95% 2.5% 2.5% +2SD 95% of data will 95% be within of data will 1.96 be within standard deviations 1.96 of sample mean
|
|
|
- William Manning
- 5 years ago
- Views:
Transcription
1 Efficient Clinical Trials John H. Powers, MD Senior Medical Scientist SAIC in support of Clinical Collaborative Research Branch NIAID/NIH Introduction Definitions what is a small clinical trial? Review of ethical considerations in designing trials Review of basic ingredients of sample size estimation and concept of power Ways to increase efficiency of trials pros and cons for various methods Focused and relevant research question Changing error rates (not suggested) Enhancing effect sizes Decreasing variability 2 Definitions Clinical trial a controlled prospective study enrolling human subjects often used to evaluate the effectiveness and/or harms of interventions in treatment, prevention or diagnosis of disease Efficiency (in physics) ratio of useful work to the energy supplied to it In clinical trials, getting valid and reliable answers to important questions with the least amount of resources Does not mean putting patients at risk because of less valid data Lower sample size does not mean less work = MORE planning 3 1
2 Lower Sample Size = More Planning Clinical trials with small numbers of participants, however, must address broad sets of issues different from those that must be addressed in trials with large numbers of participants. It is in those circumstances of trials with small sample sizes that approaches to optimization of the study design and data interpretation pose greater challenges. Small Clinical Trials: Issues and Challenges, Institute of Medicine, p. ix 4 Definitions What is a large or small clinical trial? IOM defines a large trial as one that has adequate sample size to answer the primary research question = large enough A trial with very few participants may still have adequate statistical power e.g. if effect size is large Balance between exposing research subjects to potential harms of experimental interventions with obtaining valid answers 5 Underpowered Studies and Ethics 6 2
3 Underpowered Studies and Ethics A proposed study that cannot answer the question being asked because the necessary sample size cannot be attained should not be conducted on ethical grounds. That is, it is unacceptable to expose patients or research participants to harms, even inconveniences, if there is no prospect that t useful and potentially ti generalizable information will result from the study. Small Clinical Trials: Issues and Challenges, Institute of Medicine, p Small Clinical Trials Last Resort The importance of conducting small clinical trials only when there are no alternatives cannot be overemphasized. The committee is not encouraging the use of small clinical trials, but, rather provides advice on strategies that should be considered in the design and analysis of small clinical trials when the opportunity to perform a randomized clinical trial with adequate statistical power is not possible. In doing so, it recognizes that small clinical trials frequently need to be viewed as part of a continuing process of data collection. Thus, for some trials it might be impossible to definitively answer a research question with a high degree of confidence. In those cases, perhaps the best that one can do is assess the next set of questions to be asked. Small Clinical Trials: Issues and Challenges, Institute of Medicine, p Concerns About Small Clinical Trials Small numbers increase variability and leave much to chance Statistically significant outcomes may not be generalizable (only apply to circumstances in trial) Too many variables to assess cause and effect Only able to discern gross effects and limited ability to analyze covariates Incapable of identifying adverse events Small Clinical Trials: Issues and Challenges, Institute of Medicine, p
4 Situations Where Smaller Clinical Trials Justifiable Rare diseases Unique study populations (e.g. astronauts) Individually tailored therapies Environments that are isolated Emergency situations Public health urgencies Small Clinical Trials: Issues and Challenges, Institute of Medicine, p Small vs Efficient While small clinical trials are a last resort, efficient clinical trials are always justifiable Different methods to improve efficiency are useful (or not) depending on disease under study and research question/setting 11 Review of Sample Size Considerations 4
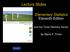
5 Normal Distribution of a Sample 95% 2.5% 2.5% +2SD 95% of data will be within 1.96 standard deviations of sample mean for large samples (>30) 13 Sample Size Superiority Trials Point estimate Of mean of One group Point estimate Of mean of Other group Clinical trials compare average effects in groups of subjects administered intervention to those not administered intervention Examine if populations differ by more than chance (for superiority trials) Example: Two groups with point estimate for means of 50 and zero, sample size of 12 per group, SD =60 14 Sample Size Superiority Trials In this example Blue area represents power (in this case 0.497) critical region of 0.05 test represented by dashed lines and red area To show difference due to greater than chance, want mean of one curve to be outside of red area 15 5
6 Sample Size Sample size in this example is 24 per group As sample size increases, overlap between curves decreases (assuming there really is a difference to show) Blue area increases = power is 0.80 Mean of one group now outside of critical area Notice still a good deal of overlap only mean value is outside critical area 16 Sample Size Sample size increased to 48 per group Distribution of data narrower and more precise Power = 0.98 Mean well outside of critical area 17 Sample Size Sample size increased to 96 per group Data still more precise Power = since no overlap of curves Mean well outside of critical area
7 Sample Size Want to select sample size large enough to show a difference if there is one to detect, but not too large Do not want to expose subjects unnecessarily to harm since this is an experiment evaluating interventions with unknown harm/benefits Use of resources - time, effort and money Sample size based on four parameters ( ingredients ) Type 1 error- usually specified as 0.05 two sided (0.025 on either side of curve) Type 2 error (1- type 2 error is power) usually specified as 0.10 to 0.20 (power of 80%-90%) Standard deviation of data (variability) Treatment difference Difference between point estimate of effect for intervention and point estimate for effect with control (delta) 19 Pictorial Representation of Sample Size 20 Pictorial Representation of Sample Size treatment effect size variability 21 7
8 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 22 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) Focusing the Research Question Research question needs to be one worth answering and of public health importance Need to focus question more questions mean greater sample size or less clear answers = simplify Many trials include measurements to try to figure out why the trial didn t work after it has failed the post-mortem on what the experiment died of 24 8
9 1. Focusing the Research Question Sample size is calculated AFTER one decides on a research question Starting out with a sample size and working back to what can I get for this is not justifiable in terms of choosing unrealistic or clinically meaningless/unachievable effect sizes 25 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) Changing Error Rates Error rates of type 1 = 0.05 and type 2 = 0.10 or 0.20 are by convention But false positive error rate of 1 in 20 trials is actually a low level l of evidence; need justification to deviate from this Increasing type 2 error rate increases likelihood of false negative conclusions = spending resources for unclear answers 27 9
