Lobular Carcinoma In Situ Variants in Breast Cores
|
|
|
- Arnold Pitts
- 6 years ago
- Views:
Transcription
1 Lobular Carcinoma In Situ Variants in Breast Cores Potential for Misdiagnosis, Upgrade Rates at Surgical Excision, and Practical Implications Megan E. Sullivan, MD; Seema A. Khan, MD; Yurdanur Sullu, MD; Carol Schiller, MD; Barbara Susnik, MD, PhD N Context. Differentiating ductal carcinoma in situ (DCIS) from lobular carcinoma in situ (LCIS) on core biopsy has important clinical implications. Lobular carcinoma in situ variants, including LCIS with necrosis and pleomorphic LCIS, share morphologic features with solid DCIS that may lead to misclassification. Objectives. (1) To review all LCIS variants diagnosed in core biopsies at Northwestern University, Feinberg School of Medicine, and determine the frequency of misinterpretation of variant LCIS as solid DCIS in archival core biopsies, and (2) to determine the frequency of upgrade to invasive carcinoma or DCIS in the surgical excision. Design. Consecutive core biopsies with original diagnoses of predominantly solid DCIS without invasion that were performed between January 2001 and December 2005 at Northwestern University, Feinberg School of Medicine, were selected for E-cadherin staining. The revised diagnosis of LCIS was based on E-cadherin negativity and morphology. The frequency of LCIS variants A diagnosis of ductal carcinoma in situ (DCIS) versus lobular carcinoma in situ (LCIS) has important clinical implications for the patient because therapeutic priorities are different for each entity. Historically, LCIS is characterized microscopically by a population of discohesive, small, monotonous cells with intracytoplasmic lumina that fill and expand the lobule. 1 The distinction from DCIS was relatively clear. However, recently described variants of LCIS, including LCIS with necrosis (LCIS-N) and pleomorphic LCIS (LCIS-P), share some morphologic features with solid DCIS, thus rendering precise diagnosis difficult on routine hematoxylin-eosin stained sections in some core biopsies. 2,3 The E-cadherin Accepted for publication August 26, From the Departments of Pathology (Drs Sullivan, Sullu, Schiller, and Susnik) and Surgery (Dr Khan), Northwestern University, Feinberg School of Medicine, Chicago, Illinois. Dr Sullu is now with the Department of Pathology, Ondokuz Mayis University, Faculty of Medicine, Samsun, Turkey. The authors have no relevant financial interest in the products or companies described in this article. Presented in part at the annual meeting of the United States and Canadian Academy of Pathology, Boston, Massachusetts, March 10, Reprints: Megan E. Sullivan, MD, Departments of Pathology, Northwestern Memorial Hospital, Feinberg Pavilion, 7th Floor, 251 E Huron St, Chicago, IL ( megan-sullivan@northwestern. edu). upgraded was then estimated from all core biopsies with original or revised diagnoses of pleomorphic LCIS or LCIS with necrosis. Results. Among 75 cases of solid DCIS, 10 (13.3%) were reclassified as LCIS, including 9 variants (5 pleomorphic LCIS, 4 LCIS with necrosis) and 1 classic LCIS. Twenty-eight patients comprised the entire group of LCIS variant cases (both reclassified and originally diagnosed cases). Seven patients with LCIS variants (25%) were upgraded to invasive lobular carcinoma in surgical excision (4 of 11 cases of LCIS with necrosis [36%] versus 3 of 17 cases of pleomorphic LCIS [18%]). Conclusions. About one-tenth of solid DCIS diagnosed in core biopsies in the past may represent LCIS variants. These show a 25% upgrade to invasive lobular carcinoma in surgical excision. The distinction of an LCIS variant from DCIS is important because of its implications for radiation therapy, although it may not affect surgical management. (Arch Pathol Lab Med. 2010;134: ) immunohistochemical stain is increasingly being used to identify lobular neoplasia and has been shown to be helpful in classifying carcinomas in situ with indeterminate features as either ductal or lobular. 4 E-cadherin is a transmembrane protein that mediates cell-to-cell adhesion as well as several signal transduction pathways. Loss of membranous E- cadherin expression is characteristic of lobular neoplasia, whereas most ductal neoplastic proliferations, as well as benign breast epithelium, maintain expression. 5 7 In addition, E-cadherin reactivity in carcinoma in situ was shown to modulate the pathobiologic potential of these lesions as a subset of LCIS with focal membranous reactivity exhibited clinical behavior similar to DCIS. 8 When LCIS-P and LCIS-N were initially described as distinct clinicopathologic entities in the literature, E- cadherin immunohistochemistry was not routinely applied for cases with overlapping morphology, and some of these lesions were diagnosed as DCIS. Therefore, we decided to identify LCIS cases that were originally interpreted and treated as DCIS and establish their frequency relative to solid DCIS. Current understanding of ductal and lobular carcinoma in situ is based on studies that included lesions that were classified solely on morphologic criteria, 9,10 with LCIS consisting entirely of lesions meeting criteria for the classic form of LCIS. Although DCIS is a known precursor to invasive 1024 Arch Pathol Lab Med Vol 134, July 2010 LCIS Variants in Breast Cores Sullivan et al
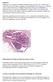
2 and LCIS-N) is less understood, but there is growing evidence that these lesions have a significant association with invasive lobular carcinoma, suggesting their potential for direct progression into invasive carcinoma.2,3,12,13 Noting this association, we decided the second aim of this study should be to determine the frequency of upgrade to lesions that require more extensive treatment (ie, DCIS or invasive malignancy) when LCIS variants are diagnosed on core biopsy. MATERIALS AND METHODS Review of Archival Core Biopsies With Original Diagnosis of Solid DCIS Figure 1. A, E-cadherin negative lobular carcinoma in situ (LCIS) with adjacent unremarkable lobule showing diffuse membranous positivity. B, Pleomorphic LCIS. C, Lobular carcinoma in situ with necrosis (E-cadherin, original magnification 310 [A]; and hematoxylineosin, original magnifications 340 [B] and 310 [C]). Following Institutional Review Board approval, consecutive needle-core biopsies of the breast with an original diagnosis of DCIS with solid architecture (including both solid-type DCIS with and without necrosis and comedo-type DCIS) without an associated invasive carcinoma performed between January 2001 and January 2005 at Northwestern University, Feinberg School of Medicine, were retrieved from the pathology files. On review, examples of DCIS with significant micropapillary or cribriform architecture and cases with insufficient diagnostic tissue remaining for immunohistochemical staining were excluded, and the remaining specimens were stained by E-cadherin. Immunohistochemical staining was performed using a dextran polymer conjugate visualization system and a primary mouse anti-human E-cadherin antibody (clone NCH-38, 1:200 dilution; DAKO, Carpinteria, California). The E-cadherin immunostaining was interpreted as positive when there was diffuse, weak, moderate, or strong membranous staining of the carcinoma in situ cells. Staining was interpreted as negative when no membranous staining of the carcinoma in situ cells was present. Adjacent benign ducts and lobules showing strong complete membranous staining served as internal and external positive controls (Figure 1, A). The diagnosis of LCIS on review was based on E-cadherin negativity as well as morphologic criteria. A proliferation was classified as LCIS-C when composed of a uniform population of small, discohesive cells within a variably distended lobule that possessed round to oval, bland nuclei and intracytoplasmic vacuoles. Although other definitions exist, the threshold used in this study for the distinction of LCIS versus atypical lobular hyperplasia was involvement of at least 75% of the units of a lobule.1 The diagnosis of LCIS-P required a variably discohesive population of pleomorphic, medium to large cells, with nuclei at least 3 times the size of a small lymphocyte that were eccentrically placed with distinct nucleoli.2 Necrosis and microcalcifications were frequently present, but not obligatory, for the diagnosis of LCIS-P. In LCIS-N, cells with classic lobular cytology displayed luminal necrosis and often marked distension of ductules.1 The necrosis was defined as either (1) a punctate type, when a collection of nuclear fragments was present focally within the LCIS; or (2) a comedo type, when prominent plugs of necrotic material were present in the lumen of the glandular spaces distended by LCIS. Although morphologic overlap existed between the LCIS variants because of the presence of necrosis in most LCIS-P cases, we distinguished LCIS-P from LCIS-N by the presence of a cytologically pleomorphic LCIS cell population that represented at least 10% of the LCIS cells in the former. Information regarding the subsequent surgical excision findings was pulled from the pathology database in all cases stained for E-cadherin, and the slides were reviewed in selected cases, including all cases with a modified diagnosis on review. Upstaging of LCIS Variants Diagnosed in Core Biopsy carcinoma, classic LCIS (LCIS-C) is considered a marker of increased risk for development of invasive breast carcinoma rather than a direct precursor lesion.9,11 The natural history of the recently described variants of LCIS (LCIS-P Arch Pathol Lab Med Vol 134, July 2010 Following Institutional Review Board approval, consecutive needle core biopsies of the breast with an original diagnosis of either LCIS-P or LCIS-N (confirmed by negative E-cadherin immunohistochemistry) without associated invasive carcinoma LCIS Variants in Breast Cores Sullivan et al 1025
