Mechanistic Modeling of in vitro Assays to Improve in vitro/in vivo Extrapolation
|
|
|
- Sharyl Underwood
- 5 years ago
- Views:
Transcription
1 Mechanistic Modeling of in vitro Assays to Improve in vitro/in vivo Extrapolation Grace Fraczkiewicz Viera Lukacova Jim Mullin 1
2 OVERVIEW MembranePlus a software platform for simulation of in vitro transport assays: Mechanisms Inputs Models Case Studies In vitro model validation In vitro/in vivo extrapolation 2
3 MECHANISMS: TRANSWELL 3
4 MECHANISMS: SANDWI C H HEPATOCYTES Collagen is assumed to not affect transport processes Model is also applicable for plated hepatocytes when bile volume is not considered 4
5 MECHANIMS: SUSPENDED HEPATOCYTES 5
6 I N P UTS: COMPOUND PROPERT I ES Physicochemical properties (in vitro or in silico) Enzyme and transporter settings applicable only to cell based assays 6
7 I N P UTS: EXPERIMENTA L S E T T I NGS Experimental Setup specific for selected Membrane type (cell monolayers, PAMPA, sandwich hepatocytes, suspended hepatocytes) Additional compound/assay specific settings 7
8 MODELS: MEMBRANE ENTRY/ E X I T R AT E STRUCTURE BASED MODEL Observed vs. Predicted on 44 training datasets V Intercept C1 logp C2 M _ RNG C3 HBDH HBD C4 HBAo log i All experimental time points to 2 hr Membrane entry and exit rates for anions and cations are determined based on logd vs. ph profile 8
9 MODELS: LYSOSOMAL TRAPPING Lipophilic Amines LogP > 1 pka > 6.5 Lysosome V i and V o correspond to the membrane/water entry and exit rate ph = 4-5 Lysosome membrane Kazmi F., Drug Metab. Disp. 41(3):897 (2013) Cell ph = 7.4 9
10 MODELS: ENZYMES AND TRANSPORT E RS Kinetics of carrier-mediated transport and metabolism is calculated using Michaelis-Menten kinetics: u metab = å i æ ç è i V max i K m intracell c u ö intracell + c u ø efflux i i V K max i m c c intracell u intracell u influx i V max units: mm/s (mmol/l/s) K m units: mm (mmol/l) General units converter allows converting these into different types of units Transporter types: Influx and Efflux Transporter locations: Apical and Basolateral (where applicable) i V max i Km c c buffer u buffer u 10
11 Case Studies: in vitro Model Validation 11
12 Case Study 1: Sodium Taurocholate Uptake into Bile 12
13 S ODIUM TAUROCHOLAT E Guo, ISSX 2014 OST alpha/beta 13
14 MEMBRANEPLUS MODEL ASSUMPTIONS Assumptions No protein binding for Na Taurocholate No stirring Complete monolayer of cells (100% viability) ADMET Predictor values for properties and transport model parameters calculated using calibration Literature values for K m were used as a starting point for building the model. K m values are similar across species Swift-Mol-Pharm (2) Transporter K m (mm) Cells Literature Source Cell Assay Inputs Feed Solution Conc. 1,2.5 mm BSA 4 % Well size 24 well Volume 0.3 ml Cell Vol pl Cell Layer Thickness 18.6 micron Cell Den 0.4 Mcell/well OST alpha/beta 25.8 Human Swift-Mol-Pharm (2) Overall Uptake 19 Rat Schwarz-Eur-J-Biochem NTCP 6 Human J-Exp-Biol BSEP 5 Rat J-Exp-Biol
15 S I MULAT I ON RES ULT Experimental K m values utilized from literature and V max values fit to data (all remaining properties were predicted by ADMET Predictor). Transporter Type K m (Exp) (mm) V max (mmol/s/l) BSEP Efflux E-03 OST alpha/beta Efflux E-02 NTCP Influx E-02 Efflux Phase 15
16 Case Study 2: Quantification of Influx Transport vs. Metabolism Statin Compounds Suspended Hepatocytes 16
17 Q UA NTIFY THE RELAT I VE IMPORTANCE OF INFLUX T R ANSPORT (OAT P 1 B1) VS. METABOLISM Paine, DMD (2008) 36: Media and whole cell concentration data for atorvastatin, cerivastatin, and indomethacin. Used a simple compartmental model to extract clearance values 17
18 MEMBRANEPLUS MODEL RES ULTS I Atorvastatin Cells Media Media Cells Cerivastatin Cells For this compound, lipid bilayer permeability model was fitted due to poor prediction from structure Cells Media Media 18
19 Metabolism Rate (ul/min/10 6 cells) MEMBRANEPLUS MODEL RES ULTS II Influx Rate (ul/min/10 6 cells) Metabolism Rate (ul/min/10 6 cells) Influx Rate (ul/min/10 6 cells) Paine, DMD (2008) 36: Influx Influx Metabolism Atorvastatin Results Metabolism Cerivastatin Results V max 3A4 (umol/s/l) V max 3A4 (umol/s/l) 3.74E-02 V max OATP1B1(umol/s/L) 31.9 V max OATP1B1(umol/s/L) Cl met (ul/min/mcell) 63 (68 Lit.) Cl met (ul/min/mcell) 20 (17 Lit.) Cl upt (ul/min/mcell) 471 (375 Lit.) Cl upt (ul/min/mcell) 704 (413 Lit.) 19
20 MODEL COMPARISON The mechanistic model in MembranePlus achieved similar result as the simpler compartmental model with fewer fitted parameters Compartmenal model* Membrane Plus Intracellular volume Fitted System parameter Cell membrane volume Fitted System parameter Membrane/water partitioning (kmem) Fitted Predicted from compound properties Active uptake Fitted Fitted Passive diffusion Fitted Predicted from compound properties (atorvastatin only) Metabolism Fitted Fitted *Paine, DMD (2008) 36:
21 Case Studies: in vitro to in vivo Extrapolation 21
22 Case Study 3: Metabolic IVIVE 22
23 P ROPAFENONE HUMAN HEPATOCYTE DATA Reports indicate it is a CYP2D6 substrate and has saturable dose dependent kinetics. In vitro data from literature was used to fit intracellular unbound Km and Vmax Komura, Drug metabolism and disposition 33.6 (2005):
24 F I T Km AND Vmax TO i n v i t r o DATA K m = mm V max = 9.27E-02 mmol/s/l cytosol (Converts to: 2.17E-02 nmol/min/10^6 cells) K m fitted in mechanistic model was lower than the one reported in paper which is indicative of K m based on unbound intracellular concentration 24
25 P R E DICT in vivo PK PBPK model: Kps predicted using default (Lukacova) method in GastroPlus CYP2D6 clearance extrapolated from fitted in vitro values (shown on previous slide) 70 mg IV bolus dose Data: Hollmann, Cardiac Arrhythmias, Springer
26 Case Study 4: Transporter IVIVE 26
27 D I GOX I N: FIT INTRACELLULAR Km i n v i t r o Published data on nonlinear Papp vs. donor concentration for Digoxin were used to fit Pgp Km (intracellular unbound) and Vmax Data from: Troutman and Thakker, Pharm. Res., Vol. 20, No. 8:
28 Papp [10^6 x cm/s] D I GOX I N: MODEL VERIFICAT I ON Km obtained from fitting to a published dataset was used to predict concentration-time profiles from another dataset Vmax was adjusted to account for different expression levels of Pgp in different systems Donor (Apical) Concentration [um] 28
29 D I GOX I N: PREDICT i n v i v o A B S ORPTION A B C A: Observed (symbols) vs. predicted plasma conc. (blue) and urinary excretion (red) of digoxin (Ochs, 1978). B: Observed (symbols) vs. predicted plasma conc. (blue) of digoxin for a PO formulation with 6.5 mm radius particle size (Jounela, 1975). C: Observed (symbols) vs. predicted plasma conc. (blue) of digoxin for a PO formulation with 51 mm radius particle size (Jounela, 1975). All simulations are using the fitted intracellular unbound P-gp Km value of 95.3 mm 29
30 Case Studies 5: Lysosomal Trapping and Absorption 30
31 LYSOSOMAL TRAPPING OF LIPOPHILIC CAT I ONS Drug Log P Basic pka T max (h) Lipophilic Amines LogP > 1 pka > Kazmi F., Drug Metab. Disp. 41(3):897 (2013) 31
32 LYSOSOMAL TRAPPING OF LIPOPHILIC CAT I ONS Hepatic perfusion experiment with and without H+ ionophore Monensin Empty symbols represent controls and solid symbols represent monensin treatment. Dashed and solid lines stand for fitted data in control and treatments, respectively. Drug Antipyrine Atenolol Propranolol Monensin Effect No Effect Minor Effect Strong Effect Siebert GA, JPET 308(1):228 (2004)) 32
33 LYSOSOMAL TRAPPING OF LIPOPHILIC CAT I ONS Caco-2 Apical-to-Basolateral permeability experiment Propranolol Propranolol + Bafilomycin Ibuprofen Testosterone Solid squares Apical compartment Solid triangles Basolateral compartment Empty squares Cell monolayer Lines represent fitted model results Heikkinen AT, JPET 328: 882 (2009) 33
34 CONSEQUENCES i n v i v o : D ESIPRAMI NE Desipramine physicochemical properties Samant T et al. CPT: PSP, 6(5): , High permeability and intestinal solubility Late Tmax Kurtz D.L. et al. CPT 1997, 62:
35 50 mg dose CONSEQUENCES i n v i v o : D ESIPRAMI NE Fu ent = 0.55% Fu ent = 100% 100 mg dose Samant T et al. CPT: PSP, 6(5): ,
36 50 mg dose CONSEQUENCES i n v i v o : D ESIPRAMI NE Fu ent = 0.55% Fu ent = 100% 100 mg dose Samant T et al. CPT: PSP, 6(5): ,
37 METHYLPHENIDAT E MEMBRANEPLUS SIMULAT I ON Lysosome S+LogP = 2.02 (AP 7.2) S+pKa = 8.56 (Base) (AP 7.2) Cytosol Cell Membrane Total cell Lysosome Membrane Apical Compartment Basolateral Compartment 37
38 METHYLPHENIDAT E MEMBRANEPLUS SIMULAT I ON S+LogP = 2.02 (AP 7.2) S+pKa = 8.56 (Base) (AP 7.2) Cytosol Fu = 4.37% Total cell 38
39 METHYLPHENIDAT E GASTROPLUS SIMULAT I ON Fu,ent = 100% Fu,ent = 4.37% 39
40 S UMMARY Mechanistic cellular simulations: Help to separate drug and system parameter inputs (similar to in vivo PBPK models) for easier translation between systems Are important to assess critical mechanisms affecting drug absorption, distribution and elimination pathways: Contributions of passive and active drug transport Interplay between drug metabolism and cellular uptake Disposition in bile Distribution within cellular structures (lipid bilayers, lysosomes) Facilitate extraction of variety of relevant parameters for more accurate IVIVE 40
41
42 Additional Slides 42
43 COMPOUND X - C L ASS II/IV Lipophilic (log P > 4) Moderate base (pka 3.2 and 6.2) Low (0.001 mg/ml), ph dependent solubility Moderate gut permeability (1.48 x 10-4 cm/s) Estimated bioavailability ~30% Are the different (fitted) precipitation and gastric emptying times under fasted & fed conditions masking something else in the model? Zhang et al. AAPS PharmSciTech 2014 January 17 43
44 COMPOUND X FA STED STAT E MODEL DEVELOPMENT Measured & ACAT Default Model Parameters Default precipitation Optimized precipitation Dissolved Absorbed Enterocyte Portal Vein Systemic Circulation Absorption through enterocyte to portal vein is too fast predicting Precipitation low can t Tmax account even for with delayed onset alone precipitation. Hence, need to optimize gastric emptying time 44
45 COMPOUND X : GASTROPLUS SIMULAT I ONS WITH MEMBRANEPLUS fu, E NTEROCYTE = 3.47% Optimized precipitation & simulated Fu, Enterocyte Dissolved Absorbed Enterocyte Portal Vein Systemic Circulation The lag between absorption into enterocyte and basolateral clearance into portal vein captures the extended Tmax No changes to default GI physiology required 45
46 COMPOUND X FOOD EFFEC T PREDICTIONS ACROSS D OSES simulated MembranePlus Fu, Enterocyte input Optimized precipitation from low dose/fasted state PK data >> predicted remaining 3 study arms Default ACAT fasted/fed physiology parameters 46
47 MEMBRANEPLUS Data Analysis Instant permeability output from molecular structure or experimental data. Unbound Intracellular concentrations (Membrane, cytosol, lysosome, etc.) In vitro K m and V max for enzymes and transporters Parameter sensitivity analysis (on logp, shaking rate, ph etc.) Transwell, sandwich hepatocyte, and suspended hepatocyte models Assay Prediction Permeability Concentrations in cellular structures (Membrane, cytosol, lysosome) 47
Modeling of Ac-ve Transport and Metabolism for in vitro Suspended and Sandwich Hepatocyte Assays U-lizing MembranePlus
 Modeling of Ac-ve Transport and Metabolism for in vitro Suspended and Sandwich Hepatocyte Assays U-lizing MembranePlus James Mullin Principal Scien1st Simula1ons Plus, Inc. Overview MembranePlus a so:ware
Modeling of Ac-ve Transport and Metabolism for in vitro Suspended and Sandwich Hepatocyte Assays U-lizing MembranePlus James Mullin Principal Scien1st Simula1ons Plus, Inc. Overview MembranePlus a so:ware
Stimulate your kinetic understanding Permeability Binding Metabolism Transporters
 Stimulate your kinetic understanding Permeability Binding Metabolism Transporters www.simulations-plus.com +1-661-723-7723 What is MembranePlus? MembranePlus is an advanced, yet easy-to-use, modeling and
Stimulate your kinetic understanding Permeability Binding Metabolism Transporters www.simulations-plus.com +1-661-723-7723 What is MembranePlus? MembranePlus is an advanced, yet easy-to-use, modeling and
HTPK: Conducting PK modeling and
 HTPK: Conducting PK modeling and simulations at high speed November 5, 2018 Robert Fraczkiewicz, David Miller, Marvin Waldman, Robert D. Clark Slide 1 Session Description and Objectives HTPK lightens the
HTPK: Conducting PK modeling and simulations at high speed November 5, 2018 Robert Fraczkiewicz, David Miller, Marvin Waldman, Robert D. Clark Slide 1 Session Description and Objectives HTPK lightens the
Pharmacokinetic Modeling & Simulation in Discovery and non-clinical Development
 Pharmacokinetic Modeling & Simulation in Discovery and non-clinical Development Where do we stand? Disclaimer I am not a bioinformatician, mathematician or biomedical engineer. I am a simple minded pharmacist,
Pharmacokinetic Modeling & Simulation in Discovery and non-clinical Development Where do we stand? Disclaimer I am not a bioinformatician, mathematician or biomedical engineer. I am a simple minded pharmacist,
Viera Lukacova Director, Simulation Sciences
 Use of In Silico Mechanistic Models to Support Interspecies Extrapolation of Oral Bioavailability and Formulation Optimization: Model Example Using GastroPlus Viera Lukacova Director, Simulation Sciences
Use of In Silico Mechanistic Models to Support Interspecies Extrapolation of Oral Bioavailability and Formulation Optimization: Model Example Using GastroPlus Viera Lukacova Director, Simulation Sciences
Metformin: Mechanistic Absorption Modeling and IVIVC Development
 Metformin: Mechanistic Absorption Modeling and IVIVC Development Maziar Kakhi *, Ph.D. FDA Silver Spring, MD 20993 Maziar.kakhi@fda.hhs.gov Viera Lukacova, Ph.D. Simulations Plus Lancaster, CA 93534 viera@simulations-plus.com
Metformin: Mechanistic Absorption Modeling and IVIVC Development Maziar Kakhi *, Ph.D. FDA Silver Spring, MD 20993 Maziar.kakhi@fda.hhs.gov Viera Lukacova, Ph.D. Simulations Plus Lancaster, CA 93534 viera@simulations-plus.com
Use of PBPK in simulating drug concentrations in pediatric populations: Case studies of Midazolam and Gabapentin
 Use of PBPK in simulating drug concentrations in pediatric populations: Case studies of Midazolam and Gabapentin WORKSHOP ON MODELLING IN PAEDIATRIC MEDICINES April 2008 Viera Lukacova, Ph.D. Walter Woltosz,
Use of PBPK in simulating drug concentrations in pediatric populations: Case studies of Midazolam and Gabapentin WORKSHOP ON MODELLING IN PAEDIATRIC MEDICINES April 2008 Viera Lukacova, Ph.D. Walter Woltosz,
Simulation of the Nonlinear PK of Gabapentin and Midazolam in. Adult and Pediatric Populations. Population Approach Group in Europe (PAGE)
 Simulation of the Nonlinear PK of Gabapentin and Midazolam in Adult and Pediatric Populations Population Approach Group in Europe (PAGE) Meeting June 2006 Brugge, Belgium Walter Woltosz, M.S., M.A.S. Viera
Simulation of the Nonlinear PK of Gabapentin and Midazolam in Adult and Pediatric Populations Population Approach Group in Europe (PAGE) Meeting June 2006 Brugge, Belgium Walter Woltosz, M.S., M.A.S. Viera
Simulation of Membrane and Cell Culture Permeability and Transport. Michael B. Bolger and Viera Lukacova Simulations Plus, Inc.
 Simulation of Membrane and Cell Culture Permeability and Transport Michael B. Bolger and Viera Lukacova Simulations Plus, Inc. utline Models of microscopic membrane kinetics Calculation of membrane entry
Simulation of Membrane and Cell Culture Permeability and Transport Michael B. Bolger and Viera Lukacova Simulations Plus, Inc. utline Models of microscopic membrane kinetics Calculation of membrane entry
PK-UK Challenges and benefits of using PBPK to evaluate an IVIVC for drugs with nonideal solubility and/or permeability. Bath, November 2014
 PK-UK 2014 Challenges and benefits of using PBPK to evaluate an IVIVC for drugs with nonideal solubility and/or permeability Bath, November 2014 Prof. Dr. Jennifer Dressman Dressman Bath 2014 Why IVIVC
PK-UK 2014 Challenges and benefits of using PBPK to evaluate an IVIVC for drugs with nonideal solubility and/or permeability Bath, November 2014 Prof. Dr. Jennifer Dressman Dressman Bath 2014 Why IVIVC
Assessing the role of hepatic uptake in drug clearance - Pharmacokinetic and experimental considerations
 Assessing the role of hepatic uptake in drug clearance - Pharmacokinetic and experimental considerations Peter Webborn ISSX Short course Toronto 2013 1 Defining the why, when and how of Transporter studies
Assessing the role of hepatic uptake in drug clearance - Pharmacokinetic and experimental considerations Peter Webborn ISSX Short course Toronto 2013 1 Defining the why, when and how of Transporter studies
Building innovative drug discovery alliances. Hepatic uptake and drug disposition o in vitro and in silico approaches
 Building innovative drug discovery alliances Hepatic uptake and drug disposition o in vitro and in silico approaches Dr Beth Williamson Evotec AG, 2017 Outline Importance of predicting clearance In vitro
Building innovative drug discovery alliances Hepatic uptake and drug disposition o in vitro and in silico approaches Dr Beth Williamson Evotec AG, 2017 Outline Importance of predicting clearance In vitro
The ADME properties of most drugs strongly depends on the ability of the drug to pass through membranes via simple diffusion.
 1 MEDCHEM 562 Kent Kunze Lecture 1 Physicochemical Properties and Drug Disposition The ADME properties of most drugs strongly depends on the ability of the drug to pass through membranes via simple diffusion.
1 MEDCHEM 562 Kent Kunze Lecture 1 Physicochemical Properties and Drug Disposition The ADME properties of most drugs strongly depends on the ability of the drug to pass through membranes via simple diffusion.
Advances in Prediction of Food Effects
 Advances in Prediction of Food Effects John Crison APS Biopharmaceutics Focus Group MSD Innovation Centre, Hoddesdon, UK June 9, 2011 Outline Introduction/Theory Physiological and Physical Chemical Parameters
Advances in Prediction of Food Effects John Crison APS Biopharmaceutics Focus Group MSD Innovation Centre, Hoddesdon, UK June 9, 2011 Outline Introduction/Theory Physiological and Physical Chemical Parameters
BASIC PHARMACOKINETICS
 BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
Detail features... 1
 PBPK Modelling and its Applications to Predict Transporter-Mediated Drug-Drug Interactions Masoud Jamei Senior Scientific Advisor, Head of M&S M.Jamei@Simcyp.com NEDMDG 14 th June 211, New England, USA
PBPK Modelling and its Applications to Predict Transporter-Mediated Drug-Drug Interactions Masoud Jamei Senior Scientific Advisor, Head of M&S M.Jamei@Simcyp.com NEDMDG 14 th June 211, New England, USA
Cryo Characterization Report (CCR)
 Human Cryopreserved Hepatocytes Lot number: HUM4061B Date: October 19, 2014 Cryo Characterization Report (CCR) Lot Overview Qualification Catalog Number Quantity Cryopreserved human hepatocytes, Qualyst
Human Cryopreserved Hepatocytes Lot number: HUM4061B Date: October 19, 2014 Cryo Characterization Report (CCR) Lot Overview Qualification Catalog Number Quantity Cryopreserved human hepatocytes, Qualyst
Supplemental Information
 Supplemental Information Article Title:The Proton Pump Inhibitor, Omeprazole, but not Lansoprazole or Pantoprazole, is a Metabolism-Dependent Inhibitor of CYP2C19: Implications for Co-Administration with
Supplemental Information Article Title:The Proton Pump Inhibitor, Omeprazole, but not Lansoprazole or Pantoprazole, is a Metabolism-Dependent Inhibitor of CYP2C19: Implications for Co-Administration with
Early BioPharm Risk Assessment in Discovery. Linette Ruston PCF9 17 th September 2010, Barcelona
 Early BioPharm Risk Assessment in Discovery Linette Ruston PCF9 17 th September 2010, Barcelona Outline Background Why? How? cmad and early absorption simulation Case Studies & Examples Summary 2 Poor
Early BioPharm Risk Assessment in Discovery Linette Ruston PCF9 17 th September 2010, Barcelona Outline Background Why? How? cmad and early absorption simulation Case Studies & Examples Summary 2 Poor
USING PBPK MODELING TO SIMULATE THE DISPOSITION OF CANAGLIFLOZIN
 USING PBPK MODELING TO SIMULATE THE DISPOSITION OF CANAGLIFLOZIN Christophe Tistaert PDMS Pharmaceutical Sciences Preformulation & Biopharmaceutics AAPS 2015, FLORIDA (USA) Canagliflozin An orally active
USING PBPK MODELING TO SIMULATE THE DISPOSITION OF CANAGLIFLOZIN Christophe Tistaert PDMS Pharmaceutical Sciences Preformulation & Biopharmaceutics AAPS 2015, FLORIDA (USA) Canagliflozin An orally active
Development of Canagliflozin: Mechanistic Absorption Modeling During Late-Stage Formulation and Process Optimization
 Development of Canagliflozin: Mechanistic Absorption Modeling During Late-Stage Formulation and Process Optimization Nico Holmstock Scientist, Janssen R&D M CERSI 2017, BALTIMORE (USA) Canagliflozin An
Development of Canagliflozin: Mechanistic Absorption Modeling During Late-Stage Formulation and Process Optimization Nico Holmstock Scientist, Janssen R&D M CERSI 2017, BALTIMORE (USA) Canagliflozin An
Define the terms biopharmaceutics and bioavailability.
 Pharmaceutics Reading Notes Define the terms biopharmaceutics and bioavailability. Biopharmaceutics: the area of study concerning the relationship between the physical, chemical, and biological sciences
Pharmaceutics Reading Notes Define the terms biopharmaceutics and bioavailability. Biopharmaceutics: the area of study concerning the relationship between the physical, chemical, and biological sciences
Altered GI absorption in special populations: An industry perspective
 Altered GI absorption in special populations: An industry perspective Cordula Stillhart and Neil J. Parrott F. Hoffmann La Roche Ltd, Basel (CH) UNGAP WG meeting, Leuven (B) 8 March 2018 Current challenges
Altered GI absorption in special populations: An industry perspective Cordula Stillhart and Neil J. Parrott F. Hoffmann La Roche Ltd, Basel (CH) UNGAP WG meeting, Leuven (B) 8 March 2018 Current challenges
Complexities of Hepatic Drug Transport: How Do We Sort It All Out?
 Complexities of Hepatic Drug Transport: How Do We Sort It All Out? Keith A. Hoffmaster Pfizer Research Technology Center Cambridge, MA NEDMDG 2005 Summer Symposium 06.08.2005 The Challenge Intestinal uptake
Complexities of Hepatic Drug Transport: How Do We Sort It All Out? Keith A. Hoffmaster Pfizer Research Technology Center Cambridge, MA NEDMDG 2005 Summer Symposium 06.08.2005 The Challenge Intestinal uptake
Lecture 1: Physicochemical Properties of Drugs and Drug Disposition
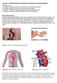 Lecture 1: Physicochemical Properties of Drugs and Drug Disposition Key objectives: 1. Be able to explain the benefits of oral versus IV drug administration 2. Be able to explain the factors involved in
Lecture 1: Physicochemical Properties of Drugs and Drug Disposition Key objectives: 1. Be able to explain the benefits of oral versus IV drug administration 2. Be able to explain the factors involved in
DMPK. APRIL 27 TH 2017 Jan Neelissen Scientific Adviser Science & Technology
 DMPK APRIL 27 TH 2017 Jan Neelissen Scientific Adviser Science & Technology What I learned is a good DMPK profile have acceptable water solubility for development be completely absorbed, preferably via
DMPK APRIL 27 TH 2017 Jan Neelissen Scientific Adviser Science & Technology What I learned is a good DMPK profile have acceptable water solubility for development be completely absorbed, preferably via
Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook with Computer Simulations
 Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook with Computer Simulations Rosenbaum, Sara E. ISBN-13: 9780470569061 Table of Contents 1 Introduction to Pharmacokinetics and Pharmacodynamics.
Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook with Computer Simulations Rosenbaum, Sara E. ISBN-13: 9780470569061 Table of Contents 1 Introduction to Pharmacokinetics and Pharmacodynamics.
Biopharmaceutics Classification System: Defining a Permeability Class
 Biopharmaceutics Classification System: Defining a Permeability Class Blair Miezeiewski, M.S. Senior Scientist, In Vitro Permeability Lab Definition of Bioequivalence The United States Food and Drug Administration
Biopharmaceutics Classification System: Defining a Permeability Class Blair Miezeiewski, M.S. Senior Scientist, In Vitro Permeability Lab Definition of Bioequivalence The United States Food and Drug Administration
EVALUATION OF DRUG-DRUG INTERACTION POTENTIAL BETWEEN SACUBITRIL/VALSARTAN (LCZ696) AND STATINS USING A PHYSIOLOGICALLY- BASED PHARMACOKINETIC MODEL
 Drug metabolism and Pharmacokinetics/PK Sciences EVALUATIN F DRUG-DRUG INTERACTIN PTENTIAL BETWEEN SACUBITRIL/VALSARTAN (LCZ696) AND STATINS USING A PHYSILGICALLY- BASED PHARMACKINETIC MDEL Imad Hanna,
Drug metabolism and Pharmacokinetics/PK Sciences EVALUATIN F DRUG-DRUG INTERACTIN PTENTIAL BETWEEN SACUBITRIL/VALSARTAN (LCZ696) AND STATINS USING A PHYSILGICALLY- BASED PHARMACKINETIC MDEL Imad Hanna,
BIOPHARMACEUTICS and CLINICAL PHARMACY
 11 years papers covered BIOPHARMACEUTICS and CLINICAL PHARMACY IV B.Pharm II Semester, Andhra University Topics: Absorption Distribution Protein binding Metabolism Excretion Bioavailability Drug Interactions
11 years papers covered BIOPHARMACEUTICS and CLINICAL PHARMACY IV B.Pharm II Semester, Andhra University Topics: Absorption Distribution Protein binding Metabolism Excretion Bioavailability Drug Interactions
Exploiting BDDCS and the Role of Transporters
 Exploiting BDDCS and the Role of Transporters (Therapeutic benefit of scientific knowledge of biological transporters, understanding the clinical relevant effects of active transport on oral drug absorption)
Exploiting BDDCS and the Role of Transporters (Therapeutic benefit of scientific knowledge of biological transporters, understanding the clinical relevant effects of active transport on oral drug absorption)
cationic molecule, paracellular diffusion would be thought of as its primary mode of transport across epithelial cells.
 ABSTRACT Metformin is the most widely prescribed anti-hyperglycemic agent for Type 2 Diabetes Mellitus (T2DM). Despite its frequent use, the intestinal absorption mechanism of this orally administered
ABSTRACT Metformin is the most widely prescribed anti-hyperglycemic agent for Type 2 Diabetes Mellitus (T2DM). Despite its frequent use, the intestinal absorption mechanism of this orally administered
Many drugs have both lipophilic and hydrophilic chemical substituents. Those drugs that are more lipid soluble tend to traverse cell membranes more
 Lecture-4 Many drugs have both lipophilic and hydrophilic chemical substituents. Those drugs that are more lipid soluble tend to traverse cell membranes more easily than less lipid-soluble or more water-soluble
Lecture-4 Many drugs have both lipophilic and hydrophilic chemical substituents. Those drugs that are more lipid soluble tend to traverse cell membranes more easily than less lipid-soluble or more water-soluble
Invited Review. Introduction
 BIOPHARMACEUTICS & DRUG DISPOSITION Biopharm. Drug Dispos. 34: 2 28 (2013) Published online 8 October 2012 in Wiley Online Library (wileyonlinelibrary.com).1810 Invited Review Absolute abundance and function
BIOPHARMACEUTICS & DRUG DISPOSITION Biopharm. Drug Dispos. 34: 2 28 (2013) Published online 8 October 2012 in Wiley Online Library (wileyonlinelibrary.com).1810 Invited Review Absolute abundance and function
Biopharmaceutics Drug Disposition Classification System (BDDCS) --- Its Impact and Application
 Biopharmaceutics Drug Disposition Classification System (BDDCS) --- Its Impact and Application Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences Schools of Pharmacy and Medicine
Biopharmaceutics Drug Disposition Classification System (BDDCS) --- Its Impact and Application Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences Schools of Pharmacy and Medicine
Muhammad Fawad Rasool Feras Khalil Stephanie Läer
 Clin Pharmacokinet (2015) 54:943 962 DOI 10.1007/s40262-015-0253-7 ORIGINAL RESEARCH ARTICLE A Physiologically Based Pharmacokinetic Drug Disease Model to Predict Carvedilol Exposure in Adult and Paediatric
Clin Pharmacokinet (2015) 54:943 962 DOI 10.1007/s40262-015-0253-7 ORIGINAL RESEARCH ARTICLE A Physiologically Based Pharmacokinetic Drug Disease Model to Predict Carvedilol Exposure in Adult and Paediatric
Pharmacokinetics Dr. Iman Lec. 3
 Pharmacokinetics r. Iman Lec. 3 Pharmacokinetics A dequate drug doses must be delivered to the target organ to get therapeutic but not toxic levels. So, pharmacokinetic examines the movement of drug over
Pharmacokinetics r. Iman Lec. 3 Pharmacokinetics A dequate drug doses must be delivered to the target organ to get therapeutic but not toxic levels. So, pharmacokinetic examines the movement of drug over
Pharmacokinetics of Drugs. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Pharmacokinetics of Drugs Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Absorption Is the transfer of a drug from its site of administration to the bloodstream.
Pharmacokinetics of Drugs Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Absorption Is the transfer of a drug from its site of administration to the bloodstream.
Aug 28 th, 2017 Pierre Daublain
 Analyzing the Potential Root Causes of Variability of Pharmacokinetics in Preclinical Species to Inform Derisking Strategies in Discovery and Early Development Aug 28 th, 2017 Pierre Daublain Outline Problem
Analyzing the Potential Root Causes of Variability of Pharmacokinetics in Preclinical Species to Inform Derisking Strategies in Discovery and Early Development Aug 28 th, 2017 Pierre Daublain Outline Problem
Advantages and Limitations of In Vivo Predictive Dissolution (IPD) Systems
 Advantages and Limitations of In Vivo Predictive Dissolution (IPD) Systems November 16 th Prof. Gregory E. Amidon - University of Michigan What is an In vivo Predictive Dissolution (IPD) System? An IPD
Advantages and Limitations of In Vivo Predictive Dissolution (IPD) Systems November 16 th Prof. Gregory E. Amidon - University of Michigan What is an In vivo Predictive Dissolution (IPD) System? An IPD
WHY... 8/21/2013 LEARNING OUTCOMES PHARMACOKINETICS I. A Absorption. D Distribution DEFINITION ADME AND THERAPEUIC ACTION
 PHARMACOKINETICS I Absorption & Distribution LEARNING OUTCOMES By the end of the lecture students will be able to.. Dr Ruwan Parakramawansha MBBS, MD, MRCP(UK),MRCPE, DMT(UK) (2013/08/21) Define pharmacokinetics,
PHARMACOKINETICS I Absorption & Distribution LEARNING OUTCOMES By the end of the lecture students will be able to.. Dr Ruwan Parakramawansha MBBS, MD, MRCP(UK),MRCPE, DMT(UK) (2013/08/21) Define pharmacokinetics,
Advantages and Limitations of In Vivo Predictive Dissolution (IPD) Systems
 Advantages and Limitations of In Vivo Predictive Dissolution (IPD) Systems November 16 th Prof. Gregory E. Amidon - University of Michigan What is an In vivo Predictive Dissolution (IPD) System? An IPD
Advantages and Limitations of In Vivo Predictive Dissolution (IPD) Systems November 16 th Prof. Gregory E. Amidon - University of Michigan What is an In vivo Predictive Dissolution (IPD) System? An IPD
Summary and general discussion
 Summary and general discussion Ingestion of contaminated soil can be an important route of exposure to soil-borne contaminants, especially for children (1). To estimate the health risk associated to this
Summary and general discussion Ingestion of contaminated soil can be an important route of exposure to soil-borne contaminants, especially for children (1). To estimate the health risk associated to this
Introduction to Pharmacokinetics (PK) Anson K. Abraham, Ph.D. Associate Principal Scientist, PPDM- QP2 Merck & Co. Inc., West Point, PA 5- June- 2017
 Introduction to Pharmacokinetics (PK) Anson K. Abraham, Ph.D. Associate Principal Scientist, PPDM- QP2 Merck & Co. Inc., West Point, PA 5- June- 2017 1 Outline Definition & Relevance of Pharmacokinetics
Introduction to Pharmacokinetics (PK) Anson K. Abraham, Ph.D. Associate Principal Scientist, PPDM- QP2 Merck & Co. Inc., West Point, PA 5- June- 2017 1 Outline Definition & Relevance of Pharmacokinetics
Determination of bioavailability
 Pharmaceutics 2 Bioavailability Bioavailability is the rate and extent to which an administered drug reaches the systemic circulation. For example, if 100 mg of a drug is administered orally and 70 mg
Pharmaceutics 2 Bioavailability Bioavailability is the rate and extent to which an administered drug reaches the systemic circulation. For example, if 100 mg of a drug is administered orally and 70 mg
Strategy on Drug Transporter Investigation Why, How, Which & When. Jasminder Sahi
 Strategy on Drug Transporter Investigation Why, How, Which & When Jasminder Sahi Intestine Drug Absorption PEPT1 OATPs MCTs AE2 Epithelial Cell MCTs MRP3 Liver Excretion via Liver Kidney MRPs OATPs N PT1
Strategy on Drug Transporter Investigation Why, How, Which & When Jasminder Sahi Intestine Drug Absorption PEPT1 OATPs MCTs AE2 Epithelial Cell MCTs MRP3 Liver Excretion via Liver Kidney MRPs OATPs N PT1
Understand the physiological determinants of extent and rate of absorption
 Absorption and Half-Life Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand Objectives Understand the physiological determinants of extent and rate of absorption
Absorption and Half-Life Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand Objectives Understand the physiological determinants of extent and rate of absorption
Biopharmaceutics Drug Disposition Classification System (BDDCS) and Its Application in Drug Discovery and Development
 Biopharmaceutics Drug Disposition Classification System (BDDCS) and Its Application in Drug Discovery and Development Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences University
Biopharmaceutics Drug Disposition Classification System (BDDCS) and Its Application in Drug Discovery and Development Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences University
The Influence of Physicochemical Properties on ADME
 The Influence of Physicochemical Properties on ADME Iain Martin Iain Martin; Physchem Forum 2 1 Physchem and ADME A quick tour of the influence of physicochemical properties on: Absorption Distribution
The Influence of Physicochemical Properties on ADME Iain Martin Iain Martin; Physchem Forum 2 1 Physchem and ADME A quick tour of the influence of physicochemical properties on: Absorption Distribution
Bioinformatics Master s Thesis Freie Universität Berlin FB Mathematik und Informatik. Max von Kleist
 A Generic Physiologically Based Pharmacokinetic Model to Predict Drug Distribution in Plasma, Red Blood Cells, Interstitial and Cellular Space with Application to the Pharmacokinetics of Tolbutamide Bioinformatics
A Generic Physiologically Based Pharmacokinetic Model to Predict Drug Distribution in Plasma, Red Blood Cells, Interstitial and Cellular Space with Application to the Pharmacokinetics of Tolbutamide Bioinformatics
DRUG METABOLISM AND PHARMACOKINETICS (DMPK) Lena Gustavsson, H. Lundbeck A/S, November 2015
 DRUG METABOLISM AND PHARMACOKINETICS (DMPK), H. Lundbeck A/S, LEGU@lundbeck.com November 2015 DMPK in Drug Discovery and Development Agenda Introduction Optimizing pharmacokinetic properties Absorption
DRUG METABOLISM AND PHARMACOKINETICS (DMPK), H. Lundbeck A/S, LEGU@lundbeck.com November 2015 DMPK in Drug Discovery and Development Agenda Introduction Optimizing pharmacokinetic properties Absorption
Basic Concepts in Pharmacokinetics. Leon Aarons Manchester Pharmacy School University of Manchester
 Basic Concepts in Pharmacokinetics Leon Aarons Manchester Pharmacy School University of Manchester Objectives 1. Define pharmacokinetics 2. Describe absorption 3. Describe distribution 4. Describe elimination
Basic Concepts in Pharmacokinetics Leon Aarons Manchester Pharmacy School University of Manchester Objectives 1. Define pharmacokinetics 2. Describe absorption 3. Describe distribution 4. Describe elimination
In vitro and in silico Predictions of Hepatic Transporter-Mediated Drug Clearance and Drug-Drug Interactions in vivo
 Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 193 In vitro and in silico Predictions of Hepatic Transporter-Mediated Drug Clearance and Drug-Drug Interactions in
Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 193 In vitro and in silico Predictions of Hepatic Transporter-Mediated Drug Clearance and Drug-Drug Interactions in
COMPARTMENTAL ANALYSIS OF DRUG DISTRIBUTION Juan J.L. Lertora, M.D., Ph.D. Director Clinical Pharmacology Program September 23, 2010
 COMPARTMENTAL ANALYSIS OF DRUG DISTRIBUTION Juan J.L. Lertora, M.D., Ph.D. Director Clinical Pharmacology Program September 23, 2010 Office of Clinical Research Training and Medical Education National
COMPARTMENTAL ANALYSIS OF DRUG DISTRIBUTION Juan J.L. Lertora, M.D., Ph.D. Director Clinical Pharmacology Program September 23, 2010 Office of Clinical Research Training and Medical Education National
Lippincott Questions Pharmacology
 Lippincott Questions Pharmacology Edition Two: Chapter One: 1.Which one of the following statements is CORRECT? A. Weak bases are absorbed efficiently across the epithelial cells of the stomach. B. Coadministration
Lippincott Questions Pharmacology Edition Two: Chapter One: 1.Which one of the following statements is CORRECT? A. Weak bases are absorbed efficiently across the epithelial cells of the stomach. B. Coadministration
1. Gastric Emptying Time Anatomically, a swallowed drug rapidly reaches the stomach. Eventually, the stomach empties its content in the small
 Lecture-5 1. Gastric Emptying Time Anatomically, a swallowed drug rapidly reaches the stomach. Eventually, the stomach empties its content in the small intestine. Because the duodenum has the greatest
Lecture-5 1. Gastric Emptying Time Anatomically, a swallowed drug rapidly reaches the stomach. Eventually, the stomach empties its content in the small intestine. Because the duodenum has the greatest
Biopharmaceutical aspects of intestinal drug absorption
 Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 257 Biopharmaceutical aspects of intestinal drug absorption Regional permeability and absorption-modifying excipients
Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 257 Biopharmaceutical aspects of intestinal drug absorption Regional permeability and absorption-modifying excipients
Leslie Z. Benet, PhD. Professor of Bioengineering and Therapeutic Sciences Schools of Pharmacy and Medicine University of California San Francisco
 Biopharmaceutics Drug Disposition Classification System (BDDCS) and Drug Interactions Leslie Z. Benet, PhD Professor of Bioengineering and Therapeutic Sciences Schools of Pharmacy and Medicine University
Biopharmaceutics Drug Disposition Classification System (BDDCS) and Drug Interactions Leslie Z. Benet, PhD Professor of Bioengineering and Therapeutic Sciences Schools of Pharmacy and Medicine University
Fundamentals of Pharmacology for Veterinary Technicians Chapter 4
 (A) (B) Figure 4-1 A, B (C) FIGURE 4-1C The active transport process moves particles against the concentration gradient from a region of low concentration to a region of high concentration. Active transport
(A) (B) Figure 4-1 A, B (C) FIGURE 4-1C The active transport process moves particles against the concentration gradient from a region of low concentration to a region of high concentration. Active transport
PBPK modeling of renal impairment what is missing?
 PBPK modeling of renal impairment what is missing? Aleksandra Galetin Centre for Applied Pharmacokinetic Research, University of Manchester, UK Outline of the presentation Physiological changes in renal
PBPK modeling of renal impairment what is missing? Aleksandra Galetin Centre for Applied Pharmacokinetic Research, University of Manchester, UK Outline of the presentation Physiological changes in renal
Supporting information
 Supporting information Intracellular drug bioavailability: a new predictor of system dependent drug disposition Mateus, André 1* ; Treyer, Andrea 1* ; Wegler, Christine 1,2 ; Karlgren, Maria 1 ; Matsson,
Supporting information Intracellular drug bioavailability: a new predictor of system dependent drug disposition Mateus, André 1* ; Treyer, Andrea 1* ; Wegler, Christine 1,2 ; Karlgren, Maria 1 ; Matsson,
Biopharmaceutics. Tips Worth Tweeting. Contributor: Sandra Earle
 Biopharmaceutics C H A P T E R 2 Contributor: Sandra Earle The physiochemical properties of drugs determine how they will move and interact with the body. By understanding a few principles, predictions
Biopharmaceutics C H A P T E R 2 Contributor: Sandra Earle The physiochemical properties of drugs determine how they will move and interact with the body. By understanding a few principles, predictions
General Principles of Pharmacology and Toxicology
 General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
Prediction of THC Plasma and Brain Concentrations following. Marijuana Administration: Approach and Challenges
 Prediction of THC Plasma and Brain Concentrations following Marijuana Administration: Approach and Challenges Contents: 1. Background and Significance 1.1. Introduction 1.2. THC - the primary chemical
Prediction of THC Plasma and Brain Concentrations following Marijuana Administration: Approach and Challenges Contents: 1. Background and Significance 1.1. Introduction 1.2. THC - the primary chemical
Under prediction of hepatic clearance from in vitro studies: prospects for resolution. J Brian Houston
 DDI, Seattle, June 2018 Under prediction of hepatic clearance from in vitro studies: prospects for resolution J Brian Houston Centre for Applied Pharmacokinetic Research (CAPkR) Scaling up in vitro parameters
DDI, Seattle, June 2018 Under prediction of hepatic clearance from in vitro studies: prospects for resolution J Brian Houston Centre for Applied Pharmacokinetic Research (CAPkR) Scaling up in vitro parameters
General Principles of Pharmacology and Toxicology
 General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
Use of Target Tissue Concentrations in PK-PD Modeling of Inhaled Drugs Ramon Hendrickx, AstraZeneca, RIA IMed Gothenburg, Sweden
 Use of Target Tissue Concentrations in PK-PD Modeling of Inhaled Drugs Ramon Hendrickx, AstraZeneca, RIA IMed Gothenburg, Sweden IQ DMPK Leadership Group 31 May 2017 What s On? Lung as target organ Benefit
Use of Target Tissue Concentrations in PK-PD Modeling of Inhaled Drugs Ramon Hendrickx, AstraZeneca, RIA IMed Gothenburg, Sweden IQ DMPK Leadership Group 31 May 2017 What s On? Lung as target organ Benefit
MODULE PHARMACOKINETICS WRITTEN SUMMARY
 MODULE 2.6.4. PHARMACOKINETICS WRITTEN SUMMARY m2.6.4. Pharmacokinetics Written Summary 2013N179518_00 TABLE OF CONTENTS PAGE 1. BRIEF SUMMARY...4 2. METHODS OF ANALYSIS...5 3. ABSORPTION...6 4. DISTRIBUTION...7
MODULE 2.6.4. PHARMACOKINETICS WRITTEN SUMMARY m2.6.4. Pharmacokinetics Written Summary 2013N179518_00 TABLE OF CONTENTS PAGE 1. BRIEF SUMMARY...4 2. METHODS OF ANALYSIS...5 3. ABSORPTION...6 4. DISTRIBUTION...7
ATP-Binding-Cassette Transporters in Biliary Efflux and Drug-Induced Liver Injury
 Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 172 ATP-Binding-Cassette Transporters in Biliary Efflux and Drug-Induced Liver Injury JENNY M. PEDERSEN ACTA UNIVERSITATIS
Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 172 ATP-Binding-Cassette Transporters in Biliary Efflux and Drug-Induced Liver Injury JENNY M. PEDERSEN ACTA UNIVERSITATIS
Predicting a Drug s Membrane Permeability: A Computational Model Validated With in Vitro Permeability Assay Data
 1 Current Literature Oct. 28, 2017 Ruyin Cao Wipf group Predicting a Drug s Membrane Permeability: A Computational Model Validated With in Vitro Permeability Assay Data Bennion BJ, Be NA, McNerney MW,
1 Current Literature Oct. 28, 2017 Ruyin Cao Wipf group Predicting a Drug s Membrane Permeability: A Computational Model Validated With in Vitro Permeability Assay Data Bennion BJ, Be NA, McNerney MW,
Biopharmaceutics. Lec: 4
 64 Biopharmaceutics Physicochemical Properties of Drugs Affecting Bioavailability Lec: 4 1 Assist. Lecturer Ali Yaseen Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School
64 Biopharmaceutics Physicochemical Properties of Drugs Affecting Bioavailability Lec: 4 1 Assist. Lecturer Ali Yaseen Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School
Nonlinear Pharmacokinetics
 Nonlinear Pharmacokinetics Non linear pharmacokinetics: In some cases, the kinetics of a pharmacokinetic process change from predominantly first order to predominantly zero order with increasing dose or
Nonlinear Pharmacokinetics Non linear pharmacokinetics: In some cases, the kinetics of a pharmacokinetic process change from predominantly first order to predominantly zero order with increasing dose or
Quantitative Evaluation of the Effect of P-Glycoprotein on Oral Drug Absorption
 Quantitative Evaluation of the Effect of P-Glycoprotein on Oral Drug Absorption -Assessment of Drug Permeability to Rat Small Intestine- College of Pharmacy, Setsunan University, Osaka, Japan Yoshiyuki
Quantitative Evaluation of the Effect of P-Glycoprotein on Oral Drug Absorption -Assessment of Drug Permeability to Rat Small Intestine- College of Pharmacy, Setsunan University, Osaka, Japan Yoshiyuki
Principles of Toxicokinetics/Toxicodynanics
 Biochemical and Molecular Toxicology ENVR 442; TOXC 442; BIOC 442 Principles of Toxicokinetics/Toxicodynanics Kim L.R. Brouwer, PharmD, PhD kbrouwer@unc.edu; 919-962-7030 Pharmacokinetics/ Toxicokinetics:
Biochemical and Molecular Toxicology ENVR 442; TOXC 442; BIOC 442 Principles of Toxicokinetics/Toxicodynanics Kim L.R. Brouwer, PharmD, PhD kbrouwer@unc.edu; 919-962-7030 Pharmacokinetics/ Toxicokinetics:
Culture Hepatocytes in Human Plasma to Count the free Concentration of Drug in Evaluation of Drug-drug Interaction. Chuang Lu
 Culture Hepatocytes in Human Plasma to Count the free Concentration of Drug in Evaluation of Drug-drug nteraction Chuang Lu Millennium, The Takeda Oncology Company Cambridge, MA, USA DD 205, Seattle, 6/29/205
Culture Hepatocytes in Human Plasma to Count the free Concentration of Drug in Evaluation of Drug-drug nteraction Chuang Lu Millennium, The Takeda Oncology Company Cambridge, MA, USA DD 205, Seattle, 6/29/205
PBPK Modelling of. Food Effects. PBPK SYMPOSIUM 2018 Paris, April 4 th David Turner (Presented by Dr Sebastian Polak)
 PBPK Modelling of Food Effects PBPK SYMPOSIUM 2018 Paris, April 4 th 2018 David Turner (Presented by Dr Sebastian Polak) david.turner@certara.com Outline Modelling of Food Effects with Population PBPK
PBPK Modelling of Food Effects PBPK SYMPOSIUM 2018 Paris, April 4 th 2018 David Turner (Presented by Dr Sebastian Polak) david.turner@certara.com Outline Modelling of Food Effects with Population PBPK
Recent Advances in Palm Tocotrienol Research: Bioavailability and Health Benefits Revisited
 Recent Advances in Palm Tocotrienol Research: Bioavailability and Health Benefits Revisited Paul W. Sylvester, Ph.D. Pfizer Endowed Professor of Pharmacology, College of Pharmacy, University of Louisiana
Recent Advances in Palm Tocotrienol Research: Bioavailability and Health Benefits Revisited Paul W. Sylvester, Ph.D. Pfizer Endowed Professor of Pharmacology, College of Pharmacy, University of Louisiana
Investigating Transporter-Mediated Drug-Drug Interactions Using a Physiologically Based Pharmacokinetic Model of Rosuvastatin
 Citation: CPT Pharmacometrics Syst. Pharmacol. (2017) 6, 228 238; VC 2017 ASCPT All rights reserved doi:10.1002/psp4.12168 ORIGINAL ARTICLE Investigating Transporter-Mediated Drug-Drug Interactions Using
Citation: CPT Pharmacometrics Syst. Pharmacol. (2017) 6, 228 238; VC 2017 ASCPT All rights reserved doi:10.1002/psp4.12168 ORIGINAL ARTICLE Investigating Transporter-Mediated Drug-Drug Interactions Using
Drug Distribution. Joseph K. Ritter, Ph.D., Assoc. Prof. Medical Sciences Building, Room
 Drug Distribution Joseph K. Ritter, Ph.D., Assoc. Prof. Medical Sciences Building, Room 531 jkritter@vcu.edu 828-1022 Department of Pharmacology and Toxicology Medical College of Virginia Campus Virginia
Drug Distribution Joseph K. Ritter, Ph.D., Assoc. Prof. Medical Sciences Building, Room 531 jkritter@vcu.edu 828-1022 Department of Pharmacology and Toxicology Medical College of Virginia Campus Virginia
Pharmacokinetics I. Dr. M.Mothilal Assistant professor
 Pharmacokinetics I Dr. M.Mothilal Assistant professor DRUG TRANSPORT For a drug to produce a therapeutic effect, it must reach to its target and it must accumulate at that site to reach to the minimum
Pharmacokinetics I Dr. M.Mothilal Assistant professor DRUG TRANSPORT For a drug to produce a therapeutic effect, it must reach to its target and it must accumulate at that site to reach to the minimum
Drug disposition classification systems: A comparative review of BDDCS, ECCS and ECCCS
 Novartis Drug disposition classification systems: A comparative review of BDDCS, ECCS and ECCCS Birk Poller, Gian Camenisch - Novartis SOLVO - Meet The Experts Transporter Conference April 26, 2018 Drug
Novartis Drug disposition classification systems: A comparative review of BDDCS, ECCS and ECCCS Birk Poller, Gian Camenisch - Novartis SOLVO - Meet The Experts Transporter Conference April 26, 2018 Drug
Physiologically Based Pharmacokinetic Modeling of Intestinal First-Pass Metabolism of CYP3A Substrates with High Intestinal Extraction S
 Supplemental material to this article can be found at: http://dmd.aspetjournals.org/content/suppl/2011/06/01/dmd.111.039248.dc1 0090-9556/11/3909-1633 1642$25.00 DRUG METABOLISM AND DISPOSITION Vol. 39,
Supplemental material to this article can be found at: http://dmd.aspetjournals.org/content/suppl/2011/06/01/dmd.111.039248.dc1 0090-9556/11/3909-1633 1642$25.00 DRUG METABOLISM AND DISPOSITION Vol. 39,
RSC, February Interplay between enzymes and. clearance and intracellular concentration of drugs. Centre for Applied Pharmacokinetic Research
 RSC, February 2014 Interplay between enzymes and transporters in defining hepatic drug clearance and intracellular concentration of drugs J Brian Houston Centre for Applied Pharmacokinetic Research (CAPkR)
RSC, February 2014 Interplay between enzymes and transporters in defining hepatic drug clearance and intracellular concentration of drugs J Brian Houston Centre for Applied Pharmacokinetic Research (CAPkR)
The Future of In Vitro Systems for the Assessment of Induction and Suppression of Enzymes and Transporters
 The Future of In Vitro Systems for the Assessment of Induction and Suppression of Enzymes and Transporters AAPS, San Diego November 6 th, 2014 Andrew Parkinson, XPD Consulting Lisa Almond, Simcyp-Certara
The Future of In Vitro Systems for the Assessment of Induction and Suppression of Enzymes and Transporters AAPS, San Diego November 6 th, 2014 Andrew Parkinson, XPD Consulting Lisa Almond, Simcyp-Certara
Mechanistic Studies and Modeling of Effects of Ingested Lipids on Oral Drug Absorption. A Thesis Presented. Selena Di Maio
 Mechanistic Studies and Modeling of Effects of Ingested Lipids on Oral Drug Absorption A Thesis Presented By Selena Di Maio to The Department of Chemical Engineering In partial fulfillment of the requirements
Mechanistic Studies and Modeling of Effects of Ingested Lipids on Oral Drug Absorption A Thesis Presented By Selena Di Maio to The Department of Chemical Engineering In partial fulfillment of the requirements
Physiologically-Based Simulation of Daclatasvir Pharmacokinetics With Antiretroviral Inducers and Inhibitors of Cytochrome P450 and Drug Transporters
 Physiologically-Based Simulation of Daclatasvir Pharmacokinetics With Antiretroviral Inducers and Inhibitors of Cytochrome P450 and Drug Transporters Qi Wang, Wenying Li, Ming Zheng, Timothy Eley, Frank
Physiologically-Based Simulation of Daclatasvir Pharmacokinetics With Antiretroviral Inducers and Inhibitors of Cytochrome P450 and Drug Transporters Qi Wang, Wenying Li, Ming Zheng, Timothy Eley, Frank
The extended clearance model and its use for the interpretation of hepatobiliary elimination data
 ADMET & DMPK 3(1) (2015) 1-14; doi: 10.5599/admet.3.1.144 Open Access : ISSN : 1848-7718 Review http://www.pub.iapchem.org/ojs/index.php/admet/index The extended clearance model and its use for the interpretation
ADMET & DMPK 3(1) (2015) 1-14; doi: 10.5599/admet.3.1.144 Open Access : ISSN : 1848-7718 Review http://www.pub.iapchem.org/ojs/index.php/admet/index The extended clearance model and its use for the interpretation
Enhancement of oral bioavailability of insulin in humans
 Neuroendocrinology Letters Volume 30 No. 1 2009 Enhancement of oral bioavailability of insulin in humans Adnan Badwan 1, Mayas Remawi 1, Nidal Qinna 1,4, Amani Elsayed 1,5, Tawfiq Arafat 2, Munther Melhim
Neuroendocrinology Letters Volume 30 No. 1 2009 Enhancement of oral bioavailability of insulin in humans Adnan Badwan 1, Mayas Remawi 1, Nidal Qinna 1,4, Amani Elsayed 1,5, Tawfiq Arafat 2, Munther Melhim
PHA Second Exam. Fall On my honor, I have neither given nor received unauthorized aid in doing this assignment.
 PHA 5127 Second Exam Fall 2011 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Put all answers on the bubble sheet TOTAL /200 pts 1 Question Set I (True or
PHA 5127 Second Exam Fall 2011 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Put all answers on the bubble sheet TOTAL /200 pts 1 Question Set I (True or
Drug Absorption and Bioavailability
 Drug Absorption and Bioavailability Juan J.L. Lertora, M.D., Ph.D. Director Clinical Pharmacology Program October 4, 2012 Office of Clinical Research Training and Medical Education National Institutes
Drug Absorption and Bioavailability Juan J.L. Lertora, M.D., Ph.D. Director Clinical Pharmacology Program October 4, 2012 Office of Clinical Research Training and Medical Education National Institutes
PHA Final Exam Fall 2006
 PHA 5127 Final Exam Fall 2006 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
PHA 5127 Final Exam Fall 2006 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
Chimica Farmaceutica. Pharmacokinetics and related topics
 Chimica Farmaceutica Pharmacokinetics and related topics INTRODUCTION In order to produce its intended effect, a drug must be present at an appropriate concentration in the fluid surrounding the effect
Chimica Farmaceutica Pharmacokinetics and related topics INTRODUCTION In order to produce its intended effect, a drug must be present at an appropriate concentration in the fluid surrounding the effect
Evaluation of Drug-Drug Interactions FDA Perspective
 Evaluation of Drug-Drug Interactions FDA Perspective Kellie Schoolar Reynolds, Pharm.D. Deputy Director Division of Clinical Pharmacology IV Office of Clinical Pharmacology Office of Translational Sciences
Evaluation of Drug-Drug Interactions FDA Perspective Kellie Schoolar Reynolds, Pharm.D. Deputy Director Division of Clinical Pharmacology IV Office of Clinical Pharmacology Office of Translational Sciences
Simultaneous Assessment of Uptake and Metabolism in Rat Hepatocytes: A. Comprehensive Mechanistic Model
 JPET Fast This Forward. article has not Published been copyedited on and December formatted. The 21, final 2011 version as may DOI:10.1124/jpet.111.187112 differ from this version. Simultaneous Assessment
JPET Fast This Forward. article has not Published been copyedited on and December formatted. The 21, final 2011 version as may DOI:10.1124/jpet.111.187112 differ from this version. Simultaneous Assessment
Assem Al Refaei. Sameer Emeish. Dr.Alia. Hodaifa Ababneh & Abdullah Shurafa
 8 Assem Al Refaei Sameer Emeish Hodaifa Ababneh & Abdullah Shurafa Dr.Alia Sheet Checklist Bioequivalence and Therapeutic equivalence. Factors Influencing Absorption. Revising Bioavailability. Factors
8 Assem Al Refaei Sameer Emeish Hodaifa Ababneh & Abdullah Shurafa Dr.Alia Sheet Checklist Bioequivalence and Therapeutic equivalence. Factors Influencing Absorption. Revising Bioavailability. Factors
Practical Application of PBPK in Neonates and Infants, Including Case Studies
 Practical Application of PBPK in Neonates and Infants, Including Case Studies Presented at the conference : Innovative Approaches to Pediatric Drug Development and Pediatric Medical Countermeasures: A
Practical Application of PBPK in Neonates and Infants, Including Case Studies Presented at the conference : Innovative Approaches to Pediatric Drug Development and Pediatric Medical Countermeasures: A
Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population
 Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population Nina Isoherranen Department of Pharmaceutics University of Washington Processes driving drug disposition are
Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population Nina Isoherranen Department of Pharmaceutics University of Washington Processes driving drug disposition are
Problem Set for Fundamentals 9 Oct 2013
 20.201 Problem Set for Fundamentals 9 ct 2013 Please prepare your answers in electronic format and submit the answers or on ctober 11. While this is not a graded problem set, you will get cr completing
20.201 Problem Set for Fundamentals 9 ct 2013 Please prepare your answers in electronic format and submit the answers or on ctober 11. While this is not a graded problem set, you will get cr completing
The use of Saliva instead of Plasma as a Surrogate in Drug Bioavailability and Bioequivalence Studies in Humans
 The use of Saliva instead of Plasma as a Surrogate in Drug Bioavailability and Bioequivalence Studies in Humans ا.د. ناصر محمد ياسر نمرادكيدك Prof. Nasir M. Idkaidek University of Petra Amman - Jordan
The use of Saliva instead of Plasma as a Surrogate in Drug Bioavailability and Bioequivalence Studies in Humans ا.د. ناصر محمد ياسر نمرادكيدك Prof. Nasir M. Idkaidek University of Petra Amman - Jordan
Pharmacokinetics in Drug Development. Edward P. Acosta, PharmD Professor & Director Division of Clinical Pharmacology Director, CCC PK/PD Core
 Pharmacokinetics in Drug Development Edward P. Acosta, PharmD Professor & Director Division of Clinical Pharmacology Director, CCC PK/PD Core Finding new drugs: A crap shoot Clinical Development Phase
Pharmacokinetics in Drug Development Edward P. Acosta, PharmD Professor & Director Division of Clinical Pharmacology Director, CCC PK/PD Core Finding new drugs: A crap shoot Clinical Development Phase
