ORIGINAL CONTRIBUTION. Ictal Fear in Temporal Lobe Epilepsy
|
|
|
- Roderick Powell
- 5 years ago
- Views:
Transcription
1 ORIGINAL CONTRIBUTION Ictal Fear in Temporal Lobe Epilepsy Surgical Outcome and Focal Hippocampal Changes Revealed by Proton Magnetic Resonance Spectroscopy Imaging Michael Feichtinger, MD; Elisabeth Pauli, PhD; Iris Schäfer; Knut W. Eberhardt, MD; Bernd Tomandl, MD; Johannes Huk, MD; Hermann Stefan, MD Background: Ictal fear (IF) is most frequently associated with epileptic discharges from mesial temporal areas. Objectives: To determine whether patients with IF were more likely to become seizure free after anteromesial temporal lobe resection compared with those without IF and whether they show more anteriorly pronounced metabolic changes assessed by means of multivoxel magnetic resonance spectroscopy (MRS) along the hippocampal axis. Methods: Surgical outcome was assessed in 33 consecutive patients with temporal lobe epilepsy after a mean follow-up of 25 months (range, months). Proton multivoxel MRS of the hippocampal formation was applied to detect regional differences along the axis of the hippocampus in patients with and without IF. Magnetic resonance tomography showed typical features of hippocampal sclerosis in all patients. Results: Twelve (36%) of the 33 patients reported fear at the beginning of their habitual seizures. Eleven of these patients were seizure free postoperatively. In contrast, only 11 of 21 patients without IF had a favorable outcome. Results of MRS revealed significantly higher pathologic N-acetylaspartate choline ratios in the anterior portion of the hippocampal formation in patients with than in those without IF, indicating focal metabolic and/or morphologic changes in the head of the hippocampus. Conclusions: These results indicate the importance of diagnosing auras with IF to provide a more detailed prognosis of the surgical outcome. In addition, our data emphasize that multivoxel MRS is a valuable tool in the presurgical evaluation, as it may reveal different topographical patterns of hippocampal sclerosis. Arch Neurol. 2001;58: From the Department of Neurology, University Erlangen Nuremberg, Erlangen, Germany. FEAR IS AMONG the most common experiential phenomena elicited by temporal lobe epileptic discharge. 1-7 The leading role of the amygdala, as well as that of the hippocampus and the parahippocampal gyrus, in the mechanisms of inducing a fearful perception is well established by studies of electrical stimulation performed intraoperatively 8 or during presurgical evaluation using intracranial depth electrodes. 2,9-12 Surgical resection of the temporal lobe became a widely accepted therapeutic approach to control complex partial seizures. 13 In patients with fear as an initial manifestation of their epileptic seizures (IF patients), ictal discharges were reported to originate from the amygdala, the hippocampus, or its near vicinity, 2,14 ie, regions that are removed by temporal lobectomy. We therefore investigated whether the presence of IF may predict a favorable outcome of surgical therapy. Proton ( 1 H) magnetic resonance spectra obtained by magnetic resonance spectroscopic imaging (MRSI) provide detailed information about metabolic changes of the mesial temporal region using signals from N-acetylaspartate (NAA)-, choline (Cho)-, and creatine (Cr)-containing compounds Because NAA is primarily located in mature neurons, a reduced NAA signal is generally interpreted as neuronal loss or dysfunction Concentrations of Cho and Cr are reported to be higher in the glia 22,23 ; thus, a decrease of levels of Cho and, to a lesser extent, Cr 24 may indicate astrocytosis. Unilateral changes (a reduced NAA/ Cho ratio) have been shown to be of value for preoperative focus localization in epilepsy surgery. 16,17,20,24,25 Because atrophy of the amygdala and the hippocampus correlates with the presence of IF, 26 analysis of these structures by means of MRS could confirm the involvement of these regions in the perception of IF. Unfortunately, MRS of the amygdala is not applicable because of its small size, its difficult differentiation to adjacent structures, 27,28 and the interference of neighboring cerebral and extracerebral tis- 771
2 PATIENTS AND METHODS PATIENTS We included 33 consecutive patients with unilateral medically intractable temporal lobe epilepsy (TLE) (19 male and 14 female) considered for surgical treatment at the Epilepsy Center of the Department of Neurology, University Erlangen Nuremberg, Erlangen, Germany. The mean age was 34 years (range, years). The epileptogenic temporal lobe was identified by means of a comprehensive evaluation, including a detailed history and neurologic examination, prolonged electroencephalographic and video monitoring with scalp and sphenoidal electrodes for ictal analysis, neuropsychological assessment, and MRI. In 5 patients, electrodes for recording intracranially ictal activity were implanted. Only patients with distinct unilateral focus within the temporal lobe were scheduled for epilepsy surgery. These patients were also included in the current study, since they fulfilled the following inclusion criteria: (1) typical seizure symptoms for TLE, (2) all seizures arising from a single temporal lobe, and (3) typical findings of mesial temporal sclerosis on MRI (ie, atrophy and/or decreased signal on T1- and/or increased signal on T2-weighted images of the hippocampus and/or amygdala). Thus, these patients represented a homogeneous group of good candidates for temporal lobectomy. This homogeneity is crucial for determination of whether IF is a prognostic variable independent from already established predicting factors. SURGICAL PROCEDURE All 33 patients underwent anteromesial temporal resection performed by the same neurosurgeon. All patients received at least a selective amygdalohippocampectomy, including the parahippocampal gyrus. Additional neocortical resection was individually tailored, guided by intraoperative electrocorticography to keep resected brain volume restricted to epileptogenic tissue. 30 The mean extent of the resected hippocampus was 24 mm (range, mm). Postoperative seizure control outcome was assessed 3 and 6 months and each year after the operation, using the classification according to Engel. 13 Only patients with a follow-up of at least 12 months were included in the study. For further statistical analysis, outcome was divided into a seizure-free group (Engel class 1) and a non seizurefree group (Engel classes 2-4). PATHOLOGICAL FINDINGS All available brain specimens were prepared and stained for histopathological examination performed by a pathologist blinded to the results of MRSI. Hippocampal sclerosis (HS) was defined as neuronal loss and marked proliferation of astrocytes involving the hippocampus, preferentially the sectors CA1 and CA3/4. In 3 cases, hippocampal sections did not allow a proper diagnosis of an HS owing to fragmentation of the specimen. Histopathological analysis of the amygdala was not available. CHARACTERIZATION AND CLASSIFICATION OF IF AND AURAS Assessment of the presence of IF included a detailed review of available medical records and an accurate inquiry about the type of auras. A standardized interview was used, including the assessment of fear and fear-related emotional, cognitive, and autonomous signs as part of an aura or in the course of a habitual seizure. Care was taken to avoid suggestive questioning. For further investigation, the term ictal fear was accepted only (1) if it was reported as being concomitant with an epileptic seizure; (2) if it arose spontaneously, out of context; and (3) if it could be clearly distinguished from the fear of a seizure. Type and presence of any other sensations or phenomena associated with seizures were assessed in a similar manner. ROUTINE MRI Routine MRI examinations were performed on a 1.5-T whole-body MR system (Siemens Magnetom Vision; Siemens, Erlangen, Germany) according to a standard protocol, including transversal and coronal T2-weighted targeted systemic exposure, coronal fluid-attenuated inversion recovery, coronal T1-weighted 3-dimensional magnetization-prepared rapid acquisition gradient echo, and coronal inversion recovery. Atrophy of the hippocampus and/or amygdala seen by means of visual inspection and/or as hyperintensity inside the hippocampus on T2- weighted images was taken as criterion for mesial temporal sclerosis. MULTIVOXEL 1 H-MRSI During presurgical evaluation, 27 patients underwent multivoxel 1 H-MRSI of the hippocampal formation after written informed consent was obtained. We acquired MRSI using a 1.5-T whole-body MR system (Siemens Magnetom Vision). To establish normative data, an additional 30 healthy volunteers (17 male and 13 female subjects; age range, years; mean age, 31 years) underwent MRSI examinations. Acquisition of MR spectra was performed using a 2-dimensional (2-D) hybrid chemical shift imaging (CSI) technique. Three orthogonal slice-selective high-frequency pulses were applied for preselection of a region of excitation, avoiding the intensive signal of subcutaneous fat. Phase encoding (16 16 matrix) was used for in-plane spatial resolution and chemical shift-selective gaussian pulses for water suppression. To optimize the homogeneity of the sue. 29 Thus, we focused on the hippocampal formation. Since sclerosis of amygdala and hippocampus was observed more often in IF patients, 26 we hypothesized that in these patients, neuronal loss and astrogliosis could be more pronounced in the head of the hippocampus. In other words, MRSI analysis aimed to detect regional differences in hippocampal metabolism. RESULTS Twelve patients (36%) reported fear as an initial manifestation of their habitual seizures. Five of these patients described an accompanying epigastric sensation; 2 patients, a cephalic aura; and 3 patients, fear as a single phenomenon. The remaining symptoms included déja vu 772
3 Postoperative outcome assessment was possible in all 33 patients (average postoperative period, 25 months [range, months]). According to the Engel classification, 22 patients became seizure free (Engel class 1), 8 patients had rare disabling seizures (Engel class 2), 1 pamagnetic field, an automatic multiangle-projection shim was applied. A series of orthogonal T1-weighted images for localization were obtained, with the transaxial plane approximately parallel to the long axis of the hippocampus. The preselected region of excitation was positioned on 3 orthogonal base images in the left and right temporal lobe. The volume of interest was chosen, including a column of 3 voxels (voxels 1-3) in the transversal plane placed in the hippocampal formation. As shown in Figure 1, this column was chosen to obtain data mainly from the hippocampus with little or no interference from adjacent regions. The position was carefully checked and meticulously adjusted in all 3 orthogonal sections to ensure identical placement in all subjects. After interactive volume-selective shimming and water suppression (frequency-selective 90 Gauss pulse), a 2-D 1 H CSI spinecho protocol was selected (acquisition variables: repetition time, 1500 milliseconds; echo time, 135 milliseconds; field of view, 200 mm 2 ; matrix, 16 16). The nominal resultant size of individual voxels was cm in the sagittal and coronal planes and 1.5 cm in the transversal plane (2.16 cm 3 ). The acquisition time for each hippocampus was 6 minutes 31 seconds. The total time required for MRS measurement was about 45 minutes. MRS DATA PROCESSING The CSI raw data were evaluated using a standard automatic CSI postprocessing program (Siemens Magnetom Vision). Spectra were displayed on transversal T1- weighted MRI scans with an overlaid grid indicating the anatomic location from where the data were derived. The postprocessing protocol used k-space filtering, 2-D fast Fourier transformation to real space, gaussian filtering of the time-domain data in each voxel, fast Fourier transformation to the frequency domain, baseline and phase correction, and curve fitting for the peaks of NAA and Cho. The ratios of the fitted peak integrals for NAA and Cho were evaluated and integration of the Cr peak was omitted. Although many studies included Cr for assessment, we preferred the NAA/Cho ratio, as this ratio is reported to be highly reliable in detecting HS in TLE. 16,20,24 Spectra of poor quality were excluded from the analyses, for instance, when Cr and Cho peaks were not resolved. The NAA/Cho ratios of each voxel were compared with the average ratio of the corresponding, homotopically located voxel in the control subjects. Pathologic voxel values (P X patient ) in patients were defined as differences between the patient s NAA/Cho ratio (R) of the specific voxel (X = 1-3) and the 2-SD threshold of the corresponding voxel (X = 1-3) in controls, 20,31 as seen in the following equation: P X patient =[M(R Xcontrol ) 2SD(R Xcontrol )] R X patient, where M indicates the mean voxel value of the controls. Deviation from the healthy controls is more pronounced as the negative pathologic value (P X patient ) increases. The value 0 was assigned when no abnormality was found. A CSI score representing the mean pathologic values of voxels 1, 2, and 3 (,, and ) was calculated using the following equation 20,31,32 : CSI=(P + P + P )/ 3. To determine ipsilateral, contralateral, and bilateral abnormalities, we computed the laterality ratio (r) using the following equation: r = (CSI ipsi CSI contra )/(CSI ipsi + CSI contra ), where ipsi and contra refer to the ipsilateral and contralateral sides of resection. Thus, a CSI laterality ratio of r =1 reflects ipsilateral CSI changes, and a ratio of r = 1 reflects exclusively contralateral CSI changes. A ratio of r = 0 corresponds to bilateral CSI changes but would also be seen in cases of no abnormality. For practical reasons, results were classified as strictly ipsilateral (0.25 r 1), strictly contralateral ( 0.25 r 1), and bilateral ( 0.25 r 0.25). To evaluate whether anterior parts of the hippocampus (predominantly the head) show metabolic changes different from those of the posterior parts, separate comparison of single-voxel data (P, P, and P ) in patients with and without IF was performed. To assess the prevalence of different distribution patterns, we used the following equation to compute difference score (I ap ) of the pathologic values of the anterior (P and P ) and posterior voxels (P ): STATISTICAL ANALYSIS I ap =[(P +P )/2] P. Statistical analysis was performed using commercially available software (SPSS E 8.0 for Windows 95; SPSS Inc, Chicago, Ill). Inference statistical decisions were based on a significance level of.05. We performed 2 tests (Fisher exact test) to evaluate the relation between the presence or absence of IF with seizure outcome and the prevalence of different spectroscopic results. For evaluation of the severity of abnormality ipsilateral to the side undergoing operation in IF and non-if patients, 1-way analysis of variance (ANOVA) with IF as between-subject factor was computed separately for the following dependent variables: CSI score; pathologic values of voxels 1, 2, and 3; and the NAA/Cho ratio of voxels 1, 2, and 3. and autonomous signs in 1 case each. Between IF and non-if patients, there was no difference in age at operation, duration of epilepsy, or age at onset of epilepsy. The extent of resected hippocampus did not differ between patient groups. There was no significant difference between IF and non-if patients with regard to results of histopathological examination (ie, presence or absence of HS). IF AND OUTCOME 773
4 A 12 Seizure Free Not Seizure Free 10 No. of Patients Non-IF Patients IF Patients Right Figure 2. Relationship between ictal fear (IF) and seizure outcome. B 3.0 Cho NAA Cho NAA NAA/Cho Ratio Control Subjects (n = 30) Non-IF Patients (n = 17) IF Patients (n = 10) Normal Pathologic Figure 1. A, Proton magnetic resonance spectroscopic imaging ( 1 H-MRSI) regions of interest in patient 2 with ictal fear and an epileptic focus within the right mesiotemporal region. Transversal T1-weighted MRI scan with the original phase-encoding grid superimposed is seen. The volume of interest was outlined (black line). Spectra of voxels 1 to 3 (,, and ) placed exactly in the hippocampal formation are displayed. In the 2 anterior voxels ( and ), the peak level of N-acetylaspartate (NAA) is clearly reduced compared with that of choline (Cho), leading to a lower NAA/Cho ratio. This indicates focal accentuated neuronal loss (or dysfunction) and/or reactive gliosis in the anterior portion of the hippocampus. B, Demonstration of a normal vs a pathologic spectrum. tient had an improvement in seizure frequency of greater than 80% (Engel class 3), and 2 patients reported no improvement (Engel class 4). Eleven of 12 IF patients had an excellent outcome, whereas only 11 of 21 non-if patients were classified as seizure free (Engel class 1) (2-sided Fisher exact test, P=.03) (Figure 2). No other aura showed a clear relation to postoperative outcome. PATHOLOGIC CSI SCORES AND THEIR RELATION TO HS AND IF In 24 patients, comparison of ipsilateral CSI scores with the histopathological diagnosis of an HS was possible. Pathologic CSI scores were measured in 10 (59%) of 17 cases with histologically proven HS; normal CSI scores were found in 6 of 7 patients without HS. Two of 3 cases with insufficient specimen for histological analysis also showed pathologic CSI scores ipsilaterally. Figure 3. Box and whisker plot of the N-acetylaspartate choline (NAA/Cho) ratio of each voxel (,, and ) in patients with and without ictal fear (IF) compared with healthy control subjects. White boxes represent the 1-SD range; the whiskers, the 2-SD range of NAA/Cho ratios of controls. The black line is positioned at the median. Shaded boxes extend from the 25th to the 75th percentile with a horizontal black line at the median in the IF and non-if patient groups. The whiskers represent the range of the data; small black squares, extremes. Lower NAA/Cho ratios in voxels 1 and 2 are seen in the IF group compared with the non-if group, reflecting a higher degree of neuron loss and/or reactive gliosis in the anterior part of the hippocampus of these patients. Concerning the frequency of bilateral or contralateral CSI score changes, no significant difference could be found between the IF and the non-if patient groups. IPSILATERAL NAA/Cho RATIOS AND IF Separate comparison of the ipsilateral NAA/Cho ratio in voxels 1, 2, and 3 revealed marked differences between IF and non-if patients; results of 1-way ANOVA showed significantly lower ratios in the anterior part of the hippocampus (voxels 1 and 2) in IF patients (voxel 1, F 1,25 =4.78 [P=.04]; voxel 2, F 1,26 =9.29 [P=.005]) (Figure 3 and Table 1). IPSILATERAL PATHOLOGIC CSI VALUES AND IF A similar relation was found when ipsilateral pathologic values for each voxel (P, P, and P ) were compared between groups (voxel 1, F 1,25 =5.93 [P=.02]; voxel 2, F 1,26 = [P=.001]) (Table 1). Pathologic values of voxel 3 did not differ significantly between IF and non-if patients. 774
5 The mean pathologic value of all 3 voxels (CSI score) also showed significantly lower scores (ie, more abnormal compared with the controls 2-SD threshold) in IF patients compared with non-if patients (1-way ANOVA, F 1,26 =5.41 [P=.03]) (Table 1). In all 6 IF patients with a pathologic CSI score, changes in the anterior hippocampus (I ap 0) were more pronounced, whereas in 6 of 7 non-if patients, abnormality was predominantly located posteriorly (I ap 0). One non-if patient had higher pathologic values in the anterior hippocampus (2-sided Fisher exact test, P=.005) (Table 2). COMMENT There is no doubt that the mesial temporal regions and especially the amygdala play a pivotal role in the mediation of fearful emotion. This is supported by numerous studies on the production of fear by electrical stimulation of the amygdala. 2,3,11 Stimulation of the hippocampus and the parahippocampal gyrus produced fear less often. 2,12 However, this research indicates that these structures are involved in the mediation of fear, at least under certain circumstances. We hypothesized that the presence of IF could be related to a satisfying seizure outcome after surgical treatment, when resections include the amygdala, the hippocampus, and the parahippocampal gyrus. Our study confirmed this hypothesis in demonstrating a close relation between favorable postoperative seizure control and the presence of IF. This was not seen for any other aura. Previous studies 33,34 have addressed the prognostic significance of the type of aura concerning postoperative seizure control and could not find any aura to be of predictive value. This discrepancy might be explained by differences in patient selection criteria, 34 classification of aura types, 33 or assessment of IF. The term ictal fear should be used exclusively for a sudden, often short, fearful affect at the beginning of or during an epileptic seizure, without context or any relation to a precedent causal perception or cognition. The intensity may vary from a slight trace of anxiety to a feeling of intense terror. 27 The predominance of other auras or anterograde amnesia may reduce subjective awareness of fear in some individuals. Thus, only a direct and standardized questioning, as performed in our study, enables the most reliable assessment of IF. Another interesting relation to the presence of IF could be detected when we analyzed 1 H-MRS data of the ipsilateral hippocampus (related to the side that underwent operation). The IF patients had significantly higher pathologic values in the anterior 2 voxels (voxels 1 and 2). These results indicate different distribution patterns concerning the degree of neuronal loss (or dysfunction) and reactive astrogliosis along the hippocampal axis in IF and non-if patients. On the basis of postmortem 35 or postoperative 36,37 histopathological examination, topographical differences of neuronal loss within the hippocampus have been described repeatedly. When qualitative and volumetric MRI techniques were applied for differentiation between focal and diffuse HS, contradicting results were reported. Several studies described Table 1. NAA/Cho Ratio, Pathologic Values for Voxels, and CSI Score in Relation to Ictal Fear* Control Group (n = 30) IF Patients (n = 10) Non-IF Patients (n = 17) NAA/Cho ratio Voxel ± Voxel ± Voxel ± Pathologic values Voxel Voxel Voxel CSI score *NAA indicates N-acetylaspartate; Cho, choline; CSI, chemical shift imaging; and IF, ictal fear. Values represent mean NAA/Cho ratio±1sd. P.05. Values represent mean pathologic value of voxels 1 to 3 in IF and non-if groups; pathologic voxel values are calculated as difference between each patient s NAA/Cho ratio of the specific voxel (1 to 3) and the 2-SD threshold of the corresponding voxel of controls. Lower score stands, therefore, for higher pathologic metabolic changes in voxel area compared with healthy control subjects. Calculated as ( P + P + P )/3 where P indicates pathologic values;, voxel 1,, voxel 2; and, voxel 3. Table 2. Frequencies of Different Distribution Patterns in Patients With and Without Ictal Fear* Distribution Pattern IF Patients Non-IF Patients Anterior (I ap 0) 6 1 Posterior (I ap 0) 0 6 *IF indicates ictal fear. P =.005, 2-sided Fisher exact test. Calculated applying a difference score ( I ap ) between pathological value of the 2 anterior and the posterior voxels ( P, P, and P ), according to the following formula: I ap =[(P + P )/2] P. Because pathologic voxel values have negative signs, I ap 0 refers to predominantly anterior abnormality and I ap 0 refers to posteriorly accentuated abnormality. a focal accentuated atrophy in the anterior portion of the hippocampus in most of their patients. However, 2 studies 41,42 reported diffuse atrophy to be the most frequent HS variant. Actually, it is controversial whether a different distribution of sclerotic changes bears any significance for seizure outcome. Kim et al 41 suggested that distinction of focal and diffuse abnormality did not appear to be essential for predicting postoperative seizure control. In contrast, Babb et al 36,37 classified the following 2 patterns of HS with implications concerning surgical outcome: a diffuse pattern (ie, equally distributed neuronal loss in the hippocampal head and body) with less favorable surgical outcome, and a focal pattern (neuronal loss predominantly in the head of the hippocampus). The present study confirms the latter observation and stresses the value of the assessment of different distribution patterns. That IF is related to focal anterior abnormality is important, as the resected brain volume should be restricted to epileptogenic tissue. Moreover, the extent of resection influences the degree of postoperative neuropsychological dysfunction. 43,44 Previous studies assessing the value of MRS in TLE have paid little attention to the detection of different 775
6 regional distribution patterns along the hippocampal axis. Vermathen et al 45 reported a strong anteriorposterior difference within the hippocampus in patients with epilepsy and in healthy individuals using multivoxel 1 H-MRSI. Similar observations were made by Hsu et al. 32 When analyzing our control data, we could also observe an anterior-posterior gradient with a higher NAA/Cho ratio in the posteriorly located voxels. We took this into account when we calculated the pathologic voxel values. Because the total volume of all 3 selected voxels is somewhat larger ( 6.48 cm 3 ) than the estimated volume of the hippocampus (approximately 4 cm 3 ), a certain influence of adjacent tissue could not be prevented. This may be particularly true in cases with atrophied hippocampus. To assess whether this volume loss influences MRS data, further correlation studies between MRSI and volumetric measurements along the hippocampal axis are needed. In our patient population, atrophy (assessed by means of visual inspection) was not related to a higher prevalence of pathologic MRSI spectra. When looking for differences along the hippocampal axis in IF patients, we assumed the anterior voxel (voxel 1) would show the highest pathologic values because of its close vicinity to the amygdala. Indeed, our results demonstrate a significant relation of anterior abnormality in the IF patients, which allows speculations about a concomitant sclerosis of the amygdala. The fact that voxels 1 and 2 are similarly associated with IF argues for higher involvement of the anterior hippocampal portion in the mechanisms of fear than previously presumed. Interestingly, Miller et al 46 found IF in a higher percentage when amygdala and hippocampus were both sclerotic compared with sole sclerosis of amygdala. Actually, several authors estimated sclerosis of the amygdala to be combined with HS in up to 60% of patients. 28,47-50 To confirm our findings, future studies concerning the correlation between amygdala volume and spectroscopic findings of the hippocampus and studies on higher numbers of patients (to rule out possible sample bias due to small patient number) are currently planned. CONCLUSIONS Our study supports the concept that IF is related to a measurable abnormality within the anterior mesial temporal regions. It underlines the role of the hippocampus in the presence of this affective ictal state and emphasizes the importance of MRS for detecting different topographical patterns of HS. Our observation that IF patients represent a population with favorable seizure relief after anteromesial lobectomy may be clinically significant when planning surgical therapy and when counseling patients with TLE. Accepted for publication October 4, Corresponding author and reprints: Hermann Stefan, MD, Department of Neurology, University Erlangen Nuremberg, Schwabachanlage 6, D Erlangen, Germany ( hermann.stefan@neuro.med.uni-erlangen.de). REFERENCES 1. Daly DD. Ictal clinical manifestations of complex partial seizures. Adv Neurol. 1975;11: Gloor P. Temporal lobe epilepsy: its possible contribution to the understanding of the functional significance of the amygdala and of its interaction with neocorticaltemporal mechanisms. In: Eleftheriou B, ed. The Neurobiology of the Amygdala: Advances in Behavioural Biology. New York, NY: Plenum Publishing Corp; 1972: Gloor P, Olivier A, Quesney LF, Andermann F, Horowitz S. The role of the limbic system in experiential phenomena of temporal lobe epilepsy. Ann Neurol. 1982; 12: Gloor P. Experiential phenomena of temporal lobe epilepsy: facts and hypotheses. Brain. 1990;113: Gloor P. Neurobiological substrates of ictal behavioral changes. Adv Neurol. 1991; 55: Taylor DC, Lochery M. Temporal lobe epilepsy: origin and significance of simple and complex auras. J Neurol Neurosurg Psychiatry. 1987;50: Wieser HG. Depth recorded limbic seizures and psychopathology. Neurosci Biobehav Rev. 1983;7: Feindel W, Penfield W. Localization of discharge in temporal lobe automatism. Arch Neurol Psychiatry. 1954;72: Fish DR, Gloor P, Quesney FL, Olivier A. Clinical responses to electrical brain stimulation of the temporal and frontal lobes in patients with epilepsy: pathophysiological implications. Brain. 1993;116: Gloor P, Olivier A, Quesney FL. The role of the amygdala in the expression of psychic phenomena in temporal lobe seizures. In: Ben-Ari Y, ed. The Amygdaloid Complex. Amsterdam, the Netherlands: Elsevier Science Publishers; 1981: Gloor P. Role of the amygdala in temporal lobe epilepsy. In: Aggleton JP, ed. The Amygdala: Neurobiological Aspects of Emotion, Memory, and Mental Dysfunction. New York, NY: John Wiley & Sons Inc; 1992: Halgren E, Walter RD, Cherlow DG, Crandall PH. Mental phenomena evoked by electrical stimulation of the human hippocampal formation and amygdala. Brain. 1978;101: Engel J Jr. Outcome with respect to epileptic seizures. In: Engel J Jr, ed. Surgical Treatment of the Epilepsies. New York, NY: Raven Press; 1993: Palmini A, Gloor P. The localizing value of auras in partial seizures: a prospective and retrospective study. Neurology. 1992;42: Cendes F, Andermann F, Preul MC, Arnold DL. Lateralization of temporal lobe epilepsy based on regional metabolic abnormalities in proton magnetic resonance spectroscopic images. Ann Neurol. 1994;35: Ende GR, Laxer KD, Knowlton RC, et al. Temporal lobe epilepsy: bilateral hippocampal metabolite changes revealed at proton MR spectroscopic imaging. Radiology. 1997;202: Gadian DG, Connelly A, Duncan JS, et al. 1 H magnetic resonance spectroscopy in the investigation of intractable epilepsy. Acta Neurol Scand Suppl. 1994;152: Hugg JW, Laxer KD, Matson GB, Maudsley AA, Weiner MW. Neuron loss localizes human temporal lobe epilepsy by in vivo proton magnetic resonance spectroscopic imaging. Ann Neurol. 1993;34: Matthews PM, Andermann F, Arnold DL. A proton magnetic resonance spectroscopy study of focal epilepsy in humans. Neurology. 1990;40: Ng TC, Comair YG, Xue M, et al. Temporal lobe epilepsy: presurgical localization with proton chemical shift imaging. Radiology. 1994;193: Birken DL, Oldendorf WH. N-Acetyl-L-aspartic acid: a literature review of a compound prominent in 1 H-NMR spectroscopic studies of brain. Neurosci Biobehav Rev. 1989;13: Urenjak J, Williams SR, Gadian DG, Noble M. Specific expression of N- acetylaspartate in neurons, oligodendrocyte-type-2 astrocyte progenitors, and immature oligodendrocytes in vitro. J Neurochem. 1992;59: Urenjak J, Williams SR, Gadian DG, Noble M. Proton nuclear magnetic resonance spectroscopy unambiguously identifies different neural cell types. J Neurosci. 1993;13: Connelly A, Jackson GD, Duncan JS, King MD, Gadian DG. Magnetic resonance spectroscopy in temporal lobe epilepsy. Neurology. 1994;44: Mendes RJ, Soares R, Simoes RF, Guimaraes ML. Reduction in temporal N- acetylaspartate and creatine (or choline) ratio in temporal lobe epilepsy: does this 1 H-magnetic resonance spectroscopy finding mean poor seizure control? J Neurol Neurosurg Psychiatry. 1998;65: Cendes F, Andermann F, Gloor P, et al. Relationship between atrophy of the amygdala and ictal fear in temporal lobe epilepsy. Brain. 1994;117: Gloor P. The Temporal Lobe and Limbic System. New York, NY: Oxford University Press;
7 28. Zentner J, Wolf HK, Helmstaedter C, et al. Clinical relevance of amygdala sclerosis in temporal lobe epilepsy. J Neurosurg. 1999;91: Cendes F, Caramanos Z, Andermann F, Dubeau F, Arnold DL. Proton magnetic resonance spectroscopic imaging and magnetic resonance imaging volumetry in the lateralization of temporal lobe epilepsy: a series of 100 patients. Ann Neurol. 1997;42: Gloor P. Contributions of electroencephalography and electrocorticography to the neurosurgical treatment of the epilepsies. Adv Neurol. 1975;8: Pauli E, Eberhardt KW, Schafer I, Tomandl B, Huk WJ, Stefan H. Chemical shift imaging spectroscopy and memory function in temporal lobe epilepsy. Epilepsia. 2000;41: Hsu YY, Chang C, Chang CN, Chu NS, Lim KE, Hsu JC. Proton MR spectroscopy in patients with complex partial seizures: single-voxel spectroscopy versus chemical-shift imaging. AJNR Am J Neuroradiol. 1999;20: Fried I, Spencer DD, Spencer SS. The anatomy of epileptic auras: focal pathology and surgical outcome. J Neurosurg. 1995;83: Sperling MR, O Connor MJ. Auras and subclinical seizures: characteristics and prognostic significance. Ann Neurol. 1990;28: Dam AM. Epilepsy and neuron loss in the hippocampus. Epilepsia. 1980;21: Babb TL, Brown WJ, Pretorius J, Davenport C, Lieb JP, Crandall PH. Temporal lobe volumetric cell densities in temporal lobe epilepsy. Epilepsia. 1984;25: Babb TL, Lieb JP, Brown WJ, Pretorius J, Crandall PH. Distribution of pyramidal cell density and hyperexcitability in the epileptic human hippocampal formation. Epilepsia. 1984;25: Cook MJ, Fish DR, Shorvon SD, Straughan K, Stevens JM. Hippocampal volumetric and morphometric studies in frontal and temporal lobe epilepsy. Brain. 1992;115: Kuks JB, Cook MJ, Fish DR, Stevens JM, Shorvon SD. Hippocampal sclerosis in epilepsy and childhood febrile seizures. Lancet. 1993;342: Van-Paesschen W, Connelly A, King MD, Jackson GD, Duncan JS. The spectrum of hippocampal sclerosis: a quantitative magnetic resonance imaging study. Ann Neurol. 1997;41: Kim JH, Tien RD, Felsberg GJ, Osumi AK, Lee N, Friedman AH. Fast spin-echo MR in hippocampal sclerosis: correlation with pathology and surgery. AJNR Am J Neuroradiol. 1995;16: Bronen RA, Fulbright RK, Kim JH, Spencer SS, Spencer DD, al-rodhan NR. Regional distribution of MR findings in hippocampal sclerosis. AJNR Am J Neuroradiol. 1995;16: Smith ML, Milner B. The role of the right hippocampus in the recall of spatial location. Neuropsychologia. 1981;19: Milner B. Psychological aspects of focal epilepsy and its neurosurgical management. Adv Neurol. 1975;8: Vermathen P, Laxer KD, Matson GB, Weiner MW. Hippocampal structures: anteroposterior N-acetylaspartate differences in patients with epilepsy and control subjects as shown with proton MR spectroscopic imaging. Radiology. 2000; 214: Miller LA, McLachlan RS, Bouwer MS, Hudson LP, Munoz DG. Amygdalar sclerosis: preoperative indicators and outcome after temporal lobectomy. J Neurol Neurosurg Psychiatry. 1994;57: Cendes F, Andermann F, Gloor P, et al. MRI volumetric measurement of amygdala and hippocampus in temporal lobe epilepsy. Neurology. 1993;43: Falconer MA, Serafetinides EA, Corsellis JAN. Etiology and pathogenesis of temporal lobe epilepsy. Arch Neurol. 1964;10: Kuzniecky R, de la Sayette V, Ethier R, et al. Magnetic resonance imaging in temporal lobe epilepsy: pathological correlations. Ann Neurol. 1987;22: Margerison JH, Corsellis JA. Epilepsy and the temporal lobes: a clinical, electroencephalographic and neuropathological study of the brain in epilepsy, with particular reference to the temporal lobes. Brain. 1966;89:
José A Mendes-Ribeiro, Raquel Soares, Fernanda Simões-Ribeiro, M Luiza Guimarães
 58 Neurophysiology Unit J A Mendes-Ribeiro M L Guimarães Department of Neurology and Neurosurgery, Hospital S João, Porto, Portugal F Simões-Ribeiro Magnetic Resonance Unit, IPO, Porto, Portugal R Soares
58 Neurophysiology Unit J A Mendes-Ribeiro M L Guimarães Department of Neurology and Neurosurgery, Hospital S João, Porto, Portugal F Simões-Ribeiro Magnetic Resonance Unit, IPO, Porto, Portugal R Soares
Fig. 1. Localized single voxel proton MR spectroscopy was performed along the long axis of right hippocampus after extension of patient s head to
 125 A B C Fig. 1. Localized single voxel proton MR spectroscopy was performed along the long axis of right hippocampus after extension of patient s head to obtain entire dimension of the hippocampal body.
125 A B C Fig. 1. Localized single voxel proton MR spectroscopy was performed along the long axis of right hippocampus after extension of patient s head to obtain entire dimension of the hippocampal body.
Usefulness of Single Voxel Proton MR Spectroscopy in the Evaluation of Hippocampal Sclerosis
 Usefulness of Single Voxel Proton MR Spectroscopy in the Evaluation of Hippocampal Sclerosis 1, 2, 3 Kee-Hyun Chang, MD Hong Dae Kim, MD 1 Sun-Won Park, MD 1 In Chan Song, PhD 2 In Kyu Yu, MD 1 1, 2, 3
Usefulness of Single Voxel Proton MR Spectroscopy in the Evaluation of Hippocampal Sclerosis 1, 2, 3 Kee-Hyun Chang, MD Hong Dae Kim, MD 1 Sun-Won Park, MD 1 In Chan Song, PhD 2 In Kyu Yu, MD 1 1, 2, 3
Value of Single-Voxel Proton MR Spectroscopy in Temporal Lobe Epilepsy
 Value of Single-Voxel Proton MR Spectroscopy in Temporal Lobe Epilepsy Eric Achten, Paul Boon, Tom Van De Kerckhove, Jacques Caemaert, Jacques De Reuck, and Marc Kunnen PURPOSE: To study the value of different
Value of Single-Voxel Proton MR Spectroscopy in Temporal Lobe Epilepsy Eric Achten, Paul Boon, Tom Van De Kerckhove, Jacques Caemaert, Jacques De Reuck, and Marc Kunnen PURPOSE: To study the value of different
The Requirement for Ictal EEG Recordings Prior to Temporal Lobe Epilepsy Surgery
 Page 1 of 7 Archives of Neurology Issue: Volume 58(4), April 2001, pp 678-680 Copyright: Copyright 2001 by the American Medical Association. All Rights Reserved. Applicable FARS/DFARS Restrictions Apply
Page 1 of 7 Archives of Neurology Issue: Volume 58(4), April 2001, pp 678-680 Copyright: Copyright 2001 by the American Medical Association. All Rights Reserved. Applicable FARS/DFARS Restrictions Apply
Theroleofclinicalin vivo 1H-MR spectroscopy in the evaluation of epilepsies
 Spectroscopy 16 (2002) 297 306 297 IOS Press Theroleofclinicalin vivo 1H-MR spectroscopy in the evaluation of epilepsies T. Hammen a,,h.stefan a and B. Tomandl b a Clinics of Neurology Center Epilepsy,
Spectroscopy 16 (2002) 297 306 297 IOS Press Theroleofclinicalin vivo 1H-MR spectroscopy in the evaluation of epilepsies T. Hammen a,,h.stefan a and B. Tomandl b a Clinics of Neurology Center Epilepsy,
Morphometric MRI Analysis of the Parahippocampal Region in Temporal Lobe Epilepsy
 Morphometric MRI Analysis of the Parahippocampal Region in Temporal Lobe Epilepsy NEDA BERNASCONI, a ANDREA BERNASCONI, ZOGRAFOS CARAMANOS, FREDERICK ANDERMANN, FRANÇOIS DUBEAU, AND DOUGLAS L. ARNOLD Department
Morphometric MRI Analysis of the Parahippocampal Region in Temporal Lobe Epilepsy NEDA BERNASCONI, a ANDREA BERNASCONI, ZOGRAFOS CARAMANOS, FREDERICK ANDERMANN, FRANÇOIS DUBEAU, AND DOUGLAS L. ARNOLD Department
Lateralizing Ability of Single-voxel Proton MR Spectroscopy in Hippocampal Sclerosis: Comparison with MR Imaging and Positron Emission Tomography
 AJNR Am J Neuroradiol 22:625 631, April 2001 Lateralizing Ability of Single-voxel Proton MR Spectroscopy in Hippocampal Sclerosis: Comparison with MR Imaging and Positron Emission Tomography Sun-Won Park,
AJNR Am J Neuroradiol 22:625 631, April 2001 Lateralizing Ability of Single-voxel Proton MR Spectroscopy in Hippocampal Sclerosis: Comparison with MR Imaging and Positron Emission Tomography Sun-Won Park,
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,100 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,100 116,000 120M Open access books available International authors and editors Downloads Our
The Asymmetric Mamillary Body: Association with Medial Temporal Lobe Disease Demonstrated with MR
 The Mamillary Body: Association with Medial Temporal Lobe Disease Demonstrated with MR Alexander C. Mamourian, Lawrence Rodichok, and Javad Towfighi PURPOSE: To determine whether mamillary body atrophy
The Mamillary Body: Association with Medial Temporal Lobe Disease Demonstrated with MR Alexander C. Mamourian, Lawrence Rodichok, and Javad Towfighi PURPOSE: To determine whether mamillary body atrophy
Surgical outcome in patients with epilepsy and dual pathology
 Brain (1999), 122, 799 805 Surgical outcome in patients with epilepsy and dual pathology L. M. Li, 1 F. Cendes, 1 F. Andermann, 1 C. Watson, 2 D. R. Fish, 3 M. J. Cook, 4 F. Dubeau, 1 J. S. Duncan, 3 S.
Brain (1999), 122, 799 805 Surgical outcome in patients with epilepsy and dual pathology L. M. Li, 1 F. Cendes, 1 F. Andermann, 1 C. Watson, 2 D. R. Fish, 3 M. J. Cook, 4 F. Dubeau, 1 J. S. Duncan, 3 S.
Fernando Cendes, MD, PhD,* Zografos Caramanos, MA, Frederick Andermann, MD, FranGois Dubeau, MD, and Douglas L. Arnold, MD
 P Proton Magnetic Resonance Spectroscopic Imaging and Magnetic Resonance Imaging Volumetry in the Lateralization of Temporal Lobe Epilepsy: A Series of 100 Patiekts Fernando Cendes, MD, PhD,* Zografos
P Proton Magnetic Resonance Spectroscopic Imaging and Magnetic Resonance Imaging Volumetry in the Lateralization of Temporal Lobe Epilepsy: A Series of 100 Patiekts Fernando Cendes, MD, PhD,* Zografos
A study of the relationship between metabolism using 1 H-MRS and function using several neuropsychological
 Seizure 2001; 10: 188 193 doi:10.1053/seiz.2000.0498, available online at http://www.idealibrary.com on A study of the relationship between metabolism using 1 H-MRS and function using several neuropsychological
Seizure 2001; 10: 188 193 doi:10.1053/seiz.2000.0498, available online at http://www.idealibrary.com on A study of the relationship between metabolism using 1 H-MRS and function using several neuropsychological
Successful Treatment of Mesial Temporal Lobe Epilepsy with Bilateral Hippocampal Atrophy and False Temporal Scalp Ictal Onset: A case report
 Hiroshima J. Med. Sci. Vol. 61, No. 2, 37~41, June, 2012 HIJM 61 7 37 Successful Treatment of Mesial Temporal Lobe Epilepsy with Bilateral Hippocampal Atrophy and False Temporal Scalp Ictal Onset: A case
Hiroshima J. Med. Sci. Vol. 61, No. 2, 37~41, June, 2012 HIJM 61 7 37 Successful Treatment of Mesial Temporal Lobe Epilepsy with Bilateral Hippocampal Atrophy and False Temporal Scalp Ictal Onset: A case
Multisection Proton MR Spectroscopy for Mesial Temporal Lobe Epilepsy
 AJNR Am J Neuroradiol 23:1359 1368, September 2002 Multisection Proton MR Spectroscopy for Mesial Temporal Lobe Epilepsy Arístides A. Capizzano, Peter Vermathen, Kenneth D. Laxer, Gerald B. Matson, Andrew
AJNR Am J Neuroradiol 23:1359 1368, September 2002 Multisection Proton MR Spectroscopy for Mesial Temporal Lobe Epilepsy Arístides A. Capizzano, Peter Vermathen, Kenneth D. Laxer, Gerald B. Matson, Andrew
Cerebral MRI as an important diagnostic tool in temporal lobe epilepsy
 Cerebral MRI as an important diagnostic tool in temporal lobe epilepsy Poster No.: C-2190 Congress: ECR 2014 Type: Educational Exhibit Authors: A. Puiu, D. Negru; Iasi/RO Keywords: Neuroradiology brain,
Cerebral MRI as an important diagnostic tool in temporal lobe epilepsy Poster No.: C-2190 Congress: ECR 2014 Type: Educational Exhibit Authors: A. Puiu, D. Negru; Iasi/RO Keywords: Neuroradiology brain,
RADIOLOGY NEURORADIOLOGY. Iranian Journal of
 NEURORADIOLOGY Iranian Journal of RADIOLOGY RADIOLOGYwww.iranjradiol.com Value of Proton-MR-Spectroscopy in the Diagnosis of Temporal Lobe Epilepsy; Correlation of Metabolite Alterations With Electroencephalography
NEURORADIOLOGY Iranian Journal of RADIOLOGY RADIOLOGYwww.iranjradiol.com Value of Proton-MR-Spectroscopy in the Diagnosis of Temporal Lobe Epilepsy; Correlation of Metabolite Alterations With Electroencephalography
Cerebral structural lesions are found in approximately. Surgery of Intractable Temporal Lobe Epilepsy Presented with Structural Lesions
 Original Article J Chin Med Assoc 2003;66:565-571 Surgery of Intractable Temporal Lobe Epilepsy Presented with Structural Lesions Yang-Hsin Shih 1 Jiang-Fong Lirng 2 Der-Jen Yen 3 Donald M. Ho 4 Chun-Hing
Original Article J Chin Med Assoc 2003;66:565-571 Surgery of Intractable Temporal Lobe Epilepsy Presented with Structural Lesions Yang-Hsin Shih 1 Jiang-Fong Lirng 2 Der-Jen Yen 3 Donald M. Ho 4 Chun-Hing
MR Detection of Hippocampal Disease in Epilepsy: Factors Influencing T2 Relaxation Time
 MR Detection of Hippocampal Disease in Epilepsy: Factors Influencing T2 Relaxation Time R. A. Grunewald, G. D. Jackson, A. Connelly, and J. S. Duncan PURPOSE: To assess the reproducibility and stability
MR Detection of Hippocampal Disease in Epilepsy: Factors Influencing T2 Relaxation Time R. A. Grunewald, G. D. Jackson, A. Connelly, and J. S. Duncan PURPOSE: To assess the reproducibility and stability
An MR Protocol for Presurgical Evaluation of Patients with Complex Partial Seizures of Temporal Lobe Origin
 An MR Protocol for Presurgical Evaluation of Patients with Complex Partial Seizures of Temporal Lobe Origin Eric Achten, Paul Boon, John De Poorter, Luc Calliauw, Tom Van De Kerckhove, Jacques De Reuck,
An MR Protocol for Presurgical Evaluation of Patients with Complex Partial Seizures of Temporal Lobe Origin Eric Achten, Paul Boon, John De Poorter, Luc Calliauw, Tom Van De Kerckhove, Jacques De Reuck,
Intracranial Studies Of Human Epilepsy In A Surgical Setting
 Intracranial Studies Of Human Epilepsy In A Surgical Setting Department of Neurology David Geffen School of Medicine at UCLA Presentation Goals Epilepsy and seizures Basics of the electroencephalogram
Intracranial Studies Of Human Epilepsy In A Surgical Setting Department of Neurology David Geffen School of Medicine at UCLA Presentation Goals Epilepsy and seizures Basics of the electroencephalogram
CHAPTER 5. The intracarotid amobarbital or Wada test: unilateral or bilateral?
 CHAPTER 5 Chapter 5 CHAPTER 5 The intracarotid amobarbital or Wada test: unilateral or bilateral? SG Uijl FSS Leijten JBAM Arends J Parra AC van Huffelen PC van Rijen KGM Moons Submitted 2007. 74 Abstract
CHAPTER 5 Chapter 5 CHAPTER 5 The intracarotid amobarbital or Wada test: unilateral or bilateral? SG Uijl FSS Leijten JBAM Arends J Parra AC van Huffelen PC van Rijen KGM Moons Submitted 2007. 74 Abstract
Proton MR Spectroscopy in Patients with Acute Temporal Lobe Seizures
 AJN Am J Neuroradiol 22:152 157, January 2001 Proton M Spectroscopy in Patients with Acute Temporal obe Seizures Mauricio Castillo, J. Keith Smith, and ester Kwock BACKGOUND AND PUPOSE: Decreases in N-acetyl
AJN Am J Neuroradiol 22:152 157, January 2001 Proton M Spectroscopy in Patients with Acute Temporal obe Seizures Mauricio Castillo, J. Keith Smith, and ester Kwock BACKGOUND AND PUPOSE: Decreases in N-acetyl
Diagnosing Complicated Epilepsy: Mapping of the Epileptic Circuitry. Michael R. Sperling, M.D. Thomas Jefferson University Philadelphia, PA
 Diagnosing Complicated Epilepsy: Mapping of the Epileptic Circuitry Michael R. Sperling, M.D. Thomas Jefferson University Philadelphia, PA Overview Definition of epileptic circuitry Methods of mapping
Diagnosing Complicated Epilepsy: Mapping of the Epileptic Circuitry Michael R. Sperling, M.D. Thomas Jefferson University Philadelphia, PA Overview Definition of epileptic circuitry Methods of mapping
PRESURGICAL EVALUATION. ISLAND OF COS Hippocrates: On the Sacred Disease. Disclosure Research-Educational Grants. Patients with seizure disorders
 PRESURGICAL EVALUATION Patients with seizure disorders Gregory D. Cascino, MD Mayo Clinic Disclosure Research-Educational Grants Mayo Foundation Neuro Pace, Inc. American Epilepsy Society American Academy
PRESURGICAL EVALUATION Patients with seizure disorders Gregory D. Cascino, MD Mayo Clinic Disclosure Research-Educational Grants Mayo Foundation Neuro Pace, Inc. American Epilepsy Society American Academy
Early seizure propagation from the occipital lobe to medial temporal structures and its surgical implication
 Original article Epileptic Disord 2008; 10 (4): 260-5 Early seizure propagation from the occipital lobe to medial temporal structures and its surgical implication Naotaka Usui, Tadahiro Mihara, Koichi
Original article Epileptic Disord 2008; 10 (4): 260-5 Early seizure propagation from the occipital lobe to medial temporal structures and its surgical implication Naotaka Usui, Tadahiro Mihara, Koichi
Case reports functional imaging in epilepsy
 Seizure 2001; 10: 157 161 doi:10.1053/seiz.2001.0552, available online at http://www.idealibrary.com on Case reports functional imaging in epilepsy MARK P. RICHARDSON Medical Research Council Fellow, Institute
Seizure 2001; 10: 157 161 doi:10.1053/seiz.2001.0552, available online at http://www.idealibrary.com on Case reports functional imaging in epilepsy MARK P. RICHARDSON Medical Research Council Fellow, Institute
High Resolution Ictal SPECT: Enhanced Epileptic Source Targeting?
 High Resolution Ictal SPECT: Enhanced Epileptic Source Targeting? Marvin A Rossi MD, PhD RUSH Epilepsy Center Research Lab http://www.synapticom.net Chicago, IL USA Medically-Refractory Epilepsy 500,000-800,000
High Resolution Ictal SPECT: Enhanced Epileptic Source Targeting? Marvin A Rossi MD, PhD RUSH Epilepsy Center Research Lab http://www.synapticom.net Chicago, IL USA Medically-Refractory Epilepsy 500,000-800,000
The running down phenomenon in temporal lobe epilepsy
 Brain (1996), 119, 989-996 The running down phenomenon in temporal lobe epilepsy Vicenta Salanova,* Frederick Andermann, Theodore Rasmussen, Andre Olivier and Luis Quesney Department of Neurology and Neurosurgery,
Brain (1996), 119, 989-996 The running down phenomenon in temporal lobe epilepsy Vicenta Salanova,* Frederick Andermann, Theodore Rasmussen, Andre Olivier and Luis Quesney Department of Neurology and Neurosurgery,
Multimodal Imaging in Extratemporal Epilepsy Surgery
 Open Access Case Report DOI: 10.7759/cureus.2338 Multimodal Imaging in Extratemporal Epilepsy Surgery Christian Vollmar 1, Aurelia Peraud 2, Soheyl Noachtar 1 1. Epilepsy Center, Dept. of Neurology, University
Open Access Case Report DOI: 10.7759/cureus.2338 Multimodal Imaging in Extratemporal Epilepsy Surgery Christian Vollmar 1, Aurelia Peraud 2, Soheyl Noachtar 1 1. Epilepsy Center, Dept. of Neurology, University
Correlation between 1 H MRS and Memory before and after Surgery in Mesial Temporal Lobe Epilepsy with Hippocampal Sclerosis
 Epilepsia, 45(6):632 640, 2004 Blackwell Publishing, Inc. C 2004 International League Against Epilepsy Correlation between 1 H MRS and Memory before and after Surgery in Mesial Temporal Lobe Epilepsy with
Epilepsia, 45(6):632 640, 2004 Blackwell Publishing, Inc. C 2004 International League Against Epilepsy Correlation between 1 H MRS and Memory before and after Surgery in Mesial Temporal Lobe Epilepsy with
Spike frequency is dependent on epilepsy duration and seizure frequency in temporal lobe epilepsy
 Original article Epileptic Disord 2005; 7 (4): 355-9 Spike frequency is dependent on epilepsy duration and seizure frequency in temporal lobe epilepsy Jozsef Janszky 1,2,3, M. Hoppe 1, Z. Clemens 3, I.
Original article Epileptic Disord 2005; 7 (4): 355-9 Spike frequency is dependent on epilepsy duration and seizure frequency in temporal lobe epilepsy Jozsef Janszky 1,2,3, M. Hoppe 1, Z. Clemens 3, I.
DIAGNOSTIC NEURORADIOLOGY. sequences
 Neuroradiology (1999) 41: 471±479 Ó Springer-Verlag 1999 DIAGNOSTIC NEURORADIOLOGY C. Oppenheim D. Dormont D. Hasboun B. Bazin S. Samson S. LehØricy M. Baulac C. Marsault Bilateral mesial temporal sclerosis:
Neuroradiology (1999) 41: 471±479 Ó Springer-Verlag 1999 DIAGNOSTIC NEURORADIOLOGY C. Oppenheim D. Dormont D. Hasboun B. Bazin S. Samson S. LehØricy M. Baulac C. Marsault Bilateral mesial temporal sclerosis:
Advanced MR Imaging of Cortical Dysplasia with or without Neoplasm: A Report of Two Cases
 AJNR Am J Neuroradiol 23:1686 1691, November/December 2002 Case Report Advanced MR Imaging of Cortical Dysplasia with or without Neoplasm: A Report of Two Cases Jay J. Pillai, Richard B. Hessler, Jerry
AJNR Am J Neuroradiol 23:1686 1691, November/December 2002 Case Report Advanced MR Imaging of Cortical Dysplasia with or without Neoplasm: A Report of Two Cases Jay J. Pillai, Richard B. Hessler, Jerry
controls. <Conclusions> These data support the hypothesis that JME and FLE involve neuronal dysfunction within the temporal lobe as well as the
 A single-voxel spectroscopy study of hippocampal metabolic dysfunction in patients with juvenile myoclonic epilepsy, frontal lobe epilepsy, and psychogenic nonepileptic seizures Epilepsy Center, National
A single-voxel spectroscopy study of hippocampal metabolic dysfunction in patients with juvenile myoclonic epilepsy, frontal lobe epilepsy, and psychogenic nonepileptic seizures Epilepsy Center, National
Hamartomas and epilepsy: clinical and imaging characteristics
 Seizure 2003; 12: 307 311 doi:10.1016/s1059 1311(02)00272-8 Hamartomas and epilepsy: clinical and imaging characteristics B. DIEHL, R. PRAYSON, I. NAJM & P. RUGGIERI Departments of Neurology, Pathology
Seizure 2003; 12: 307 311 doi:10.1016/s1059 1311(02)00272-8 Hamartomas and epilepsy: clinical and imaging characteristics B. DIEHL, R. PRAYSON, I. NAJM & P. RUGGIERI Departments of Neurology, Pathology
Single-Voxel Proton MR Spectroscopy and Positron Emission Tomography for Lateralization of Refractory Temporal Lobe Epilepsy
 AJNR Am J Neuroradiol 19:1 8, January 1998 Single-Voxel Proton MR Spectroscopy and Positron Emission Tomography for Lateralization of Refractory Temporal Lobe Epilepsy Eric Achten, Patrick Santens, Paul
AJNR Am J Neuroradiol 19:1 8, January 1998 Single-Voxel Proton MR Spectroscopy and Positron Emission Tomography for Lateralization of Refractory Temporal Lobe Epilepsy Eric Achten, Patrick Santens, Paul
Early detection of abnormalities in partial epilepsy
 104 Institute of Child Health and Hospital for Sick Children, London, Neurosciences Unit J H Cross G D Jackson B G R Neville F J Kirkham Radiology and Physics Unit A Connelly D G Gadian Department of Clinical
104 Institute of Child Health and Hospital for Sick Children, London, Neurosciences Unit J H Cross G D Jackson B G R Neville F J Kirkham Radiology and Physics Unit A Connelly D G Gadian Department of Clinical
Est-ce que l'eeg a toujours sa place en 2019?
 Est-ce que l'eeg a toujours sa place en 2019? Thomas Bast Epilepsy Center Kork, Germany Does EEG still play a role in 2019? What a question 7T-MRI, fmri, DTI, MEG, SISCOM, Of ieeg course! /HFO, Genetics
Est-ce que l'eeg a toujours sa place en 2019? Thomas Bast Epilepsy Center Kork, Germany Does EEG still play a role in 2019? What a question 7T-MRI, fmri, DTI, MEG, SISCOM, Of ieeg course! /HFO, Genetics
Methods for Normalization of Hippocampal Volumes Measured with MR
 Methods for Normalization of Hippocampal Volumes Measured with MR S. L. Free, P. S. Bergin, D. R. Fish, M. J. Cook, S. D. Shorvon, and J. M. Stevens PURPOSE: To investigate the use of six cerebral measures
Methods for Normalization of Hippocampal Volumes Measured with MR S. L. Free, P. S. Bergin, D. R. Fish, M. J. Cook, S. D. Shorvon, and J. M. Stevens PURPOSE: To investigate the use of six cerebral measures
The relevance of somatosensory auras in refractory temporal lobe epilepsies
 BRIEF COMMUNICATION The relevance of somatosensory auras in refractory temporal lobe epilepsies Ghazala Perven, Ruta Yardi, Juan Bulacio, Imad Najm, William Bingaman, Jorge Gonzalez-Martinez, and Lara
BRIEF COMMUNICATION The relevance of somatosensory auras in refractory temporal lobe epilepsies Ghazala Perven, Ruta Yardi, Juan Bulacio, Imad Najm, William Bingaman, Jorge Gonzalez-Martinez, and Lara
Deakin Research Online
 Deakin Research Online This is the published version: Silberstein, Morry, Lane, Dianne, Dodd, Seetal and Opeskin, Kenneth 2002, Identification of a by-product of nitric oxide synthase activity in human
Deakin Research Online This is the published version: Silberstein, Morry, Lane, Dianne, Dodd, Seetal and Opeskin, Kenneth 2002, Identification of a by-product of nitric oxide synthase activity in human
Increased Anterior Temporal Lobe T2 Times in Cases of Hippocampal Sclerosis: A Multi-Echo T2 Relaxometry Study At 3 T
 AJNR Am J Neuroradiol 25:389 394, March 2004 Increased Anterior Temporal Lobe T2 Times in Cases of Hippocampal Sclerosis: A Multi-Echo T2 Relaxometry Study At 3 T Regula S. Briellmann, Ari Syngeniotis,
AJNR Am J Neuroradiol 25:389 394, March 2004 Increased Anterior Temporal Lobe T2 Times in Cases of Hippocampal Sclerosis: A Multi-Echo T2 Relaxometry Study At 3 T Regula S. Briellmann, Ari Syngeniotis,
9/30/2016. Advances in Epilepsy Surgery. Epidemiology. Epidemiology
 Advances in Epilepsy Surgery George Jallo, M.D. Director, Institute for Brain Protection Sciences Johns Hopkins All Children s Hospital St Petersburg, Florida Epidemiology WHO lists it as the second most
Advances in Epilepsy Surgery George Jallo, M.D. Director, Institute for Brain Protection Sciences Johns Hopkins All Children s Hospital St Petersburg, Florida Epidemiology WHO lists it as the second most
Electrocorticographic Monitoring as An Alternative Tool for the Pre-surgical Evaluation of Patients with Bi-temporal Epilepsy
 Original Article 194 Electrocorticographic Monitoring as An Alternative Tool for the Pre-surgical Evaluation of Patients with Bi-temporal Epilepsy Peng-Wei Hsu 3, MD; Hsien-Chih Chen, MD; Yin-Cheng Huang
Original Article 194 Electrocorticographic Monitoring as An Alternative Tool for the Pre-surgical Evaluation of Patients with Bi-temporal Epilepsy Peng-Wei Hsu 3, MD; Hsien-Chih Chen, MD; Yin-Cheng Huang
Approximately 70% of childhood SURGICAL TREATMENTS FOR PEDIATRIC EPILEPSY PROCEEDINGS. Ronald P. Lesser, MD KEY POINTS
 ASIM May p153-158 5/14/01 9:19 AM Page 153 SURGICAL TREATMENTS FOR PEDIATRIC EPILEPSY Ronald P. Lesser, MD KEY POINTS Most children with epilepsy refractory to drugs can improve with surgery Temporal lobe
ASIM May p153-158 5/14/01 9:19 AM Page 153 SURGICAL TREATMENTS FOR PEDIATRIC EPILEPSY Ronald P. Lesser, MD KEY POINTS Most children with epilepsy refractory to drugs can improve with surgery Temporal lobe
Association between Size of the Lateral Ventricle and Asymmetry of the Fornix in Patients with Temporal Lobe Epilepsy
 AJNR Am J Neuroradiol 19:9 13, January 1998 Association between Size of the Lateral Ventricle and Asymmetry of the Fornix in Patients with Temporal Lobe Epilepsy Alexander C. Mamourian, Charles H. Cho,
AJNR Am J Neuroradiol 19:9 13, January 1998 Association between Size of the Lateral Ventricle and Asymmetry of the Fornix in Patients with Temporal Lobe Epilepsy Alexander C. Mamourian, Charles H. Cho,
The Clinical and Electrophysiological Characteristics of Temporal Lobe Epilepsy with Normal MRI
 Journal of Clinical Neurology / Volume 2 / March, 2006 Original Articles The Clinical and Electrophysiological Characteristics of Temporal Lobe Epilepsy with Normal MRI S.E. Kim, M.D., Ph.D., F. Andermann,
Journal of Clinical Neurology / Volume 2 / March, 2006 Original Articles The Clinical and Electrophysiological Characteristics of Temporal Lobe Epilepsy with Normal MRI S.E. Kim, M.D., Ph.D., F. Andermann,
BOLD Based MRI Functional Connectivity December 2, 2011
 BOLD Based MRI Functional Connectivity December 2, 2011 Luigi Maccotta, MD, PhD Adult Epilepsy Center Washington University School of Medicine American Epilepsy Society Annual Meeting Support Disclosure
BOLD Based MRI Functional Connectivity December 2, 2011 Luigi Maccotta, MD, PhD Adult Epilepsy Center Washington University School of Medicine American Epilepsy Society Annual Meeting Support Disclosure
EPILEPSY SURGERY EVALUATION IN ADULTS WITH SCALP VIDEO-EEG MONITORING. Meriem Bensalem-Owen, MD University of Kentucky
 EPILEPSY SURGERY EVALUATION IN ADULTS WITH SCALP VIDEO-EEG MONITORING Meriem Bensalem-Owen, MD University of Kentucky DISCLOSURES Received grants for sponsored research as investigator from: UCB Eisai
EPILEPSY SURGERY EVALUATION IN ADULTS WITH SCALP VIDEO-EEG MONITORING Meriem Bensalem-Owen, MD University of Kentucky DISCLOSURES Received grants for sponsored research as investigator from: UCB Eisai
Age at onset in patients with medically refractory. temporal lobe epilepsy and mesial temporal sclerosis: impact on clinical manifestations and
 Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology 8-1-2015 Age at onset in patients with medically refractory temporal lobe epilepsy and
Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology 8-1-2015 Age at onset in patients with medically refractory temporal lobe epilepsy and
Pharmacoresistant temporal lobe epilepsy - a diagnostic performance of standardized MRI protocol in detection of epileptogenic lesion
 Pharmacoresistant temporal lobe epilepsy - a diagnostic performance of standardized MRI protocol in detection of epileptogenic lesion Poster No.: C-2226 Congress: ECR 2013 Type: Scientific Exhibit Authors:
Pharmacoresistant temporal lobe epilepsy - a diagnostic performance of standardized MRI protocol in detection of epileptogenic lesion Poster No.: C-2226 Congress: ECR 2013 Type: Scientific Exhibit Authors:
Surgery in temporal lobe epilepsy patients without cranial MRI lateralization
 Acta neurol. belg., 2006, 106, 9-14 Surgery in temporal lobe epilepsy patients without cranial MRI lateralization Y. B. GOMCELI 1, A. ERDEM 2, E. BILIR 3, G. KUTLU 1, S. KURT 4, E. ERDEN 5,A. KARATAS 2,
Acta neurol. belg., 2006, 106, 9-14 Surgery in temporal lobe epilepsy patients without cranial MRI lateralization Y. B. GOMCELI 1, A. ERDEM 2, E. BILIR 3, G. KUTLU 1, S. KURT 4, E. ERDEN 5,A. KARATAS 2,
Accepted Manuscript. Editorial. Responsive neurostimulation for epilepsy: more than stimulation. Jayant N. Acharya
 Accepted Manuscript Editorial Responsive neurostimulation for epilepsy: more than stimulation Jayant N. Acharya PII: S2467-981X(18)30022-2 DOI: https://doi.org/10.1016/j.cnp.2018.06.002 Reference: CNP
Accepted Manuscript Editorial Responsive neurostimulation for epilepsy: more than stimulation Jayant N. Acharya PII: S2467-981X(18)30022-2 DOI: https://doi.org/10.1016/j.cnp.2018.06.002 Reference: CNP
Brain Structure and Epilepsy: The Impact of Modern Imaging
 Commentary Brain Structure and Epilepsy: The Impact of Modern Imaging Frederick Andermann, Professor of Neurology and Paediatrics, Department of Neurology and Neurosurgery, McGill University, Montreal,
Commentary Brain Structure and Epilepsy: The Impact of Modern Imaging Frederick Andermann, Professor of Neurology and Paediatrics, Department of Neurology and Neurosurgery, McGill University, Montreal,
EPILEPSY. New Ideas about an Old Disease. Gregory D. Cascino, MD
 EPILEPSY New Ideas about an Old Disease Gregory D. Cascino, MD Disclosure Research-Educational Grants Neuro Pace, Inc. American Epilepsy Society American Academy of Neurology Neurology (Associate Editor)
EPILEPSY New Ideas about an Old Disease Gregory D. Cascino, MD Disclosure Research-Educational Grants Neuro Pace, Inc. American Epilepsy Society American Academy of Neurology Neurology (Associate Editor)
Blurring the Lines Between Lesional and Nonlesional MRI
 Current Literature In Clinical Science Blurring the Lines Between Lesional and Nonlesional MRI Blurring in Patients With Temporal Lobe Epilepsy: Clinical, High-field Imaging and Ultrastructural Study.
Current Literature In Clinical Science Blurring the Lines Between Lesional and Nonlesional MRI Blurring in Patients With Temporal Lobe Epilepsy: Clinical, High-field Imaging and Ultrastructural Study.
Is DTI Increasing the Connectivity Between the Magnet Suite and the Clinic?
 Current Literature In Clinical Science Is DTI Increasing the Connectivity Between the Magnet Suite and the Clinic? Spatial Patterns of Water Diffusion Along White Matter Tracts in Temporal Lobe Epilepsy.
Current Literature In Clinical Science Is DTI Increasing the Connectivity Between the Magnet Suite and the Clinic? Spatial Patterns of Water Diffusion Along White Matter Tracts in Temporal Lobe Epilepsy.
Subject: Magnetoencephalography/Magnetic Source Imaging
 01-95805-16 Original Effective Date: 09/01/01 Reviewed: 07/26/18 Revised: 08/15/18 Subject: Magnetoencephalography/Magnetic Source Imaging THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
01-95805-16 Original Effective Date: 09/01/01 Reviewed: 07/26/18 Revised: 08/15/18 Subject: Magnetoencephalography/Magnetic Source Imaging THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
Epilepsy surgery in the elderly
 Clinical commentary Epileptic Disord 2009; 11 (1): 1-6 Epilepsy surgery in the elderly An unusual case of a 75-year-old man with recurrent status epilepticus Jose F. Tellez-Zenteno 1, Venkatraman Sadanand
Clinical commentary Epileptic Disord 2009; 11 (1): 1-6 Epilepsy surgery in the elderly An unusual case of a 75-year-old man with recurrent status epilepticus Jose F. Tellez-Zenteno 1, Venkatraman Sadanand
An analysis of MRI findings in patients referred with fits
 An analysis of MRI findings in patients referred with fits Pallewatte AS 1, Alahakoon S 1, Senanayake G 1, Bulathsinghela BC 1 1 National Hospital of Sri Lanka, Colombo, Sri Lanka Abstract Introduction:
An analysis of MRI findings in patients referred with fits Pallewatte AS 1, Alahakoon S 1, Senanayake G 1, Bulathsinghela BC 1 1 National Hospital of Sri Lanka, Colombo, Sri Lanka Abstract Introduction:
Diffusion-Weighted and Conventional MR Imaging Findings of Neuroaxonal Dystrophy
 AJNR Am J Neuroradiol 25:1269 1273, August 2004 Diffusion-Weighted and Conventional MR Imaging Findings of Neuroaxonal Dystrophy R. Nuri Sener BACKGROUND AND PURPOSE: Neuroaxonal dystrophy is a rare progressive
AJNR Am J Neuroradiol 25:1269 1273, August 2004 Diffusion-Weighted and Conventional MR Imaging Findings of Neuroaxonal Dystrophy R. Nuri Sener BACKGROUND AND PURPOSE: Neuroaxonal dystrophy is a rare progressive
Medicine Review Course Approach to Transient Amnesia
 Medicine Review Course Approach to Transient Amnesia 9 Sept 2012 Dr See Siew Ju Neurology NNI@SGH A Few Questions.. What did you have for breakfast on Friday? How many red lights did you stop at on your
Medicine Review Course Approach to Transient Amnesia 9 Sept 2012 Dr See Siew Ju Neurology NNI@SGH A Few Questions.. What did you have for breakfast on Friday? How many red lights did you stop at on your
Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use
 International Congress Series 1281 (2005) 793 797 www.ics-elsevier.com Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use Ch. Nimsky a,b,
International Congress Series 1281 (2005) 793 797 www.ics-elsevier.com Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use Ch. Nimsky a,b,
Localization of Temporal Lobe Foci by Ictal EEG Patterns
 Epilepsia, 37(4):386-399, 1996 Lippincott-Raven Publishers, Philadelphia 0 International League Against Epilepsy Localization of Temporal Lobe Foci by Ictal EEG Patterns John S. Ebersole and *Steven V.
Epilepsia, 37(4):386-399, 1996 Lippincott-Raven Publishers, Philadelphia 0 International League Against Epilepsy Localization of Temporal Lobe Foci by Ictal EEG Patterns John S. Ebersole and *Steven V.
Mesial temporal lobe epilepsy with childhood febrile seizure.
 Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology 2-9-2016 Mesial temporal lobe epilepsy with childhood febrile seizure. Ali Akbar Asadi-Pooya
Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology 2-9-2016 Mesial temporal lobe epilepsy with childhood febrile seizure. Ali Akbar Asadi-Pooya
Author's response to reviews
 Author's response to reviews Title: MRI-negative PET-positive Temporal Lobe Epilepsy (TLE) and Mesial TLE differ with Quantitative MRI and PET: a case control study Authors: Ross P Carne (carnero@svhm.org.au)
Author's response to reviews Title: MRI-negative PET-positive Temporal Lobe Epilepsy (TLE) and Mesial TLE differ with Quantitative MRI and PET: a case control study Authors: Ross P Carne (carnero@svhm.org.au)
Spike voltage topography in temporal lobe epilepsy
 Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology 5-17-2016 Spike voltage topography in temporal lobe epilepsy Ali Akbar Asadi-Pooya Thomas
Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology 5-17-2016 Spike voltage topography in temporal lobe epilepsy Ali Akbar Asadi-Pooya Thomas
EEG Background Delta Activity in Temporal Lobe Epilepsy: Correlation with Volumetric and Spectroscopic Imaging
 Epilepsia, 40(11):158&1586, 1999 Lippincott Williams & Wilkins, Inc., Philadelphia 0 International League Against Epilepsy Clinical Research EEG Background Delta Activity in Temporal Lobe Epilepsy: Correlation
Epilepsia, 40(11):158&1586, 1999 Lippincott Williams & Wilkins, Inc., Philadelphia 0 International League Against Epilepsy Clinical Research EEG Background Delta Activity in Temporal Lobe Epilepsy: Correlation
Surgery for Medically Refractory Focal Epilepsy
 Surgery for Medically Refractory Focal Epilepsy Seth F Oliveria, MD PhD The Oregon Clinic Neurosurgery Director of Functional Neurosurgery: Providence Brain and Spine Institute Portland, OR Providence
Surgery for Medically Refractory Focal Epilepsy Seth F Oliveria, MD PhD The Oregon Clinic Neurosurgery Director of Functional Neurosurgery: Providence Brain and Spine Institute Portland, OR Providence
Standard magnetic resonance imaging is inadequate for patients with refractory focal epilepsy
 PAPER Standard magnetic resonance imaging is inadequate for patients with refractory focal epilepsy J von Oertzen, H Urbach, S Jungbluth, M Kurthen, M Reuber, G Fernández, C E Elger... See Editorial Commentary
PAPER Standard magnetic resonance imaging is inadequate for patients with refractory focal epilepsy J von Oertzen, H Urbach, S Jungbluth, M Kurthen, M Reuber, G Fernández, C E Elger... See Editorial Commentary
Do seizures beget seizures?
 Does MTLE cause progressive neurocognitive damage? Andrew Bleasel Westmead Do seizures beget seizures? The tendency of the disease is toward self-perpetuation; each attack facilitates occurrence of another
Does MTLE cause progressive neurocognitive damage? Andrew Bleasel Westmead Do seizures beget seizures? The tendency of the disease is toward self-perpetuation; each attack facilitates occurrence of another
Subject Index. Band of Giacomini 22 Benton Visual Retention Test 66 68
 Subject Index Adams, R.D. 4 Addenbrooke s Cognitive Examination 101 Alzheimer s disease clinical assessment histological imaging 104 neuroimaging 101 104 neuropsychological assessment 101 clinical presentation
Subject Index Adams, R.D. 4 Addenbrooke s Cognitive Examination 101 Alzheimer s disease clinical assessment histological imaging 104 neuroimaging 101 104 neuropsychological assessment 101 clinical presentation
Magnetic Resonance Imaging of Mesial Temporal Sclerosis (MTS): What radiologists ought to know?
 Magnetic Resonance Imaging of Mesial Temporal Sclerosis (MTS): What radiologists ought to know? Poster No.: C-0856 Congress: ECR 2012 Type: Educational Exhibit Authors: P. Singh, G. Mittal, R. Kaur, K.
Magnetic Resonance Imaging of Mesial Temporal Sclerosis (MTS): What radiologists ought to know? Poster No.: C-0856 Congress: ECR 2012 Type: Educational Exhibit Authors: P. Singh, G. Mittal, R. Kaur, K.
Long-term and late seizure outcome after surgery for temporal lobe epilepsy
 Original article Epileptic Disord 2010; 12 (1): 54-8 Long-term and late seizure outcome after surgery for temporal lobe epilepsy José Pimentel 1, Carla Bentes 1,2, Alexandre Campos 3, A. Gonçalves Ferreira
Original article Epileptic Disord 2010; 12 (1): 54-8 Long-term and late seizure outcome after surgery for temporal lobe epilepsy José Pimentel 1, Carla Bentes 1,2, Alexandre Campos 3, A. Gonçalves Ferreira
Memory function decline over 18 months after selective amygdalohippocampectomy
 Original article Epileptic Disord 2004; 6: 115-20 Memory function decline over 18 months after selective amygdalohippocampectomy Tatsuya Ogino 1, Yoko Ohtsuka 1, Yumiko Ido 2, Yoshiaki Mayanagi 3, Eiju
Original article Epileptic Disord 2004; 6: 115-20 Memory function decline over 18 months after selective amygdalohippocampectomy Tatsuya Ogino 1, Yoko Ohtsuka 1, Yumiko Ido 2, Yoshiaki Mayanagi 3, Eiju
Epilepsy: diagnosis and treatment. Sergiusz Jóźwiak Klinika Neurologii Dziecięcej WUM
 Epilepsy: diagnosis and treatment Sergiusz Jóźwiak Klinika Neurologii Dziecięcej WUM Definition: the clinical manifestation of an excessive excitation of a population of cortical neurons Neurotransmitters:
Epilepsy: diagnosis and treatment Sergiusz Jóźwiak Klinika Neurologii Dziecięcej WUM Definition: the clinical manifestation of an excessive excitation of a population of cortical neurons Neurotransmitters:
Refractory focal epilepsy: findings by MRI.
 Refractory focal epilepsy: findings by MRI. Doctors Nicolás Sgarbi, Osmar Telis Clinical Radiology Department Hospital de Clínicas Montevideo- Uruguay ABSTRACT Epilepsy is one of the most frequent neurological
Refractory focal epilepsy: findings by MRI. Doctors Nicolás Sgarbi, Osmar Telis Clinical Radiology Department Hospital de Clínicas Montevideo- Uruguay ABSTRACT Epilepsy is one of the most frequent neurological
Postoperative routine EEG correlates with long-term seizure outcome after epilepsy surgery
 Seizure (2005) 14, 446 451 www.elsevier.com/locate/yseiz Postoperative routine EEG correlates with long-term seizure outcome after epilepsy surgery Michelle Hildebrandt a, Reinhard Schulz b, Matthias Hoppe
Seizure (2005) 14, 446 451 www.elsevier.com/locate/yseiz Postoperative routine EEG correlates with long-term seizure outcome after epilepsy surgery Michelle Hildebrandt a, Reinhard Schulz b, Matthias Hoppe
Clinical Study Qualitative and Quantitative Hippocampal MRI Assessments in Intractable Epilepsy
 BioMed Research International Volume 2013, Article ID 480524, 9 pages http://dx.doi.org/10.1155/2013/480524 Clinical Study Qualitative and Quantitative Hippocampal MRI Assessments in Intractable Epilepsy
BioMed Research International Volume 2013, Article ID 480524, 9 pages http://dx.doi.org/10.1155/2013/480524 Clinical Study Qualitative and Quantitative Hippocampal MRI Assessments in Intractable Epilepsy
Temporal lobe epilepsy (TLE) is the most common type of
 Differential Features of Metabolic Abnormalities Between Medial and Lateral Temporal Lobe Epilepsy: Quantitative Analysis of F-FDG PET Using SPM Yu Kyeong Kim, MD 1 ; Dong Soo Lee, MD, PhD 1 ; Sang Kun
Differential Features of Metabolic Abnormalities Between Medial and Lateral Temporal Lobe Epilepsy: Quantitative Analysis of F-FDG PET Using SPM Yu Kyeong Kim, MD 1 ; Dong Soo Lee, MD, PhD 1 ; Sang Kun
Magnetic resonance imaging (MR!) provides
 0 Wallerian Degeneration of the Pyramidal Tract in Capsular Infarction Studied by Magnetic Resonance Imaging Jesiis Pujol, MD, Josep L. Marti-Vilalta, MD, Carme Junqu6, PhD, Pere Vendrell, PhD, Juan Fernandez,
0 Wallerian Degeneration of the Pyramidal Tract in Capsular Infarction Studied by Magnetic Resonance Imaging Jesiis Pujol, MD, Josep L. Marti-Vilalta, MD, Carme Junqu6, PhD, Pere Vendrell, PhD, Juan Fernandez,
Correlation of Apparent Diffusion Coefficient with Neuropsychological Testing in Temporal Lobe Epilepsy
 AJNR Am J Neuroradiol 26:1832 1839, August 2005 Correlation of Apparent Diffusion Coefficient with Neuropsychological Testing in Temporal Lobe Epilepsy Yvonne W. Lui, Annette O. Nusbaum, William B. Barr,
AJNR Am J Neuroradiol 26:1832 1839, August 2005 Correlation of Apparent Diffusion Coefficient with Neuropsychological Testing in Temporal Lobe Epilepsy Yvonne W. Lui, Annette O. Nusbaum, William B. Barr,
Henry Molaison. Biography. From Wikipedia, the free encyclopedia
 Henry Molaison From Wikipedia, the free encyclopedia Henry Gustav Molaison (February 26, 1926 December 2, 2008), known widely as H.M., was an American memory disorder patient who had a bilateral medial
Henry Molaison From Wikipedia, the free encyclopedia Henry Gustav Molaison (February 26, 1926 December 2, 2008), known widely as H.M., was an American memory disorder patient who had a bilateral medial
A Hippocampal Lesion Detected by High-Field 3 Tesla Magnetic Resonance Imaging in a Patient with Temporal Lobe Epilepsy
 Tohoku J. Exp. Med., 2005, A Hippocampal 205, 287-291 Lesion Detected by High Field 3 T MRI 287 A Hippocampal Lesion Detected by High-Field 3 Tesla Magnetic Resonance Imaging in a Patient with Temporal
Tohoku J. Exp. Med., 2005, A Hippocampal 205, 287-291 Lesion Detected by High Field 3 T MRI 287 A Hippocampal Lesion Detected by High-Field 3 Tesla Magnetic Resonance Imaging in a Patient with Temporal
References. 1) Achten E. 1998: Aspects of proton MR spectroscopy in the seizure patients. Neuroimaging clinics of N. Am. Nov. 8 (4):
 1) Achten E. 1998: Aspects of proton MR spectroscopy in the seizure patients. Neuroimaging clinics of N. Am. Nov. 8 (4): 849 862. 2) Benbadis S.R., Agrawal V.K.,2002: Partial epilepsies, Neurol Clin. Jan.
1) Achten E. 1998: Aspects of proton MR spectroscopy in the seizure patients. Neuroimaging clinics of N. Am. Nov. 8 (4): 849 862. 2) Benbadis S.R., Agrawal V.K.,2002: Partial epilepsies, Neurol Clin. Jan.
MR and Positron Emission Tomography in the Diagnosis of Surgically Correctable Temporal Lobe Epilepsy
 MR and Positron Emission Tomography in the Diagnosis of Surgically Correctable Temporal Lobe Epilepsy R. Heinz, N. Ferris, E. K. Lee, R. Radtke, B. Crain, J. M. Hoffman, M. Hanson, S. Paine, and A. Friedman
MR and Positron Emission Tomography in the Diagnosis of Surgically Correctable Temporal Lobe Epilepsy R. Heinz, N. Ferris, E. K. Lee, R. Radtke, B. Crain, J. M. Hoffman, M. Hanson, S. Paine, and A. Friedman
Diffusion Tensor Imaging 12/06/2013
 12/06/2013 Beate Diehl, MD PhD FRCP University College London National Hospital for Neurology and Neurosurgery Queen Square London, UK American Epilepsy Society Annual Meeting Disclosure None Learning
12/06/2013 Beate Diehl, MD PhD FRCP University College London National Hospital for Neurology and Neurosurgery Queen Square London, UK American Epilepsy Society Annual Meeting Disclosure None Learning
Predictors of prognosis in patients with temporal lobe epilepsy after anterior temporal lobectomy
 1896 Predictors of prognosis in patients with temporal lobe epilepsy after anterior temporal lobectomy ZHENXING SUN 1*, HUANCONG ZUO 1, DAN YUAN 2, YAXING SUN 3, KAI ZHANG 4*, ZHIQIANG CUI 1 and JIN WANG
1896 Predictors of prognosis in patients with temporal lobe epilepsy after anterior temporal lobectomy ZHENXING SUN 1*, HUANCONG ZUO 1, DAN YUAN 2, YAXING SUN 3, KAI ZHANG 4*, ZHIQIANG CUI 1 and JIN WANG
The Surgical Treatment of Epilepsy
 The Surgical Treatment of Epilepsy Jeffrey S. Schweitzer, MD, PhD Kaiser Los Angeles Medical Center Division of Restorative Neurosurgery Ancient craniotomy When Cao started complaining about splitting
The Surgical Treatment of Epilepsy Jeffrey S. Schweitzer, MD, PhD Kaiser Los Angeles Medical Center Division of Restorative Neurosurgery Ancient craniotomy When Cao started complaining about splitting
SEIZURE OUTCOME AFTER EPILEPSY SURGERY
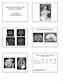 SEIZURE OUTCOME AFTER EPILEPSY SURGERY Prakash Kotagal, M.D. Head, Pediatric Epilepsy Cleveland Clinic Epilepsy Center LEFT TEMPORAL LOBE ASTROCYTOMA SEIZURE OUTCOME 1 YEAR AFTER EPILEPSY SURGERY IN ADULTS
SEIZURE OUTCOME AFTER EPILEPSY SURGERY Prakash Kotagal, M.D. Head, Pediatric Epilepsy Cleveland Clinic Epilepsy Center LEFT TEMPORAL LOBE ASTROCYTOMA SEIZURE OUTCOME 1 YEAR AFTER EPILEPSY SURGERY IN ADULTS
doi: /brain/awq006 Brain 2010: 133; Imaging memory in temporal lobe epilepsy: predicting the effects of temporal lobe resection
 doi:10.1093/brain/awq006 Brain 2010: 133; 1186 1199 1186 BRAIN A JOURNAL OF NEUROLOGY Imaging memory in temporal lobe epilepsy: predicting the effects of temporal lobe resection Silvia B. Bonelli, 1,2
doi:10.1093/brain/awq006 Brain 2010: 133; 1186 1199 1186 BRAIN A JOURNAL OF NEUROLOGY Imaging memory in temporal lobe epilepsy: predicting the effects of temporal lobe resection Silvia B. Bonelli, 1,2
The American Approach to Depth Electrode Insertion December 4, 2012
 The American Approach to Depth Electrode Insertion December 4, 2012 Jonathan Miller, MD Director, Epilepsy Surgery University Hospitals Case Medical Center/Case Western Reserve University Cleveland, Ohio
The American Approach to Depth Electrode Insertion December 4, 2012 Jonathan Miller, MD Director, Epilepsy Surgery University Hospitals Case Medical Center/Case Western Reserve University Cleveland, Ohio
doi: /brain/awt105 Brain 2013: 136;
 doi:10.1093/brain/awt105 Brain 2013: 136; 1889 1900 1889 BRAIN A JOURNAL OF NEUROLOGY Memory reorganization following anterior temporal lobe resection: a longitudinal functional MRI study Silvia B. Bonelli,
doi:10.1093/brain/awt105 Brain 2013: 136; 1889 1900 1889 BRAIN A JOURNAL OF NEUROLOGY Memory reorganization following anterior temporal lobe resection: a longitudinal functional MRI study Silvia B. Bonelli,
Semiology of Temporal Lobe Seizures: Value in Lateralizing the Seizure Focus
 Epilepsia, 39(7):721-726, 1998 Lippincott-Raven Publishers, Philadelphia 0 International League Against Epilepsy Semiology of Temporal Lobe Seizures: Value in Lateralizing the Seizure Focus William J.
Epilepsia, 39(7):721-726, 1998 Lippincott-Raven Publishers, Philadelphia 0 International League Against Epilepsy Semiology of Temporal Lobe Seizures: Value in Lateralizing the Seizure Focus William J.
Relative localizing value of amygdalo-hippocampal MR biometry in temporal lobe epilepsy
 Epilepsy Research 69 (2006) 147 164 Relative localizing value of amygdalo-hippocampal MR biometry in temporal lobe epilepsy Pedro M. Gonçalves Pereira a,b,, Eduardo Oliveira c, Pedro Rosado b,d a Department
Epilepsy Research 69 (2006) 147 164 Relative localizing value of amygdalo-hippocampal MR biometry in temporal lobe epilepsy Pedro M. Gonçalves Pereira a,b,, Eduardo Oliveira c, Pedro Rosado b,d a Department
Role of magnetic resonance imaging for preoperative evaluation of patients with refractory epilepsy
 ACF Hui JMK Lam YL Chan KM Au-Yeung KS Wong R Kay WS Poon Key words: Epilepsy; Magnetic resonance imaging; Surgery "# Hong Kong Med J 2003;9:20-4 The Chinese University of Hong Kong, Prince of Wales Hospital,
ACF Hui JMK Lam YL Chan KM Au-Yeung KS Wong R Kay WS Poon Key words: Epilepsy; Magnetic resonance imaging; Surgery "# Hong Kong Med J 2003;9:20-4 The Chinese University of Hong Kong, Prince of Wales Hospital,
Electro-clinical manifestations of the epilepsy associated to the different anatomical variants of hypothalamic hamartomas
 Electro-clinical manifestations of the epilepsy associated to the different anatomical variants of hypothalamic hamartomas Alberto JR Leal Hospital Fernando Fonseca, Dep. Neurology Lisbon. Abstract Objective
Electro-clinical manifestations of the epilepsy associated to the different anatomical variants of hypothalamic hamartomas Alberto JR Leal Hospital Fernando Fonseca, Dep. Neurology Lisbon. Abstract Objective
Anterior to posterior hippocampal MRS metabolite difference is mainly a partial volume effect
 Acta Radiologica ISSN: 0284-1851 (Print) 1600-0455 (Online) Journal homepage: http://www.tandfonline.com/loi/iard20 Anterior to posterior hippocampal MRS metabolite difference is mainly a partial volume
Acta Radiologica ISSN: 0284-1851 (Print) 1600-0455 (Online) Journal homepage: http://www.tandfonline.com/loi/iard20 Anterior to posterior hippocampal MRS metabolite difference is mainly a partial volume
The Low Sensitivity of Fluid-Attenuated Inversion-Recovery MR in the Detection of Multiple Sclerosis of the Spinal Cord
 The Low Sensitivity of Fluid-Attenuated Inversion-Recovery MR in the Detection of Multiple Sclerosis of the Spinal Cord Mark D. Keiper, Robert I. Grossman, John C. Brunson, and Mitchell D. Schnall PURPOSE:
The Low Sensitivity of Fluid-Attenuated Inversion-Recovery MR in the Detection of Multiple Sclerosis of the Spinal Cord Mark D. Keiper, Robert I. Grossman, John C. Brunson, and Mitchell D. Schnall PURPOSE:
