On the chordae structure and dynamic behaviour of the mitral valve
|
|
|
- Lesley Gray
- 5 years ago
- Views:
Transcription
1 IMA Journal of Applied Mathematics (2018) 00, 1 26 doi: /imamat/hxy035 On the chordae structure and dynamic behaviour of the mitral valve Liuyang Feng School of Mathematics and Statistics, University of Glasgow, UK Nan Qi Institute of Marine Science and Technology, Shandong University, Shandong, China and School of Mathematics and Statistics, University of Glasgow, UK Hao Gao School of Mathematics and Statistics, University of Glasgow, UK Wei Sun Wallace H Coulter Department of Biomedical Engineering, Georgia Institute of Technology, Atlanta, GA, USA Mariano Vazquez Barcelona Supercomputing Centre, IIIA-CSIC, Spain Boyce E. Griffith Departments of Mathematics, Applied Physical Sciences, and Biomedical Engineering and McAllister Heart Institute, University of North Carolina, Chapel Hill, NC, USA and Xiaoyu Luo School of Mathematics and Statistics, University of Glasgow, UK Corresponding author: xiaoyu.luo@glasgow.ac.uk [Received on 8 December 2017; revised on 29 June 2018; accepted on 29 July 2018] We develop a fluid structure interaction (FSI) model of the mitral valve (MV) that uses an anatomically and physiologically realistic description of the MV leaflets and chordae tendineae. Three different chordae models complex, pseudo-fibre and simplified chordae are compared to determine how different chordae representations affect the dynamics of the MV. The leaflets and chordae are modelled as fibrereinforced hyperelastic materials, and FSI is modelled using an immersed boundary finite element method. The MV model is first verified under static boundary conditions against the commercial finite element software ABAQUS and then used to simulate MV dynamics under physiological pressure conditions. Interesting flow patterns and vortex formulation are observed in all three cases. To quantify the highly complex system behaviour resulting from FSI, an energy budget analysis of the coupled MV FSI model is performed. Results show that the complex and pseudo-fibre chordae models yield good valve closure during systole but that the simplified chordae model leads to poorer leaflet coaptation and an unrealistic bulge in the anterior leaflet belly. An energy budget analysis shows that the MV models joint first author The Author(s) Published by Oxford University Press on behalf of the Institute of Mathematics and its Applications. This is an Open Access article distributed under the terms of the Creative Commons Attribution License ( which permits unrestricted reuse, distribution, and reproduction in any medium, provided the original work is properly cited.
2 2 L. FENG ET AL. with complex and pseudo-fibre chordae have similar energy distribution patterns but the MV model with the simplified chordae consumes more energy, especially during valve closing and opening. We find that the complex chordae and pseudo-fibre chordae have similar impact on the overall MV function but that the simplified chordae representation is less accurate. Because a pseudo-fibre chordal structure is easier to construct and less computationally intensive, it may be a good candidate for modelling MV dynamics or interaction between the MV and heart in patient-specific applications. Keywords: mitral valve; chordae tendineae; fluid structure interaction; immersed boundary method; finite element method;. 1. Introduction Mitral valve (MV) dysfunction, including MV stenosis, prolapse and regurgitation, is one of the most common valvular heart diseases and hence has attracted significant research interest. Computational modelling of human MV function (Gao et al., 2017b) can improve our understanding of MV biomechanics (Kunzelman et al., 1997, 1998), which is important for improving surgical procedures (Cochran & Kunzelman, 1998; Reimink et al., 1996) and medical therapies (Kheradvar et al., 2015). However, because of the challenges of modelling the highly complex MV structure, its deformation and its interaction with the left ventricle, only limited progress in multi-physics modelling of the MV has been made to date (Einstein et al., 2010; Gao et al., 2017b; Sun et al., 2014). Approaches to modelling the MV often fall into two categories: structural analysis and fluid structure interaction (FSI) analysis. The former is simpler and focuses on MV deformation in its fully closed state. The latter focuses on the whole cardiac cycle and is more computationally demanding but provides a more complete description of valvular function. Both approaches have been used to study MV dynamics (Einstein et al., 2010; Gao et al., 2014a; Kunzelman et al., 1993, 1997; Kunzelman & Cochran, 1992; Luo et al., 2012; Ma et al., 2013; Watton et al., 2008). For a recent review of different MV modelling strategies, the reader is referred to Gao et al. (2017b). In this study, we use an immersed boundary finite element (IB/FE) method (Griffith & Luo, 2017) to develop a dynamic MV FSI model. The chordae tendineae in the MV apparatus, which connect the papillary muscle to MV leaflets, play an important role in MV function, especially in the systolic phase to prevent the leaflets prolapse. Studies have shown that chordae structure has a substantial impact on MV function (David et al., 1984; Espino et al., 2005; Reimink et al., 1996). For example, David et al. (1984) found that to repair chronic MV regurgitation, post-operative left ventricular function benefits from preserving the chordae structure and papillary muscles when replacing the MV. Espino et al. (2005) also confirmed that anterior leaflet marginal chordae as well as commissural chordae are vital for MV competence. However, few studies have investigated the difference in MV function between different chordae structures with FSI. In our previous studies, we used various simplified chordae structures, including discrete elastic pseudofibre models (Luo et al., 2012; Ma et al., 2013; Watton et al., 2008) and fully 3D volumetric models (Gao et al., 2017a, 2014a). However, those simplified chordae structures did not distinguish the marginal, basal and strut chordae. As a result, our modelled anterior leaflet bulged towards the left atrium, which is a common artefact of many existing MV models. This study aims to overcome these defects and to investigate how different chordae representations affect valvular function. To do so, we use a physiologically detailed description of the MV leaflets and chordae structure based on multi-slice computed tomography (CT) image data (Wang & Sun, 2013). In addition, we consider three different chordae structure representations: complex, pseudo-fibre and simplified. The complex model uses a fully three-dimensional chordae structure based on the CT images,
3 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 3 in which the marginal, basal and strut chordae are all included. The pseudo-fibre model is based on the same CT data but represented using thin elastic fibres, as in a more conventional IB approach (Ma et al., 2013). The simplified chordae structure is based on our previous idealized chordae descriptions (Gao et al., 2014a), in which the chordae are also modelled using a fully three-dimensional volumetric solid mesh. In all these cases, anisotropic material laws are used for both the MV leaflets and the chordae. The paper is organized as follows: Sections 2 and 3 provide details of the IB/FE method and material properties. Section 4 describes verification studies of our IB/FE model against the commercial FE package ABAQUS ( in steady-state conditions. Sections 5 and 6 provide implementation details and describe results for FSI simulations. We compare the MV model behaviour with different chordae structures and comparisons of MV orifice area during systolic contraction of the left ventricle (LV), and fluid flow patterns during diastolic filling of the LV phase are made among those three chordae structures. We also analyse energy budget for the fully coupled MV-flow model including detailed energy distribution and its dissipation. Section 7 discusses these results and provides a concluding discussion. 2. Methodology 2.1 IB/FE formulation In the IB/FE formulation of FSI used in this study (Griffith & Luo, 2017), the structure elasticity and deformation are described in Lagrangian coordinates, and the pressure, velocity and incompressibility of the coupled fluid structure system are described in Eulerian coordinates. Let Ω R 3 denote the fixed physical domain occupied by the coupled fluid structure system, and let Ω0 s Ω be the Lagrangian reference coordinate domain for structure. x U are fixed Cartesian physical coordinates, X Ω0 s are the fixed Lagrangian reference coordinates of the structure and χ(x, t) U is the current position of material point X at time t. Thus, the region occupied by structure at time t is Ωt s = χ(ω0 s, t), and Ωt f = Ω\Ωt s is the region occupied by the fluid at time t. The Cauchy stress tensor is divided into the following two parts, σ (x, t) = { σ f (x, t) + σ e (x, t), x Ωt s, σ f (x, t), otherwise. (1) Here, σ f (x, t) is the Cauchy stress tensor for a viscous incompressible fluid, σ f (x, t) = pi + μ( u + u T ), (2) in which p and u are the material pressure and velocity, and μ is the dynamic viscosity of the fluid. The Cauchy elastic stress tensor σ e (x, t) can be related to the first Piola Kirchhoff stress tensor P e using Nanson s formula, P e (X, t)n da(x) = σ e (x, t)n da(x), (3) V χ(v,t) in which V Ω0 s is an arbitrary Lagrangian subregion, N is the outward unit vector along V and n is the outward unit vector along χ(v, t).
4 4 L. FENG ET AL. The equations for the IB/FE formulation of FSI used in this study are (Griffith & Luo, 2017) ( ) u(x, t) ρ + u(x, t) u(x, t) = p(x, t) + μ 2 u(x, t) + f(x, t), t (4) u(x, t) = 0, (5) f(x, t) = F(X, t)δ(x χ(x, t)) dx, (6) Ω0 s F(X, t) V(X) dx = P e (X, t) : X V(X) dx, V(X), (7) Ω0 s Ω0 s χ (X, t) = u(χ(x, t), t) = u(x, t)δ(χ(x, t) x) dx, t Ω (8) with V(X) an arbitrary Lagrangian test function. Equation (4) is the momentum equation for the coupled system, (5) is the incompressibility constraint and (6) and (8) are interaction equations that connect the Lagrangian and Eulerian coordinate systems, which use the delta function δ(x) to transfer quantities between the two frames. For problems discussed here, ρ and μ are assumed constants throughout the system. Equation (6) relates the Lagrangian elastic force density F(X, t) and the corresponding Eulerian elastic force density f(x, t). Equation (8) implies that the no-slip condition is satisfied at the fluid solid interface. We remark that although we used two separate set of meshes, one Eulerian and one Lagrangian, only the Eulerian momentum equation is solved for the coupled system, because the Eulerian momentum equation accounts for the momentum of the entire computational domain Ω = Ωt f Ωs t. The applied forces ensure traction continuity at the fluid solid interface. 2.2 Energy budget analysis Now we introduce the energy budget analysis for the coupled FSI model. The energy balance of the FSI system can be written as ( ) u ( ) ρ u dx + ρ(u )u u dx = p + μ 2 u u dx + f u dx, (9) Ω tube t Ω tube Ω tube Ω tube in which Ω tube is the interior of the stationary tube in which the MV is mounted (Fig. 1). Equation (9) consists of the change of the kinetic energy, KE = Ω tube ρ the kinetic energy flux of fluid on the boundary U, KF = ρ(u )u u dx = Ω tube the rate of work by the applied pressure, WP = ( p) u dx = Ω tube ( ) u u dx = d ( ) 1 ρu u dx, (10) t dt Ω tube 2 U U 1 ρ(u u)u n da, (11) 2 ( p) n u da, (12)
5 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 5 Fig. 1. Schematic illustration of the MV-fluid coupled system. the rate of energy dissipation, D = μ 2 u u dx = μ( u + u T ) : u dx, (13) Ω tube Ω tube and the rate of change of elastic strain energy in the immersed structure, ( ) E = f u dx = F(X)δ(x χ(x, t)) dx u dx (14) Ω tube Ω tube Ω0 s ( ) = u(x, t)δ(x χ(x, t)) dx F(X) dx (15) = Ω tube Ω s 0 Ω s 0 u(χ(x, t), t) F(X) dx, (16) which can be rewritten as with the help of (7). E = P e (X, t) : X u(χ(x, t), t) dx, (17) Ω0 s 2.3 Spatial and temporal discretizations A Cartesian grid is used to discretize the Eulerian domain U. We use a staggered grid to approximate the Eulerian velocity u = (u x, u y, u z ) and Eulerian force density f = (f x, f y, f z ). Standard secondorder accurate finite difference approximations to the divergence, gradient and Laplace operators
6 6 L. FENG ET AL. Fig. 2. MV models with (a) complex chordae, (b) pseudo-fibre chordae and (c) simplified chordae. In (a) the struct chordae are coloured in red, the marginal chordae are coloured in blue and the basal chordae are coloured in green (see online version). In (b) and (c), only one chordae type is considered. (Griffith, 2009) are used. The nonlinear advection terms in (4) are discretized using a version of the piecewise parabolic method (Colella & Woodward, 1984). We discretize the Lagrangian domain using the finite element method with trilinear (Q1 ) basis functions for the displacement and resultant force. Additional details of the discretization can be found in Griffith & Luo (2017). 3. MV geometry and material properties Our MV model includes a detailed MV anatomical geometry obtained from multi-slice CT images of a normal MV at mid-diastole (when the MV is open) obtained from a 61-year-old male patient (Wang & Sun, 2013). Three different models are built, as shown in Fig. 2. All three models have the same leaflets but different chordae representations. The first one, shown in Fig. 2(a), is referred to as the complex model. It has a CT-derived chordal structure that includes the marginal, basal and strut chordae, with a fork-shaped geometry at chordae leaflet connection to avoid stress concentrations. The chordae are modelled as fully three-dimensional solid bodies using a volumetric mesh discretization with a uniform cross-sectional area of 0.16 mm2 (Gao et al., 2014a). The second model, shown in Fig. 2(b), is based on the same CT scan data, but here the chordae are represented as one-dimensional elastic fibres, as in more conventional IB approaches (Ma et al., 2013). The chordae follow the same structure as in the complex model, including all three types of chordae, and a fork-shaped geometry at the chordae leaflet connection. In other words, the pseudo-fibres are the skeleton of the complex chordae derived from the CT scan data (Wang & Sun, 2013). We refer this as the pseudo-fibre model. The third model, shown in Fig. 2(c), uses a simplified chordae structure, as in our previous work (Gao et al., 2014a), which has a total of 16 evenly distributed chordae (6 on anterior leaflet and 10 on posterior leaflet) with the same uniform cross-sectional area of 0.16 mm2. The anterolateral and posterolateral papillary muscle groups are represented by two chordae attachment movements obtained from the average of chordae attachment positions from the complex model. These chordae are also modelled as volumetric solids and run through the leaflets from the free leaflet edges to the annulus ring. Details can be found in Gao et al. (2014a). This is referred to as the simplified chordae model.
