Impact of Biological and Environmental Variabilities on Biological Monitoring An Approach Using Toxicokinetic Models
|
|
|
- Matthew Holmes
- 5 years ago
- Views:
Transcription
1 ONLINE SUPPLEMENTAL APPENDIX Impact of Biological and Environmental Variabilities on Biological Monitoring An Approach Using Toxicokinetic Models by A. Berthet, A. de Batz, R. Tardif, G. Charest-Tardif, G. Truchon, D. Vernez, and P.O. Droz Table AI presents the data used in the CBTK models for all 14 chemicals. The first line for each chemical indicates the physiological identity of each compartment and chemical species. Then individual parameters are presented. Flow data both at rest and at 50W are indicated. Symbol nomenclature used in Table AI is shown in Table AII. The equations used to calculate each model parameter in Table AI are described by Pierrehumbert et al. (10) in more detail. TABLE AI. Chemical Specific Parameters Used in the Simulations for Each Substance Studied with CBTK Models Chemical/Volumes of Distribution Parameters Symbol At Rest At 50 W Arsenic (As) Central= Total body water (TBW), Metabolite 1 (M1) = TBW Exposure (µg/m 3 ) C exp 10 C Total body volume (TBV) A FV C Liver blood flow B BF M (24) 0.16 (24) Total body volume (TBV): blood partition coefficient for inorganic arsenic (-) P TBV:blood 45 (25) TBV: blood partition coefficient for monomethylarsonic acid (MMA) (-) P TBV:blood, M1 8.6 (25) TBV:blood partition coefficient for dimethylarsonic acid (DMA) (-) P TBV:blood, M2 6.7 (25) Liver permeability coefficient for inorganic arsenic (-) P p (25) Liver permeability coefficient for MMA (-) P M (25) Maximum metabolism rate of inorganic arsenic (µmol/h/kg 0.75 ) V MM (25) Michaelis-Menten constant for inorganic arsenic (µmol/l) K MM (25) Maximum metabolism rate of MMA (µmol/h/kg 0.75 ) V MM (25) Michaelis-Menten constant for MMA (µmol/l) K MM2 0.5 (25) Renal Clearance (l/h per kg -0.3 ) RC C 24 (24) Cadmium (Cd) Central = Total body water, Peripheral = kidneys Exposure (µg/m 3 ) C exp 50 D (14) - Kidneys Kidney volume A FV P (25) Kidney blood flow B BF P 0.11 (25) 0.2 (25) TBW: blood affinity coefficient (-) P TBW:blood 60 E Kidney permeability coefficient (-) P P 50 E
2 Kidney: blood affinity coefficient (-) P kidney:blood 60,000,000,000 F Renal Clearance (l/h per kg -0.3 ) RC C 200 F Carbon monoxide (CO) Central=Whole blood Exposure (ppm) C exp 35 (14) Deposit rate (%) 100 G Blood volume A FV P 0.07 (25) Blood: air affinity coefficient (-) P blood:air 500 H Hemoglobin concentration (g Hb/l) 150 (1) Link rate (%) 85 (1) Affinity (ml CO/g Hb) 1.49 (1) Chromium (Cr) Central=Total body water, Peripheral=richly perfused tissues (RP) (i.e., heart, liver, brain) Exposure (µg/m 3 ) C exp 50 I (14) - RP RP volume A FV P (10) RP blood flow B BF P 0.70 (10) 0.43 (10) TBW: blood affinity coefficient (-) P TBW:blood 17 J RP permeability coefficient (-) P P 4 K RP: blood affinity coefficient (-) P RP:blood 400,000 L Renal Clearance (l/h per kg -0.3 ) RC C (25) Cobalt (Co) Central=TBW, Peripheral=richly perfused tissues (RP) Exposure (µg/m 3 ) C exp 50 M (14) - RP RP volume A FV P (10) RP blood flow B BF P 0.70 (10) 0.43 (10) TBW: blood affinity coefficient (-) P TBW:blood 2 N RP permeability coefficient (-) P P 65 N RP: blood affinity coefficient (-) P RP:blood O Urinary excretion constant (h -1 /kg -0.3 ) k u,c P Ethylbenzene Central=Richly (RP) and slowly perfused tissues (SP), Peripheral=Fat Exposure (ppm) C exp 100 (14) Deposit rate (%) 60 (14) RP and SP volume 1 FV C Fat Fat volume A FV P 0.19 (24) Fat blood flow B BF P 0.05 (24) 0.06 (24) Liver blood flow B BF M (24) 0.16 (24) RP and SP: air partition coefficient (-) P RP and SP:air (24) Blood: air partition coefficient (-) P blood:air (24) Fat: air partition coefficient (-) P fat:air (24) Maximum metabolism rate (µmol/h/kg 0.75 ) V MM (24) Michaelis-Menten constant (µmol/l) K MM (24) Ethylbenzene fraction metabolized to mandelic acid / Ethylbenzene fraction metabolized to phenylglyoxilic acid F 2 /F 1 2 (24) Mandelic acid excretion rate (h -1 /kg -0.3 ) k u,m (24)
3 Fluorides Central=Total body water, Peripheral=bones Exposure (mg/m 3 ) C exp 2.5 Q (14) Skeleton volume A FV P (26) Skeleton blood flow B BF P (26) TBW: blood affinity coefficient (-) P TBW:blood 3 R Bones permeability coefficient (-) P P R Bones: blood affinity coefficient (-) P bones:blood 662 S Renal Clearance (l/h per kg -0.3 ) RC C T Ethyleneglycol monomethylether (EGEE) Central=Total body water, Metabolite1 (M1)=Total body water Exposure (ppm) C exp 5 (14) Deposit rate (%) 80 (27) Liver blood flow B BF P 0.26 (10,24) 0.16 (10,24) Water: air partition coefficient EGEE (-) P water:air (27) Blood: air partition coefficient EGEE (-) P blood:air (27) Water: blood partition coefficient of 2-ethoxyacetic acid (EAA) (-) P water:blood 1.00 (28) Maximum metabolism rate (µmol/h/kg 0.75 ) V MM (29) Michaelis-Menten constant (µmol/l) K MM (29) EGEE fraction metabolized to EAA / fraction metabolized to ethylene glycol (EG) (-) F 2 /F (29) EAA renal Clearance (l/h per kg -0.3 ) k u,m D Inorganic mercury (IHg) Central Total body water, Peripheral=kidneys Exposure (µg/m 3 ) C exp 25 (14) - Kidney Kidney volume A FV P (26) Kidney blood flow B BF P 0.11 (26) Kidney: blood partition coefficient (-) P kidney:blood 473,396,562 U Kidney permeability coefficient (-) P P U Renal excretion constant (h 1 per kg 0.3 ) k u,c U Biliary excretion constant (h 1 per kg 0.3 ) k f,p U Methyl isobutyl ketone (MIBK) Central=Total body water, Peripheral=Fat Exposure (ppm) C exp 50 (14) Deposit rate (%) 80 (30) Fat volume A FV P 0.19 (10) Fat blood flow B BF P 0.05 (10) 0.06 (10) Water: air partition coefficient (-) P water:air 79 (14) Blood: air partition coefficient (-) P blood:air 90 (14) Fat: air partition coefficient (-) P fat:air 926 (14) Urinary excretion constant (h -1 /kg -0.3 ) k u,p V Metabolism constant (h -1 /kg -0.3 ) K MM V Pentachlorophenol Central=Total body water, Metabolite=undefined
4 Exposure (µg/m 3 ) C exp 500 (14) Deposit rate (%) 80 W Central: air affinity coefficient (-) P central:air 125,000 X Blood: air affinity coefficient (-) P blood:air 250,000 X Urinary excretion constant (h -1 /kg -0.3 ) for free PCP k u,p X Metabolism constant (h -1 /kg -0.3 ) K MM X Urinary excretion constant (h -1 /kg -0.3 ) for combined PCP k u,p X Phenol (Ph) Central=TB Exposure (ppm) C exp 5 (14) Total body water (TBW) A FV C Renal Clearance (l/h per kg -0.3 ) RC C Y Lead (Pb) Central=Total body water minus peripheral compartment, Peripheral=skeleton Exposure (µg/m 3 ) C exp 150 (14) Total body volume (TBV) A FV C Skeleton Skeleton volume A FV P (26) Skeleton blood flow B BF P (26) TBV: blood partition coefficient (-) P TBV:blood Z Bones: blood partition coefficient (-) P bones:blood 31,881,434 Z Renal Clearance (l/h per kg -0.3 ) RC C (31) Toluene (TOL) Central=Richly (RP) and slowly perfused tissues (SP), Peripheral=Fat, Metabolite=undefined Exposure (ppm) C exp 50 (14) RP and SP volume A FV C Fat Liver Fat volume A FV P 0.19 (10) Liver volume (metabolism) A FV M (10) Fat blood flow B BF P 0.05 (24) 0.06 (24) Liver blood flow B BF M (24) 0.16 (24) Blood: air partition coefficient (-) P blood:air 15.6 (32) SP: air partition coefficient (-) P SP:air 27.7 (33) Fat: air partition coefficient (-) P fat:air (33) Michaelis-Menten maximum rate (µmol/h/kg 0.75 ) V MM (10) Michaelis-Menten constant (µmol/l) K MM (10) Fraction of TOL metabolized to hippuric acid (HA)/Fraction of TOL metabolised to o-cresol (-) F 2 /F (10) o-cresol excretion rate constant (l/h per kg -0.3 ) k u,m (34) A Expressed in fraction of body weight. B Expressed in fraction of cardiac output. C ACGIH TLV-TWA. (14) D Simulation of an exposure to 50 µg/m 3, but only 45% of dust is considered to reach the pulmonary alveoli. (26) E Determined according to the following considerations: (1) cadmium half-life in blood is 100 days (25) ; (2) cadmium blood concentration at steady state is about 50 nmol/l. (25) F Determined according to the following considerations: (1) urinary cadmium half-life is 20 years; (25) (2) cadmium half-life in body is 10 years (25).
