LAL TESTED 1 MEA TESTED 1 HSSA TESTED 1,2
|
|
|
- Jody Banks
- 5 years ago
- Views:
Transcription
1
2 MEA TESTED 1 The finished consumables are tested by an independent laboratory using a 1-cell stage mouse embryo toxicity test. Our pass criteria is 80% blastocyst after 120h LAL TESTED 1 Each sterility batch of finished product is tested for bacterial endotoxins. Our pass criteria is < 20Eu per device HSSA TESTED 1,2 Finished injection pipettes undergo and pass a sperm motility bio assay. To pass the HSSA test, the motility of sperm must be 70% after 24 hours of sample preparation 1 Refer to Sterility and Biocompatibility Testing on the website 2 Applicable to injection pipettes only
3 I appreciate the sharpness of the injection pipette and the comfort of the holding pipette during microinjection of oocytes. Maria Beatrice Dal Canto, Centro di Medicina Riproduzione BioGenesi, Italy RI PIPETTES Precise, Smooth, Consistent Micropipettes STERILISED 1 Sterilised by an approved gamma irradiation facility meeting ISO standards to a sterility assurance level (SAL) of 10-6 PRECISE Super sharp, non-sticky, with consistent geometries. Each pipette is individually inspected 5 times during manufacture EFFECTIVE Wide range of sizes and angles designed for all your assisted reproduction applications
4 RI PIPETTES OVERVIEW RI Pipettes are available in a range of configuration options to suit procedures, user preference and micromanipulator set-up 3. Configurable options include inner diameter (ID), outer diameter (OD), angle of bend and length of toe (tip to bend) 4. Please visit pipettecodes to easily select the correct pipette part number for your use 3. PRACTICAL PACKAGING Our micropipettes are packed into a high quality Twista-Pak. The Twista-Pak provides protection from tip damage and operates like a lipstick, where the simple twist action allows easy access and ensures neither end of the pipette is touched during removal. The RI micropipettes have a shelf life of three years as standard. 3 Not all configurations are held in stock - please check with your distributor for information on stock or lead times μm toe length is only available in thin glass. INJECTION PIPETTES RI ICSI-Plus Injection Pipettes can be used to immobilise, then inject spermatozoa into the cytoplasm of an oocyte during ICSI and Intracytoplasmic Morphologicallyselected Sperm Injection (IMSI). Our ICSI-Plus Injection Pipettes are designed for the smoothest control of sperm injection and cytoplasm suction, and to minimise damage to the oocyte. Multiple sperm can be loaded and injected. Injection pipettes from RI are available with a spike (SI) or without a spike (NI). For human ART, the larger 8µm ID injection pipette, is ideal for collecting sperm from a testicular tissue sample prior to ICSI. HOLDING PIPETTES These pipettes are designed for anchoring the oocyte or embryo during ICSI or other micromanipulation procedures. A smooth fire polished tip cradles the cells gently and securely. Applicable indications for use are subject to the regulations of the country into which the device is sold. Availability of a device for clinical use is dependent on the regulatory approval status of that device within the country the device is intended to be sold into. SPIKED INJECTION (SI) ID 4/5/8µm Toe Length (µm): 750, NON-SPIKED INJECTION (NI) ID 5µm Toe Length (µm): 750, HOLDING (IH) ID 15µm Toe Length (µm): 750 LARGE HOLDING (LH) ID 25/32µm Toe Length (µm): 750
5 FINISHED PRODUCT MEA TESTED 1 FINISHED PRODUCT LAL TESTED 1 FINISHED PRODUCT HSSA TESTED 1,2 BIOPSY PIPETTES RI biopsy pipettes (B) are used to remove tissue from embryos or oocytes for Pre-implantation Genetic Diagnosis (PGD). RI offer these pipettes in three profiles - flat, non-spiked or spiked. Available in a range of ID sizes suitable for a variety of biopsies including polar body (usually 14µm ID), blastomere and trophectoderm biopsies (usually between 20-40µm ID). Each biopsy pipette is fire polished to minimise damage to the cell membrane. MECHANICAL DISSECTION AND ASSISTED HATCHING PIPETTES RI mechanical dissection pipettes (MD) are used to mechanically cut an opening in the zona pellucida of an oocyte or embryo to enable assisted hatching and biopsy. The pipette is designed with a sharp tip or a strong taper section for easy control and precise manipulation. RI assisted hatching pipettes (AH) are used to direct acidic solution (acid tyrodes) onto the zona pellucida to thin the zona or create a hole to enable the embryo to hatch. BIOPSY (B) Spiked (SB) ID 14µm Non-Spiked (NB) ID 14µm Flat (FB) ID 14/20/25/ 30/35/40µm Toe Length (µm): 750 MECHANICAL DISSECTION (MD) Toe Length (µm): 750 ASSISTED HATCHING (AH) ID 10µm Toe Length (µm): 750
6 ORDER CODES ORDER CODES
7 RI Pipettes are available in a range of options, below is a table showing these options with their product codes. Please replace the XX in the code with the required bend angle to create the final code to be used for ordering. The bend options are 0, 15, 20, 25, 30, 35, 40 or 45. Injection Pipettes: Standard glass thickness is 1µm unless stated as Thin Glass (T code) which is 0.9µm. Standard toe length is 750µm, long toe length is 1000µm. Customisations are available upon request. To easily select the correct pipette part number for your use visit pipettecodes INJECTION PIPETTES BIOPSY PIPETTES PIPETTES ICSI Spiked Injection THIN GLASS INTERNAL DIAMETER (µm) LONG TOE CODE (FOR A BOX OF 20) 7-71-T4SIXX/ T5SIXX/ T8SIXX/ T4SIXXL/ T5SIXXL/20 PIPETTES Biopsy INTERNAL DIAMETER - ID (µm) PROFILE CODE (FOR A BOX OF 20) SPIKED NON-SPIKED FLAT SBXX/ NBXX/ FBXX/ FBXX/ FBXX/ FBXX/ FBXX/ FBXX/20 HOLDING PIPETTES MECHANICAL DISSECTION & ASSISTED HATCHING PIPETTES 7-71-T8SIXXL/20 PIPETTES INTERNAL DIAMETER (µm) CODE (FOR A BOX OF 20) PIPETTES INTERNAL DIAMETER (µm) CODE (FOR A BOX OF 20) ICSI Non- Spiked Injection 7-71-T5NIXX/ T5NIXXL/20 ICSI Holding IHXX/20 Large Holding LHXX/ HXX/20 Mechanical Dissection Assisted Hatching MDXX/ AHXX/20
8 QUALITY ASSURANCE STERILITY AND BIOCOMPATIBILITY TESTING ALL RI MEDICAL DEVICES CARRY THE CE MARK The CE mark shows that RI s products comply with the Essential Safety Requirements of the European Medical Device Directive (93/42/EEC) as amended. Many of our medical devices also have US FDA market clearance, Health Canada medical device licenses and certification in Taiwan and China. We are continuously working to expand our registration with the relevant medical device authorities across the world. QUALITY MANAGEMENT SYSTEM FOR MEDICAL DEVICES - EN ISO 13485:2012 At RI quality permeates all that we do. All RI products are designed, developed and manufactured within an EN ISO 13485:2012 environment. All materials and packaging used within the manufacturing process of RI medical devices are comprehensively tracked and logged to ensure full traceability. We hold ISO 9001:2008 and EN ISO 13485:2012 certification (including CMDCAS, KFDA and TFDA GMP). RI is registered with the US FDA for the supply of medical devices. Continual evaluation of our products has led to the creation of the finest range of devices on the market that clinics aspire to own and use. CLEANROOM MANUFACTURING FACILITIES RI consumables are manufactured 3 and packaged in an ISO certified cleanroom. Our highly trained manufacturing personnel inspect each individual unit at every step of the production process to ensure only the highest quality devices reach your laboratory. For example, our quality control protocols require that micropipettes are examined five times before production is completed. STERILISATION Where applicable, products are sterilised by an approved gamma irradiation facility meeting ISO standards to a sterility assurance level (SAL) of The shelf life of our pouch packed products is three years and five years for our blister packed products. PREMIER STANDARD BATCH TESTING RI s range of consumables must pass stringent testing and inspection criteria to ensure that you receive the most consistent and high quality products. RI choose to test each finished sterility batch of consumables as we consider this is a true indication of product toxicity. Many manufacturers test for toxicity at the raw material stage only which will not reflect the effect of the manufacturing process on toxicity. MOUSE EMBRYO ASSAY (MEA) FOR EMBRYO TOXICITY Every sterility batch of finished consumables is tested by an independent laboratory using a 1-cell stage mouse embryo toxicity test. Embryos are also assessed at critical development points during the MEA to ensure maximum test sensitivity. Our pass criteria is 80% blastocyst after 120h. RI s range of consumables must pass stringent testing and inspection criteria to ensure that you receive the most consistent and high quality product.
9 FINISHED PRODUCT MEA TESTED 1 FINISHED PRODUCT LAL TESTED 2,3 FINISHED PRODUCT HSSA TESTED 4,5 LIMULUS AMEBOCYTE LYSATE (LAL) FOR ENDOTOXINS Each sterility batch of finished product is tested for bacterial endotoxins. Our pass criteria is <20Eu per device. HUMAN SPERM SURVIVAL ASSAY (HSSA) Each sterility batch of finished injection pipettes within the RI Pipette range undergoes and passes a sperm motility bio assay. To pass the HSSA test, the motility of sperm must be 70% after 24 hours of sample preparation. 1 Refer to Mouse Embryo Assay (MEA) for Embryo Toxicity section 2 Refer to Limulus Amoebocyte Lysate (LAL) for Endotoxins section 3 Excludes Fertisafe Plus manufacture 4 Applicable to injection pipettes only 5 Refer to Human Sperm Survival Assay (HSSA) section For further information about our products visit:
10 Research Instruments Ltd, Bickland Industrial Park, Falmouth, Cornwall, TR11 4TA, UK tel: +44 (0) fax: +44 (0) visit: MKTG-BR /06/2016. All information correct at time of print. Specifications are subject to change without notice or obligation on the part of the manufacturer.
I can confidently use the EZ-Range for all embryological handling procedures and rest assured that the eggs/ embryos are secure during movement.
 1 I can confidently use the EZ-Range for all embryological handling procedures and rest assured that the eggs/ embryos are secure during movement. Julia Paget, Complete Fertility, Southampton, UK A RANGE
1 I can confidently use the EZ-Range for all embryological handling procedures and rest assured that the eggs/ embryos are secure during movement. Julia Paget, Complete Fertility, Southampton, UK A RANGE
Sunlight Medical. Assisted Reproduction Microtools.
 Sunlight Medical Assisted Reproduction Microtools www.sunlight-medical.com unlight Medical Inc (SLM) manufactures and supplies a complete line of micropipettes for in vitro fertilization S (IVF) procedures
Sunlight Medical Assisted Reproduction Microtools www.sunlight-medical.com unlight Medical Inc (SLM) manufactures and supplies a complete line of micropipettes for in vitro fertilization S (IVF) procedures
Precision and control.
 Precision and control. MICROMANIPULATION PIPETTES MEDICAL Precise Cook Medical micromanipulation tools are precision instruments manufactured to exacting quality standards. Specialized Pipettes are available
Precision and control. MICROMANIPULATION PIPETTES MEDICAL Precise Cook Medical micromanipulation tools are precision instruments manufactured to exacting quality standards. Specialized Pipettes are available
Sunlight Medical. Assisted Reproduction Microtools.
 Sunlight Medical Assisted Reproduction Microtools www.sunlight-medical.com unlight Medical Inc (SLM) manufactures and supplies a complete line of micropipettes for in vitro fertilization S (IVF) procedures
Sunlight Medical Assisted Reproduction Microtools www.sunlight-medical.com unlight Medical Inc (SLM) manufactures and supplies a complete line of micropipettes for in vitro fertilization S (IVF) procedures
Our Leading Range of Products. Pipettes designed for micromanipulation, denuding and handling
 Our Leading Range of Products Pipettes designed for micromanipulation, denuding and handling OOCYTE RETRIEVAL ANDROLOGY & IUI FERTILIZATION CULTURE PGD & PGS CRYOPRESERVATION EMBRYO TRANSFER The Most Comprehensive
Our Leading Range of Products Pipettes designed for micromanipulation, denuding and handling OOCYTE RETRIEVAL ANDROLOGY & IUI FERTILIZATION CULTURE PGD & PGS CRYOPRESERVATION EMBRYO TRANSFER The Most Comprehensive
micromanipulation by vitrolife A complete solution for ICSI procedures.
 micromanipulation by vitrolife A complete solution for ICSI procedures. 1 tested, tried and trusted Vitrolife micromanipulation products have been used from the beginning. Used worldwide Vitrolife is the
micromanipulation by vitrolife A complete solution for ICSI procedures. 1 tested, tried and trusted Vitrolife micromanipulation products have been used from the beginning. Used worldwide Vitrolife is the
FERTISAFE PLUS US PL AFE TIS FER
 FERTISAFE PLUS FERTISAFE PLUS FERTISAFE PLUS Broad Spectrum Antimicrobial Disinfectant Fertisafe Plus is a long lasting, CE marked, ready to use broad spectrum antimicrobial disinfectant. Quaternary Ammonium
FERTISAFE PLUS FERTISAFE PLUS FERTISAFE PLUS Broad Spectrum Antimicrobial Disinfectant Fertisafe Plus is a long lasting, CE marked, ready to use broad spectrum antimicrobial disinfectant. Quaternary Ammonium
Content: About Seaforia. Introduction Seaforia Sperm Seaparation System Seaforia procedure Seaforia Disposable Kit Seaforia Lab Kit.
 TM Content: TM About Seaforia Introduction Seaforia Sperm Seaparation System Seaforia procedure Seaforia Disposable Kit Seaforia Lab Kit 4 6 8 9 Product Catalog Seaforia Disposable Kit Seaforia Lab Kit
TM Content: TM About Seaforia Introduction Seaforia Sperm Seaparation System Seaforia procedure Seaforia Disposable Kit Seaforia Lab Kit 4 6 8 9 Product Catalog Seaforia Disposable Kit Seaforia Lab Kit
Preimplantation genetic diagnosis: polar body and embryo biopsy
 Human Reproduction, Vol. 15, (Suppl. 4), pp. 69-75, 2000 Preimplantation genetic diagnosis: polar body and embryo biopsy Luca Gianaroli SISMER, Via Mazzini 12, 40138 Bologna, Italy Scientific Director
Human Reproduction, Vol. 15, (Suppl. 4), pp. 69-75, 2000 Preimplantation genetic diagnosis: polar body and embryo biopsy Luca Gianaroli SISMER, Via Mazzini 12, 40138 Bologna, Italy Scientific Director
Assisted Reproduction
 Assisted Reproduction Embryo Transfer, Follicle Aspiration, Insemination w w w. s o m a t e x. c o m Assisted Reproduction Top Made in Germany Quality SOMATEX presents a range of products attuned to the
Assisted Reproduction Embryo Transfer, Follicle Aspiration, Insemination w w w. s o m a t e x. c o m Assisted Reproduction Top Made in Germany Quality SOMATEX presents a range of products attuned to the
REASSURING LIBERATING SECURE
 SECURE Locks the patient s identity to the sperm, eggs and embryos at every stage of the treatment, so that they are continuously monitored REASSURING Gives you and your patients increased confidence in
SECURE Locks the patient s identity to the sperm, eggs and embryos at every stage of the treatment, so that they are continuously monitored REASSURING Gives you and your patients increased confidence in
PGD & PGS. Embryo Biopsy. The full solution for efficient and effective biopsy procedures
 PGD & PGS Embryo Biopsy The full solution for efficient and effective biopsy procedures QUALITY CONTROL OOCYTE RETRIEVAL ANDROLOGY & IUI FERTILIZATION CULTURE PGD & PGS CRYOPRESERVATION EMBRYO TRANSFER
PGD & PGS Embryo Biopsy The full solution for efficient and effective biopsy procedures QUALITY CONTROL OOCYTE RETRIEVAL ANDROLOGY & IUI FERTILIZATION CULTURE PGD & PGS CRYOPRESERVATION EMBRYO TRANSFER
Seaforia Sperm Separation System. Product Catalog
 Seaforia Sperm Separation System Product Catalog TM R Content: ABOUT SEAFORIA Introduction Seaforia Sperm Separation System Seaforia procedure Seaforia Lab Kit Seaforia Lab Kit Duo Seaforia Disposable
Seaforia Sperm Separation System Product Catalog TM R Content: ABOUT SEAFORIA Introduction Seaforia Sperm Separation System Seaforia procedure Seaforia Lab Kit Seaforia Lab Kit Duo Seaforia Disposable
Graft Delivery Devices
 Graft Delivery Devices About Graft Delivery Devices Nordson MEDICAL Nordson MEDICAL is your partner in the global life sciences market providing innovative components, devices and custom OEM solutions
Graft Delivery Devices About Graft Delivery Devices Nordson MEDICAL Nordson MEDICAL is your partner in the global life sciences market providing innovative components, devices and custom OEM solutions
FROM GAMETES TO BLASTOCYSTS. Simpler Processes, Less Stress, Better Results.
