UNIVERSITY OF CINCINNATI
|
|
|
- Annabel Kelly
- 5 years ago
- Views:
Transcription
1 UNIVERSITY OF CINCINNATI Date: 5-Nov-2010 I, Justin Niehaus, hereby submit this original work as part of the requirements for the degree of: Master of Science in Aerospace Engineering It is entitled: Use of Computational Fluid Dynamics to Evaluate Energy Loss in Three Palliative Strategies of Hypoplastic Left Heart Syndrome Student Signature: Justin Niehaus This work and its defense approved by: Committee Chair: Shaaban Abdallah, PhD Shaaban Abdallah, PhD Grant Schaffner, PhD Grant Schaffner, PhD Jeffrey Shuhaiber, MD Jeffrey Shuhaiber, MD 11/12/2010 1,156
2 Use of Computational Fluid Dynamics to Evaluate Energy Loss in Three Palliative Strategies of Hypoplastic Left Heart Syndrome In partial fulfillment of requirements for the degree of Master of Science In the Department of Aerospace Engineering Of the College of Engineering 2010 By Justin Niehaus Committee: Dr. Shaaban A. Abdallah(Chair) Dr. Jeffrey Shuhaiber Dr. Grant Schaffner Previous Degree: B.S. in Aerospace Engineering, University of Cincinnati
3 Abstract Hypoplastic Left Heart Syndrome is a congenital disease that affects neonates and leaves them with an under developed left ventricle. There are three palliative strategies for correcting this disease, which all involve redirecting the blood flow of the heart. In this study, computational fluid dynamics will be used to determine the energy loss in each of the palliative strategies. The losses of the three strategies will be compared to each other, along with the shear stress on the walls. The volumetric flow rate of the systemic and pulmonary circulatory systems of each palliative strategy will also be evaluated. In addition, the differences in systemic and diastolic losses will be compared.
4
5 Acknowledgement I would like to thank my committee chair, Dr. Abdallah for presenting this interesting project to me and guiding me through to the finish. Also, my adviser from the medical side, Dr. Shuhaiber was excellent in teaching me all about the heart and being available to help out in any way. I would like to thank my third committee member, Dr. Schaffner for all the teachings he has presented me in my undergraduate career, and guiding me to being a professional engineer. I would also like to thank my friends and family for being supportive throughout this whole process.
6
7 TABLE OF CONTENTS Nomenclature 1. Introduction 1.1 Hypoplastic Left Heart Syndrome 1.2 Palliated Strategies 1.3 Using CFD to model the heart 2. Background 2.1 The Healthy Heart 2.2 The Circulatory system 2.3 Hypoplastic Left Heart Syndrome 3. CFD Modeling 3.1 Fluid Mechanics 3.2 Steady Model 3.3 Unsteady and Turbulent Model 4. CFD Analysis 4.1 Steady Results 4.2 Unsteady Results 5 Conclusion 5.1 Summary of Results 5.2 Limitations of Model 5.3 Future Work 1
8 Nomenclature: HLHS = Hypoplastic Left Heart Syndrome BTS = Classic Norwood Procedure with Blalock-Taussig Shunt RVPAS = Norwood Procedure with right ventricle to pulmonary artery shunt CFD NSE = Computational Fluid Dynamics = Navier-Stokes Equations PF = Pulmonary Flow Q p Q s = Volume Flow Rate of Pulmonary Arteries = Volume Flow Rate of Systemic Circulation or Aorta 2
9 1. INTRODUCTION This section will outline the objectives of the project. Details of hypoplastic left heart syndrome will be discussed, along with the strategies to help reroute the blood flow throughout the heart. A short background on computational fluid dynamics and how it is used for fluid mechanics problems is given. 1.1 Hypoplastic Left Heart Syndrome Hypoplastic Left Heart Syndrome (HLHS) contributes the 2-3% of all congenital diseases [1]. The disease leaves neonates with little or no function of the left ventricle, which means the right side of the heart has to perform the systolic and pulmonary circulatory functions. This presents difficulty on the heart since only one ventricle is strong enough to pump blood throughout the body. HLHS also changes how blood and oxygen is distributed about the heart, the lungs, and the rest of the body. The disease requires that the infant be under constant care, and immediate surgery is needed to reroute the circulation of blood throughout the heart. Figure 1 shows a heart with HLHS [1]. 3
10 Figure 1 Hypoplastic Left Heart Syndrome 4
11 1.2 Palliated Strategies There are three different palliative strategies for correcting HLHS. The first is the Classic Norwood Procedure (BTS). It is shown in Figure 2 [1]. This procedure involves connecting a surgical conduit called a Blalock-Taussig Shunt that directs blood from the aorta to the pulmonary arteries to become oxygenized. The aorta is also sewed up with a homograft patch to allow blood to flow into the aorta while minimizing tissue damage. The second strategy for HLHS is also a Norwood procedure, except the shunt is moved to connect the right ventricle to the pulmonary artery (RVPAS). This is shown in Figure 3 [1]. The third strategy is called the hybrid procedure. As shown in figure 4, the pulmonary arteries and the aorta remain attached [1]. Also, the atrium septum is ballooned or stented to allow for better mixing of blood and oxygen and to prevent inflow and outflow obstruction. A stent is placed in the ductus arterious. There is little circulation modeling data available to compare these strategies for best patient outcome. The ideal strategy would have the least amount of energy loss across the heart, low shear stress on the heart wall, and distribute the blood evenly throughout the aorta and the pulmonary arteries, among other things. 1.3 Using CFD to model the heart Computational fluid dynamics (CFD) is frequently used to solve complex fluid problems that before could only be solved experimentally. When used correctly, it is a powerful tool that gives a great first answer that can cut down or even eliminate experimental runs, which helps the research become time and cost efficient. The CFD code uses numerical methods to solve the full 5
12 Navier-Stokes equations (NSE), which are highly nonlinear partial differential equations. Since no close form solution exists for the Navier-Stokes equations, numerical methods must be used. These equations give information about the fluid s mass, momentum and energy, and are ultimately solved to give pressure and velocity of the fluid. CFD has traditionally been used to solve aerospace and mechanical engineering problems, but it can be used to solve any problems that involve a fluid. The heart can be compared to a complex series of pipes, in which the fluid is blood. The models of the heart can be drawn using any computer aided drawing tool. From there, a grid will need to be applied to each model, which tells the CFD program where on the geometry the NSE need to be solved. In general, the more grid points or nodes that are added, the more accurate the solution becomes. However, as the number of nodes increases, the amount of time needed to solve the equations increases. There are many commercial programs for creating a grid. The next step is using the CFD program to solve for the pressure and velocity, among other things, at each of the grid points. This usually takes the longest as there can be millions of grid points and thousands of iterations. This process will be used to solve for the pressure difference across the heart for several different models. Three models for BTS procedure using a shunt size of 3, 3.5 and 4 mm in diameter will be solved for, as well as three models of the RVPAS strategy, using 4, 5 and 6 mm in diameter. The hybrid procedure will be solved for different sizes of pulmonary artery banding. The data used from the CFD models of each of the models will be used to determine which strategy produces the lowest pressure drop across the heart, as well as the lowest shear stress on the heart wall, and the most even distribution of blood to the pulmonary arteries and the aorta. 6
13 Figure 2 Norwood Procedure with Blalock-Taussig Shunt 7
14 Figure 3 Norwood Procedure with Right Ventricle to Pulmonary Artery Shunt 8
15 Figure 4 Hybrid Procedure with Pulmonary Artery Banding 2. Background This section details the necessary information about the heart. It will cover essential parts of the heart that will need to be modeled. It will then detail the circulatory system of both the healthy heart and the HLHS heart. From there, the necessary information about the three palliative strategies will be examined. 9
16 2.1 The Healthy Heart In order to complete the investigation, knowledge of fluid mechanics, computational mechanics, and the human heart is needed. The heart is a complex muscle that pumps blood through the circulatory system of the body. It is usually about the size of a human fist and grows accordingly. Blood travels through the circulatory system to deliver oxygen and nutrients to and from cells of the body to help fight disease and stabilize body temperature. The heart is divided into four chambers. The left and right ventricles are the lower half of the heart and the left and right atria are the upper half. In a normal heart, the right ventricle pumps the same amount of blood into the lungs as the left ventricle pumps out to the body. The heart beats at an average of 120 beats per minute for an infant, which is twice the amount as an adult heart. The average blood pressure is lower than the adult, which is 70 mmhg systolic over 40 mmhg diastolic. Systolic pressure signifies when the heart beats and the volume is the smallest, and diastolic signifies when the heart relaxes and is at its greatest volume. 2.2 The Circulatory System In a healthy heart, oxygen-depleted blood first enters through the superior vena cava from the top of the body and the inferior vena cava from the bottom, as shown in figure 5 [2]. The blood then flows into the right atrium where it is held by the tricuspid valve. When that valve is released, the blood enters the right ventricle, where it is pumped to the pulmonary semilunar valve into the pulmonary arteries. The pulmonary arteries lead to the lungs where the blood becomes oxygenized. The flow of blood from the vena cava to lungs is known as the pulmonary 10
17 circulation. Its purpose is to oxygenize the blood and is supported by the pumping of the right ventricle. After the blood is oxygenized from the lungs, it returns to the heart through the pulmonary veins where it enters the left atrium. The mitral valve prevents backflow from the left ventricle to the left atrium. The now oxygen rich blood is pumped through the aortic valve where it is then released through the aorta to the rest of the body. The descending aorta pumps blood to the lower half of the body, where it returns to the heart through the inferior vena cava. The three branches of the aortic arch pumps blood to the upper half of the body and then it returns to the heart through the superior vena cava. This part of the circulatory system is referred to as the systemic system. It pumps oxygenated blood from the lungs to the rest of the body. An anterior and posterior view of the heart is shown in figures 5 and 6 respectively [2]. Figure 5 Anterior View of Normal Heart 11
18 Figure 6 Posterior View of Normal Heart The systemic circulation has a higher fluid resistance than the pulmonary system. That is because the systemic system includes all the veins and capillaries of the entire body, which are very long and have a small diameter. The left ventricle which supports the systemic system by pumping blood throughout the body is stronger than the right ventricle because it pumps blood through a resistance of approximately ten times higher than the pulmonary system. Any deficiency of the left ventricle is a serious concern to the overall cardio vascular system. 12
19 2.3 Hypoplastic Left Heart Syndrome HLHS is classified as a congenital heart disease where the left ventricle is under developed and hence dysfunctional. The blood must now be pumped entirely throughout the body by the right ventricle. Much like in a healthy heart, the blood still flows into the heart through the superior and inferior vena cava where it is held in the right atrium by the tricuspid valve. The blood is then pumped throughout the body by the right ventricle, but the blood flow splits into the pulmonary arteries and the aorta. A mixture of oxygen rich and oxygen depleted blood enters the aorta to be pumped throughout the body. The same blood mixture also enters the pulmonary arteries, where the blood goes through the lungs and the pulmonary veins and returns to the vena cava through the patent foramen ovale. This opening only exists through the development of the heart, and eventually closes up. This makes it imperative that surgery is done immediately upon birth of the infant. HLHS is fatal without intervention, usually in the first few days of life. The first step to managing the disease is to stabilize the neonate s condition so that a diagnosis can be made, along with a treatment plan. There are a few ways to go about treating HLHS but the most popular is the Norwood procedure. The goal of the surgery is to make sure the remaining ventricle is unobstructed in systemic circulation. It is also important to make sure that the there is a secure and balanced flow into the pulmonary arteries. By removing the atrial septum and reconstructing the aortic arch to remove hypoplasia, unobstructed systemic circulation can be provided. The pulmonary artery homograft is then connected into the reconstructed arch so that the right ventricle will pump blood directly into the systemic circulation. To obtain balanced flow into the pulmonary and systemic circulations (Q p /Q s = 1), a correctly sized BT shunt or RVPA shunt is used. The Norwood procedure is referred to as a 13
20 palliative strategy for HPLH because it does not restore the function of the left ventricle. The Norwood is one of the most successful operations for this disease, but it remains one of the most challenging neonatal procedures that can be performed. The most common shunt size for the BTS procedure is 3.5 mm and the most common for the RVPAS procedure is 5 mm. When comparing the two Norwood procedures, retrograde diastolic flow in the aorta can be of a concern in the BTS model. This is due to flow occurring throughout the cardiac cycle, creating a flow run-off away from the aorta, which risks coronary steal syndrome. Patients with the BTS procedure tend to have low diastolic pressures that can lead to death. With the RVPA model, since most of the flow happens during systole and because there is not flow run-off during diastole, diastolic pressure is maintained. However, the RVPAS procedure requires a ventriculotomy, which might impair ventricle function. A recent study in the New England Journal of Medicine found that the mortality rates between infants with the BTS Norwood and RVPAS Norwood procedures are very similar [3]. However, there are also more instances of earlier than planned re-interventions with the RVPAS model. An alternative to the Norwood procedure has emerged in the last few years. This procedure came about in attempt to keep cardiopulmonary bypass to a minimum, considering the fragile state of the neonates. The hybrid procedure aims for the same balance of pulmonary to systemic circulation by placing constricting bands on the pulmonary arteries to limit the flow to the lungs. A stent is then placed in the arterial duct to hold it open. To then ensure atrial mixing, a ballooned septostomy is placed in the atrial septum. The procedure has had similar success in 14
21 mortality percentage as the Norwood. One critique of the procedure is the accurate placing of the pulmonary bands on the small pulmonary arteries is a very difficult challenge. No matter what method is used to treat the infant, energy loss across the heart becomes a major concern. The heart, which is now operating with only one ventricle or pump as opposed to two, has to work a lot harder to pump blood throughout the body. Minimizing local pressure losses and pressure losses due to shear stress becomes even more vital than in a healthy heart. Not much is known about the pressure loss generated from each procedure. Being able to generate data about the pressure loss in each case would help physicians determine which procedure would be better to perform. 3. CFD Modeling This section gives the necessary background of fluid mechanics needed to complete the investigation. Details of the CFD techniques will be discussed. The metrics of the models will be given. The time steady and unsteady model will be discussed in full detail. 3.1 Fluid Mechanics In order to complete the CFD process, knowledge of fluid mechanics is needed to set up the model and evaluate the results. The primary goal of this investigation is to evaluate the energy losses across the heart. In a fluid system, energy is related to pressure. The relation can be seen 15
22 by inspection of the units. Energy is in Joules, or Newton meters. Pressure is in Pascals, or Newton per square meter. So pressure is analogous to energy per unit volume. Energy losses in the heart are often evaluated using the pressure drop from one point to another point downstream. [4], [5]. Fluid moves from high pressure to low pressure, so a high pressure is needed to drive the fluid from start to finish. This high pressure is created by the beating of the heart. The more losses that occur in the heart, the harder the heart has to beat to create the same flow rate across the circulatory system. Much like in an electrical system, fluid will attempt to flow through the path of least resistance. Equation 1 is the resistance of, it can be seen that fluid will flow through the path that has the least amount of pressure [6]. The radius of the tube the fluid follows is also a very important factor in fluid resistance, as shown in equation 1. QL R r 4 P 1. Equation 1 is known as Poiseuille s Law. Length, volume flow, and viscosity are proportional to the fluid resistance, while pressure drop is inversely proportional. The radius of the tube to the fourth power is inversely proportional to resistance making it the biggest determining factor. As the fluid goes along its path, it will lose its energy, or pressure. These losses are due to shear stress losses at the walls, local losses from flow acceleration, decelerating, or turning, and turbulence. Shear stress is the friction the fluid particles encounter when reacting with each other and the wall of the heart [7]. It is more formally defined by equation 2. u 2. y 16
23 In equation 2, u is the axial velocity relative to the wall, and y is the distance from the wall. By examination of equation 2, an increase in the rate of axial velocity along the wall will increase the shear stress. Since stress is a force per unit area, an increase in the surface area, will increase the overall viscous force and hence losses. It has been found that larger arteries produce large shear [8]. Flow accelerates when going from a larger to smaller cross sectional area to comply with the conservation of mass, so having a smaller area may increase the shear. The viscosity of the fluid is, which for all models will be constant. Local losses and turbulence are similar in their nature. Turbulence is a function of Reynolds number, which is defined in equation 3. DV Re 3. A low Reynolds number, which for internal flow is around 2300, will put the flow in a laminar flow regime. Laminar flow will follow straight path lines. As the Reynolds number increases, it enters a transitional period and then eventually becomes turbulent. In the transitional and turbulent regimes, the fluid particles become erratic and time dependent in nature. Turbulence is usually modeled as fluid particles vibrating back and forth across a mean value as shown in equations 4 and 5. The following equations outline the k- model, a popular two equation model which gives the turbulent kinetic energy and the dissipation rate, respectively [9]. k t U j k x j ij U x j i x j T k ( ) k x j 4. 17
24 U t j x j C 1 ij k U x j i C 2 2 k x j T ( ) x j 5. The eddy viscosity if defined below: T C k 2 Table 1 gives the common constants used in this model, for simple internal flow. C μ 0.09 C C σ k 1 σ ε 1.3 Table 1 Commonly used k- model constants Similar to the damping of a mechanical spring system, there are losses associated by turbulence. In the fluid system, the losses are accounted for by a loss in pressure. Just like turbulence forces fluid particles off a straight path, changes in area or curvature of the conduit the fluid is following in will change the path of the fluid particle. The losses the fluid obtains from these changes are referred to as local losses. 18
25 19 Blood flow, like most fluids, can be quantified by the continuity equation and the Navier-Stokes equations, as shown in equations 6, 7, 8, and 9 [6]. These equations hold for Newtonian fluids, and while blood is not a Newtonian fluid, it can be modeled as such due to the large chambers it passes through, relative to the size of the oxygen molecules. 0 z w y v x u 6. g x z u y u x u x p z u w y u v x u u t u g y z v y v x v y p z v w y v v x v u t v g z z w y w x w z p z w w y w v x w u t w The first equation is the continuity equation, which is a statement of the conservation of mass. The following three equations are the momentum equations, which state momentum is conserved. These four equations, along with the k and ε equations if the flow is turbulent, is what the CFD solver will numerically solve to determine information about the flow at the various grid points designated. These equations are coupled partial differential equations that can not be solved by any known analytical means. This means numerical methods such as the finite difference methods, are needed to approximate the solution to the equations. CFD modeling is used to numerically solve the NSE.
26 The commercial software Fluent and Gambit were used to construct models of a neonatal heart with HLHS. Gambit was used to draw and grid the model and Fluent was used to solve the NSE for pressure and velocity, and also for some post processing. The program Tecplot was also used for post processing. Models of the BTS, RVPA, and Hybrid were constructed. Three models for the BTS were made, using 3 mm, 3.5 mm, and 4mm diameter for the conduit. Three models were also made for the RVPA, using 4mm, 5mm, and 6 mm for the conduit size. For the hybrid model, the bands on the pulmonary arteries shrunk the arteries to half of their diameter. A steady and unsteady version of each model was made. The steady version has a constant pressure at the outlets while the unsteady model has an oscillating pressure at the outlets, to account for the time dependant beating of the heart. Table 2 outlines metrics that are used on all models. They are the diameters of the major arteries of the heart, along with the properties of the blood used [10], [11]. Tricuspid Valve (inlet) 14 mm Pulmonary Artery 5 mm Left and Right pulmonary arteries (4 outlets) 2 mm Aorta (outlet) 12 mm Left subclavian artery (outlet) 6.5 mm Left common carotid Artery (outlet) 5.1 mm Brachiocephalic Artery (outlet) 4.8 mm coronary vessels (2outlets) 0.5 mm Density of Blood 1025 kg/m 3 Viscosity of Blood kg/(m-s) Table 2 Heart Metrics 20
27 3.2 Steady Model For the steady model, no turbulence was used. It is an open circuit model with a distinct inlet and several outlets. The inlet for the model is the inlet for the right ventricle, or the tricuspid valve. The pressure set at this inlet is variable for all the models. The outlets are the descending aorta, the three main branches of the aorta, which are the brachiocephalic artery, the left common carotid artery and the left subclavian artery, and the pulmonary arteries. These outlets are set to a pressure of 70 mmhg to simulate the end systolic state of the heart. The inlet is set to a pressure that will create.035 kg/s mass flow rate, or 2000 ml/minute of blood, which is average of infant cardiac output [12]. The pressure difference in each model will be determined by taking the variable inlet pressure and subtracting it with outlet pressure. Figures 7, 8 and 9 show the gridded model of the heart system for the BTS, RVPS, and Hybrid models respectively. Figure 7 shows the 3.5 mm shunt version of the model, and figure 8 shows the 4 mm shunt version of the model. Figure 9 is the hybrid model with no pulmonary artery banding. The average number of nodes and tetrahedral elements is approximately 500,000 and 1,200,000 respectively. The nodes were chosen to surpass the amount of nodes in literature that uses similar geometry [5], [13], [14]. In Bove et al, approximately 100,000 nodes were used with grid studies including fewer nodes. In surpassing this number, the model s results should be just as accurate. More nodes were placed close to the walls to capture boundary layer flow. 21
28 Figure 7 Grid of BTS 3.5 mm model 22
29 Figure 8 Grid of RVPAS 4 mm model 23
30 Figure 8 Grid of Hybrid Model 24
31 The gridded models get exported to fluent to be run as a three dimensional model. The model is first set as steady and laminar. The fluid properties of blood are used, which are 1025 kg/m 3 for the density and N-s/kg for the viscosity. Once the boundary conditions are set, the simulation can be run till a convergence criteria is met. In this case, the simulation runs until the average value of each of the 4 equations (mass, and three momentum equations) is within 10-4 error. Error is defined as the absolute value of the left hand side of the equations minus the right hand side of the equation. Once convergence is met, the data can be analyzed. The pressure difference between the inlet and the outlets is taken, along with the shear stress of the walls. The mass flow rate going to the pulmonary arteries and going through the aorta is also calculated. 3.3 Unsteady Model There are a couple major differences between the steady and unsteady model. First, as the name implies, the pressure at the outlets are unsteady, to give the heart a pulse. The other difference is now turbulence is modeled. A k-epsilon turbulence model is used due to its simplicity and accuracy in non-reacting flows. A user defined function must be made and imported into Fluent. A cosine model is used to oscillate between 40 and 70 mmhg pressure at 120 times a minute, or twice a second. Equation 10 shows the heart beat model, with the pressure being outputted in Pascals P out 2000Cos
32 In order to simulate the difference in resistance between the pulmonary circulation and the systemic circulation, the outlets of the left and right pulmonary arteries were set to the values shown in table 3 [13]. The pressures at the coronary arteries are also given. mmhg Pa Coronary Artery LPA RPA Table 3 Pressures in Unsteady Model The same geometry and grid is used in the steady model as in the unsteady model. Once exported to Fluent, the same set up is used, and the simulation is run to the same convergence criteria as the steady model. The only difference is a time step must be set. In order to oscillate between 80 and 120 mmhg fully, or the heart to beat once, a half of a second is needed. The simulation will run with time steps of.001 seconds, for 500 time steps, which will reach a simulated time of.5 seconds, or one complete heartbeat. The pressure difference is taken between the inlet and the outlets to evaluate the energy loss. The shear stress and mass flow rate of the systemic and diastolic systems were evaluated the same as in the steady model. 26
33 4. Results and Analysis The results for the BTS models are first discussed which are followed by the results for the RVPAS models, and then the hybrid models. Information on overall pressure loss and average shear stress is then given along with flow distribution between pulmonary and systemic circulation 4.1 Steady Results Results for percent pressure loss, shear stress and volume flow ratio between the systemic circulation and the pulmonary circulation (Q p /Q s ) is shown in table 4 for the seven steady simulations. Q p /Q s Shear (Pa) % Pressure Loss BTS BTS BTS RVPAS RVPAS RVPAS Hybrid.5 PF Table 4 Steady State Results As expected, the pulmonary to systemic flow ratio increases as the conduit size increase in the BTS and RVPAS models. Also, as predicted, the pulmonary to systemic flow rates are much smaller than one, since the model used the same pressure for the aorta as in the pulmonary 27
34 arteries. In a real heart, the systemic resistance is about ten times the amount of the pulmonary resistance. However, trends can be seen from the data. As the shunt increase, the pressure loss also increases. In the BTS model, shear also increase as shunt size increase. This makes sense because the area of the model increases. However, for the RVPAS models, the highest shear is at the 5 mm model. In order to discuss these results, the unsteady models will be evaluated. The unsteady results are discussed in much more detail because they are more realistic models compared with the steady models. 4.2 Unsteady Results The time unsteady results for the BTS models for the end systole phase are given below in table 5. It gives data on flow rates, shear stress and percent pressure loss for the BTS models. Q p /Q s Shear (Pa) % Pressure Loss BTS BTS BTS Table 5 End Systole BTS Results As the conduit size increases, the shear stress decreases. This is most likely due to less flow entering the descending aorta, which has an increased area opposed to the pulmonary arteries, and hence more losses can occur in that area. A look at the contours for the pressure loss and shear stress will help give a better idea as to the relationship with conduit size and pressure loss. 28
35 Figures 9 through 14 give the total pressure contours for the BTS systolic simulations with 3mm, 3.5mm and 4mm conduits respectively. An anterior view is always given first, followed by a lateral view. The figures always go in order from smallest to largest conduit size. Figure 9 Anterior View of BTS 3 mm Model Total Pressure at End Systole in Pa 29
36 Figure 10 Lateral View of BTS 3 mm Model Total Pressure at End Systole in Pa Figure 11 Anterior View of BTS 3.5 mm Model Total Pressure at End Systole in Pa 30
37 Figure 12 Lateral View of BTS 3.5 mm Model Total Pressure at End Systole in Pa Figure 13 Anterior View of BTS 4 mm Model Total Pressure at End Systole in Pa 31
38 Figure 14 Lateral View of BTS 4 mm Model Total Pressure at End Systole in Pa It can be seen through the pressure contours that a lot of pressure is lost in the conduit to the pulmonary arteries. This is due to the high velocity in that area, as flow rushes to the low resistance pulmonary arteries. As the conduit increases, the color in the aorta gets a darker shade of red, indicating that more pressure is in that area, and hence losses are decreased, as shown in table 5. A look at the shear stress in figures 15 through 20 gives a closer look at where some of the losses occur. 32
39 Figure 15 Anterior View of BTS 3 mm Model Shear Stress at End Systole in Pa Figure 16 Lateral View of BTS 3 mm Model Shear Stress at End Systole in Pa 33
40 Figure 17 Anterior View of BTS 3.5 mm Model Shear Stress at End Systole in Pa Figure 18 Lateral View of BTS 3.5 mm Model Shear Stress at End Systole in Pa 34
41 Figure 19 Anterior View of BTS 4 mm Model Shear Stress at End Systole in Pa Figure 20 Lateral View of BTS 4 mm Model Shear Stress at End Systole in Pa 35
42 It is clear that the pressure loss in the conduit, and hence entire heart, is due largly to the increased shear in the conduit. This can be explained by an increase in velocity in the conduit which is connected to the low resistance pulmonary ciculatory system. The BTS 3 mm model has the most average shear, because it has the most flow in the aorta, which has a high area. Looking at the shear contours, lighter colors, indicating higher shear are shown in the aorta for the BTS 3 mm model. Another reason that flow to the aorta creates more losses is because shows that most of the local pressure losses that are present due to turning into the descending aorta and the increased area which deceleates the flow. Similar results can be seen for the end diastole phase BTS models. Q p /s Shear (Pa) % Pressure Loss BTS BTS BTS Table 6 End Diastole BTS Results The diastolic losses in overall pressure are significantly higher. This is because as the pressure decreases in the ventricle, there will be some backflow in the ventricle. This creates higher losses, as the flow has to move more to get to its final destination. The shear stress follows the same pattern, with the most in the BTS 3 mm model and the least in the BTS 4 mm model. Now, the pressure loss increases from the BTS 3.5 mm to the BTS 4mm. 36
43 As mentioned earlier, one of the downfalls of the BTS model is the higher retrograde flow in the aorta compared to the RVPAS model. With more flow in the aorta for the BTS 3 mm model, the retrograde flow could have an amplified effect on the pressure loss as more flow enters the pulmonary arteries. Figures 21 through 26 give the total pressure of the BTS models in end diastole, while figures 27 through 32 gives the shear stress. Figure 21 Anterior View of BTS 3 mm Model Total Pressure at End Diastole in Pa 37
44 Figure 22 Lateral View of BTS 3 mm Model Total Pressure at End Diastole in Pa Figure 23 Anterior View of BTS 3.5 mm Model Total Pressure at End Diastole in Pa 38
45 Figure 24 Lateral View of BTS 3.5 mm Model Total Pressure at End Diastole in Pa Figure 25 Anterior View of BTS 4 mm Model Total Pressure at End Diastole in Pa 39
46 Figure 26 Lateral View of BTS 4 mm Model Total Pressure at End Diastole in Pa Figure 27 Anterior View of BTS 3 mm Model Shear Stress at End Diastole in Pa 40
47 Figure 28 Lateral View of BTS 3 mm Model Shear Stress at End Diastole in Pa Figure 29 Anterior View of BTS 3.5 mm Model Shear Stress at End Diastole in Pa 41
48 Figure 30 Lateral View of BTS 3.5 mm Model Shear Stress at End Diastole in Pa Figure 31 Anterior View of BTS 4 mm Model Shear Stress at End Diastole in Pa 42
49 Figure 32 Lateral View of BTS 4 mm Model Shear Stress at End Diastole in Pa Table 7 summarized the systolic losses for the RVPA models. Comparing them to the results for the BTS shows that the Q p /Q s values are higher for the RVPA model. This makes sense, referencing that fluid resistance is a fourth order function of tube radius. With larger conduit sizes for the RVPAS, it makes sense that more flow will enter the pulmonary arteries. Due to the decreased resistance to the pulmonary arteries, fewer losses are observed as Q p /Q s increases. For the RVPAS 4 and 5 mm models, the losses are about equal. That is most likely due to the Q p /Q s values being the same distance from 1. Figures 33 through 38 below show this decrease in pressure loss as pulmonary flow increases. 43
50 Q p /Q s Shear (Pa) % Pressure Loss RVPA RVPA RVPA Table 7 End Systole RVPAS Results Figure 33 Anterior View of RVPAS 4 mm Model Total Pressure at End Systole in Pa 44
51 Figure 34 Lateral View of RVPAS 4 mm Model Total Pressure at End Systole in Pa Figure 35 Anterior View of RVPAS 5 mm Model Total Pressure at End Systole in Pa 45
52 Figure 36 Lateral View of RVPAS 4 mm Model Total Pressure at End Systole in Pa Figure 37 Anterior View of RVPAS 6 mm Model Total Pressure at End Systole in Pa 46
53 Figure 38 Lateral View of RVPAS 6 mm Model Total Pressure at End Systole in Pa As obsvered in the BTS models, increased shear can be seen in the shunt area, for the same reason of the BTS model. The shear is greatest in the RVPAS 5 mm model. Shear stress contours can be seen in figures 39 through 44 below. In the 4 mm shunt, higher shear stress can be seen in the corner of the shunt. As the shunt diameter increases, the corner becomes less sharp, and hence less shear is observed in the corner. Significant decrease in shear happens in the RVPAS 6 mm model, due to less shear in that corner and significantly less flow in the aorta. 47
54 Figure 39 Anterior View of RVPAS 4 mm Model Shear Stress at End Systole in Pa Figure 40 Lateral View of RVPAS 4 mm Model Shear Stress at End Systole in Pa 48
55 Figure 41 Anterior View of RVPAS 5 mm Model Shear Stress at End Systole in Pa Figure 42 Lateral View of RVPAS 5 mm Model Shear Stress at End Systole in Pa 49
56 Figure 43 Anterior View of RVPAS 6 mm Model Shear Stress at End Systole in Pa Figure 44 Lateral View of RVPAS 6 mm Model Shear Stress at End Systole in Pa 50
57 Similar to the BTS model, the diastolic losses are higher for the RVPA models, as observed in Table 8. Q p /Q s Shear (Pa) % Pressure Loss RVPA RVPA RVPA Table 8 End Diastole RVPAS Results The RVPA diastolic models follow a similar pattern as the systolic. Both the shear and pressure loss decrease as the flow to the pulmonary arteries increases for both end systole and end diastole phases. Just like in the BTS model, large local losses are shown in the ascending aorta. It is logical that as less flow goes through the systemic system, there will be less overall losses. This is shown in figures 45 through 50 below. 51
58 Figure 45 Anterior View of RVPAS 4 mm Model Total Pressure at End Diastole in Pa Figure 46 Lateral View of RVPAS 4 mm Model Total Pressure at End Diastole in Pa 52
