Left-right asymmetry: Nodal points
|
|
|
- Angelica Berry
- 6 years ago
- Views:
Transcription
1 Hypothesis 3251 Left-right asymmetry: Nodal points Mark Mercola Stem Cell and Regeneration Program, The Burnham Institute, N. Torrey Pines Road, La Jolla, CA 92037, USA ( Journal of Cell Science 116, The Company of Biologists Ltd doi: /jcs Summary The striking left-right asymmetry of visceral organs is known to depend on left- and right-side-specific cascades of gene expression during early embryogenesis. Now, developmental biologists are characterizing the earliest steps in asymmetry determination that dictate the sidedness of asymmetric gene expression. The proteins and structures involved control fascinating physiological processes, such as extracellular fluid flow and membrane voltage potential and yet little is known about how their activities are coordinated to control laterality. By analogy with intercellular signalling in certain epithelial and endothelial cells, however, it is reasonable to speculate that at least three of these players, monocilia, gap junction communication and the Ca 2+ channel polycystin-2, participate in a signalling pathway that propagates leftright cues through multicellular fields. Key words: Embryo, Organogenesis, Development, Asymmetry, Monocilia, Polycystin Introduction Nearly all organs of the thorax and abdomen are left-right (LR) asymmetric in anatomy, placement and, in some cases, physiology. LR asymmetry itself is a highly conserved feature of chordates, even though certain details of the anatomical asymmetry, such as sidedness of the venous system, vary between species. Perhaps the most striking aspect of LR asymmetry is that all normal individuals of a given species have identically oriented visceral asymmetry. Despite normal health, only about 1 in 8500 people exhibit complete mirror-image reversal of visceral organs (situs inversus). Whereas visceral organs show the most obvious asymmetry, stereotypic LR asymmetry extends to the brain and nervous system in humans; thus, it is intriguing to consider that LR asymmetry determination has profound implications for behavior and cognition. Conceptually, the generation of LR asymmetry can be considered to occur in three phases during early embryonic development (Fig. 1) (reviewed by Hamada et al., 2002; Mercola and Levin, 2001). During the first phase, the bilateral symmetry of the early embryo is broken and the LR axis becomes oriented with respect to the anteroposterior and dorsoventral body axes. This early pattern is processed and transduced into cascades of asymmetric gene expression that characterize the second phase. Although the asymmetric patterns of gene expression differ among species, a conserved feature in all animal species examined is the production of the secreted TGFβ-like factor Nodal (or a Nodal homologue) in the left lateral plate mesoderm. Asymmetric expression of a Nodal protein is critical as it provides a link to the third phase by inducing expression of the transcription factor Pitx2c. During the third phase, proteins such as Pitx2c control localized changes in cell migration, shape, proliferation and survival that render the stereotypic asymmetric anatomy of developing tissues. Proteins and structures involved in early LR asymmetry determination Several proteins or structures now appear to be components of the process that orients the axis of LR asymmetry (Fig. 2). Below, I focus on the proteins and structures that function upstream of asymmetric gene expression. Most of these are likely to act in the vicinity of the amniote node or analogous structures in amphibians and fish. Monocilia The mouse node is a structure at the distal tip of the E6.5 embryo consisting of a dorsal layer of ectoderm over a ventral layer of cells each with a single monocilium on their apical surface (Bellomo et al., 1996; Sulik et al., 1994) (Fig. 3). Although structurally different, the mouse node shares signalling properties with Hensen s node in the chick, Spemann s Organizer in amphibians and the shield in zebrafish. The defining characteristic of these structures is that, when grafted to an ectopic site, host tissue is induced to form a secondary body axis. The node forms at the anterior end of a furrow, known as the primitive streak, along which cells delaminate to form mesoderm. The line formed by the primitive streak and node correlates with the future dorsal midline of the embryo and lies along its axis of bilateral symmetry. The incidence of laterality defects correlates with immotile or absent node monocilia. Mutations in genes encoding dynein heavy chains in mice [lrd, also known as iv (Okada et al., 1999; Supp et al., 1997)] or humans [Dnahc5 (Olbrich et al., 2002)] are associated with mirror-image reversals of internal organs and, in mice, lead to formation of immotile node monocilia. Similarly, loss of Polaris [also known as Tg737, a hypomorphic allele of which is orpk (Murcia et al., 2000)], which is required for epithelial polarity of ventral node cells, and targeted disruption of the kinesin genes Kif3a or Kif3b (Marszalek et al., 1999; Nonaka et al., 1998; Takeda et al., 1999) in mice cause an absence of node monocilia concomitant with LR defects. In what must be one of the most elegant studies in the field, Nonaka et al. visualized the rotational movement of node monocilia by placing a fluorescent microsphere in the extracellular space of
2 3252 Journal of Cell Science 116 (16) (1) Axis of LR asymmetry is oriented and this information is disseminated through multicellular fields (2) Asymmetric pattern is amplified and propagated by cascades of gene expression (3) Proteins that influence morphogenesis are induced in target tissues Lefty2 Left Polycystin-2 and Ca 2+ signalling Gap junctions Nodal Pitx2c Morphogenetic proteins Midline barrier Monocilia, INV, LRD, Kif3A, Kif3B, Polaris Polaris L e f ft ty 1 y 1 embryos mounted in culture medium (Nonaka et al., 1998). They showed that the vortical movement of cilia creates a leftward fluid flow across the node and, on the basis of these data, proposed that the fluid flow increases the concentration of some diffusible determinant on the left side of the node. The direction of fluid flow is probably not due to the direction of ciliary rotation but to intrinsic properties of the node, such as its triangular shape or perhaps the inclination of the cilia. Support for the fluid flow model came from the observation that experimental pumping of fluid in the opposite direction tended to reverse patterns of asymmetric gene expression (Nonaka et al., 2002). This experiment is critical to the argument that the H + /K + -ATPase Gap junctions (Xenopus, chick) Phospho-Syndecan-2 Node monocilia (Mouse, chick, Xenopus, Zebrafish) (All species examined) (Xenopus, chick) Developmental stage (Xenopus) Nodal Pitx2c Cleavage Gastrulation Neurulation Organogenesis Right H + / K + -ATPase and membrane potential Fig. 1. Schematic pathway for the determination of LR asymmetry. LR asymmetry proceeds from a poorly understood initial process that orients direction (1) to propagation and amplification by cascades of gene expression that culminate in production of a Nodal protein on the left side of the embryo (2). The range of Nodal action is constrained by lefty proteins, in particular at the dorsal midline. A principal function of Nodal proteins is to regulate expression of Pitx2c and other proteins that influence morphogenesis in asymmetrically developing organs (3). Modified figure reproduced, with permission, from the Nature Publishing Group ( (Hamada et al., 2002). leftward fluid flow dictates LR asymmetry rather than occurs as an unrelated consequence of monocilia chirality. It is important to bear in mind, however, that the genetic experiments and, more to the point, the reversal of asymmetry by pumping fluid do not indicate that fluid flow initiates asymmetry, but show only that monociliary movement is important. Nonetheless, using the inherent chirality of monociliary rotation to provide LR orientation is the most conceptually satisfying model for the initiating step in the mouse. Are node monocilia involved in asymmetry determination in other species? Using an antibody to acetylated tubulin, Essner et al. saw monocilia in chick ventral node cells, dorsal blastopore cells of Xenopus early neurula, and in dorsal forerunner cells in the zebrafish shield at the end of gastrulation (Fig. 4) (Essner et al., 2002). In each case, monocilia appear before asymmetric expression of the pertinent Nodal homologue, which suggests they have an evolutionarily conserved function. However, in contrast to the situation in the mouse, asymmetric expression of other genes in chicks and Xenopus occurs prior to the appearance of monocilia (Levin, 1998; Levin et al., 1995) and asymmetry in the distribution of H + /K + -ATPase mrna in Xenopus or function in chick (see below). Moreover, transplant and regeneration studies revealed that nascent chick nodes receive LR asymmetry input from surrounding tissue and in the absence of these cues develop with abnormal asymmetry (Levin and Mercola, 1998a; Levin and Mercola, 1999; Pagan-Westphal and Tabin, 1998; Psychoyos and Stern, 1996; Yuan and Schoenwolf, 1998). Therefore, although their presence is indeed conserved, monocilia are unlikely to initiate LR asymmetry in all species. Gap junction communication Pharmacological or genetic perturbation of endogenous Fig. 2. Developmental stages when the various proteins and structures are likely to contribute to LR asymmetry determination. Animal species for which the components have been examined are listed in parentheses.
3 Left-right asymmetry 3253 Fig. 3. The mouse node and monocilia. Scanning electron micrographs (provided by Daisuke Watanabe and Hiroshi Hamada, Osaka University) showing the node at the distal tip of an E8.0 mouse embryo (A). Higher magnification views show the ventral node cells (B) and individual monocilia (C). Anteroposterior body axes (A, P) and direction of flow (arrow in B) to left side (L) are indicated. Magnifications: 200 (A); 700 (B) ; 7000 (C). gap junctional communication in early chick or Xenopus embryos disrupts expression of normally unilaterally expressed genes and alters organ situs (Levin and Mercola, 1998b; Levin and Mercola, 1999). The endogenous spatial pattern of gap junctional communication appears critical, because injection of a dominant negative connexin into dorsal blastomeres of Xenopus embryos or wild-type connexins into ventral blastomeres (but not the reverse) affects laterality; this indicates that a signal is propagated through dorsal blastomeres and impeded at the ventral midline. The effect of these manipulations on early gene expression, as well as the temporal window when embryos are sensitive to the drugs, suggests that gap junctional communication is critical in early-streak-stage chick embryos and between cleavage and the gastrula stage Xenopus (Fig. 2). Gap junctions are composed of twelve transmembrane connexin proteins, six in each apposing cell. Numerous connexins exist and, of these, Connexin43 appears crucial for chick asymmetry: it is present in cells surrounding the node and its depletion by antisense oligonucleotides unbiases the sidedness of Sonic hedgehog (Shh) and Nodal expression (Levin and Mercola, 1999). Polycystin-2 Polycystin-2, the gene product of PKD2, is mutated in autosomal dominant polycystic kidney disease and is a Ca 2+ - permeable cation channel (Cai et al., 1999; Gonzalez-Perret et al., 2001; Koulen et al., 2002). In addition to causing cardiac and kidney malformations, targeted disruption of PKD2 in mice causes hallmarks of LR disturbance that include randomization of embryo turning, right pulmonary isomerism (e.g. left lung exhibits lobation typical of the right lung), anomalous heart looping (including dextrocardia) and gut coiling defects (Pennekamp et al., 2002). Importantly, the normally left-sided nodal, lefty2 and Pitx2 expression in anterior lateral plate mesoderm is absent in PKD2 / embryos, which indicates that polycystin-2 acts upstream of asymmetric gene expression. Consistent with this possibility is the fact that polycystin-2 mrna is expressed strongly in and near the node, notochord and floorplate (Pennekamp et al., 1998) (R. D. Trelles, M. Levin and M. Mercola, unpublished). It is not yet clear how polycystin-2 influences laterality. Interestingly, elevated Ca 2+ levels prevent calmodulin from interacting with INV (Morgan et al., 2002; Yasuhiko et al., 2001), a protein that when mutated in mice causes mirrorimage organ reversals and results in slow and turbid fluid flow across the node (but does not reverse flow direction). Other Fig. 4. Conservation of node monocilia. mrna encoding endogenous left-right dynein (LRD), a protein involved in node monocilia in the mouse, is seen in Hensen s node in the chick, dorsal blastopore cells of the early neurula stage Xenopus, and the zebrafish shield (A,D,G,J; arrows). Monocilia are revealed on the apical surfaces of mouse ventral node cells and in cells of the related structures in chick, Xenopus and zebrafish embryos by immunostaining with anti-acetylated tubulin (B,E,H,K; arrows); a schematic representation is also shown (C,F,I,L). Reproduced, with permission, from the Nature Publishing Group ( (Essner et al., 2002).
4 3254 Journal of Cell Science 116 (16) than acting as a potential regulator of INV, polycystin-2 has not been linked to other proteins involved in LR asymmetry. Mutation of either polycystin-2 or INV, however, contributes to abnormal kidney tubule and duct growth, leading to formation of cysts. Monocilia on the surface of the tubule and duct epithelial cells are thought to act as sensors of tube diameter and trigger an intercellular Ca 2+ wave that negatively regulates cell proliferation and lumen expansion (Praetorius and Spring, 2001; Praetorius and Spring, 2003; Schwartz et al., 1997). The idea that a similar mechanism might operate to regulate asymmetric gene expression near the node has been proposed as an alternative to the fluid flow model (Brueckner, 2001) and is considered in more detail below. The H + /K + -ATPase A biological screen of small molecules known to affect ion channels and pumps for the ability to induce defects in heart looping, gut coiling and gall bladder position in early Xenopus embryos revealed that several compounds that cause laterality defects inhibit the H + /K + -ATPase, a pump that exchanges H + and K + across membranes (Levin et al., 2002). The window of drug sensitivity places the H + /K + -ATPase as the earliest known component of LR asymmetry determination in both Xenopus and chicks (Fig. 2). In addition to causing organ reversals, inhibition or misexpression of the H + /K + -ATPase unbiases asymmetric gene expression, including that of Shh, fgf8 (encoding fibroblast growth factor-8) and Wnt8c (encoding a secreted protein of the of the Wingless/Wnt family) in the chick node. Surprisingly, H + /K + -ATPase α mrna itself becomes selectively depleted on the left ventral side of Xenopus embryos by the 4-cell stage, well before the onset of embryonic transcription (which occurs at the mid-blastula transition when the embryo has about 1000 cells), making it the earliest known asymmetric mrna and revealing that LR patterning is present during the first cell cycle. In chick embryos, H + /K + -ATPase mrnas are first apparent adjacent to the primitive streak and developing node, but expression is symmetrical. Nonetheless, imaging studies suggest that the H + /K + -ATPase functions on the right, but not left, side of the streak and node to maintain cell membrane potentials. A domain of depolarized cells can be detected by elevated fluorescence of the potientiometric dye DiBAC 4 (3) on the left side of the developing streak and node. Exposure to BaCl 2 or omeprazole, which affects K + flux and H + /K + -ATPase activity, respectively, causes a mirror-image domain to appear on the right. This indicates that the H + /K + -ATPase normally maintains negative membrane potential on the right, which is consistent with its physiological properties. Thus, although it functions on the right side of both species, it is regulated at the mrna level in Xenopus and post-transcriptionally in chicks. Despite its importance for chick and Xenopus assymetry determination, there is no evidence indicating whether the H + /K + ATPase acts upstream of node monocilia, gap junctions or polycystin-2 or initiates a parallel signalling pathway. Epithelial structural proteins N-cadherin and claudin are also essential for normal LR asymmetry. N-cadherin is asymmetrically expressed in the chick node: it is highly expressed in the anterior right and posterior left margins of the node, corresponding to the asymmetric shape of the node itself (Garcia-Castro et al., 2000). Importantly, asymmetric expression of N-cadherin precedes asymmetric expression of Shh (left anterior node) but is also influenced by Shh. Addition of a blocking antibody to N-cadherin at stage 3-4, but not later, affects heart morphogenesis and Pitx2 expression. Curiously, Nodal and lefty expression are unaffected; thus, N-cadherin seems to function in a pathway parallel to that involving these proteins. Claudins are important cell adhesion molecules of tight junctions that are directly involved in intercellular sealing. Overexpression of either an intact protein or a C-terminally truncated version in Xenopus caused heterotaxia and bilateral expression of the Nodal homologue XNr-1 (Brizuela et al., 2001). Notwithstanding that these experiments were done in different species, the importance of N-cadherin and claudins highlight the importance of epithelial integrity in normal LR asymmetry determination. Syndecan-2 Yost and colleagues have shown by western analysis that a cytoplasmic domain of the heparin-sulphate proteoglycan syndecan-2 is phosphorylated in animal cap (presumptive ectoderm) cells on the right, but not left, half of the Xenopus embryo during gastrulation [at stage 11 (Kramer et al., 2002)]. Phosphorylation is thought to affect the ability of syndecan-2 to confer LR patterning on the mesoderm that comes into contact with the ectoderm during gastrulation. How syndecan- 2 might pass signals to the mesoderm is unclear but it might serve to localize a growth-factor-like protein within a signalling complex at the cell surface (Bernfield et al., 1999). The idea that syndecans mediate the transfer of LR information from ectoderm to mesoderm is particularly interesting and a critical question is whether or not a similar mechanism operates in species other than Xenopus. A model uniting monocilia, gap junction communication and polycystin-2 into a signalling pathway How (or, indeed, do) these very diverse proteins and structures work together to process or propagate LR asymmetry information? The fluid flow model suggests that monociliary action propels an extracellular determinant. Another possibility is that monocilia initiate a signal, such as a Ca 2+ wave, that spreads through the sheet of cells surrounding the node. Fig. 5 depicts how monocilia, gap junctions and polycystin-2 could function coordinately to propagate an intercellular Ca 2+ wave. Intercellular Ca 2+ waves are well known [for instance in endothelial, glial, and hepatic epithelial cells (for a review, see Sanderson, 1995)] and might also influence earliest asymmetric gene expression in and around the chick node, such as Shh, fgf8 and Wnt8c [a similar model has been proposed recently (Tabin and Vogan, 2003)]. Apart from the involvement of monocilia, gap junctions and polycystin-2 in LR asymmetry and expression in the vicinity of the node, evidence for the model comes from studies on autosomal dominant polycystic kidney disease, which is known to involve each of these proteins and structures. In normal kidney tubules and ducts, monocilia located on the apical surface of epithelial cells
5 Left-right asymmetry 3255 Ins(1,4,5)P 3 Ca 2+ Ca Fig. 5. Ca 2+ wave model. A hypothetical model incorporates monocilia, gap junctional communication and polycystin-2 in the vicinity of the node. By analogy with Ca 2+ waves in certain epithelial and endothelial cells, bending of monocilia on ventral node cells (shown for chick) are Ins(1,4,5)P 3 Ca 2+ proposed to trigger a Ca 2+ influx, perhaps through polycystin-2 (1), Ins(1,4,5)P 3 Ca 2+ Ca 2+ resulting in induction of a second messenger, such as Ins(1,4,5)P 3, (2) that passes through gap junctions to propagate an 3 intercellular Ca 2+ wave (3) to adjacent cells. The model does not predict whether the Ca 2+ wave is propagated leftwards, rightwards or both. Directional propagation would provide the simplest link to downstream gene expression and could be achieved in response to signals within the node, for instance if the monocilia sensed the leftward fluid flow [as proposed for the mouse (Tabin and Vogan, 2003)], or by the asymmetric localization of proteins, such as connexins, that are required to receive or propagate the Ca 2+ signal (see text). appear to sense lumen diameter by detecting changes in fluid dynamics (Nauli et al., 2003; Praetorius and Spring, 2001; Praetorius and Spring, 2003). Bending of the monocilia triggers Ca 2+ influx through ciliary or nearby channels, which in turn causes Ca 2+ release from inositol 1,4,5-trisphosphate [Ins(1,4,5)P 3 ]-sensitive intracellular stores. The signal propagates as a wave to surrounding cells by diffusion of a second messenger [such as Ins(1,4,5)P 3 ] through gap junction channels. Polycystin-2 co-localizes to kidney monocilia (Pazour et al., 2002; Yoder et al., 2002), where it might be the channel that permits entry of extracellular Ca 2+. Localization of polycystin-2, however, is still a matter of debate, and it has been proposed to function both as a Ca 2+ channel on the plasma membrane (Hanaoka et al., 2000) and as an Ins(1,4,5)P 3 - sensitive intracellular release channel on the ER (Koulen et al., 2002). In polycystic kidney disease, a lack of either polycystin-2 or monocilia causes focal proliferation of epithelial cells that eventually dilate into cysts. When applied to LR asymmetry determination at the node, the model makes several predictions: (1) a polycystin-2-dependent Ca 2+ wave should be visible in the vicinity of the node and might be asymmetric; (2) propagation of the Ca 2+ signal should be dependent on gap junctions and monocilia; and (3) the Ca 2+ signal should affect asymmetric gene expression. Of these, only monocilia are firmly linked to asymmetry determination and the remainder need to be substantiated. The model does not predict whether the Ca 2+ signal is propagated leftwards, rightwards or both. A sided pattern of propagation provides the simplest link to downstream gene expression but, if this is the case, what provides the orientation? Tabin and Vogan recently proposed that two types of node monocilia exist in the mouse node: one set to generate the leftward fluid flow and a second to sense it (Tabin and Vogan, 2003). This view, in which inherent asymmetry in node monocilary structures generates embryonic asymmetry, might apply to the mouse, where the leftward nodal fluid flow is the earliest known asymmetry. However, it is difficult to reconcile the conclusion that inherent properties of the mouse node generate the embryo s handed asymmetry with the observations that the developing chick node receives LR pattern from surrounding tissues (as discussed above). Thus, it is reasonable to propose, at least for the chick, that any sidedness to a monocilia/polycystin-2-dependent Ca 2+ signal might be dictated by an upstream signal, perhaps one dependent on the H + /K + -ATPase activity that maintains cell voltage potential on the right side of the node and streak at a developmental window that overlaps the time when monocilia are detected. Control over the propagation of a Ca 2+ signal could also be achieved by regulating some feature of epithelial structure or physiology required to receive or propagate the second messenger. It might be relevant, therefore, that Cx43 mrna is asymmetrically expressed on the right side of the chick node and might therefore impose directionality, but its role in this process is poorly understood beyond the published observation that antisense depletion of Cx43 mrna affects asymmetry of downstream gene expression (Levin and Mercola, 1999). Conclusions and perspectives The past few years have seen remarkable progress towards understanding how LR asymmetry is patterned and the next few should be as impressive. Clearly, the speculative and incomplete model that associates monocilia, gap junctions and polycystin-2 into a signalling pathway needs to be evaluated and refined. It is important to learn if (and how) upstream signals, such as generated by the H + /K + -ATPase, are coupled to signalling at the node and whether such signalling operates only in chick and Xenopus but not in the mouse. Indeed, a major challenge is to resolve whether early LR patterning information is initiated and processed similarly in different species. Differences in the timing of axial patterning, early
6 3256 Journal of Cell Science 116 (16) cleavage and gastrulation, and the anatomy of key signalling centers, such as the node or analogous structures, make the possibility of diverse mechanisms quite likely. Divergent chick, frog, fish and mouse evolution might have provided selective advantages that favored the orientation of LR asymmetry as early as the first cell cycle in animals such as the frog. Similarly, whether the node functions to initiate LR patterning, as monociliary motion might do in the mouse, or respond to earlier cues, as might occur in chick, could reflect divergent architectures of the node (Dathe et al., 2002), and this possibility needs to be addressed functionally. The application of recent advances in reverse genetics, such as RNAi, is certain to provide answers to these fascinating questions. Another longstanding issue to be resolved is why nearly 100% of homozygous inv mutant mice exhibit LR inversions (Yokoyama et al., 1993). The mutation deletes nearly the entire coding region of the cytoplasmic INV protein (Mochizuki et al., 1998; Morgan et al., 1998) and, in its presumed absence, nodal fluid flow has been described as slowed, but not reversed (Okada et al., 1999), thus frustrating attempts to couple INV to monociliary function. Perhaps related, therefore, are the observations that homozygous inv mutant embryos exhibit abnormal epithelial thickening on the edges of the node (Okada et al., 1999) and develop kidney cysts resembling those in polycystic kidney disease (PKD) (Mochizuki et al., 1998; Morgan et al., 1998; Yokoyama et al., 1993), which suggests that disturbances in epithelial character might affect the ability to propagate signalling downstream of monociliary function. Nonetheless, why these alterations would lead to reversals remains puzzling. Finally, it is important to distinguish between when LR asymmetry information is first generated and when it can be regenerated. The mammalian embryo, for instance, has a considerable ability to reconstitute axial pattern after experimental intervention, such that removal or addition of blastomeres of pre-implantation embryos can lead to perfectly normal animals. This plasticity has been misinterpreted as meaning that axial patterning occurs relatively later than in more mosaic embryos, such as those of the frog, as a result of interactions between blastomeres. Careful studies, however, have indicated that the animal-vegetal axis in the fertilized mouse egg, defined by the location of the polar body, correlates with the future long axis of bilateral symmetry in the blastocyst and, if left undisturbed, presages the anteroposterior body axis of the fetus (Gardner, 2001; Johnson, 2001). Any LR information laid down this early and perturbed experimentally might also be reconstituted during subsequent development. An example of this plasticity is the apparent de novo generation of asymmetry in Xenopus embryos by manipulations that act at multicellular stages. Fertilized Xenopus embryos normally acquire LR asymmetry by the two- to four-cell stage, as revealed by the selective depletion of H + /K + -ATPase α mrna from the ventral left blastomere (Levin et al., 2002). Yet previous studies showed that forced expression of the transcription factor Siamois (which mimics endogenous dorsal Siamois after the 1000 cell stage) induces secondary body axes on the ventral side of the embryo that are oriented ventral to ventral with respect the primary axis (belly-to-belly twins) and that these secondary axes can have normal LR asymmetry (Nascone and Mercola, 1997). The observation that manipulated embryos can bootstrap LR information well after the initial orientation event normally occurs suggests that multiple mechanisms might exist to discriminate left from right in the early embryo. I am grateful to Daisuke Watanabe and Hiroshi Hamada for providing Fig. 3, Joe Yost, Cliff Tabin and Tina Brueckner for providing Fig. 4, Nature Publishing for permission to reproduce copyrighted material, Mike Levin for comments on the manuscript and NIH/HLBI for supporting our asymmetry research (HL63271). References Bellomo, D., Lander, A., Harragan, I. and Brown, N. (1996). Cell proliferation in mammalian gastrulation: the ventral node and notochord are relatively quiescent. Dev. Dyn. 205, Bernfield, M., Gotte, M., Park, P. W., Reizes, O., Fitzgerald, M. L., Lincecum, J. and Zako, M. (1999). Functions of cell surface heparan sulfate proteoglycans. Annu. Rev. Biochem. 68, Brizuela, B., Wessely, O. and DeRobertis, E. (2001). Overexpression of the Xenopus tight-junction protein Claudin causes randomization of the leftright body axis. Dev. Biol. 230, Brueckner, M. (2001). Cilia propel the embryo in the right direction. Am. J. Med. Genet. 101, Cai, Y., Maeda, Y., Cedzich, A., Torres, V. E., Wu, G., Hayashi, T., Mochizuki, T., Park, J. H., Witzgall, R. and Somlo, S. (1999). Identification and characterization of polycystin-2, the PKD2 gene product. J. Biol. Chem. 274, Dathe, V., Gamel, A., Manner, J., Brand-Saberi, B. and Christ, B. (2002). Morphological left-right asymmetry of Hensen s node precedes the asymmetric expression of Shh and Fgf8 in the chick embryo. Anat. Embryol. 205, Essner, J. J., Vogan, K. J., Wagner, M. K., Tabin, C. J., Yost, H. J. and Brueckner, M. (2002). Conserved function for embryonic nodal cilia. Nature 418, Garcia-Castro, M. I., Vielmetter, E. and Bronner-Fraser, M. (2000). N- Cadherin, a cell adhesion molecule involved in establishment of embryonic left-right asymmetry. Science 288, Gardner, R. L. (2001). Specification of embryonic axes begins before cleavage in normal mouse development. Development 128, Gonzalez-Perret, S., Kim, K., Ibarra, C., Damiano, A. E., Zotta, E., Batelli, M., Harris, P. C., Reisin, I. L., Arnaout, M. A. and Cantiello, H. F. (2001). Polycystin-2, the protein mutated in autosomal dominant polycystic kidney disease (ADPKD), is a Ca 2+ -permeable nonselective cation channel.[comment]. Proc. Natl. Acad. Sci. USA 98, Hamada, H., Meno, C., Watanabe, D. and Saijoh, Y. (2002). Establishment of vertebrate left-right asymmetry. Nat. Rev. Genet. 3, Hanaoka, K., Qian, F., Boletta, A., Bhunia, A. K., Piontek, K., Tsiokas, L., Sukhatme, V. P., Guggino, W. B. and Germino, G. G. (2000). Coassembly of polycystin-1 and -2 produces unique cation-permeable currents. Nature 408, Johnson, M. H. (2001). Mammalian development: axes in the egg? Curr. Biol. 11, R281-R284. Koulen, P., Cai, Y., Geng, L., Maeda, Y., Nishimura, S., Witzgall, R., Ehrlich, B. E. and Somlo, S. (2002). Polycystin-2 is an intracellular calcium release channel. Nat. Cell Biol. 4, Kramer, K. L., Barnette, J. E. and Yost, H. J. (2002). PKCgamma regulates syndecan-2 inside-out signaling during xenopus left-right development. Cell 111, Levin, M. (1998). The roles of activn and follistatin signaling in chick gastrulation. Int. J. Dev. Biol. 42, Levin, M. and Mercola, M. (1998a). Evolutionary conservation of mechanisms upstream of asymmetric Nodal expression: reconciling chick and Xenopus. Dev. Genet. 23, Levin, M. and Mercola, M. (1998b). Gap junctions are involved in the early generation of left-right asymmetry. Dev. Biol. 202, Levin, M. and Mercola, M. (1999). Gap junction-mediated transfer of leftright patterning signals in the early chick blastoderm is upstream of Shh asymmetry in the node. Development 126, Levin, M., Johnson, R. L., Stern, C. D., Kuehn, M. and Tabin, C. (1995). A molecular pathway determining left-right asymmetry in chick embryogenesis. Cell 82, Levin, M., Thorlin, T., Robinson, K., Nogi, T. and Mercola, M. (2002).
7 Left-right asymmetry 3257 Asymmetries in H(+)/K(+)-ATPase and cell membrane potentials comprise a very early step in left-right patterning. Cell 111, Marszalek, J. R., Ruiz-Lozano, P., Roberts, E., Chien, K. R. and Goldstein, L. S. (1999). Situs inversus and embryonic ciliary morphogenesis defects in mouse mutants lacking the KIF3A subunit of kinesin-ii. Proc. Natl. Acad. Sci. USA 96, Mercola, M. and Levin, M. (2001). Left-right asymmetry determination in vertebrates. Annu. Rev. Cell Dev. Biol. 17, Mochizuki, T., Saijoh, Y., Tsuchiya, K., Shirayoshi, Y., Takai, S., Taya, C., Yonekawa, H., Overbeek, P., Hamada, H. and Yokoyama, T. (1998). Cloning of inv, a gene that controls left/right asymmetry and kidney development. Nature 395, Morgan, D., Turnpenny, L., Goodship, J., Dai, W., Majumder, K., Matthews, L., Gardner, A., Schuster, G., Vien, L., Harrison, W. et al. (1998). Inversin, a novel gene in the vertebrate left-right axis pathway, is partially deleted in the inv mouse. Nat. Genet. 20, Morgan, D., Goodship, J., Essner, J. J., Vogan, K. J., Turnpenny, L., Yost, H. J., Tabin, C. J. and Strachan, T. (2002). The left-right determinant inversin has highly conserved ankyrin repeat and IQ domains and interacts with calmodulin. Hum. Genet. 110, Murcia, N. S., Richards, W. G., Yoder, B. K., Mucenski, M. L., Dunlap, J. R. and Woychik, R. P. (2000). The Oak Ridge Polycystic Kidney (orpk) disease gene is required for left-right axis determination. Development 127, Nascone, N. and Mercola, M. (1997). Organizer induction determines leftright asymmetry in Xenopus. Dev. Biol. 189, Nauli, S. M., Alenghat, F. J., Luo, Y., Williams, E., Vassilev, P., Li, X., Elia, A. E., Lu, W., Brown, E. M., Quinn, S. J. et al. (2003). Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat. Genet. 33, Nonaka, S., Tanaka, Y., Okada, Y., Takeda, S., Harada, A., Kanai, Y., Kido, M. and Hirokawa, N. (1998). Randomization of left-right asymmetry due to loss of nodal cilia generating leftward flow of extraembryonic fluid in mice lacking KIF3B motor protein [published erratum appears in Cell 1999 Oct 1;99(1):117]. Cell 95, Nonaka, S., Shiratori, H., Saijoh, Y. and Hamada, H. (2002). Determination of left-right patterning of the mouse embryo by artificial nodal flow. Nature 418, Okada, Y., Nonaka, S., Tanaka, Y., Saijoh, Y., Hamada, H. and Hirokawa, N. (1999). Abnormal nodal flow precedes situs inversus in iv and inv mice. Mol. Cell 4, Olbrich, H., Haffner, K., Kispert, A., Volkel, A., Volz, A., Sasmaz, G., Reinhardt, R., Hennig, S., Lehrach, H., Konietzko, N. et al. (2002). Mutations in DNAH5 cause primary ciliary dyskinesia and randomization of left-right asymmetry. Nat. Genet. 30, Pagan-Westphal, S. M. and Tabin, C. J. (1998). The transfer of left-right positional information during chick embryogenesis. Cell 93, Pazour, G. J., San Agustin, J. T., Follit, J. A., Rosenbaum, J. L. and Witman, G. B. (2002). Polycystin-2 localizes to kidney cilia and the ciliary level is elevated in orpk mice with polycystic kidney disease. Curr. Biol. 12, R378-R380. Pennekamp, P., Bogdanova, N., Wilda, M., Markoff, A., Hameister, H., Horst, J. and Dworniczak, B. (1998). Characterization of the murine polycystic kidney disease (Pkd2) gene. Mamm. Genome 9, Pennekamp, P., Karcher, C., Fischer, A., Schweickert, A., Skryabin, B., Horst, J., Blum, M. and Dworniczak, B. (2002). The ion channel polycystin-2 is required for left-right axis determination in mice. Curr. Biol. 12, Praetorius, H. A. and Spring, K. R. (2001). Bending the MDCK cell primary cilium increases intracellular calcium. J. Membr. Biol. 184, Praetorius, H. A. and Spring, K. R. (2003). Removal of the MDCK Cell Primary Cilium Abolishes Flow Sensing. J. Membr. Biol. 191, Psychoyos, D. and Stern, C. D. (1996). Restoration of the organizer after radical ablation of Hensen s node and the anterior primitive streak in the chick embryo. Development 122, Sanderson, M. J. (1995). Intercellular calcium waves mediated by inositol trisphosphate. Ciba Found. Symp. 188, Schwartz, E. A., Leonard, M. L., Bizios, R. and Bowser, S. S. (1997). Analysis and modeling of the primary cilium bending response to fluid shear. Am. J. Physiol. 272, F132-F138. Sulik, K., Dehart, D. B., Inagaki, T., Carson, J. L., Vrablic, T., Gesteland, K. and Schoenwolf, G. C. (1994). Morphogeneisis of the murine node and notochordal plate. Dev. Dyn. 201, Supp, D., Witte, D., Potter, S. and Brueckner, M. (1997). Mutation of an axonemal dynein affects left-right asymmetry in inversus viscerum mice. Nature 389, Tabin, C. J. and Vogan, K. J. (2003). A two-cilia model for vertebrate leftright axis specification. Genes Dev. 17, 1-6. Takeda, S., Yonekawa, Y., Tanaka, Y., Okada, Y., Nonaka, S. and Hirokawa, N. (1999). Left-right asymmetry and kinesin superfamily protein KIF3A: new insights in determination of laterality and mesoderm induction by kif3a / mice analysis. J. Cell Biol. 145, Yasuhiko, Y., Imai, F., Ookubo, K., Takakuwa, Y., Shiokawa, K. and Yokoyama, T. (2001). Calmodulin binds to inv protein: implication for the regulation of inv function. Dev. Growth Differ. 43, Yoder, B. K., Hou, X. and Guay-Woodford, L. M. (2002). The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J. Am. Soc. Nephrol. 13, Yokoyama, T., Copeland, N. G., Jenkins, N. G., Montgomery, C. A., Elder, F. F. and Overbeek, P. A. (1993). Reversal of left-right asymmetry: a situs inversus mutation. Science 260, Yuan, S. and Schoenwolf, G. C. (1998). De novo induction of the organizer and formation of the primitive streak in an experimental model of notochord reconstititution in avian embryos. Development 125,
Axis Formation and Mesoderm Induction
 Developmental Biology Biology 4361 Axis Formation and Mesoderm Induction October 27, 2005 Amphibian anteroposterior specification polarized eggs animal/vegetal pigment yolk v. clear cytoplasm mitochondrial
Developmental Biology Biology 4361 Axis Formation and Mesoderm Induction October 27, 2005 Amphibian anteroposterior specification polarized eggs animal/vegetal pigment yolk v. clear cytoplasm mitochondrial
Primary Cilia of inv/inv Mouse Renal Epithelial Cells Sense Physiological Fluid Flow: Bending of Primary Cilia and Ca 2+ Influx
 CELL STRUCTURE AND FUNCTION 30: 93 100 (2005) 2005 by Japan Society for Cell Biology Primary Cilia of inv/inv Mouse Renal Epithelial Cells Sense Physiological Fluid Flow: Bending of Primary Cilia and Ca
CELL STRUCTURE AND FUNCTION 30: 93 100 (2005) 2005 by Japan Society for Cell Biology Primary Cilia of inv/inv Mouse Renal Epithelial Cells Sense Physiological Fluid Flow: Bending of Primary Cilia and Ca
Report. Cilia-Driven Leftward Flow Determines Laterality in Xenopus
 Current Biology 17, 60 66, January 9, 2007 ª2007 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.10.067 Cilia-Driven Leftward Flow Determines Laterality in Xenopus Report Axel Schweickert, 1 Thomas
Current Biology 17, 60 66, January 9, 2007 ª2007 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.10.067 Cilia-Driven Leftward Flow Determines Laterality in Xenopus Report Axel Schweickert, 1 Thomas
The left-right axis in the mouse: from origin to morphology
 REVIEW 2095 Development 133, 2095-2104 (2006) doi:10.1242/dev.02384 The left-right axis in the mouse: from origin to morphology Hidetaka Shiratori and Hiroshi Hamada* The past decade or so has seen rapid
REVIEW 2095 Development 133, 2095-2104 (2006) doi:10.1242/dev.02384 The left-right axis in the mouse: from origin to morphology Hidetaka Shiratori and Hiroshi Hamada* The past decade or so has seen rapid
Localization and Loss-of-Function Implicates Ciliary Proteins in Early, Cytoplasmic Roles in Left-Right Asymmetry
 DEVELOPMENTAL DYNAMICS 234:176 189, 2005 PATTERNS & PHENOTYPES Localization and Loss-of-Function Implicates Ciliary Proteins in Early, Cytoplasmic Roles in Left-Right Asymmetry Dayong Qiu, Shing-Ming Cheng,
DEVELOPMENTAL DYNAMICS 234:176 189, 2005 PATTERNS & PHENOTYPES Localization and Loss-of-Function Implicates Ciliary Proteins in Early, Cytoplasmic Roles in Left-Right Asymmetry Dayong Qiu, Shing-Ming Cheng,
Embryonic Left-Right Asymmetry:
 Embryonic Left-Right Asymmetry: A fundamental problem of patterning at the intersection of cell, developmental, and evolutionary biology Michael Levin The Forsyth Institute, and Harvard School of Dental
Embryonic Left-Right Asymmetry: A fundamental problem of patterning at the intersection of cell, developmental, and evolutionary biology Michael Levin The Forsyth Institute, and Harvard School of Dental
Motor protein control of ion flux is an early step in embryonic left right asymmetry
 Motor protein control of ion flux is an early step in embryonic left right asymmetry Michael Levin Summary The invariant left right asymmetry of animal body plans raises fascinating questions in cell,
Motor protein control of ion flux is an early step in embryonic left right asymmetry Michael Levin Summary The invariant left right asymmetry of animal body plans raises fascinating questions in cell,
Twinning and Embryonic Left-Right Asymmetry
 Twinning and Embryonic Left-Right Asymmetry Michael Levin Bldg. C1, 5 th floor Cell Biology dept. Harvard Medical School 240 Longwood Ave. Boston, MA 02115 U.S.A. Tel. (617) 432-0044 Fax. (617) 432-1144
Twinning and Embryonic Left-Right Asymmetry Michael Levin Bldg. C1, 5 th floor Cell Biology dept. Harvard Medical School 240 Longwood Ave. Boston, MA 02115 U.S.A. Tel. (617) 432-0044 Fax. (617) 432-1144
Wnt signaling and PKA control Nodal expression and left-right determination in the chick embryo
 Development 128, 3189-3195 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV9773 3189 Wnt signaling and PKA control Nodal expression and left-right determination in the chick embryo
Development 128, 3189-3195 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV9773 3189 Wnt signaling and PKA control Nodal expression and left-right determination in the chick embryo
Ángel Raya* and Juan Carlos Izpisúa Belmonte
 Left right asymmetry in the vertebrate embryo: from early information to higher-level integration Ángel Raya* and Juan Carlos Izpisúa Belmonte Abstract Although vertebrates seem to be essentially bilaterally
Left right asymmetry in the vertebrate embryo: from early information to higher-level integration Ángel Raya* and Juan Carlos Izpisúa Belmonte Abstract Although vertebrates seem to be essentially bilaterally
LEFT-RIGHT ASYMMETRY DETERMINATION
 Annu. Rev. Cell Dev. Biol. 2001. 17:779 805 Copyright c 2001 by Annual Reviews. All rights reserved LEFT-RIGHT ASYMMETRY DETERMINATION IN VERTEBRATES Mark Mercola Department of Cell Biology, Harvard Medical
Annu. Rev. Cell Dev. Biol. 2001. 17:779 805 Copyright c 2001 by Annual Reviews. All rights reserved LEFT-RIGHT ASYMMETRY DETERMINATION IN VERTEBRATES Mark Mercola Department of Cell Biology, Harvard Medical
The T Box Transcription Factor No Tail in Ciliated Cells Controls Zebrafish Left-Right Asymmetry
 Current Biology, Vol. 14, 685 690, April 20, 2004, 2004 Elsevier Ltd. All rights reserved. DOI 10.1016/j.cub.2004.04.002 The T Box Transcription Factor No Tail in Ciliated Cells Controls Zebrafish Left-Right
Current Biology, Vol. 14, 685 690, April 20, 2004, 2004 Elsevier Ltd. All rights reserved. DOI 10.1016/j.cub.2004.04.002 The T Box Transcription Factor No Tail in Ciliated Cells Controls Zebrafish Left-Right
Consistent Left-Right Asymmetry Cannot be Established by Late Organizers in Xenopus Unless the Late Organizer is a Conjoined Twin
 University of Massachusetts Amherst From the SelectedWorks of Laura Vandenberg April 1, 2010 Consistent Left-Right Asymmetry Cannot be Established by Late Organizers in Xenopus Unless the Late Organizer
University of Massachusetts Amherst From the SelectedWorks of Laura Vandenberg April 1, 2010 Consistent Left-Right Asymmetry Cannot be Established by Late Organizers in Xenopus Unless the Late Organizer
Urogenital Development
 2-5-03 Urogenital Development Greg Dressler Assoc. Professor Dept. of Pathology x46490 Dressler@umich.edu The Origin of the Kidney In the vertebrate embryo, the first stage of kidney development occurs
2-5-03 Urogenital Development Greg Dressler Assoc. Professor Dept. of Pathology x46490 Dressler@umich.edu The Origin of the Kidney In the vertebrate embryo, the first stage of kidney development occurs
Two Populations of Node Monocilia Initiate Left-Right Asymmetry in the Mouse
 Cell, Vol. 114, 61 73, July 11, 2003, Copyright 2003 by Cell Press Two Populations of Node Monocilia Initiate Left-Right Asymmetry in the Mouse comes restricted to the left side of the node (Collignon
Cell, Vol. 114, 61 73, July 11, 2003, Copyright 2003 by Cell Press Two Populations of Node Monocilia Initiate Left-Right Asymmetry in the Mouse comes restricted to the left side of the node (Collignon
THE EMBRYONIC ORIGINS OF LEFT-RIGHT ASYMMETRY
 CONTROVERSY The consistent asymmetry of organs such as the heart, viscera, and brain raises profound questions in developmental and cell biology. It is also of high biomedical significance, because several
CONTROVERSY The consistent asymmetry of organs such as the heart, viscera, and brain raises profound questions in developmental and cell biology. It is also of high biomedical significance, because several
Targeted deletion of the ATP binding domain of left-right dynein confirms its role in specifying development of left-right asymmetries
 Development 126, 5495-5504 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV3019 5495 Targeted deletion of the ATP binding domain of left-right dynein confirms its role in specifying
Development 126, 5495-5504 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV3019 5495 Targeted deletion of the ATP binding domain of left-right dynein confirms its role in specifying
The compulsion of chirality: toward an understanding of left right asymmetry
 PERSPECTIVE The compulsion of chirality: toward an understanding of left right asymmetry Michael Levin and Mark Mercola 1 Department of Cell Biology, Harvard Medical School, Boston, Massachusetts 02115
PERSPECTIVE The compulsion of chirality: toward an understanding of left right asymmetry Michael Levin and Mark Mercola 1 Department of Cell Biology, Harvard Medical School, Boston, Massachusetts 02115
Polaris and Polycystin-2 in dorsal forerunner cells and Kupffer s vesicle are required for specification of the zebrafish left right axis
 Developmental Biology 287 (2005) 274 288 www.elsevier.com/locate/ydbio Polaris and Polycystin-2 in dorsal forerunner cells and Kupffer s vesicle are required for specification of the zebrafish left right
Developmental Biology 287 (2005) 274 288 www.elsevier.com/locate/ydbio Polaris and Polycystin-2 in dorsal forerunner cells and Kupffer s vesicle are required for specification of the zebrafish left right
More Than 95% Reversal of Left Right Axis Induced by Right-Sided Hypodermic Microinjection of Activin into Xenopus Neurula Embryos
 Developmental Biology 221, 321 336 (2000) doi:10.1006/dbio.2000.9666, available online at http://www.idealibrary.com on More Than 95% Reversal of Left Right Axis Induced by Right-Sided Hypodermic Microinjection
Developmental Biology 221, 321 336 (2000) doi:10.1006/dbio.2000.9666, available online at http://www.idealibrary.com on More Than 95% Reversal of Left Right Axis Induced by Right-Sided Hypodermic Microinjection
Lecture IV. Mechanisms of Neural. Neural Development
 Lecture IV. Mechanisms of Neural Bio 3411 Monday 1 Readings NEUROSCIENCE: 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) References : Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994).
Lecture IV. Mechanisms of Neural Bio 3411 Monday 1 Readings NEUROSCIENCE: 5 th ed, pp 477-506 (sorta) 4 th ed, pp 545-575 (sorta) References : Fainsod, A., Steinbeisser, H., & De Robertis, E. M. (1994).
Kupffer s vesicle is a ciliated organ of asymmetry in the zebrafish embryo that initiates left-right development of the brain, heart and gut
 First posted online on 16 February 2005 as 10.1242/dev.01663 Access the most recent epress version at online http://dev.biologists.org/lookup/doi/10.1242/dev.01663 publication date 16 February 2005 1247
First posted online on 16 February 2005 as 10.1242/dev.01663 Access the most recent epress version at online http://dev.biologists.org/lookup/doi/10.1242/dev.01663 publication date 16 February 2005 1247
Determination of left/right asymmetric expression of nodal by a left side-specific enhancer with sequence similarity to a lefty-2 enhancer
 Determination of left/right asymmetric expression of nodal by a left side-specific enhancer with sequence similarity to a lefty-2 enhancer Hitoshi Adachi, Yukio Saijoh, Kyoko Mochida, Sachiko Ohishi, Hiromi
Determination of left/right asymmetric expression of nodal by a left side-specific enhancer with sequence similarity to a lefty-2 enhancer Hitoshi Adachi, Yukio Saijoh, Kyoko Mochida, Sachiko Ohishi, Hiromi
Micro-bio-fluidica: The role of fluid dynamics in embryonic development and other small biological structures.. Oreste Piro
 Micro-bio-fluidica: The role of fluid dynamics in embryonic development and other small biological structures.. Oreste Piro University of Balearic Islands, Palma, Mallorca, Spain Colaborators: Julyan H.
Micro-bio-fluidica: The role of fluid dynamics in embryonic development and other small biological structures.. Oreste Piro University of Balearic Islands, Palma, Mallorca, Spain Colaborators: Julyan H.
A study of cilia density in sea urchin embryos during gastrulation
 A study of cilia density in sea urchin embryos during gastrulation Steve Das Independent Research Project Report Bio 254 Developmental Biology May 3, 2012 Introduction Embryo growth and development is
A study of cilia density in sea urchin embryos during gastrulation Steve Das Independent Research Project Report Bio 254 Developmental Biology May 3, 2012 Introduction Embryo growth and development is
When you see this diagram, remember that you are looking at the embryo from above, through the amniotic cavity, where the epiblast appears as an oval
 When you see this diagram, remember that you are looking at the embryo from above, through the amniotic cavity, where the epiblast appears as an oval disc 2 Why the embryo needs the vascular system? When
When you see this diagram, remember that you are looking at the embryo from above, through the amniotic cavity, where the epiblast appears as an oval disc 2 Why the embryo needs the vascular system? When
Vertebrate Limb Patterning
 Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Vertebrate Limb Patterning What makes limb patterning an interesting/useful developmental system How limbs develop Key events in limb development positioning and specification initiation of outgrowth establishment
Histology = the study of tissues. Tissue = a complex of cells that have a common function
 { EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
{ EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
Pkd1l1 establishes left-right asymmetry and physically interacts with Pkd2
 Access Development the most First recent posted version epress online at on online http://dev.biologists.org/lookup/doi/10.1242/dev.058149 9 February publication 2011 as 10.1242/dev.058149 date 9 February
Access Development the most First recent posted version epress online at on online http://dev.biologists.org/lookup/doi/10.1242/dev.058149 9 February publication 2011 as 10.1242/dev.058149 date 9 February
a) They are the most common cause of pediatric kidney failure. b) They are always symptomatic. c) They can be asymmetric.
 Practice questions: 1. The paraxial mesoderm gives rise to somites. The structure of the somite a) is a loose mesenchymal sheet that will migrate toward the notochord. b) is an epithelial rosette with
Practice questions: 1. The paraxial mesoderm gives rise to somites. The structure of the somite a) is a loose mesenchymal sheet that will migrate toward the notochord. b) is an epithelial rosette with
Cell Type Nervous System I. Developmental Readout. Foundations. Stem cells. Organ formation. Human issues.
 7.72 10.11.06 Cell Type Nervous System I Human issues Organ formation Stem cells Developmental Readout Axes Cell type Axon guidance 3D structure Analysis Model + + organisms Foundations Principles 1 What
7.72 10.11.06 Cell Type Nervous System I Human issues Organ formation Stem cells Developmental Readout Axes Cell type Axon guidance 3D structure Analysis Model + + organisms Foundations Principles 1 What
Diversity and convergence in the mechanisms establishing L/R asymmetry in metazoa
 Review Diversity and convergence in the mechanisms establishing L/R asymmetry in metazoa Jean-Baptiste Coutelis 1,2,3, Nicanor González-Morales 1,2,3, Charles Géminard 1,2,3 & Stéphane Noselli 1,2,3,*
Review Diversity and convergence in the mechanisms establishing L/R asymmetry in metazoa Jean-Baptiste Coutelis 1,2,3, Nicanor González-Morales 1,2,3, Charles Géminard 1,2,3 & Stéphane Noselli 1,2,3,*
NIH Public Access Author Manuscript Nature. Author manuscript; available in PMC 2011 March 28.
 NIH Public Access Author Manuscript Published in final edited form as: Nature. 2010 July 15; 466(7304): 378 382. doi:10.1038/nature09129. Planar cell polarity breaks the bilateral symmetry by controlling
NIH Public Access Author Manuscript Published in final edited form as: Nature. 2010 July 15; 466(7304): 378 382. doi:10.1038/nature09129. Planar cell polarity breaks the bilateral symmetry by controlling
Cell cycle arrest in node cells governs ciliogenesis at the node to break left-right symmetry
 ERRATUM Development 138, 4334 (2011) doi:10.1242/dev.073650 2011. Published by The Company of Biologists Ltd Cell cycle arrest in node cells governs ciliogenesis at the node to break left-right symmetry
ERRATUM Development 138, 4334 (2011) doi:10.1242/dev.073650 2011. Published by The Company of Biologists Ltd Cell cycle arrest in node cells governs ciliogenesis at the node to break left-right symmetry
Biology 4361 Developmental Biology. October 11, Multiple choice (one point each)
 Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
Left-Right Asymmetry and Cardiac Looping: Implications for Cardiac Development and Congenital Heart Disease
 A R T I C L E American Journal of Medical Genetics (Semin. Med. Genet.) 97:271±279 (2000) Left-Right Asymmetry and Cardiac Looping: Implications for Cardiac Development and Congenital Heart Disease IRFAN
A R T I C L E American Journal of Medical Genetics (Semin. Med. Genet.) 97:271±279 (2000) Left-Right Asymmetry and Cardiac Looping: Implications for Cardiac Development and Congenital Heart Disease IRFAN
Lecture 22: A little Neurobiology
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
Neuroepithelial Cells and Neural Differentiation
 Neuroepithelial Cells and Neural Differentiation Neurulation The cells of the neural tube are NEUROEPITHELIAL CELLS Neural crest cells migrate out of neural tube Neuroepithelial cells are embryonic stem
Neuroepithelial Cells and Neural Differentiation Neurulation The cells of the neural tube are NEUROEPITHELIAL CELLS Neural crest cells migrate out of neural tube Neuroepithelial cells are embryonic stem
Inner ear development Nervous system development
 Upcoming Sessions April 22: Nervous System Development Lecture April 24: Reviews of Axonal Pathfinding in Sensory Systems April 29: Inner Ear Development Lecture May 1: May 6: May 8: Auditory System Pathfinding
Upcoming Sessions April 22: Nervous System Development Lecture April 24: Reviews of Axonal Pathfinding in Sensory Systems April 29: Inner Ear Development Lecture May 1: May 6: May 8: Auditory System Pathfinding
Research article. Summary. Introduction
 Research article 4999 Targeted deletion of the novel cytoplasmic dynein md2lic disrupts the embryonic organiser, formation of the body axes and specification of ventral cell fates Amer Ahmed Rana 1, *,
Research article 4999 Targeted deletion of the novel cytoplasmic dynein md2lic disrupts the embryonic organiser, formation of the body axes and specification of ventral cell fates Amer Ahmed Rana 1, *,
A Genetic Program for Embryonic Development
 Concept 18.4: A program of differential gene expression leads to the different cell types in a multicellular organism During embryonic development, a fertilized egg gives rise to many different cell types
Concept 18.4: A program of differential gene expression leads to the different cell types in a multicellular organism During embryonic development, a fertilized egg gives rise to many different cell types
Supplementary Figure 1. A microarray screen of organizers compared to non-organizer tissue reveals a putative organizer gene set.
 Supplementary Figure 1. A microarray screen of organizers compared to non-organizer tissue reveals a putative organizer gene set. (a, b) Venn diagrams of 31 enriched (a) and 17 depleted (b) genes significantly
Supplementary Figure 1. A microarray screen of organizers compared to non-organizer tissue reveals a putative organizer gene set. (a, b) Venn diagrams of 31 enriched (a) and 17 depleted (b) genes significantly
Polarity and Segmentation. Chapter Two
 Polarity and Segmentation Chapter Two Polarization Entire body plan is polarized One end is different than the other Head vs. Tail Anterior vs. Posterior Front vs. Back Ventral vs. Dorsal Majority of neural
Polarity and Segmentation Chapter Two Polarization Entire body plan is polarized One end is different than the other Head vs. Tail Anterior vs. Posterior Front vs. Back Ventral vs. Dorsal Majority of neural
Regionalization of the nervous system. Paul Garrity 7.68J/9.013J February 25, 2004
 Regionalization of the nervous system Paul Garrity 7.68J/9.013J February 25, 2004 Patterning along: Rostral/Caudal (AP) axis Dorsal/Ventral (DV) axis Start with DV axial patterning in Spinal Cord Dorsal/Ventral
Regionalization of the nervous system Paul Garrity 7.68J/9.013J February 25, 2004 Patterning along: Rostral/Caudal (AP) axis Dorsal/Ventral (DV) axis Start with DV axial patterning in Spinal Cord Dorsal/Ventral
Calcium fluxes in dorsal forerunner cells antagonize -catenin and alter left-right patterning
 RESEARCH ARTICLE 75 Development 135, 75-84 (2008) doi:10.1242/dev.004713 Calcium fluxes in dorsal forerunner cells antagonize -catenin and alter left-right patterning Igor Schneider 1, Douglas W. Houston
RESEARCH ARTICLE 75 Development 135, 75-84 (2008) doi:10.1242/dev.004713 Calcium fluxes in dorsal forerunner cells antagonize -catenin and alter left-right patterning Igor Schneider 1, Douglas W. Houston
Developmental Biology
 Developmental Biology 336 (2009) 183 191 Contents lists available at ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology A gap junction connexin is required in the
Developmental Biology 336 (2009) 183 191 Contents lists available at ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology A gap junction connexin is required in the
keywords: embryo, morphogenesis, axis, left-right, asymmetry, connexin-43, dynein, laterality
 Two Molecular Models of Initial Left-Right Asymmetry Generation Michael Levin 1, Ph.D. and Nanette Nascone, M.S. Cell Biology dept. Harvard Medical School 200 Longwood Ave. Boston, MA 02115 1 Author for
Two Molecular Models of Initial Left-Right Asymmetry Generation Michael Levin 1, Ph.D. and Nanette Nascone, M.S. Cell Biology dept. Harvard Medical School 200 Longwood Ave. Boston, MA 02115 1 Author for
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM
 STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
Stephan Sauer* and Amar J. S. Klar*
 ORIGINAL RESEARCH ARTICLE published: 16 November 2012 doi: 10.3389/fonc.2012.00166 Left-right symmetry breaking in mice by left-right dynein may occur via a biased chromatid segregation mechanism, without
ORIGINAL RESEARCH ARTICLE published: 16 November 2012 doi: 10.3389/fonc.2012.00166 Left-right symmetry breaking in mice by left-right dynein may occur via a biased chromatid segregation mechanism, without
Fig. S1. RT-PCR analyses of the expression and distribution of Xdscr6 transcripts during early development.
 Fig. S1. RT-PCR analyses of the expression and distribution of Xdscr6 transcripts during early development. (A) Temporal expression of Xdscr6 at various stages (numbers on the top) and its distribution
Fig. S1. RT-PCR analyses of the expression and distribution of Xdscr6 transcripts during early development. (A) Temporal expression of Xdscr6 at various stages (numbers on the top) and its distribution
Biology 4361 Developmental Biology Exam 1 ID#: October 11, 2005
 Biology 4361 Developmental Biology Name: Key Exam 1 ID#: October 11, 2005 Multiple choice (one point each) 1. Primordial germ cells a. are immortal b. produce polar bodies c. are haploid d. are somatic
Biology 4361 Developmental Biology Name: Key Exam 1 ID#: October 11, 2005 Multiple choice (one point each) 1. Primordial germ cells a. are immortal b. produce polar bodies c. are haploid d. are somatic
CNS Developmental. Anke van Eekelen, PhD. Telethon Institute for Child Health Research
 CNS Developmental Anke van Eekelen, PhD Telethon Institute for Child Health Research (Some slides are modified versions of Prof. Alan Harvey s Neuroscience lecture at ANHB and Dr. Joanne Britto s Dev Neuroscience
CNS Developmental Anke van Eekelen, PhD Telethon Institute for Child Health Research (Some slides are modified versions of Prof. Alan Harvey s Neuroscience lecture at ANHB and Dr. Joanne Britto s Dev Neuroscience
Diseases of the skeleton
 Diseases of the skeleton Flexibility Protection of vital organs Strength Skeletal defects impact human health Developmental diseases Degenerative diseases Fins regenerate and grow rapidly following amputation
Diseases of the skeleton Flexibility Protection of vital organs Strength Skeletal defects impact human health Developmental diseases Degenerative diseases Fins regenerate and grow rapidly following amputation
Problem Set 5 KEY
 2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
2006 7.012 Problem Set 5 KEY ** Due before 5 PM on THURSDAY, November 9, 2006. ** Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. You are studying the development
EMBO REPORT SUPPLEMENTARY SECTION. Quantitation of mitotic cells after perturbation of Notch signalling.
 EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
EMBO REPORT SUPPLEMENTARY SECTION Quantitation of mitotic cells after perturbation of Notch signalling. Notch activation suppresses the cell cycle indistinguishably both within and outside the neural plate
Serotonin has early, cilia-independent roles in Xenopus left-right patterning
 Disease Models & Mechanisms 6, 261-268 (2013) doi:10.1242/dmm.010256 RESEARCH REPORT Serotonin has early, cilia-independent roles in Xenopus left-right patterning Laura N. Vandenberg 1, Joan M. Lemire
Disease Models & Mechanisms 6, 261-268 (2013) doi:10.1242/dmm.010256 RESEARCH REPORT Serotonin has early, cilia-independent roles in Xenopus left-right patterning Laura N. Vandenberg 1, Joan M. Lemire
Cellular pathophysiology of cystic kidney disease: insight into future therapies
 Int. J. Dev. Biol. 43: 457-461 (1999) Cellular pathophysiology of cystic kidney desease 457 Cellular pathophysiology of cystic kidney disease: insight into future therapies ELLIS D. AVNER*, RICHARD P.
Int. J. Dev. Biol. 43: 457-461 (1999) Cellular pathophysiology of cystic kidney desease 457 Cellular pathophysiology of cystic kidney disease: insight into future therapies ELLIS D. AVNER*, RICHARD P.
Positional signalling and the development of the humerus in the chick limb bud
 Development 100, 333-338 (1987) Printed in Great Britain The Company of Biologists Limited 1987 333 Positional signalling and the development of the humerus in the chick limb bud L. WOLPERT and AMATA HORNBRUCH
Development 100, 333-338 (1987) Printed in Great Britain The Company of Biologists Limited 1987 333 Positional signalling and the development of the humerus in the chick limb bud L. WOLPERT and AMATA HORNBRUCH
Organogenesis Part 2. V. Lateral Plate Mesoderm VI. Endoderm VII. Development of the Tetrapod Limb VIII. Sex Determination. V. Lateral Plate Mesoderm
 Organogenesis Part 2 V. Lateral Plate Mesoderm VI. Endoderm VII. Development of the Tetrapod Limb VIII. Sex Determination V. Lateral Plate Mesoderm chordamesoderm paraxial mesoderm intermediate mesoderm
Organogenesis Part 2 V. Lateral Plate Mesoderm VI. Endoderm VII. Development of the Tetrapod Limb VIII. Sex Determination V. Lateral Plate Mesoderm chordamesoderm paraxial mesoderm intermediate mesoderm
Neurodevelopment II Structure Formation. Reading: BCP Chapter 23
 Neurodevelopment II Structure Formation Reading: BCP Chapter 23 Phases of Development Ovum + Sperm = Zygote Cell division (multiplication) Neurogenesis Induction of the neural plate Neural proliferation
Neurodevelopment II Structure Formation Reading: BCP Chapter 23 Phases of Development Ovum + Sperm = Zygote Cell division (multiplication) Neurogenesis Induction of the neural plate Neural proliferation
Tubulointerstitial Renal Disease. Anna Vinnikova, MD Division of Nephrology
 Tubulointerstitial Renal Disease Anna Vinnikova, MD Division of Nephrology Part I: Cystic Renal Disease www.pathguy.com Simple cysts Simple cysts May be multiple Usually 1 5cm, may be bigger Translucent,
Tubulointerstitial Renal Disease Anna Vinnikova, MD Division of Nephrology Part I: Cystic Renal Disease www.pathguy.com Simple cysts Simple cysts May be multiple Usually 1 5cm, may be bigger Translucent,
Supporting Information
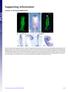 Supporting Information Creuzet 10.1073/pnas.0906072106 Fig. S1. Migrating CNC cells express Bmp inhibitors. (A) HNK1 immunolabeling of migrating CNC cells at 7 ss. (B) At this stage, Fgf8 is strongly expressed
Supporting Information Creuzet 10.1073/pnas.0906072106 Fig. S1. Migrating CNC cells express Bmp inhibitors. (A) HNK1 immunolabeling of migrating CNC cells at 7 ss. (B) At this stage, Fgf8 is strongly expressed
RECONSTITUTION OF METACHRONAL WAVES IN CILIATED CORTICAL SHEETS OF PARAMECIUM
 J. exp. Biol. 192, 73 81 (1994) Printed in Great Britain The Company of Biologists Limited 1994 73 RECONSTITUTION OF METACHRONAL WAVES IN CILIATED CORTICAL SHEETS OF PARAMECIUM II. ASYMMETRY OF THE CILIARY
J. exp. Biol. 192, 73 81 (1994) Printed in Great Britain The Company of Biologists Limited 1994 73 RECONSTITUTION OF METACHRONAL WAVES IN CILIATED CORTICAL SHEETS OF PARAMECIUM II. ASYMMETRY OF THE CILIARY
Polaris, a Protein Involved in Left-Right Axis Patterning, Localizes to Basal Bodies and Cilia
 Molecular Biology of the Cell Vol. 12, 589 599, March 2001 Polaris, a Protein Involved in Left-Right Axis Patterning, Localizes to Basal Bodies and Cilia Patrick D. Taulman,* Courtney J. Haycraft,* Daniel
Molecular Biology of the Cell Vol. 12, 589 599, March 2001 Polaris, a Protein Involved in Left-Right Axis Patterning, Localizes to Basal Bodies and Cilia Patrick D. Taulman,* Courtney J. Haycraft,* Daniel
Disease Models & Mechanisms DMM Accepted manuscript
 First posted online on 16 August 2012 as 10.1242/dmm.010256 2012. Published by The Company of Biologists Ltd. This Access is an Open the Access most article recent distributed version under at the http://dmm.biologists.org/lookup/doi/10.1242/dmm.010256
First posted online on 16 August 2012 as 10.1242/dmm.010256 2012. Published by The Company of Biologists Ltd. This Access is an Open the Access most article recent distributed version under at the http://dmm.biologists.org/lookup/doi/10.1242/dmm.010256
Signaling Vascular Morphogenesis and Maintenance
 Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Tetrapod Limb Development
 IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
IBS 8102 Cell, Molecular and Developmental Biology Tetrapod Limb Development February 11, 2008 Tetrapod Limbs Merlin D. Tuttle Vicki Lockard and Paul Barry Father Alejandro Sanchez Anne Fischer Limb Patterning
Patterning the Embryo
 Patterning the Embryo Anteroposterior axis Regional Identity in the Vertebrate Neural Tube Fig. 2.2 1 Brain and Segmental Ganglia in Drosophila Fig. 2.1 Genes that create positional and segment identity
Patterning the Embryo Anteroposterior axis Regional Identity in the Vertebrate Neural Tube Fig. 2.2 1 Brain and Segmental Ganglia in Drosophila Fig. 2.1 Genes that create positional and segment identity
Embryonic origins of spleen asymmetry
 Development 127, 167-175 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV6382 167 Embryonic origins of spleen asymmetry Kristin D. Patterson, Thomas A. Drysdale* and Paul A. Krieg
Development 127, 167-175 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV6382 167 Embryonic origins of spleen asymmetry Kristin D. Patterson, Thomas A. Drysdale* and Paul A. Krieg
Tissues. tissue = many cells w/ same structure and function. cell shape aids its function tissue shape aids its function
 Tissues tissue = many cells w/ same structure and function cell shape aids its function tissue shape aids its function Histology = study of tissues 4 types of tissues Epithelial coverings contact openings
Tissues tissue = many cells w/ same structure and function cell shape aids its function tissue shape aids its function Histology = study of tissues 4 types of tissues Epithelial coverings contact openings
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following are synthesized along various sites of the endoplasmic reticulum
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following are synthesized along various sites of the endoplasmic reticulum
Situs inversus. Dr praveena pulmonology- final year post graduate
 Situs inversus Dr praveena pulmonology- final year post graduate Definiton History Types Cause Clinical features Diagnosis Treatment Definition The term situs inversus is a short form of the latin phrase
Situs inversus Dr praveena pulmonology- final year post graduate Definiton History Types Cause Clinical features Diagnosis Treatment Definition The term situs inversus is a short form of the latin phrase
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature19102 Supplementary Discussion Benzothiazepine Binding in Ca V Ab Diltiazem and other benzothiazepines inhibit Ca V 1.2 channels in a frequency-dependent manner consistent with pore block
doi:10.1038/nature19102 Supplementary Discussion Benzothiazepine Binding in Ca V Ab Diltiazem and other benzothiazepines inhibit Ca V 1.2 channels in a frequency-dependent manner consistent with pore block
Branching morphogenesis of the lung: new molecular insights into an old problem
 86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
86 Review TRENDS in Cell Biology Vol.13 No.2 February 2003 Branching morphogenesis of the lung: new molecular insights into an old problem Pao-Tien Chuang 1 and Andrew P. McMahon 2 1 Cardiovascular Research
Skeletal Muscle Contraction 4/11/2018 Dr. Hiwa Shafiq
 Skeletal Muscle Contraction 4/11/2018 Dr. Hiwa Shafiq Skeletal Muscle Fiber About 40 per cent of the body is skeletal muscle, and 10 per cent is smooth and cardiac muscle. Skeletal muscles are composed
Skeletal Muscle Contraction 4/11/2018 Dr. Hiwa Shafiq Skeletal Muscle Fiber About 40 per cent of the body is skeletal muscle, and 10 per cent is smooth and cardiac muscle. Skeletal muscles are composed
Option A: Neurobiology & Behavior HL BIOLOGY 2 ND EDITION DAMON, MCGONEGAL, TOSTO, AND
 Option A: Neurobiology & Behavior A1: NEURAL DEVELOPMENT USE THE INFO IN THE PRESENTATION TO COMPLETE A1 NOTES GUIDE INFORMATION TAKEN FROM: HL BIOLOGY 2 ND EDITION DAMON, MCGONEGAL, TOSTO, AND WARD BIOLOGY
Option A: Neurobiology & Behavior A1: NEURAL DEVELOPMENT USE THE INFO IN THE PRESENTATION TO COMPLETE A1 NOTES GUIDE INFORMATION TAKEN FROM: HL BIOLOGY 2 ND EDITION DAMON, MCGONEGAL, TOSTO, AND WARD BIOLOGY
Name: Answer Key. Question 1.
 2007 7.013 Problem Set 6 Due before 5 PM on FRIDAY, April 27, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. Question 1. 1a. This is a diagram showing changes
2007 7.013 Problem Set 6 Due before 5 PM on FRIDAY, April 27, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. Question 1. 1a. This is a diagram showing changes
* * A3027. A4623 e A3507 A3507 A3507
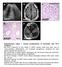 a c L A327 d e A37 A37 A37 Supplementary Figure 1. Clinical manifestations of individuals with mutations. (a) Renal ultrasound of right kidney in A327 reveals small renal cysts, loss of corticomedullary
a c L A327 d e A37 A37 A37 Supplementary Figure 1. Clinical manifestations of individuals with mutations. (a) Renal ultrasound of right kidney in A327 reveals small renal cysts, loss of corticomedullary
Mutation of the Mouse Hepatocyte Nuclear Factor/Forkhead Homologue 4 Gene Results in an Absence of Cilia and Random Left-Right Asymmetry
 Rapid Publication Mutation of the Mouse Hepatocyte Nuclear Factor/Forkhead Homologue 4 Gene Results in an Absence of Cilia and Random Left-Right Asymmetry Jianchun Chen, Heather J. Knowles, Jennifer L.
Rapid Publication Mutation of the Mouse Hepatocyte Nuclear Factor/Forkhead Homologue 4 Gene Results in an Absence of Cilia and Random Left-Right Asymmetry Jianchun Chen, Heather J. Knowles, Jennifer L.
BIOLOGY. Cell Communication CAMPBELL. Reece Urry Cain Wasserman Minorsky Jackson. Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick
 CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson 11 Cell Communication Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick Cellular Messaging Cells can signal to
CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson 11 Cell Communication Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick Cellular Messaging Cells can signal to
Chapter 11 Intercellular Communication and Tissue Architecture
 Part III Organization of Cell Populations Chapter 11 Multicellular organisms such as the human body consist of various tissues such as epithelial tissues, bones and nerves, and organs such as heart and
Part III Organization of Cell Populations Chapter 11 Multicellular organisms such as the human body consist of various tissues such as epithelial tissues, bones and nerves, and organs such as heart and
Asymmetries in H /K -ATPase and Cell Membrane Potentials Comprise a Very Early Step in Left-Right Patterning
 Cell, Vol. 111, 77 89, October 4, 2002, Copyright 2002 by Cell Press Asymmetries in H /K -ATPase and Cell Membrane Potentials Comprise a Very Early Step in Left-Right Patterning Michael Levin, 1,2 Thorleif
Cell, Vol. 111, 77 89, October 4, 2002, Copyright 2002 by Cell Press Asymmetries in H /K -ATPase and Cell Membrane Potentials Comprise a Very Early Step in Left-Right Patterning Michael Levin, 1,2 Thorleif
Developmental Biology
 Developmental Biology 367 (2012) 15 24 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Defective Nodal and Cerl2
Developmental Biology 367 (2012) 15 24 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Defective Nodal and Cerl2
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
Segmentation. Serial Homology. Hox genes
 Segmentation Hox genes Serial Homology William Bateson 1861-1926 Homeosis: a variation in which something has been changed into the likeness of something else Calvin Bridges at Columbia bithorax 1923 1
Segmentation Hox genes Serial Homology William Bateson 1861-1926 Homeosis: a variation in which something has been changed into the likeness of something else Calvin Bridges at Columbia bithorax 1923 1
University of Chicago 2012-Present Postdoctoral Research Associate Dept. of Organismal Biology and Anatomy,
 Tetsuya Nakamura, Ph.D. Organismal Biology and Anatomy, University of Chicago Culver Hall 108, 1027 E, 57 th Street Chicago, IL 60637 (773) 834-4774 tetsuya@uchicago.edu http://tetsuyanakamura.work Research
Tetsuya Nakamura, Ph.D. Organismal Biology and Anatomy, University of Chicago Culver Hall 108, 1027 E, 57 th Street Chicago, IL 60637 (773) 834-4774 tetsuya@uchicago.edu http://tetsuyanakamura.work Research
Homeostasis. Endocrine System Nervous System
 Homeostasis Endocrine System Nervous System 2004-2005 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation
Homeostasis Endocrine System Nervous System 2004-2005 Regulation Why are hormones needed? chemical messages from one body part to another communication needed to coordinate whole body homeostasis & regulation
Left/Right Patterning Signals and the Independent Regulation of Different Aspects of Situs in the Chick Embryo
 DEVELOPMENTAL BIOLOGY 189, 57 67 (1997) ARTICLE NO. DB978662 Left/Right Patterning Signals and the Independent Regulation of Different Aspects of Situs in the Chick Embryo Michael Levin,*,1 Sylvia Pagan,*
DEVELOPMENTAL BIOLOGY 189, 57 67 (1997) ARTICLE NO. DB978662 Left/Right Patterning Signals and the Independent Regulation of Different Aspects of Situs in the Chick Embryo Michael Levin,*,1 Sylvia Pagan,*
Cell Birth and Death. Chapter Three
 Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
Cell Birth and Death Chapter Three Neurogenesis All neurons and glial cells begin in the neural tube Differentiated into neurons rather than ectoderm based on factors we have already discussed If these
Edinburgh Research Explorer
 Edinburgh Research Explorer The chicken left right organizer has nonmotile cilia which are lost in a stage-dependent manner in the talpid3 ciliopathy Citation for published version: Stephen, LA, Johnson,
Edinburgh Research Explorer The chicken left right organizer has nonmotile cilia which are lost in a stage-dependent manner in the talpid3 ciliopathy Citation for published version: Stephen, LA, Johnson,
Heart Development. Origins of congenital heart defects Properties of cardiac progenitor cells. Robert G. Kelly
 ESC CBCS Summer School on Cardiovascular Sciences Heart Development 19th June 2013 Origins of congenital heart defects Properties of cardiac progenitor cells Robert G. Kelly Animal models of heart development
ESC CBCS Summer School on Cardiovascular Sciences Heart Development 19th June 2013 Origins of congenital heart defects Properties of cardiac progenitor cells Robert G. Kelly Animal models of heart development
Polycystin-1 Expression in Fetal, Adult and Autosomal Dominant Polycystic Kidney
 J Korean Med Sci 2006; 21: 425-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Polycystin-1 Expression in Fetal, Adult and Autosomal Dominant Polycystic Kidney The mutation of the PKD1
J Korean Med Sci 2006; 21: 425-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Polycystin-1 Expression in Fetal, Adult and Autosomal Dominant Polycystic Kidney The mutation of the PKD1
The mouse homeobox gene Noto regulates node morphogenesis, notochordal ciliogenesis, and left right patterning
 The mouse homeobox gene Noto regulates node morphogenesis, notochordal ciliogenesis, and left right patterning Anja Beckers*, Leonie Alten*, Christoph Viebahn, Philipp Andre, and Achim Gossler* *Institute
The mouse homeobox gene Noto regulates node morphogenesis, notochordal ciliogenesis, and left right patterning Anja Beckers*, Leonie Alten*, Christoph Viebahn, Philipp Andre, and Achim Gossler* *Institute
Animal Tissue Culture SQG 3242 Biology of Cultured Cells. Dr. Siti Pauliena Mohd Bohari
 Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Sdc2 and Tbx16 regulate Fgf2-dependent epithelial cell morphogenesis in the ciliated organ of asymmetry
 4102 140, 4102-4109 (2013) doi:10.1242/dev.096933 2013. Published by The Company of Biologists Ltd Sdc2 and Tbx16 regulate Fgf2-dependent epithelial cell morphogenesis in the ciliated organ of asymmetry
4102 140, 4102-4109 (2013) doi:10.1242/dev.096933 2013. Published by The Company of Biologists Ltd Sdc2 and Tbx16 regulate Fgf2-dependent epithelial cell morphogenesis in the ciliated organ of asymmetry
ANATOMY & PHYSIOLOGY - CLUTCH CH. 6 - CELL COMMUNICATION.
 !! www.clutchprep.com CONCEPT: CELL-TO-CELL CONNECTIONS AND SIGNALING Gap and Tight Junctions: Adjacent cells communicate and hold on to each other via junctions. Two important kinds: Gap Junctions are
!! www.clutchprep.com CONCEPT: CELL-TO-CELL CONNECTIONS AND SIGNALING Gap and Tight Junctions: Adjacent cells communicate and hold on to each other via junctions. Two important kinds: Gap Junctions are
Basics of skeletal muscle electrophysiology. Tóth András, PhD
 Basics of skeletal muscle electrophysiology Tóth András, PhD Topics Structure Contraction and relaxation Activation Excitation-contraction coupling Action potential Ion channels* Calcium homeostasis Structure
Basics of skeletal muscle electrophysiology Tóth András, PhD Topics Structure Contraction and relaxation Activation Excitation-contraction coupling Action potential Ion channels* Calcium homeostasis Structure
Phys 173 / BGGN 266. LPA Induced Cl - Oscillations in Xenopus Oocytes. Nini Huynh David Marciano Chisa Suzuki
 Phys 173 / BGGN 266 LPA Induced Cl - Oscillations in Xenopus Oocytes Nini Huynh David Marciano Chisa Suzuki If only we hadn t poked these oocytes, how cute would it be! INTRODUCTION Electrophysiology in
Phys 173 / BGGN 266 LPA Induced Cl - Oscillations in Xenopus Oocytes Nini Huynh David Marciano Chisa Suzuki If only we hadn t poked these oocytes, how cute would it be! INTRODUCTION Electrophysiology in
Mechanisms of left-right asymmetry and patterning: driver, mediator and responder Hiroshi Hamada 1 and Patrick P. L. Tam 2 *
 Published: 01 December 2014 2014 Faculty of 1000 Ltd Mechanisms of left-right asymmetry and patterning: driver, mediator and responder Hiroshi Hamada 1 and Patrick P. L. Tam 2 * Addresses: 1 Developmental
Published: 01 December 2014 2014 Faculty of 1000 Ltd Mechanisms of left-right asymmetry and patterning: driver, mediator and responder Hiroshi Hamada 1 and Patrick P. L. Tam 2 * Addresses: 1 Developmental
