STRUCTURE OF A UBIQUITIN-LOADED HECT LIGASE REVEALS THE MOLECULAR BASIS FOR CATALYTIC PRIMING SUPPLEMENTARY INFORMATION
|
|
|
- Alison Copeland
- 5 years ago
- Views:
Transcription
1 STRUCTURE OF A UBIQUITINLOADED ECT LIGASE REVEALS TE MOLECULAR BASIS FOR CATALYTIC PRIMING Elena Maspero 1, Eleonora Valentini 1, Sara Mari 1, Valentina Cecatiello 2, Paolo Soffientini 1, Sebastiano Pasqualato 2 and Simona Polo 1,3 1 Fondazione Istituto FIRC di Oncologia Molecolare, Milan, Italy. 2 Crystallography Unit, Department of Experimental Oncology, European Institute of Oncology, Milan, Italy 3 Dipartimento di Scienze della Salute, Universita degli Studi di Milano, Milan, Italy. SUPPLEMENTARY INFORMATION Short title: E3iquitin Thioester Intermediate Structure
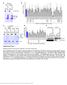
2 a b R74 c P P 7 7N 7N ut ST T 707A627AC62 C62 C62 p In G W F C Δ Δ IΔ R74 G G G76 C867 C867 E3 Thioester Bond E3 IB: Disulfide Bond time (h): ECT~ECT C76 d A280nm ECT~ ECT %B ECT~ ECT coomassie Elution volume (ml) fractions: D3 D2 D1 E1 E2 E4 E6 E8 E10 E11E12F12 F11 C lobe C lobe Cys76 C Cys867 D Cys627 Ile626 Asn627 Asn Glu629 C lobe g f D C lobe N 200 ECT~ e 10 Leu626 Pro628 Ser777 Cys778
3 Supplementary Figure 1 Preparation of ECT Nedd4 ~ D : complex. (a) Schematic drawings of the disulfide bond present in the crystal structure and the naturally occurring thioester bond. NMR studies have shown that the disulfide bond nicely mimics the native thioester bond, most likely thanks to the flexibility of Gly76 in the thioester, able to compensate for the extra bond length distance present in the SS bridge 1. (b) To favor the catalytic Cys867 for disulfide bond formation with G76C the two other ECT cysteine residues (Cys627 and Cys778) were either deleted or mutated. Mutants were tested for their ability to bind by GST pulldown assay. The indicated GSTfusion proteins were incubated for 2 hr at 4 C in YY buffer ( mm NaEPES p 7.5, 1 mm NaCl, 1 mm EDTA, 1 mm EGTA, 10% glycerol, 1% triton X100) with synthetic K63poly chains and analyzed by IB as indicated. Cys627 mutation/deletion showed impairment in the binding ability. We thus decided to substitute the residues surrounding this cysteine with the ones of Nedd4 budding yeast counterpart, Rsp5, which naturally doesn t possess it (Supplementary Figure 3), generating the ECT I CNP mutant that behaves as wildtype ECT. Coomassie staining shows comparable loading of GST proteins. (c) Disulfide bond formation between ECT I CNP and G76C was monitored using nonreducing SDSPAGE. At 48h the reaction was stopped due to monomer depletion. (d) Chromatogram from anionic exchange, showing separation of ECT I CNP ~ from ECT I CNP dimer, monomer and dimer. Lower panel shows fractions from the peak containing ECT I CNP ~, loaded on nonreducing SDSPAGE. Fractions from E1 to E11 were collected and concentrated for crystallization studies. (e) Comparison between ECT Nedd4 ~ D : and ECT Nedd4 :. Cartoon representation of the superposition of ECT Nedd4 ~ D : with ECT Nedd4 : (pdb 2xbb). The structures have been superposed through their Nlobe domains. ECT Nedd4 ~ D : is represented in the same colors as in Figure 1. The N and Clobe of ECT Nedd4 : are in dark blue and light green, respectively, whilst is in brown. (f) Same as right panel of e, but the Clobes and D are not shown, to permit a clearer view of the Nlobes and superposition. The Nlobe adopts the same conformation in the presence or absence of D (rootmeansquare deviation of 0.8 Å over 8 C ) with free kept in the binding site crafted by the Nlobe subdomains. (g) Insets display in ballandstick representation the sequences that have been mutated to generate the ECT Nedd4 ~ D : complex. Top panel highlights the three residues stretch (IleGlnPro) introduced at the place of the wild type four residues sequence (LeuCysGlnGlu) around Cys627. Bottom panel shows the Cys778 to Ser mutation.
