July 14, Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
|
|
|
- Osborn Flowers
- 5 years ago
- Views:
Transcription
1 July 14, 2015 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD Submitted via Re: Docket No. FDA-2015-D-1211 Revised Recommendations for Reducing the Risk of Human Immunodeficiency Virus Transmission by Blood and Blood Products, draft guidance. Dear Dockets Manager: We appreciate the opportunity to provide comments to the Food and Drug Administration on the draft guidance titled Revised Recommendations for Reducing the Risk of Human Immunodeficiency Virus Transmission by Blood and Blood Products The top priorities of the blood banking community are the safety of recipients of blood transfusions and the provision of useful, understandable information to donors to that end. We have long held that a recommendation to change the current indefinite deferral for men who have had sex with men (MSM) to a 12-month deferral would align the donor deferral period for MSM with criteria for other activities that may pose a similar risk of transfusion-transmissible infections, would maintain current levels of safety and would make sense to blood donors. We commend the FDA for taking this step forward and for providing a pathway for previously deferred donors to give blood. The Federal Register Notice that accompanied the draft guidance requested comments on signs and symptoms associated with HIV that would be most appropriate for inclusion in the donor education materials, as the draft guidance does not provide such a list. We continue to object to a recommendation that donors be provided with education materials that list, the signs and symptoms associated with HIV infection., as stated in Section III. Recommendations. A. Donor Education Material and Donor History Questionnaire. 1., and strongly recommend that this not be included in the final guidance Glenbrook Road Bethesda, MD MAIN FAX
2 The signs and symptoms of HIV in the current FDA accepted AABB DHQ donor education materials are: Unexplained weight loss or night sweats; Blue or purple spots in your mouth or skin Swollen lymph nodes for more than one month; White spots or unusual sores in your mouth; Cough that won t go away or shortness of breath; Diarrhea that won t go away; Fever of more than F for more than 10 days. These are nonspecific and insensitive for the intended purpose identification of HIV infected donors and largely redundant when donors are asked and required to be well on the day of donation. Prior to the 1985 advent of HIV testing and the evolution of ever more sensitive and specific serological tests and nucleic acid amplification assays, this recommendation was eminently reasonable there were few alternatives except this information and risk factor deferrals available to mitigate HIV risk. However, they are no longer relevant under the current testing regime. Donors with advanced immunodeficiency from HIV (i.e. AIDS) will be reactive in one or both screening approaches serological test and NAT. Under current GMP procedures, erroneous release of reactive donations is no longer an issue. Donors with acute retroviral syndrome, in the process of seroconverting, have positive NAT tests. They have only non-specific signs and symptoms (not likely to be included in the list above) that have a low positive predictive value for the presence of HIV infection. Further, in order for donors to be deemed eligible to donate they must respond affirmatively to the explicit question are you feeling well and healthy today? and must have a normal temperature. In the current draft guidance, the agency does not cite any literature to document the effectiveness of eliciting signs and symptoms of HIV/AIDS in promoting blood safety in the current environment. The final rule published May 22, 2015, Requirements for Blood and Blood Components Intended for Transfusion or for Further Manufacturing Use, provides citations regarding this information (references 6,7,8,31,32 and 33) I.. After review of these citations, they provide no primary data to support any assumed increment of transfusion safety from HIV transmission by providing donors signs and symptoms of HIV infection. Most donors have limited motivation to read pre-donation materials, and this issue is frequently compounded by low literacy levels. This results in skim reading and poor attention II, III,IV. These signs and symptoms are not specific for HIV infection but, more importantly, their sheer volume is distracting from more critical education needed by donors. The latter includes new information being developed to apprise donors of risks from iron depletion, a growing list of unacceptable medications, a more structured effort to educate donors about risks and responses to donor vasovagal reactions, and efforts to improve donor s provision of accurate responses to risk behavior questions. Finally, the amendments to the Code of Federal Regulations included in the final rule require that the educational materials [ (b)] provide an explanation of the readily identifiable risk factors closely associated with exposure to the relevant transfusion-transmitted infection. 2
3 Elsewhere in the same section is a clarification that HIV is the relevant transfusion-transmitted infection to which this requirement applies. The requirement in the Final Rule is for risk factors not signs and symptoms in the educational materials. The FDA-accepted AABB Donor Educational Materials do contain risk factors for HIV. Further, new (e) describes how to conduct the medical history interview with a requirement in (1)(iii) to capture any information the donor provides that would be Signs and/or symptoms of a relevant transfusiontransmitted infection. As mentioned above, the AABB Donor History Questionnaire (DHQ) requires the donor to affirm feeling well and healthy. Additionally, a temperature must be recorded. To summarize, we believe a continued recommendation to provide donors with signs and symptoms of HIV infection is obsolete, nonspecific, and no longer relevant under the current testing regime. Further, it is distracting in the face of a growing volume of more important information that donors should integrate before donation. Comments to specific recommendations in the guidance document are arranged in the following format: Section language from draft guidance reprinted. Recommendation or Request for Clarification recommendation or clarification request. Rationale/Supporting Information rationale in support of the recommendation /clarification request. Section II. Background. F. Status of Other Deferral Categories. In the case of the deferral for persons with hemophilia or related clotting disorders who have received clotting factor concentrates, the rationale for deferral has changed from prevention of HIV transmission to that of ensuring that donors are not harmed by the use of large bore needles used during the donation process. While 21 CFR 640.3(c)(3) currently requires deferral for receipt of any derivative of human blood which the FDA has advised is a possible source of viral hepatitis, given the enhanced safety measures now used in the manufacture of clotting factor concentrates (Ref. 16), FDA does not consider the receipt of FDA-licensed clotting factor concentrates to be a risk factor for hepatitis. Further, FDA has not recommended a deferral for the receipt of other FDAlicensed plasma-derivatives because of HIV or hepatitis risk, and we intend to consider revisions to the current regulations. Recommendation We recommend that the guidance be finalized with this update as written. Rationale The update to the guidance is congruent with the Final Rule mentioned above. Also, the AABB Donor History Task Force (DHTF) is updating the FDA-accepted DHQ in accordance with the draft recommendation. 3
