Oral Intake of Glucosylceramide Improves Relatively Higher Level of Transepidermal Water Loss in Mice and Healthy Human Subjects
|
|
|
- Gwen Shelton
- 5 years ago
- Views:
Transcription
1 Journal of Health Science, 54(5) (2008) 559 Oral Intake of Glucosylceramide Improves Relatively Higher Level of Transepidermal Water Loss in Mice and Healthy Human Subjects Taro Uchiyama,,a Yusuke Nakano, a Osamu Ueda, a Hiroshi Mori, a Masaya Nakashima, a Akira Noda, a Chiaki Ishizaki, b and Masako Mizoguchi c a Functional Food Development Group, Functional Food Research & Development Center, Shiseido Co., Ltd. Research Center (Kanazawa-Hakkei), Fukuura, Kanazawa-ku, Yokohama, Kanagawa , Japan, b Shinjuku Minamiguchi Hifuka, Nishishinjuku, Shinjuku-ku, Tokyo , Japan and c St. Marianna University School of Medicine, Sugao, Miyamae-ku, Kawasaki, Kanagawa , Japan (Received June 13, 2008; Accepted June 23, 2008) We examined the effect of oral intake of pure glucosylceramide derived from konjac extract on skin barrier function evaluated by transepidermal water loss (TEWL) in hairless mice with sodium dodecyl sulfate (SDS)- induced skin roughness. The difference of TEWL between SDS-treated site and untreated sites in the pure glucosylceramide-fed group was significantly lower than that in control group on day 14 of ingestion. We investigated interleukin-1α (IL-1α) production in the hairless mouse skin, and it was significantly lower in the glucosylceramide-fed group than that of control animals. This reduced IL-1α production should contribute to improvement of skin barrier function. To investigate the effect of oral intake of glucosylceramide in human, we conducted a randomized double-blind placebo-controlled study including 100 healthy subjects whose TEWL in cheek was relatively high. As a result, cheek TEWL was significantly lower in the test product group as compared with the control group in weeks 8 and 12 of ingestion (p =0.023 and p =0.002respectively). Key words konjac extract, glucosylceramide, transepidermal water loss (TEWL), itch, interleukin-1α (IL-1α), randomized double-blind placebo-controlled study INTRODUCTION The stratum corneum of skin has a barrier function to protect the organism from exogenous factors and to prevent water loss. Decrease of barrier function is associated with diseases such as atopic dermatitis (AD), 1 4) which is characterized by skin dryness and intense itching. Impairment of skin barrier function may also be caused by sudden change of the weather 5, 6) or contact with chemical agents such as cleansers. 7, 8) Severe itching may reduce the quality of life (QOL) through induction of sleep disorders. Stratum corneum lipids are known to influence skin barrier function and mainly composed of ce- To whom correspondence should be addressed: Functional Food Development Group, Functional Food Research & Development Center, Shiseido Co., Ltd. Research Center (Kanazawa-Hakkei), Fukuura, Kanazawa-ku, Yokohama, Kanagawa , Japan. Tel.: ; Fax: ; taro.uchiyama@to.shiseido.co.jp ramide (40 60%). The other components of stratum corneum lipids include cholesterol, cholesterol ester, free fatty acids, etc. The ceramide content of stratum corneum lipids decreases with aging and is also decreased in patients with atopic dermatitis, 9) and it is known that a decrease of ceramide causes impairment of the barrier function. 10) Glucosylceramide, which is a glycoside of ceramide, is a major sphingo (glycol) lipid in plants such as soybean, corn, wheat, rice, 11) and Amorphophallus konjac (A. konjac). 12) Recently, it has been reported that skin barrier function in hairless mice is improved by intake of extracts of food materials containing glucosylceramide; the purity was > 6%. 13) It has also been reported that oral intake of glucosylceramide reduced transepidermal water loss (TEWL) 14) in normal adult 12) or in AD patients. 15, 16) A. konjac, arich source of glucocylceramide, has been eaten as food for a long time in Japan. Additionally, A. konjac is extremely safe with no allergenic effects. Then, we focused our attention
2 560 Vol. 54 (2008) on konjac extract, which was fractionated from A. konjac, andstudied its efficacy. In this study, we investigated the efficacy of glucosylceramide on improving skin barrier function using pure glucosylceramide derived from konjac extract and examined the action mechanism in animal models. Moreover, we confirmed the efficacy of konjac extract including glucosylceramide in human healthy volunteers whose TEWL was relatively high, in a randomised, double-blind placebocontrolled study. MATERIALS AND METHODS Animals Five-week-old male HR-1 mice were purchased from Hoshino Laboratory Animals. They were used for the experiment at the age of 6 7 weeks. The animals were individually accommodated in plastic cages placed within a barrier system where the temperature was kept at C, and lighting was on for 12 hr/day (7:00 19:00). The relative humidity was < 10% in the dry environment study to investigate interleukin-1α (IL-1α) release from skin and was 40 70% in the other studies. Theanimals were allowed free access to a solid diet (AIN-93G, Oriental Yeast Co., Ltd., Tokyo, Japan) and tap water (after ultraviolet and microfilter treatment). During the acclimation period, the relative humidity was kept at 40 70%. The present study was approved by the ethics committee of Shiseido Research Center (Yokohama, Japan) in accordance with the guideline of the National Institute of Health (Bethesda, MD, U.S.A.). Test Substance Konjac extracts (containing 12% glucosylceramide) and konjac-derived glucosylceramide (100% pure product) were supplied from Unitika Ltd. (Kyoto, Japan). Each test substance was dispersed in 1% suspension of tragacanth gum (Wako Pure Chemical Industries, Ltd., Osaka, Japan) to make 30 µg glucosylcelamide/0.5 ml suspension for the mice in vivo study determining changes of TEWL. In the studies of IL-1 level, 250 µg glucosylceramide/0.5 ml tragacanth gum was used. One percent of tragacanth gum suspension served as control substance. Determination of Effect of Test Substance on Sodium Dodecyl Sulfate (SDS)-induced Rough Skin of Mice Administration of Sample After 1 week of acclimation, the test substance or the control substance (0.5 ml) was orally administered via a stomach tube to each animal once daily (9:00 10:00) for 14 consecutive days. SDS Treatment Under inhalational anesthesia with sevoflurane (Ishimaru Pharmaceutical Co., Ltd., Osaka, Japan), an unwoven cloth (ca cm)containing 300 µlof10%aqueous solution of SDS (Wako Pure Chemical Industries, Ltd.) was placed in contact with the left lateral area of the mouse back for 5 min. After the cloth was removed, the left lateral area of the back was gently wiped with absorbent cotton pre-immersed in warm water. Measurement of Mice TEWL Mice (test product group and control group) were orally treated with the test substance or placebo for 14 days. From the 4th day of treatment, SDS was applied daily about 3 hr after the dose (13:00) as described above. One hour after the SDS treatment on the 4th day of SDS treatment, TEWL of each mouse was measured at the right and left lateral areas of the back under identical conditions for all animals, using a Tewameter R TM-300 (Courage and Khazaka Electronics, Cologe, Germany). TEWL was calculated using the equation shown below. In this equation, SDS-treated site is the left lateral area of the back, and SDS-untreated site is the right lateral area of the back. TEWL (g/m 2 hr) = TEWL of SDS-treated site (left) (g/m 2 hr) TEWL of SDS-untreated site (right) (g/m 2 hr) All procedures for measuring skin barrier function and disrupting the barrier were carried out under anesthesia. Measurement of Skin Cytokine Release After measurement of TEWL, each mouse was euthanized without pain. The lateral back skin was collected, and subcutaneous fat removed with a knife. Test samples were prepared according to the method of Wood et al. 17) Specifically, skin pieces were placed in wells of a flat-bottomed multi-well (24-well) plate for cell culture (Becton Dickinson Labware, Franklin Lakes, NJ, U.S.A.), cooled in an ice bath. Ice-cooled Dulbecco s modified Eagle s medium (DMEM), 2 ml, was added to each well, and the plate was incubated on ice for 15 min. The culture was harvested, and stored at 80 C until measurement. IL-1α as measured by Quantikine R mouse IL-1α/IL-1F1 enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, MN, U.S.A.). Tumor necrosis factorα (TNF-α) was measured by mouse TNF-α instant ELISA (Bender MedSystems, Burlingame, CA, U.S.A.). Interferon-γ (IFN-γ)was measured by
3 No mouse IFN-γ instant ELISA (Bender MedSystems). In addition, the intake period of glucosylceramide was investigated over 29 days. Determination of Effect of Test Substance on Cytokine Release by Skin from Mice Kept in a Dry Environment After 1 week of acclimation, the test substance or the control substance (0.5 ml) was orally administered via a stomach tube to each animal in the glucosylceramide group and the control group (n = 7or8ineach group) once daily (9:00 10:00) for 29 consecutive days. The relative humidity was kept < 10% in this study to make dry environment. After the 29-day treatment, each mouse was euthanized without pain. Cytokines were measured as described in Measurement of skin cytokine release. Human Study Design This studywascarried out by KSO Corporation Co., Ltd. (Tokyo, Japan). The study began in November 2006 and ended in March To minimize the influence of changes in air temperature, weather, etc. on assessment of skin condition, each subject was acclimated to the test laboratory for 40 min, where the temperature and relative humidity were controlled at 21 ± 2 C and 45%, respectively. The study was designed as a placebo-controlled randomized double-blind parallel group comparison. Candidates were healthy individuals who selfassessed their skin roughness and itching in skin. From 256 candidates, individuals having skin abnormalities were excluded; thus, 120 individuals having high TEWL in cheek were selected. These 120 individuals were randomly divided into two groups (60 individuals/group) in such a manner as to minimize the inter-group differences in TEWL, male-to-female ratio, and age. After this grouping, they were treated in double blind manner. At the start of the study (week 0), individuals found to have skin abnormalities and those who refused to participate in the study or had low TEWL in cheek were excluded; thus the remaining 100 individuals (49 men and 51 women, age years) were finally included in the study. Test beverage (containing konjac extract) or placebo beverage was ingested for a period of 12 weeks. The 12-week ingestion period was followed by a 4-week non-ingestion period. Each subject was instructed to visit the hospital immediately before the start of ingestion (week 0), and at 4, 8, and 12 weeks after the start of ingestion and at 4 weeks after the end of ingestion (completion of the study) for evaluation of the skin condition. This study was carried out in accordance with the Declaration of Helsinki and the Ethical Guidelines on Epidemiological Studies. Test Beverage Emulsifiedkonjacextract (containing 0.3% glucosylceramide) was purchased from Unitika Ltd. The test beverage was prepared with emulsified konjac extract to contained 1.8 mg glucosylceramide, sweetener, sour seasoning, and flavor in 340 ml water. The placebo beverage was prepared with the same amounts of sweetener, sour seasoning, and flavor as the test beverage, so that they would not differ from each other in taste. Evaluation of Human TEWL TEWL was evaluated at thecheek, forearm and back using an evaporimeter (Model EP-2, Servomed, Stockholm, Sweden). Measurement was continued for 120 seconds, and the most stable value was adopted as TEWLatagiven occasion of measurement for a given subject. The variation in this parameter from baseline ( )wascompared between the two groups. Evaluation of Itching Sensation Evaluation was made over the entire face, entire arm, and entire leg. The itching scale used in this study measures the severity of itching at a specific point in time, using a horizontal visual analogue scale (VAS). Possible scores range from 0 (no itching) to 10 (severe itching). The variation in the rating from the baseline ( ) wascompared between the two groups. Statistics Data were expressed as mean ± S.D. Student s t-test was used for inter-group comparison of TEWL, itch data, and IL-1α level. Dunnett s test was used for inter-group comparison of mouse TEWL. Analysis of covariance (ANCOVA) was performed on absolute TEWL. p < 0.05 was regarded as statistically significant. RESULTS Identification of Active Component of Konjac Extract To identify the active component of konjac extract, we determined the effect on TEWL of pure glucosylceramide, which is the candidate active component of konjac extract in mice. As shown in Fig. 1, TEWL in the 100% pure product (100% glucosylceramide) ingestion group was significantly (p = 0.002) lower than the control group whereas that, in the konjac extract ingestion group was lower than control group (p =0.084). Each TEWL was 15.9 ± 4.5 g/m 2 hr in the control
