Stability and Reliability of Detection Thresholds for Human A-Beta and A-Delta Sensory Afferents Determined by Cutaneous Electrical Stimulation
|
|
|
- Bertram Sharp
- 6 years ago
- Views:
Transcription
1 64 Journal of Pain and Symptom Management Vol. 25 No. 1 January 2003 Original Article Stability and Reliability of Detection Thresholds for Human A-Beta and A-Delta Sensory Afferents Determined by Cutaneous Electrical Stimulation Christine N. Sang, MD, MPH, Mitchell B. Max, MD, and Richard H. Gracely, PhD Department of Anesthesia (C.N.S.), Massachusetts General Hospital, Harvard Medical School,Boston, Massachusetts, and Pain and Neurosensory Mechanisms Branch (M.B.M., R.H.G.), National Institute of Dental Research, National Institutes of Health, US Department of Health and Human Services, Bethesda, Maryland, USA Abstract Activity in primary afferent fibers that usually mediate fine touch can evoke sensations of pain in conditions in which there is sensitization of central neurons. Input from these large diameter A afferents may also sustain and exacerbate these central mechanisms. The role of these fibers in clinical pain syndromes can be evaluated by applications of electrical stimuli that preferentially activate A axons. This study assessed the stability and reliability of a method of electrical stimulation (ES) useful for clinical evaluation. Monopolar constantcurrent rectangular pulses were delivered to 5 equi-spaced sites on the volar aspect of the left forearm along a transverse line 5 cm distal to the antecubital crease. Current intensity was gradually increased to determine detection threshold and pain detection threshold. This study determined: 1) Effect of pulse duration (1, 2, and 5 msec); 2) the variation of detection threshold and pain threshold over repeated stimulation; 3) the effect of electrode position with respect to distance from the trunk of underlying ulnar or median nerves; and 4) the effect of repositioning the electrode on variability of detection threshold and pain threshold. There was no significant variability over time for either detection threshold (DT) or pain threshold (PT) at any of the 3 pulse durations tested. There was also no significant effect on variability of shifting the electrode between sites, nor was there a significant difference in variability between sites when placed either over or adjacent to peripheral nerves. Under simulated clinical conditions of electrode re-positioning, the mean detection threshold in 300 trials and ten subjects was 0.30 ma with an overall standard error of 0.007, standard errors of over the 10 subjects, over the 6 trials, and over the 5 locations. Similarly, mean pain threshold in these 300 trials was , with standard errors of 0.12 over the 10 subjects, over the 6 trials, and 0.13 over the 5 locations. Mean ratio of pain threshold divided by detection threshold ratio was with a range of Single pulse, constant current electrical stimulation of the skin at threshold levels is a quantifiable and reliable sensory method that is repeatable within and between testing sessions. Our results Address reprint requests to: Christine N. Sang, MD, MPH, Department of Anesthesia, Clinics 3, Massachusetts General Hospital, 55 Fruit Street, Boston, MA 02114, USA. Accepted for publication: February 25, U.S. Cancer Pain Relief Committee /03/$ see front matter Published by Elsevier. All rights reserved. PII S (02)
2 Vol. 25 No. 1 January 2003 Electrical Stimulation in the Evaluation of Pain 65 suggest that in skin unaffected by allodynia, a ratio of the two sensory thresholds (pain threshold and detection threshold) of less than 2.0 is uncommon. We propose that, in the presence of mechanical allodynia, a pain threshold/detection threshold of less than 2.0 suggests that altered central nervous system processing of A input may contribute to allodynia. J Pain Symptom Manage 2003;25: U.S. Cancer Pain Relief Committee. Published by Elsevier. All rights reserved. Key Words Electric stimulation, pain threshold, pain measurement, sensory thresholds, primary afferent, hyperesthesia Introduction Quantitative sensory testing (QST) is proving to be an indispensable tool to advance the classification of specific disorders 1 3 and illuminating underlying mechanisms of chronic pain. QST uses controlled sensory stimulation to extend components of the neurological examination to evaluate the function of primary afferents that mediate innocuous and painful sensation, and central processes that further modify the character and sensitivity of these primary afferents. A striking feature in many neuropathic and inflammatory pain conditions is mechanical allodynia, during which lightly touching the skin evokes an intense pain sensation. Calibrated nylon monofilaments are commonly used to quantify sensations evoked by controlled punctate pressure, although the adequate stimulus in many conditions may involve movement, or temporal summation such as provided by a vibratory stimulus. Unfortunately, mechanical stimuli identify the presence of mechanical allodynia, but do not identify the neural substrate mediating the allodynia. Pain triggered by light touch may be due to either altered central processing of A low-threshold mechanoreceptor (A LTM) input or activation of sensitized peripheral nociceptors. A LTM involvement in mechanical allodynia has been inferred from several lines of evidence with methods using electrical stimuli, such as intraneural stimulation, 4 cutaneous stimulation over a nerve supplying an area of spontaneous pain, 5,6 and cutaneous stimulation distant from any major nerves. 7 Inferences about mechanisms of dynamic mechanical allodynia from data in humans have been based on results of studies using transcutaneous electrical stimulation to differentiate between A and A stimulation These methods are based on the assumption that, since the largest fibers (A LTMs) are the most sensitive to electrical stimulation, they are the only fibers activated at detection-threshold levels of stimulation. 14 As stimulus intensity is increased further, A fibers are ultimately recruited and C-fibers are activated at even higher intensities. A -mediated allodynia is identified when electrical current evokes pain at or close to detection threshold, a current strength which activates only A fibers and causes an innocuous tactile sensation under normal conditions. In addition, the activation circumvents receptor transduction and thus is independent of receptor processes such as sensitization or suppression. 2 Thus, because activation of A LTMs occurs at currents well below those needed to activate A and C axons, Price et al. 6 and Gracely et al. 7 have proposed the use of electrical stimulation of the skin to identify A -mediated mechanical allodynia. ES has been useful to detect mechanisms of mechanical allodynia since it is noninvasive method to activate afferent fibers by direct depolarization of axons. Several laboratories have taken advantage of this property and used ES to distinguish whether mechanical allodynia is due to sensitization of central neurons or sensitization of peripheral receptors; 3 5,7,15 for example, if the faintest detectable current strength, which only activates A LTM fibers, causes pain, central nervous system mechanisms are implicated. Cutaneous electrical stimulation has demonstrated additional potential diagnostic utility in several studies. Pain threshold and pain tolerance measures have been selectively reduced in syndromes such as temporomandibular dysfunction, cervico-brachial syndrome, and fibro-
3 66 Sang et al. Vol. 25 No. 1 January 2003 myalgia. 11,16,17 Recent human and animal models of neural inflammation have shown increased sensitivity to A -strength electrical stimulation that coincides with the time course of the inflammation. 18,19 This accumulating evidence for clinical utility is matched by methodological advantages. Notermans 20 proposed the use of ES to determine pain threshold because 1) it is easily expressed in physical terms (milliamperes, ma); 2) it has little chance of damaging tissues; and 3) it produces an easily recognizable and easily definable pricking pain sensation. Although ES is a simple non-invasive psychophysical method, a number of factors have hindered its acceptance as a tool in pain research. The practical application of electrical stimulation as a diagnostic method in individual patients depends critically on reliability; variation in skin resistance and local physiological changes can potentially alter the results of electrical testing. 21,22 Like other sensory methods involving threshold measurements, this method is potentially limited by interaction with time or threshold intensity. Thus the basic stability and repeatability of electrical skin stimulation must be assessed if the method is to gain wide clinical use as a diagnostic sensory test. This study examined the influence of pulse duration, and both temporal and spatial variability at cutaneous sites distant from major nerves or at sites directly over major nerves. The methods simulated a clinical assessment: the skin was not pretreated, constant current stimuli were delivered through commercially available saline sponge electrodes using an ascending Method of Limits, and, in Experiment III, electrodes were removed and reapplied for each trial. left forearm along a transverse line 5 cm distal to the antecubital crease (Figure 1): 1) 1.2 to 3.5 cm (depending on forearm diameter) medial to the ulnar nerve; 2) over the ulnar nerve; 3) midway between the ulnar and median nerve; 4) over the median nerve; and 5) 1.2 to 3.5 cm lateral to the median nerve. Sites 2 and 4 were identified by moderate intensity electrical stimulation evoked sensations projected to the hypothenar eminence and midpalm, respectively. Monopolar constant-current rectangular pulses were delivered at 0.5 Hz through bipolar 6 mm diameter commercially available (Dantek, Medtronic) saline-sponge electrodes spaced 23 mm apart (center-to-center), applied along the axis of the limb with the cathode directed proximally. Subjects were seated with their arms resting outwards and supinated, and electrodes were applied at the level of the heart. 23 To avoid stimulation of nociceptors at suprathreshold intensities, an ascending series of stimulus intensities was delivered in 0.01 ma steps to each site, starting at zero and gradually increasing until a sensation was evoked (DT) and further increased in increments of ma until the sensation changed to definite pain (PT). This sequence constituted a DT-PT trial. Subjects were also asked to describe the sensations evoked by the electrical stimuli at detection and pain thresholds. Experiment I. Temporal Stability and Effect of Pulse Duration Coupling of an electrical conductor to the peripheral nervous system involves a number Methods Ten healthy normal volunteers (age 23 35) were studied in each of three experiments, evaluated under uniform conditions including ambient noise, moisture, seating, and time of day. The electrically evoked pain threshold is not influenced by skin temperature, especially when the ambient temperature is maintained between 20 and 25 C. 20 The ambient temperature in the current study was maintained within 23 1 C for all experiments. Five equi-spaced sites were identified on a selected area of skin on the volar aspect of the Fig. 1. Schematic of left arm marked by five equispaced sites. Sites 2 and 4 lie over the a) ulnar and b) median nerves, respectively.
4 Vol. 25 No. 1 January 2003 Electrical Stimulation in the Evaluation of Pain 67 of interfaces and resistive-capacitive elements. As a result, the effects of narrow pulses are variably diminished, while the cumulative effects of monopolar stimulation, which is accentuated by long pulse durations, results in polarization that also diminishes the effects of stimulation. Although the use of constant current stimulation controls for varying resistances, constant current methods do not control for the adverse effects of capacitive elements. Experiment I assessed the influence of these capacitive elements by determining the effect of pulse duration on variability of detection threshold and pain threshold. Stimuli with pulse durations of 1, 2, and 5 msec were delivered at 0.5 Hz to the site midway between the ulnar and median nerves (Site 3). Ten DT-PT trials were determined at each pulse duration. The shortest pulse duration that demonstrated maximal sensitivity with minimal variance would likely offer the best compromise between the adverse effects of short and long pulse durations. Experiment II. Temporal Stability and Spatial Variability Without Effect of Repositioning, Effect of Electrode Site In previous studies, we were concerned that, due to spatial variability in sensitivity, attempts to reposition the electrode at the same location would introduce unacceptable variability. To test the best case variance, and to assess whether phenomena such as habituation and sensitization affect measurement of detection and pain thresholds, DT and PT were determined by delivering 2 msec stimuli to each of the 5 sites at 0.5 Hz. This experiment also addressed the difference in sensitivity at defined spatial locations: over, adjacent, and between major cutaneous nerves. Each DT-PT trial was administered at each site 6 times in succession without moving the electrode. Experiment III. Effect of Repositioning the Electrode and Electrode Site This experiment replicated the clinical situation in which sensitivity is evaluated before and after a pain control intervention, or evaluated in longitudinal measurements of disease progression. To determine whether different sites have different detection and pain thresholds, and whether repositioning the electrode caused more variability than leaving it in the same place (as in Experiment III), each DT-PT trial was administered by delivering 2 msec stimuli to each of the 5 sites. Each subject was randomized to a different sequence of the 5 sites (based on a 5 5 Latin square design), with the same sequence of sites repeated 6 times. Statistical Analysis Data were processed using Stata 4.0 (College Station, TX). Thresholds were computed from the mean of 10 (Experiment I) or 6 (Experiments II and III) complete trials. The PT to DT ratios (PT:DT) were determined for each set of sensory thresholds. Data were analyzed using one-way ANOVA to evaluate the effect of pulse duration, site, and repositioning, and repeated measures ANOVA to evaluate temporal variability. Analysis by Shapiro-Wilk test 24 indicated that departures from normality were insignificant and would meet the assumptions for an ANOVA. In addition, Bartlett s Test 25 demonstrated homogeneity of variances for each set of variables analyzed by ANOVA. When normality was an issue, parametric data analysis was supplemented by nonparametric analysis. Matched-Sample Sign Test was used to determine equality of medians for those subjects who returned for both Experiments I and II. A probability value of less than 0.05 was considered to be significant. Coefficients of determination (R 2 ) for the regression models were adjusted for unnecessary variables in the model. Results Each of the subjects described the sensation at detection threshold as a tingle, tap, or flutter, and the sensation at pain threshold as a pinprick, sting, or electric shock that ceased immediately at termination of the stimulus. Using intraoperative nerve trunk stimulation and recording, Collins 14 observed that A activation produced pricking sensations lasting for the duration of the stimulus, while C-fiber activation produced prolonged burning pain. Collins also observed that the current required to activate A fibers was considerably less than that needed to activate C-fibers. Thus, we inferred that our subjects reports of pain sensation evoked by the least stimulus current represented activation of A fibers without C-fiber involvement. A total of 20 subjects participated in three experiments, each involving 10 subjects. Two subjects participated in all three experiments,
5 68 Sang et al. Vol. 25 No. 1 January subjects participated in Experiments I and II, and 4 subjects participated in Experiments II and III. Experiment I. Effect of Pulse Duration on Detection Threshold and Pain Threshold Figure 2A shows the detection and pain thresholds at three different pulse durations. Each data point represents the mean threshold of the 10 trials for each individual subject at that pulse duration. There was a statistically significant inverse relationship between pulse duration and threshold for both detection (P 0.01) and pain (P 0.01). This figure indicates that the 2-msec duration offered the best compromise between sensitivity, shortest pulse duration, and minimal variability. Figure 2B presents the ratios of pain to detection thresholds at the three different pulse durations. For these 10 subjects, for all pulse durations, mean PT:DT ratio was never less than 6.4. Fig. 3. Experiment II. Time course of detection and pain thresholds for each site averaged over the 10 subjects. Each series of connected data points represents 6 successive determinations approximately 5 minutes apart of DT and PT, without moving the electrode. Z1 through Z5 correspond to sites 1 5 in Figure 1. Experiment II. Time Course of Detection and Pain Thresholds and Effect of Location The 2-msec pulse duration was chosen for the subsequent experiments. Figure 3 shows the time course of the detection and pain thresholds. Each series of connected data points represented 6 successive determinations of DT and PT for each specific site averaged over the ten subjects. There was no significant change over time for either DT or PT at any of the 5 sites. Figure 4 shows the ratio of the pain threshold to detection threshold plotted against detection threshold for the 300 pair of responses (5 sites 6 repetitions 10 subjects). Subjects are identified by unique plot symbols. These data show that a majority of the detection Fig. 2. Experiment I. Effect of pulse duration on detection threshold and pain threshold. A: Sensory thresholds at three different pulse durations. The solid points represent the mean PT s; the hollow points represent the mean DT s. Detection threshold error bars are too small to be visible. B: Ratios of pain to detection thresholds at three different pulse durations. Each data point represents the mean ( standard error) PT:DT ratio for the 10 subjects. Fig. 4. Pain/Detection (PT:DT) ratios for all trials in Experiment II plotted against detection threshold. Data from each subject are shown by a unique symbol. Each subject received 6 DT and PT trials at each of 5 locations for a total of 30 trials for each of the 10 subjects. The line indicates the maximum possible ratio with a stimulation limit of 10 ma. Data from 3 subjects with both detection thresholds greater than 8 and ratios near the limit are classified as outliers.
6 Vol. 25 No. 1 January 2003 Electrical Stimulation in the Evaluation of Pain 69 thresholds were less than 0.8 ma and the ratios were always greater than 2.0. Inspection of the figure indicates outlier data with detection thresholds greater than 0.8 ma and high ratios resulting from pain thresholds near the maximum stimulus intensity of 10 ma. These data represented the results from a single site in two subjects, and from all sites in a third subject. Mean detection threshold, including these outlier data, was 0.48 ma with a standard error of for all 300 data points. Eliminating the outlier subjects reduces the mean and standard error to In the absence of significant differences, the influence of time or location can be evaluated by averaging these points over either subjects or the independent variables and computing standard errors that permit comparison of these sources of variability. These standard errors were over the 10 subjects, over the 6 trials, and over the 5 locations (0.036 subjects, trials, location without outliers). Similarly, mean pain threshold of all of the data was 4.70 ma with an overall standard error of 0.14 and standard errors of 0.12 over the 10 subjects, over the 6 trials, and 0.32 over the 5 locations. Without outliers the mean and standard error were , and the standard errors by source were for subjects, for trials, and 0.22 for location. Experiment III. Effect of Site and Repositioning on Detection Threshold and Pain Threshold Figure 5 shows the sensory thresholds at the five different sites. Each data point represents the average of 60 observations (6 DT-PT determinations from 10 subjects) at that site. There was no significant effect of site on either detection threshold or pain threshold. Moreover, there was no significant difference in variability between sites when placed over the nerves and elsewhere. However, inspection of the figures shows a trend for increased sensitivity over the nerve sites. This difference approached significance for both the detection (P 0.059) and pain (P 0.069) thresholds when the data were collapsed to nerve and non-nerve sites. Figure 6 shows the ratio of the pain threshold to detection threshold plotted against detection threshold for the 300 pair of responses collected in Experiment III. All detection thresholds were 0.7 ma or less and the mean pain/ detection threshold ratio was with Fig. 5. Experiment III. Effect of site on detection threshold and pain threshold. A: Sensory thresholds at the 5 sites (depicted in Figure 1). The hollow points represent mean DT s; the solid points represent mean PT s. B: Ratios of pain to detection thresholds at the 5 sites. Each data point represents the mean PT:DT ratio for each individual subject. a range from Mean detection threshold was 0.30 ma with an overall standard error of and standard errors of over the 10 subjects, over the 6 trials, and over the 5 locations. Similarly, mean pain threshold of all of the data was 3.24 ma with an overall standard error of and standard errors of 0.12 over the 10 subjects, over the 6 trials, and 0.13 over the 5 locations. Comparison of the standard errors indicates that the thresholds were more stable over time than over location. Correlations supported this difference. The correlations among all possible pairs of the 6 trials averaged over the five locations varied from (mean 0.943) for detection thresholds and (mean 0.97) for pain thresholds. In contrast, correlations among all possible pairs of the five locations, averaged over the six trials, ranged from (mean 0.56) for detection thresholds and (mean 0.88) for pain thresholds. Thus thresholds were highly repeatable after repositioning the electrode but varied among location possibly due in part to the influence of underlying nerves. Detection and pain thresholds in Experiment III were compared using both differences and ratios. The mean differences between DT and PT at the 5 sites were 4.20, 3.42, 3.75, 3.22, and 3.30 ma. The mean PT:DT ratios at the 5 sites were 12.7, 12.3, 11.9, 11.8, and 9.7 (medial to lateral). In all three experiments, the mean ( SEM) PT:DT ratio in 900 trials at 5 sites in 20 subjects and 30 visits was 11.7 ( 0.6) (range ). In all subjects, detection threshold current intensity at sites 2 and 4 (directly over ulnar and
7 70 Sang et al. Vol. 25 No. 1 January 2003 Fig. 6. Pain/Detection (PT:DT) ratios for all trials in Experiment III plotted against detection threshold. Data from each subject are shown by a unique symbol. Each subject received 6 DT and PT trials at each of 5 locations for a total of 30 trials for each of the 10 subjects. The line indicates the maximum possible ratio with a stimulation limit of 10 ma. Data from 3 subjects with both detection thresholds greater than 0.8 ma and ratios near the limit are classified as outliers. median nerves) evoked identical concurrent sensations referred to the hypothenar eminence and midpalm, respectively. In 210/240 trials (17/20 subjects) involving stimulation at these sites, the development of pain at the referred sites preceded the development of pain at the sites of contact with the electrodes, confirming that axons in passage are activated at lower current strengths than are local nociceptors. Discussion These results support the reliability of electrical stimulation at cutaneous sites both over and distant from peripheral nerves. There was no significant difference in detection thresholds at nerve and non-nerve sites. However, a trend of increased sensitivity over nerves suggests a subtle difference that may be demonstrated with a greater number of subjects. In addition to possible increased sensitivity, the nerve sites demonstrated a qualitatively different response. Stimulation at nerve sites evoked non-painful sensations at both the proximal electrode site and distally at the receptive field of the stimulated fibers. The threshold for detection at these two locations were similar. This equivalence suggests that the distribution of the A axons mediating the sensations is such that their sensitivity to electrical stimulation of the skin surface is similar at both distal end near the receptor and more proximally in the nerve trunk. However, if stimulating currents are increased to levels that evoke pain, this correspondence disappears. Pain is detected first in the distal referred region, and at higher levels, at the location of the electrode. We are not certain of the reason for this pattern of pain radiation. A possible explanation might be that when the electrode is placed over a nerve, the nociceptor axons within the nerve that innervate a distant site may be larger in diameter or more densely packed in the region activated by the electric current than the nociceptive axons that innervate the skin and soft tissue directly under the electrode. Larger fibers are more sensitive to electrical stimulation, while a dense concentration of fibers allows stimulation of a sufficient population required for perception of pain. These results support the strategy of testing the thresholds and evoked qualities of A stimulation by stimulating the proximal nerve innervating the test region. 5,6 This method avoids contact with the skin in the test regions and thus eliminates the confounds associated with this contact, such as touch-evoked pain observed in cases mechanical-allodynia. Inferences drawn from electrical stimulation of human skin depend on sufficient reliability, which until now has not been evaluated systematically. Temporal variability may be introduced by a number of mechanisms. Although low-intensity electrical stimulation activates afferent axons directly, bypassing receptor mechanisms, 2 habituation and sensitization may still influence the results of ES. 26,27 Electrical stimulation may introduce local physiological changes which may modify the ionic microenvironment and result in changes in skin resistance or impedance. 28,29 Similar to previous experiments, this study used constant current stimuli; hence, the intensity of the stimulating current was independent of skin resistance but not of impedance. 20 Tursky and Greenblatt 30 describe the electrode-skin circuit as two high impedance electrode-skin junctions connected by a subsurface low impedance network, whose configuration changes to a low impedance cir-
8 Vol. 25 No. 1 January 2003 Electrical Stimulation in the Evaluation of Pain 71 cuit with the administration of an electrical stimulus. This is at least in part due to dilatation of local blood vessels resulting from the axon reflex 30,31 or the presence of perspiration which may provide a low resistance path through the skin, or act as a lateral current path to shunt current along the surface of the skin. 30 Results of this study show that estimation of DT and PT by electrical skin stimulation is not significantly affected by potential sensitization, habituation, galvanic skin response, or alterations in the electrical microenvironment. Spatial variability could be introduced by electrode repositioning or use of an electrode site close to the location of a nerve. This may be due to either histological variation of vessels and other factors contributing to differential skin resistance, or to neurohistological differences between sites. 22 Results from this study indicate that the reliability of electrical skin stimulation is independent of site. Excessive variability was observed in Experiment II in 3 subjects, 2 of whom participated in Experiment III and showed normal responses. This variability was due to insensitive trials that were confined to a single location in 2 subjects and found at all locations in 1 subject. This insensitivity could represent a region of hypesthesia in the 2 subjects and generalized hyperesthesia or a response bias in the third subject. A common and likely explanation is that the insensitivity was due to the preparation, such as shunting of the current across the surface of the skin because of perspiration, drying of the saline electrodes, or excessive skin oil (and high impedance) at particular sites. We monitored skin impedance in Experiment I but discontinued monitoring in Experiments II and III to more closely match a clinical evaluation. Impedance monitoring in future studies would distinguish between such effects, although the necessity of such monitoring would depend on the desired comparisons. The present data indicate that the PT:DT ratios are still greater than 2.0 in these outlier trials and pain evoked at a ratio less than this ratio would still indicate A involvement. Our data indicate that investigation of trials with detection thresholds greater than 0.8 ma would likely provide sufficient quality assurance in a clinical application. The finding that pain is never evoked by a current less than twice that required to detect sensation validates the claim in previous reports 5,6,15 that if electrical stimuli at detection threshold produces pain, A low threshold mechanoreceptors (A LTMs) would be implicated in the mechanism of these painful sensations. The finding that the ratios of pain threshold to detection threshold exceed 2.0 in each trial of every subject is consistent with findings by Collins et al. 14 that considerably higher current is required to activate A and C nociceptors. These results support the inference that, in patients with pain evoked by detection threshold intensities of cutaneous electrical stimulation, altered central nervous system processing is responsible for the misinterpretation of innocuous stimuli as pain. 5,7 It is possible that this rule may not apply to patients with nerve injury, as this study and other supportive data were derived from patients without nerve injury in the area of stimulation. Additional studies in a variety of clinical pain syndromes associated with allodynia are needed. The clinical utility of A assessment is not limited to cases of mechanical allodynia. Increasing evidence supports A LTM involvement in inflammatory pain. Application of inflammatory agents such as carrageenan and Freund s complete adjuvant to paws or nerve trunks of rats results in decreased withdrawal latencies to A LTM activation produced by mechanical or electrical stimulation (Eliav, personal communication). In studies of paw inflammation, both innocuous touch stimuli and noxious pinch stimulation produced greater neural and motor reflex responses. 32 In addition to increased A sensitivity, these studies also indicate that A fibers may contribute to the maintenance of the sensitized state. Gentle mechanical stroking or A -strength electrical stimulation increases A sensitivity further, and effect described as progressive tactile hypersensitivity. 19 Under these conditions of inflammation A fibers appear to switch phenotype, releasing substance P at their terminals and behaving like nociceptors in maintaining central sensitivity. 33 In these studies, electrical stimuli are used to both evaluate A function and as an adequate A stimulus in evoking progressive tactile hypersensitivity. Electrical stimuli are ideally suited for this latter role since stimulation is easily applied and quantified in comparison to the mechanical method of gentle stroking. Exclusive stimulation of A afferents may also reveal effects not found
9 72 Sang et al. Vol. 25 No. 1 January 2003 in situations in which punctate stimuli are effective. In the rat chronic constriction model, monofilament stimulation reveals a consistent hypersensitivity, while electrical stimuli reveal a complex response of both hyper- and hyposensitivity (Eliav, personal communication). Our laboratory has recently found clinical evidence consistent with A involvement in inflammatory pain. 18 In an extensive sensory assessment of oral surgical patients at the time (48 hrs) of maximum postoperative inflammation, we observed increased sensitivity to electrical (and mechanical) stimuli applied to both the nerve territory of a single extracted lower third molar tooth and to territory of an adjacent nerve whose trunk was exposed to post extraction inflammation. Mechanical and electrical detection thresholds were normal in other divisions of the trigeminal nerve. Thermal testing of both detection and pain thresholds to cold and heat showed no effects at any location. These results suggest that the sole sensory sign of inflammatory pain may be an increased sensitivity that is restricted to A LTM afferents. Two other groups of investigators have reported a narrower spread than our mean ratio of 10:1 between group means of electrical pain and detection thresholds. Working with normals and patients with fibromyalgia and chronic shoulder and neck pain, Arroyo and Cohen, 16,17 Sheather-Reid and Cohen, 9 and Lautenbacher et al. 11 described ratios of between 1.5:1 and 3:1 at different skin sites. None of these articles reported pain/detection threshold ratios for individual patients. At least part of this difference may be due to differences in stimulation frequency. All of these other studies used 100 Hz trains of electrical pulses, while we gave single pulses spaced at two-second intervals. Our choice was based on our previous work with electrical stimulation of dental pulp. 34 Detection threshold in that study was constant across a broad range of frequencies, but pain threshold showed a U-shaped plot for current versus frequency, in which the lowest pain threshold and the narrowest spread between pain and detection threshold occurred at 100 Hz. The greatest spread occurred at 5 Hz, the lowest frequency tested. Single pulse constant current electrical skin stimulation is a robust technique for investigating the mechanisms of pain symptoms. Further studies are needed to confirm whether single pulses give a greater separation between pain and detection threshold than rapid trains of pulses. The limited temporal and spatial variability in young healthy subjects support its use as a quantitative sensory assessment tool. It can bypass receptors, stimulating A LTMs (or all A afferents) independently of peripheral processing such as receptor sensitization. It is quantifiable and easily applied with safe, battery-operated stimulators. Other sources of variability of electrical pain or detection thresholds that we did not explore (for example, age, sex, body size, or prolonged time intervals between