Nucleotide-activated sugars
|
|
|
- Katrina Lindsey
- 5 years ago
- Views:
Transcription
1 2 Donor substrates Monosaccharides are chemically inert molecules, thus they need to be activated to high-energy donor substrates for glycosyltransferase reactions. Typically, phosphate is used as reactive group, either alone or in the context of a nucleotide. Phosphate (P) acts as a versatile leaving group in the nucleophilic attacks involved in glycosylation reactions. At the endoplasmic reticulum (ER) membrane of eukaryotic cells, Man and Glc are also linked to the polyprenol dolichol-p (Dol-P). These Dol-P linked sugars are used as donor substrates for hydrophobic glycosyltransferases that are embedded in the ER membrane. The biosynthesis of donor substrates usually begins by the phosphorylation of hexoses at C1 and C6 mediated by hexokinase and phosphomutase enzymes. Hexokinase is an ubiquitous activity found from bacteria to mammals. By phosphorylating Glc at C6, hexokinase prevents the diffusion of Glc out the cytosol, where it is further processed. Hexokinase is not specific for Glc, since Man is also phosphorylated at C6 by the enzyme. The liver has an additional enzyme, glucokinase, which is specific for Glc and is important for glycogen formation. The sugar Gal is directly phosphorylated at C1 by the galactokinase enzyme. Deficiency of galactokinase leads to a form of galactosemia, which is characterized by the development of cataract due to the accumulation of galactitol in the lens. Phosphoglucomutase catalyzes the transition of phosphate in both C1 to C6 and C6 to C1 ways. The enzyme is important for the breakdown of glycogen where Glc-1-P is converted to Glc-6-P. In the liver, a Glc-6-phosphatase leads to the release of Glc, which drives the equilibrium of the phosphoglucomutase reaction towards Glc-6-P. This phosphatase is only found in the liver, which is the main organ regulating the levels of blood sugar. The deficiency of the main phosphoglucomutase isoform (PGM1) leads to a rare form of glycogen storage disease (GSD type XIV). Defects of glycogen breakdown mainly lead to hypoglycemia and muscle weakness, whereas some forms are severe and lead to death in infancy. The phosphoglucomutase isoform PGM3 also acts on GlcNAc-6-P and a dedicated phosphomannomutase (PMM2) catalyzes the formation of Man-1-P from Man-6-P. Mutations in the PMM2 gene are the main cause of congenital disorders of glycosylation (CDG).
2 Figure 1 Phosphorylation of Glc through hexokinase and phosphoglucomutase activity in the cytoplasm. Nucleotide-activated sugars The first nucleotide-activated sugar was identified by Leloir, who established UDP-Glc as substrate for the polymerization of Glc to glycogen. UDP is the most common nucleotide used to activate sugars in animals, as it is found linked to Glc, Gal, GlcNAc, GalNAc, GlcA and Xyl. Only two additional nucleotides are used in animals, GDP that is linked to Man and Fuc, and CMP that is linked to Sia. Whereas animal cells assemble their glycans from nine nucleotide-activated sugars, plants and in bacteria use many more substrates. More than 70 different nucleotide-activated sugars have been identified in bacteria to date. The profusion of nucleotide-activated sugars in plants and bacteria is puzzling. Why is a single form of activated Glc not sufficient in plants, which use UDP-Glc as substrate for cellulose biosynthesis and ADP-Glc as substrate for starch formation? Similar apparent redundancies are observed in bacteria. Could such a fragmentation of nucleotide-activated monosaccharides be related to a defense strategy against parasites, viruses and bacteriophages? Such pathogens divert the host metabolism for their need. An increased complexity in metabolic pathways may enable host cells to cope with such pathogen-driven diversions. Accordingly, the proliferation of nucleotide-activated sugars may represent a form of metabolic immunity (see glycans and evolution). Nucleotide-activated sugars are involved in the biosynthesis of large molecules like glycans, oligosaccharides and polysaccharides. Furthermore, the nucleotide-activated sugar UDP-GlcA is also widely used for the detoxification of xenobiotics by UDP-glucuronosyltransferases (UGT), which are mainly expressed in the liver in humans. The addition of GlcA to apolar molecules increases their solubility and thereby their elimination from the body. 2
3 Nucleotide-activated sugars are also important for the replication of some viruses. For example, the bacteriophages T2, T4 and T6 express glucosyltransferases that add Glc from UDP-Glc to hydroxymethylcytosine, thereby protecting the phage DNA from bacterial restriction enzymes. Similarly, baculoviruses express glucosyltransferases that transfer Glc from UDP-Glc to the molting hormone ecdysone, which inactivates the hormone and maintains the infected insect at a prepupal stage. Also, small molecules like the antibiotics aminoglycosides, e.g. neomycin, kanamycin and streptomycin, are synthesized from nucleotideactivated sugars. Figure 2 Structure of UDP- -D-Gal and list of nucleotide-activated sugars found in animal cells. In eukaryotes, nucleotide-activated sugars are synthesized in the cytosol with the exception of CMP-Sia, which is synthesized in the nucleus. The NDPpyrophosphorylase reaction is readily reversible. However, pyrophosphate is rapidly hydrolyzed in vivo to orthophosphate by an inorganic pyrophosphatase. Therefore, the nearly irreversible hydrolysis of pyrophosphate drives the reaction towards the synthesis of the nucleotide-activated sugar product. The substrates for the synthesis of nucleotide-activated sugars are sugar-1-p and NDP. Here again, Sia does not follow the rule, since Sia itself and CMP are the substrates for the formation of CMP-Sia by the N-acylneuraminate cytidylyltransferase enzyme. Not all nucleotide-activated sugars are built from monosaccharides units. For example, UDP-GlcA and UDP-Xyl are not synthesized from GlcA and Xyl, respectively, but from UDP-Glc, which is first oxidized to UDP-GlcA and then decarboxylated to UDP-Xyl. The so-called interconversion pathway plays a major role in maintaining stable intracellular levels of nucleotide-activated sugars under conditions where the supply of specific monosaccharides may be limited. In addition to the interconversion pathway, nucleotide-activated sugars can be 3
4 synthesized through the salvage pathway, i.e. through the breakdown of oligosaccharides in lysosomes and subsequent transfer of monosaccharides to the cytosol. Figure 3 Biosynthesis of nucleotide-activated sugars through the interconversion and salvage pathways. The interconversion of nucleotide-activated sugars is achieved by multiple enzymatic activities, such as 4,6-dehydratase and 4-epimerase/reductase (involved in conversion of GDP-Man to GDP-Fuc) or 2-epimerase (involved in conversion of UDP-GlcNAc to UDP-ManNAc). Bacteria and plants express a large amount of interconverting enzymes in contrast to animals, which have fewer nucleotide-activated sugars. Despite the limited set of interconverting activities in animals, these reactions are essential to the biosynthesis of nucleotide-activated sugars and thereby to the formation of glycoconjugates. For example, UDP-Glc can be formed from Glc-1-P and from the interconversion of UDP-Gal (through 4-epimerisation). In fact, the interconversion from UDP-Gal represents a major source of UDP-Glc in animal cells, as shown by the incorporation of [ 3 H]Glc in glycans after metabolic labeling of cells with [ 3 H]Gal. The interconversion of nucleotideactivated sugars in animal cells is controlled by rate-limiting enzymes, which are inhibited by the end-products of their respective pathways. Along this line, UDP- Glc dehydrogenase (UGD) is inhibited by UDP-Xyl, glutamine:fructose-6-p acetyltransferase (GFAT) by UDP-GlcNAc, UDP-GlcNAc 2-epimerase/kinase (GNE) by Sia and GDP-Man 4,6-dehydratase (GMDS) by GDP-Fuc. Deficiency of interconverting enzymes often leads to metabolic disorders characterized by the accumulation of toxic intermediates and by defects of glycosylation. For example, mutations in the UDP-Gal 4-epimerase causes a mild form of galactosemia, a disease where patients have to lower their dietary galactose intake to reduce the accumulation of toxic intermediates such as galactitol. Another defect of nucleotide-activated sugar interconversion is given by the deficiency of the rate-limiting enzyme UDP-GlcNAc 2-epimerase (GNE). The GNE protein includes two catalytic domains, the first one with the 2-epimerase 4
5 activity and the second one with a ManNAc kinase activity. Mutations in both domains lead to a reduced activity and thus to low biosynthesis of Sia. Surprisingly, the resulting disease, called Hereditary Inclusion Body Myopathy or Nonaka Myopathy, is rather mild considering the broad usage of Sia in glycoconjugates. The reason of this unexpected mild phenotype is probably related to the important contribution of the salvage pathway to generate enough CMP-Sia for incorporation into glycans. However, it is worth noting that the inactivation of the GNE gene in mice leads to embryonic lethality, which demonstrates that the importance of Sia biosynthesis for early embryonic development. Figure 4 NDP-sugar biosynthesis pathways. All reactions take place in the cytoplasma with the exception of the last step of CMP-Sia biosynthesis occuring in the nucleus. Rate-limiting enzymes are marked in red boxes. UGD: UDP-Glc dehydrogenase, GFAT: glutamine:fructose-6-p acetyltransferase, GNE: UDP-GlcNAc 2-epimerase/kinase, GMDS: GDP-Man 4,6-dehydratase. As mentioned previously, GNE is a rate-limiting activity in the synthesis of Sia since the enzyme is inhibited by CMP-Sia. Some mutations in the GNE gene in fact do not inactivate the enzymatic activity itself but abolish the allosteric effect of CMP-Sia, thereby opening the gate to a production of Sia ad libitum. Needless to say that such an excess production of Sia is not healthy and leads to the disease sialuria, which is characterized by increased presence of Sia in urine (hence the name sial-uria), hepatosplenomegaly and airway infections. Plants and bacteria produce a multitude of nucleotide-activated sugars and thereby rely extensively on interconverting enzymes to synthesize these donor substrates. These nucleotide-activated sugars are mainly involved in the biosynthesis of complex cell wall polysaccharides. Note that in plants and bacteria, a specific carbohydrate can be linked to different nucleotide. For example, in bacteria Glc is linked to UDP, GDP and TDP. In animals, carbohydrates are only found linked to a single type of nucleotides. 5
6 Figure 5 GNE mutations are associated with two diseases characterized by different symptoms. Common mutations detected in hereditary inclusion body myopathy are marked with black circle on the schematic representation of the GNE protein. Mutations detected in sialuria are marked with red triangles. A significant portion of carbohydrates are not synthesized de novo but recycled through salvage pathways. Monosaccharides like Glc and Gal are taken up in the gastrointestinal tract by dedicated transporters. By contrast, complex oligosaccharides, such as those found in maternal milk, are not digested and absorbed in the intestine. Within cells, glycoproteins are glycolipids are continuously broken down in lysosomes, thereby generating new building blocks for the synthesis of new molecules. The biological importance of glycan recycling in lysosomes is illustrated by lysosomal storage disorders such as Fabry s and Gaucher s diseases, in which deficiency of galactosidase and glucosidase enzymes lead to the toxic accumulation of glycolipids in lysosomes. As mentioned previously in the context of Hereditary Inclusion Body Myopathy, Sia is also extensively recycled in humans through the salvage pathway. Furthermore, Sia appears to be taken up from food products in the intestine and incorporated in glycans. It has been shown that NeuGc, which cannot be synthesized in humans, is indeed incorporated in the glycans of multiple human cell types after ingestion of dietary products rich in NeuGc, such as red meat. The incorporation of NeuGc in human glycans modifies the susceptibility to bacterial toxins, such as the subtilase cytotoxin SubAB produced by Shiga toxigenic Escherichia coli, which rely on this type of Sia to target host cells. Polyprenol-P-activated sugars Not all donor substrates of glycosyltransferase reactions are nucleotide-activated sugars. Polyprenol-based lipids are commonly used in bacteria, archaea and eukaryotes to build donor substrates. The glycosyltransferases using polyprenolbased substrates are mainly hydrophobic proteins with multiple trans-membrane 6
7 domains. The number of isoprene repeats varies according to the organisms, prokaryotes having isoprene units, yeasts units, animals units and plants units. In Dol, the last isoprene unit is reduced. This reduction is required for the subsequent phosphorylation step. All polyprenol-based substrates are phosphorylated with the exception of the undecaprenol substrates involved in the elongation of the LPS outer core, which carry a pyrophosphate group. Figure 6 Polyprenol-linked substrates in all domains of life. Dolichol has a saturated -isoprene unit whereas prokaryotic polyprenol have an unsaturated bond. Polyprenol and Dol share a common biosynthesis pathway with cholesterol up to the formation of farnesyl-pp. The basic isoprene unit is synthesized from acetyl-coa, which is condensed to 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) and further reduced to mevalonate. The HMG-CoA reductase activity is rate limiting in the pathway, as it is regulated at the transcription and protein levels by sterol concentrations. The HMG-CoA reductase enzyme is also the target of inhibitors called statins, which are commonly prescribed as cholesterol-lowering drugs. After reaching farnesyl-pp, the growing polyprenols are anchored in membranes and elongated by cis-prenyltransferase enzymes, also called dehydrodolichyl diphosphate synthase (DHDDS). The DHDDS enzymes are responsible for the variable chain length of polyprenols observed between prokaryotes and eukaryotes. Before receiving a monosaccharide unit, Dol undergo three modifications. First, the pyrophosphate group is removed by phosphatases, then the last isoprene unit is reduced and finally a phosphate group is added again by a dedicated kinase. The monosaccharides Man and Glc are attached to Dol-P in eukaryotes and various exotic sugars like GalA and Ara4N to polyprenols in bacteria. Dol-P-Man and Glc are substrates in the final assembly steps of lipid-linked 7
8 oligosaccharides in the N-linked glycosylation pathway, in the biosynthesis of the glycosylphosphatidylinositol (GPI) anchor, in O-mannosylation and in C- mannosylation. In bacteria, polyprenol-p linked monosaccharides are substrates for the modification of the lipid A core and for the synthesis of arabinogalactan and lipoarabinomannan in mycobacteria. Deficiencies in cis-prenyltransferase, Dol reductase, Dol kinase and Dol-P-Man synthase lead to a shortage of Dol-P linked substrates and thus to disorders of glycosylation. Figure 7 Mevalonate and dolichol biosynthesis pathway. Transporters The transporters for nucleotide-activated sugars are actually antiporters, which import nucleotide-activated sugars and export the corresponding nucleotide-monophosphates into the cytosol. Most glycosyltransferase reactions yield a nucleotide-diphosphate, meaning that a phosphatase reaction is required to generate nucleotide-monophosphates. Most transporters are specific for a given nucleotide-activated sugar but some transporters enable the import of multiple substrates such as UDP-Gal, UDP-GalNAc and UDP-GlcA. The majority of transporters are embedded in the Golgi membrane but can be localized at the ER membrane in some cases. Data from the last three years have shown that some nucleotide-activated sugars, e.g. GDP-Fuc, are imported in the ER and in the Golgi through different transporters. For example, the inactivation of the Golgi GDP-Fuc transporter leads to a defect of terminal fucosylation in N- and O-glycans, whereas core O-fucosylation, which takes place in the ER, is not affected. The substrate for glycan sulfation, 3'-phosphoadenosine 5'-phosphosulfate (PAPS), is also imported into the Golgi apparatus through an antiporter, which exports back AMP into the cytosol. A phosphate transporter ensuring the export of the phosphate anion to the cytosol is likely to exist, although such a transporter has not been identified yet. 8
9 Figure 8 Cellular distribution of nucleotide-activated sugar transporters. Distinct transporters can mediate the import of a nucleotide-activated sugar in the ER and Golgi apparatus (e.g. GDP-Fuc). 9
What sort of Science is Glycoscience? (Introductory lecture)
 Glycosciences: Glycobiology & Glycochemistry e-learning course What sort of Science is Glycoscience? (Introductory lecture) Paula Videira Faculdade de Ciências Médicas Nova University, Lisbon Portugal
Glycosciences: Glycobiology & Glycochemistry e-learning course What sort of Science is Glycoscience? (Introductory lecture) Paula Videira Faculdade de Ciências Médicas Nova University, Lisbon Portugal
Glycobiology 8130, 18/1/2005 Spring 2005
 Glycobiology 8130, 18/1/2005 Spring 2005 Nucleotide-sugar donors. Maor Bar-Peled, CCRC; Plant Biology Peled@ccrc.uga.edu ISTRY 1906 arden and Young- discovered that yeast fermented with sugar accumulate
Glycobiology 8130, 18/1/2005 Spring 2005 Nucleotide-sugar donors. Maor Bar-Peled, CCRC; Plant Biology Peled@ccrc.uga.edu ISTRY 1906 arden and Young- discovered that yeast fermented with sugar accumulate
189,311, , ,561, ,639, ,679, Ch13; , Carbohydrates
 Lecture 31 (12/8/17) Reading: Ch7; 258-267 Ch10; 371-373 Problems: Ch7 (text); 26,27,28 Ch7 (study-guide: applying); 2,5 Ch7 (study-guide: facts); 6 NEXT (LAST!) Reading: Chs4,6,8,10,14,16,17,18; 128-129,
Lecture 31 (12/8/17) Reading: Ch7; 258-267 Ch10; 371-373 Problems: Ch7 (text); 26,27,28 Ch7 (study-guide: applying); 2,5 Ch7 (study-guide: facts); 6 NEXT (LAST!) Reading: Chs4,6,8,10,14,16,17,18; 128-129,
The addition of sugar moiety determines the blood group
 The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
Fatty acid breakdown
 Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
Chapter 26 Biochemistry 5th edition. phospholipids. Sphingolipids. Cholesterol. db=books&itool=toolbar
 http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
BCH 4054 Chapter 19 Lecture Notes
 BCH 4054 Chapter 19 Lecture Notes 1 Chapter 19 Glycolysis 2 aka = also known as verview of Glycolysis aka The Embden-Meyerhoff Pathway First pathway discovered Common to almost all living cells ccurs in
BCH 4054 Chapter 19 Lecture Notes 1 Chapter 19 Glycolysis 2 aka = also known as verview of Glycolysis aka The Embden-Meyerhoff Pathway First pathway discovered Common to almost all living cells ccurs in
Lecture 34. Carbohydrate Metabolism 2. Glycogen. Key Concepts. Biochemistry and regulation of glycogen degradation
 Lecture 34 Carbohydrate Metabolism 2 Glycogen Key Concepts Overview of Glycogen Metabolism Biochemistry and regulation of glycogen degradation Biochemistry and regulation of glycogen synthesis What mechanisms
Lecture 34 Carbohydrate Metabolism 2 Glycogen Key Concepts Overview of Glycogen Metabolism Biochemistry and regulation of glycogen degradation Biochemistry and regulation of glycogen synthesis What mechanisms
Metabolism of pentoses, glycogen, fructose and galactose. Jana Novotna
 Metabolism of pentoses, glycogen, fructose and galactose Jana Novotna 1. The Pentose Phosphate Pathway The pentose phosphate pathway (PPP): (hexose monophosphate or 6-phosphogluconate patway) Process that
Metabolism of pentoses, glycogen, fructose and galactose Jana Novotna 1. The Pentose Phosphate Pathway The pentose phosphate pathway (PPP): (hexose monophosphate or 6-phosphogluconate patway) Process that
Content. Course (LV 21246): Fr. 12:00-14:00 16 Lectures
 Content Course (LV 21246): Fr. 12:00-14:00 16 Lectures Content 1: Overview Occurrence of carbohydrates in nature Glycopolymers and glycoconjugates Carbohydrates in animals Carbohydrates in bacteria and
Content Course (LV 21246): Fr. 12:00-14:00 16 Lectures Content 1: Overview Occurrence of carbohydrates in nature Glycopolymers and glycoconjugates Carbohydrates in animals Carbohydrates in bacteria and
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
Metabolism. Metabolic pathways. BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 11: Metabolic Pathways
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 11: Metabolic Pathways http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Metabolism Metabolism is the chemical change of
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 11: Metabolic Pathways http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Metabolism Metabolism is the chemical change of
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 16 Glycolysis 2013 W. H. Freeman and Company Chapter 16 Outline Why is glucose such a prominent fuel in all life forms? 1. Glucose
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 16 Glycolysis 2013 W. H. Freeman and Company Chapter 16 Outline Why is glucose such a prominent fuel in all life forms? 1. Glucose
BIOLOGY 111. CHAPTER 2: The Chemistry of Life Biological Molecules
 BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
Multiple choice: Circle the best answer on this exam. There are 12 multiple choice questions, each question is worth 3 points.
 CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
number Done by Corrected by Doctor Nayef Karadsheh
 number 16 Done Huda shaheen by Corrected by حسام أبو عوض Doctor Nayef Karadsheh 1 In the previous lecture, we talked about glycogen metabolism and regulation. In this sheet we will talk about the metabolism
number 16 Done Huda shaheen by Corrected by حسام أبو عوض Doctor Nayef Karadsheh 1 In the previous lecture, we talked about glycogen metabolism and regulation. In this sheet we will talk about the metabolism
1- Which of the following statements is TRUE in regards to eukaryotic and prokaryotic cells?
 Name: NetID: Exam 3 - Version 1 October 23, 2017 Dr. A. Pimentel Each question has a value of 4 points and there are a total of 160 points in the exam. However, the maximum score of this exam will be capped
Name: NetID: Exam 3 - Version 1 October 23, 2017 Dr. A. Pimentel Each question has a value of 4 points and there are a total of 160 points in the exam. However, the maximum score of this exam will be capped
METABOLISM Biosynthetic Pathways
 METABOLISM Biosynthetic Pathways Metabolism Metabolism involves : Catabolic reactions that break down large, complex molecules to provide energy and smaller molecules. Anabolic reactions that use ATP energy
METABOLISM Biosynthetic Pathways Metabolism Metabolism involves : Catabolic reactions that break down large, complex molecules to provide energy and smaller molecules. Anabolic reactions that use ATP energy
Glycogen Metabolism. BCH 340 lecture 9
 Glycogen Metabolism BC 340 lecture 9 Structure of glycogen Glycogen is homopolysaccharide formed of branched D-glucose units The primary glycosidic bond is 1-4-linkage Each branch is made of 6-12 glucose
Glycogen Metabolism BC 340 lecture 9 Structure of glycogen Glycogen is homopolysaccharide formed of branched D-glucose units The primary glycosidic bond is 1-4-linkage Each branch is made of 6-12 glucose
Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes
 Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes The major site of acetoacetate and 3-hydorxybutyrate production is in the liver. 3-hydorxybutyrate is the
Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes The major site of acetoacetate and 3-hydorxybutyrate production is in the liver. 3-hydorxybutyrate is the
Cellular Respiration
 Cellular Respiration 1. To perform cell work, cells require energy. a. A cell does three main kinds of work: i. Mechanical work, such as the beating of cilia, contraction of muscle cells, and movement
Cellular Respiration 1. To perform cell work, cells require energy. a. A cell does three main kinds of work: i. Mechanical work, such as the beating of cilia, contraction of muscle cells, and movement
Biosynthesis of Fatty Acids. By Dr.QUTAIBA A. QASIM
 Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
Chapter 3. Table of Contents. Section 1 Carbon Compounds. Section 2 Molecules of Life. Biochemistry
 Biochemistry Table of Contents Section 1 Carbon Compounds Section 2 Molecules of Life Section 1 Carbon Compounds Objectives Distinguish between organic and inorganic compounds. Explain the importance of
Biochemistry Table of Contents Section 1 Carbon Compounds Section 2 Molecules of Life Section 1 Carbon Compounds Objectives Distinguish between organic and inorganic compounds. Explain the importance of
Chapter 15 Homework Assignment
 Chapter 15 Homework Assignment The following problems will be due once we finish the chapter: 3, 5, 6, 8, 9 Chapter 15 1 Regulation of Metabolic Pathways Dynamic Steady State Fuels, such as glucose, enter
Chapter 15 Homework Assignment The following problems will be due once we finish the chapter: 3, 5, 6, 8, 9 Chapter 15 1 Regulation of Metabolic Pathways Dynamic Steady State Fuels, such as glucose, enter
Summary of fatty acid synthesis
 Lipid Metabolism, part 2 1 Summary of fatty acid synthesis 8 acetyl CoA + 14 NADPH + 14 H+ + 7 ATP palmitic acid (16:0) + 8 CoA + 14 NADP + + 7 ADP + 7 Pi + 7 H20 1. The major suppliers of NADPH for fatty
Lipid Metabolism, part 2 1 Summary of fatty acid synthesis 8 acetyl CoA + 14 NADPH + 14 H+ + 7 ATP palmitic acid (16:0) + 8 CoA + 14 NADP + + 7 ADP + 7 Pi + 7 H20 1. The major suppliers of NADPH for fatty
Energy storage in cells
 Energy storage in cells Josef Fontana EC - 58 Overview of the lecture Introduction to the storage substances of human body Overview of storage compounds in the body Glycogen metabolism Structure of glycogen
Energy storage in cells Josef Fontana EC - 58 Overview of the lecture Introduction to the storage substances of human body Overview of storage compounds in the body Glycogen metabolism Structure of glycogen
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
CHY2026: General Biochemistry UNIT 7& 8: CARBOHYDRATE METABOLISM
 CHY2026: General Biochemistry UNIT 7& 8: CARBOHYDRATE METABOLISM Metabolism Bioenergetics is the transfer and utilization of energy in biological systems The direction and extent to which a chemical reaction
CHY2026: General Biochemistry UNIT 7& 8: CARBOHYDRATE METABOLISM Metabolism Bioenergetics is the transfer and utilization of energy in biological systems The direction and extent to which a chemical reaction
Cholesterol and its transport. Alice Skoumalová
 Cholesterol and its transport Alice Skoumalová 27 carbons Cholesterol - structure Cholesterol importance A stabilizing component of cell membranes A precursor of bile salts A precursor of steroid hormones
Cholesterol and its transport Alice Skoumalová 27 carbons Cholesterol - structure Cholesterol importance A stabilizing component of cell membranes A precursor of bile salts A precursor of steroid hormones
Test Review Worksheet 1 Name: Per:
 Test Review Worksheet 1 Name: Per: 1. Put the following in order according to blood flow through the body, starting with the lungs: Lungs, right atrium, left atrium, right ventricle, left ventricle, aorta,
Test Review Worksheet 1 Name: Per: 1. Put the following in order according to blood flow through the body, starting with the lungs: Lungs, right atrium, left atrium, right ventricle, left ventricle, aorta,
A. Major parts 1. Nucleus 2. Cytoplasm a. Contain organelles (see below) 3. Plasma membrane (To be discussed in Cellular Transport Lecture)
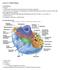 Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Link download full of Test Bank for Fundamentals of Biochemistry 4th Edition by Voet
 Link download full of Test Bank for Fundamentals of Biochemistry 4th Edition by Voet http://testbankair.com/download/test-bank-for-fundamentals-ofbiochemistry-4th-edition-by-voet/ Chapter 16: Glycogen
Link download full of Test Bank for Fundamentals of Biochemistry 4th Edition by Voet http://testbankair.com/download/test-bank-for-fundamentals-ofbiochemistry-4th-edition-by-voet/ Chapter 16: Glycogen
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Glycolysis Part 2. BCH 340 lecture 4
 Glycolysis Part 2 BCH 340 lecture 4 Regulation of Glycolysis There are three steps in glycolysis that have enzymes which regulate the flux of glycolysis These enzymes catalyzes irreversible reactions of
Glycolysis Part 2 BCH 340 lecture 4 Regulation of Glycolysis There are three steps in glycolysis that have enzymes which regulate the flux of glycolysis These enzymes catalyzes irreversible reactions of
Lecture 2: Glycogen metabolism (Chapter 15)
 Lecture 2: Glycogen metabolism (Chapter 15) First. Fig. 15.1 Review: Animals use glycogen for ENERGY STORAGE. Glycogen is a highly-branched polymer of glucose units: Basic structure is similar to that
Lecture 2: Glycogen metabolism (Chapter 15) First. Fig. 15.1 Review: Animals use glycogen for ENERGY STORAGE. Glycogen is a highly-branched polymer of glucose units: Basic structure is similar to that
Cells. Variation and Function of Cells
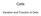 Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION
 Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Major Pathways in Carbohydrate Metabolism
 Major Pathways in Carbohydrate Metabolism 70 Stage 1: Digestion of Carbohydrates In Stage 1, the digestion of carbohydrates Begins in the mouth where salivary amylase breaks down polysaccharides to smaller
Major Pathways in Carbohydrate Metabolism 70 Stage 1: Digestion of Carbohydrates In Stage 1, the digestion of carbohydrates Begins in the mouth where salivary amylase breaks down polysaccharides to smaller
Activity: Biologically Important Molecules
 Activity: Biologically Important Molecules AP Biology Introduction We have already seen in our study of biochemistry that the molecules that comprise living things are carbon-based, and that they are thought
Activity: Biologically Important Molecules AP Biology Introduction We have already seen in our study of biochemistry that the molecules that comprise living things are carbon-based, and that they are thought
Significance and Functions of Carbohydrates. Bacterial Cell Walls
 Biochemistry 462a - Carbohydrate Function Reading - Chapter 9 Practice problems - Chapter 9: 2, 4a, 4b, 6, 9, 10, 13, 14, 15, 16a, 17; Carbohydrate extra problems Significance and Functions of Carbohydrates
Biochemistry 462a - Carbohydrate Function Reading - Chapter 9 Practice problems - Chapter 9: 2, 4a, 4b, 6, 9, 10, 13, 14, 15, 16a, 17; Carbohydrate extra problems Significance and Functions of Carbohydrates
PRINT your Name Student (FAMILY, first name) Midterm 7:00 P.M.
 PRINT your Name Student No. (FAMILY, first name) BIOCHEMISTRY 311A VERSION 1 (ONE) Midterm 7:00 P.M. Examiners: Dr. R. E. MacKenzie (69%) Dr. A. Storer (18%) Dr. W. Mushynski (13%) READ THE QUESTIONS CAREFULLY!!
PRINT your Name Student No. (FAMILY, first name) BIOCHEMISTRY 311A VERSION 1 (ONE) Midterm 7:00 P.M. Examiners: Dr. R. E. MacKenzie (69%) Dr. A. Storer (18%) Dr. W. Mushynski (13%) READ THE QUESTIONS CAREFULLY!!
In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic
 Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
An Introduction to Carbohydrates
 An Introduction to Carbohydrates Carbohydrates are a large class of naturally occurring polyhydroxy aldehydes and ketones. Monosaccharides also known as simple sugars, are the simplest carbohydrates containing
An Introduction to Carbohydrates Carbohydrates are a large class of naturally occurring polyhydroxy aldehydes and ketones. Monosaccharides also known as simple sugars, are the simplest carbohydrates containing
Enzymes Part III: regulation II. Dr. Mamoun Ahram Summer, 2017
 Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
1. Finish textbook questions from chapter 1 2. Parts of the cell lesson 3. Parts of the cell assignment
 Science 10 Dec. 3 1. Finish textbook questions from chapter 1 2. Parts of the cell lesson 3. Parts of the cell assignment Chapter Two Structure and Cell Function 2.1 The Cell as an Efficient System Thought
Science 10 Dec. 3 1. Finish textbook questions from chapter 1 2. Parts of the cell lesson 3. Parts of the cell assignment Chapter Two Structure and Cell Function 2.1 The Cell as an Efficient System Thought
Adenosine triphosphate (ATP)
 Adenosine triphosphate (ATP) 1 High energy bonds ATP adenosine triphosphate N NH 2 N -O O P O O P O- O- O O P O- O CH 2 H O H N N adenine phosphoanhydride bonds (~) H OH ribose H OH Phosphoanhydride bonds
Adenosine triphosphate (ATP) 1 High energy bonds ATP adenosine triphosphate N NH 2 N -O O P O O P O- O- O O P O- O CH 2 H O H N N adenine phosphoanhydride bonds (~) H OH ribose H OH Phosphoanhydride bonds
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
Cellular Respiration. Overview of Cellular Respiration. Lecture 8 Fall Overview of Cellular Respiration. Overview of Cellular Respiration
 Overview of Cellular Respiration 1 Cellular Respiration Lecture 8 Fall 2008 All organisms need ATP to do cellular work Cellular Respiration: The conversion of chemical energy of carbon compounds into another
Overview of Cellular Respiration 1 Cellular Respiration Lecture 8 Fall 2008 All organisms need ATP to do cellular work Cellular Respiration: The conversion of chemical energy of carbon compounds into another
number Done by Corrected by Doctor Nayef Karadsheh
 number 11 Done by حسام أبو عوض Corrected by Moayyad Al-Shafei Doctor Nayef Karadsheh 1 P a g e General Regulatory Aspects in Metabolism: We can divide all pathways in metabolism to catabolicand anabolic.
number 11 Done by حسام أبو عوض Corrected by Moayyad Al-Shafei Doctor Nayef Karadsheh 1 P a g e General Regulatory Aspects in Metabolism: We can divide all pathways in metabolism to catabolicand anabolic.
Cell Cell
 Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
4/12/17. Cells. Cell Structure. Ch. 2 Cell Structure and Func.on. Range of Cell Sizes BIOL 100
 Ch. 2 Cell Structure and Func.on BIOL 100 Cells Fundamental units of life Cell theory All living things are composed of one or more cells. The cell is the most basic unit of life. All cells come from pre-existing
Ch. 2 Cell Structure and Func.on BIOL 100 Cells Fundamental units of life Cell theory All living things are composed of one or more cells. The cell is the most basic unit of life. All cells come from pre-existing
Chapter 11. Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins.
 Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
BIOLOGY 111. CHAPTER 3: The Cell: The Fundamental Unit of Life
 BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
CELLS. Cells. Basic unit of life (except virus)
 Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
AS Level Paper 1 and 2. A2 Level Paper 1 and 3 - Topics 1-4
 Section 3.1: Biological Molecules 3.1.1 Monomers and Polymers 3.1.2 Carbohydrates 3.1.3 Lipids 3.1.4.1 Proteins 3.1.4.2 Enzymes 3.1.5.1 Nucleic acid structure 3.1.5.2 DNA Replication 3.1.6 ATP 3.1.7 Water
Section 3.1: Biological Molecules 3.1.1 Monomers and Polymers 3.1.2 Carbohydrates 3.1.3 Lipids 3.1.4.1 Proteins 3.1.4.2 Enzymes 3.1.5.1 Nucleic acid structure 3.1.5.2 DNA Replication 3.1.6 ATP 3.1.7 Water
AP Biology
 Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Tour of the Cell (1) 2007-2008 Types of cells Prokaryote bacteria cells - no organelles - organelles Eukaryote animal cells Eukaryote plant cells Cell Size Why organelles? Specialized structures - specialized
Chapter 18: Carbohydrate Metabolism
 Vocabulary Biotin: a CO2 carrier molecule Cori Cycle: a pathway in carbohydrate metabolism that links glycolysis in the liver with gluconeogenesis in the liver Debranching Enzyme: an enzyme that hydrolyzes
Vocabulary Biotin: a CO2 carrier molecule Cori Cycle: a pathway in carbohydrate metabolism that links glycolysis in the liver with gluconeogenesis in the liver Debranching Enzyme: an enzyme that hydrolyzes
Gluconeogenesis. Gluconeogenesis / TCA 11/12/2009. Free energy changes in glycolysis 11/13/2009
 Gluconeogenesis Gluconeogenesis / TCA 11/12/2009 Gluconeogenesis is the process whereby precursors such as lactate, pyruvate, glycerol, and amino acids are converted to glucose. Fasting requires all the
Gluconeogenesis Gluconeogenesis / TCA 11/12/2009 Gluconeogenesis is the process whereby precursors such as lactate, pyruvate, glycerol, and amino acids are converted to glucose. Fasting requires all the
Glycoproteins and Mucins. B.Sopko
 Glycoproteins and Mucins B.Sopko Content Glycoproteins: Structures and Linkages Interconversions and activation of dietary sugars Other pathways of sugar nucleotide metabolism Biosynthesis of oligosaccharides
Glycoproteins and Mucins B.Sopko Content Glycoproteins: Structures and Linkages Interconversions and activation of dietary sugars Other pathways of sugar nucleotide metabolism Biosynthesis of oligosaccharides
Chemical Energy. Valencia College
 9 Pathways that Harvest Chemical Energy Valencia College 9 Pathways that Harvest Chemical Energy Chapter objectives: How Does Glucose Oxidation Release Chemical Energy? What Are the Aerobic Pathways of
9 Pathways that Harvest Chemical Energy Valencia College 9 Pathways that Harvest Chemical Energy Chapter objectives: How Does Glucose Oxidation Release Chemical Energy? What Are the Aerobic Pathways of
A. Lipids: Water-Insoluble Molecules
 Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Oxidation of Long Chain Fatty Acids
 Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions
 Chemistry of Life Revision: The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions Atoms exchange electrons with other elements to form
Chemistry of Life Revision: The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions Atoms exchange electrons with other elements to form
Nucleic acids. Nucleic acids are information-rich polymers of nucleotides
 Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Biology: Life on Earth Chapter 3 Molecules of life
 Biology: Life on Earth Chapter 3 Molecules of life Chapter 3 Outline 3.1 Why Is Carbon So Important in Biological Molecules? p. 38 3.2 How Are Organic Molecules Synthesized? p. 38 3.3 What Are Carbohydrates?
Biology: Life on Earth Chapter 3 Molecules of life Chapter 3 Outline 3.1 Why Is Carbon So Important in Biological Molecules? p. 38 3.2 How Are Organic Molecules Synthesized? p. 38 3.3 What Are Carbohydrates?
WELCOME TO BIOLOGY 11. Mr. Gandha
 WELCOME TO BIOLOGY 11 Mr. Gandha TOPICS OF BIOLOGY 11 Chemicals of life Cells Evolution Taxonomy Microbio Plants Animals BIOLOGY THIS SEMESTER Review of Biology and Processes Adaptation and Evolution:
WELCOME TO BIOLOGY 11 Mr. Gandha TOPICS OF BIOLOGY 11 Chemicals of life Cells Evolution Taxonomy Microbio Plants Animals BIOLOGY THIS SEMESTER Review of Biology and Processes Adaptation and Evolution:
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
The Cell. Copyright 2003 Pearson Education, Inc. publishing as Benjamin Cummings
 The Cell Cell Theory The cell is the basic structural and functional unit of life The organism activity depends on individual and collective activity of cells Biochemical activities of cells are dictated
The Cell Cell Theory The cell is the basic structural and functional unit of life The organism activity depends on individual and collective activity of cells Biochemical activities of cells are dictated
Lipid Metabolism. Catabolism Overview
 Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
Chemical Composition of the Cell. B. Balen
 Chemical Composition of the Cell B. Balen Table 2-2 Molecular Biology of the Cell ( Garland Science 2008) 1. Water the most abundant substance in the cell! Where did it come from? several hypothesis: -
Chemical Composition of the Cell B. Balen Table 2-2 Molecular Biology of the Cell ( Garland Science 2008) 1. Water the most abundant substance in the cell! Where did it come from? several hypothesis: -
Lesson 2. Biological Molecules. Introduction to Life Processes - SCI 102 1
 Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
NBCE Mock Board Questions Biochemistry
 1. Fluid mosaic describes. A. Tertiary structure of proteins B. Ribosomal subunits C. DNA structure D. Plasma membrane structure NBCE Mock Board Questions Biochemistry 2. Where in the cell does beta oxidation
1. Fluid mosaic describes. A. Tertiary structure of proteins B. Ribosomal subunits C. DNA structure D. Plasma membrane structure NBCE Mock Board Questions Biochemistry 2. Where in the cell does beta oxidation
Metabolism Energy Pathways Biosynthesis. Catabolism Anabolism Enzymes
 Topics Microbial Metabolism Metabolism Energy Pathways Biosynthesis 2 Metabolism Catabolism Catabolism Anabolism Enzymes Breakdown of complex organic molecules in order to extract energy and dform simpler
Topics Microbial Metabolism Metabolism Energy Pathways Biosynthesis 2 Metabolism Catabolism Catabolism Anabolism Enzymes Breakdown of complex organic molecules in order to extract energy and dform simpler
cholesterol structure Cholesterol FAQs Cholesterol promotes the liquid-ordered phase of membranes Friday, October 15, 2010
 cholesterol structure most plasma cholesterol is in the esterified form (not found in cells or membranes) cholesterol functions in all membranes (drives formation of lipid microdomains) cholesterol is
cholesterol structure most plasma cholesterol is in the esterified form (not found in cells or membranes) cholesterol functions in all membranes (drives formation of lipid microdomains) cholesterol is
SBI3U7 Cell Structure & Organelles. 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells
 SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
Carbohydrates. What are they? What do cells do with carbs? Where do carbs come from? O) n. Formula = (CH 2
 Carbohydrates What are they? Formula = (C 2 O) n where n > 3 Also called sugar Major biomolecule in body What do cells do with carbs? Oxidize them for energy Store them to oxidize later for energy Use
Carbohydrates What are they? Formula = (C 2 O) n where n > 3 Also called sugar Major biomolecule in body What do cells do with carbs? Oxidize them for energy Store them to oxidize later for energy Use
BIOCHEMISTRY. How Are Macromolecules Formed? Dehydration Synthesis or condensation reaction Polymers formed by combining monomers and removing water.
 BIOCHEMISTRY Organic compounds Compounds that contain carbon are called organic. Inorganic compounds do not contain carbon. Carbon has 4 electrons in outer shell. Carbon can form covalent bonds with as
BIOCHEMISTRY Organic compounds Compounds that contain carbon are called organic. Inorganic compounds do not contain carbon. Carbon has 4 electrons in outer shell. Carbon can form covalent bonds with as
At the membrane frontier: A prospectus on the remarkable evolutionary conservation of polyprenols and polyprenylphosphates
 At the membrane frontier: A prospectus on the remarkable evolutionary conservation of polyprenols and polyprenylphosphates The MIT Faculty has made this article openly available. Please share how this
At the membrane frontier: A prospectus on the remarkable evolutionary conservation of polyprenols and polyprenylphosphates The MIT Faculty has made this article openly available. Please share how this
Chapter 9. Cellular Signaling
 Chapter 9 Cellular Signaling Cellular Messaging Page 215 Cells can signal to each other and interpret the signals they receive from other cells and the environment Signals are most often chemicals The
Chapter 9 Cellular Signaling Cellular Messaging Page 215 Cells can signal to each other and interpret the signals they receive from other cells and the environment Signals are most often chemicals The
Glycosaminoglycans: Anionic polysaccharide chains made of repeating disaccharide units
 Glycosaminoglycans: Anionic polysaccharide chains made of repeating disaccharide units Glycosaminoglycans present on the animal cell surface and in the extracellular matrix. Glycoseaminoglycans (mucopolysaccharides)
Glycosaminoglycans: Anionic polysaccharide chains made of repeating disaccharide units Glycosaminoglycans present on the animal cell surface and in the extracellular matrix. Glycoseaminoglycans (mucopolysaccharides)
Early scientists who observed cells made detailed sketches of what they saw.
 Early scientists who observed cells made detailed sketches of what they saw. Early scientists who observed cells made detailed sketches of what they saw. CORK Early scientists who observed cells made detailed
Early scientists who observed cells made detailed sketches of what they saw. Early scientists who observed cells made detailed sketches of what they saw. CORK Early scientists who observed cells made detailed
What are the molecules of life?
 Molecules of Life What are the molecules of life? Organic Compounds Complex Carbohydrates Lipids Proteins Nucleic Acids Organic Compounds Carbon- hydrogen based molecules From Structure to Function Ø Carbon
Molecules of Life What are the molecules of life? Organic Compounds Complex Carbohydrates Lipids Proteins Nucleic Acids Organic Compounds Carbon- hydrogen based molecules From Structure to Function Ø Carbon
Highlights Pentose Phosphate Pathway
 Highlights Pentose Phosphate Pathway 1. The pentose phosphate pathway (PPP) is an interchange of metabolic pathways. 2. It is important to cells as a) an important source of NADPH, b) an important source
Highlights Pentose Phosphate Pathway 1. The pentose phosphate pathway (PPP) is an interchange of metabolic pathways. 2. It is important to cells as a) an important source of NADPH, b) an important source
Topic 3.1 Nutrients. - Lipids are an essential part of the and are a part of cell in the body.
 Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
1. endoplasmic reticulum This is the location where N-linked oligosaccharide is initially synthesized and attached to glycoproteins.
 Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
Biology 4410 Name Spring 2006 Exam 2 A. Multiple Choice, 2 pt each Pick the best choice from the list of choices, and write it in the space provided. Some choices may be used more than once, and other
CHE 242 Exam 3 Practice Questions
 CHE 242 Exam 3 Practice Questions Glucose metabolism 1. Below is depicted glucose catabolism. Indicate on the pathways the following: A) which reaction(s) of glycolysis are irreversible B) where energy
CHE 242 Exam 3 Practice Questions Glucose metabolism 1. Below is depicted glucose catabolism. Indicate on the pathways the following: A) which reaction(s) of glycolysis are irreversible B) where energy
Rawan almujaibel. Ayman Musleh. Dr. Nayef
 12 Rawan almujaibel Ayman Musleh Ayman Musleh Dr. Nayef In the previous lecture we talked about digestion and absorption of carbohydrates. In this lecture we will be talking about glycolysis. Glycolysis
12 Rawan almujaibel Ayman Musleh Ayman Musleh Dr. Nayef In the previous lecture we talked about digestion and absorption of carbohydrates. In this lecture we will be talking about glycolysis. Glycolysis
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam 3 BIOL 1406, Fall 2012 HCC Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) When biologists wish to study the internal ultrastructure
Exam 3 BIOL 1406, Fall 2012 HCC Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) When biologists wish to study the internal ultrastructure
Carbohydrate Metabolism 2 Supplemental Reading
 Carbohydrate Metabolism 2 Supplemental Reading Key Concepts - Overview of glycogen metabolism - Biochemistry and regulation glycogen degradation - Biochemistry and regulation of glycogen synthesis - Control
Carbohydrate Metabolism 2 Supplemental Reading Key Concepts - Overview of glycogen metabolism - Biochemistry and regulation glycogen degradation - Biochemistry and regulation of glycogen synthesis - Control
DEPARTMENT: Chemistry
 CODE: CHEM-236 TITLE: Biochemistry Institute: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: Upon completion of this course the student will be able to recognize and draw the structure and state the nature
CODE: CHEM-236 TITLE: Biochemistry Institute: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: Upon completion of this course the student will be able to recognize and draw the structure and state the nature
Carbohydrate. Metabolism
 Carbohydrate Metabolism Dietary carbohydrates (starch, glycogen, sucrose, lactose Mouth salivary amylase Summary of Carbohydrate Utilization Utilization for energy (glycolysis) ligosaccharides and disaccharides
Carbohydrate Metabolism Dietary carbohydrates (starch, glycogen, sucrose, lactose Mouth salivary amylase Summary of Carbohydrate Utilization Utilization for energy (glycolysis) ligosaccharides and disaccharides
An aldose contains an aldehyde functionality A ketose contains a ketone functionality
 RCT Chapter 7 Aldoses and Ketoses; Representative monosaccharides. (a)two trioses, an aldose and a ketose. The carbonyl group in each is shaded. An aldose contains an aldehyde functionality A ketose contains
RCT Chapter 7 Aldoses and Ketoses; Representative monosaccharides. (a)two trioses, an aldose and a ketose. The carbonyl group in each is shaded. An aldose contains an aldehyde functionality A ketose contains
Tala Saleh. Razi Kittaneh ... Nayef Karadsheh
 Tala Saleh Razi Kittaneh... Nayef Karadsheh β-oxidation of Fatty Acids The oxidation of fatty acids occurs in 3 steps: Step 1: Activation of the Fatty acid FA + HS-CoA + ATP FA-CoA + AMP + PPi - The fatty
Tala Saleh Razi Kittaneh... Nayef Karadsheh β-oxidation of Fatty Acids The oxidation of fatty acids occurs in 3 steps: Step 1: Activation of the Fatty acid FA + HS-CoA + ATP FA-CoA + AMP + PPi - The fatty
Don t Freak Out. Test on cell organelle on Friday!
 Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Practice Quiz 1 AP Bio Sept 2016 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The element present in all organic molecules is A) hydrogen.
Practice Quiz 1 AP Bio Sept 2016 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The element present in all organic molecules is A) hydrogen.
Chapter 22. Before the class. 10 Steps of glycolysis. Outline. Can you tell the ten steps of glycolysis? Do you know how glucoses are
 Chapter 22 Gluconeogenesis, Glycogen metabolism, and the Pentose Phosphate Pathway Reginald H. Garrett Charles M. Grisham 1 Before the class Can you tell the ten steps of glycolysis? Do you know how glucoses
Chapter 22 Gluconeogenesis, Glycogen metabolism, and the Pentose Phosphate Pathway Reginald H. Garrett Charles M. Grisham 1 Before the class Can you tell the ten steps of glycolysis? Do you know how glucoses
number Done by Corrected by Doctor Faisal Al-Khatibe
 number 24 Done by Mohammed tarabieh Corrected by Doctor Faisal Al-Khatibe 1 P a g e *Please look over the previous sheet about fatty acid synthesis **Oxidation(degradation) of fatty acids, occurs in the
number 24 Done by Mohammed tarabieh Corrected by Doctor Faisal Al-Khatibe 1 P a g e *Please look over the previous sheet about fatty acid synthesis **Oxidation(degradation) of fatty acids, occurs in the
