Renal Dendritic Cells Adopt a Pro-Inflammatory Phenotype in Obstructive Uropathy to Activate T Cells but Do Not Directly Contribute to Fibrosis
|
|
|
- Roy Moore
- 5 years ago
- Views:
Transcription
1 ASIP 2012 AJP CME Program Cardiovascular, Pulmonary, and Renal Pathology The American Journal of Pathology, Vol. 180, No. 1, January 2012 Copyright 2012 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved. DOI: /j.ajpath Renal Dendritic Cells Adopt a Pro-Inflammatory Phenotype in Obstructive Uropathy to Activate T Cells but Do Not Directly Contribute to Fibrosis Sarah L. Snelgrove,* Joshua Y. Kausman,* Cecilia Lo,* Camden Lo, Joshua D. Ooi,* P. Toby Coates, Michael J. Hickey,* Stephen R. Holdsworth,* Christian Kurts, Daniel R. Engel, and A. Richard Kitching* From the Centre for Inflammatory Diseases,* Monash University Department of Medicine, and the Departments of Neophrology, and Pediatric Nephrology, Monash Medical Centre, Clayton, Victoria, Australia; Monash Microimaging, Monash University, Clayton, Victoria, Australia; the Central Northern Adelaide Renal and Transplantation Service, Royal Adelaide Hospital, South Australia and Department of Medicine, The University of Adelaide, South Australia; and the Institutes of Molecular Medicine and Experimental Immunology, University of Bonn, Bonn, Germany Unilateral ureteral obstruction (UUO) is a well-characterized murine model of renal inflammation leading to fibrosis. Renal dendritic cells (DCs) constitute a significant portion of kidney leukocytes and may participate in local inflammation and have critical roles in antigen presentation. The heterogeneity in renal DC populations and surface marker overlap with monocytes/macrophages has made studying renal DCs difficult. These studies used CD11c-promoter driven reporter/depletion mice to study DCs in vivo. Studying early local inflammatory events (day 3 of UUO), in vivo multiphoton imaging of the intact kidney of CD11c reporter mice revealed more dendrite extensions and increased activity of renal DCs in real time. Phenotypic analysis suggested resident DC maturation in obstructed kidneys with increased CD11b and less F4/80 expressed. CD11b hi Gr-1 inflammatory DCs were also present in obstructed kidneys. T-cell receptor transgenic mice revealed enhanced antigen-presenting capacity of renal DCs after UUO, with increased antigen-specific T-cell proliferation in vivo and ex vivo. However, conditional DC ablation at days 0, 2, or 4 did not attenuate fibrosis or apoptosis 7 days after UUO, and depletion at 7 days did not alter outcomes at day 14. Therefore, after UUO, renal DCs exhibit inflammatory morphological and functional characteristics and are more effective antigen-presenting cells, but they do not directly contribute to tubulointerstitial damage and fibrosis. (Am J Pathol 2012, 180:91 103; DOI: /j.ajpath ) Dendritic cells (DCs) are sentinels of the immune system, providing immunological surveillance and being capable of maintaining tolerance or initiating active immunity. These antigen-presenting cells (APCs) form heterogeneous populations with diversity in phenotype and function segregating subpopulations. Although different DC subsets and their relative functions have been extensively studied in secondary lymphoid organs, less is known about the phenotype and function of DCs in peripheral tissues such as the kidney. DCs constitute one of the major leukocyte populations in the kidney and are distributed in an organized and uniform fashion throughout the tubulointerstitium. 1 4 Like conventional DCs, renal DCs express CD11c and major histocompatibility complex class II (MHC II) but some renal DCs express markers also associated with tissue macrophages (in the mouse CD11b, F4/80 and CX3CR1), making discrimination between the two subsets more difficult. 3,4 Dendritic cells can act to promote or regulate renal disease in two ways. First, DCs have the capacity to act This work was supported by grants from the National Health and Medical Research Council of Australia (Project Grant ), the Go8 Australia- Germany DAAD Co-operation Scheme and the Deutsche Forschungsgemeinschaft grants SFBTR57 and KFO228. Accepted for publication September 27, A.R.K. and D.R.E. contributed equally to this work. CME Disclosure: None of the authors disclosed any relevant financial relationships. Supplemental material for this article can be found at amjpathol.org or at doi: /j.ajpath Address reprint requests to Richard Kitching, Ph.D., Centre for Inflammatory Diseases, Monash University Department of Medicine, Monash Medical Centre, 246 Clayton Road, Clayton, VIC 3168, Australia. Richard.Kitching@monash.edu. 91
2 92 Snelgrove et al as professional APCs to initiate or regulate antigen-specific (adaptive) immune responses by presenting antigen to T cells. In addition to this role as a bridge between innate and adaptive immunity, DCs have the capacity to act locally as innate immune cells and to directly modify local inflammation. Although renal DCs have been shown to produce inflammatory mediators, for example tumor necrosis factor (TNF), 5,6 few studies have investigated the functional role of DCs in renal disease (by the conditional ablation of DCs). In immune-mediated disease renal DCs have been reported as having early local antiinflammatory properties and later pro-inflammatory roles. 7,8 In cisplatin-induced acute kidney injury, a toxic nephropathy where there is no evidence for a role of adaptive immunity, DCs are protective via interleukin-10 production. 9 Clearly, DCs can produce pro-inflammatory and anti-inflammatory cytokines, which may exacerbate or ameliorate pathological processes. DCs have been implicated in a variety of important pathological conditions, and this article defines their contribution to the early inflammatory events in renal fibrosis. Renal fibrosis occurs as a consequence of chronic kidney disease in both children and adults, and the consequent progressive decline in kidney function may ultimately lead to renal failure. Unilateral ureteric obstruction (UUO) is a common experimental model used to study renal fibrosis. Obstruction initiates a series of pathological changes, including interstitial inflammation, pro-inflammatory cytokine release, tubular cell apoptosis, myofibroblast activation, and tubulointerstitial fibrosis, all of which have deleterious effects on kidney function. 10,11 DCs are present in the kidney and early after UUO, secrete proinflammatory cytokines. 6 Although some, 12 but not other studies 13 suggest a role for T cells in obstructive uropathy, there is no documented role for antigen-specific responses. However antigen-specific responses, including those relating to infection and to allogeneic responses, can occur in the presence of obstruction in native and transplant kidneys. The present study uses a range of techniques, including DC-specific tools, CD11c-promoter driven enhanced yellow fluorescent protein (EYFP) reporter, and diphtheria toxin receptor (DTR) depletion mice, to define the role of DCs in the kidney, using UUO as a model inflammatory disease. By means of in vivo multiphoton microscopy and ex vivo phenotypic analyses, this study provides an analysis of both renal DCs from healthy kidneys and the changes induced in this population after obstruction. Renal DCs undergo morphological changes and are functionally activated in the injured kidney, consistent with a pro-inflammatory phenotype. The low capacity of renal DCs (compared with splenic DCs) to induce T-cell proliferation is increased in the inflammatory milieu that exists after UUO. Depletion studies show that although renal CD11c cells become activated during inflammation (assessed by surface phenotype, cytokine production and antigenpresenting capacity), renal DCs do not directly contribute to the progression of apoptosis or fibrosis in the kidney in the early stages after UUO. Materials and Methods Mice and Unilateral Ureteric Obstruction C57BL/6 and OT-I mice [TCR transgenic specific for H-2K b and ovalbumin (OVA) 14 ]werebredatmonash Animal Services (Melbourne, Australia). CD11c-EYFP 15 and rat insulin promoter (RIP) mova mice expressing membrane-bound OVA under the control of the rat insulin promoter 16 were bred and maintained at Monash Medical Centre (Melbourne, Australia). CD11c- DTR mice, 17 which also express green fluorescent protein (GFP) under the control of the CD11c promoter, were bred at the Institute for Molecular Medicine and Experimental Immunology (Bonn, Germany). All mice were on a C57BL/6 background. Experiments were conducted in mice 8 to 14 weeks of age and were approved by the Monash Medical Centre Animal Ethics Committee or the German Institutional and Government Review Boards. Adult male mice were anesthetized via inhalation of sevoflurane (Abbotts Laboratories Ltd, Berkshire, UK). UUO was performed through a midline incision. After the left ureter was identified, it was tied with silk suture at two points and permanently ligated. Sham-operated mice underwent an identical procedure but without ureteric ligation. At 3, 7, or 14 days postobstruction, mice were sacrificed and kidneys collected. For DC depletion, sexmatched, adult CD11c-DTR transgenic mice were used. CD11c-DTR transgenic or control (transgene negative or wild-type) mice were injected i.p with 4 ng of diphtheria toxin (DT; Sigma-Aldrich, St. Louis, MO) per gram of body weight to specifically deplete CD11c DCs, as previously described. The administration of DT to the control mice does not deplete DCs. Depletion of renal CD11c cells in CD11c-DTR mice, measured 2 days after administration of DTR, as 72% (means of n 2depletedandn 2nondepletedmice),consistentwiththedegreeofdepletion of renal CD11c cells using the CD11c-DTR in previous studies. 