Biology 325 Fall 2003 Human anatomy and physiology
|
|
|
- Thomas Dennis
- 6 years ago
- Views:
Transcription
1 SKELETAL MUSCLE PROPERTIES Background : In this experiment, you will investigate the physiological properties of skeletal muscle from the isolated amphibian gastrocnemius. You will examine skeletal muscle functions such as the single twitch, multiple motor unit summation, the relationship between muscle length and tension generated, wave summation and tetanus, and muscle fatigue. The physiology of skeletal muscle was not fully understood until the early 20 th century. Some of the earliest experiments on muscle physiology were performed between 1661 and 1665 by Jan Swammerdam, who demonstrated that an isolated frog muscle could be made to contract when the sciatic nerve was irritated with a metal object. Later, Luigi Galvani ( ) demonstrated that frog muscle responded to electrical currents (Fulton and Wilson, 1966). The basic unit of a muscle is the muscle cell, or myofiber, and whole muscles are made up of bundles of these myofibers. A single muscle fiber has a very regular structure, and is composed of contractile units called myofibrils. Each myofibril consists of an orderly repeated arrangement of the contractile proteins actin and myosin (sarcomere), which slide past each other in the presence of Ca 2+ and ATP. A motor unit is a motor neuron and all the myofibers it innervates. Greater numbers of motor neurons associated with a muscle give finer control over that muscle. Motor neurons release the neurotransmitter acetylcholine from their synaptic bulbs onto the muscle. This junction between a nerve and a muscle is called the neuromuscular junction. The release of acetylcholine at the motor end plate depolarizes the muscle tissue and in turn leads to the release of intracellular calcium from the sarcoplasmic reticulum, a variant of smooth endoplasmic reticulum. This release of intracellular calcium sets in motion the biochemical events that allow actin and myosin to interact and slide past each other, a process ultimately driven by ATP hydrolysis. Skeletal muscle is similar to nerve tissue in that it responds to a stimulus in an all-or-none fashion. This response is called a twitch. Depending on the intensity and frequency of stimulation, greater numbers of fibers are activated. By increasing the number of active muscle fibers, the muscle is able to increase the force it generates. Muscle contractions are graded in size by wave summation, which depends on stimulus frequency, or by recruitment of more motor units. Thus, muscles with large cross-sectional areas are able to generate larger forces than those with small cross-sectional areas. In this experiment, you will examine the basic principles of skeletal muscle physiology, including the all-or-none response, the effect of stimulus intensity and frequency on contraction force and the phenomenon of muscle fatigue. These experiments illustrate the collective understanding of muscle physiology gained from over 400 years of
2 research (adapted from AD Instruments Lab Protocols, for complete document see Experimental apparatus and tissue preparation : Dissection. Double pith a 3-4" Northern Grass Frog (Rana pipiens). Cut the skin all the way around the abdomen, and peel away the skin over the legs by pulling down with a pair of tissue-gripping forceps. Sever one leg by cutting through the femur. Remove the muscles of the upper leg and trim the femur to about one inch. Identify the gastrocnemius and measure its resting length. Now separate the gastrocnemius muscle from other muscles of the calf, and cut through the Achilles tendon above the ankle leaving most of the tendon attached to the muscle preparation. Next, cut the tibiofibula at the knee, along with the other calf muscles. You should be left with the gastrocnemius and the Achilles tendon at the distal end, and an inch of femur attached to the proximal end of the muscle. Place the muscle in a bath of room temperature Ringer's solution at ph (For a diagrammatic representation see Appendix I: Dissection) The muscle will contract in vitro when stimulated by an adequate electrical stimulus, just as it would in vivo when a motor unit is stimulated by a motor neuron. Grass Stimulator. The SD9 stimulators are electronic devices that supply a known stimulus to the preparation. We can set the intensity (voltage), duration (time in msec), and frequency (number of stimulation pulses/sec) of stimulation with the dials on the front panel of the instrument. Note that the switch below each dial is a multiplier that multiplies the dial value by 0.01 to 10X. Transducer. The device on your ring stand is a semi-isometric strain gage transducer (UFI Inc. Model 1030) which converts the mechanical tension produced by muscle contraction into an electronic signal that the recording system can detect. Other kinds of transducers exist for pressure, temperature, muscle shortening, fluid flow, sound, and light--each of these forms of energy can be converted by a transducer to an electrical signal. Bridge amplifier and PowerLab. The PowerLab units contain 2 to 4 banks of analogto-digital conversion circuits which will record electrical signals from our preparation. The bridge amplifier is a Wheatstone bridge circuit which collects data from the strain gauge transducer and provides a signal to PowerLab proportional to tension produced by contracting muscle. In some of the experimental stations the transducer may be connected directly to the PowerLab unit (the amplification is built into the PowerLab unit) PC. At your table is a desktop computer that can be used to compose graphs, analyze experimental results, work with text, and record experimental data. You will record data using Chart 4.0 PowerLab software. This software simulates a pen-and-paper chart recorder.
3 Getting Started: 1. Starting up and working with PowerLab. a. Make sure the PowerLab unit in your station is turned on -- to turn it on use the power switch on the back of the unit. b. Log onto the PC. c. Launch Chart , an application that simulates a pen-and-paper chart recorder. d. The screen should look similar to Figure 1. The window shown in Figure 1 looks rather complex, but the software is very user friendly. We will introduce you to a few key features and you will gradually pick up other details as you work with the program. Figure 1 i. By selecting the Rate pop-up menu you can choose the sampling rate the number of data samples that are stored per unit time, and the time taken by each sampling division to scroll past. For details see Figure 2 below. By altering the value in this pop-up menu you are doing the same as controlling paper speed in an old-time pen-and-paper recorder.
4 Figure 2 ii. By selecting the Range pop-up menu above each channel icon (Figure 3) you can change the sensitivity of your recording. The sensitivity can be set from 2 mv to 10 V -- the higher the value the less sensitive the recording. For example, a channel set at a sensitivity of 500 mv will have lower sensitivity than if set at 100 mv. Figure 3 iii. At times throughout the experiment and data analysis procedure you will be asked to bring up the Channel Function pop-up menu (Figure 4). This will allow you to turn the channel off and access settings for the bridge amp you have connected to the PowerLab unit, among other features. Your instructor will demonstrate these features in more detail when appropriate. Figure 4
5 iv. Chart uses a visual metaphor of a mechanical chart recorder: recorded data scroll across the data display area from the right of the window as if the display area were a roll of paper in such a device, with new data being drawn at the right and old data moving left (see Figure 1). To start sampling, simply click the Start button at the bottom right of the Chart window (see Figure 1). -- this means that you will see data be acquired as it moves across the screen. Whether the data is recorded or not depends on the status of the Record/Monitor icon to the left of the Start button (Figure 1 and Figure 5). If you place a large X over the Record/Monitor button by clicking it, the data will be displayed but not recorded -- this is a great feature to preview the results of a procedure without using memory to store data. If the large X is removed from the Record/Monitor icon by clicking on it, the data will be displayed and recorded -- do this only when you are ready to record results of a specific procedure. For more details see Figure 5. Figure 5 v. Figure 6 shows the changes in appearance that occur at the bottom of the Chart window when you start sampling and in this case also recording. You can enter a comment while recording so you know what procedure or what settings the experiment involves. To enter a comment while recording, type in the text entry area at the bottom of the Chart window, and press the Enter or Return key to add the comment to the file at the time the key is pressed. This features allows you to identify the specifics of an experiment or recording when you scroll through it later. Figure 6 To look at several comments at once, locate a comment in a file, or delete or edit comments, choose Comments from the Windows menu to bring up the Comments window. The comments are listed in a scrolling field in the window in the order that they appear left to right across a file, with the comment numbers in boxes.
