Induction of Small Intestinal Damage in Rats Following Combined Treatment with Cyclooxygenase-2 and Nitric-Oxide Synthase Inhibitors
|
|
|
- Ross Campbell
- 5 years ago
- Views:
Transcription
1 /04/ $20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 310, No. 2 Copyright 2004 by The American Society for Pharmacology and Experimental Therapeutics 65961/ JPET 310: , 2004 Printed in U.S.A. Induction of Small Intestinal Damage in Rats Following Combined Treatment with Cyclooxygenase-2 and Nitric-Oxide Synthase Inhibitors Ryoko Ohno, Aya Yokota, Akiko Tanaka, and Koji Takeuchi Department of Pharmacology and Experimental Therapeutics, Kyoto Pharmaceutical University, Misasagi, Yamashina, Kyoto, Japan Received January 21, 2004; accepted March 24, 2004 ABSTRACT Nitric oxide (NO) produced by constitutively expressed NO synthase (cnos) plays an important role in maintaining the mucosal integrity of the small intestine, in collaboration with prostaglandins produced by cyclooxygenase (COX)-1. We examined whether intestinal damage is provoked in rats under inhibition of both cnos and COX-2. The animals were given L-NAME (N G -nitro-l-arginine methyl ester), aminoguanidine, or rofecoxib, either alone or in combination, and killed 24 h later. Neither L-NAME nor aminoguanidine alone provoked damage in the small intestinal mucosa within 24 h, yet L-NAME produced damage in a L-arginine-sensitive manner when administered together with rofecoxib. L-NAME up-regulated the expression of COX-2 mrna, and the prostaglandin E 2 (PGE 2 ) content following the L-NAME administration significantly increased 12 h later, in both a rofecoxib- and a L-arginine-inhibitable manner. L-NAME enhanced intestinal motility, decreased Article, publication date, and citation information can be found at DOI: /jpet mucus secretion, and increased the number of bacteria in the mucosa. The up-regulation of COX-2 expression and PGE 2 production by L-NAME was inhibited by prior administration of atropine, at a dose that inhibited the intestinal hypermotility. The intestinal lesions induced by L-NAME plus rofecoxib were prevented by pretreatment with ampicillin and aminoguanidine as well as atropine, indicating the involvement of bacteria, inducible nitric oxide synthase, and hypermotility in the pathogenesis. These results suggest that inhibition of both cnos and COX-2 provokes intestinal damage, similar to inhibition of both COX-1 and COX-2. Inhibition of cnos, similar to COX-1, upregulates COX-2 expression, the process being associated with intestinal hypermotility and bacterial invasion, and this may be a key to the occurrence of intestinal damage associated with COX-2 inhibition. Nonsteroidal anti-inflammatory drugs (NSAIDs) such as indomethacin cause gastrointestinal ulceration as an adverse reaction (Whittle, 1981; Lanza, 1984; Takuechi et al., 1986). Although a deficiency of prostaglandins (PGs) caused by cyclooxygenase-1 (COX-1) is considered to play a critical role in the pathogenesis of these lesions (O Neill and Ford-Hutchinson, 1993), recent studies showed that inhibition of both COX-1 and COX-2 is required for induction of damage in the gastrointestinal mucosa following administration of NSAIDs (Wallace et al., 2000; Tanaka et al., 2001). Since inhibition of COX-1 was accompanied by the expression of COX-2 mrna in these tissues (Tanaka et al., 2002a,b) and mucosal PGE 2 levels recovered from 6 h after administration of SC-560 in a rofecoxib-sensitive manner (Tanaka et al., 2002b), it is assumed that PGs produced by COX-2 contribute to maintaining the mucosal integrity of the gastrointestinal tract by antagonizing deleterious events caused by inhibition of COX-1. Nitric oxide (NO) plays a dual role in the pathogenesis of NSAID-induced intestinal damage: a protective role when produced by constitutively expressed NO synthase (cnos) and a pro-ulcerogenic role when produced by inducible NO synthase (inos) (Tanaka et al., 1999). Indeed, indomethacininduced intestinal lesions were prevented or aggravated by prior administration of NOR-3, the NO donor, or L-NAME (N G -nitro-l-arginine methyl ester), the nonselective NOS inhibitor, respectively (Tanaka et al., 2001). Thus, NO produced by cnos is important for maintaining the mucosal integrity of the small intestine, just mimicking the role of COX-1/PGs. If so, intestinal damage might also be induced by inhibition of both cnos and COX-2. ABBREVIATIONS: NSAID, nonsteroidal anti-inflammatory drug; PG, prostaglandin; COX, cyclooxygenase; PGE 2, prostaglandin E 2 ; SC-560, 5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluoromethyl)-1H-pyrazole; NO, nitric oxide; cnos, constitutively expressed nitric-oxide synthase; inos, inducible nitric-oxide synthase; L-NAME, N G -nitro-l-arginine methyl ester; PCR, polymerase chain reaction; G3PDH, glyceraldehyde-3- phosphate dehydrogenase; PBS, phosphate-buffered saline; CFU, colony-forming unit(s); LPS, lipopolysaccharide. 821
2 822 Ohno et al. In the present study, we therefore examined whether or not intestinal damage is provoked in rats following the combined administration of L-NAME and the selective COX-2 inhibitor rofecoxib (Chan et al., 1995). We also examined whether inhibition of NO production by L-NAME up-regulates the expression of COX-2 mrna in the intestinal mucosa, similar to inhibition of PGs by SC-560, the selective COX-1 inhibitor (Smith et al., 1998). In addition, since NSAID-induced intestinal lesions occur in association with various functional events, such as intestinal motility and bacterial counts (Tanaka et al., 2002c), we investigated the effect of COX and NOS inhibitors on these parameters at different time points, according to a previous study (Takeuchi et al., 2002). Materials and Methods Animals. Male Sprague-Dawley rats ( g; Nippon Charles River Laboratories, Inc., Shizuoka, Japan) were used. Studies were carried out using groups of four to eight animals without fasting, unless otherwise specified. All experimental procedures described here were approved by the Experimental Animal Research Committee of Kyoto Pharmaceutical University. Evaluation of Small Intestinal Ulcerogenic Properties. In the first study, the animals were given p.o. with SC-560 (a selective COX-1 inhibitor: 10 mg/kg) (Smith et al., 1998; Tanaka et al., 2002b), rofecoxib (a selective COX-2 inhibitor: 10 mg/kg) (Chan et al., 1995; Tanaka et al., 2002b), or SC-560 plus rofecoxib. In the second study, they were given s.c. with L-NAME (10, 20, and 50 mg/kg) or aminoguanidine (20 mg/kg), together with or without p.o. administration of rofecoxib (10 mg/kg). The dose of L-NAME or aminoguanidine was selected to inhibit basal NO production or inos activity in rats, respectively (Tanaka et al., 1999, 2001). In the case of L-NAME (50 mg/kg) plus rofecoxib, the protective effect of L-arginine (500 mg/kg), atropine (3 mg/kg: an anticholinergic drug), ampicillin (800 mg/kg: an antibiotics), or aminoguanidine (20 mg/kg) on the intestinal lesions was examined. L-arginine, atropine, and aminoguanidine were given s.c. 30 min before administration of L-NAME plus rofecoxib, whereas ampicillin was given p.o. twice 24 h and 30 min before. In each study, the animals were killed 24 h later under deep diethyl ether anesthesia, and the small intestine was excised and treated with 2% formalin for fixation of the tissue walls. Then, it was opened along the antimesenteric attachment and examined for damage under a dissecting microscope with square grids ( 10). The area (millimeters squared) of macroscopically visible lesions was measured, summed per small intestine, and used as a lesion score. The person measuring the lesions did not know the treatments given to the animals. In some cases, the animals were killed at various time points (3, 6, 12, and 24 h) after administration of treated with L-NAME plus rofecoxib. Determination of Mucosal PGE 2 Content. Levels of PGE 2 in the small intestinal mucosa were measured after administration of SC-560 (10 mg/kg), rofecoxib (10 mg/kg), L-NAME (50 mg/kg), or aminoguanidine (20 mg/kg). The animals were killed under deep diethyl ether anesthesia 3 h after the administration, and the small intestinal tissue was isolated, weighed, and placed in a tube containing 100% ethanol plus 0.1 M indomethacin (Futaki et al., 1994). In some cases, the mucosal PGE 2 content was measured at 12 h after administration of L-NAME (50 mg/kg), with or without rofecoxib, L-arginine (500 mg/kg, s.c.), or atropine (3 mg/kg, s.c.) given 30 min before L-NAME. Then, the tissues were homogenized using a polytron homogenizer (IKA, Tokyo, Japan) and centrifuged at 10,000 rpm for 10 min at 4 C. After the supernatant of each sample had been evaporated with N 2 gas, the residue was dissolved in assay buffer and used for determination of PGE 2. The concentration of PGE 2 was measured using a PGE 2 enzyme immunoassay kit (Amersham Biosciences UK, Ltd., Little Chalfont, Buckinghamshire, UK). Analysis of COX-2 and Glyceraldehyde-3-Phosphate Dehydrogenase (G3PDH) mrna Expression by Reverse Transcription-PCR. The animals were killed under deep diethyl ether anesthesia 6 h after administration of SC-560 (10 mg/kg), rofecoxib (10 mg/kg), or L-NAME (50 mg/kg), and their small intestines were removed, frozen in liquid nitrogen, and stored at 80 C until use. In some cases, L-NAME was administered together with L-arginine (500 mg/kg, s.c.) or atropine (3 mg/kg, s.c.) given 30 min before L-NAME. Intestinal tissue samples were pooled from two to three rats for extraction of total RNA, which was prepared by a single-step acid phenol-chloroform extraction procedure by use of TRIzol (Invitrogen, Carlsbad, CA). Total RNA primed by random hexadeoxy ribonucleotide was reverse-transcribed with the SuperScript preamplification system (Invitrogen). The sequences of sense and antisense primers for the rat COX-2 were 5 -TGATGACTGCCCAACTCCCATG-3 and 5 -AATGTTGAAG GTGTCCGGCAGC-3, respectively, giving rise to a 702-base pair PCR product (Iso et al., 1995). For the rat G3PDH, a housekeeping gene, the sequences were 5 -GAACGGGAAGCTCACT- GGCATGGC-3 for the sense primer and 5 -TGAGG TCCACCAC- CCTGTTGCTG-3 for the antisense primer, giving rise to a 310-base pair PCR product (Feng et al., 1993). An aliquot of the reverse transcription reaction product served as a template in 35 cycles of PCR with 1 min of denaturation at 94 C, 0.5 min of annealing at 58 C, and 1 min of extension at 72 C on a thermal cycler. A portion of the PCR mixture was electrophoresed in 1.8% agarose gel in Tris-EDTA-acetic acid buffer (40 mm Tris buffer, 2 mm EDTA, and 20 mm acetic acid, ph 8.1), and the gel was stained with ethidium bromide and photographed. Determination of Small Intestinal Motility. Intestinal motility was determined using a miniature balloon according to previous papers (Kunikata et al., 2002; Takeuchi et al., 2002). In brief, the rat was anesthetized with urethane (1.25 g/kg i.p.), and the trachea was cannulated to facilitate respiration. A midline incision was made to expose the small intestine, and a thin, saline-filled balloon, made from silicone rubber and attached to a polyethylene catheter, was introduced into the jejunum via a small incision and tied in place avoiding large blood vessels. The volume in the balloon was adjusted to give an initial resting pressure of 5 mm Hg, which was not sufficient to cause active distension of the intestinal wall, and after allowing the preparation to rest for 30 min, intestinal motility was monitored on a recorder (U-228; Tokai-Irika, Tokyo, Japan) as intraluminal pressure changes, through a pressure transducer and polygraph recorder (Nihon Kodan, Ibaragi, Japan). SC-560 (10 mg/ kg), rofecoxib (10 mg/kg), or L-NAME (50 mg/kg) was given i.d. or s.c., respectively, after basal intestinal motility had well stabilized, and the motility was measured for 2 h thereafter. In some cases, the effect of L-arginine (500 mg/kg s.c.) or atropine (3 mg/kg s.c.) on the intestinal hypermotility induced by L-NAME was also examined. Determination of the Number of Enterobacteria. The enterobacteria were enumerated according to a modified method originally described by Reuter et al. (1997). Six hours after administration of SC-560 (10 mg/kg), rofecoxib (10 mg/kg), or L-NAME (50 mg/kg), the animals were killed under deep diethyl ether anesthesia, and their small intestine was removed. After the rinsing of each intestine with sterile saline, the mucosa was scraped with glass slides, weighed, and homogenized in 1 ml of sterile phosphate-buffered saline (PBS) per 100 mg of wet tissue. Aliquots of the homogenate were placed on blood agar and Gifu anaerobic medium agar (Nissui, Tokyo, Japan). Blood agar plates were incubated at 37 C for 24 h under aerobic conditions, whereas Gifu anaerobic medium agar plates were incubated for 48 h under standard anaerobic conditions (BBL Gas Pack Pouch Anaerobic System; BD Biosciences, San Jose, CA). Plates containing 10 to 200 colony-forming units (CFU) were examined for numbers of enterobacteria, and the data were expressed as log CFU per gram of tissue. Determination of Intestinal Mucus Content. Two hours after administration of various COX and NOS inhibitors, the animals were killed under deep diethyl ether anesthesia, and the intestine
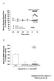
3 was removed and opened along the antimesenteric attachment. The mucus content including soluble and adherent mucus gel was measured as described by Azumi et al. (1981). Briefly, the intestinal mucosa was immersed in 20 ml of PBS containing 2% (w/w) N-acetylcysteine and gently stirred with a magnetic stirrer for 5 min at room temperature. Then, the mucosa was rinsed with 10 ml of PBS twice, and the solution recovered as the adherent samples was dissolved in 4 ml of 50 mm Tris-HCl (ph 7.2) containing 2% Triton X-100, homogenized with a Polytron tissue homogenizer (IKA), and centrifuged at 10,000 rpm for 30 min at 4 C. An aliquot of the supernatant was applied to a column of Sepharose 2B (Amersham Biosciences AB, Uppsala, Sweden) every 50 cm and eluted with 50 mm Tris-HCl (ph 7.2) containing 2% Triton X-100. The void volume fraction was collected, and the hexose content was measured as mucin by the phenol-sulfuric acid method, using galactose as the standard. Preparation of Drugs. The drugs used were SC-560 (Cayman Chemical, Ann Arbor, MI), rofecoxib (Banyu, Tokyo, Japan), L-NAME, aminoguanidine, L-arginine, atropine (Nakarai tesque, Kyoto, Japan), and urethane (Tokyo Kasei, Tokyo, Japan). Both SC-560 and rofecoxib were suspended in a hydroxy propyl cellulose solution (Wako Pure Chemicals, Osaka, Japan), whereas other drugs were dissolved in saline. All drugs were prepared immediately before use and administered p.o., i.d., or s.c. in a volume of 0.5 ml/100 g body weight. Statistics. Data are presented as the mean S.E. of four to eight rats per group. Statistical analyses were performed using the twotailed Dunnett s multiple comparison test, and values of P 0.05 were considered significant. Results Effects of COX and NOS Inhibitors on Intestinal Mucosa and PGE 2 Content. Neither SC-560 (10 mg/kg) nor rofecoxib (10 mg/kg) induced damage in the small intestine within 24 h (Fig. 1A); however, when SC-560 was given together with rofecoxib, this treatment produced hemorrhagic lesions at an incidence of 100%, the lesion score being mm 2. Likewise, L-NAME, the nonselective NOS inhibitor (50 mg/kg), did not by itself cause any damage in the intestine, yet the agent provoked damage when given together with rofecoxib, the selective COX-2 inhibitor, the damage score being mm 2 (Fig. 1B). The ulcerogenic action of L-NAME in combination with rofecoxib was observed in a dose-dependent manner, and the damage score was and mm 2 at 10 and 20 mg/kg, respectively (data not shown). Following administration of L-NAME plus rofecoxib, the intestinal lesions were first observed from 6 h later, slightly increased 12 h later, and reached maximum 24 h later, the damage score being , , , and mm 2, respectively. In contrast, aminoguanidine, the selective inos inhibitor (20 mg/kg s.c.), did not cause damage, irrespective of whether or not the agent was given together with rofecoxib. Since L-NAME is known to produce a marked increase in blood pressure in rats (Takeuchi et al., 1994), it is possible that the intestinal ulcerogenic response observed in the present study is due to such increases in blood pressure. To check this point, we examined whether intestinal damage was induced in the presence of rofecoxib (10 mg/kg) by administration of norepinephrine at the dose (1 mg/kg, s.c.) that increased arterial blood pressure similar to L-NAME; however, this treatment caused only minimal change in the intestinal mucosa, and the lesion score was mm 2, Intestinal Damage Induced by L-NAME plus Rofecoxib 823 Fig. 1. Intestinal ulcerogenic responses induced in rats by SC-560, L- NAME or aminoguanidine alone and in the presence of rofecoxib. A, the animals were administered with SC-560 (10 mg/kg) and rofecoxib (10 mg/kg) p.o., either alone or in combination, and they were killed 24 h later. B, the animals were administered L-NAME (50 mg/kg) and aminoguanidine (20 mg/kg) s.c., and the intestinal mucosa was examined 24 h later. One-half the animals were given rofecoxib (10 mg/kg) p.o. 30 min after L-NAME or aminoguanidine. Data are presented as the mean S.E. from four to seven rats. SC, SC-560; Rof, Rofecoxib. which is not significantly different from that ( mm 2 ) in animals given saline alone. On the other hand, SC-560 markedly decreased the mucosal PGE 2 content of the small intestine from ng/g tissue to less than 2 ng/g tissue within 3 h, whereas rofecoxib had no effect on the PGE 2 content (Fig. 2). Neither L-NAME nor aminoguanidine had any effect on the mucosal PGE 2 content of the small intestine when determined 3 h after the administration. Fig. 2. Effect of COX and NOS inhibitors on early measurement of mucosal PGE 2 content of the rat small intestine. Animals given SC-560 (10 mg/kg p.o.), rofecoxib (10 mg/kg p.o.), L-NAME (50 mg/kg s.c.), or aminoguanidine (20 mg/kg s.c.) were killed 3 h later, and the PGE 2 content was measured by enzyme immunoassay. Data are presented as the mean S.E. from five to seven rats., significant difference from control (saline p.o.) at P 0.05.
4 824 Ohno et al. Fig. 4. Effects of L-arginine, atropine, ampicillin, and aminoguanidine on intestinal lesions induced by L-NAME plus rofecoxib in rats. The animals were given L-NAME (50 mg/kg, s.c.) and subsequently rofecoxib (10 mg/kg, p.o.) 30 min later, and the intestinal mucosa was examined 24 h later. L-arginine (500 mg/kg), atropine (3 mg/kg), or aminoguanidine (50 mg/kg) was given s.c. 30 min before L-NAME, whereas ampicillin (800 mg/kg) was given p.o. twice 24 h and 30 min before the above treatment. Data are presented as the mean S.E. from four to eight rats. Significant difference at P 0.05: from control, # from vehicle 1, and $ from vehicle 2. Fig. 5. A, gene expression of COX-2 and G3PDH in the rat intestinal mucosa following administration of L-NAME, SC-560, or rofecoxib. B, the effect of L-arginine or atropine on the expression of COX-2 mrna induced by L-NAME. The animals were given L-NAME (50 mg/kg s.c.), SC-560 (SC; 10 mg/kg p.o.), or rofecoxib (Rof; 10 mg/kg p.o.), and killed 6 h later. L-arginine (500 mg/kg) or atropine (3 mg/kg) was given s.c. 30 min before the administration of L-NAME. M, marker; V, vehicle. Note that COX-2 mrna was clearly expressed in the intestinal mucosa after administration of L-NAME or SC-560 and that the expression of COX-2 following the L-NAME treatment was suppressed by pretreatment with either L-arginine or atropine. Fig. 3. Effects of SC-560, rofecoxib, or L-NAME on bacterial counts in the rat small intestinal mucosa. The animals were given SC-560 (10 mg/kg p.o.), rofecoxib (10 mg/kg p.o.), or L-NAME (50 mg/kg, s.c.) and killed 6 h later. All values are presented as the mean S.E. from four to eight animals per group., significant difference from control (saline, p.o.) at P Effect of COX and NOS Inhibitors on Bacterial Invasion. The number of aerobic and anaerobic enterobacteria in the normal intestinal mucosa was and log CFU/g tissue, respectively (Fig. 3). Six hours following the administration of SC-560, both values were markedly increased as compared with controls, being and log CFU/g tissue, respectively. Likewise, L-NAME also increased the number of bacteria in the mucosa, the values for both aerobic and anaerobic bacteria being equivalent to those observed in SC-560-treated animals; however, the bacterial count remained unchanged following administration of rofecoxib or aminoguanidine. Effects of Ampicillin, Aminoguanidine, L-Arginine, and Atropine on Intestinal Lesions Induced by L-NAME Plus Rofecoxib. The combined treatment with L-NAME plus rofecoxib caused the increase in mucosal bacterial invasion, similar to indomethacin at the ulcerogenic dose (Tanaka et al., 1999; Kunikata et l., 2002; Takeuchi et al., 2002). In the latter model, the bacterial invasion plays a pathogenetic role in the overproduction of NO through inos induction (Tanaka et al., 1999). Thus, we further investigated, using ampicillin the antibiotics and aminoguanidine the selective inhibitor of inos, whether the same would be applied to the pathogenesis of intestinal lesions in response to L-NAME plus rofecoxib. As shown in Fig. 4, the intestinal lesions induced by L-NAME plus rofecoxib were significantly attenuated by prior administration of ampicillin (800 mg/kg p.o.) as well as aminoguanidine (20 mg/kg s.c.), the inhibition being over 90% in both cases. Likewise, the intestinal ulcerogenic response induced by L-NAME plus rofecoxib was significantly attenuated by prior administration of L-arginine (500 mg/kg, s.c.) or atropine (3 mg/kg, s.c.), and the lesion score in both cases decreased to less than 5 mm 2. Expression of COX-2 mrna in the Intestinal Mucosa. The gene expression of COX-2 was negligible in the small intestine of normal rats; however, it was clearly up-regulated 6 h after administration of L-NAME (50 mg/kg) or SC-560 (10 mg/kg) but not rofecoxib (10 mg/kg) (Fig. 5A). By contrast, G3PDH mrna was observed in the intestinal mucosa, irrespective of whether the animal was treated with vehicle, L-NAME, SC-560, or rofecoxib. In addition, the expression of COX-2 mrna following treatment with L-NAME was curtailed when the animal was pretreated with L-arginine (500 m/kg s.c.) or atropine (3 mg/kg s.c.) (Fig. 5B). Changes in Mucosal PGE 2 Content at a Later Period. Although SC-560 at 10 mg/kg significantly decreased the mucosal PGE 2 content at 3 h after the administration, this effect was reversed by PG production due to up-regulation of COX-2 expression 12 h later (Tanaka et al., 2002b). Since we discovered in the present study that L-NAME up-regulated COX-2 expression similar to SC-560, the same might be
5 observed in the animals treated with L-NAME. Thus, we measured the mucosal PGE 2 content 12 h after the administration of L-NAME, in the absence or presence of rofecoxib. The mucosal PGE 2 content of the small intestine was significantly increased by L-NAME (50 mg/kg) when determined at 12 h after the administration (Fig. 6); however, the increase in PGE 2 content induced by L-NAME was significantly attenuated by the combined administration of rofecoxib (10 mg/kg) and L-arginine (500 mg/kg) as well as atropine (3 mg/kg). The PGE 2 content observed at 12 h after administration of SC-560 remained at the same levels as control, yet the values significantly decreased on the combined administration of rofecoxib. Effects of COX and NOS Inhibitors on Intestinal Mucous Content. The total amount of mucus in the small intestine under basal conditions was g galactose/cm tissue. The mucus content significantly decreased in response to SC-560 (10 mg/kg), reaching g galactose/cm tissue 3 h later, approximately 65.8% of basal values (Fig. 7). Likewise, s.c. administration of L-NAME (50 mg/kg) also significantly decreased the secretion of mucus ( g galactose/cm tissue). Neither rofecoxib (10 mg/kg) nor aminoguanidine (20 mg/kg) had any effect on intestinal mucus secretion. Effects of COX and NOS Inhibitors on Intestinal Motility. Since intestinal hypermotility has been implicated as one of the pathogenetic factors in NSAID-induced small intestinal lesions (Takeuchi et al., 2002), we examined the effects of various COX inhibitors on intestinal motility. Under urethane anesthesia, no clear contraction was observed in the small intestine of normal rats, resulting in a fluctuation at baseline levels; however, the intestinal motility was markedly enhanced following administration of SC-560, in regard to both the amplitude and frequency of contraction (Fig. 8A). L-NAME also caused intestinal hypermotility, similar to SC-560, and the effect persisted for over 2 h. In contrast, rofecoxib did not have any influence on intestinal motility, and no clear contraction was observed before and after administration of this agent. Likewise, aminoguanidine had no effect on intestinal motility (data not shown). The Intestinal Damage Induced by L-NAME plus Rofecoxib 825 Fig. 7. Effects of SC-560, rofecoxib, or L-NAME on mucus secretion in the rat small intestine. Animals given SC-560 (10 mg/kg p.o.), rofecoxib (10 mg/kg p.o.), or L-NAME (50 mg/kg s.c.) were killed 3 h later, and the mucus content including soluble and adherent mucus was measured. Data are presented as the mean S.E. from five to eight rats., significant difference from control (saline, p.o.) at P Fig. 6. Effect of L-NAME and SC-560 on late measurement of mucosal PGE 2 content of the rat intestine, with or without rofecoxib. The animals were administered L-NAME (50 mg/kg s.c.) or SC-560 (10 mg/kg p.o.) and killed 12 h later. In one-half the number of animals, rofecoxib (10 mg/kg) was given p.o. together with the above agents. Data are presented as the mean S.E. from five to seven rats. Significant difference at P 0.05: from control and # from vehicle. Fig. 8. Representative recordings showing the effects of SC-560, rofecoxib, or L-NAME on small intestinal motility (A) and the effect of L- arginine or atropine on the intestinal hypermotility induced by L-NAME in rats (B). The animals were given SC-560 (10 mg/kg i.d.), rofecoxib (10 mg/kg i.d.), or L-NAME (50 mg/kg s.c.), and the intestinal motility was measured for 2 h thereafter. L-arginine (500 mg/kg s.c.) or atropine (3 mg/kg s.c.) was given 1 h after the administration of L-NAME. increased motility caused by L-NAME was potently inhibited by L-arginine (500 mg/kg s.c.) or atropine (3 mg/kg s.c.) given 1 h later (Fig. 8B). Discussion The present study confirmed our previous finding that neither SC-560 nor rofecoxib alone caused intestinal damage,
6 826 Ohno et al. yet their combination provoked gross damage in the small intestine (Tanaka et al., 2002b). We further demonstrated that L-NAME given together with rofecoxib did produce intestinal damage, although inhibition of NO production by L-NAME alone induced no damage, the finding being similar to that observed under inhibition of both COX-1 and COX-2 (Tanaka et al., 2002b). Furthermore, we found that L-NAME up-regulated the expression of COX-2 mrna in the intestine, similar to SC-560, a key to the occurrence of intestinal damage associated with COX-2 inhibition. Wallace et al. (2000) recently reported that neither SC-560 nor celecoxib induced gastric lesions, but their combination caused damage in the stomach, suggesting that inhibition of both COX-1 and COX-2 is required for the occurrence of NSAID-induced gastric injury. We also showed that these two agents when administered together produced damage in the intestine, although neither of these agents alone induced any damage (Tanaka et al., 2002b). The present findings together with those data indicate that COX-2 as well as COX-1 plays a role in maintaining the mucosal integrity of the gastrointestinal tract and that the adverse reaction of NSAID is not accounted for solely by inhibition of COX-1. Interestingly, we found that inhibition of NO production by L-NAME, the nonselective NOS inhibitor, also provoked intestinal damage in the presence of rofecoxib, in an L-argininesensitive manner. This effect was not, however, mimicked by aminoguanidine, the selective inos inhibitor, given together with rofecoxib. It was also found that the administration of L-NAME, but not aminoguanidine, up-regulated COX-2 expression in the small intestine, similar to SC-560. We have previously reported that the PGE 2 content of the small intestine was markedly decreased by SC-560, yet gradually recovered from 6 h after the administration, in a rofecoxibsensitive manner (Tanaka et al., 2002b). We confirmed in the present study that the PGE 2 content in SC-560-treated animals was recovered 12 h later to the levels equivalent to those in normal rats, although it was markedly decreased at 3 h after the administration of SC-560. Interestingly, L-NAME had no effect on mucosal PGE 2 production when determined 3 h after administration, but the PGE 2 content in these rats increased 12 h later, in a COX-2-dependent manner. Certainly, L-NAME at the dose used decreases NO production in the small intestine (Tanaka et al., 1999). Since NO produced by cnos plays a role in the regulation of various functions of the small intestine, mimicking the role of COX-1/PGs (Tanaka et al., 2001), it is assumed that a rapid up-regulation of COX-2 expression might represent a redundant or compensatory response to inhibition of PG or NO biosynthesis and contribute to the maintenance of mucosal integrity under such adverse conditions. Because L-NAME causes a persistent increase in systemic blood pressure, it is possible that such blood pressure changes might account partly for the intestinal ulcerogenic action of this agent in the presence of rofecoxib; however, in a preliminary study, we did not observe any damage in the intestinal mucosa following administration of norepinephrine plus rofecoxib, arguing against the above possibility (unpublished data). We previously reported that endogenous NO exerts a dual action in the pathogenesis of indomethacin-induced intestinal lesions; NO generated by NOS expressed constitutively, cnos, is protective against indomethacin, whereas NO derived by inos plays a key pathogenetic role in the ulcerogenic process (Tanaka et al., 1999). Laszlo et al. (1994) demonstrated that following indomethacin administration, the early inhibition of NO synthase by L-NAME leads to acute microvascular injury in the rat intestine, indicating a protective role of NO formed by cnos in the intestine. Aminoguanidine is known to prevent the intestinal ulceration induced by indomethacin (Tanaka et al., 1999; Takeuchi et al., 2002). In the present study, this agent also prevented the intestinal damage induced by L-NAME plus rofecoxib, suggesting the involvement of inos/no in the pathogenesis of these lesions. At present, the exact mechanism by which COX-2 expression is up-regulated following inhibition of L-NAME or SC- 560 remains unknown. Because the expression of COX-2 induced by L-NAME was mitigated by pretreatment with L-arginine, it is likely that the process is associated with inhibition of NO production. Interestingly, the up-regulation of COX-2 expression and PGE 2 production was also attenuated by atropine at the dose that inhibited intestinal hypermotility in response to L-NAME, suggesting a relationship between the hypermotility and COX-2 expression. Since inhibition of intestinal hypermotility resulted in suppression of bacterial invasion in the mucosa (Takeuchi et al., 2002), and enterobacteria induce inos expression through release of endotoxin [lipopolysaccharide (LPS)] (Boughton-Smith et al., 1993), it is possible that COX-2 expression is up-regulated by LPS, similar to inos expression. In a preliminary study, we observed that the expression of COX-2 following SC-560 treatment was attenuated by ampicillin due to sterilizing action, suggesting a close relationship between COX-2 expression and enterobacteria (Tanaka et al., 2004). As shown in the present study, both L-NAME and SC-560 enhanced bacterial invasion in the intestinal mucosa. Thus, it is possible that COX-2 expression is up-reguated by LPS under inhibition of the production of NO or PGs. These results strongly support the idea that the expression of COX-2 induced by L-NAME is associated with hypermotility and bacterial invasion. This idea was further supported by the fact that the intestinal ulcerogenic response to L-NAME plus rofecoxib was also significantly prevented by prior administration of ampicillin as well as aminoguanidine. It is believed that NO produced by cnos contributes to maintaining the mucosal integrity of the gastrointestinal tract by modulating various functions, such as mucosal blood flow and mucus secretion (Whittle, 1994). The mucus layer acts as a barrier to bacterial invasion in the mucosa, the major pathogenetic event in NSAID-induced intestinal damage (Tanaka et al., 2001, 2002c; Kunikata et al., 2002). We observed in the present study that intestinal mucus secretion was decreased by L-NAME as well as SC-560, suggesting a role for NO in the secretion, similar to PGs. It is assumed that endogenous NO as well as PGs hamper the enterobacterial invasion in the mucosa by stimulating the secretion of mucus and by increasing the mucus gel thickness. On the other hand, we previously reported an increase of intestinal motility following administration of NSAID and suggested the involvement of a hypermotility response in the pathogenesis of NSAID-induced intestinal damage (Kunikata et al., 2002; Takeuchi et al., 2002; Tanaka et al., 2002c). In the present study, we observed that intestinal motility was increased by both SC-560 and L-NAME but not aminoguanidine, again with a relation between this response and inhibition of COX-1 and cnos. Abnormal hypermotility caused
7 by NSAID may disrupt the mucus layer over the epithelium, promoting enterobacterial invasion. In conclusion, the present study suggests that intestinal damage occurs by the inhibition of both cnos and COX-2 as well as both COX-1 and COX-2. The inhibition of NO production by L-NAME caused an increase in intestinal motility and a decrease in intestinal mucus secretion, resulting in enhancement of bacterial invasion, the events similar to those observed under conditions of PG deficiency caused by SC-560 through COX-1 inhibition; however, inhibition of cnos or COX-1 up-regulates the expression of COX-2 through intestinal hypermotility, and this may counteract the deleterious events caused by endogenous NO or PG deficiency. Although cnos exists in two isoforms, endothelial NOS and neuronal NOS, it remains unknown at present which type of cnos contribute to the integrity of small intestinal mucosa. Finally, the present findings strongly suggest a role for COX-2 in maintaining the mucosal integrity of the small intestine. References Azumi Y, Ohara S, Ishihara K, Okabe H, and Hotta K (1981) Correlation of quantitative changes of gastric mucosal glycoproteins with aspirin-induced gastric damage in rats. Gut 21: Boughton-Smith N, Evans SM, Laszlo F, Whittle BJR, and Moncada S (1993) The induction of nitric oxide synthase and intestinal vascular permeability by endotoxin in the rat. Br J Pharmacol 110: Chan CC, Boyce S, Bridear C, Ford-Hutchinson W, Gordon R, Guay D, Li CS, Mancini J, Panneton M, Prasit P, et al. (1995) Pharmacology of a selective cyclooxygenase-2 inhibitor L-745,337: a novel nonsteroidal anti-inflammatory agent with an ulcerogenic sparing effect in rat and non-human primate stomach. J Pharmacol Exp Ther 274: Feng L, Sun W, Xia Y, Tang WW, Chanmugam P, Soyoola E, Wilson CB, and Hwang D (1993) Cloning two isoforms of rat cyclooxygenase: differential regulation of their expression. Arch Biochem Biophys 307: Futaki N, Takahashi S, Yokoyama M, Arai I, Higuchi S, and Otomo S (1994) NS-398, a new antiinflammatory agent, selectively inhibits prostaglandin G/H synthase/ cyclo-oxygenase (COX-2) activity in vitro. Prostaglandins 47: Iso JY, Sun X-H, Kao T-H, Reece KS, and Wu R (1995) Isolation and characterization of rat and human glyceraldehyde-3-phosphate dehydro-genase cdnas: genomic complexity and molecular evolution of the gene. Nucleic Acids Res 13: Kunikata T, Tanaka A, Miyazawa T, Kato S, and Takeuchi K (2002) 16,16-dimethyl prostaglandin E2 inhibits indomethacin-induced small intestinal lesions through EP3 and EP4 receptors. Dig Dis Sci 47: Lanza FL (1984) Endoscopic studies of gastric and duodenal injury after the use of Intestinal Damage Induced by L-NAME plus Rofecoxib 827 ibuprofen, aspirin and other nonsteroidal antiinflammatory agents. Am J Med 13: Laszlo F, Whittle BJR, and Moncada S (1994) Time-dependent enhancement and inhibition of endotoxin-induced vascular injury in rat intestine by nitric oxide synthase inhibitors. Br J Pharmacol 111: O Neill GP and Ford-Hutchinson AW (1993) Expression of mrna for cyclooxygenase-1 and cyclooxygenase-2 in human tissues. FEBS Lett 330: Reuter BK, Davies NM, and Wallace JL (1997) Nonsteroidal anti-inflammatory drug enteropathy in rats: role of permeability, bacteria and enterohepatic circulation. Gastroenterology 112: Smith CJ, Zhang Y, Koboldt CM, Muhammad J, Zweifel BS, Shaffer A, Talley JJ, Masferrer JL, Seibert K, and Isakson PC (1998) Pharmacological analysis of cyclooxygenase-1 in inflammation. Proc Natl Acad Sci USA 95: Takeuchi K, Miyazawa T, Tanaka A, Kato S, and Kunikata T (2002) Pathogenic importance of intestinal hypermotility in NSAID-induced small intestinal damage in rats. Digestion 66: Takeuchi K, Ohuchi T, Tachibana M, and Okabe S (1994) Mechanisms underlying stimulation of gastric HCO3- secretion by NG-nitro-L-arginine methyl ester, an inhibitor of nitric oxide synthase in rats. J Gastroenterol Hepatol 9:S50 S54. Takuechi K, Ueki S, and Okabe S (1986) Importance of gastric motility in the pathogenesis of indomethacin-induced gastric lesions in rats. Dig Dis Sci 31: Tanaka A, Araki H, Hase S, and Takeuchi K (2002a) Roles of COX-1 and COX-2 inhibition in nonsteroidal antiinflammatory drug-induced gastric damage in rats: relation to functional responses. Aliment Pharmacol Ther 16: Tanaka A, Hase S, Miyazawa T, and Takeuchi K (2002b) Up-regulation of COX-2 by inhibition of COX-1: A key to NSAID-Induced intestinal damage. J Pharmacol Exp Ther 300: Tanaka A, Hase S, Ohno R, and Takeuchi K (2002c) Roles of COX-1 and COX-2 inhibition in NSAID-induced intestinal damage in rats. J Pharmacol Exp Ther 303: Tanaka A, Kunikata T, Konaka A, Kato S, and Takeuchi K (1999) Dual action of nitric oxide in pathogenesis of indomethacin-induced small intestinal ulceration in rats. J Physiol Pharmacol 50: Tanaka A, Mizoguchi H, Kunikata T, Miyazawa T, and Takeuchi K (2001) Protection by constitutively formed nitric oxide of intestinal damage induced by indomethacin in rats. J Physiol Paris 95: Tanaka A, Ohno R, Hase S, and Takeuchi K (2004) Functional mechanism underlying up-regulation of COX-2 in rat small intestine under inhibition of COX-1. J Gastroenterol Hepatol (in press) Wallace JL, McKnight W, Reuter BK, and Vergnolle N (2000) NSAID-induced gastric damage in rats: requirement for inhibition of both cyclooxygenase 1 and 2. Gastroenterology 119: Whittle BJR (1981) Temporal relationship between cyclooxygenase inhibition, as measured by prostacyclin biosynthesis and the gastrointestinal damage induced by indomethacin in the rat. Gastroenterology 80: Whittle BJR (1994) Nitric oxide in gastrointestinal physiology and pathology, in Physiology of the Gastrointestinal Tract (Johnson LR ed) ed 3, pp , Raven Press, New York. Address correspondence to: Dr. Koji Takeuchi, Department of Pharmacology and Experimental Therapeutics, Kyoto Pharmaceutical University, Misasagi, Yamashina, Kyoto 607, Japan. takeuchi@mb.kyoto-phu.ac.jp
Augmentation of Cysteamine and Mepirizole-Induced Lesions in the Rat Duodenum and Stomach by Histamine or Indomethacin
 Augmentation of Cysteamine and Mepirizole-Induced Lesions in the Rat Duodenum and Stomach by Histamine or Indomethacin Hironori TANAKA, Yoshimi KUWAHARA and Susumu OKABE Department of Applied Pharmacology,
Augmentation of Cysteamine and Mepirizole-Induced Lesions in the Rat Duodenum and Stomach by Histamine or Indomethacin Hironori TANAKA, Yoshimi KUWAHARA and Susumu OKABE Department of Applied Pharmacology,
Adaptive Gastric Cytoprotection Is Mediated by Prostaglandin EP1 Receptors: A Study Using Rats and Knockout Mice
 0022-3565/01/2973-1160 1165$3.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 297, No. 3 Copyright 2001 by The American Society for Pharmacology and Experimental Therapeutics 3543/904882
0022-3565/01/2973-1160 1165$3.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 297, No. 3 Copyright 2001 by The American Society for Pharmacology and Experimental Therapeutics 3543/904882
NSAID-Induced Small Intestinal Damage Roles of Various Pathogenic Factors
 Invited Review Digestion 215;91:218 232 DOI: 1.1159/37416 Published online: March 18, 215 NSAID-Induced Small Intestinal Damage Roles of Various Pathogenic Factors Koji Takeuchi a, b Hiroshi Satoh b, c
Invited Review Digestion 215;91:218 232 DOI: 1.1159/37416 Published online: March 18, 215 NSAID-Induced Small Intestinal Damage Roles of Various Pathogenic Factors Koji Takeuchi a, b Hiroshi Satoh b, c
Roles of Endogenous Prostaglandins and Cyclooxygenase Isozymes in Healing of Indomethacin-Induced Small Intestinal Lesions in Rats
 0022-3565/06/3182-691 699$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 318, No. 2 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 103994/3127801
0022-3565/06/3182-691 699$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 318, No. 2 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 103994/3127801
Role of Cyclooxygenase-2 in the Healing of Gastric Ulcers in Rats
 0022-3565/98/2863-1383$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 286, No. 3 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/98/2863-1383$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 286, No. 3 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Effects of NSAIDs on Bladder Function in Normal and Cystitis Rats: a Comparison Study of Aspirin, Indomethacin, and Ketoprofen
 J Pharmacol Sci 95, 458 465 (2004) Journal of Pharmacological Sciences 2004 The Japanese Pharmacological Society Full Paper Effects of NSAIDs on Bladder Function in Normal and Cystitis Rats: a Comparison
J Pharmacol Sci 95, 458 465 (2004) Journal of Pharmacological Sciences 2004 The Japanese Pharmacological Society Full Paper Effects of NSAIDs on Bladder Function in Normal and Cystitis Rats: a Comparison
ulcer healing role 118 Bicarbonate, prostaglandins in duodenal cytoprotection 235, 236
 Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
Subject Index Actin cellular forms 48, 49 epidermal growth factor, cytoskeletal change induction in mucosal repair 22, 23 wound repair 64, 65 polyamine effects on cytoskeleton 49 51 S-Adenosylmethionine
Delayed Healing of Acetic Acid-Induced Gastric Ulcers in Rats by Indomethacin
 GASTROENTEROLOGY 1989;96:393-402 Delayed Healing of Acetic Acid-Induced Gastric Ulcers in Rats by Indomethacin JIAN YING WANG, SHINICHI YAMASAKI, KGJI TAKEUCHI, and SUSUMU GKABE Department of Applied Pharmacology,
GASTROENTEROLOGY 1989;96:393-402 Delayed Healing of Acetic Acid-Induced Gastric Ulcers in Rats by Indomethacin JIAN YING WANG, SHINICHI YAMASAKI, KGJI TAKEUCHI, and SUSUMU GKABE Department of Applied Pharmacology,
JOURNAL OF PHYSIOLOGY AND PHARMACOLOGY 2013, 64, 1, K. AMAGASE, Y. YOSHIDA, D. HARA, T. MURAKAMI, K.
 JOURNAL OF PHYSIOLOGY AND PHARMACOLOGY 2013, 64, 1, 65-75 www.jpp.krakow.pl K. AMAGASE, Y. YOSHIDA, D. HARA, T. MURAKAMI, K. TAKEUCHI PROPHYLACTIC EFFECT OF EGUALEN SODIUM, A STABLE AZULENE DERIVATIVE,
JOURNAL OF PHYSIOLOGY AND PHARMACOLOGY 2013, 64, 1, 65-75 www.jpp.krakow.pl K. AMAGASE, Y. YOSHIDA, D. HARA, T. MURAKAMI, K. TAKEUCHI PROPHYLACTIC EFFECT OF EGUALEN SODIUM, A STABLE AZULENE DERIVATIVE,
Effect of taurine on ulcerogenic response and impaired ulcer healing induced by monochloramine in rat stomachs
 Aliment Pharmacol Ther 22; 16 (Suppl. 2): 35 43. Effect of taurine on ulcerogenic response and impaired ulcer healing induced by monochloramine in rat stomachs S. KATO, M. UMEDA, M. TAKEEDA, K. KANATSU
Aliment Pharmacol Ther 22; 16 (Suppl. 2): 35 43. Effect of taurine on ulcerogenic response and impaired ulcer healing induced by monochloramine in rat stomachs S. KATO, M. UMEDA, M. TAKEEDA, K. KANATSU
Research Article Animal Model of Acid-Reflux Esophagitis: Pathogenic Roles of Acid/Pepsin, Prostaglandins, and Amino Acids
 Hindawi Publishing Corporation BioMed Research International Volume 214, Article ID 532594, 1 pages http://dx.doi.org/1.1155/214/532594 Research Article Animal Model of Acid-Reflux Esophagitis: Pathogenic
Hindawi Publishing Corporation BioMed Research International Volume 214, Article ID 532594, 1 pages http://dx.doi.org/1.1155/214/532594 Research Article Animal Model of Acid-Reflux Esophagitis: Pathogenic
Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall
 Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall DAY ONE All incubations are done at room temperature unless otherwise noted. All solutions and all containers
Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall DAY ONE All incubations are done at room temperature unless otherwise noted. All solutions and all containers
JPET # Cyclooxygenase (COX)-1 and COX-2 both play an important role in the protection of the
 JPET Fast This Forward. article has not Published been copyedited on and September formatted. The 24, final 2012 version as may DOI:10.1124/jpet.112.199182 differ from this version. Title page. Title:
JPET Fast This Forward. article has not Published been copyedited on and September formatted. The 24, final 2012 version as may DOI:10.1124/jpet.112.199182 differ from this version. Title page. Title:
Exacerbation of Nonsteroidal Anti-Inflammatory Drug-Induced Small Intestinal Lesions by Antisecretory Drugs in Rats: The Role of Intestinal Motility S
 1521-0103/12/3432-270 277$25.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 343, No. 2 Copyright 2012 by The American Society for Pharmacology and Experimental Therapeutics 197475/3799140
1521-0103/12/3432-270 277$25.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 343, No. 2 Copyright 2012 by The American Society for Pharmacology and Experimental Therapeutics 197475/3799140
STUDIES ON THE EXPERIMENTAL GASTROINTESTINAL ULCERS PRODUCED BY RESERPINE AND STRESS II. ULCEROGENIC ACTIVITIES OF RESERPINE AND ITS ANALOGUES
 STUDIES ON THE EXPERIMENTAL GASTROINTESTINAL ULCERS PRODUCED BY RESERPINE AND STRESS II. ULCEROGENIC ACTIVITIES OF RESERPINE AND ITS ANALOGUES MASAMI DOTEUCHI Division of Neuropharmacology, Shionogi Research
STUDIES ON THE EXPERIMENTAL GASTROINTESTINAL ULCERS PRODUCED BY RESERPINE AND STRESS II. ULCEROGENIC ACTIVITIES OF RESERPINE AND ITS ANALOGUES MASAMI DOTEUCHI Division of Neuropharmacology, Shionogi Research
Role of Cyclooxygenase-1 and -2, Phospholipase C, and Protein Kinase C in Prostaglandin-Mediated Gastroprotection
 0022-3565/03/3053-1233 1238$7.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 305, No. 3 Copyright 2003 by The American Society for Pharmacology and Experimental Therapeutics 49650/1065399
0022-3565/03/3053-1233 1238$7.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 305, No. 3 Copyright 2003 by The American Society for Pharmacology and Experimental Therapeutics 49650/1065399
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
EFFECT OF HISTAMINE, BRADYKININ AND MORPHINE ON ADRENALINE RELEASE FROM RAT ADRENAL GLAND
 EFFECT OF HISTAMINE, BRADYKININ AND MORPHINE ON ADRENALINE RELEASE FROM RAT ADRENAL GLAND Toshio YOSHIZAKI Shionogi Research Laboratory, Shionogi & Co., Ltd., Fukushima-ku, Osaka, 553 Japan Accepted March
EFFECT OF HISTAMINE, BRADYKININ AND MORPHINE ON ADRENALINE RELEASE FROM RAT ADRENAL GLAND Toshio YOSHIZAKI Shionogi Research Laboratory, Shionogi & Co., Ltd., Fukushima-ku, Osaka, 553 Japan Accepted March
Gastrointestinal Safety of Coxibs and Outcomes Studies: What s the Verdict?
 Vol. 23 No. 4S April 2002 Journal of Pain and Symptom Management S5 Proceedings from the Symposium The Evolution of Anti-Inflammatory Treatments in Arthritis: Current and Future Perspectives Gastrointestinal
Vol. 23 No. 4S April 2002 Journal of Pain and Symptom Management S5 Proceedings from the Symposium The Evolution of Anti-Inflammatory Treatments in Arthritis: Current and Future Perspectives Gastrointestinal
Short Communication. Abstract. Introduction
 Short Communication JPP, 6: 76 7 The Authors JPP Royal Pharmaceutical Society Received November 8, Accepted February, DOI./j.-758..76.x ISSN -57 Relationship between lipophilicity and absorption from the
Short Communication JPP, 6: 76 7 The Authors JPP Royal Pharmaceutical Society Received November 8, Accepted February, DOI./j.-758..76.x ISSN -57 Relationship between lipophilicity and absorption from the
Evaluation of firocoxib inhibition of cyclooxygenase activity in alpacas. by Abigail Chadwick A THESIS. submitted to. Oregon State University
 Evaluation of firocoxib inhibition of cyclooxygenase activity in alpacas by Abigail Chadwick A THESIS submitted to Oregon State University Honors College in partial fulfillment of the requirements for
Evaluation of firocoxib inhibition of cyclooxygenase activity in alpacas by Abigail Chadwick A THESIS submitted to Oregon State University Honors College in partial fulfillment of the requirements for
Single Cell Quantitative Polymer Chain Reaction (sc-qpcr)
 Single Cell Quantitative Polymer Chain Reaction (sc-qpcr) Analyzing gene expression profiles from a bulk population of cells provides an average profile which may obscure important biological differences
Single Cell Quantitative Polymer Chain Reaction (sc-qpcr) Analyzing gene expression profiles from a bulk population of cells provides an average profile which may obscure important biological differences
Copyright 2012 by the American Society for Pharmacology and Experimental Therapeutics.
 JPET Fast This article Forward. has not been Published copyedited on and August formatted. 1, The 2012 final version as DOI:10.1124/jpet.112.197475 may differ from this version. 1. Title page. Title: Exacerbation
JPET Fast This article Forward. has not been Published copyedited on and August formatted. 1, The 2012 final version as DOI:10.1124/jpet.112.197475 may differ from this version. 1. Title page. Title: Exacerbation
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature
 PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
Manual (Second edition)
 Reagent for RNA Extraction ISOGENⅡ Manual (Second edition) Code No. 311-07361 Code No. 317-07363 NIPPON GENE CO., LTD. Table of contents I Product description 1 II Product content 1 III Storage 1 IV Precautions
Reagent for RNA Extraction ISOGENⅡ Manual (Second edition) Code No. 311-07361 Code No. 317-07363 NIPPON GENE CO., LTD. Table of contents I Product description 1 II Product content 1 III Storage 1 IV Precautions
310 bp. 887 bp. 702 bp. 310 bp. 887 bp. 702 bp
 74).2;51/;),2)4)+/;!#"5KFF"$#`& MMMFFH=MF 6)-7+1)6)))4);6) 4-.+:1116112)6/--515.5)1,1,7+-, 5)16-561),))/-,AF=HJAJB2D=H=?CO=@-NFAHEAJ=6DAH=FAKJE?I OJ2D=H=?AKJE?=7ELAHIEJOEI=I=CE;==IDE=OJ=F= IJAHE@==JEEB==JHO@HKCI5)1,IIK?D=IE@AJD=?E@A?HA=IA
74).2;51/;),2)4)+/;!#"5KFF"$#`& MMMFFH=MF 6)-7+1)6)))4);6) 4-.+:1116112)6/--515.5)1,1,7+-, 5)16-561),))/-,AF=HJAJB2D=H=?CO=@-NFAHEAJ=6DAH=FAKJE?I OJ2D=H=?AKJE?=7ELAHIEJOEI=I=CE;==IDE=OJ=F= IJAHE@==JEEB==JHO@HKCI5)1,IIK?D=IE@AJD=?E@A?HA=IA
Prostaglandins And Other Biologically Active Lipids
 Prostaglandins And Other Biologically Active Lipids W. M. Grogan, Ph.D. OBJECTIVES After studying the material of this lecture, the student will: 1. Draw the structure of a prostaglandin, name the fatty
Prostaglandins And Other Biologically Active Lipids W. M. Grogan, Ph.D. OBJECTIVES After studying the material of this lecture, the student will: 1. Draw the structure of a prostaglandin, name the fatty
Pharmacological analysis of cyclooxygenase-1 in inflammation
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 13313 13318, October 1998 Pharmacology Pharmacological analysis of cyclooxygenase-1 in inflammation CHRISTOPHER J. SMITH, YAN ZHANG, CAROL M. KOBOLDT, JERRY MUHAMMAD,
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 13313 13318, October 1998 Pharmacology Pharmacological analysis of cyclooxygenase-1 in inflammation CHRISTOPHER J. SMITH, YAN ZHANG, CAROL M. KOBOLDT, JERRY MUHAMMAD,
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A. Real-Time RT-PCR Detection.
