Challenges for use of CTCs as a Diagnostic. Farideh Z. Bischoff, Ph.D. Interim CSO Sr. Director, Translational Clinical Development Biocept, Inc.
|
|
|
- Roland McGee
- 5 years ago
- Views:
Transcription
1 Challenges for use of CTCs as a Diagnostic Farideh Z. ischoff, Ph.D. Interim CSO Sr. Director, Translational Clinical Development iocept, Inc.
2 Current Technology for CTC Testing Existing CTC testing platform is CellSearch by Veridex/J&J CellSearch provides CTC enumeration CellSearch is FDA cleared for 3 cancer types (breast, colorectal, and prostate) but limited to metastatic, late-stage patients Many supportive studies: Positive correlation between pretreatment (baseline) CTC numbers and clinical prognosis/recurrence or response to therapy
3 Ideal CTC (Diagnostic) Test Components CTC Enumeration Protein Marker analysis by ICC (e.g. ER) Gene Amplification by FISH (e.g. HER2) Mutation Analysis by PCR (e.g. EGFR)
4 CTCs as a Surrogate for Tumor Tissue Guide selection of best therapeutic for individual patients Identify patients with minimal residual disease Detect treatment-resistant profiles Improve understanding of mechanisms of biological metastatic processes Discover and identify new targets for therapeutic manipulation
5 Limits of Current CTC Technology CellSearch detects CTCs in 30-60% of blood samples from cancer patients (breast, CRC, prostate) Limited to epithelial CTCs Use of single CTC-specific capture Ab (e.g., just EpCAM) Use of single CTC detection stain (cytokeratin; CK)
6 Many CTC Recovery and Detection Methods Described Enrichment/Isolation Immunomagnetic (MACS; CellSearch ) Centrifugation/Density Separation Filtration (ISET) Red cell lysis/slide preparation (EPIC) Microfluidic (iocept, MGH, On-Q-ity) Electrophoresis Detection Cytometric (IHC, ICC, FISH) Nucleic Acid-ased
7 Inlet Silicone channel Glass slide Cover slip iocept s OncoCEE Microchannel Design Outlet Uniquely designed to maximize rare cell capture by randomizing size/shape and placement of posts: Prevent laminar flow 9,000 posts; 24?L total volume Capture and visualization of tumor cells directly within the microchannel
8 Problem 1: CTC Capture Solution: Antibody Cocktail Surface antigens are additive; enable more sensitive capture of low and high level antigen expressing cells Epithelial and/or mesenchymal-like cell types captured simultaneously (all aspects of EMT epithelial-to-mesenchymal transition) 8
9 Technical Validation Study: CTC Recovery and Her2 Determination Antibody Cocktail: EpCAM, Trop-2, c-met, Folate binding protein receptor, N-Cadherin, CD318, MSC, Her2, Muc-1,and EGFR CTC Detection (CK+/CD45-/DAPI+) 8 of 29 stage II-III cases (28%) 52 of 63 stage IV or recurrent cases (82.5%)? 60 Informative (CK+) cases CD45+ nucleated lymphocytes Standard CTC Definition CK+, CD45-, DAPI+ tumor cell
10 Determination of Her2 Status 56 of 60 CK+ Cases with Known Her2 Status (by tumor tissue) CTC Result (FISH) Patient (Tissue) Her2 Positive Her2 Negative Her2 Positive 20 4 Her2 Negative 1 31 lue = Concordant Red = Discordant Overall, 91% Concordance Her2 FISH: 83% Sensitivity, 97% Specificity
11 CK+ Her2+ CTC Composite Image DAPI Green Orange CK and Centromere 17 Her2
12 Her2 Amplification Confirms CK-negative CTC Chromosome 8 centromere (blue) Her2 gene (red), on chromosome 17 Chromosome 17 centromere (green) 12
13 Correlation of CK+ stain and Her2 Amp Among all Her2+ cells identified (n=76): 36 identified on relocation of CK+ cells (47%) 40 identified on analysis of all CK- cells (53%)
14 Problem 2: CTC Detection Solution: CK & Enhanced staining CTCs must be distinguished from non-specific background cells (e.g., WCs) Today, CTCs are detected (and defined) by cytokeratin (CK) staining; but CTCs may lose CK through EMT Additional target cell types, e.g., endothelial, mesenchymal and stem cells, may require different detection strategies (beyond CK)
15 15 CEE-Enhanced TM stain: Detecting what we catch CK+ cell CK+ cell plus CEE-Enhanced stain CK- cell plus CEE -Enhanced stain (standard method of CTC detection) C Cell Captured On Channel with Antibody cocktail A
16 Weak Staining CK+ CTCs ecome righter Weak CK+ Ca CTC DAPI-stained WCs + CEE-Enhanced WCs unchanged CK+ cell plus CEE- Enhanced stain
17 reast Cancer CTC Study (MDA): Operable Patient Cohort 25 patient blood samples taken pre-surgery Process and capture using antibody cocktail Stain using CK combined with CEE-Enhanced TM CTCs detected in 19 of 25 (76%) cases y stage: y subtype: T1N0 (9/12) Luminal A (15/19) T1N1 (2/3) Luminal (2/3) T2N0 (1/3) Her2 (1/2) T2N1 (2/2) Triple negative (1/1) T2N3 (1/1) T3N0 (1/1) T4N0 (2/2) T4N3 (1/1) 17
18 Summary Methods that enable reliable recovery and detection of CTCs exist FISH-based methods used for secondary analysis (assay development) and/or detection of clinically relevant biomarkers (CLIA; laboratory developed tests) Clinical validation of biomarkers using CTCs needed Longitudinal studies to assess clinical significance
19 FDA & LDTs Current situation Commercial sale of instrument or diagnostic kits (e.g., Veridex) requires FDA clearance Reference Laboratories offering commercial tests (lab developed tests) are under the prevue of CLIA do not require FDA submission FDA office of In Vitro Diagnostic Device Evaluation and Safety stressed that LDT oversight process will take time to develop & will include input from the industry and consumer groups Draft legislation creating a new category for both LDTs and IVDs regulated by CMS and CLIA that would include reimbursement policies Why the Difference between FDA Submission & LDTS In vitro diagnostic kits/instruments are sold across the US. LDTs are a medical service under the control of a medical director, per CLIA, in a defined setting Why the interest by the FDA Response to public health risks created by LDTs that may not provide reasonable assurances of safety and effectiveness Lack of information or accounting on LDTS provided to FDA CLIA only requires analytical validity of LDTs, not clinical validity. FDA Commissioner Margaret Hamburg believes this is needed for personalized medicine to be successful
Agenda. What is a Liquid Biopsy? Biocept technology. Concordance With Tissue. Clinical Applications. Billing and Reimbursement.
 Agenda What is a Liquid Biopsy? Biocept technology Concordance With Tissue Clinical Applications Billing and Reimbursement Recap & Questions 1 Targets of Tumor Found in Liquid Biopsy 1 Eric Topol, Professor
Agenda What is a Liquid Biopsy? Biocept technology Concordance With Tissue Clinical Applications Billing and Reimbursement Recap & Questions 1 Targets of Tumor Found in Liquid Biopsy 1 Eric Topol, Professor
A Novel CTC-Detecting Technique Using TelomeScan and Its Clinical Applications
 A Novel CTC-Detecting Technique Using TelomeScan and Its Clinical Applications Yasuo Urata CEO and President Oncolys BioPharma Inc. February 16, 2013 Telomere Length is a Limiting Factor for Cell Replication
A Novel CTC-Detecting Technique Using TelomeScan and Its Clinical Applications Yasuo Urata CEO and President Oncolys BioPharma Inc. February 16, 2013 Telomere Length is a Limiting Factor for Cell Replication
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Finite-element analysis of cell cluster dynamics in different cluster trap architectures. (a) Cluster-Chip (b) Filter (c) A structure identical to the Cluster-Chip except that one
Supplementary Figure 1 Finite-element analysis of cell cluster dynamics in different cluster trap architectures. (a) Cluster-Chip (b) Filter (c) A structure identical to the Cluster-Chip except that one
High Sensitivity Immunomagnetic CTC Isolation as Compared to Alternative Isolation Methods
 High Sensitivity Immunomagnetic CTC Isolation as Compared to Alternative Isolation Methods 1. Introduction: An overview of CTC isolation methods 2. Challenges for direct comparisons of CTC recovery 3.
High Sensitivity Immunomagnetic CTC Isolation as Compared to Alternative Isolation Methods 1. Introduction: An overview of CTC isolation methods 2. Challenges for direct comparisons of CTC recovery 3.
Circulating tumor cells/dna/etc for Radiation Oncologists
 Circulating tumor cells/dna/etc for Radiation Oncologists Andrew Z. Wang, M.D. Associate Professor Director of Clinical and Translational Research Department of Radiation Oncology Carolina Center for Cancer
Circulating tumor cells/dna/etc for Radiation Oncologists Andrew Z. Wang, M.D. Associate Professor Director of Clinical and Translational Research Department of Radiation Oncology Carolina Center for Cancer
CIRCULATING TUMOR CELL (CTC) DIAGNOSTICS: TECHNOLOGIES AND GLOBAL MARKETS
 CIRCULATING TUMOR CELL (CTC) DIAGNOSTICS: TECHNOLOGIES AND GLOBAL MARKETS PHM153A January 2014 Usha Nagavarapu Project Analyst ISBN: 1-56965-671-1 BCC Research 49 Walnut Park, Building 2 Wellesley, MA
CIRCULATING TUMOR CELL (CTC) DIAGNOSTICS: TECHNOLOGIES AND GLOBAL MARKETS PHM153A January 2014 Usha Nagavarapu Project Analyst ISBN: 1-56965-671-1 BCC Research 49 Walnut Park, Building 2 Wellesley, MA
Circulating Tumor Cells (CTC) Technologies
 Table of Contents MIR036 I. SCOPE AND METHODOLOGY Scope of the Study Analytics and data presented in this report pertain to several parameters such as - Research Methodology This report is uniquely researched
Table of Contents MIR036 I. SCOPE AND METHODOLOGY Scope of the Study Analytics and data presented in this report pertain to several parameters such as - Research Methodology This report is uniquely researched
Incorporating pharmacodynamic, response and patient selection biomarkers. Paul Elvin PhD Chief Translational Science Officer Aptus Clinical
 Incorporating pharmacodynamic, response and patient selection biomarkers Paul Elvin PhD Chief Translational Science Officer Aptus Clinical 22 Oncology drug development Biomarkers key for: Strong hypothesis
Incorporating pharmacodynamic, response and patient selection biomarkers Paul Elvin PhD Chief Translational Science Officer Aptus Clinical 22 Oncology drug development Biomarkers key for: Strong hypothesis
La biopsia liquida. Aldo Scarpa. Anatomia Patologica e ARC-NET Centro di Ricerca Applicata sul Cancro
 La biopsia liquida Aldo Scarpa Anatomia Patologica e ARC-NET Centro di Ricerca Applicata sul Cancro Azienda Ospedaliera Universitaria Integrata di Verona Obstacles to precision oncology Genomic heterogeneity
La biopsia liquida Aldo Scarpa Anatomia Patologica e ARC-NET Centro di Ricerca Applicata sul Cancro Azienda Ospedaliera Universitaria Integrata di Verona Obstacles to precision oncology Genomic heterogeneity
Supplementary Figures and Tables
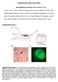 Supplementary Figures and Tables Novel platform for detection of CK+ and CK- CTCs Chad V. Pecot, Farideh Z. Bischoff, Julie Ann Mayer, Karina L. Wong, Tam Pham, Justin Bottsford-Miller, Rebecca L. Stone,
Supplementary Figures and Tables Novel platform for detection of CK+ and CK- CTCs Chad V. Pecot, Farideh Z. Bischoff, Julie Ann Mayer, Karina L. Wong, Tam Pham, Justin Bottsford-Miller, Rebecca L. Stone,
Prospective Clinical Study of Circulating Tumor Cells For Colorectal Cancer Screening
 Prospective Clinical Study of Circulating Tumor Cells For Colorectal Cancer Screening Results From a Multi-Year 620-Sample Study CRC: SLOW GROWING, PREVENTABLE POLYP TO CANCER CAN TAKE 5 TO 15 YEARS CANCER
Prospective Clinical Study of Circulating Tumor Cells For Colorectal Cancer Screening Results From a Multi-Year 620-Sample Study CRC: SLOW GROWING, PREVENTABLE POLYP TO CANCER CAN TAKE 5 TO 15 YEARS CANCER
Circulating tumor cells as biomarker for hormonal treatment in breast and prostate cancer. Michal Mego
 National Cancer Institute, Slovakia Translational Research Unit Circulating tumor cells as biomarker for hormonal treatment in breast and prostate cancer Michal Mego 2 nd Department of Oncology, Faculty
National Cancer Institute, Slovakia Translational Research Unit Circulating tumor cells as biomarker for hormonal treatment in breast and prostate cancer Michal Mego 2 nd Department of Oncology, Faculty
The Presence and Persistence of Resistant and Stem Cell- Like Tumor Cells as a Major Problem in Ovarian Cancer
 Welcome! The Presence and Persistence of Resistant and Stem Cell- Like Tumor Cells as a Major Problem in Ovarian Cancer Prof. Sabine Kasimir-Bauer Department of Gynecology and Obstetrics University Hospital
Welcome! The Presence and Persistence of Resistant and Stem Cell- Like Tumor Cells as a Major Problem in Ovarian Cancer Prof. Sabine Kasimir-Bauer Department of Gynecology and Obstetrics University Hospital
The CellCollector TM technology
 The CellCollector TM technology In vivo isolation of circulating tumor cells by the CellCollector TM system Dr. Klaus Lücke (CEO) Unmet medical need in oncology: Access to tumor cells not only at the time
The CellCollector TM technology In vivo isolation of circulating tumor cells by the CellCollector TM system Dr. Klaus Lücke (CEO) Unmet medical need in oncology: Access to tumor cells not only at the time
Cover Letter. Reviewer 1:
 Cover Letter Michael Yang, M.D., Ph.D. Managing Editor of Cancer Research Frontiers 1188 Willis Ave, #109, Albertson, NY 11507, USA Phone: +1-917-426-1571 http://cancer-research-frontiers.org/ Dear Dr.
Cover Letter Michael Yang, M.D., Ph.D. Managing Editor of Cancer Research Frontiers 1188 Willis Ave, #109, Albertson, NY 11507, USA Phone: +1-917-426-1571 http://cancer-research-frontiers.org/ Dear Dr.
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE 1 CANCER PATIENTS
 WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE 1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE 1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
Circulating Tumor Cells in non- Metastatic Triple Negative Breast Cancer
 Circulating Tumor Cells in non- Metastatic Triple Negative Breast Cancer Carolyn Hall, Ph.D. Department of Surgical Oncology The University of Texas MD Anderson Cancer Center Triple Negative Breast Cancer
Circulating Tumor Cells in non- Metastatic Triple Negative Breast Cancer Carolyn Hall, Ph.D. Department of Surgical Oncology The University of Texas MD Anderson Cancer Center Triple Negative Breast Cancer
10/15/2012. Biologic Subtypes of TNBC. Topics. Topics. Histopathology Molecular pathology Clinical relevance
 Biologic Subtypes of TNBC Andrea L. Richardson M.D. Ph.D. Brigham and Women s Hospital Dana-Farber Cancer Institute Harvard Medical School Boston, MA Topics Histopathology Molecular pathology Clinical
Biologic Subtypes of TNBC Andrea L. Richardson M.D. Ph.D. Brigham and Women s Hospital Dana-Farber Cancer Institute Harvard Medical School Boston, MA Topics Histopathology Molecular pathology Clinical
AdnaNews. News from the meeting on Advances in Circulating Tumor Cells (ACTC), Reythymnon, Crete, Greece October 8-11, 2014.
 News from the meeting on Advances in Circulating Tumor Cells (ACTC), Reythymnon, Crete, Greece October 8-11, 2014. The ACTC meeting (www.actc2014.org) is together with the ISMRC meeting one of the most
News from the meeting on Advances in Circulating Tumor Cells (ACTC), Reythymnon, Crete, Greece October 8-11, 2014. The ACTC meeting (www.actc2014.org) is together with the ISMRC meeting one of the most
1/23/2017. Alarice Lowe, MD Assistant Professor of Pathology Director, Circulating Tumor Cell Lab Brigham and Women s Hospital Harvard Medical School
 Application of Cytologic Techniques to Circulating Tumor Cell Specimens Alarice Lowe, MD Assistant Professor of Pathology Director, Circulating Tumor Cell Lab Brigham and Women s Hospital Harvard Medical
Application of Cytologic Techniques to Circulating Tumor Cell Specimens Alarice Lowe, MD Assistant Professor of Pathology Director, Circulating Tumor Cell Lab Brigham and Women s Hospital Harvard Medical
Youngnam Cho. National Cancer Center Biomarker Branch
 Youngnam Cho National Cancer Center Biomarker Branch Contents 1. Liquid Biopsy 2. Circulating Tumor Cells from Blood 3. Cell-free DNA from Blood 1. Liquid biopsy Cancer Diagnosis IMAGING TISSUE BIOPSY
Youngnam Cho National Cancer Center Biomarker Branch Contents 1. Liquid Biopsy 2. Circulating Tumor Cells from Blood 3. Cell-free DNA from Blood 1. Liquid biopsy Cancer Diagnosis IMAGING TISSUE BIOPSY
Liquid Biopsy: Implications for Cancer Staging & Therapy
 Prof. Klaus Pantel, MD, PhD Institut für Tumorbiologie Liquid Biopsy: Implications for Cancer Staging & Therapy Tumor cell dissemination and cancer dormancy Primary tumor Local relapse Cancer cells disseminate
Prof. Klaus Pantel, MD, PhD Institut für Tumorbiologie Liquid Biopsy: Implications for Cancer Staging & Therapy Tumor cell dissemination and cancer dormancy Primary tumor Local relapse Cancer cells disseminate
BIOCEPT INC FORM FWP. (Free Writing Prospectus - Filing under Securities Act Rules 163/433) Filed 01/22/15
 BIOCEPT INC FORM FWP (Free Writing Prospectus - Filing under Securities Act Rules 163/433) Filed 01/22/15 Address 5810 NANCY RIDGE DR SAN DIEGO, CA, 92121 Telephone 858-320-8200 CIK 0001044378 Symbol BIOC
BIOCEPT INC FORM FWP (Free Writing Prospectus - Filing under Securities Act Rules 163/433) Filed 01/22/15 Address 5810 NANCY RIDGE DR SAN DIEGO, CA, 92121 Telephone 858-320-8200 CIK 0001044378 Symbol BIOC
Detection of the Circulating Tumor Cells in Cancer Patients
 Detection of the Circulating Tumor Cells in Cancer Patients Athanasios Armakolas; Zacharoula Panteleakou; Adrianos Nezos; Aikaterini Tsouma; Maria Skondra; Peter Lembessis; Nikolaos Pissimissis; Michael
Detection of the Circulating Tumor Cells in Cancer Patients Athanasios Armakolas; Zacharoula Panteleakou; Adrianos Nezos; Aikaterini Tsouma; Maria Skondra; Peter Lembessis; Nikolaos Pissimissis; Michael
Inertia based microfluidic capture and characterisation of circulating tumour cells for the diagnosis of lung cancer
 Original Article Page 1 of 6 Inertia based microfluidic capture and characterisation of circulating tumour cells for the diagnosis of lung cancer Dimple Y. Chudasama 1,2,3, Daria V. Freydina 1,4, Maxim
Original Article Page 1 of 6 Inertia based microfluidic capture and characterisation of circulating tumour cells for the diagnosis of lung cancer Dimple Y. Chudasama 1,2,3, Daria V. Freydina 1,4, Maxim
Immunohistochemical classification of breast tumours
 Immunohistochemical classification of breast tumours Workshop in Diagnostic Immunohistochemistry September 19 th - 21 th 2018 Anne-Vibeke Lænkholm Department of Surgical Pathology, Zealand University Hospital,
Immunohistochemical classification of breast tumours Workshop in Diagnostic Immunohistochemistry September 19 th - 21 th 2018 Anne-Vibeke Lænkholm Department of Surgical Pathology, Zealand University Hospital,
CTC in clinical studies: Latest reports on GI cancers
 CTC in clinical studies: Latest reports on GI cancers François-Clément Bidard, MD PhD GI cancers are characterized by Multimodal treatment strategies Treatments are adapted to tumor burden & prognosis
CTC in clinical studies: Latest reports on GI cancers François-Clément Bidard, MD PhD GI cancers are characterized by Multimodal treatment strategies Treatments are adapted to tumor burden & prognosis
Corporate Overview June NASDAQ: BIOC
 Corporate Overview June 2017 NASDAQ: BIOC www.biocept.com Forward-Looking Statements This presentation contains, and any accompanying oral presentation would no doubt contain, forward-looking statements,
Corporate Overview June 2017 NASDAQ: BIOC www.biocept.com Forward-Looking Statements This presentation contains, and any accompanying oral presentation would no doubt contain, forward-looking statements,
The Avatar System TM Yields Biologically Relevant Results
 Application Note The Avatar System TM Yields Biologically Relevant Results Liquid biopsies stand to revolutionize the cancer field, enabling early detection and noninvasive monitoring of tumors. In the
Application Note The Avatar System TM Yields Biologically Relevant Results Liquid biopsies stand to revolutionize the cancer field, enabling early detection and noninvasive monitoring of tumors. In the
Colorectal cancer diagnostics: biomarkers, cellfree DNA, circulating tumor cells and defining heterogeneous populations by single-cell analysis
 Expert Review of Molecular Diagnostics ISSN: 1473-7159 (Print) 1744-8352 (Online) Journal homepage: http://www.tandfonline.com/loi/iero20 Colorectal cancer diagnostics: biomarkers, cellfree DNA, circulating
Expert Review of Molecular Diagnostics ISSN: 1473-7159 (Print) 1744-8352 (Online) Journal homepage: http://www.tandfonline.com/loi/iero20 Colorectal cancer diagnostics: biomarkers, cellfree DNA, circulating
Survey Results Q1. How would you best describe your organization?
 Survey Results Q1. How would you best describe your organization? Q2. How high would you rate the priority of Circulating Tumor Cells in you organization? Q3. What do you think is the biggest challenge
Survey Results Q1. How would you best describe your organization? Q2. How high would you rate the priority of Circulating Tumor Cells in you organization? Q3. What do you think is the biggest challenge
The Role of CTCs as Tumor Biomarkers
 The Role of CTCs as Tumor Biomarkers 21 Evi S. Lianidou, Athina Markou, and Areti Strati Abstract Detection of Circulating Tumor Cells (CTCs) in peripheral blood can serve as a liquid biopsy approach and
The Role of CTCs as Tumor Biomarkers 21 Evi S. Lianidou, Athina Markou, and Areti Strati Abstract Detection of Circulating Tumor Cells (CTCs) in peripheral blood can serve as a liquid biopsy approach and
Layered-IHC (L-IHC): A novel and robust approach to multiplexed immunohistochemistry So many markers and so little tissue
 Page 1 The need for multiplex detection of tissue biomarkers. There is a constant and growing demand for increased biomarker analysis in human tissue specimens. Analysis of tissue biomarkers is key to
Page 1 The need for multiplex detection of tissue biomarkers. There is a constant and growing demand for increased biomarker analysis in human tissue specimens. Analysis of tissue biomarkers is key to
Transform genomic data into real-life results
 CLINICAL SUMMARY Transform genomic data into real-life results Biomarker testing and targeted therapies can drive improved outcomes in clinical practice New FDA-Approved Broad Companion Diagnostic for
CLINICAL SUMMARY Transform genomic data into real-life results Biomarker testing and targeted therapies can drive improved outcomes in clinical practice New FDA-Approved Broad Companion Diagnostic for
Corporate Medical Policy
 Corporate Medical Policy Microarray-based Gene Expression Testing for Cancers of Unknown File Name: Origination: Last CAP Review: Next CAP Review: Last Review: microarray-based_gene_expression_testing_for_cancers_of_unknown_primary
Corporate Medical Policy Microarray-based Gene Expression Testing for Cancers of Unknown File Name: Origination: Last CAP Review: Next CAP Review: Last Review: microarray-based_gene_expression_testing_for_cancers_of_unknown_primary
Identifying genomic signatures in circulating breast tumour cells
 Identifying genomic signatures in circulating breast tumour cells 9 th ISMRC 2013, Paris, France September 25th, 2013 NISHA KANWAR PhD Candidate Department of Laboratory Medicine and Pathobiology University
Identifying genomic signatures in circulating breast tumour cells 9 th ISMRC 2013, Paris, France September 25th, 2013 NISHA KANWAR PhD Candidate Department of Laboratory Medicine and Pathobiology University
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE CANCER PATIENTS
 WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
Personalized medecine Biomarker
 Patient Disease Diagnosis Risk prediction Personalized medecine Biomarker Targeted therapies Diagnostic Theranostic: Efficay Toxicity Treatment 1 THE CASE FOR PERSONALIZED MEDICINE IS COMPELLING.. Toxicity
Patient Disease Diagnosis Risk prediction Personalized medecine Biomarker Targeted therapies Diagnostic Theranostic: Efficay Toxicity Treatment 1 THE CASE FOR PERSONALIZED MEDICINE IS COMPELLING.. Toxicity
Breast cancer pathology
 Breast cancer pathology Giancarlo Pruneri, M.D. National Cancer Institute (INT) Milan University of Milan, School of Medicine Giancarlo.Pruneri@unimi.it Currently accepted prognostic/predictive parameters
Breast cancer pathology Giancarlo Pruneri, M.D. National Cancer Institute (INT) Milan University of Milan, School of Medicine Giancarlo.Pruneri@unimi.it Currently accepted prognostic/predictive parameters
Detection of Circulating Tumor Cells Harboring a Unique ALK-Rearrangement in ALK- Positive Non-Small-Cell Lung Cancer.
 Detection of Circulating Tumor Cells Harboring a Unique ALK-Rearrangement in ALK- Positive Non-Small-Cell Lung Cancer Pailler, et al Data Supplement Table S1. Numbers and Percentages of ALK-Rearranged
Detection of Circulating Tumor Cells Harboring a Unique ALK-Rearrangement in ALK- Positive Non-Small-Cell Lung Cancer Pailler, et al Data Supplement Table S1. Numbers and Percentages of ALK-Rearranged
February 2, Dear Dr. Shuren,
 American Cancer Society Cancer Action Network 555 11 th Street, NW Suite 300 Washington, DC 20004 202.661.5700 www.acscan.org Jeffrey E. Shuren, M.D., J.D. Director, Center for Devices and Radiological
American Cancer Society Cancer Action Network 555 11 th Street, NW Suite 300 Washington, DC 20004 202.661.5700 www.acscan.org Jeffrey E. Shuren, M.D., J.D. Director, Center for Devices and Radiological
1. Q: What has changed from the draft recommendations posted for public comment in November/December 2011?
 Frequently Asked Questions (FAQs) in regard to Molecular Testing Guideline for Selection of Lung Cancer Patients for EGFR and ALK Tyrosine Kinase Inhibitors 1. Q: What has changed from the draft recommendations
Frequently Asked Questions (FAQs) in regard to Molecular Testing Guideline for Selection of Lung Cancer Patients for EGFR and ALK Tyrosine Kinase Inhibitors 1. Q: What has changed from the draft recommendations
Breast Cancer Interpretation Guide
 Breast Cancer Interpretation Guide UCT D O R P NEW ERBB2/ C E P S ht e ZytoLig lor Prob o C l a u 2D D17S12 ng to the i d r o c c a ting for re-tes idelines 2013 ASCO Gu Breast Cancer Interpretation Guide
Breast Cancer Interpretation Guide UCT D O R P NEW ERBB2/ C E P S ht e ZytoLig lor Prob o C l a u 2D D17S12 ng to the i d r o c c a ting for re-tes idelines 2013 ASCO Gu Breast Cancer Interpretation Guide
Laboratory Developed Tests (LDTs) and the FDA: The Impact on Clinical Laboratories
 Laboratory Developed Tests (LDTs) and the FDA: The Impact on Clinical Laboratories THSNA: Current Challenges in Coagulation Curtis A. Hanson, M.D. Professor of Laboratory Medicine and Pathology Mayo Clinic
Laboratory Developed Tests (LDTs) and the FDA: The Impact on Clinical Laboratories THSNA: Current Challenges in Coagulation Curtis A. Hanson, M.D. Professor of Laboratory Medicine and Pathology Mayo Clinic
Accelerate Your Research with Conversant Bio
 Imagination has given us the steam engine, the telephone, the talkingmachine and the automobile, for these things had to be dreamed of before they became realities. So I believe that dreams... are likely
Imagination has given us the steam engine, the telephone, the talkingmachine and the automobile, for these things had to be dreamed of before they became realities. So I believe that dreams... are likely
The OncoBEAM Platform: The Use of a High Sensitive Technology for Liquid Biopsies in Clinical Practice
 The OncoBEAM Platform: The Use of a High Sensitive Technology for Liquid Biopsies in Clinical Practice Sysmex Inostics Dr. Friederike Lehmann Head of CRO Marketing Sysmex Corporation Kobe 2 Sysmex Corporation
The OncoBEAM Platform: The Use of a High Sensitive Technology for Liquid Biopsies in Clinical Practice Sysmex Inostics Dr. Friederike Lehmann Head of CRO Marketing Sysmex Corporation Kobe 2 Sysmex Corporation
In most industrialized countries, primary lung cancer is. Circulating Tumor Cells in Pulmonary Venous Blood of Primary Lung Cancer Patients
 Circulating Tumor Cells in Pulmonary Venous Blood of Primary Lung Cancer Patients Yoshitomo Okumura, MD, Fumihiro Tanaka, MD, PhD, Kazue Yoneda, Masaki Hashimoto, MD, Teruhisa Takuwa, MD, Nobuyuki Kondo,
Circulating Tumor Cells in Pulmonary Venous Blood of Primary Lung Cancer Patients Yoshitomo Okumura, MD, Fumihiro Tanaka, MD, PhD, Kazue Yoneda, Masaki Hashimoto, MD, Teruhisa Takuwa, MD, Nobuyuki Kondo,
SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer.
 Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
Present Role of Immunohistochemistry in the. Subtypes. Beppe Viale European Institute of Oncology University of Milan Milan-Italy
 Present Role of Immunohistochemistry in the Classification of Molecular Subtypes Beppe Viale European Institute of Oncology University of Milan Milan-Italy We know it is many diseases Breast cancer is
Present Role of Immunohistochemistry in the Classification of Molecular Subtypes Beppe Viale European Institute of Oncology University of Milan Milan-Italy We know it is many diseases Breast cancer is
Welcome! HER2 TESTING DIAGNOSTIC ACCURACY 4/11/2016
 HER2 TESTING DIAGNOSTIC ACCURACY Can t We Finally Get It Right? Allen M. Gown, M.D. Medical Director and Chief Pathologist PhenoPath Laboratories Seattle, Washington Clinical Professor of Pathology University
HER2 TESTING DIAGNOSTIC ACCURACY Can t We Finally Get It Right? Allen M. Gown, M.D. Medical Director and Chief Pathologist PhenoPath Laboratories Seattle, Washington Clinical Professor of Pathology University
When starting a new line of therapy for your metastatic breast, colorectal, and prostate 1 cancer patients
 When starting a new line of therapy for your metastatic breast, colorectal, and prostate 1 cancer patients There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
When starting a new line of therapy for your metastatic breast, colorectal, and prostate 1 cancer patients There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
CanAssist-Breast: New test for prediction of risk of recurrence in ER+/Her2- early stage breast cancer patients Manjiri Bakre, Ph.D.
 CanAssist-Breast: New test for prediction of risk of recurrence in ER+/Her2- early stage breast cancer patients Manjiri Bakre, Ph.D. Founder and CEO OncoStem Diagnostics Pvt. Ltd. Treatment Decision: HR+/HER2-
CanAssist-Breast: New test for prediction of risk of recurrence in ER+/Her2- early stage breast cancer patients Manjiri Bakre, Ph.D. Founder and CEO OncoStem Diagnostics Pvt. Ltd. Treatment Decision: HR+/HER2-
Deciphering the biology that drives response to immunotherapy
 Deciphering the biology that drives response to immunotherapy Phenoptics TM Quantitative Pathology Platform Trent Norris, Field Application Scientist September 15, 2016 HUMAN HEALTH ENVIRONMENTAL HEALTH
Deciphering the biology that drives response to immunotherapy Phenoptics TM Quantitative Pathology Platform Trent Norris, Field Application Scientist September 15, 2016 HUMAN HEALTH ENVIRONMENTAL HEALTH
Proteomic Biomarker Discovery in Breast Cancer
 Proteomic Biomarker Discovery in Breast Cancer Rob Baxter Laboratory for Cellular and Diagnostic Proteomics Kolling Institute, University of Sydney, Royal North Shore Hospital robert.baxter@sydney.edu.au
Proteomic Biomarker Discovery in Breast Cancer Rob Baxter Laboratory for Cellular and Diagnostic Proteomics Kolling Institute, University of Sydney, Royal North Shore Hospital robert.baxter@sydney.edu.au
College of American Pathologists
 College of American Pathologists Comments to the Food and Drug Administration on the draft guidance In Vitro Companion Diagnostics Devices October 12, 2011 College of American Pathologists 1350 I Street,
College of American Pathologists Comments to the Food and Drug Administration on the draft guidance In Vitro Companion Diagnostics Devices October 12, 2011 College of American Pathologists 1350 I Street,
Question 1 A. ER-, PR-, HER+ B. ER+, PR+, HER2- C. ER-, PR+, HER2- D. ER-, PR-, HER2- E. ER-, PR+, HER2+
 Triple Negative Breast Cancer Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA Question 1 The tumor depicted on the next slide
Triple Negative Breast Cancer Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA Question 1 The tumor depicted on the next slide
Maximizing the Potential of Population-Based Cancer Registries to Inform Cancer Research
 Maximizing the Potential of Population-Based Cancer Registries to Inform Cancer Research Presented by: Thomas C. Tucker, PhD, MPH Associate Director for Cancer Control Markey Cancer Center University of
Maximizing the Potential of Population-Based Cancer Registries to Inform Cancer Research Presented by: Thomas C. Tucker, PhD, MPH Associate Director for Cancer Control Markey Cancer Center University of
Supplementary Online Content
 Supplementary Online Content Fumagalli D, Venet D, Ignatiadis M, et al. RNA Sequencing to predict response to neoadjuvant anti-her2 therapy: a secondary analysis of the NeoALTTO randomized clinical trial.
Supplementary Online Content Fumagalli D, Venet D, Ignatiadis M, et al. RNA Sequencing to predict response to neoadjuvant anti-her2 therapy: a secondary analysis of the NeoALTTO randomized clinical trial.
Review. Organ-specific markers in circulating tumor cell screening: an early indicator of metastasis capable malignancy.
 For reprint orders, please contact: reprints@futuremedicine.com Organ-specific markers in circulating tumor cell screening: an early indicator of metastasis capable malignancy Patrizia Paterlini-Bréchot
For reprint orders, please contact: reprints@futuremedicine.com Organ-specific markers in circulating tumor cell screening: an early indicator of metastasis capable malignancy Patrizia Paterlini-Bréchot
Detecting Oncogenic Mutations in Whole Blood
 WHITE PAPER Detecting Oncogenic Mutations in Whole Blood Analytical validation of Cynvenio Biosystems LiquidBiopsy circulating tumor cell (CTC) capture and next-generation sequencing (NGS) September 2013
WHITE PAPER Detecting Oncogenic Mutations in Whole Blood Analytical validation of Cynvenio Biosystems LiquidBiopsy circulating tumor cell (CTC) capture and next-generation sequencing (NGS) September 2013
Statistical validation of biomarkers and surogate endpoints
 Statistical validation of biomarkers and surogate endpoints Marc Buyse, ScD IDDI, Louvain-la-Neuve & Hasselt University, Belgium marc.buyse@iddi.com OUTLINE 1. Setting the scene: definitions and types
Statistical validation of biomarkers and surogate endpoints Marc Buyse, ScD IDDI, Louvain-la-Neuve & Hasselt University, Belgium marc.buyse@iddi.com OUTLINE 1. Setting the scene: definitions and types
Immune Cell Phenotyping in Solid Tumors using Quantitative Pathology
 Immune Cell Phenotyping in Solid Tumors using Quantitative Pathology James R. Mansfield Director of Quantitative Pathology Applications 2009 PerkinElmer What is Quantitative Pathology? Quantitative Pathology
Immune Cell Phenotyping in Solid Tumors using Quantitative Pathology James R. Mansfield Director of Quantitative Pathology Applications 2009 PerkinElmer What is Quantitative Pathology? Quantitative Pathology
MET as a novel treatment target- the story of the sleeping beauty. Balazs Halmos M.D. Montefiore Medical Center/Albert Einstein College of Medicine
 MET as a novel treatment target- the story of the sleeping beauty Balazs Halmos M.D. Montefiore Medical Center/Albert Einstein College of Medicine MET as a novel treatment target MET as an oncogene MET
MET as a novel treatment target- the story of the sleeping beauty Balazs Halmos M.D. Montefiore Medical Center/Albert Einstein College of Medicine MET as a novel treatment target MET as an oncogene MET
Urinary ctdna Platform for Diagnosis and Cancer Treatment Monitoring. Summit August 19,2015
 Urinary ctdna Platform for Diagnosis and Cancer Treatment Monitoring Mark G. Erlander, Ph.D., CSO CHI Next Generation Summit August 19,2015 Circulating Tumor DNA (ctdna) Tumor cells Main Advantages of
Urinary ctdna Platform for Diagnosis and Cancer Treatment Monitoring Mark G. Erlander, Ph.D., CSO CHI Next Generation Summit August 19,2015 Circulating Tumor DNA (ctdna) Tumor cells Main Advantages of
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE1 CANCER PATIENTS
 WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
The use of diagnostic FFPE material in cancer epidemiology research
 The use of diagnostic FFPE material in cancer epidemiology research Neil O Callaghan Genetic Epidemiology Laboratory Department of Pathology The University of Melbourne www.pedigree.org.au Overview Who
The use of diagnostic FFPE material in cancer epidemiology research Neil O Callaghan Genetic Epidemiology Laboratory Department of Pathology The University of Melbourne www.pedigree.org.au Overview Who
OS related to CTC response (response: 30% decline) 4 weeks 8 weeks 12 weeks
 OS related to CTC response (response: 30% decline) 4 weeks 8 weeks 12 weeks 20 CTC characterization (DNA, RNA, proteins) - Therapeutic targets - Resistance mechanisms Detection of therapeutic targets on
OS related to CTC response (response: 30% decline) 4 weeks 8 weeks 12 weeks 20 CTC characterization (DNA, RNA, proteins) - Therapeutic targets - Resistance mechanisms Detection of therapeutic targets on
7th November, Translational Science: how to move from biology to clinical applications
 7th November, 2014 Translational Science: how to move from biology to clinical applications 1 Translational science: How to move from biology to clinical applications Translational cancer genomics and
7th November, 2014 Translational Science: how to move from biology to clinical applications 1 Translational science: How to move from biology to clinical applications Translational cancer genomics and
Supplemental Information. High-Throughput Microfluidic Labyrinth for the. Label-free Isolation of Circulating Tumor Cells
 Cell Systems, Volume 5 Supplemental Information High-Throughput Microfluidic Labyrinth for the Label-free Isolation of Circulating Tumor Cells Eric Lin, Lianette Rivera-Báez, Shamileh Fouladdel, Hyeun
Cell Systems, Volume 5 Supplemental Information High-Throughput Microfluidic Labyrinth for the Label-free Isolation of Circulating Tumor Cells Eric Lin, Lianette Rivera-Báez, Shamileh Fouladdel, Hyeun
Breast cancer: Molecular STAGING classification and testing. Korourian A : AP,CP ; MD,PHD(Molecular medicine)
 Breast cancer: Molecular STAGING classification and testing Korourian A : AP,CP ; MD,PHD(Molecular medicine) Breast Cancer Theory: Halsted Operative breast cancer is a local-regional disease The positive
Breast cancer: Molecular STAGING classification and testing Korourian A : AP,CP ; MD,PHD(Molecular medicine) Breast Cancer Theory: Halsted Operative breast cancer is a local-regional disease The positive
Qué hemos aprendido hasta hoy? What have we learned so far?
 Qué hemos aprendido hasta hoy? What have we learned so far? Luís Costa Hospital de Santa Maria & Instituto de Medicina Molecular Faculdade de Medicina de Lisboa Disclosures Research Grants: Amgen; Novartis;
Qué hemos aprendido hasta hoy? What have we learned so far? Luís Costa Hospital de Santa Maria & Instituto de Medicina Molecular Faculdade de Medicina de Lisboa Disclosures Research Grants: Amgen; Novartis;
Optimal algorithm for HER2 testing
 Optimal algorithm for HER2 testing The revised definition of IHC 2+ (equivocal) is invasive breast cancer with Weak to moderate complete membrane staining observed in >10% of tumor cells. (see Figure 1
Optimal algorithm for HER2 testing The revised definition of IHC 2+ (equivocal) is invasive breast cancer with Weak to moderate complete membrane staining observed in >10% of tumor cells. (see Figure 1
Triple Negative Breast Cancer. Eric P. Winer, MD Dana-Farber Cancer Institute Harvard Medical School Boston, MA October, 2008
 Triple Negative Breast Cancer Eric P. Winer, MD Dana-Farber Cancer Institute Harvard Medical School Boston, MA October, 2008 Triple Negative Breast Cancer 15% 25% Triple Negative 20% HER2+ ER+ Low Grade
Triple Negative Breast Cancer Eric P. Winer, MD Dana-Farber Cancer Institute Harvard Medical School Boston, MA October, 2008 Triple Negative Breast Cancer 15% 25% Triple Negative 20% HER2+ ER+ Low Grade
FDA Companion Diagnostic Testing and Implications for Pharmacy and Medical Directors
 FDA Companion Diagnostic Testing and Implications for Pharmacy and Medical Directors Diana Brixner, PhD, RPh, FAMCP Professor, University of Utah Health Sciences Executive Director, Pharmacotherapy Outcomes
FDA Companion Diagnostic Testing and Implications for Pharmacy and Medical Directors Diana Brixner, PhD, RPh, FAMCP Professor, University of Utah Health Sciences Executive Director, Pharmacotherapy Outcomes
Applications of IHC. Determination of the primary site in metastatic tumors of unknown origin
 Applications of IHC Determination of the primary site in metastatic tumors of unknown origin Classification of tumors that appear 'undifferentiated' by standard light microscopy Precise classification
Applications of IHC Determination of the primary site in metastatic tumors of unknown origin Classification of tumors that appear 'undifferentiated' by standard light microscopy Precise classification
CANCER. Clinical Validation of Breast Cancer Predictive Markers
 Clinical Validation of Breast Cancer Predictive Markers David Hicks, MD Loralee McMahon, MS, HTL(ASCP) CANCER The human body is composed of billions of highly regulated cells Cancer cells no longer respond
Clinical Validation of Breast Cancer Predictive Markers David Hicks, MD Loralee McMahon, MS, HTL(ASCP) CANCER The human body is composed of billions of highly regulated cells Cancer cells no longer respond
Circulating tumor cells
 Circulating tumor cells counts and characteristics Bianca Mostert Circulating tumor cells counts and characteristics Bianca Mostert The studies described in this thesis were performed at the Department
Circulating tumor cells counts and characteristics Bianca Mostert Circulating tumor cells counts and characteristics Bianca Mostert The studies described in this thesis were performed at the Department
MEETING THE STANDARDS: FDA MANDATORY RAPID INFLUENZA DETECTION TEST (RIDTs) RECLASSIFICATION
 MEETING THE STANDARDS: FDA MANDATORY RAPID INFLUENZA DETECTION TEST (RIDTs) RECLASSIFICATION NOVEMBER 6, 2017 SALLY A. HOJVAT M.Sc., Ph.D. Retired as Director of FDA Division of Microbiology Devices, CDRH
MEETING THE STANDARDS: FDA MANDATORY RAPID INFLUENZA DETECTION TEST (RIDTs) RECLASSIFICATION NOVEMBER 6, 2017 SALLY A. HOJVAT M.Sc., Ph.D. Retired as Director of FDA Division of Microbiology Devices, CDRH
Disclosure. Summary. Circulating DNA and NGS technology 3/27/2017. Disclosure of Relevant Financial Relationships. JS Reis-Filho, MD, PhD, FRCPath
 Circulating DNA and NGS technology JS Reis-Filho, MD, PhD, FRCPath Director of Experimental Pathology, Department of Pathology Affiliate Member, Human Oncology and Pathogenesis Program Disclosure of Relevant
Circulating DNA and NGS technology JS Reis-Filho, MD, PhD, FRCPath Director of Experimental Pathology, Department of Pathology Affiliate Member, Human Oncology and Pathogenesis Program Disclosure of Relevant
Circulating Stromal Cells in Immunotherapy Utilizing a Total Blood Based Biopsy
 Circulating Stromal Cells in Immunotherapy Utilizing a Total Blood Based Biopsy Cancer associated macrophage CTCs Daniel Adams Senior Research Scientist/Head of Clinical Core Laboratory Creatv MicroTech,
Circulating Stromal Cells in Immunotherapy Utilizing a Total Blood Based Biopsy Cancer associated macrophage CTCs Daniel Adams Senior Research Scientist/Head of Clinical Core Laboratory Creatv MicroTech,
Cytogenetics 101: Clinical Research and Molecular Genetic Technologies
 Cytogenetics 101: Clinical Research and Molecular Genetic Technologies Topics for Today s Presentation 1 Classical vs Molecular Cytogenetics 2 What acgh? 3 What is FISH? 4 What is NGS? 5 How can these
Cytogenetics 101: Clinical Research and Molecular Genetic Technologies Topics for Today s Presentation 1 Classical vs Molecular Cytogenetics 2 What acgh? 3 What is FISH? 4 What is NGS? 5 How can these
Janet E. Dancey NCIC CTG NEW INVESTIGATOR CLINICAL TRIALS COURSE. August 9-12, 2011 Donald Gordon Centre, Queen s University, Kingston, Ontario
 Janet E. Dancey NCIC CTG NEW INVESTIGATOR CLINICAL TRIALS COURSE August 9-12, 2011 Donald Gordon Centre, Queen s University, Kingston, Ontario Session: Correlative Studies in Phase III Trials Title: Design
Janet E. Dancey NCIC CTG NEW INVESTIGATOR CLINICAL TRIALS COURSE August 9-12, 2011 Donald Gordon Centre, Queen s University, Kingston, Ontario Session: Correlative Studies in Phase III Trials Title: Design
Challenges and unanswered questions for the next decade of circulating tumour cell research in lung cancer
 Review Article Challenges and unanswered questions for the next decade of circulating tumour cell research in lung cancer Sumitra Mohan, Francesca Chemi, Ged Brady Clinical and Experimental Pharmacology
Review Article Challenges and unanswered questions for the next decade of circulating tumour cell research in lung cancer Sumitra Mohan, Francesca Chemi, Ged Brady Clinical and Experimental Pharmacology
Quality Assurance and Quality Control in the Pathology Dept.
