Treatment of Genital Psoriasis: A Systematic Review
|
|
|
- Adelia Simmons
- 5 years ago
- Views:
Transcription
1 Dermatol Ther (Heidelb) (2018) 8: REVIEW Treatment of Genital Psoriasis: A Systematic Review Kristen M. Beck. Eric J. Yang. Isabelle M. Sanchez. Wilson Liao Received: June 1, 2018 / Published online: August 2, 2018 Ó The Author(s) 2018 ABSTRACT Genital affects approximately 63% of patients at least once in their lifetime. More than any other area on the body, genital lesions significantly impair patients psychologic well-being and quality of life. We aimed to systematically review the published evidence on the safety, efficacy, and tolerability of treatments of genital and synthesize the available clinical data. A total of 1 randomized controlled trial, 11 open-label studies, and 26 case reports were included in our analysis, representing a total of 8 patients, of which 332 were adults and 126 were children. Topical corticosteroids were commonly used first-line for genital and were well tolerated. Nonsteroidal agents, such as topical calcineurin inhibitors or vitamin D analogs, were also efficacious, but were often irritating. One systemic agent, ixekizumab, demonstrated efficacy in Kristen M. Beck and Eric J. Yang contributed equally to the work. Enhanced digital features To view enhanced digital features for this article go to m9.figshare K. M. Beck (&) E. J. Yang I. M. Sanchez W. Liao Department of Dermatology, UCSF Medical Center, San Francisco, CA, USA Kristen.Beck@ucsf.edu reducing genital symptoms in a large, randomized, placebo-controlled trial. Various systemic and topical medications may improve genital lesions, but there is a lack of high-quality evidence to guide clinical decisionmaking. Specific reporting of efficacy for genital in larger controlled studies of treatments are necessary to improve the available evidence regarding the optimal treatment regimen for genital. Keywords: Genital ; Psoriasis; Therapy; Treatment INTRODUCTION Sixty-three percent of adults with develop psoriatic lesions in the genital area at least once during their lifetime [1]. In the presence of inverse, the prevalence of genital increases to approximately 79% [2, 3]. In 2 % of patients, lesions only occur in the genital region [3]. Genital can occur in all age groups, from newborns to geriatric patients, with a slight predilection for younger male patients with relatively severe disease []. In children under 2 years of age, genital typically presents as intense well-demarcated erythema in the diaper area, termed napkin [, 6].
2 10 Dermatol Ther (Heidelb) (2018) 8:09 2 Patients with lesions in their genital area experience significantly worse quality of life than patients with on any other areas, particularly with respect to pruritus, sexual function, sexual health, sexual distress, avoidance of sexual relationships, embarrassment, shame, and psychologic depression [, 7 9]. Despite the high prevalence of genital, almost half of patients with genital lesions do not discuss their symptoms with their physician [8, 10, 11]. Lack of communication and awareness about genital in the healthcare environment may result in underdiagnosed and under-treated genital, subsequently increasing the risk of inappropriate self-treatment [10]. Although there are many effective treatments in the therapeutic armamentarium for, treatment of lesions on the genitalia and surrounding skin folds remains challenging. Topical treatments have increased penetration through the thin, sensitive, and often occluded genital skin, increasing the risk of side effects from common medications [12]. A review of the literature in 2011 found a lack of evidence regarding the efficacy and safety of various treatments for genital [3]. This review summarizes the most current literature regarding the efficacy and safety of available therapies for affecting the genital skin. METHODS A literature search using the MEDLINE and Embase health literature databases was conducted using the terms ( ) AND ( penile OR penis OR genital* OR glans OR scrotal OR scrotum OR anal OR diaper OR napkin OR shaft OR foreskin OR prepuce OR perianal OR vulva* OR labia OR labium OR groin OR preputial OR penoscrotal ). Two independent reviewers identified the included articles (EY, IS) and extracted information. The review methods were established prior to the conduct of the review and did not deviate during the course of the study. Articles were included if they included patients with or psoriasiform napkin dermatitis affecting the genital area, discussed the results of treatment with respect to the genital lesions, and were published prior to 31 July Studies were excluded if they did not study genital, did not discuss the results of treatment in the genital region, were in a foreign language, were conference abstracts, or were a review or meta-analysis. Study results were extracted using a standardized data form recording information on the publication date, type of, site, age and gender of patients, and reported efficacy and adverse events. In addition, the quality of all studies was rated based on the Oxford Centre for Evidence-Based Medicine Levels of Evidence rating scheme [13]. The article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors. RESULTS A total of 196 potentially relevant unique citations were identified from our literature search (Fig. 1). Of these, 128 articles were selected for further evaluation based on the relevance of their title and abstract. A total of 32 articles examining the treatment of genital were ultimately included in this study. Table 1 summarizes the data for adults. Table 2 summarizes the findings for infants and children. Treatments for Genital Psoriasis in Adults A total of 21 articles examined treatment outcomes for adults with genital (Table 1), including 1 randomized controlled trial (grade 1), 8 open-label studies (grade ), and 16 case reports (grade ), representing a total of 332 patients. Of these, topical corticosteroids were used in the regimen of 201 patients for successful treatment (Table 2). Low-potency topical steroids were used in 132 patients, moderatepotency steroids were used in 120 patients, and high-potency steroids were used in 1 patients. Antifungal medications were used as part of
3 Dermatol Ther (Heidelb) (2018) 8: Fig. 1 Genital : study selection for treatment successful treatment in 3 patients, while coal tar preparations were effective in clearing genital lesions in 126 patients. Topical tacrolimus 0.1% ointment resulted in significant improvement in genital lesions in 38 patients across openlabel studies (grade ) and 2 case reports (grade ). Tacrolimus was fairly well tolerated by most patients, with side effects of mild pruritus and/ or burning sensation of limited duration. However, use precipitated extreme irritation in one case requiring discontinuation of therapy [1]. Other agents used for successful treatment of genital include topical cyclosporine, which was well tolerated in three patients [1]. Vitamin D preparations were used successfully in 0 patients, often in combination with topical corticosteroids for genital [16 18]. Additional reports of vitamin D preparation use in genital patients exist in the literature; these studies often do not report outcomes specifically for the genital area. A randomized, double-blind, head-to-head comparison (grade 1) of calcitriol ointment 3 lg/g taken twice daily was compared with tacrolimus ointment 0.3 mg/g taken twice daily for 6 weeks in 9 patients with either facial or genitofemoral [19]. Of these patients, five had genital ; two were treated with calcitriol, and three were treated with tacrolimus. Both treatments were well tolerated in the study, and tacrolimus treatment resulted in a more significant reduction of target lesion size, but results were not stratified for lesions on the face and genitalia. In addition to topical treatments, several injectable and oral medications have been reported to successfully clear genital lesions of (Table 3). Ixekizumab, an IL-17A inhibitor approved for the treatment of moderateto-severe and active psoriatic arthritis [20], is the first biologic to report formal clinical
4 12 Dermatol Ther (Heidelb) (2018) 8:09 2 Table 1 Studies on treatments for genital in adults Sources Study characteristics Treatments and outcomes Study design, level of evidence Type of Site Age (years) Cohort (M/F) Successful (side effects) Unsuccessful (side effects) Notes J Fam Pract 2016 [1] Albert et al. [1] Albert et al. [1] Bissonnette et al. [33] Cassano et al. [26] Fischer et al. [16] Foureur et al. [39] Foureur et al. [39] Foureur et al. [39] Foureur et al. [39] Guglielmetti et al. [23] Open-label case series, Open-label case series, Open-label case series, Napkin Napkin Napkin Napkin Groin 1 M Triamcinolone cream, clobetasol ointment Anogenital area, genitocrural folds Groin, genitalia extending to perianal and natal cleft Penis, scrotum F Oral doxepin, methotrexate mg/week (GI disturbance) 1% hydrocortisone cream, topical antifungals, oral antifungals Mild to ultra-potent topical corticosteroids, topical tacalcitol, topical doxepin, twice weekly PUVA for 2 months, topical 0.1% tacrolimus (extreme irritation, worsening of pruritus) 90% improvement after 1 month, 9% improvement after 2 months Oral doxepin partially helpful in alleviating itching, methotrexate improved genitocrural folds but abandoned because of GI disturbance 77 1 F Clomitrazole 1%, hydrocortisone 1% Minimal improvement (29 70) 12 M Topical 0.1% tacrolimus twice daily (mild pruritus or burning sensation of limited duration) Mean genital PASI decreased from 1.8 to 1.2 after 8 weeks Genitalia 18? 9 c Efalizumab 1 mg/kg weekly Considerable improvement in genital lesions after 12 weeks Vulva 33 (1 6) 8 F TCS, 2% LPC, topical calcipotriol Resolution of vulvitis Diaper area 77 1 F Betamethasone 0.0% cream Bifonazole cream daily Cured in 1 month Diaper area 87 1 M Bifonazole cream daily Improved in 1 month Diaper area F Betamethasone 0.0% cream Cured in 1 month Diaper area 87 1 F Bifonazole cream daily, oral fluconazole 100 mg daily Vulva, groin, perianal region 2 1 F Dapsone 100 g daily, mycophenolate mofetil 00 mg twice daily Improved with fluconazole after 1 month 20 mg methotrexate weekly (UTI) Complete clearance after weeks of dapsone, remission for 2 years using topical tacrolimus 0.1% and calcipotriol. Slow improvement with mycophenolate mofetil after 2 months, partial therapeutic response with methotrexate, stopped at months
