The Extreme Effects of Not-so-minor Concussions: Chronic Traumatic Encephalopathy Literature Review
|
|
|
- Marjory Lester
- 5 years ago
- Views:
Transcription
1 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Honors Theses, University of Nebraska-Lincoln Honors Program Spring The Extreme Effects of Not-so-minor Concussions: Chronic Traumatic Encephalopathy Literature Review Alyssa Pierson University of Nebraska - Lincoln Follow this and additional works at: Part of the Medical Neurobiology Commons Pierson, Alyssa, "The Extreme Effects of Not-so-minor Concussions: Chronic Traumatic Encephalopathy Literature Review" (2019). Honors Theses, University of Nebraska-Lincoln This Thesis is brought to you for free and open access by the Honors Program at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Honors Theses, University of Nebraska-Lincoln by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln.
2 CHRONIC TRAUMATIC ENCEPHALOPATHY 1 The Extreme Effects of Not-so-minor Concussions: Chronic Traumatic Encephalopathy Literature Review Alyssa Pierson University of Nebraska- Lincoln
3 CHRONIC TRAUMATIC ENCEPHALOPATHY 2 Abstract Many of the concerns that people have with chronic traumatic encephalopathy (CTE) have already been researched for almost a century, but recently, there has been a big push in CTE research. Previous research was done on boxing and football and has now expanded to other contact sports and the military. Risk factors for CTE include repetitive head trauma, the Apoe4 allele, and age of first exposure to brain trauma. A wide range of symptoms may present with CTE, from motor impairment to suicidal ideation. It is believed that a biopsychosocial model should be used when approaching the symptoms of CTE patients. Current research is trying to determine the main pathology or pathologies of CTE and have determined that there are both macroscopic and microscopic pathologies. The only definitive cases of CTE have been determined by an autopsy, but with the use of biomarkers and advances in neuroimaging, hopefully CTE can be diagnosed in living persons and work can be done to find a treatment.
4 CHRONIC TRAUMATIC ENCEPHALOPATHY 3 Background The overall concept of chronic traumatic encephalopathy (CTE) and brain trauma dates back almost a century ago. Osnato and Gilberti (1927) are credited with the original concept that chronic neurodegeneration might occur after minor brain trauma. They studied 100 clinical cases of concussions of the brain, which was defined back then as a blow to the head with loss of consciousness with or without posttraumatic amnesia or skull fracture. Through this study, they found that several instances in which the clinical symptoms persisted, and secondary degenerative changes developed, they termed this traumatic encephalitis. The following year, in 1928, Martland studied the effects of repetitive brain trauma in boxers, he called it punchdrunk syndrome. Boxers did not like the term punch-drunk syndrome so it was later named dementia pugilistica, which described a physical-psychic syndrome that accumulated over a lengthy boxing career. (Tharmaratnam et al., 2018). In 1973, Corsellis, Bruton and Freeman- Browne described the clinical and neurological features of 15 retired boxers. The study conducted in 1973 introduced a relatively stereotyped pattern of structural brain abnormalities. These abnormalities included: cerebral atrophy, enlargement of the lateral and third ventricles, thinning of the corpus callosum, cavum septum pellucidum with fenestrations and cerebellar scarring (Corsellis, Bruton, & Freeman, 1973). Currently, many people follow the guidelines set by Omalu and colleagues who were the first to demonstrate the CTE phenomenon in 2005 through a series of case reports based on autopsy findings of deceased players brains. CTE is currently defined as brain trauma that is either repetitive, episodic, or a single precipitating event, leading to progressive brain neurodegeneration (Tharmaratnam et al., 2018).
5 CHRONIC TRAUMATIC ENCEPHALOPATHY 4 Epidemiology and Incidence Studies aiming to identify the prevalence of CTE in football players have shown remarkable, indisputable findings. In the largest ever case series of CTE, involving 202 deceased former football players, 87% of them had signs of CTE, including 99% ex-nfl players. The magnitude of the disease burden was correlated to level of play, with high school athletes having mild CTE, and NFL players showcasing the most severe form of CTE. In this same study, it was found that Ex-NFL players were also, if over the age of 50, five times more likely to be diagnosed with dementia than the national population averages (Tharmaratnam et al., 2018). The exact incidence of CTE is still unknown but it is believed to be attributed to duration of play, position, sport, age of first exposure, and genetics. Risk Factors The primary risk factor for CTE is believed to be repetitive head trauma, which is very common in football players, especially NFL football players. In a study done on NFL football players, 61% of them have experienced at least 1 concussion within their careers; but, remember that brain trauma without a concussion can also lead to CTE. In general, most people recover from a single head impact with no CTE, however, experiencing 3 or more concussions is correlated to an increased risk of prolonged symptoms. In a sample of 1721 cases of contact sport athletes, CTE was reported to be found in 32% of the cases, whereas no cases of CTE was found in patients with no prior brain trauma in the control group. This suggests that participating in contact sports is a predisposing risk factor in the development of CTE (Tharmaratnam et al., 2018). Research has shown that there may be a genetic susceptibility risk factor for CTE. There is an apolipoprotein (Apoe), a 34-kDa glycosylated protein, that is implicated as the primary
6 CHRONIC TRAUMATIC ENCEPHALOPATHY 5 source of cholesterol transport in the brain for neuronal repair. Individuals possessing Apoe4 allele have been shown to have an increased risk of poor functional outcome. Compared to other isoforms, the Apoe4 variant has been associated with longer recovery times from neurotrauma, increased injury severity, and greater cognitive deficits in football players (Tharmaratnam et al., 2018). Another risk factor is age of first exposure (AFE) which is associated with neurological and psychiatric dysfunction later in life. Ages 9-12 have been identified as key periods corresponding to peak gray and white matter volumes and neurological maturation of the hippocampus and amygdala. AFE <12 was associated with more than double the risk of cognitive impairment later in life, which included impairments in correlates of apathy and depression, but not cognition (Tharmaratnam et al., 2018). Mechanisms of Injury The literature to date is not sufficient to determine whether the development of CTE is associated with head injury frequency or head injury type (Aldag et al., 2017). It has been demonstrated that brain trauma without concussion can also lead to CTE. Over time, tau oligomers develop into paired helical, and straight filamentous neurofibrillary tangles (NFTs), which interfere with white matter tracts in the brain and cause signaling and communication abnormalities through denervation injury (Tharmaratnam et al., 2018). The increased bloodbarrier permeability is due to the inflammatory state which activates cytokine and chemokines, allowing unwanted chemicals and hormones in. Symptoms Clinical symptoms associated with CTE include chronic psychiatric illness, headache, cognitive dysfunction, motor impairment, irritability, aggression, episodic memory impairment,
7 CHRONIC TRAUMATIC ENCEPHALOPATHY 6 suicidal ideation, rampant mood fluctuations and depression (Aldag et al., 2017). Usually, behavioral and mood changes appear before the onset of neurocognitive decline; with the most commonly described behavioral features including explosivity, impulsivity, and hopelessness followed by suicidality and anxiety (Asken et al., 2016). In 2014, Montenigro and colleagues coined the term traumatic encephalopathy syndrome (TES) along with various TES subtypes. They included five general criteria derived from calculated prevalence of cognitive and emotional deficits across reported cases of CTE, three core clinical features, and nine supportive features (Asken et al., 2016). The most recent rendition of TES has different requirement and was established by Reams and colleagues. This new criterion requires persistence of symptoms for at least two years, a history of head trauma exposure, delayed onset following head trauma, progressive course, and formal neuropsychological testing corroborating self- or observer-report of cognitive dysfunction in the executive, visuospatial, memory, and/or language domain(s) (Asken et al., 2016). Symptoms do not present immediately and typically occur in midlife, usually decades after the original injury. On average, symptom onset usually occurs around 8 years after retirement, but it is unclear why this latency period exists. Some researchers believe that it is a result of tau propagation from focal to widespread areas, as a consequence of progressive axonal disruption. Pathologies The neuropathology of CTE is usually looked at in two different categories: macroscopic and microscopic. The macroscopic changes that are seen are commonly found in the advanced forms of CTE and rarely manifest in the early stages. Macroscopically, there may be cerebral atrophy, reduced brain mass, enlarged lateral and third ventricles, cavum septum pellucidum, pale locus coeruleus, and diencephalon atrophy (Tharmaratnam et al., 2018). The microscopic
8 CHRONIC TRAUMATIC ENCEPHALOPATHY 7 pathologies are more debated and are multifactorial, they include tau protein aggregation, transactive response DNA-binding protein (TDP-43), beta-amyloid plaque (Ab), and neuroinflammation. Tau progression is classically found in the perivascular spaces around the blood vessels, deep in the cortical sulci (Tharmaratnam et al., 2018). The perivascular accumulation of hyperphosphorylated tau (p-tau) at the depths of cortical sulci is thought to be unique to CTE and has been proposed as a diagnostic requirement of CTE. The normal function of tau protein is to stabilize microtubules; however, aberrant p-tau causes the formation of protein aggregates and NFTs, which may contribute to the development of CTE (Lucke-Wold et al., 2014). TDP-43 functions as a transcriptional regulator in the central nervous system and this pathological feature is not unique to CTE, it is also observed in other neurodegenerative diseases (Armstrong 2009; Bosque, Boyer, & Mishra 2013). TDP-43 is a ubiquitously expressed nuclear protein and has a high binding affinity for either TG repeats in DNA or UG repeats in RNA. It is commonly found co-localized with p-tau NFTs (Tharmaratnam et al., 2018). TDP-43 bound to (UG)n elements at the 3 -UTR of tau mrna and promoted its instability, resulting in a negative regulation of tau expression (Gu et al., 2017). Phosphorylated TDP-43, previously identified as the common pathologic proteinopathy linking frontotemporal dementia (FTD) and amyotrophic lateral sclerosis (ALS) is found in more than 80% of CTE cases (McKee et al., 2016). As tau and TDP-43 deposition increases, it has been shown that there is a parallel increase in axonal pathology and loss (Stein, Alvarez, & McKee 2014). Beta-amyloid plaque may be found in a diffuse pattern in sporadic loci or none at all, so it is not a tell-tale sign of CTE. Neuroinflammation is associated with microglial and astroglial activation, potentially playing a role in long-term neurodegeneration (Faden, Wu, Stoica, & Loane 2015).
9 CHRONIC TRAUMATIC ENCEPHALOPATHY 8 There are four phenotypes that were proposed by Omalu and co-workers (Omalu et al., 2011) that were thought of as parallel pathologies. Phenotype I has sparse to frequent NFTs and neuritic threads (NTs) in the cerebral cortex and brainstem. Phenotype II also includes NFTs and NTs in the basal ganglia and cerebellum in addition to diffuse amyloid plaques. Phenotype III has a combination of moderate to frequent NFTs and NTs predominately in the brainstem with absent to sparse NFTs and NTs in the cerebral cortex and basal ganglia with none in the cerebellum. Phenotype IV is a combination of absent to sparse NFTs and NTs in the cerebral cortex, brainstem, and basal ganglia, an absence of NFTs and NTs in the cerebellum, and an absence of diffuse amyloid plaques in the cerebral cortex. Although the process begins focally, it gradually spreads to involve widespread regions of the brain including the frontal and temporal lobes, medial temporal lobe, diencephalon, and brainstem. McKee and colleagues (2013) determined that CTE could be classified into four pathological stages based on a stereotyped pattern of structural change and tau pathology. Stage I brains are grossly unremarkable, although mild enlargement of the frontal horns of the lateral ventricles may be found occasionally. Microscopically, there are isolated perivascular foci of p- tau NFTs, neurophil threads and astrocytic tangles. This is most commonly seen at the depths of cerebral sulci of the superior, dorsolateral, lateral, and inferior frontal cortices. P-tau-positive astrocytes are usually found in the subpial region directly overlying the perivascular foci. In stage II, macroscopic abnormalities are found in approximately one-half of the cases, including mild enlargement of the frontal horns of the lateral ventricles and third ventricle, a cavum septum, and pallor of the locus coeruleus and substantia nigra. Microscopically, multiple foci of tau pathology are found at the depths of the sulci commonly in the superior, dorsolateral, lateral and inferior frontal, the anterior inferior and lateral temporal, inferior and superior parietal,
10 CHRONIC TRAUMATIC ENCEPHALOPATHY 9 insular and septal cortices. Deep structures such as the substantia nigra, dorsal and median raphe and thalamus show mild neurofibrillary degeneration. Beta-amyloid pathology is not found in stage II CTE. Most stage III cases of CTE show a reduction in brain weight, mild atrophy of the frontal and temporal lobes and enlargement of the lateral and third ventricles. Microscopically, NFTs are present diffusely in the frontal, temporal and parietal cortices and are most concentrated around small vessels and at the depths of sulci. The majority of cases show TDP- 43-positive neurites and inclusions in the cerebral cortex, medial temporal lobe, diencephalon and brainstem. Although most stage III cases of CTE show no beta-amyloid deposition, sparse diffuse and neuritic beta-amyloid plaques are found in approximately 13%. Brain weight in stage IV is significantly decreased, there may be marked global atrophy of the brain. There is usually pronounced atrophy of the frontal and temporal lobes, medial temporal lobe and anterior thalamus. The hypothalamic floor is thinned, mammillary bodies are darkly discolored and atrophied, and there is marked enlargement of the lateral and third ventricles. Approximately two-thirds of subjects will have septal abnormalities including cavum septum, fenestrations, or absence. Microscopically, there is widespread neuronal loss, as well as severe tau deposition, as clusters of glial tangles and small NFTs in a patchy irregular distribution throughout frontal, temporal and parietal cortices. NFTs are found widely distributed throughout the hippocampal formation including the dentate gyrus, CA3, CA2, and CA4 regions. The CA1 region is typically severely sclerotic, with few remaining neurons. The fourth stage generally involves the cerebellum where there is marked loss and distortion of axons throughout the cerebral and cerebellar white matter. TDP-43 deposition is severe and widespread with dense accumulations of dot-like and thread-like inclusions in neurites and intro-neuronal cytoplasmic inclusions in all cases (McKee et al., 2013).
11 CHRONIC TRAUMATIC ENCEPHALOPATHY 10 Biopsychosocial Model Approach Many researchers believe that to obtain a more comprehensive diagnosis, a biopsychosocial model should be applied. Existing research has examined convenient samples of athletes who donated their brains and were experiencing clinical symptoms prior to their deaththis has unfortunately led to misleading headlines about the risk of developing CTE pathology and estimates of its prevalence which are almost certainly inflated (O Keefe 2015). Attribution of pathological burden and symptom expression needs to be considered in a broad biopsychosocial explanatory framework that incorporates these factors in the casual model instead of focusing only on a history of repetitive brain trauma as the sole causative factor in the development or degree of CTE symptoms (Asken et al., 2016). The environment a particular person develops in may have a large impact on their behavior or psyche. Disadvantaged SES predisposes to increased aggression and higher susceptibility to depression, two symptoms also seen in CTE (McLoyd 1997). Interaction of SES and prenatal development, as well as delayed development, could affect cognitive and neural functioning into adulthood as well. Some research takes normal aging into account, and compares CTE to the normal aging brain, but a lot of the effects seen in CTE are also found in normal aging. Structural brain changes associated with normal aging are well-known and include neurofibrillary tangle (NFT) development, decreased grey and white matter volume, increased ventricular volume, increased white matter hyperintensities on MRI, and default mode network (regions of highly correlated functional activity) dysfunction on MRI as well (Andrews-Hanna et al. 2007). Decreased white matter integrity is also associated with slowed processing speed and impaired executive function (Asken et al., 2016). NFT deposition accompanies the normal aging process and the recently
12 CHRONIC TRAUMATIC ENCEPHALOPATHY 11 proposed primary age-related tauopathy (PART) describes the continuum of severities with and without associated cognitive changes (Crary et al. 2014) However, the unique distribution of tau deposition in the perivascular spaces of cortical sulci described in CTE differentiates CTE from PART, which is associated with NFT formation in the medial temporal lobe and several subcortical nuclei. So, contrary to other research, the pathological features of CTE do not represent normal or accelerated aging. Sleep disturbances may also impact the behavior of a particular person. Sleep is very important, recent evidence indicates that sleep enhances the clearance of metabolic waste products from interstitial space in the brain via the glymphatic system (Xie et al. 2013). The glymphatic system is thought to rely on the sleep state to relax interstitial spaces enabling cerebrospinal fluid (CSF) and interstitial fluid (ISF) to move through and clear metabolic waste (Jessen, Munk, Lundgaard, & Nedergaard, 2015). Interstitial space actually expands by up to 60% during sleep which accelerates the flow of fluids through the glymphatic system. Disruption to this system may be particularly relevant to collision sport athletes, who are likely to consistently accumulate metabolic waste from concussive and subclinical brain insults, including beta-amyloid from recurrent trauma (Giza and Hovda 2014). In many cases, sleep is disturbed after a concussion and disordered sleep can be a significant factor in the development of a wide range of cognitive, mood, and behavioral problems. A Case Study A case study was reported by Mez et al. in 2016 of a 25-year-old Division I college football defensive linebacker and special teams player. He began playing football at the age of six and stopped playing at the beginning of his junior season of college because of the ongoing symptoms he experienced. In his football career, he experienced more than 10 concussions, the
13 CHRONIC TRAUMATIC ENCEPHALOPATHY 12 first of which occurring at age eight. Although CTE was considered, the lack of delay in symptom onset, his young age and his family history of depression were the reasons against CTE as a primary diagnosis for his death. Focal lesions of CTE have been found in athletes as young as 17 years; however, widespread CTE pathology is unusual in such a young football player. Prospective studies that include neuropsychological testing with imaging and fluid biomarkers will be essential to future improvement in diagnosing CTE in living patients. Prevention Currently there are no treatments for CTE, primarily because CTE hasn t been diagnosed in a living person yet. Without treatment strategies available, it is important to prevent severe head injuries to mitigate CTE. Protective head gear is important in protecting against penetrating injuries, skull fracture, and intracranial hemorrhage, but they do not appear to mitigate the incidence or severity of sports-related concussions (Harmon et al., 2013). It is important to understand that repeated blows to the head without a concussion can still lead to CTE, kids need to be protected from this rick factor. Putting kids into non-contact sports will help mitigate the incidence of blows to the head, concussions and CTE. Future Research Neuroimaging using positron emission tomography (PET), diffusion tensor imaging (DTI), functional magnetic resonance imaging (fmri) may lead to the diagnosis of CTE in living patients. One approach is the depletion of molecules associated with CTE pathology (e.g., tau, Ab). The second approach is detecting structural or molecular changes as associated with head injury, which is thought to contribute to the development of CTE (Aldag et al., 2017). PET can detect the presence and distribution of specific molecules using trace amounts of radioactive ligands that bind to molecules of interest. Investigators are developing PET radioligands to
14 CHRONIC TRAUMATIC ENCEPHALOPATHY 13 image pathology associated with CTE, such as aggregations of tau and Ab (Villemangne & Okamura 2014; Barrio et al., 1999). Several PET radioligands targeting tau have shown potential as CTE biomarkers, some are even in clinical trials. Some challenges with PET are that p-tau aggregates are expressed intracellularly, which requires the corresponding imaging ligand to cross the blood brain-barrier and cell membrane (Villemagne, Fodero-Tacoletti, Masters, & Rowe 2015). DTI revealed significant white matter changes in a high school contact sport athlete after a single concussion and have supported a link between axonal abnormalities and executive impairment after traumatic brain injuries (Bazarian et al., 2012; Lipton et al., 2009). Researchers have used functional magnetic resonance imaging (fmri) to evaluate functional disruptions in both concussive and subconcussive injury groups, even in the absence of overt clinical symptoms (Talavage et al., 2010). Biospecimen-based biomarkers would be able to provide a more accessible, costeffective, and deployable method for identifying CTE in vivo than neuroimaging modalities. Neuroimaging modalities are resource dependent, very expensive, and located in fixed buildings. Cerebral spinal fluid has been thought to be a prime biospecimen, but some challenges have been met, and not overcome. Although CSF is considered a potential source of TBI biomarkers given its direct contact with the brain and nervous system, the lumbar puncture required to sample CSF poses many disadvantages (Turner et al., 2013). Blood plasma has less risks than CSF lumbar punctures, but there are still some drawbacks including: dilution of the brain-specific protein by the large volume of plasma and in the extracellular fluid of peripheral organs, degradation of the biomarker candidate by blood proteases, clearance of the protein by hepatic metabolism or renal excretion, and analyses of brain proteins in blood that can be confounded by release of the same protein from peripheral tissues (DeKosky, Blennow, Ikonomovic, & Gandy 2013).
15 CHRONIC TRAUMATIC ENCEPHALOPATHY 14 Currently, a definitive CTE diagnosis has only been confirmed with a post-mortem autopsy and immunohistochemistry for p-tau. It is believed that in the future, the use of biomarkers in along with diagnostic imaging such as PET and MRI will offer a promising opportunity for early diagnosis, monitoring of progression, and timely therapeutic interventions (Tharmartnam et al. 2018).
16 CHRONIC TRAUMATIC ENCEPHALOPATHY 15 References (80) Aldag M, Armstrong RC, Bandak F, Bellgowan PSF, Bentley T, Biggerstaff S, Caravelli K, Cmarik J, Crowder A, DeGraba TJ, Dittmer TA, Ellenborgen RG, Greene C, Gupta RK, Hicks R, Hoffman S, Latta RC, Leggieri MJ, Marion D, Mazzoli R, McCrea M, O Donnell J, Packer M, Petro JB, Rasmussen TE, Sammons-Jackson W, Shoge R, Tepe V, Tremaine LA, Zheng J. (2017). The Biological Basis of Chronic Traumatic Encephalopathy following Blast Injury: Literature Review. Journal of Neurotrauma 34, Andrews-Hanna JR, Snyder AZ, Vincent JL, Lustig C, Head D, Raichle Marcus E, Buckner RL. (2007). Disruption of Large-Scale Brain Systems in Advanced Aging. Neuron. 56(5), Armstrong, R.A., Ellis, W., Hamilton, R.L., Mackenzie, I.R., Hedreen, J., Gearing, M., Montine, T., Vonsattel, J.P., Head, E., Lieberman, A.P., and Cairns, N.J. (2009). Neuropathological heterogeneity in frontotemporal lobar degeneration with TDP-43 proteinopathy: a quantitative study of 94 cases using principal components analysis. J. Neural. Transm. 117, Asken B, Sullan M, Snyder A, Houck Z, Bryant V, Hizel L, McLaren M, Dede D, Jaffee M, DeKosky S, Bauer R. (2016). Factors Influencing Clinical Correlates of Chronic Traumatic Encephalopathy (CTE): A Review. Neuropsychol Rev. 26(4), Barrio, J.R., Huang, S.C., Cole, G., Satyamurthy N., Petric A., Phelps M.E., and Small, G. (1999). PET imaging of tangles and plaques in Alzheimer disease with a highly hydrophobic probe. J. Labelled Compd. Radiopharm. S194 S195.
17 CHRONIC TRAUMATIC ENCEPHALOPATHY 16 Bazarian, J.J., Zhu, T., Blyth, B., Borrino, A., and Zhong, J. (2012). Subject-specific changes in brain white matter on diffusion tensor imaging after sports-related concussion. Magn. Reson. Imaging 30, Bosque, P.J., Boyer, P.J., and Mishra, P. (2013). A 43-kDa TDP-43 species is present in aggregates associated with frontotemporal lobar degeneration. PLoS ONE 8, e Chen L. (2018). What triggers tauopathy in chronic traumatic encephalopathy? Neural Regeneration Research 13(6), Corsellis JA, Bruton CJ, Freeman-Browne D. (1973) The aftermath of boxing. Psychol Med 3, Crary JF, et al. (2014). Primary age-related tauopathy (PART): a common pathology associated with human aging. Acta Neuropathol. 128(6), DeKosky, S.T., Blennow, K., Ikonomovic, M.D., and Gandy, S. (2013). Acute and chronic traumatic encephalopathies: pathogenesis and biomarkers. Nat Rev Neurol 9, Faden, A.I., Wu, J., Stoica, B.A., and Loane, D.J. (2015). Progressive inflammation-mediated neurodegeneration after traumatic brain or spinal cord injury. Br. J. Pharmacol. 173, Giza CC, Hovda DA. (2014). The new neurometabolic cascade of concussion. Neurosurgery. 75(Suppl 4), S24 33 Gu J, Wu F, Xu W, Shi J, Hu W, Jin N, Qian W, Wang X, Iqbal K, Gong CX, Liu F. (2017). TDP-43 suppresses tau expression via promoting its mrna instability. Nucleic Acids Res. 45, Harmon, K.G., Drezner, J.A., Gammons, M., Guskiewicz, K.M., Halstead, M., Herring, S.A., Kutcher, J.S., Pana, A., Putukian, M., and Roberts, W.O. (2013). American Medical
18 CHRONIC TRAUMATIC ENCEPHALOPATHY 17 Society for Sports Medicine position statement: concussion in sport. Br J Sports Med 47, Jessen NA, Munk AS, Lundgaard I, Nedergaard M. (2015). The Glymphatic System: A Beginner s Guide. Neurochem Res. 40(12), Ling JP, Pletnikova O, Troncoso JC, Wong PC. (2015). TDP-43 repression of nonconserved cryptic exons is compromised in ALS-FTD. Science. 349, Lipton, M.L., Gulko, E., Zimmerman, M.E., Friedman, B.W., Kim, M., Gellella, E., Gold, T., Shifteh, K., Ardekani, B.A., and Branch, C.A. (2009). Diffusion-tensor imaging implicates prefrontal axonal injury in executive function impairment following very mild traumatic brain injury. Radiology 252, Lucke-Wold, B.P., Turner, R.C., Logsdon, A.F., Bailes, J.E., Huber, J.D., and Rosen, C.L. (2014). Linking traumatic brain injury to chronic traumatic encephalopathy: identification of potential mechanisms leading to neurofibrillary tangle development. J. Neurotrauma 31, McKee AC, Cairns NJ, Dickson DW, Folkerth RD, Keene CD, Litvan I, Perl DP, Stein TD, Vonsattel JP, Stewart W, Tripodis Y, Crary JF, Bieniek KF, Dams-O Connor K, Alvarez VE, Gordon WA TBI/CTE group. (2016). The first NINDS/NIBIB consensus meeting to define neuropathological criteria for the diagnosis of chronic traumatic encephalopathy. Acta Neuropathol. 131, McKee AC, Stein TD, Nowinski CJ, Stern RA, Daneshvar DH, Alvarez VE, Lee H-S, Hall G, Wojtowicz SM, Baugh CM, Riley DO, Kubilus CA, Cormier KA, Jacobs MA, Martin BR, Abraham CR, Ikezu T, Reichard RR, Wolozin BL, Budson AE, Goldstein LE,
19 CHRONIC TRAUMATIC ENCEPHALOPATHY 18 Kowall NW, Cantu RC. (2013). The spectrum of disease in chronic traumatic encephalopathy. Brain 136, McLoyd VC. (1997). The impact of poverty and low socioeconomic status on the socioemotional functioning of African-American children and adolescents: Mediating effects. Social and emotional adjustment and family relations in ethnic minority families. 7, 34. Mez J, Solomon T, Daneshvar D, Stein T, McKee A. (2016). Pathologically Confirmed Chronic Traumatic Encephalopathy in a 25-Year-Old Former College Football Player. JAMA Neurol. 73(3), Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Bruce J, Schuck T, Grossman M, Clark CM, McCluskey LF, Miller BL, Masliah E, Mackenzie IR, Feldman H, Feiden W, Kretzschmar HA, Trojanowski JQ, Lee VM. (2006). Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science. 314, O Keefe, M. (2015). Study finds 96 percent of former NFL players had CTE. Omalu, B., Bailes, J., Hamilton, R.L., Kamboh, M.I., Hammers, J., Case, M., and Fitzsimmons, R. (2011). Emerging histomorphologic phenotypes of chronic traumatic encephalopathy in American athletes. Neurosurgery 69, Osnato M. (1927). Postconcussion neurosis-traumatic encephalitis a conception of postconcussion phenomena. Arch NeurPsych 18, Stein et al. (2014). Chronic traumatic encephalopathy: a spectrum of neuropathological changes following repetitive brain trauma in athletes and military personnel. Alzheimer's Research & Therapy 6, 4.
20 CHRONIC TRAUMATIC ENCEPHALOPATHY 19 Thamaratnam T, Iskandar M, Tabobondung T, Tobbia I, Gopee-Ramanan P, Tabobondung T. (2018) Chronic Traumatic Encephalopathy in Professional American Football Players: Where Are We Now? Front. Neurol. 9. Turner, R.C., Lucke-Wold, B.P., Robson, M.J., Omalu, B.I., Pet- raglia, A.L., and Bailes, J.E. (2013). Repetitive traumatic brain injury and development of chronic traumatic encephalopathy: a potential role for biomarkers in diagnosis, prognosis, and treatment? Front. Neurol. 3, 186. Villemagne, V.L. and Okamura, N. (2014). In vivo tau imaging: obstacles and progress. Alzheimers Dement. 10, Suppl, S254 S264. Xie L, et al. (2013). Sleep drives metabolite clearance from the adult brain. Science, 342(6156),
A Critical Review of Chronic Traumatic Encephalopathy
 A Critical Review of Chronic Traumatic Encephalopathy Grant L. Iverson, Ph.D. Professor, Department of Physical Medicine and Rehabilitation, Harvard Medical School; Director, MassGeneral Hospital for Children
A Critical Review of Chronic Traumatic Encephalopathy Grant L. Iverson, Ph.D. Professor, Department of Physical Medicine and Rehabilitation, Harvard Medical School; Director, MassGeneral Hospital for Children
Chronic Traumatic Encephalopathy Provider and Parent Essentials
 Chronic Traumatic Encephalopathy Provider and Parent Essentials Concussion Global Cast July 30, 2014 John Lockhart, MD Seattle Children s Hospital Chronic Traumatic Encephaly (CTE) Working Definition Chronic
Chronic Traumatic Encephalopathy Provider and Parent Essentials Concussion Global Cast July 30, 2014 John Lockhart, MD Seattle Children s Hospital Chronic Traumatic Encephaly (CTE) Working Definition Chronic
The Biological Basis of Chronic Traumatic Encephalopathy following Blast Injury: A Literature Review
 JOURNAL OF NEUROTRAUMA 34:S-26 S-43 (Supplement 1, 2017) ª Mary Ann Liebert, Inc. DOI: 10.1089/neu.2017.5218 The Biological Basis of Chronic Traumatic Encephalopathy following Blast Injury: A Literature
JOURNAL OF NEUROTRAUMA 34:S-26 S-43 (Supplement 1, 2017) ª Mary Ann Liebert, Inc. DOI: 10.1089/neu.2017.5218 The Biological Basis of Chronic Traumatic Encephalopathy following Blast Injury: A Literature
An Autopsy Proven Child Onset Chronic Traumatic Encephalopathy
 https://doi.org/10.5607/en.2017.26.3.172 Exp Neurobiol. 2017 Jun;26(3):172-177. pissn 1226-2560 eissn 2093-8144 Case Report An Autopsy Proven Child Onset Chronic Traumatic Encephalopathy Kyuho Lee 1, Seong-Ik
https://doi.org/10.5607/en.2017.26.3.172 Exp Neurobiol. 2017 Jun;26(3):172-177. pissn 1226-2560 eissn 2093-8144 Case Report An Autopsy Proven Child Onset Chronic Traumatic Encephalopathy Kyuho Lee 1, Seong-Ik
It all started in 1927 when Osnato and Gilberti. Postmortem evaluation of chronic traumatic encephalopathy
 Rom J Leg Med [24] 266-272 [2016] DOI: 10.4323/rjlm.2016.266 2016 Romanian Society of Legal Medicine Ovidiu Chiroban 1,*, Lăcrămioara Perju-Dumbravă 2 Abstract: Chronic traumatic encephalopathy (CTE) is
Rom J Leg Med [24] 266-272 [2016] DOI: 10.4323/rjlm.2016.266 2016 Romanian Society of Legal Medicine Ovidiu Chiroban 1,*, Lăcrămioara Perju-Dumbravă 2 Abstract: Chronic traumatic encephalopathy (CTE) is
Chronic traumatic encephalopathy: Emerging Concepts
 Chronic traumatic encephalopathy: Emerging Concepts Ann C. McKee M.D. Professor of Neurology and Pathology VA Boston Healthcare System Boston University School of Medicine Director of the CTE Center Boston
Chronic traumatic encephalopathy: Emerging Concepts Ann C. McKee M.D. Professor of Neurology and Pathology VA Boston Healthcare System Boston University School of Medicine Director of the CTE Center Boston
MULTIPLE CONCUSSIONS LEAD TO CHRONIC TRAUMATIC ENCEPHALOPATHY (CTE)
 MULTIPLE CONCUSSIONS LEAD TO CHRONIC TRAUMATIC ENCEPHALOPATHY (CTE) CTE may develop within a short time of trauma but usually years intervene Symptoms Cognitive: depression, dementia Movement disorders:
MULTIPLE CONCUSSIONS LEAD TO CHRONIC TRAUMATIC ENCEPHALOPATHY (CTE) CTE may develop within a short time of trauma but usually years intervene Symptoms Cognitive: depression, dementia Movement disorders:
DVHIP. TBI: Clinical Issues, Controversies, and Learning from Patients. Defense and Veterans Head Injury Program. What is Neuropsychology?
 TBI: Clinical Issues, Controversies, and Learning from Patients DVHIP Defense and Veterans Head Injury Program Richard A. Lanham, Jr., Ph.D. Assistant Professor Division of Medical Psychology Psychiatry
TBI: Clinical Issues, Controversies, and Learning from Patients DVHIP Defense and Veterans Head Injury Program Richard A. Lanham, Jr., Ph.D. Assistant Professor Division of Medical Psychology Psychiatry
Plan for Today. Brain Injury: 8/4/2017. Effective Services for People Living with Brain Injury. What is it & what causes it?
 Effective Services for People Living with Brain Injury Jean Capler, MSW, LSW Local Support Network Leader The Rehabilitation Hospital of Indiana Department of Resource Facilitation Plan for Today Brain
Effective Services for People Living with Brain Injury Jean Capler, MSW, LSW Local Support Network Leader The Rehabilitation Hospital of Indiana Department of Resource Facilitation Plan for Today Brain
The Changing Landscape of Sports Concussions
 The Changing Landscape of Sports Concussions Anthony G. Alessi MD, FAAN Director, UConn NeuroSport Sports Medicine Symposium August 2, 2016 Overview Significance Diagnosis Sideline diagnosis v Management
The Changing Landscape of Sports Concussions Anthony G. Alessi MD, FAAN Director, UConn NeuroSport Sports Medicine Symposium August 2, 2016 Overview Significance Diagnosis Sideline diagnosis v Management
Sports Concussion: What Do We Really Know?
 Sports Concussion: What Do We Really Know? Anthony G. Alessi MD, FAAN Director, UConn NeuroSport October 17, 2018 Overview Significance Diagnosis Sideline diagnosis v Management Concussion Tools Return-to-Play
Sports Concussion: What Do We Really Know? Anthony G. Alessi MD, FAAN Director, UConn NeuroSport October 17, 2018 Overview Significance Diagnosis Sideline diagnosis v Management Concussion Tools Return-to-Play
The Dangers of CTE. James Ryan Cox. Skylar Spriggs. Sawyer Solfest. Team THS131
 The Dangers of CTE James Ryan Cox Skylar Spriggs Sawyer Solfest Team THS131 Dec 10th, 2017 The human body works alike to a well-oiled machine, each vital organ working as a valuable gear or cog. Yet if
The Dangers of CTE James Ryan Cox Skylar Spriggs Sawyer Solfest Team THS131 Dec 10th, 2017 The human body works alike to a well-oiled machine, each vital organ working as a valuable gear or cog. Yet if
! slow, progressive, permanent loss of neurologic function.
 UBC ! slow, progressive, permanent loss of neurologic function.! cause unknown.! sporadic, familial or inherited.! degeneration of specific brain region! clinical syndrome.! pathology: abnormal accumulation
UBC ! slow, progressive, permanent loss of neurologic function.! cause unknown.! sporadic, familial or inherited.! degeneration of specific brain region! clinical syndrome.! pathology: abnormal accumulation
8/24/18. Dementia. Risk of Dementia Following Traumatic Brain Injury: A Review of the Literature. Media Presence. Media Presence
 Risk of Dementia Following Traumatic Brain Injury: A Review of the Literature Media Presence Carlos Marquez de la Plata, Ph.D. & Jeff Schaffert, M.S. Media Presence Dementia What is dementia? Dementia
Risk of Dementia Following Traumatic Brain Injury: A Review of the Literature Media Presence Carlos Marquez de la Plata, Ph.D. & Jeff Schaffert, M.S. Media Presence Dementia What is dementia? Dementia
the injured brain, the injured mind.
 the injured brain, the injured mind. mary et boyle, ph.d. department of cognitive science ucsd Traumatic Brain Injury (TBI) is caused by a bump, blow or jolt to the head or a penetrating head injury the
the injured brain, the injured mind. mary et boyle, ph.d. department of cognitive science ucsd Traumatic Brain Injury (TBI) is caused by a bump, blow or jolt to the head or a penetrating head injury the
Role of TDP-43 in Non-Alzheimer s and Alzheimer s Neurodegenerative Diseases
 Role of TDP-43 in Non-Alzheimer s and Alzheimer s Neurodegenerative Diseases Keith A. Josephs, MD, MST, MSc Professor of Neurology 13th Annual Mild Cognitive Impairment (MCI) Symposium: Alzheimer and Non-Alzheimer
Role of TDP-43 in Non-Alzheimer s and Alzheimer s Neurodegenerative Diseases Keith A. Josephs, MD, MST, MSc Professor of Neurology 13th Annual Mild Cognitive Impairment (MCI) Symposium: Alzheimer and Non-Alzheimer
Clinicopathologic and genetic aspects of hippocampal sclerosis. Dennis W. Dickson, MD Mayo Clinic, Jacksonville, Florida USA
 Clinicopathologic and genetic aspects of hippocampal sclerosis Dennis W. Dickson, MD Mayo Clinic, Jacksonville, Florida USA The hippocampus in health & disease A major structure of the medial temporal
Clinicopathologic and genetic aspects of hippocampal sclerosis Dennis W. Dickson, MD Mayo Clinic, Jacksonville, Florida USA The hippocampus in health & disease A major structure of the medial temporal
REVIEW. Montenigro et al. Alzheimer's Research & Therapy 2014, 6:68
 Montenigro et al. Alzheimer's Research & Therapy 2014, 6:68 REVIEW Clinical subtypes of chronic traumatic encephalopathy: literature review and proposed research diagnostic criteria for traumatic encephalopathy
Montenigro et al. Alzheimer's Research & Therapy 2014, 6:68 REVIEW Clinical subtypes of chronic traumatic encephalopathy: literature review and proposed research diagnostic criteria for traumatic encephalopathy
Concussion Update and Case Presentations
 Concussion Update and Case Presentations Cayce Onks, DO, MS, ATC Associate Professor Primary Care Sports Medicine Penn State Concussion Program Departments of Family Medicine and Orthopaedics I have no
Concussion Update and Case Presentations Cayce Onks, DO, MS, ATC Associate Professor Primary Care Sports Medicine Penn State Concussion Program Departments of Family Medicine and Orthopaedics I have no
 Johnson, V. E., and Stewart, W. (2015) Traumatic brain injury: Age at injury influences dementia risk after TBI. Nature Reviews Neurology, 11(3), pp. 128-130. (doi:10.1038/nrneurol.2014.241) There may
Johnson, V. E., and Stewart, W. (2015) Traumatic brain injury: Age at injury influences dementia risk after TBI. Nature Reviews Neurology, 11(3), pp. 128-130. (doi:10.1038/nrneurol.2014.241) There may
Chronic traumatic encephalopathy: clinical biomarker correlations and current concepts in pathogenesis
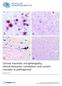 Chronic traumatic encephalopathy: clinical biomarker correlations and current concepts in pathogenesis Gandy et al. Gandy et al. Molecular Neurodegeneration 2014, 9:37 Gandy et al. Molecular Neurodegeneration
Chronic traumatic encephalopathy: clinical biomarker correlations and current concepts in pathogenesis Gandy et al. Gandy et al. Molecular Neurodegeneration 2014, 9:37 Gandy et al. Molecular Neurodegeneration
Pathogenesis of Degenerative Diseases and Dementias. D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria)
 Pathogenesis of Degenerative Diseases and Dementias D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria) Dementias Defined: as the development of memory impairment and other cognitive deficits
Pathogenesis of Degenerative Diseases and Dementias D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria) Dementias Defined: as the development of memory impairment and other cognitive deficits
Mild Cognitive Impairment
 Mild Cognitive Impairment Victor W. Henderson, MD, MS Departments of Health Research & Policy (Epidemiology) and of Neurology & Neurological Sciences Stanford University Director, Stanford Alzheimer s
Mild Cognitive Impairment Victor W. Henderson, MD, MS Departments of Health Research & Policy (Epidemiology) and of Neurology & Neurological Sciences Stanford University Director, Stanford Alzheimer s
PET Scanning of Brain Tau in Retired National Football League Players: Preliminary Findings
 PET Scanning of Brain Tau in Retired National Football League Players: Preliminary Findings Gary W. Small, M.D., Vladimir Kepe, Ph.D., Prabha Siddarth, Ph.D., Linda M. Ercoli, Ph.D., David A. Merrill,
PET Scanning of Brain Tau in Retired National Football League Players: Preliminary Findings Gary W. Small, M.D., Vladimir Kepe, Ph.D., Prabha Siddarth, Ph.D., Linda M. Ercoli, Ph.D., David A. Merrill,
Imaging of Alzheimer s Disease: State of the Art
 July 2015 Imaging of Alzheimer s Disease: State of the Art Neir Eshel, Harvard Medical School Year IV Outline Our patient Definition of dementia Alzheimer s disease Epidemiology Diagnosis Stages of progression
July 2015 Imaging of Alzheimer s Disease: State of the Art Neir Eshel, Harvard Medical School Year IV Outline Our patient Definition of dementia Alzheimer s disease Epidemiology Diagnosis Stages of progression
Neuro degenerative PET image from FDG, amyloid to Tau
 Neuro degenerative PET image from FDG, amyloid to Tau Kun Ju Lin ( ) MD, Ph.D Department of Nuclear Medicine and Molecular Imaging Center, Chang Gung Memorial Hospital ( ) Department of Medical Imaging
Neuro degenerative PET image from FDG, amyloid to Tau Kun Ju Lin ( ) MD, Ph.D Department of Nuclear Medicine and Molecular Imaging Center, Chang Gung Memorial Hospital ( ) Department of Medical Imaging
FRONTOTEMPORAL DEGENERATION: OVERVIEW, TRENDS AND DEVELOPMENTS
 FRONTOTEMPORAL DEGENERATION: OVERVIEW, TRENDS AND DEVELOPMENTS Norman L. Foster, M.D. Director, Center for Alzheimer s Care, Imaging and Research Chief, Division of Cognitive Neurology, Department of Neurology
FRONTOTEMPORAL DEGENERATION: OVERVIEW, TRENDS AND DEVELOPMENTS Norman L. Foster, M.D. Director, Center for Alzheimer s Care, Imaging and Research Chief, Division of Cognitive Neurology, Department of Neurology
Seth Smith MD, PharmD. Thank You!
 Seth Smith MD, PharmD Thank You! Concussion Headlines!!! Concussion Headlines!!! Concussion rules put kids health before winning games The Miami Herald 8/21/12 FHSAA's implementation of legislation goes
Seth Smith MD, PharmD Thank You! Concussion Headlines!!! Concussion Headlines!!! Concussion rules put kids health before winning games The Miami Herald 8/21/12 FHSAA's implementation of legislation goes
DISCLOSURES. Objectives. THE EPIDEMIC of 21 st Century. Clinical Assessment of Cognition: New & Emerging Tools for Diagnosing Dementia NONE TO REPORT
 Clinical Assessment of Cognition: New & Emerging Tools for Diagnosing Dementia DISCLOSURES NONE TO REPORT Freddi Segal Gidan, PA, PhD USC Keck School of Medicine Rancho/USC California Alzheimers Disease
Clinical Assessment of Cognition: New & Emerging Tools for Diagnosing Dementia DISCLOSURES NONE TO REPORT Freddi Segal Gidan, PA, PhD USC Keck School of Medicine Rancho/USC California Alzheimers Disease
Dementia. Jeanette Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine
 Dementia Jeanette Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine What is Dementia? Dementia is a general term referring to a decline in cognitive/mental functioning; this decline
Dementia Jeanette Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine What is Dementia? Dementia is a general term referring to a decline in cognitive/mental functioning; this decline
Neuropathology of Neurodegenerative Disorders Prof. Jillian Kril
 Neurodegenerative disorders to be discussed Alzheimer s disease Lewy body diseases Frontotemporal dementia and other tauopathies Huntington s disease Motor Neuron Disease 2 Neuropathology of neurodegeneration
Neurodegenerative disorders to be discussed Alzheimer s disease Lewy body diseases Frontotemporal dementia and other tauopathies Huntington s disease Motor Neuron Disease 2 Neuropathology of neurodegeneration
THE ASSOCIATIONS BETWEEN FOOTBALL EXPOSURE, CONCUSSION HISTORY AND PLAYING POSITION ON CEREBRAL WHITE MATTER INTEGRITY AND NEUROCOGNITIVE PERFORMANCE
 THE ASSOCIATIONS BETWEEN FOOTBALL EXPOSURE, CONCUSSION HISTORY AND PLAYING POSITION ON CEREBRAL WHITE MATTER INTEGRITY AND NEUROCOGNITIVE PERFORMANCE By Allen Anthony Champagne Senior Honors Thesis Department
THE ASSOCIATIONS BETWEEN FOOTBALL EXPOSURE, CONCUSSION HISTORY AND PLAYING POSITION ON CEREBRAL WHITE MATTER INTEGRITY AND NEUROCOGNITIVE PERFORMANCE By Allen Anthony Champagne Senior Honors Thesis Department
Many sports-related brain injuries involve mild
 neurosurgical focus Neurosurg Focus 40 (4):E14, 2016 Sports-related brain injuries: connecting pathology to diagnosis *James Pan, BS, 1 Ian D. Connolly, MS, 1 Sean Dangelmajer, BA, 2 James Kintzing, BS,
neurosurgical focus Neurosurg Focus 40 (4):E14, 2016 Sports-related brain injuries: connecting pathology to diagnosis *James Pan, BS, 1 Ian D. Connolly, MS, 1 Sean Dangelmajer, BA, 2 James Kintzing, BS,
2. Subarachnoid Hemorrhage
 Causes: 2. Subarachnoid Hemorrhage A. Saccular (berry) aneurysm - Is the most frequent cause of clinically significant subarachnoid hemorrhage is rupture of a saccular (berry) aneurysm. B. Vascular malformation
Causes: 2. Subarachnoid Hemorrhage A. Saccular (berry) aneurysm - Is the most frequent cause of clinically significant subarachnoid hemorrhage is rupture of a saccular (berry) aneurysm. B. Vascular malformation
Yin-Hui Siow MD, FRCPC Director of Nuclear Medicine Southlake Regional Health Centre
 Yin-Hui Siow MD, FRCPC Director of Nuclear Medicine Southlake Regional Health Centre Today Introduction to CT Introduction to MRI Introduction to nuclear medicine Imaging the dementias The Brain ~ 1.5
Yin-Hui Siow MD, FRCPC Director of Nuclear Medicine Southlake Regional Health Centre Today Introduction to CT Introduction to MRI Introduction to nuclear medicine Imaging the dementias The Brain ~ 1.5
HIV Neurology Persistence of Cognitive Impairment Despite cart
 HIV Neurology Persistence of Cognitive Impairment Despite cart Victor Valcour MD PhD Professor of Medicine Memory and Aging Center, Dept. of Neurology University of California San Francisco, USA 8 th International
HIV Neurology Persistence of Cognitive Impairment Despite cart Victor Valcour MD PhD Professor of Medicine Memory and Aging Center, Dept. of Neurology University of California San Francisco, USA 8 th International
Dementia. Stephen S. Flitman, MD Medical Director 21st Century Neurology
 Dementia Stephen S. Flitman, MD Medical Director 21st Century Neurology www.neurozone.org Dementia is a syndrome Progressive memory loss, plus Progressive loss of one or more cognitive functions: Language
Dementia Stephen S. Flitman, MD Medical Director 21st Century Neurology www.neurozone.org Dementia is a syndrome Progressive memory loss, plus Progressive loss of one or more cognitive functions: Language
Dementia Update. October 1, 2013 Dylan Wint, M.D. Cleveland Clinic Lou Ruvo Center for Brain Health Las Vegas, Nevada
 Dementia Update October 1, 2013 Dylan Wint, M.D. Cleveland Clinic Lou Ruvo Center for Brain Health Las Vegas, Nevada Outline New concepts in Alzheimer disease Biomarkers and in vivo diagnosis Future trends
Dementia Update October 1, 2013 Dylan Wint, M.D. Cleveland Clinic Lou Ruvo Center for Brain Health Las Vegas, Nevada Outline New concepts in Alzheimer disease Biomarkers and in vivo diagnosis Future trends
Examination of the relationship between sport concussion and long term neurodegenerative and psychological disorders: a literature review
 University of Central Florida HIM 1990-2015 Open Access Examination of the relationship between sport concussion and long term neurodegenerative and psychological disorders: a literature review 2013 Vivian
University of Central Florida HIM 1990-2015 Open Access Examination of the relationship between sport concussion and long term neurodegenerative and psychological disorders: a literature review 2013 Vivian
Alzheimer's disease (AD), also known as Senile Dementia of the Alzheimer Type (SDAT) or simply Alzheimer s is the most common form of dementia.
 CHAPTER 3 Alzheimer's disease (AD), also known as Senile Dementia of the Alzheimer Type (SDAT) or simply Alzheimer s is the most common form of dementia. This incurable, degenerative, terminal disease
CHAPTER 3 Alzheimer's disease (AD), also known as Senile Dementia of the Alzheimer Type (SDAT) or simply Alzheimer s is the most common form of dementia. This incurable, degenerative, terminal disease
Brain Concussion: A Stealth Injury. Christine Schulman, RN, MS, CNS, CCRN Trauma & Critical Care CNS Legacy Health, Portland, Oregon Director, AACN
 Brain Concussion: A Stealth Injury Christine Schulman, RN, MS, CNS, CCRN Trauma & Critical Care CNS Legacy Health, Portland, Oregon Director, AACN Legacy Emanuel Medical Center Portland, Oregon Disclosures
Brain Concussion: A Stealth Injury Christine Schulman, RN, MS, CNS, CCRN Trauma & Critical Care CNS Legacy Health, Portland, Oregon Director, AACN Legacy Emanuel Medical Center Portland, Oregon Disclosures
The role of head traume and concussions in producing chronic traumatic encephalopathy: analysis of risk factores, diagnosis, treatment, and prevention
 Boston University OpenBU Theses & Dissertations http://open.bu.edu Boston University Theses & Dissertations 2013 The role of head traume and concussions in producing chronic traumatic encephalopathy: analysis
Boston University OpenBU Theses & Dissertations http://open.bu.edu Boston University Theses & Dissertations 2013 The role of head traume and concussions in producing chronic traumatic encephalopathy: analysis
Disclosures 7/22/2015 MANAGEMENT OF SPORTS RELATED CONCUSSION. Brad Herskowitz MD Neurologist Baptist Hospital
 MANAGEMENT OF SPORTS RELATED CONCUSSION Brad Herskowitz MD Neurologist Baptist Hospital Disclosures I have no relevant financial conflicts of interest. I will not discuss off label or unapproved usage.
MANAGEMENT OF SPORTS RELATED CONCUSSION Brad Herskowitz MD Neurologist Baptist Hospital Disclosures I have no relevant financial conflicts of interest. I will not discuss off label or unapproved usage.
New life Collage of nursing Karachi
 New life Collage of nursing Karachi Presenter: Zafar ali shah Faculty: Raja khatri Subject: Pathophysiology Topic :Alzheimer s Disease Post RN BScN semester 2 nd Objective Define Alzheimer s Describe pathophysiology
New life Collage of nursing Karachi Presenter: Zafar ali shah Faculty: Raja khatri Subject: Pathophysiology Topic :Alzheimer s Disease Post RN BScN semester 2 nd Objective Define Alzheimer s Describe pathophysiology
CASE 49. What type of memory is available for conscious retrieval? Which part of the brain stores semantic (factual) memories?
 CASE 49 A 43-year-old woman is brought to her primary care physician by her family because of concerns about her forgetfulness. The patient has a history of Down syndrome but no other medical problems.
CASE 49 A 43-year-old woman is brought to her primary care physician by her family because of concerns about her forgetfulness. The patient has a history of Down syndrome but no other medical problems.
Does a Unique Neuropsychiatric Profile Currently Exist for Chronic Traumatic Encephalopathy?
 HEAD, NECK, AND SPINE Does a Unique Neuropsychiatric Profile Currently Exist for Chronic Traumatic Encephalopathy? Faith M. Hanlon, PhD 1 ; Christopher A. McGrew, MD, FACSM 2,3 ; and Andrew R. Mayer, PhD
HEAD, NECK, AND SPINE Does a Unique Neuropsychiatric Profile Currently Exist for Chronic Traumatic Encephalopathy? Faith M. Hanlon, PhD 1 ; Christopher A. McGrew, MD, FACSM 2,3 ; and Andrew R. Mayer, PhD
UPDATE ON CHRONIC TRAUMATIC ENCEPHALOPATHY (CTE)
 UPDATE ON CHRONIC TRAUMATIC ENCEPHALOPATHY (CTE) Mark A. Hogue, Psy.D. Partner: Northshore Neurosciences UPMC Neurosciences Conference, 2016 Objectives Quick review of what is a concussion. Briefly review
UPDATE ON CHRONIC TRAUMATIC ENCEPHALOPATHY (CTE) Mark A. Hogue, Psy.D. Partner: Northshore Neurosciences UPMC Neurosciences Conference, 2016 Objectives Quick review of what is a concussion. Briefly review
Delirium & Dementia. Nicholas J. Silvestri, MD
 Delirium & Dementia Nicholas J. Silvestri, MD Outline Delirium vs. Dementia Neural pathways relating to consciousness Encephalopathy Stupor Coma Dementia Delirium vs. Dementia Delirium Abrupt onset Lasts
Delirium & Dementia Nicholas J. Silvestri, MD Outline Delirium vs. Dementia Neural pathways relating to consciousness Encephalopathy Stupor Coma Dementia Delirium vs. Dementia Delirium Abrupt onset Lasts
Functional Neuroanatomy and Traumatic Brain Injury The Frontal Lobes
 Functional Neuroanatomy and Traumatic Brain Injury The Frontal Lobes Jessica Matthes, Ph.D., ABN Barrow TBI Symposium March 23, 2019 jessica.matthes@dignityhealth.org Outline TBI Mechanisms of Injury Types
Functional Neuroanatomy and Traumatic Brain Injury The Frontal Lobes Jessica Matthes, Ph.D., ABN Barrow TBI Symposium March 23, 2019 jessica.matthes@dignityhealth.org Outline TBI Mechanisms of Injury Types
Form D1: Clinician Diagnosis
 Initial Visit Packet Form D: Clinician Diagnosis NACC Uniform Data Set (UDS) ADC name: Subject ID: Form date: / / Visit #: Examiner s initials: INSTRUCTIONS: This form is to be completed by the clinician.
Initial Visit Packet Form D: Clinician Diagnosis NACC Uniform Data Set (UDS) ADC name: Subject ID: Form date: / / Visit #: Examiner s initials: INSTRUCTIONS: This form is to be completed by the clinician.
The Spectrum of Age-Associated Astroglial Tauopathies. Dennis W. Dickson MD Department of Neuroscience Mayo Clinic, Jacksonville, FL
 The Spectrum of Age-Associated Astroglial Tauopathies Dennis W. Dickson MD Mayo Clinic, Jacksonville, FL Thorn-shaped astrocytes TSA were first reported by Ikeda (1995), as tau-positive astrocytes in various
The Spectrum of Age-Associated Astroglial Tauopathies Dennis W. Dickson MD Mayo Clinic, Jacksonville, FL Thorn-shaped astrocytes TSA were first reported by Ikeda (1995), as tau-positive astrocytes in various
Disclosures. The Spectrum of Concussion: Recovery Time, Treatment and Rehabilitation, and Possible Long-Term Effects on Brain Health
 The Spectrum of Concussion: Recovery Time, Treatment and Rehabilitation, and Possible Long-Term Effects on Brain Health Grant L. Iverson, Ph.D. Professor, Department of Physical Medicine and Rehabilitation,
The Spectrum of Concussion: Recovery Time, Treatment and Rehabilitation, and Possible Long-Term Effects on Brain Health Grant L. Iverson, Ph.D. Professor, Department of Physical Medicine and Rehabilitation,
V. CENTRAL NERVOUS SYSTEM TRAUMA
 V. CENTRAL NERVOUS SYSTEM TRAUMA I. Concussion - Is a clinical syndrome of altered consiousness secondary to head injury - Brought by a change in the momentum of the head when a moving head suddenly arrested
V. CENTRAL NERVOUS SYSTEM TRAUMA I. Concussion - Is a clinical syndrome of altered consiousness secondary to head injury - Brought by a change in the momentum of the head when a moving head suddenly arrested
THE ESSENTIAL BRAIN INJURY GUIDE
 THE ESSENTIAL BRAIN INJURY GUIDE Neuroanatomy & Neuroplasticity Section 2 Contributors Erin D. Bigler, PhD Michael R. Hoane, PhD Stephanie Kolakowsky-Hayner, PhD, CBIST, FACRM Dorothy A. Kozlowski, PhD
THE ESSENTIAL BRAIN INJURY GUIDE Neuroanatomy & Neuroplasticity Section 2 Contributors Erin D. Bigler, PhD Michael R. Hoane, PhD Stephanie Kolakowsky-Hayner, PhD, CBIST, FACRM Dorothy A. Kozlowski, PhD
Concussion Management
 Concussion Management Jennifer Gray, DO Medical Co-Director, ThinkSMART! Concussion Management Program Department of Physical Medicine and Rehabilitation St. Charles Hospital Residency Program Director,
Concussion Management Jennifer Gray, DO Medical Co-Director, ThinkSMART! Concussion Management Program Department of Physical Medicine and Rehabilitation St. Charles Hospital Residency Program Director,
Gross Organization I The Brain. Reading: BCP Chapter 7
 Gross Organization I The Brain Reading: BCP Chapter 7 Layout of the Nervous System Central Nervous System (CNS) Located inside of bone Includes the brain (in the skull) and the spinal cord (in the backbone)
Gross Organization I The Brain Reading: BCP Chapter 7 Layout of the Nervous System Central Nervous System (CNS) Located inside of bone Includes the brain (in the skull) and the spinal cord (in the backbone)
Mild Cognitive Impairment (MCI)
 October 19, 2018 Mild Cognitive Impairment (MCI) Yonas E. Geda, MD, MSc Professor of Neurology and Psychiatry Consultant, Departments of Psychiatry & Psychology, and Neurology Mayo Clinic College of Medicine
October 19, 2018 Mild Cognitive Impairment (MCI) Yonas E. Geda, MD, MSc Professor of Neurology and Psychiatry Consultant, Departments of Psychiatry & Psychology, and Neurology Mayo Clinic College of Medicine
Chapter 3. Structure and Function of the Nervous System. Copyright (c) Allyn and Bacon 2004
 Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
FORENSIC SCIENCE NEWSLETTER Forensic Pathology and Neuropathology. William A. Cox, M.D., FCAP.
 NEUROPATHOLOGY FORENSIC SCIENCE NEWSLETTER Forensic Pathology and Neuropathology William A. Cox, M.D., FCAP www.forensicjournals.cm May 15, 2016 This issue of the Forensic Science Newsletter will address
NEUROPATHOLOGY FORENSIC SCIENCE NEWSLETTER Forensic Pathology and Neuropathology William A. Cox, M.D., FCAP www.forensicjournals.cm May 15, 2016 This issue of the Forensic Science Newsletter will address
Concussion. Concussion is a disturbance of brain function caused by a direct or indirect force to the head.
 Concussion Concussion is a disturbance of brain function caused by a direct or indirect force to the head. Disturbances of brain tissue is largely related to neurometabolic dysfunction rather then structural
Concussion Concussion is a disturbance of brain function caused by a direct or indirect force to the head. Disturbances of brain tissue is largely related to neurometabolic dysfunction rather then structural
Mild Traumatic Brain Injury
 Mild Traumatic Brain Injury Concussions This presentation is for information purposes only, not for any commercial purpose, and may not be sold or redistributed. David Wesley, M.D. Outline Epidemiology
Mild Traumatic Brain Injury Concussions This presentation is for information purposes only, not for any commercial purpose, and may not be sold or redistributed. David Wesley, M.D. Outline Epidemiology
Traumatic Brain Injury: Overview & Perspectives. September 27, 2013 Louis T. Giron, Jr., M.D Neurology Section VAMC Kansas City, Missouri
 Traumatic Brain Injury: Overview & Perspectives September 27, 2013 Louis T. Giron, Jr., M.D Neurology Section VAMC Kansas City, Missouri Johnny, I hardly knew ye. Popular English song, 19 th Century Definition
Traumatic Brain Injury: Overview & Perspectives September 27, 2013 Louis T. Giron, Jr., M.D Neurology Section VAMC Kansas City, Missouri Johnny, I hardly knew ye. Popular English song, 19 th Century Definition
Blood Biomarkers for Brain Injury. Dianne Langford Associate Professor Department of Neuroscience Lewis Katz School of Medicine at Temple University
 Blood Biomarkers for Brain Injury Dianne Langford Associate Professor Department of Neuroscience Lewis Katz School of Medicine at Temple University TOPICS COVERED What are we measuring? How do markers
Blood Biomarkers for Brain Injury Dianne Langford Associate Professor Department of Neuroscience Lewis Katz School of Medicine at Temple University TOPICS COVERED What are we measuring? How do markers
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative
 ORIGINAL RESEARCH E. Matsusue S. Sugihara S. Fujii T. Kinoshita T. Nakano E. Ohama T. Ogawa Cerebral Cortical and White Matter Lesions in Amyotrophic Lateral Sclerosis with Dementia: Correlation with MR
ORIGINAL RESEARCH E. Matsusue S. Sugihara S. Fujii T. Kinoshita T. Nakano E. Ohama T. Ogawa Cerebral Cortical and White Matter Lesions in Amyotrophic Lateral Sclerosis with Dementia: Correlation with MR
Concussion Management
 Concussion Management Bill Meehan, MD Director, Micheli Center for Sports Injury Prevention Co-Director, Football Players Health Study at Harvard Division of Sports Medicine, Boston Children s Hospital
Concussion Management Bill Meehan, MD Director, Micheli Center for Sports Injury Prevention Co-Director, Football Players Health Study at Harvard Division of Sports Medicine, Boston Children s Hospital
PT/MR evaluation of athletes with postconcussion syndrome
 PT/MR evaluation of athletes with postconcussion syndrome Niklas Marklund Professor@Dept. of Clinical Sciences Lund, Neurosurgery, Lund University, Sweden Sports-related concussions- basic facts Recovery
PT/MR evaluation of athletes with postconcussion syndrome Niklas Marklund Professor@Dept. of Clinical Sciences Lund, Neurosurgery, Lund University, Sweden Sports-related concussions- basic facts Recovery
Chronic Traumatic Encephalopathy
 Chronic Traumatic Encephalopathy Sandeep Gupta MD PGY-4 Clinical Instructor House Staff, Department of Psychiatry The Ohio State Wexner Medical Center 01/09/13 Objectives 1. Examine history of disorder
Chronic Traumatic Encephalopathy Sandeep Gupta MD PGY-4 Clinical Instructor House Staff, Department of Psychiatry The Ohio State Wexner Medical Center 01/09/13 Objectives 1. Examine history of disorder
Dementia and Healthy Ageing : is the pathology any different?
 Dementia and Healthy Ageing : is the pathology any different? Professor David Mann, Professor of Neuropathology, University of Manchester, Hope Hospital, Salford DEMENTIA Loss of connectivity within association
Dementia and Healthy Ageing : is the pathology any different? Professor David Mann, Professor of Neuropathology, University of Manchester, Hope Hospital, Salford DEMENTIA Loss of connectivity within association
DEMENTIA 101: WHAT IS HAPPENING IN THE BRAIN? Philip L. Rambo, PhD
 DEMENTIA 101: WHAT IS HAPPENING IN THE BRAIN? Philip L. Rambo, PhD OBJECTIVES Terminology/Dementia Basics Most Common Types Defining features Neuro-anatomical/pathological underpinnings Neuro-cognitive
DEMENTIA 101: WHAT IS HAPPENING IN THE BRAIN? Philip L. Rambo, PhD OBJECTIVES Terminology/Dementia Basics Most Common Types Defining features Neuro-anatomical/pathological underpinnings Neuro-cognitive
According to the Centers for Disease Control and. focus Neurosurg Focus 40 (4):E5, 2016
 neurosurgical focus Neurosurg Focus 40 (4):E5, 2016 Sports-related concussions: diagnosis, complications, and current management strategies Jonathan G. Hobbs, MD, 1 Jacob S. Young, BS, 1 and Julian E.
neurosurgical focus Neurosurg Focus 40 (4):E5, 2016 Sports-related concussions: diagnosis, complications, and current management strategies Jonathan G. Hobbs, MD, 1 Jacob S. Young, BS, 1 and Julian E.
Clinical diagnosis and risk factors for chronic traumatic encephalopathy
 Boston University OpenBU Theses & Dissertations http://open.bu.edu Boston University Theses & Dissertations 2016 Clinical diagnosis and risk factors for chronic traumatic encephalopathy Montenigro, Philip
Boston University OpenBU Theses & Dissertations http://open.bu.edu Boston University Theses & Dissertations 2016 Clinical diagnosis and risk factors for chronic traumatic encephalopathy Montenigro, Philip
FDG-PET e parkinsonismi
 Parkinsonismi FDG-PET e parkinsonismi Valentina Berti Dipartimento di Scienze Biomediche, Sperimentali e Cliniche Sez. Medicina Nucleare Università degli Studi di Firenze History 140 PubMed: FDG AND parkinsonism
Parkinsonismi FDG-PET e parkinsonismi Valentina Berti Dipartimento di Scienze Biomediche, Sperimentali e Cliniche Sez. Medicina Nucleare Università degli Studi di Firenze History 140 PubMed: FDG AND parkinsonism
TRAUMATIC BRAIN INJURY: CHRONIC TRAUMATIC ENCEPHALOPATHY
 TRAUMATIC BRAIN INJURY: CHRONIC TRAUMATIC ENCEPHALOPATHY Lasting Impacts of mtbi Global EOD Symposium and Exposition 2018 Alexandra M Bradley, MALS, MCHES, ATC Client Informatics Consultant Veterans CRG
TRAUMATIC BRAIN INJURY: CHRONIC TRAUMATIC ENCEPHALOPATHY Lasting Impacts of mtbi Global EOD Symposium and Exposition 2018 Alexandra M Bradley, MALS, MCHES, ATC Client Informatics Consultant Veterans CRG
Prof. Saeed Abuel Makarem & Dr.Sanaa Alshaarawy
 Prof. Saeed Abuel Makarem & Dr.Sanaa Alshaarawy 1 Objectives By the end of the lecture, you should be able to: Describe the anatomy and main functions of the thalamus. Name and identify different nuclei
Prof. Saeed Abuel Makarem & Dr.Sanaa Alshaarawy 1 Objectives By the end of the lecture, you should be able to: Describe the anatomy and main functions of the thalamus. Name and identify different nuclei
Organization of the nervous system. [See Fig. 48.1]
![Organization of the nervous system. [See Fig. 48.1] Organization of the nervous system. [See Fig. 48.1]](/thumbs/90/103926552.jpg) Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Introduction, use of imaging and current guidelines. John O Brien Professor of Old Age Psychiatry University of Cambridge
 Introduction, use of imaging and current guidelines John O Brien Professor of Old Age Psychiatry University of Cambridge Why do we undertake brain imaging in AD and other dementias? Exclude other causes
Introduction, use of imaging and current guidelines John O Brien Professor of Old Age Psychiatry University of Cambridge Why do we undertake brain imaging in AD and other dementias? Exclude other causes
TBI Update 2018: Is Dementia a Common Endpoint after TBI?
 TBI Update 2018: Is Dementia a Common Endpoint after TBI? David X. Cifu, MD Senior TBI Specialist, U.S. Department of Veterans Affairs Associate Dean for Innovation and System Integrations, Virginia Commonwealth
TBI Update 2018: Is Dementia a Common Endpoint after TBI? David X. Cifu, MD Senior TBI Specialist, U.S. Department of Veterans Affairs Associate Dean for Innovation and System Integrations, Virginia Commonwealth
The ABCs of Dementia Diagnosis
 The ABCs of Dementia Diagnosis Dr. Robin Heinrichs, Ph.D., ABPP Board Certified Clinical Neuropsychologist Associate Professor, Psychiatry & Behavioral Sciences Director of Neuropsychology Training What
The ABCs of Dementia Diagnosis Dr. Robin Heinrichs, Ph.D., ABPP Board Certified Clinical Neuropsychologist Associate Professor, Psychiatry & Behavioral Sciences Director of Neuropsychology Training What
Lecture 42: Final Review. Martin Wessendorf, Ph.D.
 Lecture 42: Final Review Martin Wessendorf, Ph.D. Lecture 33 cortex Heilbronner 5 lobes of the cortex Lateral view (left side) Mid-saggital view (right side) Cellular organization of cortex White matter
Lecture 42: Final Review Martin Wessendorf, Ph.D. Lecture 33 cortex Heilbronner 5 lobes of the cortex Lateral view (left side) Mid-saggital view (right side) Cellular organization of cortex White matter
The Primary Care Guide To Understanding The Role Of Diabetes As A Risk Factor For Cognitive Loss Or Dementia In Adults
 The Primary Care Guide To Understanding The Role Of Diabetes As A Risk Factor For Cognitive Loss Or Dementia In Adults. Introduction Glucose intolerance is common in older individuals and this metabolic
The Primary Care Guide To Understanding The Role Of Diabetes As A Risk Factor For Cognitive Loss Or Dementia In Adults. Introduction Glucose intolerance is common in older individuals and this metabolic
TRAUMATIC BRAIN INJURY
 Psychiatry and Addictions Case Conference UW Medicine Psychiatry and Behavioral Sciences TRAUMATIC BRAIN INJURY GARY STOBBE, MD UNIVERSITY OF WASHINGTON GENERAL DISCLOSURES The University of Washington
Psychiatry and Addictions Case Conference UW Medicine Psychiatry and Behavioral Sciences TRAUMATIC BRAIN INJURY GARY STOBBE, MD UNIVERSITY OF WASHINGTON GENERAL DISCLOSURES The University of Washington
Cerebral Cortex 1. Sarah Heilbronner
 Cerebral Cortex 1 Sarah Heilbronner heilb028@umn.edu Want to meet? Coffee hour 10-11am Tuesday 11/27 Surdyk s Overview and organization of the cerebral cortex What is the cerebral cortex? Where is each
Cerebral Cortex 1 Sarah Heilbronner heilb028@umn.edu Want to meet? Coffee hour 10-11am Tuesday 11/27 Surdyk s Overview and organization of the cerebral cortex What is the cerebral cortex? Where is each
Type 2 Diabetes and Brain Disease in Older Adults. Erin L. Abner, PhD, MPH Asst. Professor University Of Kentucky
 Type 2 Diabetes and Brain Disease in Older Adults Erin L. Abner, PhD, MPH Asst. Professor University Of Kentucky Disclosures to Participants Requirements for Successful Completion: For successful completion,
Type 2 Diabetes and Brain Disease in Older Adults Erin L. Abner, PhD, MPH Asst. Professor University Of Kentucky Disclosures to Participants Requirements for Successful Completion: For successful completion,
Chronic Traumatic Encephalopathy in Athletes: Progressive Tauopathy After Repetitive Head Injury
 Chronic Traumatic Encephalopathy in Athletes: Progressive Tauopathy After Repetitive Head Injury The Harvard community has made this article openly available. Please share how this access benefits you.
Chronic Traumatic Encephalopathy in Athletes: Progressive Tauopathy After Repetitive Head Injury The Harvard community has made this article openly available. Please share how this access benefits you.
Caring Sheet #11: Alzheimer s Disease:
 CARING SHEETS: Caring Sheet #11: Alzheimer s Disease: A Summary of Information and Intervention Suggestions with an Emphasis on Cognition By Shelly E. Weaverdyck, PhD Introduction This caring sheet focuses
CARING SHEETS: Caring Sheet #11: Alzheimer s Disease: A Summary of Information and Intervention Suggestions with an Emphasis on Cognition By Shelly E. Weaverdyck, PhD Introduction This caring sheet focuses
10/3/2016. T1 Anatomical structures are clearly identified, white matter (which has a high fat content) appears bright.
 H2O -2 atoms of Hydrogen, 1 of Oxygen Hydrogen just has one single proton and orbited by one single electron Proton has a magnetic moment similar to the earths magnetic pole Also similar to earth in that
H2O -2 atoms of Hydrogen, 1 of Oxygen Hydrogen just has one single proton and orbited by one single electron Proton has a magnetic moment similar to the earths magnetic pole Also similar to earth in that
Subject Index. Band of Giacomini 22 Benton Visual Retention Test 66 68
 Subject Index Adams, R.D. 4 Addenbrooke s Cognitive Examination 101 Alzheimer s disease clinical assessment histological imaging 104 neuroimaging 101 104 neuropsychological assessment 101 clinical presentation
Subject Index Adams, R.D. 4 Addenbrooke s Cognitive Examination 101 Alzheimer s disease clinical assessment histological imaging 104 neuroimaging 101 104 neuropsychological assessment 101 clinical presentation
Anatomy and Physiology (Bio 220) The Brain Chapter 14 and select portions of Chapter 16
 Anatomy and Physiology (Bio 220) The Brain Chapter 14 and select portions of Chapter 16 I. Introduction A. Appearance 1. physical 2. weight 3. relative weight B. Major parts of the brain 1. cerebrum 2.
Anatomy and Physiology (Bio 220) The Brain Chapter 14 and select portions of Chapter 16 I. Introduction A. Appearance 1. physical 2. weight 3. relative weight B. Major parts of the brain 1. cerebrum 2.
Concussion is the most common type of
 CMAJ Early release, published at www.cmaj.ca on July 22, 2013. Subject to revision. Practice Primer Concussions and their consequences: current diagnosis, management and prevention Charles H. Tator MD
CMAJ Early release, published at www.cmaj.ca on July 22, 2013. Subject to revision. Practice Primer Concussions and their consequences: current diagnosis, management and prevention Charles H. Tator MD
Overview of neurological changes in Alzheimer s disease. Eric Karran
 Overview of neurological changes in Alzheimer s disease Eric Karran Alzheimer s disease Alois Alzheimer 1864-1915 Auguste D. 1850-1906 Case presented November 26 th 1906 Guildford Talk.ppt 20 th March,
Overview of neurological changes in Alzheimer s disease Eric Karran Alzheimer s disease Alois Alzheimer 1864-1915 Auguste D. 1850-1906 Case presented November 26 th 1906 Guildford Talk.ppt 20 th March,
1 MS Lesions in T2-Weighted Images
 1 MS Lesions in T2-Weighted Images M.A. Sahraian, E.-W. Radue 1.1 Introduction Multiple hyperintense lesions on T2- and PDweighted sequences are the characteristic magnetic resonance imaging (MRI) appearance
1 MS Lesions in T2-Weighted Images M.A. Sahraian, E.-W. Radue 1.1 Introduction Multiple hyperintense lesions on T2- and PDweighted sequences are the characteristic magnetic resonance imaging (MRI) appearance
Visual Rating Scale Reference Material. Lorna Harper Dementia Research Centre University College London
 Visual Rating Scale Reference Material Lorna Harper Dementia Research Centre University College London Background The reference materials included in this document were compiled and used in relation to
Visual Rating Scale Reference Material Lorna Harper Dementia Research Centre University College London Background The reference materials included in this document were compiled and used in relation to
Parkinson e decadimento cognitivo. Stelvio Sestini
 Parkinson e decadimento cognitivo Stelvio Sestini Patients with PD can develop a spectrum of cognitive symptoms Heterogeneity of cognitive deficits The cognitive symptoms can evolve to dementia (Mov Disorder
Parkinson e decadimento cognitivo Stelvio Sestini Patients with PD can develop a spectrum of cognitive symptoms Heterogeneity of cognitive deficits The cognitive symptoms can evolve to dementia (Mov Disorder
Learning Objectives. Sports Concussion. Learning Objectives (Cont.) Definitions. Marc Norman, Ph.D. & Amanda Gooding, Ph.D. Department of Psychiatry
 Learning Objectives Sports Concussion Marc Norman, Ph.D. & Amanda Gooding, Ph.D. Department of Psychiatry Definitions Mechanisms Neuroimaging Assessment Sideline Clinic Impact Symptoms Vestibular Cognitive
Learning Objectives Sports Concussion Marc Norman, Ph.D. & Amanda Gooding, Ph.D. Department of Psychiatry Definitions Mechanisms Neuroimaging Assessment Sideline Clinic Impact Symptoms Vestibular Cognitive
Piramal Imaging to Present New Research in PET Imaging at Society of Nuclear Medicine and Molecular Imaging 2017 Annual Meeting
 FOR IMMEDIATE RELEASE Media Contacts: Nicole Fletcher Piramal Imaging nicole.fletcher@piramal.com (857) 202-1122 Piramal Imaging to Present New Research in PET Imaging at Society of Nuclear Medicine and
FOR IMMEDIATE RELEASE Media Contacts: Nicole Fletcher Piramal Imaging nicole.fletcher@piramal.com (857) 202-1122 Piramal Imaging to Present New Research in PET Imaging at Society of Nuclear Medicine and
Lecture 13. The Nervous System. Lecture 13
 Lecture 13 The Nervous System Lecture 13 1. Introduction 2. Functions of the Nervous System 3. Neurons 4. CNS Brain and Spinal Cord 5. Peripheral Nervous System 6. Nervous System Health Concerns 1 The
Lecture 13 The Nervous System Lecture 13 1. Introduction 2. Functions of the Nervous System 3. Neurons 4. CNS Brain and Spinal Cord 5. Peripheral Nervous System 6. Nervous System Health Concerns 1 The
Evolving concepts of chronic traumatic encephalopathy as a neuropathological entity
 Neuropathology and Applied Neurobiology (2017), 43, 467 476 doi: 10.1111/nan.12425 Evolving concepts of chronic traumatic encephalopathy as a neuropathological entity H. Ling*,,, J. W. Neal and T. Revesz*,,
Neuropathology and Applied Neurobiology (2017), 43, 467 476 doi: 10.1111/nan.12425 Evolving concepts of chronic traumatic encephalopathy as a neuropathological entity H. Ling*,,, J. W. Neal and T. Revesz*,,
Battlefield to Ball Field: The Science, Myth and Reality of Concussion
 Battlefield to Ball Field: The Science, Myth and Reality of Concussion David X. Cifu, MD Professor and Chairman Virginia Commonwealth University Senior Traumatic Brain Injury Expert U.S. Department of
Battlefield to Ball Field: The Science, Myth and Reality of Concussion David X. Cifu, MD Professor and Chairman Virginia Commonwealth University Senior Traumatic Brain Injury Expert U.S. Department of
Recent publications using the NACC Database. Lilah Besser
 Recent publications using the NACC Database Lilah Besser Data requests and publications Using NACC data Number of requests by year Type 2009 2010 2011 2012 2013 2014 2015 Data files* 55 85 217 174 204
Recent publications using the NACC Database Lilah Besser Data requests and publications Using NACC data Number of requests by year Type 2009 2010 2011 2012 2013 2014 2015 Data files* 55 85 217 174 204
IT S ALL IN YOUR HEAD!
 IT S ALL IN YOUR HEAD! CARING FOR CONCUSSIONS IN YOUR COMMUNITY Stephen K Stacey, DO CPT, MC, USA OUTLINE Definition Epidemiology Diagnosis Evaluation Recovery Sequelae Prevention Resources for providers
IT S ALL IN YOUR HEAD! CARING FOR CONCUSSIONS IN YOUR COMMUNITY Stephen K Stacey, DO CPT, MC, USA OUTLINE Definition Epidemiology Diagnosis Evaluation Recovery Sequelae Prevention Resources for providers