7 9 Depletion was performed on days 0, 2, 4, or 7. On day 7 (day 14 for mice depleted at day 7), mice were sacrificed and the kidneys collected for histological analysis. Kidney samples were fixed in 2% paraformaldehyde, embedded in paraffin, and sectioned at 4 m. Renal Multiphoton Microscopy Intravital multiphoton microscopy of the intact kidney was performed on mice anesthetized by i.p. injection of 150 mg/kg ketamine hydrochloride and 10 mg/kg xylazine, as described previously. 18 The jugular vein was cannulated for the administration of fluorescent reagents and additional anesthetic. The left kidney was exteriorized through a lateral skin incision and held in a kidney holder specifically molded to the shape of the left kidney. Surgical glue was used to aid in keeping the kidney in place. Mice were kept at a constant temperature of 37 C on a heating pad. The kidney was
3 Dendritic Cells in Renal Fibrosis 93 immersed in bicarbonate buffered saline at 37 C and covered with a coverslip. Time lapse recordings of EYFP DCs in the kidney of CD11c-EYFP mice were captured using a Leica SP5 mulitphoton microscope system, equipped with a NA water immersion objective (Leica Microsystems, Mannheim, Germany). Fluorophores were excited by a SpectraPhysics MaiTai pulsed infrared laser tuned to 860 nm, with emissions captured by nondescanned detectors with 525/50 nm (YFP), 590/50 nm (TRITC), and 650/50 nm (Alexa 633) filters. Images were collected with 120 m Z-depthin6- m steps, with one Z-stack every 30s for 15 to 25 minutes. Images were processed using Imaris (v7.0, Bitplane AG), and analysis was performed in Microsoft Excel (Microsoft Corporation, Redmond, WA). Specificity of EYFP expression by renal DCs in these reporter mice was confirmed by flow cytometry, where at least 95% of all EYFP cells expressed CD11c (see Supplemental Figure S1 at To determine DC activity, in vivo changes in morphology were tracked for individual DCs and a morphology index derived. Each DC three-dimensional shape can be described by the following mathematical model of sphericity, 19 where the value is between 0 (an absolute 2D object) and 1 (a perfect sphere): Sphericity 1 3 (6 x cell volume) 2 3/cell surface area. The sphericity of each DC was then tracked over time, with the sphericity difference between consecutive time points averaged over the entire time course to generate a value which describes the activity of DCs, as described by the rate of change in their morphology index ( MI). Cells with relatively low MI values change morphology less than cells with relatively high MI values. Isolation of DCs from Kidney or Lymph Nodes Kidneys and lymph nodes (LN) were digested with 1 mg/ml Collagenase D and 0.1 mg/ml DNase I (both Roche Diagnostics, Indianapolis, IN), as described previously. 3 After washing in RPMI 1640/10% fetal calf serum, kidney preparations were left to settle for 10 minutes to remove tubular debris. Erythrocytes were lysed with ammonium chloride and single cell preparations filtered through 70- m mesh. In some experiments, DCs were further sorted by flow cytometry (MoFlo XDP, Beckman Coulter, Miami, FL) by sorting on the CD45 CD11c population. Postsorting analysis of sorted DCs showed the purity was 90%. Flow Cytometry Cells were preincubated with anti-cd16/cd32 antibody (Ab) for 10 minutes to block nonspecific staining, then stained with directly conjugated primary Ab at predetermined optimal concentrations for 30 minutes. Fluorescentl labeled monoclonal Ab used were against CD45 (30-F11), CD11c (HL3), CD11b (M1/70), F4/80, MHC II (M5/ ), Gr-1 (RB6-8C5), B220 (RA3-6B2), CD19 (1D3), CD3 (17A2), CD4 (GK1.5), CD8 (53-6.7), V 2 (B20.1), TNF (MP6-XT22), or IL-12p40 (C15.6). Dead cells were excluded by staining with propidium iodide. To assess cytokine production by DCs, single cell kidney suspensions were incubated in 1mLRPMI/10%FCSwith10 g/ml Brefeldin A (4 hours at 37 C) before staining with surface markers. Cells were fixed and permeablized using the Cytofix/Cytoperm kit (BD Pharmingen, San Diego, CA) then stained intracellularly with anti-cytokine antibodies for 20 minutes. To enumerate the total number of cells, a known number of BD Calibrate Beads (BD Biosciences, San Jose, CA) was added per sample. Cells were acquired on a FACSCanto flow cytometer (BD Biosciences) and analyzed with FlowJo software (Tree Star, Ashland, OR). Stringent fluorescence minus one controls of both normal and obstructed kidney samples were used to ensure specificity in data analysis. T-Cell Isolation, ex Vivo T-Cell Stimulation, and in Vivo T-Cell Proliferation Assays CD8 T cells from naive OT-I donors were positively enriched from the spleen and LN by MACS (Miltenyi- Biotec, Auburn, CA) using anti-cd8 conjugated to microbeads. Enriched CD8 T cells were resuspended ( cells/ml), labeled with 5 mol/l carboxyfluorescein succinimidyl ester (CFSE), (Molecular Probes, Eugene, OR) then washed twice. A total quantity of enriched CFSE-labeled OT-I CD8 TCR transgenic cells was added to titrated numbers of sorted DCs that were pulsed with OVA peptide. Cells were cultured in a final volume of 200 ml RPMI 1640 containing 10% fetal calf serum, 50 mmol/l 2-ME, 2 mmol/l L-glutamine, 100 U/mL penicillin, and 100 mg/ml streptomycin in 96-well, U-bottomed plates. Cells were harvested after 60 hours and were stained with antibody, and proliferation was analyzed by flow cytometry. To assess DC function in vivo, whole-single-celllymphocyte suspensions from pooled OT-I spleen and LN were CFSE labeled and adult male RIPmOVA mice were i.v. injected with CFSE-labeled lymphocytes (equating to OT-I T cells). The left ureter of recipient mice was surgically ligated or control mice underwent sham operation. At day 3 after surgery, mice were sacrificed, and the renal LNs [UUO and contralateral (CL)] were collected. Cells were stained and then analyzed by flow cytometry as described above. Histology and Immunohistology Kidney sections were stained with Picrosirius red stain, and histological assessment of collagen deposition was determined using polarized light microscopy. Nonoverlapping images of the entire kidney section were taken using Nikon Abrio Polarized light microscope ( 200 magnification). National Institutes of Health (NIH) ImageJ software was used to analyze the images and results were expressed as percentage of total area. Interstitial myofibroblast [ -smooth muscle actin ( -SMA)], cleaved
