MHC MULTIMER PROFICIENCY PANEL 2015
|
|
|
- Roderick Williams
- 6 years ago
- Views:
Transcription
1 MHC MULTIMER PROFICIENCY PANEL 2015 July 2015 CONTACT Charlotte Halgreen FOR MORE INFORMATION
2 MHC MULTIMER PROFICIENCY PANEL 2015 This report summarizes the results of the MHC Multimer Proficiency Panel The report provides individual test results for each participating laboratory that participated in the MHC Multimer proficiency panel 2015, as well as an anonymized overview of the other participants test results. 30 laboratories from 13 countries participated in the MHC Multimer Proficiency Panel. Participants included researchers and clinicians active in such diverse fields as Cancer, HIV, CMV, and Diabetes. 30 laboratories from 13 countries participated in the MHC Multimer Proficiency Panel Participants are active in such diverse fields as Cancer, HIV, CMV, and Diabetes Immudex has taken over the MHC Multimer and Elispot proficiency panels from the CIC (Cancer Immunotherapy Consortium of the Cancer Research Institute, USA) and the CIMT (Association for Cancer Immunotherapy, Europe). The proficiency panel services offered by Immudex are open to any laboratory, independent on geographic location or field of interest, with a need to perform accurate and reproducible quantification of antigen-specific T cells. The report is provided using European numeration. The proficiency panels conducted by Immudex are non-profit services offered with the intent of testing and ensuring a high level of proficiency and reliability among the researchers and clinicians that perform the immune monitoring assays. It is the hope and expectation that better immune monitoring assays will lead to better and more efficient immunotherapies. Next MHC Multimer proficiency panel to be held in 2016 The next MHC Multimer proficiency panel will be held in Page 2 of 21
3 Contents INFORMATION ON PARTICIPANTS, PROTOCOLS, REAGENTS, CELL SAMPLES... 4 ANALYSES PERFORMED BY THE PARTICIPANTS... 4 PRESENTATION OF DATA... 5 PROFICIENCY TESTING RESULTS... 5 OVERALL PROFICIENCY GENERAL COMMENTS ACKNOWLEDGEMENTS ABOUT IMMUDEX APPENDIX 1: INSTRUCTIONS APPENDIX 2: PBMC REPORTED NUMBERS APPENDIX 3: HBC 525 REPORTED NUMBERS Page 3 of 21
4 INFORMATION ON PARTICIPANTS, PROTOCOLS, REAGENTS, CELL SAMPLES 30 laboratories participated in the proficiency panel. Each participating laboratory was assigned a confidential participant Identification Number (Lab Id), only known by the laboratory itself and Immudex. Each participant received two vials of PBMCs (human peripheral blood mononuclear cells), termed PBMC and HBC 525, respectively. The PBMCs were shipped in liquid nitrogen. A temperature logger was included in the shipment, allowing observation of vial temperature from packaging to delivery. MCH Dextramers were provided upon request by Immudex. All laboratories used MHC Dextramers. The four multimer specificities tested were EBV HLA- A*0201/GLCTLVAML, EBV HLA-A*0201/CLGGLLTMV, EBV HLA- B*0702/RPPIFIRRL and CMV HLA-B*0702/TPRVTGGGAM. A negative control multimer carrying an irrelevant peptide was also included. Each laboratory performed the Multimer assay according to their own preferred operating procedure. Instructions (see Appendix 1) including Harmonization Guidelines, were provided to all participants. Prior to the shipping of the PBMCs to the participants, the PBMCs were pretested by two labs at separate locations, in order to verify the uniformity of the PBMC vials. Thus, the Multimer assay was performed on a total of 6 vials for each PBMC, using the four multimer specificities provided. The observed variability between different PBMC vials of the same cell sample, as regards cell viability and number of antigenspecific T cells, was insignificant. PBMC samples were thoroughly pretested prior to shipping and sample temperature was monitored from packaging to delivery The PBMCs tested included a range of frequencies of antigen-specific T cells, from about 0,06 % multimer + CD8 + cells ( Low responder ) to about 0,80 % multimer + CD8 + cells ( High responder ). ANALYSES PERFORMED BY THE PARTICIPANTS Each participant received detailed instructions for carrying out the proficiency test; see Instructions (Appendix 1). The participants were asked to report back the following experimental data for PBMC and HBC 525: Number of CD8 + cells counted Page 4 of 21
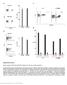
5 Number of EBV HLA-A*0201/GLCTLVAML-specific CD8 + cells counted (PBMC 13075) Number of EBV HLA-A*0201/CLGGLLTMV-specific CD8 + cells counted (PBMC 13075) Number of EBV HLA-B*0702/RPPIFIRRL-specific CD8 + cells counted (HBC 525) Number of CMV HLA-B*0702/TPRVTGGGAM-specific CD8 + cells counted (HBC 525) Number of negative control multimer + CD8 + cells counted From the reported number of CD8 + cells and multimer + CD8 + cells for each measurement, the percentage of multimer + CD8 + cells of the total number of CD8 + cells were calculated and reported as the result All measurements should be done in duplicate. PRESENTATION OF DATA The results obtained are shown in Figures 1-4 and Appendices 2-3. The 30 participants data comprised significant outliers for each of the four PBMC/multimer combinations tested. It was therefore decided to use the median, rather than the average, of the reported results (including all outliers) to represent the average result. Thus, in the following the median of the results for each PBMC/multimer combination represents the average value for all the participants for that particular PBMC/multimer combination. It should be emphasized that the average value (the median) is not necessarily the true value, as no golden standard exists for the MHC Multimer assay. Nevertheless, the median of all the participants results for a particular PBMC/multimer combination was used to calculate the Relative Accuracy of a given participant s result (see below). The median, rather than the average, of the reported results represents the average result PROFICIENCY TESTING RESULTS The results obtained by the 30 participants of the MHC Multimer Proficiency Panel 2015 are shown in Figures 1-4 and Appendices 2-3. Each of Figures 1-4 has an upper and a lower panel. The upper panel shows the percentage of antigen-specific cells (duplicate analysis indicated with two red diamonds), and the percentage of negative control multimer+ cells, i.e. background Page 5 of 21
6 staining (duplicate analysis indicated with two black diamonds). The data are presented in order of increasing Lab Id from left to right. The lower panel shows the relative accuracy, defined as percentage of antigen-specific cells determined by a given participant, divided by the median percentage of antigen-specific cells determined by all the participants. Relative accuracies of are considered in the average range and are represented by filled black columns; relative accuracies of or are considered near average and are represented by hatched columns; relative accuracies below 0.50 or above 2,0 are considered far from average and are represented by open columns. The data are presented in order of increasing relative accuracy from left to right. A relative accuracy of 1,0 indicates full agreement with the average result. Page 6 of 21
7 Relative accuracy % multimer+ PBMC 13075, EBV epitope 1-specific multimer 0,80 0,70 0,60 0,50 0,40 0,30 0,20 0,10 0,00 Lab Id PBMC / Negative control multimer PBMC / EBV epitope 1-specific multimer Median 2,50 PBMC 13075, EBV epitope 1-specific multimer 2,00 1,50 1,00 0,50 0,00 Lab Id Relative accuracy 0,66-1,5 Relative accuracy 0,50-0,65 & 1,6-2,0 Relative accuracy below 0,50 & above 2,0 Figure 1. EBV epitope 1-specific cells of PBMC Percentage of EBV epitope 1- specific CD8 + cells of total CD8 + cells, determined by the 30 participants. Upper panel: Duplicate measurements of the percentage of EBV epitope 1-specific CD8 + cells (red diamonds) and the percentage of negative control multimer + CD8 + cells (black diamonds).the median value (0,16 %) for EBV epitope 1-specific CD8 + cells is indicated with a black horizontal line. Lower panel: Relative accuracy for the measurement of EBV epitope 1-specific CD8 + cells. The relative accuracy for Lab Id 312, 303 and 325 are 0, 3,2 and 5,0, respectively. Page 7 of 21