 FROM GAMETES TO BLASTOCYSTS Simpler Processes, Less Stress, Better Results. HELP AT EVERY STEP with simpler processes, less stress and better results Multipurpose Handling Medium RETRIEVE AND RINSE OOCYTES
FROM GAMETES TO BLASTOCYSTS Simpler Processes, Less Stress, Better Results. HELP AT EVERY STEP with simpler processes, less stress and better results Multipurpose Handling Medium RETRIEVE AND RINSE OOCYTES
Medidée Services SA. Nano-Tera.ch. 05 February 2015 part 4. Intro ISO GMP - GLP Pierre-Alain Sommer
 Nano-Tera.ch 05 February 2015 part 4 Intro ISO GMP - GLP Pierre-Alain Sommer Pierre-alain.sommer@medidee.com www.medidee.com Nano-Tera 2015 05.02.2015 Introduction to ISO 13485, cgmp s and GLP s Context
Nano-Tera.ch 05 February 2015 part 4 Intro ISO GMP - GLP Pierre-Alain Sommer Pierre-alain.sommer@medidee.com www.medidee.com Nano-Tera 2015 05.02.2015 Introduction to ISO 13485, cgmp s and GLP s Context
EC Certificate - Production Quality Assurance Directive 93/42/EEC on Medical Devices, Annex V
 Directive 93/42/EEC on Medical Devices, Annex V No. CE 80978 In respect of: The manufacture of sterile cryopresevation devices (Cryopette ) and sterile micropipettes/stripper tips intended for the preparation,
Directive 93/42/EEC on Medical Devices, Annex V No. CE 80978 In respect of: The manufacture of sterile cryopresevation devices (Cryopette ) and sterile micropipettes/stripper tips intended for the preparation,
LG MEDIA & GPS DISHWARE CODA AIR
 The Future of IVF Technologies! Always a Leader! global Based on Pure Science Over 10 Years of Published Clinical Results and Consistency! The leading scientifically based and clinically proven one solution
The Future of IVF Technologies! Always a Leader! global Based on Pure Science Over 10 Years of Published Clinical Results and Consistency! The leading scientifically based and clinically proven one solution
product catalogue Quality Results for life
 product catalogue ET CATHETERS OPU NEEDLES Quality Results for life IUI CATHETERS OOCYTE RETRIEVAL NEEDLES NEEDLES oocyte retrieval needles Manufactured from steel of excellent quality, its triple cut
product catalogue ET CATHETERS OPU NEEDLES Quality Results for life IUI CATHETERS OOCYTE RETRIEVAL NEEDLES NEEDLES oocyte retrieval needles Manufactured from steel of excellent quality, its triple cut
Product: Spinal Needle. Zhejiang Runqiang Medical Instruments Co., Ltd
 Product: Spinal Needle Zhejiang Runqiang Medical Instruments Co., Ltd Parameter Method Specification Product Description - The product spinal needle is made up of three parts: - Spinal needle (needle tube
Product: Spinal Needle Zhejiang Runqiang Medical Instruments Co., Ltd Parameter Method Specification Product Description - The product spinal needle is made up of three parts: - Spinal needle (needle tube
Draft Guidance for Industry and FDA Staff
 Draft Guidance for Industry and FDA Staff Submission and Review of Sterility Information in Premarket Notification (510(k)) Submissions for Devices Labeled as Sterile DRAFT GUIDANCE This guidance document
Draft Guidance for Industry and FDA Staff Submission and Review of Sterility Information in Premarket Notification (510(k)) Submissions for Devices Labeled as Sterile DRAFT GUIDANCE This guidance document
NEW! Micromanipulator meets microscope. Eppendorf Cell Technology
 NEW! Micromanipulator meets microscope Eppendorf Cell Technology Eppendorf Systems... Intracytoplasmic sperm injection (ICSI) Intracytoplasmic sperm injection (ICSI), the injection of a single sperm into
NEW! Micromanipulator meets microscope Eppendorf Cell Technology Eppendorf Systems... Intracytoplasmic sperm injection (ICSI) Intracytoplasmic sperm injection (ICSI), the injection of a single sperm into
MEGA Assay. Modernizing quality control in IVF.
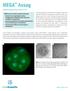 MEGA Assay Modernizing quality control in IVF MEGA assures product consistency through: Analysis of pre-implantation embryo development and health by gene expression in addition to morphology Increased
MEGA Assay Modernizing quality control in IVF MEGA assures product consistency through: Analysis of pre-implantation embryo development and health by gene expression in addition to morphology Increased
March 8, DxNow, Inc. Kevin Sly Senior Advisor to DxNow, Inc. 401 Professional Drive, Suite 130 Gaithersburg, Maryland
 March 8, 2018 Kevin Sly Senior Advisor to 401 Professional Drive, Suite 130 Gaithersburg, Maryland 20879-3429 Re: Trade/Device Name: ZyMot ICSI Sperm Separation Device, ZyMot Multi Sperm Separation Device
March 8, 2018 Kevin Sly Senior Advisor to 401 Professional Drive, Suite 130 Gaithersburg, Maryland 20879-3429 Re: Trade/Device Name: ZyMot ICSI Sperm Separation Device, ZyMot Multi Sperm Separation Device
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
I.C.E. Embryo Vitrification Kit
 I.C.E. Embryo Vitrification Kit Vitrification Media V1, V2, V3 ICE Embryo Vitrification Instructions For Use Testing and Cautions Innvative Cryo Enterprises LLC 317 Springfield Road Linden, New Jersey
I.C.E. Embryo Vitrification Kit Vitrification Media V1, V2, V3 ICE Embryo Vitrification Instructions For Use Testing and Cautions Innvative Cryo Enterprises LLC 317 Springfield Road Linden, New Jersey
Data Pack. KIMTECH PURE* G3 Sterile Latex Gloves
 KIMTECH PURE* G3 Sterile Latex Gloves Data Pack /* Trademarks of Kimberly-Clark Worldwide, Inc. or its affiliates. Marques déposées de Kimberly-Clark Worldwide, Inc. ou de ses filiales. KCWW 2011. K02306
KIMTECH PURE* G3 Sterile Latex Gloves Data Pack /* Trademarks of Kimberly-Clark Worldwide, Inc. or its affiliates. Marques déposées de Kimberly-Clark Worldwide, Inc. ou de ses filiales. KCWW 2011. K02306
FDA Compliant Patented & Patent Pending
 FDA Compliant Patented & Patent Pending ISO 13485:2003 ISO 9001:2000 Embryo Corral & Embryo GPS TM IVF Dishware Specially Designed & Tested for IVF No more droplet collapsing, running and mixing. The benefits
FDA Compliant Patented & Patent Pending ISO 13485:2003 ISO 9001:2000 Embryo Corral & Embryo GPS TM IVF Dishware Specially Designed & Tested for IVF No more droplet collapsing, running and mixing. The benefits
ASSISTED REPRODUCTIVE TECHNOLOGIES (ART)
 ASSISTED REPRODUCTIVE TECHNOLOGIES (ART) Dr. Herve Lucas, MD, PhD, Biologist, Andrologist Dr. Taher Elbarbary, MD Gynecologist-Obstetrician Geneva Foundation for Medical Education and research Definitions
ASSISTED REPRODUCTIVE TECHNOLOGIES (ART) Dr. Herve Lucas, MD, PhD, Biologist, Andrologist Dr. Taher Elbarbary, MD Gynecologist-Obstetrician Geneva Foundation for Medical Education and research Definitions
A high-level overview of the requirements of medical packaging standards
 Medical Packaging A high-level overview of the requirements of medical packaging standards Dec 2012 Thierry Wagner Regulatory Affairs Director Europe, Middle East and Africa DuPont Medical & Pharmaceutical
Medical Packaging A high-level overview of the requirements of medical packaging standards Dec 2012 Thierry Wagner Regulatory Affairs Director Europe, Middle East and Africa DuPont Medical & Pharmaceutical
» Guided Surgery. by MEDENTiKA « MEDENTIGUIDE SURGERY MANUAL MICROCONE. IPS Implant systems
 » Guided Surgery by MEDENTiKA «IPS Implant systems MEDENTIGUIDE MICROCONE SURGERY MANUAL » MedentiGuide «MedentiGuide drill sleeves support the surgeon in preparing the implant bed for MEDENTiKA implants.
» Guided Surgery by MEDENTiKA «IPS Implant systems MEDENTIGUIDE MICROCONE SURGERY MANUAL » MedentiGuide «MedentiGuide drill sleeves support the surgeon in preparing the implant bed for MEDENTiKA implants.
BONE SUBSTITUTION SPECIAL MEMBRANES SCREWS RIVETS
 BONE SUBSTITUTION SPECIAL MEMBRANES SCREWS RIVETS 1 Introduction About the company 2 Technology and quality 3 Applied raw materials 5 BIONIKA Titanium membrane meshes Titanium membranes Special membrane
BONE SUBSTITUTION SPECIAL MEMBRANES SCREWS RIVETS 1 Introduction About the company 2 Technology and quality 3 Applied raw materials 5 BIONIKA Titanium membrane meshes Titanium membranes Special membrane
Intracytoplasmic Sperm Injection (ICSI) with the Eppendorf micromanipulator TransferMan 4m
 APPLICATION NOTE No. 009 I June 2013 Intracytoplasmic Sperm Injection (ICSI) with the Eppendorf micromanipulator TransferMan 4m Verena Nordhoff, Centre of Reproductive Medicine and Andrology, University
APPLICATION NOTE No. 009 I June 2013 Intracytoplasmic Sperm Injection (ICSI) with the Eppendorf micromanipulator TransferMan 4m Verena Nordhoff, Centre of Reproductive Medicine and Andrology, University
Understanding eggs, sperm and embryos. Marta Jansa Perez Wolfson Fertility Centre
 Understanding eggs, sperm and embryos Marta Jansa Perez Wolfson Fertility Centre What does embryology involve? Aims of the embryology laboratory Creation of a large number of embryos and supporting their
Understanding eggs, sperm and embryos Marta Jansa Perez Wolfson Fertility Centre What does embryology involve? Aims of the embryology laboratory Creation of a large number of embryos and supporting their
» Guided Surgery. by MEDENTiKA « MEDENTIGUIDE SURGERY MANUAL QUATTROCONE QUATTROCONE30. IPS Implant systems
 » Guided Surgery by MEDENTiKA «IPS Implant systems MEDENTIGUIDE QUATTROCONE QUATTROCONE30 SURGERY MANUAL » MedentiGuide «MedentiGuide drill sleeves support the surgeon in preparing the implant bed for
» Guided Surgery by MEDENTiKA «IPS Implant systems MEDENTIGUIDE QUATTROCONE QUATTROCONE30 SURGERY MANUAL » MedentiGuide «MedentiGuide drill sleeves support the surgeon in preparing the implant bed for
ART TRAINING - DAILY SCHEDULE Trainee: Priyanka Sinha, MD Training Dates: September 3 - September 28, 2018
 WEEK DATE TIME FACULTY NAME PROGRAM Week 1: Module 5D Mon 9/03/2018 Tues 9/04/2018 Wed 9/05/2018 Thu 9/06/2018 Fri 9/07/2018 ART TRAINING - DAILY SCHEDULE Trainee: Priyanka Sinha, MD Training Dates: September
WEEK DATE TIME FACULTY NAME PROGRAM Week 1: Module 5D Mon 9/03/2018 Tues 9/04/2018 Wed 9/05/2018 Thu 9/06/2018 Fri 9/07/2018 ART TRAINING - DAILY SCHEDULE Trainee: Priyanka Sinha, MD Training Dates: September
Intracytoplasmic Sperm Injection (ICSI) with the Eppendorf TransferMan 4m and CellTram 4m
 APPLICATION NOTE No. 009 Intracytoplasmic Sperm Injection (ICSI) with the Eppendorf TransferMan 4m and CellTram 4m Verena Nordhoff, Centre of Reproductive Medicine and Andrology, University Hospital of
APPLICATION NOTE No. 009 Intracytoplasmic Sperm Injection (ICSI) with the Eppendorf TransferMan 4m and CellTram 4m Verena Nordhoff, Centre of Reproductive Medicine and Andrology, University Hospital of
High-end solution STEP-BY-STEP USER GUIDE
 High-end solution STEP-BY-STEP USER GUIDE System Contents REAL TIME Restoration Guided Conical drill Multi-Unit abutment, Conical connection Guided kit for conical connection implant procedure C1 & V3
High-end solution STEP-BY-STEP USER GUIDE System Contents REAL TIME Restoration Guided Conical drill Multi-Unit abutment, Conical connection Guided kit for conical connection implant procedure C1 & V3
Integra SURGICAL TECHNIQUE. MemoFix Super Elastic Nitinol Staple System. Super Elastic Nitinol Staple System
 Integra MemoFix Super Elastic Nitinol Staple System SURGICAL TECHNIQUE Super Elastic Nitinol Staple System Table of Contents Design Rationale...2 System Features...2 Indications...2 Contraindications...2
Integra MemoFix Super Elastic Nitinol Staple System SURGICAL TECHNIQUE Super Elastic Nitinol Staple System Table of Contents Design Rationale...2 System Features...2 Indications...2 Contraindications...2
Biomimetic Product catalogue
 Biomimetic Product catalogue 20 Pol. Industrial Santa Anna I Apartado 20 08251 Santpedor (Barcelona) España T. (+34) 902 38 38 48 F. (+34) 93 827 38 73 www.avinent.com avinent@avinent.com 1 CORAL is the
Biomimetic Product catalogue 20 Pol. Industrial Santa Anna I Apartado 20 08251 Santpedor (Barcelona) España T. (+34) 902 38 38 48 F. (+34) 93 827 38 73 www.avinent.com avinent@avinent.com 1 CORAL is the
ECOCERT CANADA LABELLING GUIDE. For Canadian Certified Organic Products. Version3
 ECOCERT CANADA LABELLING GUIDE For Canadian Certified Organic Products Version3 2014 TABLE OF CONTENTS Validation of label design........3 Use of the Ecocert Trademark....4 Changes to labels.... 4 Prerequisites
ECOCERT CANADA LABELLING GUIDE For Canadian Certified Organic Products Version3 2014 TABLE OF CONTENTS Validation of label design........3 Use of the Ecocert Trademark....4 Changes to labels.... 4 Prerequisites
Latest range of highest quality sterile and filtered cleanroom alcohols
 Latest range of highest quality sterile and filtered cleanroom alcohols Shield Medicare Company Profile Shield Medicare is a market leader in the supply of specialist contamination control products to
Latest range of highest quality sterile and filtered cleanroom alcohols Shield Medicare Company Profile Shield Medicare is a market leader in the supply of specialist contamination control products to
Quality Management System Certification. Understanding Quality Management System (QMS) certification
 Quality Management System Certification Understanding Quality Management System (QMS) certification The medical device manufacturing sector is one of the most regulated sectors in which significant quality
Quality Management System Certification Understanding Quality Management System (QMS) certification The medical device manufacturing sector is one of the most regulated sectors in which significant quality
Dental Instruments. Edition 2. Single Use Instruments. Absorbents. Wipes. Woundcare. Disposable Gloves. Patient Identification. First Aid.
 Single Use Instruments Absorbents Wipes Woundcare Disposable Gloves Patient Identification First Aid Dental Instruments Cotton Wool Personal Care Continence Care Edition 2 Instrapac Single Use Dental Instruments
Single Use Instruments Absorbents Wipes Woundcare Disposable Gloves Patient Identification First Aid Dental Instruments Cotton Wool Personal Care Continence Care Edition 2 Instrapac Single Use Dental Instruments
CLICK SEAL SYSTEM. Thermo Scientific Samco Clicktainer Vial. Unique click seal system providing sample protection and user safety
 LEAKPROOF 95KPA TESTED CE MARK CLICK SEAL SYSTEM Thermo Scientific Samco Clicktainer Vial Unique click seal system providing sample protection and user safety Thermo Scientific Samco Clicktainer Vial Providing
LEAKPROOF 95KPA TESTED CE MARK CLICK SEAL SYSTEM Thermo Scientific Samco Clicktainer Vial Unique click seal system providing sample protection and user safety Thermo Scientific Samco Clicktainer Vial Providing
Восток-пром CATALOGUE INSEMINATION & IN VITRO FERTILIZATION КАТАЛОГ ИНСЕМИНАЦИЯ & РЕПРОДУКТИВНАЯ МЕДИЦИНА. медикал
 CATALOGUE INSEMINATION & IN VITRO FERTILIZATION КАТАЛОГ ИНСЕМИНАЦИЯ & РЕПРОДУКТИВНАЯ МЕДИЦИНА Central HQ Karadžičová St. Nb 8-A 82108 Bratislava - SLOVAKIA Phone/Fax Land-line: +421 (0) 2 5939 6000 Fax:
CATALOGUE INSEMINATION & IN VITRO FERTILIZATION КАТАЛОГ ИНСЕМИНАЦИЯ & РЕПРОДУКТИВНАЯ МЕДИЦИНА Central HQ Karadžičová St. Nb 8-A 82108 Bratislava - SLOVAKIA Phone/Fax Land-line: +421 (0) 2 5939 6000 Fax:
Product Family Matrix Single-Use Earplugs No-roll design, enhanced communication
 Product Family Matrix Single-Use Earplugs No-roll design, enhanced communication Learn more about our products at howardleight.com > Learn more about hearing conservation at hearforever.org > View More
Product Family Matrix Single-Use Earplugs No-roll design, enhanced communication Learn more about our products at howardleight.com > Learn more about hearing conservation at hearforever.org > View More
Your guarantee of Success Since 1974
 2310 Seongnam-dong, Jungwon-gu, Seongnam-si Gyeonggi-do, 462-824, Korea Tel : 82-31-752-4070, Fax : 82-31-752-4074 http://www.suredent.com, E-mail : suredent@suredent.com Your guarantee of Success Since
2310 Seongnam-dong, Jungwon-gu, Seongnam-si Gyeonggi-do, 462-824, Korea Tel : 82-31-752-4070, Fax : 82-31-752-4074 http://www.suredent.com, E-mail : suredent@suredent.com Your guarantee of Success Since
040716_ANDI_VENTIS WEBSITE(new) Printout 1. HOME PAGE. Welcome to our world of Dry Powder Inhalers!
 1. HOME PAGE Welcome to our world of Dry Powder Inhalers! Andi-Ventis Ltd, established in 2001 is a joint venture between Medochemie Ltd, Cyprus and Miat SpA Italy, founded to carry out the pharmaceutical
1. HOME PAGE Welcome to our world of Dry Powder Inhalers! Andi-Ventis Ltd, established in 2001 is a joint venture between Medochemie Ltd, Cyprus and Miat SpA Italy, founded to carry out the pharmaceutical
Micro Production Part.
 Micro Production Part www.mipro-inc.com Worldwide activity 3 Who we are 4 Materials 5 Surface Treatment 6 Packaging 7 Letter from CEO 8 Cover 10 SOI Implant 11-12 TOI Implant 13-14 NSI Implant 15-16 SHI
Micro Production Part www.mipro-inc.com Worldwide activity 3 Who we are 4 Materials 5 Surface Treatment 6 Packaging 7 Letter from CEO 8 Cover 10 SOI Implant 11-12 TOI Implant 13-14 NSI Implant 15-16 SHI
CLICK SEAL SYSTEM. Thermo Scientific Samco Clicktainer Vial. Unique click seal system providing sample protection and user safety
 LEAKPROOF 95KPA TESTED CE MARK CLICK SEAL SYSTEM Thermo Scientific Samco Clicktainer Vial Unique click seal system providing sample protection and user safety Thermo Scientific Samco Clicktainer Vial Providing
LEAKPROOF 95KPA TESTED CE MARK CLICK SEAL SYSTEM Thermo Scientific Samco Clicktainer Vial Unique click seal system providing sample protection and user safety Thermo Scientific Samco Clicktainer Vial Providing
Isn t it worth two bags?
 Isn t it worth two bags? NEW from Miltenyi Biotec CryoMACS Freezing Bag CryoMACS Freezing Bag Designed for freezing and cryopreservation of blood components Overwrap bag protects both sample and freezing
Isn t it worth two bags? NEW from Miltenyi Biotec CryoMACS Freezing Bag CryoMACS Freezing Bag Designed for freezing and cryopreservation of blood components Overwrap bag protects both sample and freezing
TABLE OF CONTENTS P. 4-5 P. 6-7 P. 8-9 P P P P
 Full Smile Solution TABLE OF CONTENTS P. 4-5 P. 6-7 P. 8-9 P. 10-11 P. 12-13 P. 14-15 P. 16-17 FULL SMILE SOLUTION MGUIDE PROCESS ADVANTAGES CUSTOM SOLUTIONS MGUIDE SURGICAL SETS MCENTER REQUIRED SOURCES
Full Smile Solution TABLE OF CONTENTS P. 4-5 P. 6-7 P. 8-9 P. 10-11 P. 12-13 P. 14-15 P. 16-17 FULL SMILE SOLUTION MGUIDE PROCESS ADVANTAGES CUSTOM SOLUTIONS MGUIDE SURGICAL SETS MCENTER REQUIRED SOURCES
Part 1: General requirements
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-1 Second edition 2013-06-15 Corrected version 2014-01-15 Intravascular catheters Sterile and single-use catheters Part 1: General requirements
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-1 Second edition 2013-06-15 Corrected version 2014-01-15 Intravascular catheters Sterile and single-use catheters Part 1: General requirements
DUAL CHAMBER SYSTEMS
 DUAL CHAMBER SYSTEMS HEALTH is the inestimable value to which Pharmaceutical Industry is committed. BORMIOLI PHARMA contributes to this mission by following the same values that guide this industry: quality,
DUAL CHAMBER SYSTEMS HEALTH is the inestimable value to which Pharmaceutical Industry is committed. BORMIOLI PHARMA contributes to this mission by following the same values that guide this industry: quality,
QA Regulatory forum: Classification and Risk assessment according to MDR Date : T h e r e s e. A l b i n s s o m e d q t e c h.
 QA Regulatory forum: Classification and Risk assessment according to MDR Date : 05.04.2018 THERESE ALBINSSON T h e r e s e. A l b i n s s o n @ m e d q t e c h. s e MedQtech AB MedQtech AB is a consultant
QA Regulatory forum: Classification and Risk assessment according to MDR Date : 05.04.2018 THERESE ALBINSSON T h e r e s e. A l b i n s s o n @ m e d q t e c h. s e MedQtech AB MedQtech AB is a consultant
A complete surgical kit for Premium and Shelta systems
 SURGICAL KIT A complete surgical kit for Premium and Shelta systems The combined Premium Shelta surgical kit contains the instruments for the surgical and prosthetic phases of the fixtures of both implant
SURGICAL KIT A complete surgical kit for Premium and Shelta systems The combined Premium Shelta surgical kit contains the instruments for the surgical and prosthetic phases of the fixtures of both implant
THE DOSING SPECIALIST
 04 THE DOSING SPECIALIST THE COMPANY WHO ARE WE? Over the past 30 years, ROVIPHARM has become the specialist manufacturer of medical and dosing devices for the pharmaceutical and healthcare industries.
04 THE DOSING SPECIALIST THE COMPANY WHO ARE WE? Over the past 30 years, ROVIPHARM has become the specialist manufacturer of medical and dosing devices for the pharmaceutical and healthcare industries.
Qualitix reliable consumables by excellence
 Socorex compatibility chart Chart indicates optimal combination of Qualitix and Socorex pipettes. Acura manual and Acura electro 815 825 / 826 / 926 835 / 936 855 / 956 Calibra digital 822 832 852 10 µl
Socorex compatibility chart Chart indicates optimal combination of Qualitix and Socorex pipettes. Acura manual and Acura electro 815 825 / 826 / 926 835 / 936 855 / 956 Calibra digital 822 832 852 10 µl
Intracytoplasmic Sperm Injection (ICSI) in the Mouse with the Eppendorf PiezoXpert : How to Increase Oocyte Survival Rates After Injection
 APPLICATION NOTE No. 395 Intracytoplasmic Sperm Injection (ICSI) in the Mouse with the Eppendorf PiezoXpert : How to Increase Oocyte Survival Rates After Injection Nuno Costa-Borges 1*, Enric Mestres 1,
APPLICATION NOTE No. 395 Intracytoplasmic Sperm Injection (ICSI) in the Mouse with the Eppendorf PiezoXpert : How to Increase Oocyte Survival Rates After Injection Nuno Costa-Borges 1*, Enric Mestres 1,
GYNO INFO INTRAUTERINE INSEMINATION CATHETER RANGE IUI #4220. Smooze IUI #4225. Smooze IUI Medium
 GYNO INFO INTRAUTERINE INSEMINATION CATHETER RANGE PRODUCT ORDER NUMBER IUI #4220 IUI Memo IUI Plus #4220 Memo #4220 Plus Smooze IUI #4225 Smooze IUI Medium Smooze IUI Long Seminor Rigid Seminor Flexible
GYNO INFO INTRAUTERINE INSEMINATION CATHETER RANGE PRODUCT ORDER NUMBER IUI #4220 IUI Memo IUI Plus #4220 Memo #4220 Plus Smooze IUI #4225 Smooze IUI Medium Smooze IUI Long Seminor Rigid Seminor Flexible
Integra. DigiFuse Cannulated Intramedullary Fusion System SURGICAL TECHNIQUE
 Integra DigiFuse Cannulated Intramedullary Fusion System SURGICAL TECHNIQUE Table of Contents Design Rationale... 2 System Features... 2 Indications... 2 Contraindications... 2 Surgical Technique...3 Step
Integra DigiFuse Cannulated Intramedullary Fusion System SURGICAL TECHNIQUE Table of Contents Design Rationale... 2 System Features... 2 Indications... 2 Contraindications... 2 Surgical Technique...3 Step
NEPHROCHECK Calibration Verification Kit Package Insert
 NEPHROCHECK Calibration Verification Kit Package Insert Manufactured for Astute Medical, Inc. 3550 General Atomics Ct. Building 2 San Diego, CA 92121 USA Intended Use The NEPHROCHECK Calibration Verification
NEPHROCHECK Calibration Verification Kit Package Insert Manufactured for Astute Medical, Inc. 3550 General Atomics Ct. Building 2 San Diego, CA 92121 USA Intended Use The NEPHROCHECK Calibration Verification
EC-PRO Embryo Transfer CATHETERS
 EC-PRO Embryo Transfer CATHETERS Designed following the advice of some of the most reputable gynaecologists in the world. Complying with the high quality standards of Kitazato, EC-PRO catheters offer gynaecologists
EC-PRO Embryo Transfer CATHETERS Designed following the advice of some of the most reputable gynaecologists in the world. Complying with the high quality standards of Kitazato, EC-PRO catheters offer gynaecologists
BACKFLUSH INSTRUMENTS
 BACKFLUSH INSTRUMENTS Versatility designed to handle all your surgical backflush needs. About VitreQ VitreQ creates instruments for the ophthalmic society with a special focus on vitreoretinal surgery.
BACKFLUSH INSTRUMENTS Versatility designed to handle all your surgical backflush needs. About VitreQ VitreQ creates instruments for the ophthalmic society with a special focus on vitreoretinal surgery.
Race family Endodontic NiTi rotary files
 EN Race family Endodontic NiTi rotary files Foreword Race (Reamer with Alternating Cutting Edges) With the appearance of nickel-titanium (NiTi) alloy, the domain of endodontic instrument production discovered
EN Race family Endodontic NiTi rotary files Foreword Race (Reamer with Alternating Cutting Edges) With the appearance of nickel-titanium (NiTi) alloy, the domain of endodontic instrument production discovered
STERILITY TESTING OF PHARMACEUTICAL PRODUCTS
 STERILITY TESTING OF PHARMACEUTICAL PRODUCTS Tim Sandle CONTENTS Introduction xiii 1 STERILITY 1 Introduction 1 Sterility 3 Microorganisms and Microbial Growth 5 Types of microorganisms 7 Sterilization
STERILITY TESTING OF PHARMACEUTICAL PRODUCTS Tim Sandle CONTENTS Introduction xiii 1 STERILITY 1 Introduction 1 Sterility 3 Microorganisms and Microbial Growth 5 Types of microorganisms 7 Sterilization
Standard Operating Procedure Title: Bacterial Endotoxin Testing (LAL) - Gel-Clot Method
 The reagents used for testing and all the disposable equipment must be disposed of into the Biohazard Bin. Table of Contents 1. General... 2 2. Preparation of the Endotoxin... 3 3. Preparation of Test
The reagents used for testing and all the disposable equipment must be disposed of into the Biohazard Bin. Table of Contents 1. General... 2 2. Preparation of the Endotoxin... 3 3. Preparation of Test
Aer ODE B3-20. Database System for GEX B3 Dosimetry. Aérial. Optical Dosimetry Equipment. (gamma and/or electron beam versions)
 B3-20 Database System for GEX B3 Dosimetry (gamma and/or electron beam versions) and GEX combine to provide a Genesys 20 version of the flexible 21 CFR Part 11 certified database dosimetry system for use
B3-20 Database System for GEX B3 Dosimetry (gamma and/or electron beam versions) and GEX combine to provide a Genesys 20 version of the flexible 21 CFR Part 11 certified database dosimetry system for use
SEQUENTIAL CULTURE MEDIA SYSTEM
 SEQUENTIAL CULTURE MEDIA SYSTEM Gamete solutions... 7 Fertilization solutions...15 Cleavage solutions...21 Blastocyst solutions...27 STIC Media Stopper Removal Tool...35 Complete list of order numbers...36
SEQUENTIAL CULTURE MEDIA SYSTEM Gamete solutions... 7 Fertilization solutions...15 Cleavage solutions...21 Blastocyst solutions...27 STIC Media Stopper Removal Tool...35 Complete list of order numbers...36
Preparation Instructions for the CAMLOG /CONELOG Implant System
 J8000.0032 Rev.6 EN 06/2015 Preparation Instructions for the CAMLOG /CONELOG Implant System ENGLISH The following descriptions contain detailed instructions on cleaning, disinfection and sterilization
J8000.0032 Rev.6 EN 06/2015 Preparation Instructions for the CAMLOG /CONELOG Implant System ENGLISH The following descriptions contain detailed instructions on cleaning, disinfection and sterilization
Lakshy Management Consultant Pvt. Ltd.
 What is ISO 13485:2003? You can t buy trust. BUILD IT through ISO 13485. Demonstrate your ability to supply medical devices through Quality Management System for Medical Devices - ISO 13485 ISO 13485 is
What is ISO 13485:2003? You can t buy trust. BUILD IT through ISO 13485. Demonstrate your ability to supply medical devices through Quality Management System for Medical Devices - ISO 13485 ISO 13485 is
The Interpretation of the relative Cytotoxic testing for gloves for the safety of the operator
 The Interpretation of the relative Cytotoxic testing for gloves for the safety of the operator Glove Selection factors Standards and guidelines for Gloves Cytotoxic testing BioClean ISO Class 4 100kg Washer
The Interpretation of the relative Cytotoxic testing for gloves for the safety of the operator Glove Selection factors Standards and guidelines for Gloves Cytotoxic testing BioClean ISO Class 4 100kg Washer
FOR C1 & V3 IMPLANTS STEP-BY-STEP GUIDED SURGICAL PROCEDURE
 FOR C1 & V3 IMPLANTS STEP-BY-STEP GUIDED SURGICAL PROCEDURE 1 2 LOGICAL LAYOUT 1 Example: C1 Implant Ø4.20 / 11.5L ; Soft Bone Ø4.20 / 11.5L MG-D0624 MG-D1124 MG-D1133 MG-D1137 2 Example: Implant C1 Ø4.20
FOR C1 & V3 IMPLANTS STEP-BY-STEP GUIDED SURGICAL PROCEDURE 1 2 LOGICAL LAYOUT 1 Example: C1 Implant Ø4.20 / 11.5L ; Soft Bone Ø4.20 / 11.5L MG-D0624 MG-D1124 MG-D1133 MG-D1137 2 Example: Implant C1 Ø4.20
To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice.
 1.0 Purpose: To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice. Useful References: Kimura, Y & Yanagimuach1 R (1995) Intracytoplasmic sperm injection
1.0 Purpose: To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice. Useful References: Kimura, Y & Yanagimuach1 R (1995) Intracytoplasmic sperm injection
Acute Therapy Systems. provencare Catheter Sets
 Acute Therapy Systems provencare Catheter Sets Catheter Sets Contents provencare Catheter Sets 4 provencare Product Features and Accessories 6 provencare Single Lumen Catheter Sets 8 provencare Paediatric
Acute Therapy Systems provencare Catheter Sets Catheter Sets Contents provencare Catheter Sets 4 provencare Product Features and Accessories 6 provencare Single Lumen Catheter Sets 8 provencare Paediatric
DuPont Tyvek IsoClean, Model IC 458 B WH MS
 Technical data sheet DuPont Tyvek IsoClean, Model IC 458 B WH MS CAT I Option MS SAL Product Description DuPont Tyvek IsoClean boot cover model IC 458 B WH MS. Tunnelled elastication at shin. Ties. Elasticated
Technical data sheet DuPont Tyvek IsoClean, Model IC 458 B WH MS CAT I Option MS SAL Product Description DuPont Tyvek IsoClean boot cover model IC 458 B WH MS. Tunnelled elastication at shin. Ties. Elasticated
CONFIGURE YOUR OWN CUSTOM-MADE SUPERIOR FOLLICLE ASPIRATION SET IN 7 STEPS
 CONFIGURE YOUR OWN CUSTOM-MADE SUPERIOR FOLLICLE ASPIRATION SET IN 7 STEPS COPY OR DOWNLOAD AT WWW.GYNETICS.BE, COMPLETE AND RETURN FOR INQUIRY STEP 1 CHOOSE YOUR FLUSH NEEDLE TICK BOX Needle Lumen Gauge
CONFIGURE YOUR OWN CUSTOM-MADE SUPERIOR FOLLICLE ASPIRATION SET IN 7 STEPS COPY OR DOWNLOAD AT WWW.GYNETICS.BE, COMPLETE AND RETURN FOR INQUIRY STEP 1 CHOOSE YOUR FLUSH NEEDLE TICK BOX Needle Lumen Gauge
InterMedics Training Academy (ITA) Embryology Training and Lab Management
 InterMedics Training Academy (ITA) Embryology Training and Lab Management InterMedics is at the forefront of IVF service delivery in India and has recently opened its own training facility in Mumbai. Optimal
InterMedics Training Academy (ITA) Embryology Training and Lab Management InterMedics is at the forefront of IVF service delivery in India and has recently opened its own training facility in Mumbai. Optimal
Plate/Valve Specifications: Thickness: 0.9mm Width: 13.00mm Length: 16.00mm Surface Area: mm 2
 Distribué en France par FCI S.A.S. France Chirurgie Instrumentation SAS 20/22 rue Louis Armand 75015 PARIS Tél. 01.53.98.98.98 / Fax. 01.53.98.98.99 fci@fci.fr / www.fci.fr CATALOGUE DE VENTE Features:
Distribué en France par FCI S.A.S. France Chirurgie Instrumentation SAS 20/22 rue Louis Armand 75015 PARIS Tél. 01.53.98.98.98 / Fax. 01.53.98.98.99 fci@fci.fr / www.fci.fr CATALOGUE DE VENTE Features:
Sterile hypodermic needles for single use Requirements and test methods
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 7864 Fourth edition 2016-08-01 Sterile hypodermic needles for single use Requirements and test methods Aiguilles hypodermiques stériles, non réutilisables
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 7864 Fourth edition 2016-08-01 Sterile hypodermic needles for single use Requirements and test methods Aiguilles hypodermiques stériles, non réutilisables
For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges.
 MS9662 MS9663 HumaPen LUXURA INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. Your HumaPen LUXURA
MS9662 MS9663 HumaPen LUXURA INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. Your HumaPen LUXURA
Public Assessment Report for a Traditional Herbal Medicinal Product for Human Use
 IPAR Public Assessment Report for a Traditional Herbal Medicinal Product for Human Use Ginkgoforce Memory & Circulation Ginkgo Tablets Extract (as dry extract) from fresh leaves of Ginkgo biloba (Ginkgo
IPAR Public Assessment Report for a Traditional Herbal Medicinal Product for Human Use Ginkgoforce Memory & Circulation Ginkgo Tablets Extract (as dry extract) from fresh leaves of Ginkgo biloba (Ginkgo
SPERM PREPARATION, HANDLING & STORAGE
 SPERM PREPARATION, HANDLING & STORAGE Solutions for the andrology laboratory HELP AT EVERY STEP with simpler processes, less stress and better results Multipurpose Handling Medium RETRIEVE AND RINSE OOCYTES
SPERM PREPARATION, HANDLING & STORAGE Solutions for the andrology laboratory HELP AT EVERY STEP with simpler processes, less stress and better results Multipurpose Handling Medium RETRIEVE AND RINSE OOCYTES
FOOD ACT No.26 OF 1980
 FOOD ACT No.26 OF 1980 REGULATIONS made by the Minister of Health in consultation with the Food Advisory Committee under section 32 of the Food Act No. 26 of 1980. Colombo 15 th November, 2005 Nimal Siripala
FOOD ACT No.26 OF 1980 REGULATIONS made by the Minister of Health in consultation with the Food Advisory Committee under section 32 of the Food Act No. 26 of 1980. Colombo 15 th November, 2005 Nimal Siripala
For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges.
 MS9673 HumaPen LUXURA HD INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. The colour of your HumaPen
MS9673 HumaPen LUXURA HD INSULIN DELIVERY DEVICE INSTRUCTIONS FOR USE For use only with Lilly 3 ml insulin cartridges (100 units/ml). Do not use other brands of insulin cartridges. The colour of your HumaPen
Biopsy of Blastomeres from Cleavage-stage Mouse Embryos with Eppendorf PiezoXpert
 APPLICATION NOTE No. 270 Biopsy of Blastomeres from Cleavage-stage Mouse Embryos with Eppendorf PiezoXpert Norhazlin Jusoh Mohd. Yusoff and Nor Ashikin Mohamed Noor Khan, Institute of Medical Molecular
APPLICATION NOTE No. 270 Biopsy of Blastomeres from Cleavage-stage Mouse Embryos with Eppendorf PiezoXpert Norhazlin Jusoh Mohd. Yusoff and Nor Ashikin Mohamed Noor Khan, Institute of Medical Molecular
EC-PRO Routine Embryo Transfer CATHETERS
 EC-PRO Routine Kitazato s superior catheter softness. Rounded tips to minimize trauma. Keep memory when giving shape. Easy hub to introduce the catheter. Ergonomic handle for better control. Laser-printed
EC-PRO Routine Kitazato s superior catheter softness. Rounded tips to minimize trauma. Keep memory when giving shape. Easy hub to introduce the catheter. Ergonomic handle for better control. Laser-printed
15. Procuring, processing and transporting gametes and
 15. Procuring, processing and transporting gametes and embryos Version 6.0 On this page: : Extracts from the HFE Act Directions HFEA guidance: Documented procedures: general Patient selection and procurement
15. Procuring, processing and transporting gametes and embryos Version 6.0 On this page: : Extracts from the HFE Act Directions HFEA guidance: Documented procedures: general Patient selection and procurement
closed tray technique using the indirect transfer coping
 closed tray technique using the indirect transfer coping impression techniques closed tray technique using the indirect transfer coping Use this technique to make a single or multiple-unit, implant-level
closed tray technique using the indirect transfer coping impression techniques closed tray technique using the indirect transfer coping Use this technique to make a single or multiple-unit, implant-level
Cement Polished Tapered Stems of 12/14 Taper. 96 mm 98 mm 104 mm 110 mm 116 mm 122 mm 128 mm. Ceramic Femoral Head. Outer Diameter
 Design Philosophy Cementless Stems of 12/14 Taper Stem Length Neck shaft Angle STEM-N3 STEM-N4 STEM-N5 STEM-N6 STEM-N7 STEM-N8 STEM-N9 123 mm 125 mm 130 mm 135 mm 140 mm 145 mm 150 mm 132 Cement Polished
Design Philosophy Cementless Stems of 12/14 Taper Stem Length Neck shaft Angle STEM-N3 STEM-N4 STEM-N5 STEM-N6 STEM-N7 STEM-N8 STEM-N9 123 mm 125 mm 130 mm 135 mm 140 mm 145 mm 150 mm 132 Cement Polished
College of Physicians and Surgeons of Saskatchewan STANDARDS. Assisted Reproductive Technology PREAMBLE
 College of Physicians and Surgeons of Saskatchewan STANDARDS Assisted Reproductive Technology STATUS: UNDER REVIEW Approved by Council: September 2012 Amended: November 2015 To be reviewed: November 2020
College of Physicians and Surgeons of Saskatchewan STANDARDS Assisted Reproductive Technology STATUS: UNDER REVIEW Approved by Council: September 2012 Amended: November 2015 To be reviewed: November 2020
NARROW DIAMETER implant
 N NARROW IAMETER implant N Precision dental solutions C-Tech Implant is a dynamic company with aggressive growth, producing components and product lines primarily for dental implantology. International
N NARROW IAMETER implant N Precision dental solutions C-Tech Implant is a dynamic company with aggressive growth, producing components and product lines primarily for dental implantology. International
Medical Device VSP Design & Manufacturing. Virtual Surgical Planning
 Medical Device VSP Design & Manufacturing Virtual Surgical Planning Overview 3D printing technology is at an inflection point with the medical device industry rapidly adopting 3D printing technology. This
Medical Device VSP Design & Manufacturing Virtual Surgical Planning Overview 3D printing technology is at an inflection point with the medical device industry rapidly adopting 3D printing technology. This
Talon Compounding Pharmacy 10/3/17
 Talon Compounding Pharmacy 10/3/17 Office of Pharmaceutical Quality Operations, Division II 4040 N. Central Expressway, Suite 300 Dallas, Texas 75204 October 3, 2017 CMS Case # 522630 VIA UPS EXPRESS WARNING
Talon Compounding Pharmacy 10/3/17 Office of Pharmaceutical Quality Operations, Division II 4040 N. Central Expressway, Suite 300 Dallas, Texas 75204 October 3, 2017 CMS Case # 522630 VIA UPS EXPRESS WARNING
A complete surgical kit for Premium and Shelta systems
 SURGICAL KIT A complete surgical kit for Premium and Shelta systems The combined Premium Shelta surgical kit contains the instruments for the surgical and prosthetic phases of the fixtures of both implant
SURGICAL KIT A complete surgical kit for Premium and Shelta systems The combined Premium Shelta surgical kit contains the instruments for the surgical and prosthetic phases of the fixtures of both implant
What public health benefits could be secured by using universal labelling & packaging for influenza pandemic vaccines?
 What public health benefits could be secured by using universal labelling & packaging for influenza pandemic vaccines? Workshop on the joint procurement of medical countermeasures 29 April 2015, Luxembourg
What public health benefits could be secured by using universal labelling & packaging for influenza pandemic vaccines? Workshop on the joint procurement of medical countermeasures 29 April 2015, Luxembourg
Prescription Drug Importation: Can it Help America's Seniors? Safety of Imported Medications: I-SaveRx Case Study
 Prescription Drug Importation: Can it Help America's Seniors? Safety of Imported Medications: I-SaveRx Case Study Presented By: Scott McKibbin Special Advocate For Prescription Drugs State Of Illinois
Prescription Drug Importation: Can it Help America's Seniors? Safety of Imported Medications: I-SaveRx Case Study Presented By: Scott McKibbin Special Advocate For Prescription Drugs State Of Illinois
BODY FLUID TREATMENT
 BODY FLUID TREATMENT TORASET SET FOR THORACENTESIS Toracentesis Set for aspiration of pleuric exudates. - Practical system with single-acting valves. - Waste bag with non-return valve and drainage tap.
BODY FLUID TREATMENT TORASET SET FOR THORACENTESIS Toracentesis Set for aspiration of pleuric exudates. - Practical system with single-acting valves. - Waste bag with non-return valve and drainage tap.
TROUBLESHOOTING PGT CRB Workshop. PhD, HCLD, CC
 TROUBLESHOOTING PGT 2018 CRB Workshop Jean M. Popwell PhD, HCLD, CC PREIMPLANTATION GENETIC TESTING (PGT) WHAT IS TROUBLESHOOTING? Planning ahead is always a great goal but we all should learn how to TROUBLESHOOT
TROUBLESHOOTING PGT 2018 CRB Workshop Jean M. Popwell PhD, HCLD, CC PREIMPLANTATION GENETIC TESTING (PGT) WHAT IS TROUBLESHOOTING? Planning ahead is always a great goal but we all should learn how to TROUBLESHOOT