4 94 Snelgrove et al caspase-3 expression, CD4 T cells, and CD8 T cells were assessed by immunohistochemistry. For -SMA and cleaved caspase-3, paraffin-fixed sections were stained with mouse anti-human -SMA monoclonal Ab (Dako, Victoria, Australia) or polyclonal rabbit anti-mouse cleaved caspase-3 Ab (Asp175, Cell Signaling Technology, Danvers, MA) overnight at 4 C. For T cells, 6- mthick frozen kidney sections were air dried and acetone fixed, and blocked with 10% rabbit serum before incubation overnight at 4 C with either rat anti-mouse CD4 (GK1.5, American Type Culture Collection [ATCC], Manassas, VA, 40 g/ml) or CD8 (53-6.7, ATCC, 40 g/ ml; rat IgG2a and IgG2b were isotype controls). Secondary steps were, for -SMA, rabbit anti-mouse immunoglobulin horseradish peroxidase (Dako), for cleaved caspase-3, Signal Stain Boost IHC detection reagent (Cell Signaling), and for T cells, rabbit anti-rat biotinylated immunoglobulin, followed by incubation with Vectastain ABC reagent (Vector Laboratories, Burlingame, CA). Bound antibodies were detected with 3,3=diaminobenzidine (Sigma-Aldrich) and sections were counterstained with 1% nuclear fast red. For -SMA, 5-10 nonoverlapping high-power ( 400) fields (HPF) of each kidney section were captured and quantified using Metamorph analysis software (Molecular Devices, Downingtown, PA) and positively stained area expressed as percentage of total area. For cleaved caspase-3 and T cells, the number of positive cells was counted in 10 to 15 high-power fields. Sections stained with Periodic Acid- Schiff were assessed for tubular injury, including tubular dilation, cast formation, tubular atrophy and the presence of inflammatory infiltrate. To confirm the findings for cleaved caspase-3, apoptotic cells in kidneys in some experiments were identified using the ApopTag In Situ Apoptosis Detection Kit (Millipore-Chemicon, CA), using 2- m paraformaldehyde-fixed paraffin sections according to the manufacturer s instructions. In addition, 3,3=diaminobenzidine was used to reveal numbers of the tubular and interstitial TdT-dUTP nick end labeling (TUNEL) positive apoptotic cells in the cortex, assessed in at least 10 high-power fields. Statistical Analysis All values are expressed as mean SEM. Statistical analysis was performed using the unpaired t-test (GraphPad Prism 5; GraphPad Software Inc., San Diego, CA). A P value of less than 0.05 was considered significant. In all figures, *P 0.05, **P 0.01, and ***P The number of repeat experiments to ensure reproducibility of results is reported in the figure legends. Results Dendritic Cells Undergo Morphological Changes After Ureteric Obstruction To determine the behavior of renal DCs in normal and injured kidneys, kidneys of live CD11cEYFP reporter mice were exteriorized and the DCs were visualized by multiphoton microscopy. DCs were present within the cortical interstitium forming a continuous, interdigitating network (Figure 1A, concordant with previous confocal images of CX3CR1-GFP cells in the normal kidney 4 ). Some DCs showed actively probing dendrites; however, in contrast to dermal DCs, 20 renal DCs were essentially immotile (Figure 1C; see also Supplemental Video S1 at Changes in DC morphology and activity were further investigated in inflammation. Three days after UUO (d3 UUO), the highly structured arrangement of DCs seen in normal mice was disrupted with DCs clustering around damaged (more autofluorescent) tubules (Figure 1B). Similar findings were seen in paraffinfixed kidney sections (day 7 after UUO; see Supplemental Figure S2 at The morphology of the DCs present in the kidney had changed by day 3 after obstruction, becoming more stellate than in control mice (Figure 1, C and D). Individual EYFP DCs were further assessed for morphological changes over time. In vivo observations showed that DCs in the injured kidney exhibited more extension and retraction of dendrites in the renal interstitium compared with control renal DCs (Figure 1, C E; see also Supplemental Videos S1 and S2 at Some mobile DCs were also consistently identified adjacent to damaged tubules after UUO, such mobility was rarely seen in the normal kidney (see Supplemental Videos S1 and S2 at amjpathol.org). Between normal and injured kidneys, absolute numbers of DCs were similar and the length of dendrites on DCs remained unchanged (mean dendrite length 10 m; Figure 1,FandG).However,thenumberofdendrite extensions per DC was doubled in kidneys 3 days after UUO (Figure 1H). These are the first in vivo microscopy studies of renal DCs in disease and provide evidence of in vivo DC activation in obstructed kidneys. Ureteric Obstruction Promotes Leukocyte Recruitment At 3 days after UUO, leukocytes within the kidney were analyzed by flow cytometry and compared with kidneys from sham-operated mice. Leukocytes, isolated by enzymatic and mechanical digestion steps, were incubated with the pan-leukocyte marker, CD45 and a range of specific cell surface markers to determine the proportion and number of CD8 and CD4 cells, DCs, B cells, macrophages, monocytes, and neutrophils. The proportions and cell number of monocytes and neutrophils were significantly increased in obstructed kidneys, accompanied by an increase in CD8 T cells (Figure 2, AandB),consistentwithpreviousfindings. 6 Although the proportion of DCs and macrophages (of all CD45 cells) was lower in injured kidneys, reflecting the influx of other leukocyte populations to the site of inflammation, actual total numbers of both DCs and macrophages were unchanged compared with kidneys from sham-operated mice.
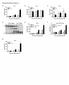
5 Dendritic Cells in Renal Fibrosis 95 Figure 1. DCs undergo morphological changes during early inflammatory events after UUO. Renal DCs were imaged using multiphoton microscopy, and DCs were assessed in the kidneys of normal mice (not operated on, n 5) and at 3 days after UUO (n 5). A representative maximum projection image (three-dimensional image x, y, and z, compressed into two dimensions, x and y) is shown for normal DCs (A) and at day 3 after UUO (B). DCs expressing EYFP are shown in yellow; the tubular epithelial cells are highlighted in red by the incorporation of a cell tracker dye; and Hoechst nuclear stain is shown in blue. Magnification of the boxed areas in panels A and B are shown as time lapse panels at four time intervals, each 2 minutes apart for normal DCs (C) and d3 UUO (D). Dotted circles encompass dendrites that exhibit morphological changes over time. The movement or activity of each individual DC was measured by the change in the morphology index over time (n 4/group). The percentage of DCs exhibiting increasing levels of cellular activity is shown (E), as is the number of DCs per field of view (F), the estimated length of the dendrites on the DCs (G), and the mean number of dendrites per DC (H), *P Phenotype of DC Subsets in Kidney Is Altered After Ureteric Obstruction Renal DCs are heterogeneous, consisting of several subsets, distinguishable by their cell surface marker expression. To characterize the phenotype of the DC subsets in the kidney during inflammation, single-cell kidney suspensions from individual mice 3 days after UUO, or shamoperated controls, were analyzed by multicolor flow cytometry. Gating on CD45 CD11c DCs, the expression of other cell surface markers were assessed (Figure 3A). In control mice, the majority of renal DCs expressed F4/80 and CD11b as previously described. 3 DCs isolated from mice 3 days after UUO exhibited different surface expression of these markers, with an increase in the both proportion and absolute number of F4/80 DCs, coinciding with fewer F4/ 80 DCs (Figure 3, AandB).AreductioninresidentDCs that were MHCII F4/80 was accompanied by an increase in the MHCII F4/80 subset (Figure 3B). As F4/80 expression is reportedly decreased on antigen uptake and DC maturation, 21 this finding suggests that some renal DCs are exhibiting a more mature phenotype. The level of CD11b expression was increased (Figure 3,A and C),and a subset of DCs expressing high levels of CD11b and Gr-1 was found almost exclusively in the obstructed kidneys (Figure 3, A and C). An increase in the number of immature CD11c MHCII DCs was also found in the injured kidneys, consistent with this subset being a DC precursor population. 3,22 Proportions of B220 plasmacytoid DCs (pdcs) and CD8 conventional DCs were also increased, with an increased absolute number of pdcs, but not CD8 DCs (Figure 3, A and D).Thus,in UUO-induced inflammation, renal DCs undergo phenotypic changes consistent with an increased level of maturation of DCs in the kidney. Cytokine production by renal DCs, specifically the production of TNF and IL-12p40 (the common chain for IL-12 and IL-23) was assessed by intracellular flow cytometry, comparing kidneys from mice 3 days after UUO with shamoperated mice. The proportion of CD11c cells producing either TNF or IL-12p40 was not altered by UUO (Figure 4,A and B). However, more CD11c F4/80 cells made TNF and IL-12p40 after UUO (though for IL-12p40, proportions were still low). TNF production by CD11c CD11b int cells was similar with and without UUO: a higher proportion of CD11c cells with hi or lo CD11b expression made TNF (Figure 4C). Fewer CD11c CD11b lo cells made IL-12p40, although proportions, even in sham-operated mice, were very low (Figure 4D).
6 96 Snelgrove et al more of this subset was present in the inflamed kidney). The differences in phenotype between sham-operated and obstructed kidneys were largely due to changes in cells expressing lower levels of CD11c. These results indicate that resident renal cells undergo maturation changes in the obstructed kidneys involving the loss of F4/80 expression and an increased level of CD11b. DCs migrate from inflamed tissue to regional LNs 27 and more DCs were present in the draining renal LN of obstructed kidneys compared with sham operated kidneys (Figure 5D). As the absolute numbers of DCs in the kidney were similar between sham operated and d3 UUO mice (Figure 2B), this efflux of resident DCs must be accompanied by recruitment into the kidney. The phenotype of immature MHC II lo,monocyte-derived inflammatory cells with lower levels of CD11c is typical of recruited inflammatory APCs and consistent with these cells as recent immigrants to the obstructed kidney. Reduced Capacity of Renal DCs to Stimulate T-Cell Responses Is Increased After UUO Figure 2. Increased numbers of CD8 T cells, monocytes, and neutrophils are recruited to the injured kidney whereas the total numbers of renal DCs are unchanged. Renal CD45 leukocyte populations were isolated from whole kidneys from mice at d3 UUO or subjected to sham operation. Cell populations were assessed by multicolor flow cytometric analysis. Staining definitions for the individual cell populations were CD8 T cells: CD45 /CD3 / CD8. CD4 T cells: CD45 /CD3 /CD4. DCs: CD45 /CD11c. B cells: CD45 /CD11c /CD19. Macrophages: CD45 /CD11c /F4/80. Monocytes: CD45 /CD11c /Gr-1 int. Neutrophils: CD45 /CD11c /CD11b hi /Gr- 1 hi.the proportions (A) and absolute cell numbers (B) of individual leukocyte subsets are shown. Each cell population is expressed as the mean SEM of five mice per group. Similar results were reproduced in three experiments. *P 0.05; **P 0.01; ***P Level of CD11c Expression Relates to Differential Expression of Markers Overlapping with Monocyte/Macrophage Lineage Monocytes can differentiate into DCs in both steady-state and inflammatory conditions Changes in DC subsets may be attributed to the recruitment of monocytic DC precursors with subsequent differentiation into DCs, activation of resident renal DCs, or both. Typically, the closer to a monocyte precursor a cell is, the lower the CD11c expression. 26 To address this issue, cells that expressed CD11c were divided into CD11c low, intermediate, or high and analyzed for the expression of markers that overlap with the monocyte/macrophage lineage (MHC II, Gr-1, F4/80 and CD11b). At day 3 after obstruction there was a threefold increase in the proportion of DCs that were CD11c high (Figure 5A). The alterations in MHC II, Gr-1, F4/80, and CD11b expression postobstruction, outlined in Figure 3, related to the degree of CD11c expression. With increasing levels of CD11c, F4/80 expression decreased and CD11b expression increased. However, MHC II lo cells, and Gr-1 cells were more often expressing low to intermediate levels of CD11c (Figure 5C). When comparing changes in the damaged kidney to the sham-operated mice, CD11c high DCs were of similar phenotype in both groups (although Given the selective increase of CD11c hi cells in the kidney and the renal LN after UUO, the antigen-presenting capacity of these cells was assessed ex vivo as a measure of their ability to stimulate antigen-specific CD8 T-cell responses. Purified syngeneic DCs, isolated from either spleen or kidney, were pulsed with OVA peptide before culture with naive, CFSE-labeled, OVA-specific OT-I T cells. Flowcytometric analyses using serial CFSE dilution showed that renal DCs from untreated kidneys were less capable of stimulating antigen-specific CD8 T-cell responses than splenic DCs (Figure 6A). The majority of renal DCs are F4/80 (unlike splenic DCs), and it has been suggested that the F4/80 molecule plays a role in tolerance induction. 28 Spleen and kidney DCs were mixed to determine whether renal DCs diminished the capacity of splenic DCs to induce OT-I proliferation. However, when stimulated by equal numbers of splenic and renal DCs (500 splenic and 500 renal DCs per culture), numbers of proliferating OT-I cells were not reduced compared with cells stimulated by 500 splenic DCs alone (Figure 6B). Similar results were obtained when using splenic and renal DCs at several other ratios (data not shown). Using UUO as a model inflammatory condition within the kidney, we assessed the classical function of DCs, that is, their capacity to stimulate T-cell responses. This function was enhanced in renal DCs 3 days after UUO, with the proportion of proliferating OT-I cells and the number of cells undergoing one to seven divisions being similar to that recorded for splenic DC stimulated T cells (Figure 6,Cand D). In vivo T-cell proliferation assays were conducted using RIP-mOVA transgenic mice that express membrane-bound OVA on kidney proximal tubule cells. 16 RIPmOVA mice received CFSE-labeled OT-I CD8 T cells followed by surgical UUO or underwent sham operation. Three days postoperatively, the draining renal LNs (UUO and CL) were collected for flow-cytometric analyses (Figure 6E). The number of OVA-specific CD8 T cells was sevenfold higher in the UUO renal LN 3 days postoperatively compared with the CL renal LN or the draining UUO LN in sham-operated mice
7 Dendritic Cells in Renal Fibrosis 97 Figure 3. Renal DCs undergo phenotypic changes after ureteric obstruction. Renal DC subsets were assessed by multicolor flow-cytometric analysis of single-cell kidney suspensions from mice at d3 UUO or sham operated controls (n 5/group). A: Representative dot plots indicate the gating strategy used to identify DCs by the expression of CD45 and the DC specific marker, CD11c. DC subsets were further assessed for the expression of F4/80, CD11b, Gr-1 versus CD11b and B220 versus CD8a. Numbers indicate the mean percentage of DCs in the given subset. B D: The absolute number of DCs categorized by the designated DC subsets is shown. Similar results were obtained in three individual experiments. **P 0.01; ***P (Figure 6F), confirming that renal DCs are functionally activated after UUO. Conditional Ablation of DCs Does Not Hinder Progression of Fibrosis in Obstructed Kidneys To determine whether DCs play a direct role in innate renal inflammation leading to fibrosis DCs were conditionally ablated during UUO. In CD11cDTR mice, DCs repopulate the kidney 3 days after DT. 7,9 In separate experiments, DT was administered to CD11cDTR mice and controls (transgene negative littermates and wild-type mice) on day 0, 2, or 4 after UUO, and renal tissue was assessed histologically on day 7 (Figure 7A). Depleting DCs at day 0 (on the day of surgery), day 2, or day 4 allowed assessment of the roles of resting tissue DCs and of more recent immigrants. By day 7, obstructed kidneys displayed tubulointerstitial damage, with dilated tubules, cast formation, tubular atrophy, and inflammatory infiltrates (Figure 7, B and C).Collagen was deposited in the interstitium, tubular cell apoptosis was present and -SMA cells (a marker for myofibroblasts) were accumulating, hallmarks in the development of renal fibrosis (Figure 7,D and H;Figure 8,A and E;and Figure 9, AandE).TheselectivedepletionofDCsatanytimepoint, initially after UUO on day 0, at an intermediate time of day 2 or in the later stages of obstruction at day 4, had no significant impact on the extent of collagen deposition in the renal cortex (Figure 7). Depleting DCs (at day 0: n 5micenot DC depleted, n 6miceDCdepleted)didnotsignificantly alter the early renal interstitial infiltration (assessed at day 4) of CD4 cells (UUO with DCs , UUO DC depleted cells/hpf, P 0.15) or CD8 cells (UUO with DCs , UUO DC depleted cells/hpf, P 0.35). Tubular apoptosis occurs before the onset of frank fibrosis. In models of ischemia/reperfusion injury and UUO, including in the current study, renal DCs produce TNF, a cytokine that can induce apoptosis. 5,6 Therefore, paraffinembedded sections were stained for cleaved caspase-3, a marker of apoptosis. Similar to the results obtained for collagen deposition, mice that were depleted of DCs at day 0, 2, or 4 during UUO showed no significant difference in tubular apoptosis compared with nondepleted control mice (Figure 8). TUNEL staining performed on sections from non DC-depleted mice and DC-depleted mice at day 0 also showed no difference in apoptosis (UUO with DCs , UUO DC day 0 depleted cells/hpf). Myofibroblast accumulation, important in renal fibrosis, can be defined by the expression of -SMA. Although, in normal kidneys, -SMA expression is restricted to the vessels, in renal fibrosis expression extends to the tubulointerstitium. The depletion of DCs at this series of time points showed that, compared with control mice given DT, there was no difference in the expression of -SMA in mice depleted of DCs at day 0, 2, or 4 (Figure 9). Collectively, the histological analysis of these three hallmarks of injury in this model
8 98 Snelgrove et al Figure 4. Renal CD11c F4/80 cells produce more cytokines after UUO. At day 3 after UUO, TNF, and IL-12p40 production was compared in kidneys from mice undergoing UUO or sham operation (n 5/group). The proportion of all CD11c cells producing either TNF (A) or IL-12p40 (B) was similar, but a higher proportion of CD11c F4/80 cells produced either cytokine after UUO. There were no consistent and marked changes when CD11c CD11b cells were examined. A higher proportion of CD11b lo and CD11b hi cells made TNF (C), and a lower proportion of CD11b lo cells made IL-12p40 (D) (although proportions were low for IL- 12p40). *P 0.05; **P collagen deposition, tubular apoptosis, and -SMA expression show that renal DCs do not alter the early renal fibrosis. To determine whether depletion of DCs at a later time point alters the development of later fibrosis, CD11c-DTR mice were given DT 7 days after UUO, and injury was studied at day 14 (Figure 10A). Collagen deposition, the accumulation of -SMA myofibroblasts and numbers of cleaved caspase-3 apoptotic cells were similar in mice in which DCs had not been depleted and in mice in which DCs were depleted at day 7 (Figure 10, B F). Discussion The current studies use a suite of in vivo and ex vivo techniques to define the phenotype and role of DCs in innate renal inflammation that results in fibrosis. The well-characterized murine model of ureteric obstruction was used to define the role of DCs in inflammatory events in the kidney. In this model, the cellular and pathological events are similar to those in human disease. 29 By 3 days after obstruction, leukocyte infiltrates are present in the obstructed kidney, 6 and overt fibrosis occurs after 7 days, with the deposition of extracellular matrix, including collagens, tubular apoptosis, and myofibroblast activation. 30,31 In this model, there is no clear evidence of the involvement of adaptive immune responses. 12,13 The key new findings in the current studies include the following: i) in vivo microscopic evidence of renal DC activation after obstruction; ii) changes in cell surface DC phenotype, reflecting increased activation and recruitment; iii) increased capacity of DCs to activate antigenspecific CD8 Tcells(potentiallyrelevantinallogeneicand infection-triggered responses associated with obstruction); and iv) no direct profibrotic effects for DCs. These results clearly show that renal DCs were activated after UUO with changes in cellular architecture, cell surface markers, and increased antigen-presenting capacity. Despite these phenotypic and functional changes, renal DCs did not aggravate apoptosis or fibrosis. Multiphoton confocal in vivo microscopy was used to visualize renal DC behavior in real-time in disease for the first time. Live imaging of renal DCs in the normal kidney showed some probing of dendrites from otherwise stationary DCs, consistent with the previous study using this technique. The previous study used CX3CR1-GFP mice, in which 70% of GFP cells were identified as CD11c DCs. 4 Our studies used mice expressing EYFP under the control of the CD11c promoter, so that at least 95% of renal EYFP cells were DCs, providing increased specificity compared with the previous study. This study further extended these findings and provides the very first intravital images of renal DCs under disease conditions. Our studies revealed behavioral changes in DCs during inflammation, including increased cellular activity and an increased number of dendrites extending from the cells, consistent with the elevated activation status of these potent APCs. The numbers of monocytes, neutrophils, and CD8 T cells were increased in obstructed kidneys 3 days postoperatively. In this respect our findings were similar to those of Dong et al (2008); however, the current study did not find significant increases in the number of DCs in obstructed kidneys. 6 Absolute cell numbers in the current studies were determined by analyzing samples with a known number of calibration beads, added to whole digested kidneys. This allows accurate assessment of leukocyte numbers between normal and diseased states. It also controls for potential alterations in leukocyte isolation between normal (or sham operated) kidneys and UUO, as damaged tubular cells may not survive the isolation process and injured tubules may be more readily filtered out. Significant differences in DC surface markers were observed after UUO. Monocyte-derived inflammatory APCs exist in inflammatory lesions, 32 and phenotypic analysis of renal CD11c cells after obstruction provided evidence for the recruitment of immature, CD11b hi Gr-1 monocyte-derived inflammatory DCs with lower levels of CD11c migrating into the obstructed kidney. These finding support links between recruited monocytes, macrophages, and DCs, at least in terms of cell surface marker changes. Maturational changes were also identified on resident renal DCs, with loss of F4/80 expression and increased CD11b expression,
9 Dendritic Cells in Renal Fibrosis 99 Figure 5. The expression level of CD11c relates to differential expression of markers overlapping with the monocyte/macrophage lineage. Renal CD11c subsets were assessed by multicolor flow-cytometric analysis of single-cell kidney suspensions from mice at d3 UUO or sham-operated controls (n 5/group). A: Representative dot plots indicate the gating strategy used to identify CD11c expression: CD11c cells were further subdivided into three groups based on the surface expression of CD11c being low, intermediate or high (lo, int, or hi). Bar graph indicates the proportion of CD11c cells expressing the differential levels of CD11c for each group. B: Subdivided based on the level of CD11c expression, the proportion of each subset expressing MHC II, Gr-1, F4/80, or CD11b are compared between sham and d3 UUO. C: Representative histograms from a d3 UUO kidney shows the differential expression of MHC II, Gr-1, F4/80, and CD11b on cells that express CD11c at lo, int, or hi levels. D: The absolute number of DCs in the draining renal LN of sham operated and d3 UUO mice (n 4/group). *P 0.05; ***P (d3 UUO versus sham, proportion of CD11c hi DCs). consistent with their measured increase in function (antigen-specific T-cell responses). In addition, a higher proportion of F4/80- DCs made TNF after UUO, in contrast to that reported by Dong et al, 6 who found that, 24 hours after UUO, F4/80 cells were the major source of TNF. Although this study demonstrates inflammatory CD11c cells in the injured kidney, further experiments are required to conclusively differentiate between resident and immigrating DCs, to discern their relative contributions in the inflammatory process and to better define the plasticity that exists be-
10 100 Snelgrove et al Figure 6. A limited capacity of renal DCs to stimulate T-cell responses is enhanced after UUO. The ability of splenic versus kidney derived DCs to stimulate T-cell responses was tested ex vivo. DCs were isolated from the spleen or kidney and pulsed with OVA peptide before culture with naïve CFSE-labeled OT-I CD8 T cells for 60 hours. A: Comparison of the number of proliferating OT-I T cells (CD8 V 2 ) when stimulated by titrating numbers of splenic versus renal DCs. B: Proliferative T-cell response when cultured with 500 splenic versus 500 renal DCs and the effect of stimulating T cells with equal numbers of both groups of DCs (500 splenic plus 500 renal DCs). Results of A and B are expressed as the mean SEM of triplicate wells. C: Representative histogram profiles and the percentage of OT-I T cells that proliferated in response to stimulation by splenic versus renal versus d3 UUO renal DCs. D: Gives the mean number of proliferating OT-I T cells that have undergone 0 to 7 divisions when stimulated by splenic or renal DCs from healthy donors versus renal DCs from a d3 UUO kidney. Each well was performed in triplicate. E: Shows the experimental protocol used to measure the in vivo proliferation of antigen-specific OT-I T cells. RIPmOVA mice were injected i.v. with CFSE-labeled lymphocytes from OT-I donors. After transfer, the left ureter of recipient mice was surgically ligated (UUO, n 6), and control mice underwent sham operation (n 6). At day 3 after surgery, mice were sacrificed and the renal lymph nodes (UUO and CL) were collected for flow-cytometric analysis. F: Proliferating OT-I T cells were identified by the dilution of CFSE, and the absolute number of proliferating OT-I T cells per lymph node was quantitated by the incorporation of a known number of Calibrate microbeads per sample. *P 0.05; ***P tween monocytes/macrophages and DCs and the nature of the turnover that exists in these cells within the kidney. Antigen-specific activation of T cells is the fundamental function of DCs. Renal DCs from noninflamed kidneys were less efficient than splenic DCs in inducing antigen-specific proliferative syngeneic OVA-specific CD8 (OT-I) T cells, consistent with findings in an allogeneic system. 3 Adding renal DCs to splenic DCs did not inhibit T-cell proliferation. After UUO, a combination of ex vivo and in vivo approaches showed a marked increase in the capacity of DCs to induce antigen-specific T-cell proliferation. Ex vivo data showed that renal DCs were capable of inducing proliferation to levels similar to those caused by splenic DCs, although in vivo studies using CFSE-labeled naive OT-I T cells transferred into syngeneic RIP-mOVA mice (expressing membrane-bound OVA on renal tubules) showed a marked increase in antigen-specific cells in the draining lymph node after UUO. Therefore, after inflammation, renal DCs become more professional in their primary function, namely, that of activating antigen-specific T cells. These findings have implications for our understanding of allogeneic and protective immune responses, particularly as urinary tract obstruction can complicate renal transplantation, and urinary tract infection may occur in the presence of obstruction. We used CD11c-DTR mice to define the roles of DCs in inflammatory renal disease leading to fibrosis. DC depletion studies in glomerulonephritis induced by an adaptive immune responses to a foreign antigen show that initially, DCs were protective 7 but later in disease acquired a pathogenic role, 8 whereas in cisplatin-induced nephrotoxicity, DCs are protective. 9 Renal DCs are sources of TNF which can induce renal epithelial cell apoptosis, 6 and TNF neutralization attenuates both apoptosis and fibrosis. 33,34 In the current studies, there was no overall increase in the proportion of renal DCs with the capacity to make TNF, although increased proportions of the CD11c F4/80 subset produced TNF by day 3. However, no difference was found in key indices of injury after DC depletion, regardless of the time point at which that DCs were ablated. The extent of collagen accumulation, the number of apoptotic cells and the expression of -SMA were similar between groups. Although CD11c cell depletion is not complete in the DTR model, studies in other experimental renal diseases have found an effect after DC depletion, 7 9 indicating the degree