3 Figure 2. Screening mammogram from patient 18 (see Table 1), a 43-year-old woman with indeterminate calcifications (Breast Imaging-Reporting and Data System 4B) biopsied using a 9-gauge needle with vacuum assistance and stereotactic guidance showing florid lobular carcinoma in situ with necrosis. Figure 3. Patient 18 had a 0.5-cm invasive lobular carcinoma in her needle-localized surgical excision with negative margins and a negative sentinel lymph node biopsy (hematoxylin-eosin, original magnification 310). performed between January 2001 and January 2009 at Northwestern University, Feinberg School of Medicine, were retrieved from the pathology records. We reviewed the pathology of the core biopsy specimens and the subsequent pathology of the surgical excision of the core biopsy site. An upgrade was defined as the finding of a lesion in the surgical specimen (adjacent to the core biopsy site) such as invasive carcinoma or DCIS that would warrant additional treatment, such as complete excision and radiation therapy. Statistical differences were assessed using the x2 test or t test (P values,.05 were considered statistically significant). RESULTS Review of Archival Core Biopsies With Original Diagnosis of Solid DCIS On review of 135 consecutive needle core biopsies of the breast with an original diagnosis of solid DCIS without an associated invasive carcinoma, those with significant micropapillary or cribriform architecture, and those with insufficient diagnostic tissue remaining for immunohistochemical staining were excluded (n 5 56; 41%). The remaining 79 core biopsy specimens were stained with E-cadherin. Four additional cases were dropped because no lesional tissue remained on the deeper level used for E-cadherin immunohistochemistry, leaving 75 core biopsies included in the study. Membranous E-cadherin positivity was present in 86.7% of cases (65 of 75) and E-cadherin was negative in 13.3% of cases (10 of 75) originally diagnosed as solid DCIS. On review of the hematoxylin-eosin stained sections and immunohistochemistry results, all E-cadherin negative cases were reclassified as LCIS, including 1 LCIS-C and 9 variants (5 LCIS-P and 4 LCIS-N), (Figure 1, B and C). The original grade of reclassified cases was 1 (1 case; 10%), 2 (7 cases; 70%), and 3 (2 cases; 20%). Eight of the 10 total reclassified cases (80%) showed necrosis, including all LCIS-N (4 of 4; 100%) and 4 LCIS-P (4 of 5; 80%). Seven of the 9 cases reclassified as LCIS variants (78%) had surgical follow-up at our institution and were included in the upgrade analysis Arch Pathol Lab Med Vol 134, July 2010 LCIS Variants Diagnosed in Core Biopsy: Clinicopathologic Features and Frequency of Upgrade in Surgical Specimens We identified 28 patients with a LCIS variant diagnosed by core biopsy and with surgical follow-up available, including 7 cases (25%) with an original diagnosis of solid DCIS revised to an LCIS variant (Table 1). Only LCIS variants without an invasive component or accompanying DCIS in the core biopsy were included. The entire group (n 5 28) consisted of 11 LCIS-N (39%) and 17 LCISP (61%). Patient ages ranged from 41 to 70 years (mean, 54.5 years). The radiologic imaging target was a mass in 3 cases (11%) and calcifications in the remaining 25 cases (89%) (Figure 2). Lesions were sampled by core biopsy with stereotactic (n 5 24; 86%), sonographic (n 5 2; 7%), or magnetic resonance imaging (n 5 2; 7%) guidance. Ten patients had a family history of either a mother or sister with breast carcinoma (10 of 28; 36%). Eight patients (8 of 28; 29%) had a personal history of ipsilateral breast carcinoma, including 3 individuals (3 of 28; 11%) with previous carcinoma and 5 patients (5 of 28; 18%) with synchronous carcinoma in another quadrant (Table 1). These ipsilateral cancers included DCIS (n 5 3) and invasive ductal (n 5 2), mammary (n 5 2) and lobular (n 5 1) carcinoma. Two patients had contralateral, but synchronous, invasive carcinoma of the ductal type (2 of 28; 7%). Overall, LCIS variants were upgraded to invasive carcinoma in 25% (7 of 28) of excisional specimens (Table 2). A comparable upgrade rate was seen in the Ecadherin positive DCIS core biopsies from our study set with available surgical excision pathology (16 of 60; 26.7%). All of the invasive cancers associated with LCIS variants were lobular type (Figure 3). There was a trend toward younger patients in the upgrade to invasive lobular carcinoma (mean, 49.7 years) as opposed to patients without an upgrade (mean, 56.1 years); however, that finding was not statistically significant (t test, P 5.09). The size of upgraded invasive lobular carcinomas in surgical specimen ranged from less than 0.1 to 1.5 cm LCIS Variants in Breast Cores Sullivan et al
4 Table 1. Case No. Age, y Characteristics of Patients With Lobular Carcinoma In Situ (LCIS) Variant Diagnosed by Core Biopsy and With Surgical Follow-up Available a,b Radiologic Findings History of Ipsilateral Cancer Original dx, Grade Reviewed dx Surgery Upgrade on Excision Radiation Therapy 1 69 Calcifications Synchronous IDC and ILC solid DCIS 3 LCIS-P M No No 2 57 Calcifications None solid DCIS 2 LCIS-N L, M ILC Yes 3 48 Calcifications Previous IMC (8 mo) solid DCIS 2 LCIS-P L No Yes (for IMC) 4 42 Calcifications None solid DCIS 3 LCIS-P L ILC Yes 5 53 Calcifications None solid DCIS 2 LCIS-N M ILC No 6* 70 Calcifications None solid DCIS 2 LCIS-N L No Yes 7 54 Calcifications Previous IMC (3 y) solid DCIS 2 LCIS-N M No No 8 61 Calcifications Synchronous IDC, DCIS LCIS-N LCIS-N M No No 9 59 Calcifications None LCIS-N LCIS-N L ILC Yes 10* 41 Calcifications None LCIS-P LCIS-P L No No Calcifications Synchronous DCIS LCIS-N LCIS-N L x2 DCIS Yes 12* 58 Mass None LCIS-P LCIS-P L No Yes 13* 45 Calcifications None LCIS-P LCIS-P L No No Calcifications Previous DCIS (6 y) LCIS-P LCIS-P L No No 15* 62 Calcifications None LCIS-P LCIS-P L No No 16* 46 Calcifications None LCIS-N LCIS-N L No Yes Calcifications None LCIS-P LCIS-P L ILC Yes Calcifications None LCIS-N LCIS-N L ILC Yes 19* 58 Calcifications None LCIS-P LCIS-P L No No Calcifications None LCIS-P LCIS-P L ILC Yes Calcifications None LCIS-P LCIS-P L DCIS Yes 22* 52 Calcifications None LCIS-P LCIS-P L No No 23* 52 Calcifications None LCIS-P LCIS-P L No No Mass None LCIS-P LCIS-P M No No 25* 64 Calcifications None LCIS-N LCIS-N L No No Calcifications Synchronous ILC LCIS-P LCIS-P M No No Calcifications Synchronous DCIS LCIS-N LCIS-N L No Yes Mass None LCIS-P LCIS-P M DCIS No Abbreviations: DCIS, ductal carcinoma in situ; dx, diagnosis; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; IMC, invasive mammary carcinoma; L, lumpectomy; LCIS-N, lobular carcinoma in situ with necrosis; LCIS-P, pleomorphic lobular carcinoma in situ; M, mastectomy. a Only cases with surgical follow-up are included in the table. b Cases with an asterisk (*) represent pure LCIS (patients with no personal history of invasive carcinoma and no upgrade in their excision). (mean, 0.4 cm). The upgraded invasive lobular carcinomas were staged as T1mic (n 5 4; 57%), T1a (n 5 2; 29%), and T1c (n 5 1; 14%). Two patients with large synchronous ipsilateral invasive carcinomas in other quadrants had positive lymph nodes. None of the 3 lesions presenting as a radiologic mass was upgraded and 6 of 25 lesions (24%) presenting with calcifications were upgraded. Even the largest upgraded invasive carcinoma (size, 1.5 cm) presented only with calcifications on the original mammogram. The calcifications were associated with the LCIS variant in 6 of the 7 upgrades (86%), and in all cases, the calcifications were present in luminal necrotic material. In the remaining patient with an upgrade at excision, the calcifications were present only in adjacent breast tissue. Upgrade to invasive carcinoma was more frequent in LCIS-N (4 of 11; 36%) than in LCIS-P (3 of 17; 18%); however, the difference was not statistically significant (x 2 test, P 5.26). A single case of LCIS-P showed no intraluminal necrosis, whereas all other LCIS variants with an upgrade showed necrosis (6 of 7; 86%). The presence of necrosis was not significantly associated with an upgrade (x 2 test, P 5.63). In 3 additional cases, DCIS was identified in the subsequent surgical specimen by hematoxylin-eosin morphology adjacent to the core biopsy site. Including all cases where a more significant lesion was present in the excisional specimen (7 invasive lobular carcinomas and 3 DCIS), the overall upgrade was 36% (10 of 28; Table 2). Overall, 61% (17 of 28) of LCIS variants were associated with malignancy in the ipsilateral breast, either DCIS or invasive carcinoma, at some point during the patient s clinical course. Focusing only on the associated invasive malignancy, 43% of LCIS variants (12 of 28) were associated with an invasive carcinoma in the same breast (Table 1). Eleven patients with LCIS variants diagnosed by core biopsy had no upgrade and no history of ipsilateral carcinoma and were representative of pure LCIS variants. One of these individuals (9%) was treated by mastectomy, and 10 patients (91%) were treated by conservative Table 2. Upgrade in Subsequent Surgical Excision of Lobular Carcinoma In Situ (LCIS) Variants Diagnosed in Core Biopsy Core Biopsy Diagnosis Upgrade to ILC in SSE, No. (%) Upgrade to DCIS in SSE, No. (%) Total Upgrade in SSE, No. (%) LCIS-N, n (36) 1 (9) 5 (45) LCIS-P, n (18) 2 (12) 5 (29) Total variant LCIS, n = 28 7 (25) 3 (11) 10 (36) Abbreviations: DCIS, ductal carcinoma in situ; ILC, invasive lobular carcinoma; LCIS-N, lobular carcinoma in situ with necrosis; LCIS-P, pleomorphic lobular carcinoma in situ; SSE, subsequent surgical excision. Arch Pathol Lab Med Vol 134, July 2010 LCIS Variants in Breast Cores Sullivan et al 1027