5 Dermatol Ther (Heidelb) (2018) 8: Table 1 continued Sources Study characteristics Treatments and outcomes Study design, level of evidence Type of Site Age (years) Cohort (M/F) Successful (side effects) Unsuccessful (side effects) Notes Jemec et al. [1] Ješe et al. [2] Kapila et al. [18] Martin- Ezquerra et al. [6] Meeuwis et al. [17] Quan et al. [3] Rallis et al. [7] Open-label case series, Open-label case series, Open-label case series, Open-label case series, Pustular Open-label case series, Glans penis, inner fold of prepuce Glans, foreskin, gluteal fold 3 3 M Topical cyclosporine solution 100 mg/ml three times daily 37 1 M Adalimumab 0 mg every 2 weeks Vulva 30 (2 8) 1 F Methylprednisolone aceponate 0.1% (110), 2% LPC (118), hydrocortisone 1% (9), calcipotriol 0.0% (10), betamethasone dipropionate 0.0% (7), betamethasone dipropionate 0.0%? calcioptriol 0.0% (), UVB phototherapy (3), clioquinol 1% (1), clobetasol propionate 0.0% (1), methotrexate (1) Intertriginous folds Genitalia 0 Glans penis, penile shaft Glans penis, scrotum 18? 8M/7 F (20 80) 2 M/ 17 F 0.1% tacrolimus ointment twice daily Low-potency corticosteroid cream with (18) and without (16) vitamin D analog ointment, moderate-potency corticosteroid cream (), daily tacrolimus with lowpotency corticosteroid cream (2), alternating mild and higher potency corticosteroid cream (1) 23 1 M Oral itraconazole 200 mg bid, triamcinolone, desonide, mometasone furoate, baking soda preparation 37 (22 72) 7 M 0.1% tacrolimus ointment twice daily for 10 days, then every 7 days thereafter Mean baseline score a decreased from.8 to 0.8 after 8 weeks, cyclosporine well tolerated Topical pimecrolimus, topical corticosteroids, methotrexate 1 mg weekly (nausea, vomiting, headache, insomnia) Methylprednisolone aceponate 0.1% (7), 2% LPC (), hydrocortisone 1% (6) Near complete regression at 90 days, complete clearance at 6 months 93.8%responded to topical treatment Improvement as early as day 1, mean total score b from 6.88 to 0.37 after 60 days Significant improvement in PASI, IA, SUM, DLQI, FSDS, sqol-m Hydrocortisone 1%, coal tar 2%, naftifine hydrochloride Some improvement with itraconazole, minimal improvement with hydrocortisone and coal tar Complete clearance after 3 weeks, 3% still clear at 12 weeks
6 1 Dermatol Ther (Heidelb) (2018) 8:09 2 Table 1 continued Sources Study characteristics Treatments and outcomes Study design, level of evidence Type of Site Age (years) Cohort (M/F) Successful (side effects) Unsuccessful (side effects) Notes Ryan et al. [0] Sezer et al. [1] Shimamoto et al. [2] Singh et al. [22] Winrauch et al. Yao et al. [0] Zampetti et al. [8] Randomized, placebocontrolled phase III clinical trial, 1 Pustular Pustular Genitalia 3 Glans penis, penile shaft (19 77) 6 M/ 19 F Ixekizumab 160 mg at week 0, then 80 mg every 2 weeks thereafter 26 1 M Betamethasone valerate, topical % salicylic acid Groin 80 1 M Oral chlorpromazine 12 mg/day Glans penis 37 1 M Dapsone 100 mg daily, doxycycline, metronidazole, penicillin, topical steroid ointments Labium majus 22 1 F Betametasone 17-valerate three times daily Glans penis 37 1 M Tacrolimus 0.1% ointment twice daily Glans penis 1 M Topical tacrolimus 0.1% ointment daily Significant improvement in genital symptoms compared with placebo as measured by spga-g 0/1 (7% vs. 8%), GenPS-SFQ item 2 score 0/1 (78% vs. 21%), and C 3 point reduction in genital itch NRS (60% vs. 8%) Marked regression of the lesions Corticosteroids, antibiotics, griseofulvin Resolution of skin symptoms several weeks after initiation of treatment, associated with reduction in manic symptoms Complete clearance in weeks with dapsone, maintenance with dapsone 0 mg daily Clearance of lesions Topical ketoconazole cream for 2 weeks Resolution of lesions after 3 weeks Complete resolution after 3 weeks PASI Psoriasis Area and Severity Index, IA investigator s assessment of affected genital skin, SUM sum of severity score for erythema, desquamation, and induration, DLQI Dermatological Life Quality Index, FSDS Female Sexual Distress Scale, SQoL-M Sexual Quality of Life questionnaire for use in men, spga-g static Physician s Global Assessment of Genitalia, GenPS-SFQ Genital Psoriasis Sexual Frequency Questionnaire, NRS numeric rating scale Measured on a 6-point scale grading redness, scaling, and maceration Measured on a 9-point scale grading erythema, infiltration, and desquamation of face, genitalia, and intertriginous areas Unspecified sex a b c
7 Dermatol Ther (Heidelb) (2018) 8: Table 2 Evidence on topical treatments for genital by medication Medication Successful Unsuccessful Zinc paste Fergusson et al. [3] Andersen et al. [28], Baggio et al. [36], Greco et al. [37] Salicylic acid Andersen et al. [28] Aniline dye Andersen et al. [28] Topical antibiotics Cretu et al. [37] Topical antifungals Watanabe et al. [38], Foureur et al. [39] Baggio et al. [36], Greco et al. [37], Foureur et al. [39], Yao et al. [0], J Fam Pract 2016 [1] Topical corticosteroids Low potency Cretu et al. [37], Kapila et al. [18], Meeuwis et al. [17], Kamer et al. [2] Andersen et al. [28], Baggio et al. [36], Cretu et al. [37], Fergusson et al. [3], Greco et al. [37], Albert et al. [1], Meeuwis et al. [17], Quan et al. [3], Singh et al. [22], J Fam Pract 2016 [1] Mid potency Afsar et al. [6], Baggio et al. [36], Kapila et al. [18], Meeuwis et al. [17], Weinrauch et al. [], J Fam Pract 2016 [1] Amichai et al. [29], Hernandez et al. [], Albert et al. [1], Meeuwis et al. [17], Ješe et al. [2] High potency Hernandez et al. [], Foureur et al. [39], J Fam Pract 2016 [1] Albert et al. [1], Meeuwis et al. [17] Topical calcineurin inhibitors Amichai et al. [29], Bissonnette et al. [33], Martin-Ezquerra et al. [6], Rallis et al. [7], Zampetti et al. [8], Jemec et al. [1], Yao et al. [0] Albert et al. [1], Ješe et al. [2] Vitamin D analogs Amichai et al. [29], Albert et al. [1] Coal tar Andersen et al. [28] Quan et al. [3] Topical doxepin Albert et al. [1] Combination therapies Topical antifungal and tar Kapila et al. [18] Low-potency TCS? topical antifungals Andersen et al. [28], Rattet et al. [9], Kapila et al. [18] Albert et al. [1] Low-potency TCS? TCIs Kapila et al. [18] Low-potency TCS? tar Kapila et al. [18] Kapila et al. [18]
8 16 Dermatol Ther (Heidelb) (2018) 8:09 2 Table 2 continued Medication Successful Unsuccessful Low-potency TCS? topical vitamin D analog Meeuwis et al. [17] Low-potency TCS? tar? vitamin D analog Fischer et al. [16] Low-potency TCS? midpotency TCS? tar Kapila et al. [18] Kapila et al. [18] Low-potency TCS? midpotency TCS? tar? vitamin D analog Kapila et al. [18] Mid-potency TCS? tar Kapila et al. [18] Kapila et al. [18] Mid-potency TCS? tar? vitamin D analog Kapila et al. [18] High-potency TCS? vitamin D? tar Kapila et al. [18]
9 Dermatol Ther (Heidelb) (2018) 8: Table 3 Evidence on systemic treatments for genital by medication Medication Successful Unsuccessful Phototherapy UVB phototherapy Kapila et al. [18] PUVA Albert et al. [1] Immunosuppressants Mycophenolate mofetil Guglielmetti et al. [23] Methotrexate Albert et al. [1], Kapila et al. [18] Guglielmetti et al. [23], Ješe et al. [2] Biologics Adalimumab Ješe et al. [2] Ixekizumab Ryan et al. [0] Efalizumab a Cassano et al. [26] Oral antifungals Foureur et al. [39], Meeuwis et al. [17] J Fam Pract 2016 [1] Oral antibiotics Quan et al. [3] Singh et al. [22] Dapsone Guglielmetti et al. [23], Singh et al. [22] Doxepin Albert et al. [1] Antihistamines Cretu et al. [37] Antipsychotics Shimamoto et al. [2] Calcium gluconate Cretu et al. [37] a Formerly available treatment trial data for the treatment of genital [21]. In this randomized, double-blind, placebocontrolled phase III trial (grade 1), adult subjects with moderate-to-severe genital were randomized to receive either placebo (n = 7) or ixekizumab 160 mg subcutaneously at week 0 followed by 80 mg every 2 weeks thereafter (n = 7). The primary outcome of patients achieving static Physician s Global Assessment of Genitalia score of clear or minimal (spga-g 0/1) by week 12 was met by significantly more subjects on ixekizumab treatment than placebo (7% vs. 8%). Additionally, significantly more patients treated with ixekizumab reported improved sexual activity as measured by Genital Psoriasis Sexual Frequency Questionnaire (GenPS-SFQ) item 2 score 0/1 (78% vs. 21%) and clinically meaningful reduction in genital itch as measured by the genital itch numeric rating scale (NRS) (60% vs. 8%) compared with placebo. Safety outcomes were similar to the overall safety profile of ixekizumab, with the most common adverse events including diarrhea, injection site reactions, nasopharyngitis, upper respiratory tract infections, and headaches. Two cases (grade ) reported that oral dapsone given 100 mg daily was found to be successful in clearing lesions within weeks, without any reported adverse events [22, 23]. Mycophenolate mofetil and oral doxepin have both been reported as successful treatments for genital in one case each as well [1, 23]. Methotrexate mg weekly improved genital symptoms in two of four genital patients [1, 18, 23, 2]. However, methotrexate use was associated with gastrointestinal disturbance, headache, insomnia, and urinary tract infections. There is one report of genital pustular clearance
