Adverse effects of endocrine disruptors on the foetal testis development: focus on the phthalates
|
|
|
- Magdalene Lynch
- 6 years ago
- Views:
Transcription
1 FOLIA HISTOCHEMICA ET CYTOBIOLOGICA Vol. 47, No. 5, 2009 pp. S67-S74 Review article Adverse effects of endocrine disruptors on the foetal testis development: focus on the phthalates René Habert 1,2,3, Vincent Muczynski 1,2,3, Abdelali Lehraiki 1,2,3, Romain Lambrot 1,2,3, Charlotte Lécureuil 1,2,3, Christine Levacher 1,2,3, Hervé Coffigny 1,2,3, Catherine Pairault 1,2,3, Delphine Moison 1,2,3, René Frydman 4 and Virginie Rouiller-Fabre 1,2,3 1Laboratory of Differentiation and Radiobiology of the Gonads, CEA DSV / ircm / SCSR, F Fontenay aux Roses, France 2Université Paris Diderot-Paris7, F-92265, Fontenay aux Roses, France 3 INSERM, Unité 967, F-92265, Fontenay aux Roses, France 4 Service de Gynécologie-Obstétrique, Université Paris Sud, Hôpital A. Béclere, F Clamart, France Abstract: There are great concerns about the increasing incidence of abnormalities in male reproductive function. Human sperm counts have markedly dropped and the rate of testicular cancer has clearly augmented over the past four decades. Moreover, the prevalence rates of cryptorchidism and hypospadias are also probably increasing. It has been hypothesized that all these adverse trends in male reproduction result from abnormalities in the development of the testis during foetal and neonatal life. Furthermore, many recent epidemiological, clinical and experimental data suggest that these male reproductive disorders could be due to the effects of xenobiotics termed endocrine disruptors, which are becoming more and more concentrated and prevalent in our environment. Among these endocrine disruptors, we chose to focus this review on the phthalates for different reasons: 1) they are widespread in the environment; 2) their concentrations in many human biological fluids have been measured; 3) the experimental data using rodent models suggesting a reprotoxicity are numerous and are the most convincing; 4) their deleterious effects on the in vivo and in vitro development and function of the rat foetal testis have been largely studied; 5) some epidemiological data in humans suggest a reprotoxic effect at environmental concentrations at least during neonatal life. However, the direct effects of phthalates on human foetal testis have never been explored. Thus, as we did for the rat in the 1990s, we recently developed and validated an organ culture system which allows maintenance of the development of the different cell types of human foetal testis. In this system, addition of 10-4 M MEHP (mono-2-ethylhexyl phthalate), the most produced phthalate, had no effect on basal or LH-stimulated production of testosterone, but it reduced the number of germ cells by increasing their apoptosis, without modification of their proliferation. This is the first experimental demonstration that phthalates alter the development of the foetal testis in humans. Using our organotypic culture system, we and others are currently investigating the effect of MEHP in the mouse and the rat, and it will be interesting to compare the results between these species to analyse the relevance of toxicological tests based on rodent models. Key words: endocrine disruptors, phthalates, environment, development, reproduction, health, foetus, testis, germ cells, testosterone Alterations in male reproductive function Changes in the environment and their consequences for male reproductive function have been of major concern for the past 20 years [1-3]. Correspondence: R. Habert, Laboratory of Differentiation and Radiobiology of the Gonads, CEA DSV/iRCM/SCSR, F Fontenay aux Roses, France; tel.: , fax.: , rene.habert@cea.fr Alterations in male reproduction were first observed in wild animals, in studies reporting the effects of accidental exposure of estrogenic chemicals on wildlife in the natural environment. These changes in male reproductive function vary from very subtle to permanent alterations, such as feminization or changes in reproductive behaviour [2,4]. Guillette et al studied the male reproductive function of alligators in two lakes in Florida. These two lakes are located very close Folia Histochem Cytobiol. 2009:47(5): S67 (S67-S74)
2 S68 to each other geographically, excluding the possibility of climate-based bias in these studies. They found that adult male alligators in Apopka Lake, which was polluted with agricultural waste and experienced a major chemical spill in 1980, had lower testosterone levels and presented micropenis and disorganized testes [5,6]. A key part of this story is that no chemicals could be detected in the water of the apparently contaminated lake and thus the alligators were being exposed simply by being at the top of the food chain. Other documented disruptions or alterations of reproductive activity and physiology have been correlated with exposure of contaminants in fish, amphibians, reptiles, birds, and mammals [4,7,8]. Most of the reported effects on wildlife have been observed on aquatic organisms and this is linked to the concentration of pollutants along the food chain. In humans, there is increasing evidence that the birth sex ratio is altered in areas close to industry and exposed to environmental and industrial chemicals. The findings of the recent report on the Aamjiwnaang First Nation community in Canada are striking [9], i.e. the proportion of male live births in this community has been decreasing continually from 1990 to 2003, the sex ratio (number of male births/total number of births) being only 0.3. The epidemiologic data have also shown an increase in human male reproductive function disorders over the past 50 years, with the suggestion of a relation with the increase in the amounts of endocrine disruptors in the environment. Testicular cancer, which is the most prevalent cancer in young men, has steadily increased in all countries studied, rising, for example, from 3.4 per 10,000 in 1973 to 5.5 per 10,000 in 1997 in North America [10]. Hypospadias and cryptorchidism also dramatically increased from 0.2 and 2%, respectively, in 1970 to 0.38 and 3.5%, respectively, in 1991 [10]. Finally, sperm count decline has been controversial, but large-scale prospective studies using standardized methodologies have shown a decline from 170 to 70 million spermatozoa per millilitre between 1940 and 1990 in Europe [1,11]. There are grounds for linking these four types of disorders. For example, a comparative study in European countries showed that the incidence of each of these four pre-cited abnormalities (sperm count decline, testicular cancer, hypospadias, and cryptorchidism) was maximal in Denmark and minimal in Finland [12]. Moreover, a history of cryptorchidism increases the risk of other three disorders [13], by a factor of 3-17 in the case of testicular cancer [14]. Similarly, hypospadias increases the chances of developing testicular cancer [1], and oligospermia is frequently observed in men who go on to develop testicular cancer [15,16]. Therefore, it has been suggested that these four alterations are symptoms of a single syndrome named the testicular dysgenesis syndrome (TDS) [16,17]. R. Habert et al. These abnormalities first arise during foetal development The foetal testis is formed by the mixing of a somatic anlage growing at the surface of the mesonephros and primordial germ cells that colonize the gonad and are called gonocytes (reviews in [18], Rouiller-Fabre et al in this issue). The foetal testis carries out crucial endocrine and gametogenic functions. Foetal Leydig cells produce the testosterone and insulin-like factor 3 (Insl3), which are absolutely necessary for phenotypic masculinisation of the embryo [19], (review in [20]). After birth, gonocytes give rise to the adult stem spermatogonia and thus correct development of the germ cell lineage during foetal/embryonic life is essential for the establishment of the ability of the individual to produce spermatozoa throughout his life. It is currently thought that TDS is probably caused by disturbances in the development of the foetal testis [16] because the origins of all four characteristics of TDS can be traced to foetal development. Hypospadias results from defects in androgen production or action during foetal development, while cryptorchidism results from abnormalities in the production and/or activity of Insl3 and/or the androgens respectively regulating the transabdominal and transinguinal descent of the testes [19]. The aetiology of testicular cancer remains unclear, but there is considerable evidence to suggest that it originates early in development [17] when gonocytes would normally have differentiated into spermatogonia. Carcinoma in situ (CIS) is a local malignant lesion that precedes testicular cancer (seminomas and nonseminomas) [21]. CIS cells closely resemble foetal germ cells in terms of morphology and immunohistological markers (c-kit, alkaline phosphatase, etc) [22]. Moreover, CIS has been reported in boys only a few months old [16]. Finally, sperm counts may have decreased for multiple reasons as the regulation of spermatogenesis remains poorly understood, but is known to involve complex endocrine, intratesticular, and intracellular regulation processes. The stock of gonocytes is determined during foetal development and takes part in determining the number of germ stem cells present in adulthood, since experimentally induced decreases in the number of gonocytes during foetal development lead to decreases in sperm count in adulthood [23,24] Similar results are obtained if the number of Sertoli cells is reduced during perinatal life [25]. Thus, adult sperm production depends partly on foetal seminiferous development. Numerous clinical, epidemiological and experimental data support the hypothesis that TDS is caused by endocrine disruptors acting during foetal or neonatal life (reviews in [2], [26-28]). Endocrine disruptors have been quantitatively and qualitatively increasing Folia Histochem Cytobiol. 2009:47(5): S68 (S67-S74)
3 Phthalates and foetal testis in our environment during the last decades. They originate from various sources including plants (phytoestrogens), agriculture (insecticides, herbicides, fumigants and fungicides) and many chemicals (pharmaceutical products, plasticisers, resins, detergents, PCB, flame retardant, bisphenol A, antimicrobial parabens, dioxins). In the present study we focused on phthalates. Environmental exposure to phthalates Phthalates (phthalic acid esters) are industrial chemicals that have been used increasingly since Their worldwide production grew from 1.8 to 4.3 million tons between 1970 and Phthalates are plasticisers that are added to polymers and essentially to PVC to make them softer and more flexible. They are widely used in a wide range of soft-pvc products including building and construction materials such as cabling, flooring, wall covering, profiles and roofs. They are components of medical equipment, hoses, shower curtains, films and plastic gloves, household furnishings, toys, car interiors, clothing, food and beverage packaging, pharmaceutical products, etc. Phthalates are also used as solvents for oil-soluble dyes, insecticides, peroxides, and other organics. They are added to paints and lacquers, adhesives and sealants, cosmetics, lubricants, putty, perfumes, deodorants, sprays. Di(2-ethylhexyl) phthalate (DEHP) is the most widely used phthalate. One and half million tons of DEHP are produced every year worldwide (40% in Europe). Phthalates are not covalently bound to the product matrix and can leach out over time from these products. As an example, they are recovered in domestic dust [29]. Dermal exposure via clothes and cosmetics may also occur. Small population groups may be exposed via medical equipment. So humans are constantly exposed to phthalates through oral, dermal and inhalation routes [30,31]. In the body, phthalates are rapidly hydrolyzed by esterases in the gut and other tissues into monoesters, which are the active molecules [32]. For example, DEHP is metabolized to its monoester metabolite, mono-(2-ethylhexyl) phthalate (MEHP), and DBP is converted into mono-butyl phthalate (MBP). Whereas many endocrine disruptors are persistent in the environment and accumulate in fat, the half-life of phthalates does not exceed 36 h in the body. In humans, 75% of the DEHP ingested is metabolized and excreted in urine within 2 days [33]. However, phthalates are so widespread in the environment that humans are largely exposed. As an example, according to a study published in 2003, 12% of the German population has a daily intake of DEHP that exceeds European recommendations [34]. In an epidemiologic study, 75% of the 289 human subjects tested were positive for the presence of four different types of phthalates in their urine samples [35]. The concentration of phthalates in biological fluids in humans show large individual variations [36-41]. Values for MEHP are reported in Table 1. Values for MBP are similar or higher, and its urinary concentration in pregnant mothers can reach M. The most recent reports indicate median levels in urine and amniotic fluid of M for MBP and M for MEHP [41]. Effects of phthalates on the development of the rat testis S69 The first observation of phthalate-induced testicular injuries was reported using adult rats in 1945 [42]. The oral administration of DEHP at dietary concentrations of 0.075, 0.75, 1.5 and 5.0% to rats for 90 days resulted in tubular atrophy and testicular degeneration at the two top dose levels. Subsequently, Harris et al. found occasional incidence of tubular atrophy in rats fed 0.5% DEHP in the diet for periods of 3 or 24 months [43]. Many other confirmed and explored the testicular effects of phthalate in experimental animals (review in [44]). Numerous studies in the rat foetus have shown that in utero exposure to di (n-butyl) phthalate (DBP) or DEHP [27,45]). Much attention has been paid to the analysis of Leydig cell development and function [46-52]. In utero exposure to phthalates induces an abnormal aggregation of the foetal Leydig cells, an occurrence of intratubular Leydig cells, a reduction of foetal testosterone production and Leydig cell Insl3 gene expression. This leads to epididymal agenesis, reduced ano-genital distance, hypospadias and cryptorchidism. Furthermore, in utero exposure to phthalates induces subnormal Sertoli cell proliferation [53], and possibly function [54,55]. Lastly, formation of multinucleated gonocytes during neonatal life and impaired spermatogenesis/infertility in adult have been observed [54,56]. Thus in utero exposure to phthalate results in a TDS-like syndrome in the male offspring, except for testicular cancer which is not induced. The in vitro approach has also been used for time and dose analyses of the effects of phthalates on the rat foetal testis. The main limitation on the toxicological use of in vitro studies is that the activities and fates of the cells in vitro must reproduce those existing in vivo. In mammalian cell culture systems, foetal Leydig cells dedifferentiate in the absence of specific gonadotropic stimulation [57-59]. Isolated gonocytes display poor survival in such systems [60-62]. Organ culture systems preserving testicular architecture and intercellular communications appear to us as a relevant method to maintain the development of the foetal, embryonic or neonatal testis. Furthermore, one testis can be cultured in the presence of the tested molecule whereas Folia Histochem Cytobiol. 2009:47(5): S69 (S67-S74)
4 S70 Table 1. Concentrations of MEHP in differents biological fluids in human the contralateral testis is cultured in its absence. Thus, an original technique using a filter system was been set up by Habert's team in 1991 [63] (Fig 1). This technique reproduces the development of the Leydig, Sertoli and germ cells observed in vivo [58,59,64-67]. Thus, this organotypic culture system is an important tool to study the age-, time- and dose-dependent direct effects of endocrine disruptors on the development of the foetal testis [67]. We named this method the rat foetal testis assay (rfeta) Then, we extended this method to mouse and human foetal testis [66,68-70] and named the methods mfeta and hfeta, respectively. Using this system, the effects of MBP and MEHP were investigated in the rat [71-74]. When explanted at 13.5 or 14.5 days post partum (dpc), one group found an MEHP-induced decrease in the number of gonocytes, and in the production of testosterone and AMH [74] which were not observed by others [72,73]. The reasons for these discrepancies are unclear, but are probably linked to experimental conditions. With foetal testes explanted at dpc, MEHP or MBP induced reductions in the proliferation and AMH expression of Sertoli cells and a reduction in LH-stimulated testosterone production [71,72]. At R. Habert et al. 3 days post partum, when proliferation of the gonocytes resumes, MEHP induced apoptosis in this cell type [72]. Effects of phthalates on the development of human foetal testis Despite the growing body of literature data on phthalate reproductive toxicity in animal models, and data demonstrating extensive human exposure, very few studies have examined the effects of these chemicals on human reproductive development. Epidemiological studies are highly difficult to perform because of the numerous factors that can act on foetal testis development, including hundreds of endocrine disruptors, alcohol, tobacco, individual stress. Furthermore, it is important to take into account the period of exposure. For instance, an association between a low sperm count in adult men with the treatment of their mother with diethylstilbestrol (DES) during pregnancy can be demonstrated only if the data relate to treatment during the first trimester [75]. Recently, anogenital distance was chosen in humans as an index of masculinisation and therefore an index of androgenic activity of the testis. An inverse correlation has been found between anogenital distance measured in male infants 2-36 months of age (mean 12.6 months) and maternal urinary concentrations of MBP and 3 other monoester phthalate metabolites measured at the end of pregnancy [38]. Interestingly, no correlation was observed for MEHP. In the same way, a concentration-dependent association between phthalates in breast milk and levels of reproductive hormones in boys at 3 months of age was also reported [39]. However, a recent paper by Huang et al 2009 reported no association between the anogenital distance measured in boys at birth and the concentration of any phthalates in the amniotic fluid or maternal urine during pregnancy. Taken together, these epidemiological studies suggest that some phthalates at environmental concentrations have antiandrogenic effects during neonatal life. Their antiandrogenic effects during foetal life are not demonstrated. Recently our team reported the first experimental demonstration of the potential deleterious effect of phthalates on human testis function or development [76]. In this study, we used the organ culture system of human foetal testes that we developed previously coupled with morphologic, functional, and molecular methods [68-70] to analyse the effects of MEHP on the development of testicular somatic and germ cells during the first trimester of pregnancy (Fig 1). This early developmental period of the testis has been shown to be a critical window for the determination of the reproductive tract [77]. Folia Histochem Cytobiol. 2009:47(5): S70 (S67-S74)
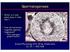
5 Phthalates and foetal testis S71 Fig. 1. Human foetal testis assay in humans (hfeta): One testis is cultured after fragmentation in the presence of the test compound and the other one from the same foetus is cultured in the absence of the test compound and served as control. MEHP was assayed and microphotographs show the reduction of the number of gonocytes after 3 days of culture in the presence of 10-4 M MEHP [76]. Gonocytes are identified as white cells inside the seminiferous cords, while the Sertoli cells have been immunostained for anti-müllerian hormone. Human foetal testes were recovered during the first trimester (7-12 weeks) of gestation and cultured for 3 days with or without MEHP in basal conditions or stimulated with luteinizing hormone (LH). Whatever the dose, MEHP treatment had no effect on basal or LH-stimulated testosterone produced by the human foetal testis in vitro. MEHP treatment did not affect the mrna expression of P450c17, P450scc, or StAR, or that of Insl3 produced by foetal Leydig cells, which is known to be involved in testicular descent and the expression of the steroidogenic enzymes. MEHP (10-4 M) did not affect proliferation or apoptosis of Sertoli cells, but it reduced the mrna expression of anti-müllerian hormone. Interestingly, MEHP (10-4 M) reduced the number of germ cells by increasing their apoptosis, measured by the detection of caspase-3-positive germ cells, without modification of their proliferation. Conclusions We review here arguments suggesting that the alterations of the male reproductive functions observed during the last decades are linked to an increasing exposure to endocrine disruptors with a focus on the phthalates. Our laboratory recently here the first experimental evidence for the potential of phthalates to impair the development of foetal testis in the human species [76]. Interestingly, the main disruption appears to be of the gametogenic function of the foetal testis, and no effect was observed on Leydig cell development and function. In the same way, epidemiological studies suggest that the antiandrogenic effects of environmental phthalates occur in the baby or the young child and not during foetal life. In rats, as in humans, phthalates act on foetal/neonatal gametogenesis, but, unlike in humans, they induce large antiandrogenic effects. Thus, the relevance of the rat model for toxicological studies related to phthalates must be debated. A recent paper reports that in utero exposure to phthalates impairs the development of gametogenesis without alterations of steroidogenesis of the foetal testis in mouse [78]. Using mouse foetal testis in culture, we also observed impairment of gametogenesis and no decrease of steroidogenesis [79]. Thus it will be important to establish whether the mouse could be an interesting model for studying the effects and mechanisms of action of phthalates in human foetal testicular development. Acknowledgements: This work was supported by Université Paris Diderot-Paris 7, CEA, INSERM, the Agence Française pour la Santé et la Sécurité dans l'environnement et le Travail (AFSSET) Folia Histochem Cytobiol. 2009:47(5): S71 (S67-S74)
6 S72 R. Habert et al. and the Agence Nationale pour la Recherche (ANR). V.M. and A.L. hold fellowships from the Ministere de l'education Nationale de la Recherche et de la Technologie. R.L. was the recipient of a fellowship from the Commissariat a l'energie Atomique. We thank the staff of the Department of Obstetrics and Gynecology of the Antoine Béclere Hospital (Clamart, France). We thank Aurélie Gouret for her skilful secretarial assistance. This review is dedicated to my mentor, JM Saez. References [1] Sharpe RM, Irvine DS. How strong is the evidence of a link between environmental chemicals and adverse effects on human reproductive health? BMJ. 2004;328: [2] Delbes G, Levacher C, Habert R. Estrogen effects on fetal and neonatal testicular development. Reproduction. 2006;132: [3] Leridon H, Slama R. The impact of a decline in fecundity and of pregnancy postponement on final number of children and demand for assisted reproduction technology. Hum Reprod. 2008;23: [4] Vos JG, Dybing E, Greim HA, Ladefoged O, Lambre C, Tarazona JV, Brandt I, Vethaak AD. Health effects of endocrinedisrupting chemicals on wildlife, with special reference to the European situation. Crit Rev Toxicol. 2000;30: [5] Guillette LJ Jr, Gross TS, Masson GR, Matter JM, Percival HF & Woodward AR. Developmental abnormalities of the gonad and abnormal sex hormone concentrations in juvenile alligators from contaminated and control lakes in Florida. Environ Health Perspect. 1994;102: [6] Guillette LJ Jr, Guillette EA. Environmental contaminants and reproductive abnormalities in wildlife: implications for public health? Toxicol Industr Health. 1996;12: [7] Oskam IC, Ropstad E, Dahl E, Lie E, Derocher AE, Wiig O, Larsen S, Wiger R & Skaare JU. Organochlorines affect the major androgenic hormone, testosterone, in male polar bears (Ursus maritimus) at Svalbard. J Toxicol Environ Health ;Part A 66: [8] Edwards TM, Moore BC, Guillette LJ Jr. Reproductive dysgenesis in wildlife: a comparative view. Int J Androl. 2006; 29: [9] Mackenzie CA, Lockridge A, Keith M. Declining sex ratio in a first nation community. Environ Health Perspect. 2005;113: [10] Toppari J. Environmental endocrine disrupters and disorders of sexual differentiation. Semin Reprod Med. 2002;20: [11] Auger J, Kunstmann JM, Czyglik F, Jouannet P. Decline in semen quality among fertile men in Paris during the past 20 years. NEJM. 1995;332: [12] Virtanen HE, Rajpert-De Meyts E, Main KM, Skakkebaek NE, Toppari J. Testicular dysgenesis syndrome and the development and occurrence of male reproductive disorders. Toxicol Applied Pharmacol. 2005; [13] Kaleva M, Virtanen HE, Haavisto AM, Main KM, Reunanen M, Skakkebaek NE & Toppari J. Circannual rhythm in the incidence of cryptorchidism in Finland. Int J Androl. 2005; [14] Davenport M. Risk of testicular cancer in boys with cryptorchidism. Study was based on small number of cancers. BMJ. 1997;315: [15] Moller H & Skakkebaek NE. Risk of testicular cancer in subfertile men: case-control study. BMJ. 1999;318: [16] Skakkebaek NE, Rajpert-De Meyts E, Main KM. Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Human Reprod. 2001;16: [17] Skakkebaek NE, Jorgensen N. Testicular dysgenesis and fertility. Andrologia. 2005;37: [18] Olaso R, Habert R. Genetic and cellular analysis of male germ cell development. J Androl. 2000;21: [19] Kubota Y, Temelcos C, Bathgate RA, Smith KJ, Scott D, Zhao C, Hutson JM. The role of insulin 3, testosterone, Mullerian inhibiting substance and relaxin in rat gubernacular growth. Mol Human Reprod. 2002;8: [20] Habert R, Lejeune H & Saez JM. Origin, differentiation and regulation of fetal and adult Leydig cells. Mol Cell Endocrinol. 2001;179: [21] Skakkebaek NE, Bancroft J, Davidson DW, Warner P. Androgen replacement with oral testosterone undecanoate in hypogonadal men: a double blind controlled study. Clin Endocrinol. 1981;14: [22] Meyts E, Jorgensen N, Brondum-Nielsen K, Muller J, Skakkebaek NE. Developmental arrest of germ cells in the pathogenesis of germ cell neoplasia. APMIS. 1998;106: (Discussion ). [23] Moreno SG, Dutrillaux B, Coffigny H. Status of p53, p21, mdm2, prb proteins, and DNA methylation in gonocytes of control and gamma-irradiated rats during testicular development. Biol Reprod 2001;64: [24] Forand A, Messiaen S, Habert R, & Bernardino-Sgherri J. Exposure of the mouse perinatal testis to radiation leads to hypospermia at sexual maturity Reproduction. (in press). [25] Orth JM, Gunsalus GL, Lamperti A. Evidence from sertoli celldepleted rats indicates that spermatid number in adults depends on numbers of sertoli cells produced during perinatal development. Endocrinology. 1988;122: [26] Sharpe RM. Pathways of endocrine disruption during male sexual differentiation and masculinization. Best Pract Res Clin Endocrinol Metab. 2006;20: [27] Sharpe RM, Skakkebaek NE. Testicular dysgenesis syndrome: mechanistic insights and potential new downstream effects. Fertil Steril. 2008;89(2 Suppl): [28] Wohlfahrt-Veje C, Main KM, Skakkebæ k NE. Testicular Dysgenesis Syndrome; Fetal origin of adult reproductive problems. Clin Endocrinol (Oxf) Feb 16. [Epub ahead of print] [29] Becker K, Seiwert M, Angerer J, Heger W, Koch HM, Nagorka R, Rosskamp E, Schlüter C, Seifert B, Ullrich D. DEHP metabolites in urine of children and DEHP in house dust. Int J Hyg Environ Health. 2004;207: [30] Koch HM, Preuss R, Angerer J. Di(2-ethylhexyl)phthalate (DEHP): human metabolism and internal exposure-- an update and latest results. Int J Androl. 2006;29: [31] Wormuth M, Scheringer M, Vollenweider M, Hungerbuhler K. What are the sources of exposure to eight frequently used phthalic acid esters in Europeans? Risk Anal. 2006;26: [32] Latini G. Monitoring phthalate exposure in humans. Clin Chim Acta. 2005;361: [33] Koch HM, Bolt HM, Preuss R, Angerer J. New metabolites of di(2- ethylhexyl)phthalate (DEHP) in human urine and serum after single oral doses of deuteriumlabeled DEHP. Arch Toxicol. 2005;79: [34] Koch HM, Drexler H, Angerer J. An estimation of the daily intake of di(2- ethylhexyl)phthalate (DEHP) and other phthlates in the general population. Int J Hyg Environ Health. 2003;206: [35] Blount BC, Silva MJ, Caudill SP, Needham LL, Pirkle JL, Sampson EJ, et al. Levels of seven urinary phthalate metabolites in a human reference population. Environ Health Perspect. 2000;108: [36] Latini G, De Felice C, Presta G, Del Vecchio A, Paris I, Ruggieri F, Mazzeo P. In utero exposure to di-(2- ethylhexyl)phthalate and duration of human pregnancy. Environ Health Perspect. 2003;111: Folia Histochem Cytobiol. 2009:47(5): S72 (S67-S74)
7 Phthalates and foetal testis S73 [37] Silva MJ, Barr DB, Reidy JA, Malek NA, Hodge CC, Caudill SP, et al. Urinary levels of seven phthalate metabolites in the U.S. population from the National Health and Nutrition Examination Survey (NHANES) Environ Health Perspect. 2004;112: [38] Swan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM, Mao CS, Redmon JB, Ternand CL, Sullivan S et al. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect. 2005; 113: [39] Main KM, Mortensen GK, Kaleva MM, Boisen KA, Damgaard IN, Chellakooty M, Schmidt IM, Suomi AM, Virtanen HE, Petersen DV, Andersson AM, Toppari J, Skakkebaek NE. Human breast milk contamination with phthalates and alterations of endogenous reproductive hormones in infants three months of age. Environ Health Perspect. 2006;11: [40] Högberg J, Hanberg A, Berglund M, Skerfving S, Remberger M, Calafat AM, Filipsson AF, Jansson B, Johansson N, Appelgren M, Hakansson H. Phthalate diesters and their metabolites in human breast milk, blood or serum, and urine as biomarkers of exposure in vulnerable populations. Environ Health Perspect. 2008;116: [41] Huang PC, Kuo PL, Chou YY, Lin SJ, Lee CC. Association between prenatal exposure to phthalates and the health of newborns. Environ Int. 2009;35: [42] Shaffer CB, Carpenter CP, Smyth HR Jr. Acute and subacute toxicity of di-(2-ethylhexyl)phthalate with note upon its metabolism. J Ind Hyg Toxicol. 1945;27: [43] Harris RS, Hodge HC, Maynard ER and Blancher HJ Jr. Chronic oral toxicity of 2-ethylhexyl phthalate in rats and dogs. Arch Ind Hyg. 1956;13: [44] Gangolli SD. Testicular effects of phthalate esters. Environ Health Perspect. 1982;45: [45] Foster PM. Disruption of reproductive development in male rat offspring following in utero exposure to phthalate esters. Int J Androl. 2006;29: ; discussion [46] Mylchreest E, Wallace DG, Cattley RC, Foster PM. Dosedependent alterations in androgen-regulated male reproductive development in rats exposed to Di(n-butyl) phthalate during late gestation. Toxicol Sci. 2000;55: [47] Parks LG, Ostby JS, Lambright CR, Abbott BD, Klinefelter GR, Barlow NJ, et al. The plasticizer diethylhexyl phthalate induces malformations by decreasing fetal testosterone synthesis during sexual differentiation in the male rat. Toxicol Sci. 2000;58: [48] Lehmann KP, Phillips S, Sar M, Foster PM, Gaido K. Dosedependent alterations in gene expression and testosterone synthesis in the fetal testes of male rats exposed to di (n-butyl) phthalate. Toxicol Sci. 2004;81: [49] Wilson VS, Lambright C, Furr J, Ostby J, Wood C, Held G, Gray LE, Jr. Phthalate ester-induced gubernacular lesions are associated with reduced insl3 gene expression in the fetal rat testis. Toxicol Lett. 2004;146: [50] Mahood IK, Hallmark N, McKinnell C, Walker M, Fisher JS, Sharpe RM. Abnormal Leydig cell aggregation in the fetal testis of rats exposed to di (n-butyl) phthalate and its possible role in testicular dysgenesis. Endocrinology. 2005;146: [51] McKinnell C, Sharpe RM, Mahood K, Hallmark N, Scott H, Ivell R, Staub C, Jegou B, Haag F, Koch-Nolte F, Hartung S. Expression of insulin-like factor 3 protein in the rat testis during fetal and postnatal development and in relation to cryptorchidism induced by in utero exposure to di (n-butyl) phthalate. Endocrinology. 2005;146: [52] Howdeshell KL, Wilson VS, Furr J, Lambright CR, Rider CV, Blystone CR, Hotchkiss AK, Gray LE Jr. A mixture of five phthalate esters inhibits fetal testicular testosterone production in the sprague-dawley rat in a cumulative, dose-additive manner. Toxicol Sci. 2008;105: [53] Scott HM, Hutchison GR, Mahood IK, Hallmark N, Welsh M, de Gendt K, et al. Role of androgens in fetal testis development and dysgenesis. Endocrinology 2007;148: [54] Fisher JS, Macpherson S, Marchetti N, Sharpe RM. Human ''testicular dysgenesis syndrome'': a possible model using inutero exposure of the rat to dibutyl phthalate. Hum Reprod. 2003;18: [55] Fisher JS. Environmental anti-androgens and male reproductive health: focus on phthalates and testicular dysgenesis syndrome. Reproduction. 2004;127: [56] Ferrara D, Hallmark N, Scott H, Brown R, McKinnell C, Mahood IK, Sharpe RM. Acute and long-term effects of in utero exposure of rats to di(n-butyl) phthalate on testicular germ cell development and proliferation. Endocrinology. 2006;147: [57] Gautier C, Levacher C, Saez JM, Habert R. Transforming Growth Factor β1 inhibits steroidogenesis in dispersed fetal testicular cells in culture. Mol Cell Endocrinol. 1997;131: [58] Rouiller-Fabre L, Carmona S, Abou-Merhi R, Cate R, Habert R, Vigier B. Effect of anti-müllerian hormone (AMH) on Sertoli and Leydig cell functions in fetal and immature rats. Endocrinology. 1998;139: [59] Rouiller-Fabre V, Lecerf L, Gautier C, Saez JM, Habert R. Expression and effect of Insulin-like Growth Factor I on rat fetal Leydig cell function and differentiation. Endocrinology. 1998;139: [60] Van Dissel-Emiliani FM, de Boer-Brouwer M, Spek ER, van der Donk JA, de Rooij DJ. Survival and proliferation of rat gonocytes in vitro. Cell Tissue Res. 1993;273: [61] Li H, Papadapoulos V, Vidic B, Dym M, Culty M. Regulation of rat testis gonocyte proliferation by platelet-derived growth factor and estradiol: identification of signaling mechanisms involved. Endocrinology. 1997;138: [62] Boulogne B, Habert R, Levacher C. Regulation of the proliferation of cocultured gonocytes and Sertoli cells by retinoids, triiodothyronine, and intracellular signaling factors: differences between fetal and neonatal cells. Mol Reprod Dev. 2003;65: [63] Habert R, Devif I, Gangnerau MN, Lecerf L. Ontogenesis of the in vitro response of rat testis to gonadotropin-releasing hormone. Mol Cell Endocrinol. 1991;82: [64] Lecerf L, Rouiller-Fabre V, Levacher C, Gautier C, Saez J, Habert R. Stimulatory effect of follicle-stimulating hormone on basal and luteinizing hormone-stimulated testosterone secretion by fetal rat testis in vitro. Endocrinology. 1993; 133: [65] Olaso R, Pairault C, Boulogne B, Durand P, Habert R. Transforming Growth Factor β1 and β2 reduce the number of gonocytes by increasing apoptosis. Endocrinology. 1998;139: [66] Livera G, Rouiller-Fabre V, Durand P, Habert R. Multiple effects of retinoids on the development of Sertoli, germ and Leydig cells of fetal and neonatal rat testis in culture. Biol Reprod. 2000;62: [67] Livera G, Delbes G, Pairault C, Rouiller-Fabre V, Habert R. Organotypic culture, a powerful model for studying rat and mouse fetal testis development. Cell Tissue Res. 2006;324: [68] Lambrot R, Coffigny H, Pairault C, Frydman R, Habert R, Rouiller-Fabre V. Use of organ culture to study the human fetal testis development: effect of retinoic acid. J Clin Endoc Metab. 2006;91: [69] Lambrot R, Coffigny H, Pairault C, Frydman R, Habert R & Rouiller-Fabre V. High radiosensitivity of germ cells in human male fetus. J Clin Endoc Metab. 2007;92: [70] Lambrot R, Livera G, Coffigny H, Pairault C, Frydman R, Habert R, Rouiller-Fabre V. A new method for toxicity assays on human and mouse fetal testis. Biochimie. 2006;88: Folia Histochem Cytobiol. 2009:47(5): S73 (S67-S74)
8 S74 R. Habert et al. [71] Hallmark N, Walker M, McKinnell C, Mahood IK, Scott H, Bayne R, Coutts S, Anderson RA, Greig I, Morris K, Sharpe RM. Effects of monobutyl and di(n-butyl) phthalate in vitro on steroidogenesis and Leydig cell aggregation in fetal testis explants from the rat: comparison with effects in vivo in the fetal rat and neonatal marmoset and in vitro in the human. Environ Health Perspect. 2007;115: [72] Li H, Kim KH. Effects of mono-(2-ethylhexyl) phthalate on fetal and neonatal rat testis organ cultures. Biol Reprod. 2003; 69: [73] Stroheker T, Regnier JF, Lassurguere J, Chagnon MC. Effect of in utero exposure to di-(2-ethylhexyl)phthalate: distribution in the rat fetus and testosterone production by rat fetal testis in culture. Food Chem Toxicol. 2006;44: [74] Chauvigné F, Menuet A, Lesné L, Chagnon MC, Chevrier C, Regnier JF, Angerer J, Jégou B. Time- and Dose-Related Effects of Di-(2-ethylhexyl) Phthalate and its Main Metabolites on the Function of the Rat Fetal Testis in vitro. Environ Health Perspect. in press. [75] Storgaard L, Bonde JP, Olsen J. Male reproductive disorders in humans and prenatal indicators of estrogen exposure. A review of published epidemiological studies. Cell Tissue Res. 2006;21: [76] Lambrot R, Muczynski V, Lécureuil C, Angenard G, Coffigny H, Pairault C, Moison D, Frydman R, Habert R, Rouiller- Fabre V. Phthalates impair germ cell development in the human fetal testis in vitro without change in testosterone production. Environ Health Perspect. 2009;117: [77] Welsh M, Saunders PT, Fisken M, Scott HM, Hutchison GR, Smith LB, et al. Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. J Clin Invest. 2008;118: [78] Gaido KW, Hensley JB, Liu D, Wallace DG, Borghoff S, Johnson KJ, Hall SJ, Boekelheide K. Fetal mouse phthalate exposure shows that Gonocyte multinucleation is not associated with decreased testicular testosterone. Toxicol Sci. 2007; 97: [79] Lehraiki A, Szenker J, Habert R, Levacher C. In vivo and in vitro adverse effects of phthalates on fetal and neonatal mouse gonocytes 15th European Testis Workshop. Naantali- Finland Miniposter N Available in miniposters&pm=main miniposters Folia Histochem Cytobiol. 2009:47(5): S74 (S67-S74)
Ontogenesis of testicular function in humans
 FOLIA HISTOCHEMICA ET CYTOBIOLOGICA Vol. 47, No. 5, 2009 pp. S19-S24 Review article Ontogenesis of testicular function in humans Virginie Rouiller-Fabre 1,2,3, Vincent Muczynski 1,2,3, Romain Lambrot 1,2,3,
FOLIA HISTOCHEMICA ET CYTOBIOLOGICA Vol. 47, No. 5, 2009 pp. S19-S24 Review article Ontogenesis of testicular function in humans Virginie Rouiller-Fabre 1,2,3, Vincent Muczynski 1,2,3, Romain Lambrot 1,2,3,
Edinburgh Research Explorer
 Edinburgh Research Explorer Effect of fetal or neonatal exposure to monobutyl phthalate (MBP) on testicular development and function in the marmoset Citation for published version: McKinnell, C, Mitchell,
Edinburgh Research Explorer Effect of fetal or neonatal exposure to monobutyl phthalate (MBP) on testicular development and function in the marmoset Citation for published version: McKinnell, C, Mitchell,
Effect of fetal or neonatal exposure to monobutyl phthalate (MBP) on testicular development and function in the marmoset
 Human Reproduction, Vol.00, No.0 pp. 1 11, 2009 doi:10.1093/humrep/dep200 Hum. Reprod. Advance Access published June 2, 2009 ORIGINAL ARTICLE Reproductive biology Effect of fetal or neonatal exposure to
Human Reproduction, Vol.00, No.0 pp. 1 11, 2009 doi:10.1093/humrep/dep200 Hum. Reprod. Advance Access published June 2, 2009 ORIGINAL ARTICLE Reproductive biology Effect of fetal or neonatal exposure to
Endocrine disrupters a role in human health?