59 Figure 47 Anterior View of RVPAS 5 mm Model Total Pressure at End Diastole in Pa Figure 48 Lateral View of RVPAS 5 mm Model Total Pressure at End Diastole in Pa 53
60 Figure 49 Anterior View of RVPAS 6 mm Model Total Pressure at End Diastole in Pa Figure 50 Lateral View of RVPAS 6 mm Model Total Pressure at End Diastole in Pa 54
61 Also just like the BTS model, the conduits produce the most shear. However, this shear is concentrated in a small area, and in both the BTS and RVPAS models, shear stress decreases as more flow enters the conduit. This shows that the most overall shear is produced by the aorta. This is shown in figures 51 through 56. Figure 51 Anterior View of RVPAS 4 mm Model Shear Stress at End Diastole in Pa 55
62 Figure 52 Lateral View of RVPAS 4 mm Model Shear Stress at End Diastole in Pa Figure 53 Anterior View of RVPAS 5 mm Model Shear Stress at End Diastole in Pa 56
63 Figure 54 Lateral View of RVPAS 5 mm Model Shear Stress at End Diastole in Pa Figure 55 Anterior View of RVPAS 6 mm Model Shear Stress at End Diastole in Pa 57
64 Figure 56 Lateral View of RVPAS 6 mm Model Shear Stress at End Diastole in Pa It is interesting to compare the BTS and RVPAS models, as they both have high shear stress conduits. This is most likely due to the high velocity seen in this section. High velocity is generated because the fluid is introduced to the low pressure pulmonary arteries from the high pressure systemic circulation. Figure 57 shows velocity cross sectional areas at the entrance of the shunts. 58
65 Figure 57 Cross Sectional Area View of Velocity in Entrance of Shunt at End Systole in m/s With the average velocity in the models at approximately.3 m/s, very high velocity can be seen at the entrance of the shunts. Much higher velocity is seen in the BTS models, while more developed flow can be seen in the RVPAS models. Velocity decreases as shunt size increases to keep with the conservation of mass. It is also interesting to note the pressures at this point as well, which can be seen in figure
66 Figure 58 Cross Sectional Area View of Total Pressure in Entrance of Shunt at End Systole in Pa The pressure at the shunt entrance is much more developed and higher in the RVPAS models. This makes sense considering RVPA sees the higher pressure of the ventricle when it enters the shunt, as opposed to the BTS seeing the lower pressure down stream of the aorta. The more developed flow in the RVPAS models is explained by noticing that at the point of the shunt, the 60
67 flow only has two options to flow through, the shunt or aorta. On the other hand, in the BTS model, the flow can flow into the shunt, or any of the arteries in the aorta, as well as the coronary arteries. Figures 59 and 60 give the pressure cross section of the entire shunt for the BTS and RVPAS models respectively. Figure 59 Cross Sectional Area View of Total Pressure in Shunt at End Systole in Pa 61
68 Figure 60 Cross Sectional Area View of Total Pressure in Shunt at End Systole in Pa A study by Ohye et al in the New England Journal of Medicine showed little difference in mortality in the two different Norwood Procedures [3]. However, in the RVPAS Norwood procedure, there was a higher occurance of unscheduled interventions. This is most likely due to the higher pressure the RVPAS is exposed to, and perhaps the decreased velocity, which keeps flow in the conduit longer. The results for the hybrid procedure are shown below for different pulmonary artery banding in Table 9. Similar to the RVPA models, losses significantly decrease when pulmonary artery flow increases. 62
69 Systolic Q p /Q s Shear (Pa) % Pressure Loss.25 PF PF PF PF Table 9 End Systole Hybrid Results The hybrid procedure produced the least overall pressure loss and shear for the systolic phase. One of the major reasons for this is that a high shear stress conduit from the heart to the pulmonary arteries is not in the procedure. However, large amounts of shear still exist in near the pulmonary artery banding. This is because the reduced area creates a larger velocity, creating more shear as the flow accelerates. The total pressure contours, as well as the shear stress contours can be seen in figures 61 through
70 Figure 61 Anterior View of Hybrid.25 PF Model Total Pressure at End Systole in Pa Figure 62 Anterior View of Hybrid.25 PF Model Shear Stress at End Systole in Pa 64
71 Figure 63 Left Pulmonary Artery Close Up of Hybrid.25 PF Model Total Pressure at End Systole in Pa Figure 64 Left Pulmonary Artery Close Up of Hybrid.25 PF Model Shear at End Systole in Pa 65
72 Figure 65 Anterior View of Hybrid.50 PF Model Total Pressure at End Systole in Pa Figure 66 Anterior View of Hybrid.50 PF Model Shear Stress at End Systole in Pa 66
73 Figure 67 Left Pulmonary Artery Close Up of Hybrid.50 PF Model Total Pressure at End Systole in Pa Figure 68 Left Pulmonary Artery Close Up of Hybrid.50 PF Model Shear Stress at End Systole in Pa 67
74 Figure 69 Anterior View of Hybrid.75 PF Model Total Pressure at End Systole in Pa Figure 70 Anterior View of Hybrid.75 PF Model Shear Stress at End Systole in Pa 68
75 Figure 71 Left Pulmonary Artery Close Up of Hybrid.75 PF Model Total Pressure at End Systole in Pa Figure 72 Left Pulmonary Artery Close Up of Hybrid.75 PF Model Shear Stress at End Systole in Pa 69
76 Figure 73 Anterior View of Hybrid 1.00 PF Model Total Pressure at End Systole in Pa Figure 74 Anterior View of Hybrid 1.00 PF Model Shear Stress at End Systole in Pa 70
77 Figure 75 Pulmonary Artery Close Up of Hybrid 1.00 PF Model Total Pressure at End Systole in Pa Figure 76 Pulmonary Artery Close Up of Hybrid 1.00 PF Model Shear Stress at End Systole in Pa 71
78 The diastolic losses, like in all models are higher for the hybrid. They follow the same trend as the systolic losses in the hybrid, as seen in table 10. Diastolic Q p /Q s Shear (Pa) % Pressure Loss.25 PF PF PF PF Table 10 End Diastole Hybrid Results Figures 77 through 92 outline the hybrid end diastolic losses. Figure 77 Anterior View of Hybrid.25 PF Model Total Pressure at End Diastole in Pa 72
79 Figure 78 Anterior View of Hybrid.25 PF Model Shear Stress at End Diastole in Pa Figure 79 Left Pulmonary Artery Close Up of Hybrid.25 PF Model Total Pressure at End Diastole in Pa 73
80 Figure 80 Left Pulmonary Artery Close Up of Hybrid.25 PF Model Shear Stress at End Diastole in Pa Figure 81 Anterior View of Hybrid.50 PF Model Total Pressure at End Diastole in Pa 74
81 Figure 82 Anterior View of Hybrid.50 PF Model Shear Stress at End Diastole in Pa Figure 83 Left Pulmonary Artery Close Up of Hybrid.50 PF Model Total Pressure at End Diastole in Pa 75
82 Figure 84 Left Pulmonary Artery Close Up of Hybrid.50 PF Model Shear Stress at End Diastole in Pa Figure 85 Anterior View of Hybrid.75 PF Model Total Pressure at End Diastole in Pa 76
83 Figure 86 Anterior View of Hybrid.75 PF Model Shear Stress at End Diastole in Pa Figure 87 Left Pulmonary Artery Close Up of Hybrid.75 PF Model Total Pressure at End Diastole in Pa 77
84 Figure 88 Left Pulmonary Artery Close Up of Hybrid.75 PF Model Shear Stress at End Diastole in Pa Figure 89 Anterior View of Hybrid 1.00 PF Model Total Pressure at End Diastole in Pa 78
85 Figure 90 Anterior View of Hybrid 1.00 PF Model Shear Stress at End Diastole in Pa Figure 91 Pulmonary Artery Close Up of Hybrid 1.00 PF Model Total Pressure at End Diastole in Pa 79
86 Figure 92 Pulmonary Artery Close Up of Hybrid 1.00 PF Model Shear Stress at End Diastole in Pa It is difficult to find a correlation in the shear stress for the hybrid. The no banding case creates the most shear, most likely due to the increased area of the high shear stress pulmonary arteries. As the pulmonary banding contracts, several things ocure in the flow. First, the overall area becomes less, creating less shear. However this creates a higher velocity at the banding point, which creates more shear. Also, as the banding contracts, less flow goes into the high velocity pulmonary artery banding, which creates less shear, however, that sends more flow to the large surface area aorta, where a lot of shear is created. This creates an interesting optimization problem in the case of minimizing shear with in a certain Q p /Q s value. 80
87 With only a couple exceptions, as more flow entered the pulmonary arteries, overall losses decreased. This most likely has to do with the decrease shear the comes with less flow to the aorta, and less local losses from turning into the decending aorta. Another hypothesis can be that as flow heads towards all pulmonary flow, or all systolic flow, the losses decrease due to a simpler system. The RVPAS models demonstrate this. In the systolic models, losses are about the same at around.7, and 1.3 Q p /Q s values, but significantly decrease at a Q p /Q s of In the diastolic models, the Q p /Q s value was close to one for the RVPAS model, and created the most losses, while the RVPAS 4 and 6 mm created less. More evidence is shown in the steady state models, which start at very low Q p /Q s values but shear and losses increase as the conduit size increases. Directly comparing the steady and unsteady model is difficult due to the difference in turbulence modeling pressure boundary conditions, but it is interesting to point out the Q p /Q s values in the set of models. The steady models had small Q p /Q s values, and as those values increased, the pressure loss increased. However, the unsteady results had higher Qp/Qs values and the pressure loss generally decreased as Q p /Q s increased. This makes sense because as the Q p /Q s approaches 0, or all the flow in the systemic aorta, the system because much simpler and hence efficient. On the other end, as Q p /Q s heads to infinity, or all the flow into the pulmonary arteries, that also simplifies the system, making it more efficient. When flow is somewhere in the middle, and flow has to split between going to the pulmonary arteries and the aorta, the system is more complex and open to a lot of local losses from turning, accelerating, and decelerating. This correlation is probably the hardest to see in the BTS models. For the RVPAS and hybrid models, if the Q p /Q s values were to head towards infinity, no flow would have to enter the aorta. However, in the BTS 81
88 models, no matter what the Q p /Q s values are, the flow must enter the ascending aorta and the branches of the aortic arch in order to enter the conduit to get to the pulmonary arteries. 5. Conclusion and Future Work 5.1 Summary From the results of the unsteady simulations, the models produced less overall pressure loss when Q p /Q s values were higher in general. The RVPAS 5 mm model produced the most even flow between the pulmonary and systemic circulation for both systolic and diastolic models. The steady simulations, produced higher losses as Q p /Q s became higher, indicating that a maximum pressure loss can be obtained at some medium value of Q p /Q s which is most likely different for each model. The hybrid procedure produces the least amount of shear over all for all cases, and the least amount of pressure loss in the most common clinically used cases. Shear is definitely a function of pulmonary flow, but it is a different correlation with each model. 5.2 Limitations While much can be learned from this investigation, there are improvements that could be made to the model to make it represent a heart better. First is the rigid wall assumption. In a real heart, the ventricle stretches, and losses occur due to elastic losses in the wall. In all the models, the heart wall is assumed ridged. The next addition to the model could be the addition of valves. No valve was used in the model to stop blood flow from returning to the ventricle as the pressure in the ventricle fell at the 82
89 diastole phase. Even if valves were to be added, diastolic losses would be higher, but would probably be limited. The heart model used in this investigation is also an open loop model. The real circulatory system is a closed loop, which means some energy will return to the ventricle with every beat of the heart. While this energy is very small by the time the flow cycles through the entire body, it could yield more accurate results. Addition of the valve is a necessity to make sure the flow in the closed loop case will be flow the correct direction. Also, in an infant, the heart does grow rapidly, and grows different with each palliative strategy. When models of the growth of the heart for each strategy become more sophisticated, they could be used to model energy losses over period of months as the heart grows. 5.3 Future Work In the immediate future, optimization studies could be used to find out what pulmonary to systemic flow ratio produces the least amount of shear or energy loss within a certain boundary. Also, optimization studies can be done on the hybrid to learn about the best pulmonary artery banding size to use to minimize shear. Since a Q p /Q s value of 1 happens between the pulmonary artery banding of.25 PF and.5 PF, more data should be found between those two points. As more data is generated off the current model, more sophisticated models can be generated. Turning the model into a closed loop will help determine if energy dissipated back to the right 83
Computational Fluid Dynamics Analysis of Blalock-Taussig Shunt
 Washington University in St. Louis Washington University Open Scholarship Mechanical Engineering and Materials Science Independent Study Mechanical Engineering & Materials Science 12-23-2017 Computational
Washington University in St. Louis Washington University Open Scholarship Mechanical Engineering and Materials Science Independent Study Mechanical Engineering & Materials Science 12-23-2017 Computational
Health Science 20 Circulatory System Notes
 Health Science 20 Circulatory System Notes Functions of the Circulatory System The circulatory system functions mainly as the body s transport system. It transports: o Oxygen o Nutrients o Cell waste o
Health Science 20 Circulatory System Notes Functions of the Circulatory System The circulatory system functions mainly as the body s transport system. It transports: o Oxygen o Nutrients o Cell waste o
The cardiovascular system is composed of a pump the heart and blood
 5 E X E R C I S E Cardiovascular Dynamics O B J E C T I V E S 1. To understand the relationships among blood flow, pressure gradient, and resistance 2. To define resistance and describe the main factors
5 E X E R C I S E Cardiovascular Dynamics O B J E C T I V E S 1. To understand the relationships among blood flow, pressure gradient, and resistance 2. To define resistance and describe the main factors
Closed-loop CFD Model of the Self-Powered Fontan Circulation for the Hypoplastic Left Heart Syndrome
 McNair Scholars Research Journal Volume 3 Article 4 016 Closed-loop CFD Model of the Self-Powered Fontan Circulation for the Hypoplastic Left Heart Syndrome Nathalie E. Quintero Embry-Riddle Aeronautical
McNair Scholars Research Journal Volume 3 Article 4 016 Closed-loop CFD Model of the Self-Powered Fontan Circulation for the Hypoplastic Left Heart Syndrome Nathalie E. Quintero Embry-Riddle Aeronautical
Chapter 14. The Cardiovascular System
 Chapter 14 The Cardiovascular System Introduction Cardiovascular system - heart, blood and blood vessels Cardiac muscle makes up bulk of heart provides force to pump blood Function - transports blood 2
Chapter 14 The Cardiovascular System Introduction Cardiovascular system - heart, blood and blood vessels Cardiac muscle makes up bulk of heart provides force to pump blood Function - transports blood 2
THE HEART. Unit 3: Transportation and Respiration
 THE HEART Unit 3: Transportation and Respiration The Circulatory System Also called the Cardiovascular System Circulates blood in the body Transports nutrients, oxygen, carbon dioxide, hormones, and blood
THE HEART Unit 3: Transportation and Respiration The Circulatory System Also called the Cardiovascular System Circulates blood in the body Transports nutrients, oxygen, carbon dioxide, hormones, and blood
THE CIRCULATORY SYSTEM
 Biology 30S THE CIRCULATORY SYSTEM Name: This module adapted from bblearn.merlin.mb.ca 1 Introduction to Circulation The first organ to form, and the last organ to die. The heart is the pump of life. The
Biology 30S THE CIRCULATORY SYSTEM Name: This module adapted from bblearn.merlin.mb.ca 1 Introduction to Circulation The first organ to form, and the last organ to die. The heart is the pump of life. The
CIRCULATORY SYSTEM BLOOD VESSELS
 Name: Block: CIRCULATORY SYSTEM Multicellular organisms (above the level of roundworms) rely on a circulatory system to bring nutrients to, and take wastes away from, cells. In higher organisms such as
Name: Block: CIRCULATORY SYSTEM Multicellular organisms (above the level of roundworms) rely on a circulatory system to bring nutrients to, and take wastes away from, cells. In higher organisms such as
CVS Hemodynamics. Faisal I. Mohammed, MD,PhD.
 CVS Hemodynamics Faisal I. Mohammed, MD,PhD. Objectives point out the physical characteristics of the circulation: distribution of blood volume total cross sectional area velocity blood pressure List the
CVS Hemodynamics Faisal I. Mohammed, MD,PhD. Objectives point out the physical characteristics of the circulation: distribution of blood volume total cross sectional area velocity blood pressure List the
Cardiovascular System Notes: Physiology of the Heart
 Cardiovascular System Notes: Physiology of the Heart Interesting Heart Fact Capillaries are so small it takes ten of them to equal the thickness of a human hair. Review What are the 3 parts of the cardiovascular
Cardiovascular System Notes: Physiology of the Heart Interesting Heart Fact Capillaries are so small it takes ten of them to equal the thickness of a human hair. Review What are the 3 parts of the cardiovascular
The Mammalian Circulatory System
 The Mammalian Heart The Mammalian Circulatory System Recall: What are the 3 cycles of the mammalian circulatory system? What are their functions? What are the three main vessel types in the mammalian circulatory
The Mammalian Heart The Mammalian Circulatory System Recall: What are the 3 cycles of the mammalian circulatory system? What are their functions? What are the three main vessel types in the mammalian circulatory
Identify and describe the circulation system that is missing from the organizer above.
 Lesson 15.1 NOTES: The Circulatory System (Unlock) Essential Question: -What are the structures and functions of the circulatory system? Learning Target(s): -I can identify structures and explain functions
Lesson 15.1 NOTES: The Circulatory System (Unlock) Essential Question: -What are the structures and functions of the circulatory system? Learning Target(s): -I can identify structures and explain functions
Cardiovascular System- Heart. Miss Wheeler Unit 8
 Cardiovascular System- Heart Miss Wheeler Unit 8 Overview CARDIOVASCULAR SYSTEM heart vessels Made up of heart, blood vessels, and blood Functions Heart- pump blood Vessels- (veins, arteries, capillaries)
Cardiovascular System- Heart Miss Wheeler Unit 8 Overview CARDIOVASCULAR SYSTEM heart vessels Made up of heart, blood vessels, and blood Functions Heart- pump blood Vessels- (veins, arteries, capillaries)
The Heart. Made up of 3 different tissue: cardiac muscle tissue, nerve tissue, and connective tissue.
 The Heart The Heart Made up of 3 different tissue: cardiac muscle tissue, nerve tissue, and connective tissue. Your heart pumps with a regular beat (Heart Rate) Your heart rate can change depending on
The Heart The Heart Made up of 3 different tissue: cardiac muscle tissue, nerve tissue, and connective tissue. Your heart pumps with a regular beat (Heart Rate) Your heart rate can change depending on
THE HEART. Structure & Function
 THE HEART Structure & Function SARAH JOHANSON BIOTECH ENGINEERING, 2015 Function of the Heart: The heart is muscular organ that sits centrally in the thorax region of the body, but is skewed and twisted
THE HEART Structure & Function SARAH JOHANSON BIOTECH ENGINEERING, 2015 Function of the Heart: The heart is muscular organ that sits centrally in the thorax region of the body, but is skewed and twisted
Biology Unit 3 The Human Heart P
 Biology 2201 Unit 3 The Human Heart P 314-321 Structure and Function of the Human Heart Structure of the Human Heart Has four Chambers (2 Atria and 2 Ventricles) Made of Cardiac Muscle Found in Chest Cavity
Biology 2201 Unit 3 The Human Heart P 314-321 Structure and Function of the Human Heart Structure of the Human Heart Has four Chambers (2 Atria and 2 Ventricles) Made of Cardiac Muscle Found in Chest Cavity
Large Arteries of Heart
 Cardiovascular System (Part A-2) Module 5 -Chapter 8 Overview Arteries Capillaries Veins Heart Anatomy Conduction System Blood pressure Fetal circulation Susie Turner, M.D. 1/5/13 Large Arteries of Heart
Cardiovascular System (Part A-2) Module 5 -Chapter 8 Overview Arteries Capillaries Veins Heart Anatomy Conduction System Blood pressure Fetal circulation Susie Turner, M.D. 1/5/13 Large Arteries of Heart
Circulatory System Review
 Circulatory System Review 1. Know the diagrams of the heart, internal and external. a) What is the pericardium? What is myocardium? What is the septum? b) Explain the 4 valves of the heart. What is their
Circulatory System Review 1. Know the diagrams of the heart, internal and external. a) What is the pericardium? What is myocardium? What is the septum? b) Explain the 4 valves of the heart. What is their
TOPIC 6: HUMAN HEALTH AND PHYSIOLOGY
 TOPIC 6: HUMAN HEALTH AND PHYSIOLOGY 6.2 Transport System/Circulatory Draw and label a diagram of the heart showing the four chambers, associated blood vessels, valves and the route of blood through the
TOPIC 6: HUMAN HEALTH AND PHYSIOLOGY 6.2 Transport System/Circulatory Draw and label a diagram of the heart showing the four chambers, associated blood vessels, valves and the route of blood through the
1. Which of the following blood vessels has a thin elastic layer? A. Aorta. B. Pulmonary artery. C. Posterior vena cava. D. Mesenteric capillary.
 CIRCULATORY SYSTEM 1. Which of the following blood vessels has a thin elastic layer? A. Aorta. B. Pulmonary artery. C. Posterior vena cava. D. Mesenteric capillary. 2. Capillary beds are equipped with
CIRCULATORY SYSTEM 1. Which of the following blood vessels has a thin elastic layer? A. Aorta. B. Pulmonary artery. C. Posterior vena cava. D. Mesenteric capillary. 2. Capillary beds are equipped with
A Computational Fluid Dynamics Study on Bidirectional Glenn Shunt Flow with an Additional Pulsatile Flow Through a modified Blalock-Taussig Shunt
 University of New Orleans ScholarWorks@UNO University of New Orleans Theses and Dissertations Dissertations and Theses Spring 5-19-2017 A Computational Fluid Dynamics Study on Bidirectional Glenn Shunt
University of New Orleans ScholarWorks@UNO University of New Orleans Theses and Dissertations Dissertations and Theses Spring 5-19-2017 A Computational Fluid Dynamics Study on Bidirectional Glenn Shunt
Physics of the Cardiovascular System
 Dentistry College Medical Physics Physics of the Cardiovascular System The cells of the body act like individual engines. In order for them to function they must have: - 1. Fuel from our food to supply
Dentistry College Medical Physics Physics of the Cardiovascular System The cells of the body act like individual engines. In order for them to function they must have: - 1. Fuel from our food to supply
The Circulatory System
 The Circulatory System Key Questions What are the functions of the circulatory system? How does the heart pump blood through the body? What are three types of blood vessels? Vocabulary myocardium atrium
The Circulatory System Key Questions What are the functions of the circulatory system? How does the heart pump blood through the body? What are three types of blood vessels? Vocabulary myocardium atrium
Cardiovascular Physiology
 Cardiovascular Physiology Lecture 1 objectives Explain the basic anatomy of the heart and its arrangement into 4 chambers. Appreciate that blood flows in series through the systemic and pulmonary circulations.
Cardiovascular Physiology Lecture 1 objectives Explain the basic anatomy of the heart and its arrangement into 4 chambers. Appreciate that blood flows in series through the systemic and pulmonary circulations.
Chapter 27 -The Heart & Blood Vessels
 Chapter 27 -The Heart & Blood Vessels 3.2 Learning Objectives 3.2.2 Organisational Complexity of the human 1. Describe the structures and organisation of tissues in the closed circulatory system. 2. Discuss
Chapter 27 -The Heart & Blood Vessels 3.2 Learning Objectives 3.2.2 Organisational Complexity of the human 1. Describe the structures and organisation of tissues in the closed circulatory system. 2. Discuss
10. Thick deposits of lipids on the walls of blood vessels, called, can lead to serious circulatory issues. A. aneurysm B. atherosclerosis C.
 Heart Student: 1. carry blood away from the heart. A. Arteries B. Veins C. Capillaries 2. What is the leading cause of heart attack and stroke in North America? A. alcohol B. smoking C. arteriosclerosis
Heart Student: 1. carry blood away from the heart. A. Arteries B. Veins C. Capillaries 2. What is the leading cause of heart attack and stroke in North America? A. alcohol B. smoking C. arteriosclerosis
Pearson's Comprehensive Medical Assisting Administrative and Clinical Competencies
 Pearson's Comprehensive Medical Assisting Administrative and Clinical Competencies THIRD EDITION CHAPTER 27 The Cardiovascular System Lesson 1: Overview of the Cardiovascular System Lesson Objectives Upon
Pearson's Comprehensive Medical Assisting Administrative and Clinical Competencies THIRD EDITION CHAPTER 27 The Cardiovascular System Lesson 1: Overview of the Cardiovascular System Lesson Objectives Upon
The Cardiovascular System (Heart)
 The Cardiovascular System The Cardiovascular System (Heart) A closed system of the heart and blood vessels The heart pumps blood Blood vessels allow blood to circulate to all parts of the body The function
The Cardiovascular System The Cardiovascular System (Heart) A closed system of the heart and blood vessels The heart pumps blood Blood vessels allow blood to circulate to all parts of the body The function
Chapter 27 The Heart and Blood Vessels
 Chapter 27 The Heart and Blood Vessels Most animals have a closed blood system. The blood flows continuously in vessels back to the heart. In an open system the blood is pumped into open ended tubes and
Chapter 27 The Heart and Blood Vessels Most animals have a closed blood system. The blood flows continuously in vessels back to the heart. In an open system the blood is pumped into open ended tubes and
Figure ) The specific chamber of the heart that is indicated by letter A is called the. Diff: 1 Page Ref: 364
 Essentials of Anatomy and Physiology, 9e (Marieb) Chapter 11 The Cardiovascular System Short Answer Figure 11.1 Using Figure 11.1, identify the following: 1) The Purkinje fibers are indicated by label.
Essentials of Anatomy and Physiology, 9e (Marieb) Chapter 11 The Cardiovascular System Short Answer Figure 11.1 Using Figure 11.1, identify the following: 1) The Purkinje fibers are indicated by label.
CIE Biology GCSE. 9: Transport in animals. Notes.
 CIE Biology GCSE 9: Transport in animals Notes The circulatory system acts as the main transport system in animals. It is made up of blood vessels such as arteries, veins and capillaries, in which blood
CIE Biology GCSE 9: Transport in animals Notes The circulatory system acts as the main transport system in animals. It is made up of blood vessels such as arteries, veins and capillaries, in which blood
CPM Specifications Document Fontan - Exercise:
 CPM Specifications Document Fontan - Exercise: OSMSC 0063_2000, 0063_3000, 0063_4000, 0064_2000, 0064_3000, 0064_4000, 0065_2000, 0065_3000, 0065_4000, 0075_2000, 0075_3000, 0075_4000, 0076_2000, 0076_3000,
CPM Specifications Document Fontan - Exercise: OSMSC 0063_2000, 0063_3000, 0063_4000, 0064_2000, 0064_3000, 0064_4000, 0065_2000, 0065_3000, 0065_4000, 0075_2000, 0075_3000, 0075_4000, 0076_2000, 0076_3000,
Ch.15 Cardiovascular System Pgs {15-12} {15-13}
 Ch.15 Cardiovascular System Pgs {15-12} {15-13} E. Skeleton of the Heart 1. The skeleton of the heart is composed of rings of dense connective tissue and other masses of connective tissue in the interventricular
Ch.15 Cardiovascular System Pgs {15-12} {15-13} E. Skeleton of the Heart 1. The skeleton of the heart is composed of rings of dense connective tissue and other masses of connective tissue in the interventricular
Cardiovascular System
 Cardiovascular System Purpose Transport oxygen and nutrients Take waste products away from tissues & organs Things we learned Blood pressure: the force of blood pushing against the walls of blood vessels
Cardiovascular System Purpose Transport oxygen and nutrients Take waste products away from tissues & organs Things we learned Blood pressure: the force of blood pushing against the walls of blood vessels
(2) (1) (3) (4) BLOOD PATHWAY ASSESSMENT RUBRIC
 BLOODPATHWAYASSESSMENT(4) BLOOD%PATHWAY%ASSESSMENT%(3)% BLOODPATHWAYASSESSMENT(3) (4) (3) (2) (1) Using a completely blank diagram of the heart, all valves, chambers, great vessels, and direction of blood
BLOODPATHWAYASSESSMENT(4) BLOOD%PATHWAY%ASSESSMENT%(3)% BLOODPATHWAYASSESSMENT(3) (4) (3) (2) (1) Using a completely blank diagram of the heart, all valves, chambers, great vessels, and direction of blood
Contents 1 Computational Haemodynamics An Introduction 2 The Human Cardiovascular System
 Contents 1 Computational Haemodynamics An Introduction... 1 1.1 What is Computational Haemodynamics (CHD)... 1 1.2 Advantages of CHD... 3 1.3 Applications in the Cardiovascular System... 4 1.3.1 CHD as
Contents 1 Computational Haemodynamics An Introduction... 1 1.1 What is Computational Haemodynamics (CHD)... 1 1.2 Advantages of CHD... 3 1.3 Applications in the Cardiovascular System... 4 1.3.1 CHD as
The Heart. Happy Friday! #takeoutyournotes #testnotgradedyet
 The Heart Happy Friday! #takeoutyournotes #testnotgradedyet Introduction Cardiovascular system distributes blood Pump (heart) Distribution areas (capillaries) Heart has 4 compartments 2 receive blood (atria)
The Heart Happy Friday! #takeoutyournotes #testnotgradedyet Introduction Cardiovascular system distributes blood Pump (heart) Distribution areas (capillaries) Heart has 4 compartments 2 receive blood (atria)
THE HEART. Structure & Function
 THE HEART Structure & Function SARAH JOHANSON BIOTECH ENGINEERING, 2015 Function of the Heart: The heart is muscular organ that sits centrally in the thorax region of the body, but is skewed and twisted
THE HEART Structure & Function SARAH JOHANSON BIOTECH ENGINEERING, 2015 Function of the Heart: The heart is muscular organ that sits centrally in the thorax region of the body, but is skewed and twisted
Circulatory System 10.1
 1 Circulatory System 10.1 2 ARTERIES Arteries-blood vessels that carry blood away from the heart Thick walls Inner & Outer layers: connective tissue Middle layers are muscle and elastic connective tissue
1 Circulatory System 10.1 2 ARTERIES Arteries-blood vessels that carry blood away from the heart Thick walls Inner & Outer layers: connective tissue Middle layers are muscle and elastic connective tissue
Your heart is a muscular pump about the size of your fist, located
 How Your Heart Works Your heart is a muscular pump about the size of your fist, located slightly to the left and behind your breastbone. Its function is to pump blood throughout your body. As your heart
How Your Heart Works Your heart is a muscular pump about the size of your fist, located slightly to the left and behind your breastbone. Its function is to pump blood throughout your body. As your heart
Mr. Epithelium s Anatomy and Physiology Test SSSS
 Mr. Epithelium s Anatomy and Physiology Test SSSS You have 50 minutes to complete this test packet. One 8.5 x 11 cheat sheet is allowed, along with 1 non-programmable calculator dedicated to computation.
Mr. Epithelium s Anatomy and Physiology Test SSSS You have 50 minutes to complete this test packet. One 8.5 x 11 cheat sheet is allowed, along with 1 non-programmable calculator dedicated to computation.
CVS Hemodynamics. Change in blood pressure:
 CVS Hemodynamics -The distribution of blood inside the circulation: The major part of blood volume is found in the venous system 60% (2/3), that s why veins are called the capacitance vessels. -Arteries
CVS Hemodynamics -The distribution of blood inside the circulation: The major part of blood volume is found in the venous system 60% (2/3), that s why veins are called the capacitance vessels. -Arteries
Today s objectives:! - Learn BASICS of circulatory system (Heart, different veins and arteries)! - Appreciate effects and treatment for
 Today s objectives:! - Learn BASICS of circulatory system (Heart, different veins and arteries)! - Appreciate effects and treatment for hyperlipidemia! Agenda! - Review objectives for 6.2! - Video of circulatory
Today s objectives:! - Learn BASICS of circulatory system (Heart, different veins and arteries)! - Appreciate effects and treatment for hyperlipidemia! Agenda! - Review objectives for 6.2! - Video of circulatory
This lab activity is aligned with Visible Body s A&P app. Learn more at visiblebody.com/professors
 1 This lab activity is aligned with Visible Body s A&P app. Learn more at visiblebody.com/professors 2 PRE-LAB EXERCISES: A. Watch the video 29.1 Heart Overview and make the following observations: 1.
1 This lab activity is aligned with Visible Body s A&P app. Learn more at visiblebody.com/professors 2 PRE-LAB EXERCISES: A. Watch the video 29.1 Heart Overview and make the following observations: 1.
The Circulatory System. Lesson Overview. Lesson Overview The Circulatory System
 33.1 THINK ABOUT IT More than one-third of the 1.2 million Americans who suffer a heart attack each year die. This grim evidence shows that the heart and the circulatory system it powers are vital to life.
33.1 THINK ABOUT IT More than one-third of the 1.2 million Americans who suffer a heart attack each year die. This grim evidence shows that the heart and the circulatory system it powers are vital to life.
CIRCULATION & GAS EXCHANGE
 AP BIOLOGY ACTIVITY2.13 Text:Campbell,v.8,chapter42 NAME DATE HOUR CIRCULATION & GAS EXCHANGE 1. In general, what is the function of transport systems? 2. What method/structure do most invertebrates use
AP BIOLOGY ACTIVITY2.13 Text:Campbell,v.8,chapter42 NAME DATE HOUR CIRCULATION & GAS EXCHANGE 1. In general, what is the function of transport systems? 2. What method/structure do most invertebrates use
Danil Hammoudi.MD 1/12/2009
 Danil Hammoudi.MD Aorta the biggest and longest artery (a blood vessel carrying blood away from the heart) in the body. It carries oxygen rich blood from the left ventricle of the heart to the body.inferior
Danil Hammoudi.MD Aorta the biggest and longest artery (a blood vessel carrying blood away from the heart) in the body. It carries oxygen rich blood from the left ventricle of the heart to the body.inferior
Cardiovascular System. Biology 105 Lecture 15 Chapter 12
 Cardiovascular System Biology 105 Lecture 15 Chapter 12 Outline I. Functions of cardiovascular system II. Components of the cardiovascular system: I. Blood vessels II. Heart III. Regulation of the heartbeat
Cardiovascular System Biology 105 Lecture 15 Chapter 12 Outline I. Functions of cardiovascular system II. Components of the cardiovascular system: I. Blood vessels II. Heart III. Regulation of the heartbeat
37 1 The Circulatory System
 H T H E E A R T 37 1 The Circulatory System The circulatory system and respiratory system work together to supply cells with the nutrients and oxygen they need to stay alive. a) The respiratory system:
H T H E E A R T 37 1 The Circulatory System The circulatory system and respiratory system work together to supply cells with the nutrients and oxygen they need to stay alive. a) The respiratory system:
Lesson 10 Circulatory System (Nelson p.88-93)
 Name: Date: Lesson 10 Circulatory System (Nelson p.88-93) Learning Goals: A. I can explain the primary functions of the circulatory system in animals. B. I can identify and explain all the parts of the
Name: Date: Lesson 10 Circulatory System (Nelson p.88-93) Learning Goals: A. I can explain the primary functions of the circulatory system in animals. B. I can identify and explain all the parts of the
Numerical Simulation of Blood Flow through Asymmetric and Symmetric Occlusion in Carotid Artery
 Proceedings of the 3 rd International Conference on Fluid Flow, Heat and Mass Transfer (FFHMT 16) Ottawa, Canada May 2 3, 2016 Paper No. 170 Numerical Simulation of Blood Flow through Asymmetric and Symmetric
Proceedings of the 3 rd International Conference on Fluid Flow, Heat and Mass Transfer (FFHMT 16) Ottawa, Canada May 2 3, 2016 Paper No. 170 Numerical Simulation of Blood Flow through Asymmetric and Symmetric
Design and Simulation of Blocked Blood Vessel for Early Detection of Heart Diseases
 Proceedings of the 215 2nd International Symposium on Physics and Technology of Sensors, 8-1th March, 215, Pune, India Design and Simulation of Blocked Blood Vessel for Early Detection of Heart Diseases
Proceedings of the 215 2nd International Symposium on Physics and Technology of Sensors, 8-1th March, 215, Pune, India Design and Simulation of Blocked Blood Vessel for Early Detection of Heart Diseases
The Heart and Cardiovascular System
 The Heart and Cardiovascular System What you will learn The location of the heart 3 layers and covering of the heart Explain the function of the heart as 2 separate pumps Identify the 4 chambers of the
The Heart and Cardiovascular System What you will learn The location of the heart 3 layers and covering of the heart Explain the function of the heart as 2 separate pumps Identify the 4 chambers of the
The Circulatory System (p )
 The Circulatory System (p. 268-281) How Does Gravity Affect Blood Circulation? As with all land animals, the giraffe and the corn snake are constantly subject to the force of gravity The circulatory system
The Circulatory System (p. 268-281) How Does Gravity Affect Blood Circulation? As with all land animals, the giraffe and the corn snake are constantly subject to the force of gravity The circulatory system
THE HEART OBJECTIVES: LOCATION OF THE HEART IN THE THORACIC CAVITY CARDIOVASCULAR SYSTEM
 BIOLOGY II CARDIOVASCULAR SYSTEM ACTIVITY #3 NAME DATE HOUR THE HEART OBJECTIVES: Describe the anatomy of the heart and identify and give the functions of all parts. (pp. 356 363) Trace the flow of blood
BIOLOGY II CARDIOVASCULAR SYSTEM ACTIVITY #3 NAME DATE HOUR THE HEART OBJECTIVES: Describe the anatomy of the heart and identify and give the functions of all parts. (pp. 356 363) Trace the flow of blood
Cardiovascular System
 Cardiovascular System angio BELLWORK Day One: Define using technology hemo/hema cardio Medical Therapeutics Standards 11) Outline the gross normal structure and function of all body systems and summarize
Cardiovascular System angio BELLWORK Day One: Define using technology hemo/hema cardio Medical Therapeutics Standards 11) Outline the gross normal structure and function of all body systems and summarize
The Cardiovascular System
 11 PART A The Cardiovascular System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Cardiovascular
11 PART A The Cardiovascular System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Cardiovascular
All About the Heart. Structures of the heart. Layers. Chambers
 All About the Heart Your heart is a muscle. It is slightly larger than your fist and weighs less than a pound. It is located to the left of the middle of your chest. Your heart pumps blood to the lungs
All About the Heart Your heart is a muscle. It is slightly larger than your fist and weighs less than a pound. It is located to the left of the middle of your chest. Your heart pumps blood to the lungs
The Function. To carry nutrients and oxygen to and remove waste from the cells of the body.
 The Function To carry nutrients and oxygen to and remove waste from the cells of the body. What makes up the circulatory system? 1. Heart 2. Blood 3. Blood vessels Blood travels from the heart to the body
The Function To carry nutrients and oxygen to and remove waste from the cells of the body. What makes up the circulatory system? 1. Heart 2. Blood 3. Blood vessels Blood travels from the heart to the body
PHYSIOEX 3.0 EXERCISE 33B: CARDIOVASCULAR DYNAMICS
 PHYSIOEX 3.0 EXERCISE 33B: CARDIOVASCULAR DYNAMICS Objectives 1. To define the following: blood flow; viscosity; peripheral resistance; systole; diastole; end diastolic volume; end systolic volume; stroke
PHYSIOEX 3.0 EXERCISE 33B: CARDIOVASCULAR DYNAMICS Objectives 1. To define the following: blood flow; viscosity; peripheral resistance; systole; diastole; end diastolic volume; end systolic volume; stroke
Cardiovascular System. Heart Anatomy
 Cardiovascular System Heart Anatomy 1 The Heart Location & general description: Atria vs. ventricles Pulmonary vs. systemic circulation Coverings Walls The heart is found in the mediastinum, the medial
Cardiovascular System Heart Anatomy 1 The Heart Location & general description: Atria vs. ventricles Pulmonary vs. systemic circulation Coverings Walls The heart is found in the mediastinum, the medial
Non-Newtonian pulsatile blood flow in a modeled artery with a stenosis and an aneurysm
 Non-Newtonian pulsatile blood flow in a modeled artery with a stenosis and an aneurysm I. Husain, C. Langdon and J. Schwark Department of Mathematics Luther College University of Regina Regina, Saskatchewan
Non-Newtonian pulsatile blood flow in a modeled artery with a stenosis and an aneurysm I. Husain, C. Langdon and J. Schwark Department of Mathematics Luther College University of Regina Regina, Saskatchewan
The Cardiovascular System
 CHAPTER 16 1 SECTION Circulation and Respiration The Cardiovascular System California Science Standards 7.5.a, 7.5.b, 7.6.j BEFORE YOU READ After you read this section, you should be able to answer these
CHAPTER 16 1 SECTION Circulation and Respiration The Cardiovascular System California Science Standards 7.5.a, 7.5.b, 7.6.j BEFORE YOU READ After you read this section, you should be able to answer these
IB TOPIC 6.2 THE BLOOD SYSTEM
 IB TOPIC 6.2 THE BLOOD SYSTEM TERMS TO KNOW circulation ventricle artery vein THE BLOOD SYSTEM 6.2.U1 - Arteries convey blood at high pressure from the ventricles to the tissues of the body Circulation
IB TOPIC 6.2 THE BLOOD SYSTEM TERMS TO KNOW circulation ventricle artery vein THE BLOOD SYSTEM 6.2.U1 - Arteries convey blood at high pressure from the ventricles to the tissues of the body Circulation
The Cardiovascular System
 Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 11 The Cardiovascular System Slides 11.1 11.19 Lecture Slides in PowerPoint by Jerry L. Cook The Cardiovascular System
Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 11 The Cardiovascular System Slides 11.1 11.19 Lecture Slides in PowerPoint by Jerry L. Cook The Cardiovascular System
the Cardiovascular System I
 the Cardiovascular System I By: Dr. Nabil A Khouri MD, MsC, Ph.D MEDIASTINUM 1. Superior Mediastinum 2. inferior Mediastinum Anterior mediastinum. Middle mediastinum. Posterior mediastinum Anatomy of
the Cardiovascular System I By: Dr. Nabil A Khouri MD, MsC, Ph.D MEDIASTINUM 1. Superior Mediastinum 2. inferior Mediastinum Anterior mediastinum. Middle mediastinum. Posterior mediastinum Anatomy of
Major Function of the Cardiovascular System. Transportation. Structures of the Cardiovascular System. Heart - muscular pump
 Structures of the Cardiovascular System Heart - muscular pump Blood vessels - network of tubes Blood - liquid transport vehicle brachiocephalic trunk superior vena cava right pulmonary arteries right pulmonary
Structures of the Cardiovascular System Heart - muscular pump Blood vessels - network of tubes Blood - liquid transport vehicle brachiocephalic trunk superior vena cava right pulmonary arteries right pulmonary
IB TOPIC 6.2 THE BLOOD SYSTEM
 IB TOPIC 6.2 THE BLOOD SYSTEM THE BLOOD SYSTEM TERMS TO KNOW circulation ventricle artery vein 6.2.U1 - Arteries convey blood at high pressure from the ventricles to the tissues of the body Circulation
IB TOPIC 6.2 THE BLOOD SYSTEM THE BLOOD SYSTEM TERMS TO KNOW circulation ventricle artery vein 6.2.U1 - Arteries convey blood at high pressure from the ventricles to the tissues of the body Circulation
Chapter 20 (1) The Heart
 Chapter 20 (1) The Heart Learning Objectives Describe the location and structure of the heart Describe the path of a drop of blood from the superior vena cava or inferior vena cava through the heart out
Chapter 20 (1) The Heart Learning Objectives Describe the location and structure of the heart Describe the path of a drop of blood from the superior vena cava or inferior vena cava through the heart out
1. Label the Diagram using the following terms: artery, arterioles, vein, venules, capillaries, valve, inner wall, middle wall, outer wall
 Bio 20 Ms. Nyboer Arteries, Veins, Capillaries, and the Heart Structure and Function Workbook Use your textbook (Ch. 10) and notes to fill in this workbook Part A: Arteries, Veins, Capillaries 1. Label
Bio 20 Ms. Nyboer Arteries, Veins, Capillaries, and the Heart Structure and Function Workbook Use your textbook (Ch. 10) and notes to fill in this workbook Part A: Arteries, Veins, Capillaries 1. Label
Anatomy of the Heart
 Biology 212: Anatomy and Physiology II Anatomy of the Heart References: Saladin, KS: Anatomy and Physiology, The Unity of Form and Function 8 th (2018). Required reading before beginning this lab: Chapter
Biology 212: Anatomy and Physiology II Anatomy of the Heart References: Saladin, KS: Anatomy and Physiology, The Unity of Form and Function 8 th (2018). Required reading before beginning this lab: Chapter
Lab 16. The Cardiovascular System Heart and Blood Vessels. Laboratory Objectives
 Lab 16 The Cardiovascular System Heart and Blood Vessels Laboratory Objectives Describe the anatomical structures of the heart to include the pericardium, chambers, valves, and major vessels. Describe
Lab 16 The Cardiovascular System Heart and Blood Vessels Laboratory Objectives Describe the anatomical structures of the heart to include the pericardium, chambers, valves, and major vessels. Describe
4. The two inferior chambers of the heart are known as the atria. the superior and inferior vena cava, which empty into the left atrium.
