... J Clin Pathol 2006;59: doi: /jcp
|
|
|
- Anissa Heath
- 5 years ago
- Views:
Transcription
1 1238 MY APPROACH Diagnosis of thymoma S Suster... The diagnosis of thymic epithelial neoplasm has been a topic of controversy for many years. Reasons for this include the lack of predictive value associated with the morphology of these tumours and the multiplicity of classification schemes and terminologies proposed over the years. Recently, a new classification schema was introduced by the World Health Organization (WHO) in an attempt to standardise nomenclature and facilitate the diagnosis of primary thymic epithelial neoplasms. This schema, although not originally intended as a new histological classification, but rather as a means for translating equivalent terms from the various existing classifications, has represented a major step forward in this direction. However, problems still exist with the WHO schema, particularly with some of the criteria for the various histological subtypes as well as with issues of interobserver reproducibility. For this reason, we favour using a much more simplified approach to the morphological classification of thymic epithelial neoplasms. A personal approach to the morphological diagnosis of thymoma is described, with a brief explanation for the rationale for simplifying the existing diagnostic categories Correspondence to: S Suster, Department of Pathology, Ohio State University, E-411 Doan Hall, 410 West 10th Avenue, Columbus, OH 43210, USA; saul.suster@odumc.edu Accepted 6 February 2006 Published Online First 5 May T he morphological diagnosis of thymoma has traditionally posed difficulties for pathologists because of the great histological variability and intratumoral heterogeneity of these tumours. A cogent and easily reproducible morphological classification for these tumors has therefore been difficult to conceptualise. Many attempts were made in the past to devise a classification based on pure morphology (ie, shape and proportion of the tumour cells), histogenesis and immunophenotype. Most of these approaches have proved cumbersome, difficult to reproduce in clinical practice and, in general, have failed to provide a sensible solution to the problem. Currently, the most widely used approach to the classification of thymic epithelial neoplasms is that proposed by the World Health Organization (WHO). 1 2 We will first review the rationale and basis for this approach. WHO CLASSIFICATION OF THYMOMA The WHO classification of thymic epithelial neoplasms was first introduced in This schema was the result of extensive deliberation by an international panel of experts. The classification used a combination of letters and J Clin Pathol 2006;59: doi: /jcp numbers to designate various common (and some uncommon) morphological appearances of thymoma. There was no anatomical, functional, histogenetic or other type of stated rationales underlying the schema, and the classification was essentially based on the shape of the neoplastic epithelial cells that is, whether they adopted an oval or spindle shape (thymoma type A), a round or polygonal shape (type B) or a combination of the two (type AB). A fourth category, thymoma type C, was also introduced and defined as a tumour showing overt cytological features of malignancy, independent of the shape of the cells. The tumours in the type C group lacked any similarity to the other groups of thymoma and resembled instead a variety of carcinomas arising in other organs. Type B thymoma was further subclassified into three types(b1,b2andb3)basedontheproportional increase (in relation to the lymphocytes) and emergence of atypia of the neoplastic epithelial cells. A disclaimer was offered in the WHO monograph by the authors to the effect that this schema was not intended to replace any of the existing classifications, but rather to serve as a means for translating the different terms used by the already existing classifications, mainly the traditional classification of Bernatz et al 3 and the histogenetic classification of Kirchner and Muller- Hermelink, 4 intoacommonlanguage.arevised version of the WHO classification was more recently published in 2004, 1 which essentially retained the three basic morphotypes introduced in the original proposal (types A, B and AB), and excluded type C, segregating it into a separate category of thymic carcinoma. The updated version also introduced several unusual morphological types of thymoma that could not be easily placed into any of the other existing categories. WHO type A thymoma is a tumour that is essentially characterised by a neoplastic population of oval to spindle cells devoid of any marked cytological atypia admixed in variable proportions with small (usually few) lymphocytes. This tumour essentially represents the counterpart of spindle-cell thymoma in the traditional classification proposed by Bernatz et al 3 from the Mayo Clinic in Type AB thymoma is defined as a tumour in which foci having features of type A thymoma are admixed with foci showing features of type B thymoma. Type B1 thymoma is defined as a tumour that resembles the normal functional thymus and combines large expanses indistinguishable from the normal thymic cortex with areas resembling the thymic medulla. The neoplastic epithelial cells are scant and composed of oval cells with pale round nuclei and Abbreviations: CAP, College of American Pathologists; WHO, World Health Organization
2 Diagnosis of thymoma 1239 small nucleoli, although it is also stated that in some cells they may be large and occasionally have conspicuous nucleoli. This tumour is regarded as the equivalent of the lymphocyte-rich thymoma in the traditional classification. 3 Type B2 thymoma is defined as a tumour in which the neoplastic thymic epithelial cells are increased in number and appear as scattered plump cells among a heavy population of lymphocytes. The epithelial cells are described as large, polygonal cells, with large vesicular nuclei and central prominent nucleoli. The tumour cells show a tendency to palisade around vessels and fibrous septa. Dilated perivascular spaces are commonly found. These tumours are regarded as the equivalent of mixed lymphoepithelial thymoma in the traditional 3 classification. The main difference between types B1 and B2 seems to be the size of the epithelial cells, these being larger and more numerous, and with no areas of medullary differentiation in type B2 compared with type B1. Type B3 thymoma corresponds to tumours composed predominantly of epithelial cells having a round or polygonal shape and exhibiting negligible or mild atypia. Type C thymoma is defined as a tumour exhibiting clear-cut cytological atypia and a set of cytoarchitectural features no longer specific to the thymus, but rather analogous to those seen in carcinomas of other organs. They lack immature T lymphocytes; the lymphocytes present are usually mature T lymphocytes or B lymphocytes and are often admixed with plasma cells. Another designation for these tumours is thymic carcinoma. A variety of histological types are recognised, most having their counterpart in identical tumours arising from other organs, including keratinising squamous cell carcinoma, non-keratinising squamous cell carcinoma, lymphoepithelioma-like carcinoma, sarcomatoid carcinoma, clear-cell carcinoma, basaloid carcinoma, mucoepidermoid carcinoma, papillary carcinoma and undifferentiated carcinoma. Although the WHO classification has been greeted enthusiastically by many investigators, 5 9 several shortcomings and problem areas have been noted that limit its application. The main problems associated with this classification seem to centre on issues of poor interobserver reproducibility and the lack of clinical relevance for some of its histological subtypes; these have been discussed elsewhere and is not reviewed in detail here Another major problem with the WHO classification is that it shows considerable overlap in morphological criteria between many of its subtypes, making precise classification difficult and sometimes arbitrary, often necessitating the help of an expert consultant for proper categorisation of the lesions. Also, as the original schema was devised as a means of decoding the terminology used by the other competing and already existing classifications, a clear rationale or common substrate to the schema was lacking. Although the authors of the new rendition of the WHO classification have repeatedly emphasised that their schema is based on the histogenetic principles of the Kirchner and Muller-Hermelink classification of thymoma (and, indeed, all of the histogenetic categories in that classification are represented in the WHO schema), 1 the fact is that the WHO schema has no demonstrable histogenetic basis. Moreover, the existence of any histogenetic correspondence has been disclaimed in both the original and the revised versions of the WHO books. 12 These, and other problems associated with the use of this classification, in my opinion, limit the value and utility of the WHO schema for clinical practice. SUSTER AND MORAN CLASSIFICATION OF THYMOMA Owing to the various difficulties that we were experiencing with the existing thymoma classifications, in 1999 we presented a novel proposal for the histological classification of thymic epithelial neoplasms. 13 This proposal essentially represented a simplified approach to the morphological classification of these tumours that separated thymic epithelial neoplasms into three broad categories based on their degrees of organotypical differentiation. In our proposal, the histological grading of these tumours was based on the premise that, as in other organs, primary thymic epithelial neoplasms form part of a continuous spectrum of lesions that range from well differentiated to moderately differentiated to poorly differentiated neoplasms. Welldifferentiated tumours would correspond to tumours designated by convention as thymoma; poorly differentiated neoplasms are those conventionally designated as thymic carcinomas; and tumours showing intermediate features of differentiation are designated as atypical thymoma (table 1). The determination of the degree of differentiation for any given tumour in this system is established on the presence or absence of the characteristic organotypical features of differentiation of the normal thymus and on the degree of cytological atypia of the proliferating epithelial cells Thus, tumours displaying most or all of the organotypical features of thymic differentiation (such as lobulation, dual cell population with a mixture of thymic epithelial cells and immature T lymphocytes, perivascular spaces, areas of socalled medullary differentiation, and so on) and absence of cytological atypia are classified as well-differentiated thymic epithelial neoplasms (ie, thymoma); tumours that retain some of the organotypical features of differentiation of the thymus, but which already display mild to moderate cytological atypia are classified as moderately differentiated thymic epithelial neoplasms (ie, atypical thymoma); and tumours characterised by total absence of the organotypical features of the thymus and showing overt cytological evidence of malignancy correspond to poorly differentiated thymic epithelial neoplasms (ie, thymic carcinoma). This classification is simple, easy to reproduce and does not depend on any purported histogenetic considerations or require the use of special stains or other specialised techniques. It can be easily applied on the basis of the examination of routinely stained slides by haematoxylin and eosin, and requires only familiarity with the organotypical features of differentiation for the different stages of maturation of the normal thymus, along with attention to the degree of cytological atypia displayed by the neoplastic epithelial cells. 13 Moreover, the division of these tumours based on degrees of differentiation (as is the norm for most other organs) provides a functional substrate for a rational classification of these tumours, as the various organotypical features of differentiation of the thymus closely parallel the functional status of the organ. 13 One of the problems for understanding the relationship between organotypical differentiation and the functional status of the organ in the past has been the failure to realise Table 1 Suster and Moran classification of thymic epithelial neoplasms Tumour type Grading Histological criteria Atypical thymoma Thymic carcinoma Well differentiated Moderately differentiated Poorly differentiated Reprinted with permission from Suster and Moran. 17 Preservation of organotypical features of thymic differentiation; no cytological evidence of atypia Partial preservation of organotypical features of differentiation; mild to moderate cytological atypia Loss of organotypical features of differentiation; presence of overt cytological evidence of malignancy
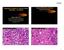
3 1240 Suster A B Figure 1 (A) Normal, mature thymus of childhood. (B) Normal thymus in a 57-year-old man. Note the massive involution of the thymus with extensive replacement by fat and thin strands of flattened, atrophic, spindle thymic epithelial cells. that the thymus is unique among human organs in that its normal morphotype can vary considerably depending on the age of the patient and the stage of maturation of the organ. 15 Unlike most other organs in the human body, the thymus reaches its functional maturity during childhood and adolescence and undergoes a process of loss of functional activity and progressive involution thereafter. This results in an organ whose normal histological appearance can vary widely depending on the age of the patient (fig 1A,B). Recent studies by Hale 16 have mapped this process and shown that with loss of functional maturity, the lymphocytic component of the thymus is progressively replaced by fat and, although the thymus retains its normal size and shape even with advancing age, the residual thymic elements become atrophic and reduced to microscopic remnants. The epithelial cell component also undergoes a process of involution and with loss of functional activity, the cells lose their round shape and become smaller, with oval to spindle nuclei and scant cytoplasm Well-differentiated thymic epithelial neoplasms (ie, thymoma) therefore can show a wide spectrum of morphological appearances that can vary depending on whether the neoplastic cells are attempting to recapitulate the normal, mature thymus of infancy and adolescence or whether they resemble the normal involuted thymus of the adult Most tumours that recapitulate the normal thymus of infants and adolescents are characterised by well-developed lobules, with a predominance of small immature T lymphocytes and few intermingled neoplastic thymic epithelial cells (fig 2). The lobules are usually separated by fibrous bands of variable thickness that are often angulated. Dilated perivascular spaces are commonly present in these tumours. Focal areas of medullary differentiation characterised by pale foci containing a smaller number of lymphocytes than the surrounding tissue can also be seen. The proportion of small lymphocytes to epithelial cells can vary widely among tumours and within different areas of the same tumour. The epithelial cells are round, with large vesicular nuclei and single small eosinophilic nucleoli, and are surrounded by abundant cytoplasm with indistinct cell borders. The cells usually do not show any marked cytological atypia and are devoid of mitotic activity. Mitoses can often be observed in the more immature lymphoid cell elements. These tumours are the equivalent of the lymphocyte-rich thymoma and mixed (lymphoepithelial) thymoma of the traditional J Clin Pathol: first published as /jcp on 5 May Downloaded from Figure 2 showing features that recapitulate the mature, functionally active thymus of childhood and adolescence, with predominance of immature T lymphocytes admixed with scattered large, round thymic epithelial cells. Figure 3 showing features that recapitulate the atrophic or involuted, non-functional thymus of the adult. Sheets of bland-appearing spindled epithelial cells with occasional small T lymphocytes can be observed. on 11 March 2019 by guest. Protected by copyright.
4 Diagnosis of thymoma 1241 Figure 4 Atypical thymoma, characterised by partial preservation of the organotypical features of the thymus (perivascular spaces and dual cell population), but showing paucity of lymphocytes and sheets of large polygonal epithelial cells with nuclear atypia and dense chromatin pattern. classification, 3 of the cortical and predominantly cortical thymoma of the Kirchner and Muller-Hermelink classification, 4 and of the types B1 and B2 in the WHO schema. 1 Tumours recapitulating the features of the normal involuted thymus of the adult are characterised by a solid proliferation of oval to spindle cells, with small elongated nuclei showing a dense chromatin pattern with occasional small, inconspicuous nucleoli and scant rim of cytoplasm (fig 3). The cells often adopt a fascicular growth pattern, but may grow as sheets admixed with variable numbers of small lymphocytes, or they can adopt a variety of unusual growth patterns, such as the creation of rosette-like structures, storiform pattern, hemangiopericytic pattern, micronodular pattern, trabecular (adenoid) pattern and others. 17 The epithelial tumour cells are completely devoid of cytological atypia or mitotic activity. These tumours are usually characterised by a paucity of lymphocytes, although some cases can show a considerable number of T lymphocytes admixed with the epithelial cells. These tumours correspond to the spindle cell type of thymoma in the traditional classification 3 and to the medullary type of thymoma in the Kirchner and Muller-Hermelink classification, 4 and are also the equivalent of type A thymoma in the WHO schema. 1 Patients showing admixtures of spindle cell areas with round cells and lymphocyte-rich areas are often encountered and correspond to the mixed category in the Kirchner and Muller- Hermelink classification 4 and to the type AB thymoma in the WHO schema. 1 Moderately differentiated thymic epithelial neoplasms (ie, atypical thymomas) are defined as tumours that retain some of the organotypical features of differentiation of the thymus, but already show some degree of cytological atypia in the neoplastic epithelial cells (fig 4). Such tumours are the equivalent of the predominantly epithelial thymomas in the traditional classification, 3 of the well-differentiated thymic carcinoma of the Kirchner and Muller-Hermelink classification 4 and of the type B3 thymoma in the WHO schema. 1 The tumours are histologically characterised by sheets of large, round to polygonal epithelial cells with large, irregular and hyperchromatic nuclei showing frequent prominent eosinophilic nucleoli and occasional mitotic figures. The nuclei often have irregular, raisin-like contours. The cytoplasm of the cells is generally abundant and deeply eosinophilic, with very sharp cell borders often imparting the lesion with a squamoid appearance. Foci of early squamous differentiation Figure 5 Thymic carcinoma characterised by absence of the organotypical features of the thymus, with overt cytological evidence of malignancy. can be encountered. The cells can sometimes show clear cytoplasm and often show a tendency to palisade around vessels or perivascular spaces. The tumours also show at least some of the organotypical features of the thymus, such as lobulation, prominent dilated perivascular spaces and an admixture of epithelial cells with small lymphocytes. These tumours are more often invasive than other types of thymoma, with a tendency for earlier recurrences. Atypical thymoma can also be composed of spindle to oval cells rather than round or polygonal cells. In such instances, the spindle cells will show increased nuclear size, with prominent nucleoli and occasional mitotic figures. Poorly differentiated thymic epithelial neoplasms (ie, thymic carcinoma) are characterised by loss of the organotypical features of differentiation of the thymus and the presence of marked or overt cytological features of malignancy (fig 5). These tumours are the equivalent of thymoma type C in the original proposal of the WHO classification 2 and they essentially resemble carcinomas similar to those arising in other epithelial organs. 17 Thymic carcinoma represents a diagnosis of exclusion. Currently, as no reliable specific markers are available that can help us determine the primary nature of these tumours, definitive diagnosis depends on the demonstration of the absence of a primary tumour elsewhere by thorough clinical and imaging studies or at the time of autopsy A large variety of histological variants have been described, including keratinising squamous cell carcinoma, non-keratinising, poorly differentiated (lymphoepitheliomalike) squamous cell carcinoma, mucoepidermoid carcinoma, clear cell carcinoma, basaloid carcinoma, spindle cell (sarcomatoid) carcinoma, anaplastic carcinoma, neuroendocrine carcinoma and others The main problem associated with Table 2 Comparison of World Health Organization schema and the Suster and Moran histological classification of thymoma WHO Type A Type AB Type B1 Type B2 Type B3 WHO, World Health Organization. Suster and Moran Atypical thymoma
5 1242 Suster their diagnosis lies in making a determination as to whether they are primary or metastatic. Although our proposal was initially severely criticised, this approach has been recently validated in a large study of thymoma in which it was shown that further simplification of the WHO schema into three subgroups led to classes with good discriminatory power with respect to survival and superior interobserver agreement. 20 Thus, by simplifying the WHO classification (ie, merging types A, AB, B1 and B2 into a single group, B3 as a separate group and type C as another group), three subgroups with distinct survival could be identified in that study. 20 The authors concluded our results are in favour of classifications that permit accurate allocation of these neoplasms into simple and reproducible diagnostic categories, as proposed by Suster and Moran. 20 This simplified approach also has the advantage of allowing general pathologists to confidently report thymomas without the need to struggle with often conflicting, confusing or ambiguous histological criteria that define some of the WHO categories (table 2). MY APPROACH TO THE DIAGNOSIS OF THYMOMA The most important step in the diagnosis of thymoma is, firstly, to make sure that we are indeed dealing with a thymoma. The most common problem brought to my attention in my personal consultation material is the failure by other pathologists to recognise a given tumour as a thymoma, or of mistakenly diagnosing something else as a thymoma. Clinical features that should raise the possibility of thymoma in the differential diagnosis of a mediastinal mass include location of the tumour in the anterior or anterior superior mediastinal compartment, the description of a wellcircumscribed, lobulated mass surrounded by a fibrous capsule, the absence of malignancy elsewhere on thorough clinical history and clinical examination, and a history of myasthenia gravis or other paraneoplastic syndromes. 17 The next step in the diagnosis of thymoma lies in the histological examination of a biopsy sample or resection specimen. The most critical step for histopathological diagnosis rests on identifying the organotypical features of thymic differentiation associated with the different stages of maturation of the normal thymus (table 1). The vast majority of thymomas are well differentiated and organotypical. The morphology of well-differentiated thymoma, however, can vary considerably depending on whether it is trying to recapitulate the normal thymus of childhood or adolescence or the normal thymus of adults. A multitude of variations on the theme can be seen, with many gradations being possible in the size and shape of the epithelial cells within the same tumour, as well as the relative proportion of thymic epithelial cells to lymphocytes. Unusual morphological variants, including cases showing a micronodular configuration with abundant B cells or plasma cells in the stroma, unusual growth patterns (ie, adenoid, trabecular, hyalinising, hemangiopericytic, rosette forming, and so on) and tumours with variable proportions of thymic epithelial cells in relation to the background lymphocytes can be seen. 14 For as long as the proliferating thymic epithelial cells lack evidence of cytological atypia, and some of the organotypical features of thymic differentiation can still be recognised, they should all be regarded as examples of well-differentiated thymomas. Increase in cytological atypia, such as increase in nuclear size, nucleolar prominence, hyperchromasia and mitotic figures, in a tumour that still retains some of the organotypical features of thymic differentiation define a moderately differentiated neoplasm (atypical thymoma). The shape of the cells in atypical thymoma can be either round polygonal or oval spindle. The important feature is the presence of cytological atypia beyond what is normally encountered in normal thymic epithelial cells, whether those of the mature thymus in childhood or adolescence, or of the involuted thymus of the adult. The diagnosis of a poorly differentiated thymic epithelial neoplasm (thymic carcinoma) is made when the cells show overt cytological evidence of malignancy and the tumour no longer shows any organotypical attributes of the normal thymus, but instead resembles a carcinoma arising in other organs. This diagnosis is more often a diagnosis of exclusion and can only be made a priori under exceptional circumstances. Differential diagnosis The differential diagnosis for thymoma is quite broad and includes several epithelial and mesenchymal tumours, both primary and metastatic. The most important differential diagnosis for well-differentiated thymoma with a predominantly lymphocytic component is with lymphoma, in particular lymphoblastic lymphoma. The most important step in the differential diagnosis in this setting is to carry out a cytokeratin stain to identify the scattered thymic epithelial cells, which are singly scattered in the background of the lymphocytic population. Misdiagnosis of thymoma for lymphoma is one of the most common pitfalls encountered in this clinical setting. The spindle cell variant of thymoma can occasionally be confused for a variety of mesenchymal spindle cell tumours, including solitary fibrous tumour and synovial sarcoma, as well as for a neuroendocrine neoplasm when the tumour is predominantly composed of rosette-like or trabecular structures. The absence of appreciable cytological atypia and identification of some of the organotypical features of thymic differentiation will help separate these tumours from other conditions. Additionally, strong and diffuse positivity for cytokeratin and negative results for other differentiation markers can help arrive at the correct diagnosis. Other immunohistochemical markers that can be of help for the diagnosis of thymoma under certain circumstances include CD1a, CD3, TdT and CD99, all of which mark immature T lymphocytes whose presence is a constant feature (in varying proportions) of well-differentiated thymic epithelial neoplasms. Surgical pathology report Once a diagnosis of thymoma has been established, the surgical pathology report should include three essential components: the main-line diagnosis; a comment; and the synoptic report (fig 6). In the line diagnosis, I simply indicate whether the tumour corresponds to a thymoma or an atypical thymoma, followed by whether the tumour is encapsulated or invasive. In the comment section (or note), I usually provide a short description of the morphology and indicate the salient histological features of the tumour, usually accompanied by a categorisation into one of the histological types in the traditional classification (if possible), and with an indication of its equivalence in the current WHO schema (if possible). I mention that in the note if the tumour cannot be fitted into any of the current WHO types or if precise categorisation is not possible. In this section, I also provide the results of any relevant immunohistochemical stains that support my diagnosis or the results of any other ancillary studies carried out (ie, electron microscopy, cytogenetics, flow cytometry and so on). In recent years, both the College of American Pathologists (CAP) and the Association of Directors of Anatomic and Surgical Pathology in the USA have proposed the use of diagnostic checklists or templates that summarise, under a separate heading, all of the relevant information on a case that will be needed by the clinician or oncologist for proper staging of the disease and for the planning of patient management. Thus, the most salient features of the tumour required for clinical staging and management are also listed in the section under synoptic report (fig 6, bottom half). I
6 Diagnosis of thymoma 1243 also recommend becoming familiarised with the protocol for the examination of specimens from patients with thymic epithelial tumours published by the Cancer Committee of the CAP. 21 The CAP developed these protocols as an educational tool to assist pathologists in providing clinically useful and relevant information when reporting results of surgical specimen examinations. The type of information necessary for producing a complete and accurate reporting of thymic epithelial neoplasms, including clinical information, macroscopic examination and microscopic evaluation, is detailed in the CAP guidelines. 21 Staging of thymoma Staging of thymoma continues to be a controversial issue. Clinical staging of thymoma was first introduced by Berg et al 22 and later modified by Wilkins and Castleman. 23 The most The Ohio State University Medical Center Department of Pathology 410 West 10th Avenue Columbus, Ohio Tel: Fax: Surgical Pathology Report Patient Name: PUBLIC, JOHN Accession #: S Med. Rec #: Client: OSU Collected: 1/19/2006 DOB: 3/2/1955 Location: DOAN1 Received: 1/19/2006 Gender: M Account #: Complete: 1/19/2006 Soc. Sec. #: OtPt Submitting Physician: John Doe, MD CLINICAL HISTORY & PREOPERATIVE DIAGNOSIS 50 year old male with anterior mediastinal mass. *****Pathologic Diagnosis***** A. Anterior mediastinal mass, excision:, encapsulated (see synoptic report). Note: the tumor is composed of sheets of small lymphocytes admixed with few scattered round epithelial cells. The tumor shows a lobular growth pattern separated by bands of fibrous tissue and is completely encapsulated. In some areas, pale foci of so-called "medullary" differentiation are noted. Other areas show dilated perivascular spaces. A cytokeratin stain highlights scattered neoplastic epithelial cells; the small lymphocytes are strongly positive for CD1a, CD3 and CD99, indicating an immature T-phenotype. The histologic features of the tumor are those of a lymphocyte-rich thymoma of the "traditional" classification, and of the type B-1 thymoma in the WHO schema. rlm/sms:1/27/2006 ***Electronically Signed By*** Saul Suster, M.D. 1/27/ :16:17 *****Synoptic Report***** SYNOPTIC REPRORT FOR THYMIC CANCER: Specimen type: Thymectomy. Tumor location: Right lobe of thymus. Tumor size: cm. Histologic type:. Tumor grade: Well differentiated (low grade). Lymphatic/vascular invasion: No. Perineural invasion: No Tumor extent: Capsular status: Encapsulated. Pleura and Lung: No. Pericardium: No Chest wall/soft tissues: No. Margins: Inked outer surface of specimen: Free of tumor. Lymph nodes: Total number examined: N/A Additional pathologic findings: Benign secondary cystic changes involving surrounding parenchyma. Special studies: Immunohistochemical stains, as reported above. Modified Masaoka Staging (Koga et al): Stage l (non invasive). PUBLIC. JOHN Figure 6 Example of a surgical pathology report for thymoma (no actual patient). The top half contains the line diagnosis and a supplementary explanatory comment; the bottom half lists the synoptic report. popular staging system for thymoma was published in 1981 by Masaoka et al 24 and this is the one that has been most widely used in most of the published studies on thymoma. A modification of the Masaoka staging scheme has been more recently introduced by Koga et al, 25 in which the four original categories (ie, stages I, II, III and IV) have been retained. However, the authors noted that the survival curves for their tumours between stages I and II as well as for the tumours between stages III and IV did not differ markedly. They therefore concluded that the staging of thymoma could be simplified into two groups: non-invasive for stages I and II (ie, tumours that are still confined to the anterior mediastinum, without invasion of neighbouring organs) and invasive for stages III and IV (ie, tumours showing invasion of adjacent organs and metastases). I use the non-invasive and invasive scheme 25 in my synoptic report, and also include
7 1244 Suster Take-home messages N A simplified three-tiered histologic classification (thymoma, atypical thymoma and thymic carcinoma; or low-, intermediate and high-grade tumours) is favoured over more complex histologic classifications for the reporting of thymic epithelial neoplasms. N The equivalent type in the current WHO schema is appended in a comment (whenever possible) to facilitate comparison with other classification schemes. N Staging is a more important predictive parameter for prognosis than histology. N The simplified modification of the Masaoka clinical staging scheme proposed by Koga et al is recommended, which categorises these tumours into noninvasive (confined to the mediastinum) and invasive (with infiltration of adjacent structures). N Use of a synoptic report to summarise the salient clinical information is recommended. it implicitly in my line diagnosis, where I always indicate whether the tumour is invasive or non-invasive. I prefer to designate stage II, however, as minimally invasive, to distinguish it from stage III, which is widely invasive, because the two are associated with dramatically different prognoses. 25 A somewhat similar, simplified system has been proposed recently by Bedini et al, 26 which stages the tumours into three tiers: locally restricted disease, locally advanced disease and systemic disease. No universally accepted staging scheme for thymic carcinoma exists yet, and these tumours are not included in the latest edition of the Cancer Staging Manual of the American Joint Commission on Cancer. 27 In the largest series published on thymic carcinoma, the authors applied the original Masaoka staging scheme for the staging of tumours in their patients. 18 Until additional information becomes available or a consensus is developed, it is probably best to use the modified Masaoka staging of thymoma for these tumours in the same way that it is applied for thymomas. SUMMARY Although much emphasis in recent years has been placed on the histological classification of thymoma, the bulk of the evidence continues to point to clinical staging as the most important parameter for prognostication, particularly for well-differentiated thymic epithelial neoplasms For this reason, I believe the most relevant information for the clinician in the main-line diagnosis of the pathology report should be whether the tumour is well, moderately or poorly differentiated, and whether the tumour is invasive or non-invasive. Assigning a tumour to any particular WHO subtype is, in my opinion, of secondary importance, particularly for types A, AB, B1 and B2. All of these tumours essentially represent examples of welldifferentiated neoplasms that retain most of the organotypical features of differentiation of the thymus and are devoid of marked cytological atypia. The status of capsular integrity in such cases will be a more accurate predictor of their biological potential and future behaviour than the histology. The specific WHO subtype in this context, although of academic interest for the pathologist, is of secondary importance to the clinician and may actually contribute to create confusion when provided in the line diagnosis. The simplified approach to the reporting of thymic epithelial neoplasms offered here provides all of the pertinent information for our clinical colleagues, including the WHO subtype (whenever applicable) in the comment section, with minimal chance for error and confusion. Owing to its simplicity and the reduced potential for confusion, this is the format that I favour and use in my clinical practice for the reporting of thymoma. Competing interests: None declared. REFERENCES 1 Travis WD, Brambilla E, Muller-Hermelink HK, et al. Pathology and genetics of tumors of the lung, pleura, thymus and heart. In: World Health Organization classification of tumours. Lyon: IARC Press, Rosai J. Histological typing of tumors of the thymus. In:World Health Organization international histological classification of tumors.2nd edn. Berlin: Springer-Verlag, Bernatz PE, Harrison EG, Claggett OT.. A clinicopathologic study. J Thorac Cardiovasc Surg 1961;42: Kirschner T, Muller-Hermelink HK. New approaches to the diagnosis of thymic epithelial tumors. Prog Surg Pathol 1989;10: Okumura M, Ohta M, Miyoshi S, et al. Oncological significance of WHO histological thymoma classification. A clinical study based on 286 patients. Jpn J Torac Cardiovasc Surg 2002;50: Kim DJ, Yang WI, Choi SS, et al. Prognostic and clinical relevance of the World Health Organization schema for the classification of thymic epithelial tumors: a clinicopathologic study of 108 patients and literature review. Chest 2005;127: Park MS, Chung KY, Kim KD, et al. Prognosis of thymic tumors according to the new World Health Organization histologic classification. Ann Thorac Surg 2004;78: Kondo K, Yoshizawa K, Tsuyuguchi M, et al. WHO histologic classification is a prognostic indicator in thymoma. Ann Thorac Surg 2004;77: Chalabreysse L, Roy P, Cordier J-F, et al. Correlation of the WHO schema for the classification of thymic epithelial neoplasms with prognosis. A retrospective study of 90 tumors. Am J Surg Pathol 2002;26: Rieker RJ, Hoegel J, Morresi-Hauf A, et al. Histologic classification of thymic epithelial tumors: comparison of established classification schemes. Int J Cancer 2002;98: Suster S, Moran CA. classification: current status and future trends. Am J Clin Pathol 2006;125: Suster S, Moran CA. Problem areas and inconsistencies in the WHO classification of thymoma. Semin Diagn Pathol 2005;22: Suster S, Moran CA., atypical thymoma and thymic carcinoma. A novel conceptual approach to the classification of neoplasms of thymic epithelium. Am J Clin Pathol 1999;111: Suster S, Moran CA. Primary thymic epithelial neoplasms. Spectrum of differentiation and histologic features. Semin Diagn Pathol 1999;16: Suster S, Rosai J. The thymus. In: Sternberg SS, ed. Histology for pathologists. 2nd edn. Philadelphia: Raven-Lippincott, 1997: Hale LP. Histologic and molecular assessment of human thymus. Ann Diagn Pathol 2004;8: Suster S, Moran CA. The mediastinum. In: Weidner N, Cote R, Suster S, Weiss LM, eds. Modern surgical pathology. Philadelphia: WB Saunders, 2003: Suster S, Rosai J. Thymic carcinoma. A clinicopathologic study of 60 cases. Cancer 1991;67: Suster S, Moran CA. Thymic carcinoma: spectrum of differentiation and histologic types. Pathology 1998;30: Rieker RJ, Hoegel J, Morresi-Hauf A, et al. Histologic classification of thymic epithelial tumors: comparison of established classification schemes. Int J Cancer 2002;98: Marchevsky AM, Hammond EH, Moran CA, et al. Protocol for the examination of specimens from patients with thymic epithelial tumors located in any area of the mediastinum. Arch Pathol Lab Med 2003;127: Berg NP, Gatzinsky P, Larsson S, et al. Tumors of the thymus and thymic region. I. Clinicopathological studies on thymomas. Ann Thorac Surg 1978;25: Wilkins EW, Castleman B. : a continuing survey at the Massachusetts General Hospital. Ann Thorac Surg 1979;28: Masaoka A, Monden Y, Nakahara K, et al. Follow-up study of thymoma with special reference to their clinical stages. Cancer 1981;48: Koga K, Matsuno Y, Noguchi M, et al. A review of 79 thymomas: modification of staging system and reappraisal of conventional division into invasive and non-invasive thymoma. Pathol Int 1994;44: Bedini AV, Andreani SM, Tavecchio L, et al. Proposal for a novel system for the staging of thymic epithelial tumors. Ann Thorac Surg 2005;80: American Joint Committee on Cancer. AJCC cancer staging manual, 6th edn. Berlin: Springer-Verlag, 2002.
Design: Retrospective, clinicopathologic analysis of our experience and a review of recent literature.
 Prognostic and clinical relevance of the World Health Organization schema for the classification of thymic epithelial tumors : a clinicopathologic study of 108 patients and literature review CHEST, March,
Prognostic and clinical relevance of the World Health Organization schema for the classification of thymic epithelial tumors : a clinicopathologic study of 108 patients and literature review CHEST, March,
THYMIC CARCINOMAS AN UPDATE
 THYMIC CARCINOMAS AN UPDATE Mark R. Wick, M.D. University of Virginia Medical Center Charlottesville, VA CARCINOMA OF THE THYMUS General Clinical Features No apparent gender predilection Age range of 35-75
THYMIC CARCINOMAS AN UPDATE Mark R. Wick, M.D. University of Virginia Medical Center Charlottesville, VA CARCINOMA OF THE THYMUS General Clinical Features No apparent gender predilection Age range of 35-75
Clinical Usefulness of the WHO Histological Classification of Thymoma
 Original Article Clinical Usefulness of the WHO Histological Classification of Thymoma Satoshi Sonobe, MD, 1 Hideaki Miyamoto, MD, 1 Hiroshi Izumi, MD, 2 Bunsei Nobukawa, MD, 2 Toshiro Futagawa, MD, 1
Original Article Clinical Usefulness of the WHO Histological Classification of Thymoma Satoshi Sonobe, MD, 1 Hideaki Miyamoto, MD, 1 Hiroshi Izumi, MD, 2 Bunsei Nobukawa, MD, 2 Toshiro Futagawa, MD, 1
WHO Histologic Classification is a Prognostic Indicator in Thymoma
 WHO Histologic Classification is a Prognostic Indicator in Thymoma Kazuya Kondo, MD, PhD, Kiyoshi Yoshizawa, MD, PhD, Masaru Tsuyuguchi, MD, PhD, Suguru Kimura, MD, PhD, Masayuki Sumitomo, MD, Junji Morita,
WHO Histologic Classification is a Prognostic Indicator in Thymoma Kazuya Kondo, MD, PhD, Kiyoshi Yoshizawa, MD, PhD, Masaru Tsuyuguchi, MD, PhD, Suguru Kimura, MD, PhD, Masayuki Sumitomo, MD, Junji Morita,
Presentation material is for education purposes only. All rights reserved URMC Radiology Page 1 of 98
 Presentation material is for education purposes only. All rights reserved. 2011 URMC Radiology Page 1 of 98 Radiology / Pathology Conference February 2011 Brooke Koltz, Cytopathology Resident Presentation
Presentation material is for education purposes only. All rights reserved. 2011 URMC Radiology Page 1 of 98 Radiology / Pathology Conference February 2011 Brooke Koltz, Cytopathology Resident Presentation
Thymoma and Thymic Carcinoma
 Thymoma and Thymic Carcinoma Protocol applies to thymic epithelial tumors located in any area of the mediastinum. Procedures Biopsy Resection Protocol revision date: January 2004 No AJCC/UICC staging system
Thymoma and Thymic Carcinoma Protocol applies to thymic epithelial tumors located in any area of the mediastinum. Procedures Biopsy Resection Protocol revision date: January 2004 No AJCC/UICC staging system
Thymomas have long attracted interest for a number
 Clinical Value of the WHO Classification System of Thymoma Frank C. Detterbeck, MD Division of Thoracic Surgery, Yale University, New Haven, Connecticut Since the World Health Organization (WHO) histologic
Clinical Value of the WHO Classification System of Thymoma Frank C. Detterbeck, MD Division of Thoracic Surgery, Yale University, New Haven, Connecticut Since the World Health Organization (WHO) histologic
3/27/2017. Pulmonary Pathology Specialty Conference. Disclosure of Relevant Financial Relationships. Clinical History:
 Pulmonary Pathology Specialty Conference Saul Suster, M.D. Medical College of Wisconsin Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position
Pulmonary Pathology Specialty Conference Saul Suster, M.D. Medical College of Wisconsin Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position
Thymomas are rare tumors arising from thymic epithelial
 ITMIG Definitions and Policies Histologic Classification of Thymoma: A Practical Guide for Routine Cases Michael A. den Bakker, MD, PhD,* Anja C. Roden, MD, Alexander Marx, MD, and Mirella Marino, MD Key
ITMIG Definitions and Policies Histologic Classification of Thymoma: A Practical Guide for Routine Cases Michael A. den Bakker, MD, PhD,* Anja C. Roden, MD, Alexander Marx, MD, and Mirella Marino, MD Key
Protocol for the Examination of Specimens From Patients With Thymic Tumors
 Protocol for the Examination of Specimens From Patients With Thymic Tumors Version: Protocol Posting Date: June 2017 Includes ptnm requirements from the 8 th Edition, AJCC Staging Manual For accreditation
Protocol for the Examination of Specimens From Patients With Thymic Tumors Version: Protocol Posting Date: June 2017 Includes ptnm requirements from the 8 th Edition, AJCC Staging Manual For accreditation
Neuroendocrine Lung Tumors Myers
 Diagnosis and Classification of Neuroendocrine Lung Tumors Jeffrey L. Myers, M.D. A. James French Professor Director, Anatomic Pathology & MLabs University of Michigan, Ann Arbor, MI myerjeff@umich.edu
Diagnosis and Classification of Neuroendocrine Lung Tumors Jeffrey L. Myers, M.D. A. James French Professor Director, Anatomic Pathology & MLabs University of Michigan, Ann Arbor, MI myerjeff@umich.edu
Protocol for the Examination of Specimens From Patients With Thymoma and Thymic Carcinoma
 Protocol for the Examination of Specimens From Patients With Thymoma and Thymic Carcinoma Protocol applies to thymic epithelial tumors located in any area of the mediastinum. No AJCC/UICC TNM Staging System
Protocol for the Examination of Specimens From Patients With Thymoma and Thymic Carcinoma Protocol applies to thymic epithelial tumors located in any area of the mediastinum. No AJCC/UICC TNM Staging System
University Journal of Pre and Para Clinical Sciences
 ISSN 2455 2879 Volume 2 Issue 1 2016 Metaplastic carcinoma breast a rare case report Abstract : Metaplastic carcinoma of the breast is a rare malignancy with two distinct cell lines described as a breast
ISSN 2455 2879 Volume 2 Issue 1 2016 Metaplastic carcinoma breast a rare case report Abstract : Metaplastic carcinoma of the breast is a rare malignancy with two distinct cell lines described as a breast
TNM classifications have been established for various
 Lymphogenous and Hematogenous Metastasis of Thymic Epithelial Tumors Kazuya Kondo, MD, PhD, and Yasumasa Monden, MD, PhD Department of Oncological and Regenerative Surgery, School of Medicine, University
Lymphogenous and Hematogenous Metastasis of Thymic Epithelial Tumors Kazuya Kondo, MD, PhD, and Yasumasa Monden, MD, PhD Department of Oncological and Regenerative Surgery, School of Medicine, University
The Role of Fine Needle Aspiration Cytology in the Diagnosis and Management of Thymic Neoplasia
 MALIGNANCIES OF THE THYMUS The Role of Fine Needle Aspiration Cytology in the Diagnosis and Management of Thymic Neoplasia Maureen F. Zakowski, MD, James Huang, MD, and Matthew P. Bramlage, MD Background:
MALIGNANCIES OF THE THYMUS The Role of Fine Needle Aspiration Cytology in the Diagnosis and Management of Thymic Neoplasia Maureen F. Zakowski, MD, James Huang, MD, and Matthew P. Bramlage, MD Background:
Thymic epithelial tumors (TETs), including thymomas,
 Evolution of Classification of Thymic Epithelial Tumors in the Era of Dr Thomas V. Colby Anja C. Roden, MD Context. Numerous histomorphologic and staging classifications of thymic epithelial tumors (TETs)
Evolution of Classification of Thymic Epithelial Tumors in the Era of Dr Thomas V. Colby Anja C. Roden, MD Context. Numerous histomorphologic and staging classifications of thymic epithelial tumors (TETs)
PATHOLOGICAL COMPARATIVE ASSESSMENT OF TWO CASES OF THYMIC CYST AND CYSTIC THYMOMA AND REVIEW OF THE LITERATURE
 Rev. Med. Chir. Soc. Med. Nat., Iaşi 2012 vol. 116, no. 3 INTERNAL MEDICINE - PEDIATRICS CASE REPORTS PATHOLOGICAL COMPARATIVE ASSESSMENT OF TWO CASES OF THYMIC CYST AND CYSTIC THYMOMA AND REVIEW OF THE
Rev. Med. Chir. Soc. Med. Nat., Iaşi 2012 vol. 116, no. 3 INTERNAL MEDICINE - PEDIATRICS CASE REPORTS PATHOLOGICAL COMPARATIVE ASSESSMENT OF TWO CASES OF THYMIC CYST AND CYSTIC THYMOMA AND REVIEW OF THE
Pathological Classification of Hepatocellular Carcinoma
 3 rd APASL Single Topic Conference: HCC in 3D Pathological Classification of Hepatocellular Carcinoma Glenda Lyn Y. Pua, M.D. HCC Primary liver cancer is the 2 nd most common cancer in Asia HCC is the
3 rd APASL Single Topic Conference: HCC in 3D Pathological Classification of Hepatocellular Carcinoma Glenda Lyn Y. Pua, M.D. HCC Primary liver cancer is the 2 nd most common cancer in Asia HCC is the
encapsulated thyroid nodule with a follicular architecture and some form of atypia. The problem is when to diagnose
 Histological Spectrum of Papillary Carcinoma of Thyroid A Two Years Study Gomathi Srinivasan 1, M. Vennila 2 1 Associate Professor Pathology, Government Medical College, Omandurar Estate, Chennai 600 002
Histological Spectrum of Papillary Carcinoma of Thyroid A Two Years Study Gomathi Srinivasan 1, M. Vennila 2 1 Associate Professor Pathology, Government Medical College, Omandurar Estate, Chennai 600 002
Papillary Lesions of the Breast A Practical Approach to Diagnosis. (Arch Pathol Lab Med. 2016;140: ; doi: /arpa.
 Papillary Lesions of the Breast A Practical Approach to Diagnosis (Arch Pathol Lab Med. 2016;140:1052 1059; doi: 10.5858/arpa.2016-0219-RA) Papillary lesions of the breast Span the spectrum of benign,
Papillary Lesions of the Breast A Practical Approach to Diagnosis (Arch Pathol Lab Med. 2016;140:1052 1059; doi: 10.5858/arpa.2016-0219-RA) Papillary lesions of the breast Span the spectrum of benign,
Surgical treatment of thymoma: an 11-year experience with 761 patients
 European Journal of Cardio-Thoracic Surgery 49 (2016) 1144 1149 doi:10.1093/ejcts/ezv288 Advance Access publication 30 August 2015 ORIGINAL ARTICLE Cite this article as: Zhao Y, Shi J, Fan L, Hu D, Yang
European Journal of Cardio-Thoracic Surgery 49 (2016) 1144 1149 doi:10.1093/ejcts/ezv288 Advance Access publication 30 August 2015 ORIGINAL ARTICLE Cite this article as: Zhao Y, Shi J, Fan L, Hu D, Yang
Papillary Lesions of the breast
 Papillary Lesions of the breast Emad Rakha Professor of Breast Pathology The University of Nottingham Papillary lesions of the breast are a heterogeneous group of disease, which are characterised by neoplastic
Papillary Lesions of the breast Emad Rakha Professor of Breast Pathology The University of Nottingham Papillary lesions of the breast are a heterogeneous group of disease, which are characterised by neoplastic
Diplomate of the American Board of Pathology in Anatomic and Clinical Pathology
 A 33-year-old male with a left lower leg mass. Contributed by Shaoxiong Chen, MD, PhD Assistant Professor Indiana University School of Medicine/ IU Health Partners Department of Pathology and Laboratory
A 33-year-old male with a left lower leg mass. Contributed by Shaoxiong Chen, MD, PhD Assistant Professor Indiana University School of Medicine/ IU Health Partners Department of Pathology and Laboratory
Case Scenario 1: Thyroid
 Case Scenario 1: Thyroid History and Physical Patient is an otherwise healthy 80 year old female with the complaint of a neck mass first noticed two weeks ago. The mass has increased in size and is palpable.
Case Scenario 1: Thyroid History and Physical Patient is an otherwise healthy 80 year old female with the complaint of a neck mass first noticed two weeks ago. The mass has increased in size and is palpable.
Case Report A case report of sclerosing thymoma of the anterior mediastinum: an exceedingly rare morphology
 Int J Clin Exp Pathol 2015;8(4):4233-4237 www.ijcep.com /ISSN:1936-2625/IJCEP0006183 Case Report A case report of sclerosing thymoma of the anterior mediastinum: an exceedingly rare morphology Shogo Tajima
Int J Clin Exp Pathol 2015;8(4):4233-4237 www.ijcep.com /ISSN:1936-2625/IJCEP0006183 Case Report A case report of sclerosing thymoma of the anterior mediastinum: an exceedingly rare morphology Shogo Tajima
Pathology of Mediastinal Tumors
 SAMO Meeting Lucerne 2009 Pathology of Mediastinal Tumors Alex Soltermann Most common lesions (adults) Clinical presentation 50% of the patients are asymptomatic, lesion discovered incidentally Symptoms
SAMO Meeting Lucerne 2009 Pathology of Mediastinal Tumors Alex Soltermann Most common lesions (adults) Clinical presentation 50% of the patients are asymptomatic, lesion discovered incidentally Symptoms
57th Annual HSCP Spring Symposium 4/16/2016
 An Unusual Malignant Spindle Cell Lesion to Involve the Breast Erinn Downs-Kelly, D.O. Associate Professor of Pathology University of Utah & ARUP Laboratories No disclosures Case 39 y/o female with no
An Unusual Malignant Spindle Cell Lesion to Involve the Breast Erinn Downs-Kelly, D.O. Associate Professor of Pathology University of Utah & ARUP Laboratories No disclosures Case 39 y/o female with no
Note: The cause of testicular neoplasms remains unknown
 - In the 15- to 34-year-old age group, they are the most common tumors of men. - Tumors of the testis are a heterogeneous group of neoplasms that include: I. Germ cell tumors : 95%; all are malignant.
- In the 15- to 34-year-old age group, they are the most common tumors of men. - Tumors of the testis are a heterogeneous group of neoplasms that include: I. Germ cell tumors : 95%; all are malignant.
The Relevance of Cytologic Atypia in Cutaneous Neural Tumors
 The Relevance of Cytologic Atypia in Cutaneous Neural Tumors Recent Findings - New Developments New Problems Zsolt B. Argenyi, M.D. Professor of Pathology & Dermatology Director of Dermatopathology Department
The Relevance of Cytologic Atypia in Cutaneous Neural Tumors Recent Findings - New Developments New Problems Zsolt B. Argenyi, M.D. Professor of Pathology & Dermatology Director of Dermatopathology Department
Using the World Health Organization Classification of Thymic Epithelial Neoplasms to Describe CT Findings
 Using the World Health Organization Classification of Thymic Epithelial Neoplasms to Describe CT Findings Noriyuki Tomiyama 1 Takeshi Johkoh 2 Naoki Mihara 2 Osamu Honda 2 Takenori Kozuka 2 Mitsuhiro Koyama
Using the World Health Organization Classification of Thymic Epithelial Neoplasms to Describe CT Findings Noriyuki Tomiyama 1 Takeshi Johkoh 2 Naoki Mihara 2 Osamu Honda 2 Takenori Kozuka 2 Mitsuhiro Koyama
Neuroendocrine carcinomas (carcinoids) of the thymus
 Neuroendocrine carcinomas (carcinoids) of the thymus Cesar A. Moran, MD Professor of Pathology Director of Thoracic Pathology M D Anderson Cancer Center Houston, TX Primary thymic neuroendocrine carcinomas
Neuroendocrine carcinomas (carcinoids) of the thymus Cesar A. Moran, MD Professor of Pathology Director of Thoracic Pathology M D Anderson Cancer Center Houston, TX Primary thymic neuroendocrine carcinomas
Case Report Langerhans cell proliferation in micronodular thymic carcinoma with lymphoid stroma: report of a rare case
 Int J Clin Exp Med 2016;9(11):22669-22673 www.ijcem.com /ISSN:1940-5901/IJCEM0036179 Case Report Langerhans cell proliferation in micronodular thymic carcinoma with lymphoid stroma: report of a rare case
Int J Clin Exp Med 2016;9(11):22669-22673 www.ijcem.com /ISSN:1940-5901/IJCEM0036179 Case Report Langerhans cell proliferation in micronodular thymic carcinoma with lymphoid stroma: report of a rare case
Original Article Thymic epithelial tumors: a clinicopathologic study of 249 cases from a single institution
 Int J Clin Exp Pathol 2014;7(11):7760-7767 www.ijcep.com /ISSN:1936-2625/IJCEP0002645 Original Article Thymic epithelial tumors: a clinicopathologic study of 249 cases from a single institution Xue-Ying
Int J Clin Exp Pathol 2014;7(11):7760-7767 www.ijcep.com /ISSN:1936-2625/IJCEP0002645 Original Article Thymic epithelial tumors: a clinicopathologic study of 249 cases from a single institution Xue-Ying
Ectopic Hamartomatous Thymoma
 The Korean Journal of Pathology 2006; 40: 292-6 Ectopic Hamartomatous Thymoma - Case Report along with a Review of the Literature Concerning the Histogenesis and New Nomenclature - Sang Hee Seok Dong Hyun
The Korean Journal of Pathology 2006; 40: 292-6 Ectopic Hamartomatous Thymoma - Case Report along with a Review of the Literature Concerning the Histogenesis and New Nomenclature - Sang Hee Seok Dong Hyun
ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY. Jonathan I. Epstein.
 1 ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY Jonathan I. Epstein Professor Pathology, Urology, Oncology The Reinhard Professor of Urological
1 ARTHUR PURDY STOUT SOCIETY COMPANION MEETING: DIFFICULT NEW DIFFERENTIAL DIAGNOSES IN PROSTATE PATHOLOGY Jonathan I. Epstein Professor Pathology, Urology, Oncology The Reinhard Professor of Urological
Clinical and pathological aspects of microscopic thymoma with myasthenia gravis and review of published reports
 Original Article Clinical and pathological aspects of microscopic thymoma with myasthenia gravis and review of published reports Mitsuro Fukuhara, Mitsunori Higuchi, Yuki Owada, Takuya Inoue, Yuzuru Watanabe,
Original Article Clinical and pathological aspects of microscopic thymoma with myasthenia gravis and review of published reports Mitsuro Fukuhara, Mitsunori Higuchi, Yuki Owada, Takuya Inoue, Yuzuru Watanabe,
FNA of Thyroid. Toward a Uniform Terminology With Management Guidelines. NCI NCI Thyroid FNA State of the Science Conference
 FNA of Thyroid NCI NCI Thyroid FNA State of the Science Conference Toward a Uniform Terminology With Management Guidelines Thyroid Thyroid FNA Cytomorphology NCI Thyroid FNA State of the Science Conference
FNA of Thyroid NCI NCI Thyroid FNA State of the Science Conference Toward a Uniform Terminology With Management Guidelines Thyroid Thyroid FNA Cytomorphology NCI Thyroid FNA State of the Science Conference
On the Histologic Heterogeneity of Thymic Epithelial Neoplasms: Impact of Sampling in Subtyping and Classification of Thymomas
 Stránka č. 1 z 8 cme.medscape.com To Print: Click your browser's PRINT button. NOTE: To view the article with Web enhancements, go to: http://www.medscape.com/viewarticle/0683 On the Histologic Heterogeneity
Stránka č. 1 z 8 cme.medscape.com To Print: Click your browser's PRINT button. NOTE: To view the article with Web enhancements, go to: http://www.medscape.com/viewarticle/0683 On the Histologic Heterogeneity
Tumors of the Lungs and Pleura
 5 Tumors of the Lungs and Pleura Saul Suster and Cesar Moran Introduction Carcinoma of the lung is a common malignant tumor. Histological typing of lung tumors has long been a source of controversy in
5 Tumors of the Lungs and Pleura Saul Suster and Cesar Moran Introduction Carcinoma of the lung is a common malignant tumor. Histological typing of lung tumors has long been a source of controversy in
Almost any suspected tumor can be aspirated easily and safely. Some masses are more risky to aspirate including:
 DOES THIS PATIENT HAVE CANCER? USING IN-HOUSE CYTOLOGY TO HELP YOU MAKE THIS DIAGNOSIS. Joyce Obradovich, DVM, Diplomate, ACVIM (Oncology) Animal Cancer & Imaging Center, Canton, Michigan Almost every
DOES THIS PATIENT HAVE CANCER? USING IN-HOUSE CYTOLOGY TO HELP YOU MAKE THIS DIAGNOSIS. Joyce Obradovich, DVM, Diplomate, ACVIM (Oncology) Animal Cancer & Imaging Center, Canton, Michigan Almost every
Kidney Case 1 SURGICAL PATHOLOGY REPORT
 Kidney Case 1 Surgical Pathology Report February 9, 2007 Clinical History: This 45 year old woman was found to have a left renal mass. CT urography with reconstruction revealed a 2 cm medial mass which
Kidney Case 1 Surgical Pathology Report February 9, 2007 Clinical History: This 45 year old woman was found to have a left renal mass. CT urography with reconstruction revealed a 2 cm medial mass which
Salivary Glands 3/7/2017
 Salivary Glands 3/7/2017 Goals and objectives Focus on the entities unique to H&N Common board type facts Information for your future practice Salivary Glands Salivary Glands Major gland. Paratid. Submandibular.
Salivary Glands 3/7/2017 Goals and objectives Focus on the entities unique to H&N Common board type facts Information for your future practice Salivary Glands Salivary Glands Major gland. Paratid. Submandibular.
Lesions Mimicking Adenoid Cystic Carcinoma. Diagnostic Problems in Salivary Gland Pathology An Update 5/29/2009
 Diagnostic Problems in Salivary Gland Pathology An Update Lesions Mimicking Adenoid Cystic Carcinoma Stacey E. Mills, M.D. W.S. Royster Professor of Pathology Director of Surgical and Cytopathology University
Diagnostic Problems in Salivary Gland Pathology An Update Lesions Mimicking Adenoid Cystic Carcinoma Stacey E. Mills, M.D. W.S. Royster Professor of Pathology Director of Surgical and Cytopathology University
CYSTIC TUMORS OF THE KIDNEY JOHN N. EBLE, M.D. CYSTIC NEPHROMA
 Page 1 CYSTIC TUMORS OF THE KIDNEY JOHN N. EBLE, M.D. Department of Pathology & Laboratory Medicine Phone (317) 274-4806 Medical Science A-128 FAX: (317) 278-2018 635 Barnhill Drive jeble @iupui.edu Indianapolis,
Page 1 CYSTIC TUMORS OF THE KIDNEY JOHN N. EBLE, M.D. Department of Pathology & Laboratory Medicine Phone (317) 274-4806 Medical Science A-128 FAX: (317) 278-2018 635 Barnhill Drive jeble @iupui.edu Indianapolis,
THYMUS AND THYMOMA: WHAT S NEW?
 THYMUS AND THYMOMA: WHAT S NEW? M. RAICA*, SVETLANA ENCICĂ**, L. MOGOANTĂ***, C. SUCIU*, ANCA MARIA CÎMPEAN*, CL. MĂRGĂRITESCU**** *Department of Histology, "Victor Babeş" University of Medicine and Pharmacy,
THYMUS AND THYMOMA: WHAT S NEW? M. RAICA*, SVETLANA ENCICĂ**, L. MOGOANTĂ***, C. SUCIU*, ANCA MARIA CÎMPEAN*, CL. MĂRGĂRITESCU**** *Department of Histology, "Victor Babeş" University of Medicine and Pharmacy,
Microcystic Squamous Cell Carcinoma of the Lung A Clinicopathologic Study of Three Cases
 Anatomic Pathology / Microcystic SCC of the Lung Microcystic Squamous Cell Carcinoma of the Lung A Clinicopathologic Study of Three Cases Annikka Weissferdt, MD, and Cesar A. Moran, MD Key Words: Squamous
Anatomic Pathology / Microcystic SCC of the Lung Microcystic Squamous Cell Carcinoma of the Lung A Clinicopathologic Study of Three Cases Annikka Weissferdt, MD, and Cesar A. Moran, MD Key Words: Squamous
Atypical Palisaded Myofibroblastoma of Lymph Node: Report of a rare case.
 ISPUB.COM The Internet Journal of Pathology Volume 10 Number 1 Atypical Palisaded Myofibroblastoma of Lymph Node: Report of a rare case. V Kinnera, R Nandyala, M Yootla, K Mandyam Citation V Kinnera, R
ISPUB.COM The Internet Journal of Pathology Volume 10 Number 1 Atypical Palisaded Myofibroblastoma of Lymph Node: Report of a rare case. V Kinnera, R Nandyala, M Yootla, K Mandyam Citation V Kinnera, R
Squamous Cell Carcinoma of Thyroid: possible thymic origin, so-called ITET/CASTLE 2012/03/22
 Squamous Cell Carcinoma of Thyroid: possible thymic origin, so-called ITET/CASTLE 2012/03/22 History of ITET/CASTLE First Report Gross Appearance and Prognosis 1) Miyauchi A et al: Intrathyroidal epithelial
Squamous Cell Carcinoma of Thyroid: possible thymic origin, so-called ITET/CASTLE 2012/03/22 History of ITET/CASTLE First Report Gross Appearance and Prognosis 1) Miyauchi A et al: Intrathyroidal epithelial
Case Report Two Invasive Thymomas Incidentally Found during Coronary Artery Bypass Graft Surgery
 Case Reports in Pathology Volume 2016, Article ID 1516521, 4 pages http://dx.doi.org/10.1155/2016/1516521 Case Report Two Invasive Thymomas Incidentally Found during Coronary Artery Bypass Graft Surgery
Case Reports in Pathology Volume 2016, Article ID 1516521, 4 pages http://dx.doi.org/10.1155/2016/1516521 Case Report Two Invasive Thymomas Incidentally Found during Coronary Artery Bypass Graft Surgery
Pitfalls in thyroid tumor pathology. Prof.Valdi Pešutić-Pisac MD, PhD
 Pitfalls in thyroid tumor pathology Prof.Valdi Pešutić-Pisac MD, PhD Too many or... Tumour herniation through a torn capsule simulating capsular invasion fibrous capsule with a sharp discontinuity, suggestive
Pitfalls in thyroid tumor pathology Prof.Valdi Pešutić-Pisac MD, PhD Too many or... Tumour herniation through a torn capsule simulating capsular invasion fibrous capsule with a sharp discontinuity, suggestive
Problem 1: Differential of Neuroendocrine Carcinoma 3/23/2017. Disclosure of Relevant Financial Relationships
 Differential of Neuroendocrine Carcinoma Alain C. Borczuk,MD Weill Cornell Medicine Disclosure of Relevant Financial Relationships USCAP requires that all faculty in a position to influence or control
Differential of Neuroendocrine Carcinoma Alain C. Borczuk,MD Weill Cornell Medicine Disclosure of Relevant Financial Relationships USCAP requires that all faculty in a position to influence or control
Normal thyroid tissue
 Thyroid Pathology Overview Normal thyroid tissue Normal thyroid tissue with follicles filled with colloid. Thyroid cells form follicles, spheres of epithelial cells (always single layered in health, usually
Thyroid Pathology Overview Normal thyroid tissue Normal thyroid tissue with follicles filled with colloid. Thyroid cells form follicles, spheres of epithelial cells (always single layered in health, usually
In the 1960s, thymomas were classified into two categories: Staging System of Thymoma MALIGNANCIES OF THE THYMUS. Akira Masaoka, MD, PhD S304
 MALIGNANCIES OF THE THYMUS Akira Masaoka, MD, PhD Introduction: Thirty years have gone by since the Masaoka staging system of thymoma was proposed in 1981. Although the Masaoka staging system has been
MALIGNANCIES OF THE THYMUS Akira Masaoka, MD, PhD Introduction: Thirty years have gone by since the Masaoka staging system of thymoma was proposed in 1981. Although the Masaoka staging system has been
Respiratory Tract Cytology
 Respiratory Tract Cytology 40 th European Congress of Cytology Liverpool, UK Momin T. Siddiqui M.D. Professor of Pathology and Laboratory Medicine Director of Cytopathology Emory University Hospital, Atlanta,
Respiratory Tract Cytology 40 th European Congress of Cytology Liverpool, UK Momin T. Siddiqui M.D. Professor of Pathology and Laboratory Medicine Director of Cytopathology Emory University Hospital, Atlanta,
Update in Salivary Gland Pathology. Benjamin L. Witt University of Utah/ARUP Laboratories February 9, 2016
 Update in Salivary Gland Pathology Benjamin L. Witt University of Utah/ARUP Laboratories February 9, 2016 Objectives Review the different appearances of a selection of salivary gland tumor types Establish
Update in Salivary Gland Pathology Benjamin L. Witt University of Utah/ARUP Laboratories February 9, 2016 Objectives Review the different appearances of a selection of salivary gland tumor types Establish
Thymic epithelial tumors mainly consist of thymoma,
 Therapy for Thymic Epithelial Tumors: A Clinical Study of 1,320 Patients From Japan Kazuya Kondo, MD, PhD, and Yasumasa Monden, MD, PhD Department of Oncological and Regenerative Surgery, School of Medicine,
Therapy for Thymic Epithelial Tumors: A Clinical Study of 1,320 Patients From Japan Kazuya Kondo, MD, PhD, and Yasumasa Monden, MD, PhD Department of Oncological and Regenerative Surgery, School of Medicine,
3/24/2017 DENDRITIC CELL NEOPLASMS: HISTOLOGY, IMMUNOHISTOCHEMISTRY, AND MOLECULAR GENETICS. Disclosure of Relevant Financial Relationships
 DENDRITIC CELL NEOPLASMS: HISTOLOGY, IMMUNOHISTOCHEMISTRY, AND MOLECULAR GENETICS Jason L. Hornick, M.D., Ph.D. Director of Surgical Pathology and Immunohistochemistry Brigham and Women s Hospital Professor
DENDRITIC CELL NEOPLASMS: HISTOLOGY, IMMUNOHISTOCHEMISTRY, AND MOLECULAR GENETICS Jason L. Hornick, M.D., Ph.D. Director of Surgical Pathology and Immunohistochemistry Brigham and Women s Hospital Professor
Treatment and prognosis of type B2 thymoma
 Song et al. World Journal of Surgical Oncology 2014, 12:291 WORLD JOURNAL OF SURGICAL ONCOLOGY RESEARCH Open Access Treatment and prognosis of type B2 thymoma Zhengbo Song 1,2, Xiangyu Jin 1,2 and Yiping
Song et al. World Journal of Surgical Oncology 2014, 12:291 WORLD JOURNAL OF SURGICAL ONCOLOGY RESEARCH Open Access Treatment and prognosis of type B2 thymoma Zhengbo Song 1,2, Xiangyu Jin 1,2 and Yiping
Thyroid pathology Practical part
 Thyroid pathology Practical part My Algorithm After a good macroscopy and a microscopic overview of the lesion, I especially look at the capsule and the thyroid just above and just beneath the capsule.
Thyroid pathology Practical part My Algorithm After a good macroscopy and a microscopic overview of the lesion, I especially look at the capsule and the thyroid just above and just beneath the capsule.
LUNG CANCER PATHOLOGY: UPDATE ON NEUROENDOCRINE LUNG TUMORS
 LUNG CANCER PATHOLOGY: UPDATE ON NEUROENDOCRINE LUNG TUMORS William D. Travis, M.D. Attending Thoracic Pathologist Memorial Sloan Kettering Cancer Center New York, NY PULMONARY NE TUMORS CLASSIFICATION
LUNG CANCER PATHOLOGY: UPDATE ON NEUROENDOCRINE LUNG TUMORS William D. Travis, M.D. Attending Thoracic Pathologist Memorial Sloan Kettering Cancer Center New York, NY PULMONARY NE TUMORS CLASSIFICATION
number Done by Corrected by Doctor Maha Shomaf
 number 16 Done by Waseem Abo-Obeida Corrected by Zeina Assaf Doctor Maha Shomaf MALIGNANT NEOPLASMS The four fundamental features by which benign and malignant tumors can be distinguished are: 1- differentiation
number 16 Done by Waseem Abo-Obeida Corrected by Zeina Assaf Doctor Maha Shomaf MALIGNANT NEOPLASMS The four fundamental features by which benign and malignant tumors can be distinguished are: 1- differentiation
Differential diagnosis of hematolymphoid tumors composed of medium-sized cells. Brian Skinnider B.C. Cancer Agency, Vancouver General Hospital
 Differential diagnosis of hematolymphoid tumors composed of medium-sized cells Brian Skinnider B.C. Cancer Agency, Vancouver General Hospital Lymphoma classification Lymphoma diagnosis starts with morphologic
Differential diagnosis of hematolymphoid tumors composed of medium-sized cells Brian Skinnider B.C. Cancer Agency, Vancouver General Hospital Lymphoma classification Lymphoma diagnosis starts with morphologic
Diagnostically Challenging Cases in Gynecologic Pathology
 Diagnostically Challenging Cases in Gynecologic Pathology Eric C. Huang, M.D., Ph.D. Department of Pathology and Laboratory Medicine University of California, Davis Medical Center Case 1 Presentation 38
Diagnostically Challenging Cases in Gynecologic Pathology Eric C. Huang, M.D., Ph.D. Department of Pathology and Laboratory Medicine University of California, Davis Medical Center Case 1 Presentation 38
Follicular Derived Thyroid Tumors
 Follicular Derived Thyroid Tumors Jennifer L. Hunt, MD, MEd Aubrey J. Hough Jr, MD, Endowed Professor of Pathology Chair of Pathology and Laboratory Medicine University of Arkansas for Medical Sciences
Follicular Derived Thyroid Tumors Jennifer L. Hunt, MD, MEd Aubrey J. Hough Jr, MD, Endowed Professor of Pathology Chair of Pathology and Laboratory Medicine University of Arkansas for Medical Sciences
My Journey into the World of Salivary Gland Sebaceous Neoplasms
 My Journey into the World of Salivary Gland Sebaceous Neoplasms Douglas R. Gnepp Warren Alpert Medical School at Brown University Rhode Island Hospital Pathology Department Providence RI Asked to present
My Journey into the World of Salivary Gland Sebaceous Neoplasms Douglas R. Gnepp Warren Alpert Medical School at Brown University Rhode Island Hospital Pathology Department Providence RI Asked to present
Neoplasia 2018 Lecture 2. Dr Heyam Awad MD, FRCPath
 Neoplasia 2018 Lecture 2 Dr Heyam Awad MD, FRCPath ILOS 1. List the differences between benign and malignant tumors. 2. Recognize the histological features of malignancy. 3. Define dysplasia and understand
Neoplasia 2018 Lecture 2 Dr Heyam Awad MD, FRCPath ILOS 1. List the differences between benign and malignant tumors. 2. Recognize the histological features of malignancy. 3. Define dysplasia and understand
DETERMINATION OF A LYMPHOID PROCESS
 Chapter 2 Applications of Touch Preparation Cytology to Intraoperative Consultations: Lymph Nodes and Extranodal Tissues for Evaluation of Hematolymphoid Disorders INTRODUCTION As discussed in Chap. 1,
Chapter 2 Applications of Touch Preparation Cytology to Intraoperative Consultations: Lymph Nodes and Extranodal Tissues for Evaluation of Hematolymphoid Disorders INTRODUCTION As discussed in Chap. 1,
Objectives. Atypical Glandular Cells. Atypical Endocervical Cells. Reactive Endocervical Cells
 2013 California Society of Pathologists 66 th Annual Meeting San Francisco, CA Atypical Glandular Cells to Early Invasive Adenocarcinoma: Cervical Cytology and Histology Christina S. Kong, MD Associate
2013 California Society of Pathologists 66 th Annual Meeting San Francisco, CA Atypical Glandular Cells to Early Invasive Adenocarcinoma: Cervical Cytology and Histology Christina S. Kong, MD Associate
Salivary Gland Cytology
 Salivary Gland Cytology Diagnostic challenges and potential pitfalls Tarik M. Elsheikh, MD Professor and Medical Director Anatomic Pathology Cleveland Clinic FNA Salivary Gland Lesions Indications Distinguish
Salivary Gland Cytology Diagnostic challenges and potential pitfalls Tarik M. Elsheikh, MD Professor and Medical Director Anatomic Pathology Cleveland Clinic FNA Salivary Gland Lesions Indications Distinguish
Neoplasia literally means "new growth.
 NEOPLASIA Neoplasia literally means "new growth. A neoplasm, defined as "an abnormal mass of tissue the growth of which exceeds and is uncoordinated with that of the normal tissues and persists in the
NEOPLASIA Neoplasia literally means "new growth. A neoplasm, defined as "an abnormal mass of tissue the growth of which exceeds and is uncoordinated with that of the normal tissues and persists in the
Gross appearance of nodular hyperplasia in material obtained from suprapubic prostatectomy. Note the multinodular appearance and the admixture of
 Tiền liệt tuyến Tiền liệt tuyến Gross appearance of nodular hyperplasia in material obtained from suprapubic prostatectomy. Note the multinodular appearance and the admixture of solid and microcystic areas.
Tiền liệt tuyến Tiền liệt tuyến Gross appearance of nodular hyperplasia in material obtained from suprapubic prostatectomy. Note the multinodular appearance and the admixture of solid and microcystic areas.
Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance
 & 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance Charles C Guo 1 and
& 2006 USCAP, Inc All rights reserved 0893-3952/06 $30.00 www.modernpathology.org Intraductal carcinoma of the prostate on needle biopsy: histologic features and clinical significance Charles C Guo 1 and
Case year old female presented with asymmetric enlargement of the left lobe of the thyroid
 Case 4 22 year old female presented with asymmetric enlargement of the left lobe of the thyroid gland. No information available relative to a prior fine needle aspiration biopsy. A left lobectomy was performed.
Case 4 22 year old female presented with asymmetric enlargement of the left lobe of the thyroid gland. No information available relative to a prior fine needle aspiration biopsy. A left lobectomy was performed.
Disorders of Cell Growth & Neoplasia. Histopathology Lab
 Disorders of Cell Growth & Neoplasia Histopathology Lab Paul Hanna April 2010 Case #84 Clinical History: 5 yr-old, West Highland White terrier. skin mass from axillary region. has been present for the
Disorders of Cell Growth & Neoplasia Histopathology Lab Paul Hanna April 2010 Case #84 Clinical History: 5 yr-old, West Highland White terrier. skin mass from axillary region. has been present for the
Case 4 Diagnosis 2/21/2011 TGB
 Case 4 22 year old female presented with asymmetric enlargement of the left lobe of the thyroid gland. No information available relative to a prior fine needle aspiration biopsy. A left lobectomy was performed.
Case 4 22 year old female presented with asymmetric enlargement of the left lobe of the thyroid gland. No information available relative to a prior fine needle aspiration biopsy. A left lobectomy was performed.
Lung Cytology: Lessons Learned from Errors in Practice
 Lung Cytology: Lessons Learned from Errors in Practice Stephen S. Raab, M.D. Department of Laboratory Medicine Eastern Health and Memorial University of Newfoundland, St. John s, NL and University of Washington,
Lung Cytology: Lessons Learned from Errors in Practice Stephen S. Raab, M.D. Department of Laboratory Medicine Eastern Health and Memorial University of Newfoundland, St. John s, NL and University of Washington,
SESSION 1: GENERAL (BASIC) PATHOLOGY CONCEPTS Thursday, October 16, :30am - 11:30am FACULTY COPY
 SESSION 1: GENERAL (BASIC) PATHOLOGY CONCEPTS Thursday, October 16, 2008 9:30am - 11:30am FACULTY COPY GOAL: Describe the basic morphologic (structural) changes which occur in various pathologic conditions.
SESSION 1: GENERAL (BASIC) PATHOLOGY CONCEPTS Thursday, October 16, 2008 9:30am - 11:30am FACULTY COPY GOAL: Describe the basic morphologic (structural) changes which occur in various pathologic conditions.
Protocol for the Examination of Specimens From Patients With Primary Malignant Tumors of the Heart
 Protocol for the Examination of Specimens From Patients With Primary Malignant Tumors of the Heart Protocol applies to primary malignant cardiac tumors. Hematolymphoid neoplasms are not included. No AJCC/UICC
Protocol for the Examination of Specimens From Patients With Primary Malignant Tumors of the Heart Protocol applies to primary malignant cardiac tumors. Hematolymphoid neoplasms are not included. No AJCC/UICC
Update on 2015 WHO Classification of Lung Adenocarcinoma 1/3/ Mayo Foundation for Medical Education and Research. All rights reserved.
 1 Our speaker for this program is Dr. Anja Roden, an associate professor of Laboratory Medicine and Pathology at Mayo Clinic as well as consultant in the Anatomic Pathology Laboratory and co-director of
1 Our speaker for this program is Dr. Anja Roden, an associate professor of Laboratory Medicine and Pathology at Mayo Clinic as well as consultant in the Anatomic Pathology Laboratory and co-director of
Sarcomatoid (spindle cell) carcinoma of the cricopharynx presenting as dysphagia
 Case Report Sarcomatoid (spindle cell) carcinoma of the cricopharynx presenting as dysphagia Jagtap Sunil V. 1, Shukla Dhirajkumar B. 2, Jagtap Swati S. 3, Havle Abhay D. 4 1 Associate Professor, Department
Case Report Sarcomatoid (spindle cell) carcinoma of the cricopharynx presenting as dysphagia Jagtap Sunil V. 1, Shukla Dhirajkumar B. 2, Jagtap Swati S. 3, Havle Abhay D. 4 1 Associate Professor, Department
Among the benign intraepithelial melanocytic proliferations, Inflamed Conjunctival Nevi. Histopathological Criteria. Resident Short Reviews
 Resident Short Reviews Inflamed conjunctival nevi (ICN) may suggest malignancy because of their rapid growth and atypical histology. The objective of this study was to characterize the diagnostic features
Resident Short Reviews Inflamed conjunctival nevi (ICN) may suggest malignancy because of their rapid growth and atypical histology. The objective of this study was to characterize the diagnostic features
G3.02 The malignant potential of the neoplasm should be recorded. CG3.02a
 G3.02 The malignant potential of the neoplasm should be recorded. CG3.02a Conventional adrenocortical neoplasm. Each of the below parameters is scored 0 when absent and 1 when present. 3 or more of these
G3.02 The malignant potential of the neoplasm should be recorded. CG3.02a Conventional adrenocortical neoplasm. Each of the below parameters is scored 0 when absent and 1 when present. 3 or more of these
International Society of Gynecological Pathologists Symposium 2007
 International Society of Gynecological Pathologists Symposium 2007 Anais Malpica, M.D. Department of Pathology The University of Texas M.D. Anderson Cancer Center Grading of Ovarian Cancer Histologic grade
International Society of Gynecological Pathologists Symposium 2007 Anais Malpica, M.D. Department of Pathology The University of Texas M.D. Anderson Cancer Center Grading of Ovarian Cancer Histologic grade
TBSRTC 1- Probabilistic approach and Relationship to Clinical Algorithms
 The Benefits of a Uniform Reporting System for Thyroid Cytopathology BETHESDA REPORTING SYSTEM Prof. Fernando Schmitt Department of Pathology and Oncology, Medical Faculty of Porto University Head of Molecular
The Benefits of a Uniform Reporting System for Thyroid Cytopathology BETHESDA REPORTING SYSTEM Prof. Fernando Schmitt Department of Pathology and Oncology, Medical Faculty of Porto University Head of Molecular
NODULAR CYSTIC HIDRADENOMA OVER THE GLUTEAL REGION: A RARE CYTOMORPHOLOGICAL DIAGNOSIS
 NODULAR CYSTIC HIDRADENOMA OVER THE GLUTEAL REGION: A RARE CYTOMORPHOLOGICAL DIAGNOSIS Abstract: The primary as well as metastatic tumours of the skin can be diagnosed by fine needle aspiration cytology
NODULAR CYSTIC HIDRADENOMA OVER THE GLUTEAL REGION: A RARE CYTOMORPHOLOGICAL DIAGNOSIS Abstract: The primary as well as metastatic tumours of the skin can be diagnosed by fine needle aspiration cytology
Part 1. Slides 1-38, Rita Alaggio Soft tissue tumors Trondheim 14. mars 2013
 Part 1 Slides 1-38, Rita Alaggio Soft tissue tumors Trondheim 14. mars 2013 Pediatric Pathology Soft Tissue Tumors AN UPDATE Rita Alaggio Azienda Ospedaliera Università di Padova Soft Tissue Tumors More
Part 1 Slides 1-38, Rita Alaggio Soft tissue tumors Trondheim 14. mars 2013 Pediatric Pathology Soft Tissue Tumors AN UPDATE Rita Alaggio Azienda Ospedaliera Università di Padova Soft Tissue Tumors More
Objectives. Salivary Gland FNA: The Milan System. Role of Salivary Gland FNA 04/26/2018
 Salivary Gland FNA: The Milan System Dr. Jennifer Brainard Section Head Cytopathology Cleveland Clinic Objectives Introduce the Milan System for reporting salivary gland cytopathology Define cytologic
Salivary Gland FNA: The Milan System Dr. Jennifer Brainard Section Head Cytopathology Cleveland Clinic Objectives Introduce the Milan System for reporting salivary gland cytopathology Define cytologic
Small cell lung cancer and large cell neuroendocrine tumours interobserver variability. Michael den Bakker Erasmus MC
 Small cell lung cancer and large cell neuroendocrine tumours interobserver variability Michael den Bakker Erasmus MC PPC London 2007 WEB presentation Pulmonary pathology Club presentation, London, November
Small cell lung cancer and large cell neuroendocrine tumours interobserver variability Michael den Bakker Erasmus MC PPC London 2007 WEB presentation Pulmonary pathology Club presentation, London, November
Recurrent adamantinoma of the tibia and lymph node metastasis
 Case Report Recurrent adamantinoma of the tibia and lymph node metastasis Sunil B Gudaganatti, Meena N Jadhav, Rashmi K Patil, Shreekant K Kittur Department of Pathology, Belgaum Institute of Medical Sciences,
Case Report Recurrent adamantinoma of the tibia and lymph node metastasis Sunil B Gudaganatti, Meena N Jadhav, Rashmi K Patil, Shreekant K Kittur Department of Pathology, Belgaum Institute of Medical Sciences,
Synchronous squamous cell carcinoma of the breast. and invasive lobular carcinoma
 Sentani K et al. 1 Letter to the editor Synchronous squamous cell carcinoma of the breast and invasive lobular carcinoma Kazuhiro Sentani, 1 Takashi Tashiro, 2 Naohide Oue, 1 Wataru Yasui 1 1 Department
Sentani K et al. 1 Letter to the editor Synchronous squamous cell carcinoma of the breast and invasive lobular carcinoma Kazuhiro Sentani, 1 Takashi Tashiro, 2 Naohide Oue, 1 Wataru Yasui 1 1 Department
PSA. HMCK, p63, Racemase. HMCK, p63, Racemase
 Case 1 67 year old male presented with gross hematuria H/o acute prostatitis & BPH Urethroscopy: small, polypoid growth with a broad base emanating from the left side of the verumontanum Serum PSA :7 ng/ml
Case 1 67 year old male presented with gross hematuria H/o acute prostatitis & BPH Urethroscopy: small, polypoid growth with a broad base emanating from the left side of the verumontanum Serum PSA :7 ng/ml
Case Presentation. Maha Akkawi, MD, Fatima Obeidat, MD, Tariq Aladily, MD. Department of Pathology Jordan University Hospital Amman, Jordan
 Case Presentation Maha Akkawi, MD, Fatima Obeidat, MD, Tariq Aladily, MD Department of Pathology Jordan University Hospital Amman, Jordan The 25th Annual Congress of the ADIAP The 8/11/2013 1 5th International
Case Presentation Maha Akkawi, MD, Fatima Obeidat, MD, Tariq Aladily, MD Department of Pathology Jordan University Hospital Amman, Jordan The 25th Annual Congress of the ADIAP The 8/11/2013 1 5th International
Basaloid carcinoma of the anal canal
 J. clin. Path. (1967), 0, 18 Basaloid carcinoma of the anal canal LILLIAN S. C. PANG AND B. C. MORSON From the Research Department, St. Mark's Hospital, London SYNOPSIS The pathology and results of treatment
J. clin. Path. (1967), 0, 18 Basaloid carcinoma of the anal canal LILLIAN S. C. PANG AND B. C. MORSON From the Research Department, St. Mark's Hospital, London SYNOPSIS The pathology and results of treatment
Disclosures. Giant Cell Rich Tumors of Bone. Outline. The osteoclast. Giant cell rich tumors 5/21/11
 Disclosures Giant Cell Rich Tumors of Bone Andrew Horvai, MD, PhD Associate Clinical Professor, Pathology This lecture discusses "off label" uses of a number of pharmaceutical agents. The speaker is describing
Disclosures Giant Cell Rich Tumors of Bone Andrew Horvai, MD, PhD Associate Clinical Professor, Pathology This lecture discusses "off label" uses of a number of pharmaceutical agents. The speaker is describing
Most primary thymic epithelial neoplasms represent
 Atypical Thymoma: A Report of Seven Patients Johanna L. Baran, BS, Cynthia M. Magro, MD, Mark A. King, MD, Thomas E. Williams, Jr, MD, PhD, and Patrick Ross, Jr, MD, PhD School of Medicine and Public Health,
Atypical Thymoma: A Report of Seven Patients Johanna L. Baran, BS, Cynthia M. Magro, MD, Mark A. King, MD, Thomas E. Williams, Jr, MD, PhD, and Patrick Ross, Jr, MD, PhD School of Medicine and Public Health,
DIAGNOSTIC SLIDE SEMINAR: PART 1 RENAL TUMOUR BIOPSY CASES
 DIAGNOSTIC SLIDE SEMINAR: PART 1 RENAL TUMOUR BIOPSY CASES Dr. Andrew J. Evans MD, PhD, FACP, FRCPC Consultant in Genitourinary Pathology University Health Network, Toronto, ON Case 1 43 year-old female,
DIAGNOSTIC SLIDE SEMINAR: PART 1 RENAL TUMOUR BIOPSY CASES Dr. Andrew J. Evans MD, PhD, FACP, FRCPC Consultant in Genitourinary Pathology University Health Network, Toronto, ON Case 1 43 year-old female,
International Journal of Pharma and Bio Sciences CHROMOPHOBE VARIANT OF RENAL CELL CARCINOMA MASQUARDING AS RENAL ONCOCYTOMA ON CYTOLOGY.
 Case Report Pathology International Journal of Pharma and Bio Sciences ISSN 0975-6299 CHROMOPHOBE VARIANT OF RENAL CELL CARCINOMA MASQUARDING AS RENAL ONCOCYTOMA ON CYTOLOGY. DR.MAMATHA K*, DR. ARAKERI
Case Report Pathology International Journal of Pharma and Bio Sciences ISSN 0975-6299 CHROMOPHOBE VARIANT OF RENAL CELL CARCINOMA MASQUARDING AS RENAL ONCOCYTOMA ON CYTOLOGY. DR.MAMATHA K*, DR. ARAKERI
Histological Type. Morphological and Molecular Typing of breast Cancer. Nottingham Tenovus Primary Breast Cancer Study. Survival (%) Ian Ellis
 Morphological and Molecular Typing of breast Cancer Ian Ellis Molecular Medical Sciences, University of Nottingham Department of Histopathology, Nottingham University Hospitals NHS Trust Histological Type
Morphological and Molecular Typing of breast Cancer Ian Ellis Molecular Medical Sciences, University of Nottingham Department of Histopathology, Nottingham University Hospitals NHS Trust Histological Type
PLEOMORPHIC ADENOMA ( BENIGN MIXED TUMOR )
 ( BENIGN MIXED TUMOR ) Grossly, the tumor is freely movable, solid, sometimes lobulated and occasionally cystic. If recurrent, multinodular masses are common. Histologically, within a fibrous capsule,
( BENIGN MIXED TUMOR ) Grossly, the tumor is freely movable, solid, sometimes lobulated and occasionally cystic. If recurrent, multinodular masses are common. Histologically, within a fibrous capsule,
Case: The patient is a 62 year old woman with a history of renal cell carcinoma that was removed years ago. A 2.4 cm liver mass was found on CT
 Case: The patient is a 62 year old woman with a history of renal cell carcinoma that was removed years ago. A 2.4 cm liver mass was found on CT during follow- up. ALT, AST, Alk Phos and bilirubin were
Case: The patient is a 62 year old woman with a history of renal cell carcinoma that was removed years ago. A 2.4 cm liver mass was found on CT during follow- up. ALT, AST, Alk Phos and bilirubin were