 9 In: Endocrine Disrupters T. Grotmol, A. Bernhoft, G.S. Eriksen and T.P. Flaten, eds. Oslo: The Norwegian Academy of Science and Letters, 2006. Endocrine disrupters a role in human health? Erik Dybing
9 In: Endocrine Disrupters T. Grotmol, A. Bernhoft, G.S. Eriksen and T.P. Flaten, eds. Oslo: The Norwegian Academy of Science and Letters, 2006. Endocrine disrupters a role in human health? Erik Dybing
I have no financial disclosures
 The Environment and Reproductive Health: Why What We Eat, Breath and Touch Matters Susan.Davidson@SSMHealth.com Susan Davidson, MD Dean Medical Center I have no financial disclosures 1 Many Complex Factors
The Environment and Reproductive Health: Why What We Eat, Breath and Touch Matters Susan.Davidson@SSMHealth.com Susan Davidson, MD Dean Medical Center I have no financial disclosures 1 Many Complex Factors
Comparison of penile length at 6 24 months between children with unilateral cryptorchidism and a healthy normal cohort
 Original Article - Pediatric Urology Investig Clin Urol 2018;59:55-60. pissn 2466-0493 eissn 2466-054X Comparison of penile length at 6 24 months between children with unilateral cryptorchidism and a healthy
Original Article - Pediatric Urology Investig Clin Urol 2018;59:55-60. pissn 2466-0493 eissn 2466-054X Comparison of penile length at 6 24 months between children with unilateral cryptorchidism and a healthy
ESPOSIZIONE PRE- E POSTNATALE A FTALATI E SALUTE DEL FETO E DEL NEONATO. Giuseppe Latini Divisione di Neonatologia Ospedale Perrino Brindisi
 ESPOSIZIONE PRE- E POSTNATALE A FTALATI E SALUTE DEL FETO E DEL NEONATO Giuseppe Latini Divisione di Neonatologia Ospedale Perrino Brindisi Background PHTHALATES ARE A GROUP OF INDUSTRIAL CHEMICALS WITH
ESPOSIZIONE PRE- E POSTNATALE A FTALATI E SALUTE DEL FETO E DEL NEONATO Giuseppe Latini Divisione di Neonatologia Ospedale Perrino Brindisi Background PHTHALATES ARE A GROUP OF INDUSTRIAL CHEMICALS WITH
EOH3101 PRINCIPLES OF ENVIRONMENTAL HEALTH ENVIRONMENTAL HEALTH ISSUES: ENVIROMENTAL HORMONES
 EOH3101 PRINCIPLES OF ENVIRONMENTAL HEALTH ENVIRONMENTAL HEALTH ISSUES: ENVIROMENTAL HORMONES INTRODUCTION Endocrine disruptors are chemicals that may interfere with the body s endocrine system and produce
EOH3101 PRINCIPLES OF ENVIRONMENTAL HEALTH ENVIRONMENTAL HEALTH ISSUES: ENVIROMENTAL HORMONES INTRODUCTION Endocrine disruptors are chemicals that may interfere with the body s endocrine system and produce
Effect of Exogenous Estrogen during Pregnancy on the Development of the Testis of Rats. Histological Considerations and Clinical Implications
 Effect of Exogenous Estrogen during Pregnancy on the Development of the Testis of Rats. Histological Considerations and Clinical Implications etwally Abd-El-Bary nsoor Anatomy Department, Faculty of edicine,
Effect of Exogenous Estrogen during Pregnancy on the Development of the Testis of Rats. Histological Considerations and Clinical Implications etwally Abd-El-Bary nsoor Anatomy Department, Faculty of edicine,
DAX1, testes development role 7, 8 DFFRY, spermatogenesis role 49 DMRT genes, male sex differentiation role 15
 Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
Tackling gender inequalities in Environmental Health
 Tackling gender inequalities in Environmental Health Madrid, 11 November 2009 Sascha Gabizon,, Executive Director WECF - Women in Europe for a Common Future Why Women? Women as full partners in policy
Tackling gender inequalities in Environmental Health Madrid, 11 November 2009 Sascha Gabizon,, Executive Director WECF - Women in Europe for a Common Future Why Women? Women as full partners in policy
Annex XV report PROPOSAL FOR IDENTIFICATION OF A SUBSTANCE OF VERY HIGH CONCERN ON THE BASIS OF THE CRITERIA SET OUT IN REACH ARTICLE 57
 Annex XV report PROPOSAL FOR IDENTIFICATION OF A SUBSTANCE OF VERY HIGH CONCERN ON THE BASIS OF THE CRITERIA SET OUT IN REACH ARTICLE 57 Substance Name(s): Bis(2-ethylhexyl) phthalate (DEHP) EC Number(s):
Annex XV report PROPOSAL FOR IDENTIFICATION OF A SUBSTANCE OF VERY HIGH CONCERN ON THE BASIS OF THE CRITERIA SET OUT IN REACH ARTICLE 57 Substance Name(s): Bis(2-ethylhexyl) phthalate (DEHP) EC Number(s):
SASCHA GABIZON Women in Europe for a Common Future Director
 Dangerous Chemicals in products targeted at Children and Women SASCHA GABIZON sascha.gabizon@wecf.org Women in Europe for a Common Future Director Children most vulnerable Persistent pesticides accumulate
Dangerous Chemicals in products targeted at Children and Women SASCHA GABIZON sascha.gabizon@wecf.org Women in Europe for a Common Future Director Children most vulnerable Persistent pesticides accumulate
SCIENTIFIC COMMITTEE ON TOXICITY, ECOTOXICITY AND THE ENVIRONMENT (CSTEE) Opinion on the results of the Risk Assessment of:
 EUROPEAN COMMISSION DIRECTORATE-GENERAL HEALTH AND CONSUMER PROTECTION Directorate C - Scientific Opinions Unit C2 Management of Scientific Committees; scientific co-operation and networks Scientific Committee
EUROPEAN COMMISSION DIRECTORATE-GENERAL HEALTH AND CONSUMER PROTECTION Directorate C - Scientific Opinions Unit C2 Management of Scientific Committees; scientific co-operation and networks Scientific Committee
Your environment: Your fertility
 Your environment: Your fertility Strong Fertility Center Education Series September 25, 2008 Shanna H. Swan, PhD Professor Obstetrics & Gynecology University of Rochester School of Medicine Has fertility
Your environment: Your fertility Strong Fertility Center Education Series September 25, 2008 Shanna H. Swan, PhD Professor Obstetrics & Gynecology University of Rochester School of Medicine Has fertility
Environmental and Developmental Origins of Ovarian Reserve. Nick Macklon Professor of Obstetrics and Gynaecology, University of Southampton, UK.
 Environmental and Developmental Origins of Ovarian Reserve Nick Macklon Professor of Obstetrics and Gynaecology, University of Southampton, UK. Why worry? Women delaying childbirth Diminished ovarian
Environmental and Developmental Origins of Ovarian Reserve Nick Macklon Professor of Obstetrics and Gynaecology, University of Southampton, UK. Why worry? Women delaying childbirth Diminished ovarian
IN SUMMARY HST 071 NORMAL & ABNORMAL SEXUAL DIFFERENTIATION Fetal Sex Differentiation Postnatal Diagnosis and Management of Intersex Abnormalities
 Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
DISORDERS OF MALE GENITALS
 Wit JM, Ranke MB, Kelnar CJH (eds): ESPE classification of paediatric endocrine diagnosis. 9. Testicular disorders/disorders of male genitals. Horm Res 2007;68(suppl 2):63 66 ESPE Code Diagnosis OMIM ICD10
Wit JM, Ranke MB, Kelnar CJH (eds): ESPE classification of paediatric endocrine diagnosis. 9. Testicular disorders/disorders of male genitals. Horm Res 2007;68(suppl 2):63 66 ESPE Code Diagnosis OMIM ICD10
Six Classes We can reduce harmful chemicals for a healthier world.
 Six Classes We can reduce harmful chemicals for a healthier world. 9 F 17 Cl 35 Br Six classes 1. Highly fluorinated chemicals water and oil repellants, surfactants 2. Antimicrobials triclosan, triclocarban
Six Classes We can reduce harmful chemicals for a healthier world. 9 F 17 Cl 35 Br Six classes 1. Highly fluorinated chemicals water and oil repellants, surfactants 2. Antimicrobials triclosan, triclocarban
Endocrine Disrupter Chemicals: Universal Access to Healthcare
 Endocrine Disrupter Chemicals: the right to know Riana Bornman, School of Health Systems and Public Health, University of Pretoria SAMA Conference 2016 Universal Access to Healthcare Concern started SOS-EDCs
Endocrine Disrupter Chemicals: the right to know Riana Bornman, School of Health Systems and Public Health, University of Pretoria SAMA Conference 2016 Universal Access to Healthcare Concern started SOS-EDCs
Di(n-butyl) phthalate has no effect on the rat prepubertal testis despite its estrogenic activity in vitro
 FOLIA HISTOCHEMICA ET CYTOBIOLOGICA Vol. 49, No. 4, 2011 pp. 685 689 ORIGINAL STUDY Di(n-butyl) phthalate has no effect on the rat prepubertal testis despite its estrogenic activity in vitro Eliza Filipiak
FOLIA HISTOCHEMICA ET CYTOBIOLOGICA Vol. 49, No. 4, 2011 pp. 685 689 ORIGINAL STUDY Di(n-butyl) phthalate has no effect on the rat prepubertal testis despite its estrogenic activity in vitro Eliza Filipiak
Endocrine Disruptors
 page 1/10 Scientific Facts on Endocrine Disruptors Source document: IPCS (2002) Summary & Details: GreenFacts Level 2 - Details on Endocrine Disruptors 1. What are Endocrine Disruptors (EDCs)?...2 1.1
page 1/10 Scientific Facts on Endocrine Disruptors Source document: IPCS (2002) Summary & Details: GreenFacts Level 2 - Details on Endocrine Disruptors 1. What are Endocrine Disruptors (EDCs)?...2 1.1
Hormones. Hormones. When you hear the word hormones, what do you think of? When you hear the word hormones, what do you think of?
 When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what Hormones As your body s chemical messengers, hormones
When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what Hormones As your body s chemical messengers, hormones
Maldescended testis in Adults. Dr. BG GAUDJI Urologist STEVE BIKO ACADEMIC HOSPITAL
 Maldescended testis in Adults Dr. BG GAUDJI Urologist STEVE BIKO ACADEMIC HOSPITAL Definitions Cryptorchid: testis neither resides nor can be manipulated into the scrotum Ectopic: aberrant course Retractile:
Maldescended testis in Adults Dr. BG GAUDJI Urologist STEVE BIKO ACADEMIC HOSPITAL Definitions Cryptorchid: testis neither resides nor can be manipulated into the scrotum Ectopic: aberrant course Retractile:
Hormonal Control of Male Sexual Function
 Hormonal Control of Male Sexual Function A majority of the control of sexual functions in the male (and the female) begins with secretions of gonadotropin-releasing hormone (GnRH) by the hypothalamus.
Hormonal Control of Male Sexual Function A majority of the control of sexual functions in the male (and the female) begins with secretions of gonadotropin-releasing hormone (GnRH) by the hypothalamus.
Sensitive and vulnerable risks groups in relation to chemical risk assessment and management
 International Research and Research Training Centre Sensitive and vulnerable risks groups in relation to chemical risk assessment and management Niels E. Skakkebaek Department of Growth and Reproduction
International Research and Research Training Centre Sensitive and vulnerable risks groups in relation to chemical risk assessment and management Niels E. Skakkebaek Department of Growth and Reproduction
ENDOCRINE DISRUPTION: ARE MALES AT RISK?
 ENDOCRINE DISRUPTION: ARE MALES AT RISK? Theo Colborn, Ph.D. TEDX The first 38 weeks of human life spent in the allegedly protected environment of the amniotic sac are medically more eventful and more
ENDOCRINE DISRUPTION: ARE MALES AT RISK? Theo Colborn, Ph.D. TEDX The first 38 weeks of human life spent in the allegedly protected environment of the amniotic sac are medically more eventful and more
NO.4 A Sea of the Synthetic Chemical Compounds
 NO.4 A Sea of the Synthetic Chemical Compounds The symptoms generally called "allergy" appear through reactions of living creatures against the foreign substance taken into the body from the environment.
NO.4 A Sea of the Synthetic Chemical Compounds The symptoms generally called "allergy" appear through reactions of living creatures against the foreign substance taken into the body from the environment.
Endocrine Disrupting Chemicals: New considerations in the toxics space TURI Annual Meeting
 Endocrine Disrupting Chemicals: New considerations in the toxics space TURI Annual Meeting Laura N. Vandenberg, PhD UMass Amherst School of Public Health April 25, 2018 Disclosure statement I am funded
Endocrine Disrupting Chemicals: New considerations in the toxics space TURI Annual Meeting Laura N. Vandenberg, PhD UMass Amherst School of Public Health April 25, 2018 Disclosure statement I am funded
L Earl Gray Jr. Research Biologist
 EFFECTS OF MIXTURES OF PHTHALATES, PESTICIDES AND TCDD ON SEXUAL DIFFERENTIATON IN RATS: A RISK FRAMEWORK BASED UPON DISRUPTION OF COMMON DEVELOPING SYSTEMS. USEPA scientist grapples with difficult environmental
EFFECTS OF MIXTURES OF PHTHALATES, PESTICIDES AND TCDD ON SEXUAL DIFFERENTIATON IN RATS: A RISK FRAMEWORK BASED UPON DISRUPTION OF COMMON DEVELOPING SYSTEMS. USEPA scientist grapples with difficult environmental
Male Anatomy. testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics
 Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
To: National Toxicology Program (NTP); Center for the Evaluation of Risks to Human Reproduction (CERHR)
 February 2, 2007 To: National Toxicology Program (NTP); Center for the Evaluation of Risks to Human Reproduction (CERHR) From: Natural Resources Defense Council These comments are submitted by Natural
February 2, 2007 To: National Toxicology Program (NTP); Center for the Evaluation of Risks to Human Reproduction (CERHR) From: Natural Resources Defense Council These comments are submitted by Natural
Hormones of brain-testicular axis
 (Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
(Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
Exposures and Heritable Health Effects
 Exposures and Heritable Health Effects Thaddeus T. Schug Population Health Branch National Institute of Environmental Health Sciences November 30, 2017 Outline Developmental exposures and health effects
Exposures and Heritable Health Effects Thaddeus T. Schug Population Health Branch National Institute of Environmental Health Sciences November 30, 2017 Outline Developmental exposures and health effects
Endocrine Disrupting Activity Associated With Oil and Natural Gas Extraction
 Endocrine Disrupting Activity Associated With Oil and Natural Gas Extraction Susan C Nagel, PhD Associate Professor Obstetrics, Gynecology, and Women s Health University of Missouri NIH R21ES026395 and
Endocrine Disrupting Activity Associated With Oil and Natural Gas Extraction Susan C Nagel, PhD Associate Professor Obstetrics, Gynecology, and Women s Health University of Missouri NIH R21ES026395 and
Experimentally induced testicular dysgenesis syndrome originates in the masculinization programming window
 Experimentally induced testicular dysgenesis syndrome originates in the masculinization programming window Sander van den Driesche, 1 Karen R. Kilcoyne, 1 Ida Wagner, 1 Diane Rebourcet, 1 Ashley Boyle,
Experimentally induced testicular dysgenesis syndrome originates in the masculinization programming window Sander van den Driesche, 1 Karen R. Kilcoyne, 1 Ida Wagner, 1 Diane Rebourcet, 1 Ashley Boyle,
Title: Report on Public Health Concerns - Phthalates and Bisphenol A
 Title: Report on Public Health Concerns - Phthalates and Bisphenol A To: Public Health Subcommittee, Health and Government Operations Committee of the Maryland General Assembly Plain Language Summary Prepared
Title: Report on Public Health Concerns - Phthalates and Bisphenol A To: Public Health Subcommittee, Health and Government Operations Committee of the Maryland General Assembly Plain Language Summary Prepared
Session 5 Children s Health
 2002 Session 5 Children s Health Regional Differences and Temporal Trends in Semen Quality Niels Jørgensen Rigshospitalet, Denmark Recent retrospective studies on secular trends in male reproductive health
2002 Session 5 Children s Health Regional Differences and Temporal Trends in Semen Quality Niels Jørgensen Rigshospitalet, Denmark Recent retrospective studies on secular trends in male reproductive health
Testicular Toxicity: Evaluation During Drug Development Guidance for Industry
 Testicular Toxicity: Evaluation During Drug Development Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this
Testicular Toxicity: Evaluation During Drug Development Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this
DOHaD: Role of environmental chemical exposures
 DOHaD: Role of environmental chemical exposures Linda S. Birnbaum, Ph.D., D.A.B.T., A.T.S. Director, National Institute of Environmental Health Sciences and National Toxicology Program PPTox-IV Boston,
DOHaD: Role of environmental chemical exposures Linda S. Birnbaum, Ph.D., D.A.B.T., A.T.S. Director, National Institute of Environmental Health Sciences and National Toxicology Program PPTox-IV Boston,
Before, During and Beyond Pregnancy The Impact of Environmental Toxins on Reproductive Health. Physicians for Social Responsibility-Los Angeles
 Before, During and Beyond Pregnancy The Impact of Environmental Toxins on Reproductive Health Physicians for Social Responsibility-Los Angeles PSR-LA 2009 1 Chemicals Are All Around Us The number of chemicals
Before, During and Beyond Pregnancy The Impact of Environmental Toxins on Reproductive Health Physicians for Social Responsibility-Los Angeles PSR-LA 2009 1 Chemicals Are All Around Us The number of chemicals
The Big Picture of Trace chemicals
 Impacts of Trace Chemicals on the Environment David O. Norris Integrative Physiology University of Colorado at Boulder david.norris@colorado.edu The Big Picture of Trace chemicals And how we all fit into
Impacts of Trace Chemicals on the Environment David O. Norris Integrative Physiology University of Colorado at Boulder david.norris@colorado.edu The Big Picture of Trace chemicals And how we all fit into
APPLICATION FOR AUTHORISATION: ESTABLISHING REFERENCE DNELS FOR BBP
 Helsinki, 2 September 20 RAC/26/20/07 Rev. (Agreed at RAC-26) APPLICATION FOR AUTHORISATION: ESTABLISHING REFERENCE DNELS FOR BBP Background At the 22 nd meeting of the Committee for Risk Assessment (RAC)
Helsinki, 2 September 20 RAC/26/20/07 Rev. (Agreed at RAC-26) APPLICATION FOR AUTHORISATION: ESTABLISHING REFERENCE DNELS FOR BBP Background At the 22 nd meeting of the Committee for Risk Assessment (RAC)
COLLECTIVE EXPERT APPRAISAL: SUMMARY AND CONCLUSIONS
 COLLECTIVE EXPERT APPRAISAL: SUMMARY AND CONCLUSIONS Related to theestablishment of a Toxicity Reference Value based on the reprotoxic effects of linuron (CAS No. 330-55-2) AFSSET Solicited Request No.
COLLECTIVE EXPERT APPRAISAL: SUMMARY AND CONCLUSIONS Related to theestablishment of a Toxicity Reference Value based on the reprotoxic effects of linuron (CAS No. 330-55-2) AFSSET Solicited Request No.
Reproduction. AMH Anti-Müllerian Hormone. Analyte Information
 Reproduction AMH Anti-Müllerian Hormone Analyte Information - 1-2011-01-11 AMH Anti-Müllerian Hormone Introduction Anti-Müllerian Hormone (AMH) is a glycoprotein dimer composed of two 72 kda monomers 1.
Reproduction AMH Anti-Müllerian Hormone Analyte Information - 1-2011-01-11 AMH Anti-Müllerian Hormone Introduction Anti-Müllerian Hormone (AMH) is a glycoprotein dimer composed of two 72 kda monomers 1.
Spermatogenesis. What is it and what does it look like? How do hormones regulate spermatogenesis?
 Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
Knockout TM SR : ; ; ; : R ; R : A : X(2013) , ,, B. , (Knockout TM
 33 1 Vol.33 No.1 013 1 Dec. 013 Reproduction & Contraception doi: 10.7669/j.issn.03-37X.013.1.0804 E-mail: randc_journal@163.com Knockout TM SR ; ; ; 400014 : FBS Knockout TM SRKSR : FBS KSR HE TUNEL RT-PCR
33 1 Vol.33 No.1 013 1 Dec. 013 Reproduction & Contraception doi: 10.7669/j.issn.03-37X.013.1.0804 E-mail: randc_journal@163.com Knockout TM SR ; ; ; 400014 : FBS Knockout TM SRKSR : FBS KSR HE TUNEL RT-PCR
Superman and the Martians
 Superman and the Martians Life on earth means exposure to chemicals and living in a chemical soup Several man-made chemicals are building up in our body that haven t been properly tested for health effects
Superman and the Martians Life on earth means exposure to chemicals and living in a chemical soup Several man-made chemicals are building up in our body that haven t been properly tested for health effects
Sex Determination and Development of Reproductive Organs
 Sex Determination and Development of Reproductive Organs Sex determination The SRY + gene is necessary and probably sufficient for testis development The earliest sexual difference appears in the gonad
Sex Determination and Development of Reproductive Organs Sex determination The SRY + gene is necessary and probably sufficient for testis development The earliest sexual difference appears in the gonad
Endocrine disrupting effects of widely used substances
 Endocrine disrupting effects of widely used substances Sarah Janssen, M.D., Ph.D., M.P.H. Science Fellow, Natural Resources Defense Council Assistant Professor, University of California, San Francisco
Endocrine disrupting effects of widely used substances Sarah Janssen, M.D., Ph.D., M.P.H. Science Fellow, Natural Resources Defense Council Assistant Professor, University of California, San Francisco
Oral glucose lowering agents in gestational diabetes. Yes: E. Sobngwi (Cameroon) No: A. Vambergue (France)
 Oral glucose lowering agents in gestational diabetes Yes: E. Sobngwi (Cameroon) No: A. Vambergue (France) CONTROVERSIES Oral glucose lowering agents in gestational diabetes «NO» Pr Anne VAMBERGUE Department
Oral glucose lowering agents in gestational diabetes Yes: E. Sobngwi (Cameroon) No: A. Vambergue (France) CONTROVERSIES Oral glucose lowering agents in gestational diabetes «NO» Pr Anne VAMBERGUE Department
Endocrine disrupting chemicals & the grain industry
 Endocrine disrupting chemicals & the grain industry 21 August 2018 Riana Bornman, SHSPH, UP riana.bornman@up.ac.za Conflict of interest statement Opinions expressed and conclusions arrived at are those
Endocrine disrupting chemicals & the grain industry 21 August 2018 Riana Bornman, SHSPH, UP riana.bornman@up.ac.za Conflict of interest statement Opinions expressed and conclusions arrived at are those
ENVIRONMENTAL AND LIFE STYLE DETERMINANTS OF SEMEN QUALITY. Jorma Toppari Departments of Physiology and Pediatrics University of Turku
 ENVIRONMENTAL AND LIFE STYLE DETERMINANTS OF SEMEN QUALITY Jorma Toppari Departments of Physiology and Pediatrics University of Turku 2013 Disclosures I have received lecture honoraria from Eli Lilly,
ENVIRONMENTAL AND LIFE STYLE DETERMINANTS OF SEMEN QUALITY Jorma Toppari Departments of Physiology and Pediatrics University of Turku 2013 Disclosures I have received lecture honoraria from Eli Lilly,
Towards 2020: Linda S. Birnbaum, Ph.D., D.A.B.T., A.T.S. Director National Institute of Environmental Health Sciences National Toxicology Program
 Towards 2020: What are the Critical Environmental Health Challenges? Linda S. Birnbaum, Ph.D., D.A.B.T., A.T.S. Director National Institute of Environmental Health Sciences National Toxicology Program
Towards 2020: What are the Critical Environmental Health Challenges? Linda S. Birnbaum, Ph.D., D.A.B.T., A.T.S. Director National Institute of Environmental Health Sciences National Toxicology Program
DANIEL R. DOERGE U.S. Food and Drug Administration National Center for Toxicological Research Jefferson, AR
 Research support by Interagency Agreement between NTP/NIEHS and NCTR/FDA The opinions presented are not necessarily those of the U.S. FDA or NTP NCTR/FDA Research on BPA: Integrating pharmacokinetics in
Research support by Interagency Agreement between NTP/NIEHS and NCTR/FDA The opinions presented are not necessarily those of the U.S. FDA or NTP NCTR/FDA Research on BPA: Integrating pharmacokinetics in
Endocrine disruptors and effects on reproduction DEMETER: a support tool for occupational risks assessment
 Endocrine disruptors and effects on reproduction DEMETER: a support tool for occupational risks assessment ISSA Symposium 1 June 2016 Stéphane Malard Medical Studies and Assistance Division, INRS, Paris
Endocrine disruptors and effects on reproduction DEMETER: a support tool for occupational risks assessment ISSA Symposium 1 June 2016 Stéphane Malard Medical Studies and Assistance Division, INRS, Paris
Adverse effects of environmental antiandrogens and androgens on reproductive development in mammals 1
 international journal of andrology ISSN 0105-6263 Adverse effects of environmental antiandrogens and androgens on reproductive development in mammals 1 L. Earl Gray Jr,* Vickie S. Wilson,* Tammy Stoker,*
international journal of andrology ISSN 0105-6263 Adverse effects of environmental antiandrogens and androgens on reproductive development in mammals 1 L. Earl Gray Jr,* Vickie S. Wilson,* Tammy Stoker,*
Justification for the selection of a candidate CoRAP substance
 Justification for the selection of a candidate CoRAP substance Substance Name (Public Name): Diethyl phthalate Chemical Group: EC Number: 201-550-6 CAS Number: 84-66-2 Submitted by: Germany/Portugal Published:
Justification for the selection of a candidate CoRAP substance Substance Name (Public Name): Diethyl phthalate Chemical Group: EC Number: 201-550-6 CAS Number: 84-66-2 Submitted by: Germany/Portugal Published:
benzyl butyl phthalate (BBP)
 EVALUATION OF NEW SCIENTIFIC EVIDENCE CONCERNING THE RESTRICTIONS CONTAINED IN ANNEX XVII TO REGULATION (EC) NO 1907/2006 (REACH) REVIEW OF NEW AVAILABLE INFORMATION FOR benzyl butyl phthalate (BBP) CAS
EVALUATION OF NEW SCIENTIFIC EVIDENCE CONCERNING THE RESTRICTIONS CONTAINED IN ANNEX XVII TO REGULATION (EC) NO 1907/2006 (REACH) REVIEW OF NEW AVAILABLE INFORMATION FOR benzyl butyl phthalate (BBP) CAS
Influence of oily vehicles on fetal testis and lipid profile of rats exposed to di-butyl phthalate
 Article Influence of oily vehicles on fetal testis and lipid profile of rats exposed to di-butyl phthalate Human and Experimental Toxicology 2014, Vol 33(1) 54 63 ª The Author(s) 2014 Reprints and permission:
Article Influence of oily vehicles on fetal testis and lipid profile of rats exposed to di-butyl phthalate Human and Experimental Toxicology 2014, Vol 33(1) 54 63 ª The Author(s) 2014 Reprints and permission:
Endocrine Disruptors (EDs) ENVIRONMENTAL EXPOSURE TO ENDOCRINE DISRUPTORS AND SELECTED METABOLIC MARKERS IN CHILDREN. Polychlorinated Biphenyls (PCBs)
 ENVIRONMENTAL EXPOSURE TO ENDOCRINE DISRUPTORS AND SELECTED METABOLIC MARKERS IN CHILDREN Palkovičov ová Ľubica, Patayová Henrieta, Lancz Kinga, Rausová Katarína na, Wimmerová Soňa, Chovancová Jana, Šovčíková
ENVIRONMENTAL EXPOSURE TO ENDOCRINE DISRUPTORS AND SELECTED METABOLIC MARKERS IN CHILDREN Palkovičov ová Ľubica, Patayová Henrieta, Lancz Kinga, Rausová Katarína na, Wimmerová Soňa, Chovancová Jana, Šovčíková
Edinburgh Research Explorer
 Edinburgh Research Explorer Anogenital distance (AGD) plasticity in adulthood Citation for published version: Mitchell, RT, Mungall, W, McKinnell, C, Sharpe, RM, Cruickshanks, L, Milne, L & Smith, LB 214,
Edinburgh Research Explorer Anogenital distance (AGD) plasticity in adulthood Citation for published version: Mitchell, RT, Mungall, W, McKinnell, C, Sharpe, RM, Cruickshanks, L, Milne, L & Smith, LB 214,
Growth pattern of the sex ducts in foetal mouse hermaphrodites
 /. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
/. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
endocrine disruptors?
 worry about endocrine disruptors? Endocrine active substances are chemicals that can interfere with the body s hormone system and may have the potential to cause harm. They are used in everyday objects
worry about endocrine disruptors? Endocrine active substances are chemicals that can interfere with the body s hormone system and may have the potential to cause harm. They are used in everyday objects
A Testing Strategy for the Identification of mammalian Endocrine Disruptors with particular focus on steroids
 A Testing Strategy for the Identification of mammalian Endocrine Disruptors with particular focus on steroids Tzutzuy Ramirez, Robert Landsiedel, Susanne Kolle, Hennicke Kamp, Bennard van Ravenzwaay EUSAAT
A Testing Strategy for the Identification of mammalian Endocrine Disruptors with particular focus on steroids Tzutzuy Ramirez, Robert Landsiedel, Susanne Kolle, Hennicke Kamp, Bennard van Ravenzwaay EUSAAT
Decreased Insulin Receptor Kinase Activity in Gestational Diabetes Mellitus
 ENDOCRINOLOGY Decreased Insulin Receptor Kinase Activity in Gestational Diabetes Mellitus during pregnancy decrease in insulin sensitivity helps provide adequate glucose for the developing fetus 14% of
ENDOCRINOLOGY Decreased Insulin Receptor Kinase Activity in Gestational Diabetes Mellitus during pregnancy decrease in insulin sensitivity helps provide adequate glucose for the developing fetus 14% of
Integrative rodent models for assessing male reproductive toxicity of environmental endocrine active substances
 (2014) 16, (60 70) 2013 AJA, SIMM & SJTU. All rights reserved 1008 682X www.asiaandro.com; www.ajandrology.com Reproductive Health Open Access INVITED REVIEW Integrative rodent models for assessing male
(2014) 16, (60 70) 2013 AJA, SIMM & SJTU. All rights reserved 1008 682X www.asiaandro.com; www.ajandrology.com Reproductive Health Open Access INVITED REVIEW Integrative rodent models for assessing male
Intratubular Germ Cell Neoplasia of the Testis
 Intratubular Germ Cell Neoplasia of the Testis KS Ngoo Department of Urology Hospital Selayang Advanced Urology Course 15 Aug 2014 MUA Office Clinical scenario A 33 years old man has bilateral testicular
Intratubular Germ Cell Neoplasia of the Testis KS Ngoo Department of Urology Hospital Selayang Advanced Urology Course 15 Aug 2014 MUA Office Clinical scenario A 33 years old man has bilateral testicular
Exposure (To Stressors) Autism Spectrum Disorders
 Exposure (To Stressors) Autism Spectrum Disorders Larry L. Needham, Ph.D. Chief, Organic Analytical Toxicology Branch National Center for Environmental Health Centers for Disease Control and Prevention
Exposure (To Stressors) Autism Spectrum Disorders Larry L. Needham, Ph.D. Chief, Organic Analytical Toxicology Branch National Center for Environmental Health Centers for Disease Control and Prevention
Why Reproduce? In order to ensure the continuation of the species and the continuation of life in general by producing offspring
 HUMAN REPRODUCTION Why Reproduce? In order to ensure the continuation of the species and the continuation of life in general by producing offspring Asexual vs Sexual Reproduction Remember: Asexual reproduction:
HUMAN REPRODUCTION Why Reproduce? In order to ensure the continuation of the species and the continuation of life in general by producing offspring Asexual vs Sexual Reproduction Remember: Asexual reproduction:
The Biology of Sex: How We Become Male or Female.
 The Biology of Sex: How We Become Male or Female. Dr. Tamatha Barbeau, Dept. of Biology Guest Lecture for Gender 200 March 2017 Objectives: 1. Sex vs. Gender defined. 2. Biological sex based on inheritance
The Biology of Sex: How We Become Male or Female. Dr. Tamatha Barbeau, Dept. of Biology Guest Lecture for Gender 200 March 2017 Objectives: 1. Sex vs. Gender defined. 2. Biological sex based on inheritance
Reproductive Endocrinology. Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007
 Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism
 Research article Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism Michelle Welsh, Philippa T.K. Saunders,
Research article Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism Michelle Welsh, Philippa T.K. Saunders,
11. SEXUAL DIFFERENTIATION. Germinal cells, gonocytes. Indifferent stage INDIFFERENT STAGE
 11. SEXUAL DIFFERENTIATION INDIFFERENT STAGE Early in pregnancy, (within 10-15 % of the pregnancy s expected length) a genital ridge is formed in the sides of the embryonic tissue, ventral to the mesonephros
11. SEXUAL DIFFERENTIATION INDIFFERENT STAGE Early in pregnancy, (within 10-15 % of the pregnancy s expected length) a genital ridge is formed in the sides of the embryonic tissue, ventral to the mesonephros
MALE INFERTILITY & SEMEN ANALYSIS
 MALE INFERTILITY & SEMEN ANALYSIS DISCLOSURE Relevant relationships with commercial entities none Potential for conflicts of interest within this presentation none Steps taken to review and mitigate potential
MALE INFERTILITY & SEMEN ANALYSIS DISCLOSURE Relevant relationships with commercial entities none Potential for conflicts of interest within this presentation none Steps taken to review and mitigate potential
W.S. O University of Hong Kong
 W.S. O University of Hong Kong Development of the Genital System 1. Sexual differentiation 2. Differentiation of the gonads a. Germ cells extragonadal in origin b. Genital ridge intermediate mesoderm consisting
W.S. O University of Hong Kong Development of the Genital System 1. Sexual differentiation 2. Differentiation of the gonads a. Germ cells extragonadal in origin b. Genital ridge intermediate mesoderm consisting
Diethylstilbestrol (DES) General Information
 Use of the DES Paradigm to Inform Risk Assessment for Weakly Estrogenic Chemicals Robert Golden PhD ToxLogic Potomac, MD Diethylstilbestrol (DES) General Information DES ranges from slightly less to several
Use of the DES Paradigm to Inform Risk Assessment for Weakly Estrogenic Chemicals Robert Golden PhD ToxLogic Potomac, MD Diethylstilbestrol (DES) General Information DES ranges from slightly less to several
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
When testes make no testosterone: Identifying a rare cause of 46, XY female phenotype in adulthood
 When testes make no testosterone: Identifying a rare cause of 46, XY female phenotype in adulthood Gardner DG, Shoback D. Greenspan's Basic & Clinical Endocrinology, 10e; 2017 Sira Korpaisarn, MD Endocrinology
When testes make no testosterone: Identifying a rare cause of 46, XY female phenotype in adulthood Gardner DG, Shoback D. Greenspan's Basic & Clinical Endocrinology, 10e; 2017 Sira Korpaisarn, MD Endocrinology
Prevalence of Hypospadias in Danish Boys: A Longitudinal Study,
 european urology 55 (2009) 1022 1026 available at www.sciencedirect.com journal homepage: www.europeanurology.com Platinum Priority Pediatric Urology Editorial by Paolo Caione on pp. 1027 1029 of this
european urology 55 (2009) 1022 1026 available at www.sciencedirect.com journal homepage: www.europeanurology.com Platinum Priority Pediatric Urology Editorial by Paolo Caione on pp. 1027 1029 of this
Male Reproduction Organs. 1. Testes 2. Epididymis 3. Vas deferens 4. Urethra 5. Penis 6. Prostate 7. Seminal vesicles 8. Bulbourethral glands
 Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
DANIEL R. DOERGE U.S. Food and Drug Administration National Center for Toxicological Research Jefferson, AR
 NCTR Research Plan for BPA: Integrating pharmacokinetics in rodent and primate species, rat toxicology studies, human biomonitoring, and PBPK modeling to assess potential human risks from dietary intake
NCTR Research Plan for BPA: Integrating pharmacokinetics in rodent and primate species, rat toxicology studies, human biomonitoring, and PBPK modeling to assess potential human risks from dietary intake
COMMENTS AND RESPONSE TO COMMENTS ON CLH: PROPOSAL AND JUSTIFICATION
 COMMENTS AND RESPONSE TO COMMENTS ON CLH: PROPOSAL AND JUSTIFICATION Comments provided during public consultation are made available in this table as submitted by the webform. Please note that the comments
COMMENTS AND RESPONSE TO COMMENTS ON CLH: PROPOSAL AND JUSTIFICATION Comments provided during public consultation are made available in this table as submitted by the webform. Please note that the comments
REVIEW ARTICLE. C. V. Rider,* J. R. Furr,* V. S. Wilson* and L. E. Gray Jr* Summary
 international journal of andrology ISSN 15-6263 REVIEW ARTICLE Cumulative effects of in utero administration of mixtures of reproductive toxicants that disrupt common target tissues via diverse mechanisms
international journal of andrology ISSN 15-6263 REVIEW ARTICLE Cumulative effects of in utero administration of mixtures of reproductive toxicants that disrupt common target tissues via diverse mechanisms
Endocrine disruptors: Science for science's sake, science for health protection
 Endocrine disruptors: Science for science's sake, science for health protection Ana M. Soto Tufts University School of Medicine (Boston) Ecole Normale Supérieure (Paris) Endocrine System The set of cells
Endocrine disruptors: Science for science's sake, science for health protection Ana M. Soto Tufts University School of Medicine (Boston) Ecole Normale Supérieure (Paris) Endocrine System The set of cells
Sperm production. Sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
 Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production. Sperm production. Controlling sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
 Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
C de Jager, C van Zijl, S van Wyk, N Aneck-Hahn University of Pretoria, South Africa
 The effects of veterinary growth stimulants from cattle feedlots in South Africa on reproductive and thyroid parameters in a rat model C de Jager, C van Zijl, S van Wyk, N Aneck-Hahn University of Pretoria,
The effects of veterinary growth stimulants from cattle feedlots in South Africa on reproductive and thyroid parameters in a rat model C de Jager, C van Zijl, S van Wyk, N Aneck-Hahn University of Pretoria,
Clinical evaluation of infertility
 Clinical evaluation of infertility DR. FARIBA KHANIPOUYANI OBSTETRICIAN & GYNECOLOGIST PRENATOLOGIST Definition: inability to achieve conception despite one year of frequent unprotected intercourse. Male
Clinical evaluation of infertility DR. FARIBA KHANIPOUYANI OBSTETRICIAN & GYNECOLOGIST PRENATOLOGIST Definition: inability to achieve conception despite one year of frequent unprotected intercourse. Male
MALE REPRODUCTIVE DISORDERS AND FERTILITY TRENDS: INFLUENCES OF ENVIRONMENT AND GENETIC SUSCEPTIBILITY
 Physiol Rev 96: 55 97, 2016 Published November 18, 2015; doi:10.1152/physrev.00017.2015 MALE REPRODUCTIVE DISORDERS AND FERTILITY TRENDS: INFLUENCES OF ENVIRONMENT AND GENETIC SUSCEPTIBILITY Niels E. Skakkebaek,
Physiol Rev 96: 55 97, 2016 Published November 18, 2015; doi:10.1152/physrev.00017.2015 MALE REPRODUCTIVE DISORDERS AND FERTILITY TRENDS: INFLUENCES OF ENVIRONMENT AND GENETIC SUSCEPTIBILITY Niels E. Skakkebaek,
Treatment of Oligospermia with Large Doses of Human Chorionic Gonadotropin
 Treatment of Oligospermia with Large Doses of Human Chorionic Gonadotropin A Preliminary Report S. J. GLASS, M.D., and H. M. HOLLAND, M.D. BEFORE discussing gonadotropic therapy of oligospermia, it is
Treatment of Oligospermia with Large Doses of Human Chorionic Gonadotropin A Preliminary Report S. J. GLASS, M.D., and H. M. HOLLAND, M.D. BEFORE discussing gonadotropic therapy of oligospermia, it is
Epigenetic Pathways Linking Parental Effects to Offspring Development. Dr. Frances A. Champagne Department of Psychology, Columbia University
 Epigenetic Pathways Linking Parental Effects to Offspring Development Dr. Frances A. Champagne Department of Psychology, Columbia University Prenatal & Postnatal Experiences Individual differences in brain
Epigenetic Pathways Linking Parental Effects to Offspring Development Dr. Frances A. Champagne Department of Psychology, Columbia University Prenatal & Postnatal Experiences Individual differences in brain
EFFECTS OF POLLUTANTS ON THE REPRODUCTIVE HEALTH OF MALE VERTEBRATE WILDLIFE - MALES UNDER THREAT SUMMARY AND OVERVIEW
 EFFECTS OF POLLUTANTS ON THE REPRODUCTIVE HEALTH OF MALE VERTEBRATE WILDLIFE - SUMMARY AND OVERVIEW A CHEM Trust report by Gwynne Lyons CHEM Trust s aim is to protect humans and wildlife from harmful chemicals.
EFFECTS OF POLLUTANTS ON THE REPRODUCTIVE HEALTH OF MALE VERTEBRATE WILDLIFE - SUMMARY AND OVERVIEW A CHEM Trust report by Gwynne Lyons CHEM Trust s aim is to protect humans and wildlife from harmful chemicals.
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
-The cause of testicular neoplasms remains unknown
 - In the 15- to 34-year-old age group, they are the most common tumors of men. - include: I. Germ cell tumors : (95%); all are malignant. II. Sex cord-stromal tumors: from Sertoli or Leydig cells; usually
- In the 15- to 34-year-old age group, they are the most common tumors of men. - include: I. Germ cell tumors : (95%); all are malignant. II. Sex cord-stromal tumors: from Sertoli or Leydig cells; usually
2 The first hints that pollutants might be able to interfere with male hormones turned up over 20 years ago. Mike Howell, a fish biologist at Samford
 1 Boys won't be boys 29 June 2002 From New Scientist Print Edition. Subscribe and get 4 free issues. Julie Wakefield http://www.newscientist.com/article/mg17423495.300;jsessionid=jgikanpnojpp PEOPLE don't
1 Boys won't be boys 29 June 2002 From New Scientist Print Edition. Subscribe and get 4 free issues. Julie Wakefield http://www.newscientist.com/article/mg17423495.300;jsessionid=jgikanpnojpp PEOPLE don't
8/26/13. Announcements
 Announcements THM questions will start for points on Wednesday. Make sure you are registered correctly! Problems registering for BioPortal? Make sure you are using the link from the syllabus or FAQ. 30
Announcements THM questions will start for points on Wednesday. Make sure you are registered correctly! Problems registering for BioPortal? Make sure you are using the link from the syllabus or FAQ. 30
Decreased Levels of Plasma Testosterone/LH Ratio in Male Mice Exposed to Sodium Arsenite
 Biomolecules & Therapeutics, 18(3), 257-261 (2010) www.biomolther.org ISSN: 1976-9148(print)/2005-4483(online) DOI: 10.4062/biomolther.2010.18.3.257 Short Communication Decreased Levels of Plasma Testosterone/LH
Biomolecules & Therapeutics, 18(3), 257-261 (2010) www.biomolther.org ISSN: 1976-9148(print)/2005-4483(online) DOI: 10.4062/biomolther.2010.18.3.257 Short Communication Decreased Levels of Plasma Testosterone/LH
Main Challenges Related to Measuring Biomarkers of Exposure of Bisphenol A and Triclosan
 Main Challenges Related to Measuring Biomarkers of Exposure of Bisphenol A and Triclosan Antonia M. Calafat, PhD Organic Analytical Toxicology Branch Division of Laboratory Sciences National Center for
Main Challenges Related to Measuring Biomarkers of Exposure of Bisphenol A and Triclosan Antonia M. Calafat, PhD Organic Analytical Toxicology Branch Division of Laboratory Sciences National Center for
TESTOSTERONE DEFINITION
 DEFINITION A hormone that is a hydroxyl steroid ketone (C19H28O2) produced especially by the testes or made synthetically and that is responsible for inducing and maintaining male secondary sex characteristics.
DEFINITION A hormone that is a hydroxyl steroid ketone (C19H28O2) produced especially by the testes or made synthetically and that is responsible for inducing and maintaining male secondary sex characteristics.
