HealthPack Medical Device Packaging Technical Committee of IoPP. March 4, 2015 Norfolk, VA
|
|
|
- Opal Baldwin
- 6 years ago
- Views:
Transcription
1 HealthPack 2015 Medical Device Packaging Technical Committee of IoPP March 4, 2015 Norfolk, VA
2 MDPTC 2015 HealthPack Meeting Mission Statement Provide a forum for packaging professionals representing the Medical Device Industry for discussion, exchange and development of information on technical issues, regulations, standards and new technologies
3 MDPTC 2015 HealthPack Meeting Vision Statement Provide the latest information on national and international regulatory requirements as they affect the packaging of medical devices. Provide a forum to voice the device industry packaging position on issues that affect them. Provide education to new members entering the medical device area of package engineering. Provide a timely list of tools and documents (guides, test methods and standards) that are helpful when performing the tasks related to the package engineering of medical devices. Exchange non- proprietary information regarding new materials, equipment and systems, which can improve the overall performance and quality of the medical device packaging industry. Act as a liaison for technical and regulatory updates between organizations of mutual interest such as AAMI, ISO, ASTM, ISTA and the U.S. FDA.
4 MDPTC 2015 HealthPack Meeting Officer Introduction Chairperson Jennifer Griffin, CPP Vice Chairperson Marco Ortiz Shields, CPP Membership/Secretary Eric Carlson, CPP Subcommittee Chair Treasurer Ombudsman Program Chairperson Webmaster Karen Greene, CPP Jennifer Griffin, CPP David W. DiVaccaro Charles Kanazeh Marco Ortiz Shields, CPP
5 MDPTC 2015 HealthPack Meeting Overview Membership Current committee membership = 255 Current membership in sub-committees = 44+ Linked-In group = 187 members
6 MDPTC 2015 HealthPack Meeting Subcommittees Temperature Controlled Packaging, Led by Jim Butschli Survey executed and published Sustainability, Led by Daphne Allen Survey executed and presented 2014 Labeling, Led by Dave Olson Developing a guidance document to identify consistent and repeatable tests and procedures to insure that terminally sterilized device package labels comply with the affixed and legible requirements of Subpart K 120(a) Sec 820 Education, Led by Charlie Robnett Developing a packaging value chain map for medical device Packaging Operations, Led by John Derek Thompson ASTM Standard Practice Development: Guide for the Development of Naked Eye Visual Inspection Criteria for the Acceptance or Rejection of Detected Debris and Particulate in Final Packaging of Terminally Sterilized Medical Devices Sterilization, Led by Mike Tokarski Sterilization database available on the on IoPP website
7 MDPTC 2015 HealthPack Meeting Committee meeting held March 3, 2015 March 3 meeting---39 participants Updates from Sustainability, Labeling and Operations subcommittees Webinars planned for 2015: Intro to Medical device packaging in Spanish by Marco Ortiz Packaging for reprocessed medical devices by Chris Sarantos
8 Medical Packaging Game Show Do you know your stuff related to aseptic presentation of sterile packages and related topics?
9 Game Show question 1 True or False Shelf life is a guarantee that items will remain sterile until the expiration date.
10 Game Show answer 1 Shelf life is a guarantee that items will remain sterile until the expiration date. False
11 Game Show question 2 True or False A stained package can be used if the package is dry and if it passes visual inspection for seal and package integrity.
12 Game Show answer 2 A stained package can be used if the package is dry and if it passes visual inspection for seal and package integrity. False The contents of wet or stained packages may have been subject to strike-through which occurs when liquids soak through a barrier. Strike-through provides for passage of microorganisms; contents must be considered contaminated.
13 Game Show question 3 True or False Drapes serve as a barrier to prevent the passage of microorganisms between sterile and non-sterile areas.
14 Game Show answer 3 Drapes serve as a barrier to prevent the passage of microorganisms between sterile and non-sterile areas. True
15 Game Show question 4 True or False Heavy, awkward, or sharp items should be gently dumped onto the sterile field.
16 Game Show answer 4 Heavy, awkward, or sharp items should be gently dumped onto the sterile field. False These items should not be tossed onto the sterile field because they may roll off the edge or displace other items or penetrate the sterile barrier. These items should be opened on a separate table or stand or be received directly by the scrub person.
17 Game Show question 5 True or False On peel-open packages, contents should be slid out over package edges to present the contents to the sterile field.
18 Game Show answer 5 On peel-open packages, contents should be slid out over package edges to present the contents to the sterile field. False The flaps on peel-open packages should be pulled back, not torn, to expose the sterile contents. The contents should not be permitted to slide over the edges. The inner edge of the heat seal is considered the line of demarcation between sterile and unsterile.
19 Game Show question 6 The illustration shows: a. Aseptic technique b. Aseptic dump c. Aseptic transfer
20 Game Show answer 6 The illustration shows: a. Aseptic technique b. Aseptic dump c. Aseptic transfer C. Aseptic transfer---removing a sterile article from an opened package via a sterile, gloved operator. The operator must be cautious not touch his/her gloved hand to any unsterile area.
21 Game Show question 7 Aseptic opening is opening a package without contaminating the contents. The most common reasons for contamination are: a. Generation of unsterile fibers or particles during the opening process which settle on the contained product b. Attached pieces or fragments on the unsterile exterior of the package which fall or curl inward and contaminate the contents c. Package design that prevents easy access and removal of the contents in an aseptic manner. d. A and B e. A and C f. B and C g. A, B and C
22 Game Show answer 7 Aseptic opening is opening a package without contaminating the contents. The most common reasons for contamination are: a. Generation of unsterile fibers or particles during the opening process which settle on the contained product b. Attached pieces or fragments on the unsterile exterior of the package which fall or curl inward and contaminate the contents c. Package design that prevents easy access and removal of the contents in an aseptic manner. d. A and B e. A and C f. B and C g. A, B and C G---A, B and C
23 Game Show question 8 True or False The scrub nurse or person ensures a sterile transfer to the sterile field.
24 Game Show answer 8 The scrub nurse or person ensures a sterile transfer to the sterile field. False---the circulating person ensures the sterile transfer
25 8 questions---how did you do?
26 References Essentials of Perioperative Nursing, Third Edition, Cynthia Spry, 2005 Surgical Technology Principles and Practice, Sixth Edition, Joanna Kotcher Fuller, 2013 Berry and Kohn s Operating Room Technique, Seventh Edition, Lucy Jo Atkinson, 1992
27 Join us! Medical Device Packaging Technical Committee of IoPP Contact Eric Carlson Only requirement is membership in IoPP
Scope This policy applies to all personnel and departments that clean, prepare and/or sterilize items intended for patient care use.
 Dental Sterilization Procedures Policy Number VIM4(4)-10 Purpose The purpose of this policy is to ensure patient and employee safety when using instruments with potential for exposure to bloodborne pathogens
Dental Sterilization Procedures Policy Number VIM4(4)-10 Purpose The purpose of this policy is to ensure patient and employee safety when using instruments with potential for exposure to bloodborne pathogens
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
INFECTION CONTROL IN PRACTICE. Continuing Education
 INFECTION CONTROL IN PRACTICE Continuing Education CONTINUING EDUCATION: INFECTION CONTROL IN PRACTICE DESCRIPTION This seminar introduces infection control principals and best practices pertaining to
INFECTION CONTROL IN PRACTICE Continuing Education CONTINUING EDUCATION: INFECTION CONTROL IN PRACTICE DESCRIPTION This seminar introduces infection control principals and best practices pertaining to
APWA FLORIDA CHAPTER MEMBERSHIP PLAN
 APWA FLORIDA CHAPTER MEMBERSHIP PLAN MEMBERSHIP COMMITTEE From APWA National: The purpose of the Membership Committee is to increase efforts which support recruitment of new and diverse members to APWA;
APWA FLORIDA CHAPTER MEMBERSHIP PLAN MEMBERSHIP COMMITTEE From APWA National: The purpose of the Membership Committee is to increase efforts which support recruitment of new and diverse members to APWA;
AST Standards of Practice for Monitoring Sterility
 AST Standards of Practice for Monitoring Sterility Introduction The following Standards of Practice were researched and authored by the AST Education and Professional Standards Committee and have been
AST Standards of Practice for Monitoring Sterility Introduction The following Standards of Practice were researched and authored by the AST Education and Professional Standards Committee and have been
ASTM Workshop on Medical Device Cleanliness
 ASTM Workshop on Medical Device Cleanliness Steve Goldstine Consultants SteveGoldstine@Yahoo.com 11/16/2010 ASTM: Cleanliness of Medical Devices ASTM Workshop on Medical Device Cleanliness Standard Practices
ASTM Workshop on Medical Device Cleanliness Steve Goldstine Consultants SteveGoldstine@Yahoo.com 11/16/2010 ASTM: Cleanliness of Medical Devices ASTM Workshop on Medical Device Cleanliness Standard Practices
Instructions for Use. For use with. 10 mg vial
 Instructions for Use For use with 10 mg vial Table of Contents Parts of the ZOMA-Jet 10... 1 Supplies you will need to mix a ZOMACTON 10 mg Vial... 2 Mix a ZOMACTON 10 mg vial... 3 Reset the ZOMA-Jet 10...
Instructions for Use For use with 10 mg vial Table of Contents Parts of the ZOMA-Jet 10... 1 Supplies you will need to mix a ZOMACTON 10 mg Vial... 2 Mix a ZOMACTON 10 mg vial... 3 Reset the ZOMA-Jet 10...
AMENDATORY SECTION (Amending WSR , filed 10/10/95, effective 11/10/95)
 AMENDATORY SECTION (Amending WSR 95-21-041, filed 10/10/95, effective 11/10/95) WAC 246-817-601 Purpose. The purpose of WAC 246-817-601 through ((246-817-630)) 246-817-660 is to establish requirements
AMENDATORY SECTION (Amending WSR 95-21-041, filed 10/10/95, effective 11/10/95) WAC 246-817-601 Purpose. The purpose of WAC 246-817-601 through ((246-817-630)) 246-817-660 is to establish requirements
IV Link Staff. Infection Prevention & Control A Learning Package for IV Link Staff
 IV Link Staff Infection Prevention & Control A Learning Package for IV Link Staff Purpose This learning package provides key infection prevention messages and direction for knowledge that you require as
IV Link Staff Infection Prevention & Control A Learning Package for IV Link Staff Purpose This learning package provides key infection prevention messages and direction for knowledge that you require as
Management of Central Venous Access Devices. Blood Glucose Monitoring
 Management of Central Venous Access Devices Blood Glucose Monitoring Purpose To provide education on the standard of care regarding the use and monitoring of the Accu- Chek Blood glucose machine, including
Management of Central Venous Access Devices Blood Glucose Monitoring Purpose To provide education on the standard of care regarding the use and monitoring of the Accu- Chek Blood glucose machine, including
Policy Title: Single-Use (Disposable) Devices Policy Number: 13. Effective Date: 6/10/2013 Review Date: 6/10/2016
 Policy Title: Single-Use (Disposable) Devices Policy Number: 13 5. ROLES AND RESPONSIBILITIES: 5.1. All dental healthcare personnel have responsibility to conform and respect all aspects of this policy.
Policy Title: Single-Use (Disposable) Devices Policy Number: 13 5. ROLES AND RESPONSIBILITIES: 5.1. All dental healthcare personnel have responsibility to conform and respect all aspects of this policy.
