Chapter 26: Lipids. Hydrophobic (non-polar, soluble in organic solvent), typically of low molecular compound or organic origin
|
|
|
- Ada Peters
- 5 years ago
- Views:
Transcription
1 hapter 26: Lipids. ydrophobic (non-polar, soluble in organic solvent), typically of low molecular compound or organic origin fatty acids and waxes essential oils many vitamins hormones (non-peptide) components of cell membranes (non-peptide) Share a common biosynthesis that ultimately derives their carbon source from glucose (glycolysis) Glucose pyruvate lactate : Acetyl oenzyme A. AcSoA is a thioester. R=, oas R= acetyl, AcSoA R S P P P Pyruvate dehydrogenase: Multi-enzyme complex that converts pyruvate to AcSoA. 2 2 oas 2 P P 2 pyruvate AD thiamin diphosphate (vitamin B 1 ) Lipoic Acid Flavin adenine diphosphate (VitaminB 2 ) S-oA 2 P P 2 2 AcSoA AD
2 Acetyl oa is a thioester. Thioesters are more reactive toward nucleophilic acyl substitution than esters, but considerably less reactive than acid chlorides and anhydrides. S-oA u- u oas S-oA ( 3 ) 3 choline choline acetyltransferase Thioester enolize more readily than esters. The enol can react with electrophile to afford α-substitution products acetylcholine ( 3 ) 3 oas S-oA S-oA E E S-oA acetyl-oa carboxylase S-oA 3 biotin, ATP S-oA Malonyl oa : Fats, ils, and Fatty Acids. Fatty acids: refers to long, straight-chain saturated and unsaturated acids, typically from (Table 26.1, p. 1069). saturated fatty acids: 3 ( 2 ) n 2 n=10, lauric acid ( 12 ) n=12, myristic acid ( 14 ) n=14, palmitic acid ( 16 ) n=16, steric acid ( 18 ) unsaturated fatty acid 2 18, oleic acid 3 polyunsaturated fatty acids (PUFA) , linolenic acid (18:3) 18, linoleic acid (18:4) 20, arachidonic acid (20:4)
3 Fats and ils: Triglycerides (triaceylglycerols) are tri-esters of glycerol (1,2,3-trihydroxypropane) and fatty acids. glycerol fatty acids R 1 R 2 R 3 The R groups can be saturated or unsaturated, the same or different 2 2 R 1 R 2 R 3 when some of the R groups are unsaturated 2, catalyst 2 2 R 1 R 2 R 3 Partially hydrogenated: some cis double bond are isomerized to trans double bonds 2, catalyst 2 2 R 1 R 2 R 3 ydrogenated- only saturated fatty acids 284 Soaps: sodium & potassium salts of fatty acid produced from the saponification (base hydrolysis) of animal fats (glycerides) 2 2 R n R n R n a 3 R n a 2 2 Soaps have a hydrophilic, polar head group (carboxylate salt) and a hydrophobic, non-polar tail. Fatty acid amides (FAA): Anandamide an ethanolamine amide of arachidonic Acid ( 20 ) tetrahydrocannabinol
4 26.3: Fatty Acid Biosynthesis. Fatty acid biosynthesis is performed by a cluster of discrete enzymes in bacteria, and a very large multi-protein assembly in animals (fatty acid synthase, FAS). The fatty acid is attached to an acyl carrier protein (AP), while other proteins perform an iterative two-carbon chain extension reaction that will yield the fatty acid. 2 S P P R S R AcS-oA P ysteine of Ketosynthase (KS) oa-s R S R AcS-KS 286 FAS chain extension reaction: Ketosynthase (KR) S-oA Malonyl oa S Phosphopantetheine P Serine of AP oa-s - 2 KS-S KS-S
5 Ketoreductase: ADP (nicotinamide adenine diphosphate phosphate) is a nucleophilic hydride ( ) donor (reducing agent) 2 P P P ADP = Ketoreductase ADP Dehydratase (D): Dehydratase - 2 Enoyl Reductase (ER) ADP Enoyl Reductase 288 Iterative two-carbon chain extension KS-S (ysteine of Ketosynthase) S-KS AP-S S-oA Malonyl oa AP-S oa-s S-KS KS - 2 KR ADP D - 2 ER ADP KS S-KS KS - 2 KR ADP D - 2 ER ADP KS S-KS
6 Thioesterase TE 2 18 Fatty acid (steric acid) S-oA R R oa-s 26.4 Phospholipids. 2 2 R 1 SoA -2 2 P 3 2 P -2 3 R 2 SoA L-glycerol-3- phosphate R 2 R 2 2 R 1 2 P -2 3 phosphatidic acid R 3 2 R 1 2 P R 3 phosphoglycerodes 2 - P 4-3 R 2 R Diacylglycerol 2 R 1 R 3 SoA 2 R 1 R 3 Triacylglycerol 290 Glycerophospholipids are important components of cell membranes. onpolar tails aggregate in the center of a bilayer ionic head is exposed to solvent ell membranes are ~5 nm thick unsaturatedr 2 2 P R saturated 2 2 ( 3 ) 3 Phosphatidylcholine (lechtins) 26.6: Waxes. esters of long chain fatty acids ( ) with long chain alcohols ( ) 3 ( 2 ) n 2 ( 2 ) n
7 26.6: Prostaglandins. (eicosanoids) 20 compounds derived from arachidonic acid and related fatty acids hormone: (Greek, horman, to set in motion) chemical messengers from one cell to another, that acts as a signal for a biochemical event. phospholipid 2 Arachidonic acid Prostaglandin F 2! (PGF 2! ) 2 prostaglandin endoperoxide reductase prostaglandin endoperoxide D isomerase prostaglandin (PG) synthase, 2 2 Prostaglandin 2 (PG 2 ) 2 prostacyclin (PGI) synthase prostaglandin endoperoxide E isomerase 2 Prostaglandin I 2 (Prostacyclin) 2 2 Prostaglandin D 2 (PGE 2 ) Prostaglandin E 2 (PGE 2 ) 292 Prostaglandin biosynthesis 2 prostaglandin synthase 2 2 Arachidonic acid cyclooxygenase (X-1 or X-2) PGG 2 PG 2 X-2 cell membrane Tyr-385 Ser acetylsalicylic acid Ser-530 X covalently modified enzyme
8 X-1 is a constitutive enzyme that is expressed in virtually all mammalian cells X-2 is an inducible enzyme that is expressed as a results of a biochemical response; expressed in phagocytes (macrophages) as part of an inflammation response. SAIDs: non-steroidal anti-inflammatory drugs Aspirin, ibuprofren, and naproxen are non-selective inhibitors of X Ac 3 elebrex, vioxx, and brextra are selective inhibitors of X-2 (coxibs) S 2 S 2 S 2 F Thromboxanes: named for their role in thrombosis, the formation of a clot inside a blood vessel PG 2 thromboxane synthase Thromboxane A 2 Thromboxane B 2 Leukotrienes 2 Lipoxygenase 2 eme, Aracidonic acid Leukotriene A S 2 2 Leukotriene D
9 26.7: Terpenes: The Isoprene Rule. Isoprenoids- 10 (terpenes), 15 (sesquiterpenes) and 20 (diterpenes) plant; essential oils Ruzicka isoprene rule: terpenoids are derived from isoprene units ( 5 ) isoprene (2-methyl-1,3-butadiene) ()-arvone (caraway seeds) (-)-arvone (spearmint) ()-limonene (oranges) (-)-limonene (lemons) itral (lempon grass) amphour patchouli alcohol (patchouli oil)!-pinene "-pinene Grandisol 296 The precursor to 10 terpenoids (monoterpenes) is geraniol diphosphate (diphosphate), which consists of two 5 isoprene units that are joined head-to-tail head tail PP = P PP head - tail head - tail P 15 sesquiterpenoids are derived from farnesyl diphosphate, which consists of three 5 isoprene units that are joined head-to-tail 20 diterpenoids are derived from geranylgeranyl diphosphate, which consists of four 5 isoprene units that are joined head-to-tail PP PP
10 25 sesterpenoids are derived from geranylfarnesyl diphosphate, which consists of five 5 isoprene units that are joined head-to-tail 30 triterpenoids and steroids are derived from squalene, which consists of two 15 farnesyl units that are joined tail-to-tail PP 40 tetraterpenoids are derived from phytocene, which consists of two 20 geranylgeranyl units that are joined tail-to-tail cedrane Taxol Lanosterol !-carotene
11 26.8: Isopentyl Diphosphate: The Biological Isoprene Unit. Mevalonic acid is the biosynthetic precursor to the actual 5 isoprene units, which are isopentyl diphosphate (IPP, tail) and dimethylallyl diphosphate (DMAPP, head) 26.10: The Pathway from Acetate to Isopentenyl Diphosphate. Mevalonate Pathway B: 2 SoA acetyl oa acetoacetyl-oa acetyltransferase 2 SoA laisen condensation SoA Enzyme-ys-S 3 SoA 3 S-ys-Enzyme acetyl oa acetoacetyl oa B: 2 SoA acetyl oa MG-oA synthase B 2 SoA aldol condensation 3 2 SoA 3-ydroxy-3-methylglutaric acid (MG-oA) MG-oA reductase 2 ADP 3 2 Mevalonic acid SoA acetoacetyl oa 300 onversion of mevalonic acid to IPP and DMAPP ATP 3 2 Mevalonic acid AMP 3 P - P - - ATP ADP 3 P 3 2- P - P P 4 3- B: 3 P - P - - B: P - P - - rearrangment P - P - - isopentenyl-pp (IPP) dimethylallyl-pp (DMAPP) 26.9: arbon-arbon Bond Formation in Terpene Biosynthesis. onversion of IPP and DMAPP to geraniol-pp and farnesyl-pp electrophilic head group DMAPP PP - PP Mg 2 PP B: IPP PP nucleophilic tail group PP geranyl pyrophosphate ( 10 ) - PP Mg 2 PP PP electrophilic head group PP B: nucleophilic tail group farnesyl pyrophosphate ( 15 )
12 PP PP squalene synthase onversion of genanyl-pp to monoterpenes Limonene & α-terpineol PP PP PP - geranyl diphosphate neryl diphosphate = bond acts as a nucleophile 2 :B limonene α-terpineol : Steroids D A 8 B holesterol biosynthesis (mechanism 26.3, p. 1089) part a: the cyclization Squalene epoxidase eme, 2 holesterol ( ) Squalene ( ) A Squalene xide ( ) Squalene cyclase Protosterol cation
13 holesterol biosynthesis, part b: the 1,2-shifts Protosterol cation ( ) 1,2-hydride shift ,2-hydride shift ,2-methyl shift ,2-methyl shift B: Lanosterol ( ) Lanosterol ( ) holesterol ( ) : Vitamin D. (please read) sunlight 7-Dehydrocholesterol Vitamin D : Bile Acids. (please read) 26.14: orticosteroids. (please read) ortisone
14 26.15: Sex hormones. (please read) androgens (male) Testosterone Androsterone Androstenedione dianabol estrogens (female) Estrone Estradiol Progesterone Ethynylestradiol 26.16: arotenoids. (please read) derived from phytocene ( 40 ) Lycopene!-carotene
Naturally occurring compounds that are typically of low molecular weight and are hydrophobic (soluble in organic solvent).
 hapter 27. Biomolecules: Lipids aturally occurring compounds that are typically of low molecular weight and are hydrophobic (soluble in organic solvent). fatty acids and waxes essential oils many vitamins
hapter 27. Biomolecules: Lipids aturally occurring compounds that are typically of low molecular weight and are hydrophobic (soluble in organic solvent). fatty acids and waxes essential oils many vitamins
26.1 Acetyl Coenzyme A
 Chapter 26 Lipids Lipids Lipids are naturally occurring substances grouped together on the basis of a common property they they are more soluble in nonpolar solvents than in water. Some of the most important
Chapter 26 Lipids Lipids Lipids are naturally occurring substances grouped together on the basis of a common property they they are more soluble in nonpolar solvents than in water. Some of the most important
Practice Problems on Lipids
 Lipids.1 Practice Problems on Lipids 1. Which one of the following compounds has the structural pattern of the naturally occurring fats or oils? a) b) c) d) 2 2 2 2 2 2 e) 2 2 2. Which of the following
Lipids.1 Practice Problems on Lipids 1. Which one of the following compounds has the structural pattern of the naturally occurring fats or oils? a) b) c) d) 2 2 2 2 2 2 e) 2 2 2. Which of the following
4,5,9/99 Neuman Chapter 21
 21: Lipids Structures of Lipids Biosynthesis of Lipids Preview Lipids are biological molecules soluble in organic solvents such as alcohols and ethers. They include fats, oils, waxes, terpenes, steroids,
21: Lipids Structures of Lipids Biosynthesis of Lipids Preview Lipids are biological molecules soluble in organic solvents such as alcohols and ethers. They include fats, oils, waxes, terpenes, steroids,
OBJECTIVE. Lipids are largely hydrocarbon derivatives and thus represent
 Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
CHAPTER 28 LIPIDS SOLUTIONS TO REVIEW QUESTIONS
 28 09/16/2013 17:44:40 Page 415 APTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents
28 09/16/2013 17:44:40 Page 415 APTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents
Chapter 26 Biochemistry 5th edition. phospholipids. Sphingolipids. Cholesterol. db=books&itool=toolbar
 http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
Functions of Lipids. - Storage Fats are long term energy (9 kcal/g) while carbohydrates are quick energy (4 kcal/g).
 Chapter 8: Lipids Functions of Lipids - Storage Fats are long term energy (9 kcal/g) while carbohydrates are quick energy (4 kcal/g). - Membrane Components Lipid barriers keep water out. - Messengers Hormones
Chapter 8: Lipids Functions of Lipids - Storage Fats are long term energy (9 kcal/g) while carbohydrates are quick energy (4 kcal/g). - Membrane Components Lipid barriers keep water out. - Messengers Hormones
CHAPTER 28 LIPIDS SOLUTIONS TO REVIEW QUESTIONS
 HAPTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents and their insolubility
HAPTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents and their insolubility
BCM 221 LECTURES OJEMEKELE O.
 BCM 221 LECTURES BY OJEMEKELE O. OUTLINE INTRODUCTION TO LIPID CHEMISTRY STORAGE OF ENERGY IN ADIPOCYTES MOBILIZATION OF ENERGY STORES IN ADIPOCYTES KETONE BODIES AND KETOSIS PYRUVATE DEHYDROGENASE COMPLEX
BCM 221 LECTURES BY OJEMEKELE O. OUTLINE INTRODUCTION TO LIPID CHEMISTRY STORAGE OF ENERGY IN ADIPOCYTES MOBILIZATION OF ENERGY STORES IN ADIPOCYTES KETONE BODIES AND KETOSIS PYRUVATE DEHYDROGENASE COMPLEX
Biological role of lipids
 Lipids Lipids Organic compounds present in living organisms, insoluble in water but able to be extracted by organic solvents such as: chloroform, acetone, benzene. Extraction = the action of taking out
Lipids Lipids Organic compounds present in living organisms, insoluble in water but able to be extracted by organic solvents such as: chloroform, acetone, benzene. Extraction = the action of taking out
Biomolecules Lecture Carbohydrates Lipids Proteins Nucleic Acids
 www.aiouinfo.com Biomolecules Lecture Carbohydrates Lipids Proteins Nucleic Acids Carbohydrates Monosaccharides 4 Examples: 1. Glucose 2. Fructose 3. Ribose 4. Deoxyribose Glucose Structure Function of
www.aiouinfo.com Biomolecules Lecture Carbohydrates Lipids Proteins Nucleic Acids Carbohydrates Monosaccharides 4 Examples: 1. Glucose 2. Fructose 3. Ribose 4. Deoxyribose Glucose Structure Function of
Chapter 8. Functions of Lipids. Structural Nature of Lipids. BCH 4053 Spring 2001 Chapter 8 Lecture Notes. Slide 1. Slide 2.
 BCH 4053 Spring 2001 Chapter 8 Lecture Notes 1 Chapter 8 Lipids 2 Functions of Lipids Energy Storage Thermal Insulation Structural Components of Membranes Protective Coatings of Plants and Insects Hormonal
BCH 4053 Spring 2001 Chapter 8 Lecture Notes 1 Chapter 8 Lipids 2 Functions of Lipids Energy Storage Thermal Insulation Structural Components of Membranes Protective Coatings of Plants and Insects Hormonal
Classification, functions and structure
 Classification, functions and structure Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine and Pharmacy "Nicolae Testemitanu" Lipids
Classification, functions and structure Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine and Pharmacy "Nicolae Testemitanu" Lipids
4,5,9/99 Neuman Chapter 21. Chapter 21 Lipids
 Chapter 21 Lipids from Organic Chemistry by Robert C. Neuman, Jr. Professor of Chemistry, emeritus University of California, Riverside orgchembyneuman@yahoo.com
Chapter 21 Lipids from Organic Chemistry by Robert C. Neuman, Jr. Professor of Chemistry, emeritus University of California, Riverside orgchembyneuman@yahoo.com
Lipid Metabolism. Catabolism Overview
 Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
CH 3. Lipids CHAPTER SUMMARY
 H 3 C H 3 C 15 H 3 C H Views of Cholesterol APTER SUMMARY 15.1 The Nature of can best be defined as biomolecules which are soluble to a great extent in solvents. In contrast to carbohydrates, proteins
H 3 C H 3 C 15 H 3 C H Views of Cholesterol APTER SUMMARY 15.1 The Nature of can best be defined as biomolecules which are soluble to a great extent in solvents. In contrast to carbohydrates, proteins
15.1 Lipids 15.2 Fatty Acids. Copyright 2009 by Pearson Education, Inc.
 Chapter 15 Lipids 15.1 Lipids 15.2 Fatty Acids Copyright 2009 by Pearson Education, Inc. 1 Lipids Lipids are biomolecules that contain fatty acids or a steroid nucleus. soluble in organic solvents, but
Chapter 15 Lipids 15.1 Lipids 15.2 Fatty Acids Copyright 2009 by Pearson Education, Inc. 1 Lipids Lipids are biomolecules that contain fatty acids or a steroid nucleus. soluble in organic solvents, but
Chapter 20 Lipids. Organic and Biochem
 Chapter 20 Lipids rganic and Biochem 20.1 Introduction Found in living organisms Insoluble in water but Soluble in non-polar substances Example of Lipid Solvent: diethyl ether Polar groups in lipids are
Chapter 20 Lipids rganic and Biochem 20.1 Introduction Found in living organisms Insoluble in water but Soluble in non-polar substances Example of Lipid Solvent: diethyl ether Polar groups in lipids are
Chapter 28: Lipids In a slightly different order than in your textbook Fatty Acids
 common name hapter 28: Lipids In a slightly different order than in your textbook Fatty Acids shorthand notations #of : # of = #of, location of = line structure comments butyric acid 4:0 in butterfat lauric
common name hapter 28: Lipids In a slightly different order than in your textbook Fatty Acids shorthand notations #of : # of = #of, location of = line structure comments butyric acid 4:0 in butterfat lauric
Introduction to the Study of Lipids
 Introduction to the Study of Lipids Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General
Introduction to the Study of Lipids Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General
Lecture 16. Finish lipid metabolism (Triglycerides, Isoprenoids/Steroids, Glyoxylate cycle) Amino acid metabolism (Urea cycle) Google Man III
 Lecture 16 Finish lipid metabolism (Triglycerides, Isoprenoids/Steroids, Glyoxylate cycle) Amino acid metabolism (Urea cycle) Google Man III The Powertrain of Human Metabolism (verview) CARBHYDRATES PRTEINS
Lecture 16 Finish lipid metabolism (Triglycerides, Isoprenoids/Steroids, Glyoxylate cycle) Amino acid metabolism (Urea cycle) Google Man III The Powertrain of Human Metabolism (verview) CARBHYDRATES PRTEINS
FAD FADH2. glycerol-3- phosphate. dehydrogenase. This DHAP is metabolically no different from that produced in glycolysis.
 1 Lipid Metabolism: ow that we are aware of the types of lipids in our bodies, it is important to see how we make them or break them. We will start our discussion with triacylglyceride degradation, and
1 Lipid Metabolism: ow that we are aware of the types of lipids in our bodies, it is important to see how we make them or break them. We will start our discussion with triacylglyceride degradation, and
BIOB111_CHBIO - Tutorial activity for Session 12
 BIOB111_CHBIO - Tutorial activity for Session 12 General topic for week 6 Session 12 Lipids Useful Links: 1. Animations on Cholesterol (its synthesis, lifestyle factors, LDL) http://www.wiley.com/college/boyer/0470003790/animations/cholesterol/cholesterol.htm
BIOB111_CHBIO - Tutorial activity for Session 12 General topic for week 6 Session 12 Lipids Useful Links: 1. Animations on Cholesterol (its synthesis, lifestyle factors, LDL) http://www.wiley.com/college/boyer/0470003790/animations/cholesterol/cholesterol.htm
MCQS ON LIPIDS. Dr. RUCHIKA YADU
 MCQS ON LIPIDS Dr. RUCHIKA YADU Q1. THE FATS AND OILS ARE RESPECTIVELY RICH IN a) Unsaturated fatty acids b) Saturated fatty acids c) Saturated and unsaturated fatty acids d) None of these Q2. ESSENTIAL
MCQS ON LIPIDS Dr. RUCHIKA YADU Q1. THE FATS AND OILS ARE RESPECTIVELY RICH IN a) Unsaturated fatty acids b) Saturated fatty acids c) Saturated and unsaturated fatty acids d) None of these Q2. ESSENTIAL
Definition: Water insoluble No common structure (though generally large R groups)
 Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Biosynthesis of Fatty Acids
 Biosynthesis of Fatty Acids Fatty acid biosynthesis takes place in the cytosol rather than the mitochondria and requires a different activation mechanism and different enzymes and coenzymes than fatty
Biosynthesis of Fatty Acids Fatty acid biosynthesis takes place in the cytosol rather than the mitochondria and requires a different activation mechanism and different enzymes and coenzymes than fatty
Lipids and Fatty Acids
 Lipids and Fatty Acids Objectives: 1. What are Lipids? properties glycerolipids vs. isoprenoids glycerolipid structure glycerolipid nomenclature 2. Fatty acid biosynthesis ellular localization Substrate
Lipids and Fatty Acids Objectives: 1. What are Lipids? properties glycerolipids vs. isoprenoids glycerolipid structure glycerolipid nomenclature 2. Fatty acid biosynthesis ellular localization Substrate
Lipids Definition. Definition: Water insoluble No common structure (though generally large R groups)
 Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Factors to Consider in the Study of Biomolecules
 Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General structure and functional groups
Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General structure and functional groups
Chemistry 506: Allied Health Chemistry 2. Chapter 17: Lipids. Biochemical Esters and Hydrocarbons
 hemistry 506 Dr. unter s lass hapter 17. hemistry 506: Allied ealth hemistry 2 1 hapter 17: Lipids Biochemical Esters and ydrocarbons Introduction to General, rganic & Biochemistry, 5 th Edition by Bettelheim
hemistry 506 Dr. unter s lass hapter 17. hemistry 506: Allied ealth hemistry 2 1 hapter 17: Lipids Biochemical Esters and ydrocarbons Introduction to General, rganic & Biochemistry, 5 th Edition by Bettelheim
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Lipids (cholesterol, cholesterol esters, phospholipids & triacylglycerols) combined with proteins (apolipoprotein) in
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Lipids (cholesterol, cholesterol esters, phospholipids & triacylglycerols) combined with proteins (apolipoprotein) in
Chapter 11: Lipids. Voet & Voet: Pages
 Chapter 11: Lipids Voet & Voet: Pages 380-394 Slide 1 Lipids Lipids are distinguished by their high solubility in non polar solvents and low solubility in H2O Diverse group of compounds including Fats,
Chapter 11: Lipids Voet & Voet: Pages 380-394 Slide 1 Lipids Lipids are distinguished by their high solubility in non polar solvents and low solubility in H2O Diverse group of compounds including Fats,
Lipids and Classification:
 Lipids and Classification: Lipids: Biological lipids are a chemically diverse group of organic compounds which are insoluble or only poorly soluble in water. They are readily soluble in non-polar solvents
Lipids and Classification: Lipids: Biological lipids are a chemically diverse group of organic compounds which are insoluble or only poorly soluble in water. They are readily soluble in non-polar solvents
Lipids and Membranes
 Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Lipids and Membranes I. overview Lipids are related
Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Lipids and Membranes I. overview Lipids are related
Chemistry 1506: Allied Health Chemistry 2. Section 8: Lipids. Biochemical Esters and Hydrocarbons. Outline
 hemistry 1506 Dr. unter s lass Section 8 Notes - Page 1/21 hemistry 1506: Allied ealth hemistry 2 Section 8: Lipids Biochemical Esters and ydrocarbons utline SETIN 8.1 INTRDUTIN...2 SETIN SETIN SETIN 8.2
hemistry 1506 Dr. unter s lass Section 8 Notes - Page 1/21 hemistry 1506: Allied ealth hemistry 2 Section 8: Lipids Biochemical Esters and ydrocarbons utline SETIN 8.1 INTRDUTIN...2 SETIN SETIN SETIN 8.2
Organic molecules highly hydrophobic and water insoluble.
 UNIT 5. LIPIDS OUTLINE 5.1. Introduction. 5.2. Fatty acids. 5.3. Eicosanoids. 5.4. Triacylglycerols = Triglycerides. 5.5. Waxes. 5.6. Membrane lipids: glycerophospholipids and sphingolipids. 5.7. Isoprenoids
UNIT 5. LIPIDS OUTLINE 5.1. Introduction. 5.2. Fatty acids. 5.3. Eicosanoids. 5.4. Triacylglycerols = Triglycerides. 5.5. Waxes. 5.6. Membrane lipids: glycerophospholipids and sphingolipids. 5.7. Isoprenoids
Chem 5 PAL Worksheet Lipids Smith text Chapter 15
 Chem 5 PAL Worksheet Lipids Smith text Chapter 15 Principle: Fatty acids are carboxylic acids with long (usually > 14) carbon chains which can be saturated (no carbon-carbon double bonds) are unsaturated
Chem 5 PAL Worksheet Lipids Smith text Chapter 15 Principle: Fatty acids are carboxylic acids with long (usually > 14) carbon chains which can be saturated (no carbon-carbon double bonds) are unsaturated
ANSC/NUTR 618 Lipids & Lipid Metabolism
 I. Overall concepts A. Definitions ANC/NUTR 618 Lipids & Lipid Metabolism 1. De novo synthesis = synthesis from non-fatty acid precursors a. Carbohydrate precursors (glucose, lactate, and pyruvate) b.
I. Overall concepts A. Definitions ANC/NUTR 618 Lipids & Lipid Metabolism 1. De novo synthesis = synthesis from non-fatty acid precursors a. Carbohydrate precursors (glucose, lactate, and pyruvate) b.
Chemistry Chapter 21
 Chemistry 2100 Chapter 21 Lipids Fa3y Acids CH oleic acid (mp 4 C) CH stearic acid (mp 70 C) Triacylglycerols Fatty Acids! The fatty acid components of triglycerides have certain things in common: 1.
Chemistry 2100 Chapter 21 Lipids Fa3y Acids CH oleic acid (mp 4 C) CH stearic acid (mp 70 C) Triacylglycerols Fatty Acids! The fatty acid components of triglycerides have certain things in common: 1.
Test Bank for Lehninger Principles of Biochemistry 5th Edition by Nelson
 Test Bank for Lehninger Principles of Biochemistry 5th Edition by Nelson Link download full: http://testbankair.com/download/test-bank-forlehninger-principles-of-biochemistry-5th-edition-by-nelson/ Chapter
Test Bank for Lehninger Principles of Biochemistry 5th Edition by Nelson Link download full: http://testbankair.com/download/test-bank-forlehninger-principles-of-biochemistry-5th-edition-by-nelson/ Chapter
Ahmad O. Olimat. Abdallah Al-Qawasmeh. Dr.Mamoun
 10 Ahmad O. Olimat Abdallah Al-Qawasmeh Mohammed Yousef Dr.Mamoun A QUICK RECAP Eicosanoids They are derived from Arachidonic acid, a fatty acid that contains 20 carbon atoms and four double bonds. They
10 Ahmad O. Olimat Abdallah Al-Qawasmeh Mohammed Yousef Dr.Mamoun A QUICK RECAP Eicosanoids They are derived from Arachidonic acid, a fatty acid that contains 20 carbon atoms and four double bonds. They
BIOCHEMISTRY. How Are Macromolecules Formed? Dehydration Synthesis or condensation reaction Polymers formed by combining monomers and removing water.
 BIOCHEMISTRY Organic compounds Compounds that contain carbon are called organic. Inorganic compounds do not contain carbon. Carbon has 4 electrons in outer shell. Carbon can form covalent bonds with as
BIOCHEMISTRY Organic compounds Compounds that contain carbon are called organic. Inorganic compounds do not contain carbon. Carbon has 4 electrons in outer shell. Carbon can form covalent bonds with as
BCH 3000 PRINCIPLES OF BIOCHEMISTRY
 BCH 3000 PRINCIPLES OF BIOCHEMISTRY (Semester 2-2012/13) 1 LIPID Learning outcome (Objectives) Function and distribution. Characteristics of fatty acids-structure and chemical properties. Saturated and
BCH 3000 PRINCIPLES OF BIOCHEMISTRY (Semester 2-2012/13) 1 LIPID Learning outcome (Objectives) Function and distribution. Characteristics of fatty acids-structure and chemical properties. Saturated and
Nebal Al - Gallab. Shatha Al - Jabri. Mamoon Ahram
 10 Nebal Al - Gallab Shatha Al - Jabri Mamoon Ahram Note: the doctor showed extra examples, they were in the slides, you can refer to them... Naming of Fatty Acids - 1 st Method ( IUPAC system ) We start
10 Nebal Al - Gallab Shatha Al - Jabri Mamoon Ahram Note: the doctor showed extra examples, they were in the slides, you can refer to them... Naming of Fatty Acids - 1 st Method ( IUPAC system ) We start
Lecture 16. Finish lipid metabolism (Triglycerides, Isoprenoids/Steroids, Glyoxylate cycle) Amino acid metabolism (Urea cycle) Google Man III
 Lecture 16 Finish lipid metabolism (Triglycerides, Isoprenoids/Steroids, Glyoxylate cycle) Amino acid metabolism (Urea cycle) Google Man III The Powertrain of Human Metabolism (verview) CARBHYDRATES PRTEINS
Lecture 16 Finish lipid metabolism (Triglycerides, Isoprenoids/Steroids, Glyoxylate cycle) Amino acid metabolism (Urea cycle) Google Man III The Powertrain of Human Metabolism (verview) CARBHYDRATES PRTEINS
I. Structure and Properties of Lipids
 I. Structure and Properties of Lipids Lipids: A diverse group of compounds characterized by their low solubility in water and a high solubility in organic solvents such as chloroform and methanol. Nonpolar
I. Structure and Properties of Lipids Lipids: A diverse group of compounds characterized by their low solubility in water and a high solubility in organic solvents such as chloroform and methanol. Nonpolar
ORGANIC CHEMISTRY. Chapter Steroids. Department of Chemistry, Xiamen University
 23.4 Steroids 23.4A Structures and Systematic Nomenclature of Steroids Steroids are derivatives of the following perhydrocyclopentanophenanthrene ring system. The four rings are designated A, B, C, and
23.4 Steroids 23.4A Structures and Systematic Nomenclature of Steroids Steroids are derivatives of the following perhydrocyclopentanophenanthrene ring system. The four rings are designated A, B, C, and
Biosynthesis of Fatty Acids
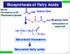 Biosynthesis of Fatty Acids CH 511 ource b-oxidation of FA Glycolysis of glucose n H CoA Acetyl CoA CoA Malonyl CoA Carboxylation of acetyl CoA H 3 C CH 2 CH 2 n-1 CoA aturated thioesters aturated fatty
Biosynthesis of Fatty Acids CH 511 ource b-oxidation of FA Glycolysis of glucose n H CoA Acetyl CoA CoA Malonyl CoA Carboxylation of acetyl CoA H 3 C CH 2 CH 2 n-1 CoA aturated thioesters aturated fatty
3.1.3 Lipids. Source: AQA Spec
 alevelbiology.co.uk SPECIFICATION Triglycerides and phospholipids are two groups of lipid. Triglycerides are formed by the condensation of one molecule of glycerol and three molecules of fatty acid. A
alevelbiology.co.uk SPECIFICATION Triglycerides and phospholipids are two groups of lipid. Triglycerides are formed by the condensation of one molecule of glycerol and three molecules of fatty acid. A
Organism. Organ and organ systems. Tissue. Cells. Carbohydrates Proteins Lipids Nucleic Acids
 hemistry of Life rganism rgan and organ systems Tissue ells arbohydrates Proteins Lipids Nucleic Acids Hydrocarbons, Alcohols, Phenols, Ethers, Thiols, Aldehydes, Ketones, arboxylic Acids, Esters, Amines,
hemistry of Life rganism rgan and organ systems Tissue ells arbohydrates Proteins Lipids Nucleic Acids Hydrocarbons, Alcohols, Phenols, Ethers, Thiols, Aldehydes, Ketones, arboxylic Acids, Esters, Amines,
Lipids. OpenStax College
 OpenStax-CNX module: m44401 1 Lipids OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be able
OpenStax-CNX module: m44401 1 Lipids OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be able
Biosynthesis of Fatty Acids. By Dr.QUTAIBA A. QASIM
 Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
LIPID METABOLISM
 LIPID METABOLISM LIPOGENESIS LIPOGENESIS LIPOGENESIS FATTY ACID SYNTHESIS DE NOVO FFA in the blood come from :- (a) Dietary fat (b) Dietary carbohydrate/protein in excess of need FA TAG Site of synthesis:-
LIPID METABOLISM LIPOGENESIS LIPOGENESIS LIPOGENESIS FATTY ACID SYNTHESIS DE NOVO FFA in the blood come from :- (a) Dietary fat (b) Dietary carbohydrate/protein in excess of need FA TAG Site of synthesis:-
Biomolecules. Biomolecules. Carbohydrates. Biol 219 Lec 3 Fall Polysaccharides. Function: Glucose storage Fig. 2.2
 Biomolecules Biomolecules Monomers Polymers Carbohydrates monosaccharides polysaccharides fatty acids triglycerides Proteins amino acids polypeptides Nucleic Acids nucleotides DNA, RNA Carbohydrates Carbohydrates
Biomolecules Biomolecules Monomers Polymers Carbohydrates monosaccharides polysaccharides fatty acids triglycerides Proteins amino acids polypeptides Nucleic Acids nucleotides DNA, RNA Carbohydrates Carbohydrates
Lipids fatty, oily, or waxy hydrophobic organic compounds.
 Lipids Lipids Lipids fatty, oily, or waxy hydrophobic organic compounds. u long hydrocarbon chain u composed of CHO Diverse group u fats u oils u waxes u steroids Do not form polymers u big molecules made
Lipids Lipids Lipids fatty, oily, or waxy hydrophobic organic compounds. u long hydrocarbon chain u composed of CHO Diverse group u fats u oils u waxes u steroids Do not form polymers u big molecules made
Lipids, Biological Membranes and Cellular Transport. 阮雪芬 May/9/2004
 Lipids, Biological Membranes and Cellular Transport 阮雪芬 May/9/2004 Outline Introduction Fatty Acids Triacylglycerols Polar lipids Steroids and other lipids Biological membranes Membrane transport Examples
Lipids, Biological Membranes and Cellular Transport 阮雪芬 May/9/2004 Outline Introduction Fatty Acids Triacylglycerols Polar lipids Steroids and other lipids Biological membranes Membrane transport Examples
Chapter 19 Lecture Outline
 Chapter 19 Lecture utline Prepared by Andrea D. Leonard University of Louisiana at Lafayette Lipids! Introduction to Lipids! Lipids are biomolecules that are soluble in organic solvents and insoluble in
Chapter 19 Lecture utline Prepared by Andrea D. Leonard University of Louisiana at Lafayette Lipids! Introduction to Lipids! Lipids are biomolecules that are soluble in organic solvents and insoluble in
Chapter 10 Homework Assignment
 hapter 10 Homework Assignment I have decided to alter the homework assignment for hapter 10. The following problems will be due once we finish the chapter: 2, 4, 5, 6, 9 Lipids are Good (and not always
hapter 10 Homework Assignment I have decided to alter the homework assignment for hapter 10. The following problems will be due once we finish the chapter: 2, 4, 5, 6, 9 Lipids are Good (and not always
TEST BANK FOR LEHNINGER PRINCIPLES OF BIOCHEMISTRY 6TH EDITION BY NELSON
 Link full download: https://testbankservice.com/download/testbank-for-lehninger-principles-of-biochemistry-6th-edition-bynelson TEST BANK FOR LEHNINGER PRINCIPLES OF BIOCHEMISTRY 6TH EDITION BY NELSON
Link full download: https://testbankservice.com/download/testbank-for-lehninger-principles-of-biochemistry-6th-edition-bynelson TEST BANK FOR LEHNINGER PRINCIPLES OF BIOCHEMISTRY 6TH EDITION BY NELSON
GUTS Lecture Syllabus for Lipid Structure and Nomenclature
 GUTS Lecture Syllabus for Lipid Structure and Nomenclature For Questions or Assistance contact: Dr. Gwen Sancar, gsancar@ad.unc.edu Learning bjectives After completing the GUTS lecture and associated self-
GUTS Lecture Syllabus for Lipid Structure and Nomenclature For Questions or Assistance contact: Dr. Gwen Sancar, gsancar@ad.unc.edu Learning bjectives After completing the GUTS lecture and associated self-
Carbohydrates, Lipids, Proteins, and Nucleic Acids
 Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
BIOSYNTHESIS OF FATTY ACIDS. doc. Ing. Zenóbia Chavková, CSc.
 BIOSYNTHESIS OF FATTY ACIDS doc. Ing. Zenóbia Chavková, CSc. The pathway for the of FAs is not the reversal of the oxidation pathway Both pathways are separated within different cellular compartments In
BIOSYNTHESIS OF FATTY ACIDS doc. Ing. Zenóbia Chavková, CSc. The pathway for the of FAs is not the reversal of the oxidation pathway Both pathways are separated within different cellular compartments In
Experiment 12 Lipids. Structures of Common Fatty Acids Name Number of carbons
 Experiment 12 Lipids Lipids are a class of biological molecules that are insoluble in water and soluble in nonpolar solvents. There are many different categories of lipids and each category has different
Experiment 12 Lipids Lipids are a class of biological molecules that are insoluble in water and soluble in nonpolar solvents. There are many different categories of lipids and each category has different
Review Session 1. Control Systems and Homeostasis. Figure 1.8 A simple control system. Biol 219 Review Sessiono 1 Fall 2016
 Control Systems and Homeostasis Review Session 1 Regulated variables are kept within normal range by control mechanisms Keeps near set point, or optimum value Control systems local and reflex Input signal
Control Systems and Homeostasis Review Session 1 Regulated variables are kept within normal range by control mechanisms Keeps near set point, or optimum value Control systems local and reflex Input signal
Good Afternoon! 11/30/18
 Good Afternoon! 11/30/18 1. The term polar refers to a molecule that. A. Is cold B. Has two of the same charges C. Has two opposing charges D. Contains a hydrogen bond 2. Electrons on a water molecule
Good Afternoon! 11/30/18 1. The term polar refers to a molecule that. A. Is cold B. Has two of the same charges C. Has two opposing charges D. Contains a hydrogen bond 2. Electrons on a water molecule
BY: RASAQ NURUDEEN OLAJIDE
 BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CLASSIFICATION OF LIPIDS PROPERTIES OF LIPIDS REACTIONS OF LIPIDS (CHEMICAL PROPERTIES) SOME QUANTITATIVE TESTS FOR LIPIDS CHEMISTRY AND PROPERTIES
BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CLASSIFICATION OF LIPIDS PROPERTIES OF LIPIDS REACTIONS OF LIPIDS (CHEMICAL PROPERTIES) SOME QUANTITATIVE TESTS FOR LIPIDS CHEMISTRY AND PROPERTIES
Chemistry B11 Chapters 15 Lipids
 Chapters 15 ipids ipids: are family of biomolecules that have the common property of being soluble in organic solvents but not in water. Role of lipids: they have three important roles in nature: 1. They
Chapters 15 ipids ipids: are family of biomolecules that have the common property of being soluble in organic solvents but not in water. Role of lipids: they have three important roles in nature: 1. They
Lipid Biosynthesis. Craig Wheelock February 2nd, 2009 Questions? Comments?
 Lipid Biosynthesis Craig Wheelock February 2nd, 2009 Questions? Comments? craig.wheelock@ki.se Why do we care about lipids? ~80% of European population overweight ~ 1/3 obese =130 million obese adults
Lipid Biosynthesis Craig Wheelock February 2nd, 2009 Questions? Comments? craig.wheelock@ki.se Why do we care about lipids? ~80% of European population overweight ~ 1/3 obese =130 million obese adults
Fatty acid breakdown
 Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
FATTY ACID SYNTHESIS
 FATTY ACID SYNTHESIS Malonyl- CoA inhibits Carni1ne Palmitoyl Transferase I. Malonyl- CoA is a precursor for fa=y acid synthesis. Malonyl- CoA is produced from acetyl- CoA by the enzyme Acetyl- CoA Carboxylase.
FATTY ACID SYNTHESIS Malonyl- CoA inhibits Carni1ne Palmitoyl Transferase I. Malonyl- CoA is a precursor for fa=y acid synthesis. Malonyl- CoA is produced from acetyl- CoA by the enzyme Acetyl- CoA Carboxylase.
Chapter 19: Lipid Metabolism
 1 hapter 19: Lipid Metabolism Triacylglycerols = triglycerides = fats - Triacylglycerols are the major biological energy source, which are water insoluble. Glycogen is also biological energy source, but
1 hapter 19: Lipid Metabolism Triacylglycerols = triglycerides = fats - Triacylglycerols are the major biological energy source, which are water insoluble. Glycogen is also biological energy source, but
Lipids are used to store and excess energy from extra carbohydrates in animals
 Lipids Lipids are a major source of energy used by cells, however lipids are more difficult for your body to break down. They produce nearly twice the amount of energy than proteins or carbohydrates. Lipids
Lipids Lipids are a major source of energy used by cells, however lipids are more difficult for your body to break down. They produce nearly twice the amount of energy than proteins or carbohydrates. Lipids
WHAT IS A LIPID? OBJECTIVE The objective of this worksheet is to understand the structure and function of lipids
 WHAT IS A LIPID? OBJECTIVE The objective of this worksheet is to understand the structure and function of lipids PART A: Understanding Lipids Lipids are more commonly known as fats and include triglycerides,
WHAT IS A LIPID? OBJECTIVE The objective of this worksheet is to understand the structure and function of lipids PART A: Understanding Lipids Lipids are more commonly known as fats and include triglycerides,
A. Structure and Function 1. Carbon a. Forms four (4) covalent bonds linked together in chains or rings Forms skeleton of basic biochemicals b.
 Biochemistry 2 A. Structure and Function 1. arbon a. Forms four (4) covalent bonds linked together in chains or rings Forms skeleton of basic biochemicals b. in three dimensions (3D) Diagrams in 2D may
Biochemistry 2 A. Structure and Function 1. arbon a. Forms four (4) covalent bonds linked together in chains or rings Forms skeleton of basic biochemicals b. in three dimensions (3D) Diagrams in 2D may
By: Dr Hadi Mozafari 1
 Biological lipids are a chemically diverse group of compounds, the common and defining feature of which is their insolubility in water. By: Dr Hadi Mozafari 1 Fats and oils are the principal stored forms
Biological lipids are a chemically diverse group of compounds, the common and defining feature of which is their insolubility in water. By: Dr Hadi Mozafari 1 Fats and oils are the principal stored forms
Chapter 2 The Chemistry of Life Part 2
 Chapter 2 The Chemistry of Life Part 2 Carbohydrates are Polymers of Monosaccharides Three different ways to represent a monosaccharide Carbohydrates Carbohydrates are sugars and starches and provide
Chapter 2 The Chemistry of Life Part 2 Carbohydrates are Polymers of Monosaccharides Three different ways to represent a monosaccharide Carbohydrates Carbohydrates are sugars and starches and provide
Biosynthesis of Triacylglycerides (TG) in liver. Mobilization of stored fat and oxidation of fatty acids
 Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
Fatty Acid and Triacylglycerol Metabolism 1
 Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Seminar 6 Theoretical part
 Seminar 6 Theoretical part Lipids are a heterogeneous group of naturally occurring organic compounds, classified together on the basis of their common solubility properties. Lipids are insoluble in water,
Seminar 6 Theoretical part Lipids are a heterogeneous group of naturally occurring organic compounds, classified together on the basis of their common solubility properties. Lipids are insoluble in water,
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
LIPID METABOLISM. Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI
 LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
MEMBRANE LIPIDS I and II: GLYCEROPHOSPHOLIPIDS AND SPHINGOLIPIDS
 December 6, 2011 Lecturer: Eileen M. Lafer MEMBRANE LIPIDS I and II: GLYCEROPHOSPHOLIPIDS AND SPHINGOLIPIDS Reading: Stryer Edition 6: Chapter 26 Images: All images in these notes were taken from Lehninger,
December 6, 2011 Lecturer: Eileen M. Lafer MEMBRANE LIPIDS I and II: GLYCEROPHOSPHOLIPIDS AND SPHINGOLIPIDS Reading: Stryer Edition 6: Chapter 26 Images: All images in these notes were taken from Lehninger,
Biochemistry Worksheet
 Biology 138 Name Section 3.1 Properties of Water Biochemistry Worksheet 1. Why is water such an important molecule to living things? 2. Describe the chemical make up and type of bonding found in water
Biology 138 Name Section 3.1 Properties of Water Biochemistry Worksheet 1. Why is water such an important molecule to living things? 2. Describe the chemical make up and type of bonding found in water
Lipids. Chapter 11: Outline. Fatty Acids. Lipid Classes. Fatty Acids-2. Fatty Acids-3. General Types eg. A fatty acid
 Chapter 11: utline Lipid Classes Fatty Acids and Derivatives Triacylglycerols Phospholipids Isoprenoids Membranes Membrane Structure Membrane Function Wax Esters Sphingolipids Lipoproteins Lipids General
Chapter 11: utline Lipid Classes Fatty Acids and Derivatives Triacylglycerols Phospholipids Isoprenoids Membranes Membrane Structure Membrane Function Wax Esters Sphingolipids Lipoproteins Lipids General
Fatty Acid and Triacylglycerol Metabolism 1
 Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Chapter 22, Fatty Acid Metabolism CH 3 (CH 2 ) 14 CO 2 R C C O2 CH 2 OH O R. Lipase + 3 H 2 O
 hapter 22, Fatty Acid Metabolism Pages: 603-613 I. Introduction - Fatty acids have 4 major physiological roles: - omponents of phospholipids and glycolipids (membranes) - Attachment to Proteins targets
hapter 22, Fatty Acid Metabolism Pages: 603-613 I. Introduction - Fatty acids have 4 major physiological roles: - omponents of phospholipids and glycolipids (membranes) - Attachment to Proteins targets
2.3 Carbon-Based Molecules CARBON BASED MOLECULES
 CARBON BASED MOLECULES KEY CONCEPTS Carbon-based molecules are the foundation of life. Lipids are one class of organic molecules. This group includes fats, oils, waxes, and steroids. Lipids are made of
CARBON BASED MOLECULES KEY CONCEPTS Carbon-based molecules are the foundation of life. Lipids are one class of organic molecules. This group includes fats, oils, waxes, and steroids. Lipids are made of
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM. Fatty Acid Elongation and Desaturation
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM I. Fatty acid elongation A. General 1. At least 60% of fatty acids in triacylglycerols are C18. 2. Free palmitic acid (16:0) synthesized in cytoplasm is elongated
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM I. Fatty acid elongation A. General 1. At least 60% of fatty acids in triacylglycerols are C18. 2. Free palmitic acid (16:0) synthesized in cytoplasm is elongated
Biomolecules: lipids
 Biomolecules: lipids Organic biomolecules: lipids Organic amphiphilic compounds insoluble in water Easily extracted from animal and vegetal cells using apolar solvents Fundamental to build cell's shape
Biomolecules: lipids Organic biomolecules: lipids Organic amphiphilic compounds insoluble in water Easily extracted from animal and vegetal cells using apolar solvents Fundamental to build cell's shape
Oxidative Phosphorylation
 Oxidative Phosphorylation Oxidative Phosphorylation - In Glycolysis and the citric acid cycle, we ve made a lot of reduced cofactors NADH and FADH 2 - In oxidative phosphorylation, we use the energy generated
Oxidative Phosphorylation Oxidative Phosphorylation - In Glycolysis and the citric acid cycle, we ve made a lot of reduced cofactors NADH and FADH 2 - In oxidative phosphorylation, we use the energy generated
Lipids. Types of lipids. Fatty acid structure. Lipid functions. Fatty acid structure
 Lipids Types of lipids lassification of Lipids Fatty Acids Glycerides Nonglycerol Lipids omplex Lipids Structure of Biological Membranes Fatty Acids Saturated Unsaturated omplex Lipids Lipoproteins Glycolipids
Lipids Types of lipids lassification of Lipids Fatty Acids Glycerides Nonglycerol Lipids omplex Lipids Structure of Biological Membranes Fatty Acids Saturated Unsaturated omplex Lipids Lipoproteins Glycolipids
3150:112 SAMPLE TEST 2. Print out a copy Answer the questions on your own. Check the answers at GOBC Ans.pdf. Good Luck!
 SAMPLE TEST 2 3150:112 Print out a copy Answer the questions on your own. Check the answers at GOBC Ans.pdf. Good Luck! QUESTIONS 1-3 REFER TO TE FOLLOWING: A. C 2 O O B. C 2 O O O C 2 O C. O C 2 O 1.
SAMPLE TEST 2 3150:112 Print out a copy Answer the questions on your own. Check the answers at GOBC Ans.pdf. Good Luck! QUESTIONS 1-3 REFER TO TE FOLLOWING: A. C 2 O O B. C 2 O O O C 2 O C. O C 2 O 1.
The Chemical Level of Organization
 2 The Chemical Level of Organization PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris Table 2-1 Principal Elements in the Human Body Table 2-1 Principal Elements
2 The Chemical Level of Organization PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris Table 2-1 Principal Elements in the Human Body Table 2-1 Principal Elements
Acid/Base chemistry. NESA Biochemistry Fall 2001 Review problems for the first exam. Complete the following sentences
 1 NESA Biochemistry Fall 2001 eview problems for the first exam Acid/Base chemistry 1. 2 3 is a weak acid. 2. The anion of a weak acid is a weak base 3. p is the measure of a solutions acidity. 4. 3 and
1 NESA Biochemistry Fall 2001 eview problems for the first exam Acid/Base chemistry 1. 2 3 is a weak acid. 2. The anion of a weak acid is a weak base 3. p is the measure of a solutions acidity. 4. 3 and
General Biochemistry-1 BCH 202
 General Biochemistry-1 BCH 202 1 I would like to acknowledge Dr. Farid Ataya for his valuable input & help in this course. 2 Outline Lipids Definition, function, fatty acids, classification: simple lipids:
General Biochemistry-1 BCH 202 1 I would like to acknowledge Dr. Farid Ataya for his valuable input & help in this course. 2 Outline Lipids Definition, function, fatty acids, classification: simple lipids:
Lipids. Polar bears have a large reserve of lipids.
 Chapter 28 Lipids Polar bears have a large reserve of lipids. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and Susan Arena Course 28.1 Lipids:
Chapter 28 Lipids Polar bears have a large reserve of lipids. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and Susan Arena Course 28.1 Lipids:
Biological Molecules
 The Chemical Building Blocks of Life Chapter 3 Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent bonds. Carbon may
The Chemical Building Blocks of Life Chapter 3 Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent bonds. Carbon may
