Policy QPCMI Page 1 of 40
|
|
|
- Sibyl Fields
- 5 years ago
- Views:
Transcription
1 Policy QPCMI Page 1 of 40 Prepared by: QA Committee Original Date: October 21, 2004 Issued by: Laboratory Manager Revision Date: April 10, 2006 Approved by: Laboratory Director Annual Review Date: April 10, 2006 Table of Contents Blood Culture...3 Enterics...4 Faeces/Rectal Swabs...4 Rectal/Large Bowel (Colon) Biopsies...5 Genital Specimens...6 Cervical Swabs...6 Group B Streptococcus Screen...6 Vaginal Swab for Vaginitis/Vaginosis Screen...6 Vaginal Swab for Culture...7 Urethral Swab...7 Penis Swab...7 Seminal Fluid...8 Endometrial Swabs, Biopsies and Curettings, Placenta Swab/Tissue,...9 Products of Conception, Endometrial/Uterine, Cul de sac/transvaginal,...9 Fallopian Tube, Tubo-Ovarian Swabs or Aspirates...9 Genital Ulcer Swab...9 Intra-Uterine Device...10 Infection Control Screening...11 Methicillin Resistant Staphylococcus aureus (MRSA) Screen...11 Vancomycin Resistant Enterococcus (VRE) Screen...11 Mycology...14 Respiratory Specimens...17 Bronchoalveolar Lavage (BAL), Bronchoscopy Aspirates/Washings Routine...17 Routine Lung Transplant Bronchoscopy Specimen...19 CMV Surveillance Bronchoscopy Specimens...20
2 Policy QPCMI Page 2 of 40 Bronchial Brush Specimens...20 Epiglottal Swabs...21 Open Lung/Transthoracic Needle/Transbronchial Lung Biopsies/Lung Aspirates...21 Mouth Swabs...22 Nasal Swabs...22 Nasopharyngeal Swabs/Auger Suctions for Bordetella pertussis...23 Oral Abscess Swabs...23 Sinus/Antral Specimens...24 Sputum including Endotracheal tube and Tracheotomy Specimens...24 Throat Swabs...26 Gastric Aspirates/Biopsies (for Helicobacter pylori)...27 Gastric Aspirates/Swabs from Neonates or Stillborn...27 Sterile Fluids...28 Cerebrospinal Fluids...28 Other Sterile Fluids Amniotic, Pleural (Thoracentesis/Empyema), Peritoneal (Ascites), Synovial (Joint), Pericardial, Tympanocentesis, Intraocular, Hydrocele Fluids etc...30 Peritoneal Dialysis Effluent...32 Predialysis Fluid...33 Bone Marrow (Aspirates or biopsies)...34 Blood, Platelets, & Other Transfusion Products...35 Urines...36 Wounds/Tissues/Aspirates...39 Duodenal or Small Bowel Aspirate/Swab/Biopsy...39 Record of Edited Revisions...40
3 Policy QPCMI Page 3 of 40 Blood Culture
4 Policy QPCMI Page 4 of 40 Enterics Faeces/Rectal Swabs a) Direct Examination: Not routinely performed. For special request, prepare a Gram stain for faecal leukocytes. MacConkey Agar (MAC) O 2, 35 o C x hours Hektoen Agar (HEK) O 2, 35 o C x hours MacConkey Sorbitol Agar (SMAC) O 2, 35 o C x hours Campylobacter Agar (CAMPY) Campy Jar 42 o C x 48 hours Selenite Broth (SEL) 1 O2, 35 o C x hours If Yersinia is requested or patient is >1 month 12 years old (except for NICU), add: Cefsulodin Irgasan Novobiocin Agar (CIN) O 2, 30 o C x 48 hours If Vibrio is requested, add: Thiosulphate Citrate Bile Salt Sucrose Agar (TCBS) O 2, 35 o C x hours Alkaline Phosphate Broth (APB) 2 O 2, 35 o C x 5-8 hours If Pleiomonas or Aeromonas is requested, add: Blood Agar (BA) O 2, 35 o C x 24 hours If Neisseria gonorrhoeae (GC) is requested (rectal swab only), inoculate only: Martin-Lewis Agar (ML) CO 2, 35 o C x 72 hours If C. difficile toxin assay is requested and appropriate specimen received, forward the specimen to the Virology section for testing. Specimen is also set up for Vancomycin Resistant Enterococcus (VRE) screen: Enterococcus Agar (6? g/ml Vancomycin) O 2 35 o C x 48 hours
5 Policy QPCMI Page 5 of 40 Notes: 1. Subculture Selenite broth following overnight incubation onto HEK. Incubate the sub-cultured HEK at 35?C in O 2 for hours. 2. Subculture APB to TCBS Agar after 5-8 hours incubation. Planter has to notify the technologist on the Enteric bench at the time of processing. Incubate the TCBS Agar at 35 o C in O 2 for hours. Rectal/Large Bowel (Colon) Biopsies a) Direct Examination: Gram stain not indicated. MacConkey Agar (MAC) Hekton Agar (HEK) MacConkey Sorbitol Agar (SMAC) Camyplobacter Agar (CAMPY) Selenite Broth (SEL) O 2 35 o C x hours O 2 35 o C x hours O 2 35 o C x hours Campy Jar 42 o C x 48 hours O 2 35 o C x hours
6 Policy QPCMI Page 6 of 40 Genital Specimens Cervical Swabs a) Direct Examination: Not indicated. Martin-Lewis Agar (ML) CO 2, 35 o C x 72 hours If Group B streptococcus is requested, refer to the Group B streptococcus screen section. Group B Streptococcus Screen a) Direct Examination: Not indicated. Colistin Nalidixic Acid Agar (CNA) Todd Hewitt Broth for Group B Strep. (TH) O 2, 35 o C x 48 hours O 2, 35 o C x 24 hours Vaginal Swab for Vaginitis/Vaginosis Screen a) Direct Examination: i. Wet mount: To be set up immediately. Gently press the swab into a drop of sterile saline on a slide. Place a cover slip on the slide and examine under the microscope using the 40X objective. Examine for the presence of Trichomonas vaginalis.. ii. Gram stain: Examine for the presence of yeast, clue cells and organisms associated with bacterial vaginosis.
7 Policy QPCMI Page 7 of 40 Vaginal Swab for Culture a) Direct Examination: not required Colistin Nalidixic Acid Agar (CNA) Todd Hewitt Broth for Group B Strep(TH) Martin-Lewis Agar (ML) (if requested) MacConkey (MAC) (for <12 years old) CO 2, 35 o C x 48 hours O 2, 35 o C x 24 hours CO 2, 35 o C x 72 hours CO 2, 35 o C x 48 hours Urethral Swab a) Direct Examination: Gram stain - Quantitate the presence of pus cells and intracellular gram negative diplococci. Martin-Lewis Agar (ML) CO 2, 35 o C x 72 hours Penis Swab a) Direct Examination: Gram stain. BA CO 2, 35 o C, x 48 hours MAC O 2, 35 o C, x 48 hours CNA O 2, 35 o C, x 48 hours ML* CO 2, 35 o C, x 72 hours *Occasionally, urethral swabs may be labelled as penile swabs. Neiserria gonorrhoeae culture is set up for this reason. The ML should be inoculated by rotating the swab in a Z streak manner. The inoculum is then streaked by the ISOplater to obtain discrete colonies. Examine ML plates at 48 and 72 hours.
8 Policy QPCMI Page 8 of 40 Seminal Fluid Set up all specimens for culture and sensitivity testing as outlined below. After inoculating the culture media below, forward the remainder of the specimen to the Virology lab for molecular detection of GC and CT. a) Direct Examination: Gram stain. Blood Agar (BA)* CO 2, 35 o C x 48 hours Martin-Lewis Agar (ML) a CO 2, 35 o C x 72 hours MacConkey Agar (MAC)* O 2, 35 o C x 48 hours *Use a 10? l disposable culture loop to inoculate media a Use a swab to inoculate media a Dilute specimen 1:2 using sterile saline before inoculating ML agar
9 Policy QPCMI Page 9 of 40 Endometrial Swabs, Biopsies and Curettings, Placenta Swab/Tissue, Products of Conception, Endometrial/Uterine, Cul de sac/transvaginal, Fallopian Tube, Tubo-Ovarian Swabs or Aspirates a) Preparation of specimen for culture: 1. If tissue is received, aseptically macerate the tissue with the use of a tissue grinder or stomacher. 2. If STAT TB is requested and approved by the Microbiologist, prepare a slide for Acid Fast Bacilli (FLUOROCHROME STAIN for MYCOBACTERIUM). A portion of the specimen should be forwarded to the Public Health Laboratory (PHL) for processing. 3. If TB culture is requested, send half of the specimen to PHL. b) Direct examination: Gram stain. c) Culture: Blood Agar (BA) CO 2, 35 o C x 48 hours Chocolate Agar (CHOC) CO 2, 35 o C x 48 hours Martin-Lewis Agar (ML) CO2, 35 o C x 72 hours MacConkey Agar (MAC) O 2, 35 o C x 48 hours Fastidious Anaerobic Agar (BRUC)* AnO 2, 35 o C x 48 hours Kanamycin-Vancomycin Agar (KV)* AnO 2, 35 o C x 48 hours Fastidious Anaerobic Broth (THIO)* O 2, 35 o C x 48 hours * If tissue/aspirate or anaerobic swab is received, add BRUC, KV and THIO. For tissues and biopsies, freeze remaining tissue in the -70 o C freezer for minimum of 3 months. Genital Ulcer Swab Refer to Ministry of Health Specimen Collection Guide
10 Policy QPCMI Page 10 of 40 Intra-Uterine Device a) Direct Examination: Gram stain of secretions. Examine for the presence of branching gram positive bacilli suggestive of Actinomyces species. Not indicated.
11 Policy QPCMI Page 11 of 40 Infection Control Screening Methicillin Resistant Staphylococcus aureus (MRSA) Screen a) Direct Examination: Not indicated Routine Screening: Mannitol Salt Agar + 10 mg/l Cefoxitin (MSFOX)* O 2, 35 o C x 48 h * If multiple swabs from a single patient are received individually, then process as separate specimens. If multiple swabs from a single patient are received as a "bundle" with a single label and order number, then process all swabs in the bundle on a single MSFOX plate. Outbreak Screening: MRSA Broth O 2, 35 o C 4 hrs or overnight on shaker then subculture from MRSA Broth to: Mannitol Salt Agar + 10 mg/l Cefoxitin (MSFOX) O 2, 35 o C x 48 h Vancomycin Resistant Enterococcus (VRE) Screen a) Direct Examination: Not indicated b) Culture in non-outbreak setting: ¼ menterococcus agar + 6mg/L vancomycin (EV) O 2, 35 o C x 72h
12 Policy QPCMI Page 12 of 40 Note: In the event of an outbreak investigation, the above protocol (b) may not apply. Process specimens as per outbreak protocol (c) below only on prior arrangement by Infection Control or Microbiologist. All swabs will arrive in the laboratory by 9a.m. on Monday or Tuesday only so that they may be processed as a batch. c) Culture in outbreak setting: i) Before 10 am, place each rectal swab into: 3 ml Brain Heart Infusion broth (BHIB) in a 10 ml tube O 2 at 35 o C x 5 hrs minimum on shaker ii) Between 3-4pm on same day, subculture swab to: CNA + 6mg/L vancomycin and Amphotericin B (CNAV6) O 2 at 35 o C x 48h Streak for single colonies Add 30? g vancomycin disc to main inoculum
13 Policy QPCMI Page 13 of 40
14 Policy QPCMI Page 14 of 40 Mycology 1. All specimens, except for skin scrapings, nail and hair (see below) are processed initially in the general planting section of the Microbiology Laboratory. Requests for skin scrapings, nails and hair should be forwarded to the Public Health Lab (PHL) for processing. Fungal smears and inoculated culture plates should be forwarded to the Mycology section for staining, interpretation, incubation and work-up. 2. Fungal smears: i) At least one, and where applicable, two smears from each specimen should be prepared and forwarded to the mycology section. One smear is for fungal staining and the second is held in reserve for special stains (if indicated). ii) In the planting area, the smears should be fixed in 99.9% methanol for 5 minutes before staining. iii) Fungal smears should be stained using Fungi-Fluor? (Refer to Staining Methods). iv) For special requests, send skin, hair and nail specimens to mycology for processing. 3. Culture: i) Refer to Bacteriology manual and/or Table 1 below, for appropriate media to be inoculated depending on specimen type. Between 0.1and 0.5 ml of fluid specimens is recommended for each media. ii) Tissue specimens should be mixed with filtered sterile saline and homogenized in the Stomacher or hand grinder before inoculating the culture media. NB: If a Zygomycete is suspected, DO NOT grind the specimen or put it into the Stomacher. Instead, cut the tissue with a sterile scalpel and use sterile forceps to inoculate the media. iii) Sterile body fluids and urine specimens should be centrifuged at 3500 rpm x 20 min prior to inoculating them. The supernate should be removed leaving sufficient volume to resuspend the sediment and inoculate the appropriate number of culture media. iv) Inoculated culture plates should be held unsealed overnight at room temperature to dry the plates. The following morning, all plates should be placed in a plastic bag and sent to Mycology to be sealed with Parafilm to prevent contamination and incubated at 28 o C. v) For special requests, send skin, hair and nail specimens to mycology for processing.
15 Policy QPCMI Page 15 of 40 Table 1 - PROCESSING AND INCUBATION OF SPECIMENS FOR MYCOLOGY SPECIMEN FS IMA EBM BEAA SAB G MYCOSEL 4 O 2, 28 o C Bone Marrow 5 X X X X 4 weeks Brain Biopsy/Abscess X X X 4 weeks Sterile Fluids: CSF 1 X X 3 weeks Pleural Fluid X X X 3 weeks Synovial Fluid X X X 3 weeks Pericardial/Ascitic Fluid X X X 3 weeks Peritoneal/Dialysis Effluent 2 X X X 3 weeks Aqueous/Vitreous X X X 3 weeks Eye/Corneal Ulcer X X 3 weeks Blood Culture 3, 5 X X X 4 weeks Lung Biopsy X X X X 4 weeks Tissue Biopsy X X X X 4 weeks Lymph Node X X X X 4 weeks Skin Biopsy X X X 3 weeks Clot/Thrombus X X X 3 weeks Bone/Joint X X X 4 weeks Heart Valves X X X 4 weeks Intravascular Grafts X X X 4 weeks Abscess/Pus/Wound 2 X X X X 3 weeks Autopsy X X X 3 weeks Respiratory Secretions: Sputum 2 X X X X 4 weeks Tracheal Aspirate 2 X X X X 4 weeks Bronchial Washing X X X X 4 weeks BAL (Routine) X X X X 4 weeks BAL(Lung Transplant Screen) X X X 2 weeks Sinus/Antral Wash X X X X 2 weeks Nose 2 X X 3 weeks Mastoid X X X 3 weeks Ear X X X 3 weeks Urine 2 X X 3 weeks Hair, Skin & Nail on special X X 3 weeks requests 1. Cryptococcal Antigen in the Planting Area 2. Only if Fungal Culture is specifically requested 3. Collected in "Isolator 10" Tube 4. If dimorphic fungus is rquested, regardless of site, media should be incubated for 6 weeks. 5. For Malassezia request, incubate IMA And IMAO for one week Only
16 Policy QPCMI Page 16 of 40 FS IMA IMAO BEAA EBM SAB G MYCOSEL Fungal Stain Inhibitory Mould Agar with Chloramphenicol Inhibitory Mould Agar with Chloramphenicol, sterile olive oil to be overlaid at Mycology bench. Blood Agar with Gentamicin, Chloramphenicol, Cyclohexamide, Egg Albumin and Avidin Esculin Base Medium S Agar with Gentamicin contains Chloramphenicol and Cyclohexamide
17 Policy QPCMI Page 17 of 40 Respiratory Specimens Bronchoalveolar Lavage (BAL), Bronchoscopy Aspirates/Washings Routine All specimens should be processed for C&S, fungus and TB. Additional processing should be performed only if requested. Bronchial specimens from the same patient should be pooled together and processed as a single sample. 1) If the specimen volume is less than the amount the requested tests, add sterile saline to meet the required volume for the tests requested. (See step 5 for required volumes) Proceed to step 4. 2) Centrifuge the entire specimen at 3500 rpm for 20 minutes. 3) Remove the supernatant in excess of 13 ml to a labelled sterile container. 4) Vortex to mix well. 5) Split the specimen into samples: - 2 ml for C & S and fungus - If PCP is requested, forward 2 ml to Virology for processing - 5 ml for TB (send to PHL) - 2 ml for Virology (if requested) - 1 ml for Legionella (if requested, send to PHL) - 1 ml for Chlamydia pneumoniae (if requested, send to PHL) 6) Direct Examination: Prepare 3 smears for: i) Gram stain Examine for presence of pus cells, epithelial cells and organisms. Report commensal flora, listing specific organism only if predominant. NB: If yeast is predominant organism seen, then report. If yeast is seen mixed with other organisms and is not the predominant organism, then report as commensal flora without specifically commenting on the presence of yeast. ii) Fungifluor stain Forward slide to Mycology Section for staining and interpretation. iii) Extra smear held in Mycology Section for special stains.
18 Policy QPCMI Page 18 of 40 7) Culture: Blood Agar (BA) Haemophilus Isolation Medium (HI) MacConkey Agar (MAC) Inihibitory Mold Agar (IMA) * Esculin Base Medium (EBM)* Blood Egg Albumin Agar (BEAA)* CO 2, CO2, CO 2, O 2, O 2, O 2, 35 o C x 48 hours 35 o C x 48 hours 35 o C x 48 hours 28 o C x 4 weeks 28 o C x 4 weeks 28 o C x 4 weeks If B. cepacia is requested or specimen is from a patient with Cystic Fibrosis, add: OF Base, Colistin, Bacitracin & Lactose Agar (OCBL) O 2, 35 o C x 5 days Keep the BA, HI and MAC plates CO 2, 35 o C x 5 days If Nocardia is requested, add: Sodium Pyruvate Agar (PYRA) O 2, 35 o C x 4 weeks * Forward inoculated fungal media to Mycology Section for incubation and work-up.
19 Policy QPCMI Page 19 of 40 Routine Lung Transplant Bronchoscopy Specimen All specimens should be processed for C&S and fungus. Additional processing should be performed only if requested. Bronchial specimens from the same patient should be pooled together a nd processed as a single sample EXCEPT specimen from Trillium Gift of Life Donor (identified by MRN prefixed with DONOR ) 1) If the specimen volume is less than 10 ml, add sterile saline to a final volume of 10 ml. Proceed to step 4. 2) Centrifuge the entire specimen at 3500 rpm for 20 minutes. 3) Remove the supernatant in excess of 13 ml to a labelled sterile container. 4) Vortex to mix well. 5) Split the specimen into samples: - 2 ml for C & S and fungus - If PCP is requested forward 2 ml to Virology for processing - 5 ml for TB if requested, (send to PHL) - 2 ml for Virology - 1 ml for Legionella (if requested, send to PHL) - 1 ml for Chlamydia pneumoniae (if requested, send to PHL) 6) Direct Examination: Prepare 3 smears for: i) Gram stain Examine for presence of pus cells, epithelial cells and organisms. Report commensal flora, listing specific organism only if predominant. NB: If yeast is predominant organism seen, then report. If yeast is seen mixed with other organisms and is not the predominant organism, then report as commensal flora without specifically commenting on the presence of yeast. ii) Fungifluor stain Forward slide to Mycology Section for staining and interpretation. iii) Extra smear held in Mycology Section for special stains. 7) Culture: Blood Agar (BA) Haemophilus Isolation Medium (HI) CO 2, 35 o C x 48 hours CO 2, 35 o C x 48 hours
20 MacConkey Agar (MAC) Policy QPCMI Page 20 of 40 CO 2, 35 o C x 48 hours Inihibitory Mold Agar (IMA) * Esculin Base Medium (EBM)* Blood Egg Albumin Agar (BEAA)* O 2, O 2, O 2, 28 o C x 2 weeks 28 o C x 2 weeks 28 o C x 2 weeks If B. cepacia is requested or specimen is from a patient with Cystic Fibrosis, add: OF Base, Colistin, Bacitracin & Lactose Agar (OCBL) O 2, 35 o C x 5 days Keep the BA, HI and MAC plates CO 2, 35 o C x 5 days If Nocardia is requested, add: Sodium Pyruvate Agar (PYRA) O 2, 35 o C x 4 weeks * Forward inoculated fungal media to Mycology Section for incubation and work-up. CMV Surveillance Bronchoscopy Specimens Specimens for routine CMV surveillance require CMV culture only. Refer specimens to Virology immediately upon receipt. Bronchial Brush Specimens All brushes received in small cap (bijou) bottles in 1 ml Ringer's lactate solution should be processed "STAT" because colony counts may change if processing is delayed. If the brush is received in <1 ml of fluid, add sufficient Ringer's lactate solution to make up 1 ml. Document in the Test Comment field of the LIS as Brush received in wrong volume of fluid. If a dry brush is received, add 1 ml of Ringer's lactate solution into the vial. Document in the Test Comment field of the LIS as Dry brush received. Mix the vial well by vortexing vigorously. Measure 0.01 ml of the sample (using a calibrated urine loop) and inoculate onto routine media for culture of aerobic bacteria.
21 Policy QPCMI Page 21 of 40 a) Direct Examination: Not indicated. Blood Agar (BA) CO 2, 35 o C x 48 hours Haemophilias Isolation Medium (HI) CO 2, 35 o C x 48 hours MacConkey Agar (MAC) CO 2, 35 o C x 48 hours If B. cepacia is required or specimen is from a patient with Cystic Fibrosis, add: Of Base, Colistin, Bacitracin & Lactose Agar (OCBL) O 2, 35 o C x 5 day Keep the BA, HI and MAC plates CO 2, 35 o C x 5 days Epiglottal Swabs a) Direct examination: Not indicated Medium Blood Agar (BA) Haemophilus Isolation Medium (HI) CO2, 35 o C x 48 hours CO 2, 35 o C x 48 hours Open Lung/Transthoracic Needle/Transbronchial Lung Biopsies/Lung Aspirates a) Direct examination: Using clean, sterile slides make 3 touch preparation smears from a cut surface of the biopsy for: i) Gram stain: Examine for presence of organisms and cells. ii) Fungifluor stain: Forward to Mycology Section for staining and interpretation. iii) Extra smear held in Mycology Section for special stains. Macerate (using a hand grinder) the remaining biopsy tissue using sterile, saline as the diluent.
22 Blood Agar (BA) Chocolate Agar (CHOC) MacConkey Agar (MAC) Fastidious Anaerobe Agar (BRUC) Fastidious Anaerobic Broth (THIO) Policy QPCMI Page 22 of 40 CO 2, 35 o C x 48 hours CO 2, 35 o C x 48 hours O 2, 35 o C x 48 hours AnO 2, 35 o C x 48 hours O 2, 35 o C x 48 hours Inhibitory Mold Agar (IMA) * Esculin Base Medium (EBM) * Blood Egg Albumin Agar (BEAA) * O2, 28 o C x 4 weeks O 2, 28 o C x 4 weeks O 2, 28 o C x 4 weeks If B. cepacia is requested or the specimen is from a patient with Cystic Fibrosis, add: OF Base, Colistin, Bacitracin & Lactose Agar (OCBL) O 2, 35 0 C x 5 days Keep the BA, HI and MAC plates for 5 days If Nocadia is requested, add: Sodium Pyruvate Agar (PYRA) O 2, 35 o C x 4 weeks * Forward inoculated fungal cultures to Mycology for incubation and work-up. Mouth Swabs Applies to both C&S and Fungus test requests: a) Direct Examination: Gram stain Not indicated. Nasal Swabs a) Direct Examination: Not indicated.
23 Policy QPCMI Page 23 of 40 If C&S is requested: Colistin-Nalidixic Agar (CNA) CO 2, 35 o C x 48 hours If N. meniningitidis is requested, add: Martin-Lewis Agar (ML) CO 2, 35 o C x 72 hours If MRSA is requested: Mannitol Salt Agar with Oxacillin (MSAOX) O 2, 35 o C x 48 hours Nasopharyngeal Swabs/Auger Suctions for Bordetella pertussis The specimen should be forwarded to the Public Health Laboratory (PHL) for processing. Oral Abscess Swabs If an anaerobic swab is received and Actinomyces is not requested, process for aerobic culture only. Do not process for anaerobic culture. Document in the TEST Comment field in the LIS as Inappropriate specimen for anaerobic culture, therefore not processed a) Direct Examination: Gram stain Blood Agar (BA) Haemophilus Isolation Medium (HI) CO 2, 35 o C x 48 hours CO 2, 35 o C x 48 hours If Actinomyces requested or suggested on Gram stain, add: Fastidious Anaerobic Agar (BRUC) Kanamycin / Vancomycin Agar (KV) AnO 2, 35 o C x 7 days AnO 2, 35 o C x 7 days
24 Policy QPCMI Page 24 of 40 Sinus/Antral Specimens a) Direct examination: i) Gram stain - Examine for and quantitate the presence of pus cells and organisms. ii) Fungifluor - Forward to Mycology Section for staining and interpretation. Blood Agar (BA) CO 2, 35 o C x 48 hours Haemophilus Isolation Medium (HI) CO 2, 35 o C x 48 hours MacConkey Agar (MAC) CO 2, 35 o C x 48 hours Inhibitory Mold agar (IMA)* O 2, 28 o C x 4 weeks Esculin Base Medium (EBM)* O 2, 28 o C x 4 weeks If anaerobic culture requested, add: Kanamycin Vancomycin Agar (KV) AnO2, 35 o C x 48 hours Fastidious Anaerobic Agar (BRUC) AnO 2, 35 o C x 48 hours Fastidious Anaerobic Broth (THIO) O 2, 35 o C x 48 hours *Forward inoculated fungal media to Mycology section for incubation and work-up. Sputum including Endotracheal tube and Tracheotomy Specimens Notes: Flush small volumes of endotracheal tube (ETT) secretions from suction tubings of neonates with a small volume of sterile trypticase soy broth (TSB) into a sterile container before processing as an unscreened sputum specimen. An ETT tip from an adult is an unacceptable specimen and will not be processed. Notify the ward immediately and issue a report stating ETT tips are not acceptable for culture. For requests for viruses, forward sample to the Virology section for processing.
25 Policy QPCMI Page 25 of 40 Requests for STAT Ziehl-Neelsen acid-fast bacilli (AFB) staining must be approved by a medical microbiologist or infection control practitioner before processing in-house. A portion of the sample should also always be forwarded to the Public Health Laboratory for processing. If mycobacteria (TB) is requested, forward a portion of the specimen to the Public Health Laboratory (PHL) for processing. If Legionella is requested, forward a portion of the specimen to the Public Health Laboratory (PHL) for processing. a) Direct Examination: i) Gram Stain Sputum is always contaminated to some degree with oropharyngeal organisms. Consequently, a screening procedure for routine culture is required to exclude grossly contaminated specimens or saliva. DO NOT screen PMH patients, endotracheal tube (ETT) aspirates, suctioned samples or any specimens requesting Mycobacterium tuberculosis (TB) only or fungus culture only. Screening Procedure Select the most purulent portion of the specimen for Gram staining and culture. Scan the smear under low power (10X magnification) as soon as possible and examine for epithelial cells. ii) Approved requests for STAT acid fast stain: Direct smear from an unconcentrated specimen. iii) Fungus requests: Prepare smear for Fungifluor and forward to Mycology Section for staining and interpretation. Blood Agar (BA) Haemophilus Isolation Medium (HI) MacConkey Agar (MAC) CO 2, CO 2, CO 2, 35 o C x 48 hours 35 o C x 48 hours 35 o C x 48 hours
26 Policy QPCMI Page 26 of 40 If B. cepacia is requested or specimen is from a patient with Cystic Fibrosis, add: OF base, colistin, bacitracin & lactose Agar (OCBL) O 2, 35 o C x 5 days Keep the BA, HI and MAC plates CO 2, 35 o C x 5 days If Nocardia culture is requested, add: Sodium Pyruvate Agar (PYRA) O 2, 35 o C x 4 weeks If fungal culture is requested, add: Inhibitory Mold Agar (IMA)* Esculin Base Medium (EBM)* Blood Egg Albumin Agar (BEAA)* O 2, O 2, O 2, 28 o C x 4 weeks 28 o C x 4 weeks 28 o C x 4 weeks * Forward inoculated fungal media to Mycology section for incubation and work-up. Throat Swabs a) Direct Examination: Not indicated for Group A streptococcus, Neisseria gonorrhoeae or Neisseria meningitidis. If yeast (thrush) is suspected / requested: Gram stain. Examine for presence of pseudohyphae and/or budding yeast. If Vincent s angina is suspected / requested: Gram stain. Examine for presence of spirochetes and/or fusiform bacilli and pus cells. Blood Agar (BA) AnO 2, 35 o C x hours If Neisseria gonorrhoeae requested, add: Martin-Lewis Agar (ML) CO 2, 35 o C x 72 hours If Corynebacterium diphtheriae requested, forward swab to Public Health Laboratory (PHL) for processing. Note: The ML plate is inoculated by rolling the swab in a Z pattern over the medium followed by cross streaking with a sterile loop over the entire plate. Set up anaerobic jar at 1600 hours and at the end of the evening shift.
27 Policy QPCMI Page 27 of 40 Gastric Aspirates/Biopsies (for Helicobacter pylori) a) Direct Examination: i) Use one half of the tissue to prepare a smear for Gram stain and to directly inoculate a urea slant. ii) Incubate the urea slant at 35 o C for 1 4 hours. iii) Macerate the remaining tissue for culture: Blood Agar (BA) Campylobacter Agar (CAMPY) Microaerophilic, 35 C x 7 days Microaerophilic, 35 C x 7 days Gastric Aspirates/Swabs from Neonates or Stillborn a) Direct Examination: Gram Stain Blood Agar (BA) Chocolate Agar (CHOC) MacConkey Agar (MAC) CO 2, 35 o C x 48 hours CO 2, 35 o C x 48 hours O 2, 35 o C x 48 hours
28 Policy QPCMI Page 28 of 40 Sterile Fluids Cerebrospinal Fluids NB: If only VDRL is requested, the specimen will NOT be cultured. All other requests require C&S. These specimens must be processed immediately. 1. Examine the fluid for colour, xanthochromia, turbidity and the presence of visible blood or clots. Record the observations in the LIS under COMMENT in the SOURCE screen. 2. If any of the following is requested: Cryptococcus or cryptococcal antigen, India ink preparation or Fungal culture perform a cryptococcal antigen test on the supernatant or from the original specimen (see Technical Manual) 3. If bacterial antigens are requested, this test is no longer available. If >1 ml of specimen is received: 4. Make a direct cytospin smear using 2-3 drops of unspun CSF. 5. Centrifuge the remaining specimen at 3500 rpm x 20 minutes. 6. Transfer the supernatant to a sterile tube which can be used for biochemical, viral, molecular, Cryptococcal antigen or serological tests if required. Label clearly remaining supernatant and refrigerate. 7. Use the sediment to inoculate the culture media listed below. Label clearly any remaining sediment and refrigerate. 8. If Mycobacteria (TB) is requested, reconstitute sediment with 1 ml of supernatant and label as such. Forward the specimen to the Public Health Laboratory (PHL) for processing. If <1 ml of specimen is received: 4. DO NOT centrifuge the specimen. Consider calling other departments for more specimen. Consult a Microbiologist if the sterility of the extra specimen is questionable.
29 Policy QPCMI Page 29 of If more than one test is ordered, call the physician to prioritize the test ordered. 6. For routine C&S, prepare a Gram smear using a drop of CSF and then directly inoculate the culture media listed below. 7. If Mycobacteria (TB) is requested, forward a portion of the specime n to the Public Health Laboratory (PHL) for processing. 8. If virology is requested, forward a portion of the specimen to the Virology Section for processing. 9. If a swab is received, a. in the Order/Entry Specimen QA Comment field of the LIS as UNST b. in the Result Comment field enter Swab received insufficient specimen. A negative result may not rule out infection. Process the swab as per routine. A. Direct Examination: Gram stain Make 2 smears if the specimen is grossly bloody. B. Culture: Blood Agar CO 2, 35 o C x 4 days Chocolate Agar CO2, 35 o C x 4 days Fastidious Anaerobic Broth O 2, 35 o C x 4 days If fungus or cryptococcus is requested, add: Inhibitory Mould Agar 2 O 2, 28 o C x 3 weeks Esculin Base Medium 2 O 2, 28 o C x 3 weeks 1 Forward inoculated fungal media to Mycology section for incubation and work-up.
30 Policy QPCMI Page 30 of 40 Other Sterile Fluids Amniotic, Pleural (Thoracentesis/Empyema), Peritoneal (Ascites), Synovial (Joint), Pericardial, Tympanocentesis, Intraocular, Hydrocele Fluids etc NB: Chest tube drainage will be treated as a wound specimen (Refer to Wound/Tissues/Aspirates Culture Manual). 1. If a clotted specimen is received, emulsify the coagulated material using a sterile swab or loop. 2. If >1 ml of fluid is received, centrifuge the specimen (3500 rpm x 20 minutes). If <1 ml of specimen is received, do NOT centrifuge the specimen. 3. Transfer the supernatant into a sterile container and refrigerate until the final report is issued. Label the container as supernatant. 4. Use the sediment to prepare the smears and inoculate the culture media. If <1 ml of specimen is received, prepare smears and inoculate culture media directly. 5. If viral culture is requested, forward a portion of the specimen to the Virology Section for processing. 6. If Mycobacteria (TB) is requested, forward a portion of the specimen to the Public Health Laboratory (PHL) for processing. 7. If a swab is received, a. In the Order/Entry Specimen QA Comment field of the LIS as UNST b. In the Result Comment field enter Swab received insufficient specimen. A negative result may not rule out infection. Process the swab as per routine. A. Direct Examination: Gram stain Calcofluor white stain (If fungus is requested). B. Culture: Blood Agar (BA) CO 2, 35 o C x 4 days Chocolate Agar (CHOC) CO 2, 35 o C x 4 days Fastidious Anaerobic Agar (BRUC) AnO 2, 35 o C x 48 hours Fastidious Anaerobic Broth (THIO) O 2, 35 o C x 4 days
31 Policy QPCMI Page 31 of 40 If fungus is requested, add: Inhibitory Mould Agar (IMA) 1 O 2, 28 o C x 3 weeks Esculin Base Medium (EBM) 1 O 2, 28 o C x 3 weeks 1 Forward inoculated fungal media to Mycology section for incubation and work-up.
32 Policy QPCMI Page 32 of 40 Peritoneal Dialysis Effluent NB: No more than one dialysis fluid per patient should be processed every other day. If a bag of cloudy fluid is received after a clean one is processed, culture and sensitivity is always done. A portion of the fluid should be sent to Hematology for cell count if requested. 1) Examine the bags noting the colour and turbidity of the fluid. Record the observations in the LIS under COMMENT in the SOURCE screen. 2) Shake bag well to mix. Working at the sink, disinfect the port with 70% ethanol and let stand for several minutes. Open clamp and run off some of the dialysate. 3) Fill a red top vacutainer tube with approximately 5 ml of dialysate and send to Hematology for cell count. 4) Fill three 50 ml sterile plastic centrifuge tubes with dialysate. Centrifuge two of the tubes (3500 rpm x 20 minutes). Store the third centrifuge tube at 4 o C for at least 7 days. Discard the bag after collecting dialysate. 5) Decant the supernatant leaving approximately 10 ml in each tube. Vortex well to resuspend the sediment and pool the two tubes into one. Prepare Gram smear and inoculate culture media as outlined below. a) Direct Examination: i) If specimen is cloudy: Gram stain ii) If specimen is clear: Gram stain is not needed. b) Culture : If specimen is clear: Bact/Alert bottles* Processed as per blood culture protocol If specimen is cloudy: Blood Agar (BA) CO 2, 35 o C x 4 days Chocolate Agar (CHOC) CO2, 35 o C x 4 days MacConkey Agar (MAC) O 2, 35 o C x 4 days BacT/Alert bottles* Processed as per blood culture protocol *Inoculate both an aerobic and anaerobic bottle with 8 to 9 ml of fluid each.
33 Policy QPCMI Page 33 of 40 Predialysis Fluid 1. Examine the fluid noting the colour and turbidity. Record the observations in the LIS under COMMENT in the SOURCE screen. 2. Centrifuge specimens (3500 rpm x 20 minutes). 3. Decant supernatant leaving approximately 2-3 ml. Vortex well to resuspend the sediment. a) Direct Examination: i) If specimen is cloudy: Gram stain ii) If specimen is clear: No Gram stain is needed. If specimen is clear: Fastidious Anaerobic Broth (THIO) If specimen is cloudy: Blood Agar (BA) MacConkey Agar (MAC) Chocolate Agar (CHOC) Fastidious Anaerobic Broth (THIO) O 2, 35 o C x 5 days CO 2, 35 o C x 4 days CO 2, 35 o C x 4 days CO 2, 35 o C x 4 days O 2, 35 o C x 4 days
34 Policy QPCMI Page 34 of 40 Bone Marrow (Aspirates or biopsies) 1. If <1 ml is received, sterile saline may be added to a final volume of 1 ml so that the media listed below can be inoculated. 2. If a clotted specimen is received, emulsify the coagulated material using a sterile swab or loop. 3. A portion of ALL specimens should be forwarded to the Public Health Laboratory (PHL) for Mycobacteria (TB) culture. a) Direct Examination: Gram stain. Calcofluor white stain. Blood Agar (BA) CO 2, 35 o C x 4 days Chocolate Agar (CHOC) CO2, 35 o C x 4 days Fastidious Anaerobic Agar (BRUC) AnO 2, 35 o C x 48 hours Kanamycin / Vancomycin Agar (KV) AnO 2, 35 o C x 48 hours Fastidous Anaerobic Broth (THIO) O 2, 35 o C x 4 days Inhibitory Mould Agar (IMA) 1 O 2, 28 o C x 4 weeks Esculin Base Medium (EBM) 1 O 2, 28 o C x 4 weeks Blood Egg Albumin Agar (BEAA) 1 O 2, 28 o C x 4 weeks 1 Forward inoculated fungal media to the Mycology Section for incubation and work-up.
35 Policy QPCMI Page 35 of 40 Blood, Platelets, & Other Transfusion Products Both a sample of the transfusion product from the bag and a segment from the tubing should be processed. With aseptic technique, use a needle and syringe to take a sample from each. Enter the order into the LIS twice: one for the tubing and the second for the bag. a) Direct Examination: Not indicated. Blood Agar (BA) CO 2, 35 o C x 4 days Chocolate Agar (CHOC) CO 2, 35 o C x 4 days FAN Aerobic Blood Culture bottle (FO2) in BacT/Alert 35 o C x 5 days
36 Policy QPCMI Page 36 of 40 Urines a) Direct Examination: Gram stain: Not routinely performed. If specifically requested, perform Gram stain directly on unspun specimen. Fungal stain: Not routinely performed. If dimorphic fungus or cryptococcus specifically requested, prepare 2 slides with spun urine (3500 rpm x 15 min.). Send unstained slides to mycology for staining and interpretation. Eosinophil stain: Not routinely performed. If requested, prepare smear by cytospin method and fix in absolute alcohol. Stain slide as per Technical manual. Mix the urine specimen and dip a sterile calibrated ml or 0.01 ml loop vertically into the sample. Streak the loop down the centre of the plate and then crossstreaked at a 90 o angle to the inoculum. (See diagram below). If using the Autostreaker, inoculate the plate with a small streak using a sterile calibrated loop (See diagram below). The amount of urine inoculated is dependent on the type of urine specimen that is processed. (See Table below) i) Autostreaker ii) Manual inoculation
37 Policy QPCMI Page 37 of 40 TABLE 1. Specimen type, inoculum, media and incubation conditions. Voided urine: use ml loop Blood Agar (BA) MacConkey Agar (MAC) In and Out catheter/catheter insertion urine : use ml loop Blood Agar (BA) MacConkey Agar (MAC) Aseptically collected urine: use ml loop Blood Agar (BA) MacConkey Agar (MAC) O 2 O 2 O 2 O 2 O 2 O 2 35 o C x hours 35 o C x hours 35 o C x hours 35 o C x hours 35 o C x hours 35 o C x hours Suprapubic Bladder Aspirate: use ml loop Blood Agar (BA) MacConkey Agar (MAC) Pre-reduced Fastidious Anaerobic Agar (BRUC) O 2 O 2 AnO 2 35 o C x 48 hours 35 o C x 48 hours 35 o C x 48 hours Segmented urines labeled as VB1, VB2, VB3: use both 0.01 ml and ml loops (2 sets) Blood Agar (BA) MacConkey Agar (MAC) Label the plates with the inoculum volume. Prostatic Fluid labeled as EPS sent together with VB1, 2 or 3 or labeled as Seminal Fluid sent together with VB1, 2 or 3: use both 0.01 ml and ml loops (2 sets) Blood Agar (BA) MacConkey Agar (MAC) Label the plates with the inoculum volume. O 2 O 2 O2 O 2 35 o C x hours 35 o C x hours 35 o C x hours 35 o C x hours Urine for routine fungus: use ml loop Blood Agar (BA) First day: Second day: Hi-light the label on the BA plate to note that the plate has to be held for the second day. MacConkey Agar (MAC) O 2 35 o C x hours Room temperature x hours O 2 35 o C x hours
38 Policy QPCMI Page 38 of 40 Urine for Cryptococcus or specific filamentous fungi: centrifuge urine at 3500 rpm, for 15 minuntes EBM IMA Inoculate 1 drop of sediment per plate. Forward plates to Mycology O 2 O 2 28 o C x 3 weeks. 28 o C x 3 weeks. Note: For specimens processed after 1600 hours, place the plates in a separate basket in the incubator. Special Requests : Eosinophil Stain Not routinely performed. If requested, prepare smear by cytospin method and fix in absolute alcohol. Stain slide as per Technical manual. Bacterial Latex Agglutination Not done due to poor sensitivity and specificity. Anaerobes Appropriate specimen is bladder suprapubic aspirate see above Table. Process these specimens immediately. If requested on other urines, consult the Medical Microbiologist. Chlamydia Detection Send the specimen to Virology immediately. Legionella Antigen Detection Send the specimen to the Provincial Health Laboratory. Leptospira Detection Send the specimen to the Provincial Health Laboratory. Cryptococcus/Systemic Fungi See above Table. TB Culture Send the specimen to the Provincial Health Laboratory. Viral Culture Send the specimen to Virology immediately. Parasitology Schistosomiasis Send the specimen to Parasitology immediately.
39 Policy QPCMI Page 39 of 40 Wounds/Tissues/Aspirates Duodenal or Small Bowel Aspirate/Swab/Biopsy a) Direct Examination: Gram stain not performed. i) Duodenal or Small Bowel Aspirates Process Duodenal and Small Bowel aspirates for O&P only. Forward the specimen to the Parasitology section immediately. If a delay is anticipated and the specimen is not received in SAF, transfer the specimen to SAF. DO NOT process these specimens routinely for bacterial culture unless requested. ii) Duodenal or Small Bowel Swab Blood Agar (BA) MacConkey Agar (MAC) Fastidious Anaerobic Agar (BRUC) Kanamycin / Vancomycin Agar (KV) O2, 35 o C x hours O 2, 35 o C x hours AnO 2, 35 o C x 48 hours AnO 2, 35 o C x 48 hours
SPECIMEN PROCESSING CHART Urines
 Policy #MI\SPEC\ Page 1 of 15 Specimen Type Gram Routine Voided urines (midstream, neonatal bagged, indwelling catheter foley catheter, ileal conduit urine) Aseptically collected urine (nephrostomy,bladder/
Policy #MI\SPEC\ Page 1 of 15 Specimen Type Gram Routine Voided urines (midstream, neonatal bagged, indwelling catheter foley catheter, ileal conduit urine) Aseptically collected urine (nephrostomy,bladder/
MICROBIOLOGY SPECIMEN COLLECTION MANUAL
 Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Microbiology Specimen Collection Instructions. Table I Culture and Sensitivities/TB
 Specimen Page 1 of 10 Source / Type of Specimen Culture and Sensitivities () Abscess aspirate, wound aspirate in sterile screw cap or swab in charcoal. Obtain anaerobic swab from lab. Pilonidal and perirectal
Specimen Page 1 of 10 Source / Type of Specimen Culture and Sensitivities () Abscess aspirate, wound aspirate in sterile screw cap or swab in charcoal. Obtain anaerobic swab from lab. Pilonidal and perirectal
Collection (Specimen Source Required on all tests) Sputum: >5 ml required. First morning specimen preferred.
 Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
Policy # MI_GEN Department of Microbiology. Page Quality Manual TABLE OF CONTENTS INTRODUCTION... 3 LOWER GENITAL TRACT... 5
 Quality Manual Version: 1.0 CURRENT 1 of 39 Prepared by QA Committee Issued by: Laboratory Manager Revision Date: 8/15/2017 Approved by Laboratory Director: Annual Review Date: 5/1/2018 Microbiologist-in-Chief
Quality Manual Version: 1.0 CURRENT 1 of 39 Prepared by QA Committee Issued by: Laboratory Manager Revision Date: 8/15/2017 Approved by Laboratory Director: Annual Review Date: 5/1/2018 Microbiologist-in-Chief
Microbiology Collection
 Microbiology Collection Notify the Microbiology Laboratory at 920-738-6317 if any of the following etiological agents are suspected to be present in the specimens sent to the laboratory. Bacillius anthracis
Microbiology Collection Notify the Microbiology Laboratory at 920-738-6317 if any of the following etiological agents are suspected to be present in the specimens sent to the laboratory. Bacillius anthracis
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department Crawley Hospital West Green Drive Crawley RH11 7DH United Kingdom Contact: Clare Reynolds Tel: +44 (0) 1293 600379
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department Crawley Hospital West Green Drive Crawley RH11 7DH United Kingdom Contact: Clare Reynolds Tel: +44 (0) 1293 600379
Effective Date: SPECIMEN COLLECTION FOR CULTURE OF BACTERIAL PATHOGENS QUICK REFERENCE
 1 of 9 Policy #: Subject: 611 (PLH-611-06) Effective Date: 9/30/2004 Reviewed Date: 8/1/2016 SPECIMEN COLLECTION FOR CULTURE OF BACTERIAL PATHOGENS QUICK REFERENCE Approved by: Laboratory Director, Jerry
1 of 9 Policy #: Subject: 611 (PLH-611-06) Effective Date: 9/30/2004 Reviewed Date: 8/1/2016 SPECIMEN COLLECTION FOR CULTURE OF BACTERIAL PATHOGENS QUICK REFERENCE Approved by: Laboratory Director, Jerry
Microbiology & Virology Resource Comprehensive
 Abscess, aspirate, drainage culture Actinomyces culture Aerobic Bacterial culture, stain AFB culture & smear (Acid Fast, TB) AFB Blood culture Anaerobic culture Blood culture Body Fluid culture, stain
Abscess, aspirate, drainage culture Actinomyces culture Aerobic Bacterial culture, stain AFB culture & smear (Acid Fast, TB) AFB Blood culture Anaerobic culture Blood culture Body Fluid culture, stain
CERTIFICATE OF ACCREDITATION
 CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
WVU Medicine Specimens Submitted for Microbiology Testing
 WVU Medicine Specimens Submitted for Microbiology Testing 1. Upper Respiratory a. anterior nares.. page 3 b. nasopharynx.. page 4 c. throat..... page 5 2. Lower Respiratory a. Sputum, tracheal aspirates,
WVU Medicine Specimens Submitted for Microbiology Testing 1. Upper Respiratory a. anterior nares.. page 3 b. nasopharynx.. page 4 c. throat..... page 5 2. Lower Respiratory a. Sputum, tracheal aspirates,
CERTIFICATE OF ACCREDITATION
 CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
MICROBIOLOGICAL TESTING IN PICU
 MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
Babak Valizadeh, DCLS
 Laboratory Diagnosis of Bacterial Infections of the Respiratory Tract Babak Valizadeh, DCLS 1391. 02. 05 2012. 04. 25 Babak_Valizadeh@hotmail.com Biological Safety Cabinet Process specimens in biological
Laboratory Diagnosis of Bacterial Infections of the Respiratory Tract Babak Valizadeh, DCLS 1391. 02. 05 2012. 04. 25 Babak_Valizadeh@hotmail.com Biological Safety Cabinet Process specimens in biological
BASIC PRINCIPLES OF SPECIMEN COLLECTION. Dr. Hala Al -Daghistani
 BASIC PRINCIPLES OF SPECIMEN COLLECTION Dr. Hala Al -Daghistani The goal of the specimen collector must be to maintain the viability of the organisms with minimal contamination Basic principles of specimen
BASIC PRINCIPLES OF SPECIMEN COLLECTION Dr. Hala Al -Daghistani The goal of the specimen collector must be to maintain the viability of the organisms with minimal contamination Basic principles of specimen
Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Special Instructions for Microbiology: Improving the Relevance of Microbiology Specimens. Importance of Proper Specimen Collection and Transport
 Special Instructions for Microbiology: Improving the Relevance of Microbiology Specimens Importance of Proper Specimen Collection and Transport The microbiology laboratory can aid in the diagnosis and
Special Instructions for Microbiology: Improving the Relevance of Microbiology Specimens Importance of Proper Specimen Collection and Transport The microbiology laboratory can aid in the diagnosis and
Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
USER GUIDE FOR SPECIMEN COLLECTION
 DIAGNOSTIC LAB SPECIMEN COLLECTION GUIDE USER GUIDE FOR SPECIMEN COLLECTION ACM GLOBAL LABORATORIES DIAGNOSTIC SERVICES ACM Global Laboratories offers a comprehensive menu of services that support many
DIAGNOSTIC LAB SPECIMEN COLLECTION GUIDE USER GUIDE FOR SPECIMEN COLLECTION ACM GLOBAL LABORATORIES DIAGNOSTIC SERVICES ACM Global Laboratories offers a comprehensive menu of services that support many
The most current laboratory testing information can be obtained at
 About Adventist Lab Partners Reference Laboratory Adventist Lab Partners (ALPs) Reference Laboratory is affiliated with Adventist Health System. Adventist Lab Partners has been a hospital-based laboratory
About Adventist Lab Partners Reference Laboratory Adventist Lab Partners (ALPs) Reference Laboratory is affiliated with Adventist Health System. Adventist Lab Partners has been a hospital-based laboratory
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 Issue No: 001 24 September 2018 Department of Microbiology Longfleet Road Poole Dorset BH15 2JB Contact: Andrew Barber Tel: +44 (0)202 448585 E-Mail: andy.barber@poole.nhs.uk Website: www.poole.nhs.uk
Issue No: 001 24 September 2018 Department of Microbiology Longfleet Road Poole Dorset BH15 2JB Contact: Andrew Barber Tel: +44 (0)202 448585 E-Mail: andy.barber@poole.nhs.uk Website: www.poole.nhs.uk
Lower Respiratory Tract Infection
 Lower Respiratory Tract Infection - Clinical Diseases and etiologic agents -Sample Collection and transport -Direct Smear (interpretation and Report) -Culture methods (interpretation and Report) The culture
Lower Respiratory Tract Infection - Clinical Diseases and etiologic agents -Sample Collection and transport -Direct Smear (interpretation and Report) -Culture methods (interpretation and Report) The culture
Clinical Microbiology and Virology Transport
 Refer to the MLabs Test Catalog for specific specimen type and volume requirements for a particular test or tests. Aerobic Transport (BD ESwab Kit) A sterile package containing a polypropylene screw-cap
Refer to the MLabs Test Catalog for specific specimen type and volume requirements for a particular test or tests. Aerobic Transport (BD ESwab Kit) A sterile package containing a polypropylene screw-cap
Clinical Microbiology CLS 2019 Clinical Microbiology Laboratory Scheme Application Form
 Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
References Required document for Laboratory Accreditation by the College of American Pathologists.
 Subject Culture: Lower Respiratory Tract Index Number Lab-3375 Section Laboratory Subsection Microbiology Category Departmental Contact Sarah Stoner Last Revised 9/11/2017 References Required document
Subject Culture: Lower Respiratory Tract Index Number Lab-3375 Section Laboratory Subsection Microbiology Category Departmental Contact Sarah Stoner Last Revised 9/11/2017 References Required document
Bacteriology, Mycology, and Mycobacteriology Specimen Collection Guidelines
 Bacteriology, Mycology, and Mycobacteriology Specimen Abscess Open Closed Bone marrow Burn Catheter: I.V. Foley Cellulitis Remove surface exudate by wiping with sterile saline or 70% ETOH a. Aspirate if
Bacteriology, Mycology, and Mycobacteriology Specimen Abscess Open Closed Bone marrow Burn Catheter: I.V. Foley Cellulitis Remove surface exudate by wiping with sterile saline or 70% ETOH a. Aspirate if
Rev. 6/18/2018. Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
V. Reporting UNIVERSITY HEALTH NETWORK/MOUNT SINAI HOSPITAL, DEPARTMENT OF MICROBIOLOGY
 Version: 1.0 CURRENT 1 of 26 TABLE OF CONTENTS Urinary Tract Infection Workup I. Introduction... 3 II. Specimen Collection and Transplant... 4 III. Reagents/Materials/Media... 4 IV. Procedure... 4 Asymptomatic
Version: 1.0 CURRENT 1 of 26 TABLE OF CONTENTS Urinary Tract Infection Workup I. Introduction... 3 II. Specimen Collection and Transplant... 4 III. Reagents/Materials/Media... 4 IV. Procedure... 4 Asymptomatic
A. Culturettes and Swabs:
 Northwest Clinical Laboratory 1550 North 115th St. Seattle, WA 98133 Phone: 206.368.1344 Fax: 206.368.1345 NWHMC/UW Medicine Laboratory (Microbiology) Policy MICROBIOLOGY SPECIMEN COLLECTION AND HANDLING
Northwest Clinical Laboratory 1550 North 115th St. Seattle, WA 98133 Phone: 206.368.1344 Fax: 206.368.1345 NWHMC/UW Medicine Laboratory (Microbiology) Policy MICROBIOLOGY SPECIMEN COLLECTION AND HANDLING
Lab 4. Blood Culture (Media) MIC AMAL-NORA-ALJAWHARA 1
 Lab 4. Blood Culture (Media) 2018 320 MIC AMAL-NORA-ALJAWHARA 1 Blood Culture 2018 320 MIC AMAL-NORA-ALJAWHARA 2 What is a blood culture? A blood culture is a laboratory test in which blood is injected
Lab 4. Blood Culture (Media) 2018 320 MIC AMAL-NORA-ALJAWHARA 1 Blood Culture 2018 320 MIC AMAL-NORA-ALJAWHARA 2 What is a blood culture? A blood culture is a laboratory test in which blood is injected
AHS Laboratory Services Central Zone Guide to Lab Services Manual Cultures/Procedures
 AEROBIC CULTURE Routine culture and routine aerobic culture are synonymous terms. Aerobic culture attempts to identify bacteria which grow in the presence of oxygen and are associated with potential infection
AEROBIC CULTURE Routine culture and routine aerobic culture are synonymous terms. Aerobic culture attempts to identify bacteria which grow in the presence of oxygen and are associated with potential infection
TISSUE COLLECTION. SCPA 603- Histopathological Techniques for Routine and Research
 TISSUE COLLECTION SCPA 603- Histopathological Techniques for Routine and Research Somphong Narkpinit, MD. Department of Pathobiology Faculty of Science, Mahidol University somphong.nar@mahidol.ac.th Learning
TISSUE COLLECTION SCPA 603- Histopathological Techniques for Routine and Research Somphong Narkpinit, MD. Department of Pathobiology Faculty of Science, Mahidol University somphong.nar@mahidol.ac.th Learning
Specimen Collection Guide
 Specimen Collection Guide 1 Blood Collection Tubes SODIUM CITRATE TUBE - PALE BLUE TOP 2.7 Coagulation studies: Prothrombin time Factor assays etc. APTT Fibrinogen Thrombophilia tests INR D-Dimer SERUM
Specimen Collection Guide 1 Blood Collection Tubes SODIUM CITRATE TUBE - PALE BLUE TOP 2.7 Coagulation studies: Prothrombin time Factor assays etc. APTT Fibrinogen Thrombophilia tests INR D-Dimer SERUM
MYCOBACTERIOLOGY SERVICE MANUAL
 MYCOBACTERIOLOGY SERVICE MANUAL The Office of Laboratory Services (OLS) provides primary isolation and identification of Mycobacterium species in human diagnostic specimens. Reference specimens of AFB
MYCOBACTERIOLOGY SERVICE MANUAL The Office of Laboratory Services (OLS) provides primary isolation and identification of Mycobacterium species in human diagnostic specimens. Reference specimens of AFB
SHASTA PATHOLOGY ASSOCIATES CYTOLOGY SPECIMEN COLLECTION
 SHASTA PATHOLOGY ASSOCIATES CYTOLOGY SPECIMEN COLLECTION I. PRINCIPLE: Proper collection and handling of all cytology specimens helps ensure the most accurate results possible from the specimen. Cytology
SHASTA PATHOLOGY ASSOCIATES CYTOLOGY SPECIMEN COLLECTION I. PRINCIPLE: Proper collection and handling of all cytology specimens helps ensure the most accurate results possible from the specimen. Cytology
Test Requested Specimen Ordering Recommendations
 Microbiology Essentials Culture and Sensitivity (C&S) Urine C&S Catheter Surgical (excluding kidney aspirates) Voided Requisition requirements o Specific method of collection MUST be indicated o Indicate
Microbiology Essentials Culture and Sensitivity (C&S) Urine C&S Catheter Surgical (excluding kidney aspirates) Voided Requisition requirements o Specific method of collection MUST be indicated o Indicate
Preferred Specimen Vaginal secretions from the lower 1/3 of the vaginal wall collected using a sterile cotton tipped applicator
 B Bacterial Vaginosis Sterile leak-proof screw cap container (SCM) Vaginal secretions from the lower 1/3 of the vaginal wall collected using a sterile cotton tipped applicator Use a sterile cotton tipped
B Bacterial Vaginosis Sterile leak-proof screw cap container (SCM) Vaginal secretions from the lower 1/3 of the vaginal wall collected using a sterile cotton tipped applicator Use a sterile cotton tipped
Culture of Throat, Sputum and Other Respiratory Specimens
 Culture of Throat, Sputum and Other Respiratory Specimens Carol Spiegel, Ph.D., D(ABMM) Professor of Pathology and Laboratory Medicine at the UW School of Medicine and Public Health and Director of Clinical
Culture of Throat, Sputum and Other Respiratory Specimens Carol Spiegel, Ph.D., D(ABMM) Professor of Pathology and Laboratory Medicine at the UW School of Medicine and Public Health and Director of Clinical
ENG MYCO WELL D- ONE REV. 1.UN 29/09/2016 REF. MS01283 REF. MS01321 (COMPLETE KIT)
 ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
Lab-15 Gram Negative Bacteria Neisseria:
 Lab-15 Gram Negative Bacteria Neisseria: د. زينب عادل چابك م. جوان احمد علي الهماوندي The genus Neisseria consists of gram-negative, catalase ve, oxidase +ve, non motile, diplococci. Grows well at aerobic
Lab-15 Gram Negative Bacteria Neisseria: د. زينب عادل چابك م. جوان احمد علي الهماوندي The genus Neisseria consists of gram-negative, catalase ve, oxidase +ve, non motile, diplococci. Grows well at aerobic
320 MBIO Microbial Diagnosis. Aljawharah F. Alabbad Noorah A. Alkubaisi 2017
 320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Blood Culture What is a blood culture? A blood culture is a laboratory test in which blood is injected into bottles with culture
320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Blood Culture What is a blood culture? A blood culture is a laboratory test in which blood is injected into bottles with culture
Providence Medford Medical Center Pathology Department
 Providence Medford Medical Center Pathology Department Anatomic pathology services including histology, cytology and autopsies are offered through Providence Medford Medical Center Pathology Department.
Providence Medford Medical Center Pathology Department Anatomic pathology services including histology, cytology and autopsies are offered through Providence Medford Medical Center Pathology Department.
MICROBIOLOGY SPECIMEN COLLECTION AND TRANSPORT GUIDELINES 9/22/2015 Page 1. Any amount but prefer > 1 ml. Any amount but prefer > 1 ml
 MICROBIOLOGY SPECIMEN COLLECTION AND TRANSPORT GUIDELINES 9/22/2015 Page 1 Abscess Remove surface exudate by wiping with sterile saline or 70% alcohol Syringe with needle removed or Sterile cup or Eswab
MICROBIOLOGY SPECIMEN COLLECTION AND TRANSPORT GUIDELINES 9/22/2015 Page 1 Abscess Remove surface exudate by wiping with sterile saline or 70% alcohol Syringe with needle removed or Sterile cup or Eswab
Collection and Transportation of Clinical Specimens
 Collection and Transportation of Clinical Specimens IDK II Faculty of Nursing Universitas Indonesia PDF Created with deskpdf PDF Writer - Trial :: http://www.docudesk.com The laboratory diagnosis of an
Collection and Transportation of Clinical Specimens IDK II Faculty of Nursing Universitas Indonesia PDF Created with deskpdf PDF Writer - Trial :: http://www.docudesk.com The laboratory diagnosis of an
LABORATORY TEST INFORMATION GUIDE Laboratory Medicine
 Alanine Aminotransferase Specimen: Blood Container: Lithium Heparin Volume: 2.5 4.5 ml Requirements: Invert 8-10 times Stability: 7 days 2-8ºC, Separated: 60 days at -20 ºC 1 of 126 Albumin Specimen: Blood
Alanine Aminotransferase Specimen: Blood Container: Lithium Heparin Volume: 2.5 4.5 ml Requirements: Invert 8-10 times Stability: 7 days 2-8ºC, Separated: 60 days at -20 ºC 1 of 126 Albumin Specimen: Blood
Microbiological Methods V-A- 1 SALMONELLA SPECIES PRESUMPTIVE AND CONFIRMATION TESTS
 Microbiological Methods V-A- 1 PRESUMPTIVE AND CONFIRMATION TESTS PRINCIPLE SCOPE Enrichment and selective procedures are used to provide a reasonably sensitive, definitive and versatile means of qualitatively
Microbiological Methods V-A- 1 PRESUMPTIVE AND CONFIRMATION TESTS PRINCIPLE SCOPE Enrichment and selective procedures are used to provide a reasonably sensitive, definitive and versatile means of qualitatively
Pathology Specimen Handling Requirements
 CONWAY REGIONAL HEALTH SYSTEM CLINICAL LABORATORY Pathology Specimen Handling Requirements POLICY: Tissue or body fluids, etc. removed or collected during any procedure for purposes of Pathologist examination
CONWAY REGIONAL HEALTH SYSTEM CLINICAL LABORATORY Pathology Specimen Handling Requirements POLICY: Tissue or body fluids, etc. removed or collected during any procedure for purposes of Pathologist examination
Storage conditions. roomtemperature or incubator at 36 C. Other: 2-8 C Blood: roomtemperature or incubator at. A.s.a.p. max 24h. A.s.a.p.
 Extract Vademecum Department of Medical Microbiology Status: Code: Version: Approval date: Geldig MMB-PR-04/1 1 Juni 2017 Name request Specimen Material needed Transport Bacteriology Bacterial culture
Extract Vademecum Department of Medical Microbiology Status: Code: Version: Approval date: Geldig MMB-PR-04/1 1 Juni 2017 Name request Specimen Material needed Transport Bacteriology Bacterial culture
Standard Operating Procedure
 Standard Operating Procedure 1. Definitions Section: Laboratory Version No. 1.2 Initials: AD Title: 2.0 Testing Algorithm Revision Date: 01 Sep 2011 1.1 SOP: Standard Operating Procedure 1.2 CRF: Case
Standard Operating Procedure 1. Definitions Section: Laboratory Version No. 1.2 Initials: AD Title: 2.0 Testing Algorithm Revision Date: 01 Sep 2011 1.1 SOP: Standard Operating Procedure 1.2 CRF: Case
35.0. ug/ml Trough. 8.0 Carbamazepine 15.0 ug/ml Digoxin 2.5 ng/ml Gentamicin Peak Random Trough
 The following are a list by department of Critical (Panic) Values: CHEMISTRY TEST LESS THAN GREATER THAN UNITS Glucose 0-7days 40 200 mg/dl Glucose > 7 days 50 400 mg/dl Glucose, CSF 25 mg/dl Sodium 121
The following are a list by department of Critical (Panic) Values: CHEMISTRY TEST LESS THAN GREATER THAN UNITS Glucose 0-7days 40 200 mg/dl Glucose > 7 days 50 400 mg/dl Glucose, CSF 25 mg/dl Sodium 121
DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations
 Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
Contents. 1. General Introduction 3
 Section 1 Medical Laboratory 1. General Introduction 3 Guidelines 3 Code of Ethics for Laboratory Technicians 3 Responsibilities of Medical Technician 3 Instructions for Laboratory Technicians 5 Arrangement
Section 1 Medical Laboratory 1. General Introduction 3 Guidelines 3 Code of Ethics for Laboratory Technicians 3 Responsibilities of Medical Technician 3 Instructions for Laboratory Technicians 5 Arrangement
BACTERIOLOGY PROGRAMME AND PLAN OF TEACHING 3 rd Semester (academic year )
 BACTERIOLOGY PROGRAMME AND PLAN OF TEACHING 3 rd Semester (academic year 2012-2013) 19. 10. 2012. Introduction in microbiology, bacterial taxonomy, general bacterial prop Bacterial structures, biosynthesis
BACTERIOLOGY PROGRAMME AND PLAN OF TEACHING 3 rd Semester (academic year 2012-2013) 19. 10. 2012. Introduction in microbiology, bacterial taxonomy, general bacterial prop Bacterial structures, biosynthesis
Beaumont. Laboratory. Quick Reference Guide: Microbiology
 Beaumont Laboratory Quick Reference Guide: Microbiology Beaumont s Clinical Microbiology and Molecular Pathology Laboratories provide a full spectrum of tests ranging from traditional culture to state-of-theart
Beaumont Laboratory Quick Reference Guide: Microbiology Beaumont s Clinical Microbiology and Molecular Pathology Laboratories provide a full spectrum of tests ranging from traditional culture to state-of-theart
DEPARTMENT OF MICROBIOLOGY
 DEPARTMENT OF MICROBIOLOGY GENERAL INSRUCTIONS FOR COLLECTION OF DIFFERENT MICROBIOLOGY SAMPLES Good quality laboratory services means good quality medicine and better patient care. The Quality of a clinical
DEPARTMENT OF MICROBIOLOGY GENERAL INSRUCTIONS FOR COLLECTION OF DIFFERENT MICROBIOLOGY SAMPLES Good quality laboratory services means good quality medicine and better patient care. The Quality of a clinical
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Issue date: 17 August 2016 Worcestershire Royal Hospital Contact: Peter Jackson Microbiology Laboratory Tel: +44 (0) 1905 760192 Ext 30659
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Issue date: 17 August 2016 Worcestershire Royal Hospital Contact: Peter Jackson Microbiology Laboratory Tel: +44 (0) 1905 760192 Ext 30659
Collection Container Category Source Tests Order Codes
 Nasal Nares culture for MRSA screen ONLY (Outpatient) NSL Room BD BBL Dual Culture Swab (Red Cap) Ear Ear culture EARC Room Eye Eye Culture EYEC Room Cultures Vaginal Genital Culture GEN or GC Room Superficial
Nasal Nares culture for MRSA screen ONLY (Outpatient) NSL Room BD BBL Dual Culture Swab (Red Cap) Ear Ear culture EARC Room Eye Eye Culture EYEC Room Cultures Vaginal Genital Culture GEN or GC Room Superficial
Rev. 7/20/2017. Specimen Collection Transport Replica. Specimen Type. Limit (if applicable) Comments. Courier/ Delayed. Device/ Min Volume.
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE Specimen Collection Transport Replica Guidelines Remove surface exudate by wiping with sterile saline or 70% ETOH Aspirate if possible or pass
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE Specimen Collection Transport Replica Guidelines Remove surface exudate by wiping with sterile saline or 70% ETOH Aspirate if possible or pass
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department Contact: Dr David Ashburn Zone 3 Raigmore Hospital Tel: +44 (0) 1463 704108 Old Perth Road Fax: +44 (0) 1463 705648
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department Contact: Dr David Ashburn Zone 3 Raigmore Hospital Tel: +44 (0) 1463 704108 Old Perth Road Fax: +44 (0) 1463 705648
Case 3: what grew? BA Gram stain morphology Mucoid colonies on BA
 Case 3: what grew? BA Gram stain morphology Mucoid colonies on BA 43 Case 3: probability split! Gram variable, grows on BA, MAC, barely on CHOC Oxidase positive Vitek: probability split, Acinetobacter
Case 3: what grew? BA Gram stain morphology Mucoid colonies on BA 43 Case 3: probability split! Gram variable, grows on BA, MAC, barely on CHOC Oxidase positive Vitek: probability split, Acinetobacter
Clostridium difficile Specimen Collection
 Clostridium difficile Specimen Collection Collect stool specimens into a clean, airtight container with no preservative. All stool specimens should be tested as soon as possible. Ideally, stool specimens
Clostridium difficile Specimen Collection Collect stool specimens into a clean, airtight container with no preservative. All stool specimens should be tested as soon as possible. Ideally, stool specimens
IMMUNOFLUORESCENT STAINING FOR RESPIRATORY VIRUS DETECTION: INFLUENZA A, INFLUENZA B AND RESPIRATORY SYNCYTIAL VIRUS
 IMMUNOFLUORESCENT STAINING FOR RESPIRATORY VIRUS DETECTION: INFLUENZA A, INFLUENZA B AND RESPIRATORY SYNCYTIAL VIRUS A. Introduction Influenza A, Influenza B and RSV are responsible for serious respiratory
IMMUNOFLUORESCENT STAINING FOR RESPIRATORY VIRUS DETECTION: INFLUENZA A, INFLUENZA B AND RESPIRATORY SYNCYTIAL VIRUS A. Introduction Influenza A, Influenza B and RSV are responsible for serious respiratory
Staphylococci. Gram stain: gram positive cocci arranged in clusters.
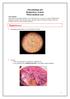 Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Laboratory Contact: Diane Dean Royal Oldham Hospital Tel: +44 (0) 161 656 1369 Rochdale Road E-Mail: diane.dean@pat.nhs.uk OL1
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Laboratory Contact: Diane Dean Royal Oldham Hospital Tel: +44 (0) 161 656 1369 Rochdale Road E-Mail: diane.dean@pat.nhs.uk OL1
Non-Gynecologic Cytology Specimen Handling & Collection Instructions
 Page 1 of 6 Non-Gynecologic Cytology Specimen Handling & Collection Instructions Labelling and Requisition Requirements Specimen Container Patients full name ( printed in same format as patients health
Page 1 of 6 Non-Gynecologic Cytology Specimen Handling & Collection Instructions Labelling and Requisition Requirements Specimen Container Patients full name ( printed in same format as patients health
Strep-a-Test Twister Test
 Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
4. RED AND BLUE TOP COLLECTION SWABS WILL NO LONGER BE SUPPLIED FOR BACTERIAL CULTURE
 Laboratory Testing Update Specimen Transport Changes dated 12-24-14 SEH Laboratory is making significant changes to specimen transports! The Laboratory is introducing new testing platforms along with working
Laboratory Testing Update Specimen Transport Changes dated 12-24-14 SEH Laboratory is making significant changes to specimen transports! The Laboratory is introducing new testing platforms along with working
Objectives 12/4/2013. Disclosure. Culture of Orthopaedic Infections. Microbiology Testing in the Diagnosis of Prosthetic Joint Infections
 Culture of Orthopaedic Infections Microbiology Testing in the Diagnosis of Prosthetic Joint Infections December 9, 2013 Raymond P. Podzorski, Ph.D., D(ABMM) Clinical Microbiologist ProHealth Care Laboratories
Culture of Orthopaedic Infections Microbiology Testing in the Diagnosis of Prosthetic Joint Infections December 9, 2013 Raymond P. Podzorski, Ph.D., D(ABMM) Clinical Microbiologist ProHealth Care Laboratories
2046: Fungal Infection Pre-Infusion Data
 2046: Fungal Infection Pre-Infusion Data Fungal infections are significant opportunistic infections affecting transplant patients. Because these infections are quite serious, it is important to collect
2046: Fungal Infection Pre-Infusion Data Fungal infections are significant opportunistic infections affecting transplant patients. Because these infections are quite serious, it is important to collect
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Issue No: 001 Issue date: 11 ugust 2017 ccredited to Department of Microbiology
Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Issue No: 001 Issue date: 11 ugust 2017 ccredited to Department of Microbiology
Perform Gram stain only. Select Survey D5, Gram Stain
 www.cap.org Bacteriology This flowchart is provided as a guide for ordering the appropriate Bacteriology Surveys for your laboratory s testing menu. In order to meet CLIA requirements for the subspecialty
www.cap.org Bacteriology This flowchart is provided as a guide for ordering the appropriate Bacteriology Surveys for your laboratory s testing menu. In order to meet CLIA requirements for the subspecialty
Medical monitoring: tests available at central hospitals
 medial monitoring: tests available at central hospitals: 1 medical monitoring: tests available at central hospitals Medical monitoring: tests available at central hospitals medial monitoring: tests available
medial monitoring: tests available at central hospitals: 1 medical monitoring: tests available at central hospitals Medical monitoring: tests available at central hospitals medial monitoring: tests available
Media Fill Test Kits. Manufactured by. Making USP <797> compliance easy!
 Media Fill Test Kits Manufactured by Making USP compliance easy! Compliance was never so easy! Microbial Contamination Testing for CSPs Hardy Diagnostics offers all the products you need to easily
Media Fill Test Kits Manufactured by Making USP compliance easy! Compliance was never so easy! Microbial Contamination Testing for CSPs Hardy Diagnostics offers all the products you need to easily
HANDLING GENERAL INFORMATION TEST REQUESTS TYPES OF TESTS TEST REQUEST FORM
 SECTION 3: SAMPLE COLLECTION & HANDLING GENERAL INFORMATION SAMPLE INTRODUCTION COLLECTION & HANDLING GENERAL results. INFORMATION The quality of laboratory test results depends greatly on the quality
SECTION 3: SAMPLE COLLECTION & HANDLING GENERAL INFORMATION SAMPLE INTRODUCTION COLLECTION & HANDLING GENERAL results. INFORMATION The quality of laboratory test results depends greatly on the quality
II- Streptococci. Practical 3. Objective: Required materials: Classification of Streptococci: Streptococci can be classified according to:
 Practical 3 II- Streptococci Objective: 1. Use of blood agar to differentiate between,, and hemolytic streptococci. 2. To know Gram reaction, shape and arrangement of streptococci. 3. To differentiate
Practical 3 II- Streptococci Objective: 1. Use of blood agar to differentiate between,, and hemolytic streptococci. 2. To know Gram reaction, shape and arrangement of streptococci. 3. To differentiate
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 Schedule of Accreditation 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK National Infection Service, Public Health England Colindale 61 Colindale Avenue London NW9 5EQ Contact: Dr Sanjiv
Schedule of Accreditation 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK National Infection Service, Public Health England Colindale 61 Colindale Avenue London NW9 5EQ Contact: Dr Sanjiv
Cytology Specimen Collection Procedures
 BERKSHIRE HEALTH SYSTEM LABORATORIES Berkshire Medical Center Cytology Specimen Collection Procedures Conventional Pap Smears SurePath Pap Smears Sputum Specimens Urine Specimens Breast Discharge Direct
BERKSHIRE HEALTH SYSTEM LABORATORIES Berkshire Medical Center Cytology Specimen Collection Procedures Conventional Pap Smears SurePath Pap Smears Sputum Specimens Urine Specimens Breast Discharge Direct
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 ccredited to Laboratory locations: Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Chester & Wirral Microbiology Service 11 Bassendale
ccredited to Laboratory locations: Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Chester & Wirral Microbiology Service 11 Bassendale
NONGYNECOLOGICAL CYTOLOGY PULMONARY SPECIMENS (Sputum, Post-Bronchoscopy Sputum, Bronchial Brushings, Bronchial Washings, Bronchoalveolar Lavage)
 NONGYNECOLOGICAL CYTOLOGY PULMONARY SPECIMENS (Sputum, Post-Bronchoscopy Sputum, Bronchial Brushings, Bronchial Washings, Bronchoalveolar Lavage) I. Purpose The adequacy of a sputum specimen is determined
NONGYNECOLOGICAL CYTOLOGY PULMONARY SPECIMENS (Sputum, Post-Bronchoscopy Sputum, Bronchial Brushings, Bronchial Washings, Bronchoalveolar Lavage) I. Purpose The adequacy of a sputum specimen is determined
Hospital-acquired Pneumonia
 Hospital-acquired Pneumonia Hospital-acquired pneumonia (HAP) Pneumonia that occurs at least 2 days after hospital admission. The second most common and the leading cause of death due to hospital-acquired
Hospital-acquired Pneumonia Hospital-acquired pneumonia (HAP) Pneumonia that occurs at least 2 days after hospital admission. The second most common and the leading cause of death due to hospital-acquired
General Guidelines for Sample Submissions
 HISTORY General Guidelines for Sample Submissions (See the Client Handbook for specific test requirements.) The history of the animal is very important, including treatments, vaccinations and feeding,
HISTORY General Guidelines for Sample Submissions (See the Client Handbook for specific test requirements.) The history of the animal is very important, including treatments, vaccinations and feeding,
IP Lab Webinar 8/23/2012
 2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Department of Microbiology Princess Alexandra Hospital Hamstel Road Harlow CM20 1QX Contact: D Orriss Tel: +44 01279 827138 E-Mail: Debbie.Orriss@pah.nhs.uk
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Department of Microbiology Princess Alexandra Hospital Hamstel Road Harlow CM20 1QX Contact: D Orriss Tel: +44 01279 827138 E-Mail: Debbie.Orriss@pah.nhs.uk
routine blood cultures. RT Blood Culture Bottles routine blood cultures. RT Blood Culture Bottles of disease
 Updated: 7/20/2015 DISEASE/ Cutaneous NPHL Quick Reference Guide to Specimen Collection of Suspected Agents of Bioterrorism & Emerging Infectious Diseases All potential agents, if not ruled out must be
Updated: 7/20/2015 DISEASE/ Cutaneous NPHL Quick Reference Guide to Specimen Collection of Suspected Agents of Bioterrorism & Emerging Infectious Diseases All potential agents, if not ruled out must be
enter the room. Persons immune from previous varicella infection may enter the room without a mask. Those immune by adenoviruses, influenza viruses.
 All clients admitted to the hospital automatically are considered to be on standard precautions. The diseases listed below require standard precautions plus additional precautions that are noted in the
All clients admitted to the hospital automatically are considered to be on standard precautions. The diseases listed below require standard precautions plus additional precautions that are noted in the
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department West Hertfordshire Hospitals NHS Trust Watford General Hospital Vicarage Road Watford Hertfordshire WD18 0HB Contact:
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department West Hertfordshire Hospitals NHS Trust Watford General Hospital Vicarage Road Watford Hertfordshire WD18 0HB Contact:
Microbiology EQA Product Portfolio
 Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
COLLECTION GUIDELINE FOR LABORATORY DIAGNOSIS OF COMMON VIRAL INFECTIONS
 1 of 5 Policy #: Subject: 609 (PLH-609-07) Effective Date: 9/30/2004 Reviewed Date: 8/1/2016 COLLECTION GUIDELINE FOR LABORATORY DIAGNOSIS OF COMMON VIRAL INFECTIONS Approved by: Laboratory Director, Jerry
1 of 5 Policy #: Subject: 609 (PLH-609-07) Effective Date: 9/30/2004 Reviewed Date: 8/1/2016 COLLECTION GUIDELINE FOR LABORATORY DIAGNOSIS OF COMMON VIRAL INFECTIONS Approved by: Laboratory Director, Jerry
Overview of Wet Preps and Gram stains. Lorna Rabe Central Lab Magee-Womens Research Institute Pittsburgh, Pa
 Overview of Wet Preps and Gram stains Lorna Rabe Central Lab Magee-Womens Research Institute Pittsburgh, Pa Vaginal Flora A secondary objective of the 035 study is to assess the effectiveness of BufferGel
Overview of Wet Preps and Gram stains Lorna Rabe Central Lab Magee-Womens Research Institute Pittsburgh, Pa Vaginal Flora A secondary objective of the 035 study is to assess the effectiveness of BufferGel
Acetylcholine Receptor Modulating Antibody
 Acetylcholine Receptor Modulating Antibody Order Name: ACETY MOD Test Number: 5516500 TEST COMPONENTS REV DATE: 09/25/2012 Acetylcholine Receptor Modulating Antibody SEMI-FC Preferred 1 ml (0.5) Serum
Acetylcholine Receptor Modulating Antibody Order Name: ACETY MOD Test Number: 5516500 TEST COMPONENTS REV DATE: 09/25/2012 Acetylcholine Receptor Modulating Antibody SEMI-FC Preferred 1 ml (0.5) Serum
Results that require action
 Following is a list of Allina Health Laboratory Results That Require Action (formerly Critical Values). Results That Require Action describes a lab test result that may indicate a lifethreatening situation.
Following is a list of Allina Health Laboratory Results That Require Action (formerly Critical Values). Results That Require Action describes a lab test result that may indicate a lifethreatening situation.
Fellowship in Cytopathology Department of Pathology. All India Institute of Medical Sciences (AIIMS) Jodhpur, Rajasthan, India
 Fellowship in Cytopathology Department of Pathology All India Institute of Medical Sciences (AIIMS) Jodhpur, Rajasthan, India Syllabus for Fellowship in Cytopathology: FNAC Direct, Guided, EUS Exfoliative
Fellowship in Cytopathology Department of Pathology All India Institute of Medical Sciences (AIIMS) Jodhpur, Rajasthan, India Syllabus for Fellowship in Cytopathology: FNAC Direct, Guided, EUS Exfoliative
Babak Valizadeh, DCLS
 Ocular Infections- Laboratory Diagnosis of Bacterial Infections of the Eye Babak Valizadeh, DCLS 1392. 02. 01 2012. 04. 21 Babak_Valizadeh@hotmail.com Discriminating Between Indigenous Microbiota and
Ocular Infections- Laboratory Diagnosis of Bacterial Infections of the Eye Babak Valizadeh, DCLS 1392. 02. 01 2012. 04. 21 Babak_Valizadeh@hotmail.com Discriminating Between Indigenous Microbiota and
MYCOBACTERIOLOGY INTRODUCTION RANGE OF LABORATORY SERVICES
 MYCOBACTERIOLOGY INTRODUCTION The Central Tuberculosis Laboratory (CTBL) of the Department of Pathology provides laboratory diagnosis of tuberculosis (TB) and other mycobacterial diseases for all hospitals,
MYCOBACTERIOLOGY INTRODUCTION The Central Tuberculosis Laboratory (CTBL) of the Department of Pathology provides laboratory diagnosis of tuberculosis (TB) and other mycobacterial diseases for all hospitals,
Issued by: LABORATORY MANAGER Original Date: June 18, 2001 Approved by: Laboratory Director RESIDENT OBJECTIVES AT MSH
 Policy # MI/EDU/08/04/v01 Page1 of 13 Section: Subject Title: Resident Objectives at MSH Issued by: LABORATORY MANAGER Original Date: June 18, 2001 Approved by: Laboratory Director Revision Date: SPECIMENS
Policy # MI/EDU/08/04/v01 Page1 of 13 Section: Subject Title: Resident Objectives at MSH Issued by: LABORATORY MANAGER Original Date: June 18, 2001 Approved by: Laboratory Director Revision Date: SPECIMENS
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Management of Needlestick and Mucous Membrane Exposures To Blood/Body Fluids
 Management of Needlestick and Mucous Membrane Exposures To Blood/Body Fluids August 2004 Table Of Contents Protocol - Management of Needlestick and Mucous Membrane Exposures to Blood/Body Fluids Assessment
Management of Needlestick and Mucous Membrane Exposures To Blood/Body Fluids August 2004 Table Of Contents Protocol - Management of Needlestick and Mucous Membrane Exposures to Blood/Body Fluids Assessment
Patient Preparation Unique patient preparation requirements are listed under each test in the Test Directory.
 Specimen Collection Lab results are only as good as the specimen provided. Patient preparation, venipuncture technique, specimen handling, and transportation can all affect the quantity of results. Health
Specimen Collection Lab results are only as good as the specimen provided. Patient preparation, venipuncture technique, specimen handling, and transportation can all affect the quantity of results. Health