10 Sample Size and Types of Error/Power Changing Error Rates Some discussion in medical literature of using one-sided rather than two-sided hypothesis testing = needs justification But.changes level of evidence to 1 in 10 false positive rate what justifies this? Assumes we are only interested in only one side of the story how much better is the test intervention, but don t we care how much harm might be done? Often statements of this intervention can t possibly be harmful are not justified by evidence So don t change error rates unless scientifically justified and even then most researchers want to see one sided error rates 29 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 30 10
11 Enhancing Effect Sizes Trials measure average effects in groups of subjects one size fits all approach may not be correct More homogenous populations can both decrease variability and increase effect sizes in presence of effect modification Effect modification is presence of quantitatively different effect sizes of an intervention based on a baseline variable Requires knowledge of natural history of disease and some evidence from prior trials Choosing population in which effect size is larger decrease sample size 31 Confounding and Effect Modification No confounding No effect modification Confounding No effect modification Outcome test control effect size Baseline variable No confounding Effect modification Confounding Effect modification 32 Examples Enrolling subjects in trial in whom effect is expected to be zero Dilutes effect size Ethical issues of exposure to harm for no benefit Trastuzumab (Herceptin) in breast cancer Mechanism of action by binding to HER2 proteins in person with specific genetic mutation 20% to 30% of person with breast cancer have this mutation Potentially harmful in those without the mutation 33 11
12 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 34 Optimizing Exposure Many trials include only one dose of an intervention with wide inter-individual individual variabililty in exposure Optimize dose based on pre-clinical and early clinical studies pharmacokinetics and pharmacodynamics Forms a hypothesis to test Not a substitute for clinical trials Standardize exposure of interventions - need unblinded third party to do this to maintain blinding in trial 35 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 36 12
13 Continuous vs Dichotomous Outcomes Continuous outcomes have more power to detect differences since uses all the data Dichotomous outcomes require 1) categorization assumes all data in a single category are similarly important which may not be true 2) choosing correct time point to evaluate if you re wrong, you miss it Requires more frequent data capture patient diaries or phone contact collected in systematic way 37 Time to Event - Cholera Saha D et al. N Engl J Med 2006;354: Time to Event - Influenza Treanor J et al. JAMA 2000;283(8):
14 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 40 Common or Composite Outcomes Choosing more common events or composite outcomes increases number of events and increases power to detect differences Some problems with interpretation: Only use when outcomes measured are of similar importance to patients if driven by less important outcomes may mask inferiority on more important outcomes Does not necessarily imply beneficial effect on all part of a composite 41 Composite Endpoints - Issues Lubsen J et al. Stat Med 2002;21:
15 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 43 More Sensitive/Specific Measures All measures composed of the true measure plus some associated error Error can be of two types: Random by chance alone Systematic bias based on individual biases and interpretations Decreasing error ( noise ) in relation to true measure ( signal ) allows smaller sample size 44 Non-Standardized Measures and Error Cochrane A et al Lancet 1951:
16 Non-Standardized Measures and Error Moertel CG and Hanley JA, Effect of measuring error on the results of therapeutic trials in advanced cancer. Cancer 1976;38: oncologists asked to measure 12 simulated tumor masses, two pairs of which were identical in size Allowed assessment of 64 measurements by same investigator and 1920 comparisons by different investigators 25% reduction as response for identical size masses = 19% response rate by same investigator, 25% between investigators (measurement error alone) 46 NMEs with PROs in approved labeling, by therapeutic category, ) 01 (Willke et al, CCT Antiinflammatory GI agents Ophthalmics CV agents Antivirals Antineoplastics Antiinfectives Anticoagulants % approvals with PRO data in label Note: Only therapeutic classes with at least 9 approvals are included. 47 Surrogate Endpoints Researchers often suggest biomarkers as surrogate endpoints in clinical trials NOT direct measures of patient benefit Idea originally was to decrease follow-up time in chronic diseases keep following subjects to validate biomarker (e.g. viral load in HIV/AIDS, cholesterol in stroke/mi prevention) Why use a surrogate in an acute disease when one can measure actual clinical outcomes? Surrogate as part of composite outcomes drive the entire outcome since more common 48 16
17 Limitations of Surrogate Endpoints 1. Surrogate endpoints may not be a true predictor of clinical outcomes Interventions may be clinically useful with no effect on surrogate Interventions may have effect on surrogate but no measurable clinical effects (or actual harms for patients) Still must measure clinical events that occur during trial and these supersede effect on surrogate May measure short term benefits that do not persist with longer followup 2. Surrogate endpoints may not yield a quantifiable clinical benefit that may be measured directly against adverse effects ICH E-9 document, p.7 3. Surrogate endpoints may hinder development of new types of therapies by insisting on only one mechanism of action by which intervention may affect disease 49 Strengths and Limitations Unmeasured benefits Intervention Surrogate Endpoint Unmeasured harm Other pathways of disease Clinical Endpoint Reasons why surrogate may not accurately predict clinical outcomes: unmeasured harms caused by intervention unmeasured benefits caused by intervention other mechanisms of disease other than those affected by intervention issues with measuring surrogate 50 issues with measuring clinical outcomes Use More Sensitive and Specific Measures From Dr Max s talk IPPCR
18 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 52 Follow-Up and Missing Data Enrolling subjects and then losing them to follow-up or missing data (failure to collect or losing it) results in effort for no gain Requires planning on part of researchers and work during the trial to make it as easy as possible for research subjects to return Phone calls and reminders Transportation Home visits Subjects who don t follow protocol are not missing and should be included in Intent to Treat (ITT) analyses 53 Efficient Clinical Trials Ways to decrease sample size 1. Focused and relevant research question 2. Changing error rates (not suggested) 3. Enhancing effect sizes More homogenous populations Choosing populations in whom effect size is larger Optimizing exposure Continuous instead of dichotomous outcomes More common or composite outcomes 4. Decreasing variability More sensitive/specific measures Assuring follow-up of enrolled subjects Study designs Cross-overover N=1 studies Sequential trial designs (e.g. dose-escalation escalation studies) 54 18
19 Study Designs Certain kinds of study designs can decrease variability and thereby decrease sample size Cross-over over and n=1 trials both use subjects as their own controls Randomize subjects to receive one intervention or the other (sometimes with wash out period in between) 55 Study Designs Limitations of cross-over over and n=1 trials Most useful in chronic illnessess with stable course of disease If effects carried over from one period treatment to the next then can bias study results Period effect for instance in seasonal diseases Most useful in diseases where treatment effect is rapid onset or rapid cessation when intervention stopped 56 Study Designs Sequential trial designs often used in Phase 1 dose escalation clinical trials Based on pre-defining what level of adverse events or not will allow progression to the next dose Makes decision based on data acquired during trial 57 19
20 Study Designs Adaptive designs - word that describes a variety of changes in trial design based on data accumulated during the trial Sequential trial dose escalation design is one form but more challenging when modifying other variables like the outcome measure Advantage = more subjects assigned to more succesful treatment Disadvantage = heterogeneity of subjects based on important risk factors which change as trial progresses introduces bias over time 58 A caveat. All the modification we have discussed apply to SUPERIORITY trials In non-inferiority trials investigators are locked into the design that was used to demonstrate the effect of the control intervention compared to placebo Disease definition Population Outcome definition and timing Concomitant medications Biases which trend results toward no difference in a superiority trial (like too small a sample size) result in false positive conclusions in non-inferiority trials 59 Non-Inferiority Trials Need to know reliable and reproducible benefit of control compared to placebo not recommended in treatment guidelines only Once this is know, need to evaluate methods used in the prior placebo controlled trials just like doing an experiment in the lab where conditions are held constant Given numerous biases should be trial design of last resort Reasons to use NI trials not consistent with reasons to do small clinical trials (presupposes an already effective intervention) 60 20
21 61 Conclusions Developing an efficient trial starts with planning and a good research question Question comes first, sample size second Various methods to increase effect sizes and decrease variability, when applied in the correct setting, can provide valid and reliable answers to important public health questions For some disease, developing the tools (better outcome measures, better data on natural history) is a good start to better trials 62 21
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON MISSING DATA
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
The RoB 2.0 tool (individually randomized, cross-over trials)
 The RoB 2.0 tool (individually randomized, cross-over trials) Study design Randomized parallel group trial Cluster-randomized trial Randomized cross-over or other matched design Specify which outcome is
The RoB 2.0 tool (individually randomized, cross-over trials) Study design Randomized parallel group trial Cluster-randomized trial Randomized cross-over or other matched design Specify which outcome is
Discrimination and Reclassification in Statistics and Study Design AACC/ASN 30 th Beckman Conference
 Discrimination and Reclassification in Statistics and Study Design AACC/ASN 30 th Beckman Conference Michael J. Pencina, PhD Duke Clinical Research Institute Duke University Department of Biostatistics
Discrimination and Reclassification in Statistics and Study Design AACC/ASN 30 th Beckman Conference Michael J. Pencina, PhD Duke Clinical Research Institute Duke University Department of Biostatistics
Assessing risk of bias
 Assessing risk of bias Norwegian Research School for Global Health Atle Fretheim Research Director, Norwegian Institute of Public Health Professor II, Uiniversity of Oslo Goal for the day We all have an
Assessing risk of bias Norwegian Research School for Global Health Atle Fretheim Research Director, Norwegian Institute of Public Health Professor II, Uiniversity of Oslo Goal for the day We all have an
Discussion Meeting for MCP-Mod Qualification Opinion Request. Novartis 10 July 2013 EMA, London, UK
 Discussion Meeting for MCP-Mod Qualification Opinion Request Novartis 10 July 2013 EMA, London, UK Attendees Face to face: Dr. Frank Bretz Global Statistical Methodology Head, Novartis Dr. Björn Bornkamp
Discussion Meeting for MCP-Mod Qualification Opinion Request Novartis 10 July 2013 EMA, London, UK Attendees Face to face: Dr. Frank Bretz Global Statistical Methodology Head, Novartis Dr. Björn Bornkamp
Indirect Treatment Comparison Without Network Meta-Analysis: Overview of Novel Techniques
 Indirect Treatment Comparison Without Network Meta-Analysis: Overview of Novel Techniques K. Jack Ishak, PhD, Executive Director, Statistics and Senior Research Scientist BACKGROUND There is generally
Indirect Treatment Comparison Without Network Meta-Analysis: Overview of Novel Techniques K. Jack Ishak, PhD, Executive Director, Statistics and Senior Research Scientist BACKGROUND There is generally
Beyond the intention-to treat effect: Per-protocol effects in randomized trials
 Beyond the intention-to treat effect: Per-protocol effects in randomized trials Miguel Hernán DEPARTMENTS OF EPIDEMIOLOGY AND BIOSTATISTICS Intention-to-treat analysis (estimator) estimates intention-to-treat
Beyond the intention-to treat effect: Per-protocol effects in randomized trials Miguel Hernán DEPARTMENTS OF EPIDEMIOLOGY AND BIOSTATISTICS Intention-to-treat analysis (estimator) estimates intention-to-treat
e.com/watch?v=hz1f yhvojr4 e.com/watch?v=kmy xd6qeass
 What you need to know before talking to your statistician about sample size Sharon D. Yeatts, Ph.D. Associate Professor of Biostatistics Data Coordination Unit Department of Public Health Sciences Medical
What you need to know before talking to your statistician about sample size Sharon D. Yeatts, Ph.D. Associate Professor of Biostatistics Data Coordination Unit Department of Public Health Sciences Medical
The Practice of Statistics 1 Week 2: Relationships and Data Collection
 The Practice of Statistics 1 Week 2: Relationships and Data Collection Video 12: Data Collection - Experiments Experiments are the gold standard since they allow us to make causal conclusions. example,
The Practice of Statistics 1 Week 2: Relationships and Data Collection Video 12: Data Collection - Experiments Experiments are the gold standard since they allow us to make causal conclusions. example,
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials
 In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
Psychology Research Process
 Psychology Research Process Logical Processes Induction Observation/Association/Using Correlation Trying to assess, through observation of a large group/sample, what is associated with what? Examples:
Psychology Research Process Logical Processes Induction Observation/Association/Using Correlation Trying to assess, through observation of a large group/sample, what is associated with what? Examples:
Assay Sensitivity.
 Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
Reflection paper on assessment of cardiovascular safety profile of medicinal products
 25 February 2016 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) Reflection paper on assessment of cardiovascular safety profile of medicinal products Draft agreed by Cardiovascular
25 February 2016 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) Reflection paper on assessment of cardiovascular safety profile of medicinal products Draft agreed by Cardiovascular
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research
 Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
EXERCISE: HOW TO DO POWER CALCULATIONS IN OPTIMAL DESIGN SOFTWARE
 ...... EXERCISE: HOW TO DO POWER CALCULATIONS IN OPTIMAL DESIGN SOFTWARE TABLE OF CONTENTS 73TKey Vocabulary37T... 1 73TIntroduction37T... 73TUsing the Optimal Design Software37T... 73TEstimating Sample
...... EXERCISE: HOW TO DO POWER CALCULATIONS IN OPTIMAL DESIGN SOFTWARE TABLE OF CONTENTS 73TKey Vocabulary37T... 1 73TIntroduction37T... 73TUsing the Optimal Design Software37T... 73TEstimating Sample
Challenges of Observational and Retrospective Studies
 Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
DRAFT GUIDANCE DOCUMENT Comparative Bioavailability Standards: Formulations Used for Systemic Effects
 1 2 3 DRAFT GUIDANCE DOCUMENT Comparative Bioavailability Standards: Formulations Used for Systemic Effects 4 This guidance document is being distributed for comment purposes only. 5 6 Published by authority
1 2 3 DRAFT GUIDANCE DOCUMENT Comparative Bioavailability Standards: Formulations Used for Systemic Effects 4 This guidance document is being distributed for comment purposes only. 5 6 Published by authority
Review. Chapter 5. Common Language. Ch 3: samples. Ch 4: real world sample surveys. Experiments, Good and Bad
 Review Ch 3: samples Sampling terminology Proportions Margin of error Ch 4: real world sample surveys Questions to ask about a study Errors in sample surveys Concerns about survey questions Probability
Review Ch 3: samples Sampling terminology Proportions Margin of error Ch 4: real world sample surveys Questions to ask about a study Errors in sample surveys Concerns about survey questions Probability
Statistics for Clinical Trials: Basics of Phase III Trial Design
 Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Study protocol v. 1.0 Systematic review of the Sequential Organ Failure Assessment score as a surrogate endpoint in randomized controlled trials
 Study protocol v. 1.0 Systematic review of the Sequential Organ Failure Assessment score as a surrogate endpoint in randomized controlled trials Harm Jan de Grooth, Jean Jacques Parienti, [to be determined],
Study protocol v. 1.0 Systematic review of the Sequential Organ Failure Assessment score as a surrogate endpoint in randomized controlled trials Harm Jan de Grooth, Jean Jacques Parienti, [to be determined],
Lecture Slides. Elementary Statistics Eleventh Edition. by Mario F. Triola. and the Triola Statistics Series 1.1-1
 Lecture Slides Elementary Statistics Eleventh Edition and the Triola Statistics Series by Mario F. Triola 1.1-1 Chapter 1 Introduction to Statistics 1-1 Review and Preview 1-2 Statistical Thinking 1-3
Lecture Slides Elementary Statistics Eleventh Edition and the Triola Statistics Series by Mario F. Triola 1.1-1 Chapter 1 Introduction to Statistics 1-1 Review and Preview 1-2 Statistical Thinking 1-3
Biases in clinical research. Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University
 Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Experimental Design. Terminology. Chusak Okascharoen, MD, PhD September 19 th, Experimental study Clinical trial Randomized controlled trial
 Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Fundamental Clinical Trial Design
 Design, Monitoring, and Analysis of Clinical Trials Session 1 Overview and Introduction Overview Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics, University of Washington February 17-19, 2003
Design, Monitoring, and Analysis of Clinical Trials Session 1 Overview and Introduction Overview Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics, University of Washington February 17-19, 2003
Department of OUTCOMES RESEARCH
 Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
Vocabulary. Bias. Blinding. Block. Cluster sample
 Bias Blinding Block Census Cluster sample Confounding Control group Convenience sample Designs Experiment Experimental units Factor Level Any systematic failure of a sampling method to represent its population
Bias Blinding Block Census Cluster sample Confounding Control group Convenience sample Designs Experiment Experimental units Factor Level Any systematic failure of a sampling method to represent its population
Cochrane Pregnancy and Childbirth Group Methodological Guidelines
 Cochrane Pregnancy and Childbirth Group Methodological Guidelines [Prepared by Simon Gates: July 2009, updated July 2012] These guidelines are intended to aid quality and consistency across the reviews
Cochrane Pregnancy and Childbirth Group Methodological Guidelines [Prepared by Simon Gates: July 2009, updated July 2012] These guidelines are intended to aid quality and consistency across the reviews
CLINICAL PROTOCOL DEVELOPMENT
 CLINICAL PROTOCOL DEVELOPMENT Clinical Protocol (1) Background/Justification --Where we are in the field --What the study will add that is important Objectives --Primary hypothesis --Secondary hypotheses
CLINICAL PROTOCOL DEVELOPMENT Clinical Protocol (1) Background/Justification --Where we are in the field --What the study will add that is important Objectives --Primary hypothesis --Secondary hypotheses
Instrumental Variables Estimation: An Introduction
 Instrumental Variables Estimation: An Introduction Susan L. Ettner, Ph.D. Professor Division of General Internal Medicine and Health Services Research, UCLA The Problem The Problem Suppose you wish to
Instrumental Variables Estimation: An Introduction Susan L. Ettner, Ph.D. Professor Division of General Internal Medicine and Health Services Research, UCLA The Problem The Problem Suppose you wish to
AP Statistics Exam Review: Strand 2: Sampling and Experimentation Date:
 AP Statistics NAME: Exam Review: Strand 2: Sampling and Experimentation Date: Block: II. Sampling and Experimentation: Planning and conducting a study (10%-15%) Data must be collected according to a well-developed
AP Statistics NAME: Exam Review: Strand 2: Sampling and Experimentation Date: Block: II. Sampling and Experimentation: Planning and conducting a study (10%-15%) Data must be collected according to a well-developed
How the ICH E9 addendum around estimands may impact our clinical trials
 Danish Society for Biopharmaceutical Statistics Copenhagen October 26, 2017 How the ICH E9 addendum around estimands may impact our clinical trials Frank Bretz (Novartis) ICH E9(R1) Expert Working Group
Danish Society for Biopharmaceutical Statistics Copenhagen October 26, 2017 How the ICH E9 addendum around estimands may impact our clinical trials Frank Bretz (Novartis) ICH E9(R1) Expert Working Group
Understanding noninferiority trials
 Review article http://dx.doi.org/10.3345/kjp.2012.55.11.403 Korean J Pediatr 2012;55(11):403-407 eissn 1738-1061 pissn 2092-7258 Understanding noninferiority trials Seokyung Hahn, PhD Department of Medicine,
Review article http://dx.doi.org/10.3345/kjp.2012.55.11.403 Korean J Pediatr 2012;55(11):403-407 eissn 1738-1061 pissn 2092-7258 Understanding noninferiority trials Seokyung Hahn, PhD Department of Medicine,
Designing Psychology Experiments: Data Analysis and Presentation
 Data Analysis and Presentation Review of Chapter 4: Designing Experiments Develop Hypothesis (or Hypotheses) from Theory Independent Variable(s) and Dependent Variable(s) Operational Definitions of each
Data Analysis and Presentation Review of Chapter 4: Designing Experiments Develop Hypothesis (or Hypotheses) from Theory Independent Variable(s) and Dependent Variable(s) Operational Definitions of each
Thursday, April 25, 13. Intervention Studies
 Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
Designing and Analyzing RCTs. David L. Streiner, Ph.D.
 Designing and Analyzing RCTs David L. Streiner, Ph.D. Emeritus Professor, Department of Psychiatry & Behavioural Neurosciences, McMaster University Emeritus Professor, Department of Clinical Epidemiology
Designing and Analyzing RCTs David L. Streiner, Ph.D. Emeritus Professor, Department of Psychiatry & Behavioural Neurosciences, McMaster University Emeritus Professor, Department of Clinical Epidemiology
Safeguarding public health Subgroup analyses scene setting from the EU regulators perspective
 Safeguarding public health Subgroup analyses scene setting from the EU regulators perspective The views expressed herein are not necessarily those of MHRA, EMA, EMA committees or their working parties.
Safeguarding public health Subgroup analyses scene setting from the EU regulators perspective The views expressed herein are not necessarily those of MHRA, EMA, EMA committees or their working parties.
Mapping the Informed Health Choices (IHC) Key Concepts (KC) to core concepts for the main steps of Evidence-Based Health Care (EBHC).
 KC No KC Short Title KC Statement EBHC concept EBHC sub concept EBHC statement EBHC Step Reported 1.01 Treatments can harm Treatments may be harmful Explain the use of harm/aetiologies for (rare) adverse
KC No KC Short Title KC Statement EBHC concept EBHC sub concept EBHC statement EBHC Step Reported 1.01 Treatments can harm Treatments may be harmful Explain the use of harm/aetiologies for (rare) adverse
Safeguarding public health Subgroup Analyses: Important, Infuriating and Intractable
 Safeguarding public health Subgroup Analyses: Important, Infuriating and Intractable The views expressed herein are not necessarily those of MHRA, EMA, EMA committees or their working parties. Rob Hemmings
Safeguarding public health Subgroup Analyses: Important, Infuriating and Intractable The views expressed herein are not necessarily those of MHRA, EMA, EMA committees or their working parties. Rob Hemmings
Modeling & Simulation to support evaluation of Safety and Efficacy of Drugs in Older Patients
 Modeling & Simulation to support evaluation of Safety and Efficacy of Drugs in Older Patients Eva Bredberg, Director Global Clinical Pharmacology, AstraZeneca On behalf of EFPIA EMA Geriatrics Workshop
Modeling & Simulation to support evaluation of Safety and Efficacy of Drugs in Older Patients Eva Bredberg, Director Global Clinical Pharmacology, AstraZeneca On behalf of EFPIA EMA Geriatrics Workshop
E11(R1) Addendum to E11: Clinical Investigation of Medicinal Products in the Pediatric Population Step4
 E11(R1) Addendum to E11: Clinical Investigation of Medicinal Products in the Step4 October 2017 International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use 1 Legal
E11(R1) Addendum to E11: Clinical Investigation of Medicinal Products in the Step4 October 2017 International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use 1 Legal
exposure/intervention
 Kathleen A. Kennedy, MD, MPH University of Texas Medical School at Houston Medical School Most clinical research evaluates an association between exposure/intervention outcome. 1 Does the investigator
Kathleen A. Kennedy, MD, MPH University of Texas Medical School at Houston Medical School Most clinical research evaluates an association between exposure/intervention outcome. 1 Does the investigator
Delfini Evidence Tool Kit
 General 1. Who is sponsoring and funding the study? What are the affiliations of the authors? Study Design Assessment Internal Validity Assessment Considerations: This can be helpful information and is
General 1. Who is sponsoring and funding the study? What are the affiliations of the authors? Study Design Assessment Internal Validity Assessment Considerations: This can be helpful information and is
Overview of Clinical Study Design Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine US National
 Overview of Clinical Study Design Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine US National Institutes of Health 2012 How Many Taken a similar course
Overview of Clinical Study Design Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine US National Institutes of Health 2012 How Many Taken a similar course
Design and analysis of clinical trials
 Design and analysis of clinical trials Lecture 3 1.Basic Design Considerations 2.Sample Size determinationa 3.Randomization 2018-01-31 Previous Lectures Definition of a clinical trial The drug development
Design and analysis of clinical trials Lecture 3 1.Basic Design Considerations 2.Sample Size determinationa 3.Randomization 2018-01-31 Previous Lectures Definition of a clinical trial The drug development
Evidence- and Value-based Solutions for Health Care Clinical Improvement Consults, Content Development, Training & Seminars, Tools
 Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
Lecture 2. Key Concepts in Clinical Research
 Lecture 2 Key Concepts in Clinical Research Outline Key Statistical Concepts Bias and Variability Type I Error and Power Confounding and Interaction Statistical Difference vs Clinical Difference One-sided
Lecture 2 Key Concepts in Clinical Research Outline Key Statistical Concepts Bias and Variability Type I Error and Power Confounding and Interaction Statistical Difference vs Clinical Difference One-sided
Thursday, April 25, 13. Intervention Studies
 Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
Intervention Studies Outline Introduction Advantages and disadvantages Avoidance of bias Parallel group studies Cross-over studies 2 Introduction Intervention study, or clinical trial, is an experiment
Designing Pharmacy Practice Research
 Designing Pharmacy Practice Research Ross T. Tsuyuki, BSc(Pharm), PharmD, MSc, FCHSP, FACC Professor of Medicine (Cardiology) Director, EPICORE Centre Faculty of Medicine and Dentistry University of Alberta
Designing Pharmacy Practice Research Ross T. Tsuyuki, BSc(Pharm), PharmD, MSc, FCHSP, FACC Professor of Medicine (Cardiology) Director, EPICORE Centre Faculty of Medicine and Dentistry University of Alberta
AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS
 AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS Susan S. Ellenberg, Ph.D. Perelman School of Medicine University of Pennsylvania FDA Clinical Investigator Course Silver Spring, MD November 14, 2018 OVERVIEW
AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS Susan S. Ellenberg, Ph.D. Perelman School of Medicine University of Pennsylvania FDA Clinical Investigator Course Silver Spring, MD November 14, 2018 OVERVIEW
Making comparisons. Previous sessions looked at how to describe a single group of subjects However, we are often interested in comparing two groups
 Making comparisons Previous sessions looked at how to describe a single group of subjects However, we are often interested in comparing two groups Data can be interpreted using the following fundamental
Making comparisons Previous sessions looked at how to describe a single group of subjects However, we are often interested in comparing two groups Data can be interpreted using the following fundamental
Clinical Trials. Susan G. Fisher, Ph.D. Dept. of Clinical Sciences
 Clinical Trials Susan G. Fisher, Ph.D. Dept. of Clinical Sciences When to Do a RCT Exposure of interest is modifiable and individuals are willing to relinquish control Legitimate uncertainty exists about
Clinical Trials Susan G. Fisher, Ph.D. Dept. of Clinical Sciences When to Do a RCT Exposure of interest is modifiable and individuals are willing to relinquish control Legitimate uncertainty exists about
Determining the size of a vaccine trial
 17th Advanced Vaccinology Course Annecy 26 th May 2016 Determining the size of a vaccine trial Peter Smith London School of Hygiene & Tropical Medicine Purposes of lecture Introduce concepts of statistical
17th Advanced Vaccinology Course Annecy 26 th May 2016 Determining the size of a vaccine trial Peter Smith London School of Hygiene & Tropical Medicine Purposes of lecture Introduce concepts of statistical
Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry
 Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Evidence Informed Practice Online Learning Module Glossary
 Term Abstract Associations Attrition Bias Background and Significance Baseline Basic Science Bias Blinding Definition An abstract is a summary of a research article. It usually includes the purpose, methods,
Term Abstract Associations Attrition Bias Background and Significance Baseline Basic Science Bias Blinding Definition An abstract is a summary of a research article. It usually includes the purpose, methods,
SHORTENING TIMELINES AND IMPROVING EFFICIENCY IN DRUG DEVELOPMENT -Seamless drug development paradigms
 SHORTENING TIMELINES AND IMPROVING EFFICIENCY IN DRUG DEVELOPMENT -Seamless drug development paradigms Claudia Filozof, MD PhD Executive Medical Director Covance Challenges in Clinical Development in NASH
SHORTENING TIMELINES AND IMPROVING EFFICIENCY IN DRUG DEVELOPMENT -Seamless drug development paradigms Claudia Filozof, MD PhD Executive Medical Director Covance Challenges in Clinical Development in NASH
DRAFT (Final) Concept Paper On choosing appropriate estimands and defining sensitivity analyses in confirmatory clinical trials
 DRAFT (Final) Concept Paper On choosing appropriate estimands and defining sensitivity analyses in confirmatory clinical trials EFSPI Comments Page General Priority (H/M/L) Comment The concept to develop
DRAFT (Final) Concept Paper On choosing appropriate estimands and defining sensitivity analyses in confirmatory clinical trials EFSPI Comments Page General Priority (H/M/L) Comment The concept to develop
EFFECTIVE MEDICAL WRITING Michelle Biros, MS, MD Editor-in -Chief Academic Emergency Medicine
 EFFECTIVE MEDICAL WRITING Michelle Biros, MS, MD Editor-in -Chief Academic Emergency Medicine Why We Write To disseminate information To share ideas, discoveries, and perspectives to a broader audience
EFFECTIVE MEDICAL WRITING Michelle Biros, MS, MD Editor-in -Chief Academic Emergency Medicine Why We Write To disseminate information To share ideas, discoveries, and perspectives to a broader audience
Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials
 Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials Dominik Heinzmann, PhD Global Development Team Leader HER2 Associate Director
Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials Dominik Heinzmann, PhD Global Development Team Leader HER2 Associate Director
Assignment #6. Chapter 10: 14, 15 Chapter 11: 14, 18. Due tomorrow Nov. 6 th by 2pm in your TA s homework box
 Assignment #6 Chapter 10: 14, 15 Chapter 11: 14, 18 Due tomorrow Nov. 6 th by 2pm in your TA s homework box Assignment #7 Chapter 12: 18, 24 Chapter 13: 28 Due next Friday Nov. 13 th by 2pm in your TA
Assignment #6 Chapter 10: 14, 15 Chapter 11: 14, 18 Due tomorrow Nov. 6 th by 2pm in your TA s homework box Assignment #7 Chapter 12: 18, 24 Chapter 13: 28 Due next Friday Nov. 13 th by 2pm in your TA
Evaluation Models STUDIES OF DIAGNOSTIC EFFICIENCY
 2. Evaluation Model 2 Evaluation Models To understand the strengths and weaknesses of evaluation, one must keep in mind its fundamental purpose: to inform those who make decisions. The inferences drawn
2. Evaluation Model 2 Evaluation Models To understand the strengths and weaknesses of evaluation, one must keep in mind its fundamental purpose: to inform those who make decisions. The inferences drawn
Evaluating and Interpreting Clinical Trials
 Article #2 CE Evaluating and Interpreting Clinical Trials Dorothy Cimino Brown, DVM, DACVS University of Pennsylvania ABSTRACT: For the practicing veterinarian, selecting the best treatment for patients
Article #2 CE Evaluating and Interpreting Clinical Trials Dorothy Cimino Brown, DVM, DACVS University of Pennsylvania ABSTRACT: For the practicing veterinarian, selecting the best treatment for patients
Statistical Considerations in Study Development: How to write good objectives!
 Statistical Considerations in Study Development: How to write good objectives! Emily Van Meter, PhD Assistant Professor, Division of Cancer Biostatistics University of Kentucky Markey Cancer Center UK
Statistical Considerations in Study Development: How to write good objectives! Emily Van Meter, PhD Assistant Professor, Division of Cancer Biostatistics University of Kentucky Markey Cancer Center UK
Statistical aspects of surgery in clinical trials. Laurence Collette, PhD Statistics Department, EORTC, Brussels (BE)
 Statistical aspects of surgery in clinical trials Laurence Collette, PhD Statistics Department, EORTC, Brussels (BE) Employee of EORTC, non profit organization No conflict of interest to declare Surgery
Statistical aspects of surgery in clinical trials Laurence Collette, PhD Statistics Department, EORTC, Brussels (BE) Employee of EORTC, non profit organization No conflict of interest to declare Surgery
Guidance Document for Claims Based on Non-Inferiority Trials
 Guidance Document for Claims Based on Non-Inferiority Trials February 2013 1 Non-Inferiority Trials Checklist Item No Checklist Item (clients can use this tool to help make decisions regarding use of non-inferiority
Guidance Document for Claims Based on Non-Inferiority Trials February 2013 1 Non-Inferiority Trials Checklist Item No Checklist Item (clients can use this tool to help make decisions regarding use of non-inferiority
Real-world data in pragmatic trials
 Real-world data in pragmatic trials Harold C. Sox,MD The Patient-Centered Outcomes Research Institute Washington, DC Presenter Disclosure Information In compliance with the accrediting board policies,
Real-world data in pragmatic trials Harold C. Sox,MD The Patient-Centered Outcomes Research Institute Washington, DC Presenter Disclosure Information In compliance with the accrediting board policies,
Glossary of Practical Epidemiology Concepts
 Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
BIOSTATISTICAL METHODS
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Designs on Micro Scale: DESIGNING CLINICAL RESEARCH THE ANATOMY & PHYSIOLOGY OF CLINICAL RESEARCH We form or evaluate a research or research
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Designs on Micro Scale: DESIGNING CLINICAL RESEARCH THE ANATOMY & PHYSIOLOGY OF CLINICAL RESEARCH We form or evaluate a research or research
Guidelines for Writing and Reviewing an Informed Consent Manuscript From the Editors of Clinical Research in Practice: The Journal of Team Hippocrates
 Guidelines for Writing and Reviewing an Informed Consent Manuscript From the Editors of Clinical Research in Practice: The Journal of Team Hippocrates 1. Title a. Emphasize the clinical utility of the
Guidelines for Writing and Reviewing an Informed Consent Manuscript From the Editors of Clinical Research in Practice: The Journal of Team Hippocrates 1. Title a. Emphasize the clinical utility of the
Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:
 Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:7332-7341 Presented by Deming Mi 7/25/2006 Major reasons for few prognostic factors to
Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:7332-7341 Presented by Deming Mi 7/25/2006 Major reasons for few prognostic factors to
Reflection paper on assessment of cardiovascular risk of medicinal products for the treatment of cardiovascular and metabolic diseases Draft
 1 2 3 21 May 2015 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Reflection paper on assessment of cardiovascular risk of medicinal products for the treatment of cardiovascular
1 2 3 21 May 2015 EMA/CHMP/50549/2015 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Reflection paper on assessment of cardiovascular risk of medicinal products for the treatment of cardiovascular
Introduction to Clinical Trials - Day 1 Session 2 - Screening Studies
 Introduction to Clinical Trials - Day 1 Session 2 - Screening Studies Presented July 23, 2018 Susanne J. May Department of Biostatistics University of Washington Daniel L. Gillen Department of Statistics
Introduction to Clinical Trials - Day 1 Session 2 - Screening Studies Presented July 23, 2018 Susanne J. May Department of Biostatistics University of Washington Daniel L. Gillen Department of Statistics
From single studies to an EBM based assessment some central issues
 From single studies to an EBM based assessment some central issues Doug Altman Centre for Statistics in Medicine, Oxford, UK Prognosis Prognosis commonly relates to the probability or risk of an individual
From single studies to an EBM based assessment some central issues Doug Altman Centre for Statistics in Medicine, Oxford, UK Prognosis Prognosis commonly relates to the probability or risk of an individual
Regulatory Hurdles for Drug Approvals
 Regulatory Hurdles for Drug Approvals William R. Hiatt, MD Professor of Medicine/Cardiology University of Colorado School of Medicine President, CPC Clinical Research 25 min Conflicts CPC Clinical Research
Regulatory Hurdles for Drug Approvals William R. Hiatt, MD Professor of Medicine/Cardiology University of Colorado School of Medicine President, CPC Clinical Research 25 min Conflicts CPC Clinical Research
PRECISION, CONFIDENCE, AND SAMPLE SIZE IN THE QUANTIFICATION OF AVIAN FORAGING BEHAVIOR
 Studies in Avian Biology No. 13:193-198, 1990. PRECISION, CONFIDENCE, AND SAMPLE SIZE IN THE QUANTIFICATION OF AVIAN FORAGING BEHAVIOR LISA J. PETIT, DANIEL R. PETIT, AND KIMBERLY G. SMITH Abstract. We
Studies in Avian Biology No. 13:193-198, 1990. PRECISION, CONFIDENCE, AND SAMPLE SIZE IN THE QUANTIFICATION OF AVIAN FORAGING BEHAVIOR LISA J. PETIT, DANIEL R. PETIT, AND KIMBERLY G. SMITH Abstract. We
CHAPTER 4 Designing Studies
 CHAPTER 4 Designing Studies 4.2 Experiments The Practice of Statistics, 5th Edition Starnes, Tabor, Yates, Moore Bedford Freeman Worth Publishers Experiments Learning Objectives After this section, you
CHAPTER 4 Designing Studies 4.2 Experiments The Practice of Statistics, 5th Edition Starnes, Tabor, Yates, Moore Bedford Freeman Worth Publishers Experiments Learning Objectives After this section, you
How to Interpret a Clinical Trial Result
 How to Interpret a Clinical Trial Result Edward V. Loftus, Jr., M.D. Professor of Medicine Division of Gastroenterology and Hepatology Mayo Clinic College of Medicine Rochester MN CP123456-1 Are results
How to Interpret a Clinical Trial Result Edward V. Loftus, Jr., M.D. Professor of Medicine Division of Gastroenterology and Hepatology Mayo Clinic College of Medicine Rochester MN CP123456-1 Are results
ICH E9(R1) Technical Document. Estimands and Sensitivity Analysis in Clinical Trials STEP I TECHNICAL DOCUMENT TABLE OF CONTENTS
 ICH E9(R1) Technical Document Estimands and Sensitivity Analysis in Clinical Trials STEP I TECHNICAL DOCUMENT TABLE OF CONTENTS A.1. Purpose and Scope A.2. A Framework to Align Planning, Design, Conduct,
ICH E9(R1) Technical Document Estimands and Sensitivity Analysis in Clinical Trials STEP I TECHNICAL DOCUMENT TABLE OF CONTENTS A.1. Purpose and Scope A.2. A Framework to Align Planning, Design, Conduct,
Type and quantity of data needed for an early estimate of transmissibility when an infectious disease emerges
 Research articles Type and quantity of data needed for an early estimate of transmissibility when an infectious disease emerges N G Becker (Niels.Becker@anu.edu.au) 1, D Wang 1, M Clements 1 1. National
Research articles Type and quantity of data needed for an early estimate of transmissibility when an infectious disease emerges N G Becker (Niels.Becker@anu.edu.au) 1, D Wang 1, M Clements 1 1. National
Estimands. EFPIA webinar Rob Hemmings, Frank Bretz October 2017
 Estimands EFPIA webinar Rob Hemmings, Frank Bretz October 2017 Why estimands? To explain which treatment effect is described to prescribers and other stakeholders. To align objectives with (design and)
Estimands EFPIA webinar Rob Hemmings, Frank Bretz October 2017 Why estimands? To explain which treatment effect is described to prescribers and other stakeholders. To align objectives with (design and)
Statistical Considerations: Study Designs and Challenges in the Development and Validation of Cancer Biomarkers
 MD-TIP Workshop at UVA Campus, 2011 Statistical Considerations: Study Designs and Challenges in the Development and Validation of Cancer Biomarkers Meijuan Li, PhD Acting Team Leader Diagnostic Devices
MD-TIP Workshop at UVA Campus, 2011 Statistical Considerations: Study Designs and Challenges in the Development and Validation of Cancer Biomarkers Meijuan Li, PhD Acting Team Leader Diagnostic Devices
Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) Additional considerations for cross-over trials
 Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) Additional considerations for cross-over trials Edited by Julian PT Higgins on behalf of the RoB 2.0 working group on cross-over trials
Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) Additional considerations for cross-over trials Edited by Julian PT Higgins on behalf of the RoB 2.0 working group on cross-over trials
(Regulatory) views on Biomarker defined Subgroups
 (Regulatory) views on Biomarker defined Subgroups Norbert Benda Disclaimer: Views expressed in this presentation are the author's personal views and not necessarily the views of BfArM Biomarker defined
(Regulatory) views on Biomarker defined Subgroups Norbert Benda Disclaimer: Views expressed in this presentation are the author's personal views and not necessarily the views of BfArM Biomarker defined
ACR OA Guideline Development Process Knee and Hip
 ACR OA Guideline Development Process Knee and Hip 1. Literature searching frame work Literature searches were developed based on the scenarios. A comprehensive search strategy was used to guide the process
ACR OA Guideline Development Process Knee and Hip 1. Literature searching frame work Literature searches were developed based on the scenarios. A comprehensive search strategy was used to guide the process
Unit 1 Exploring and Understanding Data
 Unit 1 Exploring and Understanding Data Area Principle Bar Chart Boxplot Conditional Distribution Dotplot Empirical Rule Five Number Summary Frequency Distribution Frequency Polygon Histogram Interquartile
Unit 1 Exploring and Understanding Data Area Principle Bar Chart Boxplot Conditional Distribution Dotplot Empirical Rule Five Number Summary Frequency Distribution Frequency Polygon Histogram Interquartile
HOW ARE NON-INFERIORITY MARGINS SELECTED IN NON- INFERIORITY TRIALS AND HOW DO THEY VARY WITHIN AND ACROSS FOUR MAJOR DISEASE DOMAINS?
 HOW ARE NON-INFERIORITY MARGINS SELECTED IN NON- INFERIORITY TRIALS AND HOW DO THEY VARY WITHIN AND ACROSS FOUR MAJOR DISEASE DOMAINS? A REVIEW by Ling Yang A thesis submitted to Johns Hopkins University
HOW ARE NON-INFERIORITY MARGINS SELECTED IN NON- INFERIORITY TRIALS AND HOW DO THEY VARY WITHIN AND ACROSS FOUR MAJOR DISEASE DOMAINS? A REVIEW by Ling Yang A thesis submitted to Johns Hopkins University
Glossary. Ó 2010 John Wiley & Sons, Ltd
 Glossary The majority of the definitions within this glossary are based on, but are only a selection from, the comprehensive list provided by Day (2007) in the Dictionary of Clinical Trials. We have added
Glossary The majority of the definitions within this glossary are based on, but are only a selection from, the comprehensive list provided by Day (2007) in the Dictionary of Clinical Trials. We have added
Selection and estimation in exploratory subgroup analyses a proposal
 Selection and estimation in exploratory subgroup analyses a proposal Gerd Rosenkranz, Novartis Pharma AG, Basel, Switzerland EMA Workshop, London, 07-Nov-2014 Purpose of this presentation Proposal for
Selection and estimation in exploratory subgroup analyses a proposal Gerd Rosenkranz, Novartis Pharma AG, Basel, Switzerland EMA Workshop, London, 07-Nov-2014 Purpose of this presentation Proposal for
Choosing Life: Empowerment, Action, Results! CLEAR Menu Sessions. Substance Use Risk 2: What Are My External Drug and Alcohol Triggers?
 Choosing Life: Empowerment, Action, Results! CLEAR Menu Sessions Substance Use Risk 2: What Are My External Drug and Alcohol Triggers? This page intentionally left blank. What Are My External Drug and
Choosing Life: Empowerment, Action, Results! CLEAR Menu Sessions Substance Use Risk 2: What Are My External Drug and Alcohol Triggers? This page intentionally left blank. What Are My External Drug and
GSK Medicine: Study Number: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
ICH E9 (R1) Addendum on Estimands and Sensitivity Analysis
 ICH E9 (R1) Addendum on Estimands and Sensitivity Analysis Rob Hemmings Mouna Akacha MHRA + ICH E9 (R1) Expert Working Group Novartis 1 Disclaimer (I) Not a statistical topic This impacts every clinical
ICH E9 (R1) Addendum on Estimands and Sensitivity Analysis Rob Hemmings Mouna Akacha MHRA + ICH E9 (R1) Expert Working Group Novartis 1 Disclaimer (I) Not a statistical topic This impacts every clinical
STAT 113: PAIRED SAMPLES (MEAN OF DIFFERENCES)
 STAT 113: PAIRED SAMPLES (MEAN OF DIFFERENCES) In baseball after a player gets a hit, they need to decide whether to stop at first base, or try to stretch their hit from a single to a double. Does the
STAT 113: PAIRED SAMPLES (MEAN OF DIFFERENCES) In baseball after a player gets a hit, they need to decide whether to stop at first base, or try to stretch their hit from a single to a double. Does the
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
Non-inferiority trials and switch from non-inferiority to superiority. D Costagliola U 943 INSERM and UPMC Paris 06
 Non-inferiority trials and switch from non-inferiority to superiority D Costagliola U 943 INSERM and UPMC Paris 06 References l ICH E 9 et E10 l Points to consider on biostatistical methodological issues
Non-inferiority trials and switch from non-inferiority to superiority D Costagliola U 943 INSERM and UPMC Paris 06 References l ICH E 9 et E10 l Points to consider on biostatistical methodological issues
Nonallergic Rhinitis: Developing Drug Products for Treatment Guidance for Industry
 Nonallergic Rhinitis: Developing Drug Products for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Nonallergic Rhinitis: Developing Drug Products for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Κριτική αξιολόγηση τυχαιοποιημένης κλινικής δοκιμής Άρης Λιάκος, MD MSc Ειδικευόμενος Παθολογίας, Υποψήφιος Διδάκτωρ
 Αριστοτέλειο Πανεπιστήμιο Θεσσαλονίκης Σχολή Επιστημών Υγείας Τμήμα Ιατρικής Πρόγραμμα Μεταπτυχιακών Σπουδών «Ερευνητική Μεθοδολογία στην Ιατρική και τις Επιστήμες Υγείας» Μάθημα: Ιατρική Βασισμένη στην
Αριστοτέλειο Πανεπιστήμιο Θεσσαλονίκης Σχολή Επιστημών Υγείας Τμήμα Ιατρικής Πρόγραμμα Μεταπτυχιακών Σπουδών «Ερευνητική Μεθοδολογία στην Ιατρική και τις Επιστήμες Υγείας» Μάθημα: Ιατρική Βασισμένη στην
DRAFT GUIDANCE. This guidance document is being distributed for comment purposes only.
 Inborn Errors of Metabolism That Use Dietary Management: Considerations for Optimizing and Standardizing Diet in Clinical Trials for Drug Product Development Guidance for Industry DRAFT GUIDANCE This guidance
Inborn Errors of Metabolism That Use Dietary Management: Considerations for Optimizing and Standardizing Diet in Clinical Trials for Drug Product Development Guidance for Industry DRAFT GUIDANCE This guidance
Critical appraisal: Systematic Review & Meta-analysis
 Critical appraisal: Systematic Review & Meta-analysis Atiporn Ingsathit MD.PhD. Section for Clinical Epidemiology and biostatistics Faculty of Medicine Ramathibodi Hospital Mahidol University What is a
Critical appraisal: Systematic Review & Meta-analysis Atiporn Ingsathit MD.PhD. Section for Clinical Epidemiology and biostatistics Faculty of Medicine Ramathibodi Hospital Mahidol University What is a
10 Intraclass Correlations under the Mixed Factorial Design
 CHAPTER 1 Intraclass Correlations under the Mixed Factorial Design OBJECTIVE This chapter aims at presenting methods for analyzing intraclass correlation coefficients for reliability studies based on a
CHAPTER 1 Intraclass Correlations under the Mixed Factorial Design OBJECTIVE This chapter aims at presenting methods for analyzing intraclass correlation coefficients for reliability studies based on a
Types of Data. Systematic Reviews: Data Synthesis Professor Jodie Dodd 4/12/2014. Acknowledgements: Emily Bain Australasian Cochrane Centre
 Early Nutrition Workshop, December 2014 Systematic Reviews: Data Synthesis Professor Jodie Dodd 1 Types of Data Acknowledgements: Emily Bain Australasian Cochrane Centre 2 1 What are dichotomous outcomes?
Early Nutrition Workshop, December 2014 Systematic Reviews: Data Synthesis Professor Jodie Dodd 1 Types of Data Acknowledgements: Emily Bain Australasian Cochrane Centre 2 1 What are dichotomous outcomes?
Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias.
 Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias. In the second module, we will focus on selection bias and in
Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias. In the second module, we will focus on selection bias and in