5 G Deposit rate is supposed equal to 100% because of its small molecular weight H Determined according to the following consideration: the carboxyhemoglobin half-life is 5 hours. (25) I Simulation of an exposure to 50 µg/m 3, but only 15% of smoke is considered to reach the pulmonary alveoli; (26) of this 15%, 60% is considered soluble and distributed in the central compartment directly, and the other 40% was taken to be insoluble and distributed with a half-life of 120 days, (35) which means an approximate constant flow of 0.15 µmol/hour. J Determined according to the following considerations, (1) kidney clearance is calculated from glomerular filtration; (2) the half-life of urinary elimination is 7 hours. (25) K Determined according to the following consideration: the slow half-life of central compartment is 30 hours. (25) L Determined according to the following consideration: the half-life of the peripheral compartment is 3 years. (25) M Workers are exposed to 50 µg/m 3, but only 30% of mixed dust and smoke lay down on pulmonary alveoli. (26) N Determined according to the following considerations: (1) the half-life of transfer between the central compartment and the peripheral compartment is 12 hours; (25) (2) cobalt blood concentration at steady state is about 40 nmol/l. (25) O Determined according to the following consideration: the cobalt half-life in richly perfused tissues is 2 years. (25) P Determined according to the following considerations: the urinary and blood cobalt half-life in RP tissue is 3 days. (25) Q Simulation of an exposure to 2.5 mg/m 3, but only 45% of dust is considered to reach the pulmonary alveoli. (26) R Determined according to the following considerations: (1) kidney clearance is equal to the glomerular filtration; (14) (2) fluorides half-life in TBW is 6 hours; (25) (3) 50% of fluorides are quickly eliminated and the other 50% accumulate in bones. (25) S Determined according to the following consideration: the fluorides half-life in bones is 18 days. (25) T Fluorides clearance is equal to the glomerular filtration. (31) U Determined according to following considerations: (1) at steady state, 90% of the total body burden is found in the kidney; (36) (2) the half-life of Hg in blood is 3 days; (31) (3) the half-life of Hg in kidney is 60 days; (31) (4) at steady state, 58% is excreted in urine. (36) V Determined according to following considerations: (1) elimination by exhalation represents about 10% of the absorbed dose; (2) urinary elimination represents 0.04% of the absorbed dose; (3) elimination by metabolism represents the rest of the absorbed dose. (30) W Value chosen by default in this study for solvents. X Determined according to the following considerations: (1) elimination percentages of free and combined PCP in urine are 12 and 74 % respectively; (37) (2) the rest is eliminated in the expired air; (3) free PCP clearance is l/hour; (37) and (4) plasmatic concentration at the end of shiftwork is next to 19 µmol/l. (25) Y Determined according to the half-life of Ph in the whole body hours. (14) Z Determined according to the half-life of Pb in blood, 37.5 days, and in the skeleton, 20 years. (31)
6 TABLE AII. Symbol Nomenclature Used in Table AI Symbol Definition Units BF i Fraction of cardiac output in the compartment i C exp Exposure dose µg/m 3 or ppm F 2 /F 1 Fraction differentiation between metabolite 1 and 2 FV i Volume of compartment i expressed as fraction of body weight k f,i Feces excretion rate constant for compartment i l/h per kg -0.3 K Mi Michaelis-Menten constant for metabolite i µmol/l or l/h per kg -0.3 k u,i Urine excretion rate constant for compartment i l/h per kg -0.3 P i:j Affinity or partition coefficient between media i and media j P Mi Permeability of metabolite compartment P p Permeability of peripheral compartment RC i Renal clearance from compartment i l/h per kg -0.3 V Mi Michaelis-Menten maximum rate for metabolite i µmol/h per kg 0.75
Application of BEls. rigid lines between safe and dangerous concentrations nor are they an index of toxicity.
 Application of BEls BEls' are intended as guidelines to be used in the evaluation of potential health hazards in the practice of occupational hygiene. BEls' do not indicate a sharp distinction between
Application of BEls BEls' are intended as guidelines to be used in the evaluation of potential health hazards in the practice of occupational hygiene. BEls' do not indicate a sharp distinction between
Biological Exposure Indices (BEI)
 Biological Exposure Indices (BEI) REVIEW March 2018 CONTENTS 1.0 Introduction 2 2.0 Review of BEIs 4 2.1 Arsenic (elemental and soluble inorganic compounds) 5 2.2 Benzene 6 2.3 Carbon disulphide 7 2.4
Biological Exposure Indices (BEI) REVIEW March 2018 CONTENTS 1.0 Introduction 2 2.0 Review of BEIs 4 2.1 Arsenic (elemental and soluble inorganic compounds) 5 2.2 Benzene 6 2.3 Carbon disulphide 7 2.4
Basic Concepts of TDM
 TDM Lecture 1 5 th stage What is TDM? Basic Concepts of TDM Therapeutic drug monitoring (TDM) is a branch of clinical pharmacology that specializes in the measurement of medication concentrations in blood.
TDM Lecture 1 5 th stage What is TDM? Basic Concepts of TDM Therapeutic drug monitoring (TDM) is a branch of clinical pharmacology that specializes in the measurement of medication concentrations in blood.
BASIC PHARMACOKINETICS
 BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
BASIC PHARMACOKINETICS MOHSEN A. HEDAYA CRC Press Taylor & Francis Croup Boca Raton London New York CRC Press is an imprint of the Taylor & Francis Group, an informa business Table of Contents Chapter
It the process by which a drug reversibly leaves blood and enter interstitium (extracellular fluid) and/ or cells of tissues.