8 Relative accuracy % multimer+ PBMC 13075, EBV epitope 2-specific multimer 0,40 0,35 0,30 0,25 0,20 0,15 0,10 0,05 0,00 Lab Id PBMC / Negative control multimer PBMC / EBV epitope 2-specific multimer Median 2,50 PBMC 13075, EBV epitope 2-specific multimer 2,00 1,50 1,00 0,50 0,00 Lab Id Relative accuracy 0,66-1,5 Relative accuracy 0,50-0,65 & 1,6-2,0 Relative accuracy below 0,50 & above 2,0 Figure 2. EBV epitope 2-specific cells of PBMC Percentage of EBV epitope 2- specific CD8 + cells of total CD8 + cells, determined by the 30 participants. Upper panel: Duplicate measurements of the percentage of EBV epitope 2-specific CD8 + cells (red diamonds) and the percentage of negative control multimer + CD8 + cells (black diamonds). Lab Id 325 only reported one measurement with the result 1,92 % for the EBV epitope 2-specific CD8 + cells.the median value (0,06 %) for EBV epitope 2-specific CD8 + cells is indicated with a black horizontal line. Lower panel: Relative accuracy for the measurement of EBV epitope 2-specific CD8 + cells. The relative accuracy for Lab Id 312, 333, 303 and 325 are 0, 0, 6,0 and 35, respectively. Page 8 of 21
9 Relative accuracy % multimer+ HBC 525, EBV epitope 3-specific multimer 1,00 0,90 0,80 0,70 0,60 0,50 0,40 0,30 0,20 0,10 0,00 Lab Id HBC 525 / Negative control multimer HBC 525 / EBV epitope 3-specific multimer Median 2,50 HBC 525, EBV epitope 3-specific multimer 2,00 1,50 1,00 0,50 0,00 Lab Id Relative accuracy 0,66-1,5 Relative accuracy 0,50-0,65 & 1,6-2,0 Relative accuracy below 0,50 & above 2,0 Figure 3. EBV epitope 3-specific cells of HBC 525. Percentage of EBV epitope 3-specific CD8 + cells of total CD8 + cells, determined by the 30 participants. Upper panel: Duplicate measurements of the percentage of EBV epitope 3-specific CD8 + cells (red diamonds) and the percentage of negative control multimer + CD8 + cells (black diamonds). The median value (0,35 %) for EBV epitope 3-specific CD8 + cells is indicated with a black horizontal line. Lower panel: Relative accuracy for the measurement of EBV epitope 3-specific CD8 + cells. The relative accuracy for Lab Id 312 and 303 are 0 and 2,9, respectively. Page 9 of 21
10 Relative accuracy % multimer+ HBC 525, CMV-specific multimer 1,40 1,20 1,00 0,80 0,60 0,40 0,20 0,00 Lab Id HBC 525 / Negative control multimer HBC 525 / CMV-specific multimer Median 2,50 HBC 525, CMV-specific multimer 2,00 1,50 1,00 0,50 0,00 Lab Id Relative accuracy 0,66-1,5 Relative accuracy 0,50-0,65 & 1,6-2,0 Relative accuracy below 0,50 & above 2,0 Figure 1. CMV-specific cells of HBC 525. Percentage of CMV-specific CD8 + cells of total CD8 + cells, determined by the 30 participants. Upper panel: Duplicate measurements of the percentage of CMV-specific CD8 + cells (red diamonds) and the percentage of negative control multimer + CD8 + cells (black diamonds). The median value (0,80 %) for CMV-specific CD8 + cells is indicated with a black horizontal line. Lower panel: Relative accuracy for the measurement of CMV-specific CD8 + cells. The relative accuracy for Lab Id 312 is 0. Page 10 of 21
11 OVERALL PROFICIENCY In order to describe the Overall Proficiency of each participating laboratory in enumerating the multimer + CD8 + cells, a score was assigned to each laboratory for each of the 4 measurements performed. The score 3 was assigned to results in the average range (i.e. Relative Accuracy between 0,66 and 1,5), the score 2 was assigned to results near average (i.e. Relative Accuracy 0,50-0,65 or 1,6-2,0), and finally, the score 1 was assigned to results far from average (i.e. Relative Accuracy below 0,50 or above 2,0). Overall Proficiency is defined as the average score obtained over the four measurements. Thus, a laboratory with an overall proficiency of 3 is in the average range on all four measurements and has the highest possible score, and a laboratory with an average score of 1 is far from average on all four measurements and has the lowest possible score. Overall Proficiency is shown in Figure 5. As can be seen, 10 out of 30 laboratories (33 %) are in the average range on all four measurements, and thus have the highest possible Overall Proficiency score of % of participants had results in the average range for 4 out of 4 measurements. Figure 5. Overall Proficiency. The laboratories proficiency in performing the Multimer measurements is shown. An Overall Proficiency of 3 represents the highest possible proficiency score; an Overall Proficiency of 1 represents the lowest possible Overall proficiency score. A score of 3 indicates that this laboratory was in average on all four measurements. A score of 1 indicates that this laboratory was far from average on all four measurements. Page 11 of 21
12 GENERAL COMMENTS The proficiency panel series was initiated by the CIC and CIMT in One of the goals of past proficiency panels was to harmonize procedures across laboratories. Harmonization is now considered finalized, and the responsibility of conducting the proficiency panel was handed over to Immudex in The current MHC Multimer Proficiency Panel is therefore not a harmonization panel, but rather a proficiency testing service. Consequently, harmonization and standardization is not addressed in this report. A survey was carried out in connection with the proficiency panel execution. The full set of reported data and information will be published separately. General observations and conclusions. The MHC Multimer assays performed in this proficiency panel included a range of frequencies of antigen-specific T cells from about 0,06 % multimer + CD8 + cells ( Low responder ) to about 0,80 % multimer + CD8 + cells ( High responder ). For each of the four measurements, about 60 % of the participants had a Relative Accuracy of between 0,66 and 1,5. In other words, for a given measurement, about 60 % of the participants had results in the average range. For each measurement performed, ~60 % of the participants obtained a value close to average 33 % of the participants were in the average range for four out of four measurements. For a given measurement, the 75 % of the participants that were closest to the average (median value) had results differing from 2 to 3 fold. For example, for the PBMC / EBV epitope 1 multimer combination, the 75 % of participants (i.e. 23 out of 30 participants) that were closest to the median value, detected from 0,10 % to 0,23 % multimer + CD8 + cells, and thus differed 2,3 fold. 33 % of the participants were close to average for 4 out of 4 measurements Among the 75 % of participants closest to the average, the lowest and higest percentage of multimer + CD8 + cells detected differed 2-3 fold Page 12 of 21
13 ACKNOWLEDGEMENTS We thank Tina Jakobsen/Dako for performing the pretesting of cell samples, Steffen Walter for providing PBMCs and Sine Reker and Marij Schoenmaekers- Welters for providing helpful advice. Page 13 of 21
14 ABOUT IMMUDEX Based in Copenhagen, Denmark, with North American operations based in Fairfax, Virginia, Immudex provides MHC Dextramer products for the monitoring of antigenspecific T cells, as well as provides MHC multimer and Elispot immune monitoring proficiency panel services. Immudex recognizes the need for accuracy and reproducibility in scientific and clinical research, and patient care, and has a number of research- and clinical products on the market. The goal is to enable simple, accurate and reliable monitoring of antigen-specific cellular immunity, and to promote the routine use of these technologies in diagnostics and research, and in all steps of immunotherapeutics development. Page 14 of 21
