Lecture 19, 04 Nov 2003 Chapter 13, Respiration, Gas Exchange, Acid-Base Balance. Vertebrate Physiology ECOL 437 University of Arizona Fall 2003
|
|
|
- Ashley Thomas
- 5 years ago
- Views:
Transcription
1 1 Lecture 19, 04 Nov 003 Chapter 13, Respiration, Gas Exchange, Acid-Base Balance Vertebrate Physiology ECOL 437 University of Arizona Fall 003 instr: Kevin Bonine t.a.: Bret Pasch
2 Vertebrate Physiology Blood-Gas Chemistry (CH13). Announcements... VOTE!
3 3 Term Paper Draft due Thursday 06 Nov. Turn in old, relevant, graded work. On the actual most recent draft use a CODE NAME so your paper can be anonymously reviewed by one of your peers. We will give you a paper to edit/review at the end of class on Thursday
4 Name that student: 4 Jane Davis Hematology Oncology French Katie Cox Tall Kim Hurd Air Force ROTC
5 Gravity and BP 5 Knut Schmidt_Nielsen 1997
6 6 Cardiac Output 6x Exercise Oxygen Consumption X 0 Knut Schmidt_Nielsen 1997
7 7 Chapter 13 Blood-Gas Chemistry Oxygen and Carbon Dioxide - Air vs. Water - Epithelial Transfer - Transport and Regulation ph regulation Chloride shift Carbonic Anhydrase Elevation Skip: Diving, Swimbladder, Exercise
8 8 Gas composition in air O CO N % of dry air pp at 760 mm Hg mmHg (at 6000m) Solubility in water (ml/l) 34 1, Why is po in lungs less than expected?
9 Effects of Temp and Solutes on O solubility 9 Temp (C) Fresh Sea Increase in temp Increase [ion] decrease solubility
10 10 Rate of diffusion depends on molecular weight (Graham s Law) Air Water O solubility > O rate of diffusion > Weight of medium < (amt. needed to get O ) Movement of medium tidal unidirectional (take in, (less energy expel) required)
11 11 Gas transfer 1. Breathing (supply air or water to respiratory surface). Diffusion of O & CO across resp. epithelium 3. Bulk transport of gases by blood (humans = m SA) 4. Diffusion across capillary walls (blood mitochondria)
12 13-1 1
13 Gas transport in blood 13 Respiratory pigments + + all have either Fe or Cu ions that O binds pigment increases O content of blood complex of proteins and metallic ions each has characteristic color that changes w/ O content ability to bind to O (affinity) affects carrying capacity of blood for O 98% of O transported via carrier molecules
14 hemoglobin hemocyanin hemerythrin Metal Fe Cu Fe 14 Distribution over 10 phyla phyla 4 phyla (all verts, many inverts) (arthropods, mollusks) Location RBCs (verts) dissolved in intracellular plasma Color deox maroon colorless colorless ox red blue reddish violet
15 15 Hemoglobin and other Respiratory Pigments Knut Schmidt_Nielsen 1997
16 hemoglobin 4 heme + 4 protein chains 16 can carry 4 O heme molecules
17 hemoglobin Fetal hemoglobin: 17 γ chains (not β) w/ higher affinity for O (enhance O transfer from mother to fetus) Affinity for CO = 00 x s greater than for O CO poisoning even at low partial pressures Antarctic icefish lack pigment low metabolic needs = low metabolism high cardiac output, blood volume large heart
18 O dissociation curve 18 hyperbolic sigmoidal not need lots of O to get near 100% Cooperativity -binding of 1st O facilitates more binding -oxygenation of 1st heme group increases affinity of remaining 3 for O
19 P - pp of O at which pigment is 50% saturated Pigment w/ High P : 50 low affinity high rate of O transfer to tissues Pigment w/ Low P : 50 high affinity high rate of O uptake
20 Factors that reduce affinity 0 1. low ph (increase [H+]). increase in CO 3. elevated Temp 4. organic compounds
21 Factors that reduce affinity 1 1. and. Increase in [CO ] or [H+] Bohr effect CO and H + bind to hemoglobin (allosteric site), which changes conformation of molecule and changes binding site for O at tissues: CO binds to hemoglobin, decreasing affinity for O, allowing better delivery of O Root effect fishes (skip)
22 Bohr Effect CO enters blood at tissues hemoglobin unloads O CO leaves blood at resp. surface hemoglobin uptake O CO + H O H CO H + + HCO - 3 Carbonic acid Bicarbonate 3 Inc in Pco inc [H+] dec ph reduces affinity
23 3 Bohr shift as a function of body size (small animals with greater Bohr shift [more acid sensitive] so can more readily leave oxygen at tissues at given PO) Knut Schmidt_Nielsen 1997
24 Factors that reduce affinity 4 4. organic compounds organophosphates in erythrocytes differ among spp. mammals:,3 DPG birds: IP 3 fish: ATP, GTP bind to hemoglobin as allosteric effectors used to maintain O affinity under hypoxic conditions at high altitude (low blood [O ]) increase,3 DPG to increase delivery of O to tissues
25 5 CO transport in blood + CO + H O H CO H + HCO - CO + OH HCO Proportions of CO, HCO - depend on ph, T, ionic strength of blood At normal ph, Temp: - 80% of CO in form of bicarbonate ion HCO 5-10% dissolved in blood 10% in form of carbamino groups 3 3 (bound to amino groups of hemoglobin)
26 Haldane effect 6 deox hemo has high affinity - for H creating inc. [HCO ] in 3 blood (more CO ) + recall equations on previous slide
27 7 Bohr effect + Haldane effect increasing [CO ] decreases affinity of hemoglobin for O, so binds CO more easily
28 CO transfer at tissue enters/leaves blood as CO (more rapid diffusion) passes thru RBCs CO produced = O released no change in ph -Chloride Shift -Carbonic Anhydrase 8 oxygenation of hemo: acidify interior (release H + ) only in RBC, not plasma Band III protein passive exchange, bidirectional maintain charge balance deox of hemo: + inc ph (bind H )
29 CO transfer at lung 9 facilitated diffusion Acidify RBC: facilitate HCO - CO 3 - dec. in HCO in 3 RBC: influx
30 Acid-Base balancing 30 Animal body ph: slightly alkaline (more OH - than H + ) maintain ph for stability of proteins (and function) H production / excretion produced: metabolism of ingested food ingest meat: acid ingest plants: base small overall effect on ph excreted continually via kidneys, gills, skin build-up of CO build-up of H + (acidify body) low CO low H + (alkaline body) +
31 ph buffers in blood: bicarbonate not true buffer, but CO / HCO ratio imp. to ph excretory organs (kidneys, gills, skin) proteins (hemoglobin), phosphates CO + H O H CO H + HCO Respiration and ph inc. lung ventilation (low body [CO ]) inc ph respiratory alkalosis buffer: kidney dec. ph by excreting HCO- 3 dec. lung ventilation (CO excretion dec.) dec. ph respiratory acidosis
32 ph buffers 3 If CO inc in extra., diffuse into cell - to form HCO and dec. 3 intracellular ph efflux of H +, or influx of HCO 3 - leads to rise in ph via ATPase or + coupled w/ Na influx Muscle vs. Brain
33 Need to REDO: Response to acid load in cell: 33 H + efflux + Na influx (cation-exchange) H + or both in passive diffusion out of cell plasma - - membrane HCO 3 influx + Cl efflux (anion-exchange) H + efflux = HCO - 3 influx HCO 3 - inside cell CO + OH - (inc. ph) Jacob-Stewart CO leaves cell to form HCO H + cycle p.543 buffering via proteins/phosphates in cell
34 Maintaining ph balance in the body 34 (acid production = acid excretion) Mammals: adjust CO excretion via lungs - 3 acid/hco excretion via kidneys
35 35 Jackson et al. 000 Apalone - softshell turtle Chrysemys - painted turtle Mg+, Ca+ (weak base carbonates) Lactic acid bone sequestration anoxia
36 Lung Anatomy 36 Nonrespiratory -Trachea -> -Bronchi -> -Bronchioles -> (13-1) Respiratory -Terminal bronchioles -> -Respiratory bronchioles -> -Alveoli -Cilia and Mucus
37 -Gas Diffusion Barriers: 37 (13-)
38 Lung Ventilation 38 -Small mammals with greater per gram O needs and therefore greater per gram respiratory surface area -Dead Space (anatomic and physiological) Swan (13-4)
39 Lung Ventilation 39 (13-3)
40 End
Lecture 22, 03 November 2005 Wrap up Carbon Dioxide Transport Begin Osmoregulation (Chapter 25-27)
 Lecture 22, 03 November 2005 Wrap up Carbon Dioxide Transport Begin Osmoregulation (Chapter 25-27) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 instr: Kevin
Lecture 22, 03 November 2005 Wrap up Carbon Dioxide Transport Begin Osmoregulation (Chapter 25-27) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 instr: Kevin
Gas Exchange in the Tissues
 Gas Exchange in the Tissues As the systemic arterial blood enters capillaries throughout the body, it is separated from the interstitial fluid by only the thin capillary wall, which is highly permeable
Gas Exchange in the Tissues As the systemic arterial blood enters capillaries throughout the body, it is separated from the interstitial fluid by only the thin capillary wall, which is highly permeable
PBL SEMINAR. HEMOGLOBIN, O 2 -TRANSPORT and CYANOSIS An Overview
 1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR HEMOGLOBIN, O 2 -TRANSPORT and CYANOSIS
1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR HEMOGLOBIN, O 2 -TRANSPORT and CYANOSIS
Dr. Puntarica Suwanprathes. Version 2007
 Dr. Puntarica Suwanprathes Version 2007 O 2 and CO 2 transport in blood Oxyhemoglobin dissociation curve O 2 consumption (VO 2 ) CO 2 production (VCO 2 ) O 2 capacity O 2 content: CaO 2 or CvO 2 %saturation
Dr. Puntarica Suwanprathes Version 2007 O 2 and CO 2 transport in blood Oxyhemoglobin dissociation curve O 2 consumption (VO 2 ) CO 2 production (VCO 2 ) O 2 capacity O 2 content: CaO 2 or CvO 2 %saturation
Carbon Dioxide Transport. Carbon Dioxide. Carbon Dioxide Transport. Carbon Dioxide Transport - Plasma. Hydrolysis of Water
 Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
OpenStax-CNX module: m Transport of Gases. OpenStax College. Abstract
 OpenStax-CNX module: m46545 1 Transport of Gases OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will
OpenStax-CNX module: m46545 1 Transport of Gases OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will
3. Which of the following would be inconsistent with respiratory alkalosis? A. ph = 7.57 B. PaCO = 30 mm Hg C. ph = 7.63 D.
 Pilbeam: Mechanical Ventilation, 4 th Edition Test Bank Chapter 1: Oxygenation and Acid-Base Evaluation MULTIPLE CHOICE 1. The diffusion of carbon dioxide across the alveolar capillary membrane is. A.
