysis of the 543 infants (264 in the high-flow group and 279 in the CPAP group) who were treated as specified by the protocol (Table 2). A total of 21 infants were excluded from this analysis because either they did not receive the assigned treatment (14 infants) or the assigned treatment was changed during the primary-outcome period without the protocol-specified criteria for treatment failure or nasal trauma having been met (7 infants). Secondary Outcomes and Adverse Events The most common reason for treatment failure was a fraction of inspired oxygen of 0.4 or higher (Table 3). Treatment failure due to an urgent need for intubation occurred more frequently in the CPAP group than in the high-flow group (18.4% vs. 5.6%, P = 0.03). The median duration of respiratory support was 1 day longer in the highflow group than in the CPAP group (4 vs. 3 days, P = 0.005), and infants in the high-flow group were significantly more likely to receive supple- n engl j med 375;12 nejm.org September 22,
7 The new england journal of medicine Table 2. Primary Outcome, Intubation within 72 Hours, and Outcomes in the Subgroup and Per-Protocol Analyses. High-Flow Group (N = 278) CPAP Group (N = 286) Risk Difference (95% CI)* P Value percentage points Outcome no./total no. (%) Primary intention-to-treat analysis Treatment failure within 72 hr 71/278 (25.5) 38/286 (13.3) 12.3 (5.8 to 18.7) <0.001 Gestational age <32 wk 46/140 (32.9) 27/149 (18.1) 14.7 (4.8 to 24.7) Gestational age 32 wk 25/138 (18.1) 11/137 (8.0) 10.1 (2.2 to 18.0) 0.01 Intubation within 72 hr 43/278 (15.5) 33/286 (11.5) 3.9 ( 1.7 to 9.6) 0.17 Gestational age <32 wk 30/140 (21.4) 24/149 (16.1) 5.3 ( 3.7 to 14.3) 0.25 Gestational age 32 wk 13/138 (9.4) 9/137 (6.6) 2.9 ( 3.5 to 9.3) 0.38 Per-protocol analysis Treatment failure within 72 hr 64/264 (24.2) 36/279 (12.9) 11.3 (4.8 to 17.8) <0.001 Intubation within 72 hr 39/264 (14.8) 33/279 (11.8) 2.9 ( 2.8 to 8.7) 0.31 * Positive values favor the CPAP group, and negative values favor the high-flow group. Apparent discrepancies in some of the risk differences are due to rounding. mental oxygen during their admission (78.1% vs. 69.6%, P = 0.02). Respiratory diagnoses in the participating infants are reported in Section 3.3 in the Supplementary Appendix. There was no significant difference between treatment groups in the rate of death before discharge (Table 4). Nasal trauma was significantly more common in the CPAP group than in the high-flow group (18.5% vs. 8.3%, P<0.001). The frequency of pneumothorax or other air leak from the lung was also significantly higher in the CPAP group during the assigned treatment (2.1% vs. 0.0%, P = 0.02) but not overall (3.6% in the high-flow group and 2.8% in the CPAP group, P = 0.59). Rates of other complications of prematurity did not differ significantly between the groups. The respiratory-support devices and nasal interfaces that were used initially in each treatment group are shown in Section 3.2 in the Supplementary Appendix. The calculated total cost of the tertiary hospital stay (in U.S. dollars) per infant did not differ significantly between the CPAP and high-flow groups ($32,036 and $29,785, respectively; P = 0.40). (Detailed information about the cost-effectiveness analysis is provided in Sections 4.2 and 4.3 in the Supplementary Appendix.) Discussion In this multicenter, randomized trial, high-flow treatment resulted in a significantly higher rate of treatment failure than did CPAP when used as primary respiratory support for preterm infants born at 28 weeks 0 days of gestation or later and treated in neonatal intensive care units. Enrollment was stopped after a planned interim analysis (after 75% of the target sample had been recruited), on the recommendation of the data and safety monitoring committee, owing to the between-group difference in rates of treatment failure. The difference in the primary outcome was significant in both the primary intention-totreat analysis and the per-protocol analysis. Our results contrast with those of studies of high-flow therapy initiated after extubation, which have consistently shown that the efficacy of high-flow treatment is similar to that of CPAP. 7-9,16 Unlike the infants in the trials of postextubation high-flow therapy, no infants in our study received surfactant before randomization. 7-9 The higher rate of treatment failure among infants receiving high-flow therapy in our study may reflect its reduced effectiveness in infants with surfactant-deficient lungs. Although high-flow therapy does provide some distending pressure, the higher, more consistent pressures produced during CPAP may account for the difference in treatment-failure rates that we report. We chose to include only infants with a gestational age of at least 28 weeks, on the basis of increased rates of treatment failure reported in infants with a lower gestational age who received high-flow therapy after extubation n engl j med 375;12 nejm.org September 22, 2016
8 Nasal High-Flow Therapy in Preterm Infants Table 3. Reasons for Treatment Failure and Other Secondary Outcomes.* Outcome High-Flow Group (N = 278) CPAP Group (N = 286) Percentage-Point Difference in Risk (95% CI) Difference in Median (95% CI) P Value Reason for treatment failure no./total no. (%) Apnea 9/71 (12.7) 2/38 (5.3) 7.4 ( 3.1 to 17.9) NA 0.22 Fraction of inspired oxygen /71 (74.6) 30/38 (78.9) 4.3 ( 20.7 to 12.1) NA 0.62 Respiratory acidosis 9/71 (12.7) 6/38 (15.8) 3.1 ( 17.1 to 10.8) NA 0.65 Urgent need for intubation 4/71 (5.6) 7/38 (18.4) 12.8 ( 26.2 to 0.7) NA 0.03 Clinician s decision 4/71 (5.6) 2/38 (5.3) 0.4 ( 8.5 to 9.3) NA 0.94 Median time to treatment failure after randomization (IQR) hr 7.8 (1.9 to 21.5) 9.0 (2.8 to 25.2) NA 1.2 ( 10.1 to 6.3) 0.65 Surfactant treatment no. (%) 40 (14.4) 30 (10.5) 3.9 ( 1.5 to 9.3) NA 0.16 Median no. of days of respiratory support during admission (IQR) 4 (2 to 7) 3 (2 to 6) NA 1.0 (0.3 to 1.7) Supplemental oxygen therapy Any received during admission no. (%) 217 (78.1) 199 (69.6) 8.5 (1.3 to 15.7) NA 0.02 Median age at cessation (IQR) days 2 (0 to 5) 2 (0 to 6) NA 0.0 ( 0.7 to 0.7) 1.00 Discharged home with oxygen therapy no. (%) 1 (0.4) 2 (0.7) 0.3 ( 1.5 to 0.9) NA 0.58 Median age at start of full-suck feeding (IQR) days 32 (18 to 43) 32 (17 to 45) NA 0.0 ( 3.9 to 3.9) 1.00 Discharged home with gastric-tube feeding no. (%) 21 (7.6) 17 (6.0) 1.6 ( 2.5 to 5.8) NA 0.45 Weight at discharge g 2540± ±492 NA NA 0.77 Median no. of days in tertiary care center (IQR) 20 (10 to 35) 19 (10 to 34) NA 1.0 ( 2.3 to 4.3) 0.56 Median no. of days in any hospital (IQR) 37 (23 to 50) 38 (22 to 50) NA 1.0 ( 5.4 to 3.4) 0.66 * NA denotes not applicable. Positive values favor the CPAP group, and negative values favor the high-flow group. Treatment may have failed for more than one reason. Clinician s decisions to change treatment were made in relation to the work of breathing, without fulfilling any prespecified treatment-failure criteria. The 3 infants who were discharged home with oxygen therapy (1 in the high-flow group and 2 in the CPAP group) were excluded. The 2 infants who died before discharge (1 in each treatment group) were excluded. A total of 40 infants were excluded: the 38 infants who were discharged home with gastric-tube feeding (21 in the high-flow group and 17 in the CPAP group) and the 2 infants who died before discharge (1 in each treatment group). n engl j med 375;12 nejm.org September 22,