15 APPENDIX 1: INSTRUCTIONS FOR THE General Introduction to the MHC Multimer panel All participants will receive two pre-tested donor samples. All participants must determine the percentage of CMV- and EBV-specific T cells for both donor samples using predefined MHC Multimer specificities. Analyses are done by flow cytometry. PLEASE READ ALL THE BELOW INSTRUCTIONS CAREFULLY BEFORE THAWING AND STAINING THE CELLS. If you have any questions, please contact the organizer Charlotte Halgreen Coordinator of Proficiency Panels ProficiencyPanel@immudex.com P: Materials and Reagents: Each participant receives 2 vials each comprising a donor sample, and named HBC-525 and PBMC 13075, respectively. HBC-525 contains 1ml, 20 x 10 6 PBMCs; PBMC contains 1ml, 10 x 10 6 PBMCs. Please store samples in liquid nitrogen upon arrival. MHC Multimer specificities needed for analysis: EBV HLA-A*0201/GLCTLVAML MHC Multimer EBV HLA-A*0201/CLGGLLTMV MHC Multimer EBV HLA-B*0702/RPPIFIRRL Multimer CMV HLA-B*0702/TPRVTGGGAM Multimer Negative Control MHC Multimer Participants who requested MHC Dextramers will receive the following 5 PE-labeled Dextramers: WB2130-PE HLA-A*0201/GLCTLVAML MHC Dextramer 6 tests WB2144-PE HLA-A*0201/CLGGLLTMV MHC Dextramer 6 tests WH2166-PE HLA-B*0702/RPPIFIRRL MHC Dextramer 6 tests WH2136-PE HLA-B*0702/TPRVTGGGAM MHC Dextramer 6 tests NI3233-PE General / Neg. Control MHC Dextramer 10 tests Dextramers should be stored in the dark at 2-8 C until use. Page 15 of 21
16 Overview of Required Staining reactions: Each participant has to perform a total of 12 staining reactions, corresponding to 6 staining reactions on each of the 2 supplied donor samples (HBC-525 and PBMC-13075). The 2 supplied donor samples should be analyzed as follows: PBMC Negative control; staining with Negative control MHC Multimer. Measurement of EBV-specific CD8+ T cells using EBV (HLA-A*0201/GLCTLVAML) MHC Multimer. Measurement of EBV-specific CD8+ T cells using EBV (HLA-A*0201/CLGGLLTMV) MHC Multimer. HBC-525 Negative control; staining with Negative control MHC Multimer. Measurement of EBV-specific CD8+ T cells using EBV (HLA-B*0702/RPPIFIRRL) MHC Multimer. Measurement of CMV-specific CD8+ T cells using CMV (HLA-B*0702/TPRVTGGGAM) MHC Multimer. All analyses are made in duplicates and should in addition to MHC Multimers include anti-cd8 antibody and relevant antibody marker(s) useful for exclusion or inclusion of specific cell population (e.g. anti-cd4 antibody, anti-cd3 antibody, or DEAD cell dyes) during data analysis. Below is a table overview of the required staining reactions. Indicated staining ID s must be used for naming of fcs files and for reporting results of the proficiency panel. Staining ID Donor MHC Multimer sample R Neg PBMC Negative control R Neg PBMC Negative control R EBV1 PBMC EBV (A0201/GLCTLVAML) R EBV1 PBMC EBV (A0201/GLCTLVAML) R EBV2 PBMC EBV (A0201/CLGGLLTMV) R EBV2 PBMC EBV (A0201/CLGGLLTMV) R7-525-Neg HBC-525 Negative control R8-525-Neg HBC-525 Negative control R9-525-EBV3 HBC-525 EBV (B0702/RPPIFIRRL) R EBV3 HBC-525 EBV (B0702/RPPIFIRRL) R CMV HBC-525 CMV (B0702/TPRVTGGGAM) R CMV HBC-525 CMV (B0702/TPRVTGGGAM) Page 16 of 21
17 Instructions for cell preparation and staining: 1) Thawing and Counting Thaw both vials of donor sample. Count and record total cell number after thawing and the number of viable cells for each vial. 2) Staining Please see Appendix A: Multimer Harmonization Guidelines to Optimize Assay Performance Use your own SOP (protocol) for staining and gating of MHC multimer-specific CD8+ T cells. In addition to the four virus-specific MHC, and the negative control Multimer reagents (indicated in the table overview above), the SOP must also include: anti-cd8 antibody staining Optionally, additional marker(s) for exclusion or inclusion of specific cell population(s) (e.g. anti-cd4 antibody, anti-cd3 antibody, or dead cell dyes) can be included. You are free to choose buffers, tubes, staining volume, incubation time, and use of dead cell markers in the assay. Record staining and washing conditions. You will have to perform the six staining reactions per donor outlined above, preferably with a minimum of CD8+ T cells. In order to achieve this minimum, it is recommended that you stain at least 1,5 x 10 6 viable cells per staining. Note: If using MHC-Dextramer reagents, please read the Staining Protocol that came with the Dextramers. In particular, staining and incubation with Dextramers prior to addition of antibodies (anti-cd8, etc.) is essential for optimal staining of the antigen-specific T cells. 3) Data acquisition and analysis All fcs files (flow files) must be named exactly as described in the table on page 2. Your analysis must end with a dot plot showing the CD8-staining on the x-axis and the MHC Multimer staining on the y-axis, as exemplified below. Page 17 of 21
18 Dot plot showing CD8-staining and MHC Multimer staining example: Results are recorded as: Number of CD8-positive T cells (number of events in gate R6, in the above example). Number of MHC multimer-positive T cells (number of events in gate R5 in the above example). Calculate the percentage of MHC multimer-positive T cells of total CD8-positive T cells (R5/R6x100 % in above example). Result must be recorded with two decimals. Reporting of data 1. Fill-in the PowerPoint Dot plot slide (sent by to all participants) with your own dot plot and gating strategy. 2. Create a Zip file, name it with your Lab ID, and include the following files: a. The filled-in PowerPoint Dot plot. b. The 12 fcs files, labeled as described in the table page 2 c. If acquired, include you single color compensation, too. 3. Proficiency data reporting a. Go to» Proficiency panel data registration i. If prompt to, select your region b. Upload you data Zip file, as described c. Report Proficiency Panel Data (use link to» Data report form) i. Fill-in the survey as described. Links and documents relating to Proficiency Panels can be found on Page 18 of 21
19 Appendix A Assay harmonization guidelines Multimer Harmonization Guidelines to Optimize Assay Performance A. Establish lab SOP for MHC peptide multimer staining: A1. Count at least 100,000 CD8 T cells per staining. A2. Establish adequate measures to quantify non-specific binding of Multimer to CD8- positive cells (e.g. irrelevant Multimer or autofluorescence). A3. Establish adequate measures to reduce the amount of non-specific binding of Multimer in the CD8-positive population to allow accurate quantification (e.g. DUMP channel or DEAD cell dyes). B. Establish SOP for software analyses of stained samples, including: B1. Gating strategy. B2. Rules to set the gates. C. Establish a human auditing process of all final results: C1. Are all dot plots correctly compensated? C2. Have the gates been set correctly? C3. Are the reported frequencies of multimer-positive cells plausible? D. Lab environment D1 Only let experienced personnel (per lab SOP) conduct assay. Page 19 of 21
20 APPENDIX 2: PBMC REPORTED NUMBERS PBMC 13075, Negative control multimer (Neg) PBMC 13075, EBV epitope 1-specific multimer (EBV1) PBMC 13075, EBV epitope 2-specific multimer (EBV2) % multimer + CD8 + of CD8 + cells and relative accuracy from data reported Lab Id R Neg % R Neg % R EBV1 % R EBV1 % EBV1 Relative accuracy R EBV2 % R EBV2 % EBV2 Relative accuracy 301 0,01 0,01 0,14 0,10 0,77 0,13 0,04 1, ,34 0,39 0,64 0,36 3,2 0,33 0,33 6, ,01 0,02 0,12 0,14 0,84 0,06 0,04 0, ,00 0,00 0,15 0,16 1,0 0,04 0,05 0, ,00 0,00 0,16 0,18 1,1 0,06 0,05 1, ,00 0,01 0,08 0,08 0,52 0,02 0,03 0, ,00 0,00 0,13 0,11 0,77 0,05 0,05 0, ,00 0,00 0,15 0,16 1,0 0,03 0,03 0, ,00 0,00 0,00 0,00 0 0,00 0, ,00 0,00 0,18 0,17 1,1 0,03 0,05 0, ,00 0,00 0,11 0,10 0,68 0,03 0,02 0, ,00 0,00 0,24 0,22 1,5 0,07 0,05 1, ,01 0,00 0,05 0,05 0,32 0,02 0,02 0, ,00 0,01 0,26 0,26 1,7 0,06 0,07 1, ,01 0,00 0,15 0,17 1,0 0,07 0,10 1, ,02 0,02 0,17 0,19 1,2 0,07 0,06 1, ,00 0,00 0,13 0,11 0,77 0,03 0,02 0, ,01 0,01 0,15 0,19 1,1 0,07 0,06 1, ,01 0,01 0,20 0,20 1,3 0,05 0,06 1, ,08 0,06 0,80 0,74 5,0 ANR 1, ,00 0,00 0,12 0,11 0,74 0,03 0,03 0, ,00 0,01 0,16 0,16 1,0 0,04 0,07 1, ,02 0,01 0,26 0,25 1,6 0,10 0,11 1, ,02 0,02 0,21 0,20 1,3 0,07 0,06 1, ,00 0,01 0,11 0,09 0,65 0,08 0,09 1, ,01 0,01 0,13 0,13 0,84 0,05 0,06 1, ,00 0,00 0,10 0,10 0,65 0,00 0, ,02 0,02 0,15 0,21 1,2 0,08 0,06 1, ,00 0,00 0,14 0,14 0,90 0,04 0,04 0, ,00 0,00 0,10 0,09 0,61 0,03 0,04 0,64 Page 20 of 21
21 APPENDIX 3: HBC 525 REPORTED NUMBERS HBC 525, Negative control multimer (Neg) HBC 525, EBV epitope 3-specific multimer (EBV3) HBC 525, CMV-specific multimer (CMV) % multimer + CD8 + of CD8 + cells and relative accuracy from data reported Lab Id R Neg % R Neg % R EBV3 % R EBV3 % 525- EBV3 Relative accuracy R CMV % R CMV % 525- CMV Relative accuracy 301 0,03 0,00 0,37 0,39 1,1 0,53 0,69 0, ,16 0,18 0,99 1,00 2,9 1,35 1,38 1, ,02 0,02 0,35 0,36 1,0 0,80 0,55 0, ,01 0,01 0,19 0,18 0,53 0,43 0,43 0, ,00 0,00 0,39 0,38 1,1 1,00 1,08 1, ,00 0,00 0,26 0,29 0,79 0,66 0,59 0, ,01 0,01 0,76 0,51 1,8 1,36 1,37 1, ,00 0,00 0,33 0,32 0,94 1,04 1,04 1, ,00 0,00 0,00 0,00 0 0,00 0, ,00 0,00 0,31 0,28 0,85 0,84 0,84 1, ,00 0,00 0,39 0,35 1,1 0,75 0,73 0, ,00 0,01 0,41 0,43 1,2 0,47 0,43 0, ,00 0,00 0,11 0,14 0,36 0,25 0,27 0, ,00 0,00 0,45 0,50 1,4 0,36 0,47 0, ,02 0,00 0,41 0,40 1,2 0,95 1,09 1, ,01 0,01 0,32 0,33 0,94 0,50 0,49 0, ,01 0,01 0,26 0,26 0,75 0,84 0,83 1, ,03 0,03 0,64 0,66 1,9 1,00 1,11 1, ,01 0,01 0,37 0,39 1,1 0,93 0,91 1, ,08 0,06 0,43 0,42 1,2 0,96 1,23 1, ,00 0,00 0,33 0,32 0,94 0,77 0,74 0, ,01 0,01 0,34 0,35 0,99 0,94 0,89 1, ,03 0,03 0,59 0,65 1,8 1,26 1,13 1, ,01 0,01 0,43 0,43 1,2 1,02 1,08 1, ,01 0,01 0,18 0,20 0,55 0,76 0,85 1, ,01 0,02 0,12 0,11 0,33 0,07 0,08 0, ,00 0,00 0,40 0,30 1,0 0,80 0,80 1, ,10 0,11 0,37 0,24 0,88 0,83 0,54 0, ,00 0,00 0,31 0,25 0,81 0,84 0,83 1, ,00 0,00 0,12 0,15 0,39 0,20 0,17 0,23 Page 21 of 21
MHC MULTIMER PROFICIENCY PANEL 2017
 MHC MULTIMER PROFICIENCY PANEL 2017 August 2017 CONTACT Charlotte Halgreen ProficiencyPanel@immudex.com FOR MORE INFORMATION www.proficiencypanel.com MHC MULTIMER PROFICIENCY PANEL 2017 This report summarizes
MHC MULTIMER PROFICIENCY PANEL 2017 August 2017 CONTACT Charlotte Halgreen ProficiencyPanel@immudex.com FOR MORE INFORMATION www.proficiencypanel.com MHC MULTIMER PROFICIENCY PANEL 2017 This report summarizes
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Binding capacity of DNA-barcoded MHC multimers and recovery of antigen specificity
 Supplementary Figure 1 Binding capacity of DNA-barcoded MHC multimers and recovery of antigen specificity (a, b) Fluorescent-based determination of the binding capacity of DNA-barcoded MHC multimers (+barcode)
Supplementary Figure 1 Binding capacity of DNA-barcoded MHC multimers and recovery of antigen specificity (a, b) Fluorescent-based determination of the binding capacity of DNA-barcoded MHC multimers (+barcode)
staining and flow cytometry
 Detection of influenza virus-specific T cell responses by intracellular by cytokine intracellular staining cytokine staining and flow cytometry Detection of influenza virus-specific T cell responses and
Detection of influenza virus-specific T cell responses by intracellular by cytokine intracellular staining cytokine staining and flow cytometry Detection of influenza virus-specific T cell responses and
Challenges in Development and Validation of an Intracellular Cytokine Staining assay
 Challenges in Development and Validation of an Intracellular Cytokine Staining assay Jenny Hendriks, Crucell Hatching @ EBF, Brussels, June 202 www.crucell.com Vaccines vs Protein therapeutics Protein
Challenges in Development and Validation of an Intracellular Cytokine Staining assay Jenny Hendriks, Crucell Hatching @ EBF, Brussels, June 202 www.crucell.com Vaccines vs Protein therapeutics Protein
Supplementary Figure 1. Using DNA barcode-labeled MHC multimers to generate TCR fingerprints
 Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to
 Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
7-AAD/CFSE Cell-Mediated Cytotoxicity Assay Kit
 7-AAD/CFSE Cell-Mediated Cytotoxicity Assay Kit Item No. 600120 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS
7-AAD/CFSE Cell-Mediated Cytotoxicity Assay Kit Item No. 600120 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS
HD1 (FLU) HD2 (EBV) HD2 (FLU)
 ramer staining + anti-pe beads ramer staining a HD1 (FLU) HD2 (EBV) HD2 (FLU).73.11.56.46.24 1.12 b CD127 + c CD127 + d CD127 - e CD127 - PD1 - PD1 + PD1 + PD1-1 1 1 1 %CD127 + PD1-8 6 4 2 + anti-pe %CD127
ramer staining + anti-pe beads ramer staining a HD1 (FLU) HD2 (EBV) HD2 (FLU).73.11.56.46.24 1.12 b CD127 + c CD127 + d CD127 - e CD127 - PD1 - PD1 + PD1 + PD1-1 1 1 1 %CD127 + PD1-8 6 4 2 + anti-pe %CD127
Rapid antigen-specific T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
Pro5 MHC Pentamers. Dr. Jeremy Fry. Copyright ProImmune Limited All Rights Reserved
 Pro5 MHC Pentamers Dr. Jeremy Fry Pro5 MHC Pentamers The most consistent technology for detecting antigenspecific T cells The most-cited commercially available MHC Multimer Used by most-leading academic
Pro5 MHC Pentamers Dr. Jeremy Fry Pro5 MHC Pentamers The most consistent technology for detecting antigenspecific T cells The most-cited commercially available MHC Multimer Used by most-leading academic
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4
 Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Supplementary Figure 1
 Supplementary Figure 1 FMO control (no HLA-DR antibody) FMO control (no HLA-DR antibody) Supplementary Figure 1. Gating strategy for HLA-DR+ T-cells. A gate was drawn around lymphocytes based on cell size
Supplementary Figure 1 FMO control (no HLA-DR antibody) FMO control (no HLA-DR antibody) Supplementary Figure 1. Gating strategy for HLA-DR+ T-cells. A gate was drawn around lymphocytes based on cell size
VQA Control SOP Version 4.0 Roche Amplicor HIV-1 DNA Test, v August 2007
 1. PRINCIPLE 1.1. The Virology Quality Assurance (VQA) Laboratory provides external cell pellet controls for use in the validation of assays that detect HIV proviral DNA. 1.2. HIV seronegative peripheral
1. PRINCIPLE 1.1. The Virology Quality Assurance (VQA) Laboratory provides external cell pellet controls for use in the validation of assays that detect HIV proviral DNA. 1.2. HIV seronegative peripheral
BD CBA on the BD Accuri C6: Bringing Multiplexed Cytokine Detection to the Benchtop
 BD CBA on the BD Accuri C6: Bringing Multiplexed Cytokine Detection to the Benchtop Maria Dinkelmann, PhD Senior Marketing Applications Specialist BD Biosciences, Ann Arbor, MI 23-14380-00 Cellular Communication
BD CBA on the BD Accuri C6: Bringing Multiplexed Cytokine Detection to the Benchtop Maria Dinkelmann, PhD Senior Marketing Applications Specialist BD Biosciences, Ann Arbor, MI 23-14380-00 Cellular Communication
Supplemental Figures Supplemental Figure 1:
 Supplemental Figures Supplemental Figure 1: Representative FACS data showing Concurrent Brain cell type Acquisition using either Percoll PLUS (top row) or myelin removal beads (bottom two rows). Debris
Supplemental Figures Supplemental Figure 1: Representative FACS data showing Concurrent Brain cell type Acquisition using either Percoll PLUS (top row) or myelin removal beads (bottom two rows). Debris
Polymer Technology Systems, Inc. CardioChek PA Comparison Study
 Polymer Technology Systems, Inc. CardioChek PA Comparison Study Evaluation Protocol: Accuracy Precision Clinical Correlation PTS Panels Lipid Panel Test Strips For Use in Comparisons to a Reference Laboratory
Polymer Technology Systems, Inc. CardioChek PA Comparison Study Evaluation Protocol: Accuracy Precision Clinical Correlation PTS Panels Lipid Panel Test Strips For Use in Comparisons to a Reference Laboratory
In vitro human regulatory T cell expansion
 - 1 - Human CD4 + CD25 + regulatory T cell isolation, Workflow in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation of T
- 1 - Human CD4 + CD25 + regulatory T cell isolation, Workflow in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation of T
Annexin V APC Assay Kit
 Annexin V APC Assay Kit Item No. 601410 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
Annexin V APC Assay Kit Item No. 601410 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
Sponsored and reviewed by ICCS Quality and Standards Committee Title: Verification of PNH assay sensitivity through spiking experiment Written by:
 Sponsored and reviewed by ICCS Quality and Standards Committee Title: Verification of PNH assay sensitivity through spiking experiment Written by: A Illingworth, T Oldaker, R Sutherland, S Kotanchiyev,
Sponsored and reviewed by ICCS Quality and Standards Committee Title: Verification of PNH assay sensitivity through spiking experiment Written by: A Illingworth, T Oldaker, R Sutherland, S Kotanchiyev,
LDL Uptake Flow Cytometry Assay Kit
 LDL Uptake Flow Cytometry Assay Kit Item No. 601470 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
LDL Uptake Flow Cytometry Assay Kit Item No. 601470 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
CD25-PE (BD Biosciences) and labeled with anti-pe-microbeads (Miltenyi Biotec) for depletion of CD25 +
 Supplements Supplemental Materials and Methods Depletion of CD25 + T-cells from PBMC. Fresh or HD precultured PBMC were stained with the conjugate CD25-PE (BD Biosciences) and labeled with anti-pe-microbeads
Supplements Supplemental Materials and Methods Depletion of CD25 + T-cells from PBMC. Fresh or HD precultured PBMC were stained with the conjugate CD25-PE (BD Biosciences) and labeled with anti-pe-microbeads
Supplementary Figure 1. Enhanced detection of CTLA-4 on the surface of HIV-specific
 SUPPLEMENTARY FIGURE LEGEND Supplementary Figure 1. Enhanced detection of CTLA-4 on the surface of HIV-specific CD4 + T cells correlates with intracellular CTLA-4 levels. (a) Comparative CTLA-4 levels
SUPPLEMENTARY FIGURE LEGEND Supplementary Figure 1. Enhanced detection of CTLA-4 on the surface of HIV-specific CD4 + T cells correlates with intracellular CTLA-4 levels. (a) Comparative CTLA-4 levels
Jovona Powelson, B.S. MLT (ASCP) Director of Laboratories
 Jovona Powelson, B.S. MLT (ASCP) Director of Laboratories Blood Products From the Donor to You Objectives Deliver a brief virtual tour of a blood center. Describe the number of donors needed to meet the
Jovona Powelson, B.S. MLT (ASCP) Director of Laboratories Blood Products From the Donor to You Objectives Deliver a brief virtual tour of a blood center. Describe the number of donors needed to meet the
UNISEC Universal Influenza Vaccines Secured
 UNISEC Universal Influenza Vaccines Secured Partners in the UNISEC Consortium UNISEC has been made possible by contributions from the European Commission DG-Research and the European member states Project
UNISEC Universal Influenza Vaccines Secured Partners in the UNISEC Consortium UNISEC has been made possible by contributions from the European Commission DG-Research and the European member states Project
Chapter 10. Histocompatibility Testing
 Chapter 10 Histocompatibility Testing Chapter 10 Histocompatibility Testing Table of Contents 10.1 General... 3 10.1.1 Registration of renal transplant patients...3 10.1.2 Material for histocompatibility
Chapter 10 Histocompatibility Testing Chapter 10 Histocompatibility Testing Table of Contents 10.1 General... 3 10.1.1 Registration of renal transplant patients...3 10.1.2 Material for histocompatibility
Module 3: Overview of HIV Testing Technologies
 Module 3: Overview of HIV Testing Technologies Purpose Pre-requisite Modules Module Time Learning Objectives To provide the participants with a basic knowledge of HIV testing and how HIV rapid test results
Module 3: Overview of HIV Testing Technologies Purpose Pre-requisite Modules Module Time Learning Objectives To provide the participants with a basic knowledge of HIV testing and how HIV rapid test results
In vitro human regulatory T cell expansion
 - 1 - Human CD4 + CD25 + CD127 dim/- regulatory T cell Workflow isolation, in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation
- 1 - Human CD4 + CD25 + CD127 dim/- regulatory T cell Workflow isolation, in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation
Why are validated immunogenicity assays important for HIV vaccine development?
 Why are validated immunogenicity assays important for HIV vaccine development? There is a need to compare immunogenicity of products in the pipeline, when similar or different in class when developed by
Why are validated immunogenicity assays important for HIV vaccine development? There is a need to compare immunogenicity of products in the pipeline, when similar or different in class when developed by
IFN-γ Secretion Assay Detection Kit (PE) human
 Miltenyi Biotec GmbH Friedrich-Ebert-Straße 68 51429 Bergisch Gladbach, Germany Phone +49 2204 8306-0 Fax +49 2204 85197 macs@miltenyibiotec.de Miltenyi Biotec Inc. 12740 Earhart Avenue Auburn CA 95602,
Miltenyi Biotec GmbH Friedrich-Ebert-Straße 68 51429 Bergisch Gladbach, Germany Phone +49 2204 8306-0 Fax +49 2204 85197 macs@miltenyibiotec.de Miltenyi Biotec Inc. 12740 Earhart Avenue Auburn CA 95602,
Titrations in Cytobank
 The Premier Platform for Single Cell Analysis (1) Titrations in Cytobank To analyze data from a titration in Cytobank, the first step is to upload your own FCS files or clone an experiment you already
The Premier Platform for Single Cell Analysis (1) Titrations in Cytobank To analyze data from a titration in Cytobank, the first step is to upload your own FCS files or clone an experiment you already
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
In vitro human regulatory T cell suppression assay
 Human CD4 + CD25 + regulatory T cell isolation, in vitro suppression assay and analysis In vitro human regulatory T cell suppression assay Introduction Regulatory T (Treg) cells are a subpopulation of
Human CD4 + CD25 + regulatory T cell isolation, in vitro suppression assay and analysis In vitro human regulatory T cell suppression assay Introduction Regulatory T (Treg) cells are a subpopulation of
Annexin V-FITC Apoptosis Detection Kit with SYTOX
 ab14086 Annexin V-FITC Apoptosis Detection Kit with SYTOX Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in living cells. This product is for research use only and
ab14086 Annexin V-FITC Apoptosis Detection Kit with SYTOX Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in living cells. This product is for research use only and
PROFICIENCY TESTING 2016
 Veterinary and Agrochemical Research Centre Groeselenberg 99 B 1180 Brussels (Ukkel) Tel: +32 (0)2 379 04 11 Fax : + 32 (0)2 379 06 70 http: // www.coda-cerva.be PROFICIENCY TESTING 2016 Bovine Viral Diarrhea
Veterinary and Agrochemical Research Centre Groeselenberg 99 B 1180 Brussels (Ukkel) Tel: +32 (0)2 379 04 11 Fax : + 32 (0)2 379 06 70 http: // www.coda-cerva.be PROFICIENCY TESTING 2016 Bovine Viral Diarrhea
your bioassay is in good hands: transfer from a CRO perspective MIKE MERGES March 4, 2013
 your bioassay is in good hands: transfer from a CRO perspective MIKE MERGES March 4, 2013 Contact Information Michael Merges, M.S. Director Analytical Development Solutions Development & Clinical Services
your bioassay is in good hands: transfer from a CRO perspective MIKE MERGES March 4, 2013 Contact Information Michael Merges, M.S. Director Analytical Development Solutions Development & Clinical Services
SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World
 MIT OpenCourseWare http://ocw.mit.edu SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World Spring 2009 For information about citing these materials or our
MIT OpenCourseWare http://ocw.mit.edu SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World Spring 2009 For information about citing these materials or our
Cytotoxicity assays. Rory D. de Vries, PhD 1. Viroscience lab, Erasmus MC, Rotterdam, the Netherlands
 Cytotoxicity assays Rory D. de Vries, PhD 1 1 Viroscience lab, Erasmus MC, Rotterdam, the Netherlands Anti-influenza immunity Humoral / CD4+ / CD8+ / NK? Function of CTL Elimination of virus-infected cells?
Cytotoxicity assays Rory D. de Vries, PhD 1 1 Viroscience lab, Erasmus MC, Rotterdam, the Netherlands Anti-influenza immunity Humoral / CD4+ / CD8+ / NK? Function of CTL Elimination of virus-infected cells?
altona RealStar Instructions for Use RealStar CMV PCR Kit /2017 EN DIAGNOSTICS
 altona DIAGNOSTICS Instructions for Use RealStar CMV PCR Kit 1.2 08/2017 EN RealStar RealStar CMV PCR Kit 1.2 For research use only! (RUO) 021202 INS-021200-EN-S01 48 08 2017 altona Diagnostics GmbH Mörkenstr.
altona DIAGNOSTICS Instructions for Use RealStar CMV PCR Kit 1.2 08/2017 EN RealStar RealStar CMV PCR Kit 1.2 For research use only! (RUO) 021202 INS-021200-EN-S01 48 08 2017 altona Diagnostics GmbH Mörkenstr.
VQA HIV DNA Control SOP Version 5.0 HIV DNA Testing 13 March 2012
 1. PRINCIPLE 1.1. The Virology Quality Assurance (VQA) Laboratory provides external cell pellet controls for use in the validation of assays that detect HIV proviral DNA. 1.2. HIV seronegative peripheral
1. PRINCIPLE 1.1. The Virology Quality Assurance (VQA) Laboratory provides external cell pellet controls for use in the validation of assays that detect HIV proviral DNA. 1.2. HIV seronegative peripheral
a Beckman Coulter Life Sciences: White Paper
 a Beckman Coulter Life Sciences: White Paper An 8-color DuraClone IM panel for detection of Human blood dendritic cells by flow cytometry Nathalie Dupas 1, Snehita Sattiraju 2, Neha Girish 2, Murthy Pendyala
a Beckman Coulter Life Sciences: White Paper An 8-color DuraClone IM panel for detection of Human blood dendritic cells by flow cytometry Nathalie Dupas 1, Snehita Sattiraju 2, Neha Girish 2, Murthy Pendyala
attomol HLA-B*27-Realtime LT 2 Assay for the detection of the human HLA-B*27-locus using LightCycler (Do not use for tissue typing!
 attomol HLA-B*27-Realtime LT 2 Assay for the detection of the human HLA-B*27-locus using LightCycler (Do not use for tissue typing!) For in vitro diagnostic use only! 50 determinations Order number: 95
attomol HLA-B*27-Realtime LT 2 Assay for the detection of the human HLA-B*27-locus using LightCycler (Do not use for tissue typing!) For in vitro diagnostic use only! 50 determinations Order number: 95
Lipid Droplets Fluorescence Assay Kit
 Lipid Droplets Fluorescence Assay Kit Item No. 500001 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
Lipid Droplets Fluorescence Assay Kit Item No. 500001 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
READ HIGHLIGHTED CHANGES
 BIO-FLASH anti-hbs 3000-8581 100 tests The BIO-FLASH anti-hbs is a fully automated chemiluminescent simultaneous immunoassay for quantitative measurement of antibodies to Hepatitis B surface antigen (anti-hbs)
BIO-FLASH anti-hbs 3000-8581 100 tests The BIO-FLASH anti-hbs is a fully automated chemiluminescent simultaneous immunoassay for quantitative measurement of antibodies to Hepatitis B surface antigen (anti-hbs)
Product information: Human Apolipoprotein B kit 10,000 tests 1. ASSAY DESCRIPTION AND INTENDED USE 2. PROTOCOL AT GLANCE
 Headquarters & Europe Office Cisbio Bioassays Phone: +33 ()4 66 79 67 5 Fax: +33 ()4 66 79 19 2 bioassays@cisbio.com USA Office Cisbio US, Inc. Phone: +1 888 963 4567 Fax: +1 781 687 15 htrfinfo@cisbio.us
Headquarters & Europe Office Cisbio Bioassays Phone: +33 ()4 66 79 67 5 Fax: +33 ()4 66 79 19 2 bioassays@cisbio.com USA Office Cisbio US, Inc. Phone: +1 888 963 4567 Fax: +1 781 687 15 htrfinfo@cisbio.us
RayBio Human Granzyme B ELISA Kit
 RayBio Human Granzyme B ELISA Kit Catalog #: ELH-GZMB User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross,
RayBio Human Granzyme B ELISA Kit Catalog #: ELH-GZMB User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross,
Annexin V-Cy3 Apoptosis Detection Reagent
 ab14143 Annexin V-Cy3 Apoptosis Detection Reagent Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples This product is for research use only and is not
ab14143 Annexin V-Cy3 Apoptosis Detection Reagent Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples This product is for research use only and is not
BD OneFlow LST Application Guide
 BD OneFlow LST Application Guide IVD 23-16165-00 7/2014 EC REP Becton, Dickinson and Company BD Biosciences 2350 Qume Drive San Jose, CA 95131 USA Benex Limited Pottery Road, Dun Laoghaire, Co. Dublin,
BD OneFlow LST Application Guide IVD 23-16165-00 7/2014 EC REP Becton, Dickinson and Company BD Biosciences 2350 Qume Drive San Jose, CA 95131 USA Benex Limited Pottery Road, Dun Laoghaire, Co. Dublin,
Food Proficiency Testing Program Round 36 Whole Milk Powder
 -- REPORT NO. 796 Food Proficiency Testing Program Round 36 Whole Milk Powder December 01 ACKNOWLEDGMENTS PTA wishes to gratefully acknowledge the technical assistance provided for this program by Dr R
-- REPORT NO. 796 Food Proficiency Testing Program Round 36 Whole Milk Powder December 01 ACKNOWLEDGMENTS PTA wishes to gratefully acknowledge the technical assistance provided for this program by Dr R
Human Umbilical Cord Blood CD34 + /133+ Progenitor Cell Care Manual
 Human Umbilical Cord Blood CD34 + /133+ Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.04 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+/133+ Progenitor Cells, cryopreserved Cryopreserved
Human Umbilical Cord Blood CD34 + /133+ Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.04 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+/133+ Progenitor Cells, cryopreserved Cryopreserved
# Required If available Optional MIATA Sub-Modules
 # Required If available Optional MIATA Sub-Modules. Module - Sample Module A - Donor O Essential donor info Captured in patient demographics, Table Module B Source. O Source of cell material Study subjects:
# Required If available Optional MIATA Sub-Modules. Module - Sample Module A - Donor O Essential donor info Captured in patient demographics, Table Module B Source. O Source of cell material Study subjects:
Gladstone Institutes, University of California (UCSF), San Francisco, USA
 Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
MHC Tetramers and Monomers for Immuno-Oncology and Autoimmunity Drug Discovery
 MHC Tetramers and Monomers for Immuno-Oncology and Autoimmunity Drug Discovery Your Partner in Drug Discovery and Research MHC Tetramer Background T-Cell Receptors recognize and bind to complexes composed
MHC Tetramers and Monomers for Immuno-Oncology and Autoimmunity Drug Discovery Your Partner in Drug Discovery and Research MHC Tetramer Background T-Cell Receptors recognize and bind to complexes composed
HTLV Serology Quality Assessment Program Summary for Panel HTLS Oct22
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLS425 2015Oct22
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLS425 2015Oct22
HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
Follicle Dermal Papilla Cells
 Follicle Dermal Papilla Cells Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product
Follicle Dermal Papilla Cells Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
91154 PRIME-XV T Cell CDM Liquid 1 L Additional package sizes are available at request
 PRIME-XV T CELL CDM PRIME-XV T Cell CDM is a ready-to-use chemically-defined, animal component-free medium. It is optimized and designed for the culture of T cells of human origin and recommended for use
PRIME-XV T CELL CDM PRIME-XV T Cell CDM is a ready-to-use chemically-defined, animal component-free medium. It is optimized and designed for the culture of T cells of human origin and recommended for use
Human CD4+T Cell Care Manual
 Human CD4+T Cell Care Manual INSTRUCTION MANUAL ZBM0067.04 SHIPPING CONDITIONS Human CD4+T Cells, cryopreserved Cryopreserved human CD4+T cells are shipped on dry ice and should be stored in liquid nitrogen
Human CD4+T Cell Care Manual INSTRUCTION MANUAL ZBM0067.04 SHIPPING CONDITIONS Human CD4+T Cells, cryopreserved Cryopreserved human CD4+T cells are shipped on dry ice and should be stored in liquid nitrogen
Human TSH ELISA Kit. User Manual
 Human TSH ELISA Kit User Manual Catalog number: GTX15585 GeneTex Table of Contents A. Product Description... 2 B. Kit Components... 3 C. Additional Required Materials (not included)... 3 D. Reagent Preparation...
Human TSH ELISA Kit User Manual Catalog number: GTX15585 GeneTex Table of Contents A. Product Description... 2 B. Kit Components... 3 C. Additional Required Materials (not included)... 3 D. Reagent Preparation...
MHC Multimer staining (6 colours, with fixation)
 v MH Multimer staining (6 colours, with fixation) Authors M. Odendahl,. Blum,. Busch ate 27-04-2007 Version 1.0 REAGENTS: - PBMs after Ficoll - EMA - Phosphate buffered saline (PBS), ph 7.45 - FAS staining
v MH Multimer staining (6 colours, with fixation) Authors M. Odendahl,. Blum,. Busch ate 27-04-2007 Version 1.0 REAGENTS: - PBMs after Ficoll - EMA - Phosphate buffered saline (PBS), ph 7.45 - FAS staining
EpiQuik Circulating Acetyl Histone H3K18 ELISA Kit (Colorimetric)
 EpiQuik Circulating Acetyl Histone H3K18 ELISA Kit (Colorimetric) Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE Uses: The EpiQuik Circulating Acetyl Histone H3K18 ELISA Kit (Colorimetric)
EpiQuik Circulating Acetyl Histone H3K18 ELISA Kit (Colorimetric) Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE Uses: The EpiQuik Circulating Acetyl Histone H3K18 ELISA Kit (Colorimetric)
HIV Viral Load Quality Assessment Program Summary for Panel HIVVL 2018Oct26
 1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
Peptide stimulation and Intracellular Cytokine Staining
 v Peptide stimulation and Intracellular Cytokine Staining Authors A. Cosma, S. Allgayer MATERIALS Date 01-02-2007 REAGENTS: Version 1.0 - PBMCs - Culture medium - Costimulating antibodies - Peptide pools
v Peptide stimulation and Intracellular Cytokine Staining Authors A. Cosma, S. Allgayer MATERIALS Date 01-02-2007 REAGENTS: Version 1.0 - PBMCs - Culture medium - Costimulating antibodies - Peptide pools
Naive, memory and regulatory T lymphocytes populations analysis
 Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
HIV Viral Load Quality Assessment Program Summary for Panel HIVVL 2017Oct27
 1 The National Laboratory for HIV Reference Services is Accredited to ISO 15189 and ISO 17043 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology
1 The National Laboratory for HIV Reference Services is Accredited to ISO 15189 and ISO 17043 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology
Cryopreserved Human Hepatocytes (Cat # HP-F)
 Certificate of Analysis Cryopreserved Human Hepatocytes (Cat # HP-F) This CoA represents a single Lot specific batch of cells. For details of current Lots available please contact us. Lot#: H0434 Gender
Certificate of Analysis Cryopreserved Human Hepatocytes (Cat # HP-F) This CoA represents a single Lot specific batch of cells. For details of current Lots available please contact us. Lot#: H0434 Gender
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Influenza A IgG ELISA
 Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Supplementary Figure 1. Example of gating strategy
 Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
Introduction: Table/Figure Descriptions:
 Introduction: We have completed the analysis of your HIV RNA Validation Study. The validation plan was designed to verify the installation of an unmodified FDA-approved HIV RNA assay into your laboratory.
Introduction: We have completed the analysis of your HIV RNA Validation Study. The validation plan was designed to verify the installation of an unmodified FDA-approved HIV RNA assay into your laboratory.
For the rapid, sensitive and accurate measurement of apoptosis in various samples.