4 Given the passage of time, and in order to simplify practical application of these criteria for donors and blood collection establishments, reference to since 1977 present currently for some criteria has been dropped as the period of time during which individuals are assessed to be at risk of transmitting HIV. Recommendation We recommend that the guidance be finalized with this sentence as written. Rationale The AABB DHTF is updating the FDA-accepted DHQ in accordance with the draft recommendation.. 1. We recommend that donors be provided donor education material before each donation explaining the risk of HIV transmission by blood and blood products, certain behaviors associated with the risk of HIV infection, and the signs and symptoms associated with HIV infection, so that donors can self defer. Recommendations The recommendation regarding the signs and symptoms of HIV infection should be deleted. Rationale See above 3. We recommend that the updated DHQ include the following elements to assess donors for risk: i. A history ever of a positive (footnote 4) test for HIV. Footnote 4: In this context, positive includes positive test results on an HIV diagnostic assay and repeatedly reactive or reactive results on antibody or NAT blood donor screening assays, respectively. Recommendation The footnote should include that a donor who has successfully completed a reentry program can reply No to this question. Rationale This is an ongoing problem. The FDA has previously provided personal communication on the subject and should now formalize the information in a guidance document. 3. We recommend that the updated DHQ include the following elements to assess donors for risk: 4
5 iv. A history in the past 12 months of sex with a person with a positive test for HIV, a history of exchanging sex for money or drugs, or a history of non-prescription injection drug use Recommendation Footnote 4 added to the guidance to support element i. (In this context, positive includes positive test results on an HIV diagnostic assay and repeatedly reactive or reactive results on antibody or NAT blood donor screening assays, respectively) should be expanded to allow this donor to be eligible unless the sexual contact had a confirmed positive test for HIV or has not had a negative confirmatory test result. Rationale There are few cases these days when HIV positive or reactive test results are not reflexed to confirmatory testing. Updating the footnote as recommended will allow donors to continue to donate if their partners have not received confirmed positive test results. 3. We recommend that the updated DHQ include the following elements to assess donors for risk: ix. For male donors: a history in the past 12 months of sex with another man Recommendation We support this update to the 1992 memorandum and recommend that the guidance be finalized with this element as stated. Rationale The update aligns the donor deferral period for MSM with criteria for other activities that may pose a similar risk of transfusion-transmissible infections. In addition, the AABB Donor History Task Force (DHTF) is updating the FDA-accepted DHQ in accordance with the draft recommendation. 3. We recommend that the updated DHQ include the following elements to assess donors for risk: x. For female donors: a history in the past 12 months of sex with a man who has had sex with another man Recommendation This element to assess donors for risk should be written as For female donors: a history in the past 12 months of sex with a man who has had sex with another man in the past 12 months. Rationale The final recommendation should clarify that the period of evaluation for the sexual contact between the two males is the 12 months prior to the sexual contact the female had with the male. It appears to be inferred in the requalification criteria but should be clear in the assessment elements. 5
6 Note: In the context of the donor history questionnaire, male or female gender is taken to be self-identified and self-reported. In instances where a donor has asserted a change in gender identification, medical directors may exercise discretion with regard to donor eligibility. Recommendation We recommend that the guidance be finalized with this Note as written. Rationale We appreciate that the agency has previously addressed gender identification at AABB FDA Liaison Committee meetings and find it an especially relevant addition to the guidance. Section III. Recommendations C. Donor Requalification 6. Male donors previously deferred because of a history of sex with another man, even one time, since 1977, may be eligible to donate provided that they have not had sex with another man during the past 12 months and they meet all other donor eligibility criteria. Recommendation We recommend that the guidance be finalized with this requalification criteria as written. Rationale The requalification criteria is clear and we appreciate having the information immediately available in the guidance. Section III. Recommendations C. Donor Requalification 7. Male donors deferred because of a history of sex with another man in the past 12 months may be eligible to donate provided they have not had sex with another man during the past 12 months and they meet all other donor eligibility criteria. Recommendation We recommend that the guidance be finalized with this requalification criteria as written. Rationale The requalification criteria is clear and we appreciate having the information immediately available in the guidance. AABB is an international, not-for-profit association representing individuals and institutions involved in the field of transfusion medicine and cellular therapies. The association is committed to improving health by developing and delivering standards, accreditation and educational programs that focus on optimizing patient and donor care and safety. AABB membership consists of nearly 2,000 institutions and 8,000 individuals, including physicians, nurses, 6
7 scientists, researchers, administrators, medical technologists and other health care providers. AABB members are located in more than 80 countries. Founded in 1962, America's Blood Centers is North America's largest network of communitybased, independent blood programs. The network operates more than 600 blood donor centers providing over half of the U.S., and a quarter of the Canadian blood supply. These blood centers serve more than 150 million people and provide blood products and services to more than 3,500 hospitals and healthcare facilities across North America. America's Blood Centers' U.S. members are licensed and regulated by the U.S. Food and Drug Administration. Canadian members are regulated by Health Canada. The American Red Cross shelters, feeds and provides emotional support to victims of disasters; supplies about 40 percent of the nation's blood; teaches skills that save lives; provides international humanitarian aid; and supports military members and their families. The Red Cross is a not-for-profit organization that depends on volunteers and the generosity of the American public to perform its mission. About 5.6 million units of whole blood are collected from roughly 3.3 million Red Cross volunteer donors, separated into 8 million transfusable blood products and supplied to approximately 2,700 hospitals and transfusion centers across the country for patients in need. Thank you for the opportunity to offer these comments. We look forward to continuing to work with the FDA on patient and donor safety initiatives. Questions concerning these comments may be directed to acarrgreer@aabb.org. Sincerely, M. Allene Carr-Greer Director, Regulatory Affairs I. References cited in Final Rule Requirements for Blood and Blood Components Intended for Transfusion or for Further Manufacturing Use, May 22, FDA, Guidance for Industry: Implementation of Acceptable Full-Length Donor History Questionnaire and Accompanying Materials for Use in Screening Donors of Blood and Blood Components, October 2006, ces/blood/ucm htm. 7. FDA, Guidance for Industry: Implementation of an Acceptable Full- Length and Abbreviated Donor History Questionnaires and Accompanying Materials for Use in Screening Donors of Source Plasma, February 2013, Products/ApprovedProducts/Licensed ProductsBLAs/BloodDonorScreening/ UCM pdf. 8. FDA, Guidance for Industry: Implementation of an Acceptable Abbreviated Donor History Questionnaire and Accompanying Materials for Use in Screening Frequent Donors of Blood and Blood Components, May 2013, Information/Guidances/Blood/ ucm htm. 7
8 31. AABB Association Bulletin #03-14, Deferral for Risk of Leishmaniasis Exposure, October 10, Bai, N. Donor Fatigue. The Red Cross Has Banned Chronic Fatigue Syndrome Sufferers from Giving Blood. But Does a Virus Really Cause the Disease? Scientific American July 2011;305(1): Young, S., A Fink, S. Geiger, et al. Community Blood Donors Knowledge of Anemia and Design of a Literacy- Appropriate Educational Intervention.Transfusion 2010;50:75-9. II. O Brien SF, Osmond L, Choquet K, Yi QL, Goldman M. Donor attention to reading materials. Vox Sang 2015, DOI: /vox III. Rugege-Hakiza SE, Glynn SA, Hutching ST, et al. Do blood donors read and understand screening educational materials? Transfusion 2003;43: IV. National Center for Education Statistics: (Accessed June 12, 2015). 8
23 November Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
 23 November 2016 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA-2016-N-1502,
23 November 2016 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA-2016-N-1502,
June 8, Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
 June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
Guidance for Industry
 Guidance for Industry Revised Recommendations for Donor and Product Management Based on Screening Tests for Syphilis DRAFT GUIDANCE This document is being distributed for comment purposes only. Submit
Guidance for Industry Revised Recommendations for Donor and Product Management Based on Screening Tests for Syphilis DRAFT GUIDANCE This document is being distributed for comment purposes only. Submit
[Submitted Electronically]
![[Submitted Electronically] [Submitted Electronically]](/thumbs/82/85246007.jpg) [Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
[Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
DHQ v2.0, February 2016 Table Detailing Changes from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you
 DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
Guidance for Industry
 Reprinted from FDA s website by Guidance for Industry Recommendations for Screening, Testing, and Management of Blood Donors and Blood and Blood Components Based on Screening Tests for Syphilis DRAFT GUIDANCE
Reprinted from FDA s website by Guidance for Industry Recommendations for Screening, Testing, and Management of Blood Donors and Blood and Blood Components Based on Screening Tests for Syphilis DRAFT GUIDANCE
Guidance for Industry
 Guidance for Industry Informed Consent Recommendations for Source Plasma Donors Participating in Plasmapheresis and Immunization Programs Additional copies of this guidance are available from the Office
Guidance for Industry Informed Consent Recommendations for Source Plasma Donors Participating in Plasmapheresis and Immunization Programs Additional copies of this guidance are available from the Office
Allogeneic donor selection and blood collection. by Mohammed Abu-basha
 Allogeneic donor selection and blood collection 1 An allogeneic (also called homologous) donation (allo- from the Greek meaning "other") is when a donor gives blood for storage at a blood bank for transfusion
Allogeneic donor selection and blood collection 1 An allogeneic (also called homologous) donation (allo- from the Greek meaning "other") is when a donor gives blood for storage at a blood bank for transfusion
Re: Docket No. FDA D Presenting Risk Information in Prescription Drug and Medical Device Promotion
 1201 Maryland Avenue SW, Suite 900, Washington, DC 20024 202-962-9200, www.bio.org August 25, 2009 Dockets Management Branch (HFA-305) Food and Drug Administration 5600 Fishers Lane, Rm. 1061 Rockville,
1201 Maryland Avenue SW, Suite 900, Washington, DC 20024 202-962-9200, www.bio.org August 25, 2009 Dockets Management Branch (HFA-305) Food and Drug Administration 5600 Fishers Lane, Rm. 1061 Rockville,
Hot Topics in Tissue Safety
 Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Guidance for Industry
 Guidance for Industry Use of Nucleic Acid Tests on Pooled and Individual Samples from Donors of Whole Blood and Blood Components (including Source Plasma and Source Leukocytes) to Adequately and Appropriately
Guidance for Industry Use of Nucleic Acid Tests on Pooled and Individual Samples from Donors of Whole Blood and Blood Components (including Source Plasma and Source Leukocytes) to Adequately and Appropriately
Inspections, Compliance, Enforcement, and Criminal Investigations. Central Texas Regional Blood & Tissue Center 07-Nov-03
 1 of 7 6/10/2009 2:20 PM Inspections, Compliance, Enforcement, and Criminal Investigations Department of Health and Human Services Public Health Service Food and Drug Administration Dallas District 4040
1 of 7 6/10/2009 2:20 PM Inspections, Compliance, Enforcement, and Criminal Investigations Department of Health and Human Services Public Health Service Food and Drug Administration Dallas District 4040
Guidance for Industry DRAFT GUIDANCE. This guidance document is being distributed for comment purposes only.
 Compounded Drug Products That Are Essentially Copies of a Commercially Available Drug Product Under Section 503A of the Federal Food, Drug, and Cosmetic Act Guidance for Industry DRAFT GUIDANCE This guidance
Compounded Drug Products That Are Essentially Copies of a Commercially Available Drug Product Under Section 503A of the Federal Food, Drug, and Cosmetic Act Guidance for Industry DRAFT GUIDANCE This guidance
Guidance for Industry
 Guidance for Industry Adequate and Appropriate Donor Screening Tests for Hepatitis B; Hepatitis B Surface Antigen (HBsAg) Assays Used to Test Donors of Whole Blood and Blood Components, Including Source
Guidance for Industry Adequate and Appropriate Donor Screening Tests for Hepatitis B; Hepatitis B Surface Antigen (HBsAg) Assays Used to Test Donors of Whole Blood and Blood Components, Including Source
Current Infectious Disease Screening for the Live Organ Donor
 2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
DEPARTMENT OF HEALTH AND SENIOR SERVICES PO BOX 360 TRENTON, N.J
 JON S. CORZINE Governor DEPARTMENT OF HEALTH AND SENIOR SERVICES PO BOX 360 TRENTON, N.J. 08625-0360 www.nj.gov/health HEATHER HOWARD Commissioner TO: FROM: Public Health Council Heather Howard Commissioner
JON S. CORZINE Governor DEPARTMENT OF HEALTH AND SENIOR SERVICES PO BOX 360 TRENTON, N.J. 08625-0360 www.nj.gov/health HEATHER HOWARD Commissioner TO: FROM: Public Health Council Heather Howard Commissioner
Changing eligibility criteria for MSM: The Canadian Perspective
 Changing eligibility criteria for MSM: The Canadian Perspective Mindy Goldman MD Canadian Blood Services ASFA & WAA Joint Meeting, San Francisco April 4, 2014 Outline - Historical background - International
Changing eligibility criteria for MSM: The Canadian Perspective Mindy Goldman MD Canadian Blood Services ASFA & WAA Joint Meeting, San Francisco April 4, 2014 Outline - Historical background - International
Making Your Blood Donation Safe
 BLOOD DONOR EDUCATIONAL MATERIALS Making Your Blood Donation Safe Pete Lawson Burned in a wildfire, LifeStream blood donations saved his life. WWW.LSTREAM.ORG NEW! Important Information on Iron! Recent
BLOOD DONOR EDUCATIONAL MATERIALS Making Your Blood Donation Safe Pete Lawson Burned in a wildfire, LifeStream blood donations saved his life. WWW.LSTREAM.ORG NEW! Important Information on Iron! Recent
HIV/AIDS. Kuna High School Mr. Stanley
 HIV/AIDS Kuna High School Mr. Stanley Questions 1. Write an example of how your immune system helps prevent you from getting diseases. Terms to know Epidemic - a widespread occurrence of an infectious
HIV/AIDS Kuna High School Mr. Stanley Questions 1. Write an example of how your immune system helps prevent you from getting diseases. Terms to know Epidemic - a widespread occurrence of an infectious
USER INSTRUCTIONS HPC, Cord Blood Donor History Questionnaire. Table of Contents
 Page 1 of 9 USER INSTRUCTIONS HPC, Cord Blood Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials Structure and Content Reformatting
Page 1 of 9 USER INSTRUCTIONS HPC, Cord Blood Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials Structure and Content Reformatting
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire. Table of Contents
 User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials - Structure and
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials - Structure and
Guidance for Industry
 Guidance for Industry Advisement for the Use of Gen-Probe Procleix Ultrio Plus Assay for NAT Testing on Cadaveric Donors Release Date: 08/03/14 American Medical Education and Research Association 4757
Guidance for Industry Advisement for the Use of Gen-Probe Procleix Ultrio Plus Assay for NAT Testing on Cadaveric Donors Release Date: 08/03/14 American Medical Education and Research Association 4757
Over-the-Counter Pediatric Liquid Drug Products Containing Acetaminophen
 Reprinted from FDA s website by EAS Consulting Group, LLC Over-the-Counter Pediatric Liquid Drug Products Containing Acetaminophen Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed
Reprinted from FDA s website by EAS Consulting Group, LLC Over-the-Counter Pediatric Liquid Drug Products Containing Acetaminophen Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed
Zika Virus. Report to the Standing Committee on Health. Dr. Graham Sher Chief Executive Officer, Canadian Blood Services
 Dr. Graham Sher Chief Executive Officer, Services Dr. Dana Devine Chief Medical and Scientific Officer, Services Services Services is an arm s-length organization within the larger health-care system of
Dr. Graham Sher Chief Executive Officer, Services Dr. Dana Devine Chief Medical and Scientific Officer, Services Services Services is an arm s-length organization within the larger health-care system of
DHQ Flowcharts v2.0 eff. February 2016
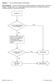 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Blood Donor Counselling
 Blood Donor Counselling A presentation for the Nepal Red Cross Society Blood Transfusion Service Dr Che Kit Lin On behalf of GAP and Hong Kong Red Cross 26 th August 2014 Purpose and Outcomes of Workshop
Blood Donor Counselling A presentation for the Nepal Red Cross Society Blood Transfusion Service Dr Che Kit Lin On behalf of GAP and Hong Kong Red Cross 26 th August 2014 Purpose and Outcomes of Workshop
Department of Health and Human Services
 Friday, August 24, 2007 Part III Department of Health and Human Services Food and Drug Administration 2 CFR Parts 606 and 60 Current Good Manufacturing Practice for Blood and Blood Components; Notification
Friday, August 24, 2007 Part III Department of Health and Human Services Food and Drug Administration 2 CFR Parts 606 and 60 Current Good Manufacturing Practice for Blood and Blood Components; Notification
Draft Guidance for Industry and FDA Staff
 Draft Guidance for Industry and FDA Staff Submission and Review of Sterility Information in Premarket Notification (510(k)) Submissions for Devices Labeled as Sterile DRAFT GUIDANCE This guidance document
Draft Guidance for Industry and FDA Staff Submission and Review of Sterility Information in Premarket Notification (510(k)) Submissions for Devices Labeled as Sterile DRAFT GUIDANCE This guidance document
Generic Drug User Fee Amendments of 2012; Regulatory Science Initiatives; Public Hearing;
 4160-01-P DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration 21 CFR Part 15 [Docket No. FDA-2013-N-0402] Generic Drug User Fee Amendments of 2012; Regulatory Science Initiatives; Public
4160-01-P DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration 21 CFR Part 15 [Docket No. FDA-2013-N-0402] Generic Drug User Fee Amendments of 2012; Regulatory Science Initiatives; Public
Guidance for Industry
 Reprinted from FDA s website by Guidance for Industry Bar Code Label Requirements Questions and Answers (Question 12 Update) DRAFT GUIDANCE This guidance document is for comment purposes only. Submit comments
Reprinted from FDA s website by Guidance for Industry Bar Code Label Requirements Questions and Answers (Question 12 Update) DRAFT GUIDANCE This guidance document is for comment purposes only. Submit comments
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire. Table of Contents
 User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Methods of Administration DHQ Materials - Structure and Content Reformatting
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Methods of Administration DHQ Materials - Structure and Content Reformatting
Facilitate physician access to compounded drugs for office-use from 503A compounding pharmacies for patients with emergent conditions;
 By Electronic Delivery Scott Gottlieb, MD Commissioner Food and Drug Administration Attn: Division of Dockets Management (HFA-305) 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: FDA-2017-N-5093 for
By Electronic Delivery Scott Gottlieb, MD Commissioner Food and Drug Administration Attn: Division of Dockets Management (HFA-305) 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: FDA-2017-N-5093 for
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services
 Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
CERTIFIED MAIL RETURN RECEIPT REQUESTED. Esther Hernandez President United States Blood Bank, Inc NW 95th Avenue Miami, Florida
 DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration Silver Spring MD 20993 CERTIFIED MAIL RETURN RECEIPT REQUESTED Esther Hernandez President United States Blood Bank, Inc. 2400 NW 95th
DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration Silver Spring MD 20993 CERTIFIED MAIL RETURN RECEIPT REQUESTED Esther Hernandez President United States Blood Bank, Inc. 2400 NW 95th
Guidance for Industry
 Guidance for Industry Lookback for Hepatitis C Virus (HCV): Product Quarantine, Consignee Notification, Further Testing, Product Disposition, and Notification of Transfusion Recipients Based on Donor Test
Guidance for Industry Lookback for Hepatitis C Virus (HCV): Product Quarantine, Consignee Notification, Further Testing, Product Disposition, and Notification of Transfusion Recipients Based on Donor Test
Revisions to Labeling Requirements for Blood and Blood Components, Including Source Plasma
 This document is scheduled to be published in the Federal Register on 01/03/2012 and available online at http://federalregister.gov/a/2011-33554, and on FDsys.gov 4160-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 01/03/2012 and available online at http://federalregister.gov/a/2011-33554, and on FDsys.gov 4160-01-P DEPARTMENT OF HEALTH AND HUMAN
Policy Document. Blood Donation Deferral. Background
 Policy Document Blood Donation Deferral Background The Australian Medical Students Association (AMSA) is the peak representative body for medical students in Australia. Accordingly, AMSA advocates on issues
Policy Document Blood Donation Deferral Background The Australian Medical Students Association (AMSA) is the peak representative body for medical students in Australia. Accordingly, AMSA advocates on issues
Guidance for Industry
 Reprinted from FDA s website by EAS Consulting Group, LLC Guidance for Industry Recommendations for Screening, Testing, and Management of Blood Donors and Blood and Blood Components Based on Screening
Reprinted from FDA s website by EAS Consulting Group, LLC Guidance for Industry Recommendations for Screening, Testing, and Management of Blood Donors and Blood and Blood Components Based on Screening
Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry
 Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
April 25, 2014 Reference No. HAMB FDA Transparency Initiative Regulations Development
 Reference No. HAMB14001 Via Email Dr. Margaret Hamburg Commissioner U.S. Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 margaret.hamburg@fda.hhs.gov SUBJECT: FDA Transparency
Reference No. HAMB14001 Via Email Dr. Margaret Hamburg Commissioner U.S. Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 margaret.hamburg@fda.hhs.gov SUBJECT: FDA Transparency
Class II Special Controls Guidance Document: Intraoral Devices for Snoring and/or Obstructive Sleep Apnea; Guidance for Industry and FDA
 Class II Special Controls Guidance Document: Intraoral Devices for Snoring and/or Obstructive Sleep Apnea; Guidance for Industry and FDA Document issued on: November 12, 2002 This document supersedes the
Class II Special Controls Guidance Document: Intraoral Devices for Snoring and/or Obstructive Sleep Apnea; Guidance for Industry and FDA Document issued on: November 12, 2002 This document supersedes the
DEPARTMENT OF VETERANS AFFAIRS SUMMARY: The Department of Veterans Affairs (VA) proposes to amend its medical
 This document is scheduled to be published in the Federal Register on 08/05/2016 and available online at http://federalregister.gov/a/2016-18660, and on FDsys.gov DEPARTMENT OF VETERANS AFFAIRS 8320-01
This document is scheduled to be published in the Federal Register on 08/05/2016 and available online at http://federalregister.gov/a/2016-18660, and on FDsys.gov DEPARTMENT OF VETERANS AFFAIRS 8320-01
Centers for Disease Control and Prevention. Recommendations for Providers Counseling Male Patients and
 This document is scheduled to be published in the Federal Register on 12/02/2014 and available online at http://federalregister.gov/a/2014-27814, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
This document is scheduled to be published in the Federal Register on 12/02/2014 and available online at http://federalregister.gov/a/2014-27814, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
Guidelines for Implementing Pre-Exposure Prophylaxis For The Prevention of HIV in Youth Peter Havens, MD MS Draft:
 Guidelines for Implementing Pre-Exposure Prophylaxis For The Prevention of HIV in Youth Peter Havens, MD MS Draft: 10-2-2015 Clinical studies demonstrate that when a person without HIV infection takes
Guidelines for Implementing Pre-Exposure Prophylaxis For The Prevention of HIV in Youth Peter Havens, MD MS Draft: 10-2-2015 Clinical studies demonstrate that when a person without HIV infection takes
QSEAL Recovered Plasma Specification
 QSEAL Recovered Plasma Specification Background The is part of a series of standards that comprise the Plasma Protein Therapeutics Association (PPTA) QSEAL Standards Program. PPTA's Voluntary Standards
QSEAL Recovered Plasma Specification Background The is part of a series of standards that comprise the Plasma Protein Therapeutics Association (PPTA) QSEAL Standards Program. PPTA's Voluntary Standards
Agency Information Collection Activities; Submission for Office of Management and Budget
 This document is scheduled to be published in the Federal Register on 01/28/2016 and available online at http://federalregister.gov/a/2016-01690, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 01/28/2016 and available online at http://federalregister.gov/a/2016-01690, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
CTYOMEGALOVIRUS (CMV) - BACKGROUND
 CTYOMEGALOVIRUS (CMV) - BACKGROUND PURPOSE The flowing information provides guidance on the use of CMV negative blood components provided by the blood bank at the Royal Children s Hospital (RCH) including
CTYOMEGALOVIRUS (CMV) - BACKGROUND PURPOSE The flowing information provides guidance on the use of CMV negative blood components provided by the blood bank at the Royal Children s Hospital (RCH) including
NATIONAL BLOOD TRANSFUSION SERVICES STRATEGY
 FEDERAL DEMOCRATIC REPUBLIC OF ETHIOPIA MINISTRY OF HEALTH NATIONAL BLOOD TRANSFUSION SERVICES STRATEGY February 2005 Addis Ababa Ethiopia Acknowledgement The Ministry of Health of the Federal Democratic
FEDERAL DEMOCRATIC REPUBLIC OF ETHIOPIA MINISTRY OF HEALTH NATIONAL BLOOD TRANSFUSION SERVICES STRATEGY February 2005 Addis Ababa Ethiopia Acknowledgement The Ministry of Health of the Federal Democratic
Directed Semen Donor Eligibility Requirements
 Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Responsibilities of Laser Light Show Projector Manufacturers, Dealers, and Distributors; Final Guidance for Industry and FDA.
 Responsibilities of Laser Light Show Projector Manufacturers, Dealers, and Distributors; Final Guidance for Industry and FDA (Laser Notice 51) Document issued on: May 27, 2001 U.S. Department of Health
Responsibilities of Laser Light Show Projector Manufacturers, Dealers, and Distributors; Final Guidance for Industry and FDA (Laser Notice 51) Document issued on: May 27, 2001 U.S. Department of Health
Center for Biologics Evaluation and Research
 Center for Biologics Evaluation and Research Current Activities Future Directions Washington, DC January 25, 2010 Karen Midthun, MD Acting Director, CBER CBER Our Mission To ensure the safety, purity,
Center for Biologics Evaluation and Research Current Activities Future Directions Washington, DC January 25, 2010 Karen Midthun, MD Acting Director, CBER CBER Our Mission To ensure the safety, purity,
NOTICE OF INITIATION OF DISQUALIFICATION PROCEEDINGS AND OPPORTUNITY TO EXPLAIN
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 1401 Rockville Pike Rockville, MD 20852-1448 September 26, 2011 By Overnight Delivery Michael Dean Berger, M.D.
