Alkaline phosphatase in stallion semen: characterization and clinical applications
|
|
|
- Abigail Robinson
- 6 years ago
- Views:
Transcription
1 Theriogenology 60 (2003) 1±10 Alkaline phosphatase in stallion semen: characterization and clinical applications R.M.O. Turner *, S.M. McDonnell Department of Clinical Studies, New Bolton Center, University of Pennsylvania School of Veterinary Medicine, Kennett Square, PA , USA Received 2 August 2001; accepted 22 January 2002 Abstract Signi cant amounts of alkaline phosphatase (AP) activity have been found in semen plasma from numerous species. In species in which the majority of semen plasma AP (SPAP) activity originates from the epididymis and testicle, SPAP activity can be used clinically as a marker to differentiate testicular origin azoospermia or oligospermia from ejaculatory failure. Information on SPAP activity in stallions to date has been limited. In this study, a standard clinical chemistry analyzer was used to determine AP activity in pre-ejaculatory uid and ejaculates from groups of normal stallions. Additionally, accessory glands, epididymides, testicles and other components of the urogenital tract of normal stallions were assayed to determine which tissues contain SPAP activity. The results indicated that levels of AP activity are low in pre-ejaculatory uid, but signi cantly higher in ejaculatory uid from normal stallions. Spermatozoa were not a signi cant source of SPAP activity. High levels of SPAP activity were found in the testes and epididymides. These ndings suggest that SPAP activity is a candidate for a sperm-independent marker for ejaculation in the stallion. Finally, AP activity was determined in ejaculatory uid from a stallion with bilaterally blocked ampullae, both before and after relief of the blockage. While the blockage was present, AP activity in ejaculatory uid was low. However, following relief of the blockage, AP activity in ejaculatory uid rose dramatically, thus suggesting that AP activity will be useful as an inexpensive, simple clinical assay for differentiating ejaculatory failure or excurrent duct blockages from testicular origin azoospermia and oligospermia. # 2002 Elsevier Science Inc. All rights reserved. Keywords: Stallion; Alkaline phosphatase; Ejaculate; Azoospermia; Oligospermia * Corresponding author. Tel.: ; fax: address: rmturner@vet.upenn.edu (R.M.O. Turner) X/02/$ ± see front matter # 2002 Elsevier Science Inc. All rights reserved. PII: S X(02)
2 2 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 1. Introduction Alkaline phosphatase (AP) is a dephosphorylating enzyme that is active in many tissues including bone, liver, kidney, intestine, lung and placenta. Variable levels of AP activity also have been reported in the semen plasma of men, dogs, toms, bulls, rabbits, rams, goats, buffalo, cocks, turkeys, boars and camels [1±9] where it is believed to be involved in sperm glycolytic reactions and fructose formation [2]. The site of production of semen plasma AP (SPAP) has been determined in some species. In dogs, the majority of SPAP is produced in the epididymis [10]. In the rabbit, the testicle, epididymis, vas deferens and ampulla all synthesize signi cant amounts of the enzyme [7,11,12]. In contrast, the majority of SPAP in bulls originates from the seminal vesicles and, to a lesser extent, from the testes and epididymides [13] while in men, the majority of SPAP activity originates from the prostate [14]. In species in which the majority of SPAP activity originates from the epididymis and testicle, SPAP activity can be used clinically as an ejaculatory marker to differentiate azoospermia or oligospermia from ejaculatory failure [15]. Little information is available on AP activity in the semen plasma of stallions. One early report suggested that a signi cant amount of SPAP activity was present in secretions from the ampullae [16]. Additionally, a preliminary study found high levels of AP activity in stallion testes and epididymides [17]. As part of an overall goal of developing better diagnostic and therapeutic approaches for stallions with ejaculatory problems, the aims of this study were: to determine if AP activity is present in the ejaculate of normal stallions and, if present, to determine its normal concentration and total activity; to determine which tissues of the stallion reproductive tract contain AP activity, and to determine if AP might be useful as a clinical marker for ejaculation in the stallion. 2. Materials and methods 2.1. Determination of presence and levels of AP activity in normal stallion ejaculates Pre-ejaculatory uid and ejaculate samples were obtained from nine horse and pony stallions actively involved in successful breeding programs. The stallions represented various breeds and ranged in age from 6 to 24 years. All stallions were classi ed as fertile based on breeding history, testicular palpation, testicular measurement and semen analysis (>60% progressively motile spermatozoa, >60% percent morphologically normal spermatozoa, total sperm numbers within normal limits for testicular size, total number of morphologically normal, progressively motile spermatozoa > in each ejaculate and satisfactory longevity of motility at 4 8C [18,19]). Samples were obtained as part of routine semen collections for shipment during April±July of a single year. Following collection, each semen sample was ltered to remove the gel portion (Kleen-Test In-Line Milk Filters, Agway, Oxford, PA). Pre-ejaculatory uid samples were collected either by manual stimulation of the erect glans penis prior to mounting or, in cases in which an unproductive mount occurred, directly into a collection bag attached to a Missouri Arti cial Vagina (MAV). Ejaculate samples were collected routinely in a MAV while stallions were mounted on a dummy or on an ovariectomized mount mare. All pre-ejaculatory uid
3 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 3 Table 1 Alkaline phosphatase (AP) activity in ejaculates and pre-ejaculatory fluid from normal stallions Stallion ID Ejaculate volume (ml) AP activity in pre-ejaculatory fluid (IU/l) AP activity in ejaculate (IU/l) Total AP activity per ejaculate (IU) Not available Not assessed Mean (S.D.) 29 (22) (12398) 1516 (1039) and ejaculate samples were stored at 4 8C for no longer than 24 h prior to being assayed for SPAP activity. The concentration of SPAP activity (IU/l) was determined in pre-ejaculatory uid and ejaculate samples for each stallion. The concentration of SPAP activity was measured using a Kodak Ektachem Clinical Chemistry Analyzer according to the manufacturer's recommendation (Eastman Kodak Company, Rochester, NY). Samples containing AP activity that exceeded the analyzer's dynamic range (>1500 IU/l) were diluted with 7% bovine serum albumin in reagent grade water and reanalyzed. The results then were multiplied by the dilution factor to obtain the original sample's AP activity. For eight of the nine stallions, total ejaculate AP activity (IU) was determined by multiplying the concentration of AP activity (IU/l) by the gel-free ejaculate volume (ml) and then dividing by 1000 (Table 1). The concentration of AP activity in pre-ejaculatory uid samples was compared to that in gel-free ejaculate samples using the paired t-test Determination of the tissue of origin of SPAP activity To rule out spermatozoa as the primary source of SPAP activity, ejaculates were collected from 11 fertile horse and pony stallions and the concentration of AP activity was compared between unprocessed ejaculates and sperm-free semen plasma. Ejaculates were collected in April and May of a single year using either a MAV while mounted on a dummy or an ovariectomized mount mare or manual stimulation while mounted on a dummy [20]. The stallions represented various breeds and ranged in age from 4 to 18 years. All ejaculates were classi ed as normal based on the criteria described previously. Half of each ejaculate was centrifuged at 16,000 g for 20min and the sperm-free semen plasma supernatant was removed from the sperm pellet. The other half of each ejaculate was left unprocessed. Unprocessed ejaculates and paired sperm-free semen plasma were stored at 4 8C for no more than 12 h before being assayed for AP activity. For 9 of the 11 stallions, total AP activity was determined in sperm-free semen plasma and in the unprocessed ejaculate by multiplying the appropriate AP concentration (IU/l) by 1/2 of the total ejaculate volume (ml) and dividing by Total ejaculate AP activity (IU) was
4 4 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 Table 2 Alkaline phosphatase (AP) activity in paired sperm-free seminal plasma and unprocessed ejaculates from normal stallions Stallion ID Ejaculate volume (ml) AP activity in sperm free seminal plasma (IU/l) AP activity in unprocessed ejaculate (IU/l) Total AP activity per ejaculate (IU) Not available Not assessed Not available Not assessed Mean (S.D.) (7075) (6391) 509 (233) determined by multiplying the concentration of AP activity in the unprocessed ejaculate (IU/l) by the original total ejaculate volume (ml) and dividing by 1000 (Table 2). The concentration of AP activity in unprocessed ejaculates was compared to that in sperm-free semen plasma using the paired t-test. Concentration of and total AP activity in unprocessed ejaculates were compared between the stallions in this group and the stallions listed in Table 1 using the unpaired t-test. Normal tissue samples from testicle (n ˆ 4), epididymal tail (n ˆ 4) and ductus deferens (n ˆ 5) were obtained from horse stallions following routine castration. Normal tissue samples from ampulla, seminal vesicle, prostate, bulbourethral gland and urethra were obtained from horse and pony stallions following euthanasia (n ˆ 5 for each tissue type). All tissues were stored at 20 8C until processing. Before processing, tissue was thawed at room temperature. A section of each tissue weighing between 125 and 140mg was homogenized manually in a glass tissue grinder in 3 ml of 0.1% Triton X-100. The resulting uid was centrifuged at 16,000 g for 20min. The supernatant was removed from the pelleted cellular debris and assayed for the concentration of AP activity as described previously. Secretions were aspirated or manually expressed from the lumina of the epididymal tail (n ˆ 4), ampulla (n ˆ 3) and seminal vesicle (n ˆ 4) of normal horse and pony stallions following euthanasia. Samples were stored at 4 8C for no more than 12 h. The concentration of AP activity was determined as described above. Signi cant differences in mean concentration of AP activity among the various tissue and luminal uid samples were determined using ANOVA with the Tukey±Kramer HSD Correction Determination of AP activity in a stallion with blocked ampullae An 11-year-old Morgan stallion presented to our clinic for evaluation of a sudden onset of infertility and azoospermia. This stallion was diagnosed with bilaterally blocked ampullae using the criteria established by Love et al. [21] and based on response to
5 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 5 treatment. The stallion's testicles were subjectively soft on palpation and total scrotal width was 7.8 cm (slightly less than the width of 8.0cm recommended by the Society for Theriogenology for classi cation as a Satisfactory Prospective Breeder [18]). Ultrasonographic examination of the testicles revealed no abnormalities. Pre-ejaculatory uid and ejaculate samples were collected prior to relief of the blockage and an ejaculate sample was obtained immediately following relief of the blockage. The concentration of AP activity was determined in each sample as described above. 3. Results 3.1. Alkaline phosphatase activity in pre-ejaculatory uid and ejaculate samples from normal stallions The concentration of AP activity in ejaculate samples was signi cantly higher than AP activity in pre-ejaculatory uid samples (P < 0:001, Table 1). Mean concentration of AP activity in normal stallion pre-ejaculatory uid was IU/l while that in ejaculate samples was 24,185 12; 398 IU/l. The range of AP activity concentration in preejaculatory uid samples was 10±90 IU/l while that in ejaculate samples was 6390± 48,700 IU/l. Mean total AP activity in normal ejaculates was IU. The range of total AP activity in normal ejaculates was 595±3409 IU Alkaline phosphatase activity in unprocessed ejaculates and sperm-free semen plasma from normal stallions Mean concentration of AP activity in the 11 unprocessed ejaculates from this group was 15, IU/l. Values ranged from 3574 to 22,180IU/l. Mean concentration of AP activity in sperm-free semen plasma from the stallions in this group was 15, IU/ l. These values ranged from 1640to 24,112 IU/l. The concentration of AP activity in unprocessed ejaculate samples was not signi cantly different from that in sperm-free semen plasma (P ˆ 0:5, Table 2). Similarly, total AP activity in unprocessed ejaculate samples and sperm-free semen plasma did not differ signi cantly (P ˆ 0:97, data not shown). The mean total ejaculate AP activity was IU. Concentration of AP activity in the unprocessed ejaculates from the stallions in this group was not signi cantly different from that in the ejaculates from the nine stallions in the group reported in Table 1 (P > 0:05). However, total ejaculate AP activity was signi cantly lower in this second group of stallions compared to the stallions listed in Table 1 (P < 0:05) Alkaline phosphatase activity in tissue and luminal uid samples from the reproductive tracts of normal stallions Mean AP activities in tissue and luminal uid samples from normal stallion reproductive tracts are shown in Table 3. Extremely high levels of AP activity were identi ed in luminal uid from the epididymal tail (mean AP activity 557, ; 066 IU/l). Although AP
6 Bulbourethral gland tissue Prostatic tissue 17 9 Seminal vesicular gland tissue 4040 Ampullary tissue Ductus deferens tissue Urethral tissue 4022 Testicular tissue Epididymal tail tissue Seminal vesicular gland fluid Ampullary fluid Epididymal tail fluid R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 Table 3 Alkaline phosphatase (AP) activity in tissue and fluid samples from normal stallions Tissue Mean AP activity (IU/l) S.D. activity in ampullary lumen uid was still high (mean 35; ; 079 IU/l) compared to semen vesicular uid (mean IU/l), it was noticeably lower than that in the lumen of the epididymal tail. Alkaline phosphatase activity in testis and epididymal tail tissue (mean concentration of AP activity and IU/l, respectively) was signi cantly higher than AP activity in the other reproductive tract tissues (P < 0:05) Alkaline phosphatase activity in ejaculate samples from a stallion with blocked ampullae Alkaline phosphatase activity was 18 IU/l in one pre-ejaculate and one ejaculate sample collected from a stallion with bilaterally blocked ampullae. These values are similar to the AP activity in normal pre-ejaculatory uid and are much lower than the AP activity in normal ejaculates. Following relief of the blockage (as evidenced by superphysiologic sperm numbers and a high percentage of detached heads), the concentration of AP activity in the ejaculate rose to 4855 IU/l (total AP activity in the ejaculate was 97 IU). 4. Discussion 4.1. High levels of AP activity are present in the normal stallion ejaculate Biochemical analysis of the ejaculates of normal stallions reveals very high levels of AP activity. These values are signi cantly higher than those found in pre-ejaculatory uid or than those reported for normal horse serum (normal serum AP activity range for our lab is 109±315 IU/l). SPAP activity was determined in two separate groups of normal stallions. SPAP concentration was not signi cantly different between these two groups (P > 0:05). However, total AP activity was signi cantly different between the two groups (P <0.05). This difference is most likely due to lower ejaculate volumes in the stallions in Group 2 (7 of the 11 stallions in Group 2 were ponies, while only two of the nine stallions in Group 1 were ponies). Total AP activity is a function of both concentration and volume and, since
7 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 7 pony stallions typically have lower ejaculate volumes than horse stallions [20], it might be expected that total AP activity in pony stallion ejaculates would be lower than that in horse stallion ejaculates The major source of SPAP activity is not spermatozoa An experiment was designed to determine how much of the SPAP activity was contributed by sperm cells. AP activity in unprocessed ejaculates did not differ signi cantly from AP activity in sperm-free semen plasma, indicating that the vast majority of AP activity in the stallion ejaculate originates from the semen plasma and not from the spermatozoa. This is consistent with studies in men in which AP activity in isolated spermatozoa was very low compared to activity in semen plasma [22] The highest tissue levels of AP activity in the stallion reproductive tract are found in the epididymis and the testicle Our data indicate that in normal stallions AP activity levels in the testis and epididymis are signi cantly higher than in other reproductive tract tissues. Additionally, our data suggest that AP activity is concentrated to extremely high levels in the lumen of the epididymal tail. After leaving the epididymal tail, AP activity is diluted to ejaculate levels probably as a result of the addition of secretions from the accessory glands. These ndings are similar to those found in dogs and rabbits [7,10] but differ from ndings in men [2] and bulls [13]. Because the highest levels of AP activity apparently originate from the testicle and epididymis in stallions, SPAP activity may be a useful marker for the presence of epididymal or testicular secretions (i.e., a marker for ejaculation). Because spermatozoa do not contribute signi cantly to SPAP activity, SPAP activity can be used as an ejaculatory marker which is independent of the presence or number of spermatozoa Bilateral blockage of the ampullae resulted in baseline SPAP activity In one stallion with bilaterally blocked ampullae, AP activity was similar in preejaculatory uid and ejaculate samples and was similar to AP activity in pre-ejaculatory uid from normal stallions. After resolution of the blockage, SPAP concentration rose to levels similar to those found in ejaculatory uid of normal stallions. The change in SPAP activity from baseline during the blockage (when contributions from the testicles and epididymides were prevented from reaching the ejaculate) to levels similar to those in the ejaculates of normal stallions following relief of the blockage (when contributions from the testicles and epididymides were contained in the ejaculate) strongly supports the hypothesis that the testicles and epididymides are the major sources of SPAP activity in the stallion. This is consistent with our ndings that the highest levels of AP activity in the reproductive tracts of normal stallions are found in the testicles and epididymides. Taken together, this information suggests that measurement of SPAP activity in stallions with azoospermia will be very useful in differentiating stallions with azoospermia resulting from ampullary blockage (good prognosis for future fertility) and those with azoospermia resulting from testicular abnormalities (poor prognosis for future fertility).
8 8 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 It should be noted that even after relief of the blockage, total SPAP activity in the blocked stallion (97 IU) remained lower than those values reported for normal stallions (range 595± 3409 IU). Nonetheless, it remained signi cantly higher than total SPAP activity in preejaculatory uid (data not shown). In this regard, even after complete resolution of the blockage, semen parameters for this stallion were well below those values required for classi cation as a Satisfactory Prospective Breeder [18]. SPAP levels were not available for subsequent ejaculates. Due to marginal semen quality and problems with recurrent ampullary blockage, this stallion was gelded at the owner's request. Histopathologic examination of the testicles revealed mild to moderate testicular degeneration. Since in normal stallions the testicles contain high levels of AP activity, it is possible that the testicular pathology present in this stallion may have resulted in a reduction in SPAP activity, even after relief of the blockage. In support of this hypothesis, we have recently examined SPAP activity in several stallions with testicular pathologies. In these stallions, preliminary data suggests that SPAP activity remains signi cantly elevated above preejaculate levels (P < 0:001) but is consistently lower than SPAP activity in normal stallions (P < 0:01) [23] Seminal plasma alkaline phosphatase as a clinical marker for ejaculation These results expand on previously reported data [17] and, together with the ready availability of inexpensive clinical assays for AP activity, suggest that SPAP activity will be useful clinically as a marker for ejaculation in stallions. SPAP activity may be useful, for example, for differentiating true azoospermia (high SPAP, no spermatozoa) from ejaculation failure (low SPAP, no spermatozoa) or azoospermia resulting from ampullary blockage (low SPAP, no spermatozoa). In the future, it will be important to examine SPAP activity in stallions with testicular origin azoospermia and oligospermia. Since the testis is a signi cant source of SPAP activity, it is possible that testicular pathology resulting in low sperm numbers also could result in reductions in SPAP activity. This may have been the case for the stallion with blocked ampullae in this report. Additionally, it has been shown in bulls that production of SPAP is, at least in part, androgen dependent [24]. If the same is true in stallions, then hormonal aberrations also could result in secondary reductions in SPAP. In spite of this, our preliminary data suggests that, even in stallions with testicular abnormalities, SPAP activity remains signi cantly higher than levels found in preejaculatory uid Normal values for AP in pre-ejaculatory and ejaculatory uid The work reported here de nes a starting point for the clinical use of SPAP activity as a marker for ejaculation in stallions. Based on the population of normal horse and pony stallions described in this manuscript, initial ranges for normal pre-ejaculatory uid and ejaculate AP activities are: AP concentration in pre-ejaculatory uid (n ˆ 9): 10±90 IU/l. AP concentration in ejaculate (n ˆ 20): 1640±48,700 IU/l. Total AP in ejaculate (n ˆ 17): 293±3409 U.
9 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 9 From these values, it is likely that AP concentrations under 100 IU/l in an unprocessed sample indicate either that ejaculation did not occur (i.e., pre-ejaculatory uid, ejaculatory dysfunction, false mount) or that epididymal and testicular secretions are not present in the ejaculate (i.e., ampullary blockage). Conversely, AP concentrations over 1000 IU/l or total AP activity over 200 IU indicates that complete ejaculation occurred (i.e., the sample contains epididymal and testicular secretions). Because of the large standard deviations observed in AP concentrations in ejaculates from normal stallions, evaluation of total AP activity rather than AP concentration may prove to be more useful once the in uence of stallion size and ejaculate volume is better de ned. The clinical signi cance of AP concentrations greater than 100 IU/l and less than 1000 IU/l is not yet known. If values within this range are obtained, it may be bene cial to repeat the assay on additional samples to better establish AP concentration. Further studies on subfertile and infertile stallions are warranted as it is possible that AP concentrations between 100 and 1000 IU/l may be indicative of hormonal abnormalities or testicular and epididymal pathology. Acknowledgements The authors thank Dr. Laura Diaz-Cueto for assistance with statistical analyses, and Drs. Eric Twitchell and Perry Habecker for assistance in obtaining and processing tissue samples. Thanks to Dr. Shirley Johnston for suggesting the project. References [1] Bell DJ, Lake PE. A comparison of phosphomonoesterase activities in the seminal plasma of the domestic cock, turkey, tom, boar, bull, buck, rabbit and man. J Fertil 1962;3:262±8. [2] Mann T. The biochemistry of semen. London: Methuen and Co. Ltd., [3] Abdou MS, el-guindi MM, Mostafa MA, el-wishy AB, Farahat AA. Comparative study of the phosphatase activity in the semen of bovines (Bos bubalis and Bos taurus) in Egypt. Zentralbl Veterinarmed [A] 1974;21:759±67. [4] El-Naggar VMA. Comparison of phosphatase activity in the seminal plasma of different types of domestic livestock in Egypt. Deutsche Tierarztliche Wochenschrift 1975;82:322±4. [5] Dubiel A. Evaluation of semen properties and ejaculation re ex in dogs with reference to fertility. In: Proceedings of the VIIIth International Congress on Animal Reproduction and Arti cial Insemination, p. 75. [6] James RW, Heywood R. Biochemical observations on beagle dog semen. Vet Rec 1979;104:480±2. [7] Dubiel A, Krolinski J, Karpiak C. Activity of gamma-glutamyltransferase, alkaline phosphatase and aspartate aminotransferase in the ejaculates of rabbits before and after elimination of the testes, epididymides and vasa deferentia secretions. Medycyna Weterynaryjna 1982;38:608±10. [8] Keller VP, Freudiger U. Enzyme activities in urine, cerebrospinal uid, bile, saliva and ejaculate of dogs. Kleintier Praxis 1984;29:15±34. [9] Mattheeuws DR, Verhaeghe BJ, Borghijs H, Comhaire F. The signi cance of alkaline phosphatase activity in dog semen and accessory sex glands. Proceedings of the IVth Congress of the International Society for Animal Clinical Biochemistry, p. 171±8. [10] Frenette G, Dube JY, Tremblay RR. Origin of alkaline phosphatase of canine seminal plasma. Arch Androl 1986;16:235±41.
10 10 R.M.O. Turner, S.M. McDonnell / Theriogenology 60 (2003) 1±10 [11] Johnson WL, Hunter AG. Distribution of seminal antigens and enzymes in the rabbit ductus epididymis. J Anim Sci 1979;49:143±50. [12] Muller B. Genital tract proteins in the male rabbit II: alkaline phosphataseðenzyme action and site of synthesis. Andrologia 1983;15:676±81. [13] Alexander FC, Zemjanis R, Graham EF, Schmehl ML. Semen characteristics and chemistry from bulls before and after seminal vesiculectomy and after vasectomy. J Dairy Sci 1971;54:1530±5. [14] Mann T. The biochemistry of semen and of the male reproductive tract. London: Methuen and Co. Ltd., [15] Olson PN. Clinical approach to evaluating dogs with azoospermia or aspermia. Vet Clin North Am 1991;21:591±608. [16] Szumowski P. Physiopathology of the secretory functions of the genital glands involved in the formation of the stallion ejaculate. In: Proceedings of the VIIth International Congress on Animal Reproduction and Arti cial Insemination, Munich, p [17] Turner RMO. Alkaline phosphatase in stallion semen: characterization and clinical applications. In: Society for Theriogenology Annual Meeting p. 284±93. [18] Kenney RM, Hurtgen JP, Pierson RH, Witherspoon D, Simons J. Clinical fertility evaluation of the stallion. J Soc Theriogenol 1983;9:7±62. [19] Love CC, Garcia MC, Riera FR, Kenney RM. Evaluation of measures taken by ultrasonography and caliper to estimate testicular volume and predict daily sperm output in the stallion. J Reprod Fertil Suppl 1991;44:99±105. [20] McDonnell SM, Love CC. Manual stimulation collection of semen from stallions: training time, sexual behavior and semen. Theriogenology 1990;33:1201±10. [21] Love CC, Riera FL, Turner RMO. Sperm occluded (plugged) ampullae in the stallion. In: Proceedings of the Annual Meeting of the Society for Theriogenology, p. 117±25. [22] Singer R, Barnet M, Allalouf D, Schwartzman S, Sagiv M, Landau B, et al. Some properties of acid and alkaline phosphatase in seminal uid and isolated sperm. Arch Androl 1980;5:195±9. [23] Turner RMO, Sertich PL. Use of alkaline phosphatase activity as a diagnostic tool in stallions with azoospermia and oligospermia. In: Third International Symposium on Stallion Reproduction, Fort Collins, CO p. 35. [24] Goyal HO, Vig MM. Histochemical activity of alkaline phosphatase and acid phosphatase in the epididymis of mature intact and androgen-deprived bulls. Am J Vet Res 1996;45:444±50.
How to Investigate Azoospermia in Stallions
 How to Investigate Azoospermia in Stallions Terry L. Blanchard, DVM, MS, Diplomate ACT; Steven P. Brinsko, DVM, MS, PhD, Diplomate ACT; Dickson D. Varner, DVM, MS, Diplomate ACT; and Charles C. Love, DVM,
How to Investigate Azoospermia in Stallions Terry L. Blanchard, DVM, MS, Diplomate ACT; Steven P. Brinsko, DVM, MS, PhD, Diplomate ACT; Dickson D. Varner, DVM, MS, Diplomate ACT; and Charles C. Love, DVM,
Male Reproductive System Dr. Gary Mumaugh
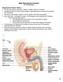 Male Reproductive System Dr. Gary Mumaugh Reproductive System Basics Primary sex organs (gonads) testes in males, ovaries in females Gonads produce sex cells called gametes (gametes means spouses) and
Male Reproductive System Dr. Gary Mumaugh Reproductive System Basics Primary sex organs (gonads) testes in males, ovaries in females Gonads produce sex cells called gametes (gametes means spouses) and
Correlations between seminal plasma enzyme activities and semen parameters in seminal fluid of Arabian horses
 Correlations between seminal plasma enzyme activities and semen parameters in seminal fluid of Arabian horses Dogan, I. 1* ; Polat, U. 2 and Nur, Z. 1 1 Department of Reproduction and Artificial Insemination,
Correlations between seminal plasma enzyme activities and semen parameters in seminal fluid of Arabian horses Dogan, I. 1* ; Polat, U. 2 and Nur, Z. 1 1 Department of Reproduction and Artificial Insemination,
Small Ruminant Reproductive Management Workshop
 Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
Male Reproductive System
 Male Reproductive System The male reproductive system consists of a number of sex organs that are part of the reproductive process. The following sections describe the function of each part of the male
Male Reproductive System The male reproductive system consists of a number of sex organs that are part of the reproductive process. The following sections describe the function of each part of the male
Study Regarding Age-Related Morphometric Features of Buffalo Sperm
 Available online at www.sciencedirect.com ScienceDirect Agriculture and Agricultural Science Procedia 6 ( 2015 ) 272 276 ST26733, International Conference "Agriculture for Life, Life for Agriculture" Study
Available online at www.sciencedirect.com ScienceDirect Agriculture and Agricultural Science Procedia 6 ( 2015 ) 272 276 ST26733, International Conference "Agriculture for Life, Life for Agriculture" Study
Animal Science 434. Semen Collection. Effect of Age on Sperm Output. Age When Semen Can Be Collected. Text: Ch. 10 and 11. Sexual Behavior (cont.
 Animal Science 434 Age When Semen Can Be Collected Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11 Bull Boar Ram Stallion Dog 12
Animal Science 434 Age When Semen Can Be Collected Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11 Bull Boar Ram Stallion Dog 12
Ultrasonographic Measurement of Stallion Accessory Sex Glands and Excurrent Ducts during Seminal Emission and Ejaculation'
 BOLOGY OF REPRODUCTON 49 267-273 (1993) Ultrasonographic Measurement of Stallion Accessory Sex Glands and Excurrent Ducts during Seminal Emission and Ejaculation' JAMES A WEBER and GORDON L WOODS 2 Northwest
BOLOGY OF REPRODUCTON 49 267-273 (1993) Ultrasonographic Measurement of Stallion Accessory Sex Glands and Excurrent Ducts during Seminal Emission and Ejaculation' JAMES A WEBER and GORDON L WOODS 2 Northwest
Animal Science 434" Semen Collection" Effect of Age on Sperm Output" Age When Semen Can Be Collected" Text: Ch. 10 and 11"
 Animal Science 434" Age When Semen Can Be Collected" Lecture 15b: Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11" "Bull "Boar
Animal Science 434" Age When Semen Can Be Collected" Lecture 15b: Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11" "Bull "Boar
The Reproductive System
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Reproductive System 16PART A The Reproductive System Gonads primary sex organs Testes in males
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Reproductive System 16PART A The Reproductive System Gonads primary sex organs Testes in males
The Repr duct ve System. Function: producing offspring
 The Repr duct ve System Function: producing offspring Anatomy of male reproductive system Location: The reproductive organs are classified as external and internal genitalia. The external genitalia are
The Repr duct ve System Function: producing offspring Anatomy of male reproductive system Location: The reproductive organs are classified as external and internal genitalia. The external genitalia are
Getting Help for Obstructive Azoospermia A BASIC GUIDE TO MALE. A doctor s guide for patients developed by the American Urological Association, Inc.
 A BASIC GUIDE TO MALE Getting Help for Obstructive Azoospermia A doctor s guide for patients developed by the American Urological Association, Inc. Based on the AUA Best Practice Policy and ASRM Practice
A BASIC GUIDE TO MALE Getting Help for Obstructive Azoospermia A doctor s guide for patients developed by the American Urological Association, Inc. Based on the AUA Best Practice Policy and ASRM Practice
STRUCTURE AND FUNCTION OF THE MALE REPRODUCTIVE SYSTEM
 Unit 7A STRUCTURE AND FUNCTION OF THE MALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the male reproductive system. 2. Learn the functions of the male reproductive system. 3. Learn
Unit 7A STRUCTURE AND FUNCTION OF THE MALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the male reproductive system. 2. Learn the functions of the male reproductive system. 3. Learn
Male Reproductive System. Dr Maan Al-Abbasi PhD, MSc, MBChB, MD
 Male Reproductive System Dr Maan Al-Abbasi PhD, MSc, MBChB, MD Learning Objectives 1. Describe the General Anatomy of the Male Reproductive System 2. Identify the structures that are related to the prostate.
Male Reproductive System Dr Maan Al-Abbasi PhD, MSc, MBChB, MD Learning Objectives 1. Describe the General Anatomy of the Male Reproductive System 2. Identify the structures that are related to the prostate.
Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization
 Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization may occur! A. Scrotum 1. Muscular pouch that holds the
Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization may occur! A. Scrotum 1. Muscular pouch that holds the
S S GURAYA and ARBANS J KAUR Department of Zoology, College of Basic Sciences and Humanities, Punjab Agricultural University, Ludhiana , India
 Proc. Indian Acad, Sci. (Anim. Sci.), Vol. 90, Number 5, SeptcIl1ber 1981, pp. 49S-soi. o Printed in India. Effect of castration on some hydrolytic enzymes in the rat vas deferens: A histochemical study
Proc. Indian Acad, Sci. (Anim. Sci.), Vol. 90, Number 5, SeptcIl1ber 1981, pp. 49S-soi. o Printed in India. Effect of castration on some hydrolytic enzymes in the rat vas deferens: A histochemical study
Primary sex organs (gonads): testes and ovaries. Accessory reproductive organs: ducts, glands, and external genitalia
 Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
- production of two types of gametes -- fused at fertilization to form zygote
 Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
Male Reproductive System. Anatomy
 Male Reproductive System Medical Terminology Chapter Seven HIT # 141 Anatomy Testis or testicle = main male sex organs, paired, oval shaped, enclosed in a sac called the scrotum. Seminiferous tubules =
Male Reproductive System Medical Terminology Chapter Seven HIT # 141 Anatomy Testis or testicle = main male sex organs, paired, oval shaped, enclosed in a sac called the scrotum. Seminiferous tubules =
Male Reproductive System
 21-1 21-2 Reproductive System Male Reproductive System Genital Tract In males the testes, held outside the body in the scrotum (optimum temp of about 35 0 C), produce sperm. Sperm mature in coiled tubes
21-1 21-2 Reproductive System Male Reproductive System Genital Tract In males the testes, held outside the body in the scrotum (optimum temp of about 35 0 C), produce sperm. Sperm mature in coiled tubes
The Reproductive System
 Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 16 The Reproductive System Slides 16.1 16.20 Lecture Slides in PowerPoint by Jerry L. Cook The Reproductive System Gonads
Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 16 The Reproductive System Slides 16.1 16.20 Lecture Slides in PowerPoint by Jerry L. Cook The Reproductive System Gonads
Student Academic Learning Services Page 1 of 5 Reproductive System Practice
 Student Academic Learning Services Page 1 of 5 Reproductive System Practice Fill in the blanks using the words listed: accessory glands, vas deferens, penis, scrotum, fallopian tube, testes, urethra, vagina,
Student Academic Learning Services Page 1 of 5 Reproductive System Practice Fill in the blanks using the words listed: accessory glands, vas deferens, penis, scrotum, fallopian tube, testes, urethra, vagina,
The Reproductive System
 16 PART A The Reproductive System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Reproductive
16 PART A The Reproductive System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Reproductive
Seminal fluid analysis
 What is semen? Semen is the fluid formed at ejaculation. Made of secretions of all the accessory glands of the male genital tract and testicular sperm component Semen quality is maintained by all the accessory
What is semen? Semen is the fluid formed at ejaculation. Made of secretions of all the accessory glands of the male genital tract and testicular sperm component Semen quality is maintained by all the accessory
A PPRAISAL OF MALE FERTILITY has mainly been based on examination of
 Biochemical Analyses of Human Semen the Study of the Physiology and Pathophysiology of the Male Accessory Genital Glands. Ln RUNE ELIASSON, M.D. A PPRAISAL OF MALE FERTILITY has mainly been based on examination
Biochemical Analyses of Human Semen the Study of the Physiology and Pathophysiology of the Male Accessory Genital Glands. Ln RUNE ELIASSON, M.D. A PPRAISAL OF MALE FERTILITY has mainly been based on examination
Student Learning Guide Health Related Technology. Basic Structure of the Human Body. Course Title: Anatomy and Physiology
 Student Learning Guide Health Related Technology Basic Structure of the Human Body Name: Date: Course Title: Anatomy and Physiology Duty/Unit: Basic structure of the Human Body Objective: Upon completion
Student Learning Guide Health Related Technology Basic Structure of the Human Body Name: Date: Course Title: Anatomy and Physiology Duty/Unit: Basic structure of the Human Body Objective: Upon completion
Reproductive System Purpose General Structures Male Structures Functions Female Anatomy Structures Functions Clinical Applications
 The Reproductive System: Male, Ch 23 Outline of class lecture After studying the male reproductive system you should be able to: 1. Define the purpose of reproduction and identify the general organs of
The Reproductive System: Male, Ch 23 Outline of class lecture After studying the male reproductive system you should be able to: 1. Define the purpose of reproduction and identify the general organs of
The Reproductive System. Presenter: Dr. Jim Hurrell
 The Reproductive System Presenter: Dr. Jim Hurrell A Warm Welcome from My Faculty TEAM and Me!!! 2 The Pledge of Allegiance 3 Veterinary Technician/NURSE Oath 4 5 Come Hang Out with Us in Our Awesome Facebook
The Reproductive System Presenter: Dr. Jim Hurrell A Warm Welcome from My Faculty TEAM and Me!!! 2 The Pledge of Allegiance 3 Veterinary Technician/NURSE Oath 4 5 Come Hang Out with Us in Our Awesome Facebook
Study Guide Answer Key Reproductive System
 Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
Male Reproductive Physiology
 Male Reproductive Physiology Overview Anatomy Function Endocrine and spermatogenesis Testis epididymus,vas deferens,seminal vesicles and prostate Hypothalamic pituitary testicular axis Hormones of the
Male Reproductive Physiology Overview Anatomy Function Endocrine and spermatogenesis Testis epididymus,vas deferens,seminal vesicles and prostate Hypothalamic pituitary testicular axis Hormones of the
There is scanty information on the biochemistry
 FERTILITY AND STERILITY Copyright 1973 by The Williams & Wilkins Co. Vol. 24, No.4, April 1973 Printed in U.S.A. ACTIVITY AND LOCALIZATION OF ISOCITRIC DEHYDROGENASE, ASPARTATE AMINOTRANSFERASE, ALANINE
FERTILITY AND STERILITY Copyright 1973 by The Williams & Wilkins Co. Vol. 24, No.4, April 1973 Printed in U.S.A. ACTIVITY AND LOCALIZATION OF ISOCITRIC DEHYDROGENASE, ASPARTATE AMINOTRANSFERASE, ALANINE
THE SPERMATIC FLUID of a normal man consists predominantly of the secretions
 The ph of Human Ejaculate JAN RABOCH, M.D., and J. skachov A, RNDr. THE SPERMATIC FLUID of a normal man consists predominantly of the secretions of the seminal vesicles and of the prostatic gland. According
The ph of Human Ejaculate JAN RABOCH, M.D., and J. skachov A, RNDr. THE SPERMATIC FLUID of a normal man consists predominantly of the secretions of the seminal vesicles and of the prostatic gland. According
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
describe the parts and function of semen and the glands that contribute to it
 You need to be able to: describe spermatogenesis (How is sperm made?) describe the anatomy of a sperm describe the parts and function of semen and the glands that contribute to it How is sperm made? Spermatogenesis
You need to be able to: describe spermatogenesis (How is sperm made?) describe the anatomy of a sperm describe the parts and function of semen and the glands that contribute to it How is sperm made? Spermatogenesis
Male Reproduction Organs. 1. Testes 2. Epididymis 3. Vas deferens 4. Urethra 5. Penis 6. Prostate 7. Seminal vesicles 8. Bulbourethral glands
 Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Examining Breeding Soundness of Beef Bulls
 Examining Breeding Soundness of Beef Bulls A herd bull that will serve a higher percentage of cows during a limited breeding season is essential to a successful cow-calf operation. In many of these operations,
Examining Breeding Soundness of Beef Bulls A herd bull that will serve a higher percentage of cows during a limited breeding season is essential to a successful cow-calf operation. In many of these operations,
Unit B Understanding Animal Body Systems. Lesson 7 Understanding Animal Reproduction
 Unit B Understanding Animal Body Systems Lesson 7 Understanding Animal Reproduction 1 Terms Anestrus Artificial insemination Castration Cervix Copulation Diestrus Egg Ejaculation Estrous cycle Estrus Fertilization
Unit B Understanding Animal Body Systems Lesson 7 Understanding Animal Reproduction 1 Terms Anestrus Artificial insemination Castration Cervix Copulation Diestrus Egg Ejaculation Estrous cycle Estrus Fertilization
Unit B Understanding Animal Body Systems. Lesson 6 Anatomy and Physiology of Animal Reproduction Systems
 Unit B Understanding Animal Body Systems Lesson 6 Anatomy and Physiology of Animal Reproduction Systems 1 Terms Alimentary canal Bladder Cervix Clitoris Cloaca Copulation Cowper s gland Epididymis Fallopian
Unit B Understanding Animal Body Systems Lesson 6 Anatomy and Physiology of Animal Reproduction Systems 1 Terms Alimentary canal Bladder Cervix Clitoris Cloaca Copulation Cowper s gland Epididymis Fallopian
Male and Female Reproduction
 Male and Female Reproduction Similarities / Differences Develop very similar structures of sex glands Two genital tubes Manufacture the hormones of the other sex. Hormones of one sex are often used to
Male and Female Reproduction Similarities / Differences Develop very similar structures of sex glands Two genital tubes Manufacture the hormones of the other sex. Hormones of one sex are often used to
Breeding Soundness Evaluation (BSE) of Bulls
 Breeding Soundness Evaluation (BSE) of Bulls Beef Bull Evaluation Reproductive merit economically > important than growth and carcass quality Evaluate Before purchase Before each breeding season If infertility
Breeding Soundness Evaluation (BSE) of Bulls Beef Bull Evaluation Reproductive merit economically > important than growth and carcass quality Evaluate Before purchase Before each breeding season If infertility
Reproductive System. Where it all begins
 Reproductive System Where it all begins When it comes the reproductive anatomy of my gender, I would rate my knowledge (1 very poor, 10 excellent) When it comes the reproductive anatomy of the opposite
Reproductive System Where it all begins When it comes the reproductive anatomy of my gender, I would rate my knowledge (1 very poor, 10 excellent) When it comes the reproductive anatomy of the opposite
Male reproduction. Cross section of Human Testis ผศ.ดร.พญ.ส ว ฒณ ค ปต ว ฒ ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล 1. Aims
 Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chris Davies & Greg Handley
 Chris Davies & Greg Handley Contents Definition Epidemiology Aetiology Conditions for pregnancy Female Infertility Male Infertility Shared infertility Treatment Definition Failure of a couple to conceive
Chris Davies & Greg Handley Contents Definition Epidemiology Aetiology Conditions for pregnancy Female Infertility Male Infertility Shared infertility Treatment Definition Failure of a couple to conceive
Efferent Ducts and Epididymis
 increase) the secretion of each of the androgen regulated proteins. Regulation of spermatogenesis is therefore an extremely complex cascade of cell-cell interactions with the Leydig cells supporting germ
increase) the secretion of each of the androgen regulated proteins. Regulation of spermatogenesis is therefore an extremely complex cascade of cell-cell interactions with the Leydig cells supporting germ
The beginning of puberty is marked by the progressive increase in the production of sex hormones.
