H. PYLORI AB, IGG HPYL
|
|
|
- Lora Richardson
- 5 years ago
- Views:
Transcription
1 H. PYLORI AB, IGG HPYL Specimen Required: 3 ml blood, Serum gel tube Analytical Time: 1 day CPT Code: HAPTOGLOBIN HAP Specimen Required: 1.5 ml blood, gel tube Reference Range: mg/dl CPT Code: HCG Pregnancy Test - Quantitative PREGQ Specimen Required: 2.5 ml blood (0.6 ml minimum), gold gel or green tube or 1 full microtainer. Reference Range: <5 miu/ml CPT Code: HDL CHOLESTEROL HDL Specimen Required: 2.5 ml blood (0.6 ml minimum), gold gel or green tube or 1 full microtainer. Reference Range: mg/dl CPT Code: HEMATOCRIT HCT Specimen Required: 1.5 ml blood, lavender top tube Reference Range: Age/sex dependent; see CBC CPT Code: 85014
2 HEMOGLOBIN HGB Specimen Required: 1.5 ml blood, lavender top tube Reference Range: Age/sex dependent; see CBC CPT Code: HEMOGLOBIN A1C See Glycohemoglobin HGBA1C HEMOGLOBIN ELECTROPHORESIS HGBE Specimen Required: 5 ml blood (0.5 ml minimum), lavender top tube Reference Range: Interpretive report with results Day(s) Test Set Up: Monday -Friday CPT Code: Test Includes: Hgb A1, Hgb A2, Hgb F, Hgb S, Hgb C, Abnormal Hgb, Hgb Phenotype HEPARIN ASSAY LMWH, FAC Xa, FACTOR 10a, LOW MOLC WT, UHF Specimen Required: 5ml blue top Reference range: IU/ml Analytical Time: 60 min CPT Code: Use for: Monitoring patients on Heparin HEPARIN INDUCED THROMBOCYTOPENIA SCREEN HIT Specimen Required: 10 ml plain red top tube (only) Reference Range: Positive (Automatically Reflex Serotonin Release Assay (SRA) Negative Analytical Time: 30min Day(s) Test Set up: Daily CPT: HEPATIC FUNCTION PANEL HEPATIC Specimen Required: Reference Range: 3.0 ml blood (0.8 ml minimum), gold gel or green tube or 1 full microtainer. See individual tests
3 CPT Code: Test Includes: Albumin, Total Bilirubin, Direct Bilirubin, Alkaline phosphatase, Total Protein, ALT (SGPT) AST (SGOT), (Calculated: Indirect Bilirubin) HEPATITIS A AB HAAB Day(s) Test Set Up: Monday-Friday CPT Code: HEPATITIS A AB - IGM HAIGM CPT Code: HEPATITIS B CORE AB HBC Specimen Required: 5.0 ml blood, gel tube Day(s) Test Set Up: Monday-Friday CPT Code: HEPATITIS B SURFACE AB HBSAB Specimen Required: 5.0 ml blood, gel tube CPT Code: HEPATITIS B SURFACE AG HBSAG Specimen Required: Reference Range: Analytical Time: 5 ml blood, gel tube Negative 4 hours
4 CPT Code: HEPATITIS BE ANTIBODY HBEAB Specimen Required: 2.5mL blood, red gel tube Day(s) Test Set Up: Monday, Wednesday and Friday CPT Code: HEPATITIS BE ANTIGEN HBEAG Specimen Required: 2.5mL blood, red gel tube Day(s) Test Set Up: Monday, Wednesday and Friday CPT Code: HEPATITIS C AB HCV Specimen Required: 1 ml blood, gel tube Special instructions: Centrifuge and refrigerate CPT Code: HEPATITIS PANEL ACUTE HEPABC Specimen Required : 7 ml gel tube Analytical Time: 2-5 days CPT Code: Tests Included: HAAb, IgM; HBcAb, IgM; HBsAG; HCV HERPES I & II SEROLOGY HERPES Specimen Required: 2.5 ml blood (1.5 ml minimum), gel tube Day(s) Test Set Up: Tuesday-Saturday CPT Code: 86695/86696
5 Test Includes: HSV-1, HSV-2 HGH (Human Growth Hormone) GH; HGH; GROWTH HORMONE Specimen Required: 1.5 ml blood, red gel tube Reference Range: Adult: 10 ng/ml or less Children: 20 ng/ml or less Analytical Time: 2-3 days Day(s) Test Set Up: Tuesday-Saturday CPT Code: HISTOPLASMA SEROLOGY HISAB; AHP; HISTO Analytical Time: 2-4 days Day(s) Test Set Up: Monday,Wednesday and Friday CPT Code: x2 Test Includes: Histo Ab mycelial, Histo Ab yeast HIT See Heparin Induced Thrombocytopenia Screen HIV-1/2 HIV Analytical Time: 1 day Day(s) Test Set Up: Monday-Friday CPT Code: NOTE: Repeatedly reactive specimens reflex to confirmation at additional charge by Western Blot. HLA-B27 HLAB27; HLA; HLAABCB Specimen Required: 10mL whole blood, yellow top tube(acd solution B Do Not Refrigerate Analytical Time: 2-4 days CPT Code: HPV(Human Papillomavirus), High Risk
6 HPV HR Specimen Required: Cervical swab submitted in liquid-based PAP container/ fresh cervical biopsy, Cervical or vaginal swab in PCR media transport. Day(s) Test Set Up: Twice/week CPT Code: HOMOCYSTEINE Specimen Required: 2.5 ml blood (0.6 ml minimum), gold gel tube or 1full microtainer. Reference Range: umol/l CPT Code: 83090
Reference Range: Age/sex dependent; appropriate normals provided with result Analytical Time: 4 hours Day(s) Test Set Up: Daily CPT Code: 84100
 Parathyroid Hormone See PTH PARIETAL CELL ANTIBODY APCAB; PARIETAL; PCA Specimen Required: 2.5 ml blood, gel tube Analytical Time: 3-5 days CPT Code: 86255 PCP SCREEN URINE PCPQU Specimen Required: 50
Parathyroid Hormone See PTH PARIETAL CELL ANTIBODY APCAB; PARIETAL; PCA Specimen Required: 2.5 ml blood, gel tube Analytical Time: 3-5 days CPT Code: 86255 PCP SCREEN URINE PCPQU Specimen Required: 50
M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017
 M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017 If laboratory results are required on a STAT basis, the designated commercial medical laboratory
M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017 If laboratory results are required on a STAT basis, the designated commercial medical laboratory
DEPARTMENT: Regulatory Compliance Support
 PAGE: 1 of 5 REPLACES POLICY DATED: 1/16/98, 3/1/99, SCOPE: All Company-affiliated hospitals performing and/or billing laboratory services. Specifically, the following departments: Business Office Admitting/Registration
PAGE: 1 of 5 REPLACES POLICY DATED: 1/16/98, 3/1/99, SCOPE: All Company-affiliated hospitals performing and/or billing laboratory services. Specifically, the following departments: Business Office Admitting/Registration
TEST REQUEST INFORMATION- VIROLOGY
 Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
C-1-Q IMMUNE COMPLEX Immune Complex, C-1-Q Binding Assay
 C-1-Q IMMUNE COMPLEX Immune Complex, C-1-Q Binding Assay 5 ml blood, gel tube Special Instructions: Avoid hemolysis. Transfer serum to plastic vial immediately after centrifugation and freeze. Less than
C-1-Q IMMUNE COMPLEX Immune Complex, C-1-Q Binding Assay 5 ml blood, gel tube Special Instructions: Avoid hemolysis. Transfer serum to plastic vial immediately after centrifugation and freeze. Less than
TEST LIST SAMPLE REQUIREMENT. 1 ml serum None
 ALBUMIN TEST NAME ALKALINE PHOSPHATASE ALLERGY PROFILE, FOOD 30 allergens ALLERGY PROFILE, INHALANT 30 Allergens ALT AMYLASE ANA ANTI- TG ANTI-GLIADIN IGG ANTI-GLIADIN IGA ANTI-HBS ANTI-HCV ANTI-TPO APOLIPOPROTEIN
ALBUMIN TEST NAME ALKALINE PHOSPHATASE ALLERGY PROFILE, FOOD 30 allergens ALLERGY PROFILE, INHALANT 30 Allergens ALT AMYLASE ANA ANTI- TG ANTI-GLIADIN IGG ANTI-GLIADIN IGA ANTI-HBS ANTI-HCV ANTI-TPO APOLIPOPROTEIN
Refugee Health Funding Models: A Review of PA Models and A Vision for the Future
 Refugee Health Funding Models: A Review of PA Models and A Vision for the Future Gretchen Shanfeld, MPH Director of Health and Wellness, Nationalities Service Center Coordinator, Philadelphia Refugee Health
Refugee Health Funding Models: A Review of PA Models and A Vision for the Future Gretchen Shanfeld, MPH Director of Health and Wellness, Nationalities Service Center Coordinator, Philadelphia Refugee Health
BASIC METABOLIC PANEL
 Update 2/12/2018 BASIC METABOLIC PANEL CPT 80048 Stability: 3 days at 15-25 C; 7 days at 2-8 C; > 7 days at -70 C Colorimetric Assay, Rate reaction, ISE Components: BUN, Calcium, Chloride, CO2, Creatinine,
Update 2/12/2018 BASIC METABOLIC PANEL CPT 80048 Stability: 3 days at 15-25 C; 7 days at 2-8 C; > 7 days at -70 C Colorimetric Assay, Rate reaction, ISE Components: BUN, Calcium, Chloride, CO2, Creatinine,
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
GSK , 2.0, 18, 2014, DAIDS
 Letter of Amendment # 1 to: HPTN 077: A Phase IIa Study to Evaluate the, Tolerability and Pharmacokinetics of the Investigational Injectable HIV Integrase Inhibitor, GSK1265744, in HIV-uninfected Men and
Letter of Amendment # 1 to: HPTN 077: A Phase IIa Study to Evaluate the, Tolerability and Pharmacokinetics of the Investigational Injectable HIV Integrase Inhibitor, GSK1265744, in HIV-uninfected Men and
To be used for the ease of test requisitioning on select patients only; all components may be ordered separately
 Panels Section To be used for the ease of test requisitioning on select patients only; all components may be ordered separately ANEMIA 1(I) PANEL (NMC & UMMC) ANEM1(I) S 8.0 ml Large Tests included are:
Panels Section To be used for the ease of test requisitioning on select patients only; all components may be ordered separately ANEMIA 1(I) PANEL (NMC & UMMC) ANEM1(I) S 8.0 ml Large Tests included are:
JAJ International, Inc./LuSys Laboratories, Inc.
 JAJ International, Inc./LuSys Laboratories, Inc. 10054 Mesa Ridge Court, Suite #120 San Diego, California 92121 USA Tel: 1(858) 866-0788, Fax: 1(858) 866-1688 Email: info@jajinternational.com Website:
JAJ International, Inc./LuSys Laboratories, Inc. 10054 Mesa Ridge Court, Suite #120 San Diego, California 92121 USA Tel: 1(858) 866-0788, Fax: 1(858) 866-1688 Email: info@jajinternational.com Website:
WOMEN'S INTERAGENCY HIV STUDY
 WOMEN'S INTERAGENCY HIV STUDY SECTION 6: OVERVIEW OF THE BASELINE VISIT FOR NEW RECRUITS I. 2001/2002 RECRUITS This section provides an overview of the components of the baseline visit for 2001/2002 new
WOMEN'S INTERAGENCY HIV STUDY SECTION 6: OVERVIEW OF THE BASELINE VISIT FOR NEW RECRUITS I. 2001/2002 RECRUITS This section provides an overview of the components of the baseline visit for 2001/2002 new
Policy # MI/SER/02/v03 Page 1 of 9 Policy & Procedure Manual
 Policy # MI/SER/02/v03 Page 1 of 9 Section: Subject Title: AxSYM System Issued by: LABORATORY MANAGER Original Date: March 14, 2001 Approved by: Laboratory Director Revision Date: October 28, 2003 AXSYM
Policy # MI/SER/02/v03 Page 1 of 9 Section: Subject Title: AxSYM System Issued by: LABORATORY MANAGER Original Date: March 14, 2001 Approved by: Laboratory Director Revision Date: October 28, 2003 AXSYM
March Immunoglobulins, Total (IgA, IgE, IgG, IgM) Interleukin-6 (IL6) Protein Electrophoresis, CSF. Thyrotropin Receptor Antibody
 University of Washington March 2012 TEST CHANGE Alpha 1-Antitrypsin Anti-IgE Receptor Antibody Anti-Mullerian Hormone Aquaporin-4 Receptor Antibody ColoSeq - Polyposis Complement testing, C3, C4 Everolimus
University of Washington March 2012 TEST CHANGE Alpha 1-Antitrypsin Anti-IgE Receptor Antibody Anti-Mullerian Hormone Aquaporin-4 Receptor Antibody ColoSeq - Polyposis Complement testing, C3, C4 Everolimus
Testing Kits. Pregnancy Test Kits. Hepatitis Test Kits. HIV - 1 & 2 Test Kits
 Pregnancy Test Kits Sr Item Name Store At Pack Size 1 hcg Card Test 2 hcg Card Test 3 Hope hcg Card Test 4 hcg Dipstick Test 5 LH Surge Card Test 5 T 6 LH Surge Card Test 10 T Hepatitis Test Kits 7 HCsAg
Pregnancy Test Kits Sr Item Name Store At Pack Size 1 hcg Card Test 2 hcg Card Test 3 Hope hcg Card Test 4 hcg Dipstick Test 5 LH Surge Card Test 5 T 6 LH Surge Card Test 10 T Hepatitis Test Kits 7 HCsAg
Please contact Customer Service with any questions. Ph: option 5
 Sarasota Memorial Laboratory Services is pleased to announce that we are now performing the following testing in house: Immature Platelet Fraction (IPF) The Immature Platelet Fraction (IPF) is used to
Sarasota Memorial Laboratory Services is pleased to announce that we are now performing the following testing in house: Immature Platelet Fraction (IPF) The Immature Platelet Fraction (IPF) is used to
Recommendations for Celiac Disease Testing. IgA & ttg IgA
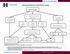 Recommendations for Celiac Disease Testing IgA & ttg IgA IgA Normal ttg IgA Negative IgA >10 mg/dl, but less than age matched range IgA
Recommendations for Celiac Disease Testing IgA & ttg IgA IgA Normal ttg IgA Negative IgA >10 mg/dl, but less than age matched range IgA
CPT Code: Serum Urea Nitrogen, Urine Urea Nitrogen in mg/dl, 24 hour Urine Urea Nitrogen, Urea Clearance
 UREA CLEARANCE UREACL Specimen Required: 2.5 ml blood, gel tube and total collection 24 hour urine Special Instructions: Refrigerate urine during collection. Provide starting date and time, and ending
UREA CLEARANCE UREACL Specimen Required: 2.5 ml blood, gel tube and total collection 24 hour urine Special Instructions: Refrigerate urine during collection. Provide starting date and time, and ending
lab Test Name notes on collection container type CBC AUTO HEMOGLOBIN & HEMATOCRIT
 lab Test Name notes on collection container type CBC AUTO HEMOGLOBIN & HEMATOCRIT CBC HEMOGRAM LAV HEMOGLOBIN HEMATOCRIT PLATELET COUNT & IPF LAV ERYTHROCYTE SEDIMENTATION RATE MALARIA SMEAR RETICULOCYTE
lab Test Name notes on collection container type CBC AUTO HEMOGLOBIN & HEMATOCRIT CBC HEMOGRAM LAV HEMOGLOBIN HEMATOCRIT PLATELET COUNT & IPF LAV ERYTHROCYTE SEDIMENTATION RATE MALARIA SMEAR RETICULOCYTE
Specific Panels. Celiac disease panel. Pancreas Panel:
 Specific Panels Celiac disease panel Anti Endomysium IgA Anti Endomysium IgG Anti Gliadin IgA Anti Gliadin IgG Anti Transglutaminase IgA Anti Transglutaminase IgG Total IgA Total IgG Stool analysis +Sudan
Specific Panels Celiac disease panel Anti Endomysium IgA Anti Endomysium IgG Anti Gliadin IgA Anti Gliadin IgG Anti Transglutaminase IgA Anti Transglutaminase IgG Total IgA Total IgG Stool analysis +Sudan
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
$ Glucose Only $ 9.00 Detects diabetes or a pre-diabetic condition.
 Glucose Only $ 9.00 Detects diabetes or a pre-diabetic condition. Hemoglobin Ale $ 19.00 Used to monitor glucose over a period of time. Detects diabetes or pre-diabetes. Insulin, Fasting $ 19.00 Used as
Glucose Only $ 9.00 Detects diabetes or a pre-diabetic condition. Hemoglobin Ale $ 19.00 Used to monitor glucose over a period of time. Detects diabetes or pre-diabetes. Insulin, Fasting $ 19.00 Used as
Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
 Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
M Series. Health Screening Just Got A Whole Lot Easier EARLY DETECTION BETTER MANAGEMENT IMPROVED OUTCOMES
 M Series Health Screening Just Got A Whole Lot Easier EARLY DETECTION BETTER MANAGEMENT IMPROVED OUTCOMES ABOUT MINMED We are a progressive medical group that enlarges organically by growing constantly,
M Series Health Screening Just Got A Whole Lot Easier EARLY DETECTION BETTER MANAGEMENT IMPROVED OUTCOMES ABOUT MINMED We are a progressive medical group that enlarges organically by growing constantly,
Lab Underwriting Puzzler. Presented by: Bill Rooney, M.D.
 Lab Underwriting Puzzler Presented by: Bill Rooney, M.D. Obtaining Best Results from this presentation For best results please do the following: Select Slide Show from the menu option on top Select From
Lab Underwriting Puzzler Presented by: Bill Rooney, M.D. Obtaining Best Results from this presentation For best results please do the following: Select Slide Show from the menu option on top Select From
ICL Integrative Laboratory Services Test Menu Contact ICL Client Care x300
 Alletess Food Sensitivity Fingerstick 96 Foods IgG with or without Wellness Program 184 Foods IgG with or without Wellness Program Alletess Food Allergy/Sensitivity Serum 96 Foods IgG with or without Wellness
Alletess Food Sensitivity Fingerstick 96 Foods IgG with or without Wellness Program 184 Foods IgG with or without Wellness Program Alletess Food Allergy/Sensitivity Serum 96 Foods IgG with or without Wellness
Alaska Native Medical Center Anchorage, AK
 ANMC Lab Test Requirements Key: Room Temp (20-25C), Refrigerated (2-8C), (-15 to -25C), Hr (Hours), D (Days), W (Weeks), Mo (Months), Yr (Years). Basic Processing Instructions: Centrifuge all blood specimens
ANMC Lab Test Requirements Key: Room Temp (20-25C), Refrigerated (2-8C), (-15 to -25C), Hr (Hours), D (Days), W (Weeks), Mo (Months), Yr (Years). Basic Processing Instructions: Centrifuge all blood specimens
IN THE NAME OF GOD. D r. MANIJE DEZFULI AZAD UNIVERCITY OF TEHRAN BOOALI HOSPITAL INFECTIOUS DISEASES SPECIALIST
 IN THE NAME OF GOD AZAD UNIVERCITY OF TEHRAN BOOALI HOSPITAL D r. MANIJE DEZFULI INFECTIOUS DISEASES SPECIALIST Acute Viral Hepatitis The Anatomy of the Liver Hepatic Physiology Liver: Largest solid organ
IN THE NAME OF GOD AZAD UNIVERCITY OF TEHRAN BOOALI HOSPITAL D r. MANIJE DEZFULI INFECTIOUS DISEASES SPECIALIST Acute Viral Hepatitis The Anatomy of the Liver Hepatic Physiology Liver: Largest solid organ
Who to Treat? Consider biopsy Treat. > 2 ULN Treat Treat Treat Treat CIRRHOTIC PATIENTS Compensated Treat HBV DNA detectable treat
 Who to Treat? Parameter AASLD US Algorithm EASL APASL HBV DNA CRITERIA HBeAg+ >, IU/mL > 2, IU/mL > 2, IU/mL >, IU/mL HBeAg- > 2, IU/mL > 2, IU/mL > 2, IU/mL > 2, IU/mL ALT CRITERIA PNALT 1-2 ULN Monitor
Who to Treat? Parameter AASLD US Algorithm EASL APASL HBV DNA CRITERIA HBeAg+ >, IU/mL > 2, IU/mL > 2, IU/mL >, IU/mL HBeAg- > 2, IU/mL > 2, IU/mL > 2, IU/mL > 2, IU/mL ALT CRITERIA PNALT 1-2 ULN Monitor
Medicaid Family Planning Waiver Services CPT Codes and ICD-10 Diagnosis Codes
 CPT Code Description of Covered Codes Evaluation and Management 99384FP 99385FP Family planning new visit 99386FP 99394FP 99395FP Family planning established visit 99396FP 99401FP HIV counseling (pre-test)
CPT Code Description of Covered Codes Evaluation and Management 99384FP 99385FP Family planning new visit 99386FP 99394FP 99395FP Family planning established visit 99396FP 99401FP HIV counseling (pre-test)
CLIA APPROVED PROFICIENCY TESTING PROGRAMS ACCUTEST, INC. P.O. Box 999 Westford, Massachusetts (800)
 ACCUTEST, INC. P.O. Box 999 Westford, Massachusetts 01886 (800) 665-2575 MICROBIOLOGY Bacteriology Aerobic Culture and Identification Antibiotic Susceptibility Testing Direct Antigen Detection Gram Stain
ACCUTEST, INC. P.O. Box 999 Westford, Massachusetts 01886 (800) 665-2575 MICROBIOLOGY Bacteriology Aerobic Culture and Identification Antibiotic Susceptibility Testing Direct Antigen Detection Gram Stain
Test Name Results Units Bio. Ref. Interval
 LL - LL-ROHINI (NATIONAL REFERENCE 135091606 Age 24 Years Gender Male 30/8/2017 92800AM 30/8/2017 94631AM 31/8/2017 90306AM Ref By Final HEATITIS A & B VIRUS EVALUATION HEATITIS A ANTIBODY (ANTI HAV),
LL - LL-ROHINI (NATIONAL REFERENCE 135091606 Age 24 Years Gender Male 30/8/2017 92800AM 30/8/2017 94631AM 31/8/2017 90306AM Ref By Final HEATITIS A & B VIRUS EVALUATION HEATITIS A ANTIBODY (ANTI HAV),
Sutter Health Plus Effective for Calendar Year 2015
 Sutter Health Plus Effective for Calendar Year 2015 CPTs CPT Descriptions 2015 Cost Under Deducible (Single Unit) Doctor's Office Visit for a New Patient (Also Urgent Care) 99201 Low Level Visit $99.00
Sutter Health Plus Effective for Calendar Year 2015 CPTs CPT Descriptions 2015 Cost Under Deducible (Single Unit) Doctor's Office Visit for a New Patient (Also Urgent Care) 99201 Low Level Visit $99.00
June Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 June 2013 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test
June 2013 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test
Controls & Calibrators. Disease Quality Controls
 Controls & Calibrators Infectious Disease Quality Controls Infectious Disease Quality Controls A broad selection of controls designed to monitor assay precision of hepatitis, retrovirus, sexually transmitted
Controls & Calibrators Infectious Disease Quality Controls Infectious Disease Quality Controls A broad selection of controls designed to monitor assay precision of hepatitis, retrovirus, sexually transmitted
Code TEST SPECIMEN REQUIREMENT TAT Referral centre. 1 week BP GM (Specimen stability: 2-8 C for 48 hours) 3240 Anti-ds DNA 3 ml Plain Blood/Serum
 B.P. CLINICAL LAB SDN. BHD. ESOTERIC TEST LIST Note: For campaign profile, please notify processing lab 3 weeks in advanced for reagent and consumables preparation. Failure to pre-notify lab will cause
B.P. CLINICAL LAB SDN. BHD. ESOTERIC TEST LIST Note: For campaign profile, please notify processing lab 3 weeks in advanced for reagent and consumables preparation. Failure to pre-notify lab will cause
A007 Allergy Basic Profile (28 parameters) 5 ml Plain Blood/Serum week BPGM
 B.P. CLINICAL LAB SDN. BHD. ESOTERIC TEST LIST Note: For campaign profile, please notify processing lab 3 weeks in advanced for reagent and consumables preparation. Failure to pre-notify lab will cause
B.P. CLINICAL LAB SDN. BHD. ESOTERIC TEST LIST Note: For campaign profile, please notify processing lab 3 weeks in advanced for reagent and consumables preparation. Failure to pre-notify lab will cause
CASE-BASED SMALL GROUP DISCUSSION
 MHD II, Session XII, STUDENT Copy Page 1 CASE-BASED SMALL GROUP DISCUSSION Session XII MHD II April 21, 2014 STUDENT COPY Helpful Resource: ACP Medicine online available through LUHS Library Infectious
MHD II, Session XII, STUDENT Copy Page 1 CASE-BASED SMALL GROUP DISCUSSION Session XII MHD II April 21, 2014 STUDENT COPY Helpful Resource: ACP Medicine online available through LUHS Library Infectious
Online catalog
 This catalog contains information about tests performed at Green Clinic Laboratory. For samples to be sent to Quest Diagnostics or any other reference lab please contact the Green Clinic Laboratory (318-251-6378)
This catalog contains information about tests performed at Green Clinic Laboratory. For samples to be sent to Quest Diagnostics or any other reference lab please contact the Green Clinic Laboratory (318-251-6378)
Ward Manual. PHC Diagnostic Virology & Reference Laboratory
 PHC Diagnostic Virology & Reference Laboratory I. General Information The SPH Virology Laboratory is located at St. Paul s Hospital 1081 Burrard Street, Room 625, Burrard Building, Vancouver, B.C. V6Z
PHC Diagnostic Virology & Reference Laboratory I. General Information The SPH Virology Laboratory is located at St. Paul s Hospital 1081 Burrard Street, Room 625, Burrard Building, Vancouver, B.C. V6Z
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates September 25, 2009 Dear Colleague: Specialty Laboratories is pleased to announce a new assay, PT and PTT-LA Mixing
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates September 25, 2009 Dear Colleague: Specialty Laboratories is pleased to announce a new assay, PT and PTT-LA Mixing
Basic Metabolic Panel
 Basic Metabolic Panel Order Name: CHEM 8 Test Number: 2028100 REV DATE:2/5/2008 Glucose Urea Nitrogen, Blood (BUN) Creatinine Sodium Potassium Serum/Plasma Chloride Bicarbonate Calcium Anion Gap Calculated
Basic Metabolic Panel Order Name: CHEM 8 Test Number: 2028100 REV DATE:2/5/2008 Glucose Urea Nitrogen, Blood (BUN) Creatinine Sodium Potassium Serum/Plasma Chloride Bicarbonate Calcium Anion Gap Calculated
VOICE Screening Part 1 Visit. Operational Walkthrough Johannesburg, South Africa November 2008
 VOICE Screening Part 1 Visit Operational Walkthrough Johannesburg, South Africa November 2008 Protocol Requirements Administrative, Behavioral, and Regulatory Procedures Informed consent for screening
VOICE Screening Part 1 Visit Operational Walkthrough Johannesburg, South Africa November 2008 Protocol Requirements Administrative, Behavioral, and Regulatory Procedures Informed consent for screening
Minimum Whole Blood Volumes for Microcollection Tubes for Neonates, Pediatrics, Patients less than 45 kg (100 lb) and Difficult Collections
 The following table outlines the minimum whole blood volume that must be drawn into a microcollection tube (unless otherwise stated) for an individual test on a neonate, pediatric, patient weighing less
The following table outlines the minimum whole blood volume that must be drawn into a microcollection tube (unless otherwise stated) for an individual test on a neonate, pediatric, patient weighing less
HEALTH ASSESSMENT FOR NEW ZEALAND VISA APPLICANTS
 INFORMATION SHEET HEALTH ASSESSMENT FOR NEW ZEALAND VISA APPLICANTS Health assessments of New Zealand visa applicants are conducted by the International Organization for Migration (IOM) on behalf of Immigration
INFORMATION SHEET HEALTH ASSESSMENT FOR NEW ZEALAND VISA APPLICANTS Health assessments of New Zealand visa applicants are conducted by the International Organization for Migration (IOM) on behalf of Immigration
Clinician Blood Panel Results
 Page 1 of 8 Blood Panel - Markers Out of Range and Patterns (Pattern: proprietary formula using one or more Blood Markers) Blood Panel: Check for Markers that are out of Lab Range ***NOTE*** Only one supplement
Page 1 of 8 Blood Panel - Markers Out of Range and Patterns (Pattern: proprietary formula using one or more Blood Markers) Blood Panel: Check for Markers that are out of Lab Range ***NOTE*** Only one supplement
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates July 27, 2009 Dear Colleague: As we prepare for the upcoming flu season, Specialty Laboratories clients should be aware
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates July 27, 2009 Dear Colleague: As we prepare for the upcoming flu season, Specialty Laboratories clients should be aware
Diagnostic Methods of HBV infection. Zohreh Sharifi,ph.D of Virology Research center, Iranian Blood Transfusion Organization (IBTO)
 Diagnostic Methods of HBV infection Zohreh Sharifi,ph.D of Virology Research center, Iranian Blood Transfusion Organization (IBTO) Hepatitis B-laboratory diagnosis Detection of HBV infection involves
Diagnostic Methods of HBV infection Zohreh Sharifi,ph.D of Virology Research center, Iranian Blood Transfusion Organization (IBTO) Hepatitis B-laboratory diagnosis Detection of HBV infection involves
Robert G. Gish MD UC San Diego
 Robert G. Gish MD UC San Diego The Pipeline of HIV-Related Point-Of- Care Tests is Growing Viral detection Examples: 2012 2013 2014 2015 2016 2017 CD4 test Examples: 2009 2010 2011 2012 2013 0 2014 Evidence
Robert G. Gish MD UC San Diego The Pipeline of HIV-Related Point-Of- Care Tests is Growing Viral detection Examples: 2012 2013 2014 2015 2016 2017 CD4 test Examples: 2009 2010 2011 2012 2013 0 2014 Evidence
Viral Hepatitis Diagnosis and Management
 Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Epic Labs Orderable As STAT PRIORITY As of 06/22/2016
 ABG+HB(CORDARTERIAL) - BABY A ABG+HB(CORD ARTERIAL)- BABY B ABG+HB(CORD ARTERIAL)- BABY C ACETAMINOPHEN LEVEL ALANINE AMINOTRANSFERASE (ALT) ALBUMIN, FLUID ALBUMIN, PLEURAL FLUID ALBUMIN, SYNOVIAL FLUID
ABG+HB(CORDARTERIAL) - BABY A ABG+HB(CORD ARTERIAL)- BABY B ABG+HB(CORD ARTERIAL)- BABY C ACETAMINOPHEN LEVEL ALANINE AMINOTRANSFERASE (ALT) ALBUMIN, FLUID ALBUMIN, PLEURAL FLUID ALBUMIN, SYNOVIAL FLUID
Procedure Description Modifier 33 Required? Screening test of visual acuity, quantitative, bilateral No Z Z00.129
 Policy Name: Preventive Health Guidelines - Newborns and Children This policy applies only to non-grandfathered plans as defined in the Affordable Care Act section 1251. The following chart contains procedure,
Policy Name: Preventive Health Guidelines - Newborns and Children This policy applies only to non-grandfathered plans as defined in the Affordable Care Act section 1251. The following chart contains procedure,
Call for Appointments
 Ala Carte Pricing For Men and Women Consultation and Full Physical $60 Doctor Examination Body Height, Weight, Blood Pressure, Pulse Rate, BMI, Waist Circumference Breast examination (Women) Prostate Examination
Ala Carte Pricing For Men and Women Consultation and Full Physical $60 Doctor Examination Body Height, Weight, Blood Pressure, Pulse Rate, BMI, Waist Circumference Breast examination (Women) Prostate Examination
Monday - Friday Monday - Friday Referred out weekdays to the Public Health Laboratory
 H. pylori (see Helicobacter Serology) H. Pylori Breath Test test only available to Grey Bruce, Owen Sound and St. Mary's, Kitchener C14 Breath test PY Test C-Urea Breath Test Toxicology/Speci al Chemistry
H. pylori (see Helicobacter Serology) H. Pylori Breath Test test only available to Grey Bruce, Owen Sound and St. Mary's, Kitchener C14 Breath test PY Test C-Urea Breath Test Toxicology/Speci al Chemistry
Maximizing Cornea and Tissue Donation through Specimen Quality
 Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
REFLEX TESTING LIST March 2012
 Aeromonas/Plesiomonas Positive findings Organism ID & Susceptibility April 2011 Culture *AFB Culture and Stain Positive findings Organism ID & Susceptibility Nov. 1999 *AFB Culture Only Positive findings
Aeromonas/Plesiomonas Positive findings Organism ID & Susceptibility April 2011 Culture *AFB Culture and Stain Positive findings Organism ID & Susceptibility Nov. 1999 *AFB Culture Only Positive findings
Reflex Test Protocols
 UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
Test Name Results Units Bio. Ref. Interval
 LL - LL-ROHINI (NATIONAL REFERENCE 135091650 Age 49 Years Gender Male 29/8/2017 120000AM 29/8/2017 100248AM 29/8/2017 105306AM Ref By Final HEATITIS, VIRAL, COMREHENSIVE ANEL HEATITIS A ANTIBODY (ANTI
LL - LL-ROHINI (NATIONAL REFERENCE 135091650 Age 49 Years Gender Male 29/8/2017 120000AM 29/8/2017 100248AM 29/8/2017 105306AM Ref By Final HEATITIS, VIRAL, COMREHENSIVE ANEL HEATITIS A ANTIBODY (ANTI
Understanding Blood Tests
 PATIENT EDUCATION patienteducation.osumc.edu Your heart pumps the blood in your body through a system of blood vessels. Blood delivers oxygen and nutrients to all parts of the body. It also carries away
PATIENT EDUCATION patienteducation.osumc.edu Your heart pumps the blood in your body through a system of blood vessels. Blood delivers oxygen and nutrients to all parts of the body. It also carries away
READ HIGHLIGHTED CHANGES
 BIO-FLASH anti-hbs 3000-8581 100 tests The BIO-FLASH anti-hbs is a fully automated chemiluminescent simultaneous immunoassay for quantitative measurement of antibodies to Hepatitis B surface antigen (anti-hbs)
BIO-FLASH anti-hbs 3000-8581 100 tests The BIO-FLASH anti-hbs is a fully automated chemiluminescent simultaneous immunoassay for quantitative measurement of antibodies to Hepatitis B surface antigen (anti-hbs)
DAT-B 2015 Direct Antiglobulin Test
 EVALUATION DAT-B 2015 Direct Antiglobulin Test INSTITUTION: ATTENTION: CAP NUMBER: 7181660-01 Kit# 1 KIT INFORMATION: Kit ID: 28002258 10/12/2015 11/16/2015 Next Mailing Date: 2/22/2016 COPIED TO: LAP
EVALUATION DAT-B 2015 Direct Antiglobulin Test INSTITUTION: ATTENTION: CAP NUMBER: 7181660-01 Kit# 1 KIT INFORMATION: Kit ID: 28002258 10/12/2015 11/16/2015 Next Mailing Date: 2/22/2016 COPIED TO: LAP
PROFILES MENU. Page no. 9 Page no. 8 Page no. 7 Page no. 6 Page no. 5. Page no. 2. Page no. 4 Page no. 3
 Health Profiles PROFILES MENU Page no. 1 Page no. 2 Page no. 4 Page no. 3 1 Basic Metabolic Panel (BMP) 2 Liver Function Tests (LFT) 3 Comprehensive Metabolic Panel (CMP) 4 Lipid Profile 5 Chem 7 Panel
Health Profiles PROFILES MENU Page no. 1 Page no. 2 Page no. 4 Page no. 3 1 Basic Metabolic Panel (BMP) 2 Liver Function Tests (LFT) 3 Comprehensive Metabolic Panel (CMP) 4 Lipid Profile 5 Chem 7 Panel
Monitoring Hepatitis C
 Monitoring Hepatitis C Section Six Monitoring Hepatitis C Screening for hepatitis C is not routinely done, so you may have to request a test from your medical provider. This usually involves an antibody
Monitoring Hepatitis C Section Six Monitoring Hepatitis C Screening for hepatitis C is not routinely done, so you may have to request a test from your medical provider. This usually involves an antibody
March Molecular diagnostics. Update distribution changes. Billing and compliance. Hematology. Immunology. Referral testing
 March 2016 Update distribution changes Billing and compliance Compliance Drug Analysis CPT change Hematology Hemoglobin A1c, Screening Reference Range Change Immunology Testing schedule changes Vitamin
March 2016 Update distribution changes Billing and compliance Compliance Drug Analysis CPT change Hematology Hemoglobin A1c, Screening Reference Range Change Immunology Testing schedule changes Vitamin
HIV Basics: Clinical Tests and Guidelines
 HIV Basics: Clinical Tests and Guidelines ACTHIV 2010 Zelalem Temesgen MD Mayo Clinic Topics Baseline laboratory evaluation Laboratory monitoring through the continuum of care Patients not on antiretroviral
HIV Basics: Clinical Tests and Guidelines ACTHIV 2010 Zelalem Temesgen MD Mayo Clinic Topics Baseline laboratory evaluation Laboratory monitoring through the continuum of care Patients not on antiretroviral
Test Definition: FNMR2 NMR LipoProfile w/ir Markers
 Reporting Title: Performing Location: Labcorp Burlington Specimen Requirements: Submit only one of the following specimens: Serum Draw blood in a plain red-top tube(s). (Serum gel tube is not acceptable.)
Reporting Title: Performing Location: Labcorp Burlington Specimen Requirements: Submit only one of the following specimens: Serum Draw blood in a plain red-top tube(s). (Serum gel tube is not acceptable.)
Laboratory Millennium Special Pathology Laboratory, Office No. 10, 11, 12, First Floor, N.K. Image, Tilak Road, 2015 Sadashiv Peth, Pune, Maharashtra
 Last Amended on - Page 1 of 18 Specific Method Range of ing / I. CLINICAL BIOCHEMISTRY ENDOCRINOLOGY TESTS THYROID HORMONES 1. SERUM Triiodothyronine (T3) Chemiluminescence 0.1 8 ng/ml 6.8 2. SERUM Thyroxine
Last Amended on - Page 1 of 18 Specific Method Range of ing / I. CLINICAL BIOCHEMISTRY ENDOCRINOLOGY TESTS THYROID HORMONES 1. SERUM Triiodothyronine (T3) Chemiluminescence 0.1 8 ng/ml 6.8 2. SERUM Thyroxine
BLOOD WORK: A USEFUL TOOL FOR MONITORING HIV
 BLOOD WORK: A USEFUL TOOL FOR MONITORING HIV A PUBLICATION FROM Information, Inspiration and Advocacy for People Living With HIV/AIDS MAY 2007 Lab tests, or blood work, can give important clues about your
BLOOD WORK: A USEFUL TOOL FOR MONITORING HIV A PUBLICATION FROM Information, Inspiration and Advocacy for People Living With HIV/AIDS MAY 2007 Lab tests, or blood work, can give important clues about your
HOVON 141 CLL. Version 3, 25JUL2018. Table Required investigations at entry, during treatment and during follow up.
 Table 10.2.1 Required investigations at entry, during treatment and during follow up At entry 1 each induction cycle cycle 2 Cycle 3 day 8 and 15 Cycle 4 day1 and 6 cycle 9 day 15 of cycle 15 9 cycle 15:
Table 10.2.1 Required investigations at entry, during treatment and during follow up At entry 1 each induction cycle cycle 2 Cycle 3 day 8 and 15 Cycle 4 day1 and 6 cycle 9 day 15 of cycle 15 9 cycle 15:
1. Purpose 1.1. To define testing locations, schedule and order priority for each test performed in the core laboratory.
 Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
NORTH SHORE - LONG ISLAND JEWISH HEALTH SYSTEM LONG ISLAND JEWISH MEDICAL CENTER DEPARTMENT OF LABORATORY MEDICINE REFERENCE RANGES
 S A2 COLUMN 0D 2.0-3.5 2.0-3.5 % ACETAMINOPHEN 0D 5-25 5-25 > 150 ug/ml > 50 ug/ml 12 hour post ingestion > 100 ug/ml 8 hour post ingestion > 200 ug/ml 4 hour post ingestion ACTIVATED PROTEIN C RESIS 0D
S A2 COLUMN 0D 2.0-3.5 2.0-3.5 % ACETAMINOPHEN 0D 5-25 5-25 > 150 ug/ml > 50 ug/ml 12 hour post ingestion > 100 ug/ml 8 hour post ingestion > 200 ug/ml 4 hour post ingestion ACTIVATED PROTEIN C RESIS 0D
Thyroid Care & Diagnostic Centre
 Thyroid Care & Diagnostic Centre Laboratory Test List DEPARTMENT OF CLINICAL PATHOLOGY BLOOD : CBC HCT/ PCV Hb, TC, DC & ESR MCV CE MCH PC MCHC MP Reticulocyte count PBF LE Cell Hb-Electrophoresis Protin
Thyroid Care & Diagnostic Centre Laboratory Test List DEPARTMENT OF CLINICAL PATHOLOGY BLOOD : CBC HCT/ PCV Hb, TC, DC & ESR MCV CE MCH PC MCHC MP Reticulocyte count PBF LE Cell Hb-Electrophoresis Protin
HEALTH SCREEN CARE. VIGNE Healthcare provides a comprehensive Health Screening Package of EXCLUSIVE HEALTH SCREENING EXPERIENCE
 HEALTH SCREEN CARE VIGNE Healthcare provides a comprehensive Health Screening Package of Silver Gold Platinum Cancer EXCLUSIVE HEALTH SCREENING EXPERIENCE Meet & Greet Medical Review with Doctor Appointment
HEALTH SCREEN CARE VIGNE Healthcare provides a comprehensive Health Screening Package of Silver Gold Platinum Cancer EXCLUSIVE HEALTH SCREENING EXPERIENCE Meet & Greet Medical Review with Doctor Appointment
ROTUNDA HOSPITAL DEPARTMENT OF LABORATORY MEDICINE
 This active test table informs the user of Biochemistry tests available in house. s referred to other sites are recorded in the Referred Table. Issue date: 4 TH April 2016 Contact Phone Number ext.1345/2522
This active test table informs the user of Biochemistry tests available in house. s referred to other sites are recorded in the Referred Table. Issue date: 4 TH April 2016 Contact Phone Number ext.1345/2522
Anti-FceR (CU Index) Antibody
 Anti-FceR (CU Index) Antibody Order Name: ANTI-FCER Test Number: 5587500 REV DATE:5/31/2007 Anti-FceR (CU Index) Antibody CU Index Preferred 2 ml (1mL) Serum Clot Activator SST (Red/Gray or Tiger Top)
Anti-FceR (CU Index) Antibody Order Name: ANTI-FCER Test Number: 5587500 REV DATE:5/31/2007 Anti-FceR (CU Index) Antibody CU Index Preferred 2 ml (1mL) Serum Clot Activator SST (Red/Gray or Tiger Top)
Sage Program Reimbursement Rates (Effective Jan 1, 2018 through Dec 31, 2018)
 Sage Program Reimbursement Rates Code Description of Service Allowable Rates New Patient 99201 History, exam, straight forward decision-making; 10 $44.47 99202 Expanded history; exam, straightforward decision-making;
Sage Program Reimbursement Rates Code Description of Service Allowable Rates New Patient 99201 History, exam, straight forward decision-making; 10 $44.47 99202 Expanded history; exam, straightforward decision-making;
GRADING CRITERIA for CMS Regulated Analytes
 CLIA '88 AND GRADING The Clinical Laboratory Improvement Amendments of 1988 (CLIA '88) were established by the federal government (CMS) to regulate clinical laboratories and proficiency test providers
CLIA '88 AND GRADING The Clinical Laboratory Improvement Amendments of 1988 (CLIA '88) were established by the federal government (CMS) to regulate clinical laboratories and proficiency test providers
PRICE LIST RELIABLE ACCURATE COST EFFECTIVE. American Technology Made in India For India. W.E.F April 2018
 PRICE LIST W.E.F April 2018 RELIABLE ACCURATE COST EFFECTIVE American Technology Made in India For India +91-44-22541131 www.athenesedx.com info@athenesedx.com Code OnSite Rapid Tests R0061C R0062C R0062C
PRICE LIST W.E.F April 2018 RELIABLE ACCURATE COST EFFECTIVE American Technology Made in India For India +91-44-22541131 www.athenesedx.com info@athenesedx.com Code OnSite Rapid Tests R0061C R0062C R0062C
Reflex Test Protocols
 Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
SIMPLIFYING DIAGNOSTICS IMPROVING GLOBAL HEALTH
 DIAGNOSTICS CATALOG SIMPLIFYING DIAGNOSTICS IMPROVING GLOBAL HEALTH What s Inside About CTK... 2 Product Lines... 3 Blood Borne... 4 Tropical/Parasitic... 6 Hepatitis... 8 Respiratory... 10 Gastrointestinal...
DIAGNOSTICS CATALOG SIMPLIFYING DIAGNOSTICS IMPROVING GLOBAL HEALTH What s Inside About CTK... 2 Product Lines... 3 Blood Borne... 4 Tropical/Parasitic... 6 Hepatitis... 8 Respiratory... 10 Gastrointestinal...
Single donator specimens as advantage in external quality control assessments of infectious diseases
 Single donator specimens as advantage in external quality control assessments of infectious diseases MD, PhD Maija Lappalainen and MD Jukka Suni HUSLAB, Department of Virology Labquality Days 10.2.2007
Single donator specimens as advantage in external quality control assessments of infectious diseases MD, PhD Maija Lappalainen and MD Jukka Suni HUSLAB, Department of Virology Labquality Days 10.2.2007
Test Result Reference Range Flag
 Date of Last Result Test Result Reference Range Flag Dec 07, 2016 25-Hydroxy Vitamin D Total 53 ng/ml 30-100 ng/ml Activated Partial Thromboplast Time Alanine Aminotransferase (ALT/SGPT) 25 sec 24-35 sec
Date of Last Result Test Result Reference Range Flag Dec 07, 2016 25-Hydroxy Vitamin D Total 53 ng/ml 30-100 ng/ml Activated Partial Thromboplast Time Alanine Aminotransferase (ALT/SGPT) 25 sec 24-35 sec
Aerobic Wound Culture and Stain
 Aerobic Wound Culture and Stain Order Name: C WOUN RTS Test Number: 6000153 Revision Date: 03/27/2014 Aerobic Wound Culture and Stain Culture Preferred 1 ml Tissue Sterile Screwtop Container Room Temperature
Aerobic Wound Culture and Stain Order Name: C WOUN RTS Test Number: 6000153 Revision Date: 03/27/2014 Aerobic Wound Culture and Stain Culture Preferred 1 ml Tissue Sterile Screwtop Container Room Temperature
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 Schedule of Accreditation 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK National Infection Service, Public Health England Colindale 61 Colindale Avenue London NW9 5EQ Contact: Dr Sanjiv
Schedule of Accreditation 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK National Infection Service, Public Health England Colindale 61 Colindale Avenue London NW9 5EQ Contact: Dr Sanjiv
Patient Information Specimen Information Client Information. Specimen: EN255254W. Requisition:
 Patient Information Specimen Information Client Information MAIL000 Requisition: 0014895 DIRECT TO PATIENT- WEST HILLS Lab Ref #: PRO76199 8401 FALLBROOK AVE WEST HILLS, CA 91304-3226 Phone: 530.347.6380
Patient Information Specimen Information Client Information MAIL000 Requisition: 0014895 DIRECT TO PATIENT- WEST HILLS Lab Ref #: PRO76199 8401 FALLBROOK AVE WEST HILLS, CA 91304-3226 Phone: 530.347.6380
ACCREDITATION DOCUMENT
 Accreditation No: Awarded to Dow Diagnostic Reference and Research Laboratory (DDRRL), Dow University of Health Sciences Suparco Road Gulzar e Hijri KDA Scheme-35, Karachi, Pakistan. The scope of accreditation
Accreditation No: Awarded to Dow Diagnostic Reference and Research Laboratory (DDRRL), Dow University of Health Sciences Suparco Road Gulzar e Hijri KDA Scheme-35, Karachi, Pakistan. The scope of accreditation
Memo No: Date: 25-Nov NEW Serology and Virology Testing
 Memo No: 2014-61 Date: 25-Nov-2014 www.hicl.on.ca Re: NEW Serology and Virology Testing In Common Laboratories has been working hard to expand our test menu. We are pleased to announce the availability
Memo No: 2014-61 Date: 25-Nov-2014 www.hicl.on.ca Re: NEW Serology and Virology Testing In Common Laboratories has been working hard to expand our test menu. We are pleased to announce the availability
BID ANNUAL CONTRACT FOR LAB TESTING
 1 ACID FAST BACILLISTAIN 100 10.48 1,048.00 8.00 800.00 2 AFP, TUMOR (CHIRON) 10 9.04 90.40 7.00 70.00 3 AMYLASE 160 3.13 500.80 2.75 440.00 4 BASIC METAB PNL 1,800 1.44 2,592.00 1.95 3,510.00 5 BZV IGGAB
1 ACID FAST BACILLISTAIN 100 10.48 1,048.00 8.00 800.00 2 AFP, TUMOR (CHIRON) 10 9.04 90.40 7.00 70.00 3 AMYLASE 160 3.13 500.80 2.75 440.00 4 BASIC METAB PNL 1,800 1.44 2,592.00 1.95 3,510.00 5 BZV IGGAB
December Metanephrines, Fractionated, Free, Plasma Plasminogen Activator Inhibitor Type 1 Stool Cultures and Sensitivities
 University of Washington December 2012 TEST change 1,25 Dihydroxy Vitamin D Frequency AFB Blood Cultures Alcohol Screen Anti Deaminated Gliadin, IgG Anti Gliadin Package Anti Mullerian Hormone Package
University of Washington December 2012 TEST change 1,25 Dihydroxy Vitamin D Frequency AFB Blood Cultures Alcohol Screen Anti Deaminated Gliadin, IgG Anti Gliadin Package Anti Mullerian Hormone Package
Viral hepatitis. Supervised by: Dr.Gaith. presented by: Shaima a & Anas & Ala a
 Viral hepatitis Supervised by: Dr.Gaith presented by: Shaima a & Anas & Ala a Etiology Common: Hepatitis A Hepatitis B Hepatitis C Hepatitis D Hepatitis E Less common: Cytomegalovirus EBV Rare: Herpes
Viral hepatitis Supervised by: Dr.Gaith presented by: Shaima a & Anas & Ala a Etiology Common: Hepatitis A Hepatitis B Hepatitis C Hepatitis D Hepatitis E Less common: Cytomegalovirus EBV Rare: Herpes
Test Definition: FMCPS Meningoencephalitis Comprehensive Panel (Serum)
 Reporting Title: Meningoencephalitis Comp Panel, S Performing Location: Focus Diagnostics, Specimen Requirements: Draw blood in a plain, red-top tube(s). (Serum gel tube is acceptable.) Spin down and send
Reporting Title: Meningoencephalitis Comp Panel, S Performing Location: Focus Diagnostics, Specimen Requirements: Draw blood in a plain, red-top tube(s). (Serum gel tube is acceptable.) Spin down and send
Directory of Services
 Directory of Services SLDOS Rev. 6/2013 TABLE OF CONTENTS Section 1- Introduction Section 2- General Test Listing Section 3- Microbiology Testing Services Section 4- Peritoneal Dialysis Testing Services
Directory of Services SLDOS Rev. 6/2013 TABLE OF CONTENTS Section 1- Introduction Section 2- General Test Listing Section 3- Microbiology Testing Services Section 4- Peritoneal Dialysis Testing Services
PROFICIENCY TESTING. Clinical Laboratory Improvement Amendments (CLIA) DOs and DON Ts. Brochure # 8
 Clinical Laboratory Improvement Amendments (CLIA) Brochure # 8 PROFICIENCY TESTING DOs and DON Ts NOTE: Congress passed the Clinical Laboratory Improvement Amendments (CLIA) in 1988 establishing quality
Clinical Laboratory Improvement Amendments (CLIA) Brochure # 8 PROFICIENCY TESTING DOs and DON Ts NOTE: Congress passed the Clinical Laboratory Improvement Amendments (CLIA) in 1988 establishing quality
Carrier state and chronic infection state. At-risk populations
 John T. Stutts, MD, MPH University of Louisville School of Medicine Department of Pediatrics Division of Pediatric Gastroenterology, Hepatology and Nutrition Carrier state and chronic infection state -
John T. Stutts, MD, MPH University of Louisville School of Medicine Department of Pediatrics Division of Pediatric Gastroenterology, Hepatology and Nutrition Carrier state and chronic infection state -
Test Bulletin. Specimen Type. Preferred Volume. PCA3 PCA3TM Urine 2.5 ml KIT Frozen. URCTFR Urine 2.5 ml Urine Refrigerated
 Bulletin New Referral ing s Effective March January 1, 2017 2014 Description Current New Specimen Type Preferred Volume Tube Type Temperature to Transport Notes/Comments Prostate Cancer Biomarker PCA3
Bulletin New Referral ing s Effective March January 1, 2017 2014 Description Current New Specimen Type Preferred Volume Tube Type Temperature to Transport Notes/Comments Prostate Cancer Biomarker PCA3
Fullerton Healthcare Screening Centres
 Fullerton Healthcare Screening Centres Fullerton Healthcare Screening Centre @ Ngee Ann City The Penthouse, #26-02 Ngee Ann City Tower B, 391B Orchard Road, Singapore 238874 Operating hours: Monday - Friday
Fullerton Healthcare Screening Centres Fullerton Healthcare Screening Centre @ Ngee Ann City The Penthouse, #26-02 Ngee Ann City Tower B, 391B Orchard Road, Singapore 238874 Operating hours: Monday - Friday
Activated Partial Thromboplastin Time (aptt)
 Activated Partial Thromboplastin Time (aptt) Order Name: PTT Test Number: 1500050 REV DATE:12/26/2008 Activated Partial Thromboplastin Time (aptt) CLOT Preferred 2.7 ml Whole Blood Sodium Citrate 3.2%
Activated Partial Thromboplastin Time (aptt) Order Name: PTT Test Number: 1500050 REV DATE:12/26/2008 Activated Partial Thromboplastin Time (aptt) CLOT Preferred 2.7 ml Whole Blood Sodium Citrate 3.2%
