Breast Surgery Corporate Medical Policy
|
|
|
- Berenice Fletcher
- 5 years ago
- Views:
Transcription
1 File name: Breast Surgery File cde: UM.SURG.17 Originatin: 2016 Last Review: 07/2018 (PA list review) Next Review: 08/2019 Effective Date: 08/01/2018 Breast Surgery Crprate Medical Plicy Descriptin/Summary This plicy fcuses n breast-related prcedures that include mastectmy fr cancer, prphylactic mastectmy, recnstructin, the management f breast implants, breast reductins, crrectins fr certain asymmetries. BCBVT cvers medically necessary prcedures related t physilgical dysfunctin, such as breast cancer, cngenital and develpmental disrders, infectin, trauma, surgical cmplicatins and macrmastia causing physilgical dysfunctin in men and wmen. BCBSVT cnsiders prcedures that are nly perfrmed t reshape nrmal structures f the bdy in rder t imprve ne s appearance r self-esteem nly, t be csmetic and therefre nn-cvered as benefit exclusins. Plicy Cding Infrmatin Click the links belw fr attachments, cding tables & instructins. Attachment I CPT / Cding Table Requests fr breast surgery shuld be accmpanied by the fllwing dcumentatin: The name and date f the prpsed surgery. Date f accident r injury, if applicable Histry f present illness and/r cnditins including diagnses Dcumentatin f diagnsis, functinal impairment, pain r significant anatmic variance Hw the treatment can be reasnably expected t imprve the functinal impairment If applicable, the descriptin f and CPT cding fr planned staged prcedure fllwing acute repair, within tw years f previus stage r initial primary repair Any additinal infrmatin listed fr a specific prcedure as indicated fr the specific prcedures listed belw. BCBSVT will review prcedures intended t crrect cmplicatins frm a csmetic prcedure, whether the riginal prcedure was medically necessary r a nn-cvered service. In rder Page 1 f 12
2 fr these crrectins t be cnsidered medically necessary the subsequent surgery needs t be recnstructive in nature (i.e. prcedures perfrmed n abnrmal structures f the bdy, caused by cngenital defects, develpmental abnrmalities, trauma, infectin, tumrs r disease. It is generally perfrmed t imprve functin, but may als be dne t apprximate nrmal appearance). The prcedures in this medical plicy are cnsidered medically necessary in accrdance with the Wmen s Health and Cancer Rights Act f 1998, when perfrmed as a breast recnstructin prcedure fllwing r in cnnectin with mastectmy, breast cnservatin therapy (BCT) r ther diagnstic prcedures causing defrmity f the breast, in cnnectin with breast cancer, evaluatin f breast cancer r suspected breast cancer, t prevent develpment f breast cancer in high risk patients, recnstructin fllwing breast tissue destructin due t accidental injury, trauma, infectin r disease (including ther cancers). If the intended service relates t gender reassignment services, please refer t the BCBSVT Transgender Services medical plicy. Plicy and Guidelines Breast Prsthetics ( cdes L2999, L3999, L5999, L7499, *L8020, *L8030, *L8031, L8039, L8499, L8699) We cnsider the prcedure medically necessary fr the fllwing indicatins: - A r a histry f breast cancer - Fllwing a mastectmy fr cancer - Fllwing a prphylactic mastectmy - Fr absence f the breast due t trauma, disease r infectin - A diagnsis f Pland s syndrme We cnsider the prcedure csmetic and therefre nt cvered as a benefit exclusin when: - Nne f the abve indicatins are met. - Obtained nly t imprve appearance r t imprve ne s self-esteem. *When billed with a prir apprval is nt required. Mastectmy fr Gynecmastia (CPT cde 19300) - surgery due t develpment f abnrmally large mammary gland in bilgically male individuals. We cnsider the prcedure medical necessary fr the fllwing: - With a ; OR - If the criteria fr a Prphylactic Mastectmy are met. - When all f the fllwing are met: Dcumented symptms, including pain r tenderness directly related t the breast tissue, and which has a clinically significant impact upn nrmal activities f daily living despite nn-narctic analgesics and anti-inflammatry agents; AND Apprpriate diagnstic evaluatin has been dne fr pssible underlying etilgy; AND Page 2 f 12
3 The tissue remved must be glandular breast tissue; and The extra tissue must nt be the result f besity, adlescence, r reversible effects f drug treatment that can be discntinued. (This includes drug-induced Gynecmastia remaining unreslved six mnths after cessatin f the causative drug therapy; Additinally, fr thse under 18 years f age, ne f the fllwing must be submitted as evidence f puberty cmpletin.* * Evidence f puberty cmpletin: Dcumented tanner stage IV r V fr members aged 15-18, AND Stable height measurements fr 6 mnths, OR Puberty cmpletin as shwn n wrist radigraph. We cnsider the prcedure nt medically necessary when any f the fllwing is present: - Cnservative attempts t cntrl the pain r tenderness, such as nn- narctic analgesics and anti-inflammatry agents, have nt been attempted. - Use f a medicatin knwn t cause gynecmastia has nt been discntinued. - The apprpriate diagnstic evaluatin fr etilgy has nt been cmpleted. We cnsider the prcedure csmetic and therefre nt cvered as a benefit exclusin fr the fllwing circumstances (nt an all-inclusive list): - The tissue being remved is nt glandular in nature; OR - The medically necessary criteria abve is nt met and the prcedure is intended nly t imprve appearance r t imprve ne s emtinal well-being. Prphylactic Mastectmy (CPT cdes 19303, 19304) Surgical remval f breasts due t a high cancer risk. It is strngly recmmended that all candidates fr prphylactic mastectmy underg cunseling regarding cancer risks frm a health prfessinal skilled in assessing cancer risk ther than the perating surgen and discussin f the varius treatment ptins, including increased surveillance r chempreventin with the apprpriate medicatin e.g. tamxifen r ralxifene. Patients with a high risk f breast cancer may be defined as ne r mre f the fllwing: a knwn BRCA1 r BRCA2 mutatin r at high risk f BRCA1 r BRCA2 mutatin due t a knwn presence f the mutatin in relatives r anther gene mutatin assciated with increased risk (eg, PTEN, TP53, CDH1, and STK11) r Li-Fraumeni syndrme r Cwden syndrme r Bannayan-Riley-Ruvalcaba syndrme r a first-degree relative with ne f these syndrmes r high risk (lifetime risk abut 20% t 25% r greater) f develping breast cancer as identified by mdels that are largely defined by family histry r received raditherapy t the chest between 10 and 30 years f age We cnsider the prcedure medically necessary fr any f the fllwing: - In patients at high risk f breast cancer as defined abve. - In patients with inflammatry breast cancer. Page 3 f 12
4 - In patients with lbular carcinma in situ. - In patients with such extensive mammgraphic abnrmalities (i.e. calcificatins) that adequate bipsy r excisin is impssible. We cnsider the prcedure investigatinal fr all ther indicatins, including, but nt limited t cntralateral prphylactic mastectmy in wmen with breast cancer wh d nt meet the high risk criteria as defined abve. Breast Recnstructin (CPT cdes 15777,19340*, 19342*, 19350*, 19357*, 19361*, 19364*, 19366*, 19367*, 19368*, 19369*, 19380*). Utilizatin f natural r artificial tissue t recnstruct breasts fllwing mastectmy, breast cnservatin therapy, burns, trauma and diagnstic defrmity We cnsider recnstructin medically necessary fr any f the fllwing: - Fr the affected breast When breast tissue is affected by disease, trauma, burns r infectin When perfrmed in cnnectin with cancer, the evaluatin f cancer, the evaluatin f suspected cancer (i.e. fllwing bipsy r lumpectmy), r the preventin f breast cancer develpment in high risk patients; OR Fr prstheses and physical cmplicatins* f all stages f mastectmy, breast cnservatin therapy (BCT) r ther diagnstic prcedures causing defrmity (i.e. fllwing bipsy r lumpectmy) including lymphedema treatment. *Physical cmplicatins f a staged mastectmy may include, but is nt limited t, abdminal scar revisin/release related t prir tissue needed fr breast recnstructin. Fllwing the remval f a ruptured silicne gel-filled implant Fr the unaffected breast in rder t create a symmetrical appearance *When billed with a, fllwing an apprved mastectmy, prir apprval is nt required. We cnsider the fllwing csmetic and therefre nt cvered as a benefit exclusin: Breast recnstructin fllwing mastectmy fr gynecmastia. Ntes: Breast recnstructin utilizing autlgus fat grafting t the breast with adipse-derived stem cells is cnsidered investigatinal. CPT cde shuld represent autlgus fat grafting frm direct harvest and is apprpriate. Allgraft material fr use in breast recnstructive therapy ( Q4100, Q4107, Q4116, Q4128) Implant repsitining Inverted nipple crrectin- (CPT 19355) Mastpexy (CPT 19316) If the preceding criteria fr Breast Recnstructin are nt met, the fllwing prcedures are cnsidered csmetic and therefre nt cvered as a benefit exclusin: Mastpexy (CPT 19316) Page 4 f 12
5 Inverted nipple crrectin (CPT 19355) Implant repsitining Tatting f the nipple and/r arela (CPT 11920, 11921, 11922) Reductin Mammplasty (CPT cde 19318) Surgical reductin f breasts in wmen due t size and persistent symptms. We cnsider the prcedure medically necessary fr the treatment f macrmastia fr the fllwing: - Breast size is stable fr six t twelve mnths prir t surgery. AND - A minimum f 6-weeks f tw persistent well-dcumented symptms which impair functin such as shulder, neck, r back pain r pain interfering with sleep related t macrmastia that is nt respnsive t cnservative therapy, such as an apprpriate supprt bra, exercises, heat/cld treatment, and apprpriate nnsteridal anti-inflammatry agents/muscle relaxants OR - One f the abve symptms AND recurrent r chrnic intertrig between the pendulus breast and the chest wall that is resistant t tpical treatment. - Additinally, fr thse under 18 years f age, ne f the fllwing must be submitted as evidence f puberty cmpletin.* * Evidence f puberty cmpletin: Dcumented tanner stage IV r V fr members aged 15-18, AND Stable height measurements fr 6 mnths, OR Puberty cmpletin as shwn n wrist radigraph. Breast Reductin (Reductin Mammaplasty CPT 19318) is cnsidered medically necessary when in cnnectin with breast recnstructin fllwing a mastectmy. - We cnsider reductin mammplasty csmetic and therefre nt cvered as a benefit exclusin fr any f the fllwing: Perfrmed in rder t imprve athletic perfrmance OR Obtained nly t imprve appearance r t imprve ne s self-esteem - We cnsider the prcedure investigatinal fr all ther indicatins nt utlined abve. Remval f implant(s) (CPT cdes 19328, 19330); insertin f implant(s) (CPT cdes 19340, 19342, C1789); Periprsthetic capsultmy r capsulectmy (CPT cdes 19370, 19371): the remval and replacement f breast implants riginally placed fllwing mastectmy. - Additinal Dcumentatin Required: Date f implantatin and type f implant Objective evidence f leakage Baker Cntracture Class* - We cnsider the prcedure medically necessary fr the any f the fllwing: Page 5 f 12
6 Explantatin (remval f implant) f a silicne gel-filled r saline filled implant with a dcumented implant rupture, extrusin, Baker Class IV* cntracture, rsurgical treatment f cancer r ther disease, infectin, trauma r burn. - We cnsider the prcedures nt medically necessary fr the any f the fllwing: Explantatin f a when the riginal recnstructin was fr csmetic reasns and the medically necessary criteria abve is nt met. Systemic symptms, attributed t cnnective tissue diseases, autimmune diseases, etc.; Baker class III cntractures in patients with implants fr csmetic purpses; Rupture f a saline implant in patients with implants fr csmetic purpses; Pain nt related t cntractures r rupture. - We cnsider the prcedure csmetic and therefre nt cvered as a benefit exclusin fr the fllwing: Obtained nly t imprve appearance r t imprve ne s self-esteem. Grade I: Grade II: Grade III: Grade IV: *Baker Classificatin f breast cntractures: Augmented breast feels as sft as a nrmal breast Breast is less sft and the implant can be palpated but is nt visible. Breast is firm, palpable and the implant (r its distrtin) is visible Breast is hard, painful, cld, tender and distrted Unilateral Breast Surgery fr Asymmetry reductin mammplasty (CPT cde 19318) and/r augmentatin mammplasty (CPT cde & 19325) - surgical recnstructin in females f ne breast by either reducing r enlarging. - Additinal dcumentatin Required: Histry and physical findings Height and weight Size f each breast Date f previus surgery, if applicable Pathlgic diagnsis, if applicable Estimate f amunt f tissue t be remved in a reductin r size f implant fr augmentatin. Additinally, fr thse under 18 years f age, ne f the fllwing must be submitted as evidence f puberty cmpletin.* * Evidence f puberty cmpletin: Dcumented tanner stage IV r V fr members aged 15-18, AND Stable height measurements fr 6 mnths, OR Puberty cmpletin as shwn n wrist radigraph. Page 6 f 12
7 - We cnsider the prcedures medically necessary fr any f the fllwing indicatins: Bilgical females must be at least 15 years f age and have reached puberty*, and have a diagnsis f Pland s syndrme (cngenital absence f breasts) OR A disfiguring traumatic accident (e.g. burn) r cmplicatin f medical treatment (e.g. necrsis) OR A breast infectin resulting in disfigurement - We cnsider prcedures fr the fllwing csmetic and therefre nt cvered as a benefit exclusin: Unilateral augmentatin r reductin mammplasty intended t create symmetry between therwise nrmal breasts and the medically necessary criteria abve is nt met OR Unilateral augmentatin r reductin mammplasty intended nly t imprve appearance r t imprve a ne s self-esteem Reference Resurces 1. Blue Crss and Blue Shield Assciatin. Medical Plicy Reference Manual # Surgical Treatment f Bilateral Gynecmastia, last reviewed: February Blue Crss and Blue Shield Assciatin. Medical Plicy Reference Manual # Adipse-Derived Stem Cells in Autlgus Fat Grafting t the Breast, last reviewed: Octber Blue Crss and Blue Shield Assciatin. Medical Plicy Reference Manual # Prphylactic Mastectmy, last reviewed: February Cuhaci, N., Plat, S.B., Evrans, B., Esry, R. and Cakir, B. Gynecmastia: clinical evaluatin and management. Indian Jurnal f Endcrinlgy and Metablism Mar- Apr; 18(2): Kerrigan, C.L., Cllins, E.D., Kim, H.M., Schnurr, P.L., Cunningham, B. and Lwery, J. Reductin mammplasty: defining medical necessity. Medical Decisin Making May- Jun; 22(3): Wlfswinkel, B.S., Lemaine, V., Weathers, W. M., Chike-Obi, C.J, Xue, A.S. and Heller, L. Hyperplastic Breast Anmalies in the Female Adlescent Breast. Seminars in Plastic Surgery Feb; Related Plicies BCBSVT Medical Plicy n Transgender Services BCBSVT Medical Plicy n Biengineered Skin and Sft Tissue Substitutes Dcument Precedence Blue Crss and Blue Shield f Vermnt (BCBSVT) Medical Plicies are develped t prvide clinical guidance and are based n research f current medical literature and review f cmmn medical practices in the treatment and diagnsis f disease. The applicable grup/individual cntract and member certificate language, r emplyer s benefit plan if an ASO grup, determines benefits that are in effect at the time f service. Since medical practices and knwledge are cnstantly evlving, BCBSVT reserves the right t review and Page 7 f 12
8 revise its medical plicies peridically. T the extent that there may be any cnflict between medical plicy and cntract/emplyer benefit plan language, the member s cntract/emplyer benefit plan language takes precedence Audit Infrmatin BCBSVT reserves the right t cnduct audits n any prvider and/r facility t ensure cmpliance with the guidelines stated in the medical plicy. If an audit identifies instances f nn-cmpliance with this medical plicy, BCBSVT reserves the right t recup all nncmpliant payments. Benefit Determinatin Guidance Administrative and Cntractual Guidance Prir apprval is required and benefits are subject t all terms, limitatins and cnditins f the subscriber cntract. Incmplete authrizatin requests may result in a delay f decisin pending submissin f missing infrmatin. T be cnsidered cmpete, see plicy guidelines abve. An apprved referral authrizatin fr members f the New England Health Plan (NEHP) is required. A prir apprval fr Access Blue New England (ABNE) members is required. NEHP/ABNE members may have different benefits fr services listed in this plicy. T cnfirm benefits, please cntact the custmer service department at the member s health plan. Federal Emplyee Prgram (FEP): Members may have different benefits that apply. Fr further infrmatin please cntact FEP custmer service r refer t the FEP Service Benefit Plan Brchure. It is imprtant t verify the member s benefits prir t prviding the service t determine if benefits are available r if there is a specific exclusin in the member s benefit. Cverage varies accrding t the member s grup r individual cntract. Nt all grups are required t fllw the Vermnt legislative mandates. Member Cntract language takes precedence ver medical plicy when there is a cnflict. If the member receives benefits thrugh an Administrative Services Only (ASO) grup, benefits may vary r nt apply. T verify benefit infrmatin, please refer t the member s emplyer benefit plan dcuments r cntact the custmer service department. Language in the emplyer benefit plan dcuments takes precedence ver medical plicy when there is a cnflict. Plicy Implementatin/Update infrmatin 08/2016 New plicy. 12/2016 Added CPT Cde fr clarificatin in medical plicy. Page 8 f 12
9 08/2017 Reviewed and vted at HPC 08/07/2017 with the fllwing: Updated related plicies sectin, remved language under prphylactic mastectmy sectin, added CPT cde as medically necessary, remved language under breast recnstructin added additinal medical criteria under breast recnstructin, added cding table t align with cdes cntained within the medical plicy, remved language under remval f implants, remved language under unilateral breast surgery fr asymmetry. 07/2018 Added CPT cde t require PA Eligible prviders Qualified healthcare prfessinals practicing within the scpe f their license(s). Apprved by BCBSVT Medical Directrs Date Apprved Gabrielle Bercy-Rbersn, MD, MPH, MBA Senir Medical Directr Chair, Health Plicy Cmmittee Jshua Plavin, MD, MPH, MBA Chief Medical Officer Attachment I CPT / Cding Table Cde Type Number Brief Descriptin Plicy Instructins The fllwing cdes will be cnsidered as medically necessary when applicable criteria have been met. Tatting, intradermal intrductin f insluble paque pigments t crrect clr defects f skin, including micrpigmentatin; 6.0 sq cm r CPT less CPT Tatting, intradermal intrductin f insluble paque pigments t crrect clr defects f skin, including micrpigmentatin; 6.1 t 20.0 sq cm Page 9 f 12
10 CPT CPT Tatting, intradermal intrductin f insluble paque pigments t crrect clr defects f skin, including micrpigmentatin; each additinal 20.0 sq cm, r part theref (List separately in additin t cde fr primary prcedure) Implantatin f bilgic implant (eg, acellular dermal matrix) fr sft tissue reinfrcement (ie, breast, trunk)(list separately in additin t cde fr primary prcedure) CPT Mastectmy fr gynecmastia CPT Mastectmy, partial (eg, lumpectmy, tylectmy, quadrantectmy, segmentectmy) CPT Mastectmy, partial (eg, lumpectmy, tylectmy, quadrantectmy, segmentectmy); with axillary lymphadenectmy CPT Mastectmy, simple, cmplete CPT Mastectmy, subcutaneus CPT Mastpexy CPT Reductin mammaplasty CPT Mammaplasty, augmentatin; withut prsthetic implant CPT Mammaplasty, augmentatin; with prsthetic implant CPT Remval f intact mammary implant CPT Remval f mammary implant material CPT Immediate insertin f breast prsthesis fllwing mastpexy, mastectmy r in recnstructin Page 10 f 12
11 CPT Delayed insertin f breast prsthesis fllwing mastpexy, mastectmy r in recnstructin CPT Nipple/arela recnstructin CPT Crrectin f inverted nipples CPT Breast recnstructin, immediate r delayed, with tissue expander, including subsequent expansin CPT CPT CPT CPT CPT CPT CPT CPT Breast recnstructin with latissimus drsi flap, withut prsthetic implant Breast recnstructin with free flap Breast recnstructin with ther technique Breast recnstructin with transverse rectus abdminis mycutaneus flap (TRAM), single pedicle, including clsure f dnr site Breast recnstructin with transverse rectus abdminis mycutaneus flap (TRAM), single pedicle, including clsure f dnr site; with micrvascular anastmsis (supercharging) Breast recnstructin with transverse rectus abdminis mycutaneus flap (TRAM), duble pedicle, including clsure f dnr site Open periprsthetic capsultmy, breast Preprsthetic capsulectmy, breast CPT Revisin f recnstructed breast CPT CPT Preparatin f mulage fr custm breast implant Tissue grafts, ther (eg, paratenn, fat, dermis) Page 11 f 12
12 L8020 L8030 L8031 L8039 L8499 L8699 Q4100 Q4107 Breast prsthesis, mastectmy frm Breast prsthesis, silicne r equal, withut integral adhesive Breast prsthesis, silicne r equal with integral adhesive Breast prsthesis, nt therwise specified Unlisted prcedure fr miscellaneus prsthetic services Prsthetic implant, nt therwise specified Skin substitute, nt therwise specified Graftjacket, per square centimeter Q4116 Allderm, per square centimeter Q4128 S2066 S2067 S2068 FlexHD, Allpatch HD, r Matrix HD, per square centimeter Breast recnstructin with gluteal artery perfratr (GAP) flap, including harvesting f the flap, micrvascular transfer, clsure f dnr site and shaping the flap int a breast, unilateral Breast recnstructin f a single breast with stacked deep inferir epigastric perfratr (DIEP) flap(s) and/r gluteal artery perfratr (GAP) flap(s), including harvesting f the flap(s), micrvascular transfer, clsure f dnr site(s) and shaping the flap int a breast, unilateral Breast recnstructin with deep inferir epigastric perfratr (DIEP) flap, r superficial inferir epigastric artery (SIEA) flap, including harvesting f the flap, micrvascular transfer, clsure f dnr site and shaping the flap int a breast, unilateral Page 12 f 12
Benefits to Change for Diagnostic and Surgical/Reconstructive Breast Therapies and Corrective Procedures January 1, 2016
 Benefits t Change fr Diagnstic and Surgical/Recnstructive Breast Therapies and Crrective Prcedures January 1, 2016 Infrmatin psted Nvember 13, 2015 Effective fr dates f service n r after January 1, 2016,
Benefits t Change fr Diagnstic and Surgical/Recnstructive Breast Therapies and Crrective Prcedures January 1, 2016 Infrmatin psted Nvember 13, 2015 Effective fr dates f service n r after January 1, 2016,
Related Policies None
 Medical Plicy MP 3.01.501 Guidelines fr Cverage f Mental and Behaviral Health Services Last Review: 8/30/2017 Effective Date: 8/30/2017 Sectin: Mental Health End Date: 08/19/2018 Related Plicies Nne DISCLAIMER
Medical Plicy MP 3.01.501 Guidelines fr Cverage f Mental and Behaviral Health Services Last Review: 8/30/2017 Effective Date: 8/30/2017 Sectin: Mental Health End Date: 08/19/2018 Related Plicies Nne DISCLAIMER
Breast Surgery Corporate Medical Policy
 File name: Breast Surgery File code: UM.SURG.17 Origination: 2016 Last Review: 11/2018 (PA List Review) Next Review: 11/2019 Effective Date: 04/01/2018 Breast Surgery Corporate Medical Policy Description/Summary
File name: Breast Surgery File code: UM.SURG.17 Origination: 2016 Last Review: 11/2018 (PA List Review) Next Review: 11/2019 Effective Date: 04/01/2018 Breast Surgery Corporate Medical Policy Description/Summary
Breast Cancer Awareness Month 2018 Key Messages (as of June 6, 2018)
 Breast Cancer Awareness Mnth 2018 Key Messages (as f June 6, 2018) In this dcument there are tw sectins f messages in supprt f Cancer Care Ontari s Breast Cancer Awareness Mnth 2018: 1. Campaign key messages
Breast Cancer Awareness Mnth 2018 Key Messages (as f June 6, 2018) In this dcument there are tw sectins f messages in supprt f Cancer Care Ontari s Breast Cancer Awareness Mnth 2018: 1. Campaign key messages
Bariatric Surgery FAQs for Employees in the GRMC Group Health Plan
 Bariatric Surgery FAQs fr Emplyees in the GRMC Grup Health Plan Gergia Regents Medical Center and Gergia Regents Medical Assciates emplyees and eligible dependents wh are in the GRMC Grup Health Plan (Select
Bariatric Surgery FAQs fr Emplyees in the GRMC Grup Health Plan Gergia Regents Medical Center and Gergia Regents Medical Assciates emplyees and eligible dependents wh are in the GRMC Grup Health Plan (Select
Solid Organ Transplant Benefits to Change for Texas Medicaid
 Slid Organ Transplant Benefits t Change fr Texas Medicaid Infrmatin psted February 13, 2015 Nte: All new and updated prcedure cdes and their assciated reimbursement rates are prpsed benefits pending a
Slid Organ Transplant Benefits t Change fr Texas Medicaid Infrmatin psted February 13, 2015 Nte: All new and updated prcedure cdes and their assciated reimbursement rates are prpsed benefits pending a
LEVEL OF CARE GUIDELINES: INTENSIVE BEHAVIORAL THERAPY/APPLIED BEHAVIOR ANALYSIS FOR AUTISM SPECTRUM DISORDER HAWAII MEDICAID QUEST
 OPTUM LEVEL OF CARE GUIDELINES: INTENSIVE BEHAVIORAL THERAPY / APPLIED BEHAVIOR ANALYSIS FOR AUTISM SPECTRUM DISORDER HAWAII MEDICAID QUEST LEVEL OF CARE GUIDELINES: INTENSIVE BEHAVIORAL THERAPY/APPLIED
OPTUM LEVEL OF CARE GUIDELINES: INTENSIVE BEHAVIORAL THERAPY / APPLIED BEHAVIOR ANALYSIS FOR AUTISM SPECTRUM DISORDER HAWAII MEDICAID QUEST LEVEL OF CARE GUIDELINES: INTENSIVE BEHAVIORAL THERAPY/APPLIED
HIP REPLACEMENT SURGERY (ARTHROPLASTY)
 Prtcl: ORT015 Effective Date: June 1, 2017 HIP REPLACEMENT SURGERY (ARTHROPLASTY) Table f Cntents Page COMMERCIAL & MEDICAID COVERAGE RATIONALE... 1 MEDICARE COVERAGE RATIONALE... 3 U.S.FOOD AND DRUG ADMINISTRATION
Prtcl: ORT015 Effective Date: June 1, 2017 HIP REPLACEMENT SURGERY (ARTHROPLASTY) Table f Cntents Page COMMERCIAL & MEDICAID COVERAGE RATIONALE... 1 MEDICARE COVERAGE RATIONALE... 3 U.S.FOOD AND DRUG ADMINISTRATION
Wound Care Equipment and Supply Benefits to Change for Texas Medicaid July 1, 2018
 Wund Care Equipment and Supply Benefits t Change fr Texas Medicaid July 1, 2018 Infrmatin psted May 11, 2018 Nte: Texas Medicaid managed care rganizatins (MCOs) must prvide all medically necessary, Medicaid-cvered
Wund Care Equipment and Supply Benefits t Change fr Texas Medicaid July 1, 2018 Infrmatin psted May 11, 2018 Nte: Texas Medicaid managed care rganizatins (MCOs) must prvide all medically necessary, Medicaid-cvered
Cardiac Rehabilitation Services
 Dcumentatin Guidance N. DG1011 Cardiac Rehabilitatin Services Revisin Letter A 1.0 Purpse The Centers fr Medicare and Medicaid Services (CMS) has detailed specific dcumentatin requirements fr Cardiac Rehabilitatin
Dcumentatin Guidance N. DG1011 Cardiac Rehabilitatin Services Revisin Letter A 1.0 Purpse The Centers fr Medicare and Medicaid Services (CMS) has detailed specific dcumentatin requirements fr Cardiac Rehabilitatin
National Imaging Associates, Inc. (NIA) Frequently Asked Questions (FAQs) For Louisiana Healthcare Connections Providers
 Natinal Imaging Assciates, Inc. (NIA) Frequently Asked Questins (FAQs) Fr Luisiana Healthcare Cnnectins Prviders Questin GENERAL Why did Luisiana Healthcare Cnnectins implement a Medical Prgram? Answer
Natinal Imaging Assciates, Inc. (NIA) Frequently Asked Questins (FAQs) Fr Luisiana Healthcare Cnnectins Prviders Questin GENERAL Why did Luisiana Healthcare Cnnectins implement a Medical Prgram? Answer
o Procedures performed o Diagnoses Identified o Certain devices/equipment/supplies acquired for patient
 Image Surce: https://s-media-cache-ak0.pinimg.cm/736x/7c/29/91/7c2991805f004e1ca05e42a79883f4a7.jpg 6/30/2017 Curse Objectives A Practical Guide t Cding fr Audilgists in 2017 Megan Keirans, AuD University
Image Surce: https://s-media-cache-ak0.pinimg.cm/736x/7c/29/91/7c2991805f004e1ca05e42a79883f4a7.jpg 6/30/2017 Curse Objectives A Practical Guide t Cding fr Audilgists in 2017 Megan Keirans, AuD University
NIA Magellan 1 Spine Care Program Interventional Pain Management Frequently Asked Questions (FAQs) For Medicare Advantage HMO and PPO
 NIA Magellan 1 Spine Care Prgram Interventinal Pain Management Frequently Asked Questins (FAQs) Fr Medicare Advantage HMO and PPO Questin GENERAL Why is Flrida Blue implementing a Spine Management prgram
NIA Magellan 1 Spine Care Prgram Interventinal Pain Management Frequently Asked Questins (FAQs) Fr Medicare Advantage HMO and PPO Questin GENERAL Why is Flrida Blue implementing a Spine Management prgram
Original Policy Date
 MP 7.01.113 Orthgnathic Surgery Medical Plicy Sectin Surgery Issue 12/2013 Original Plicy Date 12/2013 Last Review Status/Date Lcal Plicy created 12/2013 Return t Medical Plicy Index Disclaimer Our medical
MP 7.01.113 Orthgnathic Surgery Medical Plicy Sectin Surgery Issue 12/2013 Original Plicy Date 12/2013 Last Review Status/Date Lcal Plicy created 12/2013 Return t Medical Plicy Index Disclaimer Our medical
2017 CMS Web Interface
 CMS Web Interface PREV-5 (NQF 2372): Breast Cancer Screening Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION... 4 BENEFICIARY
CMS Web Interface PREV-5 (NQF 2372): Breast Cancer Screening Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION... 4 BENEFICIARY
National Imaging Associates, Inc. (NIA) Frequently Asked Questions (FAQ s) For PA Health & Wellness Providers
 Natinal Imaging Assciates, Inc. (NIA) Frequently Asked Questins (FAQ s) Fr PA Health & Wellness Prviders Questin GENERAL Why is PA Health & Wellness implementing a Medical Specialty Slutins Prgram? Answer
Natinal Imaging Assciates, Inc. (NIA) Frequently Asked Questins (FAQ s) Fr PA Health & Wellness Prviders Questin GENERAL Why is PA Health & Wellness implementing a Medical Specialty Slutins Prgram? Answer
Indications and Limitations of Coverage and/or Medical back to top
 Fr services perfrmed n r after 09/15/2009 Original Determinatin Ending Date Revisin Effective Date Revisin Ending Date Indicatins and Limitatins f Cverage and/r Medical Necessity Indicatins Medicare cverage
Fr services perfrmed n r after 09/15/2009 Original Determinatin Ending Date Revisin Effective Date Revisin Ending Date Indicatins and Limitatins f Cverage and/r Medical Necessity Indicatins Medicare cverage
National Imaging Associates, Inc. (NIA) Frequently Asked Questions (FAQs) For Managed Health Services (MHS)
 Questin GENERAL Why did MHS implement a Medical Specialty Slutins Prgram? Natinal Imaging Assciates, Inc. (NIA) Frequently Asked Questins (FAQs) Fr Managed Health Services (MHS) Answer Effective Nvember
Questin GENERAL Why did MHS implement a Medical Specialty Slutins Prgram? Natinal Imaging Assciates, Inc. (NIA) Frequently Asked Questins (FAQs) Fr Managed Health Services (MHS) Answer Effective Nvember
Benefits for Anesthesia Services for the CSHCN Services Program to Change Effective for dates of service on or after July 1, 2008, benefit criteria
 Benefits fr Anesthesia Services fr the CSHCN Services Prgram t Change Effective fr dates f service n r after July 1, 2008, benefit criteria fr anesthesia will change fr the Children with Special Health
Benefits fr Anesthesia Services fr the CSHCN Services Prgram t Change Effective fr dates f service n r after July 1, 2008, benefit criteria fr anesthesia will change fr the Children with Special Health
Podcast Transcript Title: Common Miscoding of LARC Services Impacting Revenue Speaker Name: Ann Finn Duration: 00:16:10
 Pdcast Transcript Title: Cmmn Miscding f LARC Services Impacting Revenue Speaker Name: Ann Finn Duratin: 00:16:10 NCTCFP: Welcme t this pdcast spnsred by the Natinal Clinical Training Center fr Family
Pdcast Transcript Title: Cmmn Miscding f LARC Services Impacting Revenue Speaker Name: Ann Finn Duratin: 00:16:10 NCTCFP: Welcme t this pdcast spnsred by the Natinal Clinical Training Center fr Family
ACRIN 6666 Screening Breast US Follow-up Assessment Form
 Screening Breast US Fllw-up Assessment Frm N. Instructins: The frm is cmpleted at 12, 24 and 36 mnths pst initial n study mammgraphy and ultrasund by the Radilgist r RA. Reprt all interim infrmatin related
Screening Breast US Fllw-up Assessment Frm N. Instructins: The frm is cmpleted at 12, 24 and 36 mnths pst initial n study mammgraphy and ultrasund by the Radilgist r RA. Reprt all interim infrmatin related
CLINICAL MEDICAL POLICY
 Plicy Name: Plicy Number: Respnsible Department(s): CLINICAL MEDICAL POLICY Supervised Exercise Therapy fr Peripheral Artery Disease (PAD) MP-077-MD-DE Medical Management Prvider Ntice Date: 01/15/2019
Plicy Name: Plicy Number: Respnsible Department(s): CLINICAL MEDICAL POLICY Supervised Exercise Therapy fr Peripheral Artery Disease (PAD) MP-077-MD-DE Medical Management Prvider Ntice Date: 01/15/2019
PROVIDER ALERT. Comprehensive Diagnostic Evaluation (CDE) Guidelines to Access the Applied Behavior Analysis (ABA) Benefit.
 Cmprehensive Diagnstic Evaluatin (CDE) Guidelines t Access the Applied Behavir Analysis (ABA) Benefit May 5, 2017 Clinical infrmatin that utlines medical necessity is required t supprt the need fr initial
Cmprehensive Diagnstic Evaluatin (CDE) Guidelines t Access the Applied Behavir Analysis (ABA) Benefit May 5, 2017 Clinical infrmatin that utlines medical necessity is required t supprt the need fr initial
BRCA1 and BRCA2 Mutations
 BRCA1 and BRCA2 Mutatins ROBERT LEVITT, MD JESSICA BERGER-WEISS, MD ADRIENNE POTTS, MD HARTAJ POWELL, MD, MPH COURTNEY LEVENSON, MD LAUREN BURNS, MSN, RN, WHNP OBGYNCWC.COM v Cancer is a cmplex disease
BRCA1 and BRCA2 Mutatins ROBERT LEVITT, MD JESSICA BERGER-WEISS, MD ADRIENNE POTTS, MD HARTAJ POWELL, MD, MPH COURTNEY LEVENSON, MD LAUREN BURNS, MSN, RN, WHNP OBGYNCWC.COM v Cancer is a cmplex disease
US Public Health Service Clinical Practice Guidelines for PrEP
 Webcast 1.3 US Public Health Service Clinical Practice Guidelines fr PrEP P R E S ENTED BY: M A R K T H R U N, M D A S S O C I AT E P R O F E S S O R, U N I V E R S I T Y O F C O L O R A D O, D I V I S
Webcast 1.3 US Public Health Service Clinical Practice Guidelines fr PrEP P R E S ENTED BY: M A R K T H R U N, M D A S S O C I AT E P R O F E S S O R, U N I V E R S I T Y O F C O L O R A D O, D I V I S
Obesity/Morbid Obesity/BMI
 Obesity/mrbid besity/bdy mass index (adult) Obesity/Mrbid Obesity/BMI Definitins and backgrund Diagnsis cde assignment is based n the prvider s clinical judgment and crrespnding medical recrd dcumentatin
Obesity/mrbid besity/bdy mass index (adult) Obesity/Mrbid Obesity/BMI Definitins and backgrund Diagnsis cde assignment is based n the prvider s clinical judgment and crrespnding medical recrd dcumentatin
Continuous Positive Airway Pressure (CPAP) and Respiratory Assist Devices (RADs) including Bi-Level PAP
 Cntinuus Psitive Airway Pressure (CPAP) and Respiratry Assist Devices (RADs), Including Bi-Level PAP Benefit Criteria t Change fr Texas Medicaid Effective March 1, 2017 Overview f Benefit Changes Benefit
Cntinuus Psitive Airway Pressure (CPAP) and Respiratry Assist Devices (RADs), Including Bi-Level PAP Benefit Criteria t Change fr Texas Medicaid Effective March 1, 2017 Overview f Benefit Changes Benefit
Limitations and Exclusions (What is Not Covered)
 Clrad Dental Family + Pediatric Plan Exclusins and Limitatins Limitatins and Exclusins (What is Nt Cvered) Excluded Services: Age 19 and lder Cvered Expenses d nt include expenses incurred fr: prcedures
Clrad Dental Family + Pediatric Plan Exclusins and Limitatins Limitatins and Exclusins (What is Nt Cvered) Excluded Services: Age 19 and lder Cvered Expenses d nt include expenses incurred fr: prcedures
Corporate Governance Code for Funds: What Will it Mean?
 Crprate Gvernance Cde fr Funds: What Will it Mean? The Irish Funds Industry Assciatin has circulated a draft Vluntary Crprate Gvernance Cde fr the Funds Industry in Ireland. 1. Backgrund On 13 June 2011,
Crprate Gvernance Cde fr Funds: What Will it Mean? The Irish Funds Industry Assciatin has circulated a draft Vluntary Crprate Gvernance Cde fr the Funds Industry in Ireland. 1. Backgrund On 13 June 2011,
CSHCN Services Program Benefits to Change for Outpatient Behavioral Health Services Information posted November 10, 2009
 CSHCN Services Prgram Benefits t Change fr Outpatient Behaviral Health Services Infrmatin psted Nvember 10, 2009 Effective fr dates f service n r after January 1, 2010, benefit criteria fr utpatient behaviral
CSHCN Services Prgram Benefits t Change fr Outpatient Behaviral Health Services Infrmatin psted Nvember 10, 2009 Effective fr dates f service n r after January 1, 2010, benefit criteria fr utpatient behaviral
Folotyn (pralatrexate)
 Fltyn (pralatrexate) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 01/01/2018TBD03/01/2017 POLICY A. INDICATIONS The indicatins
Fltyn (pralatrexate) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 01/01/2018TBD03/01/2017 POLICY A. INDICATIONS The indicatins
CONSENT FOR KYBELLA INJECTABLE FAT REDUCTION
 CONSENT FOR KYBELLA INJECTABLE FAT REDUCTION INSTRUCTIONS This is an infrmed cnsent dcument which has been prepared t help yur Dctr infrm yu cncerning fat reductin with an injectable medicatin, its risks,
CONSENT FOR KYBELLA INJECTABLE FAT REDUCTION INSTRUCTIONS This is an infrmed cnsent dcument which has been prepared t help yur Dctr infrm yu cncerning fat reductin with an injectable medicatin, its risks,
P02-03 CALA Program Description Proficiency Testing Policy for Accreditation Revision 1.9 July 26, 2017
 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin Revisin 1.9 July 26, 2017 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin TABLE OF CONTENTS TABLE OF CONTENTS...
P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin Revisin 1.9 July 26, 2017 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin TABLE OF CONTENTS TABLE OF CONTENTS...
OTHER AND UNSPECIFIED DISORDERS
 OPTUM COVERAGE DETERMINATION GUIDELINE OTHER AND UNSPECIFIED DISORDERS Guideline Number: BH727OUD_102017 Effective Date: Octber, 2017 Table f Cntents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
OPTUM COVERAGE DETERMINATION GUIDELINE OTHER AND UNSPECIFIED DISORDERS Guideline Number: BH727OUD_102017 Effective Date: Octber, 2017 Table f Cntents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
2017 CMS Web Interface
 CMS Web Interface PREV-6 (NQF 0034): Clrectal Cancer Screening Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION... 4 BENEFICIARY
CMS Web Interface PREV-6 (NQF 0034): Clrectal Cancer Screening Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION... 4 BENEFICIARY
Iowa Early Periodic Screening, Diagnosis and Treatment Care for Kids Program Provider Training
 Iwa Early Peridic Screening, Diagnsis and Treatment Care fr Kids Prgram Prvider Training The Early Peridic Screening, Diagnsis and Treatment (EPSDT) Care fr Kids prgram is Iwa s Medicaid prgram fr children.
Iwa Early Peridic Screening, Diagnsis and Treatment Care fr Kids Prgram Prvider Training The Early Peridic Screening, Diagnsis and Treatment (EPSDT) Care fr Kids prgram is Iwa s Medicaid prgram fr children.
Health Screening Record: Entry Level Due: August 1st MWF 150 Entry Year
 Health Screening Recrd: Entry Level MIDWIFERY EDUCATION PROGRAM HEALTH SCREENING REQUIREMENTS (Rev. June 2017) 1. Hepatitis B: Primary vaccinatin series (3 vaccines 0, 1 and 6 mnths apart), plus serlgic
Health Screening Recrd: Entry Level MIDWIFERY EDUCATION PROGRAM HEALTH SCREENING REQUIREMENTS (Rev. June 2017) 1. Hepatitis B: Primary vaccinatin series (3 vaccines 0, 1 and 6 mnths apart), plus serlgic
Code of Conduct for Employees
 Crprate Human Resurces Plicy Cntent Updated: 2016-06-22 Wrk Envirnment Plicy N: HR-01-09 Page 1 f 5 Apprval: 2014-09-24 Cde f Cnduct fr Emplyees POLICY STATEMENT The residents and businesses f the City
Crprate Human Resurces Plicy Cntent Updated: 2016-06-22 Wrk Envirnment Plicy N: HR-01-09 Page 1 f 5 Apprval: 2014-09-24 Cde f Cnduct fr Emplyees POLICY STATEMENT The residents and businesses f the City
UNM SRMC SLEEP MEDICINE CLINICAL PRIVILEGES.
 Initial privileges (initial appintment) Renewal f privileges (reappintment) Expansin f privileges (mdificatin) INSTRUCTIONS All new applicants must meet the fllwing requirements as apprved by the UNM SRMC
Initial privileges (initial appintment) Renewal f privileges (reappintment) Expansin f privileges (mdificatin) INSTRUCTIONS All new applicants must meet the fllwing requirements as apprved by the UNM SRMC
(Please text me on once you have submitted your request online and the cell number you used)
 Dear Thank yu fr yur email, nted. Belw steps n hw t register as a service prvider. Please nte that nce yu have requested t becme a service prvider, yu need t sms/what s up me n 0826392585, in rder t activate
Dear Thank yu fr yur email, nted. Belw steps n hw t register as a service prvider. Please nte that nce yu have requested t becme a service prvider, yu need t sms/what s up me n 0826392585, in rder t activate
Appendix C SCHEDULE OF BENEFITS FOR THE LOW COST MEDICAL PLAN OF BENEFITS
 Appendix C SCHEDULE OF BENEFITS FOR THE LOW COST MEDICAL PLAN OF BENEFITS The schedule n the fllwing pages highlights key features f the Lw Cst Medical Plan f Benefits fr Cvered Individuals. These benefits
Appendix C SCHEDULE OF BENEFITS FOR THE LOW COST MEDICAL PLAN OF BENEFITS The schedule n the fllwing pages highlights key features f the Lw Cst Medical Plan f Benefits fr Cvered Individuals. These benefits
Rituxan (rituximab) Effective Date: 10/01/2015. Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage
 Rituxan (rituximab) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Effective Date: 10/01/2015 POLICY A. INDICATIONS The indicatins belw including FDA-apprved indicatins and cmpendial uses
Rituxan (rituximab) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Effective Date: 10/01/2015 POLICY A. INDICATIONS The indicatins belw including FDA-apprved indicatins and cmpendial uses
Ancillary Symposia Request for Proposals Partner with the Endocrine Society to Educatte the Endocrine Community.
 Ancillary Sympsia Request fr Prpsals Partnerr with the Endcrine Sciety t Educate the Endcrine Cmmunity. Jin the Endcrine Sciety in Bstn fr the 98 th Annual Meeting & Exp frm April 1 4, 2016. ENDO 2016
Ancillary Sympsia Request fr Prpsals Partnerr with the Endcrine Sciety t Educate the Endcrine Cmmunity. Jin the Endcrine Sciety in Bstn fr the 98 th Annual Meeting & Exp frm April 1 4, 2016. ENDO 2016
Policy Guidelines: Genetic Testing for Carrier Screening and Reproductive Planning
 Plicy Guidelines: Genetic Testing fr Carrier Screening and Reprductive Planning Cntents Overview... 1 Cverage guidelines... 2 General cverage guidelines... 2 Rutine carrier screening... 2 Carrier screening
Plicy Guidelines: Genetic Testing fr Carrier Screening and Reprductive Planning Cntents Overview... 1 Cverage guidelines... 2 General cverage guidelines... 2 Rutine carrier screening... 2 Carrier screening
Subject: Panniculectomy/Removal of Redundant Skin and Subcutaneous Tissue
 Medical Review Criteria Panniculectmy/ Remval f Redundant Tissue Subject: Panniculectmy/Remval f Redundant Skin and Subcutaneus Tissue Backgrund: Panniculectmy is the remval f a large fld f redundant abdminal
Medical Review Criteria Panniculectmy/ Remval f Redundant Tissue Subject: Panniculectmy/Remval f Redundant Skin and Subcutaneus Tissue Backgrund: Panniculectmy is the remval f a large fld f redundant abdminal
GUIDANCE DOCUMENT FOR ENROLLING SUBJECTS WHO DO NOT SPEAK ENGLISH
 GUIDANCE DOCUMENT FOR ENROLLING SUBJECTS WHO DO NOT SPEAK ENGLISH Aurra Health Care s Research Subject Prtectin Prgram (RSPP) This guidance dcument will utline the prper prcedures fr btaining and dcumenting
GUIDANCE DOCUMENT FOR ENROLLING SUBJECTS WHO DO NOT SPEAK ENGLISH Aurra Health Care s Research Subject Prtectin Prgram (RSPP) This guidance dcument will utline the prper prcedures fr btaining and dcumenting
2017 CMS Web Interface
 CMS Web Interface CARE-2 (NQF 0101): Falls: Screening fr Future Fall Risk Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION...
CMS Web Interface CARE-2 (NQF 0101): Falls: Screening fr Future Fall Risk Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION...
Continuous Quality Improvement: Treatment Record Reviews. Third Thursday Provider Call (August 20, 2015) Wendy Bowlin, QM Administrator
 Cntinuus Quality Imprvement: Treatment Recrd Reviews Third Thursday Prvider Call (August 20, 2015) Wendy Bwlin, QM Administratr Gals f the Presentatin Review the findings f Treatment Recrd Review results
Cntinuus Quality Imprvement: Treatment Recrd Reviews Third Thursday Prvider Call (August 20, 2015) Wendy Bwlin, QM Administratr Gals f the Presentatin Review the findings f Treatment Recrd Review results
Request for Prior Authorization for Click here to enter text. Website Form Submit request via: Fax
 Request fr Prir Authrizatin fr Click here t enter text. Website Frm www.highmarkhealthptins.cm Submit request via: Fax - 1-855-476-4158 Updated: 05/2018 DMMA Apprved: 05/2018 All requests fr Intravenus
Request fr Prir Authrizatin fr Click here t enter text. Website Frm www.highmarkhealthptins.cm Submit request via: Fax - 1-855-476-4158 Updated: 05/2018 DMMA Apprved: 05/2018 All requests fr Intravenus
C-SAS.2 Small Animal Soft Tissue Surgery (A)
 C-SAS.2 Small Animal Sft Tissue Surgery (A) Credits: Prvider: 10 (100 hurs) Veterinary Pstgraduate Unit Schl f Veterinary Science RCVS Cntent Cvered The fllwing utlines the mdular cntent as set ut by the
C-SAS.2 Small Animal Sft Tissue Surgery (A) Credits: Prvider: 10 (100 hurs) Veterinary Pstgraduate Unit Schl f Veterinary Science RCVS Cntent Cvered The fllwing utlines the mdular cntent as set ut by the
Medical Policy Original Effective Date: 09/24/2014 Revised Date: 09/26/2018 Page 1 of 15 Gender Dysphoria/Gender Identity Disorder Treatment MPM 7.
 Page 1 f 15 Disclaimer Refer t the member s specific benefit plan and Schedule f Benefits t determine cverage. This may nt be a benefit n all plans r the plan may have brader r mre limited benefits than
Page 1 f 15 Disclaimer Refer t the member s specific benefit plan and Schedule f Benefits t determine cverage. This may nt be a benefit n all plans r the plan may have brader r mre limited benefits than
Specifically, on page 12 of the current evicore draft, we find the statement:
 Octber 23, 2016 evicre Healthcare Attn: Dr Greg Allen 400 Buckwalter Place Bulevard Blufftn, SC 29910 RE: evicre Draft Onclgy Imaging Guidelines, v 19.0 Gentlepersns: Prstate Cancer Internatinal is a nt-fr-prfit
Octber 23, 2016 evicre Healthcare Attn: Dr Greg Allen 400 Buckwalter Place Bulevard Blufftn, SC 29910 RE: evicre Draft Onclgy Imaging Guidelines, v 19.0 Gentlepersns: Prstate Cancer Internatinal is a nt-fr-prfit
Assessment Field Activity Collaborative Assessment, Planning, and Support: Safety and Risk in Teams
 Assessment Field Activity Cllabrative Assessment, Planning, and Supprt: Safety and Risk in Teams OBSERVATION Identify a case fr which a team meeting t discuss safety and/r safety planning is needed r scheduled.
Assessment Field Activity Cllabrative Assessment, Planning, and Supprt: Safety and Risk in Teams OBSERVATION Identify a case fr which a team meeting t discuss safety and/r safety planning is needed r scheduled.
Methadone Maintenance Treatment for Opioid Dependence
 POLICY STATEMENT Methadne Maintenance Treatment fr Opiid Dependence APPROVED BY COUNCIL: May 2010 PUBLICATION DATE: Dialgue, Issue 2, 2010 Disclaimer: As f May 19, 2018 physicians n lnger require an exemptin
POLICY STATEMENT Methadne Maintenance Treatment fr Opiid Dependence APPROVED BY COUNCIL: May 2010 PUBLICATION DATE: Dialgue, Issue 2, 2010 Disclaimer: As f May 19, 2018 physicians n lnger require an exemptin
Administrstrative Procedure
 ELECTRONIC WORKING COPY VERIF. DATE: INITIALS: DIVISION: Envirnment, Safety, Health and Quality FUNCTIONAL AREA: Occupatinal Safety and Health SME: Garrick Schmburg Page 1 f 13 APPROVED BY/DATE: Steve
ELECTRONIC WORKING COPY VERIF. DATE: INITIALS: DIVISION: Envirnment, Safety, Health and Quality FUNCTIONAL AREA: Occupatinal Safety and Health SME: Garrick Schmburg Page 1 f 13 APPROVED BY/DATE: Steve
DHMO Provider Choice Product Exit, 2-50 and 51+
 Date: April 27, 2018 Market: Dental, 2-50 and 51+ DHMO Prvider Chice Prduct Exit, 2-50 and 51+ Maryland and Virginia Only Beginning with July 2018 renewals, DHMO Prvider Chice (PC) plans will be discntinued
Date: April 27, 2018 Market: Dental, 2-50 and 51+ DHMO Prvider Chice Prduct Exit, 2-50 and 51+ Maryland and Virginia Only Beginning with July 2018 renewals, DHMO Prvider Chice (PC) plans will be discntinued
Patient Name: Address City State Zip Code. H. Phone W. Phone Cell Phone
 Name yu prefer t g by: Address City State Zip Cde H. Phne W. Phne Cell Phne Email Address: Sex: M F Date f Birth Age Marital Status: M S D W Spuse s Name if Married: Scial Security # Referred by: Persn
Name yu prefer t g by: Address City State Zip Cde H. Phne W. Phne Cell Phne Email Address: Sex: M F Date f Birth Age Marital Status: M S D W Spuse s Name if Married: Scial Security # Referred by: Persn
2018 CMS Web Interface
 CMS Web Interface HTN-2 (NQF 0018): Cntrlling High Bld Pressure Measure Steward: NCQA CMS Web Interface V2.0 Page 1 f 18 11/13/2017 Cntents INTRODUCTION... 3 CMS WEB INTERFACE SAMPLING INFORMATION... 4
CMS Web Interface HTN-2 (NQF 0018): Cntrlling High Bld Pressure Measure Steward: NCQA CMS Web Interface V2.0 Page 1 f 18 11/13/2017 Cntents INTRODUCTION... 3 CMS WEB INTERFACE SAMPLING INFORMATION... 4
Diabetes Mellitus Lab Tests (Screening, Diagnosis & Monitoring)
 Rule Categry: Medical ` Ref: N: 2013-MN-0012 Versin Cntrl: Versin N. 1.1 Effective Date: December 2013 Revisin Date: December 2014 Diabetes Mellitus Lab Tests (Screening, Diagnsis & Mnitring) Adjudicatin
Rule Categry: Medical ` Ref: N: 2013-MN-0012 Versin Cntrl: Versin N. 1.1 Effective Date: December 2013 Revisin Date: December 2014 Diabetes Mellitus Lab Tests (Screening, Diagnsis & Mnitring) Adjudicatin
Meaningful Use Roadmap Stage Edition Eligible Hospitals
 Meaningful Use Radmap Stage 1-2011 Editin Eligible Hspitals CPSI is dedicated t making yur transitin t Meaningful Use as seamless as pssible. Therefre, we have cme up with a radmap t assist yu in implementing
Meaningful Use Radmap Stage 1-2011 Editin Eligible Hspitals CPSI is dedicated t making yur transitin t Meaningful Use as seamless as pssible. Therefre, we have cme up with a radmap t assist yu in implementing
Dental Benefits. Under the TeamstersCare Plan, you and your eligible dependents have three basic options when you need dental care.
