Keywords: monocytes, nurse cells, osteoclasts, rheumatoid arthritis, stromal cells
|
|
|
- Lindsay McBride
- 5 years ago
- Views:
Transcription
1 Research article Differentiation of monocytes into multinucleated giant boneresorbing cells: two-step differentiation induced by nurse-like cells and cytokines Tomoko Toyosaki-Maeda*, Hiroshi Takano, Tetsuya Tomita, Yuji Tsuruta*, Miki Maeda-Tanimura*, Yasunori Shimaoka, Tetsu Takahashi, Tsunetoshi Itoh, Ryuji Suzuki* and Takahiro Ochi *Department of Immunology, Shionogi Research Laboratories, Shionogi & Co Ltd, Osaka, Japan Department of Orthopedic Surgery, Osaka University Medical School, Osaka, Japan Department of Oral Surgery, Kyushu Dental College School of Dentistry, Kokura-Kita, Kita-Kyushu, Japan Department of Immuno-Ontogeny, Tohoku University School of Medicine, Aoba-ku, Sendai, Miyagi, Japan Correspondence: Takahiro Ochi, Department of Orthopedic Surgery, Osaka University Medical School, Yamada-Oka 2-2, Suita, Osaka , Japan. Tel: ; fax: ; ochi@ort.med.osaka-u.ac.jp Received: 6 December 2000 Revisions requested: 9 February 2001 Revisions received: 12 June 2001 Accepted: 22 June 2001 Published: 2 August 2001 Arthritis Res 2001, 3: This article may contain supplementary data which can only be found online at Toyosaki-Maeda et al, licensee BioMed Central Ltd (Print ISSN ; Online ISSN ) Abstract Bone resorption in the joints is the characteristic finding in patients with rheumatoid arthritis (RA). Osteoclast-like cells are present in the synovial tissues and invade the bone of patients with RA. The characteristics of these cells are not completely known. In the work reported here, we generated these cells from peripheral-blood monocytes from healthy individuals. The monocytes were co-cultured with nurse-like cells from synovial tissues of patients with RA (RA-NLCs). Within 5 weeks of culture, the monocytes were activated and differentiated into mononuclear cells positive for CD14 and tartrateresistant acid phosphatase (TRAP). These mononuclear cells then differentiated into multinucleated giant bone-resorbing cells after stimulation with IL-3, IL-5, IL-7, and/or granulocyte-macrophagecolony-stimulating factor. TRAP-positive cells with similar characteristics were found in synovial fluid from patients with RA. These results indicate that multinucleated giant bone-resorbing cells are generated from monocytes in two steps: first, RA-NLCs induce monocytes to differentiate into TRAPpositive mononuclear cells, which are then induced by cytokines to differentiate into multinucleated giant bone-resorbing cells. Keywords: monocytes, nurse cells, osteoclasts, rheumatoid arthritis, stromal cells Our laboratory has established that nurse-like cells (NLCs) are present in the synovial tissues and bone marrow of patients with rheumatoid arthritis (RA) [1 3]. Such cells, which were first discovered in thymus, play an important role in thymocyte maturation and differentiation [4 6]. In vitro, they form unique complexes with thymocytes, which initially adhere to them and then crawl beneath them [7 9]. This phenomenon, which is unique to NLCs at various tissue sites, has been called pseudoemperipolesis. NLCs from RA synovial tissue (RA-NLCs) promote survival of B cells [2,3] and maintain the growth of myeloid cells of patients with RA [1], suggesting that they contribute profoundly to pathogenesis in RA. DMEM = Dulbecco s modified Eagle s medium; FCS = fetal calf serum; GM-CSF = granulocyte/macrophage-colony-stimulating factor; HLA = human major histocompatibility antigen; IL = interleukin; NLC = nurse-like cell; RA = rheumatoid arthritis; RANKL = receptor activator of nuclearfactor-κb ligand; RA-NLC = nurse-like cell derived from rheumatoid arthritis synovial tissue; TRAP = tartrate-resistant acid phosphatase.
2 Multinucleated cells in synovial tissues have been reported to invade the bone of patients with RA [10]. The cells expression of tartrate-resistant acid phosphatase (TRAP) and calcitonin receptor suggested that they are osteoclasts [11,12]. Although the presence of osteoclast-like cells in rheumatoid synovium is well understood, the mechanism by which they differentiate is not. In order to examine the effect of RA-NLCs on monocyte functions, we co-cultured peripheral-blood monocytes with RA-NLCs and looked for morphological and functional alterations of CD14- and TRAP-positive cells. We also found such cells in synovial fluid from patients with RA. These cells differentiated into multinucleated giant bone-resorbing cells in the presence of IL-3, IL-5, IL-7, and/or granulocyte/- macrophage-colonystimulating factor (GM-CSF). In this way we defined the process by which bone-resorbing cells are generated from monocytic cells. Materials and methods Isolation of NLCs from RA synovial tissues RA-NLCs were established from RA synovial tissues as previously described [1]. Briefly, synovial tissues were obtained from knee joints of five patients with RA who fulfilled American College of Rheumatology criteria for RA [13], after informed consent had been obtained. The cells were cultured in DMEM (Dulbecco s modified Eagle s medium [DMEM; Gibco BRL, Gaithersburg, MD, USA] supplemented with 10% fetal calf serum [FCS; Hyclone, Logan, UT, USA], 100 units/ml of penicillin [Gibco BRL], and 100 µg/ml of streptomycin [Gibco BRL] at 37 C in 7.5% CO 2. RA-NLCs were identified by their ability to support pseudoemperipolesis, seen in vitro in the migration of a T-cell lymphoma line, MOLT-17, beneath the NLCs, as previously described [3]. Isolation of mononuclear cells from RA synovial fluid Synovial fluid was obtained from patients with RA who fulfilled the American College of Rheumatology criteria for RA [13]. The infiltrating cells were collected from the fluid by centrifugation at 1900 g and were cultured in supplemented DMEM. After 3 to 5 weeks of culture, most of the lymphocytes and granulocytes disappeared and monocyte-like cells became dominant. CD14-positive monocyte-like cells were purified from this population with a magnetic-activated cell sorter (MACS; Myltenyi Biotec GmbH, Germany) using anti-cd14 antibody conjugated to magnetic beads in accordance with the manufacturer s instructions. The purity of CD14-positive cells was analyzed using a fluorescence-activated cell sorter (FACScan ; see Supplementary material). Isolation and culture of monocytes from peripheral blood Peripheral-blood monocytes were collected as plasticadherent cells, as described previously [14]. Mononuclear cells were isolated from heparinized peripheral blood from five healthy volunteers [15]. Over 97% of the adherent cells were determined to be monocytes by morphology and CD14 expression. Monocytes ( ) were co-cultured with RA-NLCs. After 3 to 5 weeks, TRAP-positive mononuclear cells with abundant cytoplasm became dominant. They were collected by gently washing the culture with warm supplemented DMEM and their purity was confirmed cytochemically. Formation of multinucleated giant cells by TRAP-positive mononuclear cells The CD14-positive and TRAP-positive mononuclear cells from the synovial fluid of patients with RA were examined for expression of surface antigen and for phagocytic activity and were stimulated with various cytokines (see Supplementary material). TRAP-positive mononuclear cells ( ) were cultured in supplemented DMEM in the presence or absence of various cytokines or in conditioned medium ([15]; and see Supplementary material) for h. In the presence of receptor activator nuclear factor κb ligand (RANKL), cultures were maintained for 14 days. At the end of the culture period, May Grunwald Giemsa (Wako Pure Chemical Co., Osaka, Japan) and TRAP staining (TRAPstaining kit; Sigma, St Louis, MO, USA) were conducted. The frequency of osteoclasts was evaluated from the fusion index, as previously described [16]. More than 1000 nuclei within TRAP-positive multinucleated cells (>4 nuclei/cell) were counted. The fusion index (%) was calculated according to the formula: total no. of nuclei within multinucleated cells 100 total no. of nuclei counted where multinucleated cells are cells with >4 nuclei. Examination of bone resorption TRAP-positive mononuclear cells ( ) were stimulated with various cytokines on a dentin slice for 7 days. In order to examine resorption areas with a scanning electron microscope, the differentiated cells were washed off the slices with distilled water. Then the slices were dehydrated, air-dried, and sputtered with gold. Results Morphological changes of peripheral-blood monocytes after co-culture with RA-NLCs After peripheral-blood monocytes had been cultured with RA-NLCs for 3 to 4 weeks, we recovered TRAP-positive mononuclear cells (Fig. 1a 1c) with abundant cytoplasm and an off-center nucleus (Fig. 1a and 1b). These cells strongly expressed CD11b, CD11c, CD14, CD45, and human major histocompatibility antigen (HLA)-DR, sug- commentary review reports research article
3 Arthritis Research Vol 3 No 5 Toyosaki-Maeda et al gesting that they were of monocyte lineage (Table 1). However, they did not express CD11a, CD35, or CD68, which are expressed on freshly isolated monocytes (see Supplementary material). Figure 1 Presence of TRAP-positive mononuclear cells in synovial fluid from patients with RA We detected monocytic cells positive for CD14 and TRAP in synovial fluids of patients with RA. These cells also strongly expressed CD11b, CD11c, CD14, CD45, and HLA-DR (see Supplementary meterial) but not CD1a, CD1b, CD2, CD5, or CD86, which are expressed on dendritic cells derived from monocytes [17]. These results indicate that TRAP-positive monocytic cells present in synovial fluid and those induced in vitro in cultures with RA-NLCs are morphplogically and phenotypically the same. These cells were obtained from the synovial fluid of all patients with RA, regardless of age and sex. CD14- positive monocyte-like cells accounted for 20 91% of the mononuclear cells freshly isolated from the synovial fluid of such patients (data not presented). Differentiation of TRAP-positive mononuclear cells into multinucleated giant bone-resorbing cells in the absence of RA-NLCs The TRAP-positive cells induced in vitro and those isolated from synovial fluid both differentiated into multinucleated cells after being cultured for 72 to 96 h with the conditioned medium (Fig. 1d). These multinucleated cells still possessed TRAP activity (data not shown) and formed resorption areas on dentin slices (Fig. 2), suggesting that they had bone-resorbing activity as osteoclasts. Induction of multinucleated cells by IL-3, IL-5, IL-7 or GM-CSF The cytokines IL-3, IL-5, IL-7, and GM-CSF induced differentiation of TRAP-positive cells induced in vitro or those isolated from synovial fluid into osteoclasts (Table 1). Regardless of which cytokine was used to stimulate differentiation of the osteoclasts, they were all positive for TRAP and formed resorption pits on dentin slices, suggesting that they were all identical to the cells induced by conditioned medium (data not shown). The fusion index of osteoclasts induced by a mixture of cytokines was higher than those stimulated with a single cytokine. The cytokines IL-6 and IL-8, which are produced by RA-NLCs [2], did not induce osteoclast formation. RANKL was recently reported to induce osteoclasts from human peripheral blood [18,19]; however, a mixture of macrophage-colonystimulating factor and RANKL exhibited only weak activity for induction of osteoclasts from the TRAP-positive mononuclear cells (Table 1). Phytohemagglutinin, which was contained in the conditioned medium, did not induce differentiation. TRAP-positive cells from the synovial fluid of patients with RA have a fusion index similar to that found for the TRAP-positive cells obtained experimentally Morphology of TRAP-positive mononuclear cells induced from peripheral-blood monocytes with RA-NLCs. (a) Phase-contrast micrograph of monocytes co-cultured with RA-NLCs. Mononuclear cells (arrow) are growing on the RA-NLCs. (b) Mononuclear cells collected from the culture shown in (a). May Grunwald Giemsa staining. (c) Detection of TRAP expressed by the mononuclear cells (TRAP-positive cells were stained red with their cytoplasm). (d) Differentiated mononuclear cells. The cells shown here are multinucleated giant cells. May Grunwald Giemsa staining. Scale lines = 100 µm. from monocytes (data not shown). Induction of the osteoclasts was completely neutralized by the antibody to each cytokine (see Supplementary material). Discussion We have shown that the novel ability of RA-NLCs may contribute to the pathogenesis of RA by encouraging the generation of TRAP-positive mononuclear cells, which are osteoclast precursors. The TRAP-positive precursor cells have phagocytic activity and are negative for CD83, suggesting that they are different from peripheral-blood monocytes and dendritic cells [14,17]. Fujikawa et al reported that synovial macrophages differentiated into osteoclasts after incubation in the presence of a rat osteoblast-like cell line [20]. The fibroblasts isolated from RA synovia induced differentiation of monocytes into multinucleated cells in the presence of 1,25-dihydroxyvitamin D 3 and macrophage-colony-
4 Figure 2 Table 1 Differentiation of human TRAP-positive mononuclear cells derived from peripheral-blood monocytes into multinucleated cells after stimulation with IL-3, IL-5, IL-7, and GM-CSF Scanning electron micrographs of dentin slices, showing (a) resorption areas formed on dentin by TRAP-positive multinucleated giant cells derived from monocytes and stimulated with granulocyte-macrophagecolony-stimulating factor for 96 h and (b) a control slice incubated with the TRAP-positive mononuclear cells in the absence of cytokines. Scale lines = 50 µm. stimulating factor [21]. Further study will be required to determine the identity of those monocytic cells and our cells. Fibroblastic cells in synovial fluid from patients with RA have been reported to support pseudoemperipolesis, which was considered to be the unique feature of the nurse cells, in the presence of IL-4 [22]. Shigeyama et al recently reported that RA synovial fluid may promote osteoclastogenesis from monocytes by expressing osteoclast differentiation factor [23]. It is likely that RA fibroblasts and RA-NLCs share several roles in the pathogenesis of RA, including activation of monocytes. However, the molecules required in our study for osteoclastogenesis from monocytes were different from those in the study of Shigeyama et al [23]. These findings suggest that multiple pathways for osteoclastogenesis in RA synovia may cause severe joint destruction. There may be two steps for generation of the osteoclasts in the joints of patients with RA: first, differentiation of monocytes into TRAP-positive mononuclear cells induced and maintained by RA-NLCs, followed by cytokineinduced differentiation of these mononuclear cells into osteoclasts. The interaction between monocytes and RA- NLCs required adhesion molecules, but RANK (receptor activator of nuclear-factor-κb) and RANKL were not necessary to induce the TRAP-positive cells in preliminary studies in our laboratory (unpublished observation). The molecules required in the interaction are under investigation. In addition, we found the presence of TRAP-positive mononuclear cells which differentiated into osteoclasts in synovial fluids of patients with RA in vitro. Monocytes may infiltrate the affected joints and differentiate into TRAPpositive mononuclear cells under the influence of RA- NLCs. This conclusion is consistent with previous findings Stimulator Concentration Fusion index (%) a None 1.8 Conditioned medium b (10% v/v) 86.1 IL-1α (1 ng/ml) 1.1 IL-1β (1 ng/ml) 1.3 IL-2 (250 U/ml) 7.7 IL-3 (5 ng/ml) 64.8 IL-4 (100 U/ml) 1.3 IL-5 (1 ng/ml) 66.1 IL-6 (20 ng/ml) 5.6 IL-6 + sil-6r (sil-6r: 200 ng/ml) 5.3 IL-7 (20 ng/ml) 72.4 IL-8 (20 ng/ml) 2.1 IFN-γ (100 U/ml) 7.1 GM-CSF (1 ng/ml) 73.8 M-CSF (25 ng/ml) 5.8 TNF-α (1 ng/ml) 6.9 VD 3 (10 7 mol/l) 3.4 Dexamethasone (10 8 mol/l) 0.8 M-CSF + IL-4 (M-CSF, 25 ng/ml; IL-4,100 U/ml) 0.1 IL-3 + IL IL-7 + GM-CSF 77.3 IL-3 + IL-7 + GM-CSF 78.7 Phytohemagglutinin (1% v/v) 2.0 RANKL c (100 ng/ml) 2.6 RANKL + M-CSF c (RANKL/ODF, 100 ng/ml; 38.1 M-CSF, 25 ng/ml) a TRAP-positive mononuclear cells were stimulated with various cytokines for h. Fusion indices were calculated using the formula given in the text and previously [16]. Data are representative of three independent experiments using TRAP-positive cells induced from monocytes. b See [15], and Supplementary material. c The culture was maintained for 14 days. GM-CSF = granulocyte macrophage-colony-stimulating factor; IFN = interferon; IL = interleukin; M-CSF = macrophage-colonystimulating factor; ODF = osteoclast differentiation factor; RANKL = receptor activator of nuclear-factor-κb ligand; sil-6r = soluble interleukin-6 receptor; TNF = tumor necrosis factor; VD 3 = 1,25-dihydroxyvitamin D 3. of TRAP-positive multinucleated giant cells in the synovial tissue of patients with RA [10 12]. Further studies are required to characterize these osteoclasts derived from the TRAP-positive mononuclear cells and to delineate the unique course of differentiation into bone-resorbing cells promoted by RA-NLCs. commentary review reports research article
5 Arthritis Research Vol 3 No 5 Toyosaki-Maeda et al Conclusion In order to elucidate the role of RA-NLCs, monocytes were co-cultured with RA-NLCs. Monocytes differentiated into TRAP-positive mononuclear cells, the precursor cells of osteoclasts. Osteoclasts were generated from TRAPpositive mononuclear cells in the presence of IL-3, IL-5, IL-7, and GM-CSF. TRAP-positive cells were also present in synovial fluids of patients with RA. RA-NLCs may play a significant role in the activation of monocytes and longterm maintenance of differentiated monocytes (osteoclast precursors). The present study suggests that monocytes may differentiate into osteoclast precursor cells in the affected joints of patients with RA. Acknowledgements This work was supported by the Program for Promotion of Fundamental Studies in Health Science of the Organization for Drug ADR Relief, R&D Promotion and Product Review of Japan. The authors are grateful to Dr Ichiro Kurane (Department of Virology 1, National Institute of Infectious Diseases, Tokyo, Japan) for helpful discussions and critical reading of the manuscript. References 1. Tomita T, Takeuchi E, Toyosaki-Maeda T, Oku H, Kaneko M, Takano, Sugamoto H, Ohzono K, Suzuki R, Ochi T: Establishment of nurse-like stromal cells from bone marrow patients with rheumatoid arthritis: indication of characteristic bone marrow microenvironment in patients with rheumatoid arthritis. Rheumatology (Oxford) 1999, 38: Takeuchi E, Tomita T, Toyosaki-Maeda T, Hashimoto H, Kaneko M, Takano H, Sugano K, Suzuki R, Ochi T: Establishment and characterization of nurse-cell like stromal cell lines from synovial tissues of patients with rheumatoid arthritis. Arthritis Rheum 1999, 42: Shimaoka Y, Attrep JF, Hirano T, Ishihara K, Suzuki R, Toyosaki T, Ochi T, Lipsky PE: Nurse-like cells from bone marrow and synovium of patients with rheumatoid arthritis promote survival and enhance function of human B cells. J Clin Invest 1998, 102: Pezzano M, Li Y, Phlip D, Omene C, Cantey M, Saunders G, Guyden JC: Thymic nurse cell rescue of early CD4+ CD8+ thymocytes from apoptosis. Cell Mol Biol 1995, 41: Pezzano M, Phlip D, Stephenson S, Li Y, Reid V, Maitta R, Guyden JC: Positive selection by thymic nurse cells requires IL-1 beta and is associated with an increased Bcl-2 expression. Cell Immunol 1996, 169: Iwabuchi K, Nakayama K, McCoy RL, Wang F, Nishimura T, Habu S, Murphy KM, Loh, DY: Cellular and peptide requirements for in vitro clonal deletion of immature thymocytes. Proc Natl Acad Sci U S A 1992, 89: Itoh T, Doi H, Chin S, Nishimura T, Kasahara S: Establishment of mouse thymic nurse cell clones from a spontaneous BALB/c thymic tumor. Eur J Immunol 1988, 18: Miyake K, Hasunuma Y, Yagita H, Kimoto, M: Requirement for VLA-4 and VLA-5 integrins in lymphoma cells binding to and migration beneath stromal cells in culture. J Cell Biol 1992, 119: Hiai H, Nishi Y, Miyazawa T, Matsudaira Y, Nishizuka Y: Mouse lymophoid leukemias: symbiotic complexes of neoplastic lymphocytes and their microenvironments. J Natl Cancer Inst 1981, 66: Gravallese EM, Harada Y, Wang JT, Gom AH, Thomhil TS, Goldring, SR: Identification of cell types responsible for bone resorption in rheumatoid arthritis and juvenile rheumatoid arthritis. Am J Pathol 1998, 152: Chang JS, Quinn JM, Demaziere A, Bulstrode CJ, Francis MJ, Duthie RB, Athanasou NA: Bone resorption by cells isolated from rheumatoid synovium. Ann Rheum Dis 1992, 51: Fujikawa Y, Shingu M, Torisu T, Itonaga I, Masumi S: Bone resorption by tartrate-resistant acid phosphatase-positive multinuclear cells isolated from rheumatoid synovium. Br J Rheumatol 1996, 35: Arnett FC, Edworthy SM, Bloch DA, Mcshane DJ, Fries JF, Cooper NS, Healey LA, Kaplan SR, Liang MH, Luthra HA, Medsger TA, Jr, Mitchell DM, Neustadt DH, Pinals RS, Schaller JG, Sharp JG, Wilder RL, Hunder GG: The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988, 31: Akagawa KS, Takatsuka N, Nozaki Y, Komuro I, Azuma M, Ueda M, Naito M, Takahashi K: Generation of CD1+RelB+ dendritic cells and tartrate-resistant acid phosphatase-positive osteoclast-like multinucleated giant cells from human monocytes. Blood 1996, 88: Toyosaki T, Tsuruta Y, Yoshioka T, Takemoto H, Suzuki R, Tomita T, Ochi T: Recognition of rheumatoid arthritis synovial antigen by CD4+, CD8 T cell clones established from rheumatoid arthritis joints. Arthritis Rheum 1998, 41: Higuchi S, Tabata N, Tajima M, Ito M, Tsurudome M, Sudo A, Uchida A, Ito Y: Induction of human osteoclast-like cells by treatment of blood monocytes with anti-fusion regulatory protein-1/cd98 monoclonal antibodies. J Bone Miner Res 1998, 13: Zhou, LJ, Tedder TF: CD14+ blood monocytes can differentiate into functionally mature CD83+ dendritic cells. Proc Natl Acad Sci U S A 1996, 93: Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, Tomoyasu A, Yano K, Goto M, Murakami A, Tsuda E, Morinaga T, Higashino K, Udagawa N, Takahashi N, Suda T: Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci U S A 1998, 95: Lacey DL, Timms E, Tan HL, Kelley MJ, Dunstan CR, Burgess T, Elliott R, Colombero A, Elliott G, Scully S, Hsu H, Sullivan J, Hawkins N, Davy E, Capparelli C, Eli A, Qian YX, Kaufman S, Sarosi I, Shalhoub V, Senaldi G, Guo J, Delaney J, Boyle WJ. Ostreoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 1998, 93: Fujikawa Y, Sabokbar A, Neale S, Athanasou NA: Human osteoclast formation and bone resorption by monocytes and synovial macrophages in rheumatoid arthritis. Ann Rheum Dis 1996, 55: Takayanagi H, Oda H, Yamamoto S, Kawaguchi H, Tanaka S, Nishikawa T, Koshihara Y: A new mechanism of bone destruction in rheumatoid arthritis: synovial fibroblasts induce osteoclastogenesis. Biochem Biophys Res Commun 1997, 240: Burger JA, Zvaifler NJ, tsukada N, Firestein GS, Kipps TJ: Fibroblast-like synoviocytes support pseudoemperipolesis via a stromal cell-derived B-cell factor-1-and CD106 (VCAM-1)- dependent mechanism. J Clin Invest 2001, 107: Shigeyama Y, Pap T, Kunzler P, Simmen BR, Gay RE, Gay S: Expression of osteoclast differentiation factor in rheumatoid arthritis. Arthritis Rheum 2000, 43: Supplementary material Supplementary materials and methods Cell lines Human lung fibroblasts CCD-19Lu were obtained from American Type Culture Collection (Rockville, MD, USA). Human T cell line MOLT-17 was a generous gift from Dr J Minowada (Fujisaki Cell Center, Okayama, Japan). These cell lines were cultured as recommended by the providers. Examination of pseudoemperipolesis Pseudoemperipolesis was measured as previously described [3]. RA synovial stromal cells ( ) were incubated in supplemented DMEM in 24-well culture
6 plates overnight. The next day, MOLT-17 cells ( ) were added to the RA-NLC culture. Stromal cells with more than three MOLT-17 cells beneath them after 6 h of incubation were considered NLCs. Long-term maintenance of monocytes by RA-NLCs Monocytes ( ) were co-cultured with RA-NLCs or CCD-19Lu with or without culture inserts (Becton Dickinson, Franklin Lakes, NJ, USA) in supplemented DMEM in 48-well culture plates at 37 C in 7.5% CO 2 for up to 70 days. Half of the medium was changed once a week. The TRAP-positive mononuclear cells were collected from the culture, stained with trypan blue, and counted for viability under a microscope. Antibodies and staining of cells The cell-surface antigens on freshly isolated monocytes and TRAP-positive mononuclear cells were examined by staining with monoclonal antibodies specific for CD1a, CD4, CD5, CD11a, CD11b, CD11c, CD13, CD14, CD15, CD16, CD19, CD20, CD34, CD45, CD45RA, CD45RO, CD54, HLA-DR (Becton Dickinson), CD1b (Nichirei, Tokyo, Japan), CD2, CD3 (Ortho Diagnostics, Raritan, NJ, USA), CD35, CD68 (DAKO Japan, Kyoto, Japan), CD51/61, CD83, HLA-A, B and C (Pharmingen, San Diego, CA, USA), CD80, and CD86 (Ancell, Bayport, MN, USA). Antigen-expression was analyzed with a FACScan flow cytometer (Becton Dickinson). Multinucleated giant bone-resorbing cells differentiated from TRAPpositive mononuclear cells were fixed with cold acetone and stained immunohistochemically with rabbit polyclonal antibodies specific for actin (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA), carbonic anhydrase II (Rockland, Gilbertsville, PA, USA), or vitronectin receptor (Chemicon International, Inc., Temecula, CA, USA). TRAP activity in TRAP-positive mononuclear cells and multinucleated giant bone-resorbing cells was examined using a TRAP-staining kit (Sigma, St Louis, MO, USA). Neutralizing antibodies to human IL-3, IL-5, IL-7, and GM-CSF were purchased from Genzyme (Cambridge, MA, USA). Cytokines and reagents Conditioned media were prepared as previously reported [15]. Briefly, a mixture of peripheral-blood mononuclear cells from 10 healthy donors was stimulated with phytohemagglutinin at 37 C for 72 h. Culture supernatant fluids were collected, filtered, and used as conditioned media. IL-1α was purchased from Immugenex (Los Angeles, CA). IL-1β, IL-2, IL-3, IL-4, IL-5, IL-6, soluble IL-6 receptor (sil- 6R), interferon gamma (IFN-γ), granulocyte/macrophagecolony stimulating factor (GM-CSF), macrophage-colony stimulating factor, and tumor necrosis factor (TNF)-α were purchased from R&D Systems, Inc. (Minneapolis, MN, USA). IL-7 and IL-8 were purchased from Genzyme. 1,25- dihydroxyvitamin D 3 and dexamethasone were purchased from Wako Pure Chemical Co. (Osaka, Japan). Receptor activator of nuclear-factor-κb ligand (RANKL) was obtained from Peprotech (London, UK). Assessment of phagocytic activity Phagocytic activity of TRAP-positive mononuclear cells was assessed from their ingestion of heat-killed yeast. TRAP-positive mononuclear cells ( ) were incubated with yeast cells in phosphate-buffered saline supplemented with 10% fresh human serum, type AB, at 37 C for 45 min. The cells were washed and stained with fuchsin (Wako Pure Chemical Co.), and the cells with ingested yeast were counted under a microscope. Inhibition of the generation of multinucleated giant cells from TRAP-positive mononuclear cells by neutralizing antibodies Neutralizing antibodies specific for IL-3, IL-5, IL-7, and GM- CSF were used for inhibition of the generation of multinucleated giant bone-resorbing cells. Irrelevant polyclonal mouse IgG from Jackson ImmunoResearch (West Grove, PA, USA) was used as a control. The TRAP-positive mononuclear cells ( ) were pre-incubated with each antibody in DMEM containing 10% FCS in microtubes at 37 C for 1 h. The cells were cultured in 4-well chamber slides, and stimulated with a cytokine for h at 37 C in 7.5% CO 2. At the end of the culture period, the cells were stained for TRAP and the fusion index was calculated as described in the main paper. Detection of calcitonin receptors Calcitonin receptors on the multinucleated giant boneresorbing cells were detected in situ using 125 I-human calcitonin were performed as described elsewhere [23]. TRAP-positive mononuclear cells ( ) were stimulated with IL-3, IL-5, IL-7, or GM-CSF at the optimal concentrations in 4-well chamber slides (Nalge Nunc International, Rochester, NY, USA) for h at 37 C in 7.5% CO 2. After formation of multinucleated giant bone-resorbing cells had been confirmed microscopically, the cells were incubated in 0.4 ml αmem (Gibco BRL) with 0.1% bovine serum albumin and 0.2 mci/ml of 125 I- labeled human calcitonin (Amersham Pharmacia Biotech, Buckinghamshire, UK) for 1 h at 22 C. Nonspecific binding was assessed on each slide in the presence of an excess amount of unlabeled human calcitonin. Then the cells were washed three times with phosphate-buffered saline solution and fixed with 2.5% formaldehyde and 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer. The slides were washed and dried as previously described [S1]. Air-dried slides were dipped in photographic emulsion (Kodak NTB3; Eastman Kodak, Rochester, NY, USA), drained, and dried for 2 h and were kept in a light-proof container with desiccant at 4 C for 10 days. The slides were developed in accordance with the manufacturer s instructions. commentary review reports research article
