NKX2.5 Mutations in Patients With Congenital Heart Disease
|
|
|
- Victoria Boone
- 6 years ago
- Views:
Transcription
1 Journal of the American College of Cardiology Vol. 42, No. 9, by the American College of Cardiology Foundation ISSN /03/$30.00 Published by Elsevier Inc. doi: /s (03) NKX2.5 Mutations in Patients With Congenital Heart Disease Congenital Heart Disease Doff B. McElhinney, MD,* Elizabeth Geiger, MS,* Joshua Blinder, BS,* D. Woodrow Benson, MD, PHD, Elizabeth Goldmuntz, MD, FACC* Philadelphia, Pennsylvania; and Cincinnati, Ohio OBJECTIVES BACKGROUND METHODS RESULTS CONCLUSIONS The purpose of this study was to estimate the frequency of NKX2.5 mutations in specific cardiovascular anomalies and investigate genotype-phenotype correlations in individuals with NKX2.5 mutations. Recent reports have implicated mutations in the transcription factor NKX2.5 as a cause of various congenital heart defects (CHD). We tested genomic deoxyribonucleic acid from 608 prospectively recruited patients with conotruncal anomalies (n 370), left-sided lesions (n 160), secundum atrial septal defect (ASD) (n 71), and Ebstein s malformation (n 7) for NKX2.5 mutations. Twelve distinct mutations in the NKX2.5 coding region were identified in 18 of 608 patients (3%), including 9 of 201 (4%) with tetralogy of Fallot, 3 of 71 (4%) with a secundum ASD, one each with truncus arteriosus, double-outlet right ventricle, L-transposition of the great arteries, interrupted aortic arch, hypoplastic left heart syndrome, and aortic coarctation, but in no patients with D-transposition of the great arteries (n 86) or valvar aortic stenosis (n 21). Eleven of the mutations were amino acid-altering missense nucleotide substitutions or deletions, and one was predicted to cause premature termination of translation. None of the mutations were in the homeodomain. Sixteen of the 18 individuals with NKX2.5 mutations in this study had no family history of congenital cardiovascular anomalies, and one had first-degree atrioventricular (AV) block. NKX2.5 mutations occur in a small percentage of patients with various CHD. Most of the mutations identified in this study were missense, outside the homeodomain, and not associated with AV block. These findings suggest that NKX2.5 mutations in nonhomeodomain regions may be important in the development of human structural cardiac defects. (J Am Coll Cardiol 2003;42:1650 5) 2003 by the American College of Cardiology Foundation GENETICS IN CONTEXT Focus on gene mutations in congenital heart disease. Although congenital heart defects (CHD) are the leading cause of morbidity and mortality in infants, the etiology of most CHD remains unknown, with the influence of genetics still a topic of debate. The increased recurrence risk for parents with an affected child suggests the potential for inherited predisposing mutations that are incompletely penetrant. Heterozygous mutations in the transcription factor, NKX2-5, were among the first evidence of a genetic cause for congenital heart disease and were initially found in pedigrees with autosomal dominant transmission of cardiac septal defects. Most reported NKX2-5 mutations were in the homeodomain, a critical part of the protein that interacts with deoxyribonucleic acid (DNA), and were typically associated with cardiac conduction anomalies. However, the impact of NKX2-5 mutations on sporadic forms of isolated CHD in a broad population was unknown. In this study by McElhinney et al., 608 children with a variety of non-familial CHD, but normal conduction, were studied for mutations in NKX2-5. Three percent of the entire cohort, a relatively large number for a single gene, had mutations that did not appear to be polymorphisms. Interestingly, none were in the critical homeodomain, suggesting that other protein domains likely affect specific developmental processes without disrupting development of the conduction system. It remains intriguing that disruption of a single gene can cause the diverse forms of CHD described here. This could be attributed to specific roles of distinct regions of the NKX2-5 protein or simply the presence of other stochastic events. Finally, the notion of CHD as an inherited disorder with incomplete penetrance is supported by the common identification of an NKX2-5 point mutation in a normal parent. Similar analyses for other critical genes may together begin to establish a genetic basis for most sporadic forms of CHD. This effort will lay the foundation for more accurate genetic counseling, clinical interventions, and most importantly, new approaches for prevention of CHD in those who are genetically susceptible. Deepak Srivastava University of Texas Southwestern Medical Center Dallas, Texas From *The Children s Hospital of Philadelphia and University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania; and Children s Hospital Medical Center, Cincinnati, Ohio. Supported by the National Institutes of Health (P50 HL62177 [E.G.] and P50 HL61006 [D.W.B.]), The Ethel Brown Foerderer Fund for Excellence (E.G.), The University of Pennsylvania Research Foundation (E.G.), and the General Clinical Research Center of The Children s Hospital of Philadelphia (M01-RR00240 [E.G.]). Present address for Dr. McElhinney: Department of Cardiology, Children s Hospital, Boston, Massachusetts. Manuscript received December 11, 2002; revised manuscript received March 18, 2003, accepted May 30, 2003.
2 JACC Vol. 42, No. 9, 2003 November 5, 2003: McElhinney et al. NKX2.5 Mutations in CHD 1651 Abbreviations and Acronyms ASD atrial septal defect AV atrioventricular CHD congenital heart defect CSGE conformation-sensitive gel electrophoresis DNA deoxyribonucleic acid PCR polymerase chain reaction Mutations of NKX2.5, primarily in the homeodomain, have been identified in at least 11 kindreds with atrioventricular (AV) conduction block and concurrent congenital heart malformations, including secundum atrial septal defect (ASD) and tetralogy of Fallot (Table 1) (1 3). For this reason, we genotyped prospectively recruited patients with tetralogy of Fallot for NKX2.5 and identified four missense mutations in six of the 114 patients (5%) (4). All of these mutations were outside of the homeodomain and in individuals without AV conduction abnormalities, as was the case in one previously reported patient with tetralogy of Fallot and an NKX2.5 mutation (2). To investigate further the genotype-phenotype correlation in patients with congenital heart disease and an NKX2.5 mutation, and to estimate the frequency of NKX2.5 mutations in individuals with specific congenital cardiovascular anomalies, we genotyped 608 prospectively recruited patients with congenital heart disease by conformation-sensitive gel electrophoresis (CSGE) and/or direct sequencing. In contrast to earlier studies, which ascertained subjects on the basis of familial congenital heart disease (1 3), the subjects in this report were enrolled on the basis of specific cardiovascular phenotypes without regard for family history or the presence of AV conduction abnormalities. METHODS Table 1. Previously Reported NKX2.5 Mutations* Mutation Site # Patients # With AVB # With ASD Patients. Genotype data from a total of 608 subjects are summarized for this report. Genomic deoxyribonucleic acid (DNA) from 474 new subjects with conotruncal cardiovascular anomalies (n 236), left-sided lesions (n 160), secundum ASD (n 71), and Ebstein s malformation (n 7) were tested for mutations in the coding region of NKX2.5. Conotruncal anomalies included new patients with tetralogy of Fallot (n 67), D-transposition of the great arteries (n 86), double-outlet right ventricle (n 31), interrupted aortic arch (n 23), truncus arteriosus (n 22), and L-transposition of the great arteries (n 7). Left-sided lesions included hypoplastic left heart syndrome (n 80), coarctation of the aorta (n 59), and valvar aortic stenosis (n 21). To estimate frequency, we included the 134 patients with tetralogy of Fallot previously studied in our laboratories and reported earlier, given that they were enrolled in the same protocols as the other subjects (2,4). Therefore, the total number of patients described in this report is 608. Subjects were drawn from a cohort of patients that were recruited prospectively into studies on the genetic basis of congenital heart disease from 1991 to Informed consent was obtained from study participants in accordance with protocols approved by the Institutional Review Board for Human Research at the Children s Hospital of Philadelphia or the Medical University of South Carolina. All subjects with conotruncal malformations were tested for a chromosome 22q11 deletion as previously described (5). Some patients underwent other cytogenetic testing if clinically indicated. Patients were invited to enroll in the aforementioned studies after admission to the hospital, exclusively on the basis of the CHD present in the proband, with no selection for or against familial congenital heart disease or syndromic associations. However, patients with a chromosome 22q11 deletion, trisomy 21, or other identified chromosomal anomaly were excluded from the present analysis. Cardiovascular diagnoses were confirmed by attending pediatric cardiologists (E.G. and D.W.B.), who reviewed echocardiograms and/or echocardiogram reports, cardiac catheterization reports, and operative notes if applicable. Electrocardiograms were reviewed for abnormalities # With Other CHD Int 1DSG 1T Splice site C554T (Gln149ter) Homeodomain TOF, VSD C618T (Gln170ter) Homeodomain C642T (Thr178Met) Homeodomain C673A (Asn188Cys) Homeodomain TV anomaly C674G (Arg189Gly) Homeodomain TV anomaly A681G (Tyr191Cys) Homeodomain VSD C701T (Gln198ter) 3 coding region C886A (Tyr259ter) 3 coding region DORV, VSD Total (98%) 37 (82%) 11 (24%) *Includes the reports of Schott et al (1), Hosoda et al. (3), and Benson et al. (2), although the 20 patients with sporadic tetralogy of Fallot in the report by Benson et al., who are included in the current study, are excluded from this table. Patients reported by Goldmuntz et al. (4); who are included in the current study, are excluded from this table as well. Two separate families have been reported with these mutations. One of the two families with this mutation included four members who were not genotyped but had tetralogy of Fallot (n 2) or a ventricular septal defect (n 2). All three of these patients had Ebstein s malformation. ASD secundum atrial septal defect; AVB atrioventricular block; CHD congenital heart defects; DORV double-outlet right ventricle; TOF tetralogy of Fallot; TV tricuspid valve; VSD ventricular septal defect.
3 1652 McElhinney et al. JACC Vol. 42, No. 9, 2003 NKX2.5 Mutations in CHD November 5, 2003: Table 2. Frequency of NKX2.5 Mutations Among 608 Patients With Congenital Heart Disease Cardiovascular Anomaly # Patients Genotyped # With Mutations Secundum atrial septal defect 71 3 (4%) Ebstein s malformation 7 0 Conotruncal anomalies Tetralogy of Fallot* (4%) D-transposition of the great arteries 86 0 Double-outlet right ventricle 31 1 (3%) Interrupted aortic arch 23 1 (4%) Truncus arteriosus 22 1 (4%) L-transposition of the great arteries 7 1 (14%) Subtotal (conotruncal anomalies) (4%) Left-sided lesions Hypoplastic left heart syndrome 80 1 (1%) Coarctation of the aorta 59 1 (2%) Valvar aortic stenosis 21 0 Subtotal (left-sided lesions) (2%) Total (3%) *Includes 134 consecutively recruited patients with tetralogy of Fallot described in the reports by Goldmuntz et al. (4) (n 114) and Benson et al. (2) (n 20) seven of whom had NKX2.5 mutations. of AV conduction in all patients with secundum ASD and Ebstein s malformation, as well as those with other anomalies who were found to have an NKX2.5 mutation. Family history of congenital heart disease was ascertained by report (i.e., clinical testing of parents was not performed for the purposes of this study), and an effort was made to obtain parental DNA for mutation analysis in all patients found to have an NKX2.5 mutation. Medical records of patients with an NKX2.5 mutation were reviewed to determine whether any non-cardiac congenital malformations or recognized genetic syndrome was present. Genotype analysis. Deoxyribonucleic acid was extracted from either whole blood or a lymphoblastoid cell line using standard techniques. The coding region of the NKX2.5 gene, including exon/intron boundaries, was amplified from genomic DNA by polymerase chain reaction (PCR) using standard techniques, as described previously (4). The PCR products were screened for sequence alterations either by CSGE, a modification of heteroduplex analysis (6), as described previously (4), or by direct sequencing. The PCR products were directly sequenced in certain cohorts (e.g., all patients with tetralogy of Fallot, Ebstein s malformation, or an ASD) and in selected patients with other diagnoses. The remaining patients were evaluated initially by CSGE, and in those with an aberrant band on the CSGE gel, the shifted region of the gene was subsequently sequenced with an automated cycle sequencer (ABI Prism 373 or 377 Sequencer, ABI BigDye Terminator Cycle Sequencing Kit, Applied Biosystems, Foster City, California), as described previously (4). Among the 608 patients, the NKX2.5 coding region was sequenced in its entirety in 316 (52%) and partially in 80 (13%). Sequence alterations were examined in the context of the open reading frame to determine whether they would alter the corresponding amino acid. RESULTS Twelve distinct mutations in the coding region of NKX2.5 were identified in 18 of the 608 (3%) probands evaluated. Four mutations in seven of these subjects were previously reported (2,4) but are included in this report to allow for estimation of frequency, as previously noted. The cardiovascular anomalies in the individuals tested and affected are summarized in Table 2. The details of the specific mutations are summarized in Table 3. Among patients with conotruncal anomalies, NKX2.5 mutations were detected in 13 patients, including 9 of 201 patients with tetralogy of Fallot (4%), and one each with truncus arteriosus, double-outlet right ventricle, interrupted aortic arch, and L-transposition of the great arteries. Notably, mutations were found among the 86 patients with Table 3. NKX2.5 Mutations Identified Among 608 Patients With Congenital Heart Disease Mutation Site* Gene Region # Patients Type of Congenital Heart Defect A44T (Lys15Ile) TN domain 1 Secundum atrial septal defect G61C (Glu21Gln) 5 coding region 1 Tetralogy of Fallot A65C (Gln22Pro) 5 coding region 1 Tetralogy of Fallot C73T (Arg25Cys) 5 coding region 7 Tetralogy of Fallot (n 4 ) Truncus arteriosus (n 1) Interrupted aortic arch (n 1) Hypoplastic left heart syndrome (n 1) C188T (Ala63Val) 5 coding region 1 L-transposition of the great arteries C380A (Ala127Glu) 5 coding region 1 Secundum atrial septal defect C646T (Arg216Cys) NK domain 1 Tetralogy of Fallot C656T (Ala219Val) NK domain 1 Tetralogy of Fallot InsTCCCT701 (D235AFSter) 3 coding region 1 Secundum atrial septal defect First-degree atrioventricular block C823A (Pro275Thr) 3 coding region 1 Coarctation of the aorta delaac871 (del291asn) 3 coding region 1 Double-outlet right ventricle G967A (Ala323Thr) 3 coding region 1 Tetralogy of Fallot *Numbering of nucleotides begins with the A of the ATG start codon as 1. Two of the patients with tetralogy of Fallot and an Arg25Cys mutation had pulmonary atresia, as did the patient with tetralogy of Fallot and an Ala219Val mutation; the other four patients with tetralogy of Fallot and NKX2.5 mutations had pulmonary stenosis. Major aortopulmonary collateral arteries were present in both patients with tetralogy of Fallot, pulmonary atresia, and an Arg25Cys mutation.
4 JACC Vol. 42, No. 9, 2003 November 5, 2003: McElhinney et al. NKX2.5 Mutations in CHD 1653 Figure 1. Diagram of the NKX2.5 gene, depicting the locations of new and previously reported mutations. Mutations identified in this report are noted above the diagram of the gene, with the number of patients having the mutation noted. The arrows below the diagram indicate previously reported mutations (1 3). Coding regions are indicated as hatched or white boxes, with the conserved TN domain (TN), homeodomain (HD), and NK2 domain (NK) indicated by labeled white boxes. The 5 and 3 untranslated regions are depicted as smaller boxes, and the single intron as a broken line. The small hearts indicate previously reported NKX2.5 mutations in which one or more affected individuals had structural cardiac anomalies other than a secundum ASD. ASD atrial septal defect; AVB atrioventricular block; COA coarctation of the aorta; DORV double-outlet right ventricle; HLHS hypoplastic left heart syndrome; IAA interrupted aortic arch; L-TGA L-transposition of the great arteries; TA truncus arteriosus; TOF tetralogy of Fallot. D-transposition of the great arteries. Among patients with conotruncal anomalies and an NKX2.5 mutation, the sidedness or branching pattern of the aortic arch was abnormal in 6 of the 11 in whom the aortic arch anatomy was defined, including 5 of the 9 mutation-positive patients with tetralogy of Fallot and the mutation-positive patient with interrupted aortic arch. Of 160 patients with left-sided lesions, two were found to have mutations, including one with hypoplastic left heart syndrome and one with coarctation of the aorta and an intact ventricular septum. Mutations were detected in 3 of 71 patients with a secundum ASD (4%), and none of 7 with Ebstein s malformation. None of the mutation-positive patients had extracardiac anomalies or syndromic features. Eleven of the mutations were missense nucleotide substitutions (n 10) or in-frame deletions (n 1) predicted to result in alteration or deletion of a single amino acid residue, and one was a five-nucleotide insertion predicted to cause premature termination of translation (Table 3). Four of the mutations were located in (n 1) or immediately 3 to (n 3) the conserved amino-terminal TN domain; one was midway between the TN domain and the homeodomain; one was located just 5 to the homeodomain; three were located in (n 2) or immediately 3 to (n 1) the conserved carboxy-terminal NK2 domain; and three were located in the distal 3 coding region (Fig. 1). None of the mutations were in the homeodomain. Abnormal AV conduction was identified in only one mutation-positive patient, who had first-degree AV block with a secundum ASD and an insertion mutation in the 3 coding region predicted to truncate the protein. Sixteen of the 18 individuals with an NKX2.5 mutation had no known family history of congenital heart disease (although parents were not assessed for the presence of occult cardiovascular anomalies in this study). The other two were known to have at least one relative with a cardiovascular defect, including a bileaflet aortic valve in the grandfather of the patient with an Ala127Glu mutation and secundum ASD, and a ventricular septal defect in the father of a patient with an Arg25Cys mutation and tetralogy of Fallot. The respective NKX2.5 mutations were carried by both affected relatives. Of the 16 patients with no family history of congenital heart disease, parental DNA was available and evaluated for the corresponding mutation by direct sequencing in six (Lys15Ile, Glu21Gln, Arg25Cys n 2, Ala219Val, D235AFSter, del291asn). In all six of these cases, one parent carried the mutation, consistent with decreased penetrance. Parental DNA was not available for the remaining 10 patients with mutations. The NKX2.5 gene was sequenced from the genomic DNA of 50 random control subjects who were not known to have congenital cardiovascular disease. None of the 12 mutations detected in our study patients were found in the 100 control chromosomes. Among the mutations identified in our study population, the Arg25Cys mutation was found in multiple subjects, whereas the other mutations were unique. Five of the seven patients with an Arg25Cys mutation in this series were of African-American ethnicity, one was Hispanic, and one was Caucasian. This mutation was identified in 2 of 43 (5%) African-American control subjects, though it was not present in 100 chromosomes from random control subjects, as previously described (4). DISCUSSION NKX2.5 is the earliest known marker of myocardial progenitor cells in all species in which it has been studied (7 10). Just as mutation of the Drosophila NKX2.5 homolog, tinman, results in failure of heart formation (11,12), homozygous inactivation of NKX2.5 in mammals results in impaired cardiac looping and embryonic lethality (13,14). Mice with a heterozygous cardiac-specific deletion of NKX2.5 exhibit a more subtle phenotype, which consists of atrial septal dysmorphogenesis, occasional abnormalities of
5 1654 McElhinney et al. JACC Vol. 42, No. 9, 2003 NKX2.5 Mutations in CHD November 5, 2003: the aortic valve, and, in females, prolongation of the PR interval (15). Numerous studies have investigated the mechanisms of NKX2.5 regulation and its interaction with other transcription factors thought to be important in early cardiac development (14,16 21). Through these and other studies, it has become clear that NKX2.5 plays a central role in the determination of myocardial cell fate, and there is evidence that it may be important during postnatal life as well (22). Accordingly, there has been considerable interest in the potential role of NKX2.5 in human cardiac development and disease. Based on our identification of NKX2.5 mutations in individuals with isolated tetralogy of Fallot (4), we extended our investigation to other conotruncal lesions. The independent finding of an Arg25Cys mutation in an individual with hypoplastic left heart syndrome provided the rationale to extend the scope of our study to encompass prospectively enrolled patients with left-sided lesions as well. The association of NKX2.5 mutations with secundum ASD and tricuspid valve anomalies (2) prompted our prospective evaluation of patients with sporadic secundum ASDs and Ebstein s malformation. In this report, we have detailed our findings in these prospectively recruited cohorts of individuals with primarily sporadic congenital cardiovascular anomalies. Among 608 patients genotyped, 18 (3%) were found to have an NKX2.5 mutation. Although NKX2.5 mutations were found in individuals with cardiovascular anomalies of different types and categories (secundum ASD, various conotruncal anomalies, and two left-sided lesions), the most notable findings of this study are the frequency of mutations in patients with secundum ASD (4%) and tetralogy of Fallot (4%), and the absence of mutations in a relatively large cohort of patients with D-transposition of the great arteries. Eleven of the 12 mutations found in this study were unique. Only the Arg25Cys mutation was found in multiple study patients (n 7). The significance of this mutation is not clear, as has been discussed in a previous report (4). Functional analysis of this mutation reported by Kasahara et al. (23) showed normal nuclear localization and normal binding of the mutant protein to monomeric DNA binding sites, but a threefold higher concentration of mutant protein was required for equally distributed binding to monomeric and dimeric DNA binding sites, suggesting a slight impairment of dimerization by the mutant NKX2.5. The effect of this mutation on NKX2.5 function will require further evaluation. In the majority of previously reported patients with an NKX2.5 mutation in conjunction with familial AV block and a secundum ASD, the NKX2.5 mutation alters coding sense in the homeodomain and/or is predicted to result in a truncated protein (1 3). In contrast, all of the mutations that we detected were in the 5 or 3 coding region of the gene, with no homeodomain mutations. Only one patient with an NKX2.5 mutation (the only patient with a truncating mutation in this series) had AV block. Experimental studies have shown that the carboxy-terminal portion of NKX2.5 is important in NKX2.5 function distinct from the role of the homeodomain (23 25). Kasahara et al. found this portion of the gene to be critical for the cooperative homodimerization and heterodimerization (with other transcription factors such as GATA4) of NKX2.5 protein on dimeric DNA binding sites (24). In functional studies of the previously reported NKX2.5 mutations, these authors demonstrated decreased binding of mutant NKX2.5 proteins with mutations outside the homeodomain to dimeric DNA binding sites despite normal binding to monomeric sites (23). In addition, Schinke et al. (25) examined the effects of cardiac-specific deletion of the NK2 domain of NKX2.5 in mice. Null mutants died in utero at E14.5 and exhibited a variety of structural cardiovascular anomalies, including ventricular septal defect, double-outlet right ventricle, and AV septal defect, as well as downregulation of the ventricular markers Irx4 and MLC-2v, specifically in the right ventricle. Heterozygous NK2 mutant mice, which survived to term, also developed various structural cardiovascular defects. Binding of the NK2 mutant protein to DNA was not impaired, and mutant mice did not demonstrate AV conduction abnormalities. These findings suggest that the NK2 domain plays an important role in cardiovascular development independent of the homeodomain. Our finding of 12 distinct mutations in the 5 and 3 coding regions of NKX2.5 in 18 of 608 patients (3%) with congenital cardiovascular anomalies, along with the aforementioned experimental findings, suggests that NKX2.5 mutations outside of the homeodomain, either alone or in conjunction with as-of-yet unidentified modifying factors, may cause structural congenital cardiovascular anomalies without affecting AV conduction. In 8 of the 16 patients with an NKX2.5 mutation, a parental carrier was identified, two of whom were known to have a congenital cardiovascular anomaly. The finding of reduced phenotypic penetrance associated with the NKX2.5 mutations identified in this study is consistent with patterns frequently observed in complex developmental anomalies and may indicate that these mutations are associated with an increased risk of congenital heart disease. We do not believe that the mutations we have detected are random changes, insofar as they were not detected among control subjects or patients with certain diagnoses (e.g., D-transposition of the great arteries). Further analysis of NKX2.5-mediated regulation of cardiovascular development, as well as functional studies of the mutations that have been identified, is necessary to determine the mechanisms by which non-homeodomain NKX2.5 mutations lead to congenital cardiovascular anomalies. Acknowledgment We thank Seigo Izumo, MD, for valuable discussion and commentary on the manuscript.
6 JACC Vol. 42, No. 9, 2003 November 5, 2003: McElhinney et al. NKX2.5 Mutations in CHD 1655 Reprint requests and correspondence: Dr. Elizabeth Goldmuntz, Division of Cardiology, The Children s Hospital of Philadelphia, Abramson Research Center 702A, 3516 Civic Center Blvd., Philadelphia, Pennsylvania goldmuntz@ .chop.edu. REFERENCES 1. Schott JJ, Benson DW, Basson CT, et al. Congenital heart disease caused by mutations in the transcription factor NKX2-5. Science 1998;281: Benson DW, Silberbach GM, Kavanaugh-McHugh A, et al. Mutations in the cardiac transcription factor NKX2.5 affect diverse cardiac developmental pathways. J Clin Invest 1999;104: Hosoda T, Komuro I, Shiojima I, et al. Familial atrial septal defect and atrioventricular conduction disturbance associated with a point mutation in the cardiac homeobox gene CSX/NKX2-5 in a Japanese patient. Jpn Circ J 1999;63: Goldmuntz E, Geiger E, Benson DW. NKX2.5 mutations in patients with tetralogy of Fallot. Circulation 2001;104: Goldmuntz E, Clark BJ, Mitchell LE, et al. Frequency of 22q11 deletions in patients with conotruncal defects. J Am Coll Cardiol 1998;32: Ganguly A, Rock MJ, Prockop DJ. Conformation-sensitive gel electrophoresis for rapid detection of single-base differences in doublestranded PCR products and DNA fragments: evidence for solventinduced bends in DNA heteroduplexes. Proc Natl Acad Sci USA 1993;90: Komuro I, Izumo S. Csx: a murine homeobox-containing gene specifically expressed in the developing heart. Proc Natl Acad Sci USA 1993;90: Harvey RP. NK-2 homeobox genes and heart development. Dev Biol 1996;178: Turbay D, Wechsler SB, Blanchard KM, Izumo S. Molecular cloning, chromosomal mapping, and characterization of the human cardiacspecific homeobox gene hcsx. Mol Med 1996;2: Tanaka M, Kasahara H, Bartunkova S, et al. Vertebrate homologs of tinman and bagpipe: roles of the homeobox genes in cardiovascular development. Dev Genet 1998;22: Azpiazu N, Frasch M. tinman and bagpipe: two homeo box genes that determine cell fates in the dorsal mesoderm of Drosophila. Genes Dev 1993;7: Bodmer R. The gene tinman is required for specification of the heart and visceral muscles in Drosophila. Development 1993;118: Lyons I, Parsons LM, Hartley L, et al. Myogenic and morphogenetic defects in the heart tubes of murine embryos lacking the homeo box gene Nkx2-5. Genes Dev 1995;9: Tanaka M, Chen Z, Bartunkova S, Yamasaki N, Izumo S. The cardiac homeobox gene Csx/NKX2.5 lies genetically upstream of multiple genes essential for heart development. Development 1999;126: Biben C, Weber R, Kesteven S, et al. Cardiac septal and valvular dysmorphogenesis in mice heterozygous for mutations in the homeobox gene Nkx2-5. Circ Res 2000;87: Schwartz RJ, Olson EN. Building the heart piece by piece: modularity of cis-elements regulating Nkx2-5 transcription. Development 1999; 126: Bruneau BG, Bao ZZ, Tanaka M, et al. Cardiac expression of the ventricle-specific homeobox gene Irx4 is modulated by Nkx2-5 and dhand. Dev Biol 2000;217: Hiroi Y, Kudoh S, Monzen K, et al. Tbx5 associates with Nkx2-5 and synergistically promotes cardiomyocyte differentiation. Nat Genet 2001;28: Jamali M, Karamboulas C, Rogerson PJ, Skerjanc IS. BMP signaling regulates Nkx2-5 activity during cardiomyogenesis. FEBS Lett 2001; 509: Yamagishi H, Yamagishi C, Nakagawa O, Harvey RP, Olson EN, Srivastava D. The combinatorial activities of NKX2.5 and dhand are essential for cardiac ventricle formation. Dev Biol 2001;239: Sepulveda JL, Vlahopoulos S, Iyer D, Belaguli N, Schwartz RJ. Combinatorial expression of GATA4, Nkx2-5 and serum response factor directs early cardiac gene activity. J Biol Chem 2002;277: Thompson JT, Rackley MS, O Brien TX. Upregulation of the cardiac homeobox gene Nkx2-5 (CSX) in feline right ventricular pressure overload. Am J Physiol 1998;274:H Kasahara H, Lee B, Schott Benson DW, Seidman JG, Seidman CE, Izumo S. Loss of function and inhibitory effects of human CSX/ NKX2.5 homeoprotein mutations associated with congenital heart disease. J Clin Invest 2000;106: Kasahara H, Usheva A, Ueyama T, Aoki H, Horikoshi N, Izumo S. Characterization of homo- and heterodimerization of cardiac Csx/ NKX2.5 homeoprotein. J Biol Chem 2001;276: Schinke M, Litovsky S, Usheva A, Kasahara H, Aoki H, Izumo S. Lack of the conserved NK2-domain of the cardiac transcription factor NKX2.5 causes multiple heart defects (abstr). Circulation 2001;104 Suppl:II27.
Cardiac Homeobox Gene NKX2-5 Mutations and Congenital Heart Disease Associations With Atrial Septal Defect and Hypoplastic Left Heart Syndrome
 Journal of the American College of Cardiology Vol. 41, No. 11, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/s0735-1097(03)00420-0
Journal of the American College of Cardiology Vol. 41, No. 11, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/s0735-1097(03)00420-0
Heart Development and Congenital Heart Disease
 Heart Development and Congenital Heart Disease Sally Dunwoodie s.dunwoodie@victorchang.edu.au Developmental and Stem Cell Biology Division Victor Chang Cardiac Research Institute for the heart of Australia...
Heart Development and Congenital Heart Disease Sally Dunwoodie s.dunwoodie@victorchang.edu.au Developmental and Stem Cell Biology Division Victor Chang Cardiac Research Institute for the heart of Australia...
Congenital Heart Disease How much of it is genetic?
 Congenital Heart Disease How much of it is genetic? Stephen Robertson Curekids Professor of Paediatric Genetics Dunedin School of Medicine University of Otago Congenital Heart Disease The most common survivable
Congenital Heart Disease How much of it is genetic? Stephen Robertson Curekids Professor of Paediatric Genetics Dunedin School of Medicine University of Otago Congenital Heart Disease The most common survivable
5/22/2013. Alan Zuckerman 1, Swapna Abhyankar 1, Tiffany Colarusso 2, Richard Olney 2, Kristin Burns 3, Marci Sontag 4
 Alan Zuckerman 1, Swapna Abhyankar 1, Tiffany Colarusso 2, Richard Olney 2, Kristin Burns 3, Marci Sontag 4 1 National Library of Medicine, NIH, Bethesda, MD, USA, 2 Centers for Disease Control and Prevention,
Alan Zuckerman 1, Swapna Abhyankar 1, Tiffany Colarusso 2, Richard Olney 2, Kristin Burns 3, Marci Sontag 4 1 National Library of Medicine, NIH, Bethesda, MD, USA, 2 Centers for Disease Control and Prevention,
The Fetal Cardiology Program
 The Fetal Cardiology Program at Texas Children s Fetal Center About the program Since the 1980s, Texas Children s Fetal Cardiology Program has provided comprehensive fetal cardiac care to expecting families
The Fetal Cardiology Program at Texas Children s Fetal Center About the program Since the 1980s, Texas Children s Fetal Cardiology Program has provided comprehensive fetal cardiac care to expecting families
Congenital Heart Defects
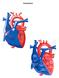 Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Maoqing Ye, Yan Yin, Kazumi Fukatsu, and Paul Grossfeld
 Evidence That Deletion of ETS-1, a Gene in the Jacobsen Syndrome (11q-) Cardiac Critical Region, Causes Congenital Heart Defects through Impaired Cardiac Neural Crest Cell Function 52 Maoqing Ye, Yan Yin,
Evidence That Deletion of ETS-1, a Gene in the Jacobsen Syndrome (11q-) Cardiac Critical Region, Causes Congenital Heart Defects through Impaired Cardiac Neural Crest Cell Function 52 Maoqing Ye, Yan Yin,
"Lecture Index. 1) Heart Progenitors. 2) Cardiac Tube Formation. 3) Valvulogenesis and Chamber Formation. 4) Epicardium Development.
 "Lecture Index 1) Heart Progenitors. 2) Cardiac Tube Formation. 3) Valvulogenesis and Chamber Formation. 4) Epicardium Development. 5) Septation and Maturation. 6) Changes in Blood Flow during Development.
"Lecture Index 1) Heart Progenitors. 2) Cardiac Tube Formation. 3) Valvulogenesis and Chamber Formation. 4) Epicardium Development. 5) Septation and Maturation. 6) Changes in Blood Flow during Development.
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT
 ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
Analysis of Cardiovascular Phenotype and Genotype-Phenotype Correlation in Individuals With a JAG1 Mutation and/or Alagille Syndrome
 Analysis of Cardiovascular Phenotype and Genotype-Phenotype Correlation in Individuals With a Mutation and/or Alagille Syndrome Doff B. McElhinney, MD; Ian D. Krantz, MD; Lynn Bason, MS; David A. Piccoli,
Analysis of Cardiovascular Phenotype and Genotype-Phenotype Correlation in Individuals With a Mutation and/or Alagille Syndrome Doff B. McElhinney, MD; Ian D. Krantz, MD; Lynn Bason, MS; David A. Piccoli,
Cardiac Catheterization Cases Primary Cardiac Diagnoses Facility 12 month period from to PRIMARY DIAGNOSES (one per patient)
 PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
Division of Pediatric Cardiology, Medical University of South Carolina, Charleston, South Carolina 29425, USA 2
 Mutations in the cardiac transcription factor NKX2.5 affect diverse cardiac developmental pathways D. Woodrow Benson, 1 G. Michael Silberbach, 2 Ann Kavanaugh-McHugh, 3 Carol Cottrill, 4 Yizhong Zhang,
Mutations in the cardiac transcription factor NKX2.5 affect diverse cardiac developmental pathways D. Woodrow Benson, 1 G. Michael Silberbach, 2 Ann Kavanaugh-McHugh, 3 Carol Cottrill, 4 Yizhong Zhang,
3/14/2011 MANAGEMENT OF NEWBORNS CARDIAC INTENSIVE CARE CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 WITH HEART DEFECTS
 CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016
 1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
MEDICAL MANAGEMENT WITH CAVEATS 1. In one study of 50 CHARGE patients with CHD, 75% required surgery. 2. Children with CHARGE may be resistant to chlo
 CARDIOLOGY IN CHARGE SYNDROME: FOR THE PHYSICIAN Angela E. Lin, M.D. Teratology Program/Active Malformation Surveillance, Brigham and Women's Hospital, Old PBBH-B501, 75 Francis St., Boston, MA 02115 alin@partners.org
CARDIOLOGY IN CHARGE SYNDROME: FOR THE PHYSICIAN Angela E. Lin, M.D. Teratology Program/Active Malformation Surveillance, Brigham and Women's Hospital, Old PBBH-B501, 75 Francis St., Boston, MA 02115 alin@partners.org
Index. cardiology.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
Heart and Soul Evaluation of the Fetal Heart
 Heart and Soul Evaluation of the Fetal Heart Ivana M. Vettraino, M.D., M.B.A. Clinical Associate Professor, Michigan State University College of Human Medicine Objectives Review the embryology of the formation
Heart and Soul Evaluation of the Fetal Heart Ivana M. Vettraino, M.D., M.B.A. Clinical Associate Professor, Michigan State University College of Human Medicine Objectives Review the embryology of the formation
Transient malformations like PDA and PDA of prematurity were not considered. We have divided cardiac malformations in 2 groups:
 CARDIAC MALFORMATIONS DETECTED AT BIRTH Anwar Dudin-MD, Annie Rambaud-Cousson-MD, Mahmoud Nashashibi-MD Pediatric Department Makassed Hospital Jerusalem Diagnosis of congenital heart disease in the neonatal
CARDIAC MALFORMATIONS DETECTED AT BIRTH Anwar Dudin-MD, Annie Rambaud-Cousson-MD, Mahmoud Nashashibi-MD Pediatric Department Makassed Hospital Jerusalem Diagnosis of congenital heart disease in the neonatal
Outflow Tracts Anomalies
 Diagnosis of Outflow Tract Anomalies in the Fetus General Framing D.Paladini Fetal Medicine & Surgery Unit Gasllini Children s Hospital - Genoa dariopaladini@ospedale-gaslini.ge.it Outflow Tracts Anomalies
Diagnosis of Outflow Tract Anomalies in the Fetus General Framing D.Paladini Fetal Medicine & Surgery Unit Gasllini Children s Hospital - Genoa dariopaladini@ospedale-gaslini.ge.it Outflow Tracts Anomalies
Before we are Born: Fetal Diagnosis of Congenital Heart Disease
 Before we are Born: Fetal Diagnosis of Congenital Heart Disease Mohamed Sulaiman, MD Pediatric cardiologist Kidsheart: American Fetal & Children's Heart Center Dubai Healthcare City, Dubai-UAE First Pediatric
Before we are Born: Fetal Diagnosis of Congenital Heart Disease Mohamed Sulaiman, MD Pediatric cardiologist Kidsheart: American Fetal & Children's Heart Center Dubai Healthcare City, Dubai-UAE First Pediatric
Heart and Lungs. LUNG Coronal section demonstrates relationship of pulmonary parenchyma to heart and chest wall.
 Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Methods PEDIATRIC CARDIOLOGY
 1805 PEDIATRIC CARDIOLOGY Trends and Outcomes After Prenatal Diagnosis of Congenital Cardiac Malformations by Fetal Echocardiography in a Well Defined Birth Population, Atlanta, Georgia, 1990 1994 EDUARDO
1805 PEDIATRIC CARDIOLOGY Trends and Outcomes After Prenatal Diagnosis of Congenital Cardiac Malformations by Fetal Echocardiography in a Well Defined Birth Population, Atlanta, Georgia, 1990 1994 EDUARDO
Screening for Critical Congenital Heart Disease
 Screening for Critical Congenital Heart Disease Caroline K. Lee, MD Pediatric Cardiology Disclosures I have no relevant financial relationships or conflicts of interest 1 Most Common Birth Defect Most
Screening for Critical Congenital Heart Disease Caroline K. Lee, MD Pediatric Cardiology Disclosures I have no relevant financial relationships or conflicts of interest 1 Most Common Birth Defect Most
Children with Single Ventricle Physiology: The Possibilities
 Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
MEDICAL SCIENCES Vol.I -Adult Congenital Heart Disease: A Challenging Population - Khalid Aly Sorour
 ADULT CONGENITAL HEART DISEASE: A CHALLENGING POPULATION Khalid Aly Sorour Cairo University, Kasr elaini Hospital, Egypt Keywords: Congenital heart disease, adult survival, specialized care centers. Contents
ADULT CONGENITAL HEART DISEASE: A CHALLENGING POPULATION Khalid Aly Sorour Cairo University, Kasr elaini Hospital, Egypt Keywords: Congenital heart disease, adult survival, specialized care centers. Contents
UPDATE FETAL ECHO REVIEW
 UPDATE 1 FETAL ECHO REVIEW Study Alert for RDCS Candidates D A V I E S P U B L I S H I N G I N C. Fetal Echo Review Study Alert U P D A T E D A U G U S T 1, 2 0 1 2 Nikki Stahl, RT(R)(M)(CT), RDMS, RVT
UPDATE 1 FETAL ECHO REVIEW Study Alert for RDCS Candidates D A V I E S P U B L I S H I N G I N C. Fetal Echo Review Study Alert U P D A T E D A U G U S T 1, 2 0 1 2 Nikki Stahl, RT(R)(M)(CT), RDMS, RVT
LES NOUVEAUX TROUBLES DE CONDUCTION HEREDITAIRES
 LES NOUVEAUX TROUBLES DE CONDUCTION HEREDITAIRES Alban Baruteau L institut du thorax, INSERM 1087 CNRS 6291 IRS Université de Nantes Centre Chirurgical Marie Lannelongue M3C Université Paris Sud Le Kremlin
LES NOUVEAUX TROUBLES DE CONDUCTION HEREDITAIRES Alban Baruteau L institut du thorax, INSERM 1087 CNRS 6291 IRS Université de Nantes Centre Chirurgical Marie Lannelongue M3C Université Paris Sud Le Kremlin
SEX, BIRTH ORDER, AND MATERNAL AGE CHARACTERISTICS OF INFANTS WITH CONGENITAL HEART DEFECTS
 AMERICAN JOURNAL OF EPIDEMIOLOGY Copyright 1 by The Johns Hopkins University School of Hygiene and Public Health Vol., Xo. Printed in U.S.A. SEX, BIRTH ORDER, AND MATERNAL AGE CHARACTERISTICS OF INFANTS
AMERICAN JOURNAL OF EPIDEMIOLOGY Copyright 1 by The Johns Hopkins University School of Hygiene and Public Health Vol., Xo. Printed in U.S.A. SEX, BIRTH ORDER, AND MATERNAL AGE CHARACTERISTICS OF INFANTS
CYANOTIC CONGENITAL HEART DISEASES. PRESENTER: DR. Myra M. Koech Pediatric cardiologist MTRH/MU
 CYANOTIC CONGENITAL HEART DISEASES PRESENTER: DR. Myra M. Koech Pediatric cardiologist MTRH/MU DEFINITION Congenital heart diseases are defined as structural and functional problems of the heart that are
CYANOTIC CONGENITAL HEART DISEASES PRESENTER: DR. Myra M. Koech Pediatric cardiologist MTRH/MU DEFINITION Congenital heart diseases are defined as structural and functional problems of the heart that are
Keeping Your Heart in Good Shape for a Lifetime
 Keeping Your Heart in Good Shape for a Lifetime Adult Congenital Heart Program Adult Congenital Heart Program Today, children with congenital heart disease are not only surviving but thriving as adults.
Keeping Your Heart in Good Shape for a Lifetime Adult Congenital Heart Program Adult Congenital Heart Program Today, children with congenital heart disease are not only surviving but thriving as adults.
ULTRASOUND OF THE FETAL HEART
 ULTRASOUND OF THE FETAL HEART Cameron A. Manbeian, MD Disclosure Statement Today s faculty: Cameron Manbeian, MD does not have any relevant financial relationships with commercial interests or affiliations
ULTRASOUND OF THE FETAL HEART Cameron A. Manbeian, MD Disclosure Statement Today s faculty: Cameron Manbeian, MD does not have any relevant financial relationships with commercial interests or affiliations
Research article. Primary detection of congenital heart diseases in the Kyrgyz Republic
 Research article Primary detection of congenital heart diseases in the Kyrgyz Republic Irina A. Akhmedova, Gulzada A. Imanalieva, Damir A.Abibillaev, Taalaibek Z. Kudaiberdiev Scientific Research Institute
Research article Primary detection of congenital heart diseases in the Kyrgyz Republic Irina A. Akhmedova, Gulzada A. Imanalieva, Damir A.Abibillaev, Taalaibek Z. Kudaiberdiev Scientific Research Institute
Making Sense of Cardiac Views and Imaging Characteristics for 13 Congenital Heart Defects (CHDs)
 Making Sense of Cardiac Views and Imaging Characteristics for 13 Congenital Heart Defects (CHDs) Manny Gaziano, MD, FACOG obimages.net obimages.net@gmail.com Acknowledgements: Krista Wald, RDMS, sonographer,
Making Sense of Cardiac Views and Imaging Characteristics for 13 Congenital Heart Defects (CHDs) Manny Gaziano, MD, FACOG obimages.net obimages.net@gmail.com Acknowledgements: Krista Wald, RDMS, sonographer,
Frequency of 22q11 Deletions in Patients With Conotruncal Defects
 492 JACC Vol. 32, No. 2 PEDIATRIC CARDIOLOGY Frequency of 22q11 Deletions in Patients With Conotruncal Defects ELIZABETH GOLDMUNTZ, MD, FACC,* BERNARD J. CLARK, MD, FACC,* LAURA E. MITCHELL, PHD, ABBAS
492 JACC Vol. 32, No. 2 PEDIATRIC CARDIOLOGY Frequency of 22q11 Deletions in Patients With Conotruncal Defects ELIZABETH GOLDMUNTZ, MD, FACC,* BERNARD J. CLARK, MD, FACC,* LAURA E. MITCHELL, PHD, ABBAS
Nkx2-5 transcription. REVIEW ARTICLE Building the heart piece by piece: modularity of cis-elements regulating
 Development 126, 4187-4192 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV3037 4187 REVIEW ARTICLE Building the heart piece by piece: modularity of cis-elements regulating Nkx2-5
Development 126, 4187-4192 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV3037 4187 REVIEW ARTICLE Building the heart piece by piece: modularity of cis-elements regulating Nkx2-5
CONGENITAL HEART DISEASE (CHD)
 CONGENITAL HEART DISEASE (CHD) DEFINITION It is the result of a structural or functional abnormality of the cardiovascular system at birth GENERAL FEATURES OF CHD Structural defects due to specific disturbance
CONGENITAL HEART DISEASE (CHD) DEFINITION It is the result of a structural or functional abnormality of the cardiovascular system at birth GENERAL FEATURES OF CHD Structural defects due to specific disturbance
Accuracy of the Fetal Echocardiogram in Double-outlet Right Ventricle
 Blackwell Publishing IncMalden, USACHDCongenital Heart Disease 2006 The Authors; Journal compilation 2006 Blackwell Publishing, Inc.? 200723237Original ArticleFetal Echocardiogram in Double-outlet Right
Blackwell Publishing IncMalden, USACHDCongenital Heart Disease 2006 The Authors; Journal compilation 2006 Blackwell Publishing, Inc.? 200723237Original ArticleFetal Echocardiogram in Double-outlet Right
Pathological physiology of cardiovascular system Congenital heart diseases
 Pathological physiology of cardiovascular system Congenital heart diseases Rácz Oliver, Sedláková Eva Institute of Pathological Physiology, Medical School, P.J. Šafárik University Oliver Rácz, Eva Sedláková
Pathological physiology of cardiovascular system Congenital heart diseases Rácz Oliver, Sedláková Eva Institute of Pathological Physiology, Medical School, P.J. Šafárik University Oliver Rácz, Eva Sedláková
Supplemental Information
 ARTICLE Supplemental Information SUPPLEMENTAL TABLE 6 Mosaic and Partial Trisomies Thirty-eight VLBW infants were identified with T13, of whom 2 had mosaic T13. T18 was reported for 128 infants, of whom
ARTICLE Supplemental Information SUPPLEMENTAL TABLE 6 Mosaic and Partial Trisomies Thirty-eight VLBW infants were identified with T13, of whom 2 had mosaic T13. T18 was reported for 128 infants, of whom
Slide 1. Slide 2. Slide 3 CONGENITAL HEART DISEASE. Papworth Hospital NHS Trust INTRODUCTION. Jakub Kadlec/Catherine Sudarshan INTRODUCTION
 Slide 1 CONGENITAL HEART DISEASE Jakub Kadlec/Catherine Sudarshan NHS Trust Slide 2 INTRODUCTION Most common congenital illness in the newborn Affects about 4 9 / 1000 full-term live births in the UK 1.5
Slide 1 CONGENITAL HEART DISEASE Jakub Kadlec/Catherine Sudarshan NHS Trust Slide 2 INTRODUCTION Most common congenital illness in the newborn Affects about 4 9 / 1000 full-term live births in the UK 1.5
Pediatric Echocardiography Examination Content Outline
 Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Potential Data Sources
 Potential Data Sources Addressing Congenital Heart Defects: Occurrence, Populations and Risk Factors Centers for Disease Control and Prevention (CDC) Birth Defects Data and Statistics http://www.cdc.gov/ncbddd/birthdefects/data.html
Potential Data Sources Addressing Congenital Heart Defects: Occurrence, Populations and Risk Factors Centers for Disease Control and Prevention (CDC) Birth Defects Data and Statistics http://www.cdc.gov/ncbddd/birthdefects/data.html
Epidemiology of congenital heart diseases
 Epidemiology of congenital heart diseases Damien Bonnet Unité médico-chirurgicale de Cardiologie Congénitale et Pédiatrique Hôpital Universitaire Necker Enfants malades APHP, Université Paris Descartes,
Epidemiology of congenital heart diseases Damien Bonnet Unité médico-chirurgicale de Cardiologie Congénitale et Pédiatrique Hôpital Universitaire Necker Enfants malades APHP, Université Paris Descartes,
Diagnosis of Congenital Cardiac Defects Between 11 and 14 Weeks Gestation in High-Risk Patients
 Article Diagnosis of Congenital Cardiac Defects Between 11 and 14 Weeks Gestation in High-Risk Patients Zeev Weiner, MD, Abraham Lorber, MD, Eliezer Shalev, MD Objective. To examine the feasibility of
Article Diagnosis of Congenital Cardiac Defects Between 11 and 14 Weeks Gestation in High-Risk Patients Zeev Weiner, MD, Abraham Lorber, MD, Eliezer Shalev, MD Objective. To examine the feasibility of
By Dickens ATURWANAHO & ORIBA DAN LANGOYA MAKchs, MBchB CONGENTAL HEART DISEASE
 By Dickens ATURWANAHO & ORIBA DAN LANGOYA MAKchs, MBchB CONGENTAL HEART DISEASE Introduction CHDs are abnormalities of the heart or great vessels that are present at birth. Common type of heart disease
By Dickens ATURWANAHO & ORIBA DAN LANGOYA MAKchs, MBchB CONGENTAL HEART DISEASE Introduction CHDs are abnormalities of the heart or great vessels that are present at birth. Common type of heart disease
September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical)
 September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical) Advances in cardiac surgery have created a new population of adult patients with repaired congenital heart
September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical) Advances in cardiac surgery have created a new population of adult patients with repaired congenital heart
Regional Prenatal Congenital Heart Disease Detection and Practices Lori Erickson MSN, RN, CPNP-PC Ward Family Heart Center
 Regional Prenatal Congenital Heart Disease Detection and Practices Lori Erickson MSN, RN, CPNP-PC Ward Family Heart Center The Children's Mercy Hospital, 2014. 05/14 Objectives Evaluate our regional prenatal
Regional Prenatal Congenital Heart Disease Detection and Practices Lori Erickson MSN, RN, CPNP-PC Ward Family Heart Center The Children's Mercy Hospital, 2014. 05/14 Objectives Evaluate our regional prenatal
Congenital Heart Defects
 C H A P T E R 46 Congenital Heart Defects Rocio Moran Department of Pediatric Genetics, Cleveland Clinic Lerner College of Medicine, Cleveland, OH, USA Nathaniel H Robin Department of Genetics and Pediatrics,
C H A P T E R 46 Congenital Heart Defects Rocio Moran Department of Pediatric Genetics, Cleveland Clinic Lerner College of Medicine, Cleveland, OH, USA Nathaniel H Robin Department of Genetics and Pediatrics,
A SURGEONS' GUIDE TO CARDIAC DIAGNOSIS
 A SURGEONS' GUIDE TO CARDIAC DIAGNOSIS PART II THE CLINICAL PICTURE DONALD N. ROSS B. Sc., M. B., CH. B., F. R. C. S. CONSULTANT THORACIC SURGEON GUY'S HOSPITAL, LONDON WITH 53 FIGURES Springer-Verlag
A SURGEONS' GUIDE TO CARDIAC DIAGNOSIS PART II THE CLINICAL PICTURE DONALD N. ROSS B. Sc., M. B., CH. B., F. R. C. S. CONSULTANT THORACIC SURGEON GUY'S HOSPITAL, LONDON WITH 53 FIGURES Springer-Verlag
Presentation of congenital heart disease in infancy: implications for routine examination
 Arch Dis Child Fetal Neonatal Ed 999;80:F49 F5 F49 Presentation of congenital heart disease in infancy: implications for routine examination Christopher Wren, Sam Richmond, Liam Donaldson Department of
Arch Dis Child Fetal Neonatal Ed 999;80:F49 F5 F49 Presentation of congenital heart disease in infancy: implications for routine examination Christopher Wren, Sam Richmond, Liam Donaldson Department of
Assessing Cardiac Anatomy With Digital Subtraction Angiography
 485 JACC Vol. 5, No. I Assessing Cardiac Anatomy With Digital Subtraction Angiography DOUGLAS S., MD, FACC Cleveland, Ohio The use of intravenous digital subtraction angiography in the assessment of patients
485 JACC Vol. 5, No. I Assessing Cardiac Anatomy With Digital Subtraction Angiography DOUGLAS S., MD, FACC Cleveland, Ohio The use of intravenous digital subtraction angiography in the assessment of patients
Dear Parent/Guardian,
 Dear Parent/Guardian, You have indicated on school records that your child has an ongoing health problem that may require medication and/or treatment during the school day with rescue medication. Attached
Dear Parent/Guardian, You have indicated on school records that your child has an ongoing health problem that may require medication and/or treatment during the school day with rescue medication. Attached
Low-dose prospective ECG-triggering dual-source CT angiography in infants and children with complex congenital heart disease: first experience
 Low-dose prospective ECG-triggering dual-source CT angiography in infants and children with complex congenital heart disease: first experience Ximing Wang, M.D., Zhaoping Cheng, M.D., Dawei Wu, M.D., Lebin
Low-dose prospective ECG-triggering dual-source CT angiography in infants and children with complex congenital heart disease: first experience Ximing Wang, M.D., Zhaoping Cheng, M.D., Dawei Wu, M.D., Lebin
Regional Prenatal Congenital Heart Disease Detection and Practices Jenny Ecord, APRN Ward Family Heart Center Wichita
 Regional Prenatal Congenital Heart Disease Detection and Practices Jenny Ecord, APRN Ward Family Heart Center Wichita The Children's Mercy Hospital, 2014. 05/14 Objectives Review current local and regional
Regional Prenatal Congenital Heart Disease Detection and Practices Jenny Ecord, APRN Ward Family Heart Center Wichita The Children's Mercy Hospital, 2014. 05/14 Objectives Review current local and regional
Absent Pulmonary Valve Syndrome
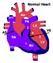 Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Introduction. Pediatric Cardiology. General Appearance. Tools of Assessment. Auscultation. Vital Signs
 Introduction Pediatric Cardiology An introduction to the pediatric patient with heart disease: M-III Lecture Douglas R. Allen, M.D. Assistant Professor and Director of Community Pediatric Cardiology at
Introduction Pediatric Cardiology An introduction to the pediatric patient with heart disease: M-III Lecture Douglas R. Allen, M.D. Assistant Professor and Director of Community Pediatric Cardiology at
DEVELOPMENT OF THE CIRCULATORY SYSTEM L E C T U R E 5
 DEVELOPMENT OF THE CIRCULATORY SYSTEM L E C T U R E 5 REVIEW OF CARDIAC ANATOMY Heart 4 chambers Base and apex Valves Pericardial sac 3 layers: epi, myo, endo cardium Major blood vessels Aorta and its
DEVELOPMENT OF THE CIRCULATORY SYSTEM L E C T U R E 5 REVIEW OF CARDIAC ANATOMY Heart 4 chambers Base and apex Valves Pericardial sac 3 layers: epi, myo, endo cardium Major blood vessels Aorta and its
Segmental Analysis. Gautam K. Singh, M.D. Washington University School of Medicine St. Louis
 Segmental Analysis Gautam K. Singh, M.D. Washington University School of Medicine St. Louis Segmental Analysis Segmental Analysis: From Veins to Ventricles Segmental Approach to Evaluation of Congenital
Segmental Analysis Gautam K. Singh, M.D. Washington University School of Medicine St. Louis Segmental Analysis Segmental Analysis: From Veins to Ventricles Segmental Approach to Evaluation of Congenital
When is Risky to Apply Oxygen for Congenital Heart Disease 부천세종병원 소아청소년과최은영
 When is Risky to Apply Oxygen for Congenital Heart Disease 부천세종병원 소아청소년과최은영 The Korean Society of Cardiology COI Disclosure Eun-Young Choi The author have no financial conflicts of interest to disclose
When is Risky to Apply Oxygen for Congenital Heart Disease 부천세종병원 소아청소년과최은영 The Korean Society of Cardiology COI Disclosure Eun-Young Choi The author have no financial conflicts of interest to disclose
DIAGNOSIS, MANAGEMENT AND OUTCOME OF HEART DISEASE IN SUDANESE PATIENTS
 434 E AST AFRICAN MEDICAL JOURNAL September 2007 East African Medical Journal Vol. 84 No. 9 September 2007 DIAGNOSIS, MANAGEMENT AND OUTCOME OF CONGENITAL HEART DISEASE IN SUDANESE PATIENTS K.M.A. Sulafa,
434 E AST AFRICAN MEDICAL JOURNAL September 2007 East African Medical Journal Vol. 84 No. 9 September 2007 DIAGNOSIS, MANAGEMENT AND OUTCOME OF CONGENITAL HEART DISEASE IN SUDANESE PATIENTS K.M.A. Sulafa,
9/8/2009 < 1 1,2 3,4 5,6 7,8 9,10 11,12 13,14 15,16 17,18 > 18. Tetralogy of Fallot. Complex Congenital Heart Disease.
 Current Indications for Pediatric CTA S Bruce Greenberg Professor of Radiology Arkansas Children s Hospital University of Arkansas for Medical Sciences greenbergsbruce@uams.edu 45 40 35 30 25 20 15 10
Current Indications for Pediatric CTA S Bruce Greenberg Professor of Radiology Arkansas Children s Hospital University of Arkansas for Medical Sciences greenbergsbruce@uams.edu 45 40 35 30 25 20 15 10
Congenital Heart Disease: Physiology and Common Defects
 Congenital Heart Disease: Physiology and Common Defects Jamie S. Sutherell, M.D, M.Ed. Associate Professor, Pediatrics Division of Cardiology Director, Medical Student Education in Pediatrics Director,
Congenital Heart Disease: Physiology and Common Defects Jamie S. Sutherell, M.D, M.Ed. Associate Professor, Pediatrics Division of Cardiology Director, Medical Student Education in Pediatrics Director,
Recent technical advances and increasing experience
 Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Burden of congenital heart diseases in a tertiary cardiac care institute in Western India: Need for a national registry
 Original Article Burden of congenital heart diseases in a tertiary cardiac care institute in Western India: Need for a national registry ABSTRACT Objective: Congenital heart disease (CHD) is very common
Original Article Burden of congenital heart diseases in a tertiary cardiac care institute in Western India: Need for a national registry ABSTRACT Objective: Congenital heart disease (CHD) is very common
Heart Development. Robert G. Kelly Developmental Biology Institute of Marseilles - Luminy
 ESC CBCS Summer School on Cardiovascular Sciences 15th June 2011 Heart Development Robert G. Kelly Developmental Biology Institute of Marseilles - Luminy Animal models of heart development Tinman/Nkx2.5
ESC CBCS Summer School on Cardiovascular Sciences 15th June 2011 Heart Development Robert G. Kelly Developmental Biology Institute of Marseilles - Luminy Animal models of heart development Tinman/Nkx2.5
(2013 ) ACHD ACHD
 (0 ). ) ) ) ) ) ) (ACHD) / ( ) ACHD ACHD 0 6 (9 ) 9 8 8 6 0 6 / ACHD ACHD Adult congenital heart disease, Pediatric cardiologists, Adult cardiologists, Emergency admission, Transfer of the patients (congenital
(0 ). ) ) ) ) ) ) (ACHD) / ( ) ACHD ACHD 0 6 (9 ) 9 8 8 6 0 6 / ACHD ACHD Adult congenital heart disease, Pediatric cardiologists, Adult cardiologists, Emergency admission, Transfer of the patients (congenital
Pulmonary Valve Morphology in Patients with Bicuspid Aortic Valves
 https://doi.org/10.1007/s00246-018-1807-x ORIGINAL ARTICLE Pulmonary Valve Morphology in Patients with Bicuspid Aortic Valves Wilke M. C. Koenraadt 1 Margot M. Bartelings 2 Adriana C. Gittenberger de Groot
https://doi.org/10.1007/s00246-018-1807-x ORIGINAL ARTICLE Pulmonary Valve Morphology in Patients with Bicuspid Aortic Valves Wilke M. C. Koenraadt 1 Margot M. Bartelings 2 Adriana C. Gittenberger de Groot
Genome - Wide Linkage Mapping
 Biological Sciences Initiative HHMI Genome - Wide Linkage Mapping Introduction This activity is based on the work of Dr. Christine Seidman et al that was published in Circulation, 1998, vol 97, pgs 2043-2048.
Biological Sciences Initiative HHMI Genome - Wide Linkage Mapping Introduction This activity is based on the work of Dr. Christine Seidman et al that was published in Circulation, 1998, vol 97, pgs 2043-2048.
Multimodality Imaging of Septal Defects
 Multimodality Imaging of Septal Defects Ohio-ACC 2018 Annual Meeting October 27, 2018 Kan N. Hor, MD Director, Cardiac Magnetic Resonance Imaging Associate Professor of Pediatrics The Heart Center, Nationwide
Multimodality Imaging of Septal Defects Ohio-ACC 2018 Annual Meeting October 27, 2018 Kan N. Hor, MD Director, Cardiac Magnetic Resonance Imaging Associate Professor of Pediatrics The Heart Center, Nationwide
Anomalous Systemic Venous Connection Systemic venous anomaly
 World Database for Pediatric and Congenital Heart Surgery Appendix B: Diagnosis (International Paediatric and Congenital Cardiac Codes (IPCCC) and definitions) Anomalous Systemic Venous Connection Systemic
World Database for Pediatric and Congenital Heart Surgery Appendix B: Diagnosis (International Paediatric and Congenital Cardiac Codes (IPCCC) and definitions) Anomalous Systemic Venous Connection Systemic
Summary. HVRA s Cardio Vascular Genetic Detailed L2 Obstetrical Ultrasound. CPT 76811, 76825, _ 90% CHD detection. _ 90% DS detection.
 What is the role of fetal echocardiography (2D 76825, cardiovascular color flow mapping 93325) as performed in conjunction with detailed fetal anatomy scan (CPT 76811) now that AIUM requires limited outflow
What is the role of fetal echocardiography (2D 76825, cardiovascular color flow mapping 93325) as performed in conjunction with detailed fetal anatomy scan (CPT 76811) now that AIUM requires limited outflow
Cardiology Competency Based Goals and Objectives
 Cardiology Competency Based Goals and Objectives COMPETENCY 1. Patient Care. Provide family centered patient care that is developmentally and age appropriate, compassionate, and effective for the treatment
Cardiology Competency Based Goals and Objectives COMPETENCY 1. Patient Care. Provide family centered patient care that is developmentally and age appropriate, compassionate, and effective for the treatment
Changing Profile of Adult Congenital Heart Disease
 Congenital Heart Disease New Developments for the General Cardiologist Changing Profile of Adult Congenital Heart Disease European Society of Cardiology August 27, 2012 Ariane Marelli MD, FRCP, FACC, MPH
Congenital Heart Disease New Developments for the General Cardiologist Changing Profile of Adult Congenital Heart Disease European Society of Cardiology August 27, 2012 Ariane Marelli MD, FRCP, FACC, MPH
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions
 Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Fetal Tetralogy of Fallot
 36 Fetal Tetralogy of Fallot E.D. Bespalova, R.M. Gasanova, O.A.Pitirimova National Scientific and Practical Center of Cardiovascular Surgery, Moscow Elena D. Bespalova, MD Professor, Director Rena M,
36 Fetal Tetralogy of Fallot E.D. Bespalova, R.M. Gasanova, O.A.Pitirimova National Scientific and Practical Center of Cardiovascular Surgery, Moscow Elena D. Bespalova, MD Professor, Director Rena M,
Born Blue. Anesthesia and CHD. Kristine Faust, CRNA, MS, MBA, DNAP
 Born Blue Anesthesia and CHD Kristine Faust, CRNA, MS, MBA, DNAP Disclosures Disclosures None to Report Objectives Review all congenital defects in which the patient is blue Describe physiology of the
Born Blue Anesthesia and CHD Kristine Faust, CRNA, MS, MBA, DNAP Disclosures Disclosures None to Report Objectives Review all congenital defects in which the patient is blue Describe physiology of the
Congenital Heart Disease
 Congenital Heart Disease Mohammed Alghamdi, MD, FRCPC, FAAP, FACC Associate Professor and Consultant Pediatric Cardiology, Cardiac Science King Fahad Cardiac Centre King Saud University INTRODUCTION CHD
Congenital Heart Disease Mohammed Alghamdi, MD, FRCPC, FAAP, FACC Associate Professor and Consultant Pediatric Cardiology, Cardiac Science King Fahad Cardiac Centre King Saud University INTRODUCTION CHD
Pattern of Congenital Heart Disease A Hospital-Based Study *Sadiq Mohammed Al-Hamash MBChB, FICMS
 Pattern of Congenital Heart Disease A Hospital-Based Study *Sadiq Mohammed Al-Hamash MBChB, FICMS ABSTRACT Background: The congenital heart disease occurs in 0,8% of live births and they have a wide spectrum
Pattern of Congenital Heart Disease A Hospital-Based Study *Sadiq Mohammed Al-Hamash MBChB, FICMS ABSTRACT Background: The congenital heart disease occurs in 0,8% of live births and they have a wide spectrum
Heart Development. Origins of congenital heart defects Properties of cardiac progenitor cells. Robert G. Kelly
 ESC CBCS Summer School on Cardiovascular Sciences Heart Development 19th June 2013 Origins of congenital heart defects Properties of cardiac progenitor cells Robert G. Kelly Animal models of heart development
ESC CBCS Summer School on Cardiovascular Sciences Heart Development 19th June 2013 Origins of congenital heart defects Properties of cardiac progenitor cells Robert G. Kelly Animal models of heart development
The most frequent c.239a>g SNP of NKX2.5 is not involved in Congenital Heart Disease
 The most frequent c.239a>g SNP of NKX2.5 is not involved in Congenital Heart Disease S. M. Dinesh, Kusuma L, and N. B. Ramachandra Genomics Laboratory, Department of Studies in Zoology, University of Mysore,
The most frequent c.239a>g SNP of NKX2.5 is not involved in Congenital Heart Disease S. M. Dinesh, Kusuma L, and N. B. Ramachandra Genomics Laboratory, Department of Studies in Zoology, University of Mysore,
Advanced Congenital Cardiac Morphology. Sunday, Oct. 22, to Thursday, Oct. 26, Learn more: chop.cloud-cme.com
 Advanced Congenital Cardiac Morphology Sunday, Oct. 22, to Thursday, Oct. 26, 2017 Ruth and Tristram Colket, Jr. Translational Research Building Learn more: chop.cloud-cme.com 1 Course Information Course
Advanced Congenital Cardiac Morphology Sunday, Oct. 22, to Thursday, Oct. 26, 2017 Ruth and Tristram Colket, Jr. Translational Research Building Learn more: chop.cloud-cme.com 1 Course Information Course
CCHD Screening with Pulse Oximetry: A Success Story!
 CCHD Screening with Pulse Oximetry: A Success Story! Nicole Spillane, MD Associate Director of Neonatology, HackensackUMC Hackensack Meridian Health Objective Recognize the contribution of pulse oximetry
CCHD Screening with Pulse Oximetry: A Success Story! Nicole Spillane, MD Associate Director of Neonatology, HackensackUMC Hackensack Meridian Health Objective Recognize the contribution of pulse oximetry
How to Recognize a Suspected Cardiac Defect in the Neonate
 Neonatal Nursing Education Brief: How to Recognize a Suspected Cardiac Defect in the Neonate https://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/
Neonatal Nursing Education Brief: How to Recognize a Suspected Cardiac Defect in the Neonate https://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/
Echocardiography in Adult Congenital Heart Disease
 Echocardiography in Adult Congenital Heart Disease Michael Vogel Kinderherz-Praxis München CHD missed in childhood Subsequent lesions after repaired CHD Follow-up of cyanotic heart disease CHD missed in
Echocardiography in Adult Congenital Heart Disease Michael Vogel Kinderherz-Praxis München CHD missed in childhood Subsequent lesions after repaired CHD Follow-up of cyanotic heart disease CHD missed in
A Search For Genetic Modifiers Responsible For Congenital Heart Disease Variability In The Presence Of Nkx2-5 Haploinsufficiency
 Washington University in St. Louis Washington University Open Scholarship All Theses and Dissertations (ETDs) 1-15-2010 A Search For Genetic Modifiers Responsible For Congenital Heart Disease Variability
Washington University in St. Louis Washington University Open Scholarship All Theses and Dissertations (ETDs) 1-15-2010 A Search For Genetic Modifiers Responsible For Congenital Heart Disease Variability
Fetal Growth Among Infants With Congenital Heart Defects by Maternal Race/Ethnicity
 BRIEF COMMUNICATION Fetal Growth Among Infants With Congenital Heart Defects by Maternal Race/Ethnicity WENDY N. NEMBHARD, PHD, AND MELISSA L. LOSCALZO, MD PURPOSE: Congenital heart defects (CHDs) are
BRIEF COMMUNICATION Fetal Growth Among Infants With Congenital Heart Defects by Maternal Race/Ethnicity WENDY N. NEMBHARD, PHD, AND MELISSA L. LOSCALZO, MD PURPOSE: Congenital heart defects (CHDs) are
Prevalence of Secundum Atrial Septal Defect and Associated Findings
 Prevalence of Secundum Atrial Septal Defect and Associated Findings 3 Mark D. Reller Oregon Health & Science University, USA 1. Introduction The goal of this chapter is to review data regarding the current
Prevalence of Secundum Atrial Septal Defect and Associated Findings 3 Mark D. Reller Oregon Health & Science University, USA 1. Introduction The goal of this chapter is to review data regarding the current
5.8 Congenital Heart Disease
 5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
Cardiology Fellowship Manual. Goals & Objectives -Cardiac Imaging- 1 P a g e
 Cardiology Fellowship Manual Goals & Objectives -Cardiac Imaging- 1 P a g e UNIV. OF NEBRASKA CHILDREN S HOSPITAL & MEDICAL CENTER DIVISION OF CARDIOLOGY FELLOWSHIP PROGRAM CARDIAC IMAGING ROTATION GOALS
Cardiology Fellowship Manual Goals & Objectives -Cardiac Imaging- 1 P a g e UNIV. OF NEBRASKA CHILDREN S HOSPITAL & MEDICAL CENTER DIVISION OF CARDIOLOGY FELLOWSHIP PROGRAM CARDIAC IMAGING ROTATION GOALS
Pattern of Congenital Heart Disease in Jordan
 Pattern of Congenital Heart Disease in Jordan Khaled Amro Department of Pediatrics at Prince Hashim military hospital in Jordan- Zarka Eur J Gen Med 2009; 6(3): 161-165 ABSTRACT Aim: Congenital heart disease
Pattern of Congenital Heart Disease in Jordan Khaled Amro Department of Pediatrics at Prince Hashim military hospital in Jordan- Zarka Eur J Gen Med 2009; 6(3): 161-165 ABSTRACT Aim: Congenital heart disease
Congenital Heart Disease
 Screening Programmes Fetal Anomaly Congenital Heart Disease Information for health professionals Publication date: April 2012 Review date: April 2013 Version 2 67 Congenital Heart Disease Information for
Screening Programmes Fetal Anomaly Congenital Heart Disease Information for health professionals Publication date: April 2012 Review date: April 2013 Version 2 67 Congenital Heart Disease Information for
CASE REPORT: DOUBLE ORIFICE MITRAL VALVE WITH CLEFT IN ANTERIOR LEAFLET OF DOMINANT VALVE IN AN AFRO-CARIBBEAN
 CASE REPORT: DOUBLE ORIFICE MITL VAE WITH CLEFT IN ANTERIOR LEAFLET OF DOMINANT VAE IN AN AFRO-CARIBBEAN Disclosure: No potential conflict of interest. Received: 27.08.13 Accepted: 23.06.14 Citation: EMJ
CASE REPORT: DOUBLE ORIFICE MITL VAE WITH CLEFT IN ANTERIOR LEAFLET OF DOMINANT VAE IN AN AFRO-CARIBBEAN Disclosure: No potential conflict of interest. Received: 27.08.13 Accepted: 23.06.14 Citation: EMJ
Adult Congenital Heart Disease: The Scope of the Problem
 Adult Congenital Heart Disease: The Scope of the Problem Elizabeth E. Adams, DO Children s Heart Center Nevada Program for Adult Congenital Cardiology Congenital Heart Disease u Recognized for centuries
Adult Congenital Heart Disease: The Scope of the Problem Elizabeth E. Adams, DO Children s Heart Center Nevada Program for Adult Congenital Cardiology Congenital Heart Disease u Recognized for centuries
Foetal Cardiology: How to predict perinatal problems. Prof. I.Witters Prof.M.Gewillig UZ Leuven
 Foetal Cardiology: How to predict perinatal problems Prof. I.Witters Prof.M.Gewillig UZ Leuven Cardiopathies Incidence : 8-12 / 1000 births ( 1% ) Most frequent - Ventricle Septum Defect 20% - Atrium Septum
Foetal Cardiology: How to predict perinatal problems Prof. I.Witters Prof.M.Gewillig UZ Leuven Cardiopathies Incidence : 8-12 / 1000 births ( 1% ) Most frequent - Ventricle Septum Defect 20% - Atrium Septum
Long Term outcomes after surgical interventions for Congenital Heart Disease (CHD)
 Long Term outcomes after surgical interventions for Congenital Heart Disease (CHD) Lazaros Kochilas, MD, MSCR, FAAP Professor of Pediatrics Director of Clinical Research The shaping of the field of Pediatric
Long Term outcomes after surgical interventions for Congenital Heart Disease (CHD) Lazaros Kochilas, MD, MSCR, FAAP Professor of Pediatrics Director of Clinical Research The shaping of the field of Pediatric
Hypoplastic Left Heart Syndrome: Echocardiographic Assessment
 Hypoplastic Left Heart Syndrome: Echocardiographic Assessment Craig E Fleishman, MD, FACC, FASE Director, Non-invasive Cardiac Imaging The Hear Center at Arnold Palmer Hospital for Children, Orlando SCAI
Hypoplastic Left Heart Syndrome: Echocardiographic Assessment Craig E Fleishman, MD, FACC, FASE Director, Non-invasive Cardiac Imaging The Hear Center at Arnold Palmer Hospital for Children, Orlando SCAI
Common Defects With Expected Adult Survival:
 Common Defects With Expected Adult Survival: Bicuspid aortic valve :Acyanotic Mitral valve prolapse Coarctation of aorta Pulmonary valve stenosis Atrial septal defect Patent ductus arteriosus (V.S.D.)
Common Defects With Expected Adult Survival: Bicuspid aortic valve :Acyanotic Mitral valve prolapse Coarctation of aorta Pulmonary valve stenosis Atrial septal defect Patent ductus arteriosus (V.S.D.)
Advanced Congenital Cardiac Morphology
 Advanced Congenital Cardiac Morphology October 18 22, 2015 Ruth and Tristram Colket, Jr. Translational Research Building at The Children s Hospital of Philadelphia Philadelphia, Pa. www.chop.edu/cme Program:
Advanced Congenital Cardiac Morphology October 18 22, 2015 Ruth and Tristram Colket, Jr. Translational Research Building at The Children s Hospital of Philadelphia Philadelphia, Pa. www.chop.edu/cme Program:
Congenitally Corrected Transposition of the Great Arteries (cctga or l-loop TGA)
 Congenitally Corrected Transposition of the Great Arteries (cctga or l-loop TGA) Mary Rummell, MN, RN, CPNP, CNS Clinical Nurse Specialist, Pediatric Cardiology/Cardiac Surgery Doernbecher Children s Hospital,
Congenitally Corrected Transposition of the Great Arteries (cctga or l-loop TGA) Mary Rummell, MN, RN, CPNP, CNS Clinical Nurse Specialist, Pediatric Cardiology/Cardiac Surgery Doernbecher Children s Hospital,
МОРФОЛОГИЯ ВРОЖДЕННЫХ ПОРОКОВ СЕРДЦА Бабий Леся Николаевна, Сосамма Йоханнан Сану Харьковский национальный медицинский университет Украина, Харьков
 МОРФОЛОГИЯ ВРОЖДЕННЫХ ПОРОКОВ СЕРДЦА Бабий Леся Николаевна, Сосамма Йоханнан Сану Харьковский национальный медицинский университет Украина, Харьков.MORPHOLOGY OF CONGENITAL HEART DEFECT Babiy L.,Sanu.S.Yohannan
МОРФОЛОГИЯ ВРОЖДЕННЫХ ПОРОКОВ СЕРДЦА Бабий Леся Николаевна, Сосамма Йоханнан Сану Харьковский национальный медицинский университет Украина, Харьков.MORPHOLOGY OF CONGENITAL HEART DEFECT Babiy L.,Sanu.S.Yohannan