 It the process by which a drug reversibly leaves blood and enter interstitium (extracellular fluid) and/ or cells of tissues. Primarily depends on: 1.Regional blood flow. 2.Capillary permeability. 3.Protein
It the process by which a drug reversibly leaves blood and enter interstitium (extracellular fluid) and/ or cells of tissues. Primarily depends on: 1.Regional blood flow. 2.Capillary permeability. 3.Protein
Naval Medical Research Unit Dayton
 Naval Medical Research Unit Dayton AVAILABILITY OF ACUTE AND/OR SUBACUTE TOXICOKI- NETIC DATA FOR SELECT COMPOUNDS FOR THE RAT AND PHYSIOLOGICALLY BASED PHARMACOKINETIC (PBPK) MODELS FOR RATS AND HUMANS
Naval Medical Research Unit Dayton AVAILABILITY OF ACUTE AND/OR SUBACUTE TOXICOKI- NETIC DATA FOR SELECT COMPOUNDS FOR THE RAT AND PHYSIOLOGICALLY BASED PHARMACOKINETIC (PBPK) MODELS FOR RATS AND HUMANS
Pharmacokinetics I. Dr. M.Mothilal Assistant professor
 Pharmacokinetics I Dr. M.Mothilal Assistant professor DRUG TRANSPORT For a drug to produce a therapeutic effect, it must reach to its target and it must accumulate at that site to reach to the minimum
Pharmacokinetics I Dr. M.Mothilal Assistant professor DRUG TRANSPORT For a drug to produce a therapeutic effect, it must reach to its target and it must accumulate at that site to reach to the minimum
Unit 2b: EXCRETION OF DRUGS. Ms.M.Gayathri Mpharm (PhD) Department of Pharmaceutics Krishna Teja Pharmacy college Subject code: 15R00603 (BPPK)
 Unit 2b: EXCRETION OF DRUGS By Ms.M.Gayathri Mpharm (PhD) Department of Pharmaceutics Krishna Teja Pharmacy college Subject code: 15R00603 (BPPK) Excretion, along with metabolism and tissue redistribution,
Unit 2b: EXCRETION OF DRUGS By Ms.M.Gayathri Mpharm (PhD) Department of Pharmaceutics Krishna Teja Pharmacy college Subject code: 15R00603 (BPPK) Excretion, along with metabolism and tissue redistribution,
Reverse Dosimetry From Human Biomonitoring and In Vitro Concentration- Response Data
 Reverse Dosimetry From Human Biomonitoring and In Vitro Concentration- Response Data Lancaster, 11 th July 2014 George Loizou The Current Environmental Risk Assessment Paradigm Humans are exposed to thousands
Reverse Dosimetry From Human Biomonitoring and In Vitro Concentration- Response Data Lancaster, 11 th July 2014 George Loizou The Current Environmental Risk Assessment Paradigm Humans are exposed to thousands
General Principles of Pharmacology and Toxicology
 General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
EVE 491/591 Toxicology. Toxicant Entry into the Body 2/19/2018. Absorption and Fate of a Toxicant
 EVE 491/591 Toxicology Lecture #7 1. Absorption of Toxicants 2. Case study Part VI Toxicant Entry into the Body Toxicants must defeat barriers to absorption The respiratory system The digestive system
EVE 491/591 Toxicology Lecture #7 1. Absorption of Toxicants 2. Case study Part VI Toxicant Entry into the Body Toxicants must defeat barriers to absorption The respiratory system The digestive system
TOXIC AND ESSENTIAL ELEMENTS
 TOXIC AND ESSENTIAL ELEMENTS Assessment of Key Minerals and Harmful Metals Whole Blood, Red Blood Cell and Serum Elements Urine Toxic and Essential Elements Creatinine Clearance Hair Elements Fecal Metals
TOXIC AND ESSENTIAL ELEMENTS Assessment of Key Minerals and Harmful Metals Whole Blood, Red Blood Cell and Serum Elements Urine Toxic and Essential Elements Creatinine Clearance Hair Elements Fecal Metals
DRUG DISTRIBUTION. Distribution Blood Brain Barrier Protein Binding
 DRUG DISTRIBUTION Distribution Blood Brain Barrier Protein Binding DRUG DISTRIBUTION Drug distribution is a reversible transport of drug through the body by the systemic circulation The drug molecules
DRUG DISTRIBUTION Distribution Blood Brain Barrier Protein Binding DRUG DISTRIBUTION Drug distribution is a reversible transport of drug through the body by the systemic circulation The drug molecules
Industrial Toxicology
 Industrial Toxicology Learning Objectives Know the assumptions of the doseresponse and time-course curves Be able to define and label key points of a curve Know the difference between potency and efficacy
Industrial Toxicology Learning Objectives Know the assumptions of the doseresponse and time-course curves Be able to define and label key points of a curve Know the difference between potency and efficacy
Renal Disease and PK/PD. Anjay Rastogi MD PhD Division of Nephrology
 Renal Disease and PK/PD Anjay Rastogi MD PhD Division of Nephrology Drugs and Kidneys Kidney is one of the major organ of drug elimination from the human body Renal disease and dialysis alters the pharmacokinetics
Renal Disease and PK/PD Anjay Rastogi MD PhD Division of Nephrology Drugs and Kidneys Kidney is one of the major organ of drug elimination from the human body Renal disease and dialysis alters the pharmacokinetics
Ecotoxicology. Toxicology Uptake and distribution of xenobiotics
 Ecotoxicology Toxicology Uptake and distribution of xenobiotics Functions of the biological membrane Maintain homeostasis Selective uptake and excretion Excitability Ca ++ ph=7.4 [conc. 2] + + + + + +
Ecotoxicology Toxicology Uptake and distribution of xenobiotics Functions of the biological membrane Maintain homeostasis Selective uptake and excretion Excitability Ca ++ ph=7.4 [conc. 2] + + + + + +
ENVIRONMENTAL TOXICOLOGY
 ENVIRONMENTAL TOXICOLOGY Chapter 4 Toxicokinetics Mohd Amir Bin Arshad Toxicokinetics study on how a substance gets into the body and what happens to it in the body" The kinetics (movement) of substances
ENVIRONMENTAL TOXICOLOGY Chapter 4 Toxicokinetics Mohd Amir Bin Arshad Toxicokinetics study on how a substance gets into the body and what happens to it in the body" The kinetics (movement) of substances
Regulation of fluid and electrolytes balance
 Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
SUPPLEMENTARY DATA. Supplementary Table S1. Clinical characteristics of the study subjects.*
 Supplementary Table S1. Clinical characteristics of the study subjects.* T2D ND n (F/M) 66 (21/45) 25 (7/18) Age (years) 61.8 ± 6.9 49.4 ± 7.3 # Body weight (kg) 95 ± 16 105 ± 13 # Body mass index (kg.
Supplementary Table S1. Clinical characteristics of the study subjects.* T2D ND n (F/M) 66 (21/45) 25 (7/18) Age (years) 61.8 ± 6.9 49.4 ± 7.3 # Body weight (kg) 95 ± 16 105 ± 13 # Body mass index (kg.
A Regional Model of Lung Metabolism for Improving Species Dependent Descriptions of 1,3-Butadiene and its Metabolites
 A Regional Model of Lung Metabolism for Improving Species Dependent Descriptions of 1,3-Butadiene and its Metabolites Harvey Clewell, Jerry Campbell, and Cynthia VanLandingham ENVIRON and The Hamner Institutes
A Regional Model of Lung Metabolism for Improving Species Dependent Descriptions of 1,3-Butadiene and its Metabolites Harvey Clewell, Jerry Campbell, and Cynthia VanLandingham ENVIRON and The Hamner Institutes
PROBLEM SET 7.1 FLUID VOLUMES, GLOMERULAR FILTRATION AND CLEARANCE
 PROBLEM SET 7.1 FLUID VOLUMES, GLOMERULAR FILTRATION AND CLEARANCE ANSWER KEY 1. The time course of decay of plasma [inulin] shown in Fig. 6.1.1 can be simultaneously used to determine the ECF volume and
PROBLEM SET 7.1 FLUID VOLUMES, GLOMERULAR FILTRATION AND CLEARANCE ANSWER KEY 1. The time course of decay of plasma [inulin] shown in Fig. 6.1.1 can be simultaneously used to determine the ECF volume and
BIO 202 HUMAN ANATOMY AND PHYSIOLOGY II BIO 201 Prefix No. Course Title Prerequisite
 Date Revised: 1980 Date Revised: 1985 Date Revised: 1985, 1998, 2002 BIO 202 HUMAN ANATOMY AND PHYSIOLOGY II BIO 201 Prefix No. Course Title Prerequisite Credit Hours: 4 Contact Hours: Lecture 3 Lab 2
Date Revised: 1980 Date Revised: 1985 Date Revised: 1985, 1998, 2002 BIO 202 HUMAN ANATOMY AND PHYSIOLOGY II BIO 201 Prefix No. Course Title Prerequisite Credit Hours: 4 Contact Hours: Lecture 3 Lab 2
IndusChemFate A PBTK-model in MS-Excel applicable for worker and consumer exposure to multiple chemicals User manual
 IndusChemFate A PBTK-model in MS-Excel applicable for worker and consumer exposure to multiple chemicals User manual version 1.5 Nijmegen-NL, August 2nd, 2010 Authors: Frans Jongeneelen Daan Huizer Wil
IndusChemFate A PBTK-model in MS-Excel applicable for worker and consumer exposure to multiple chemicals User manual version 1.5 Nijmegen-NL, August 2nd, 2010 Authors: Frans Jongeneelen Daan Huizer Wil
General renal pathophysiology
 General renal pathophysiology 1 Relationship between plasma solute concentration and its excretion by kidneys General scheme of a feedback regulation (ig 1) 1 Relationship between plasma solute concentration
General renal pathophysiology 1 Relationship between plasma solute concentration and its excretion by kidneys General scheme of a feedback regulation (ig 1) 1 Relationship between plasma solute concentration
1. If the MTC is 100 ng/ml and the MEC is 0.12 ng/ml, which of the following dosing regimen(s) are in the therapeutic window?
 Page 1 PHAR 750: Biopharmaceutics/Pharmacokinetics October 23, 2009 - Form 1 Name: Total 100 points Please choose the BEST answer of those provided. For numerical answers, choose none of the above if your
Page 1 PHAR 750: Biopharmaceutics/Pharmacokinetics October 23, 2009 - Form 1 Name: Total 100 points Please choose the BEST answer of those provided. For numerical answers, choose none of the above if your
Principles of Drug Action. Intro to Pharmacology: Principles of Courework Drug Action Intro to Pharmacology
 Principles of Drug Action Intro to Pharmacology: Principles of Courework 102.3 Drug Action Intro to Pharmacology Directions Read the PPT and complete R.E.A.D. Assignment. There are videos embedded within
Principles of Drug Action Intro to Pharmacology: Principles of Courework 102.3 Drug Action Intro to Pharmacology Directions Read the PPT and complete R.E.A.D. Assignment. There are videos embedded within
DRUG ELIMINATION II BILIARY EXCRETION MAMMARY, SALIVARY AND PULMONARY EXCRETION
 DRUG ELIMINATION II BILIARY EXCRETION MAMMARY, SALIVARY AND PULMONARY EXCRETION ROUTE OF DRUG ADMINISTRATION AND EXTRAHEPATIC DRUG METABOLISM The decline in plasma concentration after drug administration
DRUG ELIMINATION II BILIARY EXCRETION MAMMARY, SALIVARY AND PULMONARY EXCRETION ROUTE OF DRUG ADMINISTRATION AND EXTRAHEPATIC DRUG METABOLISM The decline in plasma concentration after drug administration
BIOPHARMACEUTICS and CLINICAL PHARMACY
 11 years papers covered BIOPHARMACEUTICS and CLINICAL PHARMACY IV B.Pharm II Semester, Andhra University Topics: Absorption Distribution Protein binding Metabolism Excretion Bioavailability Drug Interactions
11 years papers covered BIOPHARMACEUTICS and CLINICAL PHARMACY IV B.Pharm II Semester, Andhra University Topics: Absorption Distribution Protein binding Metabolism Excretion Bioavailability Drug Interactions
Physiologically Based Toxicokinetic Modeling of Inhaled Ethyl tertiary-butyl Ether in Humans
 TOXICOLOGICAL SCIENCES 51, 184 194 (1999) Copyright 1999 by the Society of Toxicology Physiologically Based Toxicokinetic Modeling of Inhaled Ethyl tertiary-butyl Ether in Humans Annsofi Nihlén*,1 and
TOXICOLOGICAL SCIENCES 51, 184 194 (1999) Copyright 1999 by the Society of Toxicology Physiologically Based Toxicokinetic Modeling of Inhaled Ethyl tertiary-butyl Ether in Humans Annsofi Nihlén*,1 and
Principles of Toxicology: The Study of Poisons
 Principles of Toxicology: The Study of Poisons Elizabeth Casarez Department of Pharmacology and Toxicology University it of Arizona The study of the adverse effects of a toxicant on living organisms Adverse
Principles of Toxicology: The Study of Poisons Elizabeth Casarez Department of Pharmacology and Toxicology University it of Arizona The study of the adverse effects of a toxicant on living organisms Adverse
Renal Excretion of Drugs
 Renal Excretion of Drugs 3 1 Objectives : 1 Identify main and minor routes of Excretion including renal elimination and biliary excretion 2 Describe its consequences on duration of drugs. For better understanding:
Renal Excretion of Drugs 3 1 Objectives : 1 Identify main and minor routes of Excretion including renal elimination and biliary excretion 2 Describe its consequences on duration of drugs. For better understanding:
Concurrent Exposure to Ototoxic Chemicals and Noise
 1 Concurrent Exposure to Ototoxic Chemicals and Noise Many common industrial chemicals are ototoxic (poisonous to the ears) and as damaging to employees hearing as the industrial noise to which they are
1 Concurrent Exposure to Ototoxic Chemicals and Noise Many common industrial chemicals are ototoxic (poisonous to the ears) and as damaging to employees hearing as the industrial noise to which they are
PHA Final Exam. Fall On my honor, I have neither given nor received unauthorized aid in doing this assignment.
 PHA 5127 Final Exam Fall 2010 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
PHA 5127 Final Exam Fall 2010 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
ISPUB.COM. Review Of Currently Used Inhalation Anesthetics: Part II. O Wenker SIDE EFFECTS OF INHALED ANESTHETICS CARDIOVASCULAR SYSTEM
 ISPUB.COM The Internet Journal of Anesthesiology Volume 3 Number 3 O Wenker Citation O Wenker.. The Internet Journal of Anesthesiology. 1998 Volume 3 Number 3. Abstract SIDE EFFECTS OF INHALED ANESTHETICS
ISPUB.COM The Internet Journal of Anesthesiology Volume 3 Number 3 O Wenker Citation O Wenker.. The Internet Journal of Anesthesiology. 1998 Volume 3 Number 3. Abstract SIDE EFFECTS OF INHALED ANESTHETICS
Update on PBTK model-based derivation of HBM-1 values for ethylene glycol monoethyl ether (acetate)
 Prof. Wilhelm Huisinga Computational Physiology Group Universität Potsdam Update on PBTK model-based derivation of HBM-1 values for ethylene glycol monoethyl ether (acetate) 49th Sitzung der HBM-Kommission
Prof. Wilhelm Huisinga Computational Physiology Group Universität Potsdam Update on PBTK model-based derivation of HBM-1 values for ethylene glycol monoethyl ether (acetate) 49th Sitzung der HBM-Kommission
PHA First Exam Fall 2003
 PHA 5127 First Exam Fall 2003 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Question/Points 1. /14 pts 2. /6 pts 3. /15 pts 4. /12 pts 5. /20 pts 6. /10pts
PHA 5127 First Exam Fall 2003 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Question/Points 1. /14 pts 2. /6 pts 3. /15 pts 4. /12 pts 5. /20 pts 6. /10pts
Pharmacokinetics Dr. Iman Lec. 3
 Pharmacokinetics r. Iman Lec. 3 Pharmacokinetics A dequate drug doses must be delivered to the target organ to get therapeutic but not toxic levels. So, pharmacokinetic examines the movement of drug over
Pharmacokinetics r. Iman Lec. 3 Pharmacokinetics A dequate drug doses must be delivered to the target organ to get therapeutic but not toxic levels. So, pharmacokinetic examines the movement of drug over
TWA [2] (mg/m3) [5] (ppm) [6]
![TWA [2] (mg/m3) [5] (ppm) [6] TWA [2] (mg/m3) [5] (ppm) [6]](/thumbs/76/74282081.jpg) (2-Butoxyethoxy) ethanol 112-34-5 67.5 10 101.2 15 - DIR 2006/15/CE (2-Methoxyethoxy)ethanol 111-77-3 50.1 10 - - Skin DIR 2006/15/CE Acetic acid 64-19-7 25 10 - - - DIR 91/322/CE Acetone 67-64-1 1210
(2-Butoxyethoxy) ethanol 112-34-5 67.5 10 101.2 15 - DIR 2006/15/CE (2-Methoxyethoxy)ethanol 111-77-3 50.1 10 - - Skin DIR 2006/15/CE Acetic acid 64-19-7 25 10 - - - DIR 91/322/CE Acetone 67-64-1 1210
General Principles of Pharmacology and Toxicology
 General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
Physiological Modeling of Inhalation Kinetics of Octamethylcyclotetrasiloxane in Humans during Rest and Exercise
 TOXICOLOGICAL SCIENCES 72, 3 18 (2003) DOI: 10.1093/toxsci/kfg001 Copyright 2003 by the Society of Toxicology Physiological Modeling of Inhalation Kinetics of Octamethylcyclotetrasiloxane in Humans during
TOXICOLOGICAL SCIENCES 72, 3 18 (2003) DOI: 10.1093/toxsci/kfg001 Copyright 2003 by the Society of Toxicology Physiological Modeling of Inhalation Kinetics of Octamethylcyclotetrasiloxane in Humans during
Chapter 15 Fluid and Acid-Base Balance
 Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
A Speciation Study focused on the Identification of Proximate Toxic Arsenic Metabolites. Kazuo T. Suzuki
 A Speciation Study focused on the Identification of Proximate Toxic Arsenic Metabolites Kazuo T. Suzuki Graduate School of Pharmaceutical Sciences, Chiba University Inage, Chiba 263-8522, JAPAN Arsenic
A Speciation Study focused on the Identification of Proximate Toxic Arsenic Metabolites Kazuo T. Suzuki Graduate School of Pharmaceutical Sciences, Chiba University Inage, Chiba 263-8522, JAPAN Arsenic
Drug elimination and Hepatic clearance Chapter 6
 Drug elimination and Hepatic clearance Chapter 6 DRUG ELIMINATION Drugs are removed from the body by various elimination processes. Drug elimination refers to the irreversible removal of drug from the
Drug elimination and Hepatic clearance Chapter 6 DRUG ELIMINATION Drugs are removed from the body by various elimination processes. Drug elimination refers to the irreversible removal of drug from the
ESSENTIAL TRACE ELEMENTS PREPARED BY B.KIRUTHIGA LECTURER DEPT OF PHARMACEUTICAL CHEMISTRY
 ESSENTIAL TRACE ELEMENTS PREPARED BY B.KIRUTHIGA LECTURER DEPT OF PHARMACEUTICAL CHEMISTRY Back to Basics: Elements Remember that all matter is made of elements An element is a substance in its simplest
ESSENTIAL TRACE ELEMENTS PREPARED BY B.KIRUTHIGA LECTURER DEPT OF PHARMACEUTICAL CHEMISTRY Back to Basics: Elements Remember that all matter is made of elements An element is a substance in its simplest
A. Incorrect! The alveolus is where gas exchange takes place. B. Correct! Surfactant is the lipid-rich material that permits lung inflation.
 Toxicology - Problem Drill 13: Respiratory Toxicology No. 1 of 10 1. The lipid-rich material that decreases surface tension of the alveoli, allowing sacs to inflate properly and remain inflated during
Toxicology - Problem Drill 13: Respiratory Toxicology No. 1 of 10 1. The lipid-rich material that decreases surface tension of the alveoli, allowing sacs to inflate properly and remain inflated during
Development of a Human PBBK Model for Mixtures: Trio Mixture of Mercury, Lead, and Selenium
 IOSR Journal of Environmental Science, Toxicology and Food Technology (IOSR-JESTFT) e-issn: 2319-2402,p- ISSN: 2319-2399.Volume 11, Issue 6 Ver. I (June. 2017), PP 06-13 www.iosrjournals.org Development
IOSR Journal of Environmental Science, Toxicology and Food Technology (IOSR-JESTFT) e-issn: 2319-2402,p- ISSN: 2319-2399.Volume 11, Issue 6 Ver. I (June. 2017), PP 06-13 www.iosrjournals.org Development
Ethylene Oxide
 Ethylene Oxide 75-21-8 Hazard Summary The major use of ethylene oxide is as a chemical intermediate in the manufacture of ethylene glycol. Ethylene oxide is also used as a sterilizing agent for medical
Ethylene Oxide 75-21-8 Hazard Summary The major use of ethylene oxide is as a chemical intermediate in the manufacture of ethylene glycol. Ethylene oxide is also used as a sterilizing agent for medical
Relative Pesticide and Exposure Route Contribution to Aggregate and Cumulative Dose in Young Farmworker Children
 Int. J. Environ. Res. Public Health 2012, 9, 73-96; doi:10.3390/ijerph9010073 OPEN ACCESS Article International Journal of Environmental Research and Public Health ISSN 1660-4601 www.mdpi.com/journal/ijerph
Int. J. Environ. Res. Public Health 2012, 9, 73-96; doi:10.3390/ijerph9010073 OPEN ACCESS Article International Journal of Environmental Research and Public Health ISSN 1660-4601 www.mdpi.com/journal/ijerph
Rational Dose Prediction. Pharmacology. φαρμακον. What does this mean? pharmakon. Medicine Poison Magic Spell
 1 Rational Dose Prediction Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand 2 Pharmacology Pharmacology is derived from a Greek word (pharmakon). The Greeks used
1 Rational Dose Prediction Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand 2 Pharmacology Pharmacology is derived from a Greek word (pharmakon). The Greeks used
PHA Final Exam Fall 2006
 PHA 5127 Final Exam Fall 2006 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
PHA 5127 Final Exam Fall 2006 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Please transfer the answers onto the bubble sheet. The question number refers
Element B1 / 3 Target Organs and Systems
 / 3 Target Organs and Systems Target Organs / Target Systems (Specified toxic substance exerts its effects) Target organs Liver Lungs Kidneys Brain Skin Bladder Eyes Target Organs / Target Systems Substances
/ 3 Target Organs and Systems Target Organs / Target Systems (Specified toxic substance exerts its effects) Target organs Liver Lungs Kidneys Brain Skin Bladder Eyes Target Organs / Target Systems Substances
Relation between airborne arsenic trioxide and
 British Journal of Industrial Medicine 1992;49:387-393 Relation between airborne arsenic trioxide and urinary excretion of inorganic arsenic and its methylated metabolites J A Offergelt, H Roels, J P Buchet,
British Journal of Industrial Medicine 1992;49:387-393 Relation between airborne arsenic trioxide and urinary excretion of inorganic arsenic and its methylated metabolites J A Offergelt, H Roels, J P Buchet,
The Case. The Case. The Case. Objectives. What is a solvent?
 The Case A 24 year old male who recently finished his track career at the University of Oregon decided he wanted to experiment with shoe making to create the optimal surface for the sole of a running shoe.
The Case A 24 year old male who recently finished his track career at the University of Oregon decided he wanted to experiment with shoe making to create the optimal surface for the sole of a running shoe.
Bioavailability Adjustments in Human Health Risk Assessment for Metals-Contaminated Sites
 2015/SOM3/CD/WKSP/009 Bioavailability Adjustments in Human Health Risk Assessment for Metals-Contaminated Sites Submitted by: Australia Workshop on Metals Risk Assessment Cebu, Philippines 28-29 August
2015/SOM3/CD/WKSP/009 Bioavailability Adjustments in Human Health Risk Assessment for Metals-Contaminated Sites Submitted by: Australia Workshop on Metals Risk Assessment Cebu, Philippines 28-29 August
BODY FLUID. Outline. Functions of body fluid Water distribution in the body Maintenance of body fluid. Regulation of fluid homeostasis
 BODY FLUID Nutritional Biochemistry Yue-Hwa Chen Dec 13, 2007 Chen 1 Outline Functions of body fluid Water distribution in the body Maintenance of body fluid Intake vs output Regulation of body fluid Fluid
BODY FLUID Nutritional Biochemistry Yue-Hwa Chen Dec 13, 2007 Chen 1 Outline Functions of body fluid Water distribution in the body Maintenance of body fluid Intake vs output Regulation of body fluid Fluid
APPENDIX K - Exposure Limits For Uranium
 APPENDIX K - Exposure Limits For Uranium K.1 Introduction The terminology used to describe the controls for inhalation exposures of workers to contaminants varies among countries and even between standard-making
APPENDIX K - Exposure Limits For Uranium K.1 Introduction The terminology used to describe the controls for inhalation exposures of workers to contaminants varies among countries and even between standard-making
3. Which of the following would be inconsistent with respiratory alkalosis? A. ph = 7.57 B. PaCO = 30 mm Hg C. ph = 7.63 D.
 Pilbeam: Mechanical Ventilation, 4 th Edition Test Bank Chapter 1: Oxygenation and Acid-Base Evaluation MULTIPLE CHOICE 1. The diffusion of carbon dioxide across the alveolar capillary membrane is. A.
Pilbeam: Mechanical Ventilation, 4 th Edition Test Bank Chapter 1: Oxygenation and Acid-Base Evaluation MULTIPLE CHOICE 1. The diffusion of carbon dioxide across the alveolar capillary membrane is. A.
Concepts in Toxicology Food Toxicology Instructor: Gregory Möller, Ph.D. University of Idaho
 Concepts in Toxicology Food Toxicology Instructor: Gregory Möller, Ph.D. University of Idaho Learning Objectives Define toxicology and toxicity. Discuss different types of toxic responses. Explain how
Concepts in Toxicology Food Toxicology Instructor: Gregory Möller, Ph.D. University of Idaho Learning Objectives Define toxicology and toxicity. Discuss different types of toxic responses. Explain how
Air Toxicology and Epidemiology
 OEA Air - Table of All Acute, Chronic and 8 hour eference Exposure Levels 1 of 8 6/17/2015 3:30 PM ome Air OEA Acute, 8-hour and Chronic eference Exposure Level (EL)s Air Toxicology and Epidemiology All
OEA Air - Table of All Acute, Chronic and 8 hour eference Exposure Levels 1 of 8 6/17/2015 3:30 PM ome Air OEA Acute, 8-hour and Chronic eference Exposure Level (EL)s Air Toxicology and Epidemiology All
ICRP Perspective on Internal Dosimetry OIR and Radiopharmaceuticals
 ICRP Perspective on Internal Dosimetry OIR and Radiopharmaceuticals Dietmar Noßke dnosske@web.de 1 Disclaimer The information and views set out in this presentation are those of the author and do not necessarily
ICRP Perspective on Internal Dosimetry OIR and Radiopharmaceuticals Dietmar Noßke dnosske@web.de 1 Disclaimer The information and views set out in this presentation are those of the author and do not necessarily
Introduction to. Pharmacokinetics. University of Hawai i Hilo Pre-Nursing Program NURS 203 General Pharmacology Danita Narciso Pharm D
 Introduction to 1 Pharmacokinetics University of Hawai i Hilo Pre-Nursing Program NURS 203 General Pharmacology Danita Narciso Pharm D 2 Learning objectives Understand compartment models and how they effects
Introduction to 1 Pharmacokinetics University of Hawai i Hilo Pre-Nursing Program NURS 203 General Pharmacology Danita Narciso Pharm D 2 Learning objectives Understand compartment models and how they effects
Drug Dosing in Renal Insufficiency. Coralie Therese D. Dimacali, MD College of Medicine University of the Philippines Manila
 Drug Dosing in Renal Insufficiency Coralie Therese D. Dimacali, MD College of Medicine University of the Philippines Manila Declaration of Conflict of Interest For today s lecture on Drug Dosing in Renal
Drug Dosing in Renal Insufficiency Coralie Therese D. Dimacali, MD College of Medicine University of the Philippines Manila Declaration of Conflict of Interest For today s lecture on Drug Dosing in Renal
Basic Biopharmaceutics, Pharmacokinetics, and Pharmacodynamics
 Basic Biopharmaceutics, Pharmacokinetics, and Pharmacodynamics Learning Outcomes Define biopharmaceutics Describe 4 processes of pharmacokinetics Describe factors that affect medication absorption Describe
Basic Biopharmaceutics, Pharmacokinetics, and Pharmacodynamics Learning Outcomes Define biopharmaceutics Describe 4 processes of pharmacokinetics Describe factors that affect medication absorption Describe
MATERIAL SAFETY. St. Paul, Minnesota or (651) (24 hours)
 MATERIAL SAFETY DATA SHEET 3M 3M Center St. Paul, Minnesota 55144-1000 1-800-364-3577 or (651) 737-6501 (24 hours) Copyright, 2001, Minnesota Mining and Manufacturing Company. All rights reserved. Copying
MATERIAL SAFETY DATA SHEET 3M 3M Center St. Paul, Minnesota 55144-1000 1-800-364-3577 or (651) 737-6501 (24 hours) Copyright, 2001, Minnesota Mining and Manufacturing Company. All rights reserved. Copying
Biology 105 Midterm Exam 4 Review Sheet
 Biology 105 Midterm Exam 4 Review Sheet The fourth midterm exam will cover the following lecture material (lectures 14, 15, 16, 17, 18, and 19): Blood (from chapter 11 in the textbook), Cardiovascular
Biology 105 Midterm Exam 4 Review Sheet The fourth midterm exam will cover the following lecture material (lectures 14, 15, 16, 17, 18, and 19): Blood (from chapter 11 in the textbook), Cardiovascular
Pharmacokinetics of Rifampicin in African Children Evaluation of the new WHO dosing guidelines
 Pharmacokinetics of Rifampicin in African Children Evaluation of the new WHO dosing guidelines Paolo Denti, Carmen Gonzalez-Martinez, Jana Winckler, Adrie Bekker, Heather Zar, Gerry Davies, Annelies van
Pharmacokinetics of Rifampicin in African Children Evaluation of the new WHO dosing guidelines Paolo Denti, Carmen Gonzalez-Martinez, Jana Winckler, Adrie Bekker, Heather Zar, Gerry Davies, Annelies van
Drug dosing in Extremes of Weight
 Drug dosing in Extremes of Weight The Plump & Heavy versus The Skinny & Light Maria Minerva P. Calimag, MD, MSc, PhD, DPBA, FPSECP PROFESSOR Departments of Pharmacology, Anesthesiology and Clinical Epidemiology
Drug dosing in Extremes of Weight The Plump & Heavy versus The Skinny & Light Maria Minerva P. Calimag, MD, MSc, PhD, DPBA, FPSECP PROFESSOR Departments of Pharmacology, Anesthesiology and Clinical Epidemiology
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Appendix E Choose the sign or symptom that best indicates severe respiratory distress.
 Appendix E-2 1. In Kansas EMT-B may monitor pulse oximetry: a. after they complete the EMT-B course b. when the service purchases the state approved pulse oximeters c. when the service director receives
Appendix E-2 1. In Kansas EMT-B may monitor pulse oximetry: a. after they complete the EMT-B course b. when the service purchases the state approved pulse oximeters c. when the service director receives
Chapter-V Drug use in renal and hepatic disorders. BY Prof. C.Ramasamy, Head, Dept of Pharmacy Practice SRM College of Pharmacy, SRM University
 Chapter-V Drug use in renal and hepatic disorders. BY Prof. C.Ramasamy, Head, Dept of Pharmacy Practice SRM College of Pharmacy, SRM University Estimating renal function An accurate estimation of renal
Chapter-V Drug use in renal and hepatic disorders. BY Prof. C.Ramasamy, Head, Dept of Pharmacy Practice SRM College of Pharmacy, SRM University Estimating renal function An accurate estimation of renal
PHA Spring First Exam. 8 Aminoglycosides (5 points)
 PHA 5128 Spring 2012 First Exam 1 Aminoglycosides (5 points) 2 Aminoglycosides (10 points) 3 Basic Principles (5 points) 4 Basic Principles (5 points) 5 Bioavailability (5 points) 6 Vancomycin (5 points)
PHA 5128 Spring 2012 First Exam 1 Aminoglycosides (5 points) 2 Aminoglycosides (10 points) 3 Basic Principles (5 points) 4 Basic Principles (5 points) 5 Bioavailability (5 points) 6 Vancomycin (5 points)
MOLECULAR SIZE, ELECTRICAL CHARGE, AND SHAPE DETERMINE THE FILTERABILITY OF SOLUTES ACROSS THE GLOMERULAR FILTRATION BARRIER
 MOLECULAR SIZE, ELECTRICAL CHARGE, AND SHAPE DETERMINE THE FILTERABILITY OF SOLUTES ACROSS THE GLOMERULAR FILTRATION BARRIER The glomerular filtration barrier consists of three elements: (1) endothelial
MOLECULAR SIZE, ELECTRICAL CHARGE, AND SHAPE DETERMINE THE FILTERABILITY OF SOLUTES ACROSS THE GLOMERULAR FILTRATION BARRIER The glomerular filtration barrier consists of three elements: (1) endothelial
= (6000 ml air / min * 0.04 ml CO 2 / ml air) / 54 ml CO 2 / dl plasma
 Bio390 thanks to Dr. J.F. Anderson, Dept Zoology Univ. of Florida, Gainesville RENAL PROBLEMS Calculate the rate of pulmonary clearance of CO 2 given the following information. Cardiac Output: 4.8 L 6
Bio390 thanks to Dr. J.F. Anderson, Dept Zoology Univ. of Florida, Gainesville RENAL PROBLEMS Calculate the rate of pulmonary clearance of CO 2 given the following information. Cardiac Output: 4.8 L 6
Indoor emissions. foams) > CH 2 =O; plasticizers, especially dialkyl phthalates. Especially a problem with mobile homes. - Regulations in Sweden
 CHEM/TOX 336 Lecture 9 Indoor Air Emissions Radioisotopes in the Environment Indoor emissions From synthetic materials (carpets, plywood, ureaformaldehyde foams) > CH 2 =O; plasticizers, especially dialkyl
CHEM/TOX 336 Lecture 9 Indoor Air Emissions Radioisotopes in the Environment Indoor emissions From synthetic materials (carpets, plywood, ureaformaldehyde foams) > CH 2 =O; plasticizers, especially dialkyl
Biopharmaceutics. Tips Worth Tweeting. Contributor: Sandra Earle
 Biopharmaceutics C H A P T E R 2 Contributor: Sandra Earle The physiochemical properties of drugs determine how they will move and interact with the body. By understanding a few principles, predictions
Biopharmaceutics C H A P T E R 2 Contributor: Sandra Earle The physiochemical properties of drugs determine how they will move and interact with the body. By understanding a few principles, predictions
UNIVERSITY OF THE WEST INDIES, ST AUGUSTINE
 UNIVERSITY OF THE WEST INDIES, ST AUGUSTINE FACULTY OF MEDICAL SCIENCES SCHOOL OF PHARMACY BACHELOR OF SCIENCE IN PHARMACY DEGREE COURSE SYLLABUS COURSE TITLE: COURSE CODE: BIOPHARMACEUTICS, NEW DRUG DELIVERY
UNIVERSITY OF THE WEST INDIES, ST AUGUSTINE FACULTY OF MEDICAL SCIENCES SCHOOL OF PHARMACY BACHELOR OF SCIENCE IN PHARMACY DEGREE COURSE SYLLABUS COURSE TITLE: COURSE CODE: BIOPHARMACEUTICS, NEW DRUG DELIVERY
Alabama Department of Postsecondary Education
 Date Adopted: July 1, 1980 Date Reviewed: 1985 Date Revised: 1985, 1998, 2002, 2011 Alabama Department of Postsecondary Education Representing Alabama s Public Two-Year College System BIO 202 HUMAN ANATOMY
Date Adopted: July 1, 1980 Date Reviewed: 1985 Date Revised: 1985, 1998, 2002, 2011 Alabama Department of Postsecondary Education Representing Alabama s Public Two-Year College System BIO 202 HUMAN ANATOMY
Analyte Proficiency From All Labs # Analytes: 26 Sample # Statistical Summary # Labs Reporting: 82 Urea Issue Date : 06/30/2016
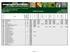 Analyte Proficiency From All Labs # Analytes: 26 Sample # 160511 Statistical Summary # Labs Reporting: 82 Urea Issue Date : 06/30/2016 Analyte Code Analyte # Tests Submitted # Tests in Robust Calculations
Analyte Proficiency From All Labs # Analytes: 26 Sample # 160511 Statistical Summary # Labs Reporting: 82 Urea Issue Date : 06/30/2016 Analyte Code Analyte # Tests Submitted # Tests in Robust Calculations
CASE 27. What is the response of the kidney to metabolic acidosis? What is the response of the kidney to a respiratory alkalosis?
 CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
MAJOR FUNCTIONS OF THE KIDNEY
 MAJOR FUNCTIONS OF THE KIDNEY REGULATION OF BODY FLUID VOLUME REGULATION OF OSMOTIC BALANCE REGULATION OF ELECTROLYTE COMPOSITION REGULATION OF ACID-BASE BALANCE REGULATION OF BLOOD PRESSURE ERYTHROPOIESIS
MAJOR FUNCTIONS OF THE KIDNEY REGULATION OF BODY FLUID VOLUME REGULATION OF OSMOTIC BALANCE REGULATION OF ELECTROLYTE COMPOSITION REGULATION OF ACID-BASE BALANCE REGULATION OF BLOOD PRESSURE ERYTHROPOIESIS
TOXICOLOGY, AND HUMAN HEALTH
 TOXICOLOGY, AND HUMAN HEALTH Toxicity measures how harmful a substance is. It mainly depends on dose the amount of absorbed matter. Three types of toxic entities: Chemical Biological Physical Chemicals
TOXICOLOGY, AND HUMAN HEALTH Toxicity measures how harmful a substance is. It mainly depends on dose the amount of absorbed matter. Three types of toxic entities: Chemical Biological Physical Chemicals
WHY... 8/21/2013 LEARNING OUTCOMES PHARMACOKINETICS I. A Absorption. D Distribution DEFINITION ADME AND THERAPEUIC ACTION
 PHARMACOKINETICS I Absorption & Distribution LEARNING OUTCOMES By the end of the lecture students will be able to.. Dr Ruwan Parakramawansha MBBS, MD, MRCP(UK),MRCPE, DMT(UK) (2013/08/21) Define pharmacokinetics,
PHARMACOKINETICS I Absorption & Distribution LEARNING OUTCOMES By the end of the lecture students will be able to.. Dr Ruwan Parakramawansha MBBS, MD, MRCP(UK),MRCPE, DMT(UK) (2013/08/21) Define pharmacokinetics,
The ADME properties of most drugs strongly depends on the ability of the drug to pass through membranes via simple diffusion.
 1 MEDCHEM 562 Kent Kunze Lecture 1 Physicochemical Properties and Drug Disposition The ADME properties of most drugs strongly depends on the ability of the drug to pass through membranes via simple diffusion.
1 MEDCHEM 562 Kent Kunze Lecture 1 Physicochemical Properties and Drug Disposition The ADME properties of most drugs strongly depends on the ability of the drug to pass through membranes via simple diffusion.
Excretion of Drugs. Prof. Hanan Hagar Pharmacology Unit Medical College
 Excretion of Drugs Prof. Hanan Hagar Pharmacology Unit Medical College Excretion of Drugs By the end of this lecture, students should be able to! Identify main and minor routes of excretion including renal
Excretion of Drugs Prof. Hanan Hagar Pharmacology Unit Medical College Excretion of Drugs By the end of this lecture, students should be able to! Identify main and minor routes of excretion including renal
Lecture 2 Chemical and Biological Agents
 Lecture 2 Chemical and Biological Agents In the Chemical ad Biological section, Routes of exposure we will be covering: Forms of Chemical agents Examples of chemical agents Forms of Biological agents Classifications
Lecture 2 Chemical and Biological Agents In the Chemical ad Biological section, Routes of exposure we will be covering: Forms of Chemical agents Examples of chemical agents Forms of Biological agents Classifications
The Respiratory System
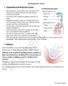 I. Organization of the Respiratory System The Respiratory System The respiratory system allows the circulation of air and the gas exchange between the body and the outside environment. It consists of the
I. Organization of the Respiratory System The Respiratory System The respiratory system allows the circulation of air and the gas exchange between the body and the outside environment. It consists of the
VOLATILE ORGANIC COMPOUNDS
 VOLATILE ORGANIC COMPOUNDS Index Page 1 Principles of volatile organic compounds 2 1.1 Introduction to volatile organic compounds 2 1.2. Classification of volatile organic compounds 2 1.3. Characteristics
VOLATILE ORGANIC COMPOUNDS Index Page 1 Principles of volatile organic compounds 2 1.1 Introduction to volatile organic compounds 2 1.2. Classification of volatile organic compounds 2 1.3. Characteristics
Applicant Name: GD Sigelei Electroinc Tech Co., Ltd B7 Building, No.1 District, Xicheng Science and Technology Park, Hengli Town, Dongguan, China
 Date: Jan.17, 2018 Applicant Name: GD Sigelei Electroinc Tech Co., Ltd Applicant Add.: B7 Building, No.1 District, Xicheng Science and Technology Park, Hengli Town, Dongguan, China Test sample(s) was(were)
Date: Jan.17, 2018 Applicant Name: GD Sigelei Electroinc Tech Co., Ltd Applicant Add.: B7 Building, No.1 District, Xicheng Science and Technology Park, Hengli Town, Dongguan, China Test sample(s) was(were)
ADME Review. Dr. Joe Ritter Associate Professor of Pharmacology
 ADME Review Dr. Joe Ritter Associate Professor of Pharmacology 828-1022 jkritter@vcu.edu What percent of a weak base (pka = 7.5) and weak acid (pka = 3.5) will be respectively ionized in urine of ph 5.5?
ADME Review Dr. Joe Ritter Associate Professor of Pharmacology 828-1022 jkritter@vcu.edu What percent of a weak base (pka = 7.5) and weak acid (pka = 3.5) will be respectively ionized in urine of ph 5.5?
PROVINCE OF BRITISH COLUMBIA ORDER OF THE LIEUTENANT GOVERNOR IN COUNCIL
 PROVINCE OF BRITISH COLUMBIA ORDER OF THE LIEUTENANT GOVERNOR IN COUNCIL Order in Council No. ' 423, Approved and Ordered APR 2 2001.01111mork Lieutenant Governor Executive Council Chambers, Victoria On
PROVINCE OF BRITISH COLUMBIA ORDER OF THE LIEUTENANT GOVERNOR IN COUNCIL Order in Council No. ' 423, Approved and Ordered APR 2 2001.01111mork Lieutenant Governor Executive Council Chambers, Victoria On
3/19/2009. The task of the kidney in acid-base balance Excretion of the daily acid load. Buffering of an acid load. A o B - + H + B - A o +OH - C +
 The task of the kidney in acid-base balance Excretion of the daily acid load Buffering of an acid load Oxidation of amino acids, fats and carbohydrates often lead to acid production. On an average American
The task of the kidney in acid-base balance Excretion of the daily acid load Buffering of an acid load Oxidation of amino acids, fats and carbohydrates often lead to acid production. On an average American
Renal Function. 1. Glomerular filtration 2. Active tubular secretion 3. Passive tubular reabsorption 4. Excretion
 59-291 Section 1, Lecture 5 Drug Excretion -most drugs are excreted in urine either as unchanged or drug metabolites Renal Function 1. Glomerular filtration 2. Active tubular secretion 3. Passive tubular
59-291 Section 1, Lecture 5 Drug Excretion -most drugs are excreted in urine either as unchanged or drug metabolites Renal Function 1. Glomerular filtration 2. Active tubular secretion 3. Passive tubular
Blood Gases, ph, Acid- Base Balance
 Blood Gases, ph, Acid- Base Balance Blood Gases Acid-Base Physiology Clinical Acid-Base Disturbances Blood Gases Respiratory Gas Exchange Chemical Control of Respiration Dyshemoglobins Oxygen Transport
Blood Gases, ph, Acid- Base Balance Blood Gases Acid-Base Physiology Clinical Acid-Base Disturbances Blood Gases Respiratory Gas Exchange Chemical Control of Respiration Dyshemoglobins Oxygen Transport
Physiology questions review
 Physiology questions review 1- The consumption of O2 by the kidney: a- decrease as blood flow increases b- regulated by erythropoiten c- remains constant as blood flow increase d- direct reflects the level
Physiology questions review 1- The consumption of O2 by the kidney: a- decrease as blood flow increases b- regulated by erythropoiten c- remains constant as blood flow increase d- direct reflects the level
Test Report No.T TC Date: JUN 29, 2018 Page 1 of 5
 Test Report No.T31820241114TC Date: JUN 29, 2018 Page 1 of 5 POLYKOTE INK & PAINT (SHENZHEN) LTD. NO. 118, HUANG PU ROAD, SHAJING SUBDISTRICT, BAOAN, SHENZHEN, 518125, CHINA The following samples were
Test Report No.T31820241114TC Date: JUN 29, 2018 Page 1 of 5 POLYKOTE INK & PAINT (SHENZHEN) LTD. NO. 118, HUANG PU ROAD, SHAJING SUBDISTRICT, BAOAN, SHENZHEN, 518125, CHINA The following samples were
OP01 [Mar96] With regards to pethidine s physical properties: A. It has an octanol coefficient of 10 B. It has a pka of 8.4
![OP01 [Mar96] With regards to pethidine s physical properties: A. It has an octanol coefficient of 10 B. It has a pka of 8.4 OP01 [Mar96] With regards to pethidine s physical properties: A. It has an octanol coefficient of 10 B. It has a pka of 8.4](/thumbs/90/104143670.jpg) Opioid MCQ OP01 [Mar96] With regards to pethidine s physical properties: A. It has an octanol coefficient of 10 B. It has a pka of 8.4 OP02 [Mar96] Which factor does NOT predispose to bradycardia with
Opioid MCQ OP01 [Mar96] With regards to pethidine s physical properties: A. It has an octanol coefficient of 10 B. It has a pka of 8.4 OP02 [Mar96] Which factor does NOT predispose to bradycardia with
Table H3-1 Values Used for Daily Intake/Absorbed Dose Inhalation of Particulates
 Table H3-1 Values Used for Daily Intake/Absorbed Dose of Particulates Exposure Point Receptor Age: Particulates See Below Adult Exposure Parameter Parameter Definition Units RME RME Equation/ Route Code
Table H3-1 Values Used for Daily Intake/Absorbed Dose of Particulates Exposure Point Receptor Age: Particulates See Below Adult Exposure Parameter Parameter Definition Units RME RME Equation/ Route Code
Terminology. Terminology. Terminology. Molarity number of moles of solute / Liter of solution. a) Terminology b) Body Fluid Compartments
 Integrative Sciences: Biological Systems A Fall 2011 Body Fluids Compartments, Renal Clearance and Renal Excretion of Drugs Monday, November 21, 2011 Lisa M. Harrison-Bernard, Ph.D. Department of Physiology;
Integrative Sciences: Biological Systems A Fall 2011 Body Fluids Compartments, Renal Clearance and Renal Excretion of Drugs Monday, November 21, 2011 Lisa M. Harrison-Bernard, Ph.D. Department of Physiology;
The Islamic University of Gaza- Civil and Environmental Engineering Department Public Health (EENV-5325) Lecture 9: Occupational health and safety
 The Islamic University of Gaza- Civil and Environmental Engineering Department Public Health (EENV-5325) Lecture 9: Occupational health and safety By Husam Al-Najar Workload: any effect that at least temporarily
The Islamic University of Gaza- Civil and Environmental Engineering Department Public Health (EENV-5325) Lecture 9: Occupational health and safety By Husam Al-Najar Workload: any effect that at least temporarily
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