5 surgery, which included radiation treatment in 3 cases (Table 1). COMMENT In our retrospective study spanning 5 years, LCIS variants were more common than anticipated, representing 12% (9 of 75) of previously diagnosed solid DCIS cases. Most revised cases were LCIS variants that would require a complete surgical excision of the core biopsy site, similar to DCIS. Although LCIS-C diagnosed in core biopsy without discordant imaging is currently not considered a surgical disease, in the past, many were managed by surgical excision. The need for complete surgical excision of LCIS variants is supported by our results showing an upgrade in 25% of LCIS variants diagnosed in core biopsy and by the data previously presented by Chivukula et al, 13 who similarly reported a 25% upgrade in LCIS-P diagnosed in core biopsy. The upgrade rate seen in LCIS variants was also similar to that seen in the E-cadherin positive, solid DCIS core biopsies (26.7%) from our initial study group. The upgrades associated with LCIS variants in our study were limited to lesions presenting with microcalcifications, appeared to be associated with a younger age, and were more common in LCIS-N than in LCIS-P. The upgraded invasive cancers were all lobular types and were identified at an early stage, most commonly microinvasive. Based on our data, as well as other reports in the literature, 13 LCIS variants diagnosed on core biopsies are frequently associated with invasive carcinoma. This association may have 2 different implications. In our study, the invasive cancers were found in different quadrants, and their diverse histologic subtypes suggest that LCIS variants, similar to classic LCIS, are markers of risk for breast cancer. However, all the adjacent invasive carcinomas identified in our study as upgrades were of the lobular type, pointing to a possible precursor role of LCIS variants. The current treatment recommendations for management of patients with a diagnosis of breast carcinoma in situ on core biopsy vary depending on whether the diagnosis rendered is DCIS, LCIS-C, or an LCIS variant. A core biopsy diagnosis of DCIS requires complete surgical excision with negative margins and subsequent radiation therapy. At the opposite end of the spectrum, some studies show that pure LCIS-C may not require any excision when the pathologic and radiologic findings are concordant 14 and can be managed by observation and endocrine chemoprophylaxis. The recommendations for LCIS-P and LCIS-N are evolving, and there is no clear consensus, but complete surgical excision is currently recommended. 15 Although more research is needed to clarify the biologic potential of LCIS variants, given that their behavior is more aggressive and DCIS-like, achieving negative surgical margins seems like a reasonable goal. As LCIS variants are still not universally accepted entities in breast pathology, one approach frequently used is to designate these lesions as mixed carcinoma in situ with ductal and lobular features, a diagnosis that triggers the management required for DCIS. 4,15 However, unlike conventional DCIS, the clinical necessity of radiation therapy for variant LCIS lesions is not clear. Although the misinterpretation of LCIS variants as solid DCIS does not usually affect surgical management, it has important treatment implications with respect to radiation therapy because LCIS-C lesions are radiotherapy resistant. 16 However, despite the paucity of data on radiotherapy effect on LCIS variants and the lack of consensus recommendations, radiation therapy is often considered. The terminology of mixed carcinoma in situ with ductal and lobular features has been supporting this approach because it advocates the management required for DCIS. Although only prospective trials may be able to establish the value of radiation therapy for LCIS variants, at present, a correct pathologic classification of these lesions is the first step toward a better understanding of their behavior. Generous use of E-cadherin immunohistochemical stain may be appropriate on core biopsy specimens with solid carcinoma in situ to exclude LCIS variants unless definitive architectural and cytologic features of ductal differentiation are present. As better immunohistochemistry-derived data regarding LCIS variants emerge, more complete outcome data will become available to guide clinical recommendations. References 1. Rosen PP. Lobular carcinoma in situ and atypical lobular hyperplasia. In: Rosen s Breast Pathology. 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009: Sneige N, Wang J, Baker BA, Krishnamurthy S, Middleton LP. Clinical, histopathologic, and biologic features of pleomorphic lobular (ductal-lobular) carcinoma in situ of the breast: a report of 24 cases. Mod Pathol. 2002;15(10): Fadare O, Dadmanesh F, Alvarado-Cabrero I, et al. Lobular intraepithelial neoplasia [lobular carcinoma in situ] with comedo-type necrosis: a clinicopathologic study of 18 cases. Am J Surg Pathol. 2006;30(11): Jacobs TW, Pliss N, Kouria G, Schnitt SJ. Carcinomas in situ of the breast with indeterminate features: role of E-cadherin staining in categorization. Am J Surg Pathol. 2001;25(2): De Leeuw WJ, Berx G, Vos CB, et al. Simultaneous loss of E-cadherin and catenins in invasive lobular breast cancer and lobular carcinoma in situ. J Pathol. 1997;183(4): Moll R, Mitze M, Frixen UH, Birchmeier W. Differential loss of E-cadherin expression in infiltrating ductal and lobular breast carcinomas. Am J Pathol. 1993;143(6): Vos CB, Cleton-Jansen AM, Berx G, et al. E-cadherin inactivation in lobular carcinoma in situ of the breast: an early event in tumorigenesis. Br J Cancer. 1997;76(9): Goldstein NS, Kestin LL, Vicini FA. Clinicopathologic implications of E- cadherin reactivity in patients with lobular carcinoma in situ of the breast. Cancer. 2001;92(4): Rosen PP, Kosloff C, Lieberman PH, Adair F, Braun DW Jr. Lobular carcinoma in situ of the breast: detailed analysis of 99 patients with average follow-up of 24 years. Am J Surg Pathol. 1978;2(3): Sanders ME, Schuyler PA, Dupont WD, Page DL. The natural history of low-grade ductal carcinoma in situ of the breast in women treated by biopsy only revealed over 30 years of long-term follow-up. Cancer. 2005;103(12): Haagensen CD, Lane N, Lattes R, Bodian C. Lobular neoplasia (so-called lobular carcinoma in situ) of the breast. Cancer. 1978;42(2): Sapino A, Frigerio A, Peterse JL, Arisio R, Coluccia C, Bussolati G. Mammographically detected in situ lobular carcinomas of the breast. Virchows Arch. 2000;436(5): Chivukula M, Haynik DM, Brufsky A, Carter G, Dabbs DJ. Pleomorphic lobular carcinoma in situ (PLCIS) on breast core needle biopsies: clinical significance and immunoprofile. Am J Surg Pathol. 2008;32(11): Hwang H, Barke LD, Mendelson EB, Susnik B. Atypical lobular hyperplasia and classic lobular carcinoma in situ in core biopsy specimens: routine excision is not necessary. Mod Pathol. 2008;21(10): Jacobs TW, Connolly JL, Schnitt SJ. Nonmalignant lesions in breast core needle biopsies: to excise or not to excise? Am J Surg Pathol. 2002;26(9): Rosen PP. Pathologic effects of therapy. In: Rosen PP, ed. Rosen s Breast Pathology. 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009: Arch Pathol Lab Med Vol 134, July 2010 LCIS Variants in Breast Cores Sullivan et al
Original Articles. Clinical Implications of Margin Involvement by Pleomorphic Lobular Carcinoma In Situ
 Original Articles Clinical Implications of Margin Involvement by Pleomorphic Lobular Carcinoma In Situ Erinn Downs-Kelly, DO; Diana Bell, MD; George H. Perkins, MD; Nour Sneige, MD; Lavinia P. Middleton,
Original Articles Clinical Implications of Margin Involvement by Pleomorphic Lobular Carcinoma In Situ Erinn Downs-Kelly, DO; Diana Bell, MD; George H. Perkins, MD; Nour Sneige, MD; Lavinia P. Middleton,
Disclosures 5/27/2012. Outline of Talk. Outline of Talk. When Is LCIS Clinically Significant? Classic LCIS. Classic LCIS
 When Is LCIS Clinically Significant? Disclosures I have nothing to disclose Yunn-Yi Chen, MD, PhD Professor Outline of Talk Outline of Talk Classic LCIS Classic LCIS Definition of lobular differentiation
When Is LCIS Clinically Significant? Disclosures I have nothing to disclose Yunn-Yi Chen, MD, PhD Professor Outline of Talk Outline of Talk Classic LCIS Classic LCIS Definition of lobular differentiation
6/3/2010. Outline of Talk. Lobular Breast Cancer: Definition of lobular differentiation. Common Problems in Diagnosing LCIS in Core Biopsies
 Outline of Talk Lobular Breast Cancer: Common Problems in Diagnosing LCIS in Core Biopsies Definition of lobular differentiation Variants of LCIS that: carry risk for unsampled invasive cancer mimic DCIS
Outline of Talk Lobular Breast Cancer: Common Problems in Diagnosing LCIS in Core Biopsies Definition of lobular differentiation Variants of LCIS that: carry risk for unsampled invasive cancer mimic DCIS
LOBULAR CARCINOMA IN SITU: WHAT DOES IT MEAN? THE SURGEON'S PERSPECTIVE
 : WHAT DOES IT MEAN? THE SURGEON'S PERSPECTIVE Benjamin O. Anderson, M.D. Director, Breast Health Clinic Professor of Surgery and Global Health, University of Washington Joint Member, Fred Hutchinson Cancer
: WHAT DOES IT MEAN? THE SURGEON'S PERSPECTIVE Benjamin O. Anderson, M.D. Director, Breast Health Clinic Professor of Surgery and Global Health, University of Washington Joint Member, Fred Hutchinson Cancer
Surgical Pathology Issues of Practical Importance
 Surgical Pathology Issues of Practical Importance Anne Moore, MD Medical Oncology Syed Hoda, MD Surgical Pathology The pathologist is central to the team approach needed to manage the patient with breast
Surgical Pathology Issues of Practical Importance Anne Moore, MD Medical Oncology Syed Hoda, MD Surgical Pathology The pathologist is central to the team approach needed to manage the patient with breast
Epithelial Columnar Breast Lesions: Histopathology and Molecular Markers
 29th Annual International Conference Advances in the Application of Monoclonal Antibodies in Clinical Oncology and Symposium on Cancer Stem Cells 25 th -27t h June, 2012, Mykonos, Greece Epithelial Columnar
29th Annual International Conference Advances in the Application of Monoclonal Antibodies in Clinical Oncology and Symposium on Cancer Stem Cells 25 th -27t h June, 2012, Mykonos, Greece Epithelial Columnar
Atypical Ductal Hyperplasia and Papillomas: A Comparison of Ultrasound Guided Breast Biopsy and Stereotactic Guided Breast Biopsy
 Atypical Ductal Hyperplasia and Papillomas: A Comparison of Ultrasound Guided Breast Biopsy and Stereotactic Guided Breast Biopsy Breast Cancer is the most common cancer diagnosed in women in the United
Atypical Ductal Hyperplasia and Papillomas: A Comparison of Ultrasound Guided Breast Biopsy and Stereotactic Guided Breast Biopsy Breast Cancer is the most common cancer diagnosed in women in the United
Proliferative Breast Disease: implications of core biopsy diagnosis. Proliferative Breast Disease
 Proliferative Breast Disease: implications of core biopsy diagnosis Jean F. Simpson, M.D. Breast Pathology Consultants, Inc. Nashville, TN Proliferative Breast Disease Must be interpreted in clinical and
Proliferative Breast Disease: implications of core biopsy diagnosis Jean F. Simpson, M.D. Breast Pathology Consultants, Inc. Nashville, TN Proliferative Breast Disease Must be interpreted in clinical and
Columnar Cell Lesions
 Columnar Cell Lesions Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Question? Columnar cell lesions are: a) Annoying lesions
Columnar Cell Lesions Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Question? Columnar cell lesions are: a) Annoying lesions
Case study 1. Rie Horii, M.D., Ph.D. Division of Pathology Cancer Institute Hospital, Japanese Foundation for Cancer Research
 NCCN/JCCNB Seminar in Japan April 15, 2012 Case study 1 Rie Horii, M.D., Ph.D. Division of Pathology Cancer Institute Hospital, Japanese Foundation for Cancer Research Present illness: A 50y.o.premenopausal
NCCN/JCCNB Seminar in Japan April 15, 2012 Case study 1 Rie Horii, M.D., Ph.D. Division of Pathology Cancer Institute Hospital, Japanese Foundation for Cancer Research Present illness: A 50y.o.premenopausal
Anatomic Pathology / Mucocele-like Lesions on Breast Core Biopsy. Mucocele-like Lesions Diagnosed on Breast Core Biopsy
 Anatomic Pathology / Mucocele-like Lesions on Breast Core Biopsy Mucocele-like Lesions Diagnosed on Breast Core Biopsy Assessment of Upgrade Rate and Need for Surgical Excision Brian Sutton, MD, 1 Simone
Anatomic Pathology / Mucocele-like Lesions on Breast Core Biopsy Mucocele-like Lesions Diagnosed on Breast Core Biopsy Assessment of Upgrade Rate and Need for Surgical Excision Brian Sutton, MD, 1 Simone
Classic lobular neoplasia on core biopsy: a clinical and radio-pathologic correlation study with follow-up excision biopsy
 762 & 2013 USCAP, Inc All rights reserved 0893-3952/13 $32.00 Classic lobular neoplasia on core biopsy: a clinical and radio-pathologic correlation study with follow-up excision biopsy Shweta Chaudhary
762 & 2013 USCAP, Inc All rights reserved 0893-3952/13 $32.00 Classic lobular neoplasia on core biopsy: a clinical and radio-pathologic correlation study with follow-up excision biopsy Shweta Chaudhary
Interpretation of Breast Pathology in the Era of Minimally Invasive Procedures
 Shahla Masood, M.D. Professor and Chair Department of Pathology and Laboratory Medicine University of Florida College of Medicine Jacksonville Medical Director, UF Health Breast Center Chief of Pathology
Shahla Masood, M.D. Professor and Chair Department of Pathology and Laboratory Medicine University of Florida College of Medicine Jacksonville Medical Director, UF Health Breast Center Chief of Pathology
Flat Epithelial Atypia
 Flat Epithelial Atypia Richard Owings, M.D. University of Arkansas for Medical Sciences Department of Pathology Flat epithelial atypia can be a difficult lesion May be a subtle diagnosis Lots of changes
Flat Epithelial Atypia Richard Owings, M.D. University of Arkansas for Medical Sciences Department of Pathology Flat epithelial atypia can be a difficult lesion May be a subtle diagnosis Lots of changes
04/10/2018 HIGH RISK BREAST LESIONS. Pathology Perspectives of High Risk Breast Lesions ELEVATED RISK OF BREAST CANCER HISTORICAL PERSPECTIVES
 Pathology Perspectives of High Risk Breast Lesions Savitri Krishnamurthy MD Professor of Pathology Deputy Division Head Director of Clinical Trials, Research and Development The University of Texas MD
Pathology Perspectives of High Risk Breast Lesions Savitri Krishnamurthy MD Professor of Pathology Deputy Division Head Director of Clinical Trials, Research and Development The University of Texas MD
Good afternoon everyone. First of all many thanks to Dr. Bonaventura and Dr. Arn for inviting
 PATHOLOGY IN-SITU CARCINOMA, ROHIT BHARGAVA, MD 1 Good afternoon everyone. First of all many thanks to Dr. Bonaventura and Dr. Arn for inviting me here, it s great to be here and I m going to talk about
PATHOLOGY IN-SITU CARCINOMA, ROHIT BHARGAVA, MD 1 Good afternoon everyone. First of all many thanks to Dr. Bonaventura and Dr. Arn for inviting me here, it s great to be here and I m going to talk about
Image guided core biopsies:
 Recommendations on the Surgical, Radiologic and Pathologic Approaches to Breast Disease: Using best practices based on multidisciplinary methodologies developed through the Allina Breast Committee. Image
Recommendations on the Surgical, Radiologic and Pathologic Approaches to Breast Disease: Using best practices based on multidisciplinary methodologies developed through the Allina Breast Committee. Image
A712(18)- Test slide, Breast cancer tissues with corresponding normal tissues
 A712(18)- Test slide, Breast cancer tissues with corresponding normal tissues (formalin fixed) For research use only Specifications: No. of cases: 12 Tissue type: Breast cancer tissues with corresponding
A712(18)- Test slide, Breast cancer tissues with corresponding normal tissues (formalin fixed) For research use only Specifications: No. of cases: 12 Tissue type: Breast cancer tissues with corresponding
A712(19)- Test slide, Breast cancer tissues with corresponding normal tissues
 A712(19)- Test slide, Breast cancer tissues with corresponding normal tissues (formalin fixed) For research use only Specifications: No. of cases: 12 Tissue type: Breast cancer tissues with corresponding
A712(19)- Test slide, Breast cancer tissues with corresponding normal tissues (formalin fixed) For research use only Specifications: No. of cases: 12 Tissue type: Breast cancer tissues with corresponding
Original Report. Mucocele-Like Tumors of the Breast: Mammographic and Sonographic Appearances. Katrina Glazebrook 1 Carol Reynolds 2
 Katrina Glazebrook 1 Carol Reynolds 2 Received January 2, 2002; accepted after revision August 28, 2002. 1 Department of Radiology, Mayo Clinic, 200 First St. S.W., Rochester, MN 55905. Address correspondence
Katrina Glazebrook 1 Carol Reynolds 2 Received January 2, 2002; accepted after revision August 28, 2002. 1 Department of Radiology, Mayo Clinic, 200 First St. S.W., Rochester, MN 55905. Address correspondence
In situ lobular neoplasia of the breast with marked myoepithelial proliferation
 In situ lobular neoplasia of the breast with marked myoepithelial proliferation Sami Shousha To cite this version: Sami Shousha. In situ lobular neoplasia of the breast with marked myoepithelial proliferation.
In situ lobular neoplasia of the breast with marked myoepithelial proliferation Sami Shousha To cite this version: Sami Shousha. In situ lobular neoplasia of the breast with marked myoepithelial proliferation.