4 a b ECTNedd4: time (sec): 120 WT Y616D Y616A Clobe D coomassie ECTNedd4: time (sec): 120 Nlobe E3~ WT R832G R832A C76 T830 C867 L841 L779 R832 E774 K598 M M835 N615 P834 Y616 Y Y610 c coomassie ECTNedd4: Y616A Y616D WT R832A R832G e2d3: time (sec): Y842 E3~ IB: coomassie Supplementary Figure 2 The Tyrlocked conformation of ECTNedd4~D: complex. (a) Details of the interactions at the interface between the ECT Clobe (green) and Nlobe (blue). Residues buried or participating at the interface are drawn in ballandstick representation and labeled. ydrogen bonds are highlighted with red dashed lines. (b) E2toECTNedd4 transthiolation assay. Upper panel, streptavidin blot. Thioester ECT~ is indicated. Lower panel, Coomassie stainig showing comparable loading of the different ECT mutants tested. (c) chain formation assay. Upper panel, kinetics of free chains formation with wildtype ECT and the indicated mutants. Lower panel, Coomassie staining showing comparable loading of proteins. Point mutations of Tyr616 to Asp or of Arg832 to Glu impair chain formation as a consequence of compromised E2 to E3 transfer. Tyr616 to Ala mutant shows reduced transthiolation, but unaffected chain formation. Less drastic mutations of Arg832 in Ala or in Gly are capable of transthiolation at the same extent of the Y616A mutant, show significantly reduced kinetic of chain formation and processivity. Nature Structural & Molecular Biology: doi: /nsmb.66
5 a snedd4_p46934 snedd4like_q69pu5 ScRsp5_P39940 sitch_q96j02 ssmurf1_q9ce7 ssmurf2_q9au4 swwp1_q90m0 swwp2_o00308 secw1_q76n89 secw2_q9p2p RDYKRKYEFFRRKLKKQNDIPNKFEMKLRRATVLEDSYRRIMGVKRADFLKARLWIEFDGEKGLDYGGVAREWFFLISKEMFNPYYGLFEYSATDNYTLQINPNSGLC REFKQKYDYFRKKLKKPADIPNRFEMKLRNNIFEESYRRIMSVKRPDVLKARLWIEFESEKGLDYGGVAREWFFLLSKEMFNPYYGLFEYSATDNYTLQINPNSGLC RDFRRKVIYFRSQPALRILPGQCIKVRRKNIFEDAYQEIMRQTPEDLKKRLMIKFDGEEGLDYGGVSREFFFLLSEMFNPFYCLFEYSAYDNYTIQINPNSGI RDFKAKVQYFRFWCQQLAMPQIKITVTRKTLFEDSFQQIMSFSPQDLRRRLWVIFPGEEGLDYGGVAREWFFLLSEVLNPMYCLFEYAGKDNYCLQINPASYI RDLVQKLKVLRELSLQQPQAGCRIEVSREEIFEESYRQIMKMRPKDLKKRLMVKFRGEEGLDYGGVAREWLYLLCEMLNPYYGLFQYSTDNIYMLQINPDSSI RDLVQKLKILRQELSQQQPQAGCRIEVSREEIFEESYRQVMKMRPKDLWKRLMIKFRGEEGLDYGGVAREWLYLLSEMLNPYYGLFQYSRDDIYTLQINPDSAV RGFRWKLAFRYLCQSNALPSVKINVSRQTLFEDSFQQIMALKPYDLRRRLYVIFRGEEGLDYGGLAREWFFLLSEVLNPMYCLFEYAGKNNYCLQINPASTI RSFRWKYQFRFLCSNALPSVKISVSRQTLFEDSFQQIMNMKPYDLRRRLYIIMRGEEGLDYGGIAREWFFLLSEVLNPMYCLFEYAGKNNYCLQINPASSI RDFEAKLRNFYRKLEAKGFGQGPGKIKLIIRRDLLEGTFNQVMAYSRKELQRNKLYVTFVGEEGLDYSGPSREFFFLLSQELFNPYYGLFEYSANDTYTVQISPMSAF RDFEAKLRNFYRKLETKGYGQGPGKLKLIIRRDLLEDAFNQIMGYSRKDLQRNKLYVTFVGEEGLDYSGPSREFFFLVSRELFNPYYGLFEYSANDTYTVQISPMSAF snedd4_p46934 snedd4like_q69pu5 ScRsp5_P39940 sitch_q96j02 ssmurf1_q9ce7 ssmurf2_q9au4 swwp1_q90m0 swwp2_o00308 secw1_q76n89 secw2_q9p2p5 NEDLSYFKFIGRVAGMAVYGKLLDGFFIRPFYKMMLKPITLDMESVDSEYYNSLRWILENDPTELDLRFIIDEELFGQTQELKNGGSEIVVTNKNKKEYIYL NEDLSYFTFIGRVAGLAVFGKLLDGFFIRPFYKMMLGKQITLNDMESVDSEYYNSLKWILENDPTELDLMFCIDEENFGQTYQVDLKPNGSEIMVTNENKREYIDL NPELNYFKFIGRVVGLGVFRRFLDAFFVGALYKMMLRKKVVLQDMEGVDAEVYNSLNWMLENSIDGVLDLTFSADDERFGEVVTVDLKPDGRNIEVTDGNKKEYVEL NPDLKYFRFIGRFIAMALFGKFIDTGFSLPFYKRILNKPVGLKDLESIDPEFYNSLIWVKENNIEECDLEMYFSVDKEILGEIKSDLKPNGGNILVTEENKEEYIRM NPDLSYFFVGRIMGLAVFGYINGGFTVPFYKQLLGKPIQLSDLESVDPELKSLVWILENDITPVLDTFCVENAFGRILQELKPNGRNVPVTEENKKEYVRL NPELSYFFVGRIMGMAVFGYIDGGFTLPFYKQLLGKSITLDDMELVDPDLNSLVWILENDITGVLDTFCVENAYGEIIQELKPNGKSIPVNEENKKEYVRL NPDLSYFCFIGRFIAMALFGKFIDTGFSLPFYKRMLSKKLTIKDLESIDTEFYNSLIWIRDNNIEECGLEMYFSVDMEILGKVTSDLKLGGSNILVTEENKDEYIGL NPDLTYFRFIGRFIAMALYGKFIDTGFTLPFYKRMLNKRPTLKDLESIDPEFYNSIVWIKENNLEECGLELYFIQDMEILGKVTTELKEGGESIRVTEENKEEYIML VENLEWFRFSGRILGLALIQYLLDAFFTRPFYKALLRLPCDLSDLEYLDEEFQSLQWMKDNNITDILDLTFTVNEEVFGQVTERELKSGGANTQVTEKNKKEYIER VDNEWFRFSGRILGLALIQYLLDAFFTRPFYKALLRILCDLSDLEYLDEEFQSLQWMKDNDIDILDLTFTVNEEVFGQITERELKPGGANIPVTEKNKKEYIER snedd4_p46934 snedd4like_q69pu5 ScRsp5_P39940 sitch_q96j02 ssmurf1_q9ce7 ssmurf2_q9au4 swwp1_q90m0 swwp2_o00308 secw1_q76n89 secw2_q9p2p VIQWRFVNRIQKQMAAFKEGFFELIPQDLIKIFDENELELLMCGLGDVDVNDWRETKYKNGYSANQVIQWFWKAVLMMDSEKRIRLLQFVTGTSRVPMNGFAELYGSN VIQWRFVNRVQKQMNAFLEGFTELLPIDLIKIFDENELELLMCGLGDVDVNDWRQSIYKNGYCPNPVIQWFWKAVLLMDAEKRIRLLQFVTGTSRVPMNGFAELYGSN YTQWRIVDRVQEQFKAFMDGFNELIPEDLVTVFDERELELLIGGIAEIDIEDWKKTDYRGYQESDEVIQWFWKCVSEWDNEQRARLLQFTTGTSRIPVNGFKDLQGSD VAEWRLSRGVEEQTQAFFEGFNEILPQQYLQYFDAKELEVLLCGMQEIDLNDWQRAIYRYARTSKQIMWFWQFVKEIDNEKRMRLLQFVTGTCRLPVGGFADLMGSN YVNWRFMRGIEAQFLALQKGFNELIPQLLKPFDQKELELIIGGLDKIDLNDWKSNTRLKCVADSNIVRWFWQAVETFDEERRARLLQFVTGSTRVPLQGFKALQGST YVNWRFLRGIEAQFLALQKGFNEVIPQLLKTFDEKELELIICGLGKIDVNDWKVNTRLKCTPDSNIVKWFWKAVEFFDEERRARLLQFVTGSSRVPLQGFKALQGAA MTEWRFSRGVQEQTKAFLDGFNEVVPLQWLQYFDEKELEVMLCGMQEVDLADWQRNTVYRYTRNSKQIIWFWQFVKETDNEVRMRLLQFVTGTCRLPLGGFAELMGSN