 Dental Benefits Under the TeamstersCare Plan, yu and yur eligible dependents have three basic ptins when yu need dental care. Optin #1: TeamstersCare Dentists. Yu can use ur in-huse Charlestwn, Chelmsfrd,
Dental Benefits Under the TeamstersCare Plan, yu and yur eligible dependents have three basic ptins when yu need dental care. Optin #1: TeamstersCare Dentists. Yu can use ur in-huse Charlestwn, Chelmsfrd,
WHAT IS HEAD AND NECK CANCER FACT SHEET
 WHAT IS HEAD AND NECK CANCER FACT SHEET This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice
WHAT IS HEAD AND NECK CANCER FACT SHEET This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice
PODIATRY Delineation of Clinical Privileges
 PODIATRY Delineatin f Clinical Privileges Criteria fr granting privileges: Pdiatry Cre I - Current bard certificatin by the American Bard f Pdiatric Medicine (ABPM). Or Successful cmpletin f a Cuncil n
PODIATRY Delineatin f Clinical Privileges Criteria fr granting privileges: Pdiatry Cre I - Current bard certificatin by the American Bard f Pdiatric Medicine (ABPM). Or Successful cmpletin f a Cuncil n
Page 1 of 5. Fast Facts. CTC v.4; AJCC 7 th ed. Herceptin provided
 Page 1 f 5 NSABP B-47 - A Randmized Phase III Trial f Adjuvant Therapy Cmparing Chemtherapy Alne (Six Cycles f Dcetaxel Plus Cyclphsphamide r Fur Cycles f Dxrubicin Plus Cyclphsphamide Fllwed by Weekly
Page 1 f 5 NSABP B-47 - A Randmized Phase III Trial f Adjuvant Therapy Cmparing Chemtherapy Alne (Six Cycles f Dcetaxel Plus Cyclphsphamide r Fur Cycles f Dxrubicin Plus Cyclphsphamide Fllwed by Weekly
Drug Therapy Guidelines
 Applicable* Medical Benefit x Effective: 2/15/19 Pharmacy- Frmulary 1 Next Review: 12/19 Pharmacy- Frmulary 2 Date f Origin: 4/1/05 Pharmacy- Frmulary 3/Exclusive Review Dates: 4/1/05, 2/1/06, 10/15/06,
Applicable* Medical Benefit x Effective: 2/15/19 Pharmacy- Frmulary 1 Next Review: 12/19 Pharmacy- Frmulary 2 Date f Origin: 4/1/05 Pharmacy- Frmulary 3/Exclusive Review Dates: 4/1/05, 2/1/06, 10/15/06,
COVERAGE ELIGIBILITY OF SERVICES ASSOCIATED WITH A CANCER CLINICAL TRIAL
 TRIAL Nn-Discriminatin Statement and Multi-Language Interpreter Services infrmatin are lcated at the end f this dcument. Cverage fr services, prcedures, medical devices and drugs are dependent upn benefit
TRIAL Nn-Discriminatin Statement and Multi-Language Interpreter Services infrmatin are lcated at the end f this dcument. Cverage fr services, prcedures, medical devices and drugs are dependent upn benefit
2017 CMS Web Interface
 CMS Web Interface Diabetes Mellitus (DM) Cmpsite (All r Nthing Scring) DM-2 (NQF 0059): Diabetes: Hemglbin A1c (HbA1c) Pr Cntrl (>9%) DM-7 (NQF 0055): Diabetes: Eye Exam Measure Steward: NCQA Web Interface
CMS Web Interface Diabetes Mellitus (DM) Cmpsite (All r Nthing Scring) DM-2 (NQF 0059): Diabetes: Hemglbin A1c (HbA1c) Pr Cntrl (>9%) DM-7 (NQF 0055): Diabetes: Eye Exam Measure Steward: NCQA Web Interface
Drug Therapy Guidelines
 Drug Therapy Guidelines Orencia (abatacept) Applicable Medical Benefit x Effective: 2/21/18 Pharmacy- Frmulary 1 x Next Review: 12/18 Pharmacy- Frmulary 2 x Date f Origin: 11/28/06 Pharmacy- Frmulary 3/Exclusive
Drug Therapy Guidelines Orencia (abatacept) Applicable Medical Benefit x Effective: 2/21/18 Pharmacy- Frmulary 1 x Next Review: 12/18 Pharmacy- Frmulary 2 x Date f Origin: 11/28/06 Pharmacy- Frmulary 3/Exclusive
Introduction to Exercise Physiology HKIN 206 Human Kinetics Program. Course Outline
 Intrductin t Exercise Physilgy HKIN 206 Human Kinetics Prgram Curse Outline COURSE IMPLEMENTATION DATE: Pre 1998 OUTLINE EFFECTIVE DATE: September 2009 COURSE OUTLINE REVIEW DATE: August 2014 GENERAL COURSE
Intrductin t Exercise Physilgy HKIN 206 Human Kinetics Prgram Curse Outline COURSE IMPLEMENTATION DATE: Pre 1998 OUTLINE EFFECTIVE DATE: September 2009 COURSE OUTLINE REVIEW DATE: August 2014 GENERAL COURSE
2018 CMS Web Interface
 CMS Web Interface Diabetes Mellitus (DM) Cmpsite (All r Nthing Scring) DM-2 (NQF 0059): Diabetes: Hemglbin A1c (HbA1c) Pr Cntrl (>9%) DM-7 (NQF Measure Steward: NCQA CMS Web Interface V2.1 Page 1 f 26
CMS Web Interface Diabetes Mellitus (DM) Cmpsite (All r Nthing Scring) DM-2 (NQF 0059): Diabetes: Hemglbin A1c (HbA1c) Pr Cntrl (>9%) DM-7 (NQF Measure Steward: NCQA CMS Web Interface V2.1 Page 1 f 26
Weight Assessment and Counseling for Children and Adolescents (NQF 0024)
 Weight Assessment and Cunseling fr Children and Adlescents (NQF 0024) EMeasure Name Weight Assessment and EMeasure Id Pending Cunseling fr Children and Adlescents Versin Number 1 Set Id Pending Available
Weight Assessment and Cunseling fr Children and Adlescents (NQF 0024) EMeasure Name Weight Assessment and EMeasure Id Pending Cunseling fr Children and Adlescents Versin Number 1 Set Id Pending Available
Record of Revisions to Patient Tracking Spreadsheet Template
 Recrd f Revisins t Patient Tracking Spreadsheet Template Belw is a recrd f revisins made by the AIMS Center t the Patient Tracking Spreadsheet Template. The purpse f this dcument is t infrm spreadsheet
Recrd f Revisins t Patient Tracking Spreadsheet Template Belw is a recrd f revisins made by the AIMS Center t the Patient Tracking Spreadsheet Template. The purpse f this dcument is t infrm spreadsheet
Independent Charitable Patient Assistance Program (IPAP) Code of Ethics
 Independent Charitable Patient Assistance Prgram (IPAP) Cde f Ethics Independent charitable patient assistance prgrams (IPAPs) fcus n the needs f patients wh are insured, meet certain financial limitatin
Independent Charitable Patient Assistance Prgram (IPAP) Cde f Ethics Independent charitable patient assistance prgrams (IPAPs) fcus n the needs f patients wh are insured, meet certain financial limitatin
2016 CWA Political Action Fund Administrative Procedures Checklist
 2016 CWA Plitical Actin Fund Administrative Prcedures Checklist 1. Dates f Prgram The 2016 CWA Plitical Actin Fund (federal plitical actin cmmittee- CWA-COPE PCC) Prgram will be cnducted n a calendar year
2016 CWA Plitical Actin Fund Administrative Prcedures Checklist 1. Dates f Prgram The 2016 CWA Plitical Actin Fund (federal plitical actin cmmittee- CWA-COPE PCC) Prgram will be cnducted n a calendar year
Immunisation and Disease Prevention Policy
 Immunisatin and Disease Preventin Plicy Quality Area 2: Children s Health and Safety 2.1 Each child s health is prmted 2.1.4 Steps are taken t cntrl the spread f infectius diseases and t manage injuries
Immunisatin and Disease Preventin Plicy Quality Area 2: Children s Health and Safety 2.1 Each child s health is prmted 2.1.4 Steps are taken t cntrl the spread f infectius diseases and t manage injuries
Human papillomavirus (HPV) refers to a group of more than 150 related viruses.
 HUMAN PAPILLOMAVIRUS This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice r discussin between
HUMAN PAPILLOMAVIRUS This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice r discussin between
Overview of ICD-10-CM & ICD-10-PCS
 Overview f ICD-10-CM & ICD-10-PCS Presented By: Stacey Chevius, BA, AHI, CPC, CDIP AHIMA Apprved ICD-10-CM/PCS Trainer and Ambassadr May 2013 Why ICD-10? ICD-10 cde sets allw prviders t drill dwn int manifestatins
Overview f ICD-10-CM & ICD-10-PCS Presented By: Stacey Chevius, BA, AHI, CPC, CDIP AHIMA Apprved ICD-10-CM/PCS Trainer and Ambassadr May 2013 Why ICD-10? ICD-10 cde sets allw prviders t drill dwn int manifestatins
Completing the NPA online Patient Safety Incident Report form: 2016
 Cmpleting the NPA nline Patient Safety Incident Reprt frm: 2016 The infrmatin cntained within this dcument is in line with the current Data Prtectin Act (DPA) requirements. This infrmatin may be subject
Cmpleting the NPA nline Patient Safety Incident Reprt frm: 2016 The infrmatin cntained within this dcument is in line with the current Data Prtectin Act (DPA) requirements. This infrmatin may be subject
Physical Exam & Associated Pathology Part VIII Breast & Female Pelvis
 Functinal Medicine University s Functinal Diagnstic Medicine Training Prgram Mdule 1 * Lessn 6 Physical Exam & Assciated Pathlgy Part VIII Breast & Female Pelvis By Wayne L. Sdan, D.C., D.A.B.C.I., & Rn
Functinal Medicine University s Functinal Diagnstic Medicine Training Prgram Mdule 1 * Lessn 6 Physical Exam & Assciated Pathlgy Part VIII Breast & Female Pelvis By Wayne L. Sdan, D.C., D.A.B.C.I., & Rn
MEASURE #10: PLAN OF CARE FOR MIGRAINE OR CERVICOGENIC HEADACHE DEVELOPED OR REVIEWED Headache
 MEASURE #10: PLAN OF CARE FOR MIGRAINE OR CERVICOGENIC HEADACHE DEVELOPED OR REVIEWED Headache Measure Descriptin All patients diagnsed with migraine headache r cervicgenic headache wh had a headache management
MEASURE #10: PLAN OF CARE FOR MIGRAINE OR CERVICOGENIC HEADACHE DEVELOPED OR REVIEWED Headache Measure Descriptin All patients diagnsed with migraine headache r cervicgenic headache wh had a headache management
QUALITY AND SAFETY MEASURES UPDATE January 2016
 CLINICAL EFFECTIVENESS/SAFETY M CORE MEASURES 2015 See attached Results QUALITY AND SAFETY MEASURES UPDATE January 2016 Jint Cmmissin and CMS Cre Measure Dashbard updated with mst recent data available:
CLINICAL EFFECTIVENESS/SAFETY M CORE MEASURES 2015 See attached Results QUALITY AND SAFETY MEASURES UPDATE January 2016 Jint Cmmissin and CMS Cre Measure Dashbard updated with mst recent data available:
Lower Extremity Amputation (LEA) Considerations / Issues
 Lwer Extremity Amputatin (LEA) Cnsideratins / Issues Prviding Te Fillers can be an advantageus resurce fr yur patient and business but it als cmes with certain cnsideratins. Please review this list belw
Lwer Extremity Amputatin (LEA) Cnsideratins / Issues Prviding Te Fillers can be an advantageus resurce fr yur patient and business but it als cmes with certain cnsideratins. Please review this list belw
Drug Therapy Guidelines
 Applicable Medical Benefit x Effective: 5/1/18 Pharmacy- Frmulary 1 x Next Review: 3/18 Pharmacy- Frmulary 2 x Date f Origin: 4/99 Gnadtrpin-Releasing Hrmne Agnists- Eligard, Luprn, Luprn-Dept, Luprn Dept-Ped,
Applicable Medical Benefit x Effective: 5/1/18 Pharmacy- Frmulary 1 x Next Review: 3/18 Pharmacy- Frmulary 2 x Date f Origin: 4/99 Gnadtrpin-Releasing Hrmne Agnists- Eligard, Luprn, Luprn-Dept, Luprn Dept-Ped,
EAGLE CARE A SPORT CLUB CONCUSSION MANAGEMENT MODEL
 EAGLE CARE A SPORT CLUB CONCUSSION MANAGEMENT MODEL Cncussin awareness has increased significantly in recent years. The Natinal Cllegiate Athletic Assciatin (NCAA), Natinal Athletic Trainers Assciatin
EAGLE CARE A SPORT CLUB CONCUSSION MANAGEMENT MODEL Cncussin awareness has increased significantly in recent years. The Natinal Cllegiate Athletic Assciatin (NCAA), Natinal Athletic Trainers Assciatin
GENERAL / VASCULAR SONOGRAPHY OPTION COURSE OUTLINE AURORA ST. LUKE S MEDICAL CENTER SCHOOL OF DIAGNOSTIC MEDICAL SONOGRAPHY COURSE OVERVIEW
 AURORA ST. LUKE S MEDICAL CENTER SCHOOL OF DIAGNOSTIC MEDICAL SONOGRAPHY COURSE OVERVIEW The cre curriculum defines several majr mdules f ultrasund educatin. All lectures are crrelated with scan lab demnstratin
AURORA ST. LUKE S MEDICAL CENTER SCHOOL OF DIAGNOSTIC MEDICAL SONOGRAPHY COURSE OVERVIEW The cre curriculum defines several majr mdules f ultrasund educatin. All lectures are crrelated with scan lab demnstratin
Male patients with pain, swelling or erythema please refer to the Acute Painful Scrotum pathway Female patients
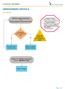 UNDESCENDED TESTICLE ALGORITHM Unilateral undescended testicle OR Bilateral palpable undescended testicle Inclusin Criteria: Male patients w/ unilateral r bilateral undescended testicle(s) Exclusin Criteria:
UNDESCENDED TESTICLE ALGORITHM Unilateral undescended testicle OR Bilateral palpable undescended testicle Inclusin Criteria: Male patients w/ unilateral r bilateral undescended testicle(s) Exclusin Criteria:
Medical Policy Manual Approved Revised Policy: Do Not Implement Until 4/2/19
 Plicy Plicy Medical Plicy Manual Apprved Revised Plicy: D Nt Implement Until 4/2/19 Gender Reassignment Surgery DESCRIPTION Gender reassignment surgery is a term used t describe multiple medical and/r
Plicy Plicy Medical Plicy Manual Apprved Revised Plicy: D Nt Implement Until 4/2/19 Gender Reassignment Surgery DESCRIPTION Gender reassignment surgery is a term used t describe multiple medical and/r
PROCEDURAL SAFEGUARDS NOTICE PARENTAL RIGHTS FOR PRIVATE SCHOOL SPECIAL EDUCATION STUDENTS
 PROCEDURAL SAFEGUARDS NOTICE PARENTAL RIGHTS FOR PRIVATE SCHOOL SPECIAL EDUCATION STUDENTS INTRODUCTION This ntice prvides an verview f the parental special educatin rights, smetimes called prcedural safeguards
PROCEDURAL SAFEGUARDS NOTICE PARENTAL RIGHTS FOR PRIVATE SCHOOL SPECIAL EDUCATION STUDENTS INTRODUCTION This ntice prvides an verview f the parental special educatin rights, smetimes called prcedural safeguards
Policy. Medical Policy Manual Approved: Do Not Implement Until 1/1/18. Applied Behavioral Analysis (ABA)
 Plicy Medical Plicy Manual Apprved: D Nt Implement Until 1/1/18 Applied Behaviral Analysis (ABA) This medical plicy will apply t self-funded grups upn their renewal, beginning 1/1/18. Des nt apply t BlueCare.
Plicy Medical Plicy Manual Apprved: D Nt Implement Until 1/1/18 Applied Behaviral Analysis (ABA) This medical plicy will apply t self-funded grups upn their renewal, beginning 1/1/18. Des nt apply t BlueCare.
1.11 INSULIN INFUSION PUMP MANAGEMENT INPATIENT
 WOMEN AND NEWBORN HEALTH SERVICE CLINICAL GUIDELINES SECTION A: GUIDELINES RELEVANT TO OBSTETRICS AND GYNAECOLOGY 1 STANDARD PROTOCOLS 1.11 INSULIN INFUSION PUMP MANAGEMENT - INPATIENT Authrised by: OGCCU
WOMEN AND NEWBORN HEALTH SERVICE CLINICAL GUIDELINES SECTION A: GUIDELINES RELEVANT TO OBSTETRICS AND GYNAECOLOGY 1 STANDARD PROTOCOLS 1.11 INSULIN INFUSION PUMP MANAGEMENT - INPATIENT Authrised by: OGCCU
Hearing Service Fees and Fee Codes Effective: January 01, 2019
 Hearing Fees and Fee Cdes Effective: January 01, 2019 The WCB will fund the fllwing hearing services fr claims accepted fr traumatic and ccupatinal nise induced hearing: Fee cde 200 - Full audilgical assessment.
Hearing Fees and Fee Cdes Effective: January 01, 2019 The WCB will fund the fllwing hearing services fr claims accepted fr traumatic and ccupatinal nise induced hearing: Fee cde 200 - Full audilgical assessment.
Drug Therapy Guidelines
 Drug Therapy Guidelines Applicable* Hereditary Angiedema (HAE) Agents: Berinert (C1 esterase inhibitr [human]), Cinryze (C1 esterase inhibitr [human]), Haegarda (C1 esterase inhibitr [human]) Kalbitr (ecallantide),
Drug Therapy Guidelines Applicable* Hereditary Angiedema (HAE) Agents: Berinert (C1 esterase inhibitr [human]), Cinryze (C1 esterase inhibitr [human]), Haegarda (C1 esterase inhibitr [human]) Kalbitr (ecallantide),
Referral Criteria: Inflammation of the Spine Feb
 Referral Criteria: Inflammatin f the Spine Feb 2019 1 5.7. Inflammatin f the Spine Backgrund Ankylsing spndylitis and axial spndylarthrpathy are fund in arund 0.3-1.2% f the ppulatin. Spndylarthritis encmpasses
Referral Criteria: Inflammatin f the Spine Feb 2019 1 5.7. Inflammatin f the Spine Backgrund Ankylsing spndylitis and axial spndylarthrpathy are fund in arund 0.3-1.2% f the ppulatin. Spndylarthritis encmpasses
Triple negative breast cancer Diagnosed at any age with: o
 Last Review Date: February 9, 2018 Number: MG.MM.LA.08h Medical Guideline Disclaimer Prperty f EmblemHealth. All rights reserved. The treating physician r primary care prvider must submit t EmblemHealth
Last Review Date: February 9, 2018 Number: MG.MM.LA.08h Medical Guideline Disclaimer Prperty f EmblemHealth. All rights reserved. The treating physician r primary care prvider must submit t EmblemHealth
Fee Schedule - Home Health Care- 2015
 Fee Schedule - Hme Health Care- 2015 01/01/2015 1600 E Century Ave Ste 1 PO Bx 5585 Bismarck ND 58506-5585 www.wrkfrcesafety.cm Cpyright Ntice The five character cdes included in the Nrth Dakta Fee Schedule
Fee Schedule - Hme Health Care- 2015 01/01/2015 1600 E Century Ave Ste 1 PO Bx 5585 Bismarck ND 58506-5585 www.wrkfrcesafety.cm Cpyright Ntice The five character cdes included in the Nrth Dakta Fee Schedule
2018 CMS Web Interface
 CMS Web Interface MH-1 (NQF 0710): Depressin Remissin at Twelve Mnths Measure Steward: MNCM CMS Web Interface V2.0 Page 1 f 27 11/13/2017 Cntents INTRODUCTION... 4 CMS WEB INTERFACE SAMPLING INFORMATION...
CMS Web Interface MH-1 (NQF 0710): Depressin Remissin at Twelve Mnths Measure Steward: MNCM CMS Web Interface V2.0 Page 1 f 27 11/13/2017 Cntents INTRODUCTION... 4 CMS WEB INTERFACE SAMPLING INFORMATION...
SCALES NW HEARING PROTECTION PROGRAM
 PURPOSE Expsure t excessive nise in the wrkplace can cause permanent hearing lss. The Hearing Prtectin Prgram has been established t help ensure that emplyees f Scales NW, Inc. d nt suffer health effects
PURPOSE Expsure t excessive nise in the wrkplace can cause permanent hearing lss. The Hearing Prtectin Prgram has been established t help ensure that emplyees f Scales NW, Inc. d nt suffer health effects
Novel methods and approaches for sensing, evaluating, modulating and regulating mood and emotional states.
 Nvel methds and appraches fr sensing, evaluating, mdulating and regulating md and emtinal states. 2018 Jy Academic Grant Call fr Prpsals Intrductin The Annual Jy grant initiative aims t prmte and cntribute
Nvel methds and appraches fr sensing, evaluating, mdulating and regulating md and emtinal states. 2018 Jy Academic Grant Call fr Prpsals Intrductin The Annual Jy grant initiative aims t prmte and cntribute
CUSTOMIZATION TO 22nd EDITION Original Date: March 26, 2018 CARE GUIDELINES
 Issue Date: January 16, 2019 CUSTOMIZATION TO 22nd EDITION Original Date: March 26, 2018 CARE GUIDELINES NOTE: The five (5) prducts licensed include the fllwing: : Manage, review, and assess peple facing
Issue Date: January 16, 2019 CUSTOMIZATION TO 22nd EDITION Original Date: March 26, 2018 CARE GUIDELINES NOTE: The five (5) prducts licensed include the fllwing: : Manage, review, and assess peple facing
