Citation. As Published Publisher. Version
|
|
|
- Brook Bryan
- 5 years ago
- Views:
Transcription
1 Helminth co-infection in Helicobacter pylori infected INS- GAS mice attenuates gastric premalignant lesions of epithelial dysplasia and glandular atrophy and preserves The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Whary, Mark T., Sureshkumar Muthupalani, Zhongming Ge, Yan Feng, Jennifer Lofgren, Hai Ning Shi, Nancy S. Taylor, et al. Helminth Co-Infection in Helicobacter Pylori Infected INS-GAS Mice Attenuates Gastric Premalignant Lesions of Epithelial Dysplasia and Glandular Atrophy and Preserves Colonization Resistance of the Stomach to Lower Bowel Microbiota. Microbes and Infection 16, no. 4 (April 2014): Elsevier Version Author's final manuscript Accessed Tue Sep 04 05:45:56 EDT 2018 Citable Link Terms of Use Creative Commons Attribution Detailed Terms
2 NIH Public Access Author Manuscript Published in final edited form as: Microbes Infect April ; 16(4): doi: /j.micinf Helminth co-infection in Helicobacter pylori infected INS-GAS mice attenuates gastric premalignant lesions of epithelial dysplasia and glandular atrophy and preserves colonization resistance of the stomach to lower bowel microbiota Mark T. Whary *,a, Sureshkumar Muthupalani a, Zhongming Ge a, Yan Feng a, Jennifer Lofgren a, Hai Ning Shi b, Nancy S. Taylor a, Pelayo Correa c, James Versalovic d, Timothy C. Wang e, and James G. Fox a a Division of Comparative Medicine, Massachusetts Institute of Technology, Cambridge, MA, USA b Mucosal Immunology Laboratory, Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA c Department of Medicine, Vanderbilt University School of Medicine, Nashville, Tennessee, USA d Department of Pathology, Baylor College of Medicine and Texas Children's Hospital, Houston, TX, USA e Division of Digestive and Liver Disease, Columbia University Medical Center, New York, NY, USA Abstract Higher prevalence of helminth infections in H. pylori infected children was suggested to potentially lower the life-time risk for gastric adenocarcinoma. In rodent models, helminth coinfection does not reduce Helicobacter-induced inflammation but delays progression of premalignant gastric lesions. Because gastric cancer in INS-GAS mice is promoted by intestinal microflora, the impact of Heligmosomoides polygyrus co-infection on H. pylori-associated gastric lesions and microflora were evaluated. Male INS-GAS mice co-infected with H. pylori and H. polygyrus for 5 months were assessed for gastrointestinal lesions, inflammation-related mrna expression, FoxP3 + cells, epithelial proliferation, and gastric colonization with H. pylori and Altered Schaedler Flora. Despite similar gastric inflammation and high levels of proinflammatory mrna, helminth co-infection increased FoxP3 + cells in the corpus and reduced H. pyloriassociated gastric atrophy (p<0.04), dysplasia (p<0.02) and prevented H. pylori-induced changes in the gastric flora (p<0.05). This is the first evidence of helminth infection reducing H. pyloriinduced gastric lesions while inhibiting changes in gastric flora, consistent with prior observations that gastric colonization with enteric microbiota accelerated gastric lesions in INS-GAS mice Elsevier Masson SAS. All rights reserved. * Corresponding Author: Mark T Whary, 77 Mass Ave A, Cambridge, MA 02139, , FAX , mwhary@mit.edu. Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
3 Whary et al. Page 2 Identifying how helminths reduce gastric premalignant lesions and impact bacterial colonization of the H. pylori infected stomach could lead to new treatment strategies to inhibit progression from chronic gastritis to cancer in humans. Keywords Helicobacter pylori; Heligmosomoides polygyrus; Altered Schaedler Flora; INS-GAS mice; gastric cancer 1. Introduction Chronic Helicobacter pylori-induced gastritis in humans can progress to gastric atrophy, intestinal metaplasia and dysplasia with increased risk for gastric adenocarcinoma [1]. Epidemiologic evidence in humans and data from rodent models support that gastric carcinogenesis is multifactorial, influenced by host genetics [2], hormones [3], H. pylori virulence properties [4], environmental co-factors [5, 6] and co-infections with enterohepatic Helicobacters [7, 8] or helminths [9]. Higher prevalence of helminth infections in H. pylori infected children was suggested to potentially lower the life-time risk for gastric adenocarcinoma [9], with serologic evidence that life-long exposure through adulthood to a variety of parasites impacts inflammatory responses to H. pylori [10]. Experimental data from rodent models further support this hypothesis: gastric atrophy was reduced in H. felis and Heligmosomoides polygyrus co-infected C57BL/6 mice [11] and in H. pylori and Brugia pahangi co-infected gerbils [12]. Notably in rodent models, helminth co-infection does not reduce Helicobacter-induced inflammation but does delay progression of pre-malignant lesions, suggesting these lesions are suppressed by an alternative mechanism not directly related to severity of the inflammatory response. Evidence from human patients [13, 14] supports that gastric cancer risk may be promoted by enteric microbiota colonizing the gastric epithelium as a sequela to H. pylori-induced gastric atrophy and hypochlorhydria. Recent results from rodent models reinforce the potential importance of non-h. pylori bacteria in accelerating gastric pathology [15][16]. Transgenic INS-GAS mice, particularly males [17-19], develop hypergastrinemia, gastric hyperplasia of the foveolar and glandular epithelium, loss of chief and parietal cells (hypochlorhydric atrophy) and severe dysplasia of gastric glands that can invade through the muscularis mucosa of the corpus. Lesion progression was delayed by months in gnotobiotic INS-GAS mice monoassociated with H. pylori compared to INS-GAS mice that commonly develop intraepithelial neoplasia [20]when colonized with H. pylori and complex (normal) enteric microbiota [15]. Additional experiments demonstrated that gnotobiotic INS-GAS colonized with H. pylori and a restricted, select set of 3 members of Altered Schaedler Flora (ASF 356 Clostridium sp., ASF 361 Lactobacillus sp., ASF 519 Bacteroides sp.), developed gastritis and premalignant gastric lesions equivalent to H. pylori infected INS-GAS mice with complex microflora (e.g. specific pathogen free mice)[16]. These results support prior findings that chronic oral administration of proton pump inhibitors to achieve gastric hypochlorhydria in H. pylori-infected gerbils promoted the progression of atrophic corpus gastritis to adenocarcinoma [21, 22].
4 Whary et al. Page 3 These studies in rodents and observations that gastric cancer patients were found to be colonized with bacteria from 5 other bacterial phyla in addition to H. pylori [23], suggest colonization efficiency, rather than pathogenic potential, of specific lower bowel microflora is an important contributor to gastric cancer development. Because H. polygyrus prevented gastric atrophy in C57BL/6 mice co-infected with H. felis [11] and promoted levels of probiotic-type bacteria in the proximal intestine of mice [24], we assessed potential changes in gastric bacterial colonization in H. pylori and H. polygyrus co-infected male INS-GAS mice at 5 months post H. pylori infection when gastric atrophy in this model is well established. 2. Materials and methods 2.1. Infection models 2.2. Necropsy Mouse model Male, transgenic INS-GAS mice on a FVB/N background (Tg (Ins1-GAS) 1Sbr) [25] were produced by in-house breeding and maintained specific pathogen-free (SPF) of exogenous murine viruses, pathogenic bacteria (including Helicobacter) and ecto- and endoparasites. Mice were housed in an Association for Assessment and Accreditation of Laboratory Animal Care-accredited facility under environmental conditions of a 12:12 light / dark cycle, temperature maintenance at 20 ± 1 C and relative humidity range of 30-70%. Mice were group housed in microisolator caging on hardwood bedding and provided reverse osmosis water and pelleted diet (ProLab 2000, Purina Mills, St. Louis, MO) ad libitum. Animal use was approved by the MIT Committee on Animal Care Experimental infection with Heligmosomoides polygyrus H. polygyrus were cultivated and prepared for dosing as previously described [26]. At 6 weeks of age, 23 male INS-GAS mice were orally dosed with 200 third stage (L3) H. polygyrus larvae in 200 μl. At 9 and 14 days post infection, mice were treated orally with 172 mg/kg of pyrantel pamoate (Apothecary Products. Minneapolis, MN) in 200 μl to eliminate adult parasites from the intestine. Mice were re-infected with 200 L3 larvae 1 week and 4 months following the first pyrantel pamoate dosing to mimic chronic exposure to helminths. Persistent infection with H. polygyrus was confirmed by elevated serum IgE levels at 2, 3 and 4 months post infection (mpi), observation of adult worms at necropsy, and histologic evidence of multiple life stages in the duodenum Experimental infection with Helicobacter pylori At 10 weeks of age, 10 of the H. polygyrus infected and 13 naïve male, INS-GAS mice were orally gavaged with colony forming units of H. pylori SS1 in 200 μl every other day for a total of 3 doses. Controls included 9 non-dosed, uninfected mice and 13 mice infected with H. polygyrus alone. Persistent infection with H. pylori was confirmed by quantitative PCR (qpcr) of gastric tissues [8]. Mice were euthanized with carbon dioxide and necropsied at 5 mpi with H. pylori except for 3 H. pylori and H. polygyrus co-infected mice that were evaluated at 4 mpi to evaluate
5 Whary et al. Page 4 progression of pathology and infection with H. pylori and H. polygyrus. The stomach was incised along the greater curvature and individual linear gastric strips from lesser curvature of the gastric cardia into the proximal duodenum were collected. These were either fixed in formalin or were flash-frozen in liquid nitrogen and subsequently stored at -80 C pending RNA and DNA extraction to quantify mrna expression and colonization levels of H. pylori and 8 ASF using qpcr. Tissues fixed in 10% buffered formalin were paraffin embedded and 5 μm sections were processed routinely for histology and immunohistochemistry Lesion scoring Tissue sections were scored for gastric lesions using previously published criteria [6] by a board certified veterinary pathologist (S.M.) blinded to sample identity. Briefly, pathology subfeatures of the gastric corpus, including inflammation (extent of inflammatory infiltrates into the mucosa and submucosa), epithelial defects (degeneration of the surface epithelium and underlying glands), mucous metaplasia, hyalinosis, epithelial hyperplasia (elongation of gastric glands lined by foveolar and glandular lining cells), pseudopyloric metaplasia (abnormal mucosa resembling pyloric antrum in glandular phenotype and mucin expression, analogous to findings in H. pylori infected humans), oxyntic gland atrophy (loss of chief and parietal cells) and dysplasia (based on a combination of the extent of glandular architectural distortion and degree of cellular atypia) were evaluated and graded on an ascending scale from 0 to 4. For all categories, an increment of 0.5 was used in some instances when the degree of histological changes was considered to fall between two grades represented by ordinal values. Dysplastic / neoplastic changes were graded as low (score= 1), moderate (score= 2), high (score=3, non-invasive high grade dysplasia, carcinoma in situ), or invasive neoplasms (score of 3.5 or 4). A score of 3.5 represented intramucosal carcinoma with invasion into lamina propria or muscularis mucosa and a score of 4 was assigned to submucosal carcinoma or beyond. Both invasive and non-invasive high grade dysplasia/gastrointraepithelial neoplasia- type (GIN) lesions were considered to be gastric cancers similar to that described in human literature [27, 28]. Differentiation of truly invasive glands from glandular herniation or pseudoinvasion into the submucosa in the murine gastrointestinal tract can be problematic and the guidelines established by Boivin et. al. [20] were specifically followed in this study for histological evaluation of the stomach. Excluding mucous metaplasia and hyalinosis which can occur independent of H. pylori infections, the scores for all other sub-feature categories were totaled to represent a gastric histopathologic activity index (GHAI) with a maximum possible score of 24 [6] Immunohistochemistry Immunohistochemistry was performed to phenotype Ki67 + and FoxP3 + cells in the gastric mucosa of 5 to 7 INS-GAS mice from each infection or control group at 5 mpi using previously described methods for antigen retrieval, blocking steps and counterstaining [29]. Formalin-fixed gastric sections were stained for the regulatory T cell marker FoxP3 using the FJK-16S antibody (ebiosciences, San Diego, CA) and nuclear cell proliferation marker Ki67 using mouse anti-human antibody (# BD Biosciences), with color development using the #K3954 Dako Art kit (Dako, Carpinteria, CA) per instructions. For assessment of epithelial cell proliferation lining the gastric glands, Ki67 labeling indices were calculated (using a 40 microscope objective lens) by the number of positively stained nuclei per distinct linear gastric gland averaged over 10 well-
