Anatomically Sound, Simplified Approach to Repair of "Complete" Atrioventricular Septal Defect
|
|
|
- Meghan Cooper
- 5 years ago
- Views:
Transcription
1 Anatomically Sound, Simplified Approach to Repair of "Complete" Atrioventricular Septal Defect Benson R. Wilcox, MD, David R. Jones, MD, Elman G. Frantz, MD, Lela W. Brink, MD, G. William Henry, MD, Michael R. Mill, MD, and Robert H. Anderson, MD Departments of Surgery and Pediatrics, University of North Carolina School of Medicine, Chapel Hill, North Carolina Background. There are few congenital anomalies of the heart that have benefited more from thorough anatomic analysis than the complex anomaly known as atrioventricular septal defect in the setting of common atrioventricular junction. Recent advances in understanding the anatomy of this lesion have led to alternative methods of repairing these defects. Methods. The medical records of 21 consecutive patients undergoing repair of complete atrioventricular septal defect have been reviewed. Nine of these patients had a standard one- or two-patch repair, and 12 had direct closure of the ventricular element of the defect. Results. Direct closure resulted in significantly shorter pump and cross-clamp times. Follow-up for an average of 34 months suggests that when direct closure can be performed, the results are comparable with those of the more standard technique. Conclusions. Our initial success with this approach is encouraging; however, longer follow-up is required to establish whether it will be broadly applicable. (Ann Thorac Surg 1997;64:487-94) 1997 by The Society of Thoracic Surgeons here are few congenital abnormalities of the heart T that have benefited more from thorough anatomic analysis than the complex anomaly known as atrioventricular septal defect. In the early years of open heart surgery, such defects were often thought of as simply another form of atrial or ventricular septal defects in the setting of common atrioventricular junction [1]. These hearts are now understood to be much more complicated in their deformation. Perhaps most important from a surgical point of view was the realization that the atrioventricular valves in such hearts are unique structures, not merely distortions of the usual mitral or tricuspid valves [2]. Becker and Anderson [3] and the European school of morphologists [41 have subsequently clone much to illuminate the various facets of this malformation. With the benefit of this clearer understanding, surgeons have been increasingly aggressive in attacking this problem at a time in the life of the patient that promises a better long-term outcome [5]. Faced with the challenge of correcting this complex problem in smaller patients, surgeons must take advantage of any options that may facilitate such an undertaking. It is the purpose of this article to present one such option that may be useful in the surgical approach to selected patients with a complete atrioventricular septal defect. Presented at the Thirty-third Annual Meeting of The Society of Thoracic Surgeons, San Diego, CA, Feb 3-5, Address reprint requests to Dr Wilcox, Division of Cardiothoracic Surgery, University of North Carolina at Chapel Hill, 108 Burnett-Womack Building, CB 7065, Chapel Hill, NC Material and Methods Patient Population A retrospective analysis was performed of 21 consecutive patients who had undergone repair of complete atrioventricular septal defect at University of North Carolina Hospitals between January 1992 and July 1995 (Table 1). All of the patients underwent operation performed by one of us (B.R.W.). The average age at operation was 7.8 months, with a range of 2 to 48 months. All except 3 patients, aged 12, 14, and 48 months, were less than I year of age. Seventeen patients had Down's syndrome. Operative Technique In 9 of these patients, a standard single- or double-patch repair was performed. Pericardium alone was used in the two single-patch repairs, and it was used in combination with Dacron in the seven double-patch operations. In the remaining 12 patients, direct closure of the ventricular component was accompanied by pericardial patch closure of the atrial component. In all patients except 1, the atrioventricular valves were altered by valvoplasty, annuloplasty, or both to effect as functional an anatomic repair as possible. Direct suture closure of the ventricular component (Fig 1A) was effected by placing 5-0 polypropylene mattress sutures with Dacron pledgets into the right ventricular This article has been selected for the open discussion forum on the STS Web site: by The Society of Thoracic Surgeons /97/$17.00 Published by Elsevier Science Inc PII S (97)
2 488 WILCOX ET AL Ann Thorac Surg REPAIR OF "COMPLETE" AVSD 1997;64: Table 1. Retrospective Analysis of 21 Consecutive Patients Undergoing Repair of Complete Atrioventricular Septal Defect (January 1992-July 1995) Patient Age Date of LV S/D PA S/D PA Mean PVR Preop No. (too) Operation Operation (ram Hg) (turn Hg) (mm Hg) (Wood units) LAWR 1 6 Direct 3/5/92 90/17 90/ Mild 2 5 Single 7/28/92 80/7 55/ NA 3 a 12 Double 9/22/92 100/8 65/ Mod 4 3 Direct 10/1[92 100/5 78/10 33 NA Mild 5 2 Direct 11[5/ / None 6 6 Single 11/30/92 88/5 82/ Mod 7 48 Direct 12/2/92 115/10 22/11 14 NA Mild 8 2 Double 3/28/93 90/10 80/ None Double 9/20/93 102/10 102/ Mild Double 10/4/93 90/8 75/ NA 11 6 Double 4/5/94 95/8 77/ Mild Direct 5/18/94 88/10 92/ Mild 13 6 Direct 6/8/94 90/8 22/ Mod Double 8/23/94 75/10 68/ NA Direct 12/22/94 85/13 81/ Mild Direct 12/29/94 73/9 73/ None 17 7 Direct 1/24/95 70/8 32/ None Direct 3/23/95 68/9 63/ None 19 6 Direct 4/18/95 NA NA NA NA NA 20 6 Direct 5/23/95 95/14 85/ Mild 21 a 14 Double 7/26/95 80/10 NA NA NA Mod a This patient had atrioventricular septal defect and tetralogy of Fallot. LAWR = left atrioventricular valve regurgitation on echocardiogram; LV = left ventricle; pulmonary artery; PVR = pulmonary vascular resistance; S/D = systole/diastole. Mod = moderate; NA = not available; PA = aspect of the muscular septum (Fig 1B). These sutures were placed well below the crest of the septum to avoid damage to the exposed conduction tissue. The sutures were then passed through the superior and inferior bridging leaflets at an appropriate point, demarcating the boundary between right and left atrial outlets. When appropriate, these sutures were placed toward the right side of the valve tissue, producing a more generous left atrioventricular valve. When possible, 7-0 stay sutures were placed in the zone of apposition, or "cleft," between the bridging leaflets before septal suture placement, in anticipation of later repair. Usually, to enhance visualization, particularly at the extremes of the septal crest beneath the bridging leaflets, these cleft sutures were not tied until after the septal sutures had been placed. After the septal sutures were passed through the leaflets, the left atrioventricular valve was almost invariably (20/21) repaired using 7-0 polypropylene on the cleft and a pledgeted 5-0 polypropylene annuloplasty suture at the commissure between the left mural leaflet and the superior bridging leaves. The valve was then sized with a Hegar dilator to assure an adequate orifice. The septal sutures, having been passed through the bridging leaflets, were then used to anchor the leading edge of a pericardial patch. Tying these sutures obliterated the ventricular septal component (Fig 1C). The atrioventricular valves were tested by gentle instillation of iced saline solution into each ventricle, and the repair of each valve was adjusted if necessary. A continuous suture was used in the remaining part of the patch to close the atrial component, usually but not always leaving the coronary sinus on the right side. A left atrial pressure line was left in place, and in cases with persistent pulmonary hypertension, pulmonary arterial pressure was monitored. Although it is our present practice to evaluate all such patients with intraoperative transesophageal echocardiography, none of the patients in this report was so examined. Patient Follow-up All patients were seen in follow-up by a cardiologist, and their findings were reviewed by one of us (E.G.F.). The patients and their medical records were examined for postoperative complications, and to assess atrioventricular and arterial valvar function. Postoperative echocardiograms were available in 19 of the 20 patients surviving the immediate postoperative period. In addition, in December 1996 the parents of 18 of the 19 patients alive at that time were contacted by telephone to assess the patients' functional status. The two groups, 11 survivors of direct and 9 of conventional repair, were contrasted in terms of pump time and cross-clamp time during their repair. Because 2 of the patients having standard repair of their atrioventricular septal defect also had tetralogy of Fallot, it could be argued that these more complex repairs caused the times
3 Ann Thorac Surg WILCOX ET AL ;64: REPAIR OF "COMPLETE" AVSD to be different. Thus, the 11 direct repairs were also compared with the 7 more straightforward standard repairs. The results were analyzed using an unpaired t test. A C Fig 1. (A) A surgeon's view through the right atrium with the patient's head to the left and feet to the right. (B) Pledgeted sutures are anchored in the right ventricular apex of the septum and passed through the bridging leaflets. (C) The same sutures passed through the pericardial patch used to close the atrial component. Tying these sutures obliterates the ventricular component. Results The average pump time for the 11 patients who underwent direct suture repair of the ventricular component was minutes. For the 9 patients having singleor double-patch closure, the pump time averaged 138 _+ 8 minutes, a significantly longer time (p < 0.001). The cross-clamp times were also significantly different: in those patients having direct repair, the aorta was clamped an average of 53 ± 3 minutes compared with minutes for those having the more complicated repair (p < ). Excluding the 2 patients with tetralogy of Fallot gave an average pump time of 139 _+ 11 minutes and a cross-clamp time of 75 _+ 4 minutes, still significant differences (p < 0.003). It should be noted that because of the extended effort to manage the severe pulmonary hypertension in the single patient dying during the operation--patient 20, a 6-month-old child with Down's syndrome and pulmonary vascular disease (pulmonary vascular resistance = 11 Wood units)--the values for this patient were not included in the calculations above. They relate less to operative technique than to the patient's underlying associated illness.* This patient had a direct repair of her defect. Intraoperative hemodynamic assessment and visual inspection attested to the adequacy of the repair and suggested that unresponsive pulmonary vascular disease was the cause of death. Postmortem examination demonstrated pulmonary vascular disease and also suggested that the repair was intact without gross evidence of arterial or atrioventricular valvar malfunction. Another patient (patient 9) died 8 months after doublepatch closure of his defect. At the time of his operation, he was known to have systemic pressures in his right side with a calculated pulmonary vascular resistance of 8 Wood units. After discharge from the hospital, he failed to thrive. Repeated examinations showed his repair to be intact, but there were continuing signs of increasing pulmonary vascular disease. He was admitted to the hospital 8 months after the operation anticipating repeat cardiac catheterization. He died before this could be accomplished. Postmortem examination demonstrated pulmonary vascular disease and an intact intracardiac repair. One patient (patient 14) has required reoperation because of stenosis of the left atrioventricular valve. At the time of this patient's double-patch closure, it was recognized that the left atrioventricular valve had a double orifice. After her discharge from the hospital after that initial operation, it became increasingly apparent that the degree of stenosis combined with insufficiency would not be tolerated. Twelve months after her original operation, she successfully underwent replacement of the left atrio- * Including this patient does not alter the statistical analysis significantly.