CLINICAL SIGNIFICANCE OF BENIGN EPITHELIAL CHANGES
 Papillomas. Papillomas are composed of multiple branching fibrovascular cores, each having a connective tissue axis lined by luminal and myoepithelial cells ( Fig. 23-11 ). Growth occurs within a dilated
Papillomas. Papillomas are composed of multiple branching fibrovascular cores, each having a connective tissue axis lined by luminal and myoepithelial cells ( Fig. 23-11 ). Growth occurs within a dilated
Proliferative Epithelial lesions of the Breast. Sami Shousha, MD, FRCPath Charing Cross Hospital & Imperial College, London
 Proliferative Epithelial lesions of the Breast Sami Shousha, MD, FRCPath Charing Cross Hospital & Imperial College, London Amman, November2013 Proliferative Epithelial Lesions of the Breast Usual type
Proliferative Epithelial lesions of the Breast Sami Shousha, MD, FRCPath Charing Cross Hospital & Imperial College, London Amman, November2013 Proliferative Epithelial Lesions of the Breast Usual type
Pathology of Lobular & Ductal Preneoplasia. Syed A Hoda, MD Weill-Cornell, New York, NY
 Pathology of Lobular & Ductal Preneoplasia Syed A Hoda, MD Weill-Cornell, New York, NY Proliferative Epithelial Changes in Breast A wide range of proliferative epithelial changes occur in the breast There
Pathology of Lobular & Ductal Preneoplasia Syed A Hoda, MD Weill-Cornell, New York, NY Proliferative Epithelial Changes in Breast A wide range of proliferative epithelial changes occur in the breast There
Promise of a beautiful day
 Promise of a beautiful day Ductal carcinoma in Situ Lobular Carcinoma in Situ Natural History Manosmed Tartous Oct 2009 Gérard ABADJIAN MD Pathology Department Hôtel-Dieu de France. Associate Professor
Promise of a beautiful day Ductal carcinoma in Situ Lobular Carcinoma in Situ Natural History Manosmed Tartous Oct 2009 Gérard ABADJIAN MD Pathology Department Hôtel-Dieu de France. Associate Professor
Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance
 & 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance Charles C Guo 1 and
& 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance Charles C Guo 1 and
Multiple Fibroadenomas Harboring Carcinoma in Situ in a Woman with a Familty History of Breast/ Ovarian Cancer
 Multiple Fibroadenomas Harboring Carcinoma in Situ in a Woman with a Familty History of Breast/ Ovarian Cancer A Kuijper SS Preisler-Adams FD Rahusen JJP Gille E van der Wall PJ van Diest J Clin Pathol
Multiple Fibroadenomas Harboring Carcinoma in Situ in a Woman with a Familty History of Breast/ Ovarian Cancer A Kuijper SS Preisler-Adams FD Rahusen JJP Gille E van der Wall PJ van Diest J Clin Pathol
Breast Cancer. Most common cancer among women in the US. 2nd leading cause of death in women. Mortality rates though have declined
 Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women Mortality rates though have declined 1 in 8 women will develop breast cancer Breast Cancer Breast cancer increases
Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women Mortality rates though have declined 1 in 8 women will develop breast cancer Breast Cancer Breast cancer increases
3/27/2017. Disclosure of Relevant Financial Relationships. Papilloma???
 Management of Papillary Lesions Diagnosed at Rad Path Concordant Core Biopsy (CNB) Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to
Management of Papillary Lesions Diagnosed at Rad Path Concordant Core Biopsy (CNB) Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to
Breast Cancer. Saima Saeed MD
 Breast Cancer Saima Saeed MD Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women 1 in 8 women will develop breast cancer Incidence/mortality rates have declined Breast
Breast Cancer Saima Saeed MD Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women 1 in 8 women will develop breast cancer Incidence/mortality rates have declined Breast
Cytyc Corporation - Case Presentation Archive - March 2002
 FirstCyte Ductal Lavage History: 68 Year Old Female Gail Index: Unknown Clinical History: Negative Mammogram in 1995 6 yrs. later presents with bloody nipple discharge Subsequent suspicious mammogram Suspicious
FirstCyte Ductal Lavage History: 68 Year Old Female Gail Index: Unknown Clinical History: Negative Mammogram in 1995 6 yrs. later presents with bloody nipple discharge Subsequent suspicious mammogram Suspicious
Ductal Proliferations of the Breast: The Good, the Bad, and the Ugly
 Ductal Proliferations of the Breast: The Good, the Bad, and the Ugly Melinda F. Lerwill, MD CRITERIA FOR DISTINGUISHING LOW-GRADE DUCTAL CARCINOMA IN SITU FROM USUAL DUCTAL HYPERPLASIA CYTOLOGY Low-grade
Ductal Proliferations of the Breast: The Good, the Bad, and the Ugly Melinda F. Lerwill, MD CRITERIA FOR DISTINGUISHING LOW-GRADE DUCTAL CARCINOMA IN SITU FROM USUAL DUCTAL HYPERPLASIA CYTOLOGY Low-grade
The management of B3 lesions with emphasis on lobular neoplasia
 The management of B3 lesions with emphasis on lobular neoplasia Abeer Shaaban Queen Elizabeth Hospital Birmingham NHSBSP core biopsy categories B1 - Normal B2 - Benign B3 Uncertain malignant potential
The management of B3 lesions with emphasis on lobular neoplasia Abeer Shaaban Queen Elizabeth Hospital Birmingham NHSBSP core biopsy categories B1 - Normal B2 - Benign B3 Uncertain malignant potential
RSNA, /radiol Appendix E1. Methods
 RSNA, 2016 10.1148/radiol.2016151097 Appendix E1 Methods US and Near-infrared Data Acquisition Four optical wavelengths (740 nm, 780 nm, 808 nm, and 830 nm) were used to sequentially deliver the light
RSNA, 2016 10.1148/radiol.2016151097 Appendix E1 Methods US and Near-infrared Data Acquisition Four optical wavelengths (740 nm, 780 nm, 808 nm, and 830 nm) were used to sequentially deliver the light
Malignant transformation of fibroadenomas
 Malignant transformation of fibroadenomas Poster No.: C-2503 Congress: ECR 2013 Type: Educational Exhibit Authors: L. N. Elias, M. A. Rudner, L. M. Yano, P. C. Moraes, Y. 1 1 1 1 1 1 2 1 2 Chang, M. B.
Malignant transformation of fibroadenomas Poster No.: C-2503 Congress: ECR 2013 Type: Educational Exhibit Authors: L. N. Elias, M. A. Rudner, L. M. Yano, P. C. Moraes, Y. 1 1 1 1 1 1 2 1 2 Chang, M. B.
Case Report Synchronous Bilateral Solid Papillary Carcinomas of the Breast
 Case Reports in Surgery Volume 2013, Article ID 812129, 4 pages http://dx.doi.org/10.1155/2013/812129 Case Report Synchronous Bilateral Solid Papillary Carcinomas of the Breast Noriko Yoshimura, 1 Shigeru
Case Reports in Surgery Volume 2013, Article ID 812129, 4 pages http://dx.doi.org/10.1155/2013/812129 Case Report Synchronous Bilateral Solid Papillary Carcinomas of the Breast Noriko Yoshimura, 1 Shigeru
Atypical papillary lesions after core needle biopsy and subsequent breast carcinoma
 Asian Biomedicine Vol. 5 No. 2 April 2011; 243-248 DOI: 10.5372/1905-7415.0502.031 Original article Atypical papillary lesions after core needle biopsy and subsequent breast carcinoma Tuenchit Khamapirad
Asian Biomedicine Vol. 5 No. 2 April 2011; 243-248 DOI: 10.5372/1905-7415.0502.031 Original article Atypical papillary lesions after core needle biopsy and subsequent breast carcinoma Tuenchit Khamapirad
Treatment options for the precancerous Atypical Breast lesions. Prof. YOUNG-JIN SUH The Catholic University of Korea
 Treatment options for the precancerous Atypical Breast lesions Prof. YOUNG-JIN SUH The Catholic University of Korea Not so benign lesions? Imaging abnormalities(10% recall) lead to diagnostic evaluation,
Treatment options for the precancerous Atypical Breast lesions Prof. YOUNG-JIN SUH The Catholic University of Korea Not so benign lesions? Imaging abnormalities(10% recall) lead to diagnostic evaluation,
Papillary Lesions of the Breast A Practical Approach to Diagnosis. (Arch Pathol Lab Med. 2016;140: ; doi: /arpa.
 Papillary Lesions of the Breast A Practical Approach to Diagnosis (Arch Pathol Lab Med. 2016;140:1052 1059; doi: 10.5858/arpa.2016-0219-RA) Papillary lesions of the breast Span the spectrum of benign,
Papillary Lesions of the Breast A Practical Approach to Diagnosis (Arch Pathol Lab Med. 2016;140:1052 1059; doi: 10.5858/arpa.2016-0219-RA) Papillary lesions of the breast Span the spectrum of benign,
ACCME/Disclosures. Cribriform Lesions of the Prostate. Case
 Cribriform Lesions of the Prostate Ming Zhou, MD, PhD Departments of Pathology and Urology New York University Langone Medical Center New York, NY Ming.Zhou@NYUMC.ORG ACCME/Disclosures The USCAP requires
Cribriform Lesions of the Prostate Ming Zhou, MD, PhD Departments of Pathology and Urology New York University Langone Medical Center New York, NY Ming.Zhou@NYUMC.ORG ACCME/Disclosures The USCAP requires
Basement membrane in lobule.
 Bahram Memar, MD Basement membrane in lobule. Normal lobule-luteal phase Normal lobule-follicular phase Lactating breast Greater than 95% are adenocarcinomas in situ carcinomas and invasive carcinomas.
Bahram Memar, MD Basement membrane in lobule. Normal lobule-luteal phase Normal lobule-follicular phase Lactating breast Greater than 95% are adenocarcinomas in situ carcinomas and invasive carcinomas.
Review Article. ISSN: (Print) ISSN: (Online)
 Scholars International Journal of Obstetrics and Gynecology Abbreviated key title: Sch. Int. J. Obstet. Gynec. A Publication by Scholars Middle East Publishers Dubai, United Arab Emirates ISSN: 2616-8235
Scholars International Journal of Obstetrics and Gynecology Abbreviated key title: Sch. Int. J. Obstet. Gynec. A Publication by Scholars Middle East Publishers Dubai, United Arab Emirates ISSN: 2616-8235
In Situ Breast Carcinoma. James L. Connolly, M.D Beth Israel Deaconess Medical Center Professor of Pathology Harvard Medical School Boston, MA
 In Situ Breast Carcinoma James L. Connolly, M.D Beth Israel Deaconess Medical Center Professor of Pathology Harvard Medical School Boston, MA Content In Situ Ductal Carcinoma In Situ Lobular Carcinoma
In Situ Breast Carcinoma James L. Connolly, M.D Beth Israel Deaconess Medical Center Professor of Pathology Harvard Medical School Boston, MA Content In Situ Ductal Carcinoma In Situ Lobular Carcinoma
Recurrence following Treatment of Ductal Carcinoma in Situ with Skin-Sparing Mastectomy and Immediate Breast Reconstruction
 Recurrence following Treatment of Ductal Carcinoma in Situ with Skin-Sparing Mastectomy and Immediate Breast Reconstruction Aldona J. Spiegel, M.D., and Charles E. Butler, M.D. Houston, Texas Skin-sparing
Recurrence following Treatment of Ductal Carcinoma in Situ with Skin-Sparing Mastectomy and Immediate Breast Reconstruction Aldona J. Spiegel, M.D., and Charles E. Butler, M.D. Houston, Texas Skin-sparing
Histological Type. Morphological and Molecular Typing of breast Cancer. Nottingham Tenovus Primary Breast Cancer Study. Survival (%) Ian Ellis
 Morphological and Molecular Typing of breast Cancer Ian Ellis Molecular Medical Sciences, University of Nottingham Department of Histopathology, Nottingham University Hospitals NHS Trust Histological Type
Morphological and Molecular Typing of breast Cancer Ian Ellis Molecular Medical Sciences, University of Nottingham Department of Histopathology, Nottingham University Hospitals NHS Trust Histological Type
High risk lesions of the breast : Review of the current diagnostic and management strategies
 High risk lesions of the breast : Review of the current diagnostic and management strategies Poster No.: C-1204 Congress: ECR 2016 Type: Educational Exhibit Authors: P. Jagmohan, F. J. Pool, P. G. Pillay,
High risk lesions of the breast : Review of the current diagnostic and management strategies Poster No.: C-1204 Congress: ECR 2016 Type: Educational Exhibit Authors: P. Jagmohan, F. J. Pool, P. G. Pillay,
Benign Breast Disease and Breast Cancer Risk
 Benign Breast Disease and Breast Cancer Risk Jean F. Simpson, M.D. Vanderbilt University Nashville, Tennessee December 1, 2011 Nashville Nashville Lebanon 1 Cedars of Lebanon State Park The American University
Benign Breast Disease and Breast Cancer Risk Jean F. Simpson, M.D. Vanderbilt University Nashville, Tennessee December 1, 2011 Nashville Nashville Lebanon 1 Cedars of Lebanon State Park The American University
CNB vs Surgical Excision
 Update on Core Needle Biopsy of Non-palpable Breast Lesions Nour Sneige, M.D. UT MD Anderson Cancer Center Houston, Tx Image-Guided CNB of Breast Lesions An alternative to surgical biospy CNB vs Surgical
Update on Core Needle Biopsy of Non-palpable Breast Lesions Nour Sneige, M.D. UT MD Anderson Cancer Center Houston, Tx Image-Guided CNB of Breast Lesions An alternative to surgical biospy CNB vs Surgical
ACRIN 6666 Therapeutic Surgery Form
 S1 ACRIN 6666 Therapeutic Surgery Form 6666 Instructions: Complete a separate S1 form for each separate area of each breast excised with the intent to treat a cancer (e.g. each lumpectomy or mastectomy).
S1 ACRIN 6666 Therapeutic Surgery Form 6666 Instructions: Complete a separate S1 form for each separate area of each breast excised with the intent to treat a cancer (e.g. each lumpectomy or mastectomy).
CPC 4 Breast Cancer. Rochelle Harwood, a 35 year old sales assistant, presents to her GP because she has noticed a painless lump in her left breast.
 CPC 4 Breast Cancer Rochelle Harwood, a 35 year old sales assistant, presents to her GP because she has noticed a painless lump in her left breast. 1. What are the most likely diagnoses of this lump? Fibroadenoma
CPC 4 Breast Cancer Rochelle Harwood, a 35 year old sales assistant, presents to her GP because she has noticed a painless lump in her left breast. 1. What are the most likely diagnoses of this lump? Fibroadenoma
Synchronous squamous cell carcinoma of the breast. and invasive lobular carcinoma
 Sentani K et al. 1 Letter to the editor Synchronous squamous cell carcinoma of the breast and invasive lobular carcinoma Kazuhiro Sentani, 1 Takashi Tashiro, 2 Naohide Oue, 1 Wataru Yasui 1 1 Department
Sentani K et al. 1 Letter to the editor Synchronous squamous cell carcinoma of the breast and invasive lobular carcinoma Kazuhiro Sentani, 1 Takashi Tashiro, 2 Naohide Oue, 1 Wataru Yasui 1 1 Department
The Hot Topic for today is a biopsy from a 58-year-old woman who had worrisome mammographic calcifications on screening.
 The Hot Topic for today is a biopsy from a 58-year-old woman who had worrisome mammographic calcifications on screening. 1 My name is Dan Visscher; I am a consultant in the Division of Anatomic Pathology
The Hot Topic for today is a biopsy from a 58-year-old woman who had worrisome mammographic calcifications on screening. 1 My name is Dan Visscher; I am a consultant in the Division of Anatomic Pathology
Evaluation of Breast Specimens Removed by Needle Localization Technique
 Evaluation of Breast Specimens Removed by Needle Localization Technique Specimen Handling: The breast specimen when received should be measured and grossly inspected for any orientation designated by the
Evaluation of Breast Specimens Removed by Needle Localization Technique Specimen Handling: The breast specimen when received should be measured and grossly inspected for any orientation designated by the
Management of Ductal Carcinoma In Situ
 Management of Ductal Carcinoma In Situ Stella Hetelekidis, MD Stuart J. Schnitt, MD Monica Morrow, MD Jay R. Harris, MD Introduction Ductal carcinoma in situ (DCIS), also known as intraductal carcinoma,
Management of Ductal Carcinoma In Situ Stella Hetelekidis, MD Stuart J. Schnitt, MD Monica Morrow, MD Jay R. Harris, MD Introduction Ductal carcinoma in situ (DCIS), also known as intraductal carcinoma,
Columnar Cell Lesions. Columnar Cell Lesions and Flat Epithelial Atypia
 Columnar Cell Lesions and Stuart J. Schnitt, M.D. Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA, USA Columnar Cell Lesions Lesions characterized by columnar epithelial cells
Columnar Cell Lesions and Stuart J. Schnitt, M.D. Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA, USA Columnar Cell Lesions Lesions characterized by columnar epithelial cells
1 NORMAL HISTOLOGY AND METAPLASIAS
 1 NORMAL HISTOLOGY AND METAPLASIAS, MD Anatomy and Histology 1 Metaplasias 2 ANATOMY AND HISTOLOGY The female breast is composed of a branching duct system, which begins at the nipple with the major lactiferous
1 NORMAL HISTOLOGY AND METAPLASIAS, MD Anatomy and Histology 1 Metaplasias 2 ANATOMY AND HISTOLOGY The female breast is composed of a branching duct system, which begins at the nipple with the major lactiferous
Ductal Carcinoma in Situ. Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA
 Ductal Carcinoma in Situ Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Definition of DCIS WHO 2012 A neoplastic proliferation
Ductal Carcinoma in Situ Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Definition of DCIS WHO 2012 A neoplastic proliferation
University of Zurich. Histology and Immunophenotype of Invasive Lobular Breast Cancer. Daily Practice and Pitfalls. Zurich Open Repository and Archive
 University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2009 Histology and Immunophenotype of Invasive Lobular Breast Cancer. Daily Practice
University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2009 Histology and Immunophenotype of Invasive Lobular Breast Cancer. Daily Practice
Significance of flat epithelial atypia on mammotome core needle biopsy: should it be excised? B
 Human Pathology (2007) 38, 35 41 www.elsevier.com/locate/humpath Original contribution Significance of flat epithelial atypia on mammotome core needle biopsy: should it be excised? B Lakshmi P. Kunju MD,
Human Pathology (2007) 38, 35 41 www.elsevier.com/locate/humpath Original contribution Significance of flat epithelial atypia on mammotome core needle biopsy: should it be excised? B Lakshmi P. Kunju MD,
Papillary Lesions of the Breast
 Papillary Lesions of the Breast Laura C. Collins, M.D. Associate Professor of Pathology Associate Director, Division of Anatomic Pathology Beth Israel Deaconess Medical Center and Harvard Medical School
Papillary Lesions of the Breast Laura C. Collins, M.D. Associate Professor of Pathology Associate Director, Division of Anatomic Pathology Beth Israel Deaconess Medical Center and Harvard Medical School
04/10/2018. Intraductal Papillary Neoplasms Of Breast INTRADUCTAL PAPILLOMA
 Intraductal Papillary Neoplasms Of Breast Savitri Krishnamurthy MD Professor of Pathology Deputy Division Head The University of Texas MD Anderson Cancer Center 25 th Annual Seminar in Pathology Pittsburgh,
Intraductal Papillary Neoplasms Of Breast Savitri Krishnamurthy MD Professor of Pathology Deputy Division Head The University of Texas MD Anderson Cancer Center 25 th Annual Seminar in Pathology Pittsburgh,
Columnar Cell Lesions and Flat Epithelial Atypia
 Columnar Cell Lesions and Flat Epithelial Atypia Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA Terminology for Columnar Cell
Columnar Cell Lesions and Flat Epithelial Atypia Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA Terminology for Columnar Cell
Advocating Nonsurgical Management of Patients With Small, Incidental Radial Scars at the Time of Needle Core Biopsy. A Study of 77 Cases
 Advocating Nonsurgical Management of Patients With Small, Incidental Radial Scars at the Time of Needle Core Biopsy A Study of 77 Cases Cathleen Matrai, MD; Timothy M. D Alfonso, MD; Lindsay Pharmer, MD;
Advocating Nonsurgical Management of Patients With Small, Incidental Radial Scars at the Time of Needle Core Biopsy A Study of 77 Cases Cathleen Matrai, MD; Timothy M. D Alfonso, MD; Lindsay Pharmer, MD;
Protocol for the Examination of Biopsy Specimens From Patients With Invasive Carcinoma of the Breast
 Protocol for the Examination of Specimens From Patients With Invasive Carcinoma of the Breast Version: BreastInvasive 1.0.0.0 Protocol Posting Date: February 2019 Accreditation Requirements The use of
Protocol for the Examination of Specimens From Patients With Invasive Carcinoma of the Breast Version: BreastInvasive 1.0.0.0 Protocol Posting Date: February 2019 Accreditation Requirements The use of
Flat Epithelial Atypia: A Review of Current Concepts
 90 The Open Breast Cancer Journal, 2010, 2, 90-94 Flat Epithelial Atypia: A Review of Current Concepts Amy L. Adams * Open Access Department of Pathology and Laboratory Medicine, Emory University School
90 The Open Breast Cancer Journal, 2010, 2, 90-94 Flat Epithelial Atypia: A Review of Current Concepts Amy L. Adams * Open Access Department of Pathology and Laboratory Medicine, Emory University School
Disclosures. Premalignant Lesions of the Breast: What Clinicians Want and Why. NY Times: Prone to Error: Earliest Steps to Find Cancer.
 Disclosures Premalignant Lesions of the Breast: What Clinicians Want and Why I have nothing to disclose Rick Baehner, MD Assistant Professor, UCSF Pathology NY Times: Prone to Error: Earliest Steps to
Disclosures Premalignant Lesions of the Breast: What Clinicians Want and Why I have nothing to disclose Rick Baehner, MD Assistant Professor, UCSF Pathology NY Times: Prone to Error: Earliest Steps to
3/28/2017. Disclosure of Relevant Financial Relationships. GU Evening Subspecialty Case Conference. Differential Diagnosis:
 GU Evening Subspecialty Case Conference Rajal B. Shah, M.D. VP, Medical Director, Urologic Pathology Miraca Life Sciences, Irving, Texas Clinical Associate Professor of Pathology Baylor College of Medicine,
GU Evening Subspecialty Case Conference Rajal B. Shah, M.D. VP, Medical Director, Urologic Pathology Miraca Life Sciences, Irving, Texas Clinical Associate Professor of Pathology Baylor College of Medicine,
Enterprise Interest None
 Enterprise Interest None B3 lesions of the breast What are they at surgery? Case 4 Edi Brogi MD PhD Attending Pathologist - Director of Breast Pathology Memorial Sloan Kettering Cancer Center New York
Enterprise Interest None B3 lesions of the breast What are they at surgery? Case 4 Edi Brogi MD PhD Attending Pathologist - Director of Breast Pathology Memorial Sloan Kettering Cancer Center New York
Case Scenario 1 History and Physical 3/15/13 Imaging Pathology
 Case Scenario 1 History and Physical 3/15/13 The patient is an 84 year old white female who presented with an abnormal mammogram. The patient has a five year history of refractory anemia with ringed sideroblasts
Case Scenario 1 History and Physical 3/15/13 The patient is an 84 year old white female who presented with an abnormal mammogram. The patient has a five year history of refractory anemia with ringed sideroblasts
Case Scenario 1: This case has been slightly modified from the case presented during the live session to add clarity.
 Case Scenario 1: This case has been slightly modified from the case presented during the live session to add clarity. Background: 46 year old married premenopausal female with dense breasts has noticed
Case Scenario 1: This case has been slightly modified from the case presented during the live session to add clarity. Background: 46 year old married premenopausal female with dense breasts has noticed
Breast Pathology. Breast Development
 Breast Pathology Lecturer: Hanina Hibshoosh, M.D. Reading: Kumar, Cotran, Robbins, Basic Pathology, 6th Edition, pages 623-635 Breast Development 5th week - thickening of the epidermis - milk line 5th
Breast Pathology Lecturer: Hanina Hibshoosh, M.D. Reading: Kumar, Cotran, Robbins, Basic Pathology, 6th Edition, pages 623-635 Breast Development 5th week - thickening of the epidermis - milk line 5th
Excisional biopsy or long term follow-up results in breast high-risk lesions diagnosed at core needle biopsy
 Excisional biopsy or long term follow-up results in breast high-risk lesions diagnosed at core needle biopsy Poster No.: C-2515 Congress: ECR 2015 Type: Authors: Scientific Exhibit Ö. S. Okcu 1, A. Oktay
Excisional biopsy or long term follow-up results in breast high-risk lesions diagnosed at core needle biopsy Poster No.: C-2515 Congress: ECR 2015 Type: Authors: Scientific Exhibit Ö. S. Okcu 1, A. Oktay
Case Scenario 1: This case has been slightly modified from the case presented during the live session to add clarity.
 Case Scenario 1: This case has been slightly modified from the case presented during the live session to add clarity. Background: 46 year old married premenopausal female with dense breasts has noticed
Case Scenario 1: This case has been slightly modified from the case presented during the live session to add clarity. Background: 46 year old married premenopausal female with dense breasts has noticed
Ana Sofia Preto 19/06/2013
 Ana Sofia Preto 19/06/2013 Understanding the underlying pathophysiologic processes leading to the various types of calcifications Description and illustration of the several types of calcifications, according
Ana Sofia Preto 19/06/2013 Understanding the underlying pathophysiologic processes leading to the various types of calcifications Description and illustration of the several types of calcifications, according
Pathology of the Prostate. PathoBasic Tatjana Vlajnic
 Pathology of the Prostate PathoBasic 24.01.17 Tatjana Vlajnic Overview Adenocarcinoma of the prostate Grading Special variants Mimickers of prostate adenocarcinoma Atrophy Inflammatory conditions Granulomatous
Pathology of the Prostate PathoBasic 24.01.17 Tatjana Vlajnic Overview Adenocarcinoma of the prostate Grading Special variants Mimickers of prostate adenocarcinoma Atrophy Inflammatory conditions Granulomatous
PSA. HMCK, p63, Racemase. HMCK, p63, Racemase
 Case 1 67 year old male presented with gross hematuria H/o acute prostatitis & BPH Urethroscopy: small, polypoid growth with a broad base emanating from the left side of the verumontanum Serum PSA :7 ng/ml
Case 1 67 year old male presented with gross hematuria H/o acute prostatitis & BPH Urethroscopy: small, polypoid growth with a broad base emanating from the left side of the verumontanum Serum PSA :7 ng/ml
INTRADUCTAL LESIONS OF THE PROSTATE. Jonathan I. Epstein
 INTRADUCTAL LESIONS OF THE PROSTATE Jonathan I. Epstein Topics Prostatic intraepithelial neoplasia (PIN) Intraductal adenocarcinoma (IDC-P) Intraductal urothelial carcinoma Ductal adenocarcinoma High Prostatic
INTRADUCTAL LESIONS OF THE PROSTATE Jonathan I. Epstein Topics Prostatic intraepithelial neoplasia (PIN) Intraductal adenocarcinoma (IDC-P) Intraductal urothelial carcinoma Ductal adenocarcinoma High Prostatic
BI-RADS Categorization As a Predictor of Malignancy 1
 Susan G. Orel, MD Nicole Kay, BA Carol Reynolds, MD Daniel C. Sullivan, MD BI-RADS Categorization As a Predictor of Malignancy 1 Index terms: Breast, biopsy, 00.1261 Breast neoplasms, localization, 00.125,
Susan G. Orel, MD Nicole Kay, BA Carol Reynolds, MD Daniel C. Sullivan, MD BI-RADS Categorization As a Predictor of Malignancy 1 Index terms: Breast, biopsy, 00.1261 Breast neoplasms, localization, 00.125,
DCIS of the Breast--MRI findings with mammographic correlation.
 DCIS of the Breast--MRI findings with mammographic correlation. Poster No.: C-1560 Congress: ECR 2013 Type: Educational Exhibit Authors: N. B. Ibrahim, P. Morris, S. ANANDAN; Burlington, MA/US Keywords:
DCIS of the Breast--MRI findings with mammographic correlation. Poster No.: C-1560 Congress: ECR 2013 Type: Educational Exhibit Authors: N. B. Ibrahim, P. Morris, S. ANANDAN; Burlington, MA/US Keywords:
Atypical proliferative lesions diagnosed on core biopsy - 6 year review
 Atypical proliferative lesions diagnosed on core biopsy - 6 year review Dr Angela Harris, Dr Julie Weigner & Dr Ricardo Vilain NSW Health Pathology Pathology North, Hunter Anatomical Pathology & Cytology
Atypical proliferative lesions diagnosed on core biopsy - 6 year review Dr Angela Harris, Dr Julie Weigner & Dr Ricardo Vilain NSW Health Pathology Pathology North, Hunter Anatomical Pathology & Cytology
Utility of Adequate Core Biopsy Samples from Ultrasound Biopsies Needed for Today s Breast Pathology
 Utility of Adequate Core Biopsy Samples from Ultrasound Biopsies Needed for Today s Breast Pathology Ugur Ozerdem, M.D. 1 Abstract Background: There is a paradigm shift in breast biopsy philosophy. In
Utility of Adequate Core Biopsy Samples from Ultrasound Biopsies Needed for Today s Breast Pathology Ugur Ozerdem, M.D. 1 Abstract Background: There is a paradigm shift in breast biopsy philosophy. In
Mammographic imaging of nonpalpable breast lesions. Malai Muttarak, MD Department of Radiology Chiang Mai University Chiang Mai, Thailand
 Mammographic imaging of nonpalpable breast lesions Malai Muttarak, MD Department of Radiology Chiang Mai University Chiang Mai, Thailand Introduction Contents Mammographic signs of nonpalpable breast cancer
Mammographic imaging of nonpalpable breast lesions Malai Muttarak, MD Department of Radiology Chiang Mai University Chiang Mai, Thailand Introduction Contents Mammographic signs of nonpalpable breast cancer
Breast Cancer. Dr. Andres Wiernik 2017
 Breast Cancer Dr. Andres Wiernik 2017 Agenda: The Facts! (Epidemiology/Risk Factors) Biological Classification/Phenotypes of Breast Cancer Treatment approach Local Systemic Agenda: The Facts! (Epidemiology/Risk
Breast Cancer Dr. Andres Wiernik 2017 Agenda: The Facts! (Epidemiology/Risk Factors) Biological Classification/Phenotypes of Breast Cancer Treatment approach Local Systemic Agenda: The Facts! (Epidemiology/Risk
Breast pathology. 2nd Department of Pathology Semmelweis University
 Breast pathology 2nd Department of Pathology Semmelweis University Breast pathology - Summary - Benign lesions - Acute mastitis - Plasma cell mastitis / duct ectasia - Fat necrosis - Fibrocystic change/
Breast pathology 2nd Department of Pathology Semmelweis University Breast pathology - Summary - Benign lesions - Acute mastitis - Plasma cell mastitis / duct ectasia - Fat necrosis - Fibrocystic change/
Papillary Lesions of the breast
 Papillary Lesions of the breast Emad Rakha Professor of Breast Pathology The University of Nottingham Papillary lesions of the breast are a heterogeneous group of disease, which are characterised by neoplastic
Papillary Lesions of the breast Emad Rakha Professor of Breast Pathology The University of Nottingham Papillary lesions of the breast are a heterogeneous group of disease, which are characterised by neoplastic
Immunohistochemical studies (ER & Ki-67) in Proliferative breast lesions adjacent to malignancy
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 13, Issue 3 Ver. IV. (Mar. 2014), PP 84-89 Immunohistochemical studies (ER & Ki-67) in Proliferative
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 13, Issue 3 Ver. IV. (Mar. 2014), PP 84-89 Immunohistochemical studies (ER & Ki-67) in Proliferative
Lobular carcinoma in situ (LCIS) was described by Foote
 Current Concepts in Diagnosis, Molecular Features, and Management of Lobular Carcinoma In Situ of the Breast With a Discussion of Morphologic Variants Paula S. Ginter, MD; Timothy M. D Alfonso, MD Context.
Current Concepts in Diagnosis, Molecular Features, and Management of Lobular Carcinoma In Situ of the Breast With a Discussion of Morphologic Variants Paula S. Ginter, MD; Timothy M. D Alfonso, MD Context.
Prostate Immunohistochemistry. Literature Interpretation: Caveats. Must be aware of staining pattern of antibody in the relevant tissue
 IHC Interpretation: General Principles (1) Prostate Immunohistochemistry Murali Varma Cardiff, UK wptmv@cf.ac.uk Sarajevo Nov 2013 Must be aware of staining pattern of antibody in the relevant tissue Nuclear/cytoplasmic/membranous
IHC Interpretation: General Principles (1) Prostate Immunohistochemistry Murali Varma Cardiff, UK wptmv@cf.ac.uk Sarajevo Nov 2013 Must be aware of staining pattern of antibody in the relevant tissue Nuclear/cytoplasmic/membranous
Consensus Guideline on Image-Guided Percutaneous Biopsy of Palpable and Nonpalpable Breast Lesions
 Consensus Guideline on Image-Guided Percutaneous Biopsy of Palpable and Nonpalpable Breast Lesions Purpose: To outline the use of minimally invasive biopsy techniques (MIBT) for palpable and nonpalpable
Consensus Guideline on Image-Guided Percutaneous Biopsy of Palpable and Nonpalpable Breast Lesions Purpose: To outline the use of minimally invasive biopsy techniques (MIBT) for palpable and nonpalpable
K. M. Sorensen Utah State University, Logan, Utah
 K. M. Sorensen Utah State University, Logan, Utah T. E. Doyle, B. D. Borget, M. Cervantes, J. A. Chappell, B. J. Curtis, M. A. Grover, J. E. Roring, J. E. Stiles, and L. A. Thompson Utah Valley University,
K. M. Sorensen Utah State University, Logan, Utah T. E. Doyle, B. D. Borget, M. Cervantes, J. A. Chappell, B. J. Curtis, M. A. Grover, J. E. Roring, J. E. Stiles, and L. A. Thompson Utah Valley University,
BREAST PATHOLOGY. Fibrocystic Changes
 BREAST PATHOLOGY Lesions of the breast are very common, and they present as palpable, sometimes painful, nodules or masses. Most of these lesions are benign. Breast cancer is the 2 nd most common cause
BREAST PATHOLOGY Lesions of the breast are very common, and they present as palpable, sometimes painful, nodules or masses. Most of these lesions are benign. Breast cancer is the 2 nd most common cause
Imaging in breast cancer. Mammography and Ultrasound Donya Farrokh.MD Radiologist Mashhad University of Medical Since
 Imaging in breast cancer Mammography and Ultrasound Donya Farrokh.MD Radiologist Mashhad University of Medical Since A mammogram report is a key component of the breast cancer diagnostic process. A mammogram
Imaging in breast cancer Mammography and Ultrasound Donya Farrokh.MD Radiologist Mashhad University of Medical Since A mammogram report is a key component of the breast cancer diagnostic process. A mammogram
Incidence of ductal lesions
 Ductal Proliferative Lesions of the Breast: From FEA to ADH to DCIS Incidence of ductal lesions Pre-mammography: DCIS < 3% of breast cancers, large palpable masses, with invasion Mammography: DCIS 25%
Ductal Proliferative Lesions of the Breast: From FEA to ADH to DCIS Incidence of ductal lesions Pre-mammography: DCIS < 3% of breast cancers, large palpable masses, with invasion Mammography: DCIS 25%
ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY. Jonathan I. Epstein.
 1 ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY Jonathan I. Epstein Professor Pathology, Urology, Oncology The Reinhard Professor of Urological
1 ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY Jonathan I. Epstein Professor Pathology, Urology, Oncology The Reinhard Professor of Urological
Original Article. Spontaneous Healing of Breast Cancer
 Breast Cancer Vol. 12 No. 2 April 2005 Original Article Rie Horii 1, 3, Futoshi Akiyama 1, Fujio Kasumi 2, Morio Koike 3, and Goi Sakamoto 1 1 Department of Breast Pathology, the Cancer Institute of the
Breast Cancer Vol. 12 No. 2 April 2005 Original Article Rie Horii 1, 3, Futoshi Akiyama 1, Fujio Kasumi 2, Morio Koike 3, and Goi Sakamoto 1 1 Department of Breast Pathology, the Cancer Institute of the
Image-guided core needle biopsy has become a standard
 Pathologic Review of Atypical Hyperplasia Identified by Image-Guided Breast Needle Core Biopsy Correlation With Excision Specimen I-Tien Yeh, MD; Diana Dimitrov, MD; Pamela Otto, MD; Alexander R. Miller,
Pathologic Review of Atypical Hyperplasia Identified by Image-Guided Breast Needle Core Biopsy Correlation With Excision Specimen I-Tien Yeh, MD; Diana Dimitrov, MD; Pamela Otto, MD; Alexander R. Miller,
Gross appearance of nodular hyperplasia in material obtained from suprapubic prostatectomy. Note the multinodular appearance and the admixture of
 Tiền liệt tuyến Tiền liệt tuyến Gross appearance of nodular hyperplasia in material obtained from suprapubic prostatectomy. Note the multinodular appearance and the admixture of solid and microcystic areas.
Tiền liệt tuyến Tiền liệt tuyến Gross appearance of nodular hyperplasia in material obtained from suprapubic prostatectomy. Note the multinodular appearance and the admixture of solid and microcystic areas.
Papillary Lesions of the Breast: WHO Update
 Papillary Lesions of the Breast: WHO Update Stuart J. Schnitt, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA, USA Papillary Lesions of the Breast
Papillary Lesions of the Breast: WHO Update Stuart J. Schnitt, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA, USA Papillary Lesions of the Breast
University Journal of Pre and Para Clinical Sciences
 ISSN 2455 2879 Volume 2 Issue 1 2016 Metaplastic carcinoma breast a rare case report Abstract : Metaplastic carcinoma of the breast is a rare malignancy with two distinct cell lines described as a breast
ISSN 2455 2879 Volume 2 Issue 1 2016 Metaplastic carcinoma breast a rare case report Abstract : Metaplastic carcinoma of the breast is a rare malignancy with two distinct cell lines described as a breast
Diagnostic benefits of ultrasound-guided. CNB) versus mammograph-guided biopsy for suspicious microcalcifications. without definite breast mass
 Volume 118 No. 19 2018, 531-543 ISSN: 1311-8080 (printed version); ISSN: 1314-3395 (on-line version) url: http://www.ijpam.eu ijpam.eu Diagnostic benefits of ultrasound-guided biopsy versus mammography-guided
Volume 118 No. 19 2018, 531-543 ISSN: 1311-8080 (printed version); ISSN: 1314-3395 (on-line version) url: http://www.ijpam.eu ijpam.eu Diagnostic benefits of ultrasound-guided biopsy versus mammography-guided
