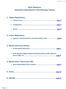
35.0. ug/ml Trough. 8.0 Carbamazepine 15.0 ug/ml Digoxin 2.5 ng/ml Gentamicin Peak Random Trough
|
|
|
- Sheena Jacobs
- 5 years ago
- Views:
Transcription
1 The following are a list by department of Critical (Panic) Values: CHEMISTRY TEST LESS THAN GREATER THAN UNITS Glucose 0-7days mg/dl Glucose > 7 days mg/dl Glucose, CSF 25 mg/dl Sodium mmol/l Potassium 0-31 days mmol/l Potassium >31 days mmol/l CO mmol/l Calcium mg/dl Lactic Acid 2.0 mmol/l Phosphorus 1.6 mg/dl Magnesium mg/dl Neonatal Bilirubin 15.0 mg/dl Total Protein, CSF 100 mg/dl Troponin I 0.06 ng/ml TDMs Acetaminophen 4hr post dose 150 ug/ml Amikacin Peak 35.0 ug/ml Trough 8.0 Carbamazepine 15.0 ug/ml Digoxin 2.5 ng/ml Gentamicin Peak Random Trough ug/ml Lithium 2.0 mmol/l Phenobarbital 40.0 ug/ml Phenytoin 20.0 ug/ml Salicylate 30.0 mg/dl Theophylline 20.0 ug/ml Tobramycin Peak 12.0 ug/ml Random 12.0 ug/ml Trough 2.0 ug/ml Valproic Acid ug/ml Vancomycin Peak Random Trough ug/ml Page 1 of 6
2 RESPIRATORY THERAPY TEST LESS THAN GREATER THAN UNITS ph PCO2 Arterial mmhg Capillary PO days mmhg PO2 >31 days 50 mmhg Carboxyhemoglobin COHB 20.0 % Methemoglobin Met Hb 10.0 % PATHOLOGY 1. Surgical Frozen Sections, Intra-operative Cytologies, and Gross Examination reports may be given directly to the physician by the pathologist, in person or by telephone. Hand written reports may also be sent directly to the operating room. Turnaround time is recorded on the Surgical Frozen Section log. 2. Results of surgical pathology and/or cytology cases that are deemed significant/unexpected findings at the time of the case review are communicated directly to the submitting provider or nurse involved in the care of the patient and recorded in the pathology report. Page 2 of 6
3 HEMATOLOGY/ COAGULATION TEST LESS THAN GREATER THAN UNITS WBC 0-4 yr x 10 3 WBC >4 yr x 10 3 Hemoglobin 0-14 days g/dl Hemoglobin 15 days- 3 mo g/dl Hemoglobin > 3 mo g/dl Platelet Count x 10 3 Blasts ANY Fibrinogen 100 mg/dl APTT 100 seconds. Protime -INR 5 INR, finger stick 5 Page 3 of 6
4 MICROBIOLOGY TEST/SITE Blood CSF Stool or other sterile site Sputum or Nasopharyngeal Culture - All sources Synovial Fluid RESULT Gram stain of positive culture MRSA positive by PCR *Follow-up call when the following organisms are isolated: Campylobacter jejuni Salmonella Shigella E.coli 0157 or Shiga toxin positive Neisseria meningitidis Haemophilus influenza Streptococcus pneumoniae Positive smear or culture Any Positive Biofire Meningitis result Salmonella (any species) Escherichia coli 0157 or Shiga toxin positive Inpatients only: Clostridium difficile Bordetella pertussis Inpatient only: Positive respiratory panel result Positive Influenza (Flu) Mycobacterium (any species) Vancomycin resistant Staph species Bioterrorism organisms Corynebacterium diphtheria Shigella (any species) Inpatients only: Vancomycin resistant Enterococcus (VRE) Methicillin resistant Staph aureus (MRSA) Carbapenemase resistant Enterbacteriaceae (CRE) Positive Stain/Culture Page 4 of 6
5 BLOOD BANK TEST RESULT Crossmatch Unable to obtain compatible blood Incompatible crossmatch noted after emergency issue of Uncrossmatched Unit Transfusion Reaction Indication of hemolytic transfusion reaction/ TRALI, Bacterial contamination or other serious adverse events related to the transfusion, the interpretation shall be reported to the patient s physician immediately Culture on issued apheresis platelet or Positive blood component Fetalscreen Positive *Follow up call after the Kleihauer Betke results are received. Recalls transfused units due to All presence of pathogen Specimen discrepancy Wrong blood in tube identified with comparison to historical type Page 5 of 6
6 RETIREMENT DATE: DISTRIBUTION: BMC General Lab Manual JMC Laboratory Administration Manual and Online Policy Manager COMPILED BY: Natasha Cortez-Caja, System Director of Laboratory Services APPROVED BY: Ryan Livengood, MD, Medical Director/Date REVIEWED BY: DATE: REVISION (ITEM): PAGE DATE REVISED BY: This list replaces the lists found in BMC GL IV.02 and JMC LAM 010. In addition, this list supersedes any and all individual department lists Microbiology: Added Blood Streptococcus pneumoniae. Added Synovial Fluid Positive Stain/Culture Page 4 TW Corrected units for Salicylate Page 1 3/20/17 GC Page 6 of 6
Results that require action
 Following is a list of Allina Health Laboratory Results That Require Action (formerly Critical Values). Results That Require Action describes a lab test result that may indicate a lifethreatening situation.
Following is a list of Allina Health Laboratory Results That Require Action (formerly Critical Values). Results That Require Action describes a lab test result that may indicate a lifethreatening situation.
PEACEHEALTH LABORATORIES
 360-414-2306 www.peacehealthlabs.org Critical Values Call List - Longview Critical values are reported per the criteria published below. Laboratory results meeting these criteria indicate potential life-threatening
360-414-2306 www.peacehealthlabs.org Critical Values Call List - Longview Critical values are reported per the criteria published below. Laboratory results meeting these criteria indicate potential life-threatening
Comments / Frequency (if not each time)
 s CHEMISTRY Acetaminophen High: > 250 ug/ml Bicarbonate Low: < 10 meq/l High: >40 meq/l Bilirubin (< 5 years old) High: > 15 mg/dl Calcium, total (serum) Low: < 6.0 mg/dl High: > 13.0 mg/dl > 15 Absurd
s CHEMISTRY Acetaminophen High: > 250 ug/ml Bicarbonate Low: < 10 meq/l High: >40 meq/l Bilirubin (< 5 years old) High: > 15 mg/dl Calcium, total (serum) Low: < 6.0 mg/dl High: > 13.0 mg/dl > 15 Absurd
PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015
 PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015 Section: Chemistry For inpatients, once 3 consecutive critical values for the same analyte (test result) have
PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015 Section: Chemistry For inpatients, once 3 consecutive critical values for the same analyte (test result) have
OHIOHEALTH Laboratory Services INPATIENT, ED AND OTHER HOSPITAL BASED PATIENT CRITICAL VALUE NOTIFICATION LIST
 11/5/2018 OHIOHEALTH Laboratory Services INPATIENT, ED AND OTHER HOSPITAL BASED PATIENT CRITICAL VALUE NOTIFICATION LIST Test Critical Low Critical High Alerting CHEMISTRY Amylase > 400 U/L Emergency Dept
11/5/2018 OHIOHEALTH Laboratory Services INPATIENT, ED AND OTHER HOSPITAL BASED PATIENT CRITICAL VALUE NOTIFICATION LIST Test Critical Low Critical High Alerting CHEMISTRY Amylase > 400 U/L Emergency Dept
MidMichigan Health LABORATORY POLICY Title: Effecti ve te: Key Words: places: Category: Applicability: reference laboratory
 Page 1 of 5 PURPOSE This policy describes the responsibilities for determining which tests performed and or provided by the MidMichigan Health Laboratories fulfill the criteria for critical values / critical
Page 1 of 5 PURPOSE This policy describes the responsibilities for determining which tests performed and or provided by the MidMichigan Health Laboratories fulfill the criteria for critical values / critical
Test Critical Low Critical High Alerting Category
 Test Critical Low Critical High Alerting CHEMISTRY Amylase > 400 U/L Emergency Dept only Amylase, Pancreatic >300 U/L Emergency Dept only Bicarb (Carbon Dioxide) 40 mmol/l Bilirubin, Total
Test Critical Low Critical High Alerting CHEMISTRY Amylase > 400 U/L Emergency Dept only Amylase, Pancreatic >300 U/L Emergency Dept only Bicarb (Carbon Dioxide) 40 mmol/l Bilirubin, Total
JOB AID CRITICAL VALUES AND TESTS W/O MICRO CRITICAL TESTS. Always call results for the following test(s):
 Facilities: NoCo Laboratories JOB AID CRITICAL VALUES AND TESTS W/O MICRO CRITICAL TESTS Always call results for the following test(s): CONSTITUENT Ethylene Glycol (Performed at CU) Time Interval (from
Facilities: NoCo Laboratories JOB AID CRITICAL VALUES AND TESTS W/O MICRO CRITICAL TESTS Always call results for the following test(s): CONSTITUENT Ethylene Glycol (Performed at CU) Time Interval (from
Laboratory Values, Critical. ThedaCare. Date Last Reviewed: 4/5/2018 Reviewing Body(s): Quality; Laboratory Leadership
 Laboratory Values, Critical ThedaCare Policy & Procedure Policy Title: Laboratory Values, Critical Policy Number: 694 Location(s): All ThedaCare Department(s): Laboratory Date Last Reviewed: 4/5/2018 Reviewing
Laboratory Values, Critical ThedaCare Policy & Procedure Policy Title: Laboratory Values, Critical Policy Number: 694 Location(s): All ThedaCare Department(s): Laboratory Date Last Reviewed: 4/5/2018 Reviewing
If a critical result is obtained for a clinic patient after the clinic has closed, we will notify the individual on-call for the clinic.
 UCSF Clinical Laboratories Critical ('Panic') and Subcritical Values Policy (Last updated 11/3/17): In collaboration with the clinical services, the following results have been determined to represent
UCSF Clinical Laboratories Critical ('Panic') and Subcritical Values Policy (Last updated 11/3/17): In collaboration with the clinical services, the following results have been determined to represent
CRITICAL VALUES, SIGNIFICANT FINDINGS LIST and STAT RESULTS
 Page 1 of 6 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: s are defined as an analytic result that suggests a clinical condition that may be lifethreatening and may require immediate
Page 1 of 6 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: s are defined as an analytic result that suggests a clinical condition that may be lifethreatening and may require immediate
CRITICAL VALUES, CRITICAL TEST RESULTS AND SIGNIFICANT FINDINGS LIST
 Page 1 of 7 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: Critical Values are defined as an analytic result that suggests a clinical condition that may be life- threatening and
Page 1 of 7 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: Critical Values are defined as an analytic result that suggests a clinical condition that may be life- threatening and
Therapeutic and Critical Drug Levels
 6 Results Reporting As a convenience for our clients and their patients, result reports are printed daily at your facility by means of remote printer/fax machine via internet platforms (4Medica, EMR, ecare/mdoffice)
6 Results Reporting As a convenience for our clients and their patients, result reports are printed daily at your facility by means of remote printer/fax machine via internet platforms (4Medica, EMR, ecare/mdoffice)
Laboratory Test Result Reporting and Inquiry Procedures
 Jump to: Pathology Information System Critical Value Reporting Policy Microbiology Result Reporting Procedures Antimicrobial Susceptibility Result Reporting Surgical Pathology Result Reporting Cytopathology
Jump to: Pathology Information System Critical Value Reporting Policy Microbiology Result Reporting Procedures Antimicrobial Susceptibility Result Reporting Surgical Pathology Result Reporting Cytopathology
Epic Labs Orderable As STAT PRIORITY As of 06/22/2016
 ABG+HB(CORDARTERIAL) - BABY A ABG+HB(CORD ARTERIAL)- BABY B ABG+HB(CORD ARTERIAL)- BABY C ACETAMINOPHEN LEVEL ALANINE AMINOTRANSFERASE (ALT) ALBUMIN, FLUID ALBUMIN, PLEURAL FLUID ALBUMIN, SYNOVIAL FLUID
ABG+HB(CORDARTERIAL) - BABY A ABG+HB(CORD ARTERIAL)- BABY B ABG+HB(CORD ARTERIAL)- BABY C ACETAMINOPHEN LEVEL ALANINE AMINOTRANSFERASE (ALT) ALBUMIN, FLUID ALBUMIN, PLEURAL FLUID ALBUMIN, SYNOVIAL FLUID
RIDGEVIEW MEDICAL CENTER AND CLINICS
 RIDGEVIEW MEDICAL CENTER AND CLINICS #5575 SUBJECT: REPORTING OF CRITICAL TEST RESULTS AND CRITICAL VALUES ORIGINATING DEPT: Laboratory DISTRIBUTION DEPTS: 7650, 8000, 7575, 7240, 7505, 7600 ACCREDITATION/REGULATORY
RIDGEVIEW MEDICAL CENTER AND CLINICS #5575 SUBJECT: REPORTING OF CRITICAL TEST RESULTS AND CRITICAL VALUES ORIGINATING DEPT: Laboratory DISTRIBUTION DEPTS: 7650, 8000, 7575, 7240, 7505, 7600 ACCREDITATION/REGULATORY
1. HISTORY. This issue publishes a revision of this publication.
 *MEDDAC MEMO 40-138 DEPARTMENT OF THE ARMY US ARMY MEDICAL DEPARTMENT ACTIVITY Fort Huachuca, Arizona 85613-7079 MEDDAC MEMORANDUM 16 August 2006 40-138 Medical Services FOLLOW-UP OF ABNORMAL TEST RESULTS
*MEDDAC MEMO 40-138 DEPARTMENT OF THE ARMY US ARMY MEDICAL DEPARTMENT ACTIVITY Fort Huachuca, Arizona 85613-7079 MEDDAC MEMORANDUM 16 August 2006 40-138 Medical Services FOLLOW-UP OF ABNORMAL TEST RESULTS
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
ALERT VALUE PROTOCOL INITIAL EVALUATION: 1. Determine the category of the alert value by reviewing the Alert Value List.
 Alert Value Protocol: Page 1 of 14 ALERT VALUE PROTOCOL PURPOSE: Alert Values are categorized into three levels of notification requirements, Red, Orange, and Yellow, with respective Communication Time
Alert Value Protocol: Page 1 of 14 ALERT VALUE PROTOCOL PURPOSE: Alert Values are categorized into three levels of notification requirements, Red, Orange, and Yellow, with respective Communication Time
Inspector's Accreditation Unit Activity Menu
 01/12/20XX 15:58:57 Laboratory Accreditation Program Page 1 of 9 CHEMISTRY 1501 ALT, serum/plasma 1502 Albumin, serum/plasma 1504 Alkaline phosphatase, serum/plasma 1506 Amylase, serum/plasma 1508 Bilirubin,
01/12/20XX 15:58:57 Laboratory Accreditation Program Page 1 of 9 CHEMISTRY 1501 ALT, serum/plasma 1502 Albumin, serum/plasma 1504 Alkaline phosphatase, serum/plasma 1506 Amylase, serum/plasma 1508 Bilirubin,
1. Purpose 1.1. To define testing locations, schedule and order priority for each test performed in the core laboratory.
 Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
GENERAL INFORMATION CLINICAL LABORATORY PHONE DIRECTORY
 GENERAL INFORMATION CLINICAL LABORATORY PHONE DIRECTORY SECTION PHONE NUMBER Clinical Pathologist 431-5888 Laboratory Main Laboratory Administrative Director Janis Nall Accessioning/Client Services Section
GENERAL INFORMATION CLINICAL LABORATORY PHONE DIRECTORY SECTION PHONE NUMBER Clinical Pathologist 431-5888 Laboratory Main Laboratory Administrative Director Janis Nall Accessioning/Client Services Section
GRADING CRITERIA for CMS Regulated Analytes
 CLIA '88 AND GRADING The Clinical Laboratory Improvement Amendments of 1988 (CLIA '88) were established by the federal government (CMS) to regulate clinical laboratories and proficiency test providers
CLIA '88 AND GRADING The Clinical Laboratory Improvement Amendments of 1988 (CLIA '88) were established by the federal government (CMS) to regulate clinical laboratories and proficiency test providers
Collection (Specimen Source Required on all tests) Sputum: >5 ml required. First morning specimen preferred.
 Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
NORTH SHORE - LONG ISLAND JEWISH HEALTH SYSTEM LONG ISLAND JEWISH MEDICAL CENTER DEPARTMENT OF LABORATORY MEDICINE REFERENCE RANGES
 S A2 COLUMN 0D 2.0-3.5 2.0-3.5 % ACETAMINOPHEN 0D 5-25 5-25 > 150 ug/ml > 50 ug/ml 12 hour post ingestion > 100 ug/ml 8 hour post ingestion > 200 ug/ml 4 hour post ingestion ACTIVATED PROTEIN C RESIS 0D
S A2 COLUMN 0D 2.0-3.5 2.0-3.5 % ACETAMINOPHEN 0D 5-25 5-25 > 150 ug/ml > 50 ug/ml 12 hour post ingestion > 100 ug/ml 8 hour post ingestion > 200 ug/ml 4 hour post ingestion ACTIVATED PROTEIN C RESIS 0D
New Mexico Emerging Infections Program Overview. Joan Baumbach NM Department of Health September 23, 2016
 New Mexico Emerging Infections Program Overview Joan Baumbach NM Department of Health September 23, 2016 Emerging Infections Program History Established in 1995 as population-based, scientific, public
New Mexico Emerging Infections Program Overview Joan Baumbach NM Department of Health September 23, 2016 Emerging Infections Program History Established in 1995 as population-based, scientific, public
lab Test Name notes on collection container type CBC AUTO HEMOGLOBIN & HEMATOCRIT
 lab Test Name notes on collection container type CBC AUTO HEMOGLOBIN & HEMATOCRIT CBC HEMOGRAM LAV HEMOGLOBIN HEMATOCRIT PLATELET COUNT & IPF LAV ERYTHROCYTE SEDIMENTATION RATE MALARIA SMEAR RETICULOCYTE
lab Test Name notes on collection container type CBC AUTO HEMOGLOBIN & HEMATOCRIT CBC HEMOGRAM LAV HEMOGLOBIN HEMATOCRIT PLATELET COUNT & IPF LAV ERYTHROCYTE SEDIMENTATION RATE MALARIA SMEAR RETICULOCYTE
What tests should be on the Alert List?
 What tests should be on the Alert List? Dr Que Lam On behalf RCPA-AACB High Risk Results Working Party: Alan McNeil, Grahame Caldwell, Craig Campbell, Penelope Coates, Robert Flatman, Andrew Georgiou,
What tests should be on the Alert List? Dr Que Lam On behalf RCPA-AACB High Risk Results Working Party: Alan McNeil, Grahame Caldwell, Craig Campbell, Penelope Coates, Robert Flatman, Andrew Georgiou,
LABORATORY NORMAL RANGES. Prepared by Date Adopted Supersedes Procedure # / Dated William T. Pope, PhD and
 Prepared by Date Adopted Supersedes Procedure # / Dated William T. Pope, PhD and 3-31-09 Normal Ranges / 12-30-08 Christy O Brien, MT (ASCP) CHEMISTRY Acetone None detected Albumin, g/dl 3.5-5.0 Adult
Prepared by Date Adopted Supersedes Procedure # / Dated William T. Pope, PhD and 3-31-09 Normal Ranges / 12-30-08 Christy O Brien, MT (ASCP) CHEMISTRY Acetone None detected Albumin, g/dl 3.5-5.0 Adult
Delta Check Calculation Guide
 Delta Check Calculation Guide National Technology 2017, All Rights Reserved By Senior Scientific Researcher, Asmaa Taher Table of Contents Definition... 2 Purpose... 2 Delta Check Research Studies... 2
Delta Check Calculation Guide National Technology 2017, All Rights Reserved By Senior Scientific Researcher, Asmaa Taher Table of Contents Definition... 2 Purpose... 2 Delta Check Research Studies... 2
cobas c 501 analyzer and cobas c 311 analyzer Within Run Imprecision Guidelines
 cobas c 501 analyzer and cobas c 311 analyzer General Information How to use these guidelines Unless otherwise indicated, the data presented is the same for both the cobas c 501 analyzer and the cobas
cobas c 501 analyzer and cobas c 311 analyzer General Information How to use these guidelines Unless otherwise indicated, the data presented is the same for both the cobas c 501 analyzer and the cobas
BACTERIOLOGY PROGRAMME AND PLAN OF TEACHING 3 rd Semester (academic year )
 BACTERIOLOGY PROGRAMME AND PLAN OF TEACHING 3 rd Semester (academic year 2012-2013) 19. 10. 2012. Introduction in microbiology, bacterial taxonomy, general bacterial prop Bacterial structures, biosynthesis
BACTERIOLOGY PROGRAMME AND PLAN OF TEACHING 3 rd Semester (academic year 2012-2013) 19. 10. 2012. Introduction in microbiology, bacterial taxonomy, general bacterial prop Bacterial structures, biosynthesis
Microbiology Specimen Collection Instructions. Table I Culture and Sensitivities/TB
 Specimen Page 1 of 10 Source / Type of Specimen Culture and Sensitivities () Abscess aspirate, wound aspirate in sterile screw cap or swab in charcoal. Obtain anaerobic swab from lab. Pilonidal and perirectal
Specimen Page 1 of 10 Source / Type of Specimen Culture and Sensitivities () Abscess aspirate, wound aspirate in sterile screw cap or swab in charcoal. Obtain anaerobic swab from lab. Pilonidal and perirectal
Fever Without a Source Age: 0-28 Day Pathway - Emergency Department Evidence Based Outcome Center
 Age: 0-28 Day Pathway - Emergency Department EXCLUSION CRITERIA Toxic appearing No fever Born < 37 weeks gestational age INCLUSION CRITERIA Non-toxic with temperature > 38 C (100.4 F) < 36 C (96.5 F) measured
Age: 0-28 Day Pathway - Emergency Department EXCLUSION CRITERIA Toxic appearing No fever Born < 37 weeks gestational age INCLUSION CRITERIA Non-toxic with temperature > 38 C (100.4 F) < 36 C (96.5 F) measured
IP Lab Webinar 8/23/2012
 2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
Alaska Native Medical Center Anchorage, AK
 ANMC Lab Test Requirements Key: Room Temp (20-25C), Refrigerated (2-8C), (-15 to -25C), Hr (Hours), D (Days), W (Weeks), Mo (Months), Yr (Years). Basic Processing Instructions: Centrifuge all blood specimens
ANMC Lab Test Requirements Key: Room Temp (20-25C), Refrigerated (2-8C), (-15 to -25C), Hr (Hours), D (Days), W (Weeks), Mo (Months), Yr (Years). Basic Processing Instructions: Centrifuge all blood specimens
Add your company slogan LOGO
 Add your company slogan LOGO Add your company slogan LOGO Louisiana State University Health Sciences Center Results of specimen that is out of normal range and probably is associated with high mortality
Add your company slogan LOGO Add your company slogan LOGO Louisiana State University Health Sciences Center Results of specimen that is out of normal range and probably is associated with high mortality
CLIA APPROVED PROFICIENCY TESTING PROGRAMS ACCUTEST, INC. P.O. Box 999 Westford, Massachusetts (800)
 ACCUTEST, INC. P.O. Box 999 Westford, Massachusetts 01886 (800) 665-2575 MICROBIOLOGY Bacteriology Aerobic Culture and Identification Antibiotic Susceptibility Testing Direct Antigen Detection Gram Stain
ACCUTEST, INC. P.O. Box 999 Westford, Massachusetts 01886 (800) 665-2575 MICROBIOLOGY Bacteriology Aerobic Culture and Identification Antibiotic Susceptibility Testing Direct Antigen Detection Gram Stain
MICROBIOLOGY SPECIMEN COLLECTION MANUAL
 Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Clinical Microbiology CLS 2019 Clinical Microbiology Laboratory Scheme Application Form
 Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
enter the room. Persons immune from previous varicella infection may enter the room without a mask. Those immune by adenoviruses, influenza viruses.
 All clients admitted to the hospital automatically are considered to be on standard precautions. The diseases listed below require standard precautions plus additional precautions that are noted in the
All clients admitted to the hospital automatically are considered to be on standard precautions. The diseases listed below require standard precautions plus additional precautions that are noted in the
PATHOGEN DETECTION WITH THE FILMARRAY
 PATHOGEN DETECTION WITH THE FILMARRAY The System Sample-to-Answer in an Hour Single sample Multiple samples The FilmArray integrates sample preparation, amplification, detection, and analysis all into
PATHOGEN DETECTION WITH THE FILMARRAY The System Sample-to-Answer in an Hour Single sample Multiple samples The FilmArray integrates sample preparation, amplification, detection, and analysis all into
Hamilton Regional Laboratory Medicine Program
 Created: April 2002 of Review: February 2004 of Review: June 2006 of Review: July 2007, St. Joseph s Healthcare went live with Meditech as of June18, 2007. of Review: August 2009 of Review: December 2011;
Created: April 2002 of Review: February 2004 of Review: June 2006 of Review: July 2007, St. Joseph s Healthcare went live with Meditech as of June18, 2007. of Review: August 2009 of Review: December 2011;
INFECTIOUS DISEASE. Page 2
 Infectious disease Advantages OF TESTING INFECTIOUS DISEASE We are in the middle of a paradigm shift in infectious disease diagnostic testing. As we move from targeted infectious disease testing to a syndromic
Infectious disease Advantages OF TESTING INFECTIOUS DISEASE We are in the middle of a paradigm shift in infectious disease diagnostic testing. As we move from targeted infectious disease testing to a syndromic
BRATTLEBORO MEMORIAL HOSPITAL 2016 LAB GUIDE GENERAL INFORMATION
 BRATTLEBORO MEMORIAL HOSPITAL 2016 LAB GUIDE ADMINISTRATIVE TEAM Medical Director of Laboratory... Christopher Appleton, D.O. Administrative Director of Laboratory Services... Imogene Drakes, PhD, FACHE
BRATTLEBORO MEMORIAL HOSPITAL 2016 LAB GUIDE ADMINISTRATIVE TEAM Medical Director of Laboratory... Christopher Appleton, D.O. Administrative Director of Laboratory Services... Imogene Drakes, PhD, FACHE
Breakout Session C: Harmonisation of the Alert Table.
 Breakout Session C: Harmonisation of the Alert Table. RCPA-AACB High Risk Results Working Party Andrew Georgiou Craig Campbell Grahame Caldwell Hans Schneider Penelope Coates Que Lam Rita Horvath Robert
Breakout Session C: Harmonisation of the Alert Table. RCPA-AACB High Risk Results Working Party Andrew Georgiou Craig Campbell Grahame Caldwell Hans Schneider Penelope Coates Que Lam Rita Horvath Robert
PATHOLOGY SPECIMEN TUBES QUICK REFERENCE GUIDE : Edition Expires July 2013
 PATHOLOGY SPECIMEN TUBES QUICK REFERENCE GUIDE : 2012 2013 Edition Expires July 2013 FOR THE MOST UP TO DATE INFORMATION AND FOR TESTS THAT ARE NOT LISTED HERE PLEASE CHECK THE INTRANET SITE AT: http://rhhlas26/path_tests/lab_default.asp
PATHOLOGY SPECIMEN TUBES QUICK REFERENCE GUIDE : 2012 2013 Edition Expires July 2013 FOR THE MOST UP TO DATE INFORMATION AND FOR TESTS THAT ARE NOT LISTED HERE PLEASE CHECK THE INTRANET SITE AT: http://rhhlas26/path_tests/lab_default.asp
MICROBIOLOGICAL TESTING IN PICU
 MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
LANE RMC TEST DICTIONARY For Lab testing not listed below - contact
 LANE RMC TEST DICTIONARY For Lab testing not listed below - contact 658-4320 Revised 03/2017 TEST NAME SPECIMEN VOLUME CONTAINER SPECIAL INSTRUCTIONS Acetone Plasma or Serum 1ml Green Gel or SST Green
LANE RMC TEST DICTIONARY For Lab testing not listed below - contact 658-4320 Revised 03/2017 TEST NAME SPECIMEN VOLUME CONTAINER SPECIAL INSTRUCTIONS Acetone Plasma or Serum 1ml Green Gel or SST Green
PAMET Continuing Education 2016
 PAMET Continuing Education 2016 Agent of gastroenteritis Medium/method] used for routine screening/detection in stool samples Salmonella, Shigella, MacConkey, Hektoen, Bismuth sulfite,etc. Plesiomonas
PAMET Continuing Education 2016 Agent of gastroenteritis Medium/method] used for routine screening/detection in stool samples Salmonella, Shigella, MacConkey, Hektoen, Bismuth sulfite,etc. Plesiomonas
MEMORANDUM. These reference ranges are effective immediately but sample requirements remain unchanged currently.
 MEMORANDUM Originating Department Chemical Pathology Issued By: Issued To: Subject: Details: Dr. Shari Srinivasan All Laboratory Service Users Change in Chemical Pathology Analysers Dear Colleague, As
MEMORANDUM Originating Department Chemical Pathology Issued By: Issued To: Subject: Details: Dr. Shari Srinivasan All Laboratory Service Users Change in Chemical Pathology Analysers Dear Colleague, As
Program Analyte Unit Target Limit Absolute SD %
 Andrology Sperm Count 1.0E06/mL Peer Group Mean SD 2 Sperm Morphology % normal Peer Group Mean SD 2 Sperm Viability % viable Peer Group Mean SD 2 Endocrinology Cortisol µg/dl Peer Group Mean % of Target
Andrology Sperm Count 1.0E06/mL Peer Group Mean SD 2 Sperm Morphology % normal Peer Group Mean SD 2 Sperm Viability % viable Peer Group Mean SD 2 Endocrinology Cortisol µg/dl Peer Group Mean % of Target
SPECTRA EAST, INC. Rockleigh, NJ
 A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Medical Testing This laboratory is accredited in accordance with the recognized International Standard ISO/IEC
A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Medical Testing This laboratory is accredited in accordance with the recognized International Standard ISO/IEC
Online catalog
 This catalog contains information about tests performed at Green Clinic Laboratory. For samples to be sent to Quest Diagnostics or any other reference lab please contact the Green Clinic Laboratory (318-251-6378)
This catalog contains information about tests performed at Green Clinic Laboratory. For samples to be sent to Quest Diagnostics or any other reference lab please contact the Green Clinic Laboratory (318-251-6378)
Hamilton Regional Laboratory Medicine Program
 Created: April 2002 of Review: February 2004 of Review: June 2006 of Review: July 2007, St. Joseph s Healthcare went live with Meditech as of June18, 2007. of Review: August 2009 of Review: December 2011;
Created: April 2002 of Review: February 2004 of Review: June 2006 of Review: July 2007, St. Joseph s Healthcare went live with Meditech as of June18, 2007. of Review: August 2009 of Review: December 2011;
BRATTLEBORO MEMORIAL HOSPITAL 2018 LAB GUIDE GENERAL INFORMATION
 BRATTLEBORO MEMORIAL HOSPITAL 2018 LAB GUIDE ADMINISTRATION Medical Director of Laboratory... Christopher Appleton, D.O. FACP Administrative Director of Laboratory Services... Imogene Drakes, PhD, FACHE
BRATTLEBORO MEMORIAL HOSPITAL 2018 LAB GUIDE ADMINISTRATION Medical Director of Laboratory... Christopher Appleton, D.O. FACP Administrative Director of Laboratory Services... Imogene Drakes, PhD, FACHE
NPQR 2018 Qualified Clinical Data Registry (QCDR) Measures 21_18247_LS.
 NPQR 2018 Qualified Clinical Data Registry (QCDR) Measures 21_18247_LS MEASURE ID: NPQR1 MEASURE TITLE: Notification to the Ordering Provider Requesting Myoglobin or CK-MB in the Diagnosis of Suspected
NPQR 2018 Qualified Clinical Data Registry (QCDR) Measures 21_18247_LS MEASURE ID: NPQR1 MEASURE TITLE: Notification to the Ordering Provider Requesting Myoglobin or CK-MB in the Diagnosis of Suspected
M O L E C U L A R G E N E T I C S
 MOLECULAR GENETICS ADVANTAGES OF MOLECULAR GENETICS Molecular genetics is a dynamic and transformative area of diagnostics, leading to insights in research and treatment in many disease states that are
MOLECULAR GENETICS ADVANTAGES OF MOLECULAR GENETICS Molecular genetics is a dynamic and transformative area of diagnostics, leading to insights in research and treatment in many disease states that are
CERTIFICATE OF ACCREDITATION
 CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Sprung CL, Annane D, Keh D, et al. Hydrocortisone therapy for
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Sprung CL, Annane D, Keh D, et al. Hydrocortisone therapy for
Antibiotic Resistance Pattern of Blood and CSF Culture Isolates At NHLS Academic Laboratories (2005)
 Antibiotic Resistance Pattern of Blood and CSF Culture Isolates At NHLS Academic Laboratories (2005) Streptococcus pneumoniae (SP) Blood Culture Isolates Penicillin intermediate Penicillin Cefotaxime 336
Antibiotic Resistance Pattern of Blood and CSF Culture Isolates At NHLS Academic Laboratories (2005) Streptococcus pneumoniae (SP) Blood Culture Isolates Penicillin intermediate Penicillin Cefotaxime 336
AMPLIRUN TOTAL A RELIABLE QUALITY CONTROL SOURCE FOR NUCLEIC ACID TESTS
 AMPLIRUN TOTAL A RELIABLE QUALITY CONTROL SOURCE FOR NUCLEIC ACID TESTS Take Control, Molecular Control. 02 04 05 06 06 07 08 08 AMPLIRUN TOTAL RESPIRATORY INFECTIONS TUBERCULOSIS INFECTIONS GASTROINTESTINAL
AMPLIRUN TOTAL A RELIABLE QUALITY CONTROL SOURCE FOR NUCLEIC ACID TESTS Take Control, Molecular Control. 02 04 05 06 06 07 08 08 AMPLIRUN TOTAL RESPIRATORY INFECTIONS TUBERCULOSIS INFECTIONS GASTROINTESTINAL
ISO 15189:2012 Internationally-Recognized Accredited Laboratory. SPECTRA EAST, INC. Rockleigh, NJ
 ISO 15189:2012 Internationally-Recognized Accredited Laboratory A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Clinical Testing This laboratory is accredited
ISO 15189:2012 Internationally-Recognized Accredited Laboratory A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Clinical Testing This laboratory is accredited
Microbiology EQA Product Portfolio
 Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
Adam Aragon Lisa Onischuk Paul Torres NM DOH, Scientific Laboratory Division
 Adam Aragon Lisa Onischuk Paul Torres NM DOH, Scientific Laboratory Division New Mexico Scientific Laboratories 1101 Camino de Salud, NE Albuquerque, NM 87102 Scientific Laboratory Division SLD Office
Adam Aragon Lisa Onischuk Paul Torres NM DOH, Scientific Laboratory Division New Mexico Scientific Laboratories 1101 Camino de Salud, NE Albuquerque, NM 87102 Scientific Laboratory Division SLD Office
Routine, Every 2 hours, Starting today, If temperature greater than 38.5 C initiate Evaluation for Possible Sepsis Physician Order #829
 Height Weight Allergies General Vital Signs [X] Frequent vital signs Indication: Q15 minutes x (# of occurrences): 4 Q30 minutes x (# of occurrences): 4 Q1 hour x (# of occurrences): Q2 hours x (# of occurrences):
Height Weight Allergies General Vital Signs [X] Frequent vital signs Indication: Q15 minutes x (# of occurrences): 4 Q30 minutes x (# of occurrences): 4 Q1 hour x (# of occurrences): Q2 hours x (# of occurrences):
Respiratory Pathogen Panel TEM-PCR Test Code:
 Respiratory Pathogen Panel TEM-PCR Test Code: 220000 Tests in this Panel Enterovirus group Human bocavirus Human coronavirus (4 types) Human metapneumovirus Influenza A - Human influenza Influenza A -
Respiratory Pathogen Panel TEM-PCR Test Code: 220000 Tests in this Panel Enterovirus group Human bocavirus Human coronavirus (4 types) Human metapneumovirus Influenza A - Human influenza Influenza A -
LABORATORY TEST INFORMATION GUIDE Laboratory Medicine
 Alanine Aminotransferase Specimen: Blood Container: Lithium Heparin Volume: 2.5 4.5 ml Requirements: Invert 8-10 times Stability: 7 days 2-8ºC, Separated: 60 days at -20 ºC 1 of 126 Albumin Specimen: Blood
Alanine Aminotransferase Specimen: Blood Container: Lithium Heparin Volume: 2.5 4.5 ml Requirements: Invert 8-10 times Stability: 7 days 2-8ºC, Separated: 60 days at -20 ºC 1 of 126 Albumin Specimen: Blood
DTH Meditech HCIS Reference Intervals
 Applicability Purpose This document applies to all Central Zone DTH Meditech HCIS personnel of AHS Laboratory Services and laboratories administered under the Covenant Health Group collectively referred
Applicability Purpose This document applies to all Central Zone DTH Meditech HCIS personnel of AHS Laboratory Services and laboratories administered under the Covenant Health Group collectively referred
An Intriguing Case of Meningitis. Tiffany Mylius MLS (ASCP)
 An Intriguing Case of Meningitis { Tiffany Mylius MLS (ASCP) A 4yo male presents with 2wk history of URI symptoms. On the day of admission, the patient woke up with a HA in the morning, took a nap later
An Intriguing Case of Meningitis { Tiffany Mylius MLS (ASCP) A 4yo male presents with 2wk history of URI symptoms. On the day of admission, the patient woke up with a HA in the morning, took a nap later
Postanalytical phase
 Postanalytical phase Test request POSTANALYTICAL Result interpretation PHASE Result Sampling Black box: the lab And the RESULT is created The technician approves the result; it is transferred to the lab
Postanalytical phase Test request POSTANALYTICAL Result interpretation PHASE Result Sampling Black box: the lab And the RESULT is created The technician approves the result; it is transferred to the lab
Sep Oct Nov Dec Total
 LB PAGE 2 LB PAGE 3 Sep Oct Nov Dec 2007 2007 2007 2007 Total Repeat Information Total Repeats 35 15 17 9 76 Repeat Rate 6.01% 0.17% 1.12% 0.39% 2.07% Repeat Chemistry 25 0 2 0 27 Repeat Extraction 1 0
LB PAGE 2 LB PAGE 3 Sep Oct Nov Dec 2007 2007 2007 2007 Total Repeat Information Total Repeats 35 15 17 9 76 Repeat Rate 6.01% 0.17% 1.12% 0.39% 2.07% Repeat Chemistry 25 0 2 0 27 Repeat Extraction 1 0
Microbiology & Virology Resource Comprehensive
 Abscess, aspirate, drainage culture Actinomyces culture Aerobic Bacterial culture, stain AFB culture & smear (Acid Fast, TB) AFB Blood culture Anaerobic culture Blood culture Body Fluid culture, stain
Abscess, aspirate, drainage culture Actinomyces culture Aerobic Bacterial culture, stain AFB culture & smear (Acid Fast, TB) AFB Blood culture Anaerobic culture Blood culture Body Fluid culture, stain
UK Meningitis Study CRF
 History Date of onset of symptoms Route of admission A and E GP Other Date of admission to hospital* / / Time of admission (24 hour clock) : *record time and date of admission to A and E if admitted ia
History Date of onset of symptoms Route of admission A and E GP Other Date of admission to hospital* / / Time of admission (24 hour clock) : *record time and date of admission to A and E if admitted ia
OMCJH.CHEM.COLL.INF.1001 Therapeutic Drug Monitoring Guidelines
 OMCJH.CHEM.COLL.INF.1001 Copy of version 1.0 (approved and current) Last Approval or Periodic Review Completed 10/5/2017 Next Periodic Review Needed On or Before 10/5/2019 Effective Date 10/5/2017 Controlled
OMCJH.CHEM.COLL.INF.1001 Copy of version 1.0 (approved and current) Last Approval or Periodic Review Completed 10/5/2017 Next Periodic Review Needed On or Before 10/5/2019 Effective Date 10/5/2017 Controlled
DEPARTMENT OF PATHOLOGY AND LABORATORY MEDICINE LABORATORY SERVICES & SPECIMEN COLLECTION MANUAL
 DEPARTMENT OF PATHOLOGY AND LABORATORY MEDICINE LABORATORY SERVICES & SPECIMEN COLLECTION MANUAL LABORATORY SERVICES MANUAL Contents GENERAL LABORATORY... 6 INPATIENT/EMERGENCY DEPARTMENT SERVICES HOURS
DEPARTMENT OF PATHOLOGY AND LABORATORY MEDICINE LABORATORY SERVICES & SPECIMEN COLLECTION MANUAL LABORATORY SERVICES MANUAL Contents GENERAL LABORATORY... 6 INPATIENT/EMERGENCY DEPARTMENT SERVICES HOURS
Types of target values, acceptable ranges
 Types of target values, acceptable ranges As the differential of rounding, maximum 1 percent point deviation is allowed from the maximum acceptable range. 100. Clinical chemistry (wet) 1. Calcium RMV 10
Types of target values, acceptable ranges As the differential of rounding, maximum 1 percent point deviation is allowed from the maximum acceptable range. 100. Clinical chemistry (wet) 1. Calcium RMV 10
Blood/Blood Component Utilization and Administration Annual Compliance Education
 Blood/Blood Component Utilization and Administration Annual Compliance Education This course contains annual compliance education necessary to meet compliance and regulatory requirements. Instructions:
Blood/Blood Component Utilization and Administration Annual Compliance Education This course contains annual compliance education necessary to meet compliance and regulatory requirements. Instructions:
Contents. 1. General Introduction 3
 Section 1 Medical Laboratory 1. General Introduction 3 Guidelines 3 Code of Ethics for Laboratory Technicians 3 Responsibilities of Medical Technician 3 Instructions for Laboratory Technicians 5 Arrangement
Section 1 Medical Laboratory 1. General Introduction 3 Guidelines 3 Code of Ethics for Laboratory Technicians 3 Responsibilities of Medical Technician 3 Instructions for Laboratory Technicians 5 Arrangement
Potential Reimbursement CPT Codes
 BioFire FilmArray Blood Culture Identification (BCID) Panel Medicare All targets (n) 87150 n x * *BioFire BCID Panel is comprised of 27 total targets. The number of targets allowed for reimbursement may
BioFire FilmArray Blood Culture Identification (BCID) Panel Medicare All targets (n) 87150 n x * *BioFire BCID Panel is comprised of 27 total targets. The number of targets allowed for reimbursement may
Mechanisms of Pathogenicity
 Mechanisms of Pathogenicity The Microbes Fight Back Medically important bacteria Salmonella Bacillus anthracis Shigella dysenteriae Campylobacter Shigella sonnei Clostridium botulinum Staphylococcus aureus
Mechanisms of Pathogenicity The Microbes Fight Back Medically important bacteria Salmonella Bacillus anthracis Shigella dysenteriae Campylobacter Shigella sonnei Clostridium botulinum Staphylococcus aureus
CERTIFICATE OF ACCREDITATION
 CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
Infection Control. Student Orientation
 Infection Control Student Orientation Basic, but important, Principles of Cross Transmission Presence of microorganisms on hands or in environment does not necessarily = cross transmission or infection
Infection Control Student Orientation Basic, but important, Principles of Cross Transmission Presence of microorganisms on hands or in environment does not necessarily = cross transmission or infection
Manufacturer Report for Siemens Unassayed Chemistry Lot Exp 30 Jun 2018
 Acetaminophen Enzymatic, colorimetric µg/ml.09 0..0.09 0..0 0. 0. 0. 0. 9.. 9.0 0.9.0..9.. Albumin Bromcresol Purple (BCP) g/dl.0 0.0..0 0.00.. 0.0.. 0.09..9 0.0..9 0.0..0 0.0..0 0.0. Alkaline Phosphatase
Acetaminophen Enzymatic, colorimetric µg/ml.09 0..0.09 0..0 0. 0. 0. 0. 9.. 9.0 0.9.0..9.. Albumin Bromcresol Purple (BCP) g/dl.0 0.0..0 0.00.. 0.0.. 0.09..9 0.0..9 0.0..0 0.0..0 0.0. Alkaline Phosphatase
Preferred Specimen Vaginal secretions from the lower 1/3 of the vaginal wall collected using a sterile cotton tipped applicator
 B Bacterial Vaginosis Sterile leak-proof screw cap container (SCM) Vaginal secretions from the lower 1/3 of the vaginal wall collected using a sterile cotton tipped applicator Use a sterile cotton tipped
B Bacterial Vaginosis Sterile leak-proof screw cap container (SCM) Vaginal secretions from the lower 1/3 of the vaginal wall collected using a sterile cotton tipped applicator Use a sterile cotton tipped
ISF criteria (International sepsis forum consensus conference of infection in the ICU) Secondary peritonitis
 Appendix with supplementary material. This appendix was part of the submitted manuscript and has been peer reviewed. It is posted as supplied by the authors. Supplementary Tables Table S1. Definitions
Appendix with supplementary material. This appendix was part of the submitted manuscript and has been peer reviewed. It is posted as supplied by the authors. Supplementary Tables Table S1. Definitions
Review the following alphabetical listing of our lab test catalog for each of our offered tests. Each page has at least the following elements:
 Site: Fremont Rideout Health Group Laboratory Services Policy and Procedure Creation Date: 08/17/2009 Subject/Title: FRHG Laboratories' Catalog of Tests Q-S Document Owner: Corson, Karen Pagination: Page
Site: Fremont Rideout Health Group Laboratory Services Policy and Procedure Creation Date: 08/17/2009 Subject/Title: FRHG Laboratories' Catalog of Tests Q-S Document Owner: Corson, Karen Pagination: Page
WVU Medicine Specimens Submitted for Microbiology Testing
 WVU Medicine Specimens Submitted for Microbiology Testing 1. Upper Respiratory a. anterior nares.. page 3 b. nasopharynx.. page 4 c. throat..... page 5 2. Lower Respiratory a. Sputum, tracheal aspirates,
WVU Medicine Specimens Submitted for Microbiology Testing 1. Upper Respiratory a. anterior nares.. page 3 b. nasopharynx.. page 4 c. throat..... page 5 2. Lower Respiratory a. Sputum, tracheal aspirates,
WSLH. Calibration Verification/ Linearity Products. roficiency. esting. Products provided in partnership with:
 WSLH PT roficiency esting Calibration Verification/ Linearity Products Products provided in partnership with: www.wslhpt.org 800-462-5261 PTService@slh.wisc.edu General Chemistry Ammonia/Ethanol - 5 x
WSLH PT roficiency esting Calibration Verification/ Linearity Products Products provided in partnership with: www.wslhpt.org 800-462-5261 PTService@slh.wisc.edu General Chemistry Ammonia/Ethanol - 5 x
6/6/2016 pg. 1 of 24
 Last Reviewed: (Updated 06.06.16) By: Dr. Heidi Budke LTR29828 (BVH and ACC shared reference ranges) Hematology Reference Ranges: WBC LH750 AND 0-1 Day 9.0-30.0 DXH800 1-3 Days 9.4-34.0 4 Days - 1 Month
Last Reviewed: (Updated 06.06.16) By: Dr. Heidi Budke LTR29828 (BVH and ACC shared reference ranges) Hematology Reference Ranges: WBC LH750 AND 0-1 Day 9.0-30.0 DXH800 1-3 Days 9.4-34.0 4 Days - 1 Month
Selected New Jersey Communicable Disease Regulations
 COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response Selected New Jersey Communicable Disease Regulations June 2013 Public Health Emergency Response Capability Requirements as stated
COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response Selected New Jersey Communicable Disease Regulations June 2013 Public Health Emergency Response Capability Requirements as stated
Controls & Calibrators Clinical Chemistry
 Controls & Calibrators Clinical Chemistry Clinical Chemistry Controls & Lipids Clinical Chemistry and lipid quality controls have been manufactured from true human serum to ensure they perform the same
Controls & Calibrators Clinical Chemistry Clinical Chemistry Controls & Lipids Clinical Chemistry and lipid quality controls have been manufactured from true human serum to ensure they perform the same
Massachusetts Department of Public Health Bureau of Communicable Disease Control Office of Integrated Surveillance and Informatics Services (ISIS)
 Massachusetts Department of Public Health Bureau of Communicable Disease Control Office of Integrated Surveillance and Informatics Services (ISIS) Summary of Amendments to 105 CMR 300.000: Reportable Diseases,
Massachusetts Department of Public Health Bureau of Communicable Disease Control Office of Integrated Surveillance and Informatics Services (ISIS) Summary of Amendments to 105 CMR 300.000: Reportable Diseases,
3/9/2018. The Power of a Gram Stain (and Other Micro Results): What your Infection Preventionist Needs to Know Now
 The Power of a Gram Stain (and Other Micro Results): What your Infection Preventionist Needs to Know Now Sara J. Blosser, Ph.D., D(ABMM) Director, Division of Clinical Microbiology Indiana State Department
The Power of a Gram Stain (and Other Micro Results): What your Infection Preventionist Needs to Know Now Sara J. Blosser, Ph.D., D(ABMM) Director, Division of Clinical Microbiology Indiana State Department
CME/SAM. An Examination of the Usefulness of Repeat Testing Practices in a Large Hospital Clinical Chemistry Laboratory
 Clinical Chemistry / Repeat Testing Practices in Clinical Chemistry An Examination of the Usefulness of Repeat Testing Practices in a Large Hospital Clinical Chemistry Laboratory Carl O. Deetz, MD, PhD,
Clinical Chemistry / Repeat Testing Practices in Clinical Chemistry An Examination of the Usefulness of Repeat Testing Practices in a Large Hospital Clinical Chemistry Laboratory Carl O. Deetz, MD, PhD,
Cleaning for Additional Precautions Table symptom based
 for Additional Precautions Table symptom based The need to wear personal protective equipment () for Routine Practices is dependent on the risk of contact or contamination with blood or body fluids. should
for Additional Precautions Table symptom based The need to wear personal protective equipment () for Routine Practices is dependent on the risk of contact or contamination with blood or body fluids. should
Pathology and Laboratory
 Pathology and Laboratory CPT CPT copyright 2011 American Medical Association. All rights reserved. Fee schedules, relative value units, conversion factors and/or related components are not assigned by
Pathology and Laboratory CPT CPT copyright 2011 American Medical Association. All rights reserved. Fee schedules, relative value units, conversion factors and/or related components are not assigned by
CHAPTER 515. COMMUNICABLE DISEASE AND INJURY REPORTING EFFECTIVE 9/12/2014
 TITLE 310. CHAPTER 515. COMMUNICABLE DISEASE AND INJURY REPORTING EFFECTIVE 9/12/2014 310:515-1-1. Purpose The rules in this Chapter implement the Communicable Diseases Reporting Regulations, 63 O.S. 1981,
TITLE 310. CHAPTER 515. COMMUNICABLE DISEASE AND INJURY REPORTING EFFECTIVE 9/12/2014 310:515-1-1. Purpose The rules in this Chapter implement the Communicable Diseases Reporting Regulations, 63 O.S. 1981,