6 Whary et al. Page 5 oriented glands in the gastric corpus. At the same magnification, cells expressing FoxP3 + in the gastric corpus were counted and averaged over 10 representative fields of the gastric mucosa and submucosa. Nuclear labeling was considered specific for regulatory T cells, whereas granular cytoplasmic staining of oxyntic cells, if any, was considered as nonspecific background staining mrna expression levels of cytokines and inos mrna expression levels in gastric sections were measured by qpcr as previously described [15]. Total RNA from flash-frozen gastric tissue was extracted with Trizol reagent (Invitrogen, Carlsbad, CA) and cdna synthesized from 5 μg of total RNA using the High Capacity cdna Archive kit (Applied Biosystems). mrna levels for IL-10, TGFβ, IL-17A, IL-17F, IL-1β, TNFα, IFNγ, inos, and GAPDH (endogenous control) were quantified using TaqMan gene expression assays (Applied Biosystems, Foster City, CA) in the 7500 FAST Sequence Detection System. mrna levels were normalized to GAPDH expression with baseline comparisons made to uninfected mice using the ΔΔCT method (User Bulletin #2, Applied Biosystems) as previously described [15]. 2.4 Colonization levels of H. pylori and Altered Schaedler Flora DNA was extracted from the stomach using the High Pure PCR Template Preparation Kit (Roche Molecular Biochemicals, Indianapolis, IN). As previously published, samples were probed with 18S rrna-based primers for quantifying mouse DNA (Applied Biosystems) and with H. pylori DNA-specific primers and probe based on the H. pylori ureb gene [8]. ASF copy numbers were measured using SyBr-based qpcr using the 7500 Fast Sequence Detection System (Applied Biosystems) [30, 31]. Plasmid DNA containing the 16S rdna of each of the 8 ASF species was used to generate standard curves of six 10-fold dilutions, ranging from 10 6 to 10 copies. Subsequently, the bacterial DNA quantity was converted into copy number of each ASF genome based on the number of 16S rrna gene copies. ASF copy numbers were then normalized to μg of mouse chromosomal DNA. 2.5 ELISA for serum IgE response to H. polygyrus 2.6 Statistical analysis Five randomly selected mice from each experimental group were bled at 2, 3 and 4 months post H. polygyrus dosing to monitor serum IgE levels in response to helminth infection as previously described [32]. The ELISA plates were coated with rat anti-mouse antibody to IgE (BD Biosciences, Franklin Lakes NJ), incubated with diluted sera, and IgE detected with biotin-conjugated rat anti-mouse IgE (BD Biosciences) and peroxidase-conjugated streptavidin (Invitrogen). Optical density values developed with O-phenylenediamine at 492 nm were converted to ng/ml of IgE by comparison with a standard curve of purified IgE (BD Biosciences) by linear regression analysis, and are expressed as the mean concentration for each group of mice ± standard deviation. Except for gastric lesion scores, all other data analyzed statistically were from mice necropsied at a time point equivalent to 5 mpi with H. pylori. Gastric lesion scores were combined from mice necropsied at both 4 and 5 mpi because scores were statistically
7 Whary et al. Page 6 3. Results equivalent. Data analysis for GHAI, subfeature lesion scores, Ki67 and FoxP3 immunohistochemistry were compared using the Mann-Whitney nonparametric test. The Fishers exact test was used to evaluate the incidence and odds ratio of developing GIN. Cytokine and inos mrna expression levels and log-transformed qpcr colonization data for H. pylori and ASF were evaluated using the Student t test. Statistical analysis was performed using GraphPad Prism 5.0 (GraphPad Software, Inc., La Jolla, CA). Results were considered significant at p< Heligmosomoides polygyrus infection caused persistent inflammatory responses INS-GAS mice were infected with L3 stage H. polygyrus, treated with an anthelminthic and orally redosed with larvae two additional times. Multiple life stages of H. polygyrus were observed grossly at necropsy (adult worms) and in histologic sections of the duodenum (degenerate larvae and adults) (not shown). The majority of H. polygyrus infected mice developed duodenitis ranging from mild neutrophilic infiltration to severe mural-serosal, pyogranulomatous lesions, consisting of neutrophilic and mononuclear inflammation in response to degenerate and necrotic nematodes in the wall of the small intestine. As additional evidence of chronic immune stimulation from H. polygyrus, the total mean ± standard deviation of serum IgE levels in H. polygyrus infected mice were 166 ± 82 ng/ml, 179 ± 82 ng/ml in H. pylori and H. polygyrus coinfected mice, compared to significantly lower levels in mice infected with H. pylori alone (19 ± 28 ng/ml) (p<0.002) Co-infection with Heligmosomoides polygyrus reduced premalignant and malignant gastric lesions in Helicobacter pylori infected INS-GAS mice Control, uninfected INS-GAS mice or mice infected with H. polygyrus alone had similar gastric histopathologic activity indices (median and range GHAI of 8 ( ) for controls; 8.5 (6-10.5) for H. polygyrus alone) with low to moderate severity scores for subfeatures of gastritis, epithelial defects, foveolar hyperplasia, glandular atrophy, pseudopyloric metaplasia and dysplasia (Fig. 1A-F, 2A-H). In the absence of H. pylori infection, lesions consisted mainly of foveolar hyperplasia (Fig. 1C, 2B) and early dysplasia (Fig. 1F, 2B) that were attributable to hypergastrinemia as previously reported [15, 19]. Compared to controls, INS-GAS mice mono-infected with H. pylori developed gastritis that was most severe in the corpus (Fig. 1A, 2C). H. pylori infected mice had higher GHAI (median and range GHAI of 16 (14-18) compared to uninfected controls (8 ( ); p<0.0001) attributable to significantly greater inflammation, epithelial defects, hyperplasia, atrophy, metaplasia and dysplasia (Fig. 2A-F) (p<0.0002). INS-GAS mice infected with H. pylori had significant distortion of normal gastric architecture with cystic mucous metaplasia of glands in the corpus (Fig. 2C-D), epithelial defects, hyperplasia, oxyntic atrophy, pseudopyloric metaplasia and dysplasia, accompanied by edema and mild inflammatory infiltrates in the submucosa (Fig. 2C-F). High grade dysplastic glands frequently contained necrotic, apoptotic cellular debris (Fig. 2E) and in some instances exhibited budding nests of invasive cells in the lamina propria consistent with the diagnosis of intramucosal carcinoma (dysplasia score of 3.5). Occasionally, one or more dysplastic glandular units were seen
8 Whary et al. Page 7 either partially of fully extending vertically down into the submucosa. Close examination confirmed the presence of a discernible peripheral muscularis mucosa and so these lesions were considered as herniated glands or pseudoinvasion, a frequent finding in mouse GI tracts which must be differentiated on the basis of true invasion [20] (Fig. 2F). Notably, 8 of 12 male INS-GAS mice infected with H. pylori alone developed non-invasive neoplasms and 2 of 12 mice developed invasion of dysplastic glands into the lamina propria, fulfilling the definition of intramucosal carcinomas (Fig. 2E). Compared to mice mono-infected with H. pylori, INS-GAS mice co-infected with H. pylori and H. polygyrus had similar degrees of inflammation, epithelial defects, hyperplasia and pseudopyloric metaplasia (Fig. 2A-E). Immunolabeling for Ki67, a marker of epithelial cell proliferation, supported lesion scoring of hematoxylin and eosin-stained sections that H. pylori infection resulted in significant epithelial hyperplasia (p<0.004) that was not reduced by H. polygyrus co-infection (Fig. 3A). Notably however, gastric atrophy (p<0.04) and dysplasia (p<0.02) were reduced by H. polygyrus co-infection (Fig. 1D, 1F and 2H). Noninvasive high grade dysplasia was evident in only 4 of 10 co-infected mice and none had developed invasive neoplasms (intramucosal carcinoma or submucosal carcinoma) (Fig. 1F). Although herniation of dysplastic glands was noted in mice infected with H. pylori and in mice co-infected with H. polygyrus, the extent of herniation and degree of cytological atypia were greater in the H. pylori infected mice. The odds ratio for developing GIN in mice mono-infected with H. pylori was 11.7 (p<0.03: 95% CI ) compared to mice coinfected with H. pylori and H. polygyrus FoxP3 + cells were elevated in number in the gastric lamina propria of INS-GAS mice co-infected with H. pylori and H. polygyrus Immunohistochemistry labeling for the regulatory T cell marker FoxP3 demonstrated that the gastric tissues from H. pylori infected INS-GAS mice contained significantly elevated numbers of FoxP3 + regulatory T cells compared to uninfected controls or mice infected with H. polygyrus alone (p<0.01)(fig. 6B). Gastric tissues from INS-GAS mice co-infected with H. pylori and H. polygyrus contained statistically higher numbers of FoxP3 + regulatory T cells than mice mono-infected with H. pylori (p<0.03) Cytokine and inos mrna responses to H. pylori were unaffected by co-infection with H. polygyrus Mice mono-infected with H. pylori developed robust (15 to 100+ fold) mrna expression levels in the gastric corpus for proinflammatory IFN-γ, IL-17F, IL-17A, TNFα, and inos (all p<0.0001)(table 1). Mice co-infected with H. polygyrus and H. pylori developed similar, elevated levels of proinflammatory gene expression that were equivalent to levels observed in mice infected with H. pylori alone. Proinflammatory IL-1β and antiinflammatory TGFβ and IL-10 mrna expression levels were statistically elevated by H. pylori and H. pylori / H. polygyrus co-infection (p<0.05) but were orders of magnitude lower than the expression levels of the pro-inflammatory genes. IL-13 expression was also assessed but mrna levels were at background levels. The duodenitis noted in H. polygyrus challenged mice did not alter expression of proinflammatory genes in the corpus as mrna
9 Whary et al. Page 8 levels were similar between mice infected with H. polygyrus alone and uninfected control mice. 4. Discussion By qpcr assay, gastric colonization of H. pylori was not detected in 2 of 12 mice dosed with H. pylori alone (Fig. 4) which is attributable to gastrin-associated gastritis that is further promoted by H. pylori infection. Gastric lesions in these mice were similar to the other mice mono-infected with H. pylori: therefore, data on these mice were included in the 5 mpi analysis. Gastric colonization levels of H. pylori were similar in the remaining H. pylori infected mice irrespective of co-infection with H. polygyrus. Seven of 8 ASF species were detected by qpcr in gastric samples; the extremely oxygen-sensitive ASF 492, which clusters taxonomically with Eubacterium plexicaudatum [33] was undetectable in all samples. H. pylori infection promoted gastric colonization levels of 4 of the 7 ASF species (ASF 356 Clostridium sp., ASF 361 Lactobacillus sp., ASF 457 Mucispirillum schaedleri, ASF 500 Clostridium sp.) by approximately 1 log (p<0.02), whereas H. polygyrus coinfection prevented increased gastric colonization with 3 of these same ASF (ASF 356, ASF 457 and ASF 500) (p<0.05). Colonization levels observed in H. pylori and H. polygyrus coinfected mice were comparable to uninfected control mice (Fig. 4). Colonization of ASF 360 Lactobacillus sp., ASF 502 Clostridium sp. and ASF 519 Bacteroides sp. were at levels comparable to the other detectable ASF but their colonization levels were unaffected by either H. pylori or H. polygyrus infection. As previously reported, H. pylori infection in male INS-GAS mice caused gastritis, foveolar hyperplasia, gastric atrophy, pseudopyloric metaplasia and progression of chronic gastritis to invasive gastrointraepithelial neoplasia (GIN) [17, 19]. GIN was first described for lower bowel carcinomas in rodents [20] and has histologic features compatible with gastric adenocarcinoma in humans [34]. The increase in gastric colonization with ASF in H. pylori infected INS-GAS mice supports the hypothesis that lower bowel microflora may contribute to gastric carcinogenesis in humans. H. polygyrus co-infection modulated both of these features; gastric atrophy, dysplasia and the incidence of invasive GIN were reduced and colonization resistance of the stomach to ASF was maintained. Dysplasia leads to non-invasive and invasive neoplasms. GIN is equivalent to high grade dysplasia. The results from the current study demonstrate that H. polygyrus infection reduced gastric atrophy and dysplasia, and in particular, reduced the extent of dysplastic glands invading through the muscularis mucosa into the lamina propria. The odds ratio for developing GIN in mice infected with H. pylori alone was significantly elevated compared to mice co-infected with H. pylori and H. polygyrus. Consistent with prior data on helminth and Helicobacter co-infection in mice [11] and in gerbils [12], H. polygyrus co-infection did not reduce gastric inflammation. Thus, in three rodent models, helminthiasis reduced Helicobacter-associated gastric atrophy without reduction in gastric inflammation per se, supporting the hypothesis that H. pylori-associated premalignant gastric lesions are prevented, at least in part, by an alternative mechanism not directly related to severity of gastric inflammation.
10 Whary et al. Page 9 Although ASF have historically been used to colonize germfree mice with a defined flora to establish normal gut physiology and to establish commercially-reared mice under SPF conditions, phylogenetic classification of these 8 anaerobes based on 16SrRNA taxonomy [33] and spatial distribution within the mouse gastrointestinal tract [30] are relatively recent. Little is known about the long-term impact on gut physiology or pathogenic potential of the 8 member species of ASF. Acknowledging that gastric colonization with various other bacterial species may have increased or decreased secondary to H. pylori or H. polygyrus infection, ASF were used in this study to monitor microbial population shifts in gastric bacteria as we have previously demonstrated altered population dynamics in the lower bowel after infection with H. trogontum in C57BL/6 IL-10 -/- mice [35] and in outbred Swiss mice infected with H. hepaticus [31]. The increase in gastric colonization with ASF in H. pylori infected INS-GAS mice supports the hypothesis that lower bowel microflora may contribute to gastric carcinogenesis in humans. The potential role for enteric bacteria not typically associated with gastric disease to promote H. pylori-associated gastritis and gastric cancer is also supported by our previous observations. Although H. pylori as a monoinfection caused gastritis in gnotobiotic INS-GAS mice, lesions were less severe in comparison to H. pylori infected SPF INS-GAS mice with a complex enteric microbiota under otherwise similar experimental conditions [15]. We also recently demonstrated that gnotobiotic INS-GAS mice colonized with just 3 members of ASF replicated the promotion of neoplastic lesions by diverse intestinal flora in the H. pylori INS-GAS mouse model [16]. In this current study, 4 ASF species colonized the H. pylori infected stomach at significantly higher levels and suggests that parietal cell loss from chronic inflammation and subsequent increase in gastric ph enables a variety of enteric bacteria to colonize the hypochlorhydric stomach, as shown in H. pylori infected INS-GAS mice in which members of the Bacteroidetes phyla were reduced and Firmicutes phyla were increased in the stomach compared to uninfected INS-GAS mice [15]. Similarly, gastric bacterial overgrowth occurs in mice with genetic or chemically induced hypochlorhydria [36] and chronic dosing of proton pump inhibitors that increase gastric ph in H. pylori infected gerbils promoted the progression of atrophic corpus gastritis to adenocarcinoma [21, 22]. Opportunistic bacteria could further drive inflammatory responses in the stomach, accelerating atrophy and dysplasia by means of mutagenic properties or virulence factors that otherwise impair DNA repair systems and thus contribute to carcinogenesis [37, 38]. Indeed, human studies profiling the gastric microbiota in H. pylori infected and uninfected subjects have demonstrated a wide diversity of bacterial phylotypes in the stomach [39, 40]. A small number of gastric cancer patients were found to be colonized with low numbers of H. pylori and significantly, bacteria from 5 other bacterial phyla [23]. The significance of these initial observations warrants further epidemiologic studies in humans and in animal models such as INS-GAS mice where genetic and environmental variability inherent in human subjects can be minimized. Notably ASF 361 (Lactobacillus sp.) colonized the stomach of H. pylori infected and H. polygyrus co-infected mice at higher levels than control mice. Others have shown that H. polygyrus infection in C57BL/6 mice increased the abundance of Lactobacillaceae in the ileum [24]. In combination with hypochlorhydria known to develop in INS-GAS mice [19,
11 Whary et al. Page 10 25], increased ASF 361 colonization in the stomach may in part be attributable to the relative oxygen resistance of ASF 361 compared to the other obligate anaerobic ASF [30]. Gastric colonization with 3 of 4 ASF were not increased in helminth co-infected mice, coincident with elevated numbers of regulatory T cells and less gastric atrophy and dysplasia. These findings evoke a putative mechanism involving helminth-mediated, regulatory T cell preservation of parietal cell mass and maintenance of colonization resistance of the acidic stomach to lower bowel microflora. The natural niche for H. polygyrus is the duodenum of the mouse, but its impact on host physiology and immune responses are systemic. H. polygyrus infection increases permeability of the colonic mucosal barrier [41], induces regulatory T cells [42] and the helminth is known to be immunosuppressive in a variety of inflammation models [11, 32, 43-45] that clearly point to a mechanism involving immune regulation [46]. H. polygyrus stimulates innate [44, 47] as well as cell-mediated and humoral immune responses [41]. The H. polygyrus re-infection protocol used in this study is designed to maintain a Th2-biased immune response but also models chronic exposure of children to helminths who have received periodic anthelminthic therapy. This reinfection with H. polygyrus may also have contributed to duodenitis in response to degenerate H. polygyrus larvae and adult worms. As this lesion was observed in H. polygyrus infected mice irrespective of H. pylori co-infection or severity of gastritis, the significance may be limited except the same lesion may develop in children living in low socioeconomic conditions where parasitism constitutes a significant morbidity in addition to endemic H. pylori infection. Alternatively, INS-GAS mice infected with H. pylori or co-infected with H. polygyrus developed similar, but very robust IL-17, IFNγ, TNFα and inos responses in gastric tissues with low expression of antiinflammatory TGFβ and IL-10 responses and minimal IL-13 expression, suggesting concurrent duodenitis may have muted the anticipated TH2-like cytokine response to helminths, particularly given the length of co-infection in the current experiment. The robust Th2-associated inflammatory response and the differentiation of IL-10 producing, FoxP3 + regulatory T cells [42] and regulatory dendritic cells [48] that result from acute H. polygyrus infection are evolutionarily important for expulsion of the parasite from the host or alternatively, for the parasite to establish persistent infection. Compared to low numbers of cells expressing the regulatory T cell marker FoxP3 in uninfected controls and mice infected with H. polygyrus alone, FoxP3 + cell numbers were increased in the gastric mucosa and submucosa of H. pylori infected INS-GAS mice and were highest in H. pylori and H. polygyrus co-infected mice. This suggests that H. polygyrus promoted regulatory T cell development in lymph nodes which were then recruited to the inflamed stomach and appear to have preserved parietal cell function and maintained acidic gastric ph. In autoimmune mice [49], regulatory T cells suppressed production of inflammatory cytokines in the stomach and prevented parietal cell loss through release of chemokines which regulated effector T cell trafficking into the stomach. These results support that H. pylori-associated gastric atrophy allow opportunistic colonization of the achlorhydric human stomach with lower bowel microflora and that ongoing gastritis and progression of premalignant lesions may occur despite clearance of H. pylori infection when gastric atrophy is irreversible [22]. Our previous studies illustrated the
12 Whary et al. Page 11 Acknowledgments References importance of enteric colonization in promoting H. pylori-associated gastric cancer in several INS-GAS mouse models [15][16]. In addition, we reported experimental evidence for this concept when INS-GAS mice underwent antibiotic eradication of H. pylori, and likely other microbiota that may have colonized the atrophic gastric epithelium, had reduced gastric cancer risk in a time-dependent manner [27, 50]. Rodent models of Helicobacter and helminth co-infection have reproduced epidemiologic observations of lower life-time risk for gastric adenocarcinoma in H. pylori infected humans living under heavy environmental exposure to parasites, particularly in young children who commonly acquire H. pylori and helminths concurrently [9]. Identifying how helminths impact bacterial colonization of the H. pylori infected stomach through immune modulation or other mechanisms could lead to new treatment strategies to reduce malignant sequelae from chronic gastritis in humans. We thank Melissa Mobley and Amanda Potter who performed the necropsies and sample collection. Supported in part by RO1-CA67529, R01DK052413, PO1CA26731, P01 CA028842, P30ESO2109, R01DK (JGF) and R01DK (HNS) from the National Institutes of Health. 1. Correa P, Piazuelo MB. The gastric precancerous cascade. J Dig Dis. 2012; 13:2 9. [PubMed: ] 2. McLean MH, El-Omar EM. Genetics of inflammation in the gastrointestinal tract and how it can cause cancer. Recent Results Cancer Res. 2011; 185: [PubMed: ] 3. Camargo MC, Goto Y, Zabaleta J, Morgan DR, Correa P, Rabkin CS. Sex hormones, hormonal interventions, and gastric cancer risk: a meta-analysis. Cancer Epidemiol Biomarkers Prev. 2012; 21: [PubMed: ] 4. de Sablet T, Piazuelo MB, Shaffer CL, Schneider BG, Asim M, Chaturvedi R, Bravo LE, Sicinschi LA, Delgado AG, Mera RM, Israel DA, Romero-Gallo J, Peek RM Jr, Cover TL, Correa P, Wilson KT. Phylogeographic origin of Helicobacter pylori is a determinant of gastric cancer risk. Gut. 2011; 60: [PubMed: ] 5. Fox JG, Dangler CA, Taylor NS, King A, Koh TJ, Wang TC. High-salt diet induces gastric epithelial hyperplasia and parietal cell loss, and enhances Helicobacter pylori colonization in C57BL/6 mice. Cancer Res. 1999; 59: [PubMed: ] 6. Rogers AB, Taylor NS, Whary MT, Stefanich ED, Wang TC, Fox JG. Helicobacter pylori but not high salt induces gastric intraepithelial neoplasia in B6129 mice. Cancer Res. 2005; 65: [PubMed: ] 7. Lemke LB, Ge Z, Whary MT, Feng Y, Rogers AB, Muthupalani S, Fox JG. Concurrent Helicobacter bilis infection in C57BL/6 mice attenuates proinflammatory H. pylori-induced gastric pathology. Infect Immun. 2009; 77: [PubMed: ] 8. Ge Z, Feng Y, Muthupalani S, Eurell LL, Taylor NS, Whary MT, Fox JG. Coinfection with enterohepatic Helicobacter species can ameliorate or promote Helicobacter pylori-induced gastric pathology in C57BL/6 mice. Infect Immun. 2011; 79: [PubMed: ] 9. Whary MT, Sundina N, Bravo LE, Correa P, Quinones F, Caro F, Fox JG. Intestinal helminthiasis in Colombian children promotes a Th2 response to Helicobacter pylori: possible implications for gastric carcinogenesis. Cancer Epidemiol Biomarkers Prev. 2005; 14: [PubMed: ] 10. Ek C, Whary MT, Ihrig M, Bravo LE, Correa P, Fox JG. Serologic evidence that ascaris and toxoplasma infections impact inflammatory responses to Helicobacter pylori in Colombians. Helicobacter. 2012; 17: [PubMed: ]
13 Whary et al. Page Fox JG, Beck P, Dangler CA, Whary MT, Wang TC, Shi HN, Nagler-Anderson C. Concurrent enteric helminth infection modulates inflammation and gastric immune responses and reduces helicobacter-induced gastric atrophy. Nat Med. 2000; 6: [PubMed: ] 12. Martin HR, Shakya KP, Muthupalani S, Ge Z, Klei TR, Whary MT, Fox JG. Brugia filariasis differentially modulates persistent Helicobacter pylori gastritis in the gerbil model. Microbes Infect. 2010; 12: [PubMed: ] 13. Kuipers EJ. Proton pump inhibitors and gastric neoplasia. Gut. 2006; 55: [PubMed: ] 14. Fox J, Sheh A. The role of the gastrointestinal microbiome in Helicobacter pylori pathogenesis. Gut Microbes. 2013; Lofgren JL, Whary MT, Ge Z, Muthupalani S, Taylor NS, Mobley M, Potter A, Varro A, Eibach D, Suerbaum S, Wang TC, Fox JG. Lack of commensal flora in Helicobacter pylori-infected INS- GAS mice reduces gastritis and delays intraepithelial neoplasia. Gastroenterology. 2011; 140: [PubMed: ] 16. Lertpiriyapong K, Whary MT, Muthupalani S, Lofgren JL, Gamazon ER, Feng Y, Ge Z, Wang TC, Fox JG. Gastric colonisation with a restricted commensal microbiota replicates the promotion of neoplastic lesions by diverse intestinal microbiota in the Helicobacter pylori INS-GAS mouse model of gastric carcinogenesis. Gut. 2014; 63: [PubMed: ] 17. Fox JG, Rogers AB, Ihrig M, Taylor NS, Whary MT, Dockray G, Varro A, Wang TC. Helicobacter pylori-associated gastric cancer in INS-GAS mice is gender specific. Cancer Res. 2003; 63: [PubMed: ] 18. Sheh A, Lee CW, Masumura K, Rickman BH, Nohmi T, Wogan GN, Fox JG, Schauer DB. Mutagenic potency of Helicobacter pylori in the gastric mucosa of mice is determined by sex and duration of infection. Proc Natl Acad Sci U S A. 2010; 107: [PubMed: ] 19. Wang TC, Dangler CA, Chen D, Goldenring JR, Koh T, Raychowdhury R, Coffey RJ, Ito S, Varro A, Dockray GJ, Fox JG. Synergistic interaction between hypergastrinemia and Helicobacter infection in a mouse model of gastric cancer. Gastroenterology. 2000; 118: [PubMed: ] 20. Boivin GP, Washington K, Yang K, Ward JM, Pretlow TP, Russell R, Besselsen DG, Godfrey VL, Doetschman T, Dove WF, Pitot HC, Halberg RB, Itzkowitz SH, Groden J, Coffey RJ. Pathology of mouse models of intestinal cancer: consensus report and recommendations. Gastroenterology. 2003; 124: [PubMed: ] 21. Hagiwara T, Mukaisho K, Nakayama T, Sugihara H, Hattori T. Long-term proton pump inhibitor administration worsens atrophic corpus gastritis and promotes adenocarcinoma development in Mongolian gerbils infected with Helicobacter pylori. Gut. 2011; 60: [PubMed: ] 22. Fox JG, Kuipers EJ. Long-term proton pump inhibitor administration, H. pylori and gastric cancer: lessons from the gerbil. Gut. 2011; 60: [PubMed: ] 23. Dicksved J, Lindberg M, Rosenquist M, Enroth H, Jansson JK, Engstrand L. Molecular characterization of the stomach microbiota in patients with gastric cancer and in controls. J Med Microbiol. 2009; 58: [PubMed: ] 24. Walk ST, Blum AM, Ewing SA, Weinstock JV, Young VB. Alteration of the murine gut microbiota during infection with the parasitic helminth Heligmosomoides polygyrus. Inflamm Bowel Dis. 2010; 16: [PubMed: ] 25. Wang TC, Koh TJ, Varro A, Cahill RJ, Dangler CA, Fox JG, Dockray GJ. Processing and proliferative effects of human progastrin in transgenic mice. J Clin Invest. 1996; 98: [PubMed: ] 26. Shi HN, Scott ME, Koski KG, Boulay M, Stevenson MM. Energy restriction and severe zinc deficiency influence growth, survival and reproduction of Heligmosomoides polygyrus (Nematoda) during primary and challenge infections in mice. Parasitology. 1995; 110(Pt 5): [PubMed: ] 27. Lee CW, Rickman B, Rogers AB, Muthupalani S, Takaishi S, Yang P, Wang TC, Fox JG. Combination of sulindac and antimicrobial eradication of Helicobacter pylori prevents progression
14 Whary et al. Page 13 of gastric cancer in hypergastrinemic INS-GAS mice. Cancer Res. 2009; 69: [PubMed: ] 28. Schlemper RJ, Kato Y, Stolte M. Diagnostic criteria for gastrointestinal carcinomas in Japan and Western countries: proposal for a new classification system of gastrointestinal epithelial neoplasia. J Gastroenterol Hepatol. 2000; 15:G Suppl. [PubMed: ] 29. Rogers AB, Cormier KS, Fox JG. Thiol-reactive compounds prevent nonspecific antibody binding in immunohistochemistry. Lab Invest. 2006; 86: [PubMed: ] 30. Sarma-Rupavtarm RB, Ge Z, Schauer DB, Fox JG, Polz MF. Spatial distribution and stability of the eight microbial species of the altered Schaedler flora in the mouse gastrointestinal tract. Appl Environ Microbiol. 2004; 70: [PubMed: ] 31. Ge Z, Feng Y, Taylor NS, Ohtani M, Polz MF, Schauer DB, Fox JG. Colonization dynamics of altered Schaedler flora is influenced by gender, aging, and Helicobacter hepaticus infection in the intestines of Swiss Webster mice. Appl Environ Microbiol. 2006; 72: [PubMed: ] 32. Shi HN, Ingui CJ, Dodge I, Nagler-Anderson C. A helminth-induced mucosal Th2 response alters nonresponsiveness to oral administration of a soluble antigen. J Immunol. 1998; 160: [PubMed: ] 33. Dewhirst FE, Chien CC, Paster BJ, Ericson RL, Orcutt RP, Schauer DB, Fox JG. Phylogeny of the defined murine microbiota: altered Schaedler flora. Appl Environ Microbiol. 1999; 65: [PubMed: ] 34. Fox JG, Wang TC. Inflammation, atrophy, and gastric cancer. J Clin Invest. 2007; 117: [PubMed: ] 35. Whary MT, Danon SJ, Feng Y, Ge Z, Sundina N, Ng V, Taylor NS, Rogers AB, Fox JG. Rapid onset of ulcerative typhlocolitis in B6.129P2-IL10tm1Cgn (IL-10-/-) mice infected with Helicobacter trogontum is associated with decreased colonization by altered Schaedler's flora. Infect Immun. 2006; 74: [PubMed: ] 36. Zavros Y, Rieder G, Ferguson A, Samuelson LC, Merchant JL. Genetic or chemical hypochlorhydria is associated with inflammation that modulates parietal and G-cell populations in mice. Gastroenterology. 2002; 122: [PubMed: ] 37. Touati E. When bacteria become mutagenic and carcinogenic: lessons from H. pylori. Mutat Res. 2010; 703: [PubMed: ] 38. Meira LB, Bugni JM, Green SL, Lee CW, Pang B, Borenshtein D, Rickman BH, Rogers AB, Moroski-Erkul CA, McFaline JL, Schauer DB, Dedon PC, Fox JG, Samson LD. DNA damage induced by chronic inflammation contributes to colon carcinogenesis in mice. J Clin Invest. 2008; 118: [PubMed: ] 39. Bik EM, Eckburg PB, Gill SR, Nelson KE, Purdom EA, Francois F, Perez-Perez G, Blaser MJ, Relman DA. Molecular analysis of the bacterial microbiota in the human stomach. Proc Natl Acad Sci U S A. 2006; 103: [PubMed: ] 40. Sheh A, Piazuelo MB, Wilson KT, Correa P, Fox JG. Draft genome sequences of Helicobacter pylori strains isolated from regions of low and high gastric cancer risk in Colombia. Genome Announcements. 2013; Su CW, Cao Y, Kaplan J, Zhang M, Li W, Conroy M, Walker WA, Shi HN. Duodenal helminth infection alters barrier function of the colonic epithelium via adaptive immune activation. Infect Immun. 2011; 79: [PubMed: ] 42. Grainger JR, Smith KA, Hewitson JP, McSorley HJ, Harcus Y, Filbey KJ, Finney CA, Greenwood EJ, Knox DP, Wilson MS, Belkaid Y, Rudensky AY, Maizels RM. Helminth secretions induce de novo T cell Foxp3 expression and regulatory function through the TGF-beta pathway. J Exp Med. 2010; 207: [PubMed: ] 43. Weng M, Huntley D, Huang IF, Foye-Jackson O, Wang L, Sarkissian A, Zhou Q, Walker WA, Cherayil BJ, Shi HN. Alternatively activated macrophages in intestinal helminth infection: effects on concurrent bacterial colitis. J Immunol. 2007; 179: [PubMed: ] 44. Hang L, Setiawan T, Blum AM, Urban J, Stoyanoff K, Arihiro S, Reinecker HC, Weinstock JV. Heligmosomoides polygyrus infection can inhibit colitis through direct interaction with innate immunity. J Immunol. 2010; 185: [PubMed: ]
15 Whary et al. Page Shi M, Wang A, Prescott D, Waterhouse CC, Zhang S, McDougall JJ, Sharkey KA, McKay DM. Infection with an intestinal helminth parasite reduces Freund's complete adjuvant-induced monoarthritis in mice. Arthritis Rheum. 2011; 63: [PubMed: ] 46. Smith KA, Hochweller K, Hammerling GJ, Boon L, MacDonald AS, Maizels RM. Chronic helminth infection promotes immune regulation in vivo through dominance of CD11c lo CD103 - dendritic cells. J Immunol. 2011; 186: [PubMed: ] 47. Sutton TL, Zhao A, Madden KB, Elfrey JE, Tuft BA, Sullivan CA, Urban JF Jr, Shea-Donohue T. Anti-Inflammatory mechanisms of enteric Heligmosomoides polygyrus infection against trinitrobenzene sulfonic acid-induced colitis in a murine model. Infect Immun. 2008; 76: [PubMed: ] 48. Li Z, Liu G, Chen Y, Liu Y, Liu B, Su Z. The phenotype and function of naturally existing regulatory dendritic cells in nematode-infected mice. Int J Parasitol. 2011; 41: [PubMed: ] 49. Nguyen TL, Sullivan NL, Ebel M, Teague RM, DiPaolo RJ. Antigen-specific TGF-beta-induced regulatory T cells secrete chemokines, regulate T cell trafficking, and suppress ongoing autoimmunity. J Immunol. 2011; 187: [PubMed: ] 50. Lee CW, Rickman B, Rogers AB, Ge Z, Wang TC, Fox JG. Helicobacter pylori eradication prevents progression of gastric cancer in hypergastrinemic INS-GAS mice. Cancer Res. 2008; 68: [PubMed: ]
16 Whary et al. Page 15 Fig. 1. Median scores for gastric inflammation, epithelial defects, epithelial (foveolar/glandular hyperplasia), gastric atrophy, pseudopyloric metaplasia and dysplasia. At 5 months post infection (mpi) with H. pylori (Hp)(n=12), median scores for all features were significantly higher than controls (n=9) or mice infected with H. polygyrus (Hpoly) (n=13) alone. Compared to Hp infected mice, gastric atrophy and dysplasia were significantly less severe in H. pylori and H. polygyrus co-infected mice (HpHpoly) and the incidence of GIN was lower (n=10).
17 Whary et al. Page 16 Fig. 2. A-H. (A) Representative hematoxylin and eosin stained image of the gastric corpus from a control, uninfected male INS-GAS mouse necropsied at a time equivalent to 5 months post infection (mpi) for H. pylori. Bar = 400 μm. (B) Gastric corpus from a male INS-GAS mouse infected with H. polygyrus 6 mpi, equivalent to 5 mpi for H. pylori. The extent of epithelial hyperplasia and oxyntic loss was low to moderate with sparse inflammation and minimal dysplasia and is similar to the uninfected control male INS-GAS mice in Panel A. Bar = 400 μm. (C-E) Representative low and high power magnification of hematoxylin and eosin stained images of the gastric corpus from a male INS-GAS mouse 5 mpi with H. pylori. In the low magnification image (C) (Bar = 400 μm), there is severe diffuse pathomorphological alterations in the corpus characterized by significant distortion of normal gastric glandular columnar architecture with numerous cystic, dilated glands,
18 Whary et al. Page 17 prominent foveolar and glandular epithelial hyperplasia, metaplasia, oxyntic loss or atrophy, with mild to moderate inflammation in the mucosa and submucosa. This lesion has high grade dysplasia with rare invasive glands in the lamina propria consistent with gastrointestinal intramucosal carcinoma as well as partially herniated glands (lined by peripheral muscularis mucosa) in the submucosa. A high magnification focus of the proximal corpus as shown in (D) (Bar = 75 μm) revealing loss of glandular columnar orientation with arborizing, dysplastic and ectatic atypical glands containing intraluminal cellular ghosts against a background of distinct stromal inflammation. (E) is a higher magnification image (Bar = 40 μm) showing dysplastic glands with cellular atypia, necrosis and apoptosis of cells that are sloughing into the lumen, loss of basal lining and invasion of dysplastic epithelial cells into the lamina propria, thus fulfilling the histological criteria for gastrointestinal intramucosal carcinoma. A high magnification image (F) (Bar = 75 μm) of the proximal corpus from a male INS-GAS mouse 5 mpi with H. pylori showing herniation of a group of dysplastic gastric glands through the muscularis mucosa into the submucosa in a region of submucosal inflammation. (G) Proximal corpus from a male INS-GAS mouse 5 mpi with H. pylori and H. polygyrus at low magnification (Bar = 400 μm). Inflammation and hyperplasia remain significant but there is a slight reduction in the overall severity of epithelial distortion/defects, glandular metaplasia, oxyntic atrophy and extent of dysplasia as compared to the age-matched H. pylori monoinfected INS-GAS as shown in (C). (H) Higher magnification image from (G) depicting an area of glandular metaplasia, dysplastic and ectatic glands with intraluminal cellular debris but absence of lamina propria invasion (Bar = 75 μm).
19 Whary et al. Page 18 Fig. 3. The gastric corpus from 5 to 7 mice per group were labeled by immunohistochemistry to enumerate Ki67 (A) and FoxP3 positive cells (B). Compared to baseline values from uninfected control INS-GAS mice, (A) the mean (± St. Dev.) Ki67 labeling index, reflecting epithelial (foveolar/glandular) hyperplasia, was significantly higher in mice infected with H. pylori (Hp). The number of Ki67+ cells was similarly high in mice co-infected with H. pylori and H. polygyrus (HpHpoly). (B). the mean (± St. Dev.) FoxP3 labeling index, reflecting regulatory T cells, was significantly higher in mice infected with H. pylori (Hp).
The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters.
 Gastric colonisation with a restricted commensal microbiota replicates the promotion of neoplastic lesions by diverse intestinal microbiota in the Helicobacter pylori The MIT Faculty has made this article
Gastric colonisation with a restricted commensal microbiota replicates the promotion of neoplastic lesions by diverse intestinal microbiota in the Helicobacter pylori The MIT Faculty has made this article
Helicobacter pylori Eradication Prevents Progression of Gastric Cancer in Hypergastrinemic INS-GAS Mice
 Research Article Helicobacter pylori Eradication Prevents Progression of Gastric Cancer in Hypergastrinemic INS-GAS Mice Chung-Wei Lee, 1,2 Barry Rickman, 1 Arlin B. Rogers, 1 Zhongming Ge, 1 Timothy C.
Research Article Helicobacter pylori Eradication Prevents Progression of Gastric Cancer in Hypergastrinemic INS-GAS Mice Chung-Wei Lee, 1,2 Barry Rickman, 1 Arlin B. Rogers, 1 Zhongming Ge, 1 Timothy C.
ESMO Preceptorship Gastrointestinal Tumours Valencia October 2017
 www.esmo.org ESMO Preceptorship Valencia 06-07 October 2017 Gastrointestinal Tumours I have no conflicts of interest to declare Fátima Carneiro Gastrointestinal tumours Multidisciplinary management, standards
www.esmo.org ESMO Preceptorship Valencia 06-07 October 2017 Gastrointestinal Tumours I have no conflicts of interest to declare Fátima Carneiro Gastrointestinal tumours Multidisciplinary management, standards
The enteric microbiota: Implications for IBD. Eugene B. Chang, M.D. University of Chicago
 The enteric microbiota: Implications for IBD Eugene B. Chang, M.D. University of Chicago On a per cell basis, humans are mostly prokaryote 100 90 80 70 60 50 40 30 20 10 0 EuK ProK The microbial flora
The enteric microbiota: Implications for IBD Eugene B. Chang, M.D. University of Chicago On a per cell basis, humans are mostly prokaryote 100 90 80 70 60 50 40 30 20 10 0 EuK ProK The microbial flora
Intestinal Microbiota in Health and Disease
 Intestinal Microbiota in Health and Disease February 27, 2015 Master s Course in Gastroenterology Prof. Kathy McCoy 1 Overview Overview of Gut Microbiota Microbiota in Health Microbiota in Disease 2 Gut
Intestinal Microbiota in Health and Disease February 27, 2015 Master s Course in Gastroenterology Prof. Kathy McCoy 1 Overview Overview of Gut Microbiota Microbiota in Health Microbiota in Disease 2 Gut
Gastric adenocarcinoma of the
 Comment From the Editors Spasmolytic Polypeptide- Expressing Metaplasia and Intestinal Metaplasia: Time for Reevaluation of Metaplasias and the Origins of Gastric Cancer Gastric adenocarcinoma of the intestinal
Comment From the Editors Spasmolytic Polypeptide- Expressing Metaplasia and Intestinal Metaplasia: Time for Reevaluation of Metaplasias and the Origins of Gastric Cancer Gastric adenocarcinoma of the intestinal
Concurrent Helicobacter bilis Infection in C57BL/6 Mice Attenuates Proinflammatory H. pylori-induced Gastric Pathology
 INFECTION AND IMMUNITY, May 2009, p. 2147 2158 Vol. 77, No. 5 0019-9567/09/$08.00 0 doi:10.1128/iai.01395-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Concurrent Helicobacter
INFECTION AND IMMUNITY, May 2009, p. 2147 2158 Vol. 77, No. 5 0019-9567/09/$08.00 0 doi:10.1128/iai.01395-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Concurrent Helicobacter
Gut Microbiota and IBD. Vahedi. H M.D Associate Professor of Medicine DDRI
 Gut Microbiota and IBD Vahedi. H M.D Associate Professor of Medicine DDRI 1393.3.1 2 GUT MICROBIOTA 100 Trillion Microbes - 10 times more than cells in our body Collective weight of about 1kg in human
Gut Microbiota and IBD Vahedi. H M.D Associate Professor of Medicine DDRI 1393.3.1 2 GUT MICROBIOTA 100 Trillion Microbes - 10 times more than cells in our body Collective weight of about 1kg in human
Accepted Manuscript. Innate immune cells regulate oncoimmunity and cancer development. Ai-Ping Bai, Yuan Guo
 Accepted Manuscript Innate immune cells regulate oncoimmunity and cancer development Ai-Ping Bai, Yuan Guo PII: S0016-5085(18)34974-6 DOI: 10.1053/j.gastro.2018.08.057 Reference: YGAST 62119 To appear
Accepted Manuscript Innate immune cells regulate oncoimmunity and cancer development Ai-Ping Bai, Yuan Guo PII: S0016-5085(18)34974-6 DOI: 10.1053/j.gastro.2018.08.057 Reference: YGAST 62119 To appear
Faecalibacterium prausnitzii
 Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients PNAS 105(43): 16731-16736, 2008. Speaker: Ming-Cheng Chen Advisor:
Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients PNAS 105(43): 16731-16736, 2008. Speaker: Ming-Cheng Chen Advisor:
Update on the pathological classification of gastritis. Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada
 Update on the pathological classification of gastritis Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada CLASSIFICATION GASTRITIS GASTROPATHY 1. Acute 2. Chronic 3. Uncommon
Update on the pathological classification of gastritis Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada CLASSIFICATION GASTRITIS GASTROPATHY 1. Acute 2. Chronic 3. Uncommon
Histopathology: gastritis and peptic ulceration
 Histopathology: gastritis and peptic ulceration These presentations are to help you identify, and to test yourself on identifying, basic histopathological features. They do not contain the additional factual
Histopathology: gastritis and peptic ulceration These presentations are to help you identify, and to test yourself on identifying, basic histopathological features. They do not contain the additional factual
New developments in pathogenesis, gastric cancer. Matthias Ebert. II. Medizinische Klinik Klinikum rechts der Isar TU München
 New developments in pathogenesis, diagnosis, therapy and prevention of gastric cancer Matthias Ebert II. Medizinische Klinik Klinikum rechts der Isar TU München Gastric Cancer Pathogenesis Diagnosis Treatment
New developments in pathogenesis, diagnosis, therapy and prevention of gastric cancer Matthias Ebert II. Medizinische Klinik Klinikum rechts der Isar TU München Gastric Cancer Pathogenesis Diagnosis Treatment
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
Genetics. Environment. You Are Only 10% Human. Pathogenesis of IBD. Advances in the Pathogenesis of IBD: Genetics Leads to Function IBD
 Advances in the Pathogenesis of IBD: Genetics Leads to Function Pathogenesis of IBD Environmental Factors Microbes Scott Plevy, MD Associate Professor of Medicine, Microbiology & Immunology UNC School
Advances in the Pathogenesis of IBD: Genetics Leads to Function Pathogenesis of IBD Environmental Factors Microbes Scott Plevy, MD Associate Professor of Medicine, Microbiology & Immunology UNC School
The effect of proton pump inhibitors on the gastric mucosal microenvironment
 Original papers The effect of proton pump inhibitors on the gastric mucosal microenvironment Yen-Chun Peng,,, A F, Lan-Ru Huang, A, C, E, Hui-Ching Ho, C, Chi-Sen Chang, C, E, Shou-Wu Lee, E, Ching-Chang
Original papers The effect of proton pump inhibitors on the gastric mucosal microenvironment Yen-Chun Peng,,, A F, Lan-Ru Huang, A, C, E, Hui-Ching Ho, C, Chi-Sen Chang, C, E, Shou-Wu Lee, E, Ching-Chang
Gastric atrophy: use of OLGA staging system in practice
 Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Histopathology of Endoscopic Resection Specimens from Barrett's Esophagus
 Histopathology of Endoscopic Resection Specimens from Barrett's Esophagus Br J Surg 38 oct. 1950 Definition of Barrett's esophagus A change in the esophageal epithelium of any length that can be recognized
Histopathology of Endoscopic Resection Specimens from Barrett's Esophagus Br J Surg 38 oct. 1950 Definition of Barrett's esophagus A change in the esophageal epithelium of any length that can be recognized
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
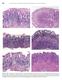 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
Supplemental Figures: Supplemental Figure 1
 Supplemental Figures: Supplemental Figure 1 Suppl. Figure 1. BM-DC infection with H. pylori does not induce cytotoxicity and treatment of BM-DCs with H. pylori sonicate, but not heat-inactivated bacteria,
Supplemental Figures: Supplemental Figure 1 Suppl. Figure 1. BM-DC infection with H. pylori does not induce cytotoxicity and treatment of BM-DCs with H. pylori sonicate, but not heat-inactivated bacteria,
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Barrett s Esophagus: Old Dog, New Tricks
 Barrett s Esophagus: Old Dog, New Tricks Stuart Jon Spechler, M.D. Chief, Division of Gastroenterology, VA North Texas Healthcare System; Co-Director, Esophageal Diseases Center, Professor of Medicine,
Barrett s Esophagus: Old Dog, New Tricks Stuart Jon Spechler, M.D. Chief, Division of Gastroenterology, VA North Texas Healthcare System; Co-Director, Esophageal Diseases Center, Professor of Medicine,
Murine Models of. Inflammatory Bowel Disease. Outline. Laura P. Hale, M.D. Ph.D. Professor of Pathology Duke University Medical Center
 Murine Models of Inflammatory Bowel Disease Laura P. Hale, M.D. Ph.D. Professor of Pathology Duke University Medical Center September 19, 2017 Outline Review of IBD in humans Murine models of IBD Tissue
Murine Models of Inflammatory Bowel Disease Laura P. Hale, M.D. Ph.D. Professor of Pathology Duke University Medical Center September 19, 2017 Outline Review of IBD in humans Murine models of IBD Tissue
Cyclooxygenase-2 Expression in Gastric Antral Mucosa Before and After Eradication of Helicobacter pylori Infection
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 94, No. 5, 1999 1999 by Am. Coll. of Gastroenterology ISSN 0002-9270/99/$20.00 Published by Elsevier Science Inc. PII S0002-9270(99)00126-4 Cyclooxygenase-2
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 94, No. 5, 1999 1999 by Am. Coll. of Gastroenterology ISSN 0002-9270/99/$20.00 Published by Elsevier Science Inc. PII S0002-9270(99)00126-4 Cyclooxygenase-2
XV Jornada de Avances en Hepatología Málaga 20 y 21 de Mayo de Barrera intestinal en la cirrosis. Agustín Albillos
 XV Jornada de Avances en Hepatología Málaga 20 y 21 de Mayo de 2016 Barrera intestinal en la cirrosis Agustín Albillos Hospital Universitario Ramón y Cajal Universidad de Alcalá, Ciberehd Madrid, Spain
XV Jornada de Avances en Hepatología Málaga 20 y 21 de Mayo de 2016 Barrera intestinal en la cirrosis Agustín Albillos Hospital Universitario Ramón y Cajal Universidad de Alcalá, Ciberehd Madrid, Spain
5/2/2018. Low Grade Dysplasia of GI Tract. High Grade Dysplasia of GI Tract. Dysplasia in Gastrointestinal Tract: Practical Pearls and Issues
 Dysplasia in Gastrointestinal Tract: Practical Pearls and Issues Arief Suriawinata, M.D. Professor of Pathology and Laboratory Medicine Geisel School of Medicine at Dartmouth Department of Pathology and
Dysplasia in Gastrointestinal Tract: Practical Pearls and Issues Arief Suriawinata, M.D. Professor of Pathology and Laboratory Medicine Geisel School of Medicine at Dartmouth Department of Pathology and
Role of the Gut Microbiota in Autoimmunity
 Role of the Gut Microbiota in Autoimmunity Pavan Bhargava, MD - Neuroimmunology Fellow Division of Neuroimmunology and Neurological Infections Johns Hopkins University, Baltimore, MD. May, 2015 None Disclosures
Role of the Gut Microbiota in Autoimmunity Pavan Bhargava, MD - Neuroimmunology Fellow Division of Neuroimmunology and Neurological Infections Johns Hopkins University, Baltimore, MD. May, 2015 None Disclosures
The surface mucous cells and the cardiac and pyloric glands secrete mucus which protects the stomach from self-digestion.
 PATHOLOGY OF THE STOMACH Stomach mucosa Gastric mucosa is covered by a layer of mucus. The mucosal glands comprise the cardiac glands, the fundic glands in the fundus and body of the stomach, and the pyloric
PATHOLOGY OF THE STOMACH Stomach mucosa Gastric mucosa is covered by a layer of mucus. The mucosal glands comprise the cardiac glands, the fundic glands in the fundus and body of the stomach, and the pyloric
Morphologic Criteria of Invasive Colonic Adenocarcinoma on Biopsy Specimens
 ISPUB.COM The Internet Journal of Pathology Volume 12 Number 1 Morphologic Criteria of Invasive Colonic Adenocarcinoma on Biopsy Specimens C Rose, H Wu Citation C Rose, H Wu.. The Internet Journal of Pathology.
ISPUB.COM The Internet Journal of Pathology Volume 12 Number 1 Morphologic Criteria of Invasive Colonic Adenocarcinoma on Biopsy Specimens C Rose, H Wu Citation C Rose, H Wu.. The Internet Journal of Pathology.
Mouse Models of Gastric Cancer
 Cancers 2013, 5, 92-130; doi:10.3390/cancers5010092 Review OPEN ACCESS cancers ISSN 2072-6694 www.mdpi.com/journal/cancers Mouse Models of Gastric Cancer Yoku Hayakawa 1, James G. Fox 2, Tamas Gonda 1,
Cancers 2013, 5, 92-130; doi:10.3390/cancers5010092 Review OPEN ACCESS cancers ISSN 2072-6694 www.mdpi.com/journal/cancers Mouse Models of Gastric Cancer Yoku Hayakawa 1, James G. Fox 2, Tamas Gonda 1,
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
NIH Public Access Author Manuscript Gastroenterology. Author manuscript; available in PMC 2013 September 11.
 NIH Public Access Author Manuscript Published in final edited form as: Gastroenterology. 2010 June ; 138(7): 2207 2210.e1. doi:10.1053/j.gastro.2010.04.023. SPEM and IM: Time for re-evaluation of metaplasias
NIH Public Access Author Manuscript Published in final edited form as: Gastroenterology. 2010 June ; 138(7): 2207 2210.e1. doi:10.1053/j.gastro.2010.04.023. SPEM and IM: Time for re-evaluation of metaplasias
Gastrointestinal pathology 2018 lecture 4. Dr Heyam Awad FRCPath
 Gastrointestinal pathology 2018 lecture 4 Dr Heyam Awad FRCPath Topics to be covered Peptic ulcer disease Hiatal hernia Gastric neoplasms Peptic ulcer disease (PUD)= chronic gastric ulcer Causes H pylori
Gastrointestinal pathology 2018 lecture 4 Dr Heyam Awad FRCPath Topics to be covered Peptic ulcer disease Hiatal hernia Gastric neoplasms Peptic ulcer disease (PUD)= chronic gastric ulcer Causes H pylori
Synergistic Inhibitory Effects of Gastrin and Histamine Receptor Antagonists on Helicobacter-Induced Gastric Cancer
 GASTROENTEROLOGY 2005;128:1965 1983 Synergistic Inhibitory Effects of Gastrin and Histamine Receptor Antagonists on Helicobacter-Induced Gastric Cancer SHIGEO TAKAISHI,* GUANGLIN CUI, DANA M. FREDERICK,
GASTROENTEROLOGY 2005;128:1965 1983 Synergistic Inhibitory Effects of Gastrin and Histamine Receptor Antagonists on Helicobacter-Induced Gastric Cancer SHIGEO TAKAISHI,* GUANGLIN CUI, DANA M. FREDERICK,
3/28/2017. Disclosure of Relevant Financial Relationships. GU Evening Subspecialty Case Conference. Differential Diagnosis:
 GU Evening Subspecialty Case Conference Rajal B. Shah, M.D. VP, Medical Director, Urologic Pathology Miraca Life Sciences, Irving, Texas Clinical Associate Professor of Pathology Baylor College of Medicine,
GU Evening Subspecialty Case Conference Rajal B. Shah, M.D. VP, Medical Director, Urologic Pathology Miraca Life Sciences, Irving, Texas Clinical Associate Professor of Pathology Baylor College of Medicine,
Helicobacter pylori Improved Detection of Helicobacter pylori
 DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
Gut Microbiome Essentials
 CORE COMPONENTS I: Gut Microbiome Essentials 2016 Tom Fabian, PhD Module Outline 1. Microbiome overview: getting a sense of the microbiome, research, what we know 2. Bacteria: features, functions, communities
CORE COMPONENTS I: Gut Microbiome Essentials 2016 Tom Fabian, PhD Module Outline 1. Microbiome overview: getting a sense of the microbiome, research, what we know 2. Bacteria: features, functions, communities
DOCTORAL THESIS SUMMARY
 UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA DOCTORAL THESIS HISTOPATHOLOGICAL AND IMMUNOHISTOCHEMICAL STUDY OF GASTRIC CARCINOMAS SUMMARY Scientific Coordinator: Univ. Prof. Dr. SIMIONESCU CRISTIANA EUGENIA
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA DOCTORAL THESIS HISTOPATHOLOGICAL AND IMMUNOHISTOCHEMICAL STUDY OF GASTRIC CARCINOMAS SUMMARY Scientific Coordinator: Univ. Prof. Dr. SIMIONESCU CRISTIANA EUGENIA
17 th International Conference of the Inflammation Research Association: Wednesday AM (Day 4) September 9-13, 2012
 17 th International Conference of the Inflammation Research Association: Wednesday AM (Day 4) September 9-13, 2012 The Sagamore Resort, Bolton Landing, NY, USA The following report includes highlights
17 th International Conference of the Inflammation Research Association: Wednesday AM (Day 4) September 9-13, 2012 The Sagamore Resort, Bolton Landing, NY, USA The following report includes highlights
Expression of the GLUT1 and p53 Protein in Atypical Mucosal Lesions Obtained from Gastric Biopsy Specimens
 The Korean Journal of Pathology 2006; 40: 32-8 Expression of the GLUT1 and p53 Protein in Atypical Mucosal Lesions Obtained from Gastric Biopsy Specimens In Gu Do Youn Wha Kim Yong-Koo Park Department
The Korean Journal of Pathology 2006; 40: 32-8 Expression of the GLUT1 and p53 Protein in Atypical Mucosal Lesions Obtained from Gastric Biopsy Specimens In Gu Do Youn Wha Kim Yong-Koo Park Department
The Pathologist s Role in the Diagnosis and Management of Neoplasia in Barrett s Oesophagus Cian Muldoon, St. James s Hospital, Dublin
 The Pathologist s Role in the Diagnosis and Management of Neoplasia in Barrett s Oesophagus Cian Muldoon, St. James s Hospital, Dublin 24.06.15 Norman Barrett Smiles [A brief digression - Chair becoming
The Pathologist s Role in the Diagnosis and Management of Neoplasia in Barrett s Oesophagus Cian Muldoon, St. James s Hospital, Dublin 24.06.15 Norman Barrett Smiles [A brief digression - Chair becoming
Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy
 Volume 2012, Article ID 368160, 4 pages doi:10.1155/2012/368160 Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy Kazuyoshi Yagi,
Volume 2012, Article ID 368160, 4 pages doi:10.1155/2012/368160 Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy Kazuyoshi Yagi,
Differential responses of human colonic ILCs to gut commensal bacteria altered during HIV infection
 Differential responses of human colonic ILCs to gut commensal bacteria altered during HIV infection Moriah J. Castleman, Ph.D. Laboratory of Dr. Cara Wilson University of Colorado Anschutz Medical Campus
Differential responses of human colonic ILCs to gut commensal bacteria altered during HIV infection Moriah J. Castleman, Ph.D. Laboratory of Dr. Cara Wilson University of Colorado Anschutz Medical Campus
Microbiome, Inflammation and Cancer
 Microbiome, Inflammation and Cancer Anita Afzali MD, MPH, FACG Medical Director, OSU Inflammatory Bowel Disease Center Abercrombie & Fitch Endowed Chair in Inflammatory Bowel Disease 4 th Annual Cancer
Microbiome, Inflammation and Cancer Anita Afzali MD, MPH, FACG Medical Director, OSU Inflammatory Bowel Disease Center Abercrombie & Fitch Endowed Chair in Inflammatory Bowel Disease 4 th Annual Cancer
Necrotizing Enterocolitis: The Role of the Immune System
 Necrotizing Enterocolitis: The Role of the Immune System Patricia Denning, M.D. Associate Professor in Pediatrics Division of Neonatology Emory University School of Medicine What is NEC? What is NEC? Necrotizing
Necrotizing Enterocolitis: The Role of the Immune System Patricia Denning, M.D. Associate Professor in Pediatrics Division of Neonatology Emory University School of Medicine What is NEC? What is NEC? Necrotizing
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology. Robin Lorenz
 Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology Robin Lorenz rlorenz@uab.edu Why do we Need to Understand How the Mucosal Immune System Works? The mucosa is the major
Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology Robin Lorenz rlorenz@uab.edu Why do we Need to Understand How the Mucosal Immune System Works? The mucosa is the major
Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
Original Article. Abstract
 Original Article Association of helicobacter pylori with carcinoma of stomach Muhammad Arif, Serajuddaula Syed Department of Pathology, Sindh Medical College, Karachi Abstract Objective: To note the association
Original Article Association of helicobacter pylori with carcinoma of stomach Muhammad Arif, Serajuddaula Syed Department of Pathology, Sindh Medical College, Karachi Abstract Objective: To note the association
Mody. AIS vs. Invasive Adenocarcinoma of the Cervix
 Common Problems in Gynecologic Pathology Michael T. Deavers, M.D. Houston Methodist Hospital, Houston, Texas Common Problems in Gynecologic Pathology Adenocarcinoma in-situ (AIS) of the Cervix vs. Invasive
Common Problems in Gynecologic Pathology Michael T. Deavers, M.D. Houston Methodist Hospital, Houston, Texas Common Problems in Gynecologic Pathology Adenocarcinoma in-situ (AIS) of the Cervix vs. Invasive
The role of gut microbiome in IBS
 The role of gut microbiome in IBS Chung Owyang, MD H. Marvin Pollard Professor of Internal Medicine Professor of Molecular and Integrative Physiology Chief, Division of Gastroenterology Director, Pollard
The role of gut microbiome in IBS Chung Owyang, MD H. Marvin Pollard Professor of Internal Medicine Professor of Molecular and Integrative Physiology Chief, Division of Gastroenterology Director, Pollard
DIGESTIVE TRACT ESOPHAGUS
 DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
DIGESTIVE TRACT From the lower esophagus to the lower rectum four fundamental layers comprise the wall of the digestive tube: mucosa, submucosa, muscularis propria (externa), and adventitia or serosa (see
Role of Th17 cells in the immunopathogenesis of dry eye disease
 Role of Th17 cells in the immunopathogenesis of dry eye disease The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Chauhan,
Role of Th17 cells in the immunopathogenesis of dry eye disease The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Chauhan,
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Common Inflammatory Gastrointestinal Disorders: Endoscopic and Pathologic Correlations
 Common Inflammatory Gastrointestinal Disorders: Endoscopic and Pathologic Correlations Nicole C. Panarelli, M.D. Attending Pathologist Montefiore Medical Center Associate Professor of Pathology - Albert
Common Inflammatory Gastrointestinal Disorders: Endoscopic and Pathologic Correlations Nicole C. Panarelli, M.D. Attending Pathologist Montefiore Medical Center Associate Professor of Pathology - Albert
Supplementary information CD4 T cells are required for both development and maintenance of disease in a new model of reversible colitis
 Supplementary information CD4 T cells are required for both development and maintenance of disease in a new model of reversible colitis rasseit and Steiner et al. .. Supplementary Figure 1 % of initial
Supplementary information CD4 T cells are required for both development and maintenance of disease in a new model of reversible colitis rasseit and Steiner et al. .. Supplementary Figure 1 % of initial
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
Helicobacter felis Eradication Restores Normal Architecture and Inhibits Gastric Cancer Progression in C57BL/6 Mice
 GASTROENTEROLOGY 2005;128:1937 1952 Helicobacter felis Eradication Restores Normal Architecture and Inhibits Gastric Cancer Progression in C57BL/6 Mice XUN CAI,* JANE CARLSON,* CALIN STOICOV,* HANCHEN
GASTROENTEROLOGY 2005;128:1937 1952 Helicobacter felis Eradication Restores Normal Architecture and Inhibits Gastric Cancer Progression in C57BL/6 Mice XUN CAI,* JANE CARLSON,* CALIN STOICOV,* HANCHEN
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
p53 expression in invasive pancreatic adenocarcinoma and precursor lesions
 Malaysian J Pathol 2011; 33(2) : 89 94 ORIGINAL ARTICLE p53 expression in invasive pancreatic adenocarcinoma and precursor lesions NORFADZILAH MY MBBCH,* Jayalakshmi PAILOOR MPath, FRCPath,* RETNESWARI
Malaysian J Pathol 2011; 33(2) : 89 94 ORIGINAL ARTICLE p53 expression in invasive pancreatic adenocarcinoma and precursor lesions NORFADZILAH MY MBBCH,* Jayalakshmi PAILOOR MPath, FRCPath,* RETNESWARI
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Studies on probiotics effects on innate immune functions in the gastrointestinal tract of broiler chicks (SUMMARY)
 Doctoral Thesis Studies on probiotics effects on innate immune functions in the gastrointestinal tract of broiler chicks (SUMMARY) ELSAYED SEDDEK IBRAHEM MOHAMMED Department of Bioresource Science Graduate
Doctoral Thesis Studies on probiotics effects on innate immune functions in the gastrointestinal tract of broiler chicks (SUMMARY) ELSAYED SEDDEK IBRAHEM MOHAMMED Department of Bioresource Science Graduate
Exosomes as a. Novel Therapeutic Approach to Gastrointestinal Diseases Rebecca Murray APRN, FNP, CDE
 Exosomes as a Novel Therapeutic Approach to Gastrointestinal Diseases Rebecca Murray APRN, FNP, CDE Endocrine Nurse Practitioner Institute for Hormonal Balance Orlando, FL Medical Director Ward-Murray
Exosomes as a Novel Therapeutic Approach to Gastrointestinal Diseases Rebecca Murray APRN, FNP, CDE Endocrine Nurse Practitioner Institute for Hormonal Balance Orlando, FL Medical Director Ward-Murray
Gastrointestinal Tract Cancer
 Gastrointestinal Tract Cancer Tumors of the Stomach Gastric adenocarcinoma Incidence and Epidemiology Incidence mortality rates USA High incidence: Japan, China, Chile, Ireland risk lower socioeconomic
Gastrointestinal Tract Cancer Tumors of the Stomach Gastric adenocarcinoma Incidence and Epidemiology Incidence mortality rates USA High incidence: Japan, China, Chile, Ireland risk lower socioeconomic
Sarkis K Mazmanian, California Institute of Technology
 Sarkis K Mazmanian, California Institute of Technology The human microbiota The human gut harbors 10 11-10 12 bacteria per gram colonic content (>10 14 total bacteria) Total bacteria outnumber human cells
Sarkis K Mazmanian, California Institute of Technology The human microbiota The human gut harbors 10 11-10 12 bacteria per gram colonic content (>10 14 total bacteria) Total bacteria outnumber human cells
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Oesophagus and Stomach update dysplasia and early cancer
 Oesophagus and Stomach update dysplasia and early cancer Dr Tim Bracey STR teaching 13/4/16 Please check pathkids.com for previous talks One of the biggest units in the country (100 major resections per
Oesophagus and Stomach update dysplasia and early cancer Dr Tim Bracey STR teaching 13/4/16 Please check pathkids.com for previous talks One of the biggest units in the country (100 major resections per
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Canberra, Australia). CD11c-DTR-OVA-GFP (B6.CD11c-OVA), B6.luc + and. Cancer Research Center, Germany). B6 or BALB/c.FoxP3-DTR-GFP mice were
 Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Dysbiosis & Inflammation
 MASTERING THE MICROBIOME: Dysbiosis & Inflammation 2017 Tom Fabian, PhD It is reasonable to propose that the composition of the microbiome and its activities are involved in most, if not all, of the biological
MASTERING THE MICROBIOME: Dysbiosis & Inflammation 2017 Tom Fabian, PhD It is reasonable to propose that the composition of the microbiome and its activities are involved in most, if not all, of the biological
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces
 Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
Association of Helicobacter pylori infection with Atrophic gastritis in patients with Dyspepsia
 ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
Innate immune regulation of T-helper (Th) cell homeostasis in the intestine
 Innate immune regulation of T-helper (Th) cell homeostasis in the intestine Masayuki Fukata, MD, Ph.D. Research Scientist II Division of Gastroenterology, Department of Medicine, F. Widjaja Foundation,
Innate immune regulation of T-helper (Th) cell homeostasis in the intestine Masayuki Fukata, MD, Ph.D. Research Scientist II Division of Gastroenterology, Department of Medicine, F. Widjaja Foundation,
Accepted Manuscript. Correlation or Causation: Sex Hormones and Microscopic Colitis. Baldeep Pabla, Reid M. Ness
 Accepted Manuscript Correlation or Causation: Sex Hormones and Microscopic Colitis Baldeep Pabla, Reid M. Ness PII: S0016-5085(18)35216-8 DOI: https://doi.org/10.1053/j.gastro.2018.11.007 Reference: YGAST
Accepted Manuscript Correlation or Causation: Sex Hormones and Microscopic Colitis Baldeep Pabla, Reid M. Ness PII: S0016-5085(18)35216-8 DOI: https://doi.org/10.1053/j.gastro.2018.11.007 Reference: YGAST
CASE REPORT. Introduction. Case Report. Kimitoshi Kubo 1, Noriko Kimura 2, Katsuhiro Mabe 1, Yusuke Nishimura 1 and Mototsugu Kato 1
 doi: 10.2169/internalmedicine.0842-18 Intern Med 57: 2951-2955, 2018 http://internmed.jp CASE REPORT Synchronous Triple Gastric Cancer Incorporating Mixed Adenocarcinoma and Neuroendocrine Tumor Completely
doi: 10.2169/internalmedicine.0842-18 Intern Med 57: 2951-2955, 2018 http://internmed.jp CASE REPORT Synchronous Triple Gastric Cancer Incorporating Mixed Adenocarcinoma and Neuroendocrine Tumor Completely
ALLERGY AND AUTOIMMUNITY
 ALLERGY AND AUTOIMMUNITY Allergies and Autoimmunity Allergies and autoimmunity can be prevented It all starts in the gut It is more than preventing leaky gut IgE allergies do not necessarily involve leaky
ALLERGY AND AUTOIMMUNITY Allergies and Autoimmunity Allergies and autoimmunity can be prevented It all starts in the gut It is more than preventing leaky gut IgE allergies do not necessarily involve leaky
Helicobacter pylori is a Gram-negative bacterium that is present
 High Dietary Salt Intake Exacerbates Helicobacter pylori-induced Gastric Carcinogenesis Jennifer A. Gaddy, a Jana N. Radin, a John T. Loh, a Feng Zhang, a M. Kay Washington, b Richard M. Peek, Jr., c,d
High Dietary Salt Intake Exacerbates Helicobacter pylori-induced Gastric Carcinogenesis Jennifer A. Gaddy, a Jana N. Radin, a John T. Loh, a Feng Zhang, a M. Kay Washington, b Richard M. Peek, Jr., c,d
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Dietary Zinc Alters the Microbiota and Decreases Resistance to Clostridium difficile Infection
 1 Dietary Zinc Alters the Microbiota and Decreases Resistance to Clostridium difficile Infection 2 3 4 5 Joseph P. Zackular 1, Jessica L. Moore 2,3, Ashley T. Jordan 1, Lillian J. Juttukonda 1, Michael
1 Dietary Zinc Alters the Microbiota and Decreases Resistance to Clostridium difficile Infection 2 3 4 5 Joseph P. Zackular 1, Jessica L. Moore 2,3, Ashley T. Jordan 1, Lillian J. Juttukonda 1, Michael
Barrett s Esophagus. Abdul Sami Khan, M.D. Gastroenterologist Aurora Healthcare Burlington, Elkhorn, Lake Geneva, WI
 Barrett s Esophagus Abdul Sami Khan, M.D. Gastroenterologist Aurora Healthcare Burlington, Elkhorn, Lake Geneva, WI A 58 year-old, obese white man has had heartburn for more than 20 years. He read a magazine
Barrett s Esophagus Abdul Sami Khan, M.D. Gastroenterologist Aurora Healthcare Burlington, Elkhorn, Lake Geneva, WI A 58 year-old, obese white man has had heartburn for more than 20 years. He read a magazine
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
Histopathological Characteristics of Atrophic Gastritis in Adult Population
 Journal of Pharmacy and Pharmacology 3 (2015) 133-138 doi: 10.17265/2328-2150/2015.03.004 D DAVID PUBLISHING Histopathological Characteristics of Atrophic Gastritis in Adult Population Marija Milićević,
Journal of Pharmacy and Pharmacology 3 (2015) 133-138 doi: 10.17265/2328-2150/2015.03.004 D DAVID PUBLISHING Histopathological Characteristics of Atrophic Gastritis in Adult Population Marija Milićević,
Role of Tyk-2 in Th9 and Th17 cells in allergic asthma
 Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
Supplementary File Role of Tyk-2 in Th9 and Th17 cells in allergic asthma Caroline Übel 1*, Anna Graser 1*, Sonja Koch 1, Ralf J. Rieker 2, Hans A. Lehr 3, Mathias Müller 4 and Susetta Finotto 1** 1 Laboratory
CINtec p16 INK4a Staining Atlas
 CINtec p16 INK4a Staining Atlas Rating Rating Positive The rating positive will be assigned if the p16 INK4a -stained slide shows a continuous staining of cells of the basal and parabasal cell layers of
CINtec p16 INK4a Staining Atlas Rating Rating Positive The rating positive will be assigned if the p16 INK4a -stained slide shows a continuous staining of cells of the basal and parabasal cell layers of
Two Cases of Primary Gastric Lymphoma, Mucosa-Associated Lymphoid Tissue (MALT)-type
 Med. J. Kagoshima Univ., Vol. 47, Suppl. 2. 93-96, November, 1995 Case Report Two Cases of Primary Gastric Lymphoma, Mucosa-Associated Lymphoid Tissue (MALT)-type Mitsuharu NOMOTO1, Hiroshi SHIRAHAMA1,
Med. J. Kagoshima Univ., Vol. 47, Suppl. 2. 93-96, November, 1995 Case Report Two Cases of Primary Gastric Lymphoma, Mucosa-Associated Lymphoid Tissue (MALT)-type Mitsuharu NOMOTO1, Hiroshi SHIRAHAMA1,
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Defining the Helper T Cell Contribution to Helicobacter pylori Gastritis. Brian M. Gray
 Defining the Helper T Cell Contribution to Helicobacter pylori Gastritis by Brian M. Gray A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Microbiology
Defining the Helper T Cell Contribution to Helicobacter pylori Gastritis by Brian M. Gray A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Microbiology
IKKα Causes Chromatin Modification on Pro-Inflammatory Genes by Cigarette Smoke in Mouse Lung
 IKKα Causes Chromatin Modification on Pro-Inflammatory Genes by Cigarette Smoke in Mouse Lung Se-Ran Yang, Samantha Valvo, Hongwei Yao, Aruna Kode, Saravanan Rajendrasozhan, Indika Edirisinghe, Samuel
IKKα Causes Chromatin Modification on Pro-Inflammatory Genes by Cigarette Smoke in Mouse Lung Se-Ran Yang, Samantha Valvo, Hongwei Yao, Aruna Kode, Saravanan Rajendrasozhan, Indika Edirisinghe, Samuel
IMO-8400, a novel TLR7, TLR8 and TLR9 antagonist, psoriasis
 IMO-8400, a novel TLR7, TLR8 and TLR9 antagonist, inhibits disease development in mouse models of psoriasis Weiwen e Ja Jiang, Fu-Gang Zhu, Dong Yu, Ekambar R. Kandimalla, a a, Nicola La Monica, and Sudhir
IMO-8400, a novel TLR7, TLR8 and TLR9 antagonist, inhibits disease development in mouse models of psoriasis Weiwen e Ja Jiang, Fu-Gang Zhu, Dong Yu, Ekambar R. Kandimalla, a a, Nicola La Monica, and Sudhir
malignant polyp Daily Challenges in Digestive Endoscopy for Endoscopists and Endoscopy Nurses BSGIE Annual Meeting 18/09/2014 Mechelen
 Plan Incidental finding of a malignant polyp 1. What is a polyp malignant? 2. Role of the pathologist and the endoscopist 3. Quantitative and qualitative risk assessment 4. How to decide what to do? Hubert
Plan Incidental finding of a malignant polyp 1. What is a polyp malignant? 2. Role of the pathologist and the endoscopist 3. Quantitative and qualitative risk assessment 4. How to decide what to do? Hubert
Correlation between Gastric Mucosal Morphologic Patterns and Histopathological Severity of
 Hindawi Publishing Corporation BioMed Research International Volume 2015, Article ID 808505, 7 pages http://dx.doi.org/10.1155/2015/808505 Research Article Correlation between Gastric Mucosal Morphologic
Hindawi Publishing Corporation BioMed Research International Volume 2015, Article ID 808505, 7 pages http://dx.doi.org/10.1155/2015/808505 Research Article Correlation between Gastric Mucosal Morphologic
The Gut Microbiota: Evidence For Gut Microbes as Contributors to Weight Gain
 The Gut Microbiota: Evidence For Gut Microbes as Contributors to Weight Gain Michael T. Bailey, Ph.D. Center for Microbial Pathogenesis The Research Institute, Nationwide Children s Hospital Department
The Gut Microbiota: Evidence For Gut Microbes as Contributors to Weight Gain Michael T. Bailey, Ph.D. Center for Microbial Pathogenesis The Research Institute, Nationwide Children s Hospital Department
Introduction. Original articles. Nicolás Rocha, 1 Sandra Huertas, 2 Rosario Albis, 3 Diego Aponte, 4 Luis Carlos Sabbagh. 5
 Original articles Correlation of endoscopic and histological findings in diagnosis of gastrointestinal metaplasia in patients referred to the Clinica Colombia for upper endoscopies Nicolás Rocha, 1 Sandra
Original articles Correlation of endoscopic and histological findings in diagnosis of gastrointestinal metaplasia in patients referred to the Clinica Colombia for upper endoscopies Nicolás Rocha, 1 Sandra
Histological and immunological characteristics of colitis associated with anti-ctla 4 antibody therapy
 Histological and immunological characteristics of colitis associated with anti-ctla 4 antibody therapy M. Perdiki 2, G. Bamias 1, D. Pouloudi 2, H. Gogas 3, I. Delladetsima 2 1 Academic Dpt. of Gastroenterology,
Histological and immunological characteristics of colitis associated with anti-ctla 4 antibody therapy M. Perdiki 2, G. Bamias 1, D. Pouloudi 2, H. Gogas 3, I. Delladetsima 2 1 Academic Dpt. of Gastroenterology,
Endoscopic Corner CASE 1. Sirimontaporn N Klaikaew N Imraporn B Rerknimitr R
 Endoscopic Corner Sirimontaporn N, et al. THAI J GASTROENTEROL 2010 Vol. 11 No. 3 Sept. - Dec. 2010 171 Sirimontaporn N Klaikaew N Imraporn B Rerknimitr R CASE 1 A 47- year-old female presented to the
Endoscopic Corner Sirimontaporn N, et al. THAI J GASTROENTEROL 2010 Vol. 11 No. 3 Sept. - Dec. 2010 171 Sirimontaporn N Klaikaew N Imraporn B Rerknimitr R CASE 1 A 47- year-old female presented to the
Pair-fed % inkt cells 0.5. EtOH 0.0
 MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
Microbiome as Predictor of Benefit and Toxicity in Cancer Immunotherapy
 Microbiome as Predictor of Benefit and Toxicity in Cancer Immunotherapy Giuliana Magri, Ph.D Optimizing Immunotherapy New Approaches, Biomarkers, Sequences and Combinations PRBB Auditorium, Barcelona October
Microbiome as Predictor of Benefit and Toxicity in Cancer Immunotherapy Giuliana Magri, Ph.D Optimizing Immunotherapy New Approaches, Biomarkers, Sequences and Combinations PRBB Auditorium, Barcelona October