 For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
Endothelium. A typical endothelial cell is about 30mm long, Accounts for 1% or less of the arterial weight
 Endothelium Discovered in 1845 A typical endothelial cell is about 30mm long, 10mm wide, and 0.2 3 mm thick Accounts for 1% or less of the arterial weight As recently as the late 1960s it was thought of
Endothelium Discovered in 1845 A typical endothelial cell is about 30mm long, 10mm wide, and 0.2 3 mm thick Accounts for 1% or less of the arterial weight As recently as the late 1960s it was thought of
Effect of rebamipide on gastric bleeding and ulcerogenic responses induced by aspirin plus clopidogrel under stimulation of acid secretion in rats
 bs_bs_banner doi:10.1111/jgh.12774 ORIGINAL ARTICLE Effect of rebamipide on gastric bleeding and ulcerogenic responses induced by aspirin plus clopidogrel under stimulation of acid secretion in rats Koji
bs_bs_banner doi:10.1111/jgh.12774 ORIGINAL ARTICLE Effect of rebamipide on gastric bleeding and ulcerogenic responses induced by aspirin plus clopidogrel under stimulation of acid secretion in rats Koji
PREVENTION BY LANSOPRAZOLE, A PROTON PUMP INHIBITOR, OF INDOMETHACIN-INDUCED SMALL INTESTINAL ULCERATION IN RATS THROUGH INDUCTION OF HEME OXYGENASE-1
 JOURNAL OF PHYSIOLOGY AND PHARMACOLOGY 2010, 61, 3, 287-294 www.jpp.krakow.pl Y. YODA 1, K. AMAGASE 2, S. KATO 2, S. TOKIOKA 1, M. MURANO 1, K. KAKIMOTO 1, H. NISHIO 2, E. UMEGAKI 1, K. TAKEUCHI 2, K.
JOURNAL OF PHYSIOLOGY AND PHARMACOLOGY 2010, 61, 3, 287-294 www.jpp.krakow.pl Y. YODA 1, K. AMAGASE 2, S. KATO 2, S. TOKIOKA 1, M. MURANO 1, K. KAKIMOTO 1, H. NISHIO 2, E. UMEGAKI 1, K. TAKEUCHI 2, K.
22 Bicozamycin (Bicyclomycin)
 22 Bicozamycin (Bicyclomycin) OH O H N O O OH HO [Summary of bicozamycin] C 12 H 18 N 2 O 7 MW: 302.3 CAS No.: 38129-37-2 Bicozamycin (BZM) is an antibiotic obtained from a fermented culture of Streptomyces
22 Bicozamycin (Bicyclomycin) OH O H N O O OH HO [Summary of bicozamycin] C 12 H 18 N 2 O 7 MW: 302.3 CAS No.: 38129-37-2 Bicozamycin (BZM) is an antibiotic obtained from a fermented culture of Streptomyces
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
EFFECTS OF L-ARIGININE SUPPLEMENTATION ON INDOMETHACIN-INDUCED GASTRIC ULCERATION IN RATS
 EFFECTS OF L-ARIGININE SUPPLEMENTATION ON INDOMETHACIN-INDUCED GASTRIC ULCERATION IN RATS 1 AJIBOYE Kolawole I., 2 OLUWOLE Francis S. 3 AJIBOYE Oyebimpe F 1 Department of Physiology, Benjamin S Carson
EFFECTS OF L-ARIGININE SUPPLEMENTATION ON INDOMETHACIN-INDUCED GASTRIC ULCERATION IN RATS 1 AJIBOYE Kolawole I., 2 OLUWOLE Francis S. 3 AJIBOYE Oyebimpe F 1 Department of Physiology, Benjamin S Carson
Ghrelin and Stress Protection
 Ghrelin and Stress Protection 3 Genetics of Stress See: Genetic Factors and Stress; Genetic Predispositions to Stressful Conditions; Gene Environment Interactions in Early Development; Genetic Variation
Ghrelin and Stress Protection 3 Genetics of Stress See: Genetic Factors and Stress; Genetic Predispositions to Stressful Conditions; Gene Environment Interactions in Early Development; Genetic Variation
Japan. J. Pharmacol. 34, (1984) 89
 Japan. J. Pharmacol. 34, 89-94 (1984) 89 Effect of an Anti-Ulcer Agent, 2'-Carboxymethoxy-4, 4'-Bis (3-Methyl-2-Butenyloxy) Chalcone (SU-88), on the Biosynthesis of Gastric Sulfated Mucosubstances in Restrained
Japan. J. Pharmacol. 34, 89-94 (1984) 89 Effect of an Anti-Ulcer Agent, 2'-Carboxymethoxy-4, 4'-Bis (3-Methyl-2-Butenyloxy) Chalcone (SU-88), on the Biosynthesis of Gastric Sulfated Mucosubstances in Restrained
COX Activity Assay Kit. Item No
 COX Activity Assay Kit Item. 760151 TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions 4 If You ave Problems 4 Storage and Stability 4 Materials Needed but t Supplied INTRODUCTION
COX Activity Assay Kit Item. 760151 TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions 4 If You ave Problems 4 Storage and Stability 4 Materials Needed but t Supplied INTRODUCTION
PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID
 GASTROENTEROLOGY Copyriht 1972 by The Williams & Wilkins Co. Vol. 62. No. 3 Printed in U.S. A. PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID LEONARD R. JOHNSON, PH.D. Department of Physiology
GASTROENTEROLOGY Copyriht 1972 by The Williams & Wilkins Co. Vol. 62. No. 3 Printed in U.S. A. PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID LEONARD R. JOHNSON, PH.D. Department of Physiology
ASSAY OF SPHINGOMYELINASE ACTIVITY
 ASSAY OF SPHINGOMYELINASE ACTIVITY Protocol for Protein Extraction Stock Solution 1. Leupeptin/hydrochloride (FW 463.0,
ASSAY OF SPHINGOMYELINASE ACTIVITY Protocol for Protein Extraction Stock Solution 1. Leupeptin/hydrochloride (FW 463.0,
Effects and mechanisms of Fenofibrate on the secretion of vascular endothelial contraction factors in hypertensive rats
 Effects and mechanisms of Fenofibrate on the secretion of vascular endothelial contraction factors in hypertensive rats Y. Zhu 1, H.-S. Wang 1, X.-M. Li 1 and C. Qu 2 1 Department of Cardiac Surgery, General
Effects and mechanisms of Fenofibrate on the secretion of vascular endothelial contraction factors in hypertensive rats Y. Zhu 1, H.-S. Wang 1, X.-M. Li 1 and C. Qu 2 1 Department of Cardiac Surgery, General
Enzyme Immunoassay for
 Enzyme Immunoassay for Prostaglandin E 2 For Research Use Only INTRODUCTION Prostaglandin E 2 EIA Kit Product Number: EA02 Store at 4 C FOR RESEARCH USE ONLY Document Control Number: EA02.120214 Page 1
Enzyme Immunoassay for Prostaglandin E 2 For Research Use Only INTRODUCTION Prostaglandin E 2 EIA Kit Product Number: EA02 Store at 4 C FOR RESEARCH USE ONLY Document Control Number: EA02.120214 Page 1
Product Manual. Omni-Array Sense Strand mrna Amplification Kit, 2 ng to 100 ng Version Catalog No.: Reactions
 Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
CHLORPROMAZINE IN RATS GIVEN TRIHEXYPHENIDYL
 Br. J. Pharmac. (1976), 56, 301-305 IMPAIRED ABSORPTION OF CHLORPROMAZINE IN RATS GIVEN TRIHEXYPHENIDYL LEONOR RIVERA-CALIMLIM Department of Pharmacology and Toxicology, University of Rochester, School
Br. J. Pharmac. (1976), 56, 301-305 IMPAIRED ABSORPTION OF CHLORPROMAZINE IN RATS GIVEN TRIHEXYPHENIDYL LEONOR RIVERA-CALIMLIM Department of Pharmacology and Toxicology, University of Rochester, School
Pinpoint Slide RNA Isolation System II Catalog No. R1007
 INSTRUCTION MANUAL Pinpoint Slide RNA Isolation System II Catalog No. R1007 Highlights Allows for the isolation of total RNA from paraffin-embedded tissue sections on glass slides Simple procedure combines
INSTRUCTION MANUAL Pinpoint Slide RNA Isolation System II Catalog No. R1007 Highlights Allows for the isolation of total RNA from paraffin-embedded tissue sections on glass slides Simple procedure combines
Aspirin antagonism in isonizaid treatment of tuberculosis in mice ACCEPTED. Department of Molecular Microbiology & Immunology, Bloomberg School of
 AAC Accepts, published online ahead of print on 4 December 2006 Antimicrob. Agents Chemother. doi:10.1128/aac.01145-06 Copyright 2006, American Society for Microbiology and/or the Listed Authors/Institutions.
AAC Accepts, published online ahead of print on 4 December 2006 Antimicrob. Agents Chemother. doi:10.1128/aac.01145-06 Copyright 2006, American Society for Microbiology and/or the Listed Authors/Institutions.
Rimoxen. Rimoxen! "The Gastro and Cardio friendly anti-inflammatory ingredient"
 Rimoxen A Botanical Anti-Inflammatory COX-2 Selective Ingredient Question: How doses Rimoxen compare to the prescription COX-2 inhibitors (Vioxx and Celebrex) and the OTC NSAID pain relievers such as aspirin
Rimoxen A Botanical Anti-Inflammatory COX-2 Selective Ingredient Question: How doses Rimoxen compare to the prescription COX-2 inhibitors (Vioxx and Celebrex) and the OTC NSAID pain relievers such as aspirin
SCFA in gut health. Kristin Verbeke. On behalf of the Prebiotic Task Force of ILSI Europe
 SCFA in gut health Kristin Verbeke Translational Research in Gastrointestinal Disorders KU Leuven On behalf of the Prebiotic Task Force of ILSI Europe Acetic acid Major anions in the large intestine Propionic
SCFA in gut health Kristin Verbeke Translational Research in Gastrointestinal Disorders KU Leuven On behalf of the Prebiotic Task Force of ILSI Europe Acetic acid Major anions in the large intestine Propionic
Study of the main chemical components of Ganoderma lucidum
 Study of the main chemical components of Ganoderma lucidum Yasuo Komota et al Tokyo Medical and Dental University [Purpose] As part of the means for exerting quality control on Ganoderma lucidum 50% ethanol
Study of the main chemical components of Ganoderma lucidum Yasuo Komota et al Tokyo Medical and Dental University [Purpose] As part of the means for exerting quality control on Ganoderma lucidum 50% ethanol
Capsaicin (3.3 mmol/l)
 A 18 16 14 1 8 6 4 Vehicle BCTC (.8 mmol/l) Sensory deafferentation Time (min) Capsaicin (3.3 mmol/l) N=3 4 * P
A 18 16 14 1 8 6 4 Vehicle BCTC (.8 mmol/l) Sensory deafferentation Time (min) Capsaicin (3.3 mmol/l) N=3 4 * P
Hepatitis B Virus Genemer
 Product Manual Hepatitis B Virus Genemer Primer Pair for amplification of HBV Viral Specific Fragment Catalog No.: 60-2007-10 Store at 20 o C For research use only. Not for use in diagnostic procedures
Product Manual Hepatitis B Virus Genemer Primer Pair for amplification of HBV Viral Specific Fragment Catalog No.: 60-2007-10 Store at 20 o C For research use only. Not for use in diagnostic procedures
EICOSANOID METABOLISM
 1 EICOSANOID METABOLISM EICOSANOIDS C20 polyunsaturated fatty acids e.g. Arachidonic acid Eicosanoids physiologically, pathologically and pharmacologically active compounds PG Prostaglandins TX - Thromboxanes
1 EICOSANOID METABOLISM EICOSANOIDS C20 polyunsaturated fatty acids e.g. Arachidonic acid Eicosanoids physiologically, pathologically and pharmacologically active compounds PG Prostaglandins TX - Thromboxanes
Study of the main chemical components of Ganoderma lucidum
 Study of the main chemical components of Ganoderma lucidum Yasuo Komota et al Tokyo Medical and Dental University [Purpose] As part of the means for exerting quality control on Ganoderma lucidum 50% ethanol
Study of the main chemical components of Ganoderma lucidum Yasuo Komota et al Tokyo Medical and Dental University [Purpose] As part of the means for exerting quality control on Ganoderma lucidum 50% ethanol
ETIOPATHOGENICAL ASPECTS OF NONSTEROIDAL ANTI- INFLAMMATORY DRUGS IN ELDERLY Mihaela Bursova¹, Gabriela Lilios², Maria Suta³, Gh.
