EXPERIMENTAL PROCEDURES
|
|
|
- Darcy Cook
- 5 years ago
- Views:
Transcription
1 Neuroscience 15 (004) FLUCTUATIONS IN SOMATOSENSORY RESPONSIVENESS AND BASELINE FIRING RATES OF NEURONS IN THE LATERAL STRIATUM OF FREELY MOVING RATS: EFFECTS OF INTRANIGRAL APOMORPHINE V. F. PROKOPENKO, A. P. PAWLAK AND M. O. WEST* Department of Psychology, Rutgers University, New Brunswick, NJ, USA Abstract Somatosensory responsiveness and baseline firing rates of 10 striatal neurons were studied in freely moving rats. For individual neurons, mean levels of responsiveness and baseline firing fluctuated unpredictably in direction and magnitude and independently of each other throughout an experiment. Following microinjections of apomorphine into the substantia nigra, which were used as a means of reducing nigral output activity, the magnitude of fluctuations in striatal somatosensory responsiveness significantly increased, while the magnitude of fluctuations in baseline firing was unaltered. The receptive zones of 54 neurons studied in control experiments remained stable, whereas receptive zones changed in 1 of 5 neurons studied after apomorphine microinjection. Normal nigrostriatal dopamine transmission appears to selectively restrict the magnitude of fluctuations in responsiveness of striatal neurons to corticostriatal synaptic input and may exert additional control over afferent projections from cutaneous receptive zones to these neurons. 004 IBRO. Published by Elsevier Ltd. All rights reserved. Key words: basal ganglia, corticostriatal, single neuron recording, substantia nigra, dopamine, microinjection. The somatosensory responsiveness of striatal neurons in behaving animals (Carelli and West, 1991; Brown, 199; Cho and West, 1997) provides an analytical tool for studying the normal operation of mechanisms governing striatal output activity, given the evidence that phasically responsive neurons are medium spiny, projection neurons (Kimura, 1990). Our aim was to examine, in freely moving rats, the time course of alterations in somatosensory responses and baseline firing of lateral striatal neurons in the normal state and during the transient lowering of general nigral output activity and nigrostriatal dopamine transmission. This was accomplished by microinjecting small doses of the dopamine receptor agonist, apomorphine (APO), into the substantia nigra (SN) in order to partially model the actions of intrinsic nigral dopamine release (Geffen et al., 1976; Cheramy et al., 1981). It was assumed that intranigral APO acting via D receptors (Seeman, 1995) on SNc neurons, which are highly sensitive to direct application of dopamine or APO (Skirboll et al., 1979; Okuyama et al., 1986; Hyland et al., 00), could depress monosynaptic nigrostriatal dopamine transmission that would be reflected in the evoked activity of striatal neurons (Freund et al., 1984). It was also assumed that intranigral APO would influence GABAergic neurons of SNr via D 1 -receptors (Seeman, 1995) on striato-nigral terminals (Martin and Waszczak, 1994; Radnikow and Misgeld, 1998) that could produce, in turn, changes in baseline activity of striatal neurons. Nigrostriatal influences regulating the processing of information flow from neocortex, thalamus, and other brain structures to the basal ganglia are mediated monosynaptically by dopaminergic neurons of substantia nigra pars compacta (SNc), and polysynaptically by GABAergic neurons of substantia nigra pars reticulata (SNr). The latter project to nonspecific midline-intralaminar nuclei of the thalamus, which, in turn, send abundantly collateralized axons to the striatum and to striatal afferent sources in cortex (Deschênes et al., 1996; Mengual et al., 1999; Gerfen, 000; Nakano, 000; Tsumory et al., 000). *Correspondence to: M. O. West, Department of Psychology, Rutgers, The State University of New Jersey, 15 Frelinghuysen Road, Piscataway, NJ , USA. Tel: ; fax: address: markwest@rci.rutgers.eduauname;(m. O. West). Abbreviations: APO, apomorphine; PSTH, peristimulus time histogram; SN, substantia nigra; SNc, substantia nigra pars compacta; SNr, substantia nigra pars reticulata; RZ, receptive zone /04$ IBRO. Published by Elsevier Ltd. All rights reserved. doi: /j.neuroscience EXPERIMENTAL PROCEDURES Experiments were conducted in accord with guidelines of the Institutional Animal Care and Use Committee of Rutgers University, the National Institutes of Health, and United States Department of Agriculture, including minimizing the number of animals used, and minimizing pain and suffering. Data were collected from 6 male Long-Evans rats ( g; Charles River Laboratories, Wilmington, MA, USA). Animals were surgically prepared for recording in the awake, unrestrained state by implantation of microdrive assembly (Josef Biela Engineering, Anaheim, CA, USA) as described previously (Carelli and West, 1991). During surgery, two bases (the first for attaching a microdrive containing a tungsten microelectrode [10 1 M ; Haer, Brunswick, ME, USA] and the second for attaching a removable intracerebral microinjector [Prokopenko at al., 1997]) were cemented to the skull above holes centered: 1) over lateral striatum, either side, perpendicular to level skull and ) over SN at an angle of 16.5 o from vertical, toward midline, ipsilateral to the microdrive base.
2 1078 V. F. Prokopenko et al. / Neuroscience 15 (004) Fig. 1. Changes in baseline firing and somatosensory responsiveness. Each column represents the data of a single striatal neuron. Left two columns: NORMAL group; right two columns: APO group. Each row (raster display and PSTH) depicts the neuron s activity recorded at one of the following time points: 0 min (onset of the experiment), 10 min, 40 min or 100 min. For a given row, 50 stimulus events contributed to the displayed firing patterns. Insets indicate position of cutaneous RZ (arrow) for that neuron. Asterisk in third column depicts shift in the neuron s RZ following APO injection. Recording sessions began 1 week after surgery. Extracellularly recorded action potentials of single neurons were amplified, filtered (bandpass Hz) and two-stage discriminated (West, 1998). For each striatal neuron that exhibited spontaneous activity, impulses were delivered in parallel to a computer and to the experimenter s headphones during visually monitored testing of responsiveness to mechanical cutaneous stimulation. Stimulation was delivered via a handheld probe ( mm in diameter), calibrated to deliver 4 g of force. The probe traveled a distance up to 3 cm in 0.15 s. Monotonous, unidirectional, stable stimuli were presented 5 10 s apart. Instantaneously on contact of the bipolar probe with the animal, a DC pulse was delivered to the computer to generate a time-stamp, or node, of the approximate onset of each stimulus. Neurons were selected if they exhibited cutaneous responsiveness and little or no correlation with active movements (e.g. neurons responsive to cutaneous stimulation on the head; Cho and West, 1997), in order to avoid difficulties in interpreting neural data in the APO group, in which abnormal movement might produce altered somatosensory feedback. For each neuron, the approximate borders of the receptive zone (RZ) in which cutaneous stimulation produced stable, repetitive responses, and any changes of this zone during the session, were recorded. Ten to 0 stimuli were applied outside the RZ to verify unresponsiveness. During each experiment, the activity of one neuron was tested at four periods. The first was termed the time 0 period. The three remaining periods occurred at 10, 40 and 100 min after 1) the end of the time 0 period, or ) the start of microinjection. Peristimulus time histograms (PSTHs) and raster displays were constructed around stimulus onset (50 stimuli were delivered over approximately 6 min at each time period). Evoked firing rates (impulses/s) were calculated during the evoked response epoch defined for each neuron by visual inspection of the time 0 PSTH. Baseline firing rates were calculated from each PSTH during the interval from 0.5 to 0.15 s before stimulus onset. A removable microinjector was used to deliver fluids (1 l, l per minute) to the SN. The interval between two experiments for each animal was at least 4 days. Each rat received nine or fewer microinjections (one injection per experiment), and histological examination revealed no damage to SN. Single intracerebral microinjections of APO (R( )-Apomorphine HCl; RBI (Nantick, MA, USA); g/ l; ph 7.0) solution were given in each APO experiment (APO group; N 48 neurons). The drug was dissolved in saline buffer (Dulbecco et al., 1979) with a supplement of 0.1% weight/volume ascorbic acid. Microinjections (same volume) of clear Dulbecco s saline buffer were administered in the second group of experiments (N 30 neurons). In the third group, injection manipulations with the empty device on the rat s head were simulated (N 4 neurons). The latter two groups did not statistically differ from each other in any parameter, which made it possible to combine them into one NORMAL group. Each animal participated in APO and NORMAL experiments. After the last experiment, each animal was anesthetized with sodium pentobarbital (150 mg/kg). A low-impedance insulated wire (00 m diameter) was placed in the following: 1) in the same location as that at which a particular neuronal recording had been obtained and ) through the microinjector base at the depth of the lowered cannula. Electrolytic lesions (0.03 ma; 10 s) were made
3 V. F. Prokopenko et al. / Neuroscience 15 (004) at the wire tips. Procedures for perfusion, histology, and identification of locations of electrolytic lesions were described previously (Carelli and West, 1991). Multiple t-tests, ANOVAs, and were used. Differences were judged significant at the 0.05 level. Neuronal activity RESULTS The firing of 10 neurons responsive to somatosensory cutaneous stimulation was studied in 10 experiments. According to histological verification, all neurons were recorded from the dorsolateral striatum (Bregma AP mm, ML mm, and DV mm from cortex surface) and the microinjector cannula tips were positioned in the SN (Bregma AP 4.8 to 5.8 mm, ML 1.8.5, DV ). At the beginning of experiments, NORMAL and APO groups showed no significant differences in baseline firing or responsiveness (P 0.05). The distribution of mean baseline and response firing rates at time 0 is shown in Fig. A. Baseline firing rate Spontaneous, non-periodic changes (fluctuations) in baseline firing (mean impulses/s) of both polarities (increases or decreases) were observed (Fig. 1). The magnitudes of these fluctuations over time are shown in Fig. B. Mean baseline activity tended to decrease non-significantly in both groups at 10 min and 40 min and to be slightly decreased or unchanged at 100 min. Separation of these fluctuations into increases versus decreases revealed no differences between mean baseline firing in NORMAL and APO groups at any time point (Fig. C). The proportions of neurons exhibiting changes in baseline firing and responsiveness are shown in Table 1. Fluctuations in neuronal responsiveness Fig.. Baseline firing (left column) and somatosensory responsiveness (right column). (A) Distribution of mean baseline and response firing rates of all neurons registered at time 0. (B) Time course of fluctuations in baseline firing and somatosensory responsiveness of individual neurons. Calculated changes in firing [B/(A B)] are displayed as a function of time. Each solid line represents a single neuron. Values 0.5 indicate increases; values 0.5 indicate decreases, relative to time zero. Time course of changes in baseline firing and somatosensory responsiveness of individual neurons in NORMAL group (top row) and APO group (bottom row). The formula used was [B/(A B)], in which A was the firing rate of the neuron at time 0 min and B was the firing rate of that neuron at subsequent times: 10 min, 40 min or 100 min. A value equal to 0.5 is indicative of no change between the B and A periods. (C) Time course of mean values of baseline and evoked neuronal activity. Mean calculated change in firing [B/(A B)] is displayed as a function of time (empty circles, NORMAL group; filled circles, APO group). Top row: mean values of neuronal activity averaged across all neurons. Bottom row: mean values of increases ( 0.5) and decreases ( 0.5), averaged separately at each time point. Asterisks indicate significant differences between NORMAL and APO groups. Fluctuations were also observed in neuronal responsiveness evoked by somatosensory cutaneous stimulation over time in all neurons studied. These fluctuations exhibited considerable variability among neurons in direction and magnitude (Fig. B, C). Averaged together, the mean magnitude of responsiveness showed insignificant decreases throughout the experiment (Fig. B). In comparison with the NORMAL group, the most marked effect of intranigral APO microinjection was the significant increase in magnitude of both positive and negative fluctuations in responsiveness to cutaneous stimulation (Figs. and 3). Each neuron recorded at all four time points in both groups (N 81) was rated as to whether its baseline and evoked firing changed in the same direction at each time point relative to the preceding time point. Thus, a neuron could receive a rating of same at none, one, two, or all three time points. Only 6% of neurons were rated as same at all three time points in the NORMAL group (i.e. chance level), compared with 19% in the APO group, which was not significantly different between groups. Thus, fluctuations in baseline and evoked firing of individual neurons varied independently in their directions and magnitudes (e.g. Figs. 1 and 3). In addition, three striatal neurons in the APO group responded to stimulation of two spatially separate RZs. The time course and polarity of changes in the same neuron s responsiveness to stimulation of each RZ were independent.
4 1080 V. F. Prokopenko et al. / Neuroscience 15 (004) Table 1. Proportions of neurons exhibiting changes in baseline firing and responsiveness a Activity Baseline firing Responsiveness Groups NORMAL APO NORMAL APO Post time 0 interval (min) Increased firing (%) 31 (57.4) 0 (40.8) 1 (31.6) 16 (33.3) 16 (34.0) 4 (54.5) 17 (31.5) 16 (3.7) 13 (34.) 11 (.9) 1 (5.5) 18 (40.9) No changes (%) 1 (1.9) (4.1) (5.3) (4.) 3 (6.4) 1 (.3) (3.7) 1 (.0) (5.3) 0 (0.0) 0 (0.0) (4.55) Decreased firing (%) (40.7) 7 (55.1) 4 (63.1) 30 (6.5) 8 (59.6) 19 (43.) 35 (64.8) 3 (65.3) 3 (60.5) 37 (77.1) 9 (74.5) 4 (54.55) Number of tested cells a There were no significant differences between NORMAL and APO groups ( -test of equality; P , corrected for six comparisons). Receptive zones RZs of 79 neurons were located on the contralateral surface of the animal s head, mainly on the animal s muzzle (71 neurons). The remaining 3 neurons exhibited RZs on other, contralateral, body parts. Ninety-nine neurons were activated by stimulation of only one RZ, while three neurons exhibited responses to stimulation of two non-neighboring RZs within the same body part. No changes were observed in the parameters of RZs for neurons in the NORMAL group throughout the time course of the experiment. In contrast, after intra-nigral microinjection of APO, changes in RZ position and/or dimensions were found in 1 of 5 cases tested. In five experiments, the RZ increased in dimensions; in five others it decreased, and in two experiments the RZ changed both its dimensions and location on the body. In these 1 cases, there was no relationship between the polarity of changes in RZ dimensions and the polarity of simultaneous changes in evoked neuronal activity (Fig. 1). Behavioral changes Muscular impairment after APO microinjection was observed in five experiments: contralateral akinesia in four cases and an observable decline in muscle tone in the other case. Nonetheless, changes in neuronal baseline firing or evoked activity in these experiments did not differ in any observable way from others in the APO group. DISCUSSION Normal baseline and evoked firing For individual somatosensory-responsive striatal neurons, the fluctuations in baseline firing and responsiveness, reported for the first time here, appear to be normal and natural. They were independent of each other and unpredictable in direction and magnitude during the time course of experiments. This independence suggests that the mechanisms underlying general excitability versus responsiveness to somatosensory cortical input of striatal neurons are largely separate, and remain to be investigated. Effects of intranigral APO microinjection Fig. 3. Comparison of changes in baseline firing and somatosensory responsiveness of each neuron. At each time point (row), each dot represents a single neuron s calculated change [B/(A B)] in responsiveness plotted against its change in baseline firing. Left column: NORMAL group; right column: APO group. The slight, insignificant increase in magnitudes of positive and negative fluctuations in baseline firing between NOR- MAL and APO groups corroborates the minimal influence of altered intrastriatal dopamine release observed on baseline striatal firing (Abercrombie and Jacobs, 1985). Our finding suggests a minor indirect influence on somato-
5 V. F. Prokopenko et al. / Neuroscience 15 (004) sensory striatal neurons of SNr-intralaminar thalamic-striatal projections that may have been altered by intranigral APO injection. At the same time, an APO influence on SNr neurons could partially explain some motoric disturbances observed, i.e. via altered SNr transmission to motor thalamo-cortical neurons. The main effect of decreasing striatal dopamine transmission was to selectively derestrict the magnitudes of fluctuations in striatal somatosensory responsiveness. Fluctuations in both directions were greater in the APO group, compared with the NORMAL group, followed by a return to a lack of difference between groups at 100 min. Thus, dopamine appears to be responsible for the normal restriction on natural fluctuations in striatal responsiveness to synaptic input from somatosensory cortex. Moreover, the changes of RZs in the APO group suggest that some striatal neurons may have undergone a reorganization of their effective afferent somatosensory inputs in the absence of dopamine transmission. After APO microinjection, the increased magnitude of fluctuations in somatosensory responsiveness, coupled with the minimal changes in baseline firing, suggests that nigrostriatal dopamine transmission mainly influences the somatosensory inputs to striatal spiny type II projection neurons (Kimura, 1990). The present data demonstrate dopamine s ability to modulate corticostriatal transmission, consistent with the suggestion of Freund et al. (1984), based on the pattern by which nigrostriatal and corticostriatal terminals innervate spines of medium spiny neurons. The actions reported here reaffirm the complexity of dopamine s modulatory actions in striatum. It has been suggested that the nigrostriatal dopaminergic system exerts tonic effects (Chase and Oh, 000), such as augmented baseline firing due to decreased striatal dopamine levels (Kish et al., 1999). During anesthesia, influences of dopamine-like agents on striatal neurons were mainly inhibitory in nature, but some excitatory actions have been shown. Stimulation of SNc attenuated the excitatory responses to cortical stimulation in the majority of striatal neurons but enhanced it in a minority of these neurons. Both effects were reduced or abolished by systemic administration of dopamine antagonists (Hirata et al., 1984; Vives and Mogenson, 1986). Alternatively, dopaminergic input may modulate the phasic responses of striatal neurons (Horvitz, 00). Both excitatory and inhibitory responses of striatal neurons to repetitive sciatic nerve stimulation were decreased by systemically injected amphetamine, while baseline activity showed little change (Abercrombie and Jacobs, 1985). This suggested that dopamine action upon target neurons might be more complex than simple inhibition or excitation, possibly modulating the intensity of neuronal responses to incoming signals. Iontophoretically applied dopamine inhibited glutamate-induced and acetylcholine-induced excitation of striatal neurons (Brown and Arbuthnott, 1983). However, dopamine ejected with lower current could potentiate glutamate-evoked neuronal activity, an effect that required simultaneous stimulation of both D 1 and D receptors (Hu and White, 1997). Under behaviorally relevant conditions, dopamine acted mainly via D 1 -receptors to provide a restraining effect on responsiveness of striatal neurons to glutamate-mediated excitatory input (Kiyatkin and Rebec, 1999). These data and our results are consistent with the interpretation that dopamine s modulatory actions include limiting the range of responsiveness of striatal neurons to cortical input. Dopamine may exert some dynamic control over the cutaneous RZ representation for each striatal neuron. Our findings regarding the influence of SNc dopamine neurons on striatal somatosensory function encourage further investigation of these complex mechanisms in freely moving animals using intranigral and intrastriatal microinjections of highly specific agonists and antagonists of dopamine receptors. Acknowledgements Supported by National Institute on Drug Abuse grant DA and the Charles and Johanna Busch Foundation. We thank Dr. Anthony T. Fabbricatore for discussion of this manuscript, and Linda King for technical assistance. REFERENCES Abercrombie ED, Jacobs BL (1985) Dopaminergic modulation of sensory responses of striatal neurons: single unit studies. Brain Res 358:7 33. Brown JR, Arbuthnott GW (1983) The electrophysiology of dopamine (D ) receptors: a study of the action of dopamine on corticostriatal transmission. Neuroscience 10: Brown LL (199) Somatotopic organization in rat striatum: evidence for a combinational map. Proc Natl Acad Sci USA 89: Carelli RM, West MO (1991) Representation of the body by single neurons in the dorsolateral striatum of the awake, unrestrained rat. J Comp Neurol 309: Chase TN, Oh JD (000) Striatal dopamine- and glutamate-mediated dysregulation in experimental parkinsonism. Trends Neurosci 3: S86 S91. Cheramy A, Leviel V, Glowinski J (1981) Dendritic release of dopamine in the substantia nigra. Nature 89: Cho J, West MO (1997) Distribution of single neurons related to body parts in the lateral striatum of the rat. Brain Res 756: Deschênes M, Bourassa J, Doan VD, Parent A (1996) A single-cell study of the axonal projections arising from the posterior intralaminar thalamic nuclei in the rat. Eur J Neurosci 8: Dulbecco R, Bologna M, Unger M (1979) Differentiation of a rat mammary cell line in vitro. Proc Natl Acad Sci USA 76: Freund TF, Powell JF, Smith AD (1984) Tyrosine hydroxylase-immunoreactive boutons in synaptic contact with identified striatonigral neurons, with particular reference to dendritic spines. Neuroscience 13: Geffen LB, Jessel TM, Cuello AC, Iversen LL (1976) Release of dopamine from dendrites in rat substantia nigra. Nature 60: Gerfen CR (000) Molecular effects of dopamine on striatal-projection pathways. Trends Neurosci 3:S64 S70. Hirata K, Yim CY, Mogenson GJ (1984) Excitatory input from sensory motor cortex to neostriatum and its modification by conditioning stimulation of the substantia nigra. Brain Res 31:1 8. Horvitz JC (00) Dopamine gating of glutamatergic sensorimotor and incentive motivation input signals to the striatum. Behav Brain Res 137: Hu XT, White FJ (1997) Dopamine enhances glutamate-induced excitation of rat striatal neurons by cooperative activation of D1 and D class receptors. Neurosci Lett 4: Hyland BI, Reynolds JNJ, Hay J, Perk CG, Miller R (00) Firing
6 108 V. F. Prokopenko et al. / Neuroscience 15 (004) modes of the midbrain dopamine cells in the freely moving rat. Neuroscience 114: Kimura M (1990) Behaviorally contingent property of movement-related activity of the primate putamen. J Neurosci 63: Kish LJ, Palmer MR, Gerhardt GA (1999) Multiple single-unit recordings in the striatum of freely moving animals: effects of apomorphine and D-amphetamine in normal and unilateral 6-hydroxydopamine-lesioned rats. Brain Res 833: Kiyatkin EA, Rebec GV (1999) Striatal neuronal activity and responsiveness to dopamine and glutamate after selective blockade of D1 and D dopamine receptors in freely moving rats. J Neurosci 19: Martin LP, Waszczak BL (1994) D 1 agonist-induced excitation of substantia nigra pars reticulata neurons: mediation by D 1 receptors on striato-nigral terminals via a pertussis toxin-sensitive coupling pathway. J Neurosci 14: Mengual E, de las Heras S, Erro E, Lanciego JL, Giménez-Amaya JM (1999) Thalamic interaction between the input and the output systems of the basal ganglia. J Chem Neuroanat 16: Nakano K (000) Neural circuits and topographic organization of the basal ganglia and related regions. Brain Dev :S5 S16. Okuyama S, Shimamura H, Hashimoto S, Aihara H (1986) Electrophysiological and behavioral assessments of dopamine autoreceptor activation to apomorphine in rats. Arch Int Pharmacodyn 84: Prokopenko VF, Shevko GN, Voloshin MYa, West MO (1997) A removable peripheral device for intracerebral microinjection in freely moving rats. Brain Res Protoc : Radnikow G, Misgeld U (1998) Dopamine D 1 receptors facilitate GABA A synaptic currents in the rat substantia nigra pars reticulata. J Neurosci 18: Seeman P (1995) Dopamine receptors: clinical correlates. In: Psychopharmacology: the fourth generation of progress (Bloom FE, Kupfer DJ, eds), pp New York: Raven Press. Skirboll LR, Grace AA, Bunney BS (1979) Dopamine auto- and postsynaptic receptors: electrophysiological evidence for differential sensitivity to dopamine agonists. Science 06:80 8. Tsumory T, Yokota S, Lai H, Yasui Y (000) Monosynaptic and disynaptic projections from the substantia nigra pars reticulata to the parafascicular thalamic nucleus in the rat. Brain Res 858: Vives F, Mogenson GJ (1986) Electrophysiological study of the effects of D 1 and D dopamine antagonists on the interaction of converging inputs from the sensory-motor cortex and substantia nigra neurons in the rat. Neuroscience 17: West MO (1998) Anesthetics eliminate somatosensory-evoked discharges of neurons in the somatotopically organized sensorimotor striatum of the rat. J Neurosci 18: (Accepted 7 February 004)
Anatomy of the basal ganglia. Dana Cohen Gonda Brain Research Center, room 410
 Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
NS219: Basal Ganglia Anatomy
 NS219: Basal Ganglia Anatomy Human basal ganglia anatomy Analagous rodent basal ganglia nuclei Basal ganglia circuits: the classical model of direct and indirect pathways + Glutamate + - GABA - Gross anatomy
NS219: Basal Ganglia Anatomy Human basal ganglia anatomy Analagous rodent basal ganglia nuclei Basal ganglia circuits: the classical model of direct and indirect pathways + Glutamate + - GABA - Gross anatomy
Basal Ganglia. Introduction. Basal Ganglia at a Glance. Role of the BG
 Basal Ganglia Shepherd (2004) Chapter 9 Charles J. Wilson Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Introduction A set of nuclei in the forebrain and midbrain area in mammals, birds, and reptiles.
Basal Ganglia Shepherd (2004) Chapter 9 Charles J. Wilson Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Introduction A set of nuclei in the forebrain and midbrain area in mammals, birds, and reptiles.
Teach-SHEET Basal Ganglia
 Teach-SHEET Basal Ganglia Purves D, et al. Neuroscience, 5 th Ed., Sinauer Associates, 2012 Common organizational principles Basic Circuits or Loops: Motor loop concerned with learned movements (scaling
Teach-SHEET Basal Ganglia Purves D, et al. Neuroscience, 5 th Ed., Sinauer Associates, 2012 Common organizational principles Basic Circuits or Loops: Motor loop concerned with learned movements (scaling
Basal Ganglia George R. Leichnetz, Ph.D.
 Basal Ganglia George R. Leichnetz, Ph.D. OBJECTIVES 1. To understand the brain structures which constitute the basal ganglia, and their interconnections 2. To understand the consequences (clinical manifestations)
Basal Ganglia George R. Leichnetz, Ph.D. OBJECTIVES 1. To understand the brain structures which constitute the basal ganglia, and their interconnections 2. To understand the consequences (clinical manifestations)
COGNITIVE SCIENCE 107A. Motor Systems: Basal Ganglia. Jaime A. Pineda, Ph.D.
 COGNITIVE SCIENCE 107A Motor Systems: Basal Ganglia Jaime A. Pineda, Ph.D. Two major descending s Pyramidal vs. extrapyramidal Motor cortex Pyramidal system Pathway for voluntary movement Most fibers originate
COGNITIVE SCIENCE 107A Motor Systems: Basal Ganglia Jaime A. Pineda, Ph.D. Two major descending s Pyramidal vs. extrapyramidal Motor cortex Pyramidal system Pathway for voluntary movement Most fibers originate
Basal ganglia macrocircuits
 Tepper, Abercrombie & Bolam (Eds.) Progress in Brain Research, Vol. 160 ISSN 0079-6123 Copyright r 2007 Elsevier B.V. All rights reserved CHAPTER 1 Basal ganglia macrocircuits J.M. Tepper 1,, E.D. Abercrombie
Tepper, Abercrombie & Bolam (Eds.) Progress in Brain Research, Vol. 160 ISSN 0079-6123 Copyright r 2007 Elsevier B.V. All rights reserved CHAPTER 1 Basal ganglia macrocircuits J.M. Tepper 1,, E.D. Abercrombie
Damage on one side.. (Notes) Just remember: Unilateral damage to basal ganglia causes contralateral symptoms.
 Lecture 20 - Basal Ganglia Basal Ganglia (Nolte 5 th Ed pp 464) Damage to the basal ganglia produces involuntary movements. Although the basal ganglia do not influence LMN directly (to cause this involuntary
Lecture 20 - Basal Ganglia Basal Ganglia (Nolte 5 th Ed pp 464) Damage to the basal ganglia produces involuntary movements. Although the basal ganglia do not influence LMN directly (to cause this involuntary
GBME graduate course. Chapter 43. The Basal Ganglia
 GBME graduate course Chapter 43. The Basal Ganglia Basal ganglia in history Parkinson s disease Huntington s disease Parkinson s disease 1817 Parkinson's disease (PD) is a degenerative disorder of the
GBME graduate course Chapter 43. The Basal Ganglia Basal ganglia in history Parkinson s disease Huntington s disease Parkinson s disease 1817 Parkinson's disease (PD) is a degenerative disorder of the
Connections of basal ganglia
 Connections of basal ganglia Introduction The basal ganglia, or basal nuclei, are areas of subcortical grey matter that play a prominent role in modulating movement, as well as cognitive and emotional
Connections of basal ganglia Introduction The basal ganglia, or basal nuclei, are areas of subcortical grey matter that play a prominent role in modulating movement, as well as cognitive and emotional
Basal Ganglia Anatomy, Physiology, and Function. NS201c
 Basal Ganglia Anatomy, Physiology, and Function NS201c Human Basal Ganglia Anatomy Basal Ganglia Circuits: The Classical Model of Direct and Indirect Pathway Function Motor Cortex Premotor Cortex + Glutamate
Basal Ganglia Anatomy, Physiology, and Function NS201c Human Basal Ganglia Anatomy Basal Ganglia Circuits: The Classical Model of Direct and Indirect Pathway Function Motor Cortex Premotor Cortex + Glutamate
Making Things Happen 2: Motor Disorders
 Making Things Happen 2: Motor Disorders How Your Brain Works Prof. Jan Schnupp wschnupp@cityu.edu.hk HowYourBrainWorks.net On the Menu in This Lecture In the previous lecture we saw how motor cortex and
Making Things Happen 2: Motor Disorders How Your Brain Works Prof. Jan Schnupp wschnupp@cityu.edu.hk HowYourBrainWorks.net On the Menu in This Lecture In the previous lecture we saw how motor cortex and
A. General features of the basal ganglia, one of our 3 major motor control centers:
 Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. March 1, 2012 THE BASAL GANGLIA Objectives: 1. What are the main
Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. March 1, 2012 THE BASAL GANGLIA Objectives: 1. What are the main
Basal Ganglia General Info
 Basal Ganglia General Info Neural clusters in peripheral nervous system are ganglia. In the central nervous system, they are called nuclei. Should be called Basal Nuclei but usually called Basal Ganglia.
Basal Ganglia General Info Neural clusters in peripheral nervous system are ganglia. In the central nervous system, they are called nuclei. Should be called Basal Nuclei but usually called Basal Ganglia.
A. General features of the basal ganglia, one of our 3 major motor control centers:
 Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. April 22, 2010 THE BASAL GANGLIA Objectives: 1. What are the
Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. April 22, 2010 THE BASAL GANGLIA Objectives: 1. What are the
Afferent Control of Nigral Dopaminergic Neurons The Role of GABAergic Inputs
 In: Graybiel, A.M. et al., (Eds.) The Basal Ganglia VI, Kluwer Academic Publishers, Norwell, 2002 pp 641-651 Afferent Control of Nigral Dopaminergic Neurons The Role of GABAergic Inputs James M. Tepper,
In: Graybiel, A.M. et al., (Eds.) The Basal Ganglia VI, Kluwer Academic Publishers, Norwell, 2002 pp 641-651 Afferent Control of Nigral Dopaminergic Neurons The Role of GABAergic Inputs James M. Tepper,
The individual animals, the basic design of the experiments and the electrophysiological
 SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
Brain anatomy and artificial intelligence. L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia
 Brain anatomy and artificial intelligence L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia The Fourth Conference on Artificial General Intelligence August 2011 Architectures
Brain anatomy and artificial intelligence L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia The Fourth Conference on Artificial General Intelligence August 2011 Architectures
COGNITIVE SCIENCE 107A. Sensory Physiology and the Thalamus. Jaime A. Pineda, Ph.D.
 COGNITIVE SCIENCE 107A Sensory Physiology and the Thalamus Jaime A. Pineda, Ph.D. Sensory Physiology Energies (light, sound, sensation, smell, taste) Pre neural apparatus (collects, filters, amplifies)
COGNITIVE SCIENCE 107A Sensory Physiology and the Thalamus Jaime A. Pineda, Ph.D. Sensory Physiology Energies (light, sound, sensation, smell, taste) Pre neural apparatus (collects, filters, amplifies)
ABSTRACT INTRODUCTION
 Neuropharmacology, 1976, 15, 755-762 Self-inhibition By Dopaminergic Neurones: Disruption By (+/-)-alpha-methylp-tyrosine Pretreatment Or Anterior Diencephalic Lesions* P. M. Groves, S. J. Young and C.
Neuropharmacology, 1976, 15, 755-762 Self-inhibition By Dopaminergic Neurones: Disruption By (+/-)-alpha-methylp-tyrosine Pretreatment Or Anterior Diencephalic Lesions* P. M. Groves, S. J. Young and C.
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND
 THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD How did I get here? What did I do? Start driving home after work Aware when you left
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD How did I get here? What did I do? Start driving home after work Aware when you left
Basal Ganglia. Steven McLoon Department of Neuroscience University of Minnesota
 Basal Ganglia Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Graduate School Discussion Wednesday, Nov 1, 11:00am MoosT 2-690 with Paul Mermelstein (invite your friends)
Basal Ganglia Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Graduate School Discussion Wednesday, Nov 1, 11:00am MoosT 2-690 with Paul Mermelstein (invite your friends)
Theme 2: Cellular mechanisms in the Cochlear Nucleus
 Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
VL VA BASAL GANGLIA. FUNCTIONAl COMPONENTS. Function Component Deficits Start/initiation Basal Ganglia Spontan movements
 BASAL GANGLIA Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University at Buffalo I) Overview How do Basal Ganglia affect movement Basal ganglia enhance cortical motor activity and facilitate movement.
BASAL GANGLIA Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University at Buffalo I) Overview How do Basal Ganglia affect movement Basal ganglia enhance cortical motor activity and facilitate movement.
Exam 2 PSYC Fall (2 points) Match a brain structure that is located closest to the following portions of the ventricular system
 Exam 2 PSYC 2022 Fall 1998 (2 points) What 2 nuclei are collectively called the striatum? (2 points) Match a brain structure that is located closest to the following portions of the ventricular system
Exam 2 PSYC 2022 Fall 1998 (2 points) What 2 nuclei are collectively called the striatum? (2 points) Match a brain structure that is located closest to the following portions of the ventricular system
Supplemental Figure 1
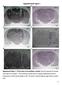 A C E Supplemental Figure 1 AP 1mm A+C 1mm Apo 1mm B D 1mm CGS F 1mm All 1mm Supplemental Figure 1. The locations of microinfusion cannulae. Each dot represents the cannula tip of a given rat in Figure.
A C E Supplemental Figure 1 AP 1mm A+C 1mm Apo 1mm B D 1mm CGS F 1mm All 1mm Supplemental Figure 1. The locations of microinfusion cannulae. Each dot represents the cannula tip of a given rat in Figure.
Visualization and simulated animations of pathology and symptoms of Parkinson s disease
 Visualization and simulated animations of pathology and symptoms of Parkinson s disease Prof. Yifan HAN Email: bctycan@ust.hk 1. Introduction 2. Biochemistry of Parkinson s disease 3. Course Design 4.
Visualization and simulated animations of pathology and symptoms of Parkinson s disease Prof. Yifan HAN Email: bctycan@ust.hk 1. Introduction 2. Biochemistry of Parkinson s disease 3. Course Design 4.
Computational cognitive neuroscience: 8. Motor Control and Reinforcement Learning
 1 Computational cognitive neuroscience: 8. Motor Control and Reinforcement Learning Lubica Beňušková Centre for Cognitive Science, FMFI Comenius University in Bratislava 2 Sensory-motor loop The essence
1 Computational cognitive neuroscience: 8. Motor Control and Reinforcement Learning Lubica Beňušková Centre for Cognitive Science, FMFI Comenius University in Bratislava 2 Sensory-motor loop The essence
Neuroscience: Exploring the Brain, 3e. Chapter 4: The action potential
 Neuroscience: Exploring the Brain, 3e Chapter 4: The action potential Introduction Action Potential in the Nervous System Conveys information over long distances Action potential Initiated in the axon
Neuroscience: Exploring the Brain, 3e Chapter 4: The action potential Introduction Action Potential in the Nervous System Conveys information over long distances Action potential Initiated in the axon
DOPAMINE DA NEURON ANATOMY AND PHYSIOLOGY
 9 DOPAMINE ANTHONY A. GRACE Studies into the regulation of the dopamine (DA) system and its postsynaptic actions are often stymied by the myriad of actions that this neurotransmitter can produce. Thus,
9 DOPAMINE ANTHONY A. GRACE Studies into the regulation of the dopamine (DA) system and its postsynaptic actions are often stymied by the myriad of actions that this neurotransmitter can produce. Thus,
PSY 315 Lecture 11 (2/23/2011) (Motor Control) Dr. Achtman PSY 215. Lecture 11 Topic: Motor System Chapter 8, pages
 Corrections: No Corrections Announcements: Exam #2 next Wednesday, March 2, 2011 Monday February 28, 2011 we will be going over the somatosensory system, and there will be time left in class to review
Corrections: No Corrections Announcements: Exam #2 next Wednesday, March 2, 2011 Monday February 28, 2011 we will be going over the somatosensory system, and there will be time left in class to review
Biological Bases of Behavior. 8: Control of Movement
 Biological Bases of Behavior 8: Control of Movement m d Skeletal Muscle Movements of our body are accomplished by contraction of the skeletal muscles Flexion: contraction of a flexor muscle draws in a
Biological Bases of Behavior 8: Control of Movement m d Skeletal Muscle Movements of our body are accomplished by contraction of the skeletal muscles Flexion: contraction of a flexor muscle draws in a
Chapter 12 Nervous Tissue
 9/12/11 Chapter 12 Nervous Tissue Overview of the nervous system Cells of the nervous system Electrophysiology of neurons Synapses Neural integration Subdivisions of the Nervous System 1 Subdivisions of
9/12/11 Chapter 12 Nervous Tissue Overview of the nervous system Cells of the nervous system Electrophysiology of neurons Synapses Neural integration Subdivisions of the Nervous System 1 Subdivisions of
神經解剖學 NEUROANATOMY BASAL NUCLEI 盧家鋒助理教授臺北醫學大學醫學系解剖學暨細胞生物學科臺北醫學大學醫學院轉譯影像研究中心.
 神經解剖學 NEUROANATOMY BASAL NUCLEI 盧家鋒助理教授臺北醫學大學醫學系解剖學暨細胞生物學科臺北醫學大學醫學院轉譯影像研究中心 http://www.ym.edu.tw/~cflu OUTLINE Components and Pathways of the Basal Nuclei Functions and Related Disorders of the Basal Nuclei
神經解剖學 NEUROANATOMY BASAL NUCLEI 盧家鋒助理教授臺北醫學大學醫學系解剖學暨細胞生物學科臺北醫學大學醫學院轉譯影像研究中心 http://www.ym.edu.tw/~cflu OUTLINE Components and Pathways of the Basal Nuclei Functions and Related Disorders of the Basal Nuclei
Towards a Better Treatment of Parkinson's disease
 Loma Linda University TheScholarsRepository@LLU: Digital Archive of Research, Scholarship & Creative Works Loma Linda University Electronic Theses, Dissertations & Projects 3-2014 Towards a Better Treatment
Loma Linda University TheScholarsRepository@LLU: Digital Archive of Research, Scholarship & Creative Works Loma Linda University Electronic Theses, Dissertations & Projects 3-2014 Towards a Better Treatment
Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817.
 Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817. Four (4) hallmark clinical signs: 1) Tremor: (Note -
Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817. Four (4) hallmark clinical signs: 1) Tremor: (Note -
Thalamo-Cortical Relationships Ultrastructure of Thalamic Synaptic Glomerulus
 Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
The Wonders of the Basal Ganglia
 Basal Ganglia The Wonders of the Basal Ganglia by Mackenzie Breton and Laura Strong /// https://kin450- neurophysiology.wikispaces.com/basal+ganglia Introduction The basal ganglia are a group of nuclei
Basal Ganglia The Wonders of the Basal Ganglia by Mackenzie Breton and Laura Strong /// https://kin450- neurophysiology.wikispaces.com/basal+ganglia Introduction The basal ganglia are a group of nuclei
Relative contributions of cortical and thalamic feedforward inputs to V2
 Relative contributions of cortical and thalamic feedforward inputs to V2 1 2 3 4 5 Rachel M. Cassidy Neuroscience Graduate Program University of California, San Diego La Jolla, CA 92093 rcassidy@ucsd.edu
Relative contributions of cortical and thalamic feedforward inputs to V2 1 2 3 4 5 Rachel M. Cassidy Neuroscience Graduate Program University of California, San Diego La Jolla, CA 92093 rcassidy@ucsd.edu
Plasticity of Cerebral Cortex in Development
 Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Neural mechanisms of reward-related motor learning Jeffery R Wickens, John N J Reynolds and Brian I Hyland
 685 Neural mechanisms of reward-related motor learning Jeffery R Wickens, John N J Reynolds and Brian I Hyland The analysis of the neural mechanisms responsible for rewardrelated learning has benefited
685 Neural mechanisms of reward-related motor learning Jeffery R Wickens, John N J Reynolds and Brian I Hyland The analysis of the neural mechanisms responsible for rewardrelated learning has benefited
Biomarkers in Schizophrenia
 Biomarkers in Schizophrenia David A. Lewis, MD Translational Neuroscience Program Department of Psychiatry NIMH Conte Center for the Neuroscience of Mental Disorders University of Pittsburgh Disease Process
Biomarkers in Schizophrenia David A. Lewis, MD Translational Neuroscience Program Department of Psychiatry NIMH Conte Center for the Neuroscience of Mental Disorders University of Pittsburgh Disease Process
Developmental regulation of Medium Spiny Neuron dendritic arborization. Lorene M. Lanier Department of Neuroscience
 Developmental regulation of Medium Spiny Neuron dendritic arborization Lorene M. Lanier Department of Neuroscience Diversity in dendritic arbors Pyramidal Purkinje Medium Spiny http://youtu.be/_tqpca6wx84
Developmental regulation of Medium Spiny Neuron dendritic arborization Lorene M. Lanier Department of Neuroscience Diversity in dendritic arbors Pyramidal Purkinje Medium Spiny http://youtu.be/_tqpca6wx84
Chapter 8. Control of movement
 Chapter 8 Control of movement 1st Type: Skeletal Muscle Skeletal Muscle: Ones that moves us Muscles contract, limb flex Flexion: a movement of a limb that tends to bend its joints, contraction of a flexor
Chapter 8 Control of movement 1st Type: Skeletal Muscle Skeletal Muscle: Ones that moves us Muscles contract, limb flex Flexion: a movement of a limb that tends to bend its joints, contraction of a flexor
The control of spiking by synaptic input in striatal and pallidal neurons
 The control of spiking by synaptic input in striatal and pallidal neurons Dieter Jaeger Department of Biology, Emory University, Atlanta, GA 30322 Key words: Abstract: rat, slice, whole cell, dynamic current
The control of spiking by synaptic input in striatal and pallidal neurons Dieter Jaeger Department of Biology, Emory University, Atlanta, GA 30322 Key words: Abstract: rat, slice, whole cell, dynamic current
Phasic Accumbal Firing May Contribute to the Regulation of Drug Taking during Intravenous Cocaine Self-administration Sessions
 Phasic Accumbal Firing May Contribute to the Regulation of Drug Taking during Intravenous Cocaine Self-administration Sessions LAURA L. PEOPLES, a ANTHONY J. UZWIAK, FRED GEE, ANTHONY T. FABBRICATORE,
Phasic Accumbal Firing May Contribute to the Regulation of Drug Taking during Intravenous Cocaine Self-administration Sessions LAURA L. PEOPLES, a ANTHONY J. UZWIAK, FRED GEE, ANTHONY T. FABBRICATORE,
Enhanced excitatory synaptic transmission in spiny neurons of rat striatum after unilateral dopamine denervation
 Neuroscience Letters 308 (2001) 201±205 www.elsevier.com/locate/neulet Enhanced excitatory synaptic transmission in spiny neurons of rat striatum after unilateral dopamine denervation Zhiping Pang, Guang
Neuroscience Letters 308 (2001) 201±205 www.elsevier.com/locate/neulet Enhanced excitatory synaptic transmission in spiny neurons of rat striatum after unilateral dopamine denervation Zhiping Pang, Guang
Modeling the interplay of short-term memory and the basal ganglia in sequence processing
 Neurocomputing 26}27 (1999) 687}692 Modeling the interplay of short-term memory and the basal ganglia in sequence processing Tomoki Fukai* Department of Electronics, Tokai University, Hiratsuka, Kanagawa
Neurocomputing 26}27 (1999) 687}692 Modeling the interplay of short-term memory and the basal ganglia in sequence processing Tomoki Fukai* Department of Electronics, Tokai University, Hiratsuka, Kanagawa
CYTOARCHITECTURE OF CEREBRAL CORTEX
 BASICS OF NEUROBIOLOGY CYTOARCHITECTURE OF CEREBRAL CORTEX ZSOLT LIPOSITS 1 CELLULAR COMPOSITION OF THE CEREBRAL CORTEX THE CEREBRAL CORTEX CONSISTS OF THE ARCHICORTEX (HIPPOCAMPAL FORMA- TION), PALEOCORTEX
BASICS OF NEUROBIOLOGY CYTOARCHITECTURE OF CEREBRAL CORTEX ZSOLT LIPOSITS 1 CELLULAR COMPOSITION OF THE CEREBRAL CORTEX THE CEREBRAL CORTEX CONSISTS OF THE ARCHICORTEX (HIPPOCAMPAL FORMA- TION), PALEOCORTEX
QUIZ YOURSELF COLOSSAL NEURON ACTIVITY
 QUIZ YOURSELF What are the factors that produce the resting potential? How is an action potential initiated and what is the subsequent flow of ions during the action potential? 1 COLOSSAL NEURON ACTIVITY
QUIZ YOURSELF What are the factors that produce the resting potential? How is an action potential initiated and what is the subsequent flow of ions during the action potential? 1 COLOSSAL NEURON ACTIVITY
Network Effects of Deep Brain Stimulation for Parkinson s Disease A Computational. Modeling Study. Karthik Kumaravelu
 Network Effects of Deep Brain Stimulation for Parkinson s Disease A Computational Modeling Study by Karthik Kumaravelu Department of Biomedical Engineering Duke University Date: Approved: Warren M. Grill,
Network Effects of Deep Brain Stimulation for Parkinson s Disease A Computational Modeling Study by Karthik Kumaravelu Department of Biomedical Engineering Duke University Date: Approved: Warren M. Grill,
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
Medical Neuroscience Tutorial
 Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
SOMATOSENSORY SYSTEMS
 SOMATOSENSORY SYSTEMS Schematic diagram illustrating the neural pathways that convey somatosensory information to the cortex and, subsequently, to the motor system. Double arrows show reciprocal connections.
SOMATOSENSORY SYSTEMS Schematic diagram illustrating the neural pathways that convey somatosensory information to the cortex and, subsequently, to the motor system. Double arrows show reciprocal connections.
Basal ganglia Sujata Sofat, class of 2009
 Basal ganglia Sujata Sofat, class of 2009 Basal ganglia Objectives Describe the function of the Basal Ganglia in movement Define the BG components and their locations Describe the motor loop of the BG
Basal ganglia Sujata Sofat, class of 2009 Basal ganglia Objectives Describe the function of the Basal Ganglia in movement Define the BG components and their locations Describe the motor loop of the BG
How strong is it? What is it? Where is it? What must sensory systems encode? 9/8/2010. Spatial Coding: Receptive Fields and Tactile Discrimination
 Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination
 Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Spatial Coding: Receptive Fields and Tactile Discrimination What must sensory systems encode? How strong is it? What is it? Where is it? When the brain wants to keep certain types of information distinct,
Nature Methods: doi: /nmeth Supplementary Figure 1. Activity in turtle dorsal cortex is sparse.
 Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
GABAergic Afferents Activate Both GABA A and GABA B Receptors in Mouse Substantia Nigra Dopaminergic Neurons In Vivo
 86 The Journal of Neuroscience, October 8, 8 8():86 98 Behavioral/Systems/Cognitive GABAergic Afferents Activate Both GABA A and GABA B Receptors in Mouse Substantia Nigra Dopaminergic Neurons In Vivo
86 The Journal of Neuroscience, October 8, 8 8():86 98 Behavioral/Systems/Cognitive GABAergic Afferents Activate Both GABA A and GABA B Receptors in Mouse Substantia Nigra Dopaminergic Neurons In Vivo
Cortical Organization. Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:.
 Cortical Organization Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:. 2. Secondary cortex: located immediately adjacent to primary cortical areas,
Cortical Organization Functionally, cortex is classically divided into 3 general types: 1. Primary cortex:. - receptive field:. 2. Secondary cortex: located immediately adjacent to primary cortical areas,
Levodopa vs. deep brain stimulation: computational models of treatments for Parkinson's disease
 Levodopa vs. deep brain stimulation: computational models of treatments for Parkinson's disease Abstract Parkinson's disease (PD) is a neurodegenerative disease affecting the dopaminergic neurons of the
Levodopa vs. deep brain stimulation: computational models of treatments for Parkinson's disease Abstract Parkinson's disease (PD) is a neurodegenerative disease affecting the dopaminergic neurons of the
The basal forebrain: Questions, chapter 29:
 The basal forebrain: Questions, chapter 29: 7) What is the "basal forebrain", and what is its involvement in Alzheimer' s Disease? The acetylcholine-containing neurons of the nucleus basalis of Meynart
The basal forebrain: Questions, chapter 29: 7) What is the "basal forebrain", and what is its involvement in Alzheimer' s Disease? The acetylcholine-containing neurons of the nucleus basalis of Meynart
Supplemental Information. Dorsal Raphe Dual Serotonin-Glutamate Neurons. Drive Reward by Establishing Excitatory Synapses
 Cell Reports, Volume 26 Supplemental Information Dorsal Raphe Dual Serotonin-Glutamate Neurons Drive Reward by Establishing Excitatory Synapses on VTA Mesoaccumbens Dopamine Neurons Hui-Ling Wang, Shiliang
Cell Reports, Volume 26 Supplemental Information Dorsal Raphe Dual Serotonin-Glutamate Neurons Drive Reward by Establishing Excitatory Synapses on VTA Mesoaccumbens Dopamine Neurons Hui-Ling Wang, Shiliang
Dynamic Nigrostriatal Dopamine Biases Action Selection
 Article Dynamic Nigrostriatal Dopamine Biases Action Selection Highlights d Nigrostriatal dopamine signaling is associated with ongoing action selection Authors Christopher D. Howard, Hao Li, Claire E.
Article Dynamic Nigrostriatal Dopamine Biases Action Selection Highlights d Nigrostriatal dopamine signaling is associated with ongoing action selection Authors Christopher D. Howard, Hao Li, Claire E.
Embryological origin of thalamus
 diencephalon Embryological origin of thalamus The diencephalon gives rise to the: Thalamus Epithalamus (pineal gland, habenula, paraventricular n.) Hypothalamus Subthalamus (Subthalamic nuclei) The Thalamus:
diencephalon Embryological origin of thalamus The diencephalon gives rise to the: Thalamus Epithalamus (pineal gland, habenula, paraventricular n.) Hypothalamus Subthalamus (Subthalamic nuclei) The Thalamus:
Chapter 2: Cellular Mechanisms and Cognition
 Chapter 2: Cellular Mechanisms and Cognition MULTIPLE CHOICE 1. Two principles about neurons were defined by Ramón y Cajal. The principle of connectional specificity states that, whereas the principle
Chapter 2: Cellular Mechanisms and Cognition MULTIPLE CHOICE 1. Two principles about neurons were defined by Ramón y Cajal. The principle of connectional specificity states that, whereas the principle
Lecture XIII. Brain Diseases I - Parkinsonism! Brain Diseases I!
 Lecture XIII. Brain Diseases I - Parkinsonism! Bio 3411! Wednesday!! Lecture XIII. Brain Diseases - I.! 1! Brain Diseases I! NEUROSCIENCE 5 th ed! Page!!Figure!!Feature! 408 18.9 A!!Substantia Nigra in
Lecture XIII. Brain Diseases I - Parkinsonism! Bio 3411! Wednesday!! Lecture XIII. Brain Diseases - I.! 1! Brain Diseases I! NEUROSCIENCE 5 th ed! Page!!Figure!!Feature! 408 18.9 A!!Substantia Nigra in
1/2/2019. Basal Ganglia & Cerebellum a quick overview. Outcomes you want to accomplish. MHD-Neuroanatomy Neuroscience Block. Basal ganglia review
 This power point is made available as an educational resource or study aid for your use only. This presentation may not be duplicated for others and should not be redistributed or posted anywhere on the
This power point is made available as an educational resource or study aid for your use only. This presentation may not be duplicated for others and should not be redistributed or posted anywhere on the
Neocortex. Cortical Structures in the Brain. Neocortex Facts. Laminar Organization. Bark-like (cortical) structures: Shepherd (2004) Chapter 12
 Neocortex Shepherd (2004) Chapter 12 Rodney Douglas, Henry Markram, and Kevan Martin Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Cortical Structures in the Brain Bark-like (cortical) structures:
Neocortex Shepherd (2004) Chapter 12 Rodney Douglas, Henry Markram, and Kevan Martin Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Cortical Structures in the Brain Bark-like (cortical) structures:
Monoamine Systems. Susan R. Sesack. Professor, Neuroscience
 Monoamine Systems Susan R. Sesack Professor, Neuroscience Nolte Nolte histidine decarboxylase Cooper, Bloom and Roth Nolte An underlying assumption in behavioral neurochemistry is that certain substances,
Monoamine Systems Susan R. Sesack Professor, Neuroscience Nolte Nolte histidine decarboxylase Cooper, Bloom and Roth Nolte An underlying assumption in behavioral neurochemistry is that certain substances,
The role of intra-thalamic and thalamocortical circuits in action selection
 The role of intra-thalamic and thalamocortical circuits in action selection M D Humphries and K N Gurney Department of Psychology, University of Sheffield, Sheffield, S10 2TP,UK E-mail: m.d.humphries@shef.ac.uk,k.gurney@shef.ac.uk
The role of intra-thalamic and thalamocortical circuits in action selection M D Humphries and K N Gurney Department of Psychology, University of Sheffield, Sheffield, S10 2TP,UK E-mail: m.d.humphries@shef.ac.uk,k.gurney@shef.ac.uk
Chemical Control of Behavior and Brain 1 of 9
 Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS
 J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
Supplementary Figure 1. Recording sites.
 Supplementary Figure 1 Recording sites. (a, b) Schematic of recording locations for mice used in the variable-reward task (a, n = 5) and the variable-expectation task (b, n = 5). RN, red nucleus. SNc,
Supplementary Figure 1 Recording sites. (a, b) Schematic of recording locations for mice used in the variable-reward task (a, n = 5) and the variable-expectation task (b, n = 5). RN, red nucleus. SNc,
Neural Communication. Central Nervous System Peripheral Nervous System. Communication in the Nervous System. 4 Common Components of a Neuron
 Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
COGS 269. Lecture 1 Spring 2018
 COGS 269 Lecture 1 Spring 2018 Psychological Experience Methods of Cognitive Neuroscience Dissociation experiments (patients with brain damage) Neuroimaging experiments Computational modeling Brain damage
COGS 269 Lecture 1 Spring 2018 Psychological Experience Methods of Cognitive Neuroscience Dissociation experiments (patients with brain damage) Neuroimaging experiments Computational modeling Brain damage
Exercise 1: (5pts) Exercise 2: (5pts) Observe the following diagram and answer the questions below.