Pilbeam: Mechanical Ventilation, 4 th Edition Test Bank Chapter 1: Oxygenation and Acid-Base Evaluation MULTIPLE CHOICE 1. The diffusion of carbon dioxide across the alveolar capillary membrane is. A.
Lecture 5. Dr. Sameh Sarray Hlaoui
 Lecture 5 Myoglobin & Hemoglobin Dr. Sameh Sarray Hlaoui Myoglobin and Hemoglobin Myoglobin - Myoglobin and Hemoglobin are (metalloprotein containing a heme prosthetic group). hemeproteins - Function as
Lecture 5 Myoglobin & Hemoglobin Dr. Sameh Sarray Hlaoui Myoglobin and Hemoglobin Myoglobin - Myoglobin and Hemoglobin are (metalloprotein containing a heme prosthetic group). hemeproteins - Function as
ATP. Chapter 7, parts of 48 Cellular Respiration: Gas Exchange, Other Metabolites & Control of Respiration. Cellular Respiration
 Chapter 7, parts of 48 Cellular Respiration: Gas Exchange, Other Metabolites & Control of Respiration Cellular Respiration ATP Gas Exchange O 2 & CO 2 exchange provides O 2 for aerobic cellular respiration
Chapter 7, parts of 48 Cellular Respiration: Gas Exchange, Other Metabolites & Control of Respiration Cellular Respiration ATP Gas Exchange O 2 & CO 2 exchange provides O 2 for aerobic cellular respiration
Globular proteins Proteins globular fibrous
 Globular proteins Globular proteins Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form in a biologically functional way. Globular
Globular proteins Globular proteins Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form in a biologically functional way. Globular
Respiratory System. Introduction. Atmosphere. Some Properties of Gases. Human Respiratory System. Introduction
 Introduction Respiratory System Energy that we consume in our food is temporarily stored in the bonds of ATP (adenosine triphosphate) before being used by the cell. Cells use ATP for movement and to drive
Introduction Respiratory System Energy that we consume in our food is temporarily stored in the bonds of ATP (adenosine triphosphate) before being used by the cell. Cells use ATP for movement and to drive
3. Which statement is false about anatomical dead space?
 Respiratory MCQs 1. Which of these statements is correct? a. Regular bronchioles are the most distal part of the respiratory tract to contain glands. b. Larynx do contain significant amounts of smooth
Respiratory MCQs 1. Which of these statements is correct? a. Regular bronchioles are the most distal part of the respiratory tract to contain glands. b. Larynx do contain significant amounts of smooth
Transport of oxygen and carbon dioxide in body fluids. Circulation and Hearts. Circulation in vertebrates and invertebrates
 Circulation Transport of oxygen and carbon dioxide in body fluids Circulation and Hearts Circulation in vertebrates and invertebrates Respiratory pigments Increase the amount of oxygen carried by blood
Circulation Transport of oxygen and carbon dioxide in body fluids Circulation and Hearts Circulation in vertebrates and invertebrates Respiratory pigments Increase the amount of oxygen carried by blood
Lecture 10. Circulatory systems; flow dynamics, flow regulation in response to environmental and internal conditions.
 Lecture 10 Circulatory systems; flow dynamics, flow regulation in response to environmental and internal conditions Professor Simchon Influence of P O2 on Hemoglobin Saturation Hemoglobin saturation plotted
Lecture 10 Circulatory systems; flow dynamics, flow regulation in response to environmental and internal conditions Professor Simchon Influence of P O2 on Hemoglobin Saturation Hemoglobin saturation plotted
Chapter 42: Circulation / Gas Exchange. d = t 2
 Chapter 42: Circulation / Gas Exchange Transport systems connect organs of exchange with body cells Diffusion Lung Blood 100 m 1 s 1 mm 100 s 1 cm 10000 s d = t 2 Bulk Flow (Pressure) Blood Cells Methods
Chapter 42: Circulation / Gas Exchange Transport systems connect organs of exchange with body cells Diffusion Lung Blood 100 m 1 s 1 mm 100 s 1 cm 10000 s d = t 2 Bulk Flow (Pressure) Blood Cells Methods
Respiratory Physiology Part II. Bio 219 Napa Valley College Dr. Adam Ross
 Respiratory Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Gas exchange Gas exchange in the lungs (to capillaries) occurs by diffusion across respiratory membrane due to differences in partial
Respiratory Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Gas exchange Gas exchange in the lungs (to capillaries) occurs by diffusion across respiratory membrane due to differences in partial
GAS EXCHANGE IB TOPIC 6.4 CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM. Terminal bronchiole Nasal cavity. Pharynx Left lung Alveoli.
 IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE
 IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
Ola Al-juneidi Abdel-Mu'ez Siyam. Dr. Nayef
 3 Ola Al-juneidi Abdel-Mu'ez Siyam Dr. Nayef Transport of CO 2 We have talked previously about the role of hemoglobin in the transport of oxygen and how it is regulated by various allosteric effectors,
3 Ola Al-juneidi Abdel-Mu'ez Siyam Dr. Nayef Transport of CO 2 We have talked previously about the role of hemoglobin in the transport of oxygen and how it is regulated by various allosteric effectors,
Module G: Oxygen Transport. Oxygen Transport. Dissolved Oxygen. Combined Oxygen. Topics to Cover
 Topics to Cover Module G: Oxygen Transport Oxygen Transport Oxygen Dissociation Curve Oxygen Transport Studies Tissue Hypoxia Cyanosis Polycythemia Oxygen Transport Oxygen is carried from the lungs to
Topics to Cover Module G: Oxygen Transport Oxygen Transport Oxygen Dissociation Curve Oxygen Transport Studies Tissue Hypoxia Cyanosis Polycythemia Oxygen Transport Oxygen is carried from the lungs to
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Fluid and Electrolytes P A R T 4
 Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Carbon Dioxide Transport and Acid-Base Balance
 CHAPTER 7 Carbon Dioxide Transport and Acid-Base Balance Carbon Dioxide Transport Dioxide Transport In plasma: Carbamino compound (bound to protein) Bicarbonate Dissolved CO 2 CO 2 Is Converted to HCO
CHAPTER 7 Carbon Dioxide Transport and Acid-Base Balance Carbon Dioxide Transport Dioxide Transport In plasma: Carbamino compound (bound to protein) Bicarbonate Dissolved CO 2 CO 2 Is Converted to HCO
Mammalian Transport and The Heart
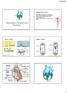 Cardiovascular System AS-G, Chapters 8-9 Blood flows through the body in a closed system (circuit) driven by the pumping power of the heart Closed vs open: does the system have vessels contained the entire
Cardiovascular System AS-G, Chapters 8-9 Blood flows through the body in a closed system (circuit) driven by the pumping power of the heart Closed vs open: does the system have vessels contained the entire
Chapter 7. Heme proteins Cooperativity Bohr effect
 Chapter 7 Heme proteins Cooperativity Bohr effect Hemoglobin is a red blood cell protein that transports oxygen from the lungs to the tissues. Hemoglobin is an allosteric protein that displays cooperativity
Chapter 7 Heme proteins Cooperativity Bohr effect Hemoglobin is a red blood cell protein that transports oxygen from the lungs to the tissues. Hemoglobin is an allosteric protein that displays cooperativity
The equilibrium between basis and acid can be calculated and termed as the equilibrium constant = Ka. (sometimes referred as the dissociation constant
 Acid base balance Dobroslav Hájek dhajek@med.muni.cz May 2004 The equilibrium between basis and acid can be calculated and termed as the equilibrium constant = Ka. (sometimes referred as the dissociation
Acid base balance Dobroslav Hájek dhajek@med.muni.cz May 2004 The equilibrium between basis and acid can be calculated and termed as the equilibrium constant = Ka. (sometimes referred as the dissociation
Acid-Base 1, 2, and 3 Linda Costanzo, Ph.D.
 Acid-Base 1, 2, and 3 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The relationship between hydrogen ion concentration and ph. 2. Production of acid
Acid-Base 1, 2, and 3 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The relationship between hydrogen ion concentration and ph. 2. Production of acid
The Respiratory System
 Elaine N. Marieb Katja Hoehn Human Anatomy & Physiology SEVENTH EDITION C H A P T E R PowerPoint Lecture Slides prepared by Vince Austin, Bluegrass Technical and Community College 22P A R T B The Respiratory
Elaine N. Marieb Katja Hoehn Human Anatomy & Physiology SEVENTH EDITION C H A P T E R PowerPoint Lecture Slides prepared by Vince Austin, Bluegrass Technical and Community College 22P A R T B The Respiratory
Renal physiology V. Regulation of acid-base balance. Dr Alida Koorts BMS
 Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Transport of Solutes and Water
 Transport of Solutes and Water Across cell membranes 1. Simple and Facilitated diffusion. 2. Active transport. 3. Osmosis. Simple diffusion Simple diffusion - the red particles are moving from an area
Transport of Solutes and Water Across cell membranes 1. Simple and Facilitated diffusion. 2. Active transport. 3. Osmosis. Simple diffusion Simple diffusion - the red particles are moving from an area
Chapter 34 Active Reading Guide Circulation and Gas Exchange
 Name: AP Biology Mr. Croft Chapter 34 Active Reading Guide Circulation and Gas Exchange Section 1 1. Gaining O 2 and nutrients while shedding CO 2 and other waste products occurs with every cell in the
Name: AP Biology Mr. Croft Chapter 34 Active Reading Guide Circulation and Gas Exchange Section 1 1. Gaining O 2 and nutrients while shedding CO 2 and other waste products occurs with every cell in the
RESPIRATORY TRACT RESPIRATORY ORGAN TGESBIOLOGY ISC 11
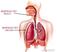 RESPIRATORY TRACT RESPIRATORY ORGAN Difference between dry and wet cough Irritation of respiratory tract Due to dust or smoke Accumulation of mucus Due to infections The respiratory tract Tracheal epithelium
RESPIRATORY TRACT RESPIRATORY ORGAN Difference between dry and wet cough Irritation of respiratory tract Due to dust or smoke Accumulation of mucus Due to infections The respiratory tract Tracheal epithelium
Chapter 9 GAS EXCHANGE & CIRCULATION
 Chapter 9 GAS EXCHANGE & CIRCULATION MECHANISMS OF GAS EXCHANGE Respiration Taking up O 2 and giving up CO 2 2 3 MECHANISMS OF GAS EXCHANGE Three phases of gas exchange Breathing Transport of oxygen and
Chapter 9 GAS EXCHANGE & CIRCULATION MECHANISMS OF GAS EXCHANGE Respiration Taking up O 2 and giving up CO 2 2 3 MECHANISMS OF GAS EXCHANGE Three phases of gas exchange Breathing Transport of oxygen and
Respiration. Chapter 37. Mader: Biology 8 th Ed.