7 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 7 Fig. 3. Two families of fibre directions shown by arrows on anterior, shown in (a) (blue online) and posterior, shown in (b) (red online) leaflet. Circumferential direction is shown by the dashed line. An invariant-based, anisotropic constitutive model based on the formulation proposed by Gasser et al. (2006) and Holzapfel et al. (2000) is used to describe the MV leaflets: W leaflet = C1 {exp[c2 (I1 3)] 1} + k1 2k2 β 2 exp k2 I4i 1 + log2 (I3 ), i=1 (18) in which I1 = trace(ft F), I3 = det(ft F) = J 2 and F = χ / X is the deformation gradient tensor. Here, β4 log2 (I3 ) is a volumetric energy that acts to penalize compressible deformations. We choose β = = max [(κi + (1 3κ)I ), 1] 500 kpa in our simulations. The modified fibre pseudo-invariants are I4i 1 4i and I4i = ei (FT F)ei, using the fibre directions ei shown in Fig. 3(a and b). Two families of fibres are included here, whose directions as well as the parameter values are directly from biaxial experimental is defined to data for a healthy human mitral leaflets (Wang & Sun, 2013). The modified invariant I4i ensure that only extended fibres contribute to the strain energy. In particular, the fibres do not support compressive loads. κ is a collagen fibre dispersion parameter, and C1, C2, k1 and k2 are the material parameters. The parameter values for the models are summarized in Table 1. We define the modified elastic stress tensor Pe via Peleaflet = 2C1 C2 exp[c2 (I1 3)]F 2C1 C2 exp[c2 (I1 3)]F T 2 + 2k1 κ(i4i 1)exp k2 I4i 2 1 F+ i= k1 (1 3κ)(I4i 1) exp k2 I4i 1 Fei ei i=1 T + β log(i3 )F. (19) The term 2C1 C2 exp[c2 (I1 3)]F T is included to ensure Peleaflet is zero when F = I. This modification is found to reduce the magnitude of spurious volume loss caused by the pressure discontinuities across the structure fluid interface (Gao et al., 2014b). In all models of the chordae tendinae, the elasticity of the chordal material is described by W chordae = C(I1 3) + β α1 exp [α2 (I4 1)2 ] 1 + log2 (I3 ), 2α2 4 (20)
8 8 L. FENG ET AL. Table 1 Material parameters of MV leaflets, from Wang & Sun (2013) C 1 (kpa) C 2 k 1 (kpa) k 2 κ Anterior Posterior Table 2 Material parameters of MV chordae C (kpa) α 1 (kpa) α 2 Basal chordae Marginal chordae Struct chordae in which C, α 1 and α 2 are material parameters whose values are summarized in Table 2, and I 1, I 3 and I4 are similarly defined without dispersion, i.e. κ = 0. The fibre direction for all the chordae tendineae is simply defined along its longitudinal direction. For the complex model, all the branches have a fibre direction defined by following the long axis at the centre of the cross-section. As in the leaflets, we use β = 500 kpa. The resulting stress tensor is [ P e chordae = 2CF 2CF T + 2α 1 exp α 2 (I4 1)2] Fe e + β log(i 3 )F T. (21) Although the pseudo-fibre model describes each chord as a one-dimensional elastic fibre, the fibre model uses the same nonlinear material description as in (20), taking the cross-sectional area of the chords to be 0.04 cm 0.04 cm. The cross-sectional area is chosen so that the total force of the pseudofibres on the MV equals to the total force exerted by the solid chordae. 4. IB/FE model verification Detailed IB/FE model verification studies are performed on a disc geometry and the MV model with complex chordae under static boundary conditions. We compare the results obtained by the IB/FE model in static conditions to a corresponding structure-only model built in ABAQUS. The models are shown to be in good agreement; additional details are provided in Appendices A and B. 5. Dynamic MV-fluid model implementation In the coupled MV-fluid system, the MV is mounted on a fixed housing structure that is placed inside a rigid tube with length 16 cm and radius 3.5 cm, as shown in Fig. 1. The whole structure sits inside an 8.6 cm 8.6 cm 16 cm computational domain filled with viscous fluid of density 1 g cm 3 and dynamic viscosity 0.04 g cm 1 s 1. The grid sizes for both fluid and solid are chosen so that good agreement is achieved in the verification problems (see Appendix for details). For the structural discretization, we use the same grid size for the leaflets, but because of the different treatment of the chordae, the total elements/nodes are / for the complex model, 97093/ for the pseudo-fibre model and 75292/92910 for the simplified model. For the Eulerian mesh, we use a twolevel block-structured adaptively refined grid with a refinement ratio 4 between the levels. The finest level has a grid of N x = 130, N y = 130 and N z = 200. Hence, the fluid mesh size is in the order of 0.07 cm 0.07 cm 0.08 cm. The time step size is chosen to be 5.0e-6 s.
9 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 9 Fig. 4. The inlet pressure profile with five phases: initialization, closing, fully closed, opening and fully open phases. We further define three time points: time A is when the MV is closing, time B is when MV is fully closed and under peak pressure (110 mmhg) and time C is when the MV is fully open. A physiological transvalvular pressure, shown in Fig. 4, is imposed at the inlet, and the outlet pressure is held zero. The modelled cardiac cycle includes the initialization phase, MV closing and fully closed phases, then MV opening and fully open phases. We also choose three time points (identified as times A, B and C as shown in Fig. 4) to perform a detailed analysis. To model the papillary muscle movements, the ends of chordae are constrained with displacement boundary conditions measured from experimental data similar to Wang & Sun (2013). The IB/FE scheme used in this study is implemented in the open-source IBAMR software ( which is an adaptive and distributed memory parallel implementation of the immersed boundary method leveraging several open-source computational frameworks, including SAMRAI ( PETSc ( libmesh ( and hypre ( to perform core functionality. The full simulations are run on the supercomputing services ARCHER ( ac.uk) and ARCHIE-WeSt ( Some early tests were also conducted at the Barcelona Supercomputing Centre ( A typical simulation of one cycle using 32 processors takes about 9 days in wall clock time on ARCHER or ARCHIE-WeSt. 6. Results 6.1 Structural deformation and fluid dynamics Figure 5 shows the deformation and the maximum principal strain and stress of the three MV models at time point B. Figure 5(a and b) shows that the complex and pseudo-fibre models generate similar closed valve configurations, but the simplified chordae model (Fig. 5(c)) is quite different. The maximum displacement of the MV leaflets for complex and pseudo-fibre models is 0.85 cm and
10 10 L. FENG ET AL. Fig. 5. Contours of the closed MV. Top row: by the displacement magnitude for MV models with (a) complex, (b) pseudo-fibre and (c) simplified chordae models. Middle row: the maximum principal strain for MV models with (d) complex, (e) pseudo-fibre and (f) simplified chordae. Bottom row: the maximum principal stress for MV models with (g) complex, (h) pseudo-fibre and (i) simplified chordae. See online version for colours cm, respectively. However, the anterior leaflet belly of the simplified model has an unrealistic large bulging into the left atrium, with a maximum displacement of 1.24 cm, which is shown in Fig. 6(c). All three models have a visible gap, which results from the regularization of the delta function (Gao et al., 2014a; Griffith et al., 2009; Ma et al., 2013); however, this gap is much larger in the simplified model, as shown in Fig. 5. The finger-like bumps in this case is resulted from the fact that the chordae run through the leaflets. In addition, MV orifice shape is compared between simulated results and segmented data from the same CT images mentioned in Section 3. Because of the image quality, only three phases in diastole can be segmented. Here, we choose the highest quality image and compare it to the complex chordae model. As shown in Figs 7 and 8, the simulated MV leaflets shape matches the CT scan reasonably well despite the fact that the segmented data doesn t have enough information
11 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 11 near the annulus ring, but it captures more details for the rest of leaflets. For example, the bumps near the chordae leaflet attachment are more pronounced in the segmented data than the simulated data. Fig. 6. Side view of the three models at time B: (a) the complex model, (b) the pseudo-fibre model and (c) the simplified model. Notice the substantial bulge in the anterior leaflet in (c) shown by an arrow. Fig. 7. Comparison of leaflet shapes at MV opening: (a) the complex model at time s and (b) the segmented MV data from CT scan. Fig. 8. Comparison of leaflet shapes at MV opening: (a) the complex model at time s and (b) the segmented MV data from CT scan. In all models, higher stress regions are found in the anterior leaflet belly region, close to the annulus ring (Fig. 5(g i)), which are in agreement with previous observations (Gao et al., 2014a; Ma et al.,
12 12 L. FENG ET AL. Table 3 Averaged maximum principle strain, stress and displacement of the anterior leaflet in a circular region defined in Fig. 5(a) Complex chordae Pseudo-fibre chordae Simplified chordae Maximum principal strain Maximum principal stress (kpa) Displacement (cm) Fig. 9. The maximum principal stress at time B: (a) complex model, (b) pseudo-fibre model and (c) simplified model. See online version for colours. 2013). However, notice that the simplified model has higher stress in the anterior leaflet than the other two models. We further compute the averaged maximum principle stress, strain and displacement of a circular region (radius 0.5 cm) in the anterior leaflet (Fig. 5(a)). The results are listed in Table 3. With the chordae stress included, the complex and pseudo-fibre models yield similar results, but the simplified model has a clear discrepancy. Figure 9(a c) show the maximum principal stress on chordae for all three models at time point B. The complex and pseudo-fibre models have very similar stress patterns: most of the chordae in the middle experience high stress whereas the chordae attached to commissure area tend to have less stress. For the simplified model, chordae attached to anterior leaflet have high stresses because of the large stretches, and near the leaflet commissures, high stresses also are observed. The average stresses of the chordae attached to the anterolateral and posterolateral papillary muscle groups are 1890 kpa and 2740 kpa, respectively, for the complex model; 2263 kpa and 2914 kpa for the pseudo-fibre model; and 3460 kpa and 3198 kpa for the simplified model. The simplified model has higher values than the other two MV models especially for the anterolateral muscle group. We also calculate the orifice area for each model by projecting the deformed leaflets onto the z = 0 plane. The results are plotted in Fig. 10. When the MV is opened at time C, the complex model has amaximumareaof5.21cm 2, while the pseudo-fibre and simplified models have slightly larger areas (maximum: 5.68 cm 2 and 5.58 cm 2 ) than the complex model. This is presumably because the pseudofibres do not constrain the MV as effectively as the solid chordae, and the simplified model does not have an effective chordae geometry, despite the fact that chordae run through the leaflets. When the MV is closed at time B, both the complex and pseudo-fibre chordae models have smaller orifice areas than the simplified chordae, as shown in Fig. 5. Further, by calculating the time that different models reach the same closure state (e.g. to an orifice area of 0.52 cm 2 at s), we can show that the complex and
13 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 13 Table 4 Fig. 10. Orifice area change of the three different models. MV orifice and closing speed comparison Complex chordae Pseudo-fibre chordae Simplified chordae Orifice area at s 4.93 cm cm cm 2 Time when orifice area = 0.52 cm s s s pseudo-fibre models have slightly faster MV closure speeds (about s faster) than the simplified model, as shown in Table 4. Figure 11 shows the flow rate through the MV. As suggested by the orifice area changes detailed in Fig. 10, faster MV closure leads to smaller regurgitant flow volumes: 8.66 ml for the complex model and 8.69 ml for the pseudo-fibre model. By contrast, the simplified model has a significantly higher regurgitation volume of ml. Figure 12 shows the streamlines of the three models at time C when the MV is fully open. Two flow jets are formed as shown by the arrows (Fig. 12(a, c and e)), one close to the anterior leaflet and the other close to the posterior leaflet. The complex and pseudo-fibre models have similar maximum filling velocities (227.1 cm/s and cm/s, respectively), and both are lower than that of the simplified model (260 cm/s). Figure 12(b, d and f) shows the corresponding flow vectors around the leaflets. It can be seen that all three models are associated with vortices behind the leaflet tips. Because the pseudo-fibre chordae are modelled as one-dimensional elastic fibres, in the continuum equations, they would not interact with the fluid. In practice, however, interaction between the flow and the chordae is mediated by regularized delta functions, and the finite extent of the delta function kernel imbues the chordae with an effective thickness. Further, at the present mesh resolutions, the Eulerian grid spacing is comparable to the cross-sectional dimensions of the pseudo-fibres. Consequently, from the standpoint of the fluid dynamics, the effective numerical thickness of the pseudo-fibre chordae is similar to that of the volumetric models. Under further grid refinement, it would be necessary to fix the support of the
14 14 L. FENG ET AL. Fig. 11. Flow rate through the MV of the three different models. regularized kernel function associated with the pseudo-fibre model, as done in prior work on modelling the interaction of thin rods with fluids using the generalized IB method (Griffith & Lim, 2012; Lim et al., 2008), to provide the pseudo-fibres with a fixed physical thickness. Figure 13 shows the pressure fields for the complex model at different time points. The other two models yield similar results. 6.2 Energy analysis To further quantify the dynamics of these different models, we perform an energy analysis. Figures 14 and 15 show the energy budget for the complex model. It is clear that in the fully open phase, kinetic energy dominates the energy distribution and consumes most of the input work (WP-KF). In the closing and opening phases, the MV strain energy (E) starts to have a bigger impact on the energy distribution. The large oscillations in E are associated with the strong motion of the MV during these phases. In the fully closed phase, the coupled system is settling down, and all energy terms gradually decay to zero. Similar energy budget patterns are found in the other two cases. Table 5 summarizes all the energy budget terms at time A (closing) and C (fully open). It shows that 80% of input work is used to increase the kinetic energy at time C and that the MV consumes 94% of the total energy from both the input work and the kinetic energy at time A. We further define the energy dissipation function, Φ = μ( u + u T ) : u, (22) and plot the dissipation field for the MV models at time C (fully open) in Fig. 16. We can see that high dissipation exists close to the MV leaflets and chordae structure. Energy is also dissipated at a higher rate close to the atrial side of the leaflet because of the high shear stress. Figure 16(g i) shows a scliced view
15 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 15 Fig. 12. Velocity streamlines and vector field of a cross-section, for the complex model (a and b), the pseudo-fibre model (c and d) and the simplified model (e and f). The two jet directions are shown by arrows, and the vortices around the leaflets can be seen in the circled areas. See online version for colours. of the dissipation field associated with the chordae. Overall, the complex and pseudo-fibre models have similar energy dissipation patterns near the chordae structure. However, the simplified model seems to cause a greater dissipation when the MV is opening.
16 16 L. FENG ET AL. Fig. 13. Pressure field for the complex model at (a) time A (closing), (b) time B (peak adverse pressure) and (c) time C (fully open). Fig. 14. Energy budget for the complex model during the initialization and closing. Figure 17 shows the total energy dissipation rate D against time. When the MV is closing or opening, D increases, as to be expected. Again, the complex and pseudo-fibre models have similar patterns, but the simplified model introduces a higher energy dissipation rate. On the other hand, during the MV closure phase, when the inlet pressure increases quickly, the pseudo-fibre model has the highest dissipation rate ( 0.31 J/s), compared to the complex and simplified ones, which are 0.23 J/s and 0.21 J/s, respectively. Figure 18 plots the rate of MV strain energy (E) for the three MV models in MV closing and fully closed phases; results in other phases are similar in all the models. It can be seen that the complex and pseudo-fibre models have similar patterns overall, with negative peaks of 3.43 J/s and 3.17 J/s,
17 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 17 Fig. 15. Energy budget for the complex model during fully closed, opening and fully open phases. Table 5 Energy budget (in J/s) for the complex model at times A and C WP-KF KE D E A (MV Closing) (6% of WP-KF-KE) (94% of WP-KF-KE) C (MV Fully Open) (80% of WP-KF) (13% ofwp-kf) (7.0% of WP-KF) respectively, when the MV is closing. However, the simplified model leads to much greater oscillations and a negative peak of 5.44 J/s. This is consistent to the fact that simplified chordae model undergoes a larger structural deformation, as is reflected in the displacement contours in Fig. 5(c). Figure 19 shows the input energy rate for the three models in the closing and fully closed phases; results in other phases are similar in all the models. As in Fig. 18, the complex and pseudo-fibre models have similar trends with maximum magnitudes: 2.07 J/s and 2.01 J/s, whereas the input energy rate of the simplified model is much greater (2.81 J/s). In other words, the simplified model requires a greater input energy compared to the other two MV models during the cardiac cycle. The model behaviours can also be seen in the integrated energy budgets. Table 6 shows the energy budgets integration during the period when the MV is closing (from to 0.25 s). The complex and pseudo-fibre models produce similar results. Part of the fluid kinetic energy is dissipated due to viscosity (40 and 46%), and the rest is transferred into the MV strain energy (43 and 38%) and to overcome the pressure loading (17 and 16%). However, the simplified model shows a totally different energy distribution: much more strain energy (60% of KE-P-F) is gained from not only the kinetic energy but also the external input energy. Compared to other two models, it also stores about twice as much strain energy during MV closing phase and requires more input energy.
18 18 L. FENG ET AL. Fig. 16. Isovolume plots for energy dissipation function (Φ) for (a) the complex model, (b) pseudo-fibre model and (c) simplified model. The corresponding 2D sliced views and contour plots around the chordal cross-sections of Φ are shown in (d, e and f) and (g, h and i), respectively. See online version for colours. 7. Discussion This paper describes an FSI model of the MV that uses an anatomically and physiologically realistic description of the MV leaflets and chordae tendineae that is based on an IB method with FE elasticity (Griffith & Luo, 2017). Our model is verified by comparing the structural results between the IB/FE model and a structure-only model implemented using ABAQUS. In this study, several advances have been made compared with our previous studies (Gao et al., 2014a; Ma et al., 2013): (1) we use physiologically detailed MV leaflets and chordae structures that are based on CT image data, (2) we compared three different chordae structure models and their effects on the MV function and (3) the FSI
19 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV Fig. 17. Dissipation Energy Rate (D) of the three models. Fig. 18. Rate of Strain Energy Change (E) in MV closing and fully closed phases of the three models. 19
20 20 L. FENG ET AL. Table 6 Integrated energy distribution (in J) over the MV closing phase WP-KF KE D E Complex chordae (17% of KE) (40% of KE) (43% of KE) Pseudo-fibre chordae (16% of KE) (46% of KE) (38% of KE) Simplified chordae (40% of KE-WP+KF) (60% of KE-WP+KF) Fig. 19. Total Input Energy Rate (WP-KF) in MV closing and fully closed phases of the three models. simulation of the MV dynamics also allows us to conduct an energy budget analysis of the dynamic MV and the flow for the first time. A realistic MV geometry is necessary for modelling MV dynamics, but it is almost impossible to reconstruct the chordae structure from low-resolution imaging data. Consequently, simplified MV models are widely used (Gao et al., 2014a; Kunzelman et al., 1997; Ma et al., 2013; Prot & Skallerud, 2009; Stevanella et al., 2009). However, researchers started to notice the limitation of such simplifications. For example, Wang & Sun (2013), using a purely structural model, showed that simplified chordae geometry leads to billowing leaflets and incomplete MV closure compared with complex chordae structures. With the help of advanced imaging techniques, more realistic chordae structures are now being reported (Lee et al., 2013, 2015; Toma et al., 2016; Wang & Sun, 2013). Our work goes further to compare the effects of three different chordae structure modelling approaches on overall MV function. In particular, because the chordae structure has often been represented by connections of elastic springs (Griffith et al., 2009; Luo et al., 2012; Ma et al., 2013) when simulating MV dynamics using immersed boundary method, it is interesting to compare the difference between the volumetric chordae and the pseudo-fibre-type representation.
21 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 21 The most notable result of this study is that the complex and pseudo-fibre chordae produce very similar dynamics and kinematics. However, the simplified chordae result in poorer leaflet coaptation and unrealistic bulging in the anterior leaflet, which is also seen in other MV modelling work (Skallerud et al., 2011; Stevanella et al., 2011). Further, compared to the simplified model, the complex and pseudo-fibre models tend to have a better performance in preventing the regurgitation, which can be seen from the orifice area profile (Fig. 10) and the volumetric flow rate (Fig. 11). It is also interesting to note that all of our MV models do not show prolapse, because of the realistic hyperelastic constitutive law used for the leaflets and volumetric structure model which automatically includes the bending stiffness. This is in agreement with our previous study (Luo et al., 2012), which showed that bending in the MV leaflets plays an important role. The complex and pseudo-fibre models also show consistent results with other published results of MV studies. Wang & Sun (2013) reported the average maximum principal stress is approximately 160 kpa on a midsection of anterior leaflet at the systolic peak. Kunzelman et al. (2007) used an FE model to find that the maximum principal stress on the anterior leaflet was 254 kpa with a transvalvular pressure of 98 mmhg. Gao et al. (2014a) also reported the peak average stress is 115 and 169 kpa, along the radial and circumferential directions, in the anterior belly region. These results are comparable to our model predictions, as shown in Table 3. The majority of MV modelling research focuses on the mechanics of the MV or the hemodynamics inside the left ventricle (Gao et al., 2014a, 2017b; Wang & Sun, 2013), and comparatively few reported studies have evaluated MV function from an energetic perspective (Choi et al., 2014). Akins et al. (2008) pointed out that evaluating energy loss during the cardiac cycle can help assess the impact of valve performance on cardiac function. Choi et al. (2014) also attempted to calculate the energy dissipation in a simplified LV-MV model to study the MV performance. Using the energy budget analyses reported in (10) (17), our work is the first effort to consider the whole cycle of the realistic MV model and FSI from an energetic perspective. This energy analysis reveals that when the MV is fully open, most of the input energy from left atrial contraction is used to increase the kinetic energy, which is then used to pump blood into the left ventricle from the left atrium. However, when the MV is closing or opening, the MV leaflet elastic energy becomes dominant. The energy distributions also show that the chordae structure plays an important role. In our simulations, even with different mesh elements, the complex chordae and pseudo-fibre chordae lead to very similar energy results, as seen in Figs Moreover, Table 6 shows that during early LV systolic phase until the time when the aortic valve opens, most of the kinetic energy is reduced via viscous dissipation and increases in the MV strain energy. However, the simplified chordae introduce more energy exchanges overall, especially when the MV is closing or opening, which can be seen from the larger magnitude peaks in Figs 18 and 19. Further, Table 6 shows that much more strain energy is stored in the simplified chordae, which may be partially responsible for the unrealistic bulging. As a result, the input energy from left ventricular contraction is also substantially increased in this model. The slower MV closing process in the simplified model also leads to a sign change in the input energy contribution in Table 6, indicating a different energy distribution from the other two models. One of the most important aspect in cardiac energy analysis is to see how energy loss (from viscosity, turbulence and flow separation) affect cardiac function, especially under pathological conditions. Although the energy dissipation term tends to have much smaller magnitude compared with other energy terms, this is the irreversible loss of the system and is thus important over the longer term. Figure 16 gives quantified description of energy loss for the fluid fields of different models. A higher dissipation rate is always found near the structure because of the high shear, particularly during opening. The simplified model shows a different energy dissipation pattern and leads to greater energy dissipation compared to other two models, which suggests decreased efficiency.
22 22 L. FENG ET AL. This study has several limitations. One of these is that homogeneous material properties are assumed for MV leaflets. Some studies (Kunzelman & Cochran, 1992; May-Newman & Yin, 1998) have suggested that this is not realistic, and regionally dependent material parameters should be considered in future works. Although we don t have sufficient experimental data at hand to directly compare with our CT-scan-based MV models at additional time points in the cardiac cycle, our results are consistent with published results of MV studies. Finally, we have shown that the energy budget analysis for the complex flow field has a great potential to provide useful pathological indicators; however, additional critical analyses will be required before it can be used for physical insights into differences between healthy and pathological cases. This requires extensive evaluation on how the energy budget is affected by sufficient number of pathological cases. Finally, we have not attempted to study pathological changes either in the chordae structure or the MV leaflets. However, with the framework developed, it is our future aim to model various sceneries with clinical significance. 8. Conclusions We have studied the effects of different chordae structures in a dynamic MV model that includes a detailed description of the valve leaflets and chordae. Our computational approach includes FSI, anisotropic hyperelastic constitutive modelling and energy budget analysis. The simulation has been carefully verified against the commercial software ABAQUS under static loading conditions. Our results show that detailed chordae structure is superior than our previously used simplified chordae structure, which, even when modelled as three-dimensional structures and embedded through the leaflets, leads to poor leaflets coaptation, large bulging on the anterior leaflet belly and different energy-transferring behaviour. On the other hand, the pseudo-fibre representation is a good approximation of the more realistic three-dimensional complex solid chordae construction. Indeed, similar MV closure shape, strain and stress patterns and energy distribution patterns are observed using either the complex or the pseudofibre models. This suggests that pseudo-fibres may be a reasonable choice to model mitral chordae in future. Acknowledgements We also wish to thank Dominic Sloan-Murphy and Richard Martin for their help with the installations of IBAMR on the ARCHER and ARCHIE-WeST supercomputers, respectively. Funding UK Engineering and Physical Sciences Research Council (EP/N014642/1 and EP/I029990/1); the British Heart Foundation (PG/14/64/31043); the National Science Foundation (ACI and ACI ); the National Institutes of Health (HL117063); China Scholarship Council Studentship to L.F.; Fee Waiver Programme of the University of Glasgow to L.F.; Fundamental Research Funds of the Shandong University, China to N.Q.; Leverhulme Trust Research Fellowship (RF to X.L.). References Akins, C. W, Travis, B. & Yoganathan, A. P. (2008) Energy loss for evaluating heart valve performance. J. Thorac. Cardiovasc. Surg., 136, Choi, Y. J., Vedula, V. & Mittal, R. (2014) Computational study of the dynamics of a bileaflet mechanical heart valve in the mitral position. Ann. Biomed. Eng., 42,
23 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 23 Cochran, R. P. & Kunzelman, K. S. (1998) Effect of papillary muscle position on mitral valve function: relationship to homografts. Ann. Thorac. Surg., 66, S155 S161. Colella, P. & Woodward, P. R. (1984) The piecewise parabolic method (PPM) for gas-dynamical simulations. J. Computat. Phys., 54, David, T. E., Burns, R. J., Bacchus, C. & Druck, M. (1984) Mitral valve replacement for mitral regurgitation with and without preservation of chordae tendineae. J. Thorac. Cardiovasc. Surg., 88, Einstein, D. R., Del Pin, F., Jiao, X., Kuprat, A. P., Carson, J. P., Kunzelman, K. S., Cochran, R. P., Guccione, J. M. & Ratcliffe, M. B. (2010) Fluid structure interactions of the mitral valve and left heart: comprehensive strategies, past, present and future. Int. J. Numer. Methods Biomed. Eng., 26, Espino, D. M., Shepherd, D., Hukins, D. & Buchan, K. G. (2005) The role of Chordae tendineae in mitral valve competence. J. Heart Valve Dis., 14, Gao, H., Feng, L., Qi, N., Berry, C., Griffith, B. & Luo, X. (2017a) A coupled mitral valve left ventricle model with fluid-structure interaction. Med. Eng. Phys., 47, Gao, H., Ma, X., Qi, N., Berry, C., Griffith, B. E. & Luo, X. (2014a) A finite strain nonlinear human mitral valve model with fluid-structure interaction. Int. J. Numer. Methods Biomed. Eng., 30, Gao, H., Qi, N., Feng, L., Ma, X., Danton, M., Berry, C. & Luo, X. (2017b) Modelling mitral valvular dynamics current trend and future directions. Int. J. Numer. Method Biomed. Eng, 33, e2858. Gao, H., Wang, H., Berry, C., Luo, X. & Griffith, B.E. (2014b) Quasi-static image-based immersed boundaryfinite element model of left ventricle under diastolic loading. Int. J. Numer. Method Biomed. Eng., 30, Gasser, T. C., Ogden, R. W. & Holzapfel, G. A. (2006) Hyperelastic modelling of arterial layers with distributed collagen fibre orientations. J. R. Soc. Interface, 3, Griffith, B. E. (2009) An accurate and efficient method for the incompressible Navier Stokes equations using the projection method as a preconditioner. J. Comput. Phys., 228, Griffith, B. E. & Lim, S. (2012) Simulating an elastic ring with bend and twist by an adaptive generalized immersed boundary method. Comm. Comput. Phys., 12, Griffith, B.E. & Luo, X. (2017) Hybrid finite difference/finite element version of the immersed boundary method. Int. J. Numer. Method Biomed. Eng., 33, e2888. Griffith, B. E., Luo, X., McQueen, D. M. & Peskin, C. S. (2009) Simulating the fluid dynamics of natural and prosthetic heart valves using the immersed boundary method. Int. J. Appl. Mech., 1, Holzapfel, G. A., Gasser, T. C. & Ogden, R. W. (2000) A new constitutive framework for arterial wall mechanics and a comparative study of material models. J. Elasticity, 61, Kheradvar, A., Groves, E. M., Dasi, L. P., Alavi, S. H., Tranquillo, R., Grande-Allen, K. J., Simmons, C. A., Griffith, B., Falahatpisheh, A., Goergen, C. J. et al. (2015) Emerging trends in heart valve engineering: part I. Solutions for future. Ann. Biomed. Eng., 43, Kunzelman, K., Cochran, R., Chuong, C., Ring, W., Verrier, E. D. & Eberhart, R. (1993) Finite element analysis of the mitral valve. J. Heart Valve Dis., 2, Kunzelman, K., Einstein, D. R. & Cochran, R. (2007) Fluid structure interaction models of the mitral valve: function in normal and pathological states. Philos. Trans. R. Soc. Lond. B Biol. Sci., 362, Kunzelman, K., Reimink, M. & Cochran, R. (1997) Annular dilatation increases stress in the mitral valve and delays coaptation: a finite element computer model. Vascular, 5, Kunzelman, K., Reimink, M. & Cochran, R. (1998) Flexible versus rigid ring annuloplasty for mitral valve annular dilatation: a finite element model. J. Heart Valve Dis., 7, Kunzelman, K. S. & Cochran, R. (1992) Stress/strain characteristics of porcine mitral valve tissue: parallel versus perpendicular collagen orientation. J. Card. Surg., 7, Lee, C.-H., Oomen, P. J., Rabbah, J. P., Yoganathan, A., Gorman, R. C., Gorman, J. H., Amini, R. & Sacks, M. S. (2013) A high-fidelity and micro-anatomically accurate 3D finite element model for simulations of functional mitral valve. International Conference on Functional Imaging and Modeling of the Heart. Berlin, Heidelberg: Springer, pp
24 24 L. FENG ET AL. Lee, C.-H., Rabbah, J.-P., Yoganathan, A. P., Gorman, R. C., Gorman, J. H. & Sacks, M. S. (2015) On the effects of leaflet microstructure and constitutive model on the closing behavior of the mitral valve. Biomech. Model. Mechanobiol., 14, Lim, S., Ferent, A., Wang, X. S. & Peskin, C. S. (2008) Dynamics of a closed rod with twist and bend in fluid. SIAM J. Sci. Comput., 31, Luo, X., Griffith, B. E., Ma, X., Yin, M., Wang, T., Liang, C., Watton, P. & Bernacca, G. (2012) Effect of bending rigidity in a dynamic model of a polyurethane prosthetic mitral valve. Biomech. Model. Mechanobiol., 11, Ma, X., Gao, H., Griffith, B. E., Berry, C. & Luo, X. (2013) Image-based fluid structure interaction model of the human mitral valve. Comput. Fluids, 71, May-Newman, K. & Yin, F. (1998) A constitutive law for mitral valve tissue. J. Biomech. Eng., 120, Prot, V. & Skallerud, B. (2009) Nonlinear solid finite element analysis of mitral valves with heterogeneous leaflet layers. Comput. Mech., 43, Reimink, M., Kunzelman, K. & Cochran, R. (1996) The effect of chordal replacement suture length on function and stresses in repaired mitral valves: a finite element study. J. Heart Valve Dis., 5, Kunzel, K., Einstein, D. R., & Coachran, R. (2007) Fluid-structure interaction models of the mitral valve: function in normal and pathological states. Philosophical Transactions of the Royal Society of London B: Biological Sciences, 362(1484), Skallerud, B., Prot, V. & Nordrum, I. (2011) Modeling active muscle contraction in mitral valve leaflets during systole: a first approach. Biomech. Model. Mechanobiol., 10, Stevanella, M., Maffessanti, F., Conti, C. A., Votta, E., Arnoldi, A., Lombardi, M., Parodi, O., Caiani, E. G. & Redaelli, A. (2011) Mitral valve patient-specific finite element modeling from cardiac MRI: application to an annuloplasty procedure. Cardiovasc. Eng. Technol., 2, Stevanella, M., Votta, E. & Redaelli, A. (2009) Mitral valve finite element modeling: implications of tissues nonlinear response and annular motion. J. Biomech. Eng., 131, Sun, W., Martin, C. & Pham, T. (2014) Computational modeling of cardiac valve function and intervention. Annu. Rev. Biomed. Eng., 16, Toma, M., Jensen, M. Ø., Einstein, D. R., Yoganathan, A. P., Cochran, R. P. & Kunzelman, K. S. (2016) Fluid structure interaction analysis of papillary muscle forces using a comprehensive mitral valve model with 3D chordal structure. Ann. Biomed. Eng., 44, Wang, Q. & Sun, W. (2013) Finite element modeling of mitral valve dynamic deformation using patient-specific multi-slices computed tomography scans. Ann. Biomed. Eng., 41, Watton, P., Luo, X., Yin, M., Bernacca, G. & Wheatley, D. (2008) Effect of ventricle motion on the dynamic behaviour of chorded mitral valves. J. Fluid Struct., 24, Appendix A. Verification test 1: deformation of a circular disc under constant pressure Model verification is first done on a simple axis symmetric geometry, as shown in Fig. A1(a and b). It is a disc-shaped structure with radius of 0.4 cm and thickness of 0.03 cm. In the IB/FE model, the disc is immersed in a viscous fluid with density of 1 g cm 3 and dynamic viscosity of 1 g. cm 1 s 1. A constant pressure of 20 mmhg is applied on the top surface of the structure in both cases, and in the IB/FE model, periodic conditions are applied on the boundaries of the computational domain Ω. Equation (18) is used for describing the disc material property. Two families of fibres are defined simply along the circumferential direction and radial direction, respectively. Figure A2(a and b) show the distribution of displacement magnitudes obtained from IB/FE and ABAQUS. The maximum displacement norm is mm in the IB/FE model, which is slightly less than the value in the ABAQUS model that is mm. It is clear that two plots have almost the same patterns.
25 ON THE CHORDAE STRUCTURE AND DYNAMIC MODELLING OF THE MV 25 Fig. A1. The disc geometry (a) side view, (b) top view. The relative difference in the displacements is computed via relative difference = norm of displacement difference 100%. average displacement norm from Abaqus (A.1) Figure A2(c) shows regions that have relative difference less than 2%, except for the inner edge; the relative difference is very small in most of the disc. Figure A3(a and b) shows the logarithmic maximum principal strain. The maximum values are in the IB/FE model and in the ABAQUS model. Using the same relative difference definition given by (A.1), Fig. A3(c) shows all the nodes with relative difference below 5%. Appendix B. Verification test 2: deformation of MV under constant pressure We use the MV with complex chordae structure to compare the IB/FE and the ABAQUS models under static loading conditions. In the IB/FE model, the MV is immersed in a 5 cm 4 cm 6 cm box filled with the same viscous fluid as in the disc case. A constant pressure of 30 mmhg is applied on the atrial Fig. A2. Normed displacement plot from (a) IB/FE and (b) ABAQUS. (c) The relative difference (only showing the regions below 2%).
26 26 L. FENG ET AL. Fig. A3. The logarithmic strain generated by (a) IB/FE and (b) ABAQUS. (c) The relative difference (only showing the regions below 5%). side of the leaflets, the annulus ring is fixed by using stiff tethering forces and displacement boundary conditions are applied to the chordae ends. Periodic boundary conditions are applied to boundaries of the computational domain Ω. The same structural constraints and loading conditions are also used in the ABAQUS model. Because of the large deformation in MV model, dynamic explicit analysis in ABAQUS is used. Equation (18) is used for the leaflets, but a Neo-Hookean material is used for the chordae structure (B.1) W = C(I1 3), with C = 740 kpa. Figure B1(a and b) shows the distribution of displacement magnitudes from the IB/FE and the ABAQUS models when the MV reaches an equilibrium state by maintaining constant loading for a long time. The maximum values are cm in the IB/FE model and cm in the ABAQUS model. Both models give good agreement in the MV closure shapes, with a mean difference of cm, except for slightly bigger gaps in the IB/FE model as shown in Fig. B1(a) due to the Eulerian mesh density and the use of delta function in the FSI model. Fig. B1. Displacement plot from (a) IB/FE and (b) ABAQUS. See online version for colours.
Dynamic finite-strain modelling of the human left ventricle in health and disease using an immersed boundary-finite element method
 IMA Journal of Applied Mathematics Advance Access published July 1, 2014 IMA Journal of Applied Mathematics (2014) Page 1 of 33 doi:10.1093/imamat/hxu029 Dynamic finite-strain modelling of the human left
IMA Journal of Applied Mathematics Advance Access published July 1, 2014 IMA Journal of Applied Mathematics (2014) Page 1 of 33 doi:10.1093/imamat/hxu029 Dynamic finite-strain modelling of the human left
PREDICTION OF BLOOD FLOW VELOCITY AND LEAFLET DEFORMATION VIA 2D MITRAL VALVE MODEL
 Journal of Mechanical Engineering and Sciences (JMES) e-issn: 2231-8380; Volume 2, pp. 217-225, June 2012 FKM, Universiti Malaysia Pahang PREDICTION OF BLOOD FLOW VELOCITY AND LEAFLET DEFORMATION VIA 2D
Journal of Mechanical Engineering and Sciences (JMES) e-issn: 2231-8380; Volume 2, pp. 217-225, June 2012 FKM, Universiti Malaysia Pahang PREDICTION OF BLOOD FLOW VELOCITY AND LEAFLET DEFORMATION VIA 2D
Effect of bending rigidity in a dynamic model of a polyurethane prosthetic mitral valve
 Biomech Model Mechanobiol (2012) 11:815 827 DOI 10.1007/s10237-011-0354-7 ORIGINAL PAPER Effect of bending rigidity in a dynamic model of a polyurethane prosthetic mitral valve X. Y. Luo B. E. Griffith
Biomech Model Mechanobiol (2012) 11:815 827 DOI 10.1007/s10237-011-0354-7 ORIGINAL PAPER Effect of bending rigidity in a dynamic model of a polyurethane prosthetic mitral valve X. Y. Luo B. E. Griffith
Finite Element Modeling of the Mitral Valve and Mitral Valve Repair
 Finite Element Modeling of the Mitral Valve and Mitral Valve Repair Iain Baxter A thesis submitted to the Faculty of Graduate and Postdoctoral Studies in partial fulfillment of the requirements for the
Finite Element Modeling of the Mitral Valve and Mitral Valve Repair Iain Baxter A thesis submitted to the Faculty of Graduate and Postdoctoral Studies in partial fulfillment of the requirements for the
Original. Stresses and Strains Distributions in Three-Dimension Three-Layer Abdominal Aortic Wall Based on in vivo Ultrasound Imaging
 Original Stresses and Strains Distributions in Three-Dimension Three-Layer Abdominal Aortic Wall Based on in vivo Ultrasound Imaging P. Khamdaengyodtai 1, T. Khamdaeng 1, P. Sakulchangsatjatai 1, N. Kammuang-lue
Original Stresses and Strains Distributions in Three-Dimension Three-Layer Abdominal Aortic Wall Based on in vivo Ultrasound Imaging P. Khamdaengyodtai 1, T. Khamdaeng 1, P. Sakulchangsatjatai 1, N. Kammuang-lue
A finite strain nonlinear human mitral valve model with fluid-structure interaction
 INTERNATIONAL JOURNAL FOR NUMERICAL METHODS IN BIOMEDICAL ENGINEERING Int. J. Numer. Meth. Biomed. Engng. 2014; 30:1597 1613 Published online in Wiley Online Library (wileyonlinelibrary.com)..2691 SELECTED
INTERNATIONAL JOURNAL FOR NUMERICAL METHODS IN BIOMEDICAL ENGINEERING Int. J. Numer. Meth. Biomed. Engng. 2014; 30:1597 1613 Published online in Wiley Online Library (wileyonlinelibrary.com)..2691 SELECTED
Anisotropic Mass-Spring Method Accurately Simulates Mitral Valve Closure from Image-Based Models
 Anisotropic Mass-Spring Method Accurately Simulates Mitral Valve Closure from Image-Based Models Peter E. Hammer 1,2,3, Pedro J. del Nido 1, and Robert D. Howe 2 1 Department of Cardiac Surgery, Children
Anisotropic Mass-Spring Method Accurately Simulates Mitral Valve Closure from Image-Based Models Peter E. Hammer 1,2,3, Pedro J. del Nido 1, and Robert D. Howe 2 1 Department of Cardiac Surgery, Children
Non-Newtonian pulsatile blood flow in a modeled artery with a stenosis and an aneurysm
 Non-Newtonian pulsatile blood flow in a modeled artery with a stenosis and an aneurysm I. Husain, C. Langdon and J. Schwark Department of Mathematics Luther College University of Regina Regina, Saskatchewan
Non-Newtonian pulsatile blood flow in a modeled artery with a stenosis and an aneurysm I. Husain, C. Langdon and J. Schwark Department of Mathematics Luther College University of Regina Regina, Saskatchewan
Design and Simulation of Blocked Blood Vessel for Early Detection of Heart Diseases
 Proceedings of the 215 2nd International Symposium on Physics and Technology of Sensors, 8-1th March, 215, Pune, India Design and Simulation of Blocked Blood Vessel for Early Detection of Heart Diseases
Proceedings of the 215 2nd International Symposium on Physics and Technology of Sensors, 8-1th March, 215, Pune, India Design and Simulation of Blocked Blood Vessel for Early Detection of Heart Diseases
WHAT CAN WE LEARN FROM COMPUTER SIMULATION?
 WHAT CAN WE LEARN FROM COMPUTER SIMULATION? Ehud Raanani, MD Gil Marom, Phd Cardiothoracic Surgery, Sheba Medical Center Sackler School of Medicine, Biomechanical Engineering, Tel Aviv University Homburg,
WHAT CAN WE LEARN FROM COMPUTER SIMULATION? Ehud Raanani, MD Gil Marom, Phd Cardiothoracic Surgery, Sheba Medical Center Sackler School of Medicine, Biomechanical Engineering, Tel Aviv University Homburg,
A novel mathematical technique to assess of the mitral valve dynamics based on echocardiography
 ORIGINAL ARTICLE A novel mathematical technique to assess of the mitral valve dynamics based on echocardiography Mersedeh Karvandi 1 MD, Saeed Ranjbar 2,* Ph.D, Seyed Ahmad Hassantash MD 2, Mahnoosh Foroughi
ORIGINAL ARTICLE A novel mathematical technique to assess of the mitral valve dynamics based on echocardiography Mersedeh Karvandi 1 MD, Saeed Ranjbar 2,* Ph.D, Seyed Ahmad Hassantash MD 2, Mahnoosh Foroughi
Patient-Specific Computational Simulation of the Mitral Valve Function Using Three-Dimensional Echocardiography
 Patient-Specific Computational Simulation of the Mitral Valve Function Using Three-Dimensional Echocardiography Y. Rim 1, S. T. Laing 1, P. Kee 1, K. B. Chandran 2, D. D. McPherson 1 and H. Kim 1 1 Department
Patient-Specific Computational Simulation of the Mitral Valve Function Using Three-Dimensional Echocardiography Y. Rim 1, S. T. Laing 1, P. Kee 1, K. B. Chandran 2, D. D. McPherson 1 and H. Kim 1 1 Department
Effect of leaflet-to-chordae contact interaction on computational mitral valve evaluation
 Rim et al. BioMedical Engineering OnLine 2014, 13:31 RESEARCH Open Access Effect of leaflet-to-chordae contact interaction on computational mitral valve evaluation Yonghoon Rim, David D McPherson and Hyunggun
Rim et al. BioMedical Engineering OnLine 2014, 13:31 RESEARCH Open Access Effect of leaflet-to-chordae contact interaction on computational mitral valve evaluation Yonghoon Rim, David D McPherson and Hyunggun
Numerical Simulation of Blood Flow through Asymmetric and Symmetric Occlusion in Carotid Artery
 Proceedings of the 3 rd International Conference on Fluid Flow, Heat and Mass Transfer (FFHMT 16) Ottawa, Canada May 2 3, 2016 Paper No. 170 Numerical Simulation of Blood Flow through Asymmetric and Symmetric
Proceedings of the 3 rd International Conference on Fluid Flow, Heat and Mass Transfer (FFHMT 16) Ottawa, Canada May 2 3, 2016 Paper No. 170 Numerical Simulation of Blood Flow through Asymmetric and Symmetric
Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture
 Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture RAMON BERGUER, a,b JOSEPH L. BULL, a,b AND KHALIL KHANAFER a a Vascular Mechanics Laboratory, Department of Biomedical Engineering,
Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture RAMON BERGUER, a,b JOSEPH L. BULL, a,b AND KHALIL KHANAFER a a Vascular Mechanics Laboratory, Department of Biomedical Engineering,
Available online at ScienceDirect. Procedia Engineering 126 (2015 )
 Available online at www.sciencedirect.com ScienceDirect Procedia Engineering 126 (2015 ) 441 445 7th International Conference on Fluid Mechanics, ICFM7 FSI modeling approach to develop right ventricle
Available online at www.sciencedirect.com ScienceDirect Procedia Engineering 126 (2015 ) 441 445 7th International Conference on Fluid Mechanics, ICFM7 FSI modeling approach to develop right ventricle
Mechanics of the mitral valve
 Biomech Model Mechanobiol (2013) 12:1053 1071 DOI 10.1007/s10237-012-0462-z ORIGINAL PAPER Mechanics of the mitral valve A critical review, an in vivo parameter identification, and the effect of prestrain
Biomech Model Mechanobiol (2013) 12:1053 1071 DOI 10.1007/s10237-012-0462-z ORIGINAL PAPER Mechanics of the mitral valve A critical review, an in vivo parameter identification, and the effect of prestrain
Accepted Manuscript. S (12)00076-X Reference: MEDIMA 694
 Accepted Manuscript An Integrated Framework for Finite-Element Modeling of Mitral Valve Biomechanics from Medical Images: Application to MitralClip Intervention Planning Tommaso Mansi, Ingmar Voigt, Bogdan
Accepted Manuscript An Integrated Framework for Finite-Element Modeling of Mitral Valve Biomechanics from Medical Images: Application to MitralClip Intervention Planning Tommaso Mansi, Ingmar Voigt, Bogdan
Valve Analysis and Pathoanatomy: THE MITRAL VALVE
 : THE MITRAL VALVE Marc R. Moon, M.D. John M. Shoenberg Chair in CV Disease Chief, Cardiac Surgery Washington University School of Medicine, St. Louis, MO Secretary, American Association for Thoracic Surgery
: THE MITRAL VALVE Marc R. Moon, M.D. John M. Shoenberg Chair in CV Disease Chief, Cardiac Surgery Washington University School of Medicine, St. Louis, MO Secretary, American Association for Thoracic Surgery
Simulating the Motion of Heart Valves Under Fluid Flows Induced by Cardiac Contraction
 Simulating the Motion of Heart Valves Under Fluid Flows Induced by Cardiac Contraction Eann A. Patterson Department of Mechanical Engineering, The University of Sheffield Mappin Street, Sheffield, S1 3JD
Simulating the Motion of Heart Valves Under Fluid Flows Induced by Cardiac Contraction Eann A. Patterson Department of Mechanical Engineering, The University of Sheffield Mappin Street, Sheffield, S1 3JD
Effect of ventricle motion on the dynamic behaviour of chorded mitral valves
 Journal of Fluids and Structures 24 (28) 58 74 www.elsevier.com/locate/jfs Effect of ventricle motion on the dynamic behaviour of chorded mitral valves P.N. Watton a,.. Luo b,, M. in c, G.M. Bernacca d,
Journal of Fluids and Structures 24 (28) 58 74 www.elsevier.com/locate/jfs Effect of ventricle motion on the dynamic behaviour of chorded mitral valves P.N. Watton a,.. Luo b,, M. in c, G.M. Bernacca d,
Contents 1 Computational Haemodynamics An Introduction 2 The Human Cardiovascular System
 Contents 1 Computational Haemodynamics An Introduction... 1 1.1 What is Computational Haemodynamics (CHD)... 1 1.2 Advantages of CHD... 3 1.3 Applications in the Cardiovascular System... 4 1.3.1 CHD as
Contents 1 Computational Haemodynamics An Introduction... 1 1.1 What is Computational Haemodynamics (CHD)... 1 1.2 Advantages of CHD... 3 1.3 Applications in the Cardiovascular System... 4 1.3.1 CHD as
Medical Engineering & Physics
 Medical Engineering & Physics 32 (2010) 1057 1064 Contents lists available at ScienceDirect Medical Engineering & Physics journal homepage: www.elsevier.com/locate/medengphy Mitral valve dynamics in structural
Medical Engineering & Physics 32 (2010) 1057 1064 Contents lists available at ScienceDirect Medical Engineering & Physics journal homepage: www.elsevier.com/locate/medengphy Mitral valve dynamics in structural
Fluid-structure interaction analysis of venous valve hemodynamics Narracott, A.J.; Keijsers, J.M.T.; Leguy, C.A.D.; Huberts, W.; van de Vosse, F.N.
 Fluid-structure interaction analysis of venous valve hemodynamics Narracott, A.J.; Keijsers, J.M.T.; Leguy, C.A.D.; Huberts, W.; van de Vosse, F.N. Published in: CMBE 2015 Proceedings Published: 01/01/2015
Fluid-structure interaction analysis of venous valve hemodynamics Narracott, A.J.; Keijsers, J.M.T.; Leguy, C.A.D.; Huberts, W.; van de Vosse, F.N. Published in: CMBE 2015 Proceedings Published: 01/01/2015
ISSUES ON COMPUTATIONAL MODELING FOR COMPUTATION-AIDED DIAGNOSIS 臨床診断支援ツールのための計算力学モデリング
 ISSUES ON COMPUTATIONAL MODELING FOR COMPUTATION-AIDED DIAGNOSIS 臨床診断支援ツールのための計算力学モデリング Hao LIU Advanced Computer and Information Division, RIKEN 2-1, Hirosawa, Wako-shi, Saitama 351-0198 JAPAN e-mail:
ISSUES ON COMPUTATIONAL MODELING FOR COMPUTATION-AIDED DIAGNOSIS 臨床診断支援ツールのための計算力学モデリング Hao LIU Advanced Computer and Information Division, RIKEN 2-1, Hirosawa, Wako-shi, Saitama 351-0198 JAPAN e-mail:
Effect of loading and geometry on functional parameters
 Effect of loading and geometry on functional parameters Piet Claus Cardiovascular Imaging and Dynamics Department of Cardiovascular Diseases Leuven University, Leuven, Belgium 5 th European Echocardiography
Effect of loading and geometry on functional parameters Piet Claus Cardiovascular Imaging and Dynamics Department of Cardiovascular Diseases Leuven University, Leuven, Belgium 5 th European Echocardiography
Effect of Congenital Anomalies of the Papillary Muscles on Mitral Valve Function
 J. Med. Biol. Eng. (2015) 35:104 112 DOI 10.1007/s40846-015-0011-1 ORIGINAL ARTICLE Effect of Congenital Anomalies of the Papillary Muscles on Mitral Valve Function Yonghoon Rim David D. McPherson Hyunggun
J. Med. Biol. Eng. (2015) 35:104 112 DOI 10.1007/s40846-015-0011-1 ORIGINAL ARTICLE Effect of Congenital Anomalies of the Papillary Muscles on Mitral Valve Function Yonghoon Rim David D. McPherson Hyunggun
Numerical Simulation of Blood Flow in the System of Human Coronary Arteries with and without Bypass Graft
 Numerical Simulation of Blood Flow in the System of Human Coronary Arteries with and without Bypass Graft BURASKORN NUNTADILOK 1, BENCHAWAN WIWATANAPATAPHEE 1 MEECHOKE CHUEDOUNG 1, THANONGCHAI SIRIAPISITH
Numerical Simulation of Blood Flow in the System of Human Coronary Arteries with and without Bypass Graft BURASKORN NUNTADILOK 1, BENCHAWAN WIWATANAPATAPHEE 1 MEECHOKE CHUEDOUNG 1, THANONGCHAI SIRIAPISITH
Two semilunar valves. Two atrioventricular valves. Valves of the heart. Left atrioventricular or bicuspid valve Mitral valve
 The Heart 3 Valves of the heart Two atrioventricular valves Two semilunar valves Right atrioventricular or tricuspid valve Left atrioventricular or bicuspid valve Mitral valve Aortic valve Pulmonary valve
The Heart 3 Valves of the heart Two atrioventricular valves Two semilunar valves Right atrioventricular or tricuspid valve Left atrioventricular or bicuspid valve Mitral valve Aortic valve Pulmonary valve
Finite Element Analysis of Patient-Specific Mitral Valve with Mitral Regurgitation
 Finite Element Analysis of Patient-Specific Mitral Valve with Mitral Regurgitation Pham Thuy, Georgia Institute of Technology Fanwei Kong, Georgia Institute of Technology Caitlin Martin, Georgia Institute
Finite Element Analysis of Patient-Specific Mitral Valve with Mitral Regurgitation Pham Thuy, Georgia Institute of Technology Fanwei Kong, Georgia Institute of Technology Caitlin Martin, Georgia Institute
Numerical simulations of fluid mechanical interactions between two abdominal aortic branches
 Korea-Australia Rheology Journal Vol. 16, No. 2, June 2004 pp. 75-83 Numerical simulations of fluid mechanical interactions between two abdominal aortic branches Taedong Kim, Taewon Seo* 1,2 and Abdul.I.
Korea-Australia Rheology Journal Vol. 16, No. 2, June 2004 pp. 75-83 Numerical simulations of fluid mechanical interactions between two abdominal aortic branches Taedong Kim, Taewon Seo* 1,2 and Abdul.I.
Ο ΡΟΛΟΣ ΤΩΝ ΚΟΛΠΩΝ ΣΤΗ ΛΕΙΤΟΥΡΓΙΚΗ ΑΝΕΠΑΡΚΕΙΑ ΤΩΝ ΚΟΛΠΟΚΟΙΛΙΑΚΩΝ ΒΑΛΒΙΔΩΝ
 Ο ΡΟΛΟΣ ΤΩΝ ΚΟΛΠΩΝ ΣΤΗ ΛΕΙΤΟΥΡΓΙΚΗ ΑΝΕΠΑΡΚΕΙΑ ΤΩΝ ΚΟΛΠΟΚΟΙΛΙΑΚΩΝ ΒΑΛΒΙΔΩΝ Ανδρέας Κατσαρός Καρδιολόγος Επιµ. Α Καρδιοχειρ/κών Τµηµάτων Γ.Ν.Α. Ιπποκράτειο ΚΑΜΙΑ ΣΥΓΚΡΟΥΣΗ ΣΥΜΦΕΡΟΝΤΩΝ ΑΝΑΦΟΡΙΚΑ ΜΕ ΤΗΝ ΠΑΡΟΥΣΙΑΣΗ
Ο ΡΟΛΟΣ ΤΩΝ ΚΟΛΠΩΝ ΣΤΗ ΛΕΙΤΟΥΡΓΙΚΗ ΑΝΕΠΑΡΚΕΙΑ ΤΩΝ ΚΟΛΠΟΚΟΙΛΙΑΚΩΝ ΒΑΛΒΙΔΩΝ Ανδρέας Κατσαρός Καρδιολόγος Επιµ. Α Καρδιοχειρ/κών Τµηµάτων Γ.Ν.Α. Ιπποκράτειο ΚΑΜΙΑ ΣΥΓΚΡΟΥΣΗ ΣΥΜΦΕΡΟΝΤΩΝ ΑΝΑΦΟΡΙΚΑ ΜΕ ΤΗΝ ΠΑΡΟΥΣΙΑΣΗ
Toward developing a finite element model of the mitral valve
 Toward developing a finite element model of the mitral valve I.A.E.W. van Loosdregt April 2011 July 2011 BMTE 11.22 Supervisors: R. Amini, Ph.D. (University of Pittsburgh) M.S. Sacks, Ph.D. (University
Toward developing a finite element model of the mitral valve I.A.E.W. van Loosdregt April 2011 July 2011 BMTE 11.22 Supervisors: R. Amini, Ph.D. (University of Pittsburgh) M.S. Sacks, Ph.D. (University
On the effects of leaflet microstructure and constitutive model on the closing behavior of the mitral valve
 Biomech Model Mechanobiol DOI 10.1007/s10237-015-0674-0 ORIGINAL PAPER On the effects of leaflet microstructure and constitutive model on the closing behavior of the mitral valve Chung-Hao Lee 1 Jean-Pierre
Biomech Model Mechanobiol DOI 10.1007/s10237-015-0674-0 ORIGINAL PAPER On the effects of leaflet microstructure and constitutive model on the closing behavior of the mitral valve Chung-Hao Lee 1 Jean-Pierre
8/31/2016. Mitraclip in Matthew Johnson, MD
 Mitraclip in 2016 Matthew Johnson, MD 1 Abnormal Valve Function Valve Stenosis Obstruction to valve flow during that phase of the cardiac cycle when the valve is normally open. Hemodynamic hallmark - pressure
Mitraclip in 2016 Matthew Johnson, MD 1 Abnormal Valve Function Valve Stenosis Obstruction to valve flow during that phase of the cardiac cycle when the valve is normally open. Hemodynamic hallmark - pressure
4D model of hemodynamics in the abdominal aorta
 Bio-Medical Materials and Engineering 26 (2015) S257 S264 DOI 10.3233/BME-151312 IOS Press S257 4D model of hemodynamics in the abdominal aorta Ireneusz Zbicinski a,*, Natalia Veshkina a and Ludomir Stefa
Bio-Medical Materials and Engineering 26 (2015) S257 S264 DOI 10.3233/BME-151312 IOS Press S257 4D model of hemodynamics in the abdominal aorta Ireneusz Zbicinski a,*, Natalia Veshkina a and Ludomir Stefa
Verification and Validation Methodologies for Prosthetic Heart Valves: Review and Considerations. ASME V&V40 Subcommittee Heart Valve Subgroup
 Verification and Validation Methodologies for Prosthetic Heart Valves: Review and Considerations. ASME V&V40 Subcommittee Heart Valve Subgroup ASME V&V 40 Heart Valve Subgroup Members Andrew Rau (Exponent
Verification and Validation Methodologies for Prosthetic Heart Valves: Review and Considerations. ASME V&V40 Subcommittee Heart Valve Subgroup ASME V&V 40 Heart Valve Subgroup Members Andrew Rau (Exponent
CFD Analysis of Pulsatile Flow and Non-Newtonian Behavior of Blood in Arteries
 Copyright 2015 Tech Science Press MCB, vol.12, no.1, pp.37-47, 2015 CFD Analysis of Pulsatile Flow and Non-Newtonian Behavior of Blood in Arteries P. Jhunjhunwala,, P.M. Padole, and S.B. Thombre, Abstract:
Copyright 2015 Tech Science Press MCB, vol.12, no.1, pp.37-47, 2015 CFD Analysis of Pulsatile Flow and Non-Newtonian Behavior of Blood in Arteries P. Jhunjhunwala,, P.M. Padole, and S.B. Thombre, Abstract:
Chen, Weiwei (2015) A coupled left ventricle and systemic arteries model. PhD thesis, University of Glasgow.
 Chen, Weiwei (2015) A coupled left ventricle and systemic arteries model. PhD thesis, University of Glasgow. http://theses.gla.ac.uk/7037/ Copyright and moral rights for this thesis are retained by the
Chen, Weiwei (2015) A coupled left ventricle and systemic arteries model. PhD thesis, University of Glasgow. http://theses.gla.ac.uk/7037/ Copyright and moral rights for this thesis are retained by the
The Edge-to-Edge Technique f For Barlow's Disease
 The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
A computational fluid dynamics simulation study of coronary blood flow affected by graft placement
 Interactive CardioVascular and Thoracic Surgery 19 (2014) 16 20 doi:10.1093/icvts/ivu034 Advance Access publication 22 April 2014 ORIGINAL ARTICLE ADULTCARDIAC A computational fluid dynamics simulation
Interactive CardioVascular and Thoracic Surgery 19 (2014) 16 20 doi:10.1093/icvts/ivu034 Advance Access publication 22 April 2014 ORIGINAL ARTICLE ADULTCARDIAC A computational fluid dynamics simulation
How does the heart pump? From sarcomere to ejection volume
 How does the heart pump? From sarcomere to ejection volume Piet Claus Cardiovascular Imaging and Dynamics Department of Cardiovascular Diseases University Leuven, Leuven, Belgium Course on deformation
How does the heart pump? From sarcomere to ejection volume Piet Claus Cardiovascular Imaging and Dynamics Department of Cardiovascular Diseases University Leuven, Leuven, Belgium Course on deformation
Outline. EuroScore II. Society of Thoracic Surgeons Score. EuroScore II
 SURGICAL RISK IN VALVULAR HEART DISEASE: WHAT 2D AND 3D ECHO CAN TELL YOU AND WHAT THEY CAN'T Ernesto E Salcedo, MD Professor of Medicine University of Colorado School of Medicine Director of Echocardiography
SURGICAL RISK IN VALVULAR HEART DISEASE: WHAT 2D AND 3D ECHO CAN TELL YOU AND WHAT THEY CAN'T Ernesto E Salcedo, MD Professor of Medicine University of Colorado School of Medicine Director of Echocardiography
Finite Element Analysis for Edge-to-Edge Technique to Treat Post-Mitral Valve Repair Systolic Anterior Motion
 Acta of Bioengineering and Biomechanics Vol. 6, No. 4, 04 Original paper DOI: 0.577/ABB-0008-04-0 Finite Element Analysis for Edge-to-Edge Technique to Treat Post-Mitral Valve Repair Systolic Anterior
Acta of Bioengineering and Biomechanics Vol. 6, No. 4, 04 Original paper DOI: 0.577/ABB-0008-04-0 Finite Element Analysis for Edge-to-Edge Technique to Treat Post-Mitral Valve Repair Systolic Anterior
Posterior leaflet prolapse is the most common lesion seen
 Techniques for Repairing Posterior Leaflet Prolapse of the Mitral Valve Robin Varghese, MD, MS, and David H. Adams, MD Posterior leaflet prolapse is the most common lesion seen in degenerative mitral valve
Techniques for Repairing Posterior Leaflet Prolapse of the Mitral Valve Robin Varghese, MD, MS, and David H. Adams, MD Posterior leaflet prolapse is the most common lesion seen in degenerative mitral valve
Finite Element Implementation of a Structurally-Motivated Constitutive Relation for the Human Abdominal Aortic Wall with and without Aneurysms
 Downloaded from orbit.dtu.dk on: Dec 18, 017 Finite Element Implementation of a Structurally-Motivated Constitutive Relation for the Human Abdominal Aortic Wall with and without Aneurysms Traberg, Marie
Downloaded from orbit.dtu.dk on: Dec 18, 017 Finite Element Implementation of a Structurally-Motivated Constitutive Relation for the Human Abdominal Aortic Wall with and without Aneurysms Traberg, Marie
Using Computational Fluid Dynamics Model to Predict Changes in Velocity properties in Stented Carotid Artery
 Excerpt from the Proceedings of the COMSOL Conference 2010 Paris (COMSOL Conference) Using Computational Fluid Dynamics Model to Predict Changes in Velocity properties in Stented Carotid Artery Vaidehi
Excerpt from the Proceedings of the COMSOL Conference 2010 Paris (COMSOL Conference) Using Computational Fluid Dynamics Model to Predict Changes in Velocity properties in Stented Carotid Artery Vaidehi
Copyright 2017 Tech Science Press MCB, vol.14, no.3, pp , 2017
 Copyright 2017 Tech Science Press MCB, vol.14, no.3, pp.137-151, 2017 Comparison of Right Ventricle Morphological and Mechanical Characteristics for Healthy and Patients with Tetralogy of Fallot: An In
Copyright 2017 Tech Science Press MCB, vol.14, no.3, pp.137-151, 2017 Comparison of Right Ventricle Morphological and Mechanical Characteristics for Healthy and Patients with Tetralogy of Fallot: An In
Supervisors: Dr. ir. Matthieu De Beule, Prof. dr. ir. Benedict Verhegghe Counsellors: Tim Dezutter, dhr. Philippe Bertrand
 Simulation tools for mitral valve repair Viktoriya Antonova Supervisors: Dr. ir. Matthieu De Beule, Prof. dr. ir. Benedict Verhegghe Counsellors: Tim Dezutter, dhr. Philippe Bertrand Master's dissertation
Simulation tools for mitral valve repair Viktoriya Antonova Supervisors: Dr. ir. Matthieu De Beule, Prof. dr. ir. Benedict Verhegghe Counsellors: Tim Dezutter, dhr. Philippe Bertrand Master's dissertation
Blood flow in vessels with artificial or pathological geometrical changes
 Blood flow in vessels with artificial or pathological geometrical changes P. Tibaut 1, B. Wiesler 1, M. Mayer 2 & R. Wegenkittel 3 1 AVL LIST GmbH, Graz, Austria 2 VRVIs, Vienna, Austria 3 Tiani Medgraph
Blood flow in vessels with artificial or pathological geometrical changes P. Tibaut 1, B. Wiesler 1, M. Mayer 2 & R. Wegenkittel 3 1 AVL LIST GmbH, Graz, Austria 2 VRVIs, Vienna, Austria 3 Tiani Medgraph
A Two-layered Model for the Analysis of Arterial Rheology
 IJCSIT International Journal of Computer Science and Information Technology, Vol., o., June, pp. 37- A Two-layered Model for the Analysis of Arterial Rheology Sapna Singh Department of Mathematics, Harcourt
IJCSIT International Journal of Computer Science and Information Technology, Vol., o., June, pp. 37- A Two-layered Model for the Analysis of Arterial Rheology Sapna Singh Department of Mathematics, Harcourt
Three dimensional fluid structural interaction of tissue valves
 University of Iowa Iowa Research Online Theses and Dissertations Spring 2013 Three dimensional fluid structural interaction of tissue valves Vijay Govindarajan University of Iowa Copyright 2013 Vijay Govindarajan
University of Iowa Iowa Research Online Theses and Dissertations Spring 2013 Three dimensional fluid structural interaction of tissue valves Vijay Govindarajan University of Iowa Copyright 2013 Vijay Govindarajan
FSI Analysis of Diseased Coronary using Patient Specific Data
 FSI Analysis of Diseased Coronary using Patient Specific Data Mingchao Cai, * Haofei Liu, a Chun Yang, a,c Jie Zheng, b Richard Bach, b Mehmet H. Kural, d Kristen L. Billiar, d David Muccigrosso, b Dongsi
FSI Analysis of Diseased Coronary using Patient Specific Data Mingchao Cai, * Haofei Liu, a Chun Yang, a,c Jie Zheng, b Richard Bach, b Mehmet H. Kural, d Kristen L. Billiar, d David Muccigrosso, b Dongsi
Image Analysis and Cytometry in Three-Dimensional Digital Reconstruction of Porcine Native Aortic Valve Leaflets
 Image Analysis and Cytometry in Three-Dimensional Digital Reconstruction of Porcine Native Aortic Valve Leaflets Introduction Chi Zheng, M1 - University of Pittsburgh; BSE - University of Michigan In association
Image Analysis and Cytometry in Three-Dimensional Digital Reconstruction of Porcine Native Aortic Valve Leaflets Introduction Chi Zheng, M1 - University of Pittsburgh; BSE - University of Michigan In association
Surgical Repair of the Mitral Valve Presenter: Graham McCrystal Cardiothoracic Surgeon Christchurch Public Hospital
 Mitral Valve Surgical intervention Graham McCrystal Chairs: Rajesh Nair & Gerard Wilkins Surgical Repair of the Mitral Valve Presenter: Graham McCrystal Cardiothoracic Surgeon Christchurch Public Hospital
Mitral Valve Surgical intervention Graham McCrystal Chairs: Rajesh Nair & Gerard Wilkins Surgical Repair of the Mitral Valve Presenter: Graham McCrystal Cardiothoracic Surgeon Christchurch Public Hospital
Keywords: Angioplasty, Explicit finite elements method, Tube hidroforming, Stents.
 Blucher Mechanical Engineering Proceedings May 2014, vol. 1, num. 1 www.proceedings.blucher.com.br/evento/10wccm AN ANALYSIS OF THE CONTACT BETWEEN THE STENT AND THE ARTERY USING TUBE HIDROFORMING SIMULATION
Blucher Mechanical Engineering Proceedings May 2014, vol. 1, num. 1 www.proceedings.blucher.com.br/evento/10wccm AN ANALYSIS OF THE CONTACT BETWEEN THE STENT AND THE ARTERY USING TUBE HIDROFORMING SIMULATION
MITRAL VALVE FORCE BALANCE: A QUANTITATIVE ASSESSMENT OF ANNULAR AND SUBVALVULAR FORCES
 MITRAL VALVE FORCE BALANCE: A QUANTITATIVE ASSESSMENT OF ANNULAR AND SUBVALVULAR FORCES A Dissertation Presented to The Academic Faculty by Andrew W. Siefert In Partial Fulfillment of the Requirements
MITRAL VALVE FORCE BALANCE: A QUANTITATIVE ASSESSMENT OF ANNULAR AND SUBVALVULAR FORCES A Dissertation Presented to The Academic Faculty by Andrew W. Siefert In Partial Fulfillment of the Requirements
By: Stephanie Bendtsen, Joseph Calderan, and Celeste Dupont Team 17 Client: Dr. Sun
 By: Stephanie Bendtsen, Joseph Calderan, and Celeste Dupont Team 17 Client: Dr. Sun Flow Loop Design Introduction Purpose: to build a pulsatile flow loop that allows for implementation and testing of mechanical
By: Stephanie Bendtsen, Joseph Calderan, and Celeste Dupont Team 17 Client: Dr. Sun Flow Loop Design Introduction Purpose: to build a pulsatile flow loop that allows for implementation and testing of mechanical
Degenerative Mitral Regurgitation: Etiology and Natural History of Disease and Triggers for Intervention
 Degenerative Mitral Regurgitation: Etiology and Natural History of Disease and Triggers for Intervention John N. Hamaty D.O. FACC, FACOI November 17 th 2017 I have no financial disclosures Primary Mitral
Degenerative Mitral Regurgitation: Etiology and Natural History of Disease and Triggers for Intervention John N. Hamaty D.O. FACC, FACOI November 17 th 2017 I have no financial disclosures Primary Mitral
3D Printing & Echocardiography
 ASE SOTA Feb 19, 2018 3D Printing & Echocardiography Stephen H. Little, MD John S. Dunn Chair in Cardiovascular Research and Education, Associate professor, Weill Cornell Medicine Disclosures Personal
ASE SOTA Feb 19, 2018 3D Printing & Echocardiography Stephen H. Little, MD John S. Dunn Chair in Cardiovascular Research and Education, Associate professor, Weill Cornell Medicine Disclosures Personal
Blood Flow Simulation toward Actual Application at Hospital
 THE 5 TH ASIAN COMPUTAITIONAL FLUID DYNAMICS BUSAN, KOREA, OCTOBER 27 ~ OCTOBER 30, 2003 Blood Flow Simulation toward Actual Application at Hospital Abstract R. Himeno 1 1. Advanced Center for Computing
THE 5 TH ASIAN COMPUTAITIONAL FLUID DYNAMICS BUSAN, KOREA, OCTOBER 27 ~ OCTOBER 30, 2003 Blood Flow Simulation toward Actual Application at Hospital Abstract R. Himeno 1 1. Advanced Center for Computing
Numerical Analysis of the Influence of Stent Parameters on the Fatigue Properties
 Numerical Analysis of the Influence of Stent Parameters on the Fatigue Properties Lin Chen, Shen Jingfeng & Chen Bing School of mechanical engineering, University of Shanghai for Science & Technology,
Numerical Analysis of the Influence of Stent Parameters on the Fatigue Properties Lin Chen, Shen Jingfeng & Chen Bing School of mechanical engineering, University of Shanghai for Science & Technology,
DYNAMIC BLOOD FLOW IN A LEFT VENTRICLE
 DYNAMIC BLOOD FLOW IN A LEFT VENTRICLE Hidehito IWASE 1, Hao LIU 2, Shinichi FUJIMOTO 3, Ryutaro HIMENO 4 1 Computer Information Center and Advanced Engineering Center, RIKEN 2-1, Hirosawa, Wako-shi, Saitam
DYNAMIC BLOOD FLOW IN A LEFT VENTRICLE Hidehito IWASE 1, Hao LIU 2, Shinichi FUJIMOTO 3, Ryutaro HIMENO 4 1 Computer Information Center and Advanced Engineering Center, RIKEN 2-1, Hirosawa, Wako-shi, Saitam
Figure 1 The Human Heart
 ARTIFICIAL HEART VALVE MODEL INTRODUCTION The heart is a muscular pump with four chambers and four heart valves. The upper chambers, the right atrium and left, are thin walled filling chambers. Blood flows
ARTIFICIAL HEART VALVE MODEL INTRODUCTION The heart is a muscular pump with four chambers and four heart valves. The upper chambers, the right atrium and left, are thin walled filling chambers. Blood flows
Biomechanical implications of congenitally bicuspid aortic valves: a finite element analysis of patient-specific geometry
 University of Iowa Iowa Research Online Theses and Dissertations Spring 2013 Biomechanical implications of congenitally bicuspid aortic valves: a finite element analysis of patient-specific geometry Paul
University of Iowa Iowa Research Online Theses and Dissertations Spring 2013 Biomechanical implications of congenitally bicuspid aortic valves: a finite element analysis of patient-specific geometry Paul
ICE: Echo Core Lab-CRF
 APPENDIX 1 ICE: Echo Core Lab-CRF Study #: - Pt Initials: 1. Date of study: / / D D M M M Y Y Y Y 2. Type of Study: TTE TEE 3. Quality of Study: Poor Moderate Excellent Ejection Fraction 4. Ejection Fraction
APPENDIX 1 ICE: Echo Core Lab-CRF Study #: - Pt Initials: 1. Date of study: / / D D M M M Y Y Y Y 2. Type of Study: TTE TEE 3. Quality of Study: Poor Moderate Excellent Ejection Fraction 4. Ejection Fraction
PROSTHETIC VALVE BOARD REVIEW
 PROSTHETIC VALVE BOARD REVIEW The correct answer D This two chamber view shows a porcine mitral prosthesis with the typical appearance of the struts although the leaflets are not well seen. The valve
PROSTHETIC VALVE BOARD REVIEW The correct answer D This two chamber view shows a porcine mitral prosthesis with the typical appearance of the struts although the leaflets are not well seen. The valve
Simulations of the blood flow in the arterio-venous fistula for haemodialysis
 Acta of Bioengineering and Biomechanics Vol. 16, No. 1, 2014 Original paper DOI: 10.5277/abb140109 Simulations of the blood flow in the arterio-venous fistula for haemodialysis DANIEL JODKO*, DAMIAN OBIDOWSKI,
Acta of Bioengineering and Biomechanics Vol. 16, No. 1, 2014 Original paper DOI: 10.5277/abb140109 Simulations of the blood flow in the arterio-venous fistula for haemodialysis DANIEL JODKO*, DAMIAN OBIDOWSKI,
Mechanical Properties and Active Remodeling of Blood Vessels. Systemic Arterial Tree. Elastic Artery Structure
 Mechanical Properties and Active Remodeling of Blood Vessels Gross anatomy of systemic and pulmonary circulation Microscopic structure Mechanical properties and testing Residual stress Remodeling Systemic
Mechanical Properties and Active Remodeling of Blood Vessels Gross anatomy of systemic and pulmonary circulation Microscopic structure Mechanical properties and testing Residual stress Remodeling Systemic
STUDY OF FLOW PHENOMENA IN AORTIC DISSECTION
 STUDY OF FLOW PHENOMENA IN AORTIC DISSECTION Maurizio Bordone *, Eugenio Oñate *, Paula Rudenick, Bart Bijnens and Eduardo Soudah * * International Centre for Numerical Methods in Engineering (CIMNE) Campus
STUDY OF FLOW PHENOMENA IN AORTIC DISSECTION Maurizio Bordone *, Eugenio Oñate *, Paula Rudenick, Bart Bijnens and Eduardo Soudah * * International Centre for Numerical Methods in Engineering (CIMNE) Campus
3D Printing & Echocardiography
 Echo Hawaii Jan 18, 2018 3D Printing & Echocardiography Stephen H. Little, MD John S. Dunn Chair in Cardiovascular Research and Education, Associate professor, Weill Cornell Medicine Rapid Prototyping
Echo Hawaii Jan 18, 2018 3D Printing & Echocardiography Stephen H. Little, MD John S. Dunn Chair in Cardiovascular Research and Education, Associate professor, Weill Cornell Medicine Rapid Prototyping
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease
 Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Echocardiographic Evaluation of Primary Mitral Regurgitation
 Echocardiographic Evaluation of Primary Mitral Regurgitation Roberto M Lang, MD 0-10 o ME 4CH Med A2 P2 50-70 o Commissural P3 P1 A2 80-100 o ME 2CH P3 A2 A1 A1 125-135 o - ME Long axis P2 A2 P3 A3 P2
Echocardiographic Evaluation of Primary Mitral Regurgitation Roberto M Lang, MD 0-10 o ME 4CH Med A2 P2 50-70 o Commissural P3 P1 A2 80-100 o ME 2CH P3 A2 A1 A1 125-135 o - ME Long axis P2 A2 P3 A3 P2
Blood flow induced wall stress in the left ventricle of the heart
 Blood flow induced wall stress in the left ventricle of the heart A. K. Macpherson 1, S. Neti 1, J. A. Mannisi 2 & P. A. Macpherson 3 1 Institute for Biomedical Engineering and Mathematical Biology, Lehigh
Blood flow induced wall stress in the left ventricle of the heart A. K. Macpherson 1, S. Neti 1, J. A. Mannisi 2 & P. A. Macpherson 3 1 Institute for Biomedical Engineering and Mathematical Biology, Lehigh
Mechanotransduction in Ischemic Cardiac Tissue: A Mechanical Bidomain Approach under Plane Stress
 Mechanotransduction in Ischemic Cardiac Tissue: A Mechanical Bidomain Approach under Plane Stress Austin Fee* & Bradley J. Roth *Department of Physics, Oakland University, Rochester, Michigan https://doi.org/10.33697/ajur.2019.001
Mechanotransduction in Ischemic Cardiac Tissue: A Mechanical Bidomain Approach under Plane Stress Austin Fee* & Bradley J. Roth *Department of Physics, Oakland University, Rochester, Michigan https://doi.org/10.33697/ajur.2019.001
Towards Patient-Specific Finite-Element Simulation of MitralClip Procedure
 Towards Patient-Specific Finite-Element Simulation of MitralClip Procedure T. Mansi 1, I. Voigt 1,2, E. Assoumou Mengue 1, R. Ionasec 1, B. Georgescu 1, T. Noack 3, J. Seeburger 3, and D. Comaniciu 1 1
Towards Patient-Specific Finite-Element Simulation of MitralClip Procedure T. Mansi 1, I. Voigt 1,2, E. Assoumou Mengue 1, R. Ionasec 1, B. Georgescu 1, T. Noack 3, J. Seeburger 3, and D. Comaniciu 1 1
Part II: Fundamentals of 3D Echocardiography: Acquisition and Application
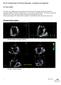 Part II: Fundamentals of 3D Echocardiography: Acquisition and Application Dr. Bruce Bollen 3D matrix array TEE probes provide options for both 2D and 3D imaging. Indeed, their utility in obtaining multiple
Part II: Fundamentals of 3D Echocardiography: Acquisition and Application Dr. Bruce Bollen 3D matrix array TEE probes provide options for both 2D and 3D imaging. Indeed, their utility in obtaining multiple
Medical Image Analysis
 Medical Image Analysis 16 (2012) 1330 1346 Contents lists available at SciVerse ScienceDirect Medical Image Analysis journal homepage: www.elsevier.com/locate/media An integrated framework for finite-element
Medical Image Analysis 16 (2012) 1330 1346 Contents lists available at SciVerse ScienceDirect Medical Image Analysis journal homepage: www.elsevier.com/locate/media An integrated framework for finite-element
Finite Element Analysis of the Human Middle Ear and an Application for Clinics for Tympanoplasty (Static and Harmonic Vibration Analysis)
 2015 American Transactions on Engineering & Applied Sciences. American Transactions on Engineering & Applied Sciences http://tuengr.com/ateas Finite Element Analysis of the Human Middle Ear and an Application
2015 American Transactions on Engineering & Applied Sciences. American Transactions on Engineering & Applied Sciences http://tuengr.com/ateas Finite Element Analysis of the Human Middle Ear and an Application
CHAPTER 34 LEFT VENTRICULAR FLOW 34-1
 CHAPTER 34 LEFT VENTRICULAR FLOW 34-1 CHAPTER 34 LEFT VENTRICULAR FLOW In Chapter 05 we studied the relationship of anterior leaflet shape to left ventricular pressure and flow from mid-diastole through
CHAPTER 34 LEFT VENTRICULAR FLOW 34-1 CHAPTER 34 LEFT VENTRICULAR FLOW In Chapter 05 we studied the relationship of anterior leaflet shape to left ventricular pressure and flow from mid-diastole through
COMPUTER SIMULATION OF BLOOD FLOW IN ARTERIES AFFECTED BY MULTIPLE ANEURYSM
 COMPUTER SIMULATION OF BLOOD FLOW IN ARTERIES AFFECTED BY MULTIPLE ANEURYSM H. GIRIJA BAI 1 and K.B. NAIDU 2 Department of Mathematics, Sathyabama University, Chennai-600 119, Tamil Nadu, India 1 girijanameprakash@gmail.com
COMPUTER SIMULATION OF BLOOD FLOW IN ARTERIES AFFECTED BY MULTIPLE ANEURYSM H. GIRIJA BAI 1 and K.B. NAIDU 2 Department of Mathematics, Sathyabama University, Chennai-600 119, Tamil Nadu, India 1 girijanameprakash@gmail.com
Mechanical Properties and Active Remodeling of Blood Vessels. Blood Vessels
 Mechanical Properties and Active Remodeling of Blood Vessels Gross anatomy of systemic and pulmonary circulation Microscopic structure Mechanical properties and testing Residual stress Remodeling Blood
Mechanical Properties and Active Remodeling of Blood Vessels Gross anatomy of systemic and pulmonary circulation Microscopic structure Mechanical properties and testing Residual stress Remodeling Blood
PHYSIOEX 3.0 EXERCISE 33B: CARDIOVASCULAR DYNAMICS
 PHYSIOEX 3.0 EXERCISE 33B: CARDIOVASCULAR DYNAMICS Objectives 1. To define the following: blood flow; viscosity; peripheral resistance; systole; diastole; end diastolic volume; end systolic volume; stroke
PHYSIOEX 3.0 EXERCISE 33B: CARDIOVASCULAR DYNAMICS Objectives 1. To define the following: blood flow; viscosity; peripheral resistance; systole; diastole; end diastolic volume; end systolic volume; stroke
Computational Simulation of Penetrating Trauma in Biological Soft Tissues using the Material Point Method
 Computational Simulation of Penetrating Trauma in Biological Soft Tissues using the Material Point Method Irina IONESCU* +, James GUILKEY* ++, Martin BERZINS*, Robert M. KIRBY*, Jeffrey WEISS* + *Scientific
Computational Simulation of Penetrating Trauma in Biological Soft Tissues using the Material Point Method Irina IONESCU* +, James GUILKEY* ++, Martin BERZINS*, Robert M. KIRBY*, Jeffrey WEISS* + *Scientific
Stretching Cardiac Myocytes: A Finite Element Model of Cardiac Tissue
 Megan McCain ES240 FEM Final Project December 19, 2006 Stretching Cardiac Myocytes: A Finite Element Model of Cardiac Tissue Cardiac myocytes are the cells that constitute the working muscle of the heart.
Megan McCain ES240 FEM Final Project December 19, 2006 Stretching Cardiac Myocytes: A Finite Element Model of Cardiac Tissue Cardiac myocytes are the cells that constitute the working muscle of the heart.
Basic principles of Rheumatic mitral valve Repair
 Basic principles of Rheumatic mitral valve Repair Prof. Gebrine El Khoury, MD DEPARTMENT OF CARDIOVASCULAR AND THORACIC SURGERY ST. LUC HOSPITAL - BRUSSELS, BELGIUM 1 Rheumatic MV disease MV repair confers
Basic principles of Rheumatic mitral valve Repair Prof. Gebrine El Khoury, MD DEPARTMENT OF CARDIOVASCULAR AND THORACIC SURGERY ST. LUC HOSPITAL - BRUSSELS, BELGIUM 1 Rheumatic MV disease MV repair confers
Copyright 2015 Tech Science Press MCB, vol.12, no.2, pp.67-85, 2015
 Copyright 2015 Tech Science Press MCB, vol.12, no.2, pp.67-85, 2015 3D Fluid-Structure Interaction Canine Heart Model with Patch to Quantify Mechanical Conditions for Optimal Myocardium Stem Cell Growth
Copyright 2015 Tech Science Press MCB, vol.12, no.2, pp.67-85, 2015 3D Fluid-Structure Interaction Canine Heart Model with Patch to Quantify Mechanical Conditions for Optimal Myocardium Stem Cell Growth
MITRAL REGURGITATION ECHO PARAMETERS TOOL
 Comprehensive assessment of qualitative and quantitative parameters, along with the use of standardized nomenclature when reporting echocardiographic findings, helps to better define a patient s MR and
Comprehensive assessment of qualitative and quantitative parameters, along with the use of standardized nomenclature when reporting echocardiographic findings, helps to better define a patient s MR and
Ischemic mitral regurgitation (MR) affects 1.2 to 2.1
 First Finite Element Model of the Left Ventricle With Mitral Valve: Insights Into Ischemic Mitral Regurgitation Jonathan F. Wenk, PhD, Zhihong Zhang, MS, Guangming Cheng, MD, PhD, Deepak Malhotra, MD,
First Finite Element Model of the Left Ventricle With Mitral Valve: Insights Into Ischemic Mitral Regurgitation Jonathan F. Wenk, PhD, Zhihong Zhang, MS, Guangming Cheng, MD, PhD, Deepak Malhotra, MD,
CVS Hemodynamics. Change in blood pressure:
 CVS Hemodynamics -The distribution of blood inside the circulation: The major part of blood volume is found in the venous system 60% (2/3), that s why veins are called the capacitance vessels. -Arteries
CVS Hemodynamics -The distribution of blood inside the circulation: The major part of blood volume is found in the venous system 60% (2/3), that s why veins are called the capacitance vessels. -Arteries
Certificate in Clinician Performed Ultrasound (CCPU) Syllabus. Rapid Cardiac Echo (RCE)
 Certificate in Clinician Performed Ultrasound (CCPU) Syllabus Rapid Cardiac Echo (RCE) Purpose: Rapid Cardiac Echocardiography (RCE) This unit is designed to cover the theoretical and practical curriculum
Certificate in Clinician Performed Ultrasound (CCPU) Syllabus Rapid Cardiac Echo (RCE) Purpose: Rapid Cardiac Echocardiography (RCE) This unit is designed to cover the theoretical and practical curriculum
Effects of Non-Newtonian Behavior of Blood on Wall Shear Stress in an Elastic Vessel with Simple and Consecutive Stenosis
 Biomedical & Pharmacology Journal Vol. 8(1), 123-131 (2015) Effects of Non-Newtonian Behavior of Blood on Wall Shear Stress in an Elastic Vessel with Simple and Consecutive Stenosis M. JAHANGIRI 1 *, M.
Biomedical & Pharmacology Journal Vol. 8(1), 123-131 (2015) Effects of Non-Newtonian Behavior of Blood on Wall Shear Stress in an Elastic Vessel with Simple and Consecutive Stenosis M. JAHANGIRI 1 *, M.
Cardiac ultrasound protocols
 Cardiac ultrasound protocols IDEXX Telemedicine Consultants Two-dimensional and M-mode imaging planes Right parasternal long axis four chamber Obtained from the right side Displays the relative proportions
Cardiac ultrasound protocols IDEXX Telemedicine Consultants Two-dimensional and M-mode imaging planes Right parasternal long axis four chamber Obtained from the right side Displays the relative proportions
Towards Patient-Specific Finite-Element Simulation of MitralClip Procedure
 Towards Patient-Specific Finite-Element Simulation of MitralClip Procedure T. Mansi 1,I.Voigt 1,2, E. Assoumou Mengue 1,R.Ionasec 1, B. Georgescu 1, T. Noack 3, J. Seeburger 3, and D. Comaniciu 1 1 Siemens
Towards Patient-Specific Finite-Element Simulation of MitralClip Procedure T. Mansi 1,I.Voigt 1,2, E. Assoumou Mengue 1,R.Ionasec 1, B. Georgescu 1, T. Noack 3, J. Seeburger 3, and D. Comaniciu 1 1 Siemens
Human Heart Blood Flow Simulations Based on CFD
 Human Heart Blood Flow Simulations Based on CFD Dean Vucinic 1, Tao Yang 1,2, Andrey A. Aksenov 3 1 Vrije Universiteit Brussel, Belgium 12 Xi an Modern Control Technology Institute, China 3 Capvidia/TESIS,
Human Heart Blood Flow Simulations Based on CFD Dean Vucinic 1, Tao Yang 1,2, Andrey A. Aksenov 3 1 Vrije Universiteit Brussel, Belgium 12 Xi an Modern Control Technology Institute, China 3 Capvidia/TESIS,
Morphological Investigations of the Anterior Leaflet and its Chordae Tendineae in Canine Mitral Valves
 Morphological Investigations of the Anterior Leaflet and its Chordae Tendineae in Canine Mitral Valves Takuma Aoki Yoko Fujii Hiroshi Sunahara Keisuke Sugimoto Yoshito Wakao Laboratory of Veterinary Surgery
Morphological Investigations of the Anterior Leaflet and its Chordae Tendineae in Canine Mitral Valves Takuma Aoki Yoko Fujii Hiroshi Sunahara Keisuke Sugimoto Yoshito Wakao Laboratory of Veterinary Surgery
CFD Challenge: Simulation of Hemodynamics in a Patient-Specific Aortic Coarctation Model
 CFD Challenge: Simulation of Hemodynamics in a Patient-Specific Aortic Coarctation Model Background Coarctation of the aorta (CoA) accounts for 8%-11% of congenital heart defects, affecting tens of thousands
CFD Challenge: Simulation of Hemodynamics in a Patient-Specific Aortic Coarctation Model Background Coarctation of the aorta (CoA) accounts for 8%-11% of congenital heart defects, affecting tens of thousands
The effect of angulation in abdominal aortic aneurysms: fluid structure interaction simulations of idealized geometries
 DOI 10.1007/s11517-010-0714-y ORIGINAL ARTICLE The effect of angulation in abdominal aortic aneurysms: fluid structure interaction simulations of idealized geometries Michalis Xenos Yared Alemu Dan Zamfir
DOI 10.1007/s11517-010-0714-y ORIGINAL ARTICLE The effect of angulation in abdominal aortic aneurysms: fluid structure interaction simulations of idealized geometries Michalis Xenos Yared Alemu Dan Zamfir
FOR many decades, cardiovascular disease has been one of
 Vol:1, No:2, 27 Effect of Non-Newtonian Behaviour of Blood on Pulsatile Flows in Stenotic Arteries Somkid Amornsamankul, Benchawan Wiwatanapataphee, Yong Hong Wu, Yongwimon Lenbury International Science
Vol:1, No:2, 27 Effect of Non-Newtonian Behaviour of Blood on Pulsatile Flows in Stenotic Arteries Somkid Amornsamankul, Benchawan Wiwatanapataphee, Yong Hong Wu, Yongwimon Lenbury International Science