7 Arthritis Research Vol 3 No 5 Toyosaki-Maeda et al Supplementary Table 1 Expression of surface antigen by TRAP-positive mononuclear cells generated by co-culture with RA-NLC % Positive a Monocytes from TRAP-positive cells induced TRAP-positive cells Antigen peripheral blood from monocytes from RA-SF CD1a CD1b CD CD CD CD CD11a LFA CD11b CR3 α chain CD11c CR4 α chain CD CD CD15 Le x CD16 FcgR III CD CD CD CD35 CR CD CD45RA RA45RO CD51/61 VNR b CD54 ICAM CD CD80 B7/BB CD CD86 B70/FUN HLA-A, -B, -C HLA-DR a Cells were stained with monoclonal antibody specific for various antigens, and analyzed by FACScan. Data are representative of four independent analyses. b Vitronectin receptor. RA-SF = synovial fluid from patients with rheumatoid arthritis. Supplementary results Phenotypic characterization of the TRAP-positive mononuclear cells induced by NLCs The TRAP-positive mononuclear cells, whether induced from monocytes or collected from the synovial fluid of patients with RA, strongly expressed CD11b, CD11c, CD14, CD45, HLA-A, HLA-B, HLA-C, and HLA-DR but did not express CD1a, CD1b, CD2, CD4, CD5, CD16, CD19, CD20, or CD83. These observations suggest that these cells belong to the monocyte/macrophage lineage (Supplementary Table 1). However, the cells did not express CD11a, CD35, or CD68, which are expressed on freshly isolated monocytes from peripheral blood (Supplementary Table 2). The TRAP-positive mononuclear cells had strong phagocytic activity against heat-killed yeast (data not presented). In addition, the cells were positive for carbonic anhydrase II, actin, and vitronectin receptor (Supplementary Fig. 1), and calcitonin receptors were also
8 Supplementary Table 2 Inhibition of formation of multinucleated giant cells by neutralizing antibodies specific for IL-3, IL-5, IL-7, and GM-CSF Fusion index (%) a Antibody b IL-3 (5 ng/ml) IL-5 (1 ng/ml) IL-7 (20 ng/ml) GM-CSF (1 ng/ml) Polyclonal mouse IgG 63.8 ± ± ± ± 6.0 Anti-IL ± 1.6 Anti-IL ± 1.8 Anti-IL ± 5.8 Anti-GM-CSF 4.0 ± 1.3 a Fusion indices were calculated using the formula given in the Methods section. Each value is the mean ± SD of three independent experiments using TRAP-positive cells induced from monocytes of three individuals. b Concentrations of cytokines: polyclonal mouse IgG, 10 mg/ml; anti-il-3, 10 mg/ml; anti-il-5, 5 mg/ml; anti-il-7, 10 mg/ml, anti-gm-csf, 2 mg/ml. = not examined; GM-CSF = granulocyte/macrophage-colonystimulating factor; IL = interleukin. Supplementary Figure 1 Immunohistochemical staining of multinucleated giant bone-resorbing cells. The cells were stained green for (a) carbonic anhydrase II and (b) vitronectin receptor. The cells were also positive for (c) actin, which showed red, ring-form staining. (d) In situ detection of calcitonin receptor using 125 I-human calcitonin. Black grains mark the cells expressing calcitonin receptor. The multinucleated giant boneresorbing cells were incubated with 125 I-human calcitonin for 1 h. The cells were washed, fixed, and dried as described in the Supplementary materials and methods section. Air-dried slides were exposed at 4 C for 10 days. Scale lines = 100 µm. Long-term maintenance of monocytes by RA-NLCs Monocytes obtained from all five healthy individuals and co-cultured with RA-NLCs grew and differentiated into TRAP-positive mononuclear cells (Supplementary Fig. 2). We also used five RA-NLC lines, each from a different patient with RA. All five cell lines induced the generation of TRAP-positive mononuclear cells from human peripheral blood monocytes (data not presented). The generation of these TRAP-positive cells required direct contact with RA-NLCs. Induction of osteoclasts by IL-3, IL-5, IL-7, or GM-CSF The conditioned medium contains several cytokines. In order to determine which cytokines induce TRAP-positive mononuclear cells to differentiate into multinucleated giant bone-resorbing cells, in vitro induced TRAP-positive cells and those isolated from synovial fluid of patients with RA were collected and stimulated with various cytokines in the absence of RA-NLCs. In a preliminary study, a highperformance liquid chromatography fraction of the conditioned medium, which was used to induce TRAP-positive mononuclear cells to differentiate into multinucleated giant bone-resorbing cells, contained proteins with a molecular weight of approximately 20 kda (data not shown). Therefore, we mainly examined cytokines with approximately that molecular weight. The activities of IL-3, IL-5, IL-7, and GM-CSF for inducing multinucleated giant bone-resorbing cells were completely neutralized by the antibody to the respective cytokine (Supplementary Table 2). Supplementary reference S1. Nicholson GC, Moseley JM, Sexton PM, Mendelsohn FAO, Martin TJ: Abundant calcitonin receptors in isolated rat osteoclasts. Biochemical and autoradiographic characterization. J Clin Invest 1986, 78: commentary review reports research article detected (Supplementary Fig. 1); these four receptors are considered characteristic of osteoclasts [10,23].
9 Arthritis Research Vol 3 No 5 Toyosaki-Maeda et al Supplementary Figure 2 Effects of culture conditions on survival of monocytes from five donors with RA-NLCs or CCD-19Lu, in the presence (separated) or absence (no indication) of culture inserts. Each culture was maintained for up to 70 days. The resulting mononuclear cells were collected, stained with trypan blue, and counted under a microscope. Each experiment was conducted in duplicate, and each point represents the mean number of viable cells in two cultures.
IRA-International Journal of Applied Sciences ISSN Vol. 03 Issue 02 (May, 2016) Paper DOI:
 IRA-International Journal of Applied Sciences ISSN 2455-4499 Vol. 03 Issue 02 (May, 2016) Paper DOI: https://dx.doi.org/10.21013/jas.v3.n2.p8 Pathophysiology of Giant Cell Formation in Giant Cell Tumor
IRA-International Journal of Applied Sciences ISSN 2455-4499 Vol. 03 Issue 02 (May, 2016) Paper DOI: https://dx.doi.org/10.21013/jas.v3.n2.p8 Pathophysiology of Giant Cell Formation in Giant Cell Tumor
Osteoclast Culture Kit
 K-ASSAY KAMIYA BIOMEDICAL COMPANY Osteoclast Culture Kit For the culture of Osteoclasts from precursor cells. Cat. No.: CC-107 Rat Osteoclast Precursor Cells, V-1 CC-109 Mouse Osteoclast Precursor Cells,
K-ASSAY KAMIYA BIOMEDICAL COMPANY Osteoclast Culture Kit For the culture of Osteoclasts from precursor cells. Cat. No.: CC-107 Rat Osteoclast Precursor Cells, V-1 CC-109 Mouse Osteoclast Precursor Cells,
Effect of Interleukin 10 on the Hematopoietic Progenitor Cells from Patients with Aplastic Anemia
 Effect of Interleukin 10 on the Hematopoietic Progenitor Cells from Patients with Aplastic Anemia YOSHINOBU ASANO, SHOICHIRO SHIBATA, SHINJI KOBAYASHI, SEIICHI OKAMURA, YOSHIYUKI NIHO First Department
Effect of Interleukin 10 on the Hematopoietic Progenitor Cells from Patients with Aplastic Anemia YOSHINOBU ASANO, SHOICHIRO SHIBATA, SHINJI KOBAYASHI, SEIICHI OKAMURA, YOSHIYUKI NIHO First Department
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Osteoclast Culture Kit
 K-ASSAY Osteoclast Culture Kit For the culture of Osteoclasts from precursor cells. Cat. No.: CC-107 Rat Osteoclast Precursor Cells, V-1 For Research Use Only. 1 Rev. 091708 K-ASSAY PRODUCT INFORMATION
K-ASSAY Osteoclast Culture Kit For the culture of Osteoclasts from precursor cells. Cat. No.: CC-107 Rat Osteoclast Precursor Cells, V-1 For Research Use Only. 1 Rev. 091708 K-ASSAY PRODUCT INFORMATION
Differentiation and function of osteoclasts. Takeshi Miyamoto and Toshio Suda
 REVIEW Differentiation and function of osteoclasts Takeshi Miyamoto and Toshio Suda The Sakaguchi Laboratory of Developmental Biology, School of Medicine, Keio University, Tokyo, Japan (Received for publication
REVIEW Differentiation and function of osteoclasts Takeshi Miyamoto and Toshio Suda The Sakaguchi Laboratory of Developmental Biology, School of Medicine, Keio University, Tokyo, Japan (Received for publication
Stimulation of osteoclast formation by inflammatory synovial fluid
 Virchows Arch (2006) 449: 69 77 DOI 10.1007/s00428-006-0200-y ORIGINAL ARTICLE Iannis E. Adamopoulos. Lynett Danks. Ichiro Itonaga. Rachel M. Locklin. Afsie Sabokbar. David J. P. Ferguson. Nicholas A.