 Respiration Chapter 37 Gas Exchange Surfaces Respiration is the sequence of events that results in gas exchange between the body s cells and the environment. Ventilation External Respiration Internal Respiration
Respiration Chapter 37 Gas Exchange Surfaces Respiration is the sequence of events that results in gas exchange between the body s cells and the environment. Ventilation External Respiration Internal Respiration
Acid-Base Tutorial 2/10/2014. Overview. Physiology (2) Physiology (1)
 Overview Acid-Base Tutorial Nicola Barlow Physiology Buffering systems Control mechanisms Laboratory assessment of acid-base Disorders of H + ion homeostasis Respiratory acidosis Metabolic acidosis Respiratory
Overview Acid-Base Tutorial Nicola Barlow Physiology Buffering systems Control mechanisms Laboratory assessment of acid-base Disorders of H + ion homeostasis Respiratory acidosis Metabolic acidosis Respiratory
thebiotutor.com AS Biology Unit 2 Exchange & Transport
 thebiotutor.com AS Biology Unit 2 Exchange & Transport 1 Exchange of materials Oxygen and Carbon dioxide are obtained passively by simple diffusion Fick s law The rate of diffusion = concentration difference
thebiotutor.com AS Biology Unit 2 Exchange & Transport 1 Exchange of materials Oxygen and Carbon dioxide are obtained passively by simple diffusion Fick s law The rate of diffusion = concentration difference
Blood Gases, ph, Acid- Base Balance
 Blood Gases, ph, Acid- Base Balance Blood Gases Acid-Base Physiology Clinical Acid-Base Disturbances Blood Gases Respiratory Gas Exchange Chemical Control of Respiration Dyshemoglobins Oxygen Transport
Blood Gases, ph, Acid- Base Balance Blood Gases Acid-Base Physiology Clinical Acid-Base Disturbances Blood Gases Respiratory Gas Exchange Chemical Control of Respiration Dyshemoglobins Oxygen Transport
There are number of parameters which are measured: ph Oxygen (O 2 ) Carbon Dioxide (CO 2 ) Bicarbonate (HCO 3 -) AaDO 2 O 2 Content O 2 Saturation
 Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Renal Physiology. April, J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine.
 Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Chapter 15 Fluid and Acid-Base Balance
 Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
1. 09/07/16 Ch 1: Intro to Human A & P 1
 Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Open Circulatory System. Closed Circulatory System
 General Types of Circulatory Systems -All circulatory systems exchange gases, nutrients, and wastes at the cellular level -Although all animals need to do this, they do so in a variety of ways: -Two types
General Types of Circulatory Systems -All circulatory systems exchange gases, nutrients, and wastes at the cellular level -Although all animals need to do this, they do so in a variety of ways: -Two types
Objectives. Blood Buffers. Definitions. Strong/Weak Acids. Fixed (Non-Volatile) Acids. Module H Malley pages
 Blood Buffers Module H Malley pages 120-126 Objectives Define a buffer system and differentiate between the buffering systems present in the body. Given an arterial blood-gas result, determine the degree
Blood Buffers Module H Malley pages 120-126 Objectives Define a buffer system and differentiate between the buffering systems present in the body. Given an arterial blood-gas result, determine the degree
Pharmacist. Drugs. body physiology. ( molecular constituents)
 Why? Pharmacist Drugs body physiology ( molecular constituents) Mechanistic levels of response: Altered patient response physiologic systems Vascular system blood, muscle, liver tissues / organs cellular
Why? Pharmacist Drugs body physiology ( molecular constituents) Mechanistic levels of response: Altered patient response physiologic systems Vascular system blood, muscle, liver tissues / organs cellular
Principles of Fluid Balance
 Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Acid/Base Balance. the concentrations of these two ions affect the acidity or alkalinity of body fluids
 Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Vertebrate Physiology 437 EXAM III NAME, Section (circle): am pm 23 November Exam is worth 100 points. You have 75 minutes.
 1 Vertebrate Physiology 437 EXAM III NAME, Section (circle): am pm 23 November 2004. Exam is worth 100 points. You have 75 minutes. True or False (please write true or false ; 10 points total; 1 point
1 Vertebrate Physiology 437 EXAM III NAME, Section (circle): am pm 23 November 2004. Exam is worth 100 points. You have 75 minutes. True or False (please write true or false ; 10 points total; 1 point
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Bio 104 Respiratory System 81
 81 Lecture Outline: Respiratory System Hole s HAP [Chapter 19] I. Introduction Respiration is the process of exchanging gases between the atmosphere and body cells. Respiration consists of: Ventilation
81 Lecture Outline: Respiratory System Hole s HAP [Chapter 19] I. Introduction Respiration is the process of exchanging gases between the atmosphere and body cells. Respiration consists of: Ventilation
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Biochemistry. Structure and function of hemoglobin M E D I C I N E. Be like stem cells, differentiate yourself from others! Editing file PO 4.
 HbA NH 2 H 2 O 2 KClO 3 Cl 2 O 7 PO 4 CH2O NAOH KMnO 4 M E D I C I N E KING SAUD UNIVERSITY Co 2 COOH MgCl 2 H 2 O Important Extra Information Doctors slides Doctors notes SO 2 HCN CCl 4 CuCl 2 Biochemistry
HbA NH 2 H 2 O 2 KClO 3 Cl 2 O 7 PO 4 CH2O NAOH KMnO 4 M E D I C I N E KING SAUD UNIVERSITY Co 2 COOH MgCl 2 H 2 O Important Extra Information Doctors slides Doctors notes SO 2 HCN CCl 4 CuCl 2 Biochemistry
Key Concepts. Learning Objectives
 Lectures 8 and 9: Protein Function, Ligand Binding -- Oxygen Binding and Allosteric Regulation in Hemoglobin [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 7, pp. 183-199 problems in textbook: chapter
Lectures 8 and 9: Protein Function, Ligand Binding -- Oxygen Binding and Allosteric Regulation in Hemoglobin [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 7, pp. 183-199 problems in textbook: chapter
UNIT VI: ACID BASE IMBALANCE
 UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
EH1008 Biomolecules. Inorganic & Organic Chemistry. Water. Lecture 2: Inorganic and organic chemistry.
 EH1008 Biomolecules Lecture 2: Inorganic and organic chemistry limian.zheng@ucc.ie 1 Inorganic & Organic Chemistry Inorganic Chemistry: generally, substances that do not contain carbon Inorganic molecules:
EH1008 Biomolecules Lecture 2: Inorganic and organic chemistry limian.zheng@ucc.ie 1 Inorganic & Organic Chemistry Inorganic Chemistry: generally, substances that do not contain carbon Inorganic molecules:
2. List seven functions performed by the respiratory system?
 The Respiratory System C23 Study Guide Tortora and Derrickson 1. In physiology we recognize that the word respiration has three meanings. What are the three different meanings of the word respiration as
The Respiratory System C23 Study Guide Tortora and Derrickson 1. In physiology we recognize that the word respiration has three meanings. What are the three different meanings of the word respiration as
Dr. Suzana Voiculescu
 Dr. Suzana Voiculescu Definition All the processes inside the body which keep the H+ concentration within normal values. Depends on water and ion balance blood gas homeostasis Blood acidity may be expressed
Dr. Suzana Voiculescu Definition All the processes inside the body which keep the H+ concentration within normal values. Depends on water and ion balance blood gas homeostasis Blood acidity may be expressed
Acids, Bases, and Salts
 Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Biology A-level: Transport. Blood. Page 1 of 22 1/18/2009. Red blood cells
 Page 1 of 22 Home A-level Biology Transport Blood Biology A-level: Transport Blood Just over half of the blood volume is made up of a pale yellow fluid called plasma. The rest of the blood is made up of
Page 1 of 22 Home A-level Biology Transport Blood Biology A-level: Transport Blood Just over half of the blood volume is made up of a pale yellow fluid called plasma. The rest of the blood is made up of
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
BIOLOGY - CLUTCH CH.42 - CIRCULATORY SYSTEM.
 !! www.clutchprep.com CONCEPT: GAS EXCHANGE AND CIRCULATION Respiratory system draws in gases from the environment, intakes O2 and outputs CO2 Circulatory system transports O2, CO2, nutrients, hormones,
!! www.clutchprep.com CONCEPT: GAS EXCHANGE AND CIRCULATION Respiratory system draws in gases from the environment, intakes O2 and outputs CO2 Circulatory system transports O2, CO2, nutrients, hormones,
Chronic Obstructive Pulmonary Disease
 136 PHYSIOLOGY CASES AND PROBLEMS Case 24 Chronic Obstructive Pulmonary Disease Bernice Betweiler is a 73-year-old retired seamstress who has never been married. She worked in the alterations department
136 PHYSIOLOGY CASES AND PROBLEMS Case 24 Chronic Obstructive Pulmonary Disease Bernice Betweiler is a 73-year-old retired seamstress who has never been married. She worked in the alterations department
O 2 O 2 O 2. Haemoglobin
 O 2 O 2 O 2 Haemoglobin O 2 O 2 O 2 98% travels in oxyhaemoglobin (in red blood cells) 2% is dissolved in plasma (compared to carbon dioxide, oxygen is relatively insoluble in plasma) O 2 is not very soluble
O 2 O 2 O 2 Haemoglobin O 2 O 2 O 2 98% travels in oxyhaemoglobin (in red blood cells) 2% is dissolved in plasma (compared to carbon dioxide, oxygen is relatively insoluble in plasma) O 2 is not very soluble
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013
 Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
1. Hemoglobin and the Movement of Oxygen. Respirator system/biochemistry
 1. Hemoglobin and the Movement of Oxygen Respirator system/biochemistry YOU MUST BE ABLE TO: Hemoglobin and the Movement of Oxygen specific aims 1. Compare structure of myoglobin and hemoglobin 2. Understand
1. Hemoglobin and the Movement of Oxygen Respirator system/biochemistry YOU MUST BE ABLE TO: Hemoglobin and the Movement of Oxygen specific aims 1. Compare structure of myoglobin and hemoglobin 2. Understand
Haemoglobin Revision. May minutes. 85 marks. Page 1 of 32
 Haemoglobin Revision May 103 97 minutes 85 marks Page 1 of 3 Q1. (a) The graph shows a dissociation curve for human haemoglobin at ph 7.4. The position of the curve is different at ph 7.. (i) (ii) Sketch
Haemoglobin Revision May 103 97 minutes 85 marks Page 1 of 3 Q1. (a) The graph shows a dissociation curve for human haemoglobin at ph 7.4. The position of the curve is different at ph 7.. (i) (ii) Sketch
Anatomy & Physiology 2 Canale. Respiratory System: Exchange of Gases
 Anatomy & Physiology 2 Canale Respiratory System: Exchange of Gases Why is it so hard to hold your breath for Discuss! : ) a long time? Every year carbon monoxide poisoning kills 500 people and sends another
Anatomy & Physiology 2 Canale Respiratory System: Exchange of Gases Why is it so hard to hold your breath for Discuss! : ) a long time? Every year carbon monoxide poisoning kills 500 people and sends another
Oxygen and CO 2 transport. Biochemistry II
 Oxygen and CO 2 transport 2 Acid- base balance Biochemistry II Lecture 9 2008 (J.S.) 1 Transport of O 2 and CO 2 O 2 INSPIRED AIR CO 2 EXPIRED AIR HCO 3 + HHb + Lungs HbO 2 + H + + HCO 3 HbO 2 + H 2 O
Oxygen and CO 2 transport 2 Acid- base balance Biochemistry II Lecture 9 2008 (J.S.) 1 Transport of O 2 and CO 2 O 2 INSPIRED AIR CO 2 EXPIRED AIR HCO 3 + HHb + Lungs HbO 2 + H + + HCO 3 HbO 2 + H 2 O
Control of Ventilation [2]
![Control of Ventilation [2] Control of Ventilation [2]](/thumbs/83/87644191.jpg) Control of Ventilation [2] สรช ย ศร ส มะ พบ., Ph.D. ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล มหาว ทยาล ยมห ดล Describe the effects of alterations in chemical stimuli, their mechanisms and response to
Control of Ventilation [2] สรช ย ศร ส มะ พบ., Ph.D. ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล มหาว ทยาล ยมห ดล Describe the effects of alterations in chemical stimuli, their mechanisms and response to
Blood Gases For beginners
 Blood Gases For beginners Lynsey ward th February 2008 4 th Aims To have a basic understanding of Blood Gas analysis. Objectives To state what acid and alkaline in the value of PH When analysing a blood
Blood Gases For beginners Lynsey ward th February 2008 4 th Aims To have a basic understanding of Blood Gas analysis. Objectives To state what acid and alkaline in the value of PH When analysing a blood
Respiration. Chapter 35
 Respiration Chapter 35 Respiration Respiration is the exchange of gasses between an organism and its environment Gas exchange occurs between the environment and blood at the lungs, and the blood and cells
Respiration Chapter 35 Respiration Respiration is the exchange of gasses between an organism and its environment Gas exchange occurs between the environment and blood at the lungs, and the blood and cells
1. Circulatory System 2. Respiration. Text: Chapters 23 & 24 (skip the invert material if you like) Then chapters 21 & 22 (skim Chapter 20)
 Lecture 19, 25 Oct 2005 Chapter 20-24 Circulation, Oxygen and Carbon Dioxide Transport 1. Circulatory System 2. Respiration Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona
Lecture 19, 25 Oct 2005 Chapter 20-24 Circulation, Oxygen and Carbon Dioxide Transport 1. Circulatory System 2. Respiration Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona
Circulation and Gas Exchange
 Circulation and Gas Exchange Sponges (porifera) Flat worms (platyhelminthes) Round worms (nematoda) Segmented worms (annelida) Stinging celled (cnidaria) Squishy (mollusca) Hard shelled (arthropods) Spiny
Circulation and Gas Exchange Sponges (porifera) Flat worms (platyhelminthes) Round worms (nematoda) Segmented worms (annelida) Stinging celled (cnidaria) Squishy (mollusca) Hard shelled (arthropods) Spiny
Lecture 25, 02 Dec 2003 Chapter 15, Feeding and Digestion Chapter 16, Energy Expenditure, Body Size
 1 Lecture 25, 02 Dec 2003 Chapter 15, Feeding and Digestion Chapter 16, Energy Expenditure, Body Size Vertebrate Physiology ECOL 437 University of Arizona Fall 2003 instr: Kevin Bonine t.a.: Bret Pasch
1 Lecture 25, 02 Dec 2003 Chapter 15, Feeding and Digestion Chapter 16, Energy Expenditure, Body Size Vertebrate Physiology ECOL 437 University of Arizona Fall 2003 instr: Kevin Bonine t.a.: Bret Pasch
Hill et al. 2004, Fig. 27.6
 Lecture 25, 15 November 2005 Osmoregulation (Chapters 25-28) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 1. Osmoregulation 2. Kidney Function Text: Chapters
Lecture 25, 15 November 2005 Osmoregulation (Chapters 25-28) Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 1. Osmoregulation 2. Kidney Function Text: Chapters
Question Expected Answers Marks Additional Guidance 1 (a) C ; E ; A ; B ; 4. PhysicsAndMathsTutor.com
 Question Expected Answers Marks Additional Guidance 1 (a) C ; E ; A ; B ; 4 Question Expected Answers Marks Additional Guidance (b) (i) P wave combined with larger peak before QRS complex ; Note: - look
Question Expected Answers Marks Additional Guidance 1 (a) C ; E ; A ; B ; 4 Question Expected Answers Marks Additional Guidance (b) (i) P wave combined with larger peak before QRS complex ; Note: - look
Omar Sami. Mustafa Khader. Yanal Shafaqouj
 8 Omar Sami Mustafa Khader Yanal Shafaqouj Let us retrieve our discussion about the ventilation-perfusion ratio (V/Q). - When (V/Q) is Zero this means that no ventilation is taking place, V is Zero; bronchial
8 Omar Sami Mustafa Khader Yanal Shafaqouj Let us retrieve our discussion about the ventilation-perfusion ratio (V/Q). - When (V/Q) is Zero this means that no ventilation is taking place, V is Zero; bronchial
Circulation and Respiration
 Lesson 10 Circulation and Respiration Introduction to Life Processes - SCI 102 1 Circulatory System: Features and Functions All circulatory systems have three major parts The heart: a pump that keeps blood
Lesson 10 Circulation and Respiration Introduction to Life Processes - SCI 102 1 Circulatory System: Features and Functions All circulatory systems have three major parts The heart: a pump that keeps blood
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Physiological Buffers
 CHM333 LECTURES 6 & 7: 9/9 9/14 FALL 2009 Professor Christine Hrycyna Physiological Buffers All about maintaining equilibrium Major buffer in blood (ph 7.4) and other extracellular fluids is the carbonic
CHM333 LECTURES 6 & 7: 9/9 9/14 FALL 2009 Professor Christine Hrycyna Physiological Buffers All about maintaining equilibrium Major buffer in blood (ph 7.4) and other extracellular fluids is the carbonic
Gas exchange Regulate blood ph Voice production Olfaction Innate immunity
 Respiration Functions Gas exchange: Grab O 2, eject CO 2 Regulate blood ph: Alters CO 2 levels Voice production: air movement past vocal cords Olfaction: in nasal cavity Innate immunity: physical protection