10 18 Dermatol Ther (Heidelb) (2018) 8:09 2 after initiation of chlorpromazine for treatment of a concurrent manic episode [2]. Adalimumab has also been shown in one case report to result in clearance of genital by 6 months without adverse effects [2]. Efalizumab considerably improved genital pruritus in 9 patients (grade ) given at a dose of 1 mg/kg weekly for 12 weeks [26]. However, 2 of patients in this study experienced adverse events requiring treatment discontinuation within 12 weeks ( flare and neurologic symptoms), and 7 patients described self-limited treatment-associated papular. Efalizumab has since been removed from the market because of safety concerns [27]. Treatment of Genital Psoriasis in the Pediatric Population A total of 12 articles examined treatments for genital in infants and children (Table ). Three of these articles were open-label studies (grade ), while the remaining ten were case reports (grade ) describing the effects of treatment on pediatric patients. Treatment outcomes were described for a total of 126 patients. Mild, topical coal tar preparations were effective in clearing genital lesions among 91 pediatric patients from two case series [16, 28]. Topical corticosteroid-based regimens led to successful treatment outcomes in 37 cases. Lowpotency topical steroids were used in 26 patients; moderate- and high-potency steroids were used in 6 patients and 1 patient, respectively. Successful treatment in six patients also included topical antifungal medications, primarily ketoconazole cream and clotrimazole cream. There was one case report (grade ) of complete resolution of psoriatic lesions with topical pimecrolimus 1% ointment treatment [29]. All of the therapies used in children were well tolerated, without any significant adverse events reported. DISCUSSION In the past several years, there has been a moderate increase in studies assessing treatments for genital. At the time of the last published review on this topic in 2011, only 6 case reports and 1 open-label study described the effects of therapies for genital, while 2 articles reflected expert onion on treatment for this disease [3]. In our analysis, we found 1 randomized controlled trial (grade 1), 11 open-label studies (grade ), and 26 case reports (grade ) describing the efficacy and safety of topical and systemic treatments for genital. Various therapies have been shown to be effective for genital in case reports and case series, but high-quality evidence in the form of randomized controlled trials remains inadequate for genital treatments. Low-to-mid-potency topical corticosteroids are recommended as the first-line treatment for genital [30] (grade of recommendation: D) and are commonly reported in the literature to be a critical component of treatment for these lesions. However, topical corticosteroids are generally approached with great caution for genital patients because of the unique environment of the genitalia [31]. The thin skin and constant occlusion of this environment cause topical medications to have increased penetration in the groin area, which is a particular problem for infants, who have a high surface area-to-body mass ratio, predisposing them to systemic side effects. Mild topical corticosteroids may not be potent enough to induce a clinically significant response in some patients [11, 32] and are often used in combination with second-line topical therapies to yield clinical benefit (Table 2). Moderate-to-high-potency corticosteroids have been used effectively in adults and children with genital, both as monotherapy and in combination with other topical agents, without reports of significant adverse effects (Table 2). There was a lack of reporting on adverse effects from topical corticosteroids in studies included our analysis; therefore, there is not enough evidence to determine whether there were no side effects with these therapies or if they simply were not mentioned. From the existing evidence, topical corticosteroids continue to be recommended as first-line treatment
11 Dermatol Ther (Heidelb) (2018) 8: Table Studies on treatments for genital in children and infants Sources Study characteristics Treatments and outcomes Study design, level of evidence Type of Site Age Cohort (M/F) Successful Unsuccessful Result Afsar et al. [6] Napkin Diaper area. months 1 M 0.0% clobetasone 17-butyrate cream daily, emollient cream Overall regression with intermittent mild flares Amichai et al. [29] Glans penis, distal parts of shaft 10 years 1 M 1% pimecrolimus cream Betamethasone 1% cream, calcipotriol ointment Resolution of psoriatic lesions after 3 weeks of topical pimecrolimus, recurrence treated successfully Andersen et al. [28] Openlabel case series, Psoriasiform napkin dermatitis Diaper area 2.1 months (0 21) 3 M/ 32 F Tar (6), corticosteroidvioform ointment (3) Zinc paste, salicylic ointment, steroid ointment, or aniline dye solutions Tar effective in 6 patients, corticosteroid-vioform ointment effective in 3 patients Baggio et al. [36] Psoriasiform napkin dermatitis Diaper area 18 months 1 F Fluticasone propionate cream Zinc oxide-based ointments, antifungal creams, desonide cream Complete resolution at 1 week Creţu et al. [37] Napkin Anogenital area, buttocks, upper 1/3 thighs months 1 M Systemic antihistamines, nonspecific desensitization treatment, rigorous hygiene, non-fluorinated topical corticosteroids Antibiotic ointment for days, calcium gluconate, topical corticosteroids Significant improvement by day 2 of treatment
12 20 Dermatol Ther (Heidelb) (2018) 8:09 2 Table continued Sources Study characteristics Treatments and outcomes Study design, level of evidence Type of Site Age Cohort (M/F) Successful Unsuccessful Result Fergusson et al. [3] Openlabel case series, Psoriasiform napkin dermatitis Diaper area Infants 22 a Zinc-oxide paste Topical steroids Complete clearance Fischer et al. [16] Openlabel case series, Vulva 6 years (2 12) 27 F LPC and topical calcipotriol with 1% hydrocortisone (23) or methylprednisolone aceponate 0.1% ointment () Symptom remission, LPC well tolerated Greco et al. [37] Napkin Diaper area 18 months 1 F Zinc oxide paste, nystatin, hydrocortisone acetate 2.% ointment Minimal improvement Hernandez et al. [] Vulvar Vulva, perianal area years 1 F Clobetasol 0.0% ointment twice daily Triamcinolone 0.1% ointment twice daily Significantly improved erythema on inner labial and perianal areas at 2 weeks, complete resolution at weeks Kamer et al. [2] Napkin Diaper area 10 weeks 1 F Low-potency topical corticosteroids Clearance of lesions at 2 weeks
13 Dermatol Ther (Heidelb) (2018) 8: Table continued Sources Study characteristics Treatments and outcomes Study design, level of evidence Type of Site Age Cohort (M/F) Successful Unsuccessful Result Rattet et al. [9] Psoriasiform napkin dermatitis Diaper area 12 months 1 F Clomitrazole 1% cream, hydrocortisone 1% cream three times daily Clearance of lesions at 2 weeks with mild erythema Rattet et al. [9] Psoriasiform napkin dermatitis Diaper area weeks 1 M Clomitrazole 1% cream, hydrocortisone 1% cream three times daily Clearance of lesions at weeks Watanabe et al. [38] Pustular Diaper area months 1 M 0.2% ketoconazole cream daily Complete clearance at 1 month LPC liquor picis carbonis Unspecified sex a