9 The new england journal of medicine Table 4. Adverse Events. Event High-Flow Group (N = 278) CPAP Group (N = 286) Risk Difference (95% CI)* P Value no. of infants (%) percentage points Death before discharge 1 (0.4) 1 (0.4) 0.0 ( 1.0 to 1.0) 0.98 Oxygen supplementation, respiratory support, or both 17 (12.1) 17 (11.4) 0.7 ( 6.7 to 8.2) 0.85 at postmenstrual age of 36 wk Pneumothorax or other air leak syndrome During assigned treatment 0 6 (2.1) 2.1 ( 3.8 to 0.4) 0.02 Any time during admission 10 (3.6) 8 (2.8) 0.8 ( 2.1 to 3.7) 0.59 Postnatal glucocorticoid treatment for lung disease 1 (0.4) 3 (1.0) 0.7 ( 2.1 to 0.7) 0.33 Nasal trauma 23 (8.3) 53 (18.5) 10.3 ( 15.8 to 4.7) <0.001 Patent ductus arteriosus treated with medication or 11 (4.0) 6 (2.1) 1.9 ( 1.0 to 4.7) 0.20 surgical ligation Confirmed sepsis 7 (2.5) 13 (4.5) 2.0 ( 5.1 to 1.0) 0.19 Necrotizing enterocolitis, Bell s stage II or III 2 (0.7) ( 0.3 to 1.7) 0.15 Isolated intestinal perforation 0 1 (0.3) 0.3 ( 1.0 to 0.3) 0.32 Laser surgery for retinopathy of prematurity 0 1 (0.7) 0.7 ( 2.0 to 0.6) 0.33 Intraventricular hemorrhage, grade 3 or 4 4 (2.9) 1 (0.7) 2.2 ( 0.9 to 5.2) 0.15 Cystic periventricular leukomalacia 3 (2.1) 2 (1.3) 0.8 ( 2.2 to 3.8) 0.60 * Positive values favor the CPAP group, and negative values favor the high-flow group. Data are reported for the 289 infants born at a gestational age of less than 32 weeks (140 infants in the high-flow group and 149 in the CPAP group). The criteria for confirmation of sepsis were a positive blood culture and treatment with intravenous antibiotics for 48 hours or longer. Modified Bell s criteria stages range from I to III, with higher stages indicating greater disease severity. In our study, intubation rates did not differ significantly between the groups, probably because the use of CPAP as rescue therapy for infants with treatment failure in the high-flow group meant that subsequent intubation was not required in 39% of those infants (28 of 71). We included rescue CPAP in our trial design because in our previous noninferiority study of high-flow treatment after extubation, 8 almost half of the infants in whom high-flow treatment failed did not require intubation after receiving rescue CPAP. Although CPAP was associated with a lower rate of treatment failure than was high-flow therapy, intubation rates did not differ significantly between the two treatment groups; in addition, infants in the high-flow group had a significantly lower rate of nasal trauma. However, infants in the high-flow group were more likely to receive brief supplemental oxygen, and the median duration of respiratory support was 1 day longer in this group. The clinical importance of these findings is uncertain. Blinding of the intervention was not possible; therefore, to minimize bias, we used prespecified, objective criteria to determine the primary outcome. We acknowledge that the use of CPAP as rescue therapy may have influenced the rates of secondary outcomes in the high-flow group. Furthermore, over half of the infants assigned to this group had received CPAP for a brief period (median, 1.6 hours) before randomization, which may also have influenced the outcomes. Our study population was limited to preterm infants in neonatal intensive care units. Further studies are required to determine the safety and efficacy of high-flow therapy in nontertiary facilities and resource-limited settings, as well as in term infants. We conclude that high-flow treatment results in a significantly higher rate of treatment failure than does CPAP, when used as primary support for preterm infants with respiratory distress. Supported by grants from the National Health and Medical Research Council ( and Centre of Research Excellence New n engl j med 375;12 nejm.org September 22, 2016
10 Nasal High-Flow Therapy in Preterm Infants born Medicine Grant ), the Royal Brisbane and Women s Hospital Foundation, and the participating neonatal units. Dr. Davis reports receiving travel support from Fisher and Paykel. No other potential conflict of interest relevant to this article was reported. Disclosure forms provided by the authors are available with the full text of this article at NEJM.org. We thank the families of all infants who participated in the study and the staff members who cared for them. References 1. Hamilton BE, Martin JA, Osterman MJK, Curtin SC. Births: preliminary data for Natl Vital Stat Rep 2015; 64(6): Chess PR, D Angio CT, Pryhuber GS, Maniscalco WM. Pathogenesis of bronchopulmonary dysplasia. Semin Perinatol 2006; 30: SUPPORT Study Group of the Eunice Kennedy Shriver NICHD Neonatal Research Network. Early CPAP versus surfactant in extremely preterm infants. N Engl J Med 2010; 362: Morley CJ, Davis PG, Doyle LW, Brion LP, Hascoet J-M, Carlin JB. Nasal CPAP or intubation at birth for very preterm infants. N Engl J Med 2008; 358: Hochwald O, Osiovich H. The use of high flow nasal cannulae in neonatal intensive care units: is clinical practice consistent with the evidence? J Neonat Perinat Med 2010; 3: Hough JL, Shearman AD, Jardine LA, Davies MW. Humidified high flow nasal cannulae: current practice in Australasian nurseries, a survey. J Paediatr Child Health 2012; 48: Collins CL, Holberton JR, Barfield C, Davis PG. A randomized controlled trial to compare heated humidified high-flow nasal cannulae with nasal continuous positive airway pressure postextubation in premature infants. J Pediatr 2013; 162(5): e1. 8. Manley BJ, Owen LS, Doyle LW, et al. High-flow nasal cannulae in very preterm infants after extubation. N Engl J Med 2013; 369: Yoder BA, Stoddard RA, Li M, King J, Dirnberger DR, Abbasi S. Heated, humidified high-flow nasal cannula versus nasal CPAP for respiratory support in neonates. Pediatrics 2013; 131(5): e Osman M, Elsharkawy A, Abdel-Hady H. Assessment of pain during application of nasal-continuous positive airway pressure and heated, humidified high-flow nasal cannulae in preterm infants. J Perinatol 2015; 35: Klingenberg C, Pettersen M, Hansen EA, et al. Patient comfort during treatment with heated humidified high flow nasal cannulae versus nasal continuous positive airway pressure: a randomised cross-over trial. Arch Dis Child Fetal Neonatal Ed 2014; 99: F Roberts CT, Manley BJ, Dawson JA, Davis PG. Nursing perceptions of highflow nasal cannulae treatment for very preterm infants. J Paediatr Child Health 2014; 50: Nair G, Karna P. Comparison of the effects of Vapotherm and nasal CPAP in respiratory distress. Presented at the Pediatric Academic Societies Meeting, Baltimore, May 14 17, abstract ( pas/ ). 14. Iranpour R, Sadeghnia A, Hesaraki M. High-flow nasal cannula versus nasal continuous positive airway pressure in the management of respiratory distress syndrome. J Isfahan Med School 2011; 29: Ciuffini F, Pietrasanta C, Lavizzari A, et al. Comparison between two different modes of non-invasive ventilatory support in preterm newborn infants with respiratory distress syndrome mild to moderate: preliminary data. Pediatr Med Chir 2014; 36: Wilkinson D, Andersen C, O Donnell CP, De Paoli AG, Manley BJ. High flow nasal cannula for respiratory support in preterm infants. Cochrane Database Syst Rev 2016; 2: CD Roberts CT, Owen LS, Manley BJ, Donath SM, Davis PG. A multicentre, randomised controlled, non-inferiority trial, comparing high flow therapy with nasal continuous positive airway pressure as primary support for preterm infants with respiratory distress (the HIPSTER trial): study protocol. BMJ Open 2015; 5(6): e Drummond MF, Sculpher MJ, Torrance GW, O Brien BJ, Stoddart GL. Methods for the economic evaluation of health care programmes. 3rd ed. Oxford, England: Oxford University Press, Piaggio G, Elbourne DR, Pocock SJ, Evans SJ, Altman DG. Reporting of noninferiority and equivalence randomized trials: extension of the CONSORT 2010 statement. JAMA 2012; 308: Kubicka ZJ, Limauro J, Darnall RA. Heated, humidified high-flow nasal cannula therapy: yet another way to deliver continuous positive airway pressure? Pediatrics 2008; 121: Spence KL, Murphy D, Kilian C, Mc- Gonigle R, Kilani RA. High-flow nasal cannula as a device to provide continuous positive airway pressure in infants. J Perinatol 2007; 27: Wilkinson DJ, Andersen CC, Smith K, Holberton J. Pharyngeal pressure with high-flow nasal cannulae in premature infants. J Perinatol 2008; 28: Copyright 2016 Massachusetts Medical Society. ARTICLE METRICS NOW AVAILABLE Visit the article page at NEJM.org and click on the Metrics tab to view comprehensive and cumulative article metrics compiled from multiple sources, including Altmetrics. Learn more at n engl j med 375;12 nejm.org September 22,
High-Flow Nasal Cannulae in Very Preterm Infants after Extubation
 original article High-Flow Nasal Cannulae in Very Preterm Infants after Extubation Brett J. Manley, M.B., B.S., Louise S. Owen, M.D., Lex W. Doyle, M.D., Chad C. Andersen, M.B., B.S., David W. Cartwright,
original article High-Flow Nasal Cannulae in Very Preterm Infants after Extubation Brett J. Manley, M.B., B.S., Louise S. Owen, M.D., Lex W. Doyle, M.D., Chad C. Andersen, M.B., B.S., David W. Cartwright,
Name and title of the investigators responsible for conducting the research: Dr Anna Lavizzari, Dr Mariarosa Colnaghi
 Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Randomised controlled trial of weaning strategies for preterm infants on nasal continuous positive airway pressure
 Tang et al. BMC Pediatrics (2015) 15:147 DOI 10.1186/s12887-015-0462-0 RESEARCH ARTICLE Open Access Randomised controlled trial of weaning strategies for preterm infants on nasal continuous positive airway
Tang et al. BMC Pediatrics (2015) 15:147 DOI 10.1186/s12887-015-0462-0 RESEARCH ARTICLE Open Access Randomised controlled trial of weaning strategies for preterm infants on nasal continuous positive airway
CPAP failure in preterm infants: incidence, predictors and consequences
 CPAP failure in preterm infants: incidence, predictors and consequences SUPPLEMENTAL TEXT METHODS Study setting The Royal Hobart Hospital has an 11-bed combined Neonatal and Paediatric Intensive Care Unit
CPAP failure in preterm infants: incidence, predictors and consequences SUPPLEMENTAL TEXT METHODS Study setting The Royal Hobart Hospital has an 11-bed combined Neonatal and Paediatric Intensive Care Unit
Nasal CPAP in Neonatology: We Can Do Better
 Nasal CPAP in Neonatology: We Can Do Better COI Disclosure I do not have any conflict of interest, nor will I be discussing any off-label product use. This class has no commercial support or sponsorship,
Nasal CPAP in Neonatology: We Can Do Better COI Disclosure I do not have any conflict of interest, nor will I be discussing any off-label product use. This class has no commercial support or sponsorship,
Kugelman A, Riskin A, Said W, Shoris I, Mor F, Bader D.
 Heated, Humidified High-Flow Nasal Cannula (HHHFNC) vs. Nasal Intermittent Positive Pressure Ventilation (NIPPV) for the Primary Treatment of RDS, A Randomized, Controlled, Prospective, Pilot Study Kugelman
Heated, Humidified High-Flow Nasal Cannula (HHHFNC) vs. Nasal Intermittent Positive Pressure Ventilation (NIPPV) for the Primary Treatment of RDS, A Randomized, Controlled, Prospective, Pilot Study Kugelman
Surfactant Administration
 Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018
 Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
This is a pre-copyedited, author-produced PDF of an article accepted for publication in Journal of Neonatal Nursing following peer review.
 This is a pre-copyedited, author-produced PDF of an article accepted for publication in Journal of Neonatal Nursing following peer review. The version of record [Journal of Neonatal Nursing (February 2013)
This is a pre-copyedited, author-produced PDF of an article accepted for publication in Journal of Neonatal Nursing following peer review. The version of record [Journal of Neonatal Nursing (February 2013)
Non-invasive ventilatory strategies, such as
 R E S E A R C H P A P E R Heated Humidified High Flow Nasal Cannula versus Nasal Continuous Positive Airway Pressure as Primary Mode of Respiratory Support for Respiratory Distress in Preterm Infants DEEPARAJ
R E S E A R C H P A P E R Heated Humidified High Flow Nasal Cannula versus Nasal Continuous Positive Airway Pressure as Primary Mode of Respiratory Support for Respiratory Distress in Preterm Infants DEEPARAJ
Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience
 Original Article Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience Pedro Paz, MD, MPH 1 Rangasamy Ramanathan, MD 1,2 Richard Hernandez, RCP 2 Manoj Biniwale, MD 1 1 Division of Neonatal
Original Article Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience Pedro Paz, MD, MPH 1 Rangasamy Ramanathan, MD 1,2 Richard Hernandez, RCP 2 Manoj Biniwale, MD 1 1 Division of Neonatal
Clinical Guideline: Heated Humidified High Flow Nasal Cannula (HHHFNC) Guideline
 EOE Neonatal ODN Clinical Guideline: Heated Humidified High Flow Nasal Cannula (HHHFNC) Guideline Authors: Dr Eliana Panayiotou and Dr Bharat Vakharia For use in: EoE Neonatal Units Guidance specific to
EOE Neonatal ODN Clinical Guideline: Heated Humidified High Flow Nasal Cannula (HHHFNC) Guideline Authors: Dr Eliana Panayiotou and Dr Bharat Vakharia For use in: EoE Neonatal Units Guidance specific to
AEROSURF Phase 2 Program Update Investor Conference Call
 AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION
 CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
Bubble CPAP for Respiratory Distress Syndrome in Preterm Infants
 R E S E A R C H P A P E R Bubble CPAP for Respiratory Distress Syndrome in Preterm Infants JAGDISH KOTI*, SRINIVAS MURKI, PRAMOD GADDAM, ANUPAMA REDDY AND M DASARADHA RAMI REDDY From Fernandez Hospital
R E S E A R C H P A P E R Bubble CPAP for Respiratory Distress Syndrome in Preterm Infants JAGDISH KOTI*, SRINIVAS MURKI, PRAMOD GADDAM, ANUPAMA REDDY AND M DASARADHA RAMI REDDY From Fernandez Hospital
Admission/Discharge Form for Infants Born in Please DO NOT mail or fax this form to the CPQCC Data Center. This form is for internal use ONLY.
 Selection Criteria Admission/Discharge Form for Infants Born in 2016 To be eligible, you MUST answer YES to at least one of the possible criteria (A-C) A. 401 1500 grams o Yes B. GA range 22 0/7 31 6/7
Selection Criteria Admission/Discharge Form for Infants Born in 2016 To be eligible, you MUST answer YES to at least one of the possible criteria (A-C) A. 401 1500 grams o Yes B. GA range 22 0/7 31 6/7
5/3/2012. Goals and Objectives HFNC. High-Flow Oxygen Therapy: Real Benefit or Just a Fad?
 High-Flow Oxygen Therapy: Real Benefit or Just a Fad? Timothy R. Myers MBA, RRT-NPS Director, Women s & Children s Respiratory Care & Procedural Services and Pediatric Heart Center Rainbow Babies & Children
High-Flow Oxygen Therapy: Real Benefit or Just a Fad? Timothy R. Myers MBA, RRT-NPS Director, Women s & Children s Respiratory Care & Procedural Services and Pediatric Heart Center Rainbow Babies & Children
COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY
 CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
Ge Zheng, 1,2 Xiao-qiu Huang, 2 Hui-hui Zhao, 2 Guo-Xing Jin, 2 and Bin Wang Introduction. 2. Methods
 Hindawi Canadian Respiratory Journal Volume 2017, Article ID 3782401, 6 pages https://doi.org/10.1155/2017/3782401 Clinical Study The Effect of the Treatment with Heated Humidified High-Flow Nasal Cannula
Hindawi Canadian Respiratory Journal Volume 2017, Article ID 3782401, 6 pages https://doi.org/10.1155/2017/3782401 Clinical Study The Effect of the Treatment with Heated Humidified High-Flow Nasal Cannula
Advantages and disadvantages of different nasal CPAP systems in newborns
 Intensive Care Med (2004) 30:926 930 DOI 10.1007/s00134-004-2267-8 N E O N A T A L A N D P A E D I A T R I C I N T E N S I V E C A R E V. Buettiker M. I. Hug O. Baenziger C. Meyer B. Frey Advantages and
Intensive Care Med (2004) 30:926 930 DOI 10.1007/s00134-004-2267-8 N E O N A T A L A N D P A E D I A T R I C I N T E N S I V E C A R E V. Buettiker M. I. Hug O. Baenziger C. Meyer B. Frey Advantages and
Research Article Timing of Caffeine Therapy and Neonatal Outcomes in Preterm Infants: A Retrospective Study
 International Pediatrics Volume 2016, Article ID 9478204, 6 pages http://dx.doi.org/10.1155/2016/9478204 Research Article Timing of Caffeine Therapy and Neonatal Outcomes in Preterm Infants: A Retrospective
International Pediatrics Volume 2016, Article ID 9478204, 6 pages http://dx.doi.org/10.1155/2016/9478204 Research Article Timing of Caffeine Therapy and Neonatal Outcomes in Preterm Infants: A Retrospective
ADMISSION/DISCHARGE FORM FOR INFANTS BORN IN 2019 DO NOT mail or fax this form to the CPQCC Data Center. This form is for internal use ONLY.