 Puberty is characterized by the changes that prepare the human body for the ability to reproduce. This stage generally occurs between the ages of 10 and 14 years old. The beginning of puberty is marked
Puberty is characterized by the changes that prepare the human body for the ability to reproduce. This stage generally occurs between the ages of 10 and 14 years old. The beginning of puberty is marked
Proceedings of the Society for Theriogenology 2012 Annual Conference
 www.ivis.org Proceedings of the Society for Theriogenology 2012 Annual Conference Aug. 20-25, 2012 Baltimore, MD, USA www.therio.org/ Next Meeting: Aug. 7-10, 2013 Louisville, KY, USA Reprinted in the
www.ivis.org Proceedings of the Society for Theriogenology 2012 Annual Conference Aug. 20-25, 2012 Baltimore, MD, USA www.therio.org/ Next Meeting: Aug. 7-10, 2013 Louisville, KY, USA Reprinted in the
Anatomy and Physiology of Farm Animals. Lecture Six. Anatomy and Physiology of Male Reproductive System II
 Anatomy and Physiology of Farm Animals Lecture Six Anatomy and Physiology of Male Reproductive System II 1 The male reproductive system Two testes Scrotum Spermatic cords Accessory glands Penis Prepuce
Anatomy and Physiology of Farm Animals Lecture Six Anatomy and Physiology of Male Reproductive System II 1 The male reproductive system Two testes Scrotum Spermatic cords Accessory glands Penis Prepuce
Chapter 22 The Reproductive System (I)
 Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Chapter 14 The Reproductive System
 Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Female and Male Reproductive Systems
 Female and Male Reproductive Systems Reproductive System: Organs that make possible the production of offspring. Female Reproductive System Female Reproductive System: Words to be familiar with ESTROGEN
Female and Male Reproductive Systems Reproductive System: Organs that make possible the production of offspring. Female Reproductive System Female Reproductive System: Words to be familiar with ESTROGEN
Semen evaluation in domestic animals I
 Reproductive Biotechnologies Andrology I I Semen evaluation in domestic animals I Prof. Alberto Contri Different aims 1. Diagnosis 2. Handling and preservation Related to the male - Breeding soundness
Reproductive Biotechnologies Andrology I I Semen evaluation in domestic animals I Prof. Alberto Contri Different aims 1. Diagnosis 2. Handling and preservation Related to the male - Breeding soundness
EFFECT OF MONTH AND STALLION ON SEMINAL CHARACTERISTICS AND SEXUAL BEHAVIOR 1, 2
 E EFFECT OF MONTH AND STALLION ON SEMINAL CHARACTERISTICS AND SEXUAL BEHAVIOR 1, 2 B. W. PICKETT, 3 L. C. FAULKNER 3 AND T. M. SUTHERLAND 4 Colorado State University, Fort Collim XTENSIVE studies have
E EFFECT OF MONTH AND STALLION ON SEMINAL CHARACTERISTICS AND SEXUAL BEHAVIOR 1, 2 B. W. PICKETT, 3 L. C. FAULKNER 3 AND T. M. SUTHERLAND 4 Colorado State University, Fort Collim XTENSIVE studies have
XVII Congresso Internazionale SIVE
 SOCIETÀ ITALIANA VETERINARI PER EQUINI SOCIETÀ FEDERATA ANMVI XVII Congresso Internazionale SIVE XVII SIVE International Congress Palacongressi d Abruzzo Montesilvano (PE) - ITALY 4-6 Febbraio 2011 February
SOCIETÀ ITALIANA VETERINARI PER EQUINI SOCIETÀ FEDERATA ANMVI XVII Congresso Internazionale SIVE XVII SIVE International Congress Palacongressi d Abruzzo Montesilvano (PE) - ITALY 4-6 Febbraio 2011 February
What You Need to Know
 UW MEDICINE PATIENT EDUCATION What You Need to Know Facts about male infertility This handout explains what causes male infertility, how it is diagnosed, and possible treatments. Infertility is defined
UW MEDICINE PATIENT EDUCATION What You Need to Know Facts about male infertility This handout explains what causes male infertility, how it is diagnosed, and possible treatments. Infertility is defined
Topic 18- Human Reproductive System. Day 2-Female (and review of) Male Reproductive Systems
 Topic 18- Human Reproductive System Day 2-Female (and review of) Male Reproductive Systems Bell Ringer (5 minutes) Bioblitz WHAT ARE WE LEARNING TODAY? Date: 12/14-12/15 Topic: Human Reproductive System
Topic 18- Human Reproductive System Day 2-Female (and review of) Male Reproductive Systems Bell Ringer (5 minutes) Bioblitz WHAT ARE WE LEARNING TODAY? Date: 12/14-12/15 Topic: Human Reproductive System
List of Equipment, Tools, Supplies, and Facilities:
 Unit B: Understanding Animal Body Systems Lesson 6: Anatomy and Physiology of Animal Reproductive Systems Student Learning Objectives: Instruction in this lesson should result in students achieving the
Unit B: Understanding Animal Body Systems Lesson 6: Anatomy and Physiology of Animal Reproductive Systems Student Learning Objectives: Instruction in this lesson should result in students achieving the
A USER S GUIDE WHAT EVERY MAN NEEDS TO KNOW
 A USER S GUIDE WHAT EVERY MAN NEEDS TO KNOW 1. Why men need to know more Good health is vital for a happy and full life. But, with work and family responsibilities, men often overlook their own health
A USER S GUIDE WHAT EVERY MAN NEEDS TO KNOW 1. Why men need to know more Good health is vital for a happy and full life. But, with work and family responsibilities, men often overlook their own health
Semen-induced ovulation in the bactrian camel (Camelus bactrianus)
 Semen-induced ovulation in the bactrian camel (Camelus bactrianus) B. X. Chen, Z. X. Yuen and G. W. Pan Department of Veterinary Medicine, Gansu Agricultural University, Wuwei, Gansu and *Haixi Institute
Semen-induced ovulation in the bactrian camel (Camelus bactrianus) B. X. Chen, Z. X. Yuen and G. W. Pan Department of Veterinary Medicine, Gansu Agricultural University, Wuwei, Gansu and *Haixi Institute
Imaging Ejaculatory Disorders and Hematospermia
 ATHENS 4-6 October 2018 European Society of Urogenital Radiology Imaging Ejaculatory Disorders and Hematospermia Parvati Ramchandani, MD Professor, Radiology and Surgery University of Pennsylvania Medical
ATHENS 4-6 October 2018 European Society of Urogenital Radiology Imaging Ejaculatory Disorders and Hematospermia Parvati Ramchandani, MD Professor, Radiology and Surgery University of Pennsylvania Medical
Human Anatomy Unit 3 REPRODUCTIVE SYSTEM
 Human Anatomy Unit 3 REPRODUCTIVE SYSTEM In Anatomy Today Male Reproductive System Gonads = testes primary organ responsible for sperm production development/maintenan ce of secondary sex characteristics
Human Anatomy Unit 3 REPRODUCTIVE SYSTEM In Anatomy Today Male Reproductive System Gonads = testes primary organ responsible for sperm production development/maintenan ce of secondary sex characteristics
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
Change in Morphology of Spermatozoa from Dismount Semen during the Breeding Season in Thoroughbred Stallions in Japan
 NOTE Change in Morphology of Spermatozoa from Dismount Semen during the Breeding Season in Thoroughbred Stallions in Japan Masanori KOYAGO 1, Ken NAKADA 1 *, Nobuo TSUNODA 2, Masaharu MORIYOSHI 1 and Yutaka
NOTE Change in Morphology of Spermatozoa from Dismount Semen during the Breeding Season in Thoroughbred Stallions in Japan Masanori KOYAGO 1, Ken NAKADA 1 *, Nobuo TSUNODA 2, Masaharu MORIYOSHI 1 and Yutaka
Chapter 14 Reproduction Review Assignment
 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Evaluation and treatment of ejaculatory duct obstruction in the infertile male
 FERTILITY AND STERILITY Copyright 99 The American Fertility Society Vol. 59, No,, February 99 Printed on acid4ree paper in U.S.A. Evaluation and treatment of ejaculatory duct obstruction in the infertile
FERTILITY AND STERILITY Copyright 99 The American Fertility Society Vol. 59, No,, February 99 Printed on acid4ree paper in U.S.A. Evaluation and treatment of ejaculatory duct obstruction in the infertile
LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM
 LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM Slide #101 (1096). Testis, rat. sustentacular ( Sertoli ) cells Nuclei of Sustentacular cells Leydig cells Spermatogonia Spermatocytes Spermatids pale
LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM Slide #101 (1096). Testis, rat. sustentacular ( Sertoli ) cells Nuclei of Sustentacular cells Leydig cells Spermatogonia Spermatocytes Spermatids pale
MALE REPRODUCTIVE SYSTEM
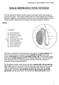 MALE REPRODUCTIVE SYSTEM The male reproductive system consists of primary sex organs (testes) and secondary or accessory sex organs. The secondary organs consist of a series of genital ducts (ductules
MALE REPRODUCTIVE SYSTEM The male reproductive system consists of primary sex organs (testes) and secondary or accessory sex organs. The secondary organs consist of a series of genital ducts (ductules
Bull BreedingSoundness Evaluation
 Bull BreedingSoundness Evaluation Scott Norman (BVSc, PhD, DipACT, GCEd) Registered Specialist in Veterinary Reproduction Associate Professor in Theriogenology, School of Animal and Veterinary Sciences
Bull BreedingSoundness Evaluation Scott Norman (BVSc, PhD, DipACT, GCEd) Registered Specialist in Veterinary Reproduction Associate Professor in Theriogenology, School of Animal and Veterinary Sciences
WJ UROGENITAL SYSTEM. UROLOGY
 WJ UROGENITAL SYSTEM. UROLOGY 1 Societies 11 History Include all aspects 13 Dictionaries. Encyclopaedias. Bibliographies Use for general works only. Classify with specific aspect where possible 15 Classification.
WJ UROGENITAL SYSTEM. UROLOGY 1 Societies 11 History Include all aspects 13 Dictionaries. Encyclopaedias. Bibliographies Use for general works only. Classify with specific aspect where possible 15 Classification.
ENZYMES IN THE SEMINAL PLASMA FROM AZOOSPERMIC MEN: CORRELATION WITH THE ORIGIN OF THEIR AZOOSPERMIA
 FERTILITY AND STERILITY Copyright C'1981 The American Fertility Society Vol. 36, No.3, September 1981 PrinU!d in UBA. ENZYMES IN THE SEMINAL PLASMA FROM AZOOSPERMIC MEN: CORRELATION WITH THE ORIGIN OF
FERTILITY AND STERILITY Copyright C'1981 The American Fertility Society Vol. 36, No.3, September 1981 PrinU!d in UBA. ENZYMES IN THE SEMINAL PLASMA FROM AZOOSPERMIC MEN: CORRELATION WITH THE ORIGIN OF
1. Describe the importance and process of animal reproduction. 2. List the sexual classification of animals for major species.
 Unit B: Understanding Animal Body Systems Lesson 7: Understanding Animal Reproduction Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives:
Unit B: Understanding Animal Body Systems Lesson 7: Understanding Animal Reproduction Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives:
Male Reproductive System
 Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
How to Obtain a Stallion Testicular Biopsy Using a Spring-Loaded Split-Needle Biopsy Instrument
 How to Obtain a Stallion Testicular Biopsy Using a Spring-Loaded Split-Needle Biopsy Instrument Lisa K. Pearson, DVM*; Jacobo S. Rodriguez, DVM, MS, Diplomate ACT; and Ahmed Tibary, DVM, MS, DSc, PhD,
How to Obtain a Stallion Testicular Biopsy Using a Spring-Loaded Split-Needle Biopsy Instrument Lisa K. Pearson, DVM*; Jacobo S. Rodriguez, DVM, MS, Diplomate ACT; and Ahmed Tibary, DVM, MS, DSc, PhD,
VETERINARY CONSIDERATIONS MALE BREEDING SOUNDNESS EXAMS AND
 VETERINARY CONSIDERATIONS MALE BREEDING SOUNDNESS EXAMS AND VENEREAL DISEASES IN BULLS Bret McNabb, DVM, MPVM, DACT University of California, Davis School of Veterinary Medicine Physical Examination A
VETERINARY CONSIDERATIONS MALE BREEDING SOUNDNESS EXAMS AND VENEREAL DISEASES IN BULLS Bret McNabb, DVM, MPVM, DACT University of California, Davis School of Veterinary Medicine Physical Examination A
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
The Male Reproductive System
 The Male Reproductive System The male reproductive system Testes Genital ducts Accessory sex glands: seminal vesicles prostate bulbourethral glands External genitalia: penis Structure of the Testis Tunica
The Male Reproductive System The male reproductive system Testes Genital ducts Accessory sex glands: seminal vesicles prostate bulbourethral glands External genitalia: penis Structure of the Testis Tunica
Chapter 26: Reproductive Systems. Male 11/29/2015. Male reproductive system is composed of... BIO 218 Fall Gonads (testes)
 Chapter 26: Reproductive Systems BIO 218 Fall 2015 Male Male reproductive system is composed of... Gonads (testes) Duct system (epididymis, ductus deferens, ejaculatory ducts, urethra) Accessory sex glands
Chapter 26: Reproductive Systems BIO 218 Fall 2015 Male Male reproductive system is composed of... Gonads (testes) Duct system (epididymis, ductus deferens, ejaculatory ducts, urethra) Accessory sex glands
Semen collection in domestic animals
 Reproductive Biotechnologies Andrology I Semen collection in domestic animals Prof. Alberto Contri Aims Technological aim Diagnostic aim Methods for semen collection Indirect collection post-coital method
Reproductive Biotechnologies Andrology I Semen collection in domestic animals Prof. Alberto Contri Aims Technological aim Diagnostic aim Methods for semen collection Indirect collection post-coital method
Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor)
 Indifferent ducts of embryo Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Y chromosome present Y chromosome absent Phenotypic sex is depends on development of external
Indifferent ducts of embryo Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Y chromosome present Y chromosome absent Phenotypic sex is depends on development of external
Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor)
 Indifferent ducts of embryo Y chromosome present Y chromosome absent Male Female penis ovary uterus vagina testis Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Phenotypic
Indifferent ducts of embryo Y chromosome present Y chromosome absent Male Female penis ovary uterus vagina testis Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Phenotypic
Reproductive system Presented by: Ms. Priya
 Reproductive system Presented by: Ms. Priya Reproductive system Female reproductive system has two functions: produce egg cells to protect and nourish the offspring (baby) until birth. Male reproductive
Reproductive system Presented by: Ms. Priya Reproductive system Female reproductive system has two functions: produce egg cells to protect and nourish the offspring (baby) until birth. Male reproductive
Science and Health THE HUMAN REPRODUCTIVE SYSTEM
 5 Module 1 Science and Health THE HUMAN REPRODUCTIVE SYSTEM Male Female A DepEd-BEAM Distance Learning Program supported by the Australian Agency for International Development NOTICE Section 9 of the Presidential
5 Module 1 Science and Health THE HUMAN REPRODUCTIVE SYSTEM Male Female A DepEd-BEAM Distance Learning Program supported by the Australian Agency for International Development NOTICE Section 9 of the Presidential
Appendix D Answers to the KAP Survey
 From Trainer s Resource Book to accompany Management of Men s Reproductive Health Problems 2003 EngenderHealth Appendix D Answers to the KAP Survey In the answer key that follows: The answers appear in
From Trainer s Resource Book to accompany Management of Men s Reproductive Health Problems 2003 EngenderHealth Appendix D Answers to the KAP Survey In the answer key that follows: The answers appear in
Chapter 22 Reproductive Systems. Male Reproductive Organs. Male Reproductive Organs. Specialized to produce, maintain the male sex cells (sperm)
 Chapter 22 Reproductive Systems Male reproductive organs 1 Male Reproductive Organs posterior view 2 Male Reproductive Organs Specialized to produce, maintain the male sex cells (sperm) Transport these
Chapter 22 Reproductive Systems Male reproductive organs 1 Male Reproductive Organs posterior view 2 Male Reproductive Organs Specialized to produce, maintain the male sex cells (sperm) Transport these
Male Anatomy. testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics
 Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Prostate Gland Disorders
 Prostate Gland Disorders THE PROSTATE GLAND A male s prostate gland is located in the floor of the pelvis surrounding the urethra between the bladder and the penis. The prostate is positioned immediately
Prostate Gland Disorders THE PROSTATE GLAND A male s prostate gland is located in the floor of the pelvis surrounding the urethra between the bladder and the penis. The prostate is positioned immediately
FEMALE REPRODUCTIVE SYSTEM
 FEMALE REPRODUCTIVE SYSTEM UTERUS (made up of muscular walls, a lining called the endometrium, and a cervix. The uterus is also called womb ) houses and protects embryo/fetus/baby allows nutrient & waste
FEMALE REPRODUCTIVE SYSTEM UTERUS (made up of muscular walls, a lining called the endometrium, and a cervix. The uterus is also called womb ) houses and protects embryo/fetus/baby allows nutrient & waste
RELATIONSHIPS BETWEEN SPERM MEMBRANE INTEGRITY AND OTHER SEMEN QUALITY CHARACTERISTICS OF THE SEMEN OF SAANEN GOAT BUCKS
 Bull Vet Inst Pulawy 49, 183-187, 2005 RELATIONSHIPS BETWEEN SPERM MEMBRANE INTEGRITY AND OTHER SEMEN QUALITY CHARACTERISTICS OF THE SEMEN OF SAANEN GOAT BUCKS ZEKARIYA NUR 1, IBRAHIM DOGAN 1, ULGEN GUNAY
Bull Vet Inst Pulawy 49, 183-187, 2005 RELATIONSHIPS BETWEEN SPERM MEMBRANE INTEGRITY AND OTHER SEMEN QUALITY CHARACTERISTICS OF THE SEMEN OF SAANEN GOAT BUCKS ZEKARIYA NUR 1, IBRAHIM DOGAN 1, ULGEN GUNAY
6.7 IN. Continuity through Reproduction. What are the differences between male and female gametes? Discuss their formation and physical attributes.
 6.7 IN What are the differences between male and female gametes? Discuss their formation and physical attributes. Males - 4 sperm per parent cell; Females - 1 ovum per parent cell Sperm - motile (tail);
6.7 IN What are the differences between male and female gametes? Discuss their formation and physical attributes. Males - 4 sperm per parent cell; Females - 1 ovum per parent cell Sperm - motile (tail);
To General Embryology Dr: Azza Zaki
 Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Male reproductive system The physiology of sexual act
 Male reproductive system The physiology of sexual act Gabriella Kékesi 65. The development and physiology of the male reproductive system. The physiology of the sexual act Define chromosomal, gonadal and
Male reproductive system The physiology of sexual act Gabriella Kékesi 65. The development and physiology of the male reproductive system. The physiology of the sexual act Define chromosomal, gonadal and
Chapter 7. Male Reproductive System
 Chapter 7 Male Reproductive System 1 Sexual Reproduction Sexual reproduction two individuals produce offspring that have genetic characteristics from both parents Sexual reproduction introduces new gene
Chapter 7 Male Reproductive System 1 Sexual Reproduction Sexual reproduction two individuals produce offspring that have genetic characteristics from both parents Sexual reproduction introduces new gene
DISINTEGRATION OF EPIDIDYMAL SPERMATOZOA BY APPLICATION OF ICE TO THE SCROTAL TESTIS
 [ DINTEGRATION OF EPIDIDYMAL SPERMATOZOA BY APPLICATION OF ICE TO THE SCROTAL TEST By MIN CHUEH CHANG, Ph.D. (Cantab.), School of Agriculture, Cambridge University (Received i May ) (With Four Text-figures)
[ DINTEGRATION OF EPIDIDYMAL SPERMATOZOA BY APPLICATION OF ICE TO THE SCROTAL TEST By MIN CHUEH CHANG, Ph.D. (Cantab.), School of Agriculture, Cambridge University (Received i May ) (With Four Text-figures)
Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature
 REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
Abnormalities of Spermatogenesis
 Abnormalities of Spermatogenesis Male Factor 40% of the cause for infertility Sperm is constantly produced by the germinal epithelium of the testicle Sperm generation time 73 days Sperm production is thermoregulated
Abnormalities of Spermatogenesis Male Factor 40% of the cause for infertility Sperm is constantly produced by the germinal epithelium of the testicle Sperm generation time 73 days Sperm production is thermoregulated
Male History, Clinical Examination and Testing
 Male History, Clinical Examination and Testing Dirk Vanderschueren, MD, PhD Case Jan is 29 years old and consults for 1 year primary subfertility partner 28 years old and normal gynaecological investigation
Male History, Clinical Examination and Testing Dirk Vanderschueren, MD, PhD Case Jan is 29 years old and consults for 1 year primary subfertility partner 28 years old and normal gynaecological investigation
to ensure the. Sexual reproduction requires the (from the mother) by a (from the father). Fertilization is the fusion of.
 The Reproductive System Fill-In Notes Purpose of life: to ensure the. Stages of Human Development Sexual reproduction requires the (from the mother) by a (from the father). Fertilization is the fusion
The Reproductive System Fill-In Notes Purpose of life: to ensure the. Stages of Human Development Sexual reproduction requires the (from the mother) by a (from the father). Fertilization is the fusion
Human Reproduction. Human Reproductive System. Scrotum. Male Reproductive System
 Human Reproductive System Human Reproduction Chapter 41 Contraceptives Scrotum Testes Epididymus Vas Deferens Seminal Vesicles Prostate Gland Bulbourethral Gland Penis Scrotum Sac of smooth muscle tissue
Human Reproductive System Human Reproduction Chapter 41 Contraceptives Scrotum Testes Epididymus Vas Deferens Seminal Vesicles Prostate Gland Bulbourethral Gland Penis Scrotum Sac of smooth muscle tissue
 MOCK TEST I (HUMAN REPRODUCTION) 1. In human transfer of sperms into female genital tract is called as 1) Fertilization 2) Implantation 3) Insemination 4) Gestation 2. In male human scrotum maintains the
MOCK TEST I (HUMAN REPRODUCTION) 1. In human transfer of sperms into female genital tract is called as 1) Fertilization 2) Implantation 3) Insemination 4) Gestation 2. In male human scrotum maintains the
Urinary System Chapter 16
 Urinary System Chapter 16 1 Urology- the branch of medicine that treats male and female urinary systems as well as the male reproductive system. Nephrology- the scientific study of the anatomy, physiology,
Urinary System Chapter 16 1 Urology- the branch of medicine that treats male and female urinary systems as well as the male reproductive system. Nephrology- the scientific study of the anatomy, physiology,