14 22 Dermatol Ther (Heidelb) (2018) 8:09 2 for genital (grade of recommendation: C). The data in this analysis do not show superior efficacy for nonsteroidal topical treatments compared with topical corticosteroids for the treatment of genital (Table 2). Topical calcineurin inhibitors did improve genital in several patients and were fairly well tolerated. Mild burning or pruritus can be associated with using these treatments in the sensitive groin region, but these symptoms are often manageable and limited in duration [33]. Topical coal tar preparations demonstrate efficacy in both adults and children with genital lesions and are not associated with significant adverse effects. Vitamin D analogs are sometimes recommended for patients with general (grade of recommendation: D), but this is less reported in the literature [11, 30]. However, these nonsteroidal topical therapies may increase the risk for lymphoma, be more irritating, and be more costly than topical corticosteroids [3]. Topical calcineurin inhibitors, coal tar preparations, and vitamin D analogs may thus be used as second-line therapies for lesions in the genital area (grade of recommendation: C). Systemic therapies such as dapsone and methotrexate can work well for patients with genital lesions and should be considered for patients with debilitating quality of life impairment (grade of recommendation: C) [30]. Our analysis did not find any evidence on the use of other traditional systemic agents, such as oral cyclosporine, acitretin, or apremilast, specifically for genital. Although numerous effective biologics are available for the treatment of, there is only one published clinical trial result on the efficacy of currently approved biologics specifically for genital. A recent study has shown ixekizumab to have high efficacy specifically for genital, with rapid improvement seen as early as 1 week into treatment [21]. This medication significantly improves genital lesion appearance, genital itch, sexual health, and quality of life and may be a promising solution for patients suffering from recalcitrant genital. Ixekizumab is currently the only medication with FDA labeling specifically mentioning genital (grade of recommendation: B). Although not specifically addressed in the majority of articles in our analysis, good hygiene and reduction of friction are essential first-line measures for the treatment of genital patients (grade of recommendation: D) [11]. Gentle, non-soap cleansers are recommended to keep the genitals clean without irritating the area, and patients should be advised to wear loose-fitting, unrestrictive clothing to avoid koebnerization and further irritation [30]. Patients with genital should always use lubricant or lubricated condoms with any sexual activity to avoid exacerbation of genital symptoms. In combination with proper pharmacologic therapy, these supportive measures are necessary for minimizing the impact of genital. Despite increased research into genital in recent years, the optimal treatment approach for affected patients remains unclear. Overall, available evidence is limited, especially regarding the efficacy of systemic agents for genital. This is most likely because systemic treatments are indicated for moderateto-severe, and efficacy evaluations typically do not specifically assess genital symptoms. Genital is ill defined, varies in characterization throughout the literature, and is not distinguished from inverse in several studies. The existing literature thus uses various different measures to evaluate treatment efficacy or assess symptom improvement qualitatively so studies can be difficult to compare. The introduction of novel assessment tools for genital will facilitate a greater understanding of how various treatments compare. Additionally, most studies included in our analysis did not assess or report safety data or side effects with treatment, which is especially important for the sensitive genital area. Most recommendations for treating genital are based on case reports or expert opinion only, and randomized controlled trials for this disease are lacking [3, 11]. Larger sample sizes and controlled studies are needed to properly assess the safety and efficacy of treatments for
15 Dermatol Ther (Heidelb) (2018) 8: genital to better understand the optimal management approach for these patients. CONCLUSIONS Recently, a growing number of studies have evaluated the efficacy of various treatments specifically for genital, including one randomized clinical trial for a biologic agent [3]. A variety of topical therapies have shown varying success for treatment of genital, while far fewer systemic and biologic therapies have been evaluated for genital. Further inquiry into the optimal treatment regimen for genital is necessary. ACKNOWLEDGEMENTS Funding. No funding or sponsorship was received for this study or publication of this article. Wilson Liao is funded in part by grants from the National Institutes of Health (R01AR0617, U01AI11912). Authorship. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published. Disclosures. Kristen M Beck, Eric J Yang, Isabelle M Sanchez, and Wilson Liao have nothing to disclose. Compliance with Ethics Guidelines. The article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors. Open Access. This article is distributed under the terms of the Creative Commons Attribution-NonCommercial.0 International License ( by-nc/.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. REFERENCES 1. Meeuwis KAP, Potts Bleakman A, van de Kerkhof PCM, Dutronc Y, Henneges C, Kornberg LJ, Menter A. Prevalence of genital in patients with. J Dermatol Treat. 2018: Wang G, Li C, Gao T, Liu Y. Clinical analysis of 8 cases of inverse : a hospital-based study. Eur J Dermatol. 200;1: Meeuwis KA, de Hullu JA, Massuger LF, van de Kerkhof PC, van Rossum MM. Genital : a systematic literature review on this hidden skin disease. Acta Derm Venereol. 2011;91: 11.. Ryan C, Sadlier M, De Vol E, Patel M, Lloyd AA, Day A, Lally A, Kirby B, Menter A. Genital is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 201;72: Morris A, Rogers M, Fischer G, Williams K. Childhood : a clinical review of 1262 cases. Pediatr Dermatol. 2001;18: Afsar FS, Uysal SS, Salis FM, Calli AO. Napkin. Pediatr Int. 2016;8: Meeuwis KA, de Hullu JA, van de Nieuwenhof HP, Evers AW, Massuger LF, van de Kerkhof PC, van Rossum MM. Quality of life and sexual health in patients with genital. Br J Dermatol. 2011;16: Zamirska A, Reich A, Berny-Moreno J, Salomon J, Szepietowski JC. Vulvar pruritus and burning sensation in women with. Acta Derm Venereol. 2008;88: Cather JC, Ryan C, Meeuwis K, Potts Bleakman AJ, Naegeli AN, Edson-Heredia E, Poon JL, Jones C, Wallace AN, Guenther L, Fretzin S. Patients perspectives on the impact of genital : a qualitative study. Dermatol Ther (Heidelb). 2017;7: Meeuwis KA, van de Kerkhof PC, Massuger LF, de Hullu JA, van Rossum MM. Patients experience of in the genital area. Dermatology. 2012;22:271 6.
16 2 Dermatol Ther (Heidelb) (2018) 8: Menter A, Korman NJ, Elmets CA, Feldman SR, Gelfand JM, Gordon KB, Gottlieb A, Koo JY, Lebwohl M, Leonardi CL, Lim HW, Van Voorhees AS, Beutner KR, Ryan C, Bhushan R. Guidelines of care for the management of and psoriatic arthritis: section 6. Guidelines of care for the treatment of and psoriatic arthritis: case-based presentations and evidence-based conclusions. J Am Acad Dermatol. 2011;6: Farage M, Maibach HI. The vulvar epithelium differs from the skin: implications for cutaneous testing to address topical vulvar exposures. Contact Dermatitis. 200;1: Howick J, Chalmers I, Glasziou P, Greenhalgh T, Heneghan C, Liberati A, Moschetti I, Phillips B, Thornton H. The 2011 Oxford CEBM levels of evidence (introductory document). Oxford Centre for Evidence-Based Medicine Albert S, Neill S, Derrick EK, Calonje E. Psoriasis associated with vulval scarring. Clin Exp Dermatol. 200;29: Jemec GB, Baadsgaard O. Effect of cyclosporine on genital and lichen planus. J Am Acad Dermatol. 1993;29: Fischer G. Chronic vulvitis in pre-pubertal girls. Aust J Dermatol. 2010;1: Meeuwis KA, de Hullu JA, IntHout J, Hendriks IM, Sparreboom EE, Massuger LF, van de Kerkhof PC, van Rossum MM. Genital awareness program: physical and psychological care for patients with genital. Acta Derm Venereol. 201;9: Kapila S, Bradford J, Fischer G. Vulvar in adults and children: a clinical audit of 19 cases and review of the literature. J Lower Genit Tract Dis. 2012;16: Liao YH, Chiu HC, Tseng YS, Tsai TF. Comparison of cutaneous tolerance and efficacy of calcitriol 3 microg g(-1) ointment and tacrolimus 0.3 mg g(-1) ointment in chronic plaque involving facial or genitofemoral areas: a double-blind, randomized controlled trial. Br J Dermatol. 2007;17: TALTZ (ixekizumab) [package insert]. Indianapolis: Eli Lilly and Company Ryan C, Menter A, Guenther L, Blauvelt A, Bissonnette R, Yang F, Potts Bleakman A. Efficacy and safety of ixekizumab in a randomized, doubleblinded, placebo-controlled, phase 3b clinical trial in patients with moderate-to-severe genital. J Sexual Med. 2018;1:S Singh N, Thappa DM. Circinate pustular localized to glans penis mimicking circinate balanitis and responsive to dapsone. Indian J Dermatol Venereol Leprol. 2008;7: Guglielmetti A, Conlledo R, Bedoya J, Ianiszewski F, Correa J. Inverse involving genital skin folds: successful therapy with dapsone. Dermatol Ther (Heidelb). 2012;2:1. 2. Jese R, Perdan-Pirkmajer K, Dolenc-Voljc M, Tomsic M. A case of inverse successfully treated with adalimumab. Acta Dermatovenerol Alp Pannonica Adriat. 201;23: Shimamoto Y, Shimamoto H. Annular pustular associated with affective psychosis. Cutis. 1990;: Cassano N, Mastrandrea V, Buquicchio R, Miracapillo A, Loconsole F, Filotico R, Vena GA. Efalizumab in moderate-to-severe plaque : a retrospective case series analysis from clinical practice. J Biol Regul Homeost Agents. 2008;22: Talamonti M, Teoli M, Botti E, Spallone G, Chimenti S, Costanzo A. Patients with moderate to severe plaque : one year after the European Medicines Agency recommendation of efalizumab suspension. Dermatology. 2011;222: Andersen SL, Thomsen K. Psoriasiform napkin dermatitis. Br J Dermatol. 1971;8: Amichai B. Psoriasis of the glans penis in a child successfully treated with Elidel (pimecrolimus) cream. J Eur Acad Dermatol Venereol. 200;18: Menter A, Korman NJ, Elmets CA, Feldman SR, Gelfand JM, Gordon KB, Gottlieb AB, Koo JY, Lebwohl M, Lim HW, Van Voorhees AS, Beutner KR, Bhushan R. Guidelines of care for the management of and psoriatic arthritis: section. Guidelines of care for the management and treatment of with traditional systemic agents. J Am Acad Dermatol. 2009;61: Kivelevitch D, Frieder J, Watson I, Paek SY, Menter MA. Pharmacotherapeutic approaches for treating in difficult-to-treat areas. Expert Opin Pharmacother. 2018;19: Czuczwar P, Stepniak A, Goren A, Wrona W, Paszkowski T, Pawlaczyk M, Piekarska-Myslinska D, Wozniak S, Pietrzak A. Genital : a hidden multidisciplinary problem a review of literature. Ginekol Pol. 2016;87: Bissonnette R, Nigen S, Bolduc C. Efficacy and tolerability of topical tacrolimus ointment for the
17 Dermatol Ther (Heidelb) (2018) 8: treatment of male genital. J Cutan Med Surg. 2008;12: Broeders JA, Ahmed Ali U, Fischer G. Systematic review and meta-analysis of randomized clinical trials (RCTs) comparing topical calcineurin inhibitors with topical corticosteroids for atopic dermatitis: a 1-year experience. J Am Acad Dermatol. 2016;7:10 19 (e13). 3. Fergusson AG, Fraser NG, Grant PW. Napkin dermatitis with psoriasiform ide. A review of fiftytwo cases. Br J Dermatol. 1966;78: Baggio R, Le Treut C, Darrieux L, Vareliette A, Safa G. Psoriasiform diaper rash possibly induced by oral propranolol in an 18-month-old girl with infantile hemangioma. Rep Dermatol. 2016;8: Greco M, Chamlin SL. An 18-month-old girl with chronic diaper dermatitis. Psoriasis presenting in the diaper area. Pediatr Ann. 2006;3(79): Watanabe M, Tabata N, Tagami H. Explosive diaper pustular. Pediatr Dermatol. United States. 2002;19: Foureur N, Vanzo B, Meaume S, Senet P. Prospective aetiological study of diaper dermatitis in the elderly. Br J Dermatol. 2006;1: Yao XJ, Zhang TD. Psoriasis localized to the glans penis in a 37-year-old man. CMAJ. 2018;190:E Usatine RP. Itchy rash in groin. J Fam Pract. 2016;6(11). 2. Kamer B, Pyziak K, Krawczyk T, Socha-Banasiak A, Rotsztejn H. A rare case of resembling diaper dermatitis. Przeglad Pediatryczny. 2012;2: Quan MB, Ruben BS. Pustular limited to the penis. Int J Dermatol. 1996;3:202.. Weinrauch L, Katz M. Psoriasis vulgaris of labium majus. Cutis. 1986;38:333.. Hernandez M, Simms-Cendan J, Zendell K. Guttate following streptococcal vulvovaginitis in a five-year-old girl. J Pediatr Adolesc Gynecol. 201;28:e Martin Ezquerra G, Sanchez Regana M, Herrera Acosta E, Umbert Millet P. Topical tacrolimus for the treatment of on the face, genitalia, intertriginous areas and corporal plaques. J Drugs Dermatol. 2006;: Rallis E, Nasiopoulou A, Kouskoukis C, Roussaki- Schulze A, Koumantaki E, Karpouzis A, Arvanitis A. Successful treatment of genital and facial with tacrolimus ointment 0.1%. Drugs Exp Clin Res. 200;31: Zampetti A, Gnarra M, Linder D, Digiuseppe MD, Carrino N, Feliciani C. Psoriatic pseudobalanitis circinata as a post-viral koebner phenomenon. Rep Dermatol. 2010;2: Rattet JP, Headley JL, Barr RJ. Diaper dermatitis with psoriasiform ID eruption. Int J Dermatol. 1981;20: Ryan C, Menter A, Guenther L, Blauvelt A, Bissonnette R, Meeuwis K, Sullivan J, Cather JC, Yosipovitch G, Gottlieb AB, Merola JF, Callis Duffin K, Fretzin S, Osuntokun OO, Burge R, Naegeli AN, Yang FE, Lin CY, Todd K, Potts Bleakman A. Efficacy and safety of ixekizumab in a randomized, doubleblinded, placebo-controlled phase 3b study of patients with moderate-to-severe genital. Br J Dermatol Sezer E, Lehman JS, Yalçın Ö, Tüfek İ, Keskin S, Durmaz EÖ, Sahin S. Oyster-shaped hyperkeratotic plaques on the penis. Dermatol Pract Concept. 201;():
Psoriasis Penis - A Two Case Report
 Article ID: WMC003176 ISSN 2046-1690 Psoriasis Penis - A Two Case Report Corresponding Author: Dr. Ilko Bakardzhiev, MD, PhD, Medical College, Medical University of Varna, 9000 - Bulgaria Submitting Author:
Article ID: WMC003176 ISSN 2046-1690 Psoriasis Penis - A Two Case Report Corresponding Author: Dr. Ilko Bakardzhiev, MD, PhD, Medical College, Medical University of Varna, 9000 - Bulgaria Submitting Author:
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar
 Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar Dr. Steven Feldman 1 Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands
Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar Dr. Steven Feldman 1 Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands
15 minute eczema consultation
 THERAPY WORKSHOP 15 minute eczema consultation History Current treatments Examination Treatment Plan Written action plan Soap substitute/bath oil Antiseptic baths Emollients Topical steroids Other treatments
THERAPY WORKSHOP 15 minute eczema consultation History Current treatments Examination Treatment Plan Written action plan Soap substitute/bath oil Antiseptic baths Emollients Topical steroids Other treatments
Vulval dermatoses. Dr Fiona Lewis, Consultant Dermatologist St John s Institute of Dermatology, London & Heatherwood & Wexham Park Hospital, Slough
 Vulval dermatoses Dr Fiona Lewis, Consultant Dermatologist St John s Institute of Dermatology, London & Heatherwood & Wexham Park Hospital, Slough Pigmentation Vulvodynia Ulcers Genetic Pruritus VULVAL
Vulval dermatoses Dr Fiona Lewis, Consultant Dermatologist St John s Institute of Dermatology, London & Heatherwood & Wexham Park Hospital, Slough Pigmentation Vulvodynia Ulcers Genetic Pruritus VULVAL
The Development of the Genital Psoriasis Sexual Frequency Questionnaire (GenPs-SFQ) to Assess the Impact of Genital Psoriasis on Sexual Health
 Dermatol Ther (Heidelb) (2018) 8:33 44 https://doi.org/10.1007/s13555-017-0212-3 ORIGINAL RESEARCH The Development of the Genital Psoriasis Sexual Frequency Questionnaire (GenPs-SFQ) to Assess the Impact
Dermatol Ther (Heidelb) (2018) 8:33 44 https://doi.org/10.1007/s13555-017-0212-3 ORIGINAL RESEARCH The Development of the Genital Psoriasis Sexual Frequency Questionnaire (GenPs-SFQ) to Assess the Impact
Topical Immunomodulator Step Therapy Program
 Topical Immunomodulator Step Therapy Program Policy Number: 5.01.557 Last Review: 8/2017 Origination: 7/2013 Next Review: 8/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) BCBSKC will provide
Topical Immunomodulator Step Therapy Program Policy Number: 5.01.557 Last Review: 8/2017 Origination: 7/2013 Next Review: 8/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) BCBSKC will provide
New Medicine Report. Pimecrolimus. RED- Hospital only Date of Last Revision 6 th March 2003
 New Medicine Report Document Status Pimecrolimus Reviewed by Suffolk D&T RED- Hospital only Date of Last Revision 6 th March 2003 Approved Name Pimecrolimus Trade Name Elidel Manufacturer Novartis Legal
New Medicine Report Document Status Pimecrolimus Reviewed by Suffolk D&T RED- Hospital only Date of Last Revision 6 th March 2003 Approved Name Pimecrolimus Trade Name Elidel Manufacturer Novartis Legal
Medication Policy Manual. Topic: Dupixent, dupilumab Date of Origin: March 10, Committee Approval: March 10, 2017 Next Review Date: May 2018
 Independent licensees of the Blue Cross and Blue Shield Association Medication Policy Manual Policy No: dru493 Topic: Dupixent, dupilumab Date of Origin: March 10, 2017 Committee Approval: March 10, 2017
Independent licensees of the Blue Cross and Blue Shield Association Medication Policy Manual Policy No: dru493 Topic: Dupixent, dupilumab Date of Origin: March 10, 2017 Committee Approval: March 10, 2017
Revisiting and old friend - pimecrolimus
 Revisiting and old friend - pimecrolimus K. A. Papp MD, PhD Probity Medical Research KAP is a consultant, investigator, speaker and advisory board member for Astellas GSK Leo Valeant 2 Warning some content
Revisiting and old friend - pimecrolimus K. A. Papp MD, PhD Probity Medical Research KAP is a consultant, investigator, speaker and advisory board member for Astellas GSK Leo Valeant 2 Warning some content
STUDY. 1% Pimecrolimus, 0.005% Calcipotriol, and 0.1% Betamethasone in the Treatment of Intertriginous Psoriasis
 STUDY 1% Pimecrolimus, 0.005% Calcipotriol, and 0.1% Betamethasone in the Treatment of Intertriginous Psoriasis A Double-blind, Randomized Controlled Study Alexander Kreuter, MD; Anna Sommer, MD; Julia
STUDY 1% Pimecrolimus, 0.005% Calcipotriol, and 0.1% Betamethasone in the Treatment of Intertriginous Psoriasis A Double-blind, Randomized Controlled Study Alexander Kreuter, MD; Anna Sommer, MD; Julia
Clinical Policy: Ixekizumab (Taltz) Reference Number: ERX.SPA.122 Effective Date:
 Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/95926
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/95926
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 2116-3 Program Prior Authorization/Medical Necessity Medications Dupixent (dupilumab) P&T Approval Date 1/2017, 5/2017, 7/2017
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 2116-3 Program Prior Authorization/Medical Necessity Medications Dupixent (dupilumab) P&T Approval Date 1/2017, 5/2017, 7/2017
Pharmacologic Treatment of Atopic Dermatitis
 J KMA Pharmacotherapeutics Pharmacologic Treatment of Atopic Dermatitis Chun Wook Park, MD Department of Dermatology, Hallym University College of Medicine E mail : dermap@paran.com J Korean Med Assoc
J KMA Pharmacotherapeutics Pharmacologic Treatment of Atopic Dermatitis Chun Wook Park, MD Department of Dermatology, Hallym University College of Medicine E mail : dermap@paran.com J Korean Med Assoc
Literature Scan: Topical Corticosteroids. Month/Year of Review: March 2015 Date of Last Review: March 2013 Source Document: OSU College of Pharmacy
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
KEY MESSAGES. Psoriasis patients are more prone to cardiovascular diseases, stroke, lymphoma and non-melanoma skin cancers, and increased mortality.
 KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
Clearance in vulvar lichen sclerosus: a realistic treatment endpoint or a chimera?
 DOI: 10.1111/jdv.14516 JEADV SHORT REPORT Clearance in vulvar lichen sclerosus: a realistic treatment endpoint or a chimera? A. Borghi,* A. Virgili, S. Minghetti, G. Toni, M. Corazza Dipartimento di Scienze
DOI: 10.1111/jdv.14516 JEADV SHORT REPORT Clearance in vulvar lichen sclerosus: a realistic treatment endpoint or a chimera? A. Borghi,* A. Virgili, S. Minghetti, G. Toni, M. Corazza Dipartimento di Scienze
An otherwise healthy 12-year-old
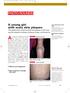 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
Conflicts of interest
 Vulvar Cases 2 nd PANHELLANIC CONGRESS on Lower Genital Tract Disorders December 14-16 Grand Hyatt Athens Lynette J. Margesson MD FRCPC Assistant Professor of Obstetrics & Gynecology and Surgery(Dermatology)
Vulvar Cases 2 nd PANHELLANIC CONGRESS on Lower Genital Tract Disorders December 14-16 Grand Hyatt Athens Lynette J. Margesson MD FRCPC Assistant Professor of Obstetrics & Gynecology and Surgery(Dermatology)
The safety and effectiveness of Dupixent in pediatric patients have not been established (1).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.30 Subject: Dupixent Page: 1 of 6 Last Review Date: September 15, 2017 Dupixent Description Dupixent
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.30 Subject: Dupixent Page: 1 of 6 Last Review Date: September 15, 2017 Dupixent Description Dupixent
Effect of Apremilast on Patient-Reported Outcomes in Patients With Moderate to Severe Plaque Psoriasis in the ESTEEM 1 Trial
 1152 Effect of Apremilast on Patient-Reported Outcomes in Patients With Moderate to Severe Plaque Psoriasis in the ESTEEM 1 Trial April W. Armstrong, MD, MPH 1 ; Christopher E.M. Griffiths, MD 2 ; Tom
1152 Effect of Apremilast on Patient-Reported Outcomes in Patients With Moderate to Severe Plaque Psoriasis in the ESTEEM 1 Trial April W. Armstrong, MD, MPH 1 ; Christopher E.M. Griffiths, MD 2 ; Tom
Skin disorders. Seborrhoeic dermatitis Search date April 2010 Luigi Naldi ...
 Seborrhoeic Search date April 21 Luigi Naldi.................................................. ABSTRACT INTRODUCTION: Seborrhoeic affects at least 1% of the population. Malassezia (Pityrosporum) ovale
Seborrhoeic Search date April 21 Luigi Naldi.................................................. ABSTRACT INTRODUCTION: Seborrhoeic affects at least 1% of the population. Malassezia (Pityrosporum) ovale
Original Policy Date
 MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
Children s Hospital Of Wisconsin
 Children s Hospital Of Wisconsin Co-Management Guidelines To support collaborative care, we have developed guidelines for our community providers to utilize when referring to, and managing patients with,
Children s Hospital Of Wisconsin Co-Management Guidelines To support collaborative care, we have developed guidelines for our community providers to utilize when referring to, and managing patients with,
The role of the practice nurse in managing psoriasis in primary care
 The role of the practice nurse in managing psoriasis in primary care Item type Authors Publisher Journal Article Buckley, David Nursing in General Practice Nursing in general practice Downloaded 16-Sep-2016
The role of the practice nurse in managing psoriasis in primary care Item type Authors Publisher Journal Article Buckley, David Nursing in General Practice Nursing in general practice Downloaded 16-Sep-2016
Ustekinumab Treatment of Erythrodermic Psoriasis Occurring after Physical Stress: A Report of Two Cases
 Published online: September 26, 2013 1662 6567/13/0053 0254$38.00/0 This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial 3.0 Unported license (CC BY-NC)
Published online: September 26, 2013 1662 6567/13/0053 0254$38.00/0 This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial 3.0 Unported license (CC BY-NC)
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 3Q17 July August
 BRAND NAME Dupixent GENERIC NAME dupilumab MANUFACTURER Regeneron DATE OF APPROVAL March 28, 2017 PRODUCT LAUNCH DATE First week of April 2017 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review
BRAND NAME Dupixent GENERIC NAME dupilumab MANUFACTURER Regeneron DATE OF APPROVAL March 28, 2017 PRODUCT LAUNCH DATE First week of April 2017 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review
Recalcitrant Warty Erythroderma With Severe Pruritus. Gil Yosipovitch Professor & Chair Department of Dermatology & Itch Center Temple University
 Recalcitrant Warty Erythroderma With Severe Pruritus Gil Yosipovitch Professor & Chair Department of Dermatology & Itch Center Temple University DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY Gil Yosipovitch,
Recalcitrant Warty Erythroderma With Severe Pruritus Gil Yosipovitch Professor & Chair Department of Dermatology & Itch Center Temple University DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY Gil Yosipovitch,
Comparative Efficacy of Topical Calcipotriol (0.005%) Versus Topical Corticosteroid (Betamethasone 0.1%) in Treating Plaque Type Psoriasis
 Original Article Comparative Efficacy of Topical Calcipotriol (0.005%) Versus Topical Corticosteroid (Betamethasone 0.1%) in Treating Plaque Type Psoriasis Admed GKA 1, Khondker L 2, Nessa M 3, Alam MN
Original Article Comparative Efficacy of Topical Calcipotriol (0.005%) Versus Topical Corticosteroid (Betamethasone 0.1%) in Treating Plaque Type Psoriasis Admed GKA 1, Khondker L 2, Nessa M 3, Alam MN
Evaluating Psoriasis: Patient Reported Outcomes and Impact of Disease
 Evaluating Psoriasis: Patient Reported Outcomes and Impact of Disease Bruce E. Strober, MD, PhD Professor and Chair Department of Dermatology University of Connecticut Farmington, Connecticut DISCLOSURE
Evaluating Psoriasis: Patient Reported Outcomes and Impact of Disease Bruce E. Strober, MD, PhD Professor and Chair Department of Dermatology University of Connecticut Farmington, Connecticut DISCLOSURE
Month/Year of Review: January 2015 Date of Last Review: January 2010
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
This PDF is available for free download from a site hosted by Medknow Publications
 Net Study Comparison of clinical efficacy of topical tazarotene.1% cream with topical clobetasol propionate.5% cream in chronic plaque psoriasis: A double-blind, randomized, right-left comparison study
Net Study Comparison of clinical efficacy of topical tazarotene.1% cream with topical clobetasol propionate.5% cream in chronic plaque psoriasis: A double-blind, randomized, right-left comparison study
Horizon Scanning Centre March Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209
 Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
EFFICACY AND SAFETY OF CLOBETASOL PROPIONATE SHAMPOO IN THERAPY OF PSORIASIS OF THE SCALP
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Platonova et al. SJIF Impact Factor 2.786 Volume 4, Issue 04, 238-246. Research Article ISSN 2278 4357 EFFICACY AND SAFETY OF CLOBETASOL PROPIONATE
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Platonova et al. SJIF Impact Factor 2.786 Volume 4, Issue 04, 238-246. Research Article ISSN 2278 4357 EFFICACY AND SAFETY OF CLOBETASOL PROPIONATE
Psoriasis. What is Psoriasis? What causes psoriasis? Medical Topics Psoriasis
 1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
Horizon Scanning Centre January Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652
 Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
Eucrisa. Eucrisa (crisaborole) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Eucrisa Page: 1 of 7 Last Review Date: June 22, 2018 Eucrisa Description Eucrisa (crisaborole)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Eucrisa Page: 1 of 7 Last Review Date: June 22, 2018 Eucrisa Description Eucrisa (crisaborole)
Comparative study of oral methotrexate and acitretin in the treatment of palmoplantar psoriasis
 International Journal of Research in Medical Sciences Parsam SB et al. Int J Res Med Sci. 2015 Jan;3(1):47-52 www.msjonline.org pissn 2320-6071 eissn 2320-6012 Research Article DOI: 10.5455/2320-6012.ijrms20150109
International Journal of Research in Medical Sciences Parsam SB et al. Int J Res Med Sci. 2015 Jan;3(1):47-52 www.msjonline.org pissn 2320-6071 eissn 2320-6012 Research Article DOI: 10.5455/2320-6012.ijrms20150109
A Vitiligo Update for Pharmacists: Current Practices and Future Advances
 A Vitiligo Update for Pharmacists: Current Practices and Future Advances Dalal Hammoudi Halat, RPh, MSc, PhD Assistant Professor School of Pharmacy, Lebanese International University Disclosure Dalal Hammoudi
A Vitiligo Update for Pharmacists: Current Practices and Future Advances Dalal Hammoudi Halat, RPh, MSc, PhD Assistant Professor School of Pharmacy, Lebanese International University Disclosure Dalal Hammoudi
Eucrisa. Eucrisa (crisaborole) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.25 Subject: Eucrisa Page: 1 of 6 Last Review Date: September 15, 2017 Eucrisa Description Eucrisa
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.25 Subject: Eucrisa Page: 1 of 6 Last Review Date: September 15, 2017 Eucrisa Description Eucrisa
Kelly H. Tyler, MD, FACOG, FAAD S052 Gender Dermatology: Diagnosis and Treatment of Genital Skin Disorders Vulvar Dermatitis
 Vulvar Dermatitis Kelly H. Tyler, MD, FACOG, FAAD Assistant Professor The Ohio State University Department of Internal Medicine, Division of Dermatology Department of Obstetrics and Gynecology Center for
Vulvar Dermatitis Kelly H. Tyler, MD, FACOG, FAAD Assistant Professor The Ohio State University Department of Internal Medicine, Division of Dermatology Department of Obstetrics and Gynecology Center for
Comparative efficacy of topical mometasone furoate 0.1% cream vs topical tacrolimus 0.03% ointment in the treatment of atopic dermatitis
 Original Article Comparative efficacy of topical mometasone furoate 0.1% cream vs topical tacrolimus 0.03% ointment in the treatment of atopic dermatitis Md Alauddin Khan *, Lubna Khondker **, Dilshad
Original Article Comparative efficacy of topical mometasone furoate 0.1% cream vs topical tacrolimus 0.03% ointment in the treatment of atopic dermatitis Md Alauddin Khan *, Lubna Khondker **, Dilshad
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Trial Report Synopsis
 Clinical Trial Report Synopsis A phase 2a, proof of concept trial, testing twice daily application of LEO 124249 ointment 30 mg/g in the treatment of mild to moderate inverse psoriasis Design of trial:
Clinical Trial Report Synopsis A phase 2a, proof of concept trial, testing twice daily application of LEO 124249 ointment 30 mg/g in the treatment of mild to moderate inverse psoriasis Design of trial:
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C
 A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Psoriasiform Dermatitis in Children: Calling in the Troops
 Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
Clinical Policy: Brodalumab (Siliq) Reference Number: CP.PHAR.375 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
What s Topical About Topicals?
 What s Topical About Topicals? Megha M. Tollefson, MD Associate Professor of Dermatology and Pediatrics July 29, 2017 2015 MFMER 3513105-1 Disclosures None 2015 MFMER 3513105-2 Outline Topical steroids
What s Topical About Topicals? Megha M. Tollefson, MD Associate Professor of Dermatology and Pediatrics July 29, 2017 2015 MFMER 3513105-1 Disclosures None 2015 MFMER 3513105-2 Outline Topical steroids
Psoriasis the latest recommendations for management: where can primary care make a real difference?
 Dermatology Psoriasis the latest recommendations for management: where can primary care make a real difference? Dr Stephen Kownacki Executive chair, Primary Care Dermatology Society (PCDS) This session
Dermatology Psoriasis the latest recommendations for management: where can primary care make a real difference? Dr Stephen Kownacki Executive chair, Primary Care Dermatology Society (PCDS) This session
Psoriasis is a chronic, inflammatory, Prescribing in children
 Psoriasis in children: current approaches to management Laura Proudfoot BSc, MRCP, Elisabeth Higgins MA, FRCP and Judy Davids RGN Our series Prescribing in children gives practical advice for successful
Psoriasis in children: current approaches to management Laura Proudfoot BSc, MRCP, Elisabeth Higgins MA, FRCP and Judy Davids RGN Our series Prescribing in children gives practical advice for successful
Combination Nonbiologic Therapy in Psoriasis. Sushil Tahiliani, MBBS, MD
 Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
The role of current biologic therapies in psoriasis
 : An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
: An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
British Association of Dermatologists guidelines for the management of lichen sclerosus 2018
 British Association of Dermatologists guidelines for the management of lichen sclerosus 2018 WEB APPENDIX SUPPLEMENTARY INFORMATION Appendix A: Review Protocol Appendix B: Clinical Evidence summary Appendix
British Association of Dermatologists guidelines for the management of lichen sclerosus 2018 WEB APPENDIX SUPPLEMENTARY INFORMATION Appendix A: Review Protocol Appendix B: Clinical Evidence summary Appendix
COMMON SKIN CONDITIONS IN PRIMARY CARE. Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio
 COMMON SKIN CONDITIONS IN PRIMARY CARE Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio DISCLOSURE The Speaker and members of the planning committee do not have a conflict of interest
COMMON SKIN CONDITIONS IN PRIMARY CARE Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio DISCLOSURE The Speaker and members of the planning committee do not have a conflict of interest
(minutes for web publishing)
 Dermatology Subcommittee of the Pharmacology and Therapeutics Advisory Committee (PTAC) Meeting held on 20 October 2017 (minutes for web publishing) Dermatology Subcommittee minutes are published in accordance
Dermatology Subcommittee of the Pharmacology and Therapeutics Advisory Committee (PTAC) Meeting held on 20 October 2017 (minutes for web publishing) Dermatology Subcommittee minutes are published in accordance
Topical Calcipotriol Algorithm
 Topical Calcipotriol Algorithm Is this patient an adult previously diagnosed with psoriasis by a doctor? Do the skin patches look the same as those diagnosed as psoriasis? Is this psoriasis covering an
Topical Calcipotriol Algorithm Is this patient an adult previously diagnosed with psoriasis by a doctor? Do the skin patches look the same as those diagnosed as psoriasis? Is this psoriasis covering an
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPA.01 Effective Date:
 Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
PUVA: Shall we still use it for psoriasis in 2019?
 PUVA: Shall we still use it for psoriasis in 2019? Ben Stoff MD, MA Associate Professor Emory Department of Dermatology Phototherapy: F003 March 1, 2019 DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY
PUVA: Shall we still use it for psoriasis in 2019? Ben Stoff MD, MA Associate Professor Emory Department of Dermatology Phototherapy: F003 March 1, 2019 DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY
Patients Perspectives on the Impact of Genital Psoriasis: A Qualitative Study
 Dermatol Ther (Heidelb) (2017) 7:447 461 DOI 10.1007/s13555-017-0204-3 ORIGINAL RESEARCH Patients Perspectives on the Impact of Genital Psoriasis: A Qualitative Study Jennifer Clay Cather. Caitriona Ryan.
Dermatol Ther (Heidelb) (2017) 7:447 461 DOI 10.1007/s13555-017-0204-3 ORIGINAL RESEARCH Patients Perspectives on the Impact of Genital Psoriasis: A Qualitative Study Jennifer Clay Cather. Caitriona Ryan.
Texas Children's Hospital Dermatology Service PCP Referral Guidelines- Atopic Dermatitis (AD)
 Diagnosis: ATOPIC DERMATITIS (AD) Texas Children's Hospital Dermatology Service PCP Referral Guidelines- Atopic Dermatitis (AD) PATIENT ADVICE: Unfortunately, there is no cure for atopic dermatitis, so
Diagnosis: ATOPIC DERMATITIS (AD) Texas Children's Hospital Dermatology Service PCP Referral Guidelines- Atopic Dermatitis (AD) PATIENT ADVICE: Unfortunately, there is no cure for atopic dermatitis, so
Bioavailability 55% - 77%
 Brand Name: Cosentyx Generic Name: secukinumab Manufacturer 1 : Novartis Pharmaceuticals Corporation Drug Class 2,3 : Antipsoriatic Agent, Interleukin-17A Receptor Antagonist Uses: Labeled Uses 1,2,3 :
Brand Name: Cosentyx Generic Name: secukinumab Manufacturer 1 : Novartis Pharmaceuticals Corporation Drug Class 2,3 : Antipsoriatic Agent, Interleukin-17A Receptor Antagonist Uses: Labeled Uses 1,2,3 :
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS
 COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS Cheryl S. Hankin PhD 1 ; Steven R. Feldman, MD, PhD 2 ; Daniel Pearce, MD 2 1 BioMedEcon, San Jose, CA, USA; 2 Wake Forest University School
COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS Cheryl S. Hankin PhD 1 ; Steven R. Feldman, MD, PhD 2 ; Daniel Pearce, MD 2 1 BioMedEcon, San Jose, CA, USA; 2 Wake Forest University School
Horizon Scanning Centre March Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798
 Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
PSORIASIS BEST PRACTICE IN MANAGEMENT
 PSORIASIS BEST PRACTICE IN MANAGEMENT Objectives Discuss pathology of psoriasis Review types of psoriasis Review triggers and factors affecting disease severity Common comorbidity review Review first and
PSORIASIS BEST PRACTICE IN MANAGEMENT Objectives Discuss pathology of psoriasis Review types of psoriasis Review triggers and factors affecting disease severity Common comorbidity review Review first and
Managing and Minimizing Flare-ups in Atopic Dermatitis
 Managing and Minimizing Flare-ups in Atopic Dermatitis Importance of the skin barrier & how commonly used drugs are impacting it Dr. Benjamin Barankin, MD FRCPC Medical Director & Founder of Toronto Dermatology
Managing and Minimizing Flare-ups in Atopic Dermatitis Importance of the skin barrier & how commonly used drugs are impacting it Dr. Benjamin Barankin, MD FRCPC Medical Director & Founder of Toronto Dermatology
What is atopic dermatitis?
 What is atopic dermatitis? Complex inflammatory skin disorder intense pruritus cutaneous hyperreactivity immune dysregulation Chronic with exacerbations and remissions Affects all ages, but more common
What is atopic dermatitis? Complex inflammatory skin disorder intense pruritus cutaneous hyperreactivity immune dysregulation Chronic with exacerbations and remissions Affects all ages, but more common
50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate).
 DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])
![Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV]) Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])](/thumbs/89/99382632.jpg) Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Details of UNCOVER-2 and UNCOVER-3 Study Results Published in The Lancet
 June 10, 2015 Eli Lilly and Company Lilly Corporate Center Indianapolis, Indiana 46285 U.S.A. +1.317.276.2000 www.lilly.com For Release: Refer to: Immediately Tim Coulom; tim.coulom@lilly.com; 317-771-2241
June 10, 2015 Eli Lilly and Company Lilly Corporate Center Indianapolis, Indiana 46285 U.S.A. +1.317.276.2000 www.lilly.com For Release: Refer to: Immediately Tim Coulom; tim.coulom@lilly.com; 317-771-2241
Risankizumab (by subcutaneous injection) for moderate to severe chronic plaque psoriasis
 NIHR Innovation Observatory Evidence Briefing: November 2017 Risankizumab (by subcutaneous injection) for moderate to severe chronic plaque psoriasis NIHRIO (HSRIC) ID: 9708 NICE ID: 9191 LAY SUMMARY Plaque
NIHR Innovation Observatory Evidence Briefing: November 2017 Risankizumab (by subcutaneous injection) for moderate to severe chronic plaque psoriasis NIHRIO (HSRIC) ID: 9708 NICE ID: 9191 LAY SUMMARY Plaque
The Itch That Rashes. Sarah D. Cipriano, MD, MPH, MS Resident, Dermatology University of Utah
 The Itch That Rashes Sarah D. Cipriano, MD, MPH, MS Resident, Dermatology University of Utah 1 Conflict of Interest No conflict of interest Will discuss off label use of medications 2 3 Most likely diagnosis?
The Itch That Rashes Sarah D. Cipriano, MD, MPH, MS Resident, Dermatology University of Utah 1 Conflict of Interest No conflict of interest Will discuss off label use of medications 2 3 Most likely diagnosis?
Etanercept: a new option in paediatric plaque psoriasis
 : a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
: a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
CADTH CANADIAN DRUG EXPERT COMMITTEE FINAL RECOMMENDATION
 CADTH CANADIAN DRUG EXPERT COMMITTEE FINAL RECOMMENDATION IXEKIZUMAB (Taltz Eli Lilly Canada Inc.) Indication: Moderate to Severe Plaque Psoriasis Recommendation: The CADTH Canadian Drug Expert Committee
CADTH CANADIAN DRUG EXPERT COMMITTEE FINAL RECOMMENDATION IXEKIZUMAB (Taltz Eli Lilly Canada Inc.) Indication: Moderate to Severe Plaque Psoriasis Recommendation: The CADTH Canadian Drug Expert Committee
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Cosentyx, Cosentyx Sensoready) Reference Number: HIM.PA.SP29 Effective Date: 05/17 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See
Clinical Policy: (Cosentyx, Cosentyx Sensoready) Reference Number: HIM.PA.SP29 Effective Date: 05/17 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See
Disclosures Dr. Lynette Margesson
 Management of Vulvar Pruritus 2 nd PANHELLANIC CONGRESS on Lower Genital Tract Disorders December 14-16 Grand Hyatt Athens Lynette J. Margesson MD FRCPC Assistant Professor of Obstetrics & Gynecology and
Management of Vulvar Pruritus 2 nd PANHELLANIC CONGRESS on Lower Genital Tract Disorders December 14-16 Grand Hyatt Athens Lynette J. Margesson MD FRCPC Assistant Professor of Obstetrics & Gynecology and
PNW EPC Drug Effectiveness Review Project Summary Report Atopic Dermatitis New Drug Evaluation: Dupilumab
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Psoriasis. Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement
 Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
DLQI (ESTEEM
 192 Apremilast, an Oral Phosphodiesterase 4 Inhibitor, in Patients With Moderate to Severe Plaque Psoriasis: Pruritus and DLQI Correlations at Week 16 (ESTEEM 1 and 2) Steven R. Feldman, MD, PhD 1 ; Diamant
192 Apremilast, an Oral Phosphodiesterase 4 Inhibitor, in Patients With Moderate to Severe Plaque Psoriasis: Pruritus and DLQI Correlations at Week 16 (ESTEEM 1 and 2) Steven R. Feldman, MD, PhD 1 ; Diamant
Supplementary Table 1: Study and sample characteristics of the included studies
 Supplementary Table 1: Study and sample characteristics of the included studies First author, Imiquimod 5% cream vs. placebo Arican et al. 2004 34 Beutner et al. 1998a 35 Tyring et al. 1998a 36 Beutner
Supplementary Table 1: Study and sample characteristics of the included studies First author, Imiquimod 5% cream vs. placebo Arican et al. 2004 34 Beutner et al. 1998a 35 Tyring et al. 1998a 36 Beutner
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
Using Your ESP* in Pharmacy: How to Improve Treatment Adherence and Patient Outcomes in Psoriasis (*Expanded Scope of Practice)
 Using Your ESP* in Pharmacy: How to Improve Treatment Adherence and Patient Outcomes in Psoriasis (*Expanded Scope of Practice) Patient Case Study in Psoriasis Patient Case Study in Psoriasis William Smith,
Using Your ESP* in Pharmacy: How to Improve Treatment Adherence and Patient Outcomes in Psoriasis (*Expanded Scope of Practice) Patient Case Study in Psoriasis Patient Case Study in Psoriasis William Smith,
THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
MALE GENITAL (PENIS) LICHEN SCLEROSUS
 MALE GENITAL (PENIS) LICHEN SCLEROSUS What are the aims of this leaflet? This leaflet has been written to help you understand more about male genital lichen sclerosus (also known as balanitis xerotica
MALE GENITAL (PENIS) LICHEN SCLEROSUS What are the aims of this leaflet? This leaflet has been written to help you understand more about male genital lichen sclerosus (also known as balanitis xerotica
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Atopic Dermatitis and Topical Antipsoriatics
 Atopic Dermatitis and Topical Antipsoriatics Goal(s): Restrict dermatological drugs only for funded OHP diagnoses. Moderate/severe psoriasis and moderate/severe atopic dermatitis treatments are funded
Atopic Dermatitis and Topical Antipsoriatics Goal(s): Restrict dermatological drugs only for funded OHP diagnoses. Moderate/severe psoriasis and moderate/severe atopic dermatitis treatments are funded
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 11.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
TCIs are only available on prescription and are usually started by a dermatology specialist.
 (TCIs) What are topical calcineurin inhibitors? Topical calcineurin inhibitors are treatments that alter the immune system and have been developed for controlling eczema. There are two types available:
(TCIs) What are topical calcineurin inhibitors? Topical calcineurin inhibitors are treatments that alter the immune system and have been developed for controlling eczema. There are two types available:
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
PNW EPC Drug Effectiveness Review Project Summary Report Atopic Dermatitis New Drug Evaluation: Dupilumab
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Prescribing Information
 Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
PACKAGE LEAFLET: INFORMATION FOR THE USER. Flutarzole 0,05% w/w cream, Fluticasone propionate
 PACKAGE LEAFLET: INFORMATION FOR THE USER Flutarzole 0,05% w/w cream, Fluticasone propionate 1. IDENTIFICATION OF THE MEDICINAL PRODUCT 1.1. Trade name Flutarzole 1.2. Composition Active substance: Fluticasone
PACKAGE LEAFLET: INFORMATION FOR THE USER Flutarzole 0,05% w/w cream, Fluticasone propionate 1. IDENTIFICATION OF THE MEDICINAL PRODUCT 1.1. Trade name Flutarzole 1.2. Composition Active substance: Fluticasone
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
Difference Between Seborrheic Dermatitis and Psoriasis
 Difference Between Seborrheic Dermatitis and Psoriasis www.differencebetween.com Key Difference Seborrheic Dermatitis vs Psoriasis Dermatological conditions are perhaps the most worrisome diseases in the
Difference Between Seborrheic Dermatitis and Psoriasis www.differencebetween.com Key Difference Seborrheic Dermatitis vs Psoriasis Dermatological conditions are perhaps the most worrisome diseases in the
Vulval disease in children
 Vulval disease in children SYDNEY MEDICAL SCHOOL NORTHERN Associate Professor Gayle Fischer MBBS MD FACD gayle.fischer@sydney.edu.au I have no conflict of interests Range of vulval diseases in children
Vulval disease in children SYDNEY MEDICAL SCHOOL NORTHERN Associate Professor Gayle Fischer MBBS MD FACD gayle.fischer@sydney.edu.au I have no conflict of interests Range of vulval diseases in children
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