 1 Any eligible inborn infant who dies in the delivery room or at any other location in your hospital within 12 hours after birth and prior to admission to the NICU is defined as a "Delivery Room Death."
1 Any eligible inborn infant who dies in the delivery room or at any other location in your hospital within 12 hours after birth and prior to admission to the NICU is defined as a "Delivery Room Death."
ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME
 INDIAN PEDIATRICS VOLUME 35-FEBRUAKY 1998 ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME Kanya Mukhopadhyay, Praveen Kumar and Anil Narang From the Division of Neonatology, Department
INDIAN PEDIATRICS VOLUME 35-FEBRUAKY 1998 ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME Kanya Mukhopadhyay, Praveen Kumar and Anil Narang From the Division of Neonatology, Department
ROLE OF PRESSURE IN HIGH FLOW THERAPY
 ROLE OF PRESSURE IN HIGH FLOW THERAPY Thomas L. Miller, PhD, MEd Director, Clinical Research and Education Vapotherm, Inc. Research Assistant Professor of Pediatrics Jefferson Medical College This information
ROLE OF PRESSURE IN HIGH FLOW THERAPY Thomas L. Miller, PhD, MEd Director, Clinical Research and Education Vapotherm, Inc. Research Assistant Professor of Pediatrics Jefferson Medical College This information
Hyaline membrane disease. By : Dr. Ch Sarishma Peadiatric Pg
 Hyaline membrane disease By : Dr. Ch Sarishma Peadiatric Pg Also called Respiratory distress syndrome. It occurs primarily in premature infants; its incidence is inversely related to gestational age and
Hyaline membrane disease By : Dr. Ch Sarishma Peadiatric Pg Also called Respiratory distress syndrome. It occurs primarily in premature infants; its incidence is inversely related to gestational age and
Newborn Life Support. NLS guidance.
 Kelly Harvey, ANNP NWNODN, previously Wythenshawe Hospital has shared this presentation with the understanding that it is for personal use following your attendance at the 8th Annual Senior Neonatal Nursing
Kelly Harvey, ANNP NWNODN, previously Wythenshawe Hospital has shared this presentation with the understanding that it is for personal use following your attendance at the 8th Annual Senior Neonatal Nursing
GS3. Understanding How to Use Statistics to Evaluate an Article. Session Summary. Session Objectives. References. Session Outline
 GS3 Understanding How to Use Statistics to Evaluate an Article Reese H. Clark, MD Director of Research Pediatrix Medical Group Neonatologist Greenville Memorial Hospital, Greenville, SC The speaker has
GS3 Understanding How to Use Statistics to Evaluate an Article Reese H. Clark, MD Director of Research Pediatrix Medical Group Neonatologist Greenville Memorial Hospital, Greenville, SC The speaker has
High-flow nasal cannula use in a paediatric intensive care unit over 3 years
 High-flow nasal cannula use in a paediatric intensive care unit over 3 years Tracey I Wraight and Subodh S Ganu Respiratory illness and/or distress is the commonest reason for non-elective paediatric intensive
High-flow nasal cannula use in a paediatric intensive care unit over 3 years Tracey I Wraight and Subodh S Ganu Respiratory illness and/or distress is the commonest reason for non-elective paediatric intensive
With wider acceptance of the early use of
 Weaning of nasal CPAP in preterm infants: who, when and how? a systematic review of literature Shaili Amatya, Deepa Rastogi, Alok Bhutada, Shantanu Rastogi New York, USA Background: There is increased
Weaning of nasal CPAP in preterm infants: who, when and how? a systematic review of literature Shaili Amatya, Deepa Rastogi, Alok Bhutada, Shantanu Rastogi New York, USA Background: There is increased
Facilitating EndotracheaL Intubation by Laryngoscopy technique and Apneic Oxygenation Within the Intensive Care Unit (FELLOW)
 Facilitating EndotracheaL Intubation by Laryngoscopy technique and Apneic Oxygenation Within the Intensive Data Analysis Plan: Apneic Oxygenation vs. No Apneic Oxygenation Background Critically ill patients
Facilitating EndotracheaL Intubation by Laryngoscopy technique and Apneic Oxygenation Within the Intensive Data Analysis Plan: Apneic Oxygenation vs. No Apneic Oxygenation Background Critically ill patients
Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome
 European Review for Medical and Pharmacological Sciences 2015; 19: 573-577 Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome B.
European Review for Medical and Pharmacological Sciences 2015; 19: 573-577 Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome B.
Prophylactic Aminophylline for Prevention of Apnea at Higher-Risk Preterm Neonates
 Iran Red Crescent Med J. 2014 August; 16(8): e12559. Published online 2014 August 5. DOI: 10.5812/ircmj.12559 Research Article Prophylactic Aminophylline for Prevention of Apnea at Higher-Risk Preterm
Iran Red Crescent Med J. 2014 August; 16(8): e12559. Published online 2014 August 5. DOI: 10.5812/ircmj.12559 Research Article Prophylactic Aminophylline for Prevention of Apnea at Higher-Risk Preterm
Nasal versus oral intubation for mechanical ventilation of newborn infants (Review)
 Nasal versus oral intubation for mechanical ventilation of newborn infants (Review) Spence K, Barr P This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration and published
Nasal versus oral intubation for mechanical ventilation of newborn infants (Review) Spence K, Barr P This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration and published
Provide guidelines for the management of mechanical ventilation in infants <34 weeks gestation.