Product Sterility Testing... To Test or Not to Test? That Is the Question
 Product Sterility Testing... To Test or Not to Test? That Is the Question Elaine Daniell, Trabue Bryans, Kimbrell Darnell, Joyce Hansen, Victoria M. Hitchins, and Manuel Saavedra Abstract The applications
Product Sterility Testing... To Test or Not to Test? That Is the Question Elaine Daniell, Trabue Bryans, Kimbrell Darnell, Joyce Hansen, Victoria M. Hitchins, and Manuel Saavedra Abstract The applications
HUMULIN 70/30 KwikPen
 1 Instructions for Use HUMULIN 70/30 KwikPen (70% human insulin isophane suspension 30% human insulin injection [rdna origin]) 100 units/ml, 3 ml pen Read the Instructions for Use before you start taking
1 Instructions for Use HUMULIN 70/30 KwikPen (70% human insulin isophane suspension 30% human insulin injection [rdna origin]) 100 units/ml, 3 ml pen Read the Instructions for Use before you start taking
How to use your gonal-f pre-filled pen
 How to use your gonal-f pre-filled pen Information in this user guide is based on the European product information for Gonal-f. It is not country specific and may vary from country to country. Please always
How to use your gonal-f pre-filled pen Information in this user guide is based on the European product information for Gonal-f. It is not country specific and may vary from country to country. Please always
AST Guidelines for Best Practices in Monitoring Sterility
 1 AST Guidelines for Best Practices in Monitoring Sterility Approved October 2008 Revised February 2015 Revised April 14, 2017 Introduction The following Guidelines for Best Practices were researched and
1 AST Guidelines for Best Practices in Monitoring Sterility Approved October 2008 Revised February 2015 Revised April 14, 2017 Introduction The following Guidelines for Best Practices were researched and
Larry! 28/06/2016. House Keeping. Welcome! From the GoToWebinar page:
 A Review of Sterilization Methods and Recommended Practices for Healthcare Facilities PART I June 30, 2016 Larry Talapa Welcome! Topic: A Review of Sterilization Methods and Recommended Practices for Healthcare
A Review of Sterilization Methods and Recommended Practices for Healthcare Facilities PART I June 30, 2016 Larry Talapa Welcome! Topic: A Review of Sterilization Methods and Recommended Practices for Healthcare
Standard operating procedures for preparation and administration of intramuscular injections. No Action Rationale
 Standard operating procedures for preparation and administration of intramuscular injections Preparation Overview No Action Rationale 1 Collect and check all equipment 2 Check that the packaging of all
Standard operating procedures for preparation and administration of intramuscular injections Preparation Overview No Action Rationale 1 Collect and check all equipment 2 Check that the packaging of all
Joint Commission In Dental Settings
 Joint Commission In Dental Settings Jay Afrow DMD, MHA Surveyor, Ambulatory Health Care The Joint Commission The Joint Commission Disclaimer These slides are current as of 1/19/2018. The Joint Commission
Joint Commission In Dental Settings Jay Afrow DMD, MHA Surveyor, Ambulatory Health Care The Joint Commission The Joint Commission Disclaimer These slides are current as of 1/19/2018. The Joint Commission
STERILITY TESTING OF PHARMACEUTICAL PRODUCTS
 STERILITY TESTING OF PHARMACEUTICAL PRODUCTS Tim Sandle CONTENTS Introduction xiii 1 STERILITY 1 Introduction 1 Sterility 3 Microorganisms and Microbial Growth 5 Types of microorganisms 7 Sterilization
STERILITY TESTING OF PHARMACEUTICAL PRODUCTS Tim Sandle CONTENTS Introduction xiii 1 STERILITY 1 Introduction 1 Sterility 3 Microorganisms and Microbial Growth 5 Types of microorganisms 7 Sterilization
Single market, regulatory environment, industries under vertical legislation Pharmaceuticals : regulatory framework and market authorisations
 EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market, regulatory environment, industries under vertical legislation Pharmaceuticals : regulatory framework and market authorisations Brussels,
EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market, regulatory environment, industries under vertical legislation Pharmaceuticals : regulatory framework and market authorisations Brussels,
Velocity Orthopedics Instrument Cleaning and Sterilization
 Velocity Orthopedics Instrument Cleaning and Sterilization It is important to read the Instructions For Use in its entirety prior to using the product. Caution: Federal law (USA) restricts this device
Velocity Orthopedics Instrument Cleaning and Sterilization It is important to read the Instructions For Use in its entirety prior to using the product. Caution: Federal law (USA) restricts this device
How Do You Know If a Sterilized Package Remains Sterile?
 How Do You Know If a Sterilized Package Remains Sterile? 1 Acknowledgments Bioaerosol and Applied Microbiology Team Brian Heimbuch, MS Group Lead Del Harnish, MS Lead Technical Mike McDonald, MS Lead Engineer
How Do You Know If a Sterilized Package Remains Sterile? 1 Acknowledgments Bioaerosol and Applied Microbiology Team Brian Heimbuch, MS Group Lead Del Harnish, MS Lead Technical Mike McDonald, MS Lead Engineer
THIS IS UNFPA. Male Latex Condom Testing Guidance UNICEF UNFPA WHO Joint Meeting September David Hill
 THIS IS UNFPA Male Latex Condom Testing Guidance UNICEF UNFPA WHO Joint Meeting September 2017 David Hill 2 Condom tests in ISO 4074:2015 The following tests are required in ISO 4074:2015:- Biocompatibility
THIS IS UNFPA Male Latex Condom Testing Guidance UNICEF UNFPA WHO Joint Meeting September 2017 David Hill 2 Condom tests in ISO 4074:2015 The following tests are required in ISO 4074:2015:- Biocompatibility
Safety First! 5/13/2018. Disclaimer. Objectives
 Safety First! OSHA and Infection Control Disclaimer Karen Gregory RN is an employee of Total Medical Compliance. Karen Gregory is a Hu-Friedy Key Opinion Leader, a consultant for SciCan and serves on the
Safety First! OSHA and Infection Control Disclaimer Karen Gregory RN is an employee of Total Medical Compliance. Karen Gregory is a Hu-Friedy Key Opinion Leader, a consultant for SciCan and serves on the
`bob`=å~ãéê~=ó=å~êéi=åäé~åáåöi= ÇáëáåÑÉÅíáçå=~åÇ=ëíÉêáäáò~íáçå
 kéï=~ë=çñw MUKOMNT `bob`=å~ãéê~=ó=å~êéi=åäé~åáåöi= ÇáëáåÑÉÅíáçå=~åÇ=ëíÉêáäáò~íáçå fåëíêìåíáçåë=ñçê=`bob`=lãåáå~ã=~åç=`bob`=_äìéå~ã Eî~äáÇ=Ñçê=rp^F båöäáëü=erpf CEREC camera care, cleaning, disinfection
kéï=~ë=çñw MUKOMNT `bob`=å~ãéê~=ó=å~êéi=åäé~åáåöi= ÇáëáåÑÉÅíáçå=~åÇ=ëíÉêáäáò~íáçå fåëíêìåíáçåë=ñçê=`bob`=lãåáå~ã=~åç=`bob`=_äìéå~ã Eî~äáÇ=Ñçê=rp^F båöäáëü=erpf CEREC camera care, cleaning, disinfection
Instructions for Use. Welcome!
 Instructions for Use Welcome! The AMJEVITA SureClick autoinjector is a single-use prefilled autoinjector. Consult your doctor if you have any questions about your dose. Your doctor has prescribed AMJEVITA
Instructions for Use Welcome! The AMJEVITA SureClick autoinjector is a single-use prefilled autoinjector. Consult your doctor if you have any questions about your dose. Your doctor has prescribed AMJEVITA
Complex Medical Devices and Steam Sterilization. By Andrew Gay Director - Sterilizer Validation Australia
 Complex Medical Devices and Steam Sterilization By Andrew Gay Director - Sterilizer Validation Australia About 10 years ago, during a routine Performance Qualification test, an observation was made that
Complex Medical Devices and Steam Sterilization By Andrew Gay Director - Sterilizer Validation Australia About 10 years ago, during a routine Performance Qualification test, an observation was made that
Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial
 Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial How do I prepare and give an injection with Enbrel multiple-dose vial? A multiple-dose vial contains
Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial How do I prepare and give an injection with Enbrel multiple-dose vial? A multiple-dose vial contains
Do not re use needles. If you do you might not get your dose (underdosing) or get too much (overdosing) as the needle could block.