Respiration Functions Gas exchange: Grab O 2, eject CO 2 Regulate blood ph: Alters CO 2 levels Voice production: air movement past vocal cords Olfaction: in nasal cavity Innate immunity: physical protection
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Biology December 2009 Exam Four FORM W KEY
 Biology 251 3 December 2009 Exam Four FORM W KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Biology 251 3 December 2009 Exam Four FORM W KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Respiratory System 1. A function of the structure labelled X is to
 1 Respiratory System 1. A function of the structure labelled X is to A. produce sound. B. exchange gases. C. carry air into and out of the lung. D. stimulate the breathing centre in the brain. 2. Identify
1 Respiratory System 1. A function of the structure labelled X is to A. produce sound. B. exchange gases. C. carry air into and out of the lung. D. stimulate the breathing centre in the brain. 2. Identify
Part 1 The Cell and the Cellular Environment
 1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
Chapter 2 Transport Systems
 Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Arterial Blood Gas Analysis
 Arterial Blood Gas Analysis L Lester www.3bv.org Bones, Brains & Blood Vessels Drawn from radial or femoral arteries. Invasive procedure Caution must be taken with patient on anticoagulants ph: 7.35-7.45
Arterial Blood Gas Analysis L Lester www.3bv.org Bones, Brains & Blood Vessels Drawn from radial or femoral arteries. Invasive procedure Caution must be taken with patient on anticoagulants ph: 7.35-7.45
Animal Form and Function. Exchange surfaces. Animal Form and Function
 Animal Form and Function Surface:Volume ratio decreases with size Today s topics: Review for exam Physical Constraints affect the design of animals Homeostasis Sensors and effectors Exchange surfaces Design
Animal Form and Function Surface:Volume ratio decreases with size Today s topics: Review for exam Physical Constraints affect the design of animals Homeostasis Sensors and effectors Exchange surfaces Design
a. Describe the physiological consequences of intermittent positive pressure ventilation and positive end-expiratory pressure.
 B. 10 Applied Respiratory Physiology a. Describe the physiological consequences of intermittent positive pressure ventilation and positive end-expiratory pressure. Intermittent positive pressure ventilation
B. 10 Applied Respiratory Physiology a. Describe the physiological consequences of intermittent positive pressure ventilation and positive end-expiratory pressure. Intermittent positive pressure ventilation
adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-
 Graphics are used with permission of : adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-74. (1) Carbon dioxide arrives at the kidney tubule cell in the proximal
Graphics are used with permission of : adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-74. (1) Carbon dioxide arrives at the kidney tubule cell in the proximal
Ch. 42 Circulatory Systems And Gas Exchange
 Ch. 42 Circulatory Systems And Gas Exchange 2008-2009 In circulation What needs to be transported nutrients & fuels from digestive system respiratory gases O 2 & CO 2 from & to gas exchange systems: lungs,
Ch. 42 Circulatory Systems And Gas Exchange 2008-2009 In circulation What needs to be transported nutrients & fuels from digestive system respiratory gases O 2 & CO 2 from & to gas exchange systems: lungs,
BUFFERING OF HYDROGEN LOAD
 BUFFERING OF HYDROGEN LOAD 1. Extracellular space minutes 2. Intracellular space minutes to hours 3. Respiratory compensation 6 to 12 hours 4. Renal compensation hours, up to 2-3 days RENAL HYDROGEN SECRETION
BUFFERING OF HYDROGEN LOAD 1. Extracellular space minutes 2. Intracellular space minutes to hours 3. Respiratory compensation 6 to 12 hours 4. Renal compensation hours, up to 2-3 days RENAL HYDROGEN SECRETION
NCERT SOLUTIONS OF Life Processes
 1 NCERT SOLUTIONS OF Life Processes Question 1: Why is diffusion insufficient to meet the oxygen requirements of multicellular organisms like humans? Answer: The body structure of multicellular organism
1 NCERT SOLUTIONS OF Life Processes Question 1: Why is diffusion insufficient to meet the oxygen requirements of multicellular organisms like humans? Answer: The body structure of multicellular organism
OXYGENATION AND ACID- BASE EVALUATION. Chapter 1
 OXYGENATION AND ACID- BASE EVALUATION Chapter 1 MECHANICAL VENTILATION Used when patients are unable to sustain the level of ventilation necessary to maintain the gas exchange functions Artificial support
OXYGENATION AND ACID- BASE EVALUATION Chapter 1 MECHANICAL VENTILATION Used when patients are unable to sustain the level of ventilation necessary to maintain the gas exchange functions Artificial support
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Membrane transport. Pharmacy Dr. Szilvia Barkó
 Membrane transport Pharmacy 04.10.2017 Dr. Szilvia Barkó Cell Membranes Cell Membrane Functions Protection Communication Import and and export of molecules Movement of the cell General Structure A lipid
Membrane transport Pharmacy 04.10.2017 Dr. Szilvia Barkó Cell Membranes Cell Membrane Functions Protection Communication Import and and export of molecules Movement of the cell General Structure A lipid