testing sessions) may be considered in future studies. This mildly painful, atraumatic, noninvasive method may provide a useful adjunct to conventional QST assessment of painful conditions. Acknowledgments The authors thank Drs. Gary Bennett and Ian Gilron for reviewing the manuscript, and Fred Brown for his technical assistance. References 1. Bennett GJ. Neuropathic pain. In: Wall PD, Melzack R, eds. Textbook of Pain. Edinburgh: Churchill Livingstone, 1994: Gracely RH. Studies of pain in normal man. In: Wall, PD, Melzack R, eds. Textbook of pain. Edinburgh: Churchill Livingstone, 1994: Gracely RH, Price DD, Roberts WJ, Bennett GJ. Quantitative sensory testing in patients with CRPS-I & II. In: Janig W, Stanton-Hicks M, eds. Reflex sympathetic dystrophy a reappraisal, Seattle: IASP Press, 1996: Torebjork HE, Lundberg LER, LaMotte RH. Central changes in processing of mechanoreceptive input in capsaicin-induced secondary hyperalgesia in humans. J Physiol 1992;448: Price DD, Bennett GJ, Rafii A. Psychophysical observations on patients with neuropathic pain relieved by a sympathetic block. Pain 1989;36: Price DD, Long S, Huit C. Sensory testing of pathophysiological mechanisms of pain in patients with reflex sympathetic dystrophy. Pain 1992;49: Gracely RH, Lynch SA, Bennett GJ. Painful neuropathy: altered central processing maintained dynamically by peripheral input. Pain 1992;51: Wasner G, Baron R, Janig W. Dynamic mechanical allodynia in humans is not mediated by a central presynaptic interaction of A beta-mechanoreceptive and nociceptive C-afferents. Pain 1999;79:
10 Vol. 25 No. 1 January 2003 Electrical Stimulation in the Evaluation of Pain Sheather-Reid RB, Cohen ML. Psychophysical evidence for a neuropathic component of chronic neck pain. Pain 1998;75: Affaitati G, Vecchiet F, Lerza R, et al. Effects of topical diclofenac on skin, subcutis and muscle pain thresholds in subjects without spontaneous pain. Drugs Exp Clin Res 2001;27: Lautenbacher S, Rollman GB, McCain GA. Multimethod assessment of experimental and clinical pain in patients with fibromyalgia. Pain 1994;59: Giamberardino MA, Berkley KJ, Iezzi S, et al. Pain threshold variations in somatic wall tissues as a function of menstrual cycle, segmental site and tissue depth in non-dysmenorrheic women, dysmenorrheic women and men. Pain 1997;71: Enggaard TP, Klitgaard NA, Gram LF, et al. Specific effect of venlafaxine on single and repetitive experimental painful stimuli in humans. Clin Pharmacol Ther 69: , Collins WF Jr. Relation of peripheral nerve fiber size and sensation in man. Arch Neurol 1960;3: Max MB, Byas-Smith MG, Gracely RH, Bennett GJ. Intravenous infusion of the NMDA antagonist, ketamine, in chronic posttraumatic pain with allodynia: a double-blind comparison to alfentanil and placebo. Clin Neuropharmacol 1995;18: Arroyo JF, Cohen ML. Unusual responses to electrocutaneous stimulation in refractory cervicobracial pain: clues to a neuropathic pathogenesis. Clin Exp Rheumatol 1992;10: Arroyo JF, Cohen ML. Abnormal responses to electrocutaneous stimulation in fibromyalgia. J Rheumatol 1992;20: Eliav E, Gracely RH. Sensory changes in the territory of the lingual and inferior alveolar nerves following lower third molar extraction. Pain 1998;77: Ma QP, Woolf CJ. Progressive tactile hypersensitivity: an inflammation-induced incremental increase in the excitability of the spinal cord. Pain 1996;67: Notermans SLH. Measurement of the pain threshold determined by electrical stimulation and its clinical application. Neurology 1966:16; Greenblatt DJ, Tursky B. Local vascular and impedance changes induced by electric shock. Am J Physiol 1969;4: Tursky B. Physical, physiological, and psychological factors that affect pain reaction to electric shock Psychophysiology 1974;11: Weinman J, Manoach M. A photoelectric approach to the study of peripheral circulation. Am Heart J 1962;63: Shapiro SS, Wilk MB. Biometrika 1965;52: Bartlett MS. Properties of sufficiency and statistical tests. Proc Royal Soc Ser A 1937;160: Price DD, Hu JW, Dubner R, Gracely RH. Peripheral suppression of first pain and central summation of second pain evoked by noxious heat pulses. Pain 1977;3: Ernst M, Lee MHM, Dworkin B, Zaretsky HH. Pain perception decrement produced through repeated stimulation. Pain 1986;26; Mueller EE, Loeffel R, Mead S. Skin impedance in relation to pain threshold testing by electrical means. J Applied Physiol 1953;5: Tursky B, Watson PD. Controlled physical and subjective intensities of electric shock. Psychophysiology 1964;1: Tursky B, Greenblatt DJ. Local vascular and thermal changes that accompany electric shock. Psychophysiology 1967;3: Lewis T, Grant RT. Vascular reactions of the skin to injury, II. Heart 1924;11: Ma QP, Woolf CJ, Tachykinin NK. 1 receptor antagonist RP67580 attenuates progressive hypersensitivity of flexor reflex during experimental inflammation in rats. Eur J Pharmacol 1997;322: Neumann S, Doubell TP, Leslie T, Woolf CJ. Inflammatory pain hypersensitivity mediated by phenotypic switch in myelinated primary sensory neurons. Nature 1996;384: McGrath PA, Gracely RH, Dubner R, Heft MW. Non-pain and pain sensations evoked by tooth pulp stimulation. Pain 1983;15:
Update on the Neurophysiology of Pain Transmission and Modulation: Focus on the NMDA-Receptor
 S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Sensory Assessment of Regional Analgesia in Humans
 REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Corporate Medical Policy
 Corporate Medical Policy Intravenous Anesthetics for the Treatment of Chronic Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravenous_anesthetics_for_the_treatment_of_chronic_pain
Corporate Medical Policy Intravenous Anesthetics for the Treatment of Chronic Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravenous_anesthetics_for_the_treatment_of_chronic_pain
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Nervous system Reflexes and Senses
 Nervous system Reflexes and Senses Physiology Lab-4 Wrood Slaim, MSc Department of Pharmacology and Toxicology University of Al-Mustansyria 2017-2018 Nervous System The nervous system is the part of an
Nervous system Reflexes and Senses Physiology Lab-4 Wrood Slaim, MSc Department of Pharmacology and Toxicology University of Al-Mustansyria 2017-2018 Nervous System The nervous system is the part of an
Prof Wayne Derman MBChB,BSc (Med)(Hons) PhD, FFIMS. Pain Management in the Elite Athlete: The 2017 IOC Consensus Statement
 Prof Wayne Derman MBChB,BSc (Med)(Hons) PhD, FFIMS Pain Management in the Elite Athlete: The 2017 IOC Consensus Statement 2 as 20 Experts published and leaders in their respective field 12 month lead in
Prof Wayne Derman MBChB,BSc (Med)(Hons) PhD, FFIMS Pain Management in the Elite Athlete: The 2017 IOC Consensus Statement 2 as 20 Experts published and leaders in their respective field 12 month lead in
Somatosensory modalities!
 Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
Compound Action Potential, CAP
 Stimulus Strength UNIVERSITY OF JORDAN FACULTY OF MEDICINE DEPARTMENT OF PHYSIOLOGY & BIOCHEMISTRY INTRODUCTION TO NEUROPHYSIOLOGY Spring, 2013 Textbook of Medical Physiology by: Guyton & Hall, 12 th edition
Stimulus Strength UNIVERSITY OF JORDAN FACULTY OF MEDICINE DEPARTMENT OF PHYSIOLOGY & BIOCHEMISTRY INTRODUCTION TO NEUROPHYSIOLOGY Spring, 2013 Textbook of Medical Physiology by: Guyton & Hall, 12 th edition
Pain teaching. Muhammad Laklouk
 Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
SYLLABUS SPRING 2011 COURSE: NSC NEUROBIOLOGY OF PAIN
 SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
Quantitative Electrophysiology
 ECE 795: Quantitative Electrophysiology Notes for Lecture #10 Wednesday, November 22, 2006 14. FUNDAMENTALS OF FUNCTIONAL ELECTRICAL STIMULATION (FES) We will look at: Design issues for FES Subthreshold
ECE 795: Quantitative Electrophysiology Notes for Lecture #10 Wednesday, November 22, 2006 14. FUNDAMENTALS OF FUNCTIONAL ELECTRICAL STIMULATION (FES) We will look at: Design issues for FES Subthreshold
MANAGEMENT OF CRPS. Brachial Neuralgia n = 81 patients. CRPS type II n = 126 patients CONTENTS. Neuropathic Pain. Introduction.
 MANAGEMENT OF CRPS Paradoxical Hypoaesthetic Skin, Painful to Touch: A Target to relieve Neuropathic Pain Spicher, C.J. et al. (2010) Paris: Sauramps medical Paradoxical Hypoaesthetic Skin, Painful to
MANAGEMENT OF CRPS Paradoxical Hypoaesthetic Skin, Painful to Touch: A Target to relieve Neuropathic Pain Spicher, C.J. et al. (2010) Paris: Sauramps medical Paradoxical Hypoaesthetic Skin, Painful to
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes
 The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes 1 Medical Hypothesis 2007, Vol. 69, pp. 1169 1178 Sota Omoigui
The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes 1 Medical Hypothesis 2007, Vol. 69, pp. 1169 1178 Sota Omoigui
Muscle Weakness Or Paralysis With Compromise Of Peripheral Nerve
 Muscle Weakness Or Paralysis With Compromise Of Peripheral Nerve Muscle weakness or complete paralysis may be secondary to an interruption in the communication between the brain and nerve cells in the
Muscle Weakness Or Paralysis With Compromise Of Peripheral Nerve Muscle weakness or complete paralysis may be secondary to an interruption in the communication between the brain and nerve cells in the
EE 791 Lecture 2 Jan 19, 2015
 EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
Bi/CNS/NB 150: Neuroscience. November 11, 2015 SOMATOSENSORY SYSTEM. Ralph Adolphs
 Bi/CNS/NB 150: Neuroscience November 11, 2015 SOMATOSENSORY SYSTEM Ralph Adolphs 1 Menu for today Touch -peripheral -central -plasticity Pain 2 Sherrington (1948): senses classified as --teloreceptive
Bi/CNS/NB 150: Neuroscience November 11, 2015 SOMATOSENSORY SYSTEM Ralph Adolphs 1 Menu for today Touch -peripheral -central -plasticity Pain 2 Sherrington (1948): senses classified as --teloreceptive
TRANSCUTANEOUS ELECTRICAL STIMULATION
 TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
Cellular Bioelectricity
 ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture #30 Thursday, March 30, 2006 Nerve excitation: To evaluate the pattern of nerve activation that is produced by a particular electrode configuration,
ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture #30 Thursday, March 30, 2006 Nerve excitation: To evaluate the pattern of nerve activation that is produced by a particular electrode configuration,
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
Chapter 14: The Cutaneous Senses
 Chapter 14: The Cutaneous Senses Somatosensory System There are three parts Cutaneous senses - perception of touch and pain from stimulation of the skin Proprioception - ability to sense position of the
Chapter 14: The Cutaneous Senses Somatosensory System There are three parts Cutaneous senses - perception of touch and pain from stimulation of the skin Proprioception - ability to sense position of the
Pain and Touch. Academic Press. Edited by Lawrence Kruger. Department of Neurobiology University of California, Los Angeles Los Angeles, California
 Pain and Touch Edited by Lawrence Kruger Department of Neurobiology University of California, Los Angeles Los Angeles, California San Diego New York Sydney Academic Press London Boston Tokyo Toronto Contributors
Pain and Touch Edited by Lawrence Kruger Department of Neurobiology University of California, Los Angeles Los Angeles, California San Diego New York Sydney Academic Press London Boston Tokyo Toronto Contributors
Pain. Types of Pain. Types of Pain 8/21/2013
 Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
How strong is it? What is it? Where is it? What must sensory systems encode? 9/8/2010. Spatial Coding: Receptive Fields and Tactile Discrimination
 Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination
 Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Chapter 13 & 14 The Peripheral Nervous System & the Autonomic Nervous System. Nervous System Organization
 Chapter 13 & 14 The Peripheral Nervous System & the Autonomic Nervous System 49 slides 1 Nervous System Organization Figure 13.1 Place of the PNS in the structural organization of the nervous system. 2
Chapter 13 & 14 The Peripheral Nervous System & the Autonomic Nervous System 49 slides 1 Nervous System Organization Figure 13.1 Place of the PNS in the structural organization of the nervous system. 2
CHAPTER 10 THE SOMATOSENSORY SYSTEM
 CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
By the end of this lecture the students will be able to:
 UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
Motor and sensory nerve conduction studies
 3 rd Congress of the European Academy of Neurology Amsterdam, The Netherlands, June 24 27, 2017 Hands-on Course 2 Assessment of peripheral nerves function and structure in suspected peripheral neuropathies
3 rd Congress of the European Academy of Neurology Amsterdam, The Netherlands, June 24 27, 2017 Hands-on Course 2 Assessment of peripheral nerves function and structure in suspected peripheral neuropathies
MEDICAL POLICY SUBJECT: KETAMINE INFUSION THERAPY FOR THE TREATMENT OF CHRONIC PAIN SYNDROMES POLICY NUMBER: CATEGORY: Technology Assessment
 Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus. MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive
 PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
GENERAL PAIN DEFINITIONS
 I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING FOR PAIN MANAGEMENT
 JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acute Pain 2 Neuropathic
JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acute Pain 2 Neuropathic
Neuropathic Pain in Palliative Care
 Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Electrophysiological Assessment of the Cutaneous Arborization of A -Fiber Nociceptors
 Electrophysiological Assessment of the Cutaneous Arborization of A -Fiber Nociceptors YUAN B. PENG, 1 MATTHIAS RINGKAMP, 1 JAMES N. CAMPBELL, 1,2 AND RICHARD A. MEYER 1,2 Departments of 1 Neurosurgery
Electrophysiological Assessment of the Cutaneous Arborization of A -Fiber Nociceptors YUAN B. PENG, 1 MATTHIAS RINGKAMP, 1 JAMES N. CAMPBELL, 1,2 AND RICHARD A. MEYER 1,2 Departments of 1 Neurosurgery
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject
 Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
 University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
Pathophysiological Classification of Pain
 PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
TRANSCUTANEOUS ELECTRICAL NERVE STIMULATION (TENS) Dr. Mohammed TA, Omar, PhD, PT Rehabilitation Science Department CAMS-KSU
 TRANSCUTANEOUS ELECTRICAL NERVE STIMULATION (TENS) Dr. Mohammed TA, Omar, PhD, PT Rehabilitation Science Department CAMS-KSU momarar@ksu.edu.sa Definition of TENS and current specifications Modes of TENS
TRANSCUTANEOUS ELECTRICAL NERVE STIMULATION (TENS) Dr. Mohammed TA, Omar, PhD, PT Rehabilitation Science Department CAMS-KSU momarar@ksu.edu.sa Definition of TENS and current specifications Modes of TENS
During a class experiment conducted by one of the authors in England in 1949
 59 J Physiol. (I953) I22, 59-65 ACTIVITY OF HUMAN SWEAT GLANDS DURING EXPOSURE TO COLD BY E. M. GLASER AND T. S. LEE From the Department of Physiology, University of Malaya, Singapore (Received 9 February
59 J Physiol. (I953) I22, 59-65 ACTIVITY OF HUMAN SWEAT GLANDS DURING EXPOSURE TO COLD BY E. M. GLASER AND T. S. LEE From the Department of Physiology, University of Malaya, Singapore (Received 9 February
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
What is Pain? An unpleasant sensory and emotional experience associated with actual or potential tissue damage. Pain is always subjective
 Pain & Acupuncture What is Pain? An unpleasant sensory and emotional experience associated with actual or potential tissue damage. NOCICEPTION( the neural processes of encoding and processing noxious stimuli.)
Pain & Acupuncture What is Pain? An unpleasant sensory and emotional experience associated with actual or potential tissue damage. NOCICEPTION( the neural processes of encoding and processing noxious stimuli.)
Guide to the use of nerve conduction studies (NCS) & electromyography (EMG) for non-neurologists
 Guide to the use of nerve conduction studies (NCS) & electromyography (EMG) for non-neurologists What is NCS/EMG? NCS examines the conduction properties of sensory and motor peripheral nerves. For both
Guide to the use of nerve conduction studies (NCS) & electromyography (EMG) for non-neurologists What is NCS/EMG? NCS examines the conduction properties of sensory and motor peripheral nerves. For both
Variety of muscle responses to tactile stimuli
 Variety of muscle responses to tactile stimuli Julita Czarkowska-Bauch Department of Neurophysiology, Nencki Institute of Experimental Biology, 3 Pasteur St., 02-093 Warsaw, Poland Abstract. Influences
Variety of muscle responses to tactile stimuli Julita Czarkowska-Bauch Department of Neurophysiology, Nencki Institute of Experimental Biology, 3 Pasteur St., 02-093 Warsaw, Poland Abstract. Influences
ANATOMY AND PHYSIOLOGY OF NEURONS. AP Biology Chapter 48
 ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
Peripheral Nervous System
 Peripheral Nervous System 1 Sensory Receptors Sensory Receptors and Sensation Respond to changes (stimuli) in the environment Generate graded potentials that can trigger an action potential that is carried
Peripheral Nervous System 1 Sensory Receptors Sensory Receptors and Sensation Respond to changes (stimuli) in the environment Generate graded potentials that can trigger an action potential that is carried
Razi Kittaneh & Leen Osama. Marah Bitar. Mohammad Khatatbeh
 11 Razi Kittaneh & Leen Osama Marah Bitar Mohammad Khatatbeh Notes on the previous lecture o Spatial summation: input (postsynaptic potentials) from multiple presynaptic neurons. These postsynaptic potentials
11 Razi Kittaneh & Leen Osama Marah Bitar Mohammad Khatatbeh Notes on the previous lecture o Spatial summation: input (postsynaptic potentials) from multiple presynaptic neurons. These postsynaptic potentials
Chapter 7. Objectives
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7. The Nervous System: Structure and Control of Movement
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
susceptibility of either the axons in the dorsal and ventral roots, or the intramedullary
 213 J. Physiol. (31958) I40, 2I3-2I9 THE SITE OF ACTION OF PROCAINE ON THE ISOLATED SPINAL CORD OF THE FROG BY M. HARMEL AND J. L. MALCOLM From the Department of Physiology, State University of New York,
213 J. Physiol. (31958) I40, 2I3-2I9 THE SITE OF ACTION OF PROCAINE ON THE ISOLATED SPINAL CORD OF THE FROG BY M. HARMEL AND J. L. MALCOLM From the Department of Physiology, State University of New York,
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM
 STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
Accelerating the Development of Enhanced Pain Treatments March 25, Bermuda
 Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Proof-of-concept trials Ian Gilron, MD,
Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Proof-of-concept trials Ian Gilron, MD,
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Testing the gate-control theory of pain in man
 Journal of Neurology, Neurosurgery, and Psychiatry, 1974, 37, 1366-1372 Testing the gate-control theory of pain in man P. W. NATHAN' AND P. RUDGE From the National Hospital, Queen Square, London SYNOPSIS
Journal of Neurology, Neurosurgery, and Psychiatry, 1974, 37, 1366-1372 Testing the gate-control theory of pain in man P. W. NATHAN' AND P. RUDGE From the National Hospital, Queen Square, London SYNOPSIS
Nerve. (2) Duration of the stimulus A certain period can give response. The Strength - Duration Curve
 Nerve Neuron (nerve cell) is the structural unit of nervous system. Nerve is formed of large numbers of nerve fibers. Types of nerve fibers Myelinated nerve fibers Covered by myelin sheath interrupted
Nerve Neuron (nerve cell) is the structural unit of nervous system. Nerve is formed of large numbers of nerve fibers. Types of nerve fibers Myelinated nerve fibers Covered by myelin sheath interrupted
Various Types of Pain Defined
 Various Types of Pain Defined Pain: The International Association for the Study of Pain describes pain as, An unpleasant sensory and emotional experience associated with actual or potential tissue damage,
Various Types of Pain Defined Pain: The International Association for the Study of Pain describes pain as, An unpleasant sensory and emotional experience associated with actual or potential tissue damage,
Pathophysiology of Pain
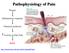 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Scheminzky's phenomenon was attempted by studying the actions of galvanic. Scheminzky (see Scheminzky, 1940, 1947, and the papers quoted therein) has
 316 J. Physiol. (I95I) II3, 3I6-32I EFFECTS OF DIRECT CURRENTS ON THE ELECTRICAL ACTIVITY OF THE SPINAL CORD BY C. AJMONE MARSAN, M. G. F. FUORTES AND F. MAROSSERO From the Clinica Malattie Nervose e Mentali,
316 J. Physiol. (I95I) II3, 3I6-32I EFFECTS OF DIRECT CURRENTS ON THE ELECTRICAL ACTIVITY OF THE SPINAL CORD BY C. AJMONE MARSAN, M. G. F. FUORTES AND F. MAROSSERO From the Clinica Malattie Nervose e Mentali,
Assay Sensitivity.
 Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
Department of Neurology/Division of Anatomical Sciences
 Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Human Anatomy - Problem Drill 11: The Spinal Cord and Spinal Nerves
 Human Anatomy - Problem Drill 11: The Spinal Cord and Spinal Nerves Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper as needed,
Human Anatomy - Problem Drill 11: The Spinal Cord and Spinal Nerves Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper as needed,
MYOFASCIAL PAIN. Dr. Janet Travell ( ) credited with bringing MTrPs to the attention of healthcare providers.
 Myofascial Trigger Points background info Laurie Edge-Hughes BScPT, MAnimSt (Animal Physio), CAFCI, CCRT History lesson Dr. Janet Travell (1901 1997) credited with bringing MTrPs to the attention of healthcare
Myofascial Trigger Points background info Laurie Edge-Hughes BScPT, MAnimSt (Animal Physio), CAFCI, CCRT History lesson Dr. Janet Travell (1901 1997) credited with bringing MTrPs to the attention of healthcare
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D.
 Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Coding of Sensory Information
 Coding of Sensory Information 22 November, 2016 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Sensory Systems Mediate Four Attributes of
Coding of Sensory Information 22 November, 2016 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Sensory Systems Mediate Four Attributes of
SOMATOSENSORY SYSTEMS
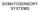 SOMATOSENSORY SYSTEMS Schematic diagram illustrating the neural pathways that convey somatosensory information to the cortex and, subsequently, to the motor system. Double arrows show reciprocal connections.
SOMATOSENSORY SYSTEMS Schematic diagram illustrating the neural pathways that convey somatosensory information to the cortex and, subsequently, to the motor system. Double arrows show reciprocal connections.
Nervous System. Master controlling and communicating system of the body. Secrete chemicals called neurotransmitters
 Nervous System Master controlling and communicating system of the body Interacts with the endocrine system to control and coordinate the body s responses to changes in its environment, as well as growth,
Nervous System Master controlling and communicating system of the body Interacts with the endocrine system to control and coordinate the body s responses to changes in its environment, as well as growth,
Trans-spinal direct current stimulation: a novel tool to promote plasticity in humans
 Trans-spinal direct current stimulation: a novel tool to promote plasticity in humans Jean-Charles Lamy, PhD Brain and Spine Institute, Paris 1 Background Grecco et al., J Neuroresto, 2015 2 Background:
Trans-spinal direct current stimulation: a novel tool to promote plasticity in humans Jean-Charles Lamy, PhD Brain and Spine Institute, Paris 1 Background Grecco et al., J Neuroresto, 2015 2 Background:
Diabetic Complications Consortium
 Diabetic Complications Consortium Application Title: Cathepsin S inhibition and diabetic neuropathy Principal Investigator: Nigel A Calcutt 1. Project Accomplishments: We investigated the efficacy of cathepsin
Diabetic Complications Consortium Application Title: Cathepsin S inhibition and diabetic neuropathy Principal Investigator: Nigel A Calcutt 1. Project Accomplishments: We investigated the efficacy of cathepsin
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Sensory Analgesia. Pain Definitions a distressing feeling due to disease, bodily injury or organic disorder. uneasiness of mind or grief.
 Sensory Analgesia Anesthesia- Analgesia- Partial or complete loss of sensation with or without loss of consciousness Relieving pain, being in a state without pain Pain Definitions a distressing feeling
Sensory Analgesia Anesthesia- Analgesia- Partial or complete loss of sensation with or without loss of consciousness Relieving pain, being in a state without pain Pain Definitions a distressing feeling
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle.
 What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
Mechanical Allodynia Definition, Assessment and Treatment
 See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/272093027 Mechanical Allodynia Definition, Assessment and Treatment Conference Paper June 2010
See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/272093027 Mechanical Allodynia Definition, Assessment and Treatment Conference Paper June 2010
Principles of Electrical Currents. HuP 272
 Principles of Electrical Currents HuP 272 Electricity is an element of PT modalities most frightening and least understood. Understanding the basis principles will later aid you in establishing treatment
Principles of Electrical Currents HuP 272 Electricity is an element of PT modalities most frightening and least understood. Understanding the basis principles will later aid you in establishing treatment
CRITICALLY APPRAISED PAPER (CAP) Evidence / Title of article
 CRITICALLY APPRAISED PAPER (CAP) Evidence / Title of article Sensory findings after stimulation of the thoracolumbar fascia with hypertonic saline suggest its contribution to low back pain Schilder A et
CRITICALLY APPRAISED PAPER (CAP) Evidence / Title of article Sensory findings after stimulation of the thoracolumbar fascia with hypertonic saline suggest its contribution to low back pain Schilder A et
Skin types: hairy and glabrous (e.g. back vs. palm of hand)
 Lecture 19 revised 03/10 The Somatic Sensory System Skin- the largest sensory organ we have Also protects from evaporation, infection. Skin types: hairy and glabrous (e.g. back vs. palm of hand) 2 major
Lecture 19 revised 03/10 The Somatic Sensory System Skin- the largest sensory organ we have Also protects from evaporation, infection. Skin types: hairy and glabrous (e.g. back vs. palm of hand) 2 major
Impact of Demyelination Disease on Neuronal Networks
 Impact of Demyelination Disease on Neuronal Networks Sandeep Adem Chiyuan Chang Mark Fleming sadem@eng.ucsd.edu chc418@eng.ucsd.edu m3flemin@eng.ucsd.edu 1. Abstract Demyelination has a detrimental impact
Impact of Demyelination Disease on Neuronal Networks Sandeep Adem Chiyuan Chang Mark Fleming sadem@eng.ucsd.edu chc418@eng.ucsd.edu m3flemin@eng.ucsd.edu 1. Abstract Demyelination has a detrimental impact
Sensory information processing, somato-sensory systems
 mm? Sensory information processing, somato-sensory systems Recommended literature 1. Kandel ER, Schwartz JH, Jessel TM (2000) Principles of Neural Science, McGraw-Hill, Ch. xx. 2. Berne EM, Levy MN, Koeppen
mm? Sensory information processing, somato-sensory systems Recommended literature 1. Kandel ER, Schwartz JH, Jessel TM (2000) Principles of Neural Science, McGraw-Hill, Ch. xx. 2. Berne EM, Levy MN, Koeppen
POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS
 J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
Basic Neuroscience. Sally Curtis
 The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
Superficial Cold And Heat Application During Transcutaneous Electrical Stimulation May Not Change Perceived Sensation Of Pain
 ISPUB.COM The Internet Journal of Pain, Symptom Control and Palliative Care Volume 9 Number 1 Superficial Cold And Heat Application During Transcutaneous Electrical Stimulation May Not Change H Jelinek,
ISPUB.COM The Internet Journal of Pain, Symptom Control and Palliative Care Volume 9 Number 1 Superficial Cold And Heat Application During Transcutaneous Electrical Stimulation May Not Change H Jelinek,
The Somatosensory System
 The Somatosensory System Reading: BCP Chapter 12 cerebrovortex.com Divisions of the Somatosensory System Somatosensory System Exteroceptive External stimuli Proprioceptive Body position Interoceptive Body
The Somatosensory System Reading: BCP Chapter 12 cerebrovortex.com Divisions of the Somatosensory System Somatosensory System Exteroceptive External stimuli Proprioceptive Body position Interoceptive Body
PART IV: NEUROPATHIC PAIN SYNDROMES JILL SINDT FEBRUARY 7, 2019
 PART IV: NEUROPATHIC PAIN SYNDROMES JILL SINDT FEBRUARY 7, 2019 NEUROPATHIC PAIN PAIN ARISING AS DIRECT CONSEQUENCE OF A LESION OR DISEASE AFFECTING THE SOMATOSENSORY SYSTEM AFFECTS 3-8% OF POPULATION
PART IV: NEUROPATHIC PAIN SYNDROMES JILL SINDT FEBRUARY 7, 2019 NEUROPATHIC PAIN PAIN ARISING AS DIRECT CONSEQUENCE OF A LESION OR DISEASE AFFECTING THE SOMATOSENSORY SYSTEM AFFECTS 3-8% OF POPULATION
راما ندى أسامة الخضر. Faisal Muhammad
 22 راما ندى أسامة الخضر Faisal Muhammad Revision Last time we started talking about sensory receptors, we defined them and talked about the mechanism of their reaction. Now we will talk about sensory receptors,
22 راما ندى أسامة الخضر Faisal Muhammad Revision Last time we started talking about sensory receptors, we defined them and talked about the mechanism of their reaction. Now we will talk about sensory receptors,
(Received 10 April 1956)
 446 J. Physiol. (I956) I33, 446-455 A COMPARISON OF FLEXOR AND EXTENSOR REFLEXES OF MUSCULAR ORIGIN BY M. G. F. FUORTES AND D. H. HUBEL From the Department ofneurophysiology, Walter Reed Army Institute
446 J. Physiol. (I956) I33, 446-455 A COMPARISON OF FLEXOR AND EXTENSOR REFLEXES OF MUSCULAR ORIGIN BY M. G. F. FUORTES AND D. H. HUBEL From the Department ofneurophysiology, Walter Reed Army Institute
Chapter 11: Functional Organization of Nervous Tissue
 Chapter 11: Functional Organization of Nervous Tissue I. Functions of the Nervous System A. List and describe the five major nervous system functions: 1. 2. 3. 4. 5. II. Divisions of the Nervous System
Chapter 11: Functional Organization of Nervous Tissue I. Functions of the Nervous System A. List and describe the five major nervous system functions: 1. 2. 3. 4. 5. II. Divisions of the Nervous System
Vibrotactile masking:,a comparison of psychophysical procedures
 Perception & Psychophysics 1977, Vol. 22 (5), 471-475 Vibrotactile masking:,a comparison of psychophysical procedures RITA E. SNYDER Indiana University, Bloomington, Indiana 47401 The amounts of ipsilateral
Perception & Psychophysics 1977, Vol. 22 (5), 471-475 Vibrotactile masking:,a comparison of psychophysical procedures RITA E. SNYDER Indiana University, Bloomington, Indiana 47401 The amounts of ipsilateral
Virtually everyone has experienced pain in one
 Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms
 DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
Ameen Alsaras. Ameen Alsaras. Mohd.Khatatbeh
 9 Ameen Alsaras Ameen Alsaras Mohd.Khatatbeh Nerve Cells (Neurons) *Remember: The neural cell consists of: 1-Cell body 2-Dendrites 3-Axon which ends as axon terminals. The conduction of impulse through
9 Ameen Alsaras Ameen Alsaras Mohd.Khatatbeh Nerve Cells (Neurons) *Remember: The neural cell consists of: 1-Cell body 2-Dendrites 3-Axon which ends as axon terminals. The conduction of impulse through
3/7/2018. IASP updated definition of pain. Nociceptive Pain. Transduction. (Nociceptors) Transmission. (Peripheral nerve) Modulation
 IASP updated definition of pain The Pain of Trauma and The Trauma of Pain: The Opioid Crisis is Not What You Think. Bennet Davis, M.D. Many people report pain in the absence of tissue damage or any likely
IASP updated definition of pain The Pain of Trauma and The Trauma of Pain: The Opioid Crisis is Not What You Think. Bennet Davis, M.D. Many people report pain in the absence of tissue damage or any likely