4 562 Vol. 54 (2008) Fig. 1. Effect of Administration of Glvcosylceramide Solution to Mice on TEWLof SDS-treated Skin Mice (test product group and control group: n = 10) were orally treated with test substance or placebo for 14 days. From day 4 of treatment, SDS was applied using the method described in Materials and Methods. One hour after SDS treatment on day 4 of SDS treatment, TEWL was measured under identical conditions, using a Tewameter. Columns indicate values of TEWL in the control group, the groups given konjac extract containing 12% glucosylceramide (glucosylceramide: 30 µg/day), and the groups given 100% pure product (=glucosylceramide) (glucosylceramide: 30 µg/day), as indicated. Dunnett s test was used for comparison between three groups. Data are presented as mean ± S.D. Asterisks indicate a statistically significant difference, p < group, 12.2 ± 3.4 g/m 2 cdothr in the konjac extract ingestion group, and 9.6 ± 3.7 g/m 2 hr in the pure glucosylceramide ingestion group. Hence konjacderived pure glucosylceramide was stronger than konjac extract in inhibitory effect of TEWL, suggesting that the elevation of TEWL associated with SDS-induced skin roughness was not suppressed by any other component except glucosylceramide contained in the ingested konjac extract. Effect of Konjac Extract on Cytokine Production in Skin Roughness Mice It has been reported that epidermal IL-1α in mice bred under dry conditions [low relative humidity (< 10%) environment] is higher than in those kept under normal conditions of humidity. 18) Therefore the hypothesis that improvement of skin barrier function by konjac extract involves modulation of the IL-1α level in skin was tested by examining the effect of glucosylceramide on release of IL- 1α in the skin of mice bred under dry conditions. Furthermore, effects of glucosylceramide on IL-1α production in SDS skin roughness mouse were investigated. As shown in Fig. 2(A), the amount of IL-1α released in the skin was 148 ± 27 pg/ml cm 2 in the control group and 98 ± 25 pg/ml cm 2 in the glucosylceramide treatment group (p = 0.003), in- Fig. 2. Effects of Glucosylceramide on IL-1α Production in Mice Skin Roughness (A) Mice (test product group and control group: n = 7 8) were fed with glucosylceramide: 250 µg/day (konjac-derived glucosylceramide) under dry conditions (relative humidity < 10%) for 29 days. The skin was then collected from each mouse and the level of IL-1α measured. (B) Mice (test product group and control group: n = 4 5) were orally treated with glucosylceramide: 250 µg/day (konjac extracts) or the placebo for 14 days. Mice were treated with SDS for 4 days as described in Materials and Methods. One hour after SDS treatment on day 4 of SDS treatment, the skin was collected from each mouse and the level of IL-1α measured. indicates the control group and indicates the glucosylceramide ingestion group. Student s t-test was used for comparison between two groups. Data are presented as mean ± S.D. Asterisks indicate a statistically significant difference, p < 0.05, p < dicating that release of IL-1α in skin under dry conditions was significantly suppressed by ingestion of konjac-derived glucosylceramide. As shown in Fig. 2(B), the amount of IL-1α released in the skin was 173 ± 49 pg/ml cm 2 in the control group and 108 ± 30 pg/ml cm 2 in the glucosylceramide treatment group (p = 0.043),indicating that release of IL-1α in skin treated with SDS was significantly suppressed by ingestion of konjac-derived glucosylceramide. These results suggest that glucosylceramide improves the skin barrier function through suppressing the formation of IL-1α. In addition, TNF-α and IFN-γ levels of both skin roughness conditions were below the determination limit.
5 No Fig. 3. Flow Chart for Selection of Subjects Table 1. Inclusion Criteria 1. Men and women aged 20 years 2. Individuals conscious of skin roughness by drying, and whose symptoms of itching are found in winter 3. Individuals who experience rough symptoms on the skin after bathing and washing face and arms 4. Individuals whose cheek TEWL at the time of measurement was > 27 (the targeted value) at 0 weeks 5. Individuals whose skin conductance (40 55 as the targeted value) was low at 0 weeks 6. Individuals who abstain from drinking alcohol from the day before measurement tothemeasurement end 7. Individuals who agree to participate in the stydy 8. Individuals judged appropriate for the study by attendant physician Subject Selection and Baseline Data in Human Study The effect of glucosylceramide in improving skin barrier function was evaluated in healthy volunteers, using konjac extract. The subjects were 100 healthy individuals who were recruited under the inclusion criteria listed in Table 1, and who were not excluded under the exclusion criteria listed in Table 2. Upon completion of the study, subjects found to fall under any of the exclusion criteria, those who had violated the instructions (Table 3), and these whose compliance with the ingestion protocol was poor were excluded from evaluation if deemed necessary at a case evaluation meeting held before the Table 2. Exclusion Criteria 1. Individuals with abnormalities of skin areas planned to be tested 2. Individuals considered likely to develop allergy to the test substance 3. Individuals with chronic cutis symptoms such as atopic dermatitis 4. Patients receiving drug therapy as outpatients 5. Individuals participating inother studies withinthe past 3months 6. Habitual smokers 7. Patients with asthma 8. Pregnant women, women desiring to become pregnant, or lactating women 9. Individuals having used cosmetics or the like possessing moisture-retaining effects on the skin areas planned to be tested in this study within 1 month before the sart of the study 10. Individuals taking or intending to take drugs against pollinosis 12. Individuals who used a ceramide-containing supplement within the past 1 month 13. Individuals who used ceramide- or retinol-containing cosmetics within the past 1 month 14. Individuals judged inappropriate for the study by attending physician key was opened. As shown in Fig. 3, excluding these subjects who dropped out of the study or violated the protocol, 83 subjects were included in the final analysis.
6 564 Vol. 54 (2008) Among these 83 subjects, there was no significant difference of mean age between the test product group (40.6 ± 9.1 years) and the control group (39.5 ± 8.9 years). Cheek TEWL at week 0 was 26.3 ± 6.8 g/m 2 hr in the test product group and 24.9 ± 8.6 g/m 2 hr in the control group (p =0.408), and forearm TEWL at week 0 was 7.6 ± 2.0 and Table 3. Instructions to Subjects during the Study Period 1. Only cosmetics which were being used before the study began may be used during the study. 2. Cutting of hair or shaving of the test areas is prohibited for 2 weeks before each occasion of measurement. 3. The use of bathing agent or the like is prohibited. 4. The use of medicines, quasi-medicines of new category, and herbal mixtures is prohibited as a rule. 5. Ingestion of food supplements or foods with healthpromoting function (specific health-promoting foods), othe than those that were being used routinely before the study, is prohibited. 6. Excessive alcohol consumption (consumption beyond the customary level) is prohibited. 7. No alcohol may be consumed on the day before each occasion of measurement. 8. Outdoor sports, exposure to artificial ultraviolet ray lamps, and other acts that might cause sunburn are prohibited. Care needs to be taken during daily living to avoid direct exposure of the test skin areas to ultraviolet rays indoors and outdoors. Concretely, it si advisable to protect the test skin areas with a cap or with clothes, or by routinely used sun-screening agents or the like. 9. Hard exercise is prohibited on the day of measurement until measurement for that day is completed. 10. Stimulating foods (hot foods, etc.) such as curry, red pepper, Tabasco, etc. may not be ingested on the day of measurement. 11. Use of thermal undershirts or the like, which exert special heat insulating effects, is prohibited. 12. A bath should be taken before going to bed on the day before measurement and bathing on the day of measurement (prior to measurement) is prohibited. 8.0 ± 2.0 g/m 2 hr, respectively (p =0.477). Therefore there was no significant difference of baseline data between the test and control groups. Sex and age composition of the analysis subjects is shown in Table 4. Effect of Konjac Extract on TEWL and Itching Sensation As shown in Fig.4(A), cheek TEWL at week 8 was 1.2 ± 5.2 g/m 2 hr in the test product group and 1.5 ± 5.5 g/m 2 hr in the control group (p =0.023), whereas that at week 12 was 2.9 ± 5.5 and 0.9 ± 5.6 g/m 2 hr, respectively (p =0.002). In ANCOVA, involving correction for baseline data, TEWL differed significantly between the two groups at weeks 8and 12 of ingestion, irrespective of the difference in baseline data (p =0.035 and p =0.004, respectively). TEWL in the forearm was also measured, but no significant difference was noted between the two groups. In addition, the back TEWL was significantly lower in the test product group (0.3 ± 2.2 g/m 2 hr) as compared with the control group (1.4 ± 2.4 g/m 2 hr) at weeks 12 of ingestion (p = 0.032). Thedegreeofimprovement of itching sensation following ingestion of konjac extract was evaluated in the face and arm of subjects by horizontal VAS. As shown in Fig. 4(B), itchiness of the arm was ameliorated in the konjac extract group after 4, 8, and 12 weeks of ingestion and at 4 weeks after the end of ingestion (week 16) (p =0.003, 0.059, 0.041, and 0.058). No marked effect on itching sensation of the face was found. Whereas, itching sensation of the leg was lower in the test product group as compared with the control group at weeks 4 and 8 of ingestion (p =0.075 and 0.084). Table 4. Sex and Average Age of Analysis Subjects Control group Konjac extract Total group Sex n Age years n Age years n Age years Male ± ± ± 10.1 Female ± 8.0 (30 57) ± 7.7 (28 56) ± 7.8 (28 57) All subjects ± ± ± 8.9 n =No.of subjects. Upper section shows mean ± S.D.; lower section shows age range.
7 No Fig. 4. Effect of Konjac Extract-containing Beverage (A) TEWL at cheeks and forearms was measured by evaporimeter at weeks 0, 4, 8, and 12 during ingestion period and week 16 in follow-up period. Variation of TEWL relative to baseline level ( TEWL) is shown. (B) Severity of itching sensation over faces and arms was evaluated by VAS at weeks 0, 4, 8, and 12 during ingestion period and week 16 in follow-up period. Variation of severity relative to baseline level ( value) is shown. indicates placebo; indicates test product. Student s t-test was used for comparison between two groups. Data are presented as mean ± S.D. Asterisks indicate a statistically significant difference, p < 0.05, p < DISCUSSION Oral intake of pure glucosylceramide improved skin roughness caused by SDS treatment in mice. This result is consistent with previous studies showing that corn and rice extracts containing glucosylceramide improve the skin condition in mouse models of chronic skin roughness induced by HR- AD and acute skin roughness induced by tape stripping. 13) Thus although there is considerable indirect evidence that glucosylceramide is effective in ameliorating skin roughness caused by environmental factors, the present study demonstrates clearly for the first time that the effect of konjac extract in decreasing TEWL is attributable to glucosylceramide contained in the extract. We additionally demonstrated that ingestion of glucosylceramide reduces the skin tissue level of IL-1α (a factor that exacerbates skin roughness) in a mouse model of skin roughness induced by a low relative humidity (< 10%) environment. In addition, we have previously observed that ingested glucosylceramide or its metabolite is distributed to the skin (data not shown). These findings suggest that glucosylceramide may reduce TEWL by suppressing induction of IL-1α in response to changes in the external environment. However, the possibility remains that glucosylceramide may play a more direct role in improving barrier function, because an increase of stratum corneum ceramide was observed following ingestion of glucosylceramide for 4 weeks. 19) Although it has been reported that barrier function is improved by uptake of konjac extract in patients with atopic dermatitis, 15, 16) we found that these effects are also observed in healthy volunteers whose daily life and activities during the study were restricted to only a small degree. Thus konjac extract also appears effective for improving skin condition in healthy subjects. However, the pattern of improvement was not similar between face and arm. Although there is not clear explanation for this effect, the correlation curve of TEWL and itching may be non-linear and the curve may differ from site to site. Further studies on IL-1α levels in skin of subjects who have taken konjac extract, and on the regulatory mechanism of IL-1α roductions are planned. In conclusion, test beverage containing konjac extract improved skin condition including barrier function, alleviated itching sensation in volunteers who took it for 12 weeks, and inhibited IL-1α pro-
8 566 Vol. 54 (2008) duction in skin. Therefore konjac extract may alleviate sleeplessness caused by itching, which should help people to lead more active daily lives and improve their QOL. REFERENCES 1) Werner, Y. and Lindberg, M. (1985) Transepidermal water loss in dry and clinically normal skin in patients with atopic dermatitis. Acta Derm. Venereol., 65, ) Loden, M., Olsson, H., Axell, T. and Linde, Y. W. (1992) Friction, capacitance and transepidermal water loss (TEWL) in dry atopic and normal skin. Br. J. Dermatol., 126, ) Thune, P. (1989) Evaluation of the hydration and the water-holding capacity in atopic skin and so-called dry skin. Acta Derm. Venereol. Suppl. (Stockh.), 144, ) Berardesca, E., Fideli, D., Borroni, G., Rabbiosi, G. and Maibach, H. (1990) In vivo hydration and waterretention capacity of stratum corneum in clinically uninvolved skin in atopic and psoriatic patients. Acta Derm. Venereol., 70, ) Sunwoo, Y., Chou, C., Takeshita, J., Murakami, M. and Tochihara, Y. (2006) Physiological and subjective responses to low relative humidity. J. Physiol. Anthropol., 25, ) Sunwoo, Y., Chou, C., Takeshita, J., Murakami, M. and Tochihara, Y. (2006) Physiological and subjective responses to low relative humidity in young and elderly men. J. Physiol. Anthropol., 25, ) Tupker, R. A., Bunte, E. E., Fidler, V., Wiechers, J. W. and Coenraads, P. J. (1999) Irritancy ranking of anionic detergents using one-time occlusive, repeated occlusive and repeated open tests. Contact Dermatitis, 40, ) Barbaro, D., Macchia, E., Orsini, P., Piazza, F., Lapi, P. and Pasquini, C. (2004) Assay of free thyroxine and free triiodothyronine in fine-needle aspiration of thyroid nodules: a useful and low-cost assessment. Endocr. Pract., 10, ) Imokawa, G., Abe, A., Jin, K., Higaki, Y., Kawashima, M. and Hidano, A. (1991) Decreased level of ceramides in stratum corneum of atopic dermatitis: an etiologic factor in atopic dry skin? J. Invest. Dermatol., 96, ) Di, Nardo, A., Wertz, P., Giannetti, A. and Seidenari, S. (1998) Ceramide and cholesterol composition of the skin of patients with atopic dermatitis. Acta Derm. Venereol., 78, ) Sugawara, T. and Miyazawa, T. (1999) Separation and determination of glycolipids from edible plant sources by high-performance liquid chromatography and evaporative light-scattering detection. Lipids, 34, ) Mukai, K. (2002) The function of an edible ceramide extracted from the tuber of konjac. Bio Industry, 19, ) Tsuji, K., Mitsutake, S., Ishikawa, J., Takagi, Y., Akiyama, M., Shimizu, H., Tomiyama, T. and Igarashi, Y. (2006) Dietary glucosylceramide improves skin barrier function in hairless mice. J. Dermatol. Sci., 44, ) Rogiers, V. and EEMCO Group. (2001) EEMCO guidance for the assessment of transepidermal water loss in cosmetic sciences. Skin Pharmacol. Appl. Skin Physiol., 14, ) Kimata, H. (2006) Improvement of atopic dermatitis and reduction of skin allergic responses by oral intake of konjac ceramide. Pediatr. Dermatol., 23, ) Miyanishi, K., Shiono, N., Shirai, H., Dombo, M. and Kimata, H. (2005) Reduction of transepidermal water loss by oral intake of glucosylceramides in patients with atopic eczema, Allergy, 60, ) Wood, L. C., Elias, P. M., Calhoun, C., Tsai, J. C., Grunfeld, C. and Feingold, K. R. (1996) Barrier disruption stimulates interleukin-1 alpha expression and release from a pre-formed pool in murine epidermis. J. Invest. Dermatol., 106, ) Ashida, Y., Ogo, M. and Denda, M. (2001) Epidermal interleukin-1 alpha generation is amplified at low humidity: implications for the pathogenesis of inflammatory dermatoses. Br. J. Dermatol., 144, ) Tsuboi, M. (2005) Validity to the skin by the student from skin-ceramide of phyto-ceramide. Food processing and ingredients, 40,
Moisturizing effects of topical nicotinamide on atopic dry skin
 Oxford, IJD International 1365-4632 Blackwell 45 UK Publishing, Journal Ltd, of Ltd. Dermatology 2004 Report Soma Reportet al. effects of topical nicotinamide Moisturizing effects of topical nicotinamide
Oxford, IJD International 1365-4632 Blackwell 45 UK Publishing, Journal Ltd, of Ltd. Dermatology 2004 Report Soma Reportet al. effects of topical nicotinamide Moisturizing effects of topical nicotinamide
Efficacy and safety of pseudo-ceramide containing moisture cream in the treatment of senile xerosis
 Hong Kong J. Dermatol. Venereol. (2009) 17, 181-189 Original Article Efficacy and safety of pseudo-ceramide containing moisture cream in the treatment of senile xerosis NM Luk, HC Lee, KLE Hon, K Ishida
Hong Kong J. Dermatol. Venereol. (2009) 17, 181-189 Original Article Efficacy and safety of pseudo-ceramide containing moisture cream in the treatment of senile xerosis NM Luk, HC Lee, KLE Hon, K Ishida
DermaPep A530. Multifunctional anti-inflammatory peptide for irritated and sensitive skin. Experience The Magic of Science
 Experience The Magic of Science DermaPep A53 Multifunctional anti-inflammatory peptide for irritated and sensitive skin DermaP ep Experience the magic of science Anti-aging DermaPep A35 DermaPep A42 DermaPep
Experience The Magic of Science DermaPep A53 Multifunctional anti-inflammatory peptide for irritated and sensitive skin DermaP ep Experience the magic of science Anti-aging DermaPep A35 DermaPep A42 DermaPep
The lipid barrier of the Stratum Corneum
 The lipid barrier of the Stratum Corneum Stratum Corneum: the "Bricks and Mortar" model Ceramides (40-65% total lipids) HN O Glucoceramides HN O O HO O cerabrosidase Cholesteryl sulphate Free fatty acids
The lipid barrier of the Stratum Corneum Stratum Corneum: the "Bricks and Mortar" model Ceramides (40-65% total lipids) HN O Glucoceramides HN O O HO O cerabrosidase Cholesteryl sulphate Free fatty acids
Application of Skin-Identical Ceramide 3 for Enhanced Skin Moisturization and Smoothness: Latest Results
 Application of Skin-Identical Ceramide 3 for Enhanced Skin Moisturization and Smoothness: Latest Results By Ute Wollenweber* and Dr. Mike Farwick* Keywords: Ceramides, Stratum corneum, skin moisturization,
Application of Skin-Identical Ceramide 3 for Enhanced Skin Moisturization and Smoothness: Latest Results By Ute Wollenweber* and Dr. Mike Farwick* Keywords: Ceramides, Stratum corneum, skin moisturization,
Skin Barrier Function as a Self-Organizing System
 Review Forma, 15, 227 232, 2000 Skin Barrier Function as a Self-Organizing System Mitsuhiro DENDA Shiseido Research Center, 2-12-1 Fukuura, Kanazawa-ku, Yokohama, Kanagawa 236-8643, Japan E-mail: mitsuhiro.denda@to.shiseido.co.jp
Review Forma, 15, 227 232, 2000 Skin Barrier Function as a Self-Organizing System Mitsuhiro DENDA Shiseido Research Center, 2-12-1 Fukuura, Kanazawa-ku, Yokohama, Kanagawa 236-8643, Japan E-mail: mitsuhiro.denda@to.shiseido.co.jp
Update on emollients
 Update on emollients Amal Mhanna, MD Pediatric Dermatologist Clemenceau Medical Center Disclosure: I was a member of an advisory board y for J&J and received honoraria. Emollients and moisturizers are
Update on emollients Amal Mhanna, MD Pediatric Dermatologist Clemenceau Medical Center Disclosure: I was a member of an advisory board y for J&J and received honoraria. Emollients and moisturizers are
DAILY CARE FOR DRY, IRRITATED AND REACTIVE SKIN. Physiogel A.I. Help your patients restore skin balance and well-being
 DAILY CARE FOR DRY, IRRITATED AND REACTIVE SKIN Help your patients restore skin balance and well-being Dry, irritated and reactive skin a question of balance Changes in the lipid barrier (1) Neurological
DAILY CARE FOR DRY, IRRITATED AND REACTIVE SKIN Help your patients restore skin balance and well-being Dry, irritated and reactive skin a question of balance Changes in the lipid barrier (1) Neurological
Objective To determine the immediate irritation effects and the irritation power of a particular materials.
 SAFETY STUDIES 1.1 Skin Irritation through Open Patch Test 1.2 Skin Irritation through Patch Test. 1.3 Human Skin Irritation Test 1.4 Repeat Open Application Test 1.5 Test For Hypoallergenicity 1.6 Modified
SAFETY STUDIES 1.1 Skin Irritation through Open Patch Test 1.2 Skin Irritation through Patch Test. 1.3 Human Skin Irritation Test 1.4 Repeat Open Application Test 1.5 Test For Hypoallergenicity 1.6 Modified
Lipo-amino acid cholesteryl derivatives promote recovery
 j. Soc. Cosmet. Chem., 47, 351-362 (November/December 1996) Lipo-amino acid cholesteryl derivatives promote recovery effect for damaged skin HIROJI ISHII, NAOKO MIKAMI, and KAZUTAMI SAKAMOTO, Ajinomoto
j. Soc. Cosmet. Chem., 47, 351-362 (November/December 1996) Lipo-amino acid cholesteryl derivatives promote recovery effect for damaged skin HIROJI ISHII, NAOKO MIKAMI, and KAZUTAMI SAKAMOTO, Ajinomoto
A unique gel matrix moisturizer delivers better skin barrier through increase of long chain acyl ceramide species
 Poster 7487 A unique gel matrix moisturizer delivers better skin barrier through increase of long chain acyl ceramide species Apostolos Pappas 1, Allison Rush 1, Anna Nicolaou 2, Julie Bianchini 1, Luke
Poster 7487 A unique gel matrix moisturizer delivers better skin barrier through increase of long chain acyl ceramide species Apostolos Pappas 1, Allison Rush 1, Anna Nicolaou 2, Julie Bianchini 1, Luke
Bio-mimetic, powerful moisturizing, and High functional Mild ingredients Made by NFC Sales Team Published date : January. 2018
 Bio-mimetic Gel Base Bio-mimetic, powerful moisturizing, and High functional Mild ingredients Made by NFC Sales Team Published date : January. 2018 Unique Efficacy of Bio-mimetic Gel Base This bio-mimetic
Bio-mimetic Gel Base Bio-mimetic, powerful moisturizing, and High functional Mild ingredients Made by NFC Sales Team Published date : January. 2018 Unique Efficacy of Bio-mimetic Gel Base This bio-mimetic
Pyranine, a fluorescent dye, detects subclinical injury to
 j. Cosmet. Sci., 49, 33-38 (January/February 1998) Pyranine, a fluorescent dye, detects subclinical injury to sodium lauryl sulfate A. PAGNONI, A.M. KLIGMAN, and T. STOUDEMAYER, S.K.I.N. Inc., 151 E. loth
j. Cosmet. Sci., 49, 33-38 (January/February 1998) Pyranine, a fluorescent dye, detects subclinical injury to sodium lauryl sulfate A. PAGNONI, A.M. KLIGMAN, and T. STOUDEMAYER, S.K.I.N. Inc., 151 E. loth
Direction for Future Actions From dermatologists standpoint
 Research and Reviews Direction for Future Actions From dermatologists standpoint JMAJ 52(3): 168 172, 29 Akira HATTORI,* 1 Masutaka FURUE,* 2 Michihiro HIDE,* 3 Masahiko IWAI,* 4 Shusho OKADA* 5 Abstract
Research and Reviews Direction for Future Actions From dermatologists standpoint JMAJ 52(3): 168 172, 29 Akira HATTORI,* 1 Masutaka FURUE,* 2 Michihiro HIDE,* 3 Masahiko IWAI,* 4 Shusho OKADA* 5 Abstract
Ceramides and barrier function in healthy skin
 Downloaded from orbit.dtu.dk on: Feb 5, 8 Ceramides and barrier function in healthy skin Jungerstedt, J; Hellgren, Lars; Drachmann, Tue; Høgh, Julie Kaae; Jemec, GBE; Agner, T Published in: Acta Dermatovenereologica
Downloaded from orbit.dtu.dk on: Feb 5, 8 Ceramides and barrier function in healthy skin Jungerstedt, J; Hellgren, Lars; Drachmann, Tue; Høgh, Julie Kaae; Jemec, GBE; Agner, T Published in: Acta Dermatovenereologica
DESCRIPTION. Dosage: % Recommended ph:
 EVOIL JJB SENSATION DESCRIPTION Consists of a multifunctional emollient obtained from natural vegetable oils. High content (ca. 25 %) of long chain length fatty acid. Contains up to 75 % of unsaturated
EVOIL JJB SENSATION DESCRIPTION Consists of a multifunctional emollient obtained from natural vegetable oils. High content (ca. 25 %) of long chain length fatty acid. Contains up to 75 % of unsaturated
Unique Plant Lipids for Skin Rejuvenation and Hydration
 http://www.lef.org/ Life Extension Magazine February 2011 Unique Plant Lipids for Skin Rejuvenation and Hydration By Robert Haas, MS Cosmetic companies around the world have devoted exhaustive research
http://www.lef.org/ Life Extension Magazine February 2011 Unique Plant Lipids for Skin Rejuvenation and Hydration By Robert Haas, MS Cosmetic companies around the world have devoted exhaustive research
Clinico Pathological Test SCPA605-Essential Pathology
 Clinico Pathological Test SCPA605-Essential Pathology Somphong Narkpinit, M.D. Department of Pathogbiology, Faculty of Science, Mahidol University e-mail : somphong.nar@mahidol.ac.th Pathogenesis of allergic
Clinico Pathological Test SCPA605-Essential Pathology Somphong Narkpinit, M.D. Department of Pathogbiology, Faculty of Science, Mahidol University e-mail : somphong.nar@mahidol.ac.th Pathogenesis of allergic
Journal of Occupational Health
 Advance Publication Journal of Occupational Health Accepted for Publication May 7, 2009 J-STAGE Advance Published Date: Jun 18, 2009 Higher prevalence of dry symptoms in skin, eyes, nose and throat among
Advance Publication Journal of Occupational Health Accepted for Publication May 7, 2009 J-STAGE Advance Published Date: Jun 18, 2009 Higher prevalence of dry symptoms in skin, eyes, nose and throat among
Canola oil and hempseed oil for atopic dermatitis skin treatment
 Canola oil and hempseed oil for atopic dermatitis skin treatment Carmen Vincent, MD, PhD Dr Carmen Vincent Clinic of General and Aesthetic Dermatology Dr Irena Eris Cosmetic Laboratories, Centre for Science
Canola oil and hempseed oil for atopic dermatitis skin treatment Carmen Vincent, MD, PhD Dr Carmen Vincent Clinic of General and Aesthetic Dermatology Dr Irena Eris Cosmetic Laboratories, Centre for Science
Assessment of the in vitro skin irritation of chemicals using the Vitrolife-Skin human skin model
 AATEX 14, Special Issue, 417-423 Proc. 6th World Congress on Alternatives & Animal Use in the Life Sciences August 21-25, 2007, Tokyo, Japan Assessment of the in vitro skin irritation of chemicals using
AATEX 14, Special Issue, 417-423 Proc. 6th World Congress on Alternatives & Animal Use in the Life Sciences August 21-25, 2007, Tokyo, Japan Assessment of the in vitro skin irritation of chemicals using
Yeast Essence Skin Care Actives. Yeast Essence C90 Yeast Essence E100 Yeast Essence N80 Yeast Essence Z20. Angel Yeast Co., Ltd.
 Yeast Essence Skin Care Actives Yeast Essence C90 Yeast Essence E100 Yeast Essence N80 Yeast Essence Z20 Angel Yeast Co., Ltd. Yeast Essence C90 Proposed INCI name: Sodium carboxymethyl beta glucan, water
Yeast Essence Skin Care Actives Yeast Essence C90 Yeast Essence E100 Yeast Essence N80 Yeast Essence Z20 Angel Yeast Co., Ltd. Yeast Essence C90 Proposed INCI name: Sodium carboxymethyl beta glucan, water
Polyaldo 10-1-CC Polyglyceryl Ester Naturally-Derived Surfactant for Excellent Mildness and Foam Properties
 Personal Care Polyaldo 10-1-CC Polyglyceryl Ester Naturally-Derived Surfactant for Excellent Mildness and Foam Properties INCI Name: Polyglyceryl-10 Caprylate/Caprate SAP Code #: 177445 Versatile Polyglyceryl
Personal Care Polyaldo 10-1-CC Polyglyceryl Ester Naturally-Derived Surfactant for Excellent Mildness and Foam Properties INCI Name: Polyglyceryl-10 Caprylate/Caprate SAP Code #: 177445 Versatile Polyglyceryl
Tetsuji WAKUDA, Masanao SHIBASAKI,Tadashi TANITSU* National University Corporation, Tsukuba University of Technology *Cooperation with research
 NTUT Education of Disabilities, 2009 Vol. 7 Effects of Acupuncture on Control of Pruritus Associated with Atopic Dermatitis Course in Acupuncture and Moxibustion, Department of Health, Tsukuba University
NTUT Education of Disabilities, 2009 Vol. 7 Effects of Acupuncture on Control of Pruritus Associated with Atopic Dermatitis Course in Acupuncture and Moxibustion, Department of Health, Tsukuba University
What is Pandoradiet TM? Standardization of Pandoradiet TM. Safety of Pandoradiet TM. Efficacies of Pandoradiet TM
 What is andoradiet TM? andoradiet TM is a proprietary extract powder of Boesenbergia pandurata, an edible plant with efficacies related to weight management and anti-skin aging. These effects have been
What is andoradiet TM? andoradiet TM is a proprietary extract powder of Boesenbergia pandurata, an edible plant with efficacies related to weight management and anti-skin aging. These effects have been
by G.L. Grove *1 C.R. Zerweck 1 J.M. Heilman 2 J.D. Pyrek 2 K.G.L. Skin Study Center; Broomall, PA 3M Health Care, St. Paul, MN
 Evaluation of Hand Skin Condition in Two 5-Day Surgical Scrub/Hand Washing Studies Comparing a New Waterless/Brushless, Chlorhexidine Gluconate/Ethanol-Emollient Antiseptic Hand Preparation and Hibiclens
Evaluation of Hand Skin Condition in Two 5-Day Surgical Scrub/Hand Washing Studies Comparing a New Waterless/Brushless, Chlorhexidine Gluconate/Ethanol-Emollient Antiseptic Hand Preparation and Hibiclens
Patients' non-compliance and
 The moisturising properties of a heel balm in patients with rough dry skin Dry plantar skin affects many older people and can result in callous and fissuring, which can reduce mobility of sufferers, cause
The moisturising properties of a heel balm in patients with rough dry skin Dry plantar skin affects many older people and can result in callous and fissuring, which can reduce mobility of sufferers, cause
AcquaSeal Algae. Defends Against Aging Skin + Cellular Hydration + Anti-Inflammation. Tomorrow s Vision Today!
 Defends Against Aging Skin + Cellular Hydration + Anti-Inflammation Tomorrow s Vision Today! Technical Information Product Code: 20852 INCI Name: Chlamydomonas Reinhardtii Extract INCI Status: Conforms
Defends Against Aging Skin + Cellular Hydration + Anti-Inflammation Tomorrow s Vision Today! Technical Information Product Code: 20852 INCI Name: Chlamydomonas Reinhardtii Extract INCI Status: Conforms
Skin Care Actives. Yeast Essence C90 Yeast Essence E100 Yeast Essence N80 Yeast Essence Z20. Ferment Essence FC01 Ferment Essence FG01
 Skin Care Actives Yeast Essence C90 Yeast Essence E100 Yeast Essence N80 Yeast Essence Z20 Ferment Essence FC01 Ferment Essence FG01 Angel Yeast Co., Ltd. Yeast Essence C90 INCI name: Sodium carboxymethyl
Skin Care Actives Yeast Essence C90 Yeast Essence E100 Yeast Essence N80 Yeast Essence Z20 Ferment Essence FC01 Ferment Essence FG01 Angel Yeast Co., Ltd. Yeast Essence C90 INCI name: Sodium carboxymethyl
Skinwell TM. Portulaca Oleracea extract. Shanghai Huiwen Biotech Co., Ltd.
 Skinwell TM Portulaca Oleracea extract Shanghai Huiwen Biotech Co., Ltd. Purslane (Portulaca Oleracea L.) Skinwell excellent anti- irritant Raw Material: Stem and leaf of Potalaca Olerace Herb. Main Active
Skinwell TM Portulaca Oleracea extract Shanghai Huiwen Biotech Co., Ltd. Purslane (Portulaca Oleracea L.) Skinwell excellent anti- irritant Raw Material: Stem and leaf of Potalaca Olerace Herb. Main Active
Emulsions. Purpose of emulsions and of emulsification:
 Pharmacist Ghada Hamid Emulsions Emulsion is a dispersion in which the dispersed phase is composed of small globules of a liquid distributed throughout a vehicle in which it is immiscible. The dispersed
Pharmacist Ghada Hamid Emulsions Emulsion is a dispersion in which the dispersed phase is composed of small globules of a liquid distributed throughout a vehicle in which it is immiscible. The dispersed
Association between itching and the serum zinc levels in patients with varicose veins
 Takai et al. Journal of Pharmaceutical Health Care and Sciences (2017) 3:24 DOI 10.1186/s40780-017-0092-9 RESEARCH ARTICLE Open Access Association between itching and the serum zinc levels in patients
Takai et al. Journal of Pharmaceutical Health Care and Sciences (2017) 3:24 DOI 10.1186/s40780-017-0092-9 RESEARCH ARTICLE Open Access Association between itching and the serum zinc levels in patients
Induction of a Hardening Phenomenon by Repeated Application of SLS: Analysis of Lipid Changes in the Stratum Corneum
 Acta Derm Venereol 2005; 85: 290 295 INVESTIGATIVE REPORT Induction of a Hardening Phenomenon by Repeated Application of SLS: Analysis of Lipid Changes in the Stratum Corneum Christian HEINEMANN 1, Christiane
Acta Derm Venereol 2005; 85: 290 295 INVESTIGATIVE REPORT Induction of a Hardening Phenomenon by Repeated Application of SLS: Analysis of Lipid Changes in the Stratum Corneum Christian HEINEMANN 1, Christiane
HL-60 ATP assay for predicting rat oral toxicity study
 AATEX 14, Special Issue, 699-703 Proc. 6th World Congress on Alternatives & Animal Use in the Life Sciences August 21-25, 2007, Tokyo, Japan HL-60 ATP assay for predicting rat oral toxicity study Yumiko
AATEX 14, Special Issue, 699-703 Proc. 6th World Congress on Alternatives & Animal Use in the Life Sciences August 21-25, 2007, Tokyo, Japan HL-60 ATP assay for predicting rat oral toxicity study Yumiko
1. Ask students to look at the skin on the backs of their hands and their arms.
 Fun in the Sun Integration: Health (Personal Health, Safety and First Aid); Science; Language Arts; Art Grade Levels: 2-3 Time: 2-3 class periods Materials: diagram of the structure of skin bottle or tube
Fun in the Sun Integration: Health (Personal Health, Safety and First Aid); Science; Language Arts; Art Grade Levels: 2-3 Time: 2-3 class periods Materials: diagram of the structure of skin bottle or tube
WHAT IS ActiLipid? WHAT IS IT FOR? HOW IS IT DIFFERENT? WHAT DOES IT DO?
 CTFA/INCI Name: Triticum Vulgare (Wheat) Germ Extract Olea Europaea (Olive) Fruit Extract Coleus Forskohlii Root Extract Polygonum Cuspidatum (Japanese Bamboo) Extract Helianthus Annuus (Sunflower) Seed
CTFA/INCI Name: Triticum Vulgare (Wheat) Germ Extract Olea Europaea (Olive) Fruit Extract Coleus Forskohlii Root Extract Polygonum Cuspidatum (Japanese Bamboo) Extract Helianthus Annuus (Sunflower) Seed
Formulation design of pasty facial-washes consisting of lowconcentration fatty acids and their effectiveness
 Formulation design of pasty facial-washes consisting of lowconcentration fatty acids and their effectiveness Ippei Tanaka 1, Emiko Hayashi 1, Takayuki Kado 1, Yukihiro Kembo 1, Cynthia Qi 2, and Toshio
Formulation design of pasty facial-washes consisting of lowconcentration fatty acids and their effectiveness Ippei Tanaka 1, Emiko Hayashi 1, Takayuki Kado 1, Yukihiro Kembo 1, Cynthia Qi 2, and Toshio
Unisooth PN-47 A complete reduction of pro-inflammatory factors for an instant soothing
 Unisooth PN-47 A complete reduction of pro-inflammatory factors for an instant soothing Skin irritation is amplified and controlled by various signaling pathways. By acting directly on the causes of skin
Unisooth PN-47 A complete reduction of pro-inflammatory factors for an instant soothing Skin irritation is amplified and controlled by various signaling pathways. By acting directly on the causes of skin
An Everyday Guide to Eczema
 An Everyday Guide to Eczema By Dr. Kristel Polder, Board-Certified Dermatologist Developed in Partnership with Who is affected by eczema? 32 million people in the US 1 in 5 children 1 in 12 adults *www.eczema.org
An Everyday Guide to Eczema By Dr. Kristel Polder, Board-Certified Dermatologist Developed in Partnership with Who is affected by eczema? 32 million people in the US 1 in 5 children 1 in 12 adults *www.eczema.org
The Ointment is back!
 Now Available for Plaque Psoriasis The Ointment is back! Bioequivalent to Dovonex * Ointment Before prescribing, please see attached full Prescribing Information. The same Delivers the reliability you
Now Available for Plaque Psoriasis The Ointment is back! Bioequivalent to Dovonex * Ointment Before prescribing, please see attached full Prescribing Information. The same Delivers the reliability you
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/20911 holds various files of this Leiden University dissertation. Author: Janssens, Michelle Title: Atopic eczema : the role of stratum corneum lipids in
Cover Page The handle http://hdl.handle.net/1887/20911 holds various files of this Leiden University dissertation. Author: Janssens, Michelle Title: Atopic eczema : the role of stratum corneum lipids in
Special Report: How Your Shower May Be Hazardous To Your Health! By Joel Kaye, MA
 Special Report: How Your Shower May Be Hazardous To Your Health! By Joel Kaye, MA www.rightbraindiet.com It's funny how people are always concerned with drinking filtered water, when in fact you absorb
Special Report: How Your Shower May Be Hazardous To Your Health! By Joel Kaye, MA www.rightbraindiet.com It's funny how people are always concerned with drinking filtered water, when in fact you absorb
PRURITOCEPTIVE AND PSYCHOGENIC PRURITUS IN LICHEN SIMPLEX CHRONICUS
 ORIGINAL ARTICLES PRURITOCEPTIVE AND PSYCHOGENIC PRURITUS IN LICHEN SIMPLEX CHRONICUS Department of Dermatology and Venerology, ABSTRACT Pruritus is a diagnostic hallmark for Lichen Simplex Chronicus (LSC).
ORIGINAL ARTICLES PRURITOCEPTIVE AND PSYCHOGENIC PRURITUS IN LICHEN SIMPLEX CHRONICUS Department of Dermatology and Venerology, ABSTRACT Pruritus is a diagnostic hallmark for Lichen Simplex Chronicus (LSC).
SAFE? ALL YOU NEED ONE LAB
 Safety tests 1 Is your product SAFE? 2 Client orientation - Mainly In- vivo Tests - Clinical control - Volunteers: - 3000 volunteers database - Caucasian, negroid, asian - Several skin types - Fast recruitment
Safety tests 1 Is your product SAFE? 2 Client orientation - Mainly In- vivo Tests - Clinical control - Volunteers: - 3000 volunteers database - Caucasian, negroid, asian - Several skin types - Fast recruitment
Supports your health with an incredible amount of ingredients and triple powers
 Supports your health with an incredible amount of ingredients and triple powers Fucoidan AF Premium Ingredients (Liquids) Okinawa Mozuku extracts (Fucoidan), Fragrant vinegar powder (rice vinegar, Dextrin),
Supports your health with an incredible amount of ingredients and triple powers Fucoidan AF Premium Ingredients (Liquids) Okinawa Mozuku extracts (Fucoidan), Fragrant vinegar powder (rice vinegar, Dextrin),
Dermatology Literary Review
 Emollients and ageing skin: optimising effectiveness and safety of Nursing, Vol. 25, No.11, pages 596-598. 6 Article outlines the causes of dryness in ageing skin and concludes that emollients are the
Emollients and ageing skin: optimising effectiveness and safety of Nursing, Vol. 25, No.11, pages 596-598. 6 Article outlines the causes of dryness in ageing skin and concludes that emollients are the
Clinical Study Report SLO-AD-1 Final Version DATE: 09 December 2013
 1. Clinical Study Report RANDOMIZED, OPEN, PARALLEL GROUP, PHASE IIIB STUDY ON THE EVALUATION OF EFFICACY OF SPECIFIC SUBLINGUAL IMMUNOTHERAPY IN PAEDIATRIC PATIENTS WITH ATOPIC DERMATITIS, WITH OR WITHOUT
1. Clinical Study Report RANDOMIZED, OPEN, PARALLEL GROUP, PHASE IIIB STUDY ON THE EVALUATION OF EFFICACY OF SPECIFIC SUBLINGUAL IMMUNOTHERAPY IN PAEDIATRIC PATIENTS WITH ATOPIC DERMATITIS, WITH OR WITHOUT
Skin care products can aggravate epidermal function: studies in a murine model suggest a pathogenic role in sensitive skin
 Contact Dermatitis Original Article COD Contact Dermatitis Skin care products can aggravate epidermal function: studies in a murine model suggest a pathogenic role in sensitive skin Zhengxiao Li 1,2,LizhiHu
Contact Dermatitis Original Article COD Contact Dermatitis Skin care products can aggravate epidermal function: studies in a murine model suggest a pathogenic role in sensitive skin Zhengxiao Li 1,2,LizhiHu
How Should We Handle Them?
 Alternative and Complementary Therapies: How Should We Handle Them? Peter A. Lio, MD Assistant Professor Clinical Dermatology & Pediatrics Northwestern University Feinberg School of Medicine DISCLOSURE
Alternative and Complementary Therapies: How Should We Handle Them? Peter A. Lio, MD Assistant Professor Clinical Dermatology & Pediatrics Northwestern University Feinberg School of Medicine DISCLOSURE
Measurement of turnover time of stratum corneum using
 j. Soc. Cosmet. Chem., 38, 321-331 (September/October 1987) Measurement of turnover time of stratum corneum using dansyl chloride fluorescence MOTOJI TAKAHASHI, YASUHIKO MACHIDA, and RONALD MARKS, Shiseido
j. Soc. Cosmet. Chem., 38, 321-331 (September/October 1987) Measurement of turnover time of stratum corneum using dansyl chloride fluorescence MOTOJI TAKAHASHI, YASUHIKO MACHIDA, and RONALD MARKS, Shiseido
A Child with Eczema: A Parent s Guide
 A Child with Eczema: A Parent s Guide What is eczema? Originally the term eczema was used to describe any itchy rash that blistered; now eczema describes skin which is red (inflamed) and intensely itchy.
A Child with Eczema: A Parent s Guide What is eczema? Originally the term eczema was used to describe any itchy rash that blistered; now eczema describes skin which is red (inflamed) and intensely itchy.
2016 HEAT SAFETY KIT
 2016 HEAT SAFETY KIT PREVENTING HEAT-RELATED ILLNESSES HEAT ILLNESS AND EMERGENCIES Many factors contribute to heat-related illnesses, which can occur when an individual is subjected to extreme temperatures
2016 HEAT SAFETY KIT PREVENTING HEAT-RELATED ILLNESSES HEAT ILLNESS AND EMERGENCIES Many factors contribute to heat-related illnesses, which can occur when an individual is subjected to extreme temperatures
AUGUST 2018 SAFETY MEETING HEAT HAZARDS
 AUGUST 2018 SAFETY MEETING HEAT HAZARDS Hot weather can make us uncomfortable, and as the temperature rises, there are hot weather related conditions we should be aware of. Nearly 70 percent of Americans
AUGUST 2018 SAFETY MEETING HEAT HAZARDS Hot weather can make us uncomfortable, and as the temperature rises, there are hot weather related conditions we should be aware of. Nearly 70 percent of Americans
Trimoist KMF Biomimetic 24 h hydrator
 Biomimetic 24 h hydrator Biomimetic 24 h hydrator An Effective Hydro-Complex with a Skin-Like Moisturizing System Trimoist KMF is a blend of lamellar lipids, CM-Glucan, humectant agents and the anti-aging
Biomimetic 24 h hydrator Biomimetic 24 h hydrator An Effective Hydro-Complex with a Skin-Like Moisturizing System Trimoist KMF is a blend of lamellar lipids, CM-Glucan, humectant agents and the anti-aging
Dr David Orchard MBBS FACD
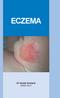 Eczema Dr David Orchard MBBS FACD What is eczema? Eczema is a particular type of inflammatory reaction in the skin. The features include redness, scaling and itch. When it is more severe the skin will
Eczema Dr David Orchard MBBS FACD What is eczema? Eczema is a particular type of inflammatory reaction in the skin. The features include redness, scaling and itch. When it is more severe the skin will
Yeast Essence C90 O O OH CH 2 O OH. OH O H Structure of Yeast Beta-Glucan C H 2 O C H 2 C O O N a
 Yeast Essence Proposed INCI name: Sodium carboxymethyl beta-glucan, water Introduction Yeast beta-glucan is a polysaccharide that exists in the cell wall of yeast, numerous clinical studies have shown
Yeast Essence Proposed INCI name: Sodium carboxymethyl beta-glucan, water Introduction Yeast beta-glucan is a polysaccharide that exists in the cell wall of yeast, numerous clinical studies have shown
Preventing irritant contact dermatitis with protective creams: influence of the application dose
 Contact Dermatitis Original Article COD Contact Dermatitis Preventing irritant contact dermatitis with protective creams: influence of the application dose Sibylle Schliemann, Maximilian Petri and Peter
Contact Dermatitis Original Article COD Contact Dermatitis Preventing irritant contact dermatitis with protective creams: influence of the application dose Sibylle Schliemann, Maximilian Petri and Peter
SWEOAT products for different uses and markets. Oats for the food industry. SWEOAT Brans for food
 SWEOAT products for different uses and markets Swedish Oat Fiber has for years been a trusted supplier to different kinds of industries locally in Sweden and globally at every continent. Our products are
SWEOAT products for different uses and markets Swedish Oat Fiber has for years been a trusted supplier to different kinds of industries locally in Sweden and globally at every continent. Our products are
The Effects of Different Intervals of Microdermabrasion Sessions on Skin Biophysical Parameters: A Randomized, Assessor-Blind, Within-Patient Trial
 Original Article The Effects of Different Intervals of Microdermabrasion Sessions on Skin Biophysical Parameters: A Randomized, Assessor-Blind, Within-Patient Trial Parastoo Davari, MD Farzam Gorouhi,
Original Article The Effects of Different Intervals of Microdermabrasion Sessions on Skin Biophysical Parameters: A Randomized, Assessor-Blind, Within-Patient Trial Parastoo Davari, MD Farzam Gorouhi,
The Aloe Vera Specialist
 The Aloe Vera Specialist Ready-To-Mix Formula Direct from Manufacturer With Ingredients Included: Aloe Solids, Flavoring, Sweeteners, Gums, ph Buffers, etc. www.aloequeen.com 1 Rise in Aloe Vera Consumption
The Aloe Vera Specialist Ready-To-Mix Formula Direct from Manufacturer With Ingredients Included: Aloe Solids, Flavoring, Sweeteners, Gums, ph Buffers, etc. www.aloequeen.com 1 Rise in Aloe Vera Consumption
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory. Light Liquid Paraffin and White Soft Paraffin Cream
 For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory OILATUM CREAM / KIDS CREAM Light Liquid Paraffin and White Soft Paraffin Cream QUALITATIVE AND QUANTITATIVE COMPOSITION
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory OILATUM CREAM / KIDS CREAM Light Liquid Paraffin and White Soft Paraffin Cream QUALITATIVE AND QUANTITATIVE COMPOSITION
TNF-alpha ELISA. For Research Use Only. Not For Use In Diagnostic Procedures.
 TNF-alpha ELISA For the quantitative determination of TNF-alpha in serum, plasma, buffered solution or cell culture medium. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number:
TNF-alpha ELISA For the quantitative determination of TNF-alpha in serum, plasma, buffered solution or cell culture medium. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number:
Morinaga Mouse C-peptide ELISA Kit
 Morinaga Mouse C-peptide ELISA Kit For the quantitative determination of C-peptide in mouse serum, plasma, and fluid 96wells For Laboratory Use Only, not for use in diagnostic procedure Please read full
Morinaga Mouse C-peptide ELISA Kit For the quantitative determination of C-peptide in mouse serum, plasma, and fluid 96wells For Laboratory Use Only, not for use in diagnostic procedure Please read full
2012 Heat Safety Kit
 2012 Heat Safety Kit PREVENTING HEAT-RELATED ILLNESSES HEAT ILLNESS AND EMERGENCIES Heat-related illnesses have many factors but can be caused when an individual is subjected to extreme temperatures and
2012 Heat Safety Kit PREVENTING HEAT-RELATED ILLNESSES HEAT ILLNESS AND EMERGENCIES Heat-related illnesses have many factors but can be caused when an individual is subjected to extreme temperatures and
Compressor Nebulizer System
 MedPro The Professional Choice Le choix des professionnels MedProTM MC Compressor Nebulizer System 705-445 Instruction Manual Index 1. Introduction...2 2. Product Identification...2 3. Important Safeguards...3
MedPro The Professional Choice Le choix des professionnels MedProTM MC Compressor Nebulizer System 705-445 Instruction Manual Index 1. Introduction...2 2. Product Identification...2 3. Important Safeguards...3
FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH
 FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH ALETA A SOURCE OF 1,3-BETA GLUCANS Aleta is highly bioavailable, offering a concentration greater than 5% of 1,3-beta glucans. Aleta provides a consistent response
FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH ALETA A SOURCE OF 1,3-BETA GLUCANS Aleta is highly bioavailable, offering a concentration greater than 5% of 1,3-beta glucans. Aleta provides a consistent response
STARCHES FOR COSMETIC INDUSTRIES CORN PO4 PH B AND RICE NS
 STARCHES FOR COSMETIC INDUSTRIES CORN PO4 PH B AND RICE NS AGRANA STARCH QUALITATIVE OPTIMIZATION OF COSMETIC EMULSIONS WITH CORN PO4 PH B OR RICE NS For many years CORN PO4 PH B and RICE NS (cross-linked
STARCHES FOR COSMETIC INDUSTRIES CORN PO4 PH B AND RICE NS AGRANA STARCH QUALITATIVE OPTIMIZATION OF COSMETIC EMULSIONS WITH CORN PO4 PH B OR RICE NS For many years CORN PO4 PH B and RICE NS (cross-linked
The Moisturizing Effects of Glycolipid Biosurfactants, Mannosylerythritol Lipids, on Human Skin
 Journal of Oleo Science Copyright 2012 by Japan Oil Chemists Society NOTE The Moisturizing Effects of Glycolipid Biosurfactants, Mannosylerythritol Lipids, on Human Skin Shuhei Yamamoto 1, Tomotake Morita
Journal of Oleo Science Copyright 2012 by Japan Oil Chemists Society NOTE The Moisturizing Effects of Glycolipid Biosurfactants, Mannosylerythritol Lipids, on Human Skin Shuhei Yamamoto 1, Tomotake Morita
Frequently Asked Questions about extreme heat and public health
 PUBLIC HEALTH DIVISION Acute and Communicable Disease Prevention Program Frequently Asked Questions about extreme heat and public health Q: Who is at greatest risk for heat-related illness? A: Those at
PUBLIC HEALTH DIVISION Acute and Communicable Disease Prevention Program Frequently Asked Questions about extreme heat and public health Q: Who is at greatest risk for heat-related illness? A: Those at
Proceedings: Indoor Air 2005
 THE EFFECTS ON THE HUMAN BODY OF BREATHING AIR POSSESSING 30% OXYGEN- EFFECTS ON BLOOD GLUCOSE LEVELS, BODY WEIGHT/BODY FAT RATIOS, VISUAL ACUITY & STRATUM CORNEUM RENEWAL T. Oketa 1*, J. Nawama1, N. Hayashi,
THE EFFECTS ON THE HUMAN BODY OF BREATHING AIR POSSESSING 30% OXYGEN- EFFECTS ON BLOOD GLUCOSE LEVELS, BODY WEIGHT/BODY FAT RATIOS, VISUAL ACUITY & STRATUM CORNEUM RENEWAL T. Oketa 1*, J. Nawama1, N. Hayashi,
Assessing the Current Treatment of Atopic Dermatitis: Unmet Needs
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Constitutional eczema
 Patient information Constitutional eczema What is constitutional eczema? Constitutional eczema, also called atopic eczema, is a form of eczema that mainly occurs in childhood. Eczema usually starts before
Patient information Constitutional eczema What is constitutional eczema? Constitutional eczema, also called atopic eczema, is a form of eczema that mainly occurs in childhood. Eczema usually starts before
FERTIUP PM 1 ml / 0.5 ml - CARD MEDIUM Set
 Product manual FERTIUP PM 1 ml / 0.5 ml - CARD MEDIUM Set Cat. No. KYD-004-EX Size: 1 SET KYD-005-EX 1 SET Department of Reproductive Engineering, Center for Animal Resources and Development, Kumamoto
Product manual FERTIUP PM 1 ml / 0.5 ml - CARD MEDIUM Set Cat. No. KYD-004-EX Size: 1 SET KYD-005-EX 1 SET Department of Reproductive Engineering, Center for Animal Resources and Development, Kumamoto
ABS Acai Sterols EFA Natural + Sustainable + Protectant. Tomorrow s Vision Today!
 ABS Acai Sterols EFA Natural + Sustainable + Protectant Tomorrow s Vision Today! ABS Acai Sterols EFA Technical Information: Product Code: 10414 INCI Name: Euterpe Oleracea Sterols INCI Status: Conforms
ABS Acai Sterols EFA Natural + Sustainable + Protectant Tomorrow s Vision Today! ABS Acai Sterols EFA Technical Information: Product Code: 10414 INCI Name: Euterpe Oleracea Sterols INCI Status: Conforms
Normal cooling mechanisms Heat-related illnesses. Evaluating the risk of heat illness Controlling heat stress First aid
 Normal cooling mechanisms Heat-related illnesses Evaluating the risk of heat illness Controlling heat stress First aid When body core temperature rises Blood flow to skin increases Sweating increases Heart
Normal cooling mechanisms Heat-related illnesses Evaluating the risk of heat illness Controlling heat stress First aid When body core temperature rises Blood flow to skin increases Sweating increases Heart
Patch testing. Dermatology Department Patient Information Leaflet
 Patch testing Dermatology Department Patient Information Leaflet Introduction This leaflet has been written to help you understand more about patch testing. It tells you what a patch test is, what is involved
Patch testing Dermatology Department Patient Information Leaflet Introduction This leaflet has been written to help you understand more about patch testing. It tells you what a patch test is, what is involved
Medescan Nebuliser Med - S600A Instructions Manual
 Medescan Nebuliser Med - S600A Instructions Manual Please read this guidebook carefully before operating this unit Your Nebuliser is intended for use in the treatment of asthma, COPD and other respiratory
Medescan Nebuliser Med - S600A Instructions Manual Please read this guidebook carefully before operating this unit Your Nebuliser is intended for use in the treatment of asthma, COPD and other respiratory
Supplemental methods. Total RNA was extracted from the stomach, liver, pancreas, pituitary, and
 Supplemental methods Real-time quantitative RT-PCR and Semi-quantitative PCR Total RNA was extracted from the stomach, liver, pancreas, pituitary, and hypothalamus as previously described (). Primers and
Supplemental methods Real-time quantitative RT-PCR and Semi-quantitative PCR Total RNA was extracted from the stomach, liver, pancreas, pituitary, and hypothalamus as previously described (). Primers and
MEADOWESTOLIDE Vs. Ceramide IIIB
 MEADWESTLIDE Vs. Ceramide IIIB USA Head ffice Europe Asia Japan Elementis Specialties, Inc. Elementis Specialties Elementis Specialties, Inc. Elementis Japan KK P.. Box 700 Stolberger Straße 370 120 Jalan
MEADWESTLIDE Vs. Ceramide IIIB USA Head ffice Europe Asia Japan Elementis Specialties, Inc. Elementis Specialties Elementis Specialties, Inc. Elementis Japan KK P.. Box 700 Stolberger Straße 370 120 Jalan
Experiment Optional #2: The Synthesis of Aspirin
 Experiment Optional #2: The Synthesis of Aspirin The natural world provides us with many of the medications in common use today. Taxol is the common name of a medication used in treating certain cancers;
Experiment Optional #2: The Synthesis of Aspirin The natural world provides us with many of the medications in common use today. Taxol is the common name of a medication used in treating certain cancers;
PATIENT INFORMATION NEUPRO [NU pro] (rotigotine transdermal system)
![PATIENT INFORMATION NEUPRO [NU pro] (rotigotine transdermal system) PATIENT INFORMATION NEUPRO [NU pro] (rotigotine transdermal system)](/thumbs/77/76620751.jpg) PATIENT INFORMATION NEUPRO [NU pro] (rotigotine transdermal system) If you have Restless Legs Syndrome (also known as Willis-Ekbom disease), read this side. If you have Parkinson s disease, read the other
PATIENT INFORMATION NEUPRO [NU pro] (rotigotine transdermal system) If you have Restless Legs Syndrome (also known as Willis-Ekbom disease), read this side. If you have Parkinson s disease, read the other
ASTAXANTHIN - THE HEALTHY TEN
 ASTAXANTHIN - THE HEALTHY TEN THE HEALTHY TEN TEN CLINICALLY-VALIDATED BENEFITS OF NATURAL ASTAXANTHIN BRAIN HEALTH 4 Human Clinical Studies 50 Supporting Pre-Clinical Trials EYE HEALTH 14 Human Clinical
ASTAXANTHIN - THE HEALTHY TEN THE HEALTHY TEN TEN CLINICALLY-VALIDATED BENEFITS OF NATURAL ASTAXANTHIN BRAIN HEALTH 4 Human Clinical Studies 50 Supporting Pre-Clinical Trials EYE HEALTH 14 Human Clinical
Development of a Food Frequency Questionnaire in Japan. Chigusa Date1, Momoko Yamaguchi2, and Heizo Tanaka3
 Journal of Epidemiology Vol. 6, No. 3 (Supplement) August NUTRITIONAL EPIDEMIOLOGY Development of a Food Frequency Questionnaire in Japan Chigusa Date1, Momoko Yamaguchi2, and Heizo Tanaka3 The three-consecutive-day
Journal of Epidemiology Vol. 6, No. 3 (Supplement) August NUTRITIONAL EPIDEMIOLOGY Development of a Food Frequency Questionnaire in Japan Chigusa Date1, Momoko Yamaguchi2, and Heizo Tanaka3 The three-consecutive-day
Gatorade Heat Safety Package
 Gatorade Heat Safety Package 1. The Four Downs of Heat Safety 2. Tips to Hydrate and Fuel for the Football Season 3. Preventing Heat-Related Illnesses The 4 Downs of Heat Safety Know the plays of heat
Gatorade Heat Safety Package 1. The Four Downs of Heat Safety 2. Tips to Hydrate and Fuel for the Football Season 3. Preventing Heat-Related Illnesses The 4 Downs of Heat Safety Know the plays of heat
e n ta d i n e Photoprotection Anti-photoaging the most natural way to reinforce skin natural defenses against outdoor and indoor radiations
 eno p r e se rvativ e n ta d i n e Photoprotection Anti-photoaging the most natural way to reinforce skin natural defenses against outdoor and indoor radiations Anti-oxidation Anti-inflammation Anti-immunosuppression
eno p r e se rvativ e n ta d i n e Photoprotection Anti-photoaging the most natural way to reinforce skin natural defenses against outdoor and indoor radiations Anti-oxidation Anti-inflammation Anti-immunosuppression
Farida Tabri, Lisa Yuniati
 International Journal of Science and Healthcare Research Vol.; Issue: 4; Oct.-Dec. 2018 Website: www.ijshr.com Original Research Article ISSN: 2455-7587 A Randomized, Double-Blind Clinical Trial Comparing
International Journal of Science and Healthcare Research Vol.; Issue: 4; Oct.-Dec. 2018 Website: www.ijshr.com Original Research Article ISSN: 2455-7587 A Randomized, Double-Blind Clinical Trial Comparing
MATERIAL SAFETY DATA SHEET (MSDS)
 MATERIAL SAFETY DATA SHEET (MSDS) HEALTH: 2 FLAMMABILITY: 1 REACTIVITY: 1 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Product Name: Trade Name: Codes and colours: Manufacturer name: Ultraviolet light
MATERIAL SAFETY DATA SHEET (MSDS) HEALTH: 2 FLAMMABILITY: 1 REACTIVITY: 1 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Product Name: Trade Name: Codes and colours: Manufacturer name: Ultraviolet light
Increases Circulation Immune Complexes, Increased Fibrin Activation and Fibrosis
 Inflammation Inflammation is a complex biological process in which the body s white blood cells and chemicals provide protection from infection and foreign substances, such as bacteria, yeast, and viruses
Inflammation Inflammation is a complex biological process in which the body s white blood cells and chemicals provide protection from infection and foreign substances, such as bacteria, yeast, and viruses
A new biomimetic ingredient of the skin hydrolipidicfilm: clinical benefits. Performance-based Natural ingredients from
 A new biomimetic ingredient of the skin hydrolipidicfilm: clinical benefits Performance-based Natural ingredients from Olive Chemistry& HydrolipidicFilm Olive chemistry Hydrolipidic film Squalene (%) 0.4-0.7
A new biomimetic ingredient of the skin hydrolipidicfilm: clinical benefits Performance-based Natural ingredients from Olive Chemistry& HydrolipidicFilm Olive chemistry Hydrolipidic film Squalene (%) 0.4-0.7
Vitamin F- glyceric ester
 SENSORIAL Vitamin F- glyceric ester www.provitalgroup.com Vitamin F-glyceric ester BOTANY Vitamin F-Glyceric ester is a mixture of polyunsaturated fatty acids esterified and biologically actives. The polyunsaturated
SENSORIAL Vitamin F- glyceric ester www.provitalgroup.com Vitamin F-glyceric ester BOTANY Vitamin F-Glyceric ester is a mixture of polyunsaturated fatty acids esterified and biologically actives. The polyunsaturated
Lecithin and Phospholipids for Cosmetics Applications
 Lecithin and Phospholipids for Cosmetics Applications Beauty comes from within. Lecithin is a natural constituent of all living cells with essential functions for humans, animals and plants. The term Lecithin
Lecithin and Phospholipids for Cosmetics Applications Beauty comes from within. Lecithin is a natural constituent of all living cells with essential functions for humans, animals and plants. The term Lecithin
Extreme Heat: A Prevention Guide to Promote Health and Safety
 Extreme Heat: A Prevention Guide to Promote Health and Safety People suffer heat-related illness when their bodies are unable to compensate and properly cool themselves. The body normally cools itself
Extreme Heat: A Prevention Guide to Promote Health and Safety People suffer heat-related illness when their bodies are unable to compensate and properly cool themselves. The body normally cools itself
Skeletal muscle metabolism was studied by measuring arterio-venous concentration differences
 Supplemental Data Dual stable-isotope experiment Skeletal muscle metabolism was studied by measuring arterio-venous concentration differences across the forearm, adjusted for forearm blood flow (FBF) (1).
Supplemental Data Dual stable-isotope experiment Skeletal muscle metabolism was studied by measuring arterio-venous concentration differences across the forearm, adjusted for forearm blood flow (FBF) (1).
Neonatal Skin Physiology. Marty Visscher, PhD, Skin Sciences Program Cincinnati Children s Hospital Medical Center May 2011
 Neonatal Skin Physiology Marty Visscher, PhD, Skin Sciences Program Cincinnati Children s Hospital Medical Center May 2011 Change the Outcome Why Study Infant Skin? Skin contact is important for the development
Neonatal Skin Physiology Marty Visscher, PhD, Skin Sciences Program Cincinnati Children s Hospital Medical Center May 2011 Change the Outcome Why Study Infant Skin? Skin contact is important for the development
Kazumi YAGASAKI, Masato NAGAOKA and Yutaka MIURA
 Kazumi YAGASAKI, Masato NAGAOKA and Yutaka MIURA Division of Agriscience and Bioscience, Institute of Symbiotic Science and Technology, Tokyo University of Agriculture and Technology, Fuchu 183-8509 ABSTRACT
Kazumi YAGASAKI, Masato NAGAOKA and Yutaka MIURA Division of Agriscience and Bioscience, Institute of Symbiotic Science and Technology, Tokyo University of Agriculture and Technology, Fuchu 183-8509 ABSTRACT
It s all in your hands: Preventing hand dermatitis. Introduction 3/5/2018. Hand hygiene is a cornerstone of effective infection control, but
 It s all in your hands: Preventing hand dermatitis Monica Herrera MHA, CDMP Introduction Hand hygiene is a cornerstone of effective infection control, but Hand sanitization and gloving are essential if
It s all in your hands: Preventing hand dermatitis Monica Herrera MHA, CDMP Introduction Hand hygiene is a cornerstone of effective infection control, but Hand sanitization and gloving are essential if
Macromolecules Materials
 Macromolecules Materials Item per bench per class Test tubes 19 a bunch Benedict s reagent 1 bottle 6 Iodine bottle 1 bottle 6 Sudan IV bottle 1 bottle 6 Biuret s Bottle 1 bottle 6 250 ml beaker 1 6 heat
Macromolecules Materials Item per bench per class Test tubes 19 a bunch Benedict s reagent 1 bottle 6 Iodine bottle 1 bottle 6 Sudan IV bottle 1 bottle 6 Biuret s Bottle 1 bottle 6 250 ml beaker 1 6 heat
Egg Protein Residue ELISA Kit
 Egg Protein Residue ELISA Kit Catalog Number KA3208 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Egg Protein Residue ELISA Kit Catalog Number KA3208 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Active Beauty Rubixyl Anti stress-ageing peptide. Crafted by synthesis
 Active Beauty Rubixyl Anti stress-ageing peptide Crafted by synthesis Focus on the product The skin barrier function The most crucial part of the skin barrier functions is the stratum corneum. The stratum
Active Beauty Rubixyl Anti stress-ageing peptide Crafted by synthesis Focus on the product The skin barrier function The most crucial part of the skin barrier functions is the stratum corneum. The stratum
Effects of Skin Surface Temperature on Epidermal Permeability Barrier Homeostasis
 ORIGINAL ARTICLE Effects of Skin Surface Temperature on Epidermal Permeability Barrier Homeostasis Mitsuhiro Denda 1, Takaaki Sokabe 2, Tomoko Fukumi-Tominaga 2 and Makoto Tominaga 2 Members of the transient
ORIGINAL ARTICLE Effects of Skin Surface Temperature on Epidermal Permeability Barrier Homeostasis Mitsuhiro Denda 1, Takaaki Sokabe 2, Tomoko Fukumi-Tominaga 2 and Makoto Tominaga 2 Members of the transient