6 Try out some of the above features of CHART -- don t hesitate to try anything! When you re done exploring, check with an instructor to see that your PowerLab is ready to record. 2. Secure the muscle to the apparatus (Figure 7) Figure 7 (autographed at request) a. Remove the muscle from its Ringer s bath. Attach the muscle to the femur clamp with the bone attached parallel to the clamp and at right angles to the muscle. b. At the top end of the muscle, attach the preparation to the isometric transducer using the fishhook thrust through the Achilles' tendon and tied to the transducer lever. Use two- three leaves of the transducer for best sensitivity and isometric recording. Stretch the muscle between the femur clamp and the transducer so that the muscle length is close to resting muscle length you measured prior to the dissection procedure. c. Attach wires from the stimulator output to the femur clamp (via a screw terminal if possible) and to the fishhook at the top (via a soldered connection). Polarity (which one is + or -) doesn't matter. d. Once you've got the muscle in place move the clamps apart so as to exert a slight tension on the resting muscle. e. Liberally douse the muscle with Ringer's saline solution now and throughout the procedure. Place a beaker underneath to catch the excess. It is extremely important that you keep the muscle wet throughout the procedure. f. In some units the transducer's output cable plugs into the bridge amplifier which in turn plugs into PowerLab Channel 1 by a BNC shielded cable. In other units the transducer's output cable plugs directly into PowerLab Channel 1.
7 2. Calibration. During the experiments, after the muscle is set in place, and between individual procedures, you will calibrate the transducer/powerlab apparatus by placing a weight (10 or 20 grams) on the end of the transducer lever and recording the displacement caused by that weight. Record at a moderate speed of 1-5 s/div, use Rate/Time pop-up menu to set this. You will use this calibration to compute the tension developed in your muscle under various conditions. Repeat the calibration procedure at frequent intervals during your work, and record the new calibration on the PowerLab record as a comment or in your notebook for reference. During experimentation, adjust sensitivity (use Range pop-up menu) as needed (except during an experimental procedure) to yield a good-sized contraction record. Always record the settings (especially sensitivity) in your written notes and/or as a comment in CHART. Liberally douse the muscle with Ringer's saline solution now and throughout the procedure. Place a beaker underneath to catch the excess. Experimental procedures : 1. Threshold and recruitment of fibers. For the first experiment, we will find the lowest stimulus voltage needed to start a muscle action potential and cause contraction. a. The sensitivity should be set relatively low. b. Make sure that the stimulator wires are properly attached to the muscle prep, and the stimulator is set in the following fashion: Duration: 30 msec Intensity: 0.1 V Frequency: single pulse c. Stimulate the muscle with single pulses using the mode switch on the stimulator. d. Record at a slow chart speed and note in the Comments the voltage applied for each stimulus. e. Gradually increase the stimulus intensity in 0.1 V or 0.2 V increments, giving a single pulse at each voltage -- keep increasing the voltage until you get a response. That stimulus intensity is the threshold for the most sensitive fibers in your muscle. f. Continue increasing the stimulus intensity ( now in V increments) until a maximal contraction is obtained.
8 g. Now, with CHART running at a slow/moderate speed, quickly repeat this stepwise progression ( V increments) from threshold to high stimulus intensities to obtain a clean, short record of the effects of recruitment of muscle fibers. Record comments that include the stimulus voltage used. h. Save your data by click-dragging from the File menu down to SAVE or SAVE AS. Give your file a new name that you ll recognize and remember. Save to the M-drive. i. Investigate the effects of applying a train of repeated subliminal or subthreshold stimuli. Do successive stimuli add together to cause a contraction? j. Does duration of the stimulus alter the threshold? Adjust duration to various values and record the voltage needed at each duration to elicit a certain level of response. 2. The muscle twitch. The individual muscle contractions seen with a single stimulation are called twitch contractions. A maximal twitch involves all the muscle's fibers contracting at once and was observed above at the point when further increases in stimulus voltage didn't cause any further increase in tension. During this procedure you will record a single maximal twitch at very high speed in Channel 1, also recording in Channel 2 the exact time when you applied the stimulus. a. Calibrate your prep as described above. b. Click Channel 2 Range pop-up menu and turn Channel 2 on. c. Use a black probe or dual-banana plug cable provided by your instructor to connect the stimulator output to PowerLab Input 2. NOTE: Do not exceed 10 V stimulus voltages when connected to PowerLab or you'll blow the A/D board, a very expensive mistake (has been done before ). d. Set up Channel 2 to record on 10 V range. The size of the stimulus wave is irrelevant; all we need is a mark time for the stimulation. Check with the Input amplifier in the Channel 2 pop-up menu to see that the stimulator pulse is displayed. e. Record several individual twitches at high speed, about msec/division -- use a maximal stimulus intensity and keep frequency and stimulus duration set as in 1. Before you start, check that the sampling rate is maximal (400 samples/div) so that you'll be able to precisely measure time in your recordings. You should see the muscle twitch in Channel 1 and the stimulus wave on Channel 2. f. Don't forget to bathe the muscle periodically with Ringer's saline solution to rinse away waste products and keep the prep moist. g. Save your data again by choosing SAVE from the File Menu.
9 h. Disconnect the stimulator from PowerLab Channel 2 and turn Channel 2 off using the Channel 2 pop-up menu. 3. Twitch fusion and tetany. a. Calibrate your prep as described above. b. Use a stimulus voltage which will produce a maximal contraction to investigate the effects of applying a series of repeated stimuli to the muscle. c. Set the stimulator frequency to 1/sec, then 2/sec, then 3/sec, 5/sec, 7/sec, 10/sec, 15/sec, 20/sec. Stimulus duration stays as above in 1 and 2. d. Apply a train of repeated stimuli by briefly switching the stimulator Mode switch to Repeat or Multiple. Use only a short period of stimulation, about 5 sec, and be consistent in applying the train of stimuli. Allow the muscle to rest for about 30 sec. between stimulations. e. Record all information in your notebook--stimulus frequency, voltage, recording speed, sensitivity -- also insert appropriate Comments during the recording. The muscle twitches you observe will fuse together, and at high frequency stimulation will reach a condition of tetanic, or constant contraction. If the recording goes off scale you ll have to reduce the sensitivity, recalibrate, and repeat the experiment for all stimulation frequencies. f. Save your data again by choosing SAVE from the File Menu. 4. Length-tension curve. You will examine the effects of changing the initial length of the muscle on the tension generated by stimulation with a maximal stimulus. a. Calibrate your prep as described above. b. Move the transducer up or down and measure the distance between clamps ( muscle length ) with a ruler or calipers. c. At each length stimulate the muscle with a single pulse using a maximal stimulus to elicit the maximum muscle tension. Use only a short range of lengths in your experiment -- perhaps cm total. Collect 8-10 data points. For each contraction, save the length value in a comment. Make sure you recalibrate every time you change muscle length. d. Save your data again by choosing SAVE from the File Menu.
10 5. Pre-loading. Examine the effects of pre-loading on muscle contraction. a. Invert the apparatus by switching the positions of the femur clamp and transducer. Stretch the muscle to the resting muscle length in the frog. b. Hang a pan for weights from the bottom of the muscle, below the transducer, start the recording at low paper speed -- record continuously throughout this procedure. Gradually increase the load by successively adding 10 to 400 g to the pan (work by increments of 10 or 20 g). Each time you add weight, let the baseline stabilize, insert an appropriate comment with weight, and stimulate muscle with a supermaximal stimulus to produce a twitch. Add the next weight increment and repeat procedure. 6. Fatigue. When you have completed all the above experiments [not before!], look at the effects of a continuous, high frequency stimulation on muscle tension. a. Calibrate your prep as described above. b. Reduce the sampling rate and recording time base so you can record over a longer period of time. c. Apply a tetanizing pulse (20-50 pulses/sec) and watch the development of fatigue in the muscle. With low paper speed, record for about 3 minutes, then turn off the stimulator and allow the muscle to relax completely before you stop the recording. d. During the stimulation apply several drops of Ringer's after tension has declined significantly. Does tension increase after a Ringer's wash? e. Save your data again by choosing SAVE from the File Menu. Clean up You have completed your experimental procedures. Before leaving do the following: 1. Remove the muscle from the apparatus and dispose of it in an animal waste bag. 2. Clean all instruments and equipment. 3. Close the Chart program, shutdown the Mac, and turn off the power in the back of the PowerLab unit. 4. Data analysis will be done next cycle.
11 Analysis : Before starting analysis covert your data to grams as necessary by using calibration waves you collected along the procedure and the Units Conversion feature of Chart Threshold and recruitment of fibers. Examine your data recording from Part 1. a. Place the marker (M) on the baseline of the waveform in the Force channel. b. Using the mouse, place the waveform cursor at the top of the contraction peak. Record the contraction amplitude in Table 1. c. From your data, determine the minimum voltage required to elicit a maximal contraction. This is the maximum excitation voltage, a maximal stimulus. Determine your value for a supramaximal stimulus by multiplying this voltage by 1.5. Record these values in Table The muscle twitch. a. Go to a recording of a high speed twitch from Part 2. Highlight both channels so that you include the full twitch. Zoom into this window and using the marker (M) and the waveform cursor, measure the duration of the latent period, contraction period, and relaxation period. Record these values in Table 3. b. Either copy or print the zoom window. Identify the latent period, contraction period, relaxation period. 3. Twitch fusion and tetany. a. Examine your data from Part 3. There will be eight blocks of recorded data. b. For each data block, determine the maximum contraction force using the marker and waveform cursor. c. Record your results in Table 4 of your Data Notebook. 4. Length-tension curve. a. Examine your data from part 4. b. For each initial muscle length, measure force produced by muscle contraction use marker (M) and waveform cursor. Don t forget to recalibrate after each trial. c. Record your data in Table 5
12 5. Pre-loading. a. Examine your data from Part 5 go to beginning of the low speed recording. b. Recalibrate your data based upon the displacement produced by your first 10 or 20 load. c. Return the marker (M) to baseline, previous to addition of any load. d. Using the waveform cursor, measure the displacement produced by addition of weight. This is the preload force. e. Next, determine the raw twitch force with the waveform cursor by placing the cursor at the peak of the contraction produced at this pre-load.. Enter your results in Table 6. f. repeat steps d and e for the other data blocks g. Calculate the net twitch force by subtracting the preload (baseline) value from the raw twitch force value. 6. Muscle fatigue a. Examine your data from Part 6. b. Place the marker (M) on the waveform immediately prior to stimulation. c. Use the waveform cursor to determine maximum contraction force. Record this value and the time of maximal stimulation in Table 7 of your Data Notebook. d. Determine the contraction force at t= 15, 30, 60, 90, 120, 150, and 180 seconds after stimulation. f. Record your results in Table 6 of your Data Notebook.
Labs #7 and #8: Vertebrate Skeletal Muscle
 Labs #7 and #8: Vertebrate Skeletal Muscle In this experiment, you will investigate the physiological properties of skeletal muscle from the isolated toad gastrocnemius. Concepts to understand include
Labs #7 and #8: Vertebrate Skeletal Muscle In this experiment, you will investigate the physiological properties of skeletal muscle from the isolated toad gastrocnemius. Concepts to understand include
1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002
 1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002 I. Introduction. Read the introductory material in your lab manual Marieb Ex 16A: Skeletal Muscle Physiology Frog
1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002 I. Introduction. Read the introductory material in your lab manual Marieb Ex 16A: Skeletal Muscle Physiology Frog
Exercise 6: Muscle Physiology II Twitch & Summation
 Exercise 6: Muscle Physiology II Twitch & Summation Text Reading: Silverthorn, 5 th ed. 412 419, 425 427; 6 th ed. pg. 410 420 In this exercise, we will investigate the physiology of contraction in the
Exercise 6: Muscle Physiology II Twitch & Summation Text Reading: Silverthorn, 5 th ed. 412 419, 425 427; 6 th ed. pg. 410 420 In this exercise, we will investigate the physiology of contraction in the
Humans make voluntary decisions to talk, walk, stand up, or sit down. The
 2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define motor unit, twitch, latent period, contraction phase, relaxation phase, threshold, summation, tetanus, fatigue, isometric contraction,
2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define motor unit, twitch, latent period, contraction phase, relaxation phase, threshold, summation, tetanus, fatigue, isometric contraction,
BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject
 Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Lab 3: Excitability & Response of Skeletal Muscle
 Lab 3: Excitability & Response of Skeletal Muscle รศ.ดร.นพ. ช ยเล ศ ชยเลศ พ ช ตพรช ย พชตพรชย ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล Objectives 1. Threshold, submaximal, maximal, supramaximal stimulus
Lab 3: Excitability & Response of Skeletal Muscle รศ.ดร.นพ. ช ยเล ศ ชยเลศ พ ช ตพรช ย พชตพรชย ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล Objectives 1. Threshold, submaximal, maximal, supramaximal stimulus
Lab #9: Muscle Physiology
 Background Overview of Skeletal Muscle Contraction Sarcomere Thick Filaments Skeletal muscle fibers are very large, elongated cells (Fig 9.1). Roughly 80% of the content of each muscle fiber consists of
Background Overview of Skeletal Muscle Contraction Sarcomere Thick Filaments Skeletal muscle fibers are very large, elongated cells (Fig 9.1). Roughly 80% of the content of each muscle fiber consists of
Muscles & Physiology
 Collin County Community College BIOL 2401 Muscles & Physiology 1 Tension Development The force exerted by a contracting muscle cell or muscle group on an object is called muscle tension, and the opposing
Collin County Community College BIOL 2401 Muscles & Physiology 1 Tension Development The force exerted by a contracting muscle cell or muscle group on an object is called muscle tension, and the opposing
Skeletal muscles are composed of hundreds to thousands of individual cells,
 2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define these terms used in describing muscle physiology: multiple motor unit summation, maximal stimulus, treppe, wave summation,
2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define these terms used in describing muscle physiology: multiple motor unit summation, maximal stimulus, treppe, wave summation,
Chapter 9 Muscle. Types of muscle Skeletal muscle Cardiac muscle Smooth muscle. Striated muscle
 Chapter 9 Muscle Types of muscle Skeletal muscle Cardiac muscle Smooth muscle Striated muscle Chapter 9 Muscle (cont.) The sliding filament mechanism, in which myosin filaments bind to and move actin
Chapter 9 Muscle Types of muscle Skeletal muscle Cardiac muscle Smooth muscle Striated muscle Chapter 9 Muscle (cont.) The sliding filament mechanism, in which myosin filaments bind to and move actin
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation
 Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Background The arterial system functions as a pressure reservoir. Blood enters via the heart and exits through the capillaries.
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Background The arterial system functions as a pressure reservoir. Blood enters via the heart and exits through the capillaries.
Exercise 5: Muscle Physiology I - Electromyography
 Exercise 5: Muscle Physiology I - Electromyography Readings: Silverthorn, 6 th ed. pg. 410 420 Your brain communicates with your muscles through action potentials on the motor neurons, which are then transmitted
Exercise 5: Muscle Physiology I - Electromyography Readings: Silverthorn, 6 th ed. pg. 410 420 Your brain communicates with your muscles through action potentials on the motor neurons, which are then transmitted
Skeletal Muscle Qiang XIA (
 Skeletal Muscle Qiang XIA ( 夏强 ), PhD Department of Physiology Rm C518, Block C, Research Building, School of Medicine Tel: 88208252 Email: xiaqiang@zju.edu.cn Course website: http://10.71.121.151/physiology
Skeletal Muscle Qiang XIA ( 夏强 ), PhD Department of Physiology Rm C518, Block C, Research Building, School of Medicine Tel: 88208252 Email: xiaqiang@zju.edu.cn Course website: http://10.71.121.151/physiology
Dynamic Muscle Recorder
 Dynamic Muscle Recorder Team 17 By: Michael Petrowicz Farrukh Rahman James Porteus Client: Biomedical Engineering Department Dr. Enderle Executive Summary The biomedical department requires a lab setup
Dynamic Muscle Recorder Team 17 By: Michael Petrowicz Farrukh Rahman James Porteus Client: Biomedical Engineering Department Dr. Enderle Executive Summary The biomedical department requires a lab setup
Exercise 2: Effects of Cold Temperature Aim: To record changes in heart rate after the heart is bathed in cold Ringer s solution.
 Experiment AM-3: Heart Muscle Warning: The heart preparation used in this experiment is functional for a limited period of time. If the muscle is bathed periodically in Ringer s solution, it will work
Experiment AM-3: Heart Muscle Warning: The heart preparation used in this experiment is functional for a limited period of time. If the muscle is bathed periodically in Ringer s solution, it will work
Warning: Avoid putting the tip of the syringe needle too deeply into the hole to avoid damage to the heart tissue.
 Experiment AM-12: Crayfish Electrocardiogram (ECG) Exercise 1: The Crayfish ECG and Heart Rate Aim: To record the electrical trace (ECG) produced by the contraction of a resting heart, and to determine
Experiment AM-12: Crayfish Electrocardiogram (ECG) Exercise 1: The Crayfish ECG and Heart Rate Aim: To record the electrical trace (ECG) produced by the contraction of a resting heart, and to determine
Lesson 2 EMG 2 Electromyography: Mechanical Work
 Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 2 EMG 2 Electromyography: Mechanical Work PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 5.23.2006 BIOPAC Systems, Inc. 42
Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 2 EMG 2 Electromyography: Mechanical Work PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 5.23.2006 BIOPAC Systems, Inc. 42
Lab 5: Electromyograms (EMGs)
 Lab 5: Electromyograms (EMGs) Overview A motorneuron and all the muscle fibers that it innervates is known as a motor unit. Under normal circumstances, a neuronal action potential activates all of the
Lab 5: Electromyograms (EMGs) Overview A motorneuron and all the muscle fibers that it innervates is known as a motor unit. Under normal circumstances, a neuronal action potential activates all of the
10 - Muscular Contraction. Taft College Human Physiology
 10 - Muscular Contraction Taft College Human Physiology Muscular Contraction Sliding filament theory (Hanson and Huxley, 1954) These 2 investigators proposed that skeletal muscle shortens during contraction
10 - Muscular Contraction Taft College Human Physiology Muscular Contraction Sliding filament theory (Hanson and Huxley, 1954) These 2 investigators proposed that skeletal muscle shortens during contraction
FT-302 Force Transducer
 Technical Note FT-302 LabScribe is a trademark of 2015 Overview The FT-302 is a high-sensitivity dual-range research grade force transducer designed to measure forces in the 0.005 to 10 gram and 0 to 100
Technical Note FT-302 LabScribe is a trademark of 2015 Overview The FT-302 is a high-sensitivity dual-range research grade force transducer designed to measure forces in the 0.005 to 10 gram and 0 to 100
Cardiovascular Effects of Exercise. Background Cardiac function
 Cardiovascular Effects of Exercise In this experiment, you will record an electrocardiogram, or ECG, and finger pulse from a healthy volunteer. You will then compare the ECG and pulse recordings when the
Cardiovascular Effects of Exercise In this experiment, you will record an electrocardiogram, or ECG, and finger pulse from a healthy volunteer. You will then compare the ECG and pulse recordings when the
Electromyography II Laboratory (Hand Dynamometer Transducer)
 (Hand Dynamometer Transducer) Introduction As described in the Electromyography I laboratory session, electromyography (EMG) is an electrical signal that can be recorded with electrodes placed on the surface
(Hand Dynamometer Transducer) Introduction As described in the Electromyography I laboratory session, electromyography (EMG) is an electrical signal that can be recorded with electrodes placed on the surface
EQA DISCUSSION QUESTIONS: INFLUENCE OF MUSCLE FIBER TYPE ON MUSCLE CONTRACTION. Influence of Muscle Fiber Type on Muscle Contraction
 0907T_c13_205-218.qxd 1/25/05 11:05 Page 209 EXERCISE 13 CONTRACTION OF SKELETAL MUSCLE 209 Aerobic cellular respiration produces ATP slowly, but can produce large amounts of ATP over time if there is
0907T_c13_205-218.qxd 1/25/05 11:05 Page 209 EXERCISE 13 CONTRACTION OF SKELETAL MUSCLE 209 Aerobic cellular respiration produces ATP slowly, but can produce large amounts of ATP over time if there is
Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue
 Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue Manual Revision PL3.7.0 M3.0.6 080706 Richard Pflanzer,
Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue Manual Revision PL3.7.0 M3.0.6 080706 Richard Pflanzer,
Chapter 9 - Muscle and Muscle Tissue
 Chapter 9 - Muscle and Muscle Tissue I. Overview of muscle tissue A. Three muscle types in the body: B. Special characteristics 1. Excitability: able to receive and respond to a stimulus 2. Contractility:
Chapter 9 - Muscle and Muscle Tissue I. Overview of muscle tissue A. Three muscle types in the body: B. Special characteristics 1. Excitability: able to receive and respond to a stimulus 2. Contractility:
Muscle and Muscle Tissue
 Muscle and Muscle Tissue Make up about half of total body mass Exerts force by converting chemical energy, ATP, to mechanical energy Muscle tissue is classified based on Shape Number and position of nuclei
Muscle and Muscle Tissue Make up about half of total body mass Exerts force by converting chemical energy, ATP, to mechanical energy Muscle tissue is classified based on Shape Number and position of nuclei
Skeletal Muscle. Connective tissue: Binding, support and insulation. Blood vessels
 Chapter 12 Muscle Physiology Outline o Skeletal Muscle Structure o The mechanism of Force Generation in Muscle o The mechanics of Skeletal Muscle Contraction o Skeletal Muscle Metabolism o Control of Skeletal
Chapter 12 Muscle Physiology Outline o Skeletal Muscle Structure o The mechanism of Force Generation in Muscle o The mechanics of Skeletal Muscle Contraction o Skeletal Muscle Metabolism o Control of Skeletal
Session 3-Part 2: Skeletal Muscle
 Session 3-Part 2: Skeletal Muscle Course: Introduction to Exercise Science-Level 2 (Exercise Physiology) Presentation Created by Ken Baldwin, M.ED, ACSM-H/FI Copyright EFS Inc. All Rights Reserved. Skeletal
Session 3-Part 2: Skeletal Muscle Course: Introduction to Exercise Science-Level 2 (Exercise Physiology) Presentation Created by Ken Baldwin, M.ED, ACSM-H/FI Copyright EFS Inc. All Rights Reserved. Skeletal
About This Chapter. Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Pearson Education, Inc.
 About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
Skeletal Muscle and the Molecular Basis of Contraction. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Lab #3: Electrocardiogram (ECG / EKG)
 Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation
 Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Exercise 1: The ECG and the Pulse in a Resting Subject Aim: To measure and correlate the ECG and the pulse in a resting individual.
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Exercise 1: The ECG and the Pulse in a Resting Subject Aim: To measure and correlate the ECG and the pulse in a resting individual.
Name: Teacher: Olsen Hour:
 Name: Teacher: Olsen Hour: Dem Bones Textbook p84-85, 88-99 61 In all exercises, quizzes and tests in this class, always answer in your own words. That is the only way that you can show that you understand
Name: Teacher: Olsen Hour: Dem Bones Textbook p84-85, 88-99 61 In all exercises, quizzes and tests in this class, always answer in your own words. That is the only way that you can show that you understand
Lab #3: Electrocardiogram (ECG / EKG)
 Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
PSK4U THE NEUROMUSCULAR SYSTEM
 PSK4U THE NEUROMUSCULAR SYSTEM REVIEW Review of muscle so we can see how the neuromuscular system works This is not on today's note Skeletal Muscle Cell: Cellular System A) Excitation System Electrical
PSK4U THE NEUROMUSCULAR SYSTEM REVIEW Review of muscle so we can see how the neuromuscular system works This is not on today's note Skeletal Muscle Cell: Cellular System A) Excitation System Electrical
iworx Sample Lab Experiment AN-5: Cockroach Leg Mechanoreceptors
 Experiment AN-5: Cockroach Leg Mechanoreceptors Exercise 1: Chordotonal Organs Aim: To explore the basic characteristics of the chordotonal organs, their response to direction and intensity of leg movement,
Experiment AN-5: Cockroach Leg Mechanoreceptors Exercise 1: Chordotonal Organs Aim: To explore the basic characteristics of the chordotonal organs, their response to direction and intensity of leg movement,
iworx Sample Lab Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study
 Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study What is Biofeedback? Biofeedback is a technique that people can use to learn to control their body's physiological functions. The definition
Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study What is Biofeedback? Biofeedback is a technique that people can use to learn to control their body's physiological functions. The definition
MUSCLE TISSUE (MUSCLE PHYSIOLOGY) PART I: MUSCLE STRUCTURE
 PART I: MUSCLE STRUCTURE Muscle Tissue A primary tissue type, divided into: skeletal muscle cardiac muscle smooth muscle Functions of Skeletal Muscles Produce skeletal movement Maintain body position Support
PART I: MUSCLE STRUCTURE Muscle Tissue A primary tissue type, divided into: skeletal muscle cardiac muscle smooth muscle Functions of Skeletal Muscles Produce skeletal movement Maintain body position Support
The organization of skeletal muscles. Excitation contraction coupling. Whole Skeletal Muscles contractions. Muscle Energetics
 Muscle and Movement The organization of skeletal muscles Excitation contraction coupling Whole Skeletal Muscles contractions Muscle Energetics The molecular bases of movement Muscular cells use molecular
Muscle and Movement The organization of skeletal muscles Excitation contraction coupling Whole Skeletal Muscles contractions Muscle Energetics The molecular bases of movement Muscular cells use molecular
The Musculoskeletal System. Chapter 46
 The Musculoskeletal System Chapter 46 Types of Skeletal Systems Changes in movement occur because muscles pull against a support structure Zoologists recognize three types: 1. Hydrostatic skeletons a fluid
The Musculoskeletal System Chapter 46 Types of Skeletal Systems Changes in movement occur because muscles pull against a support structure Zoologists recognize three types: 1. Hydrostatic skeletons a fluid
Organismic Biology Bio 207. Lecture 6. Muscle and movement; sliding filaments; E-C coupling; length-tension relationships; biomechanics. Prof.
 Organismic Biology Bio 207 Lecture 6 Muscle and movement; sliding filaments; E-C coupling; length-tension relationships; biomechanics Prof. Simchon Today s Agenda Skeletal muscle Neuro Muscular Junction
Organismic Biology Bio 207 Lecture 6 Muscle and movement; sliding filaments; E-C coupling; length-tension relationships; biomechanics Prof. Simchon Today s Agenda Skeletal muscle Neuro Muscular Junction
THE INFLUENCE OF IONS ON THE CONTRACTION OF ISOLATED EARTHWORM SMOOTH MUSCLE
 THE INFLUENCE OF IONS ON THE CONTRACTION OF ISOLATED EARTHWORM SMOOTH MUSCLE BACKGROUND READING Animal Physiology by Hill, Wyse & Anderson, 2004: pp. 484 486. ANIMALS & EQUIPMENT Living material Large
THE INFLUENCE OF IONS ON THE CONTRACTION OF ISOLATED EARTHWORM SMOOTH MUSCLE BACKGROUND READING Animal Physiology by Hill, Wyse & Anderson, 2004: pp. 484 486. ANIMALS & EQUIPMENT Living material Large
2/19/2018. Learn and Understand:
 Muscular System with Special Emphasis on Skeletal Muscle Anatomy and Physiology Learn and Understand: The definition of cell changes again The contractile unit of muscle is the sarcomere. ATP and Ca 2+
Muscular System with Special Emphasis on Skeletal Muscle Anatomy and Physiology Learn and Understand: The definition of cell changes again The contractile unit of muscle is the sarcomere. ATP and Ca 2+
CHAPTER 6 2/9/2016. Learning Objectives List the four traits that all muscle types have in common.
 Learning Objectives List the four traits that all muscle types have in common. CHAPTER 6 The Muscular System Demonstrate and explain the use of antagonistic muscle pairs. Describe the attachment of muscle
Learning Objectives List the four traits that all muscle types have in common. CHAPTER 6 The Muscular System Demonstrate and explain the use of antagonistic muscle pairs. Describe the attachment of muscle
Physiology Lessons for use with the Biopac Student Lab. Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG
 Physiology Lessons for use with the Biopac Student Lab Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine
Physiology Lessons for use with the Biopac Student Lab Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine
Muscle Tissue- 3 Types
 AN INTRODUCTION TO MUSCLE TISSUE Muscle Tissue- 3 Types Skeletal muscle (focus on these) Cardiac muscle Smooth muscle FUNCTIONS OF SKELETAL MUSCLES Produce movement of the skeleton Maintain posture and
AN INTRODUCTION TO MUSCLE TISSUE Muscle Tissue- 3 Types Skeletal muscle (focus on these) Cardiac muscle Smooth muscle FUNCTIONS OF SKELETAL MUSCLES Produce movement of the skeleton Maintain posture and
Muscle Physiology. Dr. Ebneshahidi Ebneshahidi
 Muscle Physiology Dr. Ebneshahidi Skeletal Muscle Figure 9.2 (a) Functions of the muscular system 1. Locomotion body movements are due to skeletal muscle contraction. 2. Vasoconstriction and vasodilatation
Muscle Physiology Dr. Ebneshahidi Skeletal Muscle Figure 9.2 (a) Functions of the muscular system 1. Locomotion body movements are due to skeletal muscle contraction. 2. Vasoconstriction and vasodilatation
Cardiovascular Physiology
 Cardiovascular Physiology The mammalian heart is a pump that pushes blood around the body and is made of four chambers: right and left atria and right and left ventricles. The two atria act as collecting
Cardiovascular Physiology The mammalian heart is a pump that pushes blood around the body and is made of four chambers: right and left atria and right and left ventricles. The two atria act as collecting
Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling
 Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling Background The movement of parts of the body is accomplished through a system of levers composed of skeletal muscles and bones. In a lever,
Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling Background The movement of parts of the body is accomplished through a system of levers composed of skeletal muscles and bones. In a lever,
Smooth Cardiac Skeletal Location Around tubes Heart tissue attached to skeleton Moves stuff thru Heart beat pumps Moves body parts
 Biology 067 - Muscular system A. Type of muscles: Smooth Cardiac Skeletal Location Around tubes Heart tissue attached to skeleton Function Moves stuff thru Heart beat pumps Moves body parts tubes blood
Biology 067 - Muscular system A. Type of muscles: Smooth Cardiac Skeletal Location Around tubes Heart tissue attached to skeleton Function Moves stuff thru Heart beat pumps Moves body parts tubes blood
Lab 4: Introduction to Physiological Measurements - Cardiovascular
 Lab 4: Introduction to Physiological Measurements - Cardiovascular INTRODUCTION: This lab will demonstrate cardiovascular measurements by creating an ECG with instruments used in previous labs. Students
Lab 4: Introduction to Physiological Measurements - Cardiovascular INTRODUCTION: This lab will demonstrate cardiovascular measurements by creating an ECG with instruments used in previous labs. Students
Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG
 Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Manual Revision PL3.7.0 080706 Richard Pflanzer, Ph.D.
Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Manual Revision PL3.7.0 080706 Richard Pflanzer, Ph.D.
Anatomy & Physiology Muscular System Worksheet
 Anatomy & Physiology Muscular System Worksheet 1. What are the three categories of muscle tissue? a) b) c) 2. The smallest functional unit of a muscle fiber is called a. 3. What are the four characteristics
Anatomy & Physiology Muscular System Worksheet 1. What are the three categories of muscle tissue? a) b) c) 2. The smallest functional unit of a muscle fiber is called a. 3. What are the four characteristics
Nerve regeneration. Somatic nervous system
 Somatic nervous system Signals from CNS are sent to skeletal muscles. Final result is a muscle contraction. Motor neuron starts in CNS and its axon ends at a muscle cell. Alpha motor neuron Alpha motor
Somatic nervous system Signals from CNS are sent to skeletal muscles. Final result is a muscle contraction. Motor neuron starts in CNS and its axon ends at a muscle cell. Alpha motor neuron Alpha motor
Nerve meets muscle. Nerve regeneration. Somatic nervous system
 Somatic nervous system Signals from CNS are sent to skeletal muscles. Final result is a muscle contraction. Alpha motor neurons branch into several terminals (can be over 1000), each contacting a separate
Somatic nervous system Signals from CNS are sent to skeletal muscles. Final result is a muscle contraction. Alpha motor neurons branch into several terminals (can be over 1000), each contacting a separate
Overview Active Learning
 : EMG Active Learning Updated 01-06-11 This lesson is based on a presentation given by Dr. Jennifer Lundmark and Ms. Andrea Salmi at the HAPS Conference, Maui, HI, June 2-7, 2001 Overview Active Learning
: EMG Active Learning Updated 01-06-11 This lesson is based on a presentation given by Dr. Jennifer Lundmark and Ms. Andrea Salmi at the HAPS Conference, Maui, HI, June 2-7, 2001 Overview Active Learning
iworx Sample Lab Experiment HC-3: Pulse Wave Velocity
 Experiment HC-3: Pulse Wave Velocity Exercise 1: The Pulse Wave Velocity at Rest Aim: To measure the pulse wave velocity in a resting individual. Procedure 1. Click on the Record button, located on the
Experiment HC-3: Pulse Wave Velocity Exercise 1: The Pulse Wave Velocity at Rest Aim: To measure the pulse wave velocity in a resting individual. Procedure 1. Click on the Record button, located on the
Concept 50.5: The physical interaction of protein filaments is required for muscle function
 Concept 50.5: The physical interaction of protein filaments is required for muscle function Muscle activity is a response to input from the nervous system The action of a muscle is always to contract Vertebrate
Concept 50.5: The physical interaction of protein filaments is required for muscle function Muscle activity is a response to input from the nervous system The action of a muscle is always to contract Vertebrate
Chapter 10! Chapter 10, Part 2 Muscle. Muscle Tissue - Part 2! Pages !
 ! Chapter 10, Part 2 Muscle Chapter 10! Muscle Tissue - Part 2! Pages 308-324! SECTION 10-5! Sarcomere shortening and muscle fiber stimulation produce tension! 2! Tension Production - Muscle FIBER! All-or-none
! Chapter 10, Part 2 Muscle Chapter 10! Muscle Tissue - Part 2! Pages 308-324! SECTION 10-5! Sarcomere shortening and muscle fiber stimulation produce tension! 2! Tension Production - Muscle FIBER! All-or-none
Muscle Cells & Muscle Fiber Contractions. Packet #8
 Muscle Cells & Muscle Fiber Contractions Packet #8 Skeletal muscle is attached to bones and is responsible for movement. Introduction Introduction II Skeletal muscle is composed of bundles of muscle fibers
Muscle Cells & Muscle Fiber Contractions Packet #8 Skeletal muscle is attached to bones and is responsible for movement. Introduction Introduction II Skeletal muscle is composed of bundles of muscle fibers
iworx Sample Lab Experiment HH-4: The Six-Lead Electrocardiogram
 Experiment HH-4: The Six-Lead Electrocardiogram Background The cardiac cycle involves a sequential contraction of the atria and the ventricles. These contractions are triggered by the coordinated electrical
Experiment HH-4: The Six-Lead Electrocardiogram Background The cardiac cycle involves a sequential contraction of the atria and the ventricles. These contractions are triggered by the coordinated electrical
Anatomy and Physiology 1 Chapter 10 self quiz Pro, Dima Darwish,MD.
 Anatomy and Physiology 1 Chapter 10 self quiz Pro, Dima Darwish,MD. 1) Which of the following is a recognized function of skeletal muscle? A) produce movement B) maintain posture C) maintain body temperature
Anatomy and Physiology 1 Chapter 10 self quiz Pro, Dima Darwish,MD. 1) Which of the following is a recognized function of skeletal muscle? A) produce movement B) maintain posture C) maintain body temperature
Ch 12: Muscles sarcolemma, t-tubules, sarcoplasmic reticulum, myofibrils, myofilaments, sarcomere...
 Ch 12: Muscles Review micro-anatomy of muscle tissue Terminology examples: sarcolemma, t-tubules, sarcoplasmic reticulum, myofibrils, myofilaments, sarcomere... SLOs Differentiate levels of muscle structure:
Ch 12: Muscles Review micro-anatomy of muscle tissue Terminology examples: sarcolemma, t-tubules, sarcoplasmic reticulum, myofibrils, myofilaments, sarcomere... SLOs Differentiate levels of muscle structure:
Lab #4: Physiology of the in situ Amphibian Heart
 Lab #4: Physiology of the in situ Amphibian Heart This experiment explores the basic principles of cardiac muscle physiology, including contraction force, ECG, the effect of temperature, and the effect
Lab #4: Physiology of the in situ Amphibian Heart This experiment explores the basic principles of cardiac muscle physiology, including contraction force, ECG, the effect of temperature, and the effect
Investigation of human cardiovascular physiology is very interesting, but many
 6 E X E R C I S E Frog Cardiovascular Physiology O B J E C T I V E S 1. To list the properties of cardiac muscle as automaticity and rhythmicity, and to define each. 2. To explain the statement, Cardiac
6 E X E R C I S E Frog Cardiovascular Physiology O B J E C T I V E S 1. To list the properties of cardiac muscle as automaticity and rhythmicity, and to define each. 2. To explain the statement, Cardiac
CLASS SET Unit 4: The Muscular System STUDY GUIDE
 NPHS Anatomy & Physiology Questions to answer: 1) List three functions of the muscular system. 1) movement 2) thermogenesis (generates heat) 3) posture & body/joint support CLASS SET Unit 4: The Muscular
NPHS Anatomy & Physiology Questions to answer: 1) List three functions of the muscular system. 1) movement 2) thermogenesis (generates heat) 3) posture & body/joint support CLASS SET Unit 4: The Muscular
Skeletal Muscle Tissue
 Functions of Skeletal Muscle Skeletal Muscle Tissue Keri Muma Bio 6 Movement muscles attach directly or indirectly to bone, pull on bone or tissue when they contract Maintain posture / body position muscles
Functions of Skeletal Muscle Skeletal Muscle Tissue Keri Muma Bio 6 Movement muscles attach directly or indirectly to bone, pull on bone or tissue when they contract Maintain posture / body position muscles
Muscular Tissue. Functions of Muscular Tissue. Types of Muscular Tissue. Skeletal Muscular Tissue. Properties of Muscular Tissue
 Muscular Tissue Functions of Muscular Tissue Muscle makes up a large percentage of the body s weight (40-50%) Their main functions are to: Create motion muscles work with nerves, bones, and joints to produce
Muscular Tissue Functions of Muscular Tissue Muscle makes up a large percentage of the body s weight (40-50%) Their main functions are to: Create motion muscles work with nerves, bones, and joints to produce
Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand.
 Experiment HN-4: Hand vs. Foot Reactions Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand. Procedure 1. Instruct the subject
Experiment HN-4: Hand vs. Foot Reactions Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand. Procedure 1. Instruct the subject
Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease
 Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease Lab #10 Table of Contents: Expected Learning Outcomes...... 83 Introduction....... 84 Activity 1: Collecting ECG Data..... 85 Activity
Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease Lab #10 Table of Contents: Expected Learning Outcomes...... 83 Introduction....... 84 Activity 1: Collecting ECG Data..... 85 Activity
Muscle Dr. Ted Milner (KIN 416)
 Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 10 Muscular Tissue Introduction The purpose of the chapter is to: 1. Learn about the structure and function of the 3 types of muscular tissue
Principles of Anatomy and Physiology 14 th Edition CHAPTER 10 Muscular Tissue Introduction The purpose of the chapter is to: 1. Learn about the structure and function of the 3 types of muscular tissue
Nerve Cell (aka neuron)
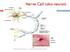 Nerve Cell (aka neuron) Neuromuscular Junction Nerve cell Muscle fiber (cell) The Nerve Stimulus and Action Potential The Nerve Stimulus and Action Potential Skeletal muscles must be stimulated by a motor
Nerve Cell (aka neuron) Neuromuscular Junction Nerve cell Muscle fiber (cell) The Nerve Stimulus and Action Potential The Nerve Stimulus and Action Potential Skeletal muscles must be stimulated by a motor
MUSCULAR TISSUE. Dr. Gary Mumaugh
 MUSCULAR TISSUE Dr. Gary Mumaugh MUSCLE OVERVIEW The three types of muscle tissue are skeletal, cardiac, and smooth These types differ in structure, location, function, and means of activation FUNCTIONAL
MUSCULAR TISSUE Dr. Gary Mumaugh MUSCLE OVERVIEW The three types of muscle tissue are skeletal, cardiac, and smooth These types differ in structure, location, function, and means of activation FUNCTIONAL
Muscle Physiology. Introduction. Four Characteristics of Muscle tissue. Skeletal Muscle
 Muscle Physiology Introduction Muscle = tissue capable of forceful shortening or contraction Converts chemical energy (ATP) into mechanical energy Important in: Respiration Urine collection & flow Gastrointestinal
Muscle Physiology Introduction Muscle = tissue capable of forceful shortening or contraction Converts chemical energy (ATP) into mechanical energy Important in: Respiration Urine collection & flow Gastrointestinal
Chapter 10 Muscle Tissue Lecture Outline
 Chapter 10 Muscle Tissue Lecture Outline Muscle tissue types 1. Skeletal muscle = voluntary striated 2. Cardiac muscle = involuntary striated 3. Smooth muscle = involuntary nonstriated Characteristics
Chapter 10 Muscle Tissue Lecture Outline Muscle tissue types 1. Skeletal muscle = voluntary striated 2. Cardiac muscle = involuntary striated 3. Smooth muscle = involuntary nonstriated Characteristics
Effects of Temperature, Stretch, and Various Drug Treatments on the
 Nicole Rodi Bio 235: Animal Physiology Heart Muscle Lab Report 10/24/2014 Effects of Temperature, Stretch, and Various Drug Treatments on the Cardiac Muscle Activity of Rana pipiens Abstract Mechanical
Nicole Rodi Bio 235: Animal Physiology Heart Muscle Lab Report 10/24/2014 Effects of Temperature, Stretch, and Various Drug Treatments on the Cardiac Muscle Activity of Rana pipiens Abstract Mechanical
Cardiac muscle is different from other types of muscle in that cardiac muscle
 6 E X E R C I S E Cardiovascular Physiology O B J E C T I V E S 1. To define autorhythmicity, sinoatrial node, pacemaker cells, and vagus nerves 2. To understand the effects of the sympathetic and parasympathetic
6 E X E R C I S E Cardiovascular Physiology O B J E C T I V E S 1. To define autorhythmicity, sinoatrial node, pacemaker cells, and vagus nerves 2. To understand the effects of the sympathetic and parasympathetic
Muscle Tissue. Muscle Tissue Outline. General Function of Muscle Tissue
 Muscle Tissue Muscle Tissue Outline General Functions of Muscle Tissue Characteristics of Muscle Tissue Classification of Muscle Tissue Skeletal Muscle Structure and Function Muscle Energetics Muscle Mechanics
Muscle Tissue Muscle Tissue Outline General Functions of Muscle Tissue Characteristics of Muscle Tissue Classification of Muscle Tissue Skeletal Muscle Structure and Function Muscle Energetics Muscle Mechanics
On which skeletal muscle filament is troponin located? What is the function of the sarcoplasmic reticulum (SR)?
 CASE 6 A 21-year-old man presents to a rural emergency center with a 1-day history of progressive stiffness of the neck and jaw, difficulty swallowing, stiff shoulders and back, and a rigid abdomen. Upon
CASE 6 A 21-year-old man presents to a rural emergency center with a 1-day history of progressive stiffness of the neck and jaw, difficulty swallowing, stiff shoulders and back, and a rigid abdomen. Upon
Microanatomy of Muscles. Anatomy & Physiology Class
 Microanatomy of Muscles Anatomy & Physiology Class Three Main Muscle Types Objectives: By the end of this presentation you will have the information to: 1. 2. 3. 4. 5. 6. Describe the 3 main types of muscles.
Microanatomy of Muscles Anatomy & Physiology Class Three Main Muscle Types Objectives: By the end of this presentation you will have the information to: 1. 2. 3. 4. 5. 6. Describe the 3 main types of muscles.
Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion
 Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion Exercise 1: Antagonistic Muscles in Forearm Aim: To study the EMG activity in muscles that work in opposition
Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion Exercise 1: Antagonistic Muscles in Forearm Aim: To study the EMG activity in muscles that work in opposition
How Do We Sense, Think, and Move? -- Lab #11 Bioelectronics Measuring Electrical Properties of the Body
 How Do We Sense, Think, and Move? -- Lab #11 Bioelectronics Measuring Electrical Properties of the Body Experiment #1 Your Body's Resistance Equipment: Digital multimeter, Banana leads Important Equipment
How Do We Sense, Think, and Move? -- Lab #11 Bioelectronics Measuring Electrical Properties of the Body Experiment #1 Your Body's Resistance Equipment: Digital multimeter, Banana leads Important Equipment
Experiment HH-2: The Electrocardiogram and Heart Sounds
 Experiment HH-2: The Electrocardiogram and Heart Sounds Exercise 1: The ECG in a Resting Subject Aim: To measure the ECG in a resting individual. Procedure 1. Click on the Record button, located on the
Experiment HH-2: The Electrocardiogram and Heart Sounds Exercise 1: The ECG in a Resting Subject Aim: To measure the ECG in a resting individual. Procedure 1. Click on the Record button, located on the
Chapter 10: Muscles. Vocabulary: aponeurosis, fatigue
 Chapter 10: Muscles 37. Describe the structural components of skeletal muscle tissue from the molecular to the organ level. 38. Describe the structure, function, and importance of sarcomeres. 39. Identify
Chapter 10: Muscles 37. Describe the structural components of skeletal muscle tissue from the molecular to the organ level. 38. Describe the structure, function, and importance of sarcomeres. 39. Identify
Muscles and Muscle Tissue
 1 Muscles and Muscle Tissue Chapter 9 2 Overview of Muscle Tissues Compare and Contrast the three basic types of muscle tissue List four important functions of muscle tissue 3 Muscle Terminology Muscle
1 Muscles and Muscle Tissue Chapter 9 2 Overview of Muscle Tissues Compare and Contrast the three basic types of muscle tissue List four important functions of muscle tissue 3 Muscle Terminology Muscle
Muscle Cell Anatomy & Function (mainly striated muscle tissue)
 Muscle Cell Anatomy & Function (mainly striated muscle tissue) General Structure of Muscle Cells (skeletal) several nuclei (skeletal muscle) skeletal muscles are formed when embryonic cells fuse together
Muscle Cell Anatomy & Function (mainly striated muscle tissue) General Structure of Muscle Cells (skeletal) several nuclei (skeletal muscle) skeletal muscles are formed when embryonic cells fuse together
Dissection and Contraction of Frog Heart
 Dissection and Contraction of Frog Heart Rob MacLeod and Alex Brownell (aabrownell@yahoo.com) March 4, 2006 1 Purpose and Background 1.1 Purpose: To examine the anatomy and basic contractile physiology
Dissection and Contraction of Frog Heart Rob MacLeod and Alex Brownell (aabrownell@yahoo.com) March 4, 2006 1 Purpose and Background 1.1 Purpose: To examine the anatomy and basic contractile physiology
Muscle Physiology. Bio 219 Dr. Adam Ross Napa Valley College
 Muscle Physiology Bio 219 Dr. Adam Ross Napa Valley College Muscle tissue Muscle is an excitable tissue capable of force production Three types Skeletal- striated, voluntary Cardiac- non-striated, involuntary
Muscle Physiology Bio 219 Dr. Adam Ross Napa Valley College Muscle tissue Muscle is an excitable tissue capable of force production Three types Skeletal- striated, voluntary Cardiac- non-striated, involuntary
Skeletal Muscle Contraction 5/11/2017 Dr. Hiwa Shafiq
 Skeletal Muscle Contraction 5/11/2017 Dr. Hiwa Shafiq Skeletal Muscle Fiber About 40 per cent of the body is skeletal muscle, and 10 per cent is smooth and cardiac muscle. Skeletal muscles are composed
Skeletal Muscle Contraction 5/11/2017 Dr. Hiwa Shafiq Skeletal Muscle Fiber About 40 per cent of the body is skeletal muscle, and 10 per cent is smooth and cardiac muscle. Skeletal muscles are composed
HASPI Medical Anatomy & Physiology 09b Lab Activity
 HASPI Medical Anatomy & Physiology 09b Lab Activity Name(s): Period: Date: Muscle Cell Structure Muscle cells are specialized to contract. An individual muscle is actually a bundle of hundreds to thousands
HASPI Medical Anatomy & Physiology 09b Lab Activity Name(s): Period: Date: Muscle Cell Structure Muscle cells are specialized to contract. An individual muscle is actually a bundle of hundreds to thousands
Lecture Overview. Muscular System. Marieb s Human Anatomy and Physiology. Chapter 9 Muscles and Muscle Tissue Lecture 16
 Marieb s Human Anatomy and Physiology Marieb Hoehn Chapter 9 Muscles and Muscle Tissue Lecture 16 1 Lecture Overview Types, characteristics, functions of muscle Structure of skeletal muscle Mechanism of
Marieb s Human Anatomy and Physiology Marieb Hoehn Chapter 9 Muscles and Muscle Tissue Lecture 16 1 Lecture Overview Types, characteristics, functions of muscle Structure of skeletal muscle Mechanism of
Chapter 10! Muscle Tissue - Part 2! Pages ! SECTION 10-5! Sarcomere shortening and muscle fiber stimulation produce tension!
 ! Chapter 10, Part 2 Muscle Chapter 10! Muscle Tissue - Part 2! Pages 308-324! SECTION 10-5! Sarcomere shortening and muscle fiber stimulation produce tension! 2! 1 Tension Production - MUSCLE FIBER! All-or-none
! Chapter 10, Part 2 Muscle Chapter 10! Muscle Tissue - Part 2! Pages 308-324! SECTION 10-5! Sarcomere shortening and muscle fiber stimulation produce tension! 2! 1 Tension Production - MUSCLE FIBER! All-or-none
MUSCULAR SYSTEM CHAPTER 09 BIO 211: ANATOMY & PHYSIOLOGY I
 1 BIO 211: ANATOMY & PHYSIOLOGY I 1 CHAPTER 09 MUSCULAR SYSTEM Part 2 of 2 Dr. Dr. Lawrence G. G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill. Some illustrations are courtesy
1 BIO 211: ANATOMY & PHYSIOLOGY I 1 CHAPTER 09 MUSCULAR SYSTEM Part 2 of 2 Dr. Dr. Lawrence G. G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill. Some illustrations are courtesy
Exercise 2: Skeletal Muscle Physiology: Activity 3: The Effect of Stimulus Frequency on Skeletal Muscle Contraction Lab Report
 Name: Courtney.Sims Exercise 2: Skeletal Muscle Physiology: Activity 3: The Effect of Stimulus Frequency on Skeletal Muscle Contraction Lab Report Pre-lab Quiz Results You scored 100% by answering 4 out
Name: Courtney.Sims Exercise 2: Skeletal Muscle Physiology: Activity 3: The Effect of Stimulus Frequency on Skeletal Muscle Contraction Lab Report Pre-lab Quiz Results You scored 100% by answering 4 out
Neurobiology: The nerve cell. Principle and task To use a nerve function model to study the following aspects of a nerve cell:
 Principle and task To use a nerve function model to study the following aspects of a nerve cell: INTRACELLULAR POTENTIAL AND ACTION POTENTIAL Comparison between low and high threshold levels Comparison
Principle and task To use a nerve function model to study the following aspects of a nerve cell: INTRACELLULAR POTENTIAL AND ACTION POTENTIAL Comparison between low and high threshold levels Comparison
The nervous system is responsible for most of the functions that characterize
 3 E X E R C I S E Neurophysiology of Nerve Impulses O B J E C T I V E S 1. To define the following: irritability, conductivity, resting membrane potential, polarized, sodium-potassium pump, threshold stimulus,
3 E X E R C I S E Neurophysiology of Nerve Impulses O B J E C T I V E S 1. To define the following: irritability, conductivity, resting membrane potential, polarized, sodium-potassium pump, threshold stimulus,
The Biomechanics of Human Skeletal Muscle
 AML2506 Biomechanics and Flow Simulation Day 03B The Biomechanics of Human Skeletal Muscle Session Speaker Dr. M. D. Deshpande 1 Session Objectives At the end of this session the delegate would have understood
AML2506 Biomechanics and Flow Simulation Day 03B The Biomechanics of Human Skeletal Muscle Session Speaker Dr. M. D. Deshpande 1 Session Objectives At the end of this session the delegate would have understood
Muscle and Neuromuscular Junction. Peter Takizawa Department of Cell Biology
 Muscle and Neuromuscular Junction Peter Takizawa Department of Cell Biology Types and structure of muscle cells Structural basis of contraction Triggering muscle contraction Skeletal muscle consists of
Muscle and Neuromuscular Junction Peter Takizawa Department of Cell Biology Types and structure of muscle cells Structural basis of contraction Triggering muscle contraction Skeletal muscle consists of
Chapter 8: Skeletal Muscle: Structure and Function
 Chapter 8: Skeletal Muscle: Structure and Function Objectives Draw & label the microstructure of skeletal muscle Outline the steps leading to muscle shortening Define the concentric and isometric Discuss:
Chapter 8: Skeletal Muscle: Structure and Function Objectives Draw & label the microstructure of skeletal muscle Outline the steps leading to muscle shortening Define the concentric and isometric Discuss:
