ARTICLE Human RFT1 Deficiency Leads to a Disorder of N-Linked Glycosylation
|
|
|
- Gabriella Norman
- 5 years ago
- Views:
Transcription
1 ARTICLE Human RFT1 Deficiency Leads to a Disorder of N-Linked Glycosylation Micha A. Haeuptle, 1,5 François M. Pujol, 2,5 Christine Neupert, 3 Bryan Winchester, 4 Alexander J. Kastaniotis, 2 Markus Aebi, 3 and Thierry Hennet 1, * N-linked glycosylation is an essential posttranslational modification of proteins in eukaryotes. The substrate of N-linked glycosylation, dolichol pyrophosphate (DolPP)-GlcNAc 2 Man 9 Glc 3, is assembled through a complex series of ordered reactions requiring the translocation of the intermediate DolPP-GlcNAc 2 Man 5 structure across the endoplasmic-reticulum membrane. A young patient diagnosed with a congenital disorder of glycosylation characterized by an intracellular accumulation of DolPP-GlcNAc 2 Man 5 was found to carry a homozygous point mutation in the RFT1 gene. The c.199c/t mutation introduced the amino acid substitution p.r67c. The human RFT1 protein shares 22% identity with its yeast ortholog, which is involved in the translocation of DolPP-GlcNAc 2 Man 5 from the cytosolic into the lumenal side of the endoplasmic reticulum. Despite the low sequence similarity between the yeast and the human RFT1 proteins, we demonstrated both their functional orthology and the pathologic effect of the human p.r67c mutation by complementation assay in Drft1 yeast cells. The causality of the RFT1 p.r67c mutation was further established by restoration of normal glycosylation profiles in patient-derived fibroblasts after lentiviral expression of a normal RFT1 cdna. The definition of the RFT1 defect establishes the functional conservation of the DolPP-GlcNAc 2 Man 5 translocation process in eukaryotes. RFT1 deficiency in both yeast and human cells leads to the accumulation of incomplete DolPP-GlcNAc 2 Man 5 and to a profound glycosylation disorder in humans. Introduction N-linked glycosylation is a ubiquitous posttranslational modification of proteins in eukaryotes. N-glycans convey essential signals for the folding and intracellular trafficking of glycoproteins. 1 N-glycans also influence the clearance of circulating glycoproteins 2 and the stability of signaling proteins at the cell surface. 3 A particular feature of N-glycans is that they are first assembled in the endoplasmic reticulum (ER) as lipid-linked oligosaccharides (LLO). This assembly proceeds through the sequential addition of monosaccharides to the growing LLO. The process begins with the addition of GlcNAc monophosphate to the lipid carrier dolichol phosphate (DolP) and ends with the formation of DolPP-GlcNAc 2 Man 9 Glc 3. The oligosaccharide is transferred to selected asparagine residues of nascent glycoproteins. 4 The assembly of LLO requires glycosyltransferases and their respective nucleotideand dolichol-activated monosaccharide substrates, but it also requires several proteins that regulate the complex topology of the process. For example, the MPDU1 protein makes the donor substrates DolP-Man and DolP-Glc available for completion of the LLO beyond DolPP-GlcNAc 2- Man Similarly, it was previously shown that in yeast the Rft1 protein is essential for translocation of the cytosolically oriented intermediate DolPP-GlcNAc 2 Man 5 into the ER lumen, where LLO assembly is completed. 8 The pathway of LLO assembly is strongly conserved among eukaryotes. Orthologous genes can be found from yeasts to humans for all glycosyltransferases involved. However, it is unclear whether the same degree of conservation applies to the accessory proteins. In fact, whereas MPDU1 orthologs can be found in metazoan and plant genomes, no orthologous gene can be identified in the yeast genome. In the case of RFT1, only genes with limited sequence similarity can be retrieved from the genome of higher organisms, thus casting doubt upon the functional significance of RFT1 in the assembly of LLO in general. The identification of N-linked glycosylation disorders in humans, often referred to as congenital disorders of glycosylation (CDG), has demonstrated the conservation of the LLO-assembly pathway between yeasts and humans. 9 The expression of human glycosyltransferase genes in glycosylation mutant yeasts demonstrated both the orthology of the glycosyltransferases in question and the pathological effect of the mutations identified in CDG cases In spite of the extensive use of the yeast as a road map, many cases of CDG have remained untyped. Clinically, these cases present the symptoms typically seen in CDG patients, i.e., neurological abnormalities, failure to thrive, and varying degrees of dysmorphism. 7,14 In the present study, we have identified a novel glycosylation defect in such an untyped CDG case, thereby establishing the importance of the RFT1 protein in human N-linked glycosylation. Material and Methods LLO and NLO Analysis The investigation of the patient material was approved by the Ethical Commission of the Kanton Zürich. Fibroblasts were grown in DMEM (GIBCO) containing 25 mm Glc and 10% FCS until 90% of 1 Institute of Physiology and Zürich Center for Integrative Human Physiology, University of Zürich, Winterthurerstrasse 190, CH-8057 Zürich, Switzerland; 2 Biocenter Oulu and Department of Biochemistry, University of Oulu, P.O. Box 3000, FIN-90014, Finland; 3 Institute of Microbiology, Swiss Federal Institute of Technology, CH-8093 Zürich, Switzerland; 4 Biochemistry Research Group, Institute of Child Health, University College London, 30 Guilford Street, London WC1N 1EH, UK 5 These authors contributed equally to this work. *Correspondence: thennet@access.uzh.ch DOI /j.ajhg ª2008 by The American Society of Human Genetics. All rights reserved. 600 The American Journal of Human Genetics 82, , March 2008
2 Figure 1. Lipid-Linked and N-Linked Oligosaccharide Profiles The LLO isolated from healthy (A) and CDG (B) fibroblasts were separated by HPLC, demonstrating the abnormal accumulation of the LLO DolPP-GlcNAc 2 Man 5 in the CDG sample. The NLO isolated from healthy (C) and CDG (D) fibroblasts were identical, showing that complete oligosaccharides were transferred to nascent proteins in the CDG cells. The retention times of DolPP-GlcNAc 2 Man 1 (M 1 ) to DolPP- GlcNAc 2 Man 9 Glc 3 (M 9 G 3 ) derived from a yeast standard LLO are marked at the top of the profiles. confluence. At that point, the fibroblasts were rinsed in PBS and incubated in Glc- and FCS-free DMEM for 45 min, then labeled by addition of 150 mci of [ 3 H]-Man (54.0 Ci/mmol, Amersham Bioscience) for 60 min. LLO and N-linked oligosaccharides (NLO) were isolated from labeled fibroblasts by chloroform-methanol-water extraction as described previously. 13 Oligosaccharides were released from LLO by mild acid hydrolysis and from NLO by N-glycosidase F (New England BioLabs) digestion, 13 then subjected to HPLC. 15 Mutation Analysis Total RNA and genomic DNA were isolated from fibroblasts and 5 ml blood samples, respectively, with the TRIzol LS reagent (Invitrogen) used according to the manufacturer s instructions. The human RFT1 cdna was prepared from 2 mg of total RNA with the primer 5 0 -GGGCTTTTGGTCTTCACT-3 0 and 2 units of Omniscript reverse transcriptase (QIAGEN). The 20 ml reaction mixtures were incubated at 37 C for 1 hr. The protein-coding region of the human RFT1 cdna was amplified by PCR from 2 mlof RT product with the primers 5 0 -GGCGGCATTTCCTGGTGTCT-3 0 and 5 0 -TGGCACTCTCTGGTGCCTCATC-3 0. The exon 3 of the human RFT1 gene was amplified by PCR from 50 ng of genomic DNA with the primers 5 0 -GGGCAATTCAGCTTTAGG-3 0 and 5 0 -CAC CACCAGTGGTTTATG-3 0. The PCR products were sequenced (Synergene Biotech, Switzerland) after removal of the unincorporated nucleotides with QIAquick columns (QIAGEN). The presence of the mutation was confirmed by detection of the Pstl site, created by the c.199c/t mutation in exon 3. Plasmid Construction For construction of the lentiviral expression vector, the human RFT1 cdna was subcloned as a PCR fragment flanked by SpeI and XhoI restriction sites into the NheI and SalI sites of the plenti6-egfp plasmid (Invitrogen), thus yielding the plenti6- hrft1 vector. The yeast rft1 gene with promoter and terminator sequences was amplified from S. cerevisiae genomic DNA by PCR and ligated into YCplac33 16,17 with the inserted 5 0 -PstI and 3 0 -BamHI restriction sites to generate YCplac33-ScRFT1. The ptsv30a-scrft1 16,17 plasmid was obtained by subcloning of ScRFT1 from YCplac33-ScRFT1 with SacI and BamHI restriction sites. Plasmid YCplac33-pGAL1 was constructed by amplification of the GAL1 promoter from pyes2 (Invitrogen) and ligation of the PCR product into YCplac33 with the inserted restriction sites 5 0 -HindIII and 3 0 -XbaI. The human RFT1 cdna was obtained via the EST clone IMAGE: (Geneservice, UK). For construction of the plasmid YCplac33 pgal1-hrft1, the human RFT1 ORF was amplified from the IMAGE clone by PCR and cloned into plasmid YCplac33 pgal1 with the inserted restriction sites 5 0 -XbaI and 3 0 -SacI. The mutant RFT1[R67C] cdna from the CDG patient was subcloned into YCplac33 pgal1 hrft1, resulting in YCplac33-pGAL1-hRFT1[R67C]. Complementation of rft1d Yeast Mutants Yeast strains W1536 5B (MATa, ade2d, ade3d, can1-100, his3-11, 15, leu2-3, 112, trp1-1, ura3-1) and W1536 8B (isogenic MATa strain) have been described earlier. 17 The diploid W1536a/a was obtained by the mating of W1536 5B and W1536 8B. W1536 5B-rft1D was generated by introduction of the rft1::kanmx4 gene-replacement cassette into W1536a/a and selection for geneticin resistance followed by tetrad dissection on a Singer MSM manual-dissection microscope (Singer Instrument). Haploid W1536 5B-rft1D mutant cells carry plasmid-borne rft1 in order to be viable. The yeast cells were grown on either rich YPD (1% yeast extract, 2% peptone, and 2% D-Glc), YPGalD (1% yeast extract, 2% peptone, 2% D-Gal, and 0.05% D-Glc), YPR (1% yeast extract, 2% peptone, and 2% D-raffinose), or synthetic complete media (Sigma or QBiogene) lacking one or more nutrients. The colonysectoring method has been described by Bender et al. 18 CPY immunoblotting was performed as described elsewhere. 19 Lentiviral-Mediated RFT1 Expression HEK293T cells ( ) were transfected with 20 mg of plenti6- hrft1 and 36 mg of the packing-plasmid mix (Invitrogen) via calcium-phosphate precipitation. Eight hours after transfection, the medium was replaced with fresh DMEM containing 10% FCS. The cell supernatant was collected after 48 hr, and lentiviruses were harvested by centrifugation at g for 5 min and filtration through 0.45 mm membranes (Schleicher & Schuell, Germany). CDG and healthy-control fibroblasts were infected The American Journal of Human Genetics 82, , March
3 with recombinant lentiviral particles including the human RFT1 cdna or the EGFP gene as controls. Infected cells were selected with 5 mg/ml blasticidin (Invitrogen) for 10 days. Results A young girl was diagnosed with a disorder of N-linked glycosylation on the basis of the detection of abnormal isoelectric focusing of serum transferrin. 20 The patient, designated by the abbreviation KS, 21 showed symptoms often encountered in CDG, namely, a marked developmental delay, hypotonia, seizures, hepatomegaly, and coagulopathy. 21 Phosphomannomutase and phosphomannose isomerase deficiencies were ruled out by the performance of enzymatic testing (data not shown). To determine whether the glycosylation disorder was related to a defect of LLO assembly, we analyzed the LLO composition in the healthy-control and patient fibroblasts. The LLO profile of control fibroblasts was dominated by the full-length DolPP-GlcNAc 2 Man 9 Glc 3 (Figure 1A). By contrast, the profile of the CDG patient was marked by an accumulation of the intermediate LLO DolPP-GlcNAc 2 Man 5 and a strong reduction of complete LLO DolPP-GlcNAc 2 Man 9 Glc 3 (Figure 1B). The analyses of NLO produced after 1 hr labeling of healthy-control and CDG fibroblasts with [ 3 H]Man were indistinguishable. Both profiles showed peaks corresponding to GlcNAc 2 Man 8 and GlcNAc 2 Man 9 oligosaccharides (Figures 1C and 1D), which are normally found on glycoproteins after the trimming of the N-linked glycans by the ER glucosidases I and II and by the ER mannosidase. The absence of GlcNAc 2 Man 5 in the NLO profile of the CDG patient suggested that only the full-length LLO DolPP- GlcNAc 2 Man 9 Glc 3 was transferred to glycoproteins. This phenotype was reminiscent of the LLO and NLO profiles described in yeast depleted for the Rft1 protein. 8 In conditions with limiting RFT1 activity, DolPP-GlcNAc 2 Man 5 accumulates at the cytosolic side of the ER membrane, whereas the small amounts of flipped oligosaccharide are extended to DolPP-GlcNAc 2 Man 9 Glc 3 and transferred to proteins. This results in the underglycosylation of N- glycoproteins. The search for a human ortholog to the yeast Rft1 protein pointed to a single gene (GenBank, NM_052859) that encodes a protein of 541 amino acids sharing 22% identity with yeast Rft1. Sequencing of the NM_ cdna in the CDG fibroblasts revealed a C-to-T transition at nucleotide position 199 (Figure 2A). This point mutation led to the amino acid substitution p.r67c in the human RFT1 protein ortholog. Analysis of the NM_ gene in the DNA of the CDG patient s parents confirmed the heterozygosity (Figure 2B). The c.199c/t mutation was not seen in 210 control chromosomes of European origin, thus excluding this mutation as a single-nucleotide polymorphism. The p.r67c mutation was localized to a 50-amino-acid-long hydrophilic stretch in the overall hydrophobic Rft1 protein ortholog (Figure 2C). Figure 2. Mutation Analysis of Human RFT1 in CDG (A) Electropherograms of RFT1 cdna surrounding nucleotide position 199, marked by an arrow, sequenced from healthy-control and patient cdna. (B) The same region was sequenced from the genomic DNA of the CDG patient s parents, showing the heterozygozity for the c.199c/t mutation. (C) The resulting p.r67c substitution, marked with a star, was predicted to be localized in a hydrophilic loop within the highly hydrophobic RFT1 protein. A potential N-glycosylation site detected at position N227 is shown schematically. Genes encoding proteins with varying degrees of similarity to the human RFT1 protein can be retrieved from all eukaryote genomes analyzed, for which the sequence identity ranges from 87% for the mouse ortholog down to 17% for fungal orthologs (Figure 3). Although the overall protein sequence identity is limited, some regions of RFT1 are strongly conserved across species. It is noteworthy that the region composing the p.r67c mutation was strongly conserved and that several arginine residues, including R67 in the human protein, were found in all sequences analyzed (Figure 3), suggesting the importance of these amino acids for proper functionality. Growth of the yeast strain W1536-5B-rft1D relies on the presence of the yeast rft1 gene on the plasmid ptsv30a- ScRFT1. Cells of this strain background carrying this 602 The American Journal of Human Genetics 82, , March 2008
4 Figure 3. RFT1 Protein-Sequence Comparison Amino acid sequences of proteins derived from Homo sapiens, Mus musculus, Danio rerio, and Caenorhabditis elegans showing similarity to the Saccharomyces cerevisiae RFT1 protein as performed by ClustalW analysis. 30 Residues conserved in all five species are shown in black. The R residue at position 67 in the human RFT1 protein (see arrow) is conserved in the five species. Total amino acid identity with the human sequence ranged from 87% for the mouse protein down to 22% for the S. cerevisiae Rft1 protein. plasmid develop a red pigment, due to the presence of the ADE3 gene on ptsv30a, whereas loss of this plasmid results in white cells. 18 Accordingly, W1536-5B-rft1D/pTSV30A- ScRFT1 forms colonies that are uniformly red. This strain was transformed with the additional plasmid YCplac33p- GAL1-hRFT1 that leads to expression of the human RFT1 cdna controlled by the galactose-inducible GAL1 promoter. Such cells formed colonies that contain white sectors, due to the fact that the ptsv30a-scrft1 plasmid was no longer essential for growth and the plasmid could be lost, as visualized by the white sectors (Figure 4A). This color change indicated functional complementation of the rft1 defect by expression of the human RFT1 cdna. This experiment demonstrated that the investigated human cdna does indeed encode the orthologous protein to yeast Rft1. The same experiment performed with a plasmid expressing the CDG RFT1[R67C] allele did not yield any sectoring colonies (Figure 4A), thus demonstrating that the p.r67c mutation led to reduced function even in the yeast system. As shown previously, 8 Rft1 depletion in yeast leads to the underglycosylation of the vacuolar N-linked glycoprotein carboxypeptidase Y. When W1536 5B-rft1D yeasts were complemented with YCplac33 pgal1-hrft1, a normal carboxypeptidase Y glycosylation profile was restored (Figure 4B), thus showing that the effect of the human RFT1 on rft1d yeast was indeed related to N-glycosylation. Finally, we introduced a normal human RFT1 cdna in the fibroblasts of the CDG patient to demonstrate that the glycosylation disorder was the consequence solely of the identified RFT1 mutation. Healthy-control and CDG fibroblasts were infected with recombinant lentiviruses expressing either the normal RFT1 cdna or EGFP as a negative control. The analysis of LLO profiles in the infected fibroblasts showed an increased formation of the full-length DolPP-GlcNAc 2 Man 9 Glc 3 and a reduced presence of DolPP-GlcNAc 2 Man 5 (Figure 5A), whereas the EGFP expression control had no effect on the LLO profile (Figure 5B). The expression of either normal human RFT1 or EGFP in healthy control fibroblasts had no effect on the LLO profiles (data not shown). Discussion The identification of a human glycosylation disorder associated with an RFT1 defect underlines the functional conservation of the RFT1 protein in eukaryotes. In spite of a limited sequence similarity between the yeast and the human RFT1 proteins, RFT1 deficiency led to identical biochemical phenotypes in both species, i.e., to an accumulation of the LLO DolPP-GlcNAc 2 Man 5 and to a severe protein underglycosylation. Clinically, the RFT1-deficient CDG patient presented with symptoms frequently encountered in CDG patients, namely, failure to thrive, psychomotor retardation, seizures, hypotonia, and coagulopathy. Among the various types of CDG, the clinical severity of the RFT1 The American Journal of Human Genetics 82, , March
5 Figure 4. Complementation of rft1d Yeasts (A) W1536 5B rft1d/ptsv30a-scrft1 cells transformed with a plasmid carrying a complementing RFT1 variant will form sectored colonies as they are allowed to lose the red pigment-inducing ptsv30a -ScRFT1 plasmid. Cells were transformed with: YCplac33 (mock), YCp33 GAL ScRFT1, YCp33 GAL hrft1[r67c], and YCp33 GAL hrft1. (B) Immunoblotting analysis of the yeast strain W1536 rftd transformed with yeast and human RFT1 expressed from the yeast GAL1 promoter showed near wild-type levels of carboxypeptidase Y glycosylation when grown on 2% Gal [G] and an accumulation of underglycosylated isoforms when RFT1 expression was repressed by 4% Glc [D]. Carbon source did not affect carboxypeptidase Y glycosylation in wild-type yeast. deficiency resembled the diseases caused by ALG3 and DPM1 deficiencies, also known as CDG-Id 22 and CDG-Ie, 23,24 respectively. The ALG3 gene encodes the DolP-Man-dependent mannosyltransferase that catalyzes the elongation of LLO from DolPP-GlcNAc 2 Man 5 to DolPP-GlcNAc 2 Man The DPM1 protein is the catalytic subunit of the DolP-Man synthase complex. 26 In CDG-Ie, the shortage in DolP-Man impairs the elongation of the LLO DolPP-GlcNAc 2 Man 5 in the ER lumen. Although DPM1, ALG3, and RFT1 defects all lead to the accumulation of the LLO DolPP-GlcNAc 2 Man 5, their impact on protein N-glycosylation is expected to be different. In fact, an RFT1 defect yields complete LLO structures for the transfer to proteins, yet in limited amount, whereas ALG3 and DPM1 defects yield low amounts of complete LLO combined with the lumenal accumulation of the DolPP-GlcNAc 2 Man 5.The clinical similarities among these three glycosylation defects suggest that the limited availability of complete LLO alone dictates the extent of the clinical manifestations. In the case of DPM1 deficiency, the decreased DolP-Man availability is expected to also affect O-mannosylation and GPIanchor formation. Yet, the comparison between the clinical features of DPM1 deficiency and those of ALG3 and RFT1 deficiencies suggests that the majority of the symptoms are due to abnormal N-glycosylation. Figure 5. Lentiviral-Mediated Complementation in CDG Fibroblasts The LLO profiles of fibroblasts infected with recombinant lentiviruses expressing either human RFT1 (A) or EGFP as negative control (B) were analyzed. The profiles show that the expression of the normal RFT1 cdna in CDG fibroblasts restored the synthesis of the complete LLO DolPP-GlcNAc 2 Man 9 Glc 3. The expression of EGFP in CDG fibroblasts had no effect on the pathologic profile characterized by the accumulation of DolPP-GlcNAc 2 Man 5. The retention times of DolPP-GlcNAc 2 Man 1 (M 1 ) to DolPP-GlcNAc 2 Man 9 Glc 3 (M 9 G 3 ) are marked at the top of the profiles. The functional conservation of the yeast and human RFT1 proteins emphasizes the essential role of RFT1 in lower and higher eukaryotes. However, the function of the RFT1 protein still remains unclear. The contribution of RFT1 to the specific translocation of DolPP-GlcNAc 2 Man 5 suggests that it may function as a flippase, 8 although such an activity could not be confirmed in vitro. A similar uncertainty relates to the function of the MPDU1 protein, 5 which is involved in making DolP-Man and DolP-Glc substrates available to ER lumenal mannosyl- and glucosyltransferases. It is presently unclear whether MPDU1 affects the flipping of these substrates across the ER membrane or whether MPDU1 affects their local concentration by a different mechanism. It is noteworthy that a deficiency of MPDU1 in humans leads to CDG, which is associated with the parallel accumulation of LLO DolPP-GlcNAc 2 Man 5 and DolPP- GlcNAc 2 Man 9. 6,7 To date, 21 forms of CDG have been divided into 13 types of CDG-I and eight types of CDG-II on the basis of the nature of the glycosylation defects In accordance with the nomenclature guidelines established previously, 27 we propose to name the RFT1 deficiency CDG-In. Acknowledgments We thank Peter Clayton for the clinical assessment of the CDG patient. F.M.P. and A.J.K. would like to thank Kalervo Hiltunen for his help with the project. This work was supported by the Körber Foundation and by grants from the Swiss National Science Foundation to T.H. (PP00A ) and to M.A. (3100A ), as well as by grants from the Academy of Finland and the Sigrid Juselius Foundation to F.M.P. and A.J.K. 604 The American Journal of Human Genetics 82, , March 2008
6 Received: October 1, 2007 Revised: November 13, 2007 Accepted: December 4, 2007 Published online: February 28, 2008 Web Resources The URLs for data presented herein are as follows: GenBank, Online Mendelian Inheritance in Man (OMIM), nlm.nih.gov/omim/ References 1. Helenius, A., and Aebi, M. (2004). Roles of N-linked glycans in the endoplasmic reticulum. Annu. Rev. Biochem. 73, Mi, Y., Shapiro, S.D., and Baenziger, J.U. (2002). Regulation of lutropin circulatory half-life by the mannose/n-acetylgalactosamine-4-so 4 receptor is critical for implantation in vivo. J. Clin. Invest. 109, Chantret, I., Dancourt, J., Dupre, T., Delenda, C., Bucher, S., Vuillaumier-Barrot, S., Ogier de Baulny, H., Peletan, C., Danos, O., Seta, N., et al. (2003). A deficiency in dolichyl-p-glucose:glc 1 Man 9 GlcNAc 2 -PP-dolichyl alpha3-glucosyltransferase defines a new subtype of congenital disorders of glycosylation. J. Biol. Chem. 278, Burda, P., and Aebi, M. (1999). The dolichol pathway of N-linked glycosylation. Biochim. Biophys. Acta 1426, Anand, M., Rush, J.S., Ray, S., Doucey, M.A., Weik, J., Ware, F.E., Hofsteenge, J., Waechter, C.J., and Lehrman, M.A. (2001). Requirement of the Lec35 gene for all known classes of monosaccharide-p-dolichol-dependent glycosyltransferase reactions in mammals. Mol. Biol. Cell 12, Kranz, C., Denecke, J., Lehrman, M.A., Ray, S., Kienz, P., Kreissel, G., Sagi, D., Peter-Katalinic, J., Freeze, H.H., Schmid, T., et al. (2001). A mutation in the human MPDU1 gene causes congenital disorder of glycosylation type If (CDG-If). J. Clin. Invest. 108, Schenk, B., Imbach, T., Frank, C.G., Grubenmann, C.E., Raymond, G.V., Hurvitz, H., Korn-Lubetzki, I., Revel-Vik, S., Raas-Rotschild, A., Luder, A.S., et al. (2001). MPDU1 mutations underlie a novel human congenital disorder of glycosylation, designated type If. J. Clin. Invest. 108, Helenius, J., Ng, D.T., Marolda, C.L., Walter, P., Valvano, M.A., and Aebi, M. (2002). Translocation of lipid-linked oligosaccharides across the ER membrane requires Rft1 protein. Nature 415, Aebi, M., and Hennet, T. (2001). Congenital disorders of glycosylation: Genetic model systems lead the way. Trends Cell Biol. 11, Imbach, T., Burda, P., Kuhnert, P., Wevers, R.A., Aebi, M., Berger, E.G., and Hennet, T. (1999). A mutation in the human ortholog of the Saccharomyces cerevisiae ALG6 gene causes carbohydrate-deficient glycoprotein syndrome type-ic. Proc. Natl. Acad. Sci. USA 96, Grubenmann, C.E., Frank, C.G., Kjaergaard, S., Berger, E.G., Aebi, M., and Hennet, T. (2002). ALG12 mannosyltransferase defect in congenital disorder of glycosylation type lg. Hum. Mol. Genet. 11, Frank, C.G., Grubenmann, C.E., Eyaid, W., Berger, E.G., Aebi, M., and Hennet, T. (2004). Identification and functional analysis of a defect in the human ALG9 gene: Definition of congenital disorder of glycosylation type-il. Am. J. Hum. Genet. 75, Grubenmann, C.E., Frank, C.G., Hülsmeier, A.J., Schollen, E., Matthijs, G., Mayatepek, E., Berger, E.G., Aebi, M., and Hennet, T. (2004). Deficiency of the first mannosylation step in the N-glycosylation pathway causes congenital disorder of glycosylation type Ik. Hum. Mol. Genet. 13, Wu, X., Rush, J.S., Karaoglu, D., Krasnewich, D., Lubinsky, M.S., Waechter, C.J., Gilmore, R., and Freeze, H.H. (2003). Deficiency of UDP-GlcNAc:Dolichol Phosphate N-Acetylglucosamine-1 Phosphate Transferase (DPAGT1) causes a novel congenital disorder of Glycosylation Type Ij. Hum. Mutat. 22, Zufferey, R., Knauer, R., Burda, P., Stagljar, I., te Heesen, S., Lehle, L., and Aebi, M. (1995). STT3, a highly conserved protein required for yeast oligosaccharyl transferase activity in vivo. EMBO J. 14, Gietz, R.D., and Sugino, A. (1988). New yeast-escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene 74, Kastaniotis, A.J., Autio, K.J., Sormunen, R.T., and Hiltunen, J.K. (2004). Htd2p/Yhr067p is a yeast 3-hydroxyacyl-ACP dehydratase essential for mitochondrial function and morphology. Mol. Microbiol. 53, Bender, A., and Pringle, J.R. (1991). Use of a screen for synthetic lethal and multicopy suppressee mutants to identify two new genes involved in morphogenesis in Saccharomyces cerevisiae. Mol. Cell. Biol. 11, Burda, P., te Heesen, S., Brachat, A., Wach, A., Dusterhoft, A., and Aebi, M. (1996). Stepwise assembly of the lipid-linked oligosaccharide in the endoplasmic reticulum of Saccharomyces cerevisiae: Identification of the ALG9 gene encoding a putative mannosyl transferase. Proc. Natl. Acad. Sci. USA 93, Stibler, H., Holzbach, U., and Kristiansson, B. (1998). Isoforms and levels of transferrin, antithrombin, alpha(1)-antitrypsin and thyroxine-binding globulin in 48 patients with carbohydrate-deficient glycoprotein syndrome type I. Scand. J. Clin. Lab. Invest. 58, Imtiaz, F., Worthington, V., Champion, M., Beesley, C., Charlwood, J., Clayton, P., Keir, G., Mian, N., and Winchester, B. (2000). Genotypes and phenotypes of patients in the UK with carbohydrate-deficient glycoprotein syndrome type 1. J. Inherit. Metab. Dis. 23, Korner, C., Knauer, R., Stephani, U., Marquardt, T., Lehle, L., and von Figura, K. (1999). Carbohydrate deficient glycoprotein syndrome type IV: Deficiency of dolichyl-p-man: Man(5)GlcNAc(2)-PP-dolichyl mannosyltransferase. EMBO J. 18, Imbach, T., Schenk, B., Schollen, E., Burda, P., Stutz, A., Grunewald, S., Bailie, N.M., King, M.D., Jaeken, J., Matthijs, G., et al. (2000). Deficiency of dolichol-phosphate-mannose synthase-1 causes congenital disorder of glycosylation type Ie. J. Clin. Invest. 105, Kim, S., Westphal, V., Srikrishna, G., Mehta, D.P., Peterson, S., Filiano, J., Karnes, P.S., Patterson, M.C., and Freeze, H.H. The American Journal of Human Genetics 82, , March
7 (2000). Dolichol phosphate mannose synthase (DPM1) mutations define congenital disorder of glycosylation Ie (CDG-Ie). J. Clin. Invest. 105, Aebi, M., Gassenhuber, J., Domdey, H., and te Heesen, S. (1996). Cloning and characterization of the ALG3 gene of Saccharomyces cerevisiae. Glycobiology 6, Maeda, Y., Tanaka, S., Hino, J., Kangawa, K., and Kinoshita, T. (2000). Human dolichol-phosphate-mannose synthase consists of three subunits, DPM1, DPM2 and DPM3. EMBO J. 19, Aebi, M., Helenius, A., Schenk, B., Barone, R., Fiumara, A., Berger, E.G., Hennet, T., Imbach, T., Stutz, A., Bjursell, C., et al. (1999). Carbohydrate-deficient glycoprotein syndromes become congenital disorders of glycosylation: An updated nomenclature for CDG. First International Workshop on CDGS. Glycoconj J. 16, Freeze, H.H. (2006). Genetic defects in the human glycome. Nat. Rev. Genet. 7, Jaeken, J., and Matthijs, G. (2007). Congenital Disorders of Glycosylation: A Rapidly Expanding Disease Family. Annu. Rev. Genomics Hum. Genet. 8, Thompson, J.D., Higgins, D.G., and Gibson, T.J. (1994). Clustal W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positionspecific gap penalties and weight matrix choice. Nucleic Acids Res. 22, The American Journal of Human Genetics 82, , March 2008
Diagnosis of CDG Enzyme Analysis and Other Investigations
 Diagnosis of CDG Enzyme Analysis and Other Investigations Biochemical Genetics Network Cambridge April 2005 Viki Worthington National Hospital for Neurology and Neurosurgery, London EUROGLYCANET European
Diagnosis of CDG Enzyme Analysis and Other Investigations Biochemical Genetics Network Cambridge April 2005 Viki Worthington National Hospital for Neurology and Neurosurgery, London EUROGLYCANET European
MUTATION UPDATE. Human Mutation
 MUTATION UPDATE Human Mutation OFFICIAL JOURNAL Congenital Disorders of Glycosylation: An Update on Defects Affecting the Biosynthesis of Dolichol-Linked Oligosaccharides www.hgvs.org Micha A. Haeuptle
MUTATION UPDATE Human Mutation OFFICIAL JOURNAL Congenital Disorders of Glycosylation: An Update on Defects Affecting the Biosynthesis of Dolichol-Linked Oligosaccharides www.hgvs.org Micha A. Haeuptle
Metadata of the chapter that will be visualized online
 Metadata of the chapter that will be visualized online Chapter Title The Alg1, Alg2, and Alg11 Mannosyltransferases of the Endoplasmic Reticulum Copyright Year 2014 Copyright Holder Springer Japan Corresponding
Metadata of the chapter that will be visualized online Chapter Title The Alg1, Alg2, and Alg11 Mannosyltransferases of the Endoplasmic Reticulum Copyright Year 2014 Copyright Holder Springer Japan Corresponding
ALG9 mannosyltransferase is involved in two different steps of lipid-linked oligosaccharide biosynthesis
 Glycobiology vol. 15 no. 11 pp. 1156 1163, 2005 doi:10.1093/glycob/cwj002 Advance Access publication on June 29, 2005 ALG9 mannosyltransferase is involved in two different steps of lipid-linked oligosaccharide
Glycobiology vol. 15 no. 11 pp. 1156 1163, 2005 doi:10.1093/glycob/cwj002 Advance Access publication on June 29, 2005 ALG9 mannosyltransferase is involved in two different steps of lipid-linked oligosaccharide
A Novel Carbohydrate-deficient Glycoprotein Syndrome Characterized by a Deficiency in Glucosylation of the Dolichol-linked Oligosaccharide
 A Novel Carbohydrate-deficient Glycoprotein Syndrome Characterized by a Deficiency in Glucosylation of the Dolichol-linked Oligosaccharide Patricie Burda,* Lubor Borsig, Joanneke de Rijk-van Andel, Ron
A Novel Carbohydrate-deficient Glycoprotein Syndrome Characterized by a Deficiency in Glucosylation of the Dolichol-linked Oligosaccharide Patricie Burda,* Lubor Borsig, Joanneke de Rijk-van Andel, Ron
Biosynthesis of N and O Glycans
 TechNote #TNGL101 Biosynthesis of N and O Glycans These suggestions and data are based on information we believe to be reliable. They are offered in good faith, but without guarantee, as conditions and
TechNote #TNGL101 Biosynthesis of N and O Glycans These suggestions and data are based on information we believe to be reliable. They are offered in good faith, but without guarantee, as conditions and
Failure to properly synthesize glycoproteins and other glycosylated molecules results in human disease
 Failure to properly synthesize glycoproteins and other glycosylated molecules results in human disease - muta6ons in genes encoding enzymes involved in glycoprotein biosynthesis: congenital disorders of
Failure to properly synthesize glycoproteins and other glycosylated molecules results in human disease - muta6ons in genes encoding enzymes involved in glycoprotein biosynthesis: congenital disorders of
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
A broad spectrum of clinical presentations in congenital disorders of glycosylation I: a series of 26 cases
 14 Pédiatrie, Hôpital des Enfants-Malades, Paris, France P de Lonlay J Laurent J Schmitz J M Saudubray Service de Biochimie, Hôpital Bichat, France N Seta S Barrot Pédiatrie, Marseille B Chabrol Génétique,
14 Pédiatrie, Hôpital des Enfants-Malades, Paris, France P de Lonlay J Laurent J Schmitz J M Saudubray Service de Biochimie, Hôpital Bichat, France N Seta S Barrot Pédiatrie, Marseille B Chabrol Génétique,
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
MPDU1 mutations underlie a novel human congenital disorder of glycosylation, designated type If
 MPDU1 mutations underlie a novel human congenital disorder of glycosylation, designated type If See related Commentary on pages 1579 1582. Barbara Schenk, 1 Timo Imbach, 2 Christian G. Frank, 1 Claudia
MPDU1 mutations underlie a novel human congenital disorder of glycosylation, designated type If See related Commentary on pages 1579 1582. Barbara Schenk, 1 Timo Imbach, 2 Christian G. Frank, 1 Claudia
Piirainen, Mari; de Ruijter, Jorg C.; Koskela, Essi V.; Frey, Alexander Glycoengineering of yeasts from the perspective of glycosylation efficiency
 Powered by TCPDF (www.tcpdf.org) This is an electronic reprint of the original article. This reprint may differ from the original in pagination and typographic detail. Piirainen, Mari; de Ruijter, Jorg
Powered by TCPDF (www.tcpdf.org) This is an electronic reprint of the original article. This reprint may differ from the original in pagination and typographic detail. Piirainen, Mari; de Ruijter, Jorg
N-Linked glycosylation
 4 N-Linked glycosylation N-linked glycosylation is a common class of glycosylation encountered in all eukaryotes as well as in Archaea and some bacteria. In eukaryotes, the assembly of N-glycans follows
4 N-Linked glycosylation N-linked glycosylation is a common class of glycosylation encountered in all eukaryotes as well as in Archaea and some bacteria. In eukaryotes, the assembly of N-glycans follows
More than 70% of the approved biopharmaceuticals are glycosylated
 A Combined System for Engineering Glycosylation Efficiency and Glycan Structure in Saccharomyces cerevisiae Farnoush Parsaie Nasab, a Markus Aebi, a Gesche Bernhard, b Alexander Daniel Frey a * Institute
A Combined System for Engineering Glycosylation Efficiency and Glycan Structure in Saccharomyces cerevisiae Farnoush Parsaie Nasab, a Markus Aebi, a Gesche Bernhard, b Alexander Daniel Frey a * Institute
7.06 Cell Biology EXAM #3 April 24, 2003
 7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004
 Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Synthesis and elongation of fatty acids
 Synthesis and elongation of fatty acids A molecular caliper mechanism for determining very long-chain fatty acid length Vladimir Denic and Jonathan S. Weissman (2007) Cell 130, 663-677 February 28, 2008
Synthesis and elongation of fatty acids A molecular caliper mechanism for determining very long-chain fatty acid length Vladimir Denic and Jonathan S. Weissman (2007) Cell 130, 663-677 February 28, 2008
2013 John Wiley & Sons, Inc. All rights reserved. PROTEIN SORTING. Lecture 10 BIOL 266/ Biology Department Concordia University. Dr. S.
 PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
PROTEIN SORTING Lecture 10 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University Introduction Membranes divide the cytoplasm of eukaryotic cells into distinct compartments. The endomembrane
TRANSPORT PROCESSES. 1b. moving proteins into membranes and organelles
 1b. moving proteins into membranes and organelles SLIDE 1 A typical mammalian cell contains up to 10,000 different kinds of proteins. The vast majority of these proteins are synthesized by cytosolic ribosomes,
1b. moving proteins into membranes and organelles SLIDE 1 A typical mammalian cell contains up to 10,000 different kinds of proteins. The vast majority of these proteins are synthesized by cytosolic ribosomes,
Glycoprotein Maturation and Quality Control in the Endoplasmic Reticulum Dr. Daniel Hebert
 Glycoprotein Maturation and Quality Control in the Endoplasmic Reticulum Department of Biochemistry and Molecular Biology University of Massachusetts, USA 1 Intracellular protein trafficking Plasma membrane
Glycoprotein Maturation and Quality Control in the Endoplasmic Reticulum Department of Biochemistry and Molecular Biology University of Massachusetts, USA 1 Intracellular protein trafficking Plasma membrane
Nucleotide-activated sugars
 2 Donor substrates Monosaccharides are chemically inert molecules, thus they need to be activated to high-energy donor substrates for glycosyltransferase reactions. Typically, phosphate is used as reactive
2 Donor substrates Monosaccharides are chemically inert molecules, thus they need to be activated to high-energy donor substrates for glycosyltransferase reactions. Typically, phosphate is used as reactive
The Ktr1p, Ktr3p, and Kre2p/Mnt1p Mannosyltransferases Participate in the Elaboration of Yeast O- and N-linked Carbohydrate Chains*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 272, No. 24, Issue of June 13, pp. 15527 15531, 1997 1997 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. The Ktr1p, Ktr3p,
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 272, No. 24, Issue of June 13, pp. 15527 15531, 1997 1997 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. The Ktr1p, Ktr3p,
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Stelter et al., http://www.jcb.org/cgi/content/full/jcb.201105042/dc1 S1 Figure S1. Dyn2 recruitment to edid-labeled FG domain Nups.
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Stelter et al., http://www.jcb.org/cgi/content/full/jcb.201105042/dc1 S1 Figure S1. Dyn2 recruitment to edid-labeled FG domain Nups.
Dolichol phosphate mannose synthase (DPM1) mutations define congenital disorder of glycosylation Ie (CDG-Ie)
 Dolichol phosphate mannose synthase (DPM1) mutations define congenital disorder of glycosylation Ie (CDG-Ie) Soohyun Kim, 1 Vibeke Westphal, 1 Geetha Srikrishna, 1 Darshini P. Mehta, 1 Sandra Peterson,
Dolichol phosphate mannose synthase (DPM1) mutations define congenital disorder of glycosylation Ie (CDG-Ie) Soohyun Kim, 1 Vibeke Westphal, 1 Geetha Srikrishna, 1 Darshini P. Mehta, 1 Sandra Peterson,
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Congenital disorders of glycosylation type Ia and Ib
 DOCTOR OF MEDICAL SCIENCE Congenital disorders of glycosylation type Ia and Ib Genetic, biochemical and clinical studies Susanne Kjærgaard This review has been accepted as a thesis together with eight
DOCTOR OF MEDICAL SCIENCE Congenital disorders of glycosylation type Ia and Ib Genetic, biochemical and clinical studies Susanne Kjærgaard This review has been accepted as a thesis together with eight
~Lentivirus production~
 ~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
PROTEIN TRAFFICKING. Dr. SARRAY Sameh, Ph.D
 PROTEIN TRAFFICKING Dr. SARRAY Sameh, Ph.D Overview Proteins are synthesized either on free ribosomes or on ribosomes bound to endoplasmic reticulum (RER). The synthesis of nuclear, mitochondrial and peroxisomal
PROTEIN TRAFFICKING Dr. SARRAY Sameh, Ph.D Overview Proteins are synthesized either on free ribosomes or on ribosomes bound to endoplasmic reticulum (RER). The synthesis of nuclear, mitochondrial and peroxisomal
What sort of Science is Glycoscience? (Introductory lecture)
 Glycosciences: Glycobiology & Glycochemistry e-learning course What sort of Science is Glycoscience? (Introductory lecture) Paula Videira Faculdade de Ciências Médicas Nova University, Lisbon Portugal
Glycosciences: Glycobiology & Glycochemistry e-learning course What sort of Science is Glycoscience? (Introductory lecture) Paula Videira Faculdade de Ciências Médicas Nova University, Lisbon Portugal
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
Heterodimeric Alg13/Alg14 UDP-GlcNAc transferase
 Heterodimeric Alg13/Alg14 UDP-GlcNAc transferase Introduction As one of the most frequent and common posttranslational modifications in eukaryotic cell, protein N-glycosylation begins with the multistep
Heterodimeric Alg13/Alg14 UDP-GlcNAc transferase Introduction As one of the most frequent and common posttranslational modifications in eukaryotic cell, protein N-glycosylation begins with the multistep
Hands-On Ten The BRCA1 Gene and Protein
 Hands-On Ten The BRCA1 Gene and Protein Objective: To review transcription, translation, reading frames, mutations, and reading files from GenBank, and to review some of the bioinformatics tools, such
Hands-On Ten The BRCA1 Gene and Protein Objective: To review transcription, translation, reading frames, mutations, and reading files from GenBank, and to review some of the bioinformatics tools, such
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine
 Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
Protein sorting (endoplasmic reticulum) Dr. Diala Abu-Hsasan School of Medicine dr.abuhassand@gmail.com An overview of cellular components Endoplasmic reticulum (ER) It is a network of membrane-enclosed
Glycans linked to lipids and lipid precursors
 Glycobiology BCMB 8130 Clinical Correlations! -sign up for 1st and 2nd choice (1/2)! *********************************! Glycosylation in Diabetes! O-Mannosylation in Human Pathophysiology! Glycoconjugate
Glycobiology BCMB 8130 Clinical Correlations! -sign up for 1st and 2nd choice (1/2)! *********************************! Glycosylation in Diabetes! O-Mannosylation in Human Pathophysiology! Glycoconjugate
Intracellular Compartments and Protein Sorting
 Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
Intracellular Compartments and Protein Sorting Intracellular Compartments A eukaryotic cell is elaborately subdivided into functionally distinct, membrane-enclosed compartments. Each compartment, or organelle,
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Significance and Functions of Carbohydrates. Bacterial Cell Walls
 Biochemistry 462a - Carbohydrate Function Reading - Chapter 9 Practice problems - Chapter 9: 2, 4a, 4b, 6, 9, 10, 13, 14, 15, 16a, 17; Carbohydrate extra problems Significance and Functions of Carbohydrates
Biochemistry 462a - Carbohydrate Function Reading - Chapter 9 Practice problems - Chapter 9: 2, 4a, 4b, 6, 9, 10, 13, 14, 15, 16a, 17; Carbohydrate extra problems Significance and Functions of Carbohydrates
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
SACCHAROMYCES CEREVISIAE GENOMIC LIBRARY SCREENING IN SEARCH FOR THE GENE RESPONSIBLE FOR INDUCTIVE ACTIVE GLYCEROL
 SACCHAROMYCES CEREVISIAE GENOMIC LIBRARY SCREENING IN SEARCH FOR THE GENE RESPONSIBLE FOR INDUCTIVE ACTIVE GLYCEROL UPTAKE R.P. Oliveira, and C. Lucas Departamento de Biologia da Universidade do Minho.
SACCHAROMYCES CEREVISIAE GENOMIC LIBRARY SCREENING IN SEARCH FOR THE GENE RESPONSIBLE FOR INDUCTIVE ACTIVE GLYCEROL UPTAKE R.P. Oliveira, and C. Lucas Departamento de Biologia da Universidade do Minho.
W I S S E N T E C H N I K L E I D E N S C H A F T MOL.911. Cell Engineering. u
 1 W I S S E N T E C H N I K L E I D E N S C H A F T MOL.911 Cell Engineering u www.tugraz.at MOL.911 Molecular Biotechnology I 2 Cell Engineering General strategies: Knock out of specific genes - Gene
1 W I S S E N T E C H N I K L E I D E N S C H A F T MOL.911 Cell Engineering u www.tugraz.at MOL.911 Molecular Biotechnology I 2 Cell Engineering General strategies: Knock out of specific genes - Gene
Supplementary Information
 Supplementary Information Archaeal Elp3 catalyzes trna wobble uridine modification at C5 via a radical mechanism Kiruthika Selvadurai, Pei Wang, Joseph Seimetz & Raven H Huang* Department of Biochemistry,
Supplementary Information Archaeal Elp3 catalyzes trna wobble uridine modification at C5 via a radical mechanism Kiruthika Selvadurai, Pei Wang, Joseph Seimetz & Raven H Huang* Department of Biochemistry,
Introduction CASE REPORT. Key Clinical Message
 CASE REPORT A novel homozygous mutation in the mannose phosphate isomerase gene causing congenital disorder of glycation and hyperinsulinemic hypoglycemia in an infant Asma Deeb & Abdulla Al Amoodi Paediatric
CASE REPORT A novel homozygous mutation in the mannose phosphate isomerase gene causing congenital disorder of glycation and hyperinsulinemic hypoglycemia in an infant Asma Deeb & Abdulla Al Amoodi Paediatric
Manja Henze, Dorothee Merker and Lothar Elling. 1. Characteristics of the Recombinant β-glycosidase from Pyrococcus
 S1 of S17 Supplementary Materials: Microwave-Assisted Synthesis of Glycoconjugates by Transgalactosylation with Recombinant Thermostable β-glycosidase from Pyrococcus Manja Henze, Dorothee Merker and Lothar
S1 of S17 Supplementary Materials: Microwave-Assisted Synthesis of Glycoconjugates by Transgalactosylation with Recombinant Thermostable β-glycosidase from Pyrococcus Manja Henze, Dorothee Merker and Lothar
TECHNICAL BULLETIN. R 2 GlcNAcβ1 4GlcNAcβ1 Asn
 GlycoProfile II Enzymatic In-Solution N-Deglycosylation Kit Product Code PP0201 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Glycosylation is one of the most common posttranslational
GlycoProfile II Enzymatic In-Solution N-Deglycosylation Kit Product Code PP0201 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Glycosylation is one of the most common posttranslational
In Saccharomyces cerevisiae, as in other eukaryotes, the
 Published Online: 7 September, 1998 Supp Info: http://doi.org/10.1083/jcb.142.5.1223 Downloaded from jcb.rupress.org on December 11, 2018 Degradation of Misfolded Endoplasmic Reticulum Glycoproteins in
Published Online: 7 September, 1998 Supp Info: http://doi.org/10.1083/jcb.142.5.1223 Downloaded from jcb.rupress.org on December 11, 2018 Degradation of Misfolded Endoplasmic Reticulum Glycoproteins in
MCB140: Second Midterm Spring 2010
 MCB140: Second Midterm Spring 2010 Before you start, print your name and student identification number (S.I.D) at the top of each page. There are 11 pages including this page. You will have 150 minutes
MCB140: Second Midterm Spring 2010 Before you start, print your name and student identification number (S.I.D) at the top of each page. There are 11 pages including this page. You will have 150 minutes
ALG6-CDG in South Africa: Genotype-Phenotype Description of Five Novel Patients
 JIMD Reports DOI 10.1007/8904_2012_150 CASE REPORT ALG6-CDG in South Africa: Genotype-Phenotype Description of Five Novel Patients M. Dercksen A. C. Crutchley E. M. Honey M. M. Lippert G. Matthijs L. J.
JIMD Reports DOI 10.1007/8904_2012_150 CASE REPORT ALG6-CDG in South Africa: Genotype-Phenotype Description of Five Novel Patients M. Dercksen A. C. Crutchley E. M. Honey M. M. Lippert G. Matthijs L. J.
Metadata of the chapter that will be visualized online
 Metadata of the chapter that will be visualized online Chapter Title Dolichyl-Phosphate (UDP-N-Acetylglucosamine) N- Acetylglucosaminephospho transferase 1 (GlcNAc-1-P Transferase) (DPAGT1) Copyright Year
Metadata of the chapter that will be visualized online Chapter Title Dolichyl-Phosphate (UDP-N-Acetylglucosamine) N- Acetylglucosaminephospho transferase 1 (GlcNAc-1-P Transferase) (DPAGT1) Copyright Year
The yeast ALG11 gene specifies addition of the terminal 1,2-Man to the. cytosolic side of the endoplasmic reticulum.*
 JBC Papers in Press. Published on February 15, 2001 as Manuscript M010896200 MO: 10896 Print Version The yeast ALG11 gene specifies addition of the terminal 1,2-Man to the Man 5 GlcNAc 2 -PP-dolichol N-glycosylation
JBC Papers in Press. Published on February 15, 2001 as Manuscript M010896200 MO: 10896 Print Version The yeast ALG11 gene specifies addition of the terminal 1,2-Man to the Man 5 GlcNAc 2 -PP-dolichol N-glycosylation
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
The Essential Smp3 Protein Is Required for Addition of the Sidebranching Fourth Mannose during Assembly of Yeast Glycosylphosphatidylinositols*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 276, No. 29, Issue of July 20, pp. 27731 27739, 2001 2001 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. The Essential Smp3
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 276, No. 29, Issue of July 20, pp. 27731 27739, 2001 2001 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. The Essential Smp3
Very-Long Chain Fatty Acid Biosynthesis
 Very-Long Chain Fatty Acid Biosynthesis Objectives: 1. Review information on the isolation of mutants deficient in VLCFA biosynthesis 2. Generate hypotheses to explain the absence of mutants with lesions
Very-Long Chain Fatty Acid Biosynthesis Objectives: 1. Review information on the isolation of mutants deficient in VLCFA biosynthesis 2. Generate hypotheses to explain the absence of mutants with lesions
Posttranslational Modification and Targeting of Proteins
 Posttranslational Modification and Targeting of Proteins Graduate Biochemistry Term 2/2016 Assist. Prof. Dr. Panida Khunkaewla School of Chemistry, Institute of Science Suranaree University of Technology
Posttranslational Modification and Targeting of Proteins Graduate Biochemistry Term 2/2016 Assist. Prof. Dr. Panida Khunkaewla School of Chemistry, Institute of Science Suranaree University of Technology
October 26, Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
DNA context and promoter activity affect gene expression in lentiviral vectors
 ACTA BIOMED 2008; 79: 192-196 Mattioli 1885 O R I G I N A L A R T I C L E DNA context and promoter activity affect gene expression in lentiviral vectors Gensheng Mao 1, Francesco Marotta 2, Jia Yu 3, Liang
ACTA BIOMED 2008; 79: 192-196 Mattioli 1885 O R I G I N A L A R T I C L E DNA context and promoter activity affect gene expression in lentiviral vectors Gensheng Mao 1, Francesco Marotta 2, Jia Yu 3, Liang
Structural Variation and Medical Genomics
 Structural Variation and Medical Genomics Andrew King Department of Biomedical Informatics July 8, 2014 You already know about small scale genetic mutations Single nucleotide polymorphism (SNPs) Deletions,
Structural Variation and Medical Genomics Andrew King Department of Biomedical Informatics July 8, 2014 You already know about small scale genetic mutations Single nucleotide polymorphism (SNPs) Deletions,
Alteration of mannose transport in broblasts from type I carbohydrate de cient glycoprotein syndrome patients
 Biochimica et Biophysica Acta 1453 (1999) 369^377 Alteration of mannose transport in broblasts from type I carbohydrate de cient glycoprotein syndrome patients T. Dupre a; *, E. Ogier-Denis b, S.E.H. Moore
Biochimica et Biophysica Acta 1453 (1999) 369^377 Alteration of mannose transport in broblasts from type I carbohydrate de cient glycoprotein syndrome patients T. Dupre a; *, E. Ogier-Denis b, S.E.H. Moore
Chapter 3. Expression of α5-megfp in Mouse Cortical Neurons. on the β subunit. Signal sequences in the M3-M4 loop of β nachrs bind protein factors to
 22 Chapter 3 Expression of α5-megfp in Mouse Cortical Neurons Subcellular localization of the neuronal nachr subtypes α4β2 and α4β4 depends on the β subunit. Signal sequences in the M3-M4 loop of β nachrs
22 Chapter 3 Expression of α5-megfp in Mouse Cortical Neurons Subcellular localization of the neuronal nachr subtypes α4β2 and α4β4 depends on the β subunit. Signal sequences in the M3-M4 loop of β nachrs
Envelope glycans of immunodeficiency virions are almost entirely oligomannose antigens
 Supporting Information for: Envelope glycans of immunodeficiency virions are almost entirely oligomannose antigens Katie J. Doores *1,2, Camille Bonomelli *3, David J. Harvey 3, Snezana Vasiljevic 3, Raymond
Supporting Information for: Envelope glycans of immunodeficiency virions are almost entirely oligomannose antigens Katie J. Doores *1,2, Camille Bonomelli *3, David J. Harvey 3, Snezana Vasiljevic 3, Raymond
joint corresponding authors: and
 A Zebrafish Model Of PMM2-CDG Reveals Altered Neurogenesis And A Substrate-Accumulation Mechanism For N-Linked Glycosylation Deficiency Revised Abigail Cline *, Ningguo Gao, Heather Flanagan-Steet*, Vandana
A Zebrafish Model Of PMM2-CDG Reveals Altered Neurogenesis And A Substrate-Accumulation Mechanism For N-Linked Glycosylation Deficiency Revised Abigail Cline *, Ningguo Gao, Heather Flanagan-Steet*, Vandana
Supplementary Figure 1
 Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
The dolichol-linked oligosaccharide Glc3Man9Glc-
 Published Online: 2 April, 2007 Supp Info: http://doi.org/10.1083/jcb.200611079 Downloaded from jcb.rupress.org on August 24, 2018 JCB: ARTICLE Dolichol-linked oligosaccharide selection by the oligosaccharyltransferase
Published Online: 2 April, 2007 Supp Info: http://doi.org/10.1083/jcb.200611079 Downloaded from jcb.rupress.org on August 24, 2018 JCB: ARTICLE Dolichol-linked oligosaccharide selection by the oligosaccharyltransferase
Nucleic acids. Nucleic acids are information-rich polymers of nucleotides
 Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
APPENDIX -1 LIST OF TABLES. 01 The blood group systems Blood group collections
 APPENDIX -1 LIST OF TABLES Table 01 The blood group systems. 014 005 02 Blood group collections. 016 008 03 Low frequency antigens: the 700 series. 017 009 04 Frequencies of low frequency antigens. 017
APPENDIX -1 LIST OF TABLES Table 01 The blood group systems. 014 005 02 Blood group collections. 016 008 03 Low frequency antigens: the 700 series. 017 009 04 Frequencies of low frequency antigens. 017
Very-Long Chain Fatty Acid Biosynthesis
 Very-Long Chain Fatty Acid Biosynthesis Objectives: 1. Review information on the isolation of mutants deficient in VLCFA biosynthesis 2. Generate hypotheses to explain the absence of mutants with lesions
Very-Long Chain Fatty Acid Biosynthesis Objectives: 1. Review information on the isolation of mutants deficient in VLCFA biosynthesis 2. Generate hypotheses to explain the absence of mutants with lesions
Designing Tools for Studying the Dynamic Glycome
 Marshall University Marshall Digital Scholar Chemistry Faculty Research Chemistry Winter 12-2012 Designing Tools for Studying the Dynamic Glycome John F. Rakus Marshall University, rakus@marshall.edu Follow
Marshall University Marshall Digital Scholar Chemistry Faculty Research Chemistry Winter 12-2012 Designing Tools for Studying the Dynamic Glycome John F. Rakus Marshall University, rakus@marshall.edu Follow
Computational Systems Biology: Biology X
 Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#4:(October-0-4-2010) Cancer and Signals 1 2 1 2 Evidence in Favor Somatic mutations, Aneuploidy, Copy-number changes and LOH
Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#4:(October-0-4-2010) Cancer and Signals 1 2 1 2 Evidence in Favor Somatic mutations, Aneuploidy, Copy-number changes and LOH
A misfolded protein conformation is not a sufficient condition for in vivo glucosylation by the UDP-Glc:glycoprotein glucosyltransferase
 The EMBO Journal Vol.17 No.20 pp.5877 5886, 1998 A misfolded protein conformation is not a sufficient condition for in vivo glucosylation by the UDP-Glc:glycoprotein glucosyltransferase Fabiana Fernández,
The EMBO Journal Vol.17 No.20 pp.5877 5886, 1998 A misfolded protein conformation is not a sufficient condition for in vivo glucosylation by the UDP-Glc:glycoprotein glucosyltransferase Fabiana Fernández,
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
An evolving view of the eukaryotic oligosaccharyltransferase
 Glycobiology Advance Access published November 29, 2005 An evolving view of the eukaryotic oligosaccharyltransferase Daniel J. Kelleher and Reid Gilmore*Department of Biochemistry and Molecular PharmacologyUniversity
Glycobiology Advance Access published November 29, 2005 An evolving view of the eukaryotic oligosaccharyltransferase Daniel J. Kelleher and Reid Gilmore*Department of Biochemistry and Molecular PharmacologyUniversity
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
7.012 Problem Set 6 Solutions
 Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent Protein.
 prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
John F. Cipollo 1,3 and Robert B. Trimble 2,3,4
 Glycobiology vol. 12 no. 11 pp. 749 762, 2002 The Saccharomyces cerevisiae alg12 mutant reveals a role for the middle-arm α1,2manand upper-arm α1,2manα1,6man- residues of Glc 3 Man 9 GlcNAc 2 -PP-Dol in
Glycobiology vol. 12 no. 11 pp. 749 762, 2002 The Saccharomyces cerevisiae alg12 mutant reveals a role for the middle-arm α1,2manand upper-arm α1,2manα1,6man- residues of Glc 3 Man 9 GlcNAc 2 -PP-Dol in
Practice Problems 8. a) What do we define as a beneficial or advantageous mutation to the virus? Why?
 Life Sciences 1a Practice Problems 8 1. You have two strains of HIV one is a wild type strain of HIV and the second has acquired a mutation in the gene encoding the protease. This mutation has a dual effect
Life Sciences 1a Practice Problems 8 1. You have two strains of HIV one is a wild type strain of HIV and the second has acquired a mutation in the gene encoding the protease. This mutation has a dual effect
Physical interactions between the Alg1, Alg2, and Alg11 mannosyltransferases of the endoplasmic reticulum
 Glycobiology vol. 14 no. 6 pp. 559±570, 2004 DOI: 10.1093/glycob/cwh072 Advance Access publication on March 24, 2004 Physical interactions between the Alg1, Alg2, and Alg11 mannosyltransferases of the
Glycobiology vol. 14 no. 6 pp. 559±570, 2004 DOI: 10.1093/glycob/cwh072 Advance Access publication on March 24, 2004 Physical interactions between the Alg1, Alg2, and Alg11 mannosyltransferases of the
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
8 Suppression Analysis
 Genetic Techniques for Biological Research Corinne A. Michels Copyright q 2002 John Wiley & Sons, Ltd ISBNs: 0-471-89921-6 (Hardback); 0-470-84662-3 (Electronic) 8 Suppression Analysis OVERVIEW Suppression
Genetic Techniques for Biological Research Corinne A. Michels Copyright q 2002 John Wiley & Sons, Ltd ISBNs: 0-471-89921-6 (Hardback); 0-470-84662-3 (Electronic) 8 Suppression Analysis OVERVIEW Suppression
Supplemental Materials and Methods Plasmids and viruses Quantitative Reverse Transcription PCR Generation of molecular standard for quantitative PCR
 Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
(Stratagene, La Jolla, CA) (Supplemental Fig. 1A). A 5.4-kb EcoRI fragment
 SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
Insulin Resistance. Biol 405 Molecular Medicine
 Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
4.32 Glycoengineering: Recombinant Glycoproteins
 4.32 Glycoengineering: Recombinant Glycoproteins M. J. Betenbaugh, N. Tomiya, and S. Narang, Johns Hopkins University, Baltimore, MD, USA ß 2007 Elsevier Ltd. All rights reserved. 4.32.1 Introduction 607
4.32 Glycoengineering: Recombinant Glycoproteins M. J. Betenbaugh, N. Tomiya, and S. Narang, Johns Hopkins University, Baltimore, MD, USA ß 2007 Elsevier Ltd. All rights reserved. 4.32.1 Introduction 607
Communication. Identification of Methionine N -Acetyltransferase from Saccharomyces cerevisiae
 Communication THE JOURNAL OP BIOLOGICAL CHEMISTRY Vol. 265, No. 7, Issue of March 5, pp. 3603-3606,lSSO 0 1990 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U. S. A. Identification
Communication THE JOURNAL OP BIOLOGICAL CHEMISTRY Vol. 265, No. 7, Issue of March 5, pp. 3603-3606,lSSO 0 1990 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U. S. A. Identification
Problem Set #5 4/3/ Spring 02
 Question 1 Chloroplasts contain six compartments outer membrane, intermembrane space, inner membrane, stroma, thylakoid membrane, and thylakoid lumen each of which is populated by specific sets of proteins.
Question 1 Chloroplasts contain six compartments outer membrane, intermembrane space, inner membrane, stroma, thylakoid membrane, and thylakoid lumen each of which is populated by specific sets of proteins.
Eukaryotic transcription (III)
 Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
Cell wall components:
 Main differences between plant and animal cells: Plant cells have: cell walls, a large central vacuole, plastids and turgor pressure. The Cell Wall The primary cell wall is capable of rapid expansion during
Main differences between plant and animal cells: Plant cells have: cell walls, a large central vacuole, plastids and turgor pressure. The Cell Wall The primary cell wall is capable of rapid expansion during
Main differences between plant and animal cells: Plant cells have: cell walls, a large central vacuole, plastids and turgor pressure.
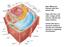 Main differences between plant and animal cells: Plant cells have: cell walls, a large central vacuole, plastids and turgor pressure. Animal cells have a lysosome (related to vacuole) and centrioles (function
Main differences between plant and animal cells: Plant cells have: cell walls, a large central vacuole, plastids and turgor pressure. Animal cells have a lysosome (related to vacuole) and centrioles (function
1. This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Chapter 11. Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins.
 Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
The Biology and Genetics of Cells and Organisms The Biology of Cancer
 The Biology and Genetics of Cells and Organisms The Biology of Cancer Mendel and Genetics How many distinct genes are present in the genomes of mammals? - 21,000 for human. - Genetic information is carried
The Biology and Genetics of Cells and Organisms The Biology of Cancer Mendel and Genetics How many distinct genes are present in the genomes of mammals? - 21,000 for human. - Genetic information is carried
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
The growth analysis of ylsnf1 gene deletion mutant of Yarrowia lipolytica on hydrophobic substrate media
 The growth analysis of ylsnf1 gene deletion mutant of Yarrowia lipolytica on hydrophobic substrate media Napapol Poopanitpan 1,*, Threesara Lertvatasilp 1, Narisa Poonsub 1, Prapasiri Jantanee 1, Sasimanas
The growth analysis of ylsnf1 gene deletion mutant of Yarrowia lipolytica on hydrophobic substrate media Napapol Poopanitpan 1,*, Threesara Lertvatasilp 1, Narisa Poonsub 1, Prapasiri Jantanee 1, Sasimanas
The addition of sugar moiety determines the blood group
 The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
Phosphate buffered saline (PBS) for washing the cells TE buffer (nuclease-free) ph 7.5 for use with the PrimePCR Reverse Transcription Control Assay
 Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Genetic defects in dolichol metabolism
 J Inherit Metab Dis (2015) 38:157 169 DOI 10.1007/s10545-014-9760-1 COMPLEX LIPIDS Genetic defects in dolichol metabolism Anna Buczkowska & Ewa Swiezewska & Dirk J. Lefeber Received: 21 May 2014 /Revised:
J Inherit Metab Dis (2015) 38:157 169 DOI 10.1007/s10545-014-9760-1 COMPLEX LIPIDS Genetic defects in dolichol metabolism Anna Buczkowska & Ewa Swiezewska & Dirk J. Lefeber Received: 21 May 2014 /Revised:
Golgi nucleotide sugar transport and leukocyte adhesion deficiency II
 Golgi nucleotide sugar transport and leukocyte adhesion deficiency II Carlos B. Hirschberg J Clin Invest. 2001;108(1):3-6. https://doi.org/10.1172/jci13480. Spotlight In 1992 a new adhesion deficiency
Golgi nucleotide sugar transport and leukocyte adhesion deficiency II Carlos B. Hirschberg J Clin Invest. 2001;108(1):3-6. https://doi.org/10.1172/jci13480. Spotlight In 1992 a new adhesion deficiency
Luminescent platforms for monitoring changes in the solubility of amylin and huntingtin in living cells
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
This PDF file includes: Supplementary Figures 1 to 6 Supplementary Tables 1 to 2 Supplementary Methods Supplementary References
 Structure of the catalytic core of p300 and implications for chromatin targeting and HAT regulation Manuela Delvecchio, Jonathan Gaucher, Carmen Aguilar-Gurrieri, Esther Ortega, Daniel Panne This PDF file
Structure of the catalytic core of p300 and implications for chromatin targeting and HAT regulation Manuela Delvecchio, Jonathan Gaucher, Carmen Aguilar-Gurrieri, Esther Ortega, Daniel Panne This PDF file