 Page 1 of 5 PURPOSE: Provide guidelines for the management of mechanical ventilation in infants
Page 1 of 5 PURPOSE: Provide guidelines for the management of mechanical ventilation in infants
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/46692 holds various files of this Leiden University dissertation Author: Zanten, Henriëtte van Title: Oxygen titration and compliance with targeting oxygen
Cover Page The handle http://hdl.handle.net/1887/46692 holds various files of this Leiden University dissertation Author: Zanten, Henriëtte van Title: Oxygen titration and compliance with targeting oxygen
ORIGINAL ARTICLE. DD Woodhead, DK Lambert, JM Clark and RD Christensen. Intermountain Healthcare, McKay-Dee Hospital, Ogden, UT, USA
 ORIGINAL ARTICLE Comparing two methods of delivering high-flow gas therapy by nasal cannula following endotracheal extubation: a prospective, randomized, masked, crossover trial DD Woodhead, DK Lambert,
ORIGINAL ARTICLE Comparing two methods of delivering high-flow gas therapy by nasal cannula following endotracheal extubation: a prospective, randomized, masked, crossover trial DD Woodhead, DK Lambert,
Recommendations for Hospital Quality Measures in 2011:
 Pediatric Measures: Recommendations for Hospital Quality Measures in 2011: Based on the input of a group of healthcare stakeholders, the following new hospital measures are recommended: 1) Home Management
Pediatric Measures: Recommendations for Hospital Quality Measures in 2011: Based on the input of a group of healthcare stakeholders, the following new hospital measures are recommended: 1) Home Management
SARASOTA MEMORIAL HOSPITAL DEPARTMENT POLICY
 PS1006 SARASOTA MEMORIAL HOSPITAL DEPARTMENT POLICY TITLE: NON-INVASIVE VENTILATION FOR THE Job Title of Reviewer: EFFECTIVE DATE: REVISED DATE: Director, Respiratory Care Services 126.685 (neo) 3/26/15
PS1006 SARASOTA MEMORIAL HOSPITAL DEPARTMENT POLICY TITLE: NON-INVASIVE VENTILATION FOR THE Job Title of Reviewer: EFFECTIVE DATE: REVISED DATE: Director, Respiratory Care Services 126.685 (neo) 3/26/15
Short-Term Outcome Of Different Treatment Modalities Of Patent Ductus Arteriosus In Preterm Infants. Five Years Experiences In Qatar
 ISPUB.COM The Internet Journal of Cardiovascular Research Volume 7 Number 2 Short-Term Outcome Of Different Treatment Modalities Of Patent Ductus Arteriosus In Preterm Infants. Five Years Experiences In
ISPUB.COM The Internet Journal of Cardiovascular Research Volume 7 Number 2 Short-Term Outcome Of Different Treatment Modalities Of Patent Ductus Arteriosus In Preterm Infants. Five Years Experiences In
A Trial Comparing Noninvasive Ventilation Strategies in Preterm Infants
 T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article A Trial Comparing Noninvasive Ventilation Strategies in Preterm Infants Haresh Kirpalani, B.M., M.Sc., David Millar, M.B., Brigitte
T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article A Trial Comparing Noninvasive Ventilation Strategies in Preterm Infants Haresh Kirpalani, B.M., M.Sc., David Millar, M.B., Brigitte
Prophylactic nasal continuous positive airway pressure for preventing morbidity and mortality in very preterm infants(review)
 Cochrane Database of Systematic Reviews Prophylactic nasal continuous positive airway pressure for preventing morbidity and mortality in very preterm infants (Review) SubramaniamP,HoJJ,DavisPG Subramaniam
Cochrane Database of Systematic Reviews Prophylactic nasal continuous positive airway pressure for preventing morbidity and mortality in very preterm infants (Review) SubramaniamP,HoJJ,DavisPG Subramaniam
SURFACTANT UPDATE. George Mandy, M.D. Nationwide Children s Hospital The Ohio State University
 SURFACTANT UPDATE George Mandy, M.D. Nationwide Children s Hospital The Ohio State University Surfactant Update Objectives History Meta-analysis of surfactant therapy New synthetic surfactant Genetic disorders
SURFACTANT UPDATE George Mandy, M.D. Nationwide Children s Hospital The Ohio State University Surfactant Update Objectives History Meta-analysis of surfactant therapy New synthetic surfactant Genetic disorders
NON INVASIVE LIFE SAVERS. Non Invasive Ventilation (NIV)
 Table 1. NIV: Mechanisms Of Action Decreases work of breathing Increases functional residual capacity Recruits collapsed alveoli Improves respiratory gas exchange Reverses hypoventilation Maintains upper
Table 1. NIV: Mechanisms Of Action Decreases work of breathing Increases functional residual capacity Recruits collapsed alveoli Improves respiratory gas exchange Reverses hypoventilation Maintains upper
NEONATAL NEWS Here s Some More Good Poop
 NEONATALNEWS Here ssomemoregoodpoop WINTEREDITION2010 THISNEWSLETTERISPUBLISHEDPERIODICALLYBYTHENEONATOLOGISTSOF ASSOCIATESINNEWBORNMEDICINETOCONVEYNEWANDUPDATEDPOLICIES ANDGUIDELINESANDPROVIDEGENERALEDUCATIONTONICUCARETAKERSAT
NEONATALNEWS Here ssomemoregoodpoop WINTEREDITION2010 THISNEWSLETTERISPUBLISHEDPERIODICALLYBYTHENEONATOLOGISTSOF ASSOCIATESINNEWBORNMEDICINETOCONVEYNEWANDUPDATEDPOLICIES ANDGUIDELINESANDPROVIDEGENERALEDUCATIONTONICUCARETAKERSAT
Systems differ in their ability to deliver optimal humidification
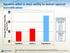 Average Absolute Humidity (mg H 2 O/L) Systems differ in their ability to deliver optimal humidification 45 Flows Tested 40 35 30 Optiflow Airvo 2 Vapotherm Vapotherm 5 L/min 10L/min 20L/min 30L/min 40L/min
Average Absolute Humidity (mg H 2 O/L) Systems differ in their ability to deliver optimal humidification 45 Flows Tested 40 35 30 Optiflow Airvo 2 Vapotherm Vapotherm 5 L/min 10L/min 20L/min 30L/min 40L/min
STOP ROP The STOP-ROP Multicenter Study Group: Pediatrics 105:295, 2000 Progression to Threshold Conventional Sat 89-94% STOP ROP
 Hrs TcPO2 > 80 nnhg (weeks 1 4) OXYGEN TARGETS: HOW GOOD ARE WE IN ACHIEVING THEM Oxygen Dependency GA wks Eduardo Bancalari MD University of Miami Miller School of Medicine Jackson Memorial Medical Center
Hrs TcPO2 > 80 nnhg (weeks 1 4) OXYGEN TARGETS: HOW GOOD ARE WE IN ACHIEVING THEM Oxygen Dependency GA wks Eduardo Bancalari MD University of Miami Miller School of Medicine Jackson Memorial Medical Center
Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome in premature infants
 Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
Original Policy Date
 MP 8.01.17 Inhaled Nitric Oxide Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy Index
MP 8.01.17 Inhaled Nitric Oxide Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy Index
INFANTS WITH birth weights less
 CLINICAL SCIENCES of Retinopathy of Prematurity Michael X. Repka, MD; Earl A. Palmer, MD; Betty Tung, MS; for the Cryotherapy for Retinopathy of Prematurity Cooperative Group Objective: To report the timing
CLINICAL SCIENCES of Retinopathy of Prematurity Michael X. Repka, MD; Earl A. Palmer, MD; Betty Tung, MS; for the Cryotherapy for Retinopathy of Prematurity Cooperative Group Objective: To report the timing
Home-Made Continuous Positive Airways Pressure Device may Reduce Mortality in Neonates with Respiratory Distress in Low-Resource Setting
 JOURNAL OF TROPICAL PEDIATRICS, VOL. 60, NO. 5, 2014 Home-Made Continuous Positive Airways Pressure Device may Reduce Mortality in Neonates with Respiratory Distress in Low-Resource Setting by Subhashchandra
JOURNAL OF TROPICAL PEDIATRICS, VOL. 60, NO. 5, 2014 Home-Made Continuous Positive Airways Pressure Device may Reduce Mortality in Neonates with Respiratory Distress in Low-Resource Setting by Subhashchandra
Hazards and Benefits of Postnatal Steroids. David J. Burchfield, MD Professor and Chief, Neonatology University of Florida
 Hazards and Benefits of Postnatal Steroids David J. Burchfield, MD Professor and Chief, Neonatology University of Florida Disclosures I have no financial affiliations or relationships to disclose. I will
Hazards and Benefits of Postnatal Steroids David J. Burchfield, MD Professor and Chief, Neonatology University of Florida Disclosures I have no financial affiliations or relationships to disclose. I will
Rango de saturacion de oxigeno: Cual es la evidencia?