Virchows Arch (2006) 449: 69 77 DOI 10.1007/s00428-006-0200-y ORIGINAL ARTICLE Iannis E. Adamopoulos. Lynett Danks. Ichiro Itonaga. Rachel M. Locklin. Afsie Sabokbar. David J. P. Ferguson. Nicholas A.
Increased osteoclastic activity in acute Charcot s osteoarthopathy: the role of receptor activator of nuclear factor-kappab ligand
 Diabetologia (28) 51:135 1 DOI 1.17/s125-8-992-1 ARTICLE Increased osteoclastic activity in acute Charcot s osteoarthopathy: the role of receptor activator of nuclear factor-kappab ligand G. Mabilleau
Diabetologia (28) 51:135 1 DOI 1.17/s125-8-992-1 ARTICLE Increased osteoclastic activity in acute Charcot s osteoarthopathy: the role of receptor activator of nuclear factor-kappab ligand G. Mabilleau
IL-17 in synovial fluids from patients with rheumatoid arthritis is a potent stimulator of osteoclastogenesis
 IL-17 in synovial fluids from patients with rheumatoid arthritis is a potent stimulator of osteoclastogenesis Shigeru Kotake, 1 Nobuyuki Udagawa, 2 Naoyuki Takahashi, 2 Kenichiro Matsuzaki, 2 Kanami Itoh,
IL-17 in synovial fluids from patients with rheumatoid arthritis is a potent stimulator of osteoclastogenesis Shigeru Kotake, 1 Nobuyuki Udagawa, 2 Naoyuki Takahashi, 2 Kenichiro Matsuzaki, 2 Kanami Itoh,
Rheumatoid arthritis ( RA) is a systemic autoimmune lymphocyte growth and development [ 17 21]. In a
![Rheumatoid arthritis ( RA) is a systemic autoimmune lymphocyte growth and development [ 17 21]. In a Rheumatoid arthritis ( RA) is a systemic autoimmune lymphocyte growth and development [ 17 21]. In a](/thumbs/89/97927013.jpg) Rheumatology 1999;38:854 863 Establishment of nurse-like stromal cells from bone marrow of patients with rheumatoid arthritis: indication of characteristic bone marrow microenvironment in patients with
Rheumatology 1999;38:854 863 Establishment of nurse-like stromal cells from bone marrow of patients with rheumatoid arthritis: indication of characteristic bone marrow microenvironment in patients with
Effect of corticosteroids on human osteoclast formation and activity
 See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/11079968 Effect of corticosteroids on human osteoclast formation and activity Article in Journal
See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/11079968 Effect of corticosteroids on human osteoclast formation and activity Article in Journal
Generation of post-germinal centre myeloma plasma B cell.
 Generation of post-germinal centre myeloma. DNA DAMAGE CXCR4 Homing to Lytic lesion activation CD38 CD138 CD56 Phenotypic markers Naive Secondary lymphoid organ Multiple myeloma is a malignancy of s caused
Generation of post-germinal centre myeloma. DNA DAMAGE CXCR4 Homing to Lytic lesion activation CD38 CD138 CD56 Phenotypic markers Naive Secondary lymphoid organ Multiple myeloma is a malignancy of s caused
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MACROPHAGE "MONOCYTES" SURFACE RECEPTORS
 LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
Tartrate resistant acid phosphatase (TRAP) positive cells in rheumatoid synovium may induce the destruction of articular cartilage
 196 EXTENDED REPORT Tartrate resistant acid phosphatase (TRAP) positive cells in rheumatoid synovium may induce the destruction of articular cartilage H Tsuboi, Y Matsui, K Hayashida, S Yamane, M Maeda-Tanimura,
196 EXTENDED REPORT Tartrate resistant acid phosphatase (TRAP) positive cells in rheumatoid synovium may induce the destruction of articular cartilage H Tsuboi, Y Matsui, K Hayashida, S Yamane, M Maeda-Tanimura,
Extensive osteolysis adjacent to implants is often
 The osteoclastogenic molecules RANKL and RANK are associated with periprosthetic osteolysis D. R. Haynes, T. N. Crotti, A. E. Potter, M. Loric G. J. Atkins, D. W. Howie, D. M. Findlay From the University
The osteoclastogenic molecules RANKL and RANK are associated with periprosthetic osteolysis D. R. Haynes, T. N. Crotti, A. E. Potter, M. Loric G. J. Atkins, D. W. Howie, D. M. Findlay From the University
Osteoclast Activity Assay Substrate
 Osteoclast Activity Assay Substrate For Research Use Only OSCOTECT INC. #3201 Trade Tower Samsung-dong 159, Kangnam-ku Seoul 135-729, Korea Tel: +82-2-6000-7666 / Fax: +82-2-6000-7667 customer@oscotec.com
Osteoclast Activity Assay Substrate For Research Use Only OSCOTECT INC. #3201 Trade Tower Samsung-dong 159, Kangnam-ku Seoul 135-729, Korea Tel: +82-2-6000-7666 / Fax: +82-2-6000-7667 customer@oscotec.com
B cells: a fundamental role in the pathogenesis of rheumatoid arthritis?
 Rheumatology 2005;44(Suppl. 2):ii3 ii7 B cells: a fundamental role in the pathogenesis of rheumatoid arthritis? doi:10.1093/rheumatology/keh616 The role of T cells in the pathogenesis of RA is well established,
Rheumatology 2005;44(Suppl. 2):ii3 ii7 B cells: a fundamental role in the pathogenesis of rheumatoid arthritis? doi:10.1093/rheumatology/keh616 The role of T cells in the pathogenesis of RA is well established,
SUPPLEMENTARY INFORMATION
 1. Supplementary Figures and Legends Supplementary Fig. 1. S1P-mediated transcriptional regulation of integrins expressed in OP/monocytoid cells. Real-time quantitative PCR analyses of mrna for two integrins,
1. Supplementary Figures and Legends Supplementary Fig. 1. S1P-mediated transcriptional regulation of integrins expressed in OP/monocytoid cells. Real-time quantitative PCR analyses of mrna for two integrins,
Supporting Information
 Supporting Information Bellora et al. 10.1073/pnas.1007654108 SI Materials and Methods Cells. NK cells purified from peripheral blood mononuclear cells (PBMC) of healthy donors (Human NK Cell Isolation
Supporting Information Bellora et al. 10.1073/pnas.1007654108 SI Materials and Methods Cells. NK cells purified from peripheral blood mononuclear cells (PBMC) of healthy donors (Human NK Cell Isolation
The Pathogenesis of Bone Erosions in RA FULL VERSION
 The Pathogenesis of Bone Erosions in RA FULL VERSION 1 Key Learning Objectives Understand the role of osteoclasts in normal bone remodeling Comprehend the key processes in pathologic osteoclast functions
The Pathogenesis of Bone Erosions in RA FULL VERSION 1 Key Learning Objectives Understand the role of osteoclasts in normal bone remodeling Comprehend the key processes in pathologic osteoclast functions
Expression of Osteoprotegerin and RANK Ligand in Breast Cancer Bone Metastasis
 J Korean Med Sci 2003; 18: 541-6 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Expression of Osteoprotegerin and RANK Ligand in Breast Cancer Bone Metastasis Bone destruction is primarily
J Korean Med Sci 2003; 18: 541-6 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Expression of Osteoprotegerin and RANK Ligand in Breast Cancer Bone Metastasis Bone destruction is primarily
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Role of JAKs in myeloid cells and autoimmune diseases. Satoshi Kubo, Kunihiro Yamaoka and Yoshiya Tanaka
 131 Mini Review Role of JAKs in myeloid cells and autoimmune diseases Satoshi Kubo, Kunihiro Yamaoka and Yoshiya Tanaka The First Department of Internal Medicine, University of Occupational and Environmental
131 Mini Review Role of JAKs in myeloid cells and autoimmune diseases Satoshi Kubo, Kunihiro Yamaoka and Yoshiya Tanaka The First Department of Internal Medicine, University of Occupational and Environmental
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serafino et al. Thymosin α1 activates complement receptor-mediated phagocytosis in human monocyte-derived macrophages. SUPPLEMENTARY FIGURES
 Supplementary Fig. S1. Evaluation of the purity and maturation of macrophage cultures tested by flow cytometry. The lymphocytic/monocytic cellular fraction was isolated from buffy coats of healthy donors
Supplementary Fig. S1. Evaluation of the purity and maturation of macrophage cultures tested by flow cytometry. The lymphocytic/monocytic cellular fraction was isolated from buffy coats of healthy donors
D.R.Haynes,T.N.Crotti,M.Loric,G.I.Bain 1,G.J.Atkins andd.m.findlay 1
 Rheumatology 2001;40:623±630 Osteoprotegerin and receptor activator of nuclear factor kappab ligand RANKL) regulate osteoclast formation by cells in the human rheumatoid arthritic joint D.R.Haynes,T.N.Crotti,M.Loric,G.I.Bain
Rheumatology 2001;40:623±630 Osteoprotegerin and receptor activator of nuclear factor kappab ligand RANKL) regulate osteoclast formation by cells in the human rheumatoid arthritic joint D.R.Haynes,T.N.Crotti,M.Loric,G.I.Bain
Immunological Aspect of Ozone in Rheumatic Diseases
 Immunological Aspect of Ozone in Rheumatic Diseases Prof. Dr. med. Z. Fahmy Chief Consulting Rheumatologist Augusta Clinic for Rheumatic Diseases And Rehabilitation Bad Kreuznach Germany Rheumatoid arthritis
Immunological Aspect of Ozone in Rheumatic Diseases Prof. Dr. med. Z. Fahmy Chief Consulting Rheumatologist Augusta Clinic for Rheumatic Diseases And Rehabilitation Bad Kreuznach Germany Rheumatoid arthritis
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
IL-17 in health and disease. March 2014 PSO13-C051n
 IL-17 in health and disease March 2014 PSO13-C051n Originally Researchers Suggested That IL-12 and IL-4 drove Th Cell Differentiation Naïve CD4 + T cell Question: Which of these cell types is responsible
IL-17 in health and disease March 2014 PSO13-C051n Originally Researchers Suggested That IL-12 and IL-4 drove Th Cell Differentiation Naïve CD4 + T cell Question: Which of these cell types is responsible
Generation of monocytederived Dendritic Cells (modcs)
 monocytederived Dendritic (modcs) Application Note Background Dendritic (DCs) are so called because of their characteristic cell surface projections that resemble the dendrites of neurons (see Fig 1 and
monocytederived Dendritic (modcs) Application Note Background Dendritic (DCs) are so called because of their characteristic cell surface projections that resemble the dendrites of neurons (see Fig 1 and
McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells
 Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival
 Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
The Adaptive Immune Responses
 The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
Siglec-15 Is A Potential Therapeutic Target For Postmenopausal Osteoporosis
 Siglec-15 Is A Potential Therapeutic Target For Postmenopausal Osteoporosis Yusuke Kameda, Masahiko Takahata, Tomohiro Shimizu, Hiroki Hamano, Norimasa Iwasaki. Department of Orthopedic Surgery, Hokkaido
Siglec-15 Is A Potential Therapeutic Target For Postmenopausal Osteoporosis Yusuke Kameda, Masahiko Takahata, Tomohiro Shimizu, Hiroki Hamano, Norimasa Iwasaki. Department of Orthopedic Surgery, Hokkaido
Autoimmune Diseases. Betsy Kirchner CNP The Cleveland Clinic
 Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Single mammalian cell encapsulation
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Single mammalian cell encapsulation
Potential Role of Sphingosine 1-Phosphate in the. Pathogenesis of Rheumatoid Arthritis
 Potential Role of Sphingosine 1-Phosphate in the Pathogenesis of Rheumatoid Arthritis COMMENTARY for Zhao, C., Fernandes, M.J., Turgeon, M., Tancrede, S., Di Battista, J., Poubelle, P.E. and Bourgoin,
Potential Role of Sphingosine 1-Phosphate in the Pathogenesis of Rheumatoid Arthritis COMMENTARY for Zhao, C., Fernandes, M.J., Turgeon, M., Tancrede, S., Di Battista, J., Poubelle, P.E. and Bourgoin,
TRACP & ALP double-stain Kit
 Table of Content I. Description... 2 II. Introduction... 2 III. Principles... 2 IV. Kit components... 3 V. Storage... 3 VI. Preparation of reagents... 3 VII. Methods... 4-7 Cell fixation... 4 Activity
Table of Content I. Description... 2 II. Introduction... 2 III. Principles... 2 IV. Kit components... 3 V. Storage... 3 VI. Preparation of reagents... 3 VII. Methods... 4-7 Cell fixation... 4 Activity
Sipper BK Experimental Animal Co. (Shanghai, China) and bred in a specific. pathogen-free environment. The animal study protocol was approved by the
 Supplementary information, Data S1 Materials and Methods Mice, Ad vectors and reagents Female C57BL/6 mice, 8-10 weeks of age, were purchased from Joint Ventures Sipper BK Experimental Animal Co. (Shanghai,
Supplementary information, Data S1 Materials and Methods Mice, Ad vectors and reagents Female C57BL/6 mice, 8-10 weeks of age, were purchased from Joint Ventures Sipper BK Experimental Animal Co. (Shanghai,
SUPPLEMENTARY INFORMATION. Involvement of IL-21 in the epidermal hyperplasia of psoriasis
 SUPPLEMENTARY INFORMATION Involvement of IL-21 in the epidermal hyperplasia of psoriasis Roberta Caruso 1, Elisabetta Botti 2, Massimiliano Sarra 1, Maria Esposito 2, Carmine Stolfi 1, Laura Diluvio 2,
SUPPLEMENTARY INFORMATION Involvement of IL-21 in the epidermal hyperplasia of psoriasis Roberta Caruso 1, Elisabetta Botti 2, Massimiliano Sarra 1, Maria Esposito 2, Carmine Stolfi 1, Laura Diluvio 2,
Macrophage-lymphocyte clustering in rheumatoid arthritis
 Antn. rheum. Dis. (1975), 34, 38 Macrophage-lymphocyte clustering in rheumatoid arthritis F. W. S. WEBB, M. BAKER, R. WEISBART, R. BLUESTONE, AND L. GOLDBERG From the Department of Medicine, Rheumatology
Antn. rheum. Dis. (1975), 34, 38 Macrophage-lymphocyte clustering in rheumatoid arthritis F. W. S. WEBB, M. BAKER, R. WEISBART, R. BLUESTONE, AND L. GOLDBERG From the Department of Medicine, Rheumatology
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT
 168 Fluoride Vol. 33 No. 4 168-173 2 Research Report THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT Boguslaw Machaliński, a Maria Zejmo,
168 Fluoride Vol. 33 No. 4 168-173 2 Research Report THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT Boguslaw Machaliński, a Maria Zejmo,
- 1 - Cell types Monocytes THP-1 cells Macrophages. LPS Treatment time (Hour) IL-6 level (pg/ml)
 Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Review Bone loss Factors that regulate osteoclast differentiation: an update Sophie Roux* and Philippe Orcel
 http://arthritis-research.com/content/2/6/451 Review Bone loss Factors that regulate osteoclast differentiation: an update Sophie Roux* and Philippe Orcel * Lariboisière Hospital, Paris, and *Bicêtre Hospital,
http://arthritis-research.com/content/2/6/451 Review Bone loss Factors that regulate osteoclast differentiation: an update Sophie Roux* and Philippe Orcel * Lariboisière Hospital, Paris, and *Bicêtre Hospital,
Adaptive Immunity. Jeffrey K. Actor, Ph.D. MSB 2.214,
 Adaptive Immunity Jeffrey K. Actor, Ph.D. MSB 2.214, 500-5344 Lecture Objectives: Understand role of various molecules including cytokines, chemokines, costimulatory and adhesion molecules in the development
Adaptive Immunity Jeffrey K. Actor, Ph.D. MSB 2.214, 500-5344 Lecture Objectives: Understand role of various molecules including cytokines, chemokines, costimulatory and adhesion molecules in the development
Title: NATURAL KILLER CELL FUNCTIONS AND SURFACE RECEPTORS
 LECTURE: 14 Title: NATURAL KILLER CELL FUNCTIONS AND SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the general morphology of the NK-cells. Enumerate the different functions
LECTURE: 14 Title: NATURAL KILLER CELL FUNCTIONS AND SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the general morphology of the NK-cells. Enumerate the different functions
EXPERIMENTAL SALMONELLOSIS
 EXPERIMENTAL SALMONELLOSIS INTRACELLULAR GROWTH OF Salmonella enteritidis INGESTED IN MONONUCLEAR PHAGOCYTES OF MICE, AND CELLULAR BASIS OF IMMUNITY SUSUMU MITSUHASHI, ICHIEI SATO, AND TOKUMITSU TANAKA
EXPERIMENTAL SALMONELLOSIS INTRACELLULAR GROWTH OF Salmonella enteritidis INGESTED IN MONONUCLEAR PHAGOCYTES OF MICE, AND CELLULAR BASIS OF IMMUNITY SUSUMU MITSUHASHI, ICHIEI SATO, AND TOKUMITSU TANAKA
Synovial fluid macrophages are capable of osteoclast formation and resorption
 Journal of Pathology Published online 8 November 2005 in Wiley InterScience (www.interscience.wiley.com). DOI: 10.1002/path.1891 Original Paper Synovial fluid macrophages are capable of osteoclast formation
Journal of Pathology Published online 8 November 2005 in Wiley InterScience (www.interscience.wiley.com). DOI: 10.1002/path.1891 Original Paper Synovial fluid macrophages are capable of osteoclast formation
CD14 + S100A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer
 CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study
 Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study Antigen Species Clone Source Dilution Insulin Guinea pig - Dako, Carpinteria, 1:200 Glucagon Rabbit - Dako, Carpinteria,
Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study Antigen Species Clone Source Dilution Insulin Guinea pig - Dako, Carpinteria, 1:200 Glucagon Rabbit - Dako, Carpinteria,
MAF Shalaby Prof. Rheumatology Al Azhar University, Cairo, Egypt.
 MAF Shalaby Prof. Rheumatology Al Azhar University, Cairo, Egypt. AUTOIMMUNE DISEASE RA SLE VASCULITIS RELAPSING POLYCHONDRITIS SS DM/PM SJOGREN S SYNDROME RHEUMATOID ARTHRITIS Classically immune mediated
MAF Shalaby Prof. Rheumatology Al Azhar University, Cairo, Egypt. AUTOIMMUNE DISEASE RA SLE VASCULITIS RELAPSING POLYCHONDRITIS SS DM/PM SJOGREN S SYNDROME RHEUMATOID ARTHRITIS Classically immune mediated
Bone resorption by cells isolated from rheumatoid synovium
 Annals of the Rheumatic Diseases 1992; 51: 1223-1229 1223 Nuffield Department of Orthopaedic Surgery and Department of Pathology, Nuffield Orthopaedic Centre, Oxford, United Kingdom J S Chang C J Buistrode
Annals of the Rheumatic Diseases 1992; 51: 1223-1229 1223 Nuffield Department of Orthopaedic Surgery and Department of Pathology, Nuffield Orthopaedic Centre, Oxford, United Kingdom J S Chang C J Buistrode
Sunitinib, an orally available receptor tyrosine kinase inhibitor, induces monocytic
 Sunitinib, an orally available receptor tyrosine kinase inhibitor, induces monocytic differentiation of acute myeogenouse leukemia cells that is enhanced by 1,25-dihydroxyviatmin D 3. To the Editor: Sunitinib,
Sunitinib, an orally available receptor tyrosine kinase inhibitor, induces monocytic differentiation of acute myeogenouse leukemia cells that is enhanced by 1,25-dihydroxyviatmin D 3. To the Editor: Sunitinib,
Bisphosphonate-induced differential modulation of immune cell function in gingiva and bone marrow in vivo: Role in osteoclastmediated
 /, Vol. 6, No. 24 Bisphosphonate-induced differential modulation of immune cell function in gingiva and bone marrow in vivo: Role in osteoclastmediated NK cell activation Han-Ching Tseng 1,*, Keiichi Kanayama
/, Vol. 6, No. 24 Bisphosphonate-induced differential modulation of immune cell function in gingiva and bone marrow in vivo: Role in osteoclastmediated NK cell activation Han-Ching Tseng 1,*, Keiichi Kanayama
Ultrastructural Study of Human Natural Killer CNK) Cell*)
 Hiroshima Journal of Medical Sciences Vol. 31, No. 1, March, 1982 HJIM 31-6 31 Ultrastructural Study of Human Natural Killer CNK) Cell*) Yoshinori KAWAGUCHI, Eishi KITTAKA, Yoshito TANAKA, Takeo TANAKA
Hiroshima Journal of Medical Sciences Vol. 31, No. 1, March, 1982 HJIM 31-6 31 Ultrastructural Study of Human Natural Killer CNK) Cell*) Yoshinori KAWAGUCHI, Eishi KITTAKA, Yoshito TANAKA, Takeo TANAKA
Interleukin-l Production in Patients with Nonlymphocytic Leukemia and Myelodysplastic Syndromes
 Interleukin-l Production in Patients with Nonlymphocytic Leukemia and Myelodysplastic Syndromes N. J. Simbirtseva 1 A common feature of all cases of myeloid leukemia is a block in normal maturation of
Interleukin-l Production in Patients with Nonlymphocytic Leukemia and Myelodysplastic Syndromes N. J. Simbirtseva 1 A common feature of all cases of myeloid leukemia is a block in normal maturation of
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3
 EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell?
 Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
ImageStream cytometer analysis. Cells were cultured as described above in vented-cap
 ImageStream cytometer analysis. Cells were cultured as described above in vented-cap polypropylene tubes, stained with αcd66b-fitc, αm-dc8-pe and αcd56-pe-cy5.5 mabs, washed and fixed with 4 % (w/v) paraformaldehyde.
ImageStream cytometer analysis. Cells were cultured as described above in vented-cap polypropylene tubes, stained with αcd66b-fitc, αm-dc8-pe and αcd56-pe-cy5.5 mabs, washed and fixed with 4 % (w/v) paraformaldehyde.
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
silent epidemic,. (WHO),
 Tel: 02-740-8686; E-mail: hhbkim@snu.ac.kr silent epidemic,. (WHO),. 5 3, 1. 50 70. 50%, 25%, 20% (12~35%). 2.8% 0.7% 4. ( ). bone remodeling (osteoblast), (osteoclast),.. 3~4.. 70% (osteocyte) (bone lining
Tel: 02-740-8686; E-mail: hhbkim@snu.ac.kr silent epidemic,. (WHO),. 5 3, 1. 50 70. 50%, 25%, 20% (12~35%). 2.8% 0.7% 4. ( ). bone remodeling (osteoblast), (osteoclast),.. 3~4.. 70% (osteocyte) (bone lining
Lack of association of IL-2RA and IL-2RB polymorphisms with rheumatoid arthritis in a Han Chinese population
 Lack of association of IL-2RA and IL-2RB polymorphisms with rheumatoid arthritis in a Han Chinese population J. Zhu 1 *, F. He 2 *, D.D. Zhang 2 *, J.Y. Yang 2, J. Cheng 1, R. Wu 1, B. Gong 2, X.Q. Liu
Lack of association of IL-2RA and IL-2RB polymorphisms with rheumatoid arthritis in a Han Chinese population J. Zhu 1 *, F. He 2 *, D.D. Zhang 2 *, J.Y. Yang 2, J. Cheng 1, R. Wu 1, B. Gong 2, X.Q. Liu
ulation of NK cells that retain the capability of expressing the HNK-1 differentiation antigen. Children with the Chediak-Higashi (CH)' syndrome,
 RAPID PUBLICATIONS Natural Killer (HNK-1l) Cells in Chediak-Higashi Patients Are Present in Numbers but Are Abnormal in Function and Morphology TORu ABO, JOHN C. RODER, WATARU ABO, MAX D. COOPER, and CHARLES
RAPID PUBLICATIONS Natural Killer (HNK-1l) Cells in Chediak-Higashi Patients Are Present in Numbers but Are Abnormal in Function and Morphology TORu ABO, JOHN C. RODER, WATARU ABO, MAX D. COOPER, and CHARLES
were isolated from the freshly drawn blood of healthy donors and ACS patients using the
 Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
Hematopoiesis. Hematopoiesis. Hematopoiesis
 Chapter. Cells and Organs of the Immune System Hematopoiesis Hematopoiesis- formation and development of WBC and RBC bone marrow. Hematopoietic stem cell- give rise to any blood cells (constant number,
Chapter. Cells and Organs of the Immune System Hematopoiesis Hematopoiesis- formation and development of WBC and RBC bone marrow. Hematopoietic stem cell- give rise to any blood cells (constant number,
/00/$03.00/0 Vol. 141, No. 9. Osteoprotegerin Produced by Osteoblasts Is an Important Regulator in Osteoclast Development and Function*
 0013-7227/00/$03.00/0 Vol. 141, No. 9 Endocrinology Printed in U.S.A. Copyright 2000 by The Endocrine Society Osteoprotegerin Produced by Osteoblasts Is an Important Regulator in Osteoclast Development
0013-7227/00/$03.00/0 Vol. 141, No. 9 Endocrinology Printed in U.S.A. Copyright 2000 by The Endocrine Society Osteoprotegerin Produced by Osteoblasts Is an Important Regulator in Osteoclast Development
LIGHT (TNFSF14), a Novel Mediator of Bone Resorption, Is Elevated in Rheumatoid Arthritis
 ARTHRITIS & RHEUMATISM Vol. 54, No. 5, May 2006, pp 1451 1462 DOI 10.1002/art.21821 2006, American College of Rheumatology LIGHT (TNFSF14), a Novel Mediator of Bone Resorption, Is Elevated in Rheumatoid
ARTHRITIS & RHEUMATISM Vol. 54, No. 5, May 2006, pp 1451 1462 DOI 10.1002/art.21821 2006, American College of Rheumatology LIGHT (TNFSF14), a Novel Mediator of Bone Resorption, Is Elevated in Rheumatoid
Patients with knee OA and with joint pain, stiffness and/or functional impairment interfering
 Supplementary Material to Alunno et al. Platelets Contribute to the Accumulation of Matrix Metalloproteinase Type 2 in Synovial Fluid in Osteoarthritis (https://doi.org/10.1160/th17-06-0379) Supplemental
Supplementary Material to Alunno et al. Platelets Contribute to the Accumulation of Matrix Metalloproteinase Type 2 in Synovial Fluid in Osteoarthritis (https://doi.org/10.1160/th17-06-0379) Supplemental
The IL-7 Receptor A Key Factor in HIV Pathogenesis
 The IL-7 Receptor A Key Factor in HIV Pathogenesis Paul MacPherson PhD, MD, FRCPC Associate Professor Division of Infectious Diseases Ottawa Hospital, General Campus Ottawa Hospital Research Institute
The IL-7 Receptor A Key Factor in HIV Pathogenesis Paul MacPherson PhD, MD, FRCPC Associate Professor Division of Infectious Diseases Ottawa Hospital, General Campus Ottawa Hospital Research Institute
Although this presentation includes information regarding pharmaceuticals (including products under development), the information is not intended as
 Although this presentation includes information regarding pharmaceuticals (including products under development), the information is not intended as any advertisement and/or medical advice. Forward-Looking
Although this presentation includes information regarding pharmaceuticals (including products under development), the information is not intended as any advertisement and/or medical advice. Forward-Looking
Quantitative Assay of Paravaccinia Virus Based
 APPrU MICROBIOLOGY, JUly 1972, p. 138-142 Copyright 1972 American Society for Microbiology Vol. 24, No. 1 Printed in U.S.A. Quantitative Assay of Paravaccinia Virus Based on Enumeration of Inclusion-Containing
APPrU MICROBIOLOGY, JUly 1972, p. 138-142 Copyright 1972 American Society for Microbiology Vol. 24, No. 1 Printed in U.S.A. Quantitative Assay of Paravaccinia Virus Based on Enumeration of Inclusion-Containing
Bihong Zhao, M.D, Ph.D Department of Pathology
 Bihong Zhao, M.D, Ph.D Department of Pathology 04-28-2009 Is tumor self or non-self? How are tumor antigens generated? What are they? How does immune system respond? Introduction Tumor Antigens/Categories
Bihong Zhao, M.D, Ph.D Department of Pathology 04-28-2009 Is tumor self or non-self? How are tumor antigens generated? What are they? How does immune system respond? Introduction Tumor Antigens/Categories
International Cartilage Repair Society
 OsteoArthritis and Cartilage (2007) 15, 291e299 ª 2006 Osteoarthritis Research Society International. Published by Elsevier Ltd. All rights reserved. doi:10.1016/j.joca.2006.08.001 Induction of osteoclastlike
OsteoArthritis and Cartilage (2007) 15, 291e299 ª 2006 Osteoarthritis Research Society International. Published by Elsevier Ltd. All rights reserved. doi:10.1016/j.joca.2006.08.001 Induction of osteoclastlike
Department of Immunology, Faculty of Medicine and Graduate School of Medicine, University of Tokyo, Tokyo, Japan
 Available online http://arthritis-research.com/content/4/s3/s227 Supplement Review Signaling crosstalk between RANKL and interferons in osteoclast differentiation Hiroshi Takayanagi, Sunhwa Kim and Tadatsugu
Available online http://arthritis-research.com/content/4/s3/s227 Supplement Review Signaling crosstalk between RANKL and interferons in osteoclast differentiation Hiroshi Takayanagi, Sunhwa Kim and Tadatsugu
IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS
 LECTURE: 07 Title: IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS LEARNING OBJECTIVES: The student should be able to: The chemical nature of the cellular surface receptors. Define the location of the
LECTURE: 07 Title: IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS LEARNING OBJECTIVES: The student should be able to: The chemical nature of the cellular surface receptors. Define the location of the
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: van Seters M, van Beurden M, ten Kate FJW, et al. Treatment
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: van Seters M, van Beurden M, ten Kate FJW, et al. Treatment
L6 GLUT4myc Cell Growth Protocol
 L6 GLUT4myc Cell Growth Protocol Background: Parental L6 cells selected for high fusion (2, 3) were stably transfected with a rat GLUT4 cdna carrying a myc epitope (recognized by the commercially available
L6 GLUT4myc Cell Growth Protocol Background: Parental L6 cells selected for high fusion (2, 3) were stably transfected with a rat GLUT4 cdna carrying a myc epitope (recognized by the commercially available
Microbiology 204: Cellular and Molecular Immunology
 Microbiology 204: Cellular and Molecular Immunology Class meets MWF 1:00-2:30PM (*exceptions: no class Fri Sept 23, Fri Oct 14, Nov 11, or Wed Nov 23) Lectures are open to auditors and will be live-streamed
Microbiology 204: Cellular and Molecular Immunology Class meets MWF 1:00-2:30PM (*exceptions: no class Fri Sept 23, Fri Oct 14, Nov 11, or Wed Nov 23) Lectures are open to auditors and will be live-streamed
Interferon γ regulates idiopathic pneumonia syndrome, a. Th17 + CD4 + T-cell-mediated GvH disease
 Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration
 Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small cell lung cancer
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Experimental Methods Cell culture B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Experimental Methods Cell culture B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
Cytokines, adhesion molecules and apoptosis markers. A comprehensive product line for human and veterinary ELISAs
 Cytokines, adhesion molecules and apoptosis markers A comprehensive product line for human and veterinary ELISAs IBL International s cytokine product line... is extremely comprehensive. The assays are
Cytokines, adhesion molecules and apoptosis markers A comprehensive product line for human and veterinary ELISAs IBL International s cytokine product line... is extremely comprehensive. The assays are
Principles of Adaptive Immunity
 Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology
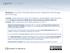 Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
The Adaptive Immune Response. T-cells
 The Adaptive Immune Response T-cells T Lymphocytes T lymphocytes develop from precursors in the thymus. Mature T cells are found in the blood, where they constitute 60% to 70% of lymphocytes, and in T-cell
The Adaptive Immune Response T-cells T Lymphocytes T lymphocytes develop from precursors in the thymus. Mature T cells are found in the blood, where they constitute 60% to 70% of lymphocytes, and in T-cell
Convenient large-scale differentiation of fully customized monocyte-derived macrophages from human PBMC in vitro
 Monocyte Attachment Medium Addition by user: SupplementMix: Date: Cat. No.: C-28051 Lot No.: Sample Volume 250 ml Exp.: Sample Store at: + 4 C For research use only for research use only for research use
Monocyte Attachment Medium Addition by user: SupplementMix: Date: Cat. No.: C-28051 Lot No.: Sample Volume 250 ml Exp.: Sample Store at: + 4 C For research use only for research use only for research use
Supplemental Data Figure S1 Effect of TS2/4 and R6.5 antibodies on the kinetics of CD16.NK-92-mediated specific lysis of SKBR-3 target cells.
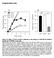 Supplemental Data Figure S1. Effect of TS2/4 and R6.5 antibodies on the kinetics of CD16.NK-92-mediated specific lysis of SKBR-3 target cells. (A) Specific lysis of IFN-γ-treated SKBR-3 cells in the absence
Supplemental Data Figure S1. Effect of TS2/4 and R6.5 antibodies on the kinetics of CD16.NK-92-mediated specific lysis of SKBR-3 target cells. (A) Specific lysis of IFN-γ-treated SKBR-3 cells in the absence
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Primary Adult Naïve CD4+ CD45RA+ Cells. Prepared by: David Randolph at University of Alabama, Birmingham
 Primary Adult Naïve CD4+ CD45RA+ Cells Prepared by: David Randolph (drdrdr@uab.edu) at University of Alabama, Birmingham Goal: To obtain large numbers of highly pure primary CD4+ CD45RO- CD25- cells from
Primary Adult Naïve CD4+ CD45RA+ Cells Prepared by: David Randolph (drdrdr@uab.edu) at University of Alabama, Birmingham Goal: To obtain large numbers of highly pure primary CD4+ CD45RO- CD25- cells from
9607 Dr. Perry Road Suite 103. Ijamsville, MD Tel: (301) Fax: (301) Ultrasensitive Samoa Human IL-33 Immunoassay
 AssayGate, Inc. 9607 Dr. Perry Road Suite 103. Ijamsville, MD 21754. Tel: (301)874-0988. Fax: (301)560-8288. Ultrasensitive Samoa Human IL-33 Immunoassay INTRODUCTION Interleukin-33 (IL-33), a cytokine
AssayGate, Inc. 9607 Dr. Perry Road Suite 103. Ijamsville, MD 21754. Tel: (301)874-0988. Fax: (301)560-8288. Ultrasensitive Samoa Human IL-33 Immunoassay INTRODUCTION Interleukin-33 (IL-33), a cytokine
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Aggregation of Sphingosine-DNA and cell construction using components from egg white
 Integrative Molecular Medicine Research Article ISSN: 2056-6360 Aggregation of Sphingosine-DNA and cell construction using components from egg white Shoshi Inooka* The Institute of Japan Applied Food Materials
Integrative Molecular Medicine Research Article ISSN: 2056-6360 Aggregation of Sphingosine-DNA and cell construction using components from egg white Shoshi Inooka* The Institute of Japan Applied Food Materials
SUPPLEMENTARY INFORMATION. CXCR4 inhibitors could benefit to HER2 but not to Triple-Negative. breast cancer patients
 SUPPLEMENTARY INFORMATION CXCR4 inhibitors could benefit to HER2 but not to Triple-Negative breast cancer patients Lefort S. 1,2, Thuleau A. 3, Kieffer Y. 1,2, Sirven P. 1,2, Bieche I. 4, Marangoni E.
SUPPLEMENTARY INFORMATION CXCR4 inhibitors could benefit to HER2 but not to Triple-Negative breast cancer patients Lefort S. 1,2, Thuleau A. 3, Kieffer Y. 1,2, Sirven P. 1,2, Bieche I. 4, Marangoni E.
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
The interleukin-23/interleukin-17 axis and the role of Treg/ Th17 cells in rheumatoid arthritis and joint destruction
 Rheumatic Diseases Page 1 of 6 The interleukin-23/interleukin-17 axis and the role of Treg/ Th17 cells in rheumatoid arthritis and joint destruction S AlFadhli* Abstract Introduction Rheumatoid arthritis
Rheumatic Diseases Page 1 of 6 The interleukin-23/interleukin-17 axis and the role of Treg/ Th17 cells in rheumatoid arthritis and joint destruction S AlFadhli* Abstract Introduction Rheumatoid arthritis