11 Dendritic Cells in Renal Fibrosis 101 Figure 8. DC depletion does not reduce the number of apoptosing cells in the obstructed kidney. The experimental protocol used to establish the role of DCs in the progression of kidney fibrosis is outlined in Figure 6A. The same kidney samples were stained with an anticleaved caspase-3 Ab to enumerate the number of apoptosing cells per kidney. Representative photomicrographs showing caspase-3 staining. UUO kidney of a control mouse (A), mouse depleted of DCs at day 0 (B) mouse depleted of DCs at day 2 (C), mouse depleted of DCs at day 4 (D) and the CL kidney of a control mouse (E) and a mouse depleted of DCs (F). G: The number of cells that stained for caspase-3 was manually counted (mean taken from 10 to 15 nonoverlapping high-power fields [HPF] of view; magnification, 400). For presentation purposes, results are collated on one graph in which controls (UUO with DCs, CL with DCs, and CL DC depleted) are combined from the individual experiments. Statistical analysis compared each experimental group to the control group from the same experiment. Data points represent individual mice. Figure 7. DC depletion at different time points does not attenuate collagen deposition in the obstructed kidney. A: Experimental protocol used to establish the role of DCs in the progression of kidney fibrosis. The left kidney of CD11cDTR mice (transgene negative or wild-type mice as controls) was obstructed on day 0. At day 0, 2 or 4, diphtheria toxin was administered to deplete DCs, and at day 7 kidneys were collected for histological analysis. Both the UUO and CL kidneys were collected for histological analysis. Representative photomicrographs of periodic acid- Schiff stained kidney sections of a CL kidney (B)and an obstructed kidney at day 7(C). Original magnification, 200. Representative photomicrographs showing picrosirius red staining for collagen fibers are shown. UUO kidney of a control mouse (D) mousedepletedofdcsatday0(e), mouse depleted of DCs at day 2 (F), mouse depleted of DCs at day 4 (G), and CL kidneys of a control mouse (H)and amousedepletedofdcs(i). J: Results are expressed as the percentage area of the kidney comprising collagen (mean taken from five to 10 nonoverlapping fields of view); magnification, 400. Depletion of DCs at each time point was performed as aseparateexperimentwithnondepletedcontrols.forpresentationpurposes,results are collated on one graph where controls (UUO with DCs, CL with DCs, and CL DC depleted) are combined from the individual experiments. Statistical analysis compared each experimental group to the control group from the same experiment. Data points represent individual mice. of depletion achieved by this model is biologically relevant. Nonetheless, it is possible that the remaining DCs in UUO may play a role in this condition. Plasmacytoid DCs, a minor population of renal DCs, may or may not be effectively depleted by DT, 35,36 and although they traditionally play a role in antiviral responses, it remains possible that pdcs may directly influence fibrosis. Our study primarily investigated the role of DCs in the early inflammatory events. Although we also examined the effect of DC depletion at day 7 on outcomes at day 14, it remains possible that DCs may play a significant functional role at a late stage of fibrosis. Previous studies that have used less specific depletion studies have also affected renal DCs. One used liposomal clodronate to deplete phagocytic cells (including F4/80
12 102 Snelgrove et al macrophages and DCs, and potentially neutrophils) after UUO, showing attenuated fibrosis and suggested a collective pro-inflammatory role for phagocytes. 37 Similar results are reported in studies that conditionally ablated CD11bexpressing cells, 38,39 which include renal macrophages and some DCs. However, these studies could not address arolefordcsintheprogressionoffibrosis,distinctfromthe contributions of macrophages (and potentially neutrophils). Therefore, the results of the current studies implicate macrophages more directly in the pathogenesis of renal fibrosis, although the diverse phenotype of tissue macrophages implies divergent roles for different macrophage subsets that may be time dependent. Most recently, CD11c-DTR chimeric mice (to allow repeated administration of DT) were depleted of DCs 17,39 before a 5-day model of UUO, with no effect on collagen accumulation. 39 In summary, in the context of interstitial inflammation, renal DCs are activated and function as more potent APCs and better stimulate T-cell responses. Their pro-inflammatory functions are related largely to their classical role as activators of T cells, and they do not directly contribute to Figure 9. Myofibroblast formation is not inhibited by the depletion of DCs during UUO. The experimental protocol used to establish the role of DCs in the progression of kidney fibrosis is outlined in Figure 7A. Kidney samples were also stained with anti- SMA a marker for myofibroblasts. Representative photomicrographs showing SMA staining. UUO kidney of a control mouse (A), mouse depleted of DCs at day 0 (B), mouse depleted of DCs at day 2 (C), mouse depleted of DCs at day 4 (D), and CL kidney of a control mouse (E) and a mouse depleted of DCs (F). G: Results are expressed as the percentage area of the kidney comprising -SMA as determined using Meta- Morph (mean taken from five to 10 nonoverlapping fields of view; magnification 400). For presentation purposes, results are collated on one graph in which controls (UUO with DCs, CL with DCs, and CL DC depleted) are combined from the individual experiments. Statistical analysis compared each experimental group to the control group from the same experiment. Data points represent individual mice. Figure 10. The effect of DC depletion 7 days after UUO on collagen deposition, myofibroblast accumulation, and apoptosis at day 14. A: Experimental protocol used to establish the role of DCs in the progression of kidney fibrosis. The left kidney of CD11cDTR mice (n 6, transgene negative or wild-type mice as controls, n 6) was obstructed on day 0. At day 7, diphtheria toxin was administered to deplete the DCs and kidneys were collected for histological analysis on day 14. Both the UUO and CL kidneys were collected for histological analysis. Representative photomicrographs showing picrosirius red staining for collagen fibers are shown, with (B) a UUO kidney of a control mouse at day 14 and (C) a mouse depleted of DCs at day 7, studied at day 14. There was no significant difference in collagen accumulation (D), the number of cleaved-caspase-3 apoptotic cells, expressed as cells per high-power field (HPF; E) or myofibroblast accumulation (F) after DC depletion. Results are expressed as the percentage area of the kidney comprising collagen or -SMA (mean taken from five to 10 nonoverlapping fields of view; magnification 400) and for caspase-3 the number of positive cells in 10 fields. Data points represent individual mice.
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supporting Information
 Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
Canberra, Australia). CD11c-DTR-OVA-GFP (B6.CD11c-OVA), B6.luc + and. Cancer Research Center, Germany). B6 or BALB/c.FoxP3-DTR-GFP mice were
 Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Hua Tang, Weiping Cao, Sudhir Pai Kasturi, Rajesh Ravindran, Helder I Nakaya, Kousik
 SUPPLEMENTARY FIGURES 1-19 T H 2 response to cysteine-proteases requires dendritic cell-basophil cooperation via ROS mediated signaling Hua Tang, Weiping Cao, Sudhir Pai Kasturi, Rajesh Ravindran, Helder
SUPPLEMENTARY FIGURES 1-19 T H 2 response to cysteine-proteases requires dendritic cell-basophil cooperation via ROS mediated signaling Hua Tang, Weiping Cao, Sudhir Pai Kasturi, Rajesh Ravindran, Helder
Supporting Information
 Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supplemental Table 1. Primer sequences for transcript analysis
 Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
B220 CD4 CD8. Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN
 B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
B6/COLODR/SPL/11C/83/LAP/#2.006 B6/COLODR/SPL/11C/86/LAP/#2.016 CD11C B6/COLODR/SPL/11C/80/LAP/#2.011 CD11C
 CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
Figure S1 Generation of γ-gt DTR transgenic mice. (A) Schematic construct of the transgene. (B)
 Figure S1 Generation of γ-gt DTR transgenic mice. (A) Schematic construct of the transgene. (B) PCR identified expected hhb-egf band (left panel) and HA tag band (right) in kidneys of transgenic (TG) mice
Figure S1 Generation of γ-gt DTR transgenic mice. (A) Schematic construct of the transgene. (B) PCR identified expected hhb-egf band (left panel) and HA tag band (right) in kidneys of transgenic (TG) mice
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice
 Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
As outlined under External contributions (see appendix 7.1), the group of Prof. Gröne at the
 3 RESULTS As outlined under External contributions (see appendix 7.1), the group of Prof. Gröne at the DKFZ in Heidelberg (Dept. of Cellular and Molecular pathology) contributed to this work by performing
3 RESULTS As outlined under External contributions (see appendix 7.1), the group of Prof. Gröne at the DKFZ in Heidelberg (Dept. of Cellular and Molecular pathology) contributed to this work by performing
activation with anti-cd3/cd28 beads and 3d following transduction. Supplemental Figure 2 shows
 Supplemental Data Supplemental Figure 1 compares CXCR4 expression in untreated CD8 + T cells, following activation with anti-cd3/cd28 beads and 3d following transduction. Supplemental Figure 2 shows the
Supplemental Data Supplemental Figure 1 compares CXCR4 expression in untreated CD8 + T cells, following activation with anti-cd3/cd28 beads and 3d following transduction. Supplemental Figure 2 shows the
Eosinophils are required. for the maintenance of plasma cells in the bone marrow
 Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Nature Immunology: doi: /ni Supplementary Figure 1. Examples of staining for each antibody used for the mass cytometry analysis.
 Supplementary Figure 1 Examples of staining for each antibody used for the mass cytometry analysis. To illustrate the functionality of each antibody probe, representative plots illustrating the expected
Supplementary Figure 1 Examples of staining for each antibody used for the mass cytometry analysis. To illustrate the functionality of each antibody probe, representative plots illustrating the expected
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and
 Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary information
 Supplementary information Intrahepatic myeloid cell-aggregates enable local CD8 + T cell expansion and successful immunotherapy against chronic viral liver infection Li- Rung Huang, Dirk Wohlleber, Florian
Supplementary information Intrahepatic myeloid cell-aggregates enable local CD8 + T cell expansion and successful immunotherapy against chronic viral liver infection Li- Rung Huang, Dirk Wohlleber, Florian
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
SUPPLEMENTARY FIGURE 1
 SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Combined Rho-kinase inhibition and immunogenic cell death triggers and propagates immunity against cancer
 Supplementary Information Combined Rho-kinase inhibition and immunogenic cell death triggers and propagates immunity against cancer Gi-Hoon Nam, Eun-Jung Lee, Yoon Kyoung Kim, Yeonsun Hong, Yoonjeong Choi,
Supplementary Information Combined Rho-kinase inhibition and immunogenic cell death triggers and propagates immunity against cancer Gi-Hoon Nam, Eun-Jung Lee, Yoon Kyoung Kim, Yeonsun Hong, Yoonjeong Choi,
Supplemental Information. Human CD1c + Dendritic Cells Drive. the Differentiation of CD103 + CD8 + Mucosal Effector T Cells via the Cytokine TGF-
 Immunity, Volume 38 Supplemental Information Human CD1c + Dendritic Cells Drive the Differentiation of CD103 + CD8 + Mucosal Effector T Cells via the Cytokine TGF- Chun I. Yu Christian Becker Yuanyuan
Immunity, Volume 38 Supplemental Information Human CD1c + Dendritic Cells Drive the Differentiation of CD103 + CD8 + Mucosal Effector T Cells via the Cytokine TGF- Chun I. Yu Christian Becker Yuanyuan
Supplementary Information. Tissue-wide immunity against Leishmania. through collective production of nitric oxide
 Supplementary Information Tissue-wide immunity against Leishmania through collective production of nitric oxide Romain Olekhnovitch, Bernhard Ryffel, Andreas J. Müller and Philippe Bousso Supplementary
Supplementary Information Tissue-wide immunity against Leishmania through collective production of nitric oxide Romain Olekhnovitch, Bernhard Ryffel, Andreas J. Müller and Philippe Bousso Supplementary
CD14 + S100A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer
 CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Pearson r = P (one-tailed) = n = 9
 8F4-Specific Lysis, % 1 UPN1 UPN3 8 UPN7 6 Pearson r =.69 UPN2 UPN5 P (one-tailed) =.192 4 UPN8 n = 9 2 UPN9 UPN4 UPN6 5 1 15 2 25 8 8F4, % Max MFI Supplementary Figure S1. AML samples UPN1-UPN9 show variable
8F4-Specific Lysis, % 1 UPN1 UPN3 8 UPN7 6 Pearson r =.69 UPN2 UPN5 P (one-tailed) =.192 4 UPN8 n = 9 2 UPN9 UPN4 UPN6 5 1 15 2 25 8 8F4, % Max MFI Supplementary Figure S1. AML samples UPN1-UPN9 show variable
Measuring Dendritic Cells
 Measuring Dendritic Cells A.D. Donnenberg, V.S. Donnenberg UNIVERSITY of PITTSBURGH CANCER INSTITUTE CCS Longbeach 10_04 Measuring DC Rare event detection The basics of DC measurement Applications Cancer
Measuring Dendritic Cells A.D. Donnenberg, V.S. Donnenberg UNIVERSITY of PITTSBURGH CANCER INSTITUTE CCS Longbeach 10_04 Measuring DC Rare event detection The basics of DC measurement Applications Cancer
NK cell flow cytometric assay In vivo DC viability and migration assay
 NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
Supplementary Figure 1. Example of gating strategy
 Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs
 Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
DC were seeded into tissue culture dishes in IMDM 2% FCS, and added with PMN. (1:1; PMN: DC) for 16h also in the presence of DNAse (100 U/ml); DC were
 Supplementary methods Flow cytometric analysis of DCs. DC were seeded into tissue culture dishes in IMDM 2% FCS, and added with PMN (1:1; PMN: DC) for 16h also in the presence of DNAse (100 U/ml); DC were
Supplementary methods Flow cytometric analysis of DCs. DC were seeded into tissue culture dishes in IMDM 2% FCS, and added with PMN (1:1; PMN: DC) for 16h also in the presence of DNAse (100 U/ml); DC were
Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation
 SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
BMDCs were generated in vitro from bone marrow cells cultured in 10 % RPMI supplemented
 Supplemental Materials Figure S1. Cultured BMDCs express CD11c BMDCs were generated in vitro from bone marrow cells cultured in 10 % RPMI supplemented with 15 ng/ml GM-CSF. Media was changed and fresh
Supplemental Materials Figure S1. Cultured BMDCs express CD11c BMDCs were generated in vitro from bone marrow cells cultured in 10 % RPMI supplemented with 15 ng/ml GM-CSF. Media was changed and fresh
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Fig. S1 A. week 4 week 6
 Fig. S1 Trabecular Number Trabecular Thickness number/mm 3.5 3. 2.5 2. 1.5 1..5 mm.45.4.35.3.25.2.15.1.5 SKG-c SKG-A mm 1.4 1.2 1..8.6.4.2 Trabecular Spacing D. week 4 week 6 Figure S1. MicroCT analysis
Fig. S1 Trabecular Number Trabecular Thickness number/mm 3.5 3. 2.5 2. 1.5 1..5 mm.45.4.35.3.25.2.15.1.5 SKG-c SKG-A mm 1.4 1.2 1..8.6.4.2 Trabecular Spacing D. week 4 week 6 Figure S1. MicroCT analysis
Multi-Parameter Apoptosis Assay Kit
 Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
were isolated from the freshly drawn blood of healthy donors and ACS patients using the
 Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
January 25, 2017 Scientific Research Process Name of Journal: ESPS Manuscript NO: Manuscript Type: Title: Authors: Correspondence to
 January 25, 2017 Scientific Research Process Name of Journal: World Journal of Gastroenterology ESPS Manuscript NO: 31928 Manuscript Type: ORIGINAL ARTICLE Title: Thiopurine use associated with reduced
January 25, 2017 Scientific Research Process Name of Journal: World Journal of Gastroenterology ESPS Manuscript NO: 31928 Manuscript Type: ORIGINAL ARTICLE Title: Thiopurine use associated with reduced
Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic
 Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic Reticulum Stress Lokesh Makhija, BE, Veda Krishnan, MSc, Rakhshinda Rehman, MTech, Samarpana Chakraborty, MSc, Shuvadeep Maity,
Chemical Chaperones Mitigate Experimental Asthma By Attenuating Endoplasmic Reticulum Stress Lokesh Makhija, BE, Veda Krishnan, MSc, Rakhshinda Rehman, MTech, Samarpana Chakraborty, MSc, Shuvadeep Maity,
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1 Protease allergens induce IgE and IgG1 production. (a-c)
 1 Supplementary Figure 1 Protease allergens induce IgE and IgG1 production. (a-c) Serum IgG1 (a), IgM (b) and IgG2 (c) concentrations in response to papain immediately before primary immunization (day
1 Supplementary Figure 1 Protease allergens induce IgE and IgG1 production. (a-c) Serum IgG1 (a), IgM (b) and IgG2 (c) concentrations in response to papain immediately before primary immunization (day
L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity
 Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection
 Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY
 ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
Supplementary Information:
 Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Interferon γ regulates idiopathic pneumonia syndrome, a. Th17 + CD4 + T-cell-mediated GvH disease
 Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Supplementary Figure 1 Lymphocytes can be tracked for at least 4 weeks after
 Supplementary Figure 1 Lymphocytes can be tracked for at least 4 weeks after photoconversion by using H2B-Dendra2. 4-5 PPs of H2B-Dendra2 BM chimeras were photoconverted and analyzed 7 days (upper panel)
Supplementary Figure 1 Lymphocytes can be tracked for at least 4 weeks after photoconversion by using H2B-Dendra2. 4-5 PPs of H2B-Dendra2 BM chimeras were photoconverted and analyzed 7 days (upper panel)
Supplemental Information. Aryl Hydrocarbon Receptor Controls. Monocyte Differentiation. into Dendritic Cells versus Macrophages
 Immunity, Volume 47 Supplemental Information Aryl Hydrocarbon Receptor Controls Monocyte Differentiation into Dendritic Cells versus Macrophages Christel Goudot, Alice Coillard, Alexandra-Chloé Villani,
Immunity, Volume 47 Supplemental Information Aryl Hydrocarbon Receptor Controls Monocyte Differentiation into Dendritic Cells versus Macrophages Christel Goudot, Alice Coillard, Alexandra-Chloé Villani,
Supplementary Figure 1. Nature Neuroscience: doi: /nn.4547
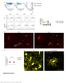 Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Role of Tyk-2 in Th9 and Th17 cells in allergic asthma
 Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Characterization of the effect of LPS on dendritic cell subset discrimination in spleen
 Short Communication J. Cell. Mol. Med. Vol 18, No 9, 2014 pp. 1908-1912 Characterization of the effect of LPS on dendritic cell subset discrimination in spleen Kristin L Griffiths, Jonathan KH Tan, Helen
Short Communication J. Cell. Mol. Med. Vol 18, No 9, 2014 pp. 1908-1912 Characterization of the effect of LPS on dendritic cell subset discrimination in spleen Kristin L Griffiths, Jonathan KH Tan, Helen
MACROPHAGE "MONOCYTES" SURFACE RECEPTORS
 LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
SUPPLEMENTARY METHODS
 SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
PBS Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs , 27-30
 PBS 803 - Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs. 15-25, 27-30 Learning Objectives Compare and contrast the maturation of B and T lymphocytes Compare and contrast
PBS 803 - Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs. 15-25, 27-30 Learning Objectives Compare and contrast the maturation of B and T lymphocytes Compare and contrast
Jean Wu,* Cindy Zhou,* Julie Robertson,* Connie C. Y. Weng, Marvin L. Meistrich, Ramesh C. Tailor, and Ya-Huan Lou*,1
 Article Identification of a bone marrow-derived CD8 dendritic cell-like population in inflamed autoimmune target tissue with capability of inducing T cell apoptosis Jean Wu,* Cindy Zhou,* Julie Robertson,*
Article Identification of a bone marrow-derived CD8 dendritic cell-like population in inflamed autoimmune target tissue with capability of inducing T cell apoptosis Jean Wu,* Cindy Zhou,* Julie Robertson,*
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured
 Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
Dendritic cell subsets and CD4 T cell immunity in Melanoma. Ben Wylie 1 st year PhD Candidate
 Dendritic cell subsets and CD4 T cell immunity in Melanoma Ben Wylie 1 st year PhD Candidate Melanoma Melanoma is the 4 th most common cancer in Australia. Current treatment options are ineffective resulting
Dendritic cell subsets and CD4 T cell immunity in Melanoma Ben Wylie 1 st year PhD Candidate Melanoma Melanoma is the 4 th most common cancer in Australia. Current treatment options are ineffective resulting
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
effect on the upregulation of these cell surface markers. The mean peak fluorescence intensity
 SUPPLEMENTARY FIGURE 1 Supplementary Figure 1 ASIC1 disruption or blockade does not effect in vitro and in vivo antigen-presenting cell activation. (a) Flow cytometric analysis of cell surface molecules
SUPPLEMENTARY FIGURE 1 Supplementary Figure 1 ASIC1 disruption or blockade does not effect in vitro and in vivo antigen-presenting cell activation. (a) Flow cytometric analysis of cell surface molecules
Downregulation of angiotensin type 1 receptor and nuclear factor-κb. by sirtuin 1 contributes to renoprotection in unilateral ureteral
 Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
Supplementary Figure 1 Chemokine and chemokine receptor expression during muscle regeneration (a) Analysis of CR3CR1 mrna expression by real time-pcr
 Supplementary Figure 1 Chemokine and chemokine receptor expression during muscle regeneration (a) Analysis of CR3CR1 mrna expression by real time-pcr at day 0, 1, 4, 10 and 21 post- muscle injury. (b)
Supplementary Figure 1 Chemokine and chemokine receptor expression during muscle regeneration (a) Analysis of CR3CR1 mrna expression by real time-pcr at day 0, 1, 4, 10 and 21 post- muscle injury. (b)
LPS CD40 + IL-4. Vorinostat (24 Hours) Vorinostat (24 Hours) Panobinostat (24 Hours) Panobinostat (24 Hours) Romidepsin (48 Hours)
 A) CD + IL- B) LPS ( Hours) ( Hours) Cell number (x1-3 ) 1 1 3.7 M 1. M. M.1 M Cell number (x1 - ) 1 1 3. M 1. M.7 M.38 M Cell number (x1-3 ) Cell number (x1-3 ) 3 1 1 1 ( Hours) 7.nM.nM 1.7nM.nM Romidepsin
A) CD + IL- B) LPS ( Hours) ( Hours) Cell number (x1-3 ) 1 1 3.7 M 1. M. M.1 M Cell number (x1 - ) 1 1 3. M 1. M.7 M.38 M Cell number (x1-3 ) Cell number (x1-3 ) 3 1 1 1 ( Hours) 7.nM.nM 1.7nM.nM Romidepsin
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with
 Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or
 Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Supplemental Information. CD4 + CD25 + Foxp3 + Regulatory T Cells Promote. Th17 Cells In Vitro and Enhance Host Resistance
 Immunity, Volume 34 Supplemental Information D4 + D25 + + Regulatory T ells Promote Th17 ells In Vitro and Enhance Host Resistance in Mouse andida albicans Th17 ell Infection Model Pushpa Pandiyan, Heather
Immunity, Volume 34 Supplemental Information D4 + D25 + + Regulatory T ells Promote Th17 ells In Vitro and Enhance Host Resistance in Mouse andida albicans Th17 ell Infection Model Pushpa Pandiyan, Heather
Bead Based Assays for Cytokine Detection
 Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
pro-b large pre-b small pre-b CCCP (µm) Rag1 -/- ;33.C9HCki
 a TMRM FI (Median) b TMRM FI (Median) c 20 15 10 5 0 8 6 4 2 0 pro-b large pre-b small pre-b 0 10 20 30 40 50 60 70 80 90 100 TMRM (nm) pro-b large pre-b small pre-b 0 1 2 4 8 16 32 64 128 256 CCCP (mm)
a TMRM FI (Median) b TMRM FI (Median) c 20 15 10 5 0 8 6 4 2 0 pro-b large pre-b small pre-b 0 10 20 30 40 50 60 70 80 90 100 TMRM (nm) pro-b large pre-b small pre-b 0 1 2 4 8 16 32 64 128 256 CCCP (mm)
Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell?
 Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Assays for Immunology Research IncuCyte S3 Live-Cell Analysis System
 ssays for Immunology Research IncuCyte S3 Live-Cell nalysis System Immune system characterization of health and disease states requires quantitative data from dynamic experimental models of the cells that
ssays for Immunology Research IncuCyte S3 Live-Cell nalysis System Immune system characterization of health and disease states requires quantitative data from dynamic experimental models of the cells that
Cytokine Complex Expanded Natural Killer Cells Improve Allogeneic Lung. Transplant Function via Depletion of Donor Dendritic Cells
 Cytokine Complex Expanded Natural Killer Cells Improve Allogeneic Lung Transplant Function via Depletion of Donor Dendritic Cells Wolfgang Jungraithmayr, Laura Codarri, Gregory Bouchaud,Carsten Krieg,
Cytokine Complex Expanded Natural Killer Cells Improve Allogeneic Lung Transplant Function via Depletion of Donor Dendritic Cells Wolfgang Jungraithmayr, Laura Codarri, Gregory Bouchaud,Carsten Krieg,
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4
 Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
The Annexin V Apoptosis Assay
 The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
renoprotection therapy goals 208, 209
 Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
Subject Index Aldosterone, plasminogen activator inhibitor-1 induction 163, 164, 168 Aminopeptidases angiotensin II processing 64 66, 214 diabetic expression 214, 215 Angiotensin I intrarenal compartmentalization
% of live splenocytes. STAT5 deletion. (open shapes) % ROSA + % floxed
 Supp. Figure 1. a 14 1 1 8 6 spleen cells (x1 6 ) 16 % of live splenocytes 5 4 3 1 % of live splenocytes 8 6 4 b 1 1 c % of CD11c + splenocytes (closed shapes) 8 6 4 8 6 4 % ROSA + (open shapes) % floxed
Supp. Figure 1. a 14 1 1 8 6 spleen cells (x1 6 ) 16 % of live splenocytes 5 4 3 1 % of live splenocytes 8 6 4 b 1 1 c % of CD11c + splenocytes (closed shapes) 8 6 4 8 6 4 % ROSA + (open shapes) % floxed
Rapid antigen-specific T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Supplementary Information. A vital role for IL-2 trans-presentation in DC-mediated T cell activation in humans as revealed by daclizumab therapy
 Supplementary Information A vital role for IL-2 trans-presentation in DC-mediated T cell activation in humans as revealed by daclizumab therapy Simone C. Wuest 1, Jehad Edwan 1, Jayne F. Martin 1, Sungpil
Supplementary Information A vital role for IL-2 trans-presentation in DC-mediated T cell activation in humans as revealed by daclizumab therapy Simone C. Wuest 1, Jehad Edwan 1, Jayne F. Martin 1, Sungpil
The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF
 CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
- 1 - Cell types Monocytes THP-1 cells Macrophages. LPS Treatment time (Hour) IL-6 level (pg/ml)
 Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
