Clinical and genetic basis of congenital myasthenic syndromes
|
|
|
- Melvyn Wilson
- 6 years ago
- Views:
Transcription
1 VIEW AND REVIEW DOI: / X Clinical and genetic basis of congenital myasthenic syndromes Bases clínicas e genéticas das síndromes miastênicas congênitas Paulo Victor Sgobbi de Souza 1, Gabriel Novaes de Rezende Batistella 1, Valéria Cavalcante Lino 1, Wladimir Bocca Vieira de Rezende Pinto 1, Marcelo Annes 1, Acary Souza Bulle Oliveira 1 ABSTRACT Neuromuscular junction disorders represent a wide group of neurological diseases characterized by weakness, fatigability and variable degrees of appendicular, ocular and bulbar musculature involvement. Its main group of disorders includes autoimmune conditions, such as autoimmune acquired myasthenia gravis and Lambert-Eaton syndrome. However, an important group of diseases include congenital myasthenic syndromes with a genetic and sometimes hereditary basis that resemble and mimick many of the classic myasthenia neurological manifestations, but also have different presentations, which makes them a complex clinical, therapeutic and diagnostic challenge for most clinicians. We conducted a wide review of congenital myasthenic syndromes in their clinical, genetic and therapeutic aspects. Keywords: myasthenia gravis; congenital myasthenic syndromes, genetics. RESUMO Distúrbios da junção neuromuscular representam um grupo amplo de doenças neruológicas caracterizadas por fraqueza, fadigabilidade e graus variados de envolvimento das musculaturas apendicular, ocular e bulbar. Os principais grupos de doenças deste grupo incluem condições auto-imunes, como a miastenia gravis auto-imune adquirida e a síndrome de Lambert-Eaton. Entretanto, um outro grupo importante de doenças incluem as sindromes miastênicas congênitas com uma base genética e eventualmente hereditária que lembra e mimetiza muitas das manifestações neurológicas clássicas das miastenias, mas também se apresentam de diferentes formas tornando um desafio clínico, terapêutico e diagnóstico complexo para a maioria dos clínicos. Realizamos ampla revisão sobre as síndromes miastênicas congênitas em seus aspectos clínicos, genéticos e terapêuticos. Palavras-chave: miastenia gravis; síndromes miastênicas congênitas, genética. In 1895, Friedrich Jolly using Greek prefixes for muscle and weakness created the term myasthenia and used the Latin word gravis to describe the severity of weakness that can often lead patients to death 1. Myasthenia is described as a clinical group of conditions affecting neuromuscular transmission and can occur in the context of acquired and hereditary congenital conditions. Congenital myasthenic syndromes (CMS) comprise a heterogeneous group of rare inherited diseases in which the neuromuscular transmission in the motor plate is compromised by one or more genetic pathophysiological specific mechanisms 2. Congenital myasthenic syndromes can be classified according to the pattern of inheritance, based on the altered protein involved in the motor plate, or by taking into account the site at the neuromuscular junction (pre-synaptic, synaptic, or postsynaptic) involved with the dysfunction (Table 1) 3. Although most commonly considered in the differential diagnosis of early-onset hypotonia in the infant and neonate, CMS must be considered in a wide group of clinical and electroneuromyographic scenarios, even including cases with juvenile and adult-onset symptomatology 2,3. It is essential to consider CMS as an important differential diagnosis, as many presentations represent well-responsive therapeutic forms and different inheritance patterns are involved, emphasizing the crucial role of genetic counseling. DIAGNOSIS AND CLINICAL FEATURES At present there are no well-defined diagnostic criteria for CMS. Congenital myasthenic syndromes should be suspected in cases of: (i) early-onset fatigable muscle weakness mainly 1 Universidade Federal de São Paulo, Departamento de Neurologia e Neurocicurgia, Divisão de Doenças Neuromusculares, São Paulo SP, Brasil. Correspondence: Wladimir Bocca Vieira de Rezende Pinto; Departamento de Neurologia e Neurocicurgia, Universidade Federal de São Paulo (UNIFESP); Rua Estado de Israel, 899; São Paulo SP, Brasil. wladimirbvrpinto@gmail.com Conflict of interest: There is no conflict of interest to declare. Received 09 September 2015; Received in final form 05 March 2016; Accepted 03 May
2 Table 1. Classification of congenital myasthenic syndromes (CMS) related to pattern of inheritance and molecular targets at the neuromuscular junction. I. Pattern of inheritance Autosomal dominant (gain-of-function) Slow-channel syndrome, SNAP25B*, SYT2* Autosomal recessive (loss-of-function) All other subtypes II. Site of defect and molecular targets at the neuromuscular junction Presynaptic defects ChAT deficiency, SNAP25B deficiency, synaptotagmin-2 deficiency Primary deficiency, slow-channel syndrome (CHRNA1, CHRNB, CHRND, CHRNE, CHRNG), Acetylcholine receptor defects fast-channel syndrome (CHRNA, CHRND, CHRNE) Synaptic basal lamina defects Acetylcholinesterase deficiency (ColQ), β2-laminin deficiency Endplate development and maintenance congenital defects Metabolic and mitochondrial disorders Others *extremely rare presentations. Agrin deficiency, MuSK deficiency, LRP4 deficiency, Dok-7 deficiency, rapsyn deficiency, COL13A1 mutations Congenital disorders of glycosylation, SLC25A1 gene mutations Congenital myopathies with secondary neuromuscular transmission compromise (MTM1, RYR1, DNM2, TPM3, BIN1); PREPL deletion; plectin deficiency involving ocular, bulbar and proximal limb musculature (generally varying from birth to late childhood); (ii) a positive family history of a specific disorder or sometimes only the history of a hypotonic infant; (iii) clinical and neurophysiological myasthenic findings with a negative antibody testing profile; (iv) electromyography (EMG) studies showing decremental responses of 10% or more in the amplitude or in the quarter of the area from the first evoked compound motor action potential (CMAP), or single-fiber EMG studies compatible with a neuromuscular junction dysfunction; and (v) the presence of a specific clinical syndromic phenotype (i.e. Escobar syndrome, Pierson syndrome) 2,3,4. However, in some congenital myasthenic syndromes, the onset of clinical manifestations may be late with involvement in adolescence or adulthood, other family members may not have been affected by the disease and electromyography changes may not be present in all muscles or occur intermittently 4. Therefore, although some clinical, therapeutic and neurophysiological clues allow a high clinical suspicion for each CMS subtype (Tables 2 and 3), in many cases of rapsyn deficiency, primary deficiency of endplate acetylcholine receptor and fast-channel syndrome, there are frequently no specific hallmarks in presentations 2,3,4. The list of differential diagnoses of CMS is huge and includes some of the following conditions: congenital myopathies, congenital muscular dystrophy, limb-girdle muscular dystrophy, mitochondrial myopathies, facioscapulohumeral dystrophy, myotonic dystrophy and autoimmune myasthenia gravis, motor neuron disease and peripheral neuropathies (Table 4) 2. Epidemiology The prevalence of CMS is very difficult to estimate due to the clinical variability of cases and the fact that many cases have no specific etiologic diagnosis or are undiagnosed. Since they are rare medical conditions in which definite diagnosis rests on clinical, electromyography and specific genetic testing, few data are available. Furthermore, there are few series of patients where this complete diagnostic profile has been established, and most of the current knowledge has been obtained by reports of isolated case reports 4. A study in the UK estimated that the prevalence of CMS with a defined genetic diagnosis is approximately 9.2 cases per million children under 18 years old in the population 5. There are also some ethnic variations in the genetic diagnosis of CMS with some mutations being more common in some populations, such as the 1293insG mutation in the gene CHRNE coding the epsilon subunit of the acetylcholine receptor, more frequently found in families from north Africa 6, and the 1267delG mutation also in the CHRNE in gypsy families from southeast Europe 7. An important epidemiological profile on CMS was obtained in a study performed at the Mayo Clinic. Most cases occured as a consequence of postsynaptic defects (68%), and basal lamina defects (13.7%), development and maintenance of the end plate defects (12.5%), pure presynaptic defects (5.9-8%) and congenital myopathies with secondary neuromuscular junction transmission defects (0.3%) also represent other rare congenital myasthenic syndromes. Thus, postsynaptic forms represent up to 75-80% of all CMS cases 3,4,8. In southern Brazil, in the state of Paraná, a minimum prevalence of 0.18/ of CMS is estimated 9. Pathophysiology To understand the pathophysiological mechanisms involved in CMS, it is essential to recognize key aspects of the neuromuscular junction structure and function. The neuromuscular junction has three basic components: (i) the presynaptic nerve terminal, where there is the biosynthesis process, storage and release of acetylcholine, the main neurotransmitter involved in primary muscular contraction process; (ii) the synaptic space or synaptic cleft, where acetylcholine is released by the presynaptic nerve terminal and where there is a complex network of local proteins responsible for maintenance of the structure of the neuromuscular junction; and (iii) the postsynaptic muscle membrane, where there are acetylcholine receptors responsible for the action potential deflagration, endplate Souza PVSS et al. Congenital myasthenic syndromes 751
3 Table 2. Main diagnostic clues to guide differential diagnosis of congenital myasthenic syndromes (CMS) subtypes. 2,4 I. Clinical clues of CMS subtypes Early-onset phenotypes (neonatal, lactancy, early infancy) Late-onset phenotypes (late infancy, adolescence, juvenile, adult) LGMD-like phenotype Apneic episodes Laryngeal stridor and vocal cord palsy Epilepsy with intellectual disability Cerebellar ataxia Pupillary defects Ptosis Ophthalmoparesis (± strabismus) Facial palsy Cervical weakness Arthrogryposis multiplex congenital-like phenotype and early-onset joint contractures Dysmorphic features Congenital malformations Anoxic encephalopathy-like phenotype Epidermolysis bullosa simplex II. Histopathological clues of CMS subtypes Tubular aggregates in the sarcoplasmic reticulum Vacuolar autophagic myopathy LMGD: limb-girdle muscular dystrophy. ChAT deficiency, synaptotagmin-2 deficiency, acetylcholinesterase, slow-channel syndrome, fast-channel syndrome, agrin deficiency, LRP4 deficiency, MuSK deficiency, Dok-7 deficiency, rapsyn deficiency, GFPT1, ALG2, ALG14, GMPPB, PREPL deficiency, plectin deficiency, SCN4A, COL13A1 Acetylcholinesterase deficiency (variants), slow-channel syndrome, agrin deficiency, SCN4A GFPT1, LRP4 deficiency, plectin deficiency ChAT deficiency, SCN4A Dok-7 deficiency DPAGT1 deficiency SNAP25B ColQ mutations Primary AChR deficiency, Dok-7 deficiency, SNAP25B, agrin deficiency, MuSK deficiency, rapsyn deficiency, PREPL, congenital myopathies, COL13A1 Rapsyn deficiency, primary AChR deficiency, MuSK deficiency, Dok-7 deficiency, congenital myopathies Agrin deficiency, MuSK deficiency, Dok-7 deficiency, rapsyn deficiency, PREPL, congenital myopathies Slow-channel syndrome, Dok-7 deficiency, COL13A1 Rapsyn deficiency, ChAT deficiency, AChR deficiency (CHRND, CHRNG), SNAP25B, synaptotagmin-2 Rapsyn deficiency, β2-laminin deficiency, COL13A1 Pierson syndrome (β2-laminin: ocular malformation, congenital nephrosis); rapsyn deficiency; GFPT1 deficiency; SLC25A1 (optic nerve hypoplasia, corpus callosum agenesis) Rapsyn deficiency, SCN4A Plectin deficiency GFPT1, DPAGT1 and ALG2-related CMS subtypes GFPT1 and DPAGT1-related subtypes potential and the acetylcholinesterase enzyme, involved with the breakdown of acetylcholine and subsequent restoration of the resting potential of the membrane postsynaptic potential 8,10,11,12. Mutations in the genes encoding proteins related to any one of these three components of the neuromuscular junction could give rise to different CMS phenotypes. Currently, major defects involved in the etiology of CMS occur 2,4 : (A) in the presynaptic terminal; (B) associated with the synaptic basal lamina membrane; (C) the acetylcholine receptor; (D) shortcomings in the maintenance and development of the neuromuscular junction; (E) congenital defects in glycosylation; and (F) other sites and mechanisms (Figure). Clinical and genetic basis of congenital myasthenic syndromes Currently, more than 20 different genes are involved as monogenic causes of CMS, with an increasing number of new discoveries every year driven by next-generation sequencing with complete exome sequencing (whole-exome sequencing). This has clarified the pathophysiological mechanisms of this spectrum of disorders, not restricted to the neuromuscular junction structures and pointing to new opportunities and approaches for future therapeutic modalities 4,13,14. PRE-SYNAPTIC SYNDROMES Deficiency of choline acetyltransferase (ChAT) (OMIM #254210) Presynaptic defects represent less than 6% of all CMS cases. The enzyme choline acetyltransferase (encoded by the CHAT gene, 10q11.23), located in the presynaptic terminal region of the axon of the lower motor neuron, is involved in the biosynthesis of the neurotransmitter acetylcholine from acetyl-coenzyme A (acetyl-coa) and choline 2,4. The severity of ChAT deficiency depends on the type and site of mutation affecting the enzyme structure, the 752 Arq Neuropsiquiatr 2016;74(9):
4 Table 3. Therapeutic clues in the differential diagnosis of congenital myasthenic syndromes (CMS) subtypes 2,4. Quinidine-responsive phenotype Slow-channel syndrome Fluoxetine-responsive phenotype Slow-channel syndrome Acetazolamide-responsive phenotype Sodium channel Amifampridine-responsive phenotype (3,4-diaminopyridine) Albuterol-responsive phenotype Salbutamol-responsive phenotype Ephedrine-responsive phenotype Pyridostigmine-responsive phenotype Pyridostigmine-avoidance phenotype (clinical worsen) Amifampridine-avoidance phenotype (clinical worsen) Fluoxetine-avoidance phenotype (clinical worsen) Quinidine-avoidance phenotype (clinical worsen) Clinical refractory phenotype Primary AChR deficiency, SNAP25, fast channel syndrome, MuSK, rapsyn, ALG2, ALG14, GFPT1, DPAGT1, COL13A1 Dok-7, Primary AChR deficiency, MuSK (partial), acetylcholinesterase deficiency Rapsyn, acetylcholinesterase, primary AChR deficiency, MuSK, DPAGT1 (partial), COL13A1 Laminin-β2 deficiency, agrin, acetylcholinesterase, Dok-7 GFPT1, sodium channel (SCN4A), centronuclear congenital myopathy, ChAT deficiency, fast-channel syndrome, AChR deficiency, paucity of synaptic vesicles and reduced quantal release (partial), congenital myopathies (partial: centronuclear myopathy), PREPL (transient) Acetylcholinesterase deficiency, Laminin-β2 deficiency, slow-channel syndrome, LRP4, Dok-7 and MuSK deficiencies; agrin deficiency Acetylcholinesterase deficiency, slow channel syndrome, LRP4 mutations Fast channel syndrome Fast channel syndrome Plectin deficiency (pyridostigmine, amifampridine), laminin-β2, plectin, acetylcolinesterase (pyridostigmine), DPAGT1 (partial), GMPPB Table 4. Main differential diagnosis of congenital myasthenic syndromes (CMS) of early- and late-onset presentations. I. Early-onset (neonatal, infant) Motor neuron disease: spinal muscular atrophy (types I-III) Congenital muscular dystrophy, infantile myotonic dystrophy type I, congenital myopathies (central core disease, centronuclear, nemaline, myotubular, congenital fiber-type disproportion) Neonatal myasthenia gravis, seropositive and seronegative acquired autoimmune myasthenia gravis, infantile botulism Mitochondrial myopathies Toxic exogenous poisoning Miscellaneous: Möbius syndrome, brainstem anomalies, congenital fibrosis of extraocular muscles II. Late-onset (teenager, young adult) Motor neuron Disease Metabolic myopathies: mitochondrial myopathies, muscle lipidosis LGMDs, FSHD, congenital myopathies Seropositive and seronegative acquired autoimmune myasthenia gravis Toxic exogenous poisoning Chronic fatigue syndrome FSHD: facioscapulohumeral muscular dystrophy; LGMD: limb-girdle muscular dystrophy 2,4. most severe clinical manifestations being found in mutations that affect the catalytic domain of the enzyme or the binding site to the substrate 2,4,8. Mutations in the CHAT gene produce a classical CMS phenotype with early onset in the neonatal period and associated with serious and threatening episodes of apnea 2,3,4. Episodes of apnea may have abrupt onset and can be triggered after episodes of physical or emotional stress or illnesses in patients with few or no prior myasthenic symptoms 2,15,16. Variable ptosis without ophthalmoparesis is common. Some patients may present with apnea and marked hypotonia at birth with difficulty in maintaining spontaneous breathing and they can also develop secondary brain atrophy due to hypoxemia in the neonatal period, while other patients are normal at birth and rarely begin to manifest apneic episodes during childhood or early adolescence. Recurrent acute attacks of respiratory failure lasting for weeks have also been described. Severe presentations with arthrogryposis multiplex-like phenotype have also been described 2,15,16. Souza PVSS et al. Congenital myasthenic syndromes 753
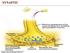
5 Rapsyn Dok-7 MuSK Lrp4 AchR Agrin ColQ AChE Plectin 1 Nerve terminal 1 5 Presynaptic vesicles Schwann cell 8 Laminin-β2 MASC ErbBR Na* channels (Nav1:4) SNAP25B, synaptotagmin-2 Muscle cystoskeleton ChAT Rough endoplasmic reticulum & ribosomes Perlecan 2 Muscle Fiber 4 3 SARCOMERE Figure. Representation of the main pathophysiological mechanisms involved in different forms of CMS: (1) acetylcholine biosynthesis defects and vesicular transport and fusion defects; (2) acetylcholinesterase deficiency; (3) acetylcholine receptor defects; (4) agrin deficiency; (5) congenital disorders of glycosylation; (6) channelopathies; (7) congenital myopathies with secondary neuromuscular transmission defects; and (8) mitochondrial dysfunction. Legends: ChAT: choline acetyltransferase; ErbBR: epidermal growth factor receptor; MASC: muscle-associated specificity component; AChE: acetylcholinesterase; Lrp4: low-density lipoprotein receptor-related protein 4; ColQ: collagen-like tail subunit or acetylcholinesterase-associated collagen; MuSK: muscle-specific tyrosine kinase receptor; Dok-7: downstream of tyrosine kinase 7 2,3,4. An indicator to consider CMS by mutations in the CHAT gene is the presence of a decrease in the amplitude of the CMAP associated with a potential endplate 50% below the baseline (normal decrease is less than 30%) followed by a very slow recovery 5-10 minutes after subtetanic stimulation 2. CMAP amplitude decrease is not a pathognomonic finding of ChAT deficiency, being present in other CMS. However, the recovery is usually complete in less than five minutes. 2,4 In such cases, the amplitudes of the CMAP, endplate potential and miniature endplate potentials are normal at rest, decreasing after a repetitive stimulation test at 10 Hz for five minutes and marked reduction of CMAPs with slow recovery to the basal line in 10 to 15 minutes 3. Deficiency of SNAP25B (OMIM #616330) The proteins of the SNARE class (soluble N-ethylmaleimidesensitive factor attachment protein receptors) constitute the core of the intracellular machinery of fusion of synaptic vesicles 17. SNAP25 (synaptosomal-associated protein 25-Kd, encoded by the SNAP25 gene, 20p12.2) and the protein syntaxin bind to target cell membranes and are termed t-snares, which have an important role in protein and vesicles anchoring, initiation of intracellular vesicles and the rapid triggering the exocytosis process 17. Deficiency of SNAP25B was reported with an autosomal dominant pattern of CMS presenting in an 11-year-old African-American female patient with a history of intrauterine hypotonia, cyanosis at birth, delayed psychomotor development, multiple joint contractures, muscle weakness, fatigable ptosis, associated with ataxia and early childhood epilepsy, whose biomolecular studies revealed a decrease in the amount of acetylcholine released from the presynaptic nerve terminal from each motor nerve impulse 4,18. Deficiency of synaptotagmin-2 (OMIM #616040) Synaptotagmin-2 protein is a synaptic vesicle protein that acts as a calcium sensor during the neuromuscular transmission process. 19 Mutations have been described in two families with a motor neuropathy phenotype characterized by hollow feet and hammer toes with diminished or abolished reflexes associated with a fatigability of proximal and distal muscles with weakness. Electromyography tests demonstrated low amplitude CMAP with a marked increase in the amplitude of the CMAP after performing a fast exercise, mimicking the findings of Lambert-Eaton myasthenic syndrome 2,20. This presentation has been linked to autosomal dominant inheritance presentations of CMS linked to SYT2 gene (1q32.1). 754 Arq Neuropsiquiatr 2016;74(9):
6 SYNDROMES ASSOCIATED WITH THE SYNAPTIC BASAL LAMINA Deficiency of endplate acetylcholinesterase by mutations in COLQ (OMIM #603034) The acetylcholinesterase in the neuromuscular plate is an asymmetrical enzyme which may be constituted by one, two or three catalytic subunits tetramers that are anchored in the basal membrane with a triple-stranded collagen tail called ColQ (acetylcholinesterase-associated collagen). Mutations in the COLQ gene (3p25.1) promote a prolongation of the residence time of acetylcholine in the neuromuscular junction, producing a desensitization of muscle acetylcholine receptor and a secondary myasthenic syndrome 21,22. This presentation corresponds to 13,4% of all CMS cases 3. The clinical picture begins at birth or in early childhood as an autosomal recessive CMS, characterized by progressive muscle weakness affecting all voluntary muscles and can spare the extrinsic ocular muscles. There is a direct correlation of site of mutations and clinical presentations and course. Most cases associated with C-terminal mutations have a light clinical course and late-onset of symptoms. Most cases related to N-terminal mutations or rod-domain of ColQ have early-onset severe presentations. Diagnosis is essential as genetic counseling represents an important clinical step and as some cases present good clinical responses to the use of albuterol or ephedrin 21,22. Electrophysiological studies typically confirm the presence of a decremental response pattern of the repetitive CMAPs greater than 10% 23. Deficiency of laminin-β2 (OMIM #609049) Laminin-β2 protein is encoded by the LAMB2 gene (3p21.31) and expressed in the basal lamina of the endplate terminal of the neuromuscular junction in the eyes and kidneys participating in the differentiation process of the pre-synaptic region as well as the alignment of the presynaptic motor terminal with the post-synaptic region in the muscle membrane 3. Only one case has been reported of autosomal recessive congenital myasthenic syndrome resulting from deficiency of laminin-β2 (less than 0,5% of all CMS cases) in a 20-year-old female patient with eye and kidney malformations, compatible with Pierson syndrome or microcoria-congenital nephrotic syndrome. Classically it represents a pyridostigmine-refractory form of CMS with predominant proximal limb weakness, where ocular abnormalities (including nonreactive fixed narrowing of the pupils, hypoplasia of the iris and the ciliary body, and lenticonus posterior) and congenital kidney disease (including neonatal-onset congenital nephrotic syndrome, early-onset end-stage renal disease and diffuse mesangial sclerosis) represent major cardinal signs. Another allelic presentation may be as hereditary nephrotic syndrome type 5 (MIM #614199) 2,24. Defects of the acetylcholine receptor The nicotinic acetylcholine receptor is a pentameric complex comprising four transmembrane subunits, composed of α2βδε in the adult neuromuscular plate and α2βδγ in the fetal neuromuscular plate, as well in extrajunctional regions 2,4. The genes encoding the α (CHRNA1 gene, 2q31.1), δ (CHRND gene, 2q37.1) and γ (CHRNG gene, 2q37.1) chains are located on the long arm of chromosome 2 at different genetic loci, while the genes encoding the β chain (CHRNB1 gene) and ε (CHRNE gene) are located in different loci on the short arm of chromosome 17p13.2 2,4. In southern Brazil, as a consequence of founder effect mutations of Hispanic settlers origin, CHRNE gene recessive mutations represent the main cause of CMS 9. Mutations in genes encoding the acetylcholine receptor subunits can produce CMS by means of three mechanisms: (1) reducing the number of acetylcholine receptors in the postsynaptic membrane; (2) extended opening called the slow-channel syndrome; (3) short opening of the receptors known as the fast-channel syndrome. 2,4 Primary deficiency of the acetylcholine receptor Primary deficiency of the acetylcholine receptor represents the most common form of CMS (34 to 50% of all cases) and results from mutations in genes coding any of the subunits of the acetylcholine receptor, with those related to the ε subunit being the most common and severe 2,4. The clinical picture is characterized by ptosis, refractory marked ophthalmoparesis and severe muscle weakness of the limbs. There is generally a partially responsive pattern to pyridostigmine, amifampridine and albuterol use in clinical practice 2,4,25. Mutations in both alleles of CHRNA1, CHRNB or CHRND genes are usually incompatible with life, resulting in death in the fetal period 2,4,25. Slow-channel syndrome Slow-channel syndrome is part of the kinetic defects of the acetylcholine receptor due to dominant mutations in the domains or pore receptor ligands of the receptorbinding portion. It resembles, in many aspects, the acetylcholinesterase deficiency. This typically presents with symptoms in the first decade of life with prominent and severe involvement of the scapular, cervical and dorsal forearm (wrist and finger extensors) muscle groups, sparing extrinsic ocular muscles and only exceptionally presenting with mild asymmetric ptosis 2,26,27. Atypical late-onset presentations have also been described as mimicking generalized autoimmune myasthenia gravis with associated mild late-onset myopathic changes and rarely with mild to moderately raised serum creatine kinase levels. However, the good therapeutic response profile to fluoxetine and quinidine and unresponsiveness to amifampridine and pyridostigmine distinguishes this condition from acquired forms 26,27,28. Souza PVSS et al. Congenital myasthenic syndromes 755
7 Fast-channel syndrome Fast-channel syndrome arises from autosomal recessive mutations in different domains of the acetylcholine receptor subunits and clinically mimicks a typical autoimmune acquired myasthenia gravis starting in the first decade of life with good clinical response to treatment with pyridostigmine and amiframpidine. Fluoxetin and quinidine use must be avoided. Allelic conditions include the multiple pterygium syndrome (lethal type) 2,3,29. Defects in the development and maintenance of the endplate Congenital myasthenic syndromes can arise by the effect of mutations in genes related to the development and maintenance of the endplate, including AGRN (agrin; 1p36.33), MUSK (9q31.3), LRP4 (11p11.2), DOK7 (4p16.3), RAPSN (11p11.2) and COL13A1 (10q22.1) genes. The agrin protein is secreted in the terminal nerve synaptic cleft and binds to the LRP4 (low density lipoprotein receptorrelated protein 4) protein of the postsynaptic membrane, originating a secondary complex that activates a MuSK (muscle skeletal receptor tyrosine kinase) receptor with tyrosine kinase activity, allowing the phosphorylation of Dok-7 (downstream of tyrosine kinase 7) and rapsyn activation (Figure). Thus, modulatory effects occur and the concentration of acetycholine in the postsynaptic membrane receptor alters the expression of nuclear genes related to the differentiation of the endplate and promoting the maintenance of the postsynaptic membrane 1,2. Agrin deficiency (OMIM #615120) Agrin is a proteoglycan secreted in the synaptic basal laminae by the terminal nerve, phosphorylating and activating MuSK by the LRP4 receptor. Mutations of the AGRN (1p36.33) gene, coding agrin, originates a typical late-onset CMS phenotype starting between the fourth and fifth decades of life and an early-onset variant in infants and neonates. The typical form originates ptosis and mild weakness in the face and proximal limbs weakness, starting from childhood and resulting from homozygous missense mutations (Gly1709Arg) and other types with more severe phenotypes (Gln353X and Val1725Phe). The subtype of early-onset CMS relates to generalized amyotrophy in lower limbs and slowly progressive weakness with lipossubstitution of the posterior compartment of the leg, sparing cranial nerves and axial muscles and without ophthalmoparesis (despite ptosis being frequently found). There is a good response to the use of ephedrine and generally failure to the use of amifampridine and pyridostigmine 3,30,31. LRP4 deficiency (OMIM #616304) The LRP4 deficiency was described in a 14-year-old patient with neonatal respiratory failure, delayed motor development and muscle weakness and fatigability of pelvic and shoulder girdle musculature. The clinical phenotype results from missense mutations Glu1233Lys and Arg1277His. Allelic conditions include sclerosteosis type 2 and Cenani-Lenz syndactyly syndrome 2,32. MuSK deficiency (OMIM #616325) The MuSK deficiency is extremely rare, results from mutations of the MUSK gene (9q31.3) and manifests through respiratory failure with neonatal ptosis or in the infant with proximal appendiceal involvement, facial and ocular and bulbar variable 33. MuSK is involved in end-plate maturation and maintenance processes, and aids rapsyn proper function and its complex interactions with AChR in the postsynaptic membrane. The allelic condition is the fetal akinesia deformation sequence 2,33. Dok-7 deficiency (OMIM #254300) Dok-7 deficiency represents less than 10% of all CMS cases. It represents the second most common cause of CMS in southern Brazil 9. Different mutations have been described in the DOK7 gene (4p16.3), the most common being the c.1124_1127duptgcc mutation related to autosomal recessive CMS. The clinical spectrum is very wide, occurring with wrist weakness and lower cervical and facial involvement and some forms with bulbar and ophthalmoparesis involvement. It represents an important differential diagnosis of limb-girdle muscular dystrophies (LGMD) and initiates predominance of type 1 fibers, atrophy of type 2 fibers, and target fiber formations 34,35. An LGMD-like pattern of weakness, ptosis and mild facial weakness with moderate to severe bulbar symptoms and laryngeal stridor and vocal cord palsy occurs in mutations of the last codon. Other cases present as lower limb dominant progressive amyotrophy. Muscle biopsy discloses generally unspecific findings. Clinical worsening is common after pyridostigmine use and clinical improvement frequently observed after ephedrine, salbutamol and albuterol use 34,35,36. Rapsyn deficiency (OMIM #616326) Rapsyn (receptor-associated protein of the synapse, 43-Kd) deficiency results from homozygous mutations in the RAPSN gene (11p11.2) and represents around 15% of all CMS cases. This autosomal recessive CMS manifests most commonly in infants during the first year of life (rarely in scholars and young adults), the most common pattern in one third of patients being an arthrogryposis phenotype at birth, multiple congenital contractures and other congenital malformations, frequently in the context of patients with post-anoxic encephalopathy after myasthenic crisis due to infectious complications 2,3,4. It is remarkable that normal adduction and abduction of the eye with strabismus and ophthalmoparesis is found in less than 25% of cases. There is some regional segregation of mutations in India and Europe (Asn88Lys mutation). 756 Arq Neuropsiquiatr 2016;74(9):
8 Another important finding relates to clinical-genetic correlations: the c.38a>g mutation presents with a milder phenotype with ptosis, prognathism, facial and masticatory muscle weakness and nasal voice; the N88K mutation represents most neonatal and early-infancy-onset cases; A38G mutation commonly presents with ptosis, hypernasal speech, facial weakness and prominent masticatory dysfunction 37. The allelic condition leads to to fetal akinesia deformation sequence. Thus, the presence of an early-onset phenotype mimicking chonic nonprogressive hypoxic-ischaemic clinical findings with neuromuscular junction dysfunction signs should raise the possibility of rapsyn deficiency. COL13A1-related CMS (OMIM #616720) A new autosomal recessive very early-onset CMS has been recently described resulting from homozygous mutations in the COL13A1 gene (10q22.1), coding the alpha-1 chain of the nonfibrillar transmembrane collagen type XIII, involved in autocrine control of the development and maturation of the neuromuscular junction 38. Clinical presentation is broad and includes ptosis, proximal and distal limb hypotonia, delayed motor milestones, poor head control, feeding difficulties, childhood-onset spinal rigidity, respiratory insufficiency, dyspnea on exertion with exercise intolerance, dysphagia, gastroesophageal reflux and recurrent lower respiratory tract infections. Mild dysmorphic features have also been reported, including pes cavus without early joint contractures, low-set ears, micrognathia, high-arched palate and pectus carinatum. Neurophysiological studies show significant decremental response on repetitive nerve stimulation testing 38. Congenital disorders of glycosylation Congenital disorders of glycosylation (CDGs) relate to different systemic and neurometabolic syndromes with different congenital malformations, subtypes of congenital muscular dystrophy and congenital myasthenic syndromes by mutations in the genes encoding the enzymes GFPT1 (glutamine:fructose-6-phosphate amidotransferase 1, 2p13.3), DPAGT1 (dolichyl-phosphate UDP-N-acetylglucosamine N-acetylglucosamine-1-phosphotransferase, 11q23.3) ALG2 (alpha-1,3-mannosyltransferase, 9q22.33) and ALG14 (UDP-Nacetylglucosaminyltransferase, 1p21.3). The histopathological presence of muscle tubular aggregates in the setting of characteristic neurophysiological findings in pre- and postsynaptic impaired neuromuscular junction must indicate genetic evaluation for CDG-related CMS 2,3. Recently, a fifth glycosylation gene called GMPPB (GDP-mannose pyrophosphorylase B, 3p21.31) has also been linked to new CMS phenotypes 39. GFPT1 deficiency (OMIM #610542) The GFPT1 (glutamine:fructose-6-phosphate amidotransferase-1) enzyme regulates glucose entering in the hexosamine pathway for the formation of precursor components for processes of O-linked and N-linked glycosylation of basic proteins. Mutations in the GFPT1 gene (2p13.3) represents less than 3% of all CMS cases. Typically, disability originates slowly progressive muscle weakness with an LGMD-like phenotype responsive to pyridostigmine use and tubular aggregates of sarcoplasmic reticulum in muscle biopsy. Some variants showed multiple arthrogryposis at birth, slow progression until eight years old with severe dysphagia and severe myopathy with autophagic vacuoles in muscle specimens. Ptosis and respiratory insufficiency are rarely found. Raised serum CK is found in around 25% of cases 2,3,40. Deficiency of DPAGT1 (OMIM #614750) The enzyme DPAGT1 (dolichyl-phosphate N-acetylglucosamine phosphotransferase) catalyzes the first step in glycosylation of N-linked proteins. The resulting clinical spectrum of mutations in the DPAGT1 (11q23.3) gene initiates moderate or severe muscle weakness and cognitive impairment, with a therapeutic profile unresponsive to pyridostigmine and amifampridine, and partially responsive to salbutamol. The most common mutations involve His375Tyr and Val264Met. Allelic conditions include CDG type Ij. Muscle biopsy shows small tubular aggregates, fiber type disproportion with small caliber type 1 fibers and autophagic vacuolar myopathy, resembling similar patterns to STIM1-related myopathies 3,41,42. ALG2 and ALG14 deficiencies (OMIM #616228; OMIM #616227) The ALG2 (asparagine-linked glycosylation 2) enzyme participates directly in the second and third steps of N-glycosylation. ALG14 forms a complex with the multiglucosyltransferase ALG13. DPAGT1 participates indirectly in the first step of N-glycosylation. Both syndromes arising from ALG2 (9q22.33) and ALG14 (1p21.3) genes mutations have an early-onset LGMD-like CMS starting in the preschooler with typical myasthenic syndrome complaints. ALG14 deficiency presents with a similar pattern to ALG2-related CMS, despite the absence of tubular aggregates in muscle biopsy. Pyridostigmine may benefit some patients. Allelic conditions include CDG type Ii. Raised serum CK levels can be observed in both cases 2,43. Deficiency of GMPPB GMPPB gene (3p21.31) mutations have recently been described as a cause of autosomal recessive CMS. Cases present with a notably exclusive appendicular (mainly proximal) weakness phenotype without facial and eye muscle involvement. They also present with typical myopathic changes in different laboratory evaluations, including high serum creatine kinase levels and unspecific myopathic changes in muscle biopsy and muscle MRI studies 39. Mutations in this same gene also give rise to a broad clinical spectrum with congenital muscular dystrophies from the dystroglycanopathy family, including allelic Souza PVSS et al. Congenital myasthenic syndromes 757
9 conditions such as autosomal recessive congenital muscular dystrophies from the dystroglycanopathy group (types A, B and C). Despite its rarity, GMPPB-related myasthenic syndromes should be considered in the setting of a nearly proximal appendicular myopathic patient in whom therapeutic strategies have failed or clinical outcomes have unexpectedly worsened 39. Other myasthenic syndromes PREPL Deletion Syndrome (OMIM #606407) The PREPL (prolyl endopeptidase-like) protein, coded by the PREPL gene (2p21), is an essential activator of clathrin adapter protein associated with type 1 (AP1), involved in vesicular transport and filling with acetylcholine and other neurotransmitters. The hypotonia-cystinuria syndrome is caused by deletions in the SLC3A1 gene and recessive mutations in the PREPL gene, originating cystinuria type A, deficiency of growth hormone, ptosis, and limb, facial and bulbar weakness, and neonatal hypotonia. The symptoms are moderate and transiently responsive to pyridostigmine during childhood 2,4,44. Deficiency of plectin Mutations in the PLEC gene (8q24.3), coding plectin 1, initiate a rare complex spectrum of clinical manifestations, involving epidermolysis bullosa simplex with nail dystrophy, limb-girdle muscular dystrophy type 2Q and congenital myasthenic syndrome. The neuromuscular phenotypes develop in patients with epidermolysis bullosa in childhood that, in the course of life, evolves into a progressive proximal myopathic phenotype refractory to pyridostigmine and decremental response pattern in neurophysiological studies 2,45. Defects in sodium channels (SCN4A) (OMIM #614198) An extremely rare form of CMS (less than 0,5% of all cases) was originally associated with a 20-year-old female patient with brief and abrupt attacks of muscle weakness (including bulbar palsy) and respiratory failure from birth that led to hypoxic-ischemic encephalopathy resulting from Val1442Glu mutation in the SCN4A gene (17q23.3), resulting in dysfunction of neuromuscular transmission in the unexcitability rest state 3,45. Each acute attack typically lasts from three to 30 minutes, similar to choline acetyltransferase deficiency. Allelic conditions include hyperkalemic periodic paralysis type 2, hypokalemic periodic paralysis type 2, atypical acetazolamide-responsive myotonia congenita and paramyotonia congenita 4,46. Congenital myopathies with secondary defects of neuromuscular transmission Congenital myopathies represent an extremely rare cause of congenital myasthenic syndrome-like phenotype (less than 0,5% of all causes). There are several reports in the literature of congenital myopathies with mutations that can lead to secondary impairment of the neuromuscular junction from school age to young adult, notably MTM1 (coding myotubularin, Xq28), RYR1 (coding ryanodine receptor 1, 19q13.2), DNM2 (coding dynamin 2, 19p13.2), TPM3 (coding tropomyosin 3, 1q21.3) and BIN1 (coding bridging integrator 1 or amphiphysin II, 2q14.3). This results in variable combinations of myasthenic syndrome (with ptosis, ophthalmoparesis and facial paresis), exercise intolerance, variable partial responsiveness to piridostigmin or corticosteroids (especially in centronuclear myopathies) and variable decremental responses (19-35% from baseline) or non-existent response in neurophysiological studies. Muscle biopsy in such cases usually shows atrophy of type 1 fibers with occasional loss of mitochondria and central myofibrillar degeneration 2,3,47. It is essential to highlight that most genes related to these phenotypes are involved with different neuromuscular phenotypes, including nemaline myopathy, centronuclear myopathy, CAP myopathy, fiber-type disproportion congenital myopathy, lethal congenital contracture syndrome type 5 and Charcot- Marie-Tooth disease spectrum. Mutations in the SLC25A1 gene The SLC25A1 (solute carrier family 25, mitocondrial carrier; citrate transporter, member 1) protein mediates mitochondrial citrate/isocitrate shuttle promoting cytosolic malate re-entry into the citric acid cycle and the movement of citrate across the mitochondrial inner membrane. Mutations in the SLC25A1 gene (22q11.21) initiate cognitive impairment, myasthenic symptoms, hypoplasia of the optic nerve, corpus callosum agenesis and eventually secondary 2-hydroxyglutaric aciduria. 2,48 An allelic condition includes the autosomal recessive combined D-2- and L-2- hydroxyglutaric aciduria (OMIM #615182). Thus, a CMS complex phenotype linked with different central nervous system malformations should make clinicians aware of the possibility of SLC25A1 gene mutations. In conclusion, CMS represents a genetically heterogeneous and important differential diagnosis group of early-onset peripheral hypotonic disorders. As a potentially treatable group of neuromuscular diseases, clinicians should be aware of this possibility when facing a hypotonic baby or infant with ophthalmoparesis, eyelid ptosis, facial palsy, recurrent unprovoked apneic episodes, dysphagia, dysphonia and early-onset distal joint contractures, sometimes mimicking congenital arthrogryposis multiplex. Late-onset presentations of CMS may also be considered, especially in cases of refractory seronegative myasthenia gravis, highly suggestive phenotypes or positive family history of myasthenia. 758 Arq Neuropsiquiatr 2016;74(9):
10 References 1. Berrih-Aknin S, Le Panse R. Myasthenia gravis: a comprehensive review of immune dysregulation and etiological mechanisms. J Autoimmun. 2014;52: doi: /j.jaut Engel AG, Shen XM, Selcen D, Sine SM. Congenital myasthenic syndromes: pathogenesis, diagnosis, and treatment. Lancet Neurol. 2015;14(4): doi: /s (14) Engel AG. Congenital myasthenic syndromes. In: Katirji B, Kaminski HJ, Ruff RL, editors. Neuromuscular disorders in clinical practice. New York: Springer; p Rodríguez Cruz PM, Palace J, Beeson D. Inherited disorders of the neuromuscular junction: an update. J Neurol. 2014;261(11): doi: /s Parr JR, Andrew MJ, Finnis M, Beeson D, Vincent A, Jayawant S. How common is childhood myasthenia? The UK incidence and prevalence of autoimmune and congenital myasthenia. Arch Dis Child. 2014;99(6): doi: /archdischild Richard P, Gaudon K, Haddad H, Ammar AB, Genin E, Bauché S et al. The CHRNE 1293insG founder mutation is a frequent cause of congenital myasthenia in North Africa. Neurology. 2008;71(24): doi: /01.wnl b 7. Abicht A, Stucka R, Karcagi V, Herczegfalvi A, Horváth R, Mortier W et al. A common mutation (epsilon1267delg) in congenital myasthenic patients of Gypsy ethnic origin. Neurology 1999;53(7): doi: /wnl Lorenzoni PJ, Scola RH, Kay CS, Werneck LC. Congenital myasthenic syndrome: a brief review. Pediatr Neurol. 2012;46(3): doi: /j.pediatrneurol Mihaylova V, Scola RH, Gervini B, Lorenzoni PJ, Kay CK, Werneck LC et al. Molecular characterisation of congenital myasthenic syndromes in Southern Brazil. J Neurol Neurosurg Psychiatry. 2010;81(9): doi: /jnnp Conti-Fine BM, Milani M, Kaminski HJ. Myasthenia gravis: past, present and future. J Clin Invest. 2006;116(11): doi: /jci Meriggioli MN, Sanders DB. Autoimmune myasthenia gravis: emerging clinical and biological heterogeneity. Lancet Neurol. 2009;8(5): doi: /s (09) Pinto WBVR, Souza PVS, Oliveira ASB. Normal muscle structure, growth, development and regeneration. Curr Rev Musculoskelet Med. 2015;8(2): doi: /s x 13. Das AS, Agamanolis DP, Cohen BH. Use of next-generation sequencing as a diagnostic tool for congenital myasthenic syndrome. Pediatr Neurol. 2014;51(5): doi: /j.pediatrneurol Tei S, Ishii HT, Mitsuhashi H, Ishiura S. Antisense oligonucleotidemediated exon skipping of CHRNA1 pre-mrna as potential therapy for Congenital Myasthenic Syndromes. Biochem Biophys Res Commun. 2015;461(3): doi: /j.bbrc Kraner S, Laufenberg I, Strassburg HM, Sieb JP, Steinlein OK. Congenital myasthenic syndrome with episodic apnea in patients homozygous for a CHAT missense mutation. Arch Neurol. 2003;60(5): doi: /archneur Ohno K, Tsujino A, Brengman JM, Harper CM, Bajzer Z, Udd B et al. Choline acetyltransferase mutations cause myasthenic syndrome associated with episodic apnea in humans. Proc Natl Acad Sci USA. 2001;98(4): doi: /pnas Mohrmann R, de Wit H, Connell E, Pinheiro PS, Leese C, Bruns D et al. Synaptotagmin interaction with SNAP-25 governs vesicle docking, priming, and fusion triggering. J Neurosci. 2013;33(36): doi: /jneurosci Shen XM, Selcen D, Brengman J, Engel AG. Mutant SNAP25B causes myasthenia, cortical hyperexcitability, ataxia, and intellectual disability. Neurology. 2014;83(24): doi: /wnl Herrmann DN, Horvath R, Sowden JE, Gonzalez M, Sanchez- Mejias A, Guan Z et al. Synaptotagmin 2 mutations cause an autosomal-dominant form of lambert-eaton myasthenic syndrome and nonprogressive motor neuropathy. Am J Hum Genet. 2014;95(3): doi: /j.ajhg Baker K, Gordon SL, Grozeva D, Kogelenberg M, Roberts NY, Pike M et al. Identification of a human synaptotagmin-1 mutation that perturbs synaptic vesicle cycling. J Clin Invest. 2015;125(4): doi: /jci Ohno K, Brengman J, Tsujino A, Engel AG. Human endplate acetylcholinesterase deficiency caused by mutations in the collagen-like tail subunit (ColQ) of the asymmetric enzyme. Proc Natl Acad Sci USA. 1998;95(16): doi: /pnas Mihaylova V, Müller JS, Vilchez JJ, Salih MA, Kabiraj MM, D Amico A et al. Clinical and molecular genetic findings in COLQ-mutant congenital myasthenic syndromes. Brain. 2008;131(3): doi: /brain/awm Lorenzoni PJ, Scola RH, Gervini BL, Kay CSK, Werneck LC. Electrophysiological study in synaptic congenital myasthenic syndrome: end-plate acetylcholinesterase deficiency. Arq Neuropsiquiatr. 2009;67(2B): doi: /s x Maselli RA, Ng JJ, Anderson JA, Cagney O, Arredondo J, Williams C et al. Mutations in LAMB2 causing a severe form of synaptic congenital myasthenic syndrome. J Med Genet. 2009;46(3): doi: /jmg Ohno K, Quiram P, Milone M, Wang HL, Harper MC, Pruitt JN 2nd et al. Congenital myasthenic syndromes due to heteroallelic nonsense/ missense mutations in the acetylcholine receptor ε subunit gene: identification and functional characterization of six new mutations. Hum Mol Genet. 1997;6(5): doi: /hmg/ Ohno K, Hutchinson DO, Milone M, Brengman JM, Bouzat C, Sine SM et al. Congenital myasthenic syndrome caused by prolonged acetylcholine receptor channel openings due to a mutation in the M2 domain of the ε subunit. Proc Natl Acad Sci USA. 1995;92(3): doi: /pnas Ohno K, Wang HL, Milone M, Bren N, Brengman JM, Nakano S et al. Congenital myasthenic syndrome caused by decreased agonist binding affinity due to a mutation in the acetylcholine receptor ε subunit. Neuron. 1996;17(1): doi: /s (00) Chaouch A, Müller JS, Guergueltcheva V, Dusl M, Schara U, Rakocević-Stojanović V et al. A retrospective clinical study of the treatment of slow-channel congenital myasthenic syndrome. J Neurol. 2012;259(3): doi: /s Shen XM, Brengman JM, Edvardson S, Sine SM, Engel AG. Highly fatal fast-channel syndrome caused by AChR ε subunit mutation at the agonist binding site. Neurology. 2012;79(5): doi: /wnl.0b013e31825b5bda 30. Nicole S, Chaouch A, Torbergsen T, Bauché S, de Bruyckere E, Fontenille MJ et al. Agrin mutations lead to a congenital myasthenic syndrome with distal muscle weakness and atrophy. Brain. 2014;137(9): doi: /brain/awu Huzé C, Bauché S, Richard P, Chevessier F, Goillot E, Gaudon K et al. Identification of an agrin mutation that causes congenital myasthenia and affects synapse function. Am J Hum Genet. 2009;85(2): doi: /j.ajhg Ohkawara B, Cabrera-Serrano M, Nakata T, Milone M, Asai N, Ito K et al. LRP4 third β-propeller domain mutations cause novel congenital myasthenia by compromising agrin-mediated MuSK signaling in a position-specific manner. Hum Mol Genet. 2014;23(7): doi: /hmg/ddt578 Souza PVSS et al. Congenital myasthenic syndromes 759
Congenital Myasthenic Syndromes or Inherited Disorders of Neuromuscular Transmission: Recent Discoveries and Open Questions
 Congenital Myasthenic Syndromes or Inherited Disorders of Neuromuscular Transmission: Recent Discoveries and Open Questions Sophie Nicole, Yoshiteru Azuma, Stéphanie Bauché, Bruno Eymard, Hanns Lochmuller,
Congenital Myasthenic Syndromes or Inherited Disorders of Neuromuscular Transmission: Recent Discoveries and Open Questions Sophie Nicole, Yoshiteru Azuma, Stéphanie Bauché, Bruno Eymard, Hanns Lochmuller,
Neonatal Hypotonia. Encephalopathy acute No encephalopathy. Neurology Chapter of IAP
 The floppy infant assumes a frog legged position. On ventral suspension, the baby can not maintain limb posture against gravity and assumes the position of a rag doll. Encephalopathy acute No encephalopathy
The floppy infant assumes a frog legged position. On ventral suspension, the baby can not maintain limb posture against gravity and assumes the position of a rag doll. Encephalopathy acute No encephalopathy
The Therapy of Congenital Myasthenic Syndromes
 Neurotherapeutics: The Journal of the American Society for Experimental NeuroTherapeutics The Therapy of Congenital Myasthenic Syndromes Andrew G. Engel Department of Neurology, Mayo Clinic, Rochester,
Neurotherapeutics: The Journal of the American Society for Experimental NeuroTherapeutics The Therapy of Congenital Myasthenic Syndromes Andrew G. Engel Department of Neurology, Mayo Clinic, Rochester,
Diseases of Muscle and Neuromuscular Junction
 Diseases of Muscle and Neuromuscular Junction Diseases of Muscle and Neuromuscular Junction Neuromuscular Junction Muscle Myastenia Gravis Eaton-Lambert Syndrome Toxic Infllammatory Denervation Atrophy
Diseases of Muscle and Neuromuscular Junction Diseases of Muscle and Neuromuscular Junction Neuromuscular Junction Muscle Myastenia Gravis Eaton-Lambert Syndrome Toxic Infllammatory Denervation Atrophy
Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report
 Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report Roshanak Jazayeri, MD, PhD Assistant Professor of Medical Genetics Faculty of Medicine, Alborz University of Medical Sciences
Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report Roshanak Jazayeri, MD, PhD Assistant Professor of Medical Genetics Faculty of Medicine, Alborz University of Medical Sciences
Neonatal Hypotonia Guideline Prepared by Dan Birnbaum MD August 27, 2012
 Neonatal Hypotonia Guideline Prepared by Dan Birnbaum MD August 27, 2012 Hypotonia: reduced tension or resistance to range of motion Localization can be central (brain), peripheral (spinal cord, nerve,
Neonatal Hypotonia Guideline Prepared by Dan Birnbaum MD August 27, 2012 Hypotonia: reduced tension or resistance to range of motion Localization can be central (brain), peripheral (spinal cord, nerve,
Stanley Iyadurai, PhD MD. Assistant Professor of Neurology/Neuromuscular Medicine Nationwide Children s Hospital Myology Course 2015
 1 Stanley Iyadurai, PhD MD Assistant Professor of Neurology/Neuromuscular Medicine Nationwide Children s Hospital Myology Course 2015 Motor unit motor neuron, its axon, and nerve terminals, and muscle
1 Stanley Iyadurai, PhD MD Assistant Professor of Neurology/Neuromuscular Medicine Nationwide Children s Hospital Myology Course 2015 Motor unit motor neuron, its axon, and nerve terminals, and muscle
Congenital Myasthenic Syndromes with Predominant Limb Girdle Weakness
 Journal of Neuromuscular Diseases 2 (2015) S21 S29 DOI 10.3233/JND-150098 IOS Press Research Report S21 Congenital Myasthenic Syndromes with Predominant Limb Girdle Weakness Teresinha Evangelista a, Mike
Journal of Neuromuscular Diseases 2 (2015) S21 S29 DOI 10.3233/JND-150098 IOS Press Research Report S21 Congenital Myasthenic Syndromes with Predominant Limb Girdle Weakness Teresinha Evangelista a, Mike
Congenital myasthenic syndromes: pathogenesis, diagnosis, and treatment
 Lancet Neurol 2015; 14: 420 34 This online publication has been corrected. The corrected version first appeared at thelancet.com/ neurology on March 26, 2015 Department of Neurology (A G Engel MD, X-M
Lancet Neurol 2015; 14: 420 34 This online publication has been corrected. The corrected version first appeared at thelancet.com/ neurology on March 26, 2015 Department of Neurology (A G Engel MD, X-M
Neuromuscular Junction Testing ELBA Y. GERENA MALDONADO, MD ACTING ASSISTANT PROFESSOR UNIVERSITY OF WASHINGTON MEDICAL CENTER
 Neuromuscular Junction Testing ELBA Y. GERENA MALDONADO, MD ACTING ASSISTANT PROFESSOR UNIVERSITY OF WASHINGTON MEDICAL CENTER Objectives Neurophysiology Electrodiagnostic Evaluation Clinical Application
Neuromuscular Junction Testing ELBA Y. GERENA MALDONADO, MD ACTING ASSISTANT PROFESSOR UNIVERSITY OF WASHINGTON MEDICAL CENTER Objectives Neurophysiology Electrodiagnostic Evaluation Clinical Application
A nomenclature and classification for the congenital myasthenic syndromes: preparing for FAIR data in the genomic era
 Thompson et al. Orphanet Journal of Rare Diseases (2018) 13:211 https://doi.org/10.1186/s13023-018-0955-7 RESEARCH A nomenclature and classification for the : preparing for FAIR data in the genomic era
Thompson et al. Orphanet Journal of Rare Diseases (2018) 13:211 https://doi.org/10.1186/s13023-018-0955-7 RESEARCH A nomenclature and classification for the : preparing for FAIR data in the genomic era
A rare case of muscular dystrophy with POMT2 and FKRP gene mutation. Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband
 A rare case of muscular dystrophy with POMT2 and FKRP gene mutation Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband Index : Congenital muscular dystrophy (CMD) Dystroglycanopathies Walker-Warburg
A rare case of muscular dystrophy with POMT2 and FKRP gene mutation Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband Index : Congenital muscular dystrophy (CMD) Dystroglycanopathies Walker-Warburg
Movement Disorders. Psychology 372 Physiological Psychology. Background. Myasthenia Gravis. Many Types
 Background Movement Disorders Psychology 372 Physiological Psychology Steven E. Meier, Ph.D. Listen to the audio lecture while viewing these slides Early Studies Found some patients with progressive weakness
Background Movement Disorders Psychology 372 Physiological Psychology Steven E. Meier, Ph.D. Listen to the audio lecture while viewing these slides Early Studies Found some patients with progressive weakness
Synapse Formation. Steven McLoon Department of Neuroscience University of Minnesota
 Synapse Formation Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Midterm Exam Monday, Nov 13 9:30-11:30am Bring a #2 pencil!! 2 Course News Lecture schedule: Mon (Oct 31)
Synapse Formation Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Midterm Exam Monday, Nov 13 9:30-11:30am Bring a #2 pencil!! 2 Course News Lecture schedule: Mon (Oct 31)
Peripheral neuropathies, neuromuscular junction disorders, & CNS myelin diseases
 Peripheral neuropathies, neuromuscular junction disorders, & CNS myelin diseases Peripheral neuropathies according to which part affected Axonal Demyelinating with axonal sparing Many times: mixed features
Peripheral neuropathies, neuromuscular junction disorders, & CNS myelin diseases Peripheral neuropathies according to which part affected Axonal Demyelinating with axonal sparing Many times: mixed features
Hole s Human Anatomy and Physiology Eleventh Edition. Mrs. Hummer. Chapter 9 Muscular System
 Hole s Human Anatomy and Physiology Eleventh Edition Mrs. Hummer Chapter 9 Muscular System 1 Chapter 9 Muscular System Skeletal Muscle usually attached to bones under conscious control striated Three Types
Hole s Human Anatomy and Physiology Eleventh Edition Mrs. Hummer Chapter 9 Muscular System 1 Chapter 9 Muscular System Skeletal Muscle usually attached to bones under conscious control striated Three Types
CONGENITAL MYASTHENIC SYNDROMES: PROGRESS OVER THE PAST DECADE
 INVITED REVIEW ABSTRACT: Congenital myasthenic syndromes (CMS) stem from defects in presynaptic, synaptic basal lamina, and postsynaptic proteins. The presynaptic CMS are associated with defects that curtail
INVITED REVIEW ABSTRACT: Congenital myasthenic syndromes (CMS) stem from defects in presynaptic, synaptic basal lamina, and postsynaptic proteins. The presynaptic CMS are associated with defects that curtail
Neuromuscular Junction Disorders
 Neuromuscular Junction Disorders HMS Child Neurology CME Course September 7 th, 2017 Amanda C. Guidon, MD Neuromuscular Diagnostic Center Director Myasthenia Gravis Clinic Massachusetts General Hospital
Neuromuscular Junction Disorders HMS Child Neurology CME Course September 7 th, 2017 Amanda C. Guidon, MD Neuromuscular Diagnostic Center Director Myasthenia Gravis Clinic Massachusetts General Hospital
Disorders of Muscle. Disorders of Muscle. Muscle Groups Involved in Myopathy. Needle Examination of EMG. History. Muscle Biopsy
 Disorders of Muscle Disorders of Muscle Zakia Bell, M.D. Associate Professor of Neurology and Physical Medicine & Rehabilitation Virginia Commonwealth University Cardinal symptom of diseases of the muscle
Disorders of Muscle Disorders of Muscle Zakia Bell, M.D. Associate Professor of Neurology and Physical Medicine & Rehabilitation Virginia Commonwealth University Cardinal symptom of diseases of the muscle
Neuromuscular in the Pediatric Clinic: Recognition and Referral
 Neuromuscular in the Pediatric Clinic: Recognition and Referral Matthew Harmelink, MD Assistant Professor, Pediatric Neurology Medical College of Wisconsin Objectives: 1. Understand common presentations
Neuromuscular in the Pediatric Clinic: Recognition and Referral Matthew Harmelink, MD Assistant Professor, Pediatric Neurology Medical College of Wisconsin Objectives: 1. Understand common presentations
Myasthenia gravis. David Hilton-Jones Oxford Neuromuscular Centre
 Myasthenia gravis David Hilton-Jones Oxford Neuromuscular Centre SWIM, Taunton, 2018 Myasthenia gravis Autoimmune disease Nature of Role of thymus Myasthenia gravis Autoimmune disease Nature of Role of
Myasthenia gravis David Hilton-Jones Oxford Neuromuscular Centre SWIM, Taunton, 2018 Myasthenia gravis Autoimmune disease Nature of Role of thymus Myasthenia gravis Autoimmune disease Nature of Role of
Evaluation of the Hypotonic Infant and Child
 Evaluation of the Hypotonic Infant and Child Basil T. Darras, M.D. Neuromuscular Program Boston Children s Hospital Harvard Medical School Boston, MA, USA Classification and General Clinical Evaluation
Evaluation of the Hypotonic Infant and Child Basil T. Darras, M.D. Neuromuscular Program Boston Children s Hospital Harvard Medical School Boston, MA, USA Classification and General Clinical Evaluation
variant led to a premature stop codon p.k316* which resulted in nonsense-mediated mrna decay. Although the exact function of the C19L1 is still
 157 Neurological disorders primarily affect and impair the functioning of the brain and/or neurological system. Structural, electrical or metabolic abnormalities in the brain or neurological system can
157 Neurological disorders primarily affect and impair the functioning of the brain and/or neurological system. Structural, electrical or metabolic abnormalities in the brain or neurological system can
General introduction
 General introduction P. N. Thompson 1 1 Epidemiology Section, Department of Production Animal Studies, Faculty of Veterinary Science, University of Pretoria, Onderstepoort, South Africa 7 8 Introduction
General introduction P. N. Thompson 1 1 Epidemiology Section, Department of Production Animal Studies, Faculty of Veterinary Science, University of Pretoria, Onderstepoort, South Africa 7 8 Introduction
CLINICAL PRESENTATION
 MYASTHENIA GRAVIS INTRODUCTION Most common primary disorder of neuromuscular transmission Usually due to acquired immunological abnormality Also due to genetic abnormalities at neuromuscular junction.
MYASTHENIA GRAVIS INTRODUCTION Most common primary disorder of neuromuscular transmission Usually due to acquired immunological abnormality Also due to genetic abnormalities at neuromuscular junction.
What effect would an AChE inhibitor have at the neuromuscular junction?
 CASE 4 A 32-year-old woman presents to her primary care physician s office with difficulty chewing food. She states that when she eats certain foods that require a significant amount of chewing (meat),
CASE 4 A 32-year-old woman presents to her primary care physician s office with difficulty chewing food. She states that when she eats certain foods that require a significant amount of chewing (meat),
MYASTHENIA GRAVIS. Mr. D.Raju, M.pharm, Lecturer
 MYASTHENIA GRAVIS Mr. D.Raju, M.pharm, Lecturer OUTLINE Background Anatomy Pathophysiology Clinical Presentation Treatment BACKGROUND Acquired autoimmune disorder Clinically characterized by: Weakness
MYASTHENIA GRAVIS Mr. D.Raju, M.pharm, Lecturer OUTLINE Background Anatomy Pathophysiology Clinical Presentation Treatment BACKGROUND Acquired autoimmune disorder Clinically characterized by: Weakness
Homeostatic regulation of synaptic strength and the safety factor for neuromuscular transmission
 The Life Cycle of Neuromuscular Synapses Homeostatic regulation of synaptic strength and the safety factor for neuromuscular transmission 1. Synaptic transmission, safety factor and sizestrength relationships
The Life Cycle of Neuromuscular Synapses Homeostatic regulation of synaptic strength and the safety factor for neuromuscular transmission 1. Synaptic transmission, safety factor and sizestrength relationships
Autonomic Nervous System. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
Clinical and pathologic aspects of congenital myopathies
 Neurol J Southeast Asia 2001; 6 : 99 106 88 Clinical and pathologic aspects of congenital myopathies Ikuya NONAKA MD National Center of Neurology and Psychiatry, Kodaira, Tokyo, Japan Abstract The term
Neurol J Southeast Asia 2001; 6 : 99 106 88 Clinical and pathologic aspects of congenital myopathies Ikuya NONAKA MD National Center of Neurology and Psychiatry, Kodaira, Tokyo, Japan Abstract The term
Critical Illness Polyneuropathy CIP and Critical Illness Myopathy CIM. Andrzej Sladkowski
 Critical Illness Polyneuropathy CIP and Critical Illness Myopathy CIM Andrzej Sladkowski Potential causes of weakness in the ICU-1 Muscle disease Critical illness myopathy Inflammatory myopathy Hypokalemic
Critical Illness Polyneuropathy CIP and Critical Illness Myopathy CIM Andrzej Sladkowski Potential causes of weakness in the ICU-1 Muscle disease Critical illness myopathy Inflammatory myopathy Hypokalemic
Activity Dependent Changes At the Developing Neuromuscular Junction
 Activity Dependent Changes At the Developing Neuromuscular Junction (slides 16, 17 and 18 have been slightly modified for clarity) MCP Lecture 2-3 9.013/7.68 04 Neuromuscular Junction Development 1. Muscle
Activity Dependent Changes At the Developing Neuromuscular Junction (slides 16, 17 and 18 have been slightly modified for clarity) MCP Lecture 2-3 9.013/7.68 04 Neuromuscular Junction Development 1. Muscle
Skeletal Muscle and the Molecular Basis of Contraction. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
NEUROMUSCULAR BLOCKING AGENTS
 NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
Synaptic communication
 Synaptic communication Objectives: after these lectures you should be able to: - explain the differences between an electrical and chemical synapse - describe the steps involved in synaptic communication
Synaptic communication Objectives: after these lectures you should be able to: - explain the differences between an electrical and chemical synapse - describe the steps involved in synaptic communication
Cellular Bioelectricity
 ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture 22 Friday, February 28, 2014 10. THE NEUROMUSCULAR JUNCTION We will look at: Structure of the neuromuscular junction Evidence for the quantal nature
ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture 22 Friday, February 28, 2014 10. THE NEUROMUSCULAR JUNCTION We will look at: Structure of the neuromuscular junction Evidence for the quantal nature
Myasthenia Gravis. Mike Gilchrist 10/30/06
 Myasthenia Gravis Mike Gilchrist 10/30/06 Overview Background Pathogenesis Clinical Manifestations Diagnosis Treatment Associated Conditions Background Severe muscle disease Most common disorder of neuromuscular
Myasthenia Gravis Mike Gilchrist 10/30/06 Overview Background Pathogenesis Clinical Manifestations Diagnosis Treatment Associated Conditions Background Severe muscle disease Most common disorder of neuromuscular
EDX in Myopathies Limitations. EDX in Myopathies Utility Causes of Myopathy. Myopathy: Issues for Electromyographers
 Electrodiagnostic Assessment of Myopathy Myopathy: Issues for Electromyographers Often perceived as challenging Ian Grant Division of Neurology QEII Health Sciences Centre Halifax NS CNSF EMG Course June
Electrodiagnostic Assessment of Myopathy Myopathy: Issues for Electromyographers Often perceived as challenging Ian Grant Division of Neurology QEII Health Sciences Centre Halifax NS CNSF EMG Course June
TREAT-NMD Care and Trial Sites Registry Information Chart
 TREAT-NMD Care and Trial Sites Registry Information Chart Below you find a list of the information that is asked in the CTSR of all registered neuromuscular disease sites. For more information please visit
TREAT-NMD Care and Trial Sites Registry Information Chart Below you find a list of the information that is asked in the CTSR of all registered neuromuscular disease sites. For more information please visit
Primary Periodic Paralysis. A Complex Disorder. A Challenging Diagnosis.
 Primary Periodic Paralysis A Complex Disorder. A Challenging Diagnosis. A Complex Disorder A Group of Rare Channelopathies With Varying Subtypes and Triggers Primary periodic paralysis causes recurrent,
Primary Periodic Paralysis A Complex Disorder. A Challenging Diagnosis. A Complex Disorder A Group of Rare Channelopathies With Varying Subtypes and Triggers Primary periodic paralysis causes recurrent,
1/28/2019. OSF HealthCare INI Care Center Team. Neuromuscular Disease: Muscular Dystrophy. OSF HealthCare INI Care Center Team: Who are we?
 Neuromuscular Disease: Muscular Dystrophy Muscular Dystrophy Association (MDA) and OSF HealthCare Illinois Neurological Institute (INI) Care Center Team The Neuromuscular clinic is a designated MDA Care
Neuromuscular Disease: Muscular Dystrophy Muscular Dystrophy Association (MDA) and OSF HealthCare Illinois Neurological Institute (INI) Care Center Team The Neuromuscular clinic is a designated MDA Care
Muscular Dystrophy. Biol 405 Molecular Medicine
 Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
Guide to the use of nerve conduction studies (NCS) & electromyography (EMG) for non-neurologists
 Guide to the use of nerve conduction studies (NCS) & electromyography (EMG) for non-neurologists What is NCS/EMG? NCS examines the conduction properties of sensory and motor peripheral nerves. For both
Guide to the use of nerve conduction studies (NCS) & electromyography (EMG) for non-neurologists What is NCS/EMG? NCS examines the conduction properties of sensory and motor peripheral nerves. For both
Muscle and Neuromuscular Junction. Peter Takizawa Department of Cell Biology
 Muscle and Neuromuscular Junction Peter Takizawa Department of Cell Biology Types and structure of muscle cells Structural basis of contraction Triggering muscle contraction Skeletal muscle consists of
Muscle and Neuromuscular Junction Peter Takizawa Department of Cell Biology Types and structure of muscle cells Structural basis of contraction Triggering muscle contraction Skeletal muscle consists of
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/20412 holds various files of this Leiden University dissertation. Author: Niks, E.H. Title: Myasthenia gravis with antibodies to muscle-specific kinase
Cover Page The handle http://hdl.handle.net/1887/20412 holds various files of this Leiden University dissertation. Author: Niks, E.H. Title: Myasthenia gravis with antibodies to muscle-specific kinase
A very simplified introduction to neuromuscular illnesses (NMIs) with a particular focus on the muscular dystrophies.
 1 A very simplified introduction to neuromuscular illnesses (NMIs) with a particular focus on the muscular dystrophies. Available on-line at: Bill Tillier Calgary Alberta September, 2008. Terminology.
1 A very simplified introduction to neuromuscular illnesses (NMIs) with a particular focus on the muscular dystrophies. Available on-line at: Bill Tillier Calgary Alberta September, 2008. Terminology.
III./10.4. Diagnosis. Introduction. A.) Laboratory tests. Laboratory tests, electrophysiology, muscle biopsy, genetic testing, imaging techniques
 III./10.4. Diagnosis Laboratory tests, electrophysiology, muscle biopsy, genetic testing, imaging techniques After studying this chapter, you will become familiar with the most commonly used diagnostic
III./10.4. Diagnosis Laboratory tests, electrophysiology, muscle biopsy, genetic testing, imaging techniques After studying this chapter, you will become familiar with the most commonly used diagnostic
Synthesis. Storage. Physiology and Pathophysiology of Neuromuscular Transmission. Release. Action. Inactivation. Myasthenia Gravis Before
 Synthesis Physiology and Pathophysiology of Neuromuscular Transmission Storage Release Action Inactivation Myasthenia gravis and LEMS are autoimmune diseases Myasthenia Gravis Before LEMS: Ca channel antibodies
Synthesis Physiology and Pathophysiology of Neuromuscular Transmission Storage Release Action Inactivation Myasthenia gravis and LEMS are autoimmune diseases Myasthenia Gravis Before LEMS: Ca channel antibodies
The Floppy Baby. Clare Betteridge
 The Floppy Baby Clare Betteridge The floppy baby Identification Evaluation Investigation Diagnosis Examples What is a floppy baby? Elbows and knees loosely extended. Head control is usually poor or absent.
The Floppy Baby Clare Betteridge The floppy baby Identification Evaluation Investigation Diagnosis Examples What is a floppy baby? Elbows and knees loosely extended. Head control is usually poor or absent.
Muscle Physiology. Dr. Ebneshahidi Ebneshahidi
 Muscle Physiology Dr. Ebneshahidi Skeletal Muscle Figure 9.2 (a) Functions of the muscular system 1. Locomotion body movements are due to skeletal muscle contraction. 2. Vasoconstriction and vasodilatation
Muscle Physiology Dr. Ebneshahidi Skeletal Muscle Figure 9.2 (a) Functions of the muscular system 1. Locomotion body movements are due to skeletal muscle contraction. 2. Vasoconstriction and vasodilatation
Myasthenia gravis. Page 1 of 7
 Myasthenia gravis What is myasthenia gravis? Myasthenia gravis (sometimes abbreviated to MG) is a chronic, autoimmune condition that causes muscle weakness and excessive muscle fatigue. It is rare, affecting
Myasthenia gravis What is myasthenia gravis? Myasthenia gravis (sometimes abbreviated to MG) is a chronic, autoimmune condition that causes muscle weakness and excessive muscle fatigue. It is rare, affecting
PSK4U THE NEUROMUSCULAR SYSTEM
 PSK4U THE NEUROMUSCULAR SYSTEM REVIEW Review of muscle so we can see how the neuromuscular system works This is not on today's note Skeletal Muscle Cell: Cellular System A) Excitation System Electrical
PSK4U THE NEUROMUSCULAR SYSTEM REVIEW Review of muscle so we can see how the neuromuscular system works This is not on today's note Skeletal Muscle Cell: Cellular System A) Excitation System Electrical
Index. Phys Med Rehabil Clin N Am 14 (2003) Note: Page numbers of article titles are in boldface type.
 Phys Med Rehabil Clin N Am 14 (2003) 445 453 Index Note: Page numbers of article titles are in boldface type. A Acid maltase deficiencies, electrodiagnosis of, 420, 422 Acquired peripheral neuropathy,
Phys Med Rehabil Clin N Am 14 (2003) 445 453 Index Note: Page numbers of article titles are in boldface type. A Acid maltase deficiencies, electrodiagnosis of, 420, 422 Acquired peripheral neuropathy,
Outline. Neuron Structure. Week 4 - Nervous System. The Nervous System: Neurons and Synapses
 Outline Week 4 - The Nervous System: Neurons and Synapses Neurons Neuron structures Types of neurons Electrical activity of neurons Depolarization, repolarization, hyperpolarization Synapses Release of
Outline Week 4 - The Nervous System: Neurons and Synapses Neurons Neuron structures Types of neurons Electrical activity of neurons Depolarization, repolarization, hyperpolarization Synapses Release of
Chapter 10: Muscles. Vocabulary: aponeurosis, fatigue
 Chapter 10: Muscles 37. Describe the structural components of skeletal muscle tissue from the molecular to the organ level. 38. Describe the structure, function, and importance of sarcomeres. 39. Identify
Chapter 10: Muscles 37. Describe the structural components of skeletal muscle tissue from the molecular to the organ level. 38. Describe the structure, function, and importance of sarcomeres. 39. Identify
NERVOUS SYSTEM NERVOUS SYSTEM. Somatic nervous system. Brain Spinal Cord Autonomic nervous system. Sympathetic nervous system
 SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
Muscular Dystrophies. Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018
 Muscular Dystrophies Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018 Definition and classification Clinical guide to recognize muscular
Muscular Dystrophies Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018 Definition and classification Clinical guide to recognize muscular
A NEW STRATEGY TO ESCAPE FROM EXPRESSION LINES V.0
 A NEW STRATEGY TO ESCAPE FROM EXPRESSION LINES V.0 WRINKLES: THE HALLMARK OF AGING The face is the most visible part of our body and increased wrinkling is one of the most striking signs of aging Repeated
A NEW STRATEGY TO ESCAPE FROM EXPRESSION LINES V.0 WRINKLES: THE HALLMARK OF AGING The face is the most visible part of our body and increased wrinkling is one of the most striking signs of aging Repeated
Myasthenia Gravis and Lambert-Eaton Myasthenic Syndrome Michael W. Nicolle, MD
 Review Article Downloaded from https://journals.lww.com/continuum by maxwo3znzwrcfjddvmduzvysskax4mzb8eymgwvspgpjoz9l+mqfwgfuplwvy+jmyqlpqmifewtrhxj7jpeo+505hdqh14pdzv4lwky42mcrzqckilw0d1o4yvrwmuvvhuyo4rrbviuuwr5dqytbtk/icsrdbt0hfryk7+zagvaltkgnudxdohhaxffu/7kno26hifzu/+bcy16w7w1bdw==
Review Article Downloaded from https://journals.lww.com/continuum by maxwo3znzwrcfjddvmduzvysskax4mzb8eymgwvspgpjoz9l+mqfwgfuplwvy+jmyqlpqmifewtrhxj7jpeo+505hdqh14pdzv4lwky42mcrzqckilw0d1o4yvrwmuvvhuyo4rrbviuuwr5dqytbtk/icsrdbt0hfryk7+zagvaltkgnudxdohhaxffu/7kno26hifzu/+bcy16w7w1bdw==
BIPN140 Lecture 13: Synapse Formation (Synaptogenesis)
 BIPN140 Lecture 13: Synapse Formation (Synaptogenesis) 1. Neuromuscular Junction (NMJ) Development 2. Synaptogenesis at Central Synapses Su (FA16) Ultrastructural Image of an NMJ Active Zone Basal Lamina
BIPN140 Lecture 13: Synapse Formation (Synaptogenesis) 1. Neuromuscular Junction (NMJ) Development 2. Synaptogenesis at Central Synapses Su (FA16) Ultrastructural Image of an NMJ Active Zone Basal Lamina
ME Farrugia Consultant Neurologist, Institute of Neurological Sciences, Southern General Hospital, Glasgow, UK
 CME doi:10.4997/jrcpe.2011.111 2011 Royal College of Physicians of Edinburgh Myasthenic syndromes Consultant Neurologist, Institute of Neurological Sciences, Southern General Hospital, Glasgow, UK ABSTRACT
CME doi:10.4997/jrcpe.2011.111 2011 Royal College of Physicians of Edinburgh Myasthenic syndromes Consultant Neurologist, Institute of Neurological Sciences, Southern General Hospital, Glasgow, UK ABSTRACT
Muscle Physiology. Introduction. Four Characteristics of Muscle tissue. Skeletal Muscle
 Muscle Physiology Introduction Muscle = tissue capable of forceful shortening or contraction Converts chemical energy (ATP) into mechanical energy Important in: Respiration Urine collection & flow Gastrointestinal
Muscle Physiology Introduction Muscle = tissue capable of forceful shortening or contraction Converts chemical energy (ATP) into mechanical energy Important in: Respiration Urine collection & flow Gastrointestinal
Muscular System. Disorders & Conditions
 Muscular System Disorders & Conditions Fibromyalgia Fibromyalgia is a disorder characterized by widespread musculoskeletal pain accompanied by fatigue, sleep, memory and mood issues. Often is described
Muscular System Disorders & Conditions Fibromyalgia Fibromyalgia is a disorder characterized by widespread musculoskeletal pain accompanied by fatigue, sleep, memory and mood issues. Often is described
Index. Note: Page numbers of article titles are in boldface type.
 Neurol Clin N Am 20 (2002) 605 617 Index Note: Page numbers of article titles are in boldface type. A ALS. See Amyotrophic lateral sclerosis (ALS) Amyotrophic lateral sclerosis (ALS) active denervation
Neurol Clin N Am 20 (2002) 605 617 Index Note: Page numbers of article titles are in boldface type. A ALS. See Amyotrophic lateral sclerosis (ALS) Amyotrophic lateral sclerosis (ALS) active denervation
1GENERAL INFORMATION
 1 GENERAL INFORMATION General information 11 1.1 Diagnosis Some children will have a diagnosis of their muscle-wasting condition when they begin their school life. Others will still be having investigations
1 GENERAL INFORMATION General information 11 1.1 Diagnosis Some children will have a diagnosis of their muscle-wasting condition when they begin their school life. Others will still be having investigations
1/4/2017. Introduction. Connective Tissue Coverings. 9.1: Structure of a Skeletal Muscle. Skeletal Muscle Fibers. Connective Tissue Coverings
 Introduction Chapter 09 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright McGraw-Hill Education. Permission required for reproduction
Introduction Chapter 09 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright McGraw-Hill Education. Permission required for reproduction
Electrodiagnosis of Neuromuscular Junction Disorders
 NMT overview Electrodiagnosis of Neuromuscular Junction Disorders Motor NAP arrives at nerve terminal Voltage-gated calcium channels open Ca +2 moves into presynaptic nerve terminal SNARE proteins elicit
NMT overview Electrodiagnosis of Neuromuscular Junction Disorders Motor NAP arrives at nerve terminal Voltage-gated calcium channels open Ca +2 moves into presynaptic nerve terminal SNARE proteins elicit
PRIMARY DISEASES OF MYELIN. By: Shifaa Al Qa qa
 PRIMARY DISEASES OF MYELIN By: Shifaa Al Qa qa Most diseases of myelin are primarily white matter disorders??? Myelinated axons most diseases of CNS myelin do not involve the peripheral nerves to any significant
PRIMARY DISEASES OF MYELIN By: Shifaa Al Qa qa Most diseases of myelin are primarily white matter disorders??? Myelinated axons most diseases of CNS myelin do not involve the peripheral nerves to any significant
Neurons Chapter 7 2/19/2016. Learning Objectives. Cells of the Nervous System. Cells of the Nervous System. Cells of the Nervous System
 Learning Objectives Neurons Chapter 7 Identify and describe the functions of the two main divisions of the nervous system. Differentiate between a neuron and neuroglial cells in terms of structure and
Learning Objectives Neurons Chapter 7 Identify and describe the functions of the two main divisions of the nervous system. Differentiate between a neuron and neuroglial cells in terms of structure and
Chapter 10 Muscle Tissue Lecture Outline
 Chapter 10 Muscle Tissue Lecture Outline Muscle tissue types 1. Skeletal muscle = voluntary striated 2. Cardiac muscle = involuntary striated 3. Smooth muscle = involuntary nonstriated Characteristics
Chapter 10 Muscle Tissue Lecture Outline Muscle tissue types 1. Skeletal muscle = voluntary striated 2. Cardiac muscle = involuntary striated 3. Smooth muscle = involuntary nonstriated Characteristics
Myasthenia Gravis What is Myasthenia Gravis? Who is at risk of developing MG? Is MG hereditary? What are the symptoms of MG? What causes MG?
 Myasthenia Gravis What is Myasthenia Gravis? Myasthenia Gravis (MG) is a chronic, autoimmune disease that causes muscle weakness and excessive muscle fatigue. It is uncommon, affecting about 15 in every
Myasthenia Gravis What is Myasthenia Gravis? Myasthenia Gravis (MG) is a chronic, autoimmune disease that causes muscle weakness and excessive muscle fatigue. It is uncommon, affecting about 15 in every
Introduction to Neurobiology
 Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Clinical Summaries. CLN1 Disease, infantile onset and others
 Clinical Summaries CLN1 Disease, infantile onset and others The gene called CLN1 lies on chromosome 1. CLN1 disease is inherited as an autosomal recessive disorder, which means that both chromosomes carry
Clinical Summaries CLN1 Disease, infantile onset and others The gene called CLN1 lies on chromosome 1. CLN1 disease is inherited as an autosomal recessive disorder, which means that both chromosomes carry
Chapter 8 Notes. Muscles
 Chapter 8 Notes Muscles 8.1 Intro Three muscle types Skeletal Smooth cardiac 8.2 Structure of Skeletal Muscle Composition Skeletal muscle tissue Nervous tissue Blood Connective tissue Connective tissue
Chapter 8 Notes Muscles 8.1 Intro Three muscle types Skeletal Smooth cardiac 8.2 Structure of Skeletal Muscle Composition Skeletal muscle tissue Nervous tissue Blood Connective tissue Connective tissue
Types of Muscle. Skeletal striated & voluntary Smooth involuntary Cardiac - heart
 Muscular System Types of Muscle Skeletal striated & voluntary Smooth involuntary Cardiac - heart The word striated means striped. Skeletal muscle appears striped under a microscope. Muscles and Muscle
Muscular System Types of Muscle Skeletal striated & voluntary Smooth involuntary Cardiac - heart The word striated means striped. Skeletal muscle appears striped under a microscope. Muscles and Muscle
THIAMINE TRANSPORTER TYPE 2 DEFICIENCY
 THIAMINE TRANSPORTER TYPE 2 DEFICIENCY WHAT IS THE THIAMINE TRANSPORTER TYPE 2 DEFICIENCY (hthtr2)? The thiamine transporter type 2 deficiency (hthtr2) is a inborn error of thiamine metabolism caused by
THIAMINE TRANSPORTER TYPE 2 DEFICIENCY WHAT IS THE THIAMINE TRANSPORTER TYPE 2 DEFICIENCY (hthtr2)? The thiamine transporter type 2 deficiency (hthtr2) is a inborn error of thiamine metabolism caused by
Recent Advances in Neurology 2014: Neuromuscular Case Presentations
 Recent Advances in Neurology 2014: Neuromuscular Case Presentations Jeffrey W. Ralph, MD Associate Clinical Professor Patient #1: Young woman with severe polyneuropathy 25 year-old woman Normal motor and
Recent Advances in Neurology 2014: Neuromuscular Case Presentations Jeffrey W. Ralph, MD Associate Clinical Professor Patient #1: Young woman with severe polyneuropathy 25 year-old woman Normal motor and
NEUROCHEMISTRY Brief Review
 NEUROCHEMISTRY Brief Review UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY PBL MBBS YEAR V SEMINAR VJ Temple 1 Membrane potential Membrane potential:
NEUROCHEMISTRY Brief Review UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY PBL MBBS YEAR V SEMINAR VJ Temple 1 Membrane potential Membrane potential:
Lumbosacral plexus lesion Lumbosacral plexus disorders G54.1 Neuralgic amyotrophy Neuralgic amyotrophy G
 ICD-9-CM and ICD-10-CM NEUROMUSCULAR DIAGNOSIS CODES Focal Neuropathy ICD-9-CM ICD-10-CM Mononeuropathy G56.00 Carpal tunnel syndrome 354.00 Other median nerve lesion 354.10 Lesion of ulnar nerve 354.20
ICD-9-CM and ICD-10-CM NEUROMUSCULAR DIAGNOSIS CODES Focal Neuropathy ICD-9-CM ICD-10-CM Mononeuropathy G56.00 Carpal tunnel syndrome 354.00 Other median nerve lesion 354.10 Lesion of ulnar nerve 354.20
Anatomy and Physiology 1 Chapter 10 self quiz Pro, Dima Darwish,MD.
 Anatomy and Physiology 1 Chapter 10 self quiz Pro, Dima Darwish,MD. 1) Which of the following is a recognized function of skeletal muscle? A) produce movement B) maintain posture C) maintain body temperature
Anatomy and Physiology 1 Chapter 10 self quiz Pro, Dima Darwish,MD. 1) Which of the following is a recognized function of skeletal muscle? A) produce movement B) maintain posture C) maintain body temperature
Muscle Pathology Surgical Pathology Unknown Conference. November, 2008 Philip Boyer, M.D., Ph.D.
 Muscle Pathology Surgical Pathology Unknown Conference November, 2008 Philip Boyer, M.D., Ph.D. Etiologic Approach to Differential Diagnosis Symptoms / Signs / Imaging / Biopsy / CSF Analysis Normal Abnormal
Muscle Pathology Surgical Pathology Unknown Conference November, 2008 Philip Boyer, M.D., Ph.D. Etiologic Approach to Differential Diagnosis Symptoms / Signs / Imaging / Biopsy / CSF Analysis Normal Abnormal
Connective tissue MUSCLE TISSUE
 Connective tissue MUSCLE TISSUE Part 1 General features of MT Develop from mesoderm Many cells, less intercellular matrix Function contraction (shortening) Skeletal (striated, voluntary) Types of MT Cardiac
Connective tissue MUSCLE TISSUE Part 1 General features of MT Develop from mesoderm Many cells, less intercellular matrix Function contraction (shortening) Skeletal (striated, voluntary) Types of MT Cardiac
Nerve Muscle Relationship and Neural Muscular Junction Quiz. Remember, you need to know the structure and the function!
 Nerve Muscle Relationship and Neural Muscular Junction Quiz Remember, you need to know the structure and the function! What is this called? What is this? Schwann cell What is this called? Basal lamina
Nerve Muscle Relationship and Neural Muscular Junction Quiz Remember, you need to know the structure and the function! What is this called? What is this? Schwann cell What is this called? Basal lamina
Alterations in Synaptic Strength Preceding Axon Withdrawal
 Alterations in Synaptic Strength Preceding Axon Withdrawal H. Colman, J. Nabekura, J.W. Lichtman presented by Ana Fiallos Synaptic Transmission at the Neuromuscular Junction Motor neurons with cell bodies
Alterations in Synaptic Strength Preceding Axon Withdrawal H. Colman, J. Nabekura, J.W. Lichtman presented by Ana Fiallos Synaptic Transmission at the Neuromuscular Junction Motor neurons with cell bodies
Drugs Affecting the Autonomic Nervous System-1. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Drugs Affecting the Autonomic Nervous System-1 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia The autonomic nervous system, along with the endocrine system,
Drugs Affecting the Autonomic Nervous System-1 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia The autonomic nervous system, along with the endocrine system,
Congenital myopathies: clinical phenotypes and new diagnostic tools
 Cassandrini et al. Italian Journal of Pediatrics (2017) 43:101 DOI 10.1186/s13052-017-0419-z REVIEW Congenital myopathies: clinical phenotypes and new diagnostic tools Open Access Denise Cassandrini 1,
Cassandrini et al. Italian Journal of Pediatrics (2017) 43:101 DOI 10.1186/s13052-017-0419-z REVIEW Congenital myopathies: clinical phenotypes and new diagnostic tools Open Access Denise Cassandrini 1,
Portions from Chapter 6 CHAPTER 7. The Nervous System: Neurons and Synapses. Chapter 7 Outline. and Supporting Cells
 CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
Spinal Muscular Atrophy: Case Study. Spinal muscular atrophy (SMA) is a fairly common genetic disorder, affecting
 Spinal Muscular Atrophy: Case Study Spinal muscular atrophy (SMA) is a fairly common genetic disorder, affecting approximately one in 6,000 babies. It is estimated that one in every 40 Americans carries
Spinal Muscular Atrophy: Case Study Spinal muscular atrophy (SMA) is a fairly common genetic disorder, affecting approximately one in 6,000 babies. It is estimated that one in every 40 Americans carries
Case 1 A 65 year old college professor came to the neurology clinic referred by her family physician because of frequent falling. She had a history of
 Peripheral Nervous System Case 1 A 65 year old college professor came to the neurology clinic referred by her family physician because of frequent falling. She had a history of non-insulin dependent diabetes
Peripheral Nervous System Case 1 A 65 year old college professor came to the neurology clinic referred by her family physician because of frequent falling. She had a history of non-insulin dependent diabetes
Effects of Temperature on Neuromuscular Function. Jon Marsden School of Health Professions University of Plymouth
 Effects of Temperature on Neuromuscular Function Jon Marsden School of Health Professions University of Plymouth http://www.visualphotos.com/image/2x2696104/test_tube_over_a_bunsen_burner http://www.doc.ic.ac.uk/~nd/surprise_95/journal/vol4/ykl/report.html
Effects of Temperature on Neuromuscular Function Jon Marsden School of Health Professions University of Plymouth http://www.visualphotos.com/image/2x2696104/test_tube_over_a_bunsen_burner http://www.doc.ic.ac.uk/~nd/surprise_95/journal/vol4/ykl/report.html
Warm Up! Test review (already! ;))
 Warm Up! Test review (already! ;)) Write a question you might find on the Unit 5 test next week! (Multiple choice, matching, fill in, or short answer!) - challenge yourself and be ready to share!!! PowerPoint
Warm Up! Test review (already! ;)) Write a question you might find on the Unit 5 test next week! (Multiple choice, matching, fill in, or short answer!) - challenge yourself and be ready to share!!! PowerPoint
Essentials of Human Anatomy & Physiology. The Muscular System
 Essentials of Human Anatomy & Physiology The Muscular System The Muscular System Muscles are responsible for all types of body movement they contract or shorten and are the machine of the body Three basic
Essentials of Human Anatomy & Physiology The Muscular System The Muscular System Muscles are responsible for all types of body movement they contract or shorten and are the machine of the body Three basic
CLASS SET Unit 4: The Muscular System STUDY GUIDE
 NPHS Anatomy & Physiology Questions to answer: 1) List three functions of the muscular system. 1) movement 2) thermogenesis (generates heat) 3) posture & body/joint support CLASS SET Unit 4: The Muscular
NPHS Anatomy & Physiology Questions to answer: 1) List three functions of the muscular system. 1) movement 2) thermogenesis (generates heat) 3) posture & body/joint support CLASS SET Unit 4: The Muscular
Index. derm.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adhesion and migration, the diverse functions of the laminin a3 subunit, 79 87 Alopecia in epidermolysis bullosa, 165 169 Amblyopia and inherited
Note: Page numbers of article titles are in boldface type. A Adhesion and migration, the diverse functions of the laminin a3 subunit, 79 87 Alopecia in epidermolysis bullosa, 165 169 Amblyopia and inherited
Neurological channelopathies: new insights into disease mechanisms and ion channel function
 Neurologicalchannelopathies:newinsightsintodiseasemechanismsand ionchannelfunction DimitriMKullmann 1 andstephengwaxman 2 1 InstituteofNeurology,UniversityCollegeLondon,London,UK 2 DepartmentofNeurology,YaleUniversitySchoolofMedicine,NewHaven,
Neurologicalchannelopathies:newinsightsintodiseasemechanismsand ionchannelfunction DimitriMKullmann 1 andstephengwaxman 2 1 InstituteofNeurology,UniversityCollegeLondon,London,UK 2 DepartmentofNeurology,YaleUniversitySchoolofMedicine,NewHaven,
Genetics of Inclusion Body Myositis
 Genetics of Inclusion Body Myositis Thomas Lloyd, MD, PhD Associate Professor of Neurology and Neuroscience Co-director, Johns Hopkins Myositis Center Sporadic IBM (IBM) Age at onset usually > 50 Prevalence
Genetics of Inclusion Body Myositis Thomas Lloyd, MD, PhD Associate Professor of Neurology and Neuroscience Co-director, Johns Hopkins Myositis Center Sporadic IBM (IBM) Age at onset usually > 50 Prevalence
REPORT Mutation Analysis of CHRNA1, CHRNB1, CHRND, and RAPSN Genes in Multiple Pterygium Syndrome/Fetal Akinesia Patients
 REPORT Mutation Analysis of CHRNA1, CHRNB1, CHRND, and RAPSN Genes in Multiple Pterygium Syndrome/Fetal Akinesia Patients Julie Vogt, 1,2 Benjamin J. Harrison, 1 Hayley Spearman, 3 Judy Cossins, 3 Sascha
REPORT Mutation Analysis of CHRNA1, CHRNB1, CHRND, and RAPSN Genes in Multiple Pterygium Syndrome/Fetal Akinesia Patients Julie Vogt, 1,2 Benjamin J. Harrison, 1 Hayley Spearman, 3 Judy Cossins, 3 Sascha
Genetics of Hereditary Spastic Paraplegia Dr. Arianna Tucci
 Genetics of Hereditary Spastic Paraplegia 1 Clinical Research Fellow Institute of Neurology University College London Hereditary spastic paraplegia: definition Clinical designation for neurologic syndromes
Genetics of Hereditary Spastic Paraplegia 1 Clinical Research Fellow Institute of Neurology University College London Hereditary spastic paraplegia: definition Clinical designation for neurologic syndromes
Epileptogenesis: A Clinician s Perspective
 Epileptogenesis: A Clinician s Perspective Samuel F Berkovic Epilepsy Research Centre, University of Melbourne Austin Health Epileptogenesis The process of development and sustaining the propensity to
Epileptogenesis: A Clinician s Perspective Samuel F Berkovic Epilepsy Research Centre, University of Melbourne Austin Health Epileptogenesis The process of development and sustaining the propensity to