 ETIOPATHOGENICAL ASPECTS OF NONSTEROIDAL ANTI- INFLAMMATORY DRUGS IN ELDERLY Mihaela Bursova¹, Gabriela Lilios², Maria Suta³, Gh. Taralunga² 1.CLINICAL EMERGENCY COUNTY HOSPITAL CONSTANŢA, GERIATRICS DEPARTAMENT
ETIOPATHOGENICAL ASPECTS OF NONSTEROIDAL ANTI- INFLAMMATORY DRUGS IN ELDERLY Mihaela Bursova¹, Gabriela Lilios², Maria Suta³, Gh. Taralunga² 1.CLINICAL EMERGENCY COUNTY HOSPITAL CONSTANŢA, GERIATRICS DEPARTAMENT
Subject Index. ATP, see Mucosal energy metabolism Autophagy gastric adenocarcinoma 208, 209 gastric carcinoid
 Subject Index Acetic acid ulcer model gastric cancer removal 38, 39 healing studies 33 36 overview 32, 33 treatment studies 36 38 Achillea millefolium, acetic acid ulcer Adrenergic receptors, gastroprotection
Subject Index Acetic acid ulcer model gastric cancer removal 38, 39 healing studies 33 36 overview 32, 33 treatment studies 36 38 Achillea millefolium, acetic acid ulcer Adrenergic receptors, gastroprotection
Name: Class: "Pharmacology NSAIDS (1) Lecture
 I Name: Class: "Pharmacology NSAIDS (1) Lecture د. احمد الزهيري Inflammation is triggered by the release of chemical mediators from injured tissues and migrating cells. The specific mediators vary with
I Name: Class: "Pharmacology NSAIDS (1) Lecture د. احمد الزهيري Inflammation is triggered by the release of chemical mediators from injured tissues and migrating cells. The specific mediators vary with
Supplemental methods. Total RNA was extracted from the stomach, liver, pancreas, pituitary, and
 Supplemental methods Real-time quantitative RT-PCR and Semi-quantitative PCR Total RNA was extracted from the stomach, liver, pancreas, pituitary, and hypothalamus as previously described (). Primers and
Supplemental methods Real-time quantitative RT-PCR and Semi-quantitative PCR Total RNA was extracted from the stomach, liver, pancreas, pituitary, and hypothalamus as previously described (). Primers and
Yasunori Nagaki, Seiji Hayasaka, Chiharu Kadoi, Nobuo Nakamura and Yoriko Hayasaka
 Effects of Scutellariae Radix Extract and its Components (Baicalein, Baicalin, and Wogonin) on the Experimental of in Pigmented Yasunori Nagaki, Seiji Hayasaka, Chiharu Kadoi, Nobuo Nakamura and Yoriko
Effects of Scutellariae Radix Extract and its Components (Baicalein, Baicalin, and Wogonin) on the Experimental of in Pigmented Yasunori Nagaki, Seiji Hayasaka, Chiharu Kadoi, Nobuo Nakamura and Yoriko
Beneficial Effects of Vegetable Oils (Rice Bran and Mustard Oils) on Anti-inflammatory and Gastro Intestinal Profiles of Indomethacin in Rats
 Biomedical & Pharmacology Journal Vol. 6(2), 375-379 (2013) Beneficial Effects of Vegetable Oils (Rice Bran and Mustard Oils) on Anti-inflammatory and Gastro Intestinal Profiles of Indomethacin in Rats
Biomedical & Pharmacology Journal Vol. 6(2), 375-379 (2013) Beneficial Effects of Vegetable Oils (Rice Bran and Mustard Oils) on Anti-inflammatory and Gastro Intestinal Profiles of Indomethacin in Rats
PEDV Research Updates 2013
 PEDV Research Updates 2013 Porcine Epidemic Diarrhea virus (PEDV) has caused significant challenges to the swine industry. The virus had not been previously identified in the United States prior to April
PEDV Research Updates 2013 Porcine Epidemic Diarrhea virus (PEDV) has caused significant challenges to the swine industry. The virus had not been previously identified in the United States prior to April
possibility that the "gastric hormone" may not as yet have been extracted investigation of any part of the stomach other than the pyloric mucosa.
 234 6I2.32.014.2I :547x78I.5 HISTAMINE IN CANINE GASTRIC TISSUES. BY GERTRUDE GAVIN, E. W. McHENRY AmD M. J. WILSON. (From the Department of Physiological Hygiene, School of Hygiene, University of Toronto.)
234 6I2.32.014.2I :547x78I.5 HISTAMINE IN CANINE GASTRIC TISSUES. BY GERTRUDE GAVIN, E. W. McHENRY AmD M. J. WILSON. (From the Department of Physiological Hygiene, School of Hygiene, University of Toronto.)
NSAIDs: Side Effects and Guidelines
 NSAIDs: Side Effects and James J Hale FY1 Department of Anaesthetics Introduction The non-steroidal anti-inflammatory drugs (NSAIDs) are a diverse group of drugs that have analgesic, antipyretic and anti-inflammatory
NSAIDs: Side Effects and James J Hale FY1 Department of Anaesthetics Introduction The non-steroidal anti-inflammatory drugs (NSAIDs) are a diverse group of drugs that have analgesic, antipyretic and anti-inflammatory
APPENDIX-I. The compositions of media used for the growth and differentiation of Pseudomonas aeruginosa are as follows:
 APPENDIX-I The compositions of media used for the growth and differentiation of Pseudomonas aeruginosa are as follows: COMPOSITION OF DIFFERENT MEDIA STOCK CULTURE AGAR (AYERS AND JOHNSON AGAR) Gms/Litre
APPENDIX-I The compositions of media used for the growth and differentiation of Pseudomonas aeruginosa are as follows: COMPOSITION OF DIFFERENT MEDIA STOCK CULTURE AGAR (AYERS AND JOHNSON AGAR) Gms/Litre
Effects of Novel Anti-Inflammatory Compounds on Healing of Acetic Acid-Induced Gastric Ulcer in Rats
 0022-3565/98/2871-0301$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 287, No. 1 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/98/2871-0301$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 287, No. 1 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Metabolism of Extremophiles
 1 Microbial Metabolism Metabolism of Extremophiles Ching-Tsan Huang ( 黃慶璨 ) Office: Agronomy Building, Room 111 Tel: (02) 33664454 E-mail: cthuang@ntu.edu.tw Extremophiles 2 Definition Inhabit some of
1 Microbial Metabolism Metabolism of Extremophiles Ching-Tsan Huang ( 黃慶璨 ) Office: Agronomy Building, Room 111 Tel: (02) 33664454 E-mail: cthuang@ntu.edu.tw Extremophiles 2 Definition Inhabit some of
Product Use HPSC-CC are for research use only. It is not approved for human or animal use, or for application in in vitro diagnostic procedures.
 HPSC-derived Cardiomyocyte Cells (HPSC-CC) Catalog #6240 Cell Specification Human primary cardiomyocytes and cardiac tissue are superior modeling systems for heart disease studies, drug discovery and toxicity
HPSC-derived Cardiomyocyte Cells (HPSC-CC) Catalog #6240 Cell Specification Human primary cardiomyocytes and cardiac tissue are superior modeling systems for heart disease studies, drug discovery and toxicity
EFFECT OF CARBENOXOLONE ON THE GASTRIC MUCOSAL BARRIER IN MAN AFTER ADMINISTRATION OF TAUROCHOLIC ACID
 GASTROENTEROLOGY 64: 1101-1105, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64 No.6 Printed in U.S.A. EFFECT OF CARBENOXOLONE ON THE GASTRIC MUCOSAL BARRIER IN MAN AFTER ADMINISTRATION OF TAUROCHOLIC
GASTROENTEROLOGY 64: 1101-1105, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64 No.6 Printed in U.S.A. EFFECT OF CARBENOXOLONE ON THE GASTRIC MUCOSAL BARRIER IN MAN AFTER ADMINISTRATION OF TAUROCHOLIC
In 1971, Sir John Vane proposed that nonsteroidal
 Non-steroidal antiinflammatory drugs: facts and fallacies This article serves to review NSAIDs, their mechanism of action and adverse effects, as well as to establish the therapeutic position of the new-generation
Non-steroidal antiinflammatory drugs: facts and fallacies This article serves to review NSAIDs, their mechanism of action and adverse effects, as well as to establish the therapeutic position of the new-generation
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES
 CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
Gut Microbiota and IBD. Vahedi. H M.D Associate Professor of Medicine DDRI
 Gut Microbiota and IBD Vahedi. H M.D Associate Professor of Medicine DDRI 1393.3.1 2 GUT MICROBIOTA 100 Trillion Microbes - 10 times more than cells in our body Collective weight of about 1kg in human
Gut Microbiota and IBD Vahedi. H M.D Associate Professor of Medicine DDRI 1393.3.1 2 GUT MICROBIOTA 100 Trillion Microbes - 10 times more than cells in our body Collective weight of about 1kg in human
Interrelationship between Angiotensin Catecholamines. Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D.
 Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
Phospholipid Association Reduces the Gastric Mucosal Toxicity of Aspirin in Human Subjects
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 94, No. 7, 1999 1999 by Am. Coll. of Gastroenterology ISSN 0002-9270/99/$20.00 Published by Elsevier Science Inc. PII S0002-9270(99)00267-1 Phospholipid Association
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 94, No. 7, 1999 1999 by Am. Coll. of Gastroenterology ISSN 0002-9270/99/$20.00 Published by Elsevier Science Inc. PII S0002-9270(99)00267-1 Phospholipid Association
Product Contents. 1 Specifications 1 Product Description. 2 Buffer Preparation... 3 Protocol. 3 Ordering Information 4 Related Products..
 INSTRUCTION MANUAL Quick-RNA MidiPrep Catalog No. R1056 Highlights 10 minute method for isolating RNA (up to 1 mg) from a wide range of cell types and tissue samples. Clean-Spin column technology allows
INSTRUCTION MANUAL Quick-RNA MidiPrep Catalog No. R1056 Highlights 10 minute method for isolating RNA (up to 1 mg) from a wide range of cell types and tissue samples. Clean-Spin column technology allows
Rectal Administration of Nonsteroidal Antiinflammatory Drugs. Effect on Rat Gastric Ulcerogenicity and Prostaglandin E2 Synthesis
 GASTROENTEROLOGY 199;98:1245-1249 Rectal Administration of Nonsteroidal Antiinflammatory Drugs Effect on Rat Gastric Ulcerogenicity and Prostaglandin E2 Synthesis M. LIGUMSKY, M. SESTIERI, F. KARMELI,
GASTROENTEROLOGY 199;98:1245-1249 Rectal Administration of Nonsteroidal Antiinflammatory Drugs Effect on Rat Gastric Ulcerogenicity and Prostaglandin E2 Synthesis M. LIGUMSKY, M. SESTIERI, F. KARMELI,
CHAPTER 4 RESULTS. showed that all three replicates had similar growth trends (Figure 4.1) (p<0.05; p=0.0000)
 CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
Supplementary Information. Sonorensin: A new bacteriocin with potential of an anti-biofilm agent and a food
 Supplementary Information Sonorensin: A new bacteriocin with potential of an anti-biofilm agent and a food biopreservative Lipsy Chopra, Gurdeep Singh, Kautilya Kumar Jena and Debendra K. Sahoo* Biochemical
Supplementary Information Sonorensin: A new bacteriocin with potential of an anti-biofilm agent and a food biopreservative Lipsy Chopra, Gurdeep Singh, Kautilya Kumar Jena and Debendra K. Sahoo* Biochemical
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
RELEASE OF HISTAMINE INTO GASTRIC VENOUS BLOOD FOLLOWING INJURY BY ACETIC OR SALICYLIC ACID
 GASTROENTEROLOGY Copyright 1967 by The Williams & Wilkins Co. Vol. 52, No.3 Printed in U.S.A. RELEASE OF HISTAMINE INTO GASTRIC VENOUS BLOOD FOLLOWING INJURY BY ACETIC OR SALICYLIC ACID LEONARD R. JOHNSON
GASTROENTEROLOGY Copyright 1967 by The Williams & Wilkins Co. Vol. 52, No.3 Printed in U.S.A. RELEASE OF HISTAMINE INTO GASTRIC VENOUS BLOOD FOLLOWING INJURY BY ACETIC OR SALICYLIC ACID LEONARD R. JOHNSON
THERMALLY OXIDIZED SOYA BEAN OIL
 THERMALLY OXIDIZED SOYA BEAN OIL Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at the 55th JECFA (2000). An ADI of 0-3 mg/kg bw was established
THERMALLY OXIDIZED SOYA BEAN OIL Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at the 55th JECFA (2000). An ADI of 0-3 mg/kg bw was established
OBJECTIVE. Lipids are largely hydrocarbon derivatives and thus represent
 Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
BIOCHEMISTRY and MOLECULAR BIOLOGY INTERNATIONAL
 Vol. 44, No. 6, May 1998 BIOCHEMISTRY and MOLECULAR BIOLOGY INTERNATIONAL Pages 1187-1192 EFFECTS OF SHORT- AND LONG-TERM EXPOSURE TO L1NOLEIC ACID HYDROPEROXIDE ON CYTOSOLIC CALCIUM ION LEVEL OF HUMAN
Vol. 44, No. 6, May 1998 BIOCHEMISTRY and MOLECULAR BIOLOGY INTERNATIONAL Pages 1187-1192 EFFECTS OF SHORT- AND LONG-TERM EXPOSURE TO L1NOLEIC ACID HYDROPEROXIDE ON CYTOSOLIC CALCIUM ION LEVEL OF HUMAN
Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples:
 Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
21 Virginiamycin OH O. For chickens (except for broilers) broilers. Added amount 5~15 5~15 10~20 10~20
 21 Virginiamycin H H H H H H Virginiamycin M 1 C 28 H 35 3 7 MW: 525.6 CAS o.: 21411-53-0 Virginiamycin S 1 C 43 H 49 7 10 MW: 823.9 CAS o.: 23152-29-6 [Summary of virginiamycin] Virginiamycin (VM) is
21 Virginiamycin H H H H H H Virginiamycin M 1 C 28 H 35 3 7 MW: 525.6 CAS o.: 21411-53-0 Virginiamycin S 1 C 43 H 49 7 10 MW: 823.9 CAS o.: 23152-29-6 [Summary of virginiamycin] Virginiamycin (VM) is
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Detection of Low Level of Chloramphenicol in Milk and Honey with MIP SPE and LC-MS-MS
 Detection of Low Level of Chloramphenicol in Milk and Honey with MIP SPE and LC-MS-MS Olga Shimelis, An Trinh, and Michael Ye Supelco, Div. of Sigma-Aldrich, Bellefonte, PA T407125 Introduction Molecularly
Detection of Low Level of Chloramphenicol in Milk and Honey with MIP SPE and LC-MS-MS Olga Shimelis, An Trinh, and Michael Ye Supelco, Div. of Sigma-Aldrich, Bellefonte, PA T407125 Introduction Molecularly
-Mohammad Ashraf. -Anas Raed. -Alia Shatnawi. 1 P a g e
 -1 -Mohammad Ashraf -Anas Raed -Alia Shatnawi 1 P a g e Dr. Alia started the lecture by talking about subjects we are going to cover through this course; you can refer to the record if you are interested.
-1 -Mohammad Ashraf -Anas Raed -Alia Shatnawi 1 P a g e Dr. Alia started the lecture by talking about subjects we are going to cover through this course; you can refer to the record if you are interested.
19 Nosiheptide S O. For chickens (excluding broilers) For broilers. Finishing period broilers Growing period broilers. Stating chicks Growing chicks
 19 osiheptide H S H H S S H H 2 H S S H S H H H [Summary of nosiheptide] C 51 H 43 13 12 S 6 MW: 1222 CAS o.: 56377-79-8 osiheptide (H) is a polypeptide antibiotic obtained by the incubation of Streptomyces
19 osiheptide H S H H S S H H 2 H S S H S H H H [Summary of nosiheptide] C 51 H 43 13 12 S 6 MW: 1222 CAS o.: 56377-79-8 osiheptide (H) is a polypeptide antibiotic obtained by the incubation of Streptomyces
Non-steroidal anti-inflammatory drugs and gastrointestinal damage problems and solutions
 82 University of Glasgow and Department of Gastroenterology, Royal Infirmary, Glasgow Correspondence to: Professor R I Russell, 28 Ralston Road, Bearsden, Glasgow G61 3BA rirla@aol.com Submitted 26 October
82 University of Glasgow and Department of Gastroenterology, Royal Infirmary, Glasgow Correspondence to: Professor R I Russell, 28 Ralston Road, Bearsden, Glasgow G61 3BA rirla@aol.com Submitted 26 October
Luminescent platforms for monitoring changes in the solubility of amylin and huntingtin in living cells
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
4. Determination of fat content (AOAC, 2000) Reagents
 94 ANALYTICAL METHODS 1. Determination of moisture content (AOAC, 2000) 1. Dry the empty dish and lid in the oven at 105 C for 3 h and transfer to desiccator to cool. Weigh the empty dish and lid. 2. Weigh
94 ANALYTICAL METHODS 1. Determination of moisture content (AOAC, 2000) 1. Dry the empty dish and lid in the oven at 105 C for 3 h and transfer to desiccator to cool. Weigh the empty dish and lid. 2. Weigh
Helicobacter pylori:an Emerging Pathogen
 Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
A. Incorrect! Histamine is a secretagogue for stomach acid, but this is not the only correct answer.
 Pharmacology - Problem Drill 21: Drugs Used To Treat GI Disorders No. 1 of 10 1. Endogenous secretagogues for stomach acid include: #01 (A) Histamine (B) Gastrin (C) PGE1 (D) A and B (E) A, B and C Histamine
Pharmacology - Problem Drill 21: Drugs Used To Treat GI Disorders No. 1 of 10 1. Endogenous secretagogues for stomach acid include: #01 (A) Histamine (B) Gastrin (C) PGE1 (D) A and B (E) A, B and C Histamine
BGG s FucoMAX TM Fucoidan
 BGG s FucoMAX TM Fucoidan Protects Gastrointestinal Tract BGG s FucoMAX TM Fucoidan Protects Gastrointestinal Tract 1 BGG s FucoMAX TM the Proprietary and Potent Source and Formula of Fucoidan, and the
BGG s FucoMAX TM Fucoidan Protects Gastrointestinal Tract BGG s FucoMAX TM Fucoidan Protects Gastrointestinal Tract 1 BGG s FucoMAX TM the Proprietary and Potent Source and Formula of Fucoidan, and the
Cell-Derived Inflammatory Mediators
 Cell-Derived Inflammatory Mediators Introduction about chemical mediators in inflammation Mediators may be Cellular mediators cell-produced or cell-secreted derived from circulating inactive precursors,
Cell-Derived Inflammatory Mediators Introduction about chemical mediators in inflammation Mediators may be Cellular mediators cell-produced or cell-secreted derived from circulating inactive precursors,
PAF Acetylhydrolase Assay Kit
 PAF Acetylhydrolase Assay Kit Catalog Number KA1354 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay... 3 General
PAF Acetylhydrolase Assay Kit Catalog Number KA1354 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay... 3 General
Competing with antibiotic growth promoters the issues... Aoife Corrigan, Ph.D. Alltech Bioscience Centre Ireland November 2012
 Competing with antibiotic growth promoters the issues... Aoife Corrigan, Ph.D. Alltech Bioscience Centre Ireland November 2012 Are we ready for 9 billion??? The World needs to double the meat production
Competing with antibiotic growth promoters the issues... Aoife Corrigan, Ph.D. Alltech Bioscience Centre Ireland November 2012 Are we ready for 9 billion??? The World needs to double the meat production
prostaglandins on alkali secretion by rabbit gastric
 Gut, 1983, 24, 784-789 Effects of non-steroidal anti-inflammatory drugs and prostaglandins on alkali secretion by rabbit gastric fundus in vitro W D W REES, L C GIBBONS, AND L A TURNBERG From the University
Gut, 1983, 24, 784-789 Effects of non-steroidal anti-inflammatory drugs and prostaglandins on alkali secretion by rabbit gastric fundus in vitro W D W REES, L C GIBBONS, AND L A TURNBERG From the University
(b) Stomach s function 1. Dilution of food materials 2. Acidification of food (absorption of dietary Fe in small intestine) 3. Partial chemical digest
 (1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
(1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
New Topics in Aspirin Therapy
 Aspirin Therapy New Topics in Aspirin Therapy JMAJ 47(12): 566 572, 2004 Makoto HANDA Director and Associate Professor, Department of Transfusion Medicine and Cell Therapy, School of Medicine, Keio University
Aspirin Therapy New Topics in Aspirin Therapy JMAJ 47(12): 566 572, 2004 Makoto HANDA Director and Associate Professor, Department of Transfusion Medicine and Cell Therapy, School of Medicine, Keio University