 ab14082 500X Annexin V-FITC Apoptosis Detection Reagent Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples. This product is for research use only and
ab14082 500X Annexin V-FITC Apoptosis Detection Reagent Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples. This product is for research use only and
SERION ELISA classic CMV Avidity Reagent B109 AVID
 SERION ELISA classic CMV Avidity Reagent B109 AVID SERION ELISA classic Avidity Reagents are complementary components which, in combination with the corresponding SERION ELISA classic, enables the avidity
SERION ELISA classic CMV Avidity Reagent B109 AVID SERION ELISA classic Avidity Reagents are complementary components which, in combination with the corresponding SERION ELISA classic, enables the avidity
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
Chapter 10. Histocompatibility Testing
 Chapter 10 Histocompatibility Testing Change record Date Author Version Change reference 08-03-2013 J. de Boer 2.0 Textual Adjustments 13-09-2012 C.M. Tieken 1.1 Text added page 2 10-03-2012 I. Doxiadis
Chapter 10 Histocompatibility Testing Change record Date Author Version Change reference 08-03-2013 J. de Boer 2.0 Textual Adjustments 13-09-2012 C.M. Tieken 1.1 Text added page 2 10-03-2012 I. Doxiadis
THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT TO PERFORM THERAPEUTIC DONOR INSEMINATION WITH ANONYMOUS DONOR SPERM
 THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT TO PERFORM THERAPEUTIC DONOR INSEMINATION WITH ANONYMOUS DONOR SPERM Partner #1 Last Name (Surname): Partner #1 First Name: Partner
THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT TO PERFORM THERAPEUTIC DONOR INSEMINATION WITH ANONYMOUS DONOR SPERM Partner #1 Last Name (Surname): Partner #1 First Name: Partner
Follicle Dermal Papilla Cell
 Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer
 AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke,. CONTRACTING ORGANIZATION: Johns Hopkins
AD Award Number: W8-XWH-5-- TITLE: Development of Antigen Presenting Cells for adoptive immunotherapy in prostate cancer PRINCIPAL INVESTIGATOR: Mathias Oelke,. CONTRACTING ORGANIZATION: Johns Hopkins
MATERIALS. Peptide stimulation and Intracellular Cytokine Staining- EFFECTOR 45RA PANEL
 v Peptide stimulation and Intracellular Cytokine Staining- EFFECTOR 45RA PANEL Authors S. Kutscher, A. Cosma, MATERIALS REAGENTS: - PBMCs - Culture medium - Costimulating antibodies - Peptide pools including
v Peptide stimulation and Intracellular Cytokine Staining- EFFECTOR 45RA PANEL Authors S. Kutscher, A. Cosma, MATERIALS REAGENTS: - PBMCs - Culture medium - Costimulating antibodies - Peptide pools including
ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3*
 ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3* 1 Department of Microbiology, Li Ka Shing Faculty of Medicine, University
ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3* 1 Department of Microbiology, Li Ka Shing Faculty of Medicine, University
Supplementary Figure 1
 Supplementary Figure 1 Identification of IFN-γ-producing CD8 + and CD4 + T cells with naive phenotype by alternative gating and sample-processing strategies. a. Contour 5% probability plots show definition
Supplementary Figure 1 Identification of IFN-γ-producing CD8 + and CD4 + T cells with naive phenotype by alternative gating and sample-processing strategies. a. Contour 5% probability plots show definition
GEX Recommended Procedure Eff. Date: 07/27/07 Rev.: C Pg. 1 of 10
 GEX Recommended Procedure Eff. Date: 07/27/07 Rev.: C Pg. 1 of 10 NOTICE: This document is version controlled and was produced as a part of the GEX Information Program which requires that all Series 100
GEX Recommended Procedure Eff. Date: 07/27/07 Rev.: C Pg. 1 of 10 NOTICE: This document is version controlled and was produced as a part of the GEX Information Program which requires that all Series 100
Instructions. Fuse-It-mRNA easy. Shipping and Storage. Overview. Kit Contents. Specifications. Note: Important Guidelines
 Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
A step-by-step approach to build and analyze a multicolor panel
 Analyze A step-by-step approach to build and analyze a multicolor panel For Research Use Only. Not for use in diagnostic or therapeutic procedures. Alexa Fluor is a registered trademark of Life Technologies
Analyze A step-by-step approach to build and analyze a multicolor panel For Research Use Only. Not for use in diagnostic or therapeutic procedures. Alexa Fluor is a registered trademark of Life Technologies
Concentration Estimation from Flow Cytometry Exosome Data Protocol
 Concentration Estimation from Flow Cytometry Exosome Data Protocol 1. STANDARD CURVE Create a standard curve for the target exosome by plotting the mean fluorescence (y axis) against the protein concentration
Concentration Estimation from Flow Cytometry Exosome Data Protocol 1. STANDARD CURVE Create a standard curve for the target exosome by plotting the mean fluorescence (y axis) against the protein concentration
ADVL0411 study. (temozolomide in patients with leukemia)
 ADVL0411 study (temozolomide in patients with leukemia) I. Overview of study: We are interested in determining the level of MGMT activity and MRS mutations in leukemic samples (plasma and lymphoblasts)
ADVL0411 study (temozolomide in patients with leukemia) I. Overview of study: We are interested in determining the level of MGMT activity and MRS mutations in leukemic samples (plasma and lymphoblasts)
For research or further manufacturing use only. Not for injection or diagnostic procedures.
 PRIME-XV T cell Expansion XSFM PRIME-XV T Cell Expansion XSFM is a xeno-free, serum-free medium optimized for the activation and expansion of human T lymphocytes. This medium contains gentamicin and requires
PRIME-XV T cell Expansion XSFM PRIME-XV T Cell Expansion XSFM is a xeno-free, serum-free medium optimized for the activation and expansion of human T lymphocytes. This medium contains gentamicin and requires
Procine sphingomyelin ELISA Kit
 Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
10/18/2012. A primer in HLA: The who, what, how and why. What?
 A primer in HLA: The who, what, how and why What? 1 First recognized in mice during 1930 s and 1940 s. Mouse (murine) experiments with tumors Independent observations were made in humans with leukoagglutinating
A primer in HLA: The who, what, how and why What? 1 First recognized in mice during 1930 s and 1940 s. Mouse (murine) experiments with tumors Independent observations were made in humans with leukoagglutinating
Human HBcAb IgM ELISA kit
 Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Clinical Evaluation for. Embrace No Code Blood Glucose Monitoring System
 Embrace No Code Report Clinical Evaluation for Embrace No Code Blood Glucose Monitoring System Applicant: Apex Biotechnology Corp. Test Date: 2007/8/28~2007/9/21 Clinical Site: SCHMIDT Group Practice Clinic
Embrace No Code Report Clinical Evaluation for Embrace No Code Blood Glucose Monitoring System Applicant: Apex Biotechnology Corp. Test Date: 2007/8/28~2007/9/21 Clinical Site: SCHMIDT Group Practice Clinic
CONTRACTING ORGANIZATION: Johns Hopkins University School of Medicine Baltimore, MD 21205
 AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AviagenBrief. Evaluating Comparative Broiler Performance through Trials INTRODUCTION KEY CONSIDERATIONS. November 2018
 AviagenBrief Evaluating Comparative Broiler Performance through Trials INTRODUCTION November 2018 Carefully planned broiler trials will allow the customer to clearly evaluate the effect of the treatment,
AviagenBrief Evaluating Comparative Broiler Performance through Trials INTRODUCTION November 2018 Carefully planned broiler trials will allow the customer to clearly evaluate the effect of the treatment,
FINAL REPORT HBV Serology External Quality Assessment Scheme
 FINAL REPORT HBV Serology External Quality Assessment Scheme Programme codes: HBVC4310, HBVC435 Panel ID: 2008/Mar/11 HBV Serology EQAS Panel ID 2008/Mar/11 Page 1 of 9 The NRL is a: NATA-accredited proficiency
FINAL REPORT HBV Serology External Quality Assessment Scheme Programme codes: HBVC4310, HBVC435 Panel ID: 2008/Mar/11 HBV Serology EQAS Panel ID 2008/Mar/11 Page 1 of 9 The NRL is a: NATA-accredited proficiency
Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12
 1 Supplementary Data Figure legends Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12 serum levels measured by multiplex ELISA (Luminex) in FL patients before
1 Supplementary Data Figure legends Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12 serum levels measured by multiplex ELISA (Luminex) in FL patients before
RayBio Human ENA-78 ELISA Kit
 RayBio Human ENA-78 ELISA Kit Catalog #: ELH-ENA78 User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross,
RayBio Human ENA-78 ELISA Kit Catalog #: ELH-ENA78 User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross,
Application Information Bulletin: Human NK Cells Phenotypic characterizing of human Natural Killer (NK) cell populations in peripheral blood
 Application Information Bulletin: Human NK Cells Phenotypic characterizing of human Natural Killer (NK) cell populations in peripheral blood Christopher A Fraker, Ph.D., University of Miami - Miami, Florida
Application Information Bulletin: Human NK Cells Phenotypic characterizing of human Natural Killer (NK) cell populations in peripheral blood Christopher A Fraker, Ph.D., University of Miami - Miami, Florida
EXOTESTTM. ELISA assay for exosome capture, quantification and characterization from cell culture supernatants and biological fluids
 DATA SHEET EXOTESTTM ELISA assay for exosome capture, quantification and characterization from cell culture supernatants and biological fluids INTRODUCTION Exosomes are small endosome-derived lipid nanoparticles
DATA SHEET EXOTESTTM ELISA assay for exosome capture, quantification and characterization from cell culture supernatants and biological fluids INTRODUCTION Exosomes are small endosome-derived lipid nanoparticles
Hyaluronan Quantification Kit
 Product manual (ELISA-like assay for HA / Hyaluronic Acid) Updated on Jan. 6th, 207 - Background Hyaluronan (HA) is an unbranched glycosaminoglycan composed of repeating disaccharide units of D-glucronic
Product manual (ELISA-like assay for HA / Hyaluronic Acid) Updated on Jan. 6th, 207 - Background Hyaluronan (HA) is an unbranched glycosaminoglycan composed of repeating disaccharide units of D-glucronic
Human Thyroid-Peroxidase Antibody, TPO-Ab ELISA Kit
 Human Thyroid-Peroxidase Antibody, TPO-Ab ELISA Kit Catalog No: E0442h 96 Tests Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Human Thyroid-Peroxidase Antibody, TPO-Ab ELISA Kit Catalog No: E0442h 96 Tests Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