 What are the ingredients in TOUJEO? Active ingredient: insulin glargine Inactive ingredients: zinc, m cresol, glycerol and water for injection Hydrochloric acid and sodium hydroxide may be added to adjust
What are the ingredients in TOUJEO? Active ingredient: insulin glargine Inactive ingredients: zinc, m cresol, glycerol and water for injection Hydrochloric acid and sodium hydroxide may be added to adjust
Operational Guideline
 1 of 6 PURPOSE Prevention of the spread of sexually transmitted and blood-borne infections (STBBI) through distribution of harm reduction supplies SCOPE & GOAL Harm reduction supplies and condoms will
1 of 6 PURPOSE Prevention of the spread of sexually transmitted and blood-borne infections (STBBI) through distribution of harm reduction supplies SCOPE & GOAL Harm reduction supplies and condoms will
How Do You Know If Your Sterilized Instruments Remain Sterile?
 How Do You Know If Your Sterilized Instruments Remain Sterile? Disclosure Harry Shaffer is the first author of the sterility maintenance study covered in this presentation and has been compensated by Halyard
How Do You Know If Your Sterilized Instruments Remain Sterile? Disclosure Harry Shaffer is the first author of the sterility maintenance study covered in this presentation and has been compensated by Halyard
Instructions for Use. HUMALOG KwikPen. insulin lispro injection (rdna origin) 100 units/ml, 3 ml pen
 1 Instructions for Use HUMALOG KwikPen insulin lispro injection (rdna origin) 100 units/ml, 3 ml pen Read the Instructions for Use before you start taking HUMALOG and each time you get another KwikPen.
1 Instructions for Use HUMALOG KwikPen insulin lispro injection (rdna origin) 100 units/ml, 3 ml pen Read the Instructions for Use before you start taking HUMALOG and each time you get another KwikPen.
A high-level overview of the requirements of medical packaging standards
 Medical Packaging A high-level overview of the requirements of medical packaging standards Dec 2012 Thierry Wagner Regulatory Affairs Director Europe, Middle East and Africa DuPont Medical & Pharmaceutical
Medical Packaging A high-level overview of the requirements of medical packaging standards Dec 2012 Thierry Wagner Regulatory Affairs Director Europe, Middle East and Africa DuPont Medical & Pharmaceutical
Instructional Guide RECONSTITUTION OF IDELVION USING THE MIX2VIAL TRANSFER SET
 Instructional Guide RECONSTITUTION OF IDELVION USING THE MIX2VIAL TRANSFER SET The only FDA-approved treatment for hemophilia B with up to 14-day dosing* * In appropriate people 12 years and older. Talk
Instructional Guide RECONSTITUTION OF IDELVION USING THE MIX2VIAL TRANSFER SET The only FDA-approved treatment for hemophilia B with up to 14-day dosing* * In appropriate people 12 years and older. Talk
EU Regulatory Update & GMP Deficiencies. Bryan J Wright July 2017
 EU Regulatory Update & GMP Deficiencies Bryan J Wright July 2017 Slide 1 PharmOut 2017 Outline Update on changes in EU GMP Guide Summary of GMP changes to the Guide Important details of changes over the
EU Regulatory Update & GMP Deficiencies Bryan J Wright July 2017 Slide 1 PharmOut 2017 Outline Update on changes in EU GMP Guide Summary of GMP changes to the Guide Important details of changes over the
Doxpal. Self-retaining Retractor
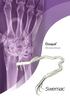 Doxpal Self-retaining Retractor Doxpal Easy to use and reposition Doxpal retractors are easy to use and easy to reposition due to the elastic properties of the plastic. There are no locking devices to
Doxpal Self-retaining Retractor Doxpal Easy to use and reposition Doxpal retractors are easy to use and easy to reposition due to the elastic properties of the plastic. There are no locking devices to
INSTRUCTIONS FOR USE
 Comfort Injects 50 Comfort-in INSTRUCTIONS FOR USE English user manual 1 Introduction Thank you for choosing the Comfort in Needle free Injection system, developed with your comfort and convenience in
Comfort Injects 50 Comfort-in INSTRUCTIONS FOR USE English user manual 1 Introduction Thank you for choosing the Comfort in Needle free Injection system, developed with your comfort and convenience in
To standardize wound care and prevent infection in compromised patients who have a Berlin Heart Ventricular Assist Device (VAD).
 PURPOSE To standardize wound care and prevent infection in compromised patients who have a Berlin Heart Ventricular Assist Device (VAD). POLICY STATEMENTS Dressing change should be done no sooner than
PURPOSE To standardize wound care and prevent infection in compromised patients who have a Berlin Heart Ventricular Assist Device (VAD). POLICY STATEMENTS Dressing change should be done no sooner than
Instructions for Use. Wright Medical Technology 1023 Cherry Road Memphis, TN USA
 Instructions for Use Wright Medical Technology 1023 Cherry Road Memphis, TN 38117 USA 1-800-238-7117 Processed from Donated Human Tissue for Wright Medical Technology by LifeCell Corporation One Millennium
Instructions for Use Wright Medical Technology 1023 Cherry Road Memphis, TN 38117 USA 1-800-238-7117 Processed from Donated Human Tissue for Wright Medical Technology by LifeCell Corporation One Millennium
AMENDATORY SECTION (Amending WSR , filed 10/10/95, effective. WAC Purpose. The purpose of WAC through
 AMENDATORY SECTION (Amending WSR 95-21-041, filed 10/10/95, effective 11/10/95) WAC 246-817-601 Purpose. The purpose of WAC 246-817-601 through ((246-817-630)) 246-817-660 is to establish requirements
AMENDATORY SECTION (Amending WSR 95-21-041, filed 10/10/95, effective 11/10/95) WAC 246-817-601 Purpose. The purpose of WAC 246-817-601 through ((246-817-630)) 246-817-660 is to establish requirements
CARE OF A TUNNELED CATHETER (HICKMAN & BROVIAC ) with a Needleless Connector (MicroClave Clear)
 CARE OF A TUNNELED CATHETER (HICKMAN & BROVIAC ) with a Needleless Connector (MicroClave Clear) Table of Contents Part 1 Learning about the Catheter...2 Part 2 Caring for Your Child s Catheter...3 A. Preventing
CARE OF A TUNNELED CATHETER (HICKMAN & BROVIAC ) with a Needleless Connector (MicroClave Clear) Table of Contents Part 1 Learning about the Catheter...2 Part 2 Caring for Your Child s Catheter...3 A. Preventing
It is essential to have the victim(s) examined by a medical
 Sexual Offenses Chapter 7 It is essential to have the victim(s) examined by a medical professional as soon as possible after the assault and before the affected areas (pubic area, vagina, rectum, etc.)
Sexual Offenses Chapter 7 It is essential to have the victim(s) examined by a medical professional as soon as possible after the assault and before the affected areas (pubic area, vagina, rectum, etc.)
STANDARD OPERATING PROCEDURE #203 LARGE ANIMAL SURGERY
 STANDARD OPERATING PROCEDURE #203 LARGE ANIMAL SURGERY 1. PURPOSE This Standard Operating Procedure (SOP) describes procedures for general surgery of large animal species such as swine, dogs, rabbits,
STANDARD OPERATING PROCEDURE #203 LARGE ANIMAL SURGERY 1. PURPOSE This Standard Operating Procedure (SOP) describes procedures for general surgery of large animal species such as swine, dogs, rabbits,
The term Routine Practices is used to describe practices that were previously known as Universal Precautions.
 Health & Safety Manual Health Promotion & Wellness ROUTINE PRACTICES PROCEDURES INTRODUCTION The term Routine Practices is used to describe practices that were previously known as Universal Precautions.
Health & Safety Manual Health Promotion & Wellness ROUTINE PRACTICES PROCEDURES INTRODUCTION The term Routine Practices is used to describe practices that were previously known as Universal Precautions.
Infection Prevention Checklist for Dental Settings. Basic Expectations for Safe Care
 Infection Prevention Checklist for Dental Settings Basic Expectations for Safe Care Infection Prevention Checklist for Dental Settings: Basic Expectations for Safe Care The following is a companion to
Infection Prevention Checklist for Dental Settings Basic Expectations for Safe Care Infection Prevention Checklist for Dental Settings: Basic Expectations for Safe Care The following is a companion to
Old Dominion University Norfolk, VA DENTAL HYGIENE PROGRAM CANDIDATED INFORMATION FOR THE CITA ADMINISTERED ADEX HYGIENE EXAMINATION
 Old Dominion University Norfolk, VA DENTAL HYGIENE PROGRAM CANDIDATED INFORMATION FOR THE CITA ADMINISTERED ADEX HYGIENE EXAMINATION Updated: 2/16/18 PLEASE READ! The intent of the information contained
Old Dominion University Norfolk, VA DENTAL HYGIENE PROGRAM CANDIDATED INFORMATION FOR THE CITA ADMINISTERED ADEX HYGIENE EXAMINATION Updated: 2/16/18 PLEASE READ! The intent of the information contained
Instructions for Use. BASAGLAR KwikPen. insulin glargine injection (100 units/ml, 3 ml pen)
 Instructions for Use BASAGLAR KwikPen insulin glargine injection (100 units/ml, 3 ml pen) 1 Read the Instructions for Use before you start using BASAGLAR and each time you get another BASAGLAR KwikPen.
Instructions for Use BASAGLAR KwikPen insulin glargine injection (100 units/ml, 3 ml pen) 1 Read the Instructions for Use before you start using BASAGLAR and each time you get another BASAGLAR KwikPen.
CHAPTER 7 Medical/Surgical Asepsis and Infection Control
 CHAPTER 7 Medical/Surgical Asepsis and Infection Control 1 Slide 1 Microorganisms Microscopic. Naturally present on and in the human body and environment. Some microorganisms (pathogens) cause specific
CHAPTER 7 Medical/Surgical Asepsis and Infection Control 1 Slide 1 Microorganisms Microscopic. Naturally present on and in the human body and environment. Some microorganisms (pathogens) cause specific
Blood is to be collected only by trained personnel working under the direction of a qualified licensed physician.
 Collection of Blood Blood is to be collected only by trained personnel working under the direction of a qualified licensed physician. Blood collection must be by aseptic methods, using a sterile closed
Collection of Blood Blood is to be collected only by trained personnel working under the direction of a qualified licensed physician. Blood collection must be by aseptic methods, using a sterile closed
Parametric Release Regulatory Requirements. Terry E. Munson Technical Vice President PAREXEL International, LLC
 Parametric Release Regulatory Requirements Terry E. Munson Technical Vice President History First application approved for parametric release in 1985 Formed the basis for the FDA Compliance Policy Guide
Parametric Release Regulatory Requirements Terry E. Munson Technical Vice President History First application approved for parametric release in 1985 Formed the basis for the FDA Compliance Policy Guide
Patient/Carer instructions for the administration of Subcutaneous Cytarabine
 Patient/Carer instructions for the administration of Subcutaneous Cytarabine This document covers the following information: What cytarabine is What subcutaneous means What happens if you decide to inject
Patient/Carer instructions for the administration of Subcutaneous Cytarabine This document covers the following information: What cytarabine is What subcutaneous means What happens if you decide to inject
Table 1 Model of STRIDESMOOTH (Stride) and STRIDESMOOTH+ Microcatheter
 AMM-Q269 Ver.7 Page 2 of 5 Table 1 Model of STRIDESMOOTH (Stride) and STRIDESMOOTH+ Microcatheter Product name STRIDESMOOTH (Stride) Model No. STD105-22S STD125-22S STD125-22A STD130-22S STD150-22S STD105-26S
AMM-Q269 Ver.7 Page 2 of 5 Table 1 Model of STRIDESMOOTH (Stride) and STRIDESMOOTH+ Microcatheter Product name STRIDESMOOTH (Stride) Model No. STD105-22S STD125-22S STD125-22A STD130-22S STD150-22S STD105-26S
Sample Enhanced Visual Inspection of medical devices. SUBJECT: Enhanced Visual Inspection of medical devices
 Sample Enhanced Visual Inspection of medical devices SUBJECT: Enhanced Visual Inspection of medical devices DEPARTMENT: Sterile Processing Area** APPROVED BY: EFFECTIVE: REVISED: 9/2017 PURPOSE: The purpose
Sample Enhanced Visual Inspection of medical devices SUBJECT: Enhanced Visual Inspection of medical devices DEPARTMENT: Sterile Processing Area** APPROVED BY: EFFECTIVE: REVISED: 9/2017 PURPOSE: The purpose
Sharps Container (not included) 1 Gather and check supplies. Gather supplies
 Instructions for Use BENLYSTA (ben-list-ah) (belimumab) injection, for subcutaneous use Prefilled Syringe Read this Instructions for Use before you start to use BENLYSTA and each time you refill your prescription.
Instructions for Use BENLYSTA (ben-list-ah) (belimumab) injection, for subcutaneous use Prefilled Syringe Read this Instructions for Use before you start to use BENLYSTA and each time you refill your prescription.