LTDWRFTRGVEEQTKAFLDGFNEVAPLEWLRYFDEKELELMLCGMQEIDMSDWQKSTIYRYTKNSKQIQWFWQVVKEMDNEKRIRLLQFVTGTCRLPVGGFAELIGSN MVKWRVERGVVQQTEALVRGFYEVVDSRLVSVFDARELELVIAGTAEIDLNDWRNNTEYRGGYDGLVIRWFWAAVERFNNEQRLRLLQFVTGTSSVPYEGFAALRGSN MVKWRIERGVVQQTESLVRGFYEVVDARLVSVFDARELELVIAGTAEIDLSDWRNNTEYRGGYDNIVIRWFWAAVERFNNEQRLRLLQFVTGTSSIPYEGFASLRGSN snedd4_p46934 snedd4like_q69pu5 ScRsp5_P39940 sitch_q96j02 ssmurf1_q9ce7 ssmurf2_q9au4 swwp1_q90m0 swwp2_o00308 secw1_q76n89 secw2_q9p2p5 GPQSFTVEQWGTPEKLPRATCFNRLDLPPYESFEELWDKLQMAIENTQGFDGVD GPQLFTIEQWGSPEKLPRATCFNRLDLPPYETFEDLREKLLMAVENAQGFEGVD GPRRFTIEKAGEVQQLPKSTCFNRVDLPQYVDYDSMKQKLTLAVEETIGFGQE GPQKFCIEKVGKENWLPRSTCFNRLDLPPYKSYEQLKEKLLFAIEETEGFGQE GAAGPRLFTILIDANTDNLPKATCFNRIDIPPYESYEKLYEKLLTAVEETCGFAVE GPRLFTIQIDACTNNLPKATCFNRIDIPPYESYEKLYEKLLTAIEETCGFAVE GPQKFCIEKVGKDTWLPRSTCFNRLDLPPYKSYEQLKEKLLFAIEETEGFGQE GPQKFCIDKVGKETWLPRSTCFNRLDLPPYKSYEQLREKLLYAIEETEGFGQE GLRRFCIEKWGKITSLPRATCFNRLDLPPYPSYSMLYEKLLTAVEETSTFGLE GPRRFCVEKWGKITALPRATCFNRLDLPPYPSFSMLYEKLLTAVEETSTFGLE Nlobe Clobe % sequence identity catalytic cysteine Cterminal residues mutated in this study
6 b RDYKRKYEFFRRKLKKQNDIPNKFEMKLRRATVLEDSYRRIMGVKRADFLKARLWIEFDGEKGLDYGGVAREWFFLISKEMFNPYYGLFEYSATDNYTLQ α1 β1 α1 α2 β2 α3 α3 β3 β INPNSGINPDLSYFKFIGRVAGMAVYGKLLDGFFIRPFYKMMLKPITLDMESVDSEYYNSLRWILENDPTELDLRFIIDEELFGQTQELKNGG β4 α4 α4 α5 α6 α7 β5 β SEIVVTNKNKKEYIYLVIQWRFVNRIQKQMAAFKEGFFELIPQDLIKIFDENELELLMSGLGDVDVNDWRETKYKNGYSANQVIQWFWKAVLMMDSEK α8 α9 α10 α10 α11 β7 α12 α RIRLLQFVTGTSRVPMNGFAELYGSNGPQSFTVEQWGTPEKLPRATCFNRLDLPPYESFEELWDKLQMAIENTQGFDGVD S α13 α13 β8 S β9 β10 α14 Nlobe residues interacting with Clobe Clobe residues interacting with Nlobe ECT residues interacting thiolester ECT residues interacting with noncovalently bound Nlobe Buried Surface Area %33% engaged in an bond 33%66% S engaged in a salt bridge 66%100% % sequence identity Clobe Supplementary Figure 3 Sequence alignment of the ECT domains. (a) Sequence alignment of ECT domains of human Nedd4 family members and yeast Rsp5, performed with Muscle 2. Uniprot accession codes are provided with the protein names. (b) Sequence of Nedd4 ECT domain, colorcoded according to sequence conservation. Secondary structure elements are depicted, together with colored circles indicating residues involved in the interaction with ubiquitin or other lobe s residues. The radius of the circles is proportional to the buried area percentage, as calculated by PISA 3. Mutations introduced to produce a stable disulfide bridge between ECT and D are indicated in red. The catalytic cysteine is highlighted by a red triangle.
7 a b Clobe8896 Clobe snedd4 snedd4like swwp1 swwp2 secw2 secw1 sitch ssmurf2 ssmurf1 ScRsp5 suwe1 sace1 se6ap serc3 serc4 serc5 serc6 8 catalytic Cys FAELYGSNGPQSFTVEQWGTPEKLPRATCFNRLDLPPYESFEELWDKLQMAIENTQGFDGVDFAELYGSNGPQLFTIEQWGSPEKLPRATCFNRLDLPPYETFEDLREKLLMAVENAQGFEGVDFAELMGSNGPQKFCIEKVGKDTWLPRSTCFNRLDLPPYKSYEQLKEKLLFAIEETEGFGQEFAELIGSNGPQKFCIDKVGKETWLPRSTCFNRL DLPPYKSYEQLREKLLYAIEETEGFGQEFASLRGSNGPRRFCVEKWGKITALPRATCFNRLDLPPYPSFSMLYEKLLTAVEETSTFGLEFAALRGSNGLRRFCIEKWGKITSLPRATCFNRLDLPPYPSYSMLYEKLLTAVEETSTFGLEFADLMGSNGPQKFCIEKVGKENWLPRSTCFNRLDLPPYKSYEQLKEKLLFAIEETEGFGQEFKALQGAAGPRLFTIQIDACTNNLPKATCFNRI DIPPYESYEKLYEKLLTAIEETCGFAVEFKALQGSTGAAGPRLFTILIDANTDNLPKATCFNRI DIPPYESYEKLYEKLLTAVEETCGFAVEFKDLQGSDGPRRFTIEKAGEVQQLPKSTCFNRVDLPQYVDYDSMKQKLTLAVEETIGFGQEFAALEGMNGIQKFQIRDDRSTDRLPSATCFNQL DLPAYESFEKLRMLLLAIQECSEGFGLAFANIMGGSGLQNFTIAAVPYTPNLLPTSSTCINMLKLPEYPSK EILKDRLLVALCGSYGYTMALGKLKMIIAKNGPDTERLPTSTCFNVLLLPEYSSK EKLKERLLKAITYAKGFGMLMASLQIVIQSTASGEEYLPVATCYNLL DLPKYSSKEILSARLTQALDNYEGFSLAMKSLKLVIQSTGGGEEYLPVSTCFNLL DLPKYTEKETLRSKLIQAIDNEGFSLILNNMKITFCCPESWNERDPIRALTCFSVLFLPKYSTM ETVEEALQEAINNNRGFGIQKMEIVFRCPETFSERDPTSITCNILSLPKYSTM ERMEEALQVAINNNRGFVSPMLTQS D Nlobe Supplementary Figure 4 Structural location of the last sixty amino acids responsible for chain specificity. (a) Multiple sequence alignment of the last sixty residues of ECT domains, colored according to Clustal X color scheme. (b) Surface/cartoon representation of ECTNedd4~D:. Colors are as in Figure 1, except for the last sixty residues of the Clobe, which have been depicted in pink. The catalytic Cys is shown in yellow. Data obtained in this paper together with previously published data strongly indicate that these sixty amino acids of the Clobe are capable of binding both D and A to achieve chain specificity.
8 a b C lobe A889F C lobe WWP1 (1nd7) A889F WWP1 (1nd7) Cys867 Cys890 C Phe889 Phe 826 N 14 C lobe A889F Supplementary Figure 5 Structure of ECTNedd4 A889F. (a) Cartoon representation of ECTNedd4 A889F structure superposed to ECTWWP1 (pdb 1nd7). The orientation and colors of ECTNedd4 are the same of Figure 1, with the Nlobe of WWP1 ECT domain depicted in dark blue and the Clobe in light green. The catalytic cysteines are highlighted in ballandstick representation. The overall superposition of the two ECT domains, performed via secondary b) Cartoon representation of the ECTNedd4 A889F Clobe contoured by the final 2FoFc map of the representation.
9 SUPPLEMENTARY REFERENCES 1. Serniwka, S.A. & Shaw, G.S. The structure of the c8ubiquitin complex shows a unique ubiquitin interaction site. Biochemistry 48, (2009). 2. Edgar, R.C. MUSCLE: a multiple sequence alignment method with reduced time and space complexity. BMC Bioinformatics 5, 113 (2004). 3. Krissinel, E. & enrick, K. Inference of macromolecular assemblies from crystalline state. J Mol Biol 2, (2007).
Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular
 Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular weight markers (M). Supplementary Figure-2. Overlay of
Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular weight markers (M). Supplementary Figure-2. Overlay of
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
(B D) Three views of the final refined 2Fo-Fc electron density map of the Vpr (red)-ung2 (green) interacting region, contoured at 1.4σ.
 Supplementary Figure 1 Overall structure of the DDB1 DCAF1 Vpr UNG2 complex. (A) The final refined 2Fo-Fc electron density map, contoured at 1.4σ of Vpr, illustrating well-defined side chains. (B D) Three
Supplementary Figure 1 Overall structure of the DDB1 DCAF1 Vpr UNG2 complex. (A) The final refined 2Fo-Fc electron density map, contoured at 1.4σ of Vpr, illustrating well-defined side chains. (B D) Three
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase. Biol 405 Molecular Medicine
 Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site.
 Supplementary Figure 1 Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site. (a) Gel filtration profiles of purified 5 TMD samples at 100 nm, heated beforehand for
Supplementary Figure 1 Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site. (a) Gel filtration profiles of purified 5 TMD samples at 100 nm, heated beforehand for
Transient β-hairpin Formation in α-synuclein Monomer Revealed by Coarse-grained Molecular Dynamics Simulation
 Transient β-hairpin Formation in α-synuclein Monomer Revealed by Coarse-grained Molecular Dynamics Simulation Hang Yu, 1, 2, a) Wei Han, 1, 3, b) Wen Ma, 1, 2 1, 2, 3, c) and Klaus Schulten 1) Beckman
Transient β-hairpin Formation in α-synuclein Monomer Revealed by Coarse-grained Molecular Dynamics Simulation Hang Yu, 1, 2, a) Wei Han, 1, 3, b) Wen Ma, 1, 2 1, 2, 3, c) and Klaus Schulten 1) Beckman
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 The UBL and RING1 interface remains associated in the complex structures of Parkin and pub. a) Asymmetric Unit of crystal structure of UBLR0RBR and pub complex showing UBL (green),
Supplementary Figure 1 The UBL and RING1 interface remains associated in the complex structures of Parkin and pub. a) Asymmetric Unit of crystal structure of UBLR0RBR and pub complex showing UBL (green),
Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.
 Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.5 % SDS-PAGE gel under reducing and non-reducing conditions.
Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.5 % SDS-PAGE gel under reducing and non-reducing conditions.
Structure of the measles virus hemagglutinin bound to the CD46 receptor. César Santiago, María L. Celma, Thilo Stehle and José M.
 Supporting Figures and Table for Structure of the measles virus hemagglutinin bound to the CD46 receptor César Santiago, María L. Celma, Thilo Stehle and José M. Casasnovas This PDF file includes: Supplementary
Supporting Figures and Table for Structure of the measles virus hemagglutinin bound to the CD46 receptor César Santiago, María L. Celma, Thilo Stehle and José M. Casasnovas This PDF file includes: Supplementary
File name: Supplementary Information Description: Supplementary figures and supplementary tables. File name: Peer review file Description:
 File name: Supplementary Information Description: Supplementary figures and supplementary tables. File name: Peer review file Description: Supplementary Figure 1. Schematic of Ras biochemical coupled assay.
File name: Supplementary Information Description: Supplementary figures and supplementary tables. File name: Peer review file Description: Supplementary Figure 1. Schematic of Ras biochemical coupled assay.
Supplementary Information
 Supplementary Information Two common structural motifs for TCR recognition by staphylococcal enterotoxins Karin Erica Johanna Rödström 1, Paulina Regenthal 1, Christopher Bahl 2, Alex Ford 2, David Baker
Supplementary Information Two common structural motifs for TCR recognition by staphylococcal enterotoxins Karin Erica Johanna Rödström 1, Paulina Regenthal 1, Christopher Bahl 2, Alex Ford 2, David Baker
Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels. Supplementary Information
 Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels Craig T. Armstrong, Philip E. Mason, J. L. Ross Anderson and Christopher E. Dempsey
Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels Craig T. Armstrong, Philip E. Mason, J. L. Ross Anderson and Christopher E. Dempsey
Supplementary Figure 1 (previous page). EM analysis of full-length GCGR. (a) Exemplary tilt pair images of the GCGR mab23 complex acquired for Random
 S1 Supplementary Figure 1 (previous page). EM analysis of full-length GCGR. (a) Exemplary tilt pair images of the GCGR mab23 complex acquired for Random Conical Tilt (RCT) reconstruction (left: -50,right:
S1 Supplementary Figure 1 (previous page). EM analysis of full-length GCGR. (a) Exemplary tilt pair images of the GCGR mab23 complex acquired for Random Conical Tilt (RCT) reconstruction (left: -50,right:
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 Design of isolated protein and RNC constructs, and homogeneity of purified RNCs. (a) Schematic depicting the design and nomenclature used for all the isolated proteins and RNCs used
Supplementary Figure 1 Design of isolated protein and RNC constructs, and homogeneity of purified RNCs. (a) Schematic depicting the design and nomenclature used for all the isolated proteins and RNCs used
Supplementary Information Janssen et al.
 Supplementary Information Janssen et al. Insights into complement convertase formation based on the structure of the factor B CVF complex Bert J.C. Janssen 1, Lucio Gomes 1, Roman I. Koning 2, Dmitri I.
Supplementary Information Janssen et al. Insights into complement convertase formation based on the structure of the factor B CVF complex Bert J.C. Janssen 1, Lucio Gomes 1, Roman I. Koning 2, Dmitri I.
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/3/eaaq0762/dc1 Supplementary Materials for Structures of monomeric and oligomeric forms of the Toxoplasma gondii perforin-like protein 1 Tao Ni, Sophie I. Williams,
advances.sciencemag.org/cgi/content/full/4/3/eaaq0762/dc1 Supplementary Materials for Structures of monomeric and oligomeric forms of the Toxoplasma gondii perforin-like protein 1 Tao Ni, Sophie I. Williams,
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/2/4/e1500980/dc1 Supplementary Materials for The crystal structure of human dopamine -hydroxylase at 2.9 Å resolution Trine V. Vendelboe, Pernille Harris, Yuguang
advances.sciencemag.org/cgi/content/full/2/4/e1500980/dc1 Supplementary Materials for The crystal structure of human dopamine -hydroxylase at 2.9 Å resolution Trine V. Vendelboe, Pernille Harris, Yuguang
Practice Problems 3. a. What is the name of the bond formed between two amino acids? Are these bonds free to rotate?
 Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
SUPPORTING INFORMATION FOR. A Computational Approach to Enzyme Design: Using Docking and MM- GBSA Scoring
 SUPPRTING INFRMATIN FR A Computational Approach to Enzyme Design: Predicting ω- Aminotransferase Catalytic Activity Using Docking and MM- GBSA Scoring Sarah Sirin, 1 Rajesh Kumar, 2 Carlos Martinez, 2
SUPPRTING INFRMATIN FR A Computational Approach to Enzyme Design: Predicting ω- Aminotransferase Catalytic Activity Using Docking and MM- GBSA Scoring Sarah Sirin, 1 Rajesh Kumar, 2 Carlos Martinez, 2
Nature Structural and Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
2. Which of the following amino acids is most likely to be found on the outer surface of a properly folded protein?
 Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Atypical Natural Killer T-cell receptor recognition of CD1d-lipid antigens supplementary Information.
 Atypical Natural Killer T-cell receptor recognition of CD1d-lipid antigens supplementary Information. Supplementary Figure 1. Phenotypic analysis of TRBV25-1 + and TRBV25-1 - CD1d-α-GalCerreactive cells.
Atypical Natural Killer T-cell receptor recognition of CD1d-lipid antigens supplementary Information. Supplementary Figure 1. Phenotypic analysis of TRBV25-1 + and TRBV25-1 - CD1d-α-GalCerreactive cells.
Protein Investigator. Protein Investigator - 3
 Protein Investigator Objectives To learn more about the interactions that govern protein structure. To test hypotheses regarding protein structure and function. To design proteins with specific shapes.
Protein Investigator Objectives To learn more about the interactions that govern protein structure. To test hypotheses regarding protein structure and function. To design proteins with specific shapes.
Supplementary Information
 Supplementary Information Structural basis of improved second generation 3-nitro-tyrosine trna synthetases Richard B. Cooley, Jessica L. Feldman, Camden M. Driggers, Taylor Bundy, Audrey L. Stokes, P.
Supplementary Information Structural basis of improved second generation 3-nitro-tyrosine trna synthetases Richard B. Cooley, Jessica L. Feldman, Camden M. Driggers, Taylor Bundy, Audrey L. Stokes, P.
LAB#23: Biochemical Evidence of Evolution Name: Period Date :
 LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is worth 2 points.
 MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
Supplementary Material
 Supplementary Material Materials and methods Enzyme assay The enzymatic activity of -glucosidase toward salicin was measured with the Miller method (Miller, 1959) using glucose as the standard. A total
Supplementary Material Materials and methods Enzyme assay The enzymatic activity of -glucosidase toward salicin was measured with the Miller method (Miller, 1959) using glucose as the standard. A total
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10913 Supplementary Figure 1 2F o -F c electron density maps of cognate and near-cognate trna Leu 2 in the A site of the 70S ribosome. The maps are contoured at 1.2 sigma and some of
doi:10.1038/nature10913 Supplementary Figure 1 2F o -F c electron density maps of cognate and near-cognate trna Leu 2 in the A site of the 70S ribosome. The maps are contoured at 1.2 sigma and some of
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
scientific report Structure of the HECT:ubiquitin complex and its role in ubiquitin chain elongation scientificreport EMBO open
 EMBO open scientificreport Structure of the HET:ubiquitin complex and its role in ubiquitin chain elongation Elena Maspero 1, Sara Mari 1, Eleonora Valentini 1, Andrea Musacchio 2,AlexanderFish 3, Sebastiano
EMBO open scientificreport Structure of the HET:ubiquitin complex and its role in ubiquitin chain elongation Elena Maspero 1, Sara Mari 1, Eleonora Valentini 1, Andrea Musacchio 2,AlexanderFish 3, Sebastiano
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Nature Structural & Molecular Biology: doi: /nsmb.1933
 The structural basis of open channel block in a prokaryotic pentameric ligand-gated ion channel Ricarda J. C. Hilf, Carlo Bertozzi, Iwan Zimmermann, Alwin Reiter, Dirk Trauner and Raimund Dutzler a GLIC
The structural basis of open channel block in a prokaryotic pentameric ligand-gated ion channel Ricarda J. C. Hilf, Carlo Bertozzi, Iwan Zimmermann, Alwin Reiter, Dirk Trauner and Raimund Dutzler a GLIC
Structural Analysis of TCRpMHC Complexes Using Computational Tools. Feroze Mohideen Briarcliff High School
 Structural Analysis of TCRpMHC Complexes Using Computational Tools Feroze Mohideen Briarcliff High School TCR-pMHC Complexes Peptide Structure of TCR-pMHC complex PDB AO7 Crossreactivity is the ability
Structural Analysis of TCRpMHC Complexes Using Computational Tools Feroze Mohideen Briarcliff High School TCR-pMHC Complexes Peptide Structure of TCR-pMHC complex PDB AO7 Crossreactivity is the ability
Amino Acids. Review I: Protein Structure. Amino Acids: Structures. Amino Acids (contd.) Rajan Munshi
 Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
1. Describe the relationship of dietary protein and the health of major body systems.
 Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
PROTEINS. Amino acids are the building blocks of proteins. Acid L-form * * Lecture 6 Macromolecules #2 O = N -C -C-O.
 Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
The Basics: A general review of molecular biology:
 The Basics: A general review of molecular biology: DNA Transcription RNA Translation Proteins DNA (deoxy-ribonucleic acid) is the genetic material It is an informational super polymer -think of it as the
The Basics: A general review of molecular biology: DNA Transcription RNA Translation Proteins DNA (deoxy-ribonucleic acid) is the genetic material It is an informational super polymer -think of it as the
Cahn - Ingold - Prelog system. Proteins: Evolution, and Analysis Lecture 7 9/15/2009. The Fischer Convention (1) G (2) (3)
 Chapter 4 (1) G Proteins: Evolution, and Analysis Lecture 7 9/15/2009 A V L I M P F W Chapter 4 (2) S (3) T N Q Y C K R H D E The Fischer Convention Absolute configuration about an asymmetric carbon related
Chapter 4 (1) G Proteins: Evolution, and Analysis Lecture 7 9/15/2009 A V L I M P F W Chapter 4 (2) S (3) T N Q Y C K R H D E The Fischer Convention Absolute configuration about an asymmetric carbon related
Introduction to Protein Structure Collection
 Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Structural analysis of fungus-derived FAD glucose dehydrogenase
 Structural analysis of fungus-derived FAD glucose dehydrogenase Hiromi Yoshida 1, Genki Sakai 2, Kazushige Mori 3, Katsuhiro Kojima 3, Shigehiro Kamitori 1, and Koji Sode 2,3,* 1 Life Science Research
Structural analysis of fungus-derived FAD glucose dehydrogenase Hiromi Yoshida 1, Genki Sakai 2, Kazushige Mori 3, Katsuhiro Kojima 3, Shigehiro Kamitori 1, and Koji Sode 2,3,* 1 Life Science Research
Chemical Mechanism of Enzymes
 Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10962 Supplementary Figure 1. Expression of AvrAC-FLAG in protoplasts. Total protein extracted from protoplasts described in Fig. 1a was subjected to anti-flag immunoblot to detect AvrAC-FLAG
doi:10.1038/nature10962 Supplementary Figure 1. Expression of AvrAC-FLAG in protoplasts. Total protein extracted from protoplasts described in Fig. 1a was subjected to anti-flag immunoblot to detect AvrAC-FLAG
reads observed in trnas from the analysis of RNAs carrying a 5 -OH ends isolated from cells induced to express
 Supplementary Figure 1. VapC-mt4 cleaves trna Ala2 in E. coli. Histograms representing the fold change in reads observed in trnas from the analysis of RNAs carrying a 5 -OH ends isolated from cells induced
Supplementary Figure 1. VapC-mt4 cleaves trna Ala2 in E. coli. Histograms representing the fold change in reads observed in trnas from the analysis of RNAs carrying a 5 -OH ends isolated from cells induced
Supplementary Table 1. Data collection and refinement statistics (molecular replacement).
 Supplementary Table 1. Data collection and refinement statistics (molecular replacement). Data set statistics HLA A*0201- ALWGPDPAAA PPI TCR PPI TCR/A2- ALWGPDPAAA PPI TCR/A2- ALWGPDPAAA Space Group P2
Supplementary Table 1. Data collection and refinement statistics (molecular replacement). Data set statistics HLA A*0201- ALWGPDPAAA PPI TCR PPI TCR/A2- ALWGPDPAAA PPI TCR/A2- ALWGPDPAAA Space Group P2
BIOCHEMISTRY REVIEW. Overview of Biomolecules. Chapter 4 Protein Sequence
 BIOCHEMISTRY REVIEW Overview of Biomolecules Chapter 4 Protein Sequence 2 3 4 Are You Getting It?? A molecule of hemoglobin is compared with a molecule of lysozyme. Which characteristics do they share?
BIOCHEMISTRY REVIEW Overview of Biomolecules Chapter 4 Protein Sequence 2 3 4 Are You Getting It?? A molecule of hemoglobin is compared with a molecule of lysozyme. Which characteristics do they share?
Table S1. X-ray data collection and refinement statistics
 Table S1. X-ray data collection and refinement statistics Data collection H7.167 Fab-Sh2/H7 complex Beamline SSRL 12-2 Wavelength (Å) 0.97950 Space group I2 1 3 Unit cell parameters (Å, º) a = b = c=207.3,
Table S1. X-ray data collection and refinement statistics Data collection H7.167 Fab-Sh2/H7 complex Beamline SSRL 12-2 Wavelength (Å) 0.97950 Space group I2 1 3 Unit cell parameters (Å, º) a = b = c=207.3,
BIO 311C Spring Lecture 15 Friday 26 Feb. 1
 BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
SUPPLEMENTARY INFORMATION
 Supplementary Discussion The HGGG sequence motif forms an oxyanion hole in HLSs. In the case of GID1, the third Gly residue of the sequence motif is replaced with Ser (Ser123 in OsGID1), i.e., the sequence
Supplementary Discussion The HGGG sequence motif forms an oxyanion hole in HLSs. In the case of GID1, the third Gly residue of the sequence motif is replaced with Ser (Ser123 in OsGID1), i.e., the sequence
Table S1: Kinetic parameters of drug and substrate binding to wild type and HIV-1 protease variants. Data adapted from Ref. 6 in main text.
 Dynamical Network of HIV-1 Protease Mutants Reveals the Mechanism of Drug Resistance and Unhindered Activity Rajeswari Appadurai and Sanjib Senapati* BJM School of Biosciences and Department of Biotechnology,
Dynamical Network of HIV-1 Protease Mutants Reveals the Mechanism of Drug Resistance and Unhindered Activity Rajeswari Appadurai and Sanjib Senapati* BJM School of Biosciences and Department of Biotechnology,
3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B
 3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B 3.2.1 Characterization of the Ligand Binding Site at GPR109A Receptor Ligands of GPR109A Receptor are Carboxylic Acids Nicotinic acid (pyridine-3-carboxylic
3.2 Ligand-Binding at Nicotinic Acid Receptor Subtypes GPR109A/B 3.2.1 Characterization of the Ligand Binding Site at GPR109A Receptor Ligands of GPR109A Receptor are Carboxylic Acids Nicotinic acid (pyridine-3-carboxylic
Supplementary Information for Structural basis for the inhibition of Polo-like kinase 1
 Supplementary Information for Structural basis for inhibition of Polo-like kinase 1 Jun Xu, Chen Shen, Tao Wang & Junmin Quan Correspondence authors: Tao Wang, taowang@pkusz.edu.cn; Junmin Quan, quanjm@szpku.edu.cn
Supplementary Information for Structural basis for inhibition of Polo-like kinase 1 Jun Xu, Chen Shen, Tao Wang & Junmin Quan Correspondence authors: Tao Wang, taowang@pkusz.edu.cn; Junmin Quan, quanjm@szpku.edu.cn
paper and beads don t fall off. Then, place the beads in the following order on the pipe cleaner:
 Beady Pipe Cleaner Proteins Background: Proteins are the molecules that carry out most of the cell s dayto-day functions. While the DNA in the nucleus is "the boss" and controls the activities of the cell,
Beady Pipe Cleaner Proteins Background: Proteins are the molecules that carry out most of the cell s dayto-day functions. While the DNA in the nucleus is "the boss" and controls the activities of the cell,
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB
 Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
Reading from the NCBI
 Reading from the NCBI http://www.ncbi.nlm.nih.gov/books/bv.fcgi?highlight=thermodyn amics&rid=stryer.section.156#167 http://www.ncbi.nlm.nih.gov/books/bv.fcgi?highlight=stability,pr otein&rid=stryer.section.365#371
Reading from the NCBI http://www.ncbi.nlm.nih.gov/books/bv.fcgi?highlight=thermodyn amics&rid=stryer.section.156#167 http://www.ncbi.nlm.nih.gov/books/bv.fcgi?highlight=stability,pr otein&rid=stryer.section.365#371
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
Supplementary Information A Hydrophobic Barrier Deep Within the Inner Pore of the TWIK-1 K2P Potassium Channel Aryal et al.
 Supplementary Information A Hydrophobic Barrier Deep Within the Inner Pore of the TWIK-1 K2P Potassium Channel Aryal et al. Supplementary Figure 1 TWIK-1 stability during MD simulations in a phospholipid
Supplementary Information A Hydrophobic Barrier Deep Within the Inner Pore of the TWIK-1 K2P Potassium Channel Aryal et al. Supplementary Figure 1 TWIK-1 stability during MD simulations in a phospholipid
SDS-Assisted Protein Transport Through Solid-State Nanopores
 Supplementary Information for: SDS-Assisted Protein Transport Through Solid-State Nanopores Laura Restrepo-Pérez 1, Shalini John 2, Aleksei Aksimentiev 2 *, Chirlmin Joo 1 *, Cees Dekker 1 * 1 Department
Supplementary Information for: SDS-Assisted Protein Transport Through Solid-State Nanopores Laura Restrepo-Pérez 1, Shalini John 2, Aleksei Aksimentiev 2 *, Chirlmin Joo 1 *, Cees Dekker 1 * 1 Department
Supplementary Figure 1 Overall structure of the transmembrane pore of FraC. a, Crystal packing of the pore along the y-z, b, the z-x, and c, the y-x
 Supplementary Figure 1 Overall structure of the transmembrane pore of FraC. a, Crystal packing of the pore along the y-z, b, the z-x, and c, the y-x planes. The N-terminal and the β-core regions are depicted
Supplementary Figure 1 Overall structure of the transmembrane pore of FraC. a, Crystal packing of the pore along the y-z, b, the z-x, and c, the y-x planes. The N-terminal and the β-core regions are depicted
Amino acids. Side chain. -Carbon atom. Carboxyl group. Amino group
 PROTEINS Amino acids Side chain -Carbon atom Amino group Carboxyl group Amino acids Primary structure Amino acid monomers Peptide bond Peptide bond Amino group Carboxyl group Peptide bond N-terminal (
PROTEINS Amino acids Side chain -Carbon atom Amino group Carboxyl group Amino acids Primary structure Amino acid monomers Peptide bond Peptide bond Amino group Carboxyl group Peptide bond N-terminal (
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Term Definition Example Amino Acids
 Name 1. What are some of the functions that proteins have in a living organism. 2. Define the following and list two amino acids that fit each description. Term Definition Example Amino Acids Hydrophobic
Name 1. What are some of the functions that proteins have in a living organism. 2. Define the following and list two amino acids that fit each description. Term Definition Example Amino Acids Hydrophobic
PROTEINS. Building blocks, structure and function. Aim: You will have a clear picture of protein construction and their general properties
 PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
Biochemistry 2 Recita0on Amino Acid Metabolism
 Biochemistry 2 Recita0on Amino Acid Metabolism 04-20- 2015 Glutamine and Glutamate as key entry points for NH 4 + Amino acid catabolism Glutamine synthetase enables toxic NH 4 + to combine with glutamate
Biochemistry 2 Recita0on Amino Acid Metabolism 04-20- 2015 Glutamine and Glutamate as key entry points for NH 4 + Amino acid catabolism Glutamine synthetase enables toxic NH 4 + to combine with glutamate
Cbl ubiquitin ligase: Lord of the RINGs
 Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
Modeling holo-acp:dh and holo-acp:kr complexes of modular polyketide synthases: a docking and molecular dynamics study
 Anand and Mohanty BMC Structural Biology 2012, 12:10 RESEARCH ARTICLE Open Access Modeling holo-acp:dh and holo-acp:kr complexes of modular polyketide synthases: a docking and molecular dynamics study
Anand and Mohanty BMC Structural Biology 2012, 12:10 RESEARCH ARTICLE Open Access Modeling holo-acp:dh and holo-acp:kr complexes of modular polyketide synthases: a docking and molecular dynamics study
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Excerpt from J. Mol. Biol. (2002) 320, :
 Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
Previous Class. Today. Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism. Protein Kinase Catalytic Properties
 Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
The mechanism of patellamide macrocyclization revealed by study of the Prochloron sp PatG macrocyclase domain
 The mechanism of patellamide macrocyclization revealed by study of the Prochloron sp PatG macrocyclase domain Jesko Koehnke 1,2, Andrew Bent 1,2, Wael E. Houssen 2,3,4, David Zollman 1, Falk Morawitz 1,
The mechanism of patellamide macrocyclization revealed by study of the Prochloron sp PatG macrocyclase domain Jesko Koehnke 1,2, Andrew Bent 1,2, Wael E. Houssen 2,3,4, David Zollman 1, Falk Morawitz 1,
Crystallization-grade After D After V3 cocktail. Time (s) Time (s) Time (s) Time (s) Time (s) Time (s)
 Ligand Type Name 6 Crystallization-grade After 447-52D After V3 cocktail Receptor CD4 Resonance Units 5 1 5 1 5 1 Broadly neutralizing antibodies 2G12 VRC26.9 Resonance Units Resonance Units 3 1 15 1 5
Ligand Type Name 6 Crystallization-grade After 447-52D After V3 cocktail Receptor CD4 Resonance Units 5 1 5 1 5 1 Broadly neutralizing antibodies 2G12 VRC26.9 Resonance Units Resonance Units 3 1 15 1 5
Protein Structure Monday, March
 Michael Morales ffice: 204/202 ary; knock hard if you visit as my office is some door. Lab: 238 & 205 ary ffice hours Either make an appointment or just drop by Phone: 829-3965 E-Mail: moralesm@buffalo.edu
Michael Morales ffice: 204/202 ary; knock hard if you visit as my office is some door. Lab: 238 & 205 ary ffice hours Either make an appointment or just drop by Phone: 829-3965 E-Mail: moralesm@buffalo.edu
1. (38 pts.) 2. (25 pts.) 3. (15 pts.) 4. (12 pts.) 5. (10 pts.) Bonus (12 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Supplementary Information
 Supplementary Information Archaeal Elp3 catalyzes trna wobble uridine modification at C5 via a radical mechanism Kiruthika Selvadurai, Pei Wang, Joseph Seimetz & Raven H Huang* Department of Biochemistry,
Supplementary Information Archaeal Elp3 catalyzes trna wobble uridine modification at C5 via a radical mechanism Kiruthika Selvadurai, Pei Wang, Joseph Seimetz & Raven H Huang* Department of Biochemistry,
Chemistry 135, First Exam. September 23, Chem 135, Exam 1 SID:
 Chemistry 135, First Exam September 23, 2015 This exam will be worth 15% of your overall grade. Please read all instructions/questions carefully and provide answers in the space provided. There should
Chemistry 135, First Exam September 23, 2015 This exam will be worth 15% of your overall grade. Please read all instructions/questions carefully and provide answers in the space provided. There should
Supplementary Materials. High affinity binding of phosphatidylinositol-4-phosphate. by Legionella pneumophila DrrA
 Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Biology Open (2014) 000, 1 10 doi: /bio
 (2014) 000, 1 10 doi:10.1242/bio.201410041 Supplementary Material Michael Brauchle et al. doi: 10.1242/bio.201410041 Fig. S1. Alignment of GFP, sfgfp, egfp, eyfp, mcherry and mruby2. Sequence-based alignment
(2014) 000, 1 10 doi:10.1242/bio.201410041 Supplementary Material Michael Brauchle et al. doi: 10.1242/bio.201410041 Fig. S1. Alignment of GFP, sfgfp, egfp, eyfp, mcherry and mruby2. Sequence-based alignment
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Amino acids-incorporated nanoflowers with an
 Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Supplementary Materials for
 www.sciencemag.org/cgi/content/full/science.aal4326/dc1 Supplementary Materials for Structure of a eukaryotic voltage-gated sodium channel at near-atomic resolution Huaizong Shen, Qiang Zhou, Xiaojing
www.sciencemag.org/cgi/content/full/science.aal4326/dc1 Supplementary Materials for Structure of a eukaryotic voltage-gated sodium channel at near-atomic resolution Huaizong Shen, Qiang Zhou, Xiaojing
HOMEWORK II and Swiss-PDB Viewer Tutorial DUE 9/26/03 62 points total. The ph at which a peptide has no net charge is its isoelectric point.
 BIOCHEMISTRY I HOMEWORK II and Swiss-PDB Viewer Tutorial DUE 9/26/03 62 points total 1). 8 points total T or F (2 points each; if false, briefly state why it is false) The ph at which a peptide has no
BIOCHEMISTRY I HOMEWORK II and Swiss-PDB Viewer Tutorial DUE 9/26/03 62 points total 1). 8 points total T or F (2 points each; if false, briefly state why it is false) The ph at which a peptide has no
Chapter 10. Regulatory Strategy
 Chapter 10 Regulatory Strategy Regulation of enzymatic activity: 1. Allosteric Control. Allosteric proteins have a regulatory site(s) and multiple functional sites Activity of proteins is regulated by
Chapter 10 Regulatory Strategy Regulation of enzymatic activity: 1. Allosteric Control. Allosteric proteins have a regulatory site(s) and multiple functional sites Activity of proteins is regulated by
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature22394 Supplementary Table 1 Observed intermolecular interactions within the GLP-1:GLP-1R TMD interface. Superscripts refer to the Wootten residue numbering system
SUPPLEMENTARY INFORMATION doi:10.1038/nature22394 Supplementary Table 1 Observed intermolecular interactions within the GLP-1:GLP-1R TMD interface. Superscripts refer to the Wootten residue numbering system
Supplementary Figure 1. Heavy chain sequences of 2G1 and 8M2 aligned with V H 1-69
 Supplementary Figure 1. Heavy chain sequences of 2G1 and 8M2 aligned with V H 1-69 germline gene sequence. 2G1 and 8M2 acquired 14 and 18 mutations from the germline gene sequence, respectively. In 2G1,
Supplementary Figure 1. Heavy chain sequences of 2G1 and 8M2 aligned with V H 1-69 germline gene sequence. 2G1 and 8M2 acquired 14 and 18 mutations from the germline gene sequence, respectively. In 2G1,
Proteins. Amino acids, structure and function. The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka
 Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Chapter 3: Amino Acids and Peptides
 Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
A Chemical Look at Proteins: Workhorses of the Cell
 A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
Classification of amino acids: -
 Page 1 of 8 P roteinogenic amino acids, also known as standard, normal or primary amino acids are 20 amino acids that are incorporated in proteins and that are coded in the standard genetic code (subunit
Page 1 of 8 P roteinogenic amino acids, also known as standard, normal or primary amino acids are 20 amino acids that are incorporated in proteins and that are coded in the standard genetic code (subunit
AP Bio. Protiens Chapter 5 1
 Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Chapter 4: Information and Knowledge in the Protein Insulin
 Chapter 4: Information and Knowledge in the Protein Insulin This chapter will calculate the information and molecular knowledge in a real protein. The techniques discussed in this chapter to calculate
Chapter 4: Information and Knowledge in the Protein Insulin This chapter will calculate the information and molecular knowledge in a real protein. The techniques discussed in this chapter to calculate
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
CHAPTER 21: Amino Acids, Proteins, & Enzymes. General, Organic, & Biological Chemistry Janice Gorzynski Smith
 CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
Amino Acids. Amino Acids. Fundamentals. While their name implies that amino acids are compounds that contain an NH. 3 and CO NH 3
 Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
The three important structural features of proteins:
 The three important structural features of proteins: a. Primary (1 o ) The amino acid sequence (coded by genes) b. Secondary (2 o ) The interaction of amino acids that are close together or far apart in
The three important structural features of proteins: a. Primary (1 o ) The amino acid sequence (coded by genes) b. Secondary (2 o ) The interaction of amino acids that are close together or far apart in
Four melanocyte-stimulating hormones have the following amino acid sequences:
 Assignment 14: Melanocyte-stimulating hormone belongs to a group called the melanocortins. This group includes ACTH, alpha-msh, beta-msh and gamma-msh; these peptides are all cleavage products of a large
Assignment 14: Melanocyte-stimulating hormone belongs to a group called the melanocortins. This group includes ACTH, alpha-msh, beta-msh and gamma-msh; these peptides are all cleavage products of a large
