Serious Adverse Event
|
|
|
- Victoria Fowler
- 5 years ago
- Views:
Transcription
1 Adverse Event Non- Adverse Event Expected Unexpected Unexpected Non- Serious Serious Serious Harm or Increased Risk of Harm Unrelated Possibly Related or Related Possibly Related or Related Unrelated Possibly Related or Related Non-Serious Adverse Event Serious Adverse Event Unanticipated Problem
2 Unanticipated Problem (UP): Internal or External SAE: Internal or External, Related or Possibly Related, Unexpected SAE: Internal, all other situations SAE: External, requiring change in protocol or ICFs but NOT considered an UP SAE: External, all other situations Non-Serious AE: Internal, all situations not considered an Unanticipated Problem Non-Serious AE: External, all situations not considered an Unanticipated Problem Immediate reporting; completed SAE and Unanticipated Problem Reporting Form with supporting documents submitted to IRB within 5 business days Meets criteria for an Unanticipated Problem and is to be reported as such Complete Event Reporting Form and submit to IRB within 15 business days Complete SAE and Unanticipated Problem Reporting Form and submit to IRB within 15 business days Submit a summary using the External SAE Summary Reporting Form for all interval events 1) at the time of study Continuing Review, or 2) at the time of study termination (if before CR), or 3) as required by the IRB approved study protocol Clinically significant AEs may be summarized at the time of Continuing Review, or study termination if before the next scheduled Continuing Review Not required to be reported to the IRB
3 An investigator conducting behavioral research collects individually identifiable sensitive information about illicit drug use and other illegal behaviors by surveying college students. The data are stored on a laptop computer without encryption, and the laptop computer is stolen from the investigator s car on the way home from work. Non-Adverse Event (no harm done yet) Unanticipated Problem: unexpected (nature), related to study participation, results in increased risk for harm than previously recognized Psychological and social harm from the breach in confidentiality
4 Subjects with coronary artery disease presenting with unstable angina are enrolled in a multicenter clinical trial evaluating the safety and efficacy of an investigational vascular stent. Based on prior studies in animals and humans, the investigators anticipate that up to 5% of subjects receiving the investigational stent will require emergency coronary artery bypass graft (CABG) surgery because of acute blockage of the stent. The risk of needing emergency CABG surgery is described in the IRB-approved protocol and ICF. After the first 20 subjects are enrolled in the study, a DSMB conducts an interim analysis, as required by the IRB-approved protocol, and notes that 10 subjects have needed to undergo emergency CABG surgery soon after placement of the investigational stent. Serious Adverse Events Unanticipated Problem: unexpected (frequency), related to study participation, serious
5 As a result of a processing error by a pharmacy technician, a subject enrolled in a multicenter clinical trial receives a dose of an experimental agent that is 10- times higher than the dose dictated by the IRBapproved protocol. Fortunately, the subject does not experience any side effects, and no detectable adverse physical or laboratory abnormalities are identified. Non-Adverse Event Unanticipated Problem: unexpected (nature), related to study participation, results in increased risk for harm than previously recognized Side-effects of experimental agent
6 A subject participating in a phase III, randomized, doubleblind, controlled clinical trial comparing the relative safety and efficacy of a new chemotherapy agent versus placebo added to standard chemotherapy treatment for multiple myeloma develops neutropenia and sepsis. The subject subsequently develops multiorgan failure and dies. Prolonged bone marrow suppression resulting in neutropenia and risk of life-threatening infections are known complications of the chemotherapy regimens being tested in this clinical trial and these risks are described in the IRB-approved protocol and ICFs. Serious Adverse Event Not Unanticipated Problem: expected, related to study participation, serious
7 A subject with chronic gastroesophageal reflux disease enrolls in a randomized, placebo-controlled, double-blind, phase III clinical trial evaluating an investigational agent that blocks acid release in the stomach. Two weeks after being randomized and started on the study intervention the subject develops acute kidney failure as evidenced by an increase in serum creatinine from 1.0 mg/dl pre-randomization to 5.0 mg/dl. The known risk profile of the investigational agent does not include renal toxicity, and the IRB-approved protocol and ICF for the study do not identify kidney damage as a risk of the research. Evaluation of the subject reveals no other obvious cause for acute renal failure. Serious Adverse Event Unanticipated Problem: unexpected (nature), related to study participation, serious
8 An investigator is conducting a psychology study evaluating the factors that affect reaction times in response to auditory stimuli. In order to perform the reaction time measurements, subjects are placed in a small, windowless soundproof booth and asked to wear headphones. The IRB-approved protocol and ICF describe claustrophobic reactions as one of the risks of the research. The 20 th subject enrolled in the research experiences significant claustrophobia, resulting in the subject withdrawing from the research. Adverse Event Not Unanticipated Problem: expected, related to study participation, serious
9 An investigator performs prospective medical chart reviews to collect medical data on premature infants in a neonatal intensive care unit (NICU) for a research registry. An infant, whose medical data are being collected for the registry, dies as the result of an infection that commonly occurs in the NICU setting. Serious Adverse Event Not Unanticipated Problem: expected, not related to study participation, serious
10 Subjects with cancer are enrolled in a phase II clinical trial evaluating an investigational biologic product derived from human sera. After several subjects are enrolled and receive the investigational product, a study audit reveals that the investigational product administered to subjects was obtained from donors who were not appropriately screened and tested for several potential viral contaminants, including the human immunodeficiency virus and the hepatitis B virus. Non-Adverse Event (no harm done yet) Unanticipated Problem: unexpected (nature), related to study participation, results in increased risk for harm than previously recognized Infection risk
11 A subject with seizures enrolls in a randomized, phase III clinical trial comparing an investigational anti-seizure agent to a standard, FDA-approved anti-seizure medication. The subject is randomized to the group receiving the investigational agent. One month after enrollment, the subject is hospitalized with severe fatigue and on further evaluation is noted to have severe anemia (hematocrit decreased from 45% pre-randomization to 20%). Further hematologic evaluation suggests an immune-mediated hemolytic anemia. The known risk profile of the investigational agent does not include anemia, and the IRB-approved protocol and ICF for the study do not identify anemia as a risk of the research. Serious Adverse Event Unanticipated Problem: unexpected (nature), related to study participation, serious
12 The 5 th subject enrolled in a phase II, open-label, uncontrolled clinical study evaluating the safety and efficacy of an investigational oral agent administered daily for treatment of severe psoriasis unresponsive to FDA-approved treatments, develops severe hepatic failure complicated by encephalopathy one month after starting the oral agent. The known risk profile of the investigational oral agent prior to this event included mild elevation of serum liver enzymes in 10% of subjects receiving the agent during previous clinical studies; but, there was no other history of subjects developing clinically significant liver disease. The IRB-approved protocol and ICF for the study identifies mild liver injury as a risk of the research. The investigators identify no other etiology for the liver failure. Serious Adverse Event Unanticipated Problem: unexpected (severity), related to study participation, serious
13 A subject with advanced renal cell carcinoma is enrolled in a study evaluating the effects of hypnosis for the management of chronic pain in cancer patients. During the subject s initial hypnosis session in the pain clinic, the subject suddenly develops acute chest pain and shortness of breath, followed by loss of consciousness. The subject suffers a cardiac arrest and dies. An autopsy reveals that the patient died from a massive pulmonary embolus, related to the underlying renal cell carcinoma. Serious Adverse Event Not Unanticipated Problem: unexpected (for the study), not related to study participation, serious
Unanticipated Problems and Adverse Events
 Unanticipated Problems and Adverse Events David Borasky, MPH, CIP Office of Research Protections RTI International Research Triangle Park, North Carolina, USA Introduction IEC review of unanticipated problems
Unanticipated Problems and Adverse Events David Borasky, MPH, CIP Office of Research Protections RTI International Research Triangle Park, North Carolina, USA Introduction IEC review of unanticipated problems
SYNOPSIS CLINICAL STUDY REPORT
 SYNOPSIS CLINICAL STUDY REPORT Non-interventional observational study on efficacy, safety and practicability of MIRCERA in kidney transplanted patients (BEAT) Report on non-interventional study ML21386
SYNOPSIS CLINICAL STUDY REPORT Non-interventional observational study on efficacy, safety and practicability of MIRCERA in kidney transplanted patients (BEAT) Report on non-interventional study ML21386
ClinicalTrials.gov Identifier: NCT Sponsor/company: Sanofi-Aventis. Date: 08/02/ 2008
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Sanofi-Aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Sanofi-Aventis ClinicalTrials.gov
GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS
 CHILDREN S HOSPITALS AND CLINICS OF MINNESOTA Introduction: GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS These guidelines have been developed in conjunction with the hospital Transfusion Committee.
CHILDREN S HOSPITALS AND CLINICS OF MINNESOTA Introduction: GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS These guidelines have been developed in conjunction with the hospital Transfusion Committee.
Adverse Experience Reporting
 Managing and reporting adverse events Key learning objectives By the end of this session, you will be able to: Define and classify an adverse event; Differentiate between serious and severe adverse events;
Managing and reporting adverse events Key learning objectives By the end of this session, you will be able to: Define and classify an adverse event; Differentiate between serious and severe adverse events;
Measure #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care
 Measure #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
Measure #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE
GSK Medicine: Study Number: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objective:
 GSK Medicine: abacavir (ABC)/dolutegravir (DTG)/lamivudine (3TC) Study Number: 201147 Title: A IIIb, randomized, open-label study of the safety, efficacy, and tolerability of switching to a fixed-dose
GSK Medicine: abacavir (ABC)/dolutegravir (DTG)/lamivudine (3TC) Study Number: 201147 Title: A IIIb, randomized, open-label study of the safety, efficacy, and tolerability of switching to a fixed-dose
Supplementary material 1. Definitions of study endpoints (extracted from the Endpoint Validation Committee Charter) 1.
 Rationale, design, and baseline characteristics of the SIGNIFY trial: a randomized, double-blind, placebo-controlled trial of ivabradine in patients with stable coronary artery disease without clinical
Rationale, design, and baseline characteristics of the SIGNIFY trial: a randomized, double-blind, placebo-controlled trial of ivabradine in patients with stable coronary artery disease without clinical
Epogen / Procrit. Epogen / Procrit (epoetin alfa) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.06 Section: Prescription Drugs Effective Date: April1, 2014 Subject: Epogen / Procrit Page: 1 of 7
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.06 Section: Prescription Drugs Effective Date: April1, 2014 Subject: Epogen / Procrit Page: 1 of 7
PAGE 1 NEURO-OPHTHALMIC QUESTIONNAIRE NAME: AGE: DATE OF EXAM: CHART #: (Office Use Only)
 PAGE 1 NEURO-OPHTHALMIC QUESTIONNAIRE NAME: AGE: DATE OF EXAM: CHART #: (Office Use Only) 1. What is the main problem that you are having? (If additional space is required, please use the back of this
PAGE 1 NEURO-OPHTHALMIC QUESTIONNAIRE NAME: AGE: DATE OF EXAM: CHART #: (Office Use Only) 1. What is the main problem that you are having? (If additional space is required, please use the back of this
This was a randomized, double-blind, placebo-controlled, fixed-dose, parallel-group study.
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Full Novartis CTRD Results Template
 Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23138E1 Title A
Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23138E1 Title A
Safety of Treatment in Cirrhotics in the Era of New Antiviral Therapies for Hepatitis C Virus
 Safety of Treatment in Cirrhotics in the Era of New Antiviral Therapies for Hepatitis C Virus JEFFREY NADELSON MD, ALAN EPSTEIN MD, THOMAS SEPE MD BOSTON UNIVERSITY SCHOOL OF MEDICINE ROGER WILLIAMS MEDICAL
Safety of Treatment in Cirrhotics in the Era of New Antiviral Therapies for Hepatitis C Virus JEFFREY NADELSON MD, ALAN EPSTEIN MD, THOMAS SEPE MD BOSTON UNIVERSITY SCHOOL OF MEDICINE ROGER WILLIAMS MEDICAL
Clinical Trial Synopsis TL-OPI-516, NCT#
 Clinical Trial Synopsis, NCT#00225277 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl Versus
Clinical Trial Synopsis, NCT#00225277 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl Versus
Link between effectiveness and cost data Costing was conducted prospectively on the same patient sample as that used in the effectiveness analysis.
 Heparin after percutaneous intervention (HAPI): a prospective multicenter randomized trial of three heparin regimens after successful coronary intervention Rabah M, Mason D, Muller D W, Hundley R, Kugelmass
Heparin after percutaneous intervention (HAPI): a prospective multicenter randomized trial of three heparin regimens after successful coronary intervention Rabah M, Mason D, Muller D W, Hundley R, Kugelmass
Safety Assessment in Clinical Trials and Beyond
 Safety Assessment in Clinical Trials and Beyond Yuliya Yasinskaya, MD Medical Team Leader Division of Anti-Infective Products Center for Drug Evaluation and Research FDA Clinical Investigator Training
Safety Assessment in Clinical Trials and Beyond Yuliya Yasinskaya, MD Medical Team Leader Division of Anti-Infective Products Center for Drug Evaluation and Research FDA Clinical Investigator Training
Primary Endpoint The primary endpoint is overall survival, measured as the time in weeks from randomization to date of death due to any cause.
 CASE STUDY Randomized, Double-Blind, Phase III Trial of NES-822 plus AMO-1002 vs. AMO-1002 alone as first-line therapy in patients with advanced pancreatic cancer This is a multicenter, randomized Phase
CASE STUDY Randomized, Double-Blind, Phase III Trial of NES-822 plus AMO-1002 vs. AMO-1002 alone as first-line therapy in patients with advanced pancreatic cancer This is a multicenter, randomized Phase
Forward-looking Statements
 Forward-looking Statements This presentation contains forward-looking statements. All statements other than statements of historical facts contained in this presentation, including statements regarding
Forward-looking Statements This presentation contains forward-looking statements. All statements other than statements of historical facts contained in this presentation, including statements regarding
Guidance on Unanticipated Problems (UPs) and Adverse Events (AEs) Bertha delanda IRB Training Specialist Research Compliance Office March 2010
 Guidance on Unanticipated Problems (UPs) and Adverse Events (AEs) Bertha delanda IRB Training Specialist Research Compliance Office March 2010 OUTLINE - AE (adverse event) - SAE (serious adverse event)
Guidance on Unanticipated Problems (UPs) and Adverse Events (AEs) Bertha delanda IRB Training Specialist Research Compliance Office March 2010 OUTLINE - AE (adverse event) - SAE (serious adverse event)
Clinical Trial Results Summary Study EN3409-BUP-305
 Title of Study: A 52-Week, Open-Label, Long-Term Treatment Evaluation of the Safety and Efficacy of BEMA Buprenorphine in Subjects with Moderate to Severe Chronic Pain Coordinating Investigator: Martin
Title of Study: A 52-Week, Open-Label, Long-Term Treatment Evaluation of the Safety and Efficacy of BEMA Buprenorphine in Subjects with Moderate to Severe Chronic Pain Coordinating Investigator: Martin
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Plattenepithelkarzinom des Ösophagus, 1 st -line
 Plattenepithelkarzinom des Ösophagus, 1 st -line AIO-STO-0309 An open-label, randomized phase III trial of cisplatin and 5-fluorouracil with or without panitumumab for patients with nonresectable, advanced
Plattenepithelkarzinom des Ösophagus, 1 st -line AIO-STO-0309 An open-label, randomized phase III trial of cisplatin and 5-fluorouracil with or without panitumumab for patients with nonresectable, advanced
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Quote Request. Advisor Information. Client Information. Medical History. Driving History. Advisor Company Date. Phone Fax.
 Advisor Information Advisor Company Date E-Mail Phone Fax Client Information Client Name Date of Birth Occupation Plan and amount of insurance requested: Has the case been submitted to other companies
Advisor Information Advisor Company Date E-Mail Phone Fax Client Information Client Name Date of Birth Occupation Plan and amount of insurance requested: Has the case been submitted to other companies
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Keeping track of your numbers
 Keeping track of your numbers If you have relapsed or refractory multiple myeloma, keeping track of your numbers can help you take an active role in your care. It s also one way you and your doctor can
Keeping track of your numbers If you have relapsed or refractory multiple myeloma, keeping track of your numbers can help you take an active role in your care. It s also one way you and your doctor can
Adverse Events- Love them, hate them, report them!
 Adverse Events- Love them, hate them, report them! Billi Tatum, RN, CCRC Regional Vascular Center Harborview Medical Center and the University of Washington Medical Center Objectives Define and identify
Adverse Events- Love them, hate them, report them! Billi Tatum, RN, CCRC Regional Vascular Center Harborview Medical Center and the University of Washington Medical Center Objectives Define and identify
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
WASHINGTON UNIVERSITY SCHOOL OF MEDICINE. Cranial Health History Form
 WASHINGTON UNIVERSITY SCHOOL OF MEDICINE Cranial Health History Form Welcome to the Neurosurgery Department at Washington University. To help us treat you, please fill this form out completely. Your Name:
WASHINGTON UNIVERSITY SCHOOL OF MEDICINE Cranial Health History Form Welcome to the Neurosurgery Department at Washington University. To help us treat you, please fill this form out completely. Your Name:
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Investigators, study sites Multicenter, 35 US sites. Coordinating Investigator: Richard Bergenstal, MD
 STUDY SYNOPSIS Study number Title HMR1964A/3502 Apidra (insulin glulisine) administered in a fixed-bolus regimen vs. variable-bolus regimen based on carbohydrate counting in adult subjects with type 2
STUDY SYNOPSIS Study number Title HMR1964A/3502 Apidra (insulin glulisine) administered in a fixed-bolus regimen vs. variable-bolus regimen based on carbohydrate counting in adult subjects with type 2
Summary of the risk management plan (RMP) for Cresemba (isavuconazole)
 EMA/576866/2015 Summary of the risk management plan (RMP) for Cresemba (isavuconazole) This is a summary of the risk management plan (RMP) for Cresemba, which details the measures to be taken in order
EMA/576866/2015 Summary of the risk management plan (RMP) for Cresemba (isavuconazole) This is a summary of the risk management plan (RMP) for Cresemba, which details the measures to be taken in order
Guidance on Unanticipated Problems (UPs) and Adverse Events (AEs) Bertha delanda IRB Training Specialist Research Compliance Office March 2010
 Guidance on Unanticipated Problems (UPs) and Adverse Events (AEs) Bertha delanda IRB Training Specialist Research Compliance Office March 2010 OUTLINE - AE (adverse event) - SAE (serious adverse event)
Guidance on Unanticipated Problems (UPs) and Adverse Events (AEs) Bertha delanda IRB Training Specialist Research Compliance Office March 2010 OUTLINE - AE (adverse event) - SAE (serious adverse event)
N Engl J Med 2018;378: DOI: /NEJMoa Lin, Wan-Ting 2018/06/27
 N Engl J Med 2018;378:1200-10. DOI: 10.1056/NEJMoa1710895 Lin, Wan-Ting 2018/06/27 1 Introduction Gout is a chronic illness characterized by hyperuricemia, arthropathy, tophus development, and urolithiasis
N Engl J Med 2018;378:1200-10. DOI: 10.1056/NEJMoa1710895 Lin, Wan-Ting 2018/06/27 1 Introduction Gout is a chronic illness characterized by hyperuricemia, arthropathy, tophus development, and urolithiasis
Pharmacotherapy Handbook
 Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
GSK Medicine: Study Number: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Supplemental Appendix. 1. Protocol Definition of Sustained Virologic Response. A patient has a sustained virologic response if:
 Supplemental Appendix 1. Protocol Definition of Sustained Virologic Response A patient has a sustained virologic response if: 1. The patient is a responder at the end of treatment and all subsequent planned
Supplemental Appendix 1. Protocol Definition of Sustained Virologic Response A patient has a sustained virologic response if: 1. The patient is a responder at the end of treatment and all subsequent planned
Clinical trials: Role of a DSMB
 Clinical trials: Role of a DSMB 15 th Advanced Course of Vaccinology May 15, 2014 Penny M. Heaton, MD Director of Vaccine Development Vaccine Development: Catalyze development of low cost vaccines that
Clinical trials: Role of a DSMB 15 th Advanced Course of Vaccinology May 15, 2014 Penny M. Heaton, MD Director of Vaccine Development Vaccine Development: Catalyze development of low cost vaccines that
UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES
 DISCLOSURES UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES Nothing to disclose Kylie Mueller, Pharm.D., BCPS Clinical Specialist, Infectious Diseases Spartanburg Regional Medical Center LEARNING OBJECTIVES
DISCLOSURES UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES Nothing to disclose Kylie Mueller, Pharm.D., BCPS Clinical Specialist, Infectious Diseases Spartanburg Regional Medical Center LEARNING OBJECTIVES
Lipoplatin monotherapy for oncologists
 Lipoplatin monotherapy for oncologists Dr. George Stathopoulos demonstrated that Lipoplatin monotherapy against adenocarcinomas of the lung can have very high efficacy (38% partial response, 43% stable
Lipoplatin monotherapy for oncologists Dr. George Stathopoulos demonstrated that Lipoplatin monotherapy against adenocarcinomas of the lung can have very high efficacy (38% partial response, 43% stable
NASDAQ: ZGNX. Company Presentation. October 2017
 NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
2.0 Synopsis. Paricalcitol Capsules M Clinical Study Report R&D/15/0380. (For National Authority Use Only)
 2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
Safety of Single- Versus Multi-vessel Angioplasty for Patients with AMI and Multi-vessel CAD
 Safety of Single- Versus Multi-vessel Angioplasty for Patients with AMI and Multi-vessel CAD Mun K. Hong, MD Associate Professor of Medicine Director, Cardiovascular Intervention and Research Weill Cornell
Safety of Single- Versus Multi-vessel Angioplasty for Patients with AMI and Multi-vessel CAD Mun K. Hong, MD Associate Professor of Medicine Director, Cardiovascular Intervention and Research Weill Cornell
KIDNEY FAILURE. What causes kidney failure People who are most at risk for kidney failure usually have one or more of the following causes:
 KIDNEY FAILURE Your kidneys are a pair of organs located toward your lower back. One kidney is on each side of your spine. They filter your blood and remove toxins from your body. Your kidneys send toxins
KIDNEY FAILURE Your kidneys are a pair of organs located toward your lower back. One kidney is on each side of your spine. They filter your blood and remove toxins from your body. Your kidneys send toxins
HEALTH SERVICES POLICY & PROCEDURE MANUAL
 PAGE 1 of 6 PURPOSE To establish basic understanding of indications and contraindications for transplantation of various organs. POLICY The N.C. Department of Correction, Division of Prisons, Health Services
PAGE 1 of 6 PURPOSE To establish basic understanding of indications and contraindications for transplantation of various organs. POLICY The N.C. Department of Correction, Division of Prisons, Health Services
ST. MICHAEL S HOSPITAL Guidelines for Reporting Serious Adverse Events / Unanticipated Problems to the SMH Research Ethics Board (REB) July 09, 2014
 ST. MICHAEL S HOSPITAL Guidelines for Reporting Serious Adverse Events / Unanticipated Problems to the SMH Research Ethics Board (REB) July 09, 2014 1. Introduction The St. Michael s Hospital (SMH) REB
ST. MICHAEL S HOSPITAL Guidelines for Reporting Serious Adverse Events / Unanticipated Problems to the SMH Research Ethics Board (REB) July 09, 2014 1. Introduction The St. Michael s Hospital (SMH) REB
Patient Information Last Name: First Name: Middle Initial: Address: City: State: Zip Code:
 Patient Information Last Name: First Name: Middle Initial: Address: City: State: Zip Code: Date of Birth (MM/DD/YY): Social Security #: Sex: Male Female Home Phone #: Mobile Phone #: Email Address: Marital
Patient Information Last Name: First Name: Middle Initial: Address: City: State: Zip Code: Date of Birth (MM/DD/YY): Social Security #: Sex: Male Female Home Phone #: Mobile Phone #: Email Address: Marital
Clinical Trial Synopsis TL-OPI-518, NCT#
 Clinical Trial Synopsis, NCT# 00225264 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl vs Glimepiride
Clinical Trial Synopsis, NCT# 00225264 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl vs Glimepiride
PRASUGREL HYDROCHLORIDE (Effient Eli Lilly Canada Inc.) Indication: Acute Coronary Syndrome
 CEDAC FINAL RECOMMENDATION PRASUGREL HYDROCHLORIDE (Effient Eli Lilly Canada Inc.) Indication: Acute Coronary Syndrome Recommendation: The Canadian Expert Drug Advisory Committee (CEDAC) recommends that
CEDAC FINAL RECOMMENDATION PRASUGREL HYDROCHLORIDE (Effient Eli Lilly Canada Inc.) Indication: Acute Coronary Syndrome Recommendation: The Canadian Expert Drug Advisory Committee (CEDAC) recommends that
2.0 Synopsis. Choline fenofibrate capsules (ABT-335) M Clinical Study Report R&D/06/772. (For National Authority Use Only) Name of Study Drug:
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Choline Fenofibrate (335) Name of Active Ingredient:
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Choline Fenofibrate (335) Name of Active Ingredient:
PFIZER INC. Study Center(s): A total of 6 centers took part in the study, including 2 in France and 4 in the United States.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
To provide information on the use of acetyl salicylic acid in the treatment and prevention of vascular events.
 ACETYL SALICYLIC ACID TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To provide information on the use of acetyl salicylic acid in the treatment and prevention of vascular events.
ACETYL SALICYLIC ACID TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To provide information on the use of acetyl salicylic acid in the treatment and prevention of vascular events.
SYNOPSIS. Clinical Study Report IM Double-blind Period
 Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier SYNOPSIS (For National Authority
Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier SYNOPSIS (For National Authority
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Appropriate Use Criteria for Initial Transthoracic Echocardiography in Outpatient Pediatric Cardiology (scores listed by Appropriate Use rating)
 Appropriate Use Criteria for Initial Transthoracic Echocardiography in Outpatient Pediatric Cardiology (scores listed by Appropriate Use rating) Table 1: Appropriate indications (median score 7-9) Indication
Appropriate Use Criteria for Initial Transthoracic Echocardiography in Outpatient Pediatric Cardiology (scores listed by Appropriate Use rating) Table 1: Appropriate indications (median score 7-9) Indication
STUDY 1 PHASE 3 TOP-LINE RESULTS. September 2017
 STUDY 1 PHASE 3 TOP-LINE RESULTS September 2017 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
STUDY 1 PHASE 3 TOP-LINE RESULTS September 2017 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
MEDICATION GUIDE. Epogen (Ee-po-jen) (epoetin alfa)
 MEDICATION GUIDE Epogen (Ee-po-jen) (epoetin alfa) Read this Medication Guide before you start Epogen, each time you refill your prescription, and if you are told by your healthcare provider that there
MEDICATION GUIDE Epogen (Ee-po-jen) (epoetin alfa) Read this Medication Guide before you start Epogen, each time you refill your prescription, and if you are told by your healthcare provider that there
10. Has your child ever been diagnosed with an unexplained seizure disorder or exercise-induced asthma?
 PLAYING IT SAFE Cardiac Screening Intake Form Patient Information: First Name: MI Last Name: Date of Birth Month Day Year Address: City State Zip Telephone: Second Phone Parent/Guardian Name: Primary Physician:
PLAYING IT SAFE Cardiac Screening Intake Form Patient Information: First Name: MI Last Name: Date of Birth Month Day Year Address: City State Zip Telephone: Second Phone Parent/Guardian Name: Primary Physician:
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Durlaza. Durlaza (aspirin) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.13 Subject: Durlaza Page: 1 of 4 Last Review Date: September 15, 2016 Durlaza Description Durlaza
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.13 Subject: Durlaza Page: 1 of 4 Last Review Date: September 15, 2016 Durlaza Description Durlaza
This was a multi-center study conducted at 44 study centers. There were 9 centers in the United States and 35 centers in Europe.
 Protocol CAM307: A Phase 3 Study to Evaluate the Efficacy and Safety of Frontline Therapy with Alemtuzumab (Campath ) vs Chlorambucil in Patients with Progressive B-Cell Chronic Lymphocytic Leukemia These
Protocol CAM307: A Phase 3 Study to Evaluate the Efficacy and Safety of Frontline Therapy with Alemtuzumab (Campath ) vs Chlorambucil in Patients with Progressive B-Cell Chronic Lymphocytic Leukemia These
Study No: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Blood Components & Indications for Transfusion. Neda Kalhor
 Blood Components & Indications for Transfusion Neda Kalhor Blood products Cellular Components: Red blood cells - Leukocyte-reduced RBCs - Washed RBCs - Irradiated RBCs Platelets - Random-donor platelets
Blood Components & Indications for Transfusion Neda Kalhor Blood products Cellular Components: Red blood cells - Leukocyte-reduced RBCs - Washed RBCs - Irradiated RBCs Platelets - Random-donor platelets
Study No Title : Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Cancer and the Risk of Blood Clots
 Patient & Family Guide 2018 Cancer and the Risk of Blood Clots www.nscancercare.ca Table of Contents Terms Used in this Booklet 1 What are Blood Clots? 2 When are Cancer Patients at the Highest Risk for
Patient & Family Guide 2018 Cancer and the Risk of Blood Clots www.nscancercare.ca Table of Contents Terms Used in this Booklet 1 What are Blood Clots? 2 When are Cancer Patients at the Highest Risk for
Questions and Answers: The NIH Trial of EDTA Chelation Therapy for Coronary Heart Disease
 Questions and Answers: The NIH Trial of EDTA Chelation Therapy for Coronary Heart Disease Results from the Trial to Assess Chelation Therapy will be published in the Journal of the American Medical Associatio
Questions and Answers: The NIH Trial of EDTA Chelation Therapy for Coronary Heart Disease Results from the Trial to Assess Chelation Therapy will be published in the Journal of the American Medical Associatio
Managing Major Adverse Events in Relapsed/Refractory Multiple Myeloma
 Welcome to Managing Myeloma. I am Dr. Ajay Nooka. In today's presentation, I will discuss managing major adverse events in relapsed/refractory multiple myeloma. In this video, I will provide you with information
Welcome to Managing Myeloma. I am Dr. Ajay Nooka. In today's presentation, I will discuss managing major adverse events in relapsed/refractory multiple myeloma. In this video, I will provide you with information
Sponsor/Company: sanofi-aventis Drug substance: Elitek/Fasturtec (rasburicase, SR29142)
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: sanofi-aventis Drug
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: sanofi-aventis Drug
Sildenafil or Revatio REVERSE-DBMD Trial
 Sildenafil or Revatio REVERSE-DBMD Trial Daniel P. Judge, MD Associate Professor; Cardiology Director, Center for Inherited Heart Disease Johns Hopkins University Kennedy-Krieger Institute Annual Connect
Sildenafil or Revatio REVERSE-DBMD Trial Daniel P. Judge, MD Associate Professor; Cardiology Director, Center for Inherited Heart Disease Johns Hopkins University Kennedy-Krieger Institute Annual Connect
Supplementary Online Content
 Supplementary Online Content Powles T, O Donnell PH, Massard C, et al. Efficacy and safety of durvalumab in locally advanced or metastatic urothelial carcinoma: updated results from a phase 1/2 openlabel
Supplementary Online Content Powles T, O Donnell PH, Massard C, et al. Efficacy and safety of durvalumab in locally advanced or metastatic urothelial carcinoma: updated results from a phase 1/2 openlabel
AEROSURF Phase 2 Program Update Investor Conference Call
 AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
UW MEDICINE PATIENT EDUCATION. Treatment for blocked heart arteries DRAFT. What are arteries? How do heart arteries become blocked?
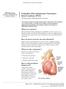 UW MEDICINE PATIENT EDUCATION Complex Percutaneous Coronary Intervention (PCI) Treatment for blocked heart arteries This handout explains complex percutaneous intervention (PCI) treatment of a coronary
UW MEDICINE PATIENT EDUCATION Complex Percutaneous Coronary Intervention (PCI) Treatment for blocked heart arteries This handout explains complex percutaneous intervention (PCI) treatment of a coronary
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Solomon SD, Uno H, Lewis EF, et al. Erythropoietic response
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Solomon SD, Uno H, Lewis EF, et al. Erythropoietic response
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Outcome
 Measure #445 (NQF 0119): Risk-Adjusted Operative Mortality for Coronary Artery Bypass Graft (CABG) National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Measure #445 (NQF 0119): Risk-Adjusted Operative Mortality for Coronary Artery Bypass Graft (CABG) National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Proper Completion of a Death Certificate"
 Proper Completion of a Death Certificate" Pennsylvania Department of Health Bureau of Health Statistics and Research Division of Statistical Registries Division of Vital Records Why should you care? 1.
Proper Completion of a Death Certificate" Pennsylvania Department of Health Bureau of Health Statistics and Research Division of Statistical Registries Division of Vital Records Why should you care? 1.
Summary of the risk management plan (RMP) for Kyprolis (carfilzomib)
 EMA/639793/2015 Summary of the risk management plan (RMP) for Kyprolis (carfilzomib) This is a summary of the risk management plan (RMP) for Kyprolis, which details the measures to be taken in order to
EMA/639793/2015 Summary of the risk management plan (RMP) for Kyprolis (carfilzomib) This is a summary of the risk management plan (RMP) for Kyprolis, which details the measures to be taken in order to
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Cornely OA, Maertens J, Winston DJ, et al. Posaconazole vs.
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Cornely OA, Maertens J, Winston DJ, et al. Posaconazole vs.
Definitions. Peace of mind today and tomorrow. CRITICAL ILLNESS Basic benefit Deluxe benefit. CRITICAL ILLNESS MULTI-PROTECTION (per child)
 Definitions Peace of mind today and tomorrow CRITICAL ILLNESS Basic benefit Deluxe benefit CRITICAL ILLNESS MULTI-PROTECTION (per child) Here are the definitions of the critical and non-critical illnesses
Definitions Peace of mind today and tomorrow CRITICAL ILLNESS Basic benefit Deluxe benefit CRITICAL ILLNESS MULTI-PROTECTION (per child) Here are the definitions of the critical and non-critical illnesses
NAME DATE Page 1. Other. Kidney Removed (Right, Left) Bladder Removed. Ovaries Removed for Endometriosis Breast Biopsy
 NAME DATE Page 1 Past Medical History: (please circle ALL that apply) Anxiety Hepatitis Arthritis Hypertension Artificial joints HIV/AIDS Asthma Hypercholesterolemia Atrial fibrillation Hyperthyroidism
NAME DATE Page 1 Past Medical History: (please circle ALL that apply) Anxiety Hepatitis Arthritis Hypertension Artificial joints HIV/AIDS Asthma Hypercholesterolemia Atrial fibrillation Hyperthyroidism
Does Adding Examples to the American Society of Anesthesiologists Physical Status Classification Improve Consistency in Assignment to Patients?
 Does Adding Examples to the American Society of Anesthesiologists Physical Status Classification Improve Consistency in Assignment to Patients? Submitted Abstract to the 2015 ASA Annual Meeting 10 Hypothetical
Does Adding Examples to the American Society of Anesthesiologists Physical Status Classification Improve Consistency in Assignment to Patients? Submitted Abstract to the 2015 ASA Annual Meeting 10 Hypothetical
Learning Objectives: At the end of this exercise, the student will be able to:
 Applications in Transfusion Medicine- A CBL Exercise- Student Guide 1 Title: Applications in Transfusion Medicine A CBL Exercise Purpose: At the conclusion of this exercise, students will be able to apply
Applications in Transfusion Medicine- A CBL Exercise- Student Guide 1 Title: Applications in Transfusion Medicine A CBL Exercise Purpose: At the conclusion of this exercise, students will be able to apply
Sponsor / Company: Sanofi Drug substance(s): Insulin Glargine. According to template: QSD VERSION N 4.0 (07-JUN-2012) Page 1
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: Not Applicable
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Patient Education. Transplant Services. Benefits and. Of a kidney/pancreas transplant
 Patient Education Benefits and Risks Of a kidney/pancreas transplant This chapter discusses the benefits as well as the risks of a kidney and/or pancreas transplant. The complications of transplant and
Patient Education Benefits and Risks Of a kidney/pancreas transplant This chapter discusses the benefits as well as the risks of a kidney and/or pancreas transplant. The complications of transplant and
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Outcome High Priority
 Quality ID #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Preventable Healthcare Harm
Quality ID #167 (NQF 0114): Coronary Artery Bypass Graft (CABG): Postoperative Renal Failure National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Preventable Healthcare Harm
CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS
 CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS Guideline Title Clinical Investigation of Anti-Anginal Medicinal Products in Stable Angina Pectoris Legislative basis
CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS Guideline Title Clinical Investigation of Anti-Anginal Medicinal Products in Stable Angina Pectoris Legislative basis
Thrombocytopenia and Chronic Liver Disease
 Thrombocytopenia and Chronic Liver Disease Severe thrombocytopenia (platelet count
Thrombocytopenia and Chronic Liver Disease Severe thrombocytopenia (platelet count
Summary of the risk management plan (RMP) for Imlygic (talimogene laherparepvec)
 EMA/713818/2015 Summary of the risk management plan (RMP) for Imlygic ( laherparepvec) This is a summary of the risk management plan (RMP) for Imlygic, which details the measures to be taken in order to
EMA/713818/2015 Summary of the risk management plan (RMP) for Imlygic ( laherparepvec) This is a summary of the risk management plan (RMP) for Imlygic, which details the measures to be taken in order to
Phase I Study of Carfilzomib and Panobinostat for Patients with Relapsed and Refractory Myeloma: A Multicenter MMRC Clinical Trial
 Phase I Study of Carfilzomib and Panobinostat for Patients with Relapsed and Refractory Myeloma: A Multicenter MMRC Clinical Trial Jonathan L. Kaufman, Todd Zimmerman, Cara A. Rosenbaum, Anuj Mahindra,
Phase I Study of Carfilzomib and Panobinostat for Patients with Relapsed and Refractory Myeloma: A Multicenter MMRC Clinical Trial Jonathan L. Kaufman, Todd Zimmerman, Cara A. Rosenbaum, Anuj Mahindra,
LIVE LONGER. Chart a course that could help you. If multiple myeloma comes back:
 1 If multiple myeloma comes back: Chart a course that could help you 1 LIVE LONGER -3 In two clinical studies with patients with relapsed multiple myeloma, two KYPROLIS -based combinations kept multiple
1 If multiple myeloma comes back: Chart a course that could help you 1 LIVE LONGER -3 In two clinical studies with patients with relapsed multiple myeloma, two KYPROLIS -based combinations kept multiple
Hydroxyurea Treatment for Sickle Cell Disease
 Hydroxyurea Treatment for Sickle Cell Disease Before Hydroxyurea After Hydroxyurea Hydroxyurea Treatment for Sickle Cell Disease This document is not intended to take the place of the care and attention
Hydroxyurea Treatment for Sickle Cell Disease Before Hydroxyurea After Hydroxyurea Hydroxyurea Treatment for Sickle Cell Disease This document is not intended to take the place of the care and attention
Blood is serious business
 Transfusion at RCH BLOOD TRANSFUSION Anthea Greenway Dept of Clinical Haematology >10000 fresh blood products per year Supports craniofacial and cardiac surgery Support bone marrow, liver transplant and
Transfusion at RCH BLOOD TRANSFUSION Anthea Greenway Dept of Clinical Haematology >10000 fresh blood products per year Supports craniofacial and cardiac surgery Support bone marrow, liver transplant and
This was a multicenter study conducted at 11 sites in the United States and 11 sites in Europe.
 Protocol CAM211: A Phase II Study of Campath-1H (CAMPATH ) in Patients with B- Cell Chronic Lymphocytic Leukemia who have Received an Alkylating Agent and Failed Fludarabine Therapy These results are supplied
Protocol CAM211: A Phase II Study of Campath-1H (CAMPATH ) in Patients with B- Cell Chronic Lymphocytic Leukemia who have Received an Alkylating Agent and Failed Fludarabine Therapy These results are supplied
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS EPTIFIBATIDE (INTEGRILIN) PROTOCOL
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
Frequently Asked Questions
 Frequently Asked Questions What is this new technique and how does it work? Researchers are testing two new investigational devices - the ResQPump and ResQPOD. The ResQPump assists EMS providers in performing
Frequently Asked Questions What is this new technique and how does it work? Researchers are testing two new investigational devices - the ResQPump and ResQPOD. The ResQPump assists EMS providers in performing
1.0 Abstract. Title. Keywords
 1.0 Abstract Title Real World Evidence of the Effectiveness of Paritaprevir/r Ombitasvir, ± Dasabuvir, ± Ribavirin in Patients with Chronic Hepatitis C - An Observational Study in Austria (REAL) Keywords
1.0 Abstract Title Real World Evidence of the Effectiveness of Paritaprevir/r Ombitasvir, ± Dasabuvir, ± Ribavirin in Patients with Chronic Hepatitis C - An Observational Study in Austria (REAL) Keywords
Fuel your determination to live longer with KYPROLIS. Look inside to learn more.
 F O R R E L A P S E D MULT IP L E M Y E L OM A APPROVED USES KYPROLIS is a prescription medication used to treat patients with relapsed or refractory multiple myeloma who have received one to three previous
F O R R E L A P S E D MULT IP L E M Y E L OM A APPROVED USES KYPROLIS is a prescription medication used to treat patients with relapsed or refractory multiple myeloma who have received one to three previous
CARDIOVASCULAR SAFETY OF FEBUXOSTAT OR ALLOPURINOL IN PATIENTS WITH GOUT AND CARDIOVASCULAR DISEASE (The CARES Trial)
 CARDIOVASCULAR SAFETY OF FEBUXOSTAT OR ALLOPURINOL IN PATIENTS WITH GOUT AND CARDIOVASCULAR DISEASE (The CARES Trial) William B. White, MD for the CARES Investigators Calhoun Cardiology Center University
CARDIOVASCULAR SAFETY OF FEBUXOSTAT OR ALLOPURINOL IN PATIENTS WITH GOUT AND CARDIOVASCULAR DISEASE (The CARES Trial) William B. White, MD for the CARES Investigators Calhoun Cardiology Center University
