Authors Chappuis, F; Stivanello, E; Adams, K; Kidane, S; Pittet, A; Bovier, P A. Published by: American Society of Tropical Medicine and Hygiene
|
|
|
- Lorena Snow
- 5 years ago
- Views:
Transcription
1 MSF Field Research Card Agglutination Test for Trypanosomiasis (CATT) End- Dilution Titer and Cerebrospinal Fluid Cell Count as Predictors of Human African Trypanosomiasis (Trypanosoma brucei gambiense) Among Serologically Suspected Individuals in Southern Sudan. Authors Chappuis, F; Stivanello, E; Adams, K; Kidane, S; Pittet, A; Bovier, P A Citation Publisher Journal Rights Card Agglutination Test for Trypanosomiasis (CATT) End-Dilution Titer and Cerebrospinal Fluid Cell Count as Predictors of Human African Trypanosomiasis (Trypanosoma brucei gambiense) Among Serologically Suspected Individuals in Southern Sudan. 2004, 71 (3):313-7 Am. J. Trop. Med. Hyg. Published by: American Society of Tropical Medicine and Hygiene American Journal of Tropical Medicine and Hygiene Archived on this site with the kind permission of the American Society of Tropical Medicine and Hygiene, Downloaded 26-Aug :24:40 Link to item
2 Am. J. Trop. Med. Hyg., 71(3), 2004, pp Copyright 2004 by The American Society of Tropical Medicine and Hygiene CARD AGGLUTINATION TEST FOR TRYPANOSOMIASIS (CATT) END-DILUTION TITER AND CEREBROSPINAL FLUID CELL COUNT AS PREDICTORS OF HUMAN AFRICAN TRYPANOSOMIASIS (TRYPANOSOMA BRUCEI GAMBIENSE) AMONG SEROLOGICALLY SUSPECTED INDIVIDUALS IN SOUTHERN SUDAN FRANÇOIS CHAPPUIS, ELISA STIVANELLO, KAREN ADAMS, SOLON KIDANE, ANNE PITTET, AND PATRICK A. BOVIER Médecins Sans Frontières, Swiss Section, Geneva, Switzerland; Department of Community Medicine, Geneva University Hospital, Geneva, Switzerland Abstract. The diagnosis of human African trypanosomiasis (HAT) due to Trypanosoma brucei gambiense relies on an initial serologic screening with the card agglutination test for trypanosomiasis (CATT) for T. b. gambiense, followed by parasitologic confirmation in most endemic areas. Unfortunately, field parasitologic methods lack sensitivity and the management of serologically suspected individuals (i.e., individuals with a positive CATT result but negative parasitology) remains controversial. In Kajo-Keji County in southern Sudan, we prospectively collected sociodemographic and laboratory data of a cohort of 2,274 serologically suspected individuals. Thirty-three percent (n 749) attended at least one follow-up visit and HAT was confirmed in 64 (9%) cases. Individuals with lower initial CATT-plasma (CATT-P) end-dilution titers had lowest risks (10.4 and 13.8/100 person-years for 1:4 and 1:8 titers, respectively) that significantly increased for higher dilutions: relative risks 5.1 (95% confidence interval [CI] ) and 4.6 (95% CI ) for 1:16 and 1:32 titers, respectively. The cumulative yearly risk was also high (76%) in individuals found with cells in the cerebrospinal fluid, but this involved only eight patients. Adjustment for potential confounders did not affect the results. In conclusion, treatment with pentamidine should be considered for all serologically suspected individuals with a CATT-P end-dilution titer 1:16 in areas of a moderate to high prevalence of HAT. INTRODUCTION 313 Field diagnosis of human African trypanosomiasis (HAT) due to Trypanosoma brucei gambiense relies on the initial screening with the card agglutination trypanosomiasis test (CATT/T. b. gambiense) in most endemic areas. The CATT/ T. b. gambiense is a cheap, quick, and practical serologic test that has been widely used since it was developed in It is a highly sensitive test when applied on undiluted whole blood (CATT-WB). 2 Despite its good specificity, the positive predictive value (PPV) of the CATT-WB is limited because the test is used for mass screening of populations in which the prevalence of HAT rarely exceeds 5%. 2,3 The specificity of the CATT can be further improved when performed on plasma (CATT-P) or serum diluted to 1:4, 4 but this is still insufficient. Confirmatory diagnosis thus relies on the finding of trypanosomes in the lymph nodes, blood, or cerebrospinal fluid (CSF). However, despite the use of concentration methods to detect trypanosomes in the blood or CSF, the field parasitologic methods have a limited sensitivity and fail to detect all infected individuals. 5,6 In Kajo-Keji County in southern Sudan, Médecins Sans Frontières has been conducting since June 2000 a sleeping sickness control program that relies on the systematic screening of the entire county population and treatment of all infected patients. The main objective of the program is to decrease the prevalence of HAT to less than 0.5% in all villages. Individuals with a positive CATT-P titer 1:4 without parasitologic confirmation are considered as serologically suspected and are followed-up for one year. However, the efficiency of this strategy remains controversial, particularly in regions such as southern Sudan where follow-up is difficult because of long distances, lack of transport, and population movements. In such a setting, the determination of a CATT-P dilution titer above which individuals are likely to have the disease would allow the treatment of these infected individuals at an earlier stage and avoid the risk of losing these patients during follow-up. Moreover, with the treatment of a higher proportion of infected individuals who are the main reservoir of T. b. gambiense, control of the disease may be accelerated. The few published studies addressing this question have led to controversial results. Of 86 serologically suspected individuals (CATT-P titer 1/4) followed in Angola, Simarro and others found that 52% of the individuals with a CATT end titer 1/16 were diagnosed with HAT during follow-up, but none of the individuals with lower CATT end-titers were diagnosed. 7 These investigators recommended treatment with pentamidine of all serologically suspected individuals with a CATT end titer 1/16 in this region. Conversely, Garcia and others found only one patient with HAT during a two-year longitudinal follow-up of 77 serologically suspected individuals in a low prevalence area of Côte d Ivoire. 8 However, in the latter study, the CATT was performed on undiluted plasma and on blood with a maximum dilution of 1:4. In this study, we retrospectively analyzed the initial CATT-P end-dilution titer and other biologic parameters as possible determinants of outcome in a large cohort of serologically suspected individuals followed-up for one year in Kajo-Keji County in southern Sudan. MATERIALS AND METHODS Between June 2000 and March 2002, all individuals presenting at the Sleeping Sickness Treatment Center (SSTC) located in Kiri in Kajo-Keji County were screened for HAT using a screening (passive) and diagnostic protocol described elsewhere. 9 Active screening was carried out with two mobile teams who systematically visited all villages since April All individuals found with a positive CATT-P titer of 1:4 during active screening were referred to the SSTC laboratory for further diagnostic procedures. Serologically suspected individuals were defined as indi-
3 314 CHAPPUIS AND OTHERS viduals with a positive CATT-P titer of 1/4 with no trypanosomes found in the cervical lymph nodes, blood (by the quantitative buffy coat technique or the hematocrit centrifugation technique), or CSF (after double centrifugation), and a CSF cell count 20/mm 3. Using the same plasma sample, a serial two-fold dilution in CATT buffer up to a titer of 1:32 was performed for all serologically suspected individuals between June 2000 and March 2002, and the end-dilution titer was recorded. All serologically suspected individuals were then discharged from the SSTC laboratory and were requested to come back for control visits after 3, 6, and 12 months. The screening and diagnostic algorithm, as performed during the initial evaluation, 9 was repeated at each control visit. Individuals who did not attend follow-up were traced at home by locally trained Sleeping Sickness Assistants. Vehicle transport was not provided because of logistic constraints. The Ethical Review Board of Médecins Sans Frontières did not require formal ethical review because the analysis was performed on data routinely collected during the program. Data collection. All sociodemographic and laboratory data, including the results of the CATT-P end-dilution titer and the laboratory results of the follow-up visits, were entered into YoTryp, a Microsoft Access-based software (Microsoft, Redmond, WA) specifically designed for the program. In March 2003, all data was transferred onto SPSS version 11.0 statistical software (SPSS, Inc., Chicago, IL) and analyzed. Statistical analysis. Descriptive statistical analysis of sociodemographic characteristics and laboratory test results at screening of all serologically suspected individuals were performed using frequency tables. All subjects with at least one follow-up and the respective outcome were selected to calculate the incidence of HAT, using the number of HAT cases detected during follow-up as the numerator and the total person-year as the denominator. To assess whether selection bias occurred, sociodemographic characteristics and laboratory test results at screening of subjects with at least one follow-up were compared with those lost to follow-up, using crosstabulations and chi-square tests. To study the association between laboratory test results at screening and the risk of developing HAT during follow-up, we calculated the incidence of HAT across four levels of CATT end-dilution serologic results (1:4, 1:8, 1:16, and 1:32) and three levels of CSF cell counts (0 5, 6 10, and 11 20) and the corresponding relative risks. Cox models were used to calculate relative risks adjusted for other significant factors and cumulative hazard over time of follow-up. The follow-up attendance of the serologically suspected individuals was low and decreased over time: 553 individuals (24%) were controlled at 3 months, 255 (11%) at 6 months, and 92 (4%) at 12 months. A total of 749 individuals (33%) were controlled at least once during follow-up. When compared with the 1,525 individuals who were lost to follow-up, these 749 individuals had a similar age, sex, CATT enddilution titer distribution, and CSF cell count, but were statistically (P < 0.05) more likely to be displaced Sudanese, to come from the Bamurye camp, and to be detected by passive screening (Table 1). Human African trypanosomiasis was diagnosed in 64 serologically suspected individuals during follow-up: 28 patients (44%) with first-stage illness and 36 patients (56%) with second-stage (or neurologic stage) illness. This represented 4% (64 of 1,658) of all patients diagnosed with HAT in the Kiri SSTC during the same period, 3% (64 of 2,274) of all individuals initially defined as serologically suspected, and 9% (64 of 749) of those who attended at least one follow-up. Predictors of a diagnosis of HAT during follow-up of serologically suspected patients. Among the 749 serologically suspected individuals who were controlled at least once during follow-up, the sociodemographic characteristics and mode of screening of the 64 individuals diagnosed with HAT during follow-up were similar to those of the other 685 individuals. The proportion of serologically suspected individuals eventually diagnosed with HAT increased with higher initial CATT-P end-dilution titers (5%, 6.7%, 23.3%, and 19.6% for 1:4, 1:8, 1:16, and 1:32 titers, respectively) and with higher initial CSF cell counts (7.3%, 16.7%, and 34.8% for 0 5, 6 10, and cells, respectively). Of the 64 HAT cases, 29 (45%) patients had an initial CATT end-titer of 1:16 or 1:32, while 8 (13%) patients (including 7 with second-stage illness) had an initial CSF cell counts of cells. The 749 serologically suspected individuals were followed for a total of 129,201 days. The overall risk/person-year of being diagnosed with HAT during follow-up was 18.1% and increased for higher CATT-P dilutions and higher CSF cell counts (Table 2). The risk was significantly higher in individuals with a CATT end-dilution titer 1/16 (Figure 1A) and in individuals with a CSF cell count of (Figure 1B). This difference remained significant after adjustment for age, sex, mode of screening, CSF cell count, and CATT end-dilution titer. The combination of a CATT titer 1/16 and a CSF cell count of had an additive effect on the cumulative risk, but this was found in only four individuals. RESULTS Between June 2000 and March 2002, 1,658 patients were diagnosed with HAT (740 with stage 1 disease and 918 with stage 2 disease) in the Kiri SSTC, while 2,355 individuals were defined as serologically suspected. The CATT-P end-dilution titer was determined in 2,274 of these 2,355 individuals (97%): the end-dilution titer was 1:4 in 1,086 (48%), 1:8 in 761 (33%), 1:16 in 264 (12%), and 1:32 in 163 (7%). The CATT-P end-dilution titer was not determined in 84 individuals (3%), who were therefore excluded from further analysis. The sociodemographic and laboratory characteristics of the 2,274 serologically suspected individuals included in the analysis are shown in Table 1. DISCUSSION Because of the insufficient sensitivity of the current parasitologic field techniques and the limited specificity of the CATT-WB and CATT-P 1/4, 4 many African patients infected with T. b. gambiense go home undetected and untreated after completion of investigations. This situation is unsatisfactory not only for the infected individual, who will be more likely to be diagnosed at a later stage or simply die at home, but also for the community. Indeed, these individuals might maintain the human reservoir of the parasite and contribute to further transmission of the disease in the community. A possible approach is to follow-up and re-examine all serologically suspected individuals at regular intervals to detect and treat
4 PREDICTORS OF SLEEPING SICKNESS IN SUDAN 315 TABLE 1 Sociodemographic characteristics and laboratory test results for African trypanomiasis at screening at 2,274 serologically suspect individuals, 749 individuals with at least one follow-up, and 1,525 individuals lost to follow-up in Kajo-Keji County, southern Sudan* Characteristics All serologically suspected no. (%) 1 Control visit during follow-up no. (%) Lost to follow-up no. (%) P Sex 0.97 Male 994 (44) 327 (44) 667 (44) Female 1,280 (56) 422 (56) 858 (56) Age (years) (4) 20 (3) 62 (4) (27) 209 (28) 403 (26) >14 1,580 (70) 520 (69) 1,060 (70) Status <0.001 Kajo-Keji resident 1,521 (67) 448 (60) 1,073 (70) Sudanese from displaced camp 686 (30) 279 (37) 407 (27) Non-Kajo-Keji residents 67 (3) 22 (3) 45 (3) Payam of residence <0.001 Bamurye 570 (25) 260 (35) 310 (20) Kangapo (20) 139 (19) 327 (21) Kangapo (41) 220 (29) 701 (46) Lirye 111 (5) 54 (7) 57 (4) Livolo 105 (5) 43 (6) 62 (4) Lobonok/Ngepo 37 (2) 15 (2) 22 (2) Other county or Uganda 64 (3) 18 (2) 46 (3) Mode of screening <0.001 Passive 1,360 (60) 539 (72) 821 (54) Active 914 (40) 210 (28) 704 (46) CATT end dilution :4 1,086 (48) 364 (49) 722 (47) 1:8 761 (33) 253 (34) 508 (33) 1: (12) 86 (11) 178 (12) 1: (7) 46 (6) 117 (8) CSF cells ,104 (93) 683 (92) 1,421 (94) (5) 35 (5) 70 (5) (2) 23 (3) 26 (2) * CATT card agglutination test for trypanosomiasis; CSF cerebrospinal fluid. by Pearson chi square test. Sixteen missing values. those with a parasitologic confirmation during follow-up. Unfortunately, this strategy is very difficult to implement in the field and its effectiveness is low, as shown in our program in Kajo-Keji County in southern Sudan. Despite an effective team of 30 community-based sleeping sickness assistants, all well-motivated and regularly paid, the follow-up attendance of serologically suspected individuals and the overall yield of this strategy remained low. Therefore, the early identification of serologically suspected individuals likely to be infected with T. b. gambiense TABLE 2 Risk of developing human African trypanosomiasis according to CATT end dilution titer and CSF cell count at screening in 749 serologically suspected patients in Kajo-Keji county, southern Sudan* Parameter No. of cases of HAT diagnosed No. serologically suspected Total days of follow-up Risk/100 person-years Relative risk (95% CI) Adjusted relative risk (95% CI) CATT end dilution titer 1: , : , ( ) 1.1 ( ) 1: , ( ) 4.4 ( ) 1: , ( ) 4.1 ( ) CSF cell count , , ( ) 2.0 ( ) , ( ) 4.4 ( ) Combination CATT 1:4 1:8 and cells , CATT 1:16 1:32 and cells , ( ) 4.5 ( ) CATT 1:4 1:8 and cells , ( ) 5.0 ( ) CATT 1:16 1:32 and cells , ( ) 15.5 ( ) * CATT card agglutination test for trypanosomiasis test; CSF cerebrospinal fluid; CI confidence interval. All relative risk adjusted for age, sex, mode of screening, CATT end dilution titer, and CSF cell count. Five missing values.
5 316 CHAPPUIS AND OTHERS FIGURE 1. Cumulative risk of being diagnosed with human African trypanosomiasis (HAT) in relation to A, the card agglutination trypanosomiasis test (CATT) plasma end-dilution titer and B, the cerebrospinal fluid (CSF) cell count during a one-year follow-up of 749 serologically suspected individuals in Kajo-Keji County in southern Sudan. would be very useful. In this study, we analyzed the initial CATT end-dilution titers and the CSF cell counts of all serologically suspected individuals (n 749) examined at least once during follow-up. We found that individuals with a CATT end-dilution titer of 1:16 or 1:32 had a higher risk of being diagnosed with HAT during follow-up (risk for 100 person-years 53% and 47%, respectively), a significantly higher risk (adjusted relative risk [RR] 4.4 and 4.1, respectively) than individuals with a CATT end-dilution titer of 1:4 (risk for 100 person-years 10%). Similar findings have been reported by Simarro and others in a smaller study in Angola. 7 If the 132 individuals with a CATT titer 1/16 had been treated after initial evaluation, 29 (22%) individuals subsequently diagnosed with HAT would have been treated and cured (provided that treatment was 100% efficient). However, 103 (78%) individuals would have been treated in excess. The PPV of a CATT titer 1/16, like any other diagnostic test, depends on the prevalence of the disease in the tested population. The PPV was moderate (22%) in Kajo- Keji, where the mean prevalence of HAT found during active screening sessions was 1%, but was higher (52%) in the study by Simarro and others in Angola, in which the prevalence of HAT in the studied focus was 1.9%. 7 This concept is crucial for policy makers and it would be extremely useful in the future to gather and analyze all published and unpublished data on that issue to determine a prevalence cut-off above which treatment of individuals with a CATT titer 1/16 is worthwhile. The decision should also depend on other factors such as the availability and cost of treatment, the capacity to perform repeated active screening sessions (potentially limited by geographic, safety, time, and/or transport constraints), and the feasibility and expected effectiveness of a follow-up strategy. As suggested by Simarro and others, pentamidine is the only therapeutic option for treating individuals with a CATT titer 1/16. 7 Lumbar puncture should be performed in these individuals to rule out second-stage illness (presence of trypanosomes and/or >20 cells in the CSF). Despite the painful daily intramuscular injections and the possible occurrence of side effects such as hypoglycemia and hypotension, pentamidine therapy is relatively short (seven days) and very safe. In Kajo-Keji, only 2 of more than 2,000 patients treated with pentamidine died during treatment, and this was due to non drug-related causes. Pentamidine would not only be very efficient in treating undiagnosed first-stage patients (CSF cell counts 0 5 cells), who would represent the highest proportion of individuals (92% in Kajo-Keji), but would also have some degree of efficiency in undiagnosed early second-stage patients (CSF cell counts 6 20 cells). 10,11 Ongoing separate studies are currently assessing the efficacy of a shortened schedule (three days) of pentamidine and an oral drug, DB 289, for the treatment of first-stage HAT. The availability of an oral treatment or, to a lesser extent, a shorter parenteral treatment would greatly facilitate the treatment of both firststage and serologically suspected patients in the field. An initial CSF cell count of was also found to be a strong risk factor for serologically suspected individuals diagnosed with HAT during follow-up with a risk for 100 personyears of 76% and an adjusted RR of 4.4 compared with individuals with 0 5 cells in the CSF. However, this risk factor was only found in 8 (13%) individuals diagnosed with HAT during follow-up, all but one with second-stage disease. If one considers the high toxicity of melarsoprol and the complicated schedule of eflornithine (14 days of 4 daily slow infusions), the only two drugs currently registered for the treatment of second-stage illness, their use should be restricted to confirmed cases. Our study had one main potential limitation. The extrapolation of our results to the whole population of serologically suspected individuals could be limited by the low attendance rate obtained during follow-up. Individuals with an unknown outcome (lost to follow-up) had a significantly different status, location of residence, and mode of screening, but had comparable CATT-P end-dilution titers and CSF cell counts, the only factors found to be associated with a higher risk of HAT. The attendance of follow-up visits was therefore influenced more by factors such as distance, mobility and motivation. Therefore, we believe that it is reasonable to extrapolate
6 PREDICTORS OF SLEEPING SICKNESS IN SUDAN 317 our results to the whole population of serologically suspected individuals diagnosed during the study period in Kajo-Keji. In conclusion, we showed that the follow-up of serologically suspected individuals had a low effectiveness and yield in a field setting such as southern Sudan. We identified a CATT-P titer 1/16 as a useful predictor of HAT in this population. We suggest that treatment of these individuals with pentamidine is worthwhile, both on an individual and public health basis, if the prevalence of HAT in the population investigated is sufficiently high. Which cut-off of prevalence to choose remains an open question. It should probably not be less than 1% and the decision should be tempered by other factors such as distance to the treatment center and means of transportation. Based on these results, all serologically suspected individuals with a CATT-P titer 1/16 diagnosed in Kajo-Keji County during passive screening or firstround active screening (the settings where the highest prevalence of HAT is found) since March 2002 have received treatment with pentamidine. Received February 10, Accepted for publication April 5, Acknowledgments: We thank the Sudanese medical and non-medical staff of the Kajo-Keji Sleeping Sickness Control Program for their dedicated work. Financial support: This study was supported by Médecins Sans Frontières, Swiss Section. Authors addresses: François Chappuis, Médecins Sans Frontières, Swiss Section, Rue de Lausanne 78, 1203 Geneva, Switzerland and Travel and Migration Medicine Unit, Department of Community Medicine, Geneva University Hospital, Rue Micheli-du-Crest 24, 1211 Geneva, Switzerland, Telephone: , Fax: , francois.chappuis@hcuge.ch. Elisa Stivanello, Karen Adams, Solon Kidane, and Anne Pittet, Médecins Sans Frontières, Swiss Section, Rue de Lausanne 78, 1203 Geneva, Switzerland, Telephone: , Fax: Patrick A. Bovier, Department of Community Medicine, Geneva University Hospital, Rue Micheli-du-Crest 24, 1211 Geneva 14, Switzerland, Telephone: , Fax: Reprint requests: François Chappuis, Travel and Migration Medicine Unit, Department of Community Medicine, Geneva University Hospital, Rue Micheli-du-Crest 24, 1211 Geneva, Switzerland. REFERENCES 1. Magnus E, Vervoort T, van Meirvenne N, A cardagglutination test with stained trypanosomes (C.A.T.T.) for the serological diagnosis of T. b. gambiense trypanosomiasis. Ann Soc Belg Med Trop 58: Truc P, Lejon V, Magnus E, Jamonneau V, Nangouma A, Verloo D, Penchenier L, Büscher P, Evaluation of the micro- CATT, CATT/Trypanosoma brucei gambiense, and LATEX/ T. b. gambiense methods for serodiagnosis and surveillance of human African trypanosomiasis in west and central Africa. Bull World Health Organ 80: Jamonneau V, Truc P, Garcia A, Magnus E, Büscher P, Preliminary evaluation of latex/t. b. gambiense and alternative versions of CATT/T. b. gambiense for the serodiagnosis of human African trypanosomiasis of a population at risk in Côte d Ivoire: considerations for mass-screening. Acta Trop 76: van Meirvenne N, Biological diagnosis of human African trypanosomiasis. Dumas M, Bouteille B, Buguet A, eds. Progress in Human African Trypanosomiasis, Sleeping Sickness. Paris: Springer Verlag, World Health Organization, Control and surveillance of African trypanosomiasis. Report of a WHO Expert Committee. World Health Organ Tech Report Series 881: Ancelle T, Paugam A, Bourlioux F, Merad A, Vigier JP, Détection des trypanosomes dans le sang par la technique du quantitative buffy coat (QBC): évaluation expérimentale. Med Trop (Mars) 57: Simarro PP, Ruiz JA, Franco JR, Josenando T, Attitude towards CATT-positive individuals without confirmation in the African trypanosomiasis (T.b.gambiense) focus of Quiçama (Angola). Trop Med Int Health 4: Garcia A, Jamonneau V, Magnus E, Laveissière C, Lejon V, Nguessan P, Ndri L, Vanmeirvenne N, Büscher P, Follow-up of card agglutination trypanosomiasis test (CATT) positive but apparently aparasitaemic individuals in Côte d Ivoire: evidence for a complex and heterogeneous population. Trop Med Int Health 5: Chappuis F, Pittet A, Bovier PA, Adams K, Godineau V, Hwang SY, Magnus E, Büscher P, Field Evaluation of the CATT/Trypanosoma brucei gambiense on blood-impregnated filter papers for diagnosis of human African trypanosomiasis in southern Sudan. Trop Med Int Health 11: Doua F, Miezan TW, Sanon Singaro JR, Boa Yapo F, Baltz T, The efficacy of pentamidine in the treatment of early-late stage Trypanosoma brucei gambiense trypanosomiasis. Am J Trop Med Hyg 55: Lejon V, Legros D, Savignoni A, Etchegorry MG, Mbulamberi D, Büscher P, Neuro-inflammatory risk factors for treatment failure in early second stage sleeping sickness patients treated with pentamidine. J Neuroimmunol 144:
F. Chappuis 1, A. Pittet 1, P. A. Bovier 2, K. Adams 1, V. Godineau 1, S. Y. Hwang 1, E. Magnus 3 and P. Büscher 3
 Tropical Medicine and International Health volume 7 no 11 pp 942 948 november 2002 Field evaluation of the CATT/Trypanosoma brucei gambiense on blood-impregnated filter papers for diagnosis of human African
Tropical Medicine and International Health volume 7 no 11 pp 942 948 november 2002 Field evaluation of the CATT/Trypanosoma brucei gambiense on blood-impregnated filter papers for diagnosis of human African
Authors Chappuis, F; Udayraj, N; Stietenroth, K; Meussen, A; Bovier, P A. Published by: Infectious Diseases Society of America
 MSF Field Research Eflornithine is Safer Than Melarsoprol for the Treatment of Second-Stage Trypanosoma Brucei Gambiense Human African Trypanosomiasis. Authors Chappuis, F; Udayraj, N; Stietenroth, K;
MSF Field Research Eflornithine is Safer Than Melarsoprol for the Treatment of Second-Stage Trypanosoma Brucei Gambiense Human African Trypanosomiasis. Authors Chappuis, F; Udayraj, N; Stietenroth, K;
Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta State, Nigeria
 JMBR: A Peer-review Journal of Biomedical Sciences December 2006 Vol. 5 No.2 pp-28-32 Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta
JMBR: A Peer-review Journal of Biomedical Sciences December 2006 Vol. 5 No.2 pp-28-32 Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta
Evaluation of an EDTA version of CATT/Trypanosoma brucei gambiense. Prince Leopold Institute of Tropical Medicine, Department of Parasitology,
 Evaluation of an EDTA version of CATT/Trypanosoma brucei gambiense for serological screening of human blood samples Eddy Magnus 1 *, Veerle Lejon 1, Dominique Bayon 2, Dirk Buyse 3, Pere Simarro 4, Didier
Evaluation of an EDTA version of CATT/Trypanosoma brucei gambiense for serological screening of human blood samples Eddy Magnus 1 *, Veerle Lejon 1, Dominique Bayon 2, Dirk Buyse 3, Pere Simarro 4, Didier
BMC Infectious Diseases
 BMC Infectious Diseases BioMed Central Research article Estimates of the duration of the early and late stage of gambiense sleeping sickness Francesco Checchi* 1, João AN Filipe 2, Daniel T Haydon 3, Daniel
BMC Infectious Diseases BioMed Central Research article Estimates of the duration of the early and late stage of gambiense sleeping sickness Francesco Checchi* 1, João AN Filipe 2, Daniel T Haydon 3, Daniel
TRYPANOSOMA BRUCEI GAMBIENSE TRYPANOSOMIASIS IN TEREGO COUNTY, NORTHERN UGANDA, 1996: A LOT QUALITY ASSURANCE SAMPLING SURVEY
 Am. J. Trop. Med. Hyg., 70(4), 2004, pp. 390 394 Copyright 2004 by The American Society of Tropical Medicine and Hygiene TRYPANOSOMA BRUCEI GAMBIENSE TRYPANOSOMIASIS IN TEREGO COUNTY, NORTHERN UGANDA,
Am. J. Trop. Med. Hyg., 70(4), 2004, pp. 390 394 Copyright 2004 by The American Society of Tropical Medicine and Hygiene TRYPANOSOMA BRUCEI GAMBIENSE TRYPANOSOMIASIS IN TEREGO COUNTY, NORTHERN UGANDA,
Cost-effectiveness of Algorithms for Confirmation Test of Human African Trypanosomiasis
 Cost-effectiveness of Algorithms for Confirmation Test of Human African Trypanosomiasis Pascal Lutumba,* Filip Meheus, Jo Robays, Constantin Miaka,* Victor Kande,* Philippe Büscher, Bruno Dujardin, and
Cost-effectiveness of Algorithms for Confirmation Test of Human African Trypanosomiasis Pascal Lutumba,* Filip Meheus, Jo Robays, Constantin Miaka,* Victor Kande,* Philippe Büscher, Bruno Dujardin, and
Confirmation of the use of Latex IgM on cerebrospinal fluid for improving stage determination of Human African Trypanosomiasis
 African Journal of Biotechnology Vol. 4 (6), pp. 517-521, June 2005 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2005 Academic Journals Full Length Research Paper Confirmation
African Journal of Biotechnology Vol. 4 (6), pp. 517-521, June 2005 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2005 Academic Journals Full Length Research Paper Confirmation
V. Jamonneau 1, P. Solano 1, A. Garcia 2, V. Lejon 3, N. Djé 4, T. W. Miezan 4, P. N Guessan 1, G. Cuny 5 and P. Büscher 3
 Tropical Medicine and International Health volume 8 no 7 pp 589 594 july 2003 Stage determination and therapeutic decision in human African trypanosomiasis: value of polymerase chain reaction and immunoglobulin
Tropical Medicine and International Health volume 8 no 7 pp 589 594 july 2003 Stage determination and therapeutic decision in human African trypanosomiasis: value of polymerase chain reaction and immunoglobulin
IgM quantification in the cerebrospinal fluid of sleeping sickness patients by a latex card agglutination test
 Tropical Medicine and International Health volume 7 no 8 pp 685 692 august 2002 IgM quantification in the cerebrospinal fluid of sleeping sickness patients by a latex card agglutination test V. Lejon 1,2,
Tropical Medicine and International Health volume 7 no 8 pp 685 692 august 2002 IgM quantification in the cerebrospinal fluid of sleeping sickness patients by a latex card agglutination test V. Lejon 1,2,
Articles. Copyright Büscher et al. Open Access article distributed under the terms of CC BY-NC-ND.
 Sensitivity and specificity of HAT Sero-K-SeT, a rapid diagnostic test for serodiagnosis of sleeping sickness caused by Trypanosoma brucei gambiense: a case-control study Philippe Büscher, Pascal Mertens,
Sensitivity and specificity of HAT Sero-K-SeT, a rapid diagnostic test for serodiagnosis of sleeping sickness caused by Trypanosoma brucei gambiense: a case-control study Philippe Büscher, Pascal Mertens,
Received 19 February 2007/Returned for modification 2 April 2007/Accepted 4 April 2007
 CLINICAL AND VACCINE IMMUNOLOGY, June 2007, p. 732 737 Vol. 14, No. 6 1556-6811/07/$08.00 0 doi:10.1128/cvi.00103-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Treatment Failure
CLINICAL AND VACCINE IMMUNOLOGY, June 2007, p. 732 737 Vol. 14, No. 6 1556-6811/07/$08.00 0 doi:10.1128/cvi.00103-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Treatment Failure
HAT. Platform EDITORIAL CONTENT P. 2 P. 2 P. 3 P. 3 P. 4 P. 4 P. 5 P. 5 P. 6 P. 6 TRAINING WORKSHOP FOR MEMBERS OF THE STEERING COMMITTEE MEETING
 HAT Platform EDITORIAL Dr Augustin Kadima Ebeja HAT Platform Coordinator We are proud to present our design for a bilingual logo that will help underscore our efforts to conduct clinical trials against
HAT Platform EDITORIAL Dr Augustin Kadima Ebeja HAT Platform Coordinator We are proud to present our design for a bilingual logo that will help underscore our efforts to conduct clinical trials against
P PDF [Page: 1 of 8]
![P PDF [Page: 1 of 8] P PDF [Page: 1 of 8]](/thumbs/94/122070429.jpg) P152603.PDF [Page: 1 of 8] P152603.PDF [Page: 2 of 8] P152603.PDF [Page: 3 of 8] Am. J. Trop. Med. Hyg., 52(3), 1995, pp. 281-286 Copyright C 1995 by The American Society of Tropical Medicine and Hygie-ne
P152603.PDF [Page: 1 of 8] P152603.PDF [Page: 2 of 8] P152603.PDF [Page: 3 of 8] Am. J. Trop. Med. Hyg., 52(3), 1995, pp. 281-286 Copyright C 1995 by The American Society of Tropical Medicine and Hygie-ne
Options for Field Diagnosis of Human African Trypanosomiasis
 CLINICAL MICROBIOLOGY REVIEWS, Jan. 2005, p. 133 146 Vol. 18, No. 1 0893-8512/05/$08.00 0 doi:10.1128/cmr.18.1.133 146.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Options
CLINICAL MICROBIOLOGY REVIEWS, Jan. 2005, p. 133 146 Vol. 18, No. 1 0893-8512/05/$08.00 0 doi:10.1128/cmr.18.1.133 146.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Options
WHO/CDS/NTD/IDM/ Recommendations of the Informal Consultation on Issues for Clinical Product Development for Human African Trypanosomiasis
 WHO/CDS/NTD/IDM/2007.1 Recommendations of the Informal Consultation on Issues for Clinical Product Development for Human African Trypanosomiasis Geneva, Switzerland 9 10 September 2004 World Health Organization
WHO/CDS/NTD/IDM/2007.1 Recommendations of the Informal Consultation on Issues for Clinical Product Development for Human African Trypanosomiasis Geneva, Switzerland 9 10 September 2004 World Health Organization
Performance of Parasitological and Molecular Techniques for the Diagnosis and Surveillance of Gambiense Sleeping Sickness
 Performance of Parasitological and Molecular Techniques for the Diagnosis and Surveillance of Gambiense Sleeping Sickness Dieudonné Mumba Ngoyi 1,2, Rosine Ali Ekangu 1, Marie France Mumvemba Kodi 1, Patient
Performance of Parasitological and Molecular Techniques for the Diagnosis and Surveillance of Gambiense Sleeping Sickness Dieudonné Mumba Ngoyi 1,2, Rosine Ali Ekangu 1, Marie France Mumvemba Kodi 1, Patient
M E M O R A N D U M. No new drug is in clinical development for second stage HAT treatment.
 M E M O R A N D U M From: Director, NTD To: Secretary, Expert Committee on the Selection and Use of Essential Medicines Our ref: EML 2009 Attention: Dr Suzanne Hill Date: 13 February 2009 Your ref: E19/81/17
M E M O R A N D U M From: Director, NTD To: Secretary, Expert Committee on the Selection and Use of Essential Medicines Our ref: EML 2009 Attention: Dr Suzanne Hill Date: 13 February 2009 Your ref: E19/81/17
How to Shorten Patient Follow-Up after Treatment for Trypanosoma brucei gambiense Sleeping Sickness
 MAJOR ARTICLE How to Shorten Patient Follow-Up after Treatment for Trypanosoma brucei gambiense Sleeping Sickness Dieudonné Mumba Ngoyi, 1,4 Veerle Lejon, 4 Pati Pyana, 1,4 Marleen Boelaert, 5 Médard Ilunga,
MAJOR ARTICLE How to Shorten Patient Follow-Up after Treatment for Trypanosoma brucei gambiense Sleeping Sickness Dieudonné Mumba Ngoyi, 1,4 Veerle Lejon, 4 Pati Pyana, 1,4 Marleen Boelaert, 5 Médard Ilunga,
Confirmation of antibodies against L-tryptophan-like epitope in human African trypanosomosis serological diagnostic
 Vol. 15(36), pp. 1986-1990, 7 September, 2016 DOI: 10.5897/AJB2016.15575 Article Number: 273864A60390 ISSN 1684-5315 Copyright 2016 Author(s) retain the copyright of this article http://www.academicjournals.org/ajb
Vol. 15(36), pp. 1986-1990, 7 September, 2016 DOI: 10.5897/AJB2016.15575 Article Number: 273864A60390 ISSN 1684-5315 Copyright 2016 Author(s) retain the copyright of this article http://www.academicjournals.org/ajb
The development of new diagnostic tools for sleeping sickness
 The development of new diagnostic tools for sleeping sickness P. Büscher Institute of Tropical Medicine, Antwerp WHO CC for Research and Training on Human African Trypanosomiasis Diagnosis 1 1 Distribution
The development of new diagnostic tools for sleeping sickness P. Büscher Institute of Tropical Medicine, Antwerp WHO CC for Research and Training on Human African Trypanosomiasis Diagnosis 1 1 Distribution
TDR/DDR/98.1 WHO, GENEVA, 14 JULY 1998 CONCLUSIONS AND RECOMMENDATIONS RELAPSING PATIENTS. The 14-day regimen is highly effective.
 TDR/DDR/98.1 REPORT ON A MEETING OF THE PRODUCT DEVELOPMENT TEAM FOR AFRICAN TRYPANOSOMIASIS CHEMOTHERAPY TO REVIEW THE COMPARATIVE STUDY OF 14-DAY VERSUS 7-DAY TREATMENT OF LATE STAGE T.B. GAMBIENSE AFRICAN
TDR/DDR/98.1 REPORT ON A MEETING OF THE PRODUCT DEVELOPMENT TEAM FOR AFRICAN TRYPANOSOMIASIS CHEMOTHERAPY TO REVIEW THE COMPARATIVE STUDY OF 14-DAY VERSUS 7-DAY TREATMENT OF LATE STAGE T.B. GAMBIENSE AFRICAN
Authors Chappuis, François; Alirol, Emilie; Worku, Dagemlidet T; Mueller, Yolanda; Ritmeijer, Koert
 MSF Field Research High mortality among older patients treated with pentavalent antimonials for visceral leishmaniasis in East Africa and rationale for switch to liposomal amphotericin B Item type Article
MSF Field Research High mortality among older patients treated with pentavalent antimonials for visceral leishmaniasis in East Africa and rationale for switch to liposomal amphotericin B Item type Article
The effectiveness of active population screening and treatment for sleeping sickness control in the Democratic Republic of Congo
 Tropical Medicine and International Health volume 9 no 5 pp 542 550 may 2004 The effectiveness of active population screening and treatment for sleeping sickness control in the Democratic Republic of Congo
Tropical Medicine and International Health volume 9 no 5 pp 542 550 may 2004 The effectiveness of active population screening and treatment for sleeping sickness control in the Democratic Republic of Congo
Trypanosomiasis. By Ahmed Faris Alila Ahmed Laith Al-Nuaimi Ahmed Mohammed Al-juboory Ahmed Naaif Talib Ahmed Nadhem Al-Obeidy Osama Ahmed Al-Obeidy
 Trypanosomiasis By Ahmed Faris Alila Ahmed Laith Al-Nuaimi Ahmed Mohammed Al-juboory Ahmed Naaif Talib Ahmed Nadhem Al-Obeidy Osama Ahmed Al-Obeidy Ahmed Faris Alila Trypanosomiasis Kingdom: Protisata
Trypanosomiasis By Ahmed Faris Alila Ahmed Laith Al-Nuaimi Ahmed Mohammed Al-juboory Ahmed Naaif Talib Ahmed Nadhem Al-Obeidy Osama Ahmed Al-Obeidy Ahmed Faris Alila Trypanosomiasis Kingdom: Protisata
HAT DNDi program progress and evolution in the path to elimination. Dr Antoine Tarral. HAT Platform, Kampala October 2018
 HAT DNDi program progress and evolution in the path to elimination Dr Antoine Tarral HAT Platform, Kampala October 2018 DNDi s Mission To develop new drugs or new formulations of existing drugs for people
HAT DNDi program progress and evolution in the path to elimination Dr Antoine Tarral HAT Platform, Kampala October 2018 DNDi s Mission To develop new drugs or new formulations of existing drugs for people
WRAIR- GEIS 'Operational Clinical Infectious Disease' Course
 Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course The opinions or assertions contained herein are the private views of the author, and are not to be construed as official, or
Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course The opinions or assertions contained herein are the private views of the author, and are not to be construed as official, or
Reviewer No. 1 checklist for application of: inclusion of Nifurtimox + eflornithine in the WHO Essential Medicines List
 Reviewer No. 1 checklist for application of: inclusion of Nifurtimox + eflornithine in the WHO Essential Medicines List (1) Have all important studies that you are aware of been included? No additional
Reviewer No. 1 checklist for application of: inclusion of Nifurtimox + eflornithine in the WHO Essential Medicines List (1) Have all important studies that you are aware of been included? No additional
Kinetoplastids Handout
 Kinetoplastids Handout 1 Kinetoplastids widespread group of flagellated protozoa parasitize virtually all animal groups as well as plants and insects 3 distinct kinetoplastid species cause human disease
Kinetoplastids Handout 1 Kinetoplastids widespread group of flagellated protozoa parasitize virtually all animal groups as well as plants and insects 3 distinct kinetoplastid species cause human disease
Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course
 Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Disclaimer The views expressed in this presentation are those of the speaker and do not reflect the official policy
Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Disclaimer The views expressed in this presentation are those of the speaker and do not reflect the official policy
NECT Is Next: Implementing the New Drug Combination Therapy for Trypanosoma brucei gambiense Sleeping Sickness
 Viewpoints NECT Is Next: Implementing the New Drug Combination Therapy for Trypanosoma brucei gambiense Sleeping Sickness Oliver Yun 1 *, Gerardo Priotto 2, Jacqueline Tong 3, Laurence Flevaud 4, François
Viewpoints NECT Is Next: Implementing the New Drug Combination Therapy for Trypanosoma brucei gambiense Sleeping Sickness Oliver Yun 1 *, Gerardo Priotto 2, Jacqueline Tong 3, Laurence Flevaud 4, François
Evaluation of a rapid screening test for sleeping sickness. Lumbala C, Bisser S, Nguertoum E, Flevaud L, Jacquet D, Büscher P, Biéler S, Ndung u JM
 Evaluation of a rapid screening test for sleeping sickness Lumbala C, Bisser S, Nguertoum E, Flevaud L, Jacquet D, Büscher P, Biéler S, Ndung u JM 1 Performance evaluation of prototype in the field Prototype
Evaluation of a rapid screening test for sleeping sickness Lumbala C, Bisser S, Nguertoum E, Flevaud L, Jacquet D, Büscher P, Biéler S, Ndung u JM 1 Performance evaluation of prototype in the field Prototype
Mapping the capacities of fixed health facilities to cover people at risk of gambiense human African trypanosomiasis. Simarro et al.
 INTERNATIONAL JOURNAL OF HEALTH GEOGRAPHICS Mapping the capacities of fixed health facilities to cover people at risk of gambiense human African trypanosomiasis Simarro et al. Simarro et al. International
INTERNATIONAL JOURNAL OF HEALTH GEOGRAPHICS Mapping the capacities of fixed health facilities to cover people at risk of gambiense human African trypanosomiasis Simarro et al. Simarro et al. International
Authors Priotto, G; Fogg, C; Balasegaram, M; Erphas, O; Louga, A; Checchi, F; Ghabri, S; Piola, P. Archived with thanks to PLoS clinical trials
 MSF Field Research Three drug combinations for late-stage Trypanosoma brucei gambiense sleeping sickness: a randomized clinical trial in Uganda. Authors Priotto, G; Fogg, C; Balasegaram, M; Erphas, O;
MSF Field Research Three drug combinations for late-stage Trypanosoma brucei gambiense sleeping sickness: a randomized clinical trial in Uganda. Authors Priotto, G; Fogg, C; Balasegaram, M; Erphas, O;
Revisiting the Immune Trypanolysis Test to Optimise Epidemiological Surveillance and Control of Sleeping Sickness in West Africa
 Revisiting the Immune Trypanolysis Test to Optimise Epidemiological Surveillance and Control of Sleeping Sickness in West Africa Vincent Jamonneau 1,2 *, Bruno Bucheton 1,2, Jacques Kaboré 2, Hamidou Ilboudo
Revisiting the Immune Trypanolysis Test to Optimise Epidemiological Surveillance and Control of Sleeping Sickness in West Africa Vincent Jamonneau 1,2 *, Bruno Bucheton 1,2, Jacques Kaboré 2, Hamidou Ilboudo
Human African trypanosomiasis
 Human African trypanosomiasis Reto Brun, Johannes Blum, Francois Chappuis, Christian Burri Lancet 2010; 375: 148 59 Published Online October 14, 2009 DOI:10.1016/S0140-6736(09)60829-1 See Editorial page
Human African trypanosomiasis Reto Brun, Johannes Blum, Francois Chappuis, Christian Burri Lancet 2010; 375: 148 59 Published Online October 14, 2009 DOI:10.1016/S0140-6736(09)60829-1 See Editorial page
Introduction. Research
 Evaluation of the micro-catt, CATT/Trypanosoma brucei gambiense, and LATEX/T. b. gambiense methods for serodiagnosis and surveillance of human African trypanosomiasis in West and Central Africa Philippe
Evaluation of the micro-catt, CATT/Trypanosoma brucei gambiense, and LATEX/T. b. gambiense methods for serodiagnosis and surveillance of human African trypanosomiasis in West and Central Africa Philippe
RESEARCH. Safety and effectiveness of first line eflornithine for Trypanosoma brucei gambiense sleeping sickness in Sudan: cohort study
 1 Epicentre, 75011 Paris, France 2 Médecins Sans Frontières, Paris, France Correspondence to: G Priotto gpriotto@epicentre.msf.org doi:10.1136/bmj.39485.592674.be RESEARCH Safety and effectiveness of first
1 Epicentre, 75011 Paris, France 2 Médecins Sans Frontières, Paris, France Correspondence to: G Priotto gpriotto@epicentre.msf.org doi:10.1136/bmj.39485.592674.be RESEARCH Safety and effectiveness of first
ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS
 Molecular Diagnostics Symposium 2014 Zurich, 27 February 2014 ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS Stijn Deborggraeve Parasite Diagnostics Unit, Institute of Tropical Medicine
Molecular Diagnostics Symposium 2014 Zurich, 27 February 2014 ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS Stijn Deborggraeve Parasite Diagnostics Unit, Institute of Tropical Medicine
APPLICATION FOR REVISION OF ANTITRYPANOSOMAL MEDICINES IN WHO MODEL LIST OF ESSENTIAL MEDICINES
 APPLICATION FOR REVISION OF ANTITRYPANOSOMAL MEDICINES IN WHO MODEL LIST OF ESSENTIAL MEDICINES The objective of this application is to assure consistency among the antitrypanosomal medicines in the WHO
APPLICATION FOR REVISION OF ANTITRYPANOSOMAL MEDICINES IN WHO MODEL LIST OF ESSENTIAL MEDICINES The objective of this application is to assure consistency among the antitrypanosomal medicines in the WHO
Nucleic Acid Sequence-Based Amplification with Oligochromatography for Detection of Trypanosoma brucei in Clinical Samples
 JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2009, p. 630 635 Vol. 47, No. 3 0095-1137/09/$08.00 0 doi:10.1128/jcm.01430-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Nucleic Acid
JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2009, p. 630 635 Vol. 47, No. 3 0095-1137/09/$08.00 0 doi:10.1128/jcm.01430-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Nucleic Acid
اعداد رغداحمد رغد جمال الدين
 اعداد رغداحمد رغد جمال الدين Trypanosoma Causes Trypanosomiasis West African Trypanosomiasis T.brucei gambiense Sleeping sickness East African Trypanosomiasis T.brucei rhodesiense American Trypanosomiasis
اعداد رغداحمد رغد جمال الدين Trypanosoma Causes Trypanosomiasis West African Trypanosomiasis T.brucei gambiense Sleeping sickness East African Trypanosomiasis T.brucei rhodesiense American Trypanosomiasis
Human African trypanosomiasis: quantitative and qualitative assessment of intrathecal immune response
 European Journal of Neurology 2003, 10: 1 9 Human African trypanosomiasis: quantitative and qualitative assessment of intrathecal immune response V. Lejon a, C. J. M. Sindic b, M.-P. Van Antwerpen b, F.
European Journal of Neurology 2003, 10: 1 9 Human African trypanosomiasis: quantitative and qualitative assessment of intrathecal immune response V. Lejon a, C. J. M. Sindic b, M.-P. Van Antwerpen b, F.
J. Jannin, P. Simarro, J.R. Franco Neglected Tropical Diseases
 J. Jannin, P. Simarro, J.R. Franco Neglected Tropical Diseases Decision of fighting HAT IOM Public Workshop on Causes and Consequences of Neglected Tropical Diseases -Washington DC, USA. 21-22 September
J. Jannin, P. Simarro, J.R. Franco Neglected Tropical Diseases Decision of fighting HAT IOM Public Workshop on Causes and Consequences of Neglected Tropical Diseases -Washington DC, USA. 21-22 September
Intrathecal Immune Response Pattern for Improved Diagnosis of Central Nervous System Involvement in Trypanosomiasis
 MAJOR ARTICLE Intrathecal Immune Response Pattern for Improved Diagnosis of Central Nervous System Involvement in Trypanosomiasis Veerle Lejon, 1 Hansotto Reiber, 3 Dominique Legros, 4 Norbert Djé, 5 Eddy
MAJOR ARTICLE Intrathecal Immune Response Pattern for Improved Diagnosis of Central Nervous System Involvement in Trypanosomiasis Veerle Lejon, 1 Hansotto Reiber, 3 Dominique Legros, 4 Norbert Djé, 5 Eddy
/j x
 REVIEW 10.1111/j.1469-0691.2011.03536.x Sleeping sickness D. Malvy 1,2 and F. Chappuis 3,4 1) Travel Clinics and Division of Tropical Medicine and Imported Diseases, Department of Internal Medicine and
REVIEW 10.1111/j.1469-0691.2011.03536.x Sleeping sickness D. Malvy 1,2 and F. Chappuis 3,4 1) Travel Clinics and Division of Tropical Medicine and Imported Diseases, Department of Internal Medicine and
Report of the second WHO stakeholders meeting on gambiense human African trypanosomiasis elimination. Geneva, March 2016
 Report of the second WHO stakeholders meeting on gambiense human African trypanosomiasis elimination Geneva, 21 23 March 2016 WHO Library Cataloguing-in-Publication Data: Report of the second WHO stakeholders
Report of the second WHO stakeholders meeting on gambiense human African trypanosomiasis elimination Geneva, 21 23 March 2016 WHO Library Cataloguing-in-Publication Data: Report of the second WHO stakeholders
KINETOPLASTIDS. Kinetoplast. Nucleus
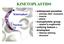 KINETOPLASTIDS Kinetoplast Nucleus widespread parasites animals (fish humans) insects plants monophyletic group related to euglenoids unifying feature = kinetoplast Giemsa staining structure KINETOPLAST
KINETOPLASTIDS Kinetoplast Nucleus widespread parasites animals (fish humans) insects plants monophyletic group related to euglenoids unifying feature = kinetoplast Giemsa staining structure KINETOPLAST
Challenges in Diagnosing Human African Trypanosomiasis
 Challenges in Diagnosing Human African Trypanosomiasis Evaluation of the MSF OCG project in Dingila, DRC by Simon Van Nieuwenhove September 2015, public version by Paul Grohma Medical anthropologist Study
Challenges in Diagnosing Human African Trypanosomiasis Evaluation of the MSF OCG project in Dingila, DRC by Simon Van Nieuwenhove September 2015, public version by Paul Grohma Medical anthropologist Study
Challenges of controlling sleeping sickness in areas of violent conflict: experience in the Democratic Republic of Congo
 Conflict and Health This Provisional PDF corresponds to the article as it appeared upon acceptance. Fully formatted PDF and full text (HTML) versions will be made available soon. Challenges of controlling
Conflict and Health This Provisional PDF corresponds to the article as it appeared upon acceptance. Fully formatted PDF and full text (HTML) versions will be made available soon. Challenges of controlling
Diagnostic Accuracy of Loopamp Trypanosoma brucei Detection Kit for Diagnosis of Human African Trypanosomiasis in Clinical Samples
 Diagnostic Accuracy of Loopamp Trypanosoma brucei Detection Kit for Diagnosis of Human African Trypanosomiasis in Clinical Samples Patrick Mitashi 1,2,3, Epco Hasker 1, Dieudonné Mumba Ngoyi 2,3, Pati
Diagnostic Accuracy of Loopamp Trypanosoma brucei Detection Kit for Diagnosis of Human African Trypanosomiasis in Clinical Samples Patrick Mitashi 1,2,3, Epco Hasker 1, Dieudonné Mumba Ngoyi 2,3, Pati
Immunoglobulin and haematological profile of Nigerians with Trypanosoma brucei gambiense infection
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Archives of Applied Science Research, 2010, 2 (5):177-183 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-508X
Available online at www.scholarsresearchlibrary.com Scholars Research Library Archives of Applied Science Research, 2010, 2 (5):177-183 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-508X
Characterization of Trypanosoma brucei gambiense variant surface glycoprotein LiTat 1.5
 Short Communication Characterization of Trypanosoma brucei gambiense variant surface glycoprotein LiTat 1.5 L. Van Nieuwenhove 1 *, S. Rogé 1 *, V. Lejon 1, Y. Guisez 2 and P. Büscher 1 1 Department of
Short Communication Characterization of Trypanosoma brucei gambiense variant surface glycoprotein LiTat 1.5 L. Van Nieuwenhove 1 *, S. Rogé 1 *, V. Lejon 1, Y. Guisez 2 and P. Büscher 1 1 Department of
African trypanosomiasis or Sleeping sickness. A bio-mathematical study of the disease
 African trypanosomiasis or Sleeping sickness A bio-mathematical study of the disease YASMINE SAMIA 008 African trypanosomiasis or Sleeping sickness A bio-mathematical study of the disease African trypanosomiasis,
African trypanosomiasis or Sleeping sickness A bio-mathematical study of the disease YASMINE SAMIA 008 African trypanosomiasis or Sleeping sickness A bio-mathematical study of the disease African trypanosomiasis,
Downloaded from:
 Palmer, JJ; Surur, EI; Checchi, F; Ahmad, F; Ackom, FK; Whitty, CJ (2014) A mixed methods study of a health worker training intervention to increase syndromic referral for gambiense human african trypanosomiasis
Palmer, JJ; Surur, EI; Checchi, F; Ahmad, F; Ackom, FK; Whitty, CJ (2014) A mixed methods study of a health worker training intervention to increase syndromic referral for gambiense human african trypanosomiasis
Detection of trypanosome-specific antibodies in saliva, towards non-invasive serological diagnosis of sleeping sickness
 Tropical Medicine and International Health doi:10.1111/j.1365-3156.2006.01620.x volume 11 no 5 pp 620 627 may 2006 Detection of trypanosome-specific antibodies in saliva, towards non-invasive serological
Tropical Medicine and International Health doi:10.1111/j.1365-3156.2006.01620.x volume 11 no 5 pp 620 627 may 2006 Detection of trypanosome-specific antibodies in saliva, towards non-invasive serological
PARASITOLOGY CASE HISTORY #4 (BLOOD PARASITES) (Lynne S. Garcia)
 PARASITOLOGY CASE HISTORY #4 (BLOOD PARASITES) (Lynne S. Garcia) A 52-year old male presented after complaining of a week of fever and headaches. Other symptoms included malaise and lack of appetite, and
PARASITOLOGY CASE HISTORY #4 (BLOOD PARASITES) (Lynne S. Garcia) A 52-year old male presented after complaining of a week of fever and headaches. Other symptoms included malaise and lack of appetite, and
Untreated Human Infections by Trypanosoma brucei gambiense Are Not 100% Fatal
 Untreated Human Infections by Trypanosoma brucei gambiense Are Not 100% Fatal Vincent Jamonneau 1,2 *, Hamidou Ilboudo 2, Jacques Kaboré 2, Dramane Kaba 3, Mathurin Koffi 4, Philippe Solano 1,2, André
Untreated Human Infections by Trypanosoma brucei gambiense Are Not 100% Fatal Vincent Jamonneau 1,2 *, Hamidou Ilboudo 2, Jacques Kaboré 2, Dramane Kaba 3, Mathurin Koffi 4, Philippe Solano 1,2, André
PLoS Negl Trop Dis 2010;4(12):e783. PLoS Neglected Tropical Diseases. Archived with thanks to PLoS Neglected Tropical Diseases
 MSF Field Research The Unknown Risk of Vertical Transmission in Sleeping Sickness--a Literature Review Item type Authors Citation DOI Journal Rights Article Lindner, A K; Priotto, G PLoS Negl Trop Dis
MSF Field Research The Unknown Risk of Vertical Transmission in Sleeping Sickness--a Literature Review Item type Authors Citation DOI Journal Rights Article Lindner, A K; Priotto, G PLoS Negl Trop Dis
Stage determination in sleeping sickness: comparison of two cell counting and two parasite detection techniques
 Tropical Medicine and International Health doi:1.1111/tmi.11 volume 1 no pp 77 7 june 13 Stage determination in sleeping sickness: comparison of two cell counting and two parasite detection techniques
Tropical Medicine and International Health doi:1.1111/tmi.11 volume 1 no pp 77 7 june 13 Stage determination in sleeping sickness: comparison of two cell counting and two parasite detection techniques
Reviewer 2: NIFURTIMOX+EFLORNITHINE(N+E)
 Reviewer 2: NIFURTIMOX+EFLORNITHINE(N+E) 1. The multicenter clinical trial of N+E combination therapy for second-stage sleeping sickness presented on behalf of G. Priotto and the NECT Study Team offers
Reviewer 2: NIFURTIMOX+EFLORNITHINE(N+E) 1. The multicenter clinical trial of N+E combination therapy for second-stage sleeping sickness presented on behalf of G. Priotto and the NECT Study Team offers
Accepted 3 December 2003
 African Journal of Biotechnology Vol. 3 (1), pp. 94-98, January 2004 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2004 Academic Journals Full Length Research Paper Characterization
African Journal of Biotechnology Vol. 3 (1), pp. 94-98, January 2004 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2004 Academic Journals Full Length Research Paper Characterization
Media Briefing Notes UN Palais press corps, Geneva. Mortality Projections for Darfur 15 October 2004
 Media Briefing Notes UN Palais press corps, Geneva Mortality Projections for Darfur 15 October 24 Presented by David Nabarro Representative of the World Health Organization Director-General Health Action
Media Briefing Notes UN Palais press corps, Geneva Mortality Projections for Darfur 15 October 24 Presented by David Nabarro Representative of the World Health Organization Director-General Health Action
Molecular analysis of archived blood slides reveals an atypical human Trypanosoma. infection
 Molecular analysis of archived blood slides reveals an atypical human Trypanosoma infection Running head: Molecular analysis of Trypanosoma infection Stijn Deborggraeve a,f,*, Mathurin Koffi b, Vincent
Molecular analysis of archived blood slides reveals an atypical human Trypanosoma infection Running head: Molecular analysis of Trypanosoma infection Stijn Deborggraeve a,f,*, Mathurin Koffi b, Vincent
Trypanosomiasis Research Centre, Kenya Agricultural Research Institute, P.O. Box , Kikuyu, Kenya
 JCM Accepts, published online ahead of print on 9 February 2011 J. Clin. Microbiol. doi:10.1128/jcm.01817-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All
JCM Accepts, published online ahead of print on 9 February 2011 J. Clin. Microbiol. doi:10.1128/jcm.01817-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All
Clinical Infectious Diseases Advance Access published October 16, Nifurtimox Eflornithine Combination Therapy (NECT) for second stage gambiense
 Clinical Infectious Diseases Advance Access published October 16, 2012 1 Nifurtimox Eflornithine Combination Therapy (NECT for second stage gambiense human African trypanosomiasis: MSF experience in the
Clinical Infectious Diseases Advance Access published October 16, 2012 1 Nifurtimox Eflornithine Combination Therapy (NECT for second stage gambiense human African trypanosomiasis: MSF experience in the
Trypanosomiasis. Introduction. Epidemiology. Global Epidemiology. Trypanosomiasis Risk in UK Travellers
 Trypanosomiasis Introduction Epidemiology Risk for travellers Transmission Signs and symptoms Treatment Prevention References Reading list Links Introduction Trypanosomiasis is caused by parasitic protozoa
Trypanosomiasis Introduction Epidemiology Risk for travellers Transmission Signs and symptoms Treatment Prevention References Reading list Links Introduction Trypanosomiasis is caused by parasitic protozoa
African Trypanosomiasis Detection Using Dempster-Shafer Theory Andino Maseleno, 2 Md. Mahmud Hasan
 African Trypanosomiasis Detection Using Dempster-Shafer Theory 1 Andino Maseleno, 2 Md. Mahmud Hasan 1 Department of Computer Science, Faculty of Science, Universiti Brunei Darussalam 2 Jalan Tungku Link,
African Trypanosomiasis Detection Using Dempster-Shafer Theory 1 Andino Maseleno, 2 Md. Mahmud Hasan 1 Department of Computer Science, Faculty of Science, Universiti Brunei Darussalam 2 Jalan Tungku Link,
Optimizing Population Screening for Infectious Diseases
 Optimizing Population Screening for Infectious Diseases Harwin de Vries MSc., Prof. Albert Wagelmans, Prof. Joris van de Klundert June 7 th, 2017 De Vries, Wagelmans, Van de Klundert Optimizing Population
Optimizing Population Screening for Infectious Diseases Harwin de Vries MSc., Prof. Albert Wagelmans, Prof. Joris van de Klundert June 7 th, 2017 De Vries, Wagelmans, Van de Klundert Optimizing Population
Antitrypanosomal Activity of Fexinidazole, a New Oral Nitroimidazole Drug Candidate for Treatment of Sleeping Sickness
 ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Dec. 2011, p. 5602 5608 Vol. 55, No. 12 0066-4804/11/$12.00 doi:10.1128/aac.00246-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Antitrypanosomal
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Dec. 2011, p. 5602 5608 Vol. 55, No. 12 0066-4804/11/$12.00 doi:10.1128/aac.00246-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Antitrypanosomal
Clinical Development of New Treatments for Sleeping Sickness. Sleeping sickness: New oral treatments
 Clinical Development of New Treatments for Sleeping Sickness Supporting WHO HAT elimination goals WHO set goals for Global Elimination of sleeping sickness by 2020, supported by London Declaration (2012)
Clinical Development of New Treatments for Sleeping Sickness Supporting WHO HAT elimination goals WHO set goals for Global Elimination of sleeping sickness by 2020, supported by London Declaration (2012)
Laboratory diagnosis of Blood and tissue flagellates
 Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
HAEMOFLAGELLATES. Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine
 HAEMOFLAGELLATES Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of Leishmania spp. and Trypanosoma
HAEMOFLAGELLATES Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of Leishmania spp. and Trypanosoma
New biomarkers for stage determination in Trypanosoma brucei rhodesiense sleeping sickness patients
 Tiberti et al. Clinical and Translational Medicine 2013, 2:1 RESEARCH New biomarkers for stage determination in Trypanosoma brucei rhodesiense sleeping sickness patients Natalia Tiberti 1, Enock Matovu
Tiberti et al. Clinical and Translational Medicine 2013, 2:1 RESEARCH New biomarkers for stage determination in Trypanosoma brucei rhodesiense sleeping sickness patients Natalia Tiberti 1, Enock Matovu
Are 10-year booster doses of yellow fever vaccine necessary? Peter Teitelbaum, MD Committee to Advise on Tropical Medicine and Travel (CATMAT)
 Are 10-year booster doses of yellow fever vaccine necessary? Peter Teitelbaum, MD Committee to Advise on Tropical Medicine and Travel (CATMAT) Disclosure of Potential for Conflict of Interest Peter Teitelbaum,
Are 10-year booster doses of yellow fever vaccine necessary? Peter Teitelbaum, MD Committee to Advise on Tropical Medicine and Travel (CATMAT) Disclosure of Potential for Conflict of Interest Peter Teitelbaum,
Scaling Up Routine Viral Load in Resource Limited Settings. MSF experience in Zimbabwe
 Scaling Up Routine Viral Load in Resource Limited Settings MSF experience in Zimbabwe Outline 1. Why do we need VL 2. What are the barriers to roll out 3. MSF experience 4. Summary samumsf.org 1. Why do
Scaling Up Routine Viral Load in Resource Limited Settings MSF experience in Zimbabwe Outline 1. Why do we need VL 2. What are the barriers to roll out 3. MSF experience 4. Summary samumsf.org 1. Why do
Cholera in South Sudan
 Republic of South Sudan Cholera in South Sudan Situation Report # 16 as at 23:59 Hours, 31 May 2014 Background On 29 April 2014, a suspected case of cholera was reported from the MSF clinic at UN House/Juba
Republic of South Sudan Cholera in South Sudan Situation Report # 16 as at 23:59 Hours, 31 May 2014 Background On 29 April 2014, a suspected case of cholera was reported from the MSF clinic at UN House/Juba
Visceral leishmaniasis and HIV in Tigray, Ethiopia
 Tropical Medicine and International Health volume 8 no 8 pp 733 739 august 2003 Visceral leishmaniasis and HIV in Tigray, Ethiopia Suzi Lyons 1, Hans Veeken 2 and Jean Long 3 1 Department of Public Health
Tropical Medicine and International Health volume 8 no 8 pp 733 739 august 2003 Visceral leishmaniasis and HIV in Tigray, Ethiopia Suzi Lyons 1, Hans Veeken 2 and Jean Long 3 1 Department of Public Health
Luminescent Multiplex Viability Assay for T.b. gambiense
 Luminescent Multiplex Viability Assay for T.b. gambiense Van Reet N., Pyana P., Rogé S., Claes F. and Büscher, P. Institute of Tropical Medicine Antwerp 32nd ISCTRC conference 8th 12th September 2013 Introduction
Luminescent Multiplex Viability Assay for T.b. gambiense Van Reet N., Pyana P., Rogé S., Claes F. and Büscher, P. Institute of Tropical Medicine Antwerp 32nd ISCTRC conference 8th 12th September 2013 Introduction
STRATEGY FOR NEGLECTED TROPICAL DISEASES
 STRATEGY FOR NEGLECTED TROPICAL DISEASES 2015-2020 1 ABOUT FIND FIND was established in 2003 to bridge existing development gaps for essential diagnostics by initiating and coordinating research and development
STRATEGY FOR NEGLECTED TROPICAL DISEASES 2015-2020 1 ABOUT FIND FIND was established in 2003 to bridge existing development gaps for essential diagnostics by initiating and coordinating research and development
EBOLA SITUATION REPORT
 EBOLA SITUATION REPORT 11 FEBRUARY 2015 CASES/ DEATHS (data up to 8 February 2015) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 1 0 1 0
EBOLA SITUATION REPORT 11 FEBRUARY 2015 CASES/ DEATHS (data up to 8 February 2015) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 1 0 1 0
Meningitis Outbreak Response intervention thresholds in sub-saharan Africa
 Meningitis Outbreak Response intervention thresholds in sub-saharan Africa Report for the WHO Meningitis Guideline Revision May 2014 Prepared by Dr Caroline Trotter (clt56@cam.ac.uk) Recommendation question:
Meningitis Outbreak Response intervention thresholds in sub-saharan Africa Report for the WHO Meningitis Guideline Revision May 2014 Prepared by Dr Caroline Trotter (clt56@cam.ac.uk) Recommendation question:
The elimination of Trypanosoma brucei gambiense sleeping sickness in the focus of Luba, Bioko Island, Equatorial Guinea
 Tropical Medicine and International Health doi:10.1111/j.1365-3156.2006.01624.x volume 11 no 5 pp 636 646 may 2006 The elimination of Trypanosoma brucei gambiense sleeping sickness in the focus of Luba,
Tropical Medicine and International Health doi:10.1111/j.1365-3156.2006.01624.x volume 11 no 5 pp 636 646 may 2006 The elimination of Trypanosoma brucei gambiense sleeping sickness in the focus of Luba,
Clinic of Ruminant, Faculty of Veterinary Medicine, Chiang Mai University,
 เช ยงใหม ส ตวแพทยสาร 2549;4(2):101-106. Original article COMPARATIVE STUDY ON PREVALENCE OF TRYPANOSOMOSIS IN DAIRY CATTLE IN CHIANG MAI AND LAM PHUN PROVINCES DIAGNOSED BY HEMATOCRIT CENTRIFUGATION TECHNIQUE
เช ยงใหม ส ตวแพทยสาร 2549;4(2):101-106. Original article COMPARATIVE STUDY ON PREVALENCE OF TRYPANOSOMOSIS IN DAIRY CATTLE IN CHIANG MAI AND LAM PHUN PROVINCES DIAGNOSED BY HEMATOCRIT CENTRIFUGATION TECHNIQUE
Ten Years of Tryps and Tsetses. Lucille Blumberg & John Frean National Institute for Communicable Diseases
 Ten Years of Tryps and Tsetses Lucille Blumberg & John Frean National Institute for Communicable Diseases Source: Simarro et al. PLoS Negl Trop Dis 2011; 5: e1007 Country of acquisition Number of cases
Ten Years of Tryps and Tsetses Lucille Blumberg & John Frean National Institute for Communicable Diseases Source: Simarro et al. PLoS Negl Trop Dis 2011; 5: e1007 Country of acquisition Number of cases
Hypocretin and Human African Trypanosomiasis
 Narcolepsy Hypocretin and Human African Trypanosomiasis Yves Dauvilliers, MD, PhD 1 ; Sylvie Bisser, MD, PhD 2 ; Florian Chapotot, PhD 3 ; Gedeao Vatunga, MD 4 ; Raymond Cespuglio, PhD 5 ; Téofilo Josenando,
Narcolepsy Hypocretin and Human African Trypanosomiasis Yves Dauvilliers, MD, PhD 1 ; Sylvie Bisser, MD, PhD 2 ; Florian Chapotot, PhD 3 ; Gedeao Vatunga, MD 4 ; Raymond Cespuglio, PhD 5 ; Téofilo Josenando,
Choosing the right study design
 Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
World Health Organization Department of Communicable Disease Surveillance and Response
 WHO Report on Global Surveillance of Epidemic-prone Infectious Diseases World Health Organization Department of Communicable Disease Surveillance and Response This document has been downloaded from the
WHO Report on Global Surveillance of Epidemic-prone Infectious Diseases World Health Organization Department of Communicable Disease Surveillance and Response This document has been downloaded from the
Blood Smears Only 3 February Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 3 February 2015 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 3 February 2015 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only
Control and surveillance of human African trypanosomiasis
 In the 1960s, it appeared that human African trypanosomiasis (HAT) could be effectively controlled, but by the beginning of the twenty-first century several decades of neglect had led to alarming numbers
In the 1960s, it appeared that human African trypanosomiasis (HAT) could be effectively controlled, but by the beginning of the twenty-first century several decades of neglect had led to alarming numbers
DOWNLOAD OR READ : WEST AFRICAN HYGIENE PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : WEST AFRICAN HYGIENE PDF EBOOK EPUB MOBI Page 1 Page 2 west african hygiene west african hygiene pdf west african hygiene West African Ebola virus epidemic Simplified Ebola virus epidemic
DOWNLOAD OR READ : WEST AFRICAN HYGIENE PDF EBOOK EPUB MOBI Page 1 Page 2 west african hygiene west african hygiene pdf west african hygiene West African Ebola virus epidemic Simplified Ebola virus epidemic
Authors Alberti, K; Guthmann, J P; Fermon, F; Nargaye, K D; Grais, R. Transactions of the Royal Society of Tropical Medicine and Hygiene
 MSF Field Research Use of Lot Quality Assurance Sampling (LQAS) to estimate vaccination coverage helps guide future vaccination efforts. Authors Alberti, K; Guthmann, J P; Fermon, F; Nargaye, K D; Grais,
MSF Field Research Use of Lot Quality Assurance Sampling (LQAS) to estimate vaccination coverage helps guide future vaccination efforts. Authors Alberti, K; Guthmann, J P; Fermon, F; Nargaye, K D; Grais,
Human African Trypanosomiasis
 Human African Trypanosomiasis Facing the challenges caused by neglect: The need for new treatment and diagnostics Alix Feuton 2 Preface This document provides an overview of the current issues surrounding
Human African Trypanosomiasis Facing the challenges caused by neglect: The need for new treatment and diagnostics Alix Feuton 2 Preface This document provides an overview of the current issues surrounding
THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA
 THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA Imna Malele, Hamisi Nyingilili, Eugen Lyaruu, Winston Kitwika Tsetse & Trypanosomiasis Research Institute Majani Mapana, Off Korogwe
THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA Imna Malele, Hamisi Nyingilili, Eugen Lyaruu, Winston Kitwika Tsetse & Trypanosomiasis Research Institute Majani Mapana, Off Korogwe
FINDs programme on diagnostics for NTDs
 FINDs programme on diagnostics for NTDs JM Ndung u, A. Picado, I Cruz & S Biéler Presentation at IRD, Montpellier, France, 12March 2018 FIND who we are & what we do A not-for-profit international organization
FINDs programme on diagnostics for NTDs JM Ndung u, A. Picado, I Cruz & S Biéler Presentation at IRD, Montpellier, France, 12March 2018 FIND who we are & what we do A not-for-profit international organization
Diphtheria Outbreak Response Cox s Bazar Bangladesh
 Diphtheria Outbreak Response Cox s Bazar Bangladesh 1 Movement of population from Rakhine (Myanmar) to Cox s Bazar (Bangladesh) Cox s Bazar district Bangladesh Rakhine Province Myanmar Began on 25 Aug
Diphtheria Outbreak Response Cox s Bazar Bangladesh 1 Movement of population from Rakhine (Myanmar) to Cox s Bazar (Bangladesh) Cox s Bazar district Bangladesh Rakhine Province Myanmar Began on 25 Aug
Diagnostic Accuracy of Two rk39 Antigen-Based Dipsticks and the Formol Gel Test for Rapid Diagnosis of Visceral Leishmaniasis in Northeastern Uganda
 JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2005, p. 5973 5977 Vol. 43, No. 12 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.12.5973 5977.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2005, p. 5973 5977 Vol. 43, No. 12 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.12.5973 5977.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
Protozoa from tissues. Leishmania spp. Naegleria fowleri Toxoplasma gondii Trichomonas vaginalis Trypanosoma spp.
 Protozoa from tissues Leishmania spp. Naegleria fowleri Toxoplasma gondii Trichomonas vaginalis Trypanosoma spp. Leishmaniasis Leishmania infantum, Leishmania donovani, in macrophages of man. Female sandflies:
Protozoa from tissues Leishmania spp. Naegleria fowleri Toxoplasma gondii Trichomonas vaginalis Trypanosoma spp. Leishmaniasis Leishmania infantum, Leishmania donovani, in macrophages of man. Female sandflies:
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
EBOLA VIRUS DISEASE. Democratic Republic of the Congo. External Situation Report 19. Date of information: 01 June
 EBOLA VIRUS DISEASE Democratic Republic of the Congo External Situation Report 19 Date of information: 01 June 2017 1. Situation update WHO, UN Agencies, international organizations, non-governmental organizations
EBOLA VIRUS DISEASE Democratic Republic of the Congo External Situation Report 19 Date of information: 01 June 2017 1. Situation update WHO, UN Agencies, international organizations, non-governmental organizations