 Rango de saturacion de oxigeno: Cual es la evidencia? Wally Carlo, M.D. University of Alabama at Birmingham Department of Pediatrics Division of Neonatology wcarlo@peds.uab.edu 1 2 Stevie Wonder 4 Objectives
Rango de saturacion de oxigeno: Cual es la evidencia? Wally Carlo, M.D. University of Alabama at Birmingham Department of Pediatrics Division of Neonatology wcarlo@peds.uab.edu 1 2 Stevie Wonder 4 Objectives
heated humidified high-flow nasal cannula therapy in children F A Hutchings, 1 T N Hilliard, 1 P J Davis 2 Review
 1 Department of Paediatric Respiratory Medicine, Bristol Royal Hospital for Children, Bristol, UK 2 Department of Paediatric Intensive Care, Bristol Royal Hospital for Children, Bristol, UK Correspondence
1 Department of Paediatric Respiratory Medicine, Bristol Royal Hospital for Children, Bristol, UK 2 Department of Paediatric Intensive Care, Bristol Royal Hospital for Children, Bristol, UK Correspondence
Continuous distending pressure for respiratory distress in preterm infants(review)
 Cochrane Database of Systematic Reviews Continuous distending pressure for respiratory distress in preterm infants(review) HoJJ,SubramaniamP,DavisPG Ho JJ, Subramaniam P, Davis PG.. Cochrane Database of
Cochrane Database of Systematic Reviews Continuous distending pressure for respiratory distress in preterm infants(review) HoJJ,SubramaniamP,DavisPG Ho JJ, Subramaniam P, Davis PG.. Cochrane Database of
Title Neonatal and Paediatric High-Flow Nasal Cannula Oxygen Therapy Guideline. Department Paediatrics / Neonates Date Issued
 Document Control Title Author Neonatal and Paediatric High-Flow Nasal Cannula Oxygen Therapy Guideline Author s job title Directorate Medical Version Department Paediatrics / Neonates Date Issued Status
Document Control Title Author Neonatal and Paediatric High-Flow Nasal Cannula Oxygen Therapy Guideline Author s job title Directorate Medical Version Department Paediatrics / Neonates Date Issued Status
Pneumothorax in preterm babies has been associated with an increased risk of intraventricular hemorrhage (IVH)
 Identification of Pneumothorax in Very Preterm Infants Risha Bhatia, MB, MA, MRCPCH, Peter G. Davis, MD, FRACP, Lex W. Doyle, MD, FRACP, Connie Wong, RN, and Colin J. Morley, MD, FRACP, FRCPCH Objective
Identification of Pneumothorax in Very Preterm Infants Risha Bhatia, MB, MA, MRCPCH, Peter G. Davis, MD, FRACP, Lex W. Doyle, MD, FRACP, Connie Wong, RN, and Colin J. Morley, MD, FRACP, FRCPCH Objective
Acute Paediatric Respiratory Pathway
 Guideline for the use of high Flow Nasal Cannula Oxygen Therapy (Optiflow or Airvo) in Children with Bronchiolitis or an acute respiratory illness Introduction: High flow nasal cannula (HFNC) oxygen enables
Guideline for the use of high Flow Nasal Cannula Oxygen Therapy (Optiflow or Airvo) in Children with Bronchiolitis or an acute respiratory illness Introduction: High flow nasal cannula (HFNC) oxygen enables
Early nasal intermittent positive pressure ventilation(nippv) versus early nasal continuous positive airway pressure(ncpap) for preterm
 Cochrane Database of Systematic Reviews Early nasal intermittent positive pressure ventilation(nippv) versus early nasal continuous positive airway pressure (NCPAP) for preterm infants(review) LemyreB,LaughonM,BoseC,DavisPG
Cochrane Database of Systematic Reviews Early nasal intermittent positive pressure ventilation(nippv) versus early nasal continuous positive airway pressure (NCPAP) for preterm infants(review) LemyreB,LaughonM,BoseC,DavisPG
Bronchopulmonary dysplasia: incidence and risk factors
 Original article Arch Argent Pediatr 2017;115(5):476-482 / 476 Bronchopulmonary dysplasia: incidence and risk factors Pablo H. Brener Dik, M.D. a, Yeimy M. Niño Gualdron, M.D. a, María F. Galletti, M.D.
Original article Arch Argent Pediatr 2017;115(5):476-482 / 476 Bronchopulmonary dysplasia: incidence and risk factors Pablo H. Brener Dik, M.D. a, Yeimy M. Niño Gualdron, M.D. a, María F. Galletti, M.D.
ARTIFICIAL INTELLIGENCE FOR PREDICTION OF SEPSIS IN VERY LOW BIRTH WEIGHT INFANTS
 ARTIFICIAL INTELLIGENCE FOR PREDICTION OF SEPSIS IN VERY LOW BIRTH WEIGHT INFANTS Markus Leskinen MD PhD, Neonatologist Children s Hospital, University of Helsinki and Helsinki University Hospital The
ARTIFICIAL INTELLIGENCE FOR PREDICTION OF SEPSIS IN VERY LOW BIRTH WEIGHT INFANTS Markus Leskinen MD PhD, Neonatologist Children s Hospital, University of Helsinki and Helsinki University Hospital The
Patent Ductus Arteriosus: Philosophy or Pathology?
 Patent Ductus Arteriosus: Philosophy or Pathology? Disclosure Ray Sato, MD is a speaker for Prolacta Biosciences, Inc. This presentation will discuss off-label uses of acetaminophen and ibuprofen. RAY
Patent Ductus Arteriosus: Philosophy or Pathology? Disclosure Ray Sato, MD is a speaker for Prolacta Biosciences, Inc. This presentation will discuss off-label uses of acetaminophen and ibuprofen. RAY
Exclusion Criteria 1. Operator or supervisor feels specific intra- procedural laryngoscopy device will be required.
 FELLOW Study Data Analysis Plan Direct Laryngoscopy vs Video Laryngoscopy Background Respiratory failure requiring endotracheal intubation occurs in as many as 40% of critically ill patients. Procedural
FELLOW Study Data Analysis Plan Direct Laryngoscopy vs Video Laryngoscopy Background Respiratory failure requiring endotracheal intubation occurs in as many as 40% of critically ill patients. Procedural
SECTION 1: INCLUSION, EXCLUSION & RANDOMISATION INFORMATION
 SECTION 1: INCLUSION, EXCLUSION & RANDOMISATION INFORMATION DEMOGRAPHIC INFORMATION Given name Family name Date of birth Consent date Gender Female Male Date of surgery INCLUSION & EXCLUSION CRITERIA YES
SECTION 1: INCLUSION, EXCLUSION & RANDOMISATION INFORMATION DEMOGRAPHIC INFORMATION Given name Family name Date of birth Consent date Gender Female Male Date of surgery INCLUSION & EXCLUSION CRITERIA YES
Nottingham Children s Hospital
 High Flow Nasal Cannula Therapy Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Guide line for the use of HFNCT (High Flow Nasal Cannula Therapy) Contact Name
High Flow Nasal Cannula Therapy Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Guide line for the use of HFNCT (High Flow Nasal Cannula Therapy) Contact Name
Von Reuss and CPAP, Disclosures CPAP. Noninvasive respiratory therapieswhy bother? Noninvasive respiratory therapies- types
 Noninvasive respiratory therapiesby a nose? NEO- The Conference for Neonatology February 21, 2014 Disclosures I have no relevant financial relationships to disclose or conflicts of interest to release.
Noninvasive respiratory therapiesby a nose? NEO- The Conference for Neonatology February 21, 2014 Disclosures I have no relevant financial relationships to disclose or conflicts of interest to release.
Non Invasive Ventilation In Preterm Infants. Manuel Sanchez Luna Hospital General Universitario Gregorio Marañón Complutense University Madrid
 Non Invasive Ventilation In Preterm Infants Manuel Sanchez Luna Hospital General Universitario Gregorio Marañón Complutense University Madrid Summary Noninvasive ventilation begings in the delivery room
Non Invasive Ventilation In Preterm Infants Manuel Sanchez Luna Hospital General Universitario Gregorio Marañón Complutense University Madrid Summary Noninvasive ventilation begings in the delivery room
Critical Appraisal Practicum. Fabio Di Bello Medical Implementation Manager
 Critical Appraisal Practicum Fabio Di Bello Medical Implementation Manager fdibello@ebsco.com What we ll talk about today: DynaMed process for appraising randomized trials and writing evidence summaries
Critical Appraisal Practicum Fabio Di Bello Medical Implementation Manager fdibello@ebsco.com What we ll talk about today: DynaMed process for appraising randomized trials and writing evidence summaries
Received: Jun 15, 2013; Accepted: Nov 08, 2013; First Online Available: Jan 24, 2014
 Original Article Iran J Pediatr Feb 2014; Vol 24 (No 1), Pp: 57-63 Management of Neonatal Respiratory Distress Syndrome Employing ACoRN Respiratory Sequence Protocol versus Early Nasal Continuous Positive
Original Article Iran J Pediatr Feb 2014; Vol 24 (No 1), Pp: 57-63 Management of Neonatal Respiratory Distress Syndrome Employing ACoRN Respiratory Sequence Protocol versus Early Nasal Continuous Positive
SWISS SOCIETY OF NEONATOLOGY. Neonatal gastric perforation
 SWISS SOCIETY OF NEONATOLOGY Neonatal gastric perforation September 2002 2 Zankl A, Stähelin J, Roth K, Boudny P and Zeilinger G, Children s Hospital of Aarau (ZA, SJ, RK, ZG) and Institute of Pathology
SWISS SOCIETY OF NEONATOLOGY Neonatal gastric perforation September 2002 2 Zankl A, Stähelin J, Roth K, Boudny P and Zeilinger G, Children s Hospital of Aarau (ZA, SJ, RK, ZG) and Institute of Pathology
SAMPLE. V.12.1 Special Report: Very Low Birthweight Neonates. I. Introduction
 I. Introduction V.12.1 Special Report: Very Low Birthweight Neonates The delivery of a very low birth weight infant continues to present many challenges to families and health care providers in spite of
I. Introduction V.12.1 Special Report: Very Low Birthweight Neonates The delivery of a very low birth weight infant continues to present many challenges to families and health care providers in spite of
Update on mangement of patent ductus arteriosus in preterm infants. Dr. Trinh Thi Thu Ha
 Update on mangement of patent ductus arteriosus in preterm infants Dr. Trinh Thi Thu Ha Outline 1. Overview of PDA 2. Timing of screening PDA? 3. When to treat PDA? Timing of ductal closure Prenatal
Update on mangement of patent ductus arteriosus in preterm infants Dr. Trinh Thi Thu Ha Outline 1. Overview of PDA 2. Timing of screening PDA? 3. When to treat PDA? Timing of ductal closure Prenatal
SWISS SOCIETY OF NEONATOLOGY. Spontaneous intestinal perforation or necrotizing enterocolitis?
 SWISS SOCIETY OF NEONATOLOGY Spontaneous intestinal perforation or necrotizing enterocolitis? June 2004 2 Stocker M, Berger TM, Neonatal and Pediatric Intensive Care Unit, Children s Hospital of Lucerne,
SWISS SOCIETY OF NEONATOLOGY Spontaneous intestinal perforation or necrotizing enterocolitis? June 2004 2 Stocker M, Berger TM, Neonatal and Pediatric Intensive Care Unit, Children s Hospital of Lucerne,
Transnasal Humidified Rapid Insufflation Ventilatory Exchange (THRIVE): An Optimal Method of Preoxygenation for General Anaesthesia in Obstetrics
 Transnasal Humidified Rapid Insufflation Ventilatory Exchange (THRIVE): An Optimal Method of Preoxygenation for General Anaesthesia in Obstetrics Dr E McMaster, Dr E Gent, Dr T Mahendrayogam, Dr A Surendran
Transnasal Humidified Rapid Insufflation Ventilatory Exchange (THRIVE): An Optimal Method of Preoxygenation for General Anaesthesia in Obstetrics Dr E McMaster, Dr E Gent, Dr T Mahendrayogam, Dr A Surendran
FANNP 28TH NATIONAL NNP SYMPOSIUM: CLINICAL UPDATE AND REVIEW OCTOBER 17-21, 2017
 Pulse Oximetry in the Delivery Room: Principles and Practice GS2 3 Jonathan P. Mintzer, MD, FAAP Assistant Professor of Pediatrics Stony Brook Children s Hospital, Division of Neonatal-Perinatal Medicine,
Pulse Oximetry in the Delivery Room: Principles and Practice GS2 3 Jonathan P. Mintzer, MD, FAAP Assistant Professor of Pediatrics Stony Brook Children s Hospital, Division of Neonatal-Perinatal Medicine,
By: Armend Lokku Supervisor: Dr. Lucia Mirea. Maternal-Infant Care Research Center, Mount Sinai Hospital
 By: Armend Lokku Supervisor: Dr. Lucia Mirea Maternal-Infant Care Research Center, Mount Sinai Hospital Background My practicum placement was at the Maternal-Infant Care Research Center (MiCare) at Mount
By: Armend Lokku Supervisor: Dr. Lucia Mirea Maternal-Infant Care Research Center, Mount Sinai Hospital Background My practicum placement was at the Maternal-Infant Care Research Center (MiCare) at Mount
9/15/2017. Disclosures. Heated High Flow Nasal Cannula: Hot Air or Optimal Noninvasive Support? Objectives. Aerogen Pharma
 Heated High Flow Nasal Cannula: Hot Air or Optimal Noninvasive Support? Rob DiBlasi RRT-NPS, FAARC Program Manager Research/QI, Respiratory Therapy Principle Investigator, Seattle Children s Research Institute
Heated High Flow Nasal Cannula: Hot Air or Optimal Noninvasive Support? Rob DiBlasi RRT-NPS, FAARC Program Manager Research/QI, Respiratory Therapy Principle Investigator, Seattle Children s Research Institute
Tehran University of Medical Sciences, Tehran, Iran. Received: 22 May 2013; Received in revised form: 12 Sep. 2013; Accepted: 17 Sep.
 ORIGINAL ARTICLE Comparison of INSURE Method with Conventional Mechanical Ventilation after Surfactant Administration in Preterm Infants with Respiratory Distress Syndrome: Therapeutic Challenge Fatemeh
ORIGINAL ARTICLE Comparison of INSURE Method with Conventional Mechanical Ventilation after Surfactant Administration in Preterm Infants with Respiratory Distress Syndrome: Therapeutic Challenge Fatemeh
Early (< 8 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants (Review)
 Early (< 8 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants (Review) Halliday HL, Ehrenkranz RA, Doyle LW This is a reprint of a Cochrane review, prepared and maintained
Early (< 8 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants (Review) Halliday HL, Ehrenkranz RA, Doyle LW This is a reprint of a Cochrane review, prepared and maintained
Clinicoetiological profile and risk assessment of newborn with respiratory distress in a tertiary care centre in South India
 International Journal of Contemporary Pediatrics Sahoo MR et al. Int J Contemp Pediatr. 2015 Nov;2(4):433-439 http://www.ijpediatrics.com pissn 2349-3283 eissn 2349-3291 Research Article DOI: http://dx.doi.org/10.18203/2349-3291.ijcp20150990
International Journal of Contemporary Pediatrics Sahoo MR et al. Int J Contemp Pediatr. 2015 Nov;2(4):433-439 http://www.ijpediatrics.com pissn 2349-3283 eissn 2349-3291 Research Article DOI: http://dx.doi.org/10.18203/2349-3291.ijcp20150990
O) or to bi-level NCPAP (group B; n=20, lower CPAP level=4.5 cm H 2
 1 NICU, Children s Hospital, Via Castelvetro, Milan, Italy 2 Laboratory, V.Buzzi Children s Hospital ICP, Milan, Italy 3 Department of Statistics, Catholic University, Milan, Italy Correspondence to Gianluca
1 NICU, Children s Hospital, Via Castelvetro, Milan, Italy 2 Laboratory, V.Buzzi Children s Hospital ICP, Milan, Italy 3 Department of Statistics, Catholic University, Milan, Italy Correspondence to Gianluca
Research Roundtable Summary
 Research Roundtable Summary 10 TENTH in a Series of Seminars on MCHB-funded Research Projects Early Cortisol Deficiency and Bronchopulmonary Dysplasia October 18, 1995 Parklawn Building Potomac Conference
Research Roundtable Summary 10 TENTH in a Series of Seminars on MCHB-funded Research Projects Early Cortisol Deficiency and Bronchopulmonary Dysplasia October 18, 1995 Parklawn Building Potomac Conference
1
 1 2 3 RIFAI 5 6 Dublin cohort, retrospective review. Milrinone was commenced at an initial dose of 0.50 μg/kg/minute up to 0.75 μg/kg/minute and was continued depending on clinical response. No loading
1 2 3 RIFAI 5 6 Dublin cohort, retrospective review. Milrinone was commenced at an initial dose of 0.50 μg/kg/minute up to 0.75 μg/kg/minute and was continued depending on clinical response. No loading
Neonatal Resuscitation in What is new? How did we get here? Steven Ringer MD PhD Harvard Medical School May 25, 2011
 Neonatal Resuscitation in 2011- What is new? How did we get here? Steven Ringer MD PhD Harvard Medical School May 25, 2011 Conflicts I have no actual or potential conflict of interest in relation to this
Neonatal Resuscitation in 2011- What is new? How did we get here? Steven Ringer MD PhD Harvard Medical School May 25, 2011 Conflicts I have no actual or potential conflict of interest in relation to this
Dr. AM MAALIM KPA 2018
 Dr. AM MAALIM KPA 2018 Journey Towards Lung protection Goals of lung protection Strategies Summary Conclusion Before 1960: Oxygen; impact assessed clinically. The 1960s:President JFK, Ventilators mortality;
Dr. AM MAALIM KPA 2018 Journey Towards Lung protection Goals of lung protection Strategies Summary Conclusion Before 1960: Oxygen; impact assessed clinically. The 1960s:President JFK, Ventilators mortality;
Objectives. Apnea Definition and Pitfalls. Pathophysiology of Apnea. Apnea of Prematurity and hypoxemia episodes 5/18/2015
 Apnea of Prematurity and hypoxemia episodes Deepak Jain MD Care of Sick Newborn Conference May 2015 Objectives Differentiating between apnea and hypoxemia episodes. Pathophysiology Diagnosis of apnea and
Apnea of Prematurity and hypoxemia episodes Deepak Jain MD Care of Sick Newborn Conference May 2015 Objectives Differentiating between apnea and hypoxemia episodes. Pathophysiology Diagnosis of apnea and
