Sonic hedgehog Is Required Early in Pancreatic Islet Development
|
|
|
- Isaac Allen
- 6 years ago
- Views:
Transcription
1 Developmental Biology 244, (2002) doi: /dbio , available online at on Sonic hedgehog Is Required Early in Pancreatic Islet Development Philip J. diiorio,*,1,2 Jennifer B. Moss,*,1 Jennifer L. Sbrogna, Rolf O. Karlstrom, and Larry G. Moss*,,3 *Department of Medicine and Department of Physiology, Tufts University School of Medicine, Boston, Massachusetts 02111; and Department of Biology, Morrill Science Center, University of Massachusetts, Amherst, Massachusetts Pancreatic organogenesis relies on a complex interplay of cell-autonomous and extracellular signals. We demonstrate that the morphogen sonic hedgehog (Shh) is required for pancreatic development in zebrafish. Genetic mutants of Shh and its signaling pathway establish this dependence as specific to endocrine, but not exocrine, pancreas. Using cyclopamine to inhibit hedgehog signaling, we show that transient Shh signaling is necessary during gastrulation for subsequent differentiation of endoderm into islet tissue. A second hedgehog-dependent activity occurring later in development was also identified and may be analogous to the known action of Shh in gut endoderm to direct localization of pancreatic development. The early action of Shh may be part of a more general process allowing neuroendocrine cells to originate in nonneuroectodermally derived tissues Elsevier Science (USA) Key Words: pancreas; islet; zebrafish; hedgehog; insulin. INTRODUCTION The exocrine and endocrine cells of the pancreas arise from common precursors present in the early gut endoderm (Le Douarin, 1988; Percival and Slack, 1999). Studies in chick and mouse have begun to uncover a complex program dependent on numerous transcription (Edlund, 2001) and signaling factors (Kim and Hebrok, 2001) required for the formation of the pancreas and differentiation of its component cell types. FGF-mediated interactions with adjacent mesoderm direct early anteroposterior (A-P) patterning of the endoderm (Wells and Melton, 2000). Later, communication from the notochord and local mesenchyme dictate and refine subsequent organ-specific commitment (Kim et al., 1997a). The pancreatic diverticulum evaginates from the posterior foregut endoderm at a characteristic position, within a broader domain defined by the expression of a 1 These authors contributed equally to this work. 2 Present address: Diabetes Division, University of Massachusetts Medical School, Worcester, MA To whom correspondence should be addressed at Department of Physiology, Tufts University School of Medicine, 136 Harrison Avenue, Boston, MA Fax: (617) larry. moss@tufts.edu. homeodomain transcription factor, Pdx-1 (Idx-1, Ipf-1) (Ohlsson et al., 1993; Miller et al., 1994). The genetic ablation of Pdx-1 results in pancreatic agenesis, although early endocrine precursor cells do form (Ahlgren et al., 1996; Offield et al., 1996). While much is now known regarding later steps in pancreatic development, the early processes by which gut endoderm acquires competence to respond to later specific signals are not well understood. Sonic hedgehog (Shh) is a member of a family of secreted morphogens that has been demonstrated to play an important role in pancreatic development. The dorsal pancreatic bud forms within a gap in the endodermal expression of shh, within the larger domain of Pdx-1 expression (Bitgood and McMahon, 1995; Apelqvist et al., 1997). Pancreatic development in the adjacent shh-expressing endoderm may operate through the suppression of essential inductive signals, such as activin, from the overlying notochord (Kim et al., 1997a; Hebrok et al., 1998). For example, overexpression of Shh within the Pdx-1-expressing endoderm blocks pancreas formation, although differentiated exocrine and endocrine cells form and are mislocalized to the intestinal wall (Apelqvist et al., 1997). Inhibition of Shh signaling by the alkaloid cyclopamine in 10-somite chick embryos causes anterior and posterior expansion of pancreatic endoderm and a relative hypertro /02 $ Elsevier Science (USA) All rights reserved. 75
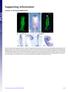
2 76 diiorio et al. FIG. 1. Disruption of Shh signaling alters pancreas development. (A) Homozygous 48-hpf INS-GFP transgenic zebrafish. Untreated embryos (left) contain a compact multicellular cluster of GFP-expressing cells. Addition of 50 M cyclopamine at 4 hpf (right) blocks formation of GFP islets. Some isolated, single GFP-expressing cells remain (arrows). (B) 48 hpf WT zebrafish in situ hybridized as whole-mount embryos with a proinsulin riboprobe depict normal clusters of insulin-expressing cells in the pancreas budding from the right side of the endoderm (left). 48 hpf Syu t4 / embryos have lost all insulin expression (middle) or exhibit expression (right) in isolated single cells (arrowheads). (C) Early insulin expression in a 24-hpf WT (left) and syu embryo (right). (D) Hybridization of WT embryos with a proglucagon probe (left) reveals cells in the perimeter of the islet as well as in an isolated cell within the gut at 48 hpf (arrow). No mrna for this neuroendocrine hormone is seen in syu embryos (right). (B D) Views are dorsal, with anterior to the left. phy of islet endocrine cell types (Kim et al., 1997b). In mice, two hedgehog (hh) genes, indian (ihh) and shh, are expressed in gut endoderm. Due to lethality before pancreas development, shh / ihh / mice are uninformative (Hebrok et al., 2000). However, the viable shh / ihh / mouse develops an annular pancreas and generates a relative excess of pancre-
3 Shh Promotes Zebrafish Pancreatic Islet Development 77 FIG. 2. Nature of the islet defect in the zebrafish mutant syu t4. The morphology of WT (A) vs syu embryos (B) was compared. Paraffin transverse cut sections (5 m), stained with hematoxylin and eosin. (A) WT islet (arrow). (B) The islet is selectively missing in syu embryos (arrow). (C) Carboxypeptidase A expression (arrow) in 72-hpf syu embryos embedded in paraffin, sectioned, and counterstained with Vector Green. (D I) Whole-mount in situ hybridization analysis of Wt and syu / embryos. Views are dorsal with anterior to the left, except (I), which is a lateral view. (D) Carboxypeptidase A expression in 48-hpf WT and syu / embryos. (E) Expression 72 hpf of transferrin (F) Pdx-1 expression in 48-hpf WT pancreas (arrow) and duodenum (arrowhead). (G) Isl-1 expression in 48-hpf syu / embryos. Arrows denote loss of expression. Foregut expression (arrowheads) is retained. (H) Loss of Beta2/NeuroD expression within the islet at 48 hpf (arrow) in syu / embryos. (I) Nkx2.2 (nk2.2) expression in WT 24-hpf embryos in early gut endoderm where the pancreatic diverticulum will form (arrow). FIG. 3. Insulin expression in hh signal transduction mutants. Insulin expression in 48-hpf WT (A), homozygous yot ty17a / (B) and smu b577 / embryos (C). Arrows denote the presence of insulin-labeled cells in the mutants (B, C). atic endocrine tissue (Hebrok et al., 2000). Thus, the established role of Shh signaling, when evaluated after the formation of the gut endoderm, appears to be inhibitory or restrictive to pancreas cell development. We used the transparent zebrafish embryo to address the possible role of Shh signaling at earlier stages of development. The zebrafish pancreas forms as a single dorsal evagination within a domain of Pdx-1-expressing endoderm located adjacent to the A-P boundary of somites 3 and 4 (Argenton et al., 1999). Islet precursor cells first express insulin at the 12-somite stage, h postfertilization (hpf) (Biemar et al., 2001). Clusters of insulin-positive cells subsequently increase in number, fuse into a single group by 24 hpf (30-somite stage), and condense into an islet surrounded by exocrine pancreatic tissue by 48 hpf (hatching stage) (Biemar et al., 2001). We generated a zebrafish transgenic line, in which insulin expression is represented by green fluorescent protein (GFP), and discovered that an inhibitor of hh signaling, cyclopamine, blocks formation of the pancreatic islet. A requirement for Shh in the pancreas was confirmed by analysis of zebrafish mutants that disrupt hh signaling. This novel requirement for shh early in embryogenesis may provide a clue to a long unanswered question of how islet endocrine cells, with their striking neuronal character, originate within the endoderm. MATERIALS AND METHODS GFP Transgenic Zebrafish Production and Imaging A 960-bp zebrafish insulin promoter was generated by PCR based on a published sequence (Milewski et al., 1998) and was cloned into a Tc3 expression vector (Raz et al., 1998) along with egfp (Clontech) to produce INS-GFP founders. Approximately 5 nl containing 20 ng/ml cesium-purified plasmid DNA and 50 ng/ml of capped Tc3A transposase mrna (mmessage mmachine; Ambion) was injected into one- to two-cell zebrafish embryos, generating singlecopy insertions. Embryos used in this study are generation 5 INS-GFP homozygotes. For fluorescence imaging, we used a Leica
4 78 diiorio et al. MZFI III dissecting microscope, a Hamamatsu Orca CCD camera, and Openlab software (Improvision) on a Macintosh G3 computer. Bright-field microscopy was recorded with a Zeiss Axiophot. Photoshop v.6 was used to prepare figures. Zebrafish Strains, Embryo Maintenance, and Cyclopamine Treatment Wildtype (WT) embryos were derived from the Oregon strain AB. Stages were assessed according to Kimmel et al. (1995) and are as follows: 4 hpf, blastula; 6 and 8 hpf, gastrula; 10 hpf, gastrulation complete; 12 hpf, 6 somites; 16 hpf, 14 somites; and 20 hpf, 24 somites. Syu t4 (Schauerte et al., 1998) is a deletion mutation in the shh gene, and Yot ty17a (Karlstrom et al., 1999) is a mutation in the gli-2 gene. Smu b577 (Barresi et al., 2000), a mutation in the smo gene, was a gift from S. Devoto. These homozygous lethal strains were maintained as heterozygotes and identified by their curved body axis. Homozygous INS-GFP embryos were grown at 28 C on agarosetreated dishes in embryo media and imaged (Westerfield, 1995). For cyclopamine treatment, chorions were removed by using pronase (Westerfield, 1995) and were grown on agarose-covered dishes. A 10 mm stock of cyclopamine (gift of W. Gaffield) was prepared in 95% ethanol and diluted to 50 M in embryo media. Histology and Whole-Mount in Situ Hybridization Five-micrometer paraffin sections were stained with hematoxylin and eosin. Whole-mount in situ hybridization was performed essentially as described (Oxtoby and Jowett, 1993) at 65 C, except for insulin (60 C). At 72 hpf, syu embryos were hybridized with carboxypeptidase A probe, embedded in paraffin, sectioned, and counterstained with vector green to indicate the presence of exocrine pancreas in the mutant. Insulin-hybridized mutant embryos were also sectioned (data not shown). Hybridization Probes Total RNA was prepared by using the Trizol method from adult pancreatic tissue, identifiable within the gut of INS-GFP zebrafish under the fluorescent dissecting microscope. Following reverse transcription using poly(dt) oligos and Superscript II RNA polymerase (Life Technologies), the cdna was used as a template to amplify pancreas-specific genes by using specific primers based on a published sequence. We generated PCR products for a 300-bp proinsulin (Milewski et al., 1998), a 650-bp proglucagon (Argenton et al., 1999), a 488-bp carboxypeptidase A, and a 540-bp Beta2/ NeuroD cdna (Liao et al., 1999) that were cloned into pcrii (Invitrogen). The inserts were sequenced and confirmed by using NCBI Blast ( Overexpression of Sonic Hedgehog mrna Capped shh mrna was synthesized from the expression plasmid pt7ts-shh by using the SP6 Message Machine (Ambion), and approximately 0.5 nl of 1 mg/ml solution was injected into yolks of embryos at the two- to four-cell stage. Injection of 0.5 nl of 1 mg/ml GFP mrna or -galactosidase ( -gal) mrna caused no detectable morphological abnormalities and served as a control for RNA toxicity. RESULTS Shh Is Required for Islet Formation An INS-GFP transgenic zebrafish line in which the zebrafish insulin promoter targets expression of GFP to the insulin-expressing cells of the islet was generated. GFP expression was detected at the 17-somite stage (18 hpf) in cells of the midline gut endoderm ventral to somite 4 (data not shown). By 48 hpf, the free-swimming zebrafish contains an islet (Biemar et al., 2001), represented by a discrete cluster of GFP-positive cells (Fig. 1A). Formation of these GFP-positive islets was blocked when 4-hpf transgenic embryos were exposed to cyclopamine (Fig. 1A), a potent inhibitor of hh signaling (Cooper et al., 1998; Incardona et al., 1998). The islet cells were either absent or detectable as one or a few isolated cells in the gut endoderm. Cyclopamine is a general inhibitor of hh action that interferes with smoothened, a plasma membrane protein that interacts with the hh receptor patched and is required for hh signal transduction (Taipale et al., 2000). To verify that the defects seen after cyclopamine treatment are indeed due to defects in Shh signaling, we examined insulin gene expression and islet development in sonic you (syu) embryos, which carry a loss-of-function mutation in the shh gene (Schauerte et al., 1998). As was seen after cyclopamine treatment, the insulin-expressing islet cell cluster fails to develop in syu embryos. When observed at 48 hpf, insulin expression is either completely eliminated (34%; 14/41 embryos) or reduced to one or more isolated single cells (66%; 27/41 embryos) (Fig. 1B). Disruption of normal islet development is also detected earlier at 24 hpf (Fig. 1C), indicating that the loss of insulin-expressing cells is not due to degeneration. Thus, we conclude that Shh signaling is a primary requirement for normal islet development. To determine whether islet cell types other than insulinexpressing cells are lost in syu / mutants, we analyzed the expression of glucagon, an endocrine hormone secreted by pancreatic cells. In 48-hpf WT embryos, glucagon expression was localized to cells which have acquired their characteristic location around the perimeter of the islet (Fig. 1D). No glucagon expression was observed in syu / (0/20) embryos, suggesting that the entire islet endocrine cell population was affected by the absence of sonic hedgehog. Blocking hh Signaling Specifically Disrupts Endocrine but Not Exocrine Islet Development Hematoxylin and eosin staining of paraffin sections shows that the 72-hpf WT pancreas consists of a single dorsal islet surrounded by basophilic exocrine tissue (Fig. 2A). In syu embryos, only basophilic tissue is seen in the expected location of the pancreas (Fig. 2B). This structure expresses carboxypeptidase A, a marker for exocrine pancreas (Fig. 2C), whereas insulin-expressing cells are not present (data not shown). Consistent with this observation, carboxypeptidase A expression in WT embryos encircles
5 Shh Promotes Zebrafish Pancreatic Islet Development 79 the islet and extends caudally along the gut to somite 9 (Fig. 2D, top). Carboxypeptidase A is also expressed in syu embryos (Fig. 2D, bottom), although expression is reduced in the anterior component of exocrine tissue, perhaps due to the absence of the normally underlying islet mass. The effects of Shh action on the liver, another organ derived from adjacent gut endoderm, were monitored by transferrin expression. No major effect resulting from the loss of Shh signaling on liver development was apparent in either WT or syu embryos at 72 hpf (Fig. 2E, top and bottom). Thus, the absence of Shh does not result in a generalized defect of gut endoderm patterning or differentiation, and the pancreatic defect is specific to the endocrine islet. We next evaluated the expression of transcription factors involved in pancreatic islet development in other organisms. Pdx-1 controls early pancreatic bud development and the ultimate morphogenesis of the pancreas but is not required for the emergence of early precursor cells in the gut endoderm (Ahlgren et al., 1998; Offield et al., 1996). In 48-hpf WT zebrafish, Pdx-1 is present in the duodenum and the entire pancreatic bud (Fig. 2F, top), which is predominately islet tissue at this stage (Biemar et al., 2001). In syu embryos, Pdx-1 expression is retained in the duodenum (Fig. 2F, bottom) but not in the bud (arrows). In addition, Isl-1 is required for the development in mouse of the entire dorsal, but not the ventral, pancreatic lobe as well as for all islet endocrine tissue (Ahlgren et al., 1997). Isl-1 is expressed in the pancreatic islet and in the gut immediately anterior to the pancreatic bud in WT zebrafish embryos (Fig. 2G, top). However, syu embryos lack islet expression of Isl-1, whereas it is retained in the adjacent gut endoderm (Fig. 2G, bottom). Another factor known to contribute to normal islet development is Beta2/NeuroD, a bhlh transcription factor expressed in early islet precursors and mature cells (Naya et al., 1997) that is expressed in the zebrafish islet (Fig 2h, top, arrow) yet is absent in the pancreas of 48-hpf syu embryos (Fig. 2H, bottom). Finally, we examined the expression of the homeodomain transcription factor Nkx2.2 (nk2.2), which is necessary for the establishment of insulin-expressing islet cells in mice (Sussel et al., 1998). Nkx2.2 is expressed within the developing pancreatic endoderm in 24-hpf WT embryos but not in syu embryos (Fig. 2I). The failure to detect any Nkx2.2 and Beta2/NeuroD expression in the syu gut endoderm suggests that the loss of hh signaling may selectively inhibit formation of early pancreatic endocrine precursors. Effects of Other hh Signaling Pathway Mutations on Pancreatic Islet Development Because additional hh family members, such as tiggywinkle and echidna hh, are expressed in zebrafish (Ekker et al., 1995; Norris et al., 2000), we analyzed mutations in downstream components of hh signaling. You-too (yot) mutations disrupt zebrafish gli-2, one of a family of transcription factors required for mediating hh action in the nucleus (Karlstrom et al., 1999). Compared with WT embryos (Fig. 3A), yot embryos (Fig. 3B) contain anterior, mislocalized insulin-expressing cells (arrow) that are reduced in number, indicating a role for gli genes in interpreting hh signals in developing islet cells. In contrast to syu embryos, insulin expression is retained in all yot mutants examined (22/22 embryos). Compensation by other gli family members may be responsible for the reduced severity of the phenotype compared with syu mutants. Therefore, we evaluated a mutation in slow muscle omitted (smu) embryos that contain a loss-of-function mutation in Smoothened, an extracellular membrane protein required for all hh signal transduction (Varga et al., 2001). Smu embryos exhibit a more profound disruption of development affecting additional aspects of embryogenesis than syu, such as ventral cyclopia (Barresi et al., 2000). However, the defect in insulin expression seen in smu embryos (Fig. 3C) is similar to that seen in syu embryos (Fig. 1B) in that insulin expression is either absent or found in single, isolated cells. Because the observed effect on insulin expression in yot, smu, or cyclopamine-treated embryos is no more severe than that in syu embryos, the effects on islet development appear to depend primarily on the action of Shh. However, subtle contributions from other hh family members during normal development cannot be ruled out. Overexpression of shh Expands Pancreatic Islet Development The inhibition in islet development caused by decreased hh signaling led us to examine whether overexpression of Shh protein early in development would affect islet phenotype. Injection of shh mrna is consistently associated with a dramatic increase in the number of insulinexpressing cells (Fig. 4; 151/154 embryos). Associated with this increase, the shh-injected embryos contain significant numbers of ectopic insulin-positive cells (35/154 embryos) extending anteriorly as far as somite 1 (Fig. 4F). In WT and control-injected embryos, occasional insulin-positive cells could be identified in the gut endoderm just anterior to the developing islet but within the normal domain of insulin expression (Fig. 4E; 2/56 embryos). In addition, both pancreatic and duodenal expression of Pdx-1 were increased in shh-injected embryos (48/51 embryos) when viewed at 48 hpf (Figs. 4G and 4H). Separate Early and Late Actions of hh Signaling on Islet Development To evaluate the stage-specific requirement for Shh action on the pancreas, we treated INS-GFP or WT embryos with cyclopamine at a series of early time points. The interruption of hh signaling on islet development when observed at 48 hpf for GFP fluorescence or in situ insulin expression is comparable (Figs. 5A and 5E; Table 1). When cyclopamine is administered at the beginning of gastrulation (6 hpf; shield stage), insulin expression is ablated or reduced to single-cell loci (Figs. 4B and 4F; Table 1). This phenotype is similar to
6 80 diiorio et al. FIG. 4. Insulin and Pdx-1 expression after shh overexpression in WT embryos. Left: mrna control-injected embryos. Right; shh mrna-injected embryos. After mrna injection at the two- to four-cell stage, embryos developed for 24 or 48 hpf and were subjected to whole-mount in situ analysis using the insulin or Pdx-1 probe. (A, C, E) Control GFP mrna-injected embryos hybridized at 24 hpf with the insulin probe. Positive cells are centered ventral to somite 4 (4). (B, D, F) Shh mrna-injected embryos hybridized with the insulin probe. Note the anterior extension of labeled cells. (G) Control -gal mrna-injected embryo hybridized with the Pdx-1 probe. (H) Shh mrna-injected embryo hybridized with the Pdx-1 probe. (A, B, E, F) Ventral views. (C, D) Lateral views. (G, H) Dorsal views. that observed in syu / embryos. When administered at 8 hpf (late gastrulation), insulin expression becomes detectable in an increasing percentage of embryos, and multicellular clusters are occasionally detected (Figs. 5C and 5G; Table 1). Treatment at 12 hpf (6-somite stage) permits insulin expression in all embryos, most of which form normal-sized, multicellular clusters (Figs. 5D and 5H; Table 1). At later time points (16 hpf, 14 somites; and 20 hpf, 24 somites), islet development appears to be unaffected by cyclopamine administration (Table 1). These data reveal a requirement during gastrulation for hh signaling in pancreatic islet development. Another consequence of cyclopamine treatment is the formation of multiple loci of insulin-positive cells at ectopic sites anterior to the normal islet location at the somite 3/4 boundary (Fig. 5; Table 1). Importantly, treatment at 12 hpf (6 somites) leads to normal-sized islet clusters, but the percentage of embryos with islet cells shifted to anterior positions is similar to that caused by treatment during gastrulation. This suggests the presence of two separate actions of shh during pancreas development. An early Shh activity appears to be required for the differentiation of endocrine cells and formation of endocrine islets. Without this early shh activity, only single-cell loci
7 Shh Promotes Zebrafish Pancreatic Islet Development 81 FIG. 5. Shh signaling is required early for patterning the endocrine islet. Living fifth-generation homozygous INS-GFP or WT fixed embryos imaged at 48 hpf untreated (A, E) or treated with 50 M cyclopamine at 6 (B, F), 8 (C, G), or 12 (D, H) hpf. Representative islet phenotypes are shown in living embryos as fluorescent (A D) or as bright-field images after fixation and in situ hybridization with insulin probe (E H). (A) 48-hpf untreated INS-GFP embryos. (B) Exposure to cyclopamine at 6 hpf. (C) Treatment at 8 hpf. (D) Cyclopamine exposure at 12 hpf. (E) Endogenous insulin expression in control ethanol-treated embryos. (F H) Cyclopamine-treated embryos. Cyclopamine was added at 5 hpf (F); branchial arches (arrowhead), 8 (G) and 12 hpf (H). Abbreviations: s4, somite 4; s1, somite1; n, notochord; arrows, position of INS-GFP fluorescent cells; arrowheads, position of insulin-expressing cells. can arise, often at anterior locations. When Shh activity is interrupted after gastrulation (once endoderm formation is complete), normal islets are observed, indicating that Shh is involved in the correct localization of the pancreatic islet. DISCUSSION We report a previously undetected early requirement for Shh in the specification of the pancreas from gut endoderm. This early role for Shh is distinct from any later role of hh signaling in restricting pancreas to a discrete endodermal location. The early action of Shh during pancreatic organogenesis is specific to endocrine islet cells and does not directly affect the exocrine pancreas, duodenum, or liver. Our initial observation that cyclopamine treatment of presomitic zebrafish embryos does not cause an increase in the amount of insulin-expressing islet tissue was unexpected since previous results for cyclopamine-treated 10-somite chick embryos (Kim and Melton, 1998) defined a repressive activity of Shh on pancreas development. One explanation is that the observed Shh signaling in the pancreas is unique to zebrafish. Alternatively, the zebrafish system allows us to look both genetically and pharmacologically to uncover a requirement for hh signaling in vertebrate pancreas development not observable in mammals. We were able to confirm our results by using a direct examination in INS- GFP transgenic embryos to determine the precise timing of the cyclopamine effect based on a temporal analysis of inhibition. We found that the requirement for Shh in islet development is transient and appears to be exerted prior to the end of gastrulation, before notochord development and during endoderm formation. At this stage, the known sources of Shh are the embryonic shield, which acts as a dorsal organizer, and an axially extending mesoendoderm similar to the chick primitive streak (Krauss et al., 1993). Fate mapping in zebrafish embryos shows that pancreatic precursors originate in the blastoderm margin at 40% epiboly, prior to the onset of gastrulation (Warga and Nusslein-Volhard, 1999). After involution, these cells migrate dorsally past the shield and arrive at the midline mesoendoderm by the end of gastrulation. Thus, pancreatic precursors normally migrate in close proximity to Shhexpressing cells at a time when our data show that they are dependent on Shh for specification of the pancreas. Early Shh signaling may act to maintain or induce proliferation in a naive and receptive subset of potential endodermal pancreatic precursors (Rowitch et al., 1999). Alternatively, a transient Shh signal during gastrulation could endow a subset of endodermal cells with competence to respond to later signals that can trigger the generation of the pancreatic endocrine phenotype. This possibility is supported by the observation that the region-specific differentiation of neurons in the chick ventral telencephalon depends on Shh signaling during gastrulation (Gunhaga et al., 2000). Furthermore, shh appears to play a similar early role in the development of the zebrafish adenohypophysis/ pituitary gland (data not shown). Novel experimental strat-
8 82 diiorio et al. TABLE 1 Insulin Expression in Developing, Cyclopamine-Treated Embryos Number of embryos Cyclopamine addition (hpf) Pattern of insulin-gfp expression observed at 72 hpf GFP Loci/embryo No expression Single cell loci Small clusters WT clusters (16) 54 (2) (8) 25 (4) 43 (4) (3) 114 (13) (26) (58) 75 1 Vehicle only 121 (180) 121 Note. Alteration of pancreatic islet development by cyclopamine is dependent on the developmental stage of treatment. Homozygous INS-GFP embryos treated at six time points with 50 M cyclopamine were allowed to develop for 72 hpf and imaged or fixed for insulin in situ analysis. Small clusters contain at least 1 locus of 2 4 fluorescent cells. A WT cluster contains at least 1 locus of 5 cells. Untreated embryos have a single locus of 5 12 cells at 72 hpf (vehicle). Treatment at early stages (4 and 6 hpf) generates no WT clusters and results in anterior repositioning of GFP cells. At the 5- to 7-somite stage (12 hpf), the inhibitory effect of cyclopamine on islet development sharply declines. The number of independent GFP loci/embryo disappears by the 13- to 15-somite stage (20 hpf). Numbers in parentheses indicate embryos analyzed by insulin in situ hybridization. egies will be required to confirm whether Shh also directs early mammalian pancreatic development. Our time series of cyclopamine treatment also reveals a second hh activity once gastrulation is complete. This activity appears to function in the correct anatomical localization of the islet within the gut endoderm since cyclopamine treatment during somitogenesis stages results in the anterior mislocalization of normal-sized insulinexpressing cell clusters. Because this anteriorization is seen after formation of the primitive gut endoderm, it is probably not due to abnormal migration of pancreatic precursors. It is likely that the loss of hh signaling after the endoderm has formed affects inductive processes, perhaps via a release of inhibition anteriorly. This potentially restrictive effect of hh signaling occurring after gastrulation in zebrafish could be analogous to the previously described antagonistic role of Shh identified in chick (Kim and Melton, 1998) and mouse (Apelqvist et al., 1997; Hebrok et al., 2000). However, different mechanisms may be involved for A-P positioning in zebrafish because, unlike in mouse and chick, we cannot detect Shh expression in the gut endoderm prior to 24 hpf (30 somites; data not shown). Early overexpression of Shh also results in islet cells located anterior to somite 4, suggesting the override of an anterior limit of pancreas induction. The interrelationship between early and late hedgehog activity may prove to be complex. However, the generation of differential effects by hh signaling on islet development or function is not unexpected. For example, in differentiated cell lines, insulin gene expression is increased by hh-stimulated Idx-1 (Pdx-1) expression (Thomas et al., 2001). In our experiments, Shh overexpression also increases Pdx-1 expression within the gut endoderm and the pancreas (Figs. 4G and 4H). Because cyclopamine treatment after 24 hpf is not associated with a decrease in the number of insulin-expressing islet cells, hh signaling may not be required for later islet growth in the zebrafish (Table 1). Our overexpression experiments suggest that the early action of Shh to promote islet development could be mediated through Pdx-1. However, the presence of Pdx-1 as well as Isl-1, another potential target of Shh action, in their respective gut domains in the syu / embryos suggests that the islet defect may not be due to a primary failure of Shh to activate Pdx-1 or Isl-1 during development. Recent work (Grapin-Botton et al., 2001) indicates that overexpression of Pdx-1 in chick gut endoderm is not sufficient to induce the formation of islet endocrine cells. Alternatively, the known Shh target genes Nkx2.2 and Pax 6 may play a role in the development of the pancreas (Sussel et al., 1998; Sander et al., 1997; St-Onge et al., 1997) that can be compared with central nervous system development (Barth and Wilson, 1995). We observed that the early pancreatic defect seen in zebrafish mutants deficient in Shh signaling differs significantly from pancreatic phenotypes of mouse genetic knockouts of candidate target genes, including Pdx-1 (Ahlgren et al., 1996; Offield et al., 1996), Hb9 (Li et al., 1999; Harrison et al., 1999), Isl-1 (Ahlgren et al., 1998), NeuroD/Beta2 (Naya et al., 1997), Nkx2.2 (Sussel et al., 1998), Nkx6.1 (Sander et al., 2000), or Pax6 (St-Onge et al., 1997; Sander et al., 1997). Importantly, the selective failure of endocrine islet development we observed does resemble the phenotype of neurogenin 3 inactivation in mice (Gradwohl et al., 2000). This proneural transcription factor functions early in pancreatic development as part of a notch-based mechanism that directs pancreatic precursors to differentiate into endocrine instead of exocrine lineages (Apelqvist et al., 1999; Jensen et al., 2000). Additionally, ectopic overexpression of neurogenin 3 can induce islet cells in early chick gut
9 Shh Promotes Zebrafish Pancreatic Islet Development 83 endoderm (Grapin-Botton et al., 2001). Finally, it has been demonstrated that the expression of another proneural family member, neurogenin 1, is regulated by Shh in the developing zebrafish central nervous system (Blader et al., 1997). Unfortunately, the identity of a zebrafish neurogenin 3 homolog has not yet been established. Islet endocrine cells have long been recognized as structurally and functionally similar to neurons. These peptide hormone-secreting cells express many proteins characteristic of a neural phenotype such as ion channels and key components of a calcium-stimulated, granular secretion apparatus. The resemblance between neural cells and islet cells is further highlighted by the fact that many transcription factors involved in islet cell development are shared by developing neurons. These include Hb9, Isl-1, Nkx2.2, Nkx6.1, and Pax6 that participate in the hh-dependent differentiation of ventral spinal cord motor neurons (Briscoe and Ericson, 2001). While embryonic islet cells were once believed to arise from neural crest cells migrating into the endoderm, it is now clear that both endocrine and exocrine pancreatic cells arise from the endoderm (Le Douarin, 1988; Percival and Slack, 1999). The expression of a neuronal phenotype within endodermal neuroendocrine cells may point to a common mechanism shared between islet cells and neurons. Our studies show that Shh activity may be a mechanistic link between these two differentiation events. Clarification of the role Shh plays in this process awaits the identification of the early endocrine-competent precursor cells along with the specific genes that are targeted by the shh signal. ACKNOWLEDGMENTS Syu and Smu zebrafish strains were kindly provided by A. Chandrasekhar and S. DeVoto, respectively. D. Melton, L. Zon, R. Moon, M. Pack, C. Burgtorf, and H. Lerach shared DNA reagents. We thank N. Roy, A. Goff, A. Klein, J. Weng, J. Dowd, and L. Etheridge for their technical expertise. These studies were supported by Juvenile Diabetes Foundation (to L.G.M.), NIH DK55370 (to L.G.M.), NIH F32DK09743 (to P.J.D.), NIH NS39994 (to R.O.K.), NIH GRASP Digestive Disease Center P30 DK34928, New England Medical Center Research Fund, and a generous gift from Mr. Edmund Lynch. Note added in proof. After submission of our manuscript, a brief communication by Roy et al. [Roy, S., Qiao, T., Wolff, C., and Ingham, P. (2001) Hedgehog signaling pathway is essential for pancreas specification in the zebrafish embryo. Curr. Biol. 11, ] also reported that pancreatic development in zebrafish is dependent on hh signaling. REFERENCES Ahlgren, U., Jonsson, J., and Edlund, H. (1996). The morphogenesis of the pancreatic mesenchyme is uncoupled from that of the pancreatic epithelium in IPF1/PDX1-deficient mice. Development 122, Ahlgren, U., Jonsson, J., Jonsson, L., Simu, K., and Edlund, H. (1998). -Cell-specific inactivation of the mouse Ipf1/Pdx1 gene results in loss of the -cell phenotype and maturity onset diabetes. Genes Dev. 12, Ahlgren, U., Pfaff, S. L., Jessell, T. M., Edlund, T., and Edlund, H. (1997). Independent requirement for ISL1 in formation of pancreatic mesenchyme and islet cells. Nature 385, Apelqvist, A., Ahlgren, U., and Edlund, H. (1997). Sonic hedgehog directs specialised mesoderm differentiation in the intestine and pancreas. Curr. Biol. 7, Apelqvist, A., Li, H., Sommer, L., Beatus, P., Anderson, D. J., Honjo, T., Hrabe de Angelis, M., Lendahl, U., and Edlund, H. (1999). Notch signalling controls pancreatic cell differentiation. Nature 400, Argenton, F., Zecchin, E., and Bortolussi, M. (1999). Early appearance of pancreatic hormone-expressing cells in the zebrafish embryo. Mech. Dev. 87, Barresi, M. J., Stickney, H. L., and Devoto, S. H. (2000). The zebrafish slow-muscle-omitted gene product is required for Hedgehog signal transduction and the development of slow muscle identity. Development 127, Barth, K. A., and Wilson, S. W. (1995). Expression of zebrafish nk2.2 is influenced by sonic hedgehog/vertebrate hedgehog-1 and demarcates a zone of neuronal differentiation in the embryonic forebrain. Development 121, Biemar, F., Argenton, F., Schmidtke, R., Epperlein, S., Peers, B., and Driever, W. (2001). Pancreas development in zebrafish: Early dispersed appearance of endocrine hormone expressing cells and their convergence to form the definitive islet. Dev. Biol. 230, Bitgood, M. J., and McMahon, A. P. (1995). Hedgehog and Bmp genes are coexpressed at many diverse sites of cell-cell interaction in the mouse embryo. Dev. Biol. 172, Blader, P., Fischer, N., Gradwohl, G., Guillemont, F., and Strahle, U. (1997). The activity of neurogenin1 is controlled by local cues in the zebrafish embryo. Development 124, Briscoe, J., and Ericson, J. (2001). Specification of neuronal fates in the ventral neural tube. Curr. Opin. Neurobiol. 11, Cooper, M. K., Porter, J. A., Young, K. E., and Beachy, P. A. (1998). Teratogen-mediated inhibition of target tissue response to Shh signaling. Science 280, Edlund, H. (2001). Developmental biology of the pancreas. Diabetes 50 (Suppl. 1), S5 S9. Ekker, S. C., McGrew, L. L., Lai, C. J., Lee, J. J., von Kessler, D. P., Moon, R. T., and Beachy, P. A. (1995). Distinct expression and shared activities of members of the hedgehog gene family of Xenopus laevis. Development 121, Gradwohl, G., Dierich, A., LeMeur, M., and Guillemot, F. (2000). neurogenin3 is required for the development of the four endocrine cell lineages of the pancreas. Proc. Natl. Acad. Sci. USA 97, Grapin-Botton, A., Majithia, A. R., and Melton, D. A. (2001). Key events of pancreas formation are triggered in gut endoderm by ectopic expression of pancreatic regulatory genes. Genes Dev. 15, Gunhaga, L., Jessell, T. M., and Edlund, T. (2000). Sonic hedgehog signaling at gastrula stages specifies ventral telencephalic cells in the chick embryo. Development 127, Harrison, K. A., Thaler, J., Pfaff, S. L., Gu, H., and Kehrl, J. H. (1999). Pancreas dorsal lobe agenesis and abnormal islets of Langerhans in Hlxb9-deficient mice. Nat. Genet. 23,
10 84 diiorio et al. Hebrok, M., Kim, S. K., and Melton, D. A. (1998). Notochord repression of endodermal Sonic hedgehog permits pancreas development. Genes Dev. 12, Hebrok, M., Kim, S. K., St. Jacques, B., McMahon, A. P., and Melton, D. A. (2000). Regulation of pancreas development by hedgehog signaling. Development 127, Incardona, J. P., Gaffield, W., Kapur, R. P., and Roelink, H. (1998). The teratogenic Veratrum alkaloid cyclopamine inhibits sonic hedgehog signal transduction. Development 125, Jensen, J., Pedersen, E. E., Galante, P., Hald, J., Heller, R. S., Ishibashi, M., Kageyama, R., Guillemot, F., Serup, P., and Madsen, O. D. (2000). Control of endodermal endocrine development by Hes-1. Nat. Genet. 24, Karlstrom, R. O., Talbot, W. S., and Schier, A. F. (1999). Comparative synteny cloning of zebrafish you-too: Mutations in the Hedgehog target gli2 affect ventral forebrain patterning. Genes Dev. 13, Kim, S. K., and Hebrok, M. (2001). Intercellular signals regulating pancreas development and function. Genes Dev. 15, Kim, S. K., Hebrok, M., and Melton, D. A. (1997a). Notochord to endoderm signaling is required for pancreas development. Development 124, Kim, S. K., Hebrok, M., and Melton, D. A. (1997b). Pancreas development in the chick embryo. Cold Spring Harbor. Symp. Quant. Biol. 62, Kim, S. K., and Melton, D. A. (1998). Pancreas development is promoted by cyclopamine, a hedgehog signaling inhibitor. Proc. Natl. Acad. Sci. USA 95, Kimmel, C. B., Ballard, W. W., Kimmel, S. R., Ullmann, B., and Schilling, T. F. (1995). Stages of embryonic development of the zebrafish. Dev. Dyn. 203, Krauss, S., Concordet, J. P., and Ingham, P. W. (1993). A functionally conserved homolog of the Drosophila segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 75, Le Douarin, N. (1988). On the origin of pancreatic endocrine cells. Cell 53, Li, H., Arber, S., Jessell, T. M., and Edlund, H. (1999). Selective agenesis of the dorsal pancreas in mice lacking homeobox gene Hlxb9. Nat. Genet. 23, Liao, J., He, J., Yan, T., Korzh, V., and Gong, Z. (1999). A class of neurod-related basic helix-loop-helix transcription factors expressed in developing central nervous system in zebrafish. DNA Cell Biol. 18, Milewski, W. M., Duguay, S. J., Chan, S. J., and Steiner, D. F. (1998). Conservation of PDX-1 structure, function, and expression in zebrafish. Endocrinology 139, Miller, C. P., McGehee Jr., R. E., and Habener, J. F. (1994). IDX-1: A new homeodomain transcription factor expressed in rat pancreatic islets and duodenum that transactivates the somatostatin gene. EMBO J. 13, Naya, F. J., Huang, H. P., Qiu, Y., Mutoh, H., DeMayo, F. J., Leiter, A. B., and Tsai, M. J. (1997). Diabetes, defective pancreatic morphogenesis, and abnormal enteroendocrine differentiation in BETA2/neuroD-deficient mice. Genes Dev. 11, Norris, W., Neyt, C., Ingham, P. W., and Currie, P. D. (2000). Slow muscle induction by Hedgehog signalling in vitro. J. Cell Sci. 113, Offield, M. F., Jetton, T. L., Labosky, P. A., Ray, M., Stein, R. W., Magnuson, M. A., Hogan, B. L., and Wright, C. V. (1996). PDX-1 is required for pancreatic outgrowth and differentiation of the rostral duodenum. Development 122, Ohlsson, H., Karlsson, K., and Edlund, T. (1993). IPF1, a homeodomain-containing transactivator of the insulin gene. EMBO J. 12, Oxtoby, E., and Jowett, T. (1993). Cloning of the zebrafish krox-20 gene (krx-20) and its expression during hindbrain development. Nucleic Acids Res. 21, Percival, A. C., and Slack, J. M. (1999). Analysis of pancreatic development using a cell lineage label. Exp. Cell Res. 247, Raz, E., van Luenen, H. G., Schaerringer, B., Plasterk, R. H., and Driever, W. (1998). Transposition of the nematode Caenorhabditis elegans Tc3 element in the zebrafish Danio rerio. Curr. Biol. 8, Rowitch, D. H., S-Jacques, B., Lee, S. M., Flax, J. D., Snyder, E. Y., and McMahon, A. P. (1999). Sonic hedgehog regulates proliferation and inhibits differentiation of CNS precursor cells. J. Neurosci. 19, Sander, M., Neubuser, A., Kalamaras, J., Ee, H. C., Martin, G. R., and German, M. S. (1997). Genetic analysis reveals that PAX6 is required for normal transcription of pancreatic hormone genes and islet development. Genes Dev. 11, Sander, M., Sussel, L., Conners, J., Scheel, D., Kalamaras, J., Dela Cruz, F., Schwitzgebel, V., Hayes-Jordan, A., and German, M. (2000). Homeobox gene Nkx6.1 lies downstream of Nkx2.2 in the major pathway of beta-cell formation in the pancreas. Development 127, Schauerte, H. E., van Eeden, F. J., Fricke, C., Odenthal, J., Strahle, U., and Haffter, P. (1998). Sonic hedgehog is not required for the induction of medial floor plate cells in the zebrafish. Development 125, St-Onge, L., Sosa-Pineda, B., Chowdhury, K., Mansouri, A., and Gruss, P. (1997). Pax6 is required for differentiation of glucagonproducing alpha-cells in mouse pancreas. Nature 387, Sussel, L., Kalamaras, J., Hartigan O Connor, D. J., Meneses, J. J., Pedersen, R. A., Rubenstein, J. L., and German, M. S. (1998). Mice lacking the homeodomain transcription factor Nkx2.2 have diabetes due to arrested differentiation of pancreatic beta cells. Development 125, Taipale, J., Chen, J. K., Cooper, M. K., Wang, B., Mann, R. K., Milenkovic, L., Scott, M. P., and Beachy, P. A. (2000). Effects of oncogenic mutations in Smoothened and Patched can be reversed by cyclopamine. Nature 406, Thomas, M., Lee, J., Rastalsky, N., and Habener, J. (2001). Hedgehog signaling regulation of homeodomain protein islet duodenum homeobox-1 expression in pancreatic -cells. Endocrinology 142, Varga, Z. M., Amores, A., Lewis, K. E., Yan, Y. L., Postlethwait, J. H., Eisen, J. S., and Westerfield, M. (2001). Zebrafish smoothened functions in ventral neural tube specification and axon tract formation. Development 128, Warga, R. M., and Nusslein-Volhard, C. (1999). Origin and development of the zebrafish endoderm. Development 126, Wells, J. M., and Melton, D. A. (2000). Early mouse endoderm is patterned by soluble factors from adjacent germ layers. Development 127, Westerfield, M. (1995). The Zebrafish Book: Guide for the Laboratory Use of Zebrafish (Danio rerio). Univ. of Oregon Press, Eugene. Received for publication September 28, 2001 Accepted December 19, 2001 Published online February 25, 2002
Homeobox gene Nkx6.1 lies downstream of Nkx2.2 in the major pathway of β-cell formation in the pancreas
 Development 127, 5533-5540 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV6501 5533 Homeobox gene Nkx6.1 lies downstream of Nkx2.2 in the major pathway of β-cell formation in
Development 127, 5533-5540 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV6501 5533 Homeobox gene Nkx6.1 lies downstream of Nkx2.2 in the major pathway of β-cell formation in
CNS Developmental. Anke van Eekelen, PhD. Telethon Institute for Child Health Research
 CNS Developmental Anke van Eekelen, PhD Telethon Institute for Child Health Research (Some slides are modified versions of Prof. Alan Harvey s Neuroscience lecture at ANHB and Dr. Joanne Britto s Dev Neuroscience
CNS Developmental Anke van Eekelen, PhD Telethon Institute for Child Health Research (Some slides are modified versions of Prof. Alan Harvey s Neuroscience lecture at ANHB and Dr. Joanne Britto s Dev Neuroscience
Regionalization of the nervous system. Paul Garrity 7.68J/9.013J February 25, 2004
 Regionalization of the nervous system Paul Garrity 7.68J/9.013J February 25, 2004 Patterning along: Rostral/Caudal (AP) axis Dorsal/Ventral (DV) axis Start with DV axial patterning in Spinal Cord Dorsal/Ventral
Regionalization of the nervous system Paul Garrity 7.68J/9.013J February 25, 2004 Patterning along: Rostral/Caudal (AP) axis Dorsal/Ventral (DV) axis Start with DV axial patterning in Spinal Cord Dorsal/Ventral
Neuroepithelial Cells and Neural Differentiation
 Neuroepithelial Cells and Neural Differentiation Neurulation The cells of the neural tube are NEUROEPITHELIAL CELLS Neural crest cells migrate out of neural tube Neuroepithelial cells are embryonic stem
Neuroepithelial Cells and Neural Differentiation Neurulation The cells of the neural tube are NEUROEPITHELIAL CELLS Neural crest cells migrate out of neural tube Neuroepithelial cells are embryonic stem
Supplementary Fig. 1 Blocking shh function at the protein level confirms its role as a guidance cue for postcommissural axons.
 Supplementary Fig. 1 Blocking shh function at the protein level confirms its role as a guidance cue for postcommissural axons. As an alternative method to demonstrate the role of shh as a guidance cue
Supplementary Fig. 1 Blocking shh function at the protein level confirms its role as a guidance cue for postcommissural axons. As an alternative method to demonstrate the role of shh as a guidance cue
A Cxcl12-Cxcr4 Chemokine Signaling Pathway Defines
 Supplemental Data A Cxcl12-Cxcr4 Chemokine Signaling Pathway Defines the Initial Trajectory of Mammalian Motor Axons Ivo Lieberam, Dritan Agalliu, Takashi Nagasawa, Johan Ericson, and Thomas M. Jessell
Supplemental Data A Cxcl12-Cxcr4 Chemokine Signaling Pathway Defines the Initial Trajectory of Mammalian Motor Axons Ivo Lieberam, Dritan Agalliu, Takashi Nagasawa, Johan Ericson, and Thomas M. Jessell
Cell Type Nervous System I. Developmental Readout. Foundations. Stem cells. Organ formation. Human issues.
 7.72 10.11.06 Cell Type Nervous System I Human issues Organ formation Stem cells Developmental Readout Axes Cell type Axon guidance 3D structure Analysis Model + + organisms Foundations Principles 1 What
7.72 10.11.06 Cell Type Nervous System I Human issues Organ formation Stem cells Developmental Readout Axes Cell type Axon guidance 3D structure Analysis Model + + organisms Foundations Principles 1 What
Patterning the Embryo
 Patterning the Embryo Anteroposterior axis Regional Identity in the Vertebrate Neural Tube Fig. 2.2 1 Brain and Segmental Ganglia in Drosophila Fig. 2.1 Genes that create positional and segment identity
Patterning the Embryo Anteroposterior axis Regional Identity in the Vertebrate Neural Tube Fig. 2.2 1 Brain and Segmental Ganglia in Drosophila Fig. 2.1 Genes that create positional and segment identity
Hedgehog signaling regulates expansion of pancreatic epithelial cells
 Developmental Biology 280 (2005) 111 121 www.elsevier.com/locate/ydbio Hedgehog signaling regulates expansion of pancreatic epithelial cells Hiroshi Kawahira, David W. Scheel, Stuart B. Smith, Michael
Developmental Biology 280 (2005) 111 121 www.elsevier.com/locate/ydbio Hedgehog signaling regulates expansion of pancreatic epithelial cells Hiroshi Kawahira, David W. Scheel, Stuart B. Smith, Michael
Combined activities of hedgehog signaling inhibitors regulate pancreas development
 Research article 4871 Combined activities of hedgehog signaling inhibitors regulate pancreas development Hiroshi Kawahira 1, Nancy H. Ma 1, Emmanouhl S. Tzanakakis 1, Andrew P. McMahon 2, Pao-Tien Chuang
Research article 4871 Combined activities of hedgehog signaling inhibitors regulate pancreas development Hiroshi Kawahira 1, Nancy H. Ma 1, Emmanouhl S. Tzanakakis 1, Andrew P. McMahon 2, Pao-Tien Chuang
Hepatogenesis I Liver development
 Hepatogenesis I Liver development HB 308 George Yeoh Room 2.59 MCS Building yeoh@cyllene.uwa.edu.au Topics Early liver development Tissue interaction - role of morphogens and cytokines Liver enriched transcription
Hepatogenesis I Liver development HB 308 George Yeoh Room 2.59 MCS Building yeoh@cyllene.uwa.edu.au Topics Early liver development Tissue interaction - role of morphogens and cytokines Liver enriched transcription
Developing Molecularly Targeted Therapies for Basal Cell Carcinoma. Ivor Caro, MD, FAAD
 Developing Molecularly Targeted Therapies for Basal Cell Carcinoma Ivor Caro, MD, FAAD Disclosures Genentech, Inc Medical Director, Dermatology (employee) Stock holder Hedgehog Signaling Pathway Fundamental
Developing Molecularly Targeted Therapies for Basal Cell Carcinoma Ivor Caro, MD, FAAD Disclosures Genentech, Inc Medical Director, Dermatology (employee) Stock holder Hedgehog Signaling Pathway Fundamental
DEVELOPMENT. Shelley B. Nelson, Ashleigh E. Schaffer and Maike Sander*
 RESEARCH ARTICLE 2491 Development 134, 2491-2500 (2007) doi:10.1242/dev.002691 The transcription factors Nkx6.1 and Nkx6.2 possess equivalent activities in promoting beta-cell fate specification in Pdx1
RESEARCH ARTICLE 2491 Development 134, 2491-2500 (2007) doi:10.1242/dev.002691 The transcription factors Nkx6.1 and Nkx6.2 possess equivalent activities in promoting beta-cell fate specification in Pdx1
Chapter 5. Summary and Future directions
 95 Chapter 5 Summary and Future directions 96 Much of our knowledge about glial development in the vertebrate CNS comes from studies of purified oligodendrocyte precursor cells (Raff 1989; Pfeiffer et
95 Chapter 5 Summary and Future directions 96 Much of our knowledge about glial development in the vertebrate CNS comes from studies of purified oligodendrocyte precursor cells (Raff 1989; Pfeiffer et
Pancreas Development in Zebrafish: Early Dispersed Appearance of Endocrine Hormone Expressing Cells and Their Convergence to Form the Definitive Islet
 Developmental Biology 230, 189 203 (2001) doi:10.1006/dbio.2000.0103, available online at http://www.idealibrary.com on Pancreas Development in Zebrafish: Early Dispersed Appearance of Endocrine Hormone
Developmental Biology 230, 189 203 (2001) doi:10.1006/dbio.2000.0103, available online at http://www.idealibrary.com on Pancreas Development in Zebrafish: Early Dispersed Appearance of Endocrine Hormone
Myf5 is a direct target of long-range Shh signaling and Gli regulation for muscle specification
 Myf5 is a direct target of long-range Shh signaling and Gli regulation for muscle specification Marcus K. Gustafsson, 1,3,4 Hua Pan, 1,3,4 Deborah F. Pinney, 1,3 Yongliang Liu, 1,3 Anna Lewandowski, 1,3
Myf5 is a direct target of long-range Shh signaling and Gli regulation for muscle specification Marcus K. Gustafsson, 1,3,4 Hua Pan, 1,3,4 Deborah F. Pinney, 1,3 Yongliang Liu, 1,3 Anna Lewandowski, 1,3
Vertebrate Limb Patterning
 Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Defining the cell lineages of the islets of langerhans using transgenic mice
 Int. J. Dev. Biol. 46: 97-103 (2002) Defining the cell lineages of the islets of langerhans using transgenic mice PEDRO L. HERRERA* Department of Morphology, University of Geneva Medical School. 1 Rue
Int. J. Dev. Biol. 46: 97-103 (2002) Defining the cell lineages of the islets of langerhans using transgenic mice PEDRO L. HERRERA* Department of Morphology, University of Geneva Medical School. 1 Rue
Developmental Biology
 Developmental Biology 362 (2012) 141 153 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Dual function of suppressor of
Developmental Biology 362 (2012) 141 153 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Dual function of suppressor of
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
Polarity and Segmentation. Chapter Two
 Polarity and Segmentation Chapter Two Polarization Entire body plan is polarized One end is different than the other Head vs. Tail Anterior vs. Posterior Front vs. Back Ventral vs. Dorsal Majority of neural
Polarity and Segmentation Chapter Two Polarization Entire body plan is polarized One end is different than the other Head vs. Tail Anterior vs. Posterior Front vs. Back Ventral vs. Dorsal Majority of neural
Reciprocal endoderm-mesoderm interactions mediated by fgf24 and fgf10 govern pancreas development
 RESEARCH ARTICLE 4011 Development 134, 4011-4021 (2007) doi:10.1242/dev.007823 Reciprocal endoderm-mesoderm interactions mediated by fgf24 and fgf10 govern pancreas development Isabelle Manfroid 1, *,
RESEARCH ARTICLE 4011 Development 134, 4011-4021 (2007) doi:10.1242/dev.007823 Reciprocal endoderm-mesoderm interactions mediated by fgf24 and fgf10 govern pancreas development Isabelle Manfroid 1, *,
Fgf10 is essential for maintaining the proliferative capacity of epithelial
 Development 128, 5109-5117 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV6542 5109 Fgf10 is essential for maintaining the proliferative capacity of epithelial progenitor cells
Development 128, 5109-5117 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV6542 5109 Fgf10 is essential for maintaining the proliferative capacity of epithelial progenitor cells
NKX6 transcription factor activity is required for α- and β-cell development in the pancreas
 Research article and disease 3139 NKX6 transcription factor activity is required for α- and β-cell development in the pancreas Korinna D. Henseleit 1,2, Shelley B. Nelson 1, *, Kirsten Kuhlbrodt 2, *,
Research article and disease 3139 NKX6 transcription factor activity is required for α- and β-cell development in the pancreas Korinna D. Henseleit 1,2, Shelley B. Nelson 1, *, Kirsten Kuhlbrodt 2, *,
Urogenital Development
 2-5-03 Urogenital Development Greg Dressler Assoc. Professor Dept. of Pathology x46490 Dressler@umich.edu The Origin of the Kidney In the vertebrate embryo, the first stage of kidney development occurs
2-5-03 Urogenital Development Greg Dressler Assoc. Professor Dept. of Pathology x46490 Dressler@umich.edu The Origin of the Kidney In the vertebrate embryo, the first stage of kidney development occurs
Biochemical and Biophysical Research Communications
 Biochemical and Biophysical Research Communications 399 (2010) 440 445 Contents lists available at ScienceDirect Biochemical and Biophysical Research Communications journal homepage: www.elsevier.com/locate/ybbrc
Biochemical and Biophysical Research Communications 399 (2010) 440 445 Contents lists available at ScienceDirect Biochemical and Biophysical Research Communications journal homepage: www.elsevier.com/locate/ybbrc
BRAIN DEVELOPMENT I: ESTABLISHMENT OF BASIC ARCHITECTURE. Thomas Marino, Ph.D.