 Answer each statement true or false. If the statement is false, change the underlined word to make it true. 1. The heart is located approximately between the second and fifth ribs and posterior to the
Answer each statement true or false. If the statement is false, change the underlined word to make it true. 1. The heart is located approximately between the second and fifth ribs and posterior to the
CIRCULATORY SYSTEM TASK CARDS Worksheet
 CIRCULATORY SYSTEM TASK CARDS Worksheet Name: Date: Instructions: Put the answers to each task card in the numbered boxes on the chart. 1 a) left semilunar valve / aortic valve b) blood would backflow
CIRCULATORY SYSTEM TASK CARDS Worksheet Name: Date: Instructions: Put the answers to each task card in the numbered boxes on the chart. 1 a) left semilunar valve / aortic valve b) blood would backflow
Cardiovascular System. Supplementary Information
 Cardiovascular System Supplementary Information THE CARDIOVASCULAR SYSTEM - 1 - THE CARDIOVASCULAR SYSTEM FUNCTION Transport system carrying nutrient, gases, hormones and waste products to and from the
Cardiovascular System Supplementary Information THE CARDIOVASCULAR SYSTEM - 1 - THE CARDIOVASCULAR SYSTEM FUNCTION Transport system carrying nutrient, gases, hormones and waste products to and from the
Blood Flow, Blood Pressure, Cardiac Output. Blood Vessels
 Blood Flow, Blood Pressure, Cardiac Output Blood Vessels Blood Vessels Made of smooth muscle, elastic and fibrous connective tissue Cells are not electrically coupled Blood Vessels Arteries arterioles
Blood Flow, Blood Pressure, Cardiac Output Blood Vessels Blood Vessels Made of smooth muscle, elastic and fibrous connective tissue Cells are not electrically coupled Blood Vessels Arteries arterioles
The Circulatory System
 The Circulatory System Science Matters Chapter 8 Introduction Living things need a transport system to carry things around the body. In humans its called The Circulatory system. The parts of the system
The Circulatory System Science Matters Chapter 8 Introduction Living things need a transport system to carry things around the body. In humans its called The Circulatory system. The parts of the system
Do Now. Get out work from last class to be checked
 Do Now Get out work from last class to be checked Heart Actions Cardiac Cycle: One complete heartbeat. The contraction of a heart chamber is called systole and the relaxation of a chamber is called diastole.
Do Now Get out work from last class to be checked Heart Actions Cardiac Cycle: One complete heartbeat. The contraction of a heart chamber is called systole and the relaxation of a chamber is called diastole.
Children with Single Ventricle Physiology: The Possibilities
 Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
ANATOMY AND PHYSIOLOGY HOMEWORK CHAPTER 11 AND 12
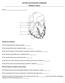 ANATOMY AND PHYSIOLOGY HOMEWORK CHAPTER 11 AND 12 Name Identify the following: 1) The Purkinje fibers are indicated by label. 2) The sinoatrial (SA) node is indicated by letter. 3) The specific chamber
ANATOMY AND PHYSIOLOGY HOMEWORK CHAPTER 11 AND 12 Name Identify the following: 1) The Purkinje fibers are indicated by label. 2) The sinoatrial (SA) node is indicated by letter. 3) The specific chamber
Circulatory Systems. All cells need to take in nutrients and expel metabolic wastes.
 Circulatory Systems All cells need to take in nutrients and expel metabolic wastes. Single celled organisms: nutrients from the environment can diffuse (or be actively transported) directly in to the cell
Circulatory Systems All cells need to take in nutrients and expel metabolic wastes. Single celled organisms: nutrients from the environment can diffuse (or be actively transported) directly in to the cell
Chapter 2 The Human Cardiovascular System
 Chapter 2 The Human Cardiovascular System 2.1 Introduction Before delving into the computational methods of CHD, this chapter provides a preliminary understanding of the circulatory system from a physiological
Chapter 2 The Human Cardiovascular System 2.1 Introduction Before delving into the computational methods of CHD, this chapter provides a preliminary understanding of the circulatory system from a physiological
End of chapter exercises
 End of chapter exercises Problem 1: The following diagrams show the heart during the cardiac cycle. The arrows represent the flow of blood. Study the diagrams and answer the questions that follow: Figure
End of chapter exercises Problem 1: The following diagrams show the heart during the cardiac cycle. The arrows represent the flow of blood. Study the diagrams and answer the questions that follow: Figure
Lab Activity 23. Cardiac Anatomy. Portland Community College BI 232
 Lab Activity 23 Cardiac Anatomy Portland Community College BI 232 Cardiac Muscle Histology Branching cells Intercalated disc: contains many gap junctions connecting the adjacent cell cytoplasm, creates
Lab Activity 23 Cardiac Anatomy Portland Community College BI 232 Cardiac Muscle Histology Branching cells Intercalated disc: contains many gap junctions connecting the adjacent cell cytoplasm, creates
Circulation. Circulation = is a process used for the transport of oxygen, carbon! dioxide, nutrients and wastes through-out the body
 Circulation Circulation = is a process used for the transport of oxygen, carbon! dioxide, nutrients and wastes through-out the body Heart = muscular organ about the size of your fist which pumps blood.
Circulation Circulation = is a process used for the transport of oxygen, carbon! dioxide, nutrients and wastes through-out the body Heart = muscular organ about the size of your fist which pumps blood.
CPM Specifications Document Aortic Coarctation: Exercise
 CPM Specifications Document Aortic Coarctation: Exercise OSMSC 0091_2000 0102_2000 0107_0000 0111_0000 May 29, 2013 Version 1 Open Source Medical Software Corporation 2013 Open Source Medical Software
CPM Specifications Document Aortic Coarctation: Exercise OSMSC 0091_2000 0102_2000 0107_0000 0111_0000 May 29, 2013 Version 1 Open Source Medical Software Corporation 2013 Open Source Medical Software
Anatomy Review: The Heart Graphics are used with permission of A.D.A.M. Software, Inc. and Benjamin/Cummings Publishing Co.
 Anatomy Review: The Heart Graphics are used with permission of A.D.A.M. Software, Inc. and Benjamin/Cummings Publishing Co. Anatomy Views Label the diagrams of the heart below: Interactive Physiology Study
Anatomy Review: The Heart Graphics are used with permission of A.D.A.M. Software, Inc. and Benjamin/Cummings Publishing Co. Anatomy Views Label the diagrams of the heart below: Interactive Physiology Study
CPM Specifications Document Fontan Aorta - Exercise:
 CPM Specifications Document Fontan Aorta - Exercise: OSMSC 0063_1000, 0064_1000, 0065_1000, 0075_1000, 0076_1000, 0077_1000 May 1, 2013 Version 1 Open Source Medical Software Corporation 2013 Open Source
CPM Specifications Document Fontan Aorta - Exercise: OSMSC 0063_1000, 0064_1000, 0065_1000, 0075_1000, 0076_1000, 0077_1000 May 1, 2013 Version 1 Open Source Medical Software Corporation 2013 Open Source
Unit 6: Circulatory System. 6.2 Heart
 Unit 6: Circulatory System 6.2 Heart Functions of Circulatory System 1. The heart is the pump necessary to circulate blood to all parts of the body 2. Arteries, veins and capillaries are the structures
Unit 6: Circulatory System 6.2 Heart Functions of Circulatory System 1. The heart is the pump necessary to circulate blood to all parts of the body 2. Arteries, veins and capillaries are the structures
14-1: The Body s Transport System. 7 th Grade Life Science
 7 th Grade Life Science Cardiovascular System The cardiovascular system, also called circulatory system, is made of your heart blood vessels, and blood. It is like the network of highways on Earth, where
7 th Grade Life Science Cardiovascular System The cardiovascular system, also called circulatory system, is made of your heart blood vessels, and blood. It is like the network of highways on Earth, where
How does the Circulatory System maintain
 How does the Circulatory System maintain -It supplies cells throughout the body with oxygen and nutrients while also ridding them of wastes. Transports materials such as: (to cells) (to attack foreign
How does the Circulatory System maintain -It supplies cells throughout the body with oxygen and nutrients while also ridding them of wastes. Transports materials such as: (to cells) (to attack foreign
THE VESSELS OF BLOOD CIRCULATION
 THE VESSELS OF BLOOD CIRCULATION scientistcindy.com /the-vessels-of-blood-circulation.html NOTE: You should familiarize yourself with the anatomy of the heart and have a good understanding of the flow
THE VESSELS OF BLOOD CIRCULATION scientistcindy.com /the-vessels-of-blood-circulation.html NOTE: You should familiarize yourself with the anatomy of the heart and have a good understanding of the flow
Bellwork Define: hemostasis anticoagulation hemophilia (Then write the underline portion of the two state standards in your notes).
 Bellwork Define: hemostasis anticoagulation hemophilia (Then write the underline portion of the two state standards in your notes). A&P Standards 31) Identify the liquid and cellular components of blood
Bellwork Define: hemostasis anticoagulation hemophilia (Then write the underline portion of the two state standards in your notes). A&P Standards 31) Identify the liquid and cellular components of blood
The cardiovascular system is composed of the heart and blood vessels that carry blood to and from the body s organs. There are 2 major circuits:
 1 The cardiovascular system is composed of the heart and blood vessels that carry blood to and from the body s organs. There are 2 major circuits: pulmonary and systemic. The pulmonary goes out to the
1 The cardiovascular system is composed of the heart and blood vessels that carry blood to and from the body s organs. There are 2 major circuits: pulmonary and systemic. The pulmonary goes out to the
The Cardiovascular System
 Essentials of Human Anatomy & Physiology Elaine N. Marieb Slides 11.1 11.19 Seventh Edition Chapter 11 The Cardiovascular System Functions of the Cardiovascular system Function of the heart: to pump blood
Essentials of Human Anatomy & Physiology Elaine N. Marieb Slides 11.1 11.19 Seventh Edition Chapter 11 The Cardiovascular System Functions of the Cardiovascular system Function of the heart: to pump blood
Cardiovascular. Function of the cardiovascular system is to transport blood containing: Nutrients Waste Hormones Immune cells Oxygen
 Cardiovascular The Cardiovascular System - Arteries Arteries Cardiovascular System Function of the cardiovascular system is to transport blood containing: Carry blood away from heart Carotid arteries Deliver
Cardiovascular The Cardiovascular System - Arteries Arteries Cardiovascular System Function of the cardiovascular system is to transport blood containing: Carry blood away from heart Carotid arteries Deliver
11/10/2014. Muscular pump Two atria Two ventricles. In mediastinum of thoracic cavity 2/3 of heart's mass lies left of midline of sternum
 It beats over 100,000 times a day to pump over 1,800 gallons of blood per day through over 60,000 miles of blood vessels. During the average lifetime, the heart pumps nearly 3 billion times, delivering
It beats over 100,000 times a day to pump over 1,800 gallons of blood per day through over 60,000 miles of blood vessels. During the average lifetime, the heart pumps nearly 3 billion times, delivering
Cardiovascular system
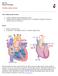 BIO 301 Human Physiology Cardiovascular system The Cardiovascular System: consists of the heart plus all the blood vessels transports blood to all parts of the body in two 'circulations': pulmonary (lungs)
BIO 301 Human Physiology Cardiovascular system The Cardiovascular System: consists of the heart plus all the blood vessels transports blood to all parts of the body in two 'circulations': pulmonary (lungs)
CFD Analysis of Pulsatile Flow and Non-Newtonian Behavior of Blood in Arteries
 Copyright 2015 Tech Science Press MCB, vol.12, no.1, pp.37-47, 2015 CFD Analysis of Pulsatile Flow and Non-Newtonian Behavior of Blood in Arteries P. Jhunjhunwala,, P.M. Padole, and S.B. Thombre, Abstract:
Copyright 2015 Tech Science Press MCB, vol.12, no.1, pp.37-47, 2015 CFD Analysis of Pulsatile Flow and Non-Newtonian Behavior of Blood in Arteries P. Jhunjhunwala,, P.M. Padole, and S.B. Thombre, Abstract:
