Absence of histone F1 in a mitotically dividing, genetically inactive
|
|
|
- Beryl Harrington
- 5 years ago
- Views:
Transcription
1 Proc. Nat. Acad. Sci. USA Vol. 72, No. 7, pp , July 1975 Cell Biology Absence of histone F1 in a mitotically dividing, genetically inactive nucleus (chromatin/phosphorylation/ Tetrahymena /macronucleus/micronucleus) MARTIN A. GOROVSKY AND JOSEPHINE BOWEN KEEVERT Department of Biology, University of Rochester, Rochester, New York Communicated by Hewson Swift, March 17,1975 ABSTRACT Histones were extracted from macro- and micronuclear chromatin of the ciliated protozoan Tetrahymena pyriformis. Conditions that resulted in macronuclear chromatin containing large amounts of histone F1 yielded micronuclear chromatin in which this histone was absent. Evidence is presented indicating that the absence of F1 from micronuclei is not a preparative artifact and that histone F1 is replaced by other tistone fractions. Since micronuclei divide mitotically, while macronuclei divide amitotically, these results suggest that histone F1 and its phosphorylation do not play an indispensable role in the process of mitotic chromosome condensation, in chromosome replication, or in the separation of newly synthesized chromatids. Like most ciliated protozoans, the vegetative cells of Tetrahymena pyriformis contain a macro- and a micronucleus. These two nuclei are formed from the daughter products of a single mitotic division during the sexual process of conjugation. It is likely, therefore, that they contain the same, or closely related, genetic information. In spite of their common origin and the fact that they reside in the same cytoplasmic millieu, macro- and micronuclei differ considerably in their structure and function (see ref. 1 for review). For example, macronuclei divide amitotically, with no sign of mitotic chromosomes; micronuclei divide mitotically (2-7). In addition, the two nuclei differ greatly in their genetic activity. Macronuclei are sites of intense RNA synthesis while micronuclei synthesize little, if any, detectable RNA (8, 9). We have recently reported that the macronucleus of Tetrahymena contains a histone with properties similar to those of histone F1 of higher organisms (1). Macronuclear F1 isolated from rapidly growing cells was found to be highly phosphorylated, while histones isolated from slowly growing cells contained only small amounts of phosphorylated FL. Thus, the relationship between the rate of cell replication and the extent of phosphorylation of histone F1 observed in a variety of other cell types (11-16) was also observed in Tetrahymena. Recent studies of histone phosphorylation in different cells have led to the various suggestions that phosphorylation of histone F1 may be involved in the alteration of chromatin structure during interphase (17), in chromosome replication (18, 19), in the sorting out of newly synthesized chromatids during S-phase (2), in the process of mitotic chromosome condensation (21-23) or in the control of genetic activity (24-26). Since macronuclei divide amitotically, we concluded from these studies that phosphorylation of histone F1 probably does not play a role exclusively in the process of mitotic chromosome condensation. Due to the striking functional and structural differences between macro- and micronuclei, it seemed that further analysis of histone F1 and its phosphorylation in Tetrahymena micronuclei might shed additional light on the function of this histone in eukaryotic nuclei. In this report, we describe the surprising finding that isolated micronuclei of Tetrahymena do not contain any detectable histone FL. MATERIALS AND METHODS Cell Culture. Tetrahymena pyriformis, strains WH-6 (Syngen 1, mating type I; a micronucleate strain) and GL (an amicronucleate strain), were cultured as described (27). Isolation of Nuclei and Chromatin. Nuclei were isolated as described (27, 28). Chromatin was isolated by a modification of the method of Panyim et al. (29). Chromatin deficient in histone fraction F1 was prepared by washing isolated nuclei repeatedly in.5 M NaCl. This treatment was found to remove most of histone F1 from macronuclei while extracting little of the other histone fractions. Isolation of Histones. Histones were isolated from whole nuclei or from chromatin by extraction with either 2.4 M urea/.2 M H2SO4 or with.2 M H2SO4 (1, 27). Thermal Denaturation of Isolated Chromatin. Solubilized macro- or micronuclear chromatin was fixed in.91% formaldehyde for at least 15 hr and then dialyzed for at least 5 hr against three changes (1 volumes each) of.2 mm EDTA. DNA isolated from Tetrahymena macronuclei was similarly treated and served as a control. Increase in absorbance at 26 nm with heating was measured in a Gilford 24 spectrophotometer. The heating rate was approximately.3 per min. Turbidity was monitored at 32 nm and was found to be negligible. Buoyant Density of Isolated Chromatin in CsCl. Solubilized chromatin was fixed for at least 24 hr in.91% formaldehyde and was then centrifuged to equilibrium in gradients containing 5.3 ml of 38.5% CsCl (p = 1.4) in.2 mm EDTA. Centrifugation was performed in a Beckman SW-5 rotor at 32, rpm at 2 for at least 6 hr. Fractions were collected directly into scintillation vials, and radioactivity was determined as described below. Acrylamide Gel Electrophoresis. Electrophoresis in long (25 cm) 2.5 M urea/15% polyacrylamide gels was performed by the method of Panyim and Chalkley (3). Gels were stained in fast green and subjected to densitometric analysis as described (1, 31). Isotopic Labeling and Counting Procedures. For labeling histones, cells of strain WH-6 were grown for at least 3 generations in medium containing 1,uCi/ml of [3H]lysine (L-[G-3H]lysine, 3.2 Ci/mmol, 1 mci/ml, New England Nuclear Corp.). To measure the amount of macronuclear contamination in the micronuclear preparation, we similarly labeled a culture of the amicronucleate strain GL with
2 Cell Biology: Gorovsky and Keevert AsCi/ml of [14C]lysine (L-[U-'4C]lysine, 312 mci/mmol,. ;1 mci/ml, New England Nuclear). The two cell types were mixed immediately before isolation of nuclei. Any 14C appearing in micronuclear histones must be due to contamination (the _4C-labeled GL cells have no micronuclei), and the degree of contamination could be estimated by comparison with the amount of 14C in the macronuclear histones. The DNA of chromatin was labeled by growing cells in enriched proteose peptone (from which yeast extract had been omitted) containing either.2,uci/ml of [14C]thymidine ([methyl-14c]thymidine, 52.7 mci/mmol, 1 gci/ ml, New England Nuclear) or 1. /ACi/ml of [3H]thymidine ([methyl-3h]thymidine, 5.7 Ci/mmol,.5 mci/ml, New England Nuclear). Polyacrylamide gels were sliced into 1-mm segments and counted in either 5 or 1 ml of fluor (Spectrafluor, Amersham-Searle, Arlington Heights, Ill.), containing.25 ml of NCS (Amersham-Searle). Fractions from CsCl gradients were counted in PCS (Amersham-Searle). All dual isotope experiments were corrected for spillover of 14C into the 3H channel by use of the automatic external standard on a Nuclear Chicago Isocap 3 scintillation counter. In later experiments, efficiency corrections were performed with a Nova 2 computer (Data General Corp.). RESULTS Histone Fraction F1 Is Absent in Micronuclei. We have previously reported (1) that F1 isolated from Tetrahymena macronuclei is similar to the Fis of other organisms in most of its chemical and physiological properties (perchloric acid solubility, molecular weight, amino-acid composition, phosphorylation associated with cell division, etc.). However, Tetrahymena F1 differs from other Fis in having a unique electrophoretic mobility in urea/acrylamide gels of low ph, migrating between fraction F2B and the unacetylated form of fraction F2A1 (1). In macronuclei (Fig. 1A) this fraction is intensely labeled when cells are grown in radioactive lysine. However, there is little or no radioactivity in this region of a gel containing histones extracted from micronuclear chromatin (Fig. 1B). The absence in this gel of any detectable '4C-labeled histones from the macronuclei of the amicronucleate cells attests to the purity of the micronuclear histones. While the experiment in Fig. 1 illustrates one of our best efforts in preparing micronuclear histones completely free of macronuclear contamination, histone preparations isolated from micronuclear chromatin routinely contain little or no Fl. The variable amounts of F1 found in some preparations of micronuclear histones can probably be accounted for by low levels (-2%) of macronuclear contamination frequently found in micronuclear preparations. Absence of Histone F1 from Micronuclei Is Not a Preparative Artifact. To demonstrate that the isolation of micronuclei does not lead to an artifactual loss of F1, we performed a mixing experiment. Cells of strain WH-6 were labeled with [14C]lysine as described for strain GL. Macronuclei were isolated and fragmented by blending in a Virtis blender. The "4C-labeled macronuclear fragments were then added to [3H]lysine-labeled cells (strain WH-6) and macroand micronuclei were isolated. Carrier (unlabeled) macronuclei were then added to the micronuclear fraction, chromatin was isolated, and the histones were extracted and analyzed. Fig. 2 shows that "4C-labeled F1 derived from the added macronuclear fragments was present in normal amounts (relative to the other macronuclear histones) in the Proc. Nat. Acad. Sci. USA 72 (1975) SLICE NUMBER FIG. 1. Electrophoretic comparison of macro- and micronuclear histones. (A) [3H]Lysine-labeled macronuclear histones (-) from strain WH-6 and [14C]lysine-labeled histones () from strain GL. (B) [3H]Lysine-labeled histones () from micronuclei of strain WH-6 and [14C]lysine-labeled histones from strain GL (). The identification of the electrophoretic fractions (except for X and Y, which remain uncharacterized) of histones extracted from macronuclei of strain WH-6 has been described (1, 35). The differences in the electrophoretic profiles of histones isolated from macronuclei of strains GL and WH-6 will be discussed elsewhere (C. A. Johmann and M. A. Gorovsky, in preparation). Other differences between the histone complements of macro- and micronuclei, such as the absence of fractions F3 and Y and of acetylated subspecies, are the subject of other communications (ref. 25; M. A. Gorovsky and J. B. Keevert, in preparation). micronuclear fraction. The histones extracted from [3H]lysine-labeled micronuclei, on the other hand, were deficient in Fl. Since the 3H-labeled micronuclei and the "4C-labeled macronuclear fragments should have been subjected to identical conditions during isolation, it is unlikely that the absence of histone F1 from micronuclei is due to loss during isolation. Comparison of Micronuclear Chromatin with Fl-Deficient Macronuclear Chromatin. The thermal denaturation and buoyant density of DNA in chromatin are greatly influenced by the amount of protein with which the DNA is associated (32-35). Therefore, we compared the thermal denaturation properties and the buoyant density of micronuclear chromatin with macronuclear chromatin and with macronuclear chromatin from which histone F1 was removed. Table 1 and Fig. 3 demonstrate that the half melting temperature (Tm) of micronuclear chromatin is essentially the same as that of macronuclear chromatin, and is distinctly different from that of macronuclear chromatin from which histone F1 has been removed by.5 M NaCl. Washing micronuclei with.5 M NaCl prior to isolating chromatin results in only a small change in its thermal denaturation properties (Fig. 3).
3 2674 Cell Biology: Gorovsky and Keevert Proc. Nat. Acad. Sci. USA 72 (1975). al SLICE NUMBER FIG. 2. Electrophoretic comparison of macro- and micronuclear histones. (A) [3H]Lysine-labeled macronuclear histones () from strain WH-6 and [14C]lysine-labeled histones () from added macronuclei of strain WH-6. (B) [3H]Lysine-labeled histones () from micronuclei of strain WH-6 and [14C]lysine-labeled histones () from added macronuclei of strain WH-6. All samples in panel B were counted for 1 min. The small deviations below zero in this figure result from the fact that all vials containing NCS and fluor were first counted before the gel slices were added and digested. Background counts were then subtracted for each vial. Fig. 4 demonstrates that the buoyant density of formaldehyde-fixed micronuclear chromatin in CsCl is different from that of macronuclear chromatin from which histone F1 has been removed. The difference in buoyant densities of macro- and micronuclear chromatin seen in Fig. 4 may be due to different amounts of non-histone protein associated with the two types of chromatin (see below). Washing micronuclei with.5 M NaCl results in little change in the buoyant density of micronuclear chromatin. Histone:DNA Ratios of Macro- and Micronuclear Chromatins. Since the thermal denaturation properties and the buoyant density of micronuclear chromatin were unlike those of macronuclear chromatin devoid of histone F1, it seemed likely that increases in the amounts of other micronuclear histones served to compensate for the absence of histone Fl. To see if this was the case, we have compared the amounts of acid-soluble material extracted from macro- and micronuclear chromatin which migrates in histone regions of polyacrylamide gels with the amounts of DNA of those chromatins. In Exps. 1 and 2 (Table 2) histones were measured by quantitative microdensitometry of gels stained with fast-green (31). Because macro- and micronuclei contain different histone fractions (Fig. 1; ref. 27) and the color yields of the individual histone-fast-green complexes are unknown, we also compared the histone:dna ratio of the two 5 Table 1. Summary of thermal denaturation properties of macro- and micronuclear chromatin No. of Hyper- Chromatin exps. a Tmn chromicityb Macronucleus Micronucleus Macronucleus salt-washed Micronucleus salt-washed DNAC a Values are for independent isolations of chromatin. Values for DNA represent 11 experiments on the same sample. b Calculated as % increase in absorbance at 26 nm at,86 over that at room temperature. Values are uncorrected for thermal expansion of water. c DNA was from isolated macronuclei. The thermal denaturation properties of macro- and micronuclear DNAs are indistinguishable. chromatins by isotopic techniques (Exp. 3, Table 2); it seemed unlikely that the incorporation of a mixture of radioactive amino acids would lead to the same systematic errors as the staining technique. The data of Table 2 show that the histone:dna ratios of macro- and micronuclear chromatins are essentially the same. DISCUSSION In some experiments we noted that histones extracted from isolated micronuclear preparations contained less F1 than might be expected from the amounts of macronuclear contamination. Similarly, when histones were extracted from small quantities of macronuclei, less F1 (relative to the other histone fractions) was often recovered than if histones were extracted from a larger number of nuclei. The reason for this failure to recover small amounts of F1 is unknown. Histone F1 is also highly susceptible to proteolytic degradation (36) and is the easiest histone to dissociate from the chromatin with salts (37) and with acids (38). Therefore, it was necessary to demonstrate that the absence of F1 in histones extracted from micronuclei was not due to artifactual loss during isolation of nuclei or extraction of histones. In this regard, it should be noted that (i) comparisons have only been made between macro- and micronuclei isolated from the same cells, (Hi) the methods used to separate macro- and micronuclei during isolation are physical techniques (centrifugation, filtration) which would not be expected to dissociate any histones from chromatin, and (Mi) the isolation procedures are known to inhibit proteolysis of histone F1 in macronuclei (1). We have also performed a mixing experiment in which 4C-labeled macronuclei were added to a micronuclear Table 2. Histone/DNA in macro- and micronuclear chromatin Macro- Micro- Exp. no. nucleusa, b nucleusa 1cC d e a Values in arbitrary units. b Value set = 1.. c DNA by diphenylamine; histone by fast green. d DNA by A26; histone by fast green. e DNA by [L4C]thymidine; histone by [3Hjaminoacid mixture.
4 Cell Biology: Gorovsky and Keevert Proc. Nat. Acad. Sc. USA 72 (1975) ' Y Ar~~~~~~~~~~~~~~C 4~~~~~~~~~ H~~~~~~~~~~~~~~~ 5 C- 1 _ ~ F- 1 5 UI 1 z w w TEMPERATURE FIG. 3. Thermal denaturation profile of macro- and micronuclear chromatins. (A) Denaturation of macronuclear chromatin () has been compared with that of macronuclear chromatin isolated from nuclei that had been washed with.5 M NaCl to remove histone F1 (). (B) Thermal denaturation profiles of micronuclear chromatin (-) and micronuclear chromatin from salt-washed nuclei (3) are similarly compared. The thermal denaturation profile of purified macronuclear DNA (A) is included for comparison with the various chromatins. The thermal denaturation profile of purified micronuclear DNA is indistinguishable from that of macronuclear DNA (M. -C. Yao and M. A. Gorovsky, unpublished observations). All data points are average values from at least three experiments (see text and Table 1 for additional details). preparation that had been labeled with a tritiated amino acid. In this experiment, care was also taken to add large amounts of carrier (unlabeled) macronuclei to insure good recovery of Fl. The 3H profile of gels containing histones from such a mixture of nuclei showed a typical micronuclear pattern, while the 14C profile (and the absorbance) showed a typical macronuclear pattern, with normal amounts of histone F1 (Fig. 2). We conclude from this mixing experiment that the absence of F1 from micronuclei reflects an intrinsic difference between these two nuclei. In addition, the thermal denaturation properties and the buoyant density of micronuclear chromatin are different from those of macronuclear chromatin from which histone F1 has been removed by salt, and the histone:dna ratios of macroand micronuclear chromatins are similar. We have also examined the (unlikely) possibility that micronuclear chromatin contains histone F1, but in a form that is not easily extracted with acids. Accordingly, we have extracted the acidinsoluble residues of macro- and micronuclear chromatins with.1% sodium dodecyl sulfate. No additional histone-like material was found when these extracts were examined on polyacrylamide gels containing dodecyl sulfate. Taken together, these results argue strongly against the possibility that the absence of histone F1 from micronuclear chromatin is a preparative artifact. Thus, while studies on isolated cellular components cannot unequivocally rule out the possibility that, in vivo, micronuclear chromatin also contains histone F1 and has a higher histone:dna ratio than macronuclear chromatin, there is no evidence that micronuclear chromatin is in any way deficient in its histone content FRACTION NUMBER FIG. 4. Buoyant density of macro- and micronuclear chromatin in CsCl. (A) [3H]Thymidine-labeled macronuclear chromatin () and ["4C]thymidine-labeled chromatin from salt-washed macronuclei (). (B) [3H]Thymidine-labeled micronuclear chromatin (-) and [14C]thymidine-labeled chromatin from salt-washed macronuclei (). The gradient contained 38.5% CsCl in.2 mm EDTA. Centrifugation was in a Spinco SW-5 rotor at 32, rpm for 63 hr at 18. Approximately 37, 3H counts and 19, 14C counts were in gradient A, while approximately 23, 3H counts and 18, 14C counts were in gradient B. when compared to macronuclear chromatin. It is more likely that histone F1 has been replaced in micronuclear chromatin by increased amounts of the other micronuclear histones (X, F2B, and unacetylated F2A1; the properties of histone F3 and Y that also seem to be absent in micronuclei will be discussed in a later communication). The observation that chromatin isolated from micronuclei does not contain any detectable histone fraction F1 reinforces our previous suggestion (1) that histone F1 and its phosphorylation do not function uniquely in the process of mitotic chromosome condensation (16, 21-23). Thus, histone fraction F1 is present and is phosphorylated in macronuclei, which divide amitotically, with little or no change in chromatin organization; F1 is absent in micronuclei, which divide mitotically and show distinct changes in chromatin condensation during the cell cycle (2-7). Similarly, the absence of histone F1 from micronuclear chromatin makes it unlikely that this histone fraction (or its phosphorylation) plays an indispensable role in the process of chromosome replication (18), or in the sorting out of sister chromatids (2). Recent reports (39, 4) that F1 is not found in the histone complement of some lower eukaryotes also support these conclusions. Of course, it is possible that, if present, F1 must be phosphorylated for any or all of these processes to occur. Our findings of extensive phosphorylation of F1 in macronuclei coupled with the absence of F1 in micronuclei would seem to argue against the recent suggestion that phosphorylation of F1 is an important step in the initiation of mitosis (21, 41). However, since macronuclei control the vegetative functions of Tetrahymena, it is still possible that phosphorylation of F1 plays some role in the control of cell divi-
5 2676 Cell Biology: Gorovsky and Keevert sion. Such a role, however, would have to be independent of whether the nuclear division occurs by a mitotic or an amitotic mechanism. Finally, because macro- and micronuclei show extreme differences in their genetic activity, we are tempted to suggest that histone F1 functions in the control of genetic activity in eukaryotic nuclei. Phosphorylation of histone F1 is clearly correlated with the rate of cell division in genetically active nuclei of a variety of cell types, including Tetrahymena (1-16). This process could be associated with an event, occurring once per cell cycle, during which the specific DNA sequences that code for RNA within a genome might be altered. Such a function for Fl-phosphorylation would account for its correlation with cell division and for the fact that it seems to occur at different times in the cell cycle in different cell types. It should be noted that the phosphorylation of F1 itself is probably not gene specific (2). It could, nonetheless, be a necessary step in changing the state of activity of specific genes. In this respect, it is interesting to note that cells frequently must undergo a normal division cycle before differentiating (42). This work was supported by National Science Foundation Grant GB Gorovsky, M. A. (1973) J. Protozool. 2, Elliott, A. M. (1963) in The Cell in Mitosis, ed. Levine, L. (Academic Press, New York), pp Flickinger, C. J. (1965) J. Cell Biol. 27, Nilsson, J. R. (197) J. Protozool. 17, Stevenson, I. & Lloyd, F. P. (1971) Aust. J. Biol. Sci. 24, Stevenson, I. & Lloyd, F. P. (1971) Aust. J. Biol. Sci. 24, Ray, C., Jr. (1956) J. Protozool. 3, Gorovsky, M. A. & Woodard, J. (1969) J. Cell Biol. 42, Murti, K. G. & Prescott, D. M. (197) J. Cell Biol. 47, Gorovsky, M. A., Keevert, J. B. & Pleger, G. L. (1974) J. Cell Biol. 61, Stevely, W. S. & Stocken, L. A. (1968) Biochem. J. 11, Sherod, D., Johnson, G. & Chalkley, R. (197) Biochemistry 9, Balhorn, R., Rieke, W.. & Chalkley, R. (1971) Biochemistry 1, Balhorn, R., Balhorn, M. & Chalkley, R. (1972) Dev. Biol. 29, Proc. Nat. Acad. Sci. USA 72 (1975) 15. Balhorn, R., Chalkley, R. & Granner, D. (1972) Biochemistry 11, Bradbury, E. M., Inglis, R. J., Matthews, H. R. & Sarner, N. (1973) Eur. J. Biochem. 33, Gurley, L. R., Walters, R. A. & Tobey, R. A. (1974) J. Cell Biol. 6, Oliver, D., Balhorn, R., Granner, D. & Chalkley, R. (1972) Biochemistry 11, Ord, M. B. & Stocken, L. A. (1969) Biochem. J. 112, Marks, D. B., Paik, W. K. & Borun, T. W. (1973) J. Biol. Chem. 248, Bradbury, E. M., Inglis, R. J. & Matthews, H. R. (1974) Nature 247, Lake, R. S. & Salzman, N. P. (1972) Biochemistry 11, Lake, R. S., Goidl, J. A. & Salzman, N. P. (1972) Exp. Cell Res. 73, Guttierrez-Cemosek, R. M. & Hnilica, L. S. (1971) Biochim. Biophys. Acta 247, Kleinsmith, L. J., Allfrey, V. G. & Mirsky, A. E. (1966) Proc. Nat. Acad. Sci. USA 55, Meisler, M. H. & Langan, T. A. (1969) J. Biol. Chem. 244, Gorovsky, M. A., Pleger, G. L., Keevert, J. B. & Johmann, C. A. (1973) J. Cell Biol. 57, Gorovsky, M. A. (197) J. Cell Biol. 47, Panyim, S., Bilek, D. & Chalkley, R. (1971) J. Biol. Chem. 246, Panyim, S. & Chalkley, R. (1969) Arch. Biochem. Biophys. 13, Gorovsky, M. A., Carlson, K. & Rosenbaum, J. L. (197) Anal. Biochem. 35, Ansevin, A. T. & Brown, B. W. (1971) Biochemistry 1, Shih, T. Y. & Bonner, J. (197) J. Mol. Biol. 48, Brutlag, D., Schlehuber, C. & Bonner, J. (1969) Biochemistry 8, Ilyin, Y. V. & Georgiev, G. P. (1969) J. Mol. Biol. 41, Bartley, J. & Chalkley, R. (197) J. Biol. Chem. 245, Ohlenbusch, H. H., Olivera, B. M., Tuan, D. & Davidson, N. (1967) J. Mol. Biol. 25, Murray, K. (1969) J. Mol. Biol. 39, Hsiang, M. W. & Cole, R. D. (1973) J. Biol. Chem. 248, Franco, L., Johns, E. W. & Navlet, J. M. (1974) Eur. J. Biochem. 45, Bradbury, E. M., Inglis, R. J., Matthews, H. R. & Langan, T. A. (1974) Nature 249, Holtzer, H., Weintraub, R., Mayne, R. & Mochan, B. (1972) Curr. Top. Dev. Biol. 7,
STUDIES ON NUCLEAR STRUCTURE AND FUNCTION IN TETRAHYMENA PYRIFORMIS
 STUDIES ON NUCLEAR STRUCTURE AND FUNCTION IN TETRAHYMENA PYRIFORMIS III. Comparison of the Histones of Macro- and Micronuclei by Quantitative Polyacrylamide Gel Electrophoresis MARTIN A. GOROVSKY From
STUDIES ON NUCLEAR STRUCTURE AND FUNCTION IN TETRAHYMENA PYRIFORMIS III. Comparison of the Histones of Macro- and Micronuclei by Quantitative Polyacrylamide Gel Electrophoresis MARTIN A. GOROVSKY From
ELECTROPHORETIC ANALYSIS OF LIVER AND TESTIS HISTONES OF THE FROG RANA PIPIENS
 ELECTROPHORETIC ANALYSIS OF LIVER AND TESTIS HISTONES OF THE FROG RANA PIPIENS DONNA ALDER and MARTIN A. GOROVSKY From the Department of Biology, University of Rochester, Rochester, New York 14627 ABSTRACT
ELECTROPHORETIC ANALYSIS OF LIVER AND TESTIS HISTONES OF THE FROG RANA PIPIENS DONNA ALDER and MARTIN A. GOROVSKY From the Department of Biology, University of Rochester, Rochester, New York 14627 ABSTRACT
Blocking by Histones of Accessibility to DNA in Chromatin (DNase/RNA polymerase/dna polymerase)
 Proc. Nat. Acad. Sci. USA Vol. 69, No. 8, pp. 2115-2119, August 1972 Blocking by Histones of Accessibility to in Chromatin (/RNA polymerase/ polymerase) ALFRED E. MIRSKY AND BERT SILVERMAN The Rockefeller
Proc. Nat. Acad. Sci. USA Vol. 69, No. 8, pp. 2115-2119, August 1972 Blocking by Histones of Accessibility to in Chromatin (/RNA polymerase/ polymerase) ALFRED E. MIRSKY AND BERT SILVERMAN The Rockefeller
Micronuclei of Tetrahymena contain two types of histone H3 (histone extraction/two-dimensional gel electrophoresis/nucleosome)
 P'roc. Natl. cad. Sci. US Vol. 76, No. 1, pp. 4857-4861, October 1979 iochemistry Micronuclei of Tetrahymena contain two types of histone H3 (histone extraction/two-dimensional gel electrophoresis/nucleosome)
P'roc. Natl. cad. Sci. US Vol. 76, No. 1, pp. 4857-4861, October 1979 iochemistry Micronuclei of Tetrahymena contain two types of histone H3 (histone extraction/two-dimensional gel electrophoresis/nucleosome)
PATTERN OF DNA INCREASE IN MACRONUCLEAR ANLAGEN OF TETRAHYMENA
 J. Cell Set. 83, 155-164 (1986) 155 Printed in Great Britain The Company of Biologists Limited 1986 PATTERN OF DNA INCREASE IN MACRONUCLEAR ANLAGEN OF TETRAHYMENA JOACHIM ROTH AND GUNTER CLEFFMANN* Institut
J. Cell Set. 83, 155-164 (1986) 155 Printed in Great Britain The Company of Biologists Limited 1986 PATTERN OF DNA INCREASE IN MACRONUCLEAR ANLAGEN OF TETRAHYMENA JOACHIM ROTH AND GUNTER CLEFFMANN* Institut
CYTOCHEMICAL STUDIES ON THE PROBLEM OF MACRONUCLEAR SUBNUCLEI IN TETRAHYMENA JOHN WOODARD, EDNA KANESHIROI AND MARTIN A.
 CYTOCHEMICAL STUDIES ON THE PROBLEM OF MACRONUCLEAR SUBNUCLEI IN TETRAHYMENA JOHN WOODARD, EDNA KANESHIROI AND MARTIN A. GOROVSKY+$ Whitman Laboratory, University of Chicago, Chicago, Illinois 60637 Manuscript
CYTOCHEMICAL STUDIES ON THE PROBLEM OF MACRONUCLEAR SUBNUCLEI IN TETRAHYMENA JOHN WOODARD, EDNA KANESHIROI AND MARTIN A. GOROVSKY+$ Whitman Laboratory, University of Chicago, Chicago, Illinois 60637 Manuscript
Sequential Phosphorylation of Histone Subfractions in the Chinese Hamster Cell Cycle*
 THE JOURNAL OF BIOLOGKXL CHEMISTRY Vol. 250, No. 10, Issue of May 25, pp. 3936-3944, 1975 Printed in U.S.A. Sequential Phosphorylation of Histone Subfractions in the Chinese Hamster Cell Cycle* (Received
THE JOURNAL OF BIOLOGKXL CHEMISTRY Vol. 250, No. 10, Issue of May 25, pp. 3936-3944, 1975 Printed in U.S.A. Sequential Phosphorylation of Histone Subfractions in the Chinese Hamster Cell Cycle* (Received
BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS
 HISTONE STOICHIOMETRY IN CHICKEN ERYTHROCYTE NUCLEI Everline B. Wright and Donald E. Olins University of Tennessee-Oak Ridge Graduate School of Biomedical Sciences Biology Division, Oak Ridge National
HISTONE STOICHIOMETRY IN CHICKEN ERYTHROCYTE NUCLEI Everline B. Wright and Donald E. Olins University of Tennessee-Oak Ridge Graduate School of Biomedical Sciences Biology Division, Oak Ridge National
This colorized scanning electron micrograph is showing a white blood cell undergoing cell division.
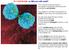 10 1 Cell Growth Why are cells small? This colorized scanning electron micrograph is showing a white blood cell undergoing cell division. This relates to cell theory because this is HOW all cells come
10 1 Cell Growth Why are cells small? This colorized scanning electron micrograph is showing a white blood cell undergoing cell division. This relates to cell theory because this is HOW all cells come
Physical properties of chemically acetylated rat liver chromatin (acetic anhydride/nucleosomes/thermal denaturation/histones)
 Proc. Nati. Acad. Sci. USA Vol. 74, No. 8, pp. 3244-3248, August 1977 Biochemistry Physical properties of chemically acetylated rat liver chromatin (acetic anhydride/nucleosomes/thermal denaturation/histones)
Proc. Nati. Acad. Sci. USA Vol. 74, No. 8, pp. 3244-3248, August 1977 Biochemistry Physical properties of chemically acetylated rat liver chromatin (acetic anhydride/nucleosomes/thermal denaturation/histones)
Single Essential Amino Acids (valine/histidine/methiotiine/high-temperature inhibition)
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 9, pp. 2057-2061, September 1971 Regulation of Protein Synthesis Initiation in HeLa Cells Deprived of Single ssential Amino Acids (valine/histidine/methiotiine/high-temperature
Proc. Nat. Acad. Sci. USA Vol. 68, No. 9, pp. 2057-2061, September 1971 Regulation of Protein Synthesis Initiation in HeLa Cells Deprived of Single ssential Amino Acids (valine/histidine/methiotiine/high-temperature
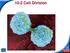
10-2 Cell Division. Chromosomes
 Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis.
Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis.
Synthesis of Proteins in Cells Infected with Herpesvirus,
 Proceedings of the National Academy of Science8 Vol. 66, No. 3, pp. 799-806, July 1970 Synthesis of Proteins in Cells Infected with Herpesvirus, VI. Characterization of the Proteins of the Viral Membrane*
Proceedings of the National Academy of Science8 Vol. 66, No. 3, pp. 799-806, July 1970 Synthesis of Proteins in Cells Infected with Herpesvirus, VI. Characterization of the Proteins of the Viral Membrane*
synthesis. Although such data strongly suggest that a protein
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1653-1657, July 1971 Mechanism of Action of Vitamin K: Evidence for the Conversion of a Precursor Protein to Prothrombin in the Rat (phylloquinone/baso4 adsorption/polyacrylamide
Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1653-1657, July 1971 Mechanism of Action of Vitamin K: Evidence for the Conversion of a Precursor Protein to Prothrombin in the Rat (phylloquinone/baso4 adsorption/polyacrylamide
Mitosis and Cytokinesis
 B-2.6 Summarize the characteristics of the cell cycle: interphase (called G1, S, G2); the phases of mitosis (called prophase, metaphase, anaphase, and telophase); and plant and animal cytokinesis. The
B-2.6 Summarize the characteristics of the cell cycle: interphase (called G1, S, G2); the phases of mitosis (called prophase, metaphase, anaphase, and telophase); and plant and animal cytokinesis. The
10-2 Cell Division mitosis. cytokinesis. Chromosomes chromosomes Slide 1 of 38
 In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis. Chromosomes
In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called cytokinesis. Chromosomes
NUCLEAR DIVISIONS WITH REDUCED NUMBERS OF MICROTUBULES IN TETRAHYMENA
 J. Cell Sci, 34, 303-319 (1978) 303 Printed in Great Britain Company of BiologiiU Limited NUCLEAR DIVISIONS WITH REDUCED NUMBERS OF MICROTUBULES IN TETRAHYMENA RUTH JAECKEL-WILLIAMS Department of Zoology,
J. Cell Sci, 34, 303-319 (1978) 303 Printed in Great Britain Company of BiologiiU Limited NUCLEAR DIVISIONS WITH REDUCED NUMBERS OF MICROTUBULES IN TETRAHYMENA RUTH JAECKEL-WILLIAMS Department of Zoology,
Prentice Hall Biology Slide 1 of 38
 Prentice Hall Biology 1 of 38 2 of 38 In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm,
Prentice Hall Biology 1 of 38 2 of 38 In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm,
N α -Acetylation of yeast ribosomal proteins and its effect on protein synthesis
 JOURNAL OF PROTEOMICS 74 (2011) 431 441 available at www.sciencedirect.com www.elsevier.com/locate/jprot N α -Acetylation of yeast ribosomal proteins and its effect on protein synthesis Masahiro Kamita
JOURNAL OF PROTEOMICS 74 (2011) 431 441 available at www.sciencedirect.com www.elsevier.com/locate/jprot N α -Acetylation of yeast ribosomal proteins and its effect on protein synthesis Masahiro Kamita
(i) List these events in the correct order, starting with D.... (1)... (1)... (1)
 Q1. (a) Boxes A to E show some of the events of the cell cycle. A Chromatids seperate B Nuclear envelopes disappears C Cytoplasm divides D Chromosomes condense and become visible E Chromosomes on the equator
Q1. (a) Boxes A to E show some of the events of the cell cycle. A Chromatids seperate B Nuclear envelopes disappears C Cytoplasm divides D Chromosomes condense and become visible E Chromosomes on the equator
10-2 Cell Division. Slide 1 of 38. End Show. Copyright Pearson Prentice Hall
 1 of 38 Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called
1 of 38 Cell Division In eukaryotes, cell division occurs in two major stages. The first stage, division of the cell nucleus, is called mitosis. The second stage, division of the cell cytoplasm, is called
Replication of Sindbis Virus V. Polyribosomes and mrna in Infected Cells
 JOURNAL OF VIROLOGY, Sept. 1974, p. 552-559 Vol. 14, No. 3 Copyright @ 1974 American Society for Microbiology Printed in U.S.A. Replication of Sindbis Virus V. Polyribosomes and mrna in Infected Cells
JOURNAL OF VIROLOGY, Sept. 1974, p. 552-559 Vol. 14, No. 3 Copyright @ 1974 American Society for Microbiology Printed in U.S.A. Replication of Sindbis Virus V. Polyribosomes and mrna in Infected Cells
THE CELL CYCLE. 1. What is meant by the cell cycle or cell division cycle?
 THE CELL CYCLE The cell cycle, or cell-division cycle, is the series of events that take place in a eukaryotic cell between its formation and the moment it replicates itself. These events can be divided
THE CELL CYCLE The cell cycle, or cell-division cycle, is the series of events that take place in a eukaryotic cell between its formation and the moment it replicates itself. These events can be divided
TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
 Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in vitro
 Radiat Environ Biophys (1986) 25:195-199 Radiation and Environmental Biophysics Springer-Verlag 1986 Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in
Radiat Environ Biophys (1986) 25:195-199 Radiation and Environmental Biophysics Springer-Verlag 1986 Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in
Histone H1 subtype synthesis in neurons and neuroblasts
 Development 115, 181-185 (1992) Printed m Great Britain The Company of Biologists Limited 1992 181 Histone H1 subtype synthesis in neurons and neuroblasts VALENTfN DOMfNGUEZ, BENJAMIN PINA and PEDRO SUAU*
Development 115, 181-185 (1992) Printed m Great Britain The Company of Biologists Limited 1992 181 Histone H1 subtype synthesis in neurons and neuroblasts VALENTfN DOMfNGUEZ, BENJAMIN PINA and PEDRO SUAU*
Regulators of Cell Cycle Progression
 Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
The Process of Cell Division
 Lesson Overview 10.2 The Process of Cell Division THINK ABOUT IT What role does cell division play in your life? Does cell division stop when you are finished growing? Chromosomes What is the role of chromosomes
Lesson Overview 10.2 The Process of Cell Division THINK ABOUT IT What role does cell division play in your life? Does cell division stop when you are finished growing? Chromosomes What is the role of chromosomes
U3.2.3: Eukaryotic chromosomes are linear DNA molecules associated with histone proteins. (Oxford Biology Course Companion page 151).
 Cell Division Study Guide U3.2.3: Eukaryotic chromosomes are linear DNA molecules associated with histone proteins. (Oxford Biology Course Companion page 151). 1. Describe the structure of eukaryotic DNA
Cell Division Study Guide U3.2.3: Eukaryotic chromosomes are linear DNA molecules associated with histone proteins. (Oxford Biology Course Companion page 151). 1. Describe the structure of eukaryotic DNA
Yeast Ribosomal Proteins are Synthesized on Small Polysomes
 Eur. J. Biochem. 62, 193-197 (1976) Yeast Ribosomal Proteins are Synthesized on Small Polysomes Willem H. MAGER and Rudi J. PLANTA Biochemisch Laboratorium, Vrije Universiteit, Amsterdam (Received September
Eur. J. Biochem. 62, 193-197 (1976) Yeast Ribosomal Proteins are Synthesized on Small Polysomes Willem H. MAGER and Rudi J. PLANTA Biochemisch Laboratorium, Vrije Universiteit, Amsterdam (Received September
T for sexual reproduction, is frequently lost from the
 . DEFECTIVE MICRONUCLEI OF Tetrahymena 509 19. Sagar, P., Krishna Murti, C. R. & Shrivastava, D. L. 1958. 23. Stavy, L., Feldman, M. & Elson, D. 1964. On ribonu- Studies in enzyme make up of Vibrio cholerae.
. DEFECTIVE MICRONUCLEI OF Tetrahymena 509 19. Sagar, P., Krishna Murti, C. R. & Shrivastava, D. L. 1958. 23. Stavy, L., Feldman, M. & Elson, D. 1964. On ribonu- Studies in enzyme make up of Vibrio cholerae.
Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples:
 Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Fig. 1. Simple columnar epithelial cells lining the small intestine.
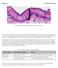 Mitosis Prelab Reading Fig. 1. Simple columnar epithelial cells lining the small intestine. The tall cells pictured in Fig. 1 form the lining of the small intestine in humans and other animals. These cells
Mitosis Prelab Reading Fig. 1. Simple columnar epithelial cells lining the small intestine. The tall cells pictured in Fig. 1 form the lining of the small intestine in humans and other animals. These cells
Why do cells divide? Cells divide in order to make more cells they multiply in order to create a larger surface to volume ratio!!!
 Why do cells divide? Cells divide in order to make more cells they multiply in order to create a larger surface to volume ratio!!! Chromosomes Are made of chromatin: a mass of genetic material composed
Why do cells divide? Cells divide in order to make more cells they multiply in order to create a larger surface to volume ratio!!! Chromosomes Are made of chromatin: a mass of genetic material composed
The Cell Cycle. Dr. SARRAY Sameh, Ph.D
 The Cell Cycle Dr. SARRAY Sameh, Ph.D Overview When an organism requires additional cells (either for growth or replacement of lost cells), new cells are produced by cell division (mitosis) Somatic cells
The Cell Cycle Dr. SARRAY Sameh, Ph.D Overview When an organism requires additional cells (either for growth or replacement of lost cells), new cells are produced by cell division (mitosis) Somatic cells
The Cell Cycle and Cell Division
 Content Vocabulary Directions: On each line, write the term from the word bank that correctly replaces the underlined words in each sentence. NOTE: You may need to change a term to its plural form. cell
Content Vocabulary Directions: On each line, write the term from the word bank that correctly replaces the underlined words in each sentence. NOTE: You may need to change a term to its plural form. cell
KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions.
 5.1 The Cell Cycle KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Objective: Cells have distinct phases of growth, reproduction and normal functions. APK: Why do
5.1 The Cell Cycle KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Objective: Cells have distinct phases of growth, reproduction and normal functions. APK: Why do
The Cell Cycle. Chapter 10
 The Cell Cycle Chapter 10 Why Do Cells Divide? Unicellular 1. Reproduction Multicellular 1. Grow 2. Repair 3. Development/reproduction Types of Division Prokaryotic cells Binary fission = asexual reproduction
The Cell Cycle Chapter 10 Why Do Cells Divide? Unicellular 1. Reproduction Multicellular 1. Grow 2. Repair 3. Development/reproduction Types of Division Prokaryotic cells Binary fission = asexual reproduction
10-2 Cell Division. Copyright Pearson Prentice Hall
 10-2 Cell Division Copyright Pearson Prentice Hall Cell Growth and Division In multicellular organisms, cell division makes new cells To replace old or damaged ones So organisms can grow In single-celled
10-2 Cell Division Copyright Pearson Prentice Hall Cell Growth and Division In multicellular organisms, cell division makes new cells To replace old or damaged ones So organisms can grow In single-celled
Nascent Subribosomal Particles in Tetrahymena pyrqormis
 Eur. J. Biochem. 13 (1970) 238-246 Nascent Subribosomal Particles in Tetrahymena pyrqormis Vagn LEICK and Jan ENCBERG Biokemisk Institut B, Krabenhavns Universitet Julius EMMERSEN Institut for Molekylax
Eur. J. Biochem. 13 (1970) 238-246 Nascent Subribosomal Particles in Tetrahymena pyrqormis Vagn LEICK and Jan ENCBERG Biokemisk Institut B, Krabenhavns Universitet Julius EMMERSEN Institut for Molekylax
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
Chapter 10 Cell Growth and Division
 Chapter 10 Cell Growth and Division 10 1 Cell Growth 2 Limits to Cell Growth The larger a cell becomes, the more demands the cell places on its DNA. In addition, the cell has more trouble moving enough
Chapter 10 Cell Growth and Division 10 1 Cell Growth 2 Limits to Cell Growth The larger a cell becomes, the more demands the cell places on its DNA. In addition, the cell has more trouble moving enough
Depleting Lipoproteins from Serum
 Depleting Lipoproteins from Serum Kathy K. Foxx Kalen Biomedical LLC President For decades, fetal bovine serum (FBS) has been used as a supplement for cell-culture media, providing the growth factors that
Depleting Lipoproteins from Serum Kathy K. Foxx Kalen Biomedical LLC President For decades, fetal bovine serum (FBS) has been used as a supplement for cell-culture media, providing the growth factors that
Title. Author(s)TAKAYANAGI, Tan. Issue Date Doc URL. Type. File Information. Mitomycin-C, Azan, MEPA and Podophyllin (With 16 Tex
 Title Cytological and Cytogenetical Studies on Paramecium Mitomycin-C, Azan, MEPA and Podophyllin (With 16 Tex Author(s)TAKAYANAG, Tan 北海道大學理學部紀要 = JOURNAL OF THE FACULTY OF SCENCE HOKKA CitationZOOLOGY,
Title Cytological and Cytogenetical Studies on Paramecium Mitomycin-C, Azan, MEPA and Podophyllin (With 16 Tex Author(s)TAKAYANAG, Tan 北海道大學理學部紀要 = JOURNAL OF THE FACULTY OF SCENCE HOKKA CitationZOOLOGY,
Cell Growth, Division, & Reproduction
 Cell Growth, Division, & Reproduction Two main reasons why cells divide rather than continue growing: A larger cell places more demand on the DNA. When a cell s size increases, its DNA does not & the extra
Cell Growth, Division, & Reproduction Two main reasons why cells divide rather than continue growing: A larger cell places more demand on the DNA. When a cell s size increases, its DNA does not & the extra
(a) Reproduction. (b) Growth and development. (c) Tissue renewal
 100 µm 200 µm 20 µm (a) Reproduction (b) Growth and development (c) Tissue renewal 1 20 µm 2 0.5 µm Chromosomes DNA molecules Chromosome arm Centromere Chromosome duplication (including DNA synthesis)
100 µm 200 µm 20 µm (a) Reproduction (b) Growth and development (c) Tissue renewal 1 20 µm 2 0.5 µm Chromosomes DNA molecules Chromosome arm Centromere Chromosome duplication (including DNA synthesis)
MITOSIS INTRODUCTION. Cytokinesis. centromere. DNA Replication S-Phase. One Chromosome Two Chromatids. One Chromosome No Chromatids 10.
 MITOSIS INTRODUCTION Cell reproduction usually involves two processes: 1) mitosis is the orderly separation and division of chromosomes in the nucleus and 2) cytokinesis is the division of the cytoplasm.
MITOSIS INTRODUCTION Cell reproduction usually involves two processes: 1) mitosis is the orderly separation and division of chromosomes in the nucleus and 2) cytokinesis is the division of the cytoplasm.
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
BIOLOGY 111. CHAPTER 9: The Links in Life s Chain Genetics and Cell Division
 BIOLOGY 111 CHAPTER 9: The Links in Life s Chain Genetics and Cell Division The Links in Life s Chain: Genetics and Cell Division 9.1 An Introduction to Genetics 9.2 An Introduction to Cell Division 9.3
BIOLOGY 111 CHAPTER 9: The Links in Life s Chain Genetics and Cell Division The Links in Life s Chain: Genetics and Cell Division 9.1 An Introduction to Genetics 9.2 An Introduction to Cell Division 9.3
Chromosomes & Cell Division
 Chromosomes & Cell Division Cell Division The growth and splitting of cells into two new, identical cells called daughter cells. Mitosis Meiosis DNA replicates Parent cell Chromosomes separate Cell division
Chromosomes & Cell Division Cell Division The growth and splitting of cells into two new, identical cells called daughter cells. Mitosis Meiosis DNA replicates Parent cell Chromosomes separate Cell division
Timing of commitment to autogamy in Paramecium bursaria
 Jpn. J. Protozool. Vol. 43, No. 2. (2010) 95 Short Communication Timing of commitment to autogamy in Paramecium bursaria Akira YANAGI Department of Biotechnology, Senshu University of Ishinomaki, Ishinomaki
Jpn. J. Protozool. Vol. 43, No. 2. (2010) 95 Short Communication Timing of commitment to autogamy in Paramecium bursaria Akira YANAGI Department of Biotechnology, Senshu University of Ishinomaki, Ishinomaki
Cell Cycle/Mitosis -Notes-
 Cell Cycle/Mitosis -Notes- LIMITS TO CELL GROWTH The a cell becomes, the more demands the cell places on DNA. Additionally, the cell has more trouble moving enough and wastes across the cell membrane.
Cell Cycle/Mitosis -Notes- LIMITS TO CELL GROWTH The a cell becomes, the more demands the cell places on DNA. Additionally, the cell has more trouble moving enough and wastes across the cell membrane.
H ISTON E acetylation, the acetylation of specific lysines
 Antibodies Specific to Acetylated Histones Document the Existence of Deposition- and Transcription-related Histone Acetylation in Tetrahymena Rueyling Lin, Joseph W. Leone,* Richard G. Cook,* and C. David
Antibodies Specific to Acetylated Histones Document the Existence of Deposition- and Transcription-related Histone Acetylation in Tetrahymena Rueyling Lin, Joseph W. Leone,* Richard G. Cook,* and C. David
Macronuclear Regeneration in Epistylis articulata
 Macronuclear Regeneration in Epistylis articulata By B. R. SESHACHAR AND C. M. S. DASS (From the Department of Zoology, University of Mysore, Central College, Bangalore) With one plate (fig. 5) SUMMARY
Macronuclear Regeneration in Epistylis articulata By B. R. SESHACHAR AND C. M. S. DASS (From the Department of Zoology, University of Mysore, Central College, Bangalore) With one plate (fig. 5) SUMMARY
MITOSIS IN ONION ROOTLET CELLS
 Lesson 6: CELL CYCLE, MITOSIS Name: Group: MITOSIS IN ONION ROOTLET CELLS Permanent slide: onion rootlet stained with acetorcein The particular mitotic phases are visible in the onion rootlet cells. Chromosomes
Lesson 6: CELL CYCLE, MITOSIS Name: Group: MITOSIS IN ONION ROOTLET CELLS Permanent slide: onion rootlet stained with acetorcein The particular mitotic phases are visible in the onion rootlet cells. Chromosomes
Mitosis. AND Cell DiVISION
 Mitosis AND Cell DiVISION Cell Division Characteristic of living things: ability to reproduce their own kind. Cell division purpose: When unicellular organisms such as amoeba divide to form offspring reproduction
Mitosis AND Cell DiVISION Cell Division Characteristic of living things: ability to reproduce their own kind. Cell division purpose: When unicellular organisms such as amoeba divide to form offspring reproduction
Organization of genetic material in eukaryotes
 Organization of genetic material in eukaryotes biologiemoleculara.usmf.md pass.: bmgu e.usmf.md 1 DNA in eukaryotes Location: In nucleus In mitochondria biologiemoleculara.usmf.md e.usmf.md pass.: bmgu
Organization of genetic material in eukaryotes biologiemoleculara.usmf.md pass.: bmgu e.usmf.md 1 DNA in eukaryotes Location: In nucleus In mitochondria biologiemoleculara.usmf.md e.usmf.md pass.: bmgu
9/3/2009 DNA i DNA n euk euk yotes Organizatio Organ izatio n of o f gen ge e n tic Locati t on: In n ucleu e s material mater in e ial
 DNA in eukaryotes Organization of genetic material in eukaryotes Location: In nucleus In mitochondria DNA in eukaryotes Nuclear DNA: Long, linear molecules; Chromatin chromosomes; 10% of DNA in genes,
DNA in eukaryotes Organization of genetic material in eukaryotes Location: In nucleus In mitochondria DNA in eukaryotes Nuclear DNA: Long, linear molecules; Chromatin chromosomes; 10% of DNA in genes,
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Chromosomes Chromosomes were first observed by the German embryologist Walther Fleming in 1882. Chromosome number varies among organisms most
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
The So-called Non-Histones from Acid-Treated Calf Thymus Chromatin
 Eur. J. Biochem. 16 (1970) GO-65 The So-called Non-Histones from Acid-Treated Calf Thymus Chromatin Johann SONNENBICHLER and Peter NOBIS Max-Planck-Institut fur Biochernie, Munchen (Received April 3/June
Eur. J. Biochem. 16 (1970) GO-65 The So-called Non-Histones from Acid-Treated Calf Thymus Chromatin Johann SONNENBICHLER and Peter NOBIS Max-Planck-Institut fur Biochernie, Munchen (Received April 3/June
The Cell Cycle. Packet #9. Thursday, August 20, 2015
 1 The Cell Cycle Packet #9 2 Introduction Cell Cycle An ordered sequence of events in the life of a dividing eukaryotic cell and is a cellular asexual reproduction. The contents of the parent s cell nucleus
1 The Cell Cycle Packet #9 2 Introduction Cell Cycle An ordered sequence of events in the life of a dividing eukaryotic cell and is a cellular asexual reproduction. The contents of the parent s cell nucleus
virus-i (RAV-1) or Rous associated virus-2 (RAV-2), do not transform but do produce
 ISOLATION OF NONINFECTIOUS PARTICLES CONTAINING ROUS SARCOMA VIRUS RNA FROM THE MEDIUM OF ROUS SARCOMA VIRUS-TRANSFORMED NONPRODUCER CELLS* BY HARRIET LATHAM ROBINSONt VIRUS LABORATORY, UNIVERSITY OF CALIFORNIA,
ISOLATION OF NONINFECTIOUS PARTICLES CONTAINING ROUS SARCOMA VIRUS RNA FROM THE MEDIUM OF ROUS SARCOMA VIRUS-TRANSFORMED NONPRODUCER CELLS* BY HARRIET LATHAM ROBINSONt VIRUS LABORATORY, UNIVERSITY OF CALIFORNIA,
Why do cells reproduce?
 Outline Cell Reproduction 1. Overview of Cell Reproduction 2. Cell Reproduction in Prokaryotes 3. Cell Reproduction in Eukaryotes 1. Chromosomes 2. Cell Cycle 3. Mitosis and Cytokinesis Examples of Cell
Outline Cell Reproduction 1. Overview of Cell Reproduction 2. Cell Reproduction in Prokaryotes 3. Cell Reproduction in Eukaryotes 1. Chromosomes 2. Cell Cycle 3. Mitosis and Cytokinesis Examples of Cell
Nature Genetics: doi: /ng Supplementary Figure 1. Assessment of sample purity and quality.
 Supplementary Figure 1 Assessment of sample purity and quality. (a) Hematoxylin and eosin staining of formaldehyde-fixed, paraffin-embedded sections from a human testis biopsy collected concurrently with
Supplementary Figure 1 Assessment of sample purity and quality. (a) Hematoxylin and eosin staining of formaldehyde-fixed, paraffin-embedded sections from a human testis biopsy collected concurrently with
CELL-CYCLE-DEPENDENT EFFECTS OF SODIUM-n-BUTYRATE IN PHYSARUM POLYCEPHALUM
 J. Cell Sri. 58, 303-3" (1982) 303 Printed in Great Britain Company of Biologists Limited 1982 CELL-CYCLE-DEPENDENT EFFECTS OF SODIUM-n-BUTYRATE IN PHYSARUM POLYCEPHALUM PETER LOIDL, PETER GROBNER, ADAM
J. Cell Sri. 58, 303-3" (1982) 303 Printed in Great Britain Company of Biologists Limited 1982 CELL-CYCLE-DEPENDENT EFFECTS OF SODIUM-n-BUTYRATE IN PHYSARUM POLYCEPHALUM PETER LOIDL, PETER GROBNER, ADAM
Cell Division. The Process of Cell Division Section Section 10.2: The Process of Cell Division 12/8/2010
 The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
Almost every cell in the human body has an identical set of 46 chromosomes, produced through the process of mitosis.
 M I T O S I S Mitosis Mitosis is the type of cell division that occurs for growth (adding new cells) and repair (replacing old or damaged cells). It results in two daughter cells that have identical chromosomes
M I T O S I S Mitosis Mitosis is the type of cell division that occurs for growth (adding new cells) and repair (replacing old or damaged cells). It results in two daughter cells that have identical chromosomes
Cell Cycle Notes --PreAP
 Cell Cycle Notes --PreAP I. DNA Deoxyribonucleic acid; located in nucleus A. Long and thread-like DNA in a non-dividing cell B. Thick, short, coiled doubled DNA in a dividing cell chromosome 1. chromosome
Cell Cycle Notes --PreAP I. DNA Deoxyribonucleic acid; located in nucleus A. Long and thread-like DNA in a non-dividing cell B. Thick, short, coiled doubled DNA in a dividing cell chromosome 1. chromosome
Cell Growth and Division
 Cell Growth and Division CHAPTER 10 EQ Why do cells grow and divide? DID YOU KNOW THAT The adult human body produces roughly 2 TRILLION cells every day. WHY DO CELLS REPRODUCE? So that the organism can
Cell Growth and Division CHAPTER 10 EQ Why do cells grow and divide? DID YOU KNOW THAT The adult human body produces roughly 2 TRILLION cells every day. WHY DO CELLS REPRODUCE? So that the organism can
Unit 6: Study Guide Cell Division. diploid gene allele interphase (G1, S, G2) prophase metaphase anaphase
 Unit 6: Study Guide Cell Division 1. Define: chromatin chromosome chromatid pair (sister chromatid) centromere spindle fibers haploid diploid gene allele interphase (G1, S, G2) prophase metaphase anaphase
Unit 6: Study Guide Cell Division 1. Define: chromatin chromosome chromatid pair (sister chromatid) centromere spindle fibers haploid diploid gene allele interphase (G1, S, G2) prophase metaphase anaphase
CHAPTER 8: CELL GROWTH AND DIVISION 8-1: CELL GROWTH 8-2: CELL DIVISION: MITOSIS AND CYTOKINESIS
 CHAPTER 8: CELL GROWTH AND DIVISION 8-1: CELL GROWTH 8-2: CELL DIVISION: MITOSIS AND CYTOKINESIS 1 LEARNING OBJECTIVES You should be able to: Give two physical reasons why mitosis must occur. Draw a chromosome
CHAPTER 8: CELL GROWTH AND DIVISION 8-1: CELL GROWTH 8-2: CELL DIVISION: MITOSIS AND CYTOKINESIS 1 LEARNING OBJECTIVES You should be able to: Give two physical reasons why mitosis must occur. Draw a chromosome
Chromosomes and Cell Cycle
 Chromosomes and Cell Cycle Cell Basics There are trillions of cells in your body Cells are microscopic Cells have DNA inside a structure called the nucleus The nucleus is enclosed by a structure called
Chromosomes and Cell Cycle Cell Basics There are trillions of cells in your body Cells are microscopic Cells have DNA inside a structure called the nucleus The nucleus is enclosed by a structure called
Omnis cellula e cellula
 Chapter 12 The Cell Cycle Omnis cellula e cellula 1855- Rudolf Virchow German scientist all cells arise from a previous cell Every cell from a cell In order for this to be true, cells must have the ability
Chapter 12 The Cell Cycle Omnis cellula e cellula 1855- Rudolf Virchow German scientist all cells arise from a previous cell Every cell from a cell In order for this to be true, cells must have the ability
Chapter 12 The Cell Cycle
 Chapter 12 The Cell Cycle Objectives Describe how cell reproduction contributes to repair and growth. Compare and contrast prokaryotic and eukaryotic cell division. Compare and contrast asexual and sexual
Chapter 12 The Cell Cycle Objectives Describe how cell reproduction contributes to repair and growth. Compare and contrast prokaryotic and eukaryotic cell division. Compare and contrast asexual and sexual
-The cell s hereditary endowment of DNA -Usually packaged into chromosomes for manageability
 Binary Fission-Bacterial Cell Division -Asexual reproduction of prokaryotes -No mitosis -Circular DNA and organelles replicate, the copies migrate to opposite sides of the elongating cell, and the cell
Binary Fission-Bacterial Cell Division -Asexual reproduction of prokaryotes -No mitosis -Circular DNA and organelles replicate, the copies migrate to opposite sides of the elongating cell, and the cell
Mitosis. An Introduction to Genetics. An Introduction to Cell Division
 Mitosis An Introduction to Genetics An Introduction to Cell Division DNA is Packaged in Chromosomes Cell Cycle Mitosis and Cytokinesis Variations in Cell Division Cell Division and Cancer An Introduction
Mitosis An Introduction to Genetics An Introduction to Cell Division DNA is Packaged in Chromosomes Cell Cycle Mitosis and Cytokinesis Variations in Cell Division Cell Division and Cancer An Introduction
CELL CYCLE INTRODUCTION PART I ANIMAL CELL CYCLE INTERPHASE
 CELL CYCLE INTRODUCTION The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes, discrete units of hereditary information consisting of double-stranded DNA. Structural proteins
CELL CYCLE INTRODUCTION The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes, discrete units of hereditary information consisting of double-stranded DNA. Structural proteins
Cell Division. Introduction. Chromatin, Chromosomes, and Chromatids, Oh My! The Cell Cycle
 Introduction Cell Division Just like a butterfly passes through different phases (such as caterpillar, chrysalis, and adult butterfly) there are a series of phases in a cell's life as it gets ready to
Introduction Cell Division Just like a butterfly passes through different phases (such as caterpillar, chrysalis, and adult butterfly) there are a series of phases in a cell's life as it gets ready to
5.1. KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. 68 Reinforcement Unit 2 Resource Book
 5.1 THE CELL CYCLE KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Cells have a regular pattern of growth, DNA duplication, and division that is called the cell cycle.
5.1 THE CELL CYCLE KEY CONCEPT Cells have distinct phases of growth, reproduction, and normal functions. Cells have a regular pattern of growth, DNA duplication, and division that is called the cell cycle.
DR. RAMESH U2 L3. MITOSIS and Cell Cycle
 DR. RAMESH U2 L3 MITOSIS and Cell Cycle Do Now! Directions: Answer the following questions in complete sentences. 1. Describe how bacteria reproduce. Describe how cats reproduce.are there any similarities
DR. RAMESH U2 L3 MITOSIS and Cell Cycle Do Now! Directions: Answer the following questions in complete sentences. 1. Describe how bacteria reproduce. Describe how cats reproduce.are there any similarities
Mitosis THE CELL CYCLE. In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for..
 Mitosis THE CELL CYCLE In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for.. Development from a fertilized cell Growth Repair Cell
Mitosis THE CELL CYCLE In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for.. Development from a fertilized cell Growth Repair Cell
Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division
 The Cell Cycle and Cellular Reproduction Chapter 9 Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division 1 2 Interphase
The Cell Cycle and Cellular Reproduction Chapter 9 Outline Interphase Mitotic Stage Cell Cycle Control Apoptosis Mitosis Mitosis in Animal Cells Cytokinesis Cancer Prokaryotic Cell Division 1 2 Interphase
Biology is the only subject in which multiplication is the same thing as division. AP Biology
 Biology is the only subject in which multiplication is the same thing as division Chapter 12. The Cell Cycle: Cell Growth, Cell Division Where it all began You started as a cell smaller than a period at
Biology is the only subject in which multiplication is the same thing as division Chapter 12. The Cell Cycle: Cell Growth, Cell Division Where it all began You started as a cell smaller than a period at
Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008
 Cell Theory 1 Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008 Cell theory: All organisms are made of cells All cells arise from preexisting cells How do new cells arise? Cell division the reproduction
Cell Theory 1 Cellular Reproduction, Part 1: Mitosis Lecture 10 Fall 2008 Cell theory: All organisms are made of cells All cells arise from preexisting cells How do new cells arise? Cell division the reproduction
[4 X 108 plaque-forming units (PFU)/ml] except in the experiment
![[4 X 108 plaque-forming units (PFU)/ml] except in the experiment [4 X 108 plaque-forming units (PFU)/ml] except in the experiment](/thumbs/82/84890494.jpg) Proc. Nat. Acad. Sci. USA Vol. 69, No. 9, pp. 2404-2409, September 1972 Extensive Symmetrical Transcription of Simian Virus 40 DNA in Virus-Yielding Cells (SV40/monkey cells/actinomycin D/RNase/hybridization)
Proc. Nat. Acad. Sci. USA Vol. 69, No. 9, pp. 2404-2409, September 1972 Extensive Symmetrical Transcription of Simian Virus 40 DNA in Virus-Yielding Cells (SV40/monkey cells/actinomycin D/RNase/hybridization)
Section Cell Growth. A. Limits to Cell Growth 1. DNA Overload 2. Exchanging Materials 3. Ratio of Surface Area to Volume 4.
 Getting Through Materials move through cells by diffusion. Oxygen and food move into cells, while waste products move out of cells. How does the size of a cell affect how efficiently materials get to all
Getting Through Materials move through cells by diffusion. Oxygen and food move into cells, while waste products move out of cells. How does the size of a cell affect how efficiently materials get to all
2.1 The Importance of Cell Division
 2.1 The Importance of Cell Division Functions of cell division Growth Repair Reproduction Growth All organisms begin as a single cell. Cell divisions will increase as an organism s size increases. There
2.1 The Importance of Cell Division Functions of cell division Growth Repair Reproduction Growth All organisms begin as a single cell. Cell divisions will increase as an organism s size increases. There
Mitosis and the Cell Cycle
 Mitosis and the Cell Cycle Chapter 12 The Cell Cycle: Cell Growth & Cell Division Where it all began You started as a cell smaller than a period at the end of a sentence Getting from there to here Cell
Mitosis and the Cell Cycle Chapter 12 The Cell Cycle: Cell Growth & Cell Division Where it all began You started as a cell smaller than a period at the end of a sentence Getting from there to here Cell
Mammalian Melanosomal Proteins: Characterization by Polyacrylamide Gel Electrophoresis
 YALE JOURNAL OF BIOLOGY AND MEDICINE 46, 553-559 (1973) Mammalian Melanosomal Proteins: Characterization by Polyacrylamide Gel Electrophoresis VINCENT J. HEARING AND MARVIN A. LUTZNER Dermatology Branch,
YALE JOURNAL OF BIOLOGY AND MEDICINE 46, 553-559 (1973) Mammalian Melanosomal Proteins: Characterization by Polyacrylamide Gel Electrophoresis VINCENT J. HEARING AND MARVIN A. LUTZNER Dermatology Branch,
Regulation of Gene Expression in Eukaryotes
 Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit User Manual (v5)
 Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit Catalog number: SM-005 Description Minute TM plasma membrane (PM) protein isolation kit is a novel and patented native PM protein
Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit Catalog number: SM-005 Description Minute TM plasma membrane (PM) protein isolation kit is a novel and patented native PM protein
The Cell Cycle CHAPTER 12
 The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
Part I: The Cell Cycle
 Cellular Differentiation Part I: The Cell Cycle During your lifetime, trillions of your cells will undergo the cell cycle. This process allows you to grow, heal, and maintain your vital tissues and organs.
Cellular Differentiation Part I: The Cell Cycle During your lifetime, trillions of your cells will undergo the cell cycle. This process allows you to grow, heal, and maintain your vital tissues and organs.
Chapter 8: Cellular Reproduction
 Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
Chapter 8: Cellular Reproduction 1. The Cell Cycle 2. Mitosis 3. Meiosis 2 Types of Cell Division 2n 1n Mitosis: occurs in somatic cells (almost all cells of the body) generates cells identical to original
An Introduction to Genetics. 9.1 An Introduction to Genetics. An Introduction to Genetics. An Introduction to Genetics. DNA Deoxyribonucleic acid
 An Introduction to Genetics 9.1 An Introduction to Genetics DNA Deoxyribonucleic acid Information blueprint for life Reproduction, development, and everyday functioning of living things Only 2% coding
An Introduction to Genetics 9.1 An Introduction to Genetics DNA Deoxyribonucleic acid Information blueprint for life Reproduction, development, and everyday functioning of living things Only 2% coding
Cell Cycle and Mitosis
 Cell Cycle and Mitosis Name Period A# THE CELL CYCLE The cell cycle, or cell-division cycle, is the series of events that take place in a eukaryotic cell between its formation and the moment it replicates
Cell Cycle and Mitosis Name Period A# THE CELL CYCLE The cell cycle, or cell-division cycle, is the series of events that take place in a eukaryotic cell between its formation and the moment it replicates
Cell cycle and apoptosis
 Cell cycle and apoptosis Cell cycle Definition Stages and steps Cell cycle Interphase (G1/G0, S, and G2) Mitosis (prophase, metaphase, anaphase, telophase, karyokinesis, cytokinesis) Control checkpoints
Cell cycle and apoptosis Cell cycle Definition Stages and steps Cell cycle Interphase (G1/G0, S, and G2) Mitosis (prophase, metaphase, anaphase, telophase, karyokinesis, cytokinesis) Control checkpoints
note on methodology I
 note on methodology I isolated per tube, and the preparation is very dilute and needs to be concentrated. We present here some modifications to this method in order to prepare large volumes of concentrated
note on methodology I isolated per tube, and the preparation is very dilute and needs to be concentrated. We present here some modifications to this method in order to prepare large volumes of concentrated
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis
 Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
Breaking Up is Hard to Do (At Least in Eukaryotes) Mitosis Prokaryotes Have a Simpler Cell Cycle Cell division in prokaryotes takes place in two stages, which together make up a simple cell cycle 1. Copy