Harvard University Exposure Control Plan
 Harvard University Exposure Control Plan Harvard University is committed to providing a safe and healthy work environment. In accordance with this goal, the Occupational Health and Safety Administration
Harvard University Exposure Control Plan Harvard University is committed to providing a safe and healthy work environment. In accordance with this goal, the Occupational Health and Safety Administration
> > > RESPIRATORY PROTECTION PANDEMIC PREPAREDNESS RESPIRATORY PRODUCTS
 > > > RESPIRATORY PROTECTION PANDEMIC PREPAREDNESS RESPIRATORY PRODUCTS According to the Occupational Health & Safety Administration (OSHA), as employers evaluate their workplace, respirators should be
> > > RESPIRATORY PROTECTION PANDEMIC PREPAREDNESS RESPIRATORY PRODUCTS According to the Occupational Health & Safety Administration (OSHA), as employers evaluate their workplace, respirators should be
Sterile hypodermic syringes for single use. Part 1: Syringes for manual use
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 7886-1 Second edition 2017-05 Sterile hypodermic syringes for single use Part 1: Syringes for manual use Seringues hypodermiques stériles, non
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 7886-1 Second edition 2017-05 Sterile hypodermic syringes for single use Part 1: Syringes for manual use Seringues hypodermiques stériles, non
BAYER: KOGENATE FS WITH BIOSET (Recombinant FVIII)
 BAYER: KOGENATE FS WITH BIOSET (Recombinant FVIII) KOGENATE FS, Recombinant Antihemophilic Factor, is indicated for the prevention and control of bleeding episodes in people with hemophilia A (classical
BAYER: KOGENATE FS WITH BIOSET (Recombinant FVIII) KOGENATE FS, Recombinant Antihemophilic Factor, is indicated for the prevention and control of bleeding episodes in people with hemophilia A (classical
Using and Referencing ISO and IEC Standards for Technical Regulation
 Using and Referencing ISO and IEC Standards for Technical Regulation Third ANSI Conference on U.S. Leadership in ISO and IEC May 23-24, 24, 2007 Chicago, IL David P. Kelly, M.I.M. Office of International
Using and Referencing ISO and IEC Standards for Technical Regulation Third ANSI Conference on U.S. Leadership in ISO and IEC May 23-24, 24, 2007 Chicago, IL David P. Kelly, M.I.M. Office of International
TJF-160F/VF Cleaning and Disinfection Checklist
 TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide)
 Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide) A Guide for Patients in the Home Phone Number: Nurse/Contact: Central Venous Catheters This manual is a guide for
Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide) A Guide for Patients in the Home Phone Number: Nurse/Contact: Central Venous Catheters This manual is a guide for
No. GA Page 1 of 7 Effective Date: 03/04/ CFR (c), ANSI/ISEA Z TITLE: Chancellor Date: 03/04/16
 TEXAS STATE TECHNICAL COLLEGE STATEWIDE OPERATING STANDARD No. GA 1.6.9 Page 1 of 7 Effective Date: 03/04/16 DIVISION: SUBJECT: AUTHORITY: General Administration Eyewash and Shower Stations 29 CFR 1910.151(c),
TEXAS STATE TECHNICAL COLLEGE STATEWIDE OPERATING STANDARD No. GA 1.6.9 Page 1 of 7 Effective Date: 03/04/16 DIVISION: SUBJECT: AUTHORITY: General Administration Eyewash and Shower Stations 29 CFR 1910.151(c),
HOW TO USE... 5mg. Pocket Guide
 HOW TO USE... Pocket Guide 4 ZOMACTON [somatropin (rdna origin)] for Injection Supplies ZOMACTON 5 mg (powder) Preparation syringe Syringe and injection needle Diluent (liquid) Diluent (liquid) contains
HOW TO USE... Pocket Guide 4 ZOMACTON [somatropin (rdna origin)] for Injection Supplies ZOMACTON 5 mg (powder) Preparation syringe Syringe and injection needle Diluent (liquid) Diluent (liquid) contains
Sterile Packaging: An Examination of Statistics and Sampling
 Sterile Packaging: An Examination of Statistics and Sampling Introduction Principles of Risk Management Basics of sampling Statistical considerations for balancing costs and risks Issues such as sterilization
Sterile Packaging: An Examination of Statistics and Sampling Introduction Principles of Risk Management Basics of sampling Statistical considerations for balancing costs and risks Issues such as sterilization
Tips for Successful Venipuncture
 Page 1 of 5 Patient Positioning Have patient lie or sit down. Never draw blood on a patient who is standing. Make the patient as comfortable as you possibly can. Always watch and ask patient if he/she
Page 1 of 5 Patient Positioning Have patient lie or sit down. Never draw blood on a patient who is standing. Make the patient as comfortable as you possibly can. Always watch and ask patient if he/she
PATIENT CARE PLAN FOR CARE OF PERIPHERAL MIDLINE. Manufacturers specific recommendations should be noted and adhered to by individual practitioners.
 PATIENT CARE PLAN FOR CARE OF PERIPHERAL MIDLINE The care plan is designed to be used in conjunction with CINS Guidelines for vascular devices. Manufacturers specific recommendations should be noted and
PATIENT CARE PLAN FOR CARE OF PERIPHERAL MIDLINE The care plan is designed to be used in conjunction with CINS Guidelines for vascular devices. Manufacturers specific recommendations should be noted and
LIFE SCIENCES INDUSTRY-SPECIFIC PROTECTION SOLUTIONS QUICK REFERENCE GUIDE
 LIFE SCIENCES INDUSTRY-SPECIFIC PROTECTION SOLUTIONS QUICK REFERENCE GUIDE FEATURED PRODUCTS TouchNTuff 73-500 STERILE Key Features Thin neoprene 5 mil Ansell ERGOFORM Technology Easy Double Donning Neoprene
LIFE SCIENCES INDUSTRY-SPECIFIC PROTECTION SOLUTIONS QUICK REFERENCE GUIDE FEATURED PRODUCTS TouchNTuff 73-500 STERILE Key Features Thin neoprene 5 mil Ansell ERGOFORM Technology Easy Double Donning Neoprene
Chapter 5: Checking and Maintaining Ultrasound Equipment
 Chapter 5: Checking and Maintaining Ultrasound Equipment Care and Cleaning (BB1564-AJ) Chapter 5: Checking and Maintaining Ultrasound Equipment 45 Ultrasound equipment requires regular checks and maintenance.
Chapter 5: Checking and Maintaining Ultrasound Equipment Care and Cleaning (BB1564-AJ) Chapter 5: Checking and Maintaining Ultrasound Equipment 45 Ultrasound equipment requires regular checks and maintenance.
Talon Compounding Pharmacy 10/3/17
 Talon Compounding Pharmacy 10/3/17 Office of Pharmaceutical Quality Operations, Division II 4040 N. Central Expressway, Suite 300 Dallas, Texas 75204 October 3, 2017 CMS Case # 522630 VIA UPS EXPRESS WARNING
Talon Compounding Pharmacy 10/3/17 Office of Pharmaceutical Quality Operations, Division II 4040 N. Central Expressway, Suite 300 Dallas, Texas 75204 October 3, 2017 CMS Case # 522630 VIA UPS EXPRESS WARNING
HumaPen LUXURA HD INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE
 1 HumaPen LUXURA HD INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. The color of your HumaPen
1 HumaPen LUXURA HD INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. The color of your HumaPen
Monitoring The Sterilization Process. Are We Over The Top?
 Monitoring The Sterilization Process. Are We Over The Top? Dr Brian Kirk, Senior Technical Services Specialist, Sterilization 3M Health Care Are We Over The Top? NO! Get it wrong and you kill the patient!
Monitoring The Sterilization Process. Are We Over The Top? Dr Brian Kirk, Senior Technical Services Specialist, Sterilization 3M Health Care Are We Over The Top? NO! Get it wrong and you kill the patient!
Unidirectional Airflow Smoke Test Protocol
 Unidirectional Airflow Smoke Test Protocol Persons signing as approvers shall have examined the Unidirectional Airflow Protocol. This approval shall signify that testing of operational parameters is adequate
Unidirectional Airflow Smoke Test Protocol Persons signing as approvers shall have examined the Unidirectional Airflow Protocol. This approval shall signify that testing of operational parameters is adequate
Mallet finger injury: non-surgical treatment
 Mallet finger injury: non-surgical treatment Information for patients from the East Kent Hand Service This leaflet provides general information about mallet finger injuries to the fingers and thumb and
Mallet finger injury: non-surgical treatment Information for patients from the East Kent Hand Service This leaflet provides general information about mallet finger injuries to the fingers and thumb and
INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 4074 First edition 2002-02-15 Natural latex rubber condoms Requirements and test methods Préservatifs masculins en latex de caoutchouc naturel Exigences et méthodes d'essai Reference
INTERNATIONAL STANDARD ISO 4074 First edition 2002-02-15 Natural latex rubber condoms Requirements and test methods Préservatifs masculins en latex de caoutchouc naturel Exigences et méthodes d'essai Reference
Bloodborne Pathogens. Post-Exposure Incident Packet. An Informational Guide
 Bloodborne Pathogens Post-Exposure Incident Packet An Informational Guide Faribault Public Schools Bloodborne Pathogens Post-Exposure Incident Packet This packet has been developed as an informational
Bloodborne Pathogens Post-Exposure Incident Packet An Informational Guide Faribault Public Schools Bloodborne Pathogens Post-Exposure Incident Packet This packet has been developed as an informational
Certifications & Approvals
 Certifications & Approvals Summary Food contact approvals. RoHS and WEEE European directives. UL/CSA. Packaging regulations. Other regulations. Where to find the ARMOR certificates. Certifications & Approvals
Certifications & Approvals Summary Food contact approvals. RoHS and WEEE European directives. UL/CSA. Packaging regulations. Other regulations. Where to find the ARMOR certificates. Certifications & Approvals
How to Use ENBREL : Vial Adapter Method
 How to Use ENBREL : Vial Adapter Method SETTING UP FOR AN INJECTION Select a clean, well-lit, flat working surface, such as a table. Take the ENBREL dose tray out of the refrigerator and place it on your
How to Use ENBREL : Vial Adapter Method SETTING UP FOR AN INJECTION Select a clean, well-lit, flat working surface, such as a table. Take the ENBREL dose tray out of the refrigerator and place it on your
DO NOT TRANSFER TO A SYRINGE SEVERE OVERDOSE CAN RESULT
 HP8824 Instructions for Use ENTUZITY KwikPen (insulin injection, human biosynthetic) 500 units/ml, 3 ml pen www.lilly.ca IMPORTANT Know your dose of ENTUZITY insulin. The Pen delivers your dose in insulin
HP8824 Instructions for Use ENTUZITY KwikPen (insulin injection, human biosynthetic) 500 units/ml, 3 ml pen www.lilly.ca IMPORTANT Know your dose of ENTUZITY insulin. The Pen delivers your dose in insulin
From Farm to Label How Nutrition gets to Point of Shelf Labeling Supermarket Dietitian Symposium 2012
 From Farm to Label How Nutrition gets to Point of Shelf Labeling Supermarket Dietitian Symposium 2012 By: Patty Packard MS, RD Director of Nutrition and Regulatory Vestcom Agenda Cover the analysis of
From Farm to Label How Nutrition gets to Point of Shelf Labeling Supermarket Dietitian Symposium 2012 By: Patty Packard MS, RD Director of Nutrition and Regulatory Vestcom Agenda Cover the analysis of
How to use your Pergoveris pre-filled pen
 How to use your Pergoveris pre-filled pen Information in this user guide is based on the European product information for Pergoveris. It is not country specific and may vary from country to country. Please
How to use your Pergoveris pre-filled pen Information in this user guide is based on the European product information for Pergoveris. It is not country specific and may vary from country to country. Please
Practical Application of Basic Sterilization Principles
 Practical Application of Basic Sterilization Principles By DORIS C. MacCLELLAND, RN, MS Commander, United States Navy Nurse Corps The opinions or assertions contained herein are those of the author and
Practical Application of Basic Sterilization Principles By DORIS C. MacCLELLAND, RN, MS Commander, United States Navy Nurse Corps The opinions or assertions contained herein are those of the author and
A GUIDE TO OUR EXCLUSIVE FAMILY OF GLOVE PRODUCTS
 A GUIDE TO OUR EXCLUSIVE FAMILY OF GLOVE PRODUCTS US FED Cleanroom Standards www.techniglove.com Delivering exceptional glove technology with industry leading features and performance for over 19 years.
A GUIDE TO OUR EXCLUSIVE FAMILY OF GLOVE PRODUCTS US FED Cleanroom Standards www.techniglove.com Delivering exceptional glove technology with industry leading features and performance for over 19 years.
SEVENTH INTERNATIONAL WORKSHOP ON DOSIMETRY FOR RADIATION PROCESSING
 INTRODUCTION ASTM Subcommittee E10.01, Radiation Processing: Dosimetry and Applications, is proud to present the seventh edition of our International Workshop on Dosimetry for Radiation Processing. This
INTRODUCTION ASTM Subcommittee E10.01, Radiation Processing: Dosimetry and Applications, is proud to present the seventh edition of our International Workshop on Dosimetry for Radiation Processing. This
Sterility Assurance and Risk Management: A CDER Microbiologist s Perspective
 Sterility Assurance and Risk Management: A CDER Microbiologist s Perspective John W. Metcalfe, Ph.D. Senior Review Microbiologist FDA/CDER/OPQ/OPF/Division of Microbiology Assessment Center for Drug Evaluation
Sterility Assurance and Risk Management: A CDER Microbiologist s Perspective John W. Metcalfe, Ph.D. Senior Review Microbiologist FDA/CDER/OPQ/OPF/Division of Microbiology Assessment Center for Drug Evaluation
Reprocessing Guide. Autoclavable Arthroscope and Hardware Sterilization Tray
 Reprocessing Guide Autoclavable Arthroscope and Hardware Sterilization Tray 0233032116 Contents Introduction...2 Intended Use of Sterilization Trays...4 Warnings...5 Cautions...5 Instructions...6 Sterilization
Reprocessing Guide Autoclavable Arthroscope and Hardware Sterilization Tray 0233032116 Contents Introduction...2 Intended Use of Sterilization Trays...4 Warnings...5 Cautions...5 Instructions...6 Sterilization
2013 USP Chapter <797> Compliance Survey & Current Practice Standards
 2013 USP Chapter Compliance Survey & Current Practice Standards Kenneth S. Latta, BS, RPh Gates Healthcare Associates, Inc Hillsborough, NC We Welcome Your Questions You can submit questions throughout
2013 USP Chapter Compliance Survey & Current Practice Standards Kenneth S. Latta, BS, RPh Gates Healthcare Associates, Inc Hillsborough, NC We Welcome Your Questions You can submit questions throughout
Uni-directional Airflow Systems Working Height Velocity (WHV) Gordon Farquharson July 2017
 Uni-directional Airflow Systems Working Height Velocity (WHV) Gordon Farquharson July 2017 Agenda: Uni-directional Airflow Systems Regulatory requirements & expectations. Measurement methods. Location
Uni-directional Airflow Systems Working Height Velocity (WHV) Gordon Farquharson July 2017 Agenda: Uni-directional Airflow Systems Regulatory requirements & expectations. Measurement methods. Location
I.C.E. Embryo Vitrification Kit
 I.C.E. Embryo Vitrification Kit Vitrification Media V1, V2, V3 ICE Embryo Vitrification Instructions For Use Testing and Cautions Innvative Cryo Enterprises LLC 317 Springfield Road Linden, New Jersey
I.C.E. Embryo Vitrification Kit Vitrification Media V1, V2, V3 ICE Embryo Vitrification Instructions For Use Testing and Cautions Innvative Cryo Enterprises LLC 317 Springfield Road Linden, New Jersey
For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges.
 MS9662 MS9663 HumaPen LUXURA INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. Your HumaPen LUXURA
MS9662 MS9663 HumaPen LUXURA INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. Your HumaPen LUXURA
Guidance for Reduced Oxygen Packaging
 Consumer Protection Division Policy, Standards, And Quality Assurance Section Public Sanitation And Retail Food Safety Unit INTRODUCTION Guidance for Reduced Oxygen Packaging While reduced oxygen packaging
Consumer Protection Division Policy, Standards, And Quality Assurance Section Public Sanitation And Retail Food Safety Unit INTRODUCTION Guidance for Reduced Oxygen Packaging While reduced oxygen packaging
The University of Toledo Medical Center and its Medical Staff
 Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
Respiratory Protection for Exposures to the Swine Influenza A (H1N1) Virus: Health Care Workers
 3M Canada Company P. O. Box / C.P. 5757 Compagnie 3M Canada London, Ontario N6A 4T1 3 April 29, 2009 Respiratory Protection for Exposures to the Swine Influenza A (H1N1) Virus: Health Care Workers Frequently
3M Canada Company P. O. Box / C.P. 5757 Compagnie 3M Canada London, Ontario N6A 4T1 3 April 29, 2009 Respiratory Protection for Exposures to the Swine Influenza A (H1N1) Virus: Health Care Workers Frequently
For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges.
 MS9673 HumaPen LUXURA HD INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. The colour of your HumaPen
MS9673 HumaPen LUXURA HD INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. The colour of your HumaPen
Oral Specimen Collection Device
 Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
Dentium Surgery Kit Dental Implant Surgical Instrument Set
 www.dentium.com Dentium Surgery Kit Dental Implant Surgical Instrument Set Dentium Surgery Kit is a cassette of surgical instruments for periodontal and dental implant surgery. The cassette is divided
www.dentium.com Dentium Surgery Kit Dental Implant Surgical Instrument Set Dentium Surgery Kit is a cassette of surgical instruments for periodontal and dental implant surgery. The cassette is divided
Sterile Product Preparation for Non-Pharmacy Personnel Dennis Sinclair, R.Ph..
 Sterile Product Preparation for Non-Pharmacy Personnel Dennis Sinclair, R.Ph.. Objectives Describe the importance of accuracy and use of proper aseptic technique when preparing IV medications. Identify
Sterile Product Preparation for Non-Pharmacy Personnel Dennis Sinclair, R.Ph.. Objectives Describe the importance of accuracy and use of proper aseptic technique when preparing IV medications. Identify
Carnitine / Acylcarnitines Dried Blood Spots LC-MS/MS Analysis Kit User Manual
 Page 1 / 14 Carnitine / Acylcarnitines Dried Blood Spots LC-MS/MS Analysis Kit User Manual ZV-3051-0200-15 200 Page 2 / 14 Table of Contents 1. INTENDED USE... 3 2. SUMMARY AND EXPLANATION... 3 3. TEST
Page 1 / 14 Carnitine / Acylcarnitines Dried Blood Spots LC-MS/MS Analysis Kit User Manual ZV-3051-0200-15 200 Page 2 / 14 Table of Contents 1. INTENDED USE... 3 2. SUMMARY AND EXPLANATION... 3 3. TEST
Capital Events. Honors & Awards Banquet. Planner of The Year - Joice Ventry (center) with Retha Nero (left) and Amy Brown (right)
 June 2010 Volume 7, Issue 6 Capital Events Honors & Awards Banquet Special Interest Articles: Annual Honors & Awards Banquet Award Recipients Congratulations! It was an exciting night for the award recipients
June 2010 Volume 7, Issue 6 Capital Events Honors & Awards Banquet Special Interest Articles: Annual Honors & Awards Banquet Award Recipients Congratulations! It was an exciting night for the award recipients
SelectTech 3.1 Bench Assembly / Owner s Manual
 SelectTech 3.1 Bench Assembly / Owner s Manual This product is compliant with the applicable CE requirements. Congratulations on your commitment to fitness and your purchase of the Bowflex SelectTech 3.1
SelectTech 3.1 Bench Assembly / Owner s Manual This product is compliant with the applicable CE requirements. Congratulations on your commitment to fitness and your purchase of the Bowflex SelectTech 3.1
Hygiene and Sanitation PDG Conference Call Minutes Date: December 13, 2013
 Hygiene and Sanitation - Conference Call 12-13-13 Mission: To provide a forum to discuss items of interest for the production and processing of safe and quality dairy products and to develop program topics
Hygiene and Sanitation - Conference Call 12-13-13 Mission: To provide a forum to discuss items of interest for the production and processing of safe and quality dairy products and to develop program topics
