Interleukin-2-inducible T Cell Kinase (Itk) Network Edge Dependence for the Maturation of i NKT Cell* Qian Qi , Mingcan Xia , Yuting Bai
|
|
|
- Stewart Atkinson
- 6 years ago
- Views:
Transcription
1 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 1, pp , January 7, by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Interleukin-2-inducible T Cell Kinase (Itk) Network Edge Dependence for the Maturation of inkt Cell * Received for publication, May 25, 2010, and in revised form, October 22, 2010 Published, JBC Papers in Press, October 29, 2010, DOI /jbc.M Qian Qi 1, Mingcan Xia 1, Yuting Bai, Sanhong Yu 1, Margherita Cantorna, and Avery August 2 From the Center for Molecular Immunology & Infectious Disease and Department of Veterinary & Biomedical Sciences and Immunology & Infectious Disease Graduate Program, The Pennsylvania State University, University Park, Pennsylvania and the Department of Microbiology & Immunology, Cornell University, Ithaca, New York Invariant natural killer T (inkt) cells are a unique subset of innate T lymphocytes that are selected by CD1d. They have diverse immune regulatory functions via the rapid production of interferon- (IFN- ) and interleukin-4 (IL-4). In the absence of signaling nodes Itk and Txk, Tec family non-receptor tyrosine kinases, mice exhibit a significant block in inkt cell development. We now show here that although the Itk node is required for inkt cell maturation, the kinase domain edge of Itk is not required for continued maturation inkt cells in the thymus compared with Itk-null mice. This rescue is dependent on the expression of the Txk node. Furthermore, this kinase domain independent edge rescue correlates with the increased expression of the transcription factors T-bet, the IL-2/IL-15 receptor chain CD122, and suppression of eomesodermin expression. By contrast, -galactosyl ceramide induced cytokine secretion is dependent on the kinase domain edge of Itk. These findings indicate that the Itk node uses a kinase domain independent edge, a scaffolding function, in the signaling pathway leading to the maturation of inkt cells. Furthermore, the findings indicate that phosphorylation of substrates by the Itk node is only partially required for maturation of inkt cells, while functional activation of inkt cells is dependent on the kinase domain/activity edge of Itk. Natural killer T cells (NKT cells) 3 are a unique population of innate-like T lymphocytes that play important functions in diverse immune responses. Invariant NKT (inkt) cells are the dominant subset of NKT cells that express an invariant TCR chain (V 14-J 18). When activated, inkt cells rapidly produce large amounts of IL-4 and IFN, along with a number of other cytokines and chemokines. inkt cells also regulate other immune cells, such as NK cells and B cells, thus playing multiple roles in immune responses. inkt cells develop from the DP (CD4 CD8 double-positive) thymocytes and are selected by the MHC class I-like molecule CD1d expressed on DP cells (1). After selection, the inkt precursors * This work was supported, in whole or in part, by National Institutes of Health Grants AI and AI (to A. A.). 1 Graduate Fellows of the Huck Institutes for the Life Sciences. 2 To whom correspondence should be addressed: Dept. of Microbiology & Immunology, College of Veterinary Medicine, Cornell University, C5 181 VMC, Ithaca, NY averyaugust@cornell.edu. 3 The abbreviations used are: NKT, natural killer T cells; inkt, invariant NKT cells; DP, CD4 and CD8 double-positive; Kin, kinase domain-deleted Itk; Itk, interleukin-2 inducible T cell kinase; SLAM, signaling lymphocyticactivation molecule. (CD44 low NK1.1 ) further develop through more mature CD44 hi NK1.1 inkt to finally mature CD44 hi NK1.1 inkt cells. A number of signaling molecules, transcription factors and cytokine receptors have been identified to be important for inkt cell development through different stages. For example, SLAM, SLAM-associated protein (SAP), and Src-family kinase FYN and NF B have been suggested to form a signaling pathway that controls inkt cell development at a very early stage (2 11). The cytokine IL-15, the vitamin D receptor, PTEN, SLP76 and transcription factors T-bet and AP-1 are all involved in the final maturation of inkt cells (12 18). Itk and Txk are two Tec family non-receptor tyrosine kinases expressed in T cells. Itk-null mice show impaired Th2 cell development, as well as cytokine secretion. In addition, both Itk and Txk regulate the development of naive phenotype CD4 and CD8 T cells (19 22). Itk-null mice have also been shown to have reduced inkt cell number, impaired maturation and cytokine secretion, which is exaggerated in Itk/Txk DKO mice (23, 24). Thus, signals through Itk and Txk nodes are important for T cell development and function. Signal transduction pathways travel via nodes along the pathway. Each node has at least 2 edges, an input edge and an output edge (25). Multi-domain signaling proteins may contain more than 1 input or output edges (26). Itk has at least 2 input edges, coming from upstream Src family kinases and from PI3 kinase (27). At least 1 output edge has been identified for Itk, tyrosine phosphorylation of PLC- 1 and subsequent calcium influx, leading to the activation of NF B, NFAT, and Ras (27). Whether the Itk node has a single edge leading to PLC- 1 activation, or whether it has multiple edges leading to other signaling pathways is not clear. There is evidence for other edges with which Itk can connect to downstream signaling pathways independent of the edge leading to PLC- 1 phosphorylation. Itk can interact with the adaptor protein SLP-76 via its SH2 and SH3 domains (28 31). This interaction is not dependent on its kinase activity and could represent additional edges that emanate from the Itk signaling node. Indeed, kinase-deleted or kinase-inactive Itk have been shown to activate the transcription factor serum response factor or induce antigen-induced actin polarization in T cell lines (29, 32, 33). These data suggest that additional edges emanate from the Itk signaling node, independent of the edge leading to tyrosine phosphorylation of PLC- 1. However, whether such edges are important in vivo is not clear. We and others (29, 32) have shown that the Itk node has edges that can activate specific pathways in a kinase inde- 138 JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 1 JANUARY 7, 2011
2 pendent manner. We have also shown that conventional T cells require the kinase domain edge of Itk for development (22). We show here that a kinase domain mutant of Itk partially rescues the maturation defect observed in Itk-null inkt cells. This rescue correlates with increased expression of CD122 and T-bet, as well as reduction in the elevated levels of eomesodermin observed in Itk-null inkt cells. However, cytokine secretion by inkt cells is dependent on the ability of Itk to phosphorylate downstream substrates. These data suggest that the Itk node downstream of the TcR has edges that are both kinase-dependent and kinase independent, which function in inkt cell development. Role of Itk Kinase in inkt Cell Maturation EXPERIMENTAL PROCEDURES Mice Wild-type (WT), Itk-null, Itk/Txk DKO (kind gift of Dr. Pamela Schwartzberg, NHGRI/NIH), Tg(Lck-Itk Kin)/ Itk /, Tg(Lck-Itk Kin)/Itk/Txk DKO, and Tg(CD2-ItkK390R)/ Itk / mice were on C57BL/6 background and kept in specific pathogen-free conditions. Tg(Lck-Itk Kin)/Itk / mice were generated as described (22). Tg(Lck-Itk Kin)/Itk/Txk DKO mice were generated by crossing Tg(Lck-Itk Kin)/Itk / mice and Itk/Txk DKO mice. Tg(CD2-ItkK390R)/Itk / mice were generated by cloning a K390R mutant of Itk into the a transgenic expression cassette driven by the CD2 promoter and CD2 enhancer (35). These mice were backcrossed to the C57Bl/6 background over five generations and crossed to Itknull mice. All the mice used were 6 8 weeks of age. Experiments were approved by the IACUC at the Pennsylvania State University. Flow Cytometry Cells were incubated with Fc block (BD Bioscience) for 10 min on ice, and then were incubated with fluorescent antibodies for 30 min on ice. PE-PBS57/CD1d tetramer and unloaded/cd1d tetramer were provided by the National Institutes of Health tetramer core facility. anti- PECy5-TCR, PECy5-CD44, and PECy7-CD4 were from BD Bioscience; anti-pecy7-nk1.1, biotin-cd122, PECy5-strepavidin, APC-alexa750-TCR, APC-alexa-CXCR3, APCalexa647-T-bet were from ebiosciences; anti-ecd CD8 was from Caltag. Cell Sorting and Real-time PCR Analysis Total inkt cells (TCR PBS-57/CD1d tetramer ) were sorted from the thymi of mice using a Cytopeia Influx Cell Sorter (Cytopeia, Seattle, WA) and RNA were extracted using RNase Mini kit (Qiagen). cdna was generated with You Prime First-Strand beads kit (GE Health) and quantitative real-time PCR was performed using primer/probe sets for T-bet, PLZF and eomesodermin, with GAPDH as a housekeeping gene (Applied Biosystems). Data were analyzed using the comparative threshold cycle CT method, normalized to the expression of GAPDH, and the values were expressed as fold increase compared with WT inkt cells, which was set as 1. In Vivo BrdU Incorporation Assay WT, Itk-null, and Tg(Lck-Itk Kin)/Itk / mice were injected intraperitoneally with 1 g of BrdU and placed on drinking water containing 0.8 mg/ml BrdU for the next 6 days. BrdU-containing drinking water was changed every 2 days. 6 days after injection, mice were sacrificed, and thymocytes isolated and stained with PE-PBS57/CD1d tetramer, followed by fixation and permeabilization. BrdU was detected using APC-BrdU kit from BD Biosciences. -Galactosyl Ceramide Stimulation and Cytokine Secretion Assays WT, Itk-null, and Tg(Lck-Itk Kin)/Itk / mice were injected intraperitoneally with 2 gof -galactosyl ceramide. After 2 h, blood was collected and serum samples were obtained. IL-4 and IFN- level were analyzed using Bioplex (Millipore). For in vitro analysis of cytokine secretion, 96-well plates were preincubated with 10 g/ml anti-cd3 and 10 g/ml anti-cd28 at 4 C overnight. Sorted hepatic inkt cells from WT, Itk-null, and Tg(Lck-Itk Kin)/Itk / mice were stimulated at /well in duplicate for 3 days, and cytokines were analyzed using Bioplex. Data Analysis Statistical evaluation was conducted for all repetitions of each experiment using Student s t test with a probability value, p 0.05, considered statistically significant. RESULTS The Kinase Domain Edge of Itk Is Required for Generating Normal Numbers of Thymic inkt Cells To determine if the kinase domain edge of Itk is required for the development of inkt cells, we compared the inkt population in WT, Itk /, Itk/Txk double knock-out (DKO) mice and mice carrying a mutant ITK lacking its kinase domain instead of WT Itk in Itk null or Itk/Txk (DKO) mice (Tg(Lck-Itk Kin)/Itk / and Tg(Lck-Itk Kin)/Itk/Txk DKO (22)). For simplicity, we refer to these as Itk / / Kin and DKO/ Kin, respectively. The expression level of the transgene in inkt cells is similar to endogenous Itk as determined by quantitative RT-PCR (data not shown). We found that the absence of Itk results in a reduction in the percent of inkt cells in the thymus, and expression of the kinase-deleted mutant of Itk could rescue this percentage to WT levels (Fig. 1A). By contrast, while the percentage of thymic inkt cells in Itk/Txk DKO was similar to that seen in the Itk / mice (i.e. reduced compared with WT mice), this percentage was not rescued by expression of the kinase-deleted mutant (Fig. 1A). The numbers of thymic inkt cells were also significantly reduced in Itk / mice compared with WT mice as previously reported (Fig. 1B and Refs. 23, 24). Expression of the kinase domain-deleted mutant in Itk / mice did not rescue the numbers of thymic inkt cells as there was no significant difference between Itk / / Kin and Itk / mice (Fig. 1B). Similarly, the numbers of thymic inkt cells was significantly reduced in Itk/Txk DKO mice compared with Itk / mice (Fig. 1B), and these numbers were not rescued by expression of the kinase domain-deleted mutant (Fig. 1, A and B). Together, these results suggest that the generation of proper numbers of inkt cells requires the kinase activity of Itk, and the expression of Txk. Role of Kinase Domain Edge of Itk in inkt Cell Maturation Analyzing the three stages of inkt cell maturation in the thymus, we found that the absence of Itk results in a significant accumulation of inkt cells at stage 2 (Fig. 2A). By contrast, the percentage of thymic inkt cells at stage 2 was reduced and the percentage at stage 3 was significantly increased in the Itk / / Kin transgenic mice compared with Itk / mice (Fig. 2A). This suggests that the kinase domain independent JANUARY 7, 2011 VOLUME 286 NUMBER 1 JOURNAL OF BIOLOGICAL CHEMISTRY 139
3 Role of Itk Kinase in inkt Cell Maturation FIGURE 1. The kinase activity edge of Itk is required for generating WT numbers of inkt cells in the thymus. A, flow cytometric analysis of inkt cells in the thymus from WT, Itk /, Itk / / Kin, and DKO/ Kin mice. Numbers indicate the percentage of inkt cells. B, numbers of inkt cells in the thymus of WT, Itk-null, Itk/Txk DKO, Itk / / Kin and DKO/ Kin mice. Data are from four independent experiments. *, p 0.05, compared with WT inkt cells;, p 0.05, compared with Itk / inkt cells. edge of Itk is sufficient to allow maturation of inkt cells through the immature stage 2 to the more mature stage 3. However, because the percentage of thymic stage 3 inkt cells in the Itk / / Kin mice is still lower than in the WT mice, the kinase activity of Itk is required for the full maturation of inkt cells through to stage 2. The percentage of stage 2 inkt cells in Itk/Txk DKO mice was similar to that seen in the absence of Itk, and expression of the kinase domain-deleted mutant did not rescue the percentage, suggesting that Txk may be involved in the non-kinase domain edge of Itk in inkt cell maturation (Fig. 2A). There are two defined subsets of murine inkt cells, CD4 or CD4 CD8 (DN). Analyzing these two populations, we found that about 50% of thymic inkt cells in WT mice were DN, which was significantly decreased to around 20% (p 0.05) in Itk / mice, demonstrating that Itk is important in inkt subset development (Fig. 2B). However, analysis of inkt cell subsets in Itk / / Kin mice revealed that the percentage of thymic DN inkt cells was similar to that in Itk / mice, indicating that the kinase activity edge of Itk is important for the generation of these two subsets of inkt (Fig. 2B). Because the kinase domain deletion mutant partly rescues the maturation of inkt cells, this also suggests that the effect of Itk on the distribution of inkt cell subsets may not be due to larger numbers of immature inkt cells in these mice. Thymic NK1.1 inkt cells have higher proliferative rates than NK1.1 inkt cells. Labeling with BrdU would therefore result in higher incorporation in the former population over the same time period. Compared with WT inkt cells, a much higher percentage of Itk / inkt cells labeled with BrdU (Fig. 2C), and as expected most of the BrdU inkt cells in Itk / mice were NK1.1, and the percentage of BrdU Itk / / Kin inkt cells was lower than those from Itk / mice, consistent with the higher percentage of mature NK1.1 inkt cells in the Itk / / Kin mice (Fig. 2C). These data add further support for a role for Itk in inkt development and maturation that is partly independent of its kinase activity. We also examined whether expression of CD1d and the SLAM family receptors, SLAM and Ly 108, on DP thymocytes are affected by Itk. We found that these receptors were expressed at similar levels on DP thymocytes from WT, Itk /, and Itk / / Kin mice, indicating that differential expression of these molecules is not responsible for the reduced inkt cell numbers in these mice (Fig. 2D). Itk is suggested to form either intramolecular folded monomers or intermolecular dimer in the cells, which may maintain Itk in the inactive state (36 39). Deletion of the kinase domain may disrupt the conformation of Itk, allowing for easier interactions between Itk and other signaling proteins, and making that edge more active. To test this possibility, we generated mice carrying an Itk mutant, K390R, which is defective in kinase activity instead of the WT Itk (Tg(CD2-ItkK390R)/ Itk /, Itk / /K390R for simplicity). This mutant of Itk has all domains intact, and instead carries a single point mutation in the kinase domain. We have shown that such a mutant likely folds in a similar fashion as the WT kinase (37). Analysis of these mice revealed that the numbers of thymic inkt cells were similar to that found in Itk / and Itk / / Kin mice, and significantly lower than WT mice (Fig. 3A). More importantly, there was a significant increase in the percentage of more mature stage 3 inkt cells in the Itk / /K390R mice, but no change in the altered CD4/DN ratio of inkt in the thymus compared with Itk / mice (Fig. 3, B and C). These data are very similar to the data from the Itk / / Kin mice, further supporting the conclusion that maturation of inkt cells is partly independent of Itk kinase activity edge. Examination of the maturation status of peripheral inkt cells reveals that expression of the Itk K390R mutant (as well as the Itk kinase mutant, data not shown) was not able to rescue the defect in maturation or numbers of splenic inkt cells (Fig. 4A). By contrast, liver inkt cell maturation is not 140 JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 1 JANUARY 7, 2011
4 Role of Itk Kinase in inkt Cell Maturation FIGURE 2. The kinase activity edge of ITK is not required for the maturation of inkt cells. A, top panel, thymocytes from WT, Itk-null, Itk/Txk DKO, Itk / / Kin, and DKO/ Kin mice were gated for inkt (tetramer TcR ) cells and examined for the maturation markers CD44 and NK1.1. Bottom panel, percentage of stage 1 (CD44 NK1.1 ), stage 2 (CD44 NK1.1 ), and stage 3 (CD44 NK1.1 ) inkt cells. *, p 0.05 compared with WT inkt cells;, p 0.05 compared with Itk / inkt cells. B, top panel, thymocytes from WT, Itk-null, and Itk / / Kin mice were gated for inkt (tetramer TcR ) cells and analyzed for CD4 and CD8. Bottom graph, percentage of thymic CD4 inkt cells. Data were from four independent experiments. C, thymocytes were gated for inkt (tetramer TcR ) cells and were analyzed for BrdU versus -Galcer/CD1d tetramer (top) or BrdU versus NK1.1 (bottom). Data were from two independent experiments. *, p 0.05 compared with WT inkt cells. D, Itk does not affect expression of CD1d or SLAM family members in inkt cells. Double-positive thymocytes from WT, Itk /, and Itk / / Kin mice were analyzed for expression of CD1d, SLAM family members SLAMf1 or SMAMf6/Ly108. JANUARY 7, 2011 VOLUME 286 NUMBER 1 FIGURE 3. Role of the kinase domain edge of Itk in inkt cell maturation. A, top panel, flow cytometric analysis of thymocytes from WT, Itk-null, and Itk / /K390R inkt mice. Bottom panel, numbers of inkt cells in the thymus. *, p 0.05 compared with WT inkt cells. B, top panel, thymocytes were gated for inkt (tetramer TcR ) cells and analyzed for the maturation markers CD44 and NK1.1. Bottom panel, percentage of stage 1 (CD44 NK1.1 ), stage 2 (CD44 NK1.1 ), and stage 3 (CD44 NK1.1 ) inkt cells. *, p 0.05 compared with WT inkt cells. **, p 0.05 compared with Itk / inkt cells. C, top panel, thymocytes were gated for inkt (tetramer TcR ) cells and analyzed for CD4 and CD8. Bottom panel, percentage of thymic CD4 inkt cells. Data were from two independent experiments. *, p 0.05 compared with WT inkt cells. affected by the absence of Itk (2, 22), and expression of the Itk K390R mutant does not affect this process (Fig. 4B). The Kinase Domain Edge of Itk Is Required for Cytokine Production of inkt Cells In the absence of Itk, inkt cells are defective in secreting IL-4 and IFN- in response to -GalCer stimulation (23, 24). To determine whether the kinase activity of Itk is important in inkt cell function, WT, Itk / and Tg(Lck-Itk Kin)/Itk / mice were injected with -GalCer JOURNAL OF BIOLOGICAL CHEMISTRY 141
5 Role of Itk Kinase in inkt Cell Maturation FIGURE 4. Kinase activity edge of Itk is required for cytokine secretion by inkt cells. A, top panel, flow cytometric analysis of splenocytes from WT, Itknull, and Itk / /K390R inkt cells. Bottom panel, numbers of inkt cells in the spleen. *, p 0.05 compared with WT inkt cells. B, liver lymphocytes were gated for inkt (tetramer TcR ) cells and analyzed for the maturation markers CD44 and NK1.1. C, WT, Itk-null, and Itk / / Kin mice were injected intraperitoneal with 2 gof -GalCer. After 2 h, serum samples from blood were collected. Cytokine levels were analyzed by Bioplex. D, hepatic inkt cells (tetramer TcR ) were sorted and stimulated with plate-bound anti-cd3 and -CD28 for 3 days. IL-4 and IFN- expression levels were analyzed by Bioplex. *, p 0.05 compared with WT inkt cells. and serum samples collected 2 h after injection. Analysis of the serum revealed that Itk / mice secreted significantly lower amounts of IL-4 and IFN- than WT mice consistent with previous reports (23, 24). Expression of kinase domaindeleted mutant in Itk-null mice did not rescue this defect in cytokine production, indicating that the kinase activity of Itk 142 JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 1 JANUARY 7, 2011
6 is required for the function of inkt cells (Fig. 4C). Because Itk-null mice (and the Itk / / Kin mice) have reduced numbers of inkt cells, we also stimulated purified hepatic inkt cells in vitro with anti-cd3 and CD28 for 3 days to confirm the reduction in cytokine secretion. We found that Itk / and Itk / / Kin inkt cells secreted comparable amounts of IL-4 and IFN-, which were significantly lower than that secreted by WT inkt cells (Fig. 4D). Altogether, these data indicate that the kinase activity edge of Itk is important for IL-4 and IFN- production of inkt cells. Kinase Domain Edge Independent Rescue of CD122 and T-bet, and Suppression of Eomesodermin Expression in Developing inkt Cells Our analysis indicates that the population of thymic CD122 (IL-2/IL-15 receptor chain) inkt cells in Itk deficient was reduced compared with WT mice (Fig. 5A). By contrast, we found that this CD122 inkt cell population was significantly higher in the Itk / / Kin mice than in Itk / mice, indicating that the increased expression of CD122 may contribute to the increased maturation of inkt cell in the mice carrying the mutant Itk. These data also suggest that Itk may regulate CD122 expression via a kinase independent edge. The transcription factor T-bet can regulate the expression of CD122, and we found that Itk / / Kin inkt cells had significantly increased levels of T-bet mrna and protein compared with Itk / inkt cells (Fig. 5, B and C). In addition, expression levels of CXCR3, another target of T-bet (40), was also rescued by the expression the equivalent K390R Itk mutant (Fig. 5D). More dramatically, the expression level of eomesodermin, another transcription factor of T-box family that also regulates CD122, was not detected in WT inkt cells, but was highly expressed in the Itk / inkt cells (Fig. 5B). Pointedly, the expression of the Itk kinase-deleted mutant significantly reduced eomesodermin expression in Itk / inkt cells (i.e. inkt cells that develop in the Itk / / Kin mice), suggesting that kinase domain independent edge signals may affect signaling pathways leading to T-bet and eomesodermin expression in inkt cells. Two recent studies have shown that that the transcription factor PLZF is important for inkt cell development at early stage (10, 11), and we found that PLZF mrna levels were significantly elevated in Itk-null inkt cells, and this was not normalized by expression of the Itk mutant (Fig. 5B). DISCUSSION We show here that the Itk node in T cell receptor signaling regulates the maturation of inkt cells in part via an edge that is kinase-independent. The partial rescue of inkt cell maturation depends on the continued expression of the related kinase node Txk, and occurs primarily by signaling the maturation of these cells through the immature stage 2 to the more mature stage 3. This correlates with increased expression of T-bet and CD122, and decreased expression of eomesodermin. Our data suggest that signals emanating from the noncatalytic domains of Itk can act as an edge in the signaling pathway that regulates the expression of these factors, thus modulating inkt cell development. Role of Itk Kinase in inkt Cell Maturation FIGURE 5. Kinase activity edge independent rescue of T-bet, CD122, and PLZF, and suppression of eomesodermin expression. A, thymocytes from WT, Itk-null, and Itk / / Kin mice were gated for inkt cells and analyzed for the expression of CD122. B, inkt cells were sorted from the thymus of WT, Itk-null, and Itk / / Kin mice and mrna levels of T-bet, PLZF, and eomesodermin were determined by quantitative real-time PCR. Data were representative of two independent experiments. *, p 0.05 compared with WT inkt cells; **, p 0.05 compared with Itk / inkt cells. C, thymocytes from WT (solid line), Itk-null (gray open line), and Itk / /K390R mice (dashed line) were gated for inkt cells and analyzed for the expression of intracellular T-bet. Tbet/Itk double knock-out mice were used as negative control cells (filled gray). D, thymocytes from WT, Itk-null, and Itk / /K390R mice were gated for inkt cells and analyzed for the expression of CXCR3. Tbet/Itk double knock-out mice were used as negative control cells to set gates (data not shown). Our analysis revealed that the number of thymic inkt cells cannot be rescued by the expression of the kinase domain mutants of Itk, indicating that the kinase activity edge is critical for transducing signals that lead to WT numbers of these cells. This could be intrinsic, or could be related to the re- JANUARY 7, 2011 VOLUME 286 NUMBER 1 JOURNAL OF BIOLOGICAL CHEMISTRY 143
7 Role of Itk Kinase in inkt Cell Maturation duced numbers of total thymocytes observed in the Itk / and Itk / / Kin mice, because the overall numbers of thymocytes, and in particular DP thymocytes, play critical roles in inkt cell development and numbers (1). Indeed, while there is a slight increase in the percentage of inkt cells in the thymus of Itk / / Kin mice, the total number of thymocytes is not rescued in these mice, and this translates into reduced numbers (although slightly higher) of inkt cells in these mice. We also tested whether the kinase-deleted mutant would behave differently from a full-length kinase that has little to no kinase activity. We compared these two mutants as it is possible that the folding of the kinase-deleted mutant may be different from the WT kinase. The structure of full-length Itk is not known, but based on a number of experiments using isolated domains, and other approaches in cells, we and others have proposed one of two models for folding of this protein, either an intramolecular folded monomer, or a intermolecular folded dimer (36, 37, 41). Deletion of the kinase domain in both models could potentially result in enhanced interactions with the SH2 and SH3 domains. However, both the kinase-deleted mutant and the kinase activity point mutant behaved in the same fashion with regards to the generation of WT numbers of inkt cells, as well as in their development and maturation, suggesting that any potential alterations in the structure of Itk does not explain our data. The related kinase Txk makes some contributions to the development of inkt cells because Itk/Txk DKO mice have significant reduction in thymic inkt cells numbers compared with both WT and in particular Itk / mice (24). We find that the kinase domain independent edge of Itk can drive the maturation of a significant percentage of stage 2 inkt cells to the more mature stage 3. However, in the absence of Txk, this does not happen. We suggest that the function of the kinase domain independent edge may be dependent on the expression of Txk. Whereas these data suggest a genetic interaction, we have not been able to get enough purified inkt cells to examine this biochemically. However, these findings suggest that kinase activity edge of Itk may be rescued by Txk, however, this may be less efficient due to the lower levels of expression of Txk in these cells (24). However, in the absence of both Itk and Txk, the kinase domain independent edge cannot drive maturation of these cells. These findings suggest that there may be some cooperation between these two nodes, Itk and Txk, such that expression of a kinase activity edge from another Tec kinase may be able to cooperate with a kinase domain independent edge of Itk in these functions. Several studies have shown that IL-15 is required for the final maturation of inkt cells and that the IL-2/IL-15 receptor -chain (CD122) is important for NKT cell development (13, 14). Thus the defect of final maturation in Itk / inkt cells may be due to the lower CD122 expression in these cells. In support of this, Felices and Berg (24) have reported that Itk-deficient inkt cells express lower levels of CD122 than WT inkt cells. Our finding that CD122 and T-bet expression are independent of the kinase domain edge of Itk suggest that the IL-15 signaling pathway may contribute to the defect of Itk-null inkt cells in maturing from stage 2 to stage 3. More dramatically, the significant increase in expression of the T-bet-related transcription factor eomesodermin in Itk / inkt cells, and its reduction upon expression of the kinase domain-deleted mutant of Itk, suggest that this kinase domain independent edge is critical in suppressing the expression of this transcription factor. It is likely that overexpression of eomesodermin alters inkt cell maturation, and that the T-bet:eomesodermin ratio is critical in inkt cell maturation. T-bet and/or eomesodermin have been shown to regulate the expression of CD122, although the exact nature of their contributions is not clear, particularly in inkt cells (42 44). It is possible that CD122 expression is strictly dependent on T-bet expression in inkt cells, and not on eomesodermin. Nevertheless, our data show that the kinase domain independent edge of Itk can partially restore this ratio or balance, thus partially restoring CD122 expression in inkt cells. By contrast, CXCR3 has been demonstrated to be a prominent target of T-bet (40), and we observed a reduction in its expression in the absence of Itk, and prominent rescue upon expression of the K390R Itk mutant, indicating that as seen for CD122, rescue of T-bet expression by the kinase domain independent edge has functional consequences in these cells. Two recent studies have shown that the transcription factor PLZF is important at an early stage of inkt cell development, with arrest at stage 1 in PLZF-null, and arrest at stage 2 in PLZF overexpressed transgenic mice (10, 11). This suggests that proper expression of PLZF is critical for normal inkt cell development and maturation. We found that PLZF mrna levels were significantly elevated in Itk-null compared with WT inkt cells, suggesting that elevated expression of PLZF may contribute to the defect in Itk / inkt cells. Indeed, transgenic overexpression of PLZF results in defects in inkt cell development, with arrest in maturation in stage 2, similar to that seen in the absence of Itk (45). In addition, inkt cells from the Itk / / Kin have levels of PLZF that is closer to that seen in Itk / inkt cells, suggesting that the interaction between the kinase domain independent edge and regulation of expression of these transcription factors may modulate the maturation of these cells. Of course, we cannot exclude the possibility that other transcription factors and signaling pathways may also be involved downstream of Itk signals to inkt cell development. Perturbation of T cell receptor signaling pathway nodes often result in different effects in inkt cell maturation and development compared with conventional T cell development (46). Indeed, the absence of the Itk node affects the development of naïve or conventional CD4 and CD8 T cell development, while leaving development of non-conventional or innate memory phenotype T cells intact (19 21). These nonconventional or innate memory phenotype T cells have properties of inkt cells, with the presence of preformed cytokine message, ability to rapidly produce cytokines, as well as the requirement for both SAP and Itk nodes for their development (47). However, while the kinase domain independent edge of Itk cannot rescue conventional T cell development (21, 22), it can partially rescue maturation of inkt cells (this work). Conventional T cells express low levels of T-bet, eome- 144 JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 1 JANUARY 7, 2011
8 sodermin, and PLZF, while non-conventional or innate memory phenotype T cells express high levels of these transcription factors (compared with naïve or conventional T cells) (19 22, 47). The ability of the kinase domain edge of Itk to regulate these factors also differs between inkt cells and non-conventional or innate memory phenotype T cells. Taken together, this suggests that while inkt cells share some characteristics with non-conventional of innate memory phenotype T cells, there are clear differences in signaling networks that control their development. Upon TcR stimulation, Itk is recruited to the cell membrane through its PH domain binding to PIP3 in the cell membrane, where Itk is phosphorylated and activated, as well as interacts with other signaling proteins, including SLP-76, LAT, GADS, PLC- 1, and Vav, to assemble the productive signaling complex and subsequently initiate the downstream signaling pathways. Itk interacts with other proteins mainly through its SH2 and SH3 domains and this adaptor function is very important for the downstream signaling pathways. Itk can regulate Vav localization and TCR-induced actin polarization independent of its kinase activity edge, but requires its PH and SH2 domains (29, 33). In addition, the SH3 domain, but not kinase activity edge of Itk, is required for antigen receptor induced transcription factor SRF activation (32). This kinase domain independent edge can partially rescue antigen receptor induced activation of Erk in Tec kinase-null DT40 cells (32). Itk may therefore utilize this edge with scaffolding function in regulating signaling pathways that contribute to the maturation of inkt cells. These pathways regulate the expression level of CD122, which in turn may be regulated by the balance of T-bet and eomesodermin expressed in these cells. The related kinase node Txk may play a part in regulating the function of this kinase independent edge. Our data indicate that the Itk kinase domain/activity edge may be targeted to affect inkt cell function, while leaving some inkt maturation intact. Acknowledgments We thank members of the August Laboratory and the CMIID for feedback and discussion. We also thank E. Kunze, N. Bem, and S. Magargee in the Center for Quantitative Cell Analysis at Penn State. Work in the Center for Molecular Immunology & Infectious Disease is supported in part under a grant from the Pennsylvania Department of Health. There are no conflicts of interests to report. REFERENCES 1. Gapin, L., Matsuda, J. L., Surh, C. D., and Kronenberg, M. (2001) Nat. Immunol. 2, Pasquier, B., Yin, L., Fondanèche, M. C., Relouzat, F., Bloch-Queyrat, C., Lambert, N., Fischer, A., de Saint-Basile, G., and Latour, S. (2005) J. Exp. Med. 201, Nichols, K. E., Hom, J., Gong, S. Y., Ganguly, A., Ma, C. S., Cannons, J. L., Tangye, S. G., Schwartzberg, P. L., Koretzky, G. A., and Stein, P. L. (2005) Nat. Med. 11, Chung, B., Aoukaty, A., Dutz, J., Terhorst, C., and Tan, R. (2005) J. Immunol. 174, Griewank, K., Borowski, C., Rietdijk, S., Wang, N., Julien, A., Wei, D. G., Mamchak, A. A., Terhorst, C., and Bendelac, A. (2007) Immunity 27, Role of Itk Kinase in inkt Cell Maturation 6. Eberl, G., Lowin-Kropf, B., and MacDonald, H. R. (1999) J. Immunol. 163, Gadue, P., Morton, N., and Stein, P. L. (1999) J. Exp. Med. 190, Stanic, A. K., Bezbradica, J. S., Park, J. J., Van Kaer, L., Boothby, M. R., and Joyce, S. (2004) J. Immunol. 172, Schmidt-Supprian, M., Tian, J., Grant, E. P., Pasparakis, M., Maehr, R., Ovaa, H., Ploegh, H. L., Coyle, A. J., and Rajewsky, K. (2004) Proc. Natl. Acad. Sci. U.S.A. 101, Savage, A. K., Constantinides, M. G., Han, J., Picard, D., Martin, E., Li, B., Lantz, O., and Bendelac, A. (2008) Immunity 29, Kovalovsky, D., Uche, O. U., Eladad, S., Hobbs, R. M., Yi, W., Alonzo, E., Chua, K., Eidson, M., Kim, H. J., Im, J. S., Pandolfi, P. P., and Sant Angelo, D. B. (2008) Nat. Immunol. 9, Townsend, M. J., Weinmann, A. S., Matsuda, J. L., Salomon, R., Farnham, P. J., Biron, C. A., Gapin, L., and Glimcher, L. H. (2004) Immunity 20, Matsuda, J. L., Gapin, L., Sidobre, S., Kieper, W. C., Tan, J. T., Ceredig, R., Surh, C. D., and Kronenberg, M. (2002) Nat. Immunol. 3, Ohteki, T., Ho, S., Suzuki, H., Mak, T. W., and Ohashi, P. S. (1997) J. Immunol. 159, Lodolce, J. P., Boone, D. L., Chai, S., Swain, R. E., Dassopoulos, T., Trettin, S., and Ma, A. (1998) Immunity 9, Yu, S., and Cantorna, M. T. (2008) Proc. Natl. Acad. Sci. U.S.A. 105, Kishimoto, H., Ohteki, T., Yajima, N., Kawahara, K., Natsui, M., Kawarasaki, S., Hamada, K., Horie, Y., Kubo, Y., Arase, S., Taniguchi, M., Vanhaesebroeck, B., Mak, T. W., Nakano, T., Koyasu, S., Sasaki, T., and Suzuki, A. (2007) Blood 109, Zullo, A. J., Benlagha, K., Bendelac, A., and Taparowsky, E. J. (2007) J. Immunol. 178, Atherly, L. O., Lucas, J. A., Felices, M., Yin, C. C., Reiner, S. L., and Berg, L. J. (2006) Immunity 25, Broussard, C., Fleischecker, C., Horai, R., Chetana, M., Venegas, A. M., Sharp, L. L., Hedrick, S. M., Fowlkes, B. J., and Schwartzberg, P. L. (2006) Immunity 25, Hu, J., and August, A. (2008) J. Immunol. 180, Hu, J., Sahu, N., Walsh, E., and August, A. (2007) Eur. J. Immunol. 37, Au-Yeung, B. B., and Fowell, D. J. (2007) J. Immunol. 179, Felices, M., and Berg, L. J. (2008) J. Immunol. 180, Kaiser, M., and Hilgetag, C. C. (2004) Biol. Cybern. 90, González-Díaz, H., González-Díaz, Y., Santana, L., Ubeira, F. M., and Uriarte, E. (2008) Proteomics 8, Berg, L. J., Finkelstein, L. D., Lucas, J. A., and Schwartzberg, P. L. (2005) Annu. Rev. Immunol. 23, Jordan, M. S., Sadler, J., Austin, J. E., Finkelstein, L. D., Singer, A. L., Schwartzberg, P. L., and Koretzky, G. A. (2006) J. Immunol. 176, Dombroski, D., Houghtling, R. A., Labno, C. M., Precht, P., Takesono, A., Caplen, N. J., Billadeau, D. D., Wange, R. L., Burkhardt, J. K., and Schwartzberg, P. L. (2005) J. Immunol. 174, Su, Y. W., Zhang, Y., Schweikert, J., Koretzky, G. A., Reth, M., and Wienands, J. (1999) Eur. J. Immunol. 29, Bunnell, S. C., Diehn, M., Yaffe, M. B., Findell, P. R., Cantley, L. C., and Berg, L. J. (2000) J. Biol. Chem. 275, Hao, S., Qi, Q., Hu, J., and August, A. (2006) FEBS Lett. 580, Grasis, J. A., Browne, C. D., and Tsoukas, C. D. (2003) J. Immunol. 170, Sahu, N., Venegas, A. M., Jankovic, D., Mitzner, W., Gomez-Rodriguez, J., Cannons, J. L., Sommers, C., Love, P., Sher, A., Schwartzberg, P. L., and August, A. (2008) J. Immunol. 181, Zhumabekov, T., Corbella, P., Tolaini, M., and Kioussis, D. (1995) J. Immunol. Methods. 185, Qi, Q., Sahu, N., and August, A. (2006) J. Biol. Chem. 281, Qi, Q., and August, A. (2009) J. Biol. Chem. 284, Colgan, J., Asmal, M., Neagu, M., Yu, B., Schneidkraut, J., Lee, Y., Sokol- JANUARY 7, 2011 VOLUME 286 NUMBER 1 JOURNAL OF BIOLOGICAL CHEMISTRY 145
9 Role of Itk Kinase in inkt Cell Maturation skaja, E., Andreotti, A., and Luban, J. (2004) Immunity 21, Andreotti, A. H., Bunnell, S. C., Feng, S., Berg, L. J., and Schreiber, S. L. (1997) Nature 385, Matsuda, J. L., Zhang, Q., Ndonye, R., Richardson, S. K., Howell, A. R., and Gapin, L. (2006) Blood 107, Brazin, K. N., Mallis, R. J., Fulton, D. B., and Andreotti, A. H. (2002) Proc. Natl. Acad. Sci. U.S.A. 99, Matsuda, J. L., George, T. C., Hagman, J., and Gapin, L. (2007) J. Immunol. 178, Intlekofer, A. M., Takemoto, N., Wherry, E. J., Longworth, S. A., Northrup, J. T., Palanivel, V. R., Mullen, A. C., Gasink, C. R., Kaech, S. M., Miller, J. D., Gapin, L., Ryan, K., Russ, A. P., Lindsten, T., Orange, J. S., Goldrath, A. W., Ahmed, R., and Reiner, S. L. (2005) Nat. Immunol. 6, Mullen, A. C., High, F. A., Hutchins, A. S., Lee, H. W., Villarino, A. V., Livingston, D. M., Kung, A. L., Cereb, N., Yao, T. P., Yang, S. Y., and Reiner, S. L. (2001) Science 292, Raberger, J., Schebesta, A., Sakaguchi, S., Boucheron, N., Blomberg, K. E., Berglöf, A., Kolbe, T., Smith, C. I., Rülicke, T., and Ellmeier, W. (2008) Proc. Natl. Acad. Sci. U.S.A. 105, Godfrey, D. I., Stankovic, S., and Baxter, A. G. (2010) Nat. Immunol. 11, Horai, R., Mueller, K. L., Handon, R. A., Cannons, J. L., Anderson, S. M., Kirby, M. R., and Schwartzberg, P. L. (2007) Immunity 27, JOURNAL OF BIOLOGICAL CHEMISTRY VOLUME 286 NUMBER 1 JANUARY 7, 2011
10 Interleukin-2-inducible T Cell Kinase (Itk) Network Edge Dependence for the Maturation of inkt Cell Qian Qi, Mingcan Xia, Yuting Bai, Sanhong Yu, Margherita Cantorna and Avery August J. Biol. Chem. 2011, 286: doi: /jbc.M originally published online October 29, 2010 Access the most updated version of this article at doi: /jbc.M Alerts: When this article is cited When a correction for this article is posted Click here to choose from all of JBC's alerts This article cites 47 references, 25 of which can be accessed free at
Tec family kinases: Itk signaling and the development of NKT ab and cd T cells
 MINIREVIEW Tec family kinases: Itk signaling and the development of NKT ab and cd T cells Qian Qi 1,2, Arun Kumar Kannan 1,2,3 and Avery August 1,2 1 Department of Veterinary & Biomedical Sciences, Center
MINIREVIEW Tec family kinases: Itk signaling and the development of NKT ab and cd T cells Qian Qi 1,2, Arun Kumar Kannan 1,2,3 and Avery August 1,2 1 Department of Veterinary & Biomedical Sciences, Center
The Adaptor Molecule Signaling Lymphocytic Activation Molecule-Associated. via Effects on GATA-3 and T-bet Expression
 This information is current as of April 21, 2018. References Subscription Permissions Email Alerts The Adaptor Molecule Signaling Lymphocytic Activation Molecule-Associated Protein (SAP) Regulates IFN-γ
This information is current as of April 21, 2018. References Subscription Permissions Email Alerts The Adaptor Molecule Signaling Lymphocytic Activation Molecule-Associated Protein (SAP) Regulates IFN-γ
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
The Tec Family Tyrosine Kinases Itk and Rlk Regulate the Development of Conventional CD8 + T Cells
 Immunity 25, 79 91, July 2006 ª2006 Elsevier Inc. DOI 10.1016/j.immuni.2006.05.012 The Tec Family Tyrosine Kinases Itk and Rlk Regulate the Development of Conventional CD8 + T Cells Luana O. Atherly, 3
Immunity 25, 79 91, July 2006 ª2006 Elsevier Inc. DOI 10.1016/j.immuni.2006.05.012 The Tec Family Tyrosine Kinases Itk and Rlk Regulate the Development of Conventional CD8 + T Cells Luana O. Atherly, 3
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
Distinct subsets of lymphocytes depend on IL-15 (1, 2). Mice
 ITK and IL-15 support two distinct subsets of CD8 T cells Sigrid Dubois, Thomas A. Waldmann*, and Jürgen R. Müller* Metabolism Branch, Center for Cancer Research, National Cancer Institute, National Institutes
ITK and IL-15 support two distinct subsets of CD8 T cells Sigrid Dubois, Thomas A. Waldmann*, and Jürgen R. Müller* Metabolism Branch, Center for Cancer Research, National Cancer Institute, National Institutes
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Altered Development of CD8 + T Cell Lineages in Mice Deficient for the Tec Kinases Itk and Rlk
 Immunity 25, 93 104, July 2006 ª2006 Elsevier Inc. DOI 10.1016/j.immuni.2006.05.011 Altered Development of CD8 + T Cell Lineages in Mice Deficient for the Tec Kinases Itk and Rlk Christine Broussard, 1,4,5
Immunity 25, 93 104, July 2006 ª2006 Elsevier Inc. DOI 10.1016/j.immuni.2006.05.011 Altered Development of CD8 + T Cell Lineages in Mice Deficient for the Tec Kinases Itk and Rlk Christine Broussard, 1,4,5
The presence of CD8 + invariant NKT cells in mice
 EXPERIMENTAL and MOLECULAR MEDICINE, Vol. 41, No. 12, 866-872, December 2009 The presence of CD8 + invariant NKT cells in mice Hyunji Lee 1, Changwan Hong 1, Junghoon Shin 1, Soohwan Oh 1, Sundo Jung 1,
EXPERIMENTAL and MOLECULAR MEDICINE, Vol. 41, No. 12, 866-872, December 2009 The presence of CD8 + invariant NKT cells in mice Hyunji Lee 1, Changwan Hong 1, Junghoon Shin 1, Soohwan Oh 1, Sundo Jung 1,
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
ROLE OF TEC FAMILY KINASE ITK IN REGULATING THE DEVELOPMENT OF T CELL SUBSETS
 The Pennsylvania State University The Graduate School The Huck Institute for Life Sciences ROLE OF TEC FAMILY KINASE ITK IN REGULATING THE DEVELOPMENT OF T CELL SUBSETS A Dissertation in Immunology and
The Pennsylvania State University The Graduate School The Huck Institute for Life Sciences ROLE OF TEC FAMILY KINASE ITK IN REGULATING THE DEVELOPMENT OF T CELL SUBSETS A Dissertation in Immunology and
Co-receptor choice by V 14i NKT cells is driven by Th-POK expression rather than avoidance of CD8-mediated negative selection
 Published Online: 19 April, 2010 Supp Info: http://doi.org/10.1084/jem.20090557 Downloaded from jem.rupress.org on December 18, 2018 Ar ticle Co-receptor choice by V 14i NKT cells is driven by Th-POK expression
Published Online: 19 April, 2010 Supp Info: http://doi.org/10.1084/jem.20090557 Downloaded from jem.rupress.org on December 18, 2018 Ar ticle Co-receptor choice by V 14i NKT cells is driven by Th-POK expression
The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep
 SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
A Role for Ly108 in the Induction of Promyelocytic Zinc Finger Transcription Factor in Developing Thymocytes
 This information is current as of August 23, 2018. Supplementary Material Subscription Permissions Email Alerts A Role for Ly108 in the Induction of Promyelocytic Zinc Finger Transcription Factor in Developing
This information is current as of August 23, 2018. Supplementary Material Subscription Permissions Email Alerts A Role for Ly108 in the Induction of Promyelocytic Zinc Finger Transcription Factor in Developing
Comparison of Invariant NKT Cells with Conventional T Cells by Using Gene Set Enrichment Analysis (GSEA)
 http://dx.doi.org/./in...6.6 pissn 9-69 eissn 9-66 ORIGINAL ARTICLE Comparison of Invariant NKT Cells with Conventional T Cells by Using Gene Set Enrichment Analysis (GSEA) Sae Jin Oh, Ji Ye Ahn and Doo
http://dx.doi.org/./in...6.6 pissn 9-69 eissn 9-66 ORIGINAL ARTICLE Comparison of Invariant NKT Cells with Conventional T Cells by Using Gene Set Enrichment Analysis (GSEA) Sae Jin Oh, Ji Ye Ahn and Doo
CD44
 MR1-5-OP-RU CD24 CD24 CD44 MAIT cells 2.78 11.2 WT RORγt- GFP reporter 1 5 1 4 1 3 2.28 1 5 1 4 1 3 4.8 1.6 8.1 1 5 1 4 1 3 1 5 1 4 1 3 3.7 3.21 8.5 61.7 1 2 1 3 1 4 1 5 TCRβ 2 1 1 3 1 4 1 5 CD44 1 2 GFP
MR1-5-OP-RU CD24 CD24 CD44 MAIT cells 2.78 11.2 WT RORγt- GFP reporter 1 5 1 4 1 3 2.28 1 5 1 4 1 3 4.8 1.6 8.1 1 5 1 4 1 3 1 5 1 4 1 3 3.7 3.21 8.5 61.7 1 2 1 3 1 4 1 5 TCRβ 2 1 1 3 1 4 1 5 CD44 1 2 GFP
Effect of IL-4 on the Development and Function of Memory-like CD8 T Cells in the Peripheral Lymphoid Tissues
 http://dx.doi.org/10.4110/in.2016.16.2.126 pissn 1598-2629 eissn 2092-6685 ORIGINAL ARTICLE Effect of IL-4 on the Development and Function of Memory-like CD8 T Cells in the Peripheral Lymphoid Tissues
http://dx.doi.org/10.4110/in.2016.16.2.126 pissn 1598-2629 eissn 2092-6685 ORIGINAL ARTICLE Effect of IL-4 on the Development and Function of Memory-like CD8 T Cells in the Peripheral Lymphoid Tissues
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
T-bet Regulates the Terminal Maturation and Homeostasis of NK and V 14i NKT Cells
 Immunity, Vol. 20, 477 494, April, 2004, Copyright 2004 by Cell Press T-bet Regulates the Terminal Maturation and Homeostasis of NK and V 14i NKT Cells Michael J. Townsend, 1 Amy S. Weinmann, 3,6 Jennifer
Immunity, Vol. 20, 477 494, April, 2004, Copyright 2004 by Cell Press T-bet Regulates the Terminal Maturation and Homeostasis of NK and V 14i NKT Cells Michael J. Townsend, 1 Amy S. Weinmann, 3,6 Jennifer
Regulation of natural killer T-cell development by deubiquitinase CYLD
 The EMBO Journal (2010) 29, 1600 1612 & 2010 European Molecular Biology Organization All Rights Reserved 0261-4189/10 www.embojournal.org Regulation of natural killer T-cell development by deubiquitinase
The EMBO Journal (2010) 29, 1600 1612 & 2010 European Molecular Biology Organization All Rights Reserved 0261-4189/10 www.embojournal.org Regulation of natural killer T-cell development by deubiquitinase
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured
 Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Page 1 of 3. Product Information Contents: ezkine CD8 Activation 2 Whole Blood Intracellular Cytokine Kit
 Page 1 of 3 ezkine CD8 Activation 2 Whole Blood Intracellular Cytokine Kit RUO: For Research Use Only. Not for use in diagnostic procedures. Staining of human whole blood with the ezkine CD8 Activation
Page 1 of 3 ezkine CD8 Activation 2 Whole Blood Intracellular Cytokine Kit RUO: For Research Use Only. Not for use in diagnostic procedures. Staining of human whole blood with the ezkine CD8 Activation
Lecture 7: Signaling Through Lymphocyte Receptors
 Lecture 7: Signaling Through Lymphocyte Receptors Questions to Consider After recognition of its cognate MHC:peptide, how does the T cell receptor activate immune response genes? What are the structural
Lecture 7: Signaling Through Lymphocyte Receptors Questions to Consider After recognition of its cognate MHC:peptide, how does the T cell receptor activate immune response genes? What are the structural
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Efficient Isolation of Mouse Liver NKT Cells by Perfusion
 Efficient Isolation of Mouse Liver NKT Cells by Perfusion Xianfeng Fang 1,2, Peishuang Du 1, Yang Liu 3 *, Jie Tang 1 * 1 Center for Infection and Immunity, Institute of Biophysics, Chinese Academy of
Efficient Isolation of Mouse Liver NKT Cells by Perfusion Xianfeng Fang 1,2, Peishuang Du 1, Yang Liu 3 *, Jie Tang 1 * 1 Center for Infection and Immunity, Institute of Biophysics, Chinese Academy of
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Granulocyte-Macrophage Colony-Stimulating Factor Regulates Effector Differentiation of Invariant Natural Killer T Cells during Thymic Ontogeny
 Immunity 25, 487 497, September 2006 ª2006 Elsevier Inc. DOI 10.1016/j.immuni.2006.06.017 Granulocyte-Macrophage Colony-Stimulating Factor Regulates Effector Differentiation of Invariant Natural Killer
Immunity 25, 487 497, September 2006 ª2006 Elsevier Inc. DOI 10.1016/j.immuni.2006.06.017 Granulocyte-Macrophage Colony-Stimulating Factor Regulates Effector Differentiation of Invariant Natural Killer
BTBD11 INTERACTS WITH PLZF AND MODULATES THE EFFECTOR FUNCTIONS OF VARIOUS INNATE LYMPHOCYTE POPULATIONS ANDREW R. CHAVKIN
 BTBD11 INTERACTS WITH PLZF AND MODULATES THE EFFECTOR FUNCTIONS OF VARIOUS INNATE LYMPHOCYTE POPULATIONS BY ANDREW R. CHAVKIN A thesis submitted to the Graduate School-New Brunswick Rutgers, The State
BTBD11 INTERACTS WITH PLZF AND MODULATES THE EFFECTOR FUNCTIONS OF VARIOUS INNATE LYMPHOCYTE POPULATIONS BY ANDREW R. CHAVKIN A thesis submitted to the Graduate School-New Brunswick Rutgers, The State
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Increased IL-12 induced STAT-4 signaling in CD8 T cells. from aged mice
 Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression.
 Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Relevant Disclosures
 6/18/215 Therapeutic developments for autoimmune demyelinating diseases: Musings from a MD (Mouse Doctor) Michael K. Racke, M.D. May 28, 215 Relevant Disclosures Editorial Boards for Journal of Neuroimmunology,
6/18/215 Therapeutic developments for autoimmune demyelinating diseases: Musings from a MD (Mouse Doctor) Michael K. Racke, M.D. May 28, 215 Relevant Disclosures Editorial Boards for Journal of Neuroimmunology,
Supplementary Figure 1. Using DNA barcode-labeled MHC multimers to generate TCR fingerprints
 Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
Tie2cre-induced inactivation of the mirna-processing enzyme Dicer disrupts invariant NKT cell development
 Tie2cre-induced inactivation of the mirna-processing enzyme Dicer disrupts invariant NKT cell development Li Zhou a,b,c,1, Kook-Heon Seo a,b,1, Hong-Zhi He a,b, Rafal Pacholczyk d,e, Dong-Mei Meng d,f,
Tie2cre-induced inactivation of the mirna-processing enzyme Dicer disrupts invariant NKT cell development Li Zhou a,b,c,1, Kook-Heon Seo a,b,1, Hong-Zhi He a,b, Rafal Pacholczyk d,e, Dong-Mei Meng d,f,
Multiple nonreceptor protein tyrosine kinases (PTKs)
 Brief Definitive Report Itk and Fyn Make Independent Contributions to T Cell Activation By X. Charlene Liao,* Dan R. Littman, and Arthur Weiss* From the *Department of Microbiology and Immunology, and
Brief Definitive Report Itk and Fyn Make Independent Contributions to T Cell Activation By X. Charlene Liao,* Dan R. Littman, and Arthur Weiss* From the *Department of Microbiology and Immunology, and
Normal GC initiation then collapse; normal mutation and 10. Constitutive signalling leads to spontaneous GC in PP, even BCR -/- 19
 S1 Genetic modifications affecting germinal-centre formation and function Gene Compartment GC Phenotype Ref LTα, LTβ, TNF, Organ Disorganized lymphoid architecture in spleens; poor 1 TNFRI, LTβR architecture
S1 Genetic modifications affecting germinal-centre formation and function Gene Compartment GC Phenotype Ref LTα, LTβ, TNF, Organ Disorganized lymphoid architecture in spleens; poor 1 TNFRI, LTβR architecture
Nature Immunology: doi: /ni Supplementary Figure 1. Id2 and Id3 define polyclonal T H 1 and T FH cell subsets.
 Supplementary Figure 1 Id2 and Id3 define polyclonal T H 1 and T FH cell subsets. Id2 YFP/+ (a) or Id3 GFP/+ (b) mice were analyzed 7 days after LCMV infection. T H 1 (SLAM + CXCR5 or CXCR5 PD-1 ), T FH
Supplementary Figure 1 Id2 and Id3 define polyclonal T H 1 and T FH cell subsets. Id2 YFP/+ (a) or Id3 GFP/+ (b) mice were analyzed 7 days after LCMV infection. T H 1 (SLAM + CXCR5 or CXCR5 PD-1 ), T FH
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
T cell and Cell-mediated immunity
 T cell and Cell-mediated immunity ( 第十章 第十二章第十二章 ) Lu Linrong ( 鲁林荣 ) PhD Laboratory of Immune Regulation Institute of Immunology Zhejiang University, School of Medicine Medical Research Building B815-819
T cell and Cell-mediated immunity ( 第十章 第十二章第十二章 ) Lu Linrong ( 鲁林荣 ) PhD Laboratory of Immune Regulation Institute of Immunology Zhejiang University, School of Medicine Medical Research Building B815-819
Immune tolerance and the prevention of autoimmune diseases
 The Journal of Immunology Deficiency of the Src Homology Region 2 Domain-Containing Phosphatase 1 (SHP-1) Causes Enrichment of CD4 CD25 Regulatory T Cells 1 Jennifer D. Carter,* Gina M. Calabrese,* Makoto
The Journal of Immunology Deficiency of the Src Homology Region 2 Domain-Containing Phosphatase 1 (SHP-1) Causes Enrichment of CD4 CD25 Regulatory T Cells 1 Jennifer D. Carter,* Gina M. Calabrese,* Makoto
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
THE. Cutting Edge: Activation by Innate Cytokines or Microbial Antigens Can Cause Arrest of Natural Killer T Cell Patrolling of Liver Sinusoids 1
 THE JOURNAL OF IMMUNOLOGY CUTTING EDGE Cutting Edge: Activation by Innate Cytokines or Microbial Antigens Can Cause Arrest of Natural Killer T Cell Patrolling of Liver Sinusoids 1 Peter Velázquez,* Thomas
THE JOURNAL OF IMMUNOLOGY CUTTING EDGE Cutting Edge: Activation by Innate Cytokines or Microbial Antigens Can Cause Arrest of Natural Killer T Cell Patrolling of Liver Sinusoids 1 Peter Velázquez,* Thomas
Akt and mtor pathways differentially regulate the development of natural and inducible. T H 17 cells
 Akt and mtor pathways differentially regulate the development of natural and inducible T H 17 cells Jiyeon S Kim, Tammarah Sklarz, Lauren Banks, Mercy Gohil, Adam T Waickman, Nicolas Skuli, Bryan L Krock,
Akt and mtor pathways differentially regulate the development of natural and inducible T H 17 cells Jiyeon S Kim, Tammarah Sklarz, Lauren Banks, Mercy Gohil, Adam T Waickman, Nicolas Skuli, Bryan L Krock,
The Transcription Factor PLZF Directs the Effector Program of the NKT Cell Lineage
 Article The Transcription Factor PLZF Directs the Effector Program of the NKT Cell Lineage Adam K. Savage, 1 Michael G. Constantinides, 1 Jin Han, 1 Damien Picard, 1 Emmanuel Martin, 2 Bofeng Li, 3 Olivier
Article The Transcription Factor PLZF Directs the Effector Program of the NKT Cell Lineage Adam K. Savage, 1 Michael G. Constantinides, 1 Jin Han, 1 Damien Picard, 1 Emmanuel Martin, 2 Bofeng Li, 3 Olivier
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
In Vivo Significance of ITK SLP-76 Interaction in Cytokine Production
 MOLECULAR AND CELLULAR BIOLOGY, July, p. 3596 3609 Vol. 30, No. 14 0270-7306/10/$12.00 doi:10.1128/mcb.01657-09 Copyright, American Society for Microbiology. All Rights Reserved. In Vivo Significance of
MOLECULAR AND CELLULAR BIOLOGY, July, p. 3596 3609 Vol. 30, No. 14 0270-7306/10/$12.00 doi:10.1128/mcb.01657-09 Copyright, American Society for Microbiology. All Rights Reserved. In Vivo Significance of
Peli1 negatively regulates T-cell activation and prevents autoimmunity
 Peli1 negatively regulates T-cell activation and prevents autoimmunity Mikyoung Chang 1,*, Wei Jin 1,5,*, Jae-Hoon Chang 1, Yi-chuan Xiao 1, George Brittain 1, Jiayi Yu 1, Xiaofei Zhou 1, Yi-Hong Wang
Peli1 negatively regulates T-cell activation and prevents autoimmunity Mikyoung Chang 1,*, Wei Jin 1,5,*, Jae-Hoon Chang 1, Yi-chuan Xiao 1, George Brittain 1, Jiayi Yu 1, Xiaofei Zhou 1, Yi-Hong Wang
Rapamycin reduced inhibition of V 1 T cells towards V 4 T cells through down-regulating IL-4
 Rapamycin reduced inhibition of V 1 T cells towards V 4 T cells through down-regulating IL-4 Wanjun Huang Biomedical Translational Research Institute, Jinan University, Guangzhou 510632, China. Abstract
Rapamycin reduced inhibition of V 1 T cells towards V 4 T cells through down-regulating IL-4 Wanjun Huang Biomedical Translational Research Institute, Jinan University, Guangzhou 510632, China. Abstract
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Development of B and T lymphocytes
 Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
Examples of questions for Cellular Immunology/Cellular Biology and Immunology
 Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Immunity and Cancer. Doriana Fruci. Lab di Immuno-Oncologia
 Immunity and Cancer Doriana Fruci Lab di Immuno-Oncologia Immune System is a network of cells, tissues and organs that work together to defend the body against attacks of foreign invaders (pathogens, cancer
Immunity and Cancer Doriana Fruci Lab di Immuno-Oncologia Immune System is a network of cells, tissues and organs that work together to defend the body against attacks of foreign invaders (pathogens, cancer
regulation of polarized membrane transport by rab GTPases Peter van der Sluijs;
 regulation of polarized membrane transport by rab GTPases Peter van der Sluijs; p.vandersluijs@umcutrecht.nl cytotoxic T cell recognizes complex of viral peptide with MHC class I and kills infected cell
regulation of polarized membrane transport by rab GTPases Peter van der Sluijs; p.vandersluijs@umcutrecht.nl cytotoxic T cell recognizes complex of viral peptide with MHC class I and kills infected cell
The Pennsylvania State University. The Graduate School. College of Agricultural Sciences
 The Pennsylvania State University The Graduate School College of Agricultural Sciences ANALYSIS OF T AND NKT CELL ACTIVATION AND FUNCTION IN RESPONSE TO SEB IN VIVO A Thesis in Pathobiology by Melanie
The Pennsylvania State University The Graduate School College of Agricultural Sciences ANALYSIS OF T AND NKT CELL ACTIVATION AND FUNCTION IN RESPONSE TO SEB IN VIVO A Thesis in Pathobiology by Melanie
Supplementary Figures. T Cell Factor-1 initiates T helper 2 fate by inducing GATA-3 and repressing Interferon-γ
 Supplementary Figures T Cell Factor-1 initiates T helper 2 fate by inducing GATA-3 and repressing Interferon-γ Qing Yu, Archna Sharma, Sun Young Oh, Hyung-Geun Moon, M. Zulfiquer Hossain, Theresa M. Salay,
Supplementary Figures T Cell Factor-1 initiates T helper 2 fate by inducing GATA-3 and repressing Interferon-γ Qing Yu, Archna Sharma, Sun Young Oh, Hyung-Geun Moon, M. Zulfiquer Hossain, Theresa M. Salay,
Signaling Through Immune System Receptors (Ch. 7)
 Signaling Through Immune System Receptors (Ch. 7) 1. General principles of signal transduction and propagation. 2. Antigen receptor signaling and lymphocyte activation. 3. Other receptors and signaling
Signaling Through Immune System Receptors (Ch. 7) 1. General principles of signal transduction and propagation. 2. Antigen receptor signaling and lymphocyte activation. 3. Other receptors and signaling
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
T cell and Cell-mediated immunity
 T cell and Cell-mediated immunity Lu Linrong ( 鲁林荣 ) PhD Laboratory of Immune Regulation Institute of Immunology Zhejiang University, it School of Medicine i Medical Research Building B815-819 Email: Lu.Linrong@gmail.com
T cell and Cell-mediated immunity Lu Linrong ( 鲁林荣 ) PhD Laboratory of Immune Regulation Institute of Immunology Zhejiang University, it School of Medicine i Medical Research Building B815-819 Email: Lu.Linrong@gmail.com
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs
 Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
Therapeutic effect of baicalin on experimental autoimmune encephalomyelitis. is mediated by SOCS3 regulatory pathway
 Therapeutic effect of baicalin on experimental autoimmune encephalomyelitis is mediated by SOCS3 regulatory pathway Yuan Zhang 1,2, Xing Li 1,2, Bogoljub Ciric 1, Cun-gen Ma 3, Bruno Gran 4, Abdolmohamad
Therapeutic effect of baicalin on experimental autoimmune encephalomyelitis is mediated by SOCS3 regulatory pathway Yuan Zhang 1,2, Xing Li 1,2, Bogoljub Ciric 1, Cun-gen Ma 3, Bruno Gran 4, Abdolmohamad
CYTOKINE RECEPTORS AND SIGNAL TRANSDUCTION
 CYTOKINE RECEPTORS AND SIGNAL TRANSDUCTION What is Cytokine? Secreted popypeptide (protein) involved in cell-to-cell signaling. Acts in paracrine or autocrine fashion through specific cellular receptors.
CYTOKINE RECEPTORS AND SIGNAL TRANSDUCTION What is Cytokine? Secreted popypeptide (protein) involved in cell-to-cell signaling. Acts in paracrine or autocrine fashion through specific cellular receptors.
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Central tolerance. Mechanisms of Immune Tolerance. Regulation of the T cell response
 Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. Mechanisms of Immune Tolerance ACTIVATION (immunity) SUPPRESSION (tolerance)
Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. Mechanisms of Immune Tolerance ACTIVATION (immunity) SUPPRESSION (tolerance)
Mechanisms of Immune Tolerance
 Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. ACTIVATION (immunity) SUPPRESSION (tolerance) Autoimmunity Immunodeficiency
Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. ACTIVATION (immunity) SUPPRESSION (tolerance) Autoimmunity Immunodeficiency
Supplementary Information:
 Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Newly Recognized Components of the Innate Immune System
 Newly Recognized Components of the Innate Immune System NOD Proteins: Intracellular Peptidoglycan Sensors NOD-1 NOD-2 Nod Protein LRR; Ligand Recognition CARD RICK I-κB p50 p65 NF-κB Polymorphisms in Nod-2
Newly Recognized Components of the Innate Immune System NOD Proteins: Intracellular Peptidoglycan Sensors NOD-1 NOD-2 Nod Protein LRR; Ligand Recognition CARD RICK I-κB p50 p65 NF-κB Polymorphisms in Nod-2
Tolerance, autoimmunity and the pathogenesis of immunemediated inflammatory diseases. Abul K. Abbas UCSF
 Tolerance, autoimmunity and the pathogenesis of immunemediated inflammatory diseases Abul K. Abbas UCSF Balancing lymphocyte activation and control Activation Effector T cells Tolerance Regulatory T cells
Tolerance, autoimmunity and the pathogenesis of immunemediated inflammatory diseases Abul K. Abbas UCSF Balancing lymphocyte activation and control Activation Effector T cells Tolerance Regulatory T cells
SUPPLEMENTARY INFORMATION. Supp. Fig. 1. Autoimmunity. Tolerance APC APC. T cell. T cell. doi: /nature06253 ICOS ICOS TCR CD28 TCR CD28
 Supp. Fig. 1 a APC b APC ICOS ICOS TCR CD28 mir P TCR CD28 P T cell Tolerance Roquin WT SG Icos mrna T cell Autoimmunity Roquin M199R SG Icos mrna www.nature.com/nature 1 Supp. Fig. 2 CD4 + CD44 low CD4
Supp. Fig. 1 a APC b APC ICOS ICOS TCR CD28 mir P TCR CD28 P T cell Tolerance Roquin WT SG Icos mrna T cell Autoimmunity Roquin M199R SG Icos mrna www.nature.com/nature 1 Supp. Fig. 2 CD4 + CD44 low CD4
Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to
 Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
Nature Immunology: doi: /ni.3631
 Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Antigen Presentation and T Lymphocyte Activation. Abul K. Abbas UCSF. FOCiS
 1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF
 CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
Fluorochrome Panel 1 Panel 2 Panel 3 Panel 4 Panel 5 CTLA-4 CTLA-4 CD15 CD3 FITC. Bio) PD-1 (MIH4, BD) ICOS (C398.4A, Biolegend) PD-L1 (MIH1, BD)
 Additional file : Table S. Antibodies used for panel stain to identify peripheral immune cell subsets. Panel : PD- signaling; Panel : CD + T cells, CD + T cells, B cells; Panel : Tregs; Panel :, -T, cdc,
Additional file : Table S. Antibodies used for panel stain to identify peripheral immune cell subsets. Panel : PD- signaling; Panel : CD + T cells, CD + T cells, B cells; Panel : Tregs; Panel :, -T, cdc,
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas
 Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance
 The EMBO Journal Peer Review Process File - EMBO-2012-80891 Manuscript EMBO-2012-80891 Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance Herbert G Kasler,
The EMBO Journal Peer Review Process File - EMBO-2012-80891 Manuscript EMBO-2012-80891 Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance Herbert G Kasler,
Adaptive Immunity. Jeffrey K. Actor, Ph.D. MSB 2.214,
 Adaptive Immunity Jeffrey K. Actor, Ph.D. MSB 2.214, 500-5344 Lecture Objectives: Understand role of various molecules including cytokines, chemokines, costimulatory and adhesion molecules in the development
Adaptive Immunity Jeffrey K. Actor, Ph.D. MSB 2.214, 500-5344 Lecture Objectives: Understand role of various molecules including cytokines, chemokines, costimulatory and adhesion molecules in the development
The Adaptive Immune Responses
 The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA
 AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
Memory CD4 T Cells Enhance Primary CD8 T-Cell Responses
 INFECTION AND IMMUNITY, July 2007, p. 3556 3560 Vol. 75, No. 7 0019-9567/07/$08.00 0 doi:10.1128/iai.00086-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Memory CD4 T Cells
INFECTION AND IMMUNITY, July 2007, p. 3556 3560 Vol. 75, No. 7 0019-9567/07/$08.00 0 doi:10.1128/iai.00086-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Memory CD4 T Cells
Wiskott-Aldrich Syndrome: a cytoskeleton disease
 Wiskott-Aldrich Syndrome: a cytoskeleton disease Elie Haddad, MD, PhD Pediatric Immunology and Rheumatology CHU Ste-Justine Université de Montréal, Montreal, QC, Canada Wiskott-Aldrich Syndrome A disease
Wiskott-Aldrich Syndrome: a cytoskeleton disease Elie Haddad, MD, PhD Pediatric Immunology and Rheumatology CHU Ste-Justine Université de Montréal, Montreal, QC, Canada Wiskott-Aldrich Syndrome A disease
NTD Vaccine Design Toolkit and Training Workshop Providence, RI January 05, 2011 Cytokines Leslie P. Cousens, PhD EpiVax, Inc.
 NTD Vaccine Design Toolkit and Training Workshop Providence, RI January 05, 2011 Cytokines Leslie P. Cousens, PhD EpiVax, Inc. Cytokines Properties of Cytokines Cytokines are proteins with specific roles
NTD Vaccine Design Toolkit and Training Workshop Providence, RI January 05, 2011 Cytokines Leslie P. Cousens, PhD EpiVax, Inc. Cytokines Properties of Cytokines Cytokines are proteins with specific roles
Human and mouse T cell regulation mediated by soluble CD52 interaction with Siglec-10. Esther Bandala-Sanchez, Yuxia Zhang, Simone Reinwald,
 Human and mouse T cell regulation mediated by soluble CD52 interaction with Siglec-1 Esther Bandala-Sanchez, Yuxia Zhang, Simone Reinwald, James A. Dromey, Bo Han Lee, Junyan Qian, Ralph M Böhmer and Leonard
Human and mouse T cell regulation mediated by soluble CD52 interaction with Siglec-1 Esther Bandala-Sanchez, Yuxia Zhang, Simone Reinwald, James A. Dromey, Bo Han Lee, Junyan Qian, Ralph M Böhmer and Leonard
Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was
 Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was sorted by FACS. Surface markers for sorting were CD11c +
Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was sorted by FACS. Surface markers for sorting were CD11c +
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
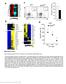 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
5/1/13. The proportion of thymus that produces T cells decreases with age. The cellular organization of the thymus
 T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
The Pennsylvania State University. The Graduate School. Department of Biochemistry, Microbiology and Molecular Biology
 The Pennsylvania State University The Graduate School Department of Biochemistry, Microbiology and Molecular Biology REGULATION OF T HELPER CELL RESPONSES AND ALLERGIC ASTHMA BY TEC KINASES, ITK AND TXK
The Pennsylvania State University The Graduate School Department of Biochemistry, Microbiology and Molecular Biology REGULATION OF T HELPER CELL RESPONSES AND ALLERGIC ASTHMA BY TEC KINASES, ITK AND TXK
An Investigation into the Effects of the Addition of Synthetic Receptor on Chemokine Induced Jurkat T-Cell Migration
 An Investigation into the Effects of the Addition of Synthetic Receptor on Chemokine Induced Jurkat T-Cell Migration Jessica Jurado, Jianfang Hu, Avery August, PhD PSU Undergraduate Animal Bioscience July
An Investigation into the Effects of the Addition of Synthetic Receptor on Chemokine Induced Jurkat T-Cell Migration Jessica Jurado, Jianfang Hu, Avery August, PhD PSU Undergraduate Animal Bioscience July
FOR REVIEW. BMB Reports - Manuscript Submission. Manuscript Draft. Manuscript Number: BMB
 BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
Optimizing Intracellular Flow Cytometry
 Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
The development of T cells in the thymus
 T cells rearrange their receptors in the thymus whereas B cells do so in the bone marrow. The development of T cells in the thymus The lobular/cellular organization of the thymus Immature cells are called
T cells rearrange their receptors in the thymus whereas B cells do so in the bone marrow. The development of T cells in the thymus The lobular/cellular organization of the thymus Immature cells are called
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
Supplementary Figure 1. Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Nature Immunology: doi: /ni.
 Supplementary Figure 1 Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Expression of Mll4 floxed alleles (16-19) in naive CD4 + T cells isolated from lymph nodes and
Supplementary Figure 1 Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Expression of Mll4 floxed alleles (16-19) in naive CD4 + T cells isolated from lymph nodes and
