Preoperative neoadjuvant chemotherapy has been used
|
|
|
- Bernard Leo Sanders
- 6 years ago
- Views:
Transcription
1 Comparison Between F-FDG PET Image Derived Indices for Early Prediction of Response to Neoadjuvant Chemotherapy in Breast Cancer Mathieu Hatt 1, David Groheux 2,3, Antoine Martineau 2, Marc Espié 4, Elif Hindié 5, Sylvie Giacchetti 4, Anne de Roquancourt 6, Dimitris Visvikis 1, and Catherine Cheze-Le Rest 1,7 1 INSERM, UMR 1101 LaTIM, Brest, France; 2 Department of Nuclear Medicine, Saint-Louis Hospital, Paris, France; 3 B2T, Doctoral School, IUH, University of Paris VII, France; 4 Breast Diseases Unit, Department of Medical Oncology, Saint-Louis Hospital, Paris, France; 5 Department of Nuclear Medicine, Haut-Lévêque Hospital, University of Bordeaux, Bordeaux, France; 6 Department of Pathology, Saint-Louis Hospital, Paris, France; and 7 Department of Nuclear Medicine, CHU Milétrie, Poitiers, France Received Jun. 12, 2012; revision accepted Oct. 2, For correspondence or reprints contact: Mathieu Hatt, INSERM, UMR 1101 LaTIM, CHRU Morvan, 5 Avenue Foch, Brest, France. hatt@univ-breast.fr Published online Jan. 17, COPYRIGHT ª 2013 by the Society of Nuclear Medicine and Molecular Imaging, Inc. The goal of this study was to determine the best predictive factor among image-derived parameters extracted from sequential F-FDG PET scans for early tumor response prediction after 2 cycles of neoadjuvant chemotherapy in breast cancer. Methods: 51 breast cancer patients were included. Responder and nonresponder status was determined by histopathologic examination according to the tumor and node Sataloff scale. PET indices (maximum and mean standardized uptake value [SUV], metabolically active tumor volume, and total lesion glycolysis [TLG]), at baseline and their variation (D) after2cyclesofneoadjuvant chemotherapy were extracted from the PET images. Their predictive value was investigated using Mann Whitney U tests and receiver-operating-characteristic analysis. Subgroup analysis was also performed by considering estrogen receptor (ER) positive/human epidermal growth factor receptor 2 (HER2) negative, triple-negative, and HER2-positive tumors separately. The impact of partial-volume correction was also investigated using an iterative deconvolution algorithm. Results: There were 24 pathologic nonresponders and 27 responders. None of the baseline PET parameters was correlated with response. After 2 neoadjuvant chemotherapy cycles, the reduction of each parameter was significantly associated with response, the best prediction of response being obtained with DTLG (96% sensitivity, 92% specificity, and 94% accuracy), which had a significantly higher area under the curve (0.91 vs. 0.82, P ) than did DSUV max (63% sensitivity, 92% specificity, and 77% accuracy). Subgroup analysis confirmed a significantly higher accuracy for DTLG than DSUV for ER-positive/ HER-negative but not for triple-negative and HER2-positive tumors. Partial-volume correction had no impact on the predictive value of any of the PET image derived parameters despite significant changes in their absolute values. Conclusion: Our results suggest that the reduction after 2 neoadjuvant chemotherapy cycles of the metabolically active volume of primary tumor measurements such as DTLG predicts histopathologic tumor response with higher accuracy than does DSUV measurements, especially for ER-positive/HER2-negative breast cancer. These results should be confirmed in a larger group of patients as they may potentially increase the clinical value and efficiency of F-FDG PET for early prediction of response to neoadjuvant chemotherapy. Key Words: breast cancer; neoadjuvant chemotherapy; F- FDG; tumor delineation; pathological response J Nucl Med 2013; 54: DOI: /jnumed Preoperative neoadjuvant chemotherapy has been used as standard treatment for inflammatory and nonoperable locally advanced breast carcinoma patients and is now increasingly being used for patients with operable but large breast tumors. This strategy allows patients to undergo breast-conserving surgery and provides information on the efficacy of chemotherapy (1). Early response prediction after 1 or 2 cycles of neoadjuvant chemotherapy might enable the selection of alternative treatment strategies (2). Breast carcinoma is a composite, and immunohistochemistry allows the definition of 3 main subgroups with different therapeutic responses and different outcomes (triple-negative, human epidermal growth factor receptor 2 (HER2) positive, and luminal tumors). Pathologic complete response (pcr) is associated with a better outcome in the HER2-overexpressed and triple-negative breast cancer patients. On the other hand, recent studies showed than in luminal tumors, especially for luminal A, the impact of pcr on a patient s survival remains less established (3,4). Thus, an intermediate response with tumor shrinkage allowing breast-conserving surgery might be considered a reasonable clinical objective for this group. Within this context, F-FDG PET has been demonstrated to be a potent predictive tool (5 8). Indeed, correlations between the pathologic tumor response after completion of neoadjuvant chemotherapy and the decrease in tumor standardized uptake values (SUVs) after SEQUENTIAL F-FDG PET FOR BREAST CANCER Hatt et al. 341
2 1 or 2 courses of chemotherapy have been demonstrated in several studies. A recent metaanalysis (8) conducted on 19 studies including 920 patients for the early prediction of primary tumor response to neoadjuvant chemotherapy reported a pooled sensitivity of 84% and specificity of 66% in identifying responders. The authors emphasized that the low pooled specificity (66%) still calls for caution. Most studies have considered SUV measurements only mostly SUV max, or SUV peak averaging the SUV max voxel with its neighboring voxels (9). On the other hand, it has been demonstrated in several recent studies and for various malignancies that other F-FDG PET image derived parameters can have a statistically significant higher predictive value than SUV in determining tumor response (10,11). These parameters, which allow for a more comprehensive tumor functional level evaluation, include metabolically active tumor volume (MATV) (11) and total lesion glycolysis (TLG), defined as the product of MATV and its associated SUV mean (12). In a recent study that compared SUV max and TLG derived using manual delineation and threshold, SUV max had a better predictive value than TLG in identifying pcr (13). To the best of our knowledge, no study has investigated the predictive value, regarding response to neoadjuvant chemotherapy for breast cancer, for all of the previously described F-FDG PET image derived parameters (SUV max,suv mean, SUV peak, TLG, MATV) within the same study, both at baseline and during treatment time points. The current study was therefore conducted with the objective of determining the predictive value of several F-FDG PET derived parameters both at baseline and after 2 neoadjuvant chemotherapy cycles. MATERIALS AND METHODS Patient Population The current study consisted of a retrospective analysis of a prospective cohort of 55 consecutive patients diagnosed with breast cancer and included in a previous clinical trial (14) performed after approval by the institutional ethical committee and with the patients written informed consent. The PET/CT image datasets of 3 patients could not be retrieved from the database because of a corrupt archive file, and 1 was excluded from the analysis because of insufficient initial uptake (SUV max,1.5)in the primary tumor. Therefore, 51 patients were included in the retrospective analysis presented in this work (Table 1). All patients underwent neoadjuvant chemotherapy with 4 cycles of epirubicin plus cyclophosphamide, followed by 4 cycles of docetaxel (plus trastuzumab in cases of HER2 overexpression [HER2-positive]). Patients underwent F-FDG PET/CT scanning at baseline and after the second cycle of neoadjuvant chemotherapy (PET 1 and PET 2, respectively). At the completion of chemotherapy, all patients underwent surgery (mastectomy or lumpectomy). F-FDG PET/CT Acquisitions All F-FDG PET/CT scans were obtained at Saint Louis Hospital in Paris between July 2007 and May A rigorous imaging protocol was designed to ensure robust SUV measurements TABLE 1 Patient Characteristics Characteristic Number of patients American Joint Committee on Cancer clinical stage* IIA 11 (21.5%) IIB 13 (25.5%) IIIA 12 (23.5%) IIIB 14 (27.5%) IIIC 1 (2%) Tumor type Invasive ductal, no special type 45 (88%) Metaplastic 3 (6%) Lobular 3 (6%) Grade 1 4 (8%) 2 28 (55%) 3 17 (33%) Unknown 2 (4%) ER status Positive 30 (59%) Negative 21 (41%) HER2 status Positive 12 (23.5%) Negative 39 (76.5%) Triple-negative status Triple-negative 13 (25.5%) Not triple-negative 38 (74.5%) Surgery Breast-conserving surgery 25 (49%) Mastectomy 26 (51%) Pathologic response Responders 27 (53%) Nonresponders 24 (47%) *Version 7 (16) according to clinical examination and conventional imaging findings. Tumors were considered positive for ER (estrogen receptor) if.10% of cells showed staining by immunohistochemistry. Tumors were considered to be positive for HER2 if.30% of invasive tumor cells showed definite membrane staining resulting in so-called fishnet appearance. across both time points. The blood glucose level had to be no more than 7 mmol/l. For both acquisitions, patients received an intravenous injection (in the arm opposite the breast tumor using a venous line) of F-FDG (5 MBq/kg) after a fasting period of 6 h. After an uptake period of 60 min, all acquisitions were performed from mid-thigh level to the base of the skull with the arms raised, on a Gemini XL PET/CT system (Philips) that combines a germanium oxyorthosilicate based PET scanner and a 16-slice Brilliance CT scanner. CT data were acquired first (120 kv, 100 mas, no contrast enhancement). PET emission list-mode data were acquired in 3-dimensional (3D) mode, with 2 min per bed position, and reconstructed using a 3D row-action maximum-likelihood algorithm with a voxel size of mm. The attenuationcorrected images were normalized for injected dose and body weight and converted into standardized uptake values (SUVs), defined as tracer concentration (kbq/ml)/(injected activity [kbq]/ patient body weight [g]). 342 THE JOURNAL OF NUCLEAR MEDICINE Vol. 54 No. 3 March 2013
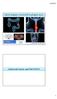
3 Tumor Histology and Immunohistochemistry Analysis Tumor type was determined on the core-needle biopsy sample obtained before neoadjuvant chemotherapy. Immunohistochemical tests were performed on formalin-fixed, paraffin-embedded tissues, using specific antibodies and an automated immunostainer (XT Immunostainer; Ventana). Tumors were considered to be HER2-positive if more than 30% of invasive tumor cells showed definite membrane staining resulting in a so-called fishnet appearance; control by fluorescence in situ hybridization or silver-enhanced in situ hybridization was done for ambiguous cases. Tumors were considered estrogen receptor (ER) negative or progesterone receptor negative if there was less than 10% staining. Three specific tumor subgroups as described previously were considered, namely triple-negative, ER-positive/HER2-negative (luminal), and HER2-positive. Pathologic Tumor Response Histopathologic response was assessed on surgical specimens at the completion of neoadjuvant chemotherapy. Response was graded in primary tumor (T) and nodes (N) according to the scale of Sataloff et al. (15), which was used in the frequently cited study by Rousseau et al. (6): TA 5 total or nearly total therapeutic effect, TB 5 greater than 50% therapeutic effect but less than total or nearly total effect, TC 5 less than 50% therapeutic effect but visible effect, TD 5 no therapeutic effect, NA 5 evidence of therapeutic effect and no residual disease, NB 5 no node metastases or therapeutic effect, NC 5 evidence of a therapeutic effect but metastasis still present, ND 5 metastasis still present and viable (no therapeutic effect). Complete and partial responders (T[A-B] with N[A-B-C]) were considered histopathologic responders, whereas patients with no response or progression (T[C-D], N[D]) were considered nonresponders. The pcr rate, defined as absence of invasive cancer cells in the primary tumor and in lymph nodes (16), was also evaluated, and the predictive power of PET parameters to predict pcr was measured. Investigated Parameters and Analysis All PET image derived parameters were extracted from the PET 1 and PET 2 images. For each patient, the primary tumor was identified on the PET image by a nuclear medicine physician with more than 10 y of experience and subsequently semiautomatically isolated in a 3D region of interest (ROI) containing the tumor and its surrounding background. Tumors were subsequently automatically delineated using the fuzzy locally adaptive bayesian (FLAB) algorithm (17) applied to the previously defined ROI. The FLAB approach allows automatic tumor delineation by computing a probability of belonging to a given class (e.g., tumor or background) for each voxel within the 3D ROI. This probability is calculated by taking into account the voxel s intensity with respect to the statistical distributions (characterized by their mean and variance) of the voxels in the various regions of the image, as well as its spatial correlation with neighboring voxels in 3 dimensions. This approach has been previously validated on simulated and clinical datasets for accuracy, robustness, and reproducibility, on both homogeneous and heterogeneous MATVs (17 19). The potential impact of partial-volume effects was also investigated by correcting the image before analysis using a state-ofthe-art iterative deconvolution previously validated for oncology PET applications (20). The analysis was subsequently performed on both the original and the partial-volume-effect corrected images. SUV max, SUV peak, and SUV mean, as well as the MATV and the TLG, were then automatically calculated from the tumor delineations. SUV peak was defined as the mean of voxel intensities in a 1.7-cm 3 spheric ROI (27 voxels) centered on SUV max (9). MATV was defined as the sum of all voxels contained in the FLAB-delineated volumes multiplied by the volume of a voxel (64 mm 3 ). SUV mean was defined as the mean of voxel intensities in the MATV delineated by FLAB. Subsequently, TLG was determined by multiplying the MATV and its associated SUV mean.thepercentage evolution of each parameter between baseline and after the second neoadjuvant chemotherapy cycle was calculated as (PET 2 parameter 2 PET 1 parameter)/pet 1 parameter 100. Statistical Analysis Statistical analyses were performed using MedCalc (MedCalc Software). All parameters distributions were expressed as median (or mean, depending on normality) 6 SD and range. Normality was tested using the D Agostino Pearson test (21). For each parameter, its correlation with patient response was tested by determining the statistical difference between responders and nonresponders distributions using a Mann Whitney U test. The absolute value of each parameter at PET 1 and PET 2 was investigated, as well as its evolution from PET 1 to PET 2. The predictive performance regarding the identification of responders was evaluated using receiver-operating-characteristic (ROC) analysis. Area under the curve (AUC), sensitivity, specificity, and accuracy were reported. Absolute and associated predictive parameter values extracted from the corrected PET images were compared with original ones. Subgroup analysis was also performed by considering triple-negative, HER2-positive, and ER-positive/HER2-negative tumors separately. All tests were 2-sided, and P values of 0.05 or less were considered statistically significant. RESULTS ER, progesterone receptor, and HER2 receptor status are provided in Table 1. Thirteen patients had triple-negative tumors, 12 had HER2-positive tumors, and 26 had ER-positive/HER2-negative tumors, which is representative (22). Considering the pathologic tumor response established at surgery (after completion of chemotherapy), there were 27 pathologic responders (53%) and 24 nonresponders (47%). pcr was found in 7 patients (13.7%), among which 3 had triple-negative and 4 had HER2-overexpressing tumors and 0 were ER-positive/HER2-negative. Figure 1 illustrates for a pathologic nonresponder and a responder the 2 scans, including delineation of the primary tumor. Predictive Value of Parameters Absolute Values According to the D Agostino Pearson test, considering the entire patient cohort (n 5 51), none of the parameters absolute values at baseline and after the second cycle or associated variation (D) were normally distributed (Table 2). Similarly, the responder and nonresponder groups were not normally distributed (Fig. 2), except for the variation values (Fig. 3). According to Mann Whitney U tests, no significant correlation (P. 0.1) was observed between the baseline values and histopathologic response (Table 2), distributions of the parameters among responders and nonresponders being SEQUENTIAL F-FDG PET FOR BREAST CANCER Hatt et al. 343
4 largely overlapped (Fig. 2A), leading to a low AUC of (Table 3). The primary tumors of nonresponders were characterized by a higher PET 2 SUV max ( vs , P ) and TLG ( vs , P ). However, no correlation (P $ 0.07) was found for any of the other parameters considered (Table 2). For all parameters, the distributions were largely overlapped (Figs. 2A 2C), as the results of ROC analysis demonstrate, with a low AUC of (Table 3). For SUV, sensitivity (57% 69%) and specificity (71% 75%) were limited. On the other hand, for volume-based measurements, sensitivity was higher (89% and 78% for MATV and TLG, respectively) but specificity was lower (42% and 63% for MATV and TLG, respectively). FIGURE 1. Illustration of PET 1 (left) and PET 2 (right) for pathologic responder (A) (DSUV max, 246%, DTLG, 289%) and nonresponder (B) (both DSUV max and DTLG, 243%). Green contours represent FLAB delineation. Evolution of Parameters and Associated Predictive Value There was a global trend of decreasing MATV and associated tumors uptake after the first 2 neoadjuvant chemotherapy cycles, although for some patients PET image derived parameters were also found to be increasing (Table 2). The lowest and largest decreases were observed for DSUV max (234% 6 32%) and DTLG (259% 6 34%), respectively. According to Mann Whitney U tests, the variation of all parameters was statistically different between responders and nonresponders, especially for DMATV and DTLG (P, ), as well as DSUV max (P ) (Table 2). According to ROC analysis, the best prediction was achieved using the DTLG, with a sensitivity of 96% and specificity of 92%, resulting in an accuracy of 94% (Table 3). The DMATV was also an accurate predictive factor (sensitivity, 93%; specificity, 88%; accuracy, 91%). On the other hand, DSUV measurements led to significantly lower AUCs (0.68 for DSUV mean to 0.82 for DSUV max vs for DMATV and 0.91 for DTLG, P # 0.01) (Fig. 4A), with significantly lower sensitivity (63% 74%), specificity (67% 92%), and resulting accuracy (71% 77%) (Table 3). The least overlapped distributions were those associated with DMATV and DTLG (Fig. 3). The optimal cutoff values maximizing sensitivity and specificity were 248%, 242%, 230%, 242%, and 256% for DSUV max, DSUV peak, DSUV mean, DMATV, and DTLG, respectively (Table 3, Fig. 3). Tumor Subgroup Analysis When considering ER-positive/HER2-negative patients (n 5 26), all AUCs were slightly higher but with the same hierarchy as for the entire cohort: DTLG and DMATV resulted in AUCs of 0.96 and 0.98, respectively, whereas DSUV measurements led to AUCs of 0.69, 0.84, and 0.88 for DSUV mean, DSUV peak,anddsuv max, respectively. The ROC analysis performed on the 12 HER2-positive and 13 triple-negative breast cancer patients resulted in TABLE 2 PET Parameter Values and Correlation with Response Parameter PET 1 P PET 2 P D(PET 1, PET 2 ) (%) P SUV max 6.8 (2.3, 27.5) (1.4, 30.7) 0.04* 234 (290, 1104) * SUV peak 4.8 (1.9, 22.0) (1.0, 24.5) (290, 173) * SUV mean 3.7 (1.3,.7) (1.0, 17.9) (287, 169) 0.026* MATV (cm 3 ) 14 (2, 227) (1, 154) (289, 174),0.0001* TLG 51 (9, 668) (1, 758) 0.05* 259 (298, 151),0.0001* *Statistically significant. Data are median, with minimum and maximum in parentheses. P values are for responders vs. nonresponders, as determined using Mann Whitney U test. 344 THE JOURNAL OF NUCLEAR MEDICINE Vol. 54 No. 3 March 2013
5 FIGURE 2. Box-and-whisker distribution plots of SUV max,suv peak,andsuv mean (A); MATV (B); and TLG (C) absolute values at PET 1 and PET 2. One patient with extremely high MATV and TLG values does not appear for readability purposes. Central box represents values from 25th to 75th percentiles. Middle line represents median. A line extends from minimum to maximum, excluding outliers, which are displayed as separate points. nonstatistically significant different AUCs for volumebased measurements and SUVs (P. 0.05) in both cases. Prediction of pcr The pcr predictive value of all parameters was reduced with respect to the prediction of partial response. The hierarchy was, however, similar, with AUCs of 0.76, 0.79, 0.67, 0.63, and 0.59 for DTLG, DMATV, DSUV max, DSUV peak, and DSUV mean, respectively (Fig. 5A). Impact of Partial-Volume-Effect Correction The correction of partial-volume effects had no significant impact on the resulting variation of either SUV or volumebased parameters, despite the significant impact on their PET 1 and PET 2 absolute values. There were therefore no statistically significant differences between the AUCs of ROC curves generated using D(%) calculated using the original and corrected PET image derived parameters (Figs. 4B and 5B). DISCUSSION The current study was the first, to our knowledge, investigation into the predictive value of F-FDG PET derived parameters on a breast cancer cohort, including MATV, TLG, and 3 different SUV measurements at baseline and their evolution. Our results contradict those of the only other study to date comparing the performance of DSUV max and DTLG a study that reported a higher predictive value for DSUV max (13). In that previous study, DSUV mean, DSUV peak, and DMATV predictive values were not considered. In addition, DTLG values were derived from tumor volumes delineated through manual contouring using a fixed threshold. Such a delineation approach has been previously shown to be inaccurate for PET (23,24). In our study, a previously validated robust and reproducible MATV segmentation algorithm was used. Another difference is that results in the previously reported study were reported for the prediction of pcr only, corresponding to 17% of the 142 patients, with a resulting cutoff DSUV max of 83% 88%, which led to high specificity (96%) but low sensitivity (67%). In our study, we considered as pathologic responders patients with complete or partial response in the primary breast cancer and lymph nodes. AccordingtothiscriterionwefoundthatMATVandTLG were more predictive than SUV. We refined a subsequent analysis considering pcr only for responders, and we observed also that volume parameters were superior (AUC, ) to SUV ( ). In contrast to results for several malignancies such as locally advanced esophageal cancer (11) ornon-hodgkin lymphoma (10), none of the absolute baseline values (with or without correction) was significantly associated with response. The absolute values of some parameters (SUV max and TLG) after the second cycle (PET 2 ) were significantly correlated with response but had limited predictive value. The most powerful predictive factors were the evolution between the baseline and the second scan. Pathologic responders were associated with a significantly higher decrease in the F-FDG PET derived indices considered. Among these, DTLG and DMATV were the best predictive factors compared with DSUV measurements, with significantly higher accuracy (91% and 94% for MATV and TLG vs. 71%, 76%, and 77% for DSUV mean, DSUV peak and DSUV max, respectively). With respect to the recent metaanalysis that reported an overall 84% sensitivity and 66% specificity based on DSUV max (8), our DSUV max results demonstrated lower sensitivity (63%) but higher specificity (92%), whereas DTLG resulted in both higher sensitivity (96%) and higher specificity (92%). The metaanalysis also demonstrated large differences in sensitivities and specificities among the various considered studies due to combinations of different patient populations, response criteria (pcr or partial response), and cutoff values. Our study is in line with previous findings that the reduction of PET indices after 2 cycles is a good predictor of response to neoadjuvant chemotherapy in breast cancer (8). We further demonstrated the added value of considering more complete tumor characterization in F-FDG PET images through the delineation of the MATV over SUV measurements only. On the one hand, these results were mostly unchanged when the analysis was performed on the subgroup SEQUENTIAL F-FDG PET FOR BREAST CANCER Hatt et al. 345
6 FIGURE 3. Box-and-whisker distribution plots of DSUV max, DSUV peak, DSUV mean, DMATV, and DTLG. Optimal cutoffs providing best accuracy in predicting response are displayed. Central box represents values from 25th to 75th percentiles. Middle line represents median. A line extends from minimum to maximum, excluding outliers, which are displayed as separate points. of ER-positive/HER2-negative patients. On the other hand, the respective predictive values of DSUV, DMATV, and DTLG were similar for the triple-negative and HER2- positive breast cancer patients. However, since this subgroup analysis included a small number of patients (13 triple-negative and 12 HER2-positive), it needs to be further validated in a larger patient population. Future validation studies on larger groups of patients will be focused on ER-positive/HER2-negative patients since it is for this subgroup that the added value of DMATV and DTLG over DSUV seems to be the most significant. This observation can be explained by the fact that ER-positive/HER2-negative tumors are known to exhibit lower initial F-FDG uptake and therefore have lower decrease margins (7,25,26). These tumors are also known to only partially respond to neoadjuvant chemotherapy, requiring finer characterization of their response (27,28). Therefore, the addition of functional tumor volume based metrics logically improves predictive accuracy for these tumors. For triple-negative and HER2-positive tumors that exhibit higher F-FDG uptake and associated decrease margins, as well as better pcr rates, the addition of volume-based metrics might not translate into a significantly improved predictive value over DSUV measurements. Another interesting result of our study was that DSUV mean had significantly lower predictive value than DSUV peak and DSUV max (AUC of 0.68 vs and 0.82, respectively, P, 0.02). Considering the use of DSUV peak relative to DSUV max, as proposed by the PERCIST recommendations (9), in this study there were no statistically significant differences between the two (AUC, 0.79 vs. 0.82, P 5 0.2), despite DSUV max leading to slightly better results than DSUV peak. Both these parameters describe the highest-activity region of the tumor, whereas the mean value is a better representative measurement of the entire tumor. A potential explanation for the lower predictive value of DSUV mean is its higher dependency on partial-volume effects compared with SUV max and SUV peak. It has, however, recently been shown in a group of 15 breast cancer patients that correction of partial-volume effect had limited impact on SUV reduction (29), without reporting, however, on the actual impact of this reduction on the overall response predictive value. Another study investigated the impact of correction on the prediction of response to neoadjuvant chemotherapy for breast cancer using dynamic F-FDG PET. The investigators found that correction eliminated significant differences in percentage F-FDG uptake measurement changes for nonresponders (NR) versus partial responders but not for pcr versus other response categories (30). In the aforementioned work, partial-volume TABLE 3 Predictive Value According to ROC Analysis Parameter Timing AUC Cutoff value Sensitivity (%) Specificity (%) Accuracy (%) SUV max PET [ ] [14 50] 83 [63 95] 55 [38 68] PET [ ] # [46 84] 75 [53 90] 71 [49 87] D (%) 0.82 [ ] # [42 81] 92 [73 99] 77 [57 90] SUV peak PET [ ] [9 42] 88 [68 97] 53 [34 69] PET [ ] # [42 81] 71 [49 87] 67 [46 85] D (%) 0.79 [ ] # [50 86] 83 [63 95] 76 [54 91] SUV mean PET [ ] # [6 38] 92 [73 99] 53 [34 69] PET [ ] #2 59 [39 78] 75 [53 90] 67 [46 85] D (%) 0.68 [ ] # [54 89] 67 [45 84] 71 [49 88] MATV (cm 3 ) PET [ ].7 85 [66 96] 42 [22 63] 65 [48 79] PET [ ] # [71 98] 42 [22 63] 67 [45 84] D (%) 0.92 [ ] # [76 99] 88 [68 97] 91 [72 98] TLG PET [ ] [76 99] 29 [13 51] 63 [42 81] PET [ ] # [58 91] 63 [41 82] 71 [49 88] D (%) 0.91 [ ] # [81 100] 92 [73 99] 94 [79 99] Data in brackets are 95% confidence intervals. 346 THE JOURNAL OF NUCLEAR MEDICINE Vol. 54 No. 3 March 2013
7 FIGURE 4. ROC curves related to prediction of responders for DSUV max, DSUV peak, DSUV mean, DMATV, and DTLG without (A) or with (B) partial-volume effect correction. effect was corrected using simplistic recovery coefficients, applied only to tumors smaller than 3 cm based on MR imaging. In our study, correction was systematically applied to all images using a voxelwise iterative image deconvolution algorithm previously validated for PET images (20). In this work, the use of correction did not change either the predictive value of the parameters or their evolution or the statistical differences between groups of response, despite a significant change in their absolute values. Finally, correction did not improve the predictive value of SUV mean or the hierarchy of performance between the 3 different SUV measurements, for either the partial or the pcr response criteria. FIGURE 5. ROC curves related to prediction of pcr for DSUV max, DSUV peak, DSUV mean, DMATV, and DTLG without (A) or with (B) partialvolume effect correction. SEQUENTIAL F-FDG PET FOR BREAST CANCER Hatt et al. 347
8 This lack of correction impact is consistent with our previous findings (31,32). The delineation of breast cancer MATVs is challenging because they often exhibit heterogeneous uptake distributions and complex shapes or low tumor-to-background ratios, particularly in the midtreatment scan. Therefore, the use of a robust algorithm is recommended. Within this context, the results presented here using FLAB may be replicated using alternative approaches, such as gradient-based (33) or improved fuzzy C-means (34) methods. Using less robust approaches might lead to significant differences in the results concerning the value of volume-derived parameters (11,13). In addition, this effect may be more important when the temporal evolution of such parameters is considered, as measurements have to be considered with respect to the known physiologic reproducibility ranges. Differences have to be larger than 630% to characterize response, because of the upper and lower physiologic reproducibility limits associated with PET-derived measurements (19). Considering MATV and TLG values, these reproducibility limits were obtained using the FLAB method, whereas less robust approaches (fixed and adaptive threshold) lead to larger values (650% 90%) (19). The FLAB method also allows for measurements that are more repeatable, with low inter- and intraobserver variability (,19). Finally, the imaging protocol in this study was specifically designed to ensure robust and reproducible tumor characterization between both time points. For most patients, measured evolutions were outside the reproducibility limits. Optimal cutoffs were also larger: 248% for SUV max, which is in accordance with previous findings (8), and 242% and 256% for MATV and TLG, respectively. One potential limitation of this study was its retrospective nature and the resulting potential bias, and the lack of reliable outcome information due to the short delay between surgery and last follow-up. Another limitation was the relatively small number of patients due to the fact that the previous prospective study was limited to 55 patients (14), 4 of whom could not be included in the present study. This small sample is especially restrictive regarding the subgroup analyses of triple-negative and HER2-positive patients. The findings of this study therefore need to be validated in larger patient cohorts. Another limitation was that although nodal status was considered to define pathologic response, only primary tumors were characterized in the images to simplify the analysis. Finally, some of the patients were characterized by DTLG and DMATV decreases that were close to the optimal cutoffs but sufficient to avoid misclassification, whereas the opposite was true for SUV max (Figs. 3). The statistical difference between AUCs of DSUV max and DTLG might therefore be lower in larger prospective studies. However, values of DMATV and DTLG present a higher spread than DSUV measurements as demonstrated in Figure 3, showing their higher discriminative power as demonstrated by Mann Whitney U tests (Table 2). CONCLUSION The reduction of metabolically active primary tumor volumes and associated activity measurements such as TLG after 2 neoadjuvant chemotherapy cycles predicts histopathologic tumor response with statistically significant higher accuracy (94%) than SUV max (77%). The advantage of TLG over SUV max was particularly evident for the 26 patients in the ER-positive/HER2-negative subgroup, and therefore we will focus on confirming these results in a larger group of ER-positive/HER2-negative patients. Such confirmation may potentially increase the clinical value and efficiency of F-FDG PET for early prediction of response to neoadjuvant chemotherapy. DISCLOSURE The costs of publication of this article were defrayed in part by the payment of page charges. Therefore, and solely to indicate this fact, this article is hereby marked advertisement in accordance with USC section No potential conflict of interest relevant to this article was reported. REFERENCES 1. Fisher B, Bryant J, Wolmark N, et al. Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. JClinOncol.1998;16: Groheux D, Giacchetti S, Espie M, Rubello D, Moretti JL, Hindie E. Early monitoring of response to neoadjuvant chemotherapy in breast cancer with F-FDG PET/CT: defining a clinical aim. Eur J Nucl Med Mol Imaging. 2011; 38: Bhargava R, Beriwal S, Dabbs DJ, et al. Immunohistochemical surrogate markers of breast cancer molecular classes predicts response to neoadjuvant chemotherapy: a single institutional experience with 359 cases. Cancer. 2010; 116: von Minckwitz G, Untch M, Blohmer JU, et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol. 2012;30: Groheux D, Hindie E, Giacchetti S, et al. Triple-negative breast cancer: early assessment with F-FDG PET/CT during neoadjuvant chemotherapy identifies patients who are unlikely to achieve a pathologic complete response and are at a high risk of early relapse. J Nucl Med. 2012;53: Rousseau C, Devillers A, Sagan C, et al. Monitoring of early response to neoadjuvant chemotherapy in stage II and III breast cancer by [ F]fluorodeoxyglucose positron emission tomography. J Clin Oncol. 2006;24: Schwarz-Dose J, Untch M, Tiling R, et al. Monitoring primary systemic therapy of large and locally advanced breast cancer by using sequential positron emission tomography imaging with [ F]fluorodeoxyglucose. J Clin Oncol. 2009;27: Wang Y, Zhang C, Liu J, Huang G. Is F-FDG PET accurate to predict neoadjuvant therapy response in breast cancer? A meta-analysis. Breast Cancer Res Treat. 2012;131: Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50(suppl 1):122S 150S. 10. Cazaentre T, Morschhauser F, Vermandel M, et al. Pre-therapy F-FDG PET quantitative parameters help in predicting the response to radioimmunotherapy in non-hodgkin lymphoma. Eur J Nucl Med Mol Imaging. 2010;37: Hatt M, Visvikis D, Pradier O, Cheze-le Rest C. Baseline F-FDG PET imagederived parameters for therapy response prediction in oesophageal cancer. Eur J Nucl Med Mol Imaging. 2011;38: Larson SM, Erdi Y, Akhurst T, et al. Tumor treatment response based on visual and quantitative changes in global tumor glycolysis using PET-FDG imaging: the visual response score and the change in total lesion glycolysis. Clin Positron Imaging. 1999;2: Tateishi U, Miyake M, Nagaoka T, et al. Neoadjuvant chemotherapy in breast cancer: prediction of pathologic response with PET/CT and dynamic contrastenhanced MR imaging prospective assessment. Radiology. 2012;263: THE JOURNAL OF NUCLEAR MEDICINE Vol. 54 No. 3 March 2013
9 14. Groheux D, Moretti J, Espié M, et al. FDG PET/CT for early prediction of the response of neoadjuvant chemotherapy for breast cancer after two courses of EC (epirubicin 1 cyclophosphamide) [abstract]. J Nucl Med. 2010;51(suppl 2):51P. 15. Sataloff DM, Mason BA, Prestipino AJ, Seinige UL, Lieber CP, Baloch Z. Pathologic response to induction chemotherapy in locally advanced carcinoma of the breast: a determinant of outcome. J Am Coll Surg. 1995;0: Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th ed. of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17: Hatt M, Cheze le Rest C, Descourt P, et al. Accurate automatic delineation of heterogeneous functional volumes in positron emission tomography for oncology applications. Int J Radiat Oncol Biol Phys. 2010;77: Hatt M, Cheze Le Rest C, Albarghach N, Pradier O, Visvikis D. PET functional volume delineation: a robustness and repeatability study. Eur J Nucl Med Mol Imaging. 2011;38: Hatt M, Cheze-Le Rest C, Aboagye EO, et al. Reproducibility of F-FDG and 39-deoxy-39- F-fluorothymidine PET tumor volume measurements. J Nucl Med. 2010;51: Boussion N, Cheze Le Rest C, Hatt M, Visvikis D. Incorporation of waveletbased denoising in iterative deconvolution for partial volume correction in whole-body PET imaging. Eur J Nucl Med Mol Imaging. 2009;36: Sheskin D. Handbook of Parametric and Nonparametric Statistical Procedures. 5th ed. Boca Raton, FL: Chapman & Hall/CRC; Straver ME, Rutgers EJ, Rodenhuis S, et al. The relevance of breast cancer subtypes in the outcome of neoadjuvant chemotherapy. Ann Surg Oncol. 2010; 17: Hatt M, Cheze-le Rest C, van Baardwijk A, Lambin P, Pradier O, Visvikis D. Impact of tumor size and tracer uptake heterogeneity in F-FDG PET and CT non-small cell lung cancer tumor delineation. J Nucl Med. 2011;52: Hatt M, Visvikis D, Le Rest CC. Autocontouring versus manual contouring [letter]. J Nucl Med. 2011;52: McDermott GM, Welch A, Staff RT, et al. Monitoring primary breast cancer throughout chemotherapy using FDG-PET. Breast Cancer Res Treat. 2007;102: Groheux D, Giacchetti S, Moretti JL, et al. Correlation of high F-FDG uptake to clinical, pathological and biological prognostic factors in breast cancer. Eur J Nucl Med Mol Imaging. 2011;38: Lips EH, Mulder L, de Ronde JJ, et al. Neoadjuvant chemotherapy in ER1 HER22 breast cancer: response prediction based on immunohistochemical and molecular characteristics. Breast Cancer Res Treat. 2012;131: de Ronde JJ, Hannemann J, Halfwerk H, et al. Concordance of clinical and molecular breast cancer subtyping in the context of preoperative chemotherapy response. Breast Cancer Res Treat. 2010;119: Hoetjes NJ, van Velden FH, Hoekstra OS, et al. Partial volume correction strategies for quantitative FDG PET in oncology. Eur J Nucl Med Mol Imaging. 2010;37: Tseng J, Dunnwald LK, Schubert EK, et al. F-FDG kinetics in locally advanced breast cancer: correlation with tumor blood flow and changes in response to neoadjuvant chemotherapy. J Nucl Med. 2004;45: Hatt M, van Stiphout R, le Pogam A, Lammering G, Visvikis D, Lambin P. Early prediction of pathological response in locally advanced rectal cancer based on sequential F-FDG PET. Acta Oncol. August 8, 2012 [Epub ahead of print]. 32. Hatt M, Le Pogam A, Visvikis D, Pradier O, Cheze le Rest C. Impact of partial volume effects correction on the predictive and prognostic value of baseline F-FDG PET images in esophageal cancer. J Nucl Med. 2012;53: Geets X, Lee JA, Bol A, Lonneux M, Gregoire V. A gradient-based method for segmenting FDG-PET images: methodology and validation. Eur J Nucl Med Mol Imaging. 2007;34: Belhassen S, Zaidi H. A novel fuzzy C-means algorithm for unsupervised heterogeneous tumor quantification in PET. Med Phys. 2010;37: SEQUENTIAL F-FDG PET FOR BREAST CANCER Hatt et al. 349
Comparison Between 18F-FDG PET Image-Derived Indices for Early Prediction of Response to Neoadjuvant Chemotherapy in Breast Cancer.
 Comparison Between 18F-FDG PET Image-Derived Indices for Early Prediction of Response to Neoadjuvant Chemotherapy in Breast Cancer. Mathieu Hatt, David Groheux, Antoine Martineau, Marc Espié, Elif Hindié,
Comparison Between 18F-FDG PET Image-Derived Indices for Early Prediction of Response to Neoadjuvant Chemotherapy in Breast Cancer. Mathieu Hatt, David Groheux, Antoine Martineau, Marc Espié, Elif Hindié,
Mathieu Hatt, Dimitris Visvikis. To cite this version: HAL Id: inserm
 Defining radiotherapy target volumes using 18F-fluoro-deoxy-glucose positron emission tomography/computed tomography: still a Pandora s box?: in regard to Devic et al. (Int J Radiat Oncol Biol Phys 2010).
Defining radiotherapy target volumes using 18F-fluoro-deoxy-glucose positron emission tomography/computed tomography: still a Pandora s box?: in regard to Devic et al. (Int J Radiat Oncol Biol Phys 2010).
Evaluation of Lung Cancer Response: Current Practice and Advances
 Evaluation of Lung Cancer Response: Current Practice and Advances Jeremy J. Erasmus I have no financial relationships, arrangements or affiliations and this presentation will not include discussion of
Evaluation of Lung Cancer Response: Current Practice and Advances Jeremy J. Erasmus I have no financial relationships, arrangements or affiliations and this presentation will not include discussion of
With a worldwide estimated 5-y survival of only 15%
 Impact of Partial-Volume Effect Correction on the Predictive and Prognostic Value of Baseline 18 F-FDG PET Images in Esophageal Cancer Mathieu Hatt 1, Adrien Le Pogam 1,2, Dimitris Visvikis 1, Olivier
Impact of Partial-Volume Effect Correction on the Predictive and Prognostic Value of Baseline 18 F-FDG PET Images in Esophageal Cancer Mathieu Hatt 1, Adrien Le Pogam 1,2, Dimitris Visvikis 1, Olivier
Reproducibility of tumor uptake heterogeneity characterization through textural feature analysis in 18 F-FDG PET imaging
 1 2 3 Reproducibility of tumor uptake heterogeneity characterization through textural feature analysis in 18 F-FDG PET imaging 4 5 6 7 Florent TIXIER 1,2, Mathieu Hatt 1, Catherine Cheze Le Rest 1, Adrien
1 2 3 Reproducibility of tumor uptake heterogeneity characterization through textural feature analysis in 18 F-FDG PET imaging 4 5 6 7 Florent TIXIER 1,2, Mathieu Hatt 1, Catherine Cheze Le Rest 1, Adrien
Inflammatory breast cancer (IBC), the rarest and most deadly
 F-FDG PET/CT to Predict Response to Neoadjuvant Chemotherapy and Prognosis in Inflammatory Breast Cancer Laurence Champion* 1, Florence Lerebours* 2, Jean-Louis Alberini 1,3, Emmanuelle Fourme 4, Eric
F-FDG PET/CT to Predict Response to Neoadjuvant Chemotherapy and Prognosis in Inflammatory Breast Cancer Laurence Champion* 1, Florence Lerebours* 2, Jean-Louis Alberini 1,3, Emmanuelle Fourme 4, Eric
8/10/2016. PET/CT Radiomics for Tumor. Anatomic Tumor Response Assessment in CT or MRI. Metabolic Tumor Response Assessment in FDG-PET
 PET/CT Radiomics for Tumor Response Evaluation August 1, 2016 Wei Lu, PhD Department of Medical Physics www.mskcc.org Department of Radiation Oncology www.umaryland.edu Anatomic Tumor Response Assessment
PET/CT Radiomics for Tumor Response Evaluation August 1, 2016 Wei Lu, PhD Department of Medical Physics www.mskcc.org Department of Radiation Oncology www.umaryland.edu Anatomic Tumor Response Assessment
Department of Breast Oncology, National Hospital Organization Shikoku Cancer Center, Matsuyama, Japan
 Predictive Ability of 18 F-fluorodeoxyglucose Positron Emission Tomography/computed Tomography for Pathological Complete Response and Prognosis after Neoadjuvant Chemotherapy in Triplenegative Breast Cancer
Predictive Ability of 18 F-fluorodeoxyglucose Positron Emission Tomography/computed Tomography for Pathological Complete Response and Prognosis after Neoadjuvant Chemotherapy in Triplenegative Breast Cancer
8/10/2016. PET/CT for Tumor Response. Staging and restaging Early treatment response evaluation Guiding biopsy
 PET/CT for Tumor Response Evaluation August 4, 2016 Wei Lu, PhD Department of Medical Physics www.mskcc.org Department of Radiation Oncology www.umaryland.edu FDG PET/CT for Cancer Imaging Staging and
PET/CT for Tumor Response Evaluation August 4, 2016 Wei Lu, PhD Department of Medical Physics www.mskcc.org Department of Radiation Oncology www.umaryland.edu FDG PET/CT for Cancer Imaging Staging and
PET/CT in breast cancer staging
 PET/CT in breast cancer staging Anni Morsing Consultant, PhD, DMSc Rigshospitalet 1 18F- FDG PET/CT for breastcancer staging Where is the clinical impact? To which women should 18F- FDG PET/CT be offered?
PET/CT in breast cancer staging Anni Morsing Consultant, PhD, DMSc Rigshospitalet 1 18F- FDG PET/CT for breastcancer staging Where is the clinical impact? To which women should 18F- FDG PET/CT be offered?
Early prediction of pathological response in locally advanced rectal cancer based on sequential (18)F-FDG PET.
 Early prediction of pathological response in locally advanced rectal cancer based on sequential (18)F-FDG PET. Mathieu Hatt, Ruud Van Stiphout, Adrien Le Pogam, Guido Lammering, Dimitris Visvikis, Philippe
Early prediction of pathological response in locally advanced rectal cancer based on sequential (18)F-FDG PET. Mathieu Hatt, Ruud Van Stiphout, Adrien Le Pogam, Guido Lammering, Dimitris Visvikis, Philippe
Utility of 18 F-FDG PET/CT in metabolic response assessment after CyberKnife radiosurgery for early stage non-small cell lung cancer
 Utility of F-FDG PET/CT in metabolic response assessment after CyberKnife radiosurgery for early stage non-small cell lung cancer Ngoc Ha Le 1*, Hong Son Mai 1, Van Nguyen Le 2, Quang Bieu Bui 2 1 Department
Utility of F-FDG PET/CT in metabolic response assessment after CyberKnife radiosurgery for early stage non-small cell lung cancer Ngoc Ha Le 1*, Hong Son Mai 1, Van Nguyen Le 2, Quang Bieu Bui 2 1 Department
Dr Sneha Shah Tata Memorial Hospital, Mumbai.
 Dr Sneha Shah Tata Memorial Hospital, Mumbai. Topics covered Lymphomas including Burkitts Pediatric solid tumors (non CNS) Musculoskeletal Ewings & osteosarcoma. Neuroblastomas Nasopharyngeal carcinomas
Dr Sneha Shah Tata Memorial Hospital, Mumbai. Topics covered Lymphomas including Burkitts Pediatric solid tumors (non CNS) Musculoskeletal Ewings & osteosarcoma. Neuroblastomas Nasopharyngeal carcinomas
The prognosis of patients with locally advanced breast
 F-FDG PET/CT in Staging Patients with Locally Advanced or Inflammatory Breast Cancer: Comparison to Conventional Staging David Groheux 1,2, Sylvie Giacchetti 3, Marc Delord 4, Elif Hindié 2,5, Laetitia
F-FDG PET/CT in Staging Patients with Locally Advanced or Inflammatory Breast Cancer: Comparison to Conventional Staging David Groheux 1,2, Sylvie Giacchetti 3, Marc Delord 4, Elif Hindié 2,5, Laetitia
Repeatability and reproducibility of 18 F-NaF PET quantitative imaging biomarkers
 Repeatability and reproducibility of 18 F-NaF PET quantitative imaging biomarkers Christie Lin, Tyler Bradshaw, Timothy Perk, Stephanie Harmon, Glenn Liu, Robert Jeraj University of Wisconsin Madison,
Repeatability and reproducibility of 18 F-NaF PET quantitative imaging biomarkers Christie Lin, Tyler Bradshaw, Timothy Perk, Stephanie Harmon, Glenn Liu, Robert Jeraj University of Wisconsin Madison,
Metabolic volume measurement (physics and methods)
 Metabolic volume measurement (physics and methods) Ronald Boellaard Department of Nuclear Medicine & PET Research, VU University Medical Center, Amsterdam r.boellaard@vumc.nl Metabolic volume vs tumor
Metabolic volume measurement (physics and methods) Ronald Boellaard Department of Nuclear Medicine & PET Research, VU University Medical Center, Amsterdam r.boellaard@vumc.nl Metabolic volume vs tumor
Breast Cancer Staging. Physiology Trumps Anatomy Author: Maxine Jochelson, MD, FSBI
 Breast Cancer Staging. Physiology Trumps Anatomy Author: Maxine Jochelson, MD, FSBI The purpose of this paper is to address the importance of physiologic imaging for the staging and follow up of patients
Breast Cancer Staging. Physiology Trumps Anatomy Author: Maxine Jochelson, MD, FSBI The purpose of this paper is to address the importance of physiologic imaging for the staging and follow up of patients
Radiological assessment of neoadjuvent chemotherapy for breast cancer
 XV th Balkan Congress of Radiology Budapest, Hungary, October 12 15, 2017 Radiological assessment of neoadjuvent chemotherapy for breast cancer V. Bešlagić C l i n i c o f R a d i o l o g y, U n i v e
XV th Balkan Congress of Radiology Budapest, Hungary, October 12 15, 2017 Radiological assessment of neoadjuvent chemotherapy for breast cancer V. Bešlagić C l i n i c o f R a d i o l o g y, U n i v e
Evaluation of Pathologic Response in Breast Cancer Treated with Primary Systemic Therapy
 Evaluation of Pathologic Response in Breast Cancer Treated with Primary Systemic Therapy Eun Yoon Cho, MD, PhD Department of Pathology and Translational Genomics Samsung Medical Center Sungkyunkwan University
Evaluation of Pathologic Response in Breast Cancer Treated with Primary Systemic Therapy Eun Yoon Cho, MD, PhD Department of Pathology and Translational Genomics Samsung Medical Center Sungkyunkwan University
ROLE OF PET-CT IN BREAST CANCER, GUIDELINES AND BEYOND. Prof Jamshed B. Bomanji Institute of Nuclear Medicine UCL Hospitals London
 ROLE OF PET-CT IN BREAST CANCER, GUIDELINES AND BEYOND Prof Jamshed B. Bomanji Institute of Nuclear Medicine UCL Hospitals London CANCER Key facts Estimated 15.2 million new cases per year in 2015 worldwide
ROLE OF PET-CT IN BREAST CANCER, GUIDELINES AND BEYOND Prof Jamshed B. Bomanji Institute of Nuclear Medicine UCL Hospitals London CANCER Key facts Estimated 15.2 million new cases per year in 2015 worldwide
Chapter 2 Staging of Breast Cancer
 Chapter 2 Staging of Breast Cancer Zeynep Ozsaran and Senem Demirci Alanyalı 2.1 Introduction Five decades ago, Denoix et al. proposed classification system (tumor node metastasis [TNM]) based on the dissemination
Chapter 2 Staging of Breast Cancer Zeynep Ozsaran and Senem Demirci Alanyalı 2.1 Introduction Five decades ago, Denoix et al. proposed classification system (tumor node metastasis [TNM]) based on the dissemination
Biases affecting tumor uptake measurements in FDG-PET
 Biases affecting tumor uptake measurements in FDG-PET M. Soret, C. Riddell, S. Hapdey, and I. Buvat Abstract-- The influence of tumor diameter, tumor-tobackground activity ratio, attenuation, spatial resolution,
Biases affecting tumor uptake measurements in FDG-PET M. Soret, C. Riddell, S. Hapdey, and I. Buvat Abstract-- The influence of tumor diameter, tumor-tobackground activity ratio, attenuation, spatial resolution,
Early monitoring of response to neoadjuvant chemotherapy in breast cancer with 18 F-FDG PET/CT: defining a clinical aim
 Eur J Nucl Med Mol Imaging (2011) 38:419 425 DOI 10.1007/s00259-010-1660-5 EDITORIAL Early monitoring of response to neoadjuvant chemotherapy in breast cancer with 18 F-FDG PET/CT: defining a clinical
Eur J Nucl Med Mol Imaging (2011) 38:419 425 DOI 10.1007/s00259-010-1660-5 EDITORIAL Early monitoring of response to neoadjuvant chemotherapy in breast cancer with 18 F-FDG PET/CT: defining a clinical
Ratio of maximum standardized uptake value to primary tumor size is a prognostic factor in patients with advanced non-small cell lung cancer
 Original Article Ratio of maximum standardized uptake value to primary tumor size is a prognostic factor in patients with advanced non-small cell lung cancer Fangfang Chen 1 *, Yanwen Yao 2 *, Chunyan
Original Article Ratio of maximum standardized uptake value to primary tumor size is a prognostic factor in patients with advanced non-small cell lung cancer Fangfang Chen 1 *, Yanwen Yao 2 *, Chunyan
Journal of Nuclear Medicine, published on August 17, 2011 as doi: /jnumed
 Downloaded from jnm.snmjournals.org by on September 4, 20. For personal use only. Journal of Nuclear Medicine, published on August 17, 2011 as doi:10.2967/jnumed.111.088914 Effects of Image Characteristics
Downloaded from jnm.snmjournals.org by on September 4, 20. For personal use only. Journal of Nuclear Medicine, published on August 17, 2011 as doi:10.2967/jnumed.111.088914 Effects of Image Characteristics
Breast Cancer. Most common cancer among women in the US. 2nd leading cause of death in women. Mortality rates though have declined
 Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women Mortality rates though have declined 1 in 8 women will develop breast cancer Breast Cancer Breast cancer increases
Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women Mortality rates though have declined 1 in 8 women will develop breast cancer Breast Cancer Breast cancer increases
Problems in staging breast carcinoma
 Problems in staging breast carcinoma Primary systemic therapy (PST) of breast carcinoma pathologists tasks Dr. Janina Kulka, 2nd Department of Pathology, Semmelweis University Budapest Austro-Hungarian
Problems in staging breast carcinoma Primary systemic therapy (PST) of breast carcinoma pathologists tasks Dr. Janina Kulka, 2nd Department of Pathology, Semmelweis University Budapest Austro-Hungarian
When do you need PET/CT or MRI in early breast cancer?
 When do you need PET/CT or MRI in early breast cancer? Elizabeth A. Morris MD FACR Chief, Breast Imaging Service Memorial Sloan-Kettering Cancer Center NY, NY Objectives What is the role of MRI in initial
When do you need PET/CT or MRI in early breast cancer? Elizabeth A. Morris MD FACR Chief, Breast Imaging Service Memorial Sloan-Kettering Cancer Center NY, NY Objectives What is the role of MRI in initial
Index. Surg Oncol Clin N Am 16 (2007) Note: Page numbers of article titles are in boldface type.
 Surg Oncol Clin N Am 16 (2007) 465 469 Index Note: Page numbers of article titles are in boldface type. A Adjuvant therapy, preoperative for gastric cancer, staging and, 339 B Breast cancer, metabolic
Surg Oncol Clin N Am 16 (2007) 465 469 Index Note: Page numbers of article titles are in boldface type. A Adjuvant therapy, preoperative for gastric cancer, staging and, 339 B Breast cancer, metabolic
PET-CT for radiotherapy planning in lung cancer: current recommendations and future directions
 PET-CT for radiotherapy planning in lung cancer: current recommendations and future directions Gerry Hanna Centre for Cancer Research and Cell Biology Queen s University of Belfast @gerryhanna Talk Outline
PET-CT for radiotherapy planning in lung cancer: current recommendations and future directions Gerry Hanna Centre for Cancer Research and Cell Biology Queen s University of Belfast @gerryhanna Talk Outline
How to Use MRI Following Neoadjuvant Chemotherapy (NAC) in Locally Advanced Breast Cancer
 Global Breast Cancer Conference 2016 & 5 th International Breast Cancer Symposium April 29 th 2016, 09:40-10:50 How to Use MRI Following Neoadjuvant Chemotherapy (NAC) in Locally Advanced Breast Cancer
Global Breast Cancer Conference 2016 & 5 th International Breast Cancer Symposium April 29 th 2016, 09:40-10:50 How to Use MRI Following Neoadjuvant Chemotherapy (NAC) in Locally Advanced Breast Cancer
FDG PET/CT for rectal carcinoma radiotherapy treatment planning: comparison of functional volume delineation algorithms and clinical challenges
 JOURNAL OF APPLIED CLINICAL MEDICAL PHYSICS, VOLUME 15, NUMBER 5, 2014 FDG PET/CT for rectal carcinoma radiotherapy treatment planning: comparison of functional volume delineation algorithms and clinical
JOURNAL OF APPLIED CLINICAL MEDICAL PHYSICS, VOLUME 15, NUMBER 5, 2014 FDG PET/CT for rectal carcinoma radiotherapy treatment planning: comparison of functional volume delineation algorithms and clinical
Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers
 日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
PET in Radiation Therapy. Outline. Tumor Segmentation in PET and in Multimodality Images for Radiation Therapy. 1. Tumor segmentation in PET
 Tumor Segmentation in PET and in Multimodality Images for Radiation Therapy Wei Lu, Ph.D. Department of Radiation Oncology Mallinckrodt Institute of Radiology Washington University in St. Louis Outline
Tumor Segmentation in PET and in Multimodality Images for Radiation Therapy Wei Lu, Ph.D. Department of Radiation Oncology Mallinckrodt Institute of Radiology Washington University in St. Louis Outline
Surgical Pathology Issues of Practical Importance
 Surgical Pathology Issues of Practical Importance Anne Moore, MD Medical Oncology Syed Hoda, MD Surgical Pathology The pathologist is central to the team approach needed to manage the patient with breast
Surgical Pathology Issues of Practical Importance Anne Moore, MD Medical Oncology Syed Hoda, MD Surgical Pathology The pathologist is central to the team approach needed to manage the patient with breast
Clinical and pathological portraits of axillary presentation breast cancer and effects of preoperative systemic therapy
 Case Series Clinical and pathological portraits of axillary presentation breast cancer and effects of preoperative systemic therapy Ling Xu 1*, Fang Li 1,2*, Yinhua Liu 1, Xuening Duan 1, Jingming Ye 1,
Case Series Clinical and pathological portraits of axillary presentation breast cancer and effects of preoperative systemic therapy Ling Xu 1*, Fang Li 1,2*, Yinhua Liu 1, Xuening Duan 1, Jingming Ye 1,
Utility of PET-CT for detection of N2 or N3 nodal mestastases in the mediastinum in patients with non-small cell lung cancer (NSCLC)
 Utility of PET-CT for detection of N2 or N3 nodal mestastases in the mediastinum in patients with non-small cell lung cancer (NSCLC) Poster No.: C-1360 Congress: ECR 2015 Type: Scientific Exhibit Authors:
Utility of PET-CT for detection of N2 or N3 nodal mestastases in the mediastinum in patients with non-small cell lung cancer (NSCLC) Poster No.: C-1360 Congress: ECR 2015 Type: Scientific Exhibit Authors:
Descriptor Definition Author s notes TNM descriptors Required only if applicable; select all that apply multiple foci of invasive carcinoma
 S5.01 The tumour stage and stage grouping must be recorded to the extent possible, based on the AJCC Cancer Staging Manual (7 th Edition). 11 (See Tables S5.01a and S5.01b below.) Table S5.01a AJCC breast
S5.01 The tumour stage and stage grouping must be recorded to the extent possible, based on the AJCC Cancer Staging Manual (7 th Edition). 11 (See Tables S5.01a and S5.01b below.) Table S5.01a AJCC breast
Breast Cancer. Saima Saeed MD
 Breast Cancer Saima Saeed MD Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women 1 in 8 women will develop breast cancer Incidence/mortality rates have declined Breast
Breast Cancer Saima Saeed MD Breast Cancer Most common cancer among women in the US 2nd leading cause of death in women 1 in 8 women will develop breast cancer Incidence/mortality rates have declined Breast
PET/CT for Therapy Assessment in Oncology
 PET/CT for Therapy Assessment in Oncology Rodolfo Núñez Miller, M.D. Nuclear Medicine Section Division of Human Health International Atomic Energy Agency Vienna, Austria Clinical Applications of PET/CT
PET/CT for Therapy Assessment in Oncology Rodolfo Núñez Miller, M.D. Nuclear Medicine Section Division of Human Health International Atomic Energy Agency Vienna, Austria Clinical Applications of PET/CT
Endobronchial Ultrasound in the Diagnosis & Staging of Lung Cancer
 Endobronchial Ultrasound in the Diagnosis & Staging of Lung Cancer Dr Richard Booton PhD FRCP Lead Lung Cancer Clinician, Consultant Respiratory Physician & Speciality Director Manchester University NHS
Endobronchial Ultrasound in the Diagnosis & Staging of Lung Cancer Dr Richard Booton PhD FRCP Lead Lung Cancer Clinician, Consultant Respiratory Physician & Speciality Director Manchester University NHS
STAGE CATEGORY DEFINITIONS
 CLINICAL Extent of disease before any treatment y clinical staging completed after neoadjuvant therapy but before subsequent surgery TX Tis Tis (DCIS) Tis (LCIS) Tis (Paget s) T1 T1mi T1a T1b T1c a b c
CLINICAL Extent of disease before any treatment y clinical staging completed after neoadjuvant therapy but before subsequent surgery TX Tis Tis (DCIS) Tis (LCIS) Tis (Paget s) T1 T1mi T1a T1b T1c a b c
PET CT for Staging Lung Cancer
 PET CT for Staging Lung Cancer Rohit Kochhar Consultant Radiologist Disclosures Neither I nor my immediate family members have financial relationships with commercial organizations that may have a direct
PET CT for Staging Lung Cancer Rohit Kochhar Consultant Radiologist Disclosures Neither I nor my immediate family members have financial relationships with commercial organizations that may have a direct
Pathology Report Patient Companion Guide
 Pathology Report Patient Companion Guide Breast Cancer - Understanding Your Pathology Report Pathology Reports can be overwhelming. They contain scientific terms that are unfamiliar and might be a bit
Pathology Report Patient Companion Guide Breast Cancer - Understanding Your Pathology Report Pathology Reports can be overwhelming. They contain scientific terms that are unfamiliar and might be a bit
A Retrospective Analysis of Clinical Utility of AJCC 8th Edition Cancer Staging System for Breast Cancer
 Elmer ress Original Article World J Oncol. 2017;8(3):71-75 A Retrospective Analysis of Clinical Utility of AJCC 8th Edition Cancer Staging System for Breast Cancer Hui Hu a, Wei Wei a, Xin Yi a, Ling Xin
Elmer ress Original Article World J Oncol. 2017;8(3):71-75 A Retrospective Analysis of Clinical Utility of AJCC 8th Edition Cancer Staging System for Breast Cancer Hui Hu a, Wei Wei a, Xin Yi a, Ling Xin
Colorectal Cancer and FDG PET/CT
 Hybrid imaging in colorectal & esophageal cancer Emmanuel Deshayes IAEA WorkShop, November 2017 Colorectal Cancer and FDG PET/CT 1 Clinical background Cancer of the colon and rectum is one of the most
Hybrid imaging in colorectal & esophageal cancer Emmanuel Deshayes IAEA WorkShop, November 2017 Colorectal Cancer and FDG PET/CT 1 Clinical background Cancer of the colon and rectum is one of the most
What is Cancer? Petra Ketterl, MD Medical Oncology and Functional Medicine
 What is Cancer? Petra Ketterl, MD Medical Oncology and Functional Medicine What is Cancer? Layman s terms: cancer starts when cells grow out of control (in any place in the body) and crowd out normal cells
What is Cancer? Petra Ketterl, MD Medical Oncology and Functional Medicine What is Cancer? Layman s terms: cancer starts when cells grow out of control (in any place in the body) and crowd out normal cells
Radiomics: a new era for tumour management?
 Radiomics: a new era for tumour management? Irène Buvat Unité Imagerie Moléculaire In Vivo (IMIV) CEA Service Hospitalier Frédéric Joliot Orsay, France irene.buvat@u-psud.fr http://www.guillemet.org/irene
Radiomics: a new era for tumour management? Irène Buvat Unité Imagerie Moléculaire In Vivo (IMIV) CEA Service Hospitalier Frédéric Joliot Orsay, France irene.buvat@u-psud.fr http://www.guillemet.org/irene
The 7th Edition of TNM in Lung Cancer.
 10th European Conference Perspectives in Lung Cancer. Brussels, March 2009. The 7th Edition of TNM in Lung Cancer. Peter Goldstraw, Consultant Thoracic Surgeon, Royal Brompton Hospital, Professor of Thoracic
10th European Conference Perspectives in Lung Cancer. Brussels, March 2009. The 7th Edition of TNM in Lung Cancer. Peter Goldstraw, Consultant Thoracic Surgeon, Royal Brompton Hospital, Professor of Thoracic
Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer
 Young Investigator Award, Global Breast Cancer Conference 2018 Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer ㅑ Running head: Revisiting estrogen positive tumors
Young Investigator Award, Global Breast Cancer Conference 2018 Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer ㅑ Running head: Revisiting estrogen positive tumors
Impact of BMI on pathologic complete response (pcr) following neo adjuvant chemotherapy (NAC) for locally advanced breast cancer
 Impact of BMI on pathologic complete response (pcr) following neo adjuvant chemotherapy (NAC) for locally advanced breast cancer Rachna Raman, MD, MS Fellow physician University of Iowa hospitals and clinics
Impact of BMI on pathologic complete response (pcr) following neo adjuvant chemotherapy (NAC) for locally advanced breast cancer Rachna Raman, MD, MS Fellow physician University of Iowa hospitals and clinics
Case Scenario 1. 2/15/2011 The patient received IMRT 45 Gy at 1.8 Gy per fraction for 25 fractions.
 Case Scenario 1 1/3/11 A 57 year old white female presents for her annual mammogram and is found to have a suspicious area of calcification, spread out over at least 4 centimeters. She is scheduled to
Case Scenario 1 1/3/11 A 57 year old white female presents for her annual mammogram and is found to have a suspicious area of calcification, spread out over at least 4 centimeters. She is scheduled to
Post Neoadjuvant therapy: issues in interpretation
 Post Neoadjuvant therapy: issues in interpretation Disclosure: Overview D Prognostic features in assessment of post treatment specimens: Tumor size Cellularity Grade Receptors LN Neoadjuvant chemotherapy:
Post Neoadjuvant therapy: issues in interpretation Disclosure: Overview D Prognostic features in assessment of post treatment specimens: Tumor size Cellularity Grade Receptors LN Neoadjuvant chemotherapy:
Accuracy of MRI in the Detection of Residual Breast Cancer after Neoadjuvant Chemotherapy
 Med. J. Cairo Univ., Vol. 85, No. 4, June: 1411-1416, 2017 www.medicaljournalofcairouniversity.net Accuracy of MRI in the Detection of Residual Breast Cancer after Neoadjuvant Chemotherapy MONA A. ABUL-ENEIN,
Med. J. Cairo Univ., Vol. 85, No. 4, June: 1411-1416, 2017 www.medicaljournalofcairouniversity.net Accuracy of MRI in the Detection of Residual Breast Cancer after Neoadjuvant Chemotherapy MONA A. ABUL-ENEIN,
LYMPHATIC DRAINAGE IN THE HEAD & NECK
 LYMPHATIC DRAINAGE IN THE HEAD & NECK Like other parts of the body, the head and neck contains lymph nodes (commonly called glands). Which form part of the overall Lymphatic Drainage system of the body.
LYMPHATIC DRAINAGE IN THE HEAD & NECK Like other parts of the body, the head and neck contains lymph nodes (commonly called glands). Which form part of the overall Lymphatic Drainage system of the body.
INSERM, UMR 1101, LaTIM, University of Brest, IBSAM, Brest, France. Academic department of nuclear Medicine, CHU Milétrie, Poitiers, France.
 Reliability of PET/CT shape and heterogeneity features in functional and morphological components of Non-Small Cell Lung Cancer tumors: a repeatability analysis in a prospective multi-center cohort Marie-Charlotte
Reliability of PET/CT shape and heterogeneity features in functional and morphological components of Non-Small Cell Lung Cancer tumors: a repeatability analysis in a prospective multi-center cohort Marie-Charlotte
PET/CT in Gynaecological Cancers. Stroobants Sigrid, MD, PhD Departement of Nuclear Medicine University Hospital,Antwerp
 PET/CT in Gynaecological Cancers Stroobants Sigrid, MD, PhD Departement of Nuclear Medicine University Hospital,Antwerp Cervix cancer Outline of this talk Initial staging Treatment monitoring/guidance
PET/CT in Gynaecological Cancers Stroobants Sigrid, MD, PhD Departement of Nuclear Medicine University Hospital,Antwerp Cervix cancer Outline of this talk Initial staging Treatment monitoring/guidance
Monitoring neo-adjuvant chemotherapy: comparison of contrast-enhanced spectral mammography (CESM) and MRI versus breast cancer characteristics
 Monitoring neo-adjuvant chemotherapy: comparison of contrast-enhanced spectral mammography (CESM) and MRI versus breast cancer characteristics Poster No.: B-1062 Congress: ECR 2016 Type: Scientific Paper
Monitoring neo-adjuvant chemotherapy: comparison of contrast-enhanced spectral mammography (CESM) and MRI versus breast cancer characteristics Poster No.: B-1062 Congress: ECR 2016 Type: Scientific Paper
Assessment of tumour size in PET/CT lung cancer studies: PET- and CT-based methods compared to pathology
 Cheebsumon et al. EJNMMI Research 2012, 2:56 ORIGINAL RESEARCH Open Access Assessment of tumour size in PET/CT lung cancer studies: PET- and CT-based methods compared to pathology Patsuree Cheebsumon 1,
Cheebsumon et al. EJNMMI Research 2012, 2:56 ORIGINAL RESEARCH Open Access Assessment of tumour size in PET/CT lung cancer studies: PET- and CT-based methods compared to pathology Patsuree Cheebsumon 1,
Quantitative Nuclear Medicine Imaging in Oncology. Susan E. Sharp, MD
 Quantitative Nuclear Medicine Imaging in Oncology Susan E. Sharp, MD Disclosures None Objectives Describe scoring systems used in oncologic nuclear medicine imaging Deauville scoring in lymphoma Curie
Quantitative Nuclear Medicine Imaging in Oncology Susan E. Sharp, MD Disclosures None Objectives Describe scoring systems used in oncologic nuclear medicine imaging Deauville scoring in lymphoma Curie
Introduction. Wilfred Truin 1 Rudi M. H. Roumen. Vivianne C. G. Tjan-Heijnen 2 Adri C. Voogd
 Breast Cancer Res Treat (2017) 164:133 138 DOI 10.1007/s10549-017-4220-x EPIDEMIOLOGY Estrogen and progesterone receptor expression levels do not differ between lobular and ductal carcinoma in patients
Breast Cancer Res Treat (2017) 164:133 138 DOI 10.1007/s10549-017-4220-x EPIDEMIOLOGY Estrogen and progesterone receptor expression levels do not differ between lobular and ductal carcinoma in patients
FDG-18 PET/CT - radiation dose and dose-reduction strategy
 FDG-18 PET/CT - radiation dose and dose-reduction strategy Poster No.: C-1856 Congress: ECR 2014 Type: Authors: Keywords: DOI: Scientific Exhibit P. Nicholson, S. McSweeney, K. O'Regan; Cork/IE Radiation
FDG-18 PET/CT - radiation dose and dose-reduction strategy Poster No.: C-1856 Congress: ECR 2014 Type: Authors: Keywords: DOI: Scientific Exhibit P. Nicholson, S. McSweeney, K. O'Regan; Cork/IE Radiation
Clinical Study The Efficacy of Neoadjuvant Chemotherapy for HER-2-Positive Locally Advanced Breast Cancer and Survival Analysis
 Hindawi Analytical Cellular Pathology Volume 2017, Article ID 1350618, 5 pages https://doi.org/10.1155/2017/1350618 Clinical Study The Efficacy of Neoadjuvant Chemotherapy for HER-2-Positive Locally Advanced
Hindawi Analytical Cellular Pathology Volume 2017, Article ID 1350618, 5 pages https://doi.org/10.1155/2017/1350618 Clinical Study The Efficacy of Neoadjuvant Chemotherapy for HER-2-Positive Locally Advanced
MEASUREMENT OF EFFECT SOLID TUMOR EXAMPLES
 MEASUREMENT OF EFFECT SOLID TUMOR EXAMPLES Although response is not the primary endpoint of this trial, subjects with measurable disease will be assessed by standard criteria. For the purposes of this
MEASUREMENT OF EFFECT SOLID TUMOR EXAMPLES Although response is not the primary endpoint of this trial, subjects with measurable disease will be assessed by standard criteria. For the purposes of this
8th Edition of the TNM Classification for Lung Cancer. Proposed by the IASLC
 8th Edition of the TNM Classification for Lung Cancer Proposed by the IASLC Introduction Stage classification - provides consistency in nomenclature - improves understanding of anatomic extent of tumour
8th Edition of the TNM Classification for Lung Cancer Proposed by the IASLC Introduction Stage classification - provides consistency in nomenclature - improves understanding of anatomic extent of tumour
Position Statement on Management of the Axilla in Patients with Invasive Breast Cancer
 - Official Statement - Position Statement on Management of the Axilla in Patients with Invasive Breast Cancer Sentinel lymph node (SLN) biopsy has replaced axillary lymph node dissection (ALND) for the
- Official Statement - Position Statement on Management of the Axilla in Patients with Invasive Breast Cancer Sentinel lymph node (SLN) biopsy has replaced axillary lymph node dissection (ALND) for the
Los Angeles Radiological Society 62 nd Annual Midwinter Radiology Conference January 31, 2010
 Los Angeles Radiological Society 62 nd Annual Midwinter Radiology Conference January 31, 2010 Self Assessment Module on Nuclear Medicine and PET/CT Case Review FDG PET/CT IN LYMPHOMA AND MELANOMA Submitted
Los Angeles Radiological Society 62 nd Annual Midwinter Radiology Conference January 31, 2010 Self Assessment Module on Nuclear Medicine and PET/CT Case Review FDG PET/CT IN LYMPHOMA AND MELANOMA Submitted
ACRIN 6666 Therapeutic Surgery Form
 S1 ACRIN 6666 Therapeutic Surgery Form 6666 Instructions: Complete a separate S1 form for each separate area of each breast excised with the intent to treat a cancer (e.g. each lumpectomy or mastectomy).
S1 ACRIN 6666 Therapeutic Surgery Form 6666 Instructions: Complete a separate S1 form for each separate area of each breast excised with the intent to treat a cancer (e.g. each lumpectomy or mastectomy).
Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE
 Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE Neoadjuvant Chemotherapy Indications: Management of locally advanced invasive breast cancers including inflammatory breast
Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE Neoadjuvant Chemotherapy Indications: Management of locally advanced invasive breast cancers including inflammatory breast
The Use of PET Scanning in Urologic Oncology
 The Use of PET Scanning in Urologic Oncology Dr Nicholas C. Buchan Uro-oncology Fellow 1 2 Aims To understand the basic concepts underlying PET scanning. Understand the emerging role of PET Scanning for
The Use of PET Scanning in Urologic Oncology Dr Nicholas C. Buchan Uro-oncology Fellow 1 2 Aims To understand the basic concepts underlying PET scanning. Understand the emerging role of PET Scanning for
The Role of Pathologic Complete Response (pcr) as a Surrogate Marker for Outcomes in Breast Cancer: Where Are We Now?
 1 The Role of Pathologic Complete Response (pcr) as a Surrogate Marker for Outcomes in Breast Cancer: Where Are We Now? Terry Mamounas, M.D., M.P.H., F.A.C.S. Medical Director, Comprehensive Breast Program
1 The Role of Pathologic Complete Response (pcr) as a Surrogate Marker for Outcomes in Breast Cancer: Where Are We Now? Terry Mamounas, M.D., M.P.H., F.A.C.S. Medical Director, Comprehensive Breast Program
Pathological complete response in younger and older breast cancer patients
 Clinical research Pathological complete response in younger and older breast cancer patients Agnieszka Kołacińska 1, Justyna Chałubińska 2, Maria Błasińska-Morawiec 3, Izabela Dowgier-Witczak 4, Wojciech
Clinical research Pathological complete response in younger and older breast cancer patients Agnieszka Kołacińska 1, Justyna Chałubińska 2, Maria Błasińska-Morawiec 3, Izabela Dowgier-Witczak 4, Wojciech
UK Interdisciplinary Breast Cancer Symposium. Should lobular phenotype be considered when deciding treatment? Michael J Kerin
 UK Interdisciplinary Breast Cancer Symposium Should lobular phenotype be considered when deciding treatment? Michael J Kerin Professor of Surgery National University of Ireland, Galway and Galway University
UK Interdisciplinary Breast Cancer Symposium Should lobular phenotype be considered when deciding treatment? Michael J Kerin Professor of Surgery National University of Ireland, Galway and Galway University
IAEA RTC. PET/CT and Planning of Radiation Therapy 20/08/2014. Sarajevo (Bosnia & Hercegovina) Tuesday, June :40-12:20 a.
 IAEA RTC PET/CT and Planning of Radiation Therapy Sarajevo (Bosnia & Hercegovina) Tuesday, June 17 2014 11:40-12:20 a.m María José García Velloso Servicio de Medicina Nuclear Clínica Universidad de Navarra
IAEA RTC PET/CT and Planning of Radiation Therapy Sarajevo (Bosnia & Hercegovina) Tuesday, June 17 2014 11:40-12:20 a.m María José García Velloso Servicio de Medicina Nuclear Clínica Universidad de Navarra
The Challenge of Individualizing Loco-Regional Treatments for Patients with Localized Breast Cancer
 The Challenge of Individualizing Loco-Regional Treatments for Patients with Localized Breast Cancer Le défi des traitements locorégionaux individualisés pour les patientes présentant un cancer du sein
The Challenge of Individualizing Loco-Regional Treatments for Patients with Localized Breast Cancer Le défi des traitements locorégionaux individualisés pour les patientes présentant un cancer du sein
Expert Review The Role of Molecular Imaging in Response Prediction in Metastatic Breast Cancer
 Expert Review The Role of Molecular Imaging in Response Prediction in Metastatic Breast Cancer Geraldine Gebhart, MD Institut Jules Bordet Brussels, Belgium Discussants Moderator Lee Lokey, MD prime Oncology
Expert Review The Role of Molecular Imaging in Response Prediction in Metastatic Breast Cancer Geraldine Gebhart, MD Institut Jules Bordet Brussels, Belgium Discussants Moderator Lee Lokey, MD prime Oncology
Retrospective analysis to determine the use of tissue genomic analysis to predict the risk of recurrence in early stage invasive breast cancer.
 Retrospective analysis to determine the use of tissue genomic analysis to predict the risk of recurrence in early stage invasive breast cancer. Goal of the study: 1.To assess whether patients at Truman
Retrospective analysis to determine the use of tissue genomic analysis to predict the risk of recurrence in early stage invasive breast cancer. Goal of the study: 1.To assess whether patients at Truman
1 Introduction. 2 Materials and methods. LI Na 1 LI Yaming 1,* YANG Chunming 2 LI Xuena 1 YIN Yafu 1 ZHOU Jiumao 1
 Nuclear Science and Techniques 20 (2009) 354 358 18 F-FDG PET/CT in diagnosis of skeletal metastases LI Na 1 LI Yaming 1,* YANG Chunming 2 LI Xuena 1 YIN Yafu 1 ZHOU Jiumao 1 1 Department of Nuclear Medicine,
Nuclear Science and Techniques 20 (2009) 354 358 18 F-FDG PET/CT in diagnosis of skeletal metastases LI Na 1 LI Yaming 1,* YANG Chunming 2 LI Xuena 1 YIN Yafu 1 ZHOU Jiumao 1 1 Department of Nuclear Medicine,
Stage: The Language of Cancer
 Stage: The Language of Cancer American Joint Committee on Cancer American College of Surgeons Chicago, IL Validating science. Improving patient care. No materials in this presentation may be repurposed
Stage: The Language of Cancer American Joint Committee on Cancer American College of Surgeons Chicago, IL Validating science. Improving patient care. No materials in this presentation may be repurposed
Claudin-4 Expression in Triple Negative Breast Cancer: Correlation with Androgen Receptors and Ki-67 Expression
 Claudin-4 Expression in Triple Negative Breast Cancer: Correlation with Androgen Receptors and Ki-67 Expression Mona A. Abd-Elazeem, Marwa A. Abd- Elazeem Pathology department, Faculty of Medicine, Tanta
Claudin-4 Expression in Triple Negative Breast Cancer: Correlation with Androgen Receptors and Ki-67 Expression Mona A. Abd-Elazeem, Marwa A. Abd- Elazeem Pathology department, Faculty of Medicine, Tanta
Computer based delineation and follow-up multisite abdominal tumors in longitudinal CT studies
 Research plan submitted for approval as a PhD thesis Submitted by: Refael Vivanti Supervisor: Professor Leo Joskowicz School of Engineering and Computer Science, The Hebrew University of Jerusalem Computer
Research plan submitted for approval as a PhD thesis Submitted by: Refael Vivanti Supervisor: Professor Leo Joskowicz School of Engineering and Computer Science, The Hebrew University of Jerusalem Computer
Evaluation the Correlation between Ki67 and 5 Years Disease Free Survival of Breast Cancer Patients
 BIOSCIENCES BIOTECHNOLOGY RESEARCH ASIA, December 2015. Vol. 12(3), 2221-2225 Evaluation the Correlation between Ki67 and 5 Years Disease Free Survival of Breast Cancer Patients S.M. Hosseini¹, H. Shahbaziyan
BIOSCIENCES BIOTECHNOLOGY RESEARCH ASIA, December 2015. Vol. 12(3), 2221-2225 Evaluation the Correlation between Ki67 and 5 Years Disease Free Survival of Breast Cancer Patients S.M. Hosseini¹, H. Shahbaziyan
WHAT DOES PET IMAGING ADD TO CONVENTIONAL STAGING OF HEAD AND NECK CANCER PATIENTS?
 doi:10.1016/j.ijrobp.2006.12.044 Int. J. Radiation Oncology Biol. Phys., Vol. 68, No. 2, pp. 383 387, 2007 Copyright 2007 Elsevier Inc. Printed in the USA. All rights reserved 0360-3016/07/$ see front
doi:10.1016/j.ijrobp.2006.12.044 Int. J. Radiation Oncology Biol. Phys., Vol. 68, No. 2, pp. 383 387, 2007 Copyright 2007 Elsevier Inc. Printed in the USA. All rights reserved 0360-3016/07/$ see front
Review of adjuvant and neo-adjuvant abstracts from SABCS 2011 January 7 th 2012
 Review of adjuvant and neo-adjuvant abstracts from SABCS 2011 January 7 th 2012 Ruth M. O Regan, MD Professor and Vice-Chair for Educational Affairs, Department of Hematology and Medical Oncology, Emory
Review of adjuvant and neo-adjuvant abstracts from SABCS 2011 January 7 th 2012 Ruth M. O Regan, MD Professor and Vice-Chair for Educational Affairs, Department of Hematology and Medical Oncology, Emory
Comparison of RECIST version 1.0 and 1.1 in assessment of tumor response by computed tomography in advanced gastric cancer
 Original Article Comparison of RECIST version 1.0 and 1.1 in assessment of tumor response by computed tomography in advanced gastric cancer Gil-Su Jang 1 *, Min-Jeong Kim 2 *, Hong-Il Ha 2, Jung Han Kim
Original Article Comparison of RECIST version 1.0 and 1.1 in assessment of tumor response by computed tomography in advanced gastric cancer Gil-Su Jang 1 *, Min-Jeong Kim 2 *, Hong-Il Ha 2, Jung Han Kim
Precision of pre-sirt predictive dosimetry
 International Course on THERANOSTICS AND MOLECULAR RADIOTHERAPY Precision of pre-sirt predictive dosimetry Hugo Levillain Department of Nuclear Medicine Medical Physics Jules Bordet Institute, Université
International Course on THERANOSTICS AND MOLECULAR RADIOTHERAPY Precision of pre-sirt predictive dosimetry Hugo Levillain Department of Nuclear Medicine Medical Physics Jules Bordet Institute, Université
Toru Nakamura 1, Takashi Fukutomi 1, Hitoshi Tsuda 2, Sadako Akashi-Tanaka 1, Kaneyuki Matsuo 1, Chikako Shimizu 1 and Kunihisa Miyakawa 3
 Jpn J Clin Oncol 2000;30(10)453 457 Changes in Findings of Mammography, Ultrasonography and Contrast-enhanced Computed Tomography of Three Histological Complete Responders with Primary Breast Cancer Before
Jpn J Clin Oncol 2000;30(10)453 457 Changes in Findings of Mammography, Ultrasonography and Contrast-enhanced Computed Tomography of Three Histological Complete Responders with Primary Breast Cancer Before
Reproducibility of Uptake Estimates in FDG PET: a Monte Carlo study
 Reproducibility of Uptake Estimates in FDG PET: a Monte Carlo study Juliette Feuardent, Marine Soret, Irène Buvat 1 Abstract Tumor glucose metabolism measurements from Fluoro-deoxyglucose (FDG) Positron
Reproducibility of Uptake Estimates in FDG PET: a Monte Carlo study Juliette Feuardent, Marine Soret, Irène Buvat 1 Abstract Tumor glucose metabolism measurements from Fluoro-deoxyglucose (FDG) Positron
Title: What is the role of pre-operative PET/PET-CT in the management of patients with
 Title: What is the role of pre-operative PET/PET-CT in the management of patients with potentially resectable colorectal cancer liver metastasis? Pablo E. Serrano, Julian F. Daza, Natalie M. Solis June
Title: What is the role of pre-operative PET/PET-CT in the management of patients with potentially resectable colorectal cancer liver metastasis? Pablo E. Serrano, Julian F. Daza, Natalie M. Solis June
ORIGINAL PAPER. Department of Radiology, Division of Nuclear Medicine and PET Center, Hyogo College of Medicine, Nishinomiya, Japan 2
 Nagoya J. Med. Sci. 80. 183 197, 2018 doi:10.18999/nagjms.80.2.183 ORIGINAL PAPER Assessment of tumor response to neoadjuvant chemotherapy in patients with breast cancer using MRI and FDG-PET/CT-RECIST
Nagoya J. Med. Sci. 80. 183 197, 2018 doi:10.18999/nagjms.80.2.183 ORIGINAL PAPER Assessment of tumor response to neoadjuvant chemotherapy in patients with breast cancer using MRI and FDG-PET/CT-RECIST
The Neoadjuvant Model as a Translational Tool for Drug and Biomarker Development in Breast Cancer
 The Neoadjuvant Model as a Translational Tool for Drug and Biomarker Development in Breast Cancer Laura Spring, MD Breast Medical Oncology Massachusetts General Hospital Primary Mentor: Dr. Aditya Bardia
The Neoadjuvant Model as a Translational Tool for Drug and Biomarker Development in Breast Cancer Laura Spring, MD Breast Medical Oncology Massachusetts General Hospital Primary Mentor: Dr. Aditya Bardia
Molecular in vitro diagnostic test for the quantitative detection of the mrna expression of ERBB2, ESR1, PGR and MKI67 in breast cancer tissue.
 Innovation for your breast cancer diagnostics PGR G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C ERBB2
Innovation for your breast cancer diagnostics PGR G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C ERBB2
Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery
 Last Review Status/Date: December 2016 Page: 1 of 6 Intraoperative Assessment of Surgical Description Breast-conserving surgery as part of the treatment of localized breast cancer is optimally achieved
Last Review Status/Date: December 2016 Page: 1 of 6 Intraoperative Assessment of Surgical Description Breast-conserving surgery as part of the treatment of localized breast cancer is optimally achieved
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/55957 holds various files of this Leiden University dissertation Author: Dekker T.J.A. Title: Optimizing breast cancer survival models based on conventional
Cover Page The handle http://hdl.handle.net/1887/55957 holds various files of this Leiden University dissertation Author: Dekker T.J.A. Title: Optimizing breast cancer survival models based on conventional
Molecular in vitro diagnostic test for the quantitative detection of the mrna expression of ERBB2, ESR1, PGR and MKI67 in breast cancer tissue.
 Innovation for your breast cancer diagnostics Now valid ated on: -Roche cob as z 480 A nalyzer -Roche L ightcycle r 480 II - ABI 750 0 Fast -Versant kpcr Cyc ler PGR ESR1 MKI67 Molecular in vitro diagnostic
Innovation for your breast cancer diagnostics Now valid ated on: -Roche cob as z 480 A nalyzer -Roche L ightcycle r 480 II - ABI 750 0 Fast -Versant kpcr Cyc ler PGR ESR1 MKI67 Molecular in vitro diagnostic
Positron emission tomography predicts survival in malignant pleural mesothelioma
 Flores et al General Thoracic Surgery Positron emission tomography predicts survival in malignant pleural mesothelioma Raja M. Flores, MD, a Timothy Akhurst, MD, b Mithat Gonen, PhD, c Maureen Zakowski,
Flores et al General Thoracic Surgery Positron emission tomography predicts survival in malignant pleural mesothelioma Raja M. Flores, MD, a Timothy Akhurst, MD, b Mithat Gonen, PhD, c Maureen Zakowski,
Whole-tumor apparent diffusion coefficient measurements in nephroblastoma: Can it identify blastemal predominance? Abstract Purpose To explore the
 Whole-tumor apparent diffusion coefficient measurements in nephroblastoma: Can it identify blastemal predominance? Abstract Purpose To explore the potential relation between whole-tumor apparent diffusion
Whole-tumor apparent diffusion coefficient measurements in nephroblastoma: Can it identify blastemal predominance? Abstract Purpose To explore the potential relation between whole-tumor apparent diffusion
INSERM : U613, Universit é de Bretagne Occidentale - Brest, Etablissement Francais du Sang 46, Rue Felix Le Dantec Brest Cedex 2,FR
 Intratumor heterogeneity characterized by textural features on baseline F-FDG PET images predicts response to concomitant radiochemotherapy in esophageal cancer Florent Tixier 1*, Catherine Cheze Le Rest,
Intratumor heterogeneity characterized by textural features on baseline F-FDG PET images predicts response to concomitant radiochemotherapy in esophageal cancer Florent Tixier 1*, Catherine Cheze Le Rest,
Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:
 Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:7332-7341 Presented by Deming Mi 7/25/2006 Major reasons for few prognostic factors to
Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:7332-7341 Presented by Deming Mi 7/25/2006 Major reasons for few prognostic factors to
