Mucosal secretory IgA and secretory piece in adult
|
|
|
- Annice Morgan
- 5 years ago
- Views:
Transcription
1 Gut, 1973, 14, Mucosal secretory IgA and secretory piece in adult coeliac disease R. J. SHINER AND JANET BALLARD' From the Medical Research Council Gastroenterology Unit, Central Middlesex Hospital, Park Royal, London SUMMARY Immunofluorescence studies with specific antisera to secretory IgA (1 IS IgA) and secretory piece were carried out on the jejunal mucosa of nine patients with adult coeliac disease (treated and untreated). The results were compared with those obtained in four normal patients and in four patients with local and systemic IgA (7S) deficiency. 11 S IgA and secretory piece were localized to the upper third of the epithelial cells of both surface and glandular epithelium in aul groups of patients. However, in the untreated coeliac patients fluorescence was also demonstrated in the basement membrane and connective tissue of the mucosa (returning to normal on treatment). On the basis of our findings, a revised pathway for the normal production of 1 1S IgA is proposed as well as an additional pathway involving a 'backflow' of 11S IgA into the lamina propria in pathological states such as coeliac disease. This backflow is reversed by an adequate gluten-free diet. It is suggested that 7S IgA and 11S IgA may be involved in immune reactions in the mucosa with antigens such as gluten. The major antibody found in human secretions is secretory IgA (11S) which differs from serum IgA (7S) in having a higher molecular weight and thus a higher sedimentation coefficient (11S rather than 7S) (Tomasi, Tan, Solomon, and Prendergast, 1965). This increase is due to a component known as secretory piece, a glycoprotein found in the gastrointestinal epithelial cells (South, Cooper, Wollheim, Hong, and Good, 1966; Hanson and Johannson, 1967; Tomasi and Bienenstock, 1968; Tourville, Adler, Bienenstock, and Tomasi, 1969). The lls IgA is formed from a dimer of 7S IgA and one molecule of secretory piece and this combination is thought to occur in the vicinity of epithelial cells (Tourville et al, 1969) before the antibody reaches the lumen.- Unlike 7S IgA, 11S IgA is resistant to digestion by trypsin (Cederblad, Johannson, and Rymo, 1966; Brown, Newcomb, and Ishizaka, 1970; Steward, 1971), thus retaining its antibody activity in the jejunal juice where the action may be directed against bacteria (Tourville, Bienenstock, and Tomasi, 1968; Girard and Kalbermatten, 1970), viruses (Balazs and Szollosy, 1962; Lipton and Steigman, 1963), parasites (Parkin, 'Send requests for reprints to J.B. Received for publication 3 July McClelland, O'Moore, Percy-Robb, Grant, and Shearman, 1972), and dietary constituents (Davis, Bierman, Pierson, Maas, and Iannetta, 1970; Ferguson and Carswell, 1972). However, the transfer of 11S IgA cannot be entirely unidirectional from epithelial cells to the lumen since several authors (Thompson, Asquith, and Cooke, 1969; Waldman, Mach, Stella, and Rowe, 1970; Thompson and Asquith, 1970; Brandtzaeg, 1971) have demonstrated it in mninute amounts in the serum of healthy subjects, and in higher concentrations in various gastrointestinal diseases, including coeliac disease. Loeb, Strober, Falchuk, and Laster (1971) demonstrated an increased synthesis of IgA, and also of IgM, by mucosal immunocytes in the adult coeliac patients after the re-introduction of gluten to their diets. In our previous publication (Shiner and Ballard, 1972b), IgA was shown to be involved in the formation of immune complexes in the jejunal mucosa of treated coeliac children after challenge with gluten. It was assumed that the only IgA involved was 7S, but the demonstration of increased amounts of lls IgA in the sera of patients with adult coeliac disease (Thompson et al, 1969) prompted us to re-investigate the type of IgA found in the jejunal mucosa of these patients. 778
2 Mucosal secretory IgA and secretory piece in adult coeliac disease This paper demonstrates that 11 S IgA and possibly atrophy of the jejunal mucosae. Six patients (nos. also free secretory piece are found in the lamina 1,4,6,7,8, and 9) were studied after treatment with a propria of untreated coeliac patients. That lls IgA gluten-free diet. Five of these showed a mild partial may be involved in subepithelial immune reactions villous atrophy on histology, but the sixth (no. 6), would seem to be a new concept. who admitted to dietary errors, had a patchy partial and subtotal villous atrophy. Materials and Methods The jejunal biopsies from eight non-coeliac patients, aged 13 to 59 (average 37 years), served as controls Nine patients with adult coeliac disease were studied, (table II). In four of these (nos. 10, 11, 12, and 13), two of whom were biopsied before and after treat- gastrointestinal disease was ruled out and the ment (table 1). They were aged 16 to 56 years, jejunal mucosa was normal. The other four (nos. 14, (average 35 years). Four patients were untreated 15, 16, and 17) had local and systemic immune (nos. 1, 2, 3, and 4), and patient no. 5 had been on deficiency disease with histological normal villous oral prednisone only. All five had a subtotal villous architecture. Patient Diagnosis of Histology of Secretory Piece IS IgA 7S IgA IgM IgG No. Coeliac Jejunal Disease Mucosa Epi- Base- Con- Epi- Base- Con- Plas- Base- Con- Plas- Base- Con- Plas- Base- Conthelial ment nective thelial ment nective ma ment nective ma ment nective ma ment nective Cells Mem- Tissue Cells Mem- Tissue Cells Mem- Tissue Cells Mem- Tissue Cells Mem- Tissue (upper brane (upper brane brane brane brane 1e ± 2 Untreated SVA Untreated SVA Untreated SVA Untreated SVA Steroids only SVA Treated PVA-SVA I Treated PVA Treated PVA Treated PVA Treated PVA Treated PVA Normal values Table I Immunofluorescence results in coeliac disease untreated and treated + +-strongly positive SVA-Subtotal villous atrophy PVA-Partial villous atrophy +-positive +-weakly positive Patient Diagnosis Histology of Secretory Piece IS IgA 7S IgA IgM IgG No. Jejunal Mucosa Epi- Base- Conn- Epi- Base- Con- Plas- Base- Con- Plas- Base- Con- Plas- Base- Conthelial ment nective thelial ment nective ma ment nective ma ment nective ma ment nective Cells Mem- Tissue Cells Mem- Tissue Cells Mem- Tissue Cells Mem- Tissue Cells Mem- Tissue (upper brane (upper brane brane brane brane 10 Normal Normal Normal Normal +± Normal Normal Normal Normal Hypogamma- Normal globulinaemia with NLH 15 IgA & IgG Normal deficiency 16 A-gamma- Normal globulinaemia 17 A-gamma- Normal globulinaemia Normal values Table II Immunofluorescence results in control patients NLH-Nodular lymphoid hyperplasia Key-see table I. 779
3 780 Jejunal mucosal biopsies (Crosby capsule) were performed on all patients as a diagnostic procedure and part of each specimen was retained, immediately frozen in liquid nitrogen, and stored at -80C for immunofluorescence studies. The antisera to 1 S IgA and secretory piece (Nordic Pharmaceuticals), both raised in sheep, were purified before use by precipitation with 40 and 50% ammonium sulphate and dialysis, giving a single gamma globulin band on clectrophoresis. The specificity of both antisera was tested by immunoelectrophoresis (fig 1). Precipitin lines identified for IgA and secretory piece excluded the possibility of reactions with other serum or milk proteins such as lactoferrin. Fig 1 Immunoelectrophoresis showing monospecific precipitin lines between normal human serum (NHS) in wells 1 and 3, normal defatted milk (NHM) in well 2 and lls IgA or secretory piece. Secretory piece antiserum showed no reaction with normal human serum. The indirect immunofluorescence technique was used as follows: sections 5g thick were cut from each biopsy at - 20C on a cryostat, fixed for 15 minutes in acetone, and washed for 45 minutes with three changes of phosphate-buffered saline (PBS) ph 7.3. The sections were then incubated for 30 to 40 minutes with antiserum to 1 IS IgA or to secretory piece followed by a 45-minute wash in phosphatebuffered saline. The slides were stained for 45 minutes with fluorescein-isothiocyanate (FITC) conjugated antisheep immunoglobulin previously diluted and absorbed with liver powder. This was followed by a further 45-minute wash in phosphate-buffered saline before mounting the sections in Polarfluor B. Slides were stored at +4 C. In addition some sections were treated with an antiserum to lactoferrin (Nordic Pharmaceuticals) because it had previously been reported (Tomasi and Bienenstock, 1968) that there is a possibility of R. J. Shiner and Janet Ballard cross reaction between ls IgA or secretory piece antisera and lactoferrin. The sections stained for the presence of lactoferrin showed fluorescence of goblet cells only, verified by simultaneous examination of the sections with phase-contrast illumination and incident light fluorescence. This contrasted with the characteristic staining pattern in the upper third of epithelial cells seen with lls IgA and secretory piece antisera. The direct staining technique using FITCconjugated 7S IgA, IgM, and IgG antisera (Wellcome reagents) was employed for the demonstration of immunoglobulins in the plasma cells of the lamina propria. The specificity of these antisera had been tested previously (unpublished data) by blocking the reaction, ie, incubating the tissue with unconjugated antiserum before staining. The blocking reactions only occurred when the unconjugated and corjugated antisera were of the same immunoglobulin class. For most specimens, both techniques were repeated and reproducible results were obtained. The antisera to 7S IgA and lls IgA were found to be specific on immunofluorescence for their particular immunoglobulin class alone. However, it seemed probable that the antiserum to secretory piece reacted with secretory piece contained in lls IgA and not only with the free molecule. All sections were studied in random order on the day of preparation or the following day with a Leitz Orthoplan microscope. Between two and six slides of each type were studied and gi.aded by one of us, without prior knowledge of the particular antisera used. The degree of staining of the upper part of epithelial cells, basement membrane, and connective tissue with 11S IgA and secretory piece antisera was expressed as strongly positive (+ +), positive (+), or weakly positive (±). The number of plasma cells stained with 7S IgA, IgM, and IgG antisera was graded 1 to 5, the normal values for this laboratory being 4 for 7S IgA, 3 for IgM, and 1 or less for IgG. Results UNTREATED COELIAC DISEASE In untreated coeliac patients (nos. 1-4) fluorescent staining with antisera to 1 IS IgA (fig 2) and secretory piece was seen in the basement membrane of surface and crypt epithelial cells and the surrounding connective tissue as well as in the upper third of epithelial cells where it is normally found (table I). Patient no. 5, treated with steroids but not with a gluten-free diet, showed staining with lls IgA and secretory piece antisera of epithelial cells and basement membrane but not connective tissue. Patient
4 Mucosal secretory IgA and secretory piece in adult coeliac disease Fig 2 Jejunal mucosa from an untreated coeliac patient (no. 1) stained with antiserum to uis IgA. Bright fluorescence is seen in the upper third of epithelial cells (E), basement membrane (BM), and connective tissue (CI) and the intercellular spaces appear outlined (arrows). L = crypt lumen. Approximately x 250. no. 6, inadequately treated with a gluten-free diet, showed a similar distribution of fluorescence to the untreated group but staining of the connective tissue with anti-secretory piece was absent. All six patients showed staining of the intercellular spaces between epithelial cells with both lls IgA and secretory piece antisera. TREATED COELIAC DISEASE In our five treated coeliac patients (nos. 1 and 4 repeated after treatment and nos. 7, 8, and 9) staining with antisera to 11S IgA and secretory piece was mainly confined to the upper third of epithelial cells, ie, normal distribution. The exception was patient no. 1 in whom connective tissue staining was observed with 11S IgA and secretory piece antisera. He had been treated with a gluten-free diet for only three months, whereas all the other patients had been on the diet for more than 12 months. None of the well treated patients showed staining of the intercellular spaces. The distribution of staining with antisera to lls IgA and secretory piece seemed to be related to the 781 histological state of the jejunal mucosa in the coeliac patients. In those patients with a subtotal villous atrophy, staining of the basement membranes and connective tissue as well as the upper third of the epithelium was observed. In those with milder mucosal abnormalities (partial villous atrophy), where at least most of the gluten was eliminated from the diet, the i is IgA and secretory piece showed a normal distribution, ie, in the epithelial cells only. CONTROL PATIENTS In our control patients (table II) staining for lls IgA (fig 3) and secretory piece was localized to the upper third of the epithelial cells only and was not seen in the intercellular spaces, basement membranes, or connective tissue. Similar staining of epithelial cells in the hypo- and agammaglobulinaemic patients (nos. 14 to 17) showed the presence of secretory piece despite an absence or virtual absence of 7S IgA in the mucosa. Fig 3 Jejunal mucosa from a control patient (no. 10) stained with 1S IgA antiserum. Only the upper third of epithelial cells (E) is fluorescent. The bright spots in the lamina propria are autofluorescent eosinophils. L = crypt lumen. Approximately x S IgA IgM, AND IgG Basement membrane and connective tissue staining was seen with 7S IgA in the untreated coeliacs (1, 2, and 3) but not in adequately treated patients (1, 4, 7, 8, 9) nor in the normal controls (10 to 13). The number of 7S IgA and IgM containing plasma
5 782 cells was increased in only one patient (no. 3, an untreated coeliac) who also had high levels of serum immunoglobulins. Patient no. 9, a well treated coeliac, had a reduced number of 7S IgA plasma cells in the lamina propria and she also had a low serum IgA level. An increase of IgG-producing plasma cells was noted in four out of five coeliacs not treated with a gluten-free diet but in none of the treated coeliac or control patients. Connective tissue staining with IgG antisera was found in all our patients except those with a serum IgG deficiency and was thought to be non-specific, as noted by us before (Shiner and Ballard, 1972a). Discussion This study has confirmed the localization of secretory piece to the upper part of the epithelial cells of the normal jejunal mucosa, as first proposed by Tourville et al (1969). The pathway of IgA from plasma cell to lumen suggested by these authors is shown in figure 4 (pathway A). The 7S IgA produced by local plasma cells is transported through the basement membrane of crypt and surface epithelium and passes either into the intercellular spaces or directly into the lower part of the epithelial cells. Dimerization and combination with secretory piece is thought to occur in the intercellular spaces. Because of the tight junctions between epithelial Pothway A Pothwoy a ( ) R. J. Shiner and Janet Ballard cells the lls IgA formed in the intercellular spaces must enter the upper part of the epithelial cells before reaching the lumen. Our hypothesis for the normal production of is IgA differs from the above and is shown as pathway B. It is based on the evidence that in our normal patients staining of the intercellular spaces was observed with 7S IgA but not with lls IgA or secretory piece antisera, and that staining of the upper third of the epithelium was seen with 1 IS IgA and secretory piece but not 7S IgA antisera. It is therefore proposed that the locally produced 7S IgA passes through the intercellular spaces to enter the upper third of the epithelium where it combines with secretory piece to form lls IgA. This is then transported across the microvilli into the lumen. On the basis of our findings in the untreated coeliac patients, an additional pathway C is suggested which may occur in pathological states. In this, a 'backflow' of lls IgA from the epithelium to the lamina propria occurs as shown by lls IgA and secretory piece staining of intercellular spaces, basement membrane, and connective tissue. The available antiserum to secretory piece unfortunately does not distinguish between the free molecule and that which is part of the lls IgA. Therefore the secretory piece staining is probably due to the presence of secretory piece in lls IgA, although the possibility that a backflow of free secretory piece Fig 4 Diagram showing the formation and distribution of IS IgA in the region of the epithelium. For explanation see text. BM = basement membrane. SP = secretory piece. = CT connective tissue. A > Sp = IS IgA. A = 7SIgA. A> Pathway C~~~~~~~~~~~~~~~~~~~ PathwayS A Pathwoy C
6 Mucosal secretory IgA and secretory piece in adult coeliac disease 783 also occurs cannot be ruled out. The exact site of formation of 11 S IgA in coeliac disease is not known but probably occurs in the upper third of epithelial cells as in normal patients although it could also be formed between epithelial cells, in the basement membrane, or in connective tissue. In the last two sites, I 1S IgA and 7S IgA may be involved in the formation of immune complexes with antigens such as gluten. This is supported by the demonstration of fluorescent staining of the lamina propria with antisera to these factors in untreated coeliac patients but not in treated or control patients. A similar localization was previously demonstrated for 7S IgA in treated coeliac children following gluten challenge (Shiner and Ballard, 1972b) and ultrastructural studies on the same patients showed cytopathological changes in the basement membrane and connective tissue (Shiner, 1973). The removal of gluten from the diet of coeliac patients apparently stops the backflow of 11S IgA into the lamina propria so that the normal pathway B remains. The localization of 11S IgA and secretory piece in well treated coeliac patients is indistinguishable from normal. The demonstration of secretory piece in the epithelial cells of our patients with a- or hypogammaglobulinaemia is in accordance with the findings of others (Brown, Butterfield, Savage, and Tada, 1972) and indicates that epithelial cells produce secretory piece regardless of any deficiency of local or systemic IgA. Plasma cell IgM was not consistently increased in any of our groups of patients, unlike Douglas, Crabbe, and Hobbs (1970) who reported an increased density of IgM-containing plasma cells in untreated coeliac patients. IgG-containing plasma cells were usually increased in the mucosae of our untreated coeliac patients and this was also reported by S0ltoft (1970). The evidence from this, and our previous publication, is very suggestive of important subephithelial events occurring in coeliac disease involving both 7S and lls IgA in response to antigenic stimulation. The demonstrated backflow of lls IgA into the lamina propria in untreated coeliac disease would explain the increased serum levels of lls IgA (Thompson, Asquith, and Cooke, 1969). We are grateful to Dr Margot Shiner for permitting us to study her patients, and for her enthusiastic encouragement, and to Dr A. D. B. Webster, Northwick Park Hospital, Harrow, Middlesex, for allowing us to include his patients with immune deficiencies. We also thank Dr J. Snoijink of Nordic Pharmaceuticals for providing us with the immunoelectrophoretic data. References Balazs, V., and Szollosy, E. (1962). Studies on the virus neutralising capacity ofantacid gastric juices. Experientia (Basel),18, Brandtzaeg, P. (1971). Human secretory immunoglobulins. V. Occurrence of secretory piece in human serum. J. Immunol, 106, Brown, W. R., Butterfield, D., Savage, D., and Tada, T. (1972). Clinical, microbiological and immunological studies inpatients with immunoglobulin deficiencies and gastrointestinal disorders. Gut, 13, Brown, W. R., Newcomb, R. W., and Ishizaka, K. (1970). Proteolytic degradation of exocrine and serum immunoglobulins. J. clin. Invest., 49, Cederblad, G., Johannson, B. G., and Rymo, L. (1966). Reduction and proteolytic degradation of immunoglobulin A from human colostrum. Acta chem. scand., 20, Davis, S. D., Bierman, C. W., Pierson, W. E., Mass, C. W., and lannetta, A. (1970). Clinical nonspecificity of milk coproantibodies in diarrheal stools. New Engl. J. Med., 282, Douglas, A. P., Crabb6, P. A., and Hobbs, J. R. (1970). Immunochemical studies of the serum, intestinal secretions and intestinal mucosa in patients with adult celiac disease and other forms of the celiac syndrome. Gastroenterology, 59, Ferguson, A., and Carswell, F. (1972). Precipitins to dietary proteins in serum and upper intestinal secretions of coeliac children. Brit. med. J., 1, Girard, J. P., and Kalbermatten, A. de (1970). Les immunoglobulines dans les s6cr6tions duodeno-pancreatiques et la bile. Schweiz. med. Wschr., 100, Hanson, L. A., and Johannson, B. G. (1967). Studies on secretory IgA. In Gamma Globulins: Structure and Control of Biosynthesis: Proceedings of the Third Nobel Symposium, edited by J. Killander, pp Interscience, New York, Almquist and Wiksel, Stockholm. Lipton, M. M., and Steigman, A. J. (1963). Human coproantibody against polioviruses. J. infect. Dis., 112, Loeb, P. M., Strober, W., Falchuk, Z. M., and Laster, L. (1971). Incorporation of L-leucine-14C into immunoglobulins by jejunal biopsies of patients with celiac sprue and other gastrointestinal diseases. J. clin. Invest., 50, Parkin, D. M., McClelland, D. B. L., O'Moore, R. R., Percy-Robb, I. W., Grant, I. W. B., and Shearman, D. J. C. (1972). Intestinal bacterial flora and bile salt studies in hypogammaglobulinaemia. Gut, 13, Shiner, M. (1973). Ultrastructural changes suggestive of immune reactions in the jejunal mucosa of coeliac children following gluten challenge. Gut, 14, Shiner, M., and Ballard, J. (1972a). Dermatitis herpetiformis-a disorder of immunity? (Letter). Lancet, 1, 796. Shiner, M., and Ballard, J. (1972b). Antigen-antibody reactions in the jejunal mucosa in childhood coeliac disease after gluten challenge. Lancet, 1, S0ltoft, J. (1970). Immunoglobulin-containing cells in non-tropical sprue. Clin. exp. Immunol., 6, South, M. A., Cooper, M. D., Wollheim, F. A., Hong, R., and Good, R. A. (1966). The IgA system. 1. Studies of the transport and immunochemistry of IgA in the saliva. J. exp. Med., 123, Steward, M. W. (1971). Resistance of rabbit secretory IgA to proteolysis. Biochim. biophys. Acta (Amst.), 236, Thompson, R. A., and Asquith, P. (1970). Quantitation of exocrine IgA in human serum in health and disease. Clin. exp. Immunol., 7, Thompson, R. A., Asquith, P., and Cooke, W. T. (1969). Secretory IgA in the serum. Lancet, 2, Tomasi, T. B., Jr., and Bienenstock, J. (1968). Secretory immunoglobulins. Advanc. Immunol., 9, Tomasi, T. B., Tan, E. M., Solomon, A., and Prendergast, R. (1965). Characteristics of an immune system common to certain external secretions. J. exp. Med., 121, Tourville, D. R., Alder, R. H., Bienenstock, J., and Tomasi,T. B., Jr. (1969). The human secretory immunoglobulin system: immunohistological localisation of va, secretory 'piece' and lactoferrin in normal human tissues. J. exp. Med., 129, Tourville, D., Beinenstock J., and Tomasi, T. B., Jr. (1968). Natural antibodies of human serum, saliva and urine reactive with Escherichia coli. Proc. soc. exp. Biol. (N. Y.), 128, Waldman, R. H., Mach, J. P., Stella, M. M., and Rowe, D. S. (1970). Secretory IgA in human serum. J. Immunol., 105,
These studies were made at a time when it was not. yet fully established that mast cells could react with
 Gut, 1975, 16, 861-866 Mast cells and immunoglobulin E in inflammatory bowel disease G. LLOYD1, F. H. Y. GREEN, H. FOX, V. MANI2, AND L. A. TURNBERG2 From the Department ofpathology, University of Manchester,
Gut, 1975, 16, 861-866 Mast cells and immunoglobulin E in inflammatory bowel disease G. LLOYD1, F. H. Y. GREEN, H. FOX, V. MANI2, AND L. A. TURNBERG2 From the Department ofpathology, University of Manchester,
The purpose of this paper is to investigate the. were carried out to determine the immunoglobulin
 Gut, 1974, 15, 284-288 Studies on the nature and significance of connective tissue antibodies in adult coeliac disease and Crohn's disease A. F. N. MAGALHAES, T. J. PETERS, AND WILLIAM F. DOE From the
Gut, 1974, 15, 284-288 Studies on the nature and significance of connective tissue antibodies in adult coeliac disease and Crohn's disease A. F. N. MAGALHAES, T. J. PETERS, AND WILLIAM F. DOE From the
The cell population of the upper jejunal mucosa in tropical sprue and postinfective malabsorption
 Gut, 1974, 15, 387-391 The cell population of the upper jejunal mucosa in tropical sprue and postinfective malabsorption R. D. MONTGOMRY AND A. C. I. SHARR From the Metabolic Unit, ast Birmingham Hospital,
Gut, 1974, 15, 387-391 The cell population of the upper jejunal mucosa in tropical sprue and postinfective malabsorption R. D. MONTGOMRY AND A. C. I. SHARR From the Metabolic Unit, ast Birmingham Hospital,
Dissecting Microscope Appearance of
 Arch. Dis. Childh., 1967, 42, 626. Dissecting Microscope Appearance of Small Bowel Mucosa in Children JOHN WALKER-SMITH From Professorial Unit, Royal Alexandra Hospital for Children, Camperdown, Sydney,
Arch. Dis. Childh., 1967, 42, 626. Dissecting Microscope Appearance of Small Bowel Mucosa in Children JOHN WALKER-SMITH From Professorial Unit, Royal Alexandra Hospital for Children, Camperdown, Sydney,
Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm
 Ann. rheum. Dis. (1973), 32, 53 Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm P. V. SHIRODARIA, K. B. FRASER, AND F. STANFORD From the Department
Ann. rheum. Dis. (1973), 32, 53 Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm P. V. SHIRODARIA, K. B. FRASER, AND F. STANFORD From the Department
Ultrastructural changes suggestive of immune. following gluten challenge. reactions in the jejunal mucosa of coeliac children
 Gut, 1973, 14, 1-12 Ultrastructural changes suggestive of immune reactions in the jejunal mucosa of coeliac children following gluten challenge MARGOT SHINER' From the Medical Research Council, Gastroenterology
Gut, 1973, 14, 1-12 Ultrastructural changes suggestive of immune reactions in the jejunal mucosa of coeliac children following gluten challenge MARGOT SHINER' From the Medical Research Council, Gastroenterology
Direct immunofluorescence of skin using formalin-fixed paraffin-embedded sections
 Direct immunofluorescence of skin using formalin-fixed paraffin-embedded sections SL MERA, EW YOUNG, AND JWB BRADFIELD J Clin Pathol 1980; 33: 365-369 From the University Department ofpathology, University
Direct immunofluorescence of skin using formalin-fixed paraffin-embedded sections SL MERA, EW YOUNG, AND JWB BRADFIELD J Clin Pathol 1980; 33: 365-369 From the University Department ofpathology, University
The mucosa associated lymphoid tissue or (MALT) Local immune component are recently being talked by immunologist as an active local immune system it
 The mucosa associated lymphoid tissue or (MALT) Local immune component are recently being talked by immunologist as an active local immune system it consist of both B- and T- Cells epithelial cell producing
The mucosa associated lymphoid tissue or (MALT) Local immune component are recently being talked by immunologist as an active local immune system it consist of both B- and T- Cells epithelial cell producing
ranged from two months to 21 years (mean 5% years)
 Gut, 1978, 19, 754-758 Response of the skin in dermatitis herpetiformis to a gluten free diet, with reference to jejunal morphology B. T. COOPER, E. MALLAS, M. D. TROTTER, AND W. T. COOKE From the Nutritional
Gut, 1978, 19, 754-758 Response of the skin in dermatitis herpetiformis to a gluten free diet, with reference to jejunal morphology B. T. COOPER, E. MALLAS, M. D. TROTTER, AND W. T. COOKE From the Nutritional
Chronic immune colitis in rabbits
 Chronic immune colitis in rabbits A. S. MEE, J. E. McLAUGHLIN, H. J. F. HODGSON, AND D. P. JEWELL Gut, 1979, 20, 1-5 From the Academic Department of Medicine and Department of Histopathology, Royal Free
Chronic immune colitis in rabbits A. S. MEE, J. E. McLAUGHLIN, H. J. F. HODGSON, AND D. P. JEWELL Gut, 1979, 20, 1-5 From the Academic Department of Medicine and Department of Histopathology, Royal Free
Immunoglobulins in chronic liver disease
 Gut, 1968, 9, 193-198 Immunoglobulins in chronic liver disease TEN FEIZI From the Department of Medicine, Royal Free Hospital, London The association of high gamma globulin levels with hepatic cirrhosis
Gut, 1968, 9, 193-198 Immunoglobulins in chronic liver disease TEN FEIZI From the Department of Medicine, Royal Free Hospital, London The association of high gamma globulin levels with hepatic cirrhosis
fixing reactions which could release substances liable to cause tissue damage in the intestine. It is therefore important to obtain information
 Gut, 1972, 13, 450-458 Bacterial agglutination studies with secretory IgA prepared from human gastrointestinal secretions and colostrum D. B. L. McCLELLAND, R. R. SAMSON, D. M. PARKIN, AND D. J. C. SHEARMAN
Gut, 1972, 13, 450-458 Bacterial agglutination studies with secretory IgA prepared from human gastrointestinal secretions and colostrum D. B. L. McCLELLAND, R. R. SAMSON, D. M. PARKIN, AND D. J. C. SHEARMAN
Bacteriology. Mycology. Patient: SAMPLE PATIENT DOB: Sex: MRN: Rare. Rare. Positive. Brown. Negative *NG. Negative
 Patient: SAMPLE PATIENT DOB: Sex: MRN: 3.2 0.9-26.8 U/g 1.2 0.2-3.3 mg/g 2.2 1.3-8.6 micromol/g 1.1 1.3-23.7 mg/g 1.1 0.2-3.5 mg/g Rare 1.0 0.2-8.8 mg/g Rare 4.4 2.6-32.4 mg/g 64.6 >= 13.6 micromol/g Bacteriology
Patient: SAMPLE PATIENT DOB: Sex: MRN: 3.2 0.9-26.8 U/g 1.2 0.2-3.3 mg/g 2.2 1.3-8.6 micromol/g 1.1 1.3-23.7 mg/g 1.1 0.2-3.5 mg/g Rare 1.0 0.2-8.8 mg/g Rare 4.4 2.6-32.4 mg/g 64.6 >= 13.6 micromol/g Bacteriology
Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology. Robin Lorenz
 Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology Robin Lorenz rlorenz@uab.edu Why do we Need to Understand How the Mucosal Immune System Works? The mucosa is the major
Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology Robin Lorenz rlorenz@uab.edu Why do we Need to Understand How the Mucosal Immune System Works? The mucosa is the major
containing immunoglobulin in the respiratory tract of normal man and class of immunoglobulin
 Thorax (1976), 31, 158 Distribution of plasma cells and other cells containing immunoglobulin in the respiratory tract of normal man and class of immunoglobulin contained therein C A SOUTAR Cardiothoracic
Thorax (1976), 31, 158 Distribution of plasma cells and other cells containing immunoglobulin in the respiratory tract of normal man and class of immunoglobulin contained therein C A SOUTAR Cardiothoracic
Protein synthesis by cultured jejunal mucosa from
 Gut, 1981, 22, 623-627 Protein synthesis by cultured jejunal mucosa from control subjects and patients with coeliac disease P E JONES,* C L L'HIRONDEL,t AND T J PETERS$ From the Department of Medicine,
Gut, 1981, 22, 623-627 Protein synthesis by cultured jejunal mucosa from control subjects and patients with coeliac disease P E JONES,* C L L'HIRONDEL,t AND T J PETERS$ From the Department of Medicine,
 SUMMARY Coeliac disease is a common food intolerance in Western populations, in which it has a prevalence of about 1%. In early infancy, when the transition is made to a gluten-containing diet (particularly
SUMMARY Coeliac disease is a common food intolerance in Western populations, in which it has a prevalence of about 1%. In early infancy, when the transition is made to a gluten-containing diet (particularly
Is a raised intraepithelial lymphocyte count with normal duodenal villous architecture clinically relevant?
 424 ORIGINAL ARTICLE Is a raised intraepithelial lymphocyte count with normal duodenal villous architecture clinically relevant? S Mahadeva, J I Wyatt, P D Howdle... See end of article for authors affiliations...
424 ORIGINAL ARTICLE Is a raised intraepithelial lymphocyte count with normal duodenal villous architecture clinically relevant? S Mahadeva, J I Wyatt, P D Howdle... See end of article for authors affiliations...
Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall
 Histol Histopath (1 990) 5: 161-1 67 Histology and Histopathology Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall Reinhold Kittelberger,
Histol Histopath (1 990) 5: 161-1 67 Histology and Histopathology Distribution of type IV collagen, laminin, nidogen and fibronectin in the haemodynamically stressed vascular wall Reinhold Kittelberger,
IMMUNOLOGIC REACTIVITY IN HUMAN BREAST CANCER AGAINST CULTURED HUMAN BREAST TUMOR CELLS
 22 IMMUNOLOGIC REACTIVITY IN HUMAN BREAST CANCER AGAINST CULTURED HUMAN BREAST TUMOR CELLS Michael P. Lerner*, J. H. Anglin, Peggy L. Munson, Peggy J. Riggs, Nancy E. Manning, and Robert E. Nordquist Departments
22 IMMUNOLOGIC REACTIVITY IN HUMAN BREAST CANCER AGAINST CULTURED HUMAN BREAST TUMOR CELLS Michael P. Lerner*, J. H. Anglin, Peggy L. Munson, Peggy J. Riggs, Nancy E. Manning, and Robert E. Nordquist Departments
Coeliac disease with severe hypogammaglobulinaemia
 Gut, 1981, 22, 153-157 Case report Coeliac disease with severe hypogammaglobulinaemia A D B WEBSTER, G SLAVIN, M SHINER, T A E PLATTS-MILLS, AND G L ASHERSON From the Divisions of Immunological Medicine
Gut, 1981, 22, 153-157 Case report Coeliac disease with severe hypogammaglobulinaemia A D B WEBSTER, G SLAVIN, M SHINER, T A E PLATTS-MILLS, AND G L ASHERSON From the Divisions of Immunological Medicine
Coeliac Disease Bible Class Questions and Answers
 Coeliac Disease Bible Class Questions and Answers Jan Hendrik Niess What is the definition of coeliac disease? Coeliac disease is an immune reaction to gluten (wheat, barely, rye) in an genetic predisposed
Coeliac Disease Bible Class Questions and Answers Jan Hendrik Niess What is the definition of coeliac disease? Coeliac disease is an immune reaction to gluten (wheat, barely, rye) in an genetic predisposed
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE
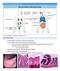 LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Rectal mucosal plasma cells in inflammatory bowel
 Gut, 1983, 24, 519-524 Rectal mucosal plasma cells in inflammatory bowel disease B B SCOTT, ANNE GOODALL, P STEPHENSON, AND D JENKINS From the Departments of Medicine and Pathology, Lincoln County Hospital,
Gut, 1983, 24, 519-524 Rectal mucosal plasma cells in inflammatory bowel disease B B SCOTT, ANNE GOODALL, P STEPHENSON, AND D JENKINS From the Departments of Medicine and Pathology, Lincoln County Hospital,
What is your diagnosis? a. Lymphocytic colitis. b. Collagenous colitis. c. Common variable immunodeficiency (CVID) associated colitis
 Case History A 24 year old male presented with fatigue, fever, watery diarrhea, and a cough with sputum production for the past three weeks. His past medical history was significant for recurrent bouts
Case History A 24 year old male presented with fatigue, fever, watery diarrhea, and a cough with sputum production for the past three weeks. His past medical history was significant for recurrent bouts
Mucosal immunity Reddy April Deveshni Reddy Allergy Meeting 13 April 2012
 Deveshni Reddy Allergy Meeting 13 April First recorded by Hippocrates over 2000 years ago. 1921: Prausnitz and Kustner demonstrated that substance responsible for Kustner s fish allergy was present in
Deveshni Reddy Allergy Meeting 13 April First recorded by Hippocrates over 2000 years ago. 1921: Prausnitz and Kustner demonstrated that substance responsible for Kustner s fish allergy was present in
Small intestine. Small intestine
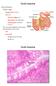 General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
General features Tubular organ longest part; 5-6 m most of chemical digestion absorption of nutrients reabsorption of H2O occurs. Two structural features; maximize the lumenal surface area villi microvilli
Quantification of enterochromaffin cells with
 Archives of Disease in Childhood, 1984, 59, 523-527 Quantification of enterochromaffin cells with serotonin immunoreactivity in the duodenal mucosa in coeliac disease E E WHEELER AND D N CHALLACOMBE Somerset
Archives of Disease in Childhood, 1984, 59, 523-527 Quantification of enterochromaffin cells with serotonin immunoreactivity in the duodenal mucosa in coeliac disease E E WHEELER AND D N CHALLACOMBE Somerset
Confirming persistence of gluten intolerance in
 Archives of Disease in Childhood, 1975, 50, 259. Confirming persistence of gluten intolerance in children diagnosed as having coeliac disease in infancy Usefulness of onehour blood xylose test C. J. ROLLES,
Archives of Disease in Childhood, 1975, 50, 259. Confirming persistence of gluten intolerance in children diagnosed as having coeliac disease in infancy Usefulness of onehour blood xylose test C. J. ROLLES,
Lecture 2. Immunoglobulin
 Lecture 2 Immunoglobulin Objectives; Define secretary IgA Describe structure & functions of IgM Compare the antigenic receptor of B lymphocyte Assess the role of IgE in Atopy Distinguish between Isotype,
Lecture 2 Immunoglobulin Objectives; Define secretary IgA Describe structure & functions of IgM Compare the antigenic receptor of B lymphocyte Assess the role of IgE in Atopy Distinguish between Isotype,
Three-dimensional structure of the human small intestinal mucosa in health and disease
 Gut, 1969, 10, 6-12 Three-dimensional structure of the human small intestinal mucosa in health and disease C. A. LOEHRY AND B. CREAMER From the Gastrointestinal Laboratory, St Thomas' Hospital, London
Gut, 1969, 10, 6-12 Three-dimensional structure of the human small intestinal mucosa in health and disease C. A. LOEHRY AND B. CREAMER From the Gastrointestinal Laboratory, St Thomas' Hospital, London
Lecture 2. Immunoglobulin
 Lecture 2 Immunoglobulin Objectives; To study the secretary IgA To know the structure & functions of IgM The antigenic receptor of B lymphocyte The role of IgE in Atopy The difference between Isotype,
Lecture 2 Immunoglobulin Objectives; To study the secretary IgA To know the structure & functions of IgM The antigenic receptor of B lymphocyte The role of IgE in Atopy The difference between Isotype,
Timing of infiltration of T lymphocytes induced by gluten into the small intestine in coeliac disease
 J Clin Pathol 1987;40:741-745 Timing of infiltration of T lymphocytes induced by gluten into the small intestine in coeliac disease A R FREEDMAN,* J C MACARTNEY,t J M NELUFER,* P J CICLITIRA* From the
J Clin Pathol 1987;40:741-745 Timing of infiltration of T lymphocytes induced by gluten into the small intestine in coeliac disease A R FREEDMAN,* J C MACARTNEY,t J M NELUFER,* P J CICLITIRA* From the
Collagenous colitis associated with small intestinal villous atrophy
 (Gut, 1986, 27, 1394-1398 Case report Collagenous colitis associated with small intestinal villous atrophy I HAMILTON, S SANDERS, D HOPWOOD, AND I A D BOUCHIER From the University Department of Medicine
(Gut, 1986, 27, 1394-1398 Case report Collagenous colitis associated with small intestinal villous atrophy I HAMILTON, S SANDERS, D HOPWOOD, AND I A D BOUCHIER From the University Department of Medicine
ANTIBODIES Jiri Mestecky, M.D., Ph.D. - Lecturer
 ANTIBODIES Jiri Mestecky, M.D., Ph.D. - Lecturer Distribution in body fluids: secretions plasma (serum), tears, saliva, milk, genitourinary, and intestinal Cells producing antibodies and their tissue distribution:
ANTIBODIES Jiri Mestecky, M.D., Ph.D. - Lecturer Distribution in body fluids: secretions plasma (serum), tears, saliva, milk, genitourinary, and intestinal Cells producing antibodies and their tissue distribution:
Human Cytomegalovirus
 JOURNAL OF CLINICAL MICROBIOLOGY, Oct. 1975, p. 332-336 Copyright ) 1975 American Society for Microbiology Vol. 2, No. 4 Printed in U.S.A. Demonstration of Immunoglobulin G Receptors Induced by Human Cytomegalovirus
JOURNAL OF CLINICAL MICROBIOLOGY, Oct. 1975, p. 332-336 Copyright ) 1975 American Society for Microbiology Vol. 2, No. 4 Printed in U.S.A. Demonstration of Immunoglobulin G Receptors Induced by Human Cytomegalovirus
Longitudinal Studies of Neutralizing Antibody Responses to Rotavirus in Stools and Sera of Children following Severe Rotavirus Gastroenteritis
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 1998, p. 897 901 Vol. 5, No. 6 1071-412X/98/$04.00 0 Copyright 1998, American Society for Microbiology. All Rights Reserved. Longitudinal Studies of
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 1998, p. 897 901 Vol. 5, No. 6 1071-412X/98/$04.00 0 Copyright 1998, American Society for Microbiology. All Rights Reserved. Longitudinal Studies of
(b) Stomach s function 1. Dilution of food materials 2. Acidification of food (absorption of dietary Fe in small intestine) 3. Partial chemical digest
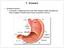 (1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
(1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
Refractory celiac disease (RCD) KASSEM BARADA LEBANESE SOCIETY OF GASTROENTEROLOGY NOVEMBER, 2014
 Refractory celiac disease (RCD) KASSEM BARADA LEBANESE SOCIETY OF GASTROENTEROLOGY NOVEMBER, 2014 Case scenario (1) A 49 year woman presents with intermittent watery diarrhea and bloating of two years
Refractory celiac disease (RCD) KASSEM BARADA LEBANESE SOCIETY OF GASTROENTEROLOGY NOVEMBER, 2014 Case scenario (1) A 49 year woman presents with intermittent watery diarrhea and bloating of two years
Light and electron microscopical studies of focal glomerular sclerosis
 J. clin. Path., 1971, 24, 846-850 Light and electron microscopical studies of focal glomerular sclerosis A. H. NAGI, F. ALEXANDER, AND R. LANNIGAN From the Department of Pathology, Queen's University of
J. clin. Path., 1971, 24, 846-850 Light and electron microscopical studies of focal glomerular sclerosis A. H. NAGI, F. ALEXANDER, AND R. LANNIGAN From the Department of Pathology, Queen's University of
Malignant histiocytosis of the intestine: the early histological lesion
 Gut, 1980, 21, 381-386 Malignant histiocytosis of the intestine: the early histological lesion P ISAACSON From the Department of Pathology, Faculty of Medicine, Southampton University Hospital, Southampton
Gut, 1980, 21, 381-386 Malignant histiocytosis of the intestine: the early histological lesion P ISAACSON From the Department of Pathology, Faculty of Medicine, Southampton University Hospital, Southampton
Distribution of Immunoglobulin-Containing Cells in Normal and Neonatally Thymectomized Rats
 INFECTION AND IMMUNITY, Nov. 1983, p. 668-674 Vol. 42, No. 2 0019-9567/83/110668-07$02.00/0 Copyright 1983, American Society for Microbiology Distribution of Immunoglobulin-Containing Cells in Normal and
INFECTION AND IMMUNITY, Nov. 1983, p. 668-674 Vol. 42, No. 2 0019-9567/83/110668-07$02.00/0 Copyright 1983, American Society for Microbiology Distribution of Immunoglobulin-Containing Cells in Normal and
Differentiation of Cytomegalovirus Antigens by Their Reactivity with Various Classes of Human Antibodies in the Indirect Fluorescent Antibody Test
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 1980, p. 88-93 0095-1 137/80/01-0088/06$02.00/0 Vol. 11, No. 1 Differentiation of Cytomegalovirus Antigens by Their Reactivity with Various Classes of Human Antibodies
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 1980, p. 88-93 0095-1 137/80/01-0088/06$02.00/0 Vol. 11, No. 1 Differentiation of Cytomegalovirus Antigens by Their Reactivity with Various Classes of Human Antibodies
Quantitation of intraepithelial lymphocytes
 Gut, 1971, 12, 988-994 Quantitation of intraepithelial lymphocytes inhuman jejunum ANNE FERGUSON AND D. MURRAY From the University Departments ofbacteriology and Immunology (Western Infirmary) and Pathology
Gut, 1971, 12, 988-994 Quantitation of intraepithelial lymphocytes inhuman jejunum ANNE FERGUSON AND D. MURRAY From the University Departments ofbacteriology and Immunology (Western Infirmary) and Pathology
Malignancy in relatives of patients with coeliac disease
 Brit. J. prev. soc. Med. (1976), 30, 17-21 Malignancy in relatives of patients with coeliac disease P. L. STOKES, PATRICIA PRIOR, T. M. SORAHAN, R. J. McWALTER* J. A. H. WATERHOUSE, AND W. T. COOKE The
Brit. J. prev. soc. Med. (1976), 30, 17-21 Malignancy in relatives of patients with coeliac disease P. L. STOKES, PATRICIA PRIOR, T. M. SORAHAN, R. J. McWALTER* J. A. H. WATERHOUSE, AND W. T. COOKE The
Astrovirus-associated gastroenteritis in children
 Journal of Clinical Pathology, 1978, 31, 939-943 Astrovirus-associated gastroenteritis in children C. R. ASHLEY, E. 0. CAUL, AND W. K. PAVER1 From the Public Health Laboratory, Myrtle Road, Bristol BS2
Journal of Clinical Pathology, 1978, 31, 939-943 Astrovirus-associated gastroenteritis in children C. R. ASHLEY, E. 0. CAUL, AND W. K. PAVER1 From the Public Health Laboratory, Myrtle Road, Bristol BS2
Laboratory Methods for Diagnosing Celiac Disease. Vijay Kumar, PhD, FACB IMMCO Diagnostics, Inc. Buffalo, NY
 Laboratory Methods for Diagnosing Celiac Disease Vijay Kumar, PhD, FACB IMMCO Diagnostics, Inc. Buffalo, NY Prevalence of Celiac Disease Group With Symptoms Adults Children Associated Symptoms Chronic
Laboratory Methods for Diagnosing Celiac Disease Vijay Kumar, PhD, FACB IMMCO Diagnostics, Inc. Buffalo, NY Prevalence of Celiac Disease Group With Symptoms Adults Children Associated Symptoms Chronic
COMMENTS DEFICIENCY OF IMMUNOGLOBULIN A IN INTESTINAL DISEASE. Characteristics of IgA Antibodies
 GASTROENTEROLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 54, No. 2 Printed in U.S.A. COMMENTS R eaders are invited to contribute Comments. If found suitable, they will be published promptly,
GASTROENTEROLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 54, No. 2 Printed in U.S.A. COMMENTS R eaders are invited to contribute Comments. If found suitable, they will be published promptly,
LECTURE: 21. Title IMMUNOGLOBULINS FUNCTIONS & THEIR RECEPTORS LEARNING OBJECTIVES:
 LECTURE: 21 Title IMMUNOGLOBULINS FUNCTIONS & THEIR RECEPTORS LEARNING OBJECTIVES: The student should be able to: Determine predominant immunoglobulin isotypes in serum. Determine the predominant immunoglobulin
LECTURE: 21 Title IMMUNOGLOBULINS FUNCTIONS & THEIR RECEPTORS LEARNING OBJECTIVES: The student should be able to: Determine predominant immunoglobulin isotypes in serum. Determine the predominant immunoglobulin
EVIDENCE THAT ACTIVATED MUCOSAL T CELLS PLAY A ROLE IN THE PATHOGENESIS OF ENTEROPATHY IN HUMAN SMALL INTESTINE
 Published Online: 1 April, 1988 Supp Info: http://doi.org/10.1084/jem.167.4.1341 Downloaded from jem.rupress.org on September 23, 2018 EVIDENCE THAT ACTIVATED MUCOSAL T CELLS PLAY A ROLE IN THE PATHOGENESIS
Published Online: 1 April, 1988 Supp Info: http://doi.org/10.1084/jem.167.4.1341 Downloaded from jem.rupress.org on September 23, 2018 EVIDENCE THAT ACTIVATED MUCOSAL T CELLS PLAY A ROLE IN THE PATHOGENESIS
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces
 Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
Dr Nadine Gravett School of Anatomical Sciences Room 2B10B
 Dr Nadine Gravett School of Anatomical Sciences Room 2B10B Nadine.Gravett@wits.ac.za Oral cavity Mechanical breakdown Formation of bolus Oesophagus Conduit from mouth to stomach Stomach Digestion Temporary
Dr Nadine Gravett School of Anatomical Sciences Room 2B10B Nadine.Gravett@wits.ac.za Oral cavity Mechanical breakdown Formation of bolus Oesophagus Conduit from mouth to stomach Stomach Digestion Temporary
Gastricsin in the benign and malignant prostate
 J Clin Pathol 1985;38:639-643 Gastricsin in the benign and malignant prostate WA REID,* CN LIDDLE,t J SVASTI,t J KAY From the *Department ofpathology, University of Leeds, Leeds, the tdepartment ofpathology,
J Clin Pathol 1985;38:639-643 Gastricsin in the benign and malignant prostate WA REID,* CN LIDDLE,t J SVASTI,t J KAY From the *Department ofpathology, University of Leeds, Leeds, the tdepartment ofpathology,
PATHOLOGY OF NON NEOPLASTIC LESIONS OF THE UPPER GASTROINTESTINAL TRACT.
 PATHOLOGY OF NON NEOPLASTIC LESIONS OF THE UPPER GASTROINTESTINAL TRACT. OESOPHAGEAL LESIONS OESOPHAGITIS AND OTHER NON NEOPLASTIC DISORDERS Corrosive Gastroesophageal reflux (GERD), Pills, Acid intake,
PATHOLOGY OF NON NEOPLASTIC LESIONS OF THE UPPER GASTROINTESTINAL TRACT. OESOPHAGEAL LESIONS OESOPHAGITIS AND OTHER NON NEOPLASTIC DISORDERS Corrosive Gastroesophageal reflux (GERD), Pills, Acid intake,
HDF Case CRYPTOSPORIDIOSE
 HDF Case 986949 CRYPTOSPORIDIOSE 45 yo male with severe diarrhea. Known HIV positive. Endoscopic biopsy of duodenum, the colon and ileum. EXUDATIVE CHANGES GRANULAR BASOPHILIC BODIES Colonic biopsy shows
HDF Case 986949 CRYPTOSPORIDIOSE 45 yo male with severe diarrhea. Known HIV positive. Endoscopic biopsy of duodenum, the colon and ileum. EXUDATIVE CHANGES GRANULAR BASOPHILIC BODIES Colonic biopsy shows
Adaptive Immunity: Specific Defenses of the Host
 17 Adaptive Immunity: Specific Defenses of the Host SLOs Differentiate between innate and adaptive immunity, and humoral and cellular immunity. Define antigen, epitope, and hapten. Explain the function
17 Adaptive Immunity: Specific Defenses of the Host SLOs Differentiate between innate and adaptive immunity, and humoral and cellular immunity. Define antigen, epitope, and hapten. Explain the function
An evaluation of two new haemagglutination tests for the rapid diagnosis of autoimmune thyroid diseases
 Journal of Clinical Pathology, 1978, 31, 1147-115 An evaluation of two new haemagglutination tests for the rapid diagnosis of autoimmune thyroid diseases I. CAYZER, S. R. CHALMERS, D. DONIACH', AND G.
Journal of Clinical Pathology, 1978, 31, 1147-115 An evaluation of two new haemagglutination tests for the rapid diagnosis of autoimmune thyroid diseases I. CAYZER, S. R. CHALMERS, D. DONIACH', AND G.
General Structure of Digestive Tract
 Dr. Nabil Khouri General Structure of Digestive Tract Common Characteristics: Hollow tube composed of a lumen whose diameter varies. Surrounded by a wall made up of 4 principal layers: Mucosa Epithelial
Dr. Nabil Khouri General Structure of Digestive Tract Common Characteristics: Hollow tube composed of a lumen whose diameter varies. Surrounded by a wall made up of 4 principal layers: Mucosa Epithelial
Immunoglobulin A and M Levels in Premature
 Archives of Disease in Childhood, 1971, 46, 671. Immunoglobulin A and M Levels in Premature Infants with Gastroenteritis P. PANAYOTOU, C. PAPADATOS, G. PAPAVANGLOU, D. ALXIOU, A. SKARDOUTSOU, and. KOURA
Archives of Disease in Childhood, 1971, 46, 671. Immunoglobulin A and M Levels in Premature Infants with Gastroenteritis P. PANAYOTOU, C. PAPADATOS, G. PAPAVANGLOU, D. ALXIOU, A. SKARDOUTSOU, and. KOURA
and Grey, 1972) there is an increased risk of cancer. The role of secretory to our knowledge, been studied but in
 Br. J. Cancer (1973) 27, 269 LOCAL IMMUNOGLOBULIN PRODUCTION IN BREAST CANCER M. MAUREEN ROBERTS, ELEANOR M. BASS, I. W. J. WALLACE AND ANN STEVENSON From the Departnment of Clinical Surgery, University
Br. J. Cancer (1973) 27, 269 LOCAL IMMUNOGLOBULIN PRODUCTION IN BREAST CANCER M. MAUREEN ROBERTS, ELEANOR M. BASS, I. W. J. WALLACE AND ANN STEVENSON From the Departnment of Clinical Surgery, University
Regulation of human gut B lymphocytes by T lymphocytes
 Regulation of human gut B lymphocytes by T lymphocytes R CLANCY, A CRIPPS, AND HEATHER CHIPCHASE Gut, 1984, 25, 47-51 From the Department of Pathology, Faculty of Medicine, The University of Newcastle,
Regulation of human gut B lymphocytes by T lymphocytes R CLANCY, A CRIPPS, AND HEATHER CHIPCHASE Gut, 1984, 25, 47-51 From the Department of Pathology, Faculty of Medicine, The University of Newcastle,
Immunofluorescent localization of Tamm-Horsfall mucoprotein in human kidney
 J. clin. Path. (1969), 22,334-339 Immunofluorescent localization of Tamm-Horsfall mucoprotein in human kidney J. K. McKENZIE1 AND E. G. McQUEEN From the Wellcome Medical Research Institute, Department
J. clin. Path. (1969), 22,334-339 Immunofluorescent localization of Tamm-Horsfall mucoprotein in human kidney J. K. McKENZIE1 AND E. G. McQUEEN From the Wellcome Medical Research Institute, Department
Simplex and Varicella-Zoster Virus Antigens in Vesicular
 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1980, p. 651-655 0095-1137/80/11-0651/05$02.00/0 Vol. 12, No. 5 Direct Immunofluorescence Staining for Detection of Herpes Simplex and Varicella-Zoster Virus Antigens
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1980, p. 651-655 0095-1137/80/11-0651/05$02.00/0 Vol. 12, No. 5 Direct Immunofluorescence Staining for Detection of Herpes Simplex and Varicella-Zoster Virus Antigens
Scanning Electron Microscopy of the Small Intestine of a Normal Unsuckled Calf and a Calf with Enteric Colibacillosis
 Vet. Pathol. 15; 400-406 (1978) Scanning Electron Microscopy of the Small Intestine of a Normal Unsuckled Calf and a Calf with Enteric Colibacillosis G. R. PEARSON. E. F. LOGAN and G. P. BRENNAN Departmcnt
Vet. Pathol. 15; 400-406 (1978) Scanning Electron Microscopy of the Small Intestine of a Normal Unsuckled Calf and a Calf with Enteric Colibacillosis G. R. PEARSON. E. F. LOGAN and G. P. BRENNAN Departmcnt
CLINICAL REVIEW. and small bowel carcinoma SIMON KENWRIGHT* M.R.C.P.
 Postgraduate Medical Journal (November 1972) 48, 673-677. Coeliac disease CLINICAL REVIEW and small bowel carcinoma SIMON KENWRIGHT* M.R.C.P. Department of Gastro-enterology, Frenchay Hospital, Bristol
Postgraduate Medical Journal (November 1972) 48, 673-677. Coeliac disease CLINICAL REVIEW and small bowel carcinoma SIMON KENWRIGHT* M.R.C.P. Department of Gastro-enterology, Frenchay Hospital, Bristol
THE USE OF HUMAN SKIN FOR THE DETECTION OF ANTI- EPITHELIAL AUTOANTIBODIES A DIAGNOSTIC AND PROGNOSTIC TEST*
 Tlia JOUStNAL OF INVF5T1WTiVF DEBM.,TLGY Copyright 1969 by The Williams & Wilkins Co. Vol. 53, No. 6 Printed 'in U.S.A. THE USE OF HUMAN SKIN FOR THE DETECTION OF ANTI- EPITHELIAL AUTOANTIBODIES A DIAGNOSTIC
Tlia JOUStNAL OF INVF5T1WTiVF DEBM.,TLGY Copyright 1969 by The Williams & Wilkins Co. Vol. 53, No. 6 Printed 'in U.S.A. THE USE OF HUMAN SKIN FOR THE DETECTION OF ANTI- EPITHELIAL AUTOANTIBODIES A DIAGNOSTIC
Proteolytic Degradation of Exocrine and Serum Immunoglobulins
 Proteolytic Degradation of Exocrine and Serum Immunoglobulins WILLIAM R. BROWN, RiCHARD W. NEWCOMB, and KIMISHIGE ISHIZAKA From the Department of Medicine, University of Colorado Medical Center, and Children's
Proteolytic Degradation of Exocrine and Serum Immunoglobulins WILLIAM R. BROWN, RiCHARD W. NEWCOMB, and KIMISHIGE ISHIZAKA From the Department of Medicine, University of Colorado Medical Center, and Children's
however, and the present communication is concerned with some of
 THE AGGLUTINATION OF HUMAN ERYTHROCYTES MODIFIED BY TREATMENT WITH NEWCASTLE DISEASE AND INFLUENZA VIRUS' ALFRED L. FLORMAN' Pediatric Service and Division of Bacteriology, The Mount Sinai Hospital, New
THE AGGLUTINATION OF HUMAN ERYTHROCYTES MODIFIED BY TREATMENT WITH NEWCASTLE DISEASE AND INFLUENZA VIRUS' ALFRED L. FLORMAN' Pediatric Service and Division of Bacteriology, The Mount Sinai Hospital, New
AUTOIMMUNE RESPONSES TO HUMAN TUMOUR ANTIGENS
 510 AUTOIMMUNE RESPONSES TO HUMAN TUMOUR ANTIGENS MADELINE HODKINSON* AND G. TAYLOR From the Immunology Department, Royal Infirmary, Manchester Received for publication May 14, 1969 THE most convincing
510 AUTOIMMUNE RESPONSES TO HUMAN TUMOUR ANTIGENS MADELINE HODKINSON* AND G. TAYLOR From the Immunology Department, Royal Infirmary, Manchester Received for publication May 14, 1969 THE most convincing
Small intestinal mucosal patterns of coeliac disease and idiopathic steatorrhoea seen in other situations
 Gut, 1964, 5, 51 Small intestinal mucosal patterns of coeliac disease and idiopathic steatorrhoea seen in other situations R. R. W. TOWNLEY, M. H. CASS, AND CHARLOTTE M. ANDERSON From the Gastroenterological
Gut, 1964, 5, 51 Small intestinal mucosal patterns of coeliac disease and idiopathic steatorrhoea seen in other situations R. R. W. TOWNLEY, M. H. CASS, AND CHARLOTTE M. ANDERSON From the Gastroenterological
Gastroporesis or Leaky Gut
 Dr Wendy Wells 8595 E Bell Rd D101 Scottsdale, AZ 85260 (480) 607-0299 Gastroporesis or Leaky Gut From the mouth to anus is an enfolding of our skin. As you know, our skin contains a layer of cells and
Dr Wendy Wells 8595 E Bell Rd D101 Scottsdale, AZ 85260 (480) 607-0299 Gastroporesis or Leaky Gut From the mouth to anus is an enfolding of our skin. As you know, our skin contains a layer of cells and
Topics and aims. Introduction. Metabolism and Excretion
 Topics and aims Introduction This section contains instructions that are applicable to all material, irrespective of your specific course. Please take note and make sure to comply. Failure to comply could
Topics and aims Introduction This section contains instructions that are applicable to all material, irrespective of your specific course. Please take note and make sure to comply. Failure to comply could
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Clinical, microbiological, and immunological studies
 Gut, 1972, 13, 441-449 Clinical, microbiological, and immunological studies in patients with immunoglobulin deficiencies and gastrointestinal disorders' W. R. BROWN2, D. BUTTERFIELD, D. SAVAGE, AND T.
Gut, 1972, 13, 441-449 Clinical, microbiological, and immunological studies in patients with immunoglobulin deficiencies and gastrointestinal disorders' W. R. BROWN2, D. BUTTERFIELD, D. SAVAGE, AND T.
A Practical Approach to Small Bowel Biopsies: All that flattens is not sprue
 A Practical Approach to Small Bowel Biopsies: All that flattens is not sprue UCSF Liver and Gastrointestinal Pathology Update Sept. 4, 2009 How to Go Wrong When Evaluating Small Bowel Biopsies, Based on
A Practical Approach to Small Bowel Biopsies: All that flattens is not sprue UCSF Liver and Gastrointestinal Pathology Update Sept. 4, 2009 How to Go Wrong When Evaluating Small Bowel Biopsies, Based on
High incidence of corneal epithelium antibodies in
 British Journal of Ophthalmology, 1988, 72, 921-925 High incidence of corneal epithelium antibodies in Fuchs' heterochromic cyclitis E LA HEY, G S BAARSMA, A ROTHOVA, L BROERSMA, R VAN DER GAAG, AND A
British Journal of Ophthalmology, 1988, 72, 921-925 High incidence of corneal epithelium antibodies in Fuchs' heterochromic cyclitis E LA HEY, G S BAARSMA, A ROTHOVA, L BROERSMA, R VAN DER GAAG, AND A
The effect of coeliac disease upon bile salts
 Gut, 1973, 14, 24-2 T. S. LOW-BEER,1 K. W. HEATON, E. W. POMARE, AND A. E. READ From the University Department of Medicine, Bristol Royal Infirmary SUMMARY The size and composition of the bile salt pool
Gut, 1973, 14, 24-2 T. S. LOW-BEER,1 K. W. HEATON, E. W. POMARE, AND A. E. READ From the University Department of Medicine, Bristol Royal Infirmary SUMMARY The size and composition of the bile salt pool
Antigen handling by the gut
 Archives of Disease in Childhood, 1978, 53, 527-531 Antigen handling by the gut The epithelial surface of the gastrointestinal tract represents an extensive surface area exposed to a hostile intraluminal
Archives of Disease in Childhood, 1978, 53, 527-531 Antigen handling by the gut The epithelial surface of the gastrointestinal tract represents an extensive surface area exposed to a hostile intraluminal
Citation Acta Medica Nagasakiensia. 1992, 37
 NAOSITE: Nagasaki University's Ac Title Author(s) A Study on the Expression of EGFR a Content in the Stomach Cancer Tissu Nakazaki Takayuki Citation Acta Medica Nagasakiensia. 1992 37 Issue Date 1992-12-25
NAOSITE: Nagasaki University's Ac Title Author(s) A Study on the Expression of EGFR a Content in the Stomach Cancer Tissu Nakazaki Takayuki Citation Acta Medica Nagasakiensia. 1992 37 Issue Date 1992-12-25
embedded tissue-sections has the advantage of permanence of the sections and is more suitable for
 Gut, 1980, 21, 941-947 Immunoglobulin containing cells in inflammatory bowel disease of the colon: a morphometric and immunohistochemical study P C M ROSEKRANS,* C J L M MEIJER, A M VAN DER WAL, C J CORNELISSE,
Gut, 1980, 21, 941-947 Immunoglobulin containing cells in inflammatory bowel disease of the colon: a morphometric and immunohistochemical study P C M ROSEKRANS,* C J L M MEIJER, A M VAN DER WAL, C J CORNELISSE,
Definition. Celiac disease is an immune-mediated enteropathy caused by a permanent sensitivity to gluten in genetically susceptible individuals.
 Definition 1 Definition Celiac disease is an immune-mediated enteropathy caused by a permanent sensitivity to gluten in genetically susceptible individuals. It occurs in symptomatic subjects with gastrointestinal
Definition 1 Definition Celiac disease is an immune-mediated enteropathy caused by a permanent sensitivity to gluten in genetically susceptible individuals. It occurs in symptomatic subjects with gastrointestinal
ROLE OF THE GUT BACTERIA
 ROLE OF THE GUT BACTERIA Our Good Bacteria In a perfect world, we would all have a proper ratio of good bacteria And what could this proper ratio do for us? The knowledge of the connections between our
ROLE OF THE GUT BACTERIA Our Good Bacteria In a perfect world, we would all have a proper ratio of good bacteria And what could this proper ratio do for us? The knowledge of the connections between our
A total of 111 subjects admitted consecutively in a. of diseases associated with malabsorption. Informed
 J Clin Pathol 1981;34:174-178 One-hour serum ylose as an absorption test in the tropics PG HILL, IN ROSS,* R JACOB, S JYOTHEESWARAN, AND VI MATHAN From the Wellcome Research Unit and The Gastroenterology
J Clin Pathol 1981;34:174-178 One-hour serum ylose as an absorption test in the tropics PG HILL, IN ROSS,* R JACOB, S JYOTHEESWARAN, AND VI MATHAN From the Wellcome Research Unit and The Gastroenterology
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
lamina propria lymphocytes
 Gut, 1984, 25, 649-655 In vitro immunoglobulin synthesis by human intestinal lamina propria lymphocytes P A DREW, J T LA BROOY, AND D J C SHEARMAN From the Department of Medicine, University ofadelaide,
Gut, 1984, 25, 649-655 In vitro immunoglobulin synthesis by human intestinal lamina propria lymphocytes P A DREW, J T LA BROOY, AND D J C SHEARMAN From the Department of Medicine, University ofadelaide,
SUBDIVISIONS OF THE MUCOSA Distinct features of type I and type II mucosal surfaces
 Alessandra Pernis P&S 9-435 X53763 abp1@columbia.edu CHALLENGES FACED BY THE MUCOSAL SYSTEM SPECIALIZATION OF CELLS INVOLVED IN MUCOSAL IMMUNITY ORGANIZATION OF THE MUCOSAL IMMUNE SYSTEM CLINICAL IMPLICATIONS
Alessandra Pernis P&S 9-435 X53763 abp1@columbia.edu CHALLENGES FACED BY THE MUCOSAL SYSTEM SPECIALIZATION OF CELLS INVOLVED IN MUCOSAL IMMUNITY ORGANIZATION OF THE MUCOSAL IMMUNE SYSTEM CLINICAL IMPLICATIONS
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Flow Cytometry. Hanan Jafar (2017)
 1 Flow Cytometry Flow cytometry is a popular laser-based technology to analyze the characteristics of cells or particles. It is predominantly used to measure fluorescence intensity produced by fluorescent-labeled
1 Flow Cytometry Flow cytometry is a popular laser-based technology to analyze the characteristics of cells or particles. It is predominantly used to measure fluorescence intensity produced by fluorescent-labeled
(Adams 8c Purves 1958), or LATS-protector (LATS-P) (Adams 8c Kennedy. 1967). The failure of the McKenzie (1958) mouse bioassay to detect LATS in
 Department of Endocrinology, Royal Prince Alfred Hospital, and Department of Medicine, University of Sydney, Sydney, Australia THE THYROTROPHIN RECEPTOR IN HUMAN THYROID PLASMA MEMBRANES: EFFECT OF SERUM
Department of Endocrinology, Royal Prince Alfred Hospital, and Department of Medicine, University of Sydney, Sydney, Australia THE THYROTROPHIN RECEPTOR IN HUMAN THYROID PLASMA MEMBRANES: EFFECT OF SERUM
Original Contribution
 Direct Immunofluorescence Test of Skin Biopsy Samples Results of 204 Cases Kabir AN, 1 Das RK, 2 Kamal M 3 Direct immunofluorescence (DIF) test of skin and renal biopsy specimens is being done on regular
Direct Immunofluorescence Test of Skin Biopsy Samples Results of 204 Cases Kabir AN, 1 Das RK, 2 Kamal M 3 Direct immunofluorescence (DIF) test of skin and renal biopsy specimens is being done on regular
Ever wonder what s really happening on the inside?
 For Practitioners Ever wonder what s really happening on the inside? Are your patients suffering from diarrhea, constipation, bloating, gas or indigestion? Rocky Mountain Analytical is now offering Gut-Well
For Practitioners Ever wonder what s really happening on the inside? Are your patients suffering from diarrhea, constipation, bloating, gas or indigestion? Rocky Mountain Analytical is now offering Gut-Well
Localization of talin in skeletal and cardiac muscles
 Volume 200, number 1 FEBS 3591 May 1986 Localization of talin in skeletal and cardiac muscles A.M. Belkin, N.I. Zhidkova and V.E. Koteliansky* Laboratory of Molecular and Cellular Cardiology, Institute
Volume 200, number 1 FEBS 3591 May 1986 Localization of talin in skeletal and cardiac muscles A.M. Belkin, N.I. Zhidkova and V.E. Koteliansky* Laboratory of Molecular and Cellular Cardiology, Institute
Reintroduction of gluten in adults and children with treated coeliac disease
 Reintroduction of gluten in adults and children with treated coeliac disease PARVEEN J. KUMAR, D. P. O'DONOGHUE, KATHY STENSON, AND A. M. DAWSON From the Department of Gastroenterology, St. Bartholomew's
Reintroduction of gluten in adults and children with treated coeliac disease PARVEEN J. KUMAR, D. P. O'DONOGHUE, KATHY STENSON, AND A. M. DAWSON From the Department of Gastroenterology, St. Bartholomew's
A CELL-MEDIATED IMMUNE MODEL OF INFLAMMATORY BOWEL DISEASE IN THE RABBIT
 0016-5085/7501-0029$02.00/0 GASTROENTEROLOGY 75:29-33, 1978 Copyright 1978 by the American Gastroenterological Association Vol. 75, No.1 Printed in U.8A. A CELL-MEDIATED IMMUNE MODEL OF INFLAMMATORY BOWEL
0016-5085/7501-0029$02.00/0 GASTROENTEROLOGY 75:29-33, 1978 Copyright 1978 by the American Gastroenterological Association Vol. 75, No.1 Printed in U.8A. A CELL-MEDIATED IMMUNE MODEL OF INFLAMMATORY BOWEL
SAM PROVIDER TOOLKIT
 THE AMERICAN BOARD OF PATHOLOGY Maintenance of Certification (MOC) Program SAM PROVIDER TOOLKIT Developing Self-Assessment Modules (SAMs) www.abpath.org The American Board of Pathology (ABP) approves educational
THE AMERICAN BOARD OF PATHOLOGY Maintenance of Certification (MOC) Program SAM PROVIDER TOOLKIT Developing Self-Assessment Modules (SAMs) www.abpath.org The American Board of Pathology (ABP) approves educational
Low gluten diet in the treatment of adult coeliac disease: effect on jejunal morphology and serum anti-gluten antibodies
 Gut, 1988, 29, 1564-1568 Low gluten diet in the treatment of adult coeliac disease: effect on jejunal morphology and serum anti-gluten antibodies A M P MONTGOMERY, A K J GOKA, P J KUMAR, M J G FARTHING,
Gut, 1988, 29, 1564-1568 Low gluten diet in the treatment of adult coeliac disease: effect on jejunal morphology and serum anti-gluten antibodies A M P MONTGOMERY, A K J GOKA, P J KUMAR, M J G FARTHING,
VIRAL AGENTS CAUSING GASTROENTERITIS
 VIRAL AGENTS CAUSING GASTROENTERITIS VIRAL AGENTS CAUSING GASTROENTERITIS Pathogens discussed in our lectures 1. Rotavirus 2. Enteric adenoviruses 3. Caliciviruses 4. Astroviruses 5. Toroviruses Viruses
VIRAL AGENTS CAUSING GASTROENTERITIS VIRAL AGENTS CAUSING GASTROENTERITIS Pathogens discussed in our lectures 1. Rotavirus 2. Enteric adenoviruses 3. Caliciviruses 4. Astroviruses 5. Toroviruses Viruses
above 110 g per 100 ml in four patients, but the
 J. clin. Path., 1971, 24, 244-249 Diagnostic value of the serum folate assay J. FORSHAW AND LILIAN HARWOOD From the Haematology Laboratory, Sefton General Hospital, Liverpool SYNOPSIS The diagnostic value
J. clin. Path., 1971, 24, 244-249 Diagnostic value of the serum folate assay J. FORSHAW AND LILIAN HARWOOD From the Haematology Laboratory, Sefton General Hospital, Liverpool SYNOPSIS The diagnostic value