 S.E. Biology Exercise 1: (5pts) Observe the following diagram and answer the questions below. 1. Give a suitable title for the given schematic drawing. 2. Indicate the type of the stimulus, the conductor,
S.E. Biology Exercise 1: (5pts) Observe the following diagram and answer the questions below. 1. Give a suitable title for the given schematic drawing. 2. Indicate the type of the stimulus, the conductor,
Microcircuitry coordination of cortical motor information in self-initiation of voluntary movements
 Y. Isomura et al. 1 Microcircuitry coordination of cortical motor information in self-initiation of voluntary movements Yoshikazu Isomura, Rie Harukuni, Takashi Takekawa, Hidenori Aizawa & Tomoki Fukai
Y. Isomura et al. 1 Microcircuitry coordination of cortical motor information in self-initiation of voluntary movements Yoshikazu Isomura, Rie Harukuni, Takashi Takekawa, Hidenori Aizawa & Tomoki Fukai
Physiology of synapses and receptors
 Physiology of synapses and receptors Dr Syed Shahid Habib Professor & Consultant Clinical Neurophysiology Dept. of Physiology College of Medicine & KKUH King Saud University REMEMBER These handouts will
Physiology of synapses and receptors Dr Syed Shahid Habib Professor & Consultant Clinical Neurophysiology Dept. of Physiology College of Medicine & KKUH King Saud University REMEMBER These handouts will
You submitted this quiz on Sun 19 May :32 PM IST (UTC +0530). You got a score of out of
 Feedback Ex6 You submitted this quiz on Sun 19 May 2013 9:32 PM IST (UTC +0530). You got a score of 10.00 out of 10.00. Question 1 What is common to Parkinson, Alzheimer and Autism? Electrical (deep brain)
Feedback Ex6 You submitted this quiz on Sun 19 May 2013 9:32 PM IST (UTC +0530). You got a score of 10.00 out of 10.00. Question 1 What is common to Parkinson, Alzheimer and Autism? Electrical (deep brain)
Basal Ganglia. Today s lecture is about Basal Ganglia and it covers:
 Basal Ganglia Motor system is complex interaction between Lower motor neurons (spinal cord and brainstem circuits) and Upper motor neurons (pyramidal and extrapyramidal tracts) plus two main regulators
Basal Ganglia Motor system is complex interaction between Lower motor neurons (spinal cord and brainstem circuits) and Upper motor neurons (pyramidal and extrapyramidal tracts) plus two main regulators
Strick Lecture 4 March 29, 2006 Page 1
 Strick Lecture 4 March 29, 2006 Page 1 Basal Ganglia OUTLINE- I. Structures included in the basal ganglia II. III. IV. Skeleton diagram of Basal Ganglia Loops with cortex Similarity with Cerebellar Loops
Strick Lecture 4 March 29, 2006 Page 1 Basal Ganglia OUTLINE- I. Structures included in the basal ganglia II. III. IV. Skeleton diagram of Basal Ganglia Loops with cortex Similarity with Cerebellar Loops
The Cerebral Cortex and Higher Intellectual Functions
 The Cerebral Cortex and Higher Intellectual Functions The Cerebral cortex consists of 2 cerebral hemisphere and each hemisphere consists of 5 lobes (frontal, parietal,temporal,occipital,insular lobe which
The Cerebral Cortex and Higher Intellectual Functions The Cerebral cortex consists of 2 cerebral hemisphere and each hemisphere consists of 5 lobes (frontal, parietal,temporal,occipital,insular lobe which
Supplementary Figure 1
 Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Short communication RESPONSES OF RED NUCLEUS NEURONS TO PERIPHERAL STIMULATION IN CHLORALOSE ANESTHETIZED CATS. Janusz RAJKOWSKI
 ACTA NEUROBIOL. EXP. 1982. 42: 195-201 Short communication RESPONSES OF RED NUCLEUS NEURONS TO PERIPHERAL STIMULATION IN CHLORALOSE ANESTHETIZED CATS Janusz RAJKOWSKI Department of Neurophysiology, Nencki
ACTA NEUROBIOL. EXP. 1982. 42: 195-201 Short communication RESPONSES OF RED NUCLEUS NEURONS TO PERIPHERAL STIMULATION IN CHLORALOSE ANESTHETIZED CATS Janusz RAJKOWSKI Department of Neurophysiology, Nencki
SAMPLE EXAMINATION QUESTIONS
 SAMPLE EXAMINATION QUESTIONS PLEASE NOTE, THE QUESTIONS BELOW SAMPLE THE ENTIRE LECTURE COURSE AND THEREORE INCLUDE QUESTIONS ABOUT TOPICS THAT WE HAVE NOT YET COVERED IN CLASS. 1. Which of the following
SAMPLE EXAMINATION QUESTIONS PLEASE NOTE, THE QUESTIONS BELOW SAMPLE THE ENTIRE LECTURE COURSE AND THEREORE INCLUDE QUESTIONS ABOUT TOPICS THAT WE HAVE NOT YET COVERED IN CLASS. 1. Which of the following
The motor regulator. 1) Basal ganglia/nucleus
 The motor regulator 1) Basal ganglia/nucleus Neural structures involved in the control of movement Basal Ganglia - Components of the basal ganglia - Function of the basal ganglia - Connection and circuits
The motor regulator 1) Basal ganglia/nucleus Neural structures involved in the control of movement Basal Ganglia - Components of the basal ganglia - Function of the basal ganglia - Connection and circuits
Lateral view of human brain! Cortical processing of touch!
 Lateral view of human brain! Cortical processing of touch! How do we perceive objects held in the hand?! Touch receptors deconstruct objects to detect local features! Information is transmitted in parallel
Lateral view of human brain! Cortical processing of touch! How do we perceive objects held in the hand?! Touch receptors deconstruct objects to detect local features! Information is transmitted in parallel
Physiology of Tactile Sensation
 Physiology of Tactile Sensation Objectives: 1. Describe the general structural features of tactile sensory receptors how are first order nerve fibers specialized to receive tactile stimuli? 2. Understand
Physiology of Tactile Sensation Objectives: 1. Describe the general structural features of tactile sensory receptors how are first order nerve fibers specialized to receive tactile stimuli? 2. Understand
9/28/2016. Neuron. Multipolar Neuron. Astrocytes Exchange Materials With Neurons. Glia or Glial Cells ( supporting cells of the nervous system)
 Neuron Multipolar Neuron https://www.youtube.com/watch?v=lw-psbnu5xago to :38 Glia or Glial Cells ( supporting cells of the nervous system) 10X more numerous than neurons but one-tenth the size make up
Neuron Multipolar Neuron https://www.youtube.com/watch?v=lw-psbnu5xago to :38 Glia or Glial Cells ( supporting cells of the nervous system) 10X more numerous than neurons but one-tenth the size make up
Neurons. Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons.
 Neurons Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons. MBL, Woods Hole R Cheung MSc Bioelectronics: PGEE11106 1 Neuron
Neurons Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons. MBL, Woods Hole R Cheung MSc Bioelectronics: PGEE11106 1 Neuron
Gangli della Base: un network multifunzionale
 Gangli della Base: un network multifunzionale Prof. Giovanni Abbruzzese Centro per la Malattia di Parkinson e i Disordini del Movimento DiNOGMI, Università di Genova IRCCS AOU San Martino IST Basal Ganglia
Gangli della Base: un network multifunzionale Prof. Giovanni Abbruzzese Centro per la Malattia di Parkinson e i Disordini del Movimento DiNOGMI, Università di Genova IRCCS AOU San Martino IST Basal Ganglia
Chapter 11: Nervous System and Nervous Tissue
 Chapter 11: Nervous System and Nervous Tissue I. Functions and divisions of the nervous system A. Sensory input: monitor changes in internal and external environment B. Integrations: make decisions about
Chapter 11: Nervous System and Nervous Tissue I. Functions and divisions of the nervous system A. Sensory input: monitor changes in internal and external environment B. Integrations: make decisions about
MOLECULAR BIOLOGY OF DRUG ADDICTION. Sylvane Desrivières, SGDP Centre
 1 MOLECULAR BIOLOGY OF DRUG ADDICTION Sylvane Desrivières, SGDP Centre Reward 2 Humans, as well as other organisms engage in behaviours that are rewarding The pleasurable feelings provide positive reinforcement
1 MOLECULAR BIOLOGY OF DRUG ADDICTION Sylvane Desrivières, SGDP Centre Reward 2 Humans, as well as other organisms engage in behaviours that are rewarding The pleasurable feelings provide positive reinforcement
Relationships between the Prefrontal Cortex and the Basal Ganglia in the Rat: Physiology of the Cortico-Nigral Circuits
 The Journal of Neuroscience, June 1, 1999, 19(11):4674 4681 Relationships between the Prefrontal Cortex and the Basal Ganglia in the Rat: Physiology of the Cortico-Nigral Circuits Nicolas Maurice, 1 Jean-Michel
The Journal of Neuroscience, June 1, 1999, 19(11):4674 4681 Relationships between the Prefrontal Cortex and the Basal Ganglia in the Rat: Physiology of the Cortico-Nigral Circuits Nicolas Maurice, 1 Jean-Michel
The Neuroscience of Addiction: A mini-review
 The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
This is a repository copy of Basal Ganglia. White Rose Research Online URL for this paper:
 This is a repository copy of. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/114159/ Version: Accepted Version Book Section: Prescott, A.J. orcid.org/0000-0003-4927-5390,
This is a repository copy of. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/114159/ Version: Accepted Version Book Section: Prescott, A.J. orcid.org/0000-0003-4927-5390,
Dr. Farah Nabil Abbas. MBChB, MSc, PhD
 Dr. Farah Nabil Abbas MBChB, MSc, PhD The Basal Ganglia *Functions in association with motor cortex and corticospinal pathways. *Regarded as accessory motor system besides cerebellum. *Receive most of
Dr. Farah Nabil Abbas MBChB, MSc, PhD The Basal Ganglia *Functions in association with motor cortex and corticospinal pathways. *Regarded as accessory motor system besides cerebellum. *Receive most of