 Quality Assurance and Quality Control in the Pathology Dept. Judith Sandbank M.D. Pathology Assaf-Harofeh Medical Center ISRAEL jsandbank@asaf.health.gov.il 2 nd IBDC, 9 th February, 2012 Pathology as
Quality Assurance and Quality Control in the Pathology Dept. Judith Sandbank M.D. Pathology Assaf-Harofeh Medical Center ISRAEL jsandbank@asaf.health.gov.il 2 nd IBDC, 9 th February, 2012 Pathology as
Contents 1 The Windows of Susceptibility to Breast Cancer 2 The So Called Pre-Neoplastic Lesions and Carcinoma In Situ
 Contents 1 The Windows of Susceptibility to Breast Cancer... 1 1.1 Introduction... 1 1.2 Risk Factor and Etiological Agents... 2 1.3 The Concept of the Windows of Susceptibility to Carcinogenesis... 5
Contents 1 The Windows of Susceptibility to Breast Cancer... 1 1.1 Introduction... 1 1.2 Risk Factor and Etiological Agents... 2 1.3 The Concept of the Windows of Susceptibility to Carcinogenesis... 5
Advances in Pathology and molecular biology of lung cancer. Lukas Bubendorf Pathologie
 Advances in Pathology and molecular biology of lung cancer Lukas Bubendorf Pathologie Agenda The revolution of predictive markers Liquid biopsies PD-L1 Molecular subtypes (non-squamous NSCLC) Tsao AS et
Advances in Pathology and molecular biology of lung cancer Lukas Bubendorf Pathologie Agenda The revolution of predictive markers Liquid biopsies PD-L1 Molecular subtypes (non-squamous NSCLC) Tsao AS et
Triple Negative Breast Cancer
 Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Recent advances in breast cancers
 Recent advances in breast cancers Breast cancer is a hetrogenous disease due to distinct genetic alterations. Similar morphological subtypes show variation in clinical behaviour especially in response
Recent advances in breast cancers Breast cancer is a hetrogenous disease due to distinct genetic alterations. Similar morphological subtypes show variation in clinical behaviour especially in response
Enterprise Interest No
 Enterprise Interest No SY-05 Pulmonary Pathology: Options for targeted therapy in lung cancer Liquid biopsy in thoracic oncology Where are we now? Paul Hofman Laboratory of Clinical and Experimental Pathology
Enterprise Interest No SY-05 Pulmonary Pathology: Options for targeted therapy in lung cancer Liquid biopsy in thoracic oncology Where are we now? Paul Hofman Laboratory of Clinical and Experimental Pathology
AVENIO family of NGS oncology assays ctdna and Tumor Tissue Analysis Kits
 AVENIO family of NGS oncology assays ctdna and Tumor Tissue Analysis Kits Accelerating clinical research Next-generation sequencing (NGS) has the ability to interrogate many different genes and detect
AVENIO family of NGS oncology assays ctdna and Tumor Tissue Analysis Kits Accelerating clinical research Next-generation sequencing (NGS) has the ability to interrogate many different genes and detect
Evolution of Pathology
 1 Traditional pathology Molecular pathology 2 Evolution of Pathology Gross Pathology Cellular Pathology Morphologic Pathology Molecular/Predictive Pathology Antonio Benivieni (1443-1502): First autopsy
1 Traditional pathology Molecular pathology 2 Evolution of Pathology Gross Pathology Cellular Pathology Morphologic Pathology Molecular/Predictive Pathology Antonio Benivieni (1443-1502): First autopsy
HSL-Advanced Diagnostics 2018 / 19 Test & Service List
 HSL-Advanced Diagnostics 2018 / 19 Test & Service List 2018/19 TEST & SERVICE LIST Haematoxylin & Eosin H&E H&E per slide Routine Immunohistochemistry Immunohistochemical demonstration of an antigen in
HSL-Advanced Diagnostics 2018 / 19 Test & Service List 2018/19 TEST & SERVICE LIST Haematoxylin & Eosin H&E H&E per slide Routine Immunohistochemistry Immunohistochemical demonstration of an antigen in
TEB. Id4 p63 DAPI Merge. Id4 CK8 DAPI Merge
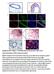 a Duct TEB b Id4 p63 DAPI Merge Id4 CK8 DAPI Merge c d e Supplementary Figure 1. Identification of Id4-positive MECs and characterization of the Comma-D model. (a) IHC analysis of ID4 expression in the
a Duct TEB b Id4 p63 DAPI Merge Id4 CK8 DAPI Merge c d e Supplementary Figure 1. Identification of Id4-positive MECs and characterization of the Comma-D model. (a) IHC analysis of ID4 expression in the
Clinical Trial Design to Expedite Drug Development Mary W. Redman, Ph.D.
 Clinical Trial Design to Expedite Drug Development Mary W. Redman, Ph.D. What do we mean by expediting drug development? Phase I Single Arm Phase II (expansion cohort) Randomized Phase II Phase III Necessary?
Clinical Trial Design to Expedite Drug Development Mary W. Redman, Ph.D. What do we mean by expediting drug development? Phase I Single Arm Phase II (expansion cohort) Randomized Phase II Phase III Necessary?
FISH mcgh Karyotyping ISH RT-PCR. Expression arrays RNA. Tissue microarrays Protein arrays MS. Protein IHC
 Classification of Breast Cancer in the Molecular Era Susan J. Done University Health Network, Toronto Why classify? Prognosis Prediction of response to therapy Pathogenesis Invasive breast cancer can have
Classification of Breast Cancer in the Molecular Era Susan J. Done University Health Network, Toronto Why classify? Prognosis Prediction of response to therapy Pathogenesis Invasive breast cancer can have
R.G.C.C.-RESEARCH GENETIC CANCER CENTRE. Florina, 28 / 12 / 2006 Dear Dr. Gilbard and Dr. Hammon, LIPOMAS, UNDIAGNOSED PAIN, FAMILY HISTORY OR CANCER
 Florina, 28 / 12 / 2006 Dear Dr. Gilbard and Dr. Hammon, LIPOMAS, UNDIAGNOSED PAIN, FAMILY HISTORY OR CANCER We send you the results from the analysis made about a patient (????????????) for screening.
Florina, 28 / 12 / 2006 Dear Dr. Gilbard and Dr. Hammon, LIPOMAS, UNDIAGNOSED PAIN, FAMILY HISTORY OR CANCER We send you the results from the analysis made about a patient (????????????) for screening.
CTC molecular characterization: Are we ready to move forward with clinical testing?
 CTC molecular characterization: Are we ready to move forward with clinical testing? Michail Ignatiadis MD, PhD Jules Bordet Institute, Université Libre de Bruxelles Brussels, Belgium Breast cancer: Diagnostics
CTC molecular characterization: Are we ready to move forward with clinical testing? Michail Ignatiadis MD, PhD Jules Bordet Institute, Université Libre de Bruxelles Brussels, Belgium Breast cancer: Diagnostics
Surgical Pathology Lab of the Future. Thomas M. Grogan, M.D. Professor of Pathology, University of Arizona Founder, Ventana Medical Systems, Inc.
 Surgical Pathology Lab of the Future Thomas M. Grogan, M.D. Professor of Pathology, University of Arizona Founder, Ventana Medical Systems, Inc. 28 April 2010 Objective Demonstrate how the next generation
Surgical Pathology Lab of the Future Thomas M. Grogan, M.D. Professor of Pathology, University of Arizona Founder, Ventana Medical Systems, Inc. 28 April 2010 Objective Demonstrate how the next generation
HER2 status assessment in breast cancer. Marc van de Vijver Academic Medical Centre (AMC), Amsterdam
 HER2 status assessment in breast cancer Marc van de Vijver Academic Medical Centre (AMC), Amsterdam 13e Bossche Mamma Congres 17 th June 2015 Modern cancer therapies are based on sophisticated molecular
HER2 status assessment in breast cancer Marc van de Vijver Academic Medical Centre (AMC), Amsterdam 13e Bossche Mamma Congres 17 th June 2015 Modern cancer therapies are based on sophisticated molecular
National Medical Policy
 National Medical Policy Subject: Circulating Tumor Cells Policy Number: NMP 513 Effective Date*: June 2012 Updated: June 2017 This National Medical Policy is subject to the terms in the IMPORTANT NOTICE
National Medical Policy Subject: Circulating Tumor Cells Policy Number: NMP 513 Effective Date*: June 2012 Updated: June 2017 This National Medical Policy is subject to the terms in the IMPORTANT NOTICE