4 490 WILCOX ET AL Ann Thorac Surg REPAIR OF "COMPLETE" AVSD 1997;64: ventricular valve. She is presently doing well 18 months after valvar replacement. One patient (patient 15) required recatheterization 18 months after her direct repair because of failure to thrive. She was found to have progressive vascular disease (pulmonary vascular resistance = 15 Wood units, compared with 4 Wood units preoperatively) and mild atrioventricular valvar regurgitation. No shunt was demonstrable by oxymetry. Two years after her operation, she remains on a regimen of digitalis and diuretics, but she is physically active. Although the remaining patients were not without some problems postoperatively, none of the problems were attributable to the particular intracardiac repair technique. All patients have been seen in follow-up, and 19 of the 20 operative survivors had postoperative echocardiographic examination (Table 2). Postoperative echocardiography showed that 4 of 11 patients who underwent direct closure had no left-sided regurgitation, and regurgitation in the remainder was judged mild to moderate. Of the 8 patients with postoperative echocardiograms in the group undergoing patch closure, 2 showed no regurgitation and 6 showed mild to moderate regurgitation. No patient in either group was found to have severe regurgitation or a significant leftto-right shunt. In addition, there was no evidence of left ventricular outflow tract obstruction seen in these patients. Telephone follow-up in December 1996 (17 to 57 months postoperative) has been possible in 18 of the 19 presently surviving patients. The patient who could not be contacted was seen 26 months after his direct repair and was fully active and receiving no medications. Of the 18 patients followed up in December 1996, 10 had undergone direct closure whereas 8 had their ventricular defects closed with a patch. Of the former group, all except 1 (patient 15, described above) are fully active. One other patient in the group undergoing direct closure is receiving cardiac medications. He is fully active, but requires digitalis. The 8 remaining patients undergoing direct closure are asymptomatic, in sinus rhythm, and receiving no cardiac medications. Comment It is general knowledge that, on occasion, when two atrioventricular valve orifices are present--so-called partial atrioventricular septal defect--there may be additional interventricular communications beneath the Table 2. Postoperative Follow-up Data (1.5 to 3.5 years) LAWR No. of Limited Cardiac (Mild to Operation Patients Activi~ Medications Moderate) Standard repair 8 ~ 1/8 1/8 6/8 Direct closure 11 b 1/11 2/11 6/11 a One late death, b One operative death. LAVVR = left atrioventricular valve regurgitation on echocardiogram. Normal AV septation ~ t/outlet ;;.';' AV septal defect Ratio = 1.0 Inlet/Outlet Ratio = 0.77 Fig 2. Diagram of the ventm'cular septum, in surgical orientation, showing the inlet, outlet, and scoop from a left ventricular point of view. bridging leaflets. In most instances, these are intercordal in nature and can be closed without incorporating a patch for the ventricular component [6]. On the other hand, when there is a single orifice and free communication beneath the bridging leaflets, prudence has dictated that a patch is necessary to close the ventricular component of the defect, the basis for such reasoning being the possibility of a greater deficiency in the ventricular septum [7-9]. As reported in these studies, measurements were taken from the apex of the left ventricle to (1) the crux of the heart (inlet), (2) the deepest part of the "scoop," and (3) the aortic valve (outlet) (Fig. 2). The measurements of the inlet and scoop were divided by the outlet measurement and expressed as ratios. The authors of these studies [6, 7] then compared such measurements in hearts with a single atrioventricular orifice ("complete" atrioventricular septal defect) with those in hearts with two orifices ("partial" or primum defects). There was no difference in the inlet/outlet ratios between the two groups, whereas the two sets of hearts differed significantly in the ratio of the scoop to outlet measurements [6,
5 Ann Thorac Surg WILCOX ET AL ;64: REPAIR OF "COMPLETE" AVSD A Fig 3. (A) A deep septal scoop seen in an anatomic specimen from the right ventricular point of view. (B) Similar view showing a "complete" atrioventricular septal defect where the scoop is less pronounced. B 7]. Thus, there is a definite likelihood that in hearts with "complete" atrioventricular septal defects, the ventricular septum may be more deficient than in hearts with "partial" defects. Ebels and colleagues [8], however, made an additional important observation. Although the mean values were quite different, rather wide variability was found among these specimens. In as many as 30% to 40% of hearts with single orifice, the scoop/outlet ratio was such (0.65 or greater) that very little "scoop" was apparent. That is, the nadir of the septal crest was approximately on a level with the inlet portion at the crux. These selected specimens, therefore, would not differ significantly from hearts with separate orifices in terms of ventricular septal architecture. Thus, one might reason, it is not always necessary to fill this potential deficiency with new material, namely, a ventricular patch. In some instances, one should be able to attach the atrioventricular valvar leaflets to the septum in such a way that they close the ventricular communication and yet allow satisfactory function of the reconstructed atrioventricular valves and do not cause arterial valve obstruction. With increasing experience, we have been more aggressive in applying this technique in the treatment of "complete" atrioventricular septal defect. Over a 3.5-year period beginning January 1, 1992, 21 patients underwent open repair of such defects at this hospital. We were able to apply the direct closure technique in 12 of these patients. Nine patients were judged unsuitable for direct closure. Two of these had tetralogy of Fallot as an accompanying problem, and 1 patient in this group had double orifices in the left atrioventricular valve. It was believed that a standard repair would be more suitable under these circumstances. All 3 of these patients are doing well at present, although the child with double orifice required replacement of the left atrioventricular valve 12 months after her original operation. Presently, 18 months after valvar replacement, she is leading an active life, and her only medication is maintenance warfarin. In the remaining 6 patients judged unsuitable for direct closure, it was believed at the time of operation that the space beneath the bridging leaflets was so large that it could not be safely closed without a patch. Important intraoperative considerations in making this decision included the tension required to bring the valve to the septum, with the accompanying concern that the sutures might pull through the muscle of the septum. In addition, if too great a space existed, then distortion of the atrioventricular valve could lead to an unacceptable level of regurgitation postoperatively. The decision to use a patch in these individuals has been modified as experience has accumulated, for we find ourselves closing larger defects by direct suture as our confidence in this procedure grows. Selecting which defects will lend themselves to direct closure remains a difficult judgment call. The preoperative echocardiogram can be useful, but it may exaggerate the size of the defect. At this time, the best predictor of the need for a patch is the presence of an exaggerated "scoop" (Fig 3A). That is, the central portion of the defect is more toward the apex than is often found in hearts with less "scooping" (Fig 3B). We continue to search for preoperative findings that will aid in predicting which patients will require a ventricular septal patch. So far we have been unsuccessful.
6 492 WILCOX ET AL Ann Thorac Surg REPAIR OF "COMPLETE" AVSD 1997;64: A C B Fig 4. (A) Operative view through the right atrium with the superior bridging leaflet retracted to show the right ventricular origin of the aortic orifice. (B) Inferior bridging leaflet retracted to show closer proximity of the leaflet to the crest of the septum. (C) Dacron patch closure of the superior part of the ventricular defect. (D) Direct suture closure of the inferior portion of the ventricular component. D Others [10] have reported a correlation between ratio of scoop to outlet septal dimension and the degree of left-axis deviation in the mean frontal QRS axis of the electrocardiogram. In an earlier work, these authors demonstrated that patients with a milder scooping of the ventricular septum and a well-developed atrial septum did not show left axis deviation [11]. Thus, with careful analysis of preoperative electrocardiographic, echocardiographic, and catheterization findings, one may be able to anticipate the degree of scooping that will be encountered. Unfortunately, none of these electrocardiographic parameters--nor indeed the degree of scooping--correlate well with the morphology of the superior bridging leaflet [10]. An example of the variability one may encounter was seen recently in a patient undergoing operation at our hospital for complete atrioventricular septal defect and double-outlet right ventricle. Because of the double outlet, we believed a patch would be necessary safely to correct left ventricular outflow to the aortic valve. At operation, the superior bridging leaflet was found to be riding high above the septal crest with the aorta dis- placed to the right (Fig 4A). Beneath the inferior bridging leaflet, however, only a narrow space connected the ventricles (Fig 4B). Therefore, we were able to patch the superior component (Fig 4C) and close directly the inferior element (Fig 4D). Also, on occasion we have been able to close directly the extreme ends of the defect and patch only the area of severe scooping. Thus, depending on the specific anatomic findings at operation, the technique of direct closure may have applications in part or entirely. Care must be taken not to extend its use beyond the bounds of prudence, particularly in the setting of severe "scooping" of the ventricular septum (see Fig 3A). Attempting to pull the bridging leaflets down to such a distant location will cause undue tension and may result in disruption of the septal closure or cause valvar malfunction. This success with our initial series suggests that this approach to selected patients is worthy of continued consideration. Follow-up evaluation up to 4.5 years (average, 34 months) shows that these patients do well. One patient (patient 15) has progressed to end-stage pulmonary vascular disease, as has 1 patient (patient 9) treated
7 Ann Thorac 5urg WILCOX ET AL ;64: REPAIR OF "COMPLETE" AVSD with the double-patch technique. One other patient (patient 1), although fully active, requires digitalis, probably because of continuing left atrioventricular valvar regurgitation. It is noteworthy that this is the only patient in whom neither a valvoplasty nor an annuloplasty was performed. It is our present practice to do both in virtually all patients undergoing repair of atrioventricular septal defect with common atrioventricular junction. The only exceptions are those patients with small orifices that might be excessively narrowed by annuloplasty. The zone of apposition between the bridging leaflets (the "cleft") is always closed. Certainly the direct method offers economies in time over the alternative approaches (see Table 2). Of course, statistical significance does not necessarily translate into clinical significance. The average savings in pump time of 38 minutes is probably of little consequence, although-- other things being equal--most would opt for the shorter time. The shorter cross-clamp time (average, 24 minutes) does hold promise for better postoperative performance hemodynamically. Although this could not be demonstrated in this small series, intuitively a 30% saving in ischemic time has great appeal. These considerations of time saved notwithstanding, the greater benefit of the direct closure technique is the resultant anatomic advantage. Placement of a single or double patch often necessitates extensive surgical manipulation, particularly of the bridging leaflets of the common atrioventricular valve. Exact sizing of the ventricular patch, or attachment of the divided leaflets to a single patch, can be technically challenging, whereas once one has determined that direct closure is feasible, the technical demands are limited. One way of looking at this is that direct closure of the ventricular component converts the defect into a lesion akin to an "incomplete" or "primum" defect. The results of repair of this type of atrioventricular septal defect with only an atrial component are uniformly good. If one can easily convert the "complete" defect to an "incomplete" configuration, then the resultant operative outcome should be comparable with that reported with the lesser lesion. We continue to be pleased with the results af- forded by this simplified approach to these complicated hearts. Only time and additional experience will tell if the technique is broadly applicable. We are grateful to Maggie Morris for aid with clinical follow-up, John Lockhart for assistance with echocardiographic review, and Betsy L. Mann for editorial assistance in the preparation of the manuscript. References 1. Hallman GL, Cooley DA. Surgical treatment of congenital heart disease. Philadelphia: Lea & Febiger, Carpentier A. Surgical anatomy and management of the mitral component of atrioventricular canal defects. In: Anderson RH, Shinebourne EA, eds. Paediatric cardiology. Edinburgh: Churchill Livingstone, 1977: Becker AE, Anderson RH. Atrioventricular septal defects: what's in a name? J Thorac Cardiovasc Surg 1982;83: Anderson RH, Baker EJ, Ho SY, Rigby ML, Ebels T. The morphology and diagnosis of atrioventricular septal defects. Cardiol Young 1991;1: Williams WH, Guyton RA, Michalik RE, et al. Individualized surgical management of complete atrioventricular canal. J Thorac Cardiovasc Surg 1983;86: Kirklin JW, Barratt-Boyes BG. Cardiac surgery. 2nd ed. New York: Churchill Livingstone, 1993: Anderson RH, Neches WH, Zuberbuhler JR. Scooping of the ventricular septum in atrioventricular septal defect [Letter]. J Thorac Cardiovasc Surg 1988;95: Ebels T, Anderson RH, Devine WA, Debich DE, Penkoske PA, Zuberbuhler JR. Anomalies of the left atrioventricular valve and related ventricular septal morphology in atrioventricular septal defects. J Thorac Cardiovasc Surg 1990;99: Penkoske PA, Neches WH, Anderson RH, Zuberbuhler JR. Further observations on the morphology of atrioventricular septal defects. J Thorac Cardiovasc Surg 1985;90: Suzuki K, Tatsuno K, Mimori S, et al. Relationship between scooping of the ventricular septum, morphology of the inferior bridging leaflet and electrocardiographic findings in atrioventricular septal defect with common valvar orifice. Cardiol Young 1996;6: Suzuki K, Murakami Y, Tatsuno K, et al. Atypical form of atrioventricular septal defect without left axis deviation: relation between morphology and unusual QRS-axis. Br Heart J 1993;70: DISCUSSION DR TJARK EBELS (Groningen, Netherlands): Doctor Wilcox, I enjoyed your paper. Please allow me a couple of questions. You said it was difficult to predict with echocardiography whether this technique is applicable or not. I was wondering whether the electrocardiogram could be of any help, because it has been shown that children with a more shallow scoop have less left-axis deviation on their electrocardiograms. And the second question is, did you see any subsequent increased flow velocity in the left ventricular outflow tract, indicative of obstruction? DR WILCOX: Thank you. Doctor Ebels has, of course, contributed much to our understanding of atrioventricular septal defect. You are right about the potential help from an electrocardiogram. And as I understand those reports dealing with that, it does help with the degree of scooping. But there is no correlation between that and the relationship of the superior bridging leaflet to the septal crest. We did take a quick look back at this to see whether it would be of any help to us in retrospect, and it really was not very useful. Of course, one of the concerns that we had when we first started doing this was that we would cause some left ventricular outflow obstruction, but we find no evidence of that whatsoever. I am sure I do not totally understand left ventricular outflow obstruction in the setting of this disease. I think it has less to do with the particular repair that is used than with the basic anatomy.
8 494 WILCOX ET AL Ann Thorac Surg REPAIR OF "COMPLETE" AVSD 1997;64: As you know, there is a wide variety of ways in which this disease presents. Doctor Willis Williams told me of a favorite quote of Dr Kirklin's on this subject. He reviewed 77 cases.., l believe I am getting this right... and found that they fit very neatly into 75 categories. So I think it has more to do with the underlying anatomy than it does with the repair technique. We have looked for it. We have not found it. DR EBELS: So if you do not just eyeball it during the operation, is there any rule of thumb to tell us when you do it and when you do not? DR WILCOX: I do not have an easy one. Obviously, in patients with associated lesions, such as tetralogy of Fallot or doubleoutlet right ventricle, we have been reluctant to apply this technique because of the magnitude of the septal defect. Recently, in a patient with a double-outlet right ventricle, we thought that we needed to put in a patch to avoid outflow obstruction, so we sewed a patch around the aortic orifice and then along the superior crest of the septum. Interestingly enough, though, the inferior bridging leaflet was relatively close to the crest. The scooping was principally beneath the superior leaf, so we were able to simply suture the inferior leaf to the septum. We have had 1 other patient recently in whom we have been able to directly close the extremities of the ventricular septal defect, and then when we got to the scoop, we put in a smaller patch. I think right now the best we can do is look at it and estimate how well it is going to work. Obviously, if you see tension when you pull the bridging leaflets down, then you probably ought to undo it and put in a patch. DR JOSEPH J. AMATO (Chicago, IL): I congratulate Dr Wilcox and his co-authors on a very ingenious technique. I have three questions to ask them: 1. I noted you have "complete" in quotation marks and wonder whether these lesions are in reality a transitional form of the atrioventricular septal defect and not a complete atrioventricular septal defect. It becomes a matter of semantics as to whether one is repairing a defect that has a small or larger ventricular component. If indeed the ventricular component is small, I would call it a transitional form. 2. Someone has already asked whether there was a formula for calculating the ventricular septal defect. I am concerned that during cardioplegia one cannot estimate the amount of tension necessary to pull the ventricular septal defect together. Do you have any solution to this problem? 3. Although I was taught never to close ventricular septal defects primarily, it would follow that if you had a formula for closing atrioventricular septal defects primarily that you could do so with simple ventricular septal defects. Finally, I believe you are in effect discussing the closure of the small ventricular septal defect component in the transitional form of atrioventricular septal defect. This is simply a matter of defining terms. Thank you for your excellent presentation. DR WILCOX: Thank you, Dr Amato. We define a complete atrioventricular septal defect as having a common atrioventricular junction, a common valve with a single orifice, as opposed to an incomplete or so-called primum, which is usually associated with having two orifices--that is, a tongue of tissue joining the superior and inferior bridging leaflets, dividing it into two orifices. Usually in the latter instance there is no ventricular septal defect. I do not have any magic number in terms of which will work and which will not, but I think if you start looking at it, you will get some sense of when it is appropriate to use this technique. The tension is apparent. Just by pulling it together a little bit, you can get a good idea of how much tension there will be. Regarding correlation to the ventricular septal defect repair, occasionally I have used the tricuspid valve to close a small ventricular septal defect, but generally I patch it. DR AMATO: But using the tricuspid valve is not pulling two pieces of ventricles together, and that is what I was wondering. Would you close a small muscular or a small ventricular septal defect by the same methodology? DR WILCOX: Well, we are closing this one by pulling the valve down over the defect. I have done that in ventricular septal defects, and I have closed some ventricular septal defects directly, if that helps. DR PEDRO J. DEL NIDO (Boston, MA): One of the underestimated problems in atrioventricular canal repair has always been tricuspid or right atrioventricular valve regurgitation, particularly in patients with pulmonary hypertension. It seems to me that with this technique, if I understand it correctly, you are actually bringing the leaflets further apart because you are pulling them down toward the crest of the septum, and therefore coaptation of the right atrioventricular valve leaflets may be a problem. Have you looked at that? Is that a real problem or is there still enough tissue to coapt so that tricuspid regurgitation is not an issue? DR WILCOX: In most instances, it has not been an issue. I make an effort to reconstruct the right atrioventricular valve to the point where it looks like a functional valve. Almost regardless of what I do, I find that the anterior leaflet is what closes the right atrioventricular valve. If there is a problem, I find it is usually the same whether I close it directly or with a patch. DR CHRISTO I. TCHERVENKOV (Montreal, Quebec, Canada): I simply rise to support Dr Wilcox's concept. Having trained in Boston and begun my career with the single-patch technique, I was impressed, over the last 10 years, that a number of patients had a relatively small ventricular septal defect component. In these patients, the complexity of the operation was increased by dividing the bridging leaflet and then resuspending it in the single-patch repair. More recently, I have had the experience in several patients with rather small ventricular septal defects with plicating the bridging leaflet onto the crest of the ventricular septal defect, greatly simplifying the repair. I had a patient with scooping of the central portion of the interventricular septum and did what you suggested, a combination of the two techniques: a partial plication at each extremity of the superior and the inferior leaflets, reducing the size of the ventricular septal defect, followed by closure with a smaller central patch. I support your concept using a simplified operation in selected patients with appropriate anatomy. I believe though, that the repair used should be individualized in each patient rather than applying a rigid approach of only one type of repair to all patients. DR WILCOX: Thank you very much.
Techniques for repair of complete atrioventricular septal
 No Ventricular Septal Defect Patch Atrioventricular Septal Defect Repair Carl L. Backer, MD *, Osama Eltayeb, MD *, Michael C. Mongé, MD *, and John M. Costello, MD For the past 10 years, our center has
No Ventricular Septal Defect Patch Atrioventricular Septal Defect Repair Carl L. Backer, MD *, Osama Eltayeb, MD *, Michael C. Mongé, MD *, and John M. Costello, MD For the past 10 years, our center has
Anatomy of Atrioventricular Septal Defect (AVSD)
 Surgical challenges in atrio-ventricular septal defect in grown-up congenital heart disease Anatomy of Atrioventricular Septal Defect (AVSD) S. Yen Ho Professor of Cardiac Morphology Royal Brompton and
Surgical challenges in atrio-ventricular septal defect in grown-up congenital heart disease Anatomy of Atrioventricular Septal Defect (AVSD) S. Yen Ho Professor of Cardiac Morphology Royal Brompton and
Repair of Complete Atrioventricular Septal Defects Single Patch Technique
 Repair of Complete Atrioventricular Septal Defects Single Patch Technique Fred A. Crawford, Jr., MD The first repair of a complete atrioventricular septal defect was performed in 1954 by Lillehei using
Repair of Complete Atrioventricular Septal Defects Single Patch Technique Fred A. Crawford, Jr., MD The first repair of a complete atrioventricular septal defect was performed in 1954 by Lillehei using
Surgical Treatment for Atrioventricular Septal Defect. Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery
 Surgical Treatment for Atrioventricular Septal Defect Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Rastelli classification (Rastelli) Pulmonary artery banding (Muller & Dammann)
Surgical Treatment for Atrioventricular Septal Defect Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Rastelli classification (Rastelli) Pulmonary artery banding (Muller & Dammann)
Tetralogy of Fallot (TOF) with atrioventricular (AV)
 Tetralogy of Fallot with Atrioventricular Canal Defect: Two Patch Repair Sitaram M. Emani, MD, and Pedro J. del Nido, MD Tetralogy of Fallot (TOF) with atrioventricular (AV) canal defect is classified
Tetralogy of Fallot with Atrioventricular Canal Defect: Two Patch Repair Sitaram M. Emani, MD, and Pedro J. del Nido, MD Tetralogy of Fallot (TOF) with atrioventricular (AV) canal defect is classified
after AV Canal Repair: When and How To Intervene
 Left Atrioventricular Valve Regurgitation after AV Canal Repair: When and How To Intervene Thomas L Spray, M.D. Chief, Cardiothoracic Surgery Alice Langdon Warner Endowed Chair The Children s Hospital
Left Atrioventricular Valve Regurgitation after AV Canal Repair: When and How To Intervene Thomas L Spray, M.D. Chief, Cardiothoracic Surgery Alice Langdon Warner Endowed Chair The Children s Hospital
The modified Konno procedure, or subaortic ventriculoplasty,
 Modified Konno Procedure for Left Ventricular Outflow Tract Obstruction David P. Bichell, MD The modified Konno procedure, or subaortic ventriculoplasty, first described by Cooley and Garrett in1986, 1
Modified Konno Procedure for Left Ventricular Outflow Tract Obstruction David P. Bichell, MD The modified Konno procedure, or subaortic ventriculoplasty, first described by Cooley and Garrett in1986, 1
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease
 Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Atrioventricular Canal (Septal) Defects. Norman H Silverman MD. D Sc (Med),FACC, FAHA
 Atrioventricular Canal (Septal) Defects Norman H Silverman MD. D Sc (Med),FACC, FAHA Embryology of the A-V Canal Looping NHS. Formation of the Atrial Septum Embryology of the A-V Canal NHS. Development
Atrioventricular Canal (Septal) Defects Norman H Silverman MD. D Sc (Med),FACC, FAHA Embryology of the A-V Canal Looping NHS. Formation of the Atrial Septum Embryology of the A-V Canal NHS. Development
The Edge-to-Edge Technique f For Barlow's Disease
 The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
An anterior aortoventriculoplasty, known as the Konno-
 The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT
 ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
Tricuspid Valve Repair for Ebstein's Anomaly
 Tricuspid Valve Repair for Ebstein's Anomaly Joseph A. Dearani, MD, and Gordon K. Danielson, MD E bstein's anomaly is a malformation of the tricuspid valve and right ventricle that is characterized by
Tricuspid Valve Repair for Ebstein's Anomaly Joseph A. Dearani, MD, and Gordon K. Danielson, MD E bstein's anomaly is a malformation of the tricuspid valve and right ventricle that is characterized by
HISTORY. Question: What category of heart disease is suggested by this history? CHIEF COMPLAINT: Heart murmur present since early infancy.
 HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
Mitral incompetence after repair of ostium
 Thorax (1965), 20, 40. Mitral incompetence after repair of ostium primum septal defects A. R. C. DOBELL, D. R. MURPHY, G. M. KARN, AND A. MARTINEZ-CARO From the Department of Cardiovascular Surgery, the
Thorax (1965), 20, 40. Mitral incompetence after repair of ostium primum septal defects A. R. C. DOBELL, D. R. MURPHY, G. M. KARN, AND A. MARTINEZ-CARO From the Department of Cardiovascular Surgery, the
The need for right ventricular outflow tract reconstruction
 Polytetrafluoroethylene Bicuspid Pulmonary Valve Implantation James A. Quintessenza, MD The need for right ventricular outflow tract reconstruction and pulmonary valve replacement is increasing for many
Polytetrafluoroethylene Bicuspid Pulmonary Valve Implantation James A. Quintessenza, MD The need for right ventricular outflow tract reconstruction and pulmonary valve replacement is increasing for many
Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016
 1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
Although most patients with Ebstein s anomaly live
 Management of Neonatal Ebstein s Anomaly Christopher J. Knott-Craig, MD, FACS Although most patients with Ebstein s anomaly live through infancy, those who present clinically as neonates are a distinct
Management of Neonatal Ebstein s Anomaly Christopher J. Knott-Craig, MD, FACS Although most patients with Ebstein s anomaly live through infancy, those who present clinically as neonates are a distinct
Ostium primum defects with cleft mitral valve
 Thorax (1965), 20, 405. VIKING OLOV BJORK From the Department of Thoracic Surgery, University Hospital, Uppsala, Sweden Ostium primum defects are common; by 1955, 37 operated cases had been reported by
Thorax (1965), 20, 405. VIKING OLOV BJORK From the Department of Thoracic Surgery, University Hospital, Uppsala, Sweden Ostium primum defects are common; by 1955, 37 operated cases had been reported by
Morphological evaluation of atrioventricular septal defects by magnetic resonance imaging
 138 Br Heart J 1990;64:138-45 Morphological evaluation of atrioventricular septal defects by magnetic resonance imaging Department of Paediatric Cardiology, Guy's Hospital,. London J M Parsons E J Baker
138 Br Heart J 1990;64:138-45 Morphological evaluation of atrioventricular septal defects by magnetic resonance imaging Department of Paediatric Cardiology, Guy's Hospital,. London J M Parsons E J Baker
Eva Maria Delmo Walter Takeshi Komoda Roland Hetzer
 Surgical repair of the congenitally malformed mitral valve leaflets in infants and children Eva Maria Delmo Walter Takeshi Komoda Roland Hetzer Deutsches Herzzentrum Berlin Germany Background and Objective
Surgical repair of the congenitally malformed mitral valve leaflets in infants and children Eva Maria Delmo Walter Takeshi Komoda Roland Hetzer Deutsches Herzzentrum Berlin Germany Background and Objective
Atrial Septal Defects
 Supplementary ACHD Echo Acquisition Protocol for Atrial Septal Defects The following protocol for echo in adult patients with atrial septal defects (ASDs) is a guide for performing a comprehensive assessment
Supplementary ACHD Echo Acquisition Protocol for Atrial Septal Defects The following protocol for echo in adult patients with atrial septal defects (ASDs) is a guide for performing a comprehensive assessment
The stentless bioprosthesis has many salient features that
 Aortic Valve Replacement with the Medtronic Freestyle Xenograft Using the Subcoronary Implantation Technique D. Michael Deeb, MD The stentless bioprosthesis has many salient features that make it an attractive
Aortic Valve Replacement with the Medtronic Freestyle Xenograft Using the Subcoronary Implantation Technique D. Michael Deeb, MD The stentless bioprosthesis has many salient features that make it an attractive
14 Valvular Stenosis
 14 Valvular Stenosis 14-1. Valvular Stenosis unicuspid valve FIGUE 14-1. This photograph shows severe valvular stenosis as it occurs in a newborn. There is a unicuspid, horseshoe-shaped leaflet with a
14 Valvular Stenosis 14-1. Valvular Stenosis unicuspid valve FIGUE 14-1. This photograph shows severe valvular stenosis as it occurs in a newborn. There is a unicuspid, horseshoe-shaped leaflet with a
Heart and Lungs. LUNG Coronal section demonstrates relationship of pulmonary parenchyma to heart and chest wall.
 Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Aortic valve repair is a technique that is gaining popularity
 Aortic Valve Repair in Children, Including Pericardial Patch Reconstruction Aditya K. Kaza, MD,* and John A. Hawkins, MD Aortic valve repair is a technique that is gaining popularity in children because
Aortic Valve Repair in Children, Including Pericardial Patch Reconstruction Aditya K. Kaza, MD,* and John A. Hawkins, MD Aortic valve repair is a technique that is gaining popularity in children because
Surgery For Ebstein Anomaly
 Surgery For Ebstein Anomaly Christian Pizarro, MD Chief, Pediatric Cardiothoracic Surgery Director, Nemours Cardiac Center Alfred I. dupont Hospital for Children Professor of Surgery and Pediatrics Sidney
Surgery For Ebstein Anomaly Christian Pizarro, MD Chief, Pediatric Cardiothoracic Surgery Director, Nemours Cardiac Center Alfred I. dupont Hospital for Children Professor of Surgery and Pediatrics Sidney
Accepted Manuscript. The Left atrioventricular valve: The Achilles Heel of incomplete endocardial cushion defects. Meena Nathan, MD, MPH
 Accepted Manuscript The Left atrioventricular valve: The Achilles Heel of incomplete endocardial cushion defects Meena Nathan, MD, MPH PII: S0022-5223(18)32898-8 DOI: https://doi.org/10.1016/j.jtcvs.2018.10.120
Accepted Manuscript The Left atrioventricular valve: The Achilles Heel of incomplete endocardial cushion defects Meena Nathan, MD, MPH PII: S0022-5223(18)32898-8 DOI: https://doi.org/10.1016/j.jtcvs.2018.10.120
Complete atrioventricular septal defect with tetralogy
 Atrioventricular Septal Defect With Tetralogy of Fallot: Results of Surgical Correction Stacey B. O Blenes, MD, David B. Ross, MD, Maurice A. Nanton, MD, and David A. Murphy, MD Divisions of Cardiovascular
Atrioventricular Septal Defect With Tetralogy of Fallot: Results of Surgical Correction Stacey B. O Blenes, MD, David B. Ross, MD, Maurice A. Nanton, MD, and David A. Murphy, MD Divisions of Cardiovascular
Partial atrioventricular septal defect (AVSD), the socalled
 Surgery for Partial Atrioventricular Septal Defect in the Adult Michael A. Gatzoulis, MD, Sloane Hechter, Gary D. Webb, MD, and William G. Williams, MD The Toronto Congenital Cardiac Centre for Adults,
Surgery for Partial Atrioventricular Septal Defect in the Adult Michael A. Gatzoulis, MD, Sloane Hechter, Gary D. Webb, MD, and William G. Williams, MD The Toronto Congenital Cardiac Centre for Adults,
Giovanni Di Salvo MD, PhD, FESC Second University of Naples Monaldi Hospital
 Giovanni Di Salvo MD, PhD, FESC Second University of Naples Monaldi Hospital VSD is one of the most common congenital cardiac abnormalities in the newborn. It can occur as an isolated finding or in combination
Giovanni Di Salvo MD, PhD, FESC Second University of Naples Monaldi Hospital VSD is one of the most common congenital cardiac abnormalities in the newborn. It can occur as an isolated finding or in combination
AMERICAN ACADEMY OF PEDIATRICS 993 THE NATURAL HISTORY OF CERTAIN CONGENITAL CARDIOVASCULAR MALFORMATIONS. Alexander S. Nadas, M.D.
 AMERICAN ACADEMY OF PEDIATRICS 993 tnicular overload is the major problem and left ventricular failure occurs. Since for many years the importance of hepatomegaly in the diagnosis of cardiac failure has
AMERICAN ACADEMY OF PEDIATRICS 993 tnicular overload is the major problem and left ventricular failure occurs. Since for many years the importance of hepatomegaly in the diagnosis of cardiac failure has
In 1980, Bex and associates 1 first introduced the initial
 Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
Cardiac Catheterization Cases Primary Cardiac Diagnoses Facility 12 month period from to PRIMARY DIAGNOSES (one per patient)
 PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
Yuichi Shiokawa, MD Anton E. Becker, MD, PhD
 THE LEFT VENTRICULAR OUTFLOW TRACT IN ATRIOVENTRICULAR SEPTAL DEFECT REVISITED: SURGICAL CONSIDERATIONS REGARDING PRESERVATION OF AORTIC VALVE INTEGRITY IN THE PERSPECTIVE OF ANATOMIC OBSERVATIONS Yuichi
THE LEFT VENTRICULAR OUTFLOW TRACT IN ATRIOVENTRICULAR SEPTAL DEFECT REVISITED: SURGICAL CONSIDERATIONS REGARDING PRESERVATION OF AORTIC VALVE INTEGRITY IN THE PERSPECTIVE OF ANATOMIC OBSERVATIONS Yuichi
Ebstein s anomaly is defined by a downward displacement
 Repair of Ebstein s Anomaly Sylvain Chauvaud, MD Ebstein s anomaly is a tricuspid valve anomaly associated with poor right ventricular contractility in severe cases. Surgery is indicated in all symptomatic
Repair of Ebstein s Anomaly Sylvain Chauvaud, MD Ebstein s anomaly is a tricuspid valve anomaly associated with poor right ventricular contractility in severe cases. Surgery is indicated in all symptomatic
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions
 Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Congenital Heart Defects
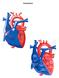 Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Recent technical advances and increasing experience
 Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT
 LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT 10-13 March 2017 Ritz Carlton, Riyadh, Saudi Arabia Zohair AlHalees, MD Consultant, Cardiac Surgery Heart Centre LEFT VENTRICULAR
LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT 10-13 March 2017 Ritz Carlton, Riyadh, Saudi Arabia Zohair AlHalees, MD Consultant, Cardiac Surgery Heart Centre LEFT VENTRICULAR
Replacement of the mitral valve in the presence of
 Mitral Valve Replacement in Patients with Mitral Annulus Abscess Christopher M. Feindel Replacement of the mitral valve in the presence of an abscess of the mitral annulus presents a major challenge to
Mitral Valve Replacement in Patients with Mitral Annulus Abscess Christopher M. Feindel Replacement of the mitral valve in the presence of an abscess of the mitral annulus presents a major challenge to
Atrioventricular valve repair: The limits of operability
 Atrioventricular valve repair: The limits of operability Francis Fynn-Thompson, MD Co-Director, Center for Airway Disorders Surgical Director, Pediatric Mechanical Support Program Surgical Director, Heart
Atrioventricular valve repair: The limits of operability Francis Fynn-Thompson, MD Co-Director, Center for Airway Disorders Surgical Director, Pediatric Mechanical Support Program Surgical Director, Heart
When to close an Atrial Septal Defect (ASD) in adulthood?
 When to close an Atrial Septal Defect (ASD) in adulthood? Philippe ALDEBERT Hôpital de la Timone, CHU Marseille Département de cardiologie pédiatrique et congénitale médico-chirurgical Abbott Incidence
When to close an Atrial Septal Defect (ASD) in adulthood? Philippe ALDEBERT Hôpital de la Timone, CHU Marseille Département de cardiologie pédiatrique et congénitale médico-chirurgical Abbott Incidence
Perioperative Management of DORV Case
 Perioperative Management of DORV Case James P. Spaeth, MD Department of Anesthesia Cincinnati Children s Hospital Medical Center University of Cincinnati Objectives: 1. Discuss considerations regarding
Perioperative Management of DORV Case James P. Spaeth, MD Department of Anesthesia Cincinnati Children s Hospital Medical Center University of Cincinnati Objectives: 1. Discuss considerations regarding
Repair of Congenital Mitral Valve Insufficiency
 Repair of Congenital Mitral Valve Insufficiency Roland Hetzer, MD, PhD, and Eva Maria Delmo Walter, MS, MD, PhD Principles of Mitral Valve Repair We believe that mitral valve repair for congenital mitral
Repair of Congenital Mitral Valve Insufficiency Roland Hetzer, MD, PhD, and Eva Maria Delmo Walter, MS, MD, PhD Principles of Mitral Valve Repair We believe that mitral valve repair for congenital mitral
Ebstein s anomaly is a congenital malformation of the right
 Cone Reconstruction of the Tricuspid Valve for Ebstein s Anomaly: Anatomic Repair Joseph A. Dearani, MD, Emile Bacha, MD, and José Pedro da Silva, MD Division of Cardiovascular Surgery, Mayo Clinic, Rochester,
Cone Reconstruction of the Tricuspid Valve for Ebstein s Anomaly: Anatomic Repair Joseph A. Dearani, MD, Emile Bacha, MD, and José Pedro da Silva, MD Division of Cardiovascular Surgery, Mayo Clinic, Rochester,
The arterial switch operation has been the accepted procedure
 The Arterial Switch Procedure: Closed Coronary Artery Transfer Edward L. Bove, MD The arterial switch operation has been the accepted procedure for the repair of transposition of the great arteries (TGA)
The Arterial Switch Procedure: Closed Coronary Artery Transfer Edward L. Bove, MD The arterial switch operation has been the accepted procedure for the repair of transposition of the great arteries (TGA)
Assessing Cardiac Anatomy With Digital Subtraction Angiography
 485 JACC Vol. 5, No. I Assessing Cardiac Anatomy With Digital Subtraction Angiography DOUGLAS S., MD, FACC Cleveland, Ohio The use of intravenous digital subtraction angiography in the assessment of patients
485 JACC Vol. 5, No. I Assessing Cardiac Anatomy With Digital Subtraction Angiography DOUGLAS S., MD, FACC Cleveland, Ohio The use of intravenous digital subtraction angiography in the assessment of patients
FUNCTIONALLY SINGLE VENTRICLE
 MORPHOLOGICAL DETERMINANTS VI TRAN EuroEcho, Budapest, 7 th December 2011 DECLARATION OF CONFLICT OF INTEREST: I have nothing to declare What is the functionally single ventricle? The heart that is incapable
MORPHOLOGICAL DETERMINANTS VI TRAN EuroEcho, Budapest, 7 th December 2011 DECLARATION OF CONFLICT OF INTEREST: I have nothing to declare What is the functionally single ventricle? The heart that is incapable
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin. Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong
 Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
Aortic valve repair is an accepted option for aortic valve
 Complex Aortic Valve Disease in Children Christopher W. Baird, MD,* and Pedro J. del Nido, MD Aortic valve repair is an accepted option for aortic valve pathologic conditions in children and young adults.
Complex Aortic Valve Disease in Children Christopher W. Baird, MD,* and Pedro J. del Nido, MD Aortic valve repair is an accepted option for aortic valve pathologic conditions in children and young adults.
Unexpected resolution of first trimester fetal valve stenosis: consequence
 Unexpected resolution of first trimester fetal valve stenosis: consequence of developmental remodeling? Gardiner, Helena M. The Fetal Center, Children s Memorial Hermann Hospital, McGovern Medical School,
Unexpected resolution of first trimester fetal valve stenosis: consequence of developmental remodeling? Gardiner, Helena M. The Fetal Center, Children s Memorial Hermann Hospital, McGovern Medical School,
Mitral valve infective endocarditis (IE) is the most
 Mitral Valve Replacement for Infective Endocarditis With Annular Abscess: Annular Reconstruction Gregory J. Bittle, MD, Murtaza Y. Dawood, MD, and James S. Gammie, MD Mitral valve infective endocarditis
Mitral Valve Replacement for Infective Endocarditis With Annular Abscess: Annular Reconstruction Gregory J. Bittle, MD, Murtaza Y. Dawood, MD, and James S. Gammie, MD Mitral valve infective endocarditis
Ventricular Septal Defect Associated with Aortic Regurgitation
 Ventricular Septal Defect Associated with Aortic Regurgitation Kouichi Hisatomi, M.D., Kenichi Kosuga, M.D., Tadashi somura, M.D., Haruo Akagawa, M.D., Kiroku Ohishi, M.D., and Michihiro Koga, M.D. ABSTRACT
Ventricular Septal Defect Associated with Aortic Regurgitation Kouichi Hisatomi, M.D., Kenichi Kosuga, M.D., Tadashi somura, M.D., Haruo Akagawa, M.D., Kiroku Ohishi, M.D., and Michihiro Koga, M.D. ABSTRACT
Pacing in patients with congenital heart disease: part 1
 Pacing in patients with congenital heart disease: part 1 September 2013 Br J Cardiol 2013;20:117 20 doi: 10.5837/bjc/2013.028 Authors: Khaled Albouaini, Archana Rao, David Ramsdale View details Only a
Pacing in patients with congenital heart disease: part 1 September 2013 Br J Cardiol 2013;20:117 20 doi: 10.5837/bjc/2013.028 Authors: Khaled Albouaini, Archana Rao, David Ramsdale View details Only a
Pediatric Echocardiography Examination Content Outline
 Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
The clinical problem of atrioventricular valve regurgitation
 Mitral Regurgitation in Congenital Heart Defects: Surgical Techniques for Reconstruction Richard G. Ohye Mitral valve regurgitation (MR) is an important source of morbidity and mortality worldwide. While
Mitral Regurgitation in Congenital Heart Defects: Surgical Techniques for Reconstruction Richard G. Ohye Mitral valve regurgitation (MR) is an important source of morbidity and mortality worldwide. While
3 Aortopulmonary Window
 0 0 0 0 0 Aortopulmonary Window Introduction Communications between the ascending aorta and pulmonary artery constitute a spectrum of malformations which is collectively designated aortopulmonary window,
0 0 0 0 0 Aortopulmonary Window Introduction Communications between the ascending aorta and pulmonary artery constitute a spectrum of malformations which is collectively designated aortopulmonary window,
Posterior leaflet prolapse is the most common lesion seen
 Techniques for Repairing Posterior Leaflet Prolapse of the Mitral Valve Robin Varghese, MD, MS, and David H. Adams, MD Posterior leaflet prolapse is the most common lesion seen in degenerative mitral valve
Techniques for Repairing Posterior Leaflet Prolapse of the Mitral Valve Robin Varghese, MD, MS, and David H. Adams, MD Posterior leaflet prolapse is the most common lesion seen in degenerative mitral valve
Diversion of the inferior vena cava following repair of atrial septal defect causing hypoxemia
 Marshall University Marshall Digital Scholar Internal Medicine Faculty Research Spring 5-2004 Diversion of the inferior vena cava following repair of atrial septal defect causing hypoxemia Ellen A. Thompson
Marshall University Marshall Digital Scholar Internal Medicine Faculty Research Spring 5-2004 Diversion of the inferior vena cava following repair of atrial septal defect causing hypoxemia Ellen A. Thompson
Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve
 OPEN ACCESS Images in cardiology Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve Ahmed Mahgoub 1, Hassan Kamel 2, Walid Simry 1, Hatem Hosny 1, * 1 Aswan Heart
OPEN ACCESS Images in cardiology Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve Ahmed Mahgoub 1, Hassan Kamel 2, Walid Simry 1, Hatem Hosny 1, * 1 Aswan Heart
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy
 Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
The outlook for patients with hypoplastic left heart syndrome (HLHS) Tricuspid valve repair in hypoplastic left heart syndrome CHD
 Ohye et al Surgery for Congenital Heart Disease Tricuspid valve repair in hypoplastic left heart syndrome Richard G. Ohye, MD a Carlen A. Gomez, MD b Caren S. Goldberg, MD, MS b Holly L. Graves, BA a Eric
Ohye et al Surgery for Congenital Heart Disease Tricuspid valve repair in hypoplastic left heart syndrome Richard G. Ohye, MD a Carlen A. Gomez, MD b Caren S. Goldberg, MD, MS b Holly L. Graves, BA a Eric
Anatomy of the coronary arteries in transposition
 Thorax, 1978, 33, 418-424 Anatomy of the coronary arteries in transposition of the great arteries and methods for their transfer in anatomical correction MAGDI H YACOUB AND ROSEMARY RADLEY-SMITH From Harefield
Thorax, 1978, 33, 418-424 Anatomy of the coronary arteries in transposition of the great arteries and methods for their transfer in anatomical correction MAGDI H YACOUB AND ROSEMARY RADLEY-SMITH From Harefield
The role of intraoperative TOE in congenital cardiac surgery
 The role of intraoperative TOE in congenital cardiac surgery Justiaan Swanevelder Dept of Anaesthesia Groote Schuur and Red Cross War Memorial Children s Hospitals University of Cape Town, South Africa
The role of intraoperative TOE in congenital cardiac surgery Justiaan Swanevelder Dept of Anaesthesia Groote Schuur and Red Cross War Memorial Children s Hospitals University of Cape Town, South Africa
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE
 SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
Adult Echocardiography Examination Content Outline
 Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
Surgical Treatment for Double Outlet Right Ventricle. Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery
 for Double Outlet Right Ventricle Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Intraventricular tunnel (Kawashima) First repair of Taussig-Bing anomaly (Kirklin) Taussig-Bing
for Double Outlet Right Ventricle Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Intraventricular tunnel (Kawashima) First repair of Taussig-Bing anomaly (Kirklin) Taussig-Bing
Implantation of Cardioverter Defibrillator After Percutaneous Closure of Atrial Septal Defect
 The Ochsner Journal 10:27 31, 2010 f Academic Division of Ochsner Clinic Foundation Implantation of Cardioverter Defibrillator After Percutaneous Closure of Atrial Septal Defect Anas Bitar, MD, Maria Malaya
The Ochsner Journal 10:27 31, 2010 f Academic Division of Ochsner Clinic Foundation Implantation of Cardioverter Defibrillator After Percutaneous Closure of Atrial Septal Defect Anas Bitar, MD, Maria Malaya
Ebstein s anomaly is characterized by malformation of
 Fenestrated Right Ventricular Exclusion (Starnes Procedure) for Severe Neonatal Ebstein s Anomaly Brian L. Reemtsen, MD,* and Vaughn A. Starnes, MD*, Ebstein s anomaly is characterized by malformation
Fenestrated Right Ventricular Exclusion (Starnes Procedure) for Severe Neonatal Ebstein s Anomaly Brian L. Reemtsen, MD,* and Vaughn A. Starnes, MD*, Ebstein s anomaly is characterized by malformation
Surgical Management of TOF in Adults. Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital
 Surgical Management of TOF in Adults Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital Tetralogy of Fallot (TOF) in Adults Most common cyanotic congenital heart
Surgical Management of TOF in Adults Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital Tetralogy of Fallot (TOF) in Adults Most common cyanotic congenital heart
Mitral Valve Disease, When to Intervene
 Mitral Valve Disease, When to Intervene Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Current ACC/AHA guideline Stages
Mitral Valve Disease, When to Intervene Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Current ACC/AHA guideline Stages
"Giancarlo Rastelli Lecture"
 "Giancarlo Rastelli Lecture" Surgical treatment of Malpositions of the Great Arteries Pascal Vouhé Giancarlo Rastelli (1933 1970) Cliquez pour modifier les styles du texte du masque Deuxième niveau Troisième
"Giancarlo Rastelli Lecture" Surgical treatment of Malpositions of the Great Arteries Pascal Vouhé Giancarlo Rastelli (1933 1970) Cliquez pour modifier les styles du texte du masque Deuxième niveau Troisième
Characteristics and Management of Cleft Mitral Valve
 Journal of the American College of Cardiology Vol. 42, No. 11, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.07.019
Journal of the American College of Cardiology Vol. 42, No. 11, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.07.019
Repair or Replacement
 Surgical intervention post MitraClip Device: Repair or Replacement Saudi Heart Association, February 21-24 Rüdiger Lange, MD, PhD Nicolo Piazza, MD, FRCPC, FESC German Heart Center, Munich, Germany Division
Surgical intervention post MitraClip Device: Repair or Replacement Saudi Heart Association, February 21-24 Rüdiger Lange, MD, PhD Nicolo Piazza, MD, FRCPC, FESC German Heart Center, Munich, Germany Division
Despite advances in our understanding of the pathophysiology
 Suture Relocation of the Posterior Papillary Muscle in Ischemic Mitral Regurgitation Benjamin B. Peeler MD,* and Irving L. Kron MD,*, *Department of Cardiovascular Surgery, University of Virginia, Charlottesville,
Suture Relocation of the Posterior Papillary Muscle in Ischemic Mitral Regurgitation Benjamin B. Peeler MD,* and Irving L. Kron MD,*, *Department of Cardiovascular Surgery, University of Virginia, Charlottesville,
COMBINED CONGENITAL SUBAORTIC STENOSIS AND INFUNDIBULAR PULMONARY STENOSIS*
 COMBINED CONGENITAL SUBAORTIC STENOSIS AND INFUNDIBULAR PULMONARY STENOSIS* BY HENRY N. NEUFELD,t PATRICK A. ONGLEY, AND JESSE E. EDWARDS From the Sections of Pa?diatrics and Pathological Anatomy, Mayo
COMBINED CONGENITAL SUBAORTIC STENOSIS AND INFUNDIBULAR PULMONARY STENOSIS* BY HENRY N. NEUFELD,t PATRICK A. ONGLEY, AND JESSE E. EDWARDS From the Sections of Pa?diatrics and Pathological Anatomy, Mayo
Aortic root enlargement is an invaluable surgical technique
 Aortic Root Enlargement in the Adult Christopher M. Feindel, MD, CM, FRCS(C) Aortic root enlargement is an invaluable surgical technique with which every cardiac surgeon performing aortic valve replacement
Aortic Root Enlargement in the Adult Christopher M. Feindel, MD, CM, FRCS(C) Aortic root enlargement is an invaluable surgical technique with which every cardiac surgeon performing aortic valve replacement
Partial anomalous pulmonary venous connection to superior
 Cavo-Atrial Anastomosis Technique for Partial Anomalous Pulmonary Venous Connection to the Superior Vena Cava The Warden Procedure Robert A. Gustafson, MD Partial anomalous pulmonary venous connection
Cavo-Atrial Anastomosis Technique for Partial Anomalous Pulmonary Venous Connection to the Superior Vena Cava The Warden Procedure Robert A. Gustafson, MD Partial anomalous pulmonary venous connection
found that some patients without stenotic lesions had blood velocity or pressure measurement across the
 Br Heart J 1985; 53: 640-4 Increased blood velocities in the heart and great vessels of patients with congenital heart disease An assessment of their significance in the absence of valvar stenosis STANLEY
Br Heart J 1985; 53: 640-4 Increased blood velocities in the heart and great vessels of patients with congenital heart disease An assessment of their significance in the absence of valvar stenosis STANLEY
JOINT MEETING 2 Tricuspid club Chairpersons: G. Athanassopoulos, A. Avgeropoulou, M. Khoury, G. Stavridis
 JOINT MEETING 2 Tricuspid club Chairpersons: G. Athanassopoulos, A. Avgeropoulou, M. Khoury, G. Stavridis Similarities and differences in Tricuspid vs. Mitral Valve Anatomy and Imaging. Echo evaluation
JOINT MEETING 2 Tricuspid club Chairpersons: G. Athanassopoulos, A. Avgeropoulou, M. Khoury, G. Stavridis Similarities and differences in Tricuspid vs. Mitral Valve Anatomy and Imaging. Echo evaluation
Introduction. Aortic Valve. Outflow Tract and Aortic Valve Annulus
 Chapter 1: Surgical anatomy of the aortic and mitral valves Jordan RH Hoffman MD, David A. Fullerton MD, FACC University of Colorado School of Medicine, Department of Surgery, Division of Cardiothoracic
Chapter 1: Surgical anatomy of the aortic and mitral valves Jordan RH Hoffman MD, David A. Fullerton MD, FACC University of Colorado School of Medicine, Department of Surgery, Division of Cardiothoracic
MEDICAL MANAGEMENT WITH CAVEATS 1. In one study of 50 CHARGE patients with CHD, 75% required surgery. 2. Children with CHARGE may be resistant to chlo
 CARDIOLOGY IN CHARGE SYNDROME: FOR THE PHYSICIAN Angela E. Lin, M.D. Teratology Program/Active Malformation Surveillance, Brigham and Women's Hospital, Old PBBH-B501, 75 Francis St., Boston, MA 02115 alin@partners.org
CARDIOLOGY IN CHARGE SYNDROME: FOR THE PHYSICIAN Angela E. Lin, M.D. Teratology Program/Active Malformation Surveillance, Brigham and Women's Hospital, Old PBBH-B501, 75 Francis St., Boston, MA 02115 alin@partners.org
Congenital pulmonary vein (PV) stenosis with anatomically
 Pulmonary Vein Stenosis With Normal Connection: Associated Cardiac Abnormalities and Variable Outcome John P. Breinholt, BS, John A. Hawkins, MD, LuAnn Minich, MD, Lloyd Y. Tani, MD, Garth S. Orsmond,
Pulmonary Vein Stenosis With Normal Connection: Associated Cardiac Abnormalities and Variable Outcome John P. Breinholt, BS, John A. Hawkins, MD, LuAnn Minich, MD, Lloyd Y. Tani, MD, Garth S. Orsmond,
Cardiac MRI in ACHD What We. ACHD Patients
 Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Chapter 24: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines Ashwin Nathan, MD, Saif Anwaruddin, MD, FACC Penn Medicine
 Chapter 24: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines Ashwin Nathan, MD, Saif Anwaruddin, MD, FACC Penn Medicine Mitral regurgitation, regurgitant flow between the
Chapter 24: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines Ashwin Nathan, MD, Saif Anwaruddin, MD, FACC Penn Medicine Mitral regurgitation, regurgitant flow between the
Anatomy of left ventricular outflow tract'
 Anatomy of left ventricular outflow tract' ROBERT WALMSLEY British Heart Journal, 1979, 41, 263-267 From the Department of Anatomy and Experimental Pathology, The University, St Andrews, Scotland SUMMARY
Anatomy of left ventricular outflow tract' ROBERT WALMSLEY British Heart Journal, 1979, 41, 263-267 From the Department of Anatomy and Experimental Pathology, The University, St Andrews, Scotland SUMMARY
S occur months or years after repair of congenital heart
 Late Reoperation for Systemic Atrioventricular Valve Regurgitation After Repair of Congenital Heart Defects John J. Lamberti, MD, Thomas S. Jensen, MD, Todd M. Grehl, MD, James H. Oury, MD, J. Deane Waldman,
Late Reoperation for Systemic Atrioventricular Valve Regurgitation After Repair of Congenital Heart Defects John J. Lamberti, MD, Thomas S. Jensen, MD, Todd M. Grehl, MD, James H. Oury, MD, J. Deane Waldman,
Biventricular Enlargement/ Hypertrophy
 Biventricular Enlargement/ Hypertrophy Keywords congenital heart disease left ventricular hypertrophy right ventricular hypertrophy SR MITTAL Abstract Electrocardiographic diagnosis of early biventricular
Biventricular Enlargement/ Hypertrophy Keywords congenital heart disease left ventricular hypertrophy right ventricular hypertrophy SR MITTAL Abstract Electrocardiographic diagnosis of early biventricular
Functional anatomy of the aortic root. ΔΡΟΣΟΣ ΓΕΩΡΓΙΟΣ Διεσθσνηής Καρδιοθωρακοτειροσργικής Κλινικής Γ.Ν. «Γ. Παπανικολάοσ» Θεζζαλονίκη
 Functional anatomy of the aortic root ΔΡΟΣΟΣ ΓΕΩΡΓΙΟΣ Διεσθσνηής Καρδιοθωρακοτειροσργικής Κλινικής Γ.Ν. «Γ. Παπανικολάοσ» Θεζζαλονίκη What is the aortic root? represents the outflow tract from the LV provides
Functional anatomy of the aortic root ΔΡΟΣΟΣ ΓΕΩΡΓΙΟΣ Διεσθσνηής Καρδιοθωρακοτειροσργικής Κλινικής Γ.Ν. «Γ. Παπανικολάοσ» Θεζζαλονίκη What is the aortic root? represents the outflow tract from the LV provides
Reoperation for Left Ventricular Outflow Tract Obstruction After Repair of Atrioventricular Septal Defect
 Reoperation for Left Ventricular Outflow Tract Obstruction After Repair of Atrioventricular Septal Defect David M. Overman Division of Pediatric Cardiac Surgery The Children s Heart Clinic Chief, Division
Reoperation for Left Ventricular Outflow Tract Obstruction After Repair of Atrioventricular Septal Defect David M. Overman Division of Pediatric Cardiac Surgery The Children s Heart Clinic Chief, Division
Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!!
 Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!! Abha'Khandelwal,'MD,'MS' 'Stanford'University'School'of'Medicine'
Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!! Abha'Khandelwal,'MD,'MS' 'Stanford'University'School'of'Medicine'
Pulmonary Valve Replacement
 Pulmonary Valve Replacement with Fascia Lata J. C. R. Lincoln, F.R.C.S., M. Geens, M.D., M. Schottenfeld, M.D., and D. N. Ross, F.R.C.S. ABSTRACT The purpose of this paper is to describe a technique of
Pulmonary Valve Replacement with Fascia Lata J. C. R. Lincoln, F.R.C.S., M. Geens, M.D., M. Schottenfeld, M.D., and D. N. Ross, F.R.C.S. ABSTRACT The purpose of this paper is to describe a technique of
The Management of the Cleft Mitral
 The Management of the Cleft Mitral Valve in Endocardia1 Cushion Defects L. H. S. Van Mierop, M.D., and Ralph D. Alley, M.D. C ustomarily, in the surgical treatment of endocardial cushion defects the cleft
The Management of the Cleft Mitral Valve in Endocardia1 Cushion Defects L. H. S. Van Mierop, M.D., and Ralph D. Alley, M.D. C ustomarily, in the surgical treatment of endocardial cushion defects the cleft
Absent Pulmonary Valve Syndrome
 Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
The pulmonary valve is the most common heart valve
 Biologic versus Mechanical Valve Replacement of the Pulmonary Valve After Multiple Reconstructions of the RVOT Tract S. Adil Husain, MD, and John Brown, MD Indiana University School of Medicine, Department
Biologic versus Mechanical Valve Replacement of the Pulmonary Valve After Multiple Reconstructions of the RVOT Tract S. Adil Husain, MD, and John Brown, MD Indiana University School of Medicine, Department
PROGRESS IN CARDIOVASCULAR SURGERY. Congenital Mitral Stenosis and Mitral Insufficiency
 PROGRESS IN CARDIOVASCULAR SURGERY Congenital Mitral Stenosis and Mitral Insufficiency GEORGE W. B. STARKEY, M.D.* Boston, Massachusetts CONGENITAL MITRAL STENOSIS AND mitral insufficiency are rare, particularly
PROGRESS IN CARDIOVASCULAR SURGERY Congenital Mitral Stenosis and Mitral Insufficiency GEORGE W. B. STARKEY, M.D.* Boston, Massachusetts CONGENITAL MITRAL STENOSIS AND mitral insufficiency are rare, particularly
An understanding of the many factors involved in the
 Atrioventricular Valve Dysfunction: Evaluation by Doppler and Cross-Sectional Ultrasound Norman H. Silverman, MD, and Doff B. McElhinney, MD Division of Pediatric Cardiology, Department of Pediatrics,
Atrioventricular Valve Dysfunction: Evaluation by Doppler and Cross-Sectional Ultrasound Norman H. Silverman, MD, and Doff B. McElhinney, MD Division of Pediatric Cardiology, Department of Pediatrics,