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 1401 Rockville Pike Rockville, MD 20852-1448 September 26, 2011 By Overnight Delivery Michael Dean Berger, M.D.
DEPARTMENT OF VETERANS AFFAIRS SUMMARY: The Department of Veterans Affairs (VA) is amending its medical
 This document is scheduled to be published in the Federal Register on 03/23/2017 and available online at https://federalregister.gov/d/2017-05799, and on FDsys.gov DEPARTMENT OF VETERANS AFFAIRS 8320-01
This document is scheduled to be published in the Federal Register on 03/23/2017 and available online at https://federalregister.gov/d/2017-05799, and on FDsys.gov DEPARTMENT OF VETERANS AFFAIRS 8320-01
Use of Light, Mild, Low, or Similar Descriptors in the Label, Labeling, or Advertising of Tobacco Products
 Guidance for Industry and FDA Staff Use of Light, Mild, Low, or Similar Descriptors in the Label, Labeling, or Advertising of Tobacco Products June 2010 For questions regarding this guidance, contact the
Guidance for Industry and FDA Staff Use of Light, Mild, Low, or Similar Descriptors in the Label, Labeling, or Advertising of Tobacco Products June 2010 For questions regarding this guidance, contact the
December 4, 2017 VIA ELECTRONIC SUBMISSION
 VIA ELECTRONIC SUBMISSION December 4, 2017 Dockets Management Staff (HFA-305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: Development of a List of pre-dietary Supplement
VIA ELECTRONIC SUBMISSION December 4, 2017 Dockets Management Staff (HFA-305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: Development of a List of pre-dietary Supplement
TOBACCO PRODUCT OR MEDICAL PRODUCT?
 TOBACCO PRODUCT OR MEDICAL PRODUCT? Priscilla Callahan-Lyon, MD Deputy Director Division of Individual Health Science Office of Science, CTP Grail Sipes, JD Director Office of Regulatory Policy, CDER Disclaimer:
TOBACCO PRODUCT OR MEDICAL PRODUCT? Priscilla Callahan-Lyon, MD Deputy Director Division of Individual Health Science Office of Science, CTP Grail Sipes, JD Director Office of Regulatory Policy, CDER Disclaimer:
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE BACKGROUND
 MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
Hypertension: Developing Fixed- Dose Combination Drugs for Treatment Guidance for Industry
 Hypertension: Developing Fixed- Dose Combination Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Hypertension: Developing Fixed- Dose Combination Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Process or performance improvement is often discussed
 Process Improvement in Transfusion Medicine Ongoing efforts to decrease costs in the clinical laboratory make continuous process improvement especially important in difficult economic times. Process improvement
Process Improvement in Transfusion Medicine Ongoing efforts to decrease costs in the clinical laboratory make continuous process improvement especially important in difficult economic times. Process improvement
May 16, Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org May 16, 2014 Division of Dockets Management (HFA-305) Food and Drug Administration
701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org May 16, 2014 Division of Dockets Management (HFA-305) Food and Drug Administration
Transfusion-transmitted HIV from seroconverted blood donors has not been identified in England since the introduction of HIV NAT screening
 Transfusion-transmitted HIV from seroconverted blood donors has not been identified in England since the introduction of HIV NAT screening Bhavita Vishram Timeline of introduction of microbiological tests
Transfusion-transmitted HIV from seroconverted blood donors has not been identified in England since the introduction of HIV NAT screening Bhavita Vishram Timeline of introduction of microbiological tests
Annual Statistical Update: HIV and AIDS
 Annual Statistical Update: HIV and AIDS 2015 Data reported to December 31, 2015 Epidemiology & Surveillance Public Health Branch Public Health and Primary Health Care Division Manitoba Health, Seniors
Annual Statistical Update: HIV and AIDS 2015 Data reported to December 31, 2015 Epidemiology & Surveillance Public Health Branch Public Health and Primary Health Care Division Manitoba Health, Seniors
FDA Reentry Guidance
 FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
As a result of this training, participants will be able to:
 Addressing Prevention with HIV Positive Clients 1 Day Training This one-day training will prepare participants to help people living with HIV to avoid sexual and substance use behaviors that can result
Addressing Prevention with HIV Positive Clients 1 Day Training This one-day training will prepare participants to help people living with HIV to avoid sexual and substance use behaviors that can result
Implementation of the Stem Cell Therapeutic and Research Act of 2005
 Implementation of the Stem Cell Therapeutic and Research Act of 2005 The 6 th Annual Somatic Cell Therapy Symposium Bethesda, MD September 26, 2006 James F. Burdick, M.D. U.S. Department of Health and
Implementation of the Stem Cell Therapeutic and Research Act of 2005 The 6 th Annual Somatic Cell Therapy Symposium Bethesda, MD September 26, 2006 James F. Burdick, M.D. U.S. Department of Health and
ANDA Submissions Refuse to Receive for Lack of Proper Justification of Impurity Limits Guidance for Industry
 ANDA Submissions Refuse to Receive for Lack of Proper Justification of Impurity Limits Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments
ANDA Submissions Refuse to Receive for Lack of Proper Justification of Impurity Limits Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments
Maximizing Cornea and Tissue Donation through Specimen Quality
 Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
A. Anti-HIV-1/2, HIV NAT Lookback: for those identified blood and blood components collected:
 PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
BAY-ARENAC BEHAVIORAL HEALTH AUTHORITY POLICIES AND PROCEDURES MANUAL
 Page: 1 of 8 Policy Bay-Arenac Behavioral Health Authority (BABHA) is committed to high standards of employee and consumer safety practices. This standard will include the prevention, surveillance, identification
Page: 1 of 8 Policy Bay-Arenac Behavioral Health Authority (BABHA) is committed to high standards of employee and consumer safety practices. This standard will include the prevention, surveillance, identification
Listing of Color Additives Exempt from Certification; Synthetic Iron Oxide
 This document is scheduled to be published in the Federal Register on 03/20/2015 and available online at http://federalregister.gov/a/2015-06418, and on FDsys.gov DEPARTMENT OF HEALTH AND HUMAN SERVICES
This document is scheduled to be published in the Federal Register on 03/20/2015 and available online at http://federalregister.gov/a/2015-06418, and on FDsys.gov DEPARTMENT OF HEALTH AND HUMAN SERVICES
Outbreak Investigation Guidance for Vectorborne Diseases
 COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
July 7, Dockets Management Branch (HFA-305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
 July 7, 2012 Dockets Management Branch (HFA-305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: Docket No. FDA 2012-N-0408: Risk Evaluation and Mitigation Strategy Assessments:
July 7, 2012 Dockets Management Branch (HFA-305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: Docket No. FDA 2012-N-0408: Risk Evaluation and Mitigation Strategy Assessments:
Guidance for Industry
 Guidance for Industry Dosage Delivery Devices for OTC Liquid Drug Products DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this
Guidance for Industry Dosage Delivery Devices for OTC Liquid Drug Products DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this
CHAPTER 2 NETWORK PROVIDER/SERVICE DELIVERY REQUIREMENTS
 CHAPTER 2 NETWORK PROVIDER/SERVICE DELIVERY REQUIREMENTS 2.9 SPECIAL POPULATIONS ADHS/DBHS receives Federal grants and State appropriations to deliver behavioral health services to special populations
CHAPTER 2 NETWORK PROVIDER/SERVICE DELIVERY REQUIREMENTS 2.9 SPECIAL POPULATIONS ADHS/DBHS receives Federal grants and State appropriations to deliver behavioral health services to special populations
MSM DONOR POLICY: STUDIES FROM THE USA AND OTHER COUNTRIES. Brian Custer IPFA PEI Workshop Rome 22 May 2014
 MSM DONOR POLICY: STUDIES FROM THE USA AND OTHER COUNTRIES Brian Custer IPFA PEI Workshop Rome 22 May 2014 Financial Disclosure Nothing to disclose International Context for Blood Donation by MSM Country
MSM DONOR POLICY: STUDIES FROM THE USA AND OTHER COUNTRIES Brian Custer IPFA PEI Workshop Rome 22 May 2014 Financial Disclosure Nothing to disclose International Context for Blood Donation by MSM Country
Nonallergic Rhinitis: Developing Drug Products for Treatment Guidance for Industry
 Nonallergic Rhinitis: Developing Drug Products for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Nonallergic Rhinitis: Developing Drug Products for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Withdrawal of Proposed Rule on Supplemental Applications Proposing Labeling Changes for
 This document is scheduled to be published in the Federal Register on 12/14/2018 and available online at https://federalregister.gov/d/2018-27098, and on govinfo.gov 4164-01-P DEPARTMENT OF HEALTH AND
This document is scheduled to be published in the Federal Register on 12/14/2018 and available online at https://federalregister.gov/d/2018-27098, and on govinfo.gov 4164-01-P DEPARTMENT OF HEALTH AND
BLOOD TRANSFUSIONS. Answers. To Your. Questions
 BLOOD TRANSFUSIONS Answers To Your Questions Answers to Your Questions This brochure is intended for people who may need a blood or blood product transfusion and for those who regularly receive transfusions.
BLOOD TRANSFUSIONS Answers To Your Questions Answers to Your Questions This brochure is intended for people who may need a blood or blood product transfusion and for those who regularly receive transfusions.
Surveillance Report 2014
 Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Overview of Blood Transfusion System of Iran:
 Report Article Overview of Blood Transfusion System of Iran: 2002-2011 AM Cheraghali High Institute for Research and Education on Transfusion Medicine, Iran Blood Transfusion Organization and University
Report Article Overview of Blood Transfusion System of Iran: 2002-2011 AM Cheraghali High Institute for Research and Education on Transfusion Medicine, Iran Blood Transfusion Organization and University
Answers. To Your. Questions
 Blood Transfusions Answers To Your Questions Answers to Your Questions This brochure is intended for people who may need a blood or blood product transfusion and for those who regularly receive transfusions.
Blood Transfusions Answers To Your Questions Answers to Your Questions This brochure is intended for people who may need a blood or blood product transfusion and for those who regularly receive transfusions.
CANADIAN HEMOPHILIA SOCIETY POSITION: CURRENT DONOR DEFERRAL CRITERIA ARE IN THE BEST INTERESTS OF BLOOD SAFETY
 CANADIAN HEMOPHILIA SOCIETY POSITION: CURRENT DONOR DEFERRAL CRITERIA ARE IN THE BEST INTERESTS OF BLOOD SAFETY The Canadian Hemophilia Society (CHS) supports the current Canadian regulations regarding
CANADIAN HEMOPHILIA SOCIETY POSITION: CURRENT DONOR DEFERRAL CRITERIA ARE IN THE BEST INTERESTS OF BLOOD SAFETY The Canadian Hemophilia Society (CHS) supports the current Canadian regulations regarding
Teva Pharmaceuticals USA Attention: Scott D. Tomsky Vice President, US Generics Regulatory Affairs 425 Privet Road Horsham, PA 19044
 DEPARTMENT OF HEALTH & HUMAN SERVICES ANDA 090783 Food and Drug Administration Silver Spring, MD 20993 Teva Pharmaceuticals USA Attention: Scott D. Tomsky Vice President, US Generics Regulatory Affairs
DEPARTMENT OF HEALTH & HUMAN SERVICES ANDA 090783 Food and Drug Administration Silver Spring, MD 20993 Teva Pharmaceuticals USA Attention: Scott D. Tomsky Vice President, US Generics Regulatory Affairs
ANDA Arthur P. Bedrosian, President Armenpharm, Ltd. 49 South Ridge Road P.O. Box D1400 Pomona, NY December 3, 2015
 DEPARTMENT OF HEALTH & HUMAN SERVICES Silver Spring, MD 20993 ANDA 060851 Arthur P. Bedrosian, President Armenpharm, Ltd. 49 South Ridge Road P.O. Box D1400 Pomona, NY 10970 Docket No. FDA-2011-P-0081
DEPARTMENT OF HEALTH & HUMAN SERVICES Silver Spring, MD 20993 ANDA 060851 Arthur P. Bedrosian, President Armenpharm, Ltd. 49 South Ridge Road P.O. Box D1400 Pomona, NY 10970 Docket No. FDA-2011-P-0081
Division of Dockets Management Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 October 31, 2017 Division of Dockets Management Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Re: Docket Nos. FDA 2012 N 1210 & FDA 2004 N 0258 Dear Sir or Madam: The (AHA)
October 31, 2017 Division of Dockets Management Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Re: Docket Nos. FDA 2012 N 1210 & FDA 2004 N 0258 Dear Sir or Madam: The (AHA)
Acknowledgement of receipt of notice of privacy practices
 Acknowledgement of receipt of notice of privacy practices NOTICE OF PRIVACY PRACTICES I acknowledge that I have received a Notice of Privacy Practices from Kettering Physician Network (dba Kettering Cancer
Acknowledgement of receipt of notice of privacy practices NOTICE OF PRIVACY PRACTICES I acknowledge that I have received a Notice of Privacy Practices from Kettering Physician Network (dba Kettering Cancer
BLOOD PROGRAM. The American Legion
 BLOOD PROGRAM The American Legion BLOOD AND COMMUNITY HEALTH The use of the blood of one human being to save the life of another is one of the world s greatest medical achievements. Thousands of people
BLOOD PROGRAM The American Legion BLOOD AND COMMUNITY HEALTH The use of the blood of one human being to save the life of another is one of the world s greatest medical achievements. Thousands of people
Nestle Infant Nutrition 10/31/14
 U.S. Food and Drug Administration Protecting and Promoting Your Health Nestle Infant Nutrition 10/31/14 OCT 31, 2014 Department of Health and Human Services Public Health Service Food and Drug Administration
U.S. Food and Drug Administration Protecting and Promoting Your Health Nestle Infant Nutrition 10/31/14 OCT 31, 2014 Department of Health and Human Services Public Health Service Food and Drug Administration
H - Human. A - Acquired I - Immunodeficiency I - Immune V - Virus D - Deficiency S - Syndrome
 Definition of Terms H - Human A - Acquired I - Immunodeficiency I - Immune V - Virus D - Deficiency S - Syndrome How is HIV Transmitted? Risk Behaviors Unprotected anal, oral, and vaginal intercourse Sharing
Definition of Terms H - Human A - Acquired I - Immunodeficiency I - Immune V - Virus D - Deficiency S - Syndrome How is HIV Transmitted? Risk Behaviors Unprotected anal, oral, and vaginal intercourse Sharing
As a result of this training, participants will be able to:
 Addressing Sexual Risk with Drug Users and their Partners 1 Day Training This one-day training will build participant knowledge and skills in offering sexual harm reduction options to substance users.
Addressing Sexual Risk with Drug Users and their Partners 1 Day Training This one-day training will build participant knowledge and skills in offering sexual harm reduction options to substance users.
DEFENSE HEALTH AGENCY 7700 ARLINGTON BOULEVARD, SUITE 5101 FALLS CHURCH, VIRGINIA
 DEFENSE HEALTH AGENCY 7700 ARLINGTON BOULEVARD, SUITE 5101 FALLS CHURCH, VIRGINIA 22042-5101 DHA-IPM 18-020 MEMORANDUM FOR ASSISTANT SECRETARY OF THE ARMY (MANPOWER AND RESERVE AFFAIRS) ASSISTANT SECRETARY
DEFENSE HEALTH AGENCY 7700 ARLINGTON BOULEVARD, SUITE 5101 FALLS CHURCH, VIRGINIA 22042-5101 DHA-IPM 18-020 MEMORANDUM FOR ASSISTANT SECRETARY OF THE ARMY (MANPOWER AND RESERVE AFFAIRS) ASSISTANT SECRETARY
Comments of the Patient, Consumer, and Public Health Coalition. Strengthening the Center for Devices and Radiological Health s 510(k) Review Process
 March 19, 2010 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Comments of the Patient, Consumer, and Public Health Coalition on Strengthening
March 19, 2010 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Comments of the Patient, Consumer, and Public Health Coalition on Strengthening
HIV and Long Term Disability
 HIV and Long Term Disability Minnesota AIDS Project The Minnesota AIDS Project envisions a world free of AIDS. Our mission is to lead Minnesota's fight to stop HIV through advocacy, education, and service.
HIV and Long Term Disability Minnesota AIDS Project The Minnesota AIDS Project envisions a world free of AIDS. Our mission is to lead Minnesota's fight to stop HIV through advocacy, education, and service.
HIV/AIDS. Communication and Prevention. Davison Community Schools Grade Six June 2018
 HIV/AIDS Communication and Prevention Davison Community Schools Grade Six June 2018 Discussing Sensitive Matters with Your Parents Parents: A child s first and most important teacher Parent s role is to
HIV/AIDS Communication and Prevention Davison Community Schools Grade Six June 2018 Discussing Sensitive Matters with Your Parents Parents: A child s first and most important teacher Parent s role is to
Name of Recipient: Recipient s DOB (if known) Relationship to Recipient: (Example: mother, father, sister, brother, friend, etc)
 The Christ Hospital Health Network DONOR REGISTRATION INFORMATION Phone: 513-585-2493 Fax: 513-585-0433 (Please be advised donor information is needed ONLY to register donor in the Christ Hospital system.
The Christ Hospital Health Network DONOR REGISTRATION INFORMATION Phone: 513-585-2493 Fax: 513-585-0433 (Please be advised donor information is needed ONLY to register donor in the Christ Hospital system.
OHRP Guidance on the Involvement of Prisoners in Research
 NOTE: THIS GUIDANCE REPLACES THE FOLLOWING OHRP GUIDANCE: "OHRP Guidance on Approving Research Involving Prisoners" (May 19, 2000) found on the OHRP website at: http://ohrp.osophs.dhhs.gov/humansubjects/guidance/prison.htm
NOTE: THIS GUIDANCE REPLACES THE FOLLOWING OHRP GUIDANCE: "OHRP Guidance on Approving Research Involving Prisoners" (May 19, 2000) found on the OHRP website at: http://ohrp.osophs.dhhs.gov/humansubjects/guidance/prison.htm
EHR Developer Code of Conduct Frequently Asked Questions
 EHR Developer Code of Conduct Frequently Asked Questions General What is the purpose of the EHR Developer Code of Conduct? EHR Association (the Association) members have a long tradition of working with
EHR Developer Code of Conduct Frequently Asked Questions General What is the purpose of the EHR Developer Code of Conduct? EHR Association (the Association) members have a long tradition of working with
OF THE REPUBLIC OF ARMENIA DECREE. 316 of 1 April 2002 Yerevan
 Ratified President of the Republic of Armenia R. Kocharyan 1 April 2002 GOVERNMENT OF THE REPUBLIC OF ARMENIA DECREE 316 of 1 April 2002 Yerevan On ratification of the National Programme on HIV/AIDS Prevention
Ratified President of the Republic of Armenia R. Kocharyan 1 April 2002 GOVERNMENT OF THE REPUBLIC OF ARMENIA DECREE 316 of 1 April 2002 Yerevan On ratification of the National Programme on HIV/AIDS Prevention
Infectious Disease Considerations in Organ Donation. Nikole A Neidlinger MD FACS Chief Medical Officer
 Infectious Disease Considerations in Organ Donation Nikole A Neidlinger MD FACS Chief Medical Officer Screening Donors and Infectious Disease considerations: Deceased Donor Screening Donors at increased
Infectious Disease Considerations in Organ Donation Nikole A Neidlinger MD FACS Chief Medical Officer Screening Donors and Infectious Disease considerations: Deceased Donor Screening Donors at increased