Children & Young People s Directorate Paediatric-Neonatal Guidelines Checklist & Version Control Sheet
 Children & Young People s Directorate Paediatric-Neonatal Guidelines Checklist & Version Control Sheet 1. Name of Guideline / Policy/ Procedure 2. Purpose of Procedure/ Guidelines/ Protocol Guideline for
Children & Young People s Directorate Paediatric-Neonatal Guidelines Checklist & Version Control Sheet 1. Name of Guideline / Policy/ Procedure 2. Purpose of Procedure/ Guidelines/ Protocol Guideline for
Form 6: Baby Outcomes Outcomes at THIS hospital
 Form 6: Baby Outcomes Outcomes at THIS hospital Please complete in black ballpoint pen Hospital name: Form 6: Baby Outcomes Please complete this form when a Baby-OSCAR participant is transferred to another
Form 6: Baby Outcomes Outcomes at THIS hospital Please complete in black ballpoint pen Hospital name: Form 6: Baby Outcomes Please complete this form when a Baby-OSCAR participant is transferred to another
GE Healthcare. Non Invasive Ventilation (NIV) For the Engström Ventilator. Relief, Relax, Recovery
 GE Healthcare Non Invasive Ventilation (NIV) For the Engström Ventilator Relief, Relax, Recovery COPD is currently the fourth leading cause of death in the world, and further increases in the prevalence
GE Healthcare Non Invasive Ventilation (NIV) For the Engström Ventilator Relief, Relax, Recovery COPD is currently the fourth leading cause of death in the world, and further increases in the prevalence
TRAINING NEONATOLOGY SILVANA PARIS
 TRAINING ON NEONATOLOGY SILVANA PARIS RESUSCITATION IN DELIVERY ROOM INTRODUCTION THE GLOBAL RESUSCITATION BURDEN IN NEWBORN 136 MILL NEWBORN BABIES EACH YEAR (WHO WORLD REPORT) 5-8 MILL NEWBORN INFANTS
TRAINING ON NEONATOLOGY SILVANA PARIS RESUSCITATION IN DELIVERY ROOM INTRODUCTION THE GLOBAL RESUSCITATION BURDEN IN NEWBORN 136 MILL NEWBORN BABIES EACH YEAR (WHO WORLD REPORT) 5-8 MILL NEWBORN INFANTS
** SURFACTANT THERAPY**
 ** SURFACTANT THERAPY** Full Title of Guideline: Surfactant Therapy Author (include email and role): Stephen Wardle (V4) Reviewed by Dushyant Batra Consultant Neonatologist Division & Speciality: Division:
** SURFACTANT THERAPY** Full Title of Guideline: Surfactant Therapy Author (include email and role): Stephen Wardle (V4) Reviewed by Dushyant Batra Consultant Neonatologist Division & Speciality: Division:
Predicting Mortality and Intestinal Failure in Neonates with Surgical Necrotizing Enterocolitis
 Predicting Mortality and Intestinal Failure in Neonates with Surgical Necrotizing Enterocolitis Darshna Bhatt MHA DO, Curtis Travers MPH, Ravi M. Patel MD MSc, Julia Shinnick MD, Kelly Arps MD, Sarah Keene,
Predicting Mortality and Intestinal Failure in Neonates with Surgical Necrotizing Enterocolitis Darshna Bhatt MHA DO, Curtis Travers MPH, Ravi M. Patel MD MSc, Julia Shinnick MD, Kelly Arps MD, Sarah Keene,
Curriculum vitae. ZohrehBadiee. MD. Personal Data: Name: zohrehbadiee. Birth Date: Sex: female
 Curriculum vitae ZohrehBadiee. MD Personal Data: Name: zohrehbadiee Birth Date: 1969 Sex: female Academic status: Professor of Pediatrics, Neonatologist, Address: neonatal intensive care unit, Pediatric
Curriculum vitae ZohrehBadiee. MD Personal Data: Name: zohrehbadiee Birth Date: 1969 Sex: female Academic status: Professor of Pediatrics, Neonatologist, Address: neonatal intensive care unit, Pediatric
Quality Improvement Approaches to BPD. Jay P. Goldsmith, M.D. Tulane University New Orleans, Louisiana
 Quality Improvement Approaches to BPD Jay P. Goldsmith, M.D. Tulane University New Orleans, Louisiana goldsmith.jay@gmail.com No conflicts of interest to declare There is nothing more dangerous to the
Quality Improvement Approaches to BPD Jay P. Goldsmith, M.D. Tulane University New Orleans, Louisiana goldsmith.jay@gmail.com No conflicts of interest to declare There is nothing more dangerous to the
Evaluating the Effect of Flow and Interface Type on Pressures Delivered With Bubble CPAP in a Simulated Model
 Evaluating the Effect of Flow and Interface Type on Pressures Delivered With Bubble CPAP in a Simulated Model Stephanie A Bailes RRT-NPS NREMT-B, Kimberly S Firestone MSc RRT, Diane K Dunn RRT-NPS, Neil
Evaluating the Effect of Flow and Interface Type on Pressures Delivered With Bubble CPAP in a Simulated Model Stephanie A Bailes RRT-NPS NREMT-B, Kimberly S Firestone MSc RRT, Diane K Dunn RRT-NPS, Neil
Effects of Indomethacin Prophylaxis Timing on IVH and PDA in Extremely Low Birth Weight (ELBW) Infants
 Effects of Indomethacin Prophylaxis Timing on IVH and PDA in Extremely Low Birth Weight (ELBW) Infants Hussnain Mirza, University of Central Florida Abbot R. Laptook, Brown University William Oh, Brown
Effects of Indomethacin Prophylaxis Timing on IVH and PDA in Extremely Low Birth Weight (ELBW) Infants Hussnain Mirza, University of Central Florida Abbot R. Laptook, Brown University William Oh, Brown
High-Flow Nasal Cannula Utilization in Pediatric Critical Care
 High-Flow Nasal Cannula Utilization in Pediatric Critical Care Kristen D Coletti MD, Dayanand N Bagdure MBBS, Linda K Walker MD, Kenneth E Remy MD MHSc, and Jason W Custer MD BACKGROUND: High-flow nasal
High-Flow Nasal Cannula Utilization in Pediatric Critical Care Kristen D Coletti MD, Dayanand N Bagdure MBBS, Linda K Walker MD, Kenneth E Remy MD MHSc, and Jason W Custer MD BACKGROUND: High-flow nasal
OmniOx (HFT 500) Jan Prepared by MEKICS. OmniOx-HFT500 Jan ver1.1 1
 OmniOx (HFT 500) Jan. 2013. Prepared by MEKICS OmniOx-HFT500 Jan. 2013 ver1.1 1 0. Index 1. Introduce of OmniOx (HTF500) 2. Specification (HFT, CPAP+) 3. The World Best Features 2 OmniOx-HFT500 Jan. 2013
OmniOx (HFT 500) Jan. 2013. Prepared by MEKICS OmniOx-HFT500 Jan. 2013 ver1.1 1 0. Index 1. Introduce of OmniOx (HTF500) 2. Specification (HFT, CPAP+) 3. The World Best Features 2 OmniOx-HFT500 Jan. 2013
Simulation 3: Post-term Baby in Labor and Delivery
 Simulation 3: Post-term Baby in Labor and Delivery Opening Scenario (Links to Section 1) You are an evening-shift respiratory therapist in a large hospital with a level III neonatal unit. You are paged
Simulation 3: Post-term Baby in Labor and Delivery Opening Scenario (Links to Section 1) You are an evening-shift respiratory therapist in a large hospital with a level III neonatal unit. You are paged
Minimal Enteral Nutrition
 Abstract Minimal Enteral Nutrition Although parenteral nutrition has been used widely in the management of sick very low birth weight infants, a smooth transition to the enteral route is most desirable.
Abstract Minimal Enteral Nutrition Although parenteral nutrition has been used widely in the management of sick very low birth weight infants, a smooth transition to the enteral route is most desirable.