 BRAIN DEVELOPMENT I: ESTABLISHMENT OF BASIC ARCHITECTURE Thomas Marino, Ph.D. Development of the Brain I. Competencies: Upon completion of this section of the course, the student must be able to: 1. Understand
BRAIN DEVELOPMENT I: ESTABLISHMENT OF BASIC ARCHITECTURE Thomas Marino, Ph.D. Development of the Brain I. Competencies: Upon completion of this section of the course, the student must be able to: 1. Understand
Mice lacking the homeodomain transcription factor Nkx2.2 have diabetes due to arrested differentiation of pancreatic β cells
 Development 125, 2213-2221 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV6347 2213 Mice lacking the homeodomain transcription factor Nkx2.2 have diabetes due to arrested differentiation
Development 125, 2213-2221 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV6347 2213 Mice lacking the homeodomain transcription factor Nkx2.2 have diabetes due to arrested differentiation
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Neurodevelopment II Structure Formation. Reading: BCP Chapter 23
 Neurodevelopment II Structure Formation Reading: BCP Chapter 23 Phases of Development Ovum + Sperm = Zygote Cell division (multiplication) Neurogenesis Induction of the neural plate Neural proliferation
Neurodevelopment II Structure Formation Reading: BCP Chapter 23 Phases of Development Ovum + Sperm = Zygote Cell division (multiplication) Neurogenesis Induction of the neural plate Neural proliferation
Tetrapod Limb Development
 IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
Pancreas development is promoted by cyclopamine, a Hedgehog signaling inhibitor
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 13036 13041, October 1998 Developmental Biology Pancreas development is promoted by cyclopamine, a Hedgehog signaling inhibitor SEUNG K. KIM* AND DOUGLAS A. MELTON
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 13036 13041, October 1998 Developmental Biology Pancreas development is promoted by cyclopamine, a Hedgehog signaling inhibitor SEUNG K. KIM* AND DOUGLAS A. MELTON
Epithelium-mesenchyme interactions play a crucial. Original Article Control of -Cell Differentiation by the Pancreatic Mesenchyme
 Original Article Control of -Cell Differentiation by the Pancreatic Mesenchyme Myriam Attali, 1 Volodymyr Stetsyuk, 1 Annie Basmaciogullari, 1 Virginie Aiello, 1 Maria A. Zanta-Boussif, 2 Bertrand Duvillie,
Original Article Control of -Cell Differentiation by the Pancreatic Mesenchyme Myriam Attali, 1 Volodymyr Stetsyuk, 1 Annie Basmaciogullari, 1 Virginie Aiello, 1 Maria A. Zanta-Boussif, 2 Bertrand Duvillie,
Gli2 is required for induction of floor plate and adjacent cells, but not most
 Development 125, 2759-2770 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV9597 2759 Gli2 is required for induction of floor plate and adjacent cells, but not most ventral neurons
Development 125, 2759-2770 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV9597 2759 Gli2 is required for induction of floor plate and adjacent cells, but not most ventral neurons
Development of the Pancreas and Response to Disease
 Chapter 1 Development of the Pancreas and Response to Disease The pancreas develops from the primitive gut, which is derived from endoderm. An interdependent series of signals is necessary to form the
Chapter 1 Development of the Pancreas and Response to Disease The pancreas develops from the primitive gut, which is derived from endoderm. An interdependent series of signals is necessary to form the
Genes controlling pancreas ontogeny. CLAIRE BONAL and PEDRO L. HERRERA*
 Int. J. Dev. Biol. 52: 823-835 (2008) doi: 10.1387/ijdb.072444cb THE INTERNATIONAL JOURNAL OF DEVELOPMENTAL BIOLOGY www.intjdevbiol.com Genes controlling pancreas ontogeny CLAIRE BONAL and PEDRO L. HERRERA*
Int. J. Dev. Biol. 52: 823-835 (2008) doi: 10.1387/ijdb.072444cb THE INTERNATIONAL JOURNAL OF DEVELOPMENTAL BIOLOGY www.intjdevbiol.com Genes controlling pancreas ontogeny CLAIRE BONAL and PEDRO L. HERRERA*
Pancreatic endocrine and exocrine cells are thought
 Changes in the Expression of Transcription Factors in Pancreatic AR42J Cells During Differentiation Into Insulin-Producing Cells You-Qing Zhang, Hirosato Mashima, and Itaru Kojima Pancreatic AR42J cells
Changes in the Expression of Transcription Factors in Pancreatic AR42J Cells During Differentiation Into Insulin-Producing Cells You-Qing Zhang, Hirosato Mashima, and Itaru Kojima Pancreatic AR42J cells
The Evolution and Development of the Gut. Dr Mike Wride School of Natural Sciences Zoology Department
 The Evolution and Development of the Gut Dr Mike Wride School of Natural Sciences Zoology Department email: wridem@tcd.ie The gut? Gut Function and Regulation (Dr. Alan Tuffery) Absorption of nutrients
The Evolution and Development of the Gut Dr Mike Wride School of Natural Sciences Zoology Department email: wridem@tcd.ie The gut? Gut Function and Regulation (Dr. Alan Tuffery) Absorption of nutrients
Axis Formation and Mesoderm Induction
 Developmental Biology Biology 4361 Axis Formation and Mesoderm Induction October 27, 2005 Amphibian anteroposterior specification polarized eggs animal/vegetal pigment yolk v. clear cytoplasm mitochondrial
Developmental Biology Biology 4361 Axis Formation and Mesoderm Induction October 27, 2005 Amphibian anteroposterior specification polarized eggs animal/vegetal pigment yolk v. clear cytoplasm mitochondrial
EMBO REPORT SUPPLEMENTARY SECTION. Quantitation of mitotic cells after perturbation of Notch signalling.
 EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
HEDGEHOG SIGNALLING IN CANCER FORMATION AND MAINTENANCE
 HEDGEHOG SIGNALLING IN CANCER FORMATION AND MAINTENANCE Marina Pasca di Magliano and Matthias Hebrok The Hedgehog signalling pathway is essential for numerous processes during embryonic development. Members
HEDGEHOG SIGNALLING IN CANCER FORMATION AND MAINTENANCE Marina Pasca di Magliano and Matthias Hebrok The Hedgehog signalling pathway is essential for numerous processes during embryonic development. Members
Preferential reduction of β cells derived from Pax6 MafB pathway in MafB deficient mice
 Available online at www.sciencedirect.com Developmental Biology 314 (2008) 443 456 www.elsevier.com/developmentalbiology Genomes & Developmental Control Preferential reduction of β cells derived from Pax6
Available online at www.sciencedirect.com Developmental Biology 314 (2008) 443 456 www.elsevier.com/developmentalbiology Genomes & Developmental Control Preferential reduction of β cells derived from Pax6
Regulation of pancreas development by hedgehog signaling
 Development 127, 4905-4913 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV3222 4905 Regulation of pancreas development by hedgehog signaling Matthias Hebrok 1, *,, Seung K. Kim
Development 127, 4905-4913 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV3222 4905 Regulation of pancreas development by hedgehog signaling Matthias Hebrok 1, *,, Seung K. Kim
Expression of neurogenin3 reveals an islet cell precursor population in the pancreas
 Development 127, 3533-3542 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV6458 3533 Expression of neurogenin3 reveals an islet cell precursor population in the pancreas Valérie
Development 127, 3533-3542 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV6458 3533 Expression of neurogenin3 reveals an islet cell precursor population in the pancreas Valérie
Lecture IV. Mechanisms of Neural. Neural Development
 Lecture IV. Mechanisms of Neural Bio 3411 Monday 1 Readings NEUROSCIENCE: 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) References : Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994).
Lecture IV. Mechanisms of Neural Bio 3411 Monday 1 Readings NEUROSCIENCE: 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) References : Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994).
Edited by Kathryn V. Anderson, Sloan Kettering Institute, New York, NY, and approved December 20, 2004 (received for review August 6, 2004)
 Lack of TCF2 vhnf1 in mice leads to pancreas agenesis C. Haumaitre*, E. Barbacci*, M. Jenny, M. O. Ott*, G. Gradwohl, and S. Cereghini* *Biologie du Développement, Unité Mixte de Recherche 7622, Centre
Lack of TCF2 vhnf1 in mice leads to pancreas agenesis C. Haumaitre*, E. Barbacci*, M. Jenny, M. O. Ott*, G. Gradwohl, and S. Cereghini* *Biologie du Développement, Unité Mixte de Recherche 7622, Centre
Embryology of the Nervous System. Steven McLoon Department of Neuroscience University of Minnesota
 Embryology of the Nervous System Steven McLoon Department of Neuroscience University of Minnesota In the blastula stage embryo, the embryonic disk has two layers. During gastrulation, epiblast cells migrate
Embryology of the Nervous System Steven McLoon Department of Neuroscience University of Minnesota In the blastula stage embryo, the embryonic disk has two layers. During gastrulation, epiblast cells migrate
Conditional inactivation of Pax6 in the pancreas causes early onset of diabetes
 Developmental Biology 269 (2004) 479 488 www.elsevier.com/locate/ydbio Conditional inactivation of Pax6 in the pancreas causes early onset of diabetes Ruth Ashery-Padan, a, * Xunlei Zhou, b Till Marquardt,
Developmental Biology 269 (2004) 479 488 www.elsevier.com/locate/ydbio Conditional inactivation of Pax6 in the pancreas causes early onset of diabetes Ruth Ashery-Padan, a, * Xunlei Zhou, b Till Marquardt,
Development of the Liver and Pancreas
 Development of the Liver and Pancreas Professor Alfred Cuschieri Department of Anatomy University of Malta Three glandular buds arise from the distal end of the foregut during the fourth week Day 22 -The
Development of the Liver and Pancreas Professor Alfred Cuschieri Department of Anatomy University of Malta Three glandular buds arise from the distal end of the foregut during the fourth week Day 22 -The
ROLE OF EPIDERMAL AND FIBROBLAST GROWTH FACTORS IN PANCREATIC DEVELOPMENT
 Helsinki University Biomedical Dissertations No. 33 ROLE OF EPIDERMAL AND FIBROBLAST GROWTH FACTORS IN PANCREATIC DEVELOPMENT Mari-Anne Pulkkinen Program for Developmental and Reproductive Biology Biomedicum
Helsinki University Biomedical Dissertations No. 33 ROLE OF EPIDERMAL AND FIBROBLAST GROWTH FACTORS IN PANCREATIC DEVELOPMENT Mari-Anne Pulkkinen Program for Developmental and Reproductive Biology Biomedicum
Supplementary Figure 1. A microarray screen of organizers compared to non-organizer tissue reveals a putative organizer gene set.
 Supplementary Figure 1. A microarray screen of organizers compared to non-organizer tissue reveals a putative organizer gene set. (a, b) Venn diagrams of 31 enriched (a) and 17 depleted (b) genes significantly
Supplementary Figure 1. A microarray screen of organizers compared to non-organizer tissue reveals a putative organizer gene set. (a, b) Venn diagrams of 31 enriched (a) and 17 depleted (b) genes significantly
Tetrapod Limb Development
 Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
SONIC HEDGEHOG (Shh) is a secreted glycoprotein
 Role of Sonic Hedgehog in the Development of the Trachea and Oesophagus By Adonis S. Ioannides, Deborah J. Henderson, Lewis Spitz, and Andrew J. Copp London, England and Newcastle, England Backround/Purpose:
Role of Sonic Hedgehog in the Development of the Trachea and Oesophagus By Adonis S. Ioannides, Deborah J. Henderson, Lewis Spitz, and Andrew J. Copp London, England and Newcastle, England Backround/Purpose:
Diabetic pdx1-mutant zebrafish show conserved responses to nutrient overload and anti-glycemic treatment
 Supplementary Information Diabetic pdx1-mutant zebrafish show conserved responses to nutrient overload and anti-glycemic treatment Robin A. Kimmel, Stefan Dobler, Nicole Schmitner, Tanja Walsen, Julia
Supplementary Information Diabetic pdx1-mutant zebrafish show conserved responses to nutrient overload and anti-glycemic treatment Robin A. Kimmel, Stefan Dobler, Nicole Schmitner, Tanja Walsen, Julia
A Genetic Program for Embryonic Development
 Concept 18.4: A program of differential gene expression leads to the different cell types in a multicellular organism During embryonic development, a fertilized egg gives rise to many different cell types
Concept 18.4: A program of differential gene expression leads to the different cell types in a multicellular organism During embryonic development, a fertilized egg gives rise to many different cell types
Emx2 patterns the neocortex by regulating FGF positional signaling
 Emx2 patterns the neocortex by regulating FGF positional signaling Tomomi Fukuchi-Shimogori and Elizabeth A Grove Presented by Sally Kwok Background Cerebral cortex has anatomically and functionally distinct
Emx2 patterns the neocortex by regulating FGF positional signaling Tomomi Fukuchi-Shimogori and Elizabeth A Grove Presented by Sally Kwok Background Cerebral cortex has anatomically and functionally distinct
Tetrapod Limb Development
 Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
Biology 4361 Developmental Biology Tetrapod Limb Development July 29, 2009 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Development - Overview
Retinoic Acid Signaling Is Required for a Critical Early Step in Zebrafish Pancreatic Development
 Current Biology, Vol. 12, 1215 1220, July 23, 2002, 2002 Elsevier Science Ltd. All rights reserved. PII S0960-9822(02)00929-6 Retinoic Acid Signaling Is Required for a Critical Early Step in Zebrafish
Current Biology, Vol. 12, 1215 1220, July 23, 2002, 2002 Elsevier Science Ltd. All rights reserved. PII S0960-9822(02)00929-6 Retinoic Acid Signaling Is Required for a Critical Early Step in Zebrafish
Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy
 Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
Kavya Puchhalapalli CALS Honors Project Report Spring 2017 Studying The Role Of DNA Mismatch Repair In Brain Cancer Malignancy Abstract Malignant brain tumors including medulloblastomas and primitive neuroectodermal
A bipotential precursor population for pancreas and liver within the embryonic endoderm
 Development 128, 871-881 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV3286 871 A bipotential precursor population for pancreas and liver within the embryonic endoderm Gail
Development 128, 871-881 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV3286 871 A bipotential precursor population for pancreas and liver within the embryonic endoderm Gail
CHAPTER 6 SUMMARIZING DISCUSSION
 CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
CHAPTER 6 SUMMARIZING DISCUSSION More than 20 years ago the founding member of the Wnt gene family, Wnt-1/Int1, was discovered as a proto-oncogene activated in mammary gland tumors by the mouse mammary
Supporting Information
 Supporting Information Creuzet 10.1073/pnas.0906072106 Fig. S1. Migrating CNC cells express Bmp inhibitors. (A) HNK1 immunolabeling of migrating CNC cells at 7 ss. (B) At this stage, Fgf8 is strongly expressed
Supporting Information Creuzet 10.1073/pnas.0906072106 Fig. S1. Migrating CNC cells express Bmp inhibitors. (A) HNK1 immunolabeling of migrating CNC cells at 7 ss. (B) At this stage, Fgf8 is strongly expressed
Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model.
 161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
Current Model for how cells become neural. Double Inhibition Model for Neural fate. Neural Development. How do cells become neurons?
 Current Model for how cells becoe neural 1) Default state is neural 2) Local secretion of BMPs by epideris inhibits neural fate 3) Local secretion of noggin, chordin by dorsal lip or esoder inhibits BMP
Current Model for how cells becoe neural 1) Default state is neural 2) Local secretion of BMPs by epideris inhibits neural fate 3) Local secretion of noggin, chordin by dorsal lip or esoder inhibits BMP
Week 14. Development of the Musculoskeletal System
 Week 14 Development of the Musculoskeletal System Skeletal System Derived from: paraxial mesoderm somites and somitomeres sclerotome sclerotome differentiation induced by SHH from notochord and floor plate
Week 14 Development of the Musculoskeletal System Skeletal System Derived from: paraxial mesoderm somites and somitomeres sclerotome sclerotome differentiation induced by SHH from notochord and floor plate
Functional Analysis of Ipf1/Pdx1, MFng and Id during Pancreatic Growth and Differentiation. Per Svensson
 Functional Analysis of Ipf1/Pdx1, MFng and Id during Pancreatic Growth and Differentiation Per Svensson Umeå Centre for Molecular Medicine Umeå University, Umeå Sweden 2008 Front cover: In situ hybridisation
Functional Analysis of Ipf1/Pdx1, MFng and Id during Pancreatic Growth and Differentiation Per Svensson Umeå Centre for Molecular Medicine Umeå University, Umeå Sweden 2008 Front cover: In situ hybridisation
a) They are the most common cause of pediatric kidney failure. b) They are always symptomatic. c) They can be asymmetric.
 Practice questions: 1. The paraxial mesoderm gives rise to somites. The structure of the somite a) is a loose mesenchymal sheet that will migrate toward the notochord. b) is an epithelial rosette with
Practice questions: 1. The paraxial mesoderm gives rise to somites. The structure of the somite a) is a loose mesenchymal sheet that will migrate toward the notochord. b) is an epithelial rosette with
Combined ectopic expression of Pdx1 and Ptf1a/p48 results in the stable conversion of posterior endoderm into endocrine and exocrine pancreatic tissue
 RESEARCH COMMUNICATION Combined ectopic expression of Pdx1 and Ptf1a/p48 results in the stable conversion of posterior endoderm into endocrine and exocrine pancreatic tissue Solomon Afelik, Yonglong Chen,
RESEARCH COMMUNICATION Combined ectopic expression of Pdx1 and Ptf1a/p48 results in the stable conversion of posterior endoderm into endocrine and exocrine pancreatic tissue Solomon Afelik, Yonglong Chen,
SUPPLEMENTARY DATA. Supplementary Table 2. Antibodies used for Immunofluoresence. Supplementary Table 3. Real-time PCR primer sequences.
 Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Tlx3 and Tlx1 are post-mitotic selector genes determining glutamatergic over GABAergic cell fates
 Tlx3 and Tlx1 are post-mitotic selector genes determining glutamatergic over GABAergic cell fates L. Cheng, A. Arata, R. Mizuguchi, Y. Qian, A. Karunaratne, P.A. Gray, S. Arata, S. Shirasawa, M. Bouchard,
Tlx3 and Tlx1 are post-mitotic selector genes determining glutamatergic over GABAergic cell fates L. Cheng, A. Arata, R. Mizuguchi, Y. Qian, A. Karunaratne, P.A. Gray, S. Arata, S. Shirasawa, M. Bouchard,
Fig. S1. RT-PCR analyses of the expression and distribution of Xdscr6 transcripts during early development.
 Fig. S1. RT-PCR analyses of the expression and distribution of Xdscr6 transcripts during early development. (A) Temporal expression of Xdscr6 at various stages (numbers on the top) and its distribution
Fig. S1. RT-PCR analyses of the expression and distribution of Xdscr6 transcripts during early development. (A) Temporal expression of Xdscr6 at various stages (numbers on the top) and its distribution
Effects of Nicotine on Embryological Neural Development. A Senior Honors Thesis. in the College of Biological Sciences of the Ohio State University
 Effects of Nicotine on Embryological Neural Development A Senior Honors Thesis Presented in Fulfillment of the Requirements for Graduation with Distinction in Biology in the College of Biological Sciences
Effects of Nicotine on Embryological Neural Development A Senior Honors Thesis Presented in Fulfillment of the Requirements for Graduation with Distinction in Biology in the College of Biological Sciences
MPB333:Molecular Endocrinology of Obesity and Diabetes
 MPB333:Molecular Endocrinology of Obesity and Diabetes The Use of Stem Cells as a Cure for Type 1 Diabetes January 15, 2010 Trish Labosky 9415C MRBIV trish.labosky@vanderbilt.edu In theory. 2. ~Easy and
MPB333:Molecular Endocrinology of Obesity and Diabetes The Use of Stem Cells as a Cure for Type 1 Diabetes January 15, 2010 Trish Labosky 9415C MRBIV trish.labosky@vanderbilt.edu In theory. 2. ~Easy and
Development of the Axial Skeleton and Limbs. Professor Alfred Cuschieri Department of Anatomy University of Malta
 Development of the Axial Skeleton and Limbs Professor Alfred Cuschieri Department of Anatomy University of Malta During the Fourth Week the Embryo Is Segmented. Each segment consists of: a segment of neural
Development of the Axial Skeleton and Limbs Professor Alfred Cuschieri Department of Anatomy University of Malta During the Fourth Week the Embryo Is Segmented. Each segment consists of: a segment of neural
Differential requirement for Gli2 and Gli3 in ventral neural cell fate specification
 Available online at www.sciencedirect.com R Developmental Biology 259 (2003) 150 161 www.elsevier.com/locate/ydbio Differential requirement for Gli2 and Gli3 in ventral neural cell fate specification Jun
Available online at www.sciencedirect.com R Developmental Biology 259 (2003) 150 161 www.elsevier.com/locate/ydbio Differential requirement for Gli2 and Gli3 in ventral neural cell fate specification Jun
The specification of neuronal identity by graded sonic hedgehog signalling
 seminars in CELL & DEVELOPMENTAL BIOLOGY, Vol 10, 1999: pp. 353 362 Article No. scdb.1999.0295, available online at http: www.idealibrary.com on The specification of neuronal identity by graded sonic hedgehog
seminars in CELL & DEVELOPMENTAL BIOLOGY, Vol 10, 1999: pp. 353 362 Article No. scdb.1999.0295, available online at http: www.idealibrary.com on The specification of neuronal identity by graded sonic hedgehog
THE ROLE OF CONNECTIVE TISSUE GROWTH FACTOR IN ISLET. Michelle Guney. Dissertation. Submitted to the Faculty of the
 THE ROLE OF CONNECTIVE TISSUE GROWTH FACTOR IN ISLET MORPHOGENESIS AND β CELL PROLIFERATION By Michelle Guney Dissertation Submitted to the Faculty of the Graduate School of Vanderbilt University In partial
THE ROLE OF CONNECTIVE TISSUE GROWTH FACTOR IN ISLET MORPHOGENESIS AND β CELL PROLIFERATION By Michelle Guney Dissertation Submitted to the Faculty of the Graduate School of Vanderbilt University In partial
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Formation of the AA5x. a, Camera lucida drawing of embryo at 48 hours post fertilization (hpf, modified from Kimmel et al. Dev Dyn. 1995 203:253-310). b, Confocal microangiogram
Supplementary Figure 1. Formation of the AA5x. a, Camera lucida drawing of embryo at 48 hours post fertilization (hpf, modified from Kimmel et al. Dev Dyn. 1995 203:253-310). b, Confocal microangiogram
Prolonged FGF signaling is necessary for lung and liver induction in Xenopus
 Shifley et al. BMC Developmental Biology 2012, 12:27 RESEARCH ARTICLE Open Access Prolonged FGF signaling is necessary for lung and liver induction in Xenopus Emily T Shifley 1, Alan P Kenny 2, Scott A
Shifley et al. BMC Developmental Biology 2012, 12:27 RESEARCH ARTICLE Open Access Prolonged FGF signaling is necessary for lung and liver induction in Xenopus Emily T Shifley 1, Alan P Kenny 2, Scott A
Studying pancreas development and diabetes using human pluripotent stem cells
 Editorial Studying pancreas development and diabetes using human pluripotent stem cells David W. Scoville, Anton M. Jetten Cell Biology Group, Immunity, Inflammation, and Disease Laboratory, National Institute
Editorial Studying pancreas development and diabetes using human pluripotent stem cells David W. Scoville, Anton M. Jetten Cell Biology Group, Immunity, Inflammation, and Disease Laboratory, National Institute
Wnt signaling. Ramray Bhat.
 Wnt signaling Ramray Bhat ramray@mrdg.iisc.ernet.in Starting with animal biology and viral infections The discovery of certain laboratory murine strains that were highly susceptible to mammary gland cancer.
Wnt signaling Ramray Bhat ramray@mrdg.iisc.ernet.in Starting with animal biology and viral infections The discovery of certain laboratory murine strains that were highly susceptible to mammary gland cancer.
The Foregut. At first the esophagus is short. but with descent of the heart and lungs it lengthens rapidly
 GI embryology 2 The Foregut At first the esophagus is short but with descent of the heart and lungs it lengthens rapidly The muscular coat, which is formed by surrounding splanchnic mesenchyme, is striated
GI embryology 2 The Foregut At first the esophagus is short but with descent of the heart and lungs it lengthens rapidly The muscular coat, which is formed by surrounding splanchnic mesenchyme, is striated
Chapter 11 Development and Diseases
 Chapter 11 Development and Diseases Fertilization Development of the Nervous System Cleavage (Blastula, Gastrula) Neuronal Induction- Neuroblast Formation Cell Migration Axon Growth/Target innervation
Chapter 11 Development and Diseases Fertilization Development of the Nervous System Cleavage (Blastula, Gastrula) Neuronal Induction- Neuroblast Formation Cell Migration Axon Growth/Target innervation
Endocrine System Hormones (Ch. 45)
 Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
Endocrine System Hormones (Ch. 45) Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body daily homeostasis & regulation of large
Segmentation. Serial Homology. Hox genes
 Segmentation Hox genes Serial Homology William Bateson 1861-1926 Homeosis: a variation in which something has been changed into the likeness of something else Calvin Bridges at Columbia bithorax 1923 1
Segmentation Hox genes Serial Homology William Bateson 1861-1926 Homeosis: a variation in which something has been changed into the likeness of something else Calvin Bridges at Columbia bithorax 1923 1
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Pancreas and beta-cell development: from the actual to the possible
 REVIEW 427 Development 134, 427-438 (2007) doi:10.1242/dev.02770 Pancreas and beta-cell development: from the actual to the possible L. Charles Murtaugh The development of insulin-producing pancreatic
REVIEW 427 Development 134, 427-438 (2007) doi:10.1242/dev.02770 Pancreas and beta-cell development: from the actual to the possible L. Charles Murtaugh The development of insulin-producing pancreatic
Development of Spinal Cord & Vertebral Column. Dr. Sanaa Alshaarawi & Prof. Ahmed Fathalla
 Development of Spinal Cord & Vertebral Column Dr. Sanaa Alshaarawi & Prof. Ahmed Fathalla OBJECTIVES At the end of the lecture, students should be able to: q Describe the development of the spinal cord
Development of Spinal Cord & Vertebral Column Dr. Sanaa Alshaarawi & Prof. Ahmed Fathalla OBJECTIVES At the end of the lecture, students should be able to: q Describe the development of the spinal cord
Shh signaling guides spatial pathfinding of raphespinal tract axons by multidirectional repulsion
 ORIGINAL ARTICLE Cell Research (2012) 22:697-716. 2012 IBCB, SIBS, CAS All rights reserved 1001-0602/12 $ 32.00 www.nature.com/cr npg Shh signaling guides spatial pathfinding of raphespinal tract axons
ORIGINAL ARTICLE Cell Research (2012) 22:697-716. 2012 IBCB, SIBS, CAS All rights reserved 1001-0602/12 $ 32.00 www.nature.com/cr npg Shh signaling guides spatial pathfinding of raphespinal tract axons
Pancreas Organogenesis: From Bud to Plexus to Gland
 a DEVELOPMENTAL DYNAMICS 240:530 565, 2011 SPECIAL ISSUE REVIEWS A PEER REVIEWED FORUM Pancreas Organogenesis: From Bud to Plexus to Gland Fong Cheng Pan and Chris Wright* Pancreas oganogenesis comprises
a DEVELOPMENTAL DYNAMICS 240:530 565, 2011 SPECIAL ISSUE REVIEWS A PEER REVIEWED FORUM Pancreas Organogenesis: From Bud to Plexus to Gland Fong Cheng Pan and Chris Wright* Pancreas oganogenesis comprises
Inner ear development Nervous system development
 Upcoming Sessions April 22: Nervous System Development Lecture April 24: Reviews of Axonal Pathfinding in Sensory Systems April 29: Inner Ear Development Lecture May 1: May 6: May 8: Auditory System Pathfinding
Upcoming Sessions April 22: Nervous System Development Lecture April 24: Reviews of Axonal Pathfinding in Sensory Systems April 29: Inner Ear Development Lecture May 1: May 6: May 8: Auditory System Pathfinding
Development of Brain Stem, Cerebellum and Cerebrum
 Development of Brain Stem, Cerebellum and Cerebrum The neural tube cranial to the 4th pair of somites develop into the brain. 3 dilatations and 2 flexures form at the cephalic end of the neural tube during
Development of Brain Stem, Cerebellum and Cerebrum The neural tube cranial to the 4th pair of somites develop into the brain. 3 dilatations and 2 flexures form at the cephalic end of the neural tube during
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Supplementary Figures Supplementary Figure 1. nrg1 bns101/bns101 embryos develop a functional heart and survive to adulthood (a-b) Cartoon of Talen-induced nrg1 mutation with a 14-base-pair deletion in
Early Embryonic Development
 Early Embryonic Development Maternal effect gene products set the stage by controlling the expression of the first embryonic genes. 1. Transcription factors 2. Receptors 3. Regulatory proteins Maternal
Early Embryonic Development Maternal effect gene products set the stage by controlling the expression of the first embryonic genes. 1. Transcription factors 2. Receptors 3. Regulatory proteins Maternal
Early Development of Neural Tube Development of Medulla Spinalis and Peripheral Nervous System. Assoc.Prof. E.Elif Güzel, M.D.
 Early Development of Neural Tube Development of Medulla Spinalis and Peripheral Nervous System Assoc.Prof. E.Elif Güzel, M.D. Third week of Embryogenesis Primitive streak/pit appears on the epiblast (day
Early Development of Neural Tube Development of Medulla Spinalis and Peripheral Nervous System Assoc.Prof. E.Elif Güzel, M.D. Third week of Embryogenesis Primitive streak/pit appears on the epiblast (day
Branching morphogenesis of the lung: new molecular insights into an old problem
 86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
Translational Embryology: Using Embryonic Principles to Generate Pancreatic Endocrine Cells From Embryonic Stem Cells
 DEVELOPMENTAL DYNAMICS 236:3218 3227, 2007 SPECIAL ISSUE REVIEWS A PEER REVIEWED FORUM Translational Embryology: Using Embryonic Principles to Generate Pancreatic Endocrine Cells From Embryonic Stem Cells
DEVELOPMENTAL DYNAMICS 236:3218 3227, 2007 SPECIAL ISSUE REVIEWS A PEER REVIEWED FORUM Translational Embryology: Using Embryonic Principles to Generate Pancreatic Endocrine Cells From Embryonic Stem Cells
Supplementary Figure S1. Monolayer differentiation of mouse ESCs into telencephalic neural precursors. (a) Schematic representation of the protocols
 Supplementary Figure S1. Monolayer differentiation of mouse ESCs into telencephalic neural precursors. (a) Schematic representation of the protocols used to differentiate mouse ESCs. (b) Representative
Supplementary Figure S1. Monolayer differentiation of mouse ESCs into telencephalic neural precursors. (a) Schematic representation of the protocols used to differentiate mouse ESCs. (b) Representative
Douglas J. Epstein 1, *, Andrew P. McMahon 2 and Alexandra L. Joyner 1,3, SUMMARY
 Development 126, 281-292 (1999) Printed in Great Britain The Company of Biologists Limited 1998 DEV4093 281 Regionalization of Sonic hedgehog transcription along the anteroposterior axis of the mouse central
Development 126, 281-292 (1999) Printed in Great Britain The Company of Biologists Limited 1998 DEV4093 281 Regionalization of Sonic hedgehog transcription along the anteroposterior axis of the mouse central
Problem Set 5 KEY
 2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